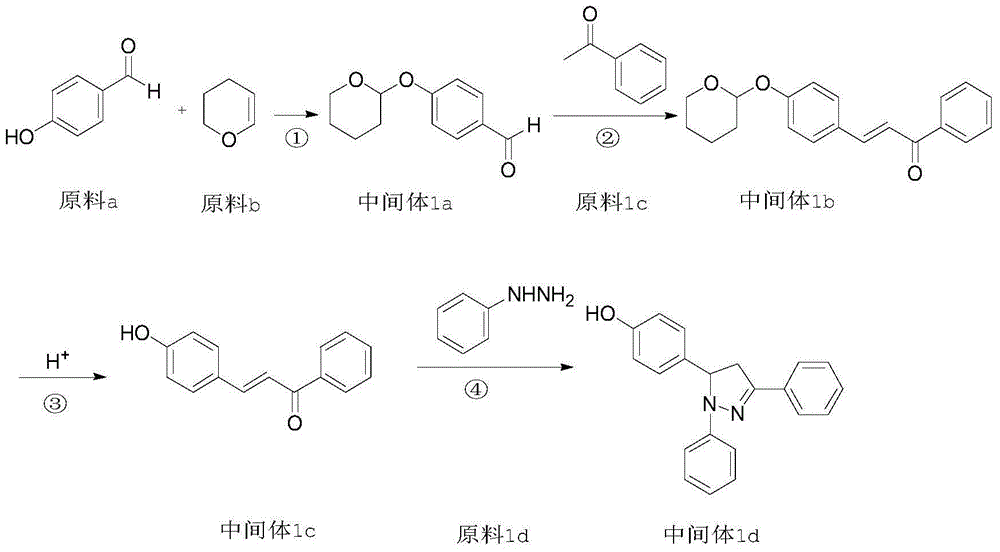
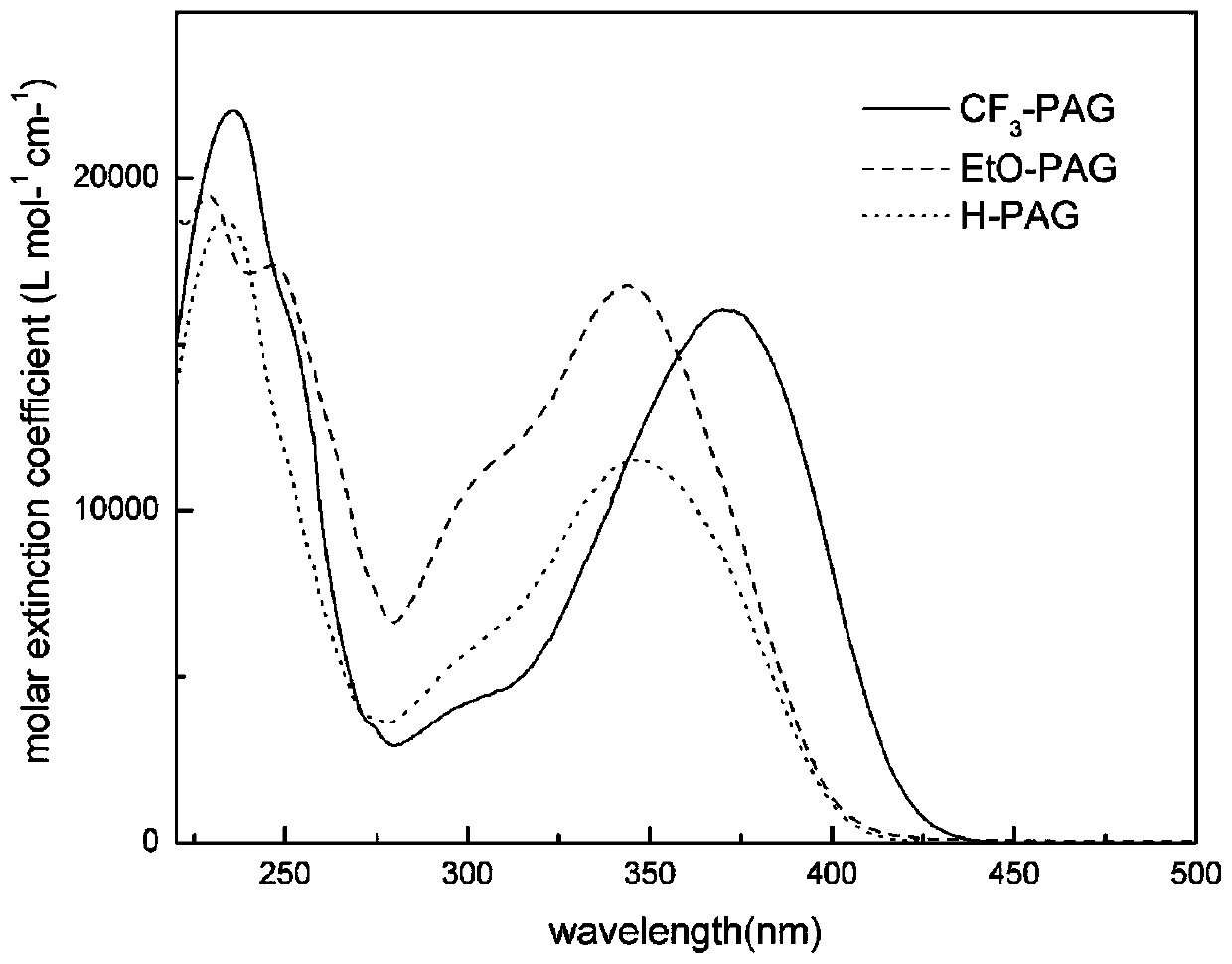
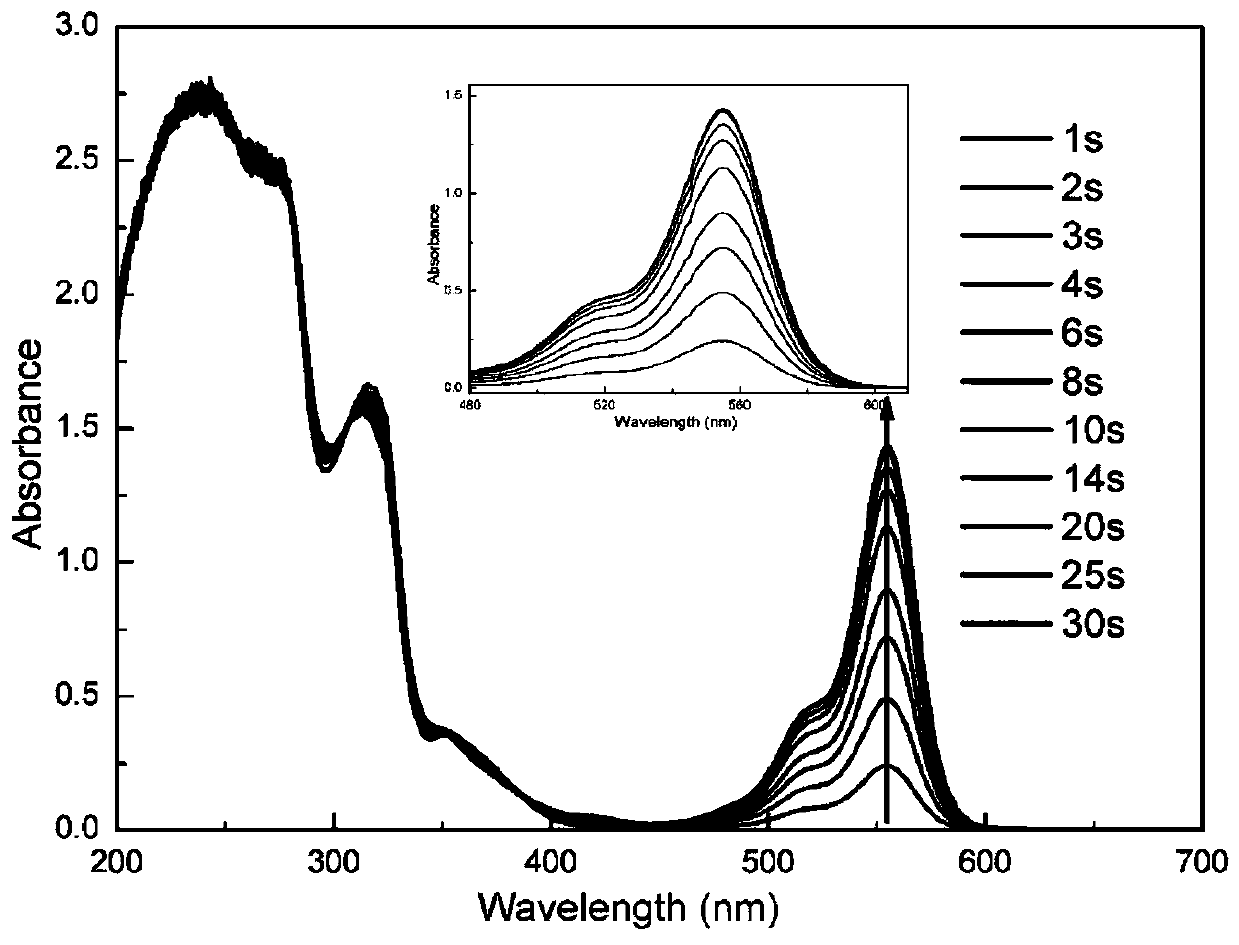
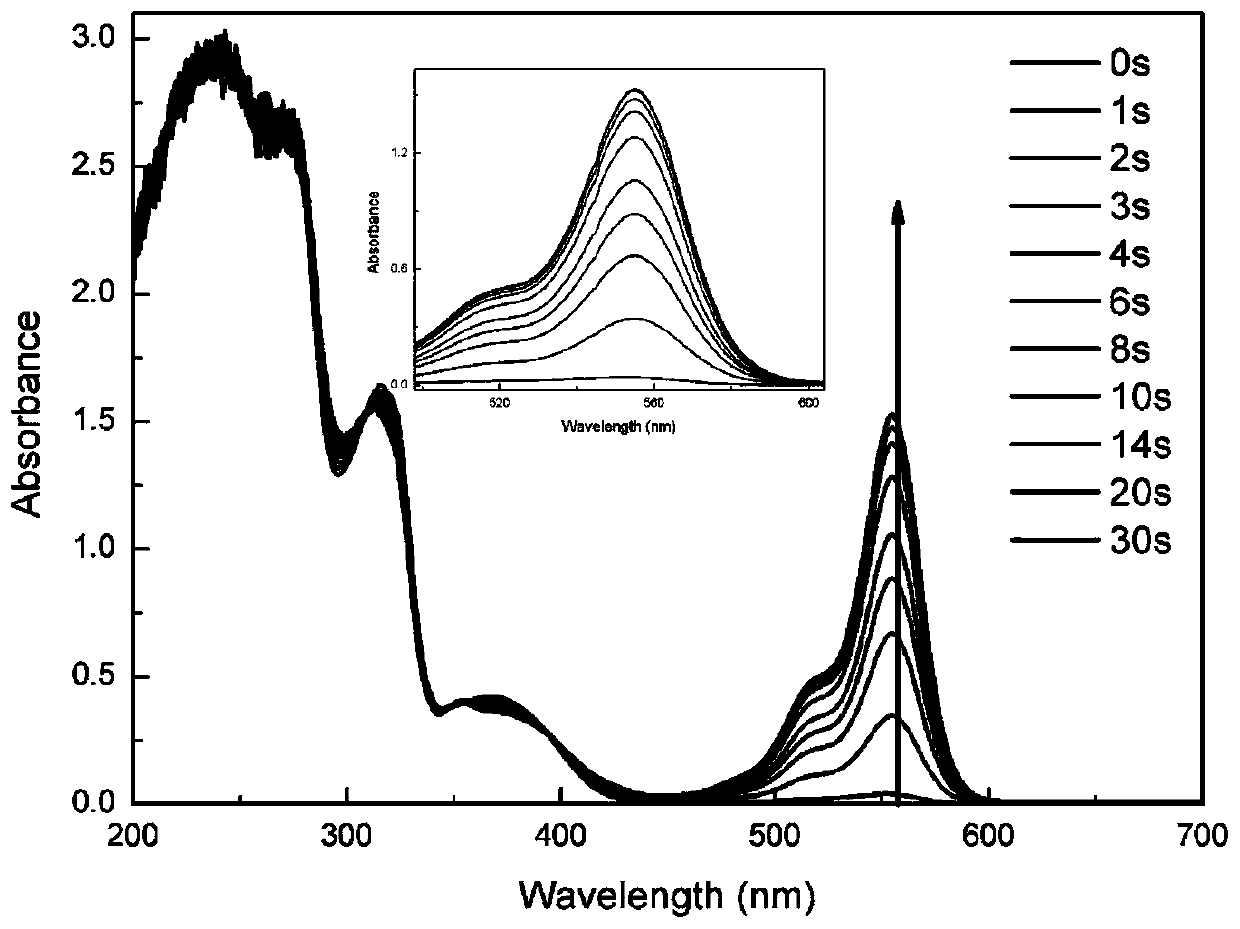
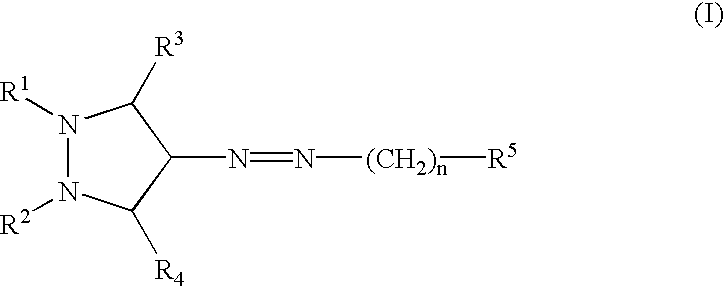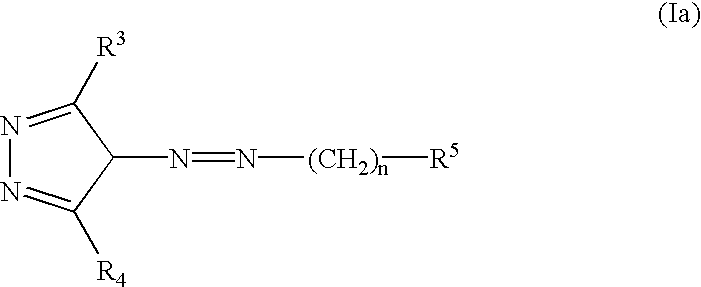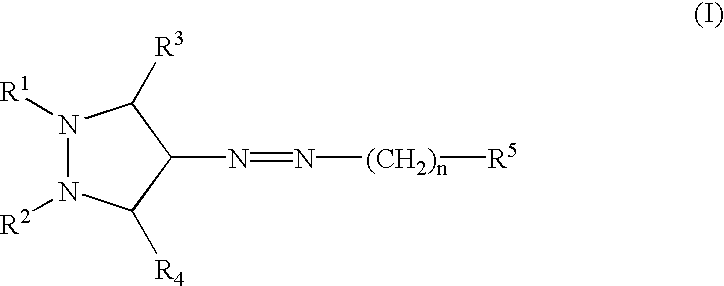Patents
Literature
187 results about "Pyrazoline" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
Pyrazoline is a heterocyclic chemical compound with the molecular formula C₃H₆N₂.
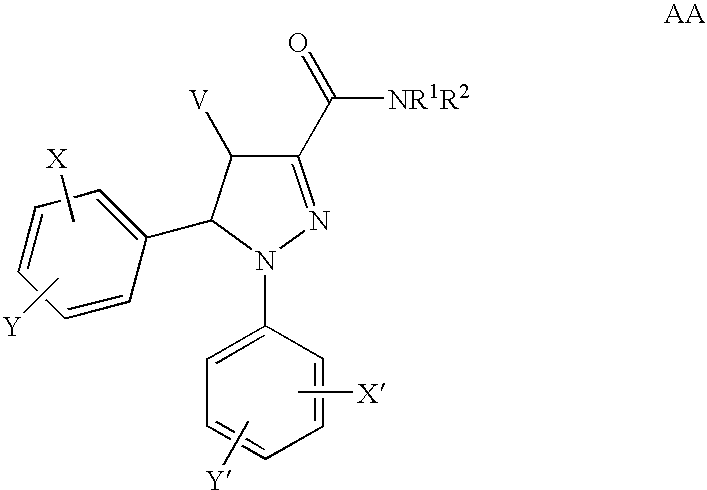
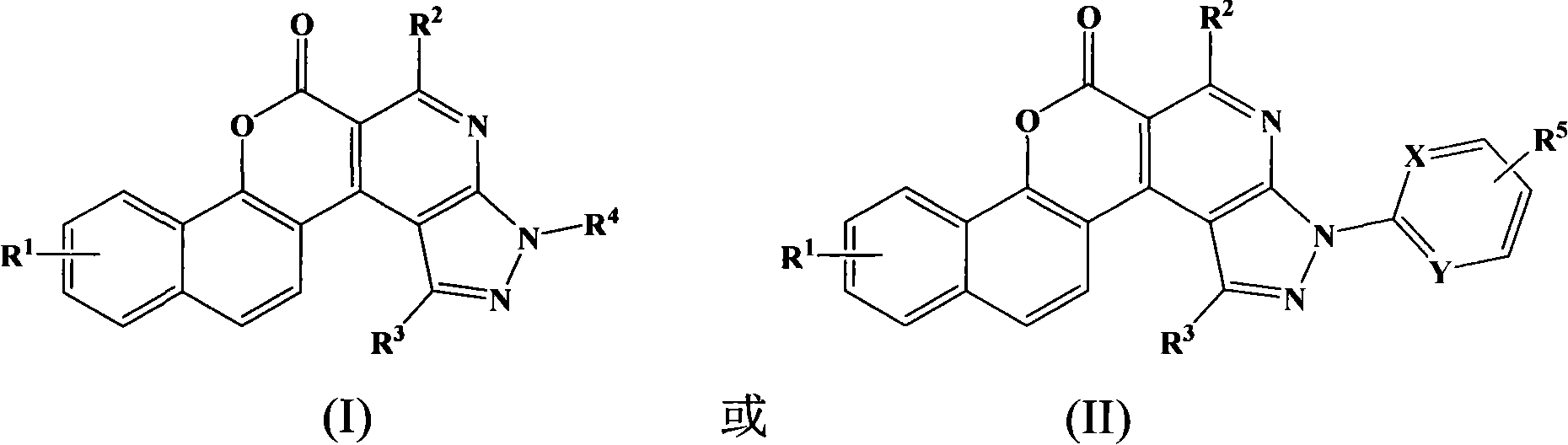
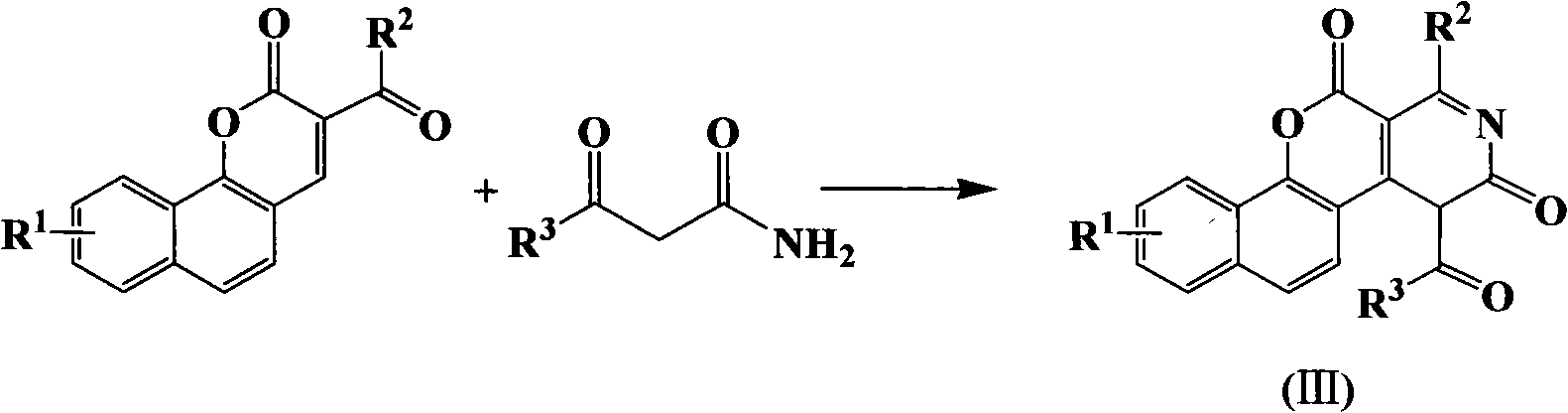
1H-pyrazolo[3,4-b]pyridines and therapeutic uses thereof
Provided herein are compounds according to Formula I and pharmaceutically acceptable salts thereof, and compositions comprising the same, for use in various methods, including treating cancers such as colon, ovarian, pancreatic, breast, liver, prostate and hematologic cancers:
Owner:BIOSPLICE THERAPEUTICS INC
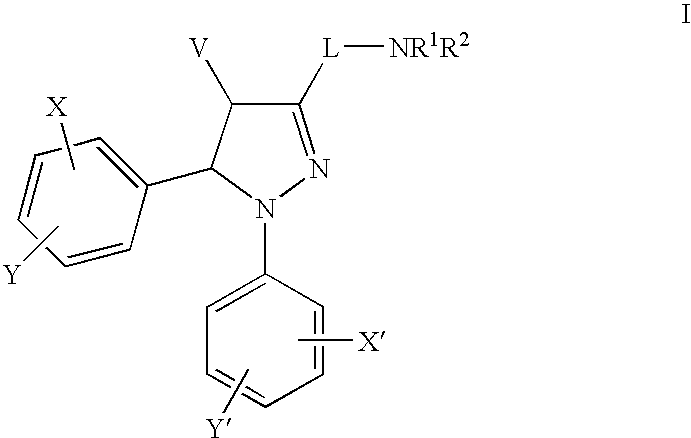
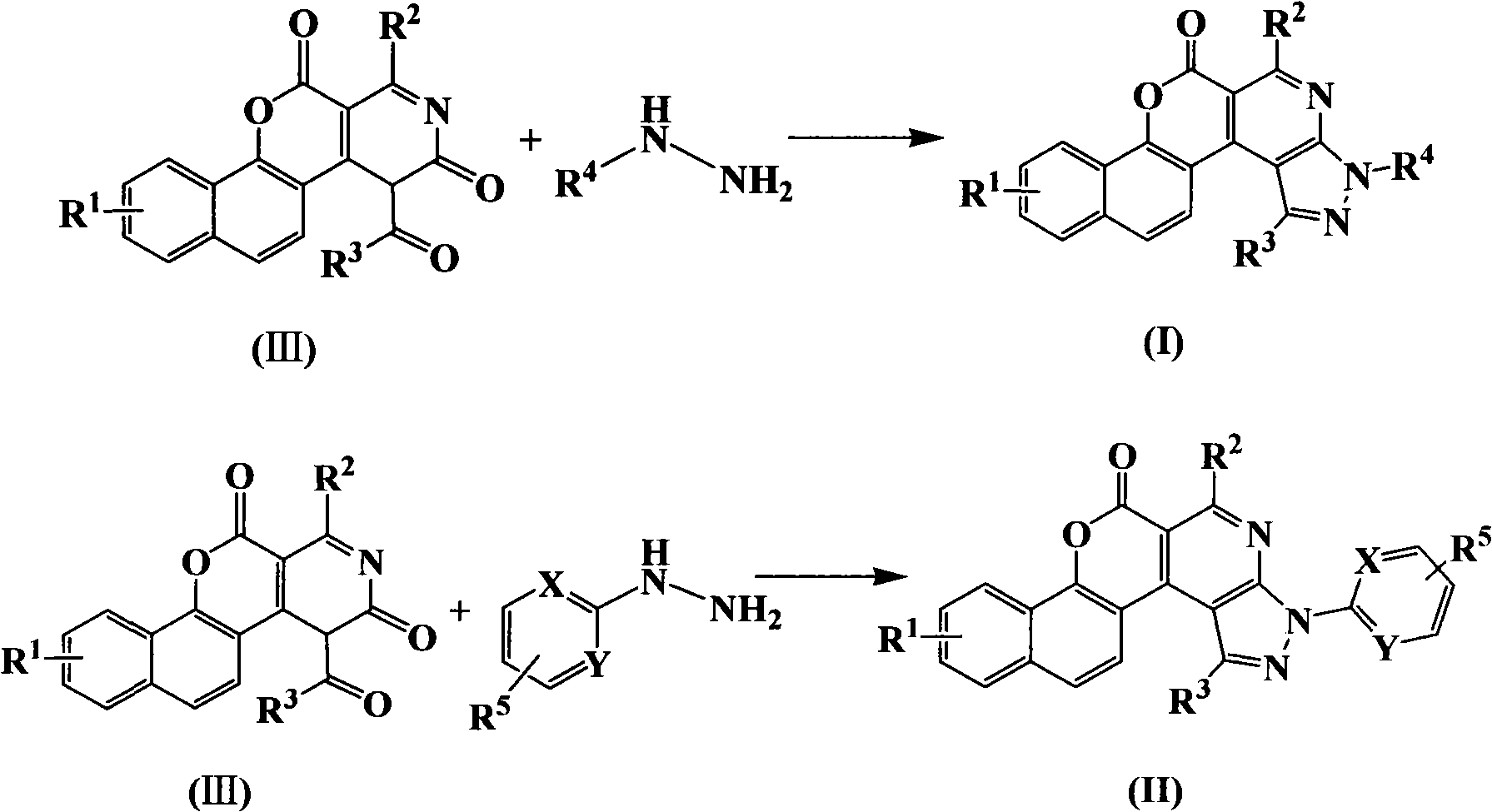
1H-pyrazolo[3,4-b]pyridines and therapeutic uses thereof
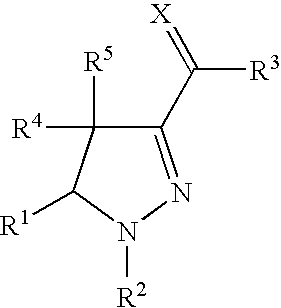
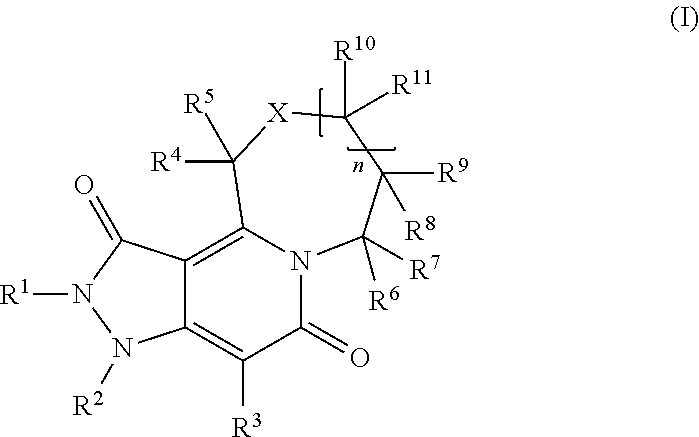
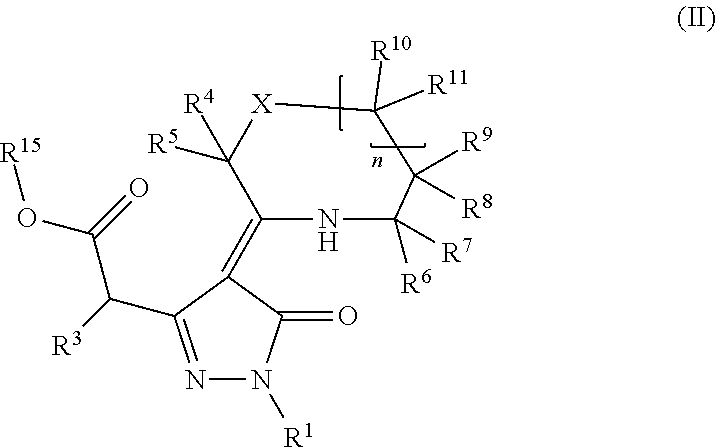
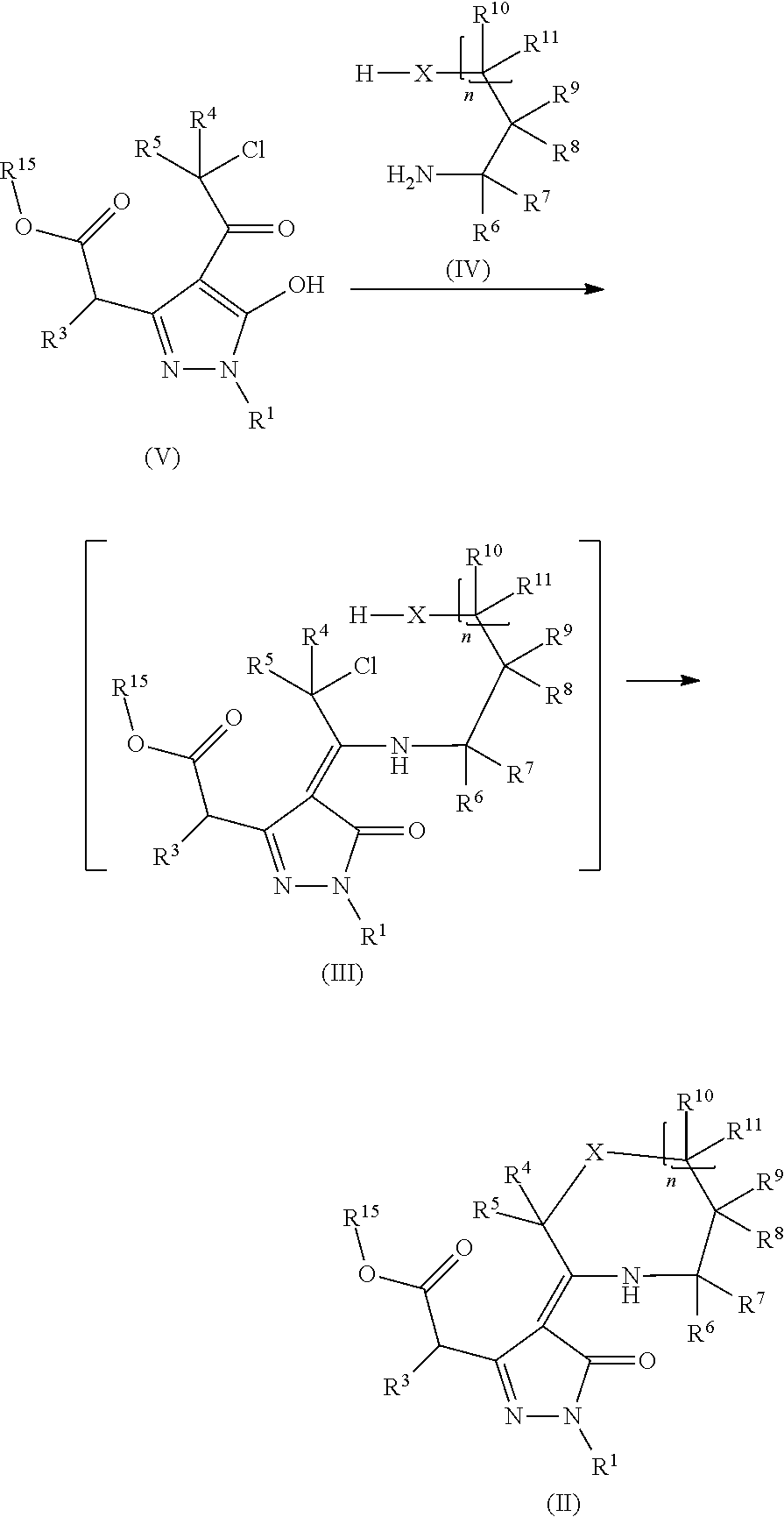
Provided herein are compounds according to Formulas (I) or (II) and pharmaceutically acceptable salts thereof, and compositions comprising the same, for use in various methods, including treating cancer, abnormal cellular proliferation, angiogenesis, Alzheimer's disease, lung disease, osteoarthritis, idiopathic pulmonary fibrosis and neurological conditions / disorders / diseases.
Owner:BIOSPLICE THERAPEUTICS INC
Pyrazole compounds
InactiveUS20030060453A1Useful in treatmentBiocideMonoazo dyesLymphoproliferative diseaseAngiogenesis growth factor
Pharmaceutical compositions and compounds are provided. The compounds of the invention demonstrate anti-proliferative activity, and may promote apoptosis in cells lacking normal regulation of cell cycle and death. In one embodiment of the invention, pharmaceutical compositions of the compounds in combination with a physiologically acceptable carrier are provided. The pharmaceutical compositions are useful in the treatment of hyperproliferative disorders, which disorders include tumor growth, lymphoproliferative diseases, angiogenesis. The compounds of the invention are substituted pyrazoles and pyrazolines.
Owner:DERMIRA CANADA
Substituted n-phenyl-5-phenyl-pyrazolin-3-yl amides as cannabinoid receptor antagonists/inverse agonists useful for treating obesity
The present invention provides novel pyrazolines that are useful as cannabinoid receptor antagonists / inverse agonists and pharmaceutical compositions thereof and methods of using the same for treating obesity, diabetes, dyslipidemias, cardiovascular disorders, hepatic disorders, and a combination thereof.
Owner:JENRIN DISCOVERY
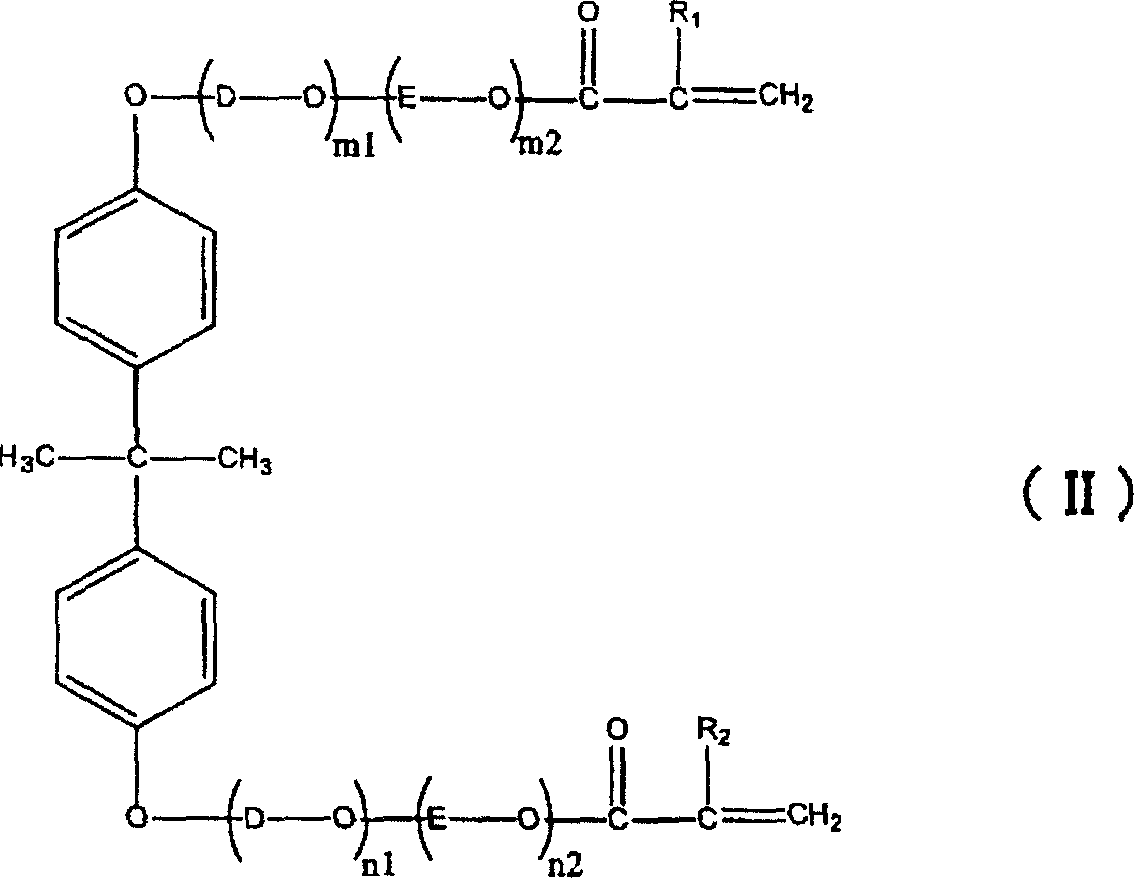
Photosensitive resin composition and photosensitive resin laminate using the same
ActiveCN1945429AIncreased sensitivityIncrease photosensitivitySemiconductor/solid-state device manufacturingPhotomechanical exposure apparatusImage resolutionAqueous solution
The present invention relates to a photosensitive resin composition superior in sensitivity with respect to an exposure light source having the wavelength of 405+ / -10 nm and resolution, having satisfactory tenting properties, and capable of being developed by an alkali aqueous solution, a photosensitive resin laminated body using the photosensitive resin composition, a method for forming a resist pattern on the substrate using the photosensitive resin stacked body, and the use of the resist pattern. The photosensitive resin composition contains: (a) a thermoplastic copolymer of 20-90% including alpha, beta-unsaturated carboxyl groups containing a monomer as a copolymerization component, and having an acid equivalent of 100-600 and weight average molecular weight of 5,000-500,000; (b) an addition polymerization monomer of 5-75% having a terminal ethylenic unsaturated group; (c) a photopolymerization initiator of 0.01-30%; and (d) a specific pyrazoline compound of 0.001-10%, and includes, (b) as the addition-polymerizable monomer containing addition-polymerizable monomer with specific structure.
Owner:ASAHI KASEI KK
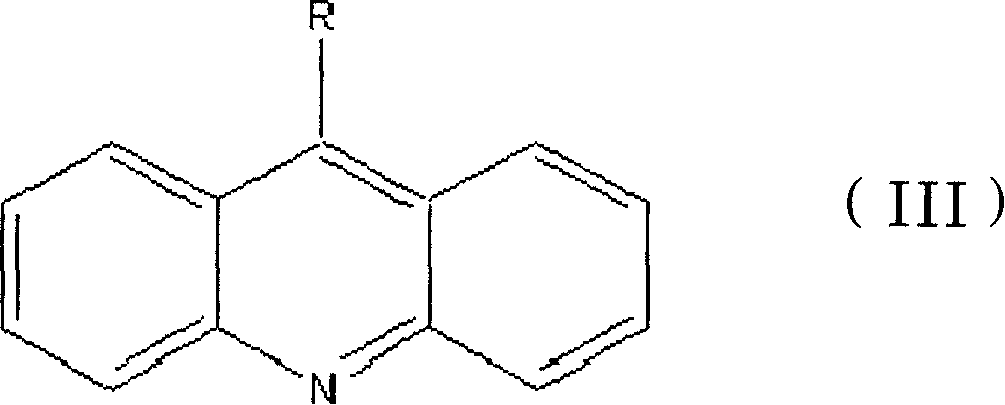
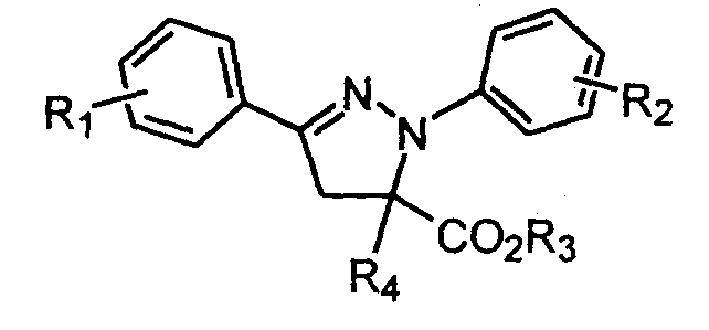
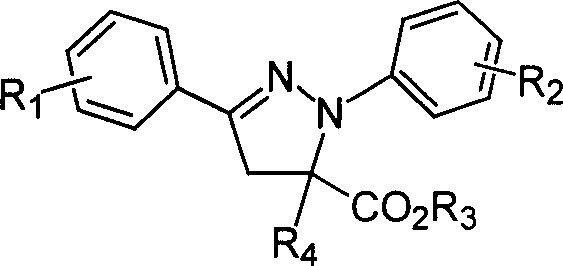
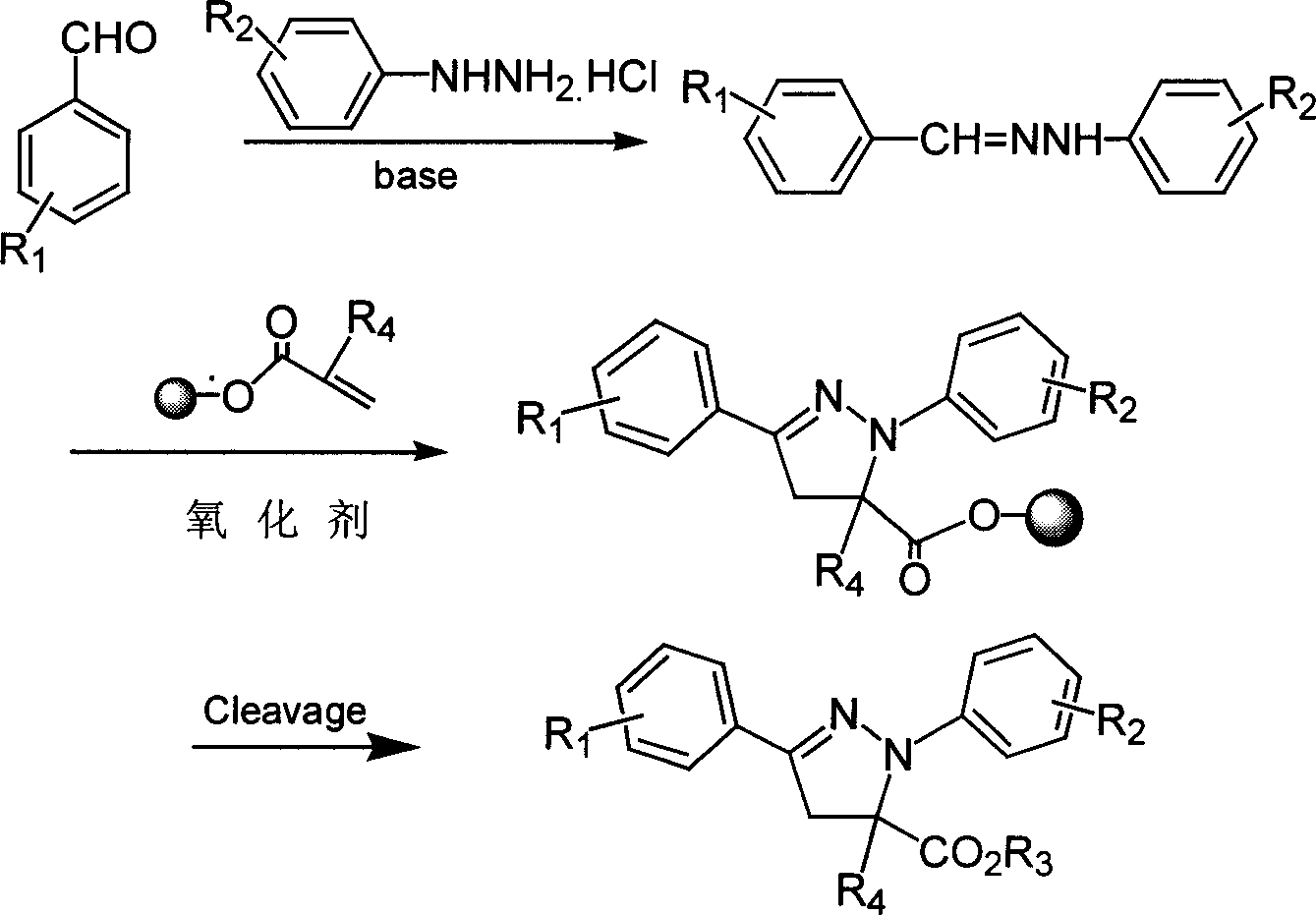
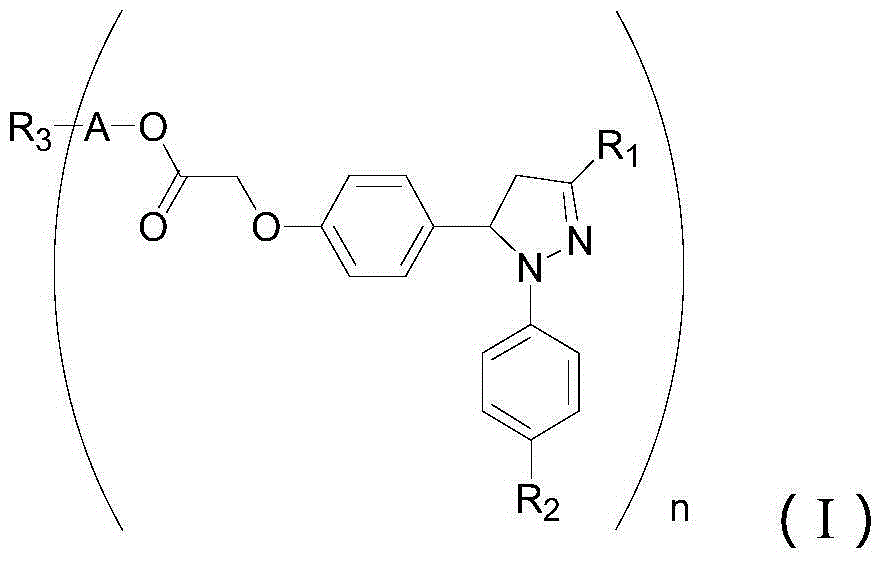
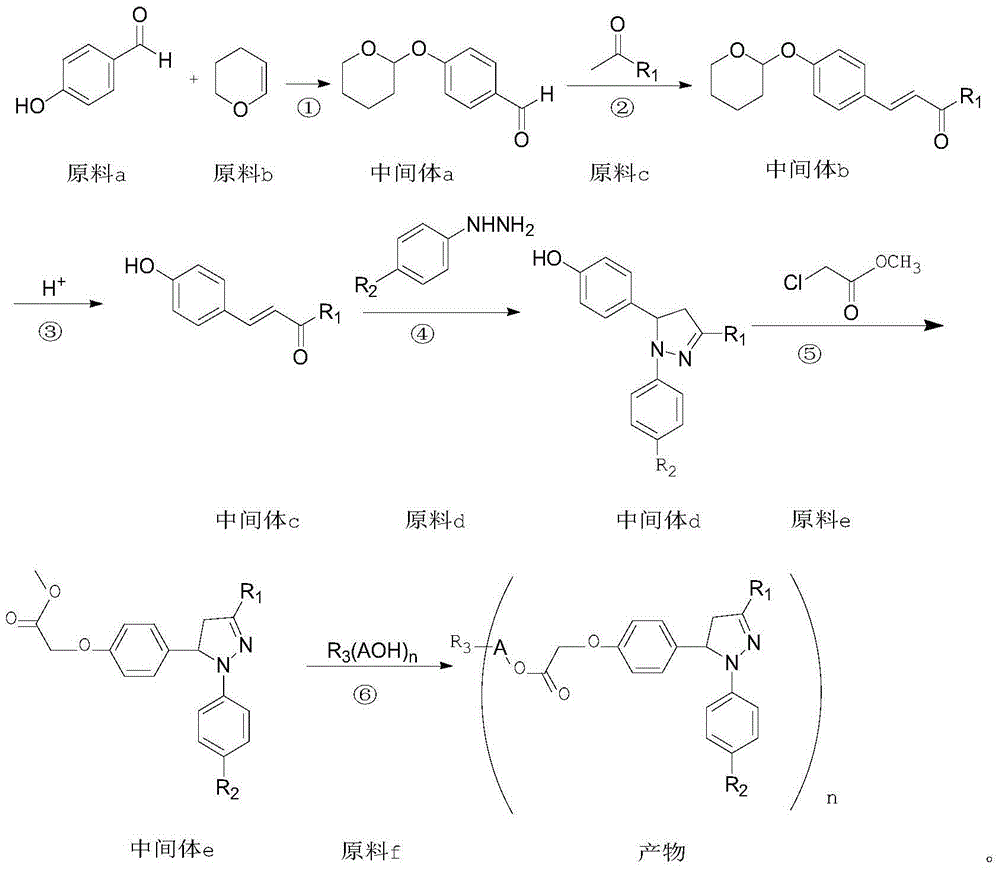
1,3-aryl group substituted pyrazoline whose 5 position contains ester gruop and its liquid phase synthesis method
InactiveCN1515557AEasy and fastHigh reaction yieldOrganic chemistryHydrazine compoundSynthesis methods
The present invention discloses a kind of 1,3-aryl substituted pyrazoline whose delta-position contains ester group and its liquid phase synthesis method. In the organic solvent, in the presence of alkali the substituted acryloyl chlorine is coupled on the soluble resin, under the action of organic alkali the isoequivalent aromatic aldehyde and aromatic hydrazine hydrochloride are mixed to obtain the correspondent hydrazone, then the substituted acrylic ester loaded by soluble resin and oxidant are added, and the above-mentioned materials are undergone the ring-forming reaction of one-pot method so as to obtain the 1,3-aryl substituted pyrazoline whose delta-position contains ester group which is loaded on the resin. In the alcohol solvent and under the catalysis of correspondent alcohol sodium the pyrazoline can be separated from soluble resin so as to obtain the invented product with high yield rate and high purity. It can be developed into medicine, pesticide and light sensitive material.
Owner:ZHEJIANG UNIV
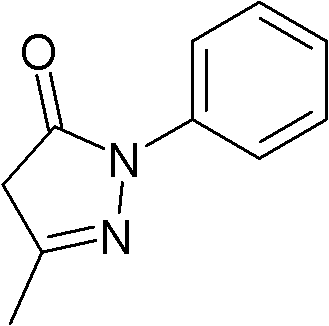
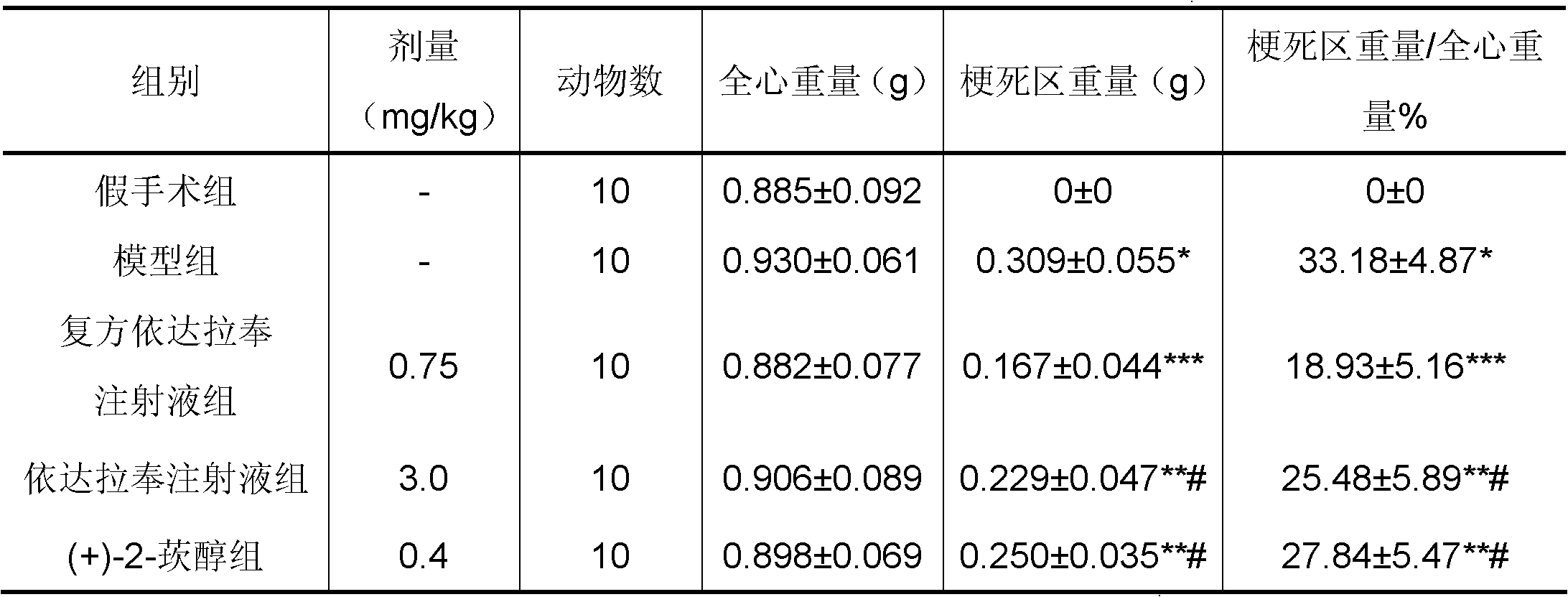
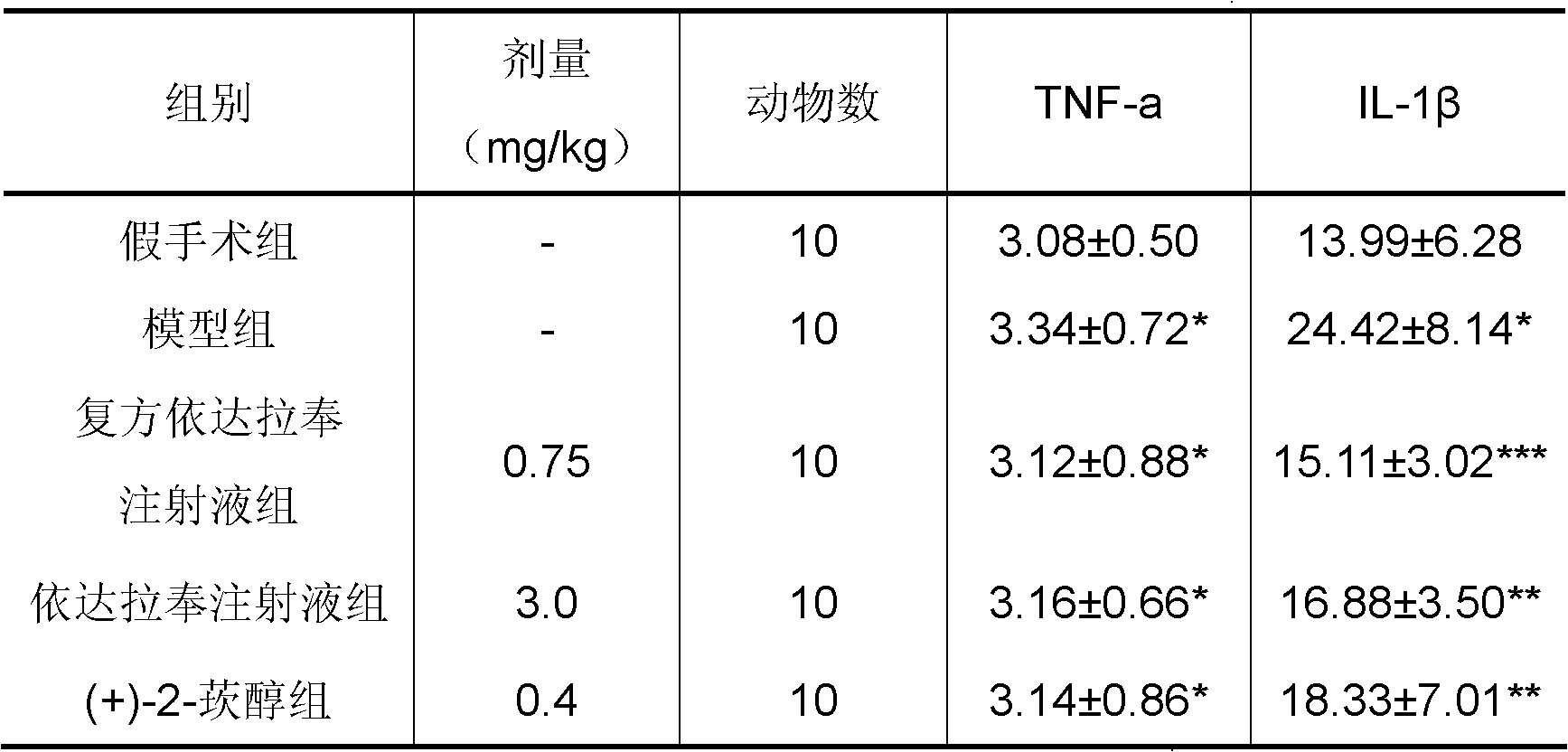
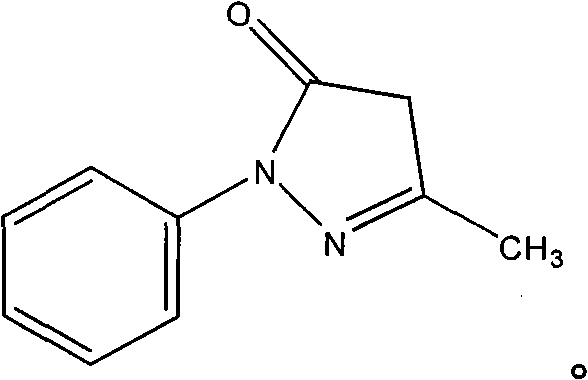
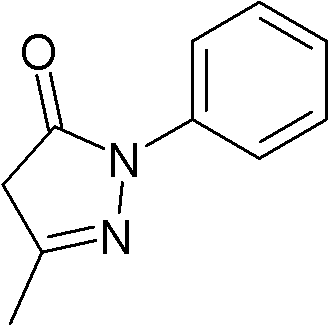
New application of 3-methyl-1-phenyl-2-pyrazoline-5-ketone and 2-borneol composition
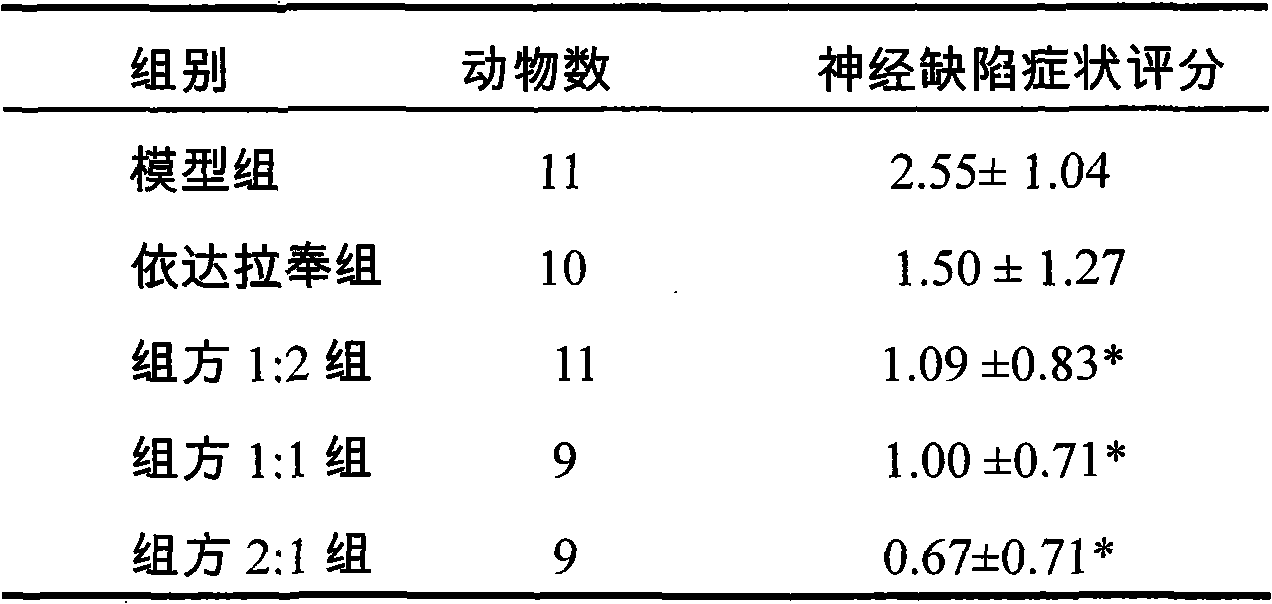
ActiveCN102579432AProtectiveLow therapeutic useHydroxy compound active ingredientsCardiovascular disorderReperfusion injuryCardiac muscle
The invention relates to application of an edaravone and borneol medicinal composition to preparation of a medicament for preventing and / or treating myocardial ischemia-reperfusion injury.
Owner:NANJING SIMCERE DONGYUAN PHARM CO LTD +1
Pharmaceutical composition and application thereof to preparation of medicament for treating cerebrovascular disease
InactiveCN102485223AReduce the effective doseGood treatment effectHydroxy compound active ingredientsCardiovascular disorderDiseaseVascular disease
A pharmaceutical composition comprises 3-methyl-1-phenyl-2-pyrazoline-5-ketone and (+)-2-borneol. The pharmaceutical composition can be used in treating cerebrovascular disease and has lower toxic and side effects.
Owner:JIANGSU SIMCERE PHARMACEUTICAL R & D CO LTD
Pyrazoline dione derivatives as nadph oxidase inhibitors
The present invention is related to pyrazoline dione derivatives of Formula (I), pharmaceutical composition thereof and to their use for the treatment and / or prophylaxis of disorders or conditions related to Nicotinamide adenine dinucleotide phosphate oxidase (NADPH Oxidase).
Owner:GENKYOTEX SUISSE SA
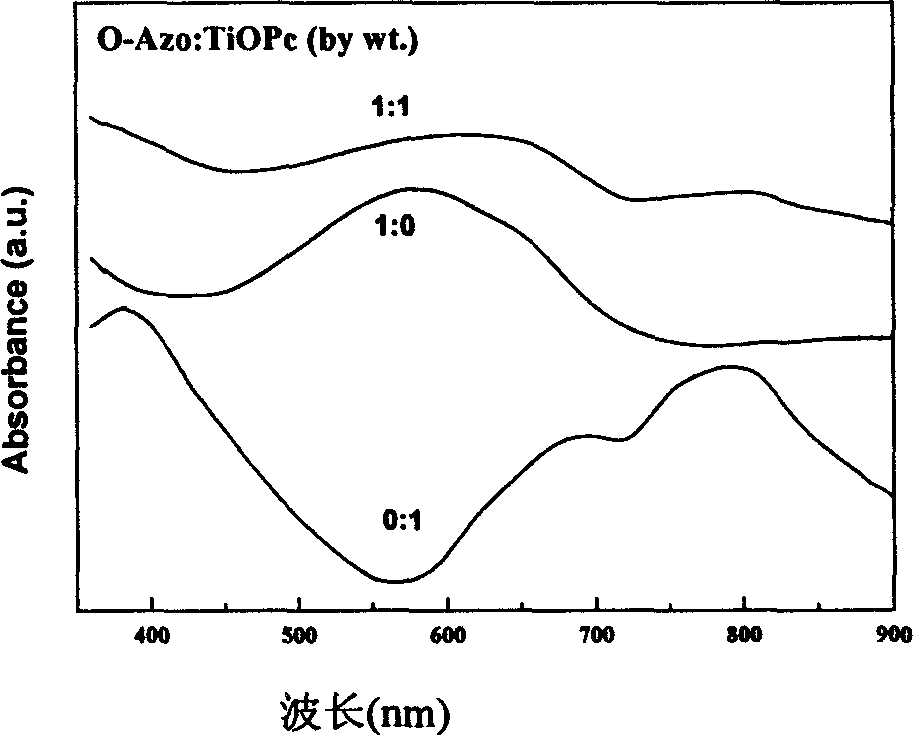
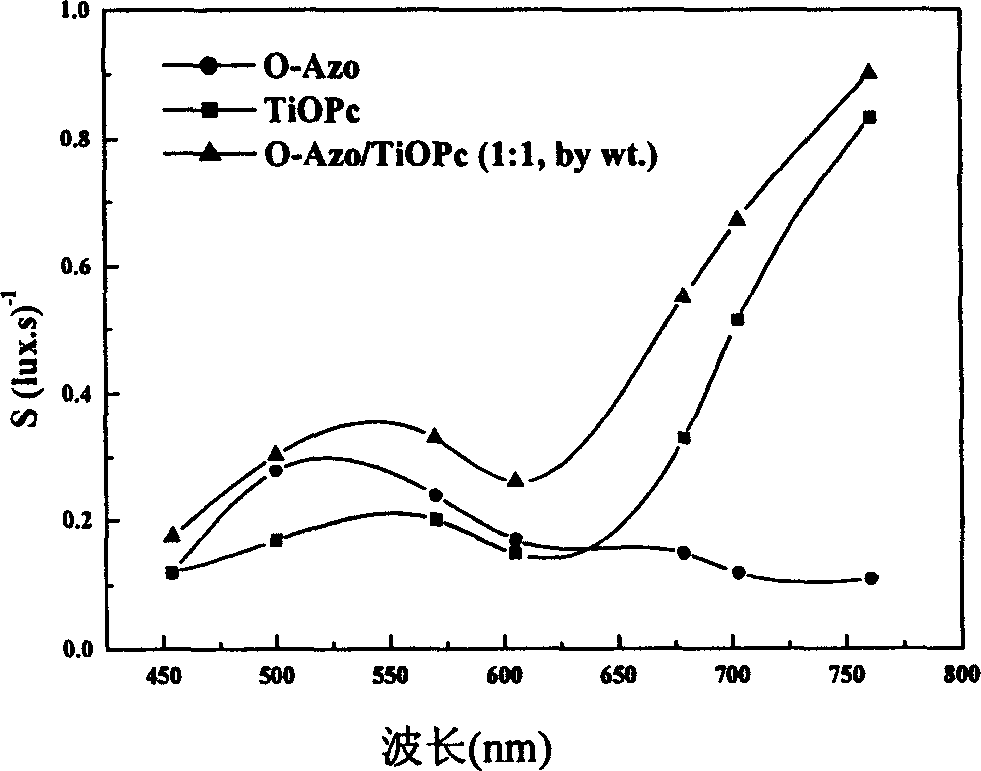
Azo/phthalocyanine composite single layer organic photoconductor and preparing method therefor
InactiveCN1632700AImprove solubilityIncrease the degree of conjugationElectrography/magnetographyElectrical conductorPhthalocyanine
This invention is a wide-frequency vibration and high light sensitive azo or phthalocyanine compound single layer organic photoelectricity conductor and its process method, which belongs to organic photoelectricity conductor technique field. The material mainly adopts charge carrier generation material and charge carrier transmission material and resin and conductive base material with certain proportion to form organic photoelectricity coating liquid and adopts the dipping and coating method. And the charge carrier generation material is the compound material of oxazolyl bisazo or metal phthalocyanine and the charge carrier transmission material adopts compounds of triaryl amine and hydrazone compound, and pyrazoline compound.
Owner:TONGJI UNIV
Burley tobacco nicotine extract product and extraction technology for same
The invention provides a burley tobacco nicotine extract product and an extraction technology for the same. The burley tobacco nicotine extract product is composed of the following components according to weight by percentage: 94 to 97% of nicotine, 0.4 to 0.6% of cotinine, 0.2 to 0.3% of N-methy pridine carboxamide, 0.1 to 0.2% of benzofuran, 0.1 to 0.2% of indol, 0.1 to 0.2% of 5.5-dimethyle pyrazoline, 0.05 to 0.15% of acetyl pridine, 0.05 to 0.1% of megastigmatrienone, 0.02 to 0.05% of hydroxide radical damascenone, 0.02 to 0.05% of palmitic acid and 0.01 to 0.04% of demethylation nicotine. The burley tobacco nicotine extract product has nigh nicotine content and has special flavor when prepared for electronic tobacco juice; electronic cigarette satisfaction can be enhanced; and the electronic cigarette can be closer to cigarette taste.
Owner:CHINA TOBACCO HUNAN INDAL CORP
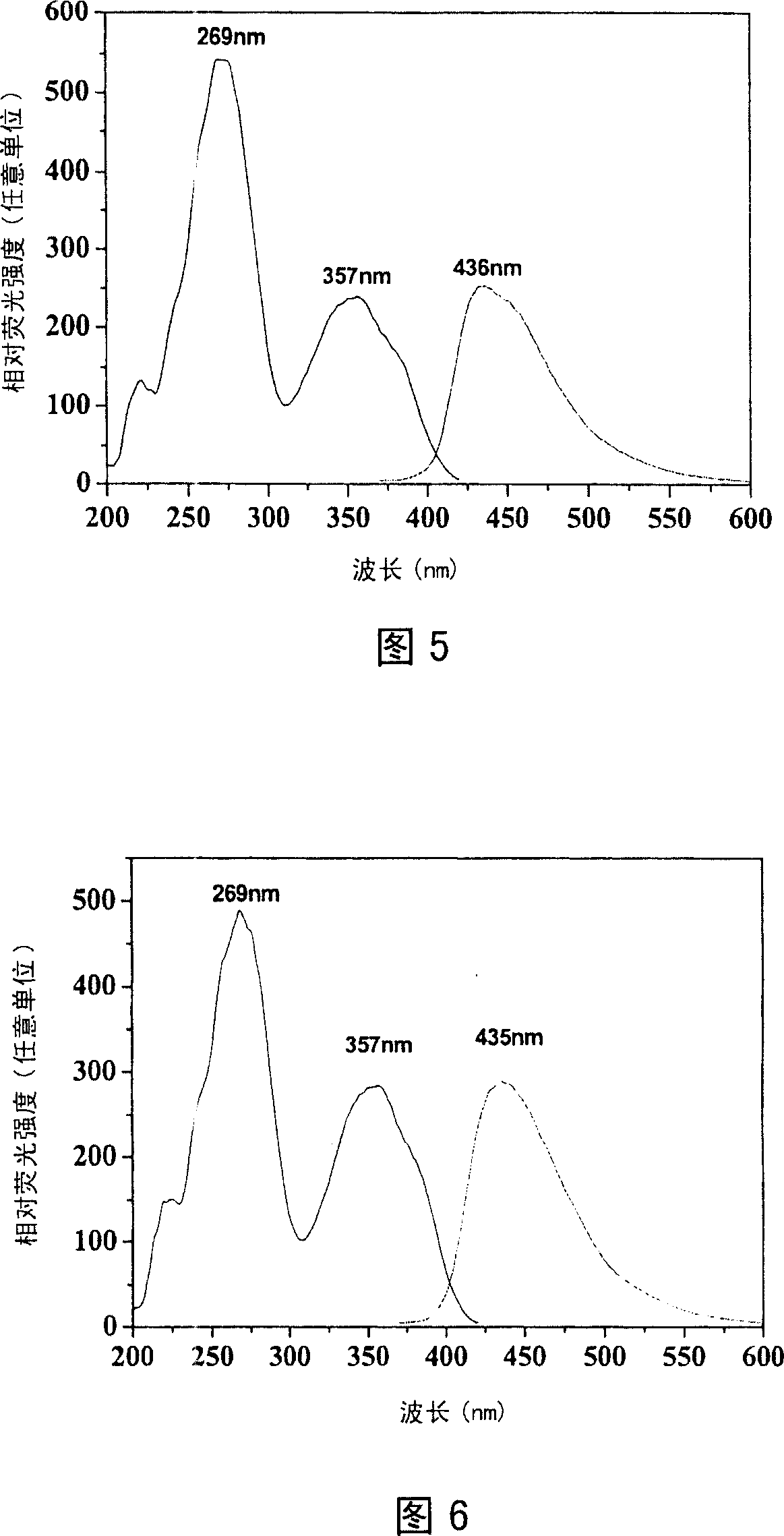
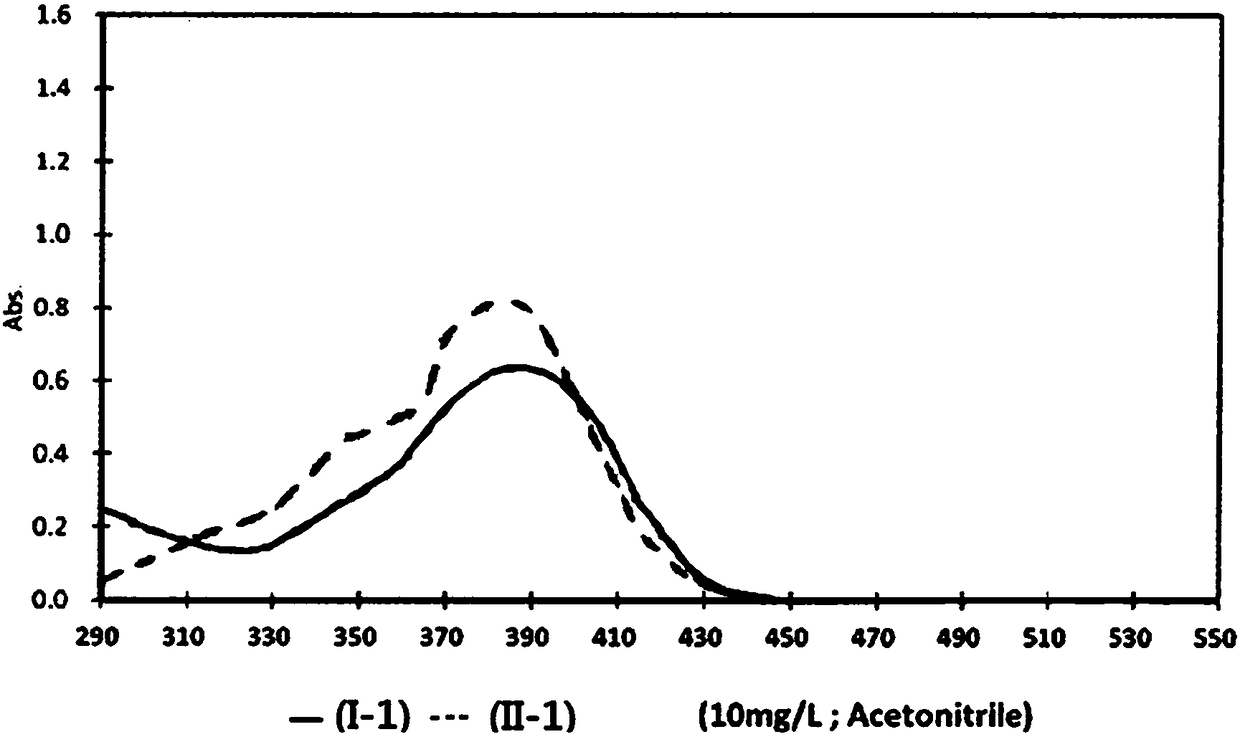
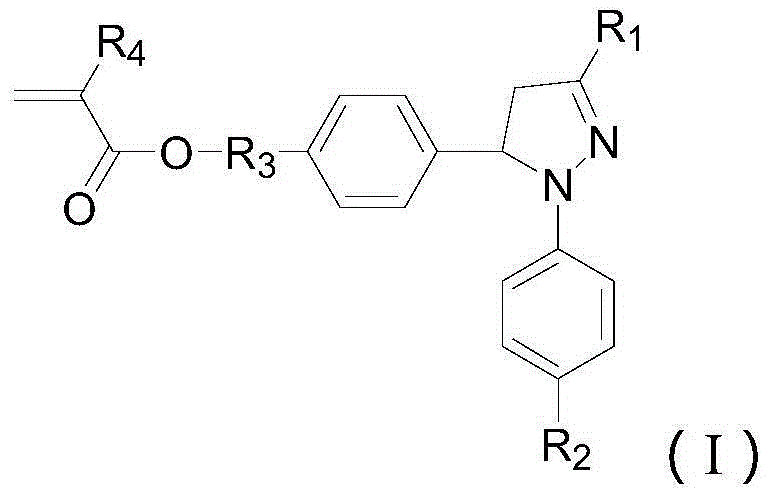
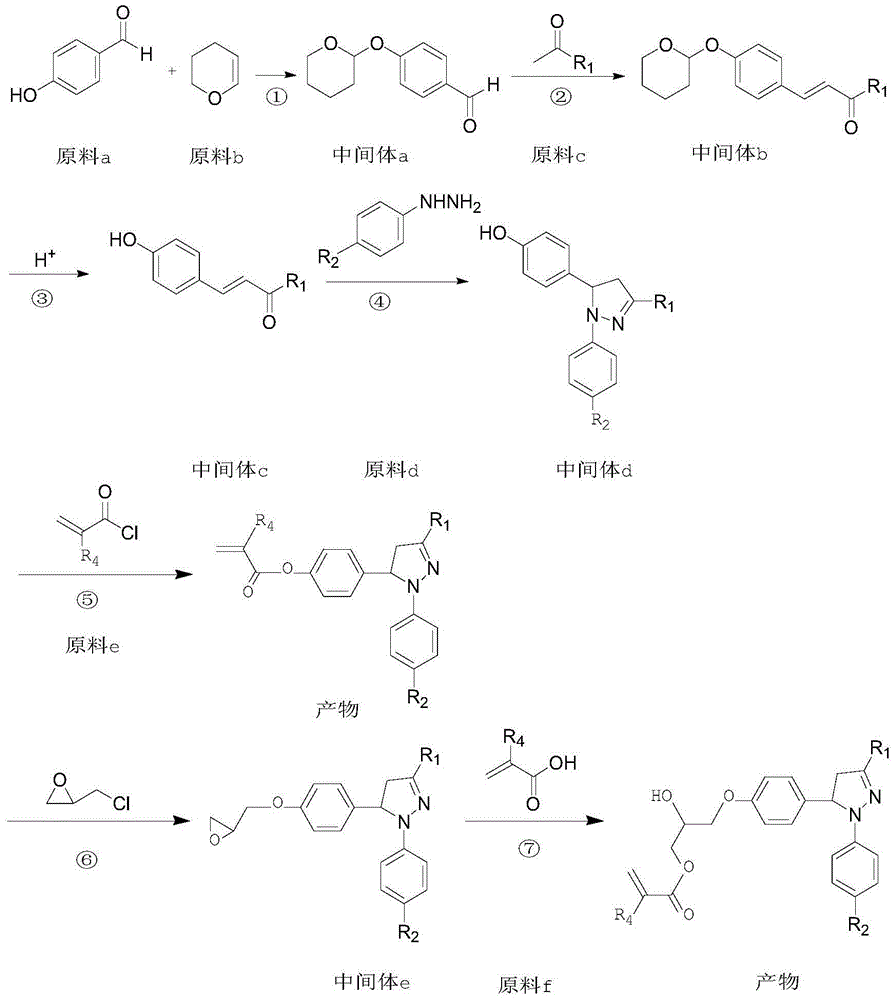
Pyrazoline sensitizer as well as preparation method and application thereof
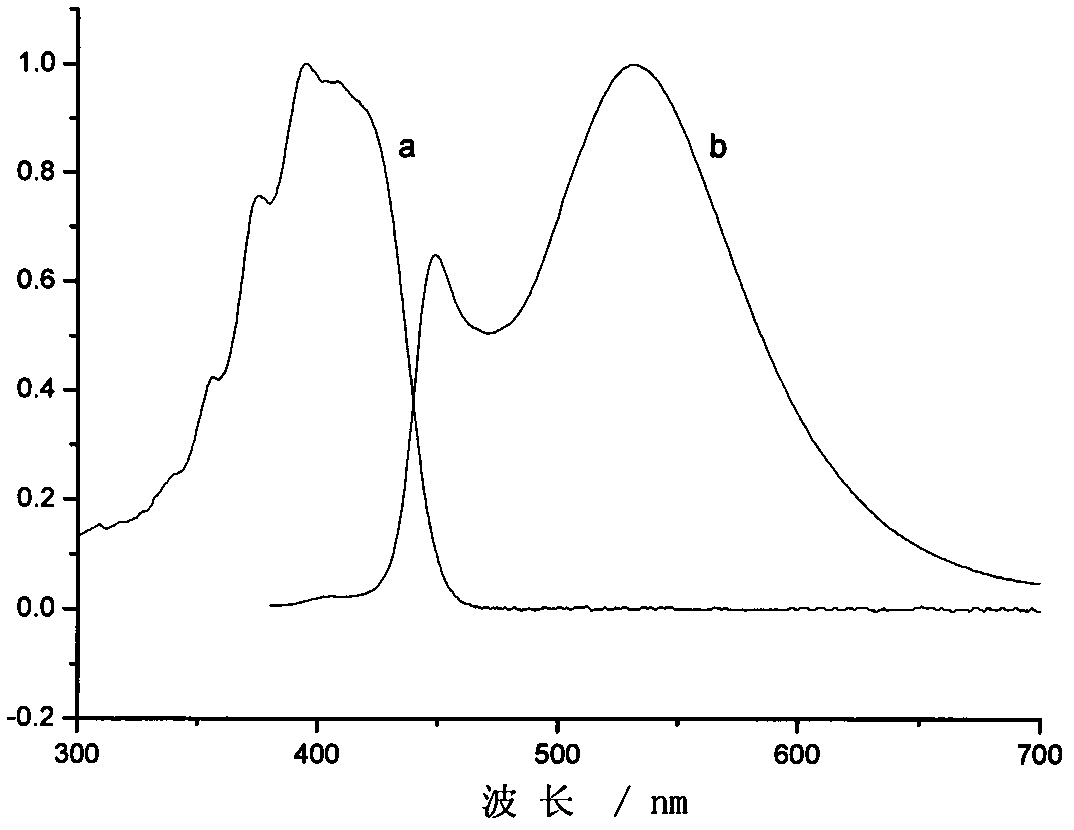
The invention provides pyrazoline sensitizer having a structure shown in the formula (I) as shown in the specification. The pyrazoline sensitizer has absorption waveband which is between 350nm and 440nm, is suitable for a light-curing system and particularly suitable for a system containing biimidazole photoinitiator. The pyrazoline sensitizer has ideal solubility and good light sensitivity improving effect. and a prepared film is good in resolution and adhesive force.
Owner:CHANGZHOU TRONLY ADVANCED ELECTRONICS MATERIALS CO LTD +1
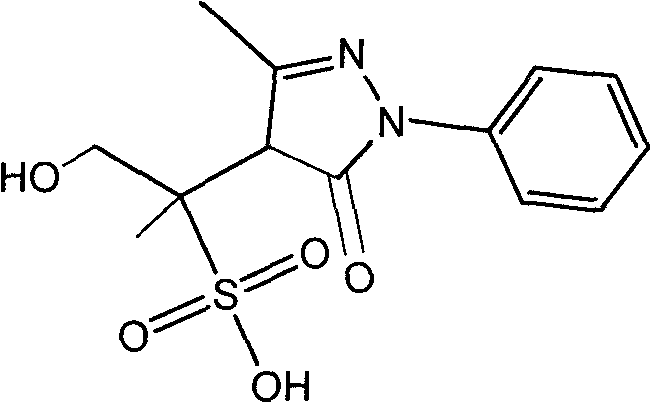
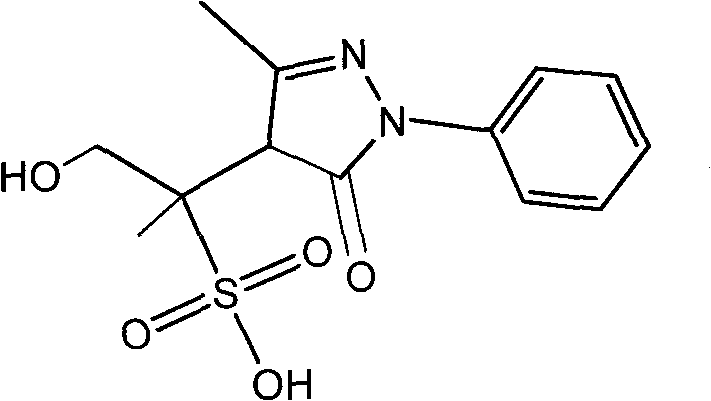
Pyrazolines compound as well as application and preparation method thereof
The invention relates to a compound having a structure as shown in a specification as well as a preparation method and application thereof. The preparation method comprises the steps of: reacting edaravone, sodium pyrosulfite and 1,2-propylene glycol; placing the solution of the reaction under an environment with the temperature of 35-55 DEG C for 96-168 hours and filtering for later use; and separating the edaravone solution by using a reversed phase high performance liquid chromatography method for preparing 4-(1-hydroxy-2-sulfo-propane-2-yl)-3-methyl-1-phenyl-2-pyrazoline-5-ketone. The compound can be used a comparison product of edaravone impurities and is convenient for controlling edaravone and the content of the edaravone in relevant preparations.
Owner:JIANGSU SIMCERE PHARMA +2
Pyrazoline pyridine-fluorescent coumarin dye derivant, synthesis and uses thereof
ActiveCN101368003AImprove luminous efficiencyCoumarine dyesLuminescent compositionsLaser dyeQuantum yield
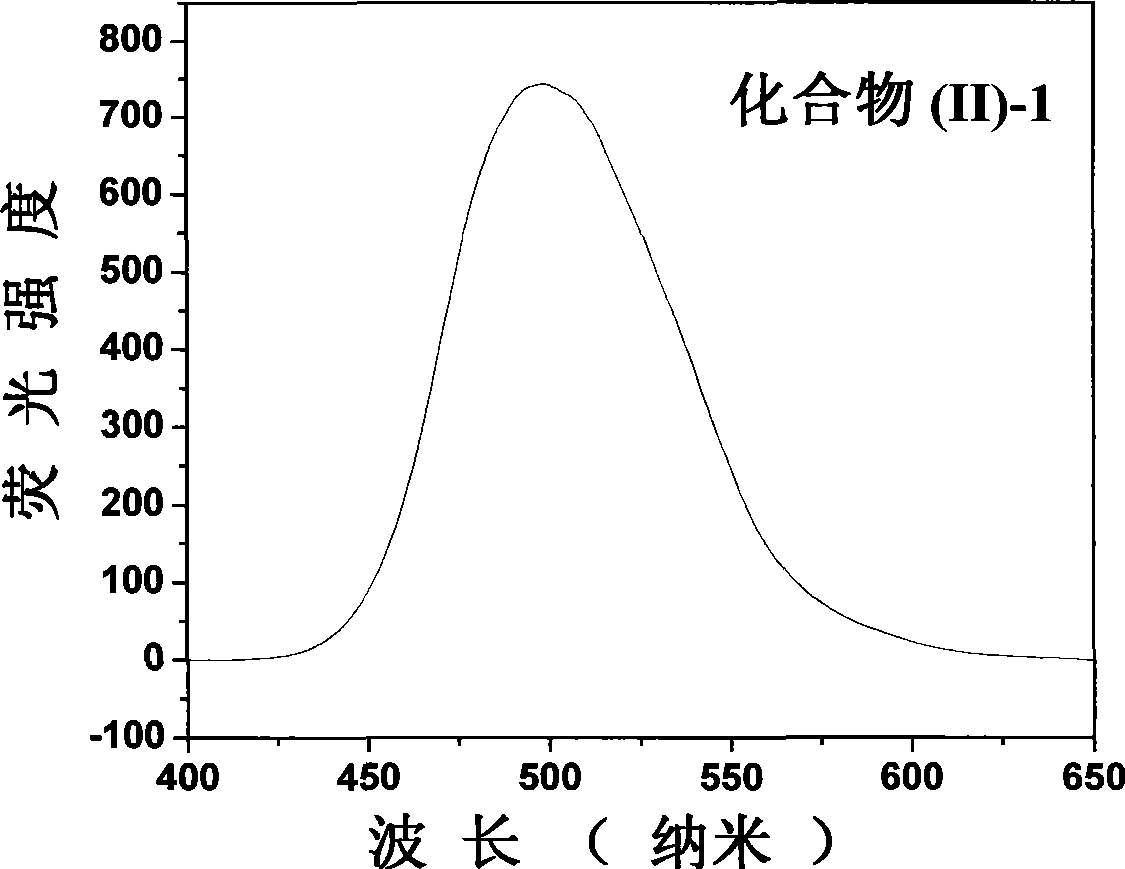
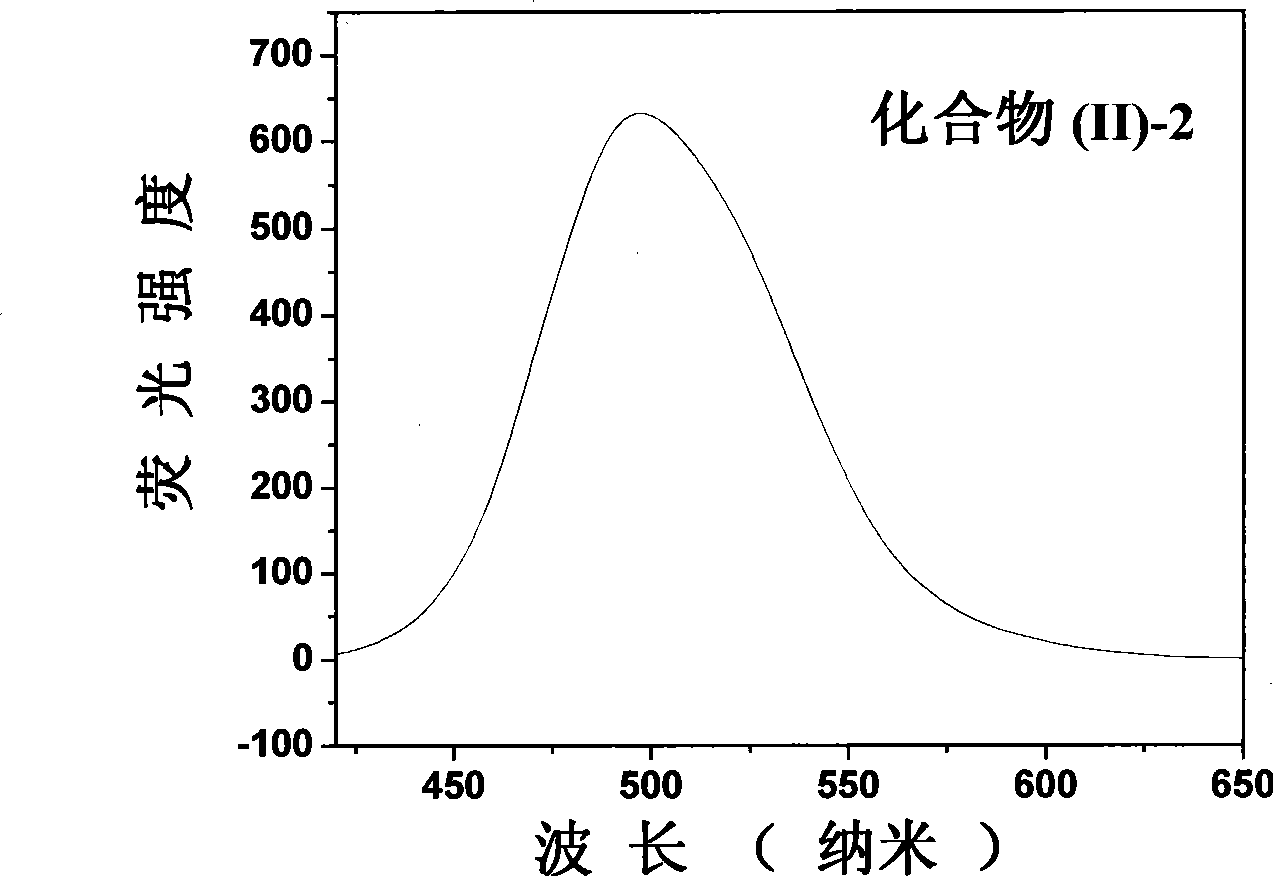
The invention belongs to the field of fluorescent dye and laser dye, and relates to coumarin fluorescent dye and laser dye, in particular to pyrazoline naphthyridine coumarin fluorescent dye derivative, and a synthetic method and an application thereof. The pyrazoline naphthyridine coumarin fluorescent dye derivative has very high fluorescent efficiency with the quantum yield of fluorescence of nearly 100 percent; meanwhile, the fluorescent material has excellent heat, light and electricity chemical stability, and can be used as fluorescent dye, laser dye, organic electroluminescent material, fluorescent marking material, fluorescent chemical sensitive material and the other. The pyrazoline naphthyridine coumarin fluorescent dye derivative has structure as seen in formula I or formula II.
Owner:TECHNICAL INST OF PHYSICS & CHEMISTRY - CHINESE ACAD OF SCI
Method for synthesizing bicycle aza ring fluorescent or phosphorescent compound
InactiveCN101113150AMild preparation conditionsEasy to separate and purifyOrganic chemistryLuminescent compositionsSynthesis methodsCycloaddition
The invention relates to a synthetic method of a dicyclic azacyclo fluorescence or phosphorescent compounds: a metal carbene compound with a dicyclic azacyclo structure skeleton is produced from a carbene compound and 2-pyrazoline-5-ketone through a cycloaddition reaction in an organic solvent, and cis- and trans- dicyclic azacyclo organic compounds are respectively prepared after oxidation and demetalization; wherein, the cis-dicyclic azacyclo compounds have strong fluorescence performance, which can be uses as a small molecule probe compound for the analysis and checking of protein and cell; trans-dicyclic azacyclo compound is a non-fluorescent compound and may has phosphorescence; the oxidant used is inorganic or organic oxidant, and the reaction solvent is polar or non-polar organic solvent. The invention is of simple operation, with material easy to be acquired, a few reaction steps, better efficiency and selectiveness than traditional method; in particularly, compared with traditional synthesis method, the invention can more easily synthesize asymmetrical dicyclic azocyclo bimane compound.
Owner:DALIAN INST OF CHEM PHYSICS CHINESE ACAD OF SCI
Concentrated softener for dry cotton fabric
The invention relates to a concentrated softener for a dry cotton fabric. The concentrated softener comprises the following components in parts by mass: 50-65 parts of dihydrogenated tallow dimethyl ammonium chloride, 6-15 parts of ester quaternary ammonium salt, 0.7-1.5 parts of polyacrylamide, 2.5-8 parts of polyethylene glycol, 18-30 parts of isopropanol, 2-8 parts of coconut oil fatty acid diethanolamide, 0.8-1.5 parts of a pyrazoline fluorescent whitening agent and 0.2-0.6 part of fragrances. The concentrated softener provided by the invention is in a concentrated state; the number of use of the concentrated softener is more than that of an existing softener under the same volume; meanwhile, the treated dry cotton fabric is soft, fluffy, fresh and bright white, and does not cause yellowing and generate static electricity.
Owner:南通信一服饰有限公司
Anti-blue-ray system containing pyrazoline or/and cinnamic acid compounds
ActiveCN109384878ALow mobilityGood compatibilityOrganic chemistryFilm/foil adhesivesAdhesiveVisual perception
Owner:江苏裕事达新材料科技有限责任公司
Alkenyl-containing pyrazoline sensitizer as well as preparation method and application thereof
ActiveCN104892513AOrganic chemistryPhotosensitive materials for photomechanical apparatusSolubilityPhotochemistry
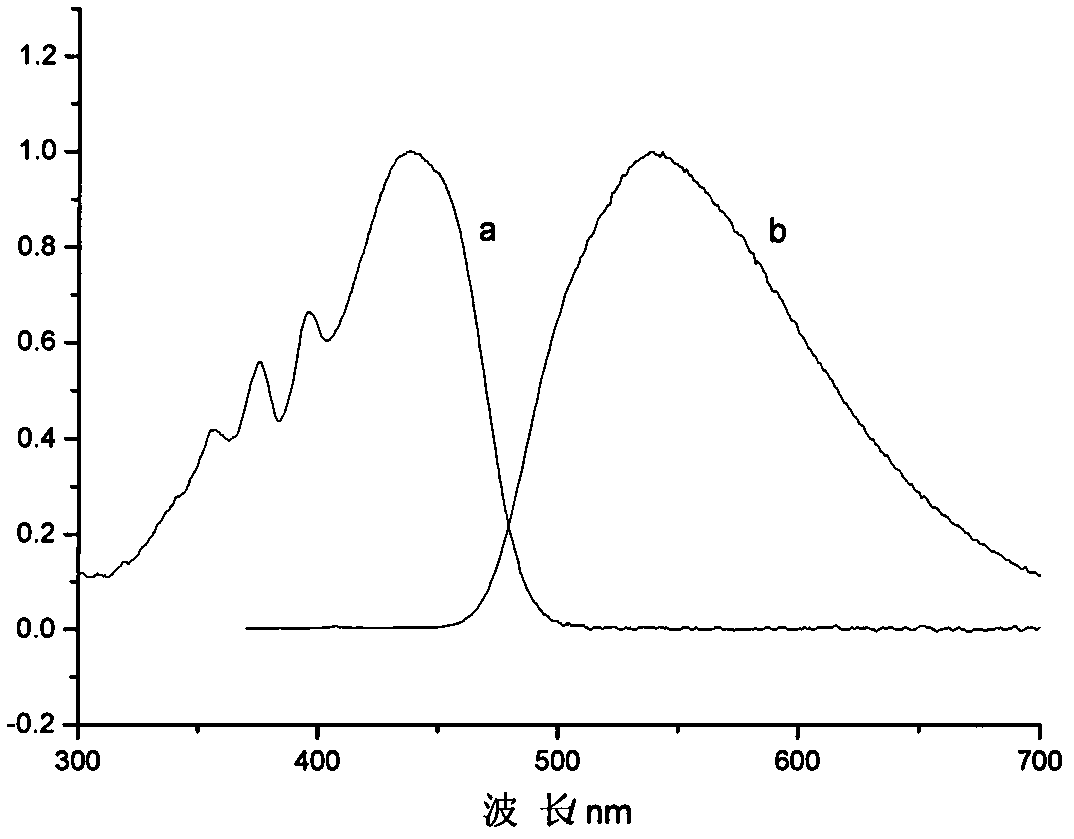
The invention discloses an alkenyl-containing pyrazoline sensitizer as well as preparation method and application thereof. When used in a photocuring system, the alkenyl-containing pyrazoline sensitizer is very ideal in dissoluvability, free of odor and excellent in sensitivity improvement effect, and a film made of the alkenyl-containing pyrazoline sensitizer is very good in resolution ratio and adhesion property.
Owner:CHANGZHOU TRONLY ADVANCED ELECTRONICS MATERIALS CO LTD +1
Process for inhibiting oxide formation on copper surfaces
InactiveUS20110045203A1Pretreated surfacesSemiconductor/solid-state device manufacturingThin layerCopper oxide
Processes are provided for inhibiting the formation of copper oxides on substantially oxide-free copper surfaces by contacting a substantially oxide-free copper surface with a pyrazoline ligand in an aqueous solution of pH 2-5. A thin layer of the ligand formed by coordination of 2-pyrazoline or 1-methyl-2-pyrazoline to the copper surface can be easily removed by exposure to a reducing plasma to regenerate a substantially oxide-free copper surface.
Owner:EI DU PONT DE NEMOURS & CO
Sulfonium salt containing pyrazoline groups and preparation method and application thereof
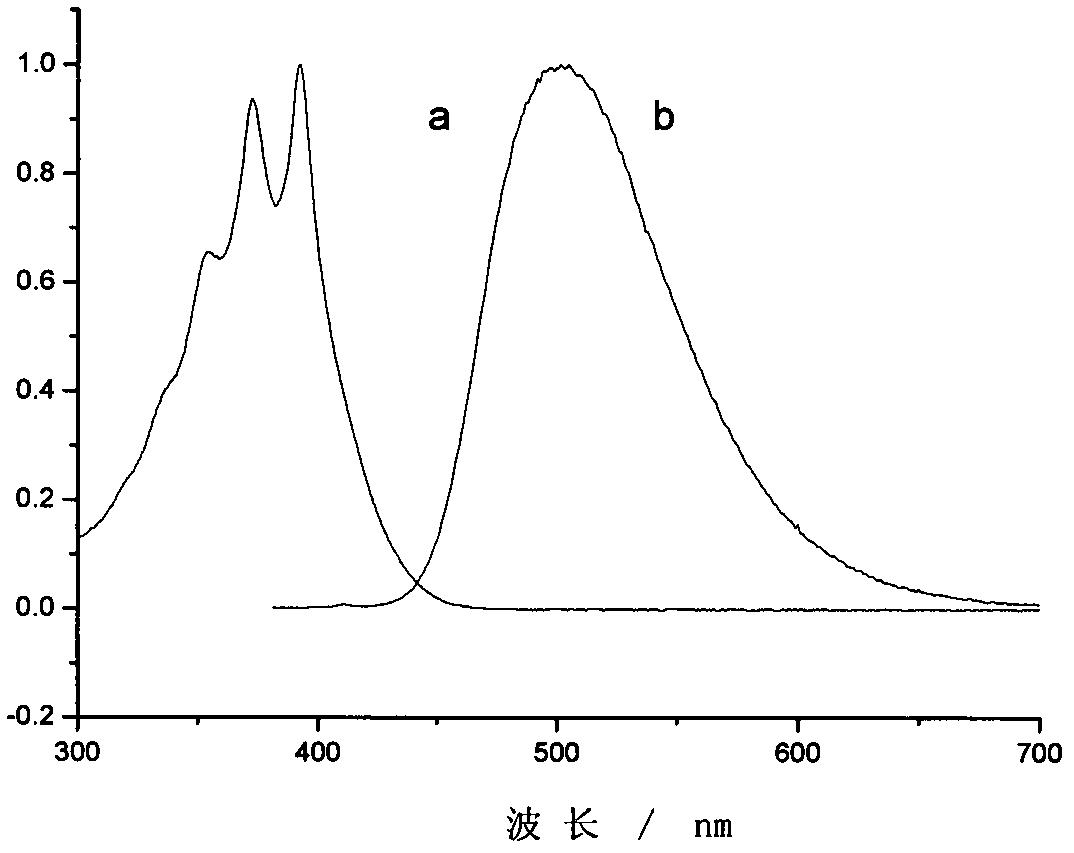
The invention provides a sulfonium salt containing pyrazoline groups and a preparation method and an application thereof, wherein the general formula of the sulfonium salt is shown in the description;R <0> and R <1> are independently selected from hydrogen, halogen atom, R', OH, OR', CH2OH, CH2OR', NR'R <0>, CH2NR'R <0>, CF3, NO2 or CN; R2 is selected from aryl groups substituted by CH2R or R4, R5, R6, R7 and R8; R3 is selected from aryl groups substituted by CH2R or R9, R10, R11, R12 and R13; the sulfonium salt is monosubstituted on the phenyl ring, and the substitution position is selectedfrom the ortho, meta or para position of the pyrazoline group; X- is selected from Cl-, CF3SO3-, CH3SO3-, BF4-, B(Ph)4-, B(PhF5)4-, PF6-, SbF6-, (CF3CF2)nPF6-n- or ((CF3)3CO) 4Al; the sulfonium salt containing the pyrazoline group can be excited by the LED, and has good light absorption and photo-acid generation properties; in addition, the preparation method has the advantages of simple process,low cost, less pollution and high yield, and can be used for industrial production.
Owner:TONGJI UNIV +1
Pyrazoline naphthyridine benzocoumarin fluorescent dye derivatives, synthesis method of same, and application of same
InactiveCN101525497ALong emission wavelengthImprove luminous efficiencyElectrical apparatusElectroluminescent light sourcesLaser dyeQuantum efficiency
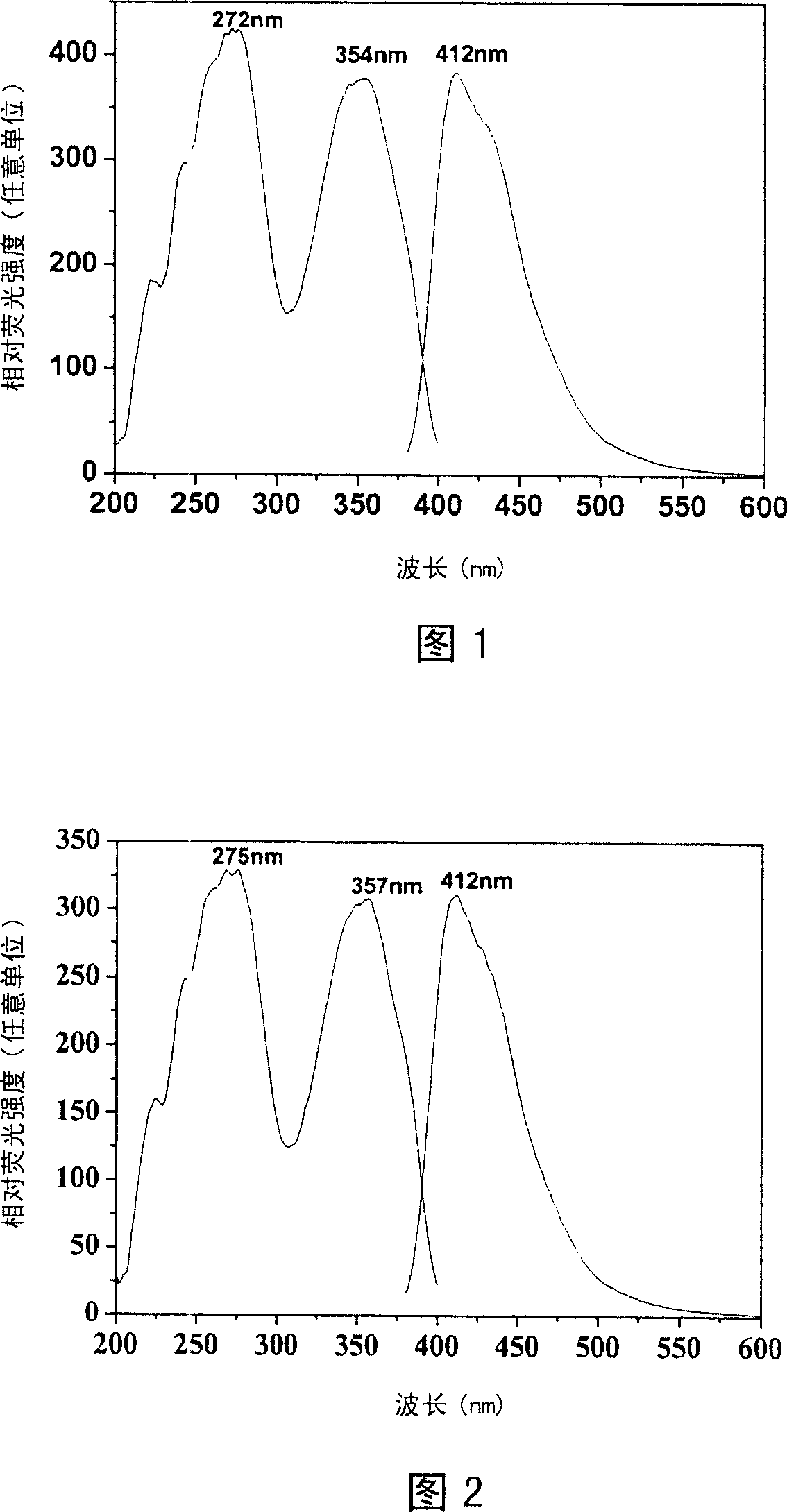
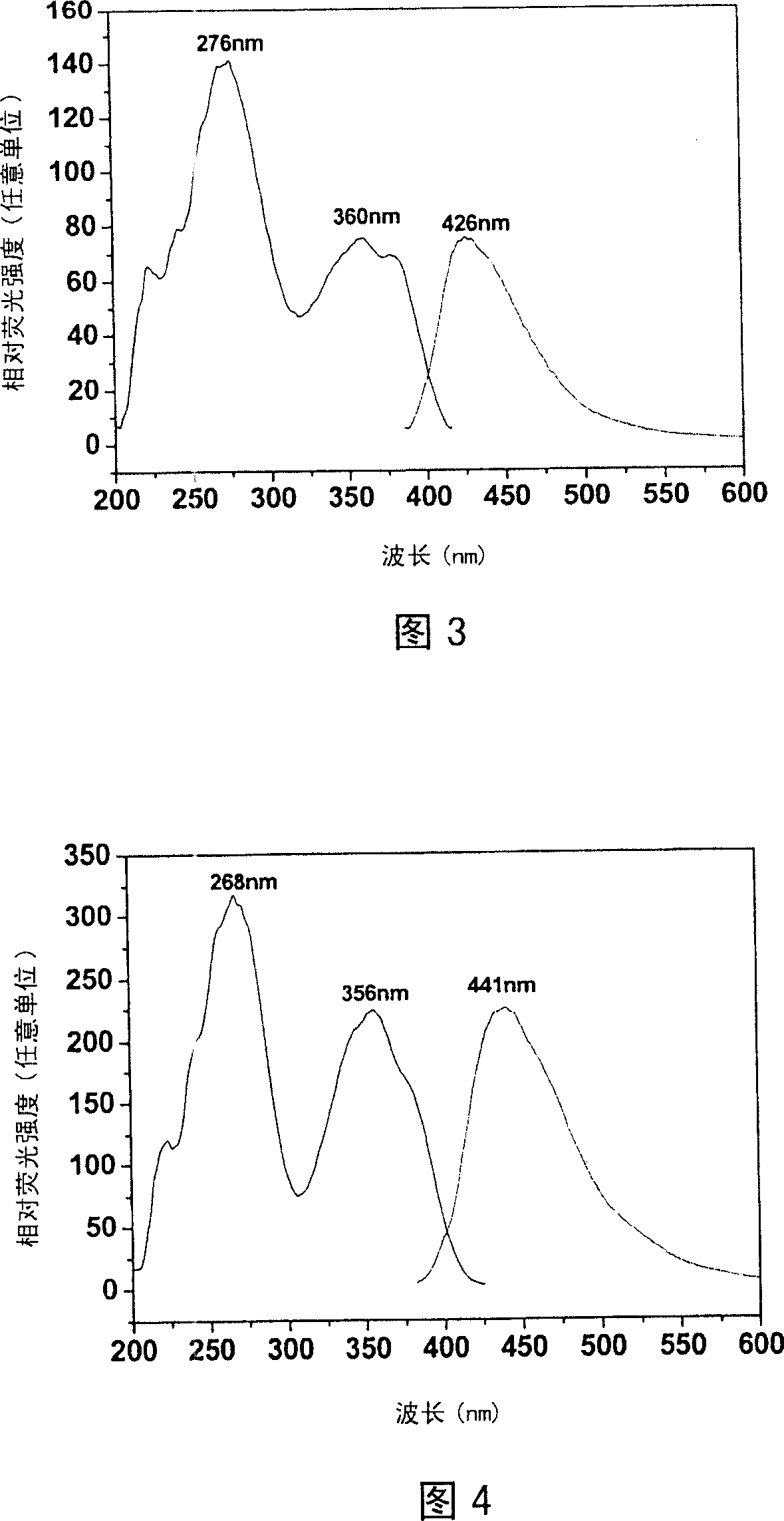
The invention belongs to the field of fluorescent dyes and laser dyes, and relates to coumarin fluorescent dyes and laser dyes, in particular to pyrazoline naphthyridine benzocoumarin fluorescent dye derivatives, a synthesis method of the same, and application of the same. The pyrazoline naphthyridine benzocoumarin fluorescent dye derivatives have longer emission wavelength and extremely high luminous efficiency, and the fluorescence quantum efficiency of the derivatives is approximately 100 percent; at the same time, a luminescent material of the derivatives has excellent thermo, photo and electrochemical stability, and can be used as fluorescent dyes, laser dyes, organic electroluminescent materials, fluorescent labeling materials, fluorescent chemosensitive materials and the like. The pyrazoline naphthyridine benzocoumarin fluorescent dye derivatives have a structure shown in a formula (I) or a formula (II).
Owner:TECHNICAL INST OF PHYSICS & CHEMISTRY - CHINESE ACAD OF SCI
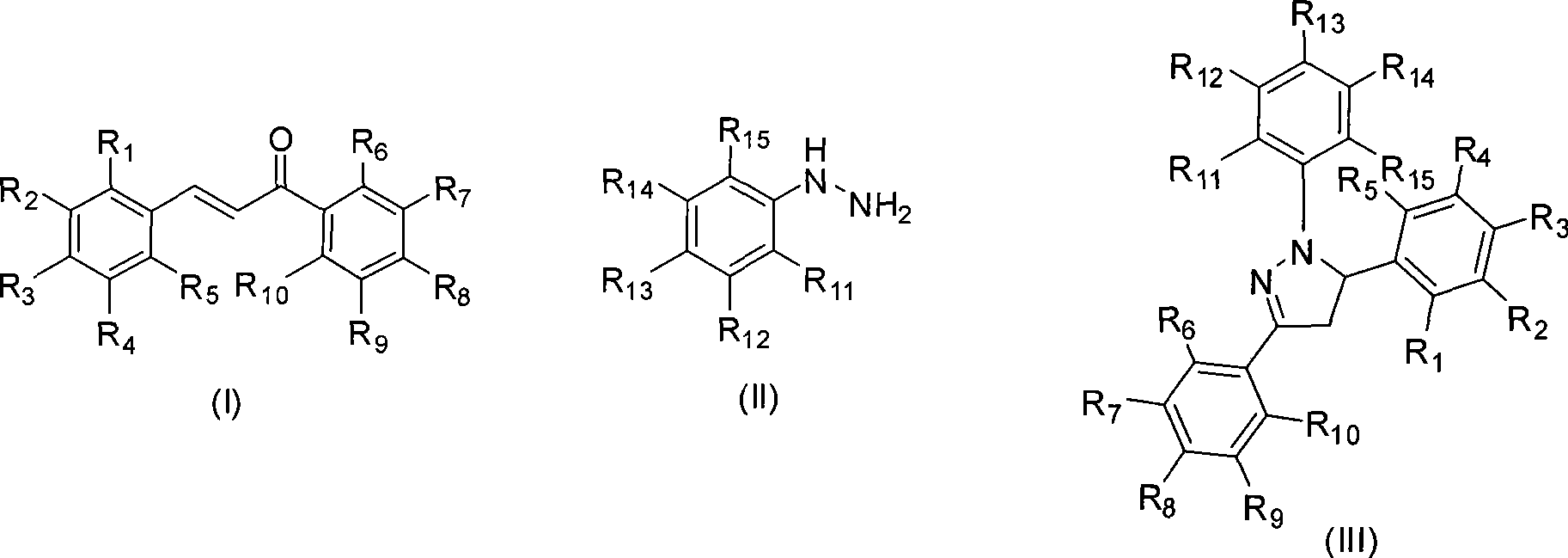
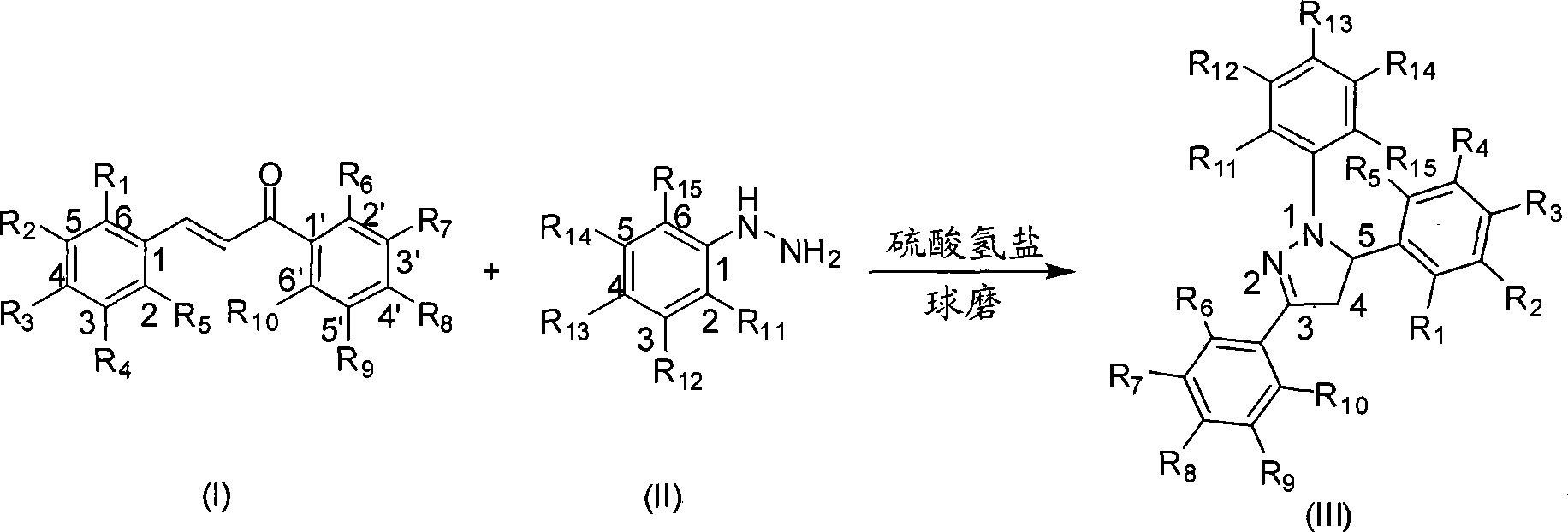
Mechanochemical preparation of 1,3,5-triaryl-2-pyrazoline compounds
InactiveCN101440065AWiden the optionsSolve the problem of large yield variationOrganic chemistrySilica gelRaw material
The invention discloses a mechanochemical preparation method for a 1, 3, 5-triaryl-2-pyrazoline compound with a structure shown in a formula (III). The method comprises the following steps: a chalcone compound with a structure shown in a formula (I) and a phenyl hydrazine compound with a structure shown in a formula (II) are taken as raw materials, hydrosulfate is taken as a catalyst, and silica gel is taken as a grinding aid to perform a ball milling reaction in a closed ball milling tank; and after the reaction is finished, a reaction mixture is separated and purified to obtain the 1, 3, 5-triaryl-2-pyrazoline compound with the structure shown in the formula (III). The method has the advantages of safe and reliable production, simple and convenient operation, short reaction time, generally higher reaction yield, low production cost, simple post-treatment, small pollution and so on, and is the preparation method for the 1, 3, 5-triaryl-2-pyrazoline compound with better popularization and application prospects.
Owner:ZHEJIANG UNIV OF TECH
New application of composition of 3-methyl-1-phenyl-2-pyrazoline-5-ketone and borneol
ActiveCN102648908ALower cAMP levelsHydroxy compound active ingredientsDermatological disorderMedicineKetone
The invention relates to a new application of composition of 3-methyl-1-phenyl-2-pyrazoline-5-ketone and borneol, namely the application in preparation of medicine for preventing and / or treating psoriasis.
Owner:NANJING SIMCERE DONGYUAN PHARM CO LTD +1
Blue hyper-branched polymer electroluminescent materials using pyrazoline unit as nuclear and preparation method thereof
InactiveCN101362820AAvoid defectsElectrical apparatusElectroluminescent light sourcesOrganic field-effect transistorBackbone chain
The invention discloses a blue hyperbranched polymer electroluminescent material with a pyrazoline unit core and a preparation method thereof, wherein, the hyperbranched polymer electroluminescent material is characterized by a chemical structural formula as shown on the right. The hyperbranched polymer electroluminescent material has the advantages that: 1. the hyperbranched polymer electroluminescent material can emit pure blue light, thereby overcoming the defect that exciplex is generated by incident primary chain cluster of the linear poly (9,9-dihexylfluorene) blue-light emitting materials; 2. The hyperbranched polymer electroluminescent material can apply to electroluminescent components, organic field effect transistors, solar batteries and biosensors.
Owner:NANCHANG HANGKONG UNIVERSITY
Preparation method of porous composite material capable of selectively absorbing aromatic hydrocarbon
InactiveCN103949219AReduce manufacturing costImprove adsorption capacityIon-exchange process apparatusOther chemical processesTetracyanoquinodimethaneAromatic solvents
The invention discloses a preparation method of a porous composite material capable of selectively absorbing aromatic hydrocarbon. The composite material is obtained from two monomer molecules by cocrystallization. Particularly, the preparation method comprises the steps of: dissolving 1-acetyl-3-(4-methoxy phenyl)-5-(9-anthryl) pyrazoline (AMPE) and 7, 7, 8, 8-TCNQ in a non-aromatic solvent, then removing the solvent, and finally drying to obtain the composite material. The composite material provided by the invention is used for only selectively absorbing aromatic hydrocarbon from a mixture of aromatic hydrocarbon and non-aromatic hydrocarbon solvents.
Owner:SOUTHEAST UNIV
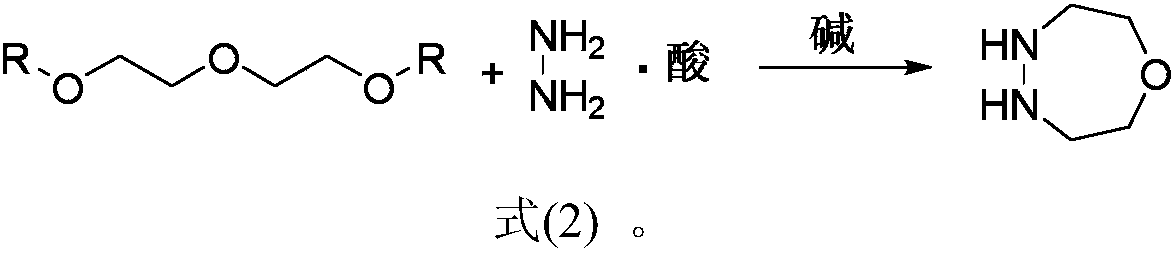
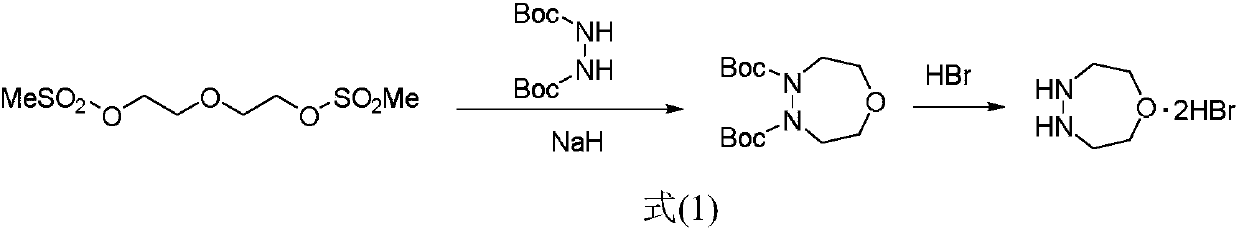
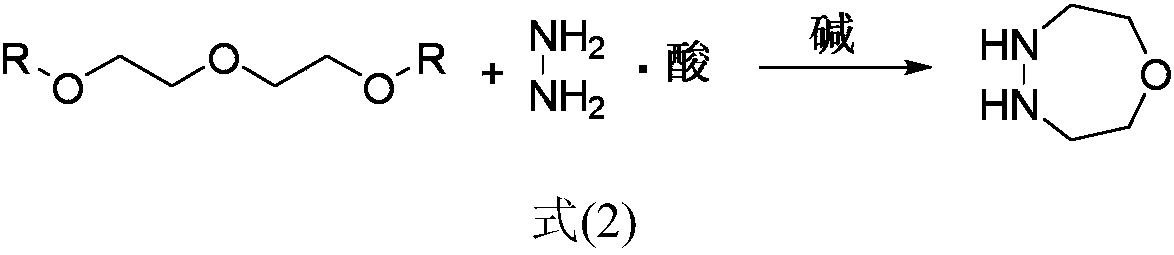
Simple and efficient 1-oxygen-4,5-diazepane synthetic method
The invention discloses a new method for preparing 1-oxygen-4,5-diazepane easily and efficiently. The method comprises the steps that hydrazine salt and the equivalent amount of diglycol sulfonic ester are added into a solvent, then the equivalent amount of alkaline media are added, a condensation reaction is conducted, and 1-oxygen-4,5-diazepane is obtained directly; a reaction equation is as shown in a formula (2) defined in the description. The product can be used for preparing phenyl pyrazoline herbicide; compared with the method in original paper, the method has the advantages of being short in step, low in raw material cost, capable of obtaining raw materials easily, safe and mild in reaction condition, low in production cost and the like, and large-scale production popularization iseasy to achieve.
Owner:SOUTHEAST UNIV
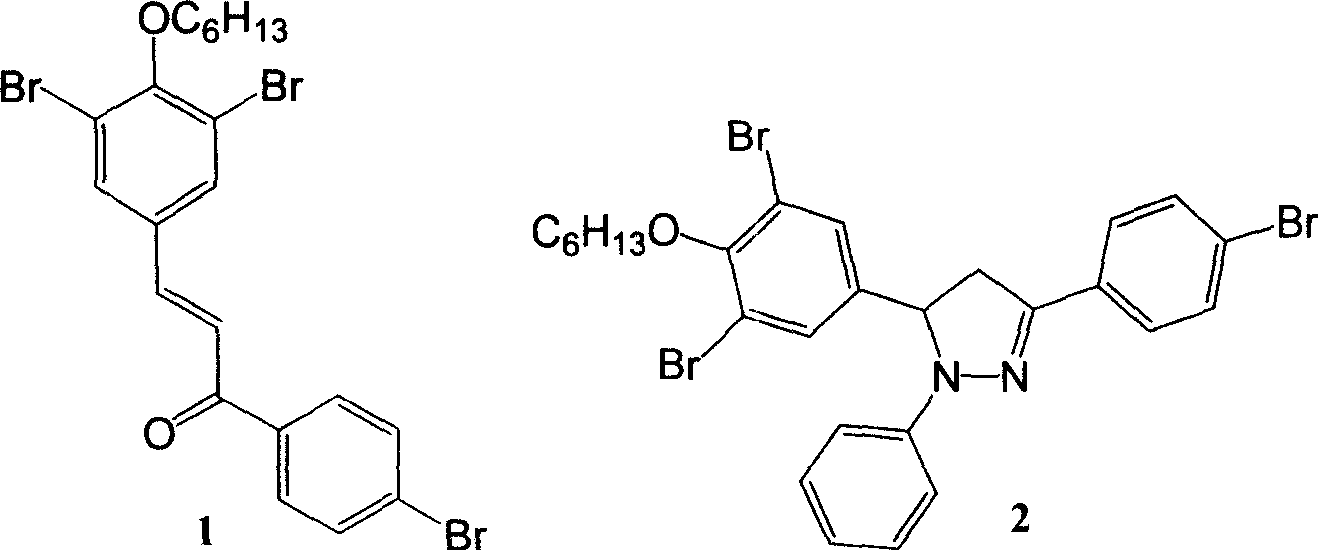
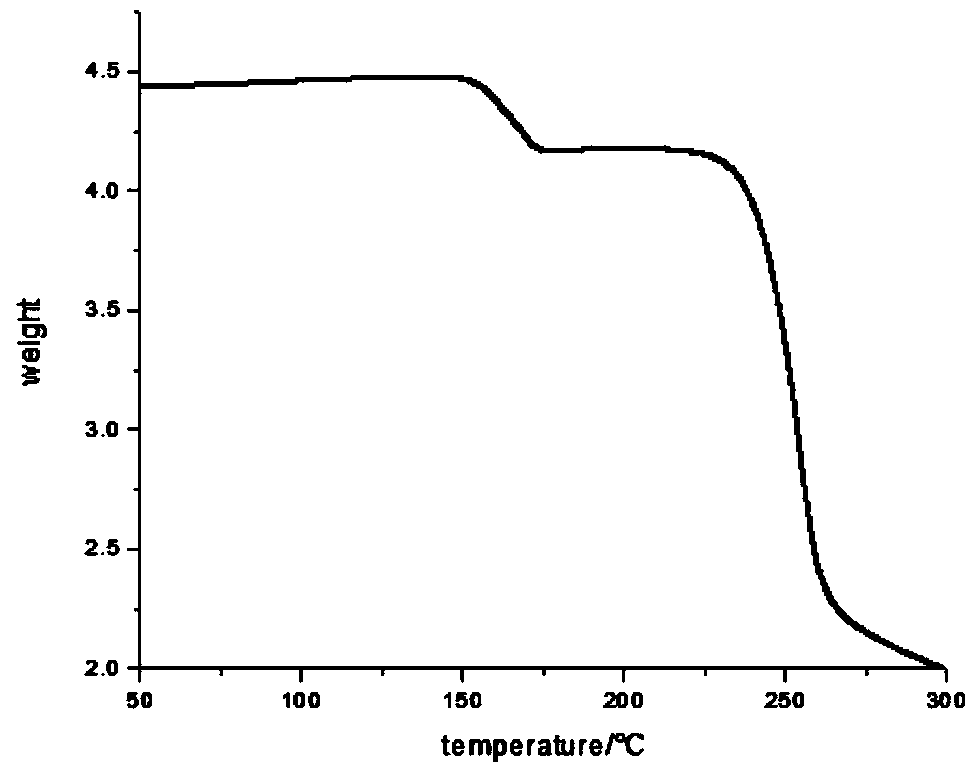
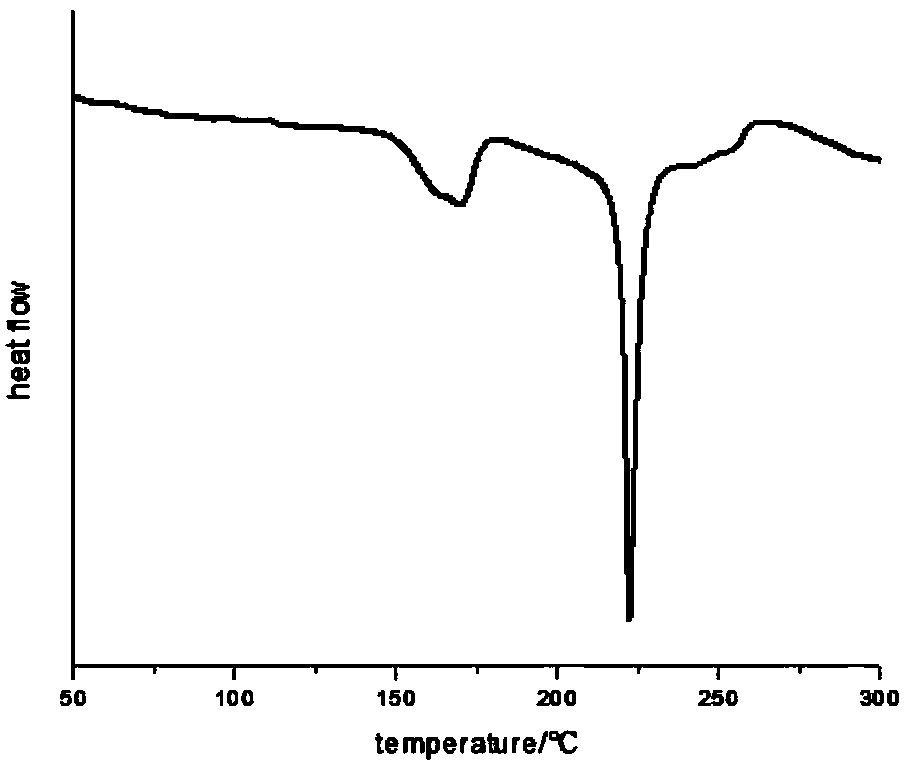
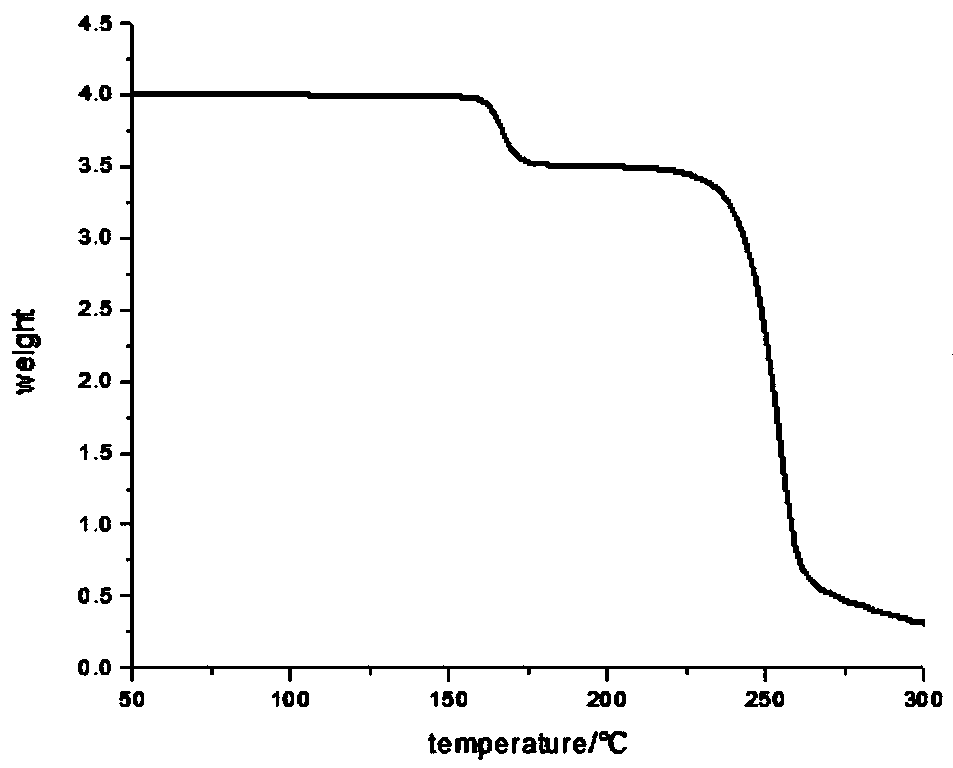
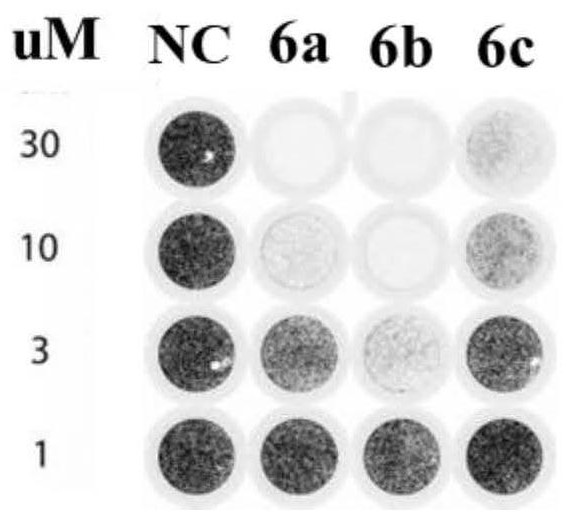
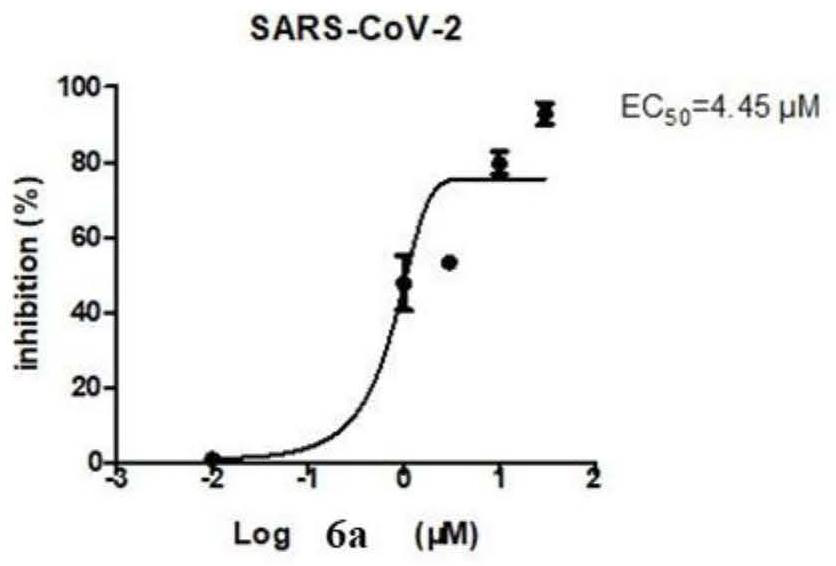
Bromophenol-pyrazoline compound and synthesis method and application thereof
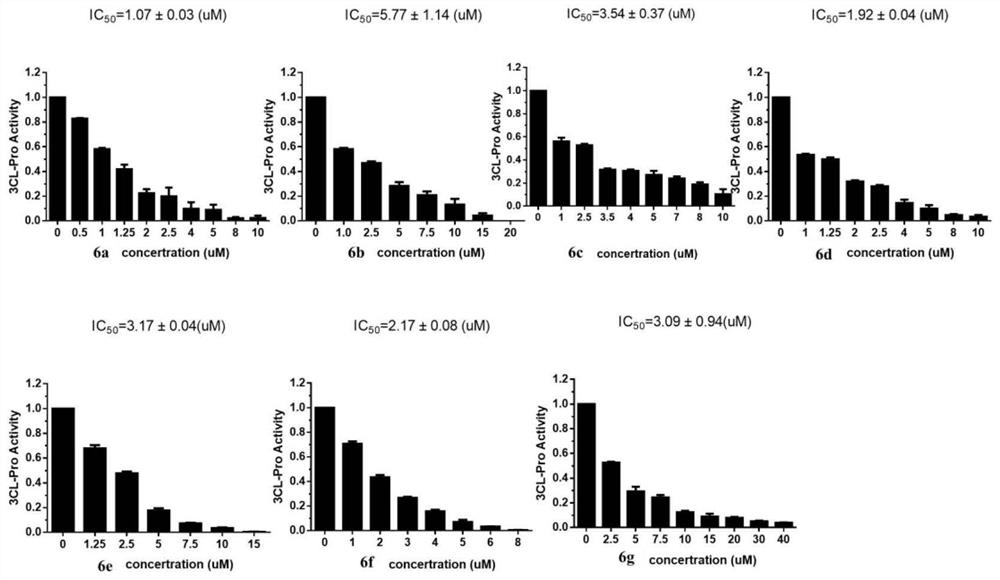
The invention relates to a compound, in particular to a bromophenol-pyrazoline compound and a synthesis method and application thereof. The bromophenol-pyrazoline compound provided by the invention has efficient main protease Mpro inhibitory activity, can interfere with the replication of coronavirus in cells, has the effect of treating coronavirus pneumonia, and has a wide application prospect inpreparation of drugs for treating coronavirus pneumonia.
Owner:LEAD HIGH TECH (QINGDAO) CO LTD
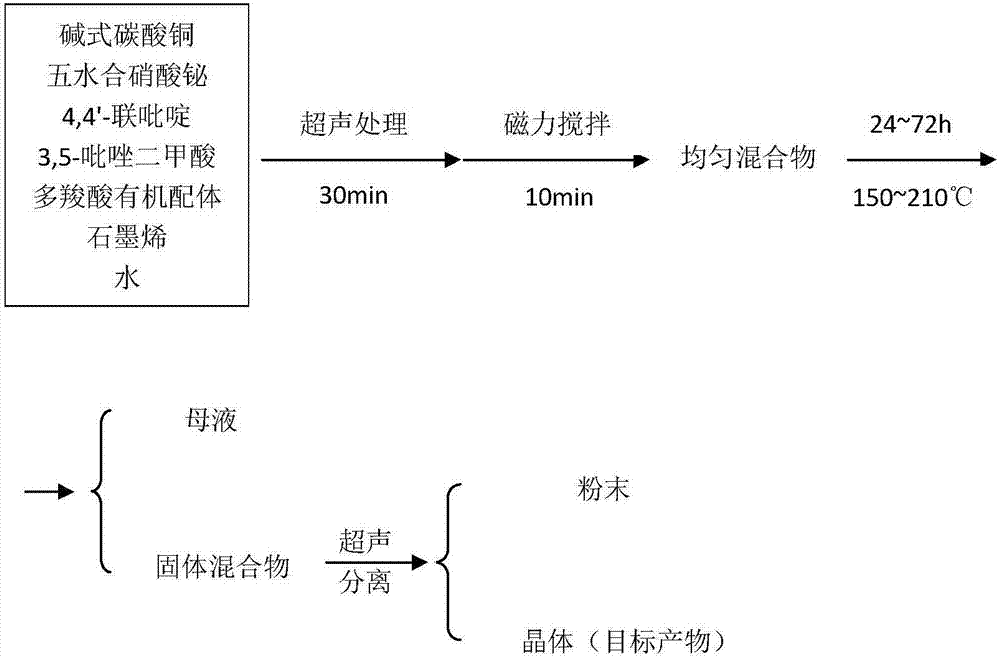
Compound of copper-bismuth double metal coordination polymer and graphene and preparation method thereof
InactiveCN106944150AEasy to prepareLow costWater/sewage treatment by irradiationWater treatment compoundsStrong acidsBimetal
The invention relates to a compound of copper-bismuth double metal coordination polymer and graphene and a preparation method thereof. The compound is prepared from the following components in parts by mass: 1-2 parts of basic cupric carbonate, 2-6 parts of bismuth nitrate pentahydrate, 2-3 parts of 4,4'-dipyridyl, 3-5 parts of 3,5-pyrazoline diformic acid and smaller than 50 parts of graphene. The material can quickly degrade a rhodamine B solution under visible light and has a good photocatalytic property. In the preparation process of the material, no strong acids or strong alkali, strong oxidants or toxic reagents are used, so that the compound has the technical advantages of low cost, environment protection and the like.
Owner:SHENYANG POLYTECHNIC UNIV
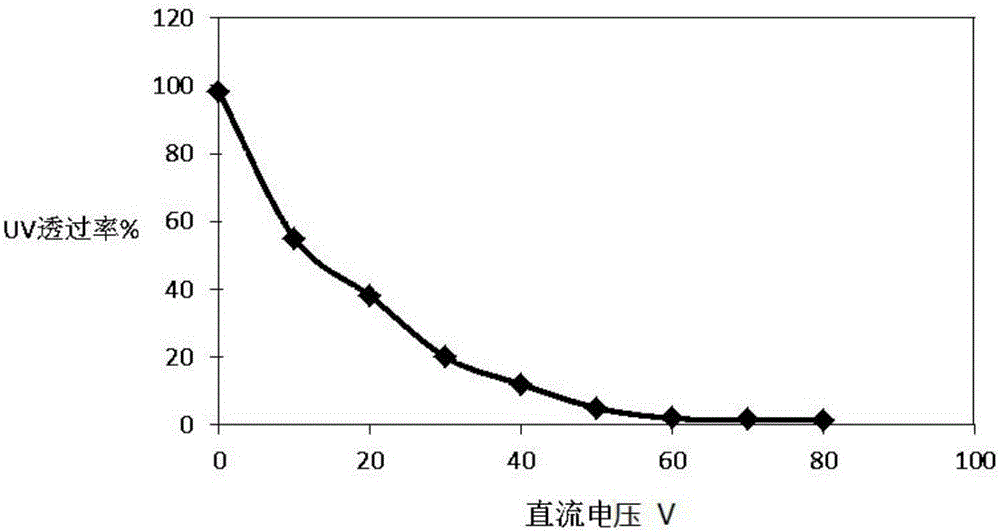
Dimming function film, dimming function assembly and dimming function device
InactiveCN106154618AControl barrierAchieve barrierStatic indicating devicesNon-linear opticsUltraviolet lightsAlternating current
The invention discloses a dimming function film, a dimming function assembly and a dimming function device. The dimming function film comprises an upper electrode layer, a dimming function layer and a lower electrode layer from top to bottom. The dimming function layer comprises a polymer dispersion liquid crystal base material and a UV blocking function compound dispersed in the polymer dispersion liquid crystal base material. The UV blocking function compound is selected from one or more of a viologen compound, dipyridine and a derivative thereof, anthraquinone and a derivative thereof, tetrathifulvalene and a derivative thereof, and pyrazoline and a derivative thereof. In the dimming function film, the compounds are dispersed in the polymer dispersion liquid crystal base material to form the dimming function layer. In the actual operation process, the compounds can be promoted to react by contorlling a direct current power source, and therefore controllable passing of ultraviolet light is achieved. The turning direction of liquid crystal molecules in a PDLC base layer is controlled by controlling the alternating current voltage, and therefore conversion between brightness and darkness is achieved.
Owner:ZHANGJIAGANG KANGDE XIN OPTRONICS MATERIAL
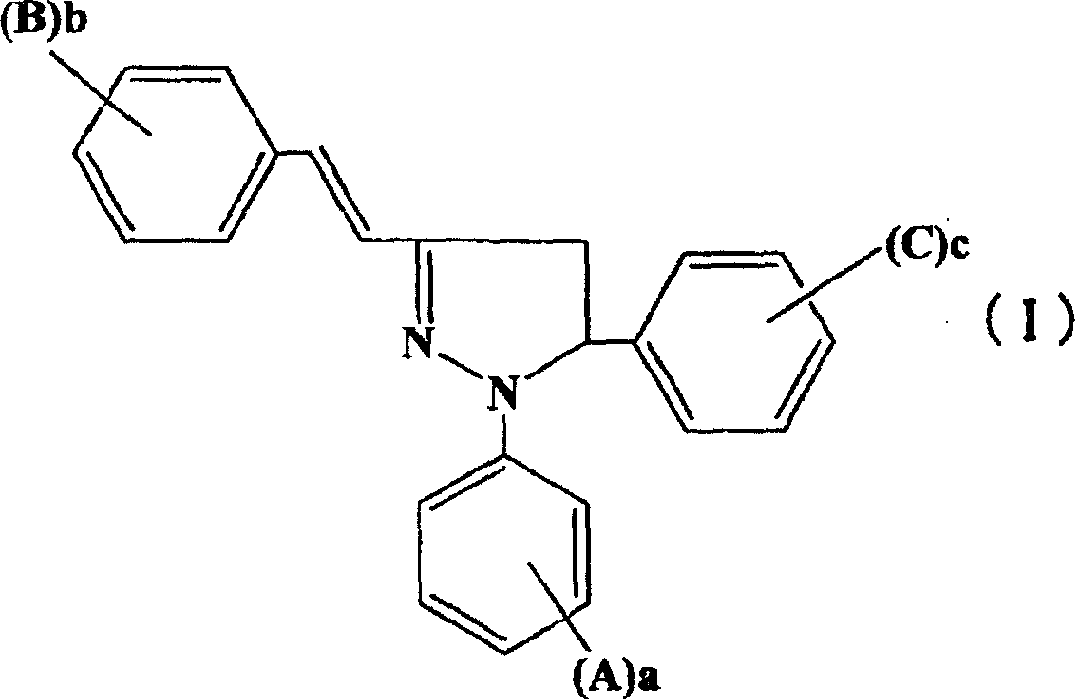
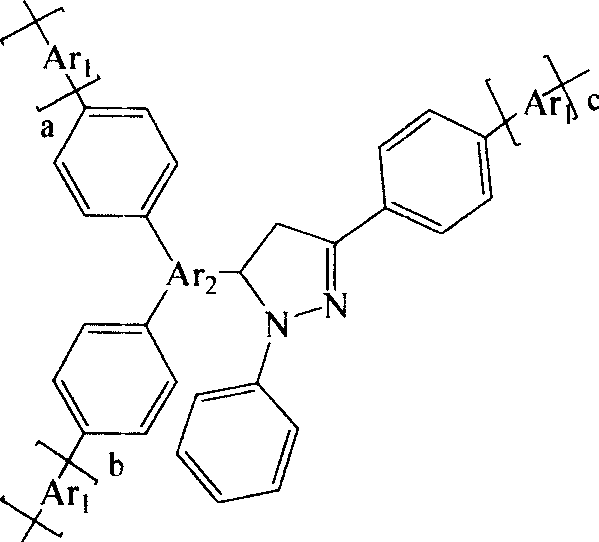
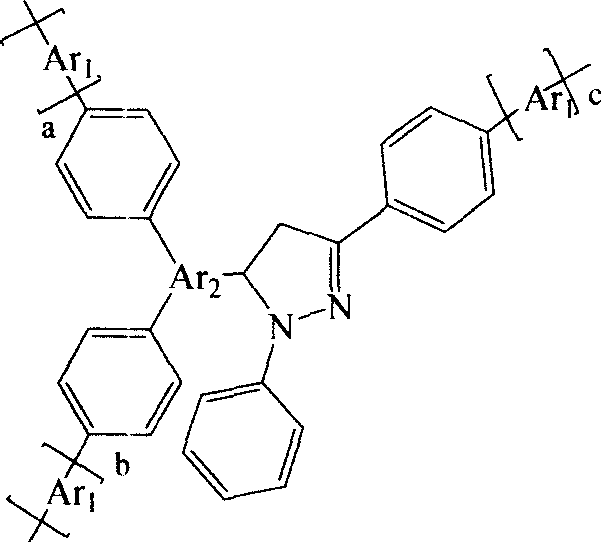
Arylpyrazoline Luminescent Compounds Substituted by 5-Position Fused Aromatic Hydrocarbons
InactiveCN102295601AEnhanced glowEnhanced Two-Photon FluorescenceOrganic chemistryLuminescent compositionsHydrazine compoundStructural formula
The general structural formula of the arylpyrazoline light-emitting compound substituted by the 5-position condensed aromatic hydrocarbon is as follows: wherein, R1 is 4-nitrophenyl, 4-cyanophenyl, 4-esterylphenyl, 2-nitro Phenyl, 2-cyanophenyl, 2-esterylphenyl, 2,4-dinitrophenyl or hydrogen atom, R2 is 4-nitro, 4-cyano, 4-esteryl, 2-nitro Group, 2-cyano group, 2-ester group or 2,4-dinitro group, R3 is 9-anthracenyl group, 9-phenanthrenyl group or 1-pyrenyl group. The preparation method is as follows: firstly, condensing aromatic aldehydes with condensed rings and aromatic ketones substituted with electron-withdrawing groups on the benzene ring to generate chalcone intermediates, and then condensing with aromatic hydrazines substituted with electron-withdrawing groups on the benzene ring to form a ring; The generated pyrazoline molecule has two chromophores, the "arylpyrazoline" and the 5-position anthracycline, which enhance charge transfer in the molecule, and the luminescence spectrum of the 5-position aromatic condensed ring is compatible with the arylpyrazoline chromophore The absorption spectra overlap to produce effective energy transfer, improve the luminescence and two-photon fluorescence of pyrazoline molecules.
Owner:SOUTHEAST UNIV


![1H-pyrazolo[3,4-<i>b</i>]pyridines and therapeutic uses thereof 1H-pyrazolo[3,4-<i>b</i>]pyridines and therapeutic uses thereof](https://images-eureka.patsnap.com/patent_img/a208a788-d164-4d46-bdfe-b503be2f9d03/US08450340-20130528-C00001.png)
![1H-pyrazolo[3,4-<i>b</i>]pyridines and therapeutic uses thereof 1H-pyrazolo[3,4-<i>b</i>]pyridines and therapeutic uses thereof](https://images-eureka.patsnap.com/patent_img/a208a788-d164-4d46-bdfe-b503be2f9d03/US08450340-20130528-C00002.png)
![1H-pyrazolo[3,4-<i>b</i>]pyridines and therapeutic uses thereof 1H-pyrazolo[3,4-<i>b</i>]pyridines and therapeutic uses thereof](https://images-eureka.patsnap.com/patent_img/a208a788-d164-4d46-bdfe-b503be2f9d03/US08450340-20130528-C00003.png)
![1H-pyrazolo[3,4-<i>b</i>]pyridines and therapeutic uses thereof 1H-pyrazolo[3,4-<i>b</i>]pyridines and therapeutic uses thereof](https://images-eureka.patsnap.com/patent_img/ab7b9af2-18c0-4461-a0e2-7121e6fc6aba/US08618128-20131231-C00001.png)
![1H-pyrazolo[3,4-<i>b</i>]pyridines and therapeutic uses thereof 1H-pyrazolo[3,4-<i>b</i>]pyridines and therapeutic uses thereof](https://images-eureka.patsnap.com/patent_img/ab7b9af2-18c0-4461-a0e2-7121e6fc6aba/US08618128-20131231-C00002.png)
![1H-pyrazolo[3,4-<i>b</i>]pyridines and therapeutic uses thereof 1H-pyrazolo[3,4-<i>b</i>]pyridines and therapeutic uses thereof](https://images-eureka.patsnap.com/patent_img/ab7b9af2-18c0-4461-a0e2-7121e6fc6aba/US08618128-20131231-C00003.png)