Patents
Literature
209 results about "Ursodeoxycholic acid" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
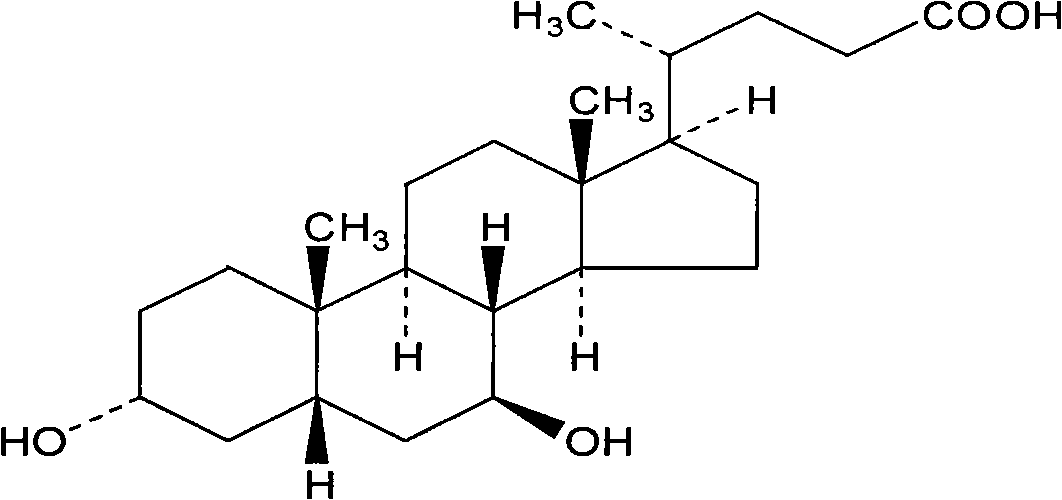
Ursodiol is used to dissolve certain types of gallstones, to prevent gallstones from forming in obese patients who are losing weight rapidly, and to treat a certain type of liver disease (primary biliary cirrhosis).
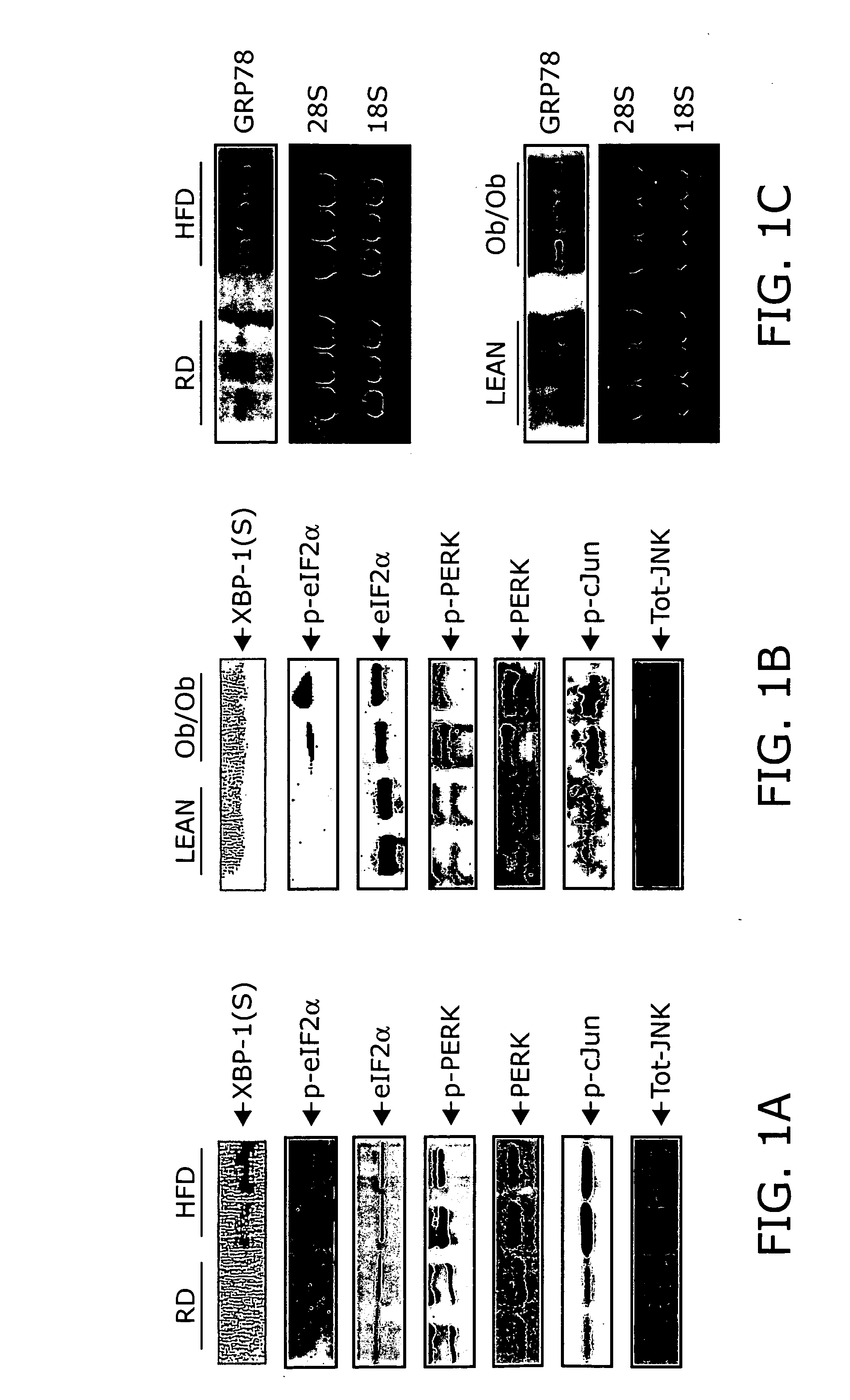
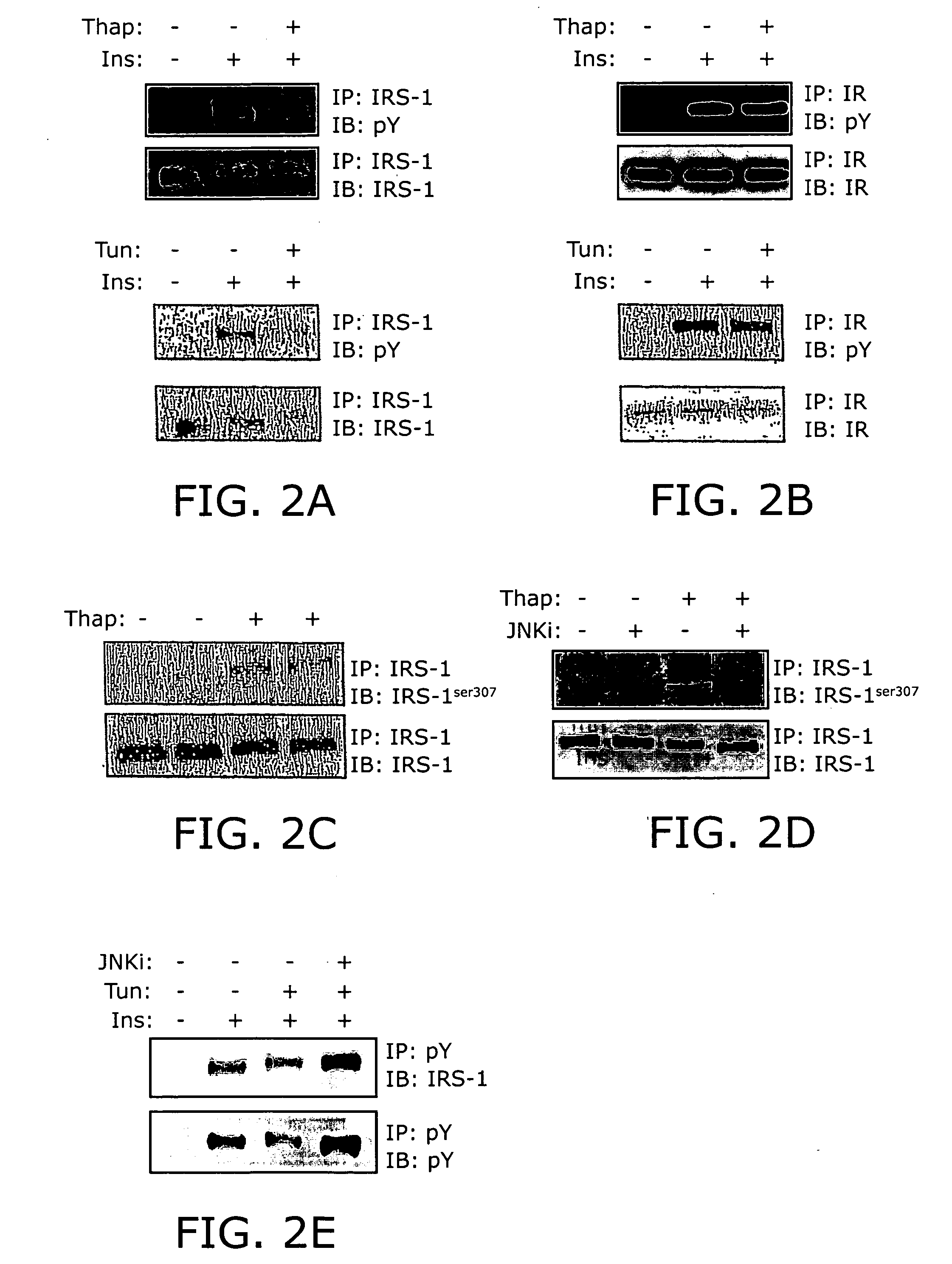
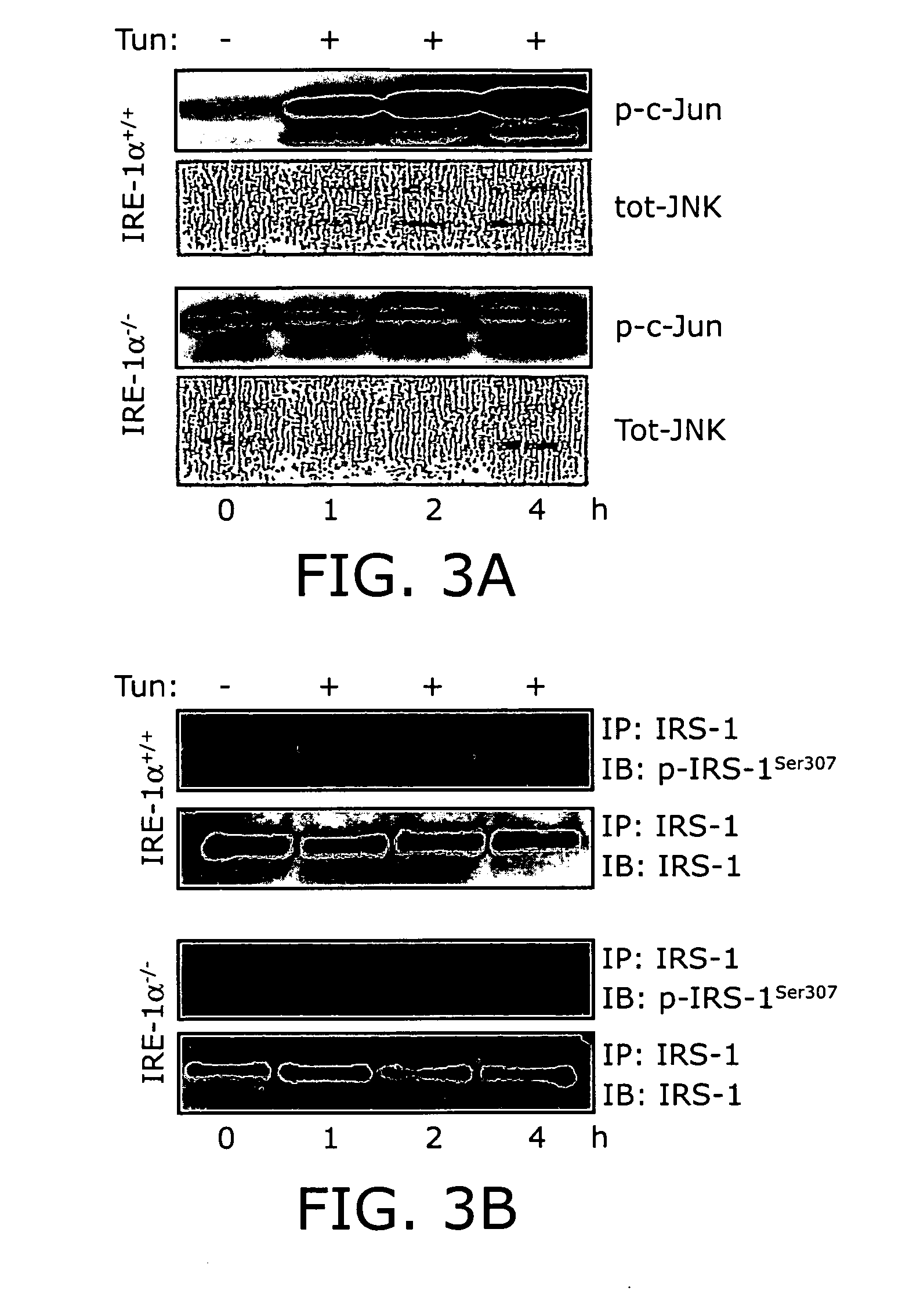
Reducing ER stress in the treatment of obesity and diabetes
InactiveUS20060073213A1Reduce ER stressIncrease insulin sensitivitySalicyclic acid active ingredientsBiocidePeripheral insulin resistanceTauroursodeoxycholic acid
Endoplasmic reticulum stress has been found to be associated with obesity. Therefore, agents that reduce or prevent ER stress may be used to treat diseases associated with obesity including peripheral insulin resistance, hypergylcemia, and type 2 diabetes. Two compounds which have been shown to reduce ER stress and to reduce blood glucose levels include 4-phenyl butyric acid (PBA), tauroursodeoxycholic acid (TUDCA), and trimethylamine N-oxide (TMAO). Other compounds useful in reducing ER stress are chemical chaperones such as trimethylamine N-oxide and glycerol. The present invention provides methods of treating a subject suffering from obesity, hyperglycemia, type 2 diabetes, or insulin resistance using ER stress reducers such as PBA, TUDCA, and TMAO. Methods of screening for ER stress reducers by identifying agents that reduce levels of ER stress markers in ER stressed cells are also provided. These agents may find use in methods and pharmaceutical compositions for treating obesity-associated diseases.
Owner:PRESIDENT & FELLOWS OF HARVARD COLLEGE
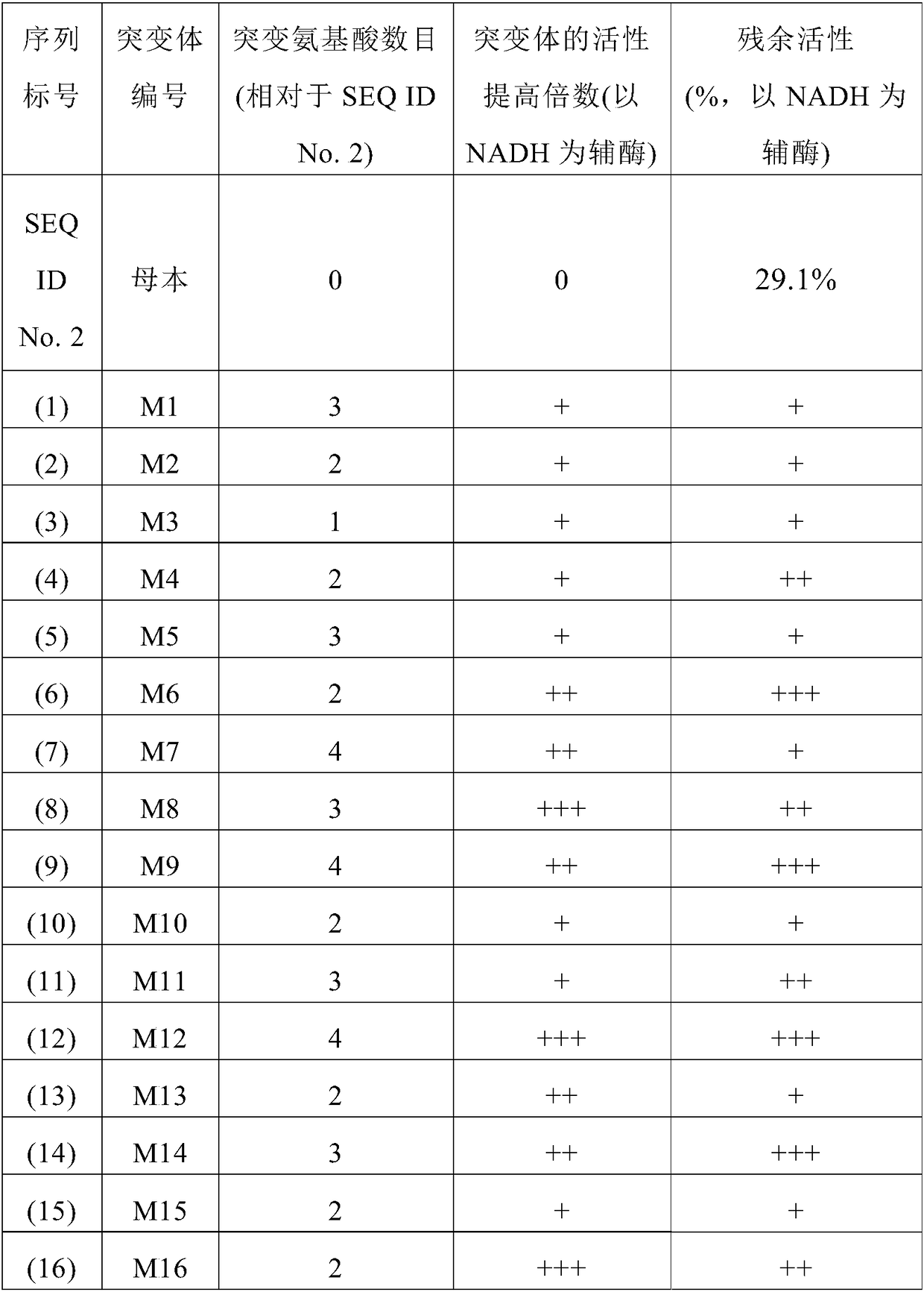
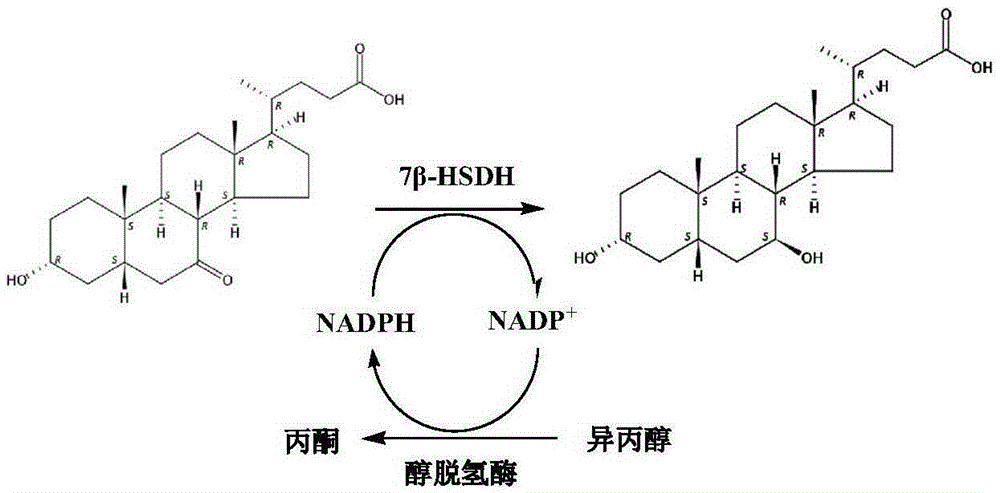
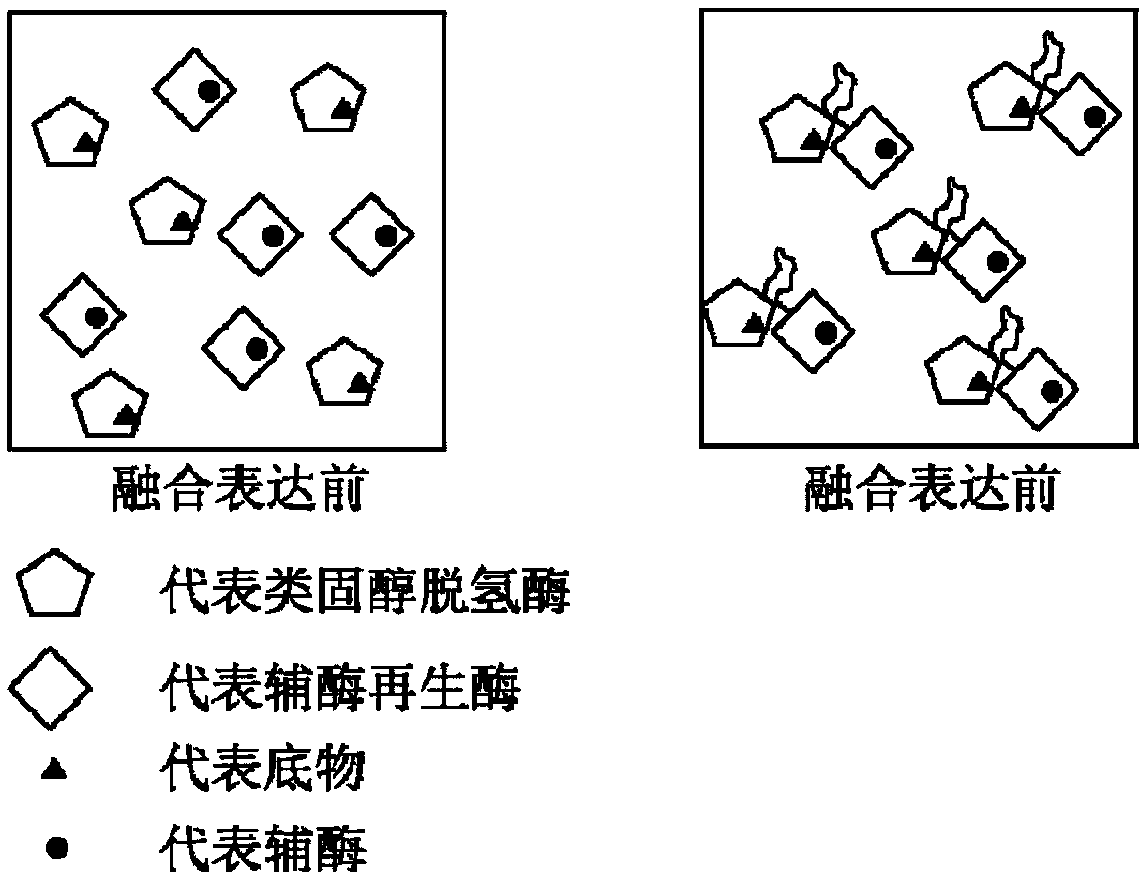
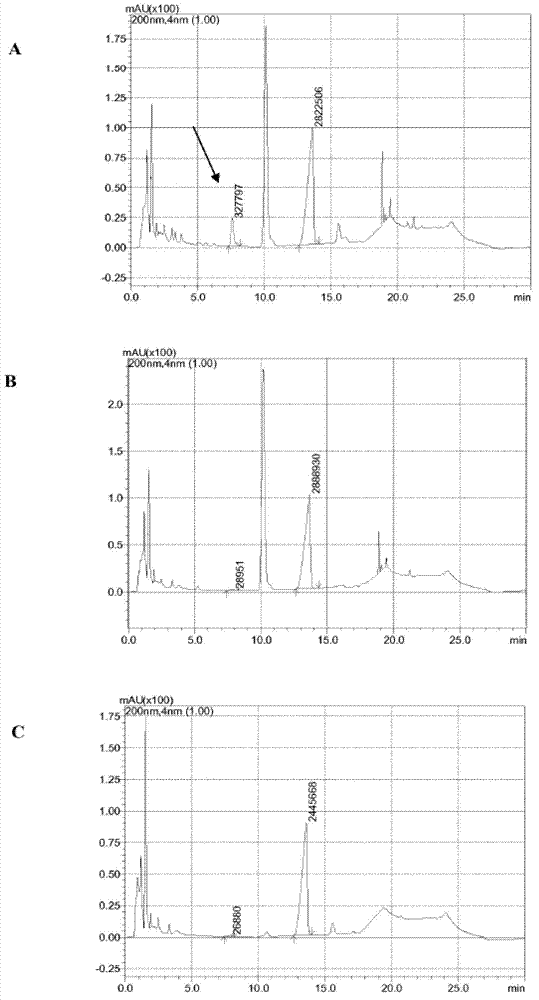
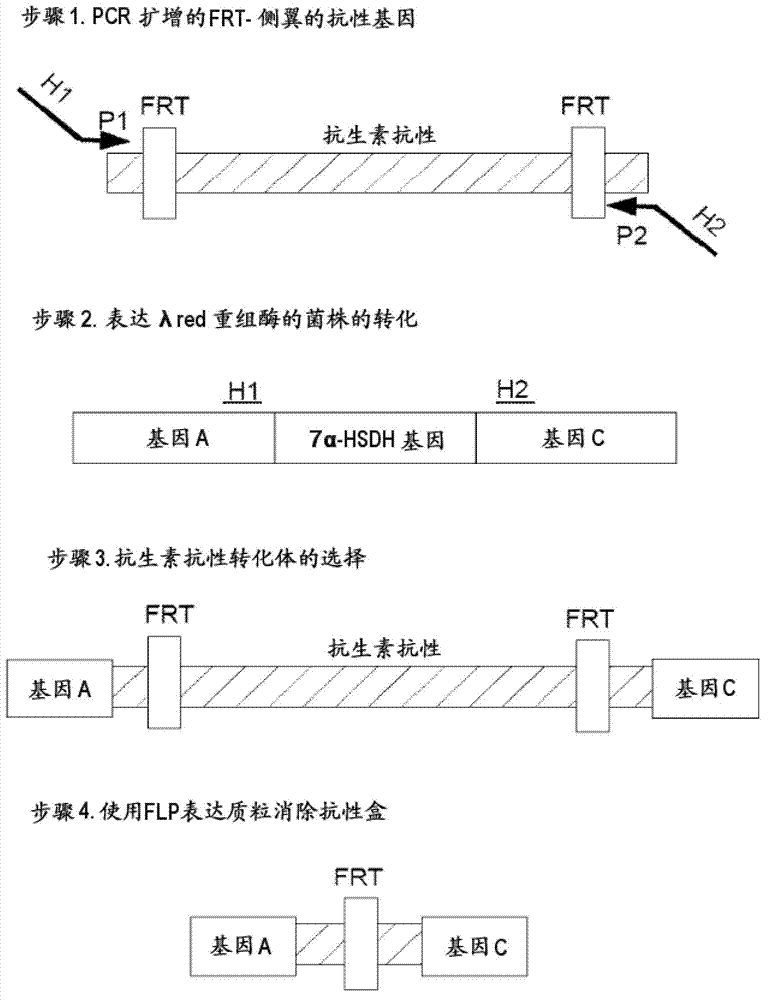
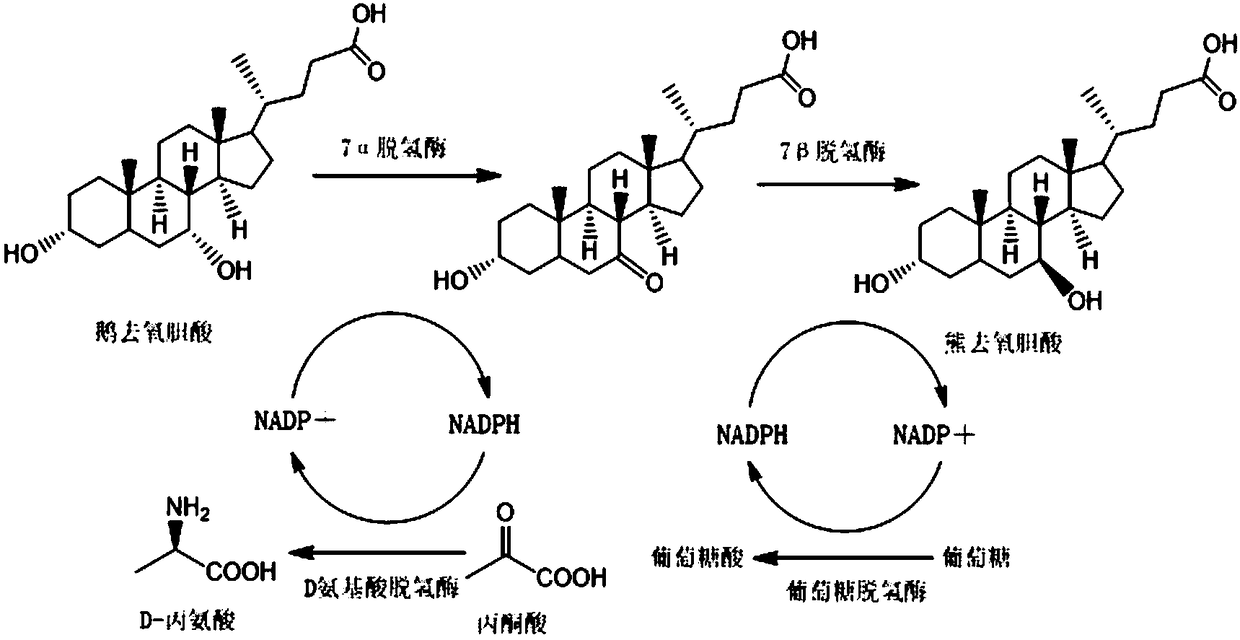
7beta-hydroxyl sterol dehydrogenase mutant and application thereof to preparation of ursodeoxycholic acid
ActiveCN108546691AEasy extractionEasy to manufactureOxidoreductasesGenetic engineeringSterolProtein engineering
The invention discloses a 7beta-hydroxyl sterol dehydrogenase mutant which is obtained by protein engineering and of which coenzyme preference is changed, a coding gene of the 7beta-hydroxyl sterol dehydrogenase mutant, a recombinant expression vector and a recombinant expression transformant which contain a sequence of the gene, a preparation method of a recombinant mutant enzyme preparation, andapplication of the recombinant mutant enzyme preparation to preparation of ursodeoxycholic acid. By co-enzyme regeneration of enzymic coupling, the recombinant mutant enzyme preparation disclosed bythe invention can efficiently utilize relatively cheap oxidized coenzyme I (NAD+) instead of very expensive oxidized coenzyme II (NADP+); asymmetric reduction of catalytic 7-hydroxyl lithocholic acideffectively reduces production cost; moreover, the recombinant mutant enzyme preparation has the advantages of simplicity for operation, mild reaction condition, environmental-friendliness, high yieldand the like, and has a good application prospect in preparation of ursodeoxycholic acid by epimerization of chenodesoxycholic acid.
Owner:EAST CHINA UNIV OF SCI & TECH +1
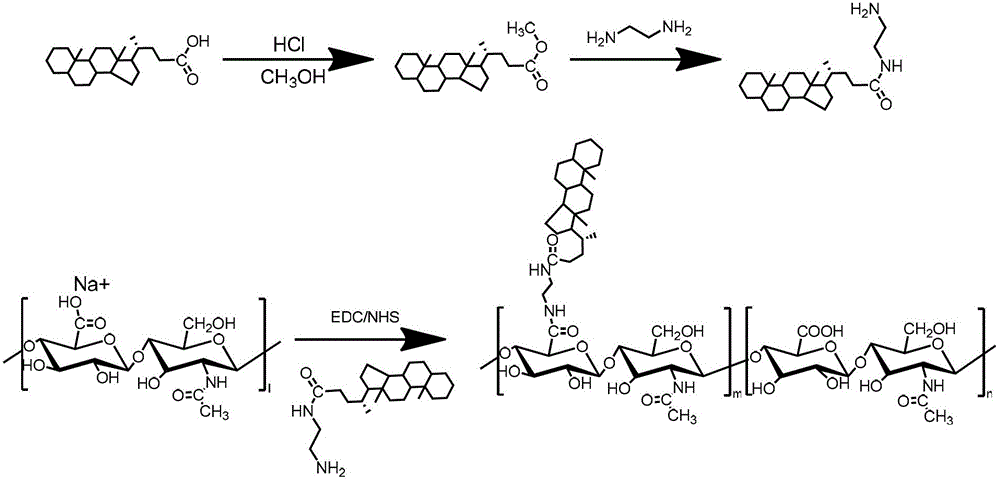
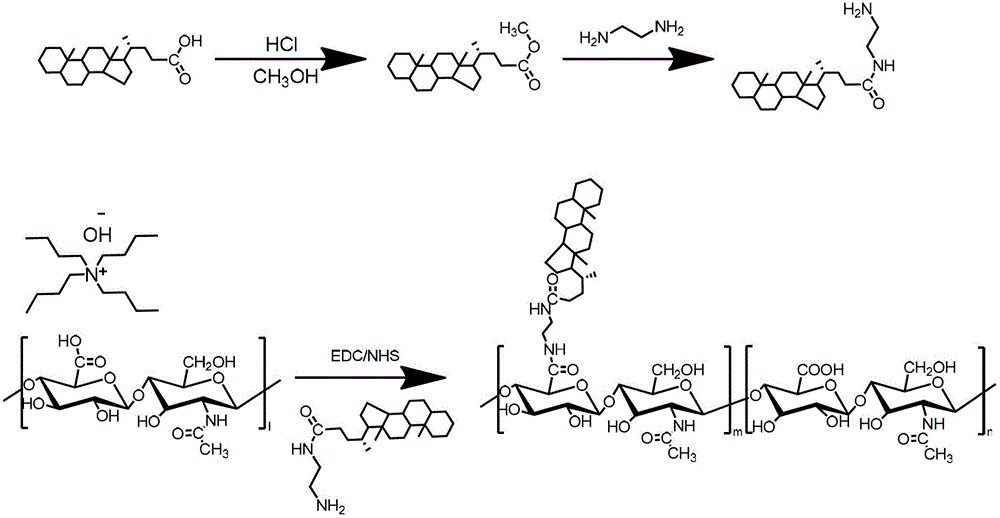
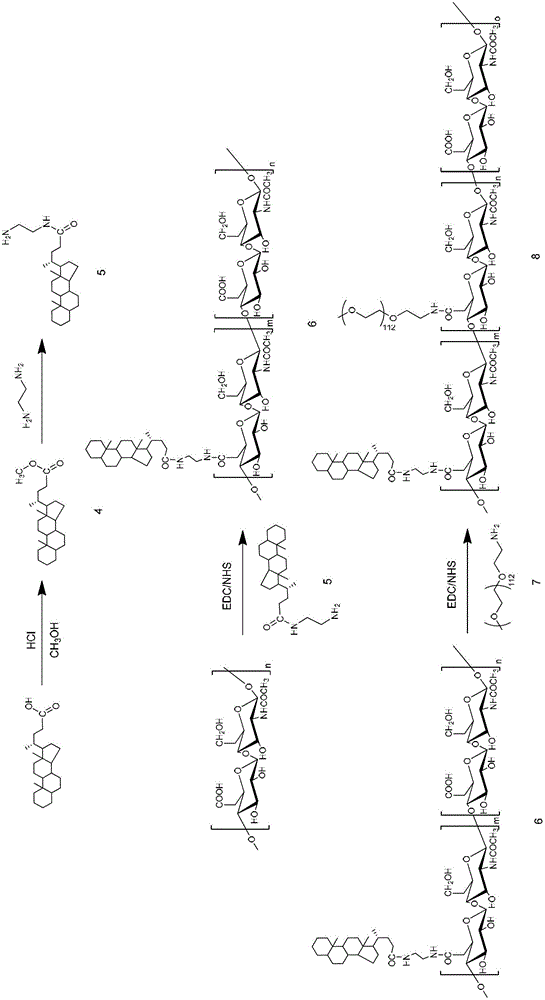
Preparation of hyaluronic-acid-based double-targeting nano-composite medicament and application of double-targeting nano-composite medicament
InactiveCN103143027AHigh mechanical strengthAccelerate the degradation rateOrganic active ingredientsPharmaceutical non-active ingredientsTumor targetDispersity
The invention relates to a hyaluronic-acid-based double-targeting nano-composite medicament and a preparation method thereof. Hydrophobic group ursodeoxycholic acid is included in a hyaluronic acid nano-polymer structure and can form an amphipathic polymer and automatically generate micelles in an aqueous solution, and polyethylene glycol can be introduced into the micelles to improve the dispersity and stability of a composite. An anti-tumor medicament can enter a nano-carrier through electrostatic adsorption or physical inclusion to generate a nano-medicament composite, wherein the nano-medicament composite is selectively concentrated in a tumor cell under an active targeting effect of hyaluronic acid and a surface CD44 receptor of a tumor cell, and promotes a tumor tissue to absorb the nano medicament-carrying composite by using a passive osmotic accumulation effect (EPR) at the same time. After an anti-tumor medicament is wrapped by a modified hyaluronic acid polymer, the anti-tumor medicament has the advantages of improving the bioavailability of the medicament, improving the targeting property, reducing the toxic and side effects, prolonging the half-life period of the medicament, being stably stored and the like, so that the tumor targeting therapy efficiency is improved in many ways.
Owner:XIAMEN UNIV
Method for producing ursodeoxycholic acid by using swine bladder as raw material
The invention provides a process of separating anthrodsoxycholic acid by using fresh swine bile as raw material, the invention further provides a process for preparing ursodeoxycholic acid by using fresh swine bile as raw material, which comprises extracting conjugated bile acid from swine bile after acidification, removing foreign materials such as aliphatic acid and protein and the like, preparing purified pig bile acid through hydrolysis (saponification) and hydroperoxide decolourization, extracting anthrodsoxycholic acid through solution immersion cleaning, preparing ursodeoxycholic acid through oxidization and reduction of anthrodsoxycholic acid which is obtained through separating. The process of the invention improves the preparation technology of ursodeoxycholic acid, and achieves positive effects on the aspects of technology simplification and output increase.
Owner:苏州天绿生物制药有限公司
Mutant of 7 beta-hydroxyl steroid dehydrogenase, application of mutant and synthesis method
ActiveCN105274070ASuitable for industrial productionEasy to controlOxidoreductasesFermentationChemical synthesisCholic acid
The invention provides a mutant of 7 beta-hydroxyl steroid dehydrogenase, application of the mutant and a synthesis method. The mutant of the 7 beta-hydroxyl steroid dehydrogenase is characterized in that amino acid sequences of the mutant are Seq ID NO:4, and coded nucleotide sequences are Seq ID NO:3; or amino acid sequences of the mutant are Seq ID NO:6, and coded nucleotide sequences are Seq ID NO:5. The mutant, the application and the synthesis method have the advantages that cholic acid compounds, particularly ursodeoxycholic acid, can be catalytically synthesized by the efficient 7 beta-hydroxyl steroid dehydrogenase, mutant enzymes of the 7 beta-hydroxyl steroid dehydrogenase and coenzyme regeneration systems, accordingly, the substrate concentration can reach 100 g / L, the conversion rate is 99.2-99.5%, and the weight yield can reach 94-96%; and the enzymes can be inexpensively and easily obtained by the aid of a fermentation process, accordingly, the production cost and the product quality are superior to the production cost and the product quality of chemical synthesis methods, and the mutant and the synthesis method are applicable to industrial production.
Owner:苏州天绿生物制药有限公司
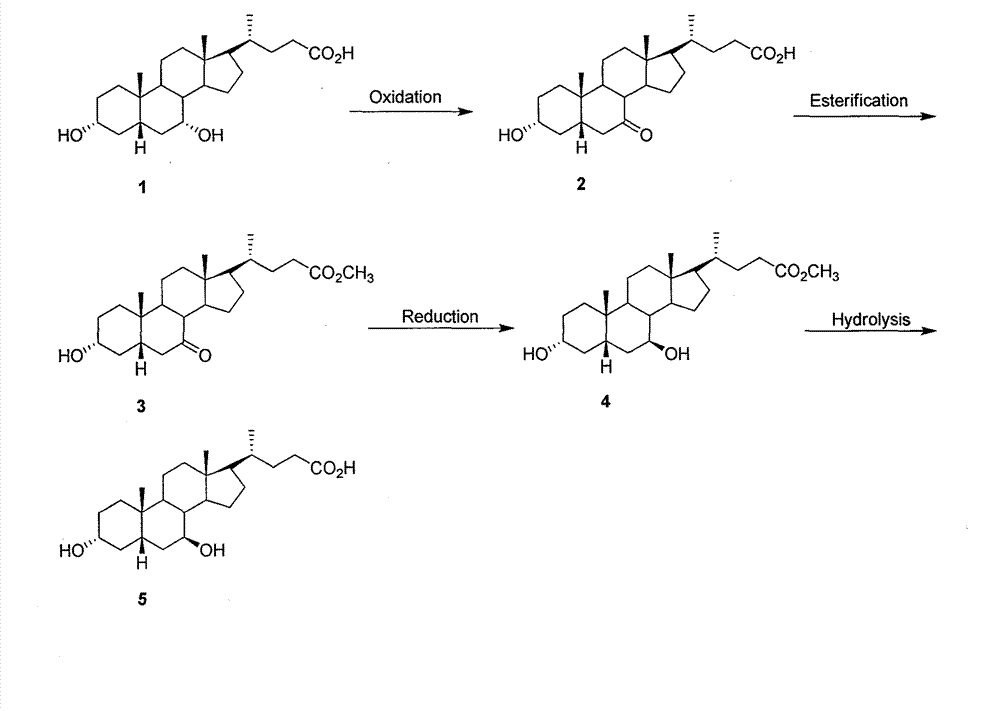
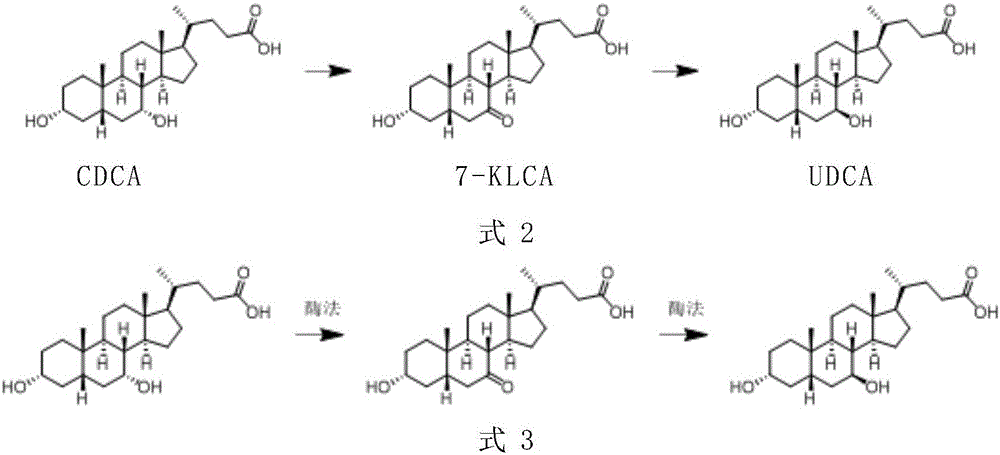
Preparation method of ursodeoxycholic acid
The invention provides a preparation method of ursodeoxycholic acid. Commercial available chenodeoxycholic acid is taken as raw materials, the ursodeoxycholic acid is obtained by four steps including selective oxidation, esterification, deoxidation and hydrolyzation, and the total yield is 85.7%. In a mixture of acetone and water, NBS is used for selective oxidation of hydroxy at C- 7 bit of the chenodeoxycholic acid, and the selective oxidation possesses excellent selectivity and high yield. NaBH14 / CeC13 may be used to deoxidize carbonyl at C-7 bit into hydroxy, and the ratio of alpha / beta is as high as 5 / 95.
Owner:EAST CHINA UNIV OF SCI & TECH
A chemical-enzyme method of preparing ursodeoxycholic acid
ActiveCN106086149AHigh ee valueEliminate enzyme inactivationSteroidsFermentationChenodeoxycholic acidOxidizing agent
A chemical-enzyme method of preparing ursodeoxycholic acid is disclosed. The method adopts chenodeoxycholic acid as an initial substrate, and prepares the ursodeoxycholic acid through a chemical process and a bio-enzyme process in order, wherein 7-KLCA reductase is adopted as a biological catalyst. A situation that an oxidant residual in a process of preparing 7-ketolithocholic acid through a chemical manner causes later enzyme inactivation in the prior art does not occur. A product prepared by the method is high in ee value and low in comprehensive cost.
Owner:ENZYMEWORKS
Berberine salts, ursodeoxycholic salts and combinations, methods of preparation and application thereof
Owner:SHENZHEN HIGHTIDE BIOPHARM
Ursodeoxycholic acid compound for preventing or treating FXR (farnesol X receptor)-mediated disease
ActiveCN106008639AGood pharmacokinetic parametersImprove securityOrganic active ingredientsMetabolism disorderFatty liverEnantiomer
Owner:SHENZHEN TARGETRX INC
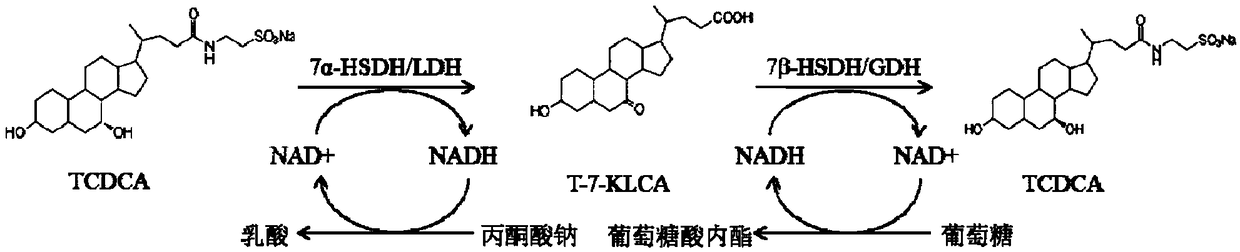
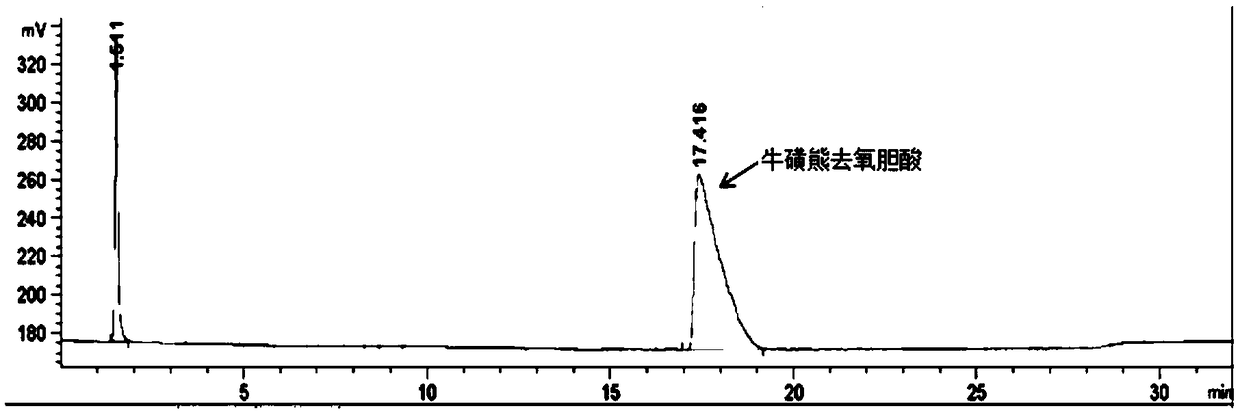
Method of preparing tauroursodeoxycholic acid by biotransformation and application of method
ActiveCN109402212AImprove conversion rateAvoid affinity purificationAntibody mimetics/scaffoldsNucleic acid vectorDrug biotransformationSubstrate concentration
The invention discloses a method of preparing tauroursodeoxycholic acid by biotransformation and application of the method. Biotransformation includes genetic codon optimization, engineered bacteria construction, engineered bacteria cultivation, substrate transformation and product preparation. Tauroursodeoxycholic acid is prepared by transforming a substrate through direct fermentation of engineered bacteria; the substrate is taurochenodeoxycholic acid. The substrate may reach 250 g / L in concentration; the reaction time is short; substrate transformation rate reaches 98% and above; the obtained product reaches 99% and above in purity; cyclic regeneration of NAD+ (nicotinamide adenine dinucleotide +) in the reaction system helps greatly reduce the usage of the coenzyme NAD+; the cost of enzymic catalytic reaction is reduced; industrial amplification is benefited. hydroxysteroid dehydrogenase and the regenerated coenzyme are connected via a flexible polypeptide sequence to form a protein fusion polymer; binding distances to the substrate and coenzyme are shorter; transformation progress is more facilitated; the number of fermenting times in industrial production is decreased; the process is simplified; time cost and material cost are saved.
Owner:JIANGSU BANGZE BIOLOGICAL MEDICINE TECH CO LTD
Ursodeoxycholic acid solid dispersoid, preparation method and solid preparation
The invention provides an ursodeoxycholic acid solid dispersoid, a preparation method and a solid preparation. The solid dispersoid comprises ursodeoxycholic acid (A), a surfactant (B) and a hydrophilic material (C), wherein the weight ratio of (A) and (C) is 1:5-1:0.1, and the content of (B) in the ursodeoxycholic acid solid dispersoid is 2-60wt%.
Owner:SHANGHAI HUIDE MEDICINE SCI & TECH +1
Method of treating nonalcoholic steatohepatitis with elevated doses of ursodeoxycholic acid
InactiveUS20120071451A1Reduces fibrosis level levelReduces level liver inflammation levelOrganic active ingredientsBiocideBlood sugarNonalcoholic steatohepatitis
The present invention is directed to a method for the treatment of nonalcoholic steatohepatitis (NASH) by administering an elevated dose of ursodeoxycholic acid (UDCA), or a pharmaceutically acceptable salt thereof, to a patient in need of such treatment, wherein the patients demonstrate a significantly improved glycemic profile during treatment.
Owner:APTALIS PHARMA CANADA
Method for extracting volatile components from Pandanus amaryllifolius Roxb leaves
ActiveCN104083900AHigh yieldImprove extraction efficiencySolid solvent extractionDihydroactinidiolideChemical composition
The invention discloses a method for extracting volatile components from Pandanus amaryllifolius Roxb leaves by use of a supercritical CO2 extraction method. The method can obtain 151 compounds from the volatile components of Pandanus amaryllifolius Roxb, but a steam distillation method only can obtain 95 compounds, and extra 56 compounds, such as n-heptatriacontanyl alcohol, methyl linolenate, phytyl acetate, 3-hydroxyl-7 (isonitroso) ursodeoxycholic acid and dihydroactinidiolide, which cannot be obtained by the steam distillation method, are obtained by the method provided by the invention. The method for extracting the volatile components from Pandanus amaryllifolius Roxb leaves is superior to the steam distillation method in the aspects of smell, yield, chemical composition, squalene yield and extracting efficiency.
Owner:GUANGZHOU ZHONGDA NANSHA TECH INNOVATION IND PARK +1
Compound feed for enhancing physiological functions of intestinal mucosa and liver/pancreas cells of fishes
InactiveCN102067955APromote growthFavorable metabolismFood processingClimate change adaptationBiotechnologySodium Bentonite
The invention belongs to a feed, and particularly relates to a compound feed for enhancing the physiological functions of intestinal mucosa and liver / pancreas cells of fishes, which contains fish meal, soybean meal, rapeseed meal, cottonseed meal, wheat, rice bran, rice bran meal, calcium biphosphate, zeolite powder, bentonite, DL carnitine, soybean oil, vitamin-mineral premix, ursodeoxycholic acid, taurine, glutamine and yeast culture. An experimental fish raised with feed, which serves as an experimental fish for the separation and primary culture of intestinal mucosa cells and liver cells,can obtain healthy intestinal mucosa cells and healthy liver cells, moreover, the physiological condition of the experimental fish can be kept in consistency, and the repeatability of a separation and primary culture experiment on the intestinal mucosa cells and the liver cells.
Owner:SUZHOU UNIV
7beta-hydroxysterol dehydrogenase mutant and application of 7beta-hydroxysterol dehydrogenase mutant in ursodeoxycholic acid synthesis
ActiveCN107099516AEasy to separate and extractOvercoming the problem of inactivation processingOxidoreductasesGenetic engineeringChenodeoxycholic acidSubstrate concentration
The invention discloses a 7beta-hydroxysterol dehydrogenase mutant with increased activity and stability which is obtained through molecular evolution, recombinant expression plasmid containing the 7beta-hydroxysterol dehydrogenase mutant gene and a recombinant expression transformant and a preparation method of a recombinant mutant enzyme preparation, and the invention also provides an application of the recombinant mutant enzyme preparation in ursodeoxycholic acid synthesis. The 7beta-hydroxysterol dehydrogenase has excellent activity and heat stability, can efficiently catalyze asymmetric reduction of 7-carbonyl lithocholic acid to prepare the ursodeoxycholic acid; the 7beta-hydroxysterol dehydrogenase is subjected to immobilization and then is subjected to couple by an enzyme method with the immobilized 7beta-hydroxysterol dehydrogenase, epimerization of a substrate chenodeoxycholic acid with low cost can be directly catalyzed, ursodeoxycholic acid can be prepared through continuous conversion, and the operation is simple. Compared with the prior art reported currently, ursodeoxycholic acid prepared by hydroxysterol dehydrogenase through catalysis has the advantages of high substrate concentration, short reaction time, complete reaction, and high product purity, and has strong industrial application prospect.
Owner:EAST CHINA UNIV OF SCI & TECH +1
NOVEL 7Beta-HYDROXYSTEROID DEHYDROGENASE MUTANTS AND PROCESS FOR THE PREPARATION OF URSODEOXYCHOLIC ACID
The invention relates to novel 7β-hydroxysteroid dehydrogenase mutants, to the sequences which encode these enzyme mutants, to processes for the preparation of the enzyme mutants and to their use in enzymatic reactions of cholic acid compounds, in particular in the preparation of ursodeoxycholic acid (UDCS). The invention also relates to novel processes for the synthesis of UDCS using the enzyme mutants; and to the preparation of UDCS using recombinant, multiply-modified microorganisms.
Owner:PHARMAZELL GMBH
Novel 7alpha-hydroxysteroid dehydrogenase knockout mutants and use thereof
The invention relates to novel microbial 7alpha-hydroxysteroid dehydrogenase (7alpha-HSDH) knockout mutants and to the use thereof for producing other HSDHs having various functionalities, such as 3alpha-, 7beta- or 12alpha-HSDH, and to the use of thus-produced HSDH enzymes in enzymatic reactions of cholic acid compounds, and in particular for producing ursodeoxycholic acid (UDCS). The invention relates in particular to novel methods for synthesizing UDCS.
Owner:CELL PHARM CO LTD
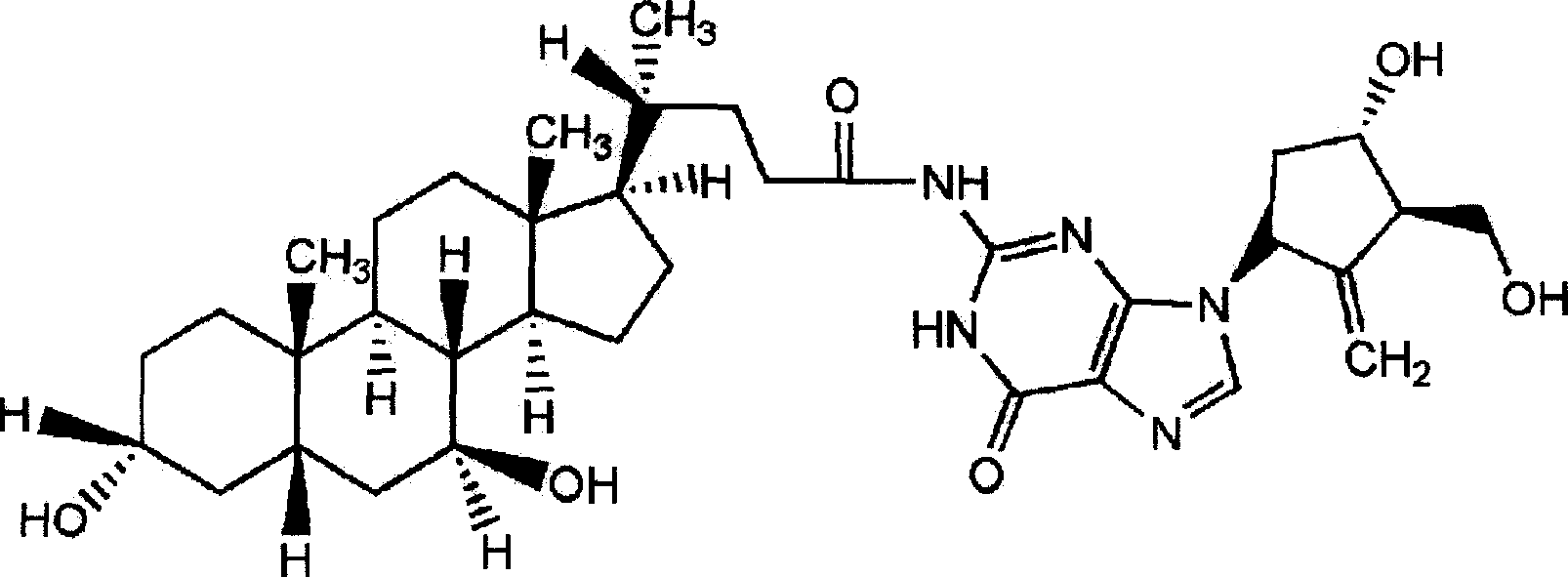
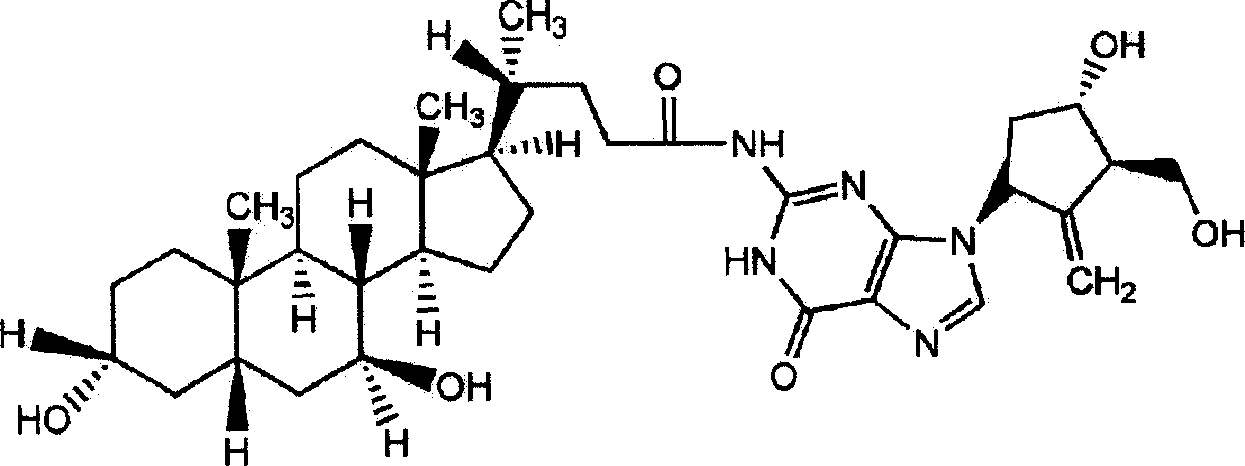
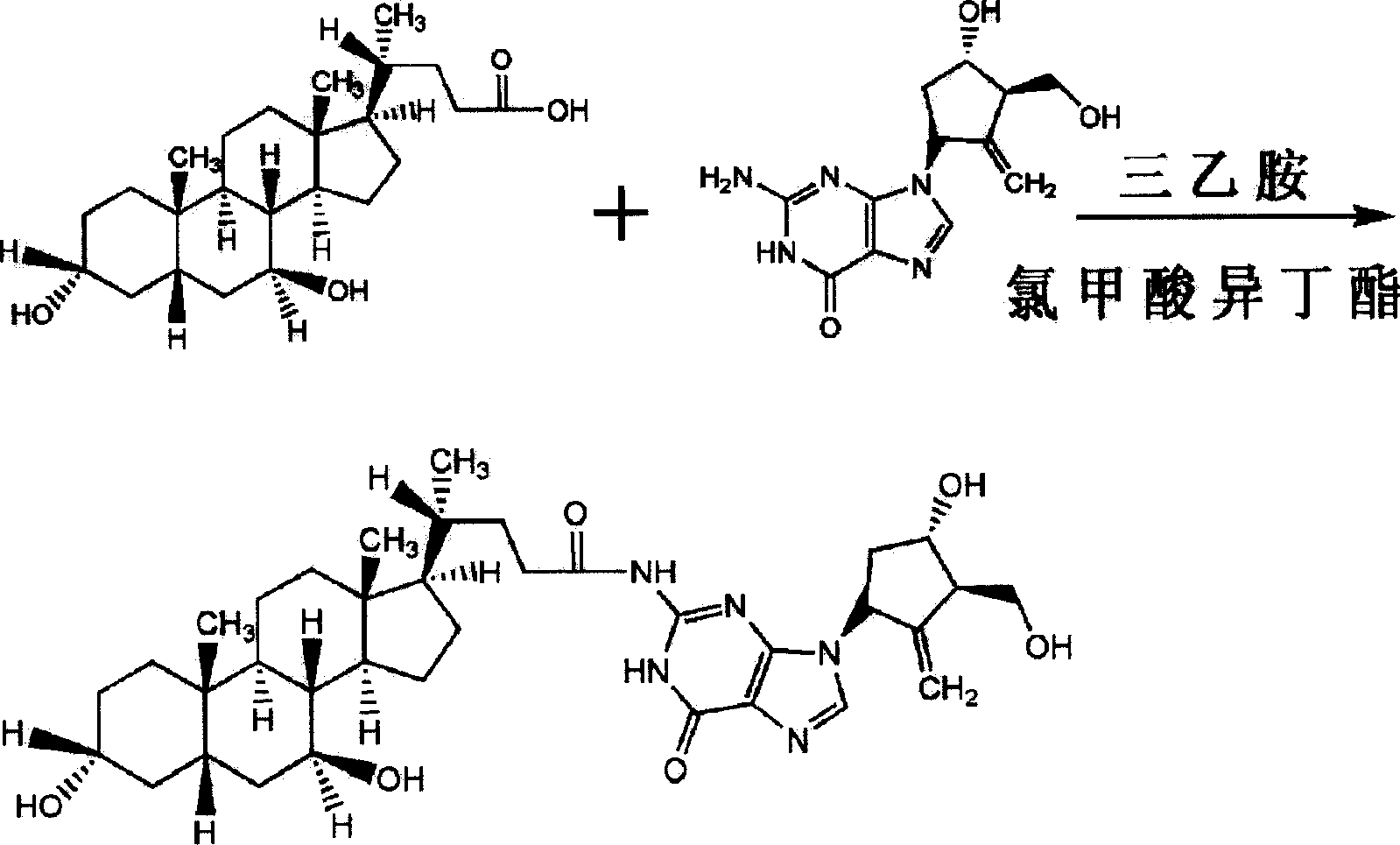
Ursodeoxycholic acid entecavir acidamide and preparation method and use thereof
InactiveCN101134769AEffective treatmentOrganic active ingredientsPowder deliveryActive componentMedicine
The present invention relates to one kind of new compound, entecavir amido ursodesoxycholate, its preparation process, medicine composition with the compound as active component and the application of the compound in preventing and treating hepatitis B.
Owner:曾昭斌
Method for synthesizing ursodeoxycholic acid through enzymatic method
The invention discloses a method for synthesizing ursodeoxycholic acid through an enzymatic method and a method for synthesizing the ursodeoxycholic acid by taking chenodeoxycholic acid as a raw material. The method takes the chenodeoxycholic acid as a base material and comprises the following steps: dissolving the chenodeoxycholic acid into a 50mM phosphate buffer solution; firstly, catalyticallyoxidizing the chenodeoxycholic acid by utilizing 7-alpha hydroxysteroid dehydrogenase in the presence of NAD, NOX2 and under the condition that oxygen is introduced, so as to obtain 7-ketolithocholicacid; then under the condition that the NAD, L-malic acid and malic dehydrogenase exist, catalytically reducing the 7-ketolithocholic acid by utilizing the 7-alpha hydroxysteroid dehydrogenase to obtain the ursodeoxycholic acid. According to the method disclosed by the invention, an organic solvent is not used and operation is simple; reaction conditions are moderate and easy to control and the utilization rate of the raw materials is high; the conversion rate reaches 99 percent or more.
Owner:ZHONGSHAN BAILING BIOTECHNOLOGY CO LTD
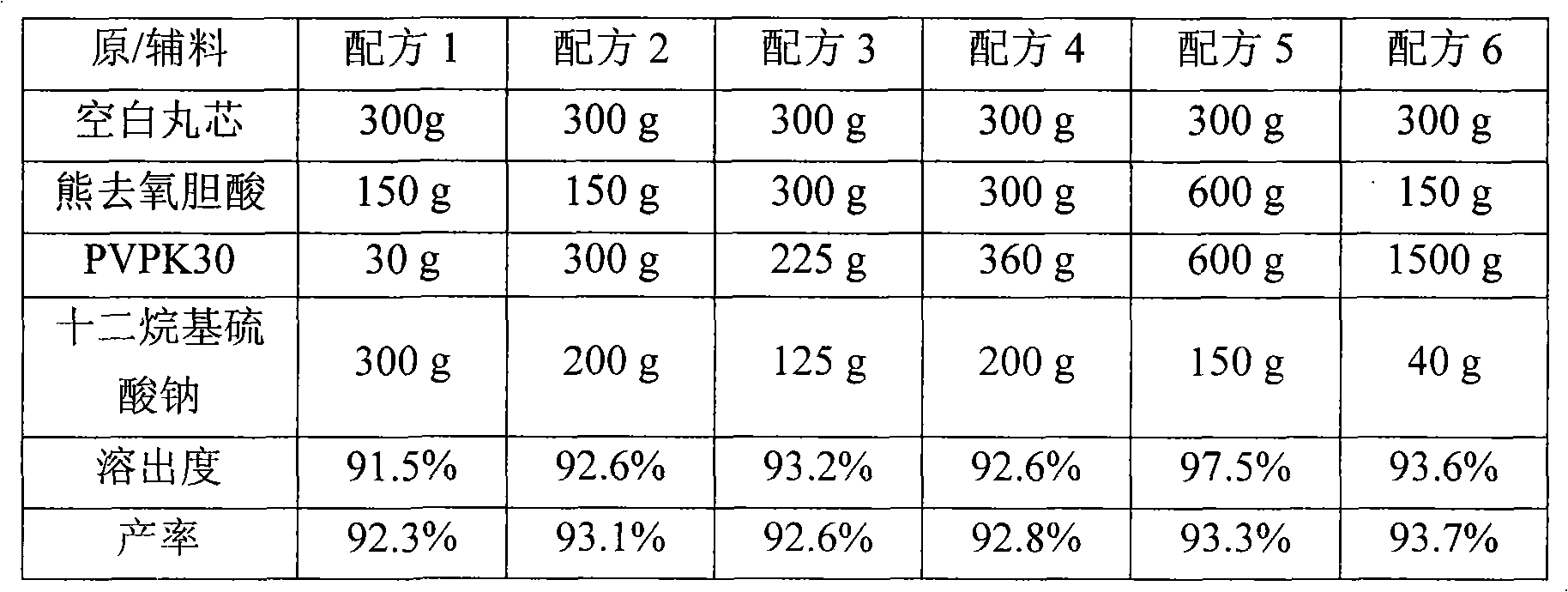
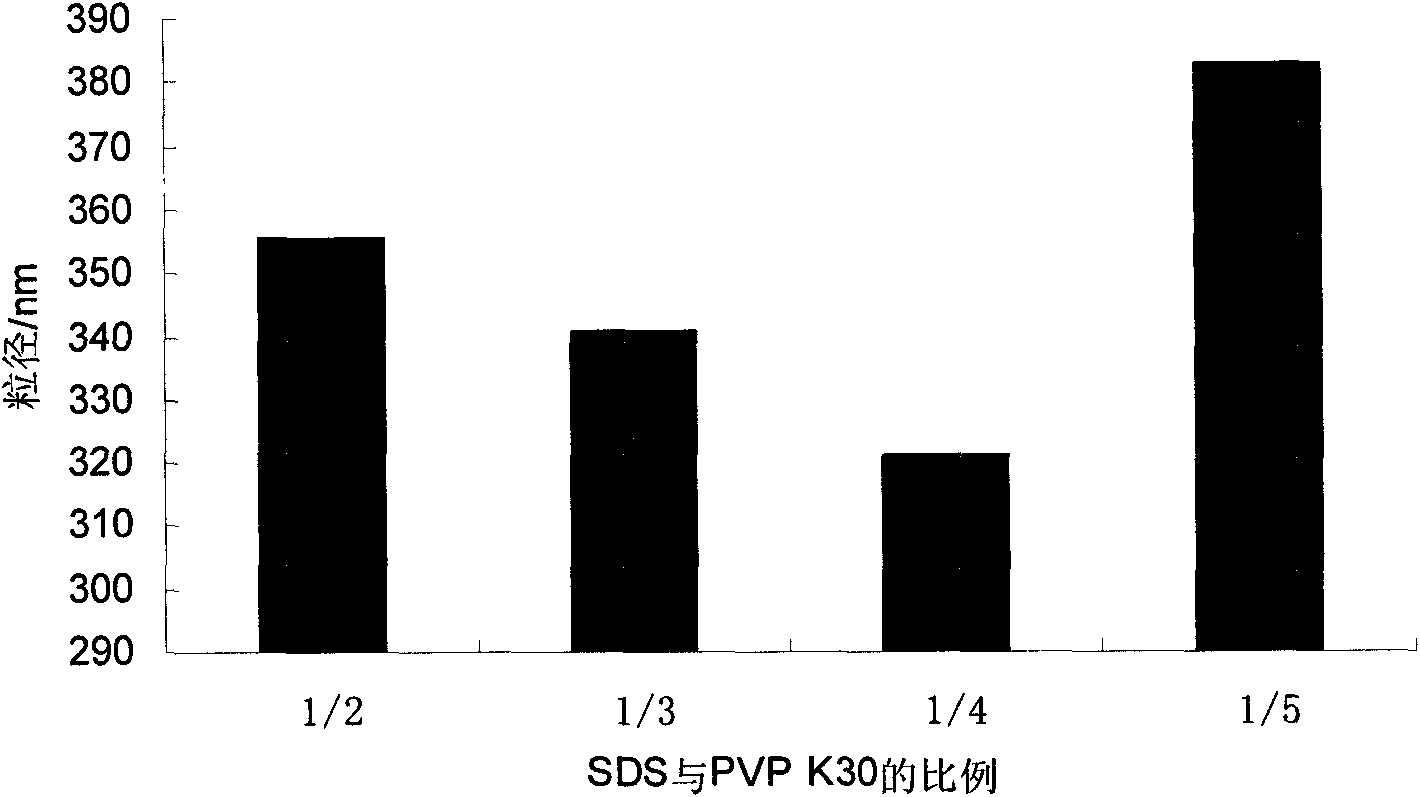
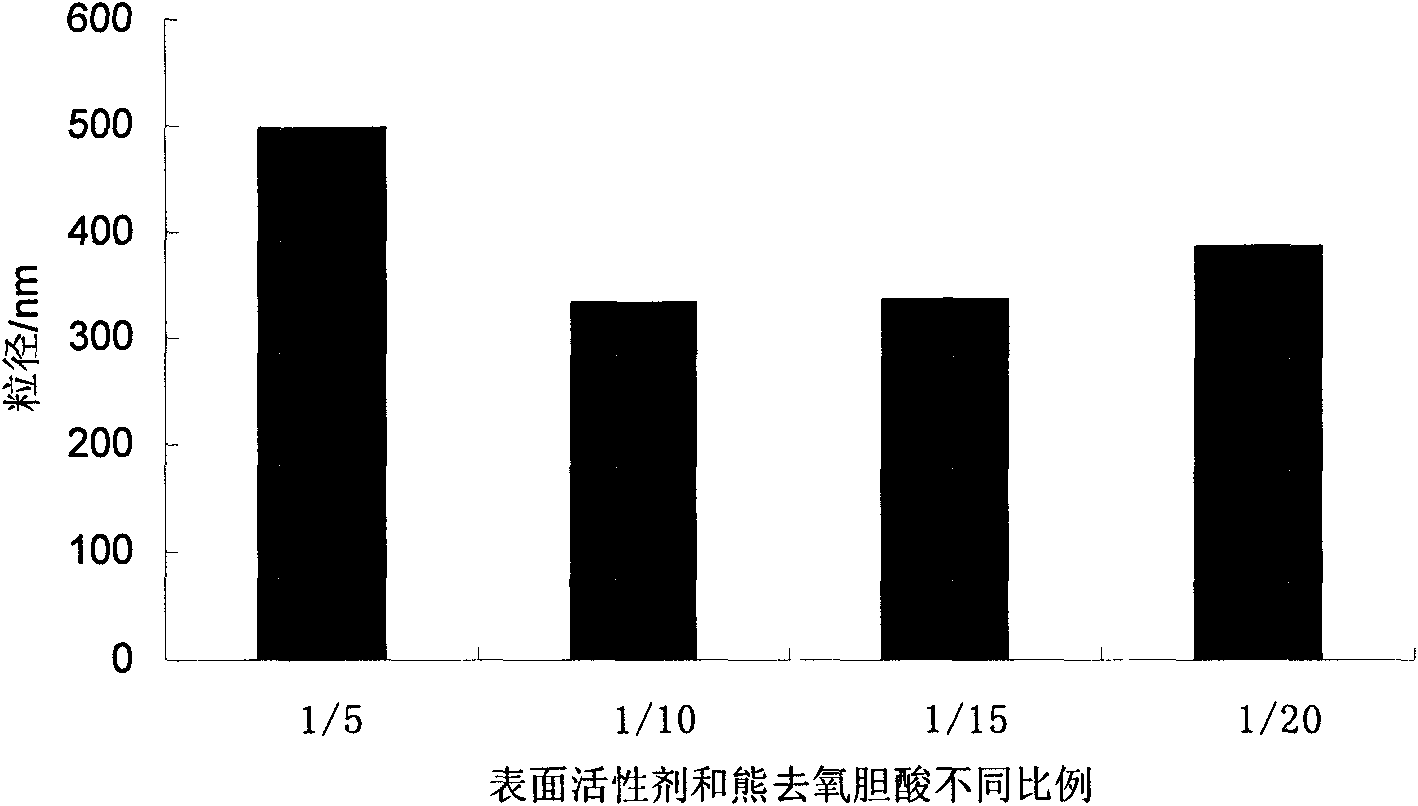
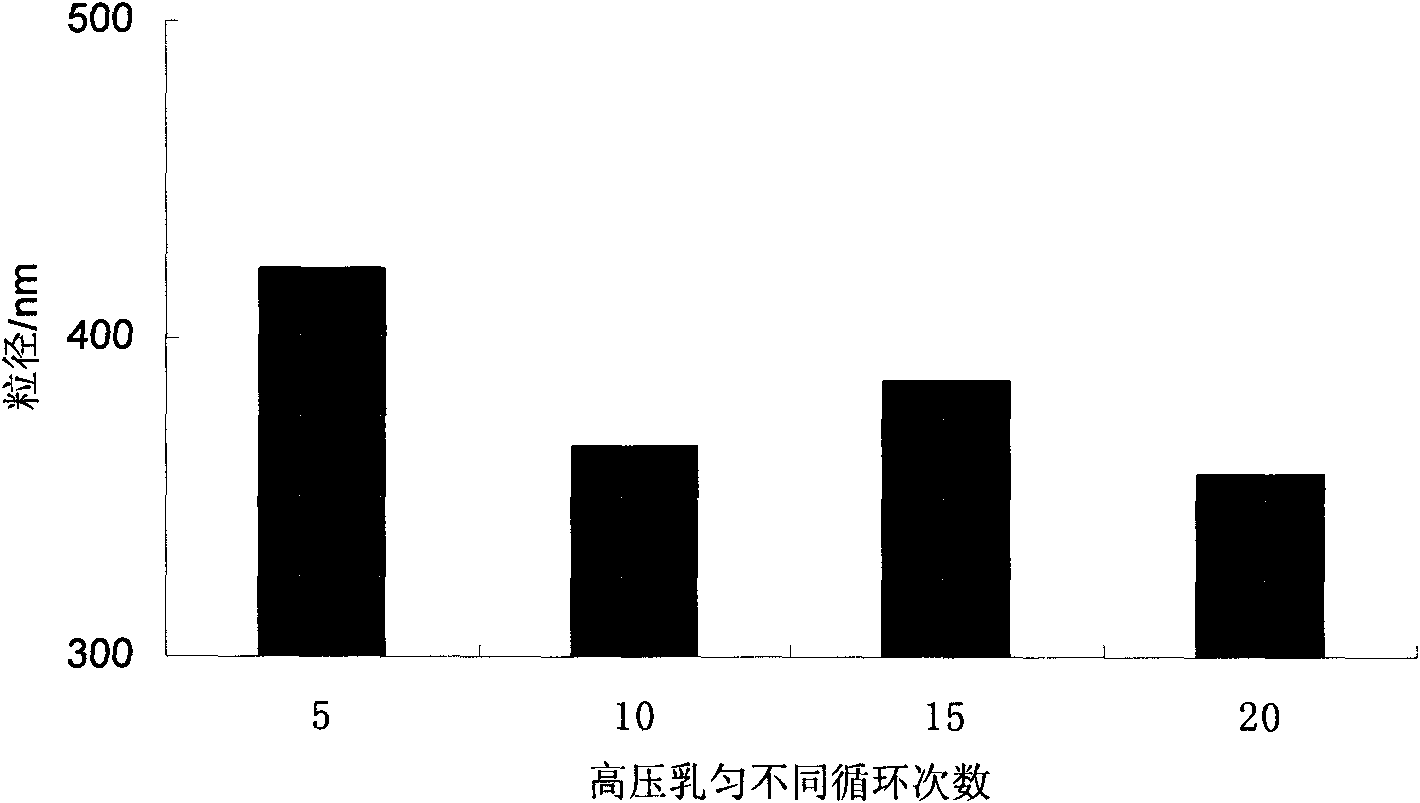
Ursodeoxycholic acid nano suspension and preparation method thereof
InactiveCN101606906AHigh dissolution rateImprove bioavailabilityPowder deliveryOrganic active ingredientsOxygenSURFACTANT BLEND
The invention discloses an ursodeoxycholic acid nano suspension and a preparation method thereof, relates to the field of pharmaceutical nano formulation, and particularly relates to an ursodeoxycholic acid nano suspension for curing cholestatic cirrhosis and dry powder thereof. The ursodeoxycholic acid nano suspension is characterized by adopting SDS and PVP K30 as surfactants and combining the SDS and the PVP K30 in a weight ratio of 1:1-1:3; and the ursodeoxycholic acid nano suspension has stable quality, with particle size of about 200-800nm. The invention also discloses a preparation method of the suspension.
Owner:302 MILITARY HOSPITAL OF CHINA
Method for producing ursodesoxycholic acid from 98.0% of cholic acid of cattle and sheep
The invention relates to a productive process for producing ursodeoxycholic acid by using 98.0% bovine and sheep cholic acid, which comprises conducting four steps of esterification, acetylization, oxidization and reduction for bovine and sheep cholic acid, transforming cholic acid with isomer into the structure of anthrodsoxycholic acid to form the compound of anthrodsoxycholic acid, obtaining ursodeoxycholic acid after reaction through utilizing anthrodsoxycholic acid, separating to obtain ursodeoxycholic acid with 98.5% purity by utilizing acetic acid ethyl ester. The productive process has simple technical process, low productive cost, and solves the problems of less fowl gall quantity and difficult collection. And the purity of ursodeoxycholic acid which is synthesized is high, which reaches to the international advanced level.
Owner:辽宁百隆生物工程有限公司
NOVEL 7beta-HYDROXYSTEROID DEHYDROGENASES AND THEIR USE
InactiveUS20130040341A1Solve problemsOxidoreductasesFermentationCholic acidHydroxysteroid Dehydrogenases
The invention relates to novel 7β-hydroxysteroid dehydrogenases which are obtainable from bacteria of the genus Collinsella, especially of the strain Collinsella aerofaciens, to the sequences encoding said enzymes, to methods for producing said enzymes and to their use in the enzymatic conversion of cholic acid compounds, and especially in the production of ursodeoxycholic acid (UDCS). The invention also relates to novel methods for the synthesis UDCS.
Owner:PHARMAZELL GMBH
Method for synthesizing ursodeoxycholic acid and high-chiral-purity D-amino acid based on enzyme-method coupling technology
The invention discloses a method for synthesizing ursodeoxycholic acid (UDCA) and high-chiral-purity D-amino acid based on an enzyme-method coupling technology. The method comprises the following steps: putting chenodeoxycholic acid and alpha-ketonic acid into a solution system containing 7alpha-HSDH (Homoserine Dehydrogenase), DAADH and NADP (Nicotinamide Adenine Dinucleotide Phosphate) and carrying out enzyme catalysis reaction; separating a reaction solution by adopting an ultra-filtration membrane to obtain a concentrated mixed enzyme solution; regulating the pH (Potential of Hydrogen) ofa dialysis solution and crystallizing; filtering and separating to obtain 7-KLCA wet powder and filtrate; carrying out chromatographic treatment on the filtrate to obtain the D-amino acid; putting the7-KLCA wet powder into a solution system containing glucose, the NADP, the 7alpha-HSDH and GDH (Glutamate Dehydrogenase) and carrying out enzyme catalysis reaction; separating the reaction solution by adopting the ultra-filtration membrane to obtain the concentrated mixed enzyme solution; crystallizing, filtering and separating the dialysis solution, so as to obtain ursodeoxycholic acid. By adopting the method provided by the invention, UDCA and the high-chiral-purity D-amino acid can be obtained at the same time, the enzyme utilization rate is high, synthesis steps are simple and the cost isreduced; meanwhile, a metal reduction reagent and an organic solvent do not need to be added in a reaction process and conditions are mild; the method is environmentally friendly and is suitable forindustrial production.
Owner:HUNAN BAOLISHI BIOTECH
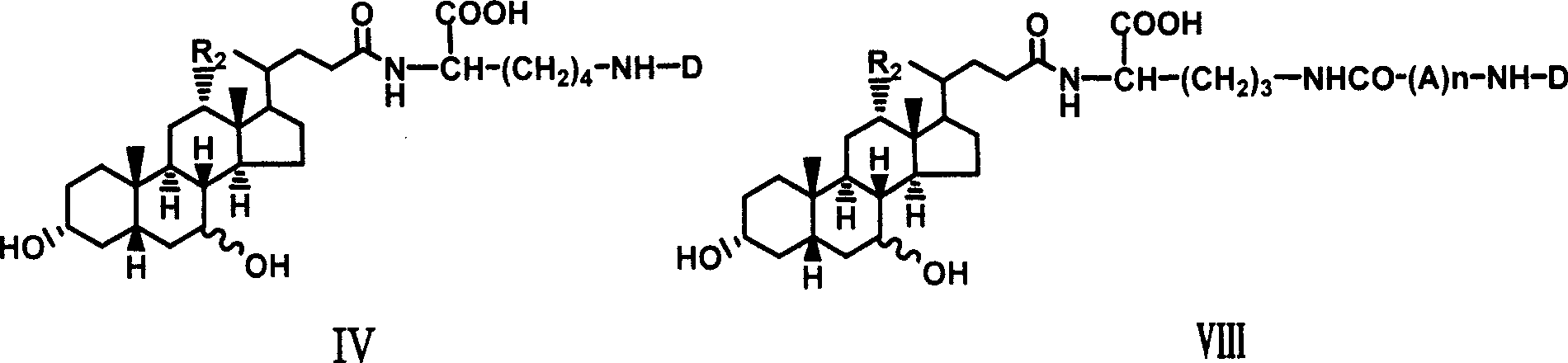
Novel coupling compound of bile acid and anti-hepatitis virus medicament and medical use thereof
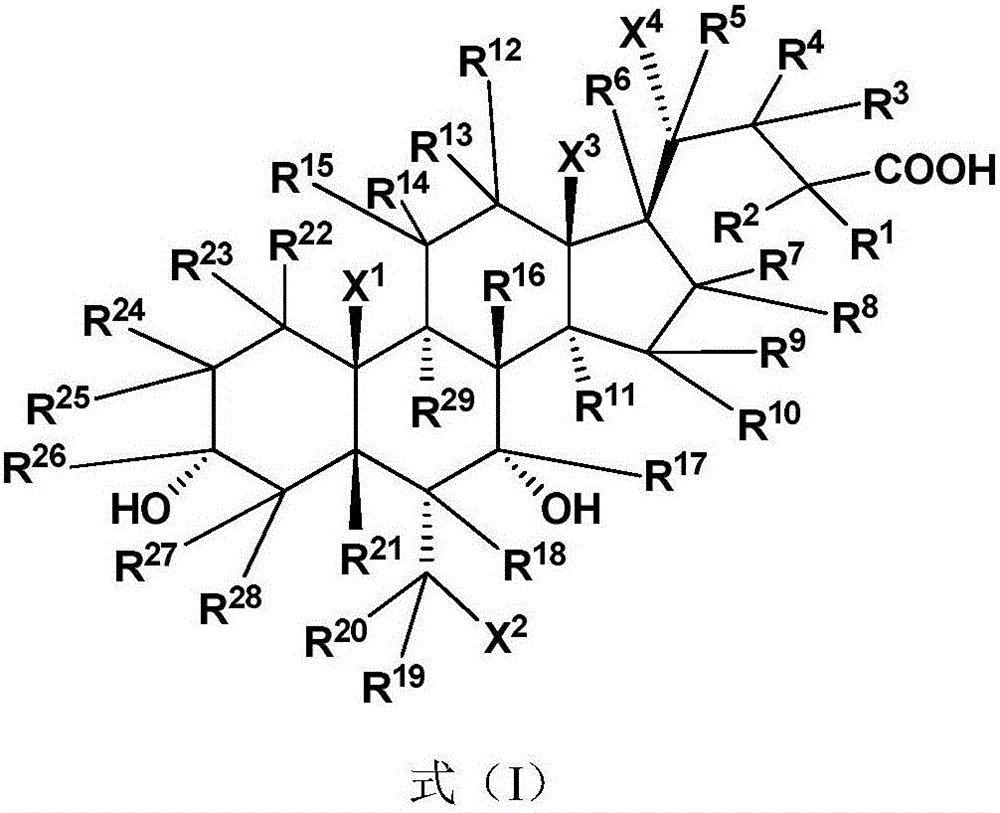
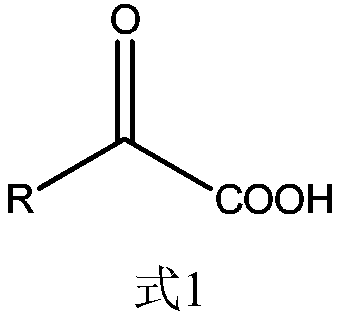
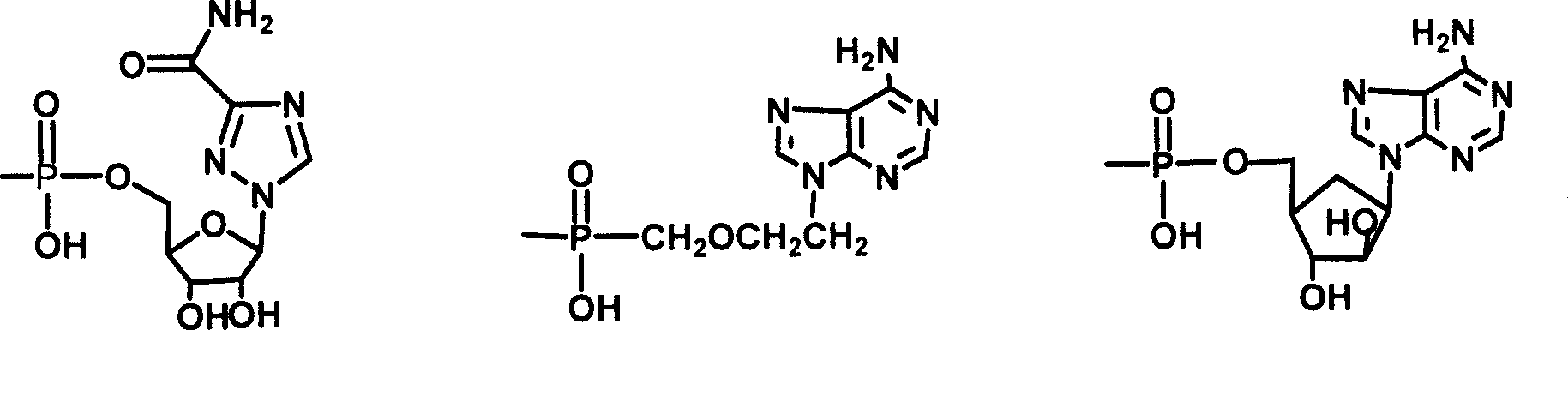
The invention relates to a bile acid-anti-hepatitis virus drug conjugate which is shown in the following formula, non-toxic pharmaceutically acceptable salts thereof, a preparation method, a pharmaceutical composition containing the compounds and a use. In the structural formula of figure (I), R represents OH (bile acid) or H (ursodeoxycholic acid), A represents amino acid, n is an integer from 1 to 3, and the structure of the amino acid is D type or L type. D represents the above structure (II).
Owner:INST OF PHARMACOLOGY & TOXICOLOGY ACAD OF MILITARY MEDICAL SCI P L A
Preparation process of deoxycholeic acid of bear
InactiveCN101289488AThe process steps are simpleReasonable process routeSteroidsChemical recyclingChenodeoxycholic acidChemical reaction
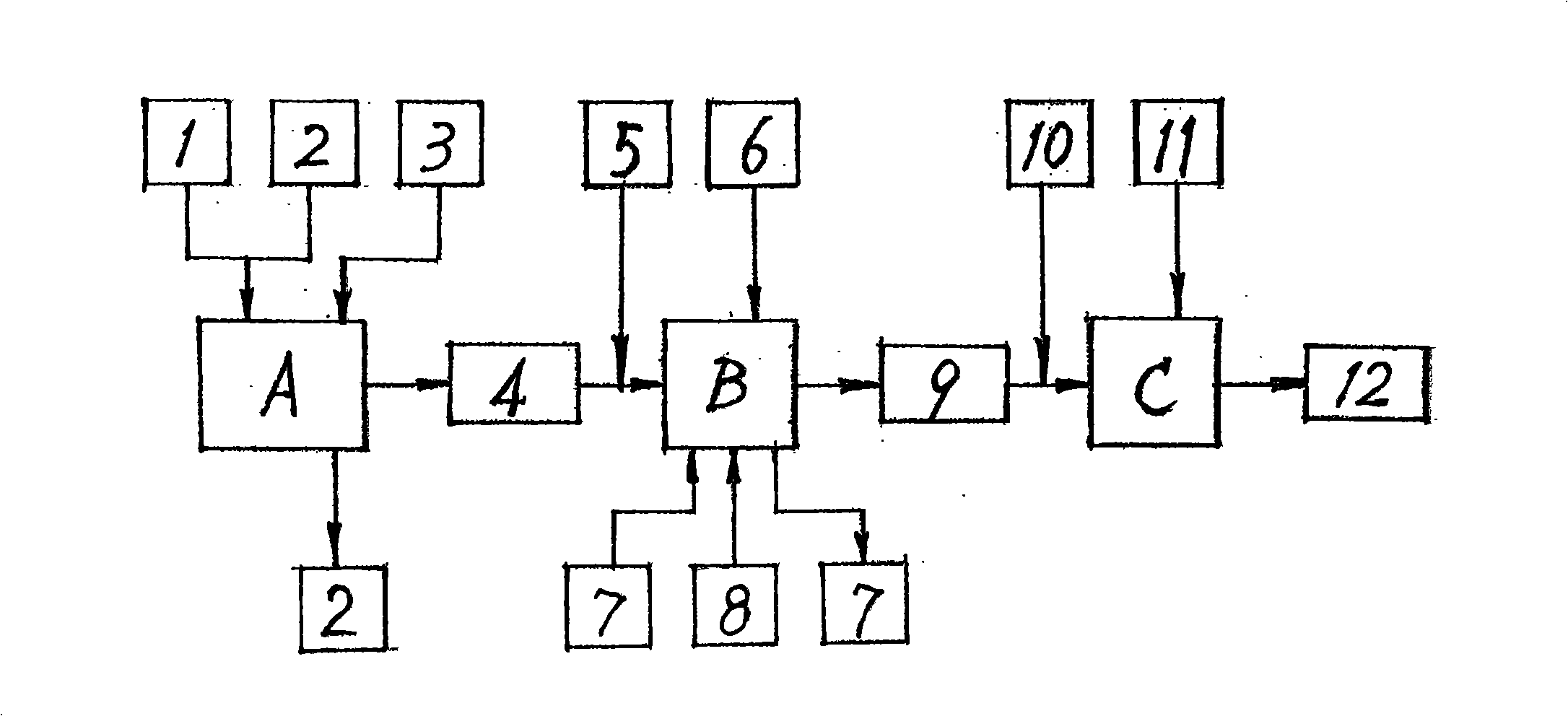
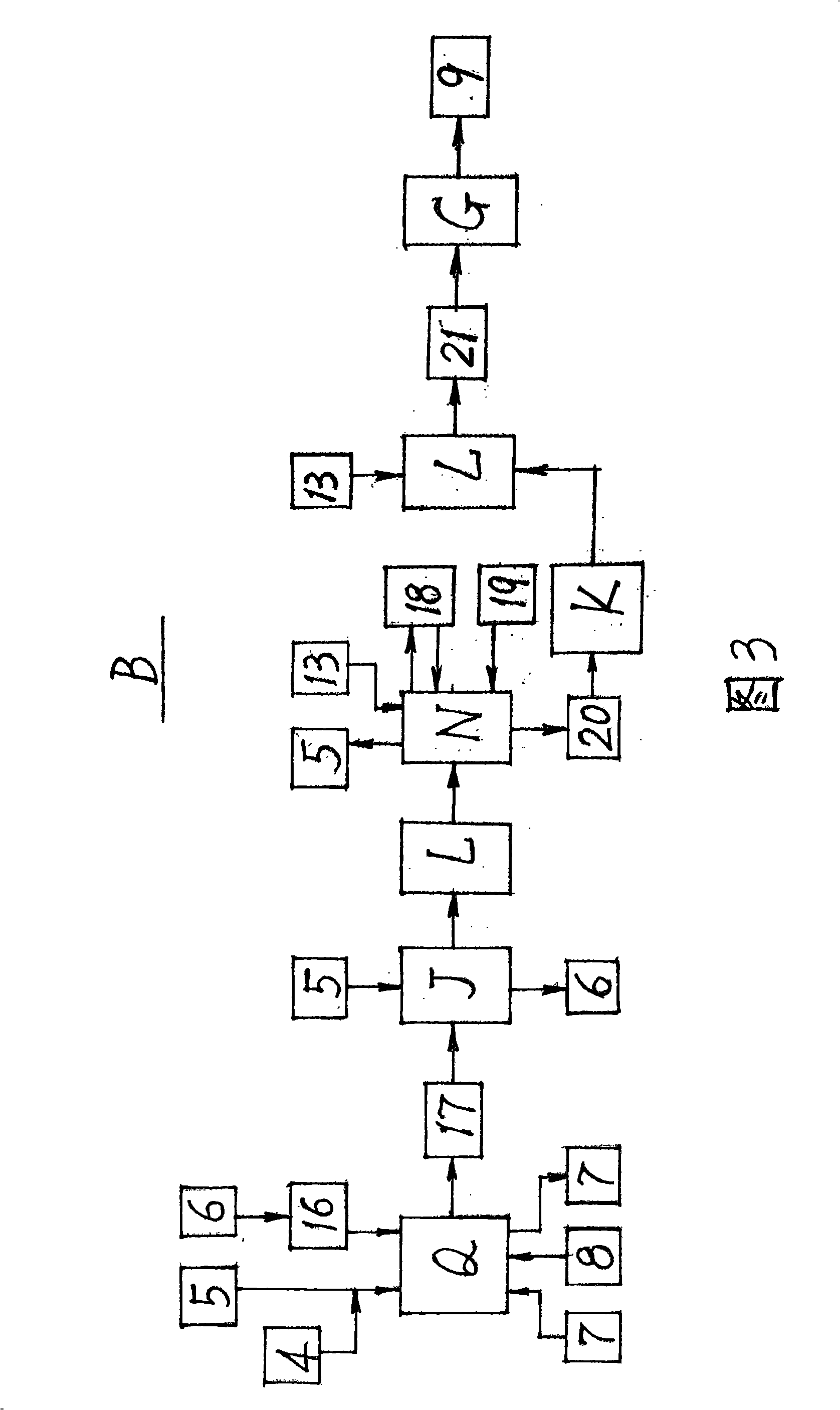
A preparation method for ursodeoxycholic acid regards chenodeoxycholic acid (1) as raw materials; in technological processes, after adopting corresponding equipment and adding excipient according to the proportional variety and dosage, chemical reactions are carried out through the process flow of an oxidation process (A), a hydrogenation process (B), and a purification process (C) in particular temperature and environmental conditions. In the oxidation process (A), an oxidant (3) and a solvent I 2 are added for oxidation reaction to generate an intermediate (4); in the hydrogenation process (B), a catalyst (6) and a solvent II(5) are added for hydrogenation reaction to get ursodeoxycholic acid crude products (9); in the purification process (C), a passivating agent (11) and a solvent III (10) are added for producing the products of ursodeoxycholic acid (12). The process route of the invention is reasonable; the chemical reactions are mild and safe; the operating conditions are easy to be controlled; the yield is high and the invention makes great progress in the research and production of artificially synthesizing ursodeoxycholic acid (12).
Owner:SHANDONG AOCTER CHEM
Composition and preparation for resisting ageing and improving male energy, preparation method of preparation and application of composition
InactiveCN109045059AReasonable compositionAppropriate compatibilityOrganic active ingredientsInorganic active ingredientsSexual functionProtopanaxadiol
The invention discloses a composition for resisting ageing and improving male energy. The composition is prepared from a raw material and an auxiliary material; the raw material is prepared from the following components in parts by weight: 1-15 parts of nicotinamide mononucleotide, 1-10 parts of protopanoxadiol, 1-9 parts of icarisid I, 2-8 parts of baohuoside I, 3-7 parts of dehydroepiandrosterone and 2-10 parts of ursodesoxycholic acid. The composition is reasonable in composing prescription and proper in compatibility; in the composition, the four of the nicotinamide mononucleotide (NMN), the icarisid I, the baohuoside I and protopanoxadiol are used as monarch drugs together, the composition is capable of comprehensively regulating gonad axis and invigorating kidney-yang and liver, candelay ageing period of the gonad axis and improve the sexual function after being taken for a long time; through intercoordination and synergistic effect of various components, the composition is comprehensive in nutrition, has very good palatability, is particularly suitable for males to take and thus relieving the symptoms of male ageing syndrome and improving the male energy. The invention alsoprovides a preparation containing the composition and a preparation method of the preparation. The preparation method is simple, is moderate in condition and is suitable for industrial batch production.
Owner:HOBOOMLIFE BIO TECH SHENZHEN CO LTD
Hydroxysteroid dehydrogenase and application thereof in synthesis of ursodeoxycholic acid precursor
ActiveCN110387360AImprove stabilityIncreased space-time yieldBacteriaMicroorganism based processesCholic acidMutant
The present invention relates to a hydroxysteroid dehydrogenase and an application thereof in synthesis of an ursodeoxycholic acid precursor, and specifically discloses a 12-alpha-hydroxysteroid dehydrogenase derived from rhodococcus ruber and a mutant thereof, encoding gene and amino acid sequences, a recombinant expression vector containing the gene, a recombinant expression transformant, and anapplication of the hydroxysteroid dehydrogenase catalyzing oxidation of 12-alpha-hydroxysteroids to form 12-carbonyl steroids. Compared with the prior art, a used recombinant steroid dehydrogenase-catalyzed oxidation reaction is NAD+coenzyme-dependent type. The hydroxysteroid dehydrogenase has advantages of low application cost, simple operation, mild reaction conditions, environmental friendliness, high yield, etc., and thus has a very good prospect in an application of cholic acid as a raw material for production and preparation of the ursodeoxycholic acid precursor.
Owner:EAST CHINA UNIV OF SCI & TECH +1
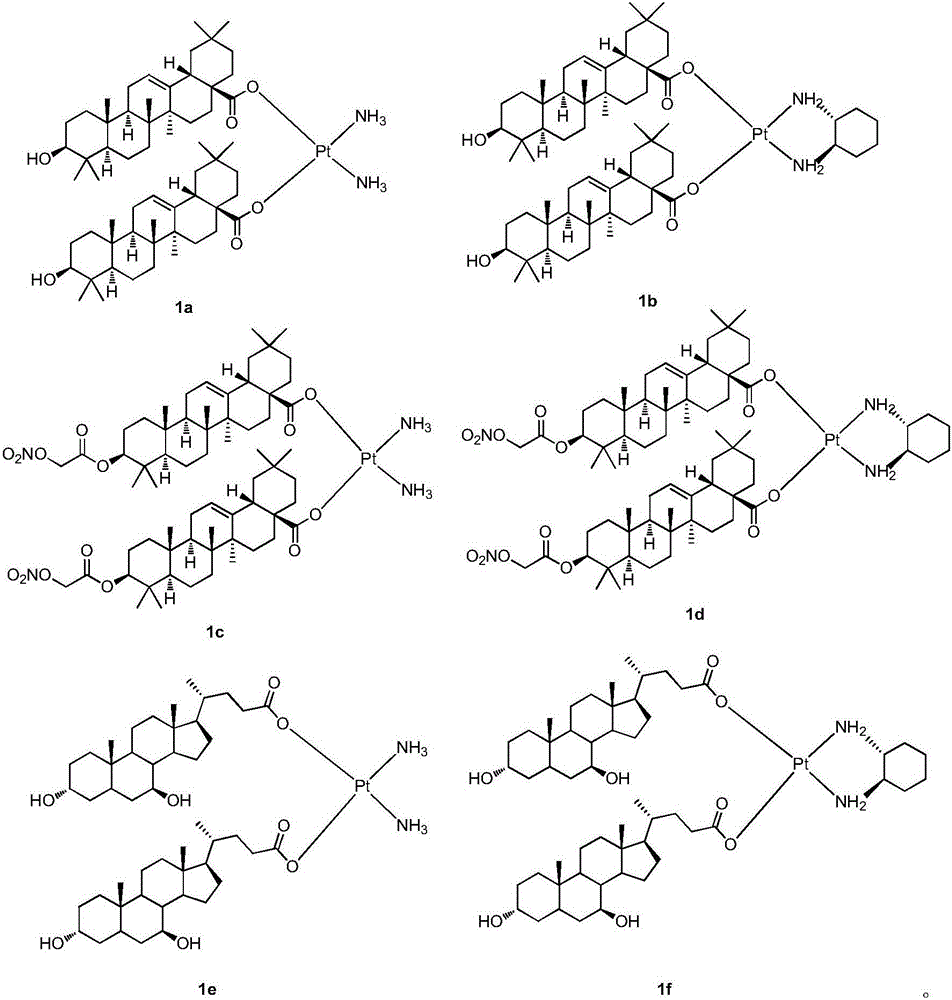
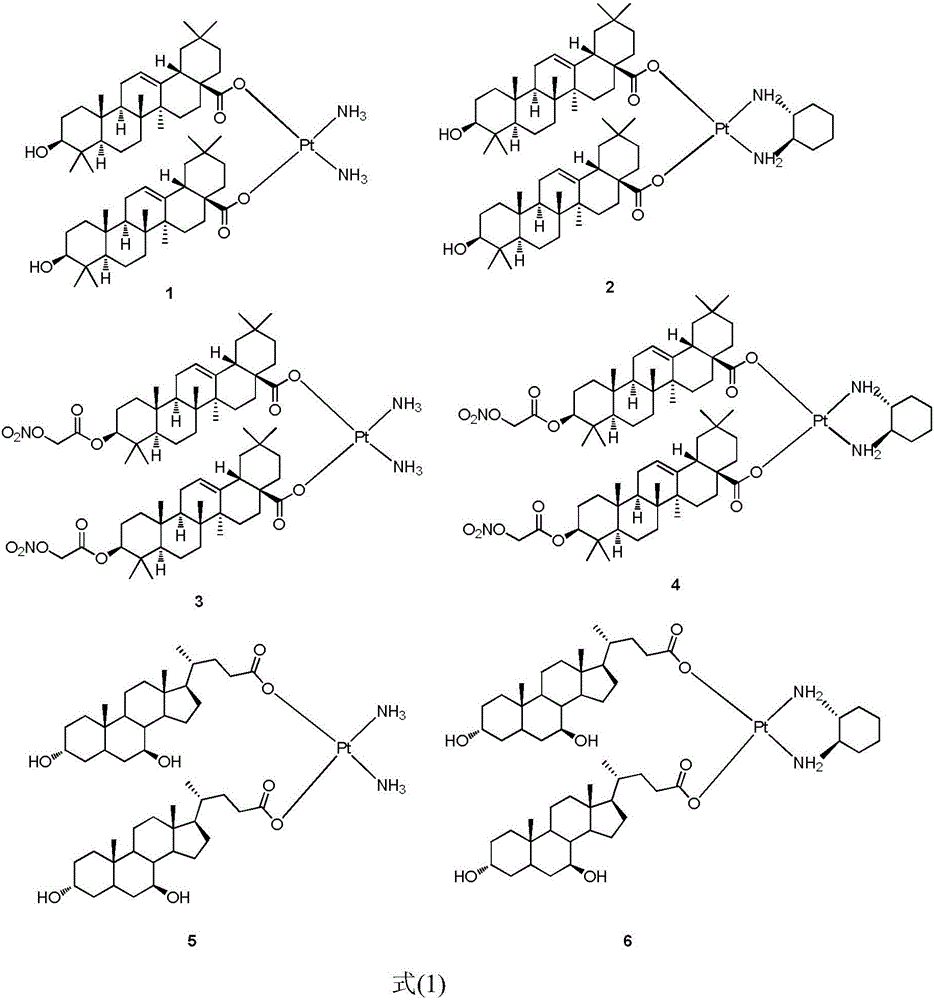
Platinum (II) coordination complex and preparing method and application thereof
ActiveCN105713047AHas antitumor potentialGood in vitro inhibitory activityPlatinum organic compoundsAntineoplastic agentsPotassium tetrachloroplatinateOxygen
The invention discloses a platinum (II) coordination complex and a preparing method and application thereof. The preparing method includes the steps that active natural products such as oleanolic acid and ursodesoxycholic acid and derivatives of the active natural products serve as ligand; potassium tetrachloroplatinate(II) and ammonia or (R,R)-cyclohexanediamine are subjected to coordination reaction under the condition of potassium iodide, then iodide ions are removed through silver nitrate under the light shielding condition, and the mixture and sodium salt of ursodesoxycholic acid or oleanolic acid or the derivatives of oleanolic acid are reacted to obtain the product. The platinum (II) coordination complex has the good in-vitro antitumor activity, and can be applied to preparing medicine for treating malignant tumor.
Owner:SOUTHEAST UNIV
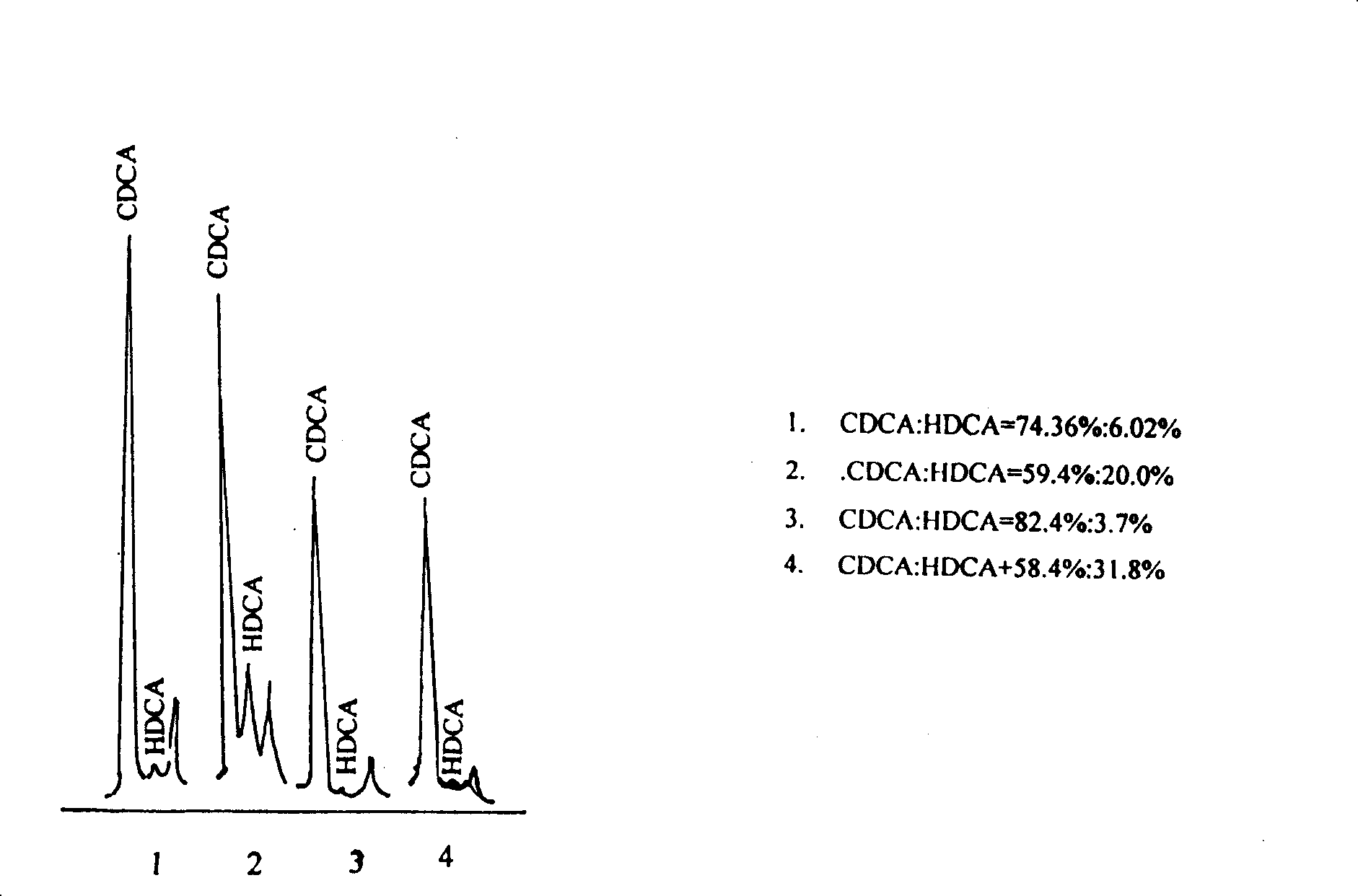
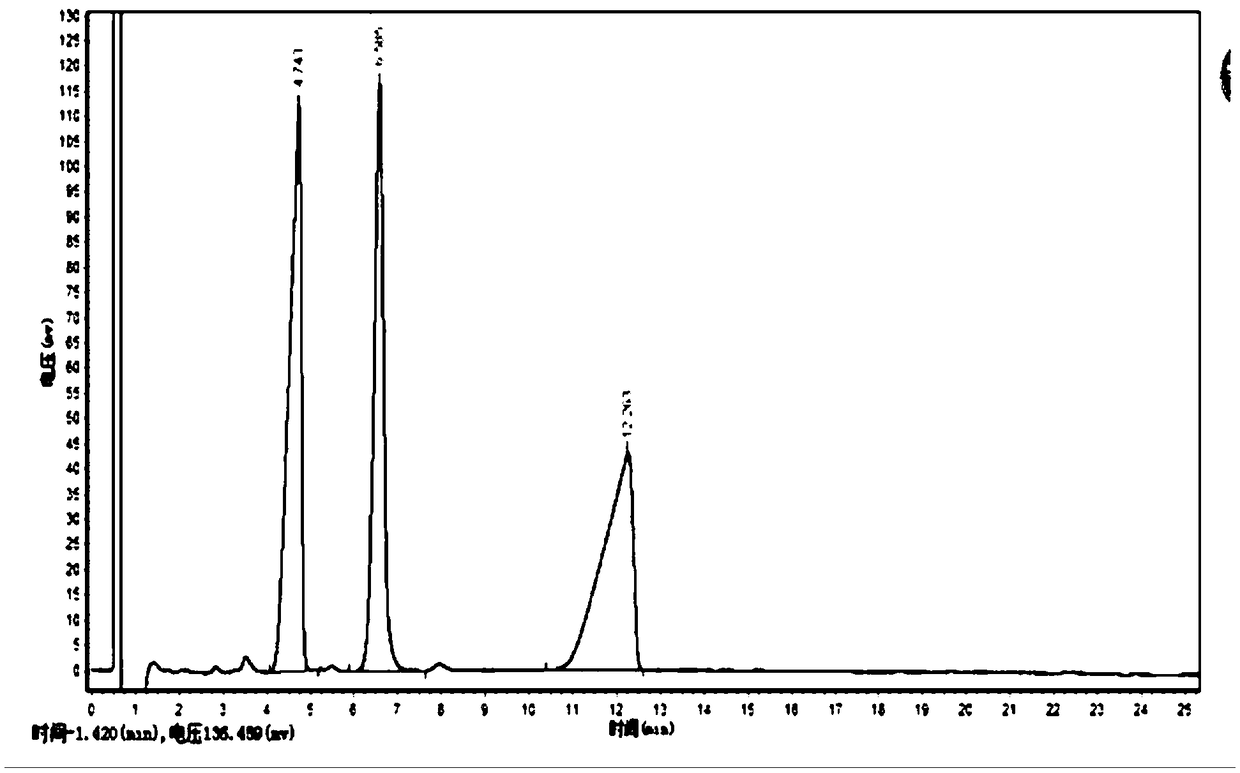
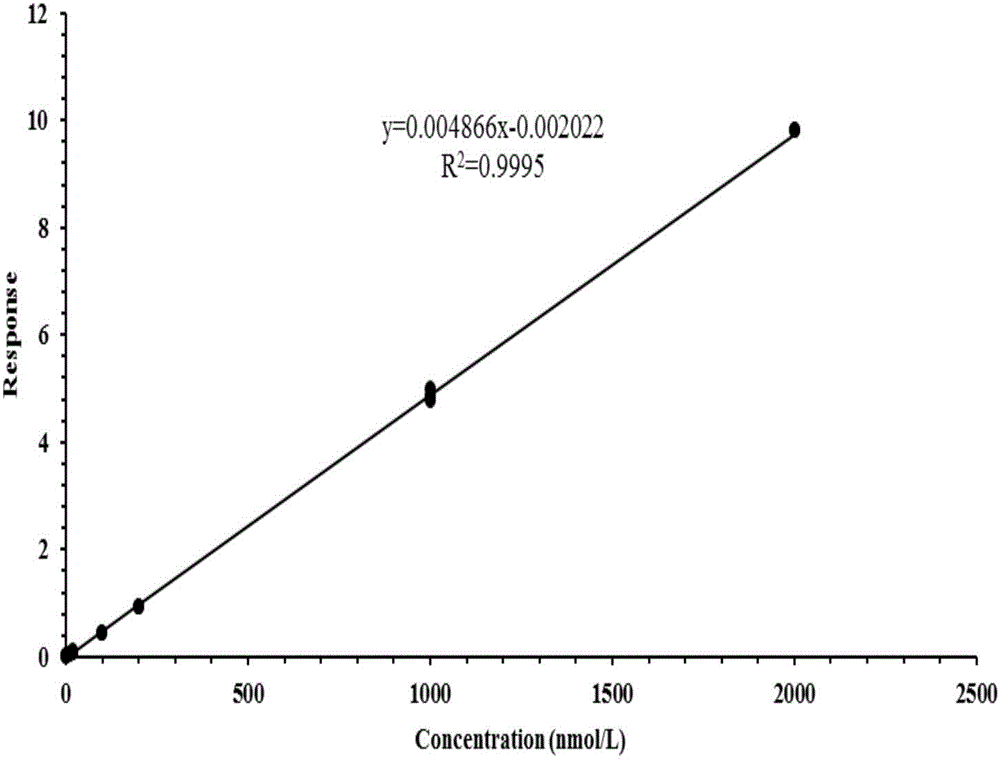
Method for detecting five free bile acids in serum through high performance liquid chromatography-tandem mass spectrum
InactiveCN106841492AEliminate distractionsSimplified processing stepsComponent separationCholic acidTandem mass spectrometry
The invention discloses a method for detecting five free bile acids in serum through high performance liquid chromatography-tandem mass spectrum. The five free bile acids comprises a cholic acid, a lithocholic acid, a deoxycholic acid, a ursodeoxycholic acid and a chenodesoxycholic acid; the method comprises the following steps of preparing a standard substance, performing high performance liquid chromatography separation, performing tandem mass spectrum detection, and detecting the free bile acids in the serum. The method can be used to simultaneously perform determination and accurately quantify the content of the free bile acids in human serum, the detection time is short, the flux is high, the detection sensitivity is high, and the specificity is good.
Owner:HANGZHOU BAICHEN MEDICAL LAB CO LTD
Chenodeoxycholic acid and preparation method thereof
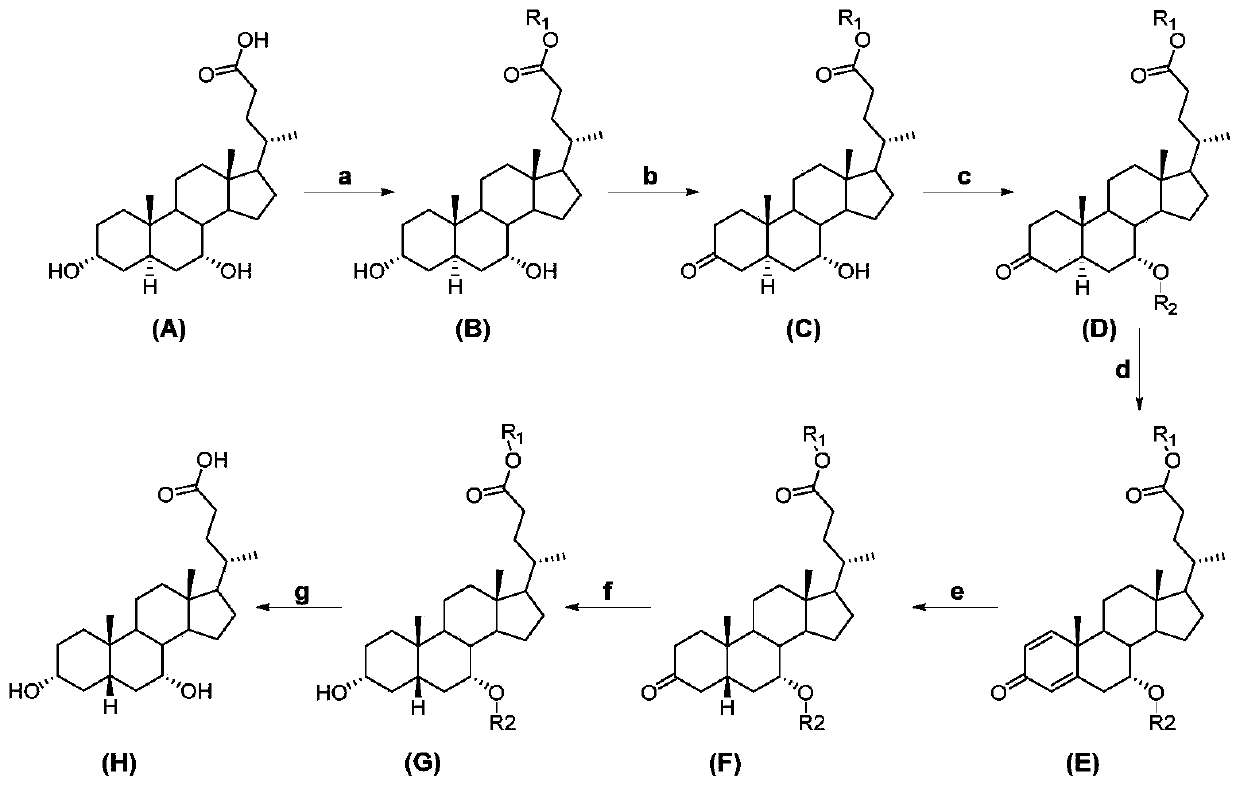
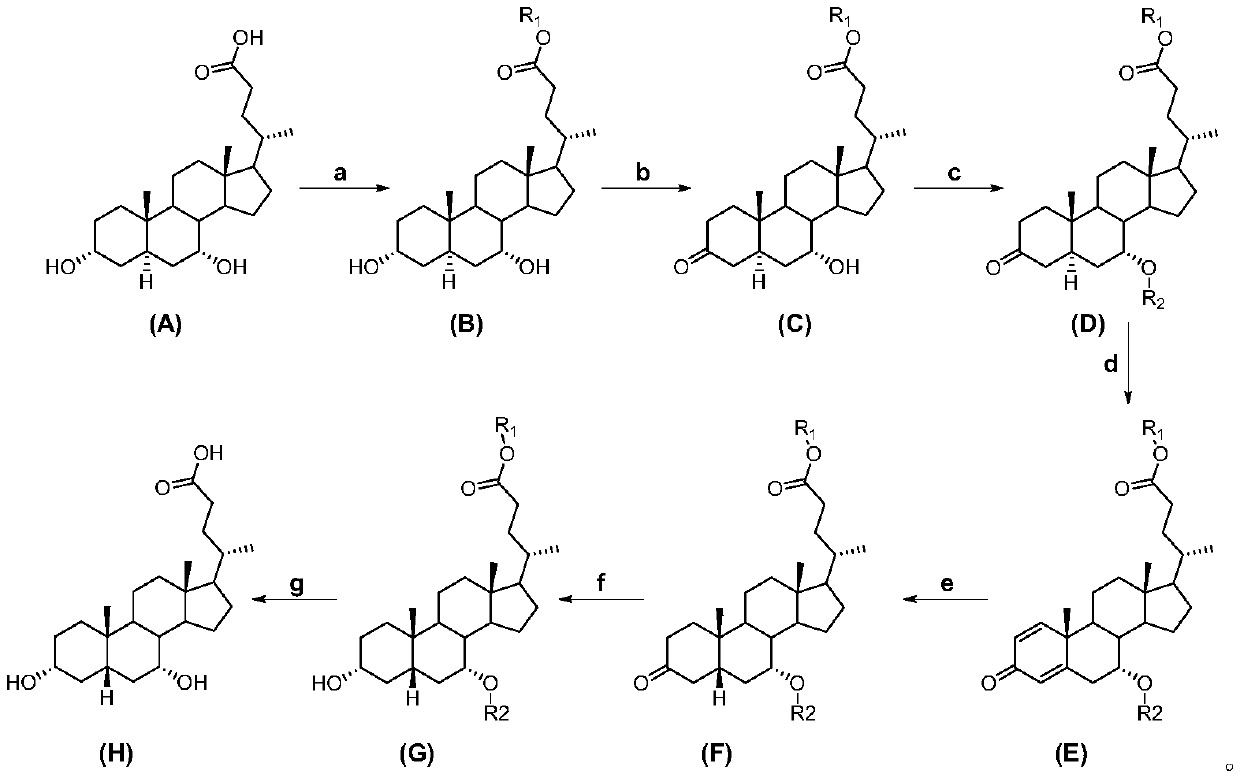
The invention relates to the field of medicine synthesis, and in particular to chenodeoxycholic acid and a preparation method thereof. The preparation method of the chenodeoxycholic acid provided by the invention comprises the following steps of: using 3 alpha,7 alpha-dihydroxy-5 alpha-ursodeoxycholic acid as raw materials to form an intermediate compound E through chemical reaction; then, performing chemical reaction on the intermediate compound E to form the chenodeoxycholic acid. The structure formula of the intermediate compound E is shown in the description, wherein R1 is alkyl, alkenylor aromatic group; R2 is acyl. The preparation method has the advantages that extracted byproducts can be used as raw materials for fast synthesizing the chenodeoxycholic acid; each step in the process is mild in condition; the yield is high; the method is suitable for mass preparation.
Owner:SOUTH CHINA UNIV OF TECH +1