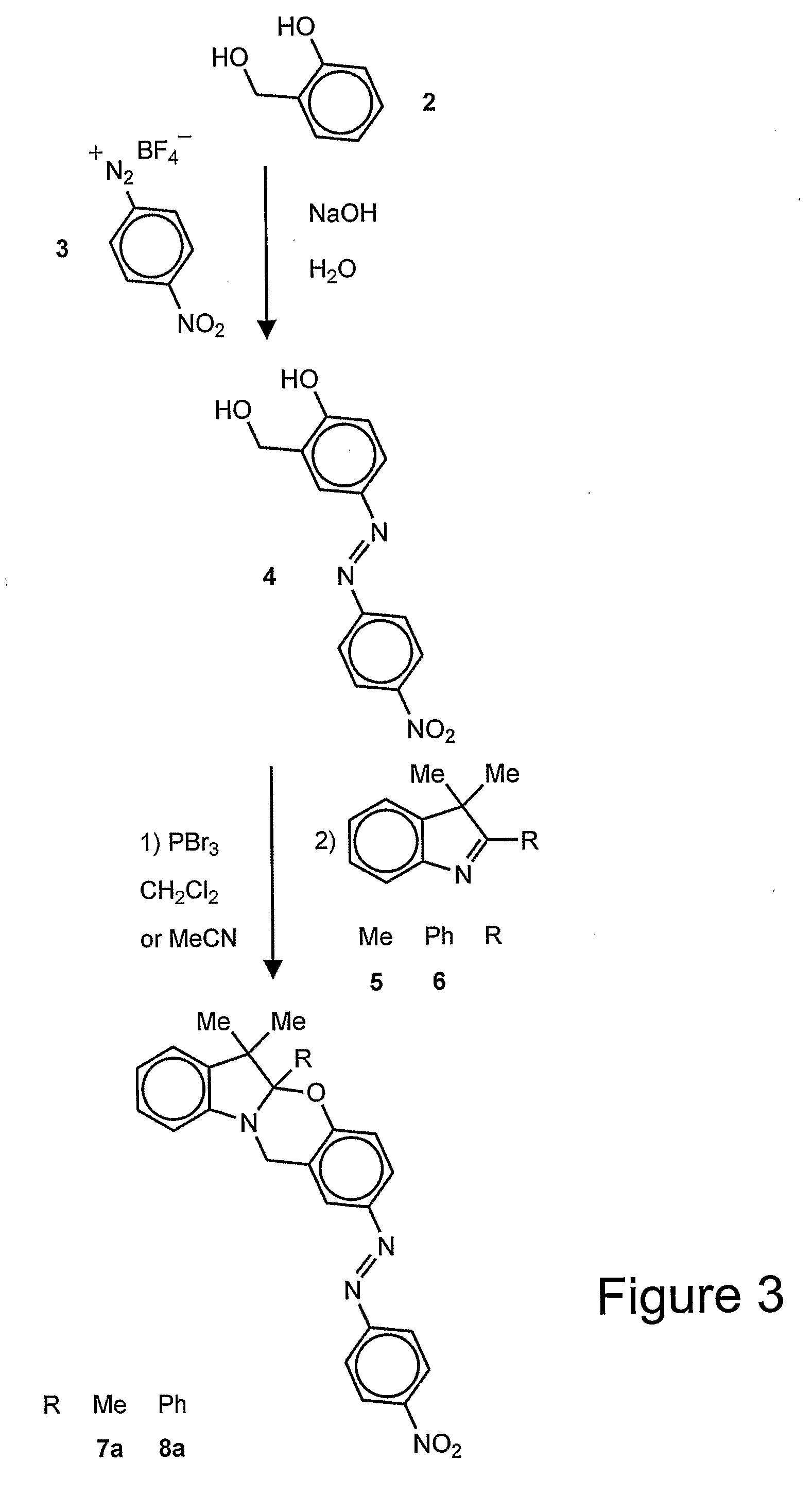
Patents
Literature
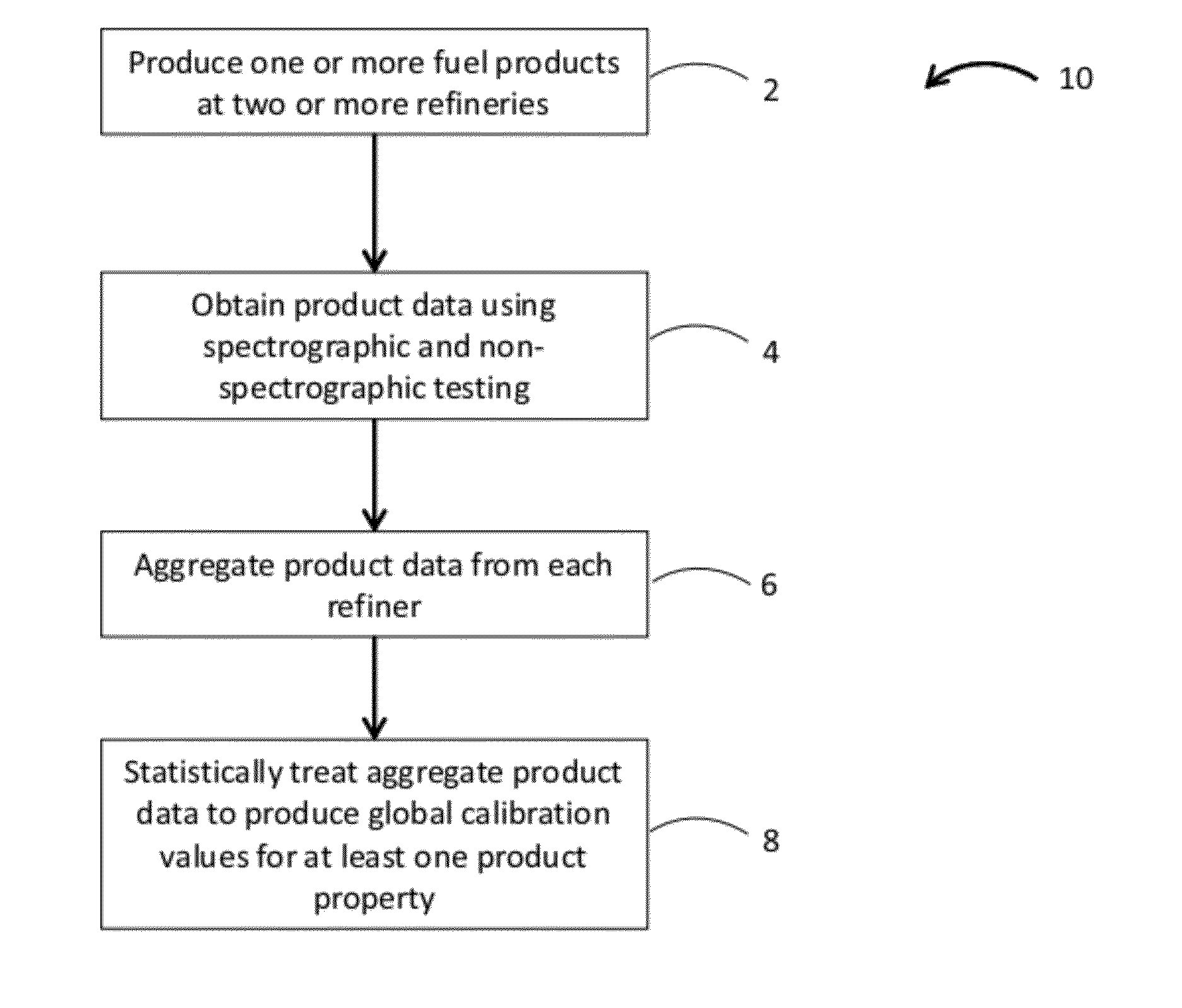
838results about "Testing organic contamination in water" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
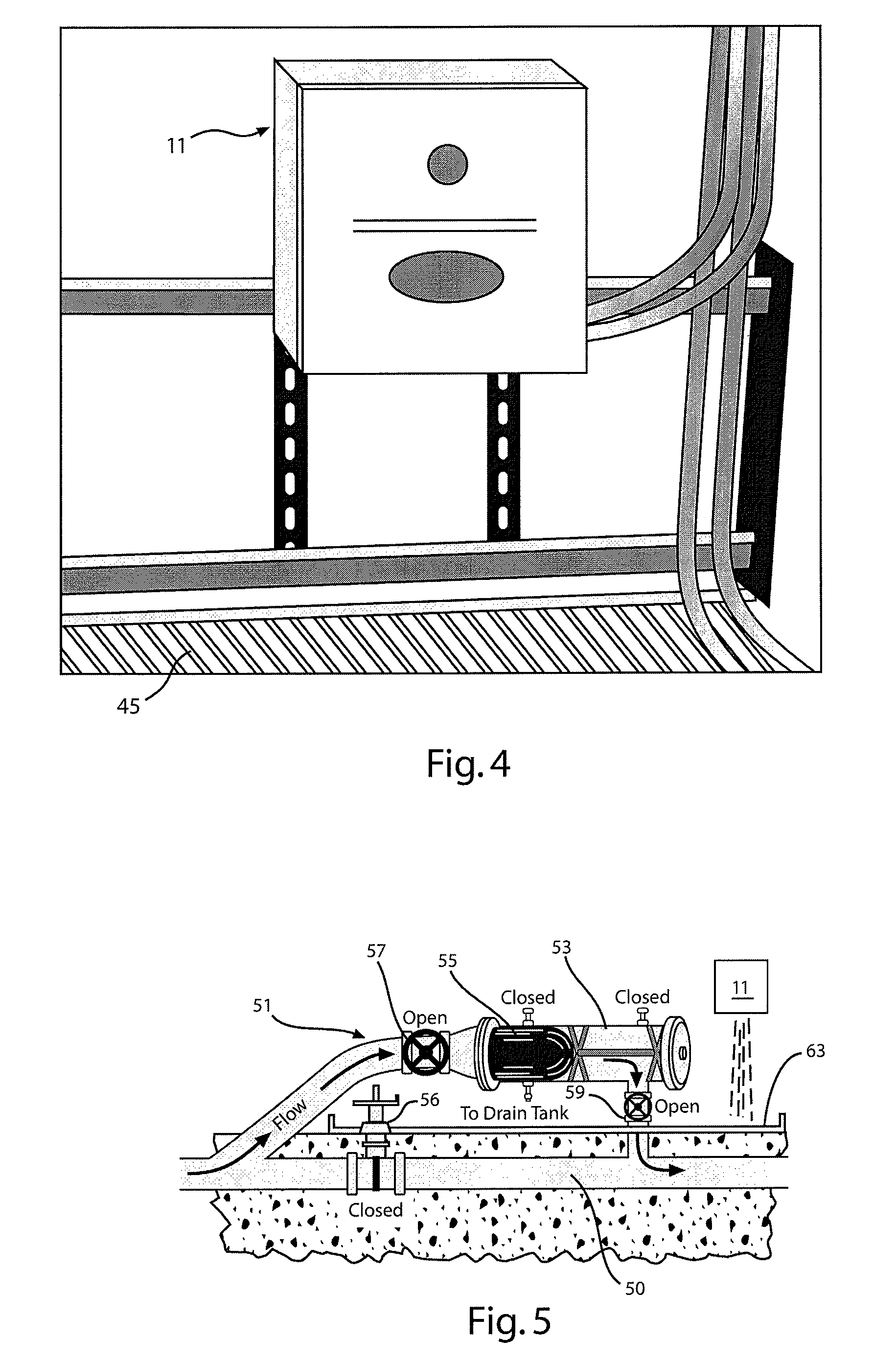
Remote monitoring system for water
InactiveUS20050009192A1Minimal disruptionFunction increaseWater treatment parameter controlControlling ratio of multiple fluid flowsControl signalMonitoring system
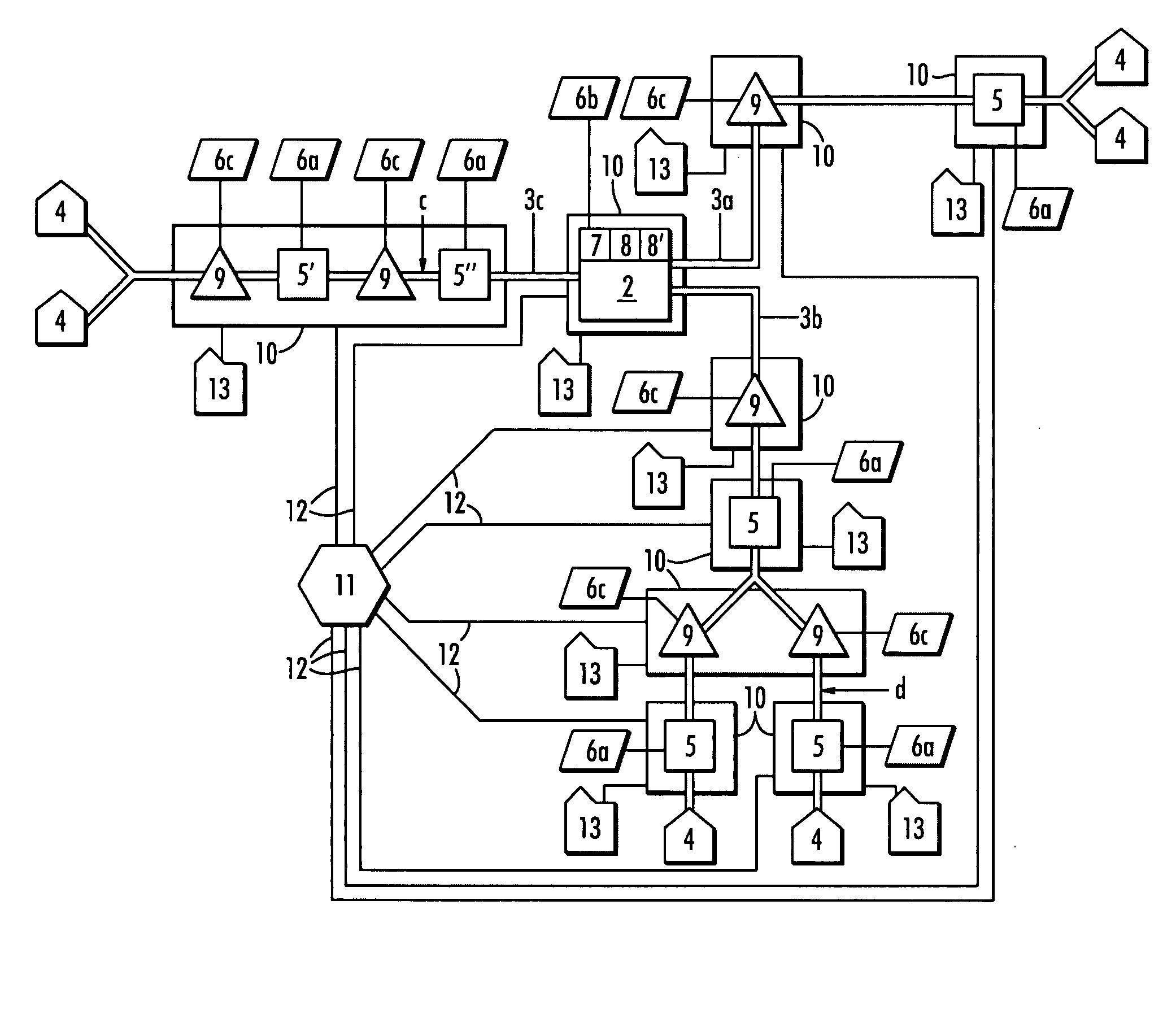
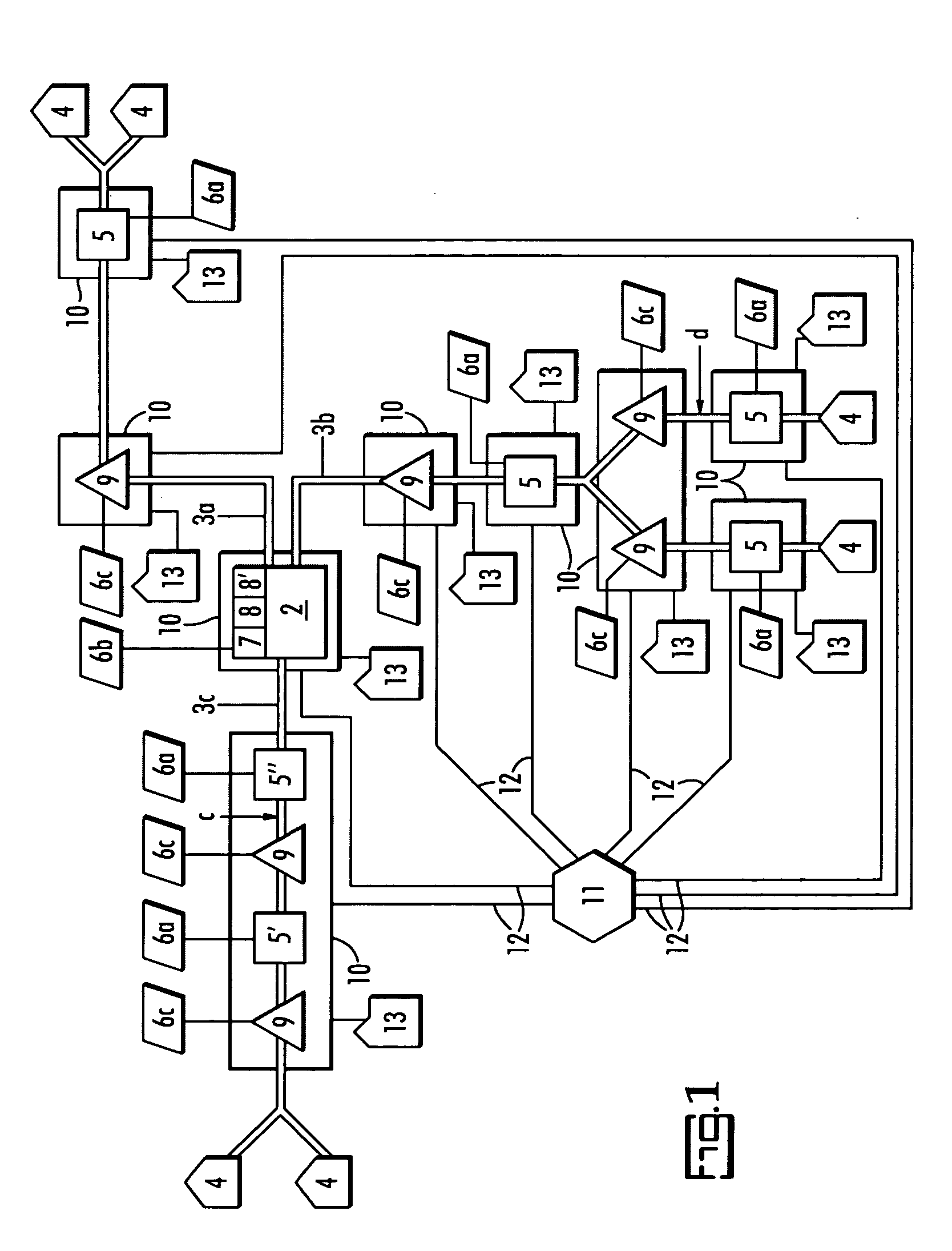
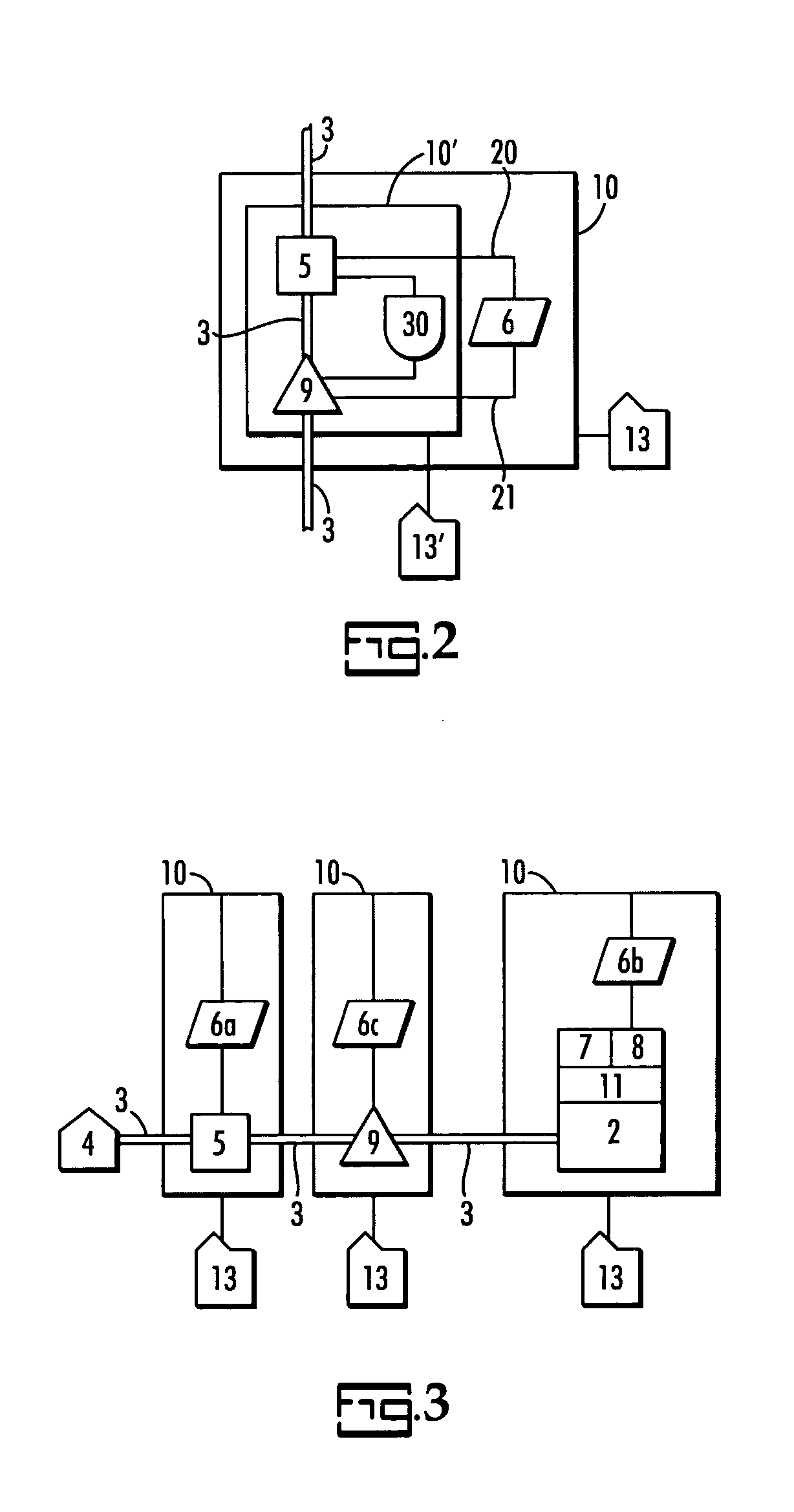
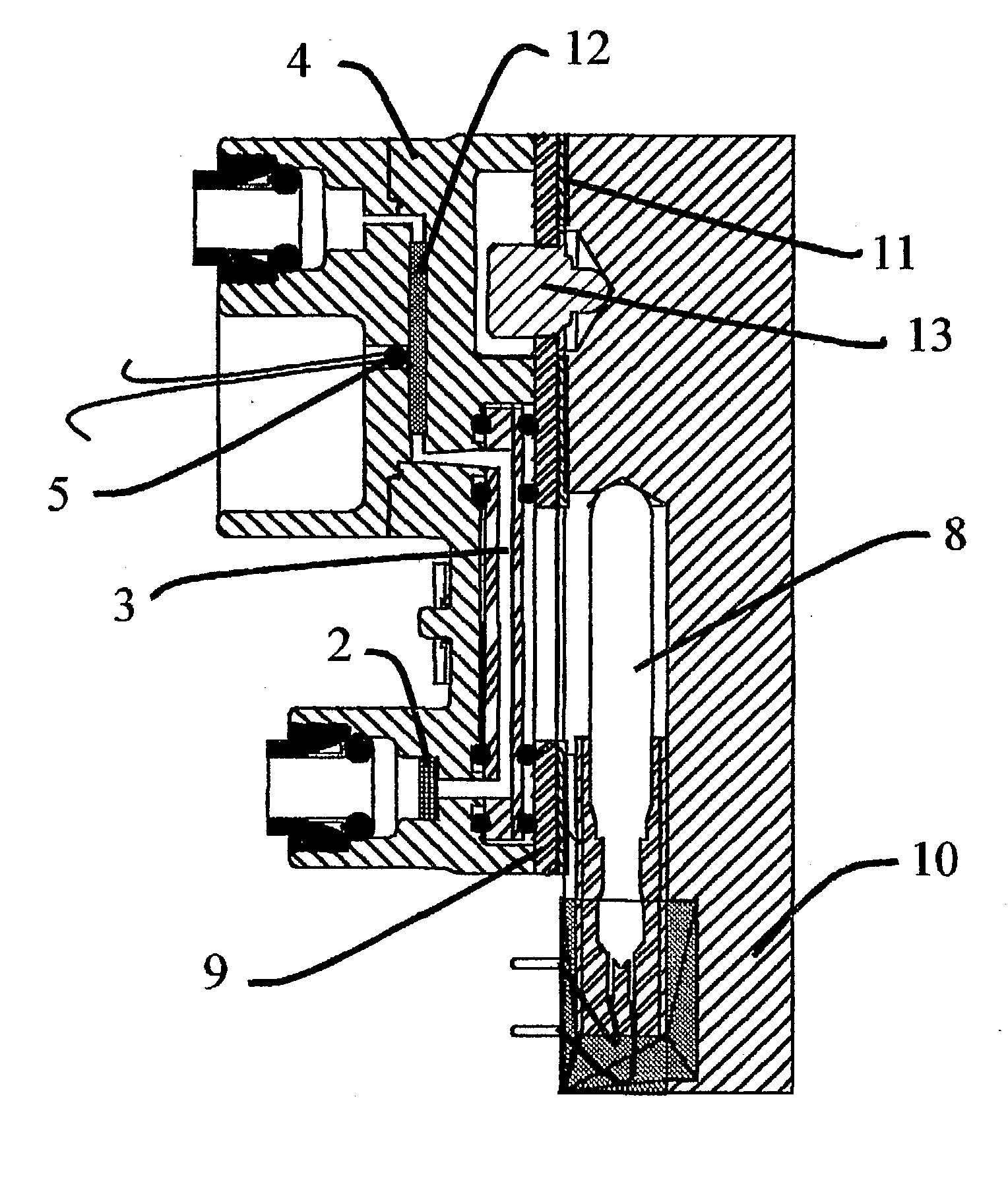
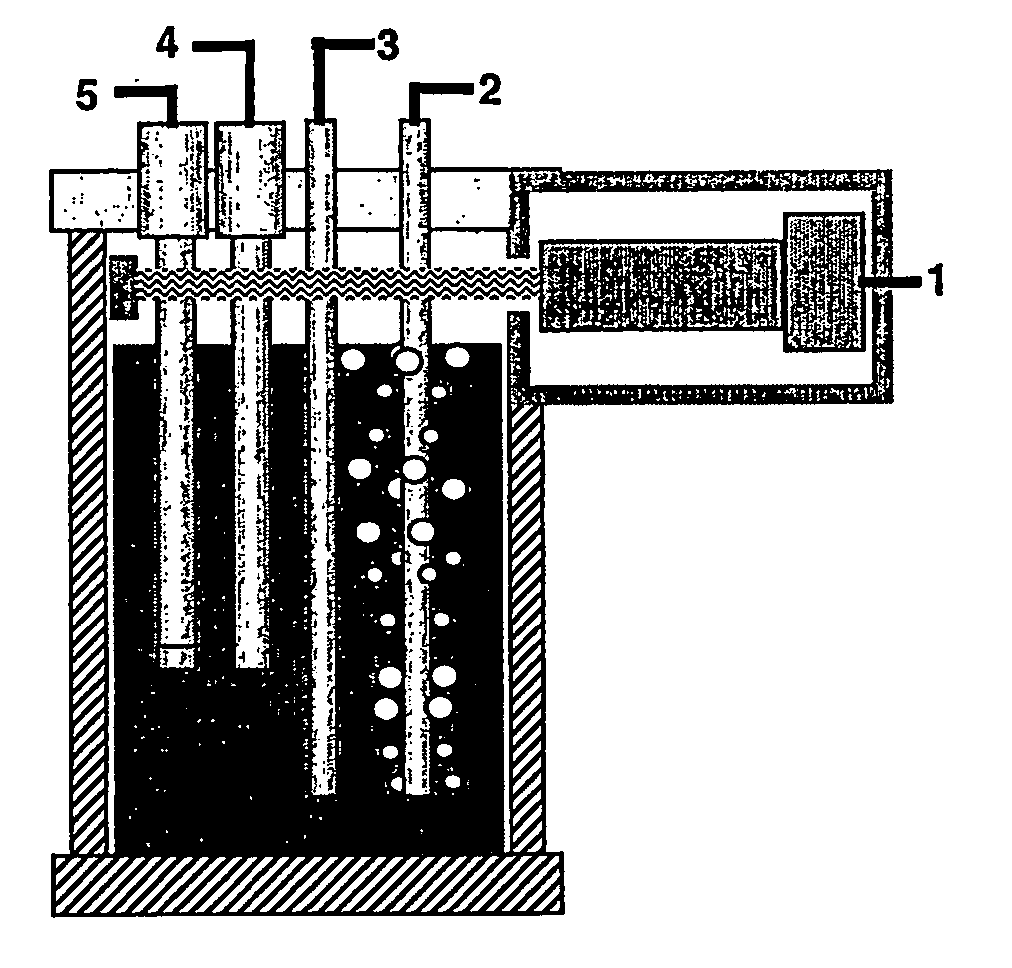
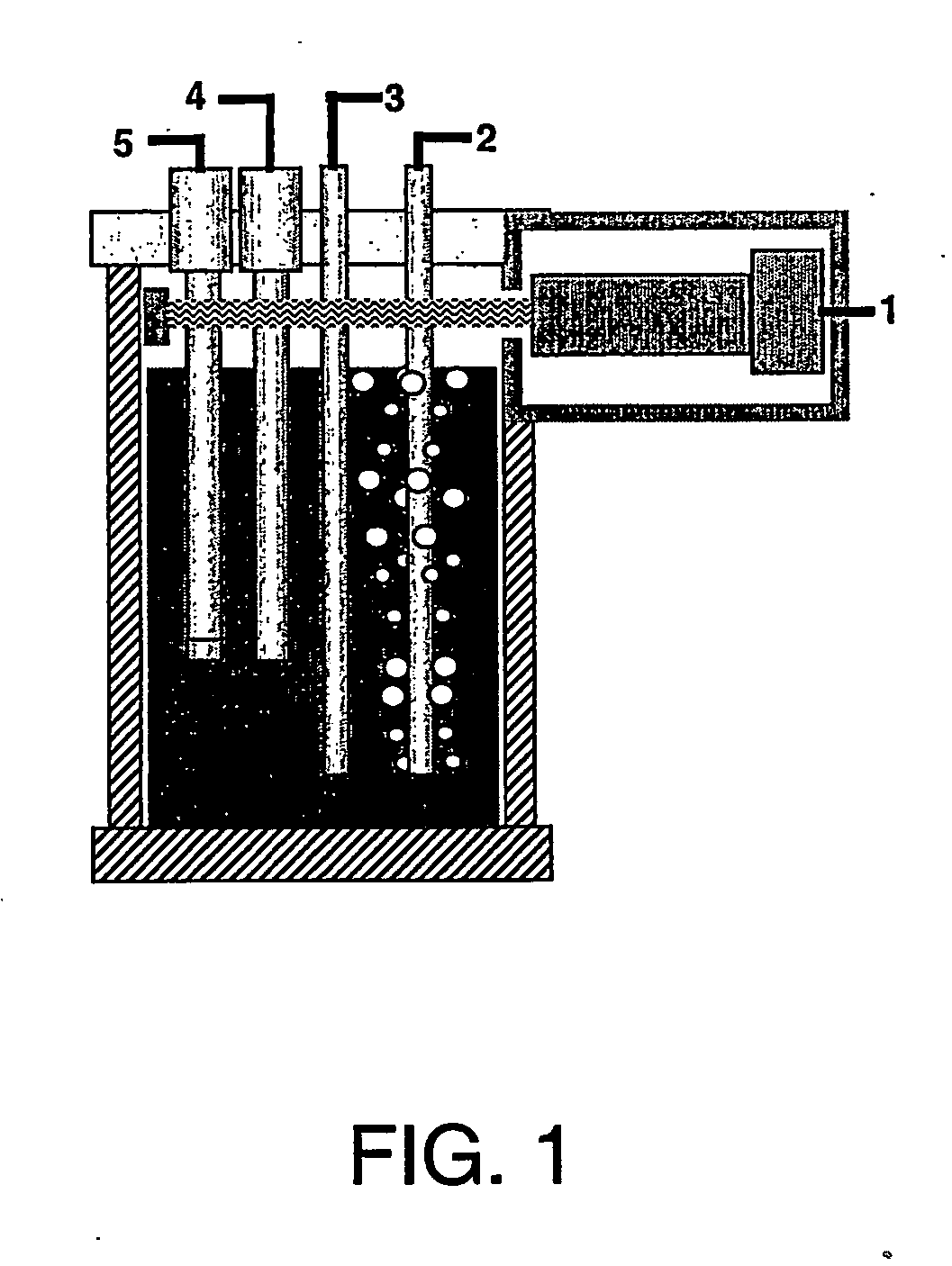
A water quality detection system for distributed water supply network. The system, 1, comprises a multiplicity of detectors, 5, wherein each detector of the detectors is capable of monitoring at least one attribute of water and providing a signal related to the attribute. A controller, 7, is provided which is capable of receiving each signal and comparing the signal to a control signal for the attribute. A response mechanism, 9, is responsive to the controller and activated when at least one signal matches the control signal. An access gate, 10, limits access to at least one of the detector, the controller or the response mechanism. An access key, 13, is provided for comparing a user attribute with a stored attribute wherein when the user attribute matches the stored attribute access is provided into the access gate.
Owner:PDA SECURITY SOLUTIONS
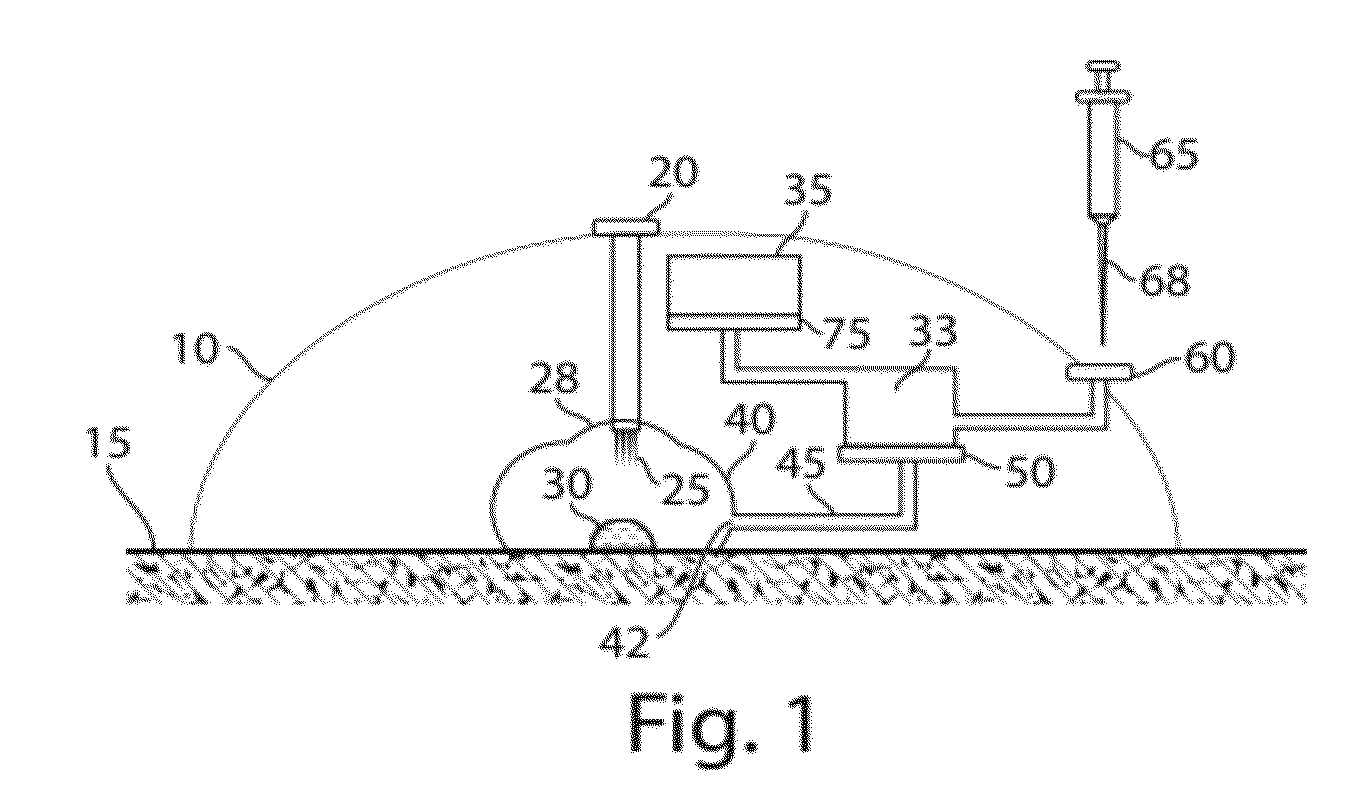
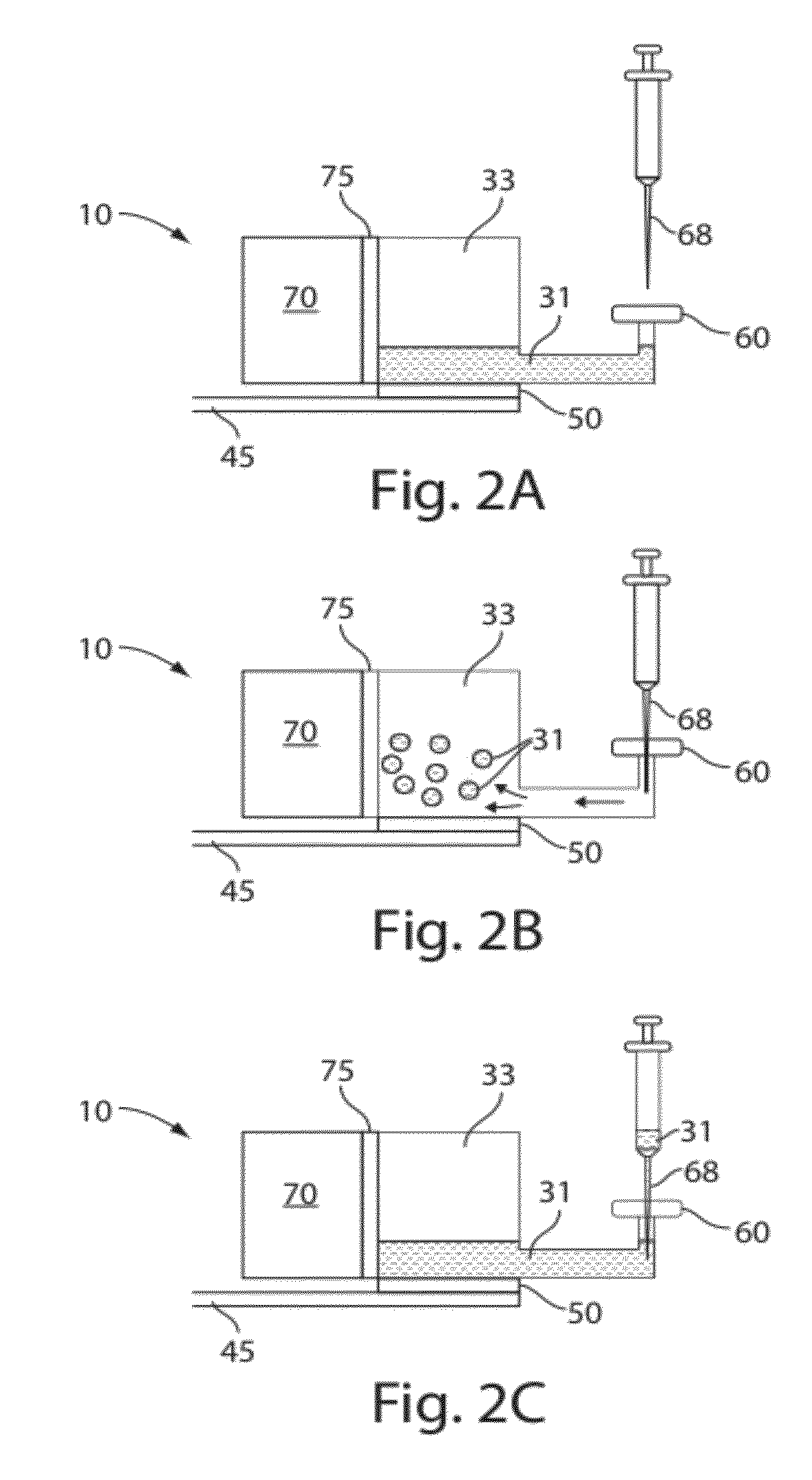
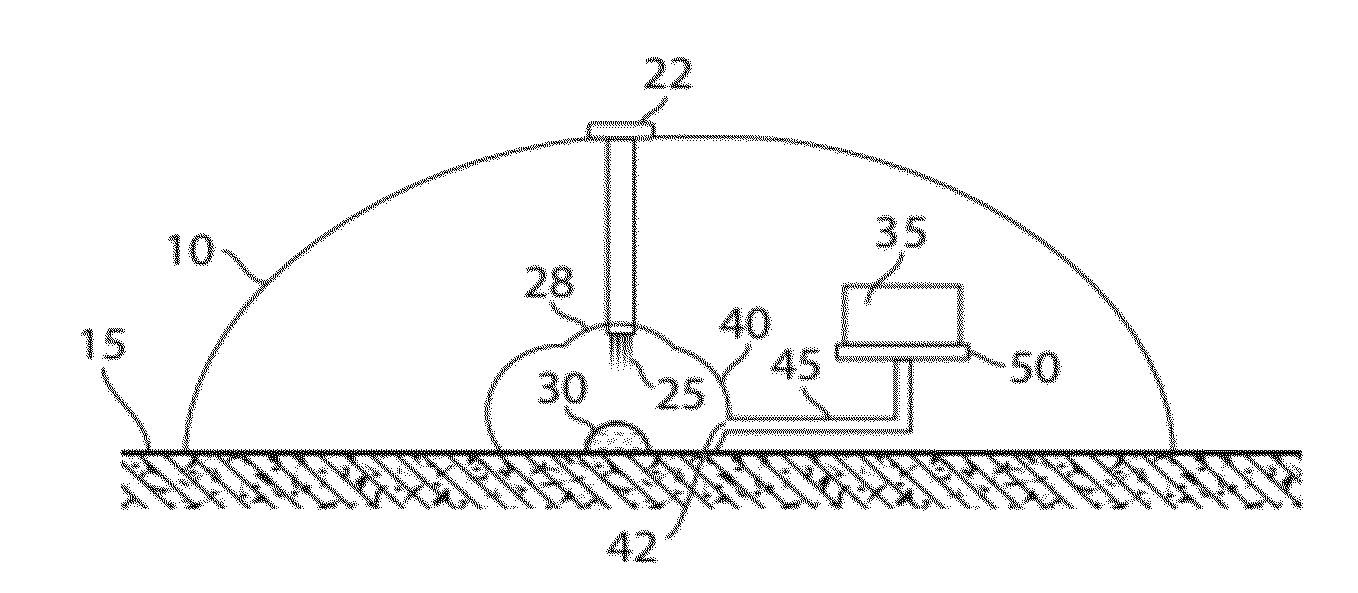
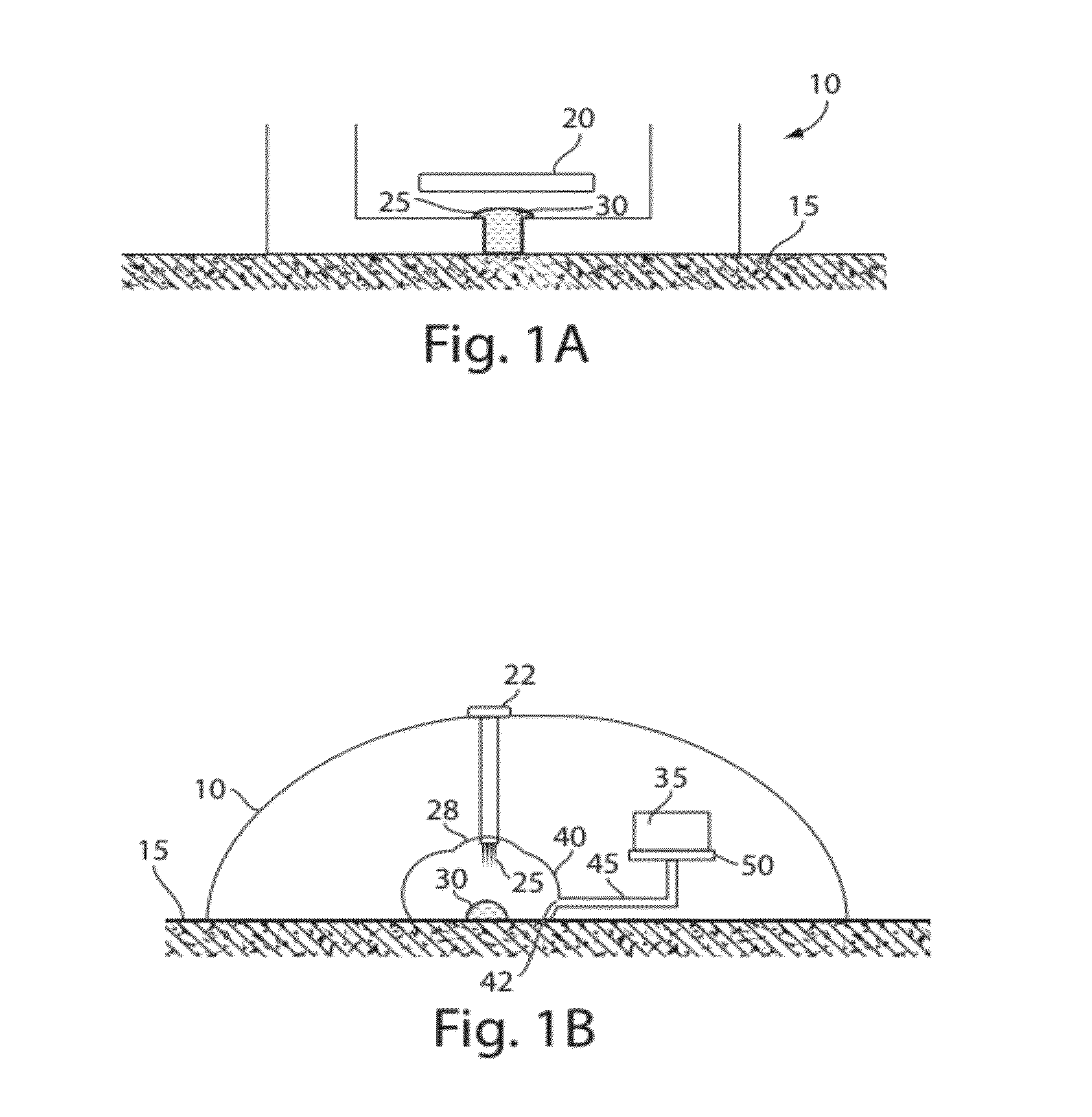
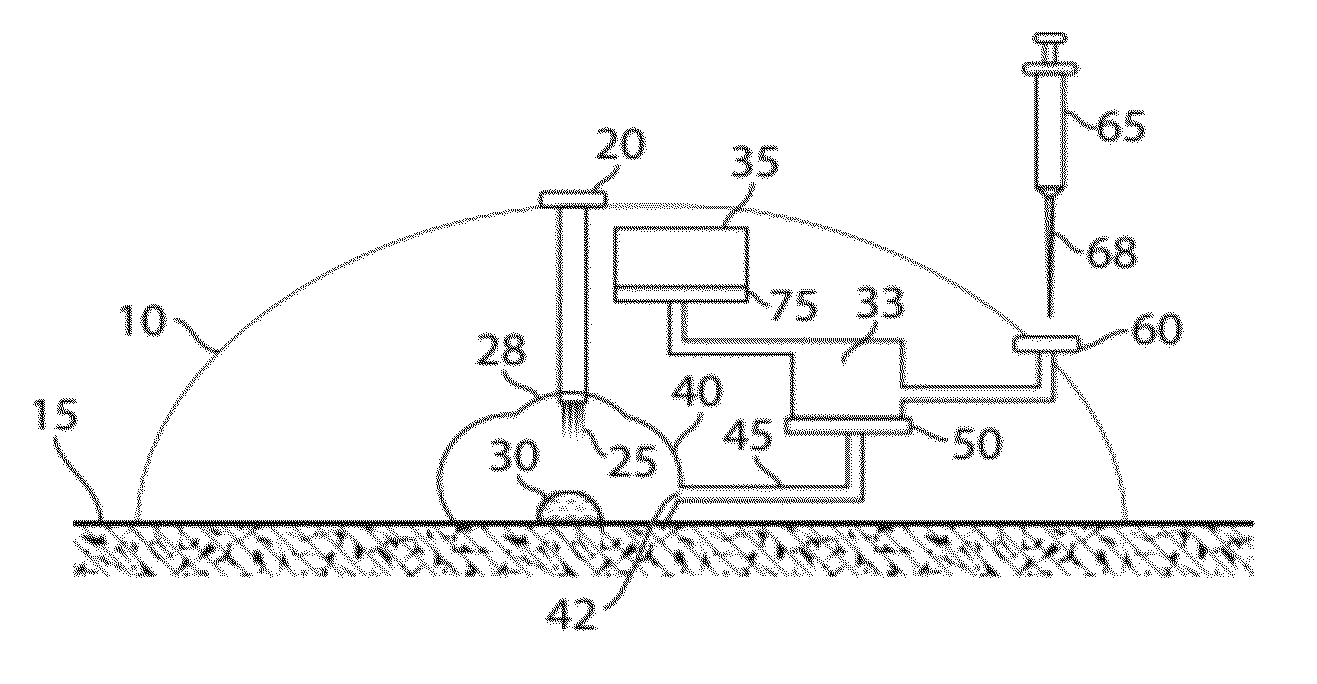
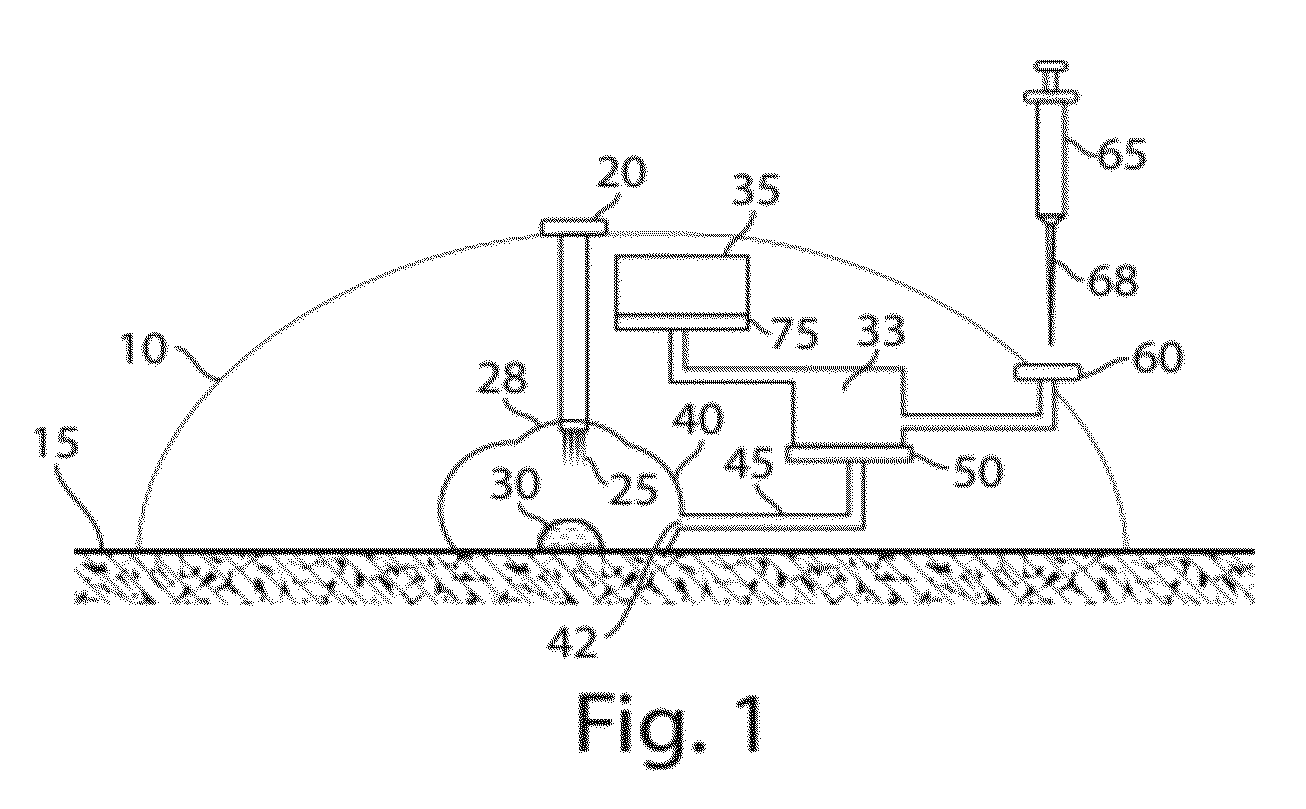
Plasma or serum production and removal of fluids under reduced pressure
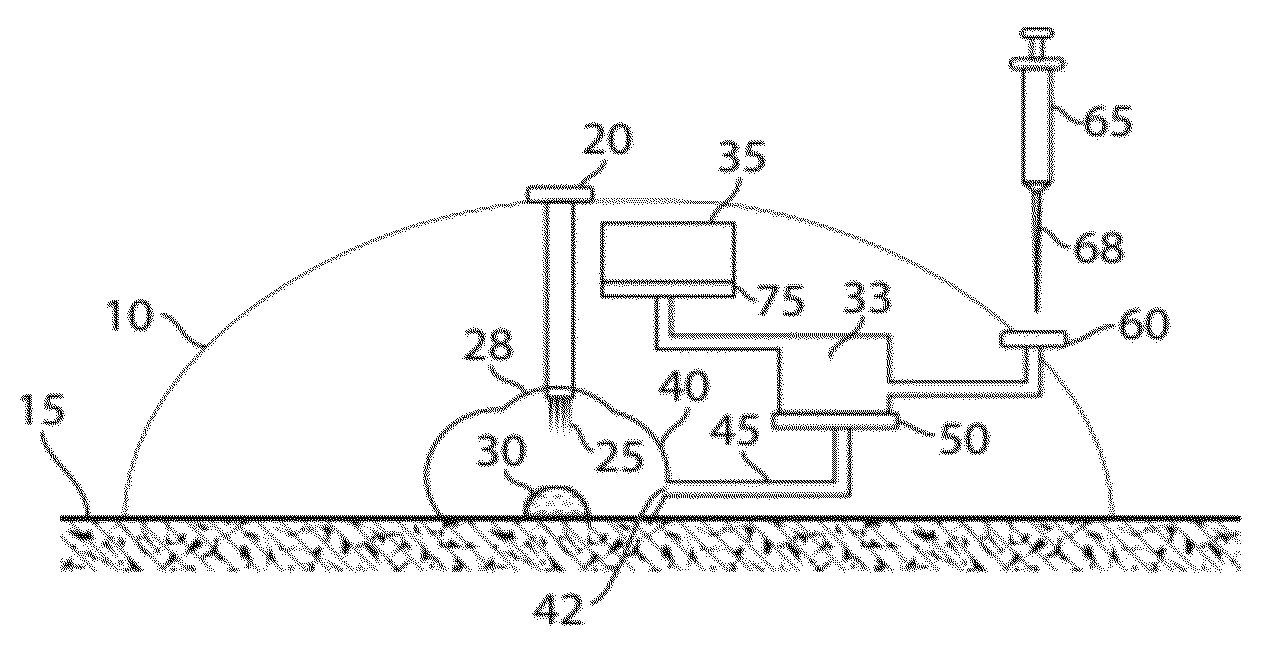
In some embodiments, the present invention generally relates to the separation of blood within a device to form plasma or serum. In some embodiments, the present invention generally relates to the removal of fluids, such as blood, contained within a device. For example, a device may be applied to the skin of a subject to receive blood from the subject and pass the blood through a separation membrane, which separates the blood into plasma and a portion concentrated in blood cells. As another example, blood or plasma may be allowed to clot within the device and serum (the unclotted portion of the blood) may be withdrawn from the device. The device may contain, in some cases, a vacuum source such as a pre-packaged vacuum to facilitate receiving of blood and / or passage of the blood through the separation membrane to produce plasma or serum.
Owner:YOURBIO HEALTH INC
Systems and methods for collection and/or manipulation of blood spots or other bodily fluids
InactiveUS20120277629A1Easy to operateReduction and elimination of painHaemofiltrationMedical devicesFilter paperBody fluid
The present invention generally relates to systems and methods for receiving blood (or other bodily fluids) from a subject, e.g., from or beneath the skin of a subject. In some cases, the blood (or other bodily fluids) may be deposited on a membrane or other substrate. For example, blood may be absorbed in a substrate, and dried in some cases to produce a dried blood spot. In one aspect, the present invention is generally directed to devices and methods for receiving blood from a subject, e.g., from the skin, using devices including a substance transfer component (which may contain, for example, one or more microneedles), and directing the blood on a substrate, e.g., for absorbing blood. The substrate, in some embodiments, may comprise filter paper or cotton-based paper. After absorption of some blood onto the substrate, the substrate may be removed from the device and shipped or analyzed.
Owner:SEVENTH SENSE BIOSYST
Apparatuses for interaction with a subterranean formation, and methods of use thereof
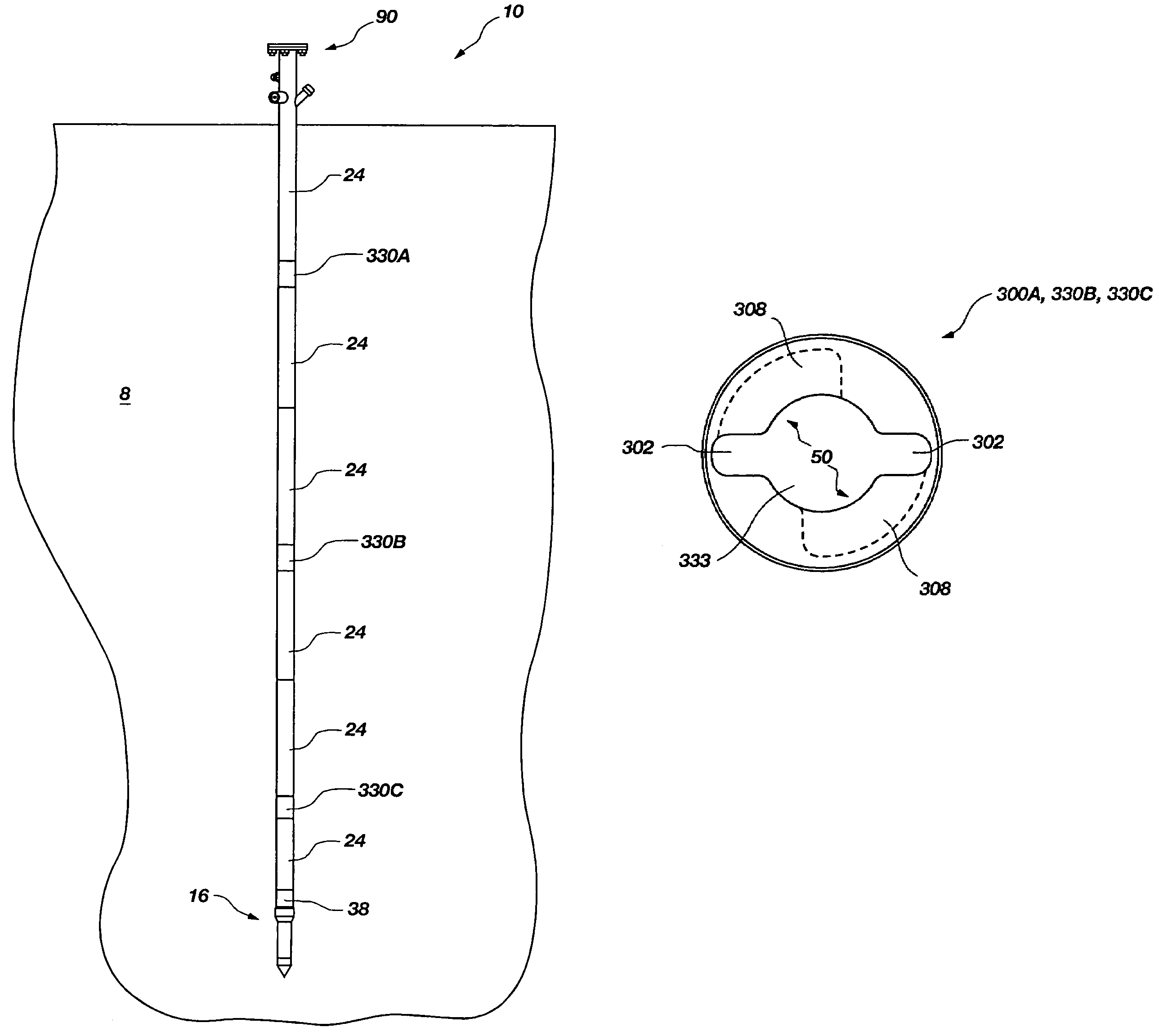
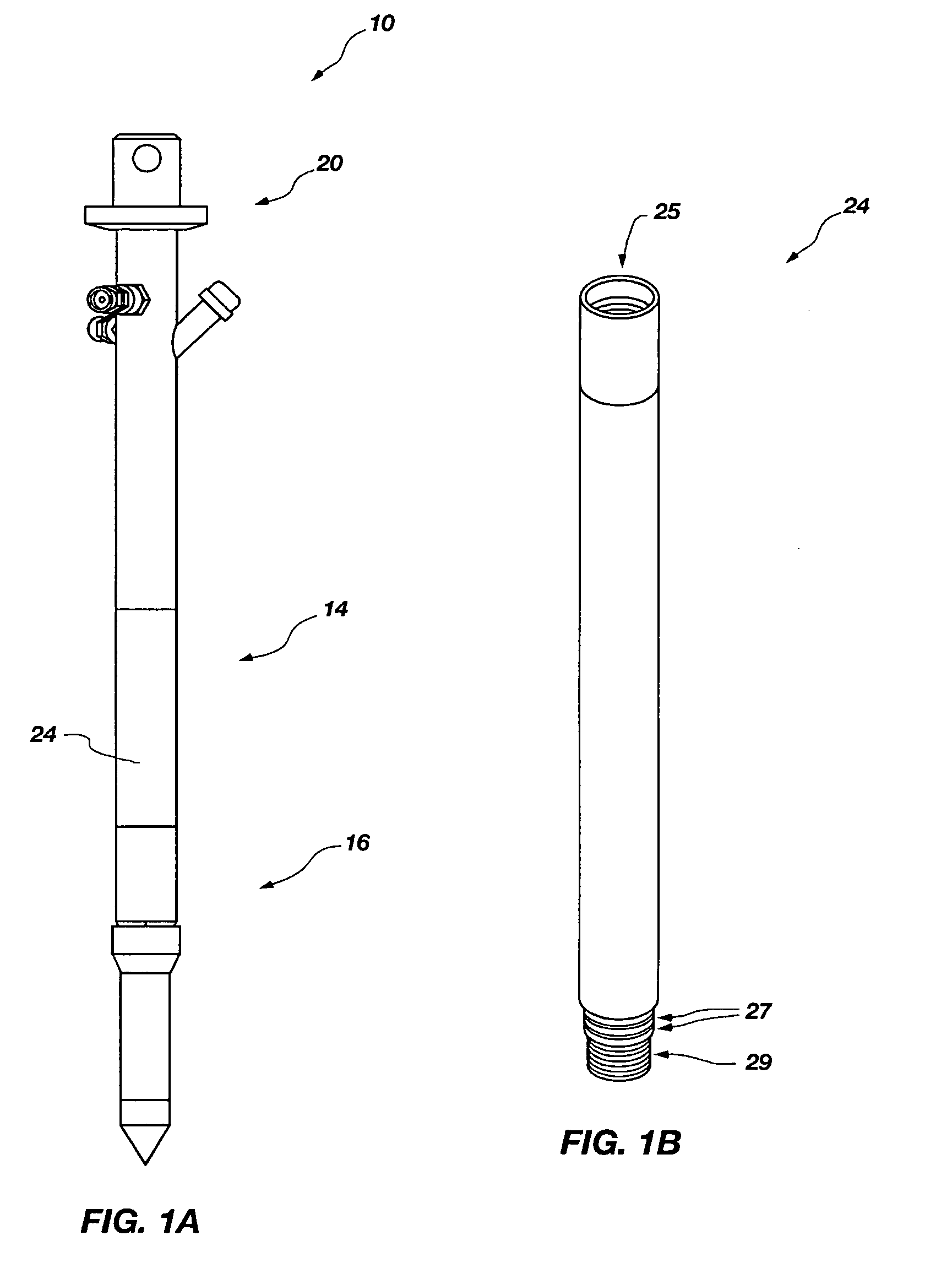
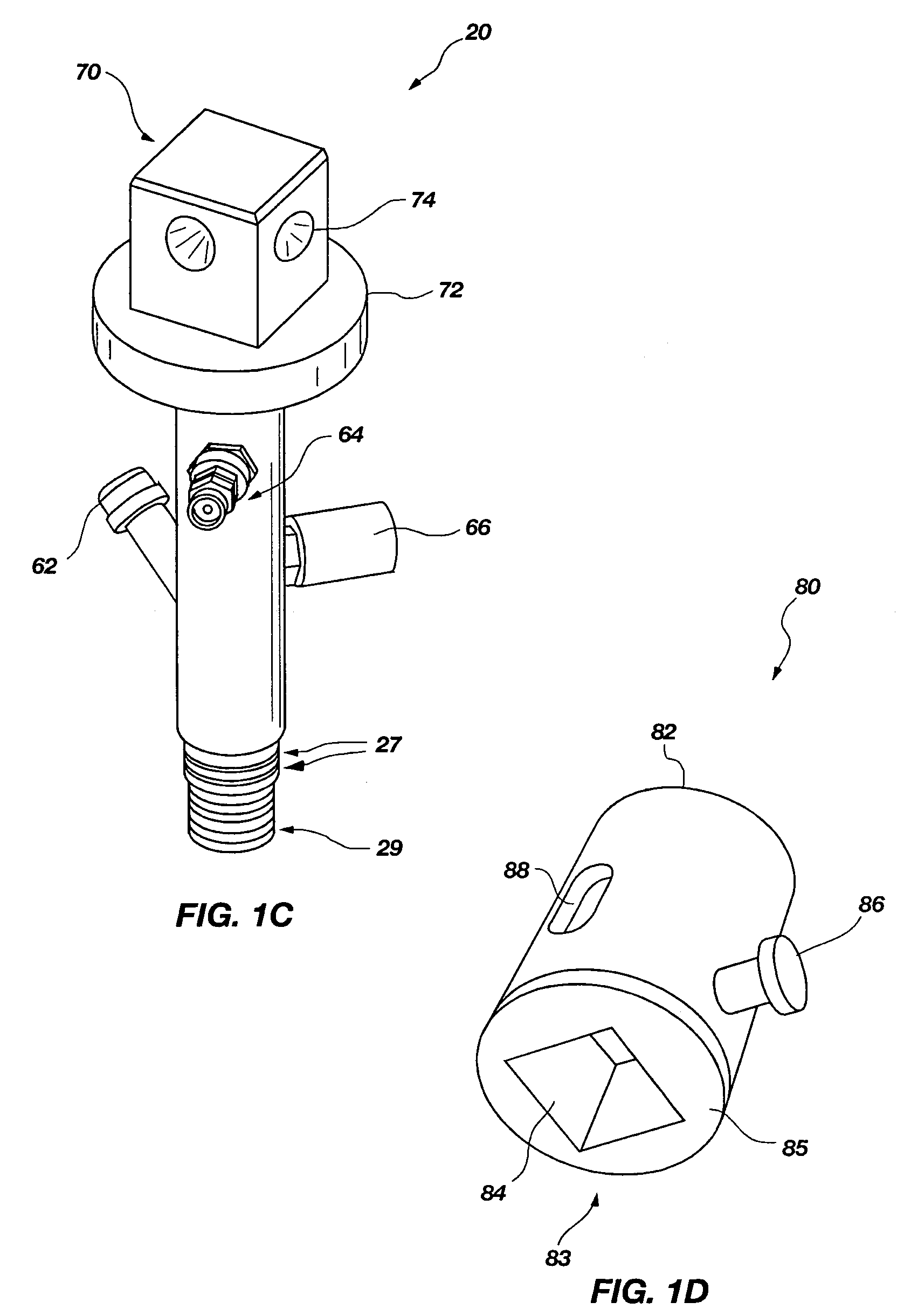
An access casing assembly structured for placement at least partially within a subterranean formation by forcing the access casing assembly thereinto, comprising a plurality of casing sections operably coupled to form a central elongated cavity for providing access to the subterranean region is disclosed. Further, a tip portion of the access casing assembly may include a porous filter through which liquid or gas may communicate with the central elongated cavity. Also, a receiving member or at least one engagement hub may form a portion of the central elongated cavity and may include an engagement feature configured for selectively and lockingly engaging a locking structure of a device to be positioned within the access casing assembly. Methods of use are disclosed. A tensiometer is disclosed including a chamber structured for allowing at least partially filling with a fluid subsequent to contact therewith.
Owner:BATTELLE ENERGY ALLIANCE LLC
Method for detection and analysis of aromatic hydrocarbons from water
InactiveUS20100068821A1Easy to judgeBiological testingTesting organic contamination in waterAromatic hydrocarbonOrganic chemistry
Methods for analyzing aromatic hydrocarbons dissolved in water are discussed. The methods include providing a substrate coated with a thin film layer of a material, wherein the material has a high affinity for at least one aromatic hydrocarbon, the material is substantially optically transparent, and the material has near-zero auto fluorescence, inserting the coated substrate directly into an environmental location including water, waiting for an exposure time permitting at least one aromatic hydrocarbon to absorb into the thin film layer, retrieving the coated substrate from the environmental location, removing any non-absorbed matter from the coated substrate, and performing fluorescence analysis on the coated substrate to detect aromatic hydrocarbons present in the thin film layer. Also methods for analyzing aromatic hydrocarbons dissolved in water contained in coated vessels are provided.
Owner:DAKOTA TECH
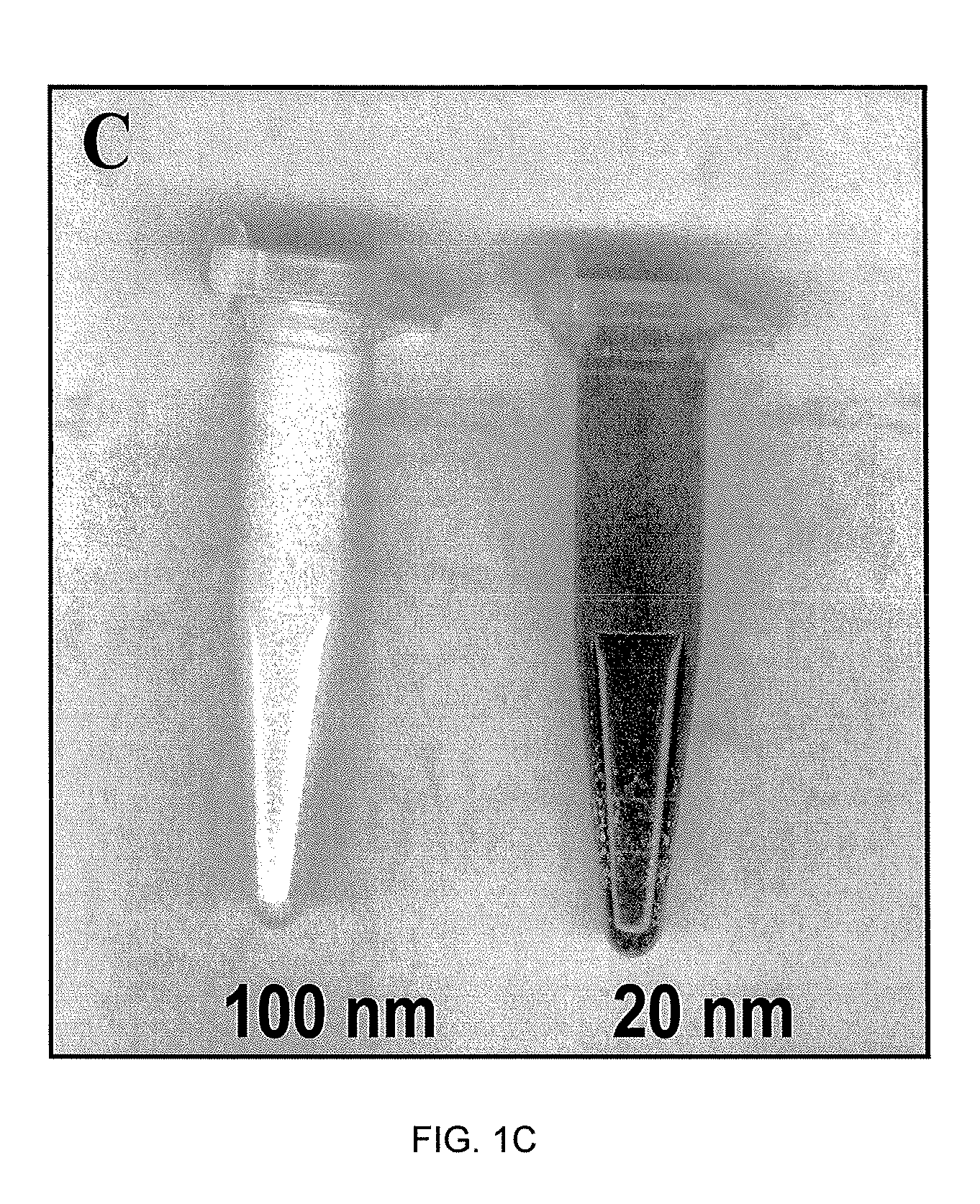
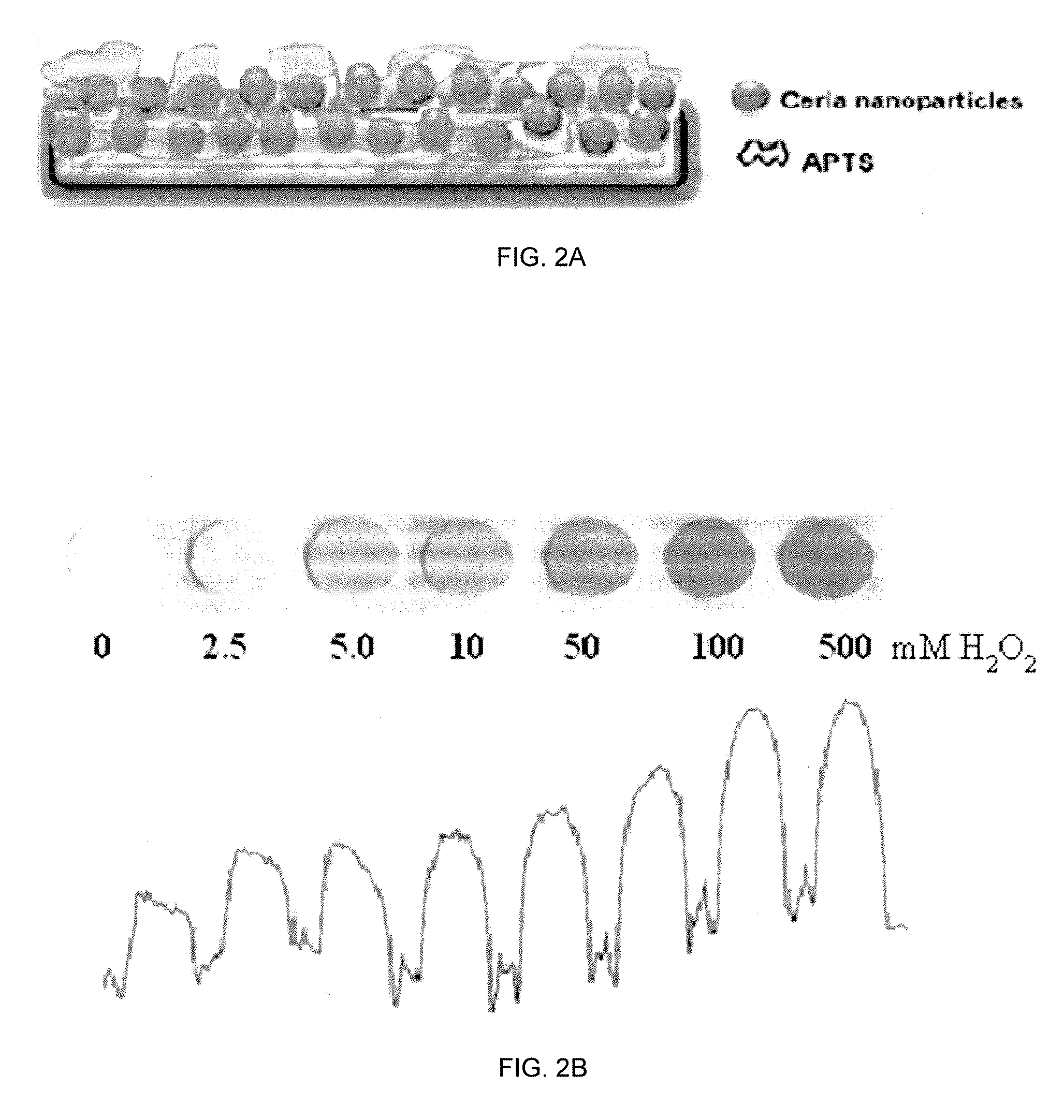
Reagentless Ceria-Based Colorimetric Sensor
ActiveUS20120315659A1Possible to detectIncrease rangeMaterial analysis by observing effect on chemical indicatorMicrobiological testing/measurementDelivery vehicleIn vivo
A colorimetric reagent in the form of nanoparticles, composite nanoparticles, and nanoparticle coatings, including methods of use, methods of preparation, deposition, and assembly of related devices and specific applications. The colorimetric reagent comprises cerium oxide nanoparticles which are used in solution or immobilized on a solid support, either alone or in conjunction with oxidase enzymes, to form an active colorimetric component that reacts with an analyte to form a colored complex. The rate of color change and the intensity of the color are proportional to the amount of analyte present in the sample. Also described is the use of ceria and doped ceria nanoparticles as an oxygen storage / delivery vehicle for oxidase enzymes and applications in biocatalytic processes in anaerobic conditions of interest in biomedicine and bioanalysis. Further described are a variety of related applications of the disclosed technology including clinical diagnosis, in vivo implantable devices, food safety, and fermentation control.
Owner:CLARKSON UNIVERSITY
Use of disulfonated anthracenes as inert fluorescent tracers
InactiveUS20050025659A1Quantity minimizationDetection of fluid at leakage pointSamplingAnthraceneSteam line
The use of an isomer of anthracene disulfonic acid as an inert fluorescent tracer is described and claimed. The utility of this type of inert fluorescent tracer in boiler systems to trace unwanted carryover of boiler water into a steam line is also described and claimed.
Owner:ECOLAB USA INC
Plasma or serum production and removal of fluids under reduced pressure
Owner:YOURBIO HEALTH INC
Method and apparatus for monitoring and measuring oil spills
InactiveUS7009550B2Improve accuracyDirection finders using radio wavesGeneral water supply conservationRadarMarine engineering
The present invention relates to an oil spill identification system and oil spill identification sensors to be used in connection with this system. The system is used primarily on fixed offshore structures, but may also be used on fixed onshore constructions. The sensor comprises a combination of a radar and at least a microwave radiometer. The data collected are transmitted to a control station. Preferably, the transmittal takes place at pixel level between the sensors and the control station. The control station processes the data received at pixel level and transmits data to an end-user, preferably through the internet. The invention also relates to a method for utilizing the system and a use of the system.
Owner:MOELLER JENSEN PETER
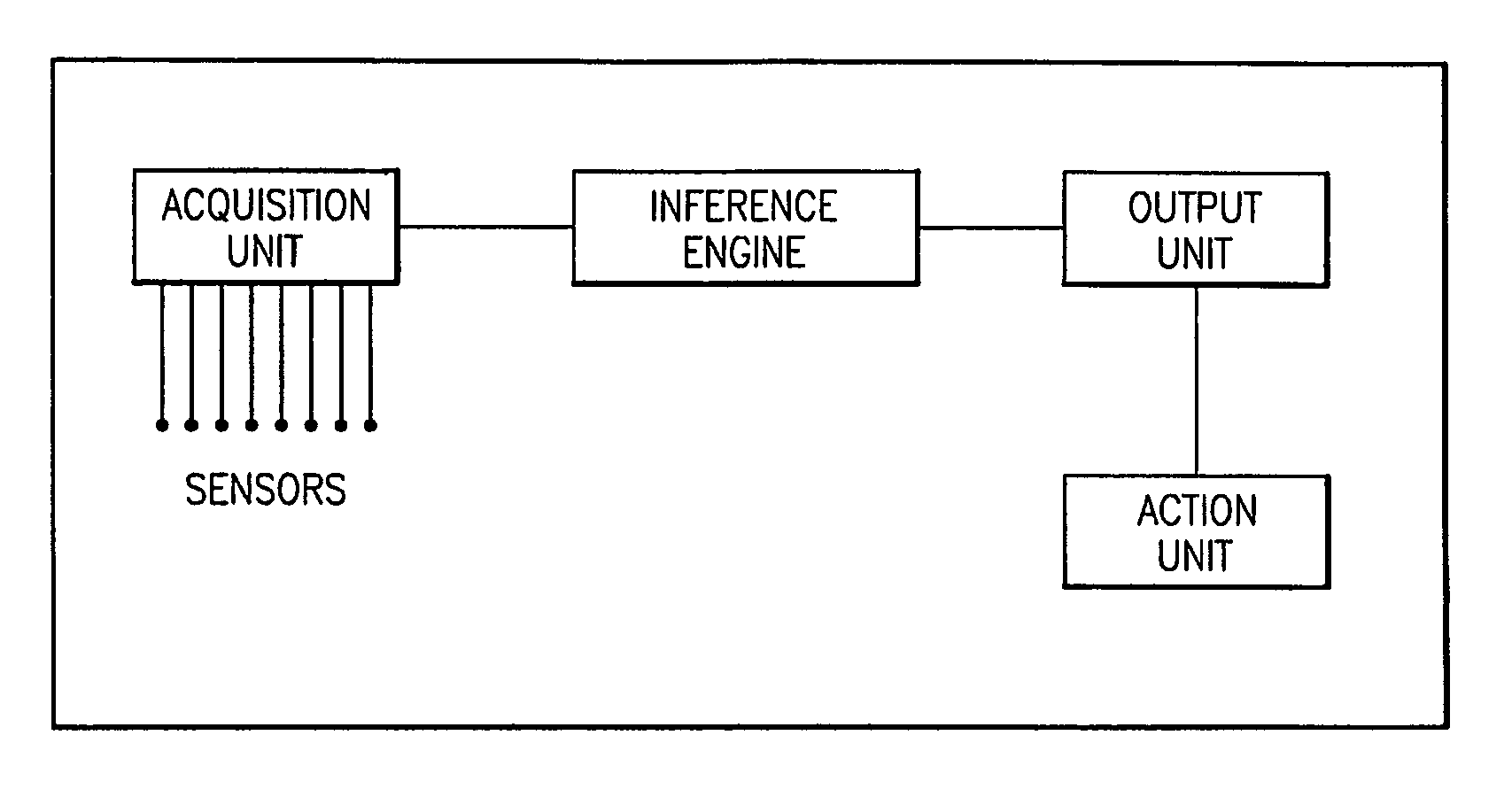
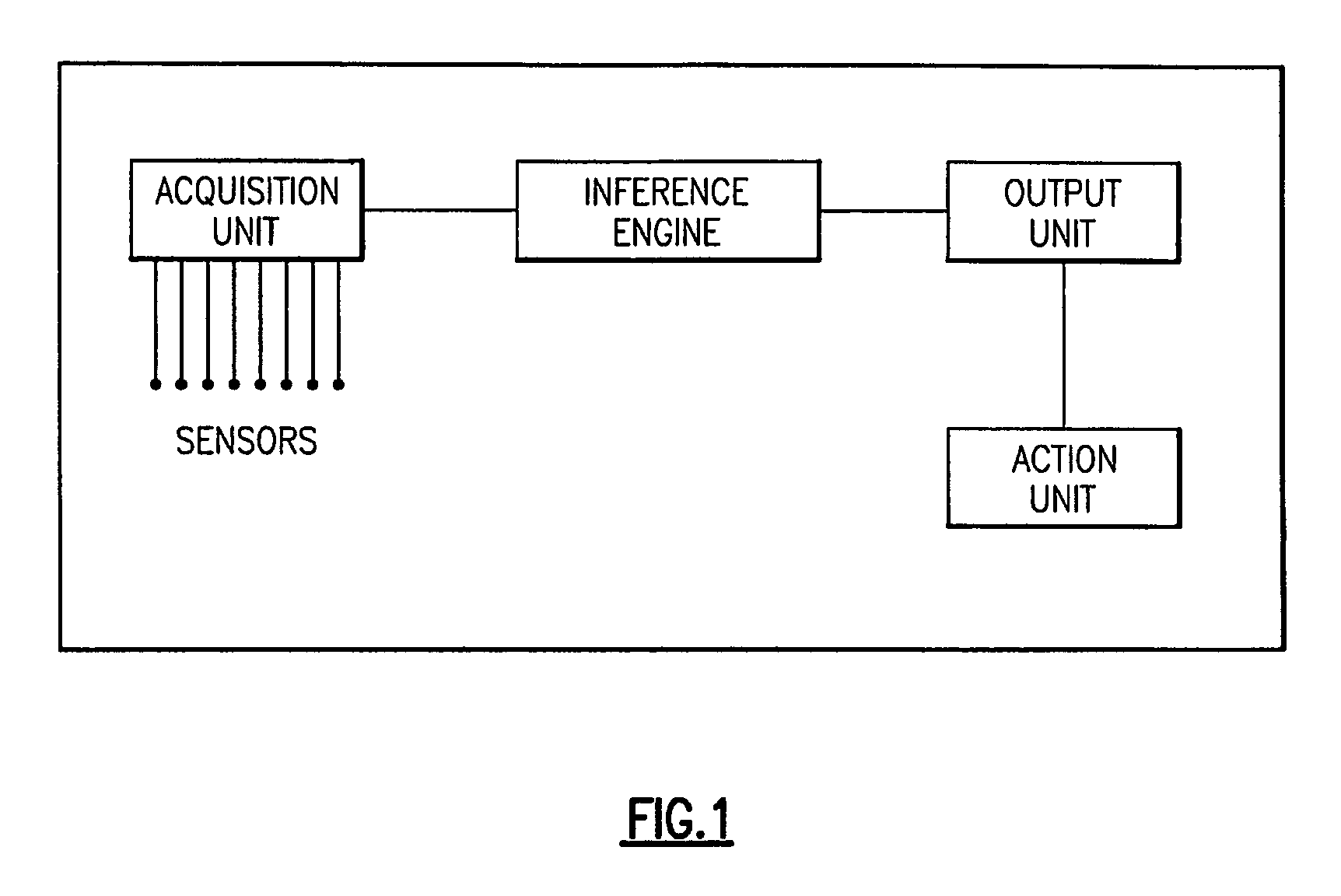
System for detection and prediction of water quality events
InactiveUS7720615B2Amplifier modifications to reduce noise influenceMicrobiological testing/measurementCorrelation coefficientPredictive systems
Owner:SENSIS CORPORATION
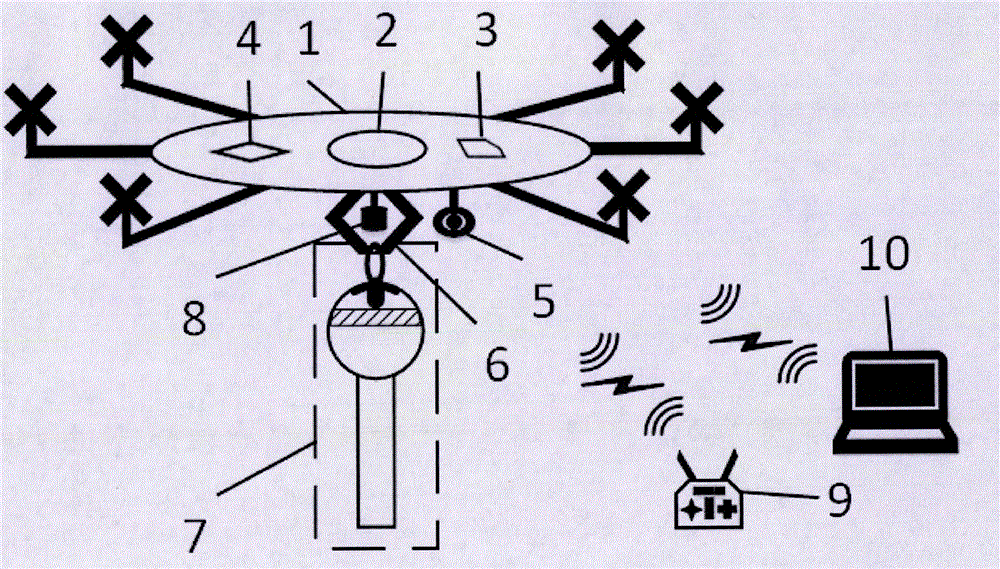
Unmanned aerial vehicle based on-line water quality testing device dropping and recovering method
PendingCN106771041AReduce in quantityApplicable to a wide range of environmentsBatteries circuit arrangementsTransmission systemsRecovery methodWater quality
The invention relates to an unmanned aerial vehicle based on-line water quality testing device dropping and recovering method. The unmanned aerial vehicle based on-line water quality testing device dropping and recovering method comprises an unmanned aerial vehicle, a remote controller and a ground monitoring station, the unmanned aerial vehicle is loaded with a camera, a function module, an onboard manipulator and a water quality testing device, the water quality testing device is connected with the unmanned aerial vehicle through the onboard manipulator, and the water quality testing device is dropped and recovered by remotely controlling the unmanned aerial vehicle. The water quality testing device tests the water quality and wirelessly sends the water quality test result to a remote monitoring system. The technological means of the unmanned aerial vehicle technique, the manipulator control technique, the remote water quality testing and the like are comprehensively utilized, the water quality testing device is arranged in an appointed water area rapidly, the water quality is tested on line, the number of immobilized water quality testing stations is saved due to the movable arrangement, the time, the labor force and the cost are saved, the method is suitable for various environments, is particularly suitable for testing the water quality of the large lake area in the remote region, and is suitable for emergent monitoring of the sudden water environment pollution.
Owner:CHINA JILIANG UNIV
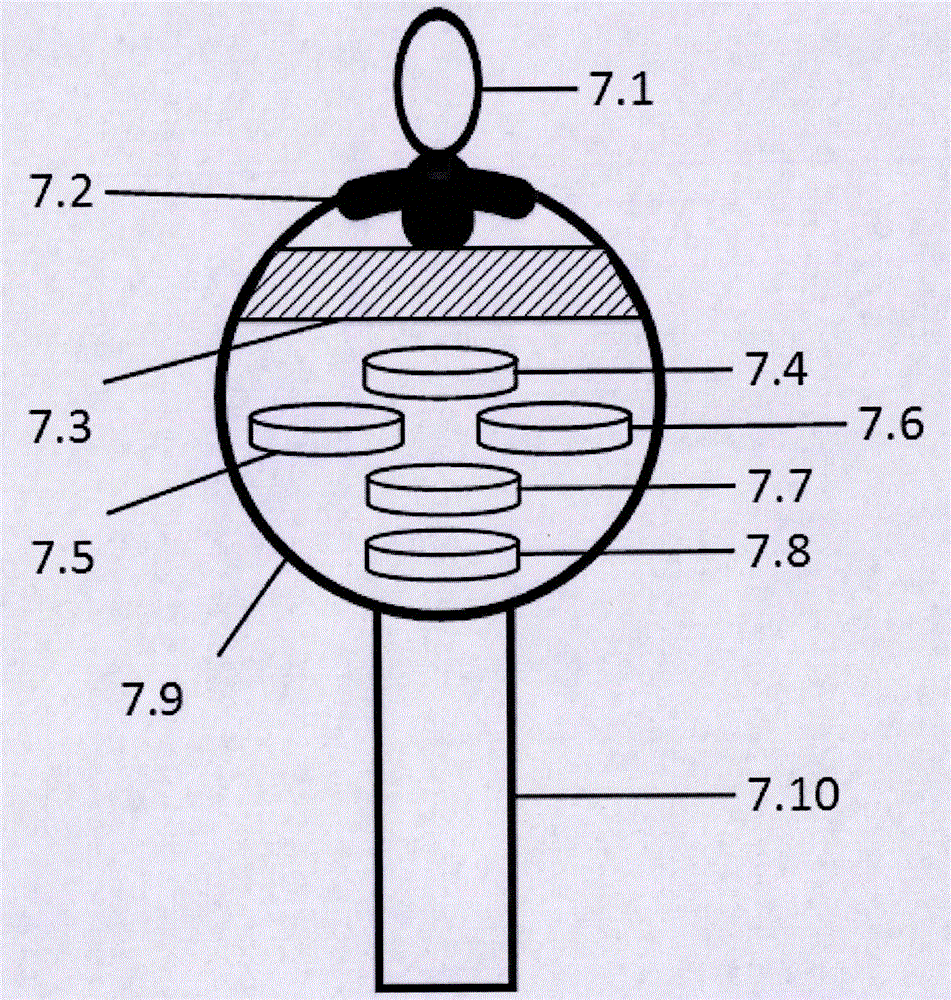
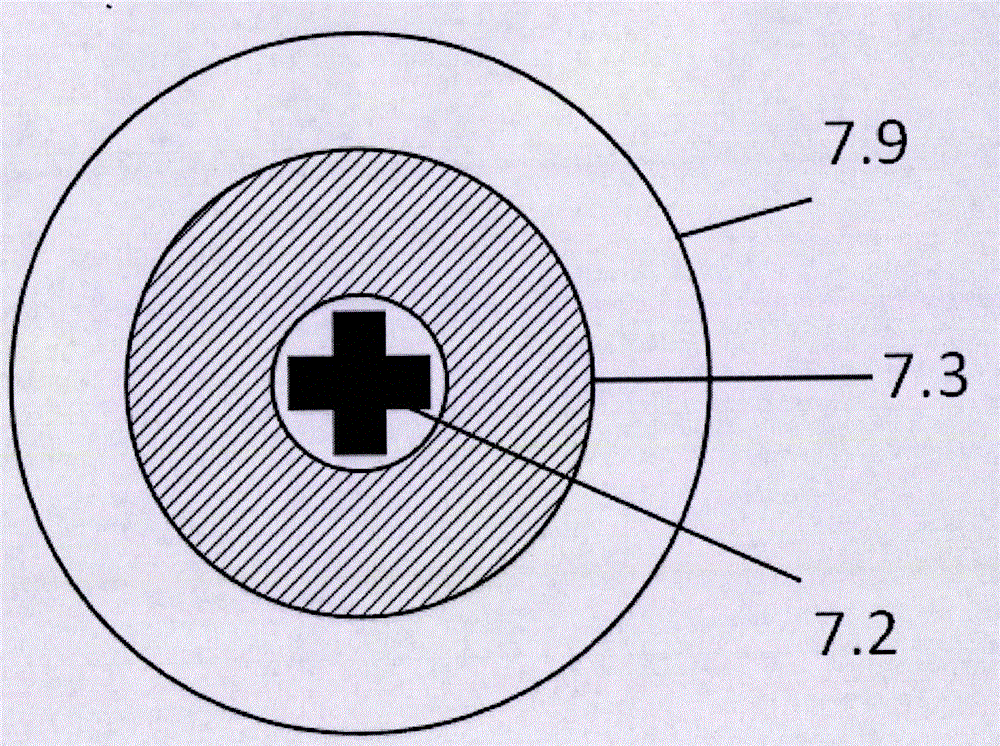
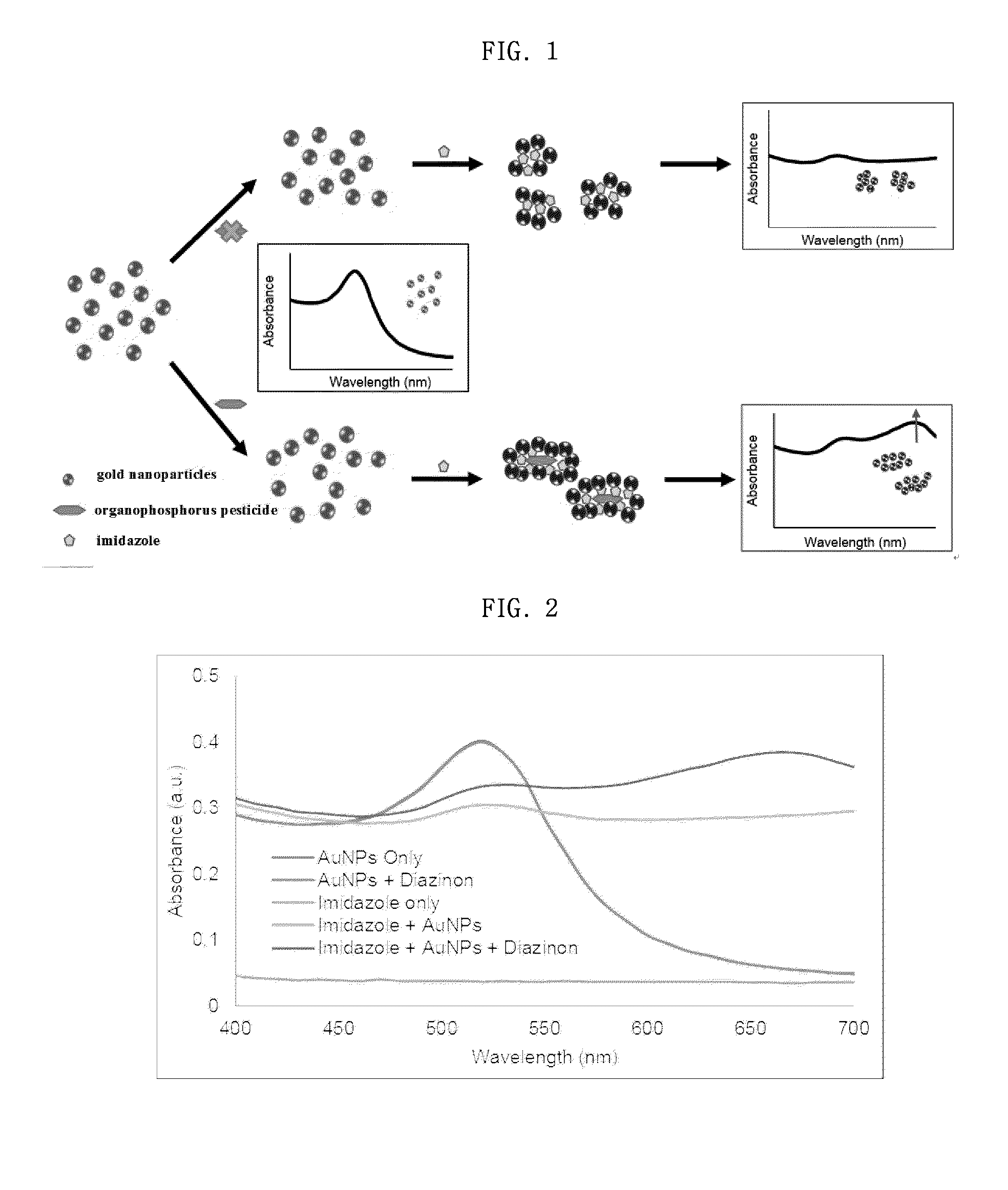
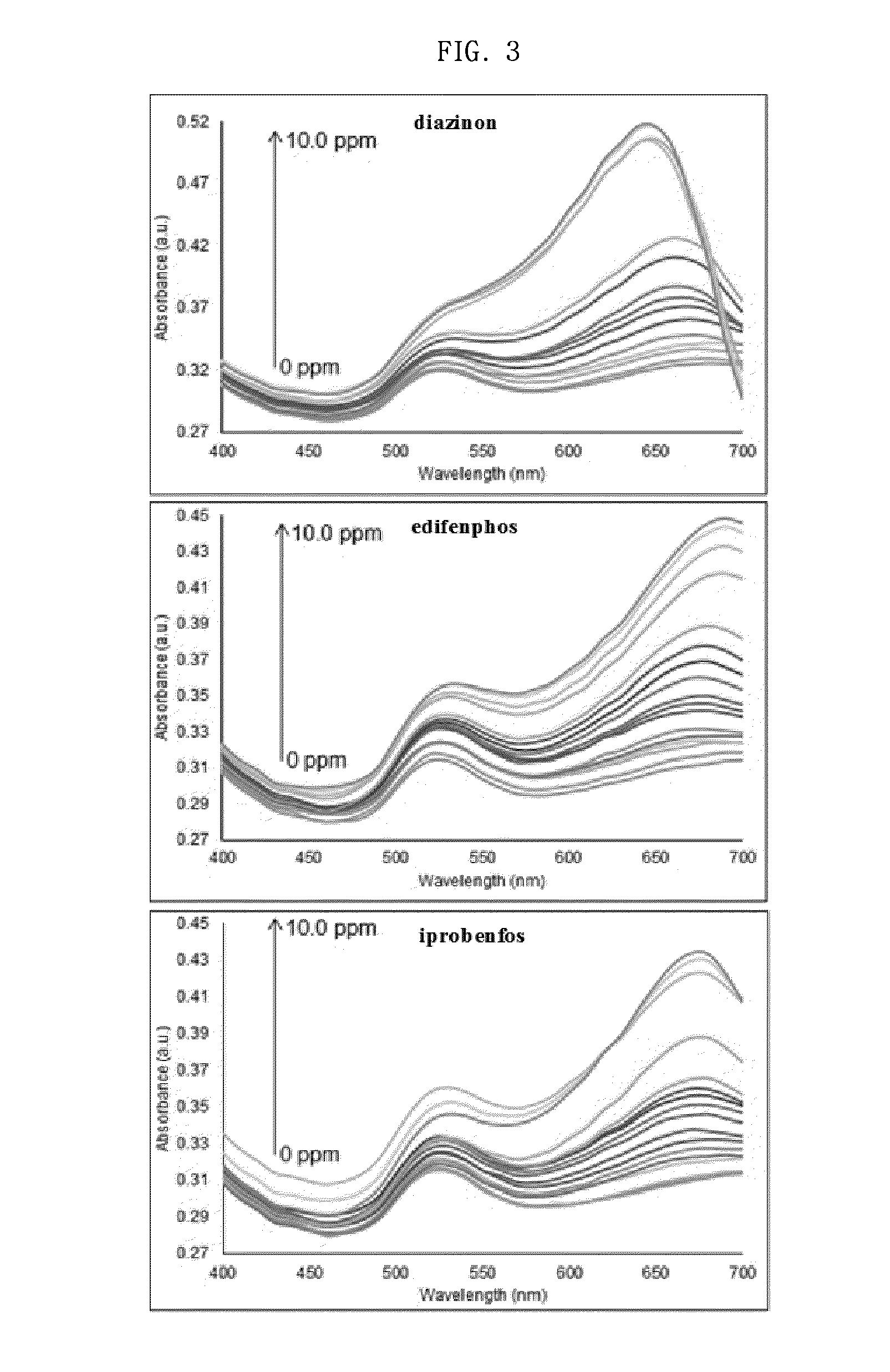
Sensor system for detecting organophosphorus residues by inducing coagulation of gold nanoparticles
ActiveUS20150355154A1Analysis using chemical indicatorsMaterial analysis by observing effect on chemical indicatorGreen fluorescent proteinPesticide residue
A sensor system detects organophosphorus pesticide residue by inducing the aggregation of gold nanoparticles. A method comprises aggregating gold nanoparticles by a reaction between an organophosphorus pesticide and imidazole or a green fluorescent protein (GFP), and detecting the organophosphorus pesticide based on a absorption spectral change resulting from the aggregation. The system for detecting pesticide residue is useful as a biosensor for analyzing pesticide residue in situ, because the optical change of the reagent by the presence of an organophosphorus pesticide is distinct, the detection speed is fast, and the range of detection limits is broad.
Owner:CHUNG ANG UNIV IND ACADEMIC COOP FOUND
Water quality analyzer
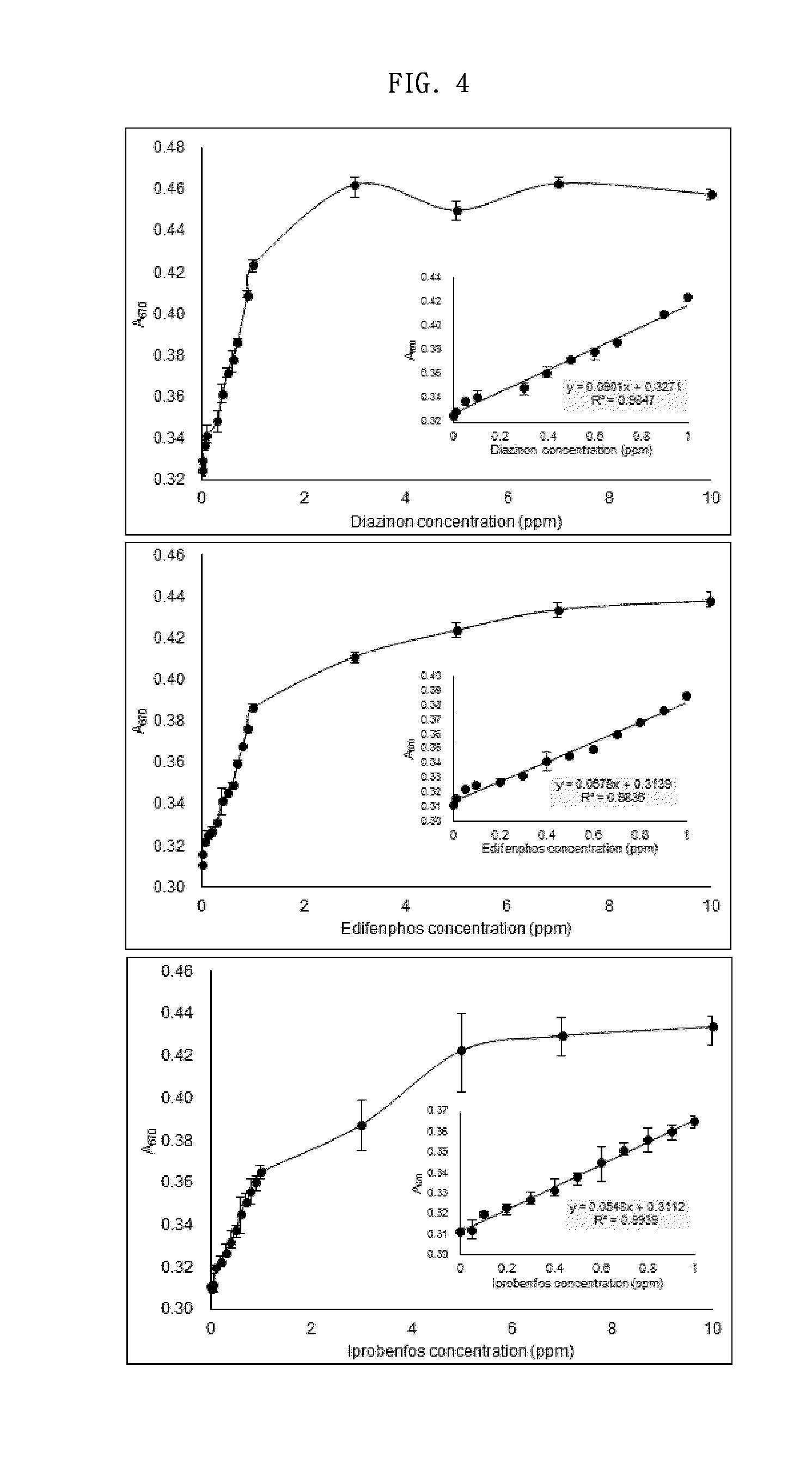
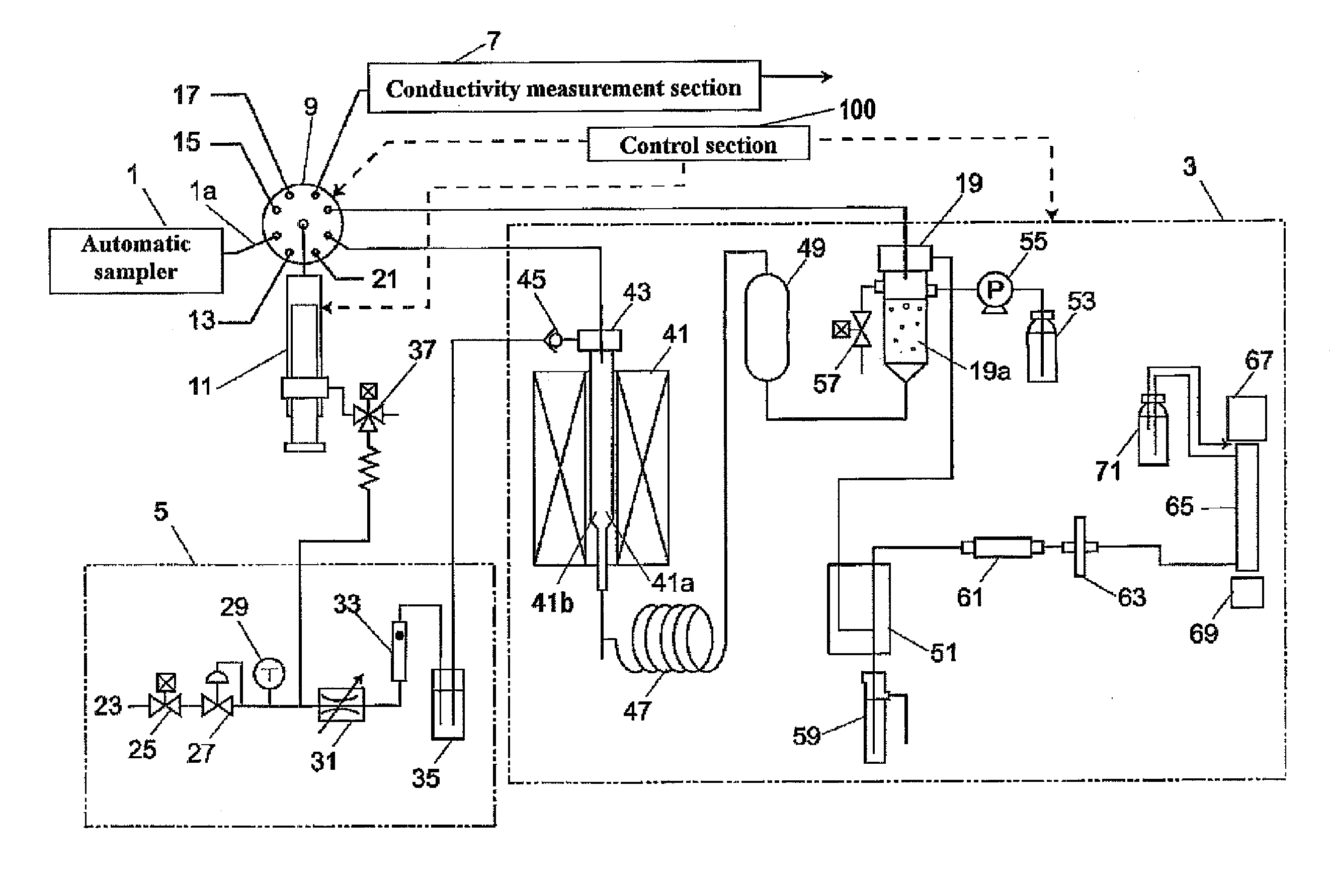
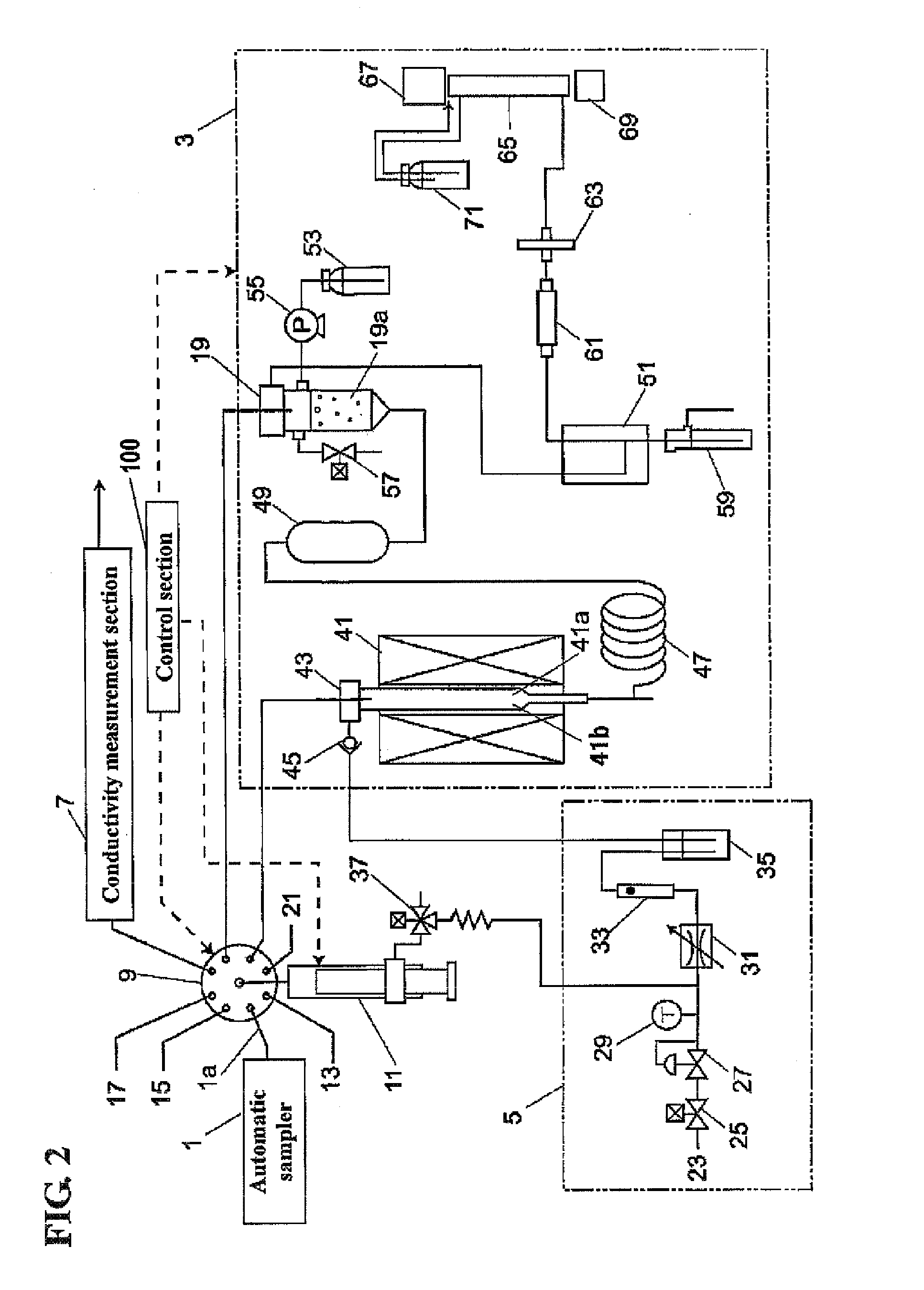
InactiveUS20070254374A1Inhibition of dissolutionEliminate riskBiological testingTesting organic contamination in waterSample waterWater quality
Disclosed is a water quality analyzer, which comprises a total organic carbon (TOC) measurement section for converting a carbon component in an aqueous sample to carbon dioxide and measuring an amount of the carbon dioxide, a conductivity measurement section for measuring a conductivity of an aqueous sample, a gas aeration mechanism for passing a degassing gas devoid of carbon dioxide gas through an aqueous sample to perform a degassing treatment of expelling carbon dioxide gas in the aqueous sample, and a control section for controlling respective operations of TOC measurement and conductivity measurement. The control section is operable, just before measuring a conductivity of a specific aqueous sample during the conductivity measurement for the specific aqueous sample, to controllably instruct the gas aeration mechanism to pass the degassing gas through the specific aqueous sample so as to perform the degassing treatment.
Owner:SHIMADZU CORP
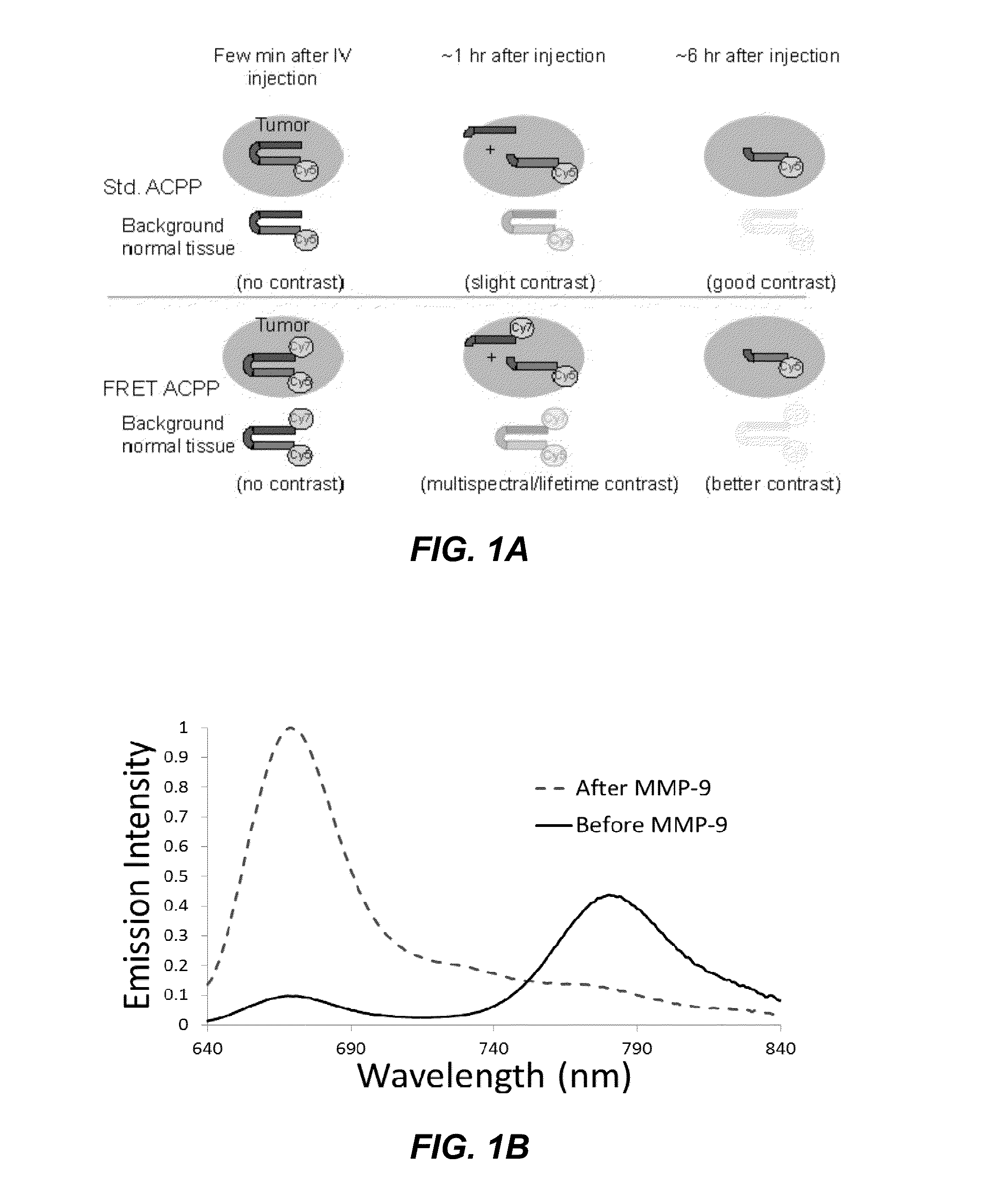
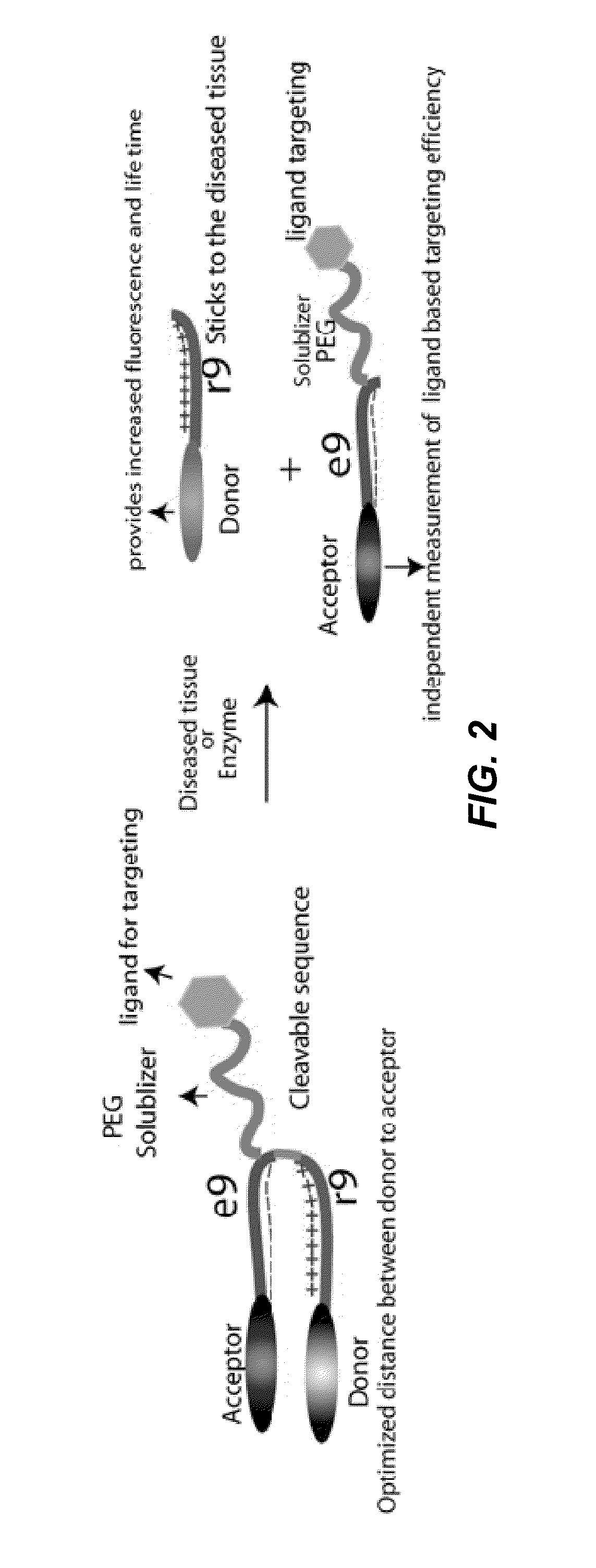
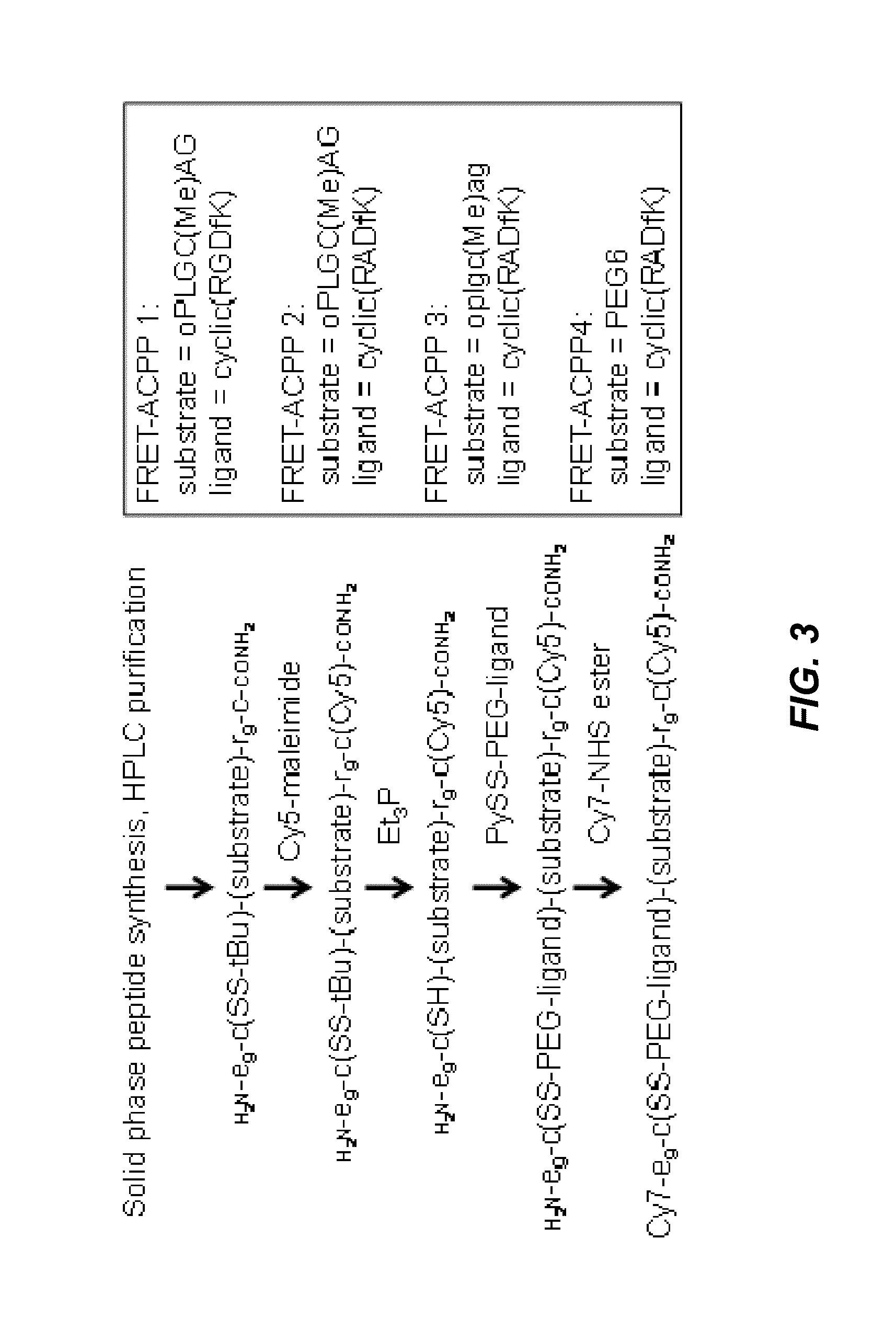
Activatable cell penetrating peptides with quenched fluorophores
ActiveUS20130078188A1Ultrasonic/sonic/infrasonic diagnosticsPeptide/protein ingredientsCyanineFluorophore
The invention provides compositions useful as molecular probes. In particular, the invention provides activatable cell penetrating peptides comprising a fluorescence donor and a fluorescence acceptor. Exemplary fluorescence donors and fluorescence acceptors include compounds derived from cyanine. Also provided are ratiometric, multispectral, and excitation lifetime imaging methods for detecting the molecular probes provided herein.
Owner:RGT UNIV OF CALIFORNIA
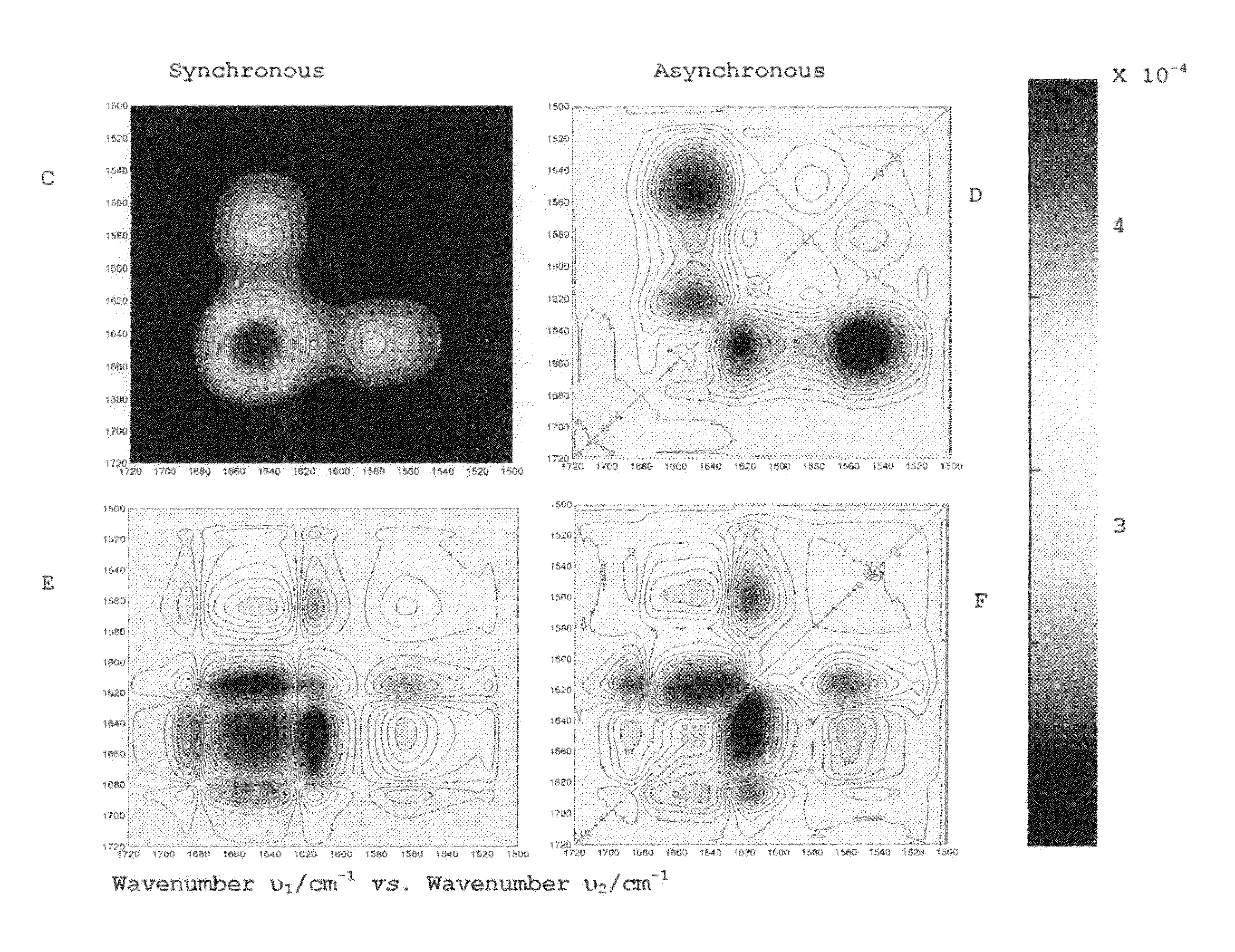
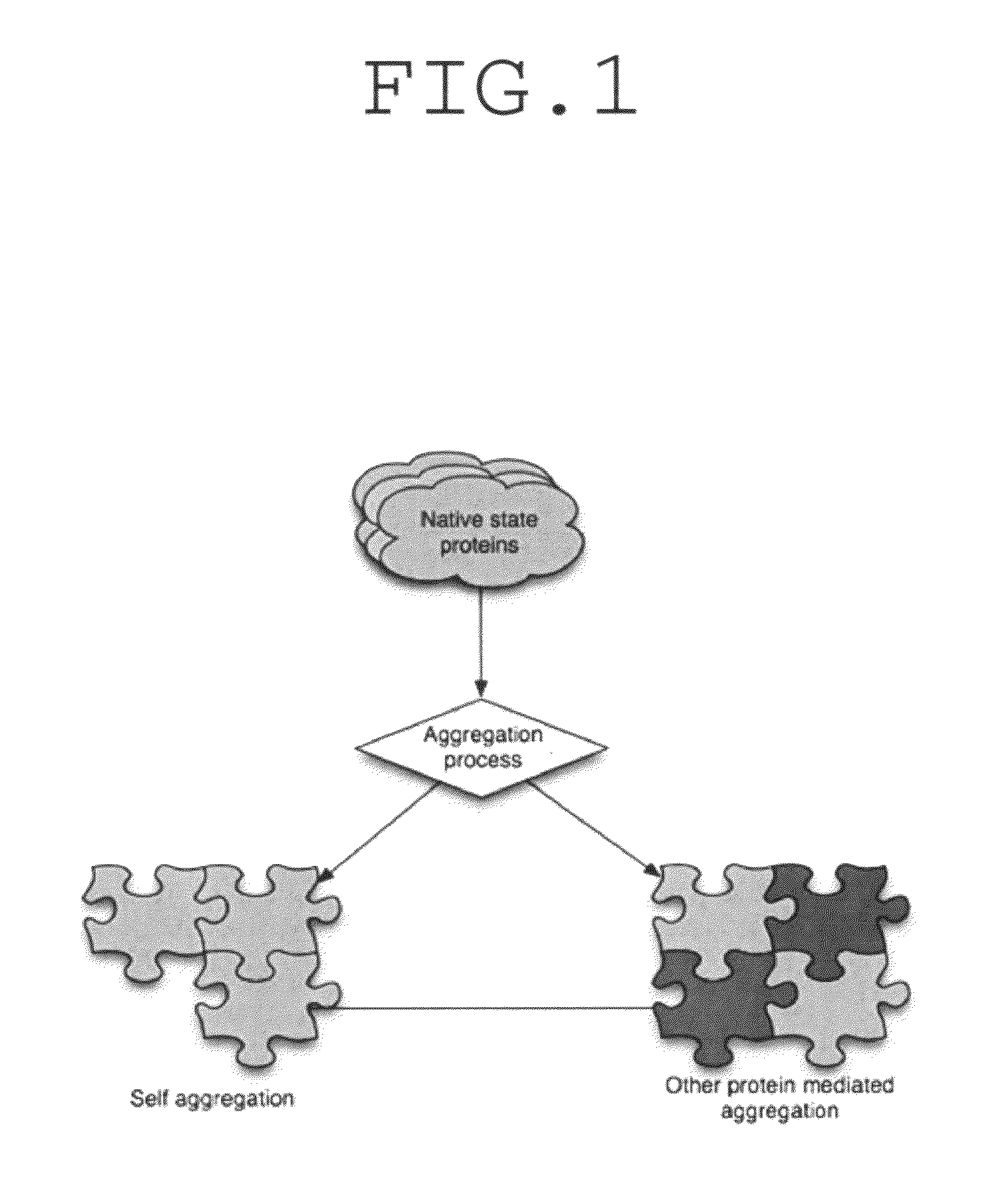
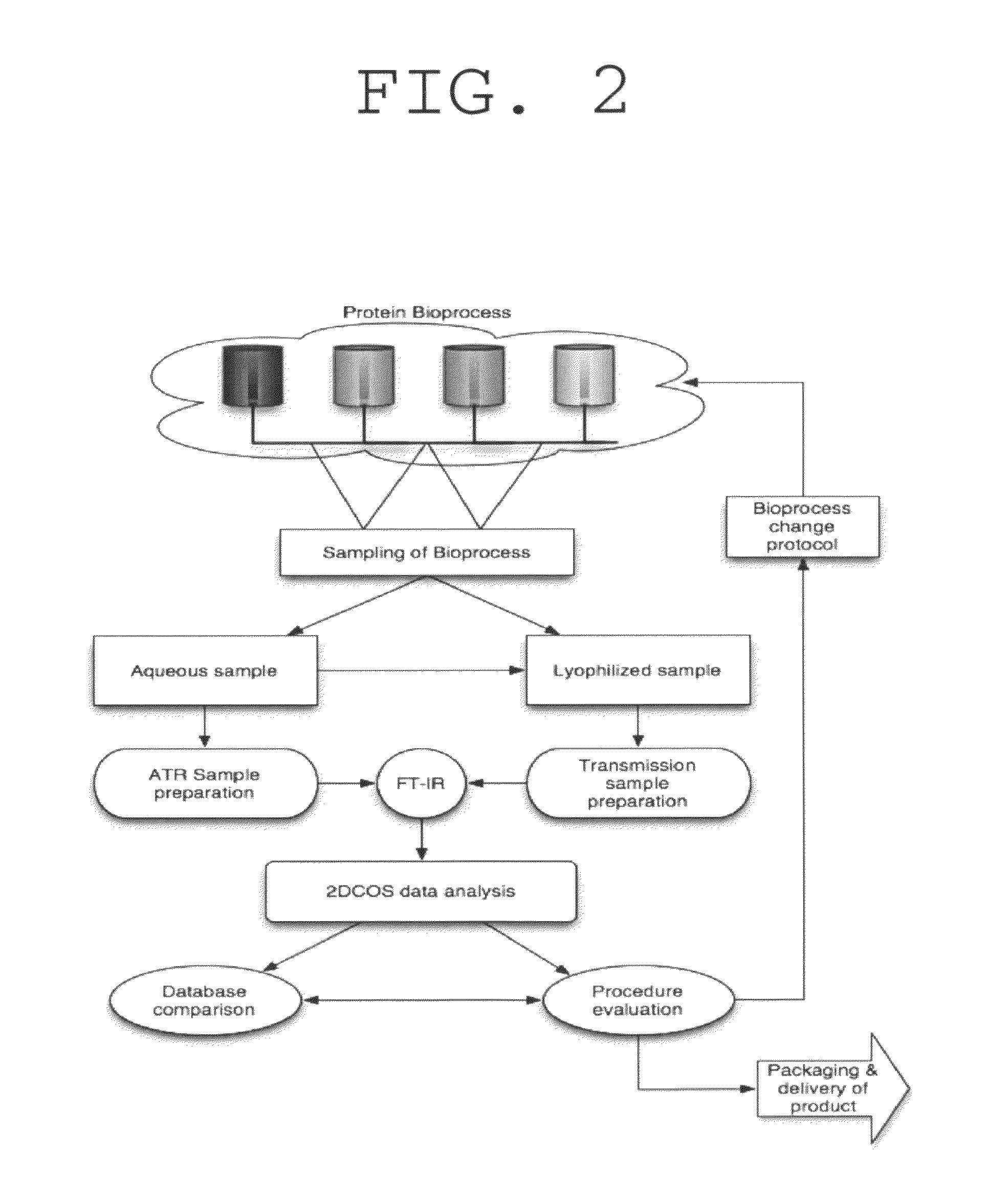
Method for determination of protein, peptide or peptoid aggregation, stability, and viability and system using the same
ActiveUS8268628B1High resolutionEasy to understandMaterial analysis by observing effect on chemical indicatorAnalysis by material excitationCorrelation analysisPeptoid
The invention describes a method for determining aggregation in protein, peptide or peptoid formulation, without the use of probes or additives. The method uses FTIR spectroscopy combined with the two-dimensional correlation analysis (2DCOS) which allows for the determination of the presence of aggregates, the determination of the mechanism of aggregation, allowing for correction in the pipeline manufacturing process of the protein to once again generate viable protein. In addition, the thermal transition of the protein can also be determined and a 2DCOS plot generated to compare with the established viable protein, allowing for quality control, stability and viability of the desired protein product. The ease of sample preparation and data analysis allows for the automation of this method.
Owner:UNIVERSITY OF PUERTO RICO +1
Breath ketone detector
InactiveUS8871521B2Analysis using chemical indicatorsMaterial analysis by observing effect on chemical indicatorKetoacidosisPowder mixture
Ketoacidosis is an extreme and uncontrolled form of ketosis, which is a normal response to prolonged fasting. Embodiments of this invention test the ketone level of a patient by measuring the ketone bodies in breath condensation. Some embodiments include a device for medical testing comprising a hollow container, comprising powder mixture of sodium nitroferricyanide, ammonium sulfate and silica and a liquid including an ammonium hydroxide solution.
Owner:AKERS BIOSCI
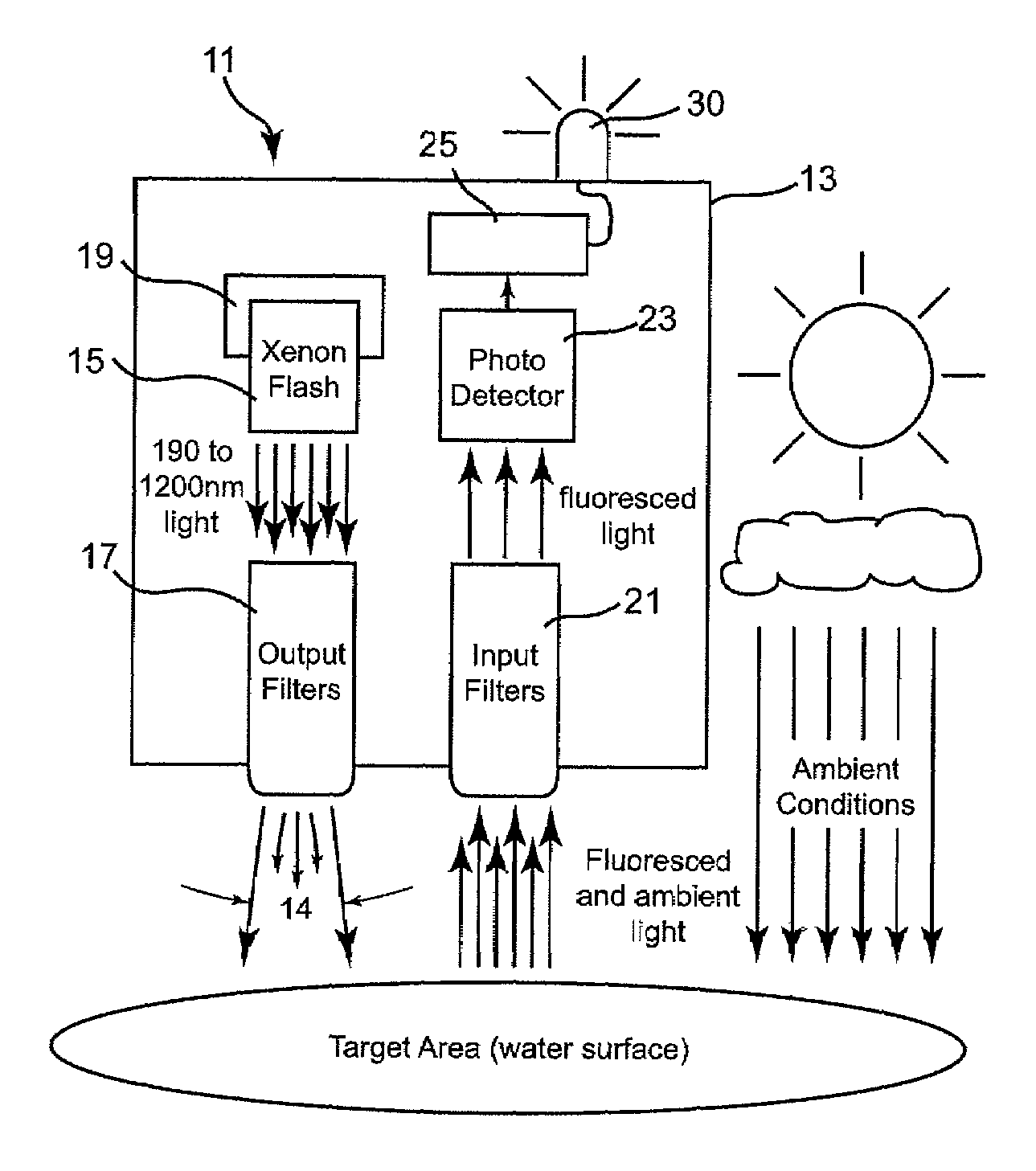
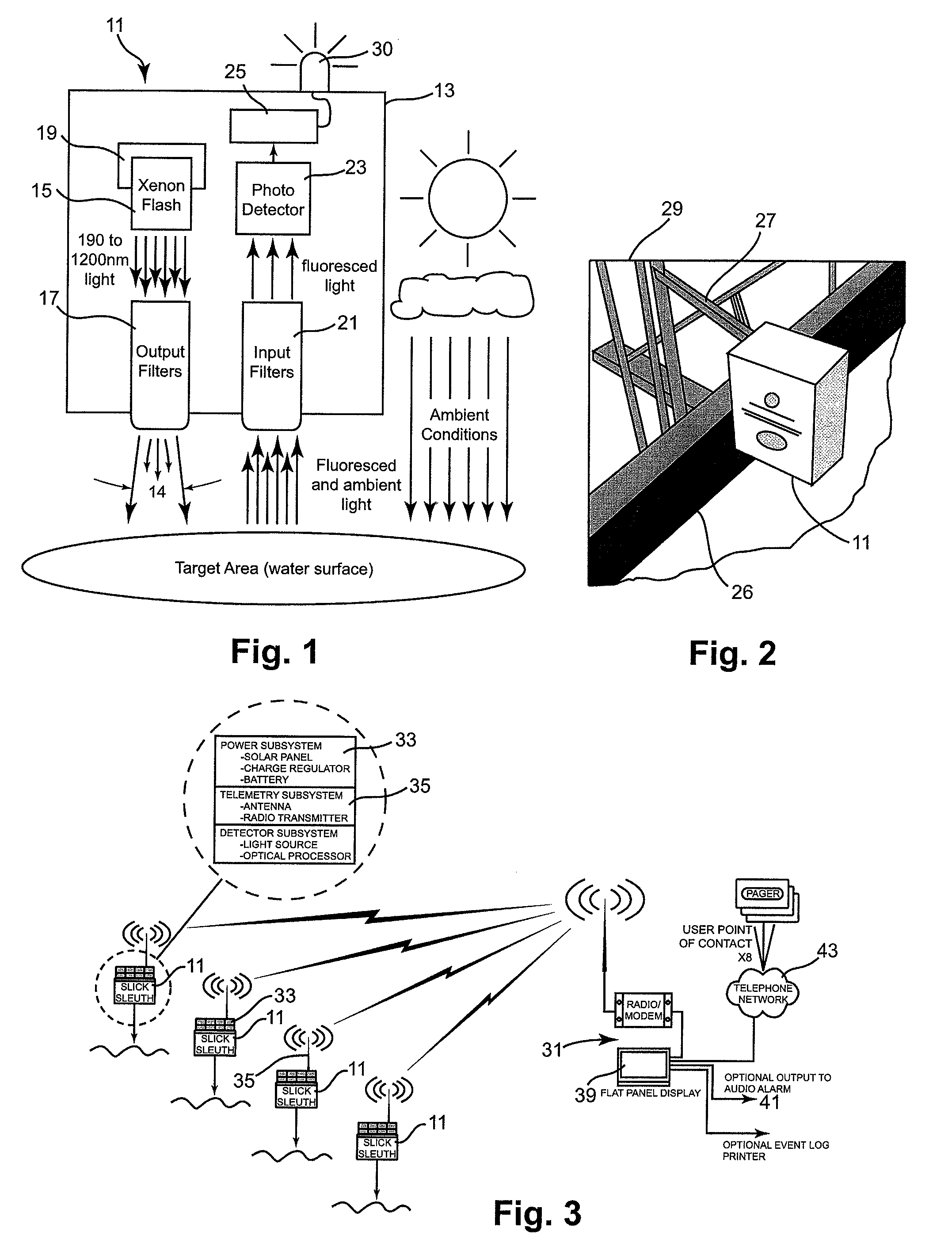
Non-contact oil spill detection apparatus and method
ActiveUS7688428B2Reliably detect signalEasy to operateRaman/scattering spectroscopyLuminescent dosimetersPhotodetectorFuel oil
Apparatus for detecting the presence of a targeted group of hydrocarbons, such as diesel / fuel oil, lube oil, motor oil, hydraulic oil, jet fuel, mineral oil and crude oil in a highly reliable manner even though present at only extremely low concentration. A high power, pulsed light source is focused into a collimated beam that is reduced by a set of filters to a band of pulsed light within a precise set of wavelengths and directed vertically onto a target surface, such as a body of water. All but a precise band of light wavelengths returning to the apparatus are blocked so that substantially all light which then reaches an internal photodetector is within such precise band of wavelengths; as a result receipt of such light programmed intervals following such pulses is indicative of the presence of a member of the targeted hydrocarbon group.
Owner:INTEROCEAN SYST LLC
Device for measuring blood alcohol concentration
InactiveUS9063120B2Quick measurementAccurate measurementPreparing sample for investigationRespiratory organ evaluationBlood collectionAlcohol
A device for measuring blood alcohol concentration for detecting the alcohol concentration from the blood of the unconscious driver as well as the driver's breath, comprises a first body including a coupling recess into which the blood collection module is coupled and a heater for heating the coupled blood collection module, and a second body including a detection probe to penetrate the blood collection module to suck the alcohol gas in the blood collection module, and an alcohol detection sensor to detect the alcohol concentration through the alcohol gas introduced through the detection probe.
Owner:DATECH
Use of disulfonated anthracenes as inert fluorescent tracers
InactiveUS7220382B2Quantity minimizationDetection of fluid at leakage pointSamplingAnthraceneSteam line
The use of an isomer of anthracene disulfonic acid as an inert fluorescent tracer is described and claimed. The utility of this type of inert fluorescent tracer in boiler systems to trace unwanted carryover of boiler water into a steam line is also described and claimed.
Owner:ECOLAB USA INC
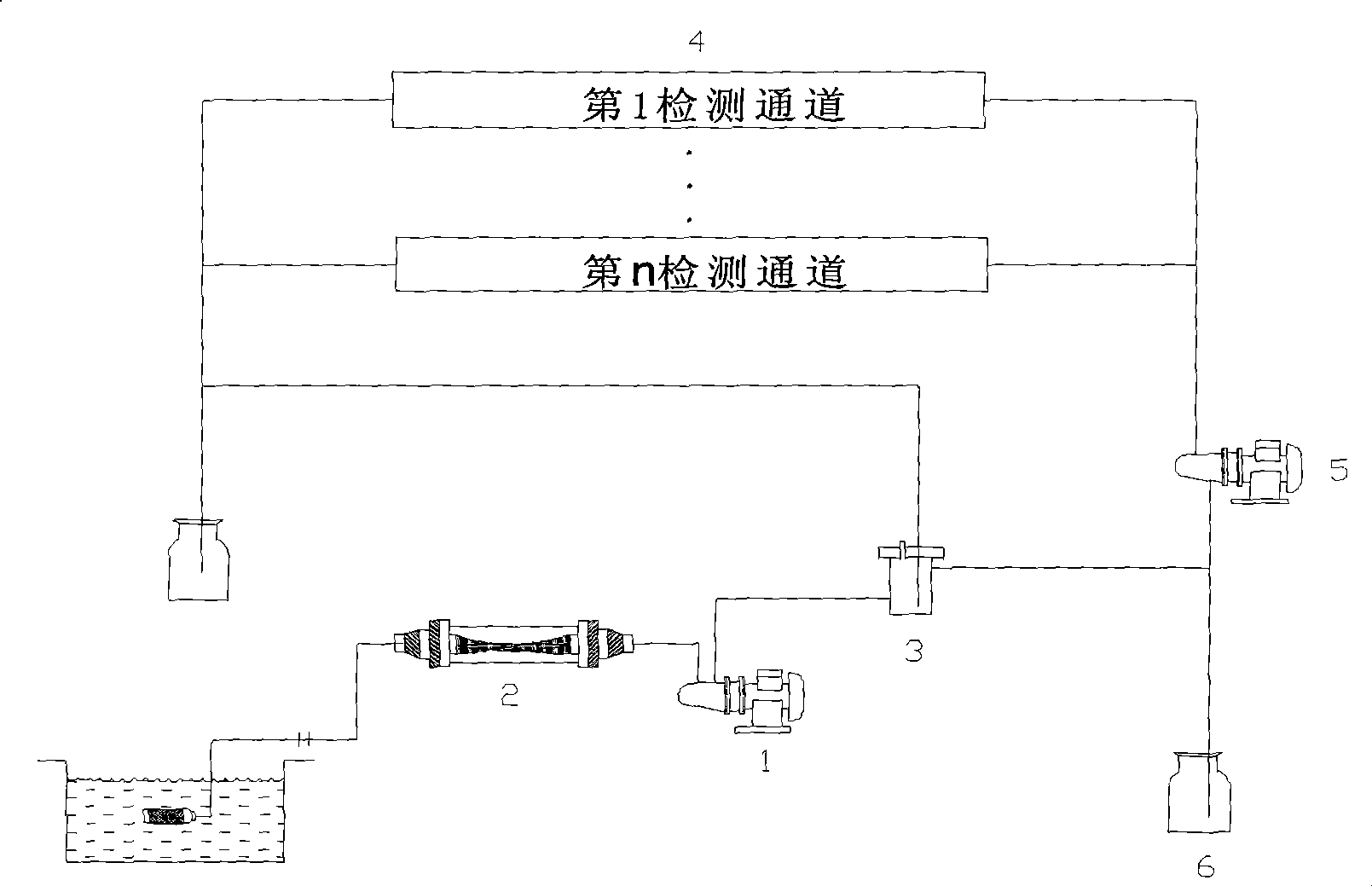
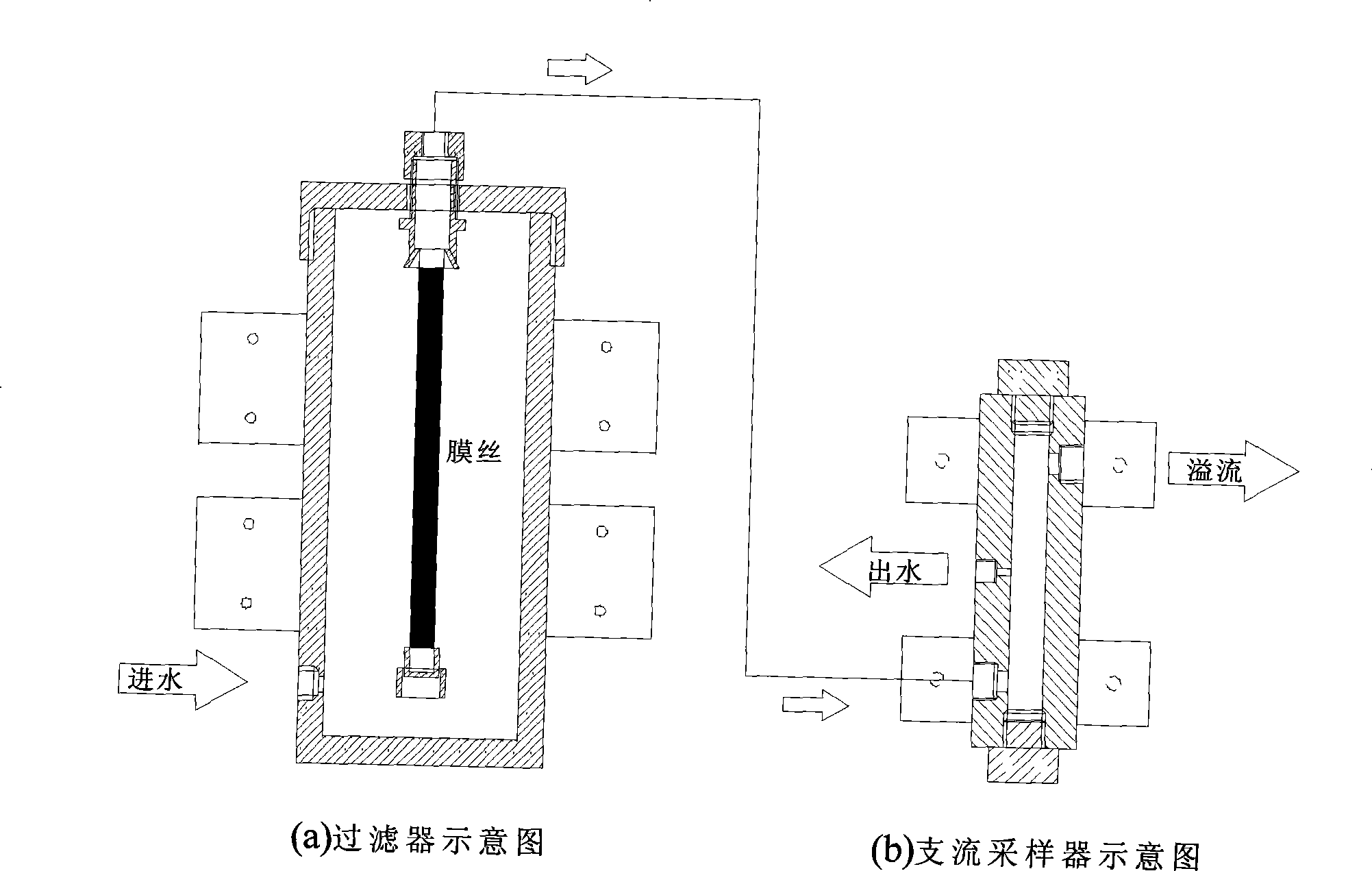
Multichannel photobacteria on-line water quality toxicity monitoring device and monitoring method
InactiveCN101477056AToxicity Rapid TestToxicity testingGeneral water supply conservationChemiluminescene/bioluminescencePeristaltic pumpFiber
The invention discloses a multi-channel photobacteria on-line water quality toxicity monitoring device and a monitoring method thereof. The device comprises a diaphragm water pump, wherein a water inlet of the diaphragm water pump is connected with a membrane filter through a water pipe and then is connected with a water sample pool; a water outlet of the diaphragm water pump is connected with an overflow type branch sampler through a water pipe; a plurality of water sample monitoring modules form a plurality of monitoring channels which are connected to the overflow type branch sampler; waste liquid outlets of the plurality of the water sample monitoring modules are parallelly connected with an external peristaltic pump; and an outlet of the peristaltic pump is connected to a waste liquid tank. The monitoring method comprises the following steps: a water sample is sucked into the membrane filter consisting of a hollow fibrous membrane by the diaphragm pump, suspensions and bacteria in water are filtered, and the water sample enters the overflow type branch sampler. Then the bioluminescent intensity of photobacteria is detected in a real-time mode through a toxicity detection module. After a detection experiment is finished, the external peristaltic pump discharges a waste liquid in a reactor into the waste liquid tank; and the device can realize the on-line detection of biologic toxicity of the water from water sources, municipal sewage, and input water of water works, and has the characteristics of simple operation, convenient maintenance, high automation degree, and so on.
Owner:YANGTZE DELTA REGION INST OF TSINGHUA UNIV ZHEJIANG
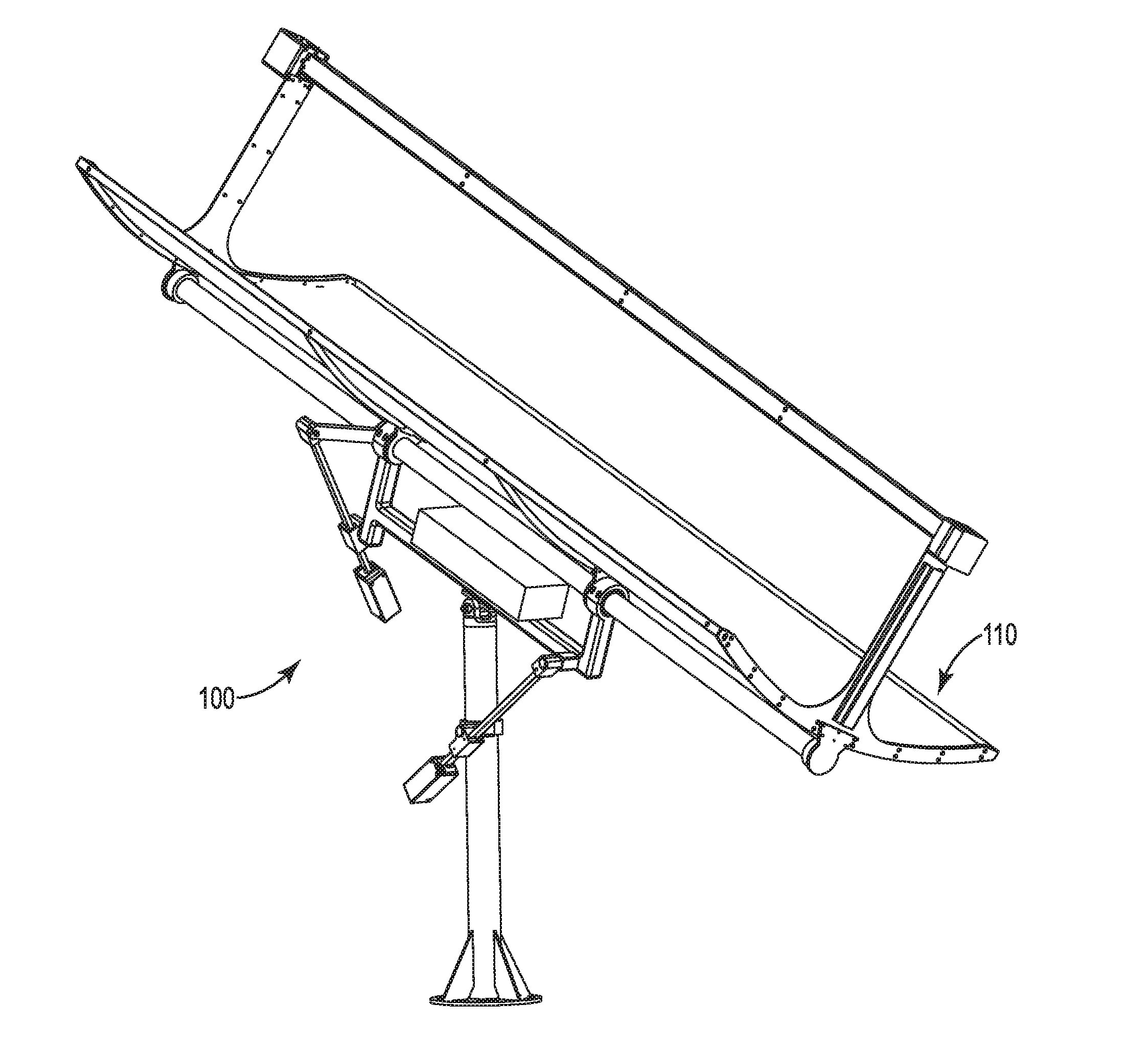
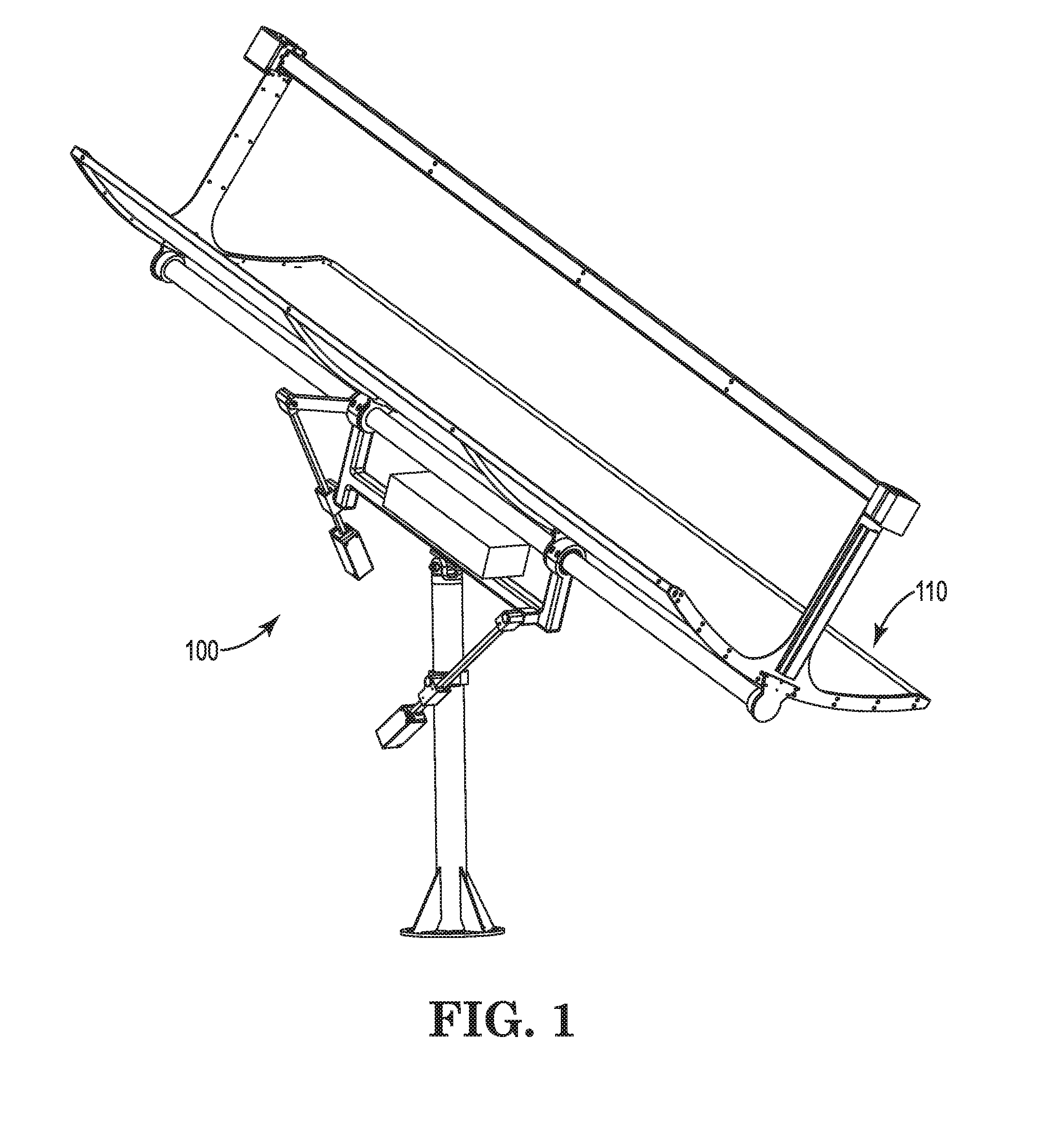
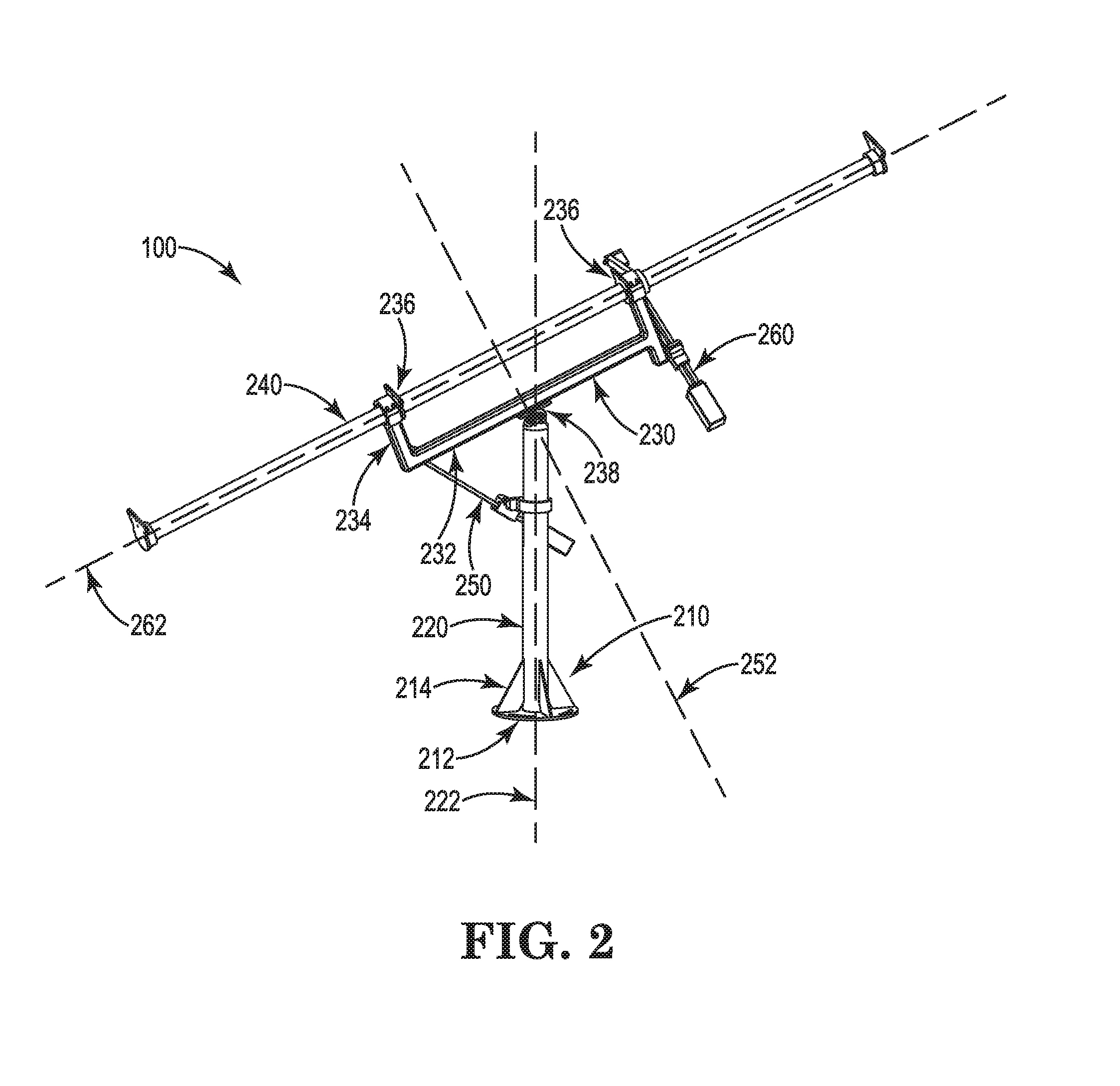
Dual axis tracking device
The disclosure relates to a tracking device configured to track an object in space, such as the sun, as the object moves across the sky. The tracking device may further be configured to direct a payload toward the object or toward an angle relative to the object. The tracking device may continuously or intermittently determine the location of the moving object, and adjust the position of the payload accordingly. The tracking device may calculate the position of the moving object based on GPS information, such as triangulated coordinates of the tracking device, date, and time. Generally, the tracking device may be capable of tracking an object such as the sun from anywhere on the earth's surface. The tracking device may employ one or more actuation assemblies to position the payload toward or relative to the moving object. The one or more actuation assemblies may operate through linear motion.
Owner:WTS
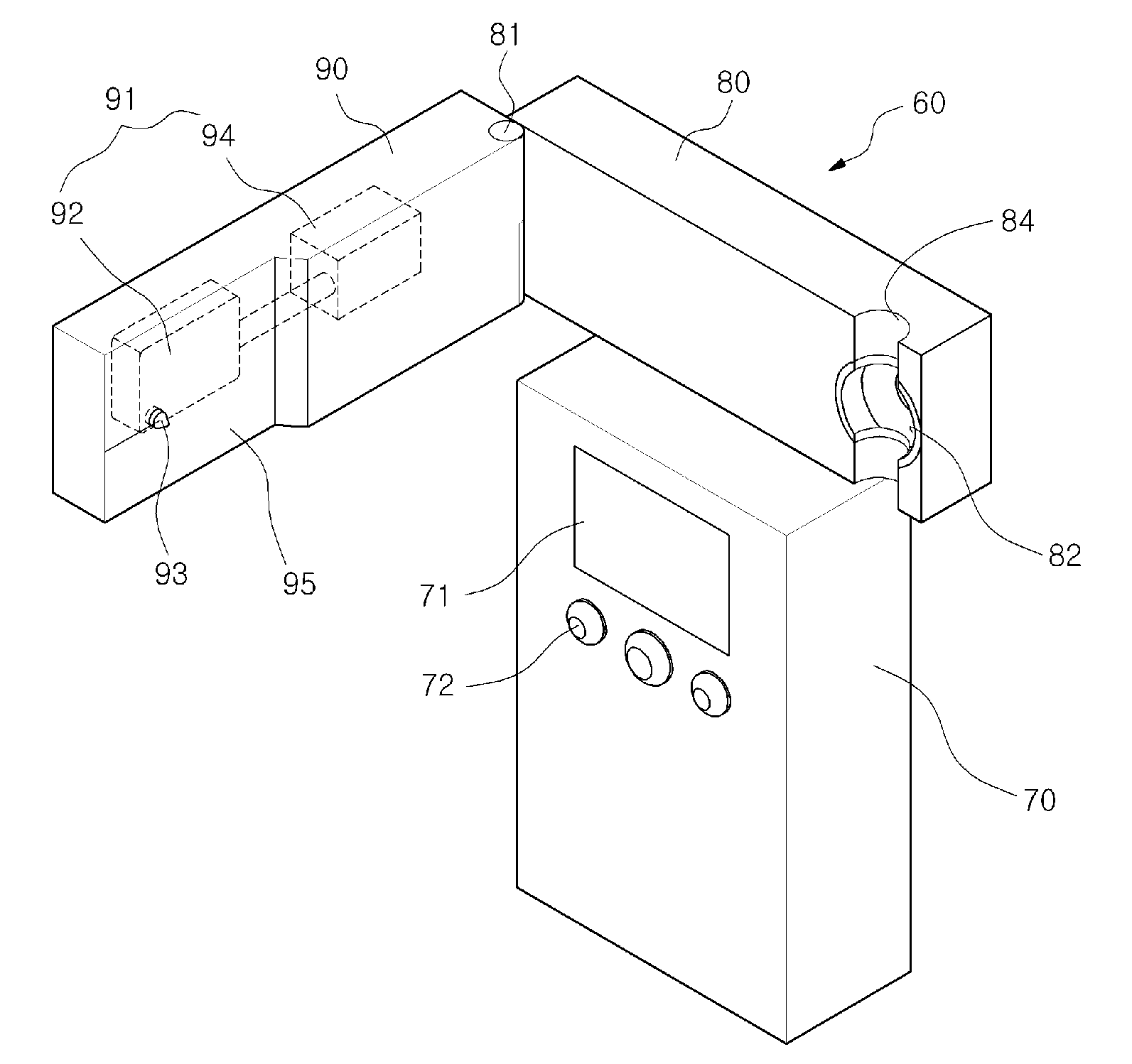
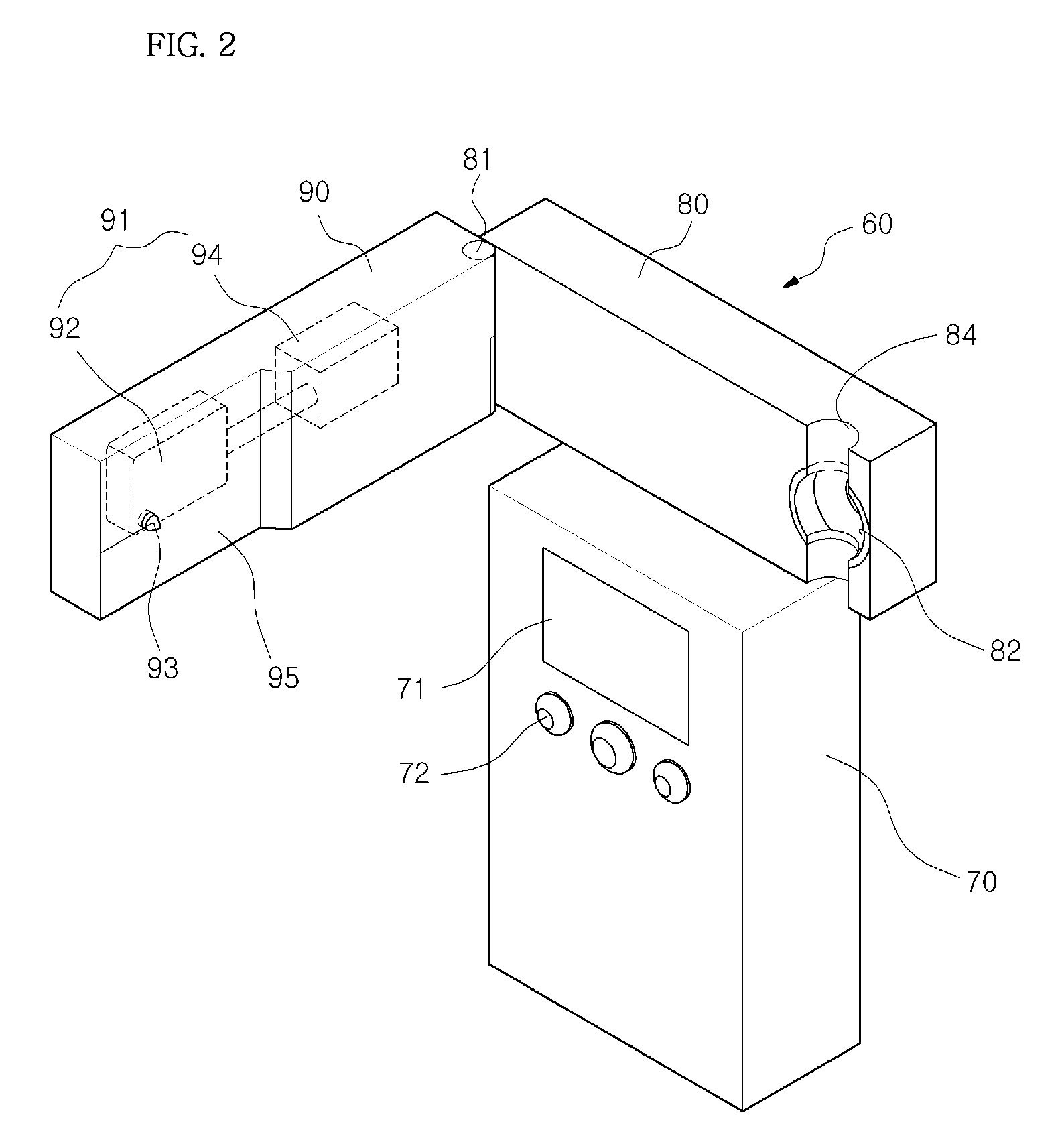
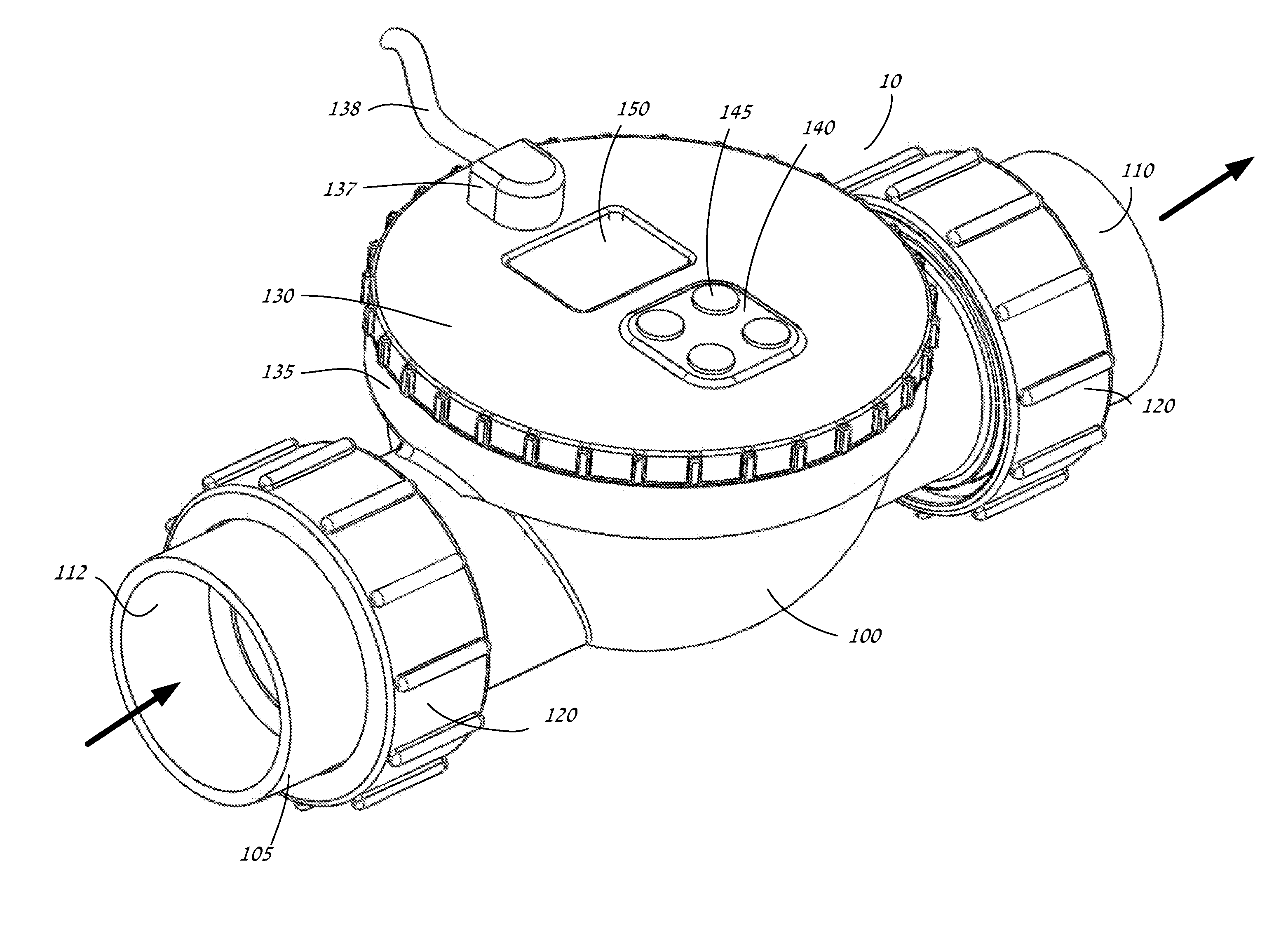
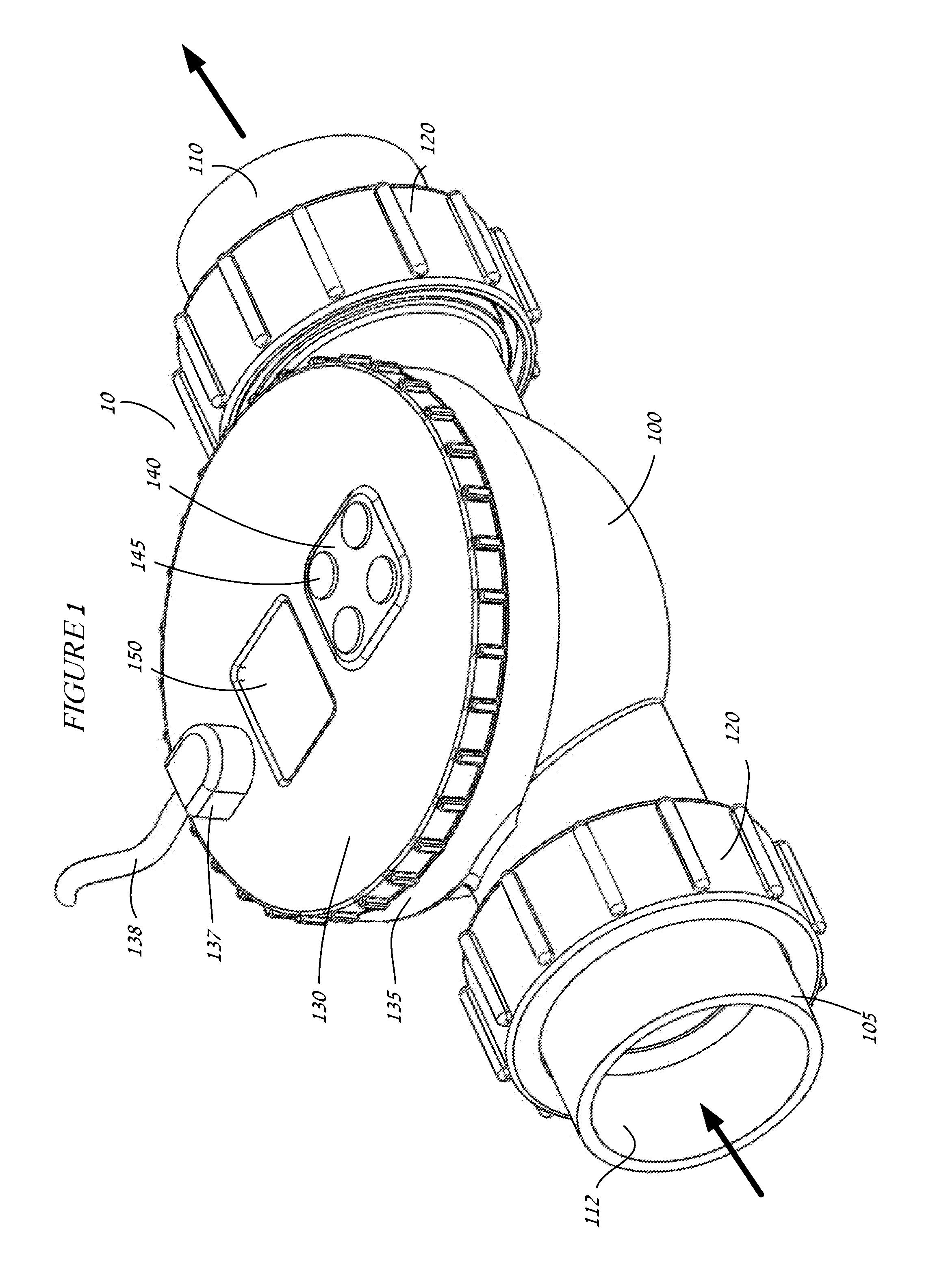
Chemical sensing apparatus having multiple immobilized reagents
InactiveUS20140273052A1More selective in sensing of analytesMore cost-effectivelyBioreactor/fermenter combinationsBiological substance pretreatmentsWater treatment systemLight energy
A sensor system in a water treatment system has a housing, controller, one or more light sources, one or more sensors and one or more targets having an immobilized reagent thereon. Light source emits light energy into the housing that is incident upon the target with the immobilized reagent and the reagent being in contact with water from the system. The immobilized reagent interacts with a reactant in the water such that the interaction changes the state of the reagent. When energy from the light source is incident on the target with the immobilized reagent the energy shows a change detectable by the sensor such that the changed energy is detectable by and collected at the sensor and data on the energy is communicated to the controller. The data is then correlated as a representation of a desired variable to be measured for the water in the water treatment system.
Owner:REDDY RAKESH +2
Wide-range TOC instrument using plasma oxidation
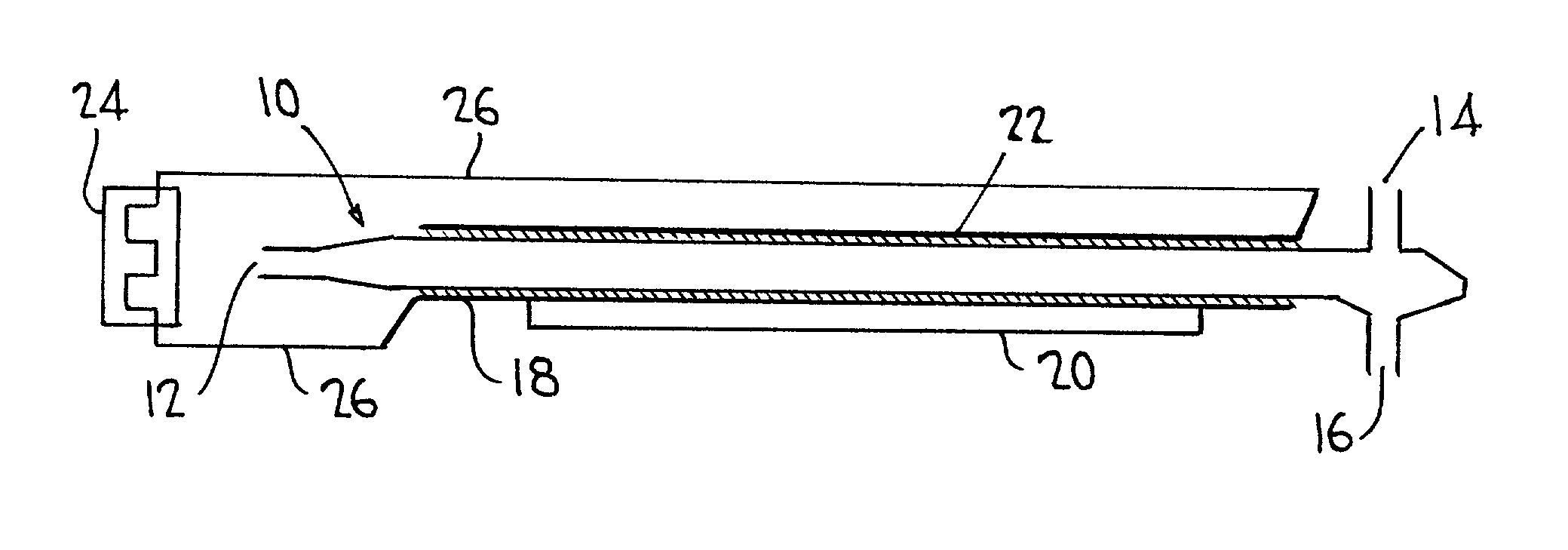
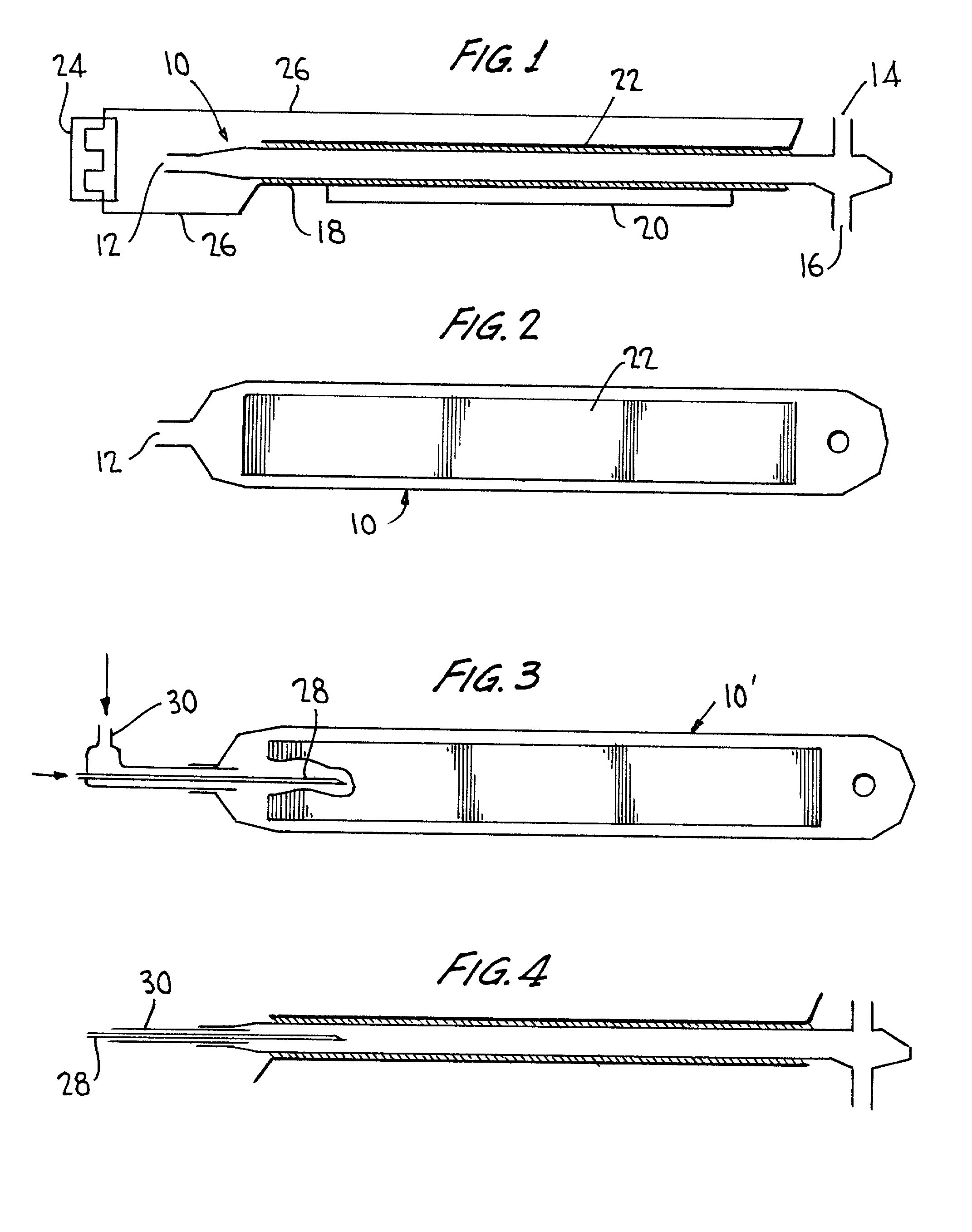
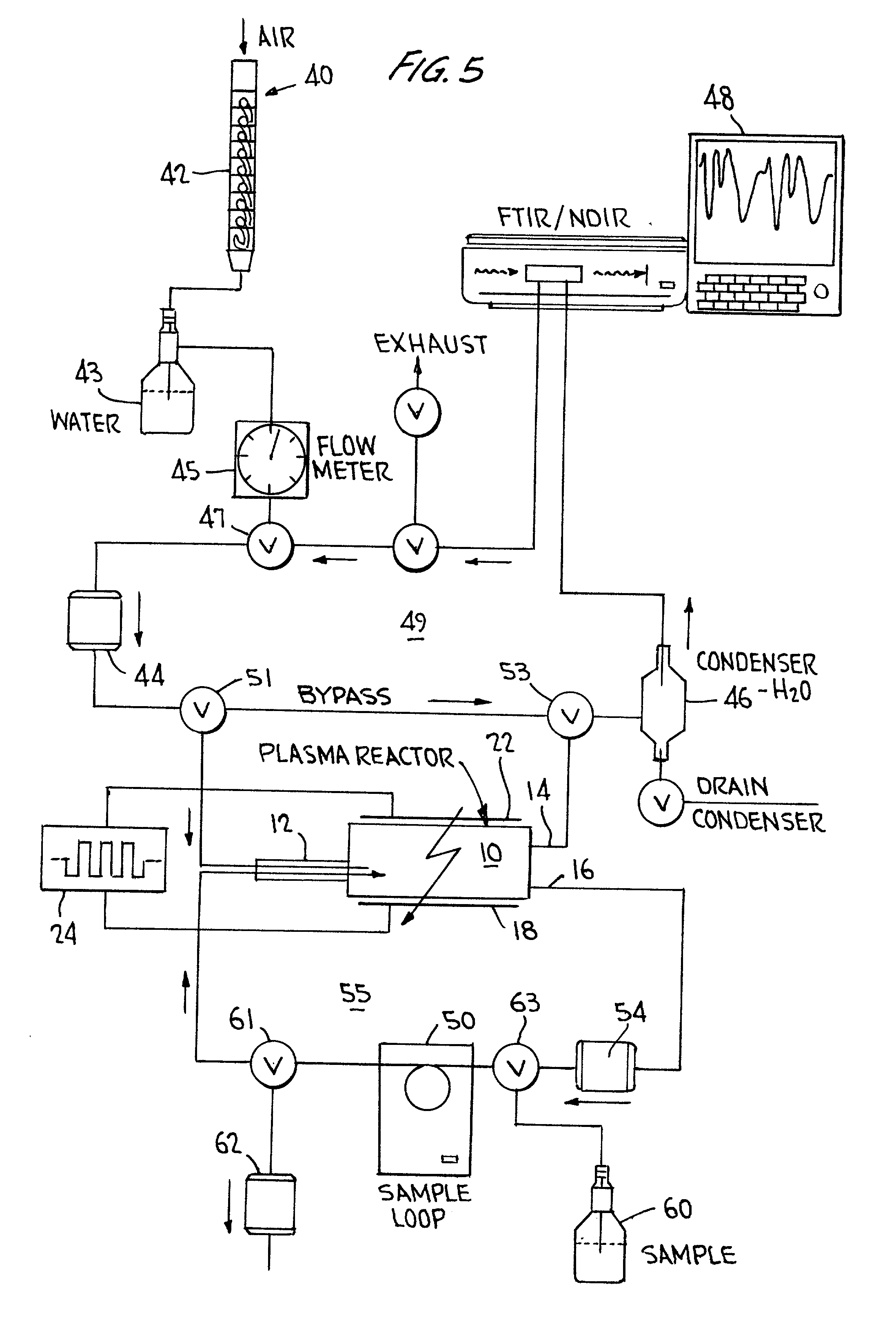
InactiveUS20020068017A1Improve efficiencyShort timeChemical analysis using combustionMaterial analysis using wave/particle radiationConductivityTotal organic carbon
Total organic carbon in an aqueous or gaseous sample is measured by admitting the sample and an oxidant gas to a cell transparent to plasma, establishing a relatively low energy plasma in the cell, and measuring the CO.sub.2 thus produced using FTIR, NDIR, or conductivity-based techniques.
Owner:HACH CO
Apparatuses for interaction with a subterranean formation, and methods of use thereof
Owner:BATTELLE ENERGY ALLIANCE LLC
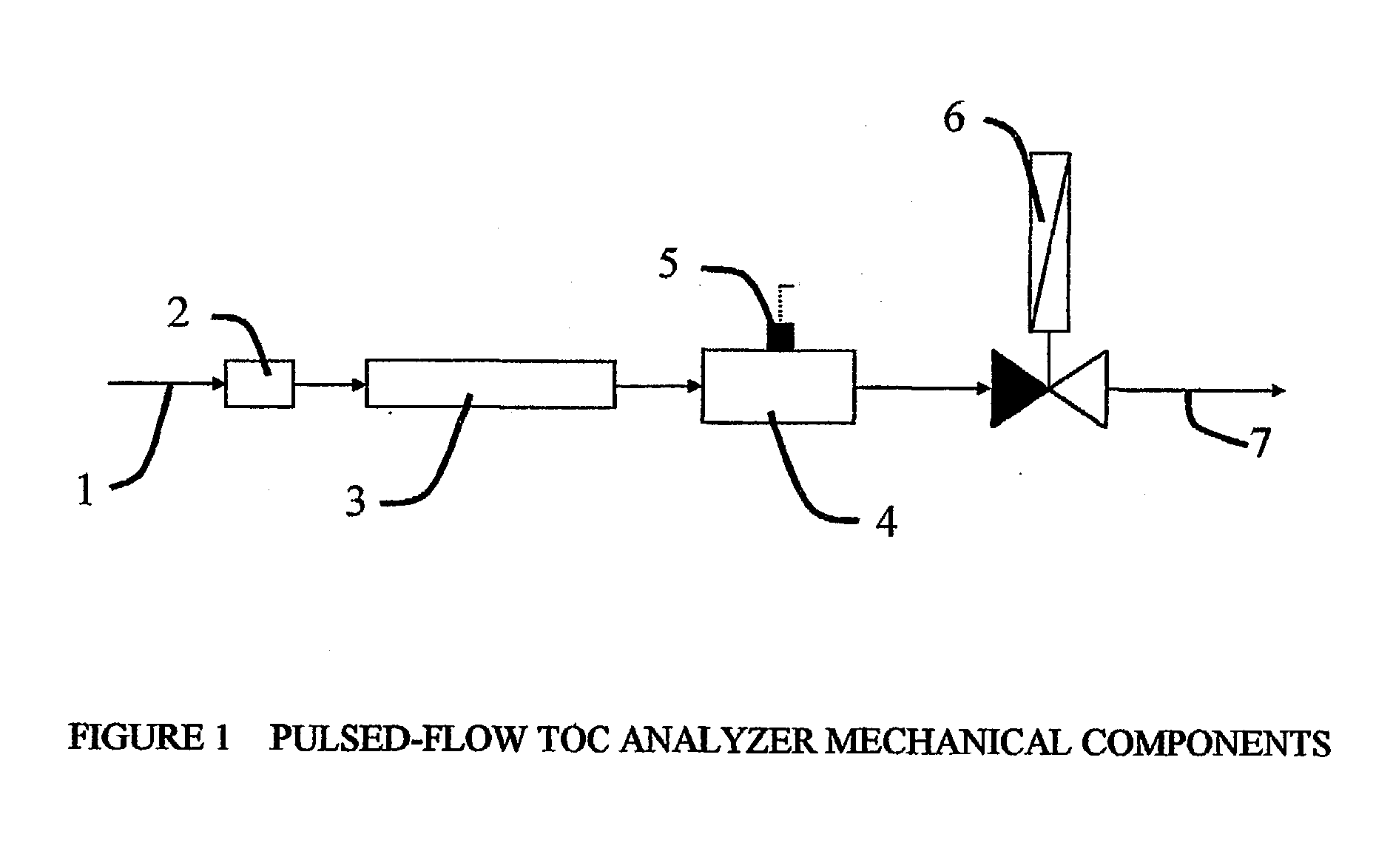
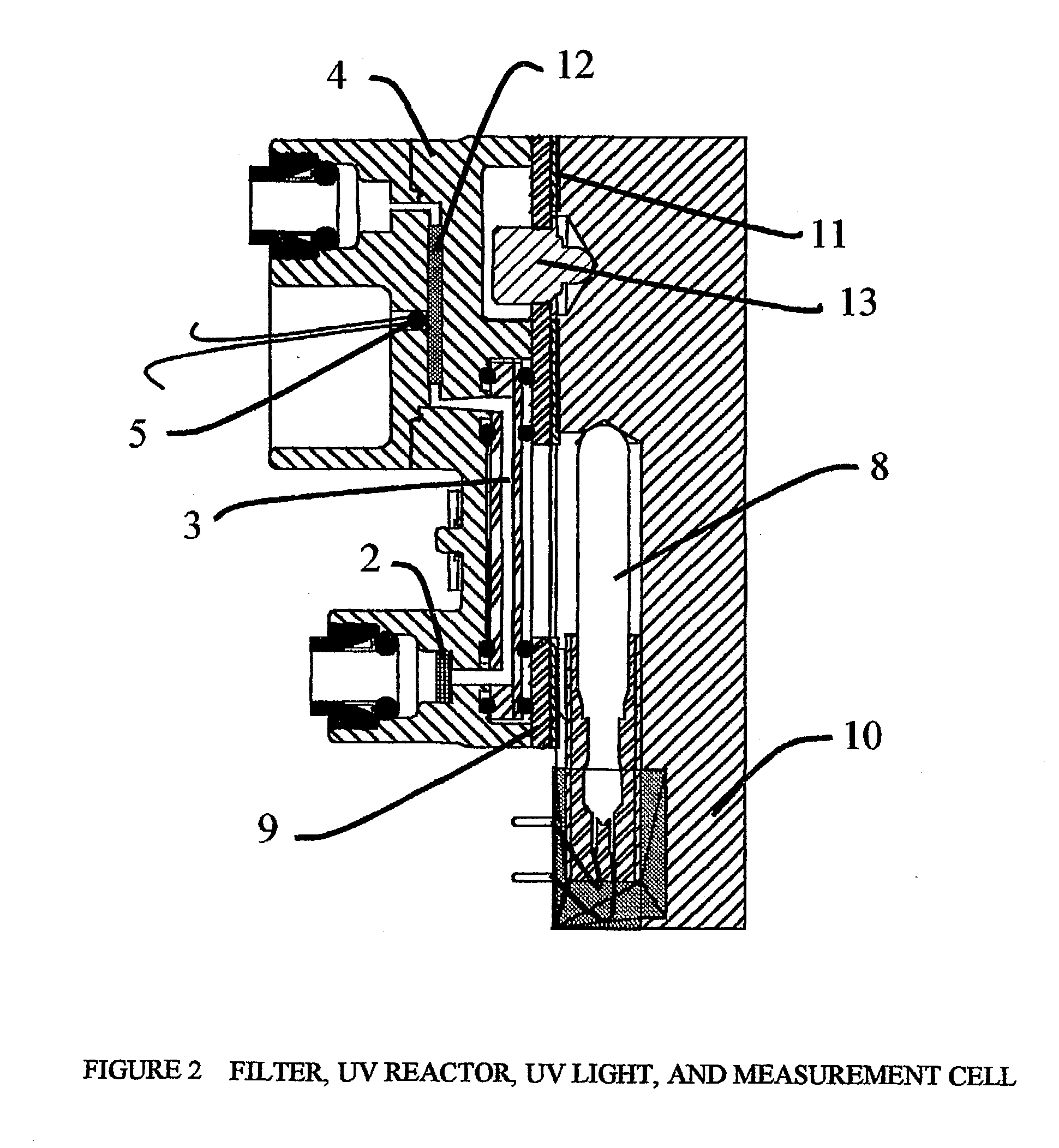
Pulsed-flow total organic carbon analyzer
InactiveUS20030211626A1Improve accuracyAvoid possibilityChemical analysis using combustionBiological testingFluenceLength wave
Methods and apparatus are disclosed for determination of the total concentration of organic carbon compounds in aqueous process streams utilizing a pulsed-flow technique for irradiating a water sample in a chamber (3) with UV or similar wavelength radiation.
Owner:SIEVERS INSTR
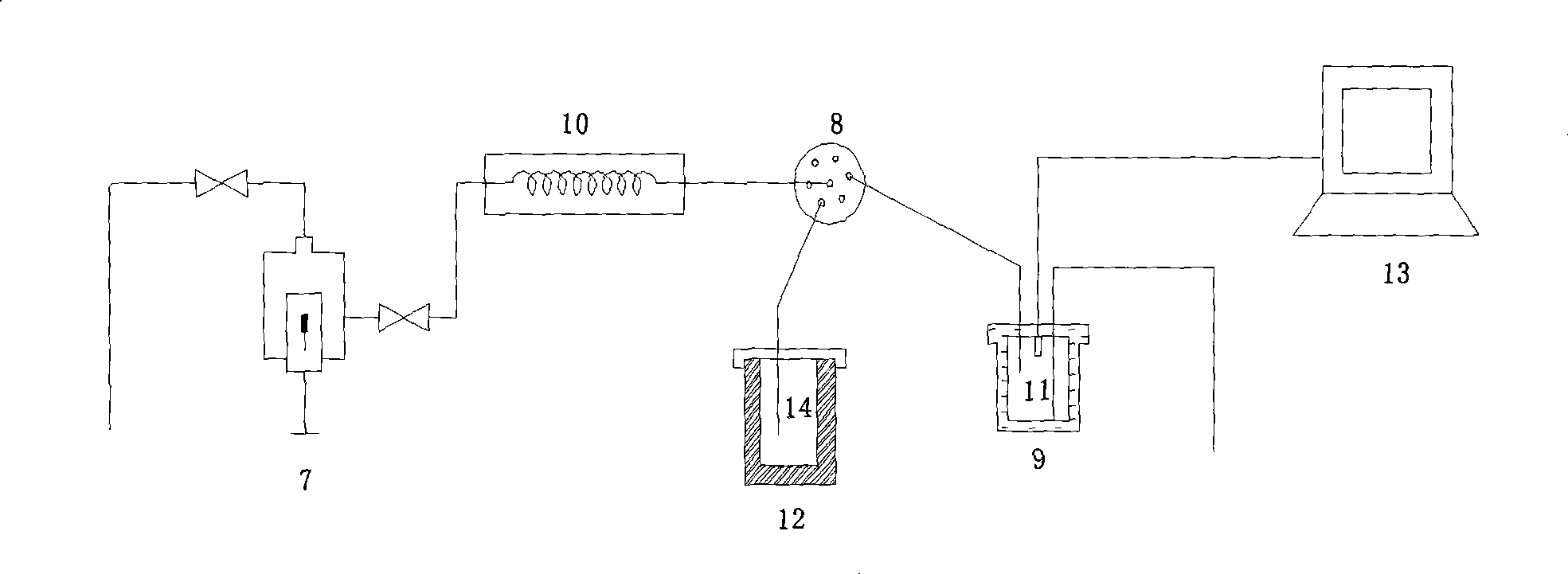
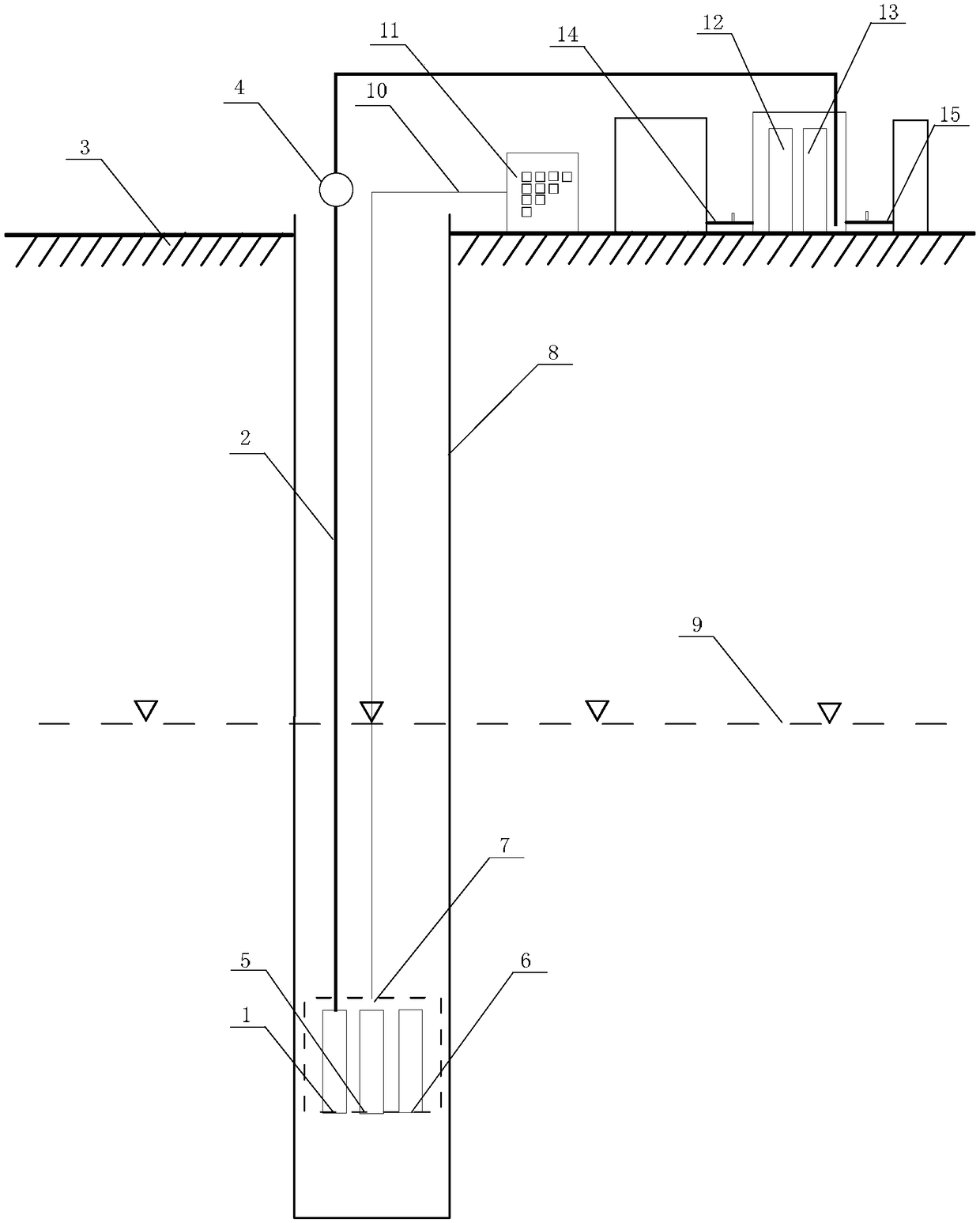
Underground water online monitoring and representative water sample collection integral self-control device and control method thereof
ActiveCN108196021AReduce uncertaintyWithdrawing sample devicesColor/spectral properties measurementsCollection systemWater quality
The invention discloses an underground water online monitoring and representative water sample collection integral self-control device and a control method thereof and relates to the technical field of underground water monitoring and water sample collection. The device comprises a control operation station, a representative water sample judgment collection system and an online in-situ monitoringsystem, wherein the control operation station is arranged on the ground; the online in-situ monitoring system is connected with the control operation station and is arranged under an underground waterlevel in a well; the online in-situ monitoring system is connected with a water pump under the underground water level through a tube; the water pump is connected with a stagnant water collector anda water sample collector through tubes; an online spectrum water quality monitor II and a common parameter water quality analyzer II of the online in-situ monitoring system are used for monitoring andanalyzing water inside the stagnant water collector and the water sample collector. To ensure that fresh water samples of water layers are monitored, and water samples are collected in multiple dimensions from multiple sites to stably judge water sample representativeness according to water quality parameters. Integration of monitoring and sampling techniques is deepened, and in-situ informationacquisition uncertainty is greatly reduced.
Owner:INST OF HYDROGEOLOGY & ENVIRONMENTAL GEOLOGY CHINESE ACAD OF GEOLOGICAL SCI
Underwater vehicle and sensor
Described herein is an underwater vehicle having a vehicle body with a buoyancy controller adapted to vary the buoyancy of the vehicle in order to control motion of the vehicle through an underwater environment. The vehicle further includes a sampling system and a sensor arrangement. The sampling system is adapted to sequentially sample fluid from the underwater environment at specified sampling times resulting in a sample sequence, each sample associated with a sample time and a fluid flow rate. The sensor arrangement includes a plurality of molecule sensors adapted to sense organic molecules in each respective sample of the sample sequence.
Owner:CHEVROU USA INC
Colorimetric detection of cyanide with a chromogenic oxazine
Owner:UNIV OF MIAMI +1
Total organic carbon (toc) analyzer
ActiveUS20050226774A1Chemical analysis using combustionMaterial analysis by observing effect on chemical indicatorBoronAqueous solution
The invention disclosed is a total organic carbon (TOC) analyzer comprised of an electrochemical cell comprising a diamond-film electrode (2) doped with boron or other conductivity including material. The diamond-film electrode is the working electrode and carries out the oxidation of TOC to produce carbon dioxide. The apparatus further comprises sensors for detecting the carbon dioxide produced. Such sensors include but are not limited to a tunable diode laser (1) and / or ion-selective electrode (5). The invention also discloses a method for measuring TOC in an aqueous solution using a total organic carbon analyzer.
Owner:TRUSTEES OF TUFTS COLLEGE TUFTS UNIV
Features
- R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
Why Patsnap Eureka
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Social media
Patsnap Eureka Blog
Learn More Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com