Patents
Literature
157 results about "Autologous blood" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
Intravascular methods and apparatus for isolation and selective cooling of the cerebral vasculature during surgical procedures
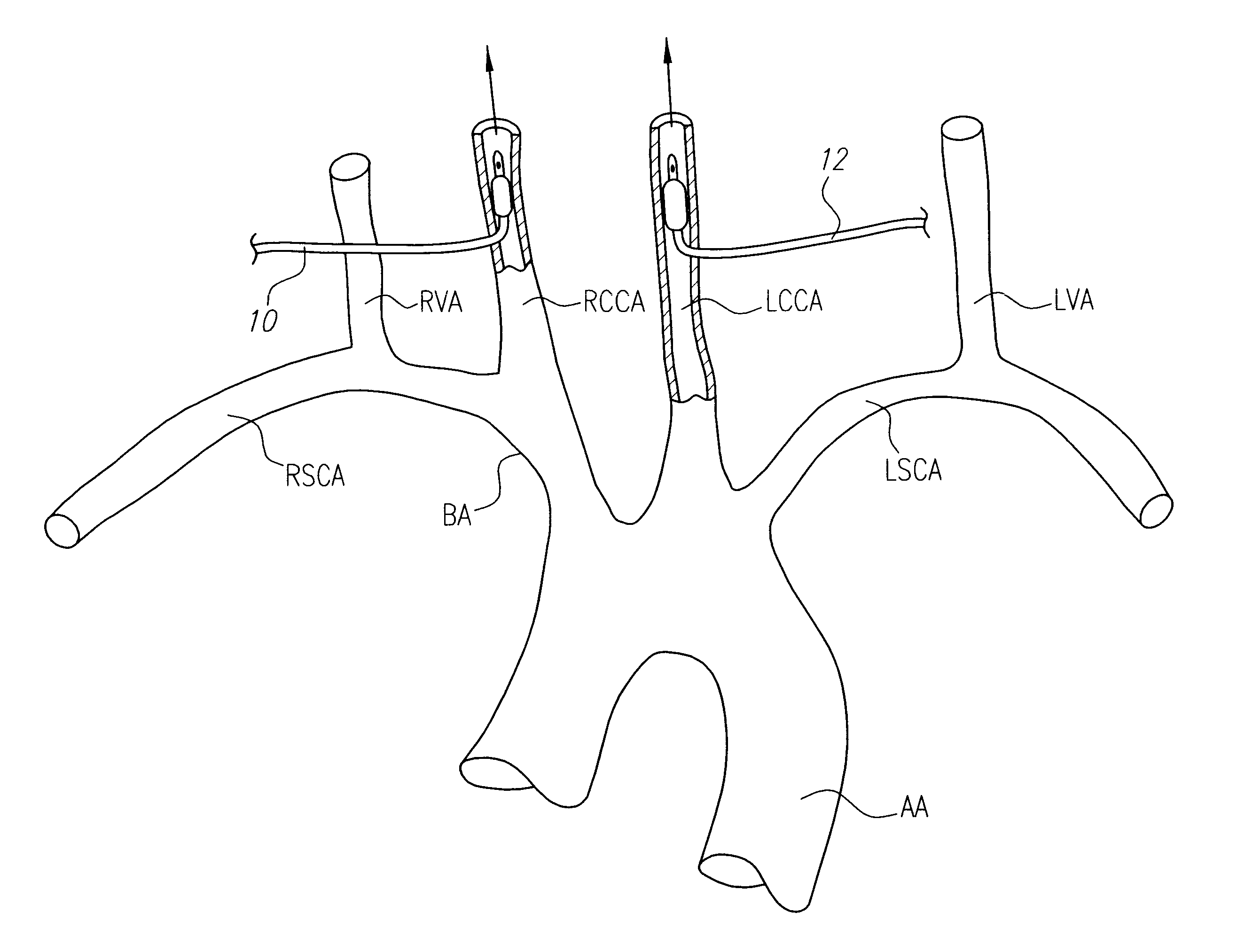
InactiveUS6555057B1Reduce riskIncrease perfusionOther blood circulation devicesMedical devicesRisk strokeSurgical department
Patients having diminished circulation in the cerebral vasculature as a result of stroke or from other causes such as cardiac arrest, shock or head trauma, or aneurysm surgery or aortic surgery, are treated by flowing an oxygenated medium through an arterial access site into the cerebral vasculature and collecting the medium through an access site in the venous site of the cerebral vasculature. Usually, the cold oxygenated medium will comprise autologous blood, and the blood will be recirculated for a time sufficient to permit treatment of the underlying cause of diminished circulation. In addition to oxygenation, the recirculating blood will also be cooled to hypothermically treat and preserve brain tissue. Isolation and cooling of cerebral vasculature in patients undergoing aortic and other procedures is achieved by internally occluding at least the right common carotid artery above the aortic arch. Blood or other oxygenated medium is perfused through the occluded common carotid artery(ies) and into the arterial cerebral vasculature. Usually, oxygen depleted blood or other medium leaving the cerebral vasculature is collected, oxygenated, and cooled in an extracorporeal circuit so that it may be returned to the patient. Occlusion of the carotid artery(ies) is preferably accomplished using expansible occluders, such as balloon-tipped cannula, catheters, or similar access devices. Access to the occlusion site(s) may be open surgical, percutaneous, or intravascular.
Owner:BARBUT DENISE +3
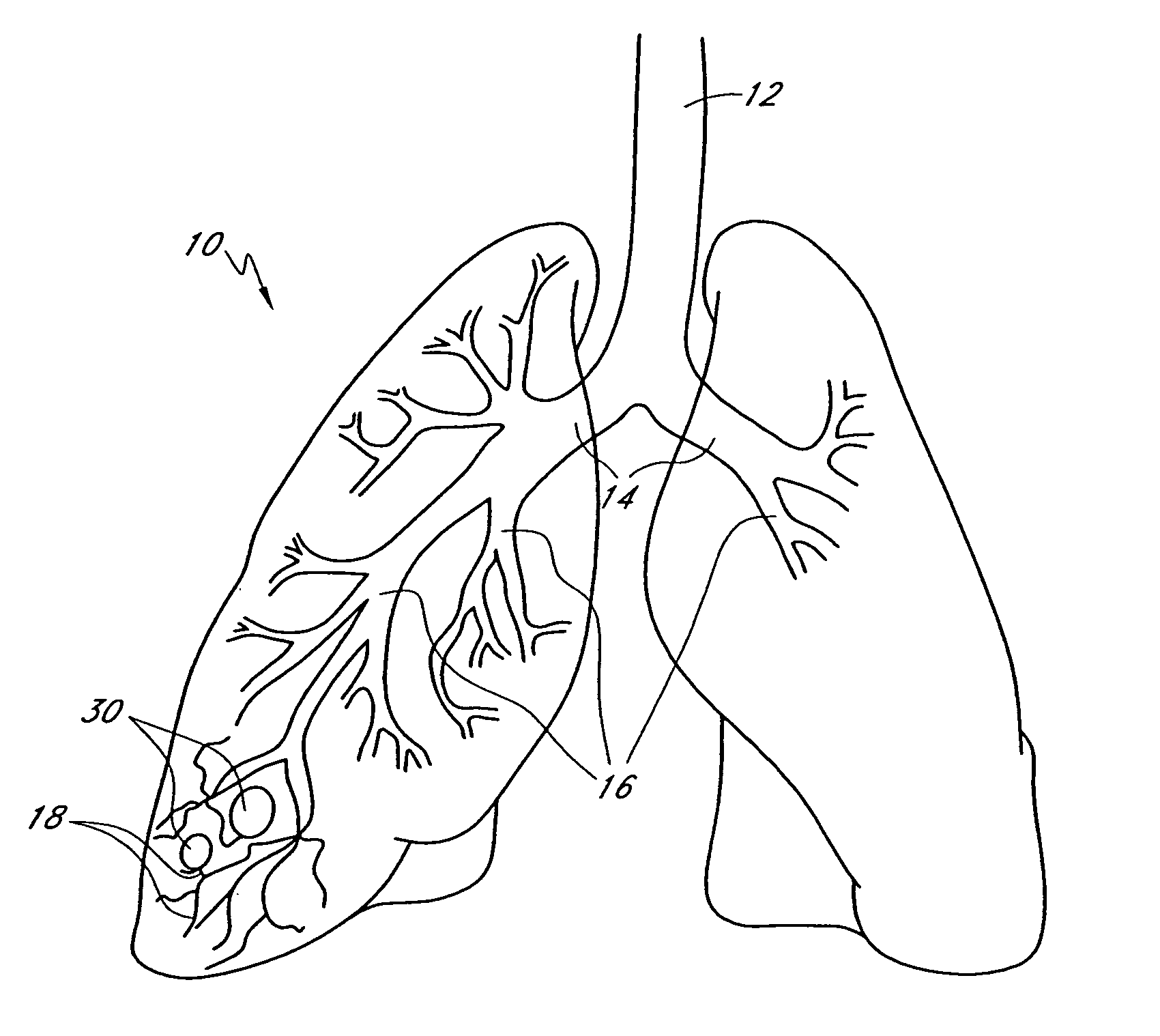
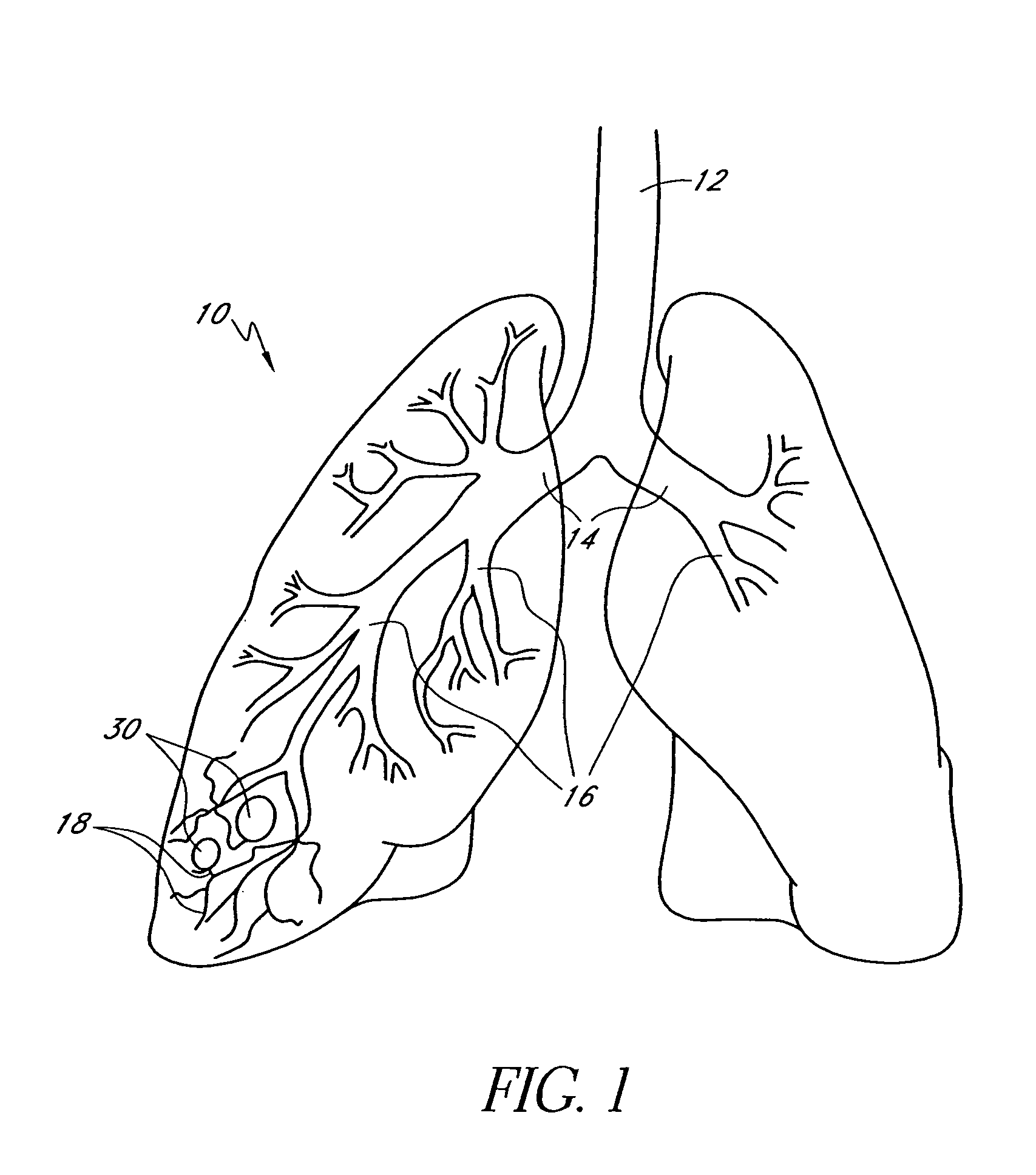
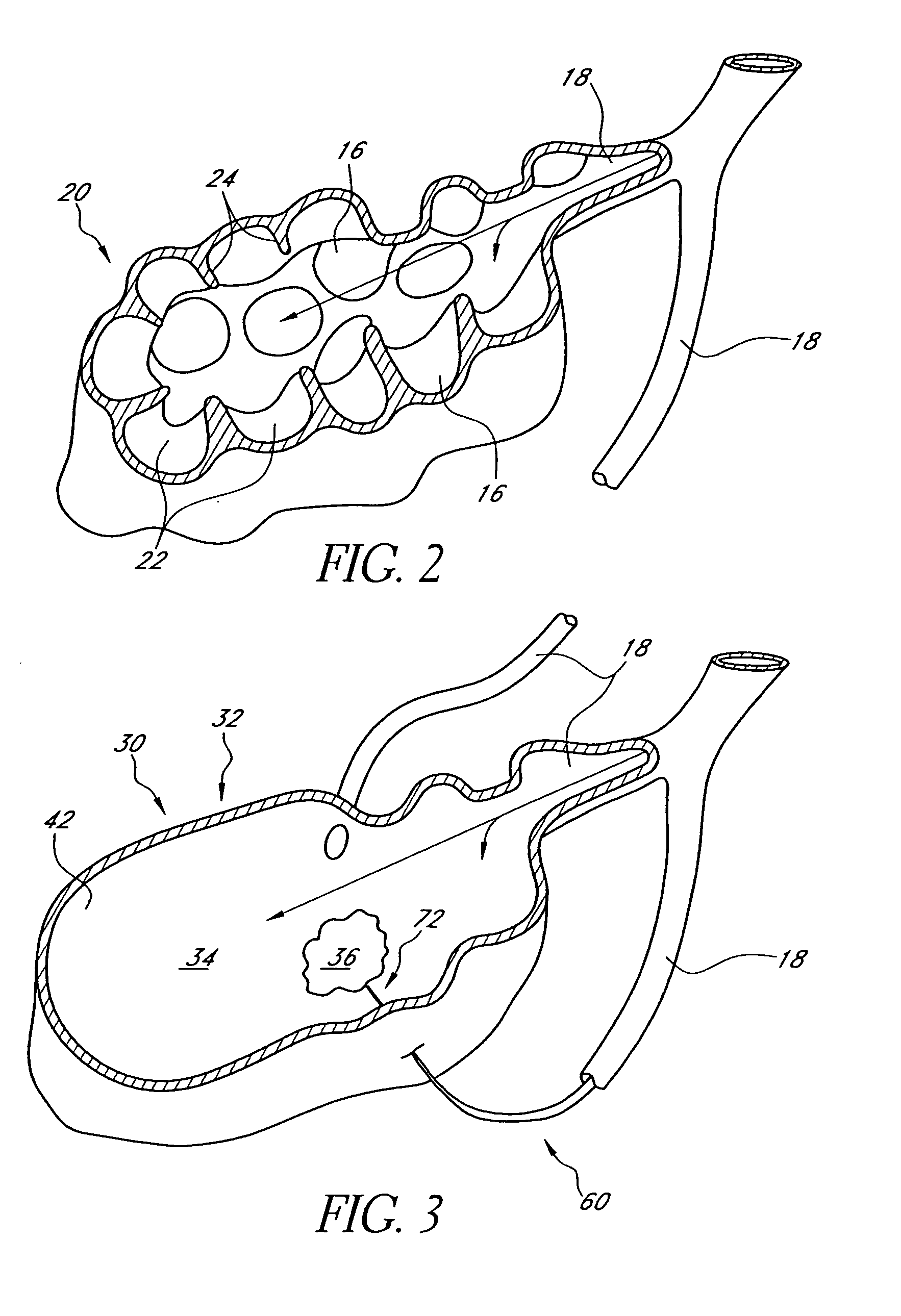
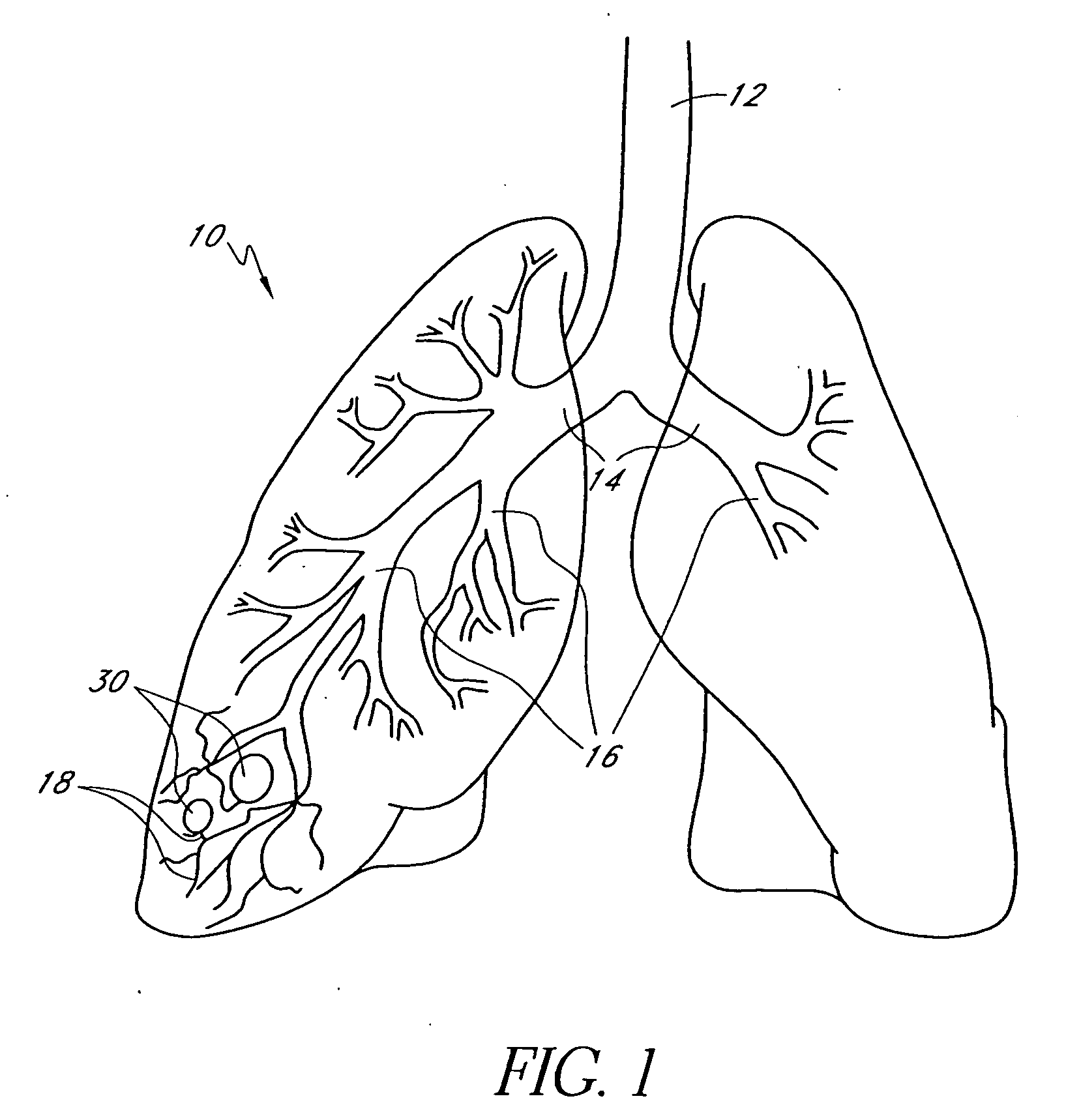
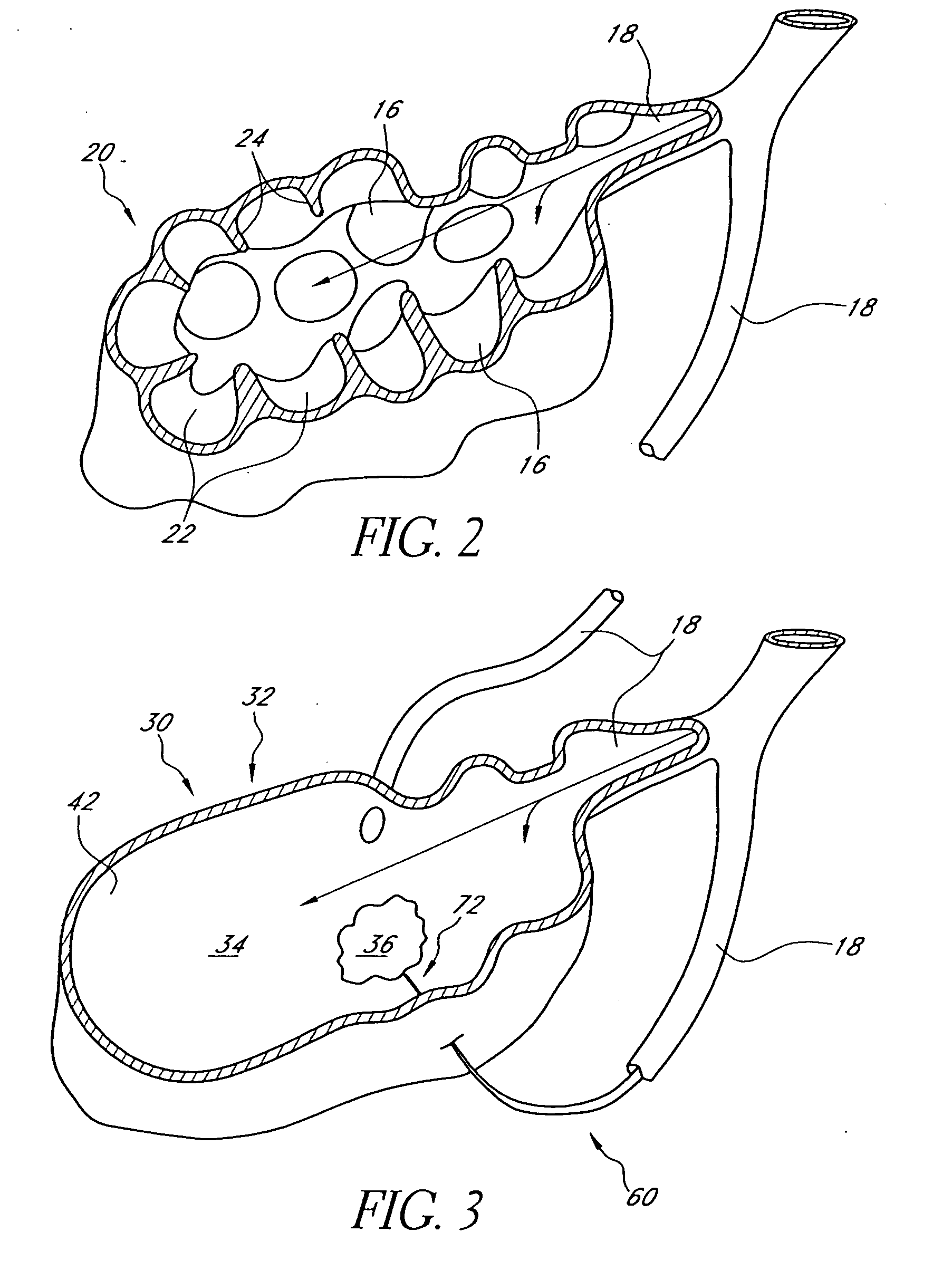
Bronchoscopic lung volume reduction method
InactiveUS7100616B2Reduce riskEffective lung volume reductionRespiratorsBronchiAirway occlusionAtmospheric air
A method of minimally invasively reducing a volume of a hyper-inflated target section of diseased lung comprising the steps of introducing a bronchoscope into a patient's airway to a position adjacent the target section and equilibrating air within the target section with atmospheric air to at least partially deflate the target lung section; injecting an inflammation-causing substance into the target section to precipitate adhesion of the walls within the target lung section, preventing substantial re-inflation of the target section by occluding an airway upstream of the target section for a period of time, and removing the airway occlusion after the target section has substantially permanently been reduced in volume. The injected substance can be autologous blood or a constituent thereof.
Owner:GYRUS ACMI INC (D B A OLYMPUS SURGICAL TECH AMERICA)
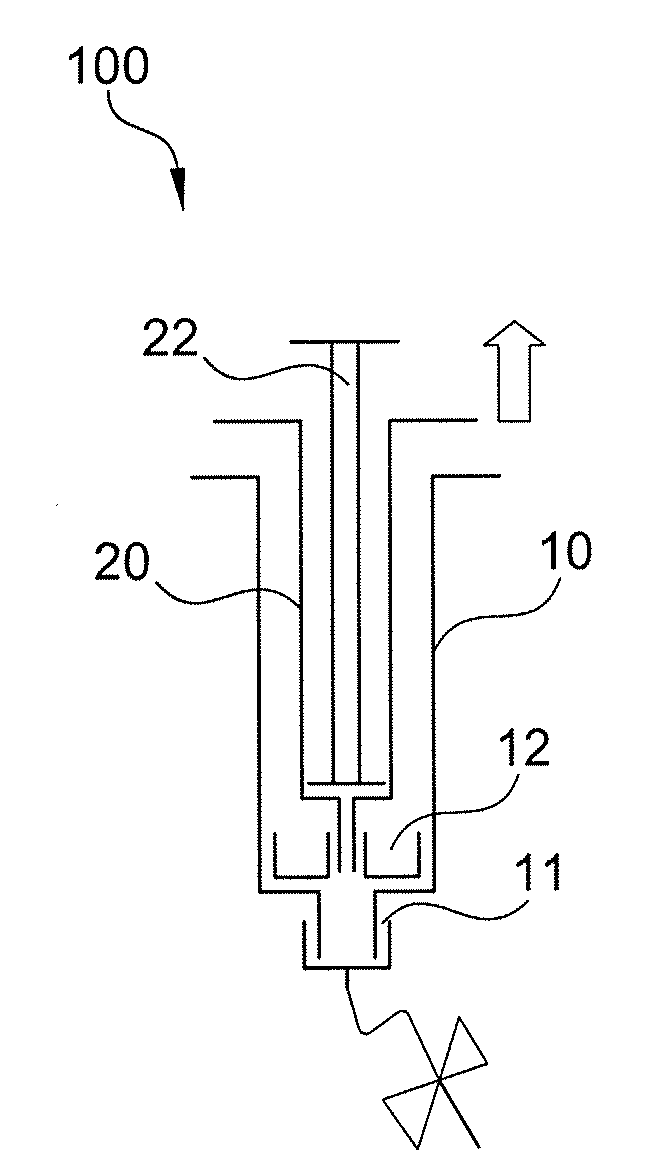
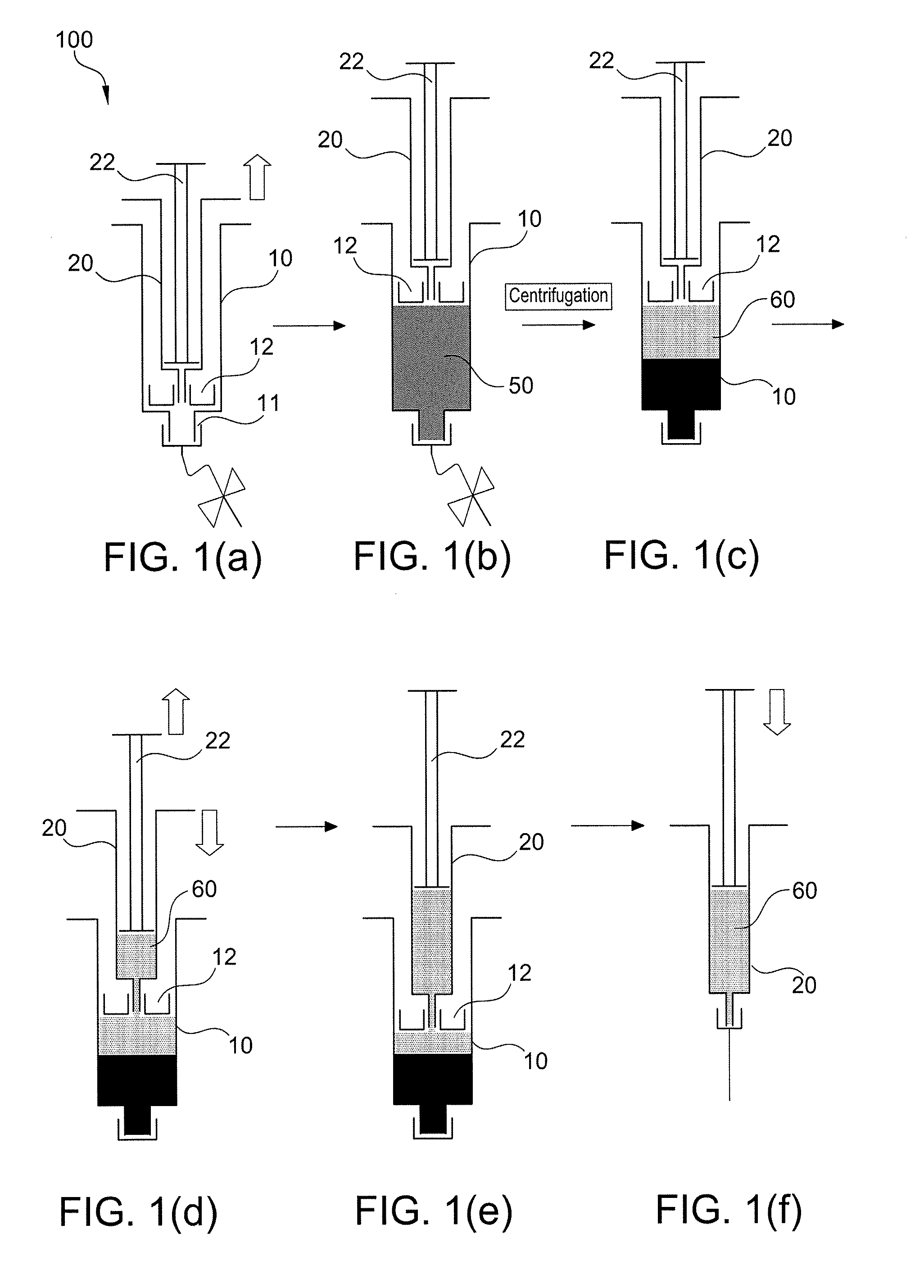
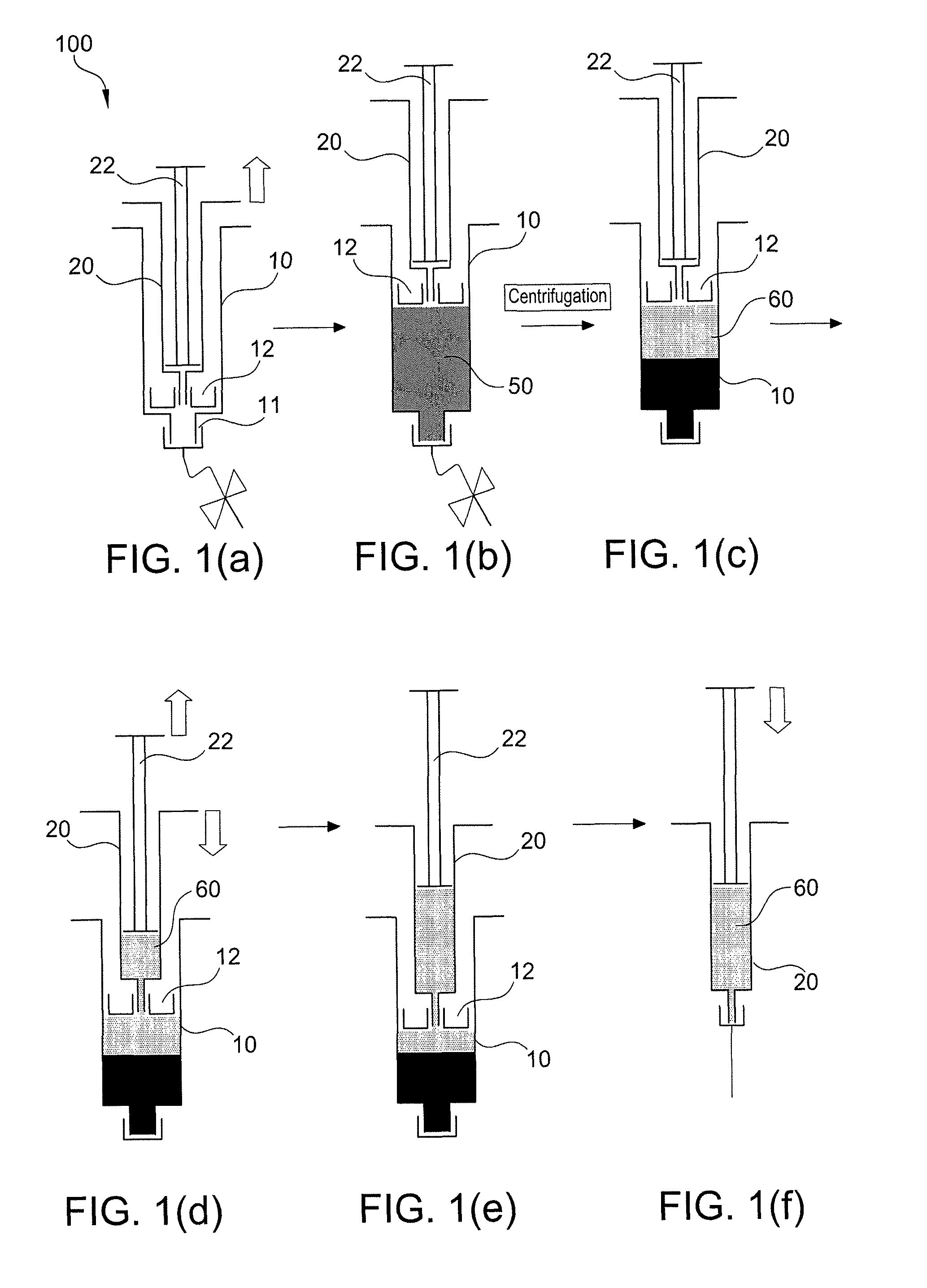
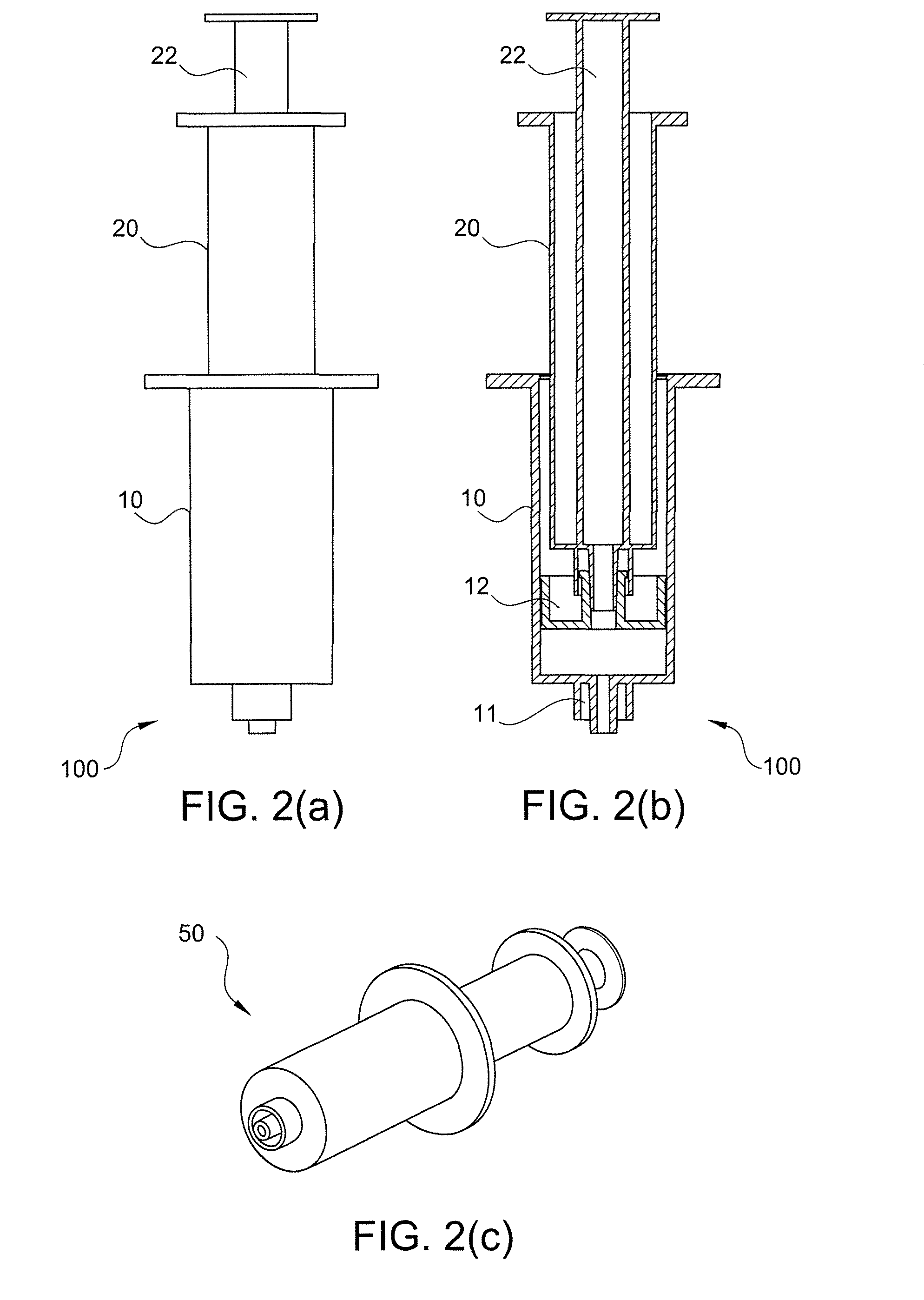
Method of surgical treatment using autologous conditioned plasma
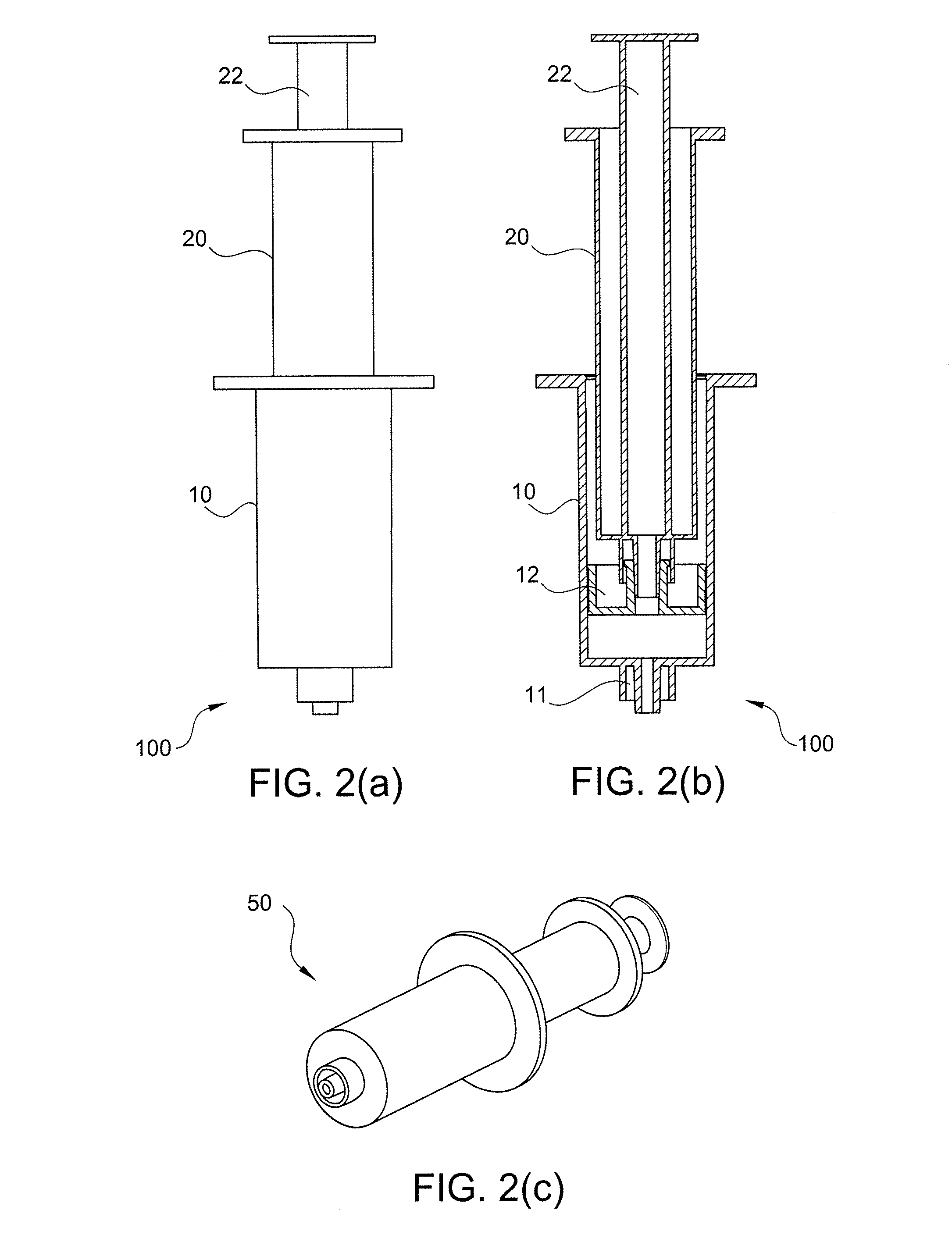
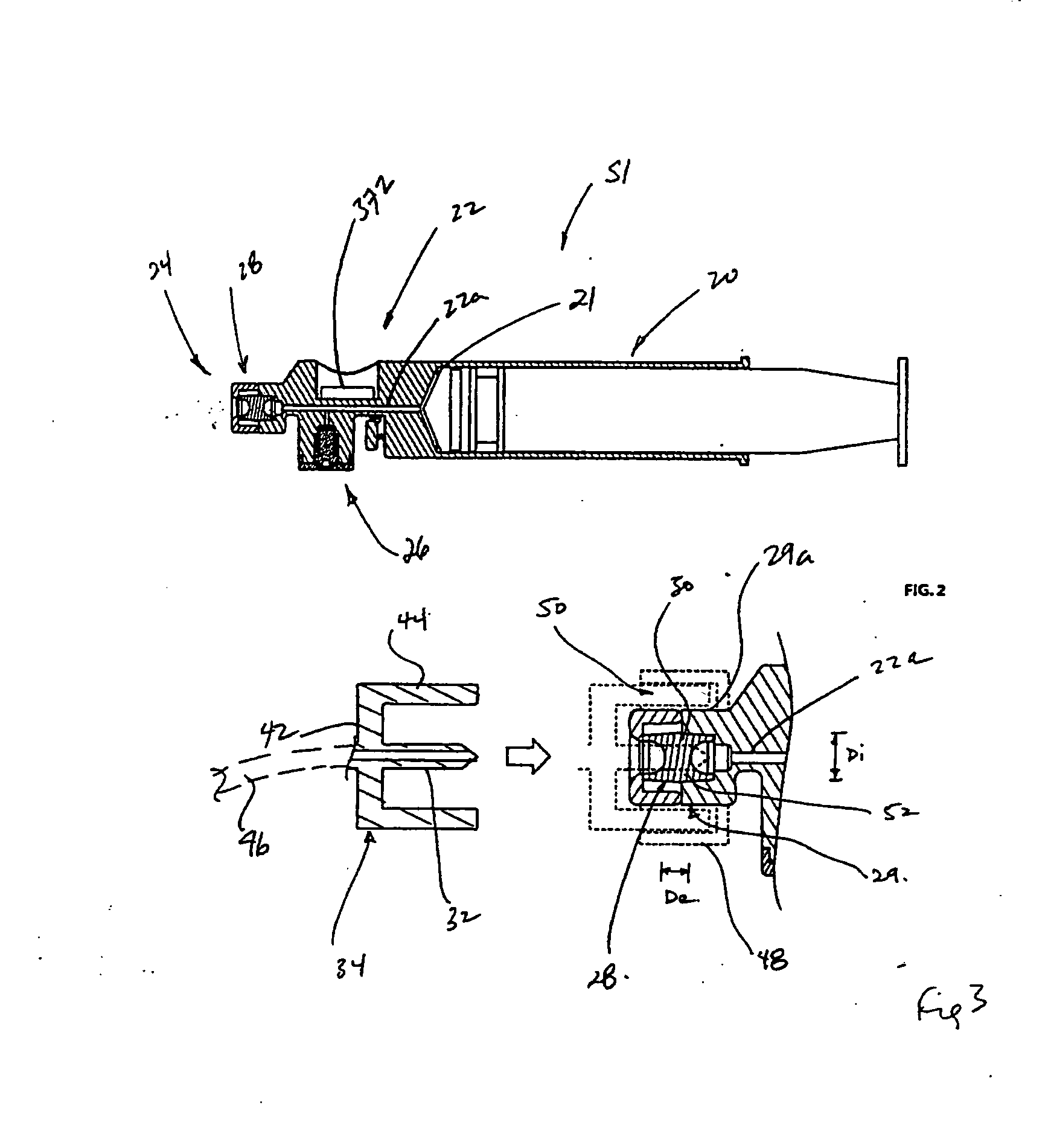
A method of providing autologous conditioned plasma (ACP) for treatment of connective tissue injuries. The method comprises the steps of: (i) providing an apparatus comprising a centrifuge and a double syringe, the double syringe including an inner syringe body and an outer syringe body; (ii) drawing autologous blood into the outer syringe body; (iii) subjecting the autologous blood to at least one centrifugation step to obtain an autologous conditioned plasma (ACP); (iv) removing, with the inner syringe body, at least a portion of autologous conditioned plasma (ACP) from the outer syringe body; and (v) inject ACP for treatment of various cartilage or tendon damage or diseases.
Owner:ARTHREX
Medical material handling systems
InactiveUS20050154368A1Inhibition releaseAvoid treatmentInfusion syringesSurgeryFluid couplingAutologous blood
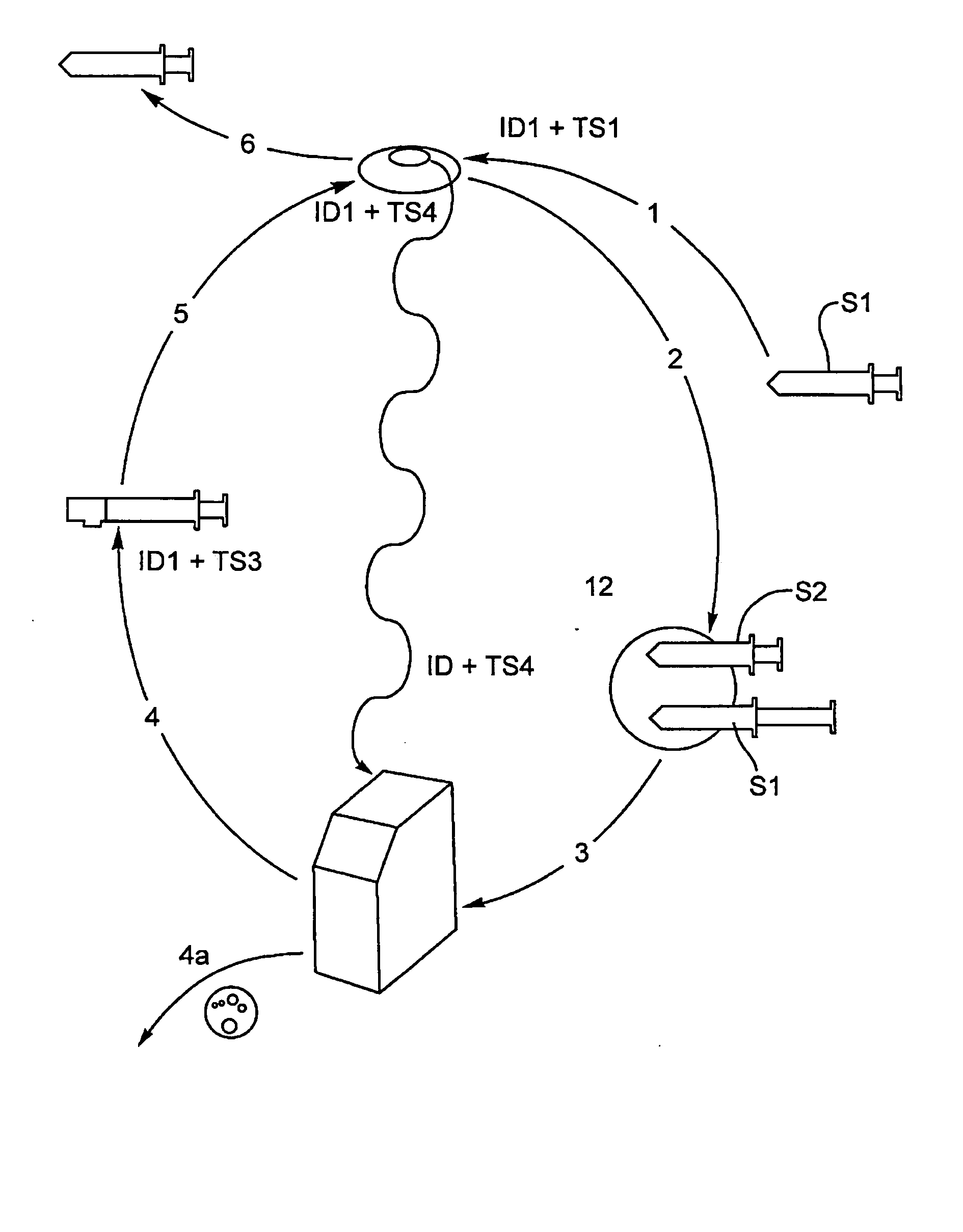
Disclosed herein is a system for the collection, treatment and delivery of an autologous blood sample, comprising a first syringe having a first body portion. A first sample transfer portion has a first syringe inlet for drawing an untreated blood sample from a patient and a first syringe outlet for dispensing the untreated blood sample. A blood sample treatment chamber having a chamber inlet, the first syringe outlet being operable to establish a dedicated first fluid coupling with the chamber inlet to dispense the untreated blood sample to the blood sample treatment chamber. The blood sample treatment chamber having a chamber outlet for dispensing a treated blood sample following treatment. The second syringe has a second body portion and a second sample transfer portion, the second sample transfer portion having a passage with a first access location which is operable to form a dedicated second fluid coupling with the chamber outlet. The second body portion has a second syringe outlet, the passage having a second access location for fluid communication with the second syringe outlet, releasable lock means for forming a locked third fluid coupling between the second access location and the second syringe outlet. The lock means is operable in response to a release signal to release the third fluid coupling, the second syringe outlet being operable when released from the third fluid coupling to form a fourth fluid coupling with a blood sample delivery unit.
Owner:VASOGEN IRELAND LTD
Verification method and system for medical treatment
InactiveUS20060264778A1Substantial accuracy of clockDecreasing clock accuracyMedical devicesCatheterBlood specimenAutologous blood
A system for the collection, treatment and delivery of an autologous blood sample, comprising a first syringe for drawing an untreated blood sample from a patient, a blood sample treatment chamber having a chamber inlet for receiving untreated blood from the first syringe and chamber outlet for passage of treated blood to a second syringe coupled thereto. The second syringe includes a releasable lock means for allowing discharge of the treated blood to the patient in response to a release signal. The release signal is issued following a positive outcome from a verification process dependent upon temporal data from certain events in the collection, treatment and delivery of the blood sample, and identity data of the patient and the second syringe with the treated blood.
Owner:VASOGEN IRELAND LTD
Bronchoscopic lung volume reduction method
A method of minimally invasively reducing a volume of a hyper-inflated target section of diseased lung comprising the steps of introducing a bronchoscope into a patient's airway to a position adjacent the target section and equilibrating air within the target section with atmospheric air to at least partially deflate the target lung section; injecting an inflammation-causing substance into the target section to precipitate adhesion of the walls within the target lung section, preventing substantial re-inflation of the target section by occluding an airway upstream of the target section for a period of time, and removing the airway occlusion after the target section has substantially permanently been reduced in volume. The injected substance can be autologous blood or a constituent thereof.
Owner:GYRUS ACMI INC (D B A OLYMPUS SURGICAL TECH AMERICA)
Thrombus aspiration system
ActiveCN103932756AGreat suctionSuction completelySurgeryIntravenous devicesTreatment effectThrombus aspiration
The invention discloses a thrombus aspiration system. The thrombus aspiration system comprises a thrombus aspiration catheter and a blood bottle connected with the thrombus aspiration catheter in a matched mode. Because the ultra-thin pipe wall, wide cavity and single-cavity piping design is adopted in the thrombus aspiration catheter, thrombus aspiration can be smoothly and efficiently carried out. Because the outer edge of the far-end face of a tapered catheter section is in the fillet shape, the whole thrombus aspiration catheter can enter the vessel easily, and injury to the human body can be reduced. Because elliptical side holes are spirally distributed in the far end, large thrombi in the vessel can be easily and smoothly aspirated, the situation that the large-sized thrombi can not be aspirated is avoided, the thrombi in the vessel can be completely aspirated, and the best treatment effect is achieved. Blood aspirated from the human body during an operation flows into the blood bottle, the aspirated thrombi are filtered out through a thrombus filter screen in the blood bottle, the filtered blood can be transfused into the human body again, and therefore the autologous blood can be quickly and effectively utilized and the adverse effects like contamination of allogeneic blood to a patient is avoided.
Owner:吴智群 +1
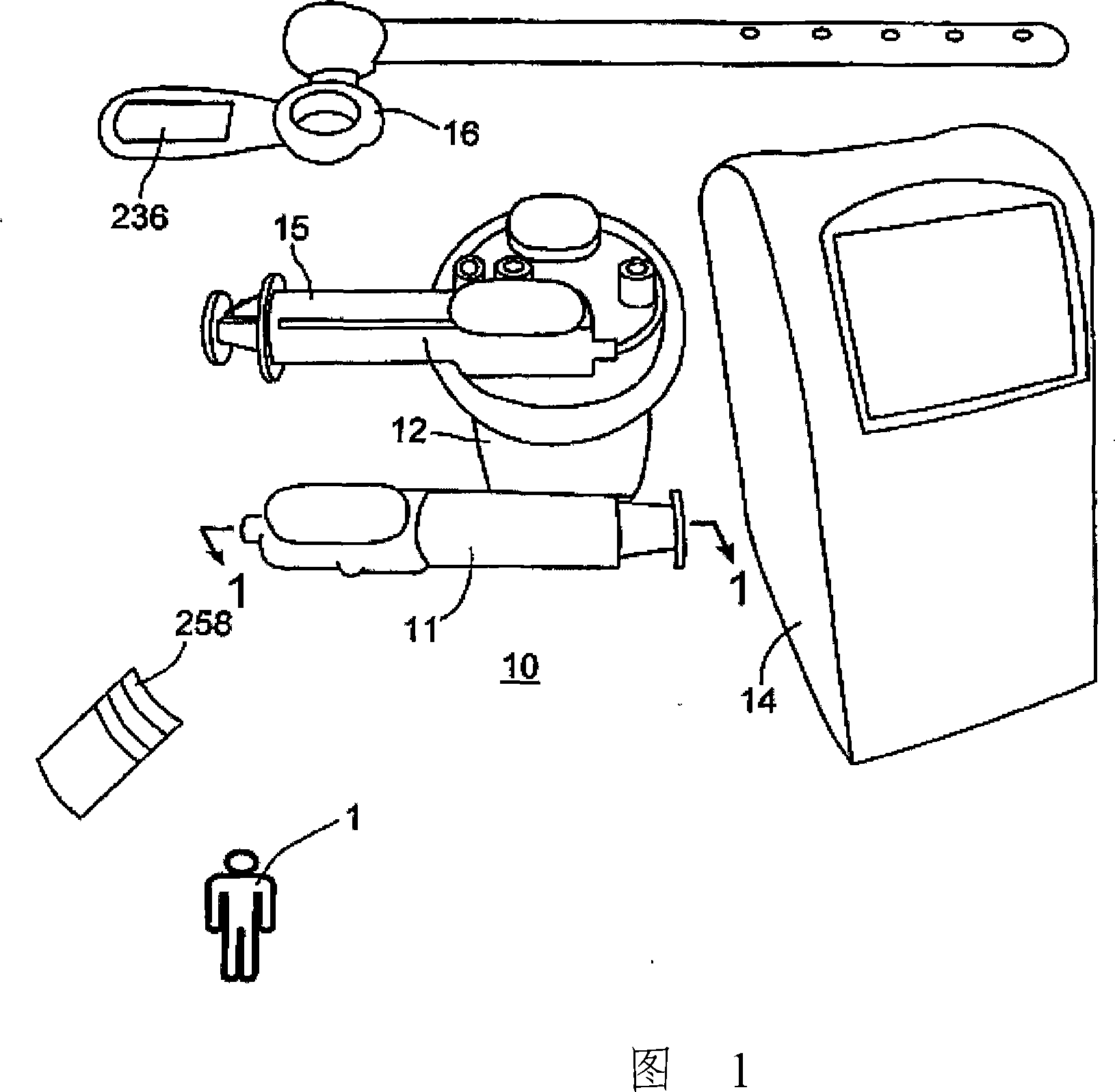
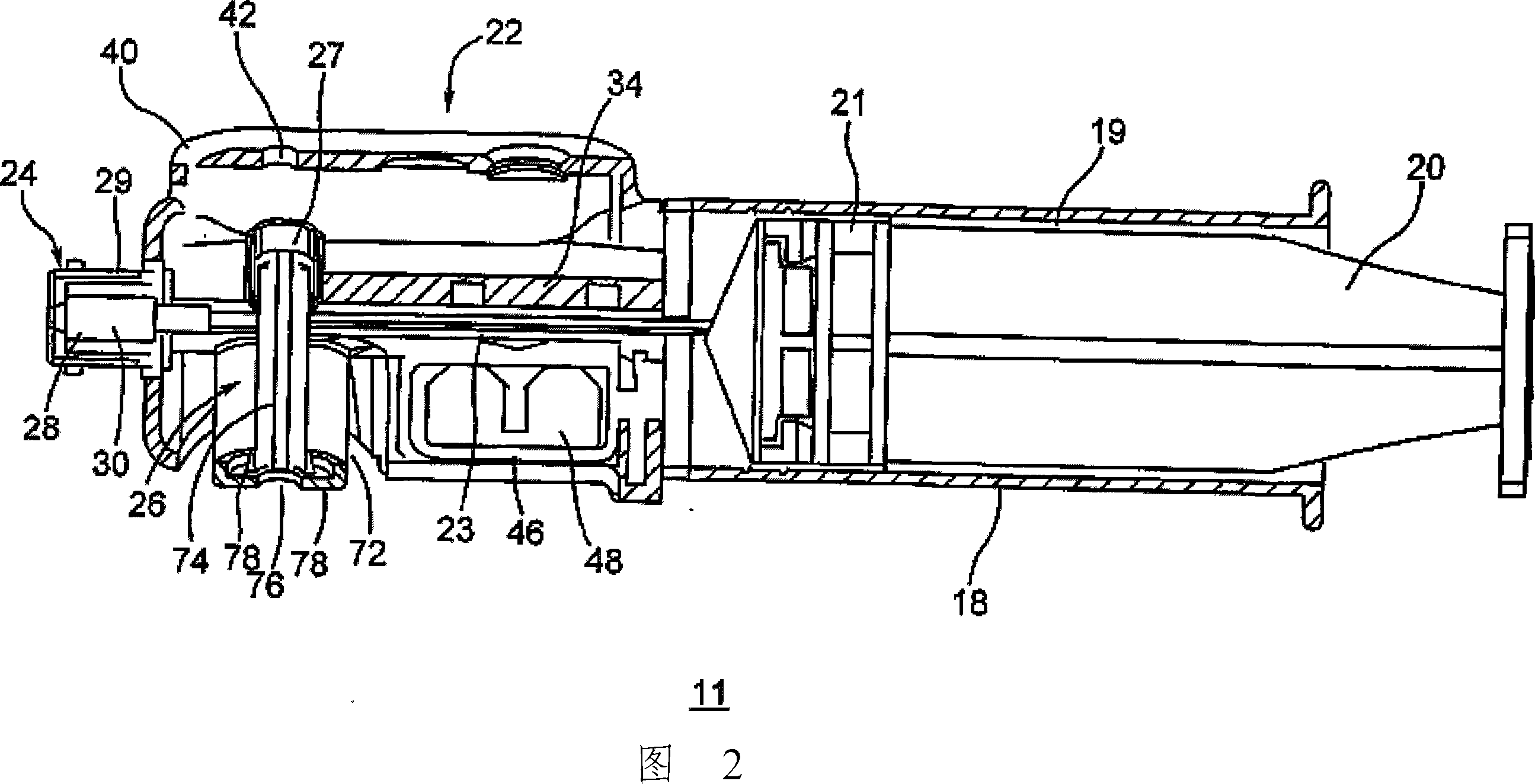
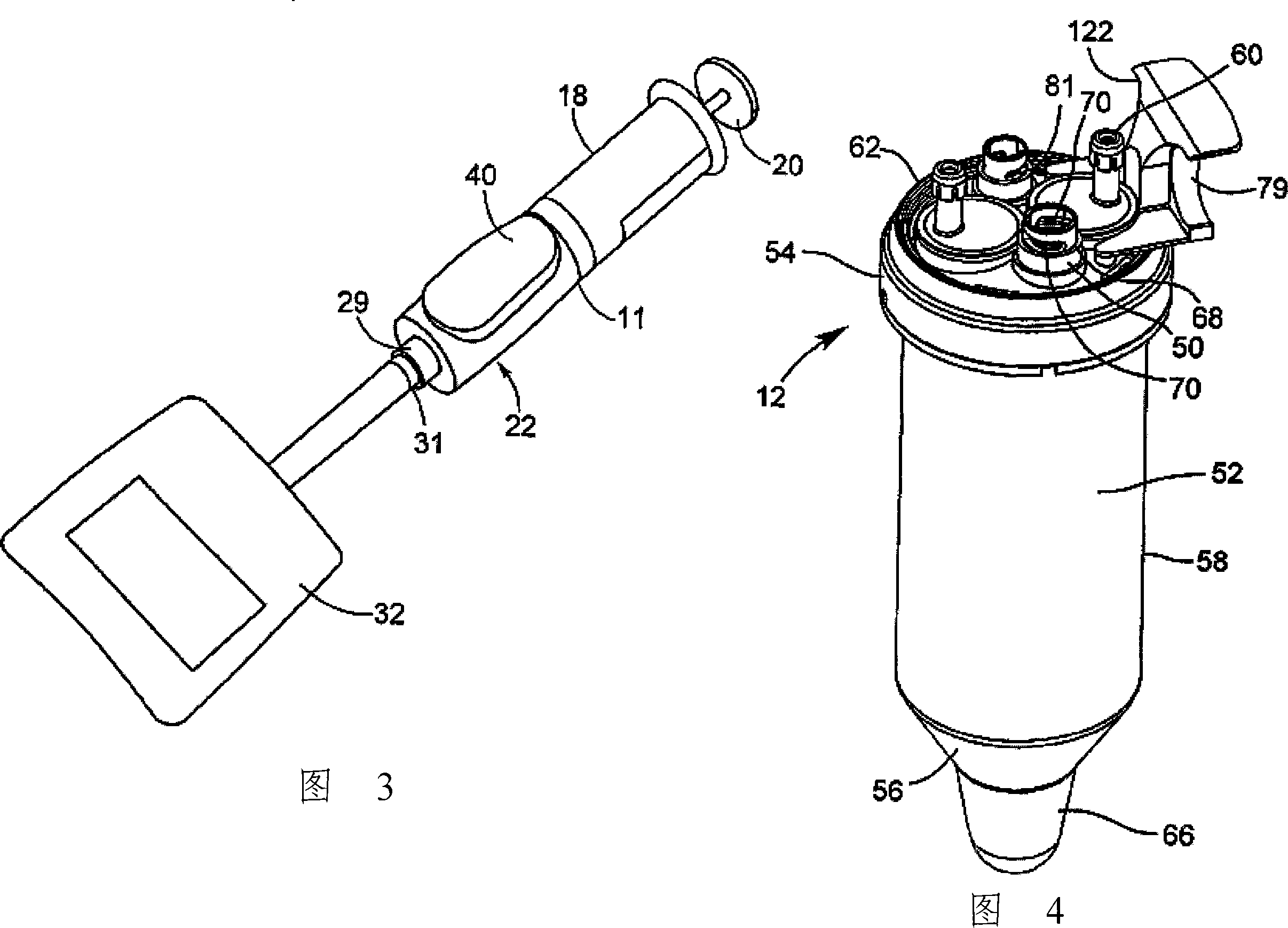
Device and method for autologous blood transfusion
InactiveUS6964646B1Avoid reactionReduce in quantityOther blood circulation devicesRotary centrifugesCell FractionLine tubing
A device for autologous blood transfusion includes a centrifuge unit with an autotransfusion set rotatably mounted thereto. The autotransfusion set includes a separation unit for concentrating a cell fraction by centrifugation and a tubing system for providing a connection to a blood supply. The tubing system includes a blood supply line leading to the separation unit for supplying blood to be processed and a return line leading away from the separation unit. A filter is integrated into the autotransfusion set in order to eliminate leukocytes and / or tumor cells, thereby yielding greater safety in autotransfusion.
Owner:FRESENIUS HEMOCARE
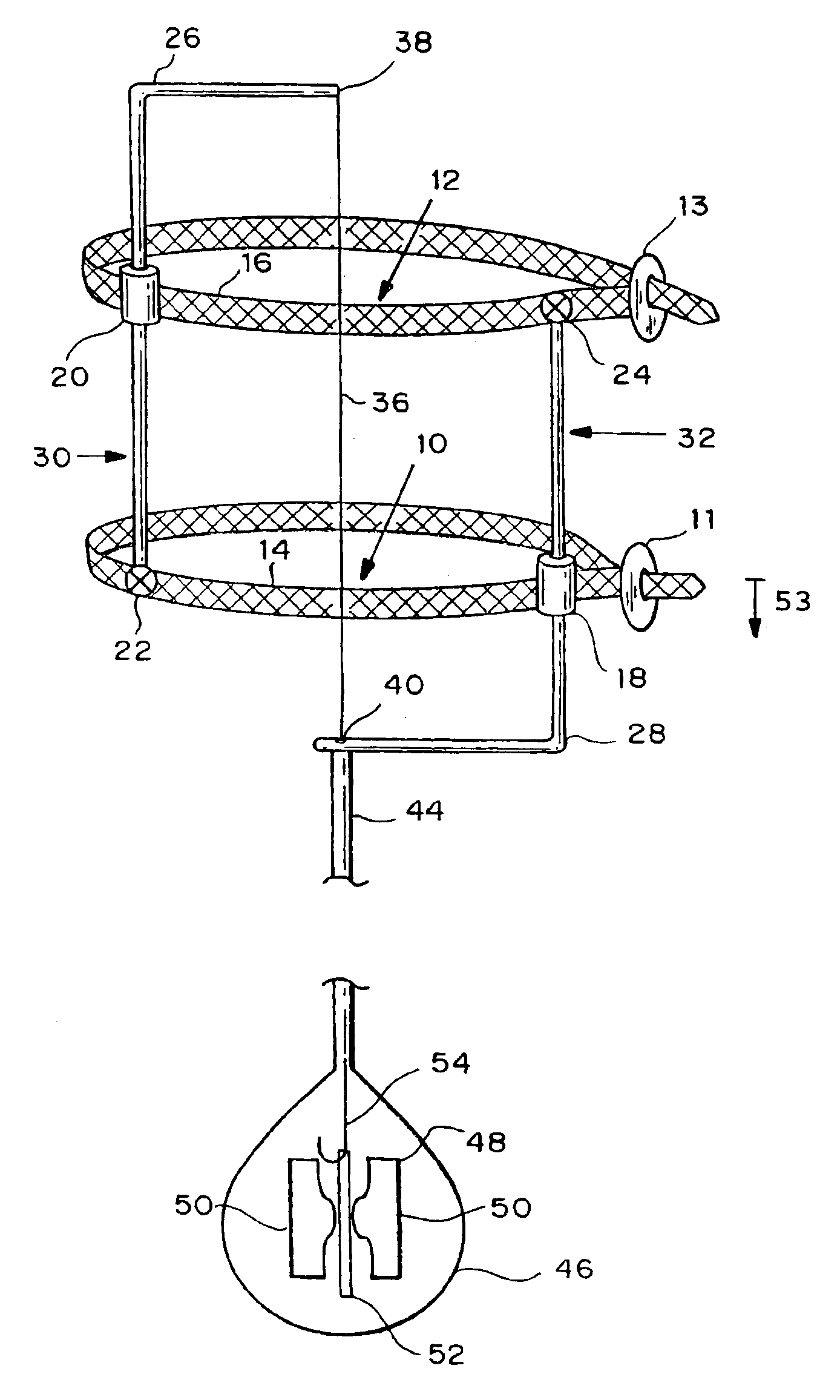
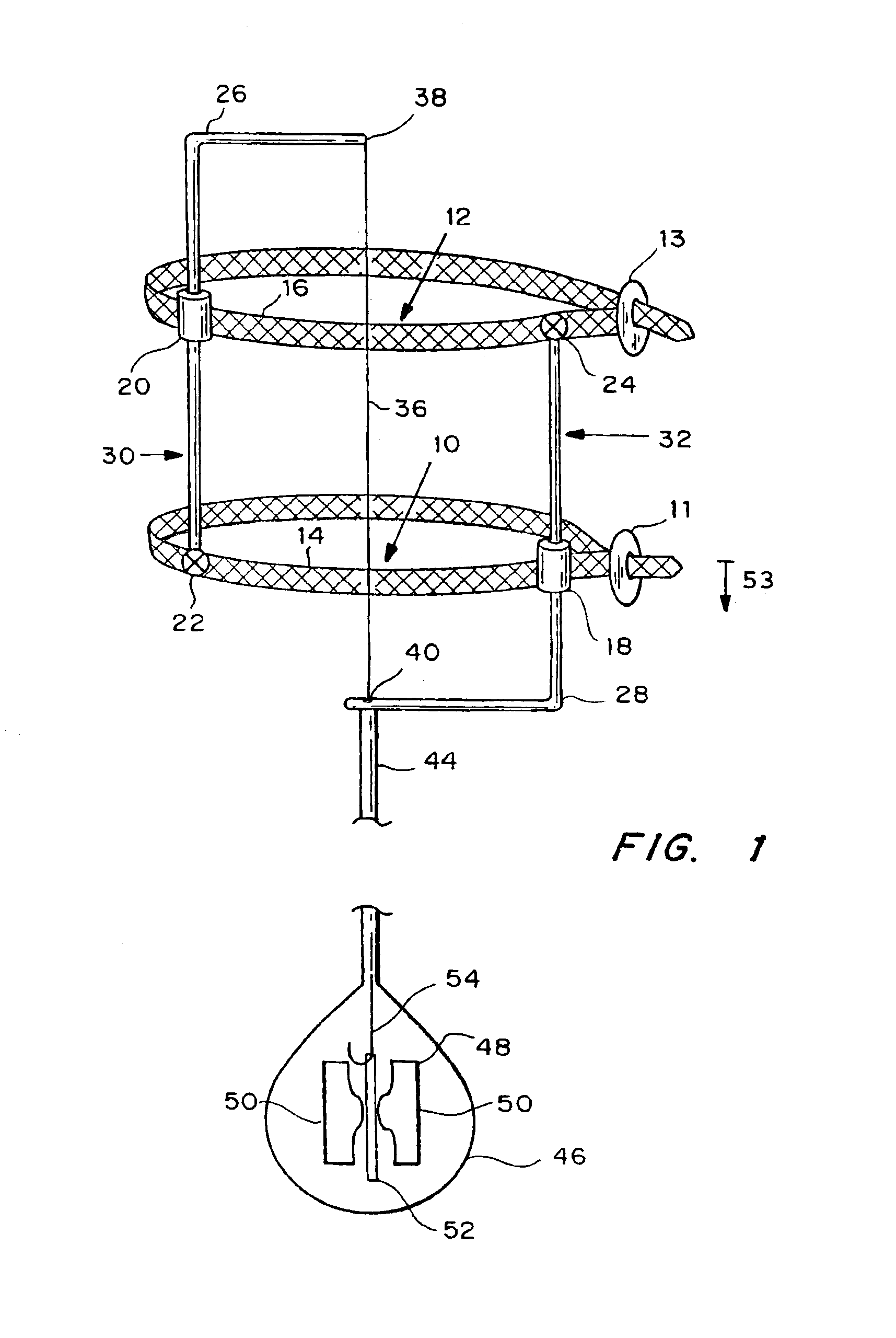
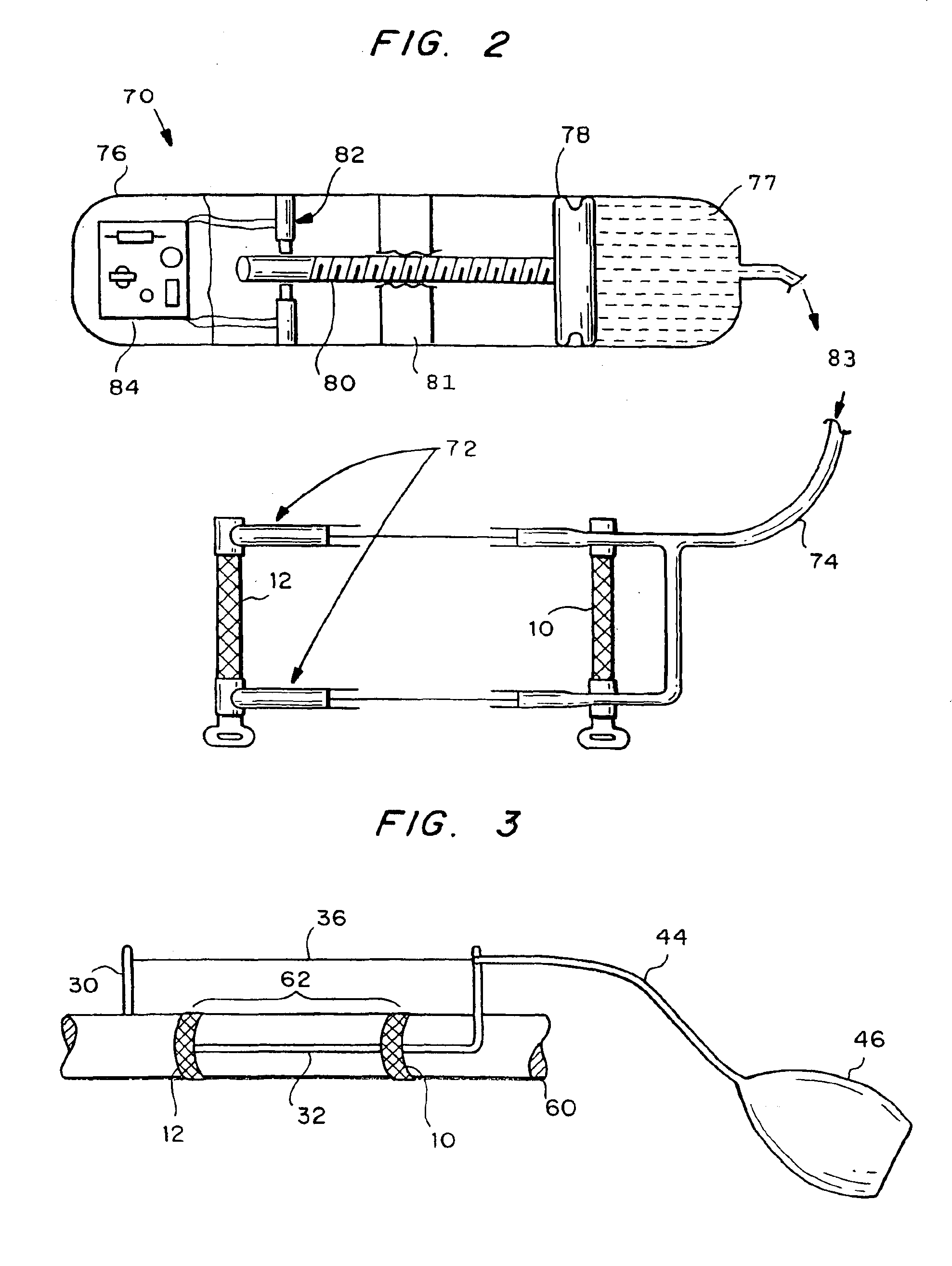
Autologous vascular grafts created by vessel distension
Devices and methods are provided for forming a vascular graft by axially distending a blood vessel to induce growth. In one embodiment, a device for distending a blood vessel of a human or animal is provided which comprises a stretching mechanism attachable directly to a blood vessel at at least two attachment positions thereon, and a means for operating the stretching mechanism to cause the blood vessel portion between said at least two attachment positions to be stretched axially. The distended portion can then be excised and used as a graft.
Owner:GEORGIA TECH RES CORP
Method of making plasma enriched with platelets using a double syringe system where the second sytringe is within the first syringe and use thereof
A method of providing autologous conditioned plasma (ACP) for treatment of connective tissue injuries. The method comprises the steps of: (i) providing an apparatus comprising a centrifuge and a double syringe, the double syringe including an inner syringe body and an outer syringe body; (ii) drawing autologous blood into the outer syringe body; (iii) subjecting the autologous blood to at least one centrifugation step to obtain an autologous conditioned plasma (ACP); (iv) removing, with the inner syringe body, at least a portion of autologous conditioned plasma (ACP) from the outer syringe body; and (v) inject ACP for treatment of various cartilage or tendon damage or diseases.
Owner:ARTHREX
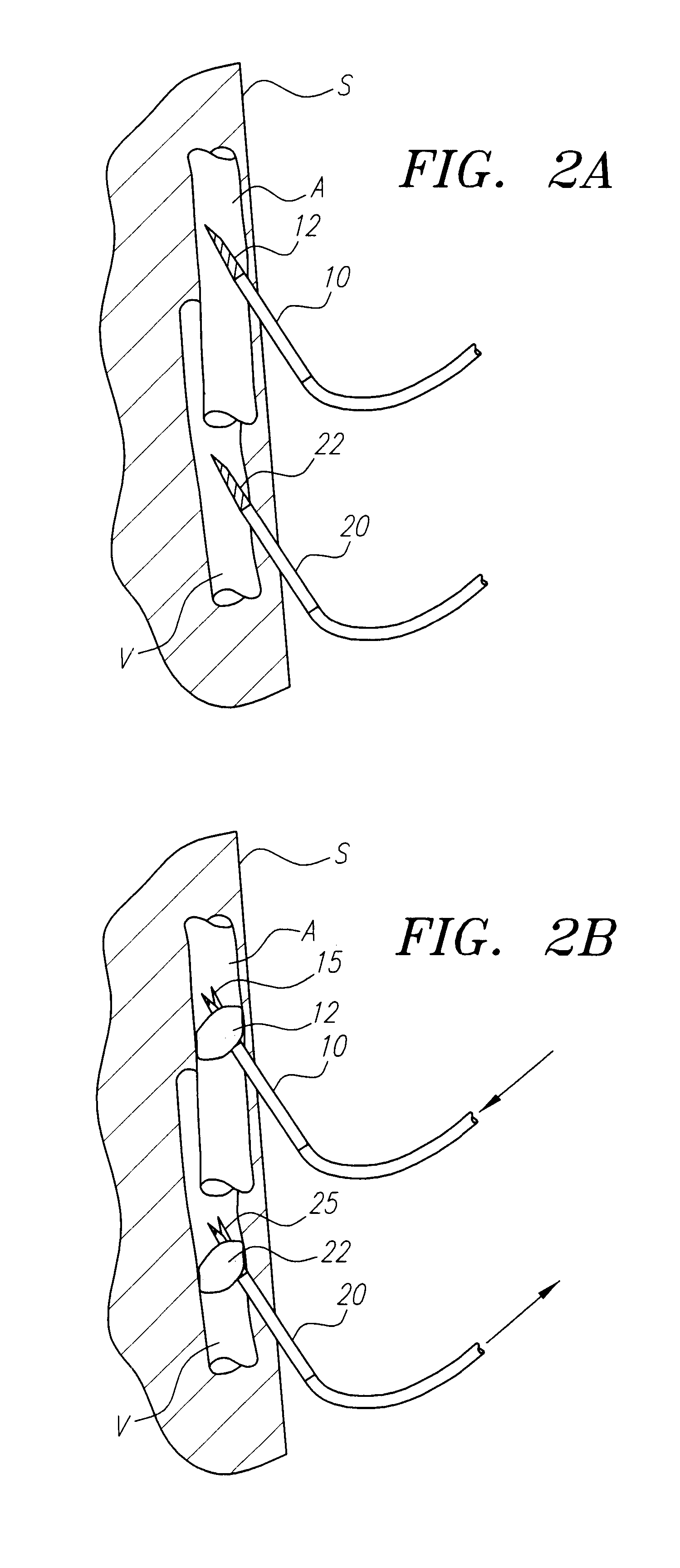
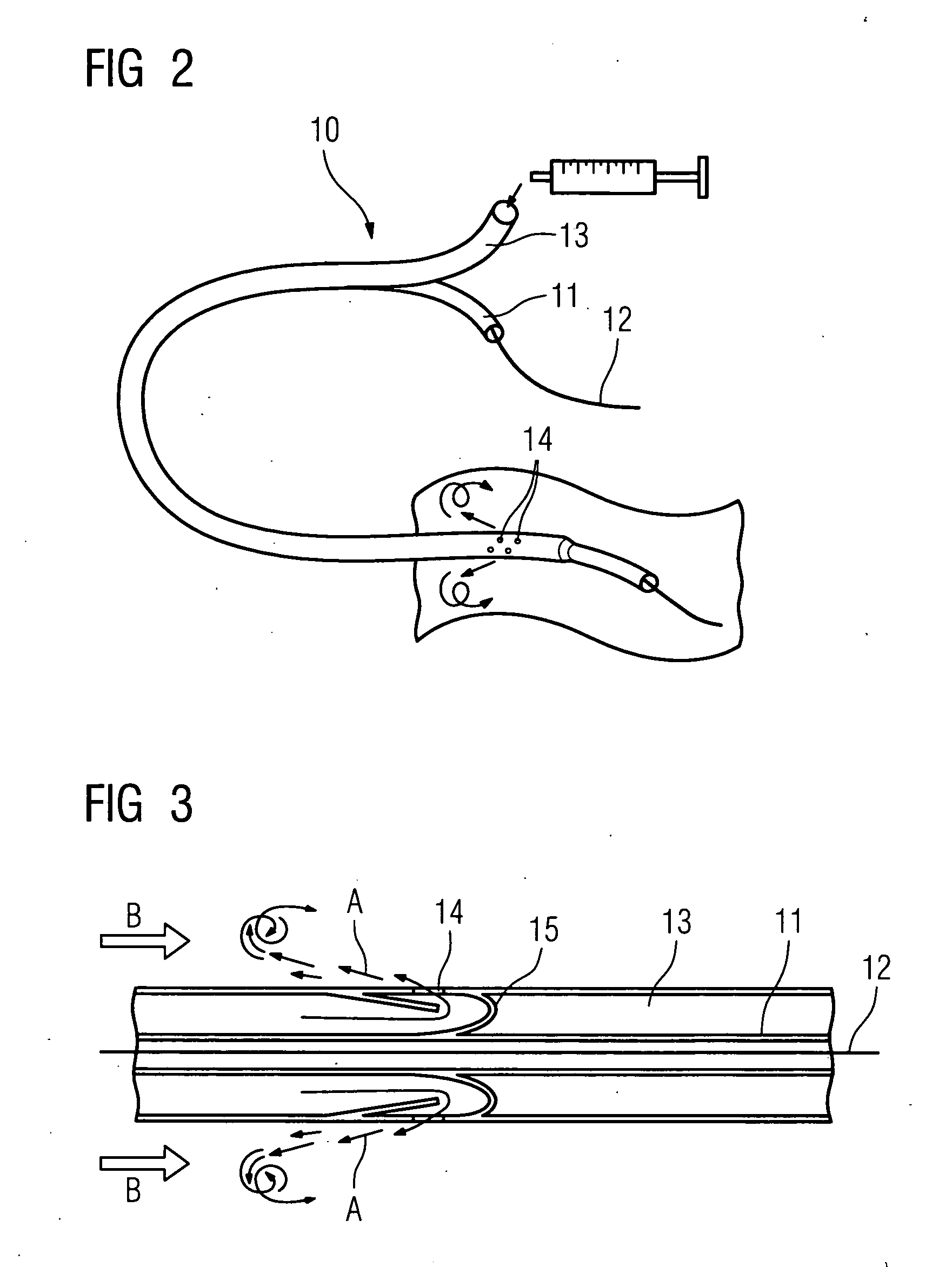
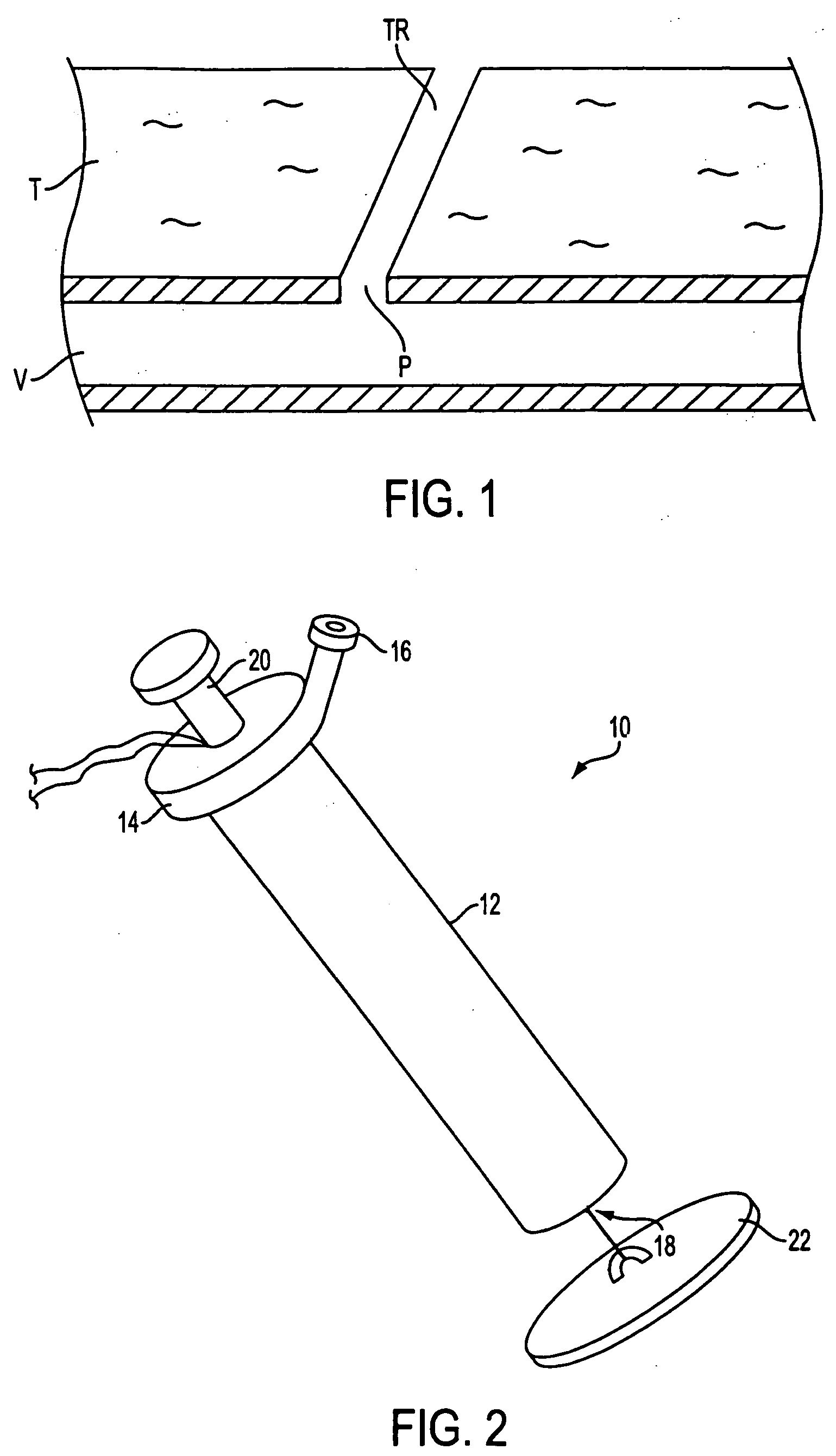
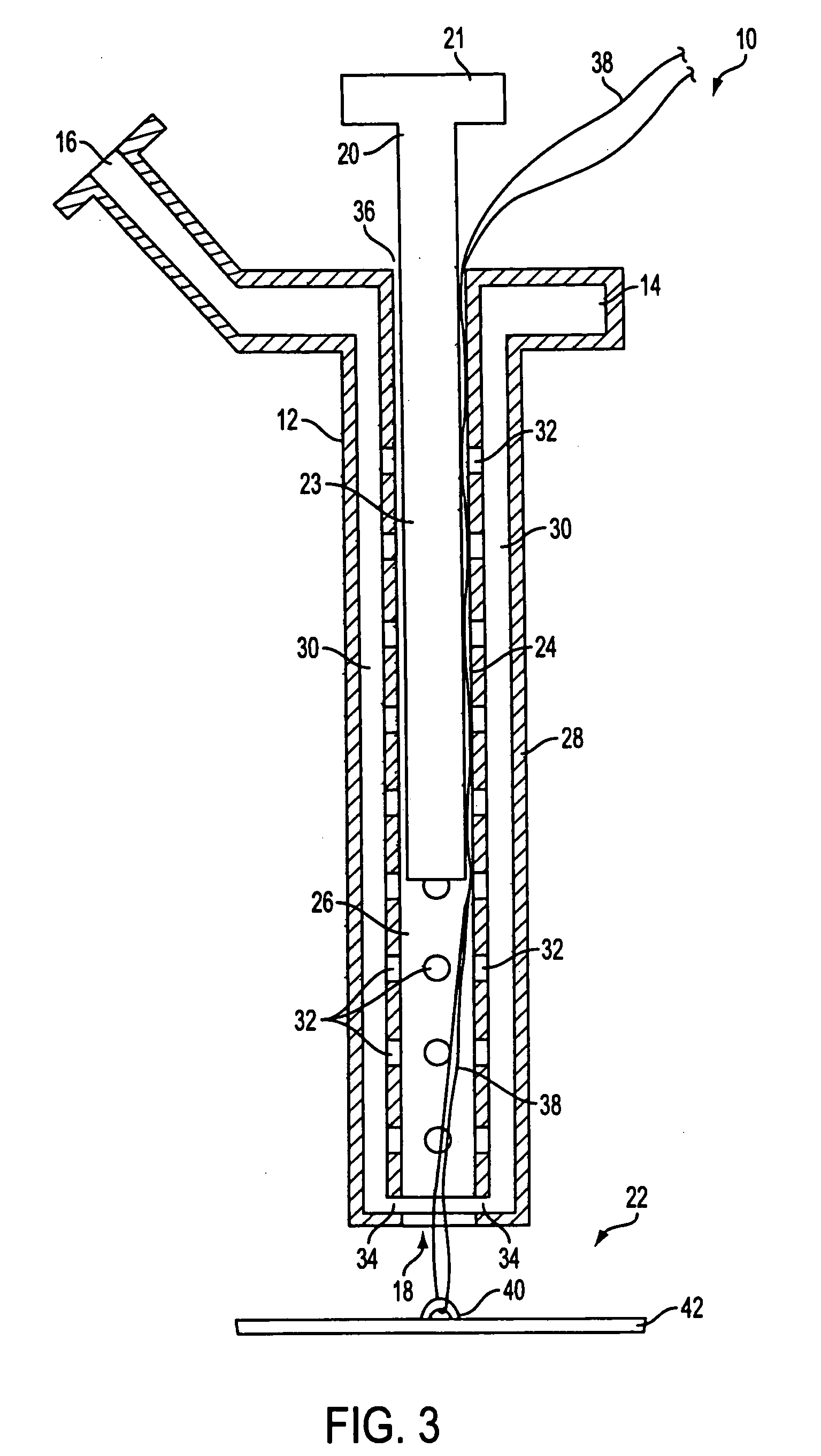
Autologous wound sealing apparatus
InactiveUS7771454B2Easy to useReduce contaminationSurgical veterinaryWound clampsAutologous bloodBlood vessel
Owner:ABBOTT LAB VASCULAR ENTERPRISE
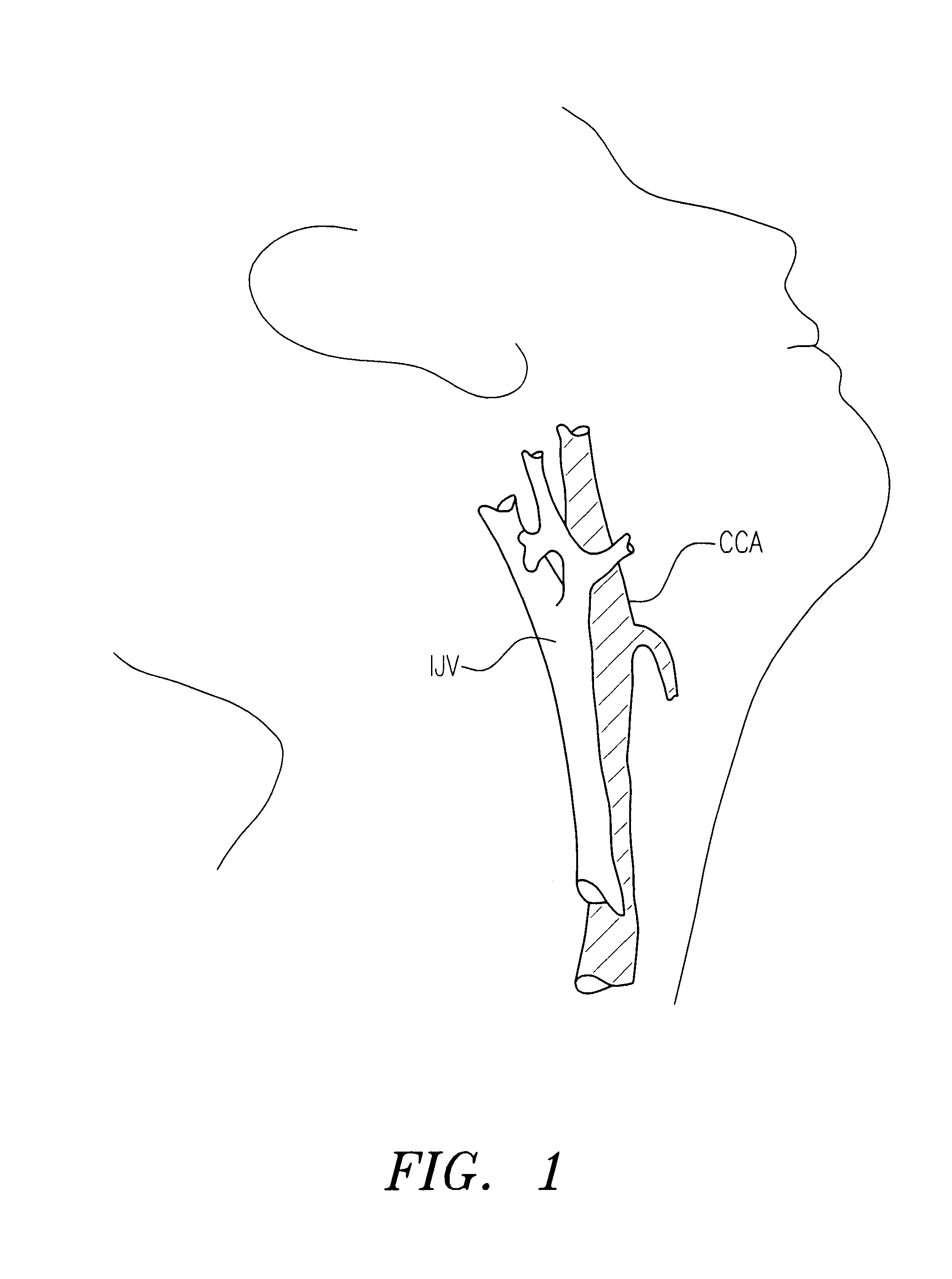
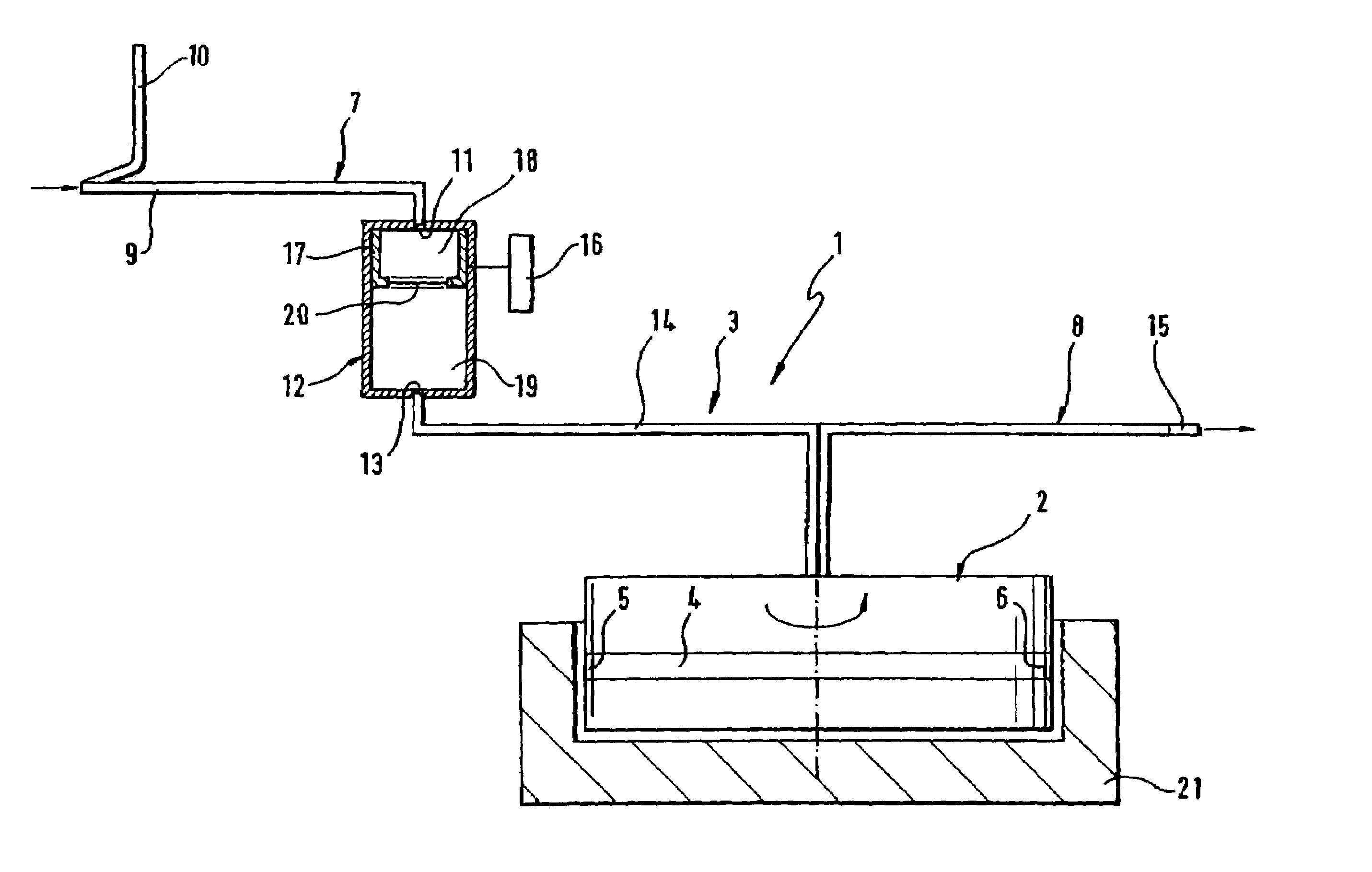
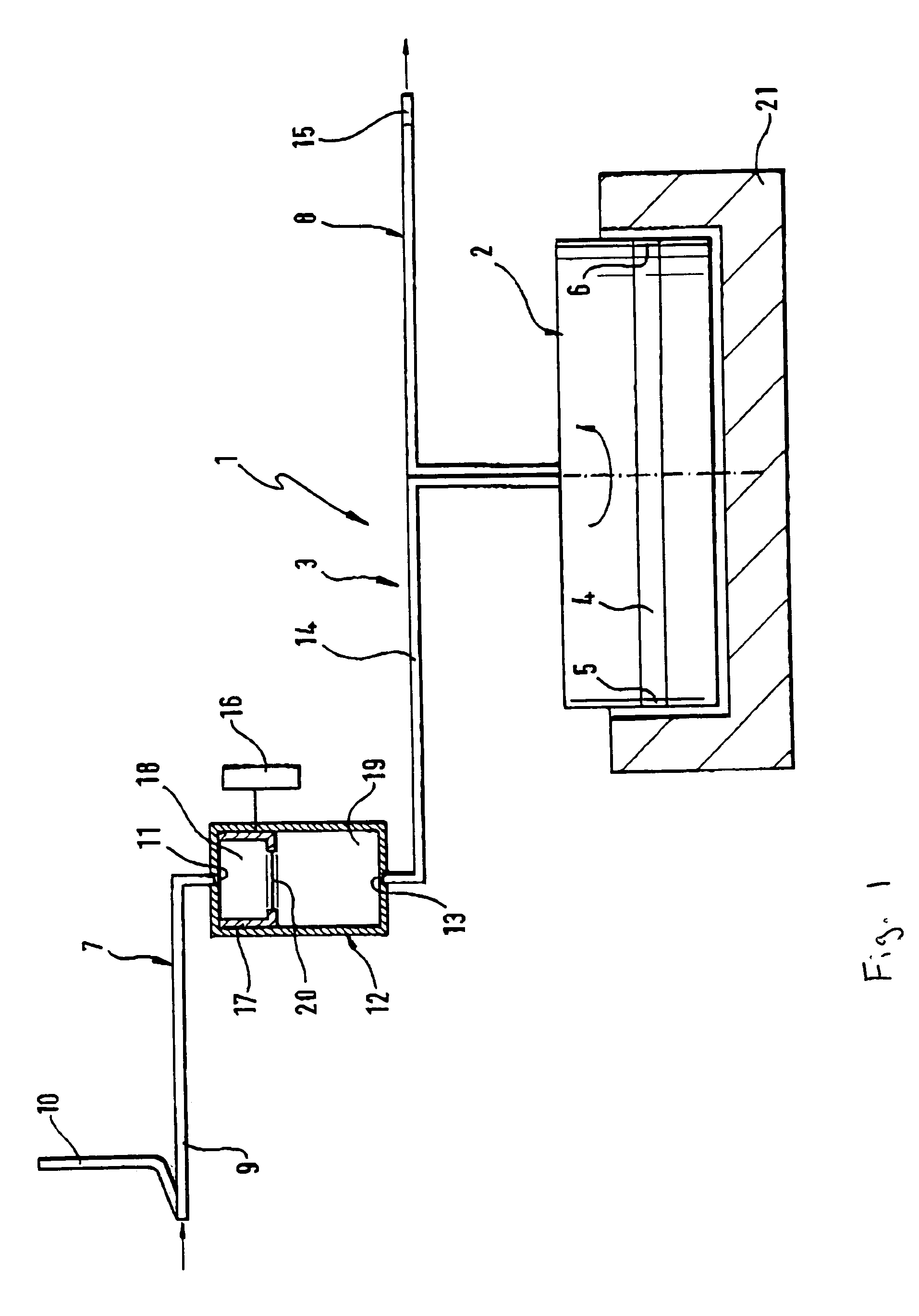
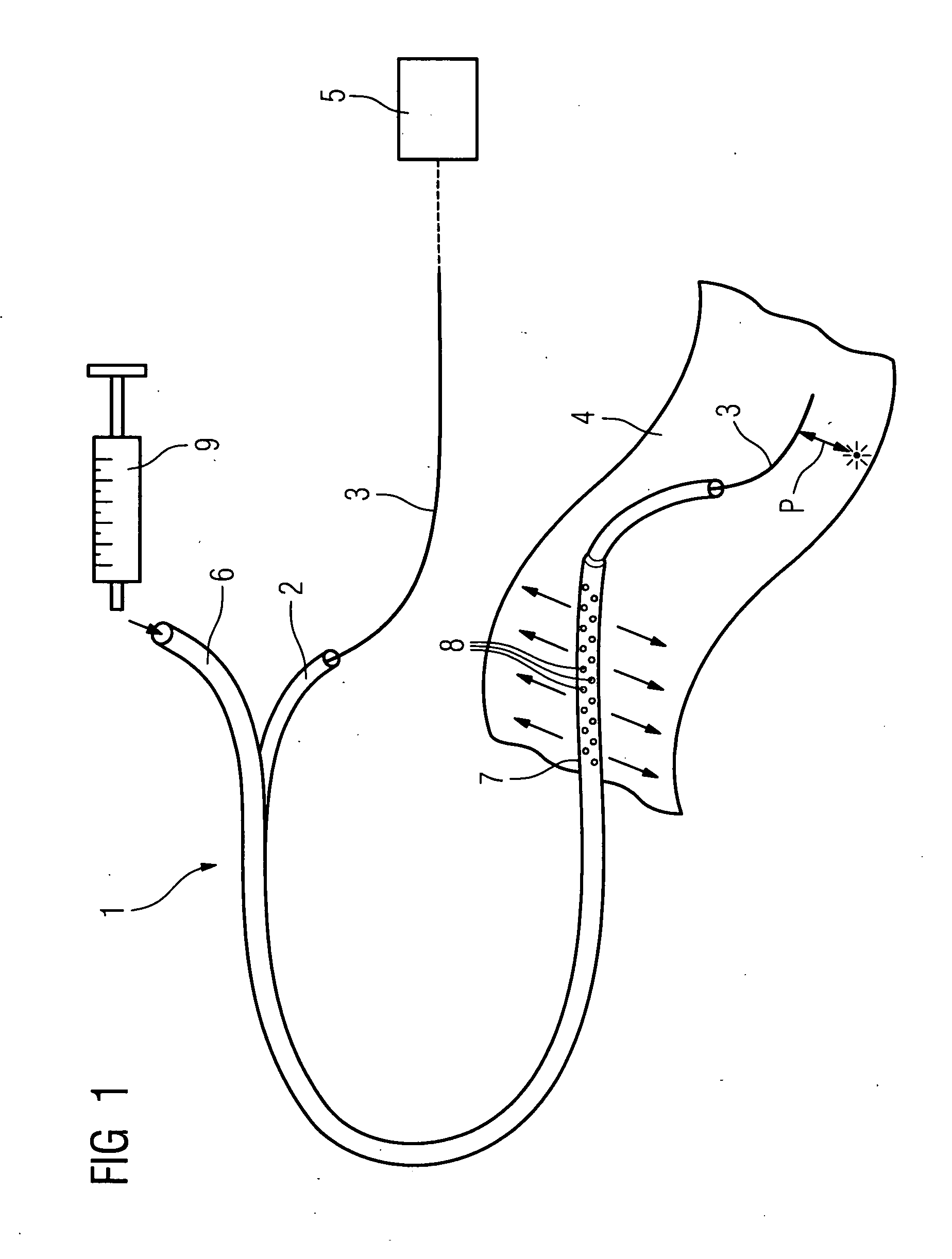
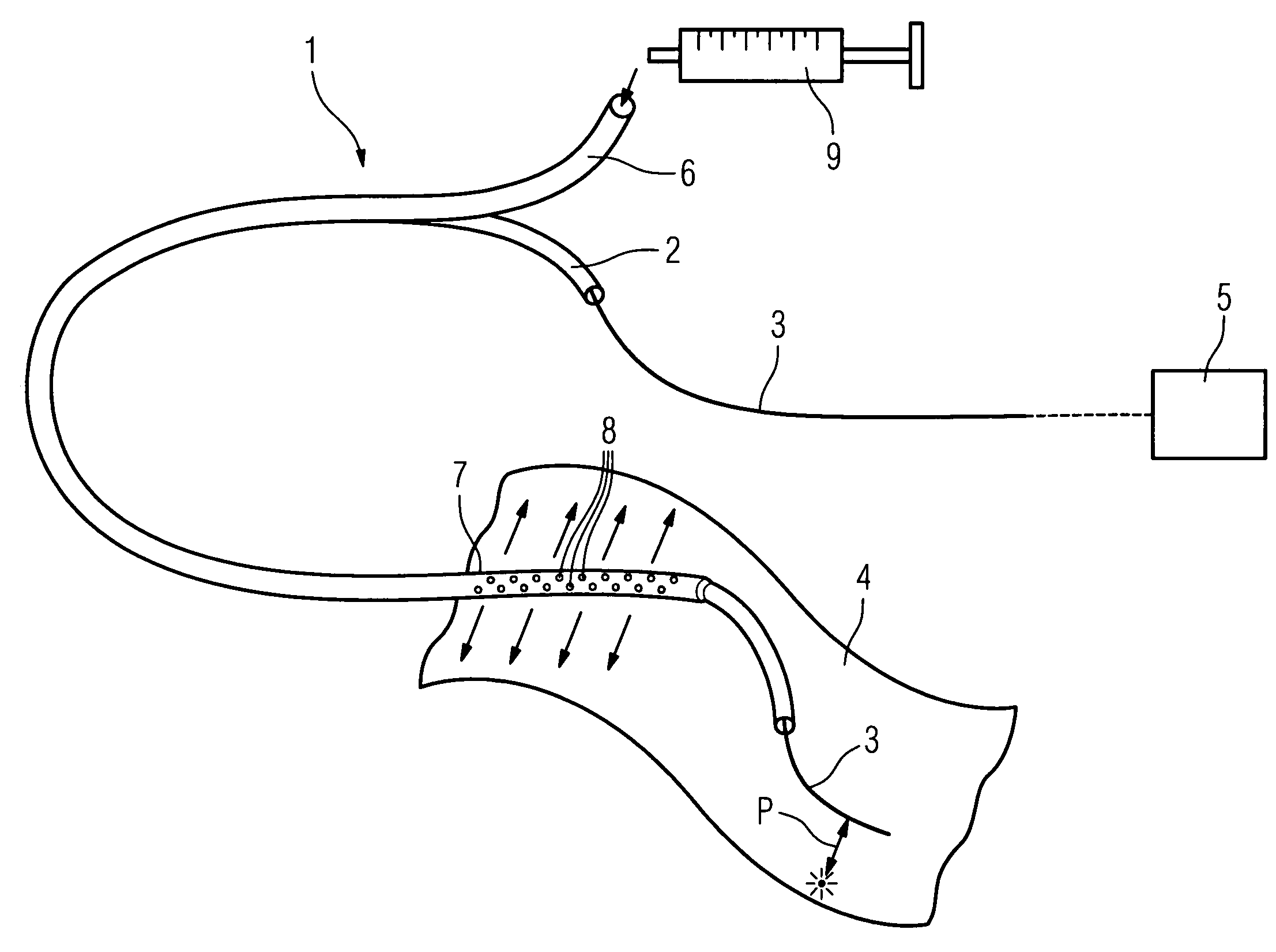
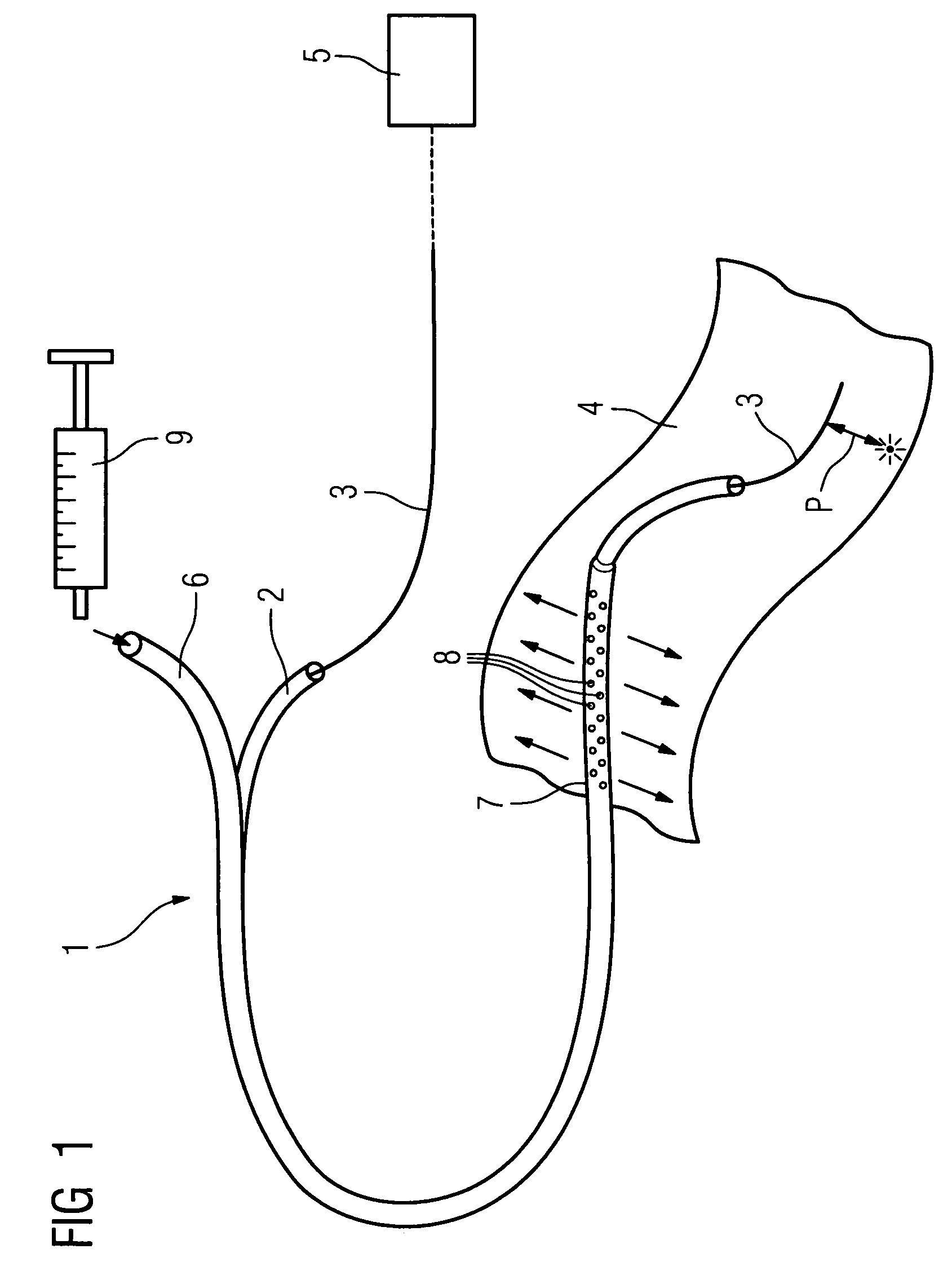
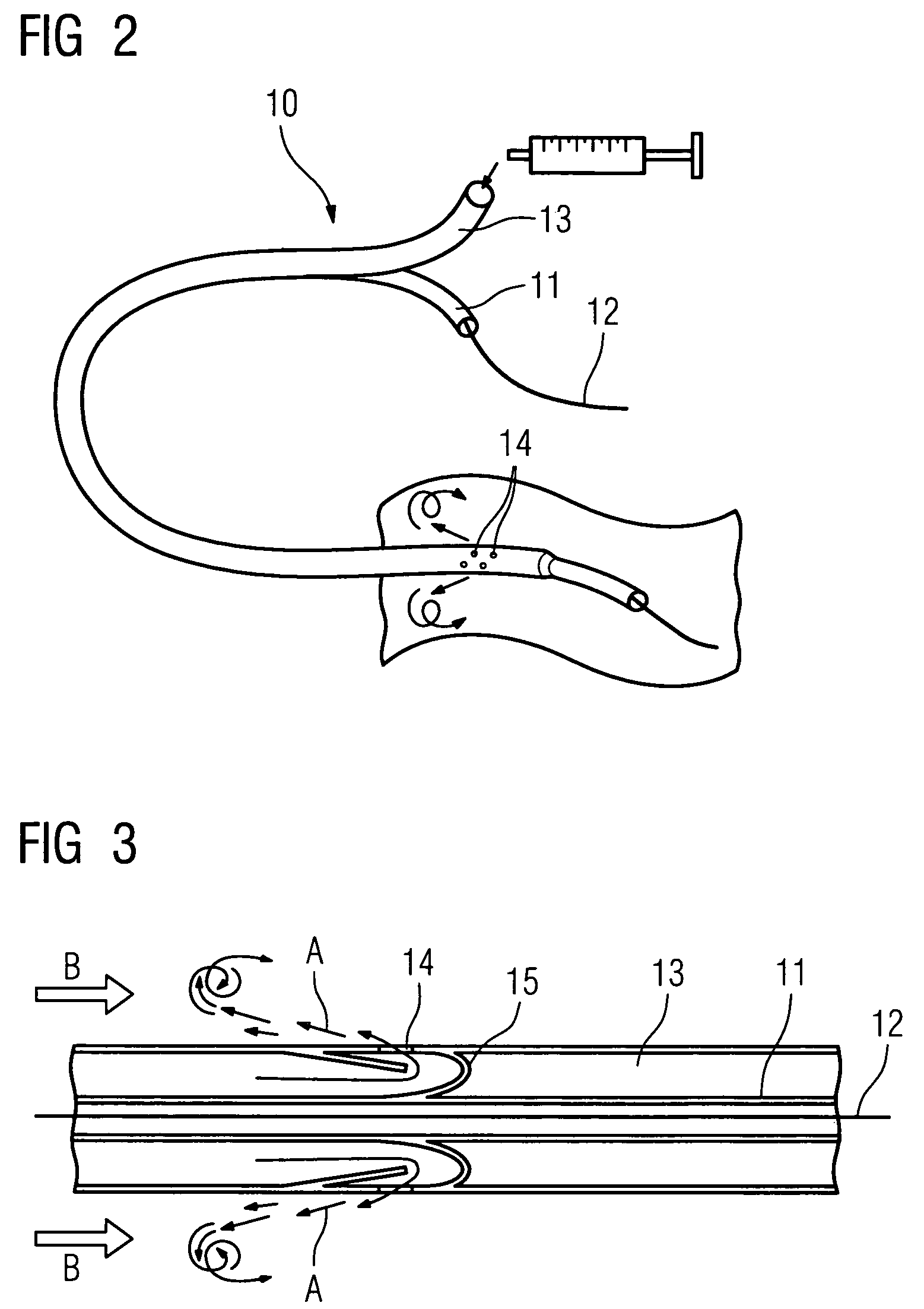
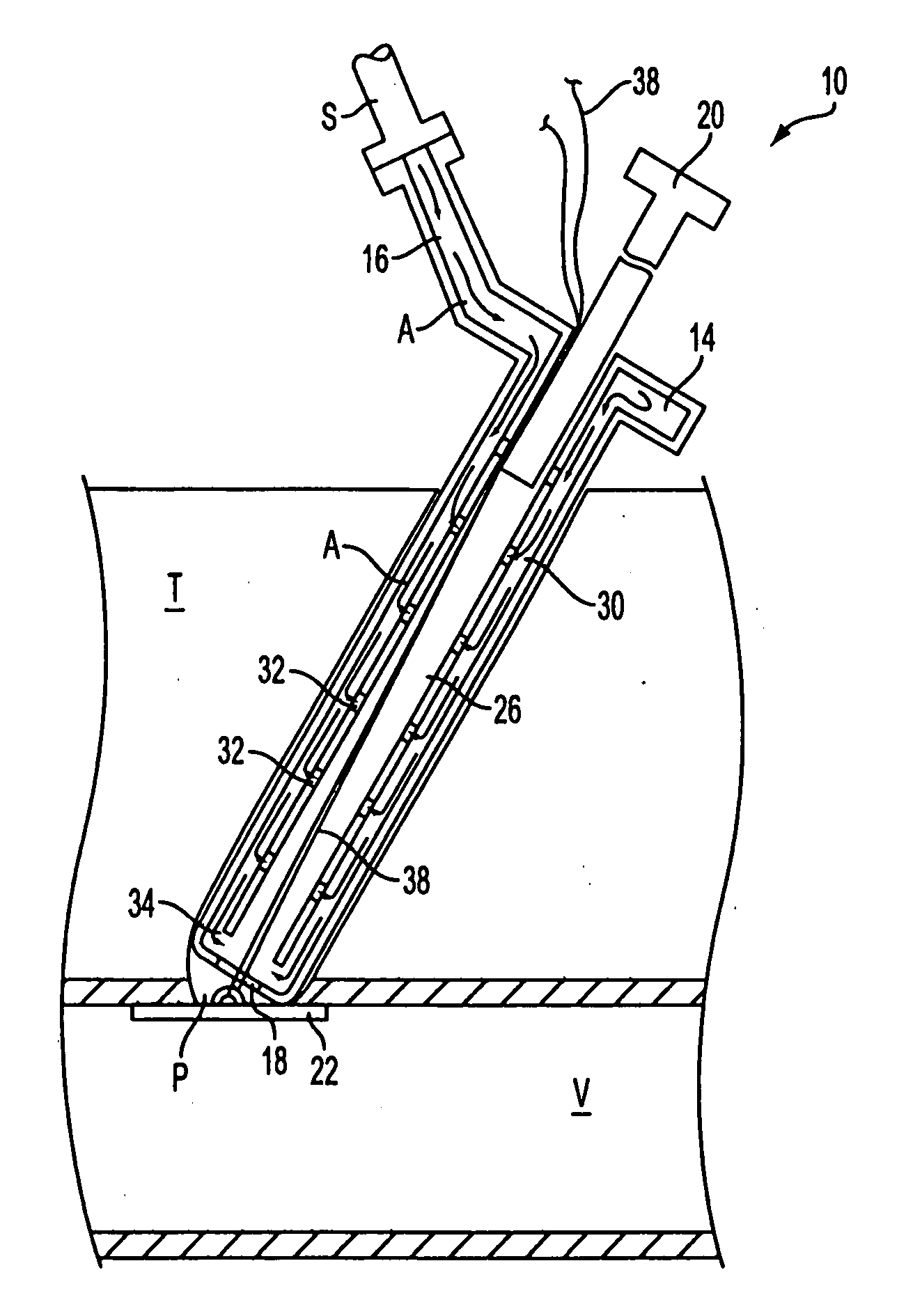
Method of recording two-dimensional images of a perfused blood vessel
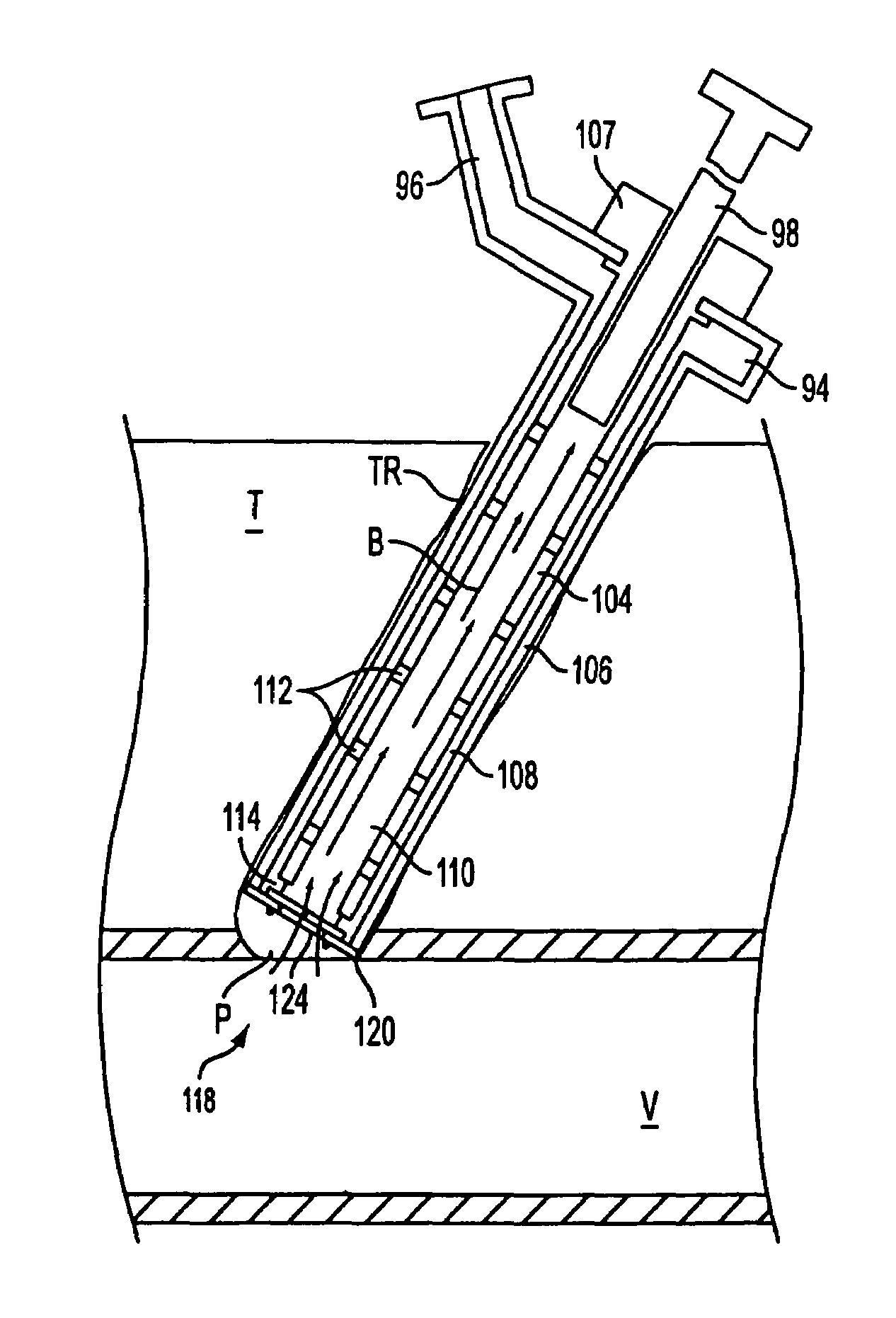
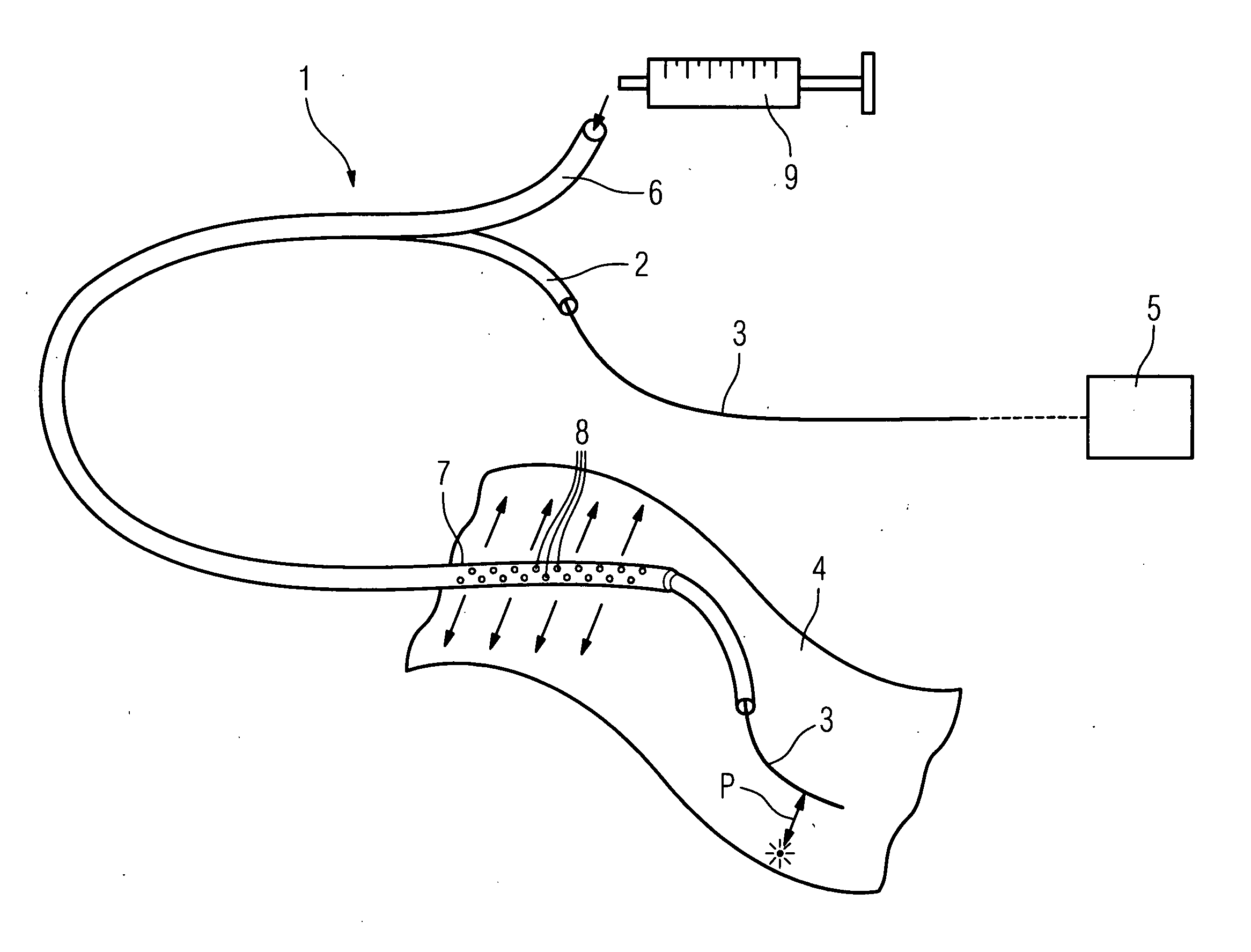
The invention relates to a method for recording two-dimensional images on the interior of a perfused blood vessel by means of optical coherence tomography using a catheter with an integrated imaging device, via which light is radiated and light reflected from the vascular wall is recorded, whereby a fluid containing a medium having a higher refractive index than blood plasma and / or cytoplasm is injected, via the catheter, into the vascular area to be recorded, to enable imaging to be carried out, whereby said fluid is a mixture of autologous blood and the medium.
Owner:SIEMENS HEALTHCARE GMBH
Medical material handling systems
Disclosed herein is a system for the collection, treatment and delivery of an autologous blood sample, comprising a first syringe having a first body portion. A first sample transfer portion has a first syringe inlet for drawing an untreated blood sample from a patient and a first syringe outlet for dispensing the untreated blood sample. A blood sample treatment chamber having a chamber inlet, the first syringe outlet being operable to establish a dedicated first fluid coupling with the chamber inlet to dispense the untreated blood sample to the blood sample treatment chamber. The blood sample treatment chamber having a chamber outlet for dispensing a treated blood sample following treatment. The second syringe has a second body portion and a second sample transfer portion, the second sample transfer portion having a passage with a first access location which is operable to form a dedicated second fluid coupling with the chamber outlet. The second body portion has a second syringe outlet, the passage having a second access location for fluid communication with the second syringe outlet, releasable lock means for forming a locked third fluid coupling between the second access location and the second syringe outlet. The lock means is operable in response to a release signal to release the third fluid coupling, the second syringe outlet being operable when released from the third fluid coupling to form a fourth fluid coupling with a blood sample delivery unit.
Owner:VASOGEN IRELAND LTD
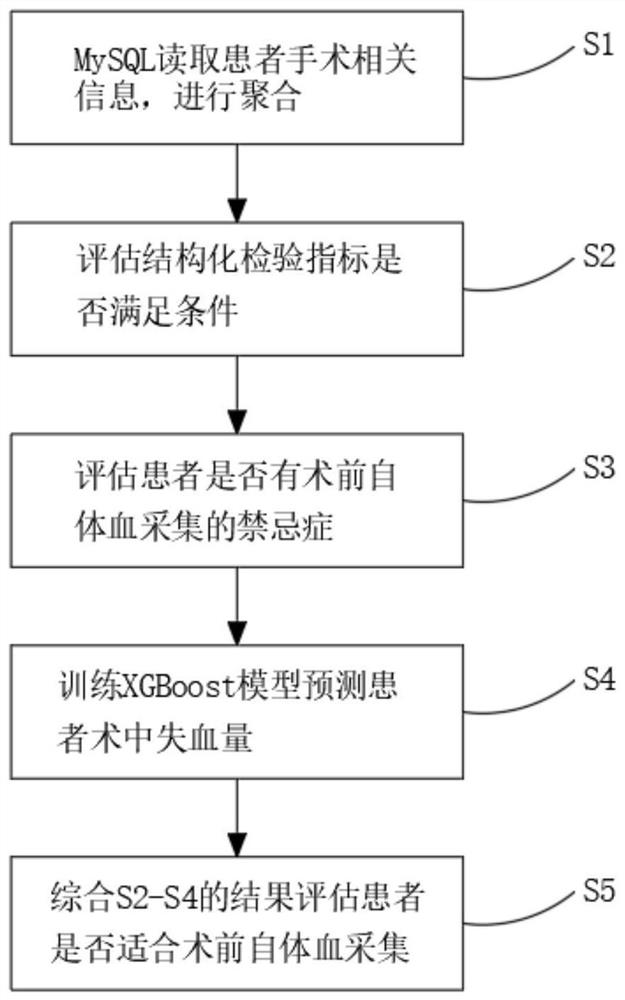
Detection and evaluation method applicable to blood management of perioperative patients
ActiveCN111724910AImprove work efficiencyAvoid callingMedical simulationMedical data miningDiseaseBlood coagulations
The invention relates to the technical field of medical treatment, and discloses a detection and evaluation method applicable to blood management of perioperative patients. The method comprises the following specific steps: S1, reading admission records, nursing documents, blood routine examination results and blood coagulation and biochemical information of a patient from a MySQL database, and conducting aggregating; and S2, evaluating whether the structured test indexes of the patient meet requirements on preoperative autologous blood collection or not. According to the invention, a doctor in a blood transfusion department can be helped to understand the blood use law of a single disease in each department of the hospital, and the doctor can be helped to formulate an optimal blood transfusion path through optimal postoperative hemoglobin analysis; clinical doctors can know the conditions of anemia and blood coagulation functions of the patient before an operation, and timely and pertinently treat the patient to ensure smooth operation in the operation, and rehabilitation of the patient can be accelerated; a doctor is helped to know an autologous blood collection scheme and intraoperative recovery feasibility in advance, the use probability of allogeneic blood is greatly reduced, and the risk of various adverse reactions is reduced.
Owner:北京和兴创联健康科技有限公司
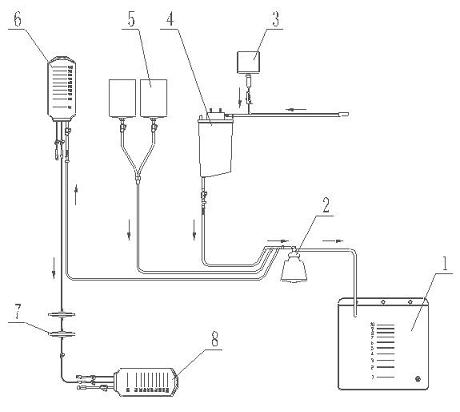
Blood processing system of autologous blood collection and method for processing blood by employing same
ActiveCN103055365AReduce the risk of infectionReduce adverse reactionsOther blood circulation devicesWhite blood cellTurbidity
The invention discloses a blood processing system of autologous blood collection and a method for processing blood by employing the same. The method includes that one end of a whole blood channel is communicated with a blood-taking needle, the other end of the whole blood channel is communicated with a centrifugal machine, a blood-separating outlet pipe is arranged on the other side of the centrifugal machine, and the blood-separating outlet pipe is provided with a turbidity detector. A platelet-collecting bag is communicated with the turbidity detector through a platelet-collecting tube, a plasma-collecting bag is communicated with the platelet-collecting tube through a plasma-collecting tube, and the platelet-collecting bag is communicated with the whole blood channel between a pump A and an anticoagulant tube through the plasma-collecting bag. The method is convenient in operation, safe and reliable. Red blood cells and platelets can be collected simultaneously, and therefore complicated separation of blood cells for a single purpose is decreased. By performing autologous blood collection and supplementing displacement liquid for a patient during the course of collection, the balance of intake and output quantity by the patient is guaranteed. Compared with the prior art, the blood processing system of autologous blood collection is more secure, and cost of medical blood transfusion is greatly reduced.
Owner:FIRST HOSPITAL AFFILIATED TO GENERAL HOSPITAL OF PLA
Human skin filler for injection and its prepn process
The human skin filler for injection consists of cell components, blood plasma with rich thrombocyte, blood coagulation factor and calcium ion. Each milliliter of the filler for injection contains cell components in (10-50)x10<6>, thrombocyte in (100-700)x10<6>, blood coagulation factor in 1-10 U, and calcium ion in 10-40 mmol. The cell components are at least one of corium stem fibroblast, fibroblast precursor and fibroblast obtained through acquiring the skin from the patient, extracorporeal no-serum culturing and amplifying, and collecting; the thrombocyte is extracted from the autoblood of the patient; and the blood coagulation factor is autogenous or extraneous. The present invention also provides preparation process of human soft tissue filler for injection. The present invention has the advantages of quick obvious filling and repairing effect, last curative effect, high safety, etc.
Owner:北京赛尔泰和生物医药科技有限公司
Improved expansion culture medium for regulatory T cells of human cord blood origin and application method of expansion culture medium
ActiveCN104357389AReduce riskPromote growth rateBlood/immune system cellsCulture mediumsClinical disease
The invention relates to an improved expansion culture medium for regulatory T cells of human cord blood origin and an application method of the expansion culture medium. According to the expansion culture medium, heparin anticoagulated autologous cord blood plasma accounting for 10%-12% of the volume of a culture medium, CD3-CD28 antibody co-expressed immunomagnetic beads, recombinant human interleukin 2, 2-mercaptoethanol, rapamycin, HEPES (4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid) and gentamicin are added into the RPMI (Roswell Park Memorial Institute)1640 culture medium; and then separated cell suspension is inoculated in a 96-well plates with a U-shaped bottom, hole-division expansion can be performed every 1-2 days, and the expansion period is 3-4 weeks. All reagents in the culture system reach the GMP (good manufacturing practice) level or are originated from autologous cord blood, so that risks caused by ingredients of animal origin are avoided, and the regulatory T cells can be used for a third-party unrelated donor and directly applied to clinical disease treatment; and compared with a traditional culture system, Treg cells (the regulatory T cells) expanded by the improved culture medium is excellent in aspects of growth speed, purity, activity, lymphocyte inhibition function and the like, and the Treg cells are expected to be used as the regulatory T cells of the third-party unrelated donor and applied to the clinical disease treatment.
Owner:HUNAN XENO LIFE SCI
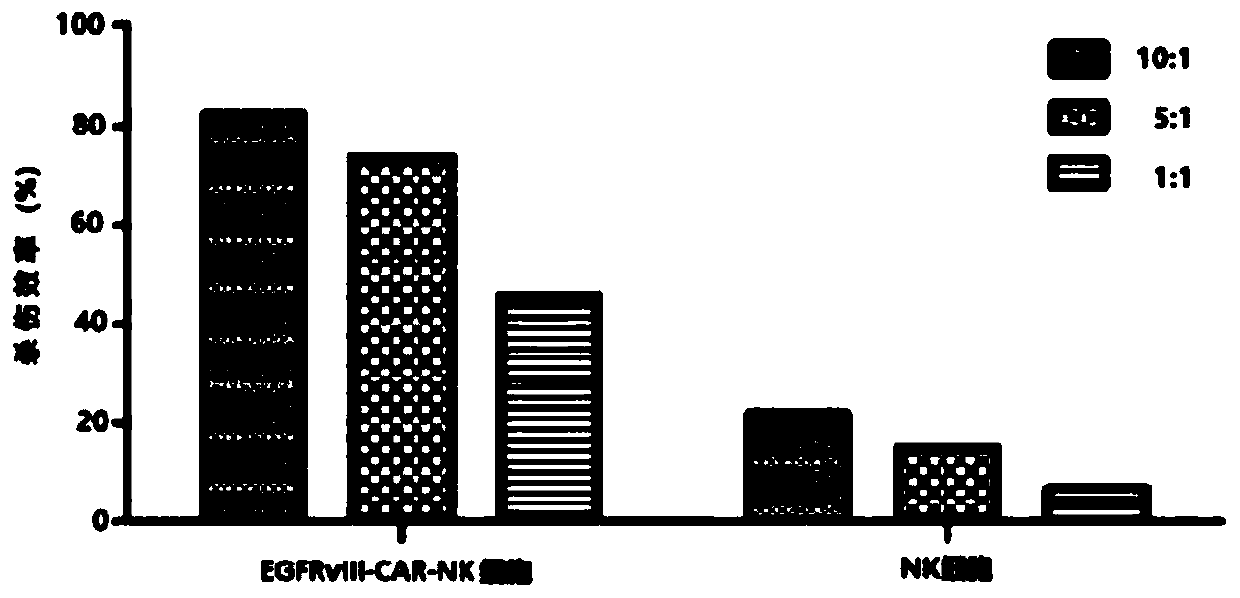
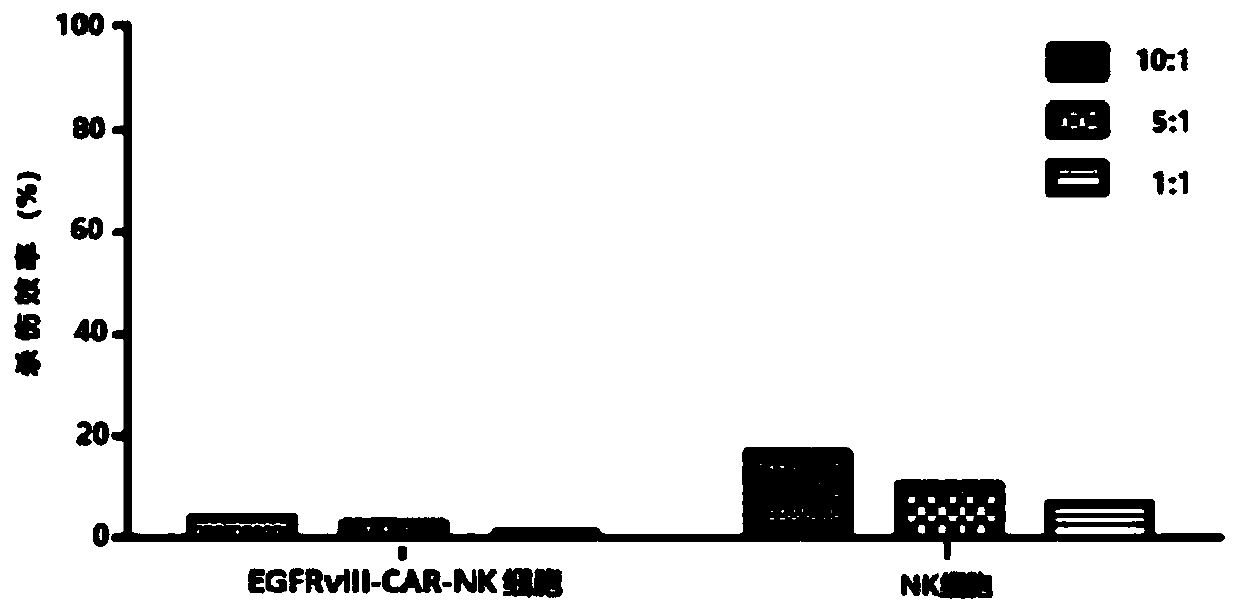
CAR-NK cell, and preparation method and application thereof
ActiveCN109536455ASolve some problemsTumor killerImmunoglobulin superfamilyGenetically modified cellsCell activityAutologous blood
The invention discloses a CAR-NK cell, and a preparation method and application thereof, and belongs to the field of cell therapy and biopharmaceutics. The method uses umbilical cord blood as a sourceof NK cells, and solves the problems of CAR-NK cells from a NK cell line, peripheral blood and autologous blood, such as complicated operation, risk of tumor formation, insufficient cell activity, asmall number of cells and GVHD caused by partial allogeneic origin cells.
Owner:WUHAN BIO RAID BIOTECH CO LTD
Autologous whole blood homogenate compound for reconstruction of autologous skin epidermis protective screen
InactiveCN102188362APromote proliferationEasy to synthesizeCosmetic preparationsPeptide/protein ingredientsAutologous Whole BloodAutografting skin
The invention discloses an autologous whole blood homogenate compound for reconstruction of an autologous skin epidermis protective screen. After the autologous whole blood is homogenized, under the synergetic effects of multiple bioactive substances and cell growth factors released by the autologous whole blood, the autoblood is effectively applied to epidermis repair and reconstruction of the skin protective screen, thereby giving better play to the maximum value and biological effects of the autoblood in autologous application; and because the autologous whole blood homogenate liquid plays a mutually cooperative protective effect with other auxiliary components and strengthens the compounding effect of the cell factors in epidermis reconstruction, the autologous whole blood homogenate liquid compound has good effects on epidermis repair and functional reconstruction of the epidermis protective screen, can improve the functional problems of the human skin protective screen in multiple aspects at different levels, restores the health of skin, and basically solves the functional problems of the epidermis protective screen. In addition, the preparation method disclosed by the invention is simple, convenient to operate, easy to learn and popularize, and suitable for medical cosmetology institutions with legal qualification for medical cosmetology.
Owner:董萍 +1
Compositions for repairing bone and methods for preparing and using such compositions
InactiveUS20060051427A1Large specific surface areaImprove propertiesPowder deliverySurgical adhesivesAutologous bloodImproved method
This invention relates to an improved method of preparing an implantable gel or paste for placement between injured bones or placement in bony voids to induce regeneration, and the compositions produced thereby. Specifically, mineral, ceramic, or processed bone particles are coated with a high molecular weight polymer capable of forming a viscous gel when reconstituted with water, saline, autologous blood, sera, or other medically acceptable solution. This high molecular weight polymer coating material may be a natural or synthetic polymeric material, producing a wettable gel upon exposure to water, saline, or another solution. In storage, the composition will be granular and dry but easily wetted. In use, the material is reconstituted to a viscous malleable paste by the simple addition of water or other medically acceptable solution without the need for aggressive mixing. The paste may be delivered by syringe or manually deposited yet will be resistant to lavage or to displacement by gravity induced flow.
Owner:NANOTHERAPEUTICS INC
Verification method and system for medical treatment
A system for the collection, treatment and delivery of an autologous blood sample, comprising a first syringe for drawing an untreated blood sample from a patient, a blood sample treatment chamber having a chamber inlet for receiving untreated blood from the first syringe and chamber outlet for passage of treated blood to a second syringe coupled thereto. The second syringe includes a releasable lock means for allowing discharge of the treated blood to the patient in response to a release signal. The release signal is issued following a positive outcome from a verification process dependent upon temporal data from certain events in the collection, treatment and delivery of the blood sample, and identity data of the patient and the second syringe with the treated blood.
Owner:VASOGEN IRELAND LTD
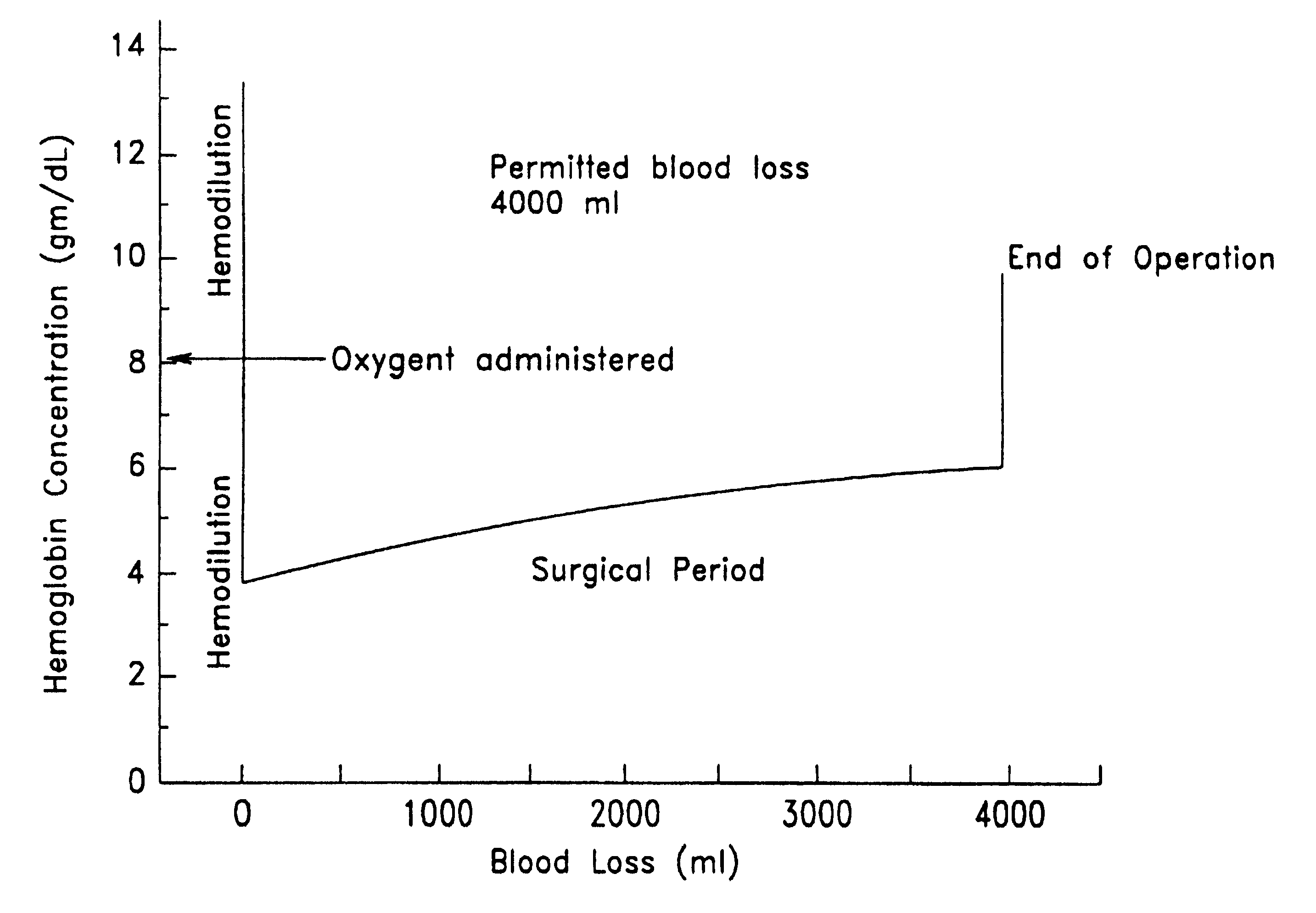
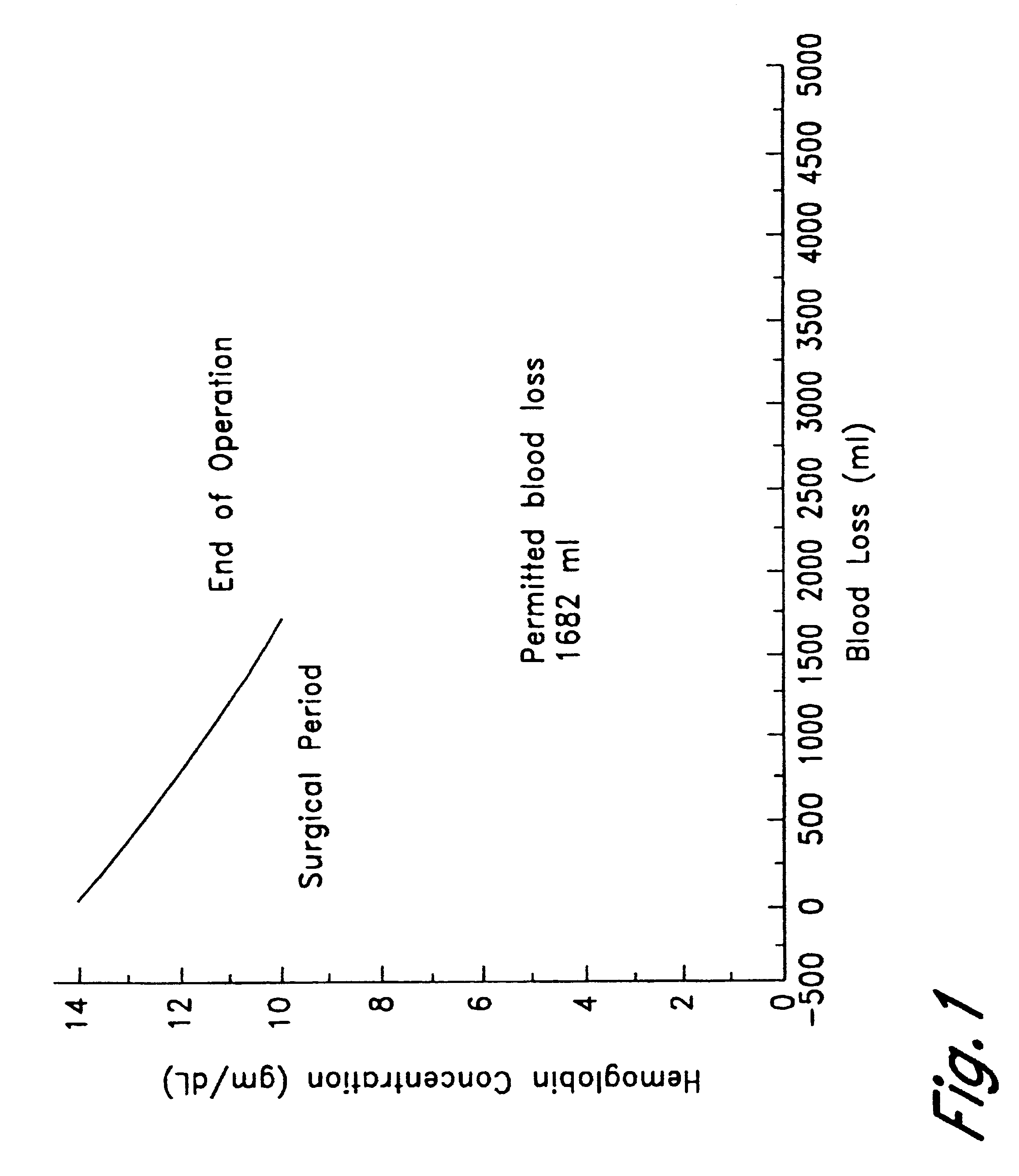
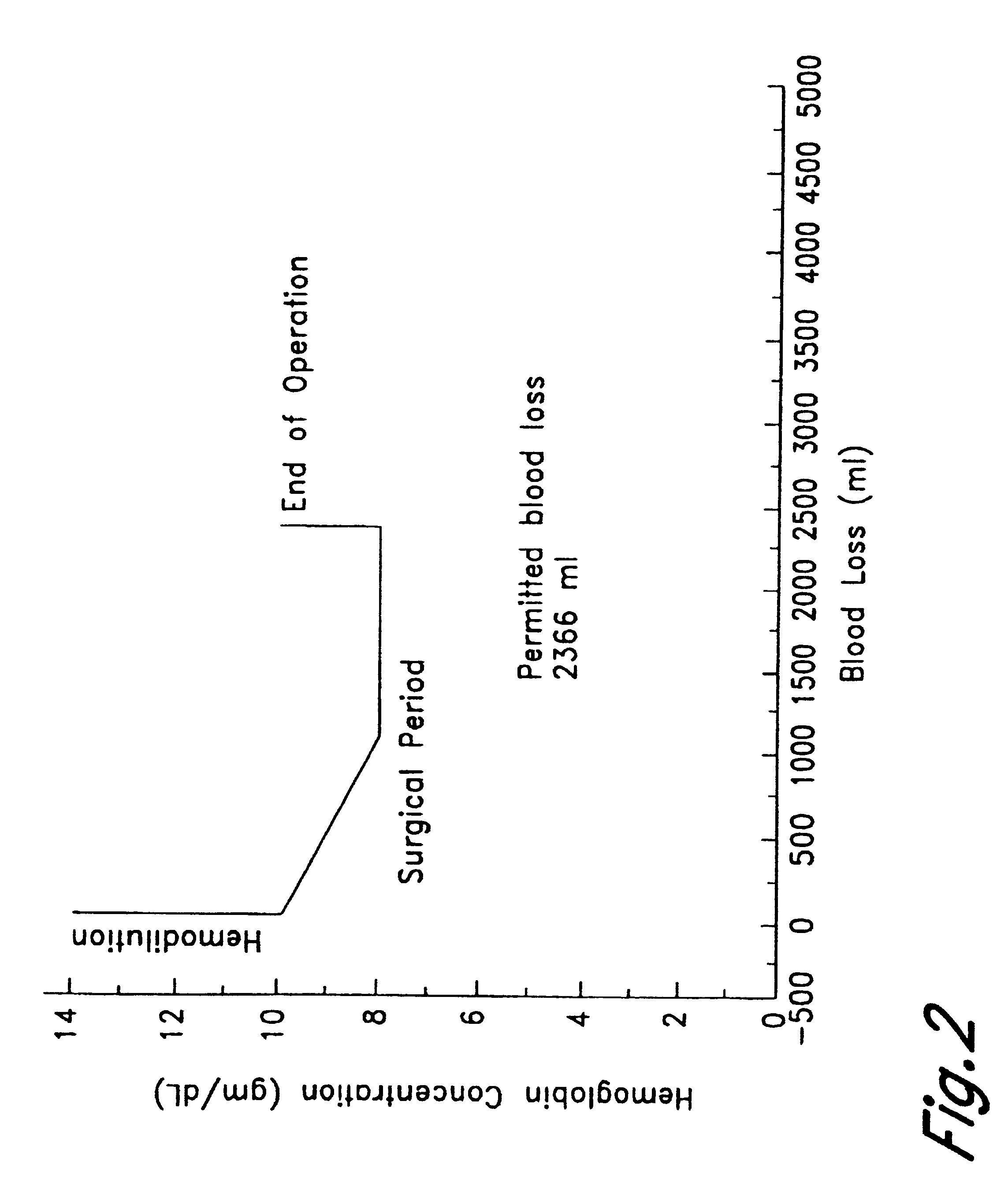
Method of hemodilution facilitated by monitoring oxygenation status
InactiveUSRE38081E1Reduce decreaseMaintain oxygenationHalogenated hydrocarbon active ingredientsPeptide/protein ingredientsHemodilutionsAutologous blood
A method for hemodiluting a patient is disclosed which includes the steps of administering a biocompatible oxygen carrier in conjunction with surgery or organ ischemia or infarct while assessing the patient's PvO2 or other oxygenation indices, and administering to the patient additional oxygen carrier or autologous blood in response to the PvO2 value if necessary to maintain the PvO2 at or above the desired level.
Owner:PFC THERAPEUTICS L L C
Exosome freeze-drying powder, preparation method of exosome freeze-drying powder and preparation comprising exosome freeze-drying powder
ActiveCN110279893ALong lastingGood filling effectPharmaceutical delivery mechanismProsthesisMedicineCentrifugation
The invention discloses exosome freeze-drying powder, a preparation method of the exosome freeze-drying powder and a preparation comprising the exosome freeze-drying powder. The preparation method of the exosome freeze-drying powder comprises the following steps of: preparing platelet factor-rich plasma: collecting autologous blood for centrifugation, preparing exosome: collecting autologous fat to prepare fat mesenchymal stem cells, and culturing the mesenchymal stem cells, preparing a freeze-drying liquid: mixing the platelet factor-rich plasma and the exosome and adding trehalose for mixing, and performing freeze-drying: performing freeze-drying on the freeze-drying solution. The preparation comprises the exosome freeze-drying powder, hyaluronic acid and saline. The preparation can effectively solve the problems that the existing cosmetic preparation has a poor filling effect, short maintenance time and high immunological rejection response.
Owner:CHENGDU QINGKE BIOTECH
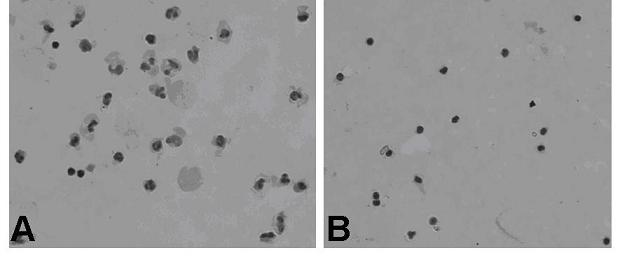
Method for recovering intraoperative autoblood
InactiveCN102430163ANumber of protectionProtective functionOther blood circulation devicesCell saverAutologous blood
The invention provides a method for recovering intraoperative autoblood. Intraoperative autoblood is recovered by using a blood recovering machine; an MAP (Mannitol-Adenine-Phosphate) erythrocyte preserving fluid is taken as a cleaning solution; an anti-freezing liquid is prepared from the MAP erythrocyte preserving fluid and sodium heparin in the ratio of 1,000ml:(200-400mg); and blood treated by using the blood recovering machine is stored in a blood bag after being filtered with an acetic ester non-woven fabric filter film which has the pore diameter of 11-20 mum and is formed by laminating 7-40 layers. Compared with the prior art, the method for recovering intranperative autoblood disclosed by the invention has the advantages that: karyocytes including tumor cells in blood are removed, the erythrocyte quantity and function are protected to the maximum extent, and the quality of the recovered blood is ensured.
Owner:WEST CHINA HOSPITAL SICHUAN UNIV +1
Methods and Apparatus for Preparing Autologous Blood Eye Drops
ActiveUS20200337946A1Improve sterilitySufficient materialSurgical furniturePharmaceutical containersKit deviceAutologous blood
Methods and apparatus for preparing autologous blood serum eye drops within a potentially contaminating environment are disclosed. Key convenience kit apparatus providing novel methodology includes a plastic bag in which an innovative tray securely holds a plurality of empty eye drop bottles and associated caps disposed and sealed therein. Providing the only access pathway into the bag is a sterilizing filter appliance which filters all fluid entering the bag and provides a nozzle for dispensing into bottles disposed in the tray. The key convenience kit apparatus is sterilized prior to use, assuring all components disposed within the bag are within a desired sterility assurance level. The sterilized state of items associated with the key apparatus is maintained by the single pathway for fluid. Capping and sealing the bottles before opening the bag permits delivery of sterile product for use outside the bag.
Owner:THORNE INTPROP HLDG LLC
Method of recording two-dimensional images of a perfused blood vessel
InactiveUS7794446B2Sufficient supplySurgeryMaterial analysis by optical meansRefractive indexAutologous blood
Owner:SIEMENS HEALTHCARE GMBH
Autologous wound sealing apparatus
InactiveUS20060155330A1Easy to useReduce contaminationSuture equipmentsSurgical veterinaryAutologous bloodBlood vessel
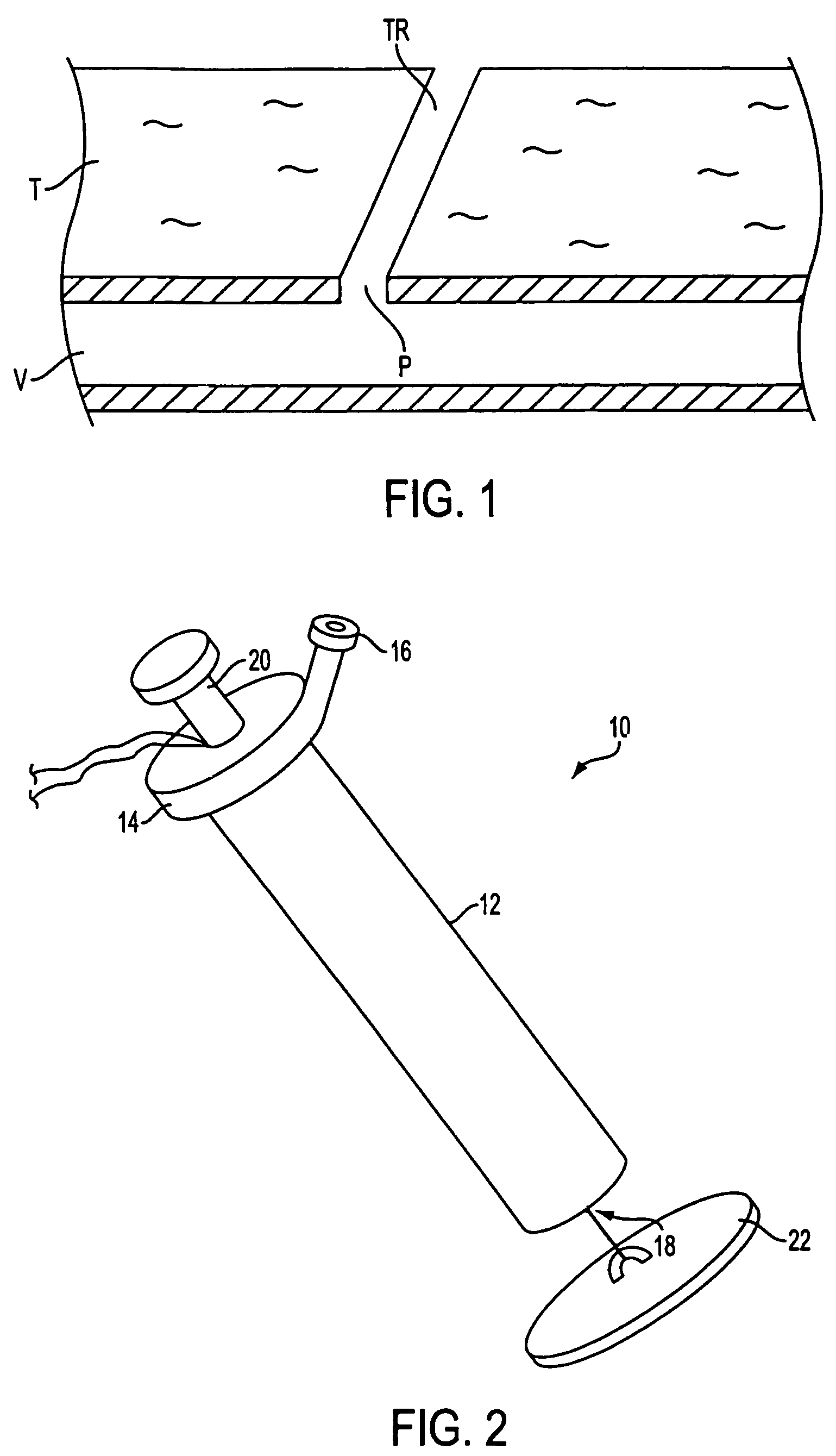
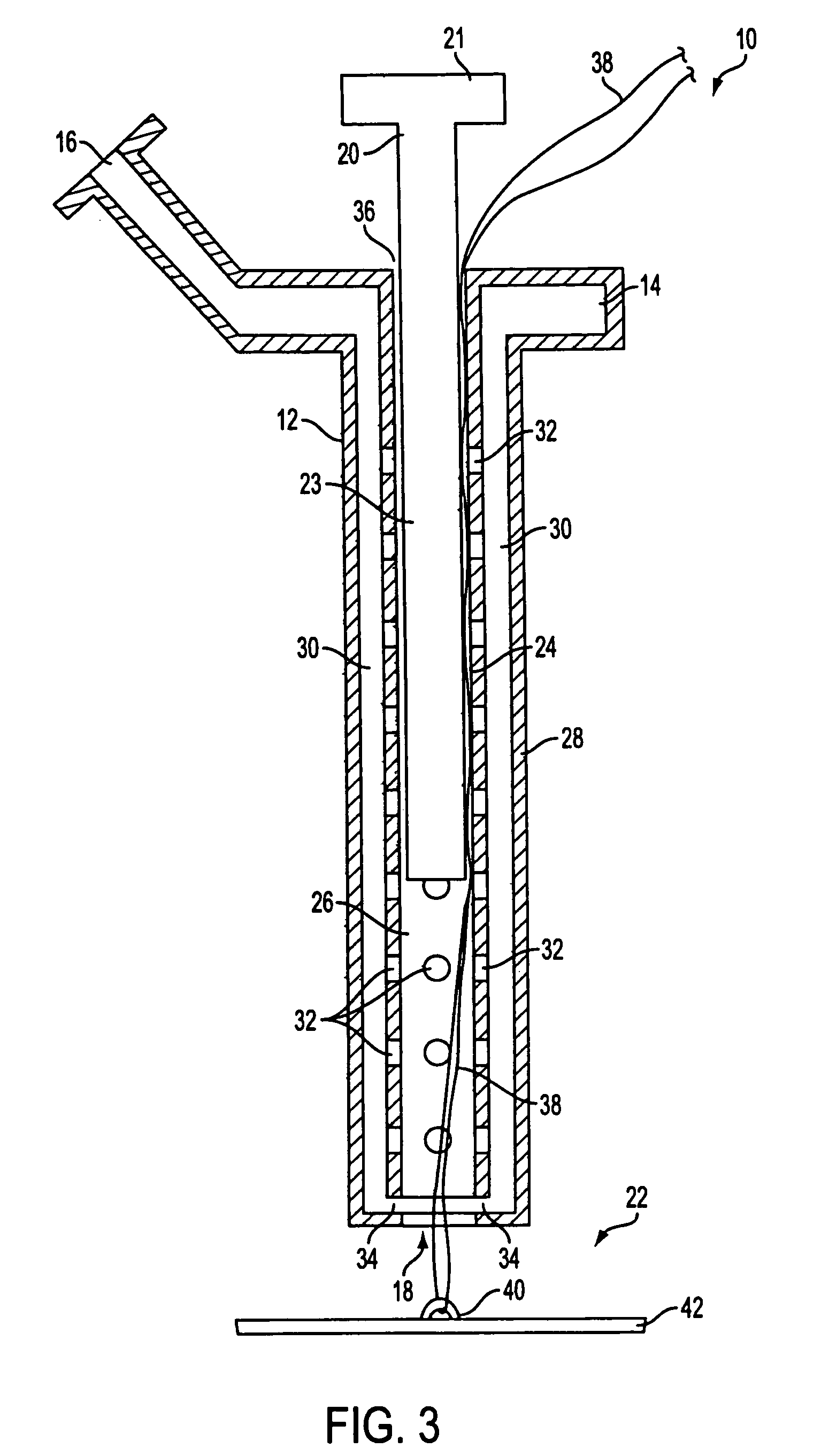
Apparatus (10) is provided for sealing a vascular puncture tract by forming the autologous plug within the puncture tract, and extruding that plug into the puncture tract. The apparatus of the present invention forms an autologous blood plug by drawing blood into the apparatus from a vessel, mixing a blood congealing agent with the drawn blood, and ejecting a plug formed from the clotted blood within the puncture tract. Also provided are various closure elements (22) to isolate the drawn blood from the vessel during mixture with the blood congealing agent, and to facilitate placement of the apparatus relative to the vessel.
Owner:ABBOTT LAB VASCULAR ENTERPRISE
Enhanced autologous growth factor production and delivery system
ActiveUS9205110B2Dead animal preservationMammal material medical ingredientsActive proteinBlood plasma
Systems and methods for producing enhanced blood serum with concentrated growth factors and / or prophylactically and therapeutically active proteins. An autologous blood product is obtained from a donor and then incubated into a containment device (sterile container) that includes a source of cells or tissue (for example, autologous, allogeneic, or xenographic tissue, or combinations thereof). The cells from the injected fluid (autologous blood product) interact / incubate with the source of cells or tissue inside the containment device, to produce a serum that is loaded with autologous proteins, growth factors, and cytokines. The autologous blood product may be a fluid and / or composition that includes blood, whole blood, autologous conditioned plasma, platelet-rich plasma, platelet-poor plasma, bone marrow aspirate, bone marrow concentrate and stem cells, among others, and combinations thereof.
Owner:ARTHREX
Hemo-concentrator system for autologous blood recovery
InactiveUS7033334B2Quickly and easily recoveredFast stabilizationOther blood circulation devicesHaemofiltrationRecovery methodEngineering
The invention is directed to a blood bag system, comprising a closed, sterile bag of substantially transparent, bio-compatible material, defining upper and lower ends; an infusion port at the upper end of the bag; an outlet port at the lower end of the bag; an inlet port at the lower end of the bag; a hemo-concentrator having an inlet port and an outlet port; a pair of inlet tubes fluidly connected to the hemo-concentrator inlet port; and a pair of outlet tubes fluidly connected to the hemo-concentrator outlet port. One of the inlet tubes is fluidly connected to the outlet port of the bag, and one of the outlet tubes fluidly connected to the inlet port of the bag. The other of the inlet tubes and the other of the outlet tubes are temporarily sealed (e.g., clamped) to prevent flow therethrough. The bag, the one outlet tube, the hemo-concentrator, and the one inlet tube thus form a closed fluid circulation circuit by which a pump can recycle the blood through the hemo-concentrator. One aspect of the invention includes a hemo-concentrator system and a specially adapted tubing set, for easily and quickly converting the hemo-concentrator from conventional use to use in the blood recovery method. Other aspects of the invention are directed to the combination of tubing set with hemo-concentrator, and to the tubing set itself.
Owner:SAMOLYK KEITH A
Small-diameter artificial blood vessel based on freeze casting technology and preparation method thereof
ActiveCN108404219AHigh tensile strengthGood anticoagulant effectCoatingsProsthesisFreeze-dryingFreeze-casting
The invention belongs to the field of biomedical materials, and particularly relates to a small-diameter artificial blood vessel based on freeze casting technology and a preparation method thereof. The preparation method comprises the following steps: dissolving a natural polymer material in water to prepare a natural polymer solution, pouring the natural polymer solution into a mold, and inserting the mold into liquid nitrogen for freezing forming; and taking a frozen formed artificial blood vessel out of the mold for freezing drying, immersing the artificial blood vessel in a glutaraldehydesolution for cross-linking treatment, and finally evenly coating the surface of the artificial blood vessel with a synthetic polymer solution to obtain the small-diameter artificial blood vessel. Theinner layer of the small-diameter artificial blood vessel is the natural polymer material which has the characteristics of short degradation period and rapid promotion of autologous blood vessel regeneration, and the outer layer of the small-diameter artificial blood vessel is a synthetic polymer material which has certain mechanical properties, and prevents blood leakage; the inner surface of thesmall-diameter artificial blood vessel has a radial structure, and can promote the adhesion of endothelial cells on the surface of the material so as to promote the endothelialization of the artificial blood vessel, and the anticoagulant ability of the artificial blood vessel can be greatly improved.
Owner:HUAZHONG UNIV OF SCI & TECH
Features
- R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
Why Patsnap Eureka
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Social media
Patsnap Eureka Blog
Learn More Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com