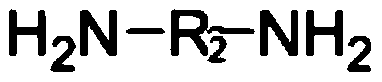Patents
Literature
55results about How to "Uniform release rate" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
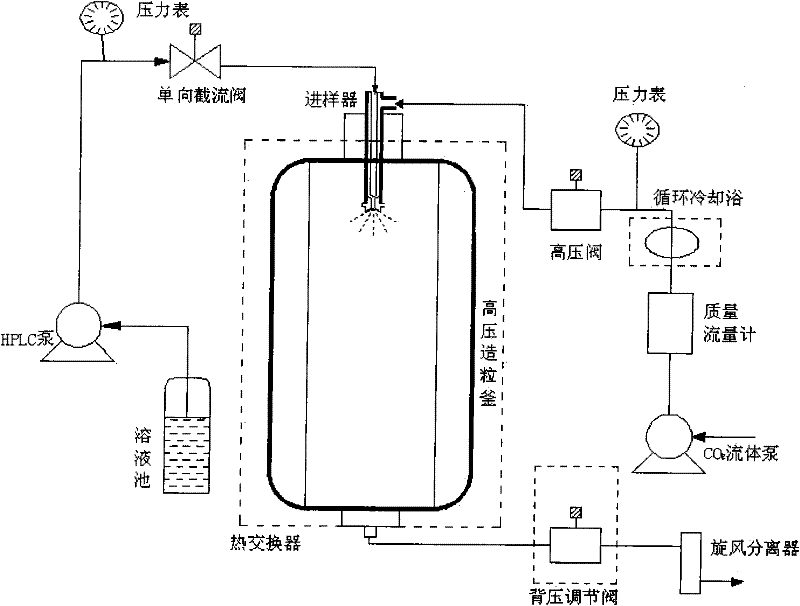
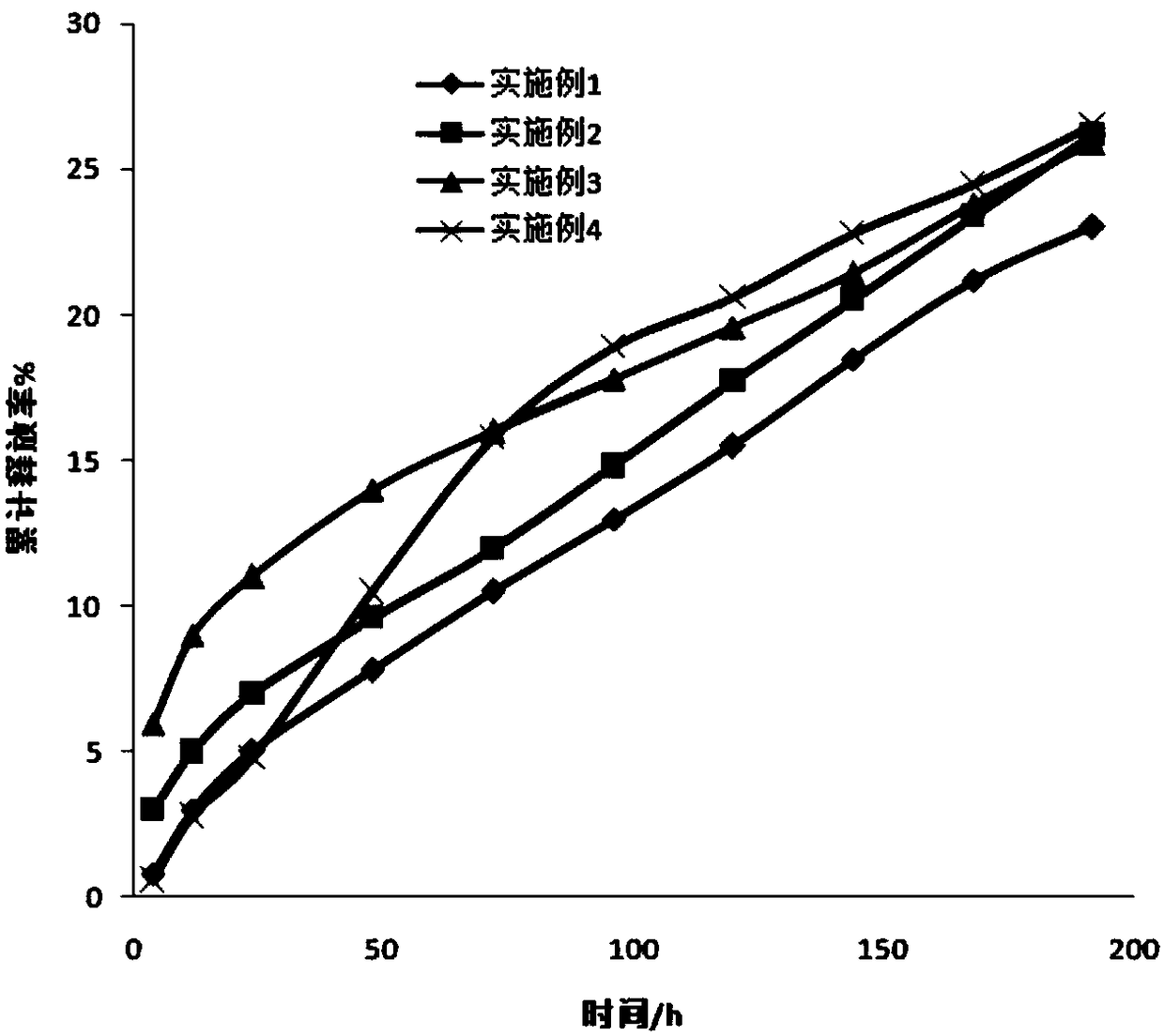
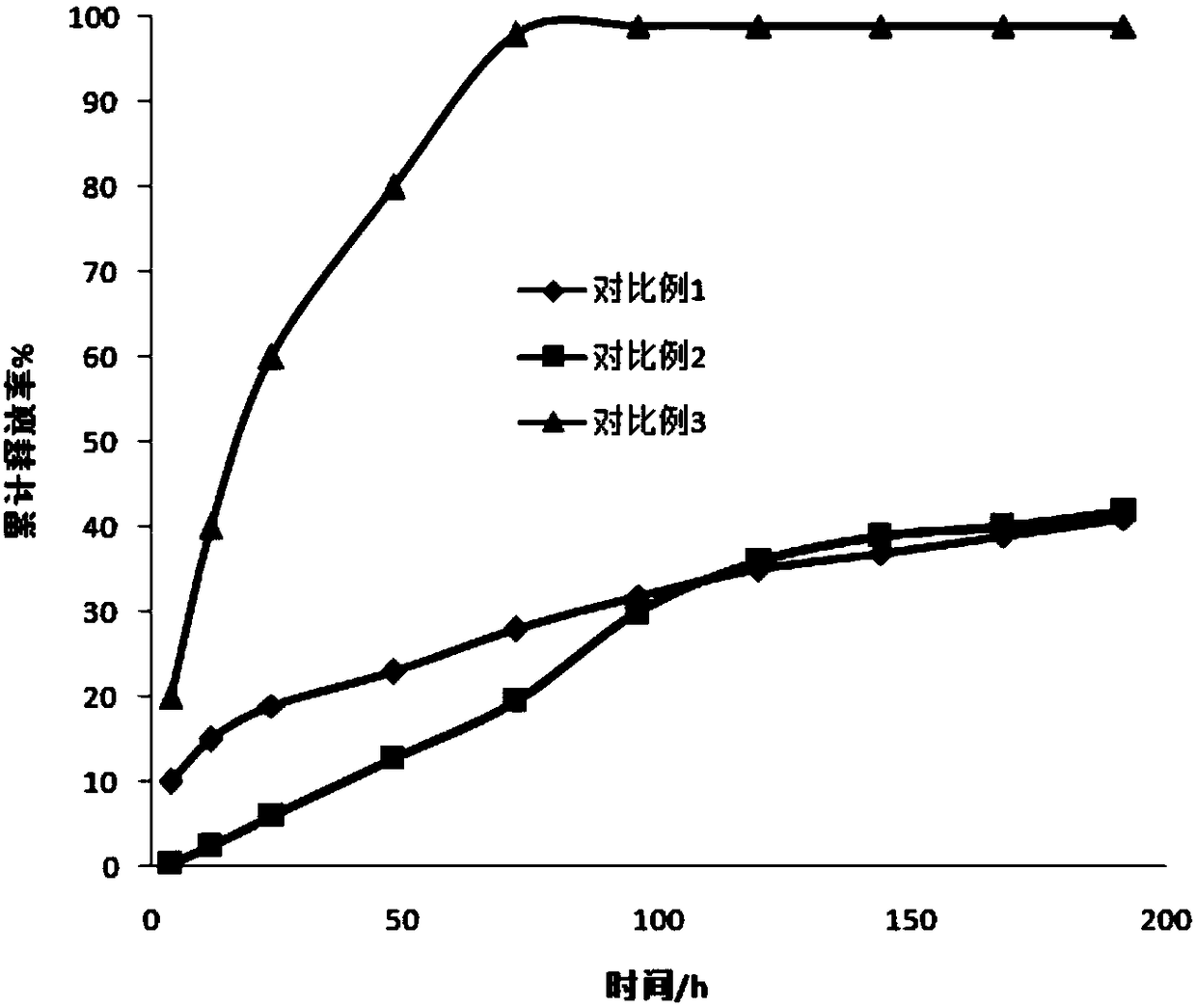
Controlled release type potassium ferrate composite as well as preparation method and application thereof
ActiveCN103896389AIssues Affecting DissolutionGuaranteed DissolutionIron compoundsWater/sewage treatment using germicide/oligodynamic-processFoaming agentControl release
The invention discloses a controlled release type potassium ferrate composite as well as a preparation method and application thereof. The preparation method is as follows: by taking a high polymer which is biodegradable and environment-friendly as a framework material, dissolving the high polymer in a low-boiling point organic solvent, adding a pore-foaming agent, a dispersing agent and high-purity potassium ferrate, adding the mixture into a medium for emulsifying and curing, and filtering and drying the cured product to obtain the controlled release type potassium ferrate composite. The composite is in a water environment, so that a net-shaped duct structure is formed in an inert framework material by virtue of the hydrophilcity of the pore-foaming agent, and therefore, the potassium ferrate is effectively aided to release from the composite, and a problem that dissolution of active ingredients is affected due to texture compaction of the framework material is avoided; the composite is different from the conventional disposable oxidant, controllable in release speed in water, uniform in velocity, long in holding time, good in storage stability, and has wide application prospect on in-situ repairing polluted underground water.
Owner:SOUTH CHINA NORMAL UNIVERSITY
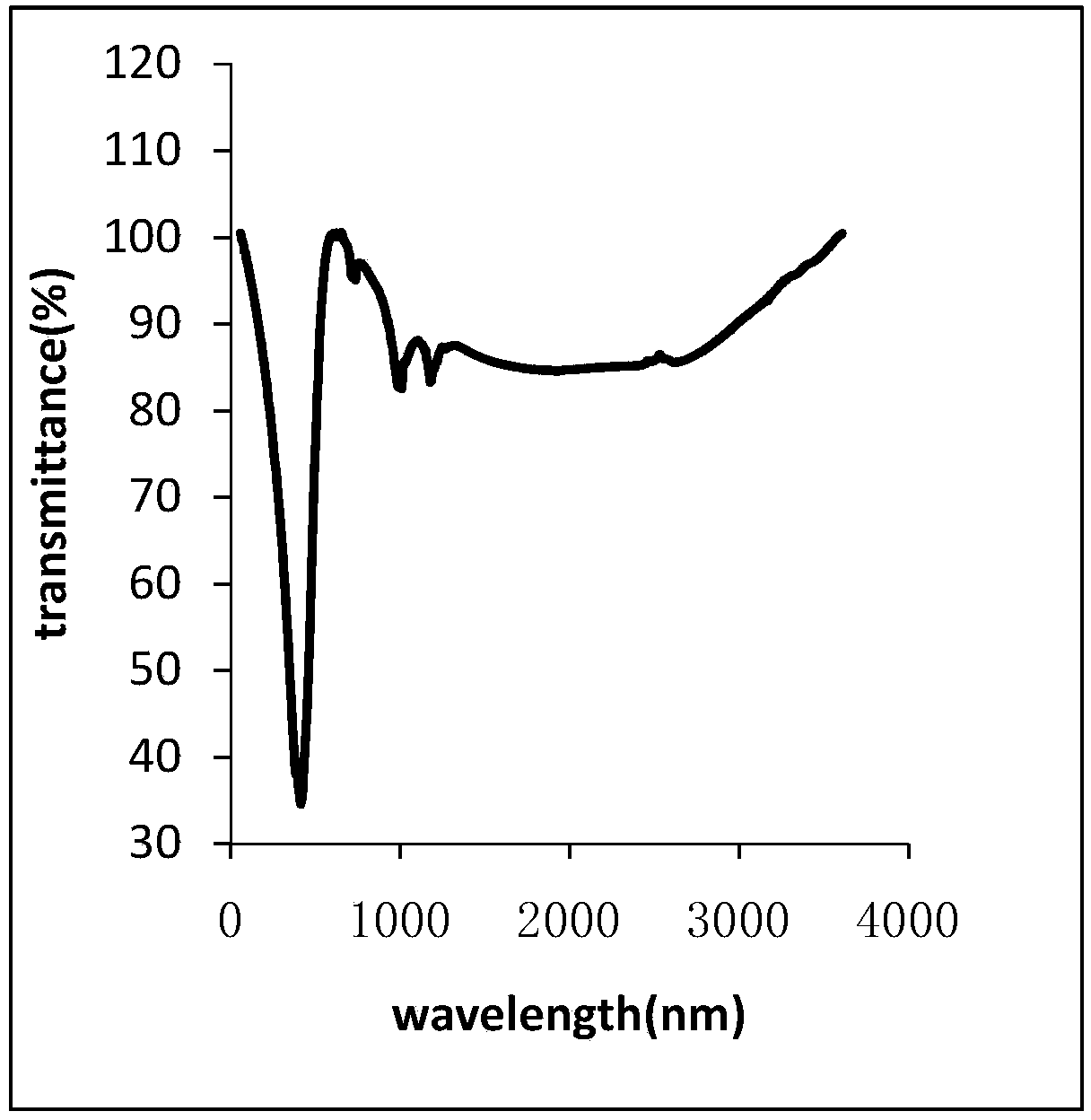
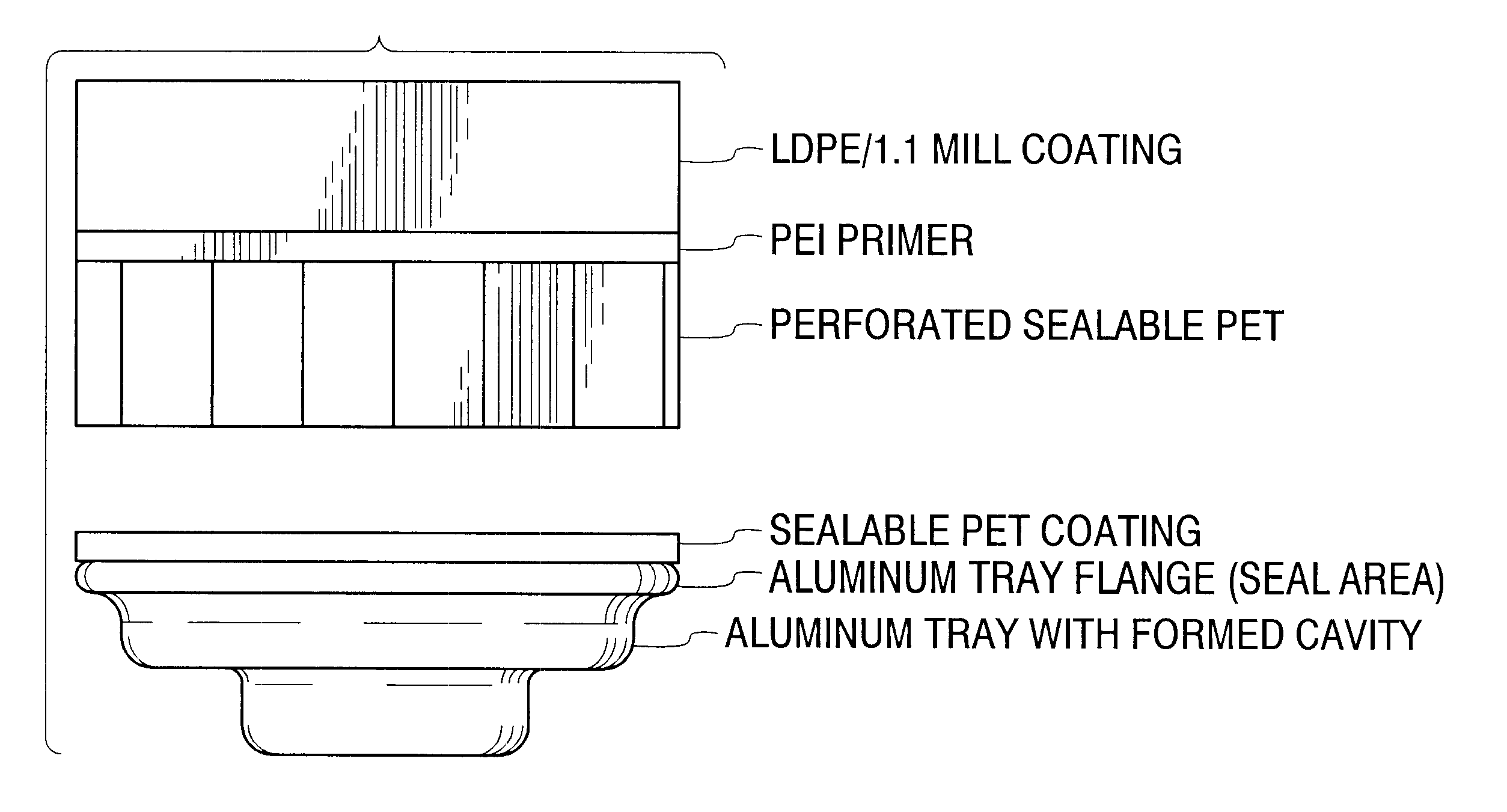
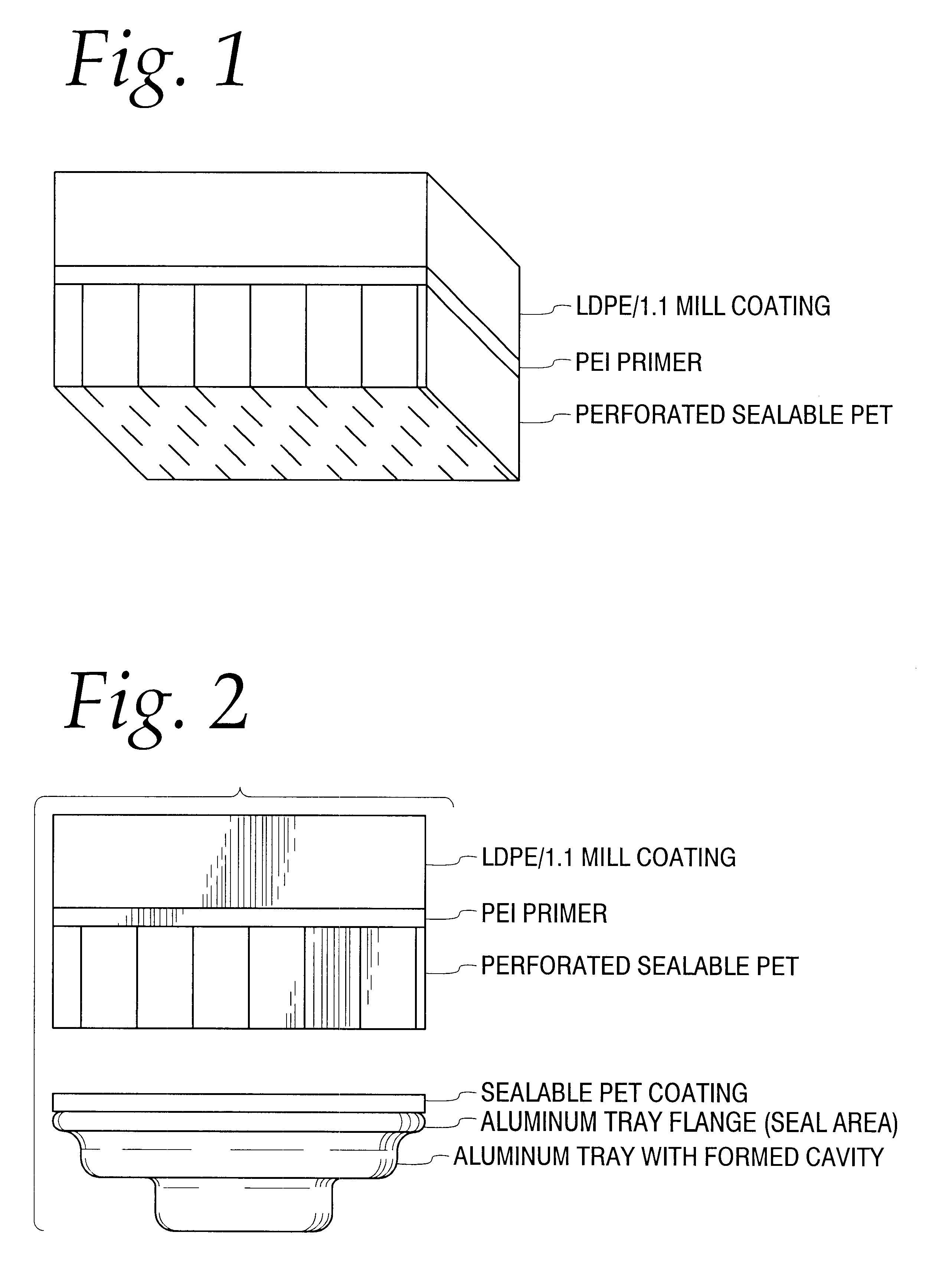
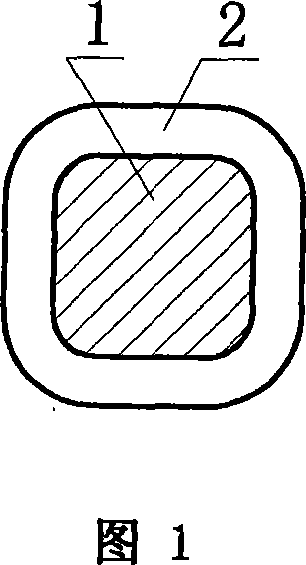
Permeable membranes having high temperature capabilities
InactiveUS6638588B1Prevents undesired spillageAvoid layeringWrappersDispensing apparatusFilm structureHigh heat
A multilayer permeable film structure having high temperature capabilities comprising a first, perforated sealable layer; a second primer layer and a third outer layer is disclosed. The multilayer permeable film structure can be heat sealed to a formed tray containing a volatile material.
Owner:PECHINEY EMBALLAGE FLEXIBLE EURO
Surface modified coat of bracket of blood vessel, and preparation method
A coated layer for modifying the surface of vascular scaffold by controlling the content of polyoxyethene in the coated layer to regulate its medicine-carrying performance and the release of medicinal is a polysulfone-polyoxyethene block copolymer with 10000-300000 for its number average molecular weight. Its preparing process features that said block copolymer is coated on the surface of said vascular scaffold directly or after the medicine is added to its solution.
Owner:SOUTHEAST UNIV
Aerogel material and preparation method and application thereof
ActiveCN104525132AImprove adsorption capacityGood slow releaseBiocideOther chemical processesOrganic chemistryThermal conductivity
The invention discloses an aerogel material. According to performance parameters of the aerogel material, the average hole diameter ranges from 5 nm to 80 nm, the specific surface area ranges from 5 m<2> / g to 300 m<2> / g, the density ranges from 50 kg / m<3> to 500 kg / m<3>, the coefficient of thermal conductivity ranges from 0.018 W / mK to 0.060 W / mK, the porosity ranges from 60% to 99%, the water absorptivity is less than 5%, and the liquid adsorption capacity is 5-15 times of gravimetric absorption. The aerogel material is good in liquid adsorption capacity and has excellent slow release performance, liquid release speed is uniform, liquid release time is long, and the aerogel material is safe, has no toxic or side effects and can be widely applied to slow release materials of parasiticides or perfume.
Owner:ZHANGJIAGANG ONECHIP BIO TECH CO LTD
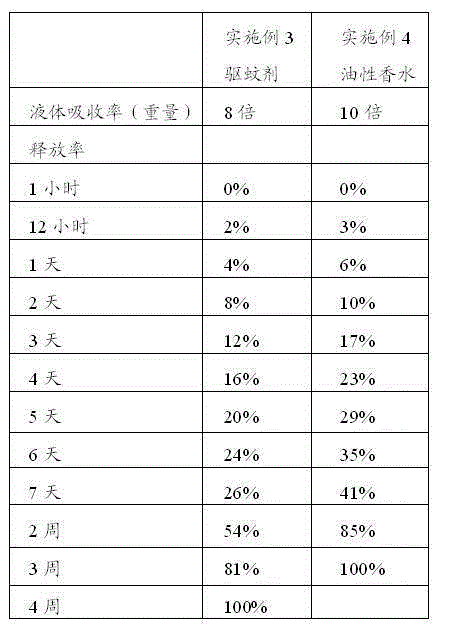
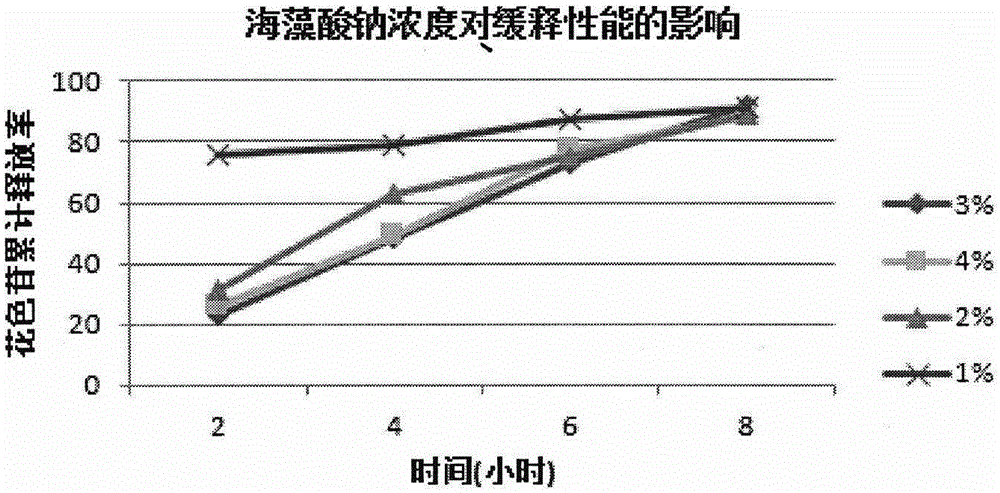
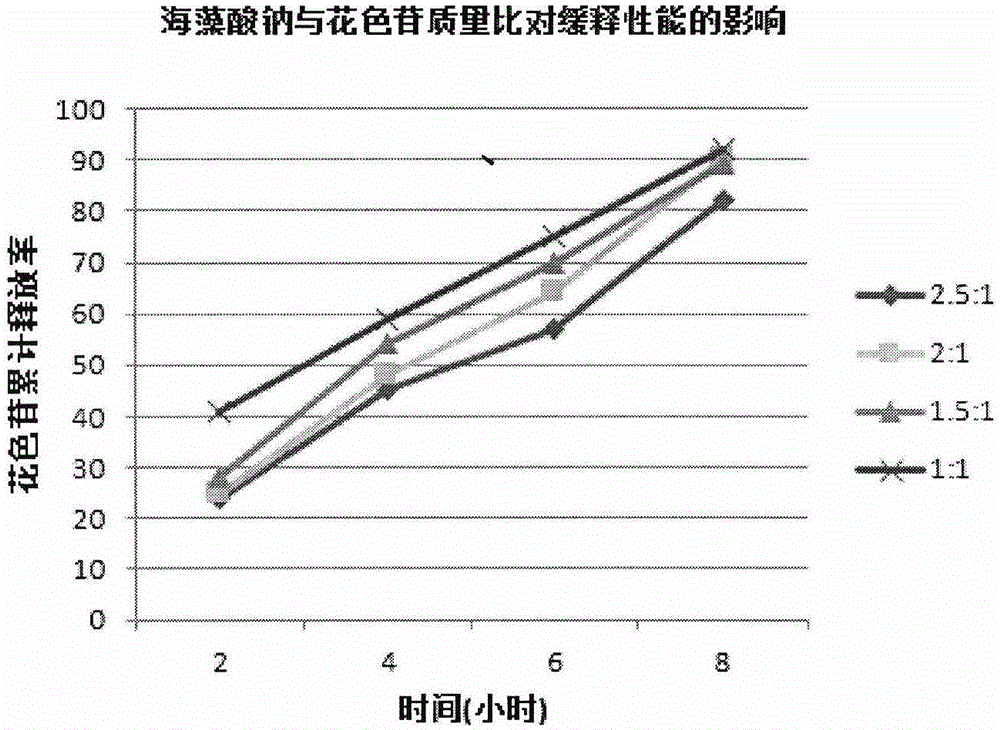
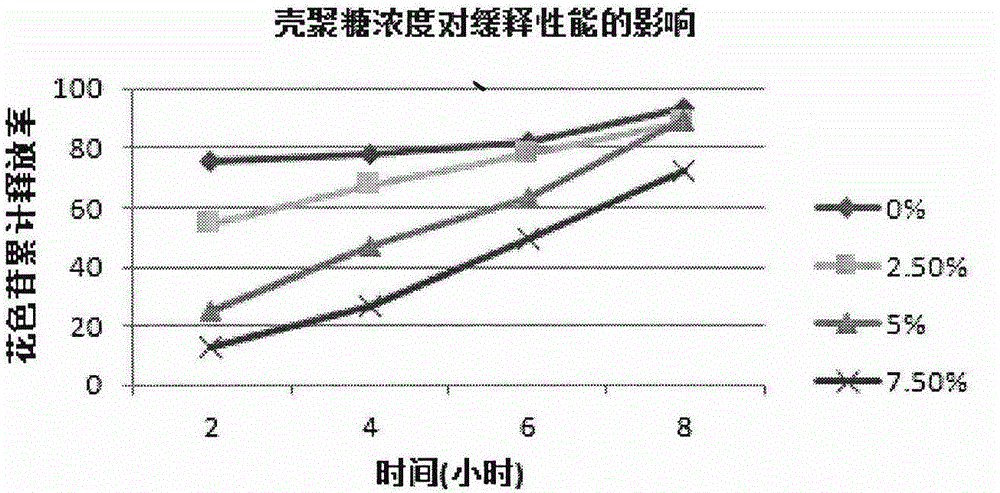
Lycium ruthenicum anthocyanin crude extract and controlled-release microcapsule thereof
ActiveCN105079281AStrong antioxidant activityAvoid purification processPharmaceutical delivery mechanismAntinoxious agentsDrug releaseControlled Release Capsule
The invention provides a lycium ruthenicum anthocyanin crude extract and a controlled-release microcapsule thereof. The anthocyanin crude extract has good antioxidant activity, so that the further purification for an anthocyanin monomeric compound is prevented, and meanwhile, a preparation method is simple and convenient, production materials are easily available, and the production and use costs of the lycium ruthenicum anthocyanin are reduced. The controlled-release microcapsule is an eight-hour controlled-release capsule, the drug release is uniform in speed and complete, the bioavailability of the drug is improved, and the utilization ratio of the drug is guaranteed.
Owner:CHINA ACAD OF SCI NORTHWEST HIGHLAND BIOLOGY INST
Ultrafine fibre preparation of taxol and method and apparatus for preparing the same
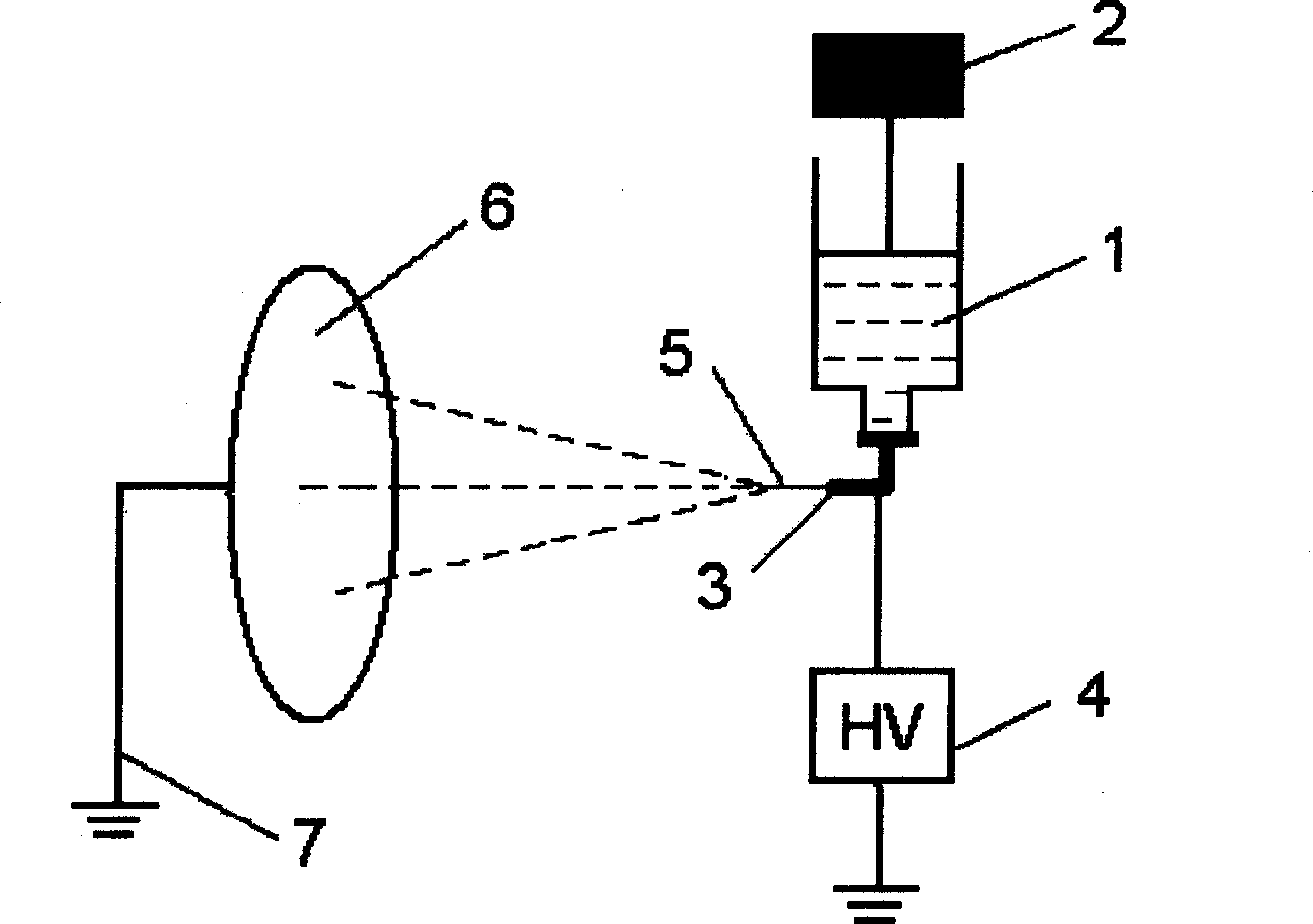
InactiveCN1543929AUniform release rateNo need for secondary surgery to removeOrganic active ingredientsFilament/thread formingNonwoven fabricBiomedical engineering
The invention relates to a superfine fiber formulation of Paclitaxel, its preparation process and apparatus, wherein the preparing process comprises dissolving the Paclitaxel into biologically degradable macromolecular solution, carrying out electrical spinning, forming superfine fiber nonwoven fabrics or fabric felt coated by Paclitaxel. The formulation is specially adapted for the localized treatment after tumor operation.
Owner:CHANGCHUN INST OF APPLIED CHEMISTRY - CHINESE ACAD OF SCI
Poly-L-lactic acid and polyethyleneglycol block copolymer (PLLA-PEG) supported 5-fluorouracil nanoparticles and preparation method thereof
InactiveCN102525933ALow toxicityReduce releaseOrganic active ingredientsPowder deliveryPolyvinyl alcoholNanoparticle
The invention relates to the field of medicinal preparations, in particular to poly-L-lactic acid and polyethyleneglycol block copolymer (PLLA-PEG) supported 5-fluorouracil (5-FU) nanoparticles and a preparation method thereof. The preparation comprises the following components in percentage by weight: 50 to 80 percent of PLLA-PEG, 5 to 20 percent of 5-FU, 1 to 5 percent of beta-cyclodextrin (Beta-CD), 5 to 15 percent of polyvinyl alcohol (PVA) and 5 to 25 percent of PEG. The PLLA-PEG supported 5-FU nanoparticles have the advantages of small average particle size, narrow particle size distribution, high medicine-loading capacity, low toxicity, good targeting performance and the like.
Owner:SHANGHAI HI TECH UNITED BIO TECHCAL RES
Trimetazidine hydrochloride bi-layer osmotic pump controlled release tablet and preparation method thereof
ActiveCN103735528ARelease constant speedSmall toxicityOrganic active ingredientsPharmaceutical delivery mechanismSide effectMotility
The invention provides a trimetazidine hydrochloride bi-layer osmotic pump controlled release tablet and a preparation method thereof. The osmotic pump controlled release tablet is formed by a drug-containing layer, a booster layer and a coating film, wherein the drug-containing layer comprises the following components in percentage by weight: 10-50% of trimetazidine hydrochloride, 30-80% of suspension agent and the balance of other auxiliary materials; the booster layer comprises the following components in percentage by weight: 20-90% of swelling agent, 5-70% of osmotic active substance and 0.5-5% of lubricating agent; the semipermeable coating film comprises 10-20g of semipermeable high polymer material and 1-5g of water-soluble pore-forming agent every 100 tablets. The trimetazidine hydrochloride bi-layer osmotic pump controlled release tablet can realizes constant release of drug in the body of a patient without being affected by pH value of a medium environment, enzyme, gastrointestinal motility and food, and is capable of maintaining the stability of plasma concentration of drug, reducing toxic and side effects of drug, decreasing dosing frequency and improving compliance of the patient.
Owner:SHENYANG PHARMA UNIVERSITY
Buccal cigarette containing tobacco full-component extract and preparation method of buccal cigarette
InactiveCN105167166AMaintenance contentConsistent and even tasteTobacco treatmentBiotechnologyMicrobiology
The invention belongs to the field of a tobacco product, and discloses a buccal cigarette containing a tobacco full-component extract and a preparation method of the buccal cigarette. The buccal cigarette containing the tobacco full-component extract consists of functional ingredients, a filling agent, a flavoring agent, a lubricating agent and a binding agent, wherein the functional ingredients include a tobacco extract and tobacco extract particles; the tobacco extract particle comprises at least two substrate layers different in tobacco extract content; the surfaces of the substrate layers are coated with coatings layer by layer, and the substrate layers contain excipient; and in the tobacco extract particle, the tobacco extract content in the substrate layers from inside to outside is gradually reduced. In the buccal cigarette containing the tobacco full-component extract, the tobacco component is provided by the tobacco extract and the tobacco extract particle which are complementary during releasing, so as to keep the content of the tobacco component in the aromatized cigarette and to offer durable and uniform taste to consumers.
Owner:GUANGDONG BRANCH OF CHINA TOBACCO GENERAL
Tablet covering equipment
InactiveCN107890425AUniform release rateGood coating effectSpray nozzlesPharmaceutical product form changeEngineeringGastric fluid
The invention discloses tablet covering equipment. The tablet covering equipment comprises an operation chamber, a spray gun, an air injection device, a feed inlet device and a recycling device; the spray gun is arranged at the bottom of the operation chamber, and the air injection device comprises a sleeve sleeving the spray gun, an air injection channel arranged in the sleeve and injecting partsconnected with the air injection channel; the air injection part is aslant arranged; since the air injection part is aslant arranged, the tilting directions among the injection parts are the same. Byaslant arranging the air injection part, after being injected, feed can rotate in the sky, and it is guaranteed that covering is uniformly conducted; the time for the feed to stay in the sky is long,the time for the feed to be in contact with a covering material is also longer, and the covering effect is good; uniform covering makes the releasing speed of tablets in gastric juice constant, and people's intestinal health is effectively protected.
Owner:陈晓盛
Slow-releasing Tamoxifen citrate tablet
InactiveCN1169523CUniform release rateSimple production processPeptide/protein ingredientsPharmaceutical delivery mechanismTamoxifen CitrateMedicine
Owner:BEIJING CREATE FORTUNE TECH IND GRP CO LTD
Mouth mucosa sustained-release preparation for treating oral and periodontal diseases by chlorhexidine
InactiveCN103110610BLong release timeUniform release rateTetracycline active ingredientsDigestive systemDiseasePolyvinyl alcohol
The invention relates to fields of a drug for treating a disease of cari oris mucosa, and preparation thereof, and particularly relates to a mouth mucosa sustained-release preparation for treating oral and periodontal diseases by chlorhexidine. The preparation is composed of a mouth mucosa contact layer and a non-adhesive seal layer; the mouth mucosa contact layer comprises the components of an antibiotic drug and an auxiliary material, of which the weight ratio is (2-20) to (80-98); the non-adhesive seal layer comprises the components of a non-adhesive low-water soluble high-molecular polymer; the non-adhesive low-water soluble high-molecular polymer is selected from polyving akohol; the mouth mucosa sustained-release preparation comprises the components of 4.0g of chlorhexidine, 12.0g of hydroxyethyl cellulose, 5.5g of modified starch, 7.0g of microcrystalline cellulose, 0.5g of grape essence and the balance of polyving akohol. The mouth mucosa sustained-release preparation has the advantages of being convenient to drug delivery, long in drug release time, even in drug release speed, strong in target, strong in mucosa adhesive force and the like; and the preparation method is simple to operate, low in cost, friendly to environment, and suitable for industrial production.
Owner:上海微丸医药开发有限公司
Gum-based chewing cigarette and preparation method thereof
InactiveCN105124747AMaintenance contentConsistent and even tasteTobacco preparationTobacco treatmentBiotechnologyTobacco product
The invention belongs to the field of tobacco products and discloses a gum-based chewing cigarette and a preparation method thereof. The gum-based chewing cigarette comprises the following substances in percentage by weight: 30%-45% of chewing gum base, 5%-15% of tobacco extractive, 10%-20% of tobacco extractive particles, 5.5%-15% of a sweetening agent, 0.5%-5% of an emulsifier, 0%-10% of spice and 0.1%-8% of an antioxidant; the tobacco extractive particles comprise at least two base material layers which are different in tobacco extractive content; the base material layers are coated layer by layer, the surfaces of the base material layers are coated by coating layers and the base material layers contain excipient; and the content of tobacco extractives in the base material layers of the tobacco extractive particles are gradually reduced from inside to outside. Tobacco ingredients in the gum-based chewing cigarette are provided by the tobacco extractives and the tobacco extractive particles, release processes of the tobacco extractives and the tobacco extractive particles complement each other, the content of tobacco ingredients in the gum-based chewing cigarette is maintained and a continuous and uniform taste is provided for consumers.
Owner:GUANGDONG BRANCH OF CHINA TOBACCO GENERAL
A kind of aromatherapy cigarette containing full-component tobacco extract and its preparation method
InactiveCN105105327BMaintenance contentConsistent and even tasteTobacco treatmentCigar manufactureFiberTobacco product
The invention belongs to the field of tobacco products and discloses a fragrance cigarette containing full-component tobacco extracts and a preparation method thereof. The fragrance cigarette containing the full-component tobacco extracts comprises functional components, carbon powder, plant fibers and a bonding agent, wherein the functional components include tobacco extracts and tobacco extract particles, the tobacco extract particles include at least two substrate layers with different tobacco extract contents, the substrate layers are coated layer by layer, the surfaces of the substrate layers are wrapped by coating layers, the substrate layers contain excipient, and the tobacco extract contents of the substrate layers in the tobacco extract particles are gradually reduced from inside to outside. According to the fragrance cigarette containing the full-component tobacco extracts, the tobacco components include the tobacco extracts and the tobacco extract particles, and the release processes of the tobacco extracts and the tobacco extract particles are complementary, so that the tobacco component content of the fragrance cigarette containing the full-component tobacco extracts is kept, and continuous and uniform taste is provided for consumers.
Owner:GUANGDONG BRANCH OF CHINA TOBACCO GENERAL
Andrographolidume sustained-release preparation as well as preparation method and use of same
InactiveCN102342964AReduce manufacturing costGuaranteed dosagePharmaceutical delivery mechanismPharmaceutical non-active ingredientsSide effectBioavailability
The invention discloses an adrographolidume sustained-release preparation as well as preparation method and use of the same. Categories and dosages of auxiliary materials are screened to prepare the adrographolidume sustained-release preparation; and the preparation can be released at a uniform releasing velocity by about 30%, 60%, 80% and above 90% in 3h, 6h, 10h and 12h respectively, and can becompletely released within predetermined 12 hours. When the adrographolidume sustained-release preparation is adopted, bioavailability of medicine is improved, toxic side effects of adrographolidume on human body is effectively reduced, and the utilization rate of the medicine is ensured as the medicine of the sustained-release preparation is completely released.
Owner:CHENGDU UNIV OF TRADITIONAL CHINESE MEDICINE
Chloroacetamide herbicide capsule suspension and production method thereof
InactiveCN109717187AEasy to adjust the release rateSmooth responseBiocideAnimal repellantsMicrobiologyChloroacetamide
The invention discloses chloroacetamide herbicide capsule suspension and a production method thereof. The produced hloroacetamide herbicide capsule suspension is stable in quality and controllable inrelease rate, and has quick release efficacy and slow release efficacy at the same time; and the production method is simple to operate, easy to control, high in production efficiency and short in production time.
Owner:JIANGSU ESSENCE AGROCHEM +1
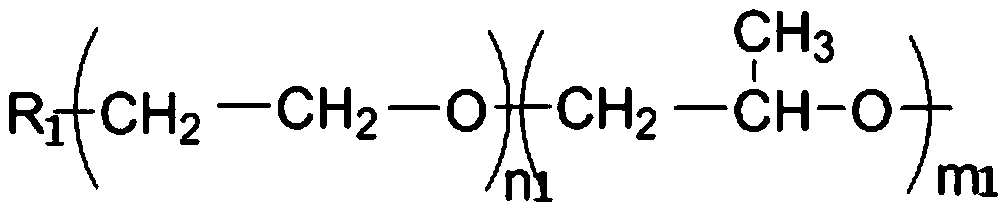
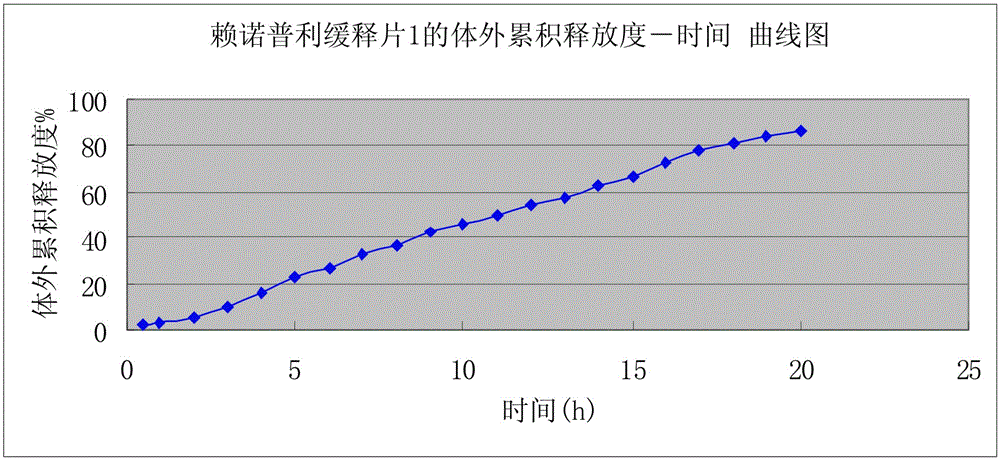
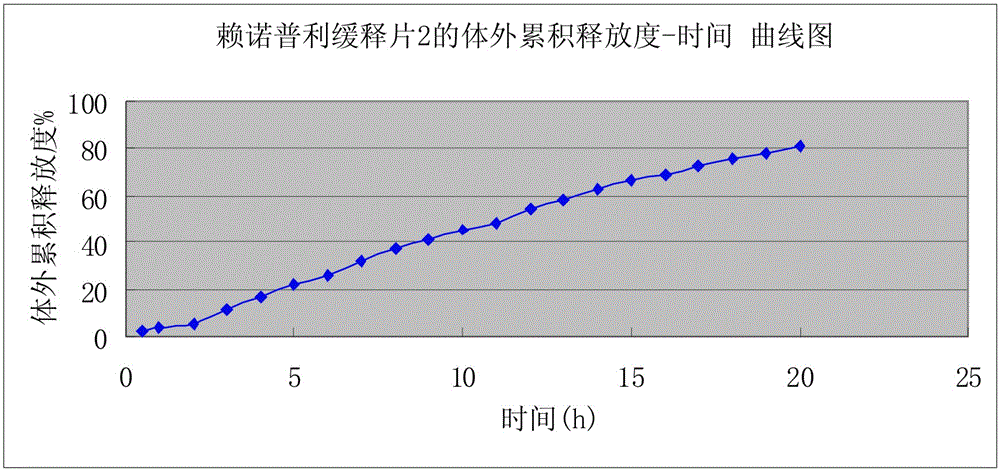
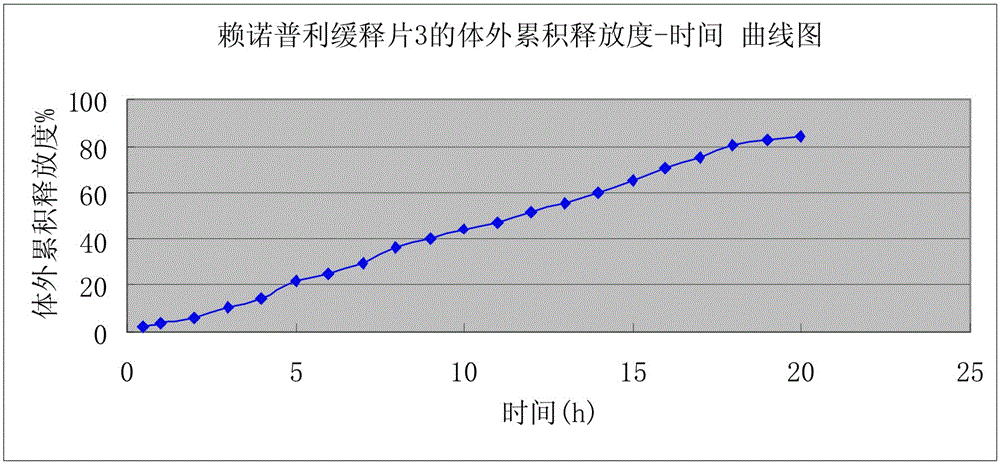
Preparation of lisinopril sustained-release tablets
ActiveCN105055358ASimple processUniform drug release rateDipeptide ingredientsPharmaceutical delivery mechanismMagnesium stearatePharmaceutical formulation
The invention belongs to the technical field of medicine preparations, and discloses preparation of lisinopril sustained-release tablets. The lisinopril sustained-release tablets comprise, by weight, 1%-4% of lisinopril, 54%-66% of matrix materials, 18%-24% of pregelatinized starch, 14.2%-17.6% of microcrystalline cellulose and 0.4%-0.8% of magnesium stearate. The matrix materials are carbomer. The preparation of the lisinopril sustained-release tablets includes smashing the lisinopril according to the weight percentage, filtering the lisinopril via a 100-mesh sieve and then adding the lisinopril into a three-dimensional movable mixer; smashing the carbomer, the pregelatinized starch, the microcrystalline cellulose and the magnesium stearate, filtering the carbomer, the pregelatinized starch, the microcrystalline cellulose and the magnesium stearate via an 80-mesh sieve and then adding the carbomer, the pregelatinized starch, the microcrystalline cellulose and the magnesium stearate into the three-dimensional movable mixer; mixing the lisinopril, the carbomer, the pregelatinized starch, the microcrystalline cellulose and the magnesium stearate with one another by the three-dimensional movable mixer for 30 minutes to obtain mixtures and then directly compressing the mixtures to obtain the lisinopril sustained-release tablets. The preparation has the advantages that the mixtures are directly compressed without coating, so that sustained-release effects can be realized; technologies are simple, and medicine is uniform in release speed.
Owner:LUOYANG JUSN PHARMA
Microcapsule nano silver bactericide preparation method
InactiveCN105284894AUniform release rateImprove the bactericidal effectBiocideDisinfectantsCentrifugationAcetic acid solution
The present invention discloses a microcapsule nano silver bactericide preparation method comprising two steps: 1) an aqueous solution of chitosan is added with acetic acid and dissolved with stirring to obtain a chitosan acetic acid solution; 0.2-0.5wt% AgNO3 solution is slowly added dropwise into the chitosan acetic acid solution, ethanol is added after the dropping addition is complete, the solution is warmed to 50-70 DEG C for constant temperature reaction for 6-10h to obtain a nano silver bactericide solution; 2) a certain amount of the nano silver bactericide solution is taken and mixed with formaldehyde, then added into to n-hexane, stirred evenly, slowly added with urea, first slowly heated to 35-45 DEG C and maintained for 20-40min, then slowly heated to 55-65 DEG C, and adjusted to pH of 3.0 with an acidic solution, and after the reaction, a dry powdery microcapsule nano silver bactericide is obtained by centrifugation and drying. The microcapsule nano silver bactericide can be efficient in sterilization and high in stability, can be slowly released, and is less limited by the media and environmental.
Owner:CNOOC TIANJIN CHEM RES & DESIGN INST +1
Oral mucosa medicament slow release preparation for treating oral periodontal disease and preparation thereof
InactiveCN101653431BLong release timeUniform release rateTetracycline active ingredientsDigestive systemDiseaseSolubility
The invention relates to an oral mucosa medicament slow release preparation for treating oral periodontal disease, which comprises an oral mucosa contact layer and an unbonded close layer, wherein the oral mucosa contact layer comprises the components of anti-biotic medicines and accessories in weight proportion of 2-20:80-98; and the unbonded close layer comprises an unbonded low water-solubility high molecular polymer. The preparation comprises the following steps: preparing a corresponding contact layer paster and water solution through uniformly mixing the anti-biotic medicine and the accessories; coating water solution of the unbonded low water-solubility high molecular polymer on a single surface of the contact layer paster; and drying to obtain the oral mucosa medicament slow release preparation. The oral mucosa medicament slow release preparation has convenient administration, long medicament release, uniform release speed, strong target property, strong bonding property with membrane, and the like; meanwhile, the preparation method has simple operation, low cost and environment protection, and is suitable to industrialized production.
Owner:ZHEJIANG HISUN PHARMA CO LTD
Finasteride sustained release tablets and preparation method thereof
ActiveCN108379236AUniform concentrationFast disintegrationOrganic active ingredientsUrinary disorderAdhesiveMale pattern alopecia
The invention discloses finasteride sustained release tablets. The finasteride sustained release tablets are prepared from the following raw materials: 100 to 150 mg of phospholipid, 40 to 70 mg of glyceride, 50 to 60 mg of ethanol, 2 to 5 mg of finasteride, 10 to 50 mg of micropowder silica gel, 20 to 60 mg of chitosan, 20 to 60 mg of an adhesive, 2 to 10 mg of low-substituted hydroxypropyl cellulose, 0.5 to 5 mg of a lubricant and 0.2 to 2 mg of a sweetening agent. The preparation method comprises the following steps: mixing the phospholipid, the glyceride and the finasteride; adding the ethanol and mixing uniformly; adding the micropowder silica gel, the chitosan and the adhesive and mixing uniformly; performing wet granulation; drying; adding the low-substituted hydroxypropyl cellulose, the lubricant and the sweetening agent and mixing uniformly; and tabletting. The finasteride sustained release tablets provided by the invention have a remarkable sustained-release effect, are uniform in release speed and can reduce the use times; the tablets have high uniform strength and good taste; and the tablets can be used for treating male pattern alopecia and benign prostatic hyperplasia.
Owner:武汉百纳礼康生物制药有限公司
Mouth mucosa sustained-release preparation for treating oral and periodontal diseases by chlorhexidine
InactiveCN103110610ALong release timeUniform release rateTetracycline active ingredientsAntisepticsDiseasePolyvinyl alcohol
The invention relates to fields of a drug for treating a disease of cari oris mucosa, and preparation thereof, and particularly relates to a mouth mucosa sustained-release preparation for treating oral and periodontal diseases by chlorhexidine. The preparation is composed of a mouth mucosa contact layer and a non-adhesive seal layer; the mouth mucosa contact layer comprises the components of an antibiotic drug and an auxiliary material, of which the weight ratio is (2-20) to (80-98); the non-adhesive seal layer comprises the components of a non-adhesive low-water soluble high-molecular polymer; the non-adhesive low-water soluble high-molecular polymer is selected from polyving akohol; the mouth mucosa sustained-release preparation comprises the components of 4.0g of chlorhexidine, 12.0g of hydroxyethyl cellulose, 5.5g of modified starch, 7.0g of microcrystalline cellulose, 0.5g of grape essence and the balance of polyving akohol. The mouth mucosa sustained-release preparation has the advantages of being convenient to drug delivery, long in drug release time, even in drug release speed, strong in target, strong in mucosa adhesive force and the like; and the preparation method is simple to operate, low in cost, friendly to environment, and suitable for industrial production.
Owner:上海微丸医药开发有限公司
Mouth mucosa drug sustained release preparation for treating mouth periodontal disease by doxycycline
InactiveCN103127034AUniform release rateBroaden applicationTetracycline active ingredientsAntisepticsDiseaseSolubility
The invention relates to drugs for treating mouth mucosa diseases and the manufacture field of the drugs for treating mouth mucosa diseases, in particular to a mouth mucosa drug sustained release preparation for treating a mouth periodontal disease by doxycycline. The preparation is composed of a mouth mucosa contacted layer and a non-adhesiveness sealing layer. The mouth mucosa contacted layer comprises, by weight, 2-20 / 80-98 of antibiotic drugs and auxiliary materials. The components of the non-adhesiveness sealing layer are non-adhesiveness low water-solubility high-molecular polymers. The non-adhesiveness low water-solubility high-molecular polymers are chosen from polyvinyl alcohol. The components of mouth mucosa drug sustained release preparation comprise 2.0g of methyl vinyl ether-maleic anhydride copolymers, 2.0g of polyoxyethylene, 2.0g of carboxymethyl cellulose sodium, 12.0g of natural starch, 2.0g of menthol and 0.5g of doxycycline, and the rest is poval. The mouth mucosa drug sustained release preparation has the advantages that drug delivering is convenient, drug releasing time is long, drug releasing speed is even, targeting is strong, and adhesive force with mucosas is strong. In addition, the manufacturing method is simple in operation, low in cost, environment-friendly and suitable for industrial production.
Owner:上海微丸医药开发有限公司
Biological fertilizer and preparation method thereof
InactiveCN112094153AEnhanced Nutrient LevelsReduce the effect of agglomerationOrganic fertilisersSolid/semi-solid fertilisersFertilizerNutrient
The invention relates to the technical field of biological fertilizers, and especially relates to a biological fertilizer and a preparation method thereof. An arc-shaped disc used for bearing the biological fertilizer comprises a carbon cover barrel, a carbon column barrel and an arc-shaped frame. Due to the fact that the added biological fertilizer is prone to loss to all positions in soil blocksunder the rainwater condition, the content of nutrients absorbed by crop root areas is reduced, nutrient supply in the crop growth process is affected, and then the use effect of the biological fertilizer is weakened. Therefore, the storage state of the biological fertilizer under different conditions of the natural environment is protected through the carbon structure of the arc-shaped disc, itis ensured that the biological fertilizer acts on a crop root system area in a land parcel, the release rate of nutrients in the biological fertilizer is balanced through gradual conversion of biological carbon nutrient substances by microbial florae in the crop growth process, the yield of the biological fertilizer is increased. and the time for finally decomposing the carbon structure of the mechanism is controlled to completely decompose the carbon structure in the biological fertilizer, so that the application effect of the biological fertilizer is improved.
Owner:何莉
Flocculating agent and preparation method thereof
InactiveCN109516535AEasy to operateGood reproducibilityWater/sewage treatment by flocculation/precipitationMaterials sciencePolymer
The invention provides a flocculating agent. The flocculating agent contains a graphene quantum dot and a polymer linked with the graphene quantum dot, wherein the graphene quantum dot is a graphene oxide quantum dot and contains a first active functional group, the polymer contains a second active functional group, and the graphene quantum dot and the polymer are linked through the condensation of the first active functional group and the second active functional group.
Owner:TCL CORPORATION
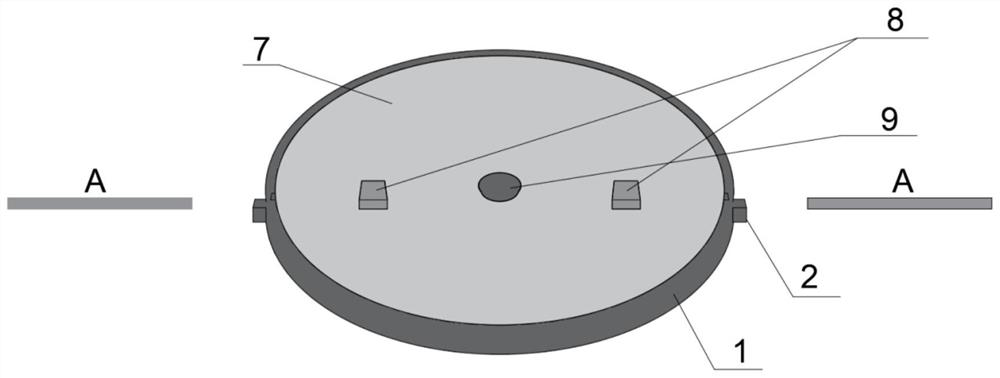
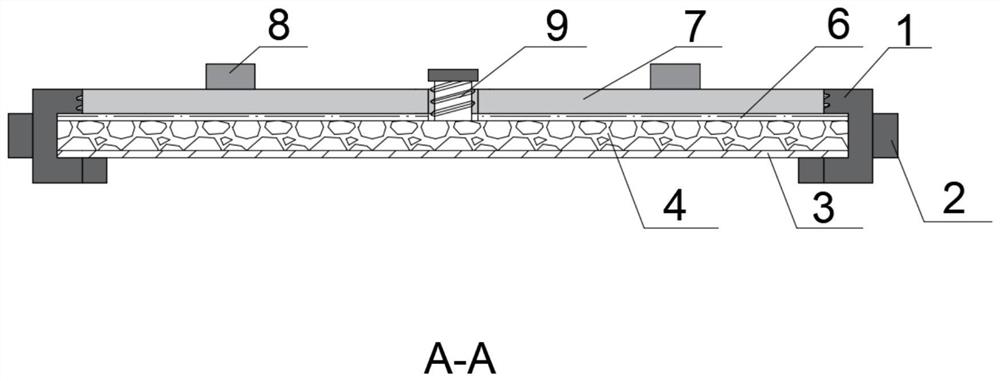
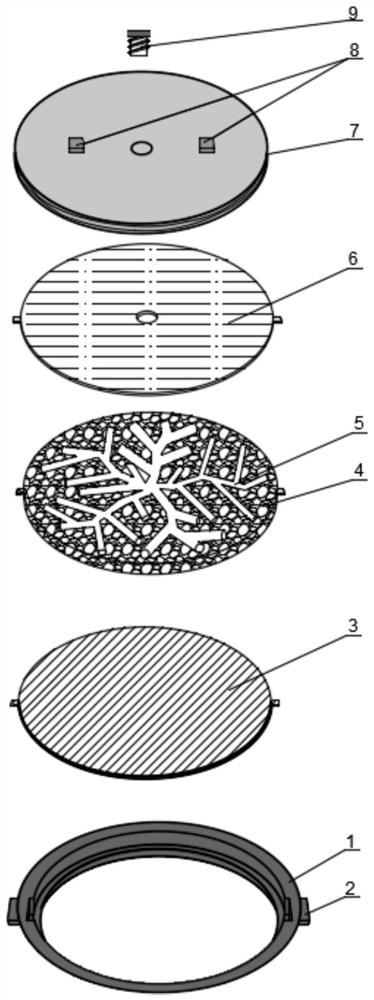
Standard plate for detecting pollution release of decoration material as well as preparation and application of standard plate
PendingCN114813277AUniform release rateStable release ratePreparing sample for investigationDiffusion analysisPollutantEnvironmental chemistry
The invention discloses a standard plate for detecting pollution release of a decoration material as well as preparation and application of the standard plate. The standard plate comprises a sealing outer sleeve, a wood plate assembly, a sealing upper cover and a sealing plug, a hole is formed in the bottom surface of the sealing outer sleeve, and the wood plate assembly is composed of a bottom plate, a core plate and a top plate, nested in the sealing outer sleeve and pressed and fixed by the sealing upper cover; the core plate contains a porous material, and radial channels are formed in the core plate; through holes are formed in the centers of the top plate and the sealing upper cover; a sealing plug is inserted into the through holes for sealing. When the standard plate is used, target pollutants are dropped into the through holes, and the target pollutants are diffused and dispersed into the porous material and other interstitial substances in the core plate through the channels by centrifugation and oscillation, so that the standard plate capable of uniformly and stably releasing the target pollutants is obtained. By changing the types and concentrations of the added target pollutants, the pollution release conditions of different product control materials can be detected.
Owner:雄安绿研检验认证有限公司
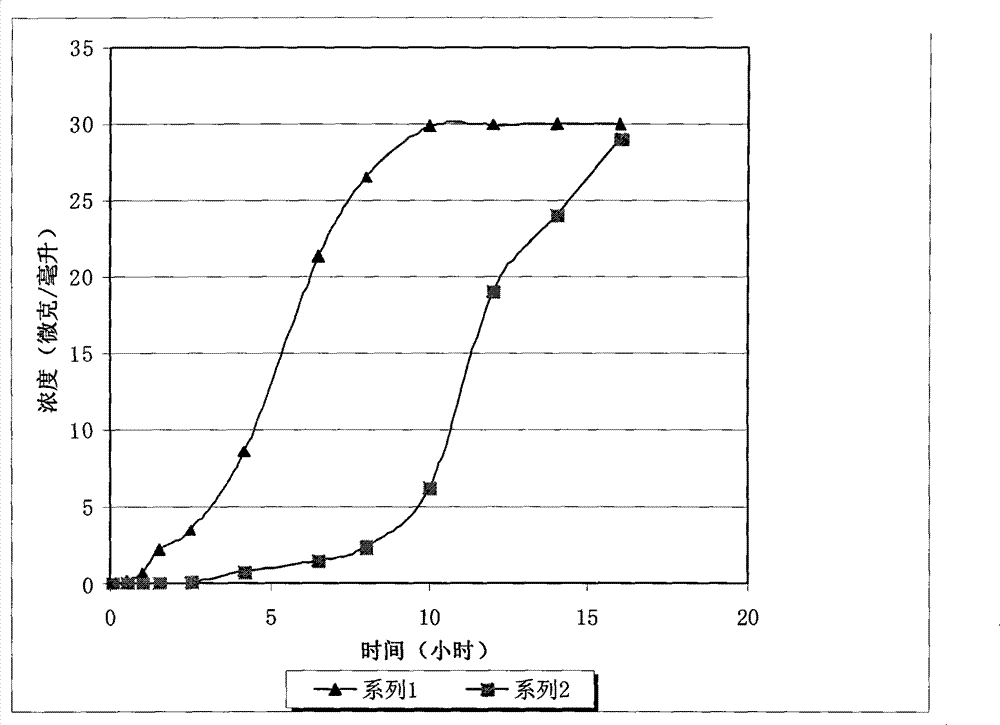
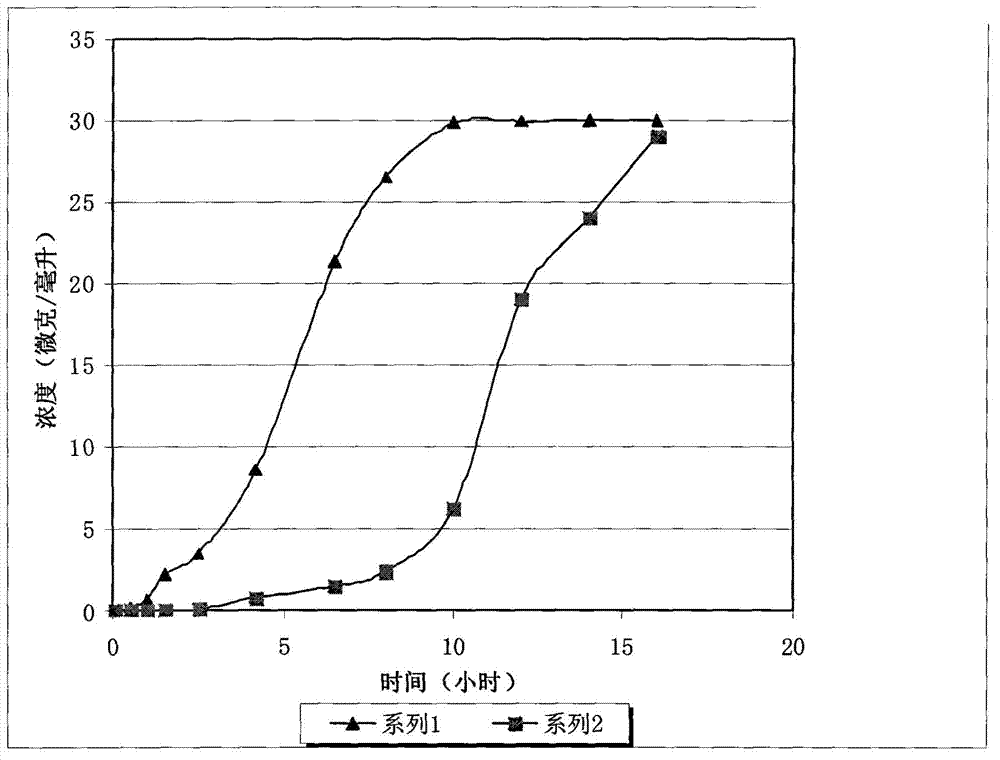
A kind of oxytocin embedding method based on cuttlefish octopus
ActiveCN107466912BLess irritatingLong duration of anesthesiaPeptide/protein ingredientsClimate change adaptationSquidBiocompatibility
The invention discloses an embedding method for oxytocin based on squid cuttlebone. Firstly, placing squids into a narcotic, and performing anaesthetization; and then embedding an oxytocin low-release agent into the cuttlebone. The method has the beneficial effects that the embedded oxytocin is located in calcareous cuttlebone of the squids, thus the embedded oxytocin has small stimulation effects on the squids, the cuttlebone has a loose and porous structure, and therefore the oxytocin can be easily released into bodies to play roles; and a certain amount of L-medetomidine is added to R-medetomidine, thus the prepared narcotic has long anaesthetization hold time and a certain analgesic effect, the stress reaction of the squids to an embedding process is greatly reduced, and ink spraying is avoided; and the GnRH low-release agent prepared by using chitosan quaternary ammonium salt as a carrier has good biocompatibility, long release time and a uniform release speed.
Owner:ZHEJIANG OCEAN UNIV
Mouth mucosa drug sustained release preparation for treating mouth periodontal disease by doxycycline
InactiveCN103127034BLong release timeUniform release rateTetracycline active ingredientsAntisepticsSolubilityDisease
The invention relates to drugs for treating mouth mucosa diseases and the manufacture field of the drugs for treating mouth mucosa diseases, in particular to a mouth mucosa drug sustained release preparation for treating a mouth periodontal disease by doxycycline. The preparation is composed of a mouth mucosa contacted layer and a non-adhesiveness sealing layer. The mouth mucosa contacted layer comprises, by weight, 2-20 / 80-98 of antibiotic drugs and auxiliary materials. The components of the non-adhesiveness sealing layer are non-adhesiveness low water-solubility high-molecular polymers. The non-adhesiveness low water-solubility high-molecular polymers are chosen from polyvinyl alcohol. The components of mouth mucosa drug sustained release preparation comprise 2.0g of methyl vinyl ether-maleic anhydride copolymers, 2.0g of polyoxyethylene, 2.0g of carboxymethyl cellulose sodium, 12.0g of natural starch, 2.0g of menthol and 0.5g of doxycycline, and the rest is poval. The mouth mucosa drug sustained release preparation has the advantages that drug delivering is convenient, drug releasing time is long, drug releasing speed is even, targeting is strong, and adhesive force with mucosas is strong. In addition, the manufacturing method is simple in operation, low in cost, environment-friendly and suitable for industrial production.
Owner:上海微丸医药开发有限公司
Filter Cap Additive Delivery System
InactiveUS20090101561A1Uniform release rateMaintain physical integrityAdditivesFiltration separationEngineeringDelivery system
A additive delivery system and method which includes a additive gel, a filter and a fluid / gel chemistry exchange region, wherein the exchange region subjects the additive gel to different types or combinations of contact with the fluid in order to control the additive release rate, maintain a uniform release rate, maintain the physical integrity of the gel or combinations thereof.
Owner:THE LUBRIZOL CORP
A kind of tobacco extract particle and gum base type mouth cigarette and its preparation method
InactiveCN105124747BMaintenance contentConsistent and even tasteTobacco preparationTobacco treatmentFlavorAntioxidant
The invention belongs to the field of tobacco products, and discloses a gum-based mouth cigarette and a preparation method thereof. A gum-based mouth cigarette, which is composed of the following substances in mass percentage: chewing gum gum base 30%-45%; tobacco extract 5%-15%; tobacco extract particles 10%-20%; sweetener 5.5%- 15%; emulsifier 0.5% to 5%; spice 0% to 10%; antioxidant 0.1% to 8%; the tobacco extract particles include at least two substrate layers with different tobacco extract contents; the substrate The material layer is coated layer by layer, the surface of the base material layer is coated with a coating layer, and the base material layer contains excipients; the content of the tobacco extract particles in the base material layer gradually decreases from the inside to the outside. Small. The tobacco ingredients in gum-based mouth cigarettes are provided by both tobacco extract and tobacco extract particles, and the release process of the two complements each other, maintaining the content of tobacco ingredients in gum-based mouth cigarettes, providing consumers with continuous and Uniform taste.
Owner:GUANGDONG BRANCH OF CHINA TOBACCO GENERAL
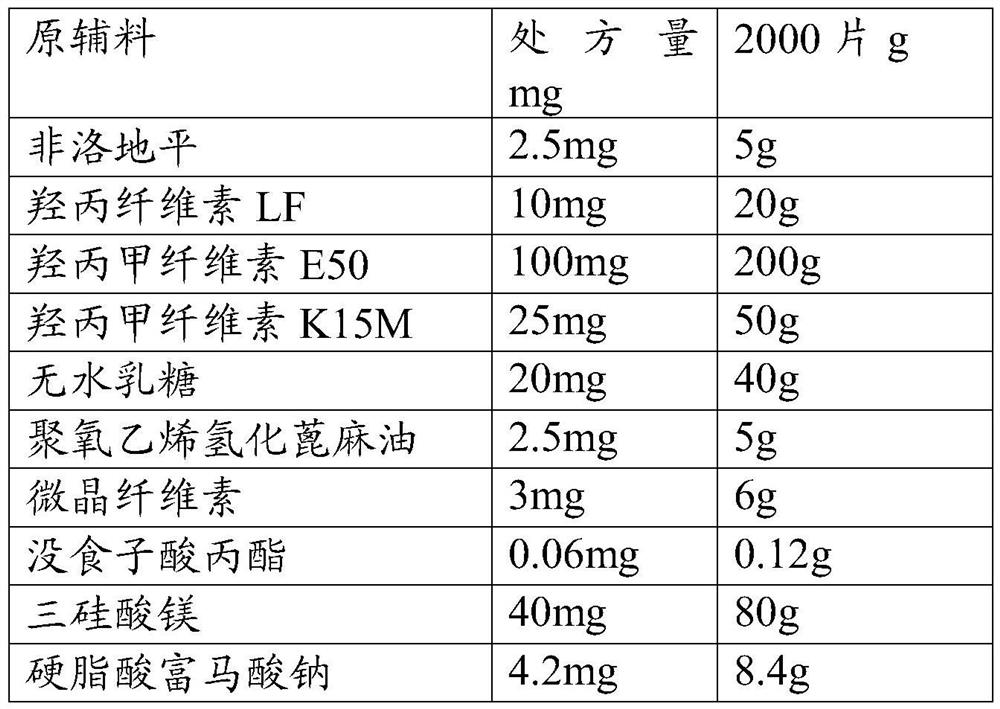
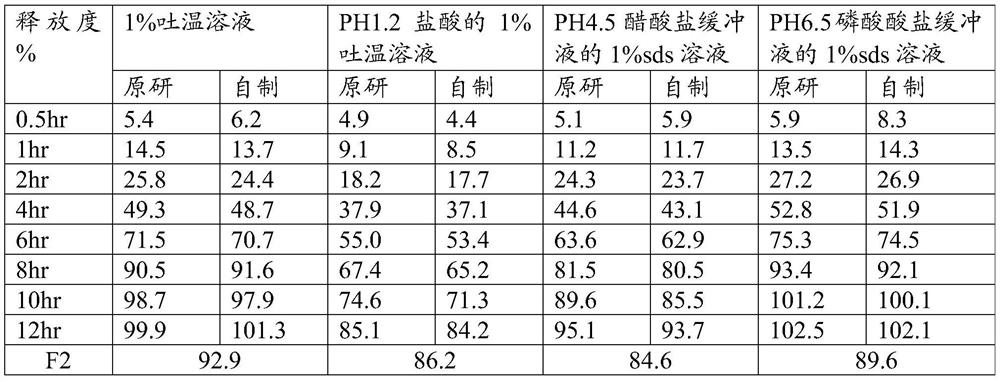
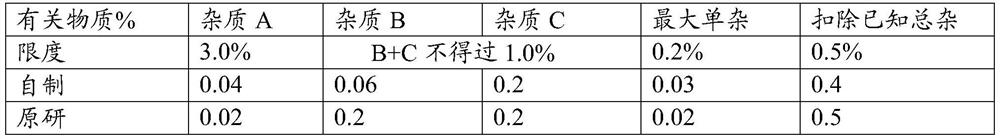
A kind of felodipine sustained-release tablet and preparation method thereof
ActiveCN109200026BEnsure safetyUniform release rateOrganic active ingredientsPill deliveryProlonged-release tabletAnoxomer
Owner:BEIJING SIHUAN KEBAO PHARM CO LTD