Patents
Literature
55 results about "Sulindac" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
Sulindac is used to reduce pain, swelling, and joint stiffness from arthritis. It is also used to treat arthritis of the spine, gouty arthritis, and shoulder bursitis/tendonitis.
Method for inhibiting the growth of gastrointestinal tract tumors
InactiveUS20050147689A1Reduce morbidityPromote growthPeptide/protein ingredientsGranular deliveryGastrointestinal tumorWilms' tumor
The present invention provides compositions and methods for preventing the development of or reducing the growth of gastrointestinal tumors. The composition comprises polymeric microspheres encapsulating sulindac, IL-12 or both. These polymeric microspheres can be administered orally to individuals to reduce the growth of or prevent the development of gastrointestinal tumors.
Owner:THERAPYX
Treatment or prevention of cancer and precancerous disorders
InactiveUS20060235080A1Increase killEfficient removalBiocidePeptide/protein ingredientsCancer preventionMetabolite
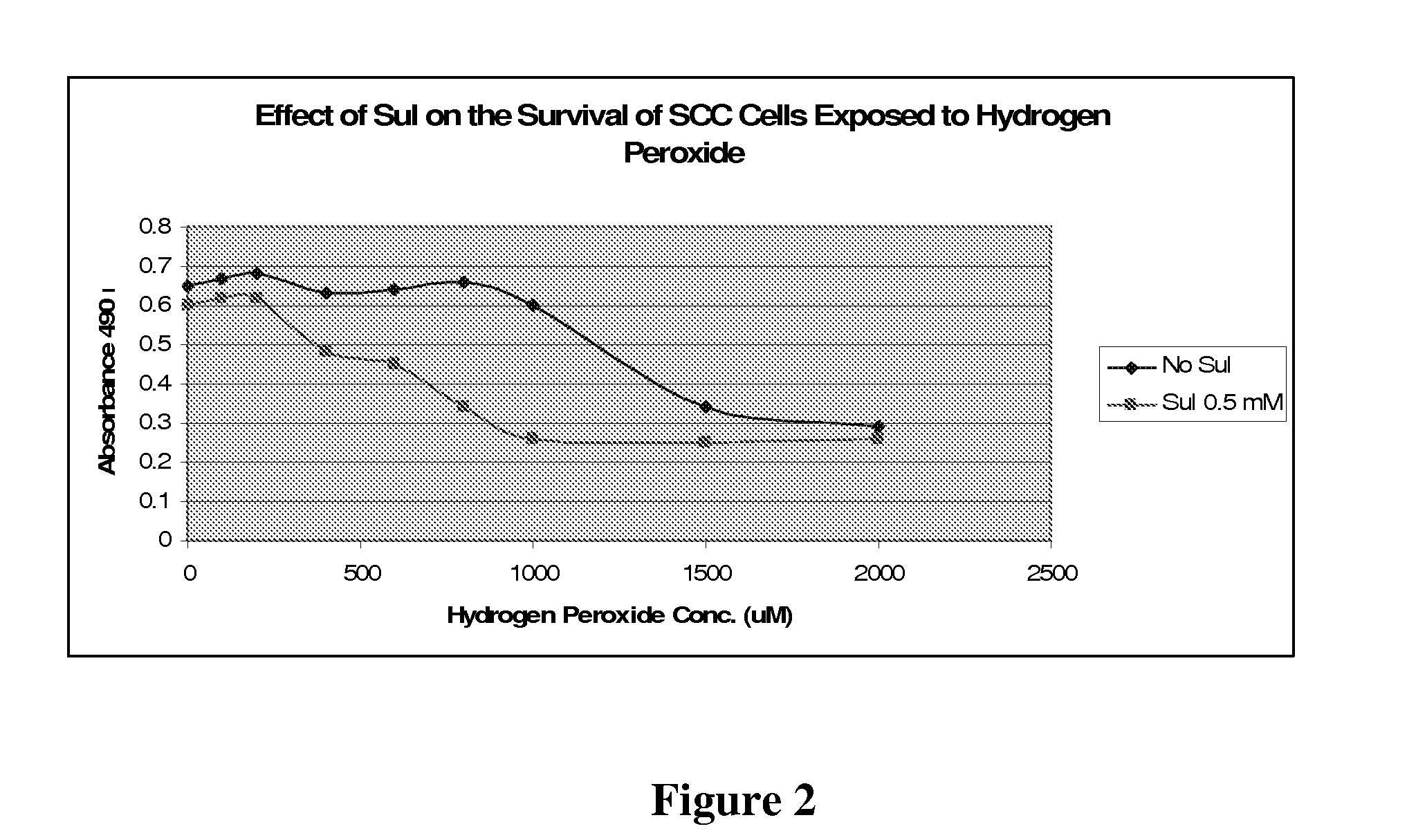
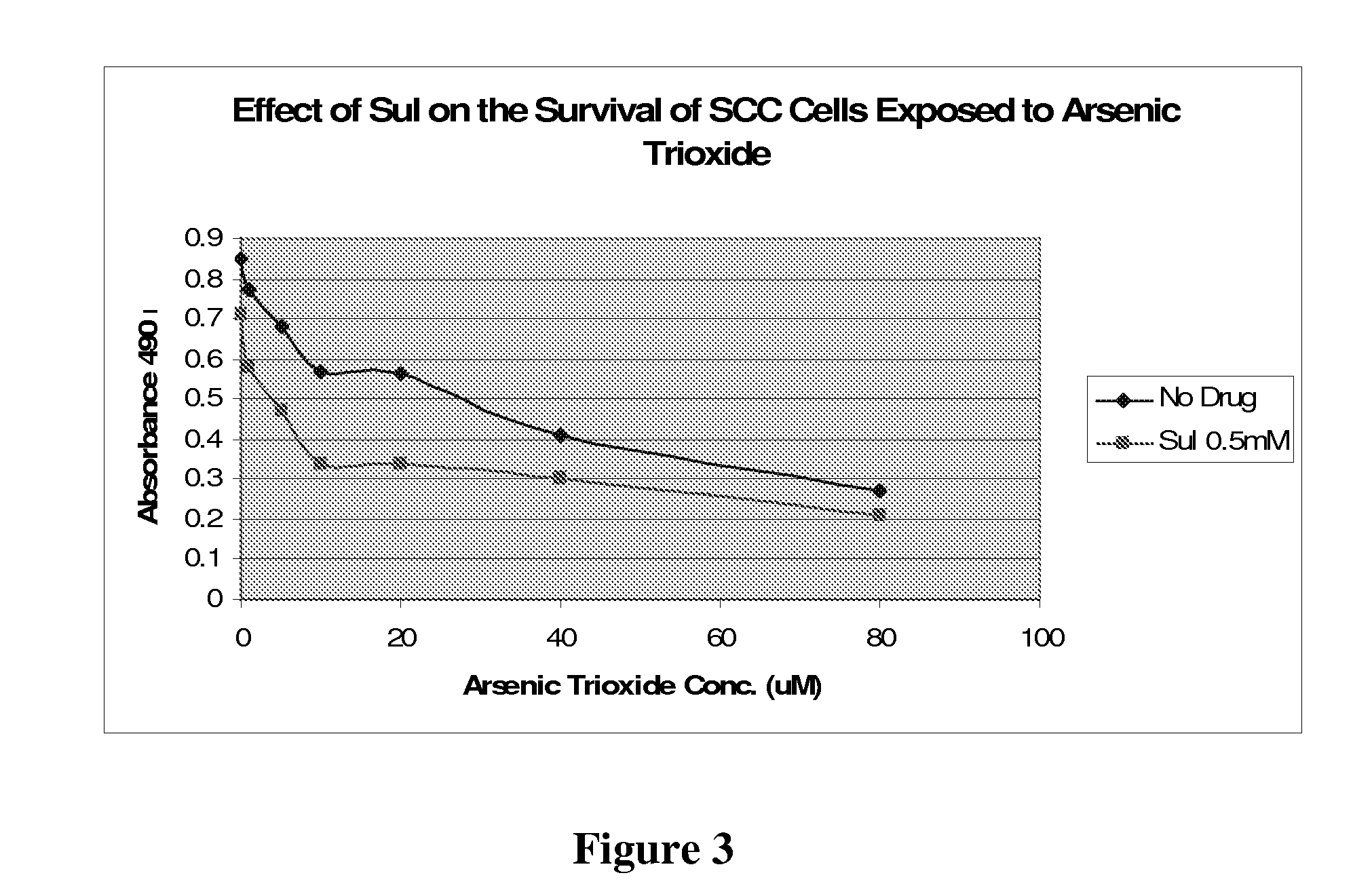
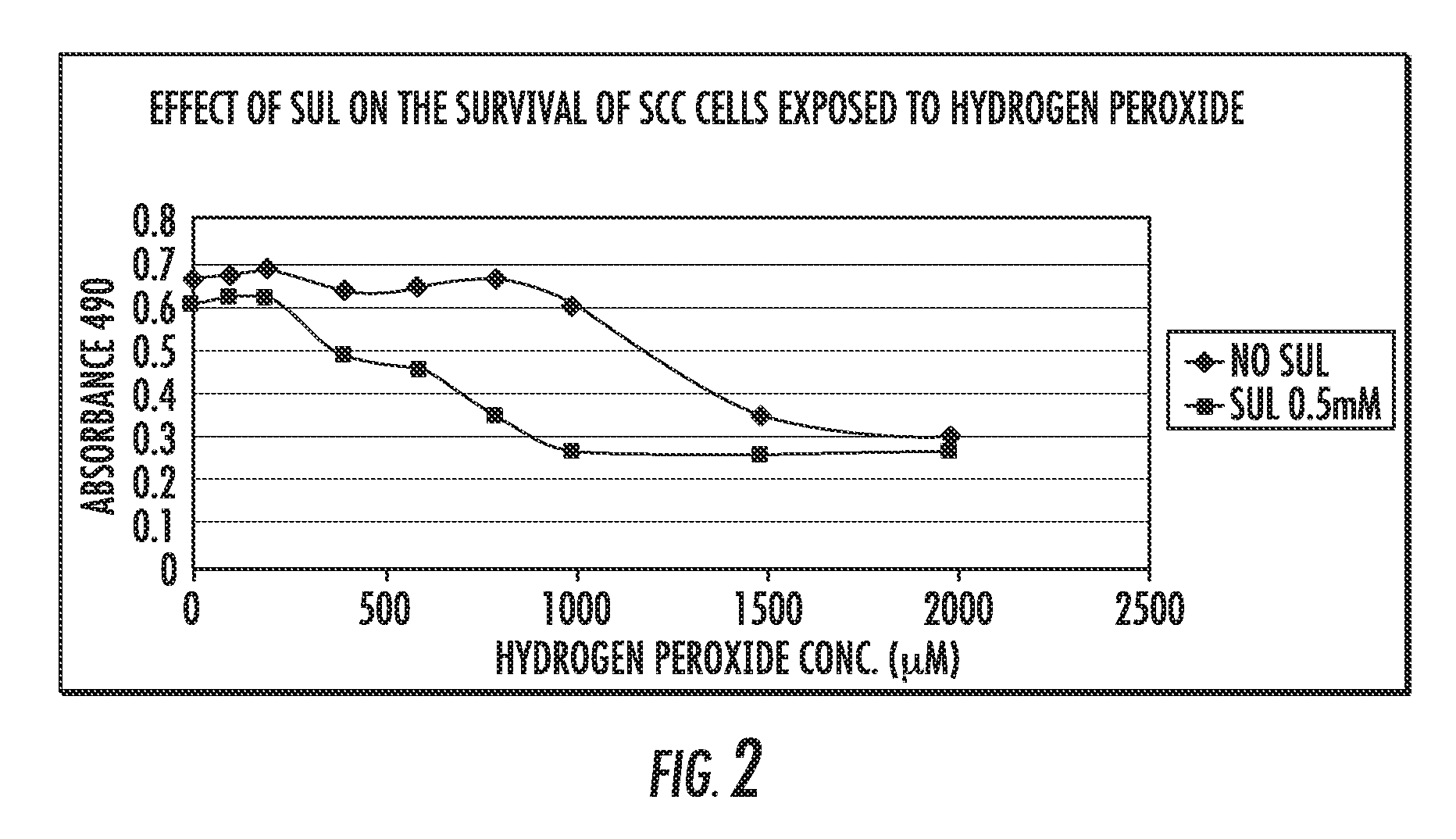
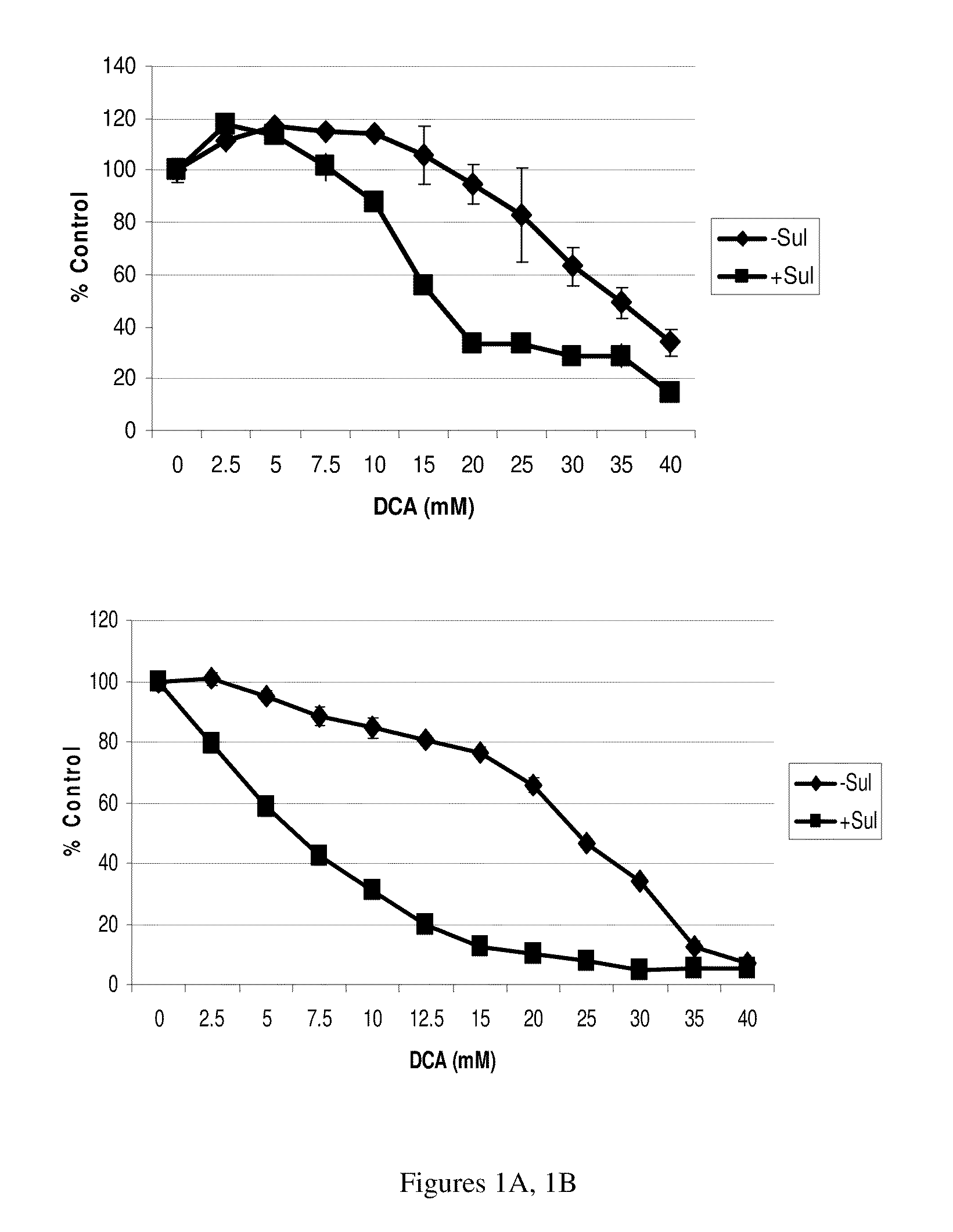
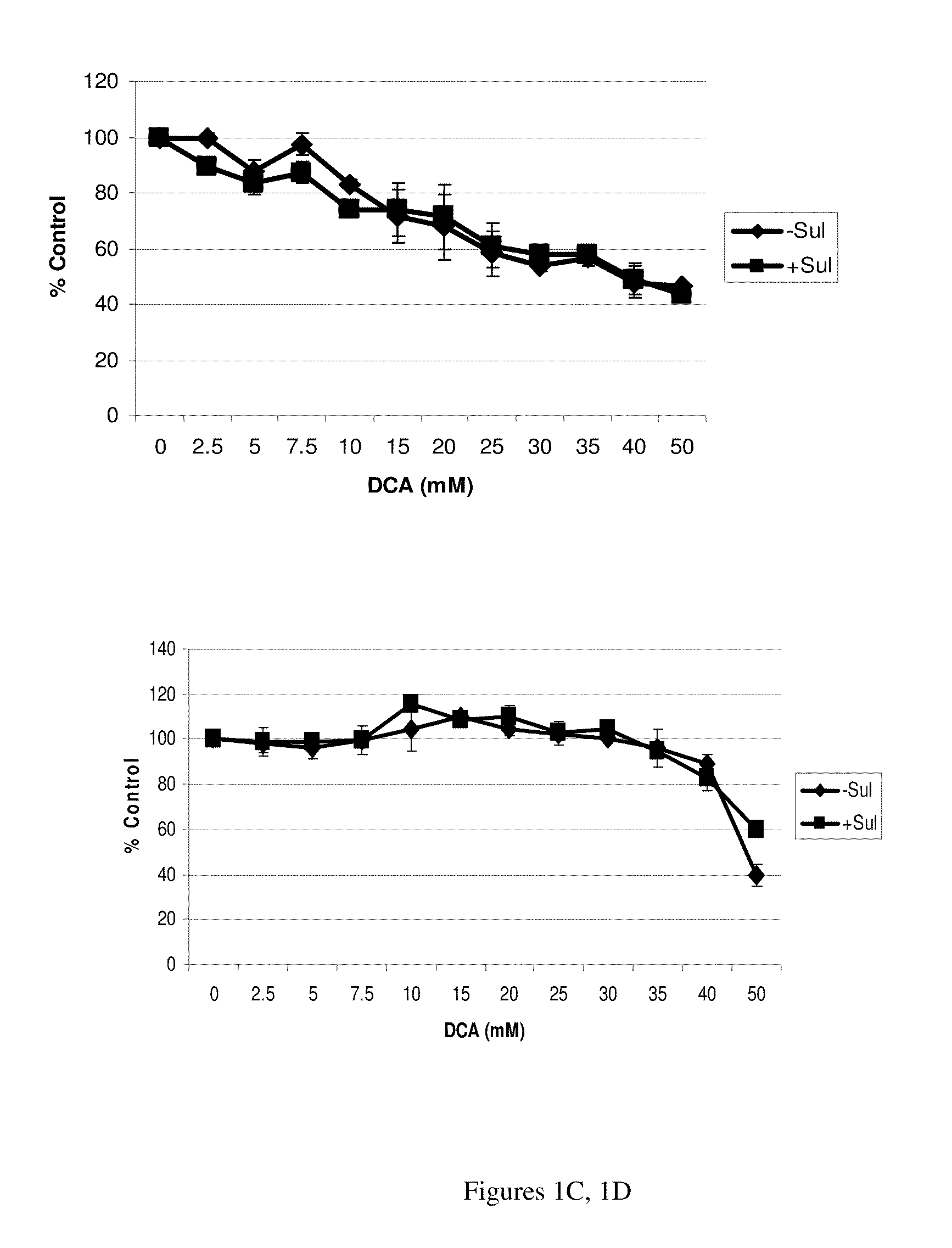
The NSAID, sulindac and / or its metabolites and derivatives, in combination with hydrogen peroxide or another oxidizing agent, such as arsenic trioxide that generates reactive oxygen species (ROS), significantly enhances the killing of cancer cells. This effect occurs at concentrations of each compound that individually have little or no activity directed against cancer cells. A skin cream has been developed and used to treat skin cancer and precancerous skin growths that effectively removes the lesions with no effect on surrounding normal skin.
Owner:CHS PHARMA
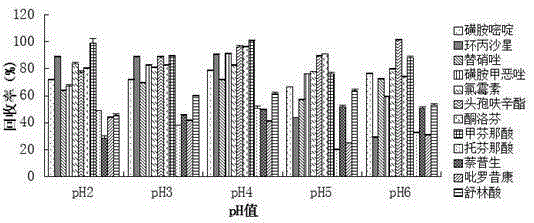
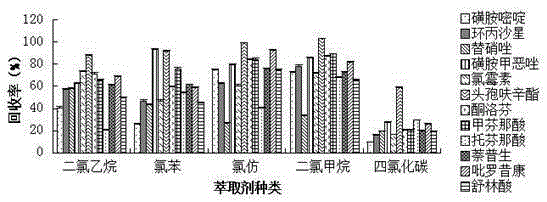
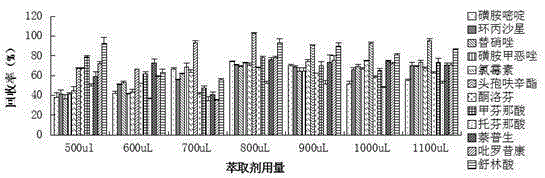
Method for measuring 12 types of remaining medicine in water environment through separation and enrichment
ActiveCN105424825ASimplified processing stepsImprove extraction efficiencyComponent separationWater dischargePretreatment method
The invention relates to a method for measuring 12 types of remaining medicine in a water environment through separation and enrichment at the same time, and belongs to the field of safety detection of a trace of organic pollutant residue in the water environment. The content of 12 types of frequently-used medicine in the water environment (drinking water, faucet water, river water and water discharged into and out of sewage treatment plants) is directly measured with an ultra performance liquid-chromatography-mass spectrometer (UPLC-MS / MS) as a detection tool after a water sample is subjected to solid phase extraction combined with ultrasonic-assisted dispersion liquid-liquid micro-extraction (UA-DLLME) separation and enrichment. The 12 types of antibiotic include ketoprofen, ciprofloxacin, tinidazole, tolfenamic acid, sulfadiazine, sulindac, naproxen, sulfamethoxazole, chloramphenicol, cefuroxime axetil, piroxicam and mefenamic acid. Inspection and optimization are conducted on a sample pretreatment method and instrument detection conditions of the water sample, and the optimal UA-DLLME method is established and is successfully applied to practical sample detection. Compared with a traditional method, the method has the advantages of being high in sensitivity, high in extraction and recycle rate, wide in suitable object, friendly to the environment, and the like.
Owner:SHENYANG PHARMA UNIVERSITY +1
Treatment or prevention of cancer and precancerous disorders
The NSAID, sulindac and / or its metabolites and derivatives, in combination with hydrogen peroxide or another oxidizing agent, such as arsenic trioxide that generates reactive oxygen species (ROS), significantly enhances the killing of cancer cells. This effect occurs at concentrations of each compound that individually have little or no activity directed against cancer cells. A skin cream has been developed and used to treat skin cancer and precancerous skin growths that effectively removes the lesions with no effect on surrounding normal skin.
Owner:CHS PHARMA
Topical formulation having effects on alleviating pain/inflammation caused by herpes virus infection
InactiveUS7132452B2Fasten skin recoveryPromote recoveryPowder deliveryBiocideComplete remissionTolmetin
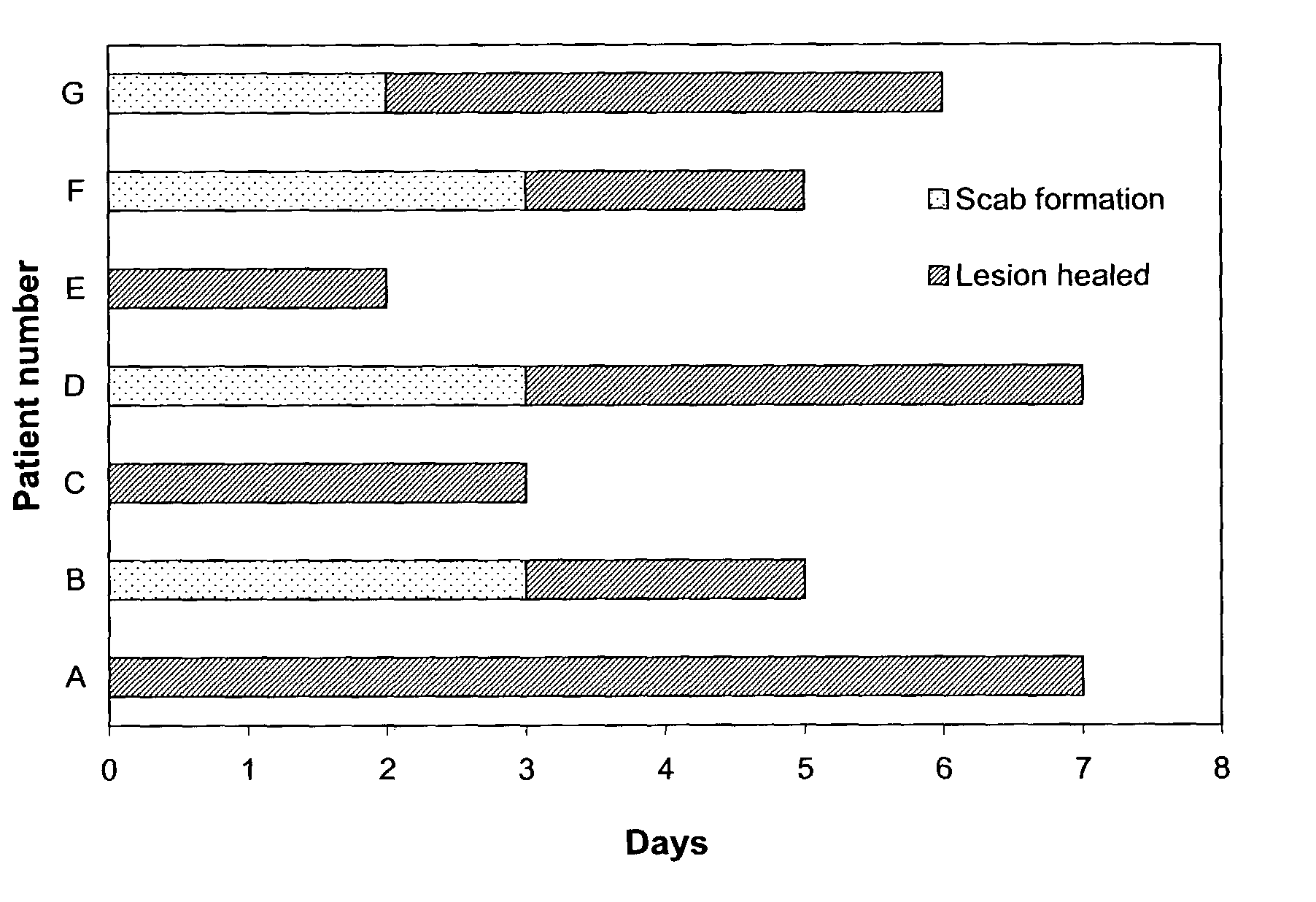
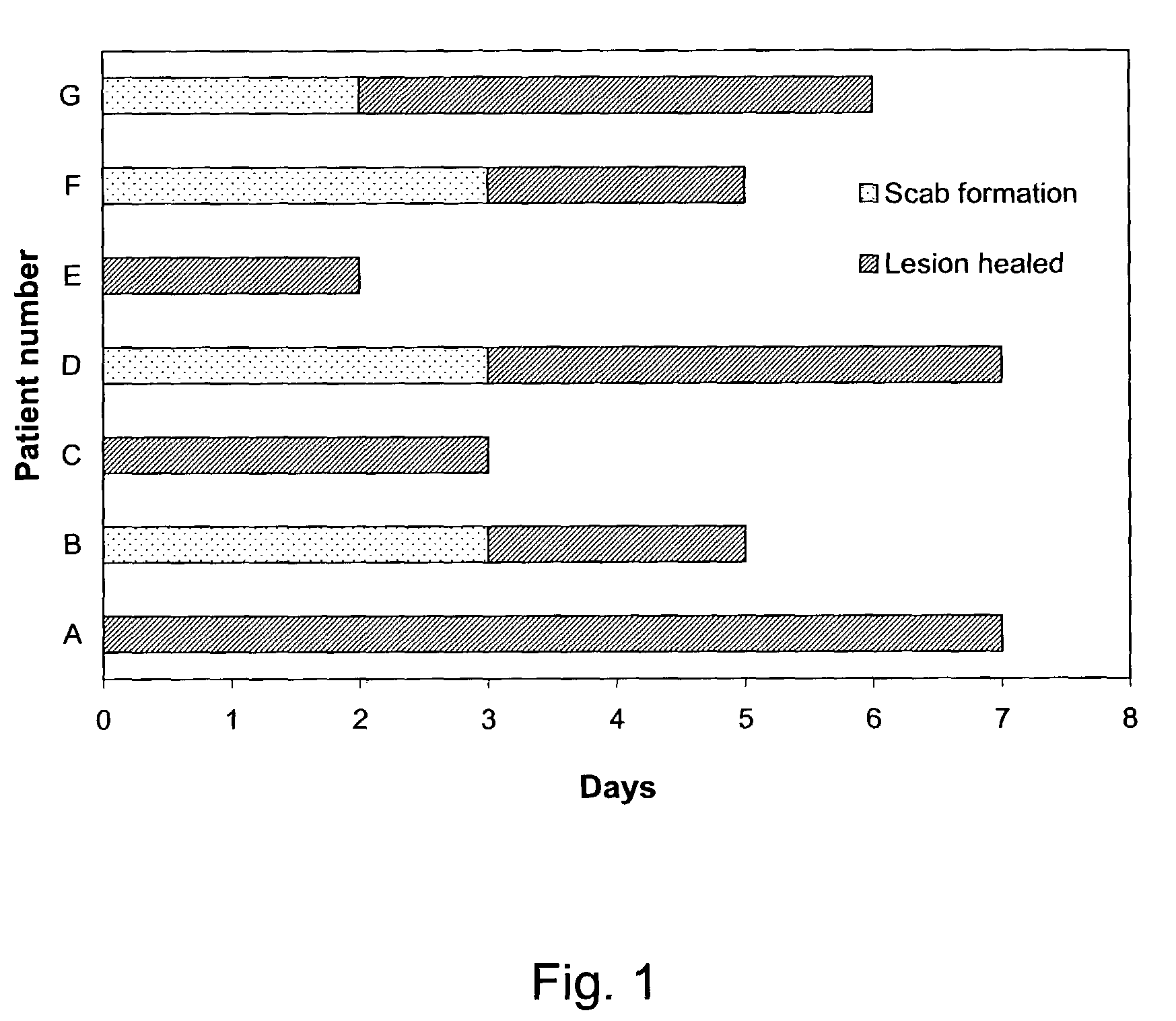
The present invention provides a topical formulation containing NSAID, particularly diclofenac. The topical formulation is particularly useful for alleviating pain / inflammation associated with infection caused by herpes virus, especially herpes simplex virus (HSV) and varicella-zoster virus (VZV). Similar relief can be achieved where diclofenac is replaced with another non-steroidal anti-inflammatory drug (NSAID), which includes, without limitation, etodolac, ketorolac, bromfenac, diflunisal, ibuprofen, fenoprofen, ketoprofen, naproxen, suprofen, meclofenamate, mefenamic acid, piroxicam, meloxicam, indomethacin, sulindac, phenylbutazone, oxyphenbutazone, and tolmetin. The topical formulation is further characterized by its fast relief on pain and / or inflammation associated with infection caused by herpes virus, i.e., a complete relief in no more than seven (7) days after the application of the topical formulation on skins of patients.
Owner:YUNG SHIN PHARMACEUTICALS INDUSTRIAL CO LTD
Preparation method of chiral sulfoxide medicament though catalysis of asymmetric oxidation of sulfides compound
ActiveCN104447692AEasy to synthesizeRaw materials are easy to getOrganic chemistryOrganic compound preparationManganeseSulfide compound
The invention provides a preparation method of a chiral sulfoxide medicament though catalysis of asymmetric oxidation of sulfides compounds. A chiral complex formed by quadridentate nitrogen organic ligand and metal manganese compound as a catalyst and hydrogen peroxide as an oxidant are used for asymmetric catalytic oxidation of prochiral thioether compound, so as to obtain the corresponding chiral sulfoxide medicament compounds including S-omeprazole, S-lansoprazole, S-pantoprazole, S-rabeprazole, R-Modafinil and R-sulindac. The reaction has the advantages of cleaness, mild reaction conditions, high conversion rate and antipodal selectivity, and shows industrial prospects.
Owner:DALIAN INST OF CHEM PHYSICS CHINESE ACAD OF SCI
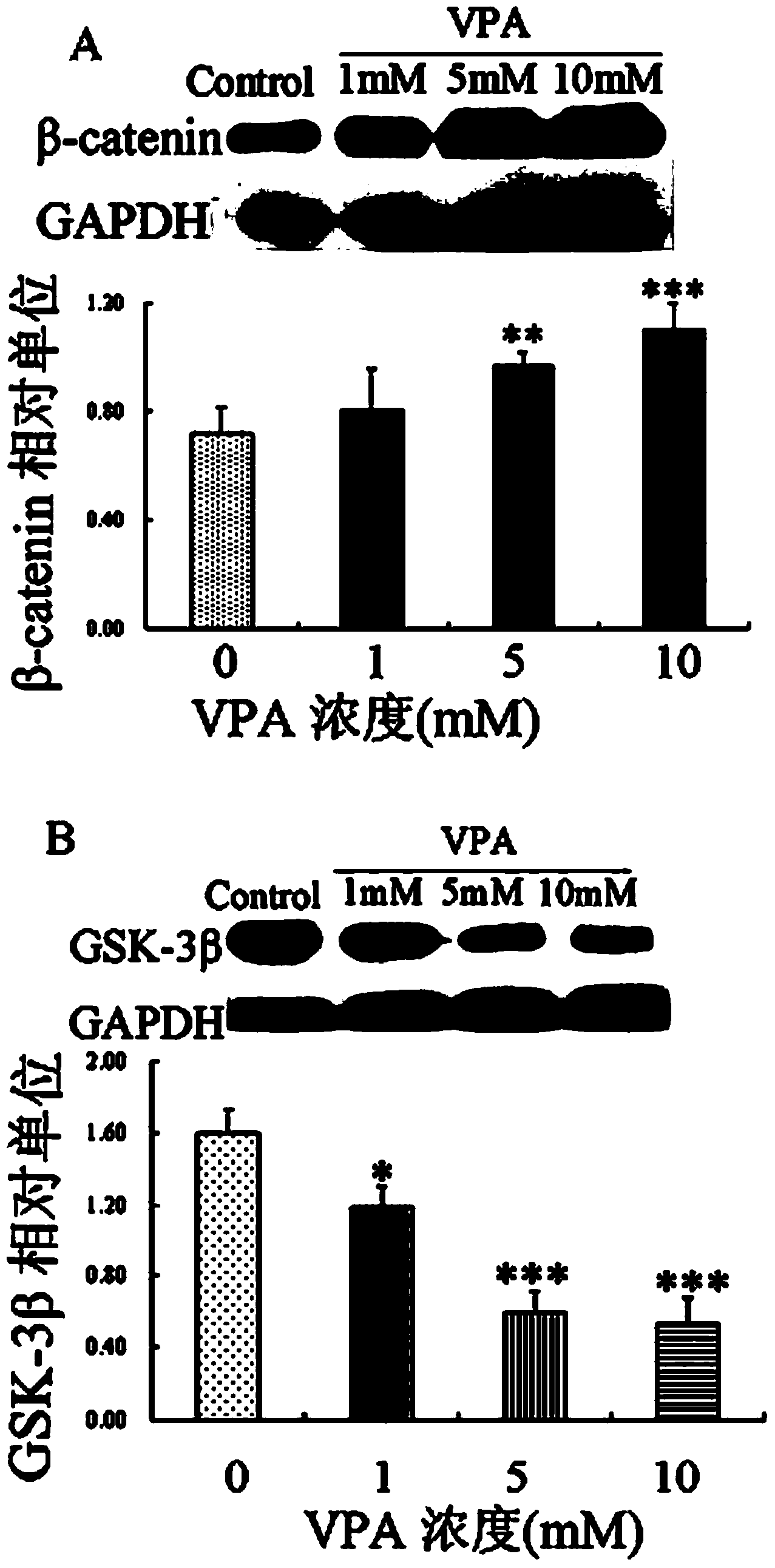
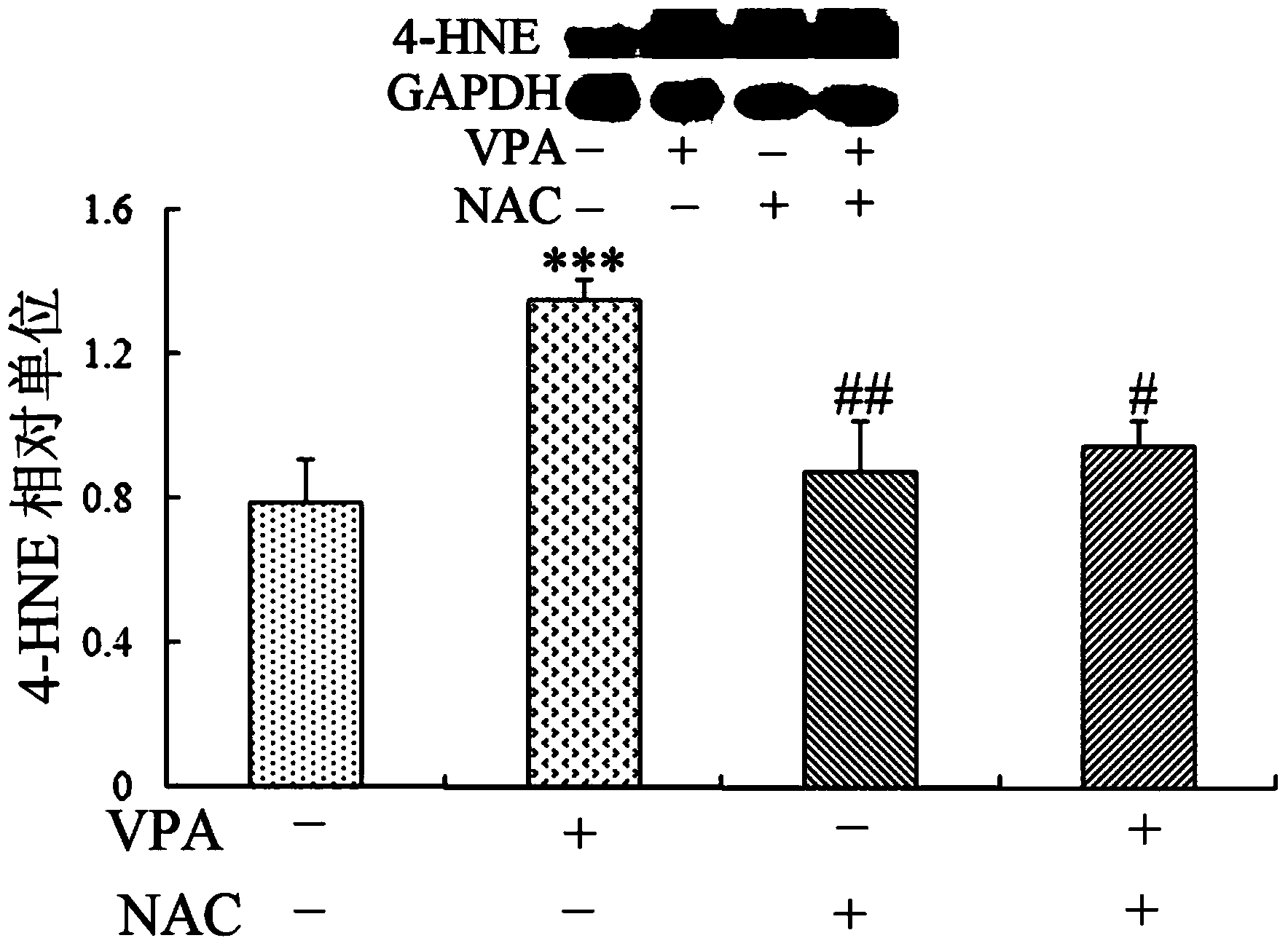
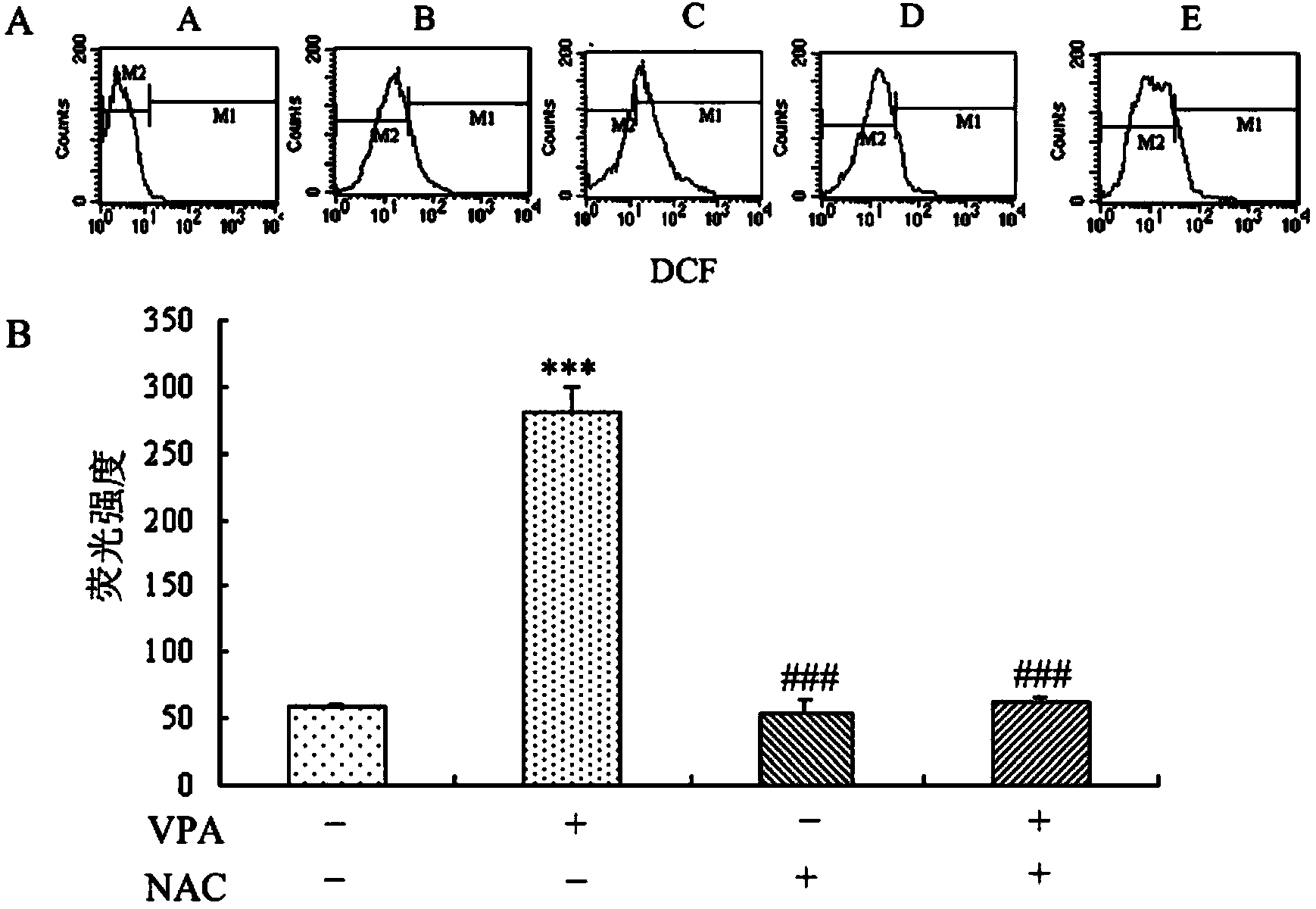
Application of sulindac in preparation of medicine for treating autism
InactiveCN103622941AHas antioxidant propertiesReduce generationOrganic active ingredientsNervous disorderValproic AcidSulindac
The invention discloses application of sulindac in preparation of a medicine for treating autism. The application of sulindac in preparation of the medicine for treating autism is characterized in that a VPA (Valproic Acid) autism animal model shows that sulindac is able to reduce abnormal behavior of an autistic, such as repeated mechanical behavior deficiency and human communication disorders; the primary culture neuron and VPA autism animal model verify the occurrence mechanism of autism as well as the functional mechanism of sulindac in treating autistic, and also prove that sulindac has oxidation resistance, and the oxidation resistance is related to down-regulating of the activity of a canonical Wnt signal channel. Therefore, sulindac is able to reduce the repeated mechanical behavior, abnormal behavior and human communication disorders of the autistic and can be used for preparing the medicine for treating autism.
Owner:XINXIANG MEDICAL UNIV
Treatment method of landfill leachate
InactiveCN103449664AGood removal effectStrong ability to change water qualityMultistage water/sewage treatmentSolubilityUltrafiltration
The invention discloses a treatment method of landfill leachate. The method comprises the following steps: removing suspended matters and floating matters in the landfill leachate by a mechanical grille, and adding PAC (polyaluminium chloride) and PAM (polyacrylamide) into the leachate to perform coagulation sedimentation and ultrafiltration treatment to remove metals, SS (sulindac sulfides) and partial organic matters in the leachate and reduce the treatment load of a follow-up membrane biological fluidized bed unit; then enabling the leachate after treatment to enter the membrane biological fluidized bed, maintaining the relatively high sludge concentration in the membrane biological fluidized bed, and performing biological treatment to remove soluble COD (chemical oxygen demand), BOD (biochemical oxygen demand) and ammonia nitrogen in the leachate; and finally removing the organic matters and salts, which are difficult to degrade, by a reverse osmosis membrane so as to realize up-to-standard emission of outlet water. The treatment method disclosed by the invention has the advantages of strong ability of being suitable for changes in water quality, high impact-resistant load, short treatment period, high treatment efficiency, good effect on removing pollutants, high removal rate of the COD, BOD and ammonia nitrogen of above 97%, small occupied area, high degree of automation and low cost.
Owner:NANJING WONDUX ENVIRONMENTAL PROTECTION TECH CO LTD
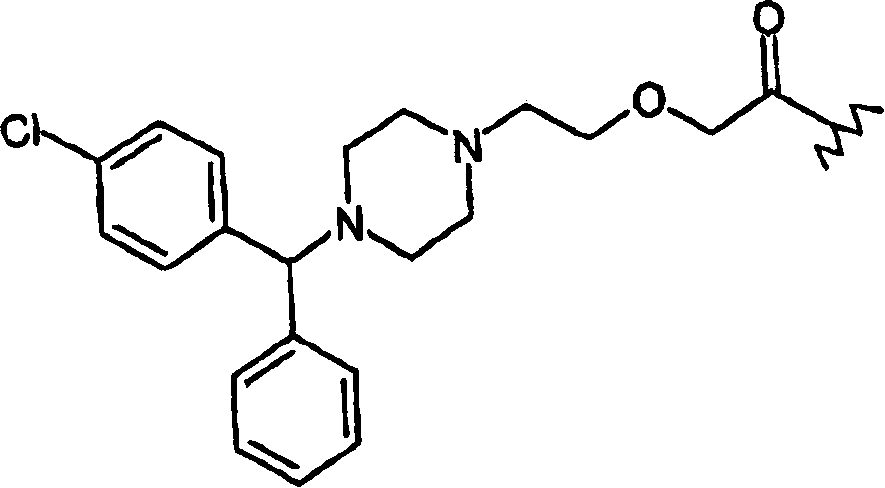
Pharmaceutical compounds
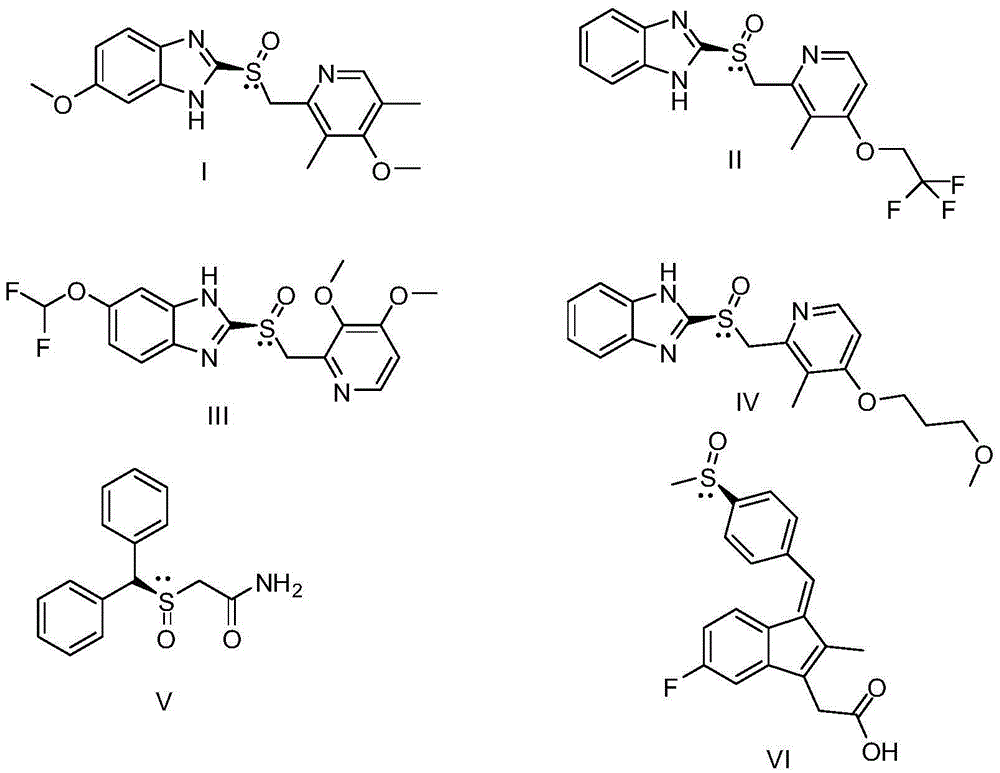
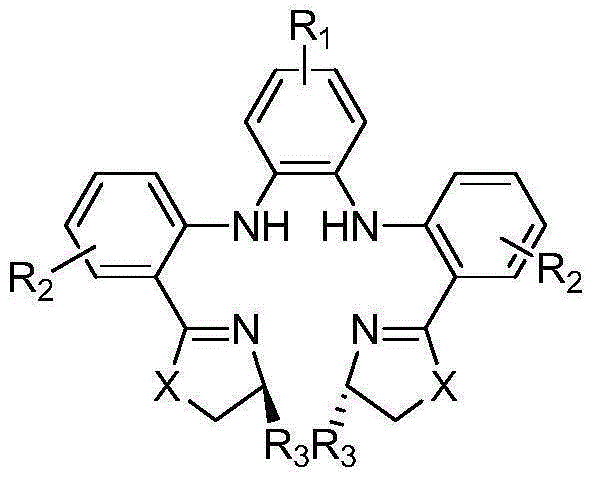
Compounds with the general formula (I): A-B-N(O)s or salts thereof, where: s is an integer equal to 1 or 2; A=R-T1-, where R is a drug residue and T1= (CO) t or (X) t′, where When t′=0, t=1; when t′=1, t=0; B=-TB-X2-O-, where TB=(CO) when t=0, when t′=0 TB=X, X is as defined above;
Owner:NICOX SA
Treatment or prevention of cancer and precancerous disorders
The NSAID, sulindac and / or its metabolites and derivatives, in combination with an agent that generates or induces reactive oxygen species (ROS), significantly enhances the killing of abnormal cells but does not affect normal cells. This effect occurs at concentrations of each compound that individually have little or no activity directed against the abnormal cells.
Owner:CHS PHARMA
Synthesis method and application of intermediate of sulindac analogue
InactiveCN102030642AReduce dosageAvoid it happening againOrganic compound preparationCarboxylic acid esters preparationKetoneSolvent
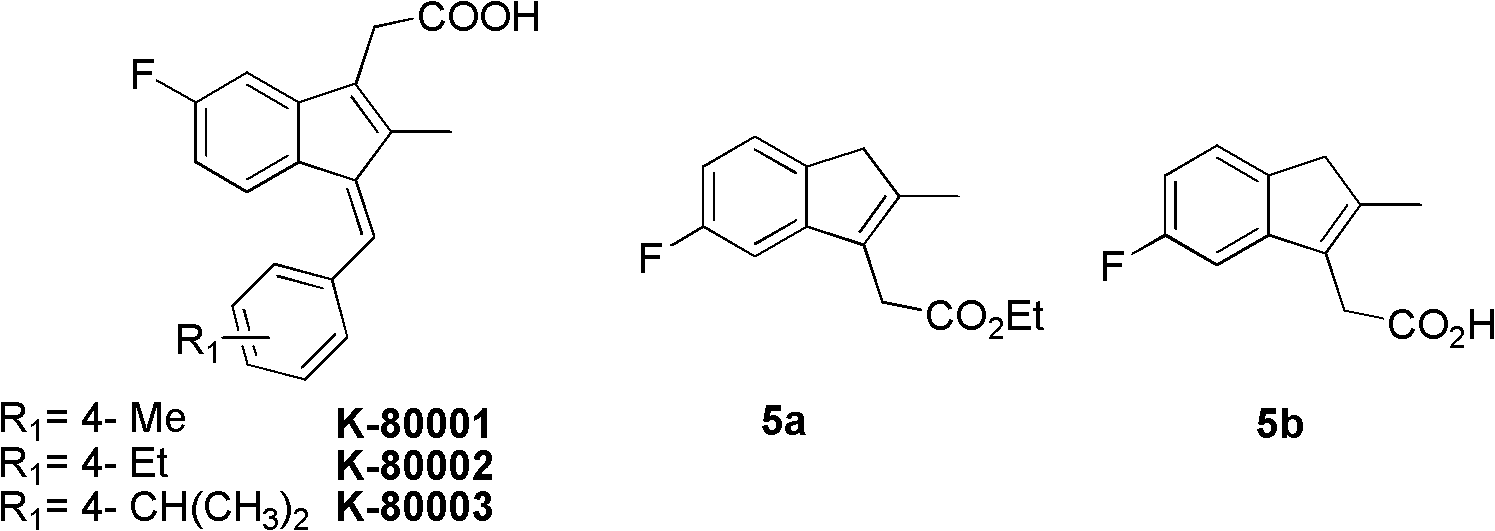
The invention discloses a synthesis method and application of intermediates of a sulindac analogue, relating to intermediates of a sulindac analogue. The intermediates are 5-fluoro-2-metyl-3-indene ethyl acetate (5a) and 5-fluoro-2-metyl-3-indene acetic acid (5b). The synthesis method comprises the following steps of: subjecting 4-fluorobenzaldehyde as an initial raw material, propionic anhydride as a solvent and propionic anhydride to a perkin reaction to obtain 4-fluoro-2-metyl-methylcinnamic acid; catalyzing the 4-fluoro-2-metyl-methylcinnamic acid with palladium carbon with the palladium content of 5-20 percent and reducing in the hydrogen gas atmosphere to obtain 3-(4-fluorine phenyl)-2-methyl propionate; subjecting the 3-(4-fluorine phenyl)-2-methyl propionate to the intramolecilar friedel-crafts acyl browning reaction under the action of polyphosphoric acid under the heating condition to form 6-fluoro-2-methyl indene ketone; and subjecting the 6-fluoro-2-methyl indene ketone and halogenated acetate to reformatsky reaction under the action of the activated zinc powder to obtain a crude product and eliminating the crude product in an acid solution to obtain 5-fluoro-2-metyl-3-indene acetate. The intermediates can be used for preparing novel sulindac analogues with anticancer activity.
Owner:XIAMEN UNIV
Positively charged water-soluble prodrugs of aryl- and heteroarylacetic acids with very fast skin penetration rate
InactiveCN101506160AGood absorption rateSignificant effectOrganic chemistryDipeptide ingredientsSolubilityZomepirac sodium
The novel positively charged pro-drugs of aryl- and heteroarylacetic acids in the general formula(1) 'Structure 1' were designed and synthesized. The compounds of the general formula(1) 'Structure 1' indicated above can be prepared from functional derivatives of tolmetin, zomepirac, etodolac, amfenac, bromofenac, alclofenac, fenclofenac, acemetacin, indomethacin, sulindac, fentiazac, lonazolac, bendazac, 6MNA, ibufenac, and related compounds, (for example acid halides or mixed anhydrides), by reaction with suitable alcohols, thiols, or amines. The positively charged amino groups of these pro-drugs not only largely increases the solubility of the drugs, but also bonds to the negative charge on the phosphate head group of membranes and pushes the pro-drug into the cytosol. The results suggest that the pro-drugs diffuses through human skin 100 times faster than does tolmetin, zomepirac, etodolac, amfenac, bromofenac, alclofenac, fenclofenac, acemetacin, indomethacin, sulindac, fentiazac, lonazolac, bendazac, or related compounds. It takes 2-4 hours for tolmetin, zomepirac, etodolac, amfenac, bromofenac, alclofenac, fenclofenac, acemetacin, indomethacin, sulindac, fentiazac, lonazolac, bendazac, 6MNA, ibufenac, and related compounds to reach the peak plasma level when they are taken orally, but these prodrugs only took about 40-50 minutes to reach the peak plasma level when they are taken transdermally. In plasma, more than 90% of these pro-drugs can change back to the drug in a few minutes. The prodrugs can be used medicinally in treating any NSAIAs-treatable conditions in humans or animals. The prodrugs can be administered not only orally, but also transdermally for any kind of medical treatments and avoid most of the side effects of NSAIAs, most notably GI disturbances such as dyspepsia, gastroduodenal bleeding, gastric ulcerations, and gastritis.
Owner:于崇曦 +1
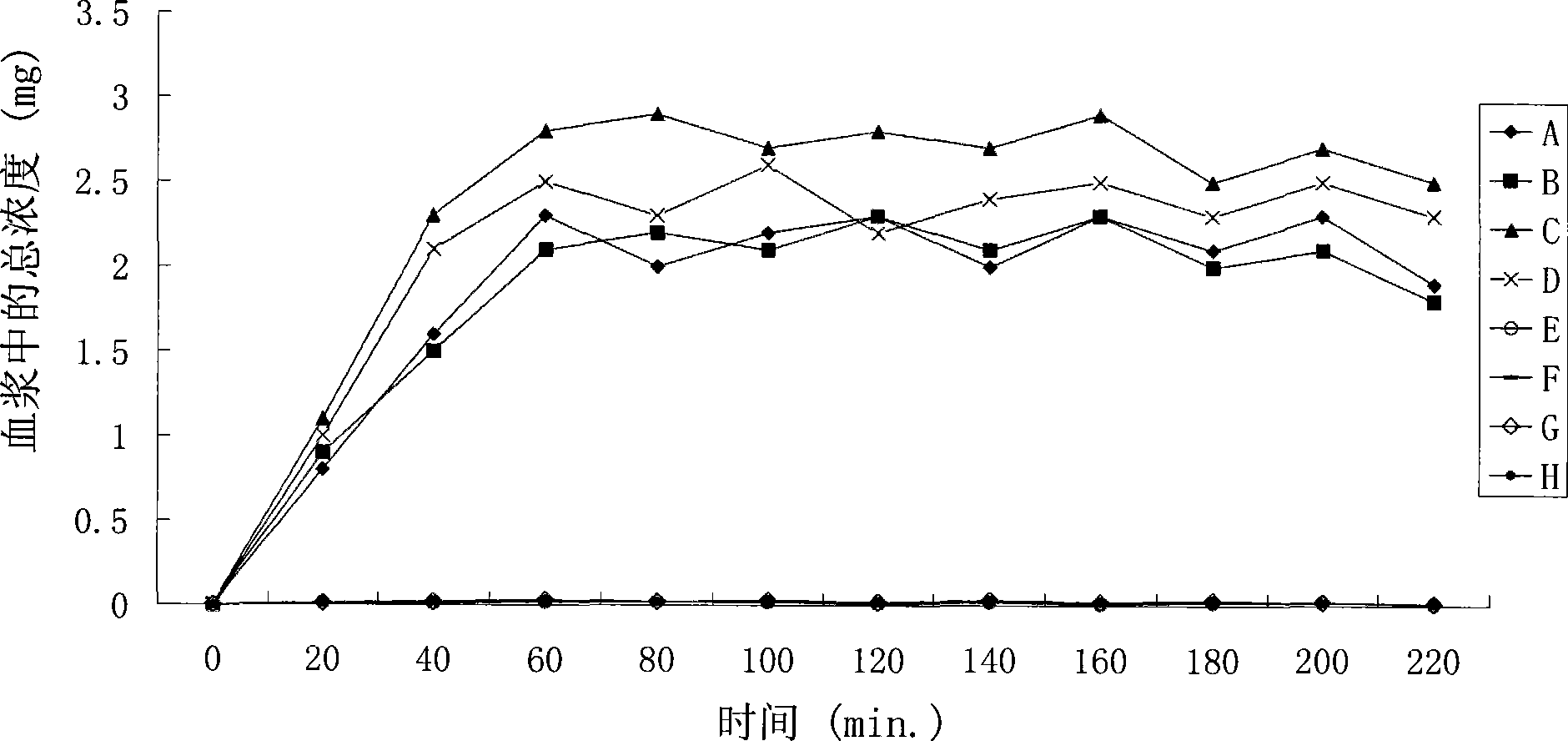
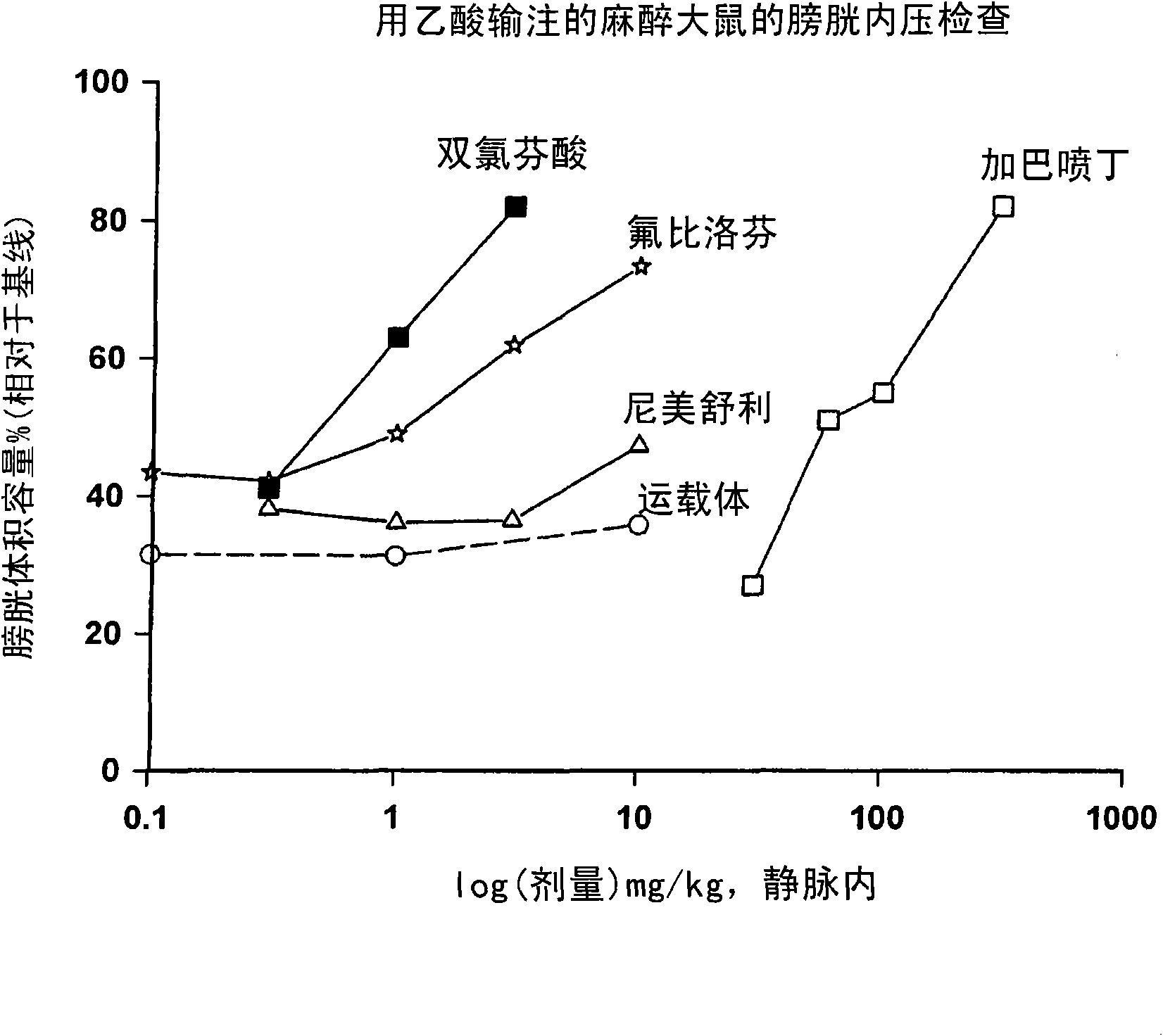
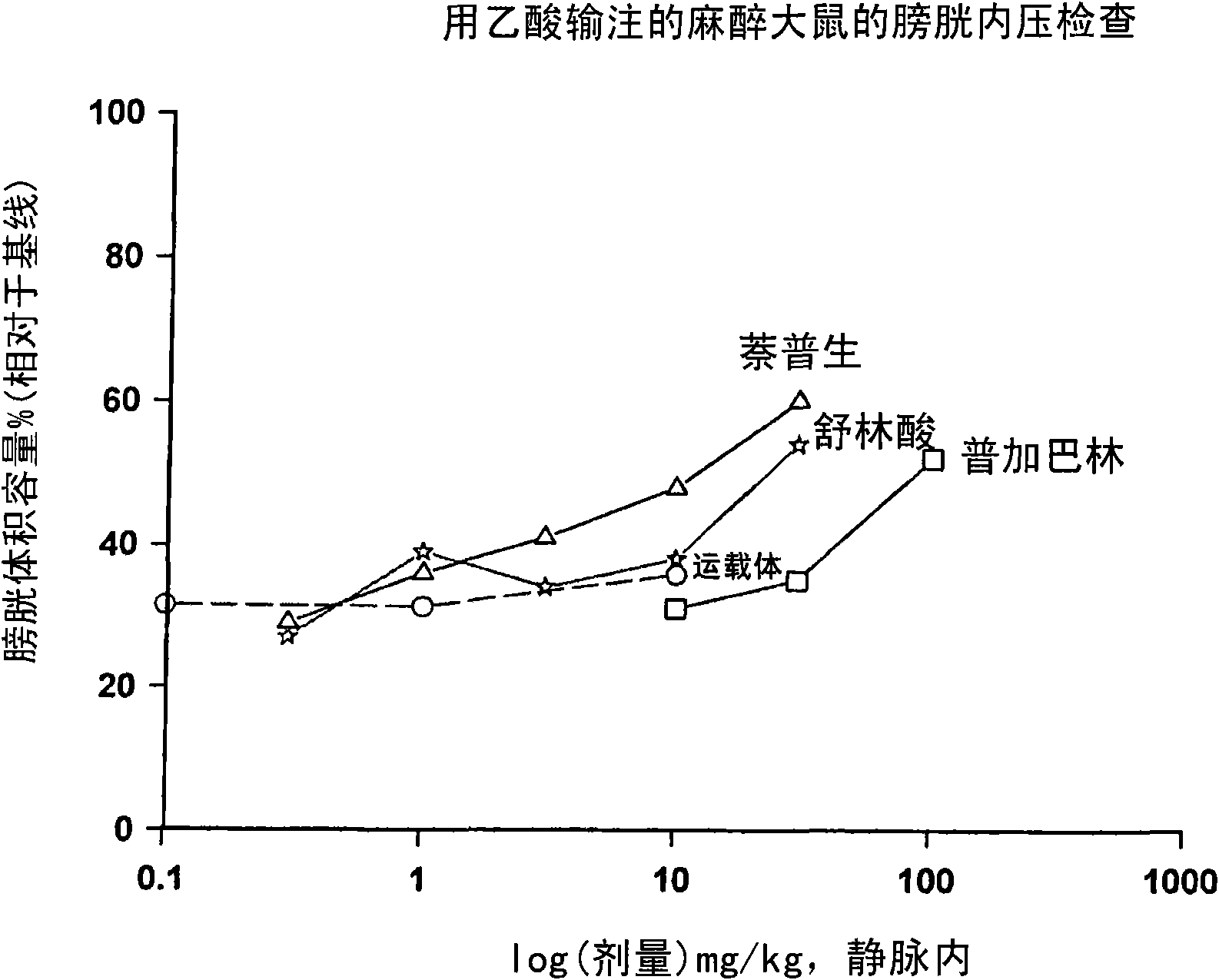
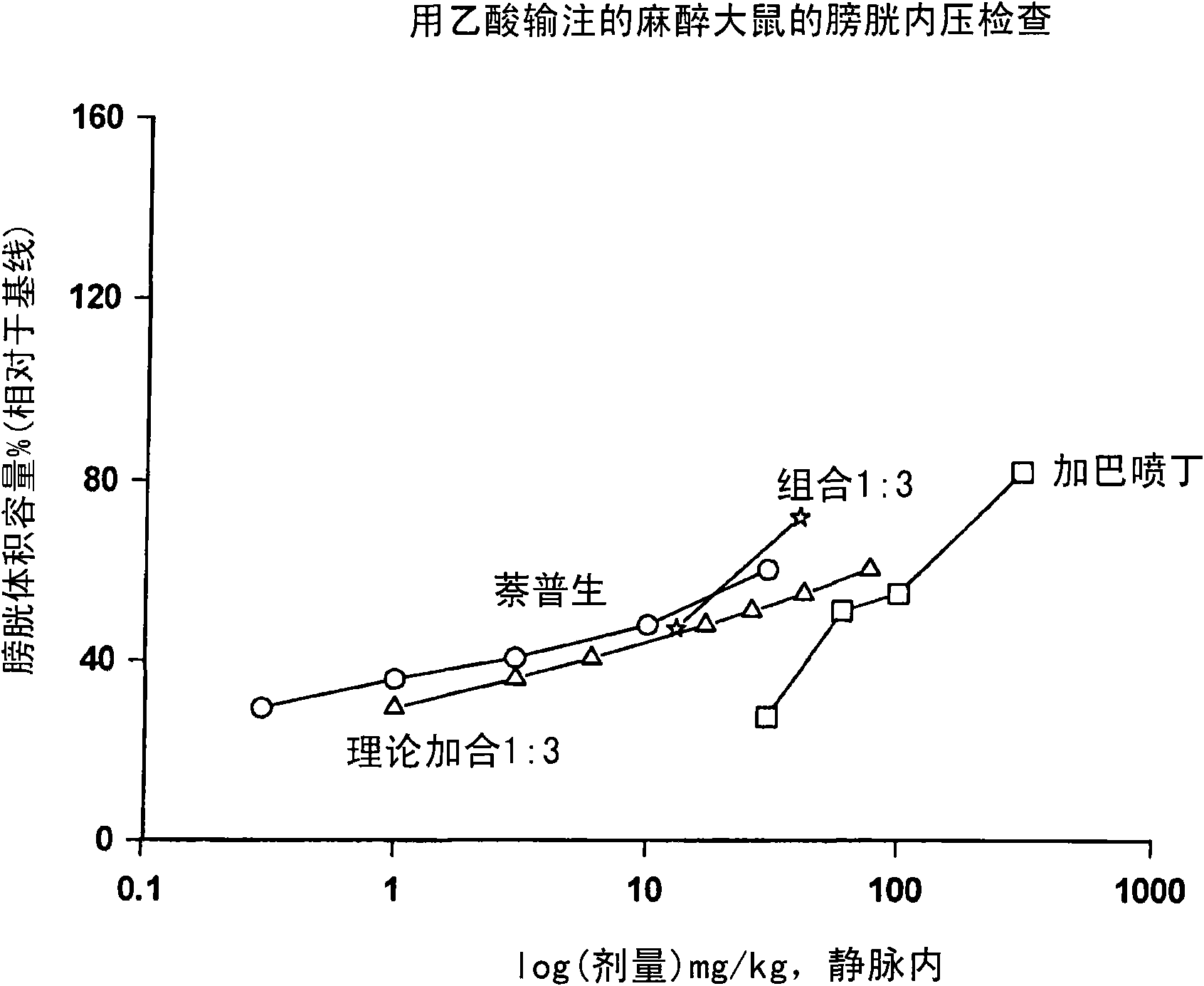
Combination therapy of lower urinary tract disorders with a2d ligands and nsaids
The administration to a mammal of a combination of compounds, at least one of which is an a2d calcium channel subunit (A2d) ligand and at least one of which is a non-steroidal anti-inflammatory drug (NSAID), provides a surprising and potent inhibition of the micturition reflex, superior to that obtained by treatment with an A2d ligand or NSAID alone. Combinations of A2d ligand and NSAIDs are thus useful for treatment of lower urinary tract disorders and symptoms thereof. Preferred A2d ligands are gabapentin and pregabalin. Preferred NSAIDs are celecoxib, diclofenac, diflunisal, flurbiprofen, naproxen, nimesulide or sulindac.
Owner:RECORDATI IRELAND LTD
Self-assembly system based on hydrophilic polymer and medicine and preparation method thereof
ActiveCN102091332AEasy to realize industrializationEasy to makeMacromolecular non-active ingredientsIndometacinPolymer science
The invention relates to a self-assembly system based on hydrophilic polymer and medicine and a preparation method thereof. The system consists of a hydrophilic polymer and a carboxyl-containing medicine, wherein the hydrophilic polymer is polymine or beta-cyclodextrin modified polymine; and the carboxyl-containing medicine is selected from ibuprofen, ketoprofen, fenoprofen, flurbiprofene, oxaprozin, naproxen, indometacin, sulindac, etodolac, diclofenac, pontal, meclofenamic acid, flufenamic acid, tolfenamic acid, lumiracoxib, licofelone, diflunisal and aspirin. The system is prepared by dissolving the hydrophilic polymer in a certain amount of water, dissolving the medicine in a water soluble solvent, slowly adding the organic solution of the medicine into the aqueous solution of the polymer under the action of ultrasound, placing the mixed solution in a dialysis bag, dialyzing in deionized water with magnetic stirring, replacing the deionized water at certain time interval, filtering dislysate after 5 to 48 hours, cooling and drying.
Owner:ARMY MEDICAL UNIV
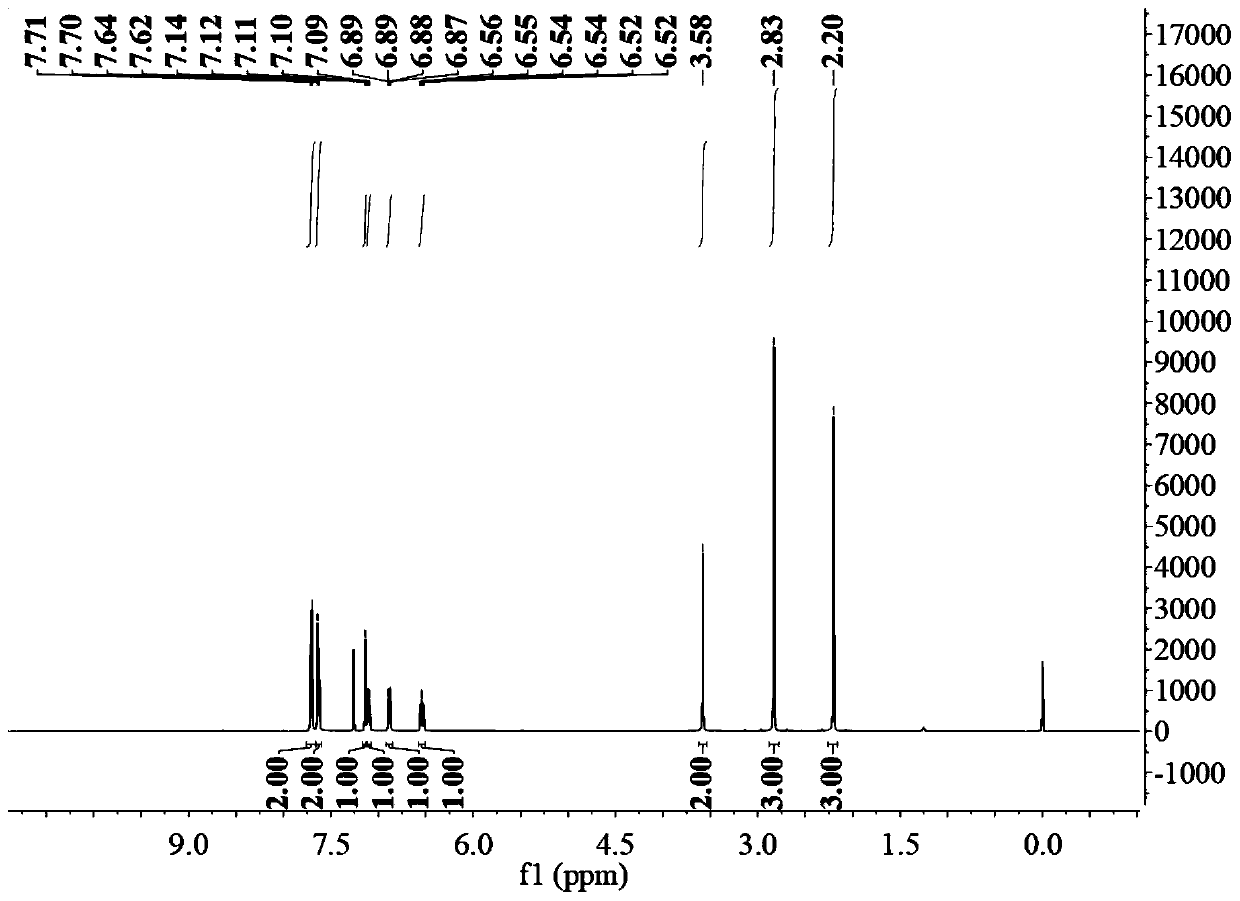
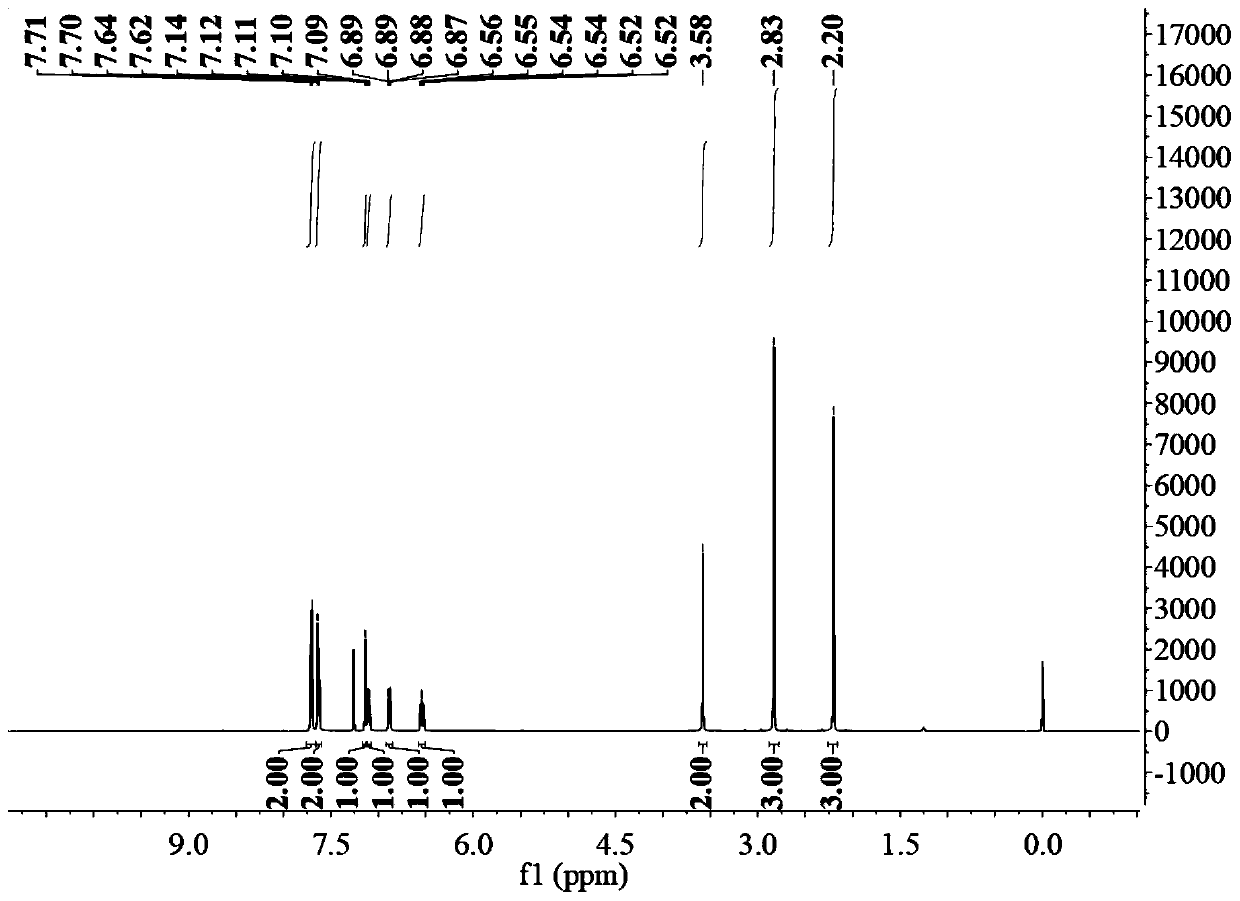
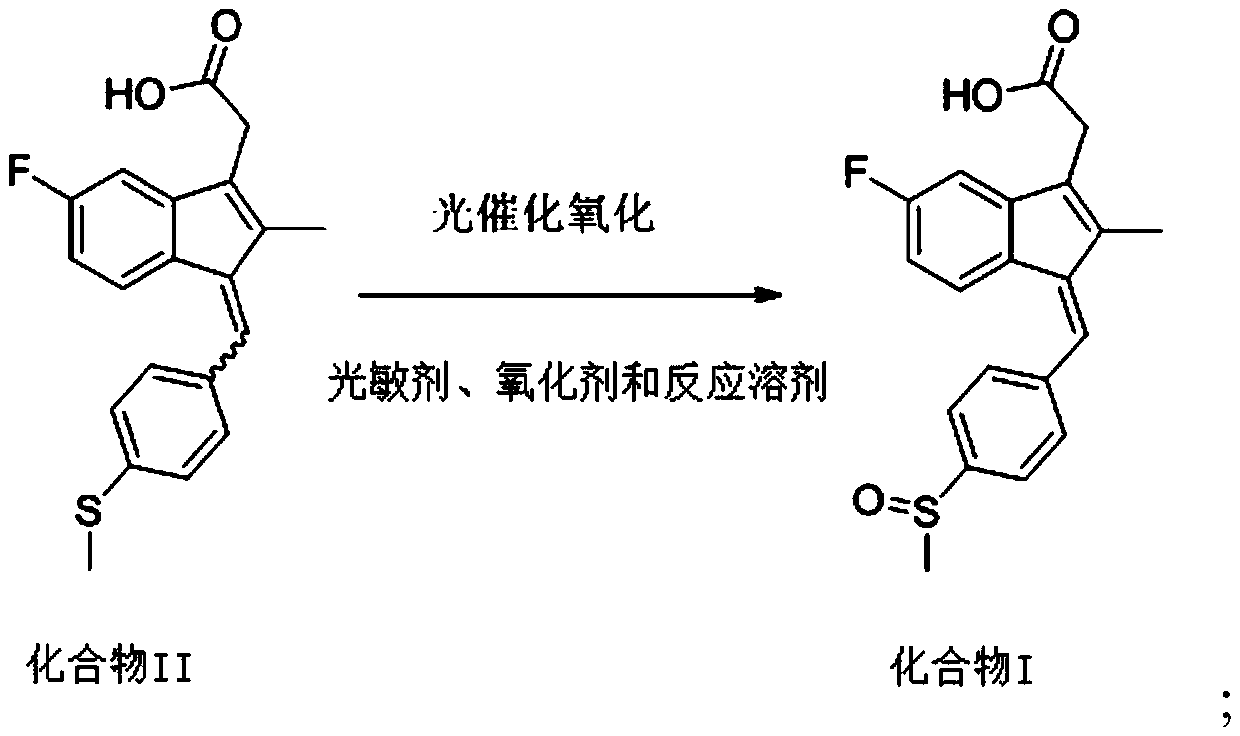
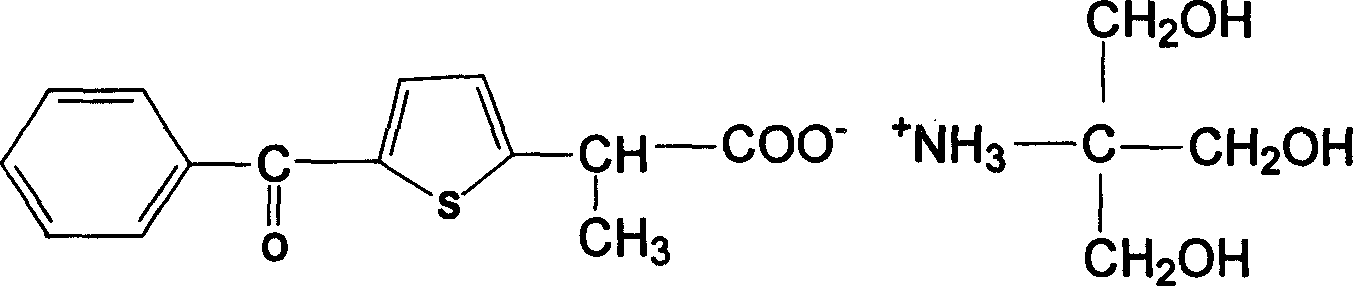
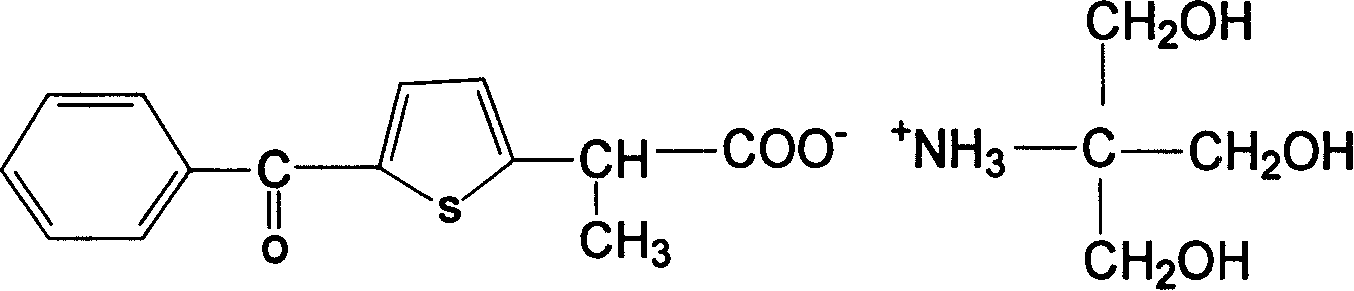
Method for preparing sulindac
ActiveCN110028430AHigh yieldHigh purityOrganic chemistryOrganic compound preparationAcetic acidPhotosensitizer
The invention relates to the technical field of medicine composition and in particular relates to a method for preparing sulindac. The method comprises the following steps: mixing 5-fluorine-2-methyl-1-(4-methyl thiobenzene methylene)-3-indene acetic acid, a photosensitizer, an oxidant and a reaction solvent, and carrying out an oxidation reaction under an ultraviolet radiation condition, therebyobtaining sulindac, wherein the 5-fluorine-2-methyl-1-(4-methyl thiobenzene methylene)-3-indene acetic acid comprises an E-shaped isomer and a Z-shaped isomer. By adopting the method provided by the invention, the sulindac is prepared from the 5-fluorine-2-methyl-1-(4-methyl thiobenzene methylene)-3-indene acetic acid of the E-shaped isomer and the Z-shaped isomer as raw materials through the oxidation reaction directly, separation purification is avoided, and the operation is simple. Experiment results of the embodiment show that when the sulindac is prepared by using the method provided by the invention, the yield is up to 98.9%, the purity is greater than 99.5%, and the method has the advantages of being high in yield and good in purity.
Owner:HANGZHOU LUPU BIOTECH CO LTD
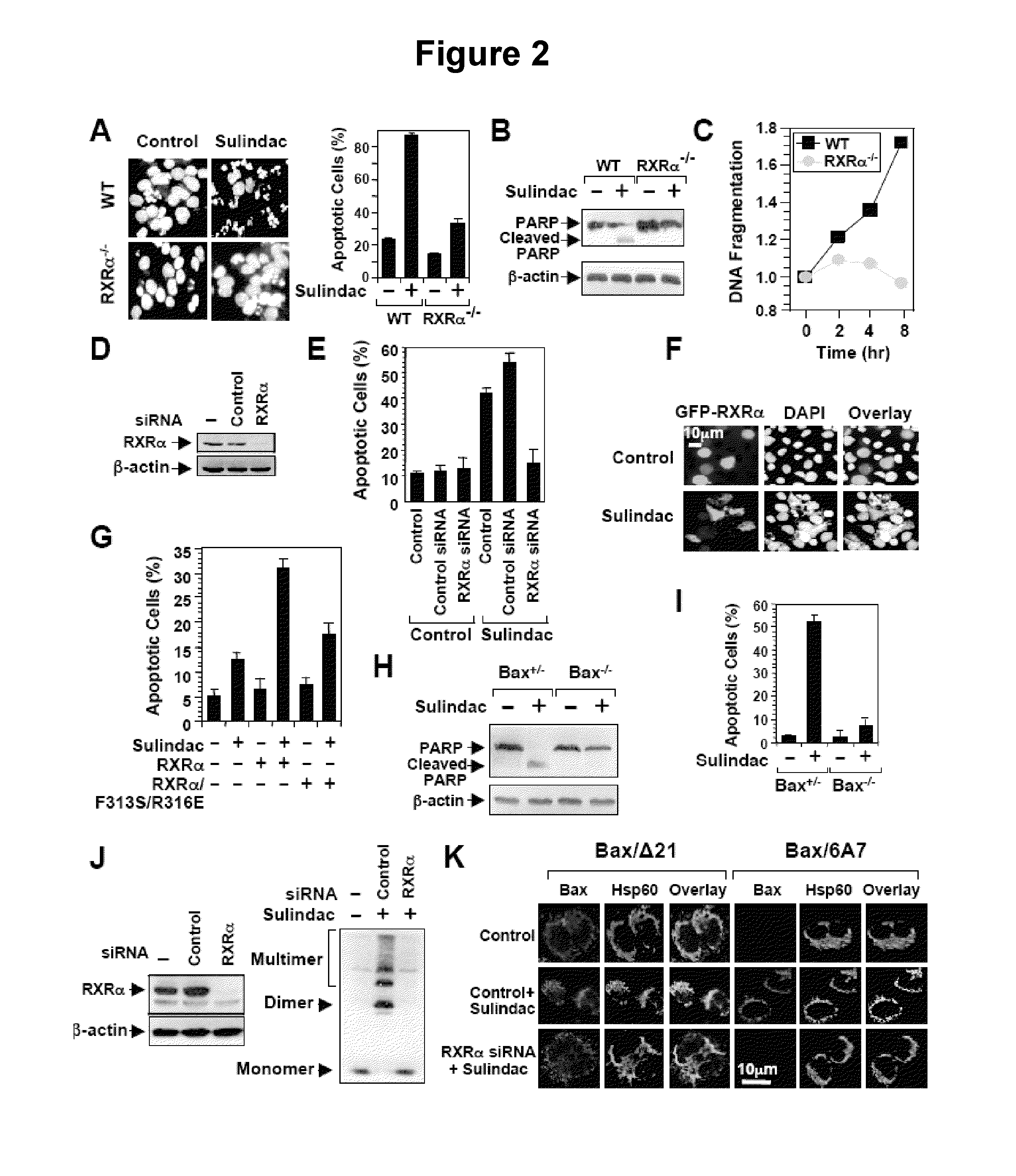
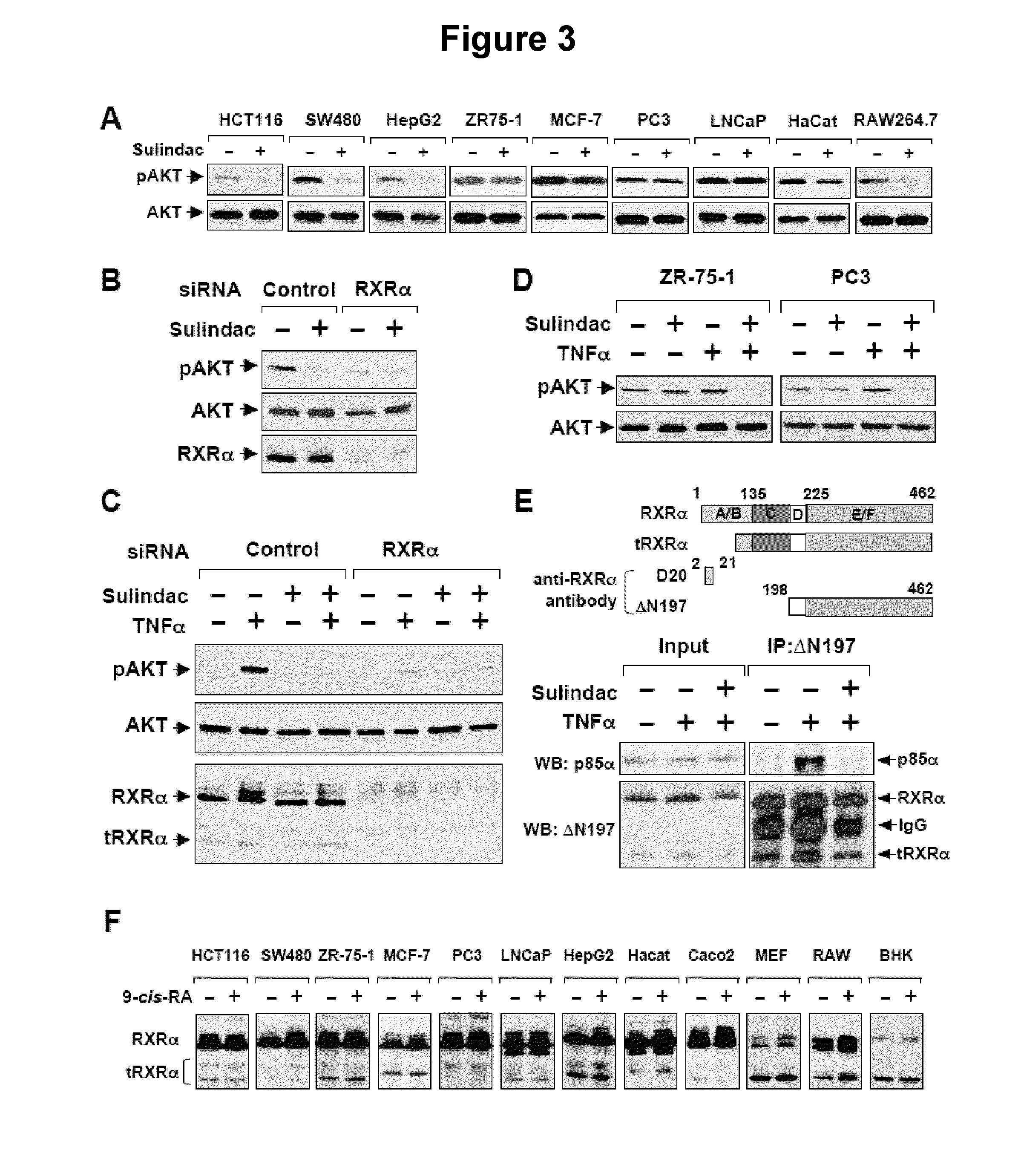
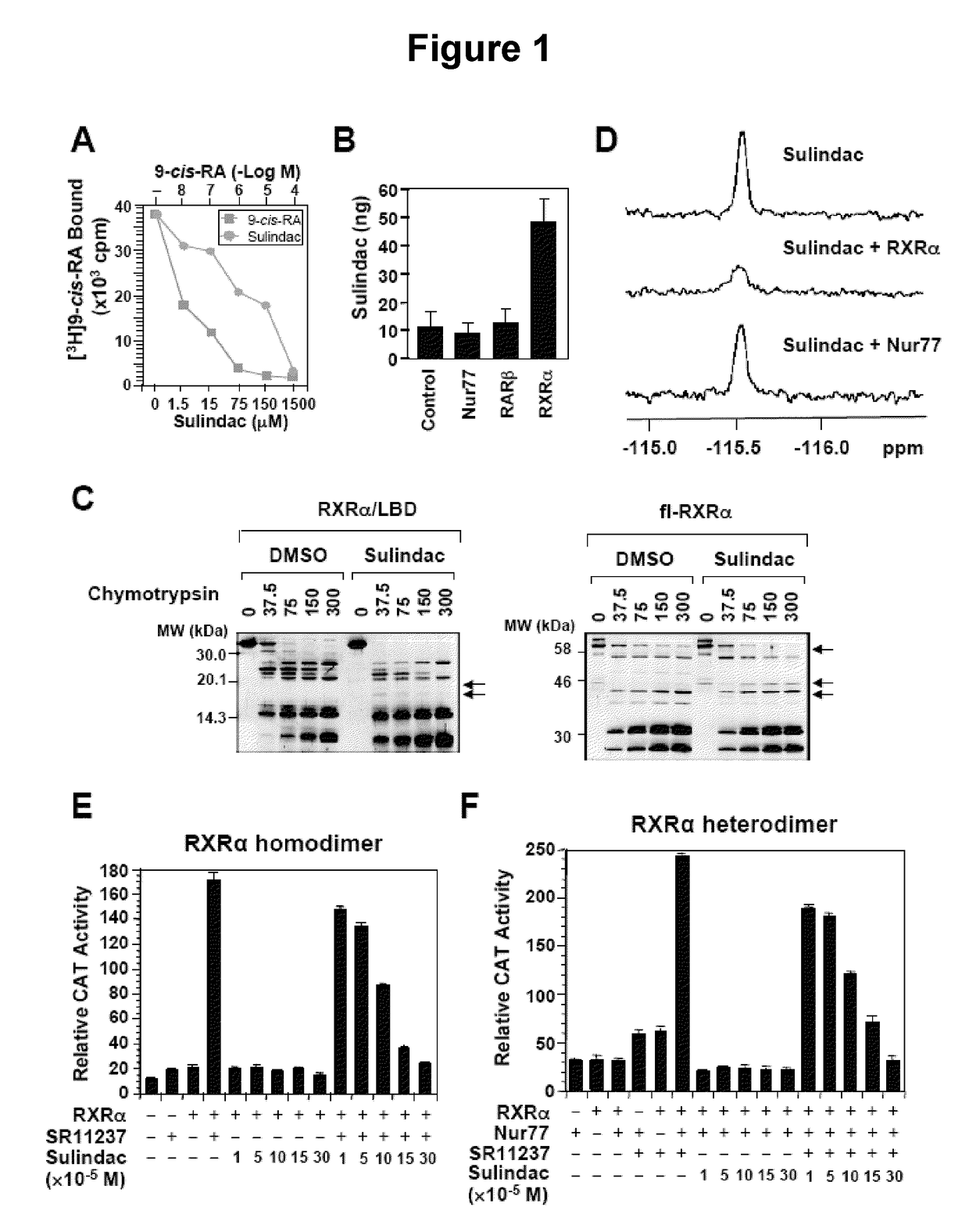
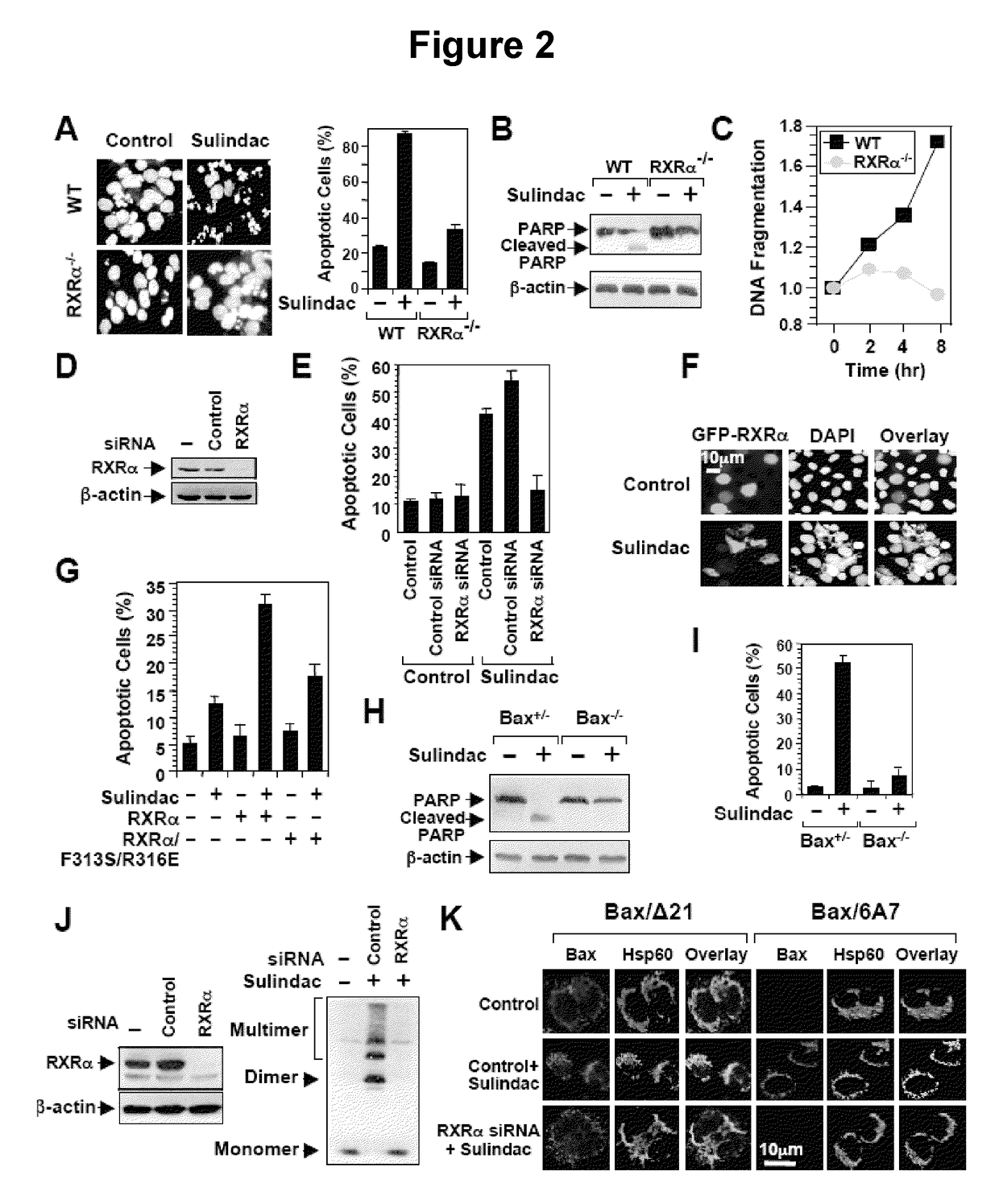
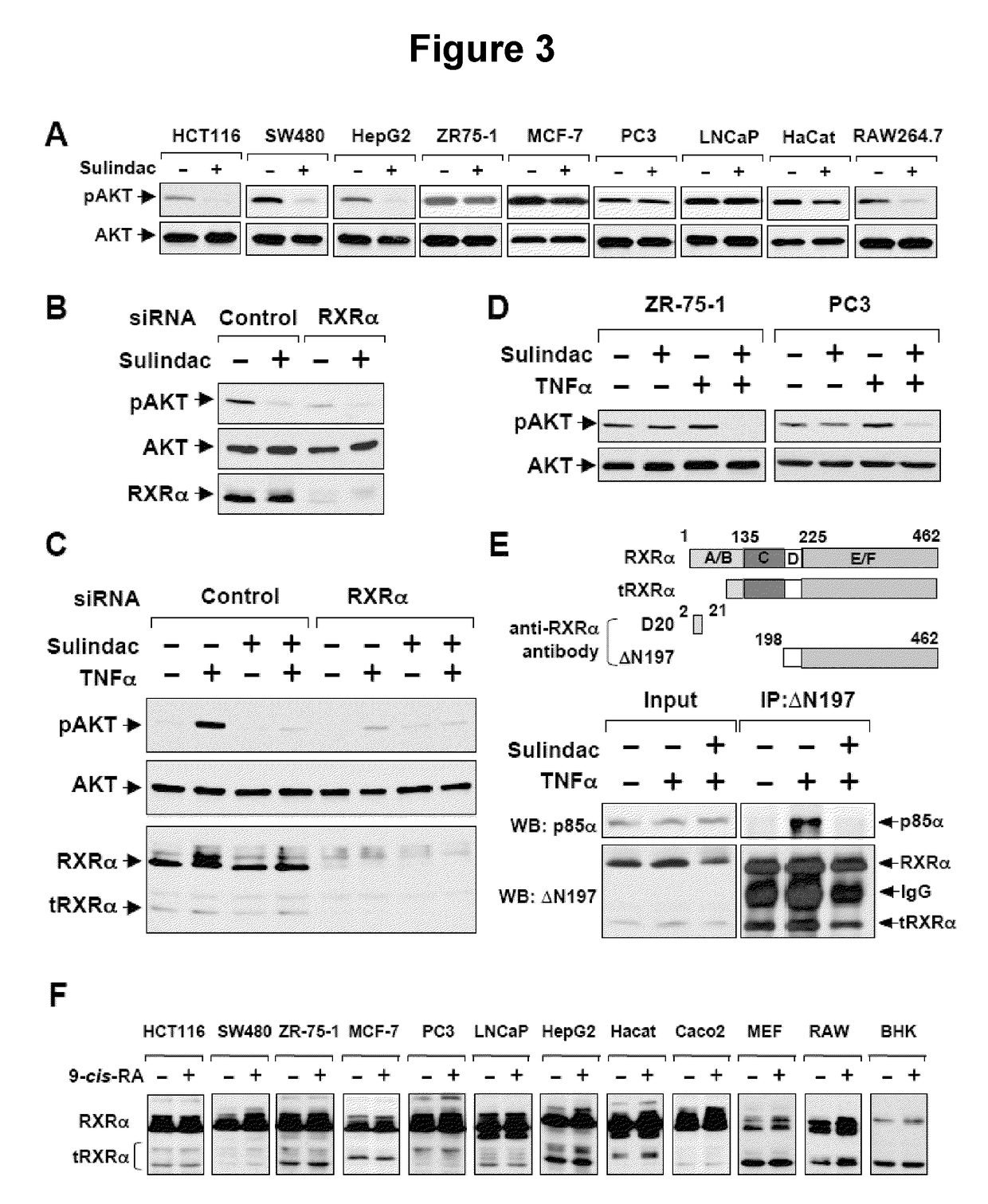
Methods and compositions related to a retinoid receptor-selective pathway
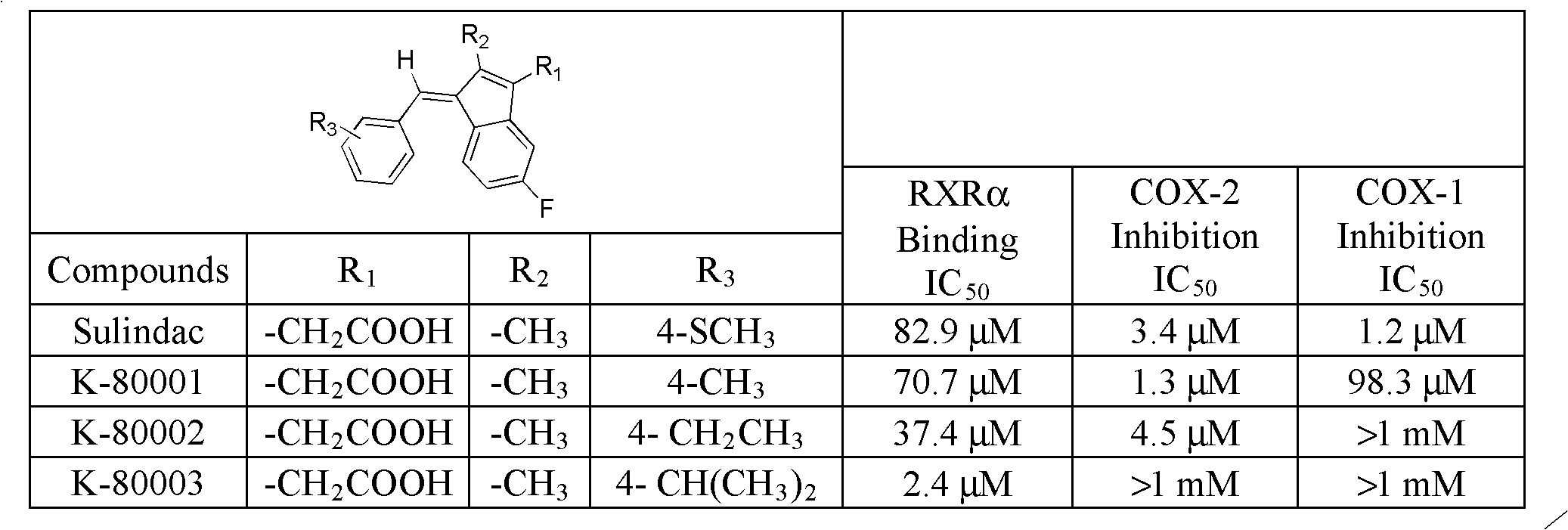
Provided herein are methods and compositions related to a retinoid receptor-selective pathway. As described herein, this pathway can be targeted to manipulate a tumor microenvironment. For example, the methods and compositions described herein can be used to induce apoptosis in a cancer cell. Further, the compositions described herein, including Sulindac and analogs thereof, can be used to target this pathway for the treatment or prevention of cancer in human patients.
Owner:XIAMEN UNIV +1
Process for preparing sulindac
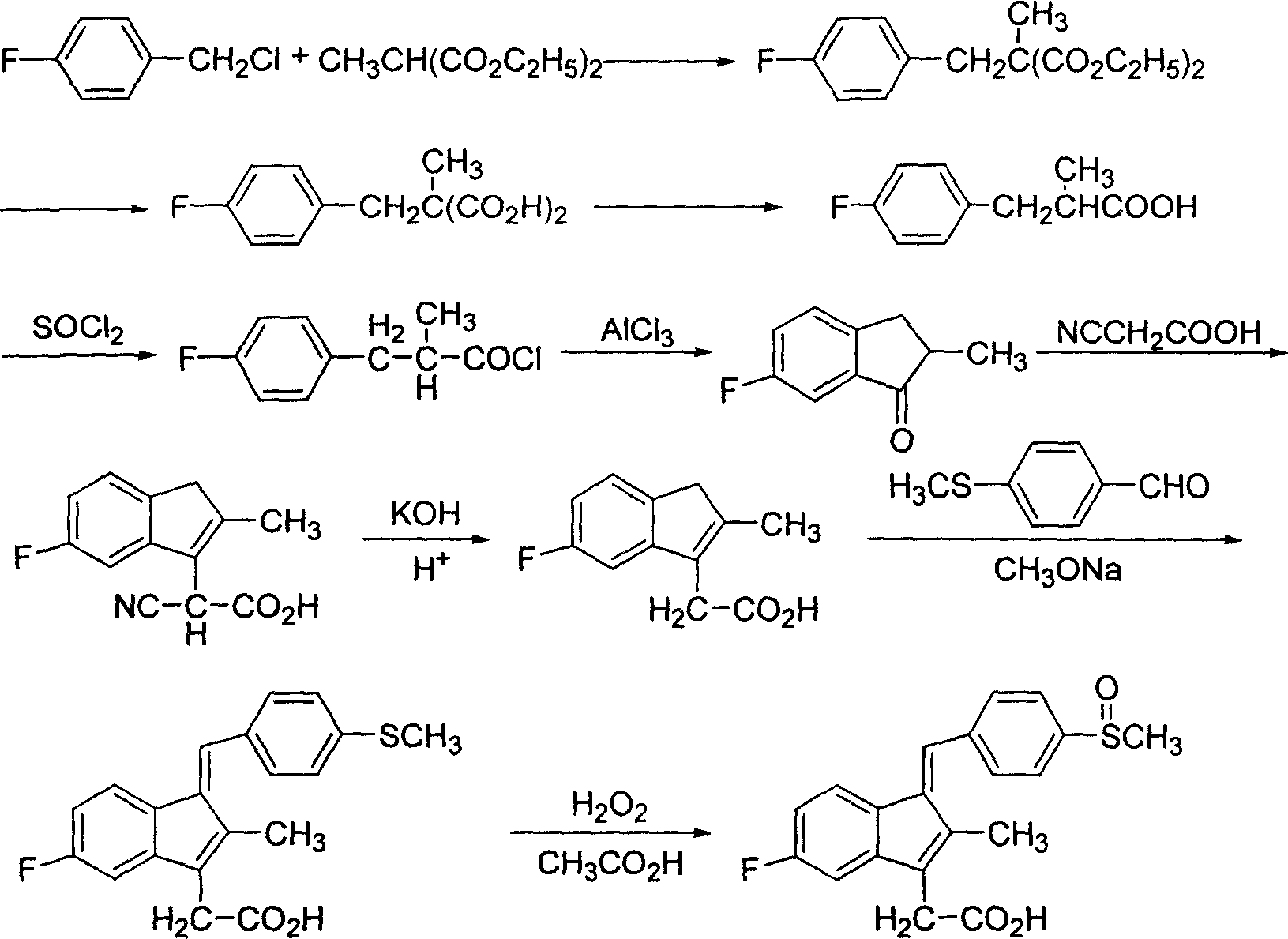
InactiveCN1699335AReduce productionHigh yieldOrganic chemistryOrganic compound preparationMethyl malonic acidCyanoacetic acid
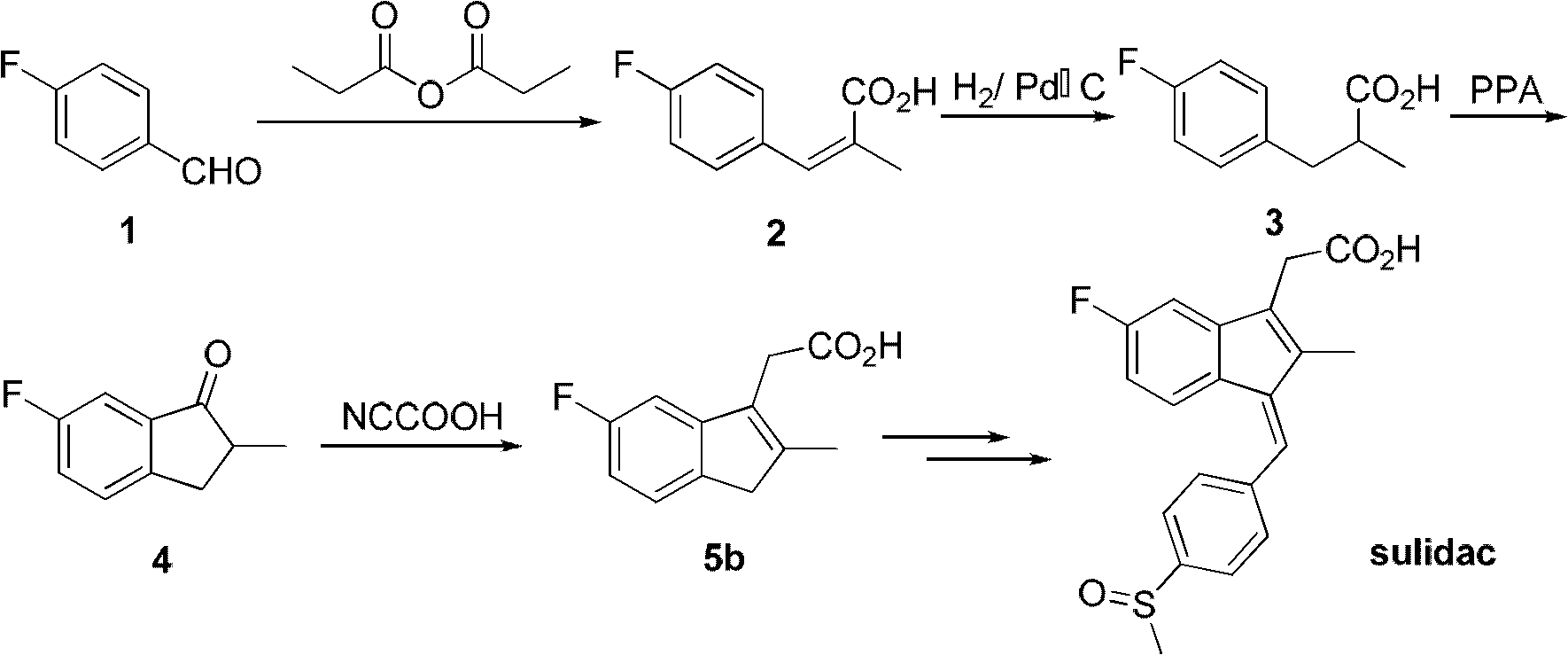
The invention discloses a process for preparing sulindac comprising the steps of, condensating 4-fluorobenzyl chloride and diethyl methylmalonate at the presence of organic base, obtaining 2-(4-fluorobenzyl)-2-diethyl methylmalonate, hydrolyzing in aqueous alkali to obtain 2-(4-fluorobenzyl)-2-methyl malonic acid, carrying out decarboxylation directly under high temperature to obtain 3-(4-fluorophenyl)-2-methylpropanoic acid, chlorinating with sulfoxide acyl chloride, then using aluminium trichloride or waterless zinc chloride as catalyst, carrying out F-C acylation to obtain 6-fluoro-2-methylindanone, condensing with cyanocetic acid, hydrolyzing to obtain 5-fluoro-methyl-3-indenes acetic acid, then condensing with p-methylthiobenzaldehyde to obtain 5-fluoro-2-methyl-1-(4-methylthiobenzal)-3-indenes acetic acid, finally using organic acid as solvent, peracid or hydroperoxide as oxidation agent to obtain the sulindac.
Owner:ZHEJIANG UNIV
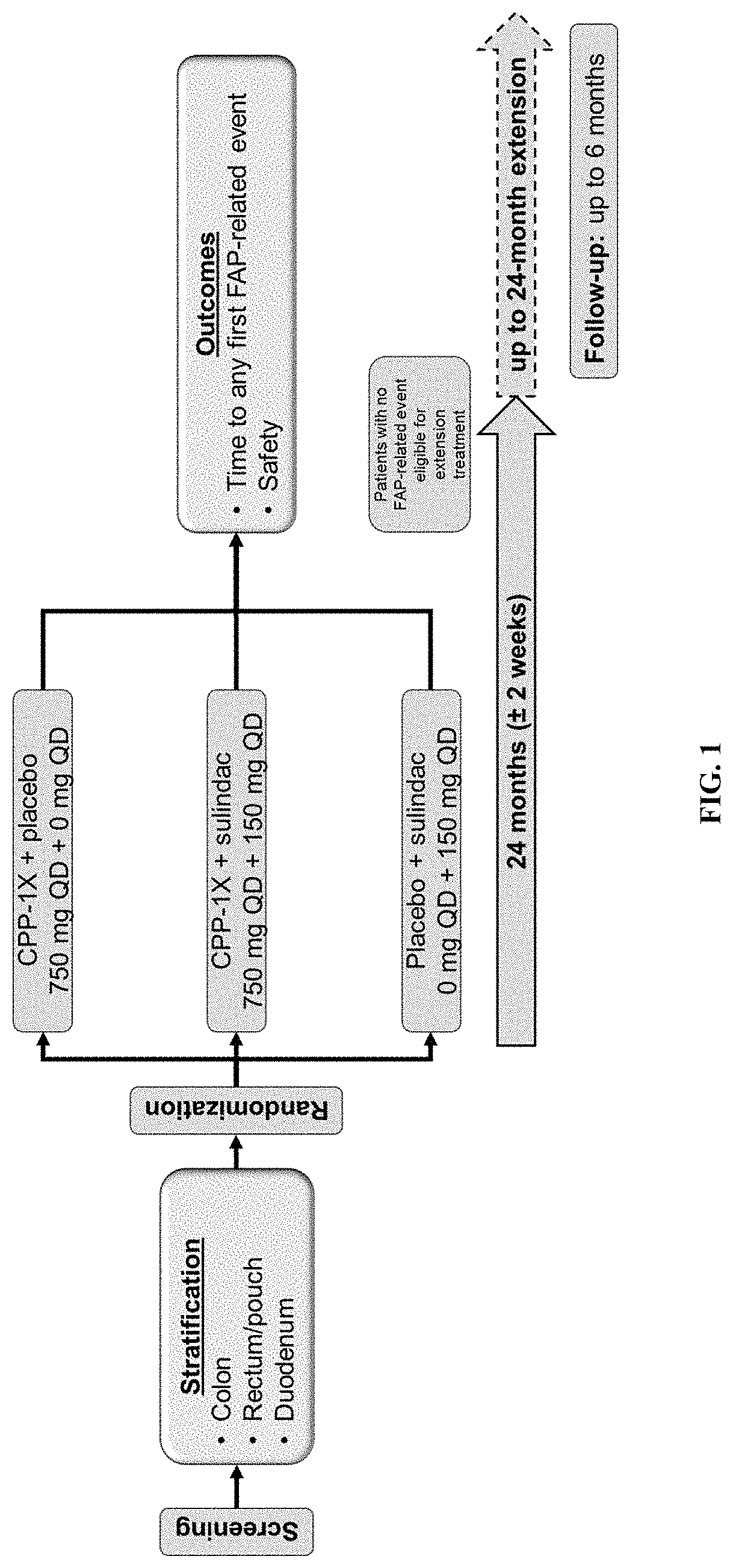
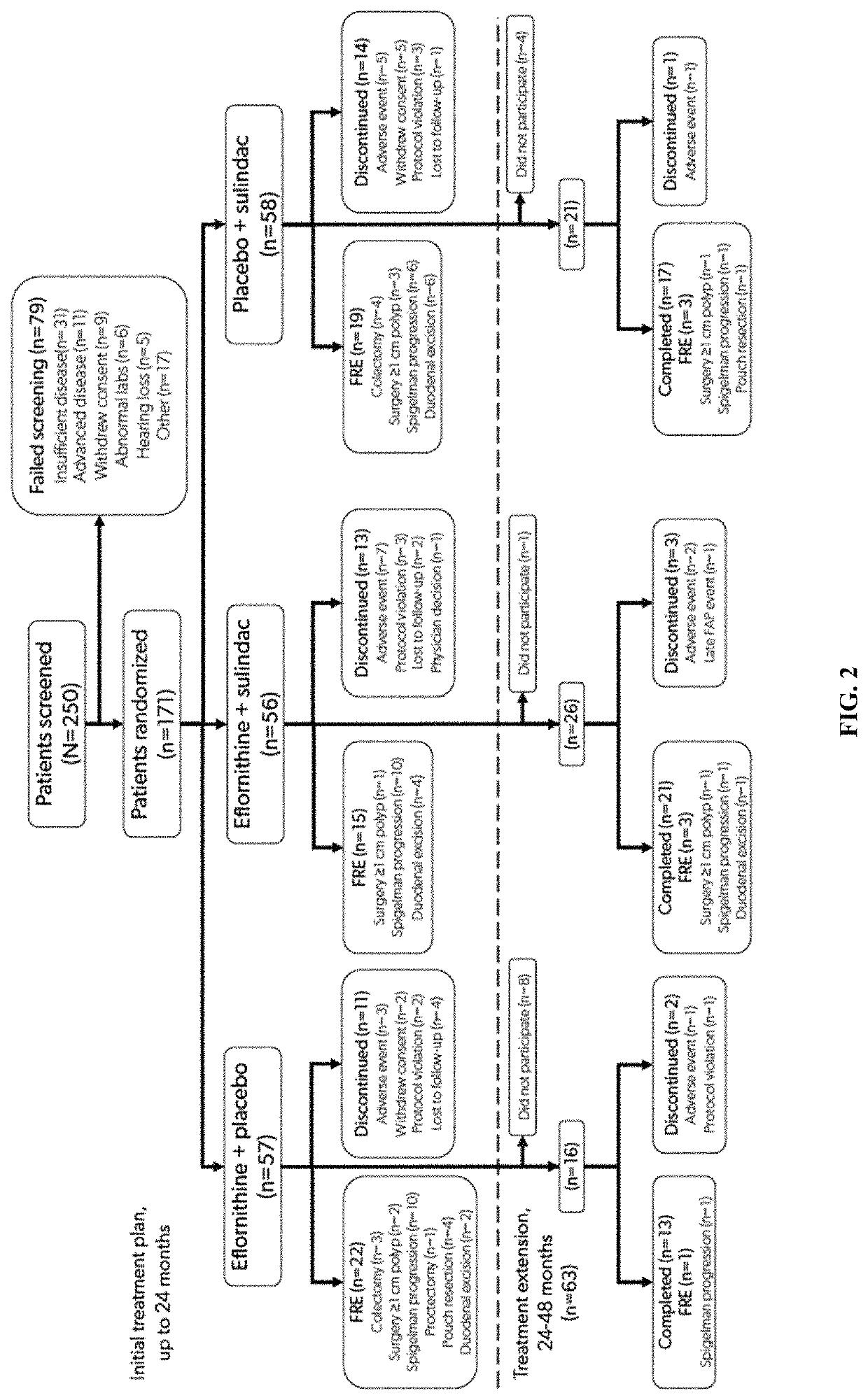
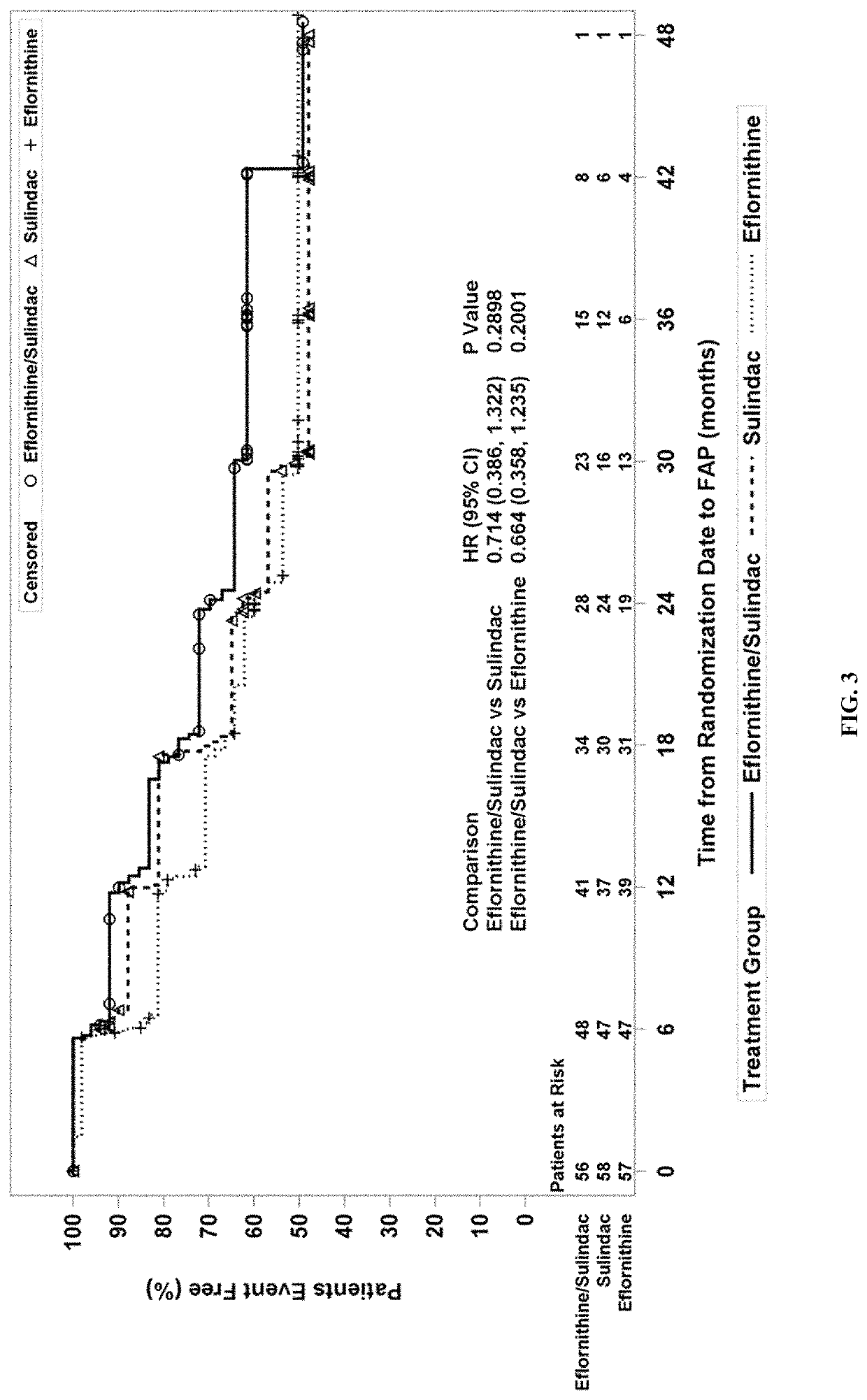
Methods for treating familial adenomatous polyposis
ActiveUS10945981B2Avoid delayInhibition formationOrganic active ingredientsDigestive systemFAP - Familial adenomatous polyposisLower Gastrointestinal Tract
Provided are methods for preventing or delaying the need for surgical intervention in a patient having familial adenomatous polyposis (FAP) and an at least partially intact lower gastrointestinal tract. Also provided are methods for preventing or delaying the formation of neoplasia and / or cancer in a patient having FAP. The methods comprise administering an effective amount of a pharmaceutical therapy that comprises eflornithine and sulindac to a patient having FAP and an intact lower gastrointestinal tract.
Owner:CANCER PREVENTION PHARMA
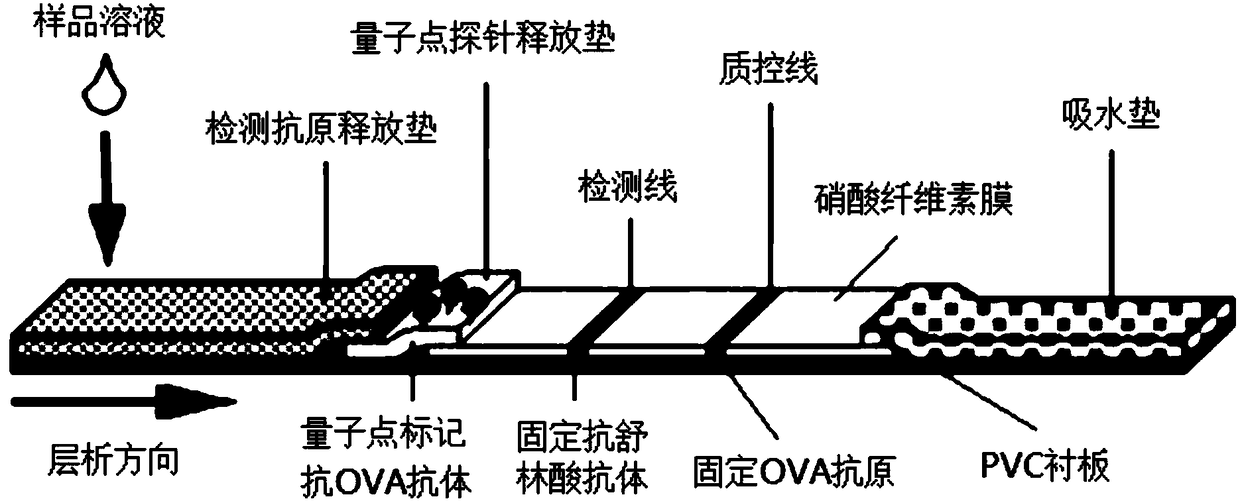
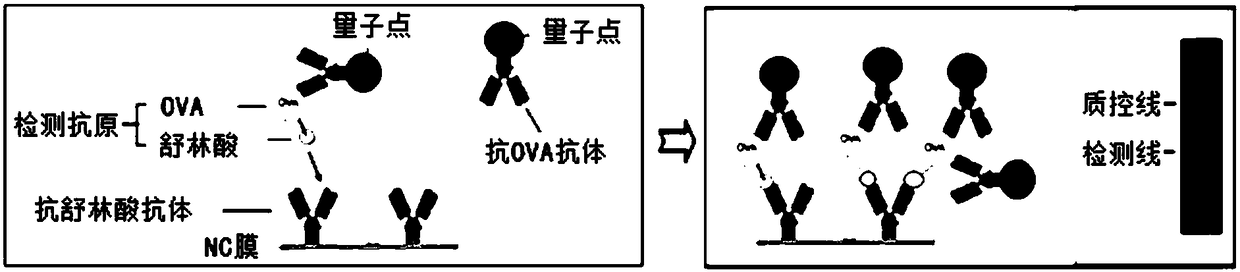
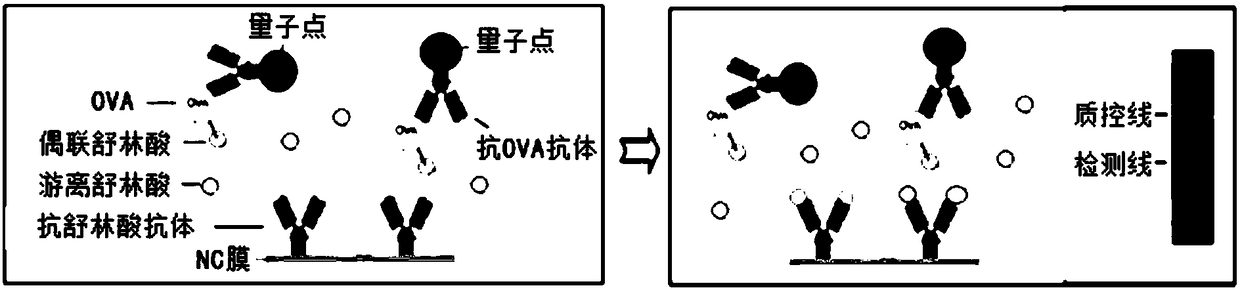
Quantum dot immunochromatography detection card and method for detecting sulindac through ternary system competitive immunoassay process
PendingCN108535475ANarrow and symmetrical emission peakImprove stabilityBiological testingAntigen releaseSignal on
Owner:SHAOGUAN COLLEGE
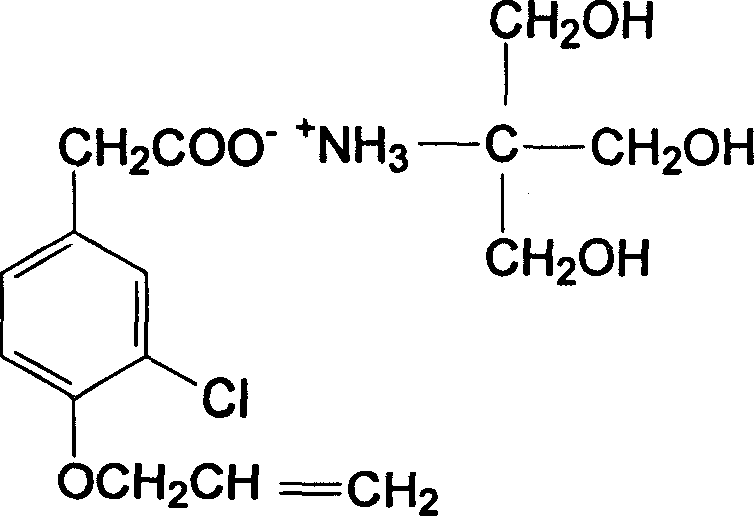
Carboxyl contained NSAIDS (nonsteroidal anti-inflammatory drugs) salt
Disclosed are non-steroidal analgesic and analgesic anti-inflammatory agents containing carboxyl including sodium, calcium, zinc, magnesium, N-n-octylgucamine, Arginine, Lycine or Trometamol salts of Loxoprofen, Ketoprofen, Pranoprofen, Tiaprofenic acid, Butibufen, Omolofen, epoxy indene acid, Lobuprofen, Clofenamic acid, Clonixin, Fenoprofen, Benorilate, Flurbiprofen, Alminoprofen, Bucloxic acid, Sulindac, Zidometacin, Acemetacin, Ketorolac, Risedronic acid, Sulindac, Lonaprofen, aspirin, Florfenicol, tiaprofenic acid, overall evaluation shows that trometamol salts are the best choice in terms of physicochemical properties, solvability, stability, local irritation, blood vessel irritation, and bioavailability for oral administration.
Owner:陈文展
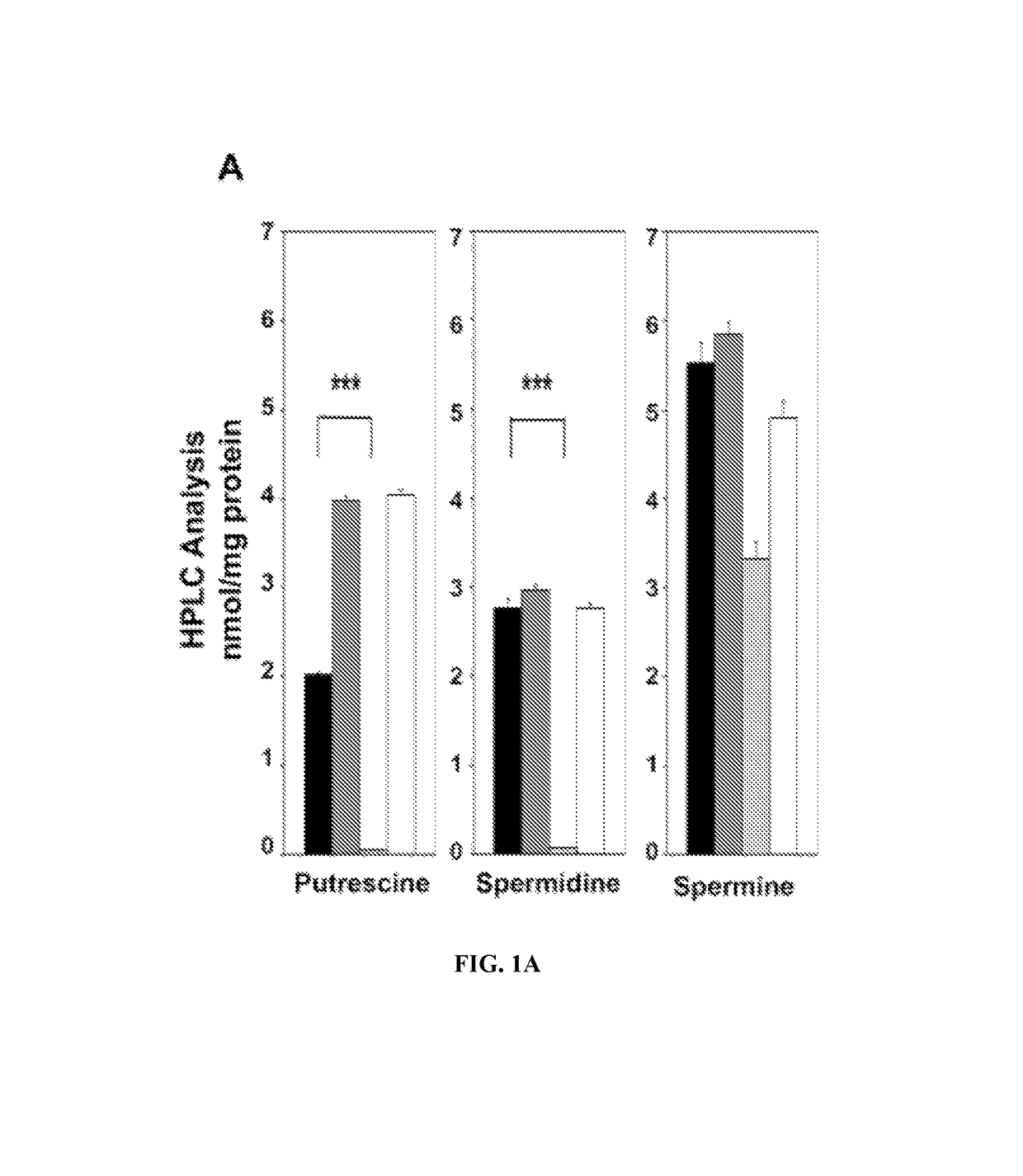
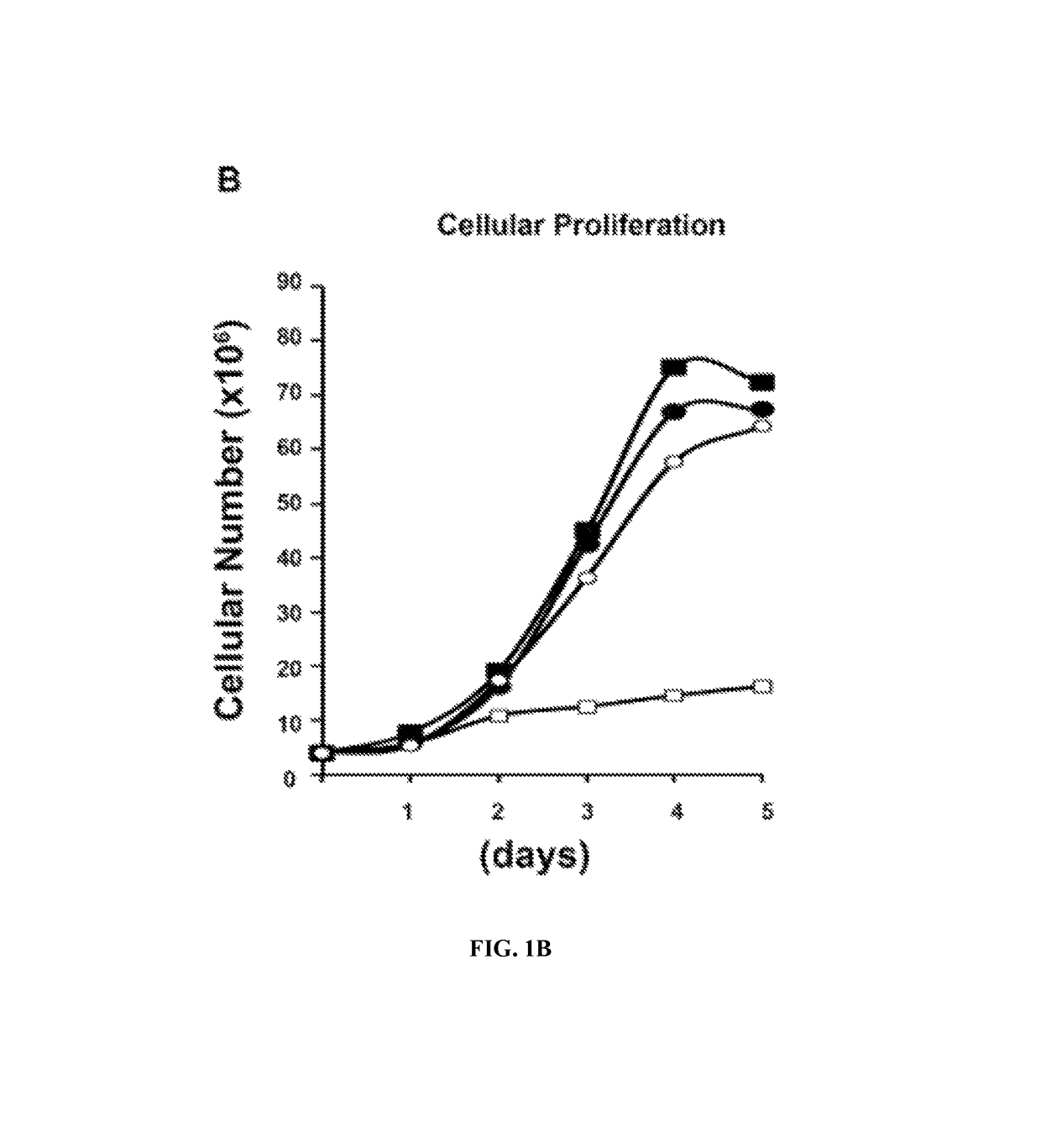
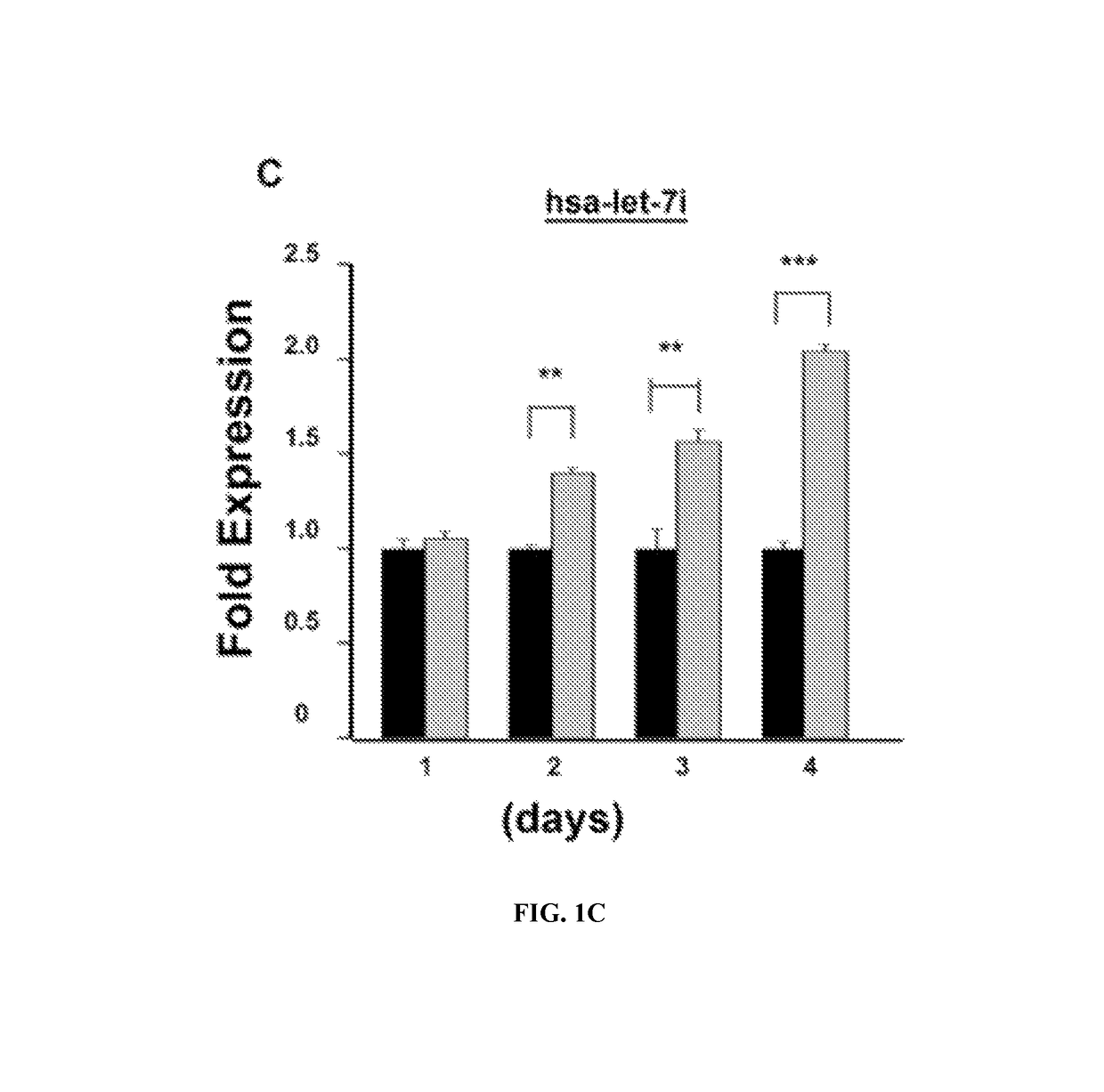
Predictive markers for polyamine inhibitor cancer therapies
The present disclosure relates to therapeutic methods and medical uses comprising the identification and use of cancer marker surrogates for increased polyamine expression. These markers may be used to identify patients who may be treated for diseases and disorders that are susceptible to polyamine synthesis inhibitors, and they can also be used to monitor therapeutic responses when such agents are used. More specifically, reduced levels of let-7 miRNA and elevated levels of LIN28 and HMGA2 proteins were found to correlate with elevated levels of polyamines and may be used for predicting the efficacy of cancer therapy using an ornithine decarboxylase (ODC1) inhibitor such as eflornithine (DFMO), suitably in combination with an NSAID such as sulindac.
Owner:THE ARIZONA BOARD OF REGENTS ON BEHALF OF THE UNIV OF ARIZONA
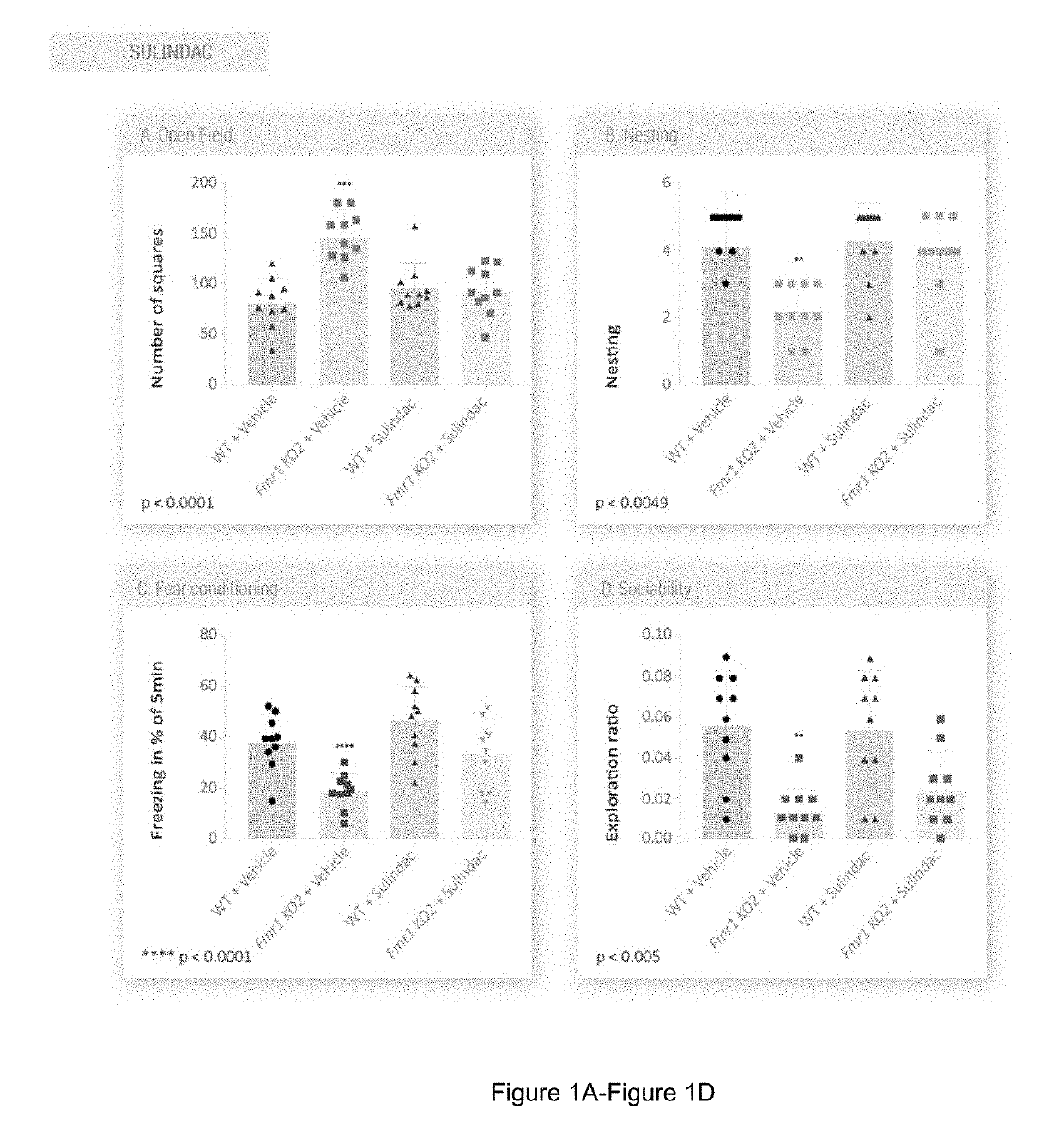
Kit, composition, and combination therapy for fragile x syndrome
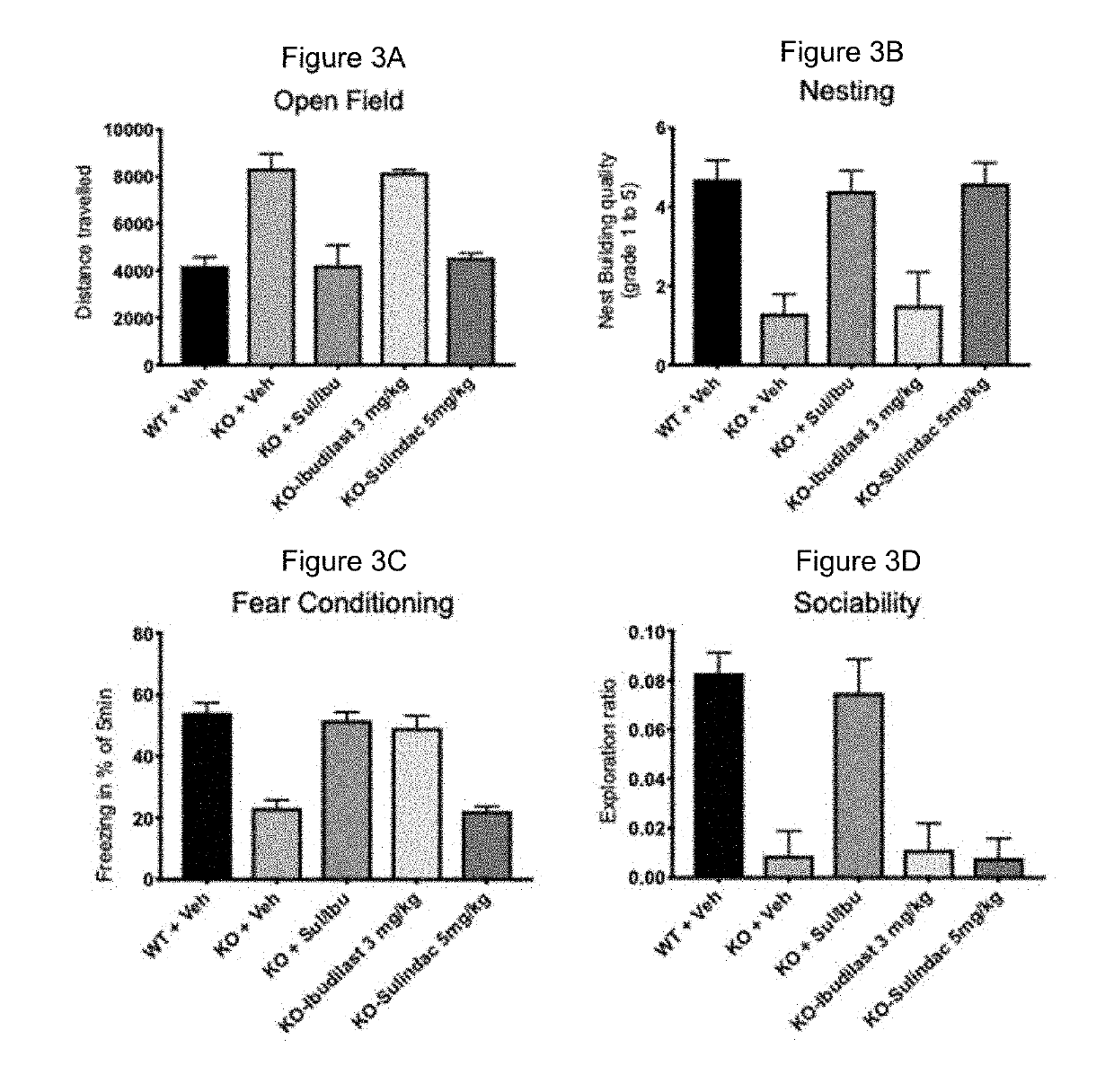
ActiveUS20190314309A1Combination is synergisticFast absorptionNervous disorderHydroxy compound active ingredientsFragile X chromosomeCombined Modality Therapy
Owner:HEALX
Treatment of fragile x syndrome
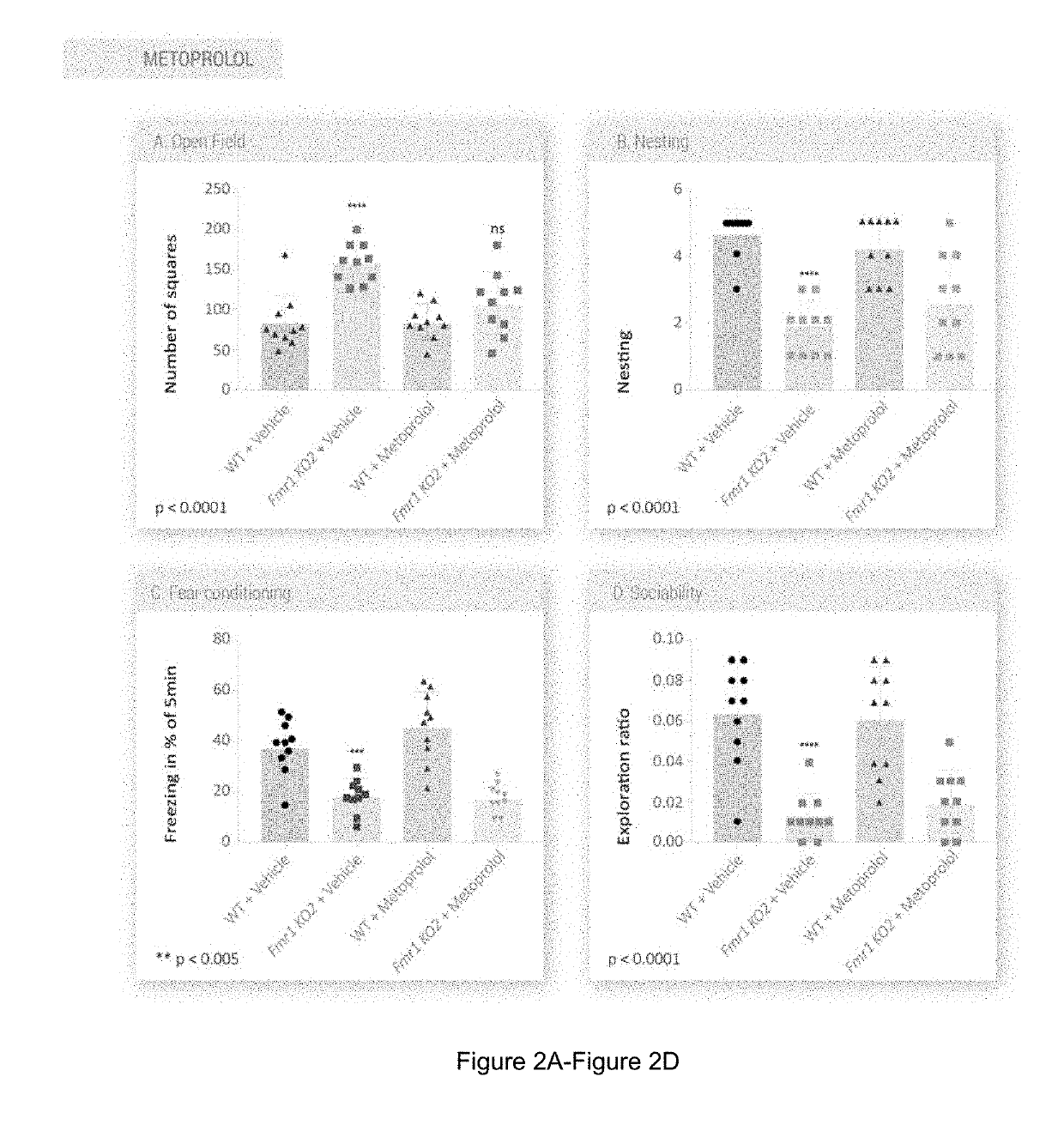
ActiveUS20190314308A1Improved the Fmr1 KO2 mouse phenotypeUseful in therapyOrganic active ingredientsNervous disorderFragile X chromosomePediatrics
Owner:HEALX
Treatment or prevention of cancer and precancerous disorders
The NSAID, sulindac and / or its metabolites and derivatives, in combination with hydrogen peroxide or another oxidizing agent, such as arsenic trioxide that generates reactive oxygen species (ROS), significantly enhances the killing of cancer cells. This effect occurs at concentrations of each compound that individually have little or no activity directed against cancer cells. A skin cream has been developed and used to treat skin cancer and precancerous skin growths that effectively removes the lesions with no effect on surrounding normal skin.
Owner:CHS PHARMA
Treatment or prevention of cancer and precancerous disorders
The NSAID, sulindac and / or its metabolites and derivatives, in combination with an agent that generates or induces reactive oxygen species (ROS), significantly enhances the killing of abnormal cells but does not affect normal cells. This effect occurs at concentrations of each compound that individually have little or no activity directed against the abnormal cells.
Owner:CHS PHARMA
Protection of normal cells
InactiveCN102112124ASmall doseReduce the incidence of adverse effectsOrganic active ingredientsNervous disorderDiseaseMetabolite
Owner:CHS PHARMA
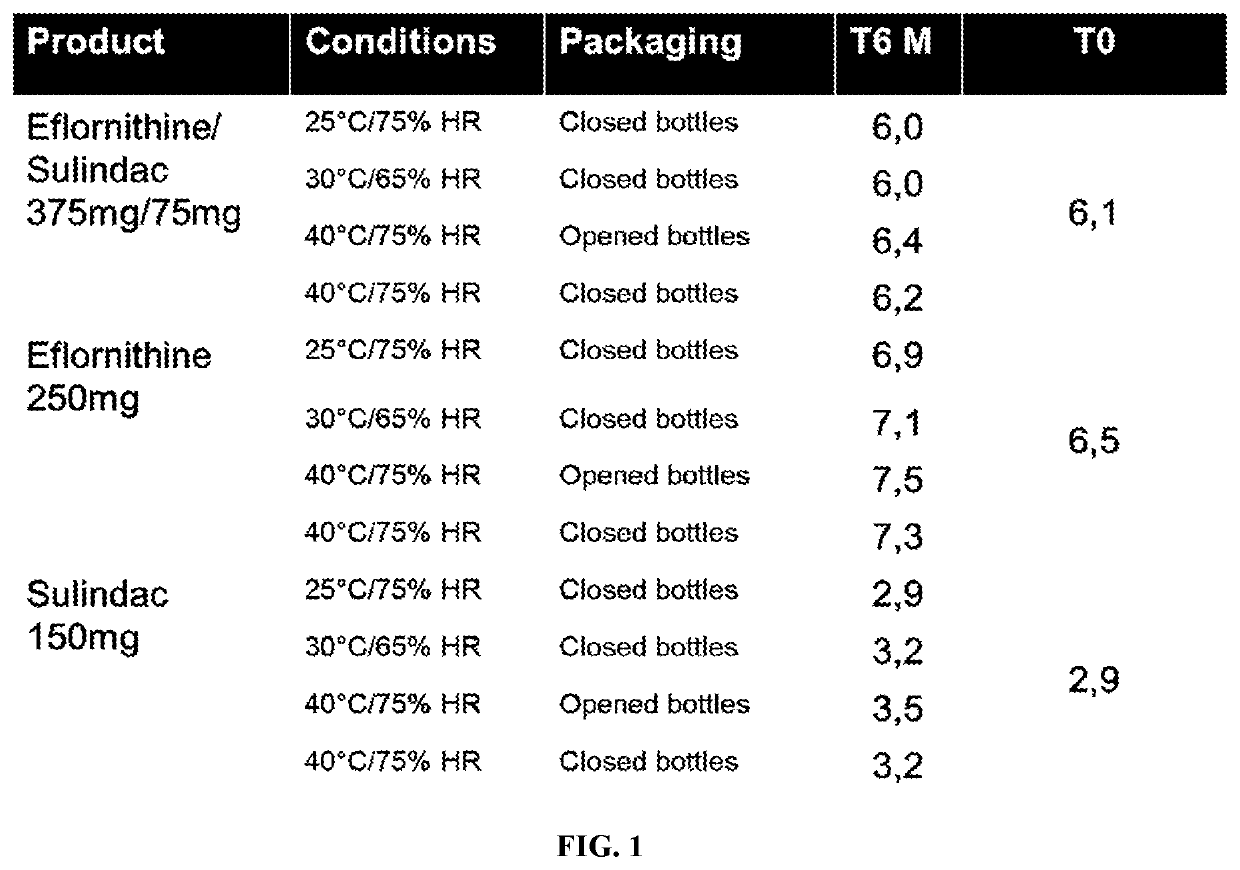
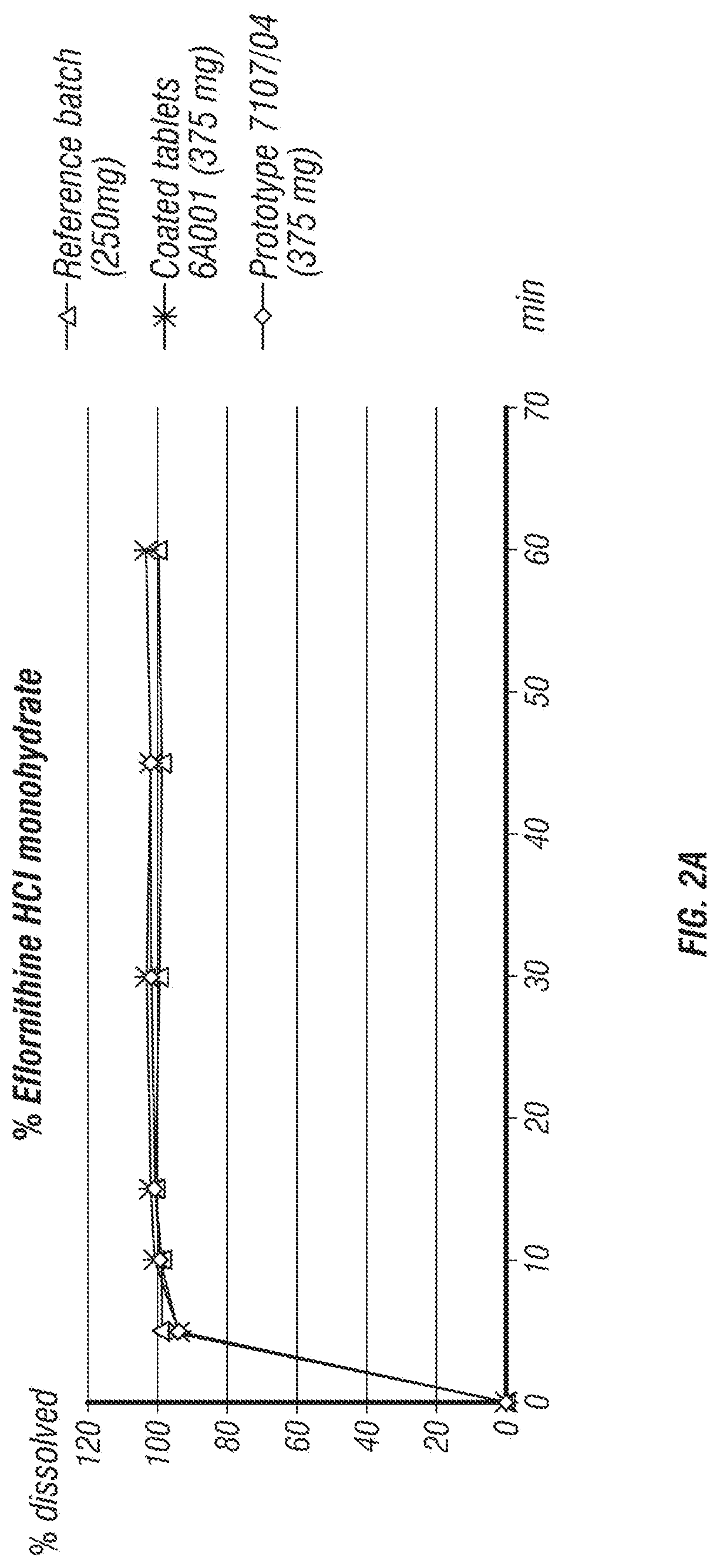
Eflornithine and sulindac, fixed dose combination formulation
ActiveUS20200009098A1Prevents tablet cappingOrganic active ingredientsNervous disorderPharmaceutical medicineMedicinal chemistry
Owner:CANCER PREVENTION PHARMA
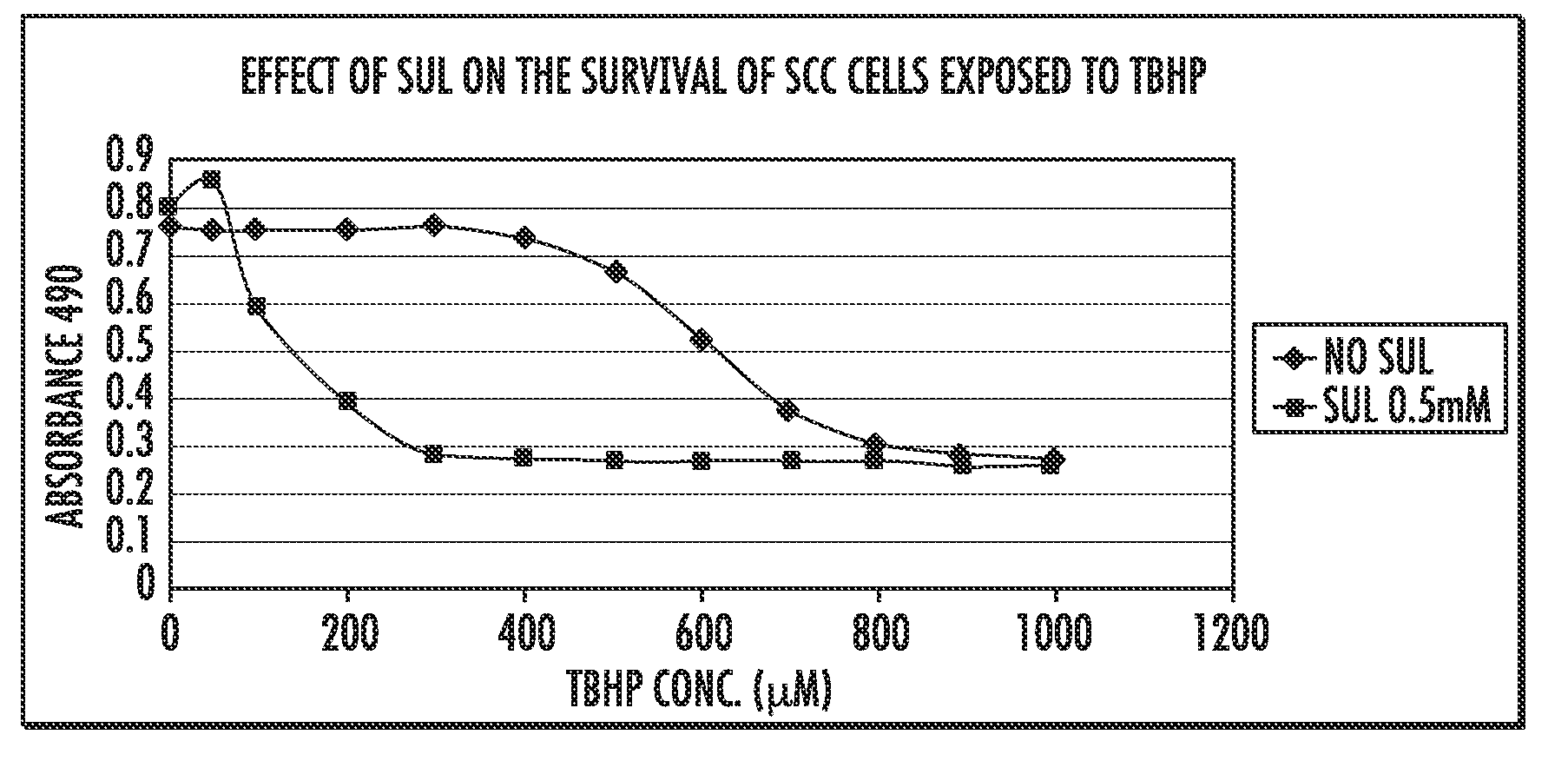
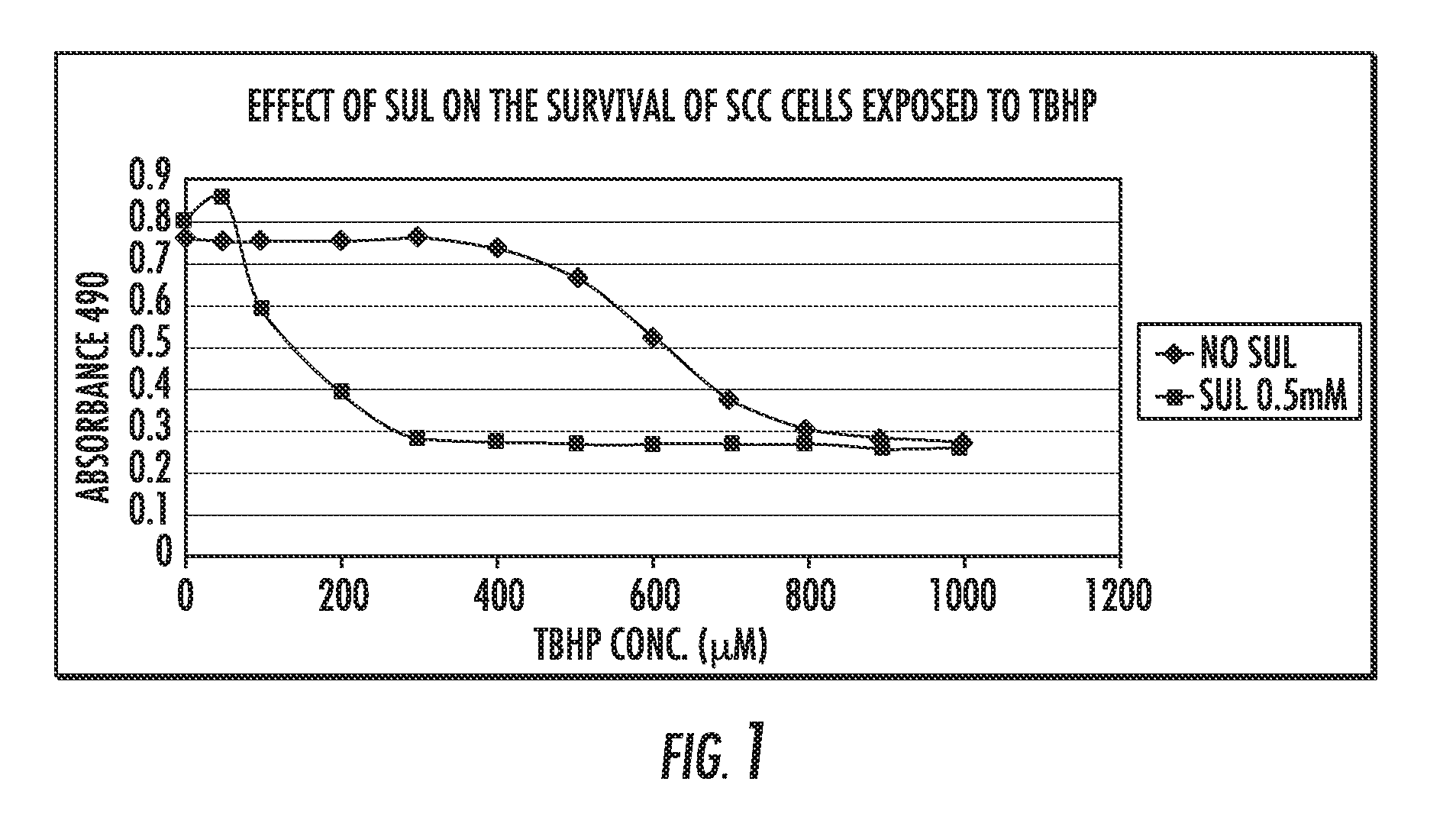
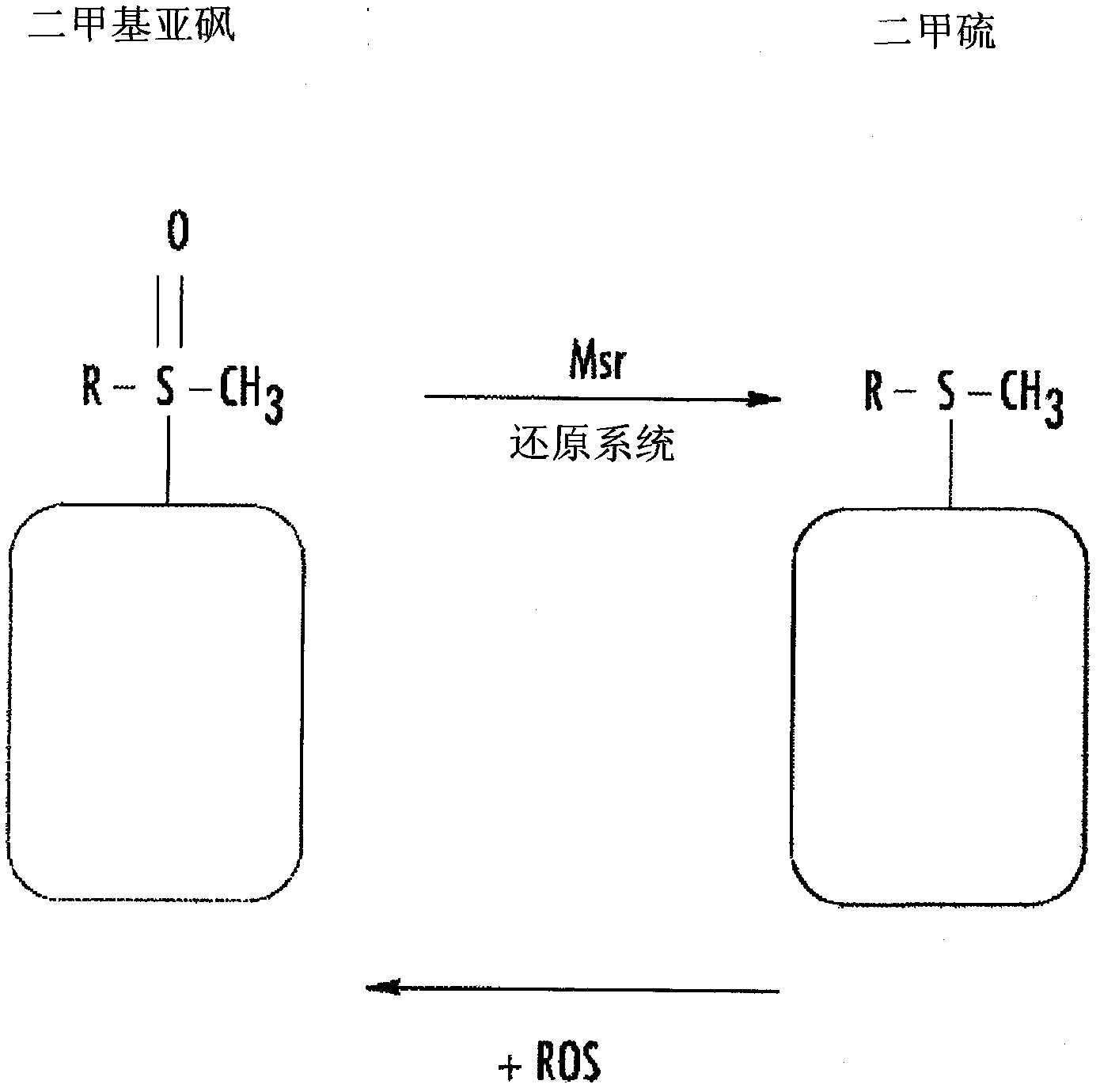
Derivatives of sulindac can protect normal cells against oxidative damage
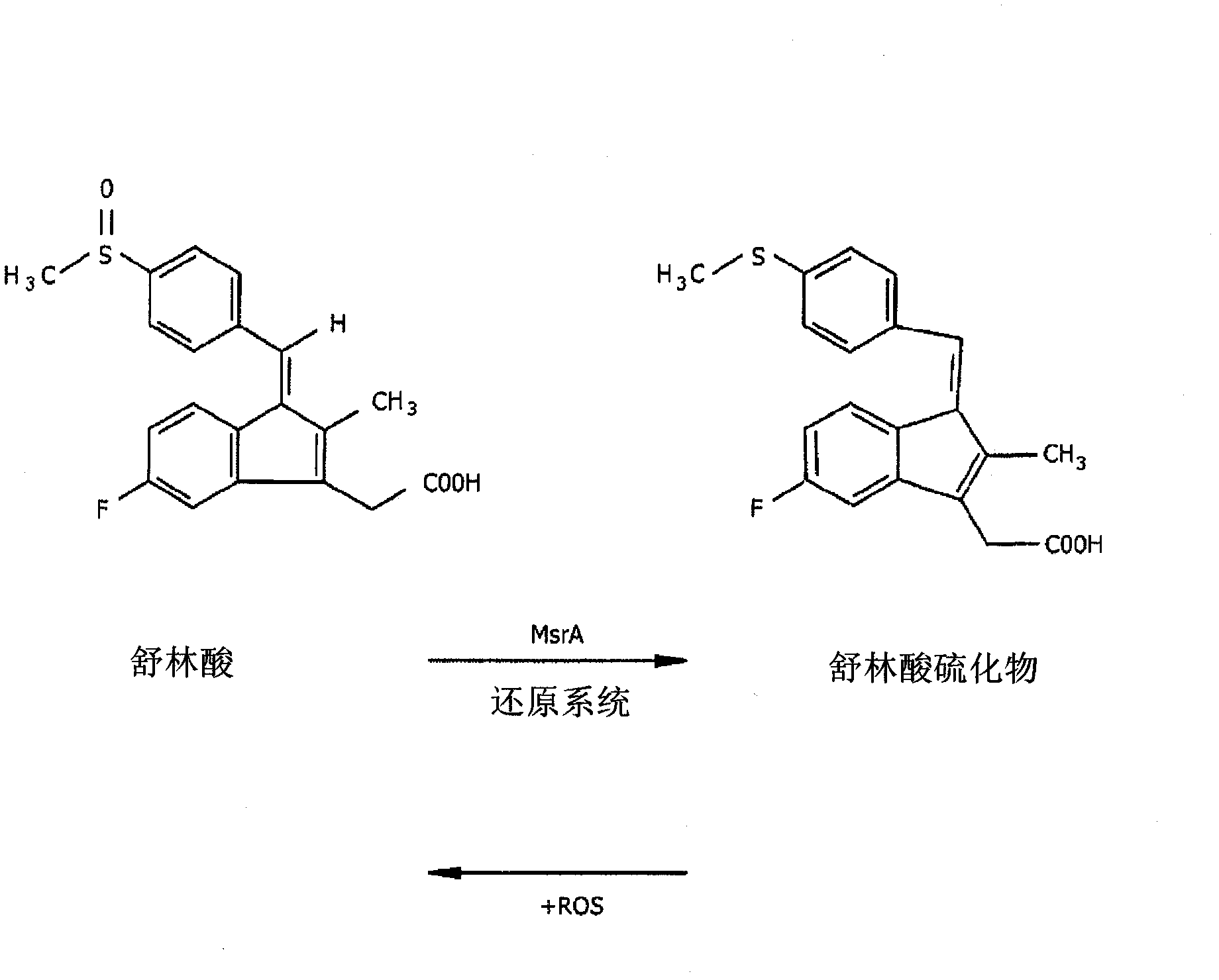
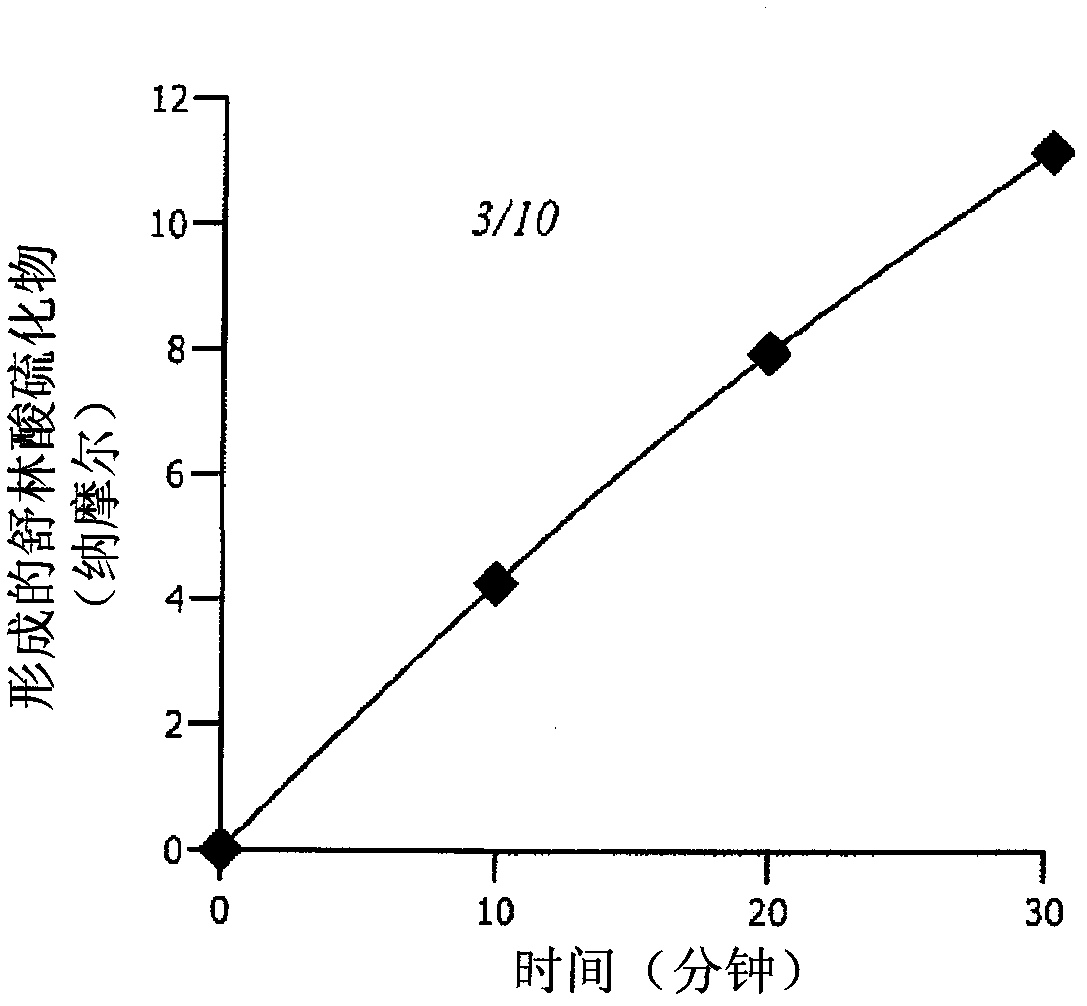
ActiveUS11186534B2Preventing or reducing the extent of tissue damage of an ischemic eventOrganic active ingredientsSenses disorderMedicineChemical compound
The disclosure provides chemical compounds possessing therapeutic and / or protective properties against oxidative damage. Methods of making such therapeutic and / or protective compounds and associated compositions are also provided, as are methods for their use, which include protecting cells from oxidative damage and / or inhibiting production of ROS in a cell or subject, as well as preventing or reducing the extent of tissue damage caused by an ischemic event in a subject at elevated risk of such an event.
Owner:UNIV OF SOUTH ALABAMA
Methods and compositions related to a retinoid receptor-selective pathway
Provided herein are methods and compositions related to a retinoid receptor-selective pathway. As described herein, this pathway can be targeted to manipulate a tumor microenvironment. For example, the methods and compositions described herein can be used to induce apoptosis in a cancer cell. Further, the compositions described herein, including Sulindac and analogs thereof, can be used to target this pathway for the treatment or prevention of cancer in human patients.
Owner:XIAMEN UNIV +1