Patents
Literature
7939 results about "Electrolytic cell" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
An electrolytic cell is an electrochemical cell that drives a non-spontaneous redox reaction through the application of electrical energy. They are often used to decompose chemical compounds, in a process called electrolysis—the Greek word lysis means to break up.
Electroactive high storage capacity polyacetylene-co-polysulfur materials and electrolytic cells containing same
InactiveUS6117590AHigh storage capacity per unit weightFacilitates electron transportElectrode manufacturing processesNon-aqueous electrolyte accumulatorsElectrochemical cellElectrode material
The present invention relates to novel electroactive energy storing polyacetylene-co-polysulfur (PAS) materials of general formula (C2Sx)n wherein x is greater than 1 to about 100, and n is equal to or greater than 2. This invention also relates to novel rechargeable electrochemical cells containing positive electrode materials comprised of said polyacetylene-co-polysulfur materials with improved storage capacity and cycle life at ambient and sub-ambient temperatures.
Owner:THE BANK OF NEW YORK +1
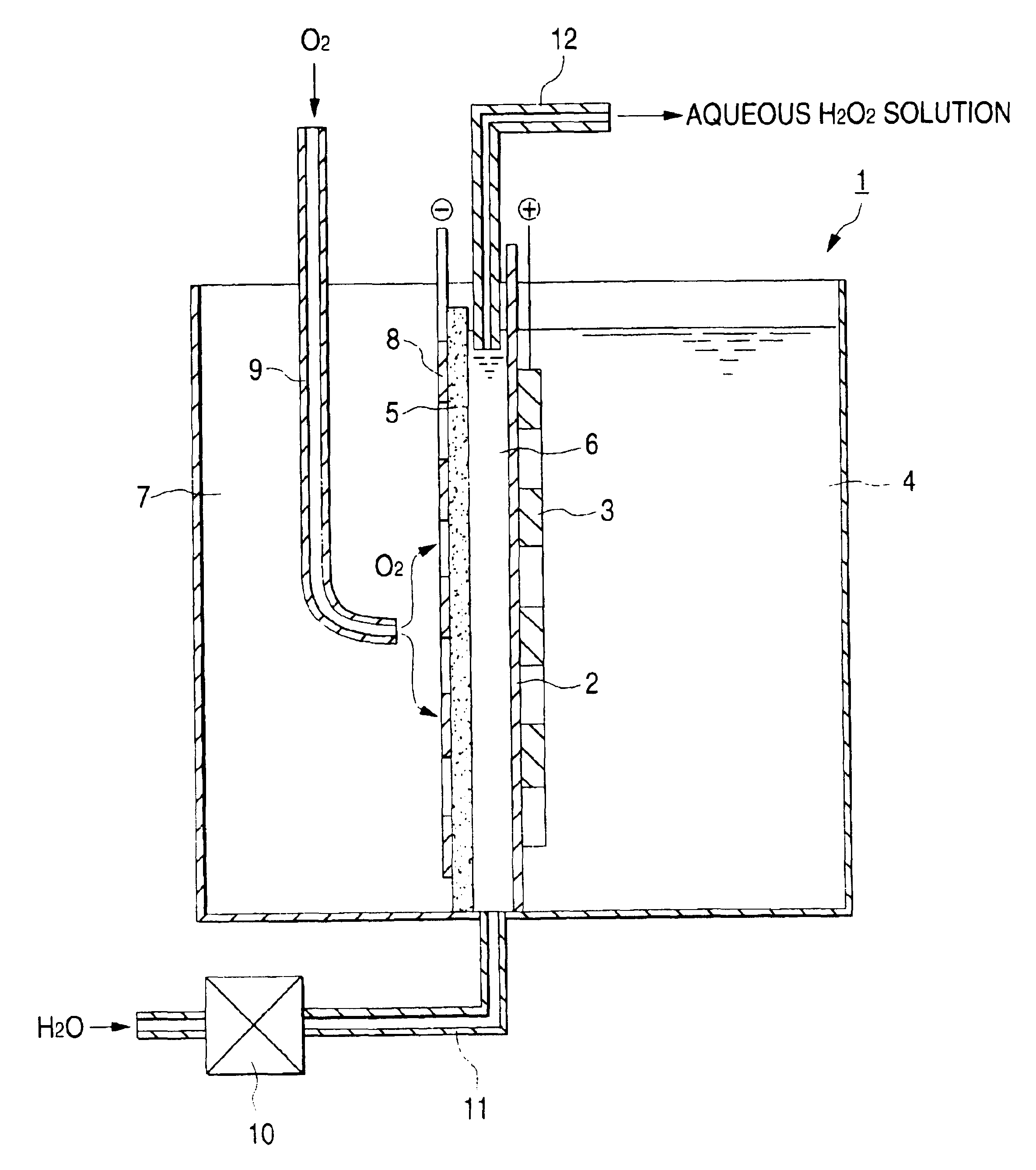
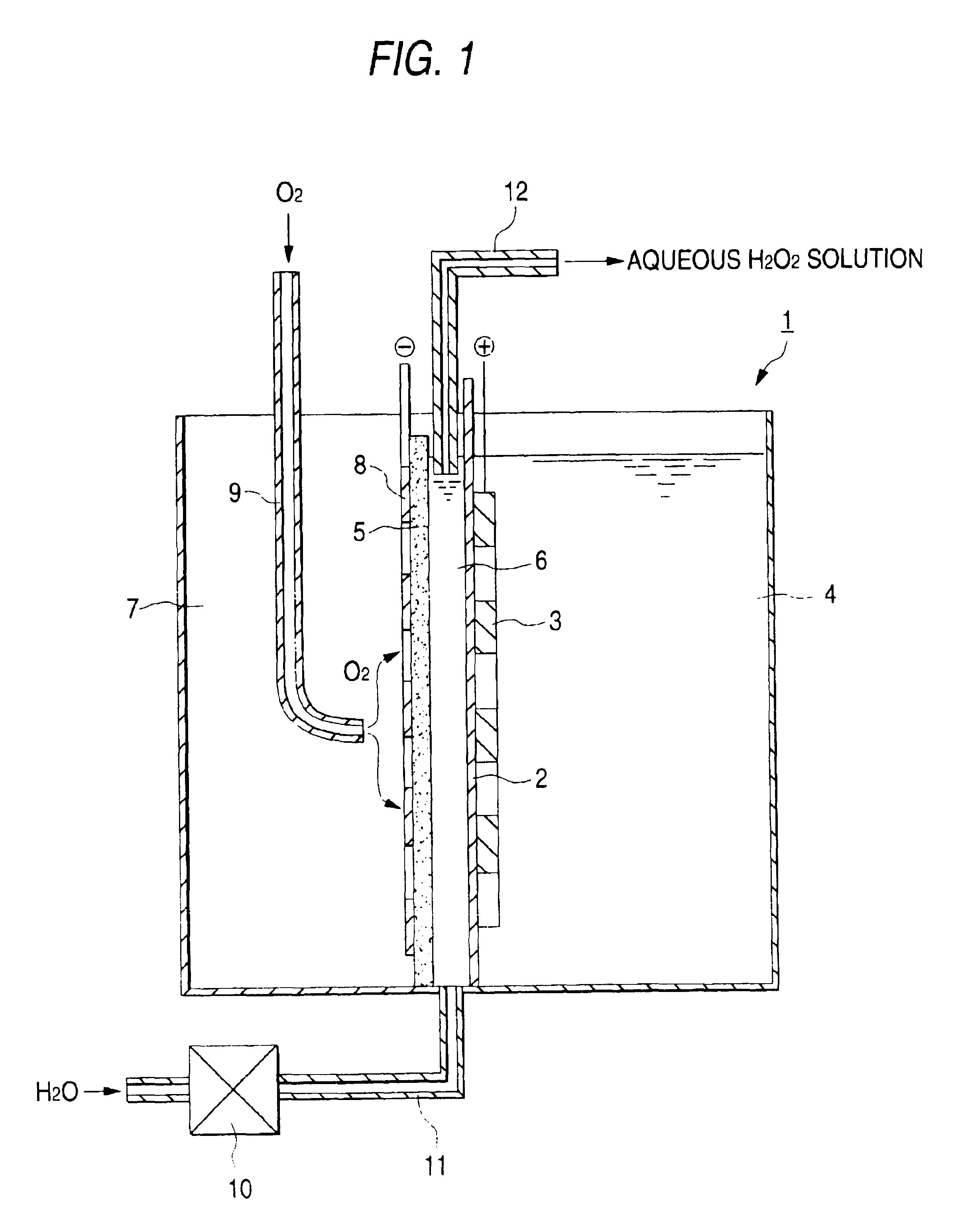
Electrolytic cell for hydrogen peroxide production and process for producing hydrogen peroxide
InactiveUS6767447B2CellsPeroxides/peroxyhydrates/peroxyacids/superoxides/ozonidesSodium sulfateNuclear chemistry
An electrolytic cell and method of electrolysis for producing hydrogen peroxide at a moderate current density while preventing metal deposition on the cathode surface. A feed water from which multivalent metal ions have been removed and in which a salt of a univalent metal, e.g., sodium sulfate, has been dissolved in a given concentration is prepared with an apparatus for removing multivalent metal ions and dissolving a salt in low concentration. The feed water is supplied to an electrolytic cell. Even when electrolysis is continued, almost no deposition of a hydroxide or carbonate occurs on the cathode because multivalent metal ions are not present in the electrolytic solution. Due to the dissolved salt, a sufficient current density is secured to prevent an excessive load from being imposed on the electrodes, etc. Thus, stable production of hydrogen peroxide is possible over a long period of time.
Owner:DE NORA PERMELEC LTD
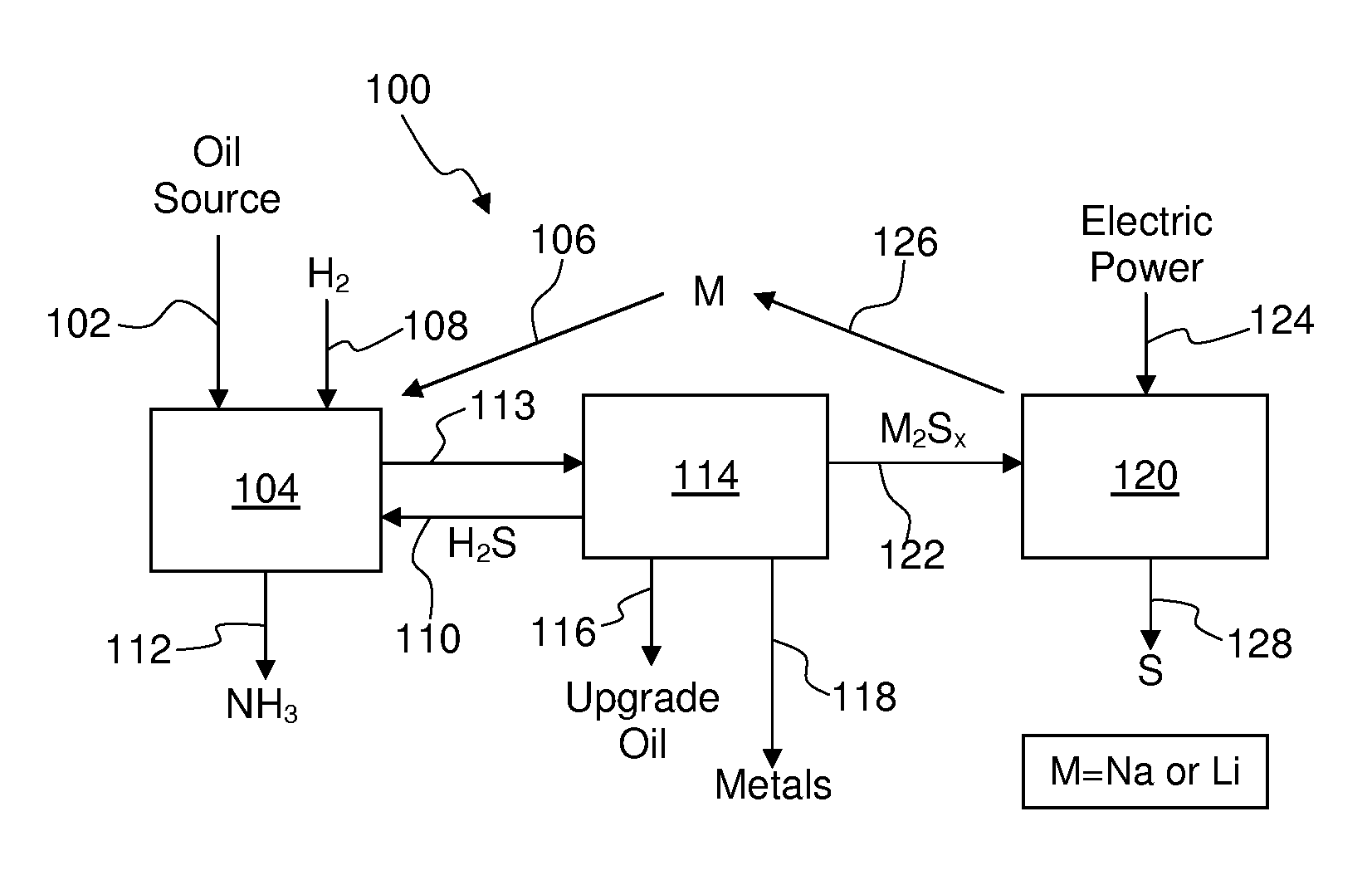
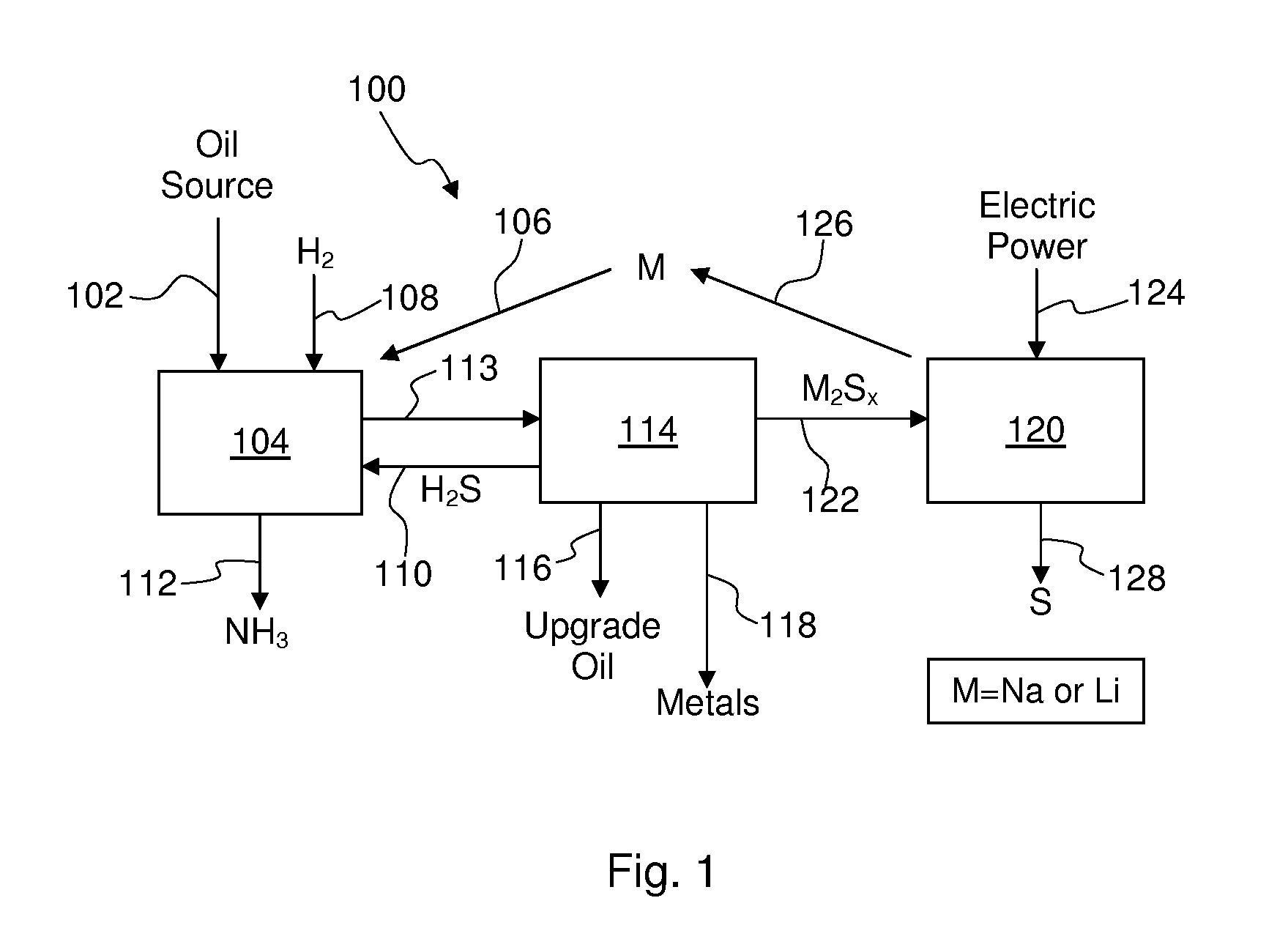
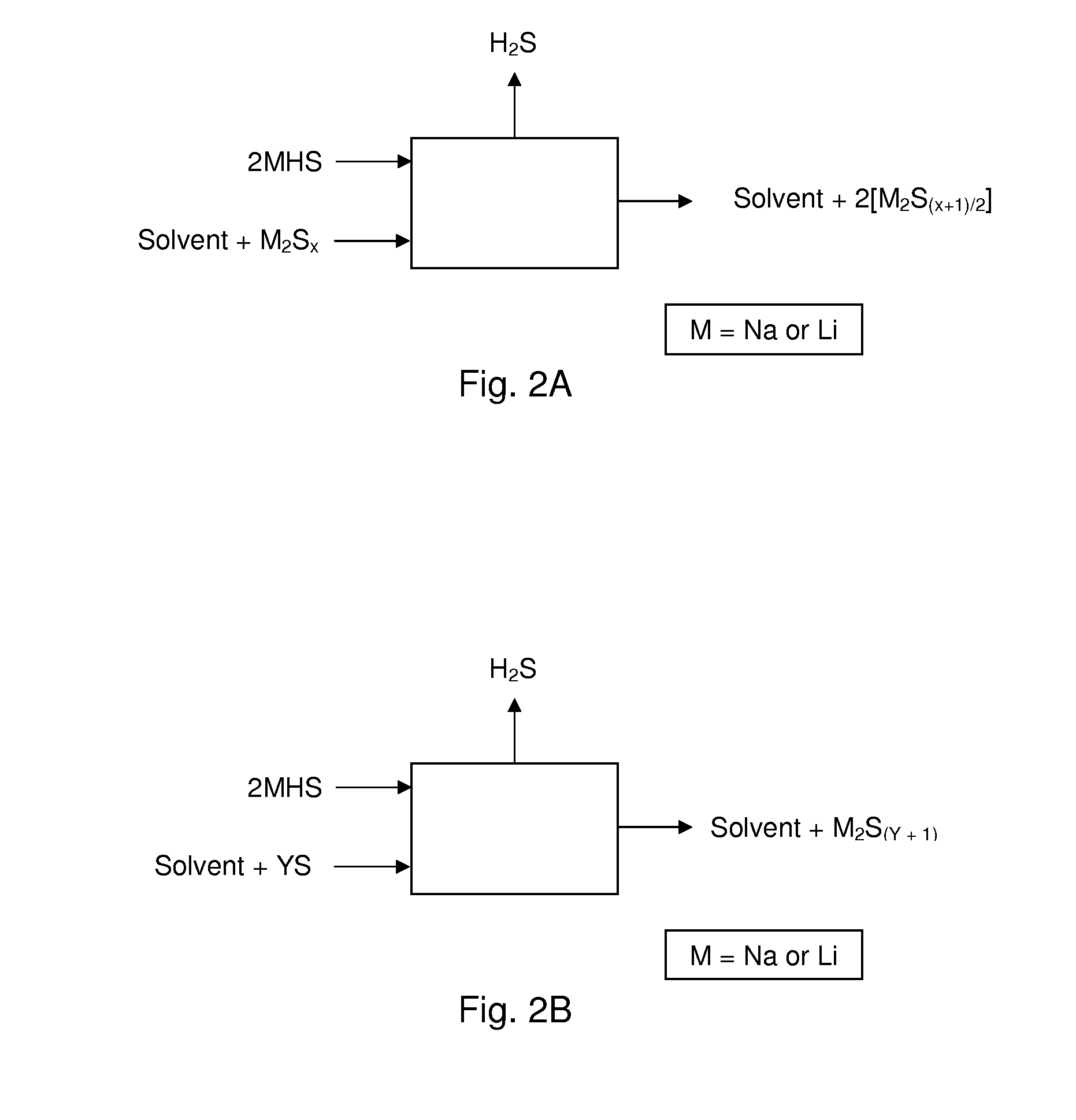
Process For Recovering Alkali Metals and Sulfur From Alkali Metal Sulfides and Polysulfides
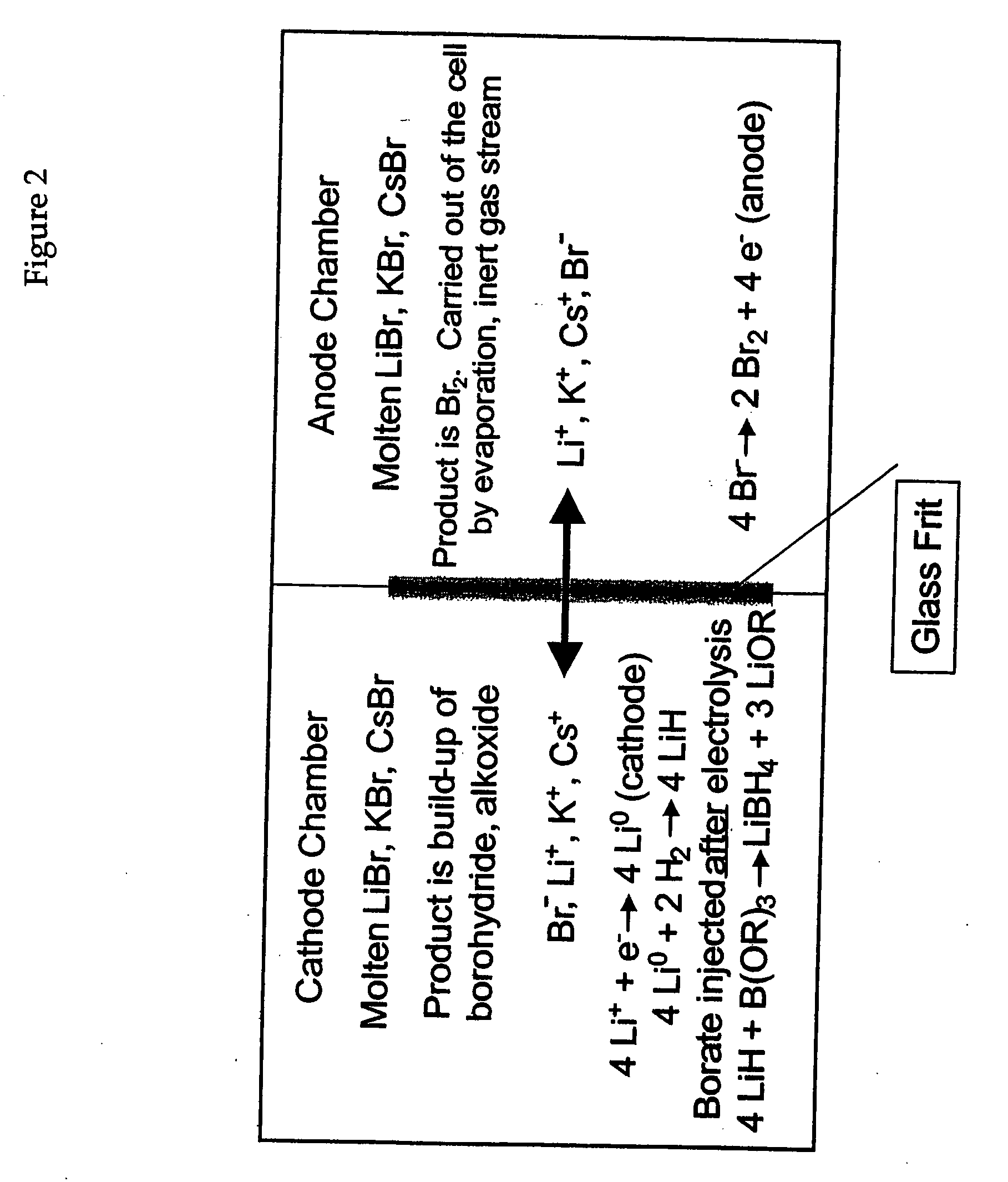
Alkali metals and sulfur may be recovered from alkali polysulfides in an electrolytic process that utilizes an electrolytic cell having an alkali ion conductive membrane. An anolyte solution includes an alkali polysulfide and a solvent that dissolves elemental sulfur. A catholyte solution includes alkali metal ions and a catholyte solvent. Applying an electric current oxidizes sulfur in the anolyte compartment, causes alkali metal ions to pass through the alkali ion conductive membrane to the catholyte compartment, and reduces the alkali metal ions in the catholyte compartment. Sulfur is recovered by removing and cooling a portion of the anolyte solution to precipitate solid phase sulfur. Operating the cell at low temperature causes elemental alkali metal to plate onto the cathode. The cathode may be removed to recover the alkali metal in batch mode or configured as a flexible band to continuously loop outside the catholyte compartment to remove the alkali metal.
Owner:ENLIGHTEN INNOVATIONS INC
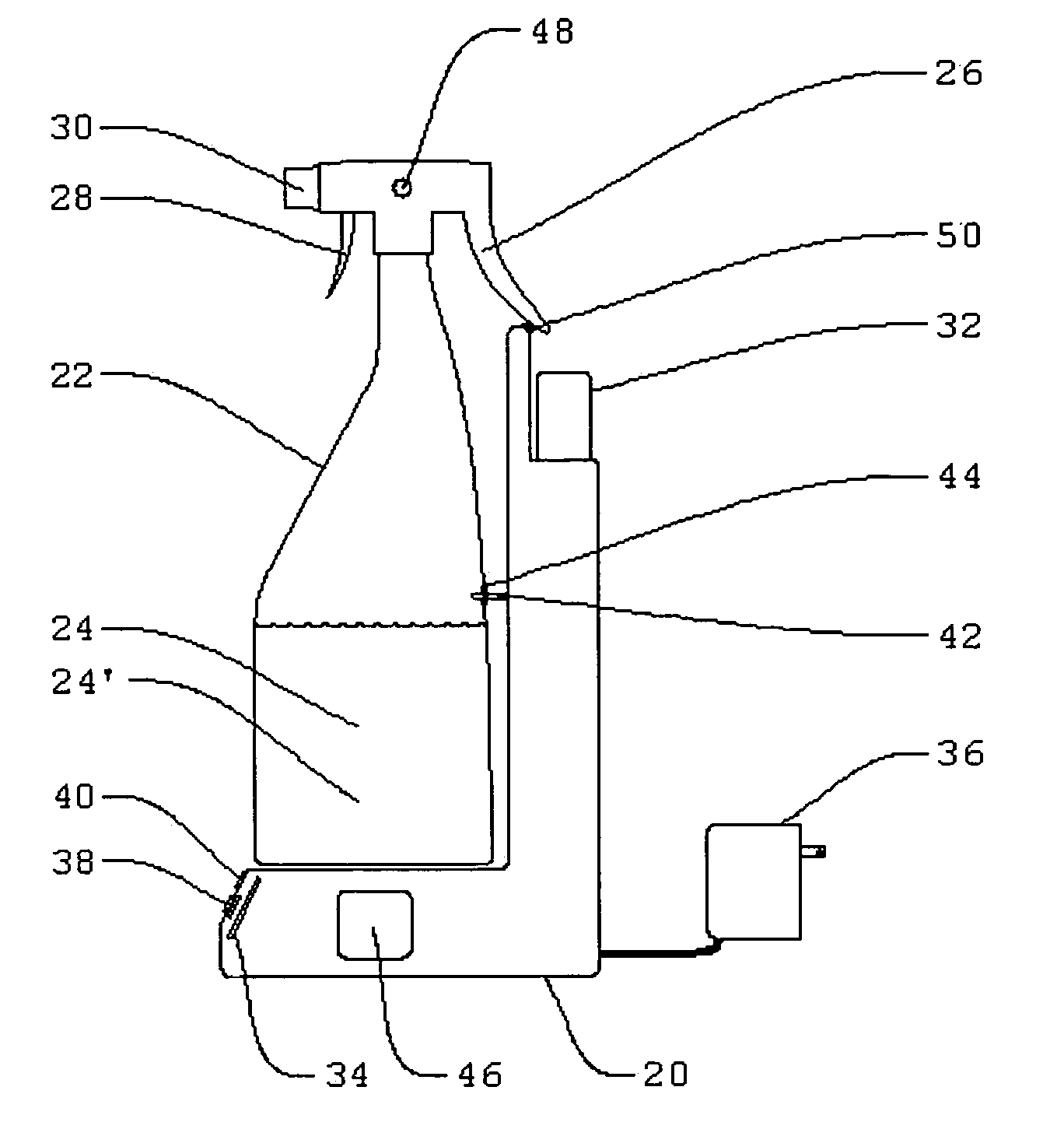
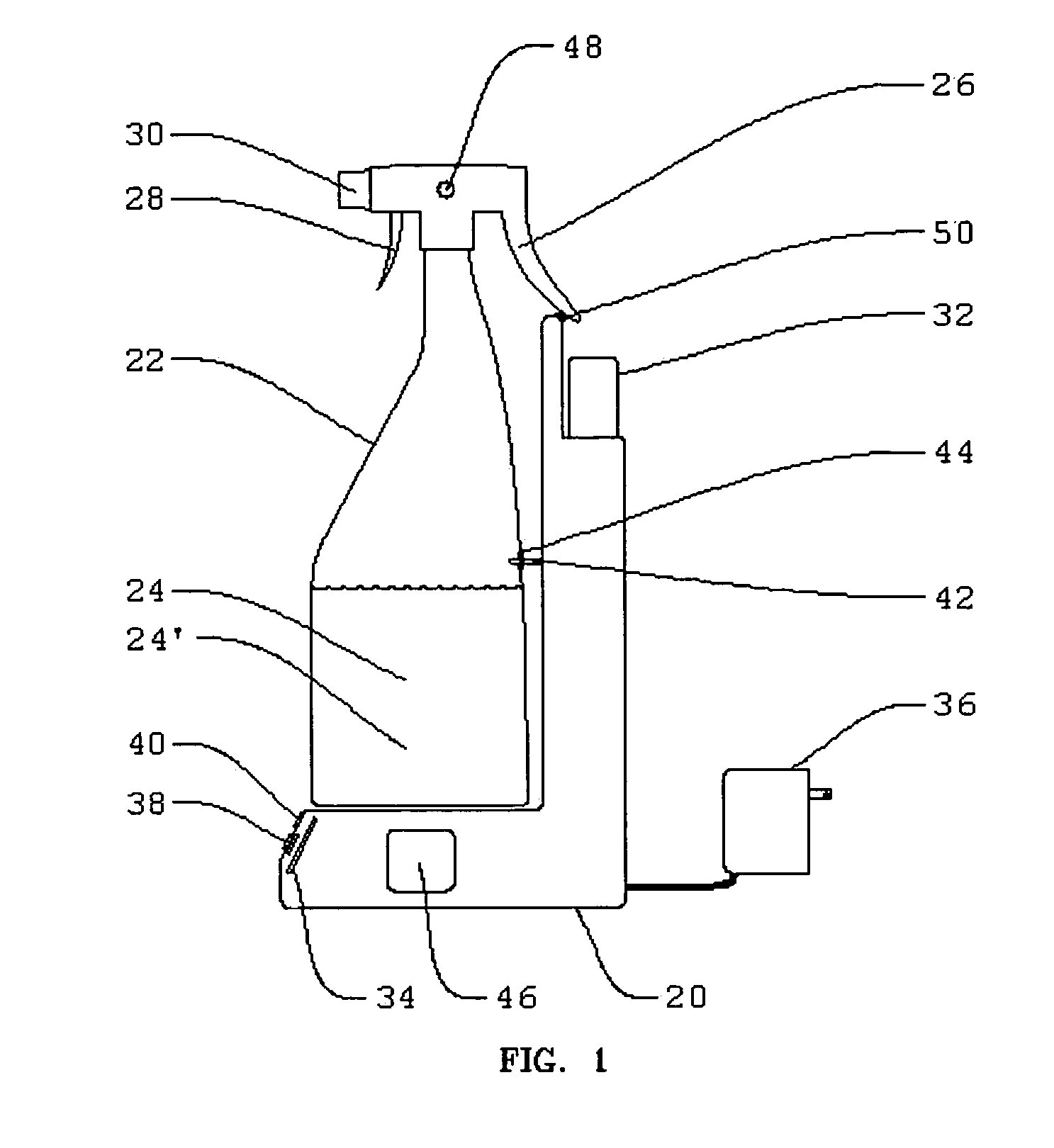
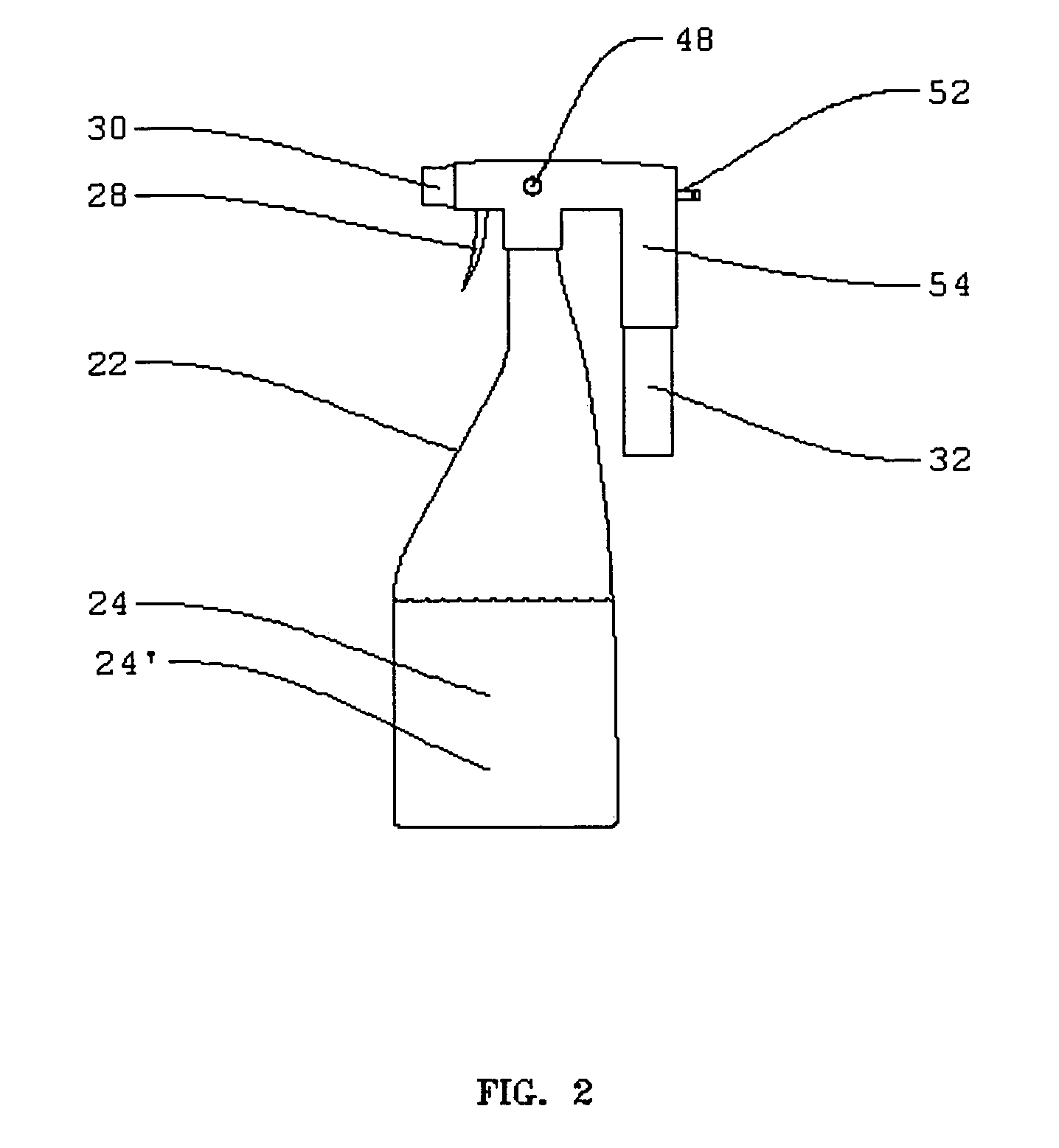
Electrolytic cell for surface and point of use disinfection
InactiveUS7008523B2Suitable for useCellsWater treatment parameter controlDisinfectantElectrical battery
The present invention is an apparatus and method for disinfecting or sanitizing a desired object. The apparatus includes a container for an aqueous solution; the container may be a spray bottle. The apparatus includes an electrolytic cell, containing an electrolyte, an electrical power source, a control circuit for providing an electric charge to the electrolyte to create an oxidant, and a fluid connection between the cell and container to permit introduction of the oxidant into the aqueous solution to create a disinfectant.
Owner:DE NORA HLDG US INC
Electric connection of electrochemical and photoelectrochemical cells
InactiveUS6657119B2Equipment is cheapReduce needLight-sensitive devicesPV power plantsPhotoelectrochemical cellEngineering
Owner:FORSKARPATENT I UPPSALA
Electrolytic method to make alkali alcoholates using ceramic ion conducting solid membranes
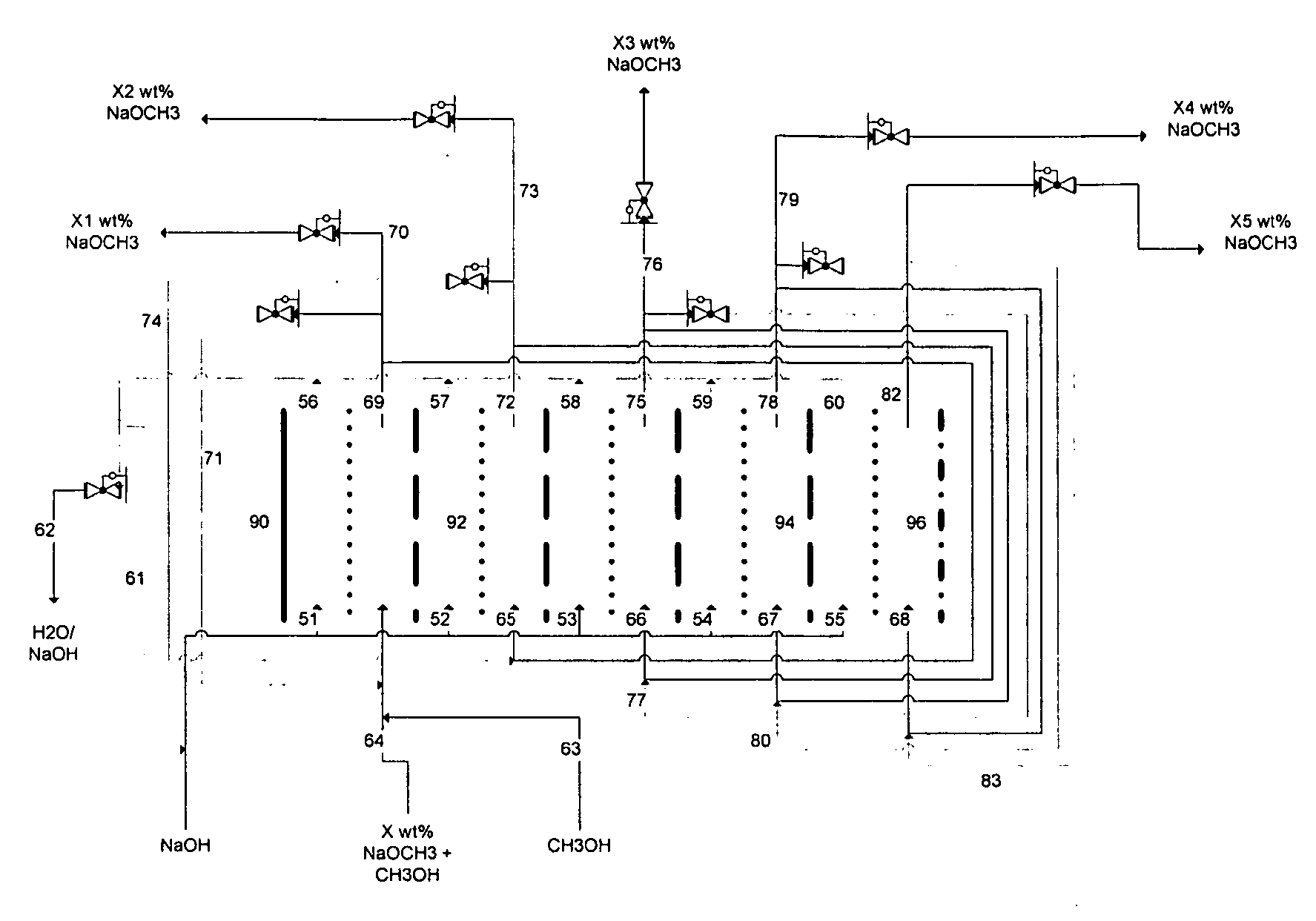
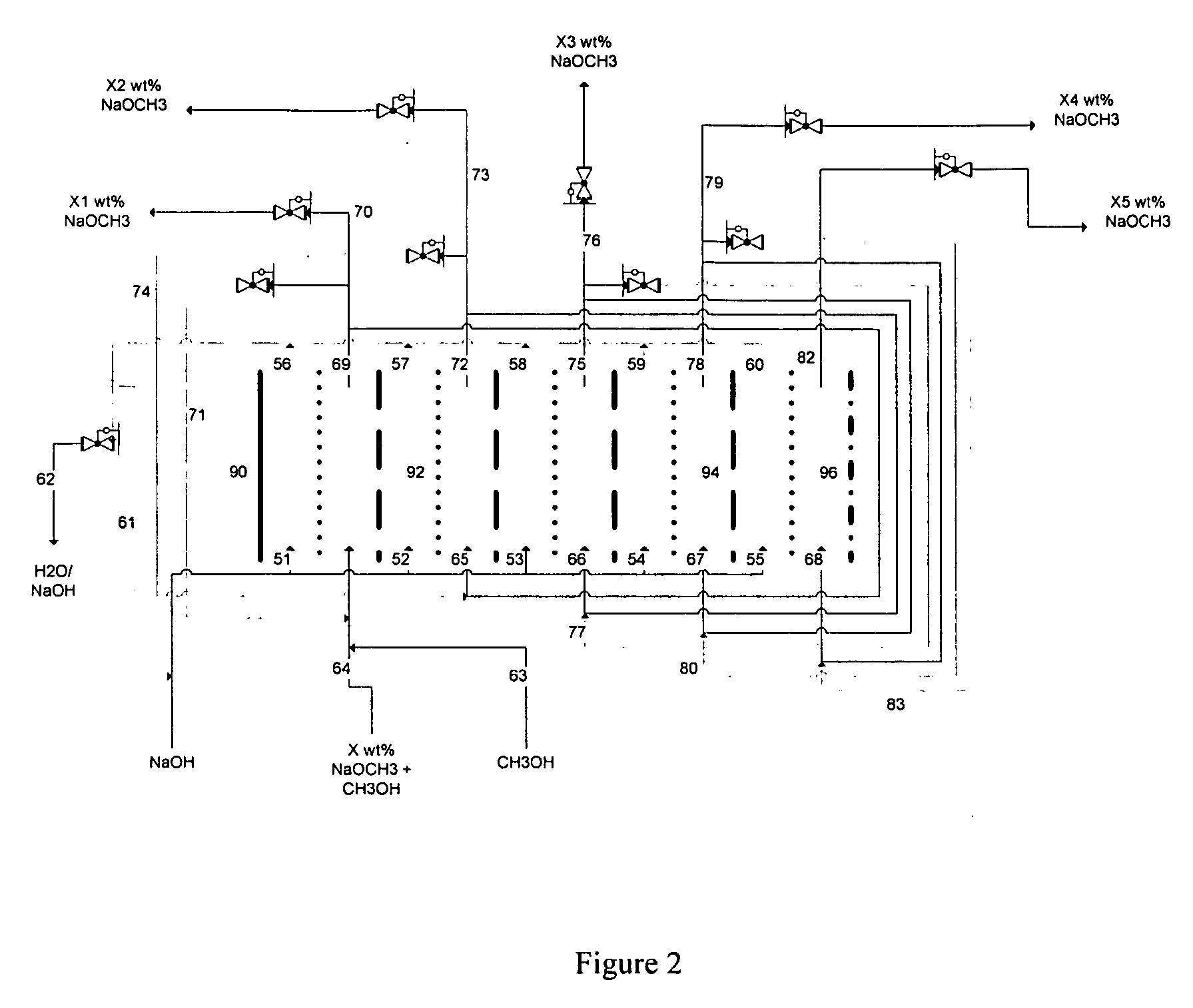
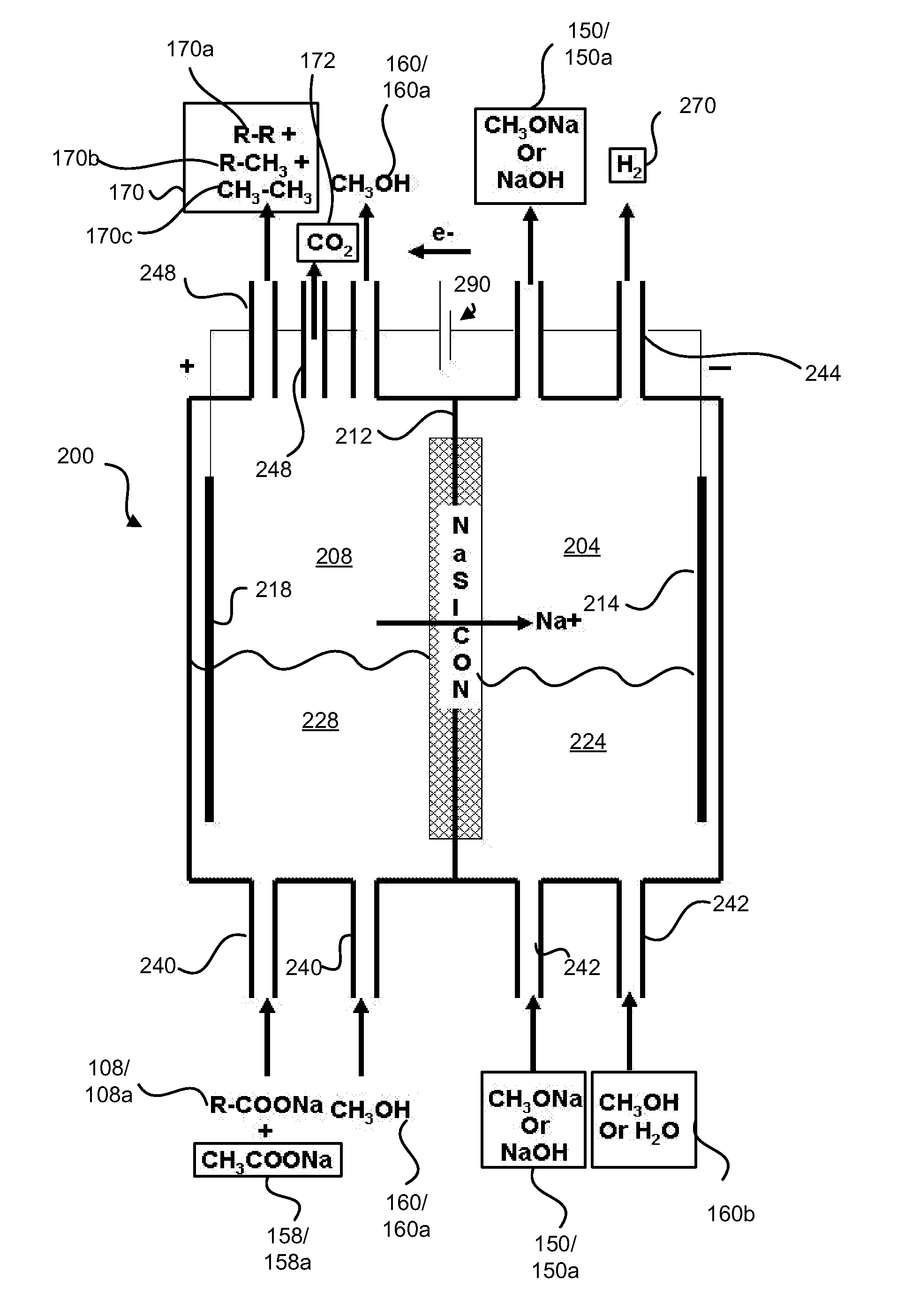
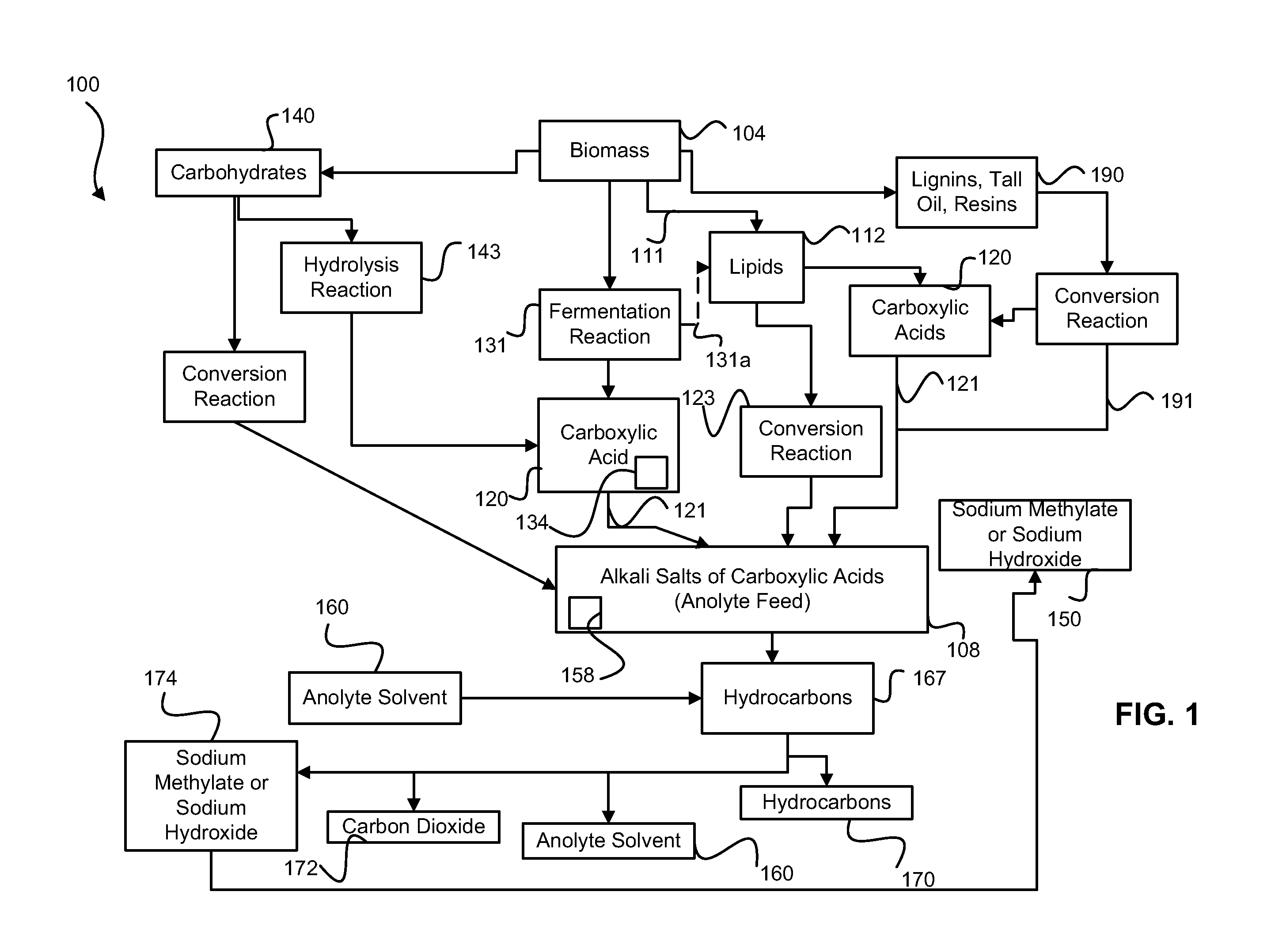
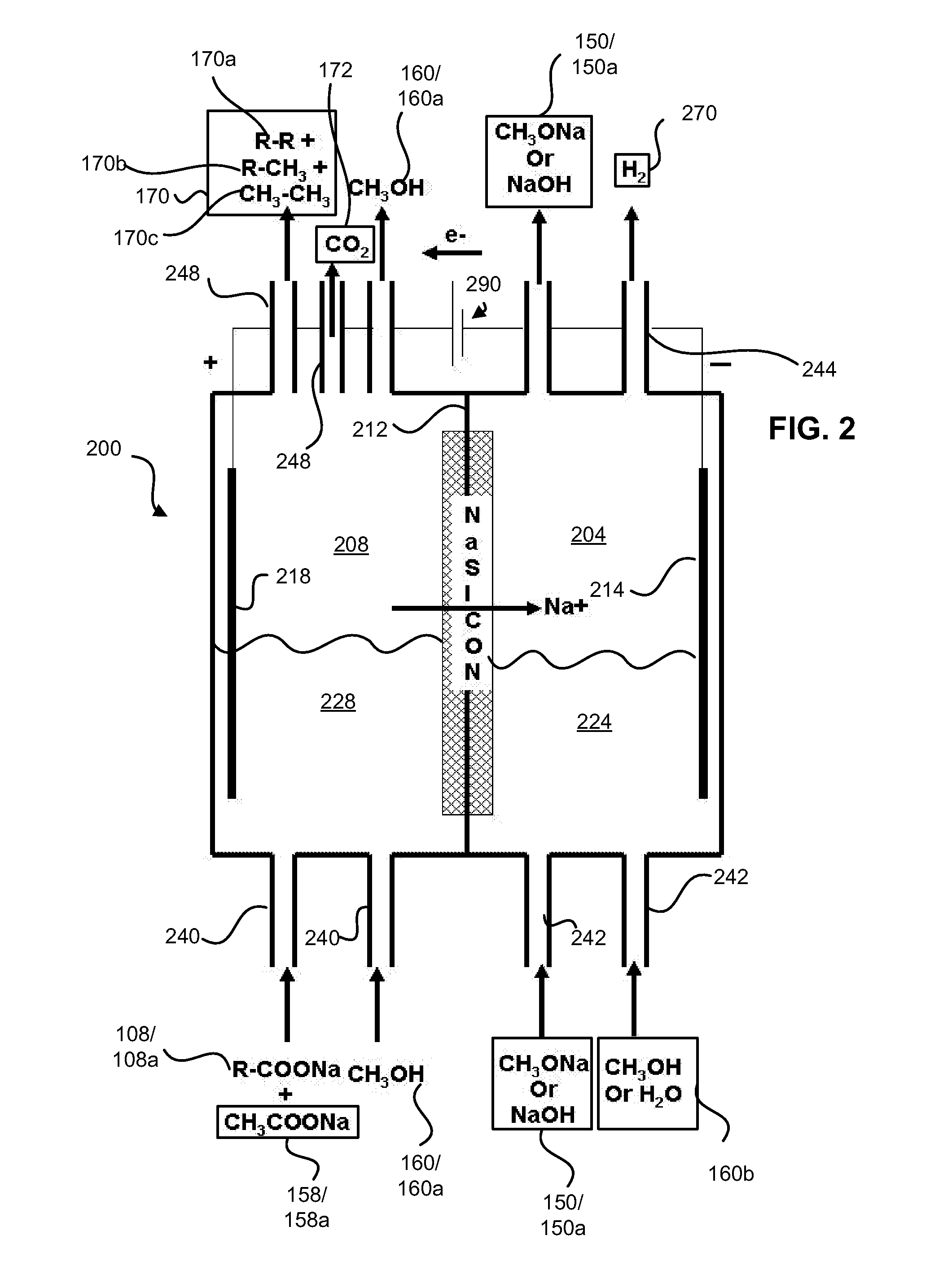
Disclosed are processes of making solutions of alkali alkoxides in their corresponding alcohols using an electrolytic process. In one embodiment, sodium methoxide in methanol is made from methanol and aqueous sodium hydroxide solution, where the aqueous sodium hydroxide solution is present in the anolyte compartment and a solution of sodium methoxide in methanol is present in the catholyte compartment, the two compartments are separated by a ceramic membrane that selectively transports sodium ions under the influence of an electric potential, and wherein the composition of the solution of sodium methoxide in methanol in the catholyte compartment of the electrolytic cell comprises between at least about 2% by weight sodium methoxide and at most about 20% by weight sodium methoxide.
Owner:ENLIGHTEN INNOVATIONS INC
NANO silicon-carbon composite material and preparation method thereof
ActiveUS20140302396A1Improve cycle stabilityShort production processMaterial nanotechnologyElectrolysis componentsCarbon compositesSilicon dioxide
The invention relates to a nano silicon-carbon composite negative material for lithium ion batteries and a preparation method thereof. A porous electrode composed of silica and carbon is taken as a raw material, and a nano silicon-carbon composite material of carbon-loaded nano silicon is formed by a molten salt electrolysis method in a manner of silica in-situ electrochemical reduction. Silicon and carbon of the material are connected by nano silicon carbide, and are metallurgical-grade combination, so that the electrochemical cycle stability of the nano silicon-carbon composite material is improved. The preparation method of the nano silicon-carbon composite material provided by the invention comprises the following steps: compounding a porous block composed of carbon and silica powder with a conductive cathode collector as a cathode; using graphite or an inert anode as an anode, and putting the cathode and anode into CaCl2 electrolyte or mixed salt melt electrolyte containing CaCl2 to form an electrolytic cell; applying voltage between the cathode and the anode; controlling the electrolytic voltage, the electrolytic current density and the electrolytic quantity, so that silica in the porous block is deoxidized into nano silicon by electrolytic reduction, and the nano silicon-carbon composite material for lithium ion batteries is prepared at the cathode.
Owner:CHINA AUTOMOTIVE BATTERY RES INST CO LTD
Electrolytic cell for producing charged anode water suitable for surface cleaning or treatment, and method for producing the same and use of the same
InactiveUS7090753B2Reduce the binding forceEasy to cleanCellsWater treatment parameter controlSurface cleaningElectrical battery
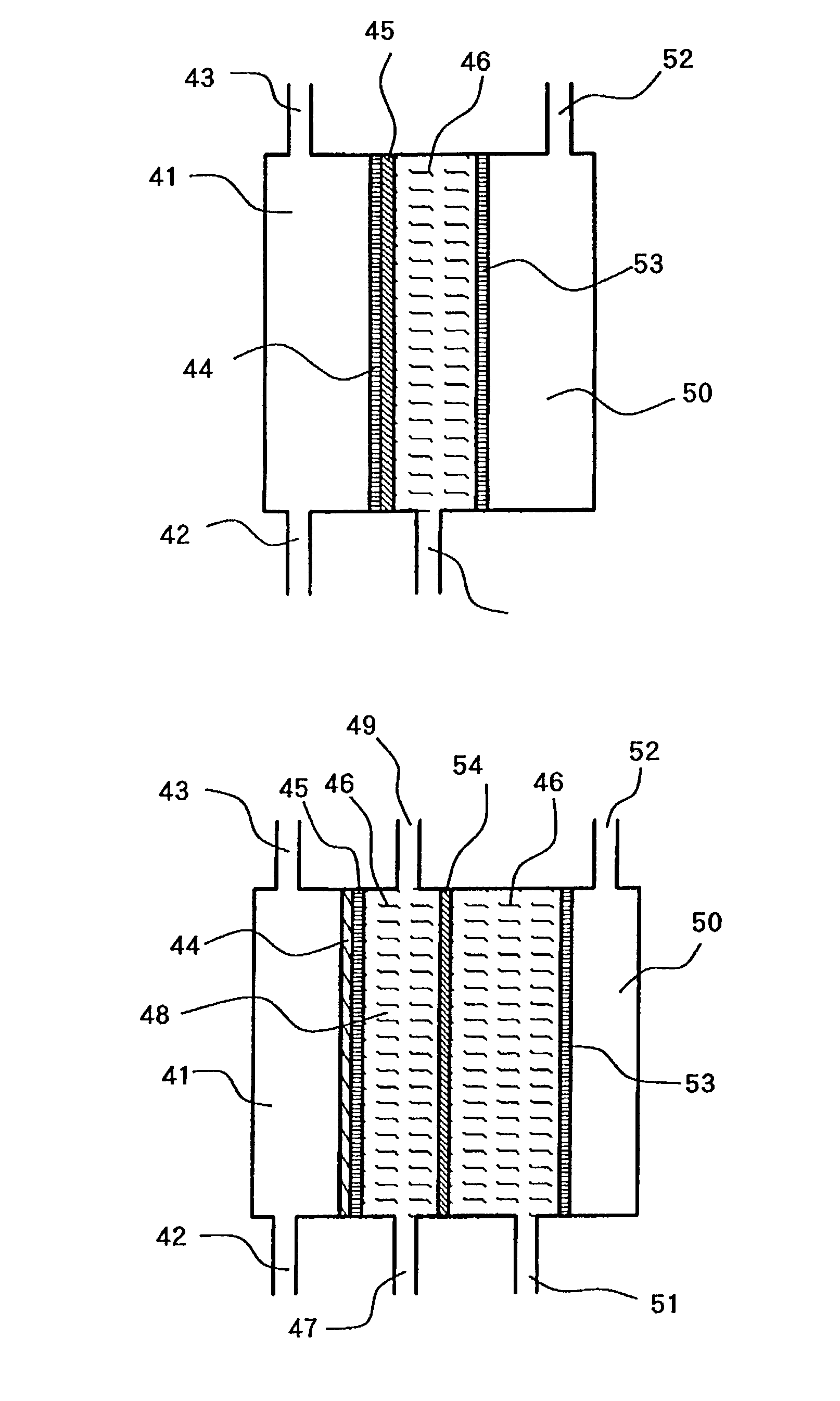
The present invention provides an electrolytic cell, which can efficiently produce, charged water having an excellent performance of improving surface cleaning or treatment of an object, e.g., semiconductor, glass, or resin and of cleaning and sterilizing medical device.The electrolytic cell of the present invention is for producing charged anode water suitable for surface cleaning or treatment, including the cathode chamber 41 and anode chamber 50, fluorinated cation-exchange membrane 46 provided to separated these chambers from each other, cathode 44 closely attaché to the cation-exchange membrane 45 on the side facing the cathode chamber 41, and middle chamber 48 filled with the cation-exchange resin 46, provided on the other side of The cation-exchange membrane 46, the cation-exchange resin 46 being arranged in such a way to come into contact with the fluorinated cation-exchange membrane 45, wherein the feed water is passed into the middle chamber 48 and passed thorough The anode chamber 50 to be recovered as the charged anode water.
Owner:SONOMA PHARMA INC
Flexible packaging for polymer electrolytic cell and method of forming same
InactiveUS6145280AEnergy Density MaximizationShrink the necessary spaceSmall-sized cells cases/jacketsPrimary cellsEngineeringMechanical engineering
Owner:NGK SPARK PLUG CO LTD
Constituting method for metal ordered structure surface reinforced base
The invention relates to a method to construct ordered structure on base, and the application of using the base to construct metal ordered structure. The method includes the following steps: adopting inorganic base or polymer base, taking base surface process to make it could conduct electricity, construct ordered microstructure of different functional group or macromolecule barrier layer; assembling the metal nanometer particle into the ordered structure to gain metal nanometer ordered structure. The invention could be widely used in making high sensitive metal sensor and detector.
Owner:JILIN UNIV
Method for preparing formic acid through electrochemical catalytic reduction of carbon dioxide
InactiveCN102190573AGood solubility and absorption propertiesImprove conductivityOrganic compound preparationCarboxylic compound preparationSupporting electrolyteOrganic solvent
The invention relates to a method for preparing formic acid through electrochemical catalytic reduction of carbon dioxide, and belongs to the technical field of carbon dioxide recycling. In the method, a proton exchange membrane separates an electrolytic tank into a cathode chamber and an anode chamber, organic solvent / ionic liquid / water mixed solution in which a large amount of carbon dioxide is dissolved is injected into the cathode chamber, and aqueous solution containing supporting electrolyte is injected into the anode chamber; and after an electrolysis power supply is connected, the carbon dioxide undergoes electroreduction reaction on the cathode to form the formic acid. By the method, the organic solvent / ionic liquid / water mixed solution with the advantages of good conductivity, low viscosity, high capacity of dissolving the carbon dioxide, wide electrochemical window, and low use cost can be obtained, and when the carbon dioxide is electrically reduced in the mixed solution, the current density in the electroreduction reaction of the carbon dioxide can be improved and the electrocatalytic activity and long-time stability of a cathode material are improved.
Owner:KUNMING UNIV OF SCI & TECH
System and Process for Treatment and De-halogenation of Ballast Water
A system and process for de-halogenating ballast water before releasing the ballast water from the vessel. In one embodiment, the system comprises a means for measuring the halogen content of the ballast water, a reducing agent source in fluid communication with the ballast water, and a means for controlling the amount of reducing agent supplied to the ballast water. In one aspect, the means for measuring the halogen content comprises one or more oxidation / reduction potential analyzers. In another embodiment, the system comprises one or more hypochlorite electrolytic cells for generating hypochlorite to treat the ballast water.One embodiment of the process for de-halogenating ballast water comprises measuring the oxidation / reduction potential of the ballast water and adding one or more reducing agents to the ballast water to de-halogenate the ballast water in response to the measured oxidation / reduction potential. In one aspect, the oxidation / reduction potential is modulated so that excess reducing agent is present in the ballast water.
Owner:DE NORA WATER TECH
Method for preparing vanadium battery solution or adjusting capacity and special device thereof
InactiveCN101619465ARapid responseContinuous operationElectrolysis componentsRegenerative fuel cellsVanadium CompoundsOxygen
The invention relates to a method for preparing vanadium battery solution or adjusting the capacity, wherein an adopted electrolysis unit comprises an electrolytic cell group, an anode electrolyte storage tank, a cathode electrolyte storage tank, a liquid conveying pipeline and a pump, a mode of forced convection is adopted so that the anode electrolyte and the cathode electrolyte stored in the anode electrolyte storage tank and the cathode electrolyte storage tank respectively flow through the anode and the cathode of the electrolytic cell group respectively, a voltage is applied between the anode and the cathode of the electrolytic cell group to generate direct current capable of generating oxygen and deoxidizing a vanadium compound, and after finishing electrochemical oxidation and reduction reaction, the anode electrolyte and the cathode electrolyte flowing through the anode and the cathode of the electrolytic cell group return to the anode electrolyte storage tank and the cathode electrolyte storage tank. The electrolysis method is applied to preparing electrolyte for a full vanadium liquid stream battery and restoring the capacity of the full vanadium liquid stream battery off line or on line.
Owner:DALIAN RONGKE ENERGY STORAGE GRP CO LTD
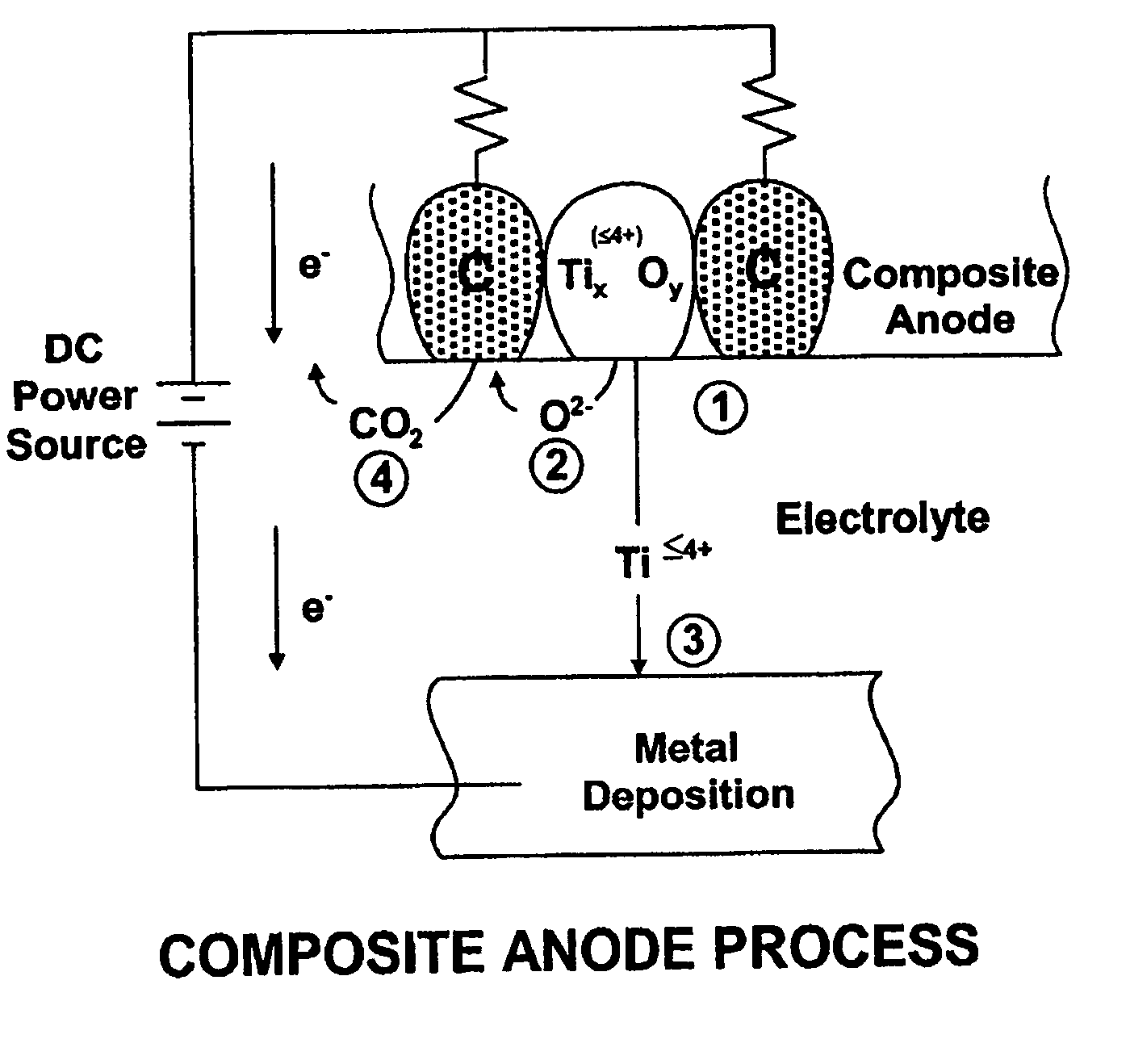
Thermal and electrochemical process for metal production
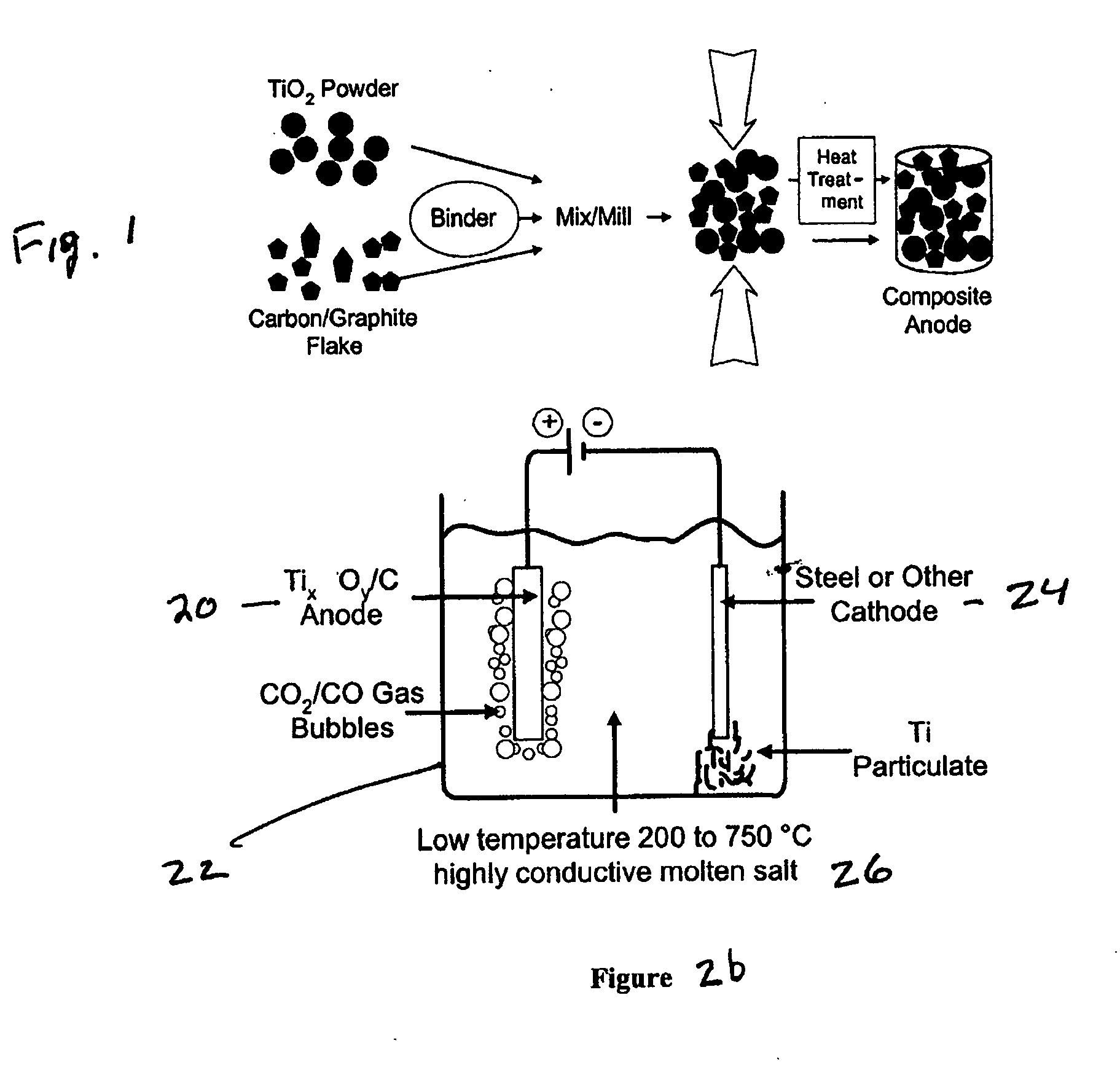
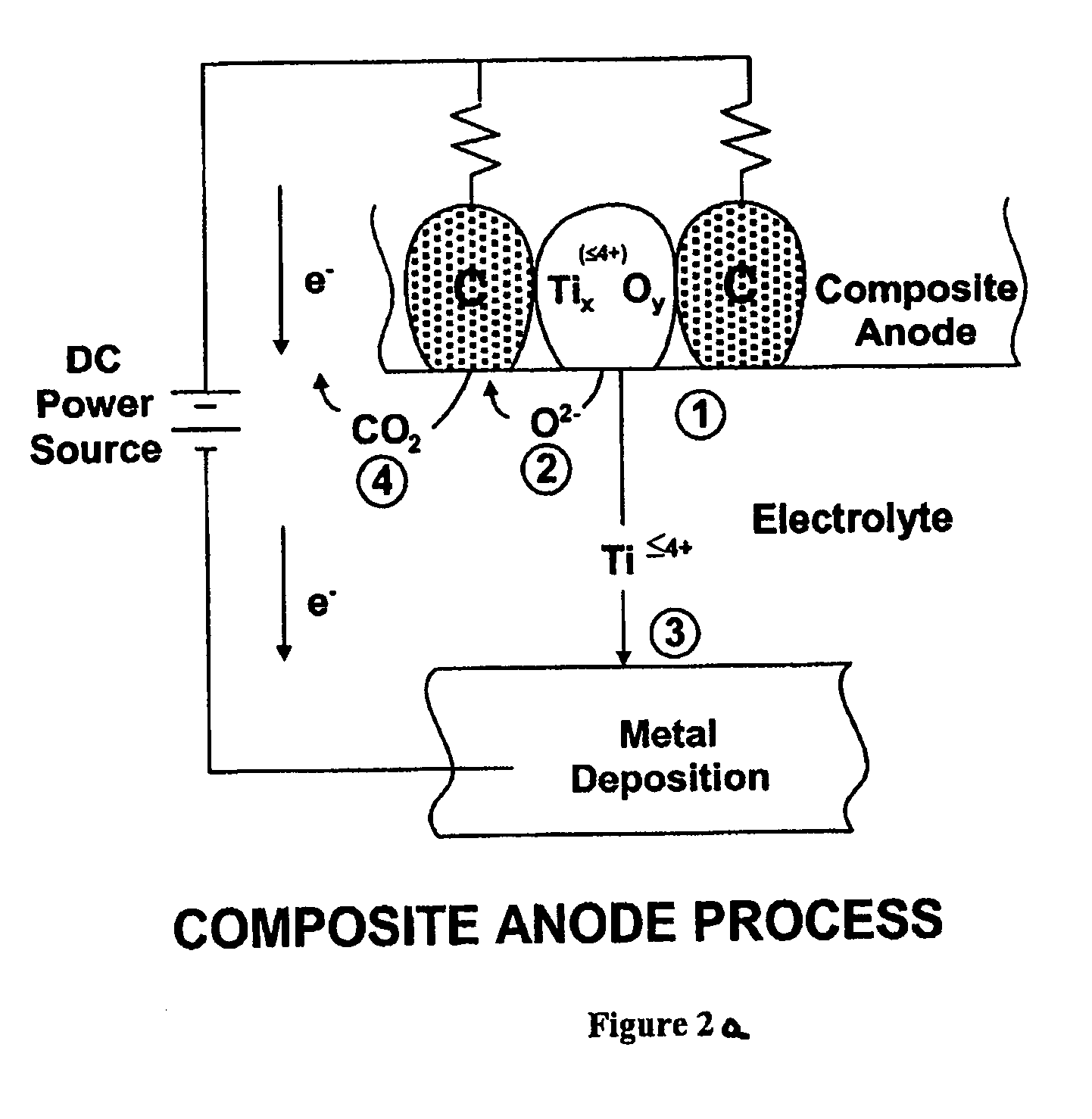
A system for purification of high value metals comprises an electrolytic cell in which an anode formed of a composite of a metal oxide of the metal of interest with carbon is electrochemically reduced in a molten salt electrolyte.
Owner:ATS MER LLC
Methods and apparatus for synthesis of metal hydrides
An electrochemical process and apparatus for preparing metal hydride compounds from metal salts under a hydrogen atmosphere are disclosed. The electrochemical process may be integrated with chemical reaction of a boron compound to produce borohydride compounds. A metal salt and a borate are charged to the cathode of an electrolytic cell wherein the borate reacts with the hydride, to produce the borohydride compound.
Owner:MILLENNIUM CELL
Decarboxylation cell for production of coupled radical products
InactiveUS20110024288A1Avoid mixingLow costCellsElectrolytic organic coupling reactionsAlkali ionsTG - Triglyceride
Owner:CERAMTEC
Electrolyzer
An ozone generator which operates at constant pressures to produce a continuous flow of ozone in an oxygen stream having from 10% to 18% by weight of ozone. The ozone generator includes one or more electrolytic cells comprising an anode / anode flowfield, a cathode / cathode flowfield, and a proton exchange medium for maintaining the separation of ozone and oxygen from hydrogen. The ozone generator also has an anode reservoir which vents oxygen and ozone and a cathode reservoir which vents hydrogen. The anode reservoir can be filled from the cathode reservoir while continuing to produce ozone. The ozone generator is readily configured for self-control using a system controller programmed to operate the anode reservoir at a constant pressure.
Owner:LYNNTECH
System and method for treatment of ballast water
ActiveUS20060113257A1Prevent outbreakLiquid separation by electricityAuxillariesHypochloriteHydrogen
A system and method for treating ballast water within an ocean going vessel by generating hypochlorite for treating the ballast water. The system comprises one or more hypochlorite electrolytic cells in fluid communication with a stream of ballast water. A chlorine analyzer is positioned downstream from the electrolytic cells to determine the chlorine concentration of the treated ballast water. A hydrogen separator is connected to the hypochlorite electrolytic cells for venting hydrogen. In the method of this invention, water is taken aboard the ship for ballast in one port. A treatment stream is separated from the ballast water stream and piped to hypochlorite electrolytic cells. Hypochlorite is generated into the treatment stream and the hydrogen byproduct is separated by the hydrogen separators. The treatment stream is then reintroduced to the ballast water to eliminate marine species and pathogenic bacteria from ballast water. The ballast water undergoes de-chlorination prior to being discharged into a new port.
Owner:DE NORA WATER TECH
Water supply system for an aircraft
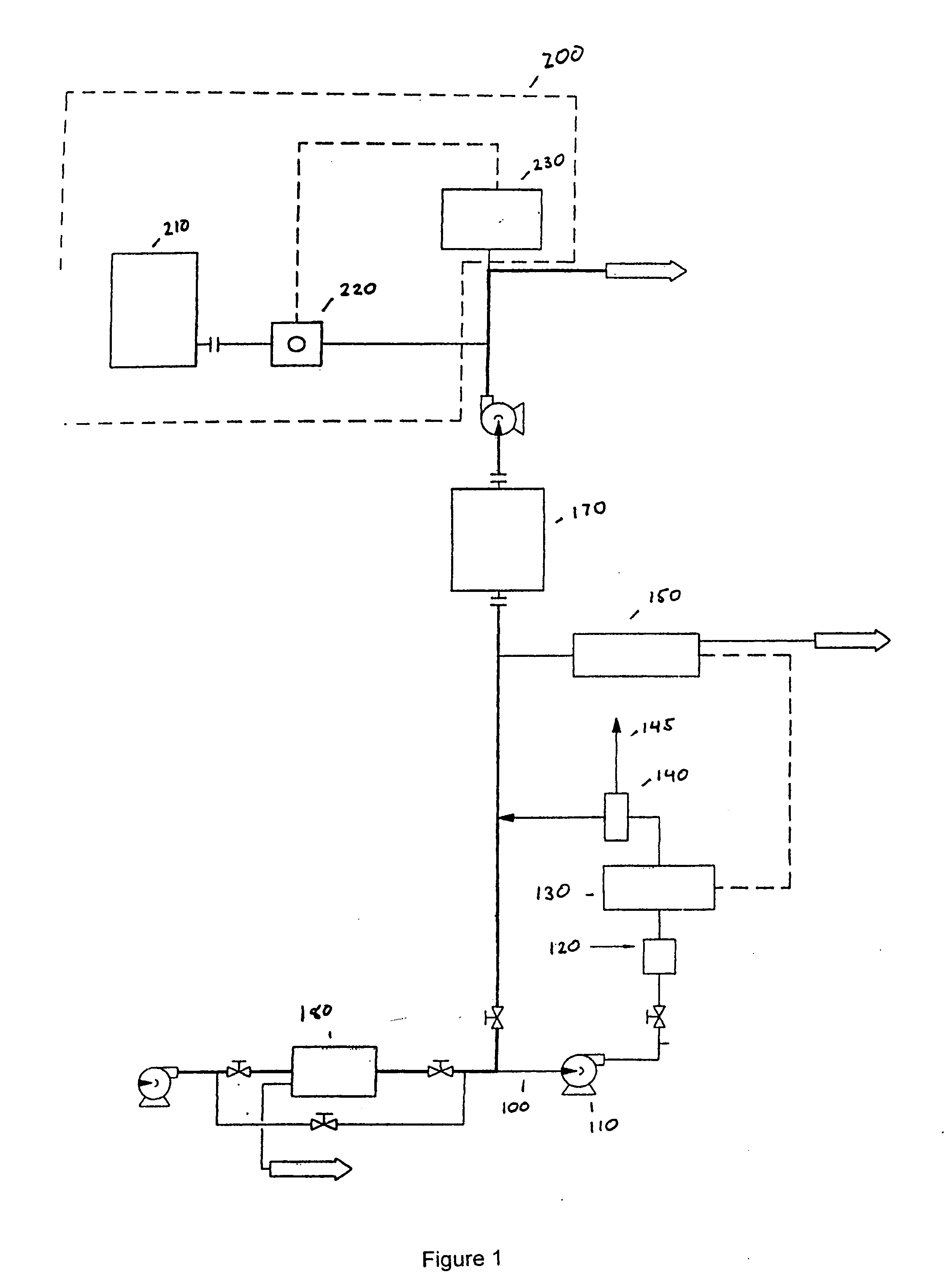
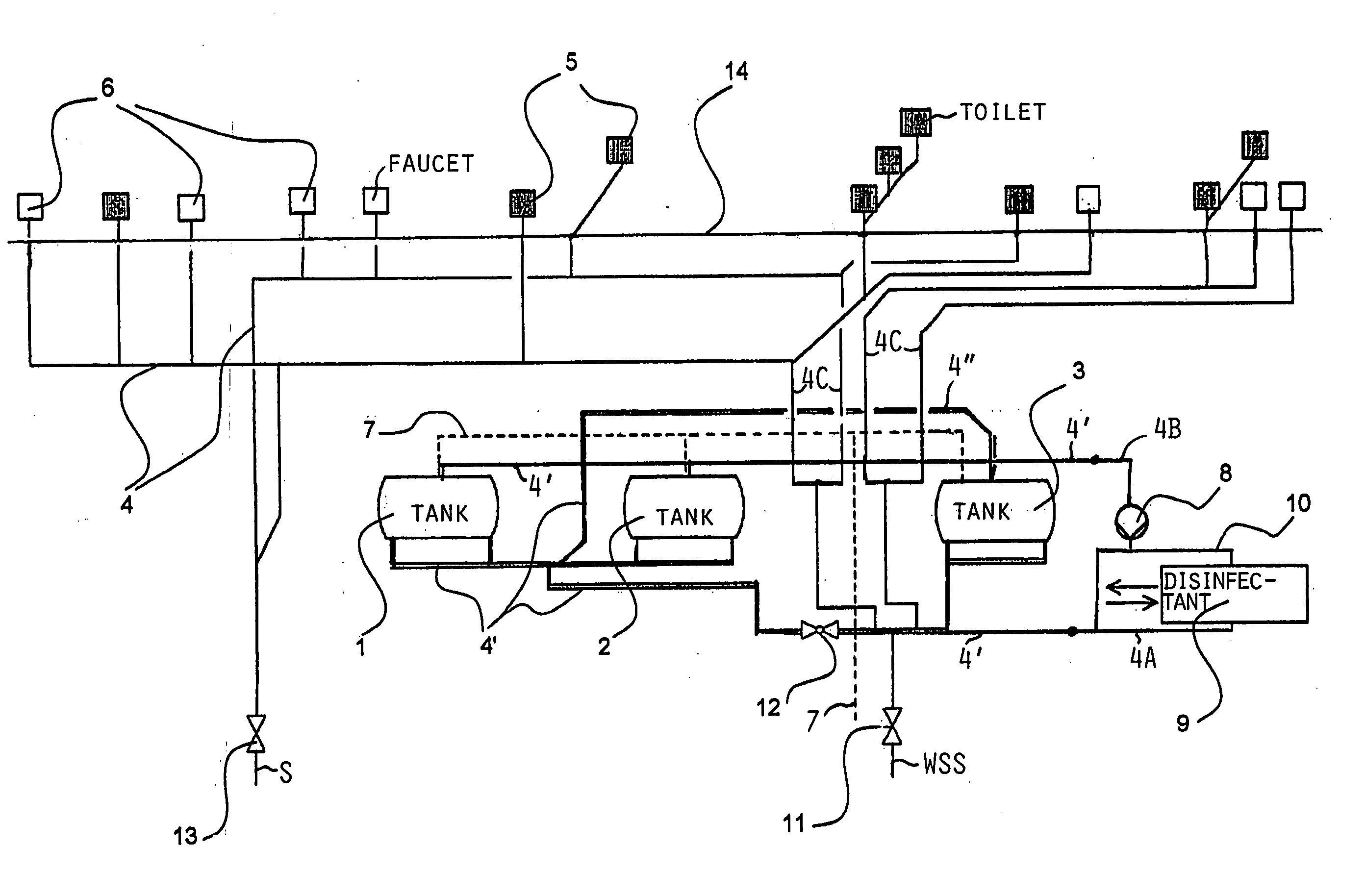
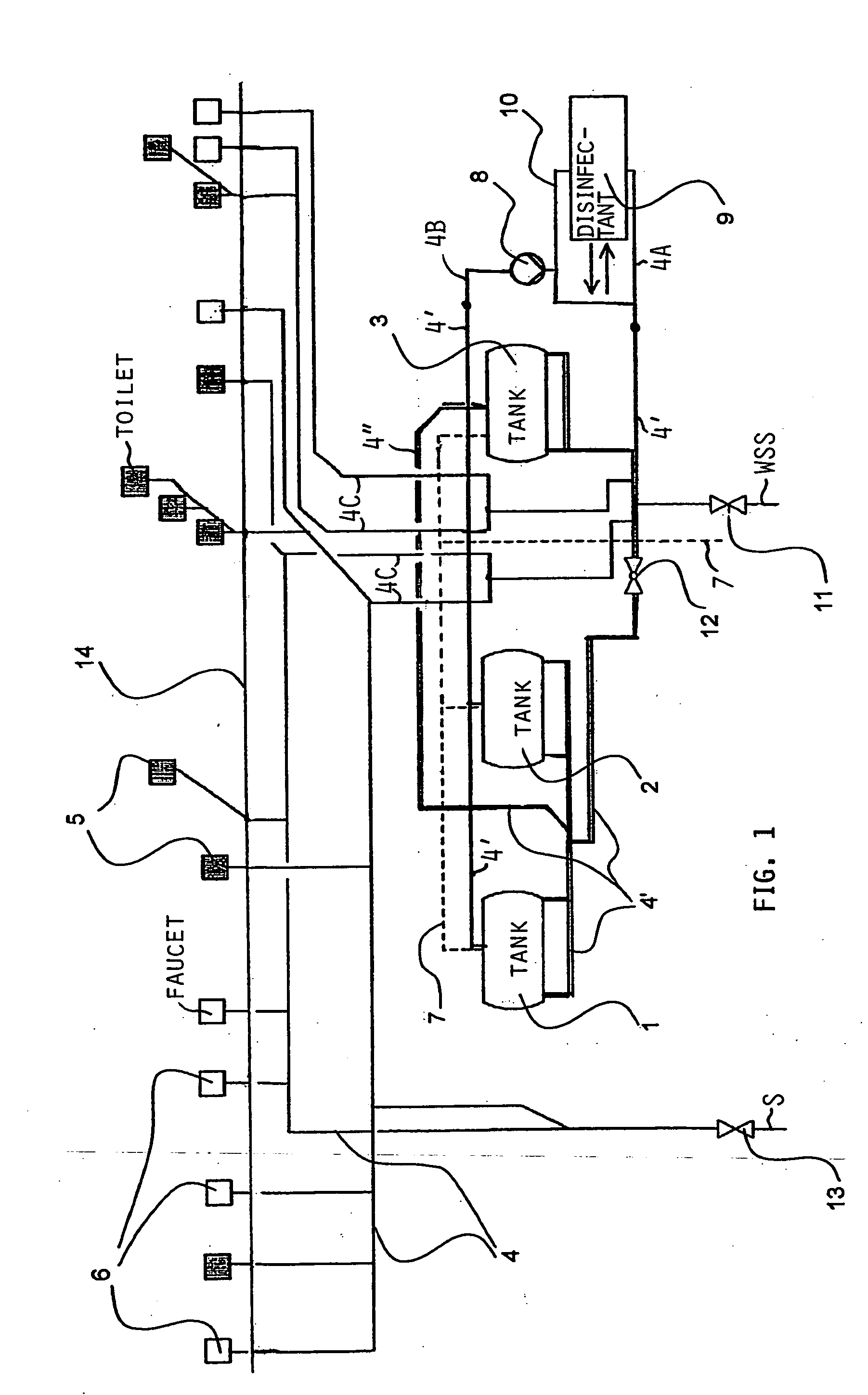
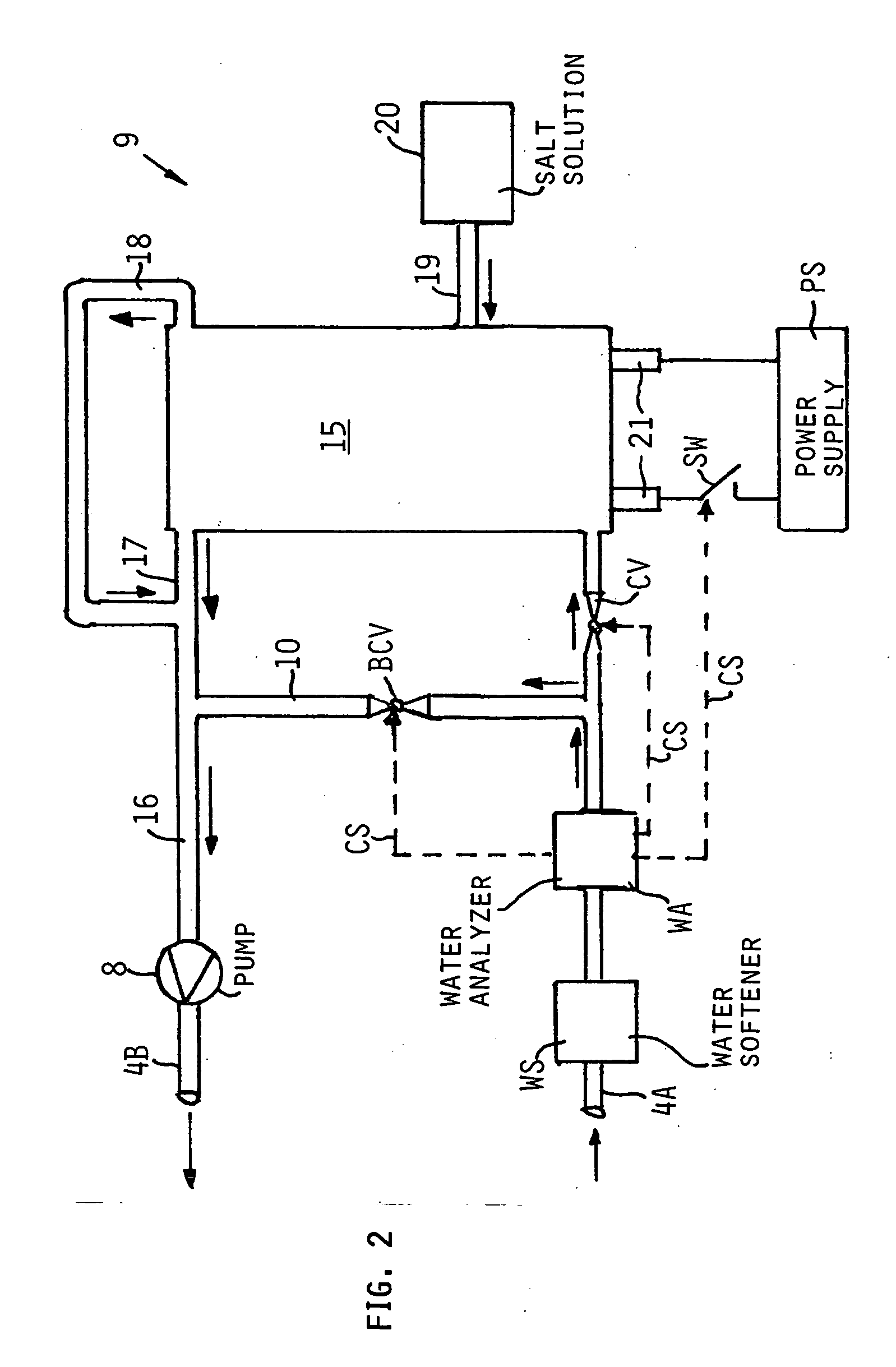
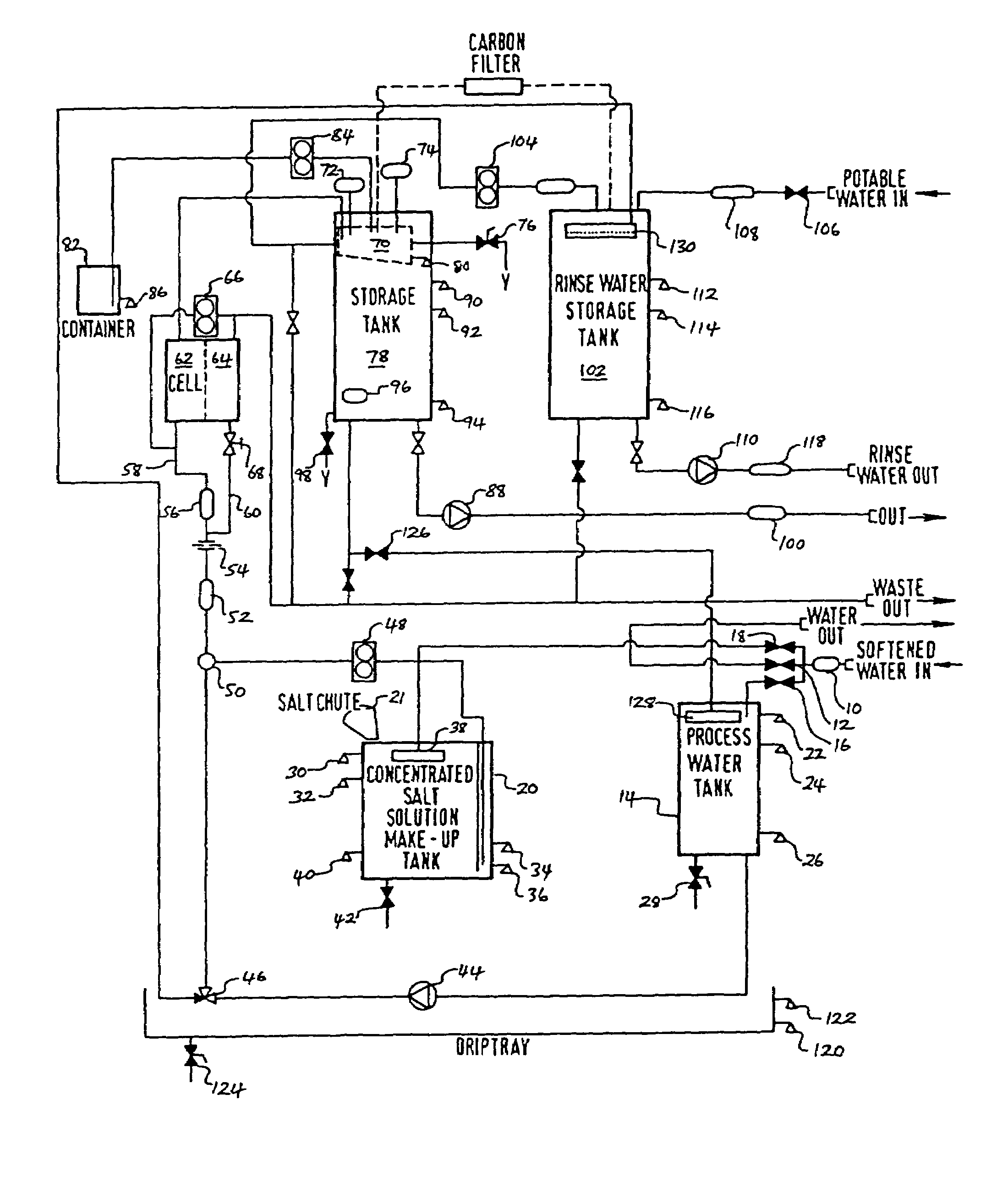
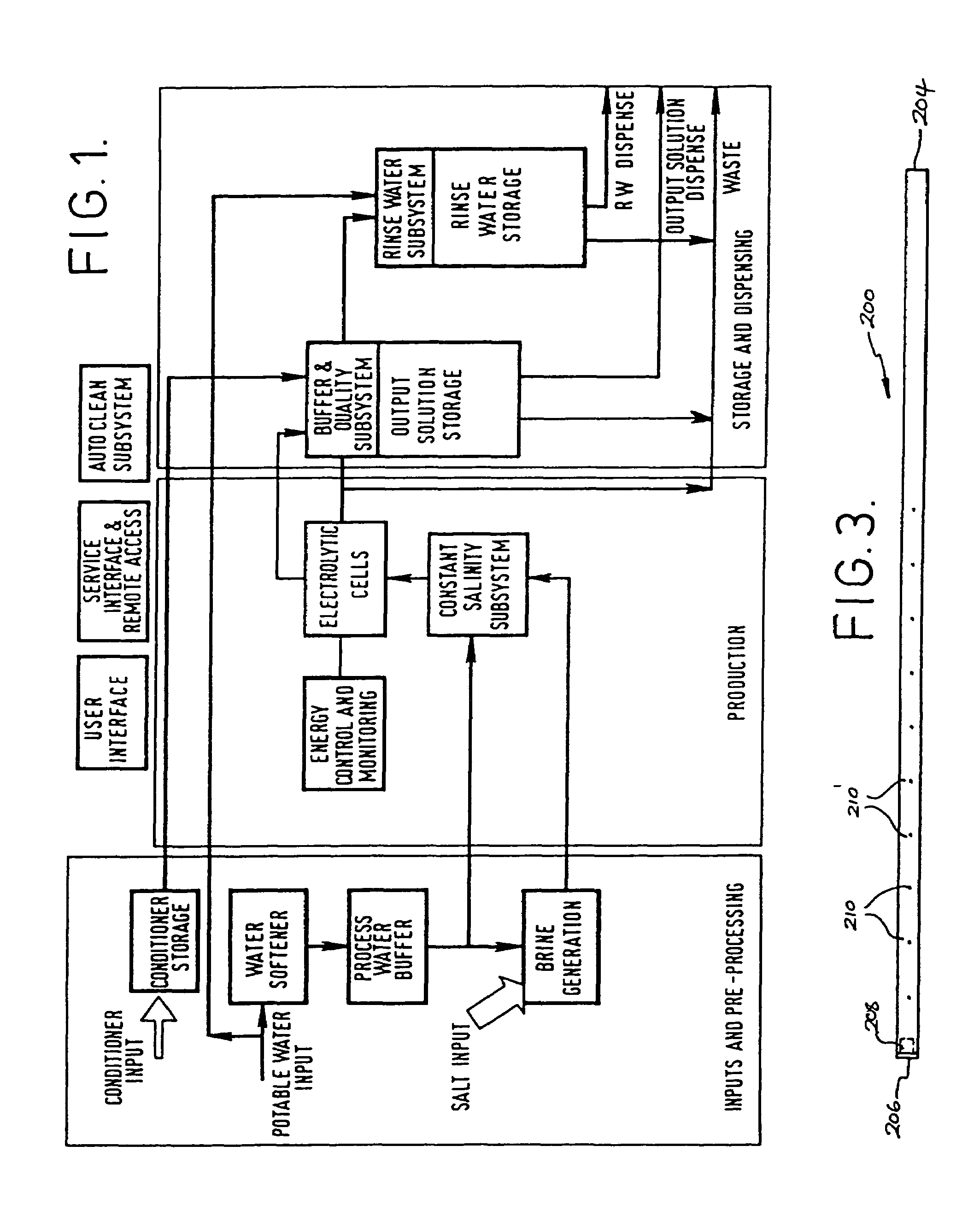
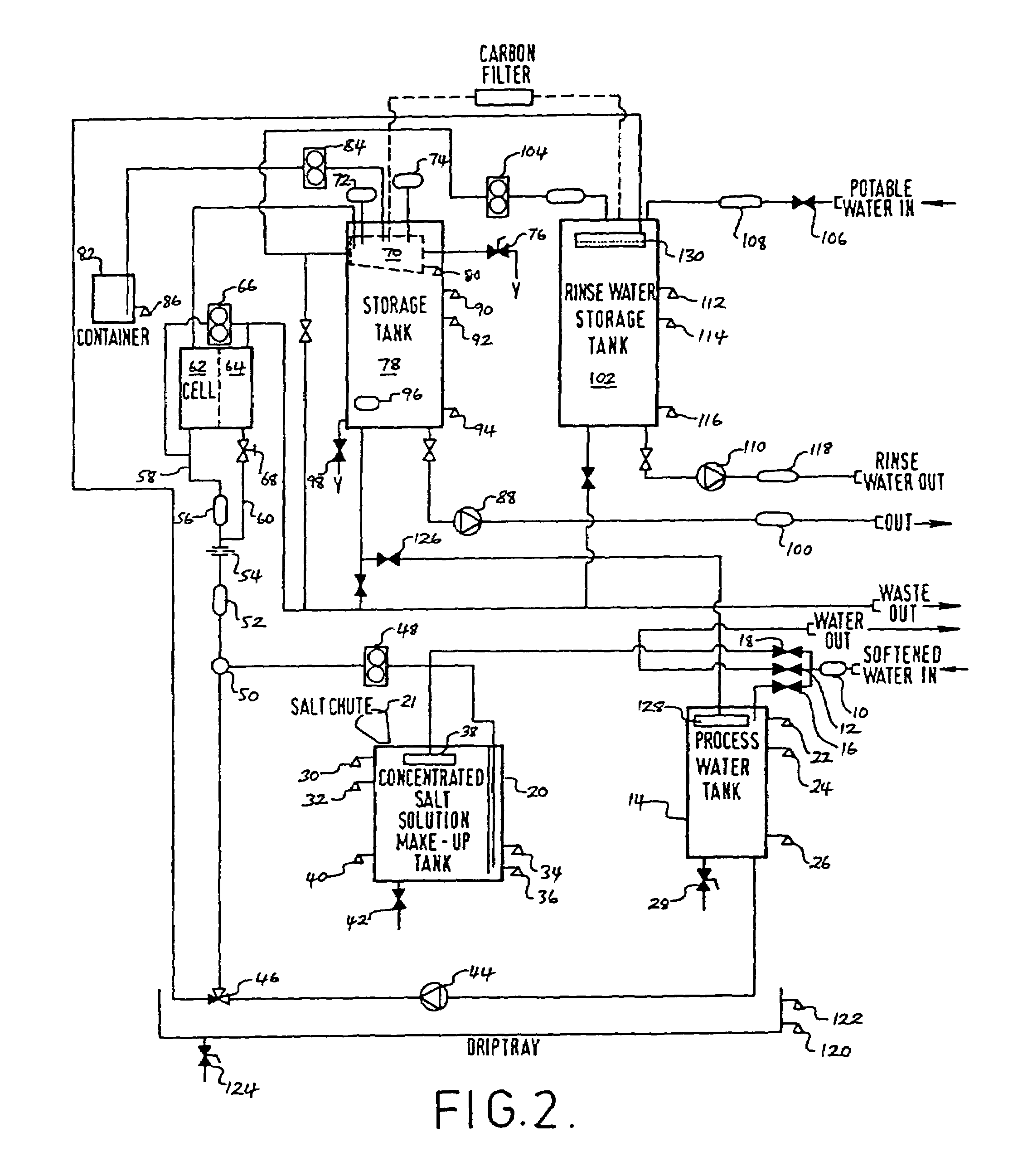
InactiveUS20050126927A1Efficient killingQuality improvementGalleysScale removal and water softeningElectrolysisWater storage tank
A water supply system, particularly for an aircraft such as a passenger aircraft, includes a disinfecting unit with an electrolysis cell connected in series with a water storage tank including a water distribution pipe system and a circulating pump. A bypass pipe with a valve permits connecting the electrolysis cell to the distribution system or bypassing the electrolysis cell. The electrolysis cell disinfects the water by anodic oxidation. A salt solution container may be connected to the electrolysis cell to enhance the anodic oxidation. The pump circulates the disinfected water through the tank and / or through the entire system, whereby the water supply is disinfected and the disinfected water in turn disinfects the entire system.
Owner:LINDAUER CHRISTIANE +1
Apparatus for purifying water
InactiveUS7238278B2Reduce concomitant disadvantageEconomical purification processLiquid solutions solvent extractionLoose filtering material filtersHypohaliteMetal salts
The invention relates to a method for purifying water by forming in an electrolytic cell molecular halogen, hypohalic acid, hypohalite ions or combinations thereof, from halide ions dissolved in the water; and dissolving one or more soluble metal salts in the water to provide corresponding metal ions. The invention also relates to a system for purifying water, having an electrolytic cell comprising a plurality of electrodes sufficient to electrolytically convert halide ion in the water into molecular halogen, hypohalic acid, or hypohalite ions, or combinations thereof; and a metal generator, which provides concentrations of one or more metals to the water.
Owner:ZODIAC POOL SYST LLC
Process for converting hydrocarbon feedstocks with electrolytic recovery of halogen
InactiveUS20080314758A1Improve efficiencyReduced Power RequirementsSolid-state devicesHydroxy compound preparationHalogenHydrocotyle bowlesioides
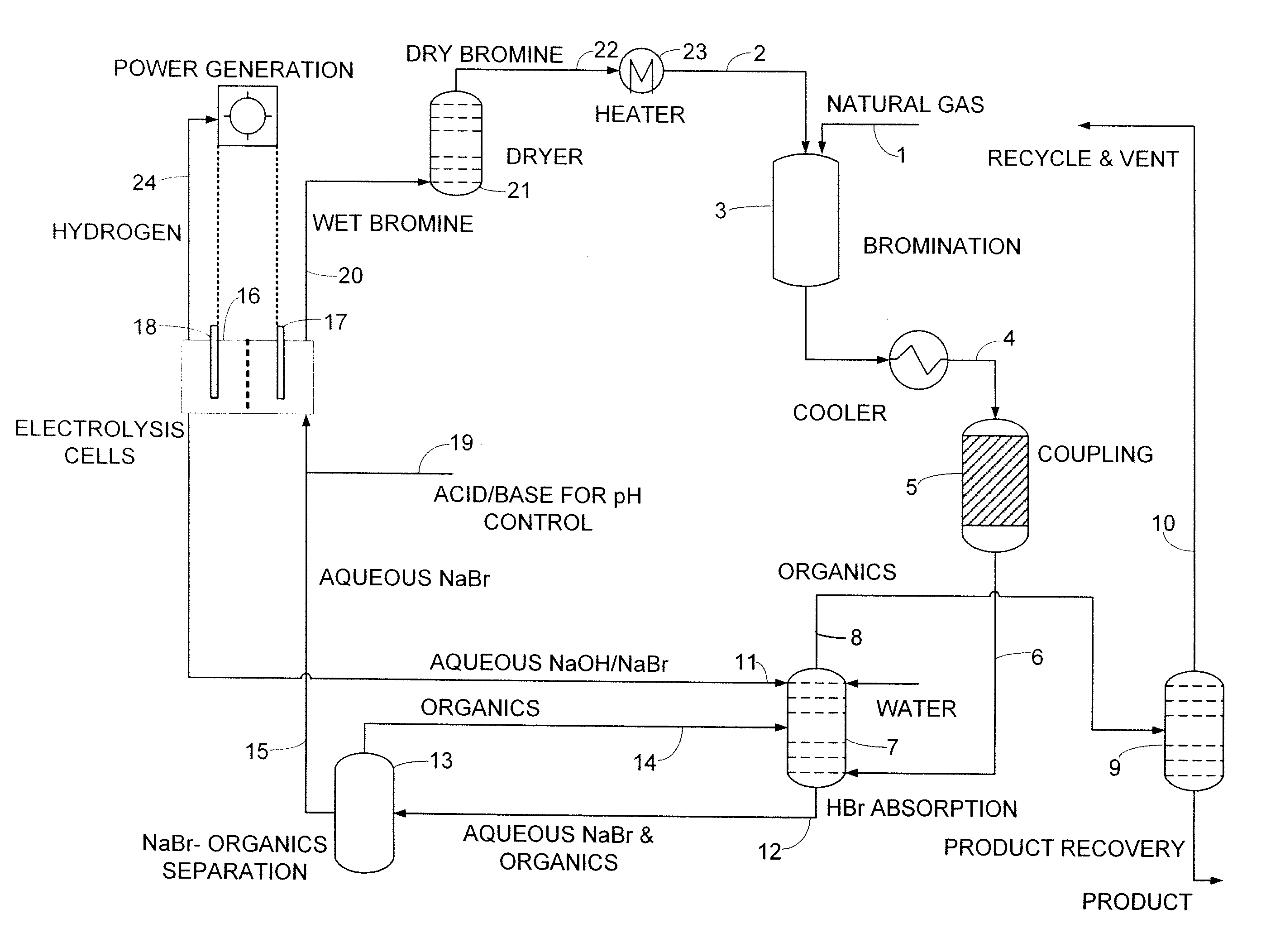
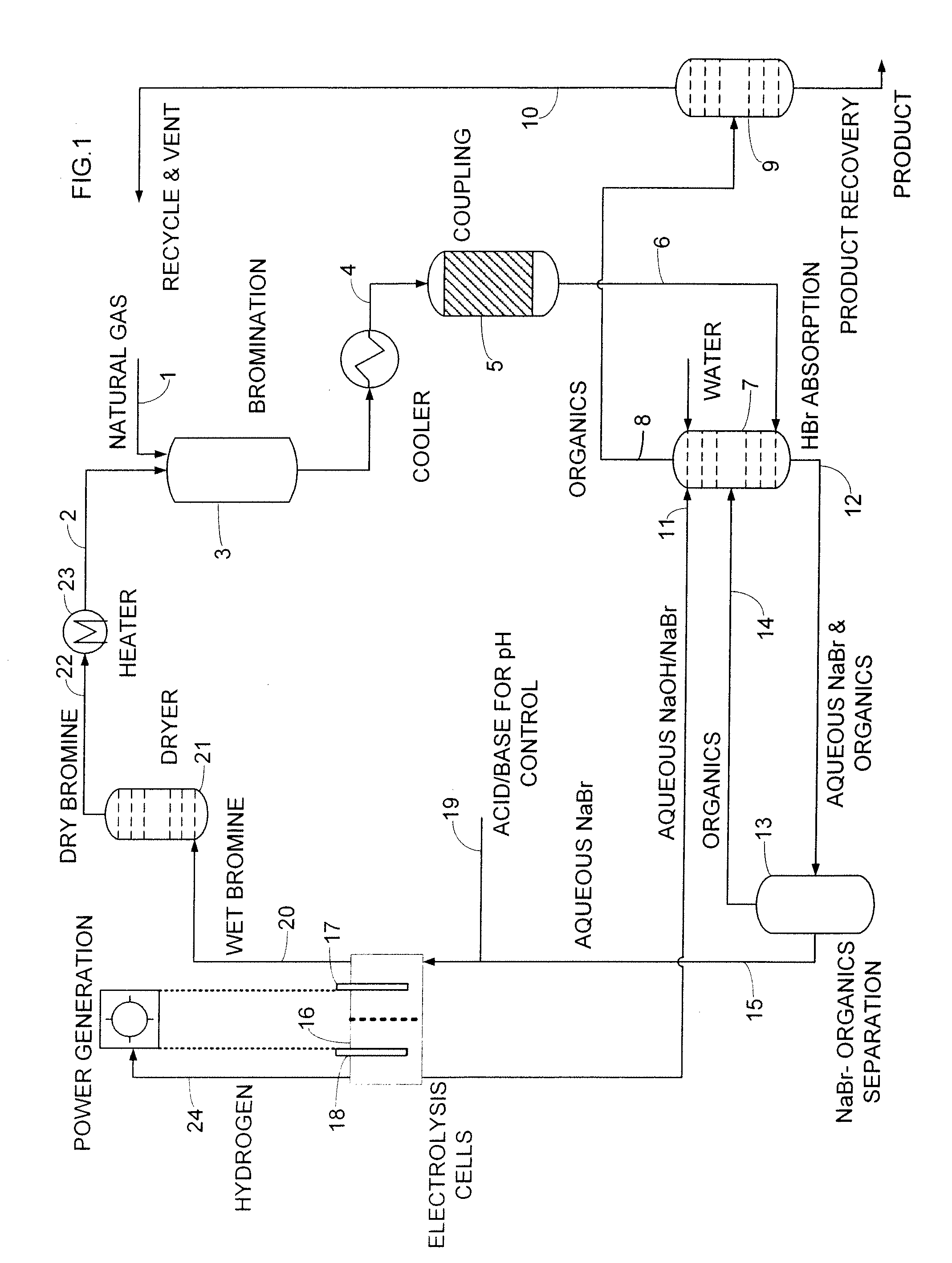
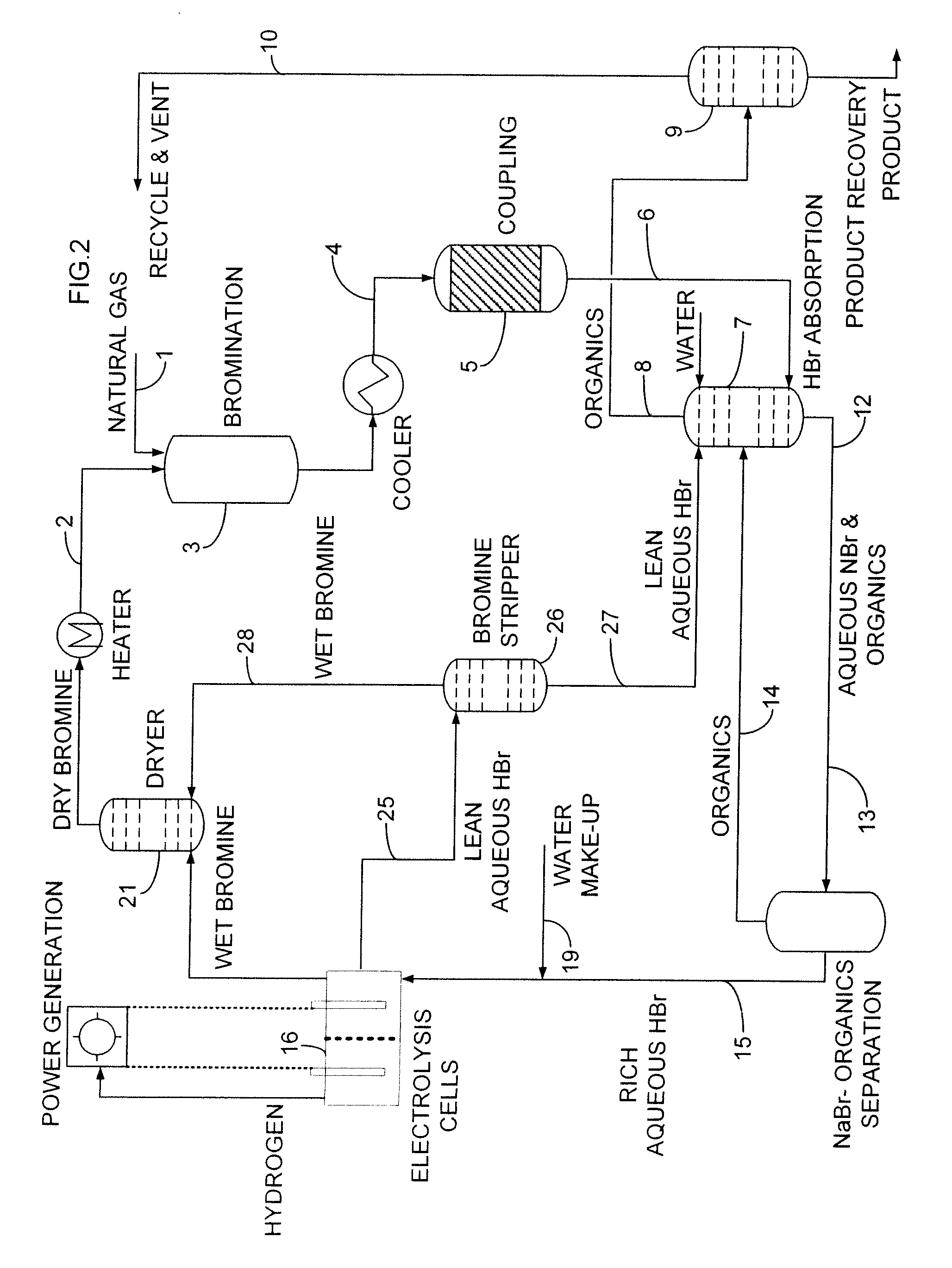
An improved continuous process for converting methane, natural gas, and other hydrocarbon feedstocks into one or more higher hydrocarbons, methanol, amines, or other products comprises continuously cycling through hydrocarbon halogenation, product formation, product separation, and electrolytic regeneration of halogen, optionally using an improved electrolytic cell equipped with an oxygen depolarized cathode.
Owner:REACTION 35 LLC
Electrochemical treatment of an aqueous solution
A method and apparatus are provided for the electrochemical treatment of an aqueous solution in an electrolytic cell. Output solution having a predetermined level of available free chlorine is produced by applying a substantially constant current across the cell between an anode and a cathode while passing a substantially constant throughput of chloride ions through the cell.
Owner:REALM THERAPEUTICS INC
Internal combustion engine/water source system
InactiveUS7302795B2Increase pointsSimple and reliable processNon-fuel substance addition to fuelInternal combustion piston enginesExternal combustion engineWater source
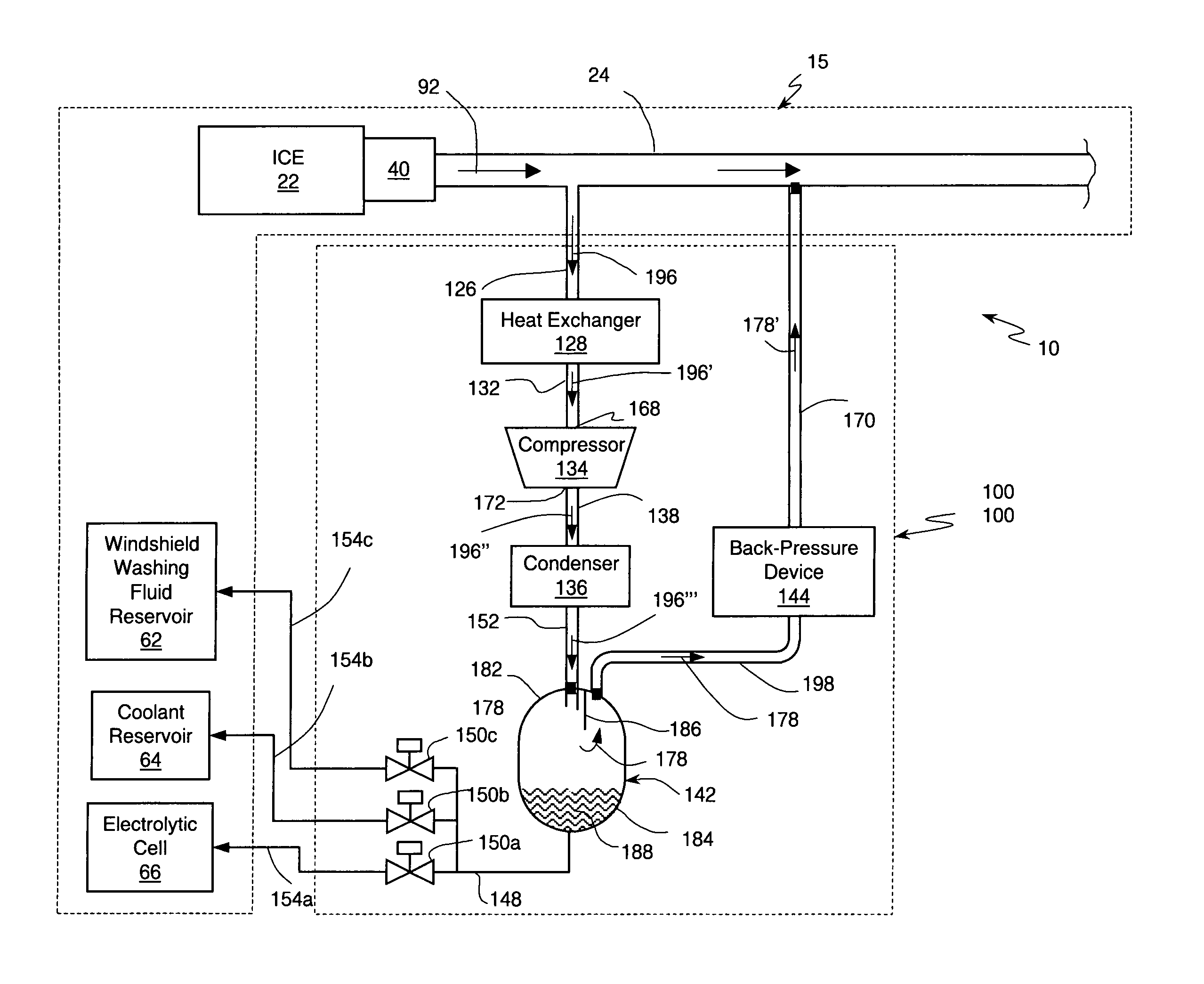
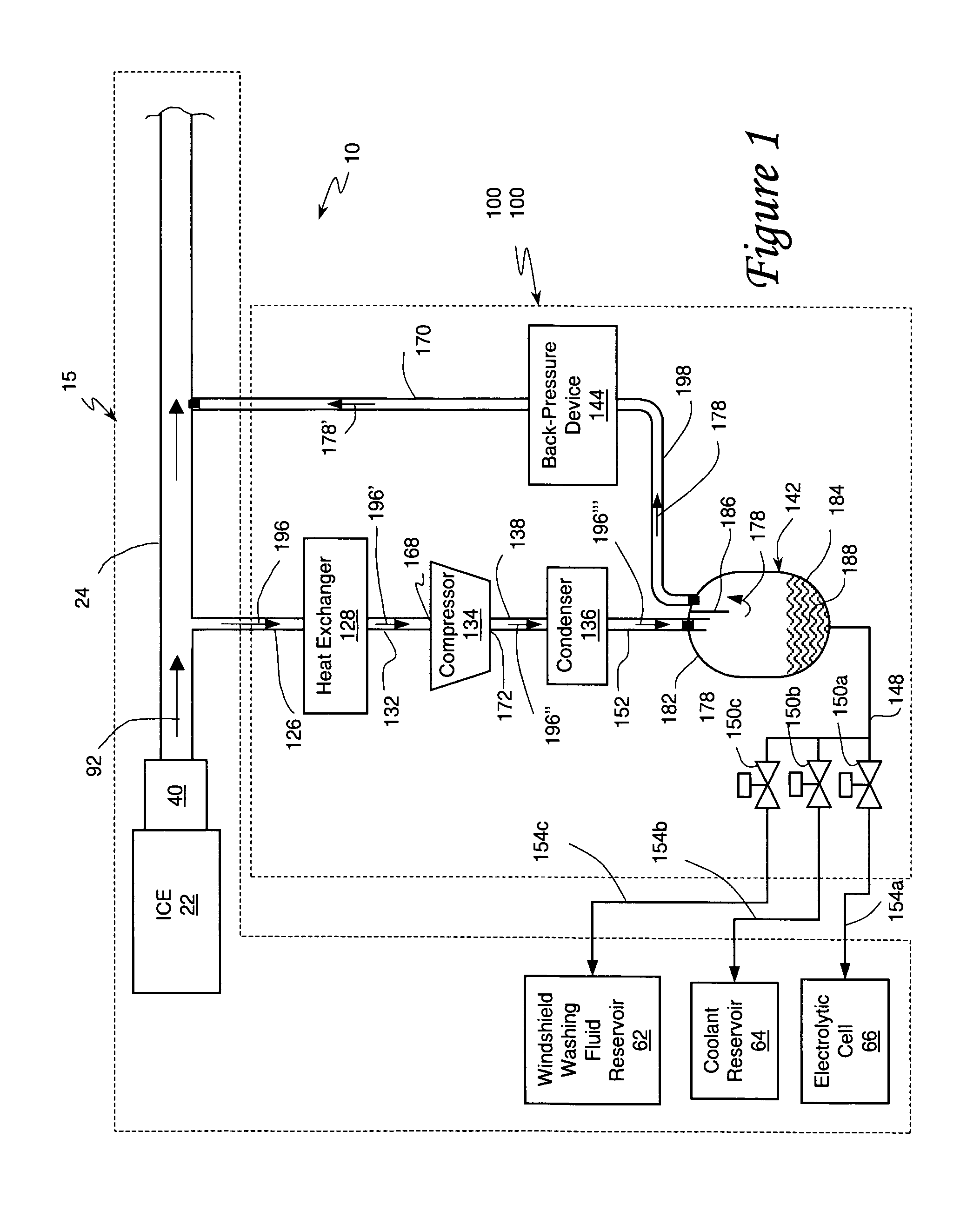
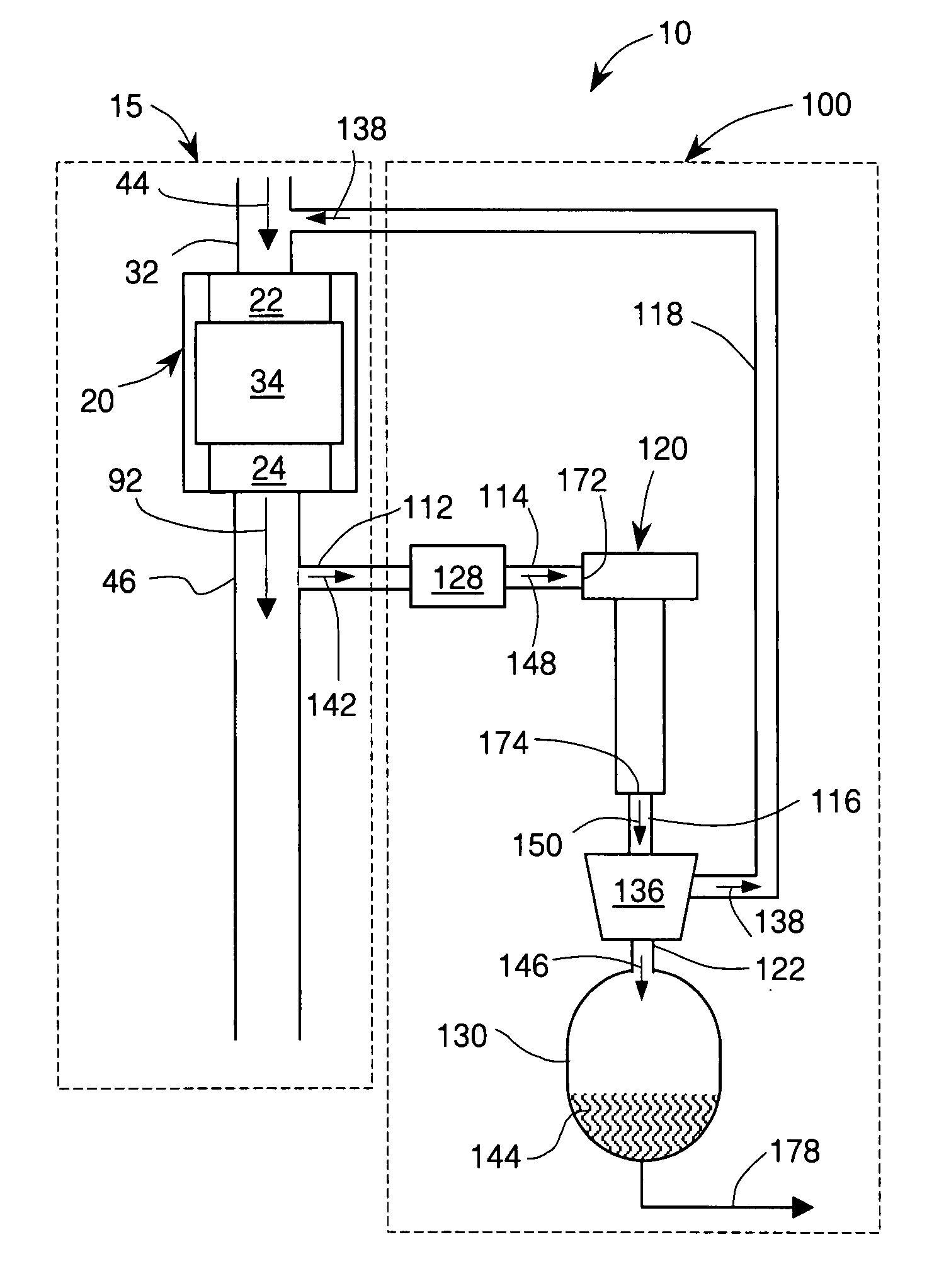
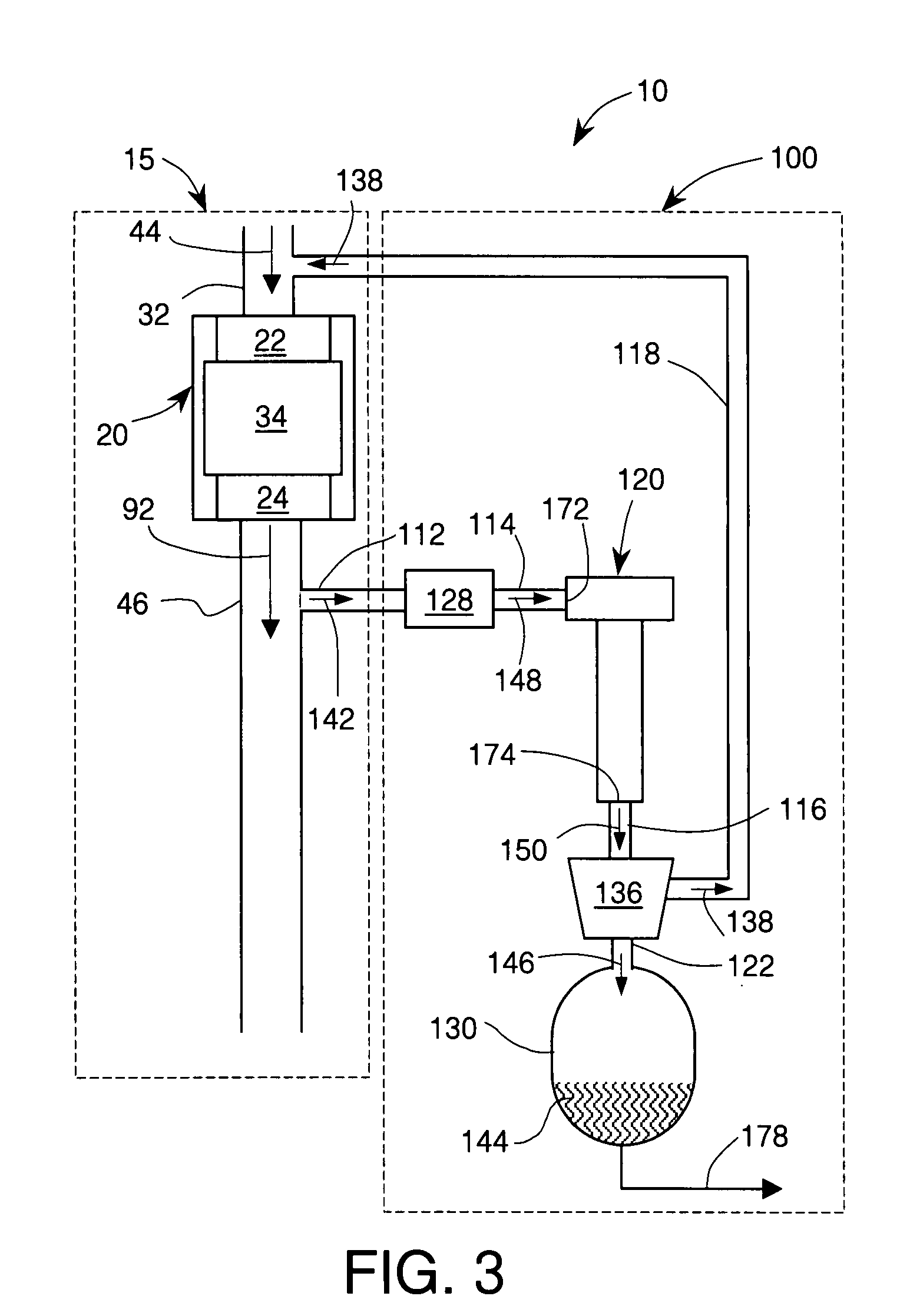
An internal combustion engine system for automotive vehicle wherein liquid water is produced by cooling a portion of exhaust gases at elevated pressure to induce condensation. The use of elevated pressure allows condensation to occur at a higher dew point which is easier to realize with cooling by ambient air. Liquid water condensate is collected and provided to an electrolytic cell for electrolysis into gaseous hydrogen and oxygen. Hydrogen gas generated by the cell is used by the internal combustion engine to reduce internal combustion engine wear and to reduce exhaust pollutants especially during start-up. Alternate uses of the liquid water include a replenishment of engine coolant and window washing fluid.
Owner:VETROVEC JAN
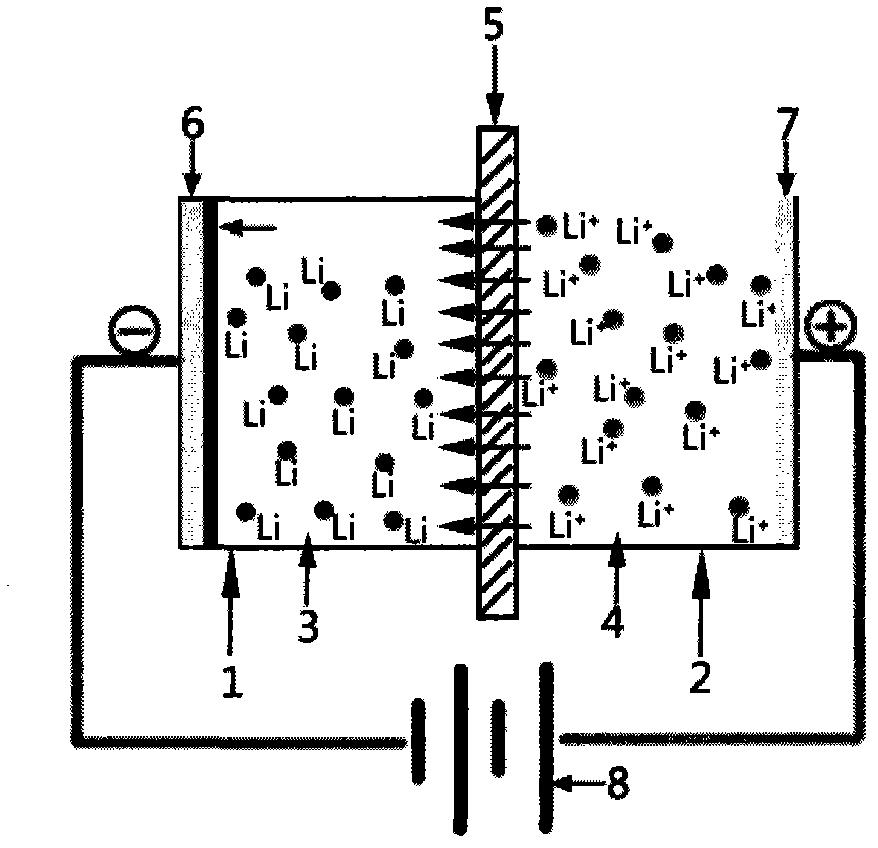
Preparation method of lithium metal through electrolysis
ActiveCN103031568AHigh purityWide variety of sourcesPhotography auxillary processesProcess efficiency improvementElectrical conductorSingle substance
The invention discloses a preparation method of lithium metal through electrolysis, which comprises the following steps: at normal temperature and normal pressure, applying direct current voltage on an anode current collector and a cathode current collector so that potassium ions in a water phase in an anode chamber penetrate through a diaphragm having lithium ion conductor characteristics under the driving of the voltage, an organic solvent in a cathode chamber is reduced to a metal lithium single substance which is deposited and enriched on the surface of the cathode current collector to obtain the product, wherein the anode chamber of an electrolysis cell is filled with an aqueous solution at least containing the lithium ions, the cathode chamber od the electrolysis cell is filled with the organic solvent having the lithium ion conductor characteristics, the diaphragm for separating the anode chamber from the cathode chamber is a lithium ion conductor ceramic membrane having the lithium ion conductor characteristics or a composite membrane of a lithium ion conductor and a polymer, and the cathode chamber is in inert gas atmosphere. The electrolysis preparation method for lithium metal avoids severe conditions which are required to prepare lithium metal by a traditional high temperature molten electrolysis process, and has the characteristics of low energy consumption, high lithium extraction efficiency and high product purity and is environment-friendly and wide in raw material sources.
Owner:QINGDAO INST OF BIOENERGY & BIOPROCESS TECH CHINESE ACADEMY OF SCI
Method for pre-lithiating electrode material of lithium ion battery
InactiveCN104201320AIncrease profitLow costFinal product manufactureCell component detailsElectrical conductorElectrical battery
The invention relates to an electrode material of a lithium ion battery, in particular to a method for pre-lithiating a cathode material. The method comprises the steps that an electrolytic cell cathode cavity is made of the electrode material such as a lithium ion cathode material and arranged in a lithium ion conductive organic electrolyte; an anode cavity is an aqueous solution containing lithium salt or an organic solution; the anode cavity is separated from the cathode cavity by a lithium ion conductor ceramic membrane or a composite membrane of lithium ion conductor ceramic and a high molecular material; an electric potential and current density are controlled by external circuit charge and discharge equipment to allow lithium ions to migrate to a cathode from an anode through the membrane; and an SEI (Solid Electrolyte Interphase) membrane is formed on the surface of the material; or the electrode material is pre-lithiated. According to the method, a cheap and safe lithium ion saline solution serves as a source of the lithium ion; the SEI membrane is generated for the cathode of the lithium ion battery in advance; or lithium is supplemented to the electrode material; the coulombic efficiency and cycling stability of the cathode material can be improved; a formation process in production of the lithium ion battery in the prior art is simplified; the electrode material and cost are saved; and the method is safe and efficient and has a large-scale application prospect.
Owner:赵前永
Internal combustion engine/water source system
InactiveUS20070137590A1Easy to operateReduce pollutantsElectrolysis componentsInternal combustion piston enginesExternal combustion engineWater source
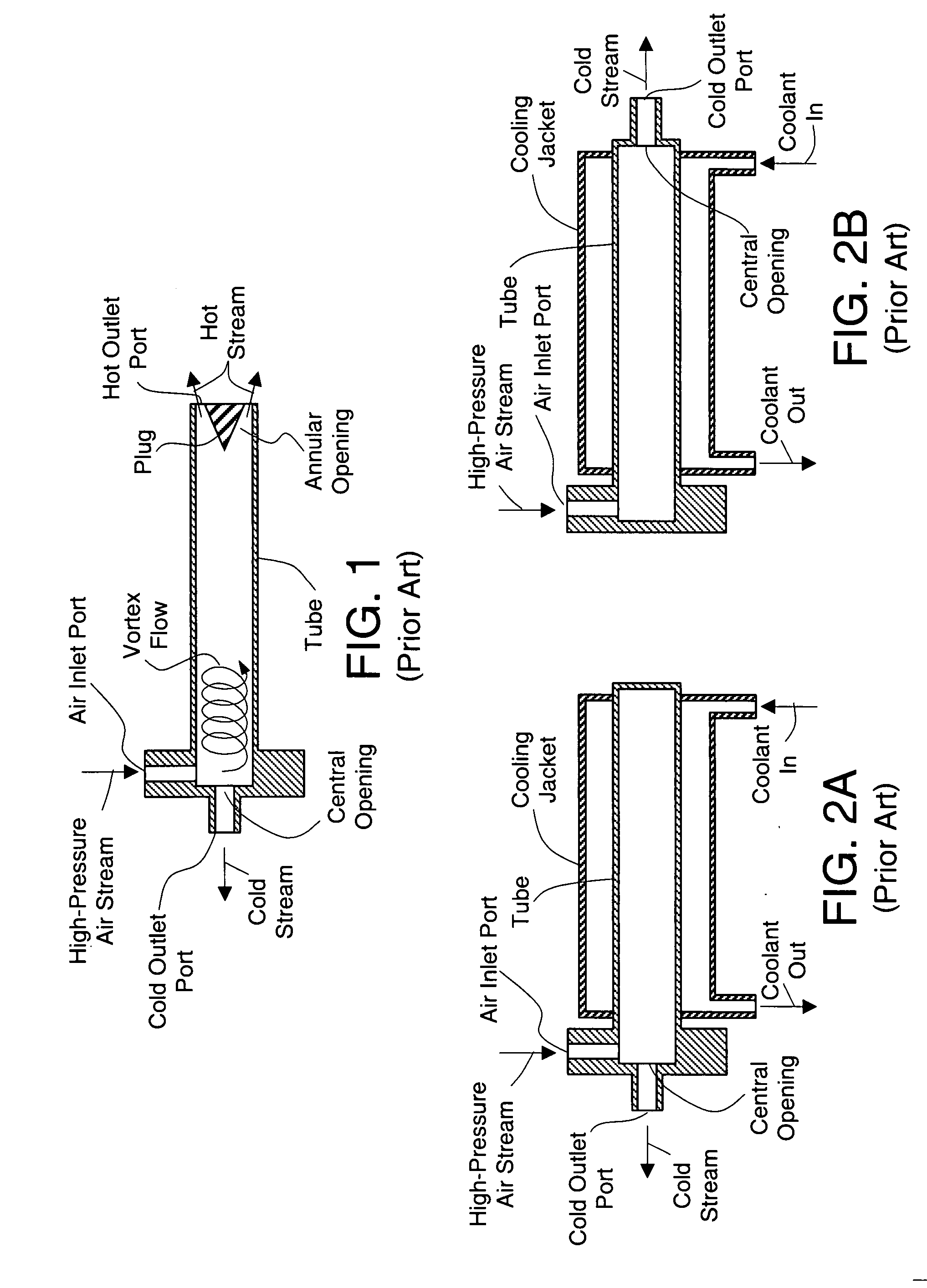
An internal combustion engine / water source system for a vehicle powered by a internal combustion engine wherein liquid water is produced by cooling a portion of engine exhaust gases in a vortex tube to induce condensation. In one embodiment, engine exhaust gases are pumped into the vortex tube by a compressor. After removing a portion of water vapor, cooled exhaust gases may be re-introduced to engine's combustion chamber thereby providing an exhaust gas recirculation. In an automotive vehicle, liquid water generated by the invention may be collected and provided to an electrolytic cell for electrolysis into gaseous hydrogen to reduce exhaust pollutants during cold engine start. Alternatively, water generated by the invention may be injected into engine combustion chamber to increase power and to reduce production of nitrogen oxides.
Owner:VETROVEC JAN
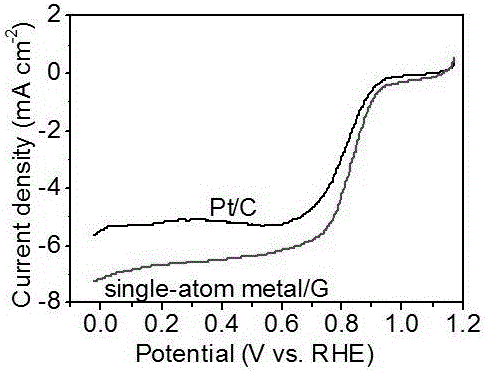
Method for preparing monodisperse metal atom/graphene composite material employing electrochemical dissolved graphite
ActiveCN106654300ALow costSimple stepsCell electrodesMetal/metal-oxides/metal-hydroxide catalystsGraphiteElectrochemistry
The invention discloses a method for preparing a monodisperse metal atom / graphene composite material employing electrochemical dissolved graphite. The method comprises the steps of (1) preparing an electrode from a graphite-based material; (2) electrolyzing the prepared electrode in an electrolytic cell, carrying out solid-liquid separation and recycling an electrolyte; (3) further stripping the solid obtained by separation and carrying out solid-liquid separation to obtain a crude monodisperse metal atom / graphene composite material; (4) separating and purifying the crude monodisperse metal atom / graphene composite material; and (5) carrying out thermal treatment on the composite material obtained in the step (4) under inert atmosphere protection and cooling and drying the material to obtain a monodisperse metal atom / graphene composite catalyst. The preparation method is simple in process steps, high in efficiency and low in energy consumption, and massive production can be achieved.
Owner:SHANXI INST OF COAL CHEM CHINESE ACAD OF SCI
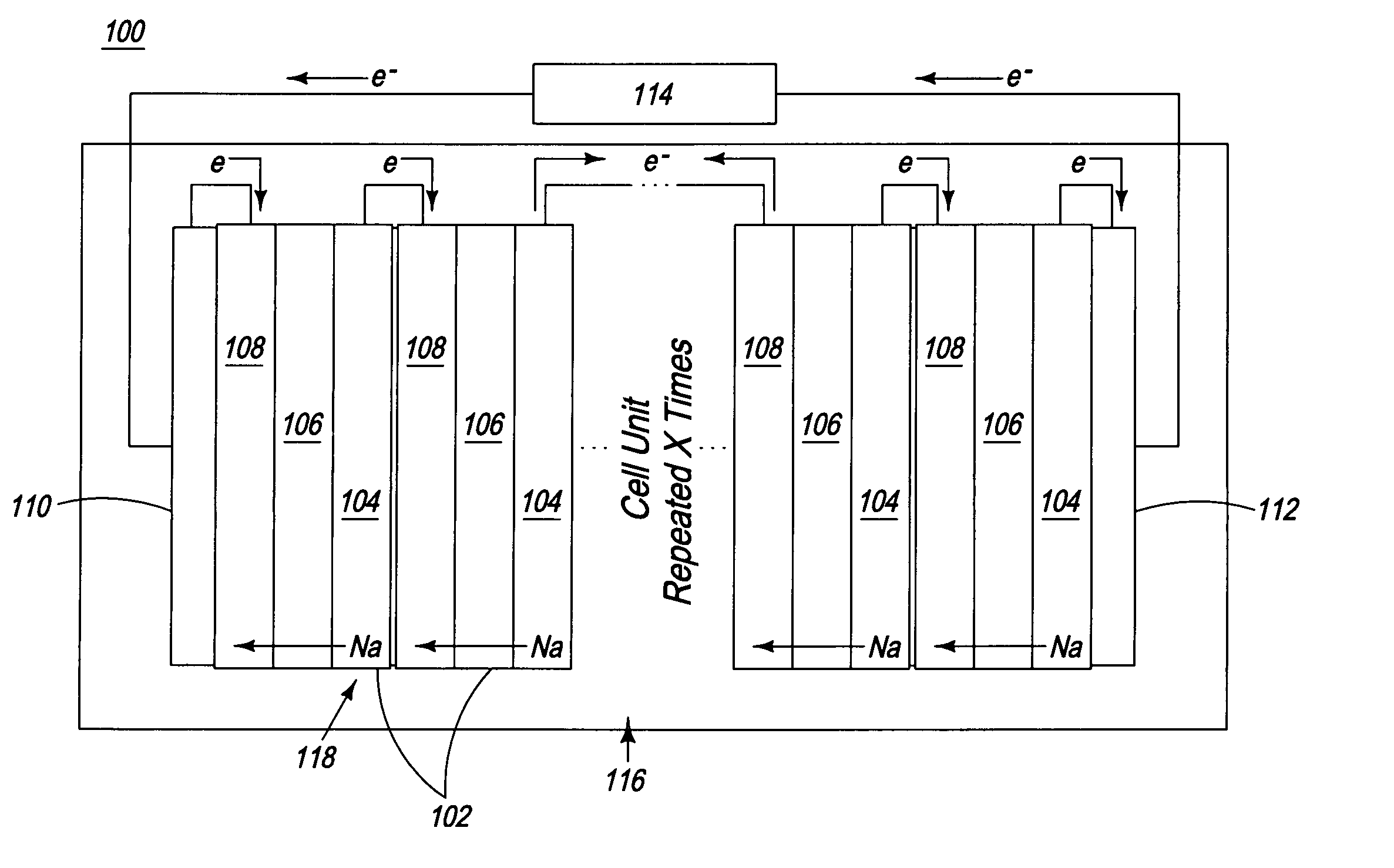
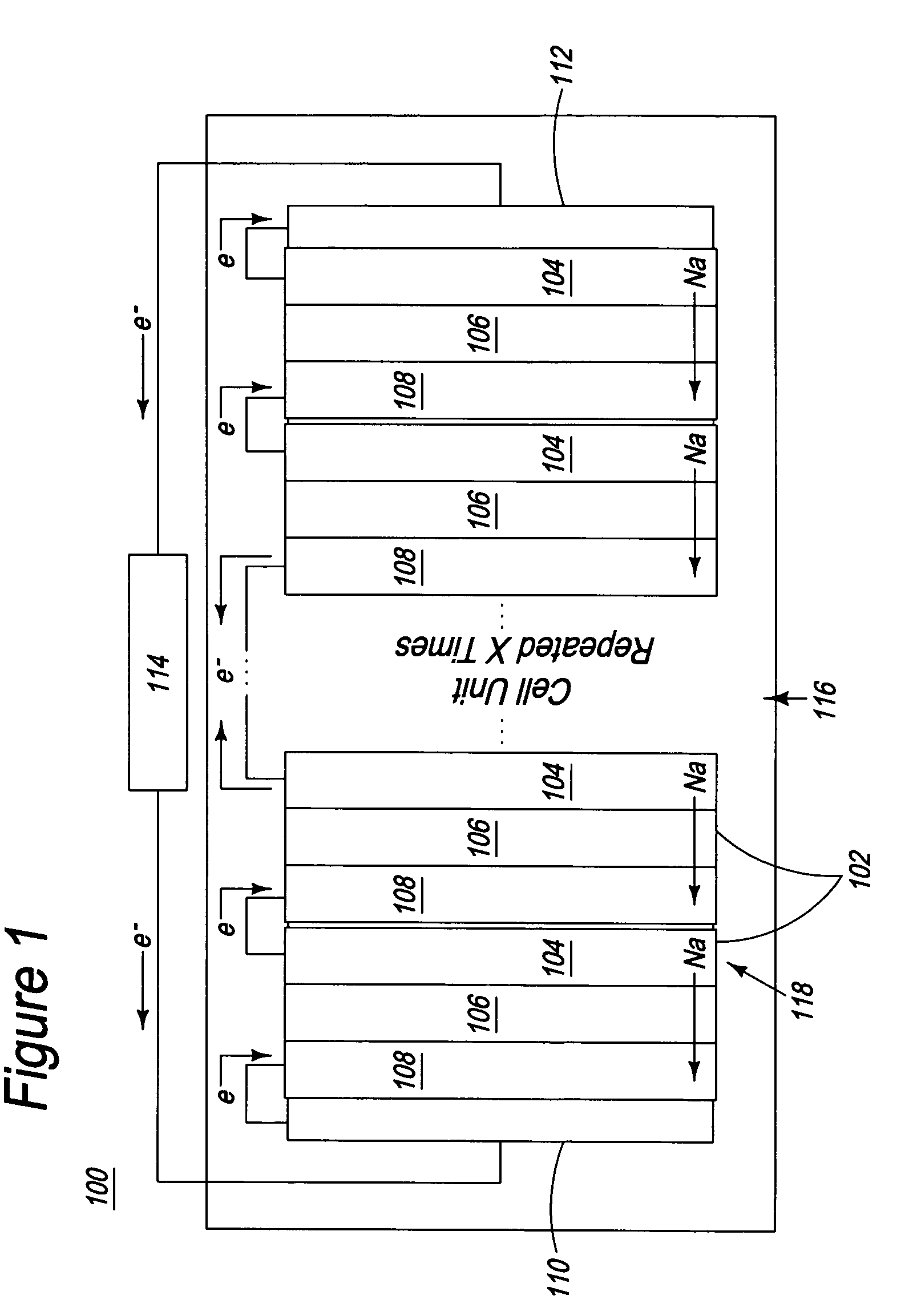
Apparatus and process for the production of metals in stacked electrolytic cells
InactiveUS20060144701A1Eliminate needPermit useCellsPhotography auxillary processesLithiumElectrical conductor
A process and apparatus for reducing an alkali metal salt to an alkali metal through electrolysis in a series of electrolytic cells are disclosed. The process employs as a separator between anode and cathode compartments a material that is both an ionic conductor of the metal ion and an electrical insulator. Preferred metals are sodium, lithium and potassium.
Owner:MILLENNIUM CELL
Method and apparatus for purifying water
The invention relates to a method for purifying water by forming in an electrolytic cell molecular halogen, hypohalic acid, hypohalite ions or combinations thereof, from halide ions dissolved in the water; and dissolving one or more soluble metal salts in the water to provide corresponding metal ions. The invention also relates to a system for purifying water, having an electrolytic cell comprising a plurality of electrodes sufficient to electrolytically convert halide ion in the water into molecular halogen, hypohalic acid, or hypohalite ions, or combinations thereof; and a metal generator, which provides concentrations of one or more metals to the water.
Owner:ZODIAC POOL SYST
Water treatment method and apparatus
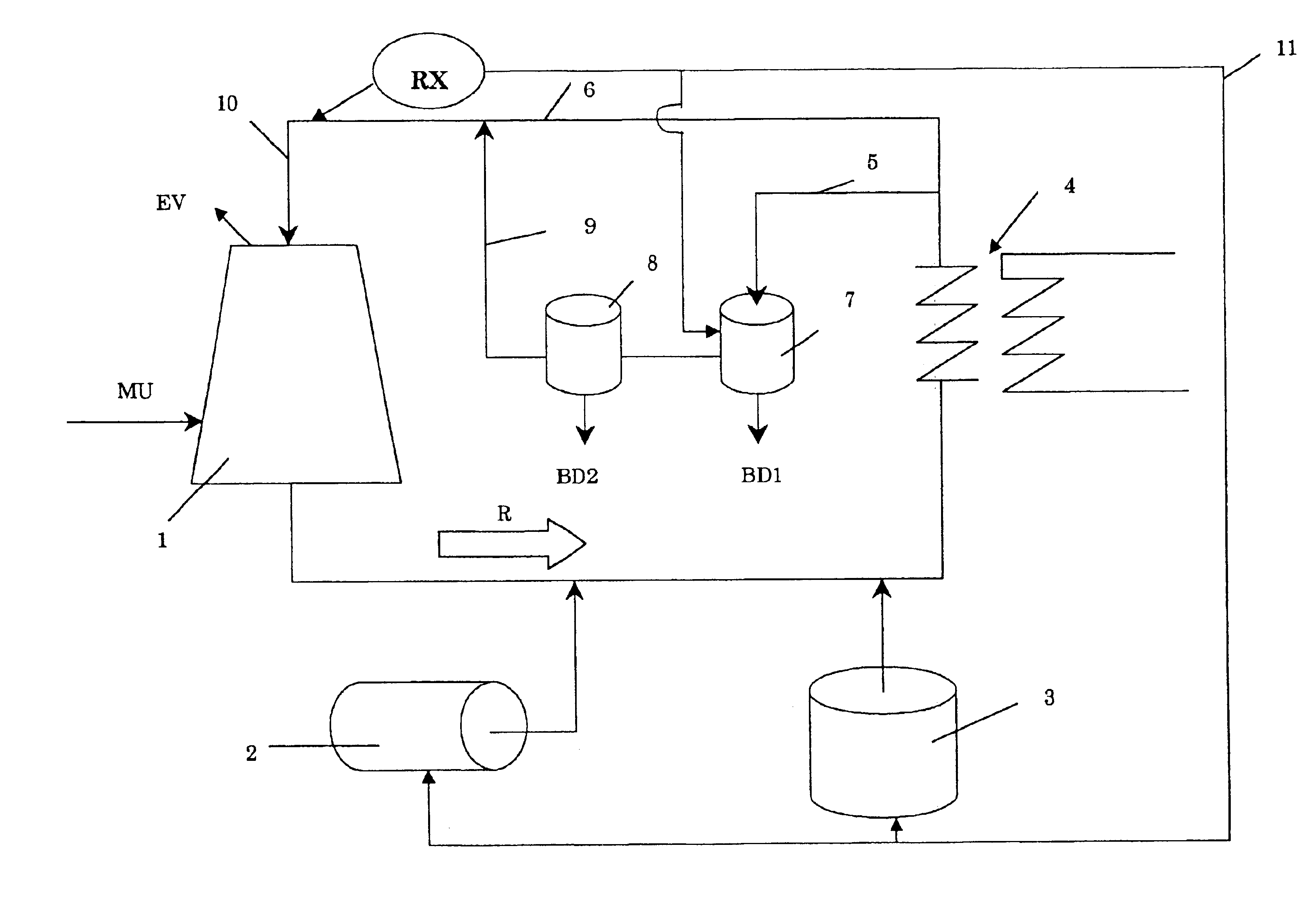
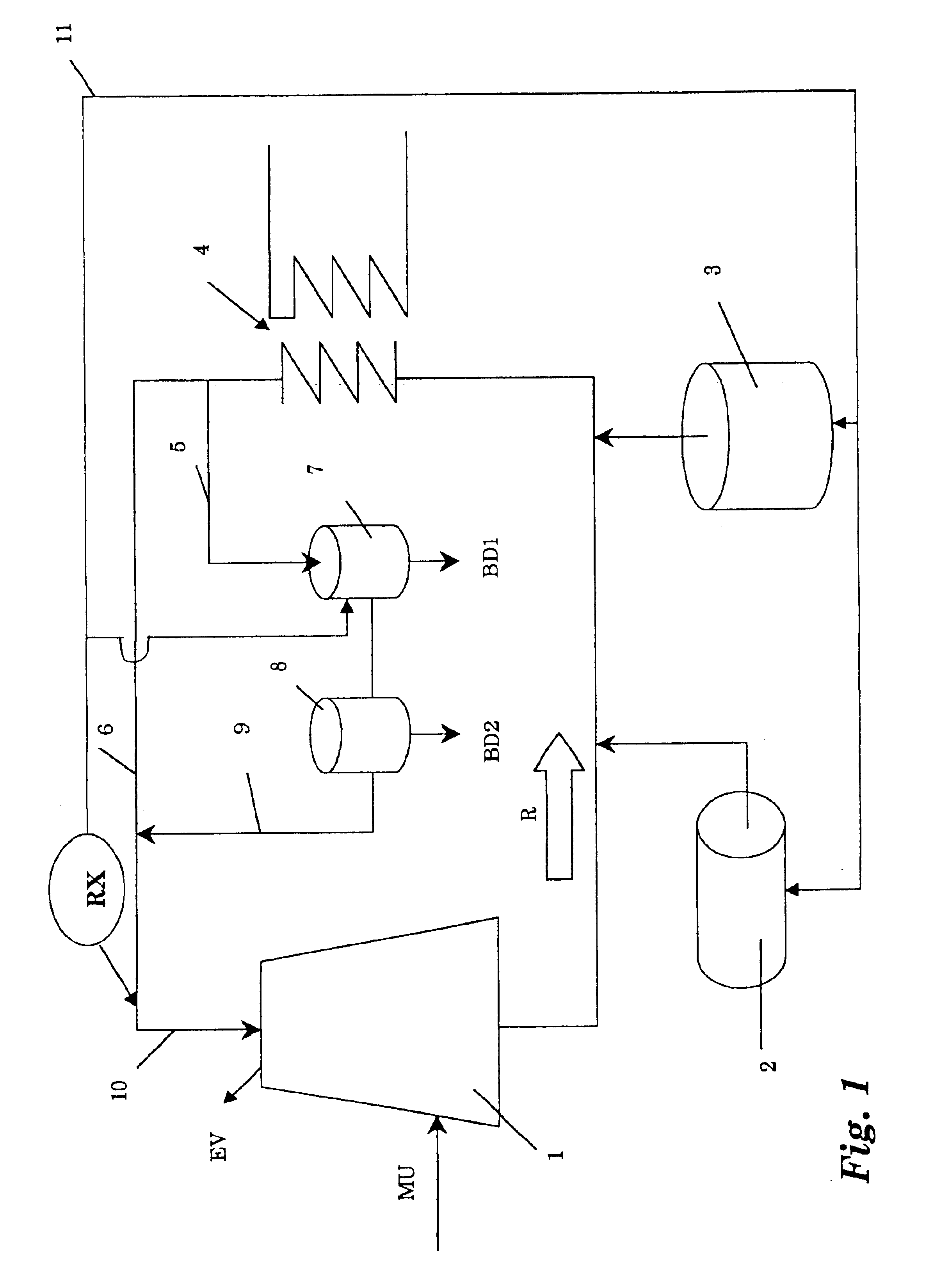
InactiveUS6733654B1Water treatment parameter controlNature of treatment waterCooling towerWater flow
A method of operating a cooling tower comprises feeding to the cooling tower a make-up stream of water containing organic and / or biological contaminants, causing a side stream taken from the recirculating stream to pass through an electrolytic cell, removing solids precipitating by the action of the cell, and remixing the treated side stream with the main stream, before feeding them to the cooling tower.
Owner:ARGAD EYAL