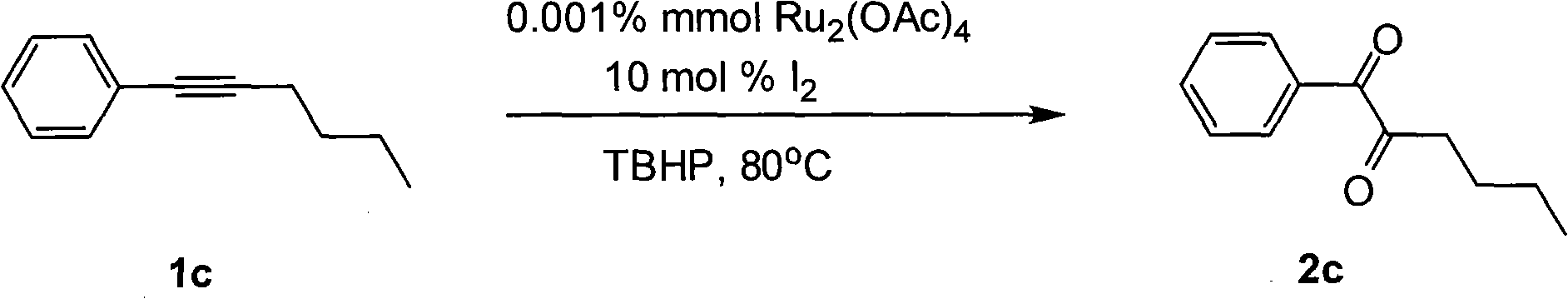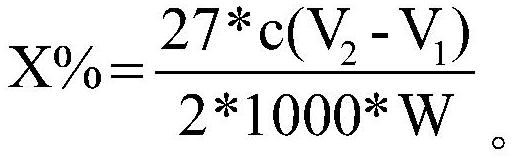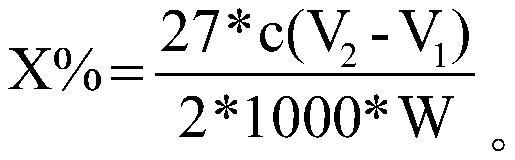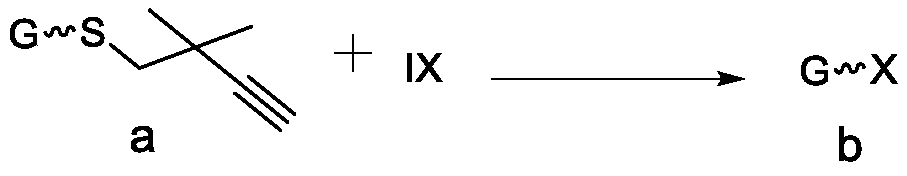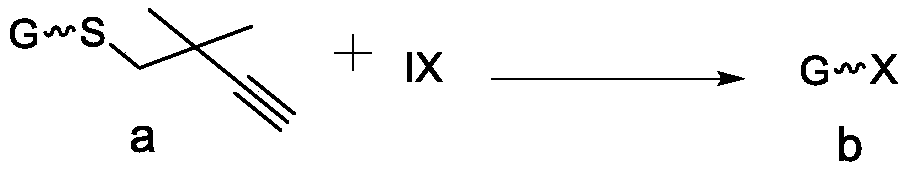Patents
Literature
33 results about "Iodine bromide" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
Bromide is a toxic halide that competes with the absorption of iodine in the body. When the body is in an iodine deficient state it attempts to substitute other halides such as bromide and fluoride to fit its needs.
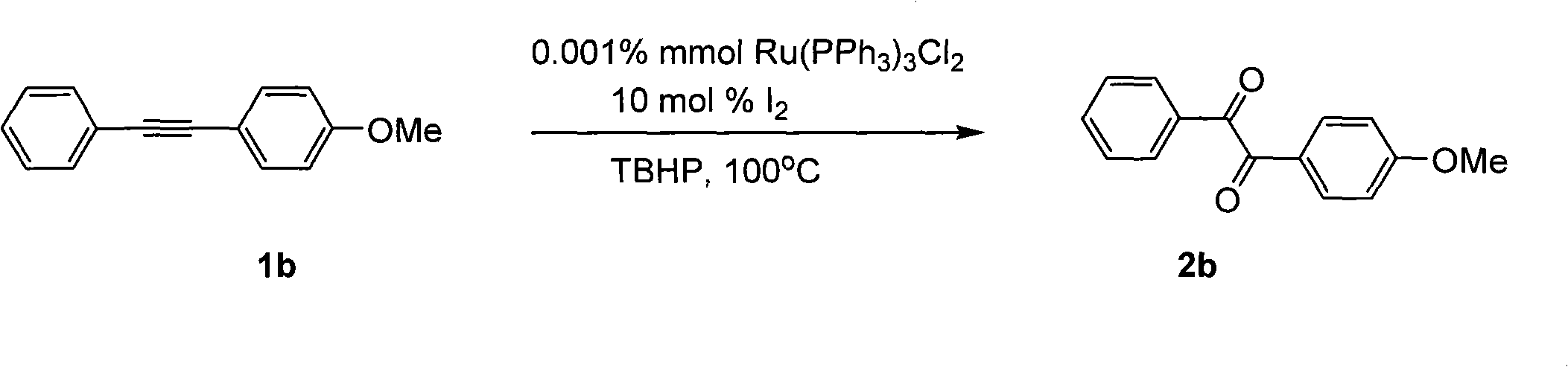
Method for preparing 1, 2-diketone by catalyzing and oxidizing alkynes
InactiveCN101624322AMild reaction conditionsMeet the requirementsCarboxylic acid nitrile preparationOrganic compound preparationCatalytic oxidationKetone
The invention belongs to the field of catalysis and oxidization, and particularly discloses a method for preparing 1, 2-diketone by catalyzing and oxidizing alkynes. The method comprises the following steps: taking alkynes R1-C-C-R2 (acetylenic link exists between C and C) as a reaction substrate, taking one of TBHP, m-chloroperoxybenzoic acid and p-benzoquinone as a oxidant, taking one of dichloro (p-cymene) ruthenium (II) dimer, tri (triphenylphosphine) ruthenous chloride, ruthenium acetate, ruthenium dichlorophenyl (II) dimer, ruthenium trichloride, BINAP ruthenous chloride, dodecacarbonyltriruthenium and tricarbonyldichlororuthenium (II) dimer as a catalyst, taking one of iodine, iodine chloride, iodine bromide, potassium iodide, tetrabutyl ammonium iodide and potassium bromide as a cocatalyst, and taking 1, 4-dioxane as a solvent to react under 40 DEG C to 100 DEG C for 4 to 24 h to prepare the 1, 2-diketone. The method is economic, environmental-friendly and mild.
Owner:SUZHOU UNIV
Composite polyamide reverse osmosis membrane showing high boron rejection and method of producing the same
InactiveUS20070227966A1Increased rejectGeneral water supply conservationSeawater treatmentReverse osmosisPolyamide
A composite polyamide reverse osmosis membrane. The membrane includes a microporous support and a polyamide layer disposed over the microporous support, the polyamide layer including iodine atoms covalently bonded thereto. The membrane is preferably prepared by a process that includes the steps of providing a microporous support, forming a polyamide layer over the microporous support, and treating the polyamide layer with an aqueous solution comprising a compound, the compound comprising at least one iodine atom. Examples of the compound comprising at least one iodine atom include molecular iodine, iodine monobromide, iodine monochloride and iodine trichloride. The iodine-containing compound may be added to an aqueous solution and dissolved therein or may be formed in situ in the aqueous solution, for example, by adding to the aqueous solution an iodide salt and an oxidizing agent.
Owner:WOONGJIN CHEM
Preparation method of efficient adsorbent for gaseous mercury sampling
ActiveCN103977763AImprove the capture effectReduce escape rateOther chemical processesWithdrawing sample devicesActivated carbonHalogen
The invention relates to a preparation method of an efficient adsorbent for gaseous mercury sampling, and particularly relates to a preparation method of the efficient adsorbent for gaseous mercury sampling. According to the invention, except halogen and compounds thereof, iodine bromide and dimercaptosuccinic acid are used for modifying; by relevant experiment, the result shows that activated functional groups of carbonyl, halogen and the like which are increased on the surface of modified activated carbon are beneficial to chemical adsorption of the element mercury and divalent mercury, and especially a symmetrical stable structure formed by the combination of dimercaptosuccinic acid and mercury enables the adsorption capability of the modified activated carbon on mercury to be improved greatly.
Owner:WUHAN TIANHONG INSTR
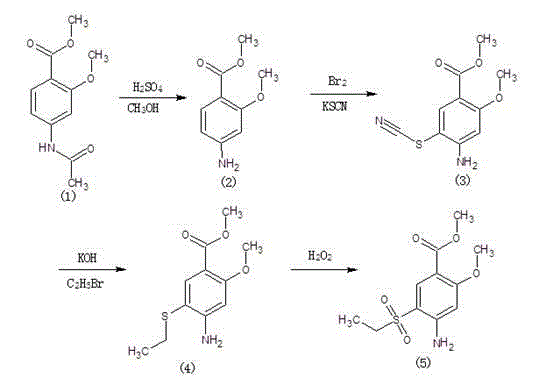
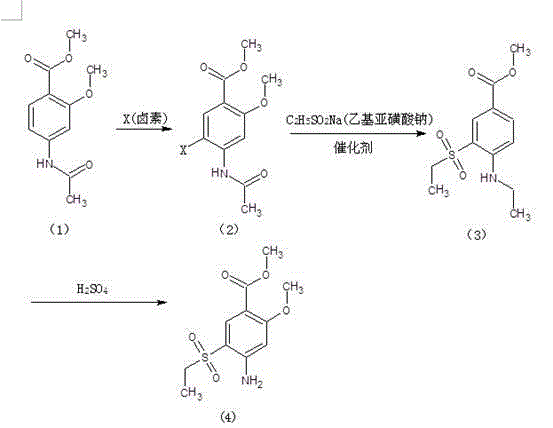
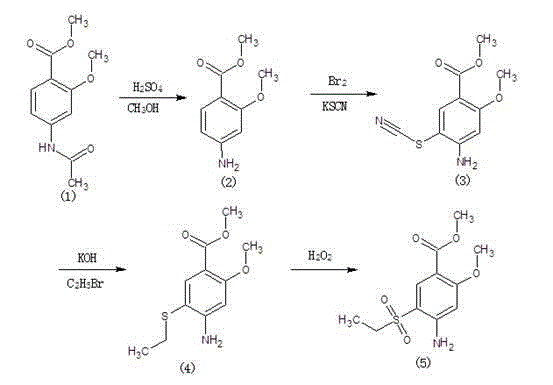
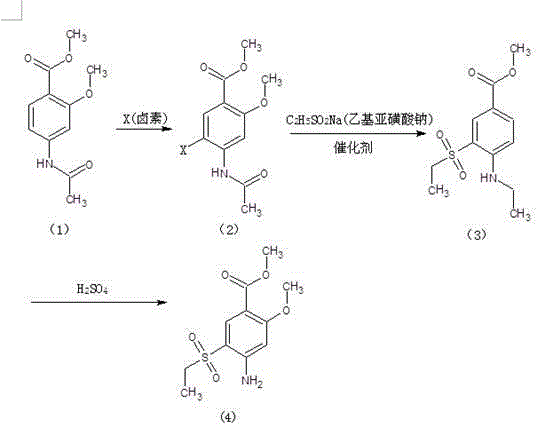
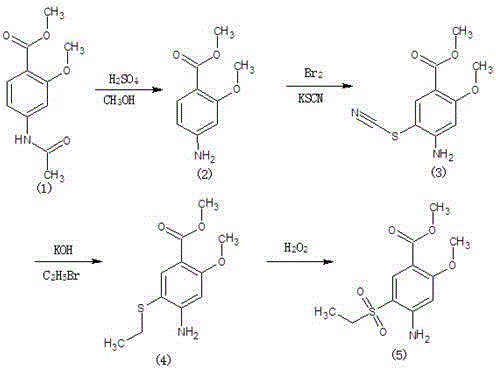
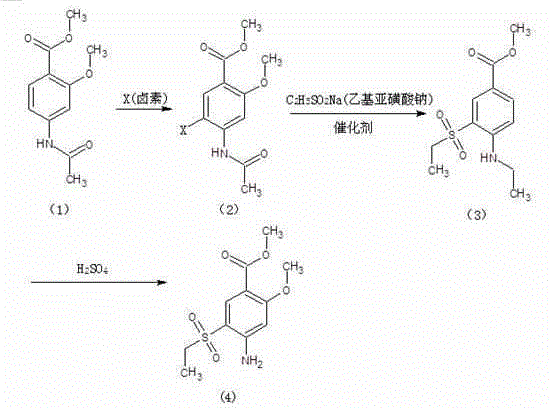
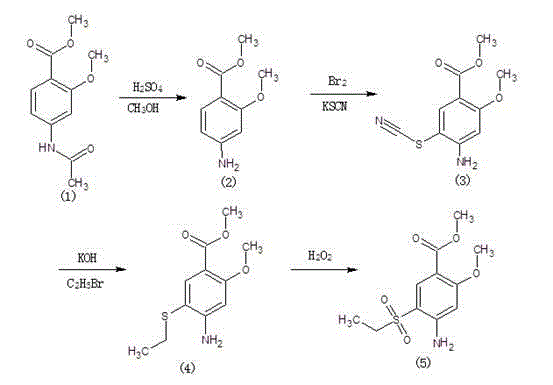
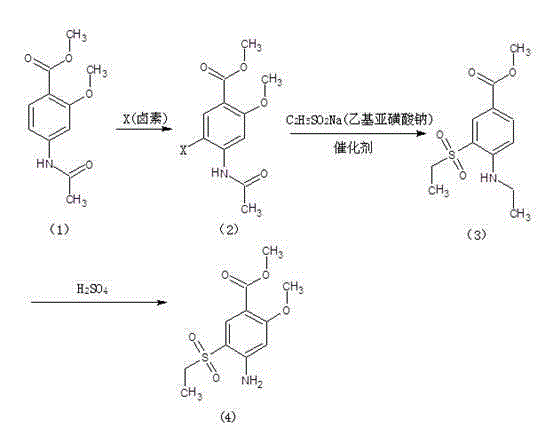
Method for synthesizing 2-methoxyl-4-amino-5-ethylsulfonyl methyl benzoate by utilizing halogenation of halogen
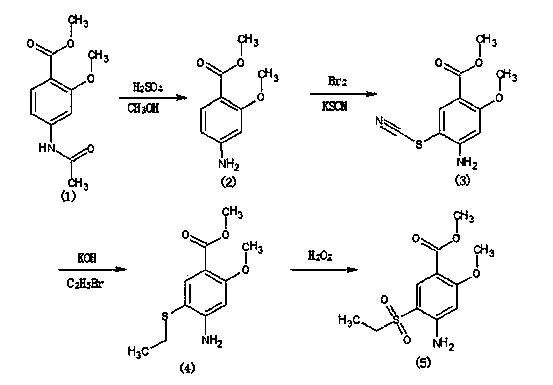
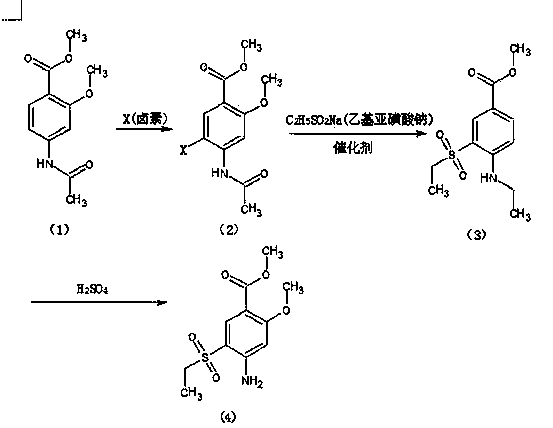
The invention relates to a method for synthesizing 2-methoxyl-4-amino-5-ethylsulfonyl methyl benzoate by utilizing halogenation of halogen. The method is characterized by comprising the following steps: halogenating (chloridizing, brominating and iodinating) 2-methoxyl-4-acetamino methyl benzoate through halogen, and then carrying out condensation with ethyl sodium sulphinate to obtain a target product. The method has the advantages that the process route is simple, the product yield is high, the total yield can reach 80 percent, the product quality is good, the purity can reach 99.5 percent, and the appearance is white.
Owner:苏州诚和医药化学有限公司
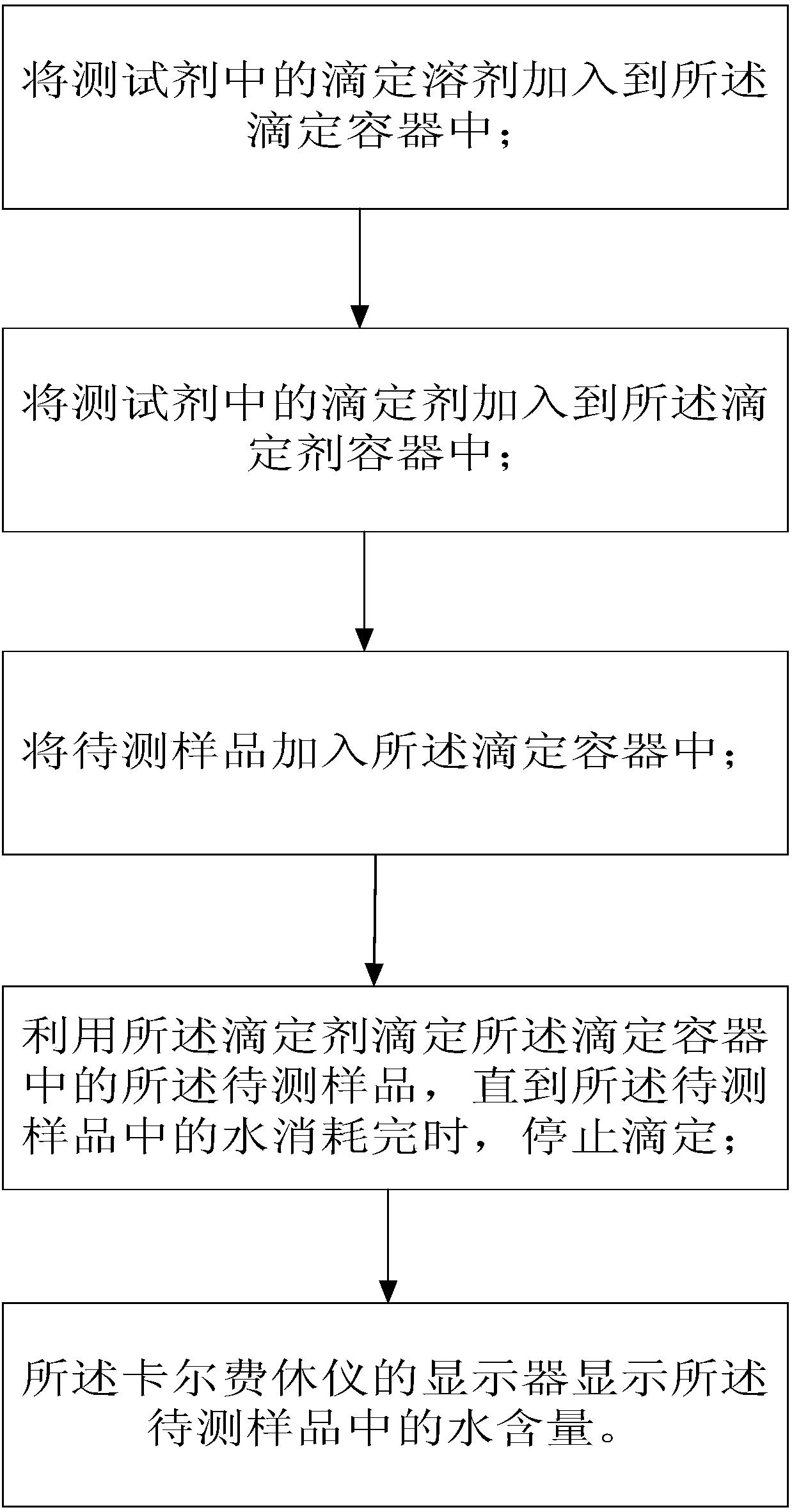
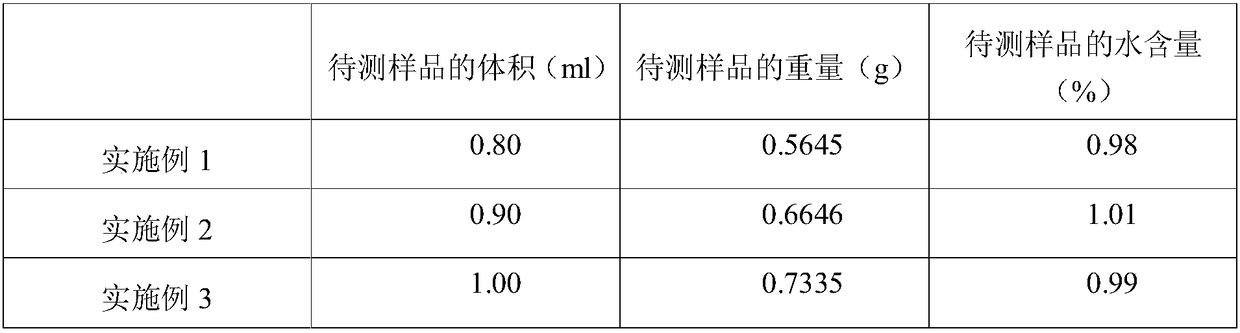
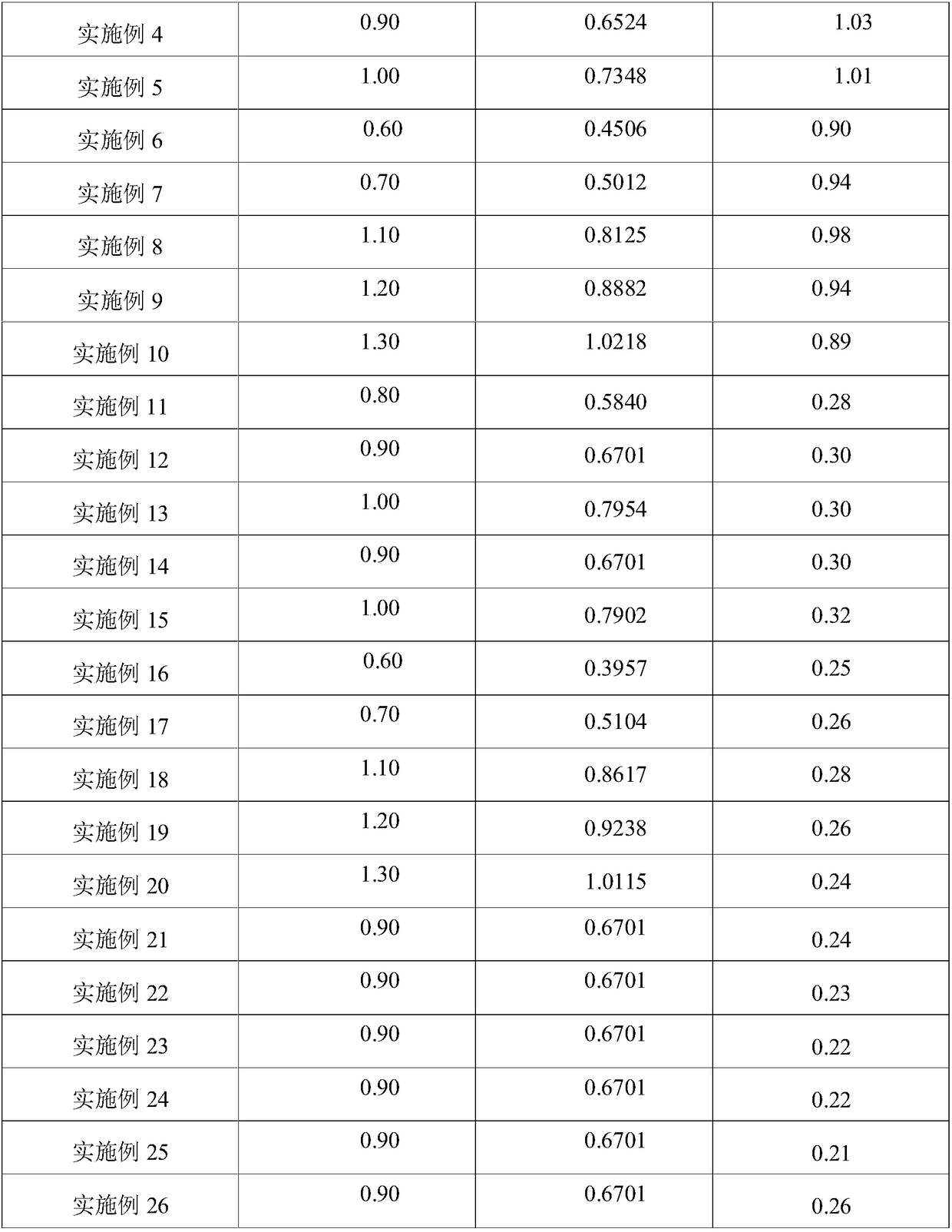
Test agent and test method for water content in aldehyde ketone
InactiveCN108426977AReached contentJudgment contentChemical analysis using titrationAlcoholTest agent
The invention provides a test agent and a test method for the water content in aldehyde ketone. The test agent comprises a titration solvent and a titrant, wherein the titration solvent comprises methoxy alcohol, imidazole and iodine bromide; the titrant comprises oxydiethanol, iodine, hydroiodic acid, imidazole, an imidazole derivative and sulfur dioxide. The test agent can quickly detect the water content in aldehyde ketone, greatly saves the test time, and avoids a problem that the system is slow or unable to achieve balance due to the conventional alcohol reacts with aldehyde ketone in a sample to be tested.
Owner:CHINA SHENHUA ENERGY CO LTD +2
Gardenia soilless culture nutrient formula and application thereof
InactiveCN104961586AMeet the needs of floating seedlingsLower control costsFertilizer mixturesZinc selenideTyrosine
The invention discloses gardenia soilless culture nutrient formula and application thereof. The gardenia soilless culture nutrient formula consists of 13-17 parts of ammonium hydrogen carbonate, 11.6-12.6 parts of ammonium ferric sulfate, 2-20 parts of auxin, 4-6 parts of tyrosine, 4-6 parts of serine, 0.4-0.6 part of palladium nitrate, 0.1-0.3 part of zinc selenide, 0.1-0.3 part of iodine bromide, 0.08-0.2 part of sodium methylate and the balance of water which are weighed according to the proportions and are uniformly mixed. By adoption of a nutrient solution, the seepage force and the diffusivity of the nutrient solution are greatly improved during seedling, so that the seeding stage is greatly shortened, the emergence rate is increased, and the quality of seedlings is improved; meanwhile, the cost is reduced; furthermore, the nutrient solution is more suitable for high-altitude and low-temperature seedling.
Owner:汤在英
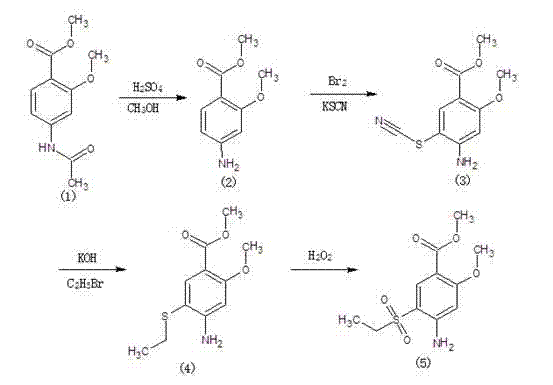
Synthetic method of 2-methoxyl-4-amino-5-ethyl sulfuryl methyl benzoate
The invention relates to a synthetic method of 2-methoxyl-4-amino-5-ethyl sulfuryl methyl benzoate. The synthetic method comprises the following steps: carrying out halogenation (chlorination, bromination and iodization) pm 2-methoxyl-4-acetamido methyl benzoate by halogens; and then, condensing the halogenated 2-methoxyl-4-acetamido methyl benzoate with ethyl sodium sulfinate to obtain a target product. The synthetic method has the advantages of simple process line, high product yield (the total yield reaches 80%) and good product quality (the purity reaches 99.5%), and the 2-methoxyl-4-amino-5-ethyl sulfuryl methyl benzoate is white in appearance.
Owner:苏州诚和医药化学有限公司
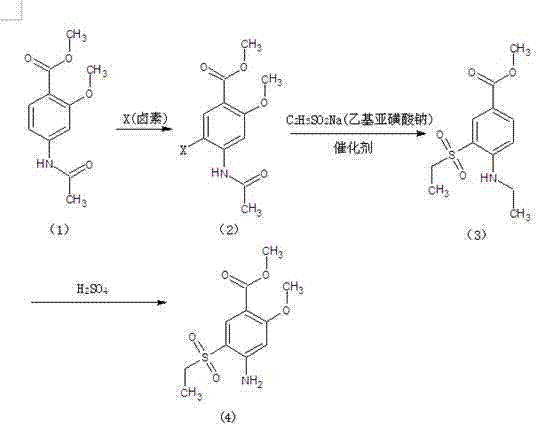
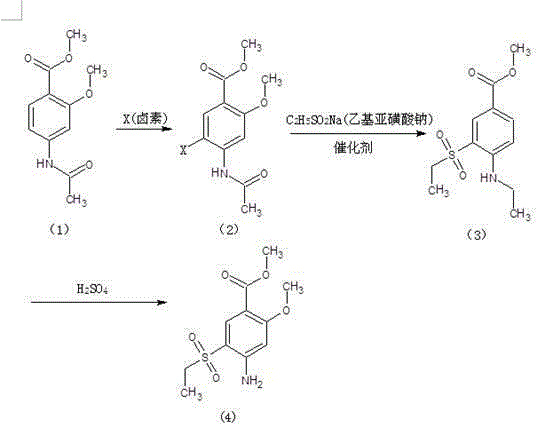
Method for preparing 2-methoxy-4-amino-5-ethysulfonyl benzoic acid methyl ester by halogen halogenation
The invention relates to a method for preparing 2-methoxy-4-amino-5-ethysulfonyl benzoic acid methyl ester by halogen halogenation. According to the invention, 2-methoxy-4-acetyl methyl anthranilate is subjected to halogen halogenation (chlorination, bromination or iodization) and then is condensed with sodium ethanesulfinate to obtain a target product. The method has the advantages that the process route is simple, the product yield is high, the total yield reaches 80 percent, the product quality is high, the purity reaches 99.5 percent, and the appearance is white.
Owner:苏州诚和医药化学有限公司
A kind of preparation method of high-efficiency adsorbent for gaseous mercury sampling
ActiveCN103977763BImprove the capture effectReduce escape rateOther chemical processesWithdrawing sample devicesActivated carbonHalogen
The invention relates to a preparation method of an efficient adsorbent for gaseous mercury sampling, and particularly relates to a preparation method of the efficient adsorbent for gaseous mercury sampling. According to the invention, except halogen and compounds thereof, iodine bromide and dimercaptosuccinic acid are used for modifying; by relevant experiment, the result shows that activated functional groups of carbonyl, halogen and the like which are increased on the surface of modified activated carbon are beneficial to chemical adsorption of the element mercury and divalent mercury, and especially a symmetrical stable structure formed by the combination of dimercaptosuccinic acid and mercury enables the adsorption capability of the modified activated carbon on mercury to be improved greatly.
Owner:WUHAN TIANHONG INSTR
Nutrient formula for soilless culture of orchids and application of nutrient formula
InactiveCN104961588AIncrease resistanceStrong resistanceFertilizer mixturesIron sulfateNutrient solution
The invention discloses a nutrient formula for the soilless culture of orchids. The nutrient formula comprises the following components in parts by weight: 13 to 20 parts of ammonium sulfamate, 11 to 13 parts of ammonium iron sulfate, 2 to 20 parts of polyamine, 4 to 5 parts of leucine, 4 to 5 parts of proline, 0.4 to 0.6 part of palladium nitrate, 0.1 to 0.3 part of barium molybdate, 0.1 to 0.3 part of iodine bromide, 0.06 to 0.2 part of calcium hypochlorite and the balance of water. A nutrient solution is obtained by weighing the components according to the proportion and uniformly mixing the components. The seepage force and the diffusivity of the nutrient solution are greatly enhanced when seedlings are cultured, so that the period of emergence is greatly shortened, the rate of emergence is increased, the emergence quality is improved, and in addition, the cost is reduced; moreover, the nutrient solution is more applicable to high-altitude low-temperature seedling culture.
Owner:汤在英
A method for detecting the vinyl content of low vinyl methyl vinyl silicone rubber
ActiveCN107664608BAvoid intractable disadvantagesDirect detection methodInvestigating material hardnessPolymer scienceVulcanization
The invention relates to a method for detecting the vinyl content of low vinyl methyl vinyl silicone rubber (referred to as "110 silicone rubber"). Molding, testing the Shore hardness, and obtaining the vinyl content according to the Shore hardness. The traditional detection method is the iodine bromide method, that is, the addition color reaction. This method is cumbersome to operate, takes a long time to detect (5 hours), and has a large detection deviation (±0.02%), especially for low vinyl content of less than 0.06%. Base 110 silicone rubber cannot be effectively identified. The new method for detecting the vinyl content of 110 silicone rubber is the application method, that is, the network structure is formed by vulcanization and cross-linking, and the vinyl content is reflected by the hardness. This method has the advantages of rapid evaluation, simple operation and direct application, and can more intuitively guide customers to use the product.
Owner:HUBEI XINGRUI SILICON MATERIAL CO LTD
Photocatalyst powder composite deodorizing-type adsorption filtering agent and manufacturing method thereof
InactiveCN104841392AExtended service lifeLarge specific surface areaDispersed particle filtrationOther chemical processesPorosityEpoxy
The invention discloses a photocatalyst powder composite deodorizing-type adsorption filtering agent which is characterized by being prepared from the following raw materials, by weight, 55-65 parts of rectorite, 24-35 parts of diatom ooze, 10-14 parts of soda lime, 1-2 parts of magnesium sulfate, 4-8 parts of a photocatalyst powder, 3.4-3.8 parts of rough-hole spherical silica gel, 2-3 parts of iodine bromide, 0.6-0.9 part of sodium dimercaptosuccinate, 5-7 parts of an epoxy resin emulsion, 6-8% of a hydrogen peroxide solution and a proper amount of water. According to the invention, rectorite, diatom ooze and soda lime are used as raw materials, and iodine bromide, the epoxy resin emulsion and the like are used along with the raw materials. Through a physical and chemical modification method, the photocatalyst powder is loaded on the surface of rectorite. Thus, organic pollutants can be decomposed into carbon dioxide and oxygen so as to reduce harmful substances adsorbed on rectorite and prolong service life of the adsorption filtering agent. The prepared adsorption filtering agent has advantages of rough and hard surface, large specific surface area, high porosity, good adsorption filtering performance and low cost. The adsorption filtering agent can release negative oxygen ions so as to effectively remove foul smell of exhaust gas and purify the air.
Owner:BENGBU DEMO FILTRATION TECH
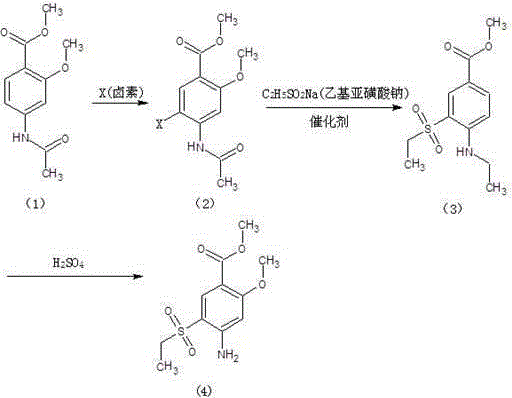
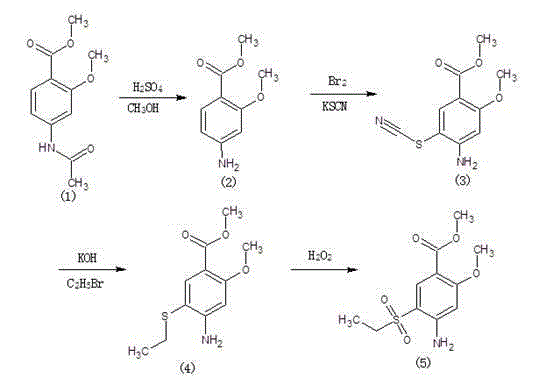
Method for synthesizing 2-methoxy-4-amino-5-ethylsulfone methyl benzoate by halogenation
The present invention relates to a method for synthesizing 2-methoxy-4-amino-5-ethylsulfone methyl benzoate by halogenation with halogen. In the present invention, the target product is condensed with sodium ethyl sulfinate by halogenation (chlorination, bromination, iodination) of 2-methoxy-4-acetylaminobenzoic acid methyl ester with halogen, and its advantage is that the process route is simple, The product yield is high, the total yield reaches 80%, the product quality is good, the purity reaches 99.5%, and the appearance is white.
Owner:苏州诚和医药化学有限公司
A method for preparing methyl 2-methoxy-4-amino-5-ethylsulfone benzoate by halogenation with halogen
The invention relates to a method for preparing methyl 2-methoxy-4-amino-5-ethylsulfone benzoate by halogenation with halogen. In the present invention, the target product is condensed with sodium ethyl sulfinate by halogenation (chlorination, bromination, iodination) of 2-methoxy-4-acetylaminobenzoic acid methyl ester with halogen, and the advantages are that the process route is simple, The product yield is high, the total yield reaches 80%, the product quality is good, the purity reaches 99.5%, and the appearance is white.
Owner:苏州诚和医药化学有限公司
Method for preparing 1-bromo-2-iodotetrafluoroethane
ActiveCN102399127BHigh selectivityEasy to separatePreparation by halogen additionTetrafluoroethyleneHydrogen
The invention discloses a method for preparing 1-bromo-2-iodotetrafluoroethane. In the method, under the existence of organic solvent or absence of solvent, tetrafluoroethylene and iodine bromide are subjected to gas-liquid phase reaction under the initial reaction temperature of 50 DEG C to 80 DEG C and the tetrafluoroethylene gas pressure of 0.1MPa to 2.0MPa, so that the 1-bromo-2-iodotetrafluoroethane is produced, and the organic solvent is C2-C4 fluorine-containing halogenated hydrocarbon or C2-C4 hydrogen fluorin-containing halogenated hydrocarbon, the boiling point of which is higher than the boiling point of the 1-bromo-2-iodotetrafluoroethane. The product selectivity of the method is high, and the product is easy to separate and purify. The purity is about 97 percent, and the price is relatively low.
Owner:CHANGSHU 3F ZHONGHAO NEW CHEM MATERIALS +1
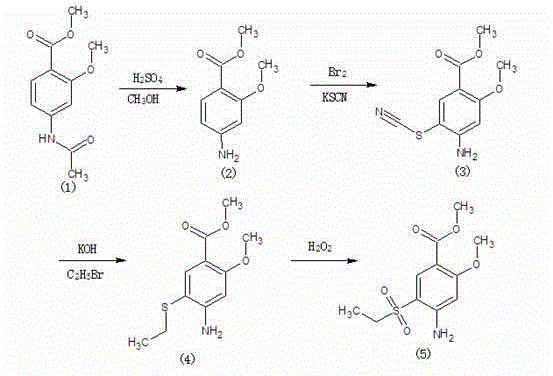
A method for preparing methyl 2-methoxy-4-amino-5-ethylsulfonyl benzoate
The invention relates to a method for preparing 2-methoxy-4-amino-5-ethylsulfonylbenzoic acid methyl ester, which is prepared by halogenating 2-methoxy-4-acetylaminobenzoic acid methyl ester with halogen ( Chlorination, bromination, iodination), and then condense the target product with sodium ethyl sulfinate, which has the advantages of simple process route, high product yield, total yield of 80%, good product quality, purity of 99.5%, and The appearance is white.
Owner:苏州诚和医药化学有限公司
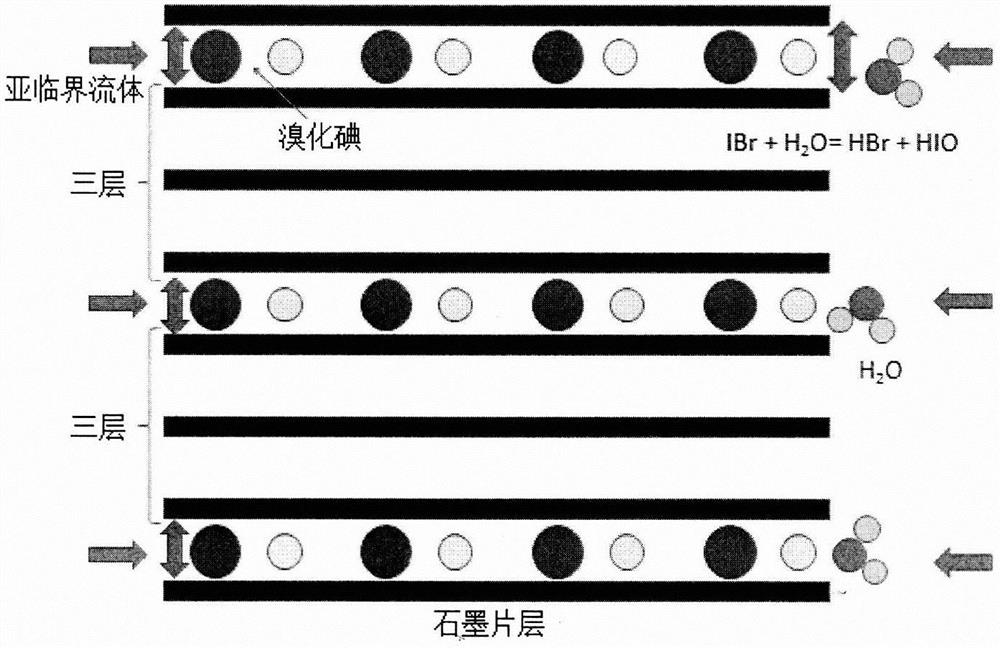
A subcritical reaction preparation method of three atomic layer high-quality graphene materials
InactiveCN108314026BSuitable for operationLower requirementGrapheneReaction temperatureTransparent conducting film
The invention provides a subcritical preparation method of three-atomic-layer high-quality graphene. The subcritical preparation method comprises the following steps: iodine bromide is used as an intercalation agent to prepare a three-order graphene intercalation compound, then the three-order graphene intercalation compound is placed in a subcritical water environment, an intercalation agent (iodine bromide) is dissolved in water and generates violent reaction with water in a graphite layer, the violent reaction and the high-pressure permeation action of the subcritical water environment acton a graphite sheet layer, and by peeling, the graphene material with the three-atomic-layer thickness is obtained. The subcritical preparation method provided by the invention has the beneficial effects that the adopted subcritical reaction temperature is 100-240 DEG C, so that the subcritical preparation method is suitable for industrial or laboratory operation; due to rapid reaction, the subcritical preparation method is suitable for preparing large-laminar graphene materials with three-atomic-layer thickness and can be used for the fields of transparent conducting films, energy storage devices and superconductors and the like.
Owner:射阳县新港污水处理有限公司
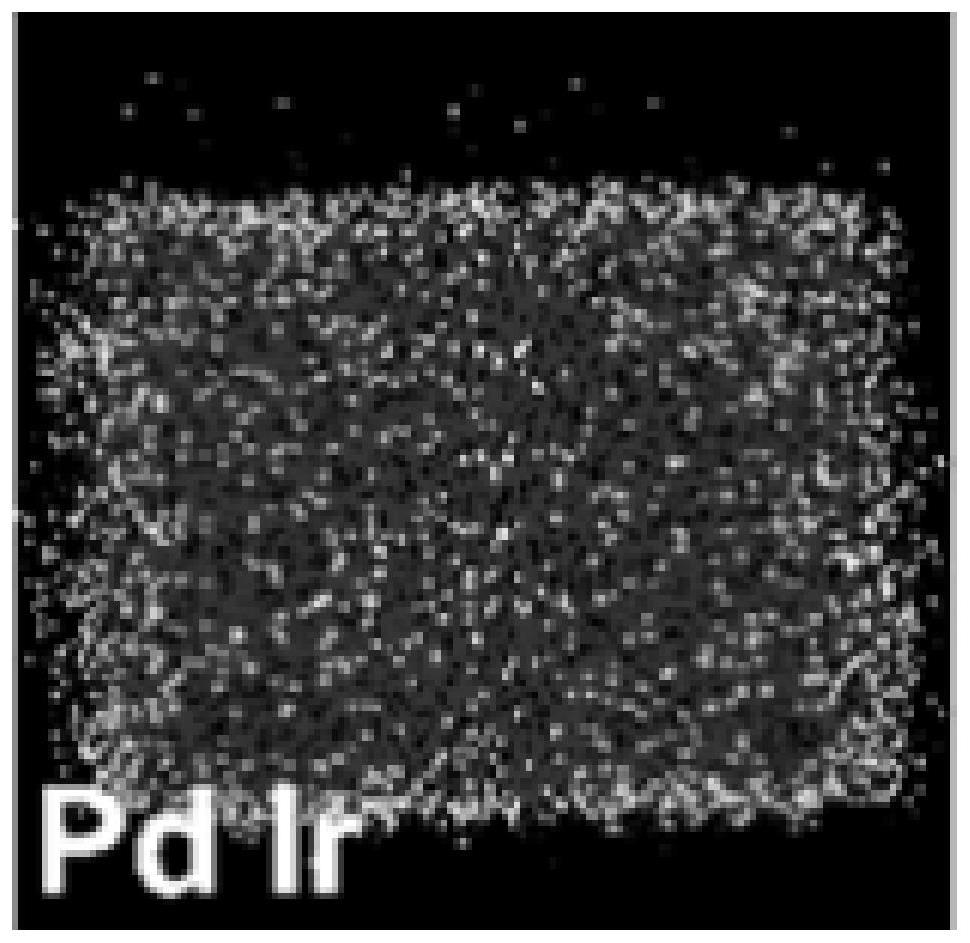
Nanoparticle catalyst with ultrathin platinum metal layer and preparation method of of nanoparticle catalyst
PendingCN113964332AEasy to prepareUniform particle sizeMaterial nanotechnologyCell electrodesPtru catalystPyrrolidinones
The invention discloses a nanoparticle catalyst with an ultrathin platinum metal layer and a preparation method of the nanoparticle catalyst, and belongs to the technical field of nano material preparation. The invention particularly relates to a simple, rapid and one-step synthesis method of ultrathin platinum metal layer nanoparticles with efficient catalytic performance. According to the invention, polyvinylpyrrolidone is used as an end-capping reagent, organic solvents such as triethylene glycol and tetraethylene glycol are used as a solvent and a reducing agent, and potassium bromide or iodine bromide is used as a synthesis blocking agent for platinum metal layer nanoparticles with efficient catalytic activity. The nanometer material with the platinum metal layer and high catalytic activity is synthesized in a green, simple, convenient and rapid hydrothermal synthesis mode, the catalytic activity of the nanometer material is remarkably higher than that of a commercial catalyst, the stability is good, and the catalytic effect is not remarkably changed after the nanometer material is placed for 9 months or above.
Owner:JIANGNAN UNIV
A kind of process of recovering iodine from medical organic waste liquid containing iodine bromide
ActiveCN112028020BNo secondary pollutionEasy to recycleIodineIncinerator apparatusIodideSodium iodide
The invention relates to a process for recovering iodine from medical organic waste liquid containing iodine bromide. First, liquid caustic soda is used to neutralize a large amount of acid in the waste liquid, and liquid caustic soda is used to maintain weak acidity, and sodium sulfite is used to reduce the water layer. The form of sodium enters the water layer, and the aqueous solution containing sodium iodide is incinerated at a high temperature (1000-1100 °C) in an incinerator to obtain an iodine-containing aqueous solution that completely removes organic matter, and then the iodine is extracted by a conventional oxidation iodine process, with a yield of more than 98%. More than 96%; the organic layer is used as a high calorific value fuel as a fuel for an incinerator, and the heat generated is used for production to reduce environmental pollution.
Owner:山东博苑医药化学股份有限公司
Quantitative determination method for carbon-carbon double bonds on surface of powder
InactiveCN112946174ALow costImprove accuracyChemical analysis using titrationDouble bondMolar concentration
The invention relates to a quantitative determination method for carbon-carbon double bonds on the surface of powder, which comprises the steps of weighing Wg of powder, adding into an iodine flask, adding an organic dispersant, and stirring; adding an excessive iodine bromide solution into the iodine flask, stirring, placing in a dark place, adding distilled water and a potassium iodide solution, and uniformly shaking; using a sodium thiosulfate solution with the molar concentration of c mol / L for titration, shaking the iodine flask till the solution becomes light yellow, adding a starch indicator, continuing the titrationtill blue disappears, repeating the experimental group for multiple times, and taking the average volume of the sodium thiosulfate solution consumed each time to be V1 mL; and taking the volume of the sodium thiosulfate solution consumed by the blank control group to be V2mL. The calculation formula of the content X% of the carbon-carbon double bonds is shown in the description. The quantitative determination method for the carbon-carbon double bonds on the surface of the powder is low in cost, high in accuracy, simple and convenient to operate, good in repeatability and suitable for determination of the content of the carbon-carbon double bonds in the polyalkyl silsesquioxane micro powder.
Owner:ZHEJIANG THIRD AGE MATERIAL TECH CO LTD
Silver bromoiodide core-shell grain emulsion
InactiveUS20040152025A1Silver halide emulsionsSilver halide subtractive colour processesEmulsionSilver iodide
The present invention relates to a light-sensitive emulsion, and a light-sensitive photographic material containing the same, comprising silver bromoiodide grains of a core shell structure comprising: a) an inner core consisting essentially of silver bromide or silver bromoiodide and having a silver iodide content within the range of 0 to 10 mole %, and b) a plurality of shells consisting essentially of silver bromide or silver bromoiodide, wherein said silver bromoiodide grains exhibit an average grain size lower than 0.60 micrometer, an average grain thickness lower than 0.15 micrometer, and an average aspect ratio lower than 5:1.
Owner:PARCO TECHCO VAL BORMIDA
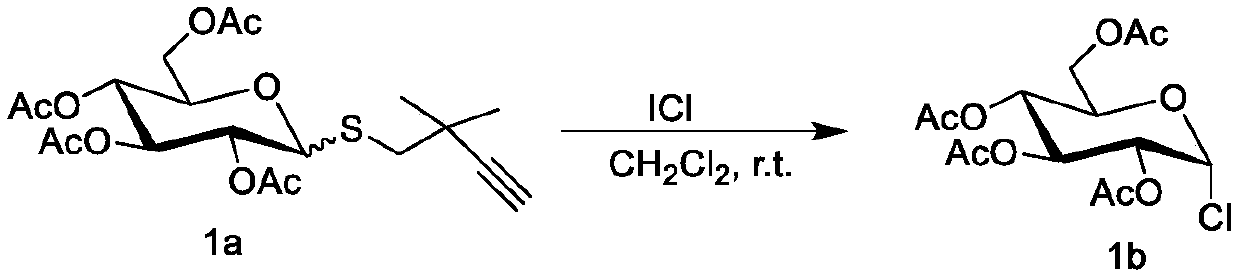
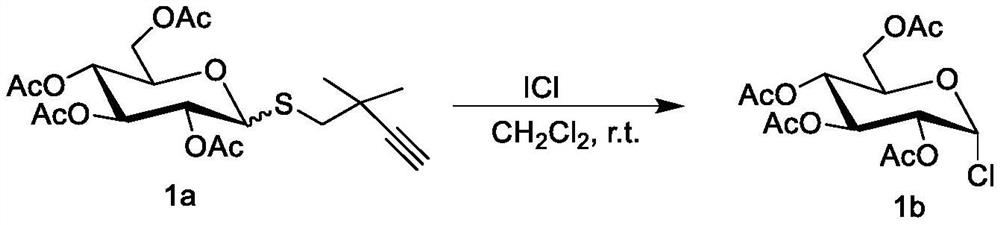
Method for preparing halogenated sugar under mild conditions
ActiveCN111269279AMild reaction conditionsThorough responseEsterified saccharide compoundsSugar derivativesPyranoseFuranose
The invention discloses a method for preparing halogenated sugar under mild conditions. The method comprises the following steps that an easily-prepared thioglycoside donor and a halogen simple substance or halogen intercompound undergo a reaction at room temperature to obtain the halogenated sugar (chlorine, bromine and iodine). The halogen simple substance or the halogen intercompound is commercial easily available iodine elementary substance, iodine bromide and iodine chloride. The method is suitable for various pyranoses and furanoses. The method has no limitation on a protecting group ofthe thioglycoside donor, and the protecting group can be an electron withdrawing group such as acetyl, benzoyl and the like, and can also be an electron donating group such as benzyl, silicon base andthe like. Meanwhile, the reaction can occur in various organic solvents such as dichloromethane, acetonitrile and methylbenzene. The preparation method of the halogenated sugar is simple, reaction conditions are mild, raw materials are easy to obtain, the application range is wide, the halogenated sugar is compatible with acid-labile groups such as isopropylidene ketal and silicon groups, and a pure product can be obtained by removing a solvent from the halogenated sugar which is not stable in the separation process.
Owner:SHAANXI NORMAL UNIV
Method for preparing methyl 4-amino-5-ethylsulfonyl-2-methoxybenzoate
The invention relates to a method for preparing methyl 4-amino-5-ethylsulfonyl-2-methoxybenzoate. The method comprises the steps of performing halogenation (chlorination, bromination and iodization) to 2-methoxy-4-acetylamine methyl benzoate, and then condensing together with ethane sulfinic acid sodium salt to obtain a target product. The method has the advantages that the technological route is simple, and the product is high in yield which achieves the total yield up to 80%, good in the quality with the purity up to 99.5%, and is white in appearance.
Owner:苏州诚和医药化学有限公司
Silver bromoiodide core-shell grain emulsion
InactiveUS6815154B2Silver halide emulsionsSilver halide subtractive colour processesEmulsionSilver iodide
The present invention relates to a light-sensitive emulsion, and a light-sensitive photographic material containing the same, comprising silver bromoiodide grains of a core shell structure comprising: a) an inner core consisting essentially of silver bromide or silver bromoiodide and having a silver iodide content within the range of 0 to 10 mole %, and b) a plurality of shells consisting essentially of silver bromide or silver bromoiodide, wherein said silver bromoiodide grains exhibit an average grain size lower than 0.60 micrometer, an average grain thickness lower than 0.15 micrometer, and an average aspect ratio lower than 5:1.
Owner:PARCO TECHCO VAL BORMIDA
Preparation method of carbon/carbon composite material MoB2 anti-oxidation outer coating
The invention provides a preparation method of a carbon / carbon composite material MoB2 anti-oxidation outer coating. The preparation method comprises the following steps of dispersing MoB2 powder in methanol, performing ultrasonic oscillation and stirring to obtain a suspension A; adding iodine bromide into the suspension A, performing ultrasonic oscillation, and stirring to obtain a solution B; pouring the solution B into a hydrothermal reaction kettle and controlling the filling degree to be 70-80%; then clamping a carbon / carbon composite material test sample with a SiC inner coating on a cathode clamp in the hydrothermal kettle, and placing the hydrothermal kettle into an oven; respectively connecting a positive electrode and a negative electrode of the hydrothermal kettle onto two corresponding electrodes of a pulsed power supply, and naturally cooling to room temperature after the end of hydrothermal electrophoresis electric arc discharge; opening the hydrothermal kettle, taking out the test sample and then drying to obtain the carbon / carbon composite material MoB2 anti-oxidation outer coating. The carbon / carbon composite material MoB2 anti-oxidation outer coating prepared by the preparation method provided by the invention has the advantages of uniform thickness and no cracks on the surface; the adopted process method has the advantages of simplicity in preparation, convenience in operation, easiness in obtainment of raw materials, short preparation period and low cost.
Owner:SHAANXI UNIV OF SCI & TECH
Method for determining vinyl content of low-vinyl methyl vinyl silicone rubber
ActiveCN107664608AAvoid intractable disadvantagesDirect detection methodInvestigating material hardnessRubber materialPolymer science
The invention relates to a method for determining the vinyl content of low-vinyl methyl vinyl silicone rubber (''110 silicone rubber'' for short). The method is characterized by comprising the steps of uniformly mixing the 110 silicone rubber with a vulcanizing agent, putting a rubber material on a mold for formation, detecting the Shore hardness, and determining the vinyl content according to theShore hardness. A traditional determination method is an iodine bromide method, namely addition developing reaction, and the traditional determination method has the disadvantages that the operationis tedious, the detection time is long (5 hours), the detection deviation is large (+ / -0.02%), and particularly, the low-vinyl 110 silicone rubber with the vinyl content lower than 0.06% cannot be effectively identified. A novel method for detecting the vinyl content of the vinyl 110 silicone rubber is an application method, namely that a network structure is formed through vulcanization and crosslinking, and the vinyl content is reflected by virtue of the hardness. The method disclosed by the invention has the advantages that the evaluation is rapid, the operation is simple, the application is direct, and meanwhile, the use of products by a client can be relatively visually guided.
Owner:HUBEI XINGRUI SILICON MATERIAL CO LTD
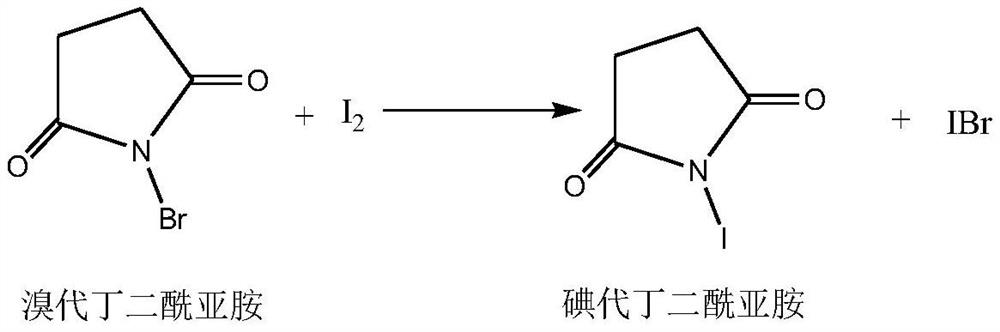
Method for recycling byproducts in preparation process of iodosuccinimide
The invention discloses a method for recycling byproducts in the preparation process of iodosuccinimide. The method comprises the following steps: recycling and refining the byproducts iodine bromidegenerated in the preparation process of iodosuccinimide into iodine, and preparing iodosuccinimide. The whole process is quick in reaction and suitable for industrial production.
Owner:启东东岳药业有限公司
Process for recovering iodine from medical organic waste liquid containing iodine bromide
The invention relates to a process for recovering iodine from medical organic waste liquid containing iodine bromide. The process comprises the following steps: neutralizing a large amount of acid inthe waste liquid by using caustic soda liquid, keeping weak acidity by using the caustic soda liquid, conducting reduction by using sodium sulfite, separating out a water layer, allowing iodine to enter the water layer in the form of sodium iodide, incinerating a sodium iodide-containing water solution at a high temperature (1000-1100 DEG C) by using an incinerator to obtain an iodine-containing water solution in which organic matters are thoroughly removed, and producing iodine by using a conventional oxidation iodine production process, wherein the yield of iodine is 98% or above and the content of iodine is 96% or above. A produced organic layer is applied as a high-calorific-value fuel for the incinerator, and generated heat is used for production, so environmental pollution is reduced.
Owner:山东博苑医药化学股份有限公司
A kind of method for preparing halogenated sugar under mild conditions
ActiveCN111269279BMild reaction conditionsThorough responseEsterified saccharide compoundsSugar derivativesPyranosePhenacyl
The invention discloses a method for preparing halogenated sugars under mild conditions. The halogenated sugars (chlorine, bromine and iodine) can be obtained by reacting an easily prepared glucosinolate donor with a halogen element or an interhalogen compound at room temperature. . The halogen element and the inter-halogen compound described therein are iodine element, iodine bromide and iodine chloride, which are readily available commercially. This method is applicable to all kinds of pyranose and furanose. And this method has no limitation on the protecting group of the glucosinolate donor, which can be an electron withdrawing group such as acetyl group, benzoyl group, etc., or an electron donating group such as benzyl group, silicon group, etc. At the same time, the reaction can occur in various organic solvents such as dichloromethane, acetonitrile, and toluene. The preparation method of the halogenated sugar of the present invention is simple, the reaction conditions are mild, the raw materials are readily available, the application range is wide, the halogenated sugar is compatible with acid-labile groups such as isopropylidene ketal and silicon groups, and is unstable in the separation process. The halogenated sugar can be purified by removing the solvent.
Owner:SHAANXI NORMAL UNIV
Synthetic method of 2-methoxyl-4-amino-5-ethyl sulfuryl methyl benzoate
The present invention relates to a kind of synthetic method of 2-methoxy-4-amino-5-ethylsulfonyl benzoic acid methyl ester, the present invention is that 2-methoxy-4-acetamido benzoic acid methyl ester is halogenated by halogen ( Chlorination, bromination, iodination), and then condense the target product with sodium ethyl sulfinate, which has the advantages of simple process route, high product yield, total yield of 80%, good product quality, purity of 99.5%, and The appearance is white.
Owner:苏州诚和医药化学有限公司