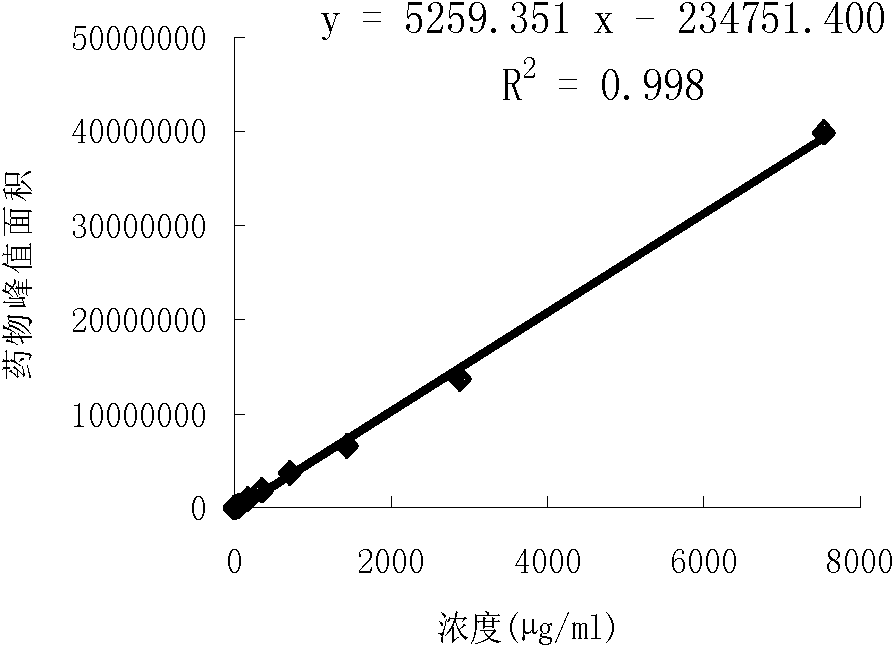Patents
Literature
33 results about "Ampelopsin" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
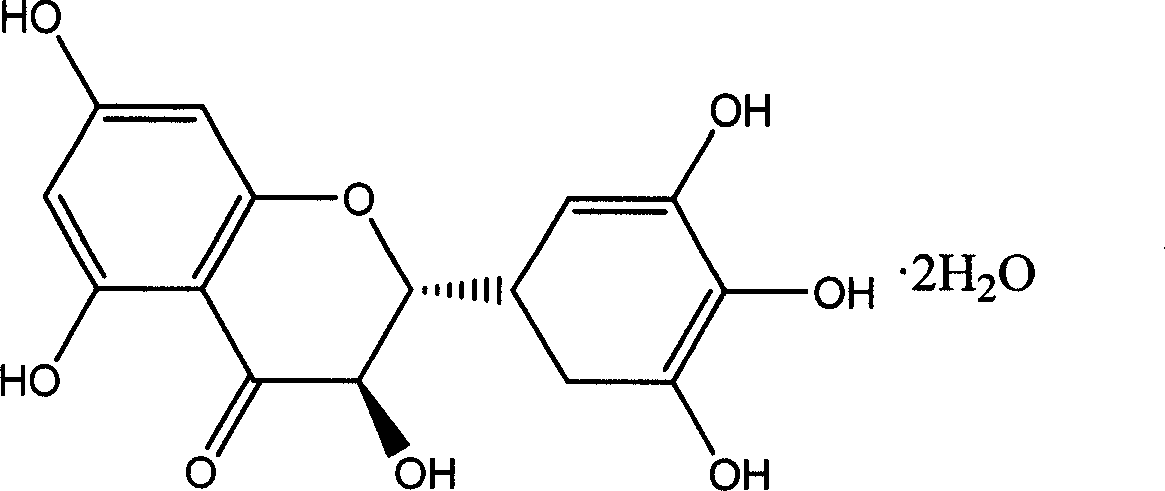
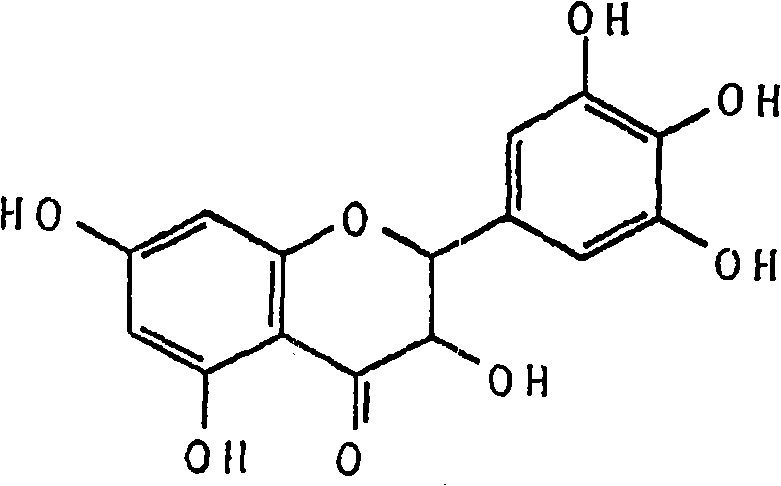
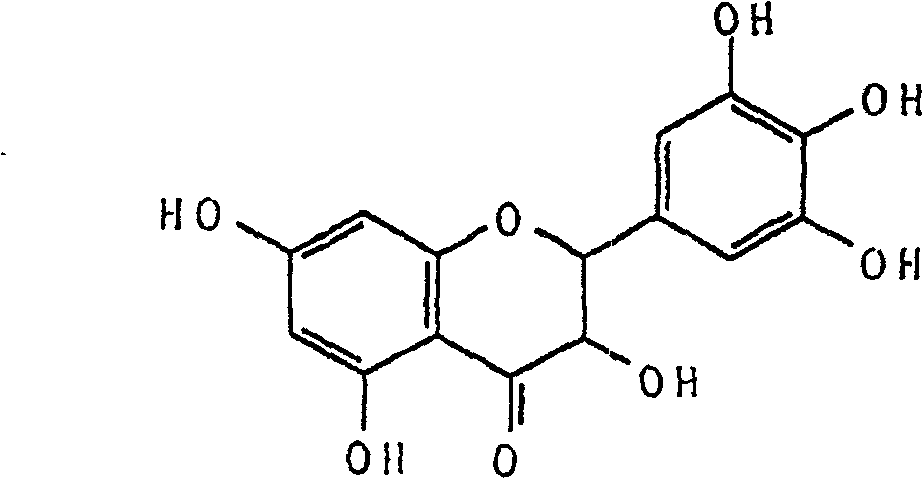
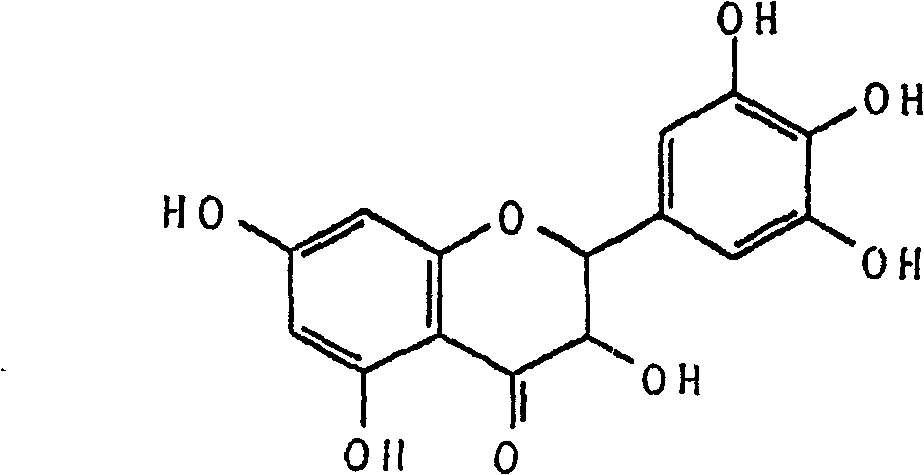
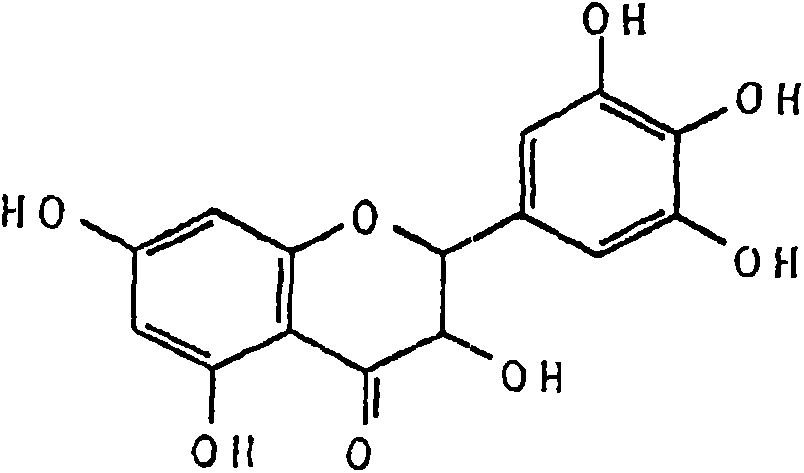
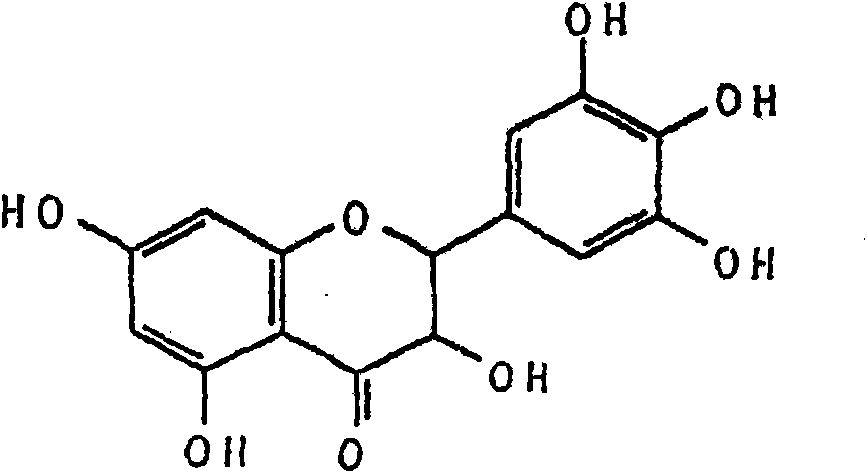
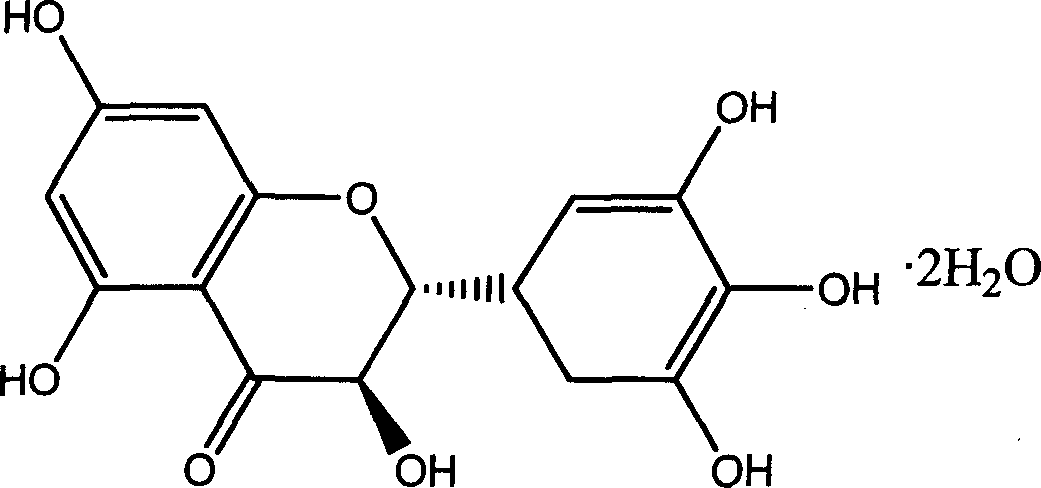
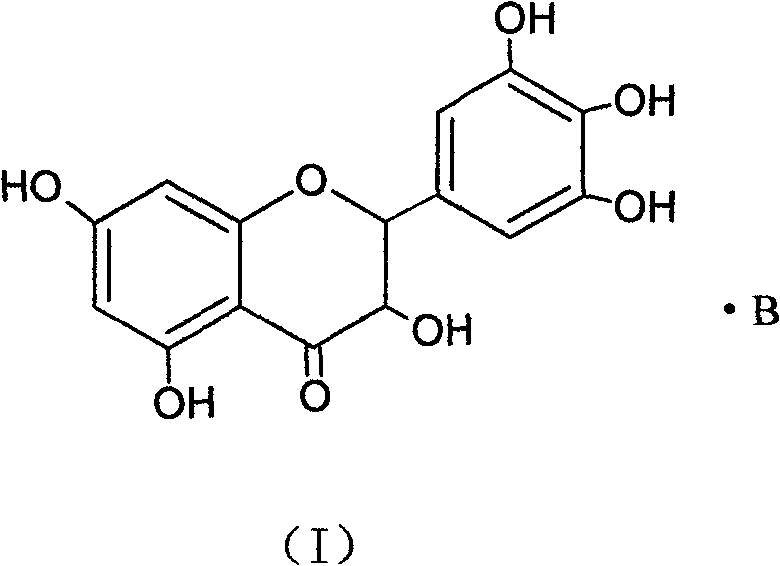
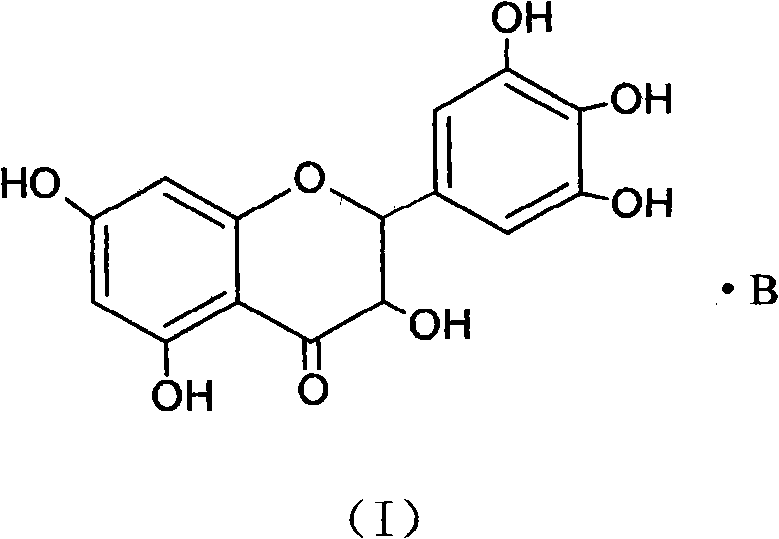
Ampelopsin, also known as dihydromyricetin, is a flavanonol, a type of flavonoid. It is found in the Ampelopsis species japonica, megalophylla, and grossedentata; Cercidiphyllum japonicum; Hovenia dulcis; Rhododendron cinnabarinum; some Pinus species; and some Cedrus species, as well as in Salix sachalinensis.
Mixed natural antibacterial agent, preparation method thereof and application of mixed natural antibacterial agent in cosmetics
InactiveCN105213250AStable in natureSimple processCosmetic preparationsToilet preparationsBiotechnologyAmpelopsin
The invention discloses a mixed natural antibacterial agent. The mixed natural antibacterial agent comprises, by weight percentage, 5-50% of ampelopsin grossedentata extract, 5-70% of one or more natural fragrant materials, and 1-80% of mixed solution of propylene glycol and water. The invention further provides a preparation method of the mixed natural antibacterial agent. The preparation method includes: weighing the ampelopsin grossedentata extract, the natural fragrant materials and the mixed solution of propylene glycol and water according to the weight percentage, and heating to dissolve the ampelopsin grossedentata extract in propylene glycol aqueous solution; adding the natural fragrant materials into the solution, and well mixing to obtain the mixed natural antibacterial agent. The invention further provides cosmetics containing the mixed natural antibacterial agent. The mixed natural antibacterial agent can effectively inhibit the microorganism reproduction in the cosmetics and can replace chemically-synthesized preservatives, and accordingly product irritation is lowered and product safety is increased.
Owner:SHANGHAI INST OF TECH +1
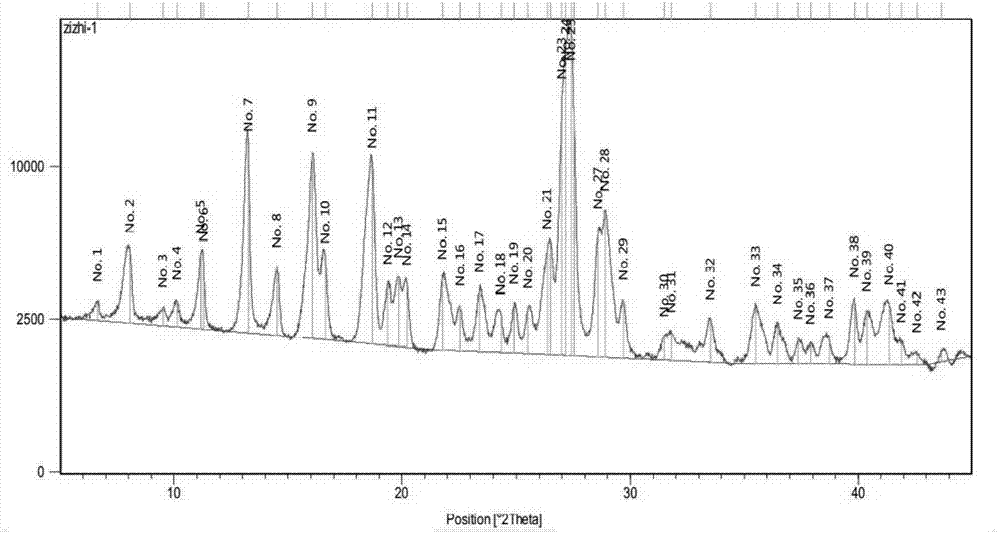
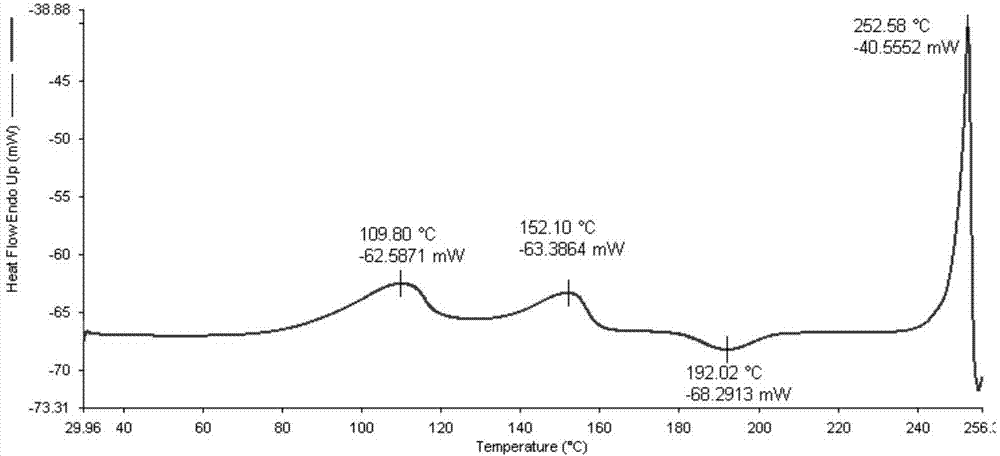
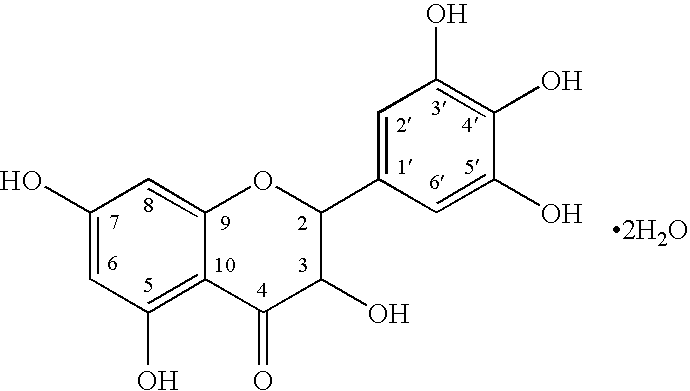
Five different crystalline form substances of dihydromyricelin
The invention discloses five crystalline form substances of dihydromyricelin (of which the chemical name is 3,5,7,3',4',5'-hexahydroxy-2,3-dihydroflavonol and the English name is dihydromyricelin or ampelopsin). The spatial configuration of dihydromyricelin is characterized by virtue of colorimetry-polarimetry and high-performance liquid-phase chiral separation methods. By virtue of solid-state analytical technologies including a powder X-ray diffraction (XRD), differential scanning calorimetry (DSC), thermal gravimetric analysis (TGA), a Karl Fischer titration and Fourier transform-attenuated total reflectance (FT-ATR) infrared spectroscopy, the crystalline forms of dihydromyricelin are characterized. The polymorph screening of dihydromyricelin is of important significance for the development of processes of active pharmaceutical ingredients, the development of dosage forms such as solid preparation, semi-solid preparation and suspension and quality control.
Owner:TONGJI HOSPITAL ATTACHED TO TONGJI MEDICAL COLLEGE HUAZHONG SCI TECH
Use of ampelopsin in preparation of angiogenesis inhibitor drug
The invention relates to the use of ampelopsin in preparation of angiogenesis inhibitor drug, wherein the suppression function of ampelopsin to propagation of blood vessel endothelial cells is reviewed, the suppression function of ampelopsin to synthesis and secretion of VEGF, bFGF by human hepatoma cells is also examined, test results show that ampelopsin has suppression function to propagation of blood vessel endothelial cells, and also suppression action to the synthesis and secretion of VEGF and bFGF.
Owner:SUN YAT SEN UNIV
Ampelopsin and basic amino acid solubilizing system
InactiveCN101513400AImprove solubilityImprove stabilityPowder deliveryOrganic active ingredientsCarboplatinDiluent
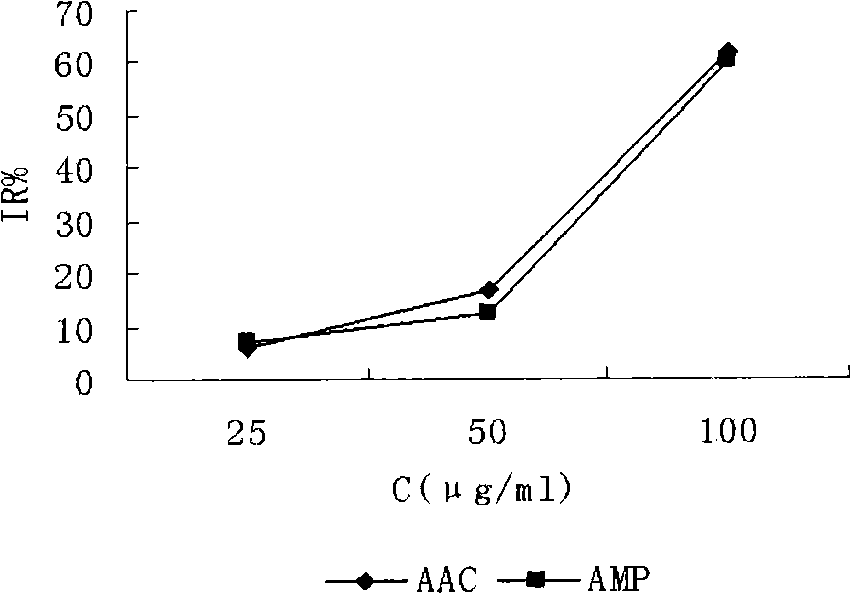
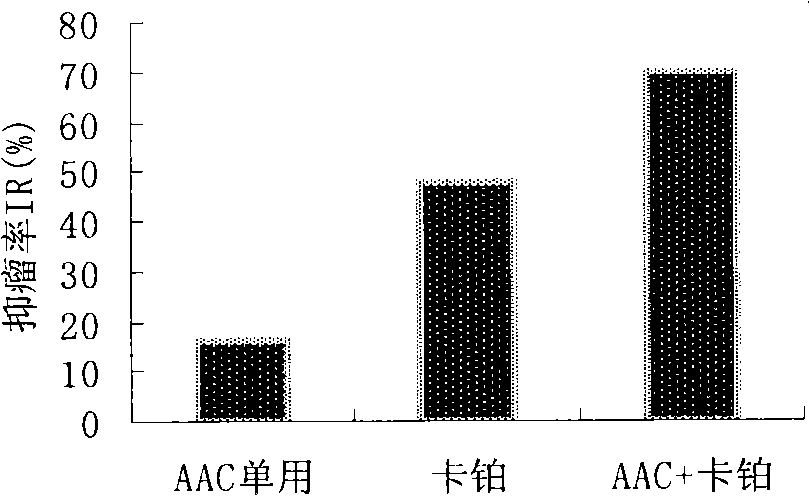
The invention provides an ampelopsin composition and a method for preparing the same. The composition comprises: a) ampelopsin and b) a basic amino acid, wherein the weight ratio the component a) to the component b) is between 1:1 and 1:10. The composition can also comprise: c) a pH regulator and or d) a pharmaceutically acceptable carrier and / or a diluting agent. The invention also relates to a medicamnet box containing the ampelopsin composition and carboplatin. The ampelopsin composition can provide improved solubility, stability and a proper pH range and is suitable for clinic use.
Owner:LANZHOU UNIVERSITY +1
Use of ampelopsin in preparation of healthcare food and medicaments for protecting vascular endothelial cells
The invention discloses the use of ampelopsin in the preparation of healthcare food and medicaments for protecting vascular endothelial cells. In the invention, purified ampelopsin or unpurified ampelopsin-containing extract is prepared into pharmaceutically allowable preparations for oral taking, injection or external application. The ampelopsin protects the vascular endothelial cells by reducing peroxidation of lipid, resisting oxidative damage and other actions, so as to prevent and treat cardiovascular and cerebrovascular diseases caused by endothelial cell injury, such as atherosclerosis, hypertension and cardiac-cerebral thrombosis.
Owner:任启生 +1
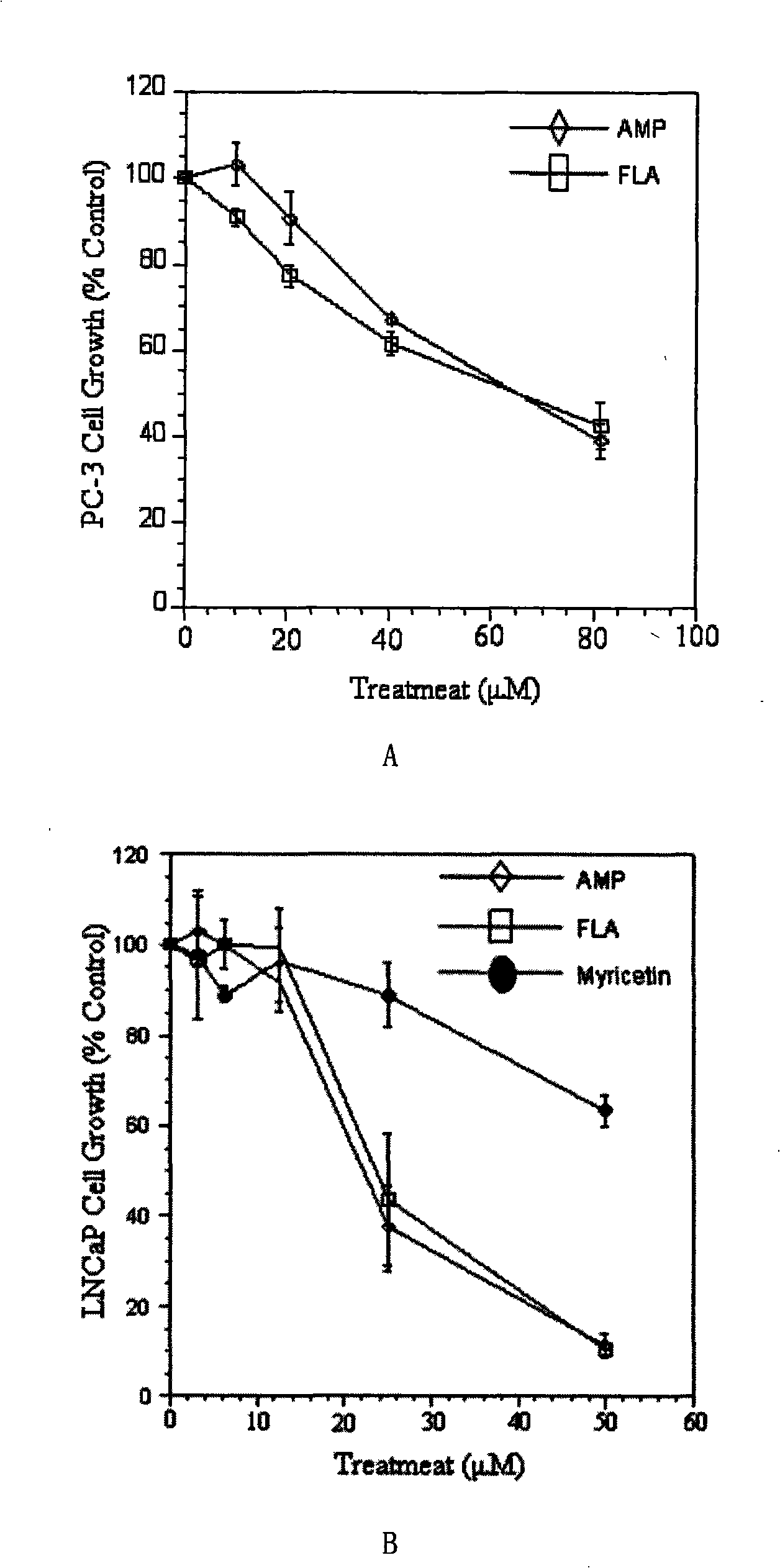
Uses of ampelopsin and total flavone valid target rich in ampelopsin in preparing medicament for preventing and treating prostate gland cancer
InactiveCN101347425ALittle side effectsPC3 tumor weight in mice significantly reduced side effectsOrganic active ingredientsAntineoplastic agentsTreatment effectSide effect
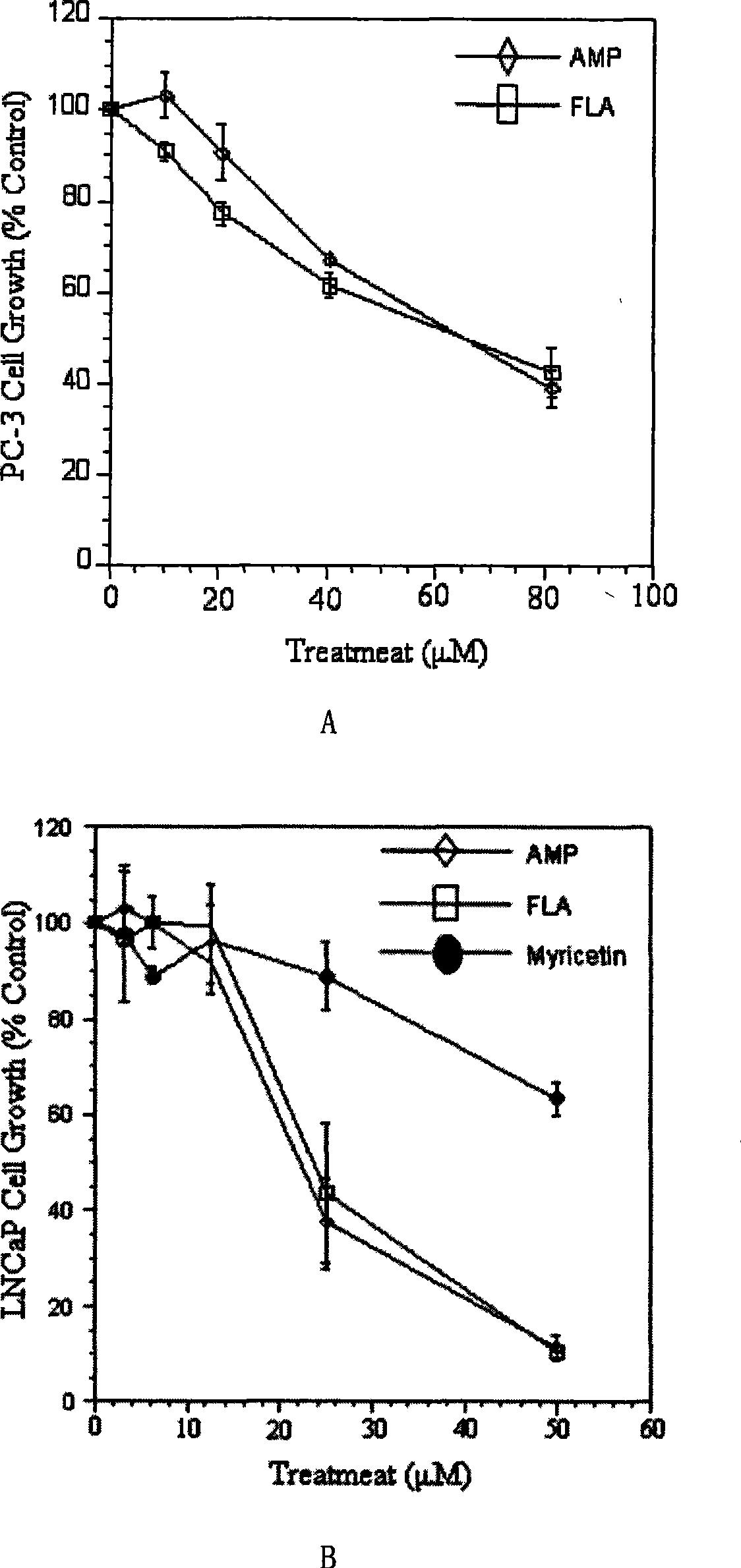
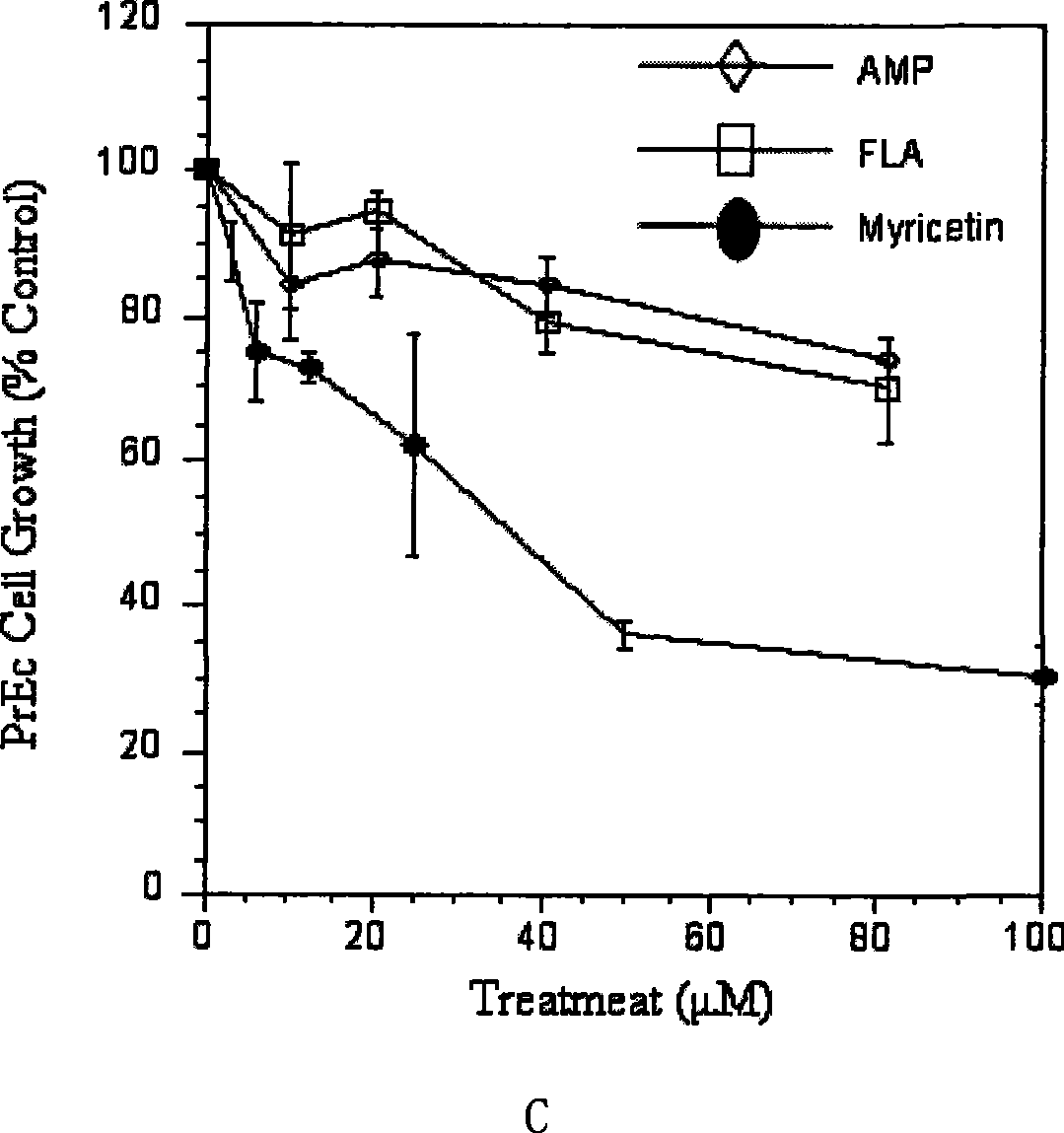
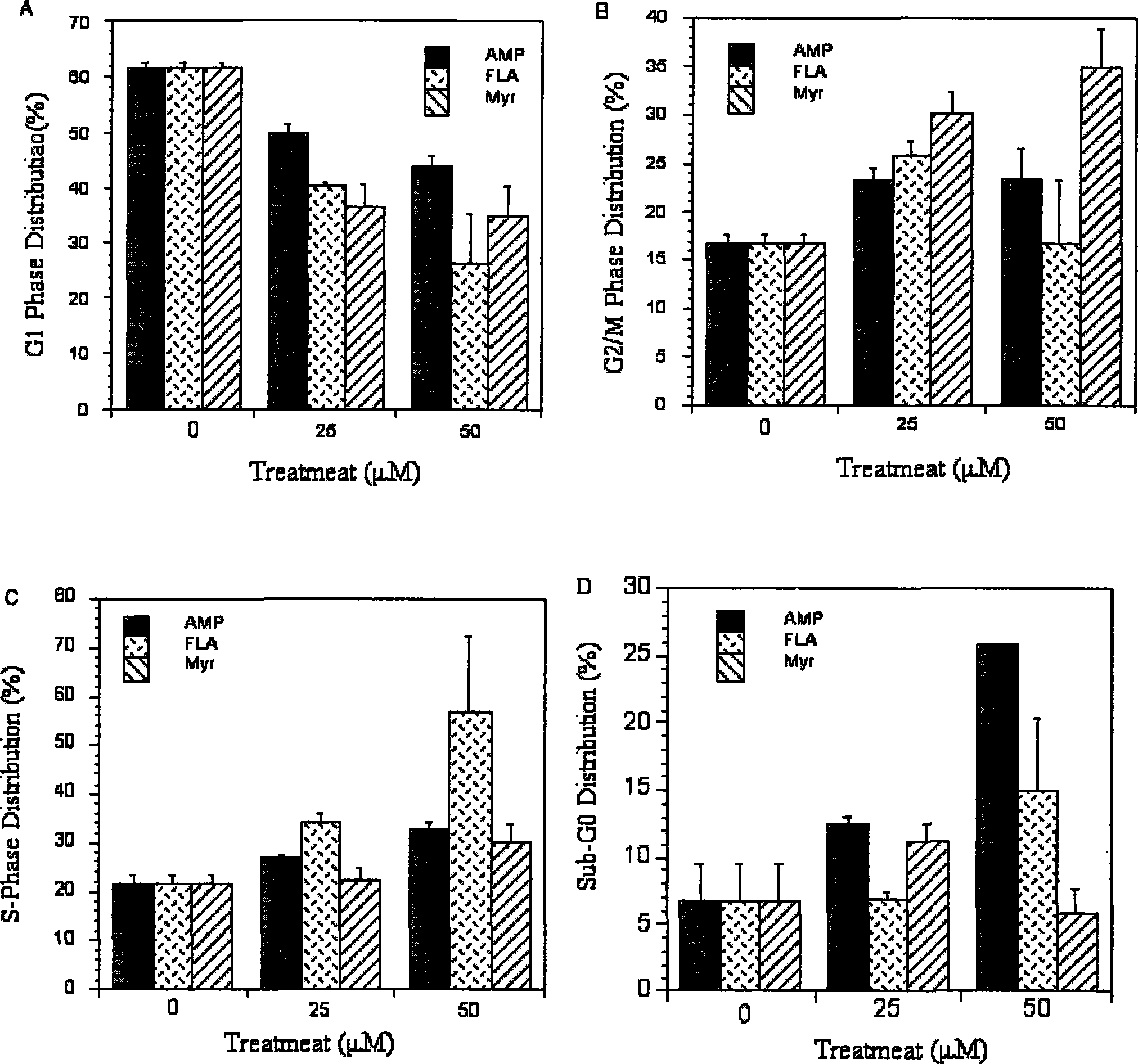
The invention provides an ampelopsin and an application of a total flavonoid effective part with rich ampelopsin in preparing a medicine for preventing and treating prostatic cancer. A new, high-efficiency, and low-toxic natural medicine resource is found out by aiming at the difficulty in treating the prostatic cancer, which has obvious curative effect and little toxic or side effect.
Owner:福建省中医药科学院 +1
Ampelopsin derivative, synthesizing method thereof and application of the same in preparing antineoplastic medicine
InactiveCN101186606AEliminate side effectsOrganic active ingredientsOrganic chemistrySynthesis methodsAmpelopsin
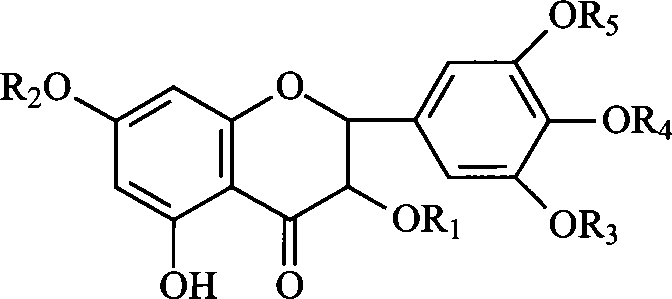
The invention relates to a ampelopsin derivative, wherein R1-R5 are represented as formula II or H, and R1-R5 are not all as H. The invention further relates to the synthesis method of the derivative and relative application for preparing anti-cancer drug.
Owner:SUN YAT SEN UNIV
Ampelopsin grossedentata light matrix seedling tray cuttage seedling method
InactiveCN105766458AImprove survival rateSaving cuttingsGrowth substratesCulture mediaAmpelopsinCladodes
The invention relates to the technical field of agricultural cultivation, in particular to an ampelopsin grossedentata light matrix seedling tray cuttage seedling method.The method includes the steps that grass carbon, perlite and vermiculite are evenly mixed and placed into a seedling tray; one-year strong fresh tender branches which are free of dehydration, diseases and pests and have leaves are selected, and stems are pruned; the pruned ampelopsin grossedentata stems are placed into a rooting powder aqueous solution to be soaked; the processed ampelopsin grossedentata branches are inserted into the light matrix seedling tray, kept vertically upwards and placed in a greenhouse after cuttage to be maintained; after the ampelopsin grossedentata branches sprout and 90% of the stems root, water-soluble fertilizer is applied; after roots of ampelopsin grossedentata plants overgrow and the length of veins is larger than or equal to 20 cm, ampelopsin grossedentata is harvested; ampelopsin grossedentata light matrix seedling tray cuttage seedling is finished.The seedling tray cuttage survival rate can reach 90% or above, scions are saved compared with land seedling cuttage, ampelopsin grossedentata roots in one month, seedlings are obtained and harvested in three months, the survival rate of transplanting in fields is up to 95%, and the advantages of land cuttage are incomparable to those of the method.
Owner:HUBEI HUAYI BIOTECH CO LTD
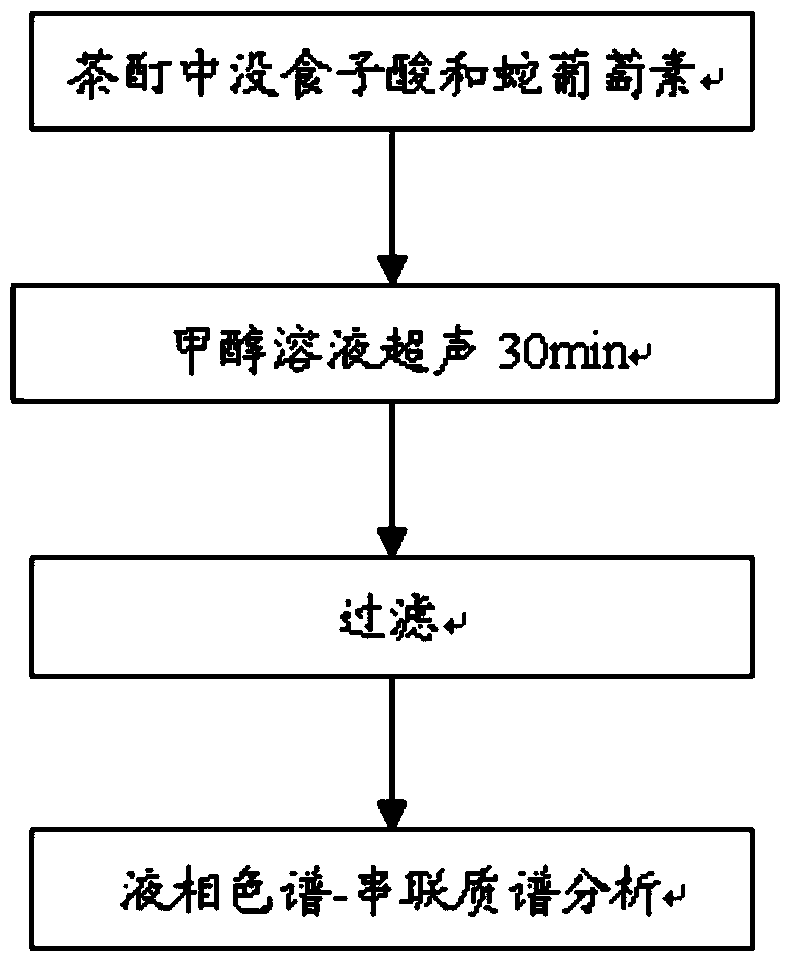
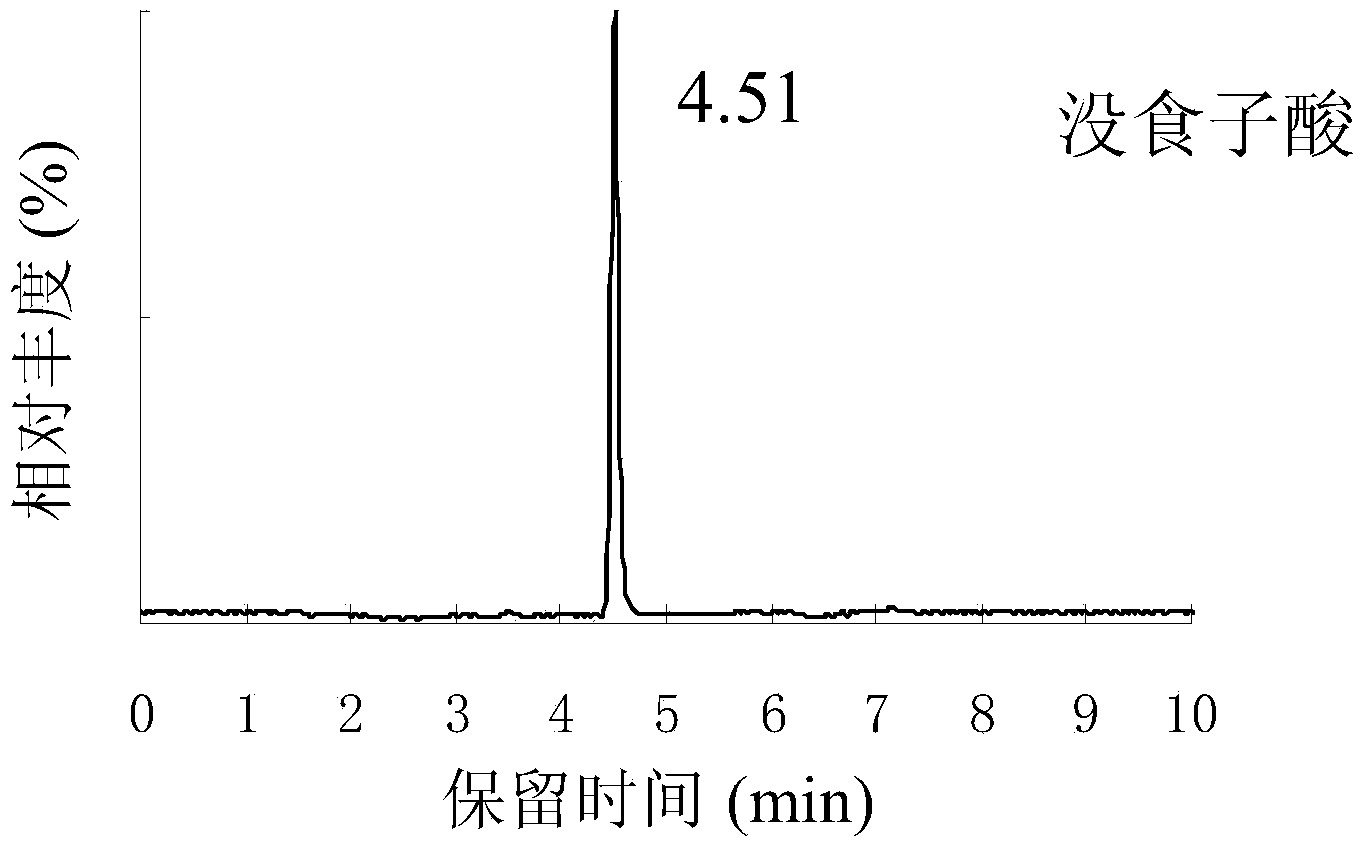
Method for detecting gallic acid and ampelopsin in tea tincture
ActiveCN104101666AEasy to handleEasy processing conditionsComponent separationGallic acid esterRelative standard deviation
The invention relates to a method for detecting gallic acid and ampelopsin in tea tincture. The method comprises performing determination through a liquid chromatography tandem mass spectrometry and performing calibration through an external standard method. The method for detecting the gallic acid and the ampelopsin in the tea tincture has the advantage s of being short in detecting time, simple in operation, high in sensitivity and recovery rate, good in repeatability and the like. Chromatographic conditions in the method enable the chromatographic peaks of the gallic acid and the ampelopsin to be well separated from those of impurities and to obtain good linearity ranges with limit of detection of 0.002 mu g / mL, the recovery rate of the gallic acid is between 91.3% and 109.2%, the recovery rate of the ampelopsin is between 84.1% and 97.8%, and the relative standard deviations of the detecting results of samples are all smaller than 5%.
Owner:CHINA TOBACCO FUJIAN IND
Application of ampelopsin for preparing medicaments for treating acne abscess, ulcer fester and piles
The invention discloses application of ampelopsin for preparing a medicament for treating acne abscess, ulcer fester and piles, and the medicament can be prepared into pharmaceutically accepted dosage forms for external use, oral administration and injection.
Owner:任启生 +1
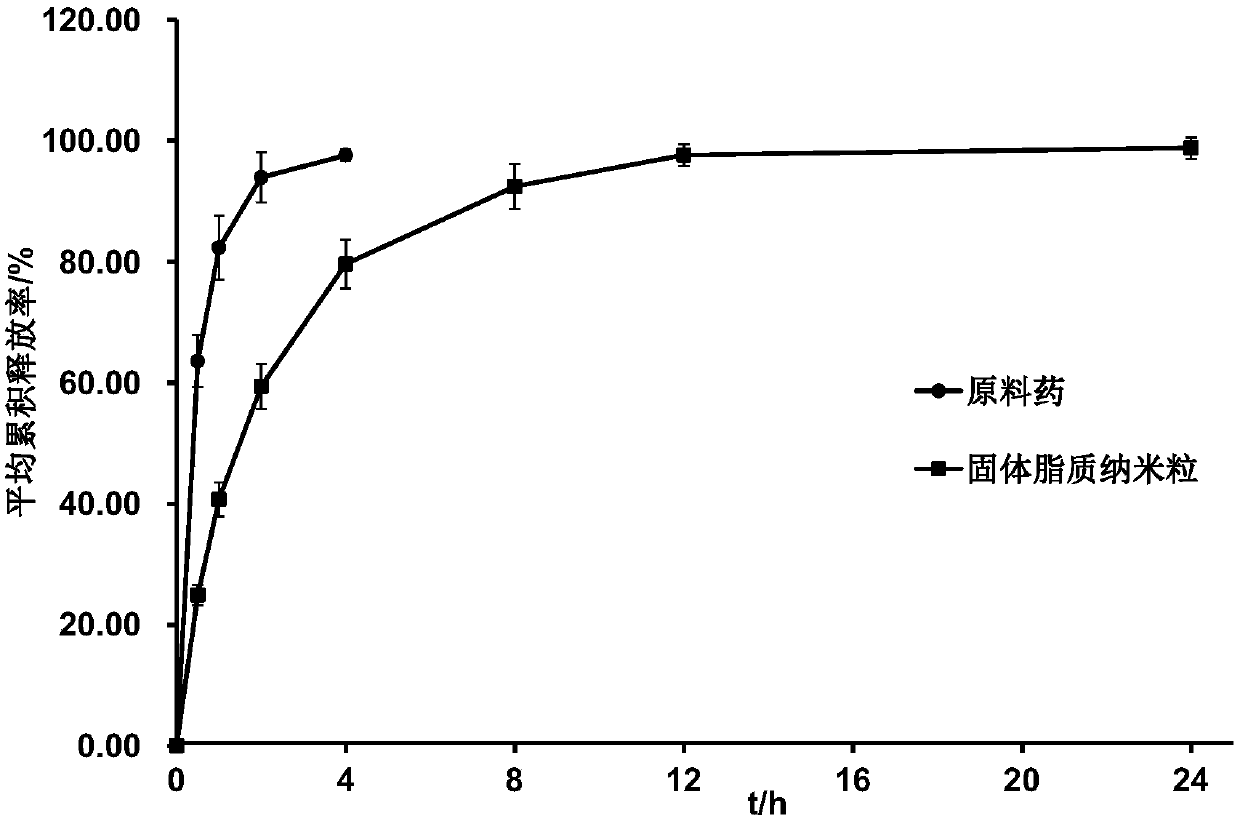
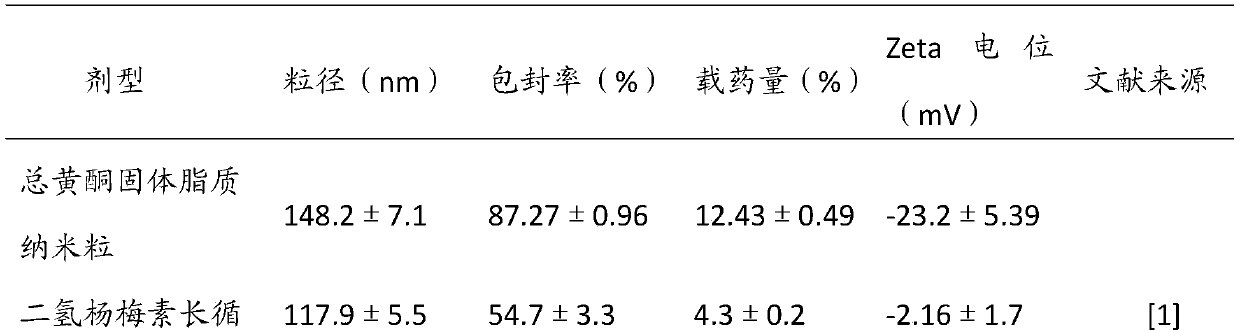
Ampelopsin grossedentata total flavonoid solid lipid nanoparticle and preparation method
InactiveCN109589367ABiodegradableGood physiological compatibilityAntibacterial agentsPowder deliveryAmpelopsinHigh pressure
The invention belongs to the field of pharmaceutical preparations and relates to an ampelopsin grossedentata total flavonoid solid lipid nanoparticle and a preparation method. The solid lipid nanoparticle takes solid natural or synthetical lipoid as a carrier and comprises the following raw materials in percentage by weight: 0.3-0.7% of ampelopsin grossedentata total flavonoids, 4-12% of a surfactant and the balance of water, wherein a drug-lipid ratio is 1:3-1:12. The preparation method employs a fusion-ultrasound method or a high-pressure homogenization method. Encapsulation efficiency and drug loading capacity of the ampelopsin grossedentata total flavonoids can be improved by selecting the appropriate solid lipid and an emolsifier and optimizing a raw material proportion; a particle size is reduced; and stability, bioavailability and targeting ability of the nanoparticle are improved. The ampelopsin grossedentata total flavonoid solid lipid nanoparticle with the small particle sizeand the higher encapsulation efficiency is prepared by the simple, convenient and operation controllable fusion-ultrasound method and shows a slow release characteristic; and a novel dosage form is provided for development and clinical application of the ampelopsin grossedentata total flavonoids.
Owner:FUJIAN UNIV OF TRADITIONAL CHINESE MEDICINE
Application of ampelopsin to preparation of anti-mutagenic health-care foods and medicaments
InactiveCN102038190AGood treatment effectOrganic active ingredientsDigestive systemMedicineAmpelopsin
The invention discloses application of ampelopsin to preparation of anti-mutagenic health-care foods and medicaments. Pharmaceutically allowable oral formulations, injection formulations or externally applied formulations are prepared from purified ampelopsin or unpurified ampelopsin-containing extracts.
Owner:任启生 +1
Application of ampelopsin in preparation of vascular endothelial cell protection health food or drugs
The present invention discloses an application of ampelopsin in preparation of vascular endothelial cell protection health food or drugs, wherein purified ampelopsin or an unpurified ampelopsin-containing extract can be adopted to prepare a pharmaceutically acceptable oral formulation, a pharmaceutically acceptable injection formulation or a pharmaceutically acceptable external application formulation, and the ampelopsin provides a vascular endothelial cell protection function through lipid peroxidation reduction, oxidative damage resistance and other effects so as to prevent and treat cardiovascular and cerebrovascular diseases caused by vascular endothelial cell damage, wherein the diseases comprise atherosclerosis, high blood pressure, heart and cerebral thrombosis, and the like.
Owner:任启生 +1
Method for extracting dihydromyricetin in ampelopsin grossedentata by means of fermentation
A method for fermenting and extracting dihydromyricetin in Maoyanberry tea relates to a method for extracting active ingredients in Maoyanberry tea, comprising the following steps: pulverizing Maoyanberry tea leaves and extracting them under reflux, concentrating the filtrate and then statically tea juice; add sucrose to the tea juice and sterilize; mix active dry yeast and sucrose solution in a constant temperature water bath to activate the active dry yeast and insert it into the sterilized tea juice; inoculate the tea juice Putting it into a shaker for fermentation; concentrating to obtain extract, drying to obtain dihydromyricetin coarse powder and recrystallizing to obtain pure dihydromyricetin. The present invention utilizes the liquid fermentation method to extract dihydromyricetin in Maoyanberry tea, which can consume nutrients such as sugar and protein in the Maoyanberry water extract, thereby reducing the viscosity of the concentrated hot water extract and making the dihydromyricetin Myricetin can settle down quickly, effectively utilizes Maoyanberry tea resources, and improves the yield of dihydromyricetin.
Owner:四川外交家酒业有限公司
Traditional Chinese medicine pill for auxiliary treatment on alcoholic fatty liver and preparation method thereof
InactiveCN104984255AHigh cure ratePromote absorptionDigestive systemPill deliveryAlcoholismsLiver and kidney
The invention discloses a traditional Chinese medicine pill for auxiliary treatment on alcoholic fatty liver and a preparation method thereof, and belongs to the field of traditional Chinese medicine. The effective component of the pill is composed of the following raw materials: radix fici simplicissimae, ampelopsin grossedentata, curcuma tuber, poncirus trifoliate, leaf and stem of ficus erecta, sweet potato vine, radix scutellariae, fingered citron, wolfberry, semen cuscutae, wrinkled giant hyssop, licorice, loofah sponge, brassica chinensis, safflower, white peony root, pine pollen, flatstem milkvetch seed, oriental waterplantain rhizome, dried orange peel, corydalis adunca, tabasheer, American ginseng, and plum blossom. The provided traditional Chinese medicine pill has a definite curative effect, has the functions of nourishing liver and kidney, invigorating the spleen, resolving dampness, clearing heat, reducing phlegm, activating blood circulation, freeing the collaterals, soothing the liver, regulating qi, promoting digestion, relieving alcoholism, harmonizing the stomach, and stopping vomiting, has the advantages of good absorption effect, prominent curative effect, no side or toxic effect, and no adverse reaction in clinic, and has a high cure rate on treating alcoholic fatty liver in clinic.
Owner:徐冬冬
Industrial extracting method for ampelopsin
The invention discloses an industrial extracting method for ampelopsin. The industrial extracting method comprises the steps of adding magnetized water into an extracting tank through a magnetizer until materials are immersed; circularly magnetizing the water, extracting under heating, and collecting an extracting solution after the extraction is ended; cooling the extracting solution, standing and extracting a clear solution to obtain a sediment; adding ethanol into the sediment, ultrasonically vibrating, extracting under heating, and extracting a supernatant liquid; carrying out vacuum concentration on the supernatant liquid to obtain a concentrated solution; adding hot water to dilute the concentrated solution, stirring, naturally cooling, separating the bottom sediment, further naturally cooling the residual clear solution to generate a yellowish white sediment, secondarily adding hot water into the yellowish white sediment, boiling, dissolving, repeating the steps many times, and drying the obtained high-purity ampelopsin. By using the industrial extracting method, the yield can be increased by more than 35%, and the extracting time can be shortened by 1 / 3; when ampelopsin is separated, fat-soluble impurities, part of water-soluble polyphenol and other impurities are removed; in addition, most of water-soluble polyphenols are removed through sectional sedimentation.
Owner:湖北金睿生物科技有限公司
Application of ampelopsin in mutation resistance composition preparation
InactiveCN103251581AGood treatment effectOrganic active ingredientsAntiviralsBiotechnologyPharmaceutical drug
The present invention discloses an application of ampelopsin in preparation of mutation resistance health food or drugs, wherein purified ampelopsin or an unpurified extract containing ampelopsin can be adopted to prepare into a pharmaceutically acceptable oral formulation, a pharmaceutically acceptable injection formulation or a pharmaceutically acceptable external application formulation.
Owner:任启生 +1
Antibacterial composition and preparation method thereof
InactiveCN109602763ANo toxicityDirect effectOrganic active ingredientsAntisepticsSilver iodideSodium iodide
The present invention relates to the technical field of medicines and particularly relates to an antibacterial composition and a preparation method thereof. The disclosed antibacterial composition isprepared from the following components: ampelopsin, an iodine-containing compound, an organic solvent, an emulsifier and water; and the iodine-containing compound is selected from one or more of potassium iodide, sodium iodide, silver iodide and calcium iodide. The ampelopsin is a natural plant extract ingredient, green and environmentally friendly, not toxic to human body skin and mucous membranes, can be directly applied to the human body epidermis and mucous membranes, directly kills bacteria at a first stage, and is more direct in effects. Experimental results show that the ampelopsin andthe iodine have a synergistic effect, a combined effect of the combination of the two materials is significant, and the antibacterial composition solves problems that existing antibacterial products are large in irritation to human body and poor in antibacterial effects on the human body epidermis and mucous membranes.
Owner:GUANGDONG UNIV OF TECH
Ampelopsin and basic amino acid solubilizing system and antitumor activity research
InactiveCN101513400BImprove solubilityImprove stabilityPowder deliveryOrganic active ingredientsCarboplatinAmpelopsin
The invention provides an ampelopsin composition and a method for preparing the same. The composition comprises: a) ampelopsin and b) a basic amino acid, wherein the weight ratio the component a) to the component b) is between 1:1 and 1:10. The composition can also comprise: c) a pH regulator and or d) a pharmaceutically acceptable carrier and / or a diluting agent. The invention also relates to a medicamnet box containing the ampelopsin composition and carboplatin. The ampelopsin composition can provide improved solubility, stability and a proper pH range and is suitable for clinic use.
Owner:LANZHOU UNIVERSITY +1
Method for improving biological activity of flavonoid compounds
InactiveCN114848631ANo chemical compatibility changesNo side effectsOrganic active ingredientsPharmaceutical delivery mechanismMetabolic enzymesAmpelopsin
The invention discloses a method for improving biological activity of flavonoid compounds, belongs to the technical field of medicines, particularly relates to the technical field of pharmaceutical compositions such as pharmaceutical dosage forms and preparations, and relates to a method for improving or improving oral absorption bioavailability of ampelopsin. The preparation is characterized in that the ampelopsin and an auxiliary material with a metabolic enzyme inhibition effect are jointly prepared into the preparation, and the concentration of the ampelopsin in plasma in a body can be effectively improved or increased, so that the oral curative effect of the ampelopsin is improved. The invention also relates to a method for improving the biological activity of the flavonoid compound. The invention has excellent technical effects as described in the specification.
Owner:FUJIAN HEALTH COLLEGE
Use of ampelopsin in preparation of angiogenesis inhibitor drug
The invention relates to the use of ampelopsin in preparation of angiogenesis inhibitor drug, wherein the suppression function of ampelopsin to propagation of blood vessel endothelial cells is reviewed, the suppression function of ampelopsin to synthesis and secretion of VEGF, bFGF by human hepatoma cells is also examined, test results show that ampelopsin has suppression function to propagation of blood vessel endothelial cells, and also suppression action to the synthesis and secretion of VEGF and bFGF.
Owner:SUN YAT SEN UNIV
Traditional Chinese medicine for treating un-ruptured tubal pregnancy and preparation method of traditional Chinese medicine
InactiveCN105687871ADefinite curative effectImprove efficiencyPowder deliveryAnthropod material medical ingredientsMedicinal herbsSide effect
The invention provides traditional Chinese medicine for treating un-ruptured tubal pregnancy and a preparation method of the traditional Chinese medicine. The traditional Chinese medicine is prepared from the following raw herbal medicine: ampelopsin grossedentata, glomeris insect, root of Japanese glorybower, all-grass of delavay pararuellia, whole plant of ficus formosana, divaricate serpentroot herb and root, Japanese raspberry herb, bamboo bee, henry sarmentose fig receptacle, four-leaf clover, root or whole herb of phlomis mongolica, rough knotweed herb, bambooleaf fig herb, whole herb of puccinellia tenuiflora and twig of Germany falsetamarisk. The traditional Chinese medicine provided by the invention has the beneficial effects that the traditional Chinese medicine and western medicine are combined to realize definite curative effect for un-ruptured tubal pregnancy; the traditional Chinese medicine is higher in effective rate, free of toxic or side effects and fewer in adverse reaction; a preparation process is simple.
Owner:石波
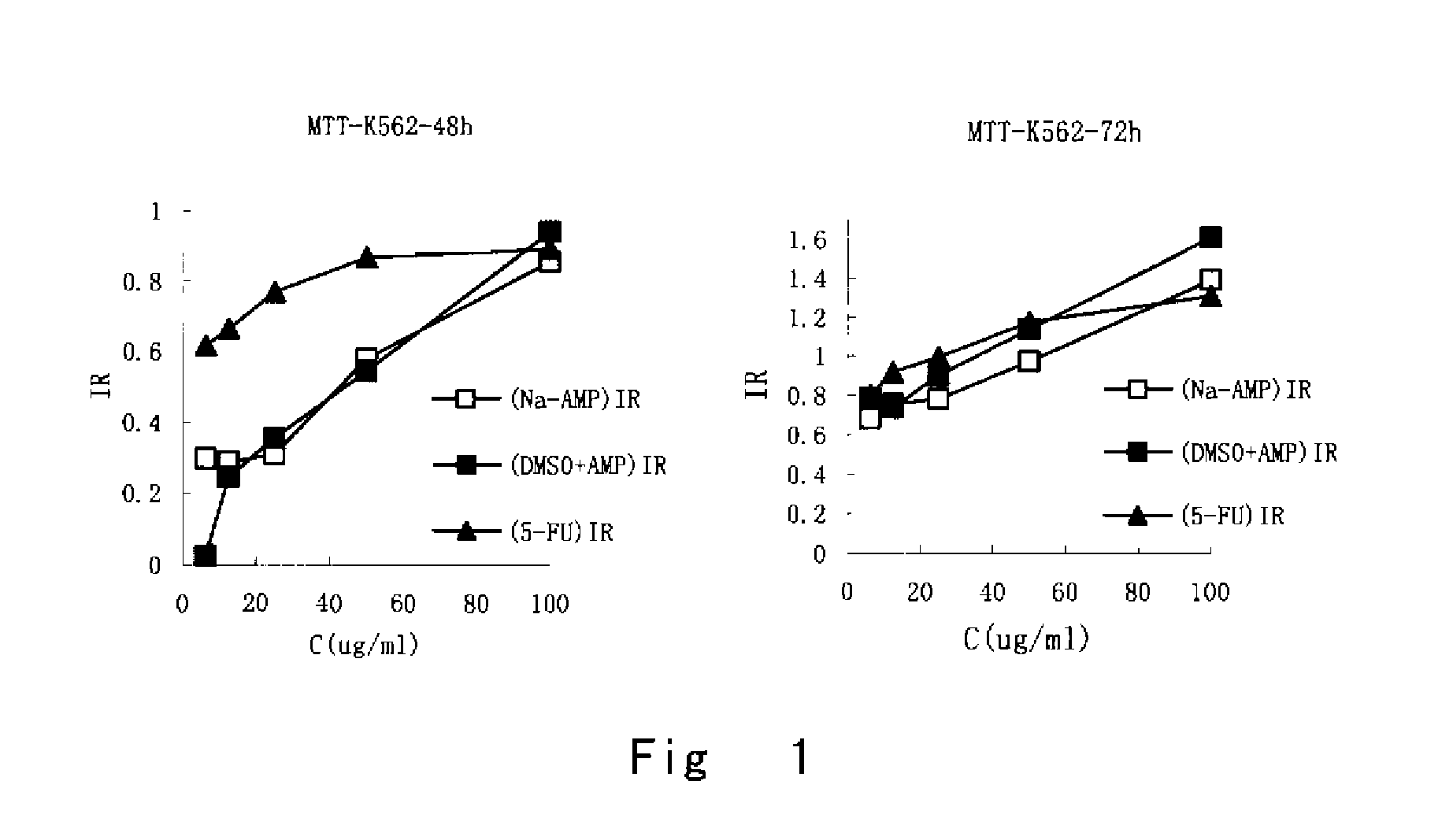
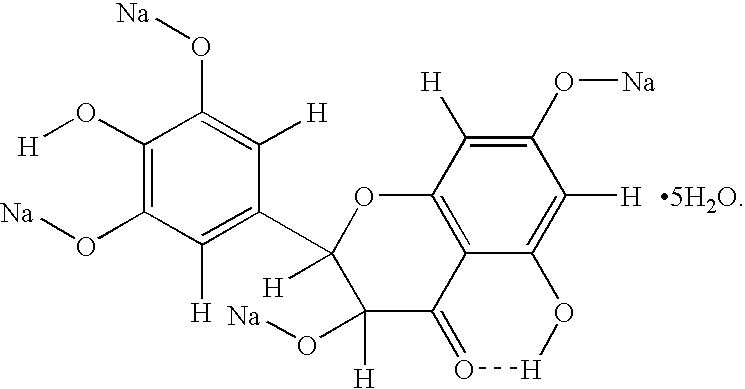
AMPelopsin unsaturated sodium salt preparation and applications thereof
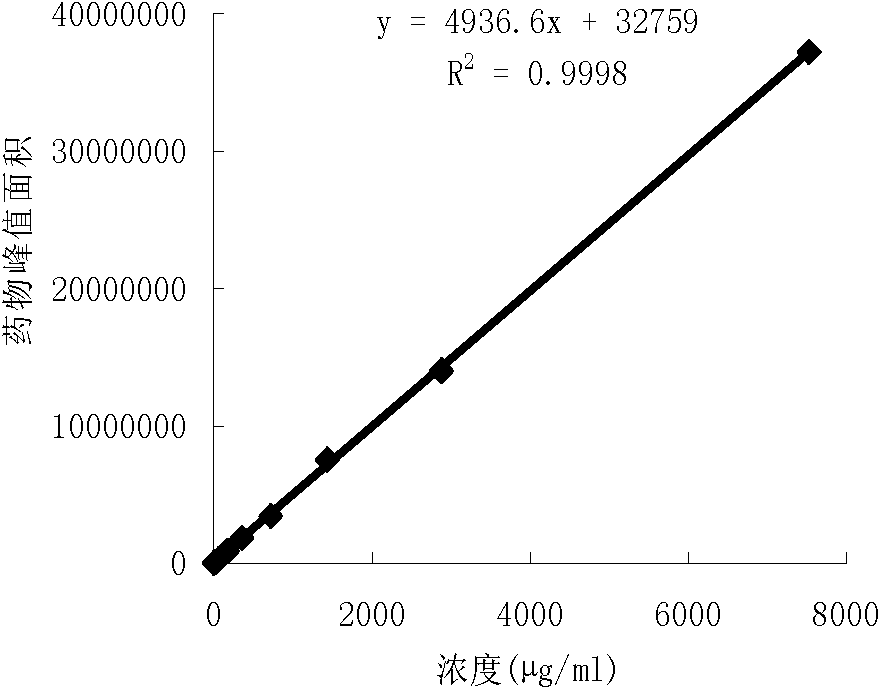
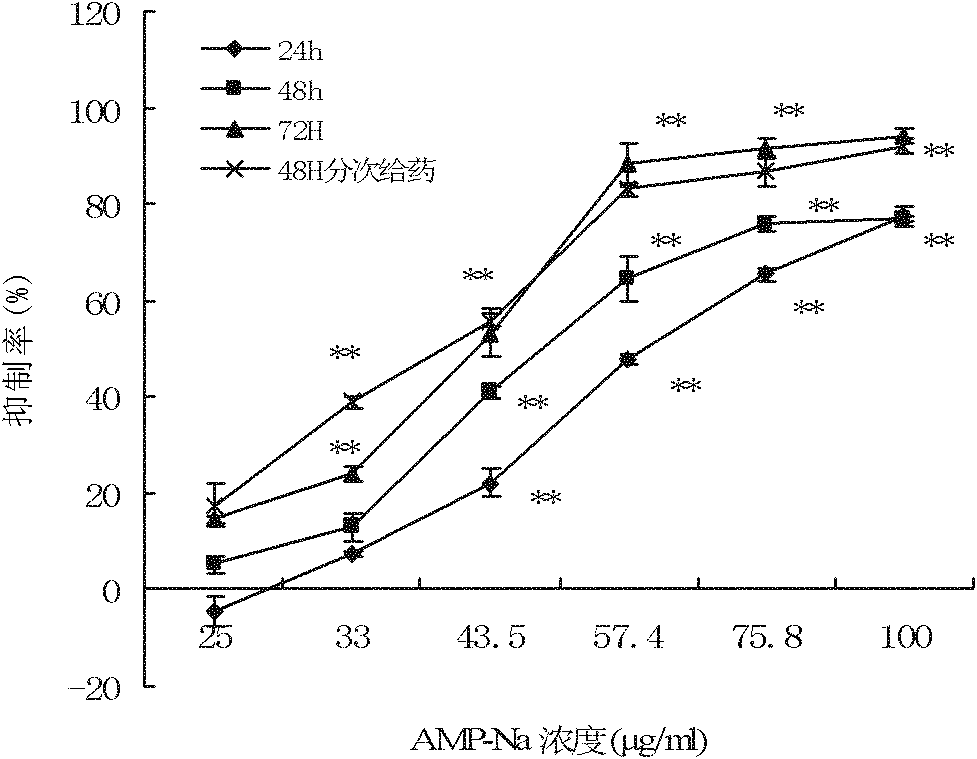
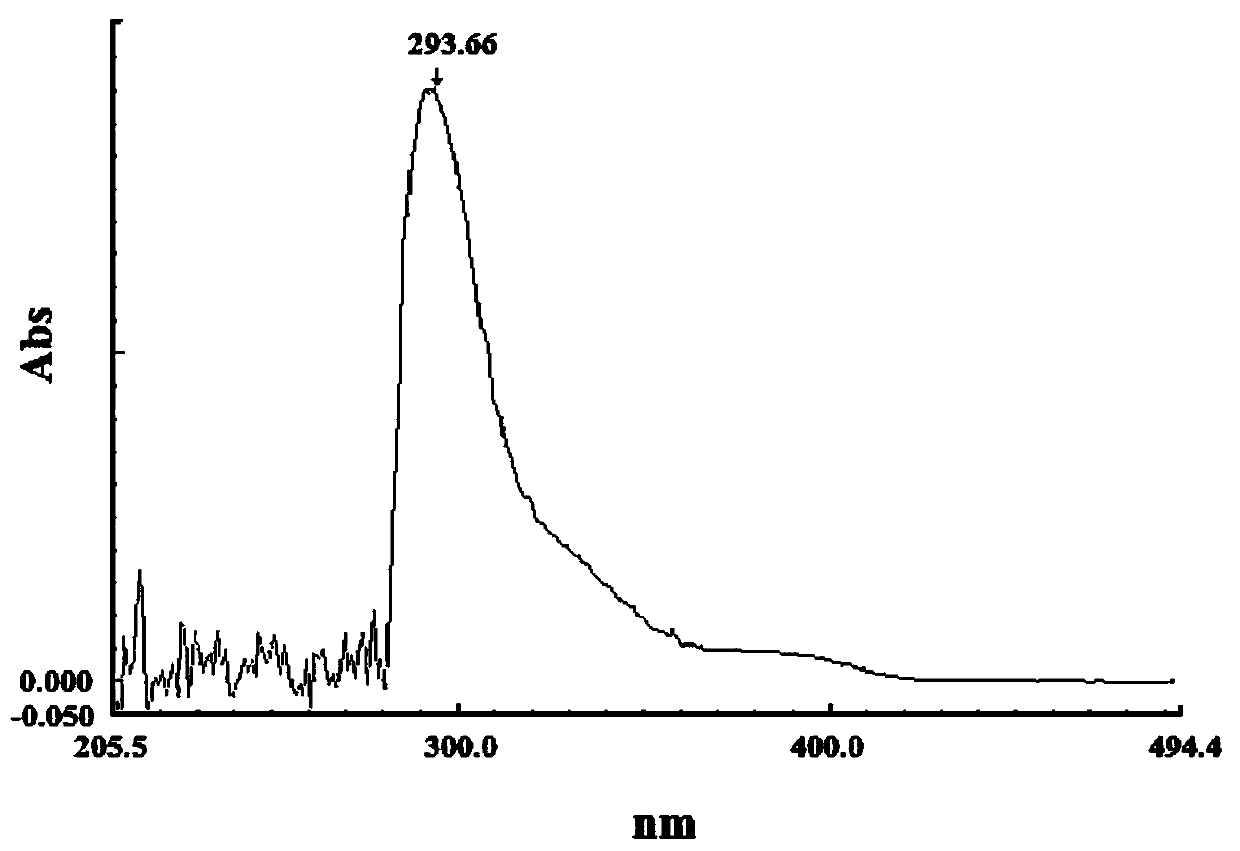
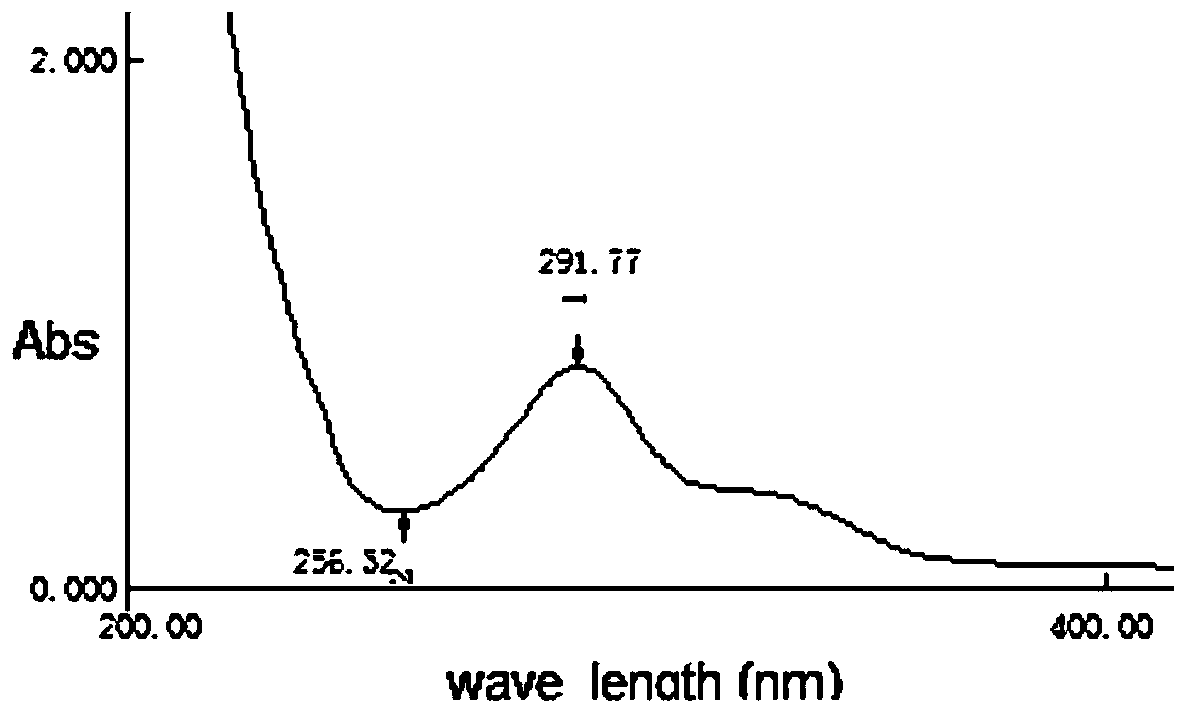
The present application relates to a novel AMPelopsin unsaturated sodium salt compound (AMP-Na), a method for preparing the same and the applications thereof. The physicochemical properties of AMP-Na according to the present application are significantly altered compared to those of AMPelopsin (AMP). AMP-Na shows significantly reduced toxicity than AMP as evidenced by the in vivo acute toxicity studies. As demonstrated by pharmacological evaluation, AMP-Na exhibits synergistic action when administered in combination with clinically used anti-cancer drugs, therefore reducing their dosages without compromising their therapeutic effects.
Owner:FAN FULIN
Application of ampelopsin in preparation of medicine for preventing and treating AIDS
InactiveCN1197570CImmune functionEnhance immune functionOrganic active ingredientsAntiviralsTherapy HIVAmpelopsin
Application of staphylophyllin in the preparation of anti-AIDS drugs. The present invention investigates the anti-HIV-1 effect of staphylosin in vitro, the regulation function of cellular immune function in the mouse immunosuppressive model, and observes the effect of staphylosin on human PBMC infected by HIV-1. S and MT-4 cell protection, and the inhibitory effect on HIV-1 co-receptor CXCR4, indicating that staphylosin has anti-HIV effect in vitro and can down-regulate PBMC S The CXCR4 on the surface can enhance the immune function of the mouse immunosuppressive model, indicating that staphylosin has the function of preventing and treating AIDS, and can be used to prepare medicaments for preventing and treating AIDS.
Owner:SUN YAT SEN UNIV
Traditional Chinese medicine for treating prosopoplegia
InactiveCN104000960ASafe and effective treatmentHigh cure rateNervous disorderPlant ingredientsAmpelopsinCimicifuga acerina
The invention relates to a traditional Chinese medicine for treating prosopoplegia. The traditional Chinese medicine is prepared from following raw medicinal materials comprising, by weight, 10 parts of keiske wormwood, 15 parts of tinospora sinensis (Lour.) Merr., 8 parts of Chinese starjasmine stem, 1 part of ampelopsin grossedentata, 80 parts of herba corydalis impatiens, 3 parts of cimicifuga acerina and 6 parts of licorice. A clinical experiment result proves that the traditional Chinese medicine in the invention can treat the prosopoplegia safely and effectively.
Owner:刘新霞
New application of ampelopsin sodium in treating bladder cancer
The invention provides a new application of ampelopsin sodium in treating bladder cancer. Particularly, the invention provides a pharmaceutical composition containing ampelopsin or its pharmaceutically acceptable salts. The medicine is administrated intravenously, can effectively treat cancers of the urinary system, especially bladder cancer.
Owner:GUANGDONG TAIHE BIOPHARML
A kind of water-soluble staphylophyllin polymer
ActiveCN106750272BProlong biological half-lifeImprove solubilityAntibacterial agentsOrganic active ingredientsDiseaseAcute hyperglycaemia
The invention discloses a water-soluble ampelopsin polymer. The polymer can be used for treating cancer, inhibiting bacteria, protecting the liver, lowering blood sugar and lowering blood fat and contains composition, namely, PGA-AMP, of a medicine conjugated with a water-soluble polymer selected from poly-1-glutamic acid, poly-d-glutamic acid and poly-d-1-glutamic acid. According to a preparation method of the water-soluble ampelopsin polymer product, polyglutamic acid and ampelopsin react to produce a polyglutamic acid derivative under the action of a condensation agent and a catalyst, the condensation agent is 1-ethyl-(3-dimethylaminopropyl)carbodiimide hydrochloride and / or N-hydroxysuccinimide and / or dicyclohexylcarbodiimide, and the catalyst is 4-dimethylamino-pyridine. The polymer can be used for treating cancer, bacterial infection, liver diseases, hyperglycemia and hyperlipidemia.
Owner:FUJIAN HEALTH COLLEGE
A health-care tea with the effects of strengthening the body and nourishing the qi and improving the body's immunity and its preparation method
ActiveCN104068159BEasy to adjustGood for healthPre-extraction tea treatmentImmunological disordersDiseaseSide effect
The invention discloses health tea with the efficacy of strengthening the body resistance, nourishing qi and improving the immunity of an organism. According to the traditional Chinese medicine theory, the health tea provided by the invention is prepared by processing ampelopsin grossedentata, American ginseng, poria cocos, northern astragalus membranaceus, rhizoma polygonati, cherokee rose fruit, emblic leafflower fruit and glycyrrhiza according to certain compatibility by utilizing the property of traditional Chinese medicine and selecting a tea leaf processing technology. Raw material medicines in the formula are matched to be used, take five internal organs into account, can regulate the relevant functions of the five internal organs well, and have the efficacy of strengthening the body resistance and nourishing qi. The health tea provided by the invention is suitable for most people, has an obvious effect of improving the immunity of human bodies and intensifying the capability of resisting diseases of an organism, is obvious in heath protection effect, is low in cost, is convenient and practical, is good in mouthfeel, and has no side effect.
Owner:刘友章
Uses of ampelopsin and total flavone valid target rich in ampelopsin in preparing medicament for preventing and treating prostate gland cancer
InactiveCN101347425BLittle side effectsPC3 tumor weight in mice significantly reduced side effectsOrganic active ingredientsAntineoplastic agentsSide effectPharmaceutical drug
Owner:福建省中医药科学院 +1
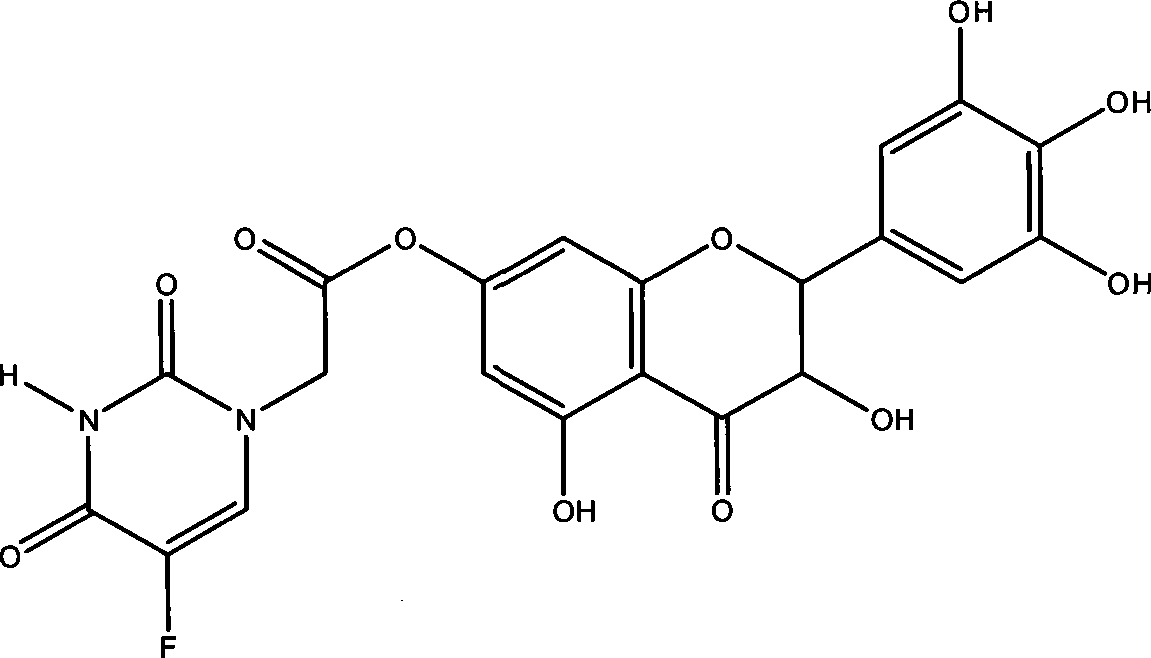
Ampelopsin pro-dug and preparing method and application thereof
InactiveCN101555241BAchieve clinical applicationImprove bioavailabilityAntibacterial agentsOrganic active ingredientsSolubilityPharmacy
The invention discloses an ampelopsin pro-drug and a preparing method and application thereof. The general formula (I) of the ampelopsin pro-drug is:, and compared with ampelopsin, the compound increases the water solubility obviously. In the preparing method, poisonous organic solvent is not used, all the solvents can be recycled, and other non-environmental-friendly reagents are not used. The ampelopsin pro-drug compound can be prepared into various dosage forms in pharmacy, including injection, tablets, capsules and granules, which is especially suitable for preparing powder-injection for injection.
Owner:FUJIAN HEALTH COLLEGE