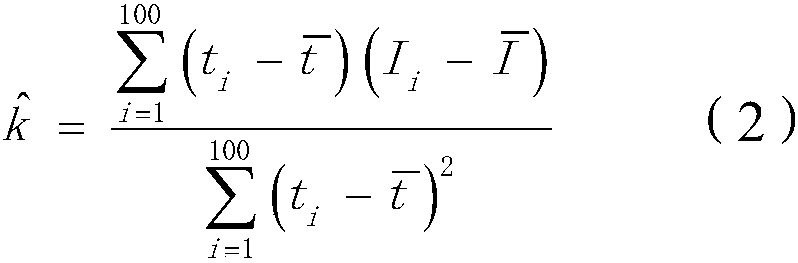Patents
Literature
51results about How to "Improve electrolysis current efficiency" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
Electric flocculation method and device for recovering and processing desulfurized wastewater
ActiveCN101817575AEliminate passivationImprove electrolysis current efficiencyWater/sewage treatmentWaste water treatment from gaseous effluentsPollutantDewatered sludge
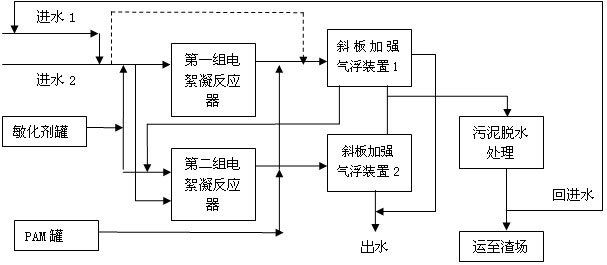
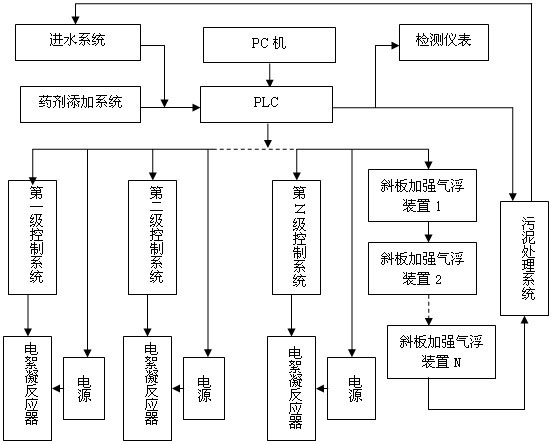
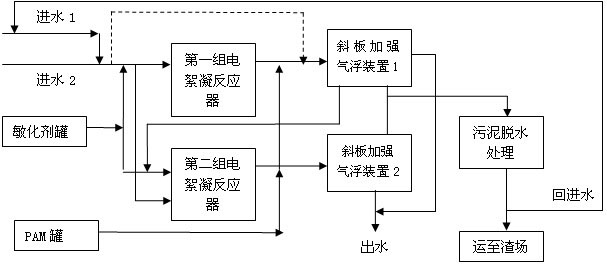
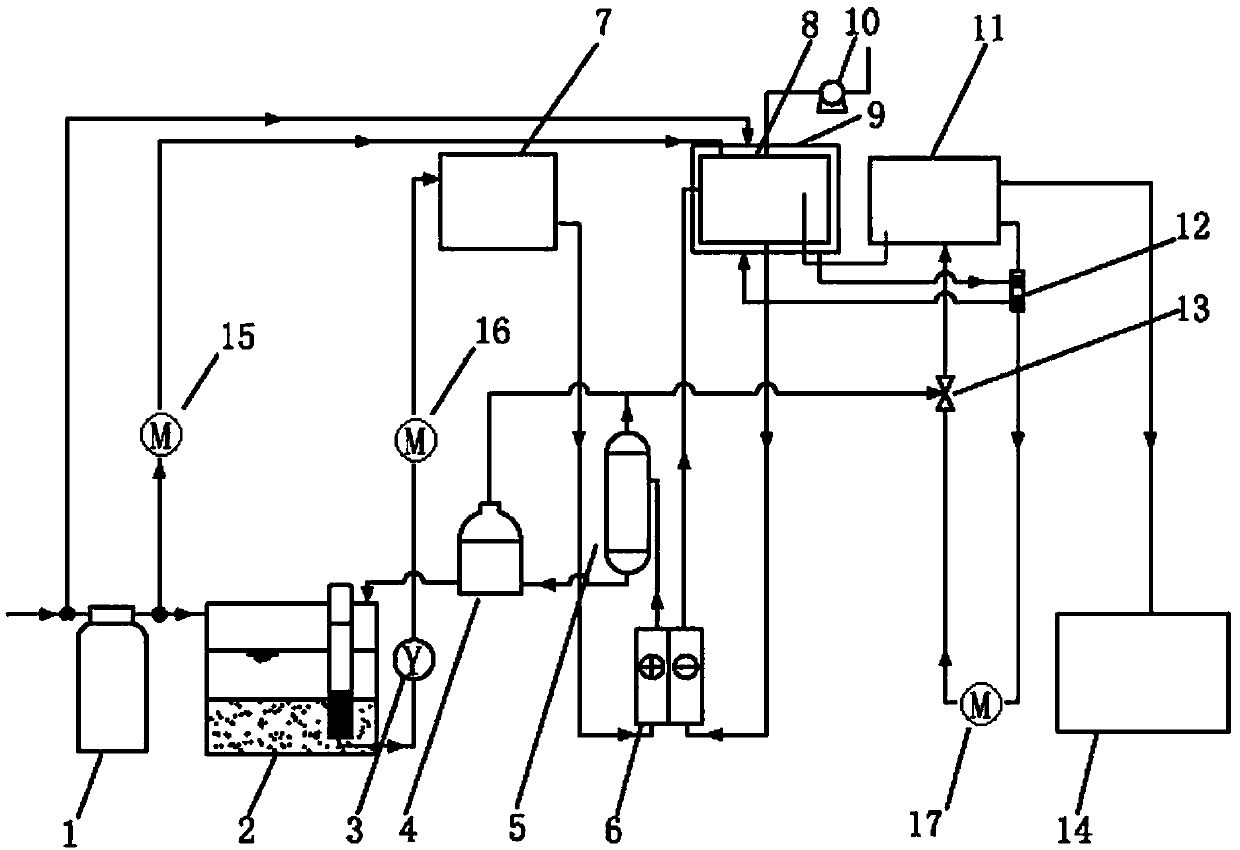
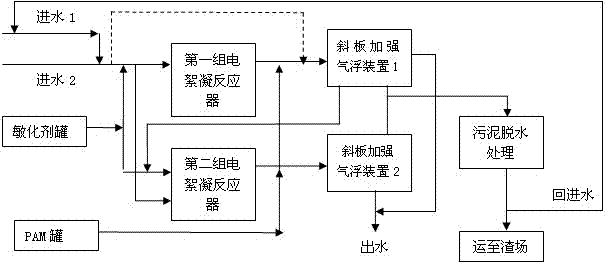
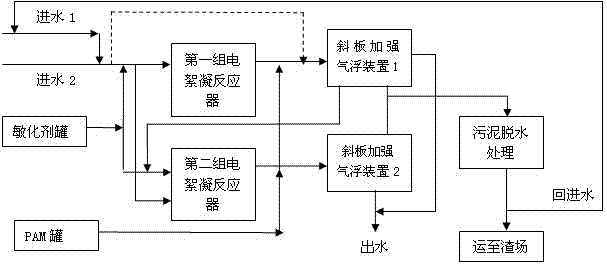
The invention discloses electric flocculation method and device for recovering and processing desulfurized wastewater. The electric flocculation method for recovering and processing desulfurized wastewater comprises the following steps of: collecting desulfurized wastewater to enter an electric flocculation reactor, then, carrying out solid-liquid separation, recovering the separated clear effluent water in a clinker-flushing system of an electric power plant or directly discharging the separated clear effluent water, conveying the sludge to dewater in a mud-processing system, and returning the effluent water of the mud-processing system to a water intake system. Compared with the prior art, the electric flocculation technology is adopted to substitute a complicated chemical method for recovering and processing the desulfurized wastewater, can effectively remove pollutants in the wastewater, reaches a heavy metal processing rate up to 95-99 percent, a chlorine-processing rate over 80 percent and removal rates of ammonian, and the like over 90 percent, greatly shortens the processing time, improves the processing efficiency and saves the investment cost. Moreover, the electric flocculation processing device has small size, extremely small floor occupation, good environmental-protection performance and low cost without additional flocculating agents.
Owner:华电水务工程有限公司
Method for electroplating titanium on surface of metal
The invention relates to a method for electroplating titanium on the surface of metal. The method is characterized in that TiOxCy is taken as a positive pole, metal to be electroplated is taken as a negative pole and molten salt is taken as an electrolyte in the titanium electroplating process. By the method provided by the invention, the aim of corrosion resistance can be achieved through electroplating titanium on the surface of metal with the melting point of 800 DEG C, such as carbon steel, stainless steel, foamy copper and foamy nickel, and other matrixes, the use cost of a titanium material can be greatly reduced, the application field of the titanium metal is widened, and the titanium is not only taken as precious space metal. The electro-deposition on general metal can be realized with least reaction steps and cheapest raw materials; the titanium electroplating is directly finished from the step of electrolyzing the raw materials of titanium, so that the step for preparing the titanium through electrolysis is omitted, the loss of the raw materials and energy sources is reduced, the efficiency of electrolytic current and the utilization ratio of the raw materials are improved, and the titanium electroplating cost is reduced.
Owner:GUIZHOU BRANCH CHINA ALUMINUM IND
Device used for producing water supply javel water
PendingCN107604377AImprove electrolysis current efficiencyEvenly distributedCellsOrganic diaphragmsGratingTotal dissolved solids
The invention provides a device used for producing water supply javel water. The device comprises a water softener, the water softener is connected with a saline preparation box, the saline preparation box is connected with a high-position saline box, the high-position saline box is connected with an ionic exchange membrane cell anode chamber with a flow guide weir, and an ionic exchange membranecell cathode chamber is connected with a high-position alkali liquor box; the anode chamber generates light salt brine and chlorine, and the cathode chamber generates hydrogen and sodium hydroxide; the anode chamber is connected with a gas-liquid separator, the gas-liquid separator is connected with a light salt brine circulation tank with a grating plate, and the light salt brine circulation tankis connected with the saline preparation box; the high-position alkali liquor box, the gas-liquid separator and the light salt brine circulation tank are connected with a chlorine negative pressure absorbing and sodium hypochlorite circular reaction system, and a sodium hypochlorite solution is generated by the chlorine negative pressure absorbing and sodium hypochlorite circular reaction system.After the javel water produced through the device is added into water, changes of indexes such as the pH value and the total dissolved solid of the water are small, generated disinfection by-productsare fewer, meanwhile, the running cost is low, and maintenance and management are convenient.
Owner:CHINA INST OF WATER RESOURCES & HYDROPOWER RES
Method for preparing TiB2 cathode coating for aluminium electrolysis
The invention provides a method for preparing a TiB2 cathode coating for aluminium electrolysis. The method comprises the following steps: grinding such materials as TiB2, a carbon-based material, an additive and the like, then mixing the ground materials proportionally and further mixing the ground materials uniformly in mixing and kneading equipment to obtain a solid powder mixture; adding a diluent into an adhesive, continuously stirring the two materials until the two materials are fully mixed uniformly; and pouring the uniformly mixed mixture of the diluent and the adhesive into the mixing and kneading equipment, nixing the mixed mixture of the diluent and the adhesive with the solid powder mixture and stirring the materials until the materials are mixed uniformly, thus obtaining the TiB2 composite required for aluminium electrolysis. The method has the following beneficial effects: after the product produced by the method is used in an aluminium electrolytic cell, the service life of the aluminium electrolytic cell is greatly prolonged and simultaneously the current efficiency of an aluminium electrolysis process is greatly improved, thus lowering the production cost of aluminium per ton; and the method has the advantages of short process flow, low production treatment cost and low environmental pollution.
Owner:KUNMING METALLURGY INST
Method for preparing battery grade lithium hydroxide monohydrate
InactiveCN110422863ARich sourcesWith resource recyclingLithium oxides/hydroxidesElectrolysisLithium carbonate
The invention provides a method for preparing battery grade lithium hydroxide monohydrate. The method utilizes crude lithium carbonate as a raw material, and includes the following steps: acid dissolution, preliminary impurity removal, deep impurity removal, evaporation concentration, preparation of lithium hydroxide by ion-exchange membrane electrolysis, evaporation crystallization, and recrystallization. The method is rich in source of raw material and has the advantages of recycling and utilizing resources, and being less in type of contained impurities and easy to remove; chlorine gas polluting the environment is not produced and product sulfuric acid can be reused to dissolve, and consumption of raw materials is reduced; and the pH of an anolyte is controlled by controlling a circulation anolyte to increase the efficiency of current.
Owner:JIANGXI RUIDA NEW ENERGY TECH CO LTD
Aluminum electrolysis method with aluminum as cathode
The invention discloses an aluminum electrolysis method with aluminum as a cathode, and relates to the improvement on a method for producing electrolysis aluminum through a molten-salt method. The aluminum electrolysis method is characterized in that in the aluminum electrolysis process, molten aluminum at the bottom of an electrolysis cell is adopted as the cathode for conducting aluminum electrolysis; and the molten aluminum at the bottom of the electrolysis cell makes contact with a cathode busbar to form the aluminum electrolysis cathode. According to the aluminum electrolysis method with the aluminum as the cathode, a conventional anode system, a discharging system and a dust purifying system can be adopted in the aluminum electrolysis cell. According to the method, the molten aluminum in which no cathode carbon block, no steel bar and no titanium boride coating exist is adopted as the cathode to replace a cathode composed of steel bars and a carbon block set. The defects that according to a traditional aluminum electrolysis cell, aluminum oxide deposit exists on cathode carbon blocks and cellular micro-batteries exist are eliminated. In the electrolysis production process, the problem that aluminum oxide and aluminum carbide which are deposited and generated at the furnace bottom pollute electrolysis molten aluminum does not exist.
Owner:XINJIANG UNIVERSITY
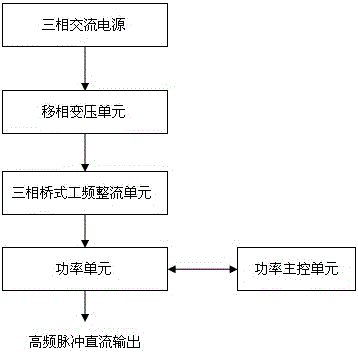
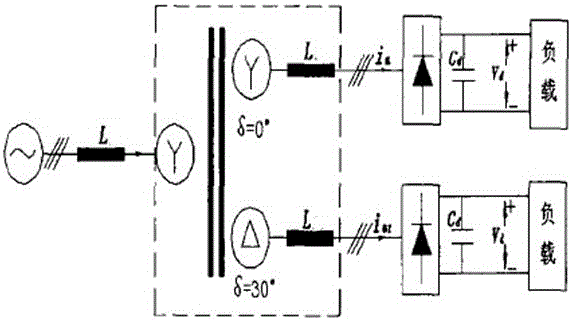
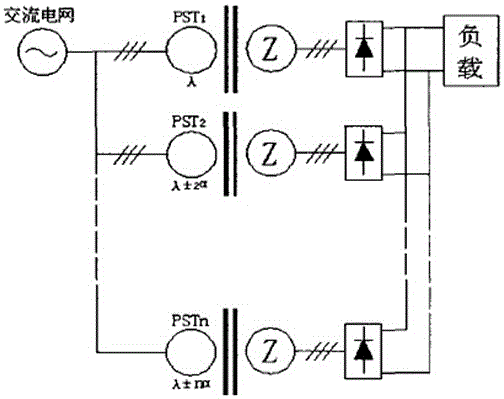
Industrial energy-saving high-frequency impulse electrolytic manganese device
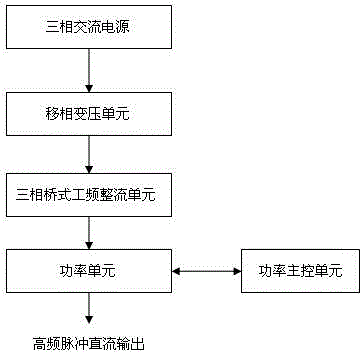
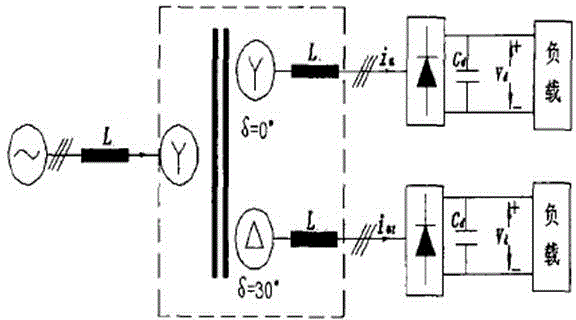
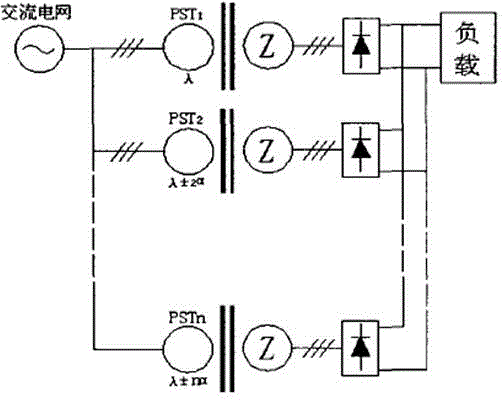
ActiveCN104831314AImprove conversion efficiencyReduce harmonic effectsPhotography auxillary processesAlternating currentPower unit
The invention discloses an industrial energy-saving high-frequency impulse electrolytic manganese device which comprises one or more phase-shifting transformers, one or more three-phase rectifier bridge assemblies, one or more power assemblies and a power master control unit, wherein the primary sides of the phase-shifting transformers are connected with the output end of a three-phase alternating-current supply and the secondary sides of the phase-shifting transformers are in one-to-one corresponding connection with the inputs of the three-phase rectifier bridge assemblies, respectively; the output ends of the three-phase rectifier bridge assemblies are correspondingly connected with the input ends of the power assemblies, and the output ends of the power units output high frequency impulse signals to electrolytic baths; the voltages output by the secondary sides of phase-shifting transforming units are determined according to the quantity of the electrolytic baths connected in series; when a plurality of the phase-shifting transformers are adopted, the phase-shifting transforming units are the phase-shifting transformers in multiple forms for reducing harmonic waves of an alternating current input current and harmonic amplitudes and current impulses of a direct current output voltage; the power master control unit is respectively connected with one or more power assemblies for current-sharing control of the power assemblies.
Owner:CHENGDU XINMING ENERGY SAVING TECH CO LTD
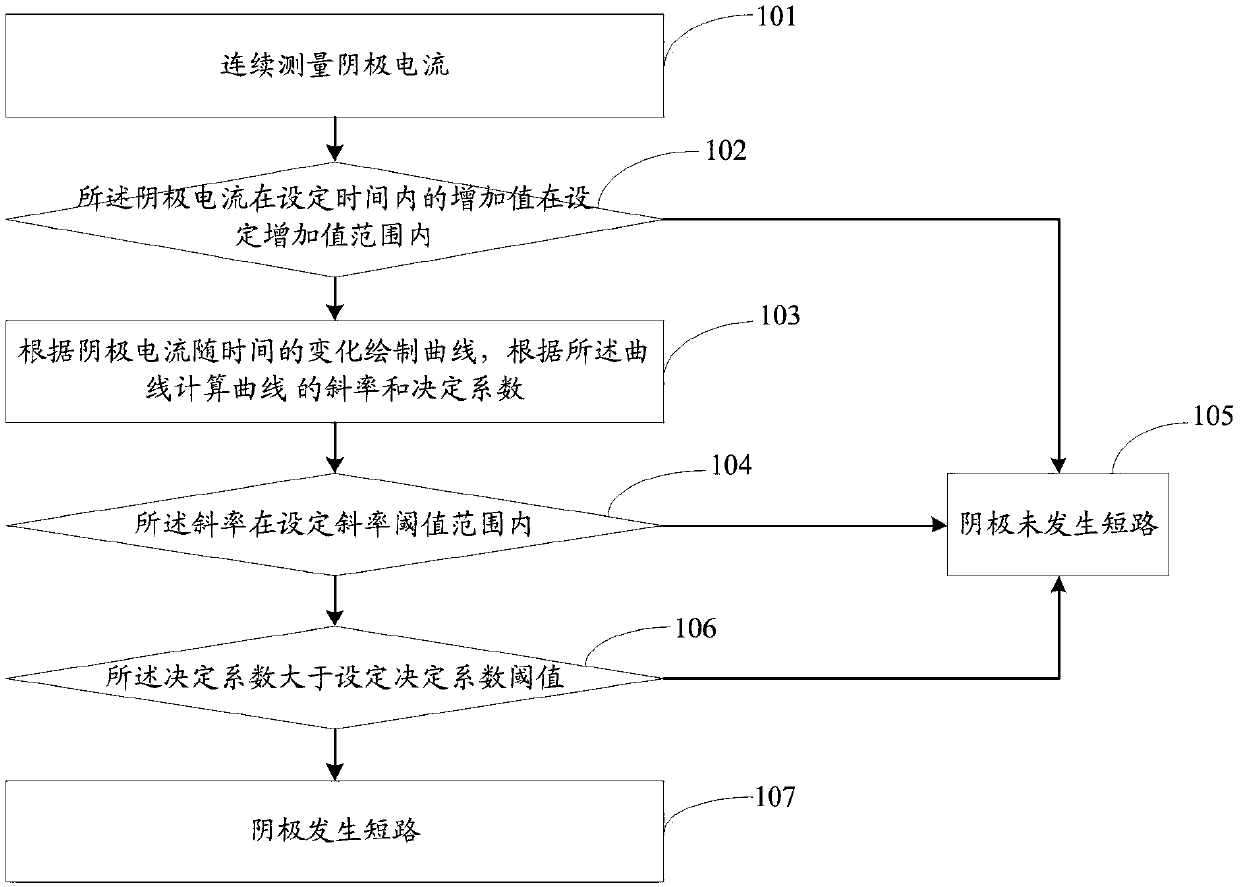
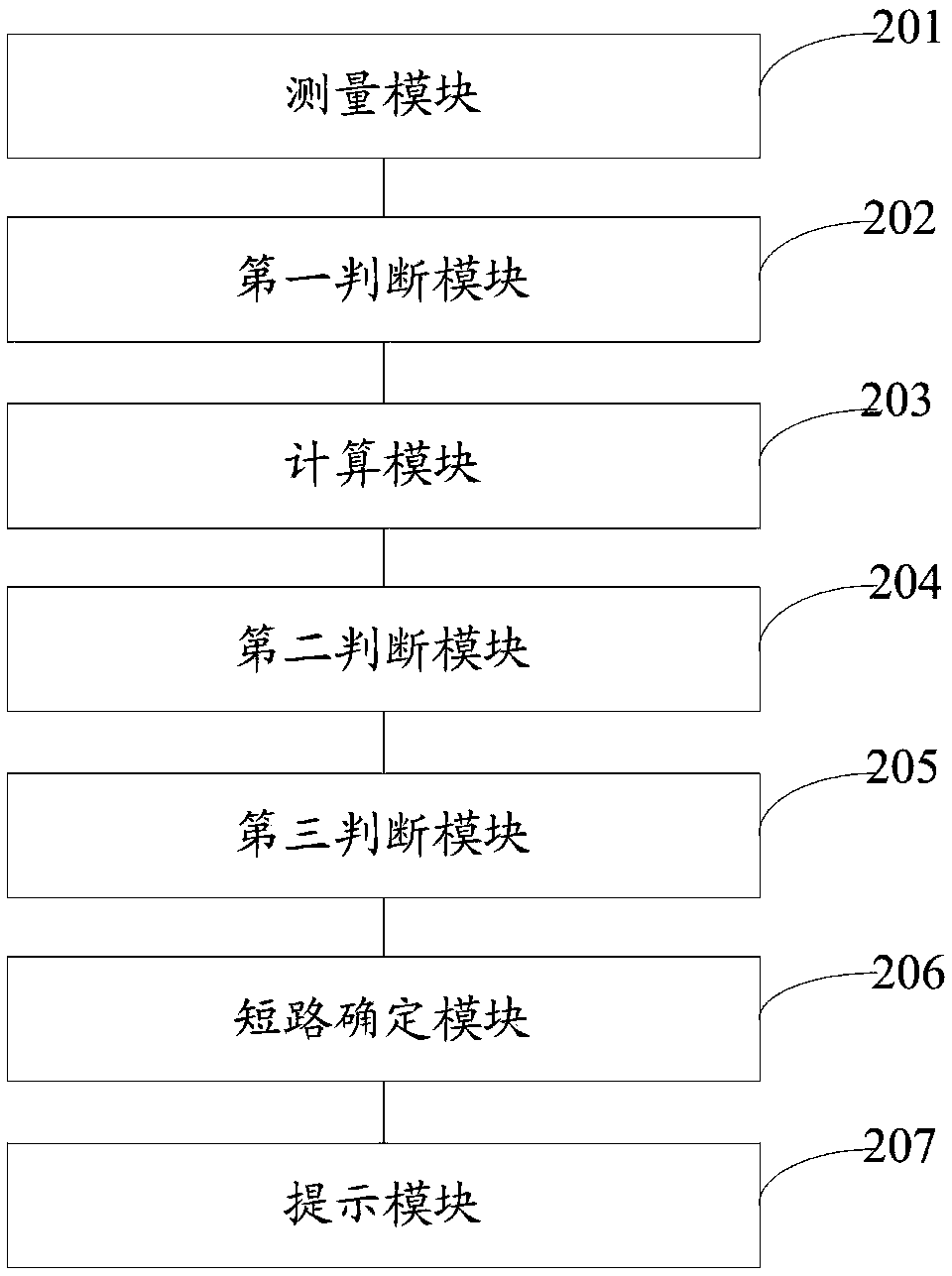
Method and system for rapidly detecting cathode short circuit
InactiveCN108896856AAvoid lostImprove electrolysis current efficiencyElectrical testingPower flowLinear model
The invention discloses a method and system for rapidly detecting a cathode short circuit. The method comprises the following steps: continuously measuring cathode current; judging whether an added value of the cathode current in pre-set time is in a pre-set added value range or not; if so, establishing a linear model according to changes of the cathode current along time and calculating a slope and a determination coefficient according to the linear model; if not, judging that the cathode is not short-circuited; judging whether the slope is in a pre-set slope threshold value range; if so, judging whether the determination coefficient is greater than a pre-set determination coefficient threshold value or not; if not, judging that the cathode is not short-circuited; if the determination coefficient is greater than or equal to the pre-set determination coefficient threshold value, judging that the cathode is short-circuited; if the determination coefficient is less than the pre-set determination coefficient threshold value, judging that the cathode is not short-circuited. By adopting the method and the system, disclosed by the invention, lasting short-circuit current loss can be avoided and the electrolytic current efficiency is improved.
Owner:NORTH CHINA UNIVERSITY OF TECHNOLOGY
Bipolar electrode plate for preparing metal aluminum by aluminum chloride electrolysis and a method of using same
ActiveCN111270271AReduce bypass currentImprove electrolysis current efficiencyElectrodesAluminium chlorideElectrolysis
The invention relates to a bipolar electrode plate for preparing metal aluminum by aluminum chloride electrolysis and a using method of the bipolar electrode plate, and belongs to the technical fieldof electrolytic aluminum. The bipolar electrode plate for preparing metal aluminum by aluminum chloride electrolysis comprises a bipolar electrode plate base block and an insulating block. The bipolarelectrode plate base block is divided into a cathode end and an anode end. In the bipolar electrode plate base block, the remaining periphery is provided with insulating blocks correspondingly exceptthe end surface of the cathode end and the end surface of the anode end. The width of the insulating block needs to comply with the following relation: (Rho<C>*h+2.15 / D*S)<<Rho<L>*(b+h). According tothe bipolar electrode plate, the insulating block is connected to the bipolar electrode plate base block, and the width of the insulating block is defined, so that when the bipolar electrode plate for preparing the metal aluminum by aluminum chloride electrolysis, bypass current can be effectively reduced, current efficiency is improved, and the electrolytic energy consumption is reduced.
Owner:NORTHEASTERN UNIV
Double-layer aluminum cathode aluminum electrolysis cell
ActiveCN105780056AEliminates contact pressure dropReduce electrolysis power consumptionElectrolysisBusbar
The invention discloses a double-layer aluminum cathode aluminum electrolysis cell, and relates to the improvement on aluminum electrolysis cell production with aluminum as a cathode, with cryolite aluminum monoxide melt as an electrolyte and through a molten salt method. The double-layer aluminum cathode aluminum electrolysis cell structurally comprises a prebaked anode, a discharging device and a dust purifying device. The double-layer aluminum cathode aluminum electrolysis cell is characterized in that the structure of a cathode system of the double-layer aluminum cathode aluminum electrolysis cell comprises an electrolysis cell body, two cathode partition walls, a cathode busbar and a cathode horizontal partition plate, wherein the two cathode partition walls are arranged in cell ledges at the two ends of the aluminum electrolysis cell body and fixedly connected with the inner walls of the side cell ledges of the electrolysis cell body, the cathode busbar enters the electrolysis cell body from side channels formed by the cathode partition walls and the inner walls of the cell ledges at the ends of the aluminum electrolysis cell body, and the cathode horizontal partition plate is located in the electrolysis cell body and provided with molten aluminum through holes. According to the double-layer aluminum cathode aluminum electrolysis cell, molten aluminum in which no cathode carbon block, no steel bar and no titanium boride coating exist is adopted as the cathode to replace a cathode composed of steel bars and a carbon block set. The defects that according to a traditional aluminum electrolysis cell, aluminum oxide deposit exists on cathode carbon blocks and cellular micro-batteries exist are eliminated. In the electrolysis production process, the problem that aluminum oxide and aluminum carbide which are deposited and generated at the furnace bottom pollute electrolysis molten aluminum does not exist.
Owner:XINJIANG UNIVERSITY
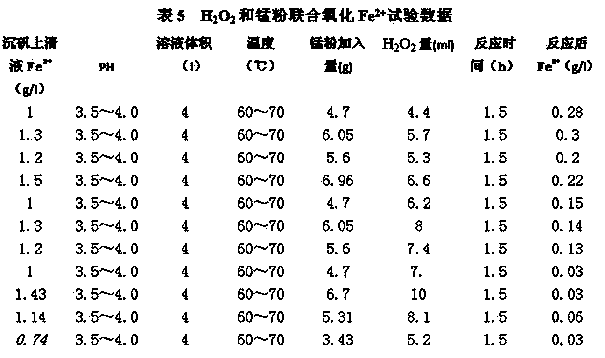
Vitriol precipitating and Fe removing process in zinc hydrometallurgy technology
ActiveCN103773957AReduce the content of manganese ionsReduce power consumptionElectrolysisZinc metal
The invention relates to a vitriol precipitating and Fe removing process in a zinc hydrometallurgy technology. The vitriol precipitating and Fe removing process comprises the following steps: adjusting the pH value of vitriol precipitating supernatant, adding an oxidizing agent for reaction, adjusting the pH value of reaction liquid to 4.8-5.2, precipitating so as to removing Fe, and adjusting the pH value of the vitriol precipitating supernatant to 3.5-4.5, wherein the oxidizing agent is an H2O2 oxidizing agent or an H2O2-manganese powder combined oxidizing agent, the reaction temperature is 60-70 DEG C, and the stirring reaction time is 40-90 minutes. According to the vitriol precipitating and Fe removing process, Fe2+ is oxidized into Fe3+ by using the H2O2 oxidizing agent or the H2O2-manganese powder combined oxidizing agent, and the formed iron vitriol is removed. The iron removal by jarosite process is low in cost and capable of reducing the content of manganese ions in a system, effectively controlling the manganese ions in the system to 5-6g / l required by the process and effectively reducing systemic circulation volume and zinc content in acid leaching slag and iron vitriol slag, thereby increasing the recovery rate of zinc metal, improving the efficiency of electrolytic currents and reducing power consumption; and production processes are simplified.
Owner:BAIYIN NONFERROUS GROUP
Sodium hypochlorite disinfectant generator
PendingCN111334819AFast productionEasy to operateCellsElectrode shape/formsElectrolysisSodium chlorate
The invention discloses a sodium hypochlorite disinfectant generator comprising a supporting shell. One end of a tubular body is connected to the supporting shell, the other end of the tubular body isconnected with a barrel for containing sodium chloride, and a through hole communicated with an inner hole of the tubular body is formed in the outer circumferential face of the tubular body. The supporting shell is further fixedly connected with a rod-shaped electrode or two rod-shaped electrodes, and the rod-shaped electrodes are arranged in the middle of the inner hole of the tubular body. When the supporting shell is fixedly connected with one rod-shaped electrode, the tubular body becomes an electrode, and when the tubular body and the barrel body filled with sodium chloride extend intowater together, the rod-shaped electrode and the tubular body are powered by a power supply for electrolysis; and when the two rod-shaped electrodes are fixedly connected to the supporting shell and the tubular body and the barrel body filled with sodium chloride extend into water together, the two rod-shaped electrodes are powered by the power supply for electrolysis. A material storage cavity isformed in the supporting shell, and materials for absorbing chlorine are contained in the material storage cavity. The sodium hypochlorite disinfectant generator is simple and convenient to operate,the preparation speed of the sodium hypochlorite disinfectant is high, and the sodium chloride consumption and the electrolysis energy consumption are reduced.
Owner:吴允苗
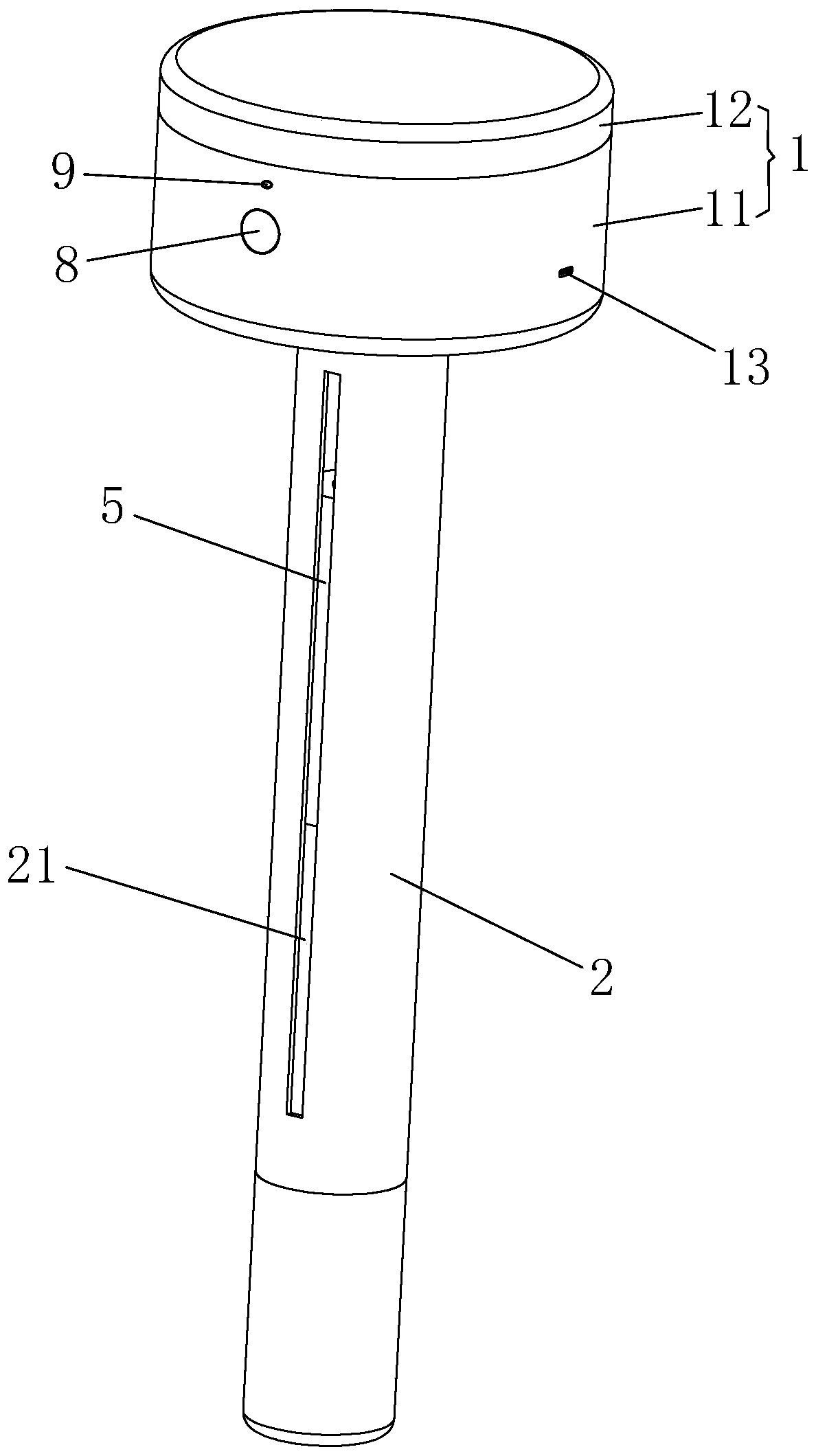
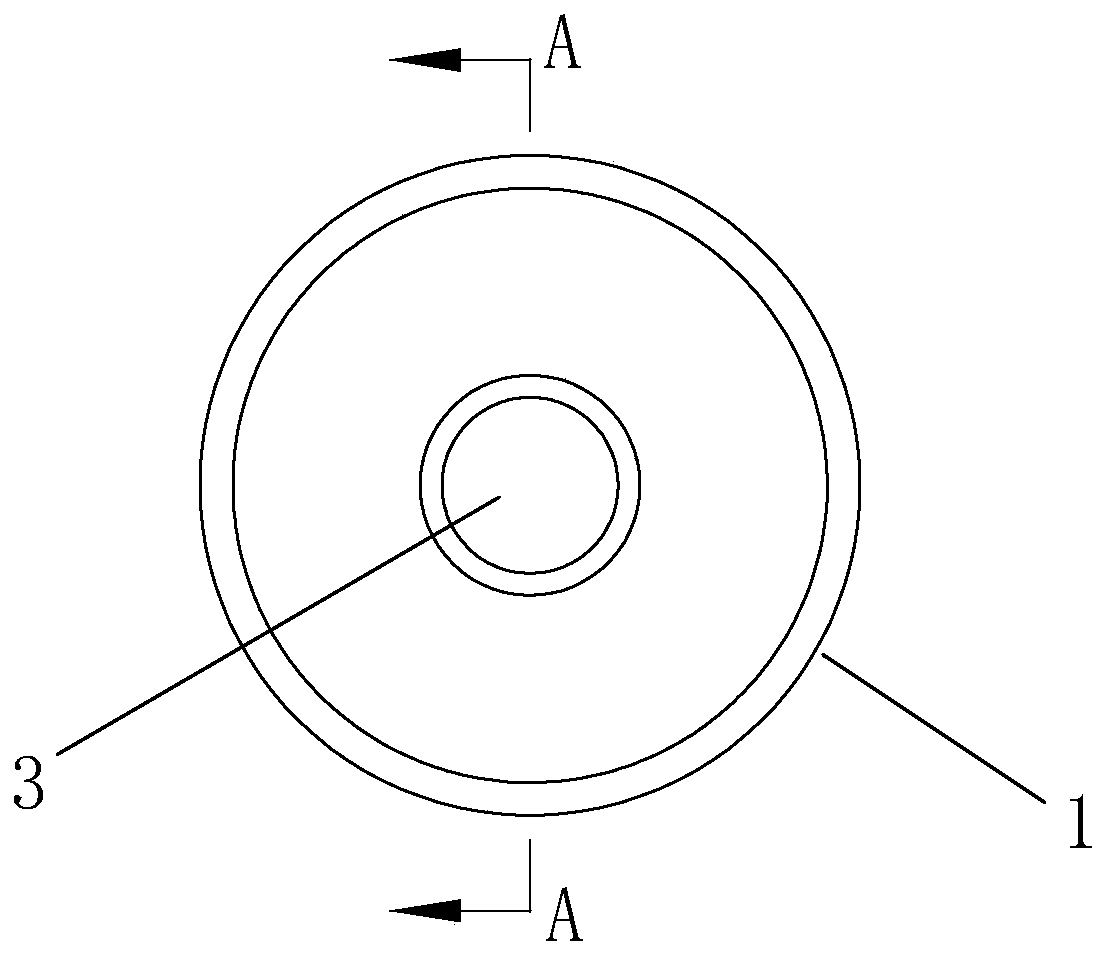
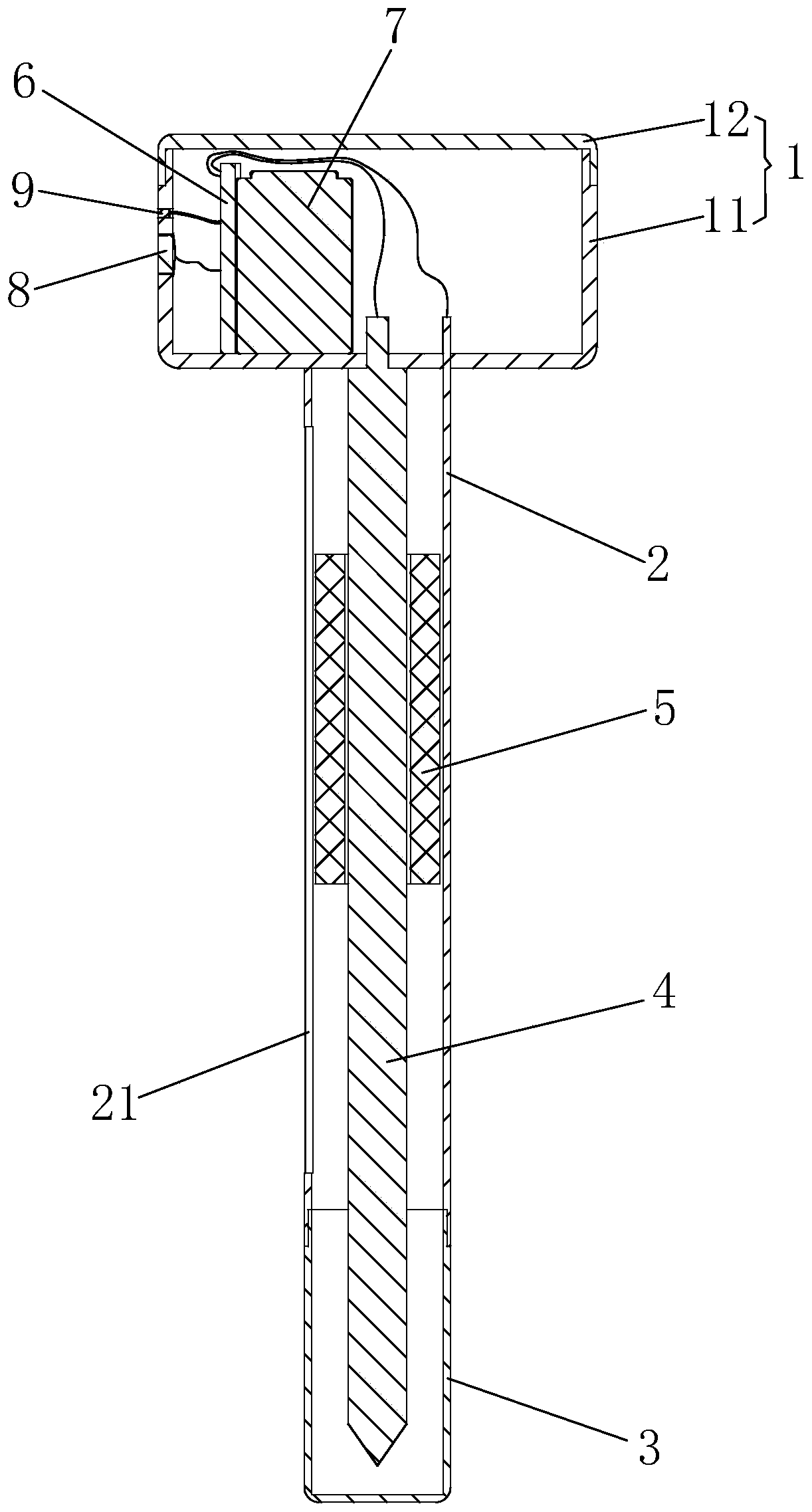
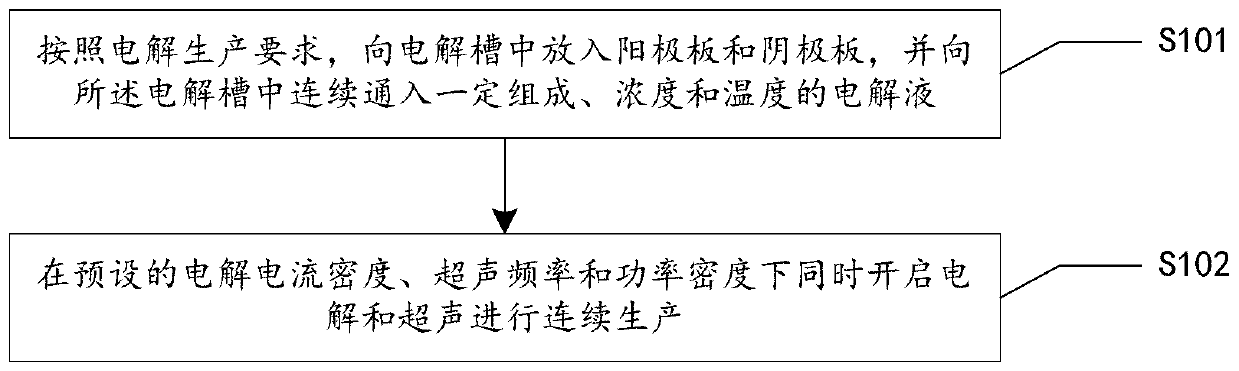
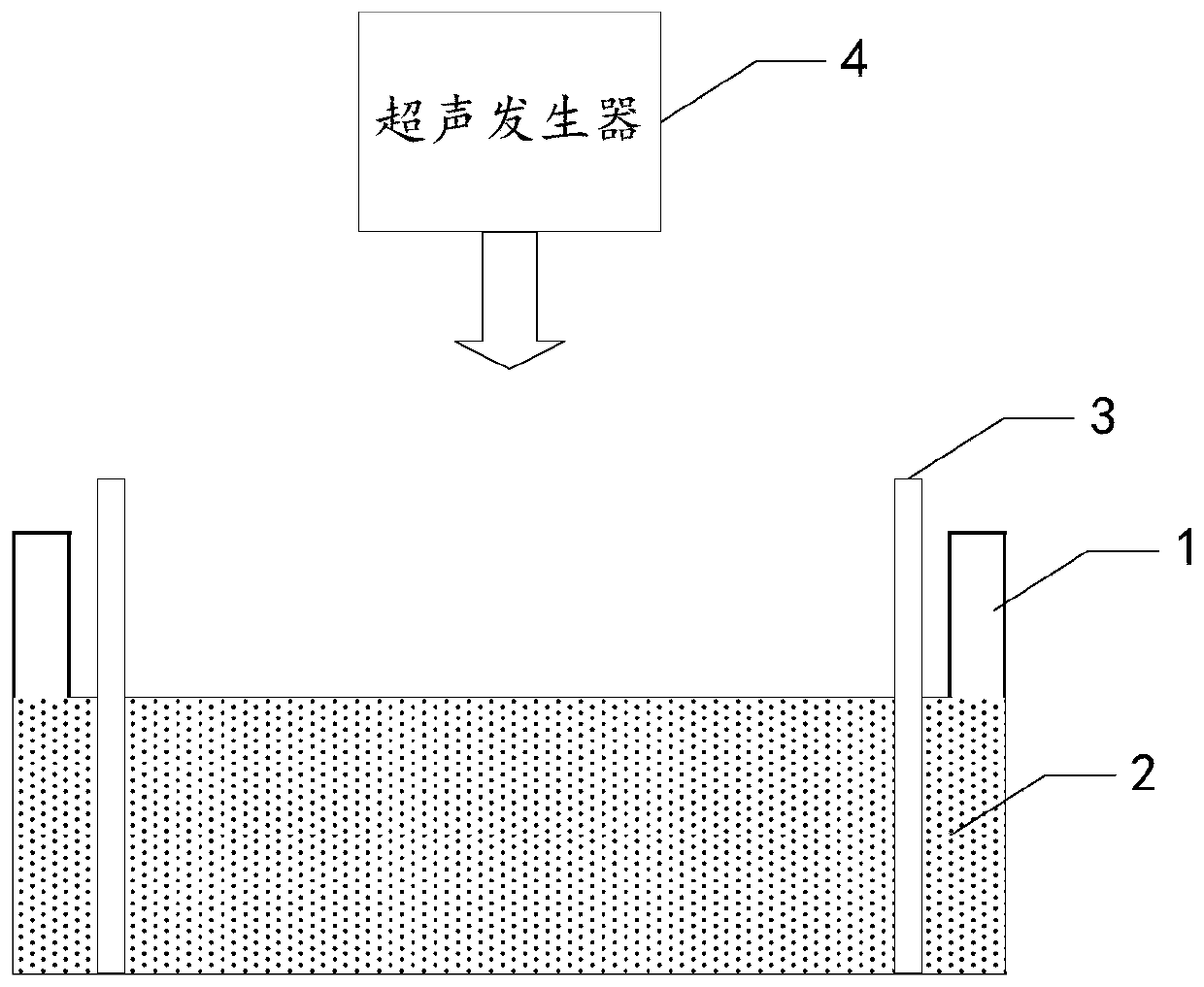
Groove surface acid mist source reduced clean production method and system in electrolysis workshop
InactiveCN110373686AReduce productionReduce coalescingElectrolysis componentsEcological environmentSlag
The invention provides a groove surface acid mist source reduced clean production method and system in an electrolysis workshop. According to the method, during normal electrolysis process, super sound with certain ultrasonic frequency and power density is applied to an electrolyte solution inside a groove and anode and cathode plates, thus achieving the purpose of controlling the size, quantity,motion speed and direction of oxygen and hydrogen bubbles which are separated out of the surface of the solution, and in this way, the subsequent process that the oxygen and hydrogen bubbles blast andrelease acid mist droplets on the free liquid surface of the electrolyte solution can be further controlled. By adopting the method, the occurrence of a large amount of acid mist caused by the blasting and sputtering of the bubbles on the free liquid surface of the electrolyte solution as well as the high-value resource loss can be reduced from the source, so that investment on end treatment facilities and high operation cost can be remarkably reduced, in this way, secondary pollution caused by acid mist treatment slag is avoided, and the damage of the groove surface acid mist to the health of workers in the workshop as well as to the ecological environment beyond the workshop can be relieved; and in addition, the electrolytic current efficiency is improved, and the electric energy consumption is reduced. The method is generally suitable for source prevention and control of the groove surface acid mist during zinc electrolysis, manganese electrolysis, nickel electrolysis, and other wet electrolytic processes.
Owner:CHINESE RES ACAD OF ENVIRONMENTAL SCI
An industrial energy-saving high-frequency pulse electrolytic manganese device
ActiveCN104831314BImprove conversion efficiencyReduce harmonic effectsPhotography auxillary processesElectrolysisAlternating current
The invention discloses an industrial energy-saving high-frequency impulse electrolytic manganese device which comprises one or more phase-shifting transformers, one or more three-phase rectifier bridge assemblies, one or more power assemblies and a power master control unit, wherein the primary sides of the phase-shifting transformers are connected with the output end of a three-phase alternating-current supply and the secondary sides of the phase-shifting transformers are in one-to-one corresponding connection with the inputs of the three-phase rectifier bridge assemblies, respectively; the output ends of the three-phase rectifier bridge assemblies are correspondingly connected with the input ends of the power assemblies, and the output ends of the power units output high frequency impulse signals to electrolytic baths; the voltages output by the secondary sides of phase-shifting transforming units are determined according to the quantity of the electrolytic baths connected in series; when a plurality of the phase-shifting transformers are adopted, the phase-shifting transforming units are the phase-shifting transformers in multiple forms for reducing harmonic waves of an alternating current input current and harmonic amplitudes and current impulses of a direct current output voltage; the power master control unit is respectively connected with one or more power assemblies for current-sharing control of the power assemblies.
Owner:CHENGDU XINMING ENERGY SAVING TECH CO LTD
Electric flocculation method and device for recovering and processing desulfurized wastewater
ActiveCN101817575BAchieve removalImplement extractionWater/sewage treatmentWaste water treatment from gaseous effluentsAmmoniacal nitrogenElectro flocculation
The invention discloses electric flocculation method and device for recovering and processing desulfurized wastewater. The electric flocculation method for recovering and processing desulfurized wastewater comprises the following steps of: collecting desulfurized wastewater to enter an electric flocculation reactor, then, carrying out solid-liquid separation, recovering the separated clear effluent water in a clinker-flushing system of an electric power plant or directly discharging the separated clear effluent water, conveying the sludge to dewater in a mud-processing system, and returning the effluent water of the mud-processing system to a water intake system. Compared with the prior art, the electric flocculation technology is adopted to substitute a complicated chemical method for recovering and processing the desulfurized wastewater, can effectively remove pollutants in the wastewater, reaches a heavy metal processing rate up to 95-99 percent, a chlorine-processing rate over 80 percent and removal rates of ammonian, and the like over 90 percent, greatly shortens the processing time, improves the processing efficiency and saves the investment cost. Moreover, the electric flocculation processing device has small size, extremely small floor occupation, good environmental-protection performance and low cost without additional flocculating agents.
Owner:华电水务工程有限公司
Low-temperature aluminum electrolysis process and electrolyte
InactiveCN101713082BReduced ability to dissolve aluminaIncreased ability to dissolve aluminaAnode effectHeat losses
The invention relates to a low-temperature aluminum electrolysis process and electrolyte. The electrolyte consists of Na3AlF6, K3AlF6, Al2O3, AlF3 and LiF; and the electrolysis temperature is between 850 and 910 DEG C, the coefficient of an anode effect is smaller than 0.1 time / tank day, and the anode current density is 0.5 to 1.2 A / cm2. The primary crystal temperature of the electrolyte adoptingthe component formula is low and changes comparatively slowly along with the changes of the components in favor of solving the problems of uneven tank bottom current, unstable tank voltage and the like caused by sharp raising of the primary crystal temperature of a melt and cathode crusting due to directional migration of Na+ and cathode enrichment. Besides, The K3AlF6 in the electrolyte can effectively increase the capability of the melt for dissolving the aluminum oxide, and solve the problem of deposition of the aluminum oxide at the bottom of a tank. Because the electrolysis temperature is lower, the heat loss of an electrolytic tank can be greatly reduced, and the energy-saving current efficiency is high. The low-temperature aluminum electrolysis process is simple and is easy to operate, the electrolyte has reasonable components, and the low-temperature aluminum electrolysis process can effectively reduce the aluminum electrolysis temperature, inhibit the cathode crusting in the process of aluminum electrolysis, improve the current efficiency of the aluminum electrolysis and reduce the energy consumption of the aluminum electrolysis, is suitable for industrial production, andcan replace the conventional aluminum electrolysis process.
Owner:CENT SOUTH UNIV
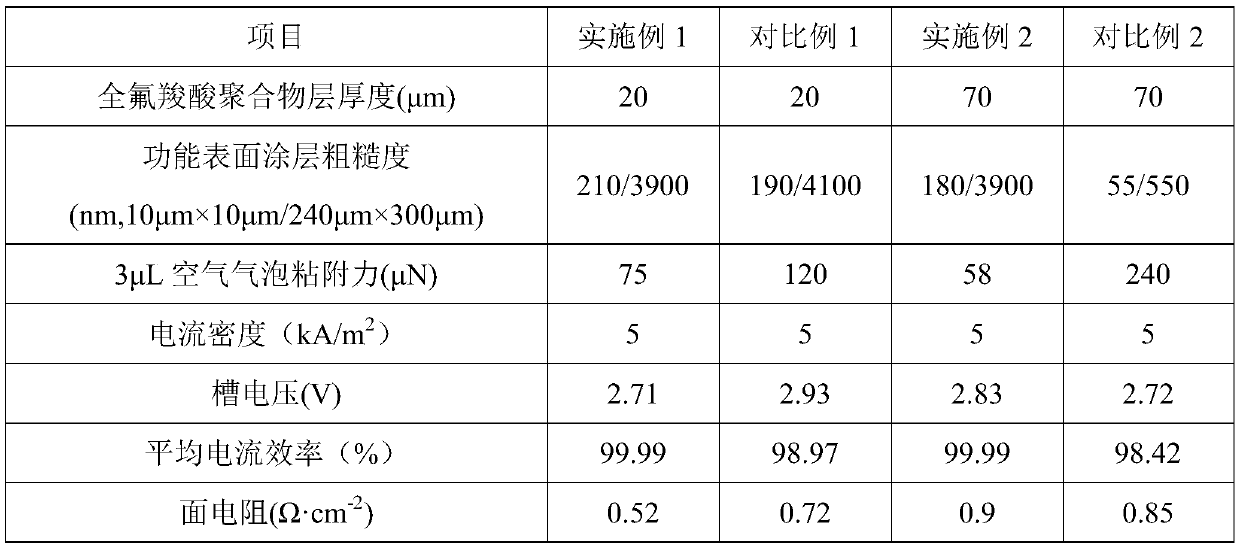
Reinforced perfluorocarboxylic acid ion exchange membrane with bubble thinning function and preparation method thereof
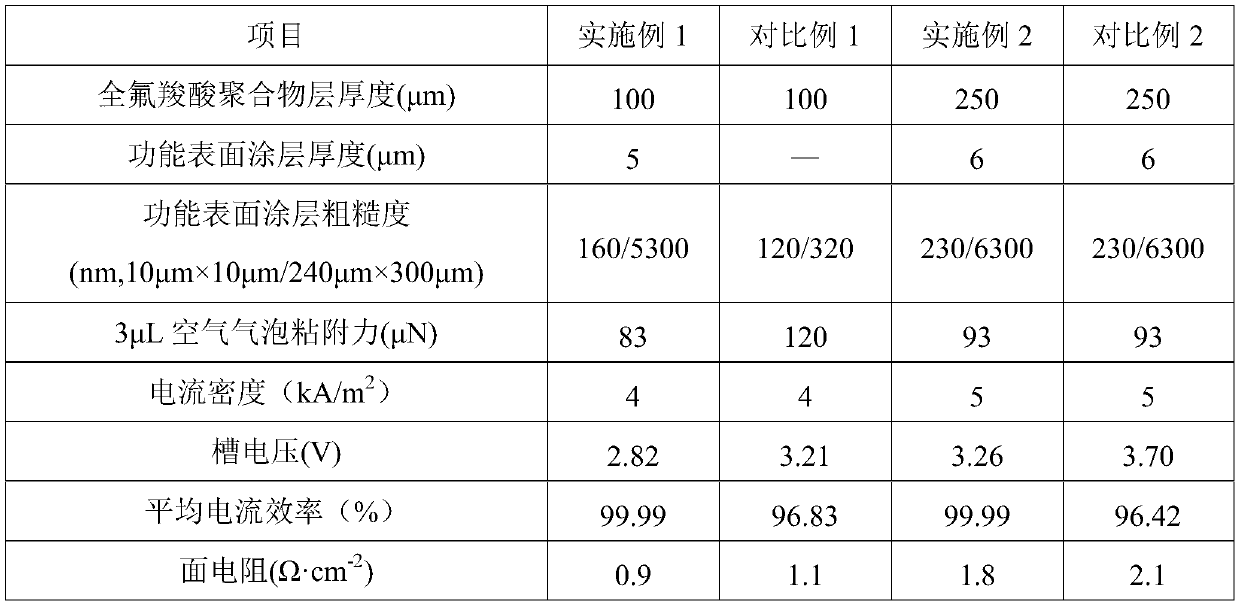
InactiveCN111118543AHinder reverse migrationSmall ion channelOrganic diaphragmsPhysical chemistryCarboxylic acid
The invention belongs to the technical field of ion exchange membranes, and particularly relates to a reinforced perfluorocarboxylic acid ion exchange membrane with a bubble dredging function and a preparation method thereof. The reinforced perfluorocarboxylic acid ion exchange membrane with the bubble thinning function is composed of a perfluorocarboxylic acid polymer layer, a porous reinforcingmaterial layer and functional surface coatings, the functional surface coatings are located on the upper surface and the lower surface of the perfluorocarboxylic acid polymer layer, the porous reinforcing material layer is embedded in the perfluorocarboxylic acid polymer layer, and the functional surface coating is of a porous rough structure composed of a perfluorinated ionic polymer. The reinforced perfluorocarboxylic acid ion exchange membrane with the bubble thinning function can effectively reduce the cell voltage, reduce the electrolysis energy consumption and reduce the production costin a novel zero-polar-distance electrolytic cell under the condition of high current density, and is suitable for the field of electrolysis of sodium chloride and potassium chloride; the invention also provides a preparation method thereof.
Owner:SHANDONG DONGYUE POLYMER MATERIAL
Aluminum electrolysis cell with aluminum as cathode
ActiveCN105780055AEliminate Aluminum Oxide PrecipitationEliminate Cellular Microbattery DefectsAluminum electrolysisAluminium oxide
The invention discloses an aluminum electrolysis cell with aluminum as a cathode, and relates to the improvement on aluminum electrolysis cell production with cryolite aluminum monoxide melt as an electrolyte and through a molten salt method. The structure of the aluminum electrolysis cell comprises a prebaked anode, a discharging device and a dust purifying device. The aluminum electrolysis cell is characterized in that the structure of a cathode system of the aluminum electrolysis cell comprises an electrolysis cell body, cathode partition walls and a cathode busbar, the cathode partition walls are arranged in cell ledges at the two ends of the aluminum electrolysis cell body, a bottom channel is formed between the bottoms of the cathode partition walls and the lower bottom of the aluminum electrolysis cell body, side channels are formed by the cathode partition walls and the inner walls of the cell ledges at the ends of the aluminum electrolysis cell body, and the cathode busbar enters the aluminum electrolysis cell body from the side channels formed by the cathode partition walls and the inner walls of the cell ledges at the ends of the aluminum electrolysis cell body. According to the aluminum electrolysis cell with aluminum as the cathode, molten aluminum in which no cathode carbon block, no steel bar and no titanium boride coating exist is adopted as the cathode to replace a cathode composed of steel bars and a carbon block set. The defects that according to a traditional aluminum electrolysis cell, aluminum oxide deposit exists on cathode carbon blocks and cellular micro-batteries exist are eliminated. In the electrolysis production process, the problem that aluminum oxide and aluminum carbide which are deposited and generated at the furnace bottom pollute electrolysis molten aluminum does not exist.
Owner:XINJIANG UNIVERSITY
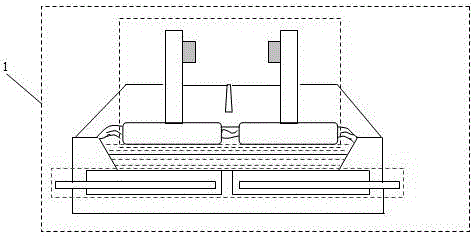
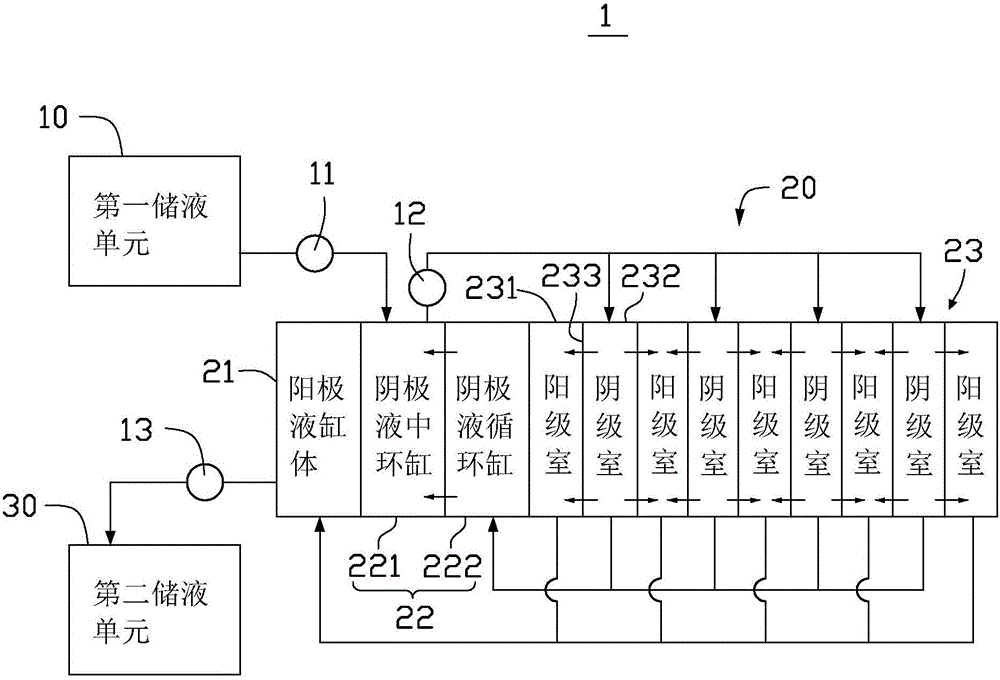
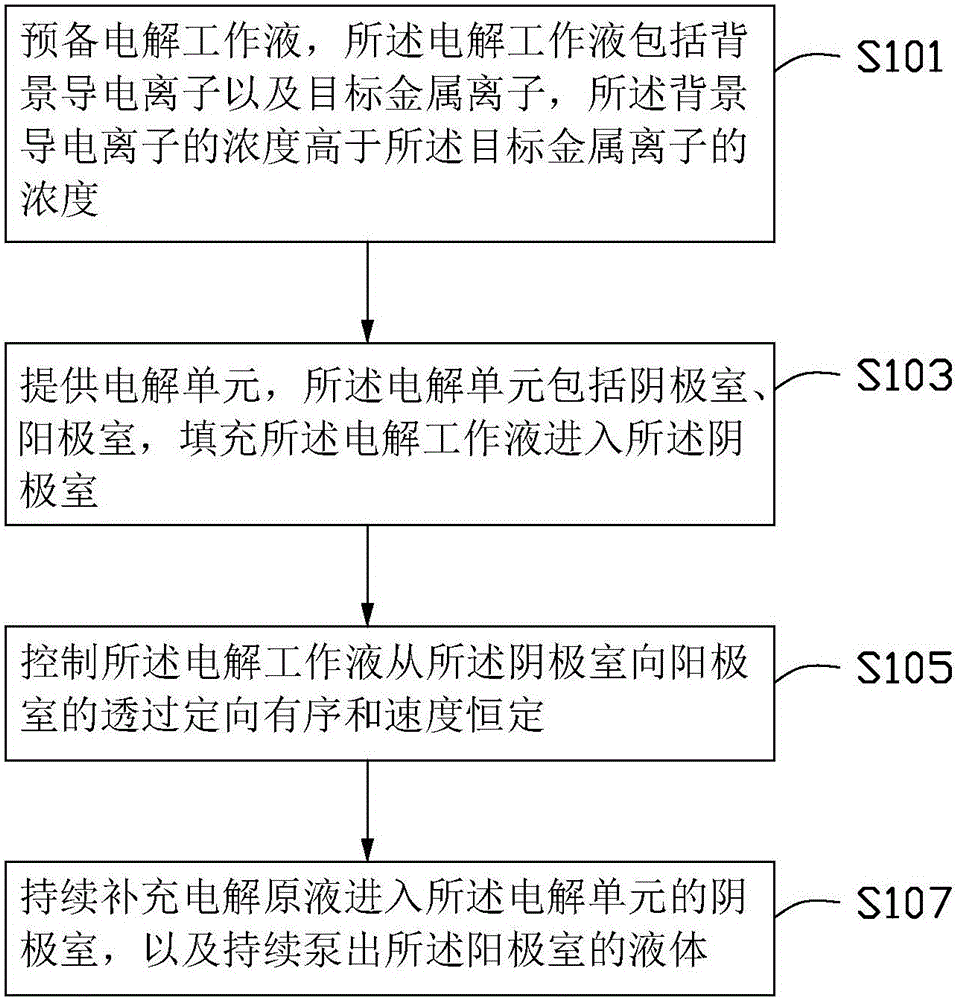
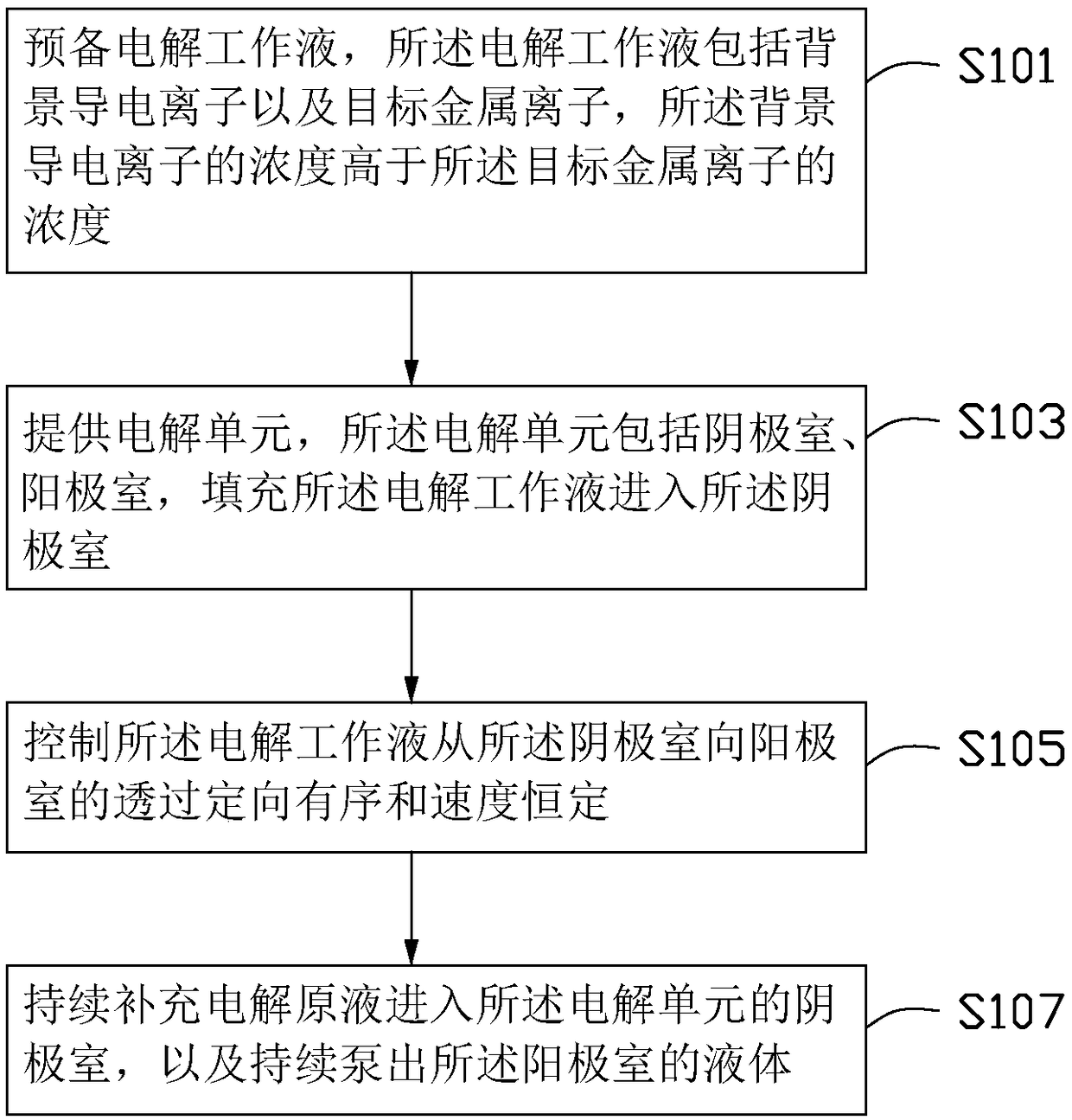
Method for improving efficiency of electrolytic current, metal electrodeposition method and metal electrodepostion device
ActiveCN106048652AImprove electrolysis current efficiencyHigh cathode current efficiencyPhotography auxillary processesProcess efficiency improvementElectrolysisElectrowinning
The invention provides a method for improving the efficiency of electrolytic current. The method includes the steps that an electrolytic working solution is prepared and includes background electric conduction ions and electrolytic target metal ions, wherein the concentration of the background electric conduction ions is higher than that of the target metal ions; the electrolytic working solution enters an electrolyzing unit in a filling manner, and the electrolyzing unit is provided with a cathode room and an anode room; and the electrolytic working solution is controlled to pass through the part between the cathode room and the anode room in a directional and orderly manner and at a constant speed. The invention further provides a method for conducting metal electrodeposition through the above method and a metal electrodeposition device.
Owner:SHENZHEN SHENTOU ENVIRONMENT TECH CO LTD
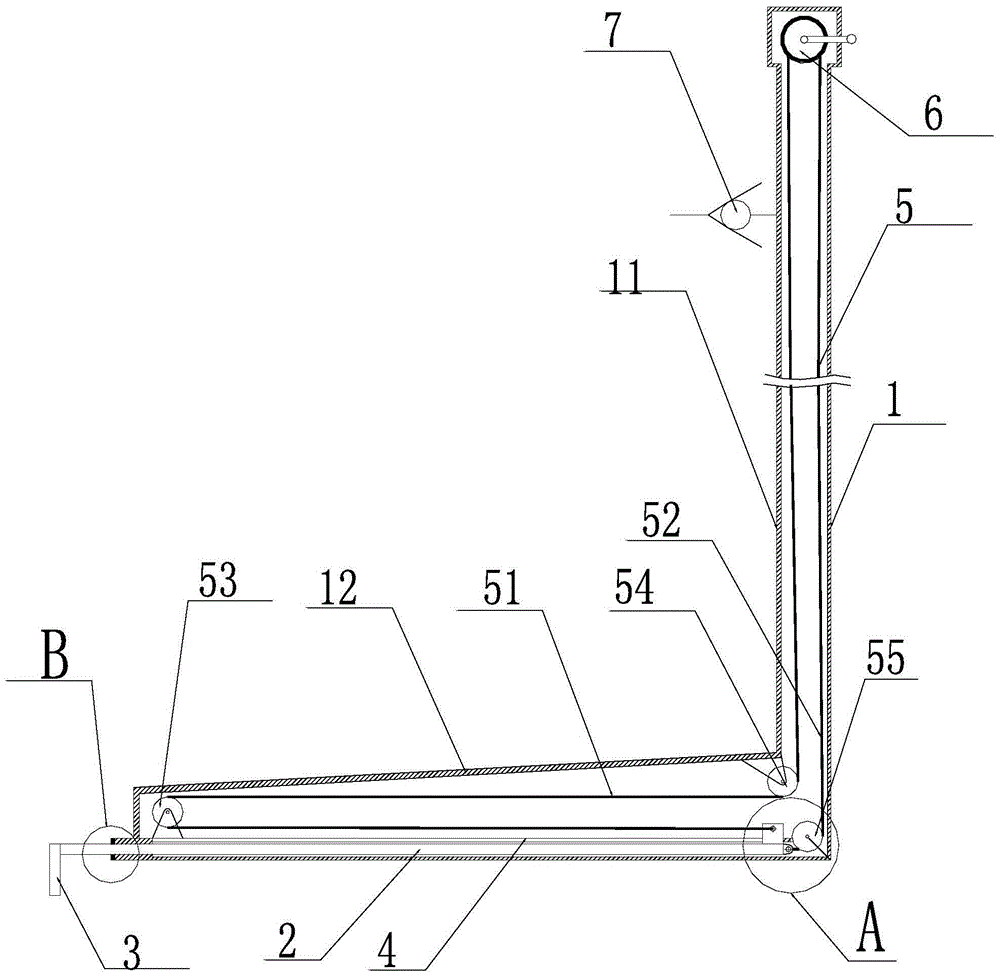
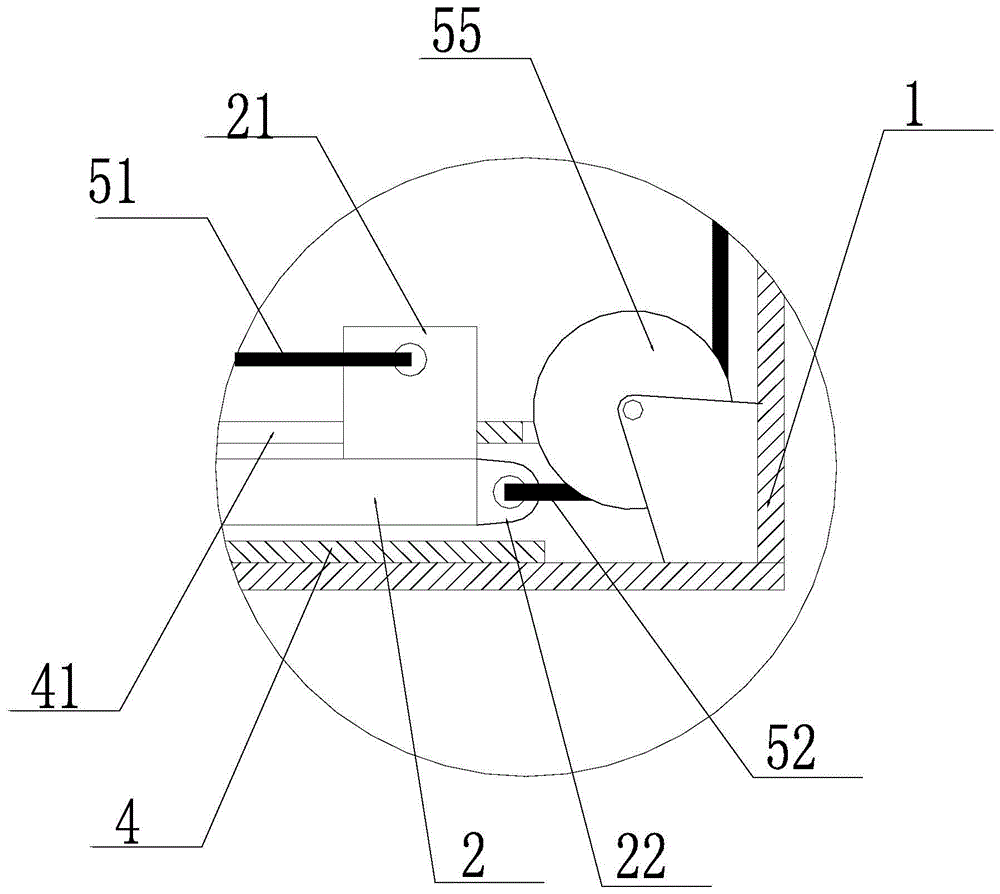
Slag removal device and method for lower inserted magnesium electrolytic cell
ActiveCN104498994BSolve the problem that it cannot be placed between the graphite electrolytic holesReduce the intensity of manual operationElectrolysis componentsElectricitySlag
The invention belongs to the field of non-ferrous metal metallurgy, and discloses a lower-insert electrolytic magnesium cell deslagging device and a deslagging method. The device comprises an L-shaped support, a push rod, a scraper and a guiding mechanism, wherein the L-shaped support comprises a handrail section and a working section connected with the handrail section; the guiding mechanism is arranged on the working section; the push rod is arranged in the guiding mechanism; the scraper is connected with the external end part of the push rod; a pulley system is arranged on the L-shaped support, and is connected with the push rod; the push rod can make a telescopic motion along the guiding mechanism under driving of the pulley system. According to the deslagging device, the push rod can make telescopic motion by utilizing driving of the pulley system, so that the scraper can go deep to the bottom of an electrolytic cell to remove residues at the circulating port, and the problem that a traditional long hook cannot be put into each graphite electrolytic hole can be solved, so that, on the one hand, the labor operation intensity can be reduced, and the electrolysis current efficiency can be greatly improved; and on the other hand, the device is applicable to multi-stage slots and has a practical application value.
Owner:PANZHIHUA IRON & STEEL RES INST OF PANGANG GROUP
Method for preparing prebaked anode through anode mud
The invention discloses a method for preparing a prebaked anode by utilizing anode slime, and belongs to the technical field of prebaked anode preparation. The present invention collects anode slime, adds sulfuric acid for immersion, mixes and stirs to collect sediment for later use, and filters to obtain a filtrate, adds citric acid to the filtrate, stirs and reacts, obtains turbid liquid through filtration, mixes with the spare sediment, kneads and presses After the green body is formed, it is kept warm in a calciner, and then the temperature is raised, and after calcined, it is immersed in water to cool to room temperature, and air-dried to obtain a prebaked anode. The beneficial effects of the present invention are: the preparation steps of the present invention are simple, the energy consumption of the obtained product is more than 30.2% lower than that of other products, no need for regular replacement, and the service period is long; the material is not easy to oxidize and fall off during use, and the electrolytic current efficiency is increased by 22-25% %.
Owner:CHANGZHOU LANXU CHEM CO LTD
Double-layer aluminum cathode aluminum electrolysis cell cathode
The invention discloses a double-layer aluminum cathode aluminum electrolysis cell cathode, and relates to the improvement on aluminum electrolysis cell production with aluminum as the cathode, with cryolite aluminum monoxide melt as an electrolyte and through a molten salt method. The double-layer aluminum cathode aluminum electrolysis cell cathode is characterized in that the structure of the cathode comprises an electrolysis cell body, two cathode partition walls, a cathode busbar and a cathode horizontal partition plate, wherein the two cathode partition walls are arranged in cell ledges at the two ends of the aluminum electrolysis cell body and fixedly connected with the inner walls of side cell ledges of the electrolysis cell body, the cathode busbar enters the electrolysis cell body from side channels formed by the cathode partition walls and the inner walls of the cell ledges at the ends of the aluminum electrolysis cell body, and the cathode horizontal partition plate is located in the electrolysis cell body and provided with molten aluminum through holes. According to the double-layer aluminum cathode aluminum electrolysis cell cathode, molten aluminum in which no cathode carbon block, no steel bar and no titanium boride coating exist is adopted as the cathode to replace a cathode composed of steel bars and a carbon block set. The defects that according to a traditional aluminum electrolysis cell, aluminum oxide deposit exists on cathode carbon blocks and cellular micro-batteries exist are eliminated. In the electrolysis production process, the problem that aluminum oxide and aluminum carbide which are deposited and generated at the furnace bottom pollute electrolysis molten aluminum does not exist.
Owner:XINJIANG UNIVERSITY
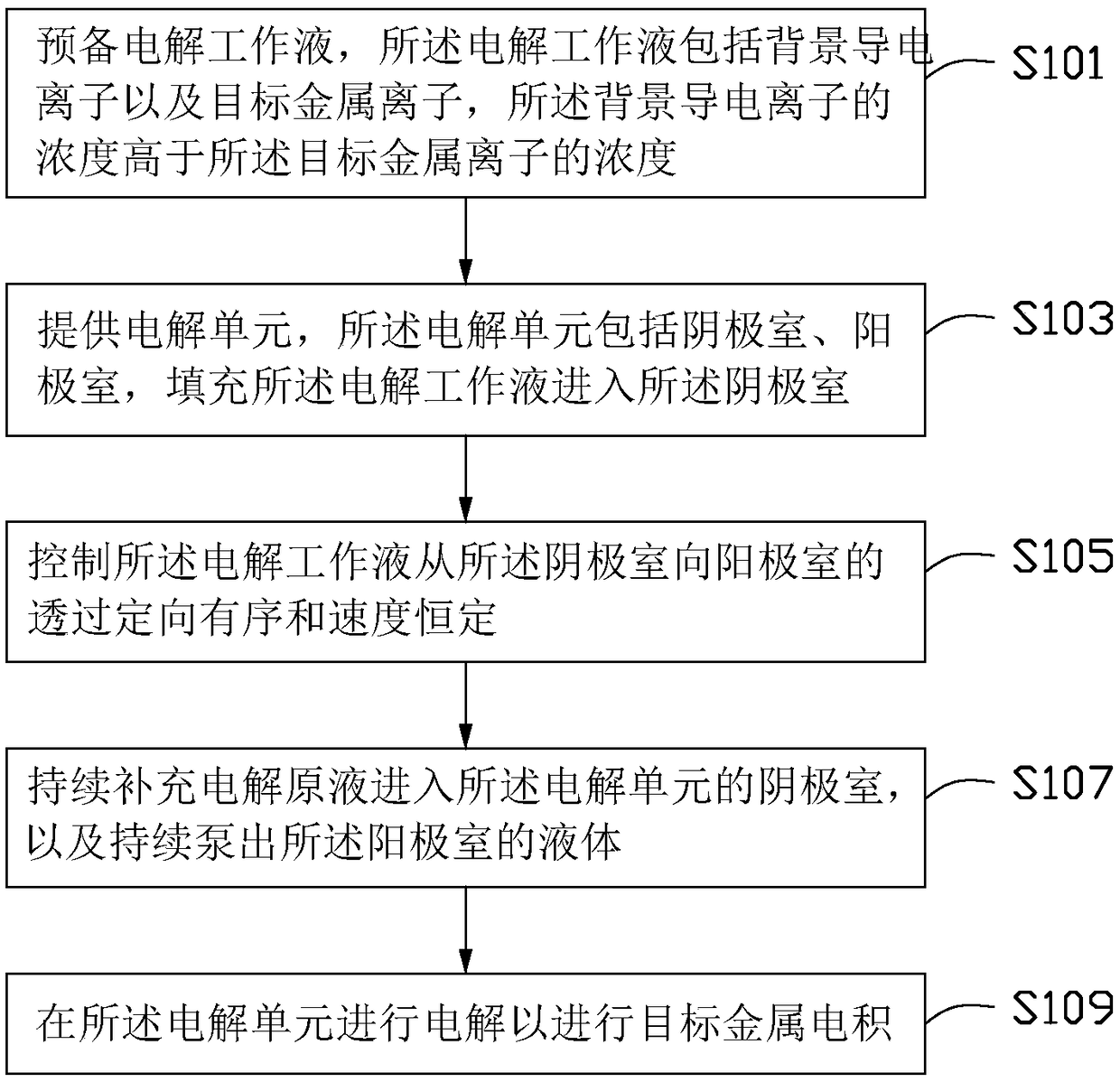
Method for improving electrolytic current efficiency, metal electrowinning method, and metal electrowinning device
ActiveCN106048652BImprove electrolysis current efficiencyHigh cathode current efficiencyPhotography auxillary processesProcess efficiency improvementElectrolysisElectrowinning
The invention provides a method for improving the efficiency of electrolytic current. The method includes the steps that an electrolytic working solution is prepared and includes background electric conduction ions and electrolytic target metal ions, wherein the concentration of the background electric conduction ions is higher than that of the target metal ions; the electrolytic working solution enters an electrolyzing unit in a filling manner, and the electrolyzing unit is provided with a cathode room and an anode room; and the electrolytic working solution is controlled to pass through the part between the cathode room and the anode room in a directional and orderly manner and at a constant speed. The invention further provides a method for conducting metal electrodeposition through the above method and a metal electrodeposition device.
Owner:SHENZHEN SHENTOU ENVIRONMENT TECH CO LTD
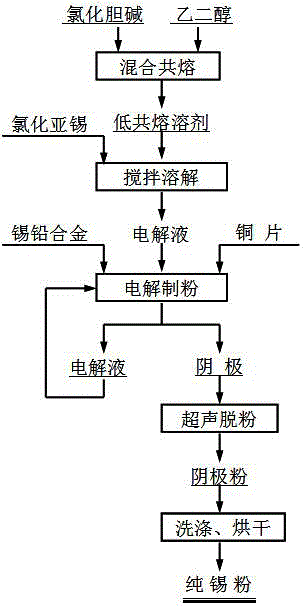
Method for electrolytically separating stannum-lead alloy through low eutectic solvent
The invention relates to a method for electrolytically separating a stannum-lead alloy through a low eutectic solvent and belongs to the technical field of ionic liquid metallurgy. Choline chloride and ethanediol are mixed in accordance with the molar ratio being 1:2 and then are subjected to heating, stirring and eutectic melting at the temperature of 80 DEG C till clear and transparent liquid is formed, and a choline chloride-ethanediol low eutectic solvent, namely 1ChC1:2EG DES for short, is synthesized; stannous chloride is added into the synthesized 1ChC1:2EG DES, stirring is conducted till even dissolution is achieved, and an electrolyte solution is prepared; the prepared electrolyte solution is heated to be 30-70 DEG C, a stannum-lead alloy block serves as a positive pole, a copper sheet serves as a negative pole, and electrolysis is conducted; and branch-shaped stannum powder is obtained on the negative pole and is slightly scraped off from the negative pole, and the stannum powder containing Sn being equal to or more than 99.95 wt% is obtained after the branch-shaped stannum powder is subjected to ultrasonic dispersion cleaning and drying. According to the electrolytically separating the stannum-lead alloy through the low eutectic solvent, the electrolyte solution is prepared by adding the moderate stannous chloride into the choline chloride-ethanediol low eutectic solvent, the steam pressure of the electrolyte solution is extremely low, the electrolyte solution is hardly volatilized, heat stability is good, an electrolyte system is environmentally friendly, and the electrolyte solution can be recycled.
Owner:KUNMING UNIV OF SCI & TECH
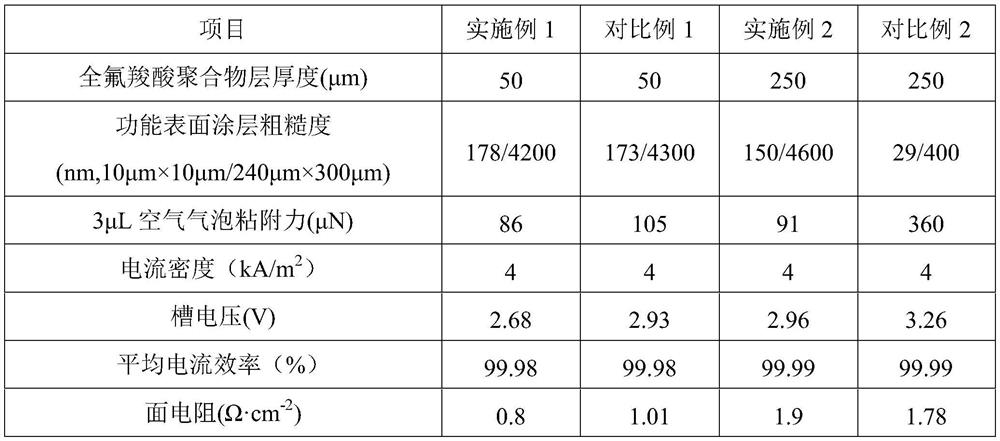
Perfluorocarboxylic acid ion exchange membrane with rough coating and method for its preparation
ActiveCN111101152BHinder reverse migrationSmall ion channelOrganic diaphragmsPhysical chemistryCarboxylic acid
The invention belongs to the technical field of ion exchange membranes, in particular to a perfluorocarboxylic acid ion exchange membrane with a rough coating and a preparation method thereof. The perfluorocarboxylic acid ion exchange membrane with rough coating of the present invention is composed of a perfluorocarboxylic acid polymer layer and a functional surface coating, wherein the functional surface coating is located on the upper and lower surfaces of the perfluorocarboxylic acid polymer layer; The thickness of the perfluorocarboxylic acid polymer layer is 50-250 μm, and the thickness of the functional surface coating is 10nm-30 μm; the functional surface coating is a porous rough structure composed of a mixture of perfluoroionic polymers and metal oxides. structure. The perfluorocarboxylic acid ion-exchange membrane with a rough coating of the present invention can effectively reduce the cell voltage, reduce electrolysis energy consumption, and reduce production costs in a zero-pole pitch electrolytic cell under novel high current density conditions; the present invention also provides its preparation method.
Owner:SHANDONG DONGYUE POLYMER MATERIAL
Pre-baked anode for aluminum electrolysis and its preparation method
The invention relates to a prebaked anode cell and the preparation method. It comprises following steps: (1) designing the shape for prebaked anode cell; (2) adding waste corner aluminum wire produced in aluminum production process during carbon anode preparation, to improve the conductivity for cabon anode, the addition amount is 1.5- 2.0%., the aluminum wire is filled into the micropores when it is conversed into liquid from solid during calcination, to increase the carrier for carbon anode base material. The invention is characterized by saved raw material cost, easy operation and management for thermal- insulating cover material, good flowability of filled material, increased currency efficiency by 0.5%, reduced resources consumption, 4.44 ton of recovery waste aluminum, and reduced carbon dioxide emmision.
Owner:YUNNAN ALUMINUM
Method for preparing Gd-Mg intermediate alloy by fluorination system coelectrodeposition
The present invention discloses a process for preparing of Gd-Mg intermediate alloy by composite electrodeposition of fluoridated systems. Gd and Mg are electrochemically separated at the cathode of a round or square electrolytic cell with the mixture of 80-95 wt% commercial Gd2O3 (purity higher than 95%) and MgO (purity higher than 95) as the raw material and GdF3-LiF or GdF3-LiF-BaF2 molten fluoride salts as the electrolytic medium and then alloyed to produce Gd-Mg intermediate alloy with the weight percentage of Gd higher than 85 percent. The prepared rear earth magnesium alloy has high magnesium content and uniform ingredients. The present invention has convenient operation that the electrolysis is continuously conducted and the tapping is conducted by the siphon method; the recovery of rear earth is higher than 90 percent, and the exhaust gas meets the requirement of environment protection; the intermediate alloy has low impurity content and low production cost.
Owner:GANNAN NORMAL UNIV
Ultrathin perfluorocarboxylic acid ion exchange membrane with rough coating and preparation method of same
ActiveCN111118542AHinder reverse migrationSmall ion channelOrganic diaphragmsTextiles and paperPhysical chemistryCarboxylic acid
The invention belongs to the technical field of ion exchange membranes, and particularly relates to an ultrathin perfluorocarboxylic acid ion exchange membrane with a rough coating and a preparation method of the same. The ultrathin perfluorocarboxylic acid ion exchange membrane with the rough coating is composed of a perfluorocarboxylic acid polymer layer, a porous non-woven polymer layer and functional surface coatings, wherein the functional surface coatings are located on the upper surface and the lower surface of the perfluorocarboxylic acid polymer layer, and the porous non-woven polymerlayer is embedded in the perfluorocarboxylic acid polymer layer; the thickness of the perfluorocarboxylic acid polymer layer is 10-80 [mu]m; the functional surface coating is of a porous rough structure formed by a mixture of a perfluorinated ionic polymer and a metal oxide. According to the invention, the ultrathin perfluorocarboxylic acid ion exchange membrane has the rough coating, so that thecell voltage can be effectively reduced, the electrolysis energy consumption can be reduced, and the production cost can be reduced in a novel zero-polar-distance electrolytic cell under the condition of high current density; the invention also provides a preparation method thereof.
Owner:SHANDONG DONGYUE POLYMER MATERIAL
Reinforced perfluorocarboxylic acid ion exchange membrane with rough coating, and preparation method thereof
InactiveCN111188062AImprove electrolysis current efficiencyIncreased durabilityOrganic diaphragmsIon-exchange membranesElectrolytic cell
The invention belongs to the technical field of ion exchange membranes, and particularly relates to a reinforced perfluorocarboxylic acid ion exchange membrane with a rough coating, and a preparationmethod thereof. The reinforced perfluorocarboxylic acid ion exchange membrane is composed of a perfluorocarboxylic acid polymer layer, a porous reinforcing material layer and functional surface coatings, wherein the functional surface coatings are located on the upper surface and the lower surface of the perfluorocarboxylic acid polymer layer, the porous reinforcing material layer is embedded in the perfluorocarboxylic acid polymer layer, and the functional surface coating is of a porous rough structure composed of a perfluorinated ionic polymer and a metal oxide. The reinforced perfluorocarboxylic acid ion exchange membrane with the rough coating can effectively reduce the cell voltage, reduce the electrolysis energy consumption and reduce the production cost in a novel zero-polar-distance electrolytic cell under the condition of high current density, and is suitable for the field of electrolysis of sodium chloride and potassium chloride. The invention also provides a preparation method of the reinforced perfluorocarboxylic acid ion exchange membrane.
Owner:SHANDONG DONGYUE POLYMER MATERIAL
Double-layer aluminum cathode aluminum electrolytic cell cathode
InactiveCN105780057BEliminates contact pressure dropReduce electrolysis power consumptionCarbideMolten salt
The invention discloses a double-layer aluminum cathode aluminum electrolysis cell cathode, and relates to the improvement on aluminum electrolysis cell production with aluminum as the cathode, with cryolite aluminum monoxide melt as an electrolyte and through a molten salt method. The double-layer aluminum cathode aluminum electrolysis cell cathode is characterized in that the structure of the cathode comprises an electrolysis cell body, two cathode partition walls, a cathode busbar and a cathode horizontal partition plate, wherein the two cathode partition walls are arranged in cell ledges at the two ends of the aluminum electrolysis cell body and fixedly connected with the inner walls of side cell ledges of the electrolysis cell body, the cathode busbar enters the electrolysis cell body from side channels formed by the cathode partition walls and the inner walls of the cell ledges at the ends of the aluminum electrolysis cell body, and the cathode horizontal partition plate is located in the electrolysis cell body and provided with molten aluminum through holes. According to the double-layer aluminum cathode aluminum electrolysis cell cathode, molten aluminum in which no cathode carbon block, no steel bar and no titanium boride coating exist is adopted as the cathode to replace a cathode composed of steel bars and a carbon block set. The defects that according to a traditional aluminum electrolysis cell, aluminum oxide deposit exists on cathode carbon blocks and cellular micro-batteries exist are eliminated. In the electrolysis production process, the problem that aluminum oxide and aluminum carbide which are deposited and generated at the furnace bottom pollute electrolysis molten aluminum does not exist.
Owner:XINJIANG UNIVERSITY