Patents
Literature
934results about "Silve compounds" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
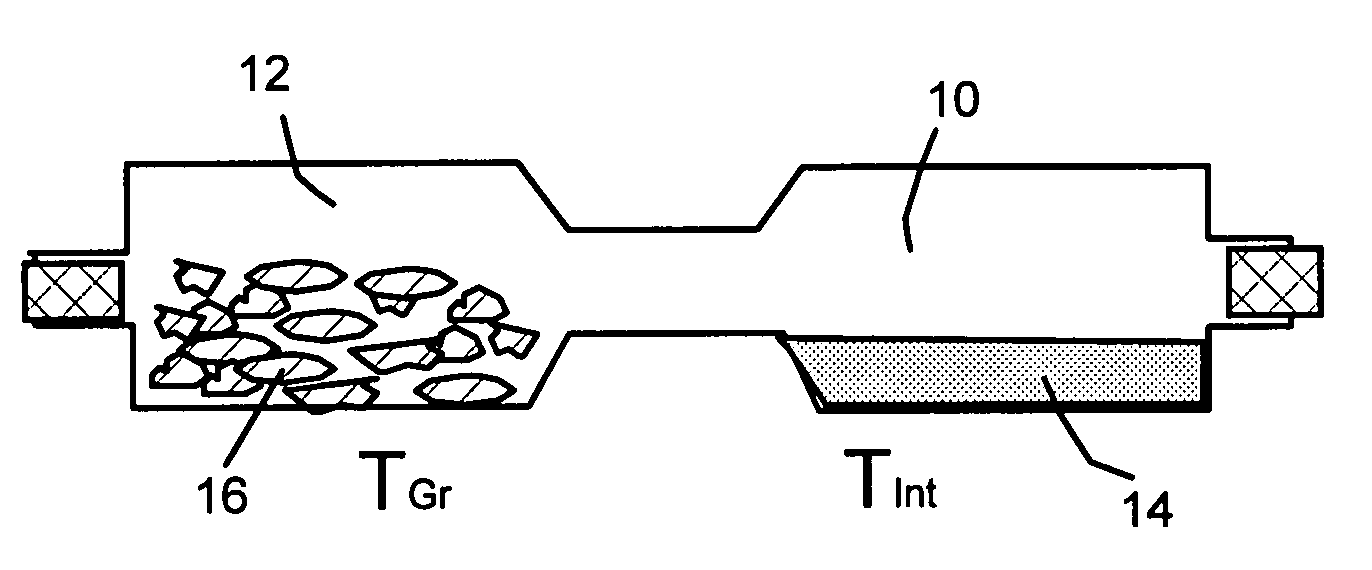
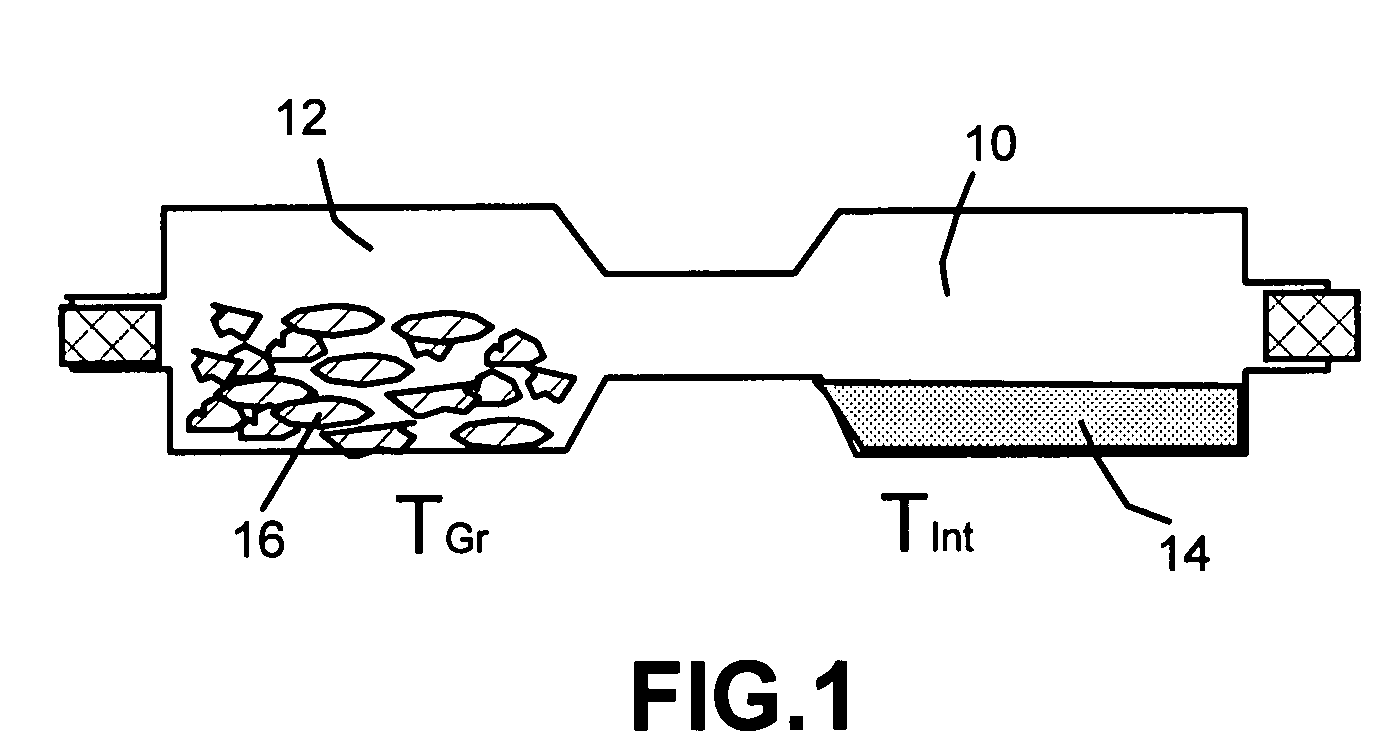
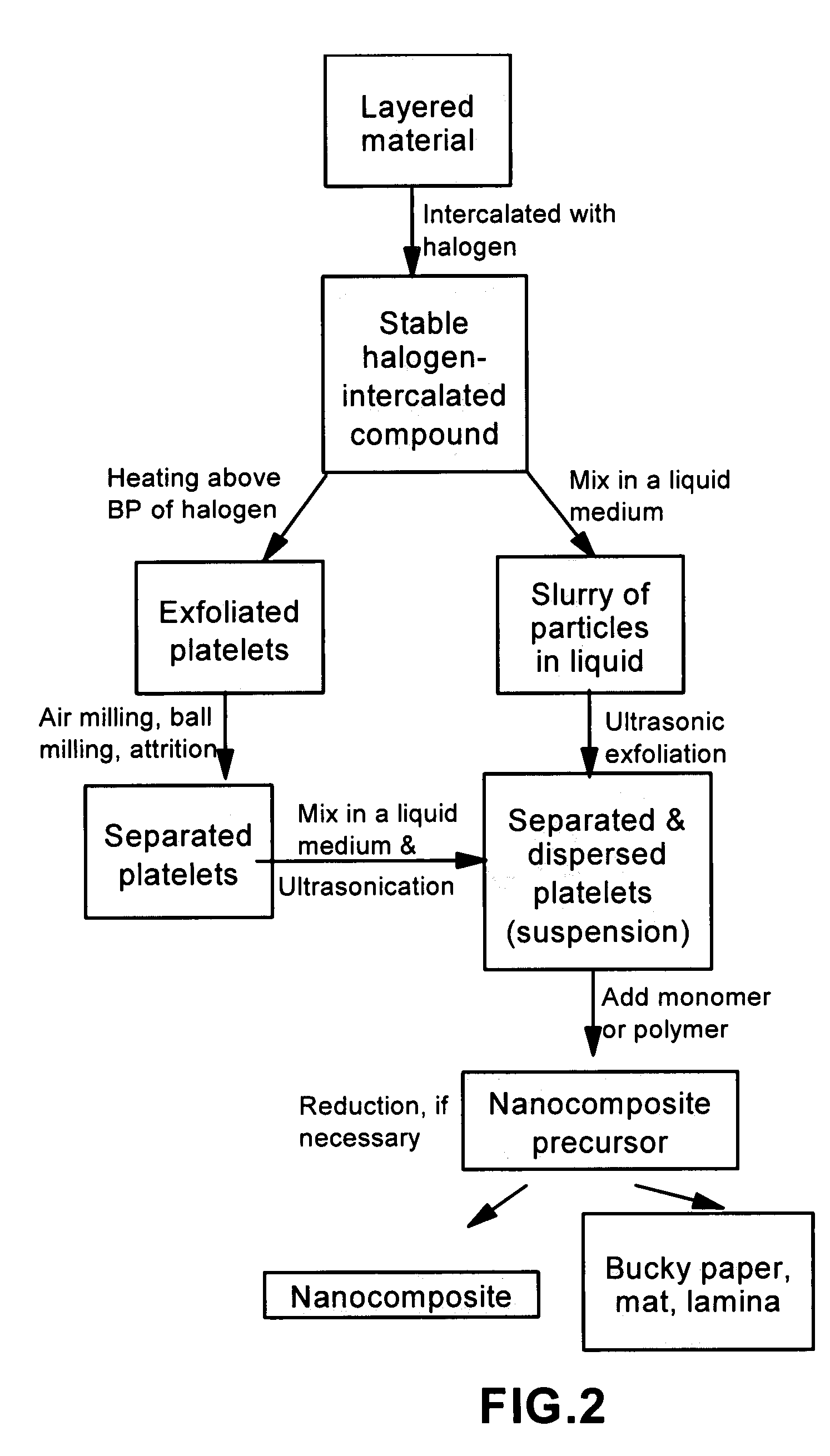
Method of producing nano-scaled graphene and inorganic platelets and their nanocomposites
ActiveUS20080206124A1Readily captured and re-usedReduce impactCarbon compoundsSelenium/tellurium compundsLiquid mediumPhysical chemistry
Disclosed is a method of exfoliating a layered material (e.g., graphite and graphite oxide) to produce nano-scaled platelets having a thickness smaller than 100 nm, typically smaller than 10 nm, and often between 0.34 nm and 1.02 nm. The method comprises: (a) subjecting the layered material in a powder form to a halogen vapor at a first temperature above the melting point or sublimation point of the halogen at a sufficient vapor pressure and for a duration of time sufficient to cause the halogen molecules to penetrate an interlayer space of the layered material, forming a stable halogen-intercalated compound; and (b) heating the halogen-intercalated compound at a second temperature above the boiling point of the halogen, allowing halogen atoms or molecules residing in the interlayer space to exfoliate the layered material to produce the platelets. Alternatively, rather than heating, step (a) is followed by a step of dispersing the halogen-intercalated compound in a liquid medium which is subjected to ultrasonication for exfoliating the halogen-intercalated compound to produce the platelets, which are dispersed in the liquid medium. The halogen can be readily captured and re-used, thereby significantly reducing the impact of halogen to the environment. The method can further include a step of dispersing the platelets in a polymer or monomer solution or suspension as a precursor step to nanocomposite fabrication.
Owner:GLOBAL GRAPHENE GRP INC
Catalysts, activating agents, support media, and related methodologies useful for making catalyst systems especially when the catalyst is deposited onto the support media using physical vapor deposition
InactiveUS20050095189A1Improve performanceEasy to useMaterial nanotechnologyInternal combustion piston enginesGas phaseAdditive ingredient
Use of physical vapor deposition methodologies to deposit nanoscale gold on activating support media makes the use of catalytically active gold dramatically easier and opens the door to significant improvements associated with developing, making, and using gold-based, catalytic systems. The present invention, therefore, relates to novel features, ingredients, and formulations of gold-based, heterogeneous catalyst systems generally comprising nanoscale gold deposited onto a nanoporous support.
Owner:3M INNOVATIVE PROPERTIES CO
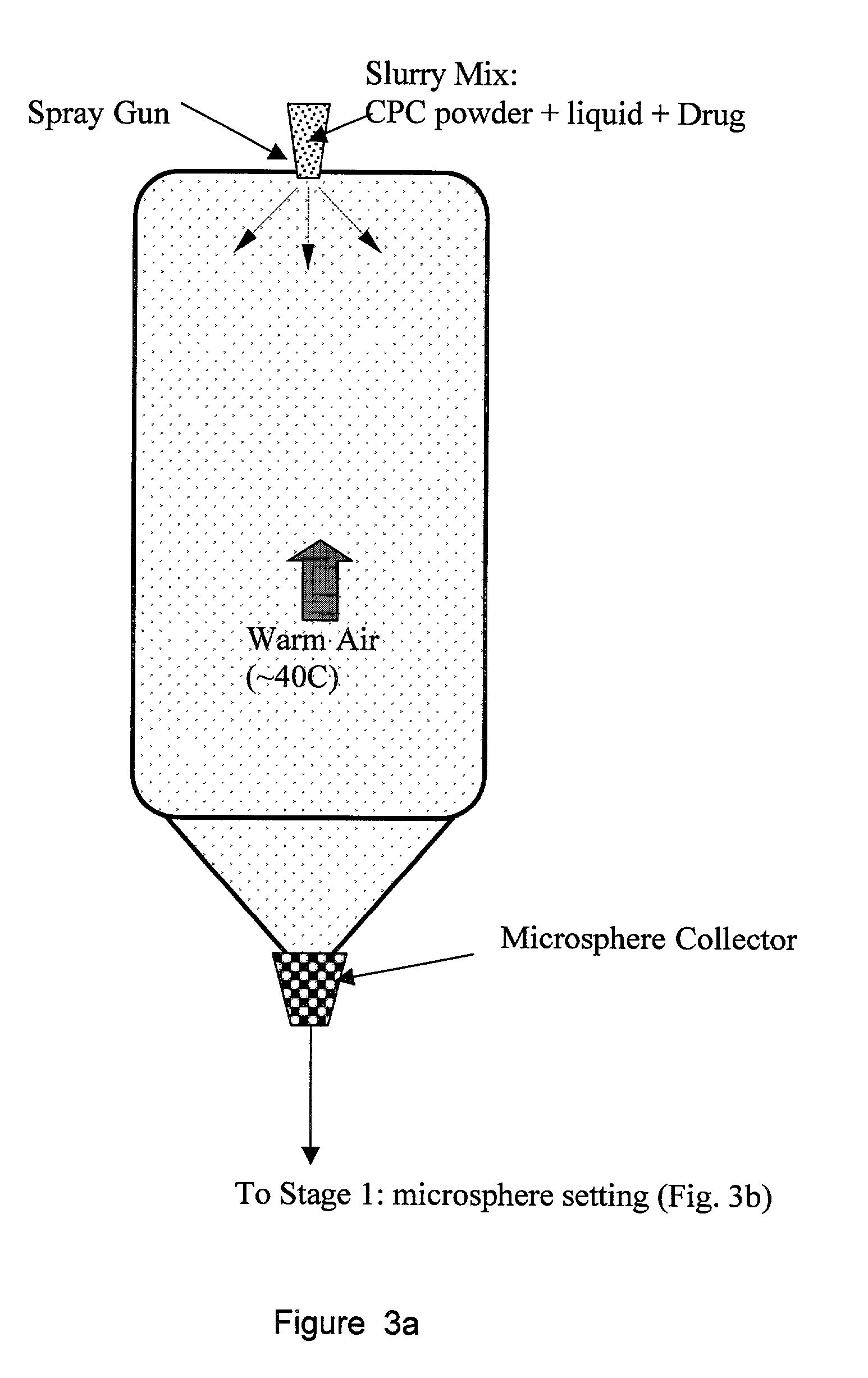
Biofunctional hydroxyapatite coatings and microspheres for in-situ drug encapsulation
InactiveUS20020155144A1Improve relationshipImprove interface strengthPowder deliveryOrganic active ingredientsGene deliverySide effect
Owner:THE UNIV OF BRITISH COLUMBIA
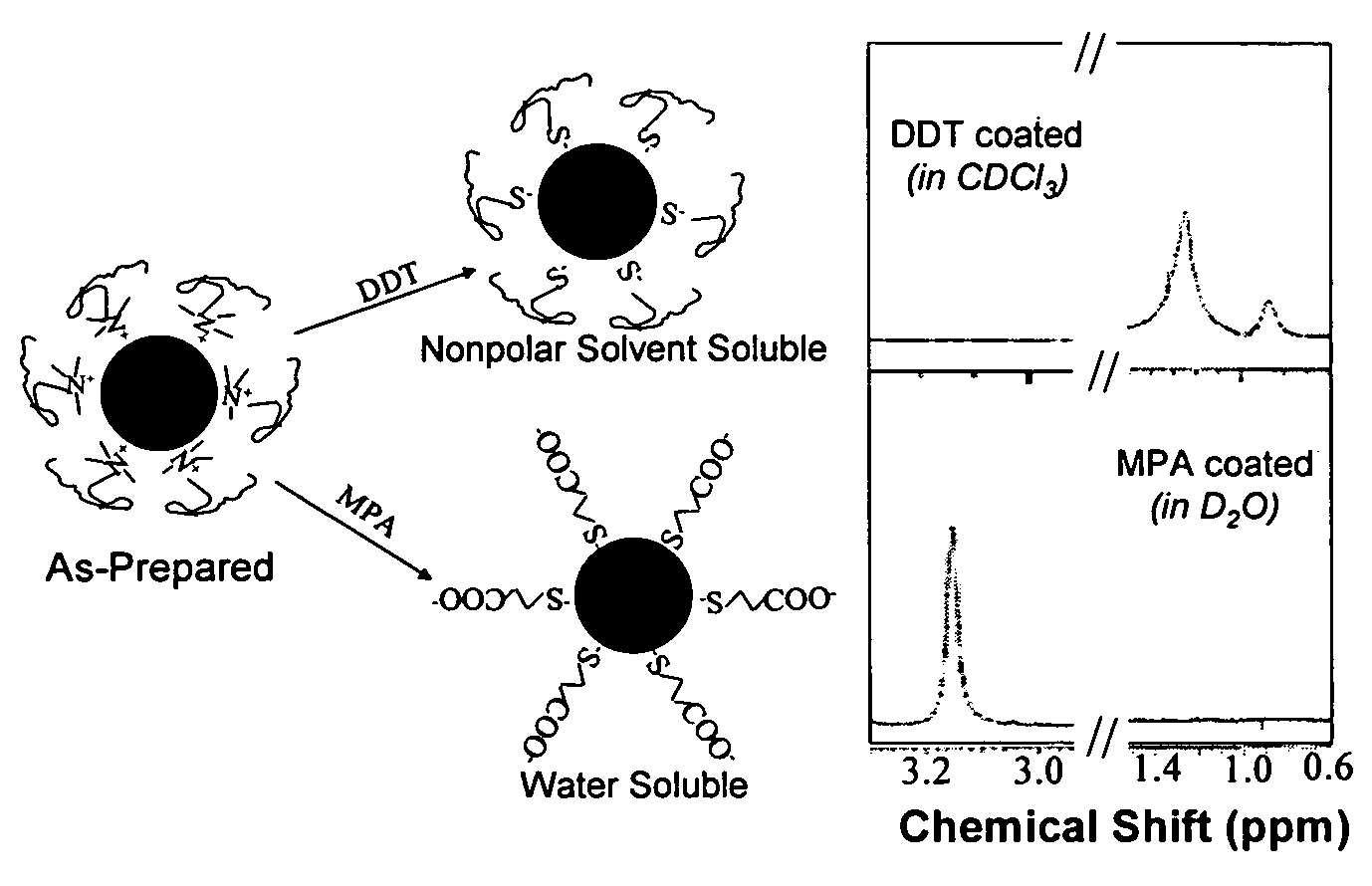
Monodisperse noble metal nanocrystals
Nanoparticle compositions of noble metals, and methods of making them, are described. The nanoparticle compositions are made by reacting a salt or complex of a noble metal, such as Au, Ag, Cu or Pt, with a weak ligand, and a reducing agent, in a single liquid phase. The noble metal is typically provided as a halide or carboxylate. The ligand is preferably a fatty acid or aliphatic amine. The reducing agent is preferably a borohydride reagent, hydrazine, or a mixture thereof. Nanocrystals in the size range of 1 nm to 20 nm are produced, and can be made in substantially monodisperse form.
Owner:THE BOARD OF TRUSTEES OF THE UNIV OF ARKANSAS
Catalysts, activating agents, support media, and related methodologies useful for making catalyst systems especially when the catalyst is deposited onto the support media using physical vapor deposition
InactiveUS7727931B2High catalytic activityTendency increaseMaterial nanotechnologyInternal combustion piston enginesGas phasePhysical chemistry
Use of physical vapor deposition methodologies to deposit nanoscale gold on activating support media makes the use of catalytically active gold dramatically easier and opens the door to significant improvements associated with developing, making, and using gold-based, catalytic systems. The present invention, therefore, relates to novel features, ingredients, and formulations of gold-based, heterogeneous catalyst systems generally comprising nanoscale gold deposited onto a nanoporous support.
Owner:3M INNOVATIVE PROPERTIES CO
Spin-coatable liquid for formation of high purity nanotube films
ActiveUS20050058590A1Lower Level RequirementsPigmenting treatmentMaterial nanotechnologyLiquid mediumSpins
Certain spin-coatable liquids and application techniques are described, which can be used to form nanotube films or fabrics of controlled properties. A spin-coatable liquid for formation of a nanotube film includes a liquid medium containing a controlled concentration of purified nanotubes, wherein the controlled concentration is sufficient to form a nanotube fabric or film of preselected density and uniformity, and wherein the spin-coatable liquid comprises less than 1×1018 atoms / cm3 of metal impurities. The spin-coatable liquid is substantially free of particle impurities having a diameter of greater than about 500 nm.
Owner:ZEON CORP
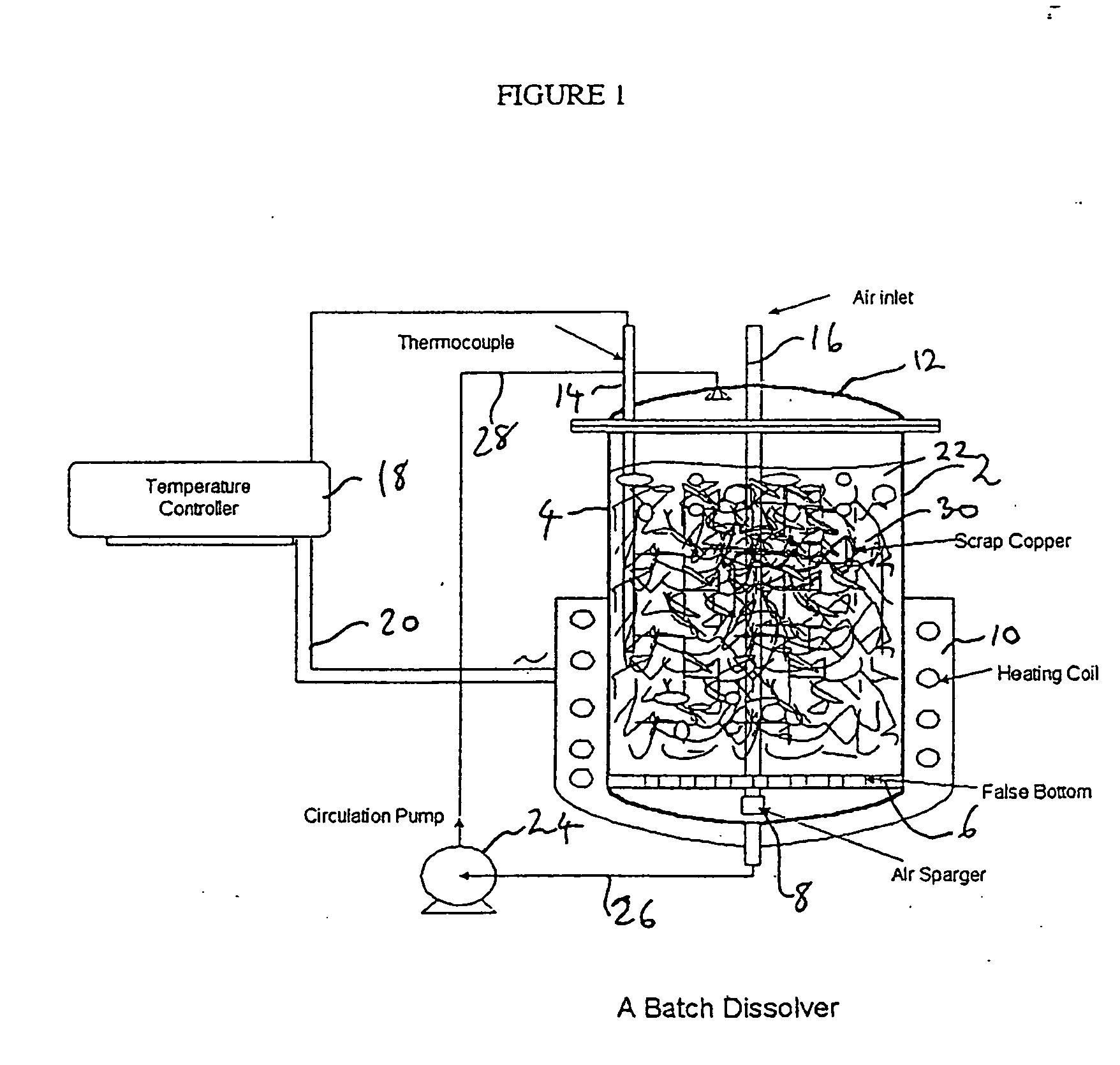
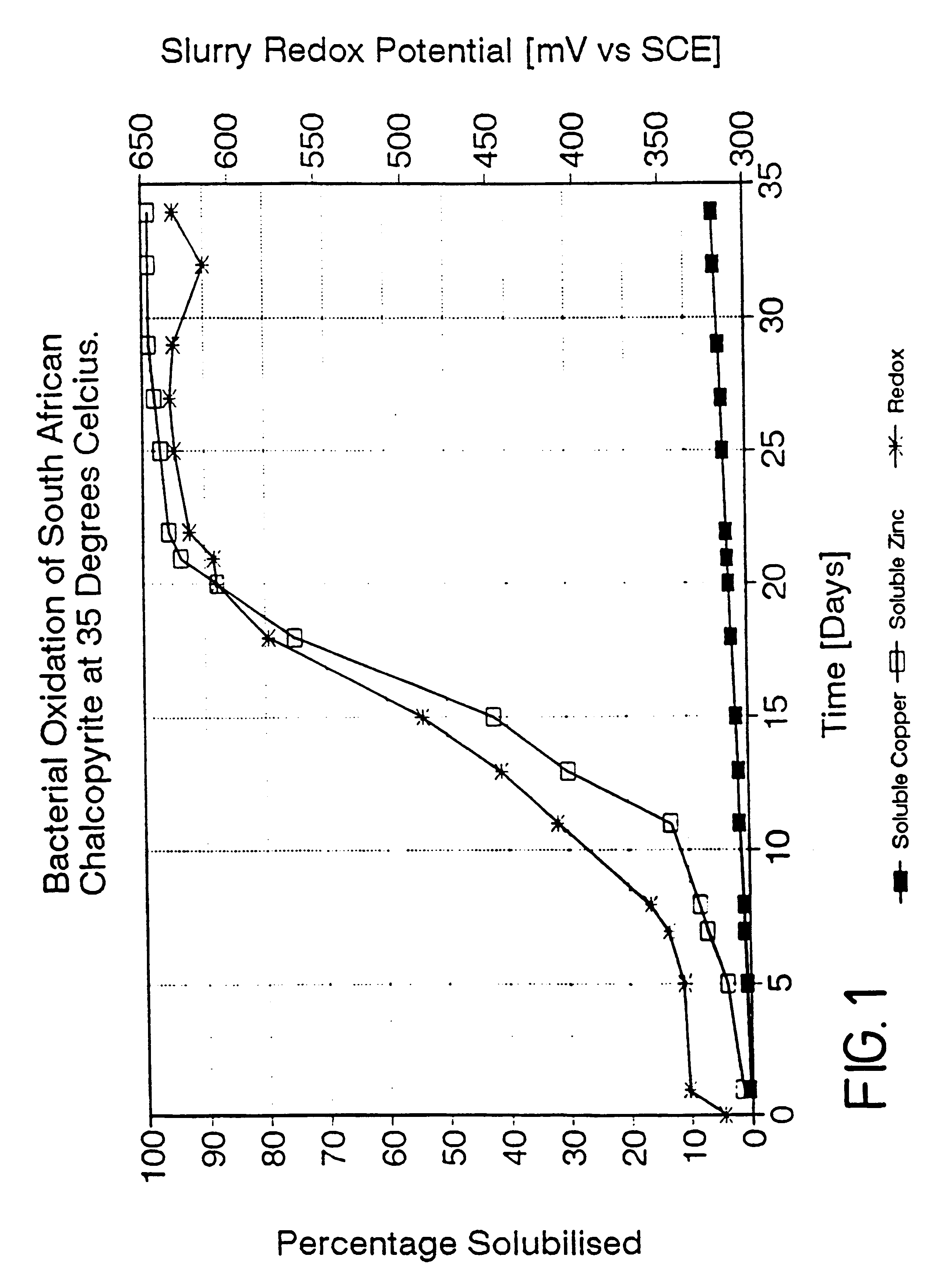
Process for the recovery of value metals from base metal sulfide ores
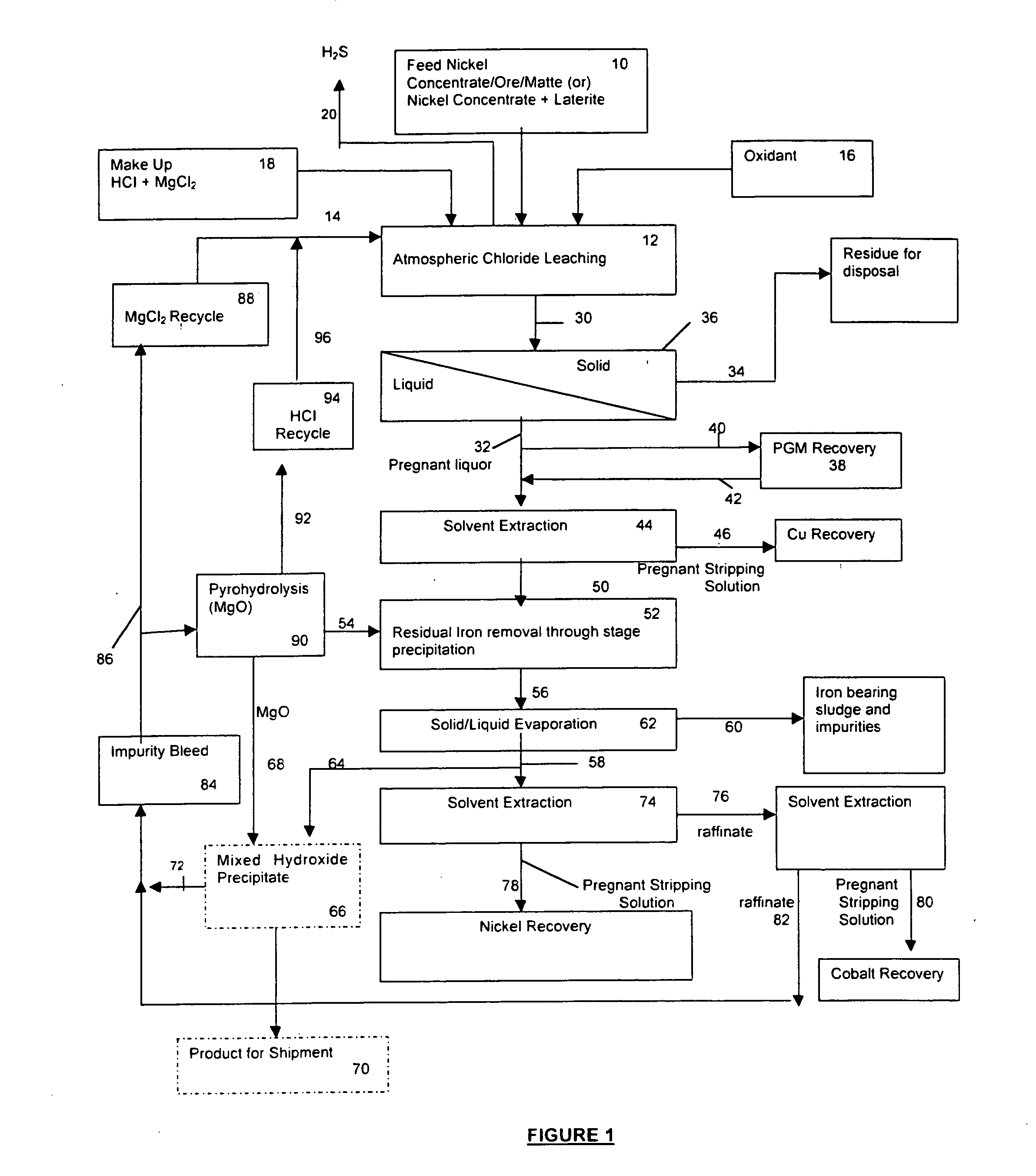
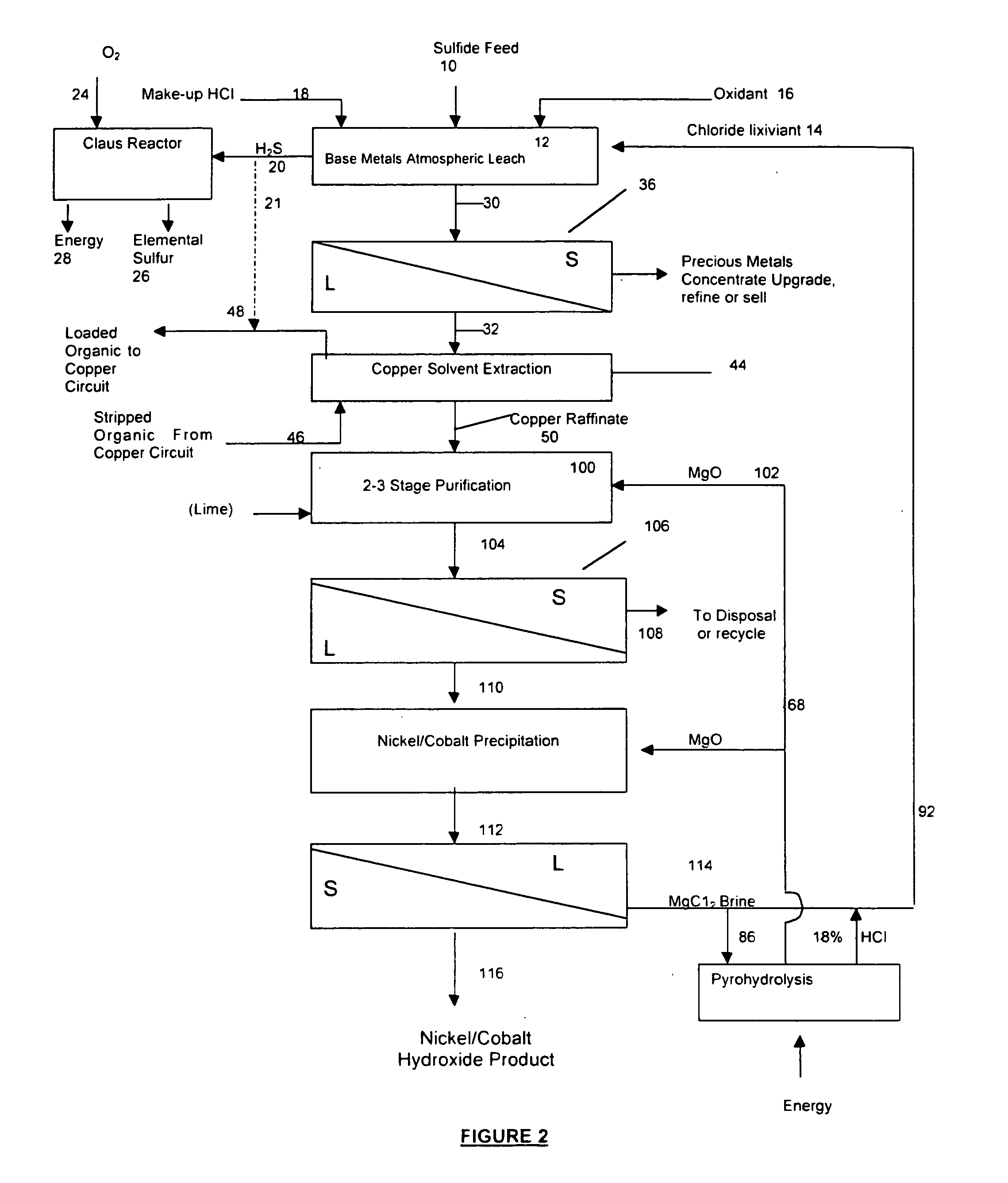
InactiveUS20050118081A1Simple gas/liquidReduce the amount requiredSulfur compoundsSolid sorbent liquid separationSulfideDissolution
A process for leaching a value metal from a base metal sulfide ore, comprising the step of leaching the ore with a lixiviant comprising a chloride, an oxidant and hydrochloric acid is disclosed. The leaching is controlled, by use of low concentrations of hydrochloric acid and a redox potential, to effect formation of hydrogen sulfide from the base metal sulfide ore. The hydrogen sulfide is stripped from the leach solution, thereby reducing the amount of sulfate generated in the leach to very low levels. The leaching may also be conducted to limit the co-dissolution of platinum group metals and gold with the base value metals. The leach forms a value metal-rich leachate and a solids residue. The solids residue may be subsequently leached to recover the platinum group metals and gold. The value metal-rich leachate can be is oxidized and neutralized to recover the value base metals. In an embodiment, the chloride is magnesium chloride and lixiviant solution is regenerated.
Owner:JAGUAR NICKEL INC
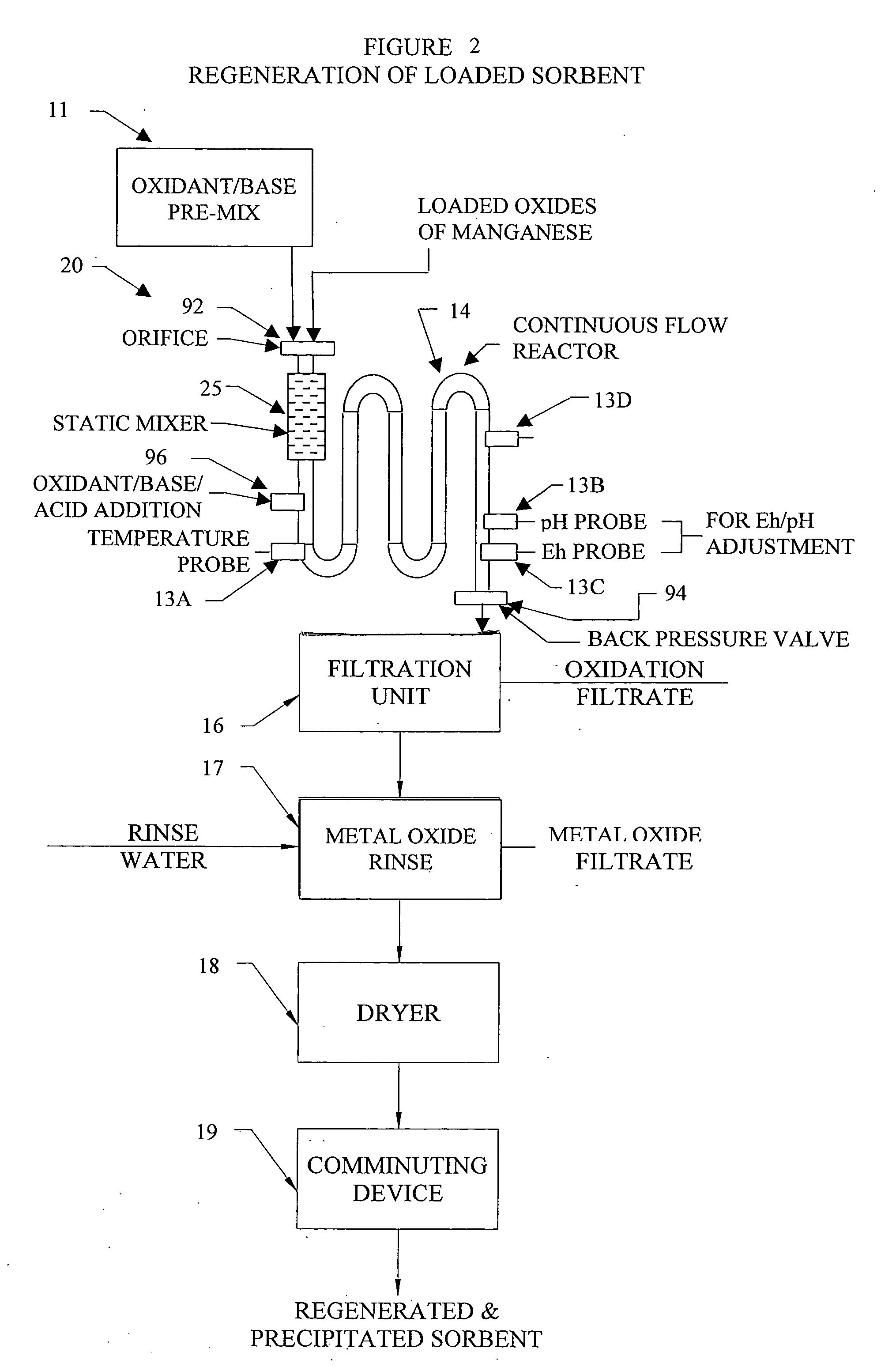
Metal oxide processing methods and systems
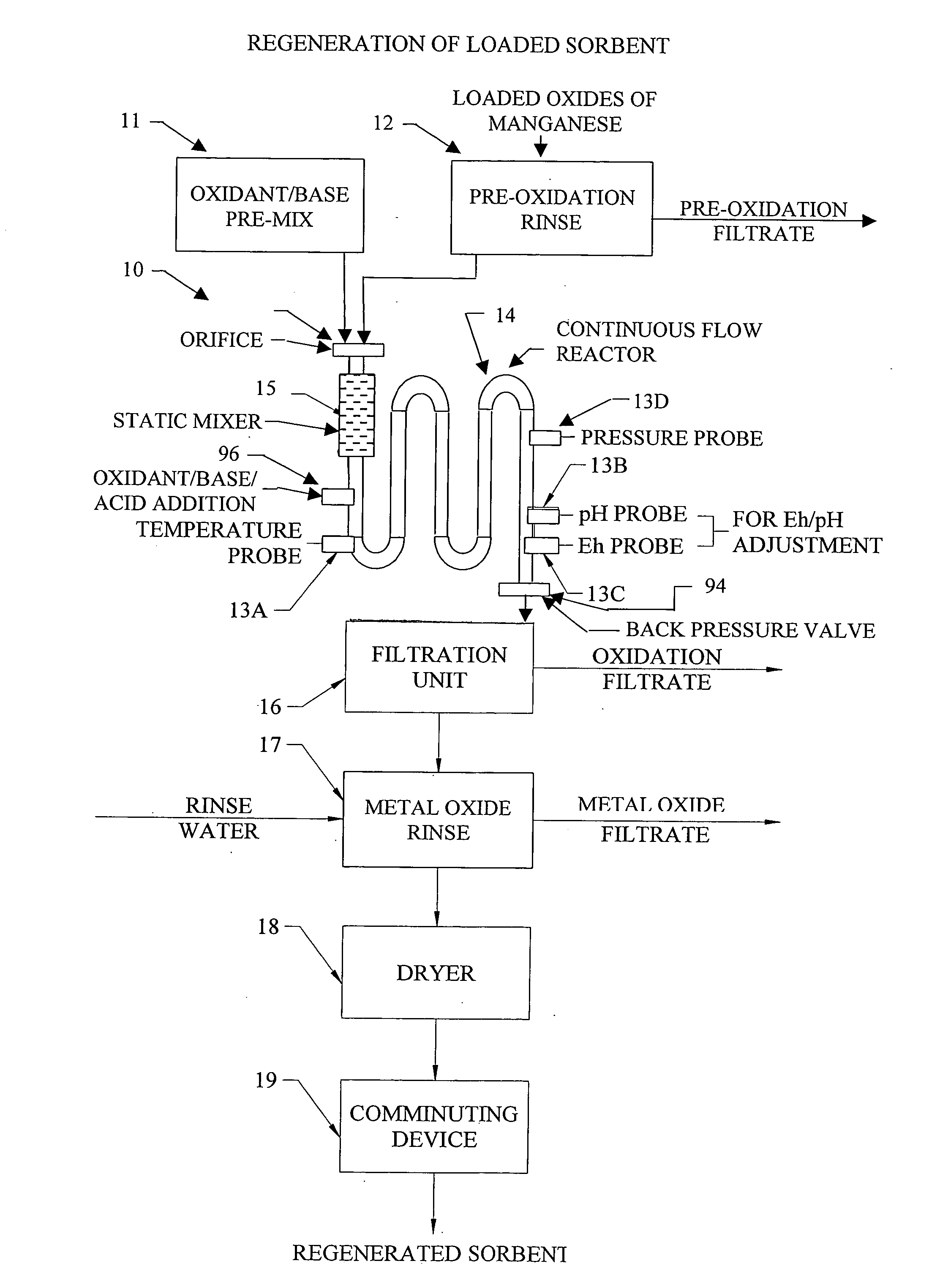
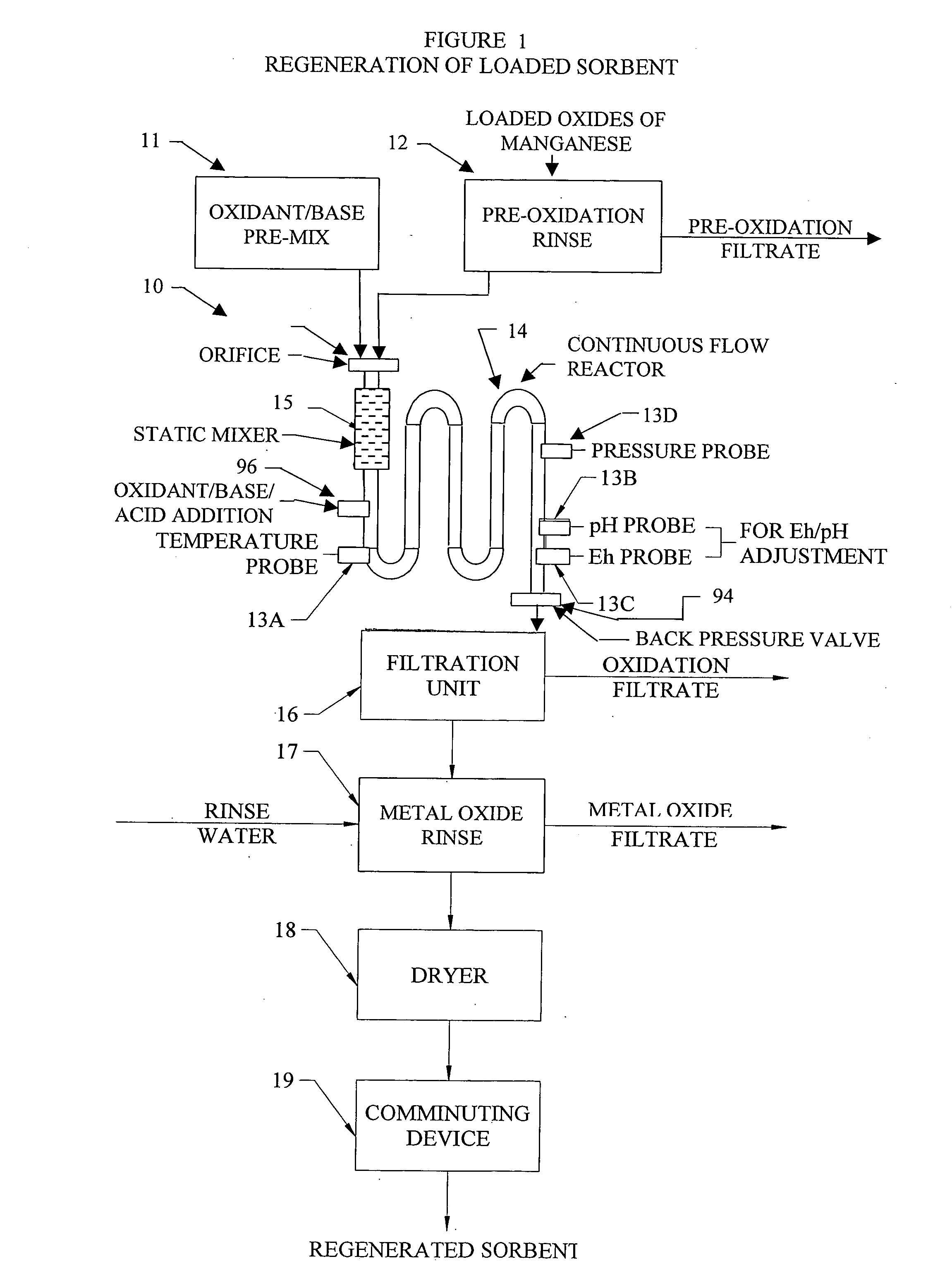
InactiveUS20050074380A1Move quicklyIncrease load capacityCombination devicesTemperatue controlIndustrial gasBatch processing
Methods and systems for processing metal oxides from metal containing solutions. Metal containing solutions are mixed with heated aqueous oxidizing solutions and processed in a continuous process reactor or batch processing system. Combinations of temperature, pressure, molarity, Eh value, and pH value of the mixed solution are monitored and adjusted so as to maintain solution conditions within a desired stability area during processing. This results in metal oxides having high or increased pollutant loading capacities and / or oxidation states. These metal oxides may be processed according to the invention to produce co-precipitated oxides of two or more metals, metal oxides incorporating foreign cations, metal oxides precipitated on active and inactive substrates, or combinations of any or all of these forms. Metal oxides thus produced are, amongst other uses; suitable for use as a sorbent for capturing or removing target pollutants from industrial gas streams or drinking water or aqueous streams or for personal protective respirators.
Owner:ENVIROSCRUB TECH CORP
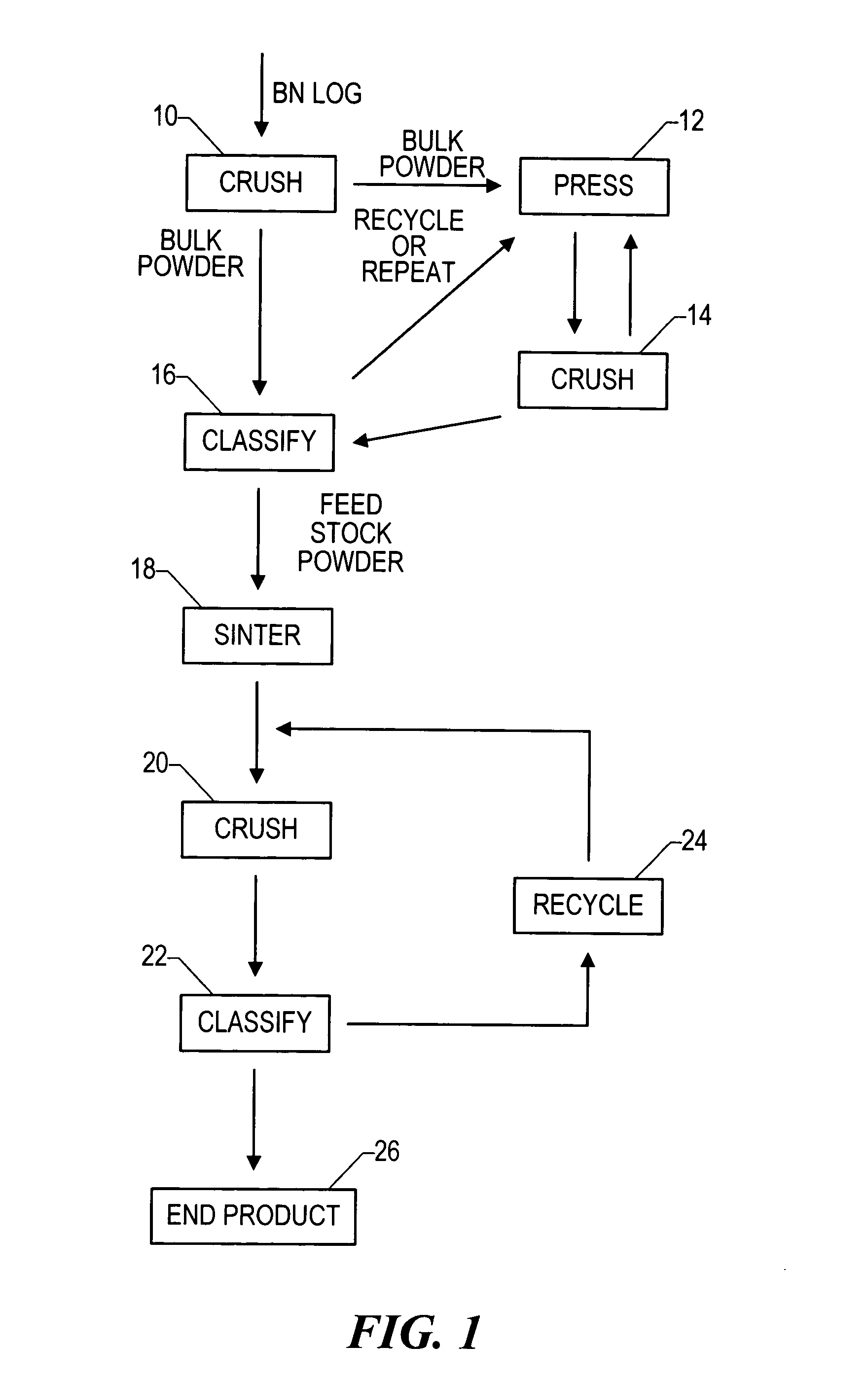
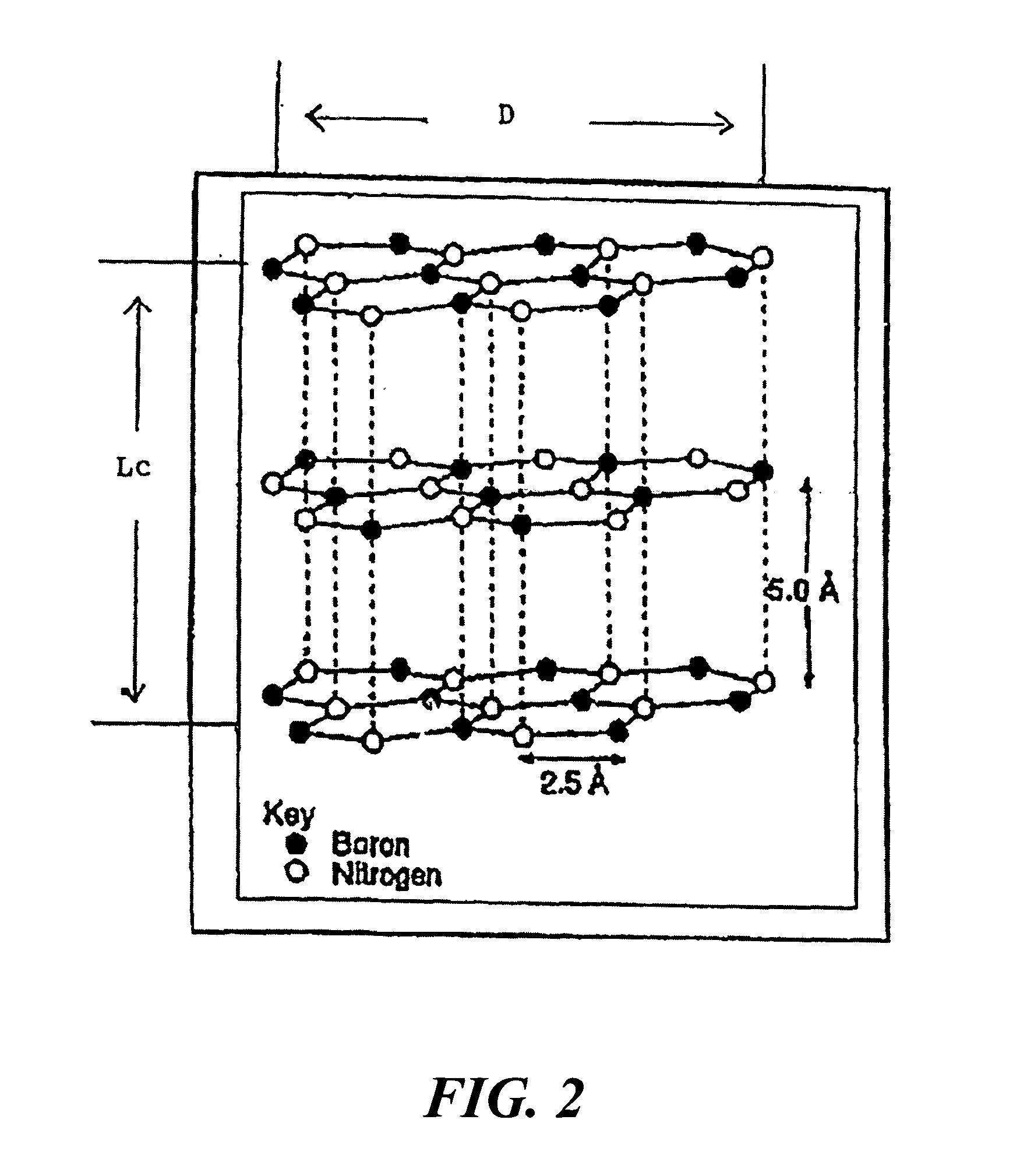
Boron nitride agglomerated powder
Novel boron nitride agglomerated powders are provided having controlled density and fracture strength features. In addition methods for producing same are provided. One method calls for providing a feedstock powder including boron nitride agglomerates, and heat treating the feedstock powder to form a heat treated boron nitride agglomerated powder. In one embodiment the feedstock powder has a controlled crystal size. In another, the feedstock powder is derived from a bulk source.
Owner:SAINT GOBAIN CERAMICS & PLASTICS INC
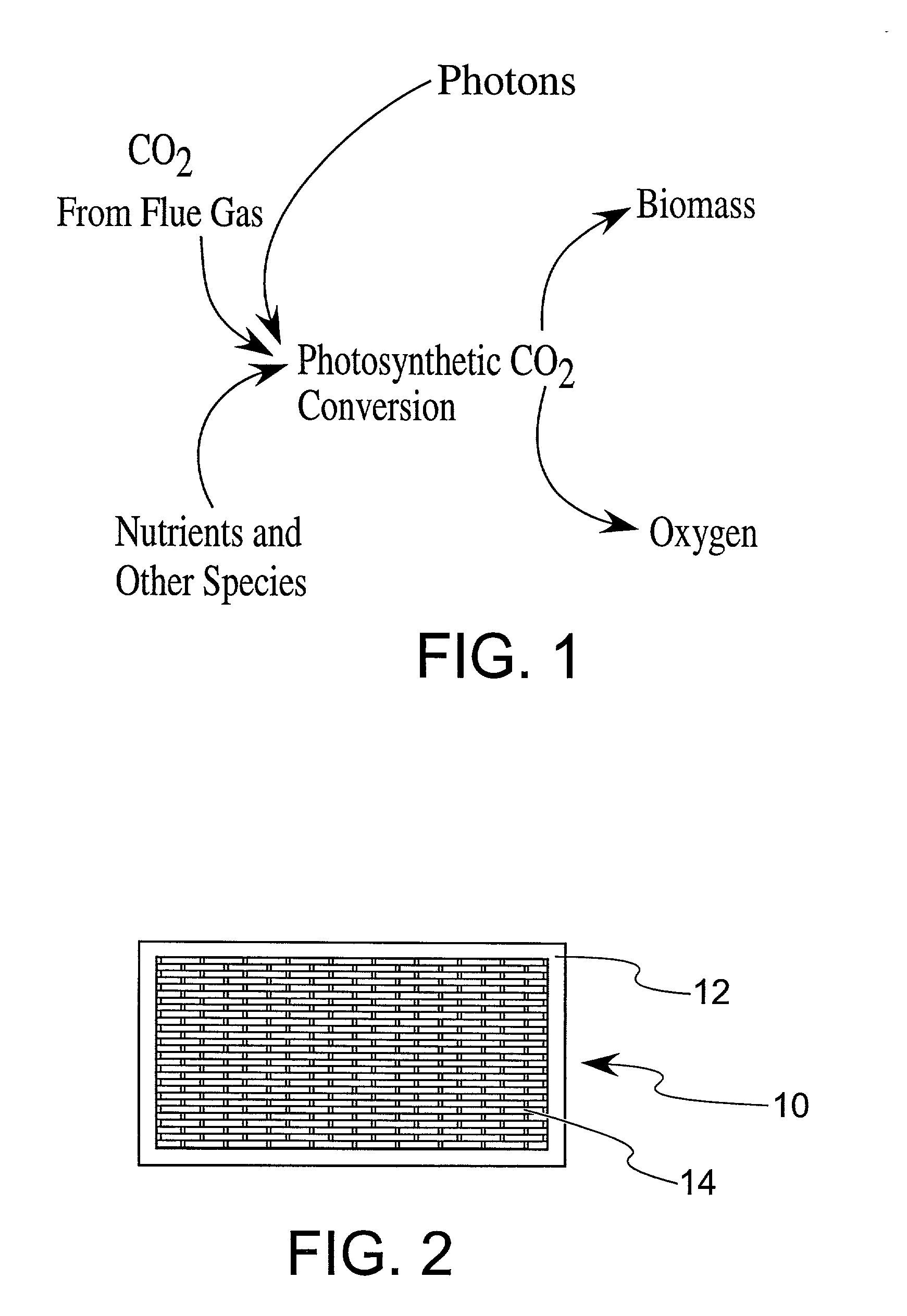
Enhanced practical photosynthetic CO2 mitigation
InactiveUS20020072109A1Increase the amount of waterBioreactor/fermenter combinationsBiological substance pretreatmentsMicroorganismCyanobacteria
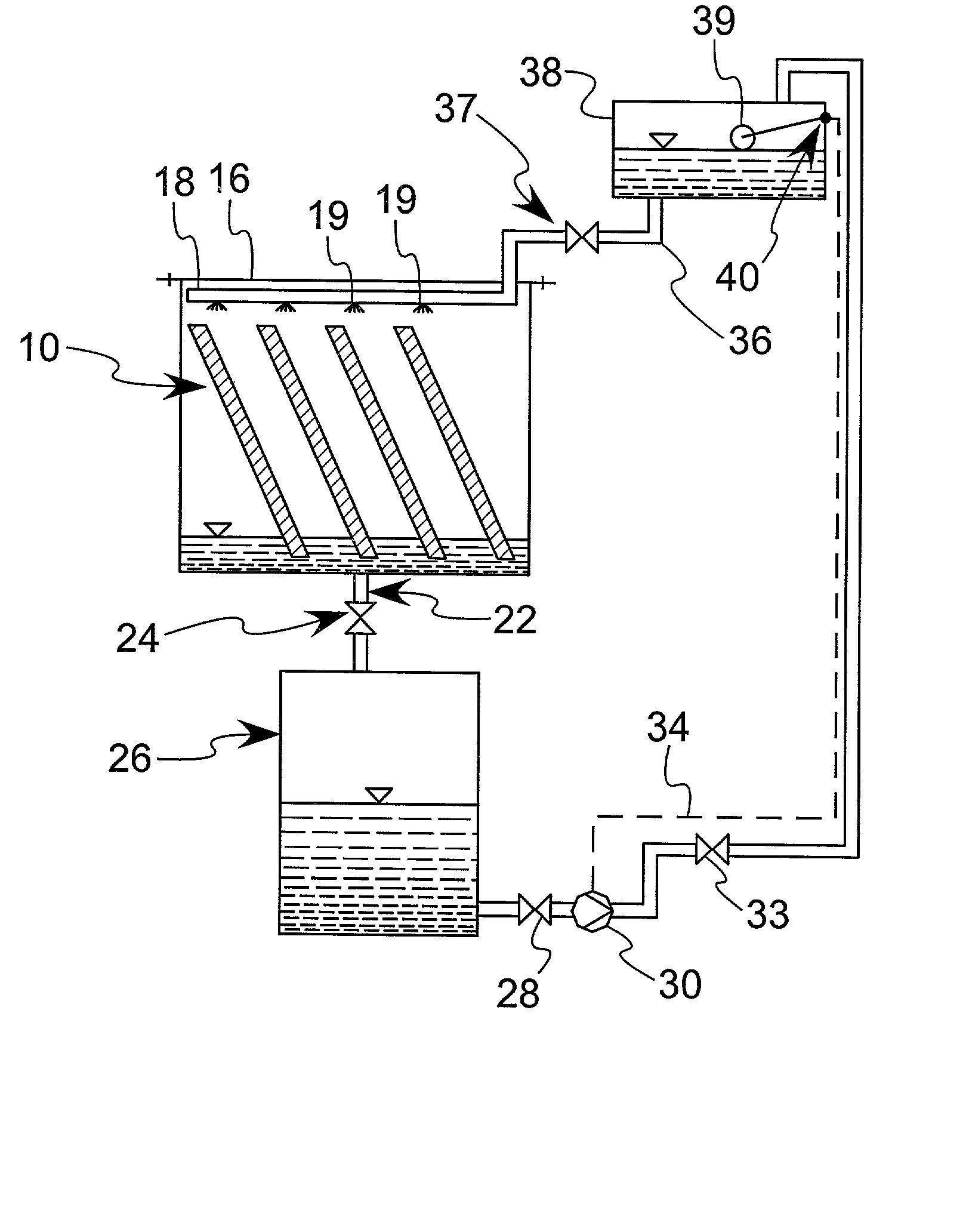
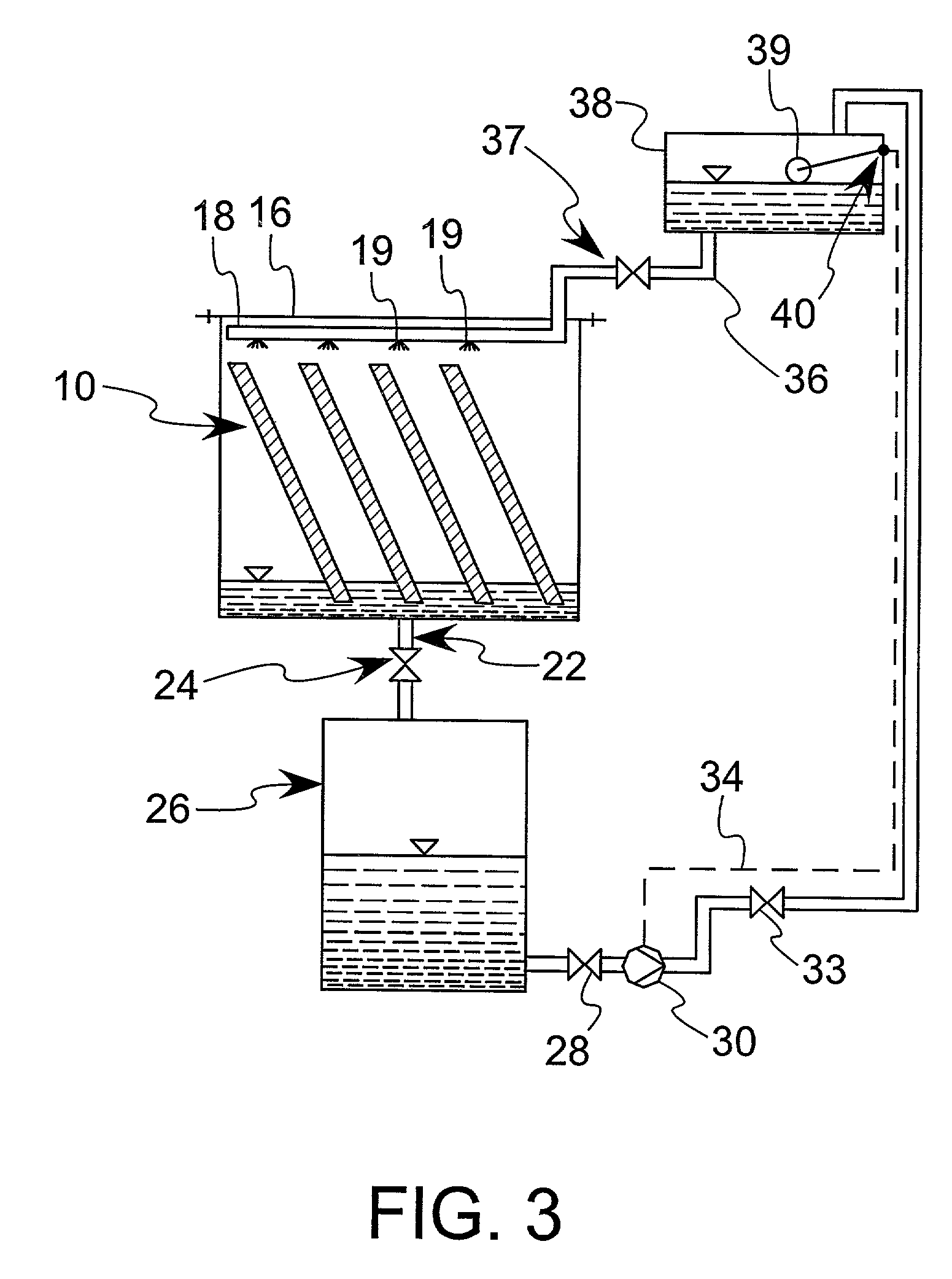
This process is unique in photosynthetic carbon sequestration. An on-site biological sequestration system directly decreases the concentration of carbon-containing compounds in the emissions of fossil generation units. In this process, photosynthetic microbes are attached to a growth surface arranged in a containment chamber that is lit by solar photons. A harvesting system ensures maximum organism growth and rate of CO2 uptake. Soluble carbon and nitrogen concentrations delivered to the cyanobacteria are enhanced, further increasing growth rate and carbon utilization.
Owner:OHIO UNIV
Continuous fiber of single-wall carbon nanotubes
InactiveUS20020127162A1High yieldCarbon compoundsElectrolytic capacitorsFiberElectric power transmission
A method for purifying a mixture comprising single-wall carbon nanotubes and amorphous carbon contaminate is disclosed. The method includes the steps of heating the mixture under oxidizing conditions sufficient to remove the amorphous carbon, followed by recovering a product comprising at least about 80% by weight of single-wall carbon nanotubes. A method for producing tubular carbon molecules of about 5 to 500 nm in length is also disclosed. The method includes the steps of cutting single-wall nanotube containing-material to form a mixture of tubular carbon molecules having lengths in the range of 5-500 nm and isolating a fraction of the molecules having substantially equal lengths The nanotubes may be used, singularly or in multiples, in power transmission cables, in solar cells, in batteries, as antennas, as molecular electronics, as probes and manipulators, and in composites.
Owner:RICE UNIV
Conductive paste
InactiveUS20060145125A1Pigmenting treatmentGroup 1/11 element organic compoundsConductive pasteAdhesive
The present invention provides an electroconductive paste that can contain a high proportion of an electroconductive powder, has excellent electroconductivity reliability and migration resistance, has a highly competitive price due to a reduced amount of silver plating, and is suitable for use in solder electrode formation, an electroconductive adhesive, etc. The electroconductive paste of the present invention comprises a binder and an electroconductive powder containing 80 to 97 wt % of a substantially spherical silver-coated copper powder in which the surface of a copper powder is coated with silver and the surface thereof is further coated with 0.02 to 0.5 wt % relative to the copper powder of a fatty acid, and 3 to 20 wt % of a flat-shaped silver-coated copper powder in which the surface of a copper powder is coated with silver and the surface thereof is further coated with 0.02 to 1.2 wt % relative to the copper powder of a fatty acid.
Owner:HITACHI CHEM CO LTD
Precipitated aragonite and a process for producing it
InactiveUS20030213937A1Less expensiveEfficient and less-expensiveCalcium/strontium/barium carbonatesInorganic/elemental detergent compounding agentsParticulatesAragonite
Disclosed is a novel form of particulate precipitated aragonite, and a novel process for producing it.
Owner:3P TECH
Process of making hydrophobic metal oxide nanoparticles
InactiveUS7081234B1Uniform coatingSulfur compoundsCopper oxides/halidesMetal oxide nanoparticlesTransport layer
A process of treating metal oxide nanoparticles that includes mixing metal oxide nanoparticles, a solvent, and a surface treatment agent that is preferably a silane or siloxane is described. The treated metal oxide nanoparticles are rendered hydrophobic by the surface treatment agent being surface attached thereto, and are preferably dispersed in a hydrophobic aromatic polymer binder of a charge transport layer of a photoreceptor, whereby π—π interactions can be formed between the organic moieties on the surface of the nanoparticles and the aromatic components of the binder polymer to achieve a stable dispersion of the nanoparticles in the polymer that is substantially free of large sized agglomerations.
Owner:XEROX CORP
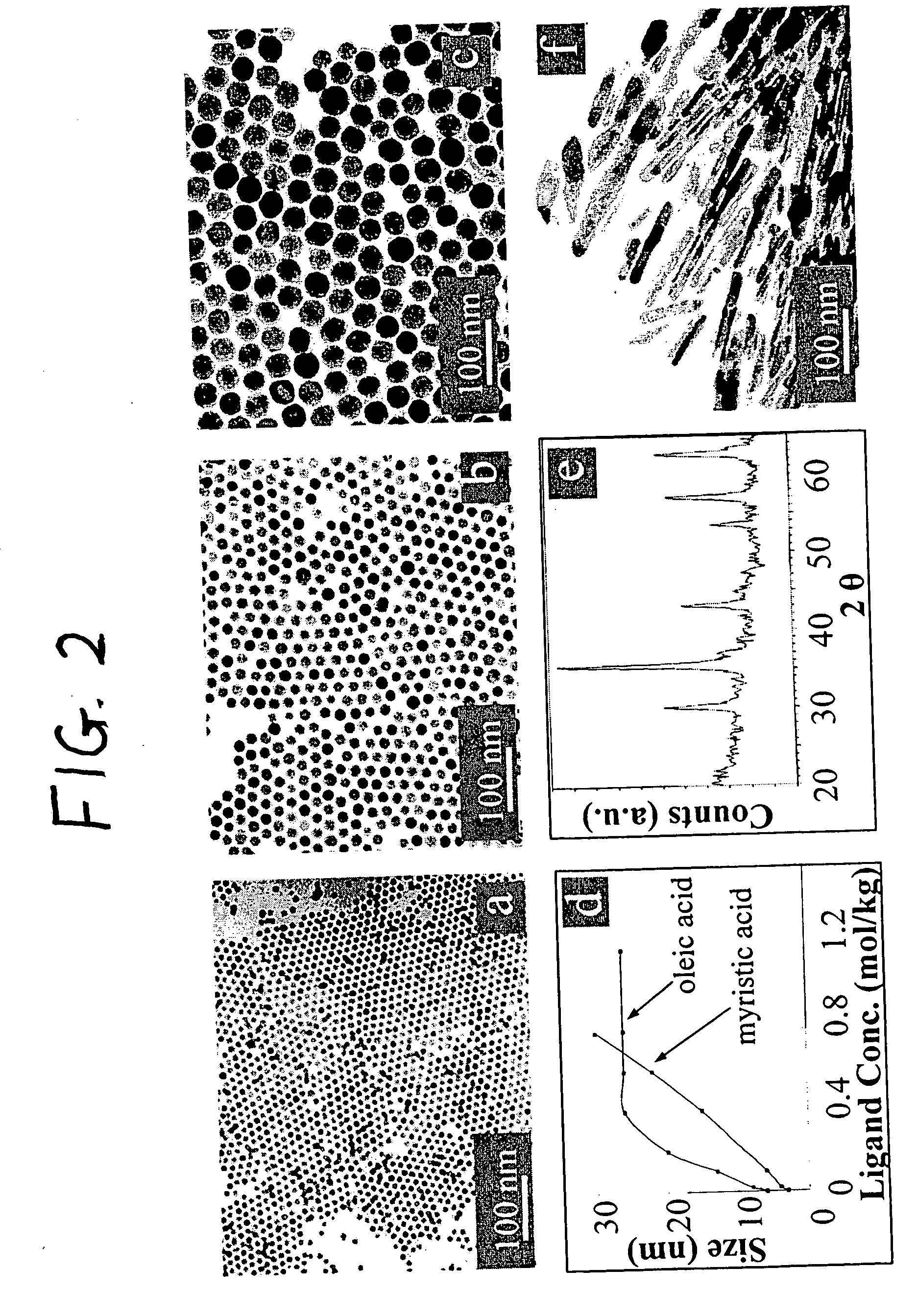
Synthetic control of metal oxide nanocrystal sizes and shapes
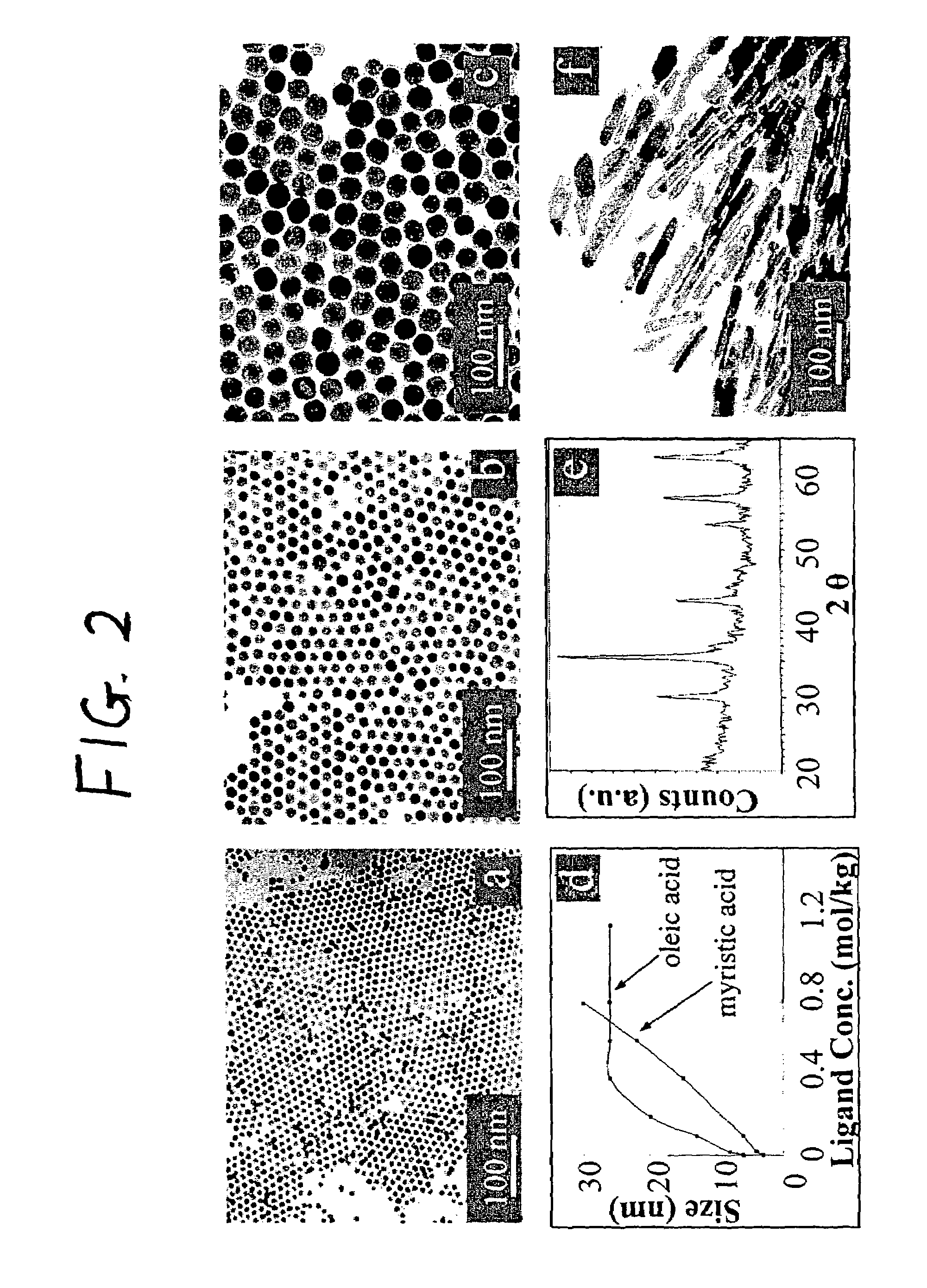
A general, reproducible, and simple synthetic method that employs readily available chemicals permits control of the size, shape, and size distribution of metal oxide nanocrystals. The synthesis entails reacting a metal fatty acid salt, the corresponding fatty acid, and a hydrocarbon solvent, with the reaction product being pyrolyzed to the metal oxide. Nearly monodisperse oxide nanocrystals of Fe3O4, Cr2O3, MnO, Co3O4, NiO, ZnO, SnO2, and In2O3, in a large size range (3-50 nm), are described. Size and shape control of the nanocrystals is achieved by varying the reactivity and concentration of the precursors.
Owner:THE BOARD OF TRUSTEES OF THE UNIV OF ARKANSAS
Methods for removing heavy metals from water using chemical precipitation and field separation methods
InactiveUS6896815B2Small sizeChemical cost reductionSolid sorbent liquid separationGold compoundsWater useSludge
A two-step chemical precipitation process involving hydroxide precipitation and sulfide precipitation combined with “field separation” technology such as magnetic separation, dissolved air flotation, vortex separation or expanded plastics flotation, effectively removes chelated and non-chelated heavy metal precipitates and other fine particles from water. In the first-step, the non-chelated heavy metals are precipitated as hydroxides and removed from the water by a conventional liquid / solids separator such as an inclined plate clarifier to remove a large percentage of the dissolved heavy metals. The cleaned water is then treated in a second precipitation step to remove the residual heavy metals to meet discharge limits. In the second precipitation step, any metal precipitant more effective than hydroxide for metal precipitation can be used. The invention improves metal removal, lowers cost because fewer chemicals are used, produces less sludge, and reduces the discharge of toxic metals and metal precipitants to the environment.
Owner:CORT STEVEN L
Methods of Making Binary Metal Oxide Nanostructures and Methods of Controlling Morphology of Same
ActiveUS20100278720A1Reduce crystallinityControl dimensionalityCopper oxides/halidesManganese oxides/hydroxidesPorous membraneNanostructure
The present invention includes a method of producing a crystalline metal oxide nanostructure. The method comprises providing a metal salt solution and providing a basic solution; placing a porous membrane between the metal salt solution and the basic solution, wherein metal cations of the metal salt solution and hydroxide ions of the basic solution react, thereby producing a crystalline metal oxide nanostructure.
Owner:WONG STANISLAUS S +1
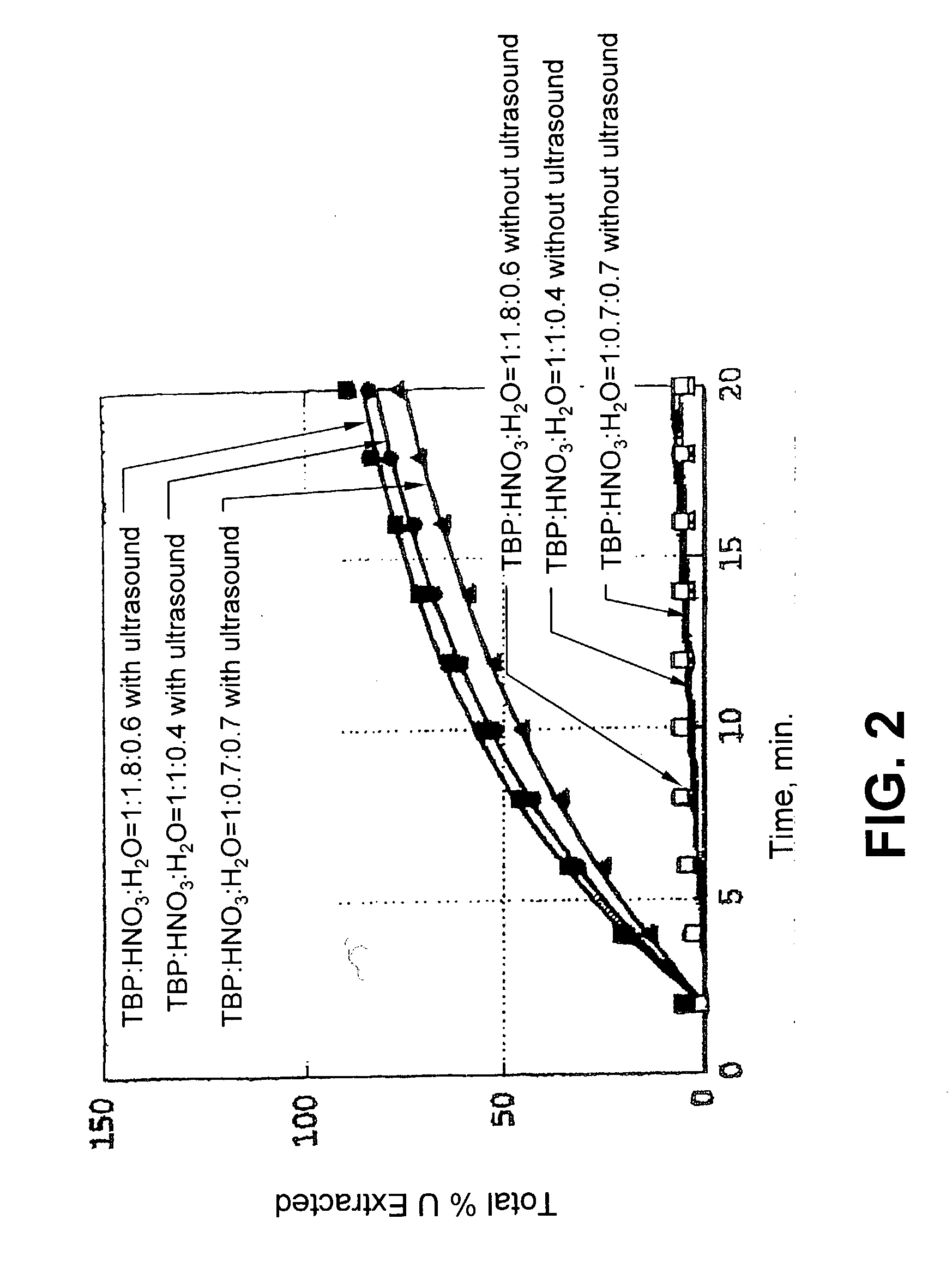
Ultrasound enhanced process for extracting metal species in supercritical fluids
InactiveUS20030183043A1Enhances rate and efficiencyReduce probabilitySolid sorbent liquid separationGold compoundsUranium oxidePresent method
Improved methods for the extraction or dissolution of metals, metalloids or their oxides, especially lanthanides, actinides, uranium or their oxides, into supercritical solvents containing an extractant are disclosed. The disclosed embodiments specifically include enhancing the extraction or dissolution efficiency with ultrasound. The present methods allow the direct, efficient dissolution of UO2 or other uranium oxides without generating any waste stream or by-products.
Owner:NAGOYA INDUSTRIAL SCIENCE RESEARCH INST +1
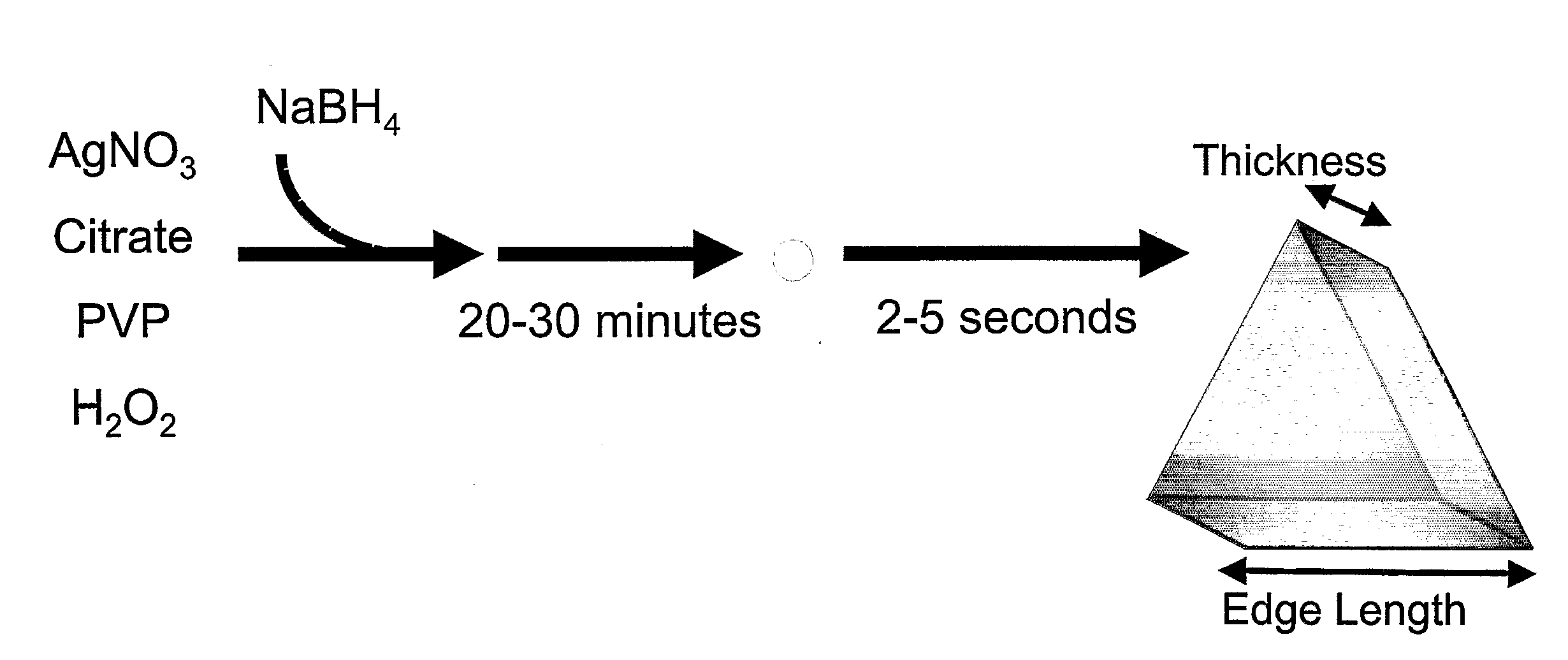
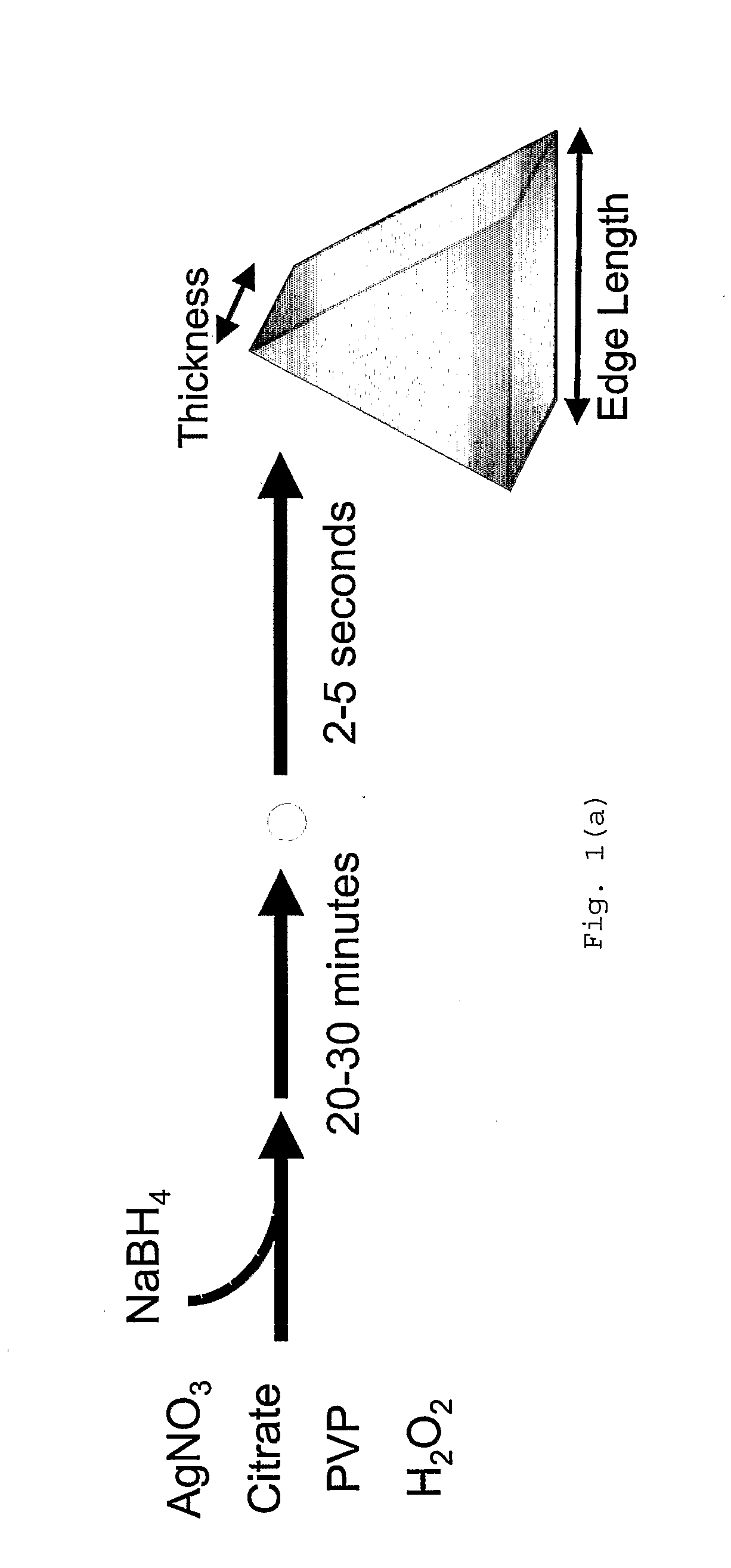
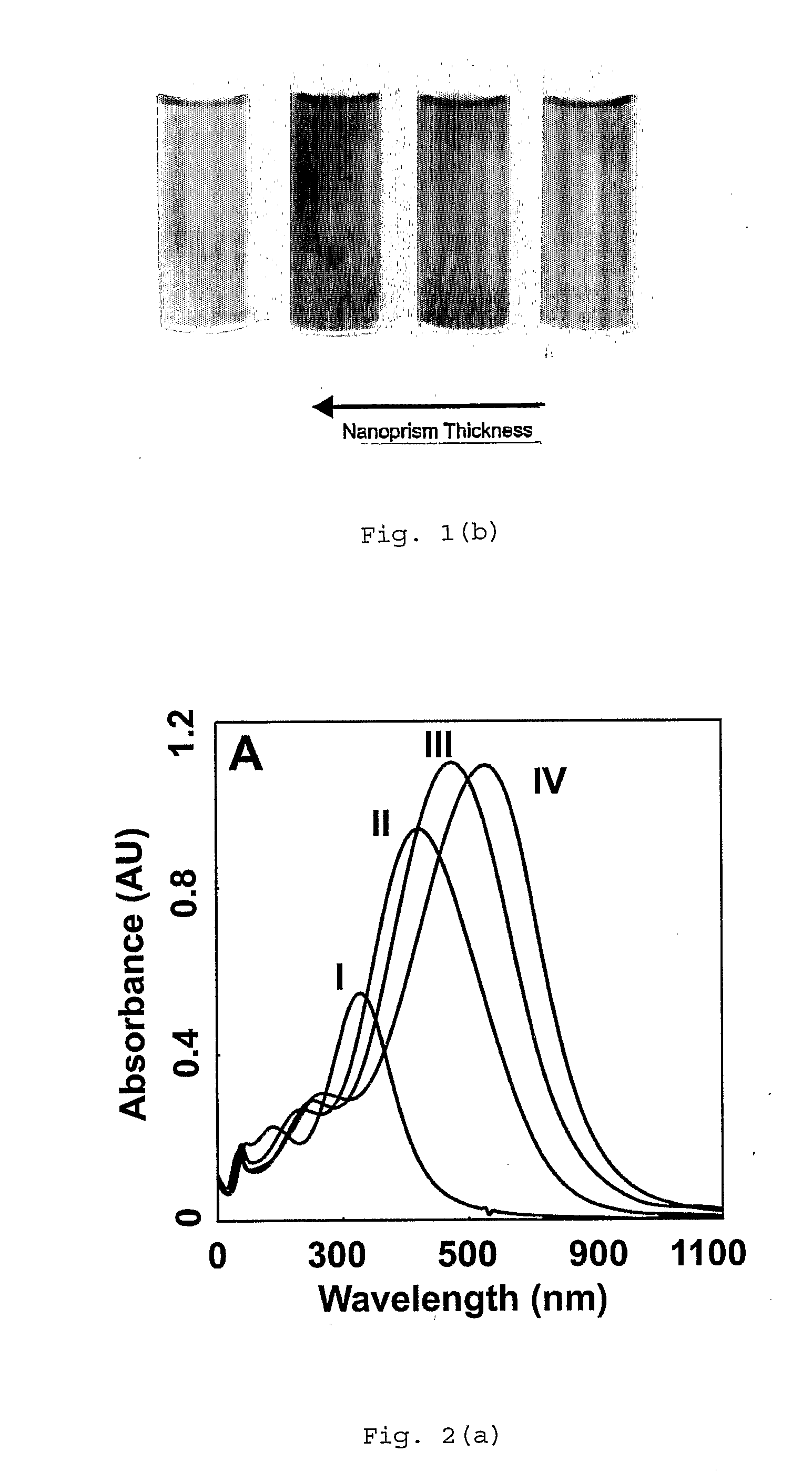
Method of Making Metal Nanoprisms Having a Predetermined Thickness
InactiveUS20080295646A1Control over edge lengthControl lengthMaterial nanotechnologyNanostructure manufacturePresent methodEngineering
A method of preparing metal nanoprisms having a unimodal size distribution and a predetermined thickness. The present method also allows control over nanoprism edge length.
Owner:NORTHWESTERN UNIV
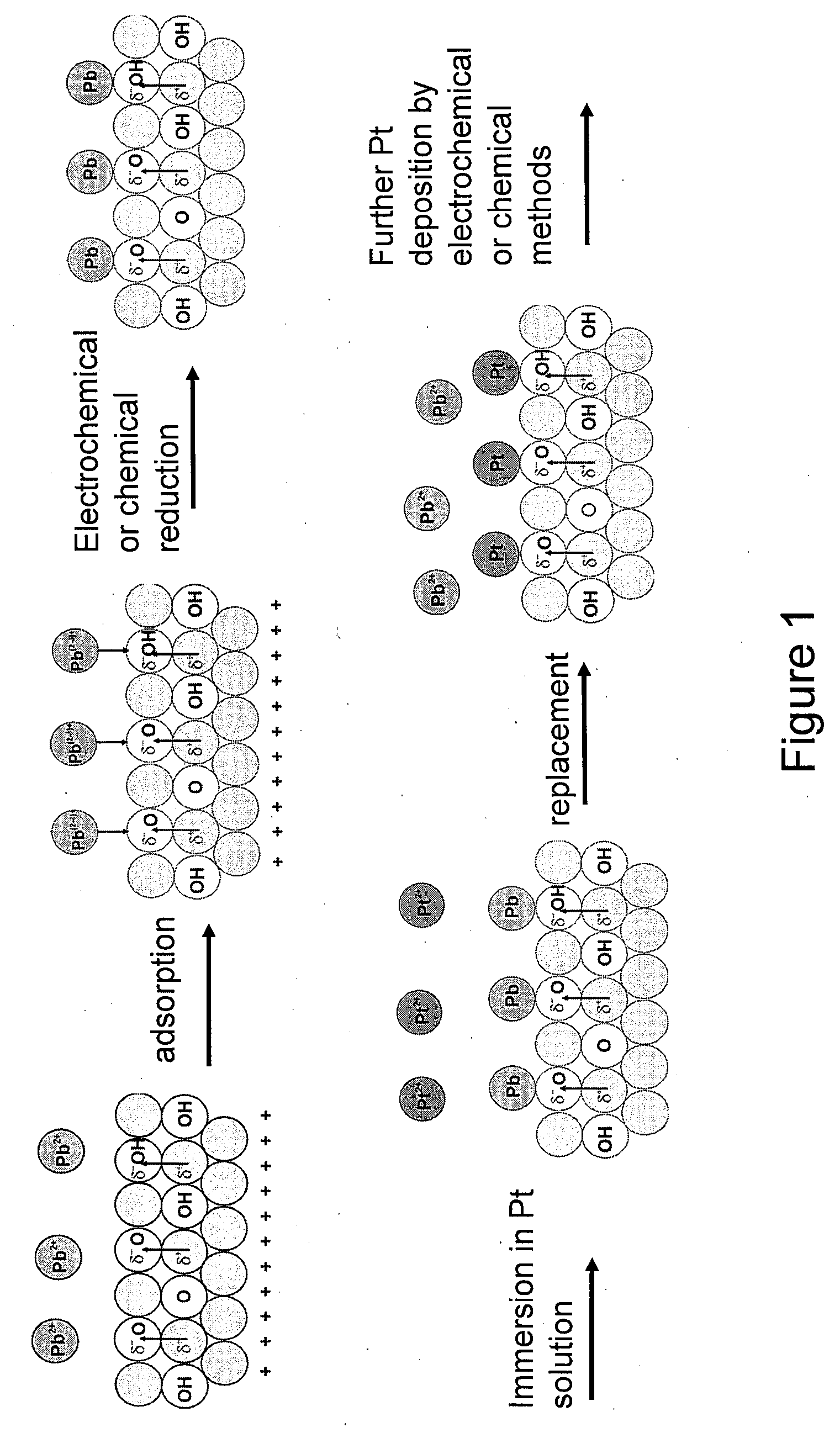
Synthesis of Metal-Metal Oxide Catalysts and Electrocatalysts Using a Metal Cation Adsorption/Reduction and Adatom Replacement by More Noble Ones
InactiveUS20070264189A1Improve stabilityReducing and preventing oxidationCell electrodesGold compoundsHydrogenFuel cells
The invention relates to platinum-metal oxide composite particles and their use as electrocatalysts in oxygen-reducing cathodes and fuel cells. The invention particularly relates to methods for preventing the oxidation of the platinum electrocatalyst in the cathodes of fuel cells by use of these platinum-metal oxide composite particles. The invention additionally relates to methods for producing electrical energy by supplying such a fuel cell with an oxidant, such as oxygen, and a fuel source, such as hydrogen. The invention also relates to methods of making the metal-metal oxide composites.
Owner:BROOKHAVEN SCI ASSOCS
Antimicrobial lubricant
InactiveUS20110301553A1Easy to insertFacilitate contact interactionBiocideSurgeryMedical deviceMedical treatment
An antimicrobial lubricant combines an antimicrobial agent, preferably silver based, with a silicone lubricant. This is achieved by first formulating a lubricant solution that has a non-volatile silicone lubricant and a volatile solvent carrier. An antimicrobial agent is added to the lubricant solution. The solution is then subjected to ultrasonic energy so that the antimicrobial agent is uniformly mixed with the lubricant in the solution. The solution containing the antimicrobial lubricant may be sprayed onto a medical device, for example the outer surface of the catheter of a peripheral IV catheter. After the solvent flashes off, a coat of uniform antimicrobial lubricant is formed on the catheter surface. The antimicrobial lubricant coating facilitates the insertion of the catheter to a patient, and at the same time prevents the growth of microorganisms from the device and infection to the patient.
Owner:SMITHS MEDICAL ASD INC
Apparatus and method for preparing cerium oxide nanoparticles
InactiveUS20050031517A1Quick responseReaction time is thus limitedMaterial nanotechnologyMixing methodsCerium nitrateNanoparticle
This invention provides a method for preparing cerium oxide nanoparticles with a narrow size distribution. The cerium oxide nanoparticles obtained by the method of the invention are nearly all crystalline. The method comprises providing a first aqueous solution comprising cerium nitrate and providing a second aqueous solution comprising hexamethylenetetramine. The first and second aqueous solutions are mixed to form a mixture, and the mixture is maintained at a temperature no higher than about 320° K to form nanoparticles. The nanoparticles that are formed are then separated from the mixture. A further aspect of the present invention is an apparatus for preparing cerium oxide nanoparticles. The apparatus comprises a mixing vessel having a first compartment for holding a first aqueous solution comprising cerium nitrate and a second compartment for holding a second aqueous solution comprising hexamethylenetetramine. The mixing vessel has a retractable partition separating the first and second compartments. When the retractable partition is retracted, rapid mixing of the first aqueous solution with the second aqueous solution takes place to form a mixture, and the mixture is maintained at a temperature no higher than about 320° K to form nanoparticles therein.
Owner:THE TRUSTEES OF COLUMBIA UNIV IN THE CITY OF NEW YORK
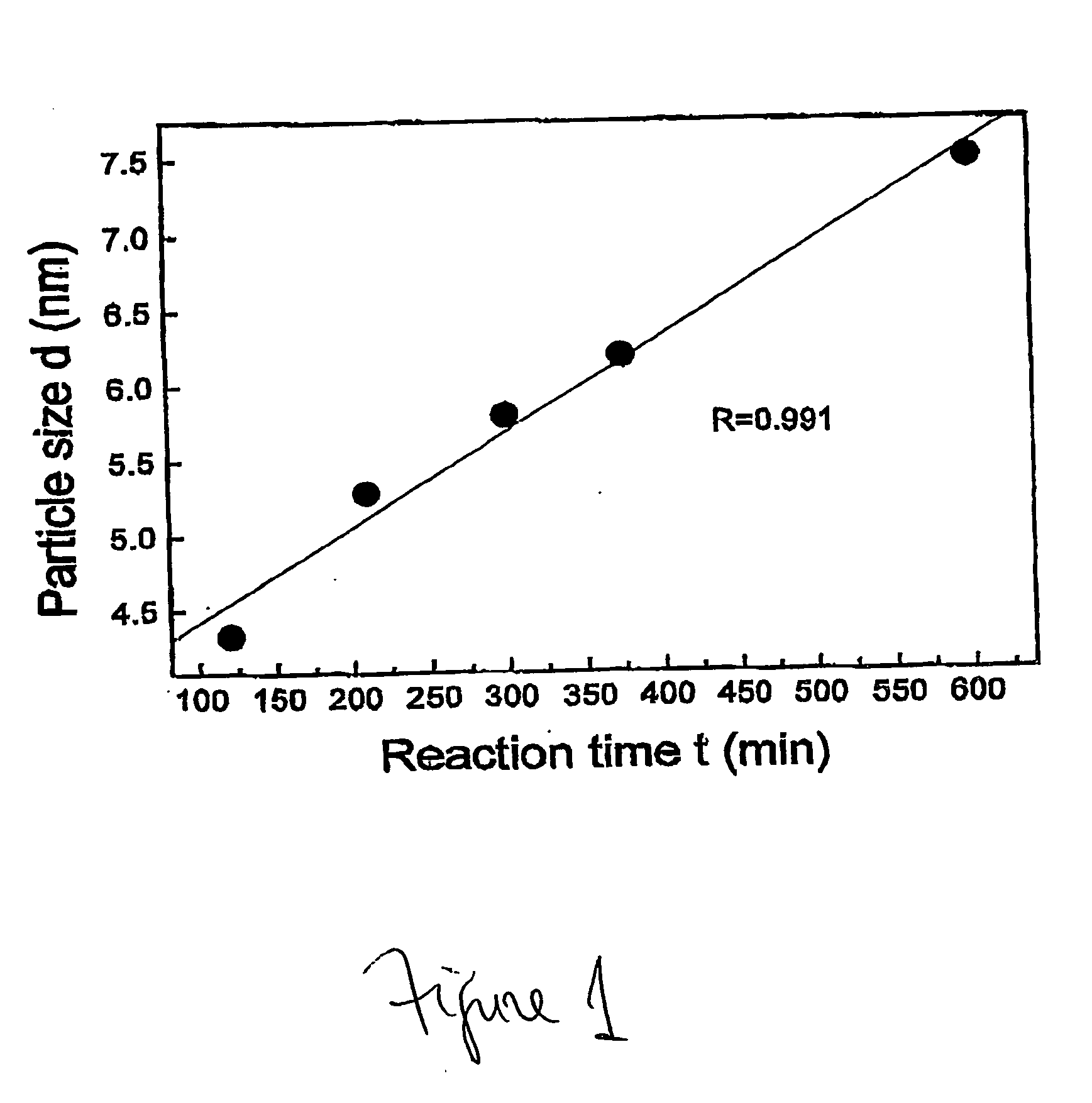
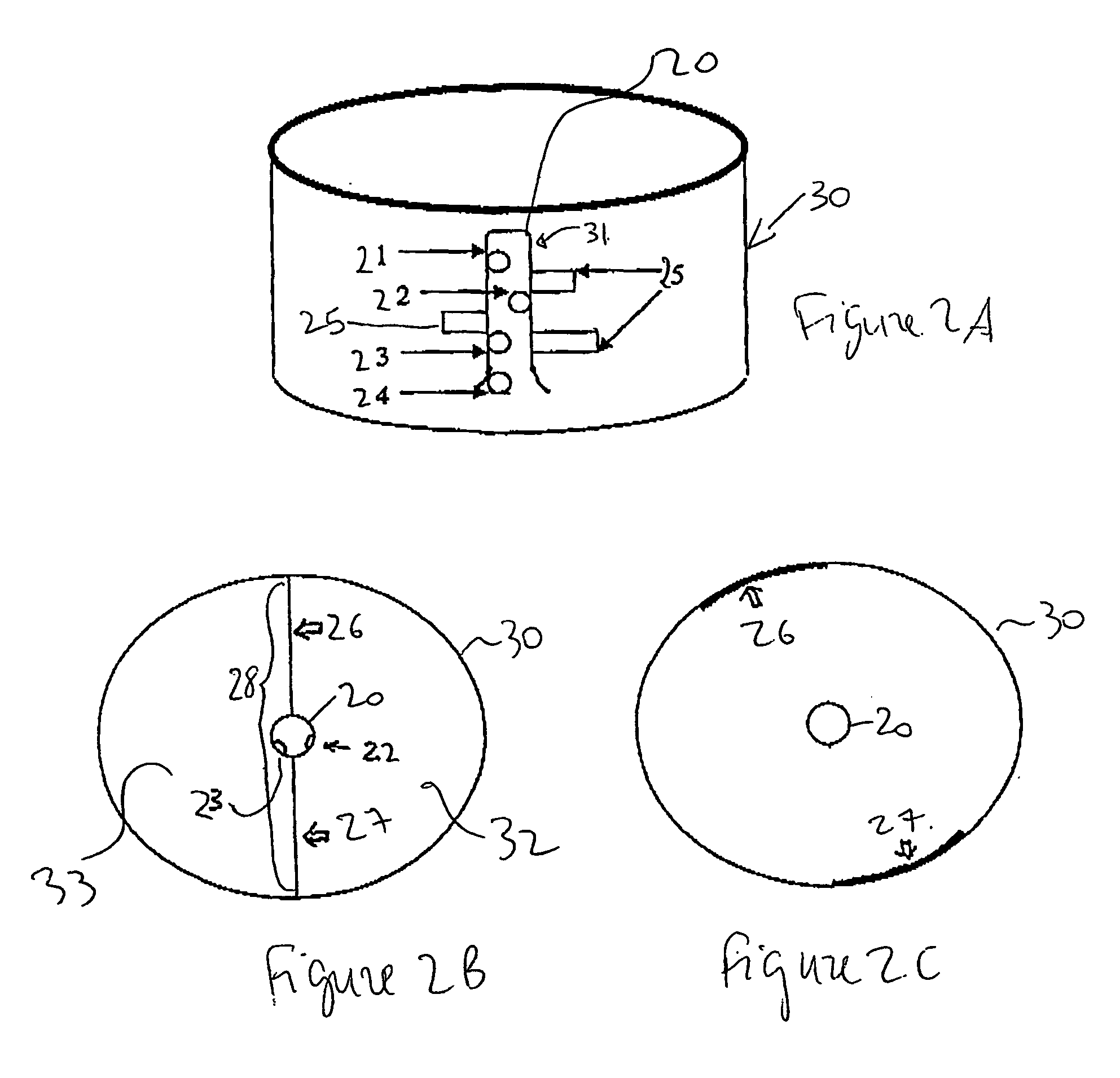
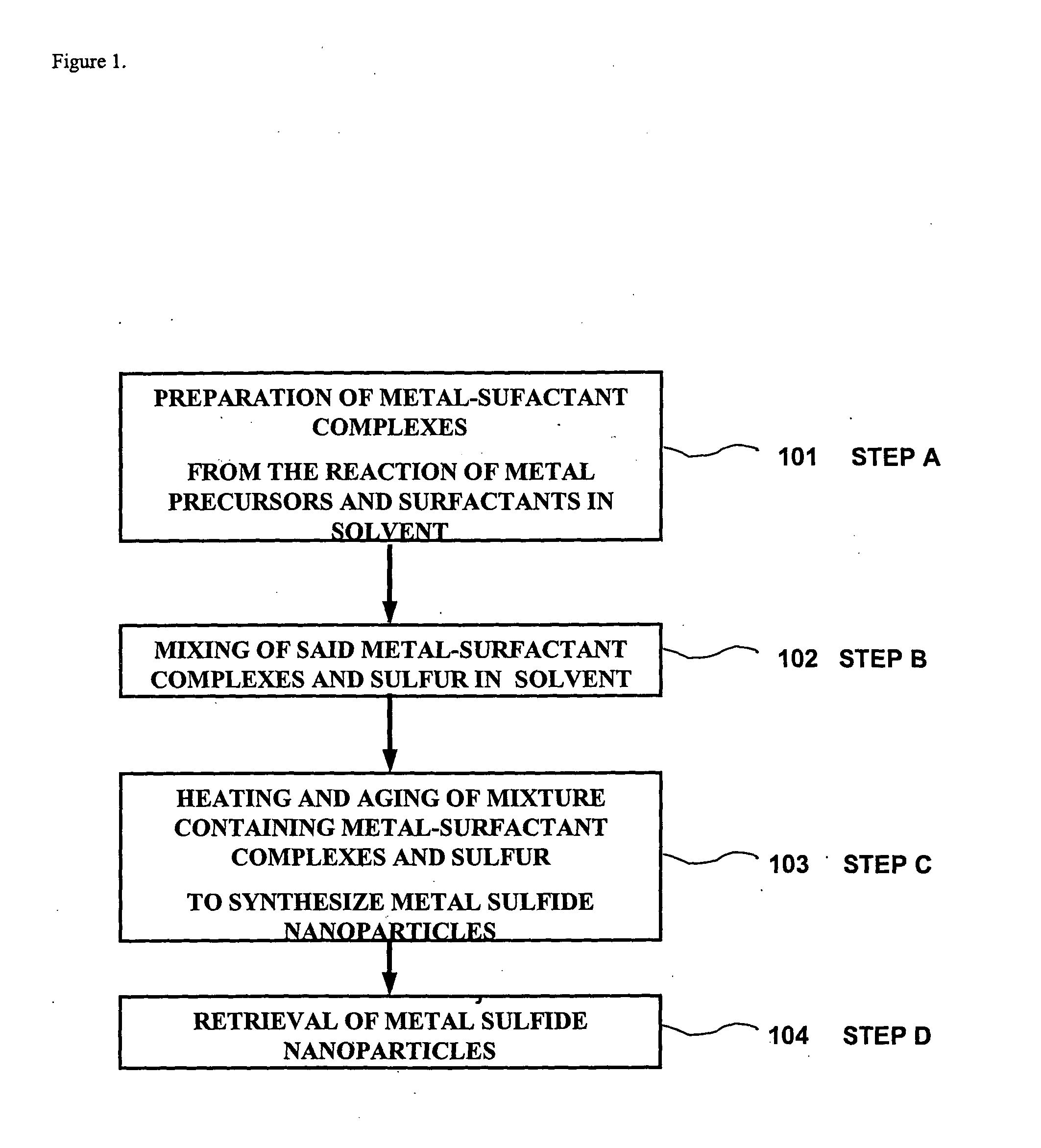
Method for synthesizing nanoparticles of metal sulfides
A synthetic method of fabricating highly crystalline and uniform nanoparticles of metal sulfides, doped metal sulfides, and multi-metallic sulfides disclosed, using no-toxic and inexpensive reagents. A typical synthetic method comprises the steps of, synthesis of metal-surfactant complexes from the reaction of metal precursors and surfactant, addition of sulfur reagent to the solution containing said metal-surfactant complexes followed by heating to high temperature, aging at that temperature to produce metal sulfide nanoparticles and completing the formation of synthesis of nanoparticles metal sulfides and multi-metallic sulfides by adding a poor solvent followed by centrifuging.
Owner:SEOUL NATIONAL UNIVERSITY
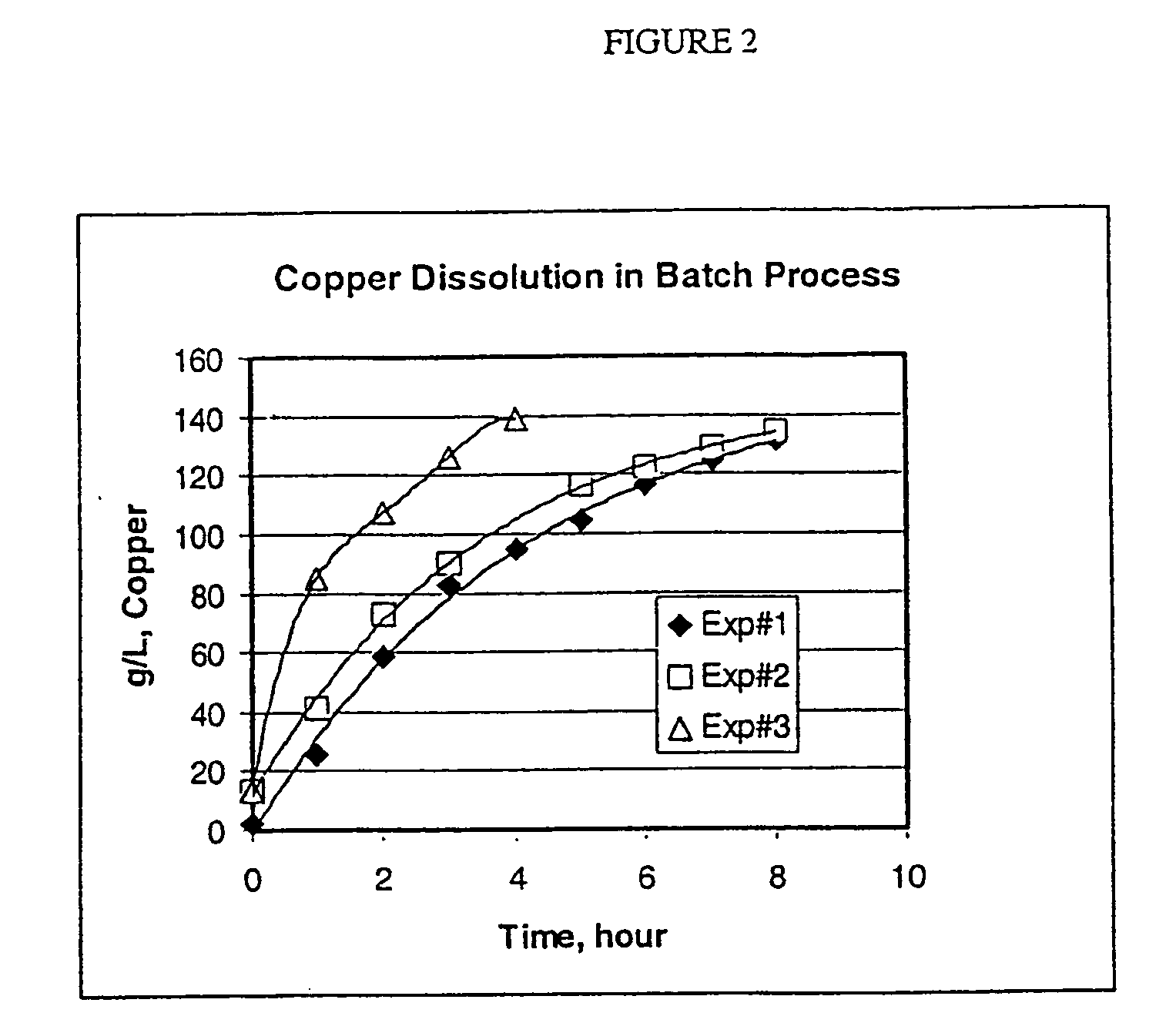
Process for the dissolution of copper metal
ActiveUS20050130866A1High initial dissolution rateShorten the timeGroup 1/11 element organic compoundsSolvent extractionGramEquivalent weight
Process for producing a copper-containing aqueous solution, in which a copper mass is dissolved in the presence of air in an aqueous leach liquor containing monoethanolamine and an acid, wherein the amount of acid equivalents is between 0.05 and about 0.7 times the equivalents of monoethanolamine, and wherein the rate of copper dissolution into the aqueous leach liquor is greater than about 4.3 grams of copper per liter of leach liquor per hour until a product having at least about 80 grams per liter is obtained.
Owner:KOPPERS PERFORMANCE CHEM
Synthetic control of metal oxide nanocrystal sizes and shapes
A general, reproducible, and simple synthetic method that employs readily available chemicals permits control of the size, shape, and size distribution of metal oxide nanocrystals. The synthesis entails reacting a metal fatty acid salt, the corresponding fatty acid, and a hydrocarbon solvent, with the reaction product being pyrolyzed to the metal oxide. Nearly monodisperse oxide nanocrystals of Fe3O4, Cr2O3, MnO, Co3O4, NiO, ZnO, SnO2, and In2O3, in a large size range (3-50 nm), are described. Size and shape control of the nanocrystals is achieved by varying the reactivity and concentration of the precursors.
Owner:THE BOARD OF TRUSTEES OF THE UNIV OF ARKANSAS
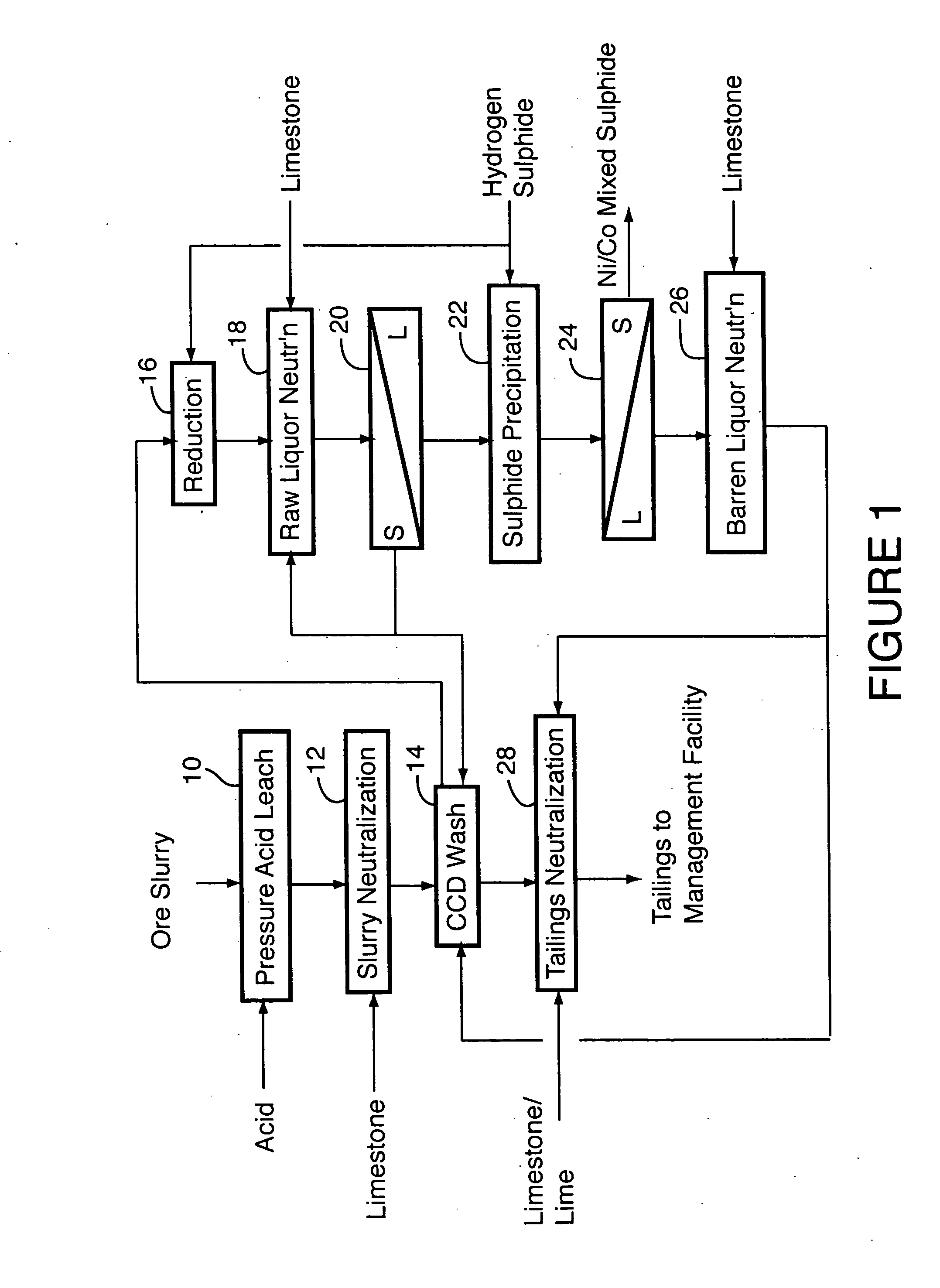
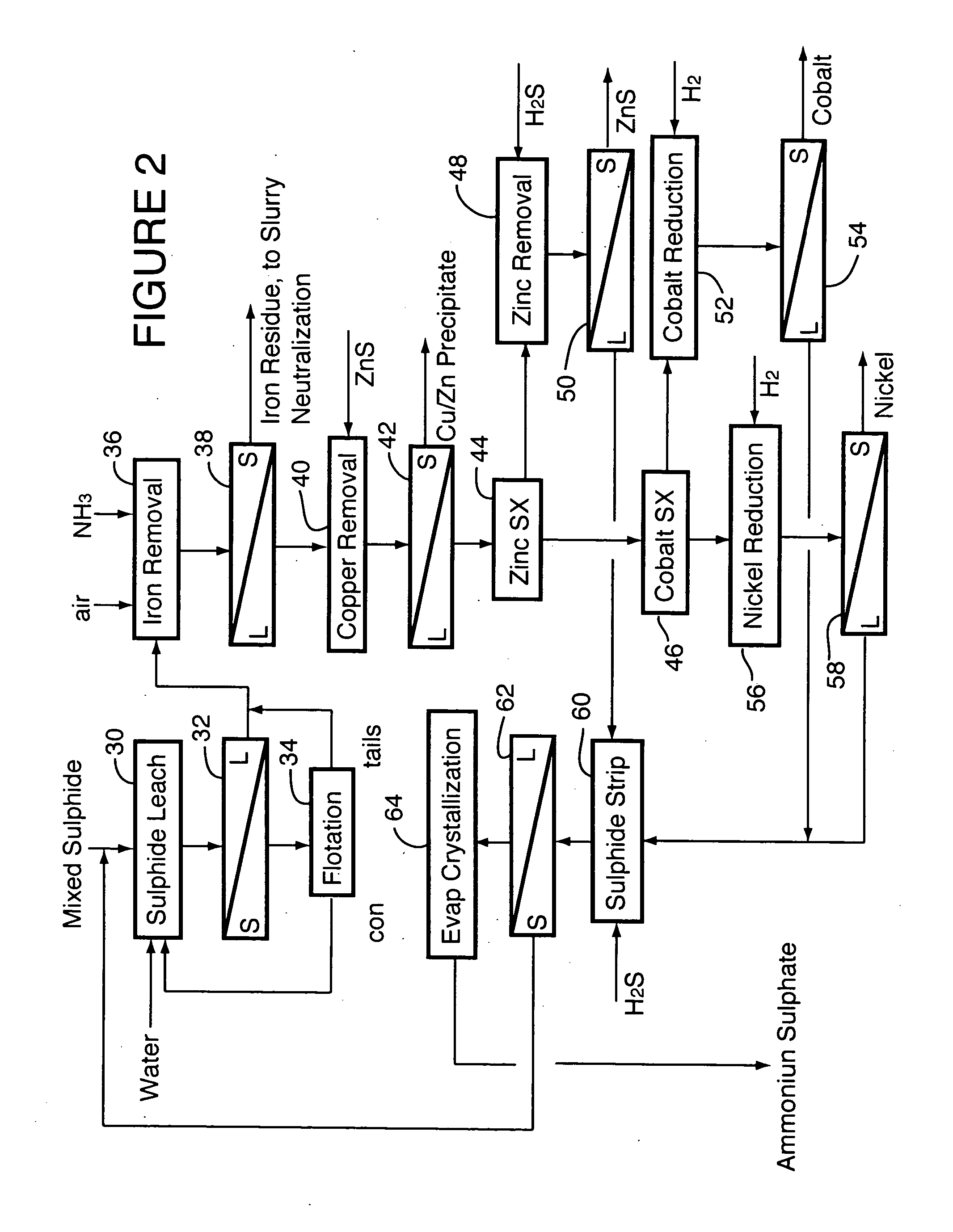
Process for recovery of nickel and cobalt from laterite ore
ActiveUS20060228279A1Efficient separation and recoveryHigh purityCobalt sulfidesSolvent extractionFree solutionSlurry
A process for recovering nickel and cobalt values from nickel- and cobalt-containing laterite ores as an enriched mixed nickel and cobalt sulphide intermediate and for producing nickel and cobalt metal from the nickel and cobalt sulphide intermediate. The laterite ore is leached as a slurry in a pressure acid leach containing an excess of aqueous sulphuric acid at high pressure and temperature, excess free acid in the leach slurry is partially neutralized to a range of 5 to 10 g / L residual free H2SO4 and washed to yield a nickel- and cobalt-containing product liquor, the product liquor is subjected to a reductant to reduce any Cr(VI) in solution to Cr(III), the reduced product liquor is neutralized to precipitate ferric iron and silicon at a pH of about 3.5 to 4.0, and the neutralized and reduced product liquor is contacted with hydrogen sulphide gas to precipitate nickel and cobalt sulphides. The precipitated nickel and cobalt sulphides can be leached in a water slurry in a pressure oxidation leach, the leach solution subjected to iron hydrolysis and precipitation, the iron-free solution contacted with zinc sulphide to precipitate copper, the iron- and copper-free solution subjected to zinc and cobalt extraction by solvent extraction to produce a nickel raffinate, the nickel raffinate contacted with hydrogen gas to produce nickel powder and the cobalt strip solution from the solvent extraction step contacted with hydrogen gas to produce cobalt powder.
Owner:SHERRITT INTERNATIONAL
Metal oxide-containing nanoparticles
InactiveUS20060084278A1Easy to modifyRaise the ratioMaterial nanotechnologyOxide/hydroxide preparationNanoparticleCopper oxide nanoparticles
The present invention provides a copper oxide-containing composition that includes copper oxide nanoparticles and one or more heteroatom donor ligands bonded to the surface of the nanoparticles, where x and y are numbers having a ratio that is equal to the ratio of the average number of M atoms to the average number of 0 atoms in the nanoparticles. The nanoparticles are stabilized by the one or more heteroatom donor ligands which act as a protective layer that cap the surface of the nanoparticles. The present invention also provides a solution of the copper oxide nanoparticles that may be applied to a substrate and then subsequently reduced to copper metal. Finally, the invention provides a method of preparing the copper oxide nanoparticles.
Owner:WAYNE STATE UNIV
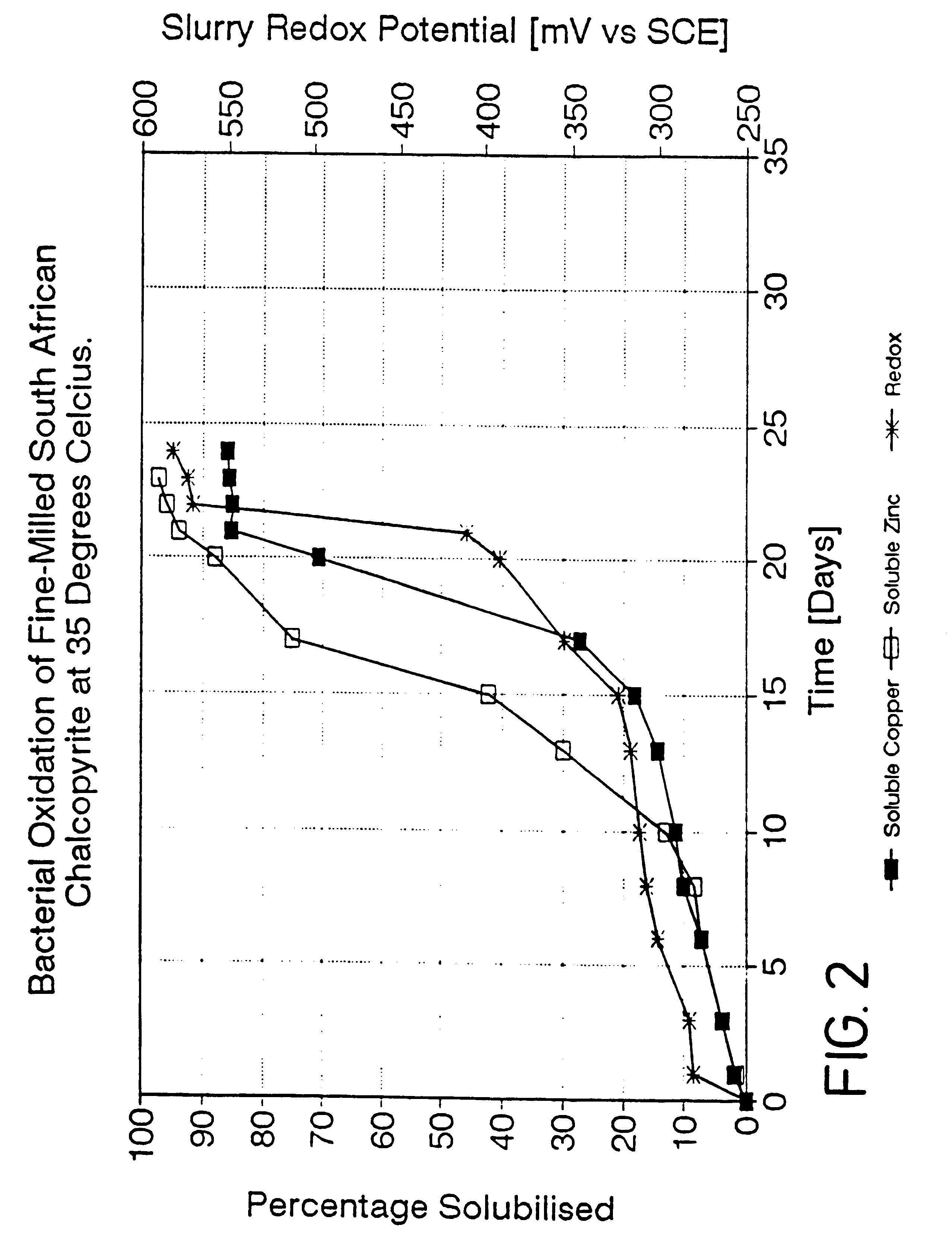
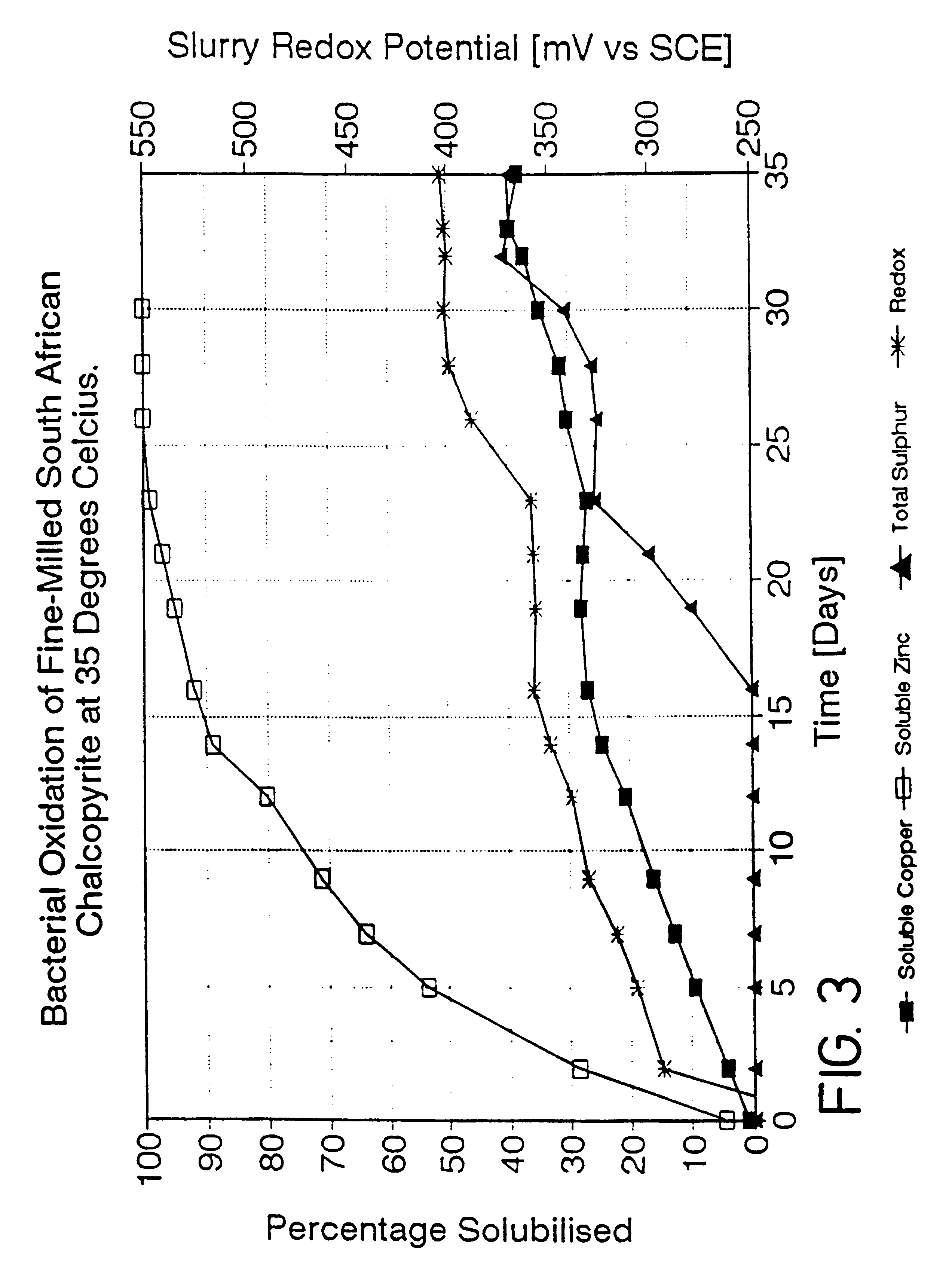
Process for the rapid leaching of chalcopyrite in the absence of catalysts
InactiveUS6277341B1Increase surface areaImprove misalignmentSolvent extractionGold compoundsPregnant leach solutionChalcopyrite
Owner:MINTEK
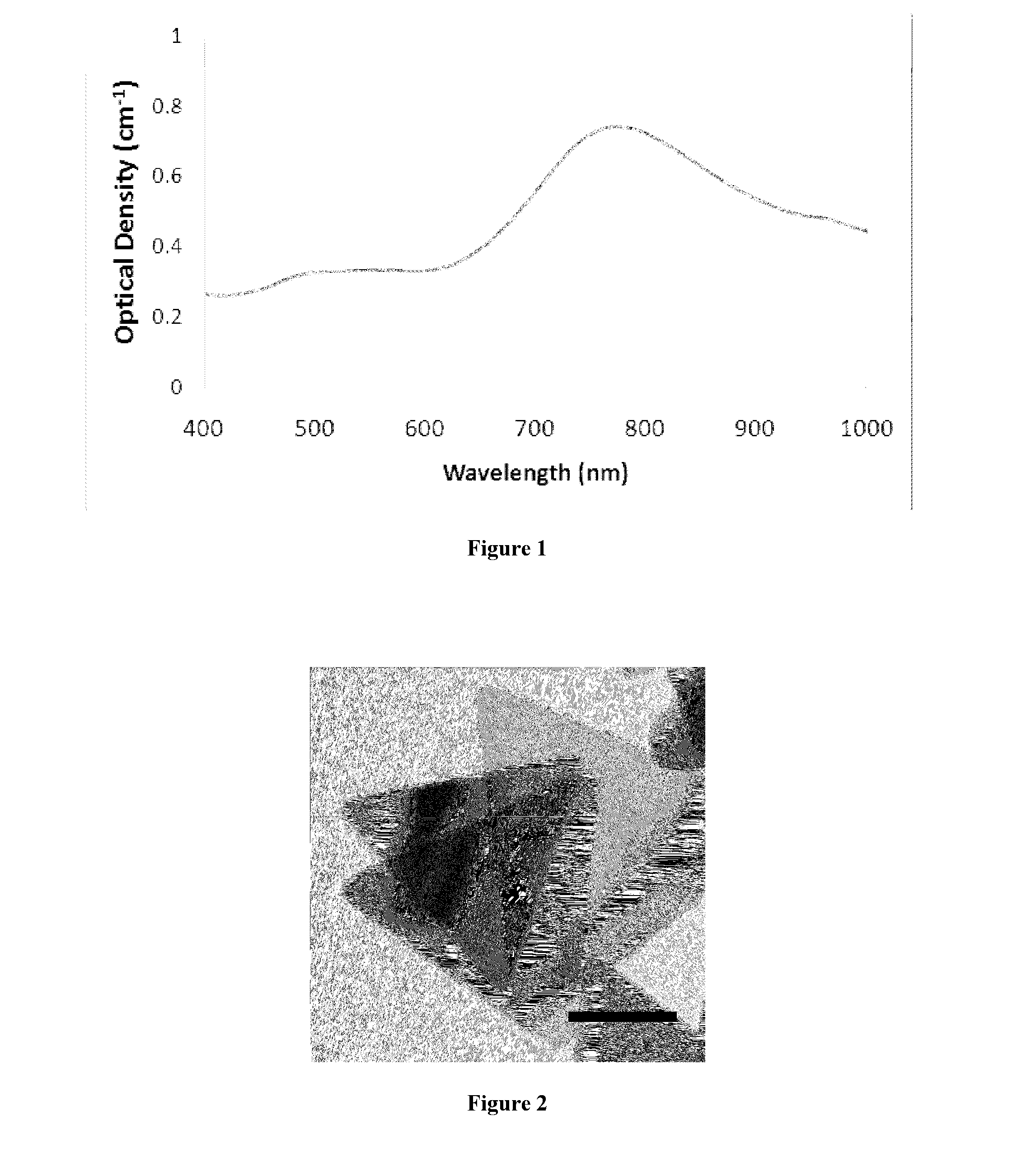
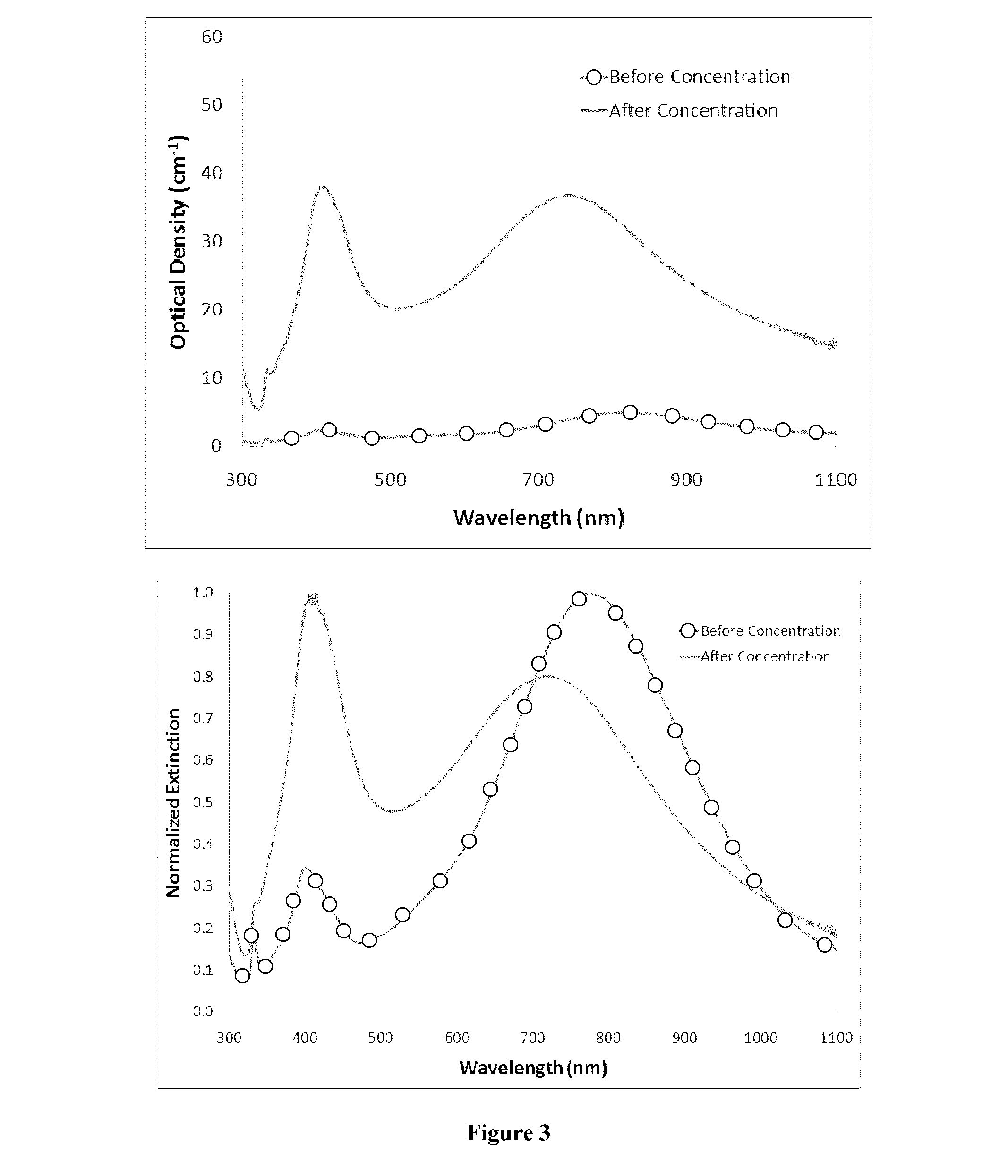
Silver nanoplate compositions and methods
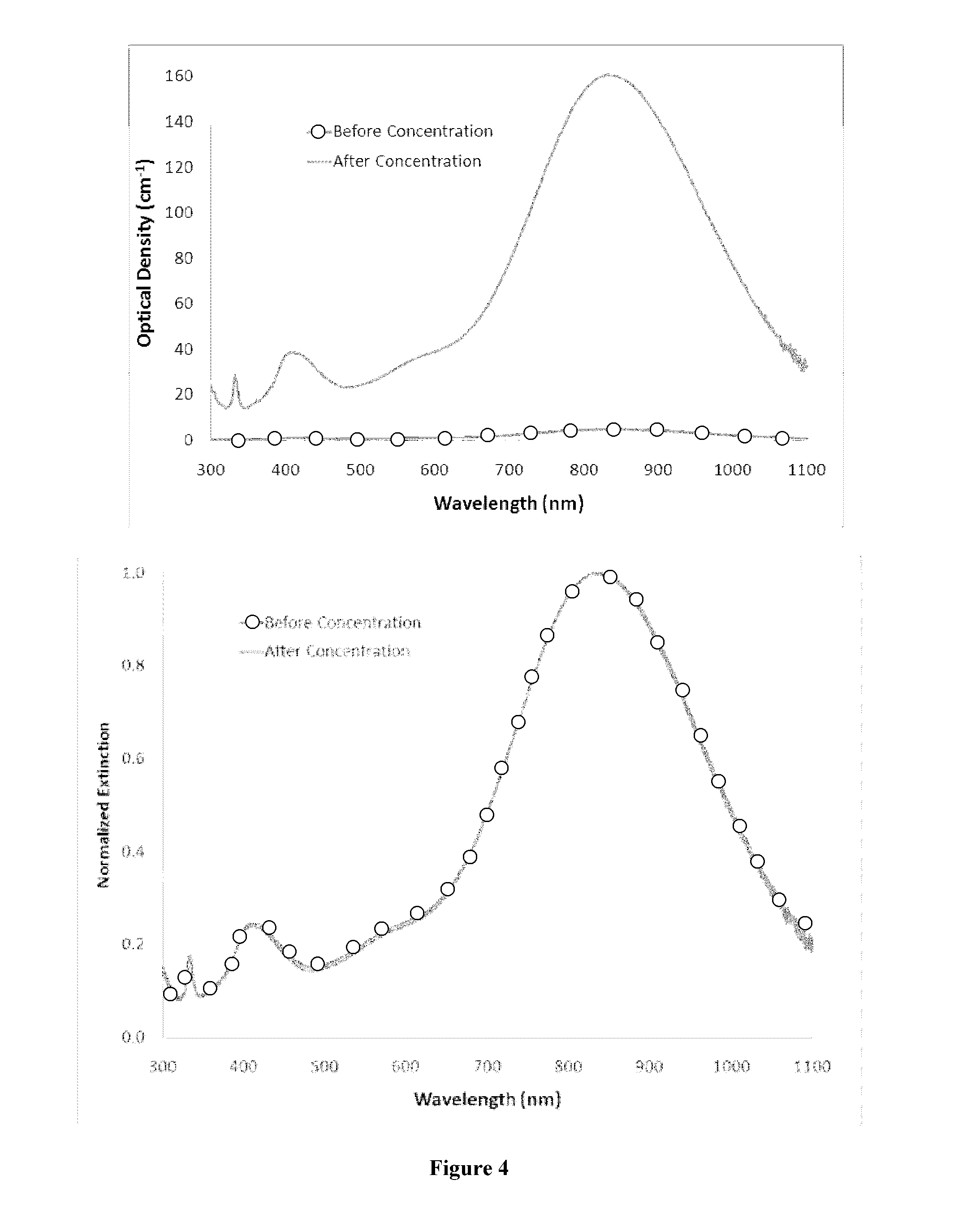
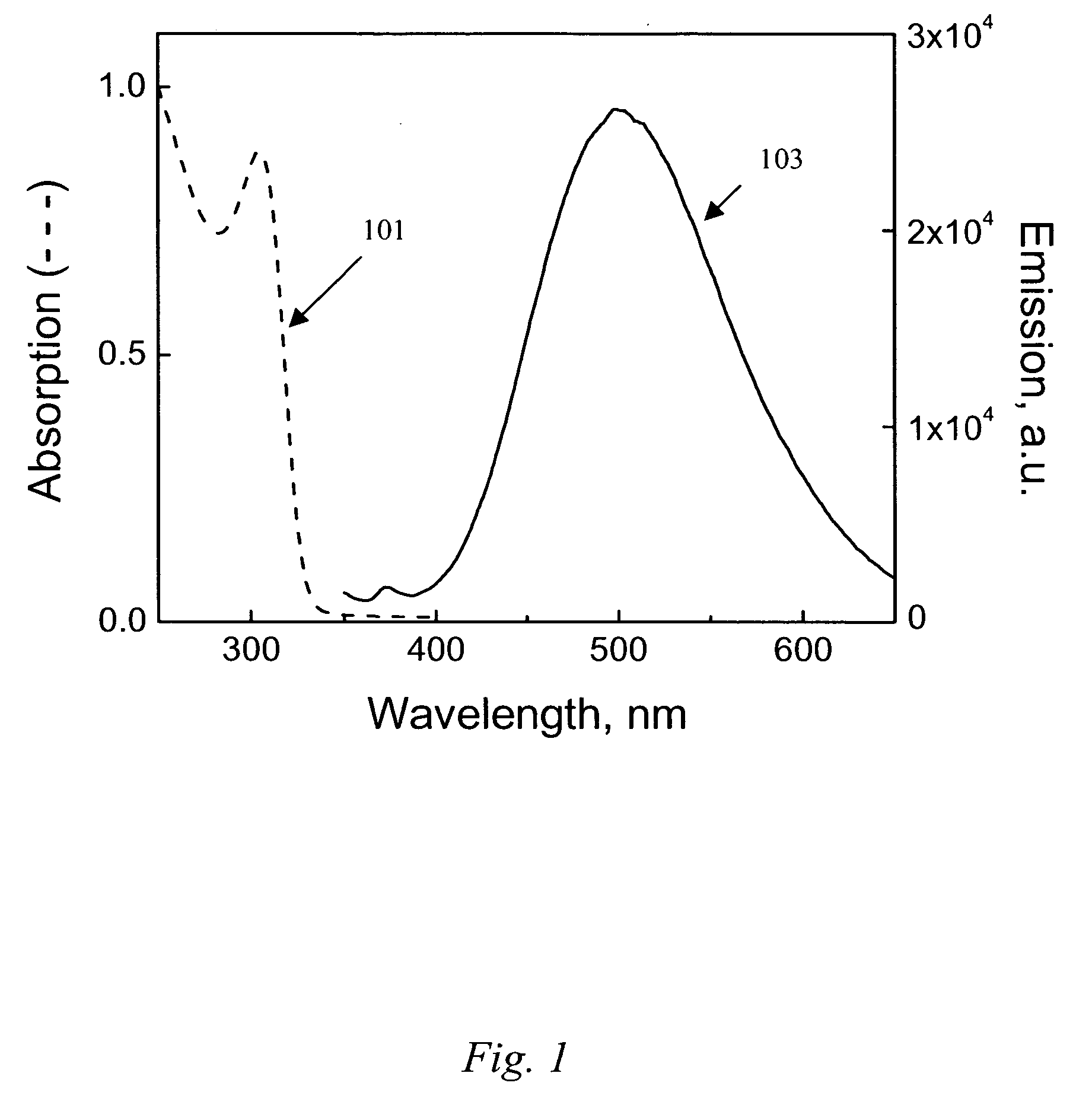
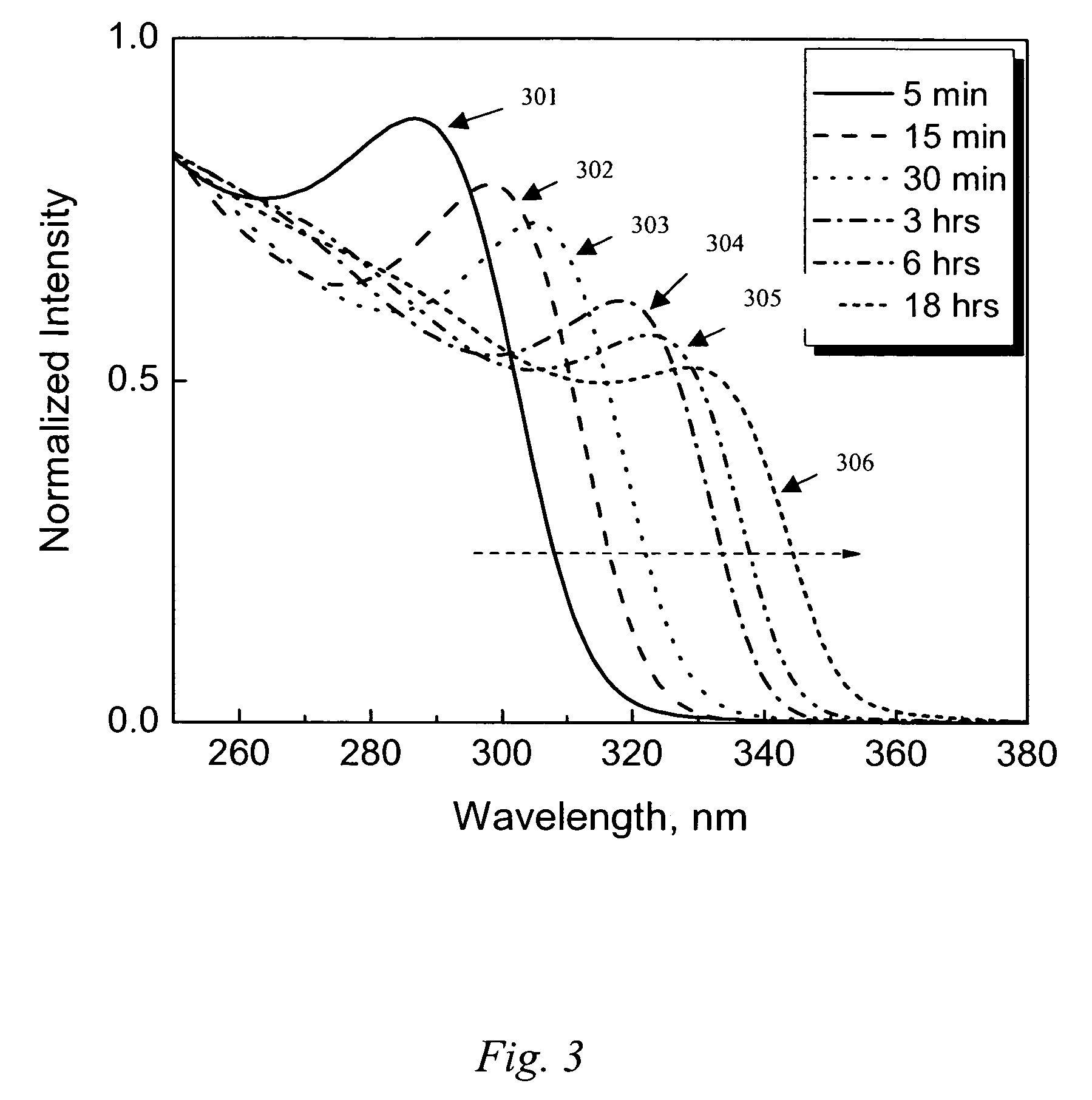
ActiveUS20140105982A1Easy loadingUndesirable degradationAntibacterial agentsBiocideNanoparticlePlatelet
Embodiments of the present invention relate to methods for preparing high optical density solutions of nanoplates, such as silver nanoplates or silver platelet nanoparticles, and to nanoparticles, solutions and substrates prepared by said methods. The process can include the addition of stabilizing agents (e.g., chemical or biological agents bound or otherwise linked to the nanoparticle surface) that stabilize the nanoparticle before, during, and / or after concentration, thereby allowing for the production of a stable, high optical density solution of silver nanoplates. The process can also include increasing the concentration of silver nanoplates within the solution, and thus increasing the solution optical density.
Owner:NANOCOMPOSIX LLC
Process for preparing nano-sized metal oxide particles
InactiveUS20050260122A1Efficiently provideNanosized metal oxide particles more efficientlyNanostructure manufactureGold compoundsHigh concentrationAlcohol
The present invention is directed to novel sol-gel methods in which metal oxide precursor and an alcohol-based solution are mixed to form a reaction mixture that is then allowed to react to produce nanosized metal oxide particles. The methods of the present invention are more suitable for preparing nanosized metal oxide than are previously-described sol-gel methods. The present invention can provide for nanosized metal oxide particles more efficiently than the previously-described sol-gel methods by permitting higher concentrations of metal oxide precursor to be employed in the reaction mixture. The foregoing is provided by careful control of the pH conditions during synthesis and by ensuring that the pH is maintained at a value of about 7 or higher.
Owner:KANEKA CORP +1