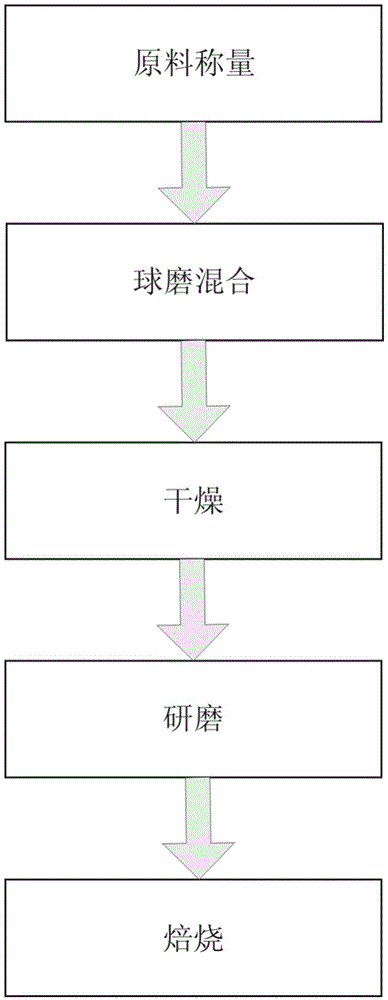
Patents
Literature
152 results about "Iron(II) phosphate" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
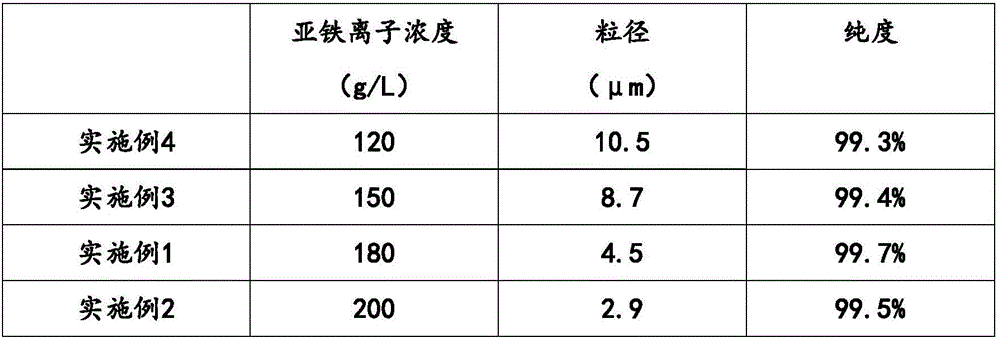
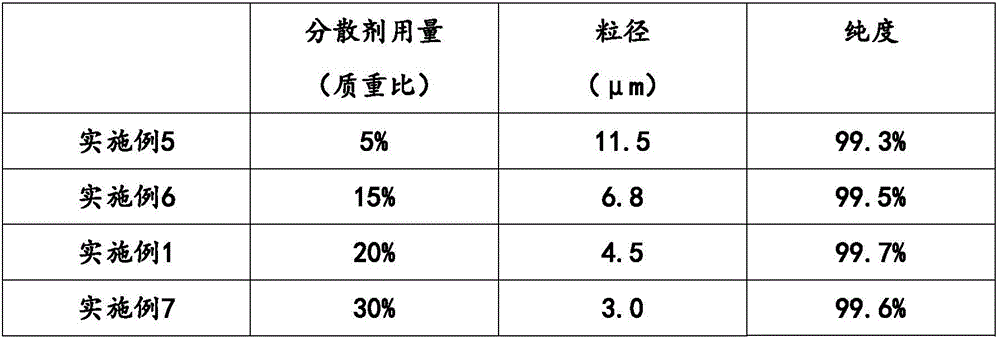
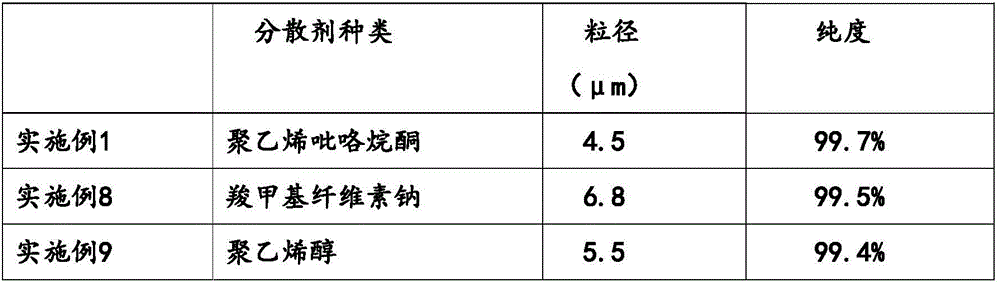
Iron(II) phosphate, also ferrous phosphate, Fe₃(PO₄)₂, is an iron salt of phosphoric acid. It is used in garden chemicals such as moss killers and slug pellets.
Method for preparing carbon coated lithium ferrous phosphate composite material including metal conductive agent
InactiveCN1649189AImprove electronic conductivityImprove protectionElectrode manufacturing processesPhosphatePhosphoric acid
This invention relates to a preparation method for LiFe PO4 / M / C compound positive material, the precursor of Li-Fe-PO4-M is synthesized with Li salt, Fe compound, phosphate, silver salt and organic acid as the raw materials by the sol-coagel method. Then the precursor mixed with pyrogenic-decomposed in inert atmosphere to get LiFePO4 / M / C compound positive material.
Owner:ZHEJIANG UNIV
Ferrous phosphate (II) powders, lithium iron phosphate powders for li-ion battery, and methods for manufacturing the same
ActiveUS20120321958A1Uniform and small grain sizeUniform and small sizePhosphatesConductive materialLithium iron phosphatePhosphate
Ferrous phosphate (II) (Fe3(PO4)2) powders, lithium iron phosphate (LiFePO4) powders for a Li-ion battery and methods for manufacturing the same are provided. The ferrous phosphate (II) powders are represented by the following formula (I):Fe(3-x)Mx(PO4)2.yH2O (I)wherein, M, x, and y are defined in the specification, the ferrous phosphate (II) powders are composed of plural flake powders, and the length of each of the flake powders is 0.5-10 μm.
Owner:NATIONAL TSING HUA UNIVERSITY +1
Ferrous phosphate doping lithium anode material and preparation process
InactiveCN1921187AImprove discharge capacityLow costElectrode manufacturing processesPhosphorus compoundsPhosphateFerrous salts
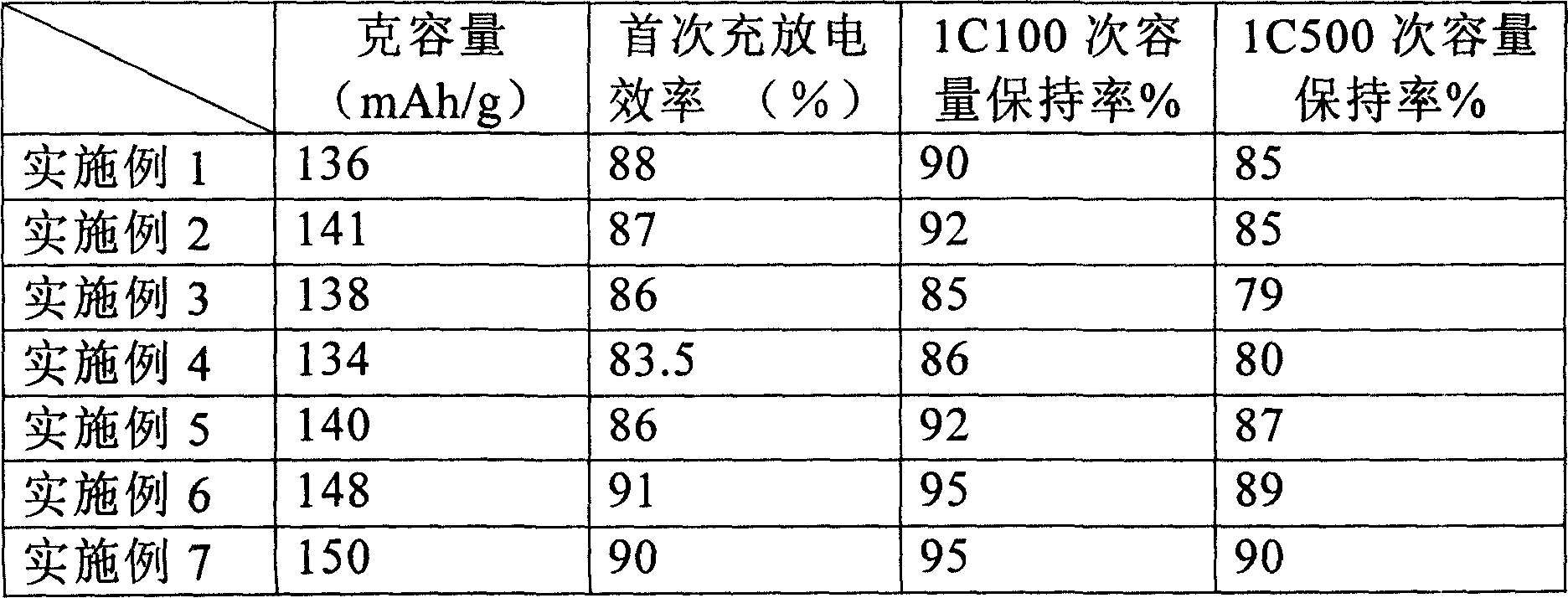
The invention relates to a lithium anode material doped with phosphate ferrous iron, and relative preparation. Wherein, the product has common advantages of lithium battery, while its discharge capacity can reach 150mAh / g, the 500-circled capacity is over 90%, with low cost. The invention is characterized in that: its formula is LiFe0.99M0.01PO4 / C; M is Cr, Zn, and Ca; the preparation comprises that (1) mixing and grinding lithium carbonate, ferrous salt, chromate salt, zinc salt, calcium salt, phosphorus source and glucose; (2), preheating the powder material in step (1) in inertia gas and low temperature; (3), grinding the material in step (2), under inertia gas, to be burnt, cooled and screened at 300-deal screen. The invention can be used in the anode of lithium battery.
Owner:XINXIANG ZHONGKE SCI&TECH
Spherical porous high-density LiFePO4 powder and process for preparing same
The invention refers to a method which prepare high-density global c, using ferrous salt, phosphorus acid, ammonia and lithium hydrate, lithium phosphate as initial material, firstly, adding ferrous salt solution and phosphor fountain in reactor by metering pump, adjusting pH value in reactor by queous alkali, occurring deposition react, the precipitate overflowing along with mother liquid, washing, filtrating and drying the effluent deposit would receive global hydrated ferrous phosphate; confecting ferrous phosphate and lithium phosphate suspend, getting mole ratoo of Li+:Fe2+:PO43- is 1:1:1, then adding carbon fountain, getting lithium phosphate and ferrous phosphate mix prodromal body after intimate mixing, solid-liquid separation; Calcining prodromal body for 5-30h in 500-800Deg C inertia and deoxidized atmosphere, cooling to room temperature with furnace, then getting global ferric lithium phosphate powder. The grain-size range of this powder is 0.5-30 mum, particle mean size is 8-13 mum, tap density is as high as 1.5- 2.0g / cm3, first discharge capacity is bigger than 120mAh / g, discharge voltage flat roof is 3.4V, hasing fine application foreground.
Owner:JIANGSU UNIV
Wet method of preparing lithium ferrous phosphate and its prepared lithium ferrous phosphate
The present invention provides a method for preparing ferrous lithium phosphate by using wet method. Said method includes the following steps: making required water-soluble compounds containing Li, Fe and P and containing alloying element M respectively be dissolved in water; stirring them and placing them into a reactor to obtain a suspension, in which the Li content, Fe content and P content must be met with the following formula: [mLi+n(1-m) / n M]; pFe:qPO4=1:1:1, in said formula n is chemical valence of alloying element M, m is mole number of Li, (1-m) / n is mole number of alloying element M and the P and q respectively are mole numbers of Fe and PO4; adding reduction electro-conductive additive, spray drying suspension, roasting and pulverizing so as to obtain the invented product.
Owner:北京环宇赛尔新能源科技有限公司
Method for measuring content of ferrous iron and ferric iron in ferrous phosphate lithium anode material
InactiveCN102323228AQuick responseResponse speed is efficientColor/spectral properties measurementsPhosphatePhenanthroline
The invention discloses a method for measuring the content of ferrous iron and ferric iron in a ferrous phosphate lithium anode material, which is used for carrying out sample treatment and sample measurement under a protective atmosphere and comprises the following steps: measuring the content of the ferrous iron: dissolving the ferrous phosphate lithium anode material by using acid; then, when the pH of a solution is regulated to 2-9, adding phenanthroline to produce an orange complex with the ferrous iron; and carrying out a sample measurement process of measuring the absorbance of the orange complex by using a spectralphotometer, and measuring the content of the ferric iron: dissolving the ferrous phosphate lithium anode material by using the acid; when the pH of the solution is 1-3, adding sulfosalicylic acid to produce a purple complex with the ferric iron; and measuring the absorbance of the purple complex by using the spectralphotometer. The invention also discloses a protective atmosphere device designed to test the method for measuring the content of the ferrous iron and the ferric iron, which comprises an admission pipe, an exhaust pipe, a box body, an operating space and operating gloves, wherein the operating space is filled with inert gases, such as argon, helium, neon, nitrogen and carbon dioxide.
Owner:NANOCHEM SYST SUZHOU
Method for preparing iron oxalate
InactiveCN101386575AMeet iron source requirementsImprove conductivityCarboxylic acid salt preparationPhosphateFiltration
The present invention discloses a method for preparing ferrous oxalate, which belongs to the technical field of powdered material preparation. The aim of the invention is to provide a preparation method of ferrous oxalate which is high in purity, controllable in granularity, even in grain size, and high in conductivity. The method comprises the following steps of locating ferrous sulfate into dilute sulphuric acid, adding scrap iron with stirring, and obtaining a ferrous sulfate solution after suction filtration; dissolving oxalic acid or / and ammonium oxalate in distilled water with stirring and heating, and obtaining a mixed solution of oxalic acid or / and ammonium oxalate through suction filtration after dissolution; and, slowly adding the ferrous sulfate solution into oxalic acid solution, keeping the temperature, stirring, separating ferrous sulfate from the original synthetic solution after keeping stand, and obtaining ferrous oxalate powder through washing and drying. The ferrous oxalate prepared by the preparation method is a powder with a medium grain diameter of between 0.5 and 80 mu m. The granularity is completely controllable. The product purity is greater than 99.0 percent. The conductivity is high. The electrochemical performance of the ferrous phosphate salt composite material is largely improved. The conductivity of lithium iron phosphate is improved by 5 orders of magnitude.
Owner:HEFEI GUOXUAN HIGH TECH POWER ENERGY
Composite material of round LiFePO4 / C in honeycomb structure, and preparation method
InactiveCN101093888AShorten the solid phase diffusion pathImprove electronic conductivityElectrode manufacturing processesChemical/physical/physico-chemical processesSucrosePhosphate
The invention solves safety issue existed in anode material of lithium ion battery currently. The issue limits lithium ion battery to be applied to electric automobile. The disclosed product is prepared from NH4FePO4.H2O, Li2CO3 and glucose or cane sugar. Mol ratio between NH4FePO4.H2O and Li2CO3 is 2:1. Mass ratio between C in glucose or cane sugar and LiFePO4 is 1:10-30. NH4FePO4.H2O is prepared from ferrous sulfate, phosphoric acid and ammonia according to mol ratio. Using ultrasonic controlled crystallization process prepares precursor, and using solid state sintering at high temperature prepares composite material. Electrolyte with cellular structure (CS) in the invention can penetrate to inside spherical granules of lithium ferrous phosphate with CS so as to shorten path of solid phase diffusion for lithium ions and to be in favor of charge and discharge in large scaling factor. The anode material is suitable to electric automobile. The method is easy of operation.
Owner:HARBIN INST OF TECH
Ferrous phosphate powders, lithium iron phosphate powders for li-ion battery, and methods for manufacturing the same
ActiveUS20150232338A1Improve conductivitySmall sizeNon-metal conductorsCell electrodesLithium iron phosphatePhosphate
Ferrous (II) phosphate (Fe3(PO4)2) powders, lithium iron phosphate (LiFePO4) powders for a Li-ion battery and methods for manufacturing the same are provided. The lithium iron phosphate powders are represented by the following formula (II):LiFe(1-a)MaPO4 (II)wherein, M, and a are defined in the specification, the lithium iron phosphate powders are composed of plural flake powders, the length of each of the flake powders is 0.1-10 μm, and a ratio of the length and the thickness of each of the flake powder is in a range from 11 to 400.
Owner:NATIONAL TSING HUA UNIVERSITY
High-purity iron phosphate used for producing lithium ion battery positive-pole material and preparation method thereof
InactiveCN102126713ALow impurity contentImprove electrochemical performancePhosphorus compoundsFerrous saltsPhosphoric acid
The invention relates to high-purity iron phosphate used for producing a lithium ion battery positive-pole material and a preparation method thereof. The preparation method is characterized by comprising the following steps: taking ferrous salt and carbonate as raw materials; reacting to generate intermediate product ferrous carbonate precipitation, wherein, the ferrous carbonate is easy to filter, and the foreign ions are easy to wash; filtering and recovering ammonium sulfate; dissolving the ferrous carbonate precipitation; adding oxidant for oxidizing; regulating the pH value of the solution; heating and preserving the temperature for a certain time to obtain the iron phosphate precipitation; filtering, drying and precipitating to obtain the high-purity iron phosphate; and mixing and calcining the high-purity iron phosphate, the lithium source and the carbon source to obtain the lithium ion battery positive-pole material with excellent electrochemistrical performance. By using the method provided by the invention, the impurity content in the iron phosphate is effectively lowered, the process time is shortened, the energy consumption is reduced, and the cost and the consumption of the raw materials are lowered; and the filter liquor is easy to recover, and the production cost is very low, thus being beneficial for large-scale production in scaled industry.
Owner:SICHUAN UNIV
Doped modified lithium iron phosphate and preparation method thereof
InactiveCN102881901AAvoid synthetic stepsSolve the problem of impurityCell electrodesPhosphorus compoundsTest batteryLithium iron phosphate
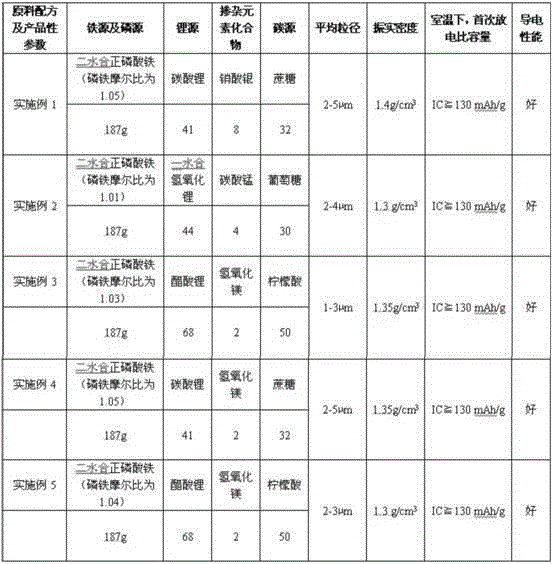
The invention discloses a raw material formula of doped modified lithium iron phosphate. Components include a ferric iron source, a phosphorus source, a lithium source, a doped element compound and excess carbon sources, wherein the mole ratio of to ferric iron source to the phosphorus source to the lithium source to the doped element compound to the carbon sources is 1:1-1.05:1-1.1:0.01-0.05:0.1-0.2. A preparation method directly adopts the ferric iron source, so that a cockamamie step of synthesizing ferrous salt by means of other synthesizing technologies is avoided, and the problem that usual products produced by means of conventional preparation methods using a ferrous salt raw material oxidizable in the air are impure is solved. A synthesized doped modified lithium iron phosphate material is small in particle diameter, narrow in distribution, high in purity, large in compaction and excellent in electrochemistry performance. A synthesized positive pole material and a carbon negative pole are assembled to form a test battery, the specific capacity is more than 130mAh / g when discharging is performed in 1C multiplying power, and excellent circulating stability performance is displayed.
Owner:绵阳天明新能源科技有限公司
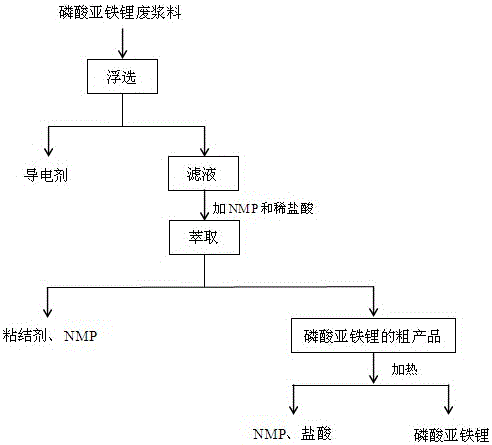
Method for recycling lithium ferrous phosphate material from waste lithium ferrous phosphate battery positive plates
ActiveCN106241769AAvoid hard-to-clean problemsSimple stepsWaste accumulators reclaimingPhosphorus compoundsPhosphateSlurry
The invention discloses a method for recycling a lithium ferrous phosphate material from waste lithium ferrous phosphate battery positive plates, wherein the method comprises the following steps: roasting the waste lithium ferrous phosphate battery positive plates, crushing, sieving and recycling positive electrode aluminum sheets to obtain a lithium ferrous phosphate waste slurry; soaking the lithium ferrous phosphate waste slurry in deionized water, and floating to remove a conductive agent, to obtain a mixture of lithium ferrous phosphate and a binder; adding the mixture to a mixed solution of N-methylpyrrolidone and hydrochloric acid, and extracting the binder, to obtain a lithium ferrous phosphate crude product; heating to remove N-methylpyrrolidon NMP and hydrochloric acid; and filtering, washing and drying to obtain lithium ferrous phosphate. The conductive agent and the binder are gradually removed or separated, so as to obtain the lithium ferrous phosphate crude product; the method is simple in steps, easy to operate and low in cost, avoids the problem of energy consumption of high temperature roasting, also avoids the problem that acid soluble precipitated impurities are difficult to remove cleanly, and is conducive to industrialization realization.
Owner:HEFEI GUOXUAN HIGH TECH POWER ENERGY
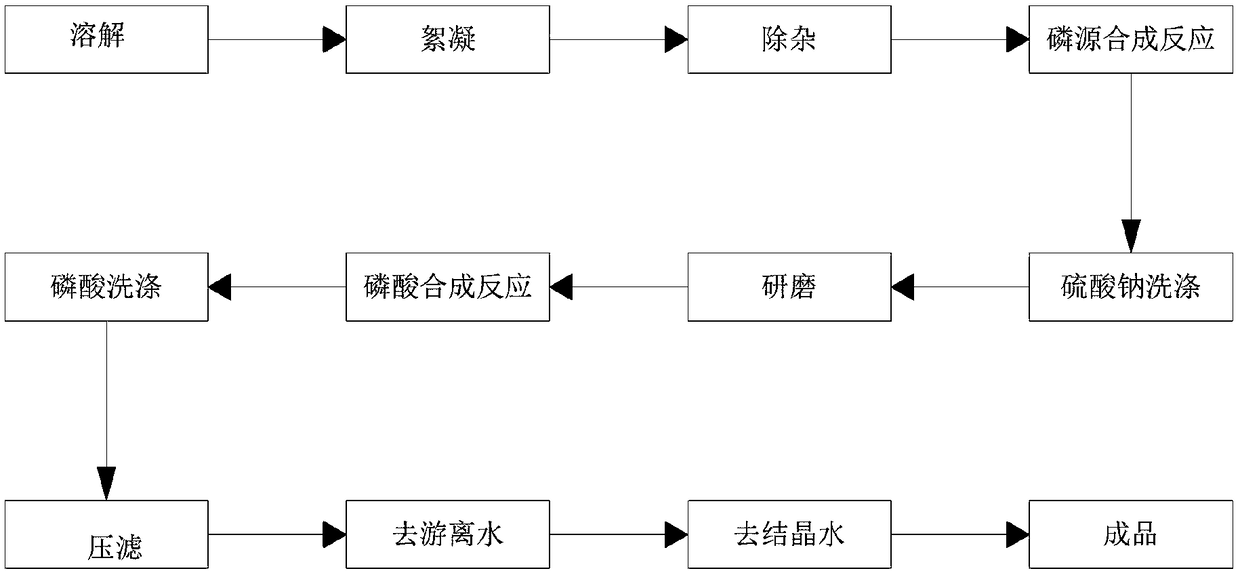
Method for producing battery-grade ferric orthophosphate from titanium dioxide solid waste
The invention belongs to the field of new energy battery materials, and comprises a method for producing battery-grade ferric orthophosphate from titanium dioxide solid waste. The method comprises thefollowing effective steps: adding water for thoroughly dissolving the titanium dioxide solid waste, adding a flocculant, and then performing multi-stage sedimentation filtration to obtain a high-purity ferrous sulfate solution; thoroughly mixing the prepared ferrous sulfate solution and a phosphorus source solution according to a molar ratio of 1:1 in a reaction kettle, then controlling the pH ofthe solution to be 5-7, controlling the temperature at 25-35 DEG C, reacting for about 2 hours while stirring to obtain a ferrous phosphate slurry and sodium sulfate; filtering and washing the ferrous phosphate slurry and the sodium sulfate after a reaction to remove sodium sulfate, slurrying a filter cake after washing, and dispersing and sanding the ferrous phosphate slurry; mixing the sanded ferrous phosphate slurry and phosphoric acid according to a molar ratio of 1:1 to 1:2 in the reaction kettle, adding an oxidizing agent, thoroughly reacting, heating the slurry to 80-100 DEG C, and aging for 2-3 hours; cooling a ferric phosphate slurry after the reaction, washing and performing tympanic membrane suction filtration, then conveying and drying to remove free water, and then removing crystal water through calcination to obtain the battery-grade ferric orthophosphate.
Owner:SHANDONG LUBEI ENTERPRISE GROUP
Lithium iron phosphate and ferrous phosphate, and preparation methods thereof
InactiveCN102070132ALow costHigh bulk densityCell electrodesPhosphorus compoundsIron saltsPhosphate ion
The invention discloses lithium iron phosphate and ferrous phosphate, and preparation methods thereof. The preparation method of the ferrous phosphate comprises the following steps of: mixing bivalent iron salt aqueous solution and phosphate ion aqueous solution under the protection of inert gas; controlling the pH value in the reaction process to be between 6.0 and 7.0; drying after the reaction is finished to obtain a spherical ferrous phosphate hydrate precursor; and performing thermal processing to obtain the ferrous phosphate. The preparation method of the lithium iron phosphate comprises the following steps of: uniformly mixing the obtained ferrous phosphate, a phosphorus source compound, a lithium source compound and a carbon source compound; and performing thermal treatment in an inert gas atmosphere at a high temperature to obtain the lithium iron phosphate. The ferrous phosphate precursor with the particle diameter of between 0.5 and 10 mu m can be prepared by the method; reduction reaction is not required in the process of preparing the lithium iron phosphate from the precursor, so the thermal treatment temperature can be greatly reduced; and the prepared lithium iron phosphate material has higher stacking density and higher electrochemical performance and has a great application prospect in the field of lithium ion batteries.
Owner:TSINGHUA UNIV
A regeneration treatment method of lithium iron phosphate waste
ActiveCN109193059AAdaptableSimple processCell electrodesWaste accumulators reclaimingScrapChemical energy
The invention discloses a regeneration treatment method of lithium iron phosphate waste, belonging to the technical field of methods and devices for directly converting chemical energy into electric energy. The method includes eight steps of dissolving, separating, adjusting composition, preparing ferrous phosphate precursor, preparing lithium phosphate precursor, preparing mixed slurry, preparinguncoated lithium iron phosphate and coating and sintering. This method can recover and dispose all kinds of abnormal production wastes, such as of unbalanced proportion, unqualified physical and chemical properties, serious oxidation, magnetic substance exceeding standard, etc., and has strong adaptability. And the process is simple, no complex smelting separation, saving a lot of chemical raw materials, low recovery cost. The lithium, iron and phosphorus elements in the waste can be recycled, ferrous phosphate and lithium phosphate can be prepared at the same time, and the prepared lithium iron phosphate positive electrode material has excellent performance, and can be directly used in the preparation of lithium iron phosphate power batteries.
Owner:嘉兴晟源工业设计有限公司
Preparation method for lithium iron phosphate/carbon composite positive material with high multiplying power
ActiveCN104743537ACoated evenlyUniform coating, which is conducive to the rapid deintercalation and uniformity of lithium ionsCell electrodesPhosphorus compoundsCarbon compositesElectrical battery
The invention relates to a preparation method for a lithium iron phosphate / carbon composite positive material with high multiplying power. The preparation method comprises the following steps: (1) stirring and mixing a phosphor source solution with an iron source solution, adding a dispersing agent, and controlling the pH value of the reaction to generate ferrous phosphate precipitate; (2) adding an oxidant after supplementing a phosphor source into the obtained ferrous phosphate in a phosphor-iron ratio of (1:1) to (7:1), and regulating the pH value to synthesize iron phosphate; (3) mixing the iron phosphate with a lithium source and a carbon source in a chemical metering ratio, carrying out ball milling, drying and roasting to obtain a sheet-layer lithium iron phosphate / carbon composite positive material with high multiplying power. The positive material has good electrochemical performances including the specific discharge capacities of 0.2C, 1C and 10C are respectively 162mAh / g, 158mAh / g and 142mAh / g. The positive material is low in cost, good in technological reproducibility, excellent in electrochemical performance, and suitable for being used as a battery positive material of large mobile equipment with high multiplying power of an electric vehicle, and the like.
Owner:山东鑫动能锂电科技有限公司
Method for preparing nanometer lithium iron phosphate/carbon compound with stable low temperature performance
ActiveCN102760879ASimple processPreparation conditions are easy to controlMaterial nanotechnologyCell electrodesCarbon layerElectrical battery
The invention belongs to a novel energy material, and particularly relates to a method for preparing a nanometer lithium iron phosphate / carbon compound with stable low temperature performance. The method comprises the following steps of: mixing an iron source, a lithium source, a phosphorous source and a compound carbon source according to a certain proportion; performing ball milling; drying; and calcining in an inertial atmosphere to obtain a lithium iron phosphate / carbon compound, wherein the particle size is smaller than 150 nanometers; carbon is uniformly coated on the surfaces of particles; the thickness of a carbon layer is about 2 nanometers; and the compound carbon source plays an important role in controlling a material structure. After the material is assembled into a button cell, the discharging capacity is 160mAh / g at the rate of 0.1C at the room temperature, the discharging capacity is 126mAh / g at the rate of 0.1C at the temperature of 20 DEG C below zero, and the capacity conservation rate is still over 97 percent after 500 cycles at the rate of 0.6C at the temperature of 20 DEG C below zero, so that the problem of unstable low temperature performance of a lithium ion battery is solved. The method has the advantages of low cost, simple production process and high safety, and the prepared compound can be applied to the field of portable equipment, power electric vehicles and the like.
Owner:长春劲能科技集团有限公司
Ferrous phosphate hydrated salt crystal, producing method thereof and method for producing Li-Fe-P composite oxides
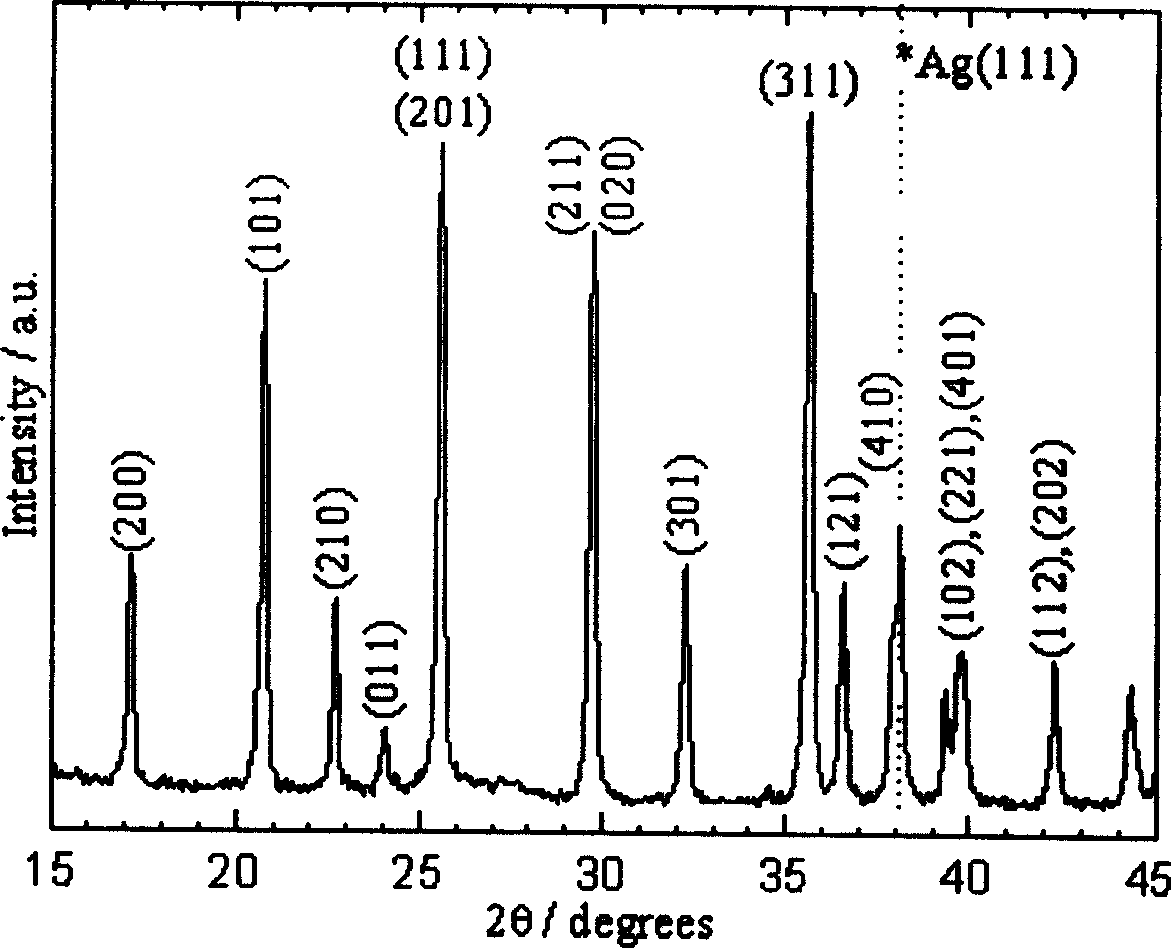
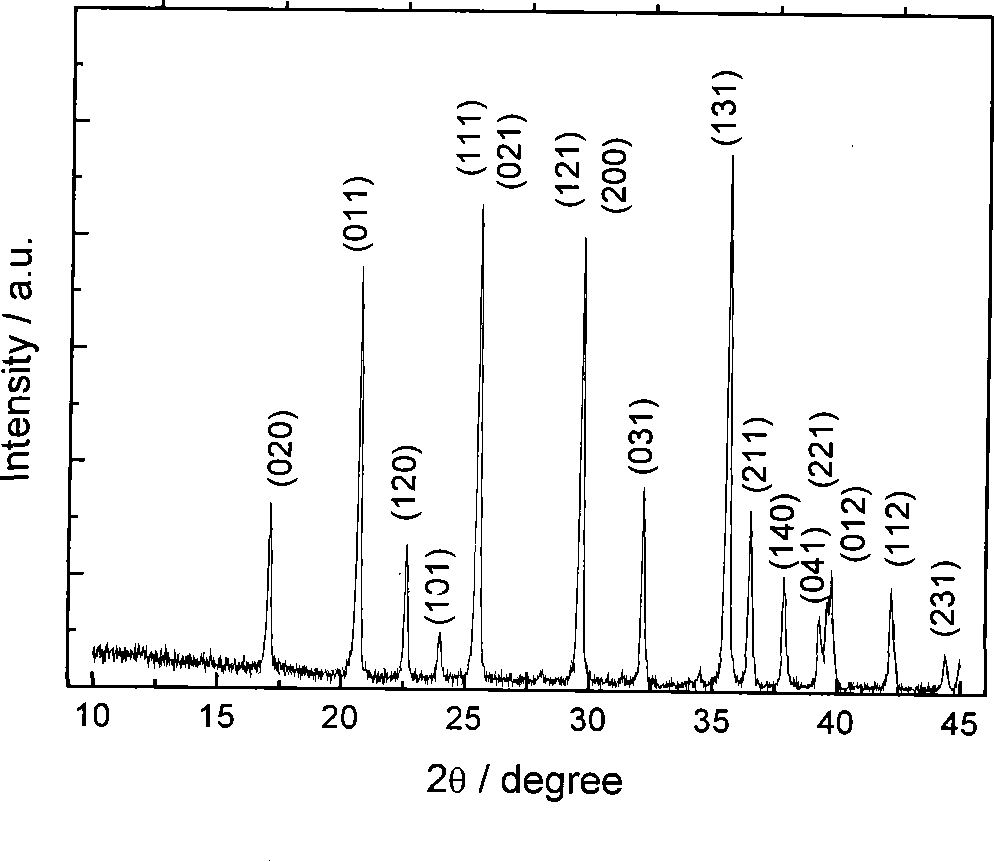
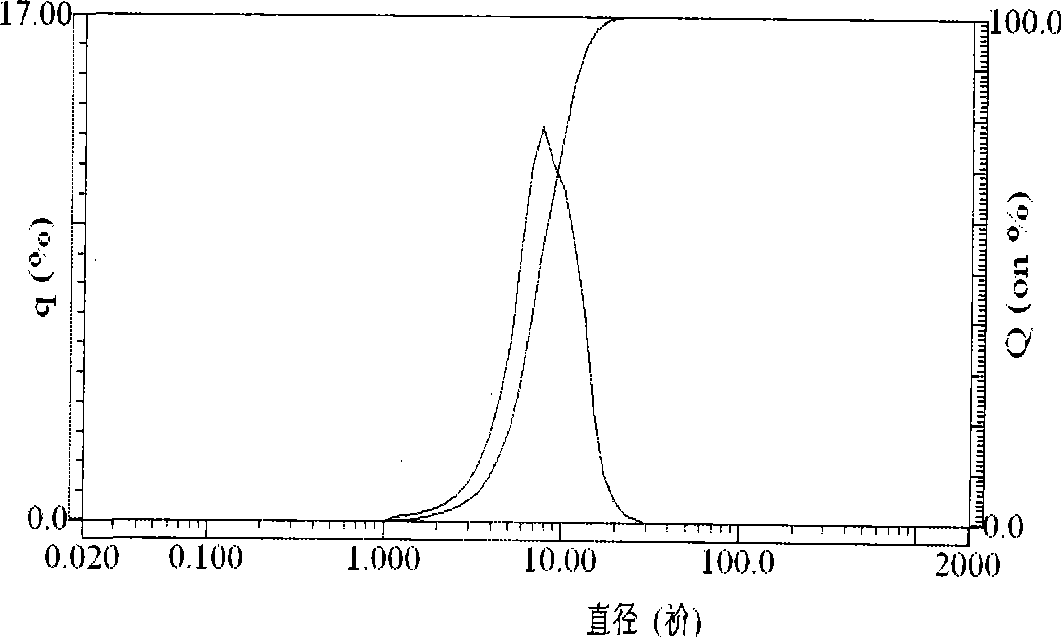
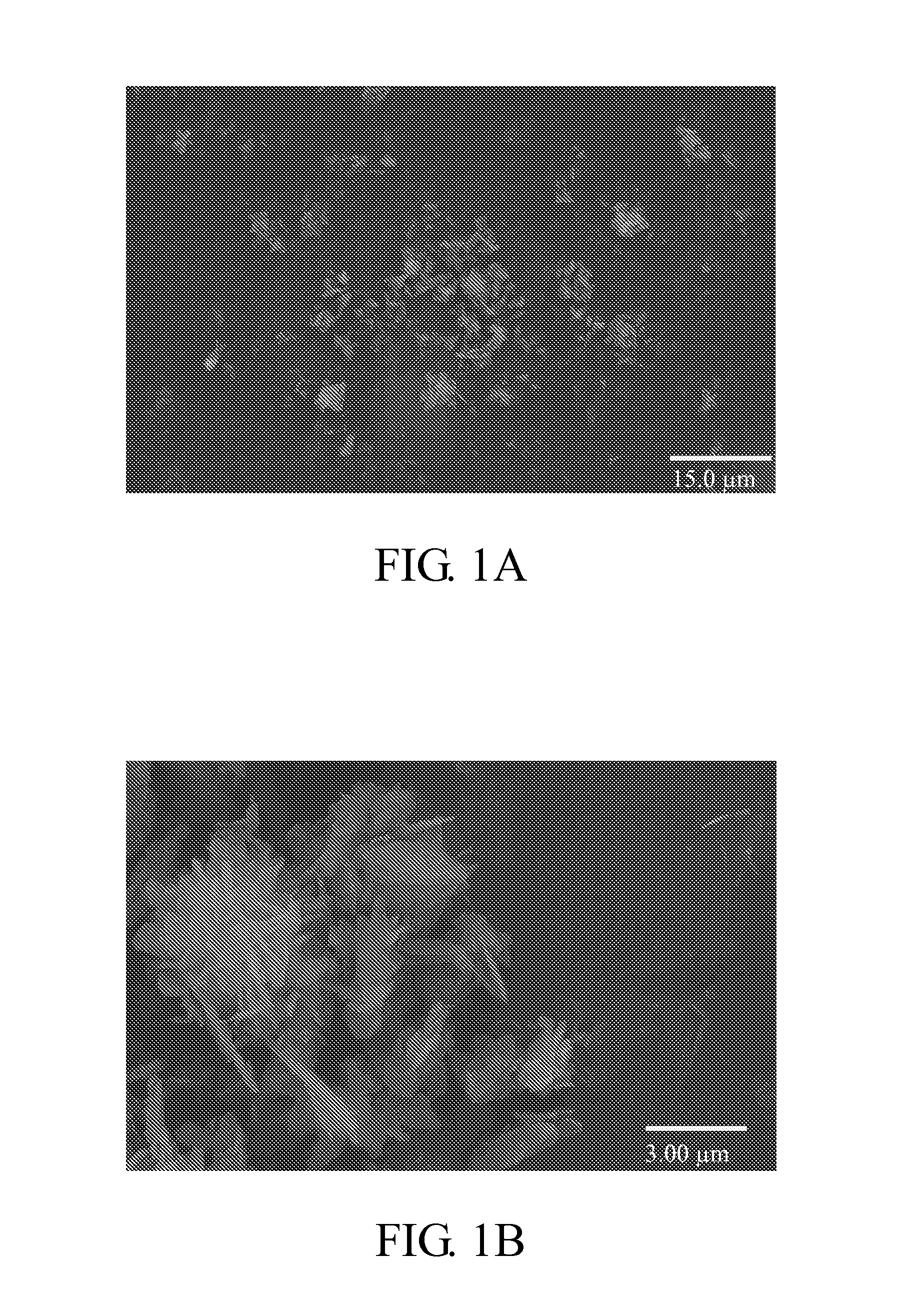
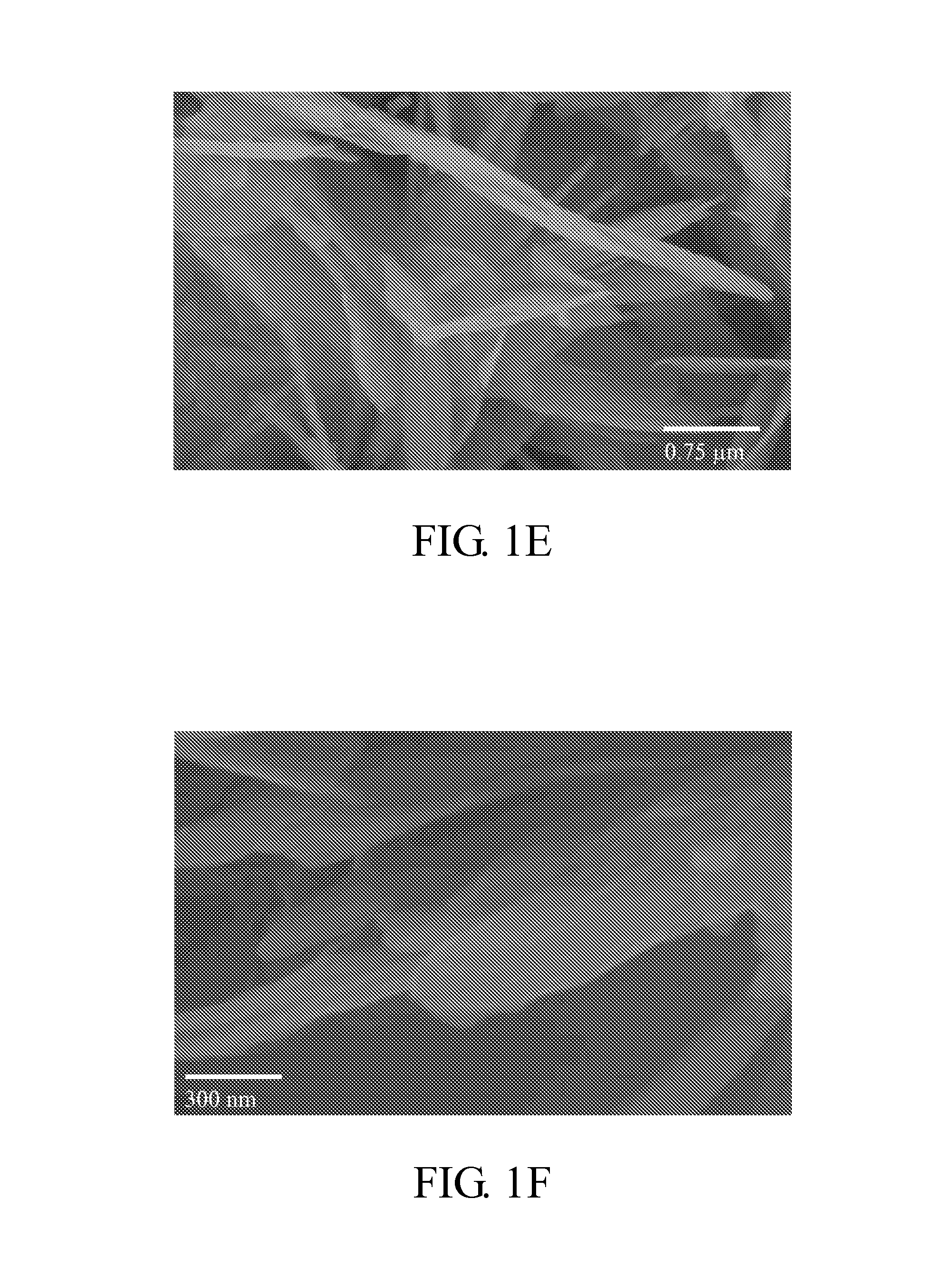
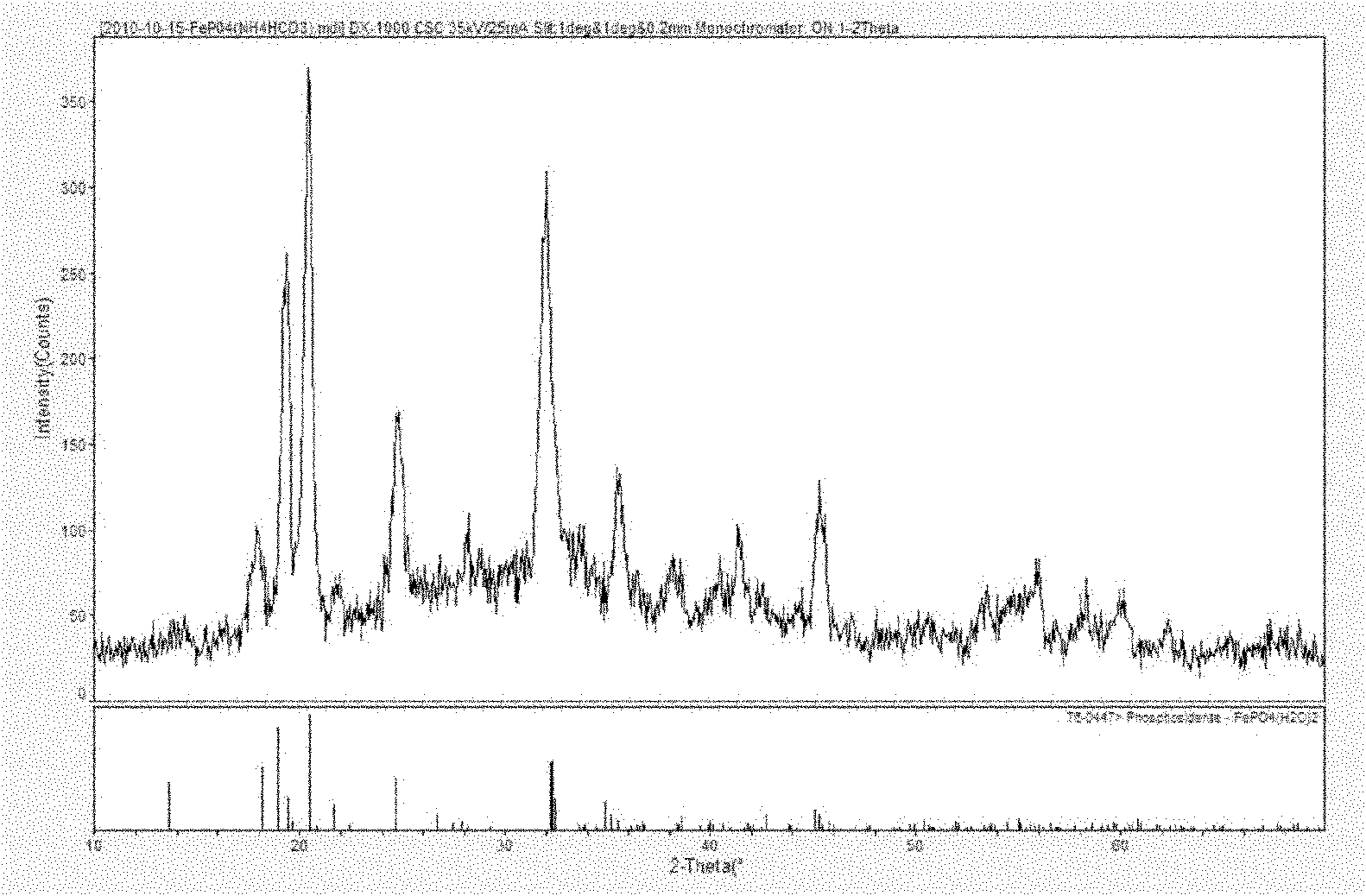
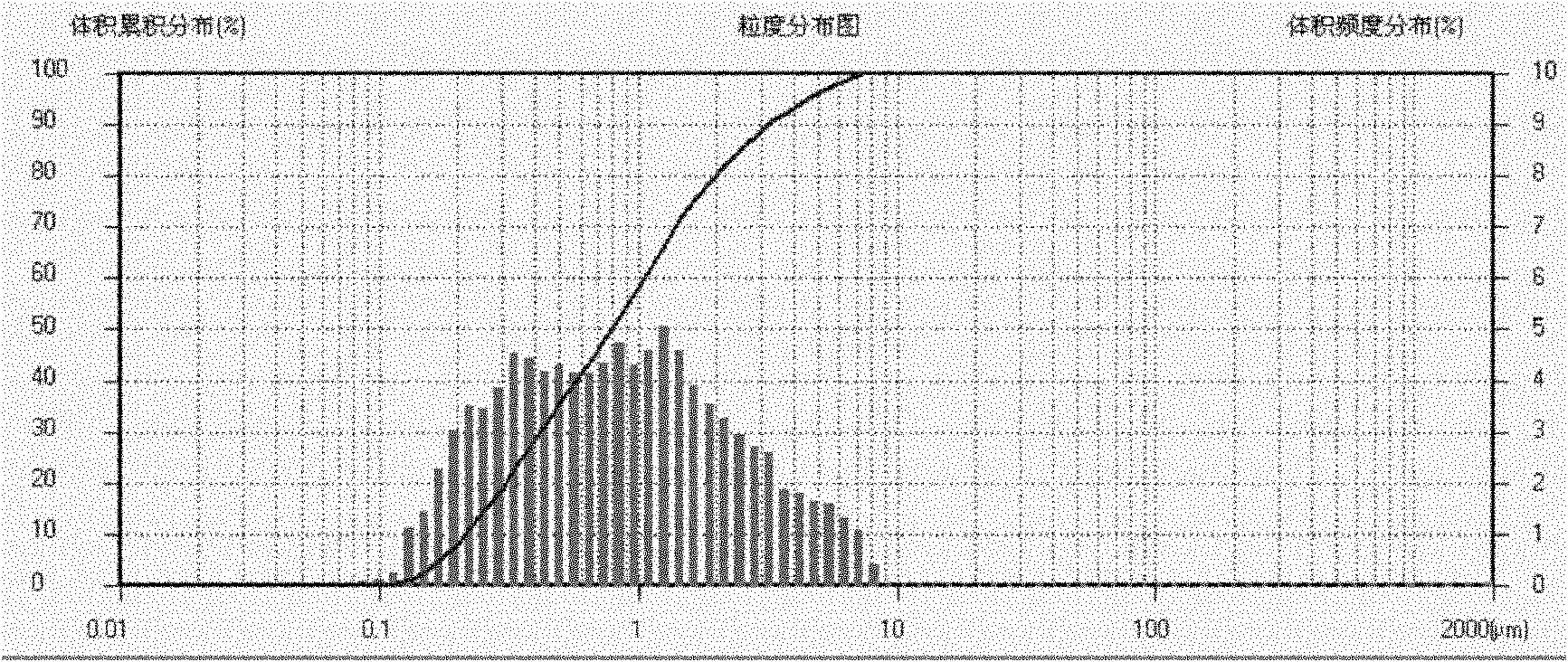
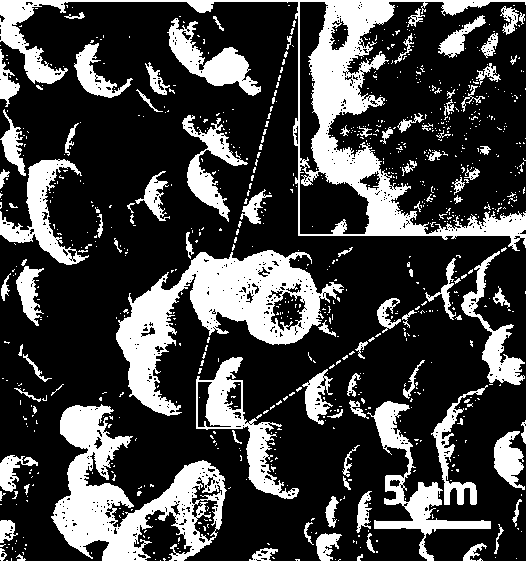
PROBLEM TO BE SOLVED: To prepare a fine ferrous phosphate hydrate crystal excellent in processability and suitable as a raw material for producing a functional inorganic material, particularly that for LiFePO<SB>4< / SB>or LiFeMePO<SB>4< / SB>(Me is at least one metal element selected from Mn, Co, Ni and Al) to be used for the positive material of a lithium secondary battery, and to provide a method for industrially advantageously producing the above crystal in a high yield and a production method for a lithium / iron / phosphorus-based complex oxide using the crystal.SOLUTION: The ferrous phosphate hydrate crystal is represented by the formula: Fe<SB>3< / SB>(PO<SB>4< / SB>)<SB>2< / SB> / 8H<SB>2< / SB>O and has an average particle size of 5 μm or less. It is preferable that the crystal has a half value width of a diffraction peak in a lattice place (020 plane) obtained by an X-ray diffraction analysis of ≥0.20°.
Owner:NIPPON CHECMICAL IND CO LTD
Preparation method of coagulant of polyphosphate ferric chloride
InactiveCN102476845AImprove the coagulation effectFast precipitationWater/sewage treatment by flocculation/precipitationIndustrial effluentPhosphate
Preparation of a coagulant of polyphosphate ferric chloride adopts waste phosphating residues produced from steel phosphating treatment as raw materials, and the coagulant is especially suitable for the treatment of oil-containing colored industrial wastewater. The product preparation of the invention adopts phosphating residues, industrial hydrochloric acid, and ferrous phosphate as raw materials, wherein the HCl content (weight ratio) is not less than 30%; the phosphating residues and industrial hydrochloric acid are polymerized according to a certain ratio so as to obtain clear orange liquid of polyphosphate ferric chloride. When the coagulant of polyphosphate ferric chloride of the invention is used in water supply and sewage treatment, when compared with existing coagulants, the product of the invention has the characteristics of high oil-removing and decoloration efficiency, few medicament adding amount, wide medicament adding range, and the like; the production processing process of the product does not cause secondary pollution; the production process is simple; waste phosphating residues are used as raw materials; the production cost is low; and thus the product of the invention has low price.
Owner:TIANJIN TANGGU XINYU ENVIRONMENTAL PROTECTION TECH
Lithium ion battery positive material lithium ferrous phosphate and preparation method
ActiveCN103531794AImprove performanceReasonable designCell electrodesSecondary cellsAdditive ingredientPhosphate
The invention discloses a preparation method for a lithium ion battery positive material LiFePO4 with iron phosphate as a raw material by utilization of nano-ceramic grinding and dispersion machine. The method comprises steps: first, lithium source, ferric phosphate and carbon source as raw materials are prepared; the raw materials are ground into nano level by a nano-ceramic grinding and dispersion machine; after desiccation, the obtained mixed material is subjected to once sintering and secondary sintering, the lithium ion battery positive material LiFePO4 is obtained. The prepared LiFePO4 has a particle size D50 of 1-6 microns, has a specific surface area of 15-25 m<2> / g, and has a tap density of being more than or equal to 1.5g / cm<3>. The process is simple and easy to control. The production cost is low. The obtained product has uniform ingredients and good physical and chemical properties and electrical properties.
Owner:CHANGSHA RES INST OF MINING & METALLURGY
Method for preparing cell grade ferrous oxalate with steel acid pickling waste liquor
ActiveCN105732359AHigh crystallinityHigh purityOrganic compound preparationCarboxylic acid salt preparationOxalateLithium iron phosphate
The invention relates to a method for preparing cell grade ferrous oxalate with steel acid pickling waste liquor.Through removing electroplating additive and adjusting the content of ferrous ions in the acid pickling waste liquor and the usage amount of dispersing agent, ferrous oxalate which is high in purity and crystallinity and even in appearance and has nanoscale can be prepared under a room temperature condition.Resource utilization optimization is adopted for the acid pickling waste liquor, in this way, economic burdens of steelworks caused by sewage treatment can be relieved, a cheap iron source is sought for preparation of cell grade lithium iron phosphate, the new method can be provided for effectively regulating the purity and particle size of cell grade ferrous oxalate, and more profits are created for the steelworks.
Owner:KUNMING UNIV OF SCI & TECH
Method for assisted preparation of carbon composite lithium iron phosphate micro-nanometer powder through industrially modified starch
InactiveCN103219514AUniform scaleCycle stability andCell electrodesPhosphorus compoundsCarbon compositesIron salts
The present invention discloses a production method for ferric phosphate nanometer powder with a characteristic of controllable particle size. In the prior art, battery grade ferric phosphate production processes commonly have disadvantages of large size, impure phase, complex process, high production cost and the like, and especially the phase carries a certain proportion of crystal water so as to directly cause instable phase, instable performances and the like of the lithium iron phosphate powder synthesized at the late stage. The synthesis process comprises: preparing a phosphate organic / pure water mixing base solution in a certain pH range, adding a plurality of additives, adding an iron salt solution at a constant temperature in a stepwise manner, adjusting the pH value of the solution during the adding process with a dropwise manner, carrying out constant temperature stirring for a certain time, carrying out suction filtration, washing and drying on the obtained product, and finally carrying out annealing for a certain time at a temperature of 300-600 DEG C to obtain the ferric phosphate nanometer powder. The ferric phosphate nanometer powder produced by the method has characteristics of controllable particle size, controllable components, simple process, low cost, high powder activity, easy production enlargement and high market competitiveness.
Owner:山东天润丰新能源科技有限公司
Production method of iron phosphate nano powder body with controllable size and granularity
InactiveCN103011117ASmall sizeGood particle size controllabilityMaterial nanotechnologyPhosphorus compoundsLithium iron phosphateIron salts
The invention discloses a production method of an iron phosphate nano powder body with controllable size and granularity. The production process of battery level iron phosphate has the defects of slightly large size, impure phase, complex process, high production cost and the like generally; and especially, a defined proportion of crystal water is carried in the phase, which directly causes phase, performance and the like of the post-synthesized lithium iron phosphate powder body to be instable. The synthetic process provided by the invention comprises the steps of: preparing a phosphate organic / pure water mixed base solution within a certain pH range; adding multiple additives; adding an iron salt solution by steps at a constant temperature; regulating pH of the solution in a dropping process; stirring for a certain time at a constant temperature; and filtering, washing and washing a product and finally annealing a temperature of 300-600 DEG C for a period of time to obtain the iron phosphate nano powder body. The iron phosphate nano powder body produced by adopting the method has the advantages of controllable size and components, simple process, low cost, high powder body activity and great market competitiveness, and is easy to produce on large scale.
Owner:山东天润丰新能源科技有限公司
Iron phosphate preparation method
PendingCN112661129AHigh purityAvoid it happening againPhosphorus compoundsPhosphateFerrous sulfate iron
An iron phosphate preparation method relates to the field of new energy material preparation, and comprises the following steps: mixing a ferrous sulfate solution and a phosphorus source in proportion, performing stirring to react for 10-120 min, adding an adjusting solution, adjusting the pH value to 3-7, performing filtering and washing to obtain ferrous phosphate; transferring ferrous phosphate into a reaction kettle, adding an impurity removing agent and a complexing agent, supplementing a phosphorus source according to a ratio of P to Fe of 1: 1-1.5: 1, then adding an oxidizing agent which is obtained by oxidizing ferrous iron into ferric iron with a stoichiometric ratio of 1.0-1.5 times, adding an adjusting solution, adjusting the pH value to 1.5-3.5, carrying out heat preservation for 30-90 minutes, heating to 80-100 DEG C, performing forming for 30-180 minutes, and carrying out precipitating, filtering, washing and drying to obtain finished ferric phosphate dihydrate; and carrying out heat preservation on the Ferric phosphate dihydrate at the temperature of 400 DEG C- and 750 DEG C for 30-360 minutes to obtain an anhydrous iron phosphate finished product. Heavy metal ions can be efficiently removed, so that the purity of iron phosphate is effectively ensured.
Owner:SICHUAN UNIV
Preparation method of high-performance ferrous phosphate
PendingCN111217346AEven growthHigh tap densityCell electrodesSecondary cellsElectrical batteryPhosphate
The invention discloses a preparation method of high-performance battery-grade ferrous phosphate. The method comprises the following steps: preparing a mixed solution of an iron source and a metal ionadditive, wherein ferrous sulfate with a concentration of 0.5-2 mol / L and a metal ion additive are mixed to prepare an iron source aqueous solution, and the molar ratio of the metal ions in the metaladditive to the iron is (0.05-0.1):1; and preparing a mixed solution of a phosphorus source, a complexing agent and a surfactant, wherein a phosphorus source, a complexing agent and a surfactant aremixed according to a certain ratio to prepare a phosphorus source aqueous solution, a molar ratio of phosphorus to iron is (0.7-0.8):1, concentrate molar ratio of the complexing agent to iron is (0.01-0.1):1, and a mass ratio of the surfactant to iron is (0.5-3%):1. According to the invention, the basic electrical property of a LiFePO4 positive electrode material can be improved, a LiFePO4 positive electrode material precursor ferrous phosphate is modified, and a mesoporous metal ion doped ferrous phosphate material is prepared by optimizing a ferrous phosphate synthesis process.
Owner:贵州唯特高新能源科技有限公司
Nanometer phosphate ferrolithium/carbon composite materials, production of solid-phase and use thereof
InactiveCN100356617CImprove cycle stabilityExcellent rate performanceElectrode manufacturing processesPhosphatesCarbon compositesPhosphate
The invention takes LiFeO4 as matrix, and uses sub-particle carbon powder to cover its surface and to fill into space between LiFeO4 particles. Grain size is 200-500um. The weight ration of carbon in composite material is 2-10%. The process of preparation feature ultrasonic wave crash and solid state reaction. The said lithium salt is FeC2O4íñH2O, and the phosphate is NH4H2PO4.
Owner:SHANGHAI INST OF MICROSYSTEM & INFORMATION TECH CHINESE ACAD OF SCI
A method for preparing nanoscale lithium iron phosphate by self-sacrificing template method
ActiveCN102267692AUniform chemical compositionControllable particle sizeNanotechnologyPhosphorus compoundsAir atmospherePhosphate
The invention relates to a self-sacrificing template method for preparing nanoscale lithium ferrous phosphate used as a cell cathode material and belongs to the field of electrochemistry. The method comprises the following specific steps: dissolving inorganic compound raw materials containing Fe<3+> and PO4<3+> in an aqueous solution, adjusting the pH value of the solution to 1-6, reacting at thetemperature of 60-150 DEG C for 3-10 hours, filtering, washing to obtain amorphous nano iron phosphate, and then baking at the temperature of 450-750 DEG C in air atmosphere for 4-16 hours to obtain a crystalline nano iron phosphate template; and on the basis of utilizing crystalline nano iron phosphate as a template, dissolving iron phosphate, a Li<+>-containing compound and a carbon-containing organic matter in water, stirring and mixing uniformly, after spray-drying, and baking at the temperature of 450-800 DEG C in a / an nitrogen / argon protective atmosphere furnace to prepare a grey-black lithium ferrous phosphate (LiFePO4) / C material. The preparation method provided by the invention is simple and low in cost; and the prepared lithium ferrous phosphate material has the advantages of high purity, good consistency, integral crystal structure, small and uniform particles and excellent electrochemical performance.
Owner:DONGFANG ELECTRIC CORP LTD
Quaternary metal phosphate lithium ion battery cathode material and preparation method thereof
InactiveCN104485441ASmall particlesImprove electrochemical performanceCell electrodesElectrical batteryPhosphate
The invention discloses a quaternary metal phosphate lithium ion battery cathode material and a preparation method thereof. The molecular formula of the quaternary metal phosphate lithium ion battery cathode material is LiNi0.05Mn0.25Fe0.3Co0.4PO4. The preparation method of the quaternary metal phosphate lithium ion battery cathode material comprises the following steps: adding reduced iron powder into a phosphoric acid solution to react so as to form a ferrous phosphate precursor, then dropwise adding lithium hydrate and a sugar solution into the ferrous phosphate precursor for reaction, adding the obtained mixture containing the ferrous phosphate precursor into a nanometer ball mill for ball milling, then adding cobaltous hydroxide, manganese carbonate and nickel protoxide for mixing and milling, after the obtained quaternary phosphate precursor solution spray is dried and placed into a crucible, controlling the temperature to be 600 to 650 DEGC, and calcining under a nitrogen atmosphere for 5 to 9 hours to obtain the quaternary metal phosphate lithium ion battery cathode material with an even spherical structure. The quaternary metal phosphate lithium ion battery cathode material has excellent electrochemical properties, has a specific capacity of 143mAh / g and an energy density of 610Wh / Kg, and has a capacity retention ratio of 96.5 percent after 10 times of circulation.
Owner:SHANGHAI INST OF TECH
Phosphorus-removing adsorbent for waterworks and preparation method thereof
InactiveCN107899547AImprove adsorption capacityWater quality up to standardOther chemical processesWater contaminantsSorbentPhosphate
The invention discloses a phosphorus-removing adsorbent for waterworks and a preparation method thereof. The phosphorus-removing adsorbent is prepared from dead branches and leaves in gardens, potassium carbonate, diatomite, ferric chloride, chitosan, citric acid and other raw materials. The adsorbent of the invention has a moderate pore structure and abundant active adsorption sites, and producesa small amount of divalent and trivalent iron ions in phosphorus-rich water for production of iron phosphate and ferrous phosphate precipitates with phosphate radicals in the water; the obtained products are conjugated with a diatomite-coated adsorption material; and the finally obtained products are recovered in virtue of superparamagnetism. The phosphorus-removing adsorbent provided by the invention can be used as a technical solution for treating sudden phosphorus pollution in the waterworks to ensure that the quality of water supplied by the waterworks meets the standard, and has the advantages of low treatment cost, simple operation, capacity of treating the waste with the waste, and no secondary pollution.
Owner:DANGTU COUNTY RUILONG FRUIT TREE PLANTING SPECIALIZED COOP
Synthetic method of lithium iron phosphate sizing agent
InactiveCN101740757AReduce manufacturing costImprove liquidityCell electrodesChemical industryCarboxymethyl cellulose
The invention discloses a synthetic method for lithium iron phosphate sizing agent, including the following steps: 1. putting sodium carboxymethyl cellulose into the water, stirring the liquid fully until sodium carboxymethyl cellulose and water turn into colloidal solution; 2. adding conductive agent and mixing the mixture thoroughly; 3. adding lithium iron phosphate and mixing the mixture evenly; 4. adding N-methylpyrrolidone and stirring the mixture evenly; and 5. adding GR-S and stirring the mixture evenly. The invention is simple in procedure and operation. Sodium carboxymethyl cellulose is used as dispersant and thickening agent, and GR-S is used as caking agent, the sodium carboxymethyl cellulose and the GR-S are both common materials in the chemical industry with wide sources and low price. Lithium iron phosphate sizing agent prepared by the synthetic method of the invention is good in fluidity, stable in solid content and viscosity and suitable for large scale industrial production, thus greatly reducing the production cost of lithium iron phosphate batteries.
Owner:东莞市金源电池科技有限公司