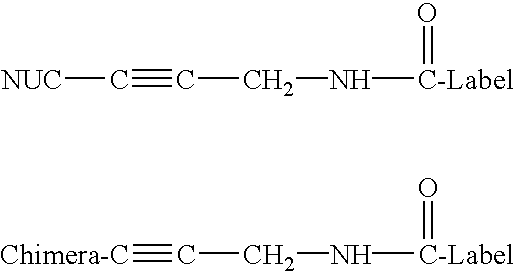Patents
Literature
787 results about "Fluorescent stain" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
Fluor·es·cent stain. (flōr-es'ĕnt stān) A staining procedure that uses a fluorescent dye or substance that combines selectively with certain tissue components and then fluoresces on irradiation with ultraviolet or violet-blue light.
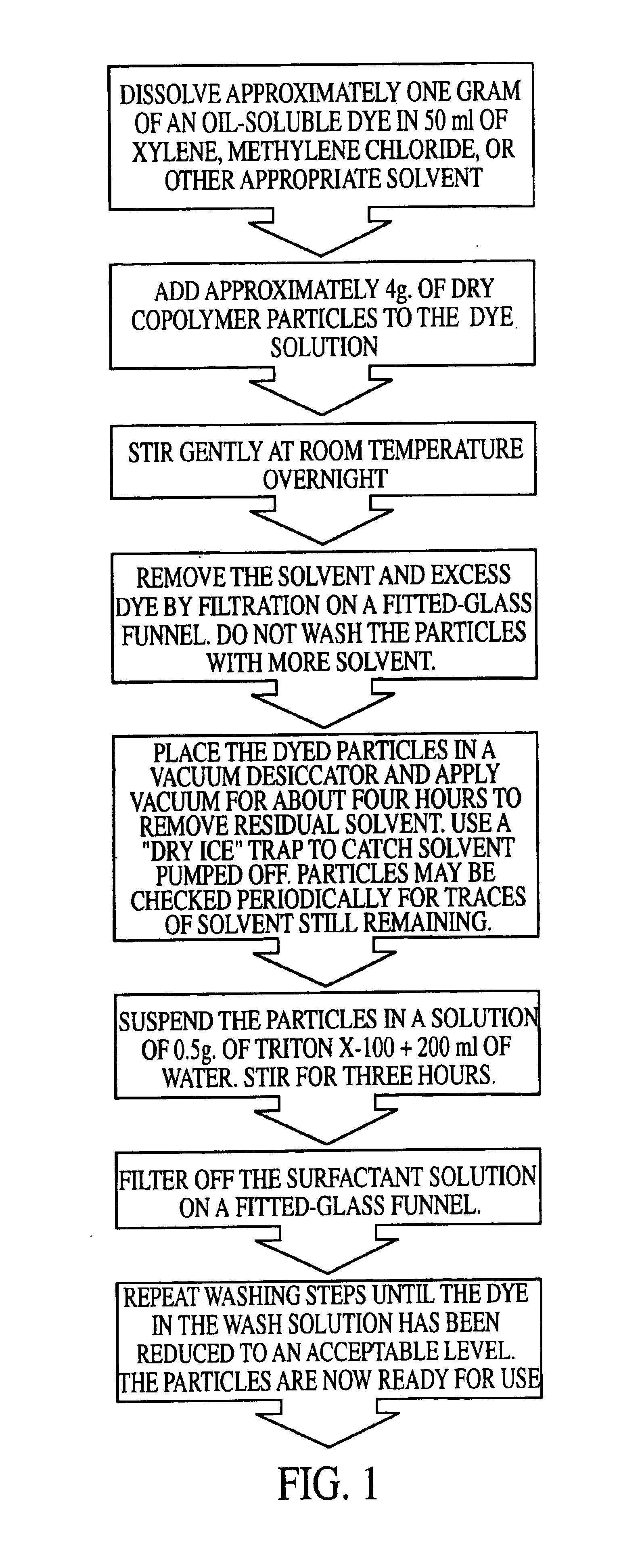
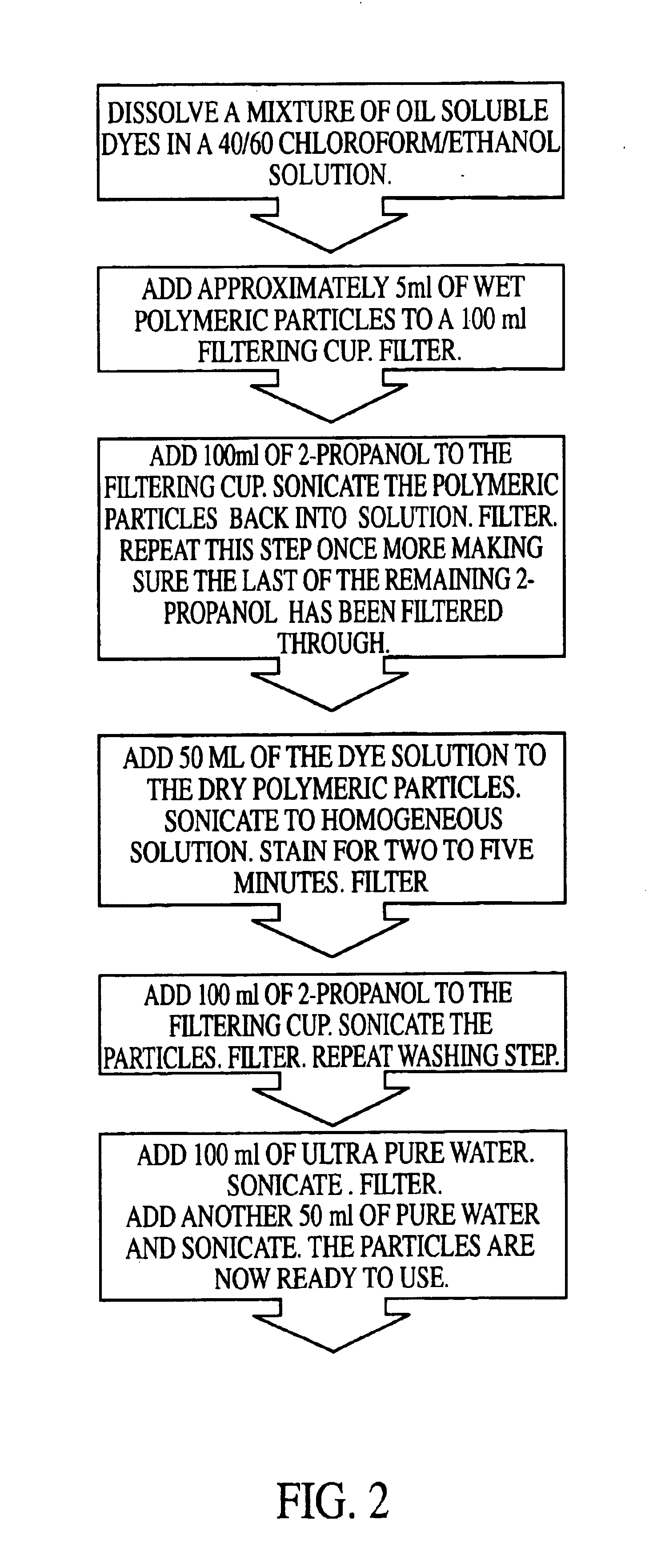
Precision fluorescently dyed particles and methods of making and using same
InactiveUS6929859B2Reduce variationIncrease rangeLiquid surface applicatorsMicrobiological testing/measurementSingle sampleAnalyte
An improved method of making a series of bead or microsphere or particle populations characterized by subtle variation in a proportion or ratio of at least two fluorescent dyes distributed within a single bead of each population is provided. These beads, when excited by a single excitation light source are capable of giving off several fluorescent signals simultaneously. A set containing as many as 64 distinct populations of multicolored, fluorescent beads is provided and when combined with analytical reagents bound to the surface of such beads is extremely useful for multiplexed analysis of a plurality of analytes in a single sample. Thus, methods of staining polymeric particles, the particles themselves, and methods of using such particles are claimed.
Owner:LUMINEX
Fluorescent compounds
ActiveUS20090305410A1Convenient and effective labelingMethine/polymethine dyesPeptide/protein ingredientsBiotechnologyDisease
Owner:BIOTIUM INC
Heterocycle-substituted xanthene dyes
ActiveUS20110136201A1Increase brightnessImprove photostabilitySugar derivativesSamplingDiseaseXanthene dye
The present invention relates to fluorescent dyes in general. The present invention provides a wide range of fluorescent dyes and kits containing the same, which are applicable for labeling a variety of biomolecules, cells and microorganisms. The present invention also provides various methods of using the fluorescent dyes for research and development, forensic identification, environmental studies, diagnosis, prognosis, and / or treatment of disease conditions.
Owner:BIOTIUM INC
Signature protected photosensitive optically variable ink compositions and process
InactiveUS20050279248A1High saturation of luminescenceHigh quantum yieldInksLuminescent compositionsRare earthDark color
Signature protected photosensitive optically variable (POV) inks are provided, which are capable of providing a unique signature in addition to other security features of POV inks. The inks contain at least two types of colorants, and a third, signature component and other ingredients to enable printing. A first colorant comprises a fluorescent dye and / or pigment emitting light within a characteristic emission band when excited by fluorescent-exciting radiation. A second colorant comprises a dye and / or pigment having a light absorption band at overlapping or longer wavelengths than the characteristic emission band of the first colorant in such a way as to result in a dark color. The third component is a fluorescent / phosphorescent rare earth composition. The inks give dark visible ink images, which also produce detectable coincident fluorescent and phosphorescent images. The inks can be used with detectors of red phosphorescence to achieve a new level of security in high speed sorting operations.
Owner:PITNEY BOWES INC
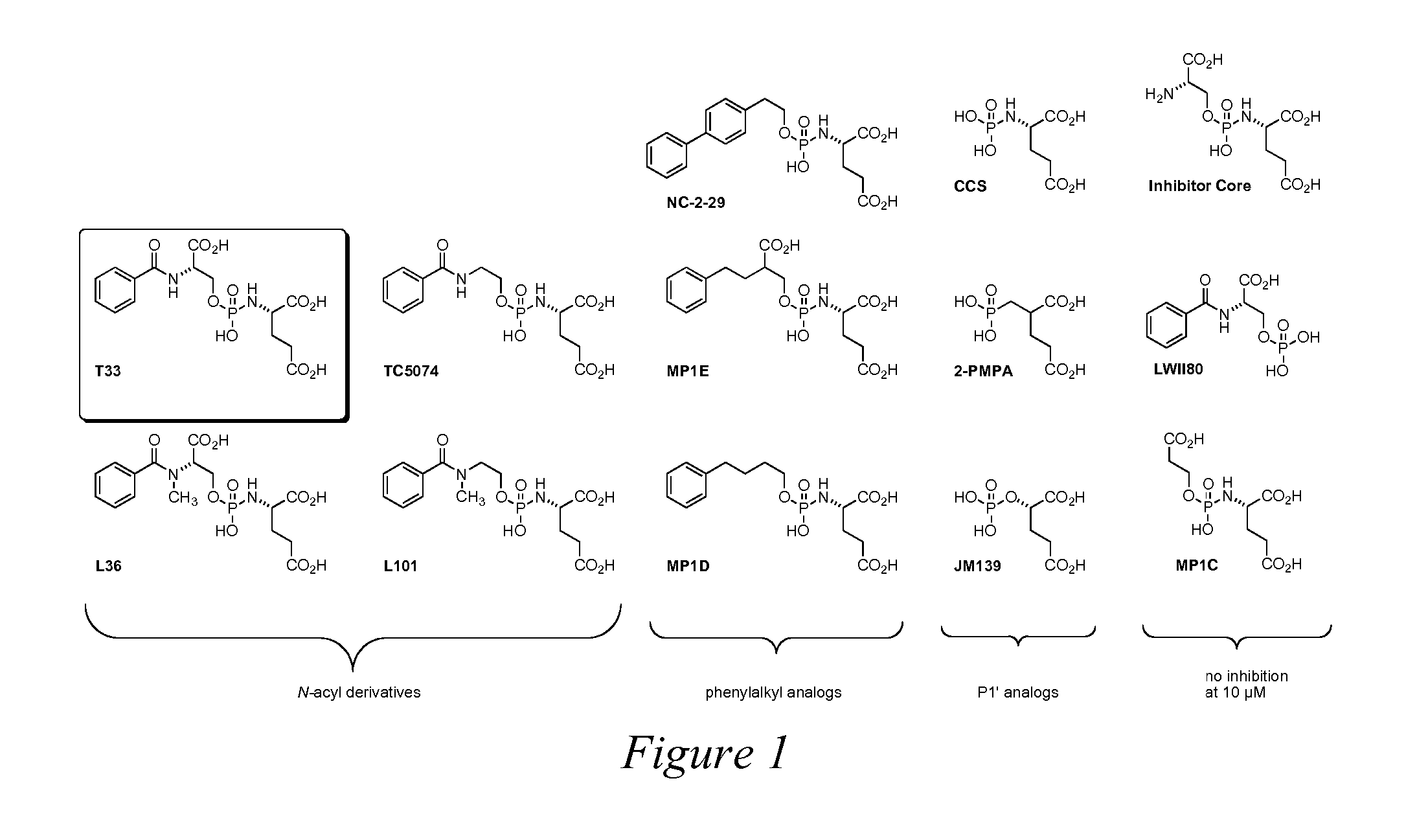
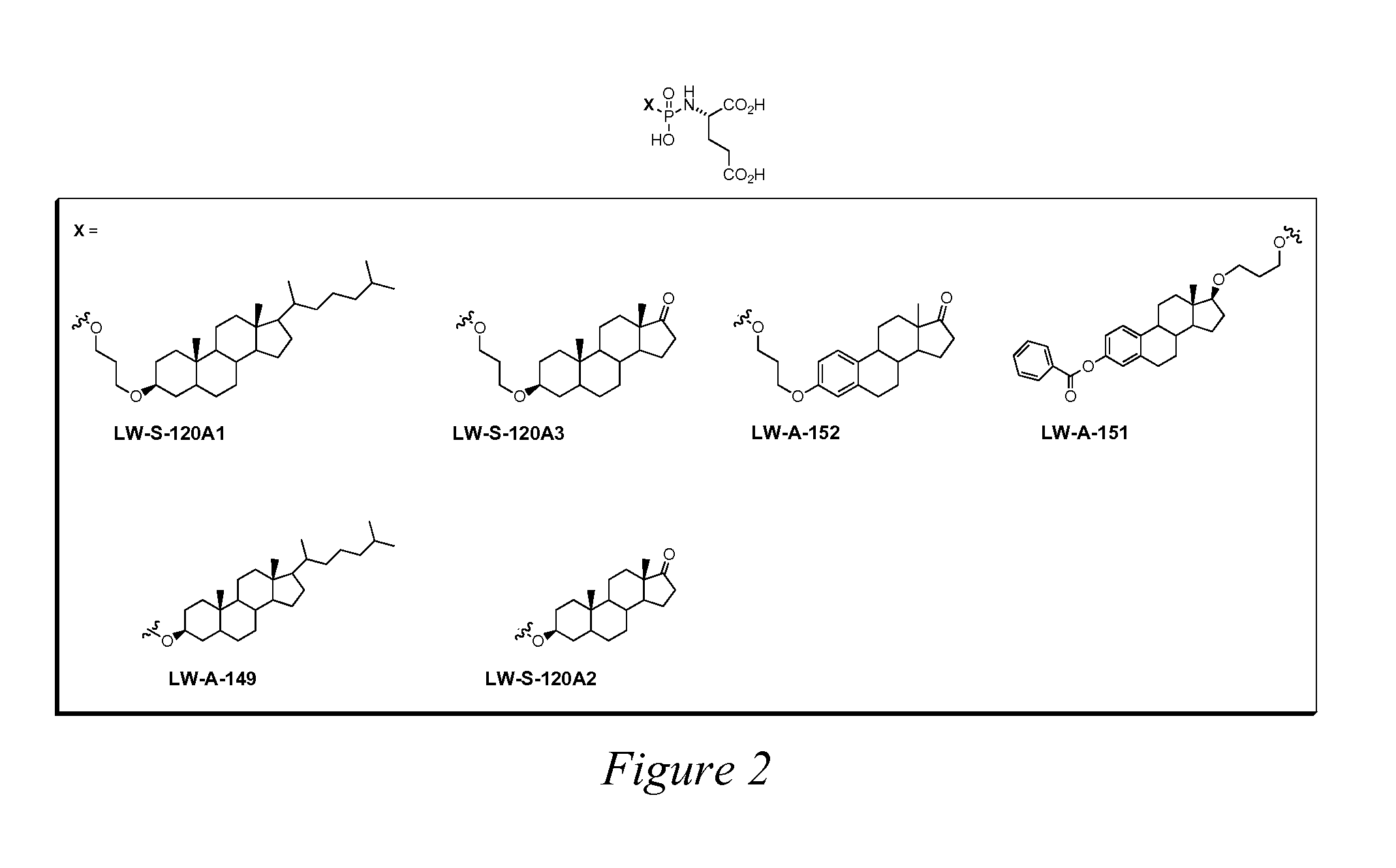
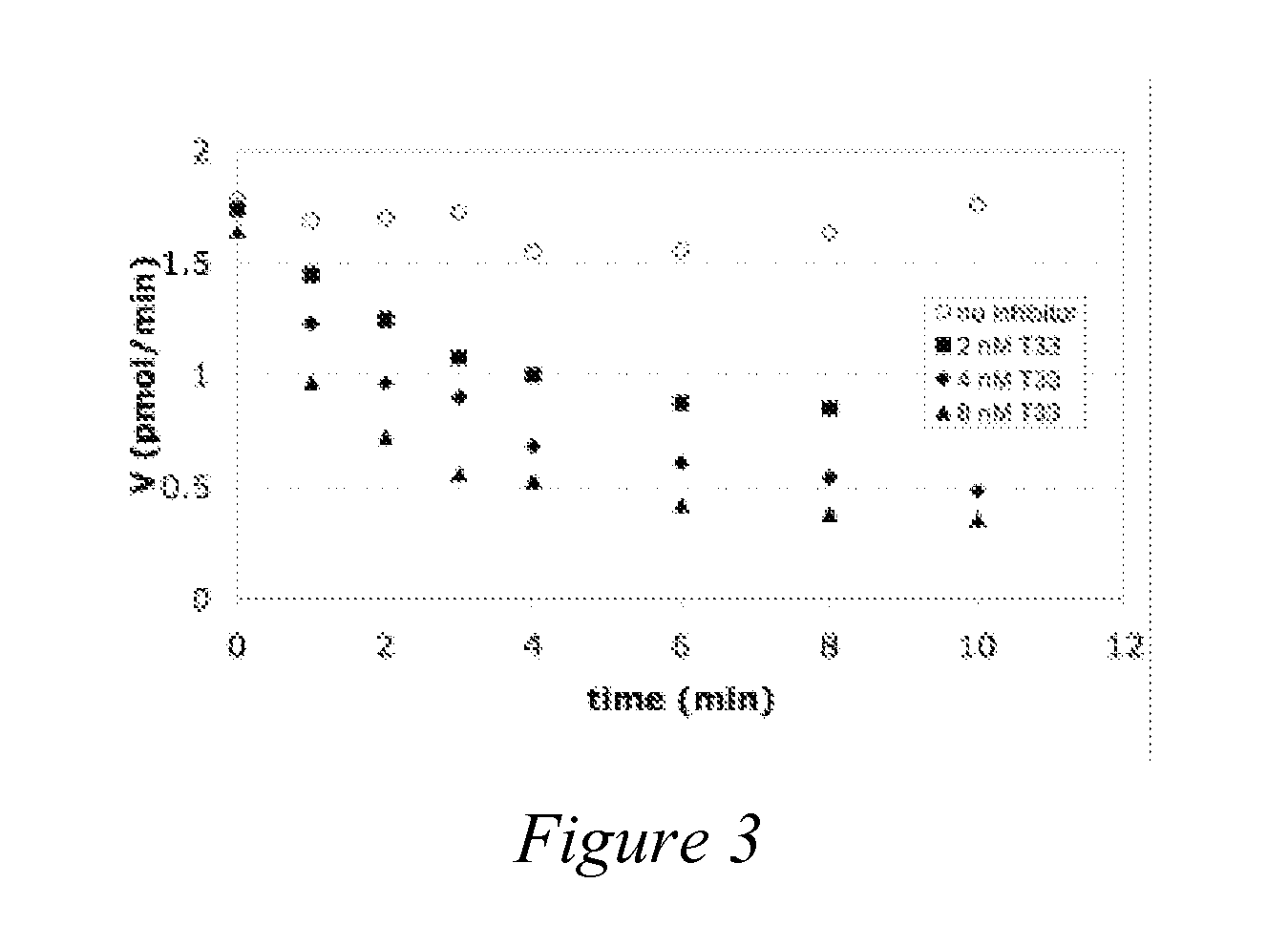
Peptidomimetic Inhibitors Of PSMA, Compounds Comprising Them, And Methods of Use
ActiveUS20070219165A1More cost-effectivelyBiocidePhosphorous compound active ingredientsAntigenProstate cancer cell
Compounds of the formula, A-L-B, wherein A is glutamate or a glutamate analog; L is a phosphoramidate or a phosphoramidate analog; and B is serine or a serine analog are described which are potent inhibitors of prostate-specific membrane antigen (PMSA). Such compounds are useful in treatment of prostate cancer; and when chemically attached to a fluorescent dye, can efficiently and selectively label prostate cancer cells for fluorescent imaging.
Owner:CANCER TARGETED TECH
Preparation method of waterborne polyurethane containing fluorescent dye
ActiveCN103275334AStrong two-photon effectStrong photoluminescent effectPolymer sciencePhotoluminescence
The invention discloses a preparation method of waterborne polyurethane containing a fluorescent dye, comprising the following steps: carrying out a reaction between the fluorescent dye containing reactive hydrogen and isocyanate and grafting parent dyes on polyurethane molecular chains, carrying out methods such as chain extension, crosslinking, side suspension of active groups and introduction of a hydrophilc agent, introducing crosslinking points, active groups and hydrophilic groups on the polyurethane chains, making molecular weight large enough, neutralizing and emulsifying to prepare the waterborne polyurethane fluorescent dye. The fluorescent polyurethane prepared by the preparation method still has strong two-photon effect and photoluminescence effect in an aqueous phase and an adhesive film. Thus, the waterborne polyurethane containing the fluorescent dye can be applied in aspects of anti-counterfeiting mark, traffic sign, biological development, biological detection, medicine tracing, chemical detection, fluorescent ink, fluorescent paint and the like.
Owner:ANHUI UNIVERSITY
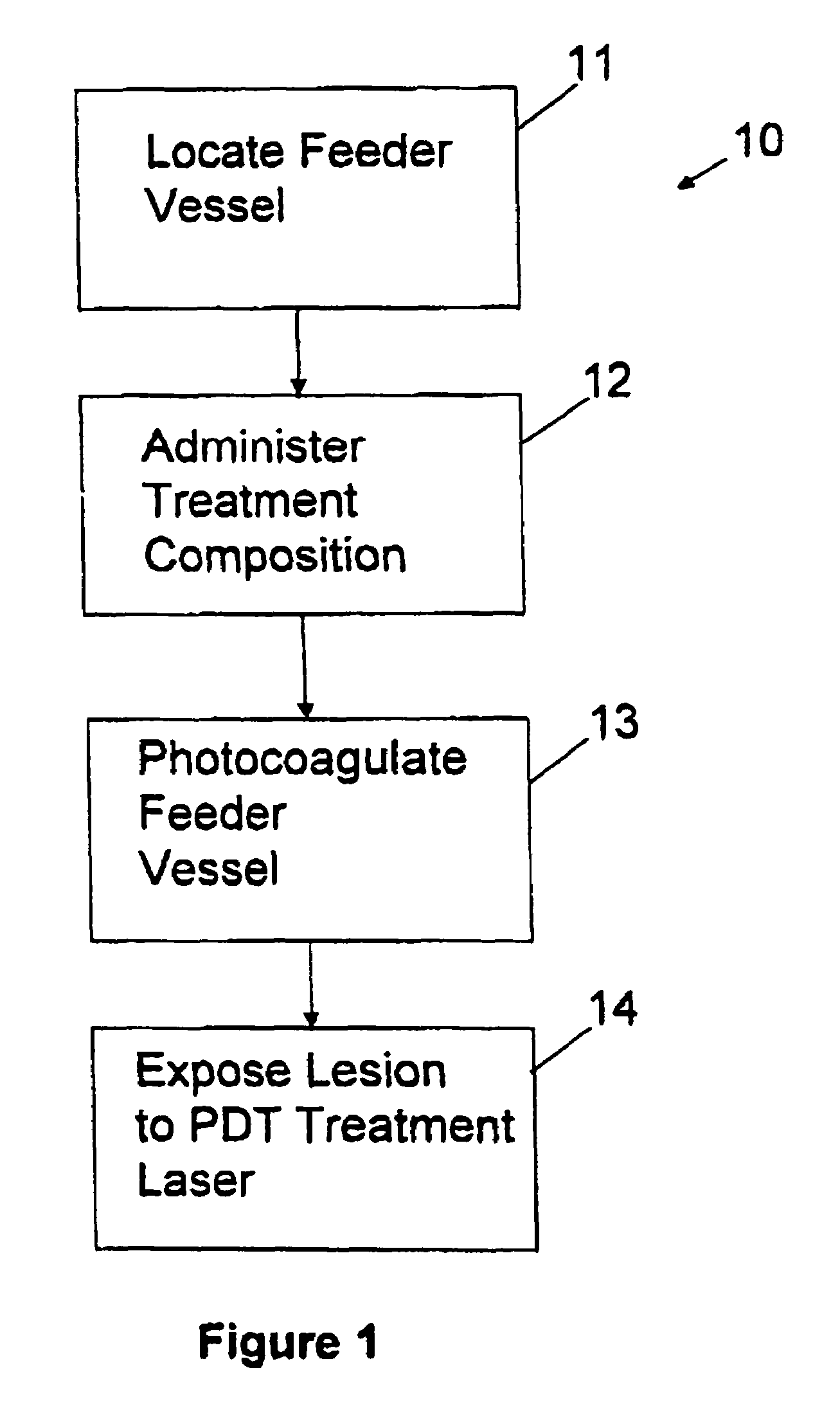
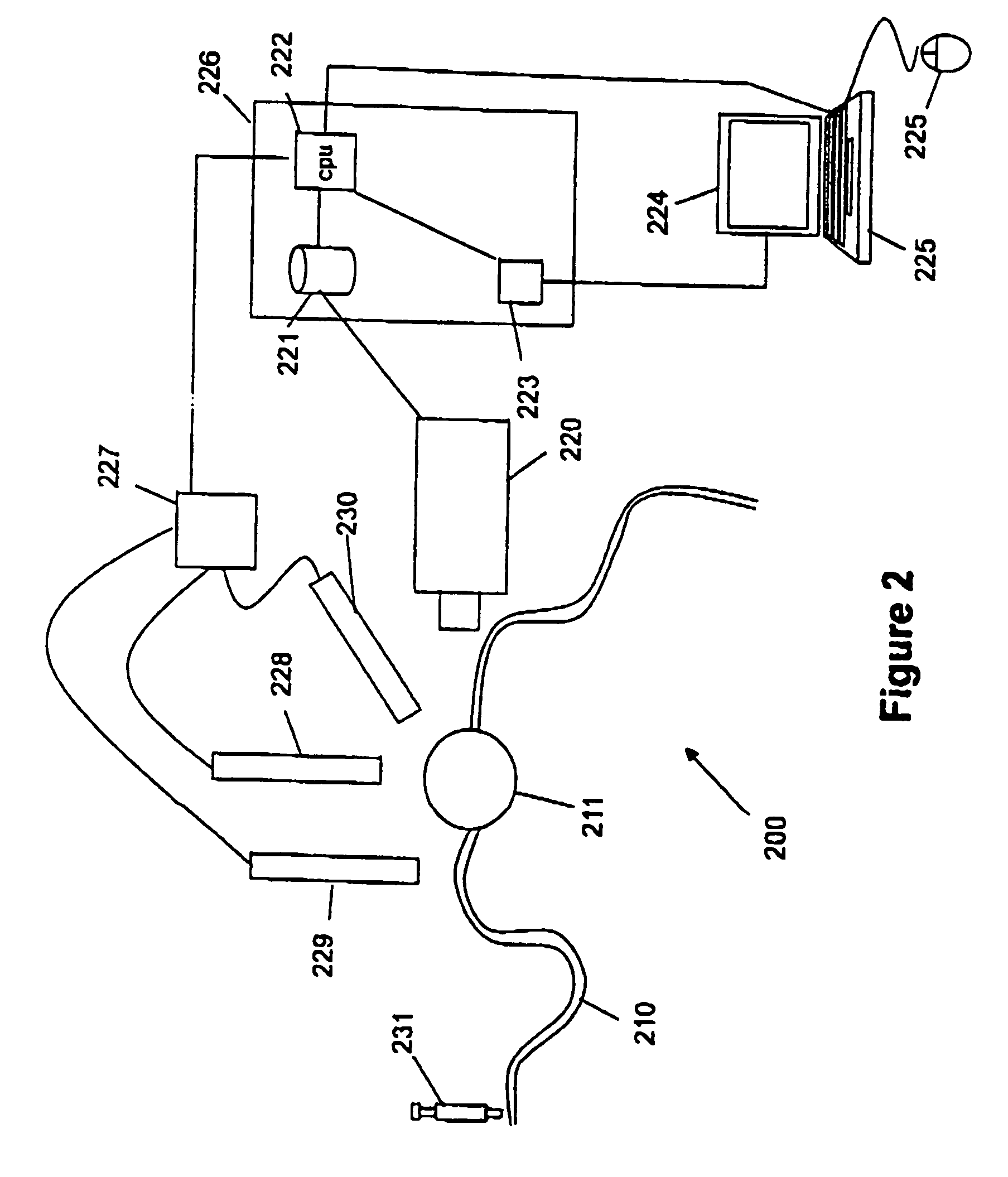
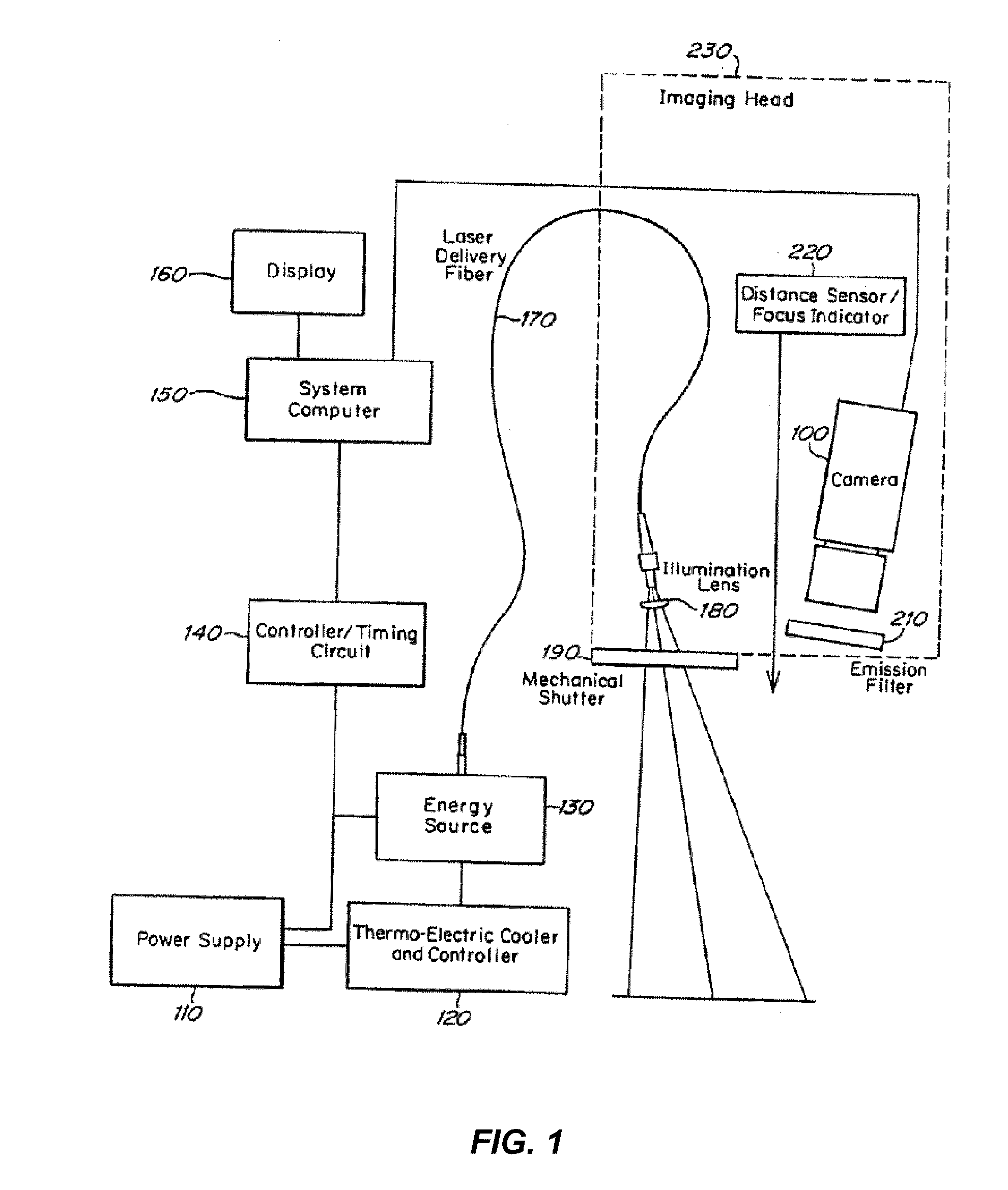
Combined photocoagulation and photodynamic therapy
InactiveUS7364574B2High flush-out rateAvoid damageLaser surgeryDiagnosticsPhotodynamic therapyLesion site
A method for treating a lesion of an animal, the animal having at least one vessel that carries blood to the lesion, comprising locating the vessel, administering a composition comprising a photodynamic agent, applying energy to the vessel to photocoagulate the vessel and thereby reduce the rate at which the treatment composition exits said lesion and applying energy to said lesion, of a type and an amount sufficient to excite the photodynamic agent, causing the lesion to undergo photodynamic therapy. Preferably, a dye that is both a fluorescent dye and a radiation absorbing dye, such as indocyanine green dye, is added to the treatment composition to allow (a) confirmation of the presence of the treatment composition in the lesion to be detected by fluorescent angiography and (b) the rate of blow flow to be reduced in the blood vessel feeding the lesion using dye enhanced photocoagulation.
Owner:NOVADAG TECH INC
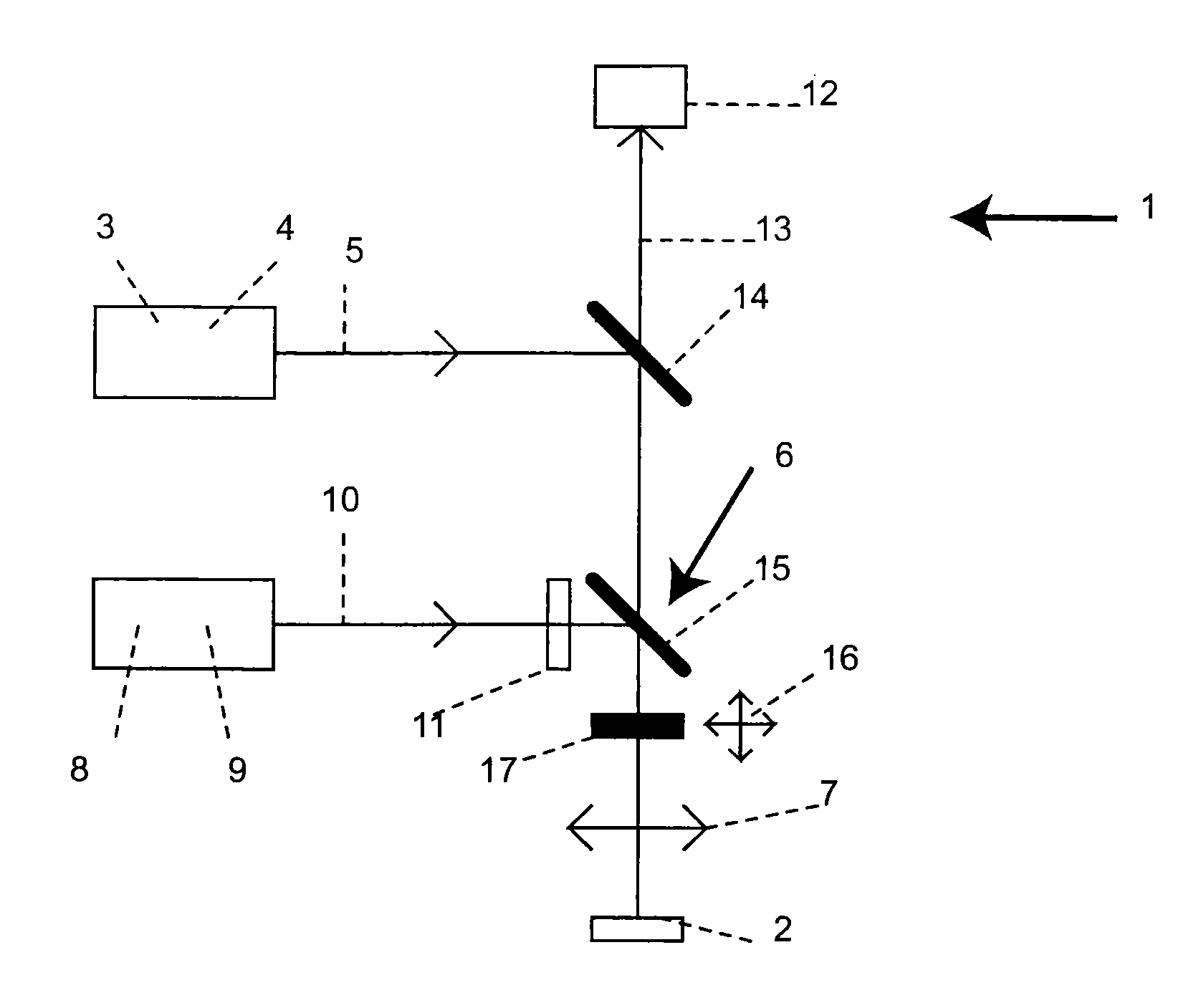
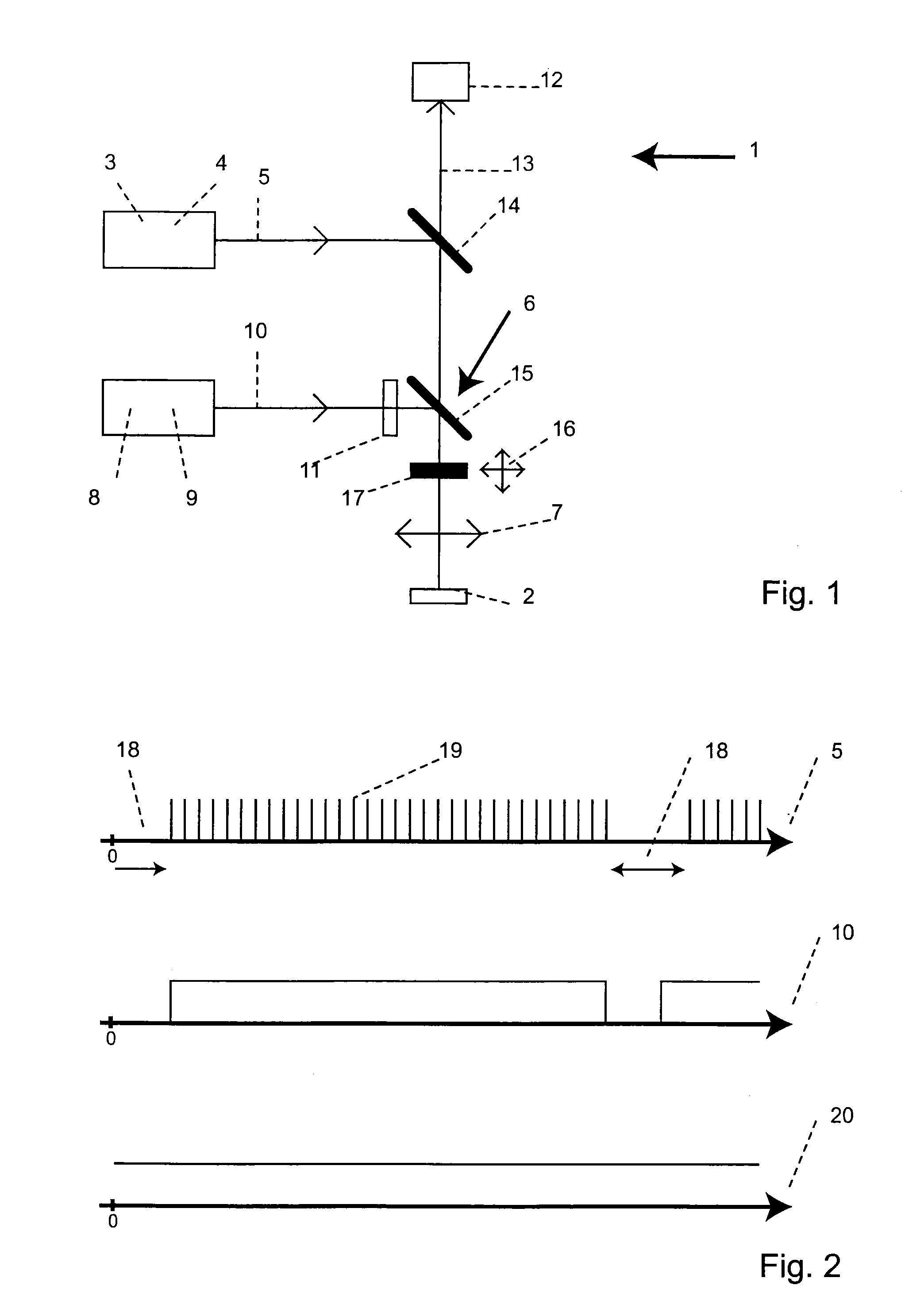
STED-Fluorescent Light Microscopy with Two-Photon Excitation
ActiveUS20100176307A1Improve spatial resolutionPhotometryLuminescent dosimetersFluorescenceImage resolution
A method of high spatial resolution imaging a structure in a sample comprises: marking the structure with molecules of a fluorescent dye; selecting a first wavelength for excitation light which excites the molecules of the fluorescent dye via a multi photon process for spontaneous emission of fluorescent light; focussing pulses of the excitation light into the sample to excite those molecules of the fluorescent dye present in a focal area of the focussed excitation light; selecting a second wavelength shorter than the first wavelength for de-excitation light which de-excites excited molecules of the fluorescent dye prior to their spontaneous emission; during a plurality of the pulses of the excitation light, continuously directing the de-excitation light onto the sample to de-excite excited molecules of the fluorescent dye, which are located outside an measurement area which is a fraction of the focal area; and recording the fluorescent light spontaneously emitted by the molecules of the fluorescent dye in the sample.
Owner:MAX PLANCK GESELLSCHAFT ZUR FOERDERUNG DER WISSENSCHAFTEN EV
Nir materials and nanomaterials for theranostic applications
InactiveUS20130039858A1Ultrasonic/sonic/infrasonic diagnosticsPowder deliveryMetal oxide nanoparticlesFluorescence
Novel fluorescent dye comprising metal oxide nanoparticles are prepared where the nanoparticles are as small as 3 nm or up to 7000 nm in diameter and where the dye is bound within the metal oxide matrix. In some embodiments the invention, novel dyes are covalently attached to the matrix and in other embodiments of the invention a dye is coordinate or ionic bound within the metal oxide matrix. A method for preparing the novel covalently bondable modified fluorescent dyes is presented. A method to prepare silica comprising nanoparticles that are 3 to 8 nm in diameter is presented. In some embodiments, the fluorescent dye comprising metal oxide nanoparticles are further decorated with functionality for use as multimodal in vitro or in vivo imaging agents. In other embodiments of the invention, the fluorescent dye comprising metal oxide nanoparticles provide therapeutic activity and incorporated therapeutic temperature monitoring.
Owner:UNIV OF FLORIDA RES FOUNDATION INC
White light organic electroluminescent device
ActiveCN106206958ALow costImprove efficiencySolid-state devicesSemiconductor/solid-state device manufacturingQuantum efficiencyMonochromatic color
The invention discloses a white light organic electroluminescent device, which comprises a light emitting layer. Multiple layers of single-color light emitting layers with complementary colors are stacked and combined to form the light emitting layer, the main material of at least one single-color light emitting layer is a thermal active delay fluorescent material, and the main material of each single-color light emitting layer is doped with a phosphorescent dye or a fluorescent dye. The thermal active delay fluorescent material is introduced to manufacturing of a white light device, the internal quantum efficiency of the material of the type is more than 25%, and the efficiency is high. As no precious metal material is used, the material cost is low.
Owner:BEIJING VISIONOX TECH +1
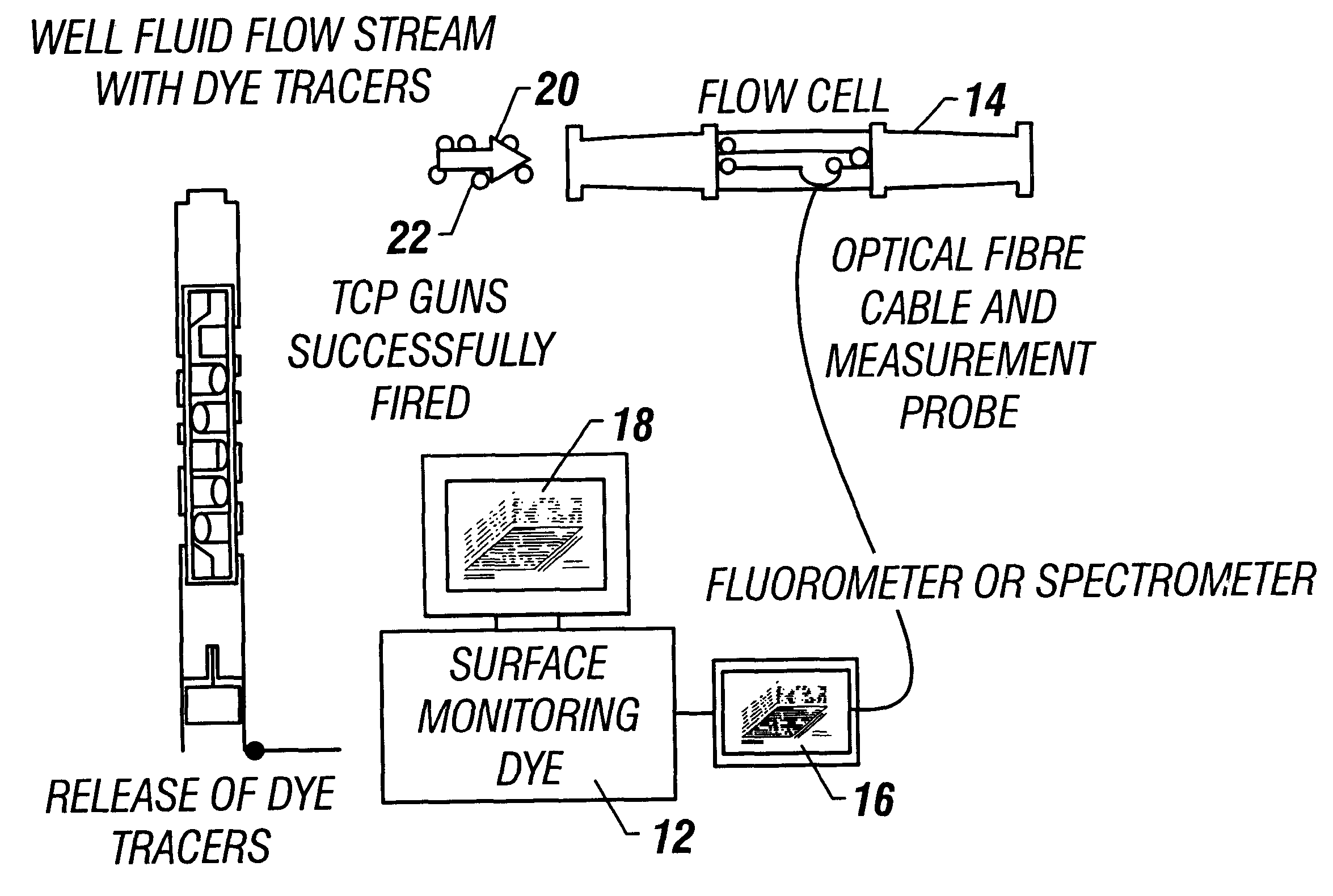
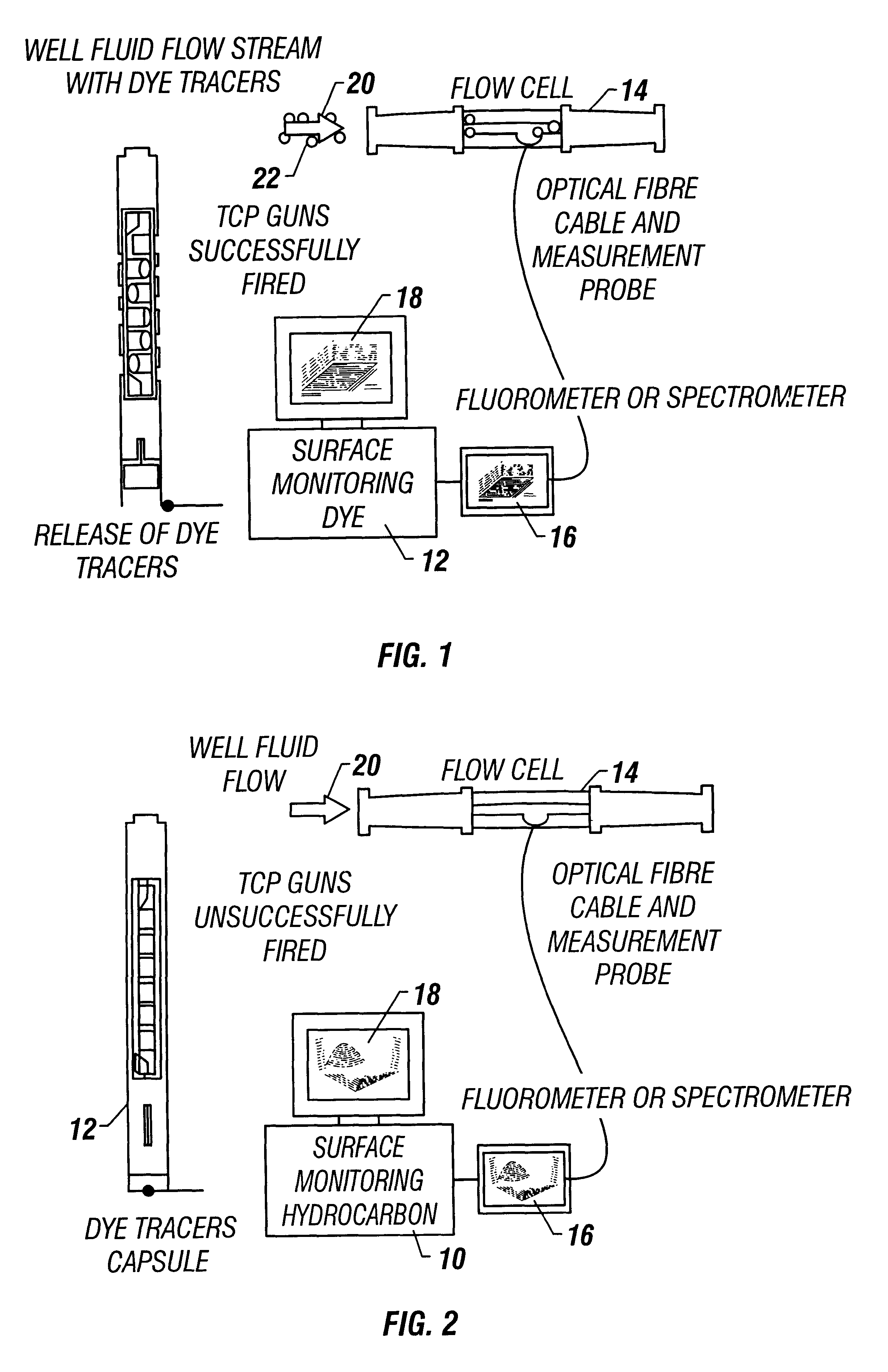
Method and apparatus for a tubing conveyed perforating guns fire identification system using enhanced marker material
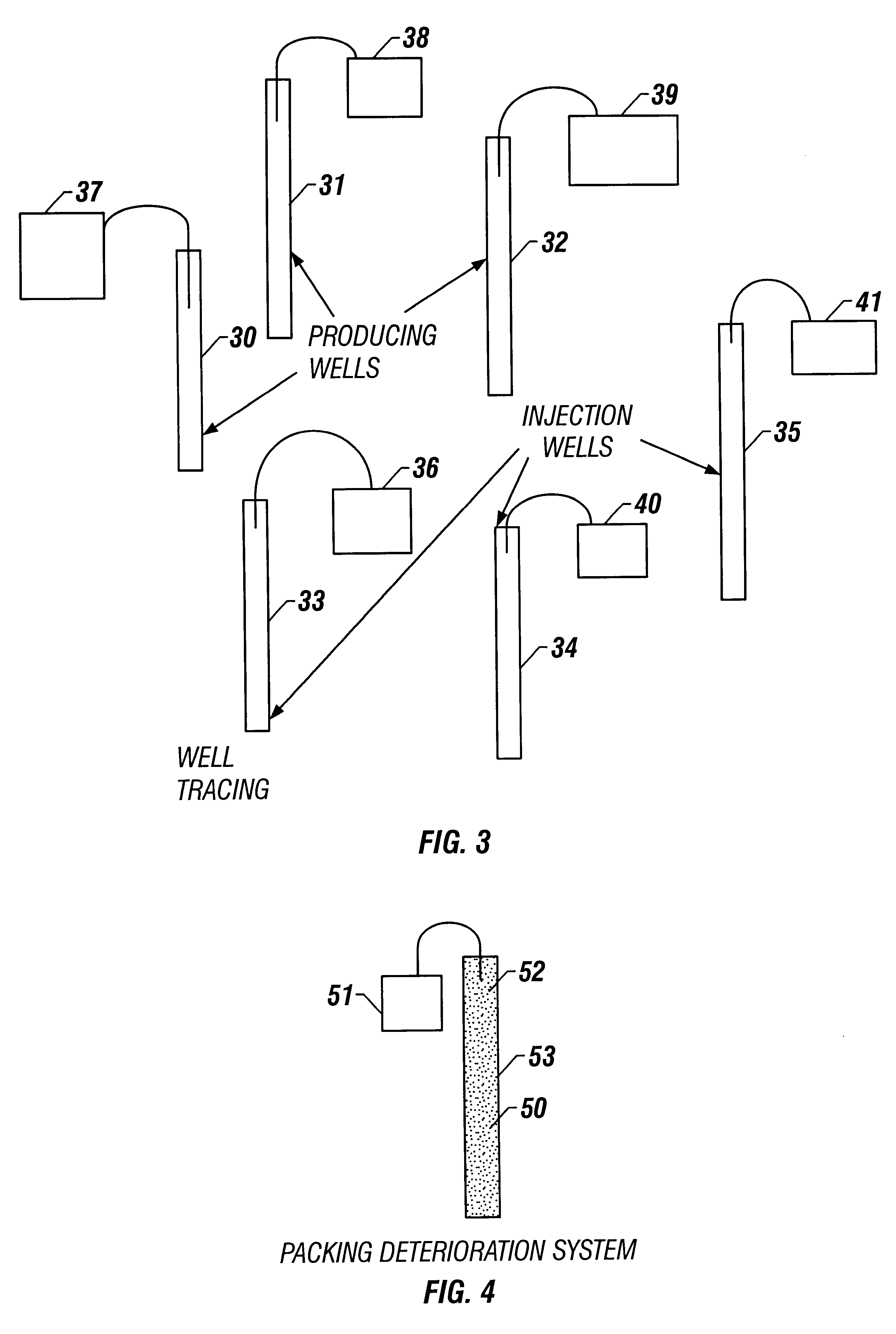
A method and apparatus detects the presence of a fluorescent tracer dye, a change in capacitance, or a change in fiber optic electrical properties to determine whether or not a TCP gun has fired. The method and apparatus detects the number of charges fired to determine whether or not all TCP guns have fired and also determines the contributions of injection wells to producing wells by introducing fluorescent tracers into injection wells and detecting the presence of the fluorescent tracers at production wells. The invention places fluorescent dye particles in a gravel pack to sense when a gravel pack is deteriorating by detecting the tracer dye particles in the well flow.
Owner:BAKER HUGHES INC
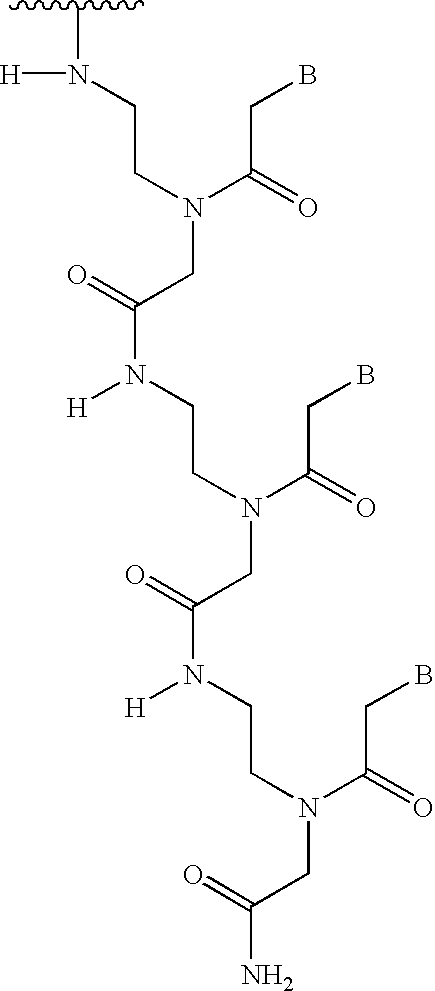
PNA-DNA chimeric probe arrays and methods of use
InactiveUS6469151B1Strong specificityHigh affinitySugar derivativesMaterial analysis by observing effect on chemical indicatorFluorescenceOligonucleotide
The invention provides methods, kits, and compositions for ligation of PNA-DNA chimeric probes and oligonucleotides when they are hybridized adjacently to template nucleic acids using ligases and ligation reagents. Structural requirements of the chimeras for ligation include 5 to 15 contiguous PNA monomer units, 2 or more contiguous nucleotides, and a 3' hydroxyl or 5' hydroxyl terminus. The chimera and / or oligonucleotide may be labelled with fluorescent dyes or other labels. The methods include, for example, oligonucleotide-ligation assays (OLA) and single nucleotide polymorphism detection.
Owner:APPL BIOSYSTEMS INC
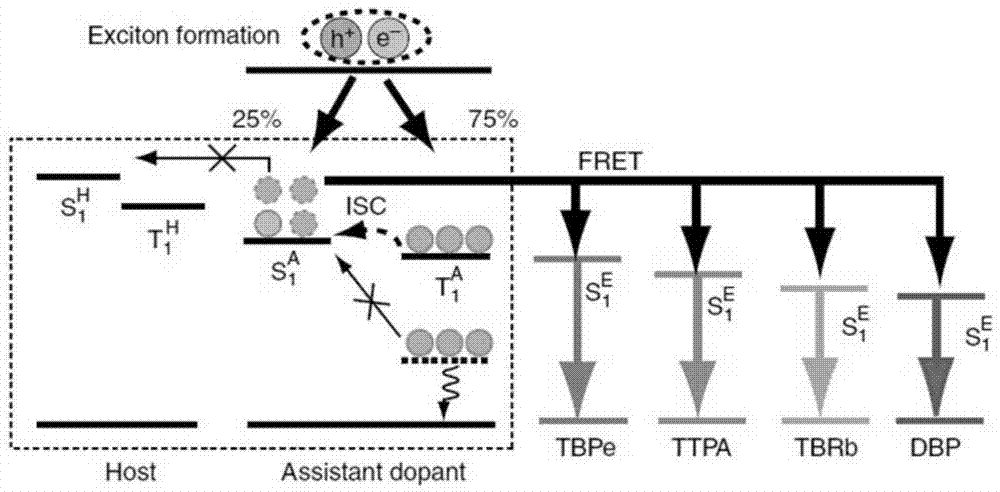
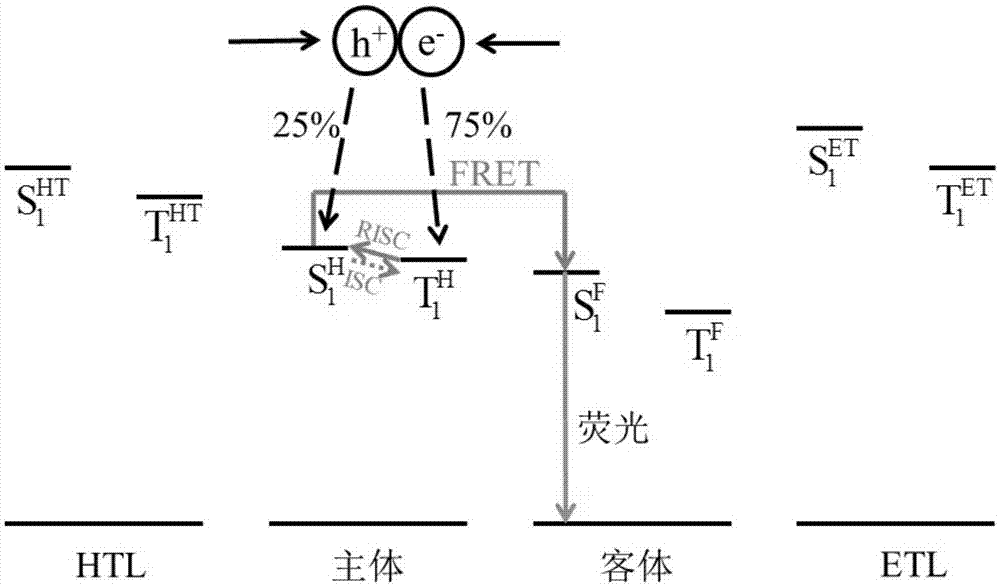
Organic light-emitting device
ActiveCN106920884AIncreased maximum theoretical external quantum efficiencyEasy transferSolid-state devicesSemiconductor/solid-state device manufacturingFluorescenceOrganic light emitting device
The invention relates to an organic light-emitting device. The main body materials of a light-emitting layer comprise materials with hole transfer capability and electron transfer capability; a triplet state T1H of at least one kind of main body material is greater than or equal to a singlet state S1F of a fluorescent dye; a triplet state energy level T1H of a CT excited state of at least one kind material of the main body materials is higher than a singlet state energy level S1H of an n-<pi> excited state, and T1H-S1H is less than or equal to 0.3eV; or the triplet state energy level T1H of the CT excited state of at least one kind of material of the main body materials is higher than a triplet state energy level S1H of the n-<pi> excited state, and T1H-S1H is greater than or equal to 1eV; in addition, the difference value between the second triplet state energy level of the n-<pi> excited state of the main body materials, and the first singlet state energy level of the CT excited state is -0.1eV to 0.1eV; and the triplet states T1 of organic functional layer materials adjacent to the light emitting layer are all higher than the singlet state S1H of the main body materials of the light emitting layer. By adopting a thermal activation delayed florescence material as the main body materials, so that recombination of excitons in the light-emitting region is limited, a phenomenon of efficiency roll-off is effectively suppressed, and the device efficiency can be improved to 13-18%.
Owner:KUNSHAN NEW FLAT PANEL DISPLAY TECH CENT
Amide-substituted xanthene dyes
Owner:ELITECHGROUP MDX LLC
Circulating tumor cell detection kit and application thereof
InactiveCN105785005AAccurate detectionEfficient enrichmentMaterial analysisLymphatic SpreadFluorescence
The invention relates to a circulating tumor cell detection kit and application thereof. The circulating tumor cell detection kit comprises a chip, sample diluent, a cell separation fluid, a cell trapping agent, a cell cleaning solution, a cell fixing agent, antibody diluent, a cell penetration agent and a fluorescence dye. The application method of the circulating tumor cell detection kit mainly comprises the several steps of separation of circulating tumor cells, trapped antibody coating, trapping of the circulating tumor cells and immuno-fluorescent staining of the circulating tumor cells. The circulating tumor cell detection kit is mainly used for tumor prognosis, reappear, metastasis, curative effect monitoring, early diagnosis and early warning, monitoring of curative effect and tumor progression situation, and utilizes the molecular subtyping, screening specificity and other characteristics of the circulating tumor cells to conduct targeted research on circulating tumor cell sensitive drug so as to supplement tumor metastasis, relapse and drug resistance mechanisms and guide individualized medical treatment.
Owner:杭州华得森生物技术有限公司
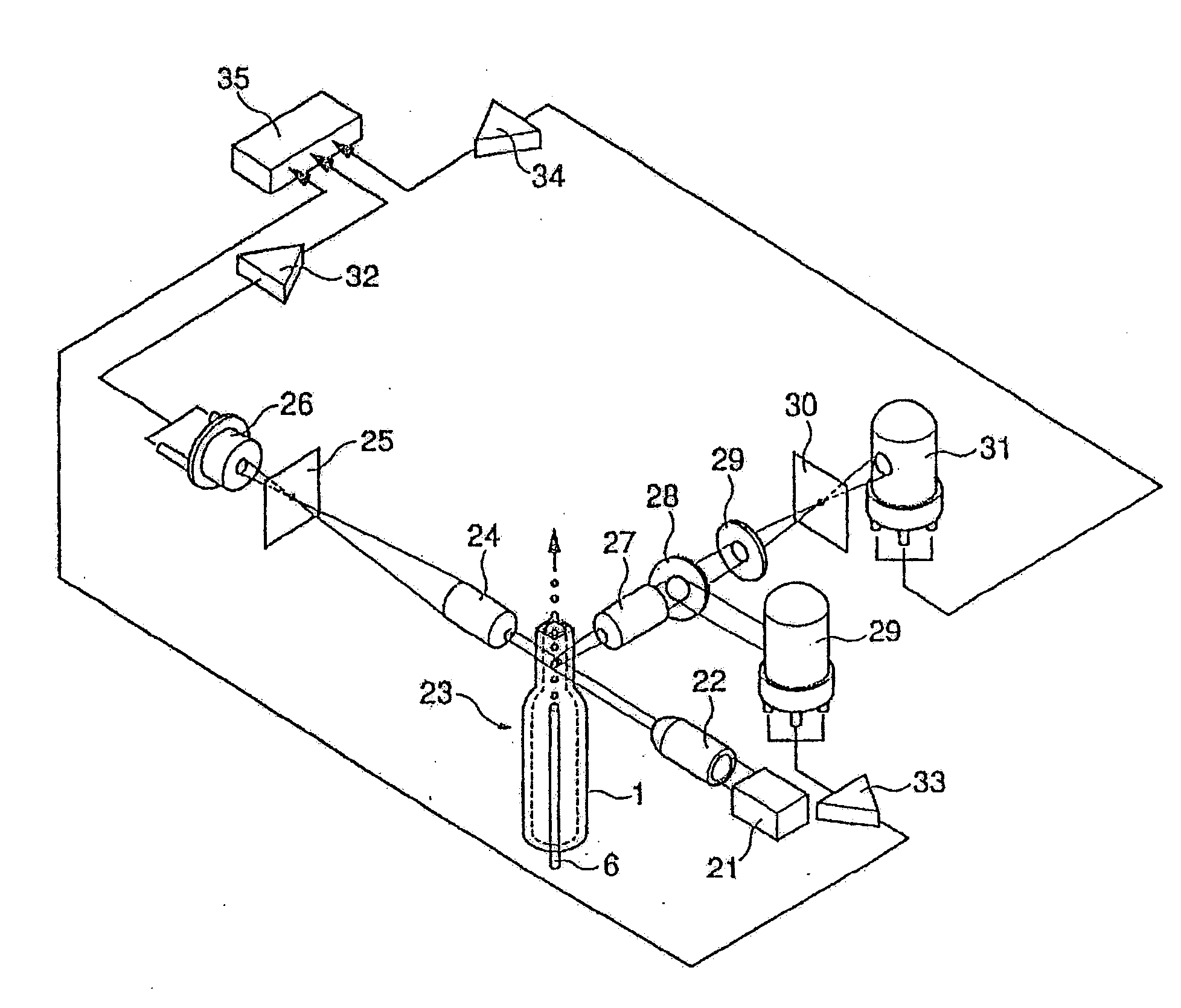
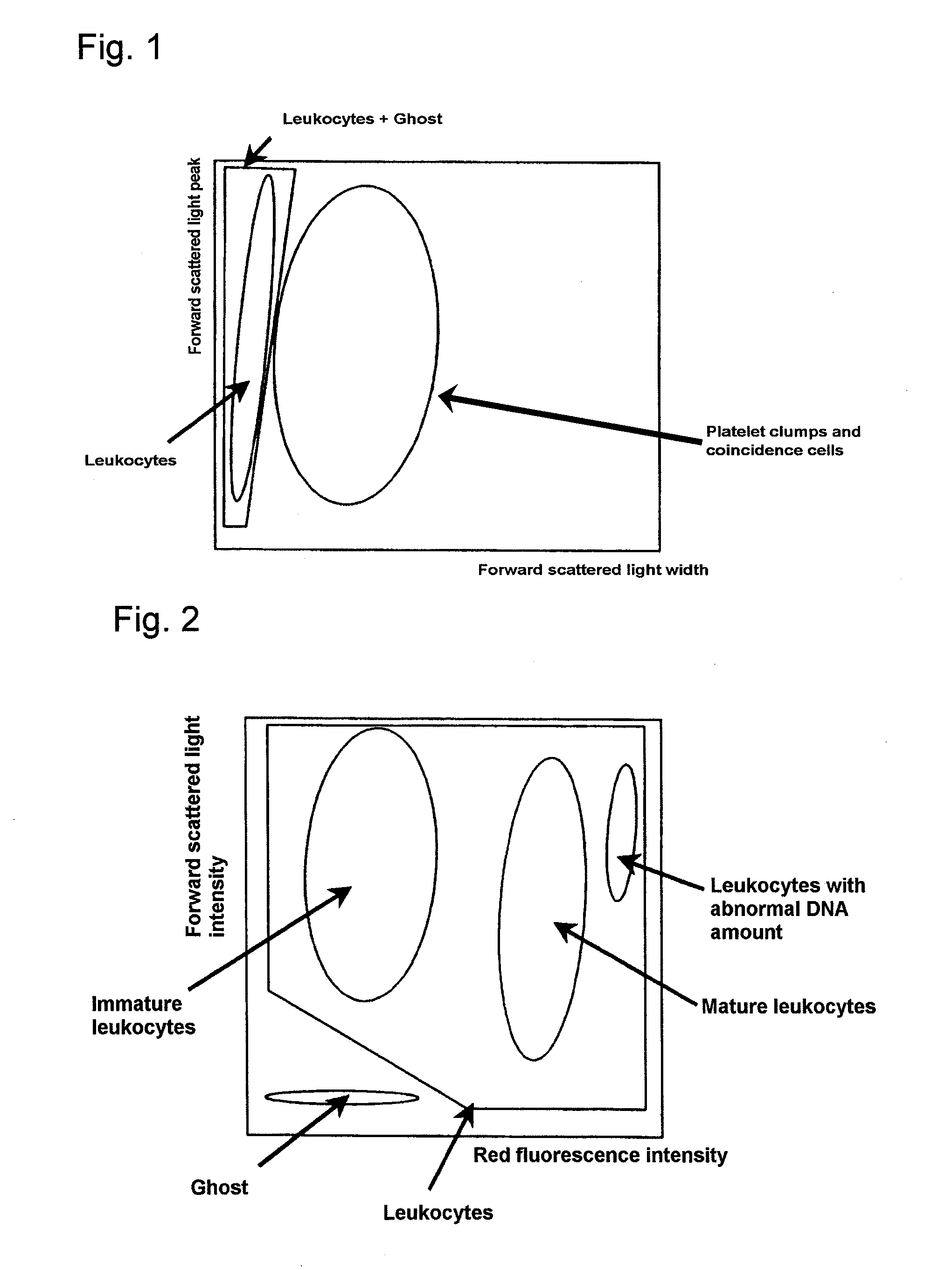
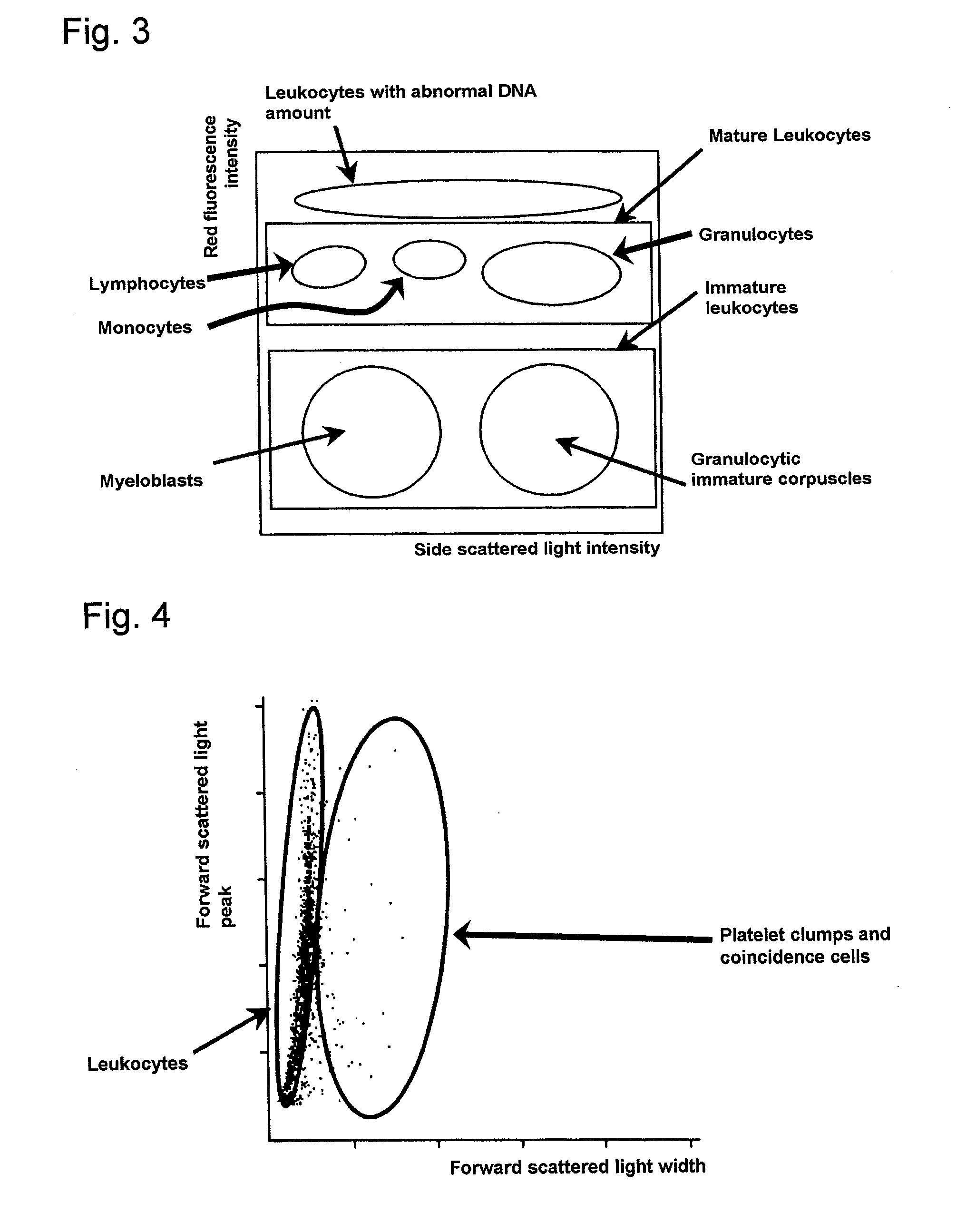
Method of classifying and counting leucocytes
ActiveUS20080176274A1Low costAccurate classificationMicrobiological testing/measurementIndividual particle analysisStainingWhite blood cell
Owner:SYSMEX CORP
Imaging methods and compositions comprising fluorescent dyes associated with viral components for nerve imaging
Disclosed herein are compositions and methods for imaging nerve cells. The composition comprises a fluorescent dye; and a viral component selected from a neurotropic, replication-defective virus, a viral protein of a neurotropic virus, and a capsid of a neurotropic virus. Although the fluorescent dye in itself cannot penetrate nerve cells, the fluorescent dye is bound to the viral component to form a dye / viral component complex that is capable of penetrating nerve cells.
Owner:STRYKER EUROPEAN OPERATIONS LIMITED
Luminescent solar collector
Owner:SABIC INNOVATIVE PLASTICS IP BV
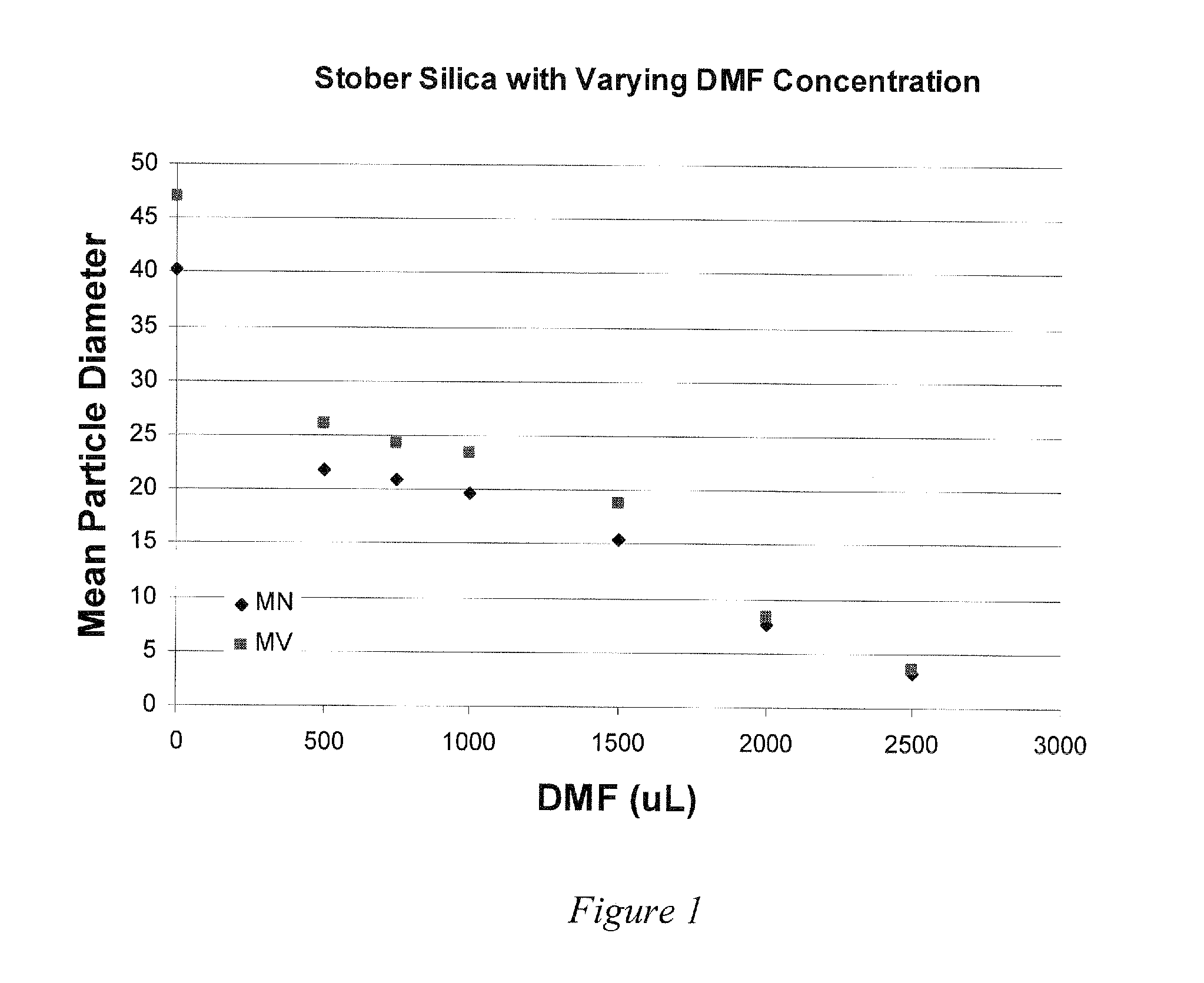
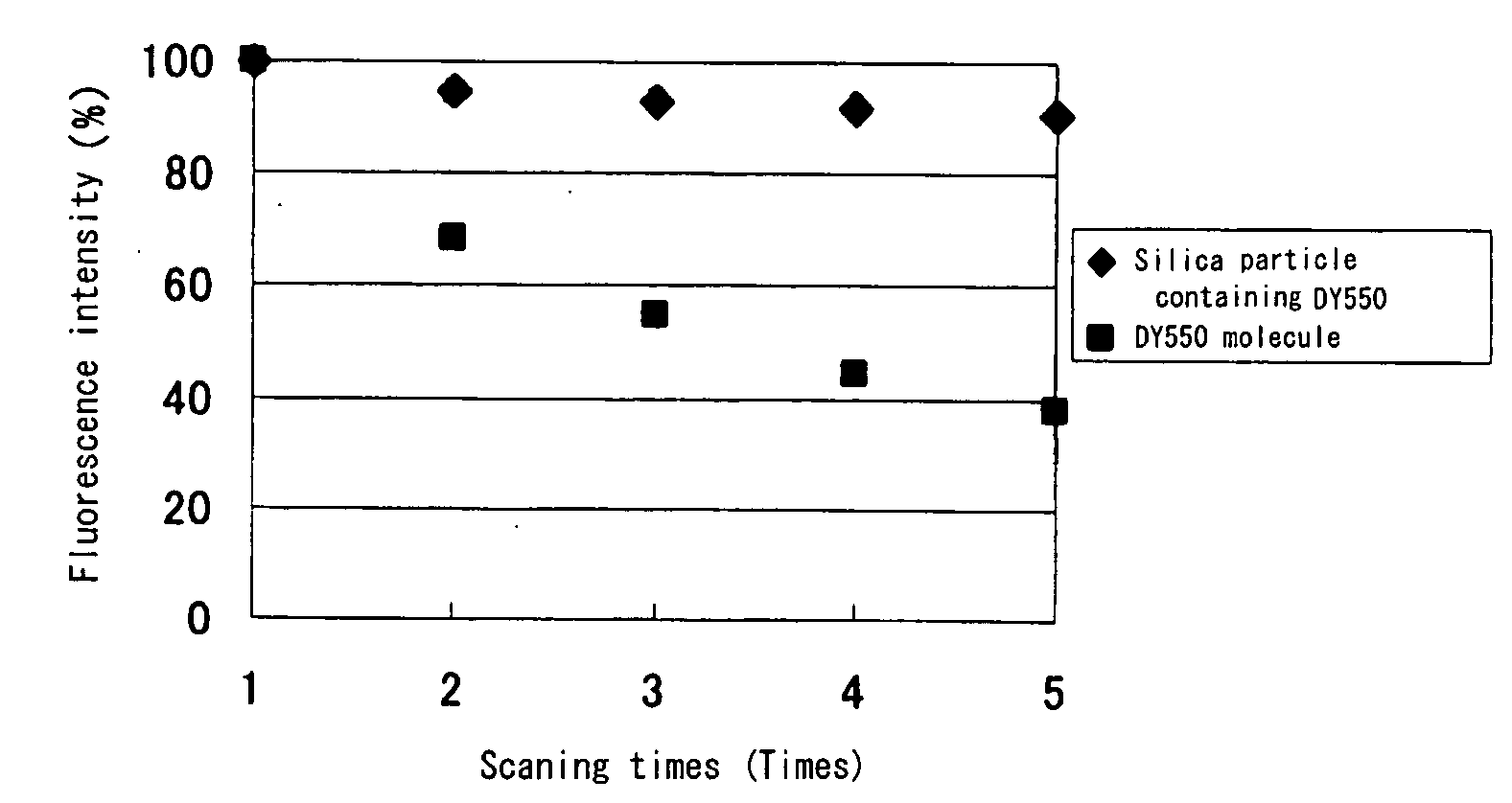
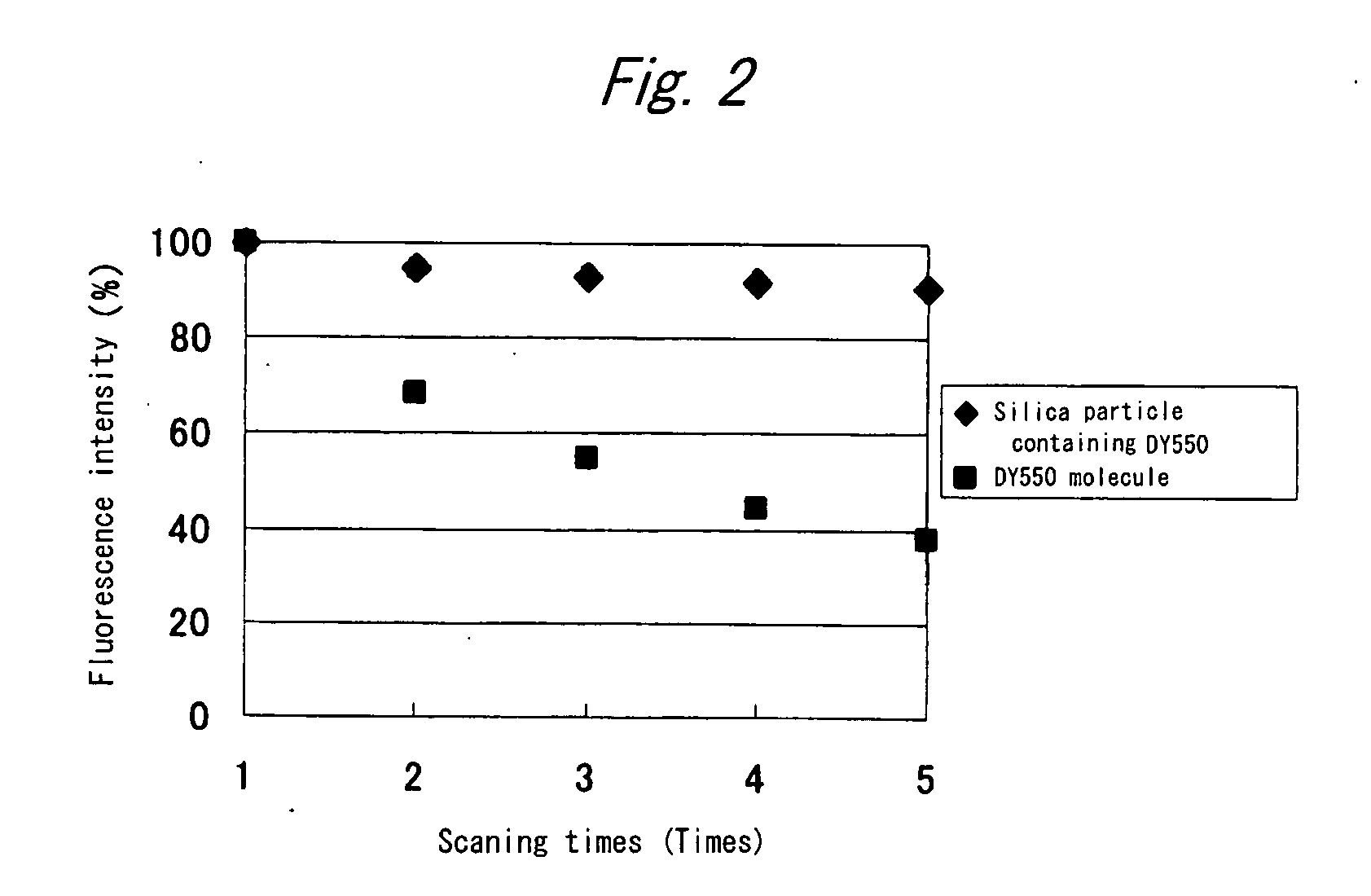
Fluorescent silica nano-particle, fluorescent nano-material, and biochip and assay using the same
ActiveUS20080293584A1Material nanotechnologyMethine/polymethine dyesChemical LinkageColloidal silica
Colloidal silica particles containing a fluorescent dye compound, composed of a silica particle containing a silica component and a fluorescent dye compound chemically bound or adsorbed thereto,wherein the colloidal silica particles containing a fluorescent dye compound have an average diameter of 30 nm or less, and wherein said silica particles are used simultaneously as fluorescent-labelling nanobeads; a fluorescent nano-material comprising said colloidal silica particles; and a biochip and an assay method using the same.
Owner:FURUKAWA ELECTRIC CO LTD +1
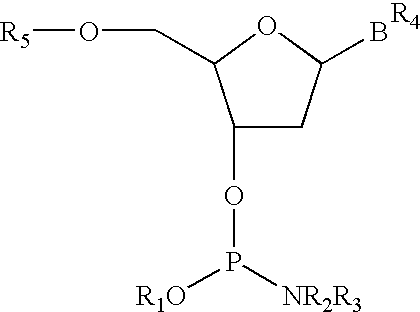
Phosphonate fluorescent dyes and conjugates
ActiveUS7671218B2Silicon organic compoundsMethine/polymethine dyesPhotochemistryAnalytical chemistry
Reagents are provided for the introduction of phosphonate groups into fluorescent dyes. Methods are also provided for preparing dye conjugates.
Owner:ELITECHGROUP MDX LLC
Organic electroluminescent device
ActiveCN106206997AEasy transferImprove quantum efficiencySolid-state devicesSemiconductor/solid-state device manufacturingQuantum efficiencyPhotoluminescence
The invention discloses an organic electroluminescent device. The organic electroluminescent device comprises a light emitting layer and is characterized in that a main body material of the light emitting layer comprises a first main body material and a second main body material, the first main body material and the second main body material are both thermal active delay fluorescent materials, the main body materials are doped with fluorescent dyes, the difference between forbidden bandwidth of the first main body material and the forbidden bandwidth of the second main body material is larger than 0.2eV, and the difference between wavelengths corresponding to peaks is within 50 nanometers after photoluminescence spectrum of the second main body material and absorption spectrums of the fluorescent dyes are highly normalized. More rapid energy transmission to the fluorescent dyes from a singlet state of the light emitting layer of the organic electroluminescent device is achieved, thus, energy transmission to the singlet state from a triplet state of a main body is promoted, high quantum efficiency is acquired, and the efficiency reduction trend is slow with increase of a current.
Owner:BEIJING VISIONOX TECH +1
Silica-coated fluorescent magnetic nanoparticle, preparation method and application
The invention relates to a silica-coated fluorescent magnetic nanoparticle modified by amino or ester dendrimer. A superparamagetism Fe3O4 inner core is prepared by using a coprecipitation method; Fe3O4 encapsulated by silicon dioxide (Fe3O4 and SiO2) is formed by alkaline hydrolyzation of tetraethoxysilane (TEOS); and a fluorescent dye (fluorescein isothiocyanate FITC) is encapsulated in the silicon dioxide layer in a covalent way so as to facilitate tracing a carrier or to be taken as a model medicament. The carrier can be used for fluorescent tracing; and the external surface of the carrier is coated with polyamide-amine (PAMAM) dendrimer, so that the carrier has a great amount of functional groups used for modifying and used for connecting antibodies easier to target a blood brain barrier, and has the potential of becoming a targeted medicament carrier. Research results show that the two materials are injected through arteria carotis communis and can pass through the blood brain barrier. The ester silica-coated fluorescent magnetic nanoparticle is easier to pass through the blood brain barrier than the amino silica-coated fluorescent magnetic nanoparticle. In addition, the carrier can also be used for marking cells.
Owner:CAPITAL UNIVERSITY OF MEDICAL SCIENCES
Heptamethine cyanine active fluorescent probe and preparation method and application thereof
InactiveCN108033907AImprove stabilityReduce background fluorescenceOrganic chemistryFluorescence/phosphorescenceChemical reactionStructural formula
The invention relates to a heptamethine cyanine active fluorescent probe and a preparation method and application thereof. The structural formula of the heptamethine cyanine active fluorescent probe is as shown in the specification, wherein X=II-IX; each of R1 and R2 is (CH2)mCH3, (CH2)nOH, (CH2CH2O)pCH3 and CH2C6H5; each of R3 and R4 is H, SO3H, SO3Na and SO3K; each of a, b, c, d, e, f and g is 2-8; each of n, m and p is 1-10. The heptamethine cyanine active fluorescent probe has the advantages that the heptamethine cyanine active fluorescent probe is based on near-infrared long-wave heptamethine cyanine dye, indoline is selected as the aroma parent nucleus to increase fluorescence intensity, and methenyl chain intermediate cyclohexene rigid bridging enhances stability; nitrogen derivatives with chemical reactivity sites are used to perform nucleophilic substitution on the meso-position of the heptamethine cyanine parent dye, and accordingly Stokes shift and active chemical groups areincreased greatly to facilitate the fluorescent labeling of various substances; the fluorescent probe is of a symmetrical structure, preparation and purification processes are simplified, and cost islowered favorably; the probe can be used as the fluorescent labeling probe of biological molecules such as high-sensitivity protein, sugar and DNA and nano carriers to perform cell or living-body horizontal fluorescence imaging, and the like.
Owner:INST OF BIOMEDICAL ENG CHINESE ACAD OF MEDICAL SCI
Acrylon fluorescent dyeing method
InactiveCN101424055ASynthetic reaction is simpleHigh yieldStyryl dyesDyeing processSodium acetateVisibility
The invention relates to a method for fluorescent staining of orlon, which belongs to the field of dye synthesis and textile dyeing. Stilbazole salt is taken as a cation fluorescent dye, 0.2 and 2.5 percent owf fluorescent dye, 0.5g / L Peregal 0, a 1.0 percent owf leveling agent and 4g / L glauber salt are mixed to prepare a dye solution, acetic acid and a sodium acetate buffer solution are used to adjust the pH value of the dye solution to 5, and the dyeing processing with the dyeing bath ratio of 100 to 1 is performed to obtain a fluorescent orlon fabric. The stilbazole salt provided by the method has simple synthesis reaction, has relative flexibility in molecular design and assembly, and also has good linear optical performance and non-linear optical performance; and under specific dye bath concentration, the dyed orlon fabric satisfies the coordinate range of orange color stipulated in European EN471 standard High-visibility Warning Clothing for Professional Use-Test Methods (2003).
Owner:SUZHOU LONGJIE SPECIAL FIBER
Fluorescent inks for writing instruments using fluorescent dyes and white pigments
A non-aqueous fluorescent ink composition suitable for use in writing instruments is described comprising a soluble fluorescent colorant, a solvent, and an opacifying agent. The ink composition may also include resins, stabilizers, surfactants, lubricants and other inert additives. The fluorescent ink composition exhibits a brilliant appearance in masscolor and when written on paper.
Owner:SOC DITE SOC BIC
Reversible solid photochromic fluorescence ink material and application thereof
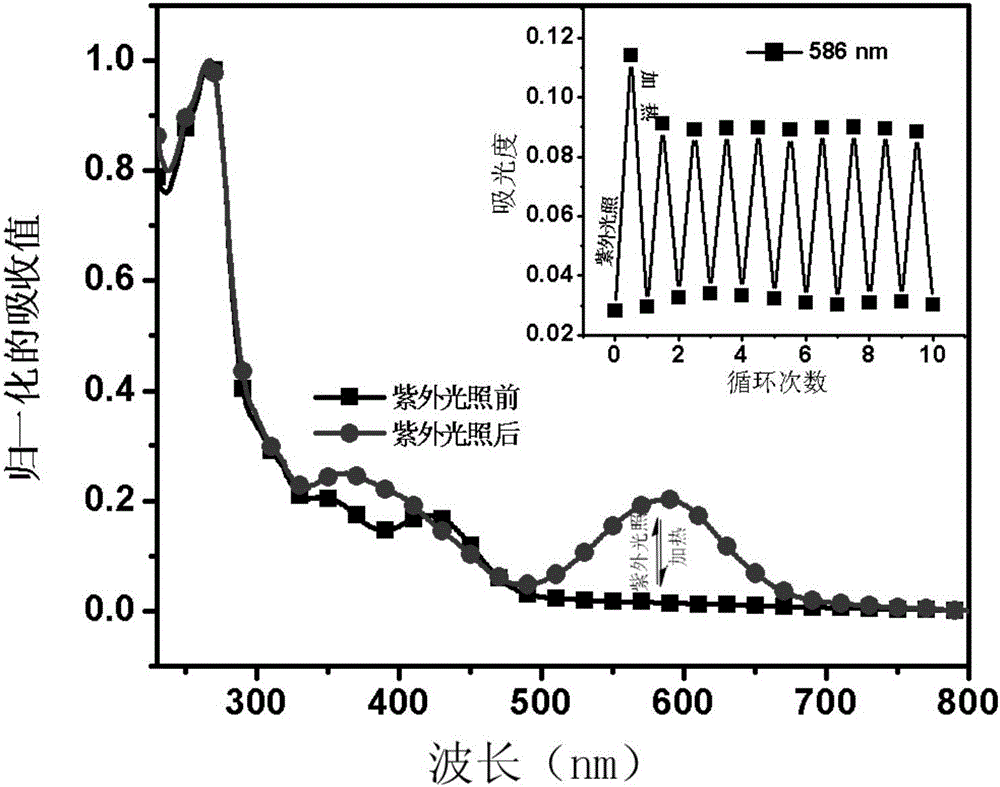
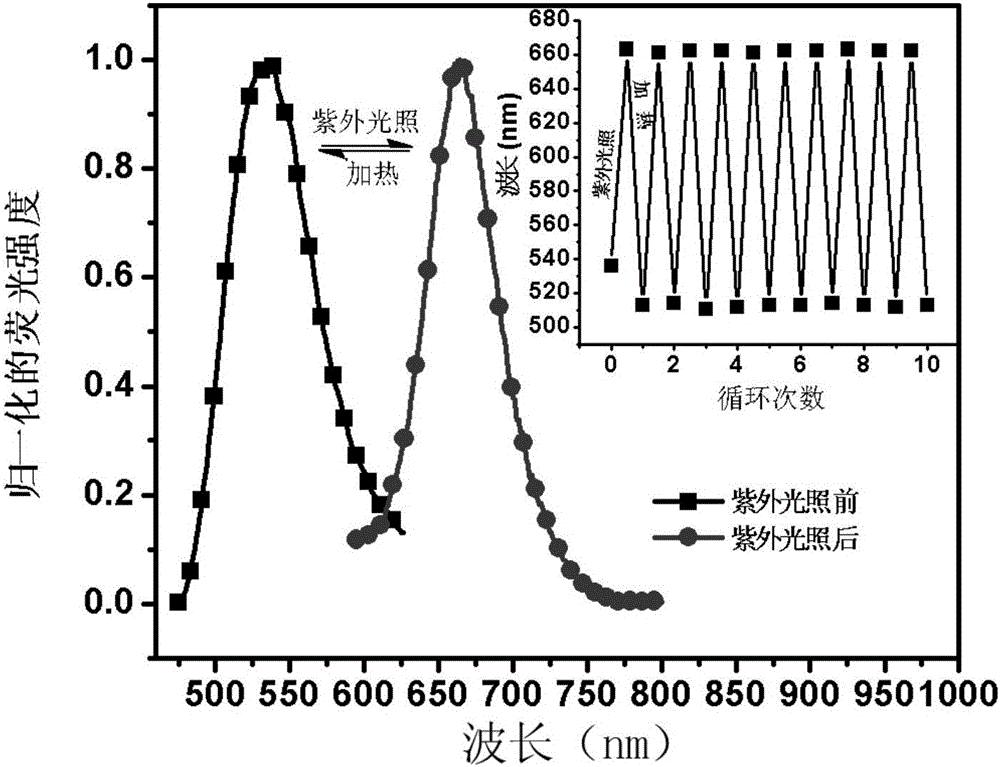
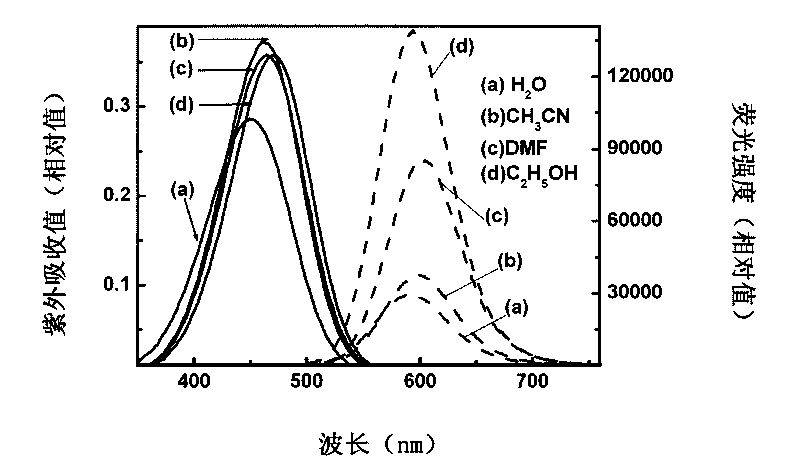
ActiveCN105670389AImprove stabilityGood reversibilityOrganic chemistryInksFluorescenceUltraviolet lights
The invention relates to a reversible solid photochromic fluorescence ink material and application thereof, belonging to the technical field of photochromic materials. The particular molecular structure uses aromatic ring or fragrant heterocyclic ring substituent group and spiropyrane as basic structural units, the structural formula is disclosed in the specification, and the material is novel fluorescent dyes synthesized by esterification reaction. The research detects that the dyes have different spectral absorption and (or) fluorescence dual color variations before and after the ultraviolet irradiation; the absorption and fluorescence color variations caused by the ultraviolet light can reversibly recover under certain temperature heating conditions to the absorption and fluorescence color before the ultraviolet irradiation; and thus, the material has excellent stability, reversibility and fatigue resistance. The ink material can be used independently, or compounded with a polymer, ink or the like to be used as a photochromic material. The ink material is applicable to the fields of safety forgery prevention, safety inks, display devices, fluorescent transduction, information storage and the like.
Owner:JILIN UNIV
Method for preparing fluorescent fiber membrane
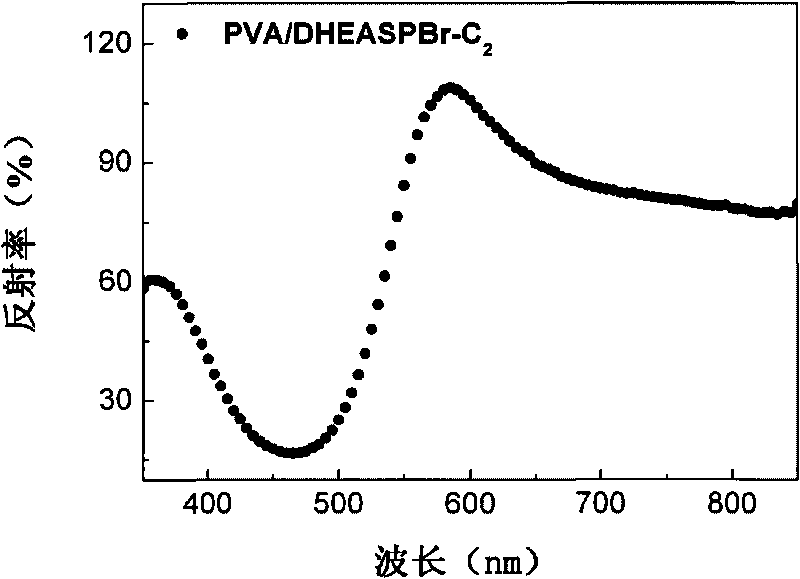
ActiveCN101735801AExcellent linear optical performanceEasy to getMonocomponent synthetic polymer artificial filamentLuminescent compositionsFiberSolubility
The invention discloses a method for preparing fluorescent fiber membrane, in particular to a method taking derivatives of styrene pyridinium as fluorescent dye to blend with polyving akohol (PVA) to prepare spinning solution and perform electrostatic spinning. The method comprises the following steps: taking styrene pyridinium as fluorescent dye, mixing the fluorescent dye with the mass concentration of 1-10% (fluorescent dye / PVA) into PVA water solution, and then performing electrostatic spinning to obtain the orange fluorescent fiber membrane. The styrene pyridinium provided by the invention has good liner optical property, good water solubility and good consistency with PVA water solution; the highest reflectivity of the obtained fibrous membranes is more than 100%, and the wavelengthcorresponding to the max peak is 605nm; and the fluorescent emission spectrum excited by laser with the excitation wavelength of 254nm is within the range of 500-750nm, and CIE color coordinate calculated value is within a yellowish orange fluorescent zone, thus having application prospect.
Owner:SUZHOU UNIV
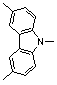
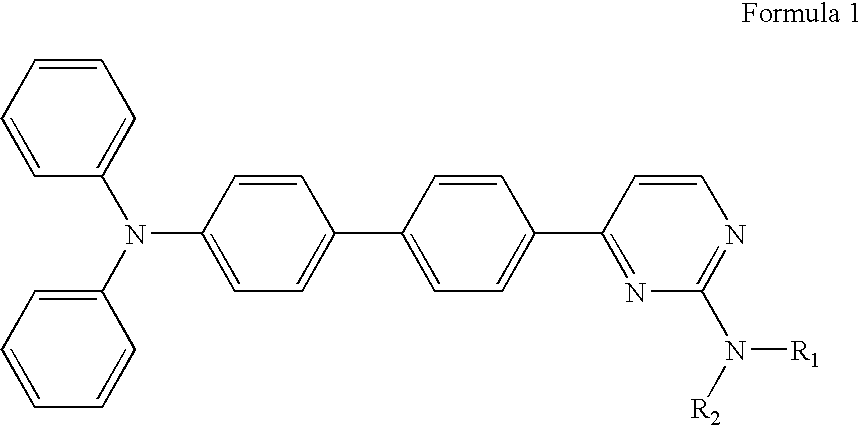
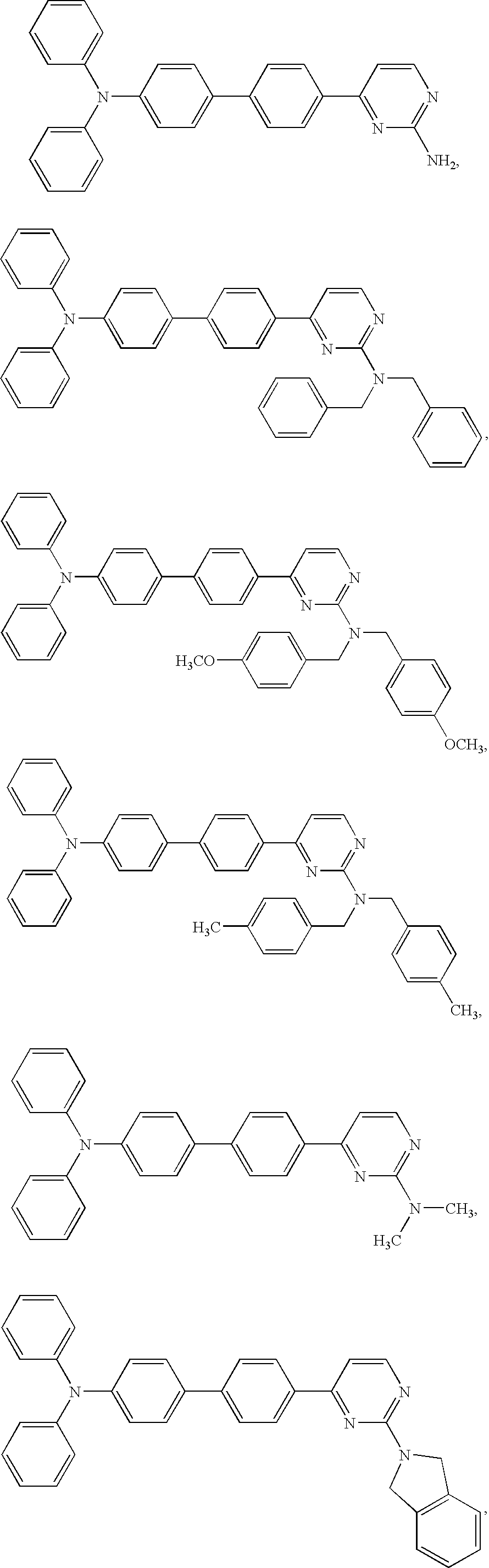
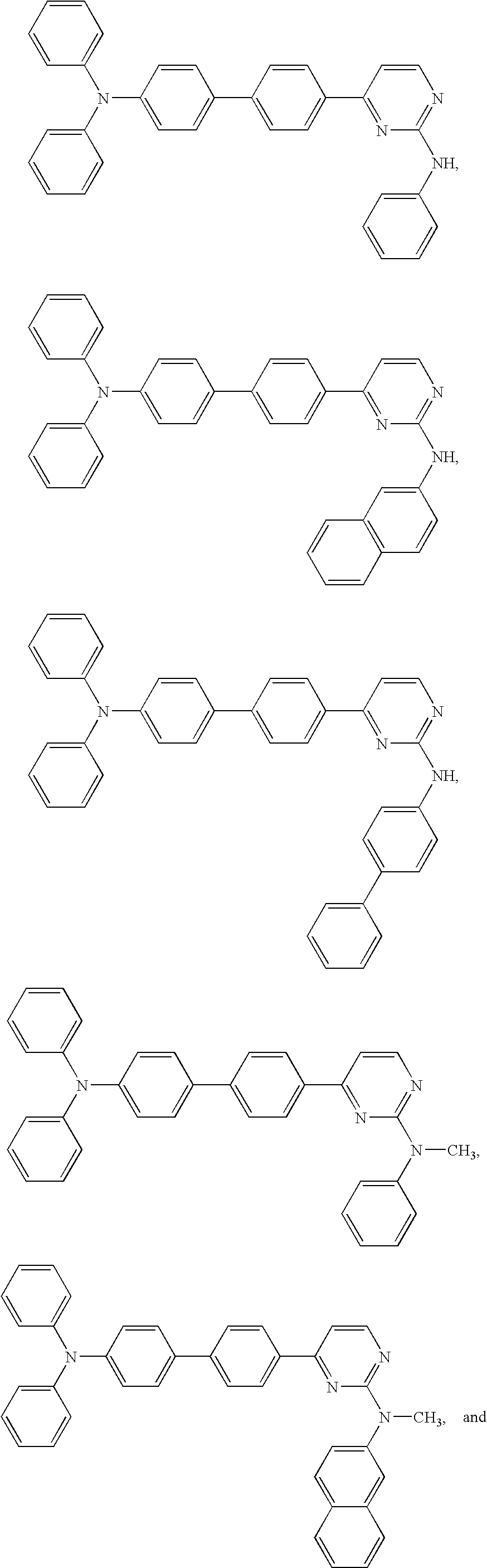
Diphenyl amine derivatives having luminescence property
ActiveUS20100063281A1Organic chemistryLuminescent compositionsOrganic electroluminescenceLuminescence
The present invention relates to diphenyl amine derivatives having luminescent properties, and particularly to diphenyl amines substituted with various electron-donating or electron-withdrawing groups. The compounds of the present invention show blue or blue-green luminescence, and luminescence of the compounds herein can be achieved by UV or visible light, thus being useful as a fluorescent dye or fluorescent material for an organic electroluminescence device or display.
Owner:KOREA INST OF SCI & TECH
High-polymer vesicle containing AIE (aggregation-induced emission) molecules as well as preparation method and application of high-polymer vesicle
InactiveCN105524441AOvercoming the disadvantages of aggregation-induced fluorescence quenchingAvoid identificationOrganic active ingredientsPowder deliveryCopolymerClinical imaging
The invention belongs to the field of high-polymer materials and discloses a high-polymer vesicle containing AIE (aggregation-induced emission) molecules as well as a preparation method and an application of the high-polymer vesicle. The high-polymer vesicle is mainly formed through self-assembly of the AIE molecules and amphiphilic block copolymers; the outer layer and the inner layer of a high-polymer vesicle film are hydrophillic layers, each hydrophillic layer is formed by hydrophilic chain sections in the amphiphilic block copolymers, and a film intermediate layer between the outer layer and the inner layer is formed by hydrophobic chain sections in the amphiphilic block copolymers and the AIE molecules. The high-polymer vesicle has high fluorescence intensity and can meet different fluorescent staining demands. The preparation method is simple and feasible, the production efficiency is high, and the repeatability is good. The high-polymer vesicle can be used for related fields of optical bioimaging, biological detection and clinical imaging, high-polymer vesicle research and the like.
Owner:SOUTH CHINA UNIV OF TECH
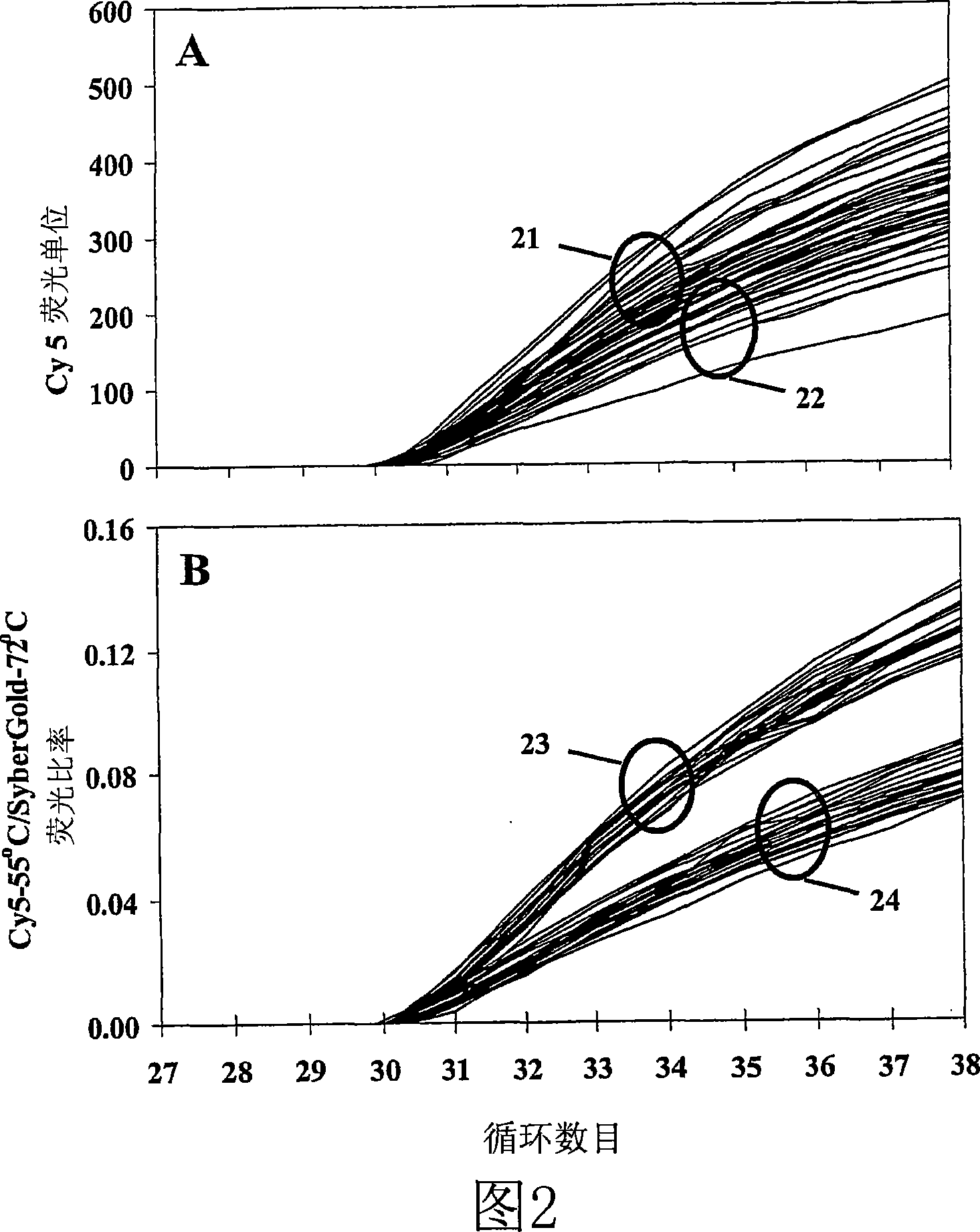
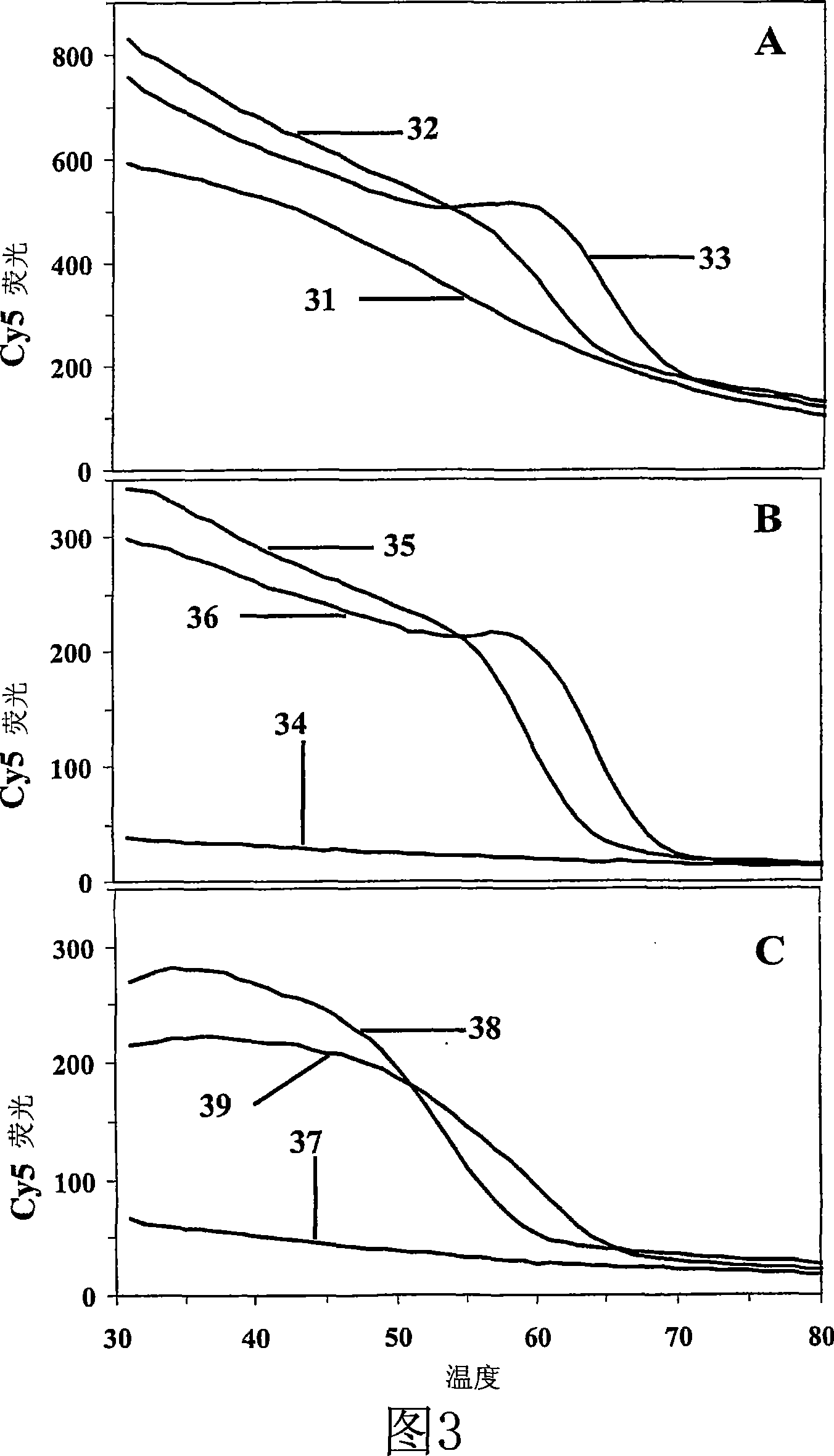
Primers, probes and methods for nucleic acid amplification
Homogenous detection during or following PCR amplification, preferably LATE-PCR, utilizing fluorescent DNA dye and indirectly excitable labeled primers and probes, improves reproducibility and quantification. Low-temperature homogeneous detection during or following non-symmetric PCR amplification, preferably LATE-PCR, utilizing fluorescent DNA dye and indirectly excitable labeled mismatch-tolerant probes permits analysis of complex targets. Sequencing sample preparation methods following LATE-PCR amplifications reduce complexity and permit ''single-tube'' processing.
Owner:BRANDEIS UNIV