Patents
Literature
5531 results about "Anodic oxidation" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
Anodic oxidation is an electrochemical method for the production of an oxide film on a metallic substrate. It removes electrons from a substance and oxidizes the anode. ... Anodic oxidation is an accelerated electrochemical oxidation process, which is intensified by the natural oxide skin of the metal. It involves the application of an electrical bias at relatively low currents while the substrate is immersed in an acid bath.
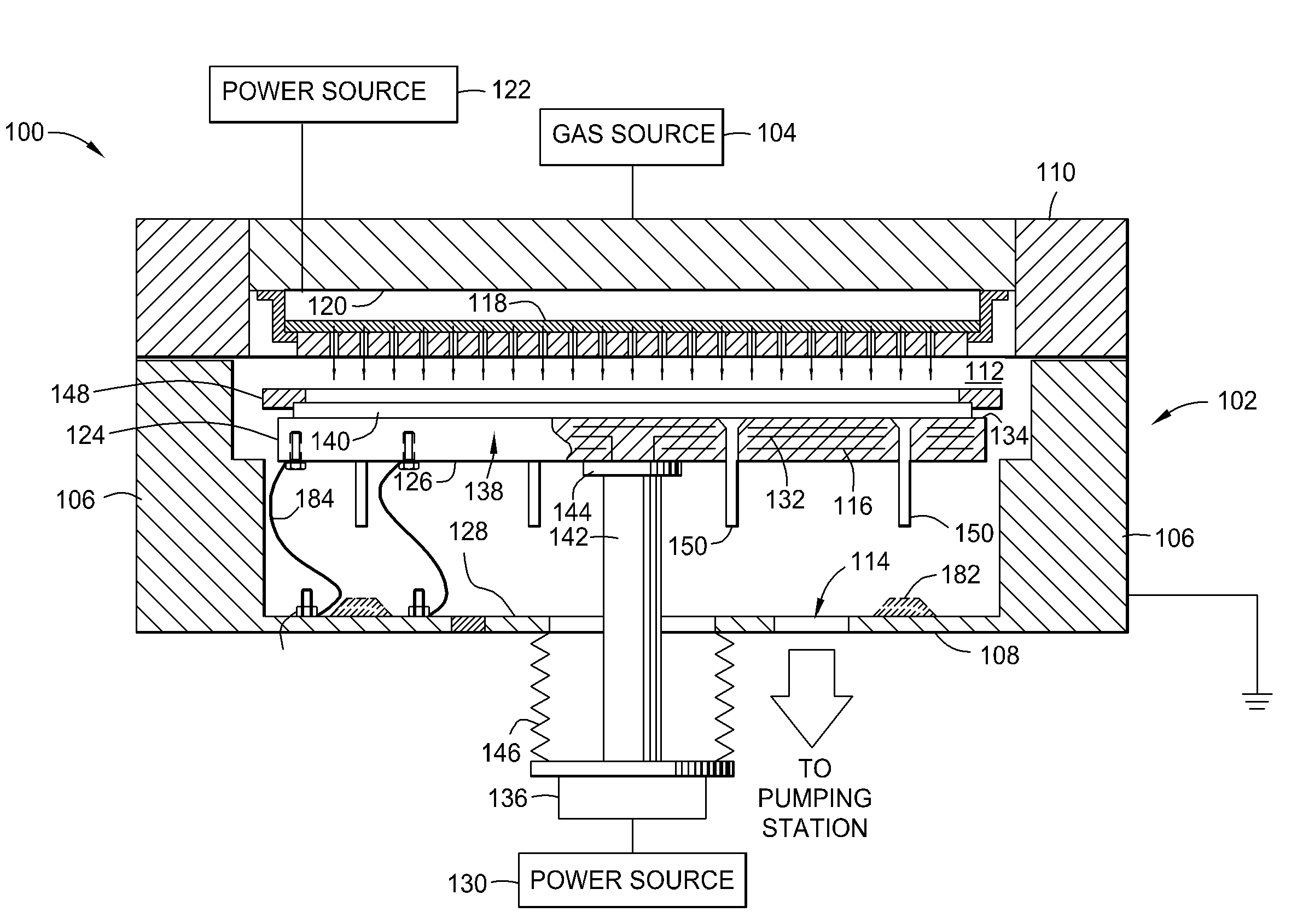
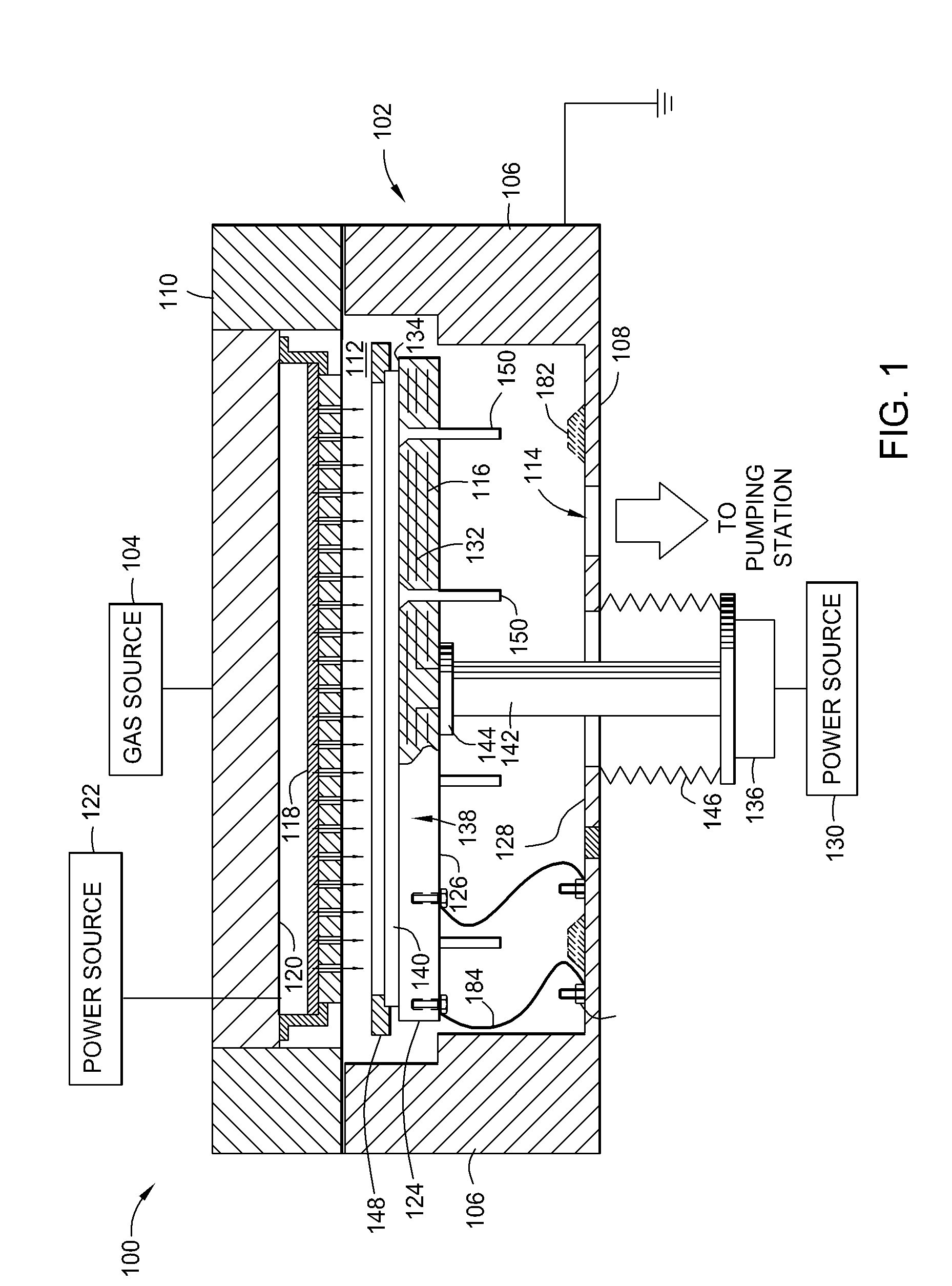
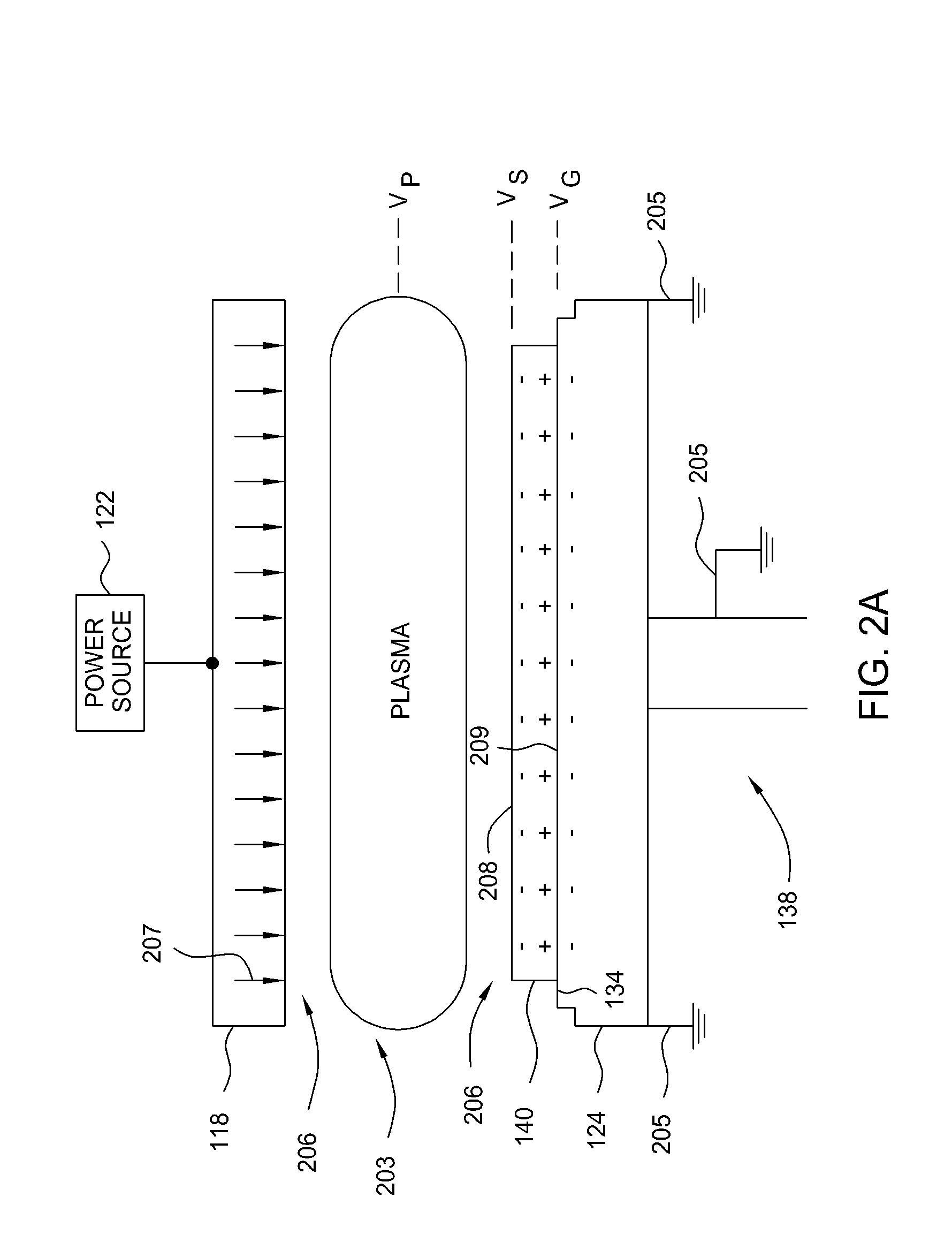
Susceptor with insulative inserts
InactiveUS20080194169A1Reduce arcingElectric discharge tubesSemiconductor/solid-state device manufacturingSusceptorMechanical engineering
A method and apparatus for reducing arcing in a plasma processing system when processing large area substrates which contain one or more holes. In one embodiment of the invention, a substrate support member includes an electrically insulating insert located beneath a hole in an insulating, large area substrate. The insulating insert is made of aluminum oxide, and is located within a hole in the support member such that the insert is disposed beneath a hole in a glass substrate. The substrate support member is made of aluminum with an anodized surface.
Owner:APPLIED MATERIALS INC
Implantable medical device having flat electrolytic capacitor fabricated with laser welded anode sheets
ActiveUS6922330B2Robust electrical and mechanical connectionMinimize damageLiquid electrolytic capacitorsCapacitor terminalsElectrolysisOptoelectronics
Implantable medical devices (IMDs) and components, including flat electrolytic capacitors and methods of making and using same, particularly an improved electrolytic capacitor fabricated of an electrode stack assembly comprising a plurality of capacitor layers stacked in registration upon one another. Each capacitor layer comprises a valve metal cathode layer having a cathode tab, a valve metal anode layer having an anode tab, and a separator layer located between the cathode layers. The anode layer is assembled from a plurality of valve metal anode sheets that are etched and anodized, stacked side-by-side, and electrically and mechanically joined together by laser weld beads. A valve metal anode tab having a thickness equal to one or more anode sheet is inserted into a tab notch in one or more stacked anode sheet and joined to the anode sheet stack by laser welding the tab and sheet edges together.
Owner:MEDTRONIC INC
Plasma etching apparatus and method for forming inner wall of plasma processing chamber
InactiveUS20070215278A1Reduce corrosionDecreasing amountElectric discharge tubesSemiconductor/solid-state device manufacturingProduction rateHeat resistance
A plasma etching apparatus is provided which can prevent corrosion of an aluminum substrate constituting an etching processing chamber or an inside component thereof, thereby avoiding a reduction in productivity due to scattering of a sprayed coating. In the plasma etching apparatus, an anodic oxide film is disposed between a ceramic sprayed coating with excellent resistance to plasma, and the etching processing chamber and the inside component thereof made of aluminum alloy. The anodic oxide film has a thickness of 5 μm or less to have heat resistance.
Owner:HITACHI HIGH-TECH CORP
Coating packaged chamber parts for semiconductor plasma apparatus
ActiveUS20140120312A1Improve service performanceImprove processing qualityVacuum evaporation coatingSurface reaction electrolytic coatingSemiconductor plasmaPhysical chemistry
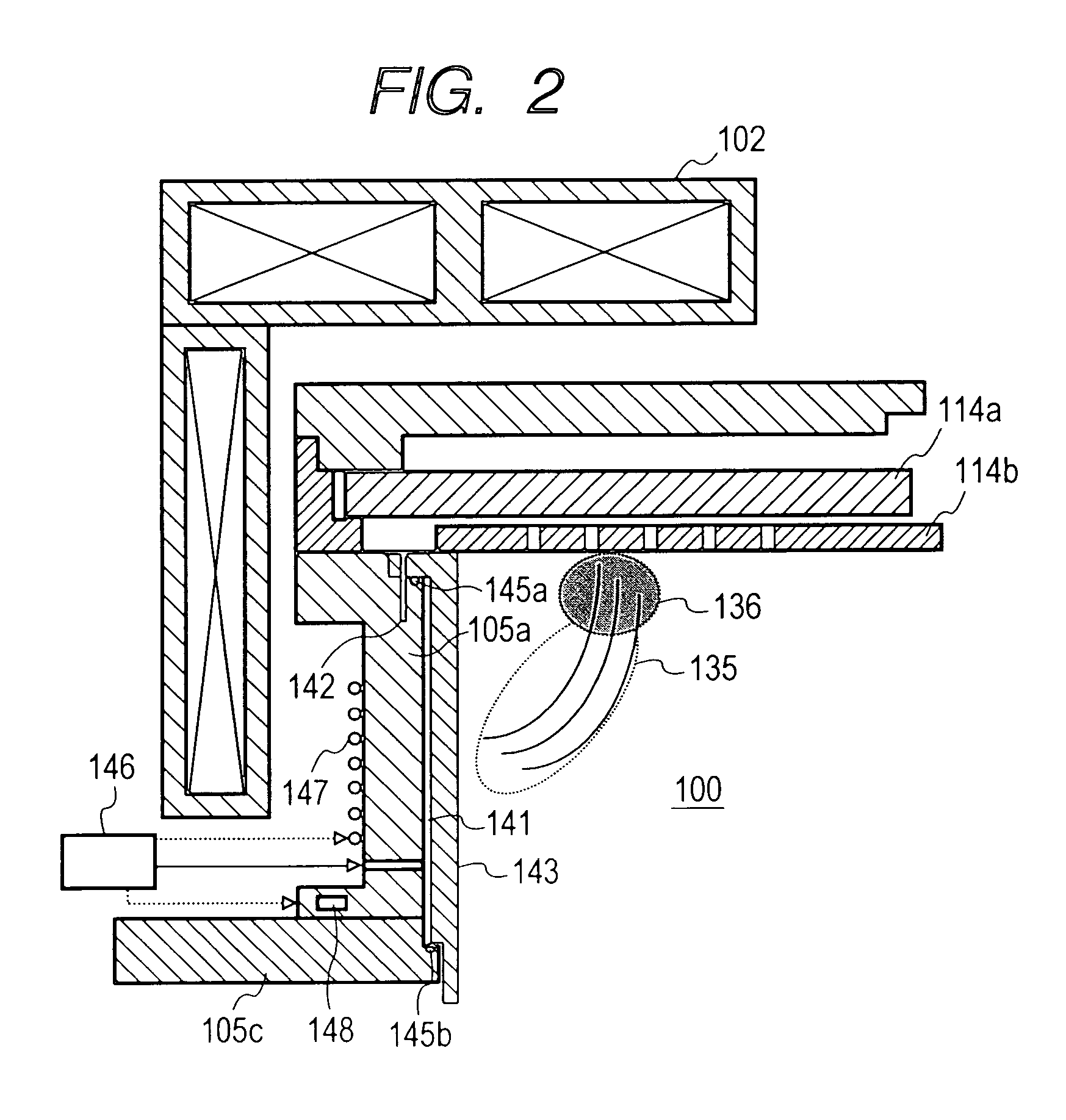
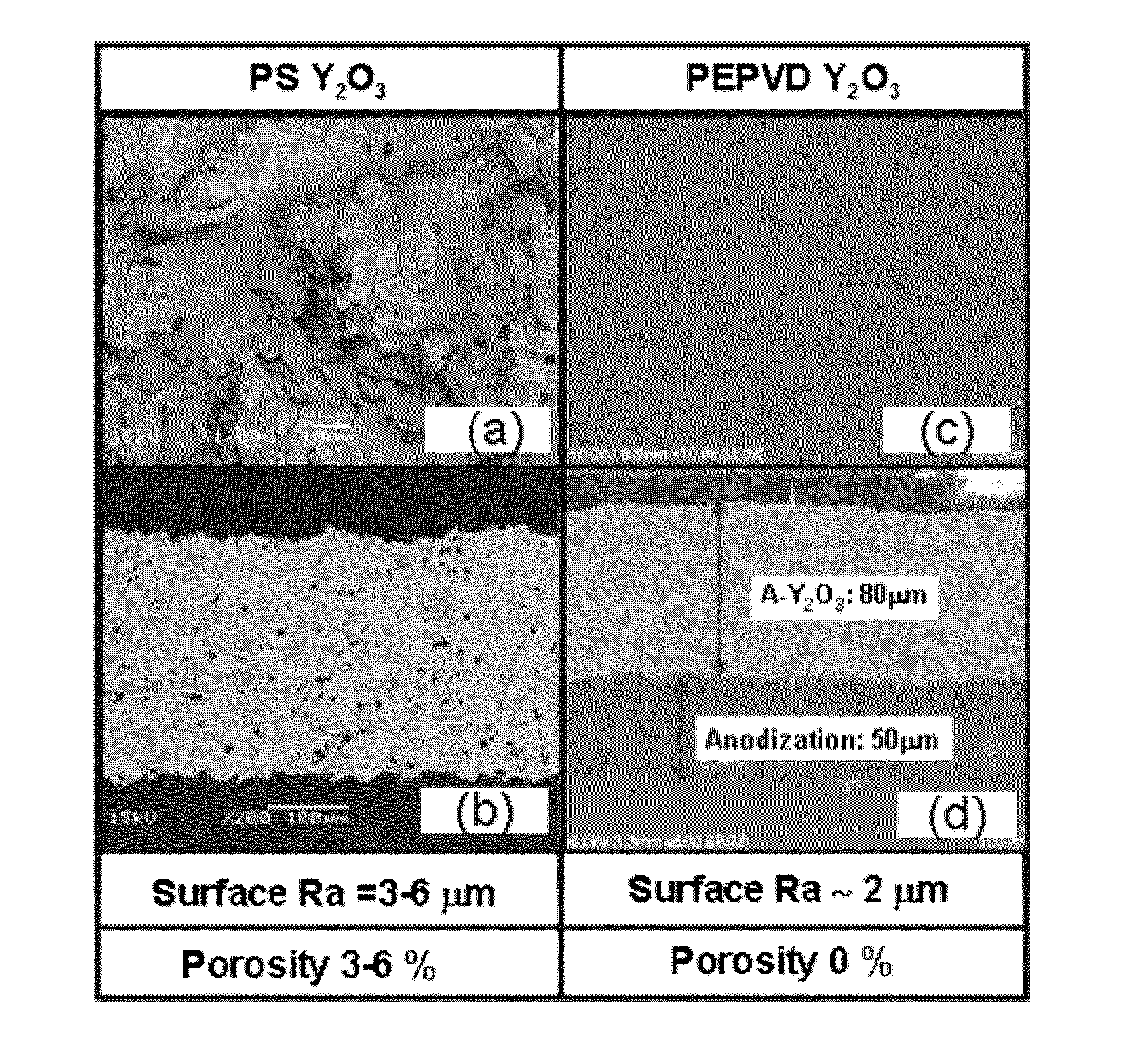
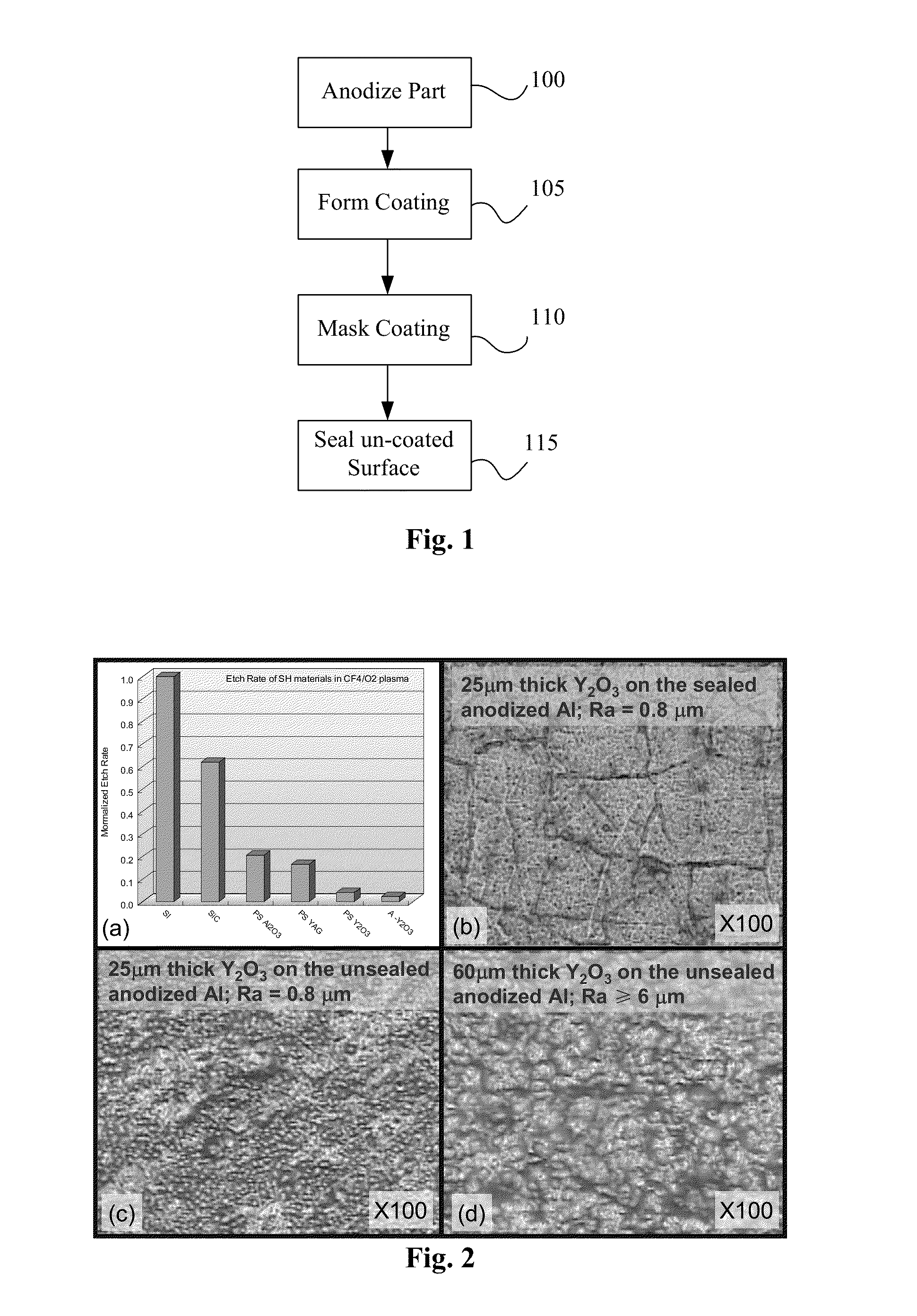
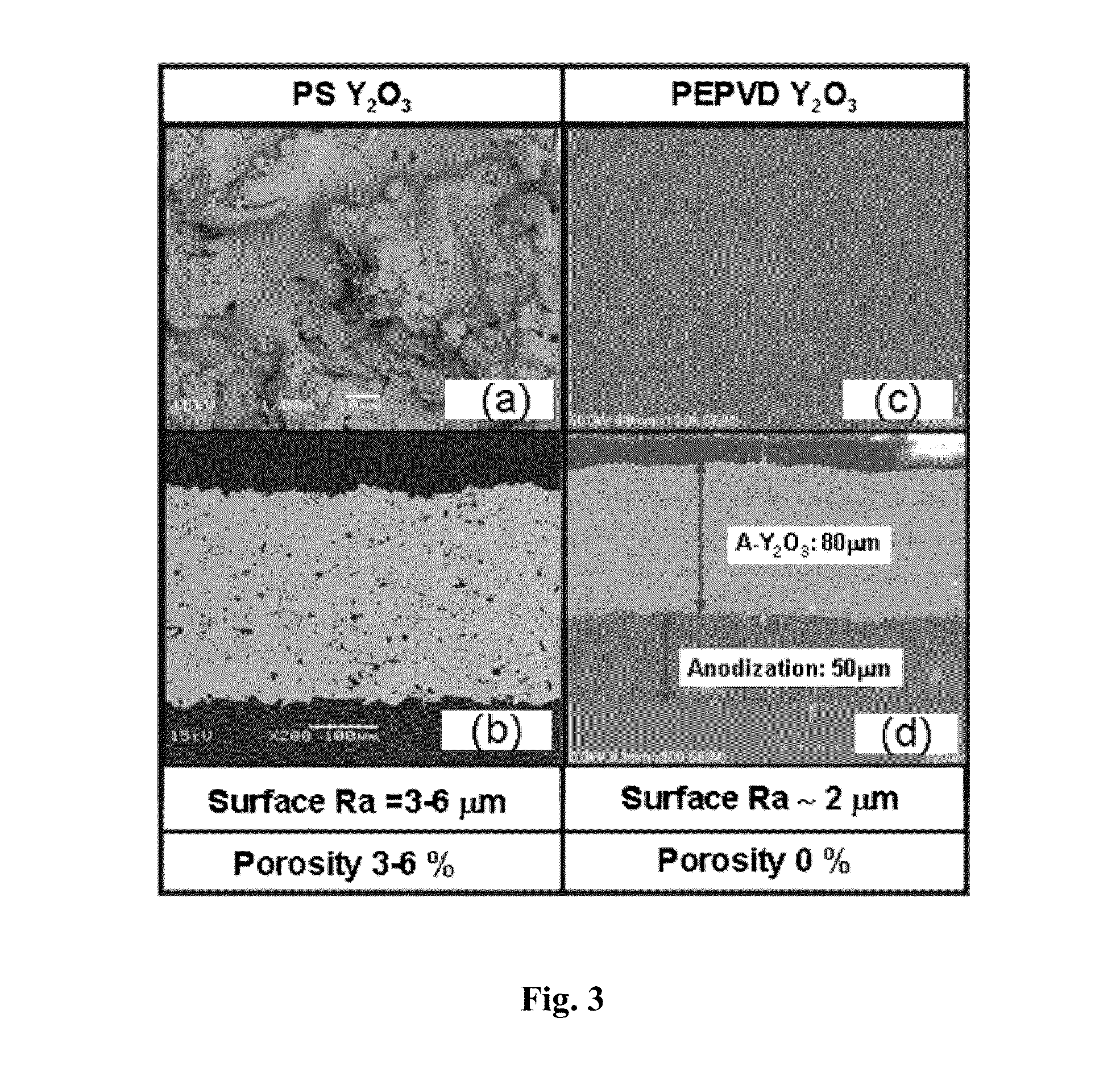
An advanced coating for parts used in plasma processing chamber. The advanced coating is formed over an anodized surface that has not been sealed. After the coating is formed, the coated area is masked, and the remaining anodized surface is sealed. The porous and rough structure of the anodized but un-sealed aluminum enhances adhesion of the coating. However, to prevent particle generation, the exposed anodized surface is sealed after formation of the coating. The coating can be of yttria, formed by plasma enhanced atomic deposition techniques which results in a dense and smooth coating.
Owner:ADVANCED MICRO FAB EQUIP INC CHINA
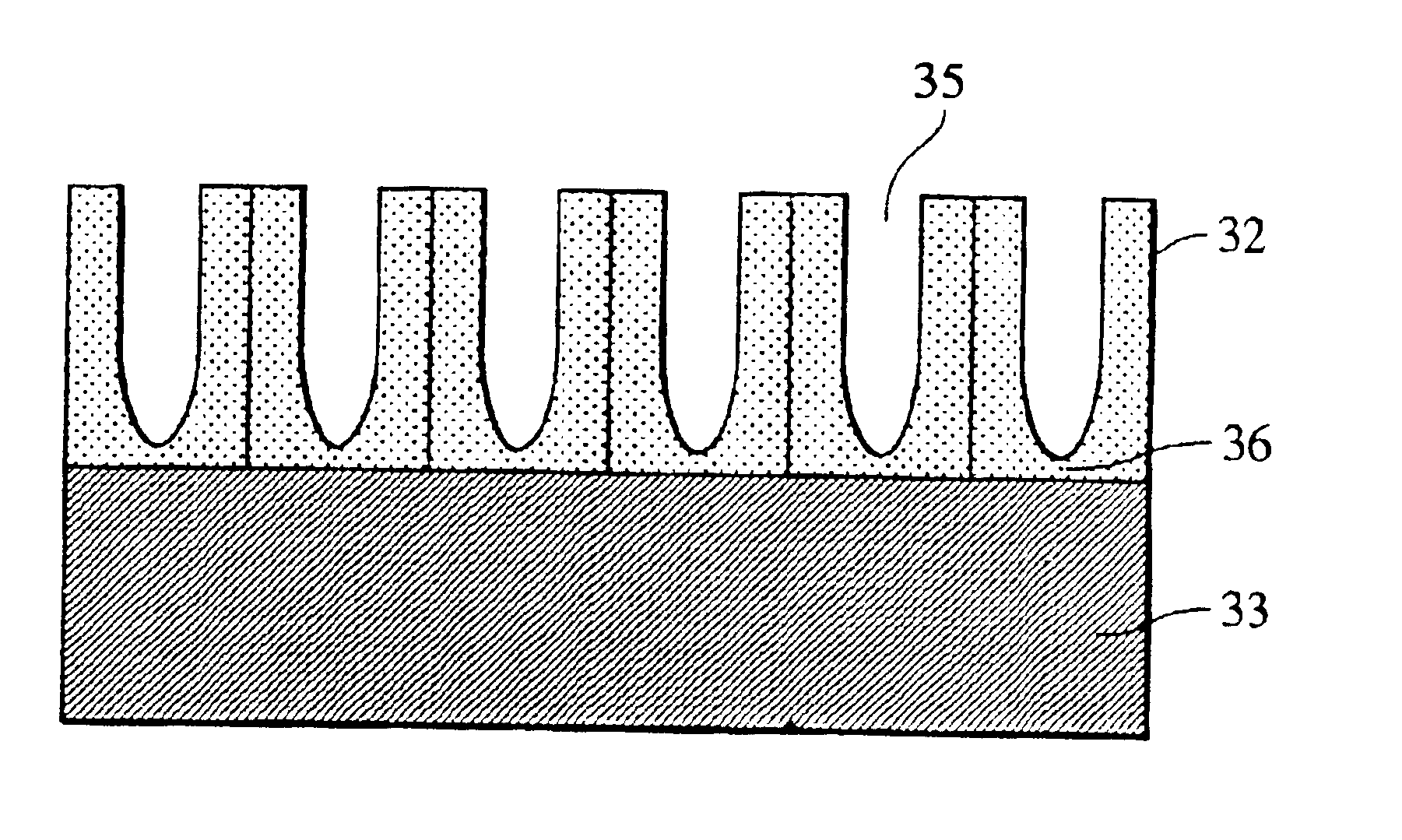
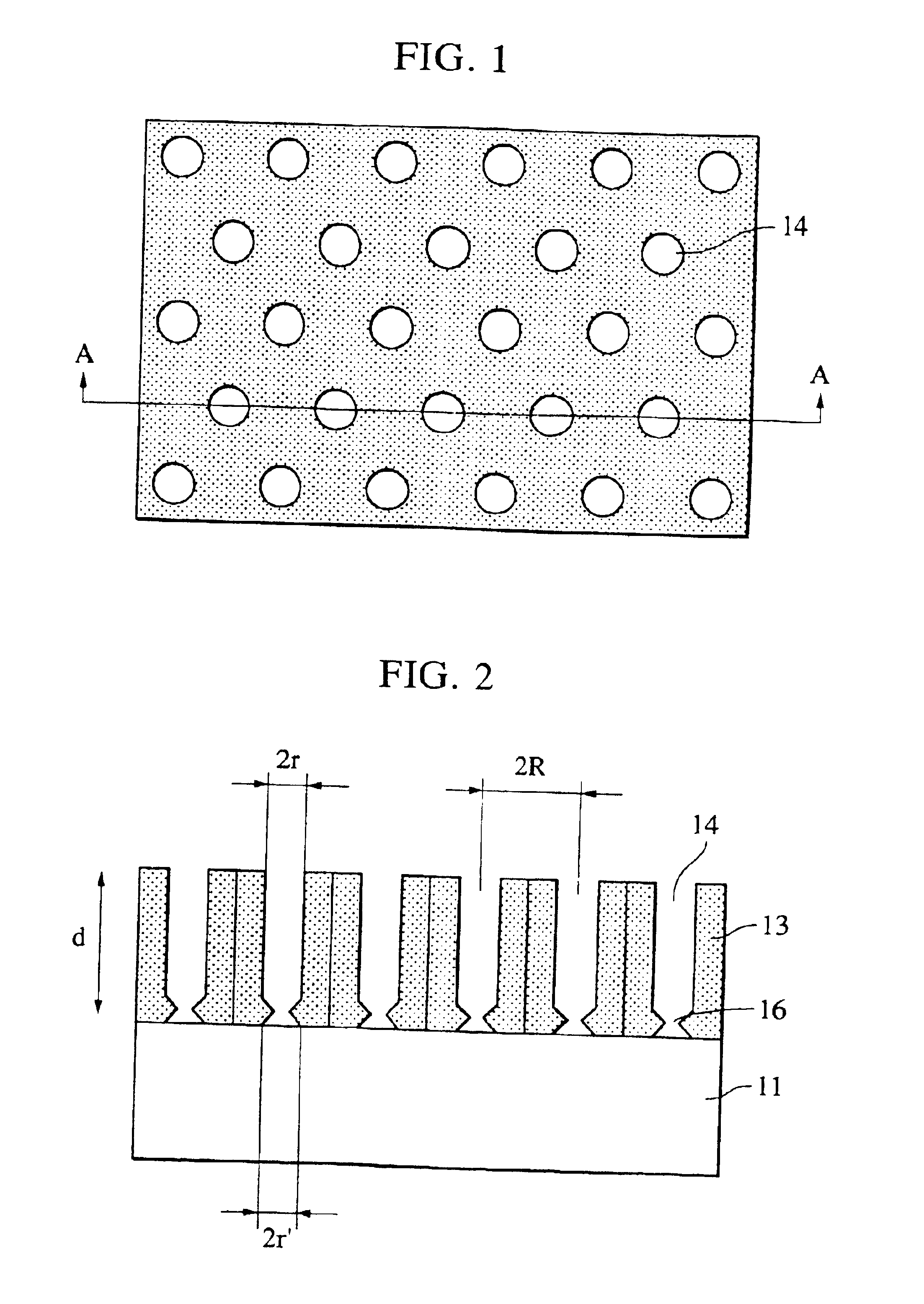
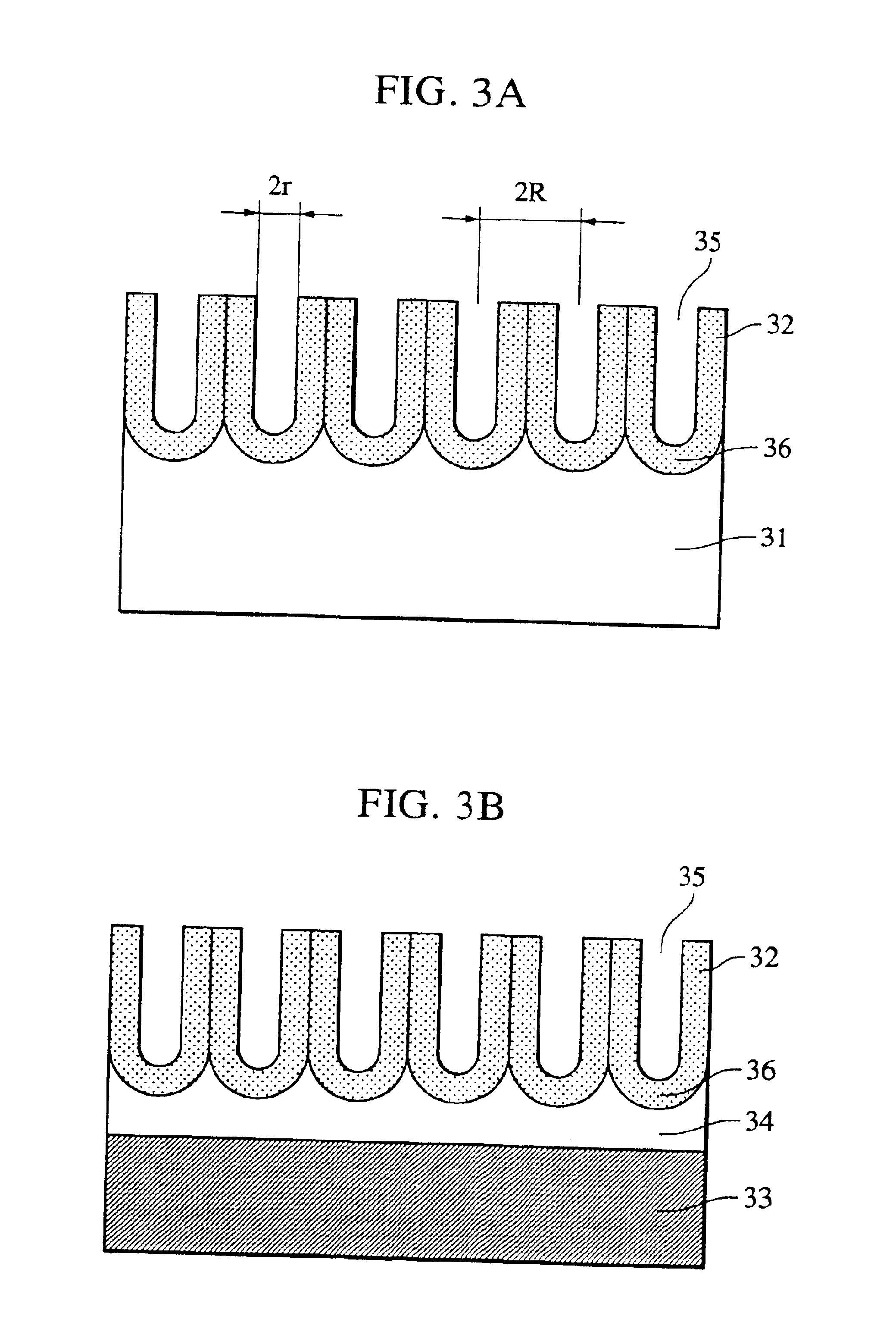
Composite of aluminium material and synthetic resin molding and process for producing the same
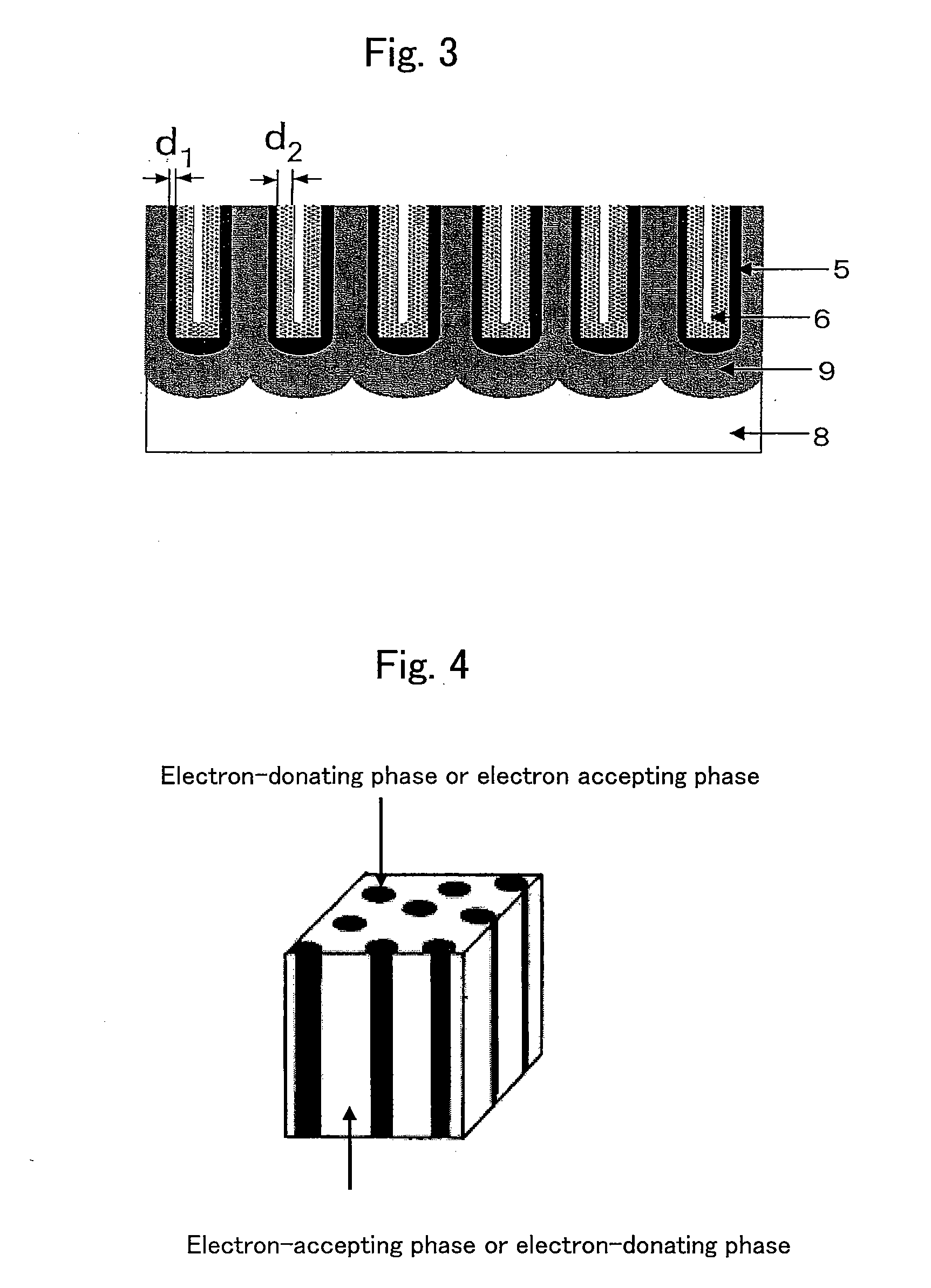
ActiveUS20060055084A1Stable and fast compositeHigh tensile strengthAnodisationNatural mineral layered productsO-Phosphoric AcidSynthetic resin
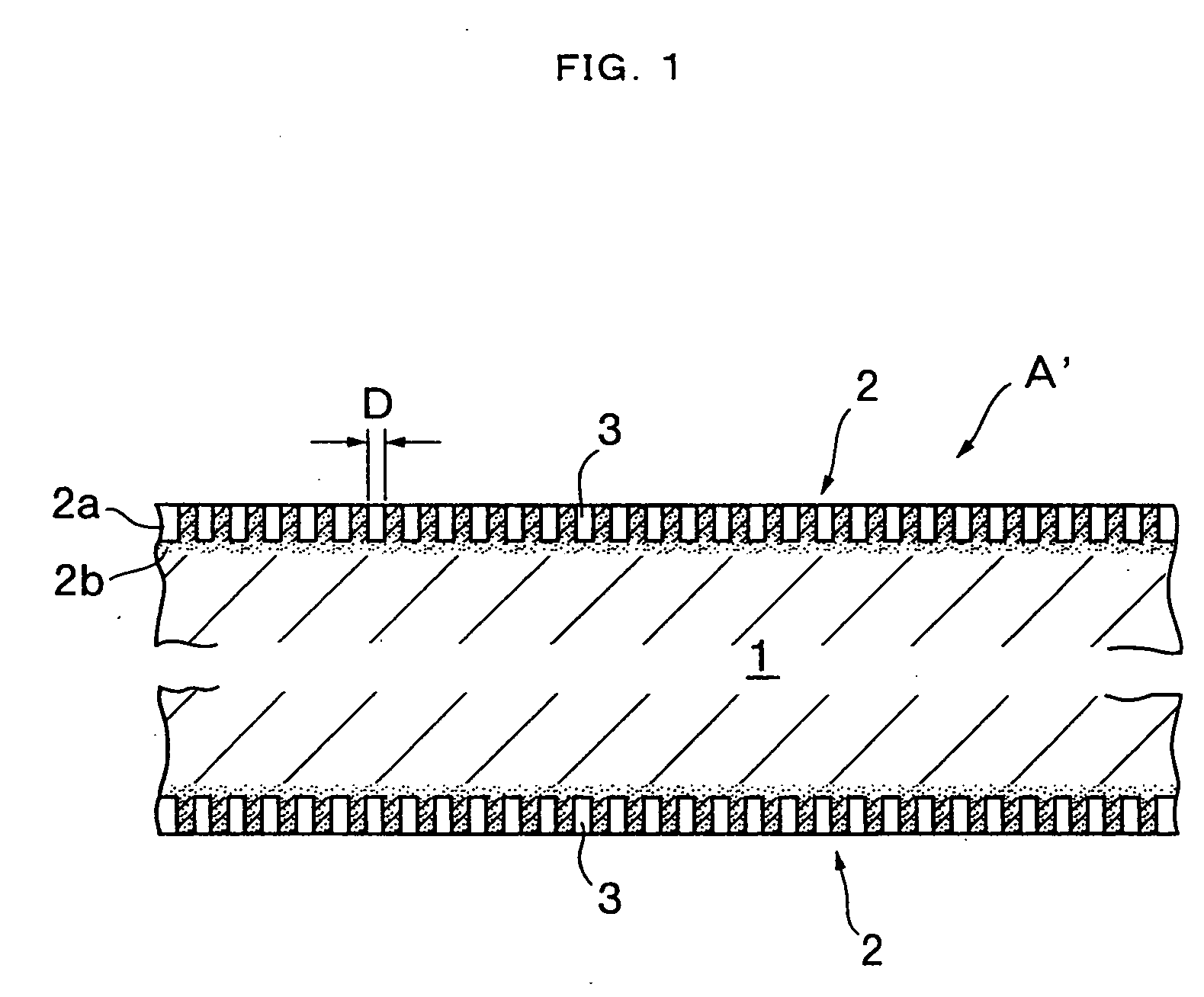
The present invention is to provide a process for producing a composite of an aluminum material and a synthetic resin molding that can be produced at a high efficiency and to provide a stable and fast composite that is large in a peel strength and a mechanical strength. The process for producing a composite according to the present invention is characterized in that an aluminum raw material is oxidized in an electrolytic bath of phosphoric acid or sodium hydride, thereby an anodic oxidation coating provided with innumerable pores 3 having a diameter of 25 nm or more made open in the surface thereof is formed thereon, and a synthetic resin mold 6 is coupled with the anodic oxidation coating 2 in such a condition that the part 6a thereof is intruded in the innumerable pores.
Owner:CHERRY CHIEF MAF CORP
Anodizing Aluminum and Alloys Thereof
This invention encompasses methods of producing a colored oxide layer on an aluminum material by anodizing the aluminum material in an electrolyte comprising water, sulfuric acid and oxalic acid. The anodizing step comprises passing at least two sequential current densities through the electrolyte. Methods of making and using article with a colored oxide layer on an aluminum material make by the methods disclosed herein are also disclosed.
Owner:DURACOUCHE INT
Nanostructure, electron emitting device, carbon nanotube device, and method of producing the same
InactiveUS6838297B2Good shape uniformityReliably producedNanostructure manufactureNanomagnetismNanoholeCarbon nanotube
Owner:CANON KK
Processing technique of cellphone shell with metal luster
ActiveCN104607884AGuaranteed StrengthReduce processing costsCoatingsNumerical controlAbrasive blasting
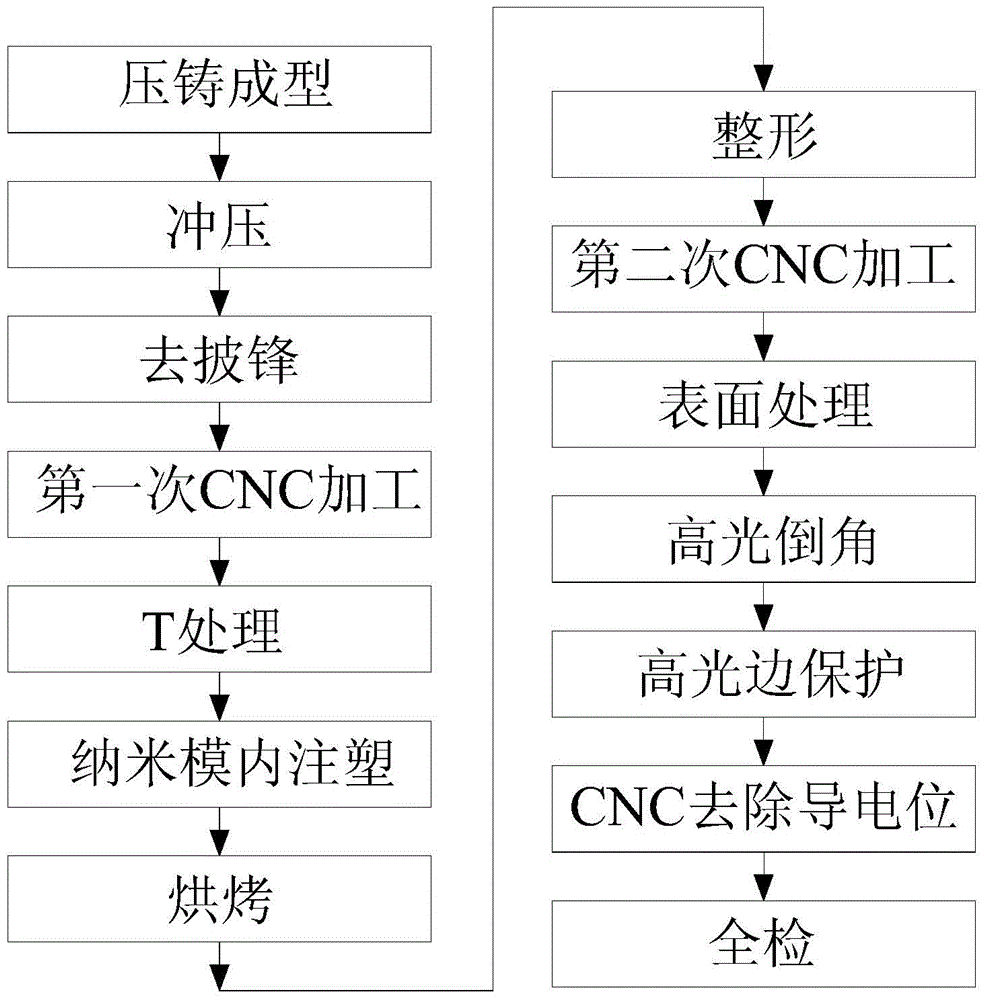
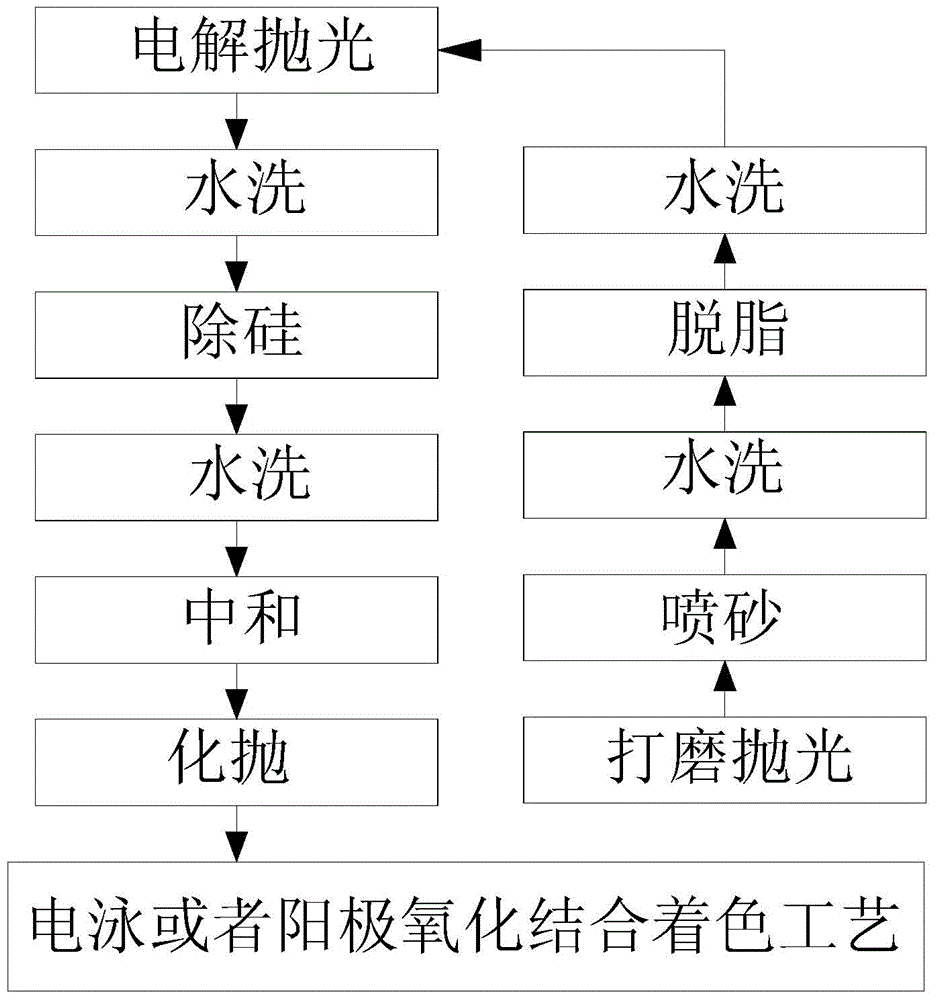
The invention provides a processing technique of a cellphone shell with metal luster. The processing technique includes the steps of S1, performing compression molding; S2, performing stamping; S3, performing primary CNC (computer numerical control) machining; S4, performing T treatment; S5, performing injection molding in a nano mold; S6, performing secondary CNC machining; S7, performing surface treatment, to be specific, performing a, polishing, b, shot-blasting, c, degreasing, d, washing, e, electrolytic polishing, f, washing, g, silicon removal aiming to decrease silicon content of the polished surface of the cellphone shell, h, neutralizing, i, chemical polishing, and j, electrophoresis or combination of anodic oxidation and coloring, aiming to impart metal luster to the polished surface of the cellphone shell. The compete cellphone shell is made through the steps S1 to S6, the silicon content of the polished surface of the cellphone shell is processed, the cellphone shell never produces compounds of intervals such as Al-Mg-Si-Fe during following anodic oxidation, defects such as oxide film darkening, bluing or milky-whiting are avoided, and the metal-lustered cellphone shell is manufactured through the complete process.
Owner:GUANGDONG OPPO MOBILE TELECOMM CORP LTD
Apparatus and process for mediated electrochemical oxidation of materials
A unique apparatus unique apparatus and process that uses mediated electrochemical oxidation (MEO) for: (1) Destruction of: a) nearly all organic solid, liquid, and gases materials, except fluorinated hydrocarbons; b) all biological solid, liquid, and gases materials; c) and / or dissolution and decontamination (such as cleaning equipment and containers, etc.) of nearly all inorganic solid, liquid, or gas where higher oxidation states exist which includes, but is not limited to, halogenated inorganic compounds (except fluorinated), inorganic pesticides and herbicides, inorganic fertilizers, carbon residues, inorganic carbon compounds, mineral formations, mining tailings, inorganic salts, metals and metal compounds, etc.); and d) combined materials (e.g. a mixture of any of the foregoing with each other); henceforth collectively referred to as materials. (2) Sterilization / disinfection of equipment, glassware, etc., by destroying all existing infectious materials. (3) Dissolution of transuranic / actinide materials and / or destruction of the oxidizable components in the hazardous waste portion of mixed waste. (4) Generation of hydrogen and oxygen from MEO of materials. (5) Alteration of organic, biological, and inorganic materials by MEO to produce other compounds from these materials. The materials are introduced into an apparatus for contacting the materials with an electrolyte containing the oxidized form of one or more reversible redox couples, at least one of which is produced electrochemically by anodic oxidation at the anode of an electrochemical cell. The oxidized forms of any other redox couples present are produced either by similar anodic oxidation or reaction with the oxidized form of other redox couples present and capable of affecting the required redox reaction. The oxidized species of the redox couples oxidize the materials molecules and are themselves converted to their reduced form, whereupon they are reoxidized by either of the aforementioned mechanisms and the redox cycle continues until all oxidizable material species, including intermediate reaction products, have undergone the desired degree of oxidation. The entire process takes place at temperatures between ambient and approximately 100° C. The oxidation process may be enhanced by the addition of reaction enhancements, such as: ultrasonic energy and / or ultraviolet radiation.
Owner:SCIMIST LNC
Damascene integration scheme for developing metal-insulator-metal capacitors
InactiveUS20040113235A1Increase capacitanceSave chip areaTransistorSolid-state devicesMetal-insulator-metalGas phase
The invention is directed to unique high-surface area BEOL capacitor structures with high-k dielectric layers and methods for fabricating the same. These high-surface area BEOL capacitor structures may be used in analog and mixed signal applications. The capacitor is formed within a trench with pedestals within the trench to provide additional surface area. The top and bottom electrodes are created using damascene integration scheme. The dielectric layer is created as a multilayer dielectric film comprising for instance Al2O3, Al2O3 / Ta2O5, Al2O3 / Ta2O5 / Al2O3 and the like. The dielectric layer may be deposited by methods like atomic layer deposition or chemical vapor deposition. The dielectric layer used in the capacitor may also be produced by anodic oxidation of a metallic precursor to yield a high dielectric constant oxide layer.
Owner:GLOBALFOUNDRIES US INC
Semiconductor device and method for forming the same
InactiveUS6624450B1Improve flatnessImprove equipment reliabilityTransistorSolid-state devicesEtchingMetallic materials
In a thin-film insulated gate type field effect transistor having a metal gate in which the surface of the gate electrode is subjected to anodic oxidation, a silicon nitride film is provided so as to be interposed between the gate electrode and the gate insulating film to prevent invasion of movable ions into a channel, and also to prevent the breakdown of the gate insulating film due to a potential difference between the gate electrode and the channel region. By coating a specific portion of the gate electrode with metal material such as chrome or the like for the anodic oxidation, and then removing only the metal material such as chrome or the like together with the anodic oxide of the metal material such as chrome or the like, an exposed portion of metal gate (e.g. aluminum) is formed, and an upper wiring is connected to the exposed portion. Further, an aluminum oxide or silicon nitride is formed as an etching stopper between the gate electrode and the gate insulating film or between the substrate and the layer on the substrate, so that the over-etching can be prevented and the flatness of the element can be improved. In addition, a contact is formed in no consideration of the concept "contact hole".
Owner:SEMICON ENERGY LAB CO LTD
Article of manufacture and process for anodically coating an aluminum substrate with ceramic oxides prior to polytetrafluoroethylene or silicone coating
InactiveUS20050115840A1AnodisationElectrolytic inorganic material coatingCeramic coatingSilicone coating
Owner:HENKEL KGAA
Strong anodic oxidation method for preparing porous pellumina
InactiveCN101139730AReduce pollutionRich sourcesSurface reaction electrolytic coatingShielding gasPhosphoric acid
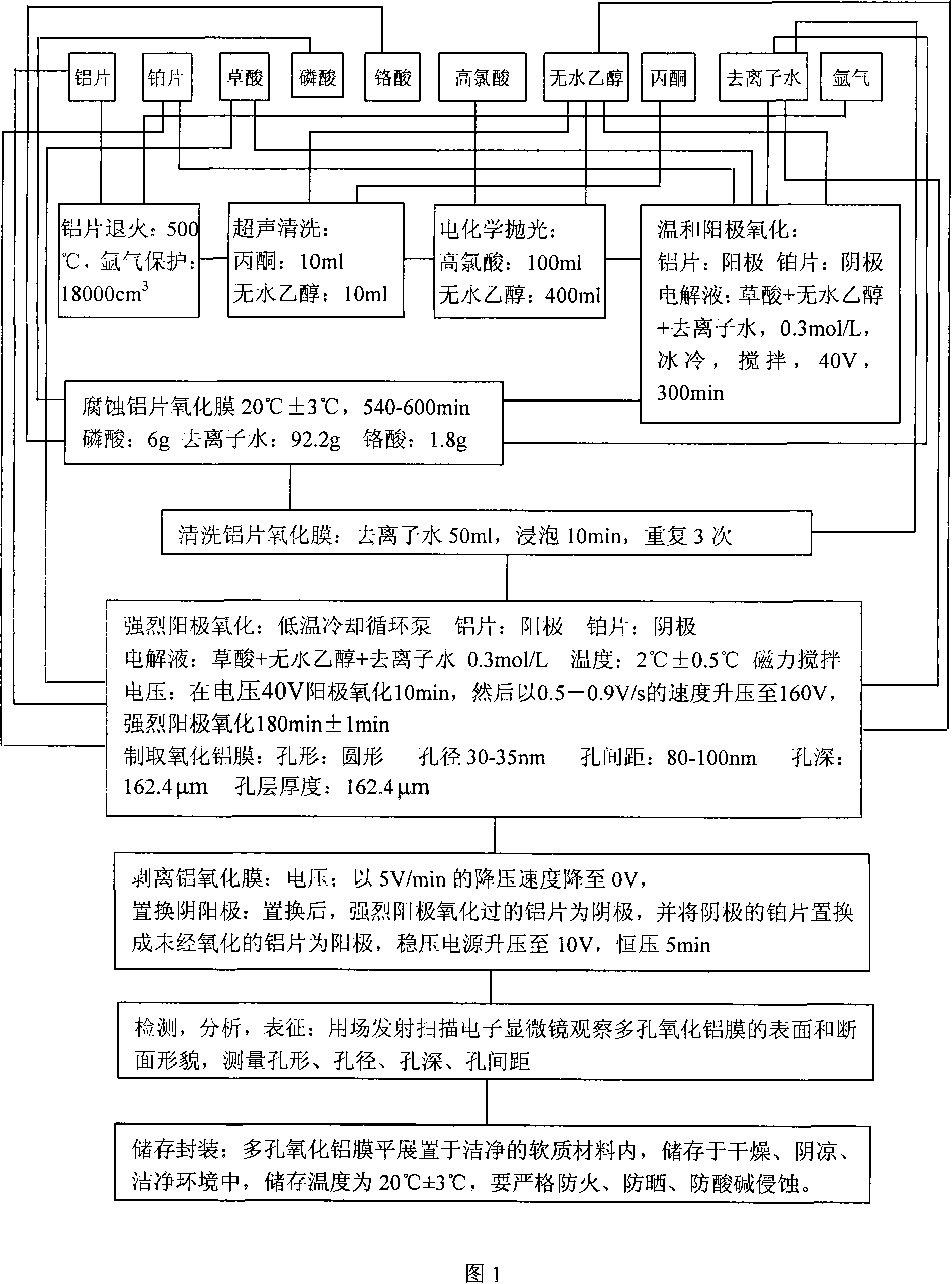
The invention relates to an intensive anodic oxidation process to prepare porous anodic alumina film. An aluminum sheet is as an anode; a platinum sheet as a cathode; argon as shielded gas; mixed solution of perchloric acid with absolute alcohol as electrochemical polishing compound for the aluminum sheet; mixed solution of oxalic acid, absolute alcohol and deionized water as electrolyte; and mixed solution of phosphoric acid, chromic acid and deionized water as alumina film corrodent. Through aluminum sheet annealing, purging, electrochemical polishing, mild anodic oxidation, corrosion on aluminum sheet oxide film, intensive anodic oxidation and stripping oxide film, and finally, pale yellow, high purity and ordered porous-structured nanometer-leveled alumina film is achieved; The alumina film pore is in circular shape sized in 30 to 35nm, pore distance in 80 to100nm and pore depth in 162.4 micrometers; growth rate of the alumina film is 54 micrometers / h, which is 27 times higher than 2 micrometers / h of growth rate of anodic oxidation aluminum film prepared through mild anodic oxidation process.
Owner:TAIYUAN UNIV OF TECH
Substrate for solar cell and solar cell
InactiveUS20100218827A1Good pressure characteristicsImprove adhesionAnodisationPhotovoltaic energy generationSolar cellAnodic oxidation
A substrate for a solar cell having a metal substrate, a first insulating oxide film formed on the metal substrate by anodic oxidation, and a second insulating film, wherein the first insulating oxide film has pores and the pores are sealed with the second insulating film at a sealing ratio of 5 to 80%.
Owner:FUJIFILM CORP
Cathode for Use in a Wet Capacitor
A cathode containing a metal substrate that possesses a micro-roughened surface imparted by spark anodization is provided. The surface is formed by contacting the substrate with an electrolytic solution and applying a voltage to form a dielectric sub-oxide layer. The voltage is raised to a sufficiently high level to initiate “sparking” at the surface of the substrate, which is believed to create high local surface temperatures sufficient to etch away the substrate. This results in the formation of a “micro-roughened” surface having a plurality elevated regions. These elevated regions can increase the effective surface area and thus allow for the formation of capacitors with increased cathode capacitance for a given size and / or capacitors with a reduced size for a given capacitance. The elevated regions may also exhibit excellent adhesion to additional electrochemically-active materials and provide enhanced stability in certain liquid electrolytes.
Owner:AVX CORP
Wet electrolytic capacitor containing a gelled working electrolyte
ActiveUS9105401B2Hybrid capacitor electrolytesCapacitor electrolytes/absorbentsElectrolysisConductive polymer
A wet electrolytic capacitor is provided. The capacitor contains an anode comprising an anodically oxidized pellet formed from a pressed and sintered powder, a cathode that contains a metal substrate coated with a conductive polymer, and a working electrolyte in communication with the anode and the cathode. The working electrolyte is in the form of a gel and comprises an ammonium salt of an organic acid, inorganic oxide particles, an acid, and a solvent system that comprises water. The working electrolyte has a pH value of from about 5.0 to about 8.0.
Owner:KYOCERA AVX COMPONENTS CORP
Biodegradable Magnesium Based Metallic Material for Medical Use
InactiveUS20090131540A1Suppressing deterioration of mechanical strengthImprove compatibilityBiocideInorganic non-active ingredientsRetention timeMetallic materials
A biodegradable magnesium based metallic material for medical use which is degraded and absorbed in vivo, characterized by comprising a film, which contains magnesium oxide and magnesium hydroxide and is formed on the surface of crystallized magnesium or a magnesium alloy by anodic oxidation. This magnesium based metallic material is capable of exhibiting desired mechanical properties such as strength and ductility at an early stage of implantation without changing the mechanical properties inherent to magnesium or its alloy and also controlling the retention time of the mechanical properties to be short or long in a desired manner.
Owner:NAT INST FOR MATERIALS SCI
Stamping tool, casting mold and methods for structuring a surface of a work piece
InactiveUS7066234B2Simple and cost-effectiveProlong lifeAnodisationMoulding toolsSurface layerCasting mold
A simple, cost-effective stamping or molding in the nanometer range is enabled using a stamping surface or molding face with a surface layer having hollow chambers that have been formed by anodic oxidation.
Owner:SHARP KK
Process method for preparing ultra-hydrophobic surface by electrochemical method
ActiveCN101665968AEndows the superior performance of "self-cleaning"Apparent nanostructureAnodisationSpecial surfacesPhysical chemistryElectrochemistry
The invention discloses a process method for preparing an ultra-hydrophobic surface by the electrochemical method. The process adopts two processing steps: forming the micro nanometer double-structurerough surface by first electrochemical etching and then oxalic acid anodic oxidation; and preparing the ultra-hydrophobic surface by modifying the surface with fluorosilane. The method is simple andpractical, uses mature electrochemical etching and anodic oxidation technology, allows for easy mass production and avoids environmental pollution.
Owner:甘肃中安瑞科科技有限公司
Method for improving alkaline resistance of anode oxide film on surface of aluminum or aluminum alloy material
InactiveCN104233428AGood alkali resistanceImprove corrosion resistanceAnodisationOrganic acidSilanes
The invention discloses a method for improving the alkaline resistance of an anode oxide film on the surface of an aluminum or aluminum alloy material. The method comprises the following steps: performing polishing pretreatment on the aluminum or aluminum alloy material, performing anodic oxidation treatment in electrolytic liquid containing organic acid, inorganic acid and soluble oxysalt, performing closing treatment on an oxide film obtained after the anodic oxidation treatment, and forming a silane film layer on the surface of the aluminum or aluminum alloy material subjected to the closing treatment. The method for surface treatment of the aluminum or aluminum alloy material is easy to perform, economic, environment-friendly and capable of effectively improving the alkaline resistance of the anode oxide film on the surface of the aluminum or aluminum alloy material.
Owner:HUNAN UNIV
Method for preparing metal titanium or titanium alloy super-oleophobic surface
InactiveCN102021628AImprove stabilityReduce resistanceSurface reaction electrolytic coatingSpecial surfacesFine structureTitanium alloy
The invention discloses a method for preparing a metal titanium or titanium alloy super-oleophobic surface. The method comprises the following steps of: performing primary anodic oxidation treatment on metal titanium or titanium alloy to obtain a roughened surface with a microstructure; forming a titanium dioxide nanotube array film on the surface with microstructure through secondary anodic oxidation so as to obtain a composite fine structured micro-nanostructure; and modifying with a low-surface-energy substance to obtain the super-oleophobic surface and the super-hydrophobic and super-oleophobic surface. The metal titanium or titanium alloy surface has super-oleophobic and super-hydrophobic characteristics for multiple kinds of organic liquid, the static contact angle is greater than 155 degrees; the rolling angle is less than 10 degrees; meanwhile, the surface also shows superior super-oleophobic and super-hydrophobic characteristics for pure water and aqueous solution of acid, alkali and salt.
Owner:LANZHOU INST OF CHEM PHYSICS CHINESE ACAD OF SCI
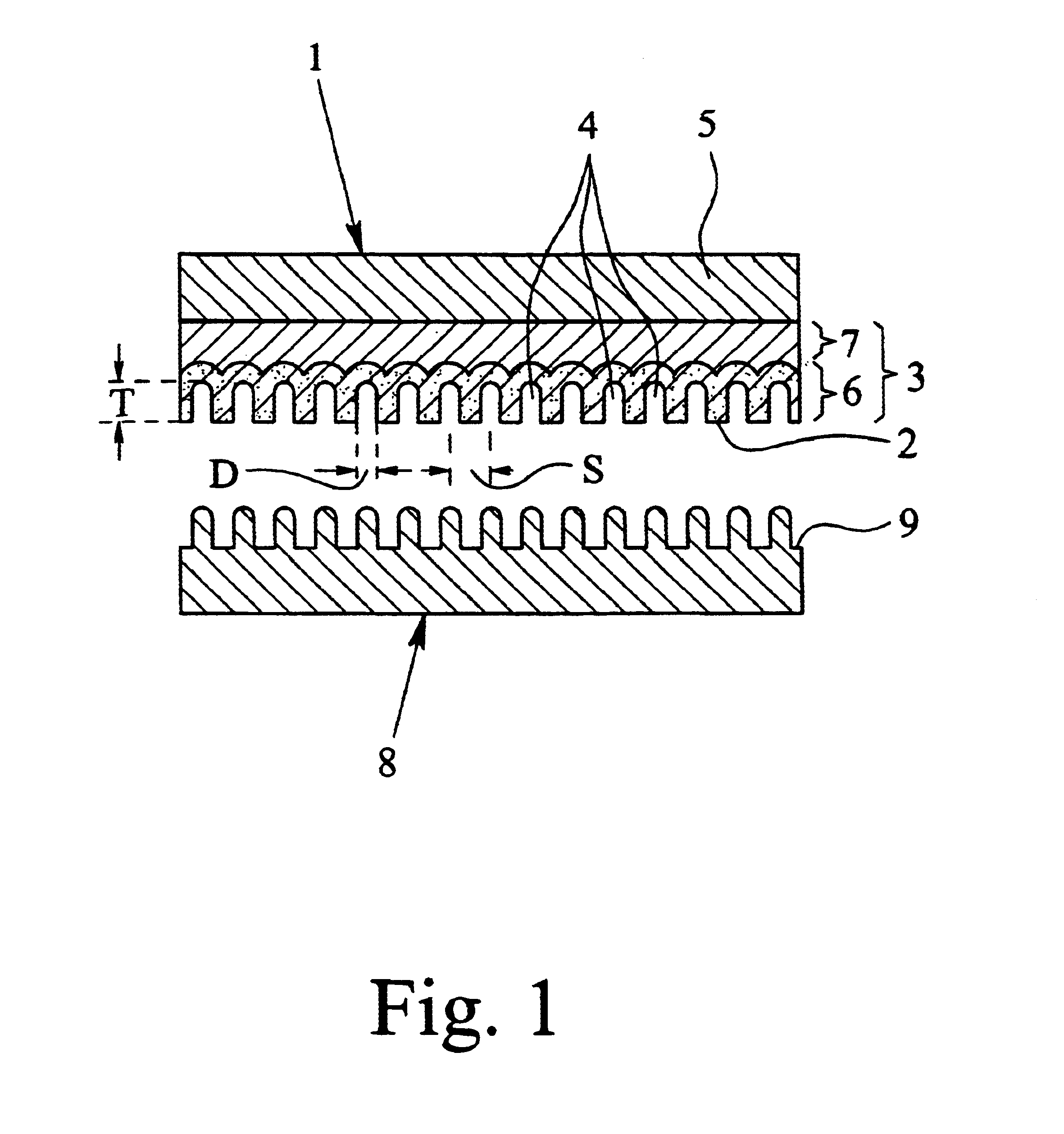
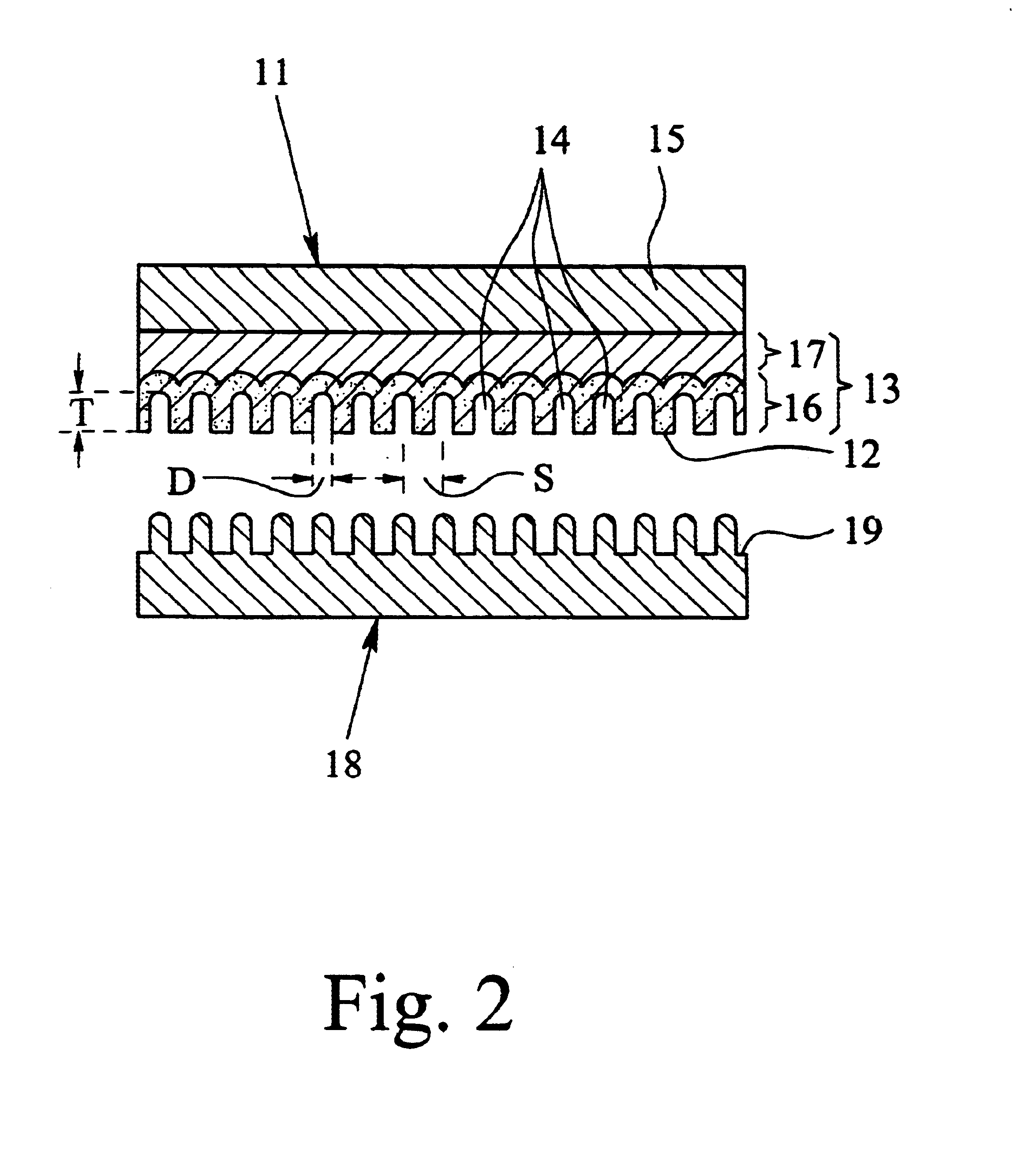
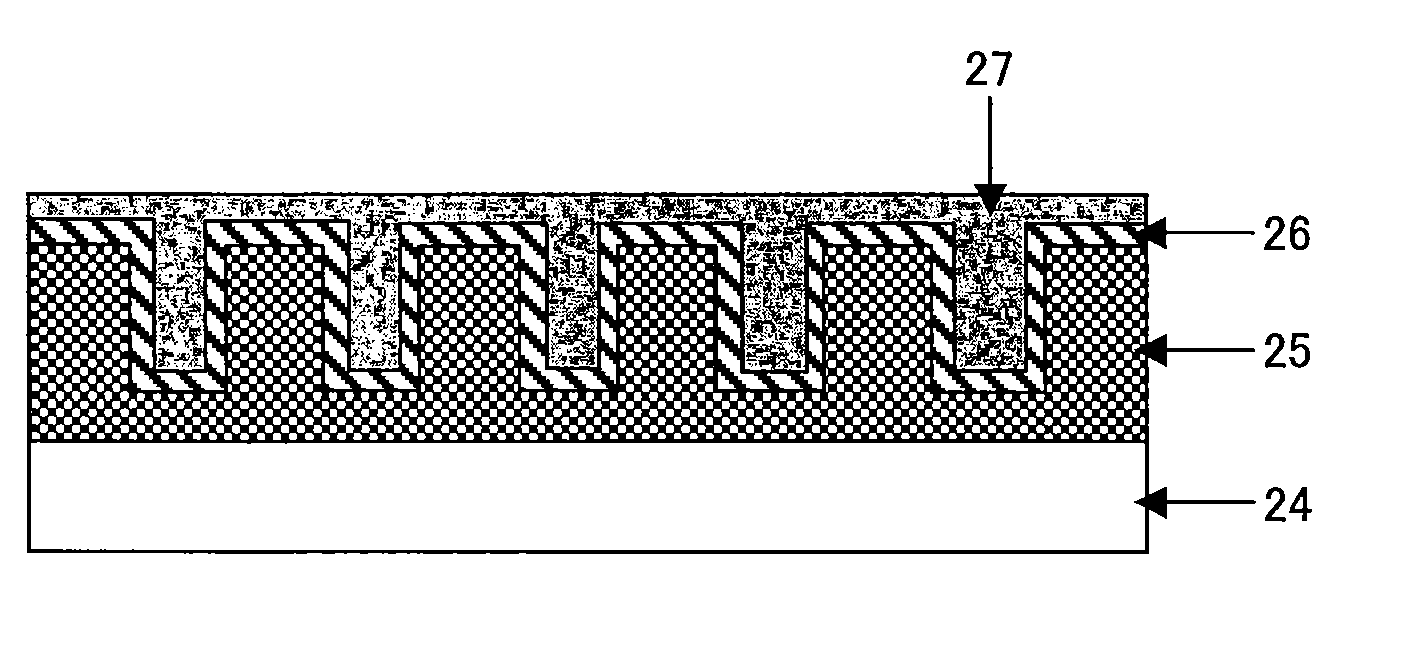
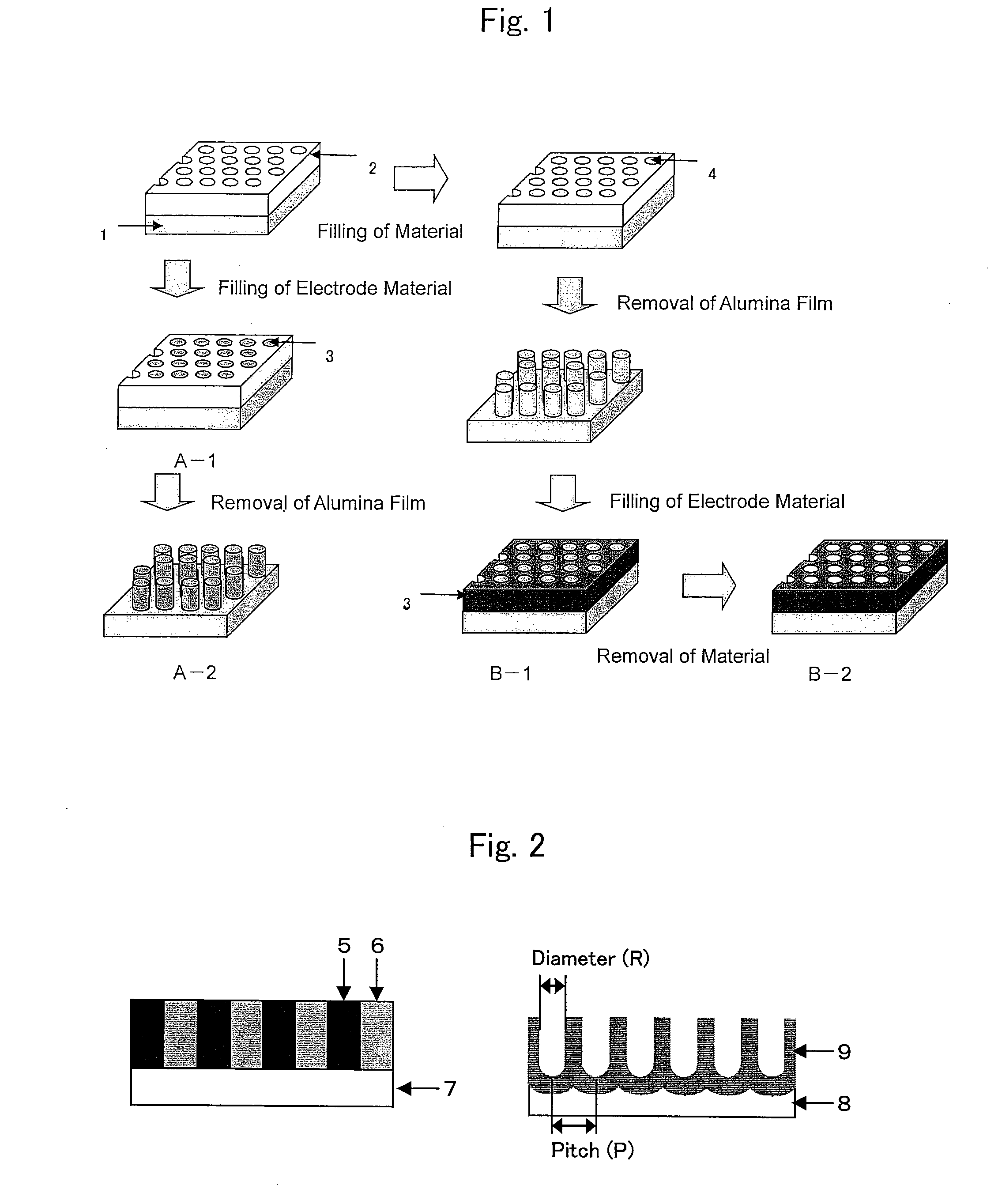
Method for manufacturing nano-array electrode and photoelectric conversion device using same
InactiveUS20060234505A1Improve efficiencyEfficient charge separationAnodisationFinal product manufactureNano structuringPhotoelectric conversion
The present invention provides a method of manufacturing a nano-array electrode with a controlled nano-structure by filling an electrode material into the fine pores of an anodic-oxide porous alumina film obtained by anodically oxidizing aluminum in electrolyte, or by filling a material into the fine pores of an anodic-oxide porous alumina film obtained by anodically oxidizing aluminum in electrolyte and then filling an electrode material into the spaces defined by the nano-array formed by removing the anodic-oxide porous alumina film, or by filling repeatedly an electrode material in the fine pores of the anodic-oxide porous alumina film to fill a plurality of electrode materials. A high-performance, high-efficiency photoelectric converting device comprising a nano-array electrode manufactured by the method is also disclosed.
Owner:NIPPON OIL CORP
Composite body of aluminum or aluminum alloy and plastic and manufacturing method thereof
InactiveCN102268183AImprove bindingAnodisationMetallic material coating processesThermoplasticMetallurgy
Owner:HONG FU JIN PRECISION IND (SHENZHEN) CO LTD +1
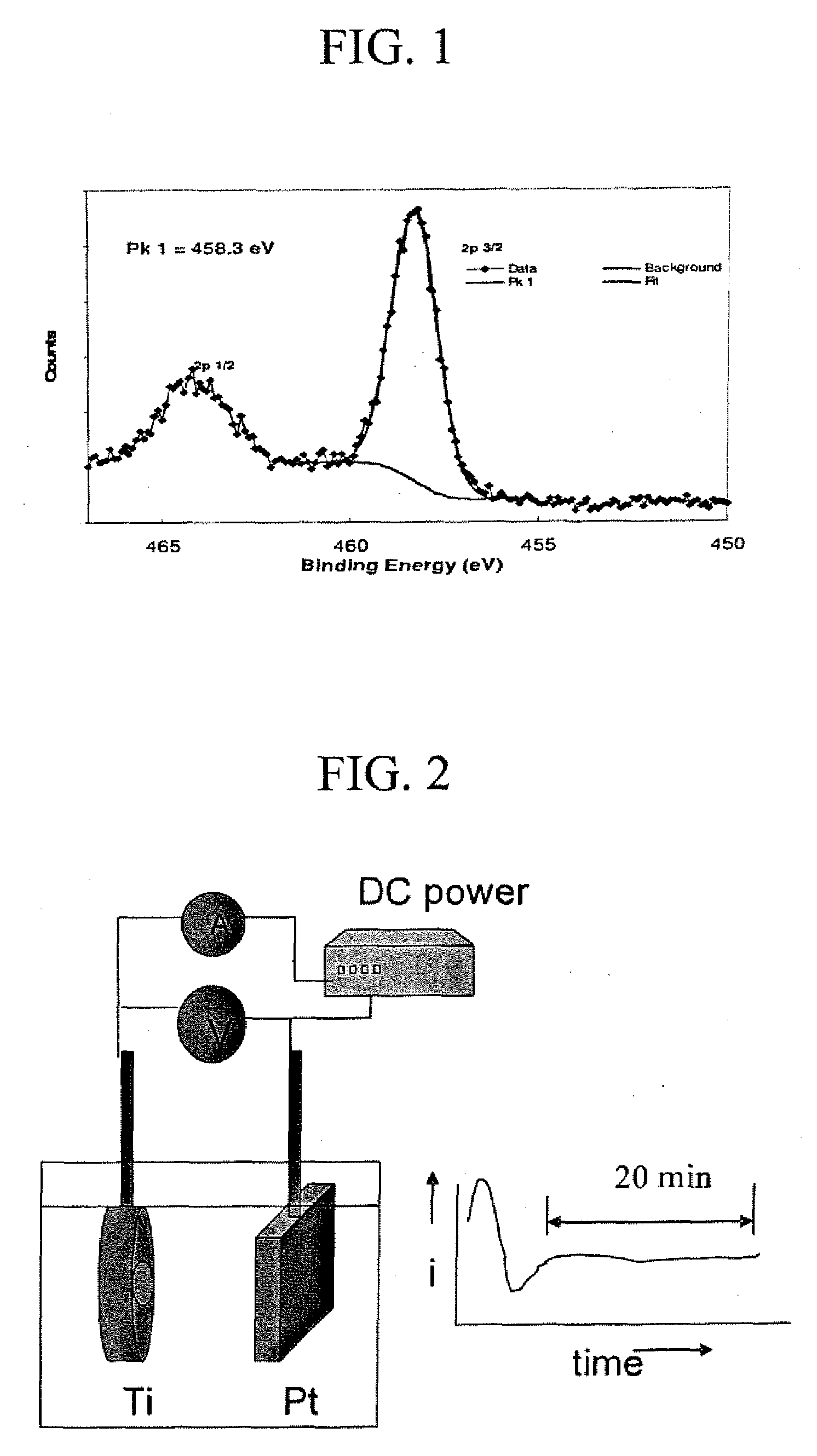
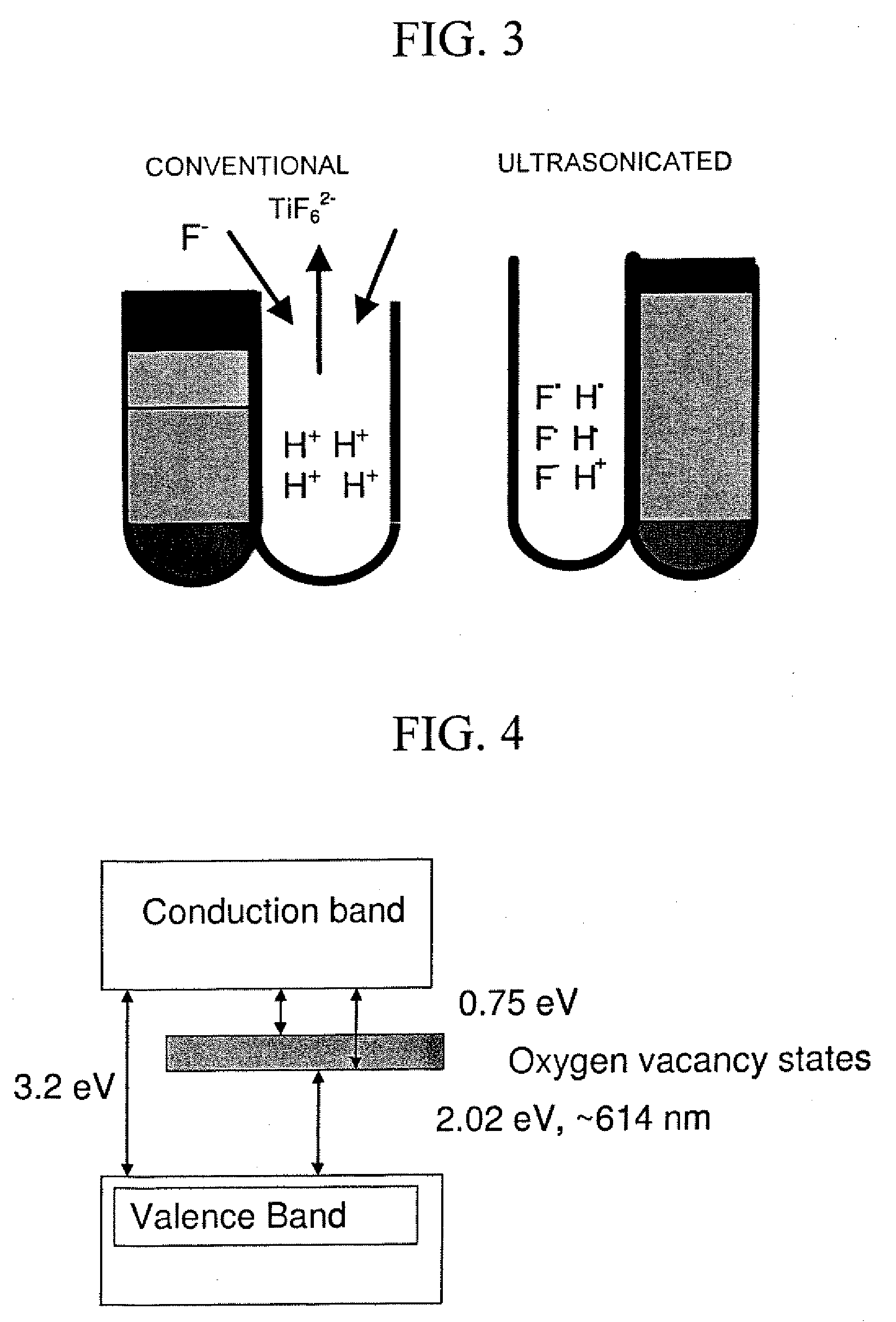
Preparation of nano-tubular titania substrate with oxygen vacancies and their use in photo-electrolysis of water
The invention relates to a method of making a nanotubular titania substrate having a titanium dioxide surface comprised of a plurality of vertically oriented titanium dioxide nanotubes containing oxygen vacancies, including the steps of anodizing a titanium metal substrate in an acidified fluoride electrolyte and annealing the titanium oxide surface in a non-oxidating atmosphere. The invention further relates to a nanotubular titania substrate having an annealed titanium dioxide surface comprised of self-ordered titanium dioxide nanotubes containing oxygen vacancies. The invention further relates to a photo-electrolysis method for generating H2 wherein the photo-anode is a nanotubular titania substrate of the invention. The invention also relates to an electrochemical method of synthesizing CdZn / CdZnTe nanowires, wherein a nanoporous TiO2 template was used in combination with non-aqueous electrolyte. The invention also relates to a nanotubular titania substrate having CdTe or CdZnTe nanowires extending therefrom.
Owner:BOARD OF RGT NEVADA SYST OF HIGHER EDUCATION ON BEHALF OF THE UNIV OF NEVADA RENO
Self-cleaning superhydrophobic surface
ActiveUS20060147634A1Material nanotechnologyNanoinformaticsPhotocatalytic reactionUltraviolet lights
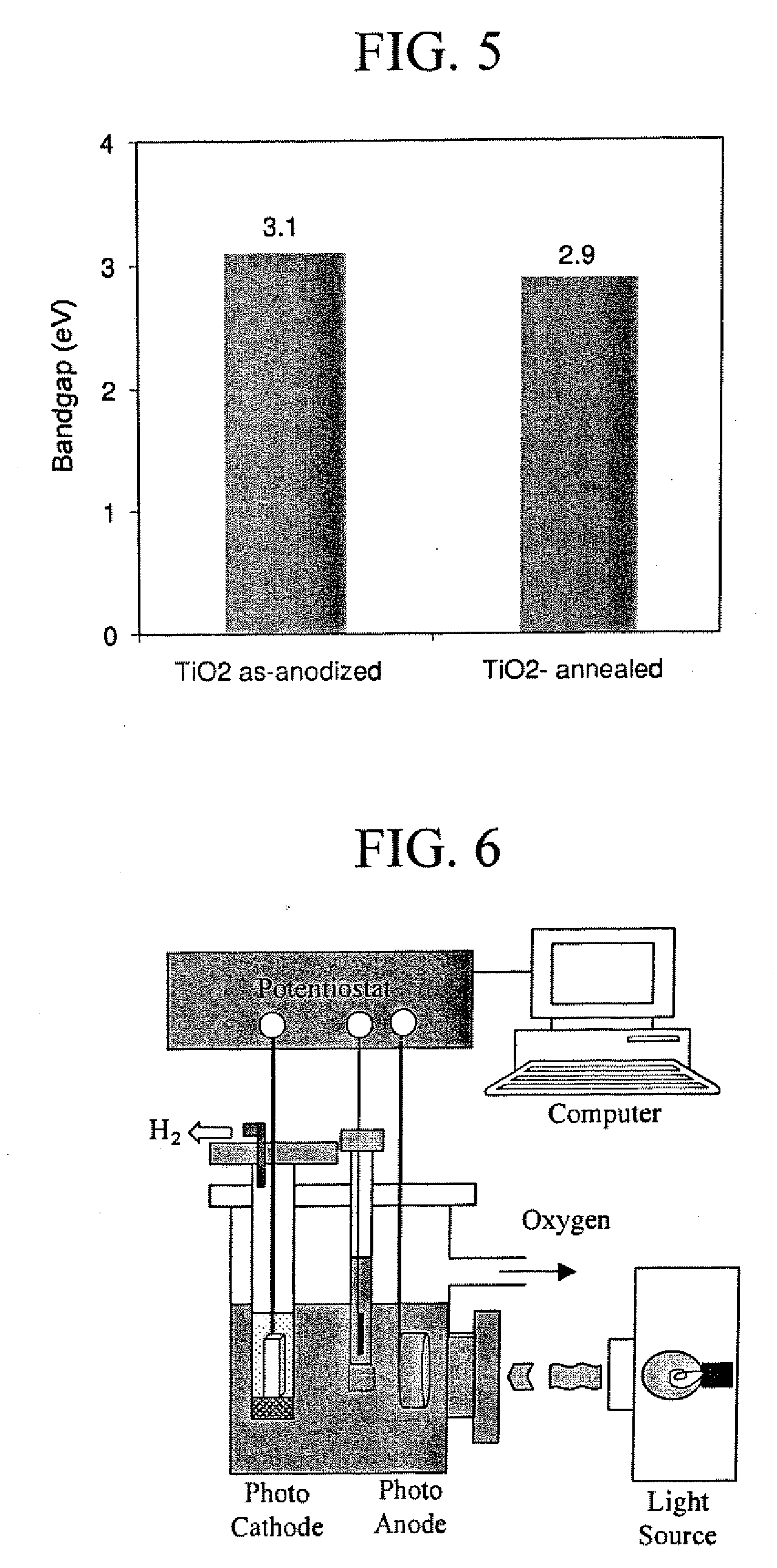
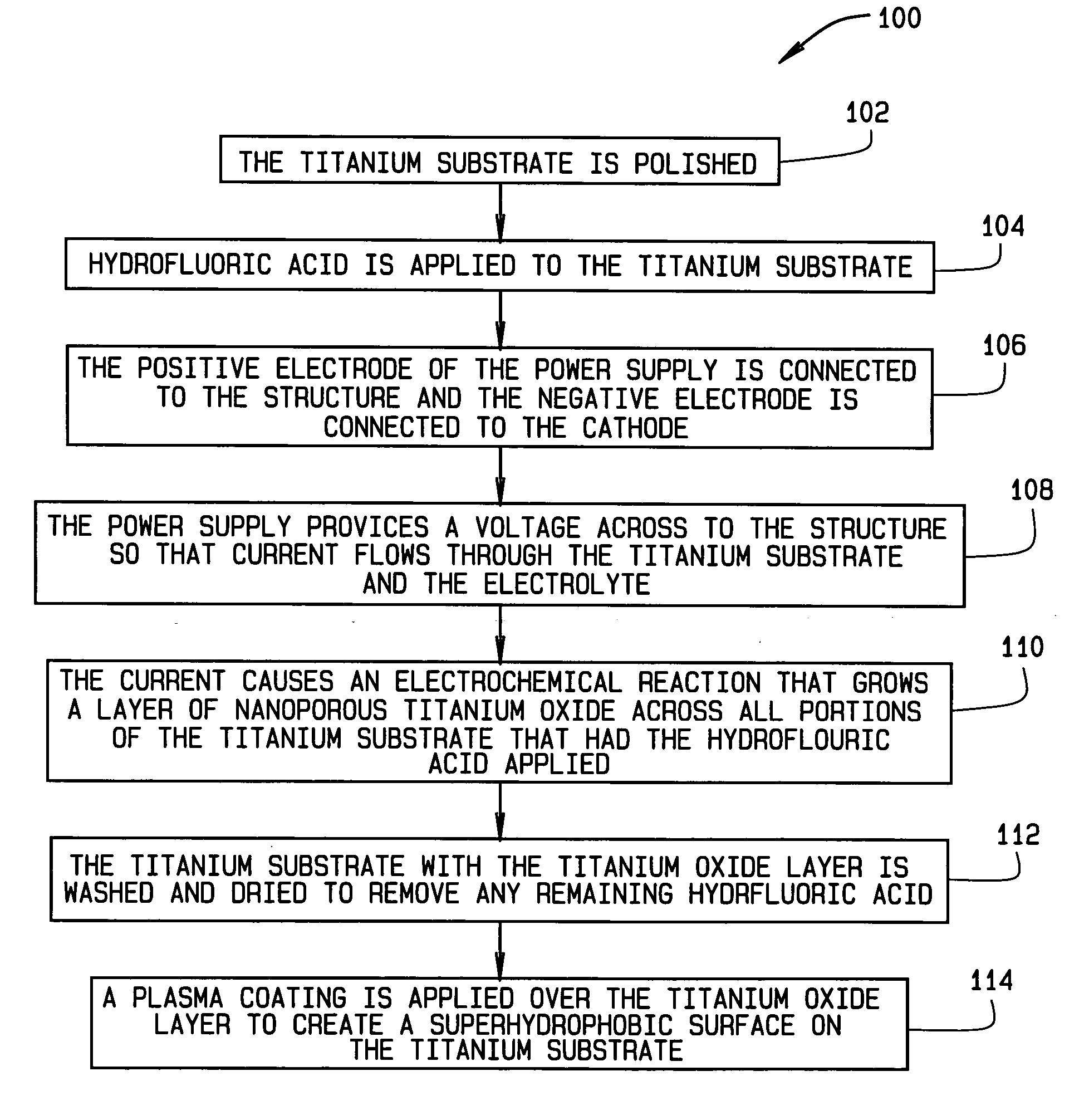
A method for providing a superhydrophobic surface on a structure, for example aircraft wings, propellers and / or rotors, is set forth. The method includes applying a coating of hydrofluoric acid over a titanium substrate. A voltage is then applied across the titanium substrate so that current flows through the titanium substrate. The current flowing through the titanium substrate causes the hydrofluoric acid to react with the titanium substrate to anodize the titanium substrate. The anodization causes a nanoporous titanium oxide layer to grow across the titanium substrate. The titanium oxide layer includes a plurality of nano-tube structures that, once the remaining hydrofluoric acid is washed away, provide a microscopically rough surface on the titanium substrate. A conformal coating of a hydrophobic compound is then desposited on the microscopically rough surface to create a superhydrophobic surface. Thus, a substantially self-cleaning superhydrophobic surface is created on the titanium substrate, whereby, when exposed to ultraviolet light, the titanium oxide layer has a photocatalytic reaction with oxygen to oxidize any organic contaminants that may gather on the superhydrophobic surface.
Owner:THE BOEING CO
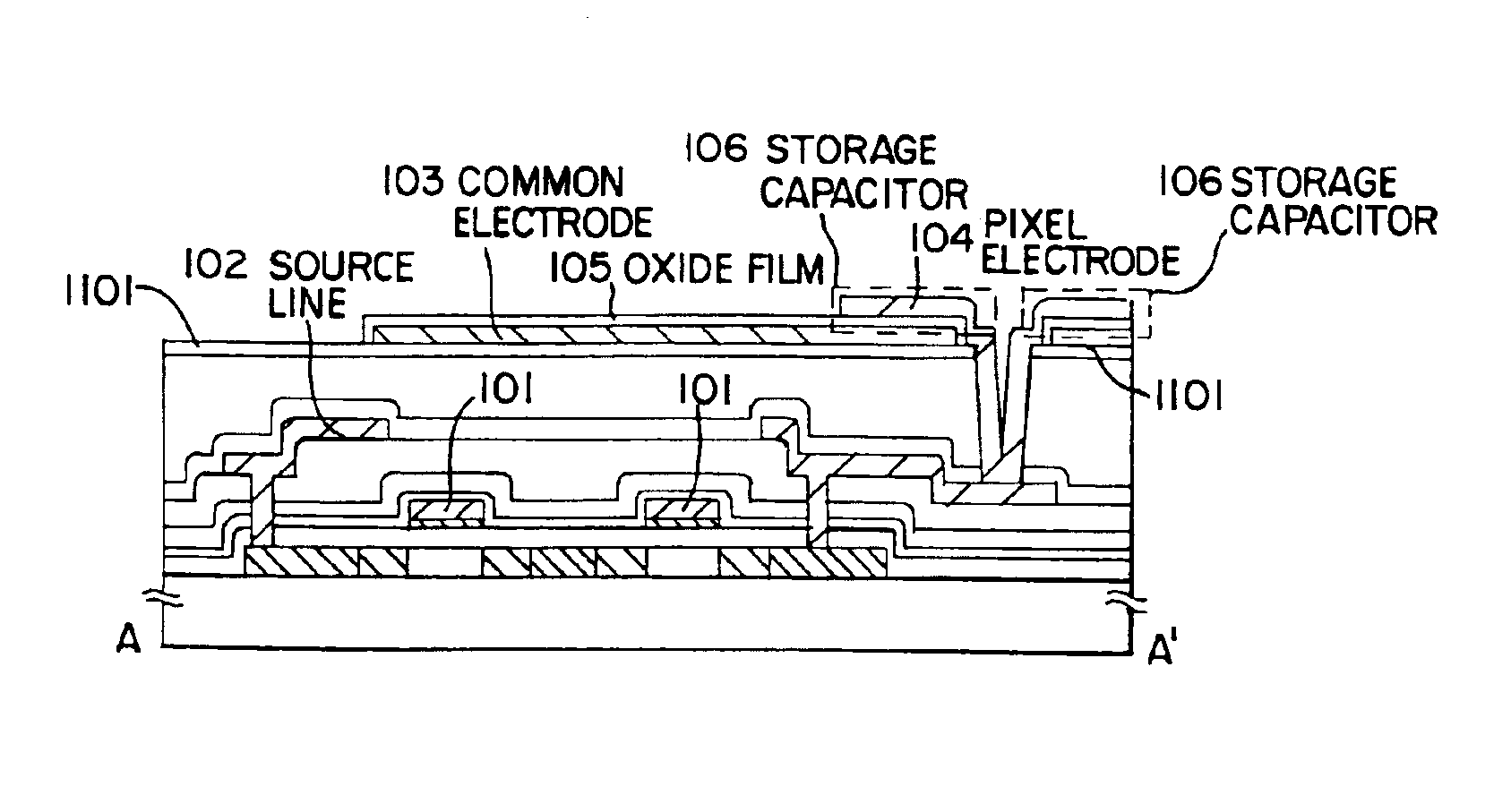
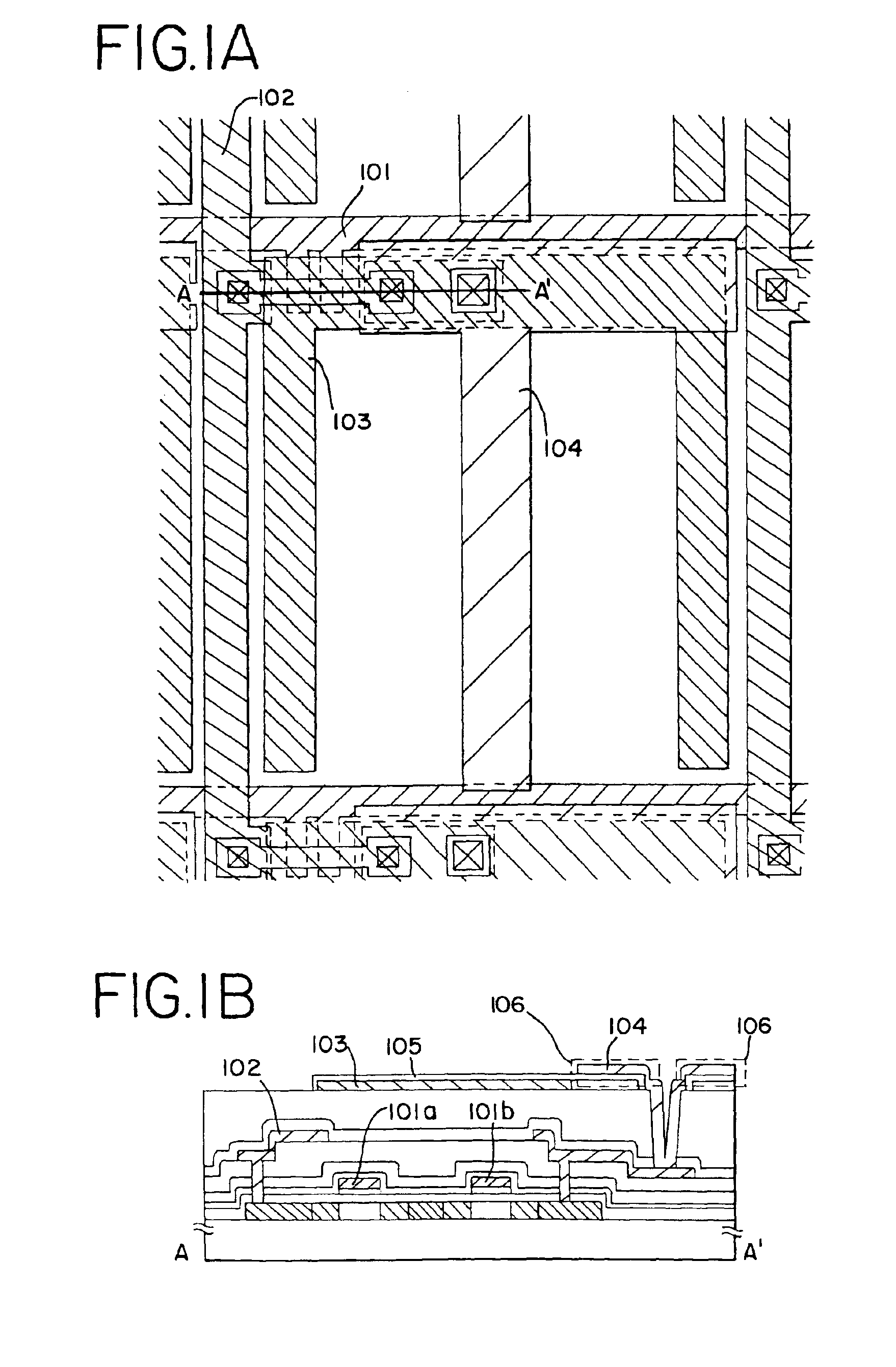
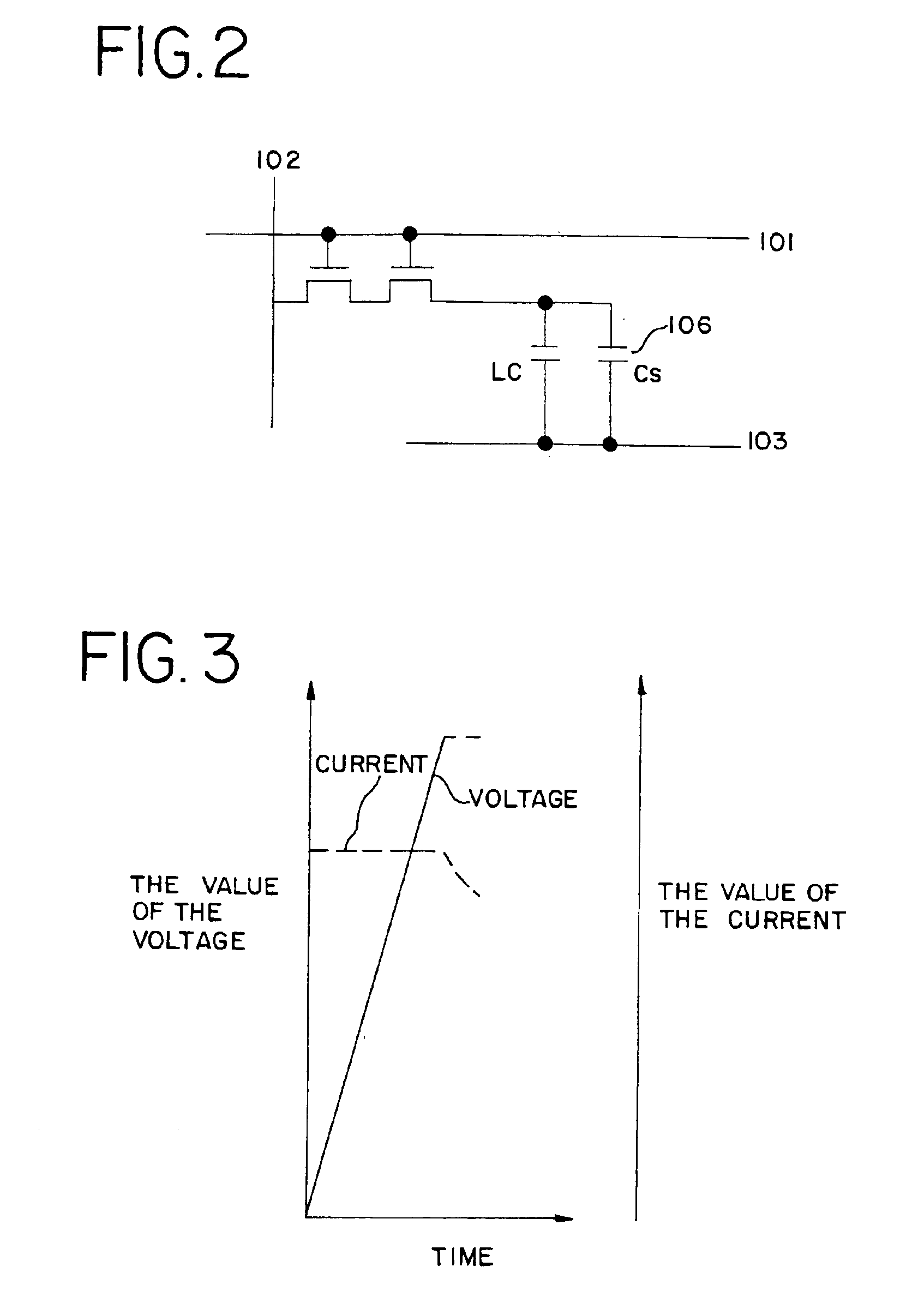
Semiconductor device with capacitor formed around contact hole
A method of forming a storage capacitor in an IPS liquid crystal display device is proposed, and a technique of forming a pixel region having a high aperture ratio is provided. An anodic oxidation process at an applied voltage / voltage supply time ratio of 11 V / min is performed for insulating films used in each circuit of an electro-optical device, typically an IPS method LCD, in particular for the surface of a common electrode formed on a resin film. The amount of formation of the extra anodic oxide film can be reduced by covering with an anodic oxide film, and a liquid crystal display device with high reliability and having an electrode with superior adhesion can be manufactured.
Owner:SEMICON ENERGY LAB CO LTD
Aluminum strip continuous anodic oxidation automatic production line
InactiveCN101676439AContinuous production saves time and laborAnodisationWinding machineProduction line
Owner:浙江聚力文化发展股份有限公司
Mediated electrochemical oxidation process used as a hydrogen fuel generator
InactiveUS20050161342A1Improve efficiencyLiquid separation by electricityWater/sewage treatment using germicide/oligodynamic-processFuranUltraviolet
A mediated electrochemical oxidation process and apparatus are used to process biological and organic materials to provide hydrogen and oxygen for use as fuel in numerous types of equipment. Waste materials are introduced into an apparatus for contacting the waste with an electrolyte containing the oxidized form of one or more reversible redox couples, at least one of which is produced electrochemically by anodic oxidation at the anode of an electrochemical cell. The oxidized species of the redox couples oxidize the organic waste molecules and are themselves converted to their reduced form, whereupon they are reoxidized and the redox cycle continues until all oxidizable waste species have undergone the desired degree of oxidation. The entire process takes place at temperatures to avoid any possible formation of either dioxins or furans. The oxidation process may be enhanced by the addition of ultrasonic energy and / or ultraviolet radiation.
Owner:SCIMIST LNC
SOI by oxidation of porous silicon
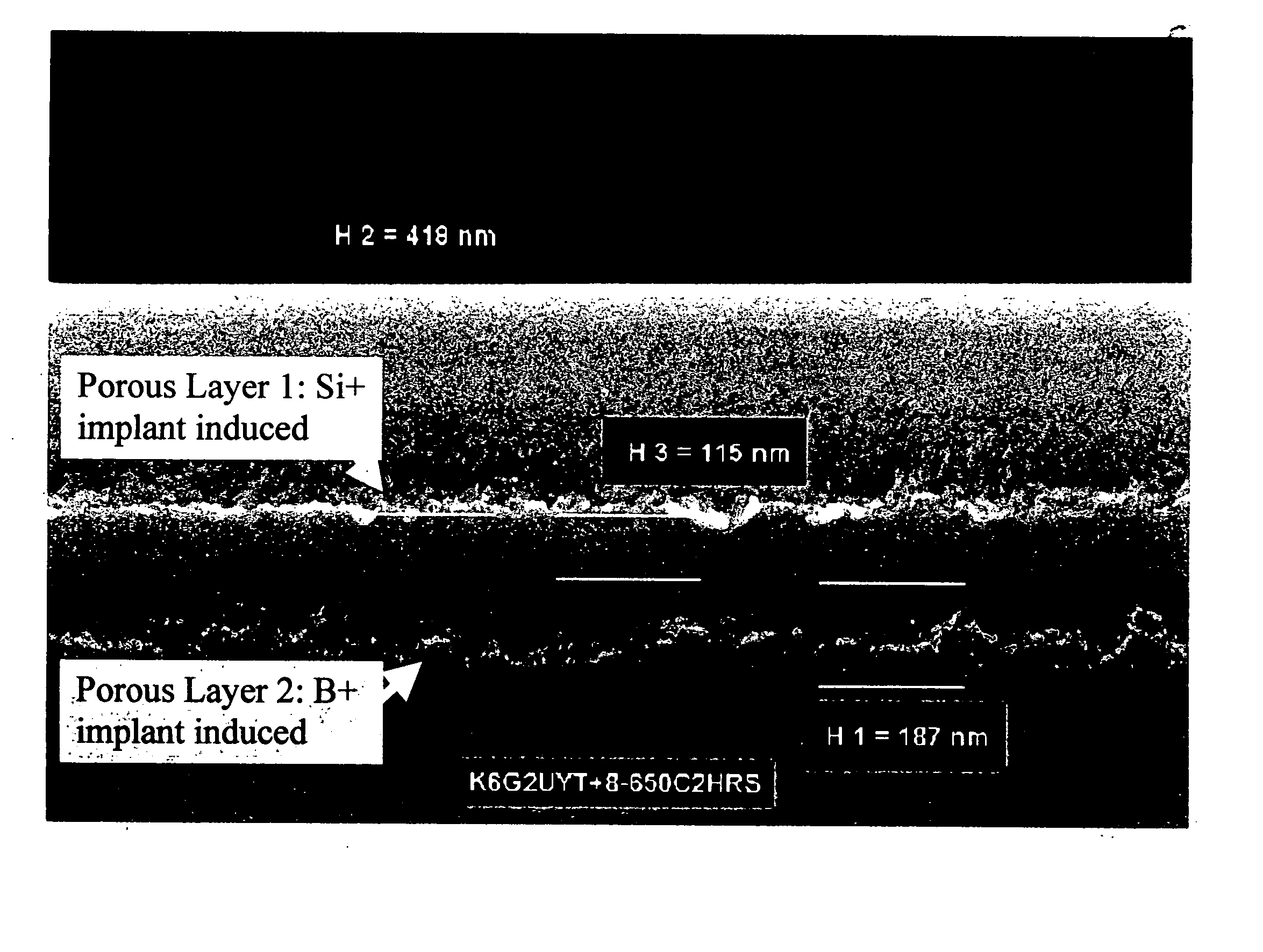
InactiveUS20050067294A1Simple and cost-effectiveReduce defect levelAnodisationVacuum evaporation coatingDopantPorous layer
A method in which a SOI substrate structure is fabricated by oxidation of graded porous Si is provided. The graded porous Si is formed by first implanting a dopant (p- or n-type) into a Si-containing substrate, activating the dopant using an activation anneal step and then anodizing the implanted and activated dopant region in a HF-containing solution. The graded porous Si has a relatively coarse top layer and a fine porous layer that is buried beneath the top layer. Upon a subsequent oxidation step, the fine buried porous layer is converted into a buried oxide, while the coarse top layer coalesces into a solid Si-containing over-layer by surface migration of Si atoms.
Owner:GLOBALFOUNDRIES INC
Method for preparing TiO2 nanotube array film
InactiveCN101514471ARegular microscopic morphologyNo reunionPolycrystalline material growthSurface reaction electrolytic coatingTio2 nanotubeTitanium
The invention provides a method for a preparing TiO2 nanotube array film, relating to a method for preparing a nanotube film. The method solves the problems of the prior art that the prepared nanotube array film features thin film, irregular micro appearance, heterogeneous length of nanotubes, reunion of tube orifice and cracking of the film. The preparation method comprises: 1. a titanium plate is cut into two titanium sheets of the same size, and polishing, ultrasound processing and washing are carried out on the two titanium sheets; 2. electrolyte is prepared; 3. primary anodic oxidation is carried out on the titanium sheets; 4. demoulding is carried out; 5. secondary anodic oxidation is carried out on the titanium sheets; 6. the titanium sheets are dried after ultrasonic processing; and 7. calcination treatment is carried out, thus obtaining the TiO2 nanotube array film. In the invention, twice anodic oxidations are carried out on the titanium sheets, thus producing the TiO2 nanotube array film featuring regular micro appearance, homogeneous, smoothness, thick film layer, reunion-free tube orifice and no cracking. The preparation method of the invention features simple technique and equipment and controllable thickness of the nanotube film.
Owner:HARBIN INST OF TECH