Patents
Literature
214results about How to "Extend cooling time" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
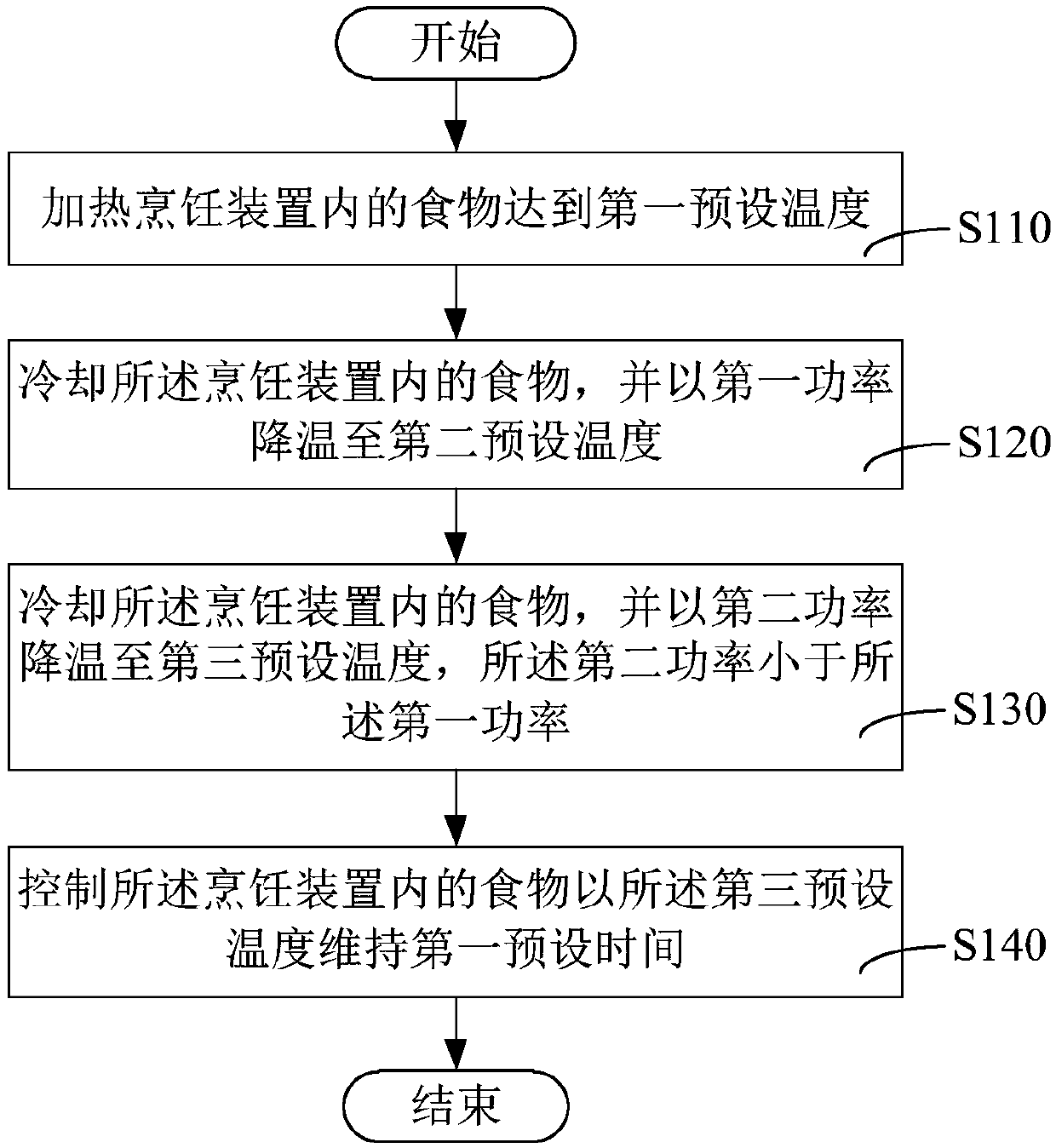
Cooking control method and cooking device
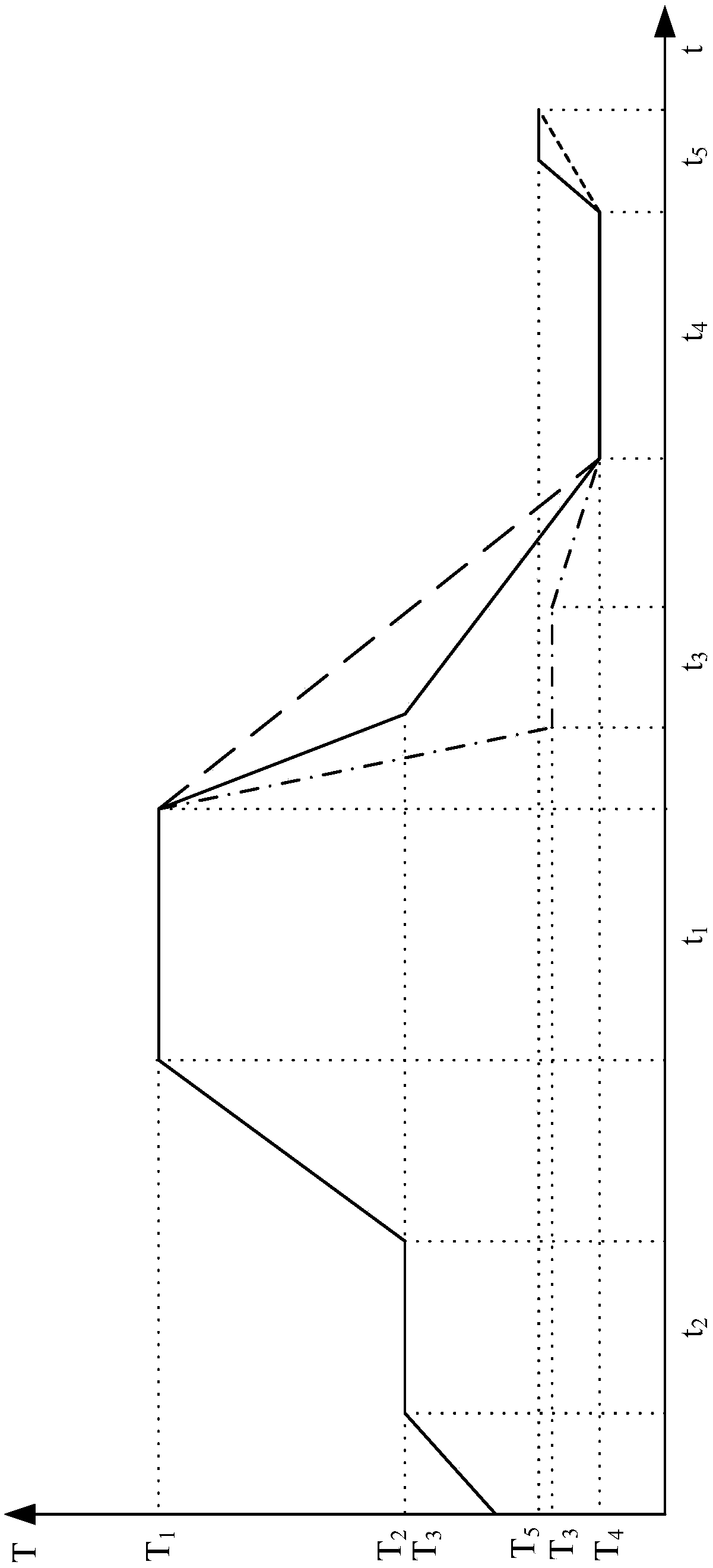
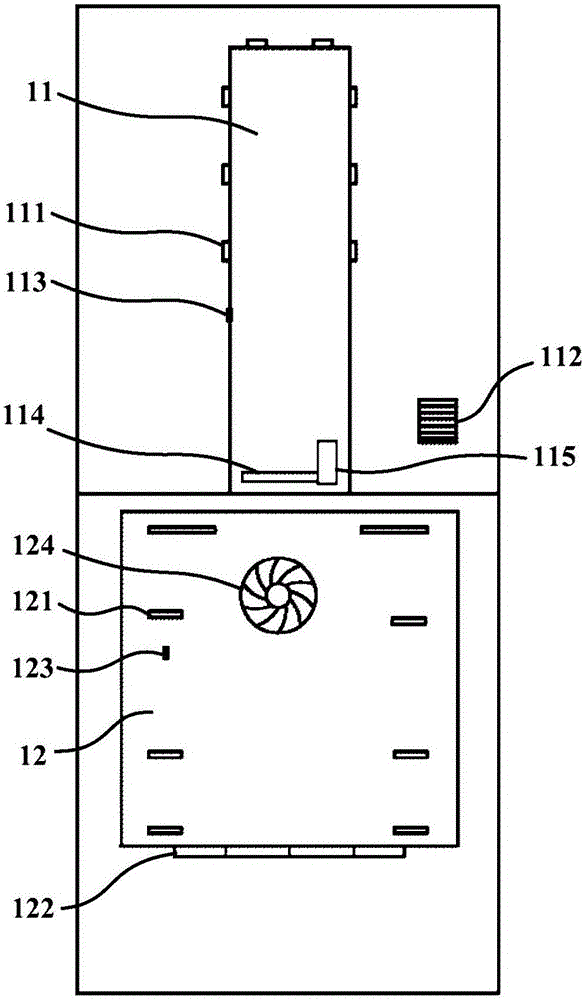
The invention relates to a cooking control method and a cooking device. The method comprises the steps that food is put into a containing cavity of a shell, and a controller controls a heater to heatfood to reach a first preset temperature; the controller controls a cooler to cool the food at the first power to a second preset temperature; furthermore, the food is cooled to a third preset temperature at the second power, wherein the second power is smaller than the first power; when the food is cooled at the first power, the food can be cooled to the second preset temperature at high speed, the efficiency of resistant starch generation is effectively improved, and the time which is not beneficial to resistant starch generation is shortened; when cooling is conducted at the second power, the cooling time can be effectively prolonged, which is more beneficial to formation of crystal nucleus of resistant starch in the food at the early stage; the food is subjected to heat preservation form first preset time at the third preset temperature, so that the food keeps at the third preset temperature which is beneficial to resistant starch generation, and the content of the resistant starchin the food can be further increased.
Owner:GREE ELECTRIC APPLIANCES INC
Preparation method of low-hydration-heat polycarboxylate water reducing agent
The invention discloses a preparation method of a low-hydration-heat polycarboxylate water reducing agent. Unsaturated carboxylic acid or unsaturated carboxylic acid anhydride performs acidylation andesterification with alcohol amine and a compound A to simultaneously prepare unsaturated monomers with acidamide groups, unsaturated monomers with amino groups and unsaturated monomers with benzene rings and phosphate groups; then, co-polymerization is performed by excessive unsaturated carboxylic acid or unsaturated carboxylic acid anhydride, unsaturated sulphonate and polyether big monomers during acidylation and esterification; acylamide groups, amino groups, phosphate groups, carboxylate groups, sulphonate groups, polyether lateral chains and benzene rings are introduced into molecular structures, so that the product has a high initial water reducing rate. The molecular structures of the water reducing agent prepared by the preparation method comprise ester groups, phenyls and phosphate groups; the ester groups hydrolyze under the basic condition of the concrete; carboxyls with the water reducing function and hydroxy phenyl phosphate groups with the hydration heat reducing function are gradually released; the concrete hydration heat can be effectively reduced.
Owner:KZJ NEW MATERIALS GROUP CO LTD
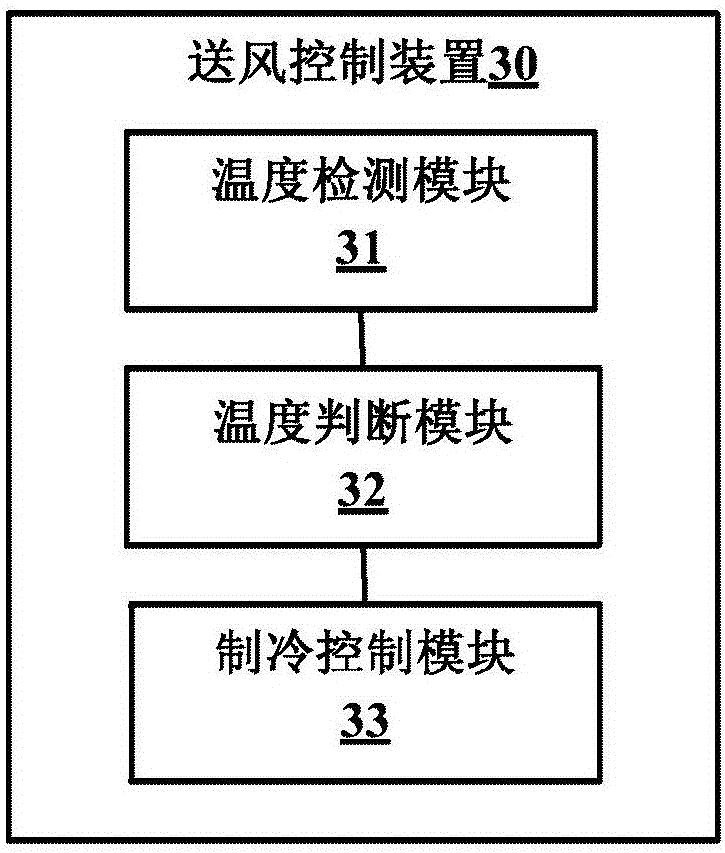
Air supply control method and device of air-cooled refrigerator
InactiveCN106595180AExtend cooling timeShorten cooling timeLighting and heating apparatusDomestic refrigeratorsRefrigerationAir door
The invention provides an air supply control method and device of an air-cooled refrigerator. The air supply control method of the air-cooled refrigerator comprises the steps that the actual temperature in a cold storage chamber and actual temperature in a freezing chamber are detected through a first temperature sensor and a second temperature sensor which are arranged in the cold storage chamber and the freezing chamber correspondingly; whether the actual temperature in the cold storage chamber is larger than or equal to the preset turn-on temperature is judged, and if yes, an air door is controlled to be opened at a preset angle, and a fan is controlled to operate at a first rotating speed; whether the actual temperature in the cold storage chamber is smaller than or equal to the first turn-off temperature is judged, the actual temperature in the freezing chamber is larger than the second shut-down temperature, and if yes, the air door is controlled to be closed and the fan is controlled to operate at a second rotating speed. According to the scheme, by shortening the negative pressure time of the cold storage chamber and reducing the negative pressure degree of the cold storage chamber, the condensation phenomenon in the cold storage chamber can be effectively relieved, and the problem that condensation affects the service life and the refrigeration effect of the refrigerator is avoided.
Owner:HAIER SMART HOME CO LTD
Phase change capsule suspension/floating heat management and cold starting system
ActiveCN106654318AReduce energy consumptionExtend cooling timeFuel cell heat exchangeSolid electrolyte fuel cellsEngineeringCooling fluid
The invention relates to a phase change capsule suspension / floating heat management and cold starting system. A suspension / floating phase change capsule and cooling liquid combined system is installed in a cooling liquid box, the density of suspension / floating phase change capsules is equivalent to that of the cooling liquid, so that the suspension / floating phase change capsules can be suspended in the cooling liquid, the density is decreased after the heat absorption, and the suspension / floating phase change capsules are floated at the surface of the cooling liquid; and the cooling liquid box is connected with a fuel cell stack to form a cooling liquid circulating loop. The suspension / floating phase change capsule and cooling liquid combined system exchanges the heat with the fuel cell stack, when a fuel cell works, the suspension / floating phase change capsules absorb a great number of heat, so that the cooling liquid circularly cools the fuel cell and stores the heat; and when the fuel cell stops working, the suspension / floating phase change capsules release the phase change latent heat to preserve the heat of the fuel cell stack, and the preheating time of the fuel cell when in cold starting can be shortened, so that a cooling and heat preservation effect for the fuel cell can be achieved, and the heat management and cold starting system of the fuel cell is optimized.
Owner:TSINGHUA UNIV
Device and method for detecting characteristics of pulse type semiconductor laser
InactiveCN101672889AExtend cooling timeImprove accuracyIndividual semiconductor device testingPhysical chemistryEngineering
The invention discloses a device and method for detecting characteristics of pulse type semiconductor laser, which relates to the technical fields of precise instruments and testing and solves the problem that the environment of a laser to be detected has vapor as the existing detector with the characteristics of a semiconductor laser has a large amount of heat accumulation. The current signal output by a current signal output end of a hardware control circuit in the detector of the invention is a pulse type current signal, and the step a of the pulse type current signal is 0.1 mA; the pulse width b of the pulse type current signal is larger than or equal to 100 ns, and is less than or equal to 20 mu s; the pulse period c of the pulse type current signal is larger than or equal to 200 ns,and is less than or equal to 20 ms; the pulse amplitude d of the pulse type current signal is larger than or equal to 0 mA, and is less than or equal to 500 mA. The detector also comprises a nitrogensource, and nitrogen is charged in the environment of the laser to be detected in the process of detection so as to test the laser to be detected in the nitrogen environment and further to avoid the generation of water vapor effectively.
Owner:HARBIN INST OF TECH
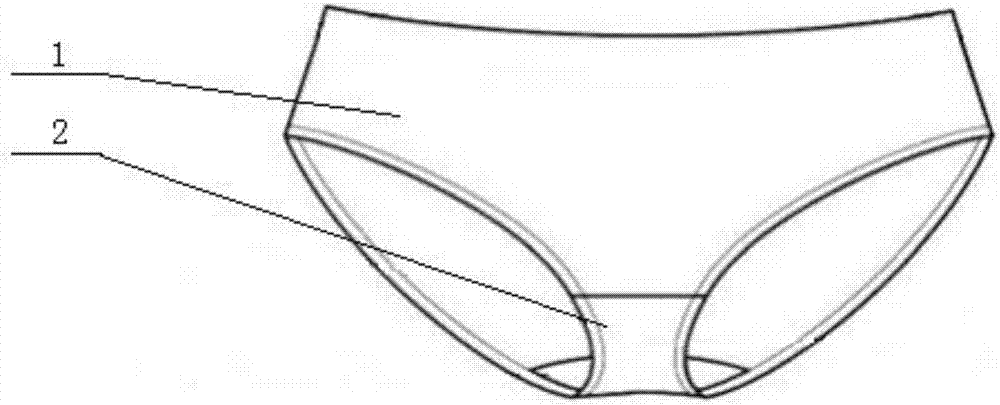
Cold therapy bag and cold therapy underpants containing same
InactiveCN106955189AReduce lossesRapid dissolution endothermic reactionTherapeutic coolingTherapeutic heatingMedicineSodium sulfate
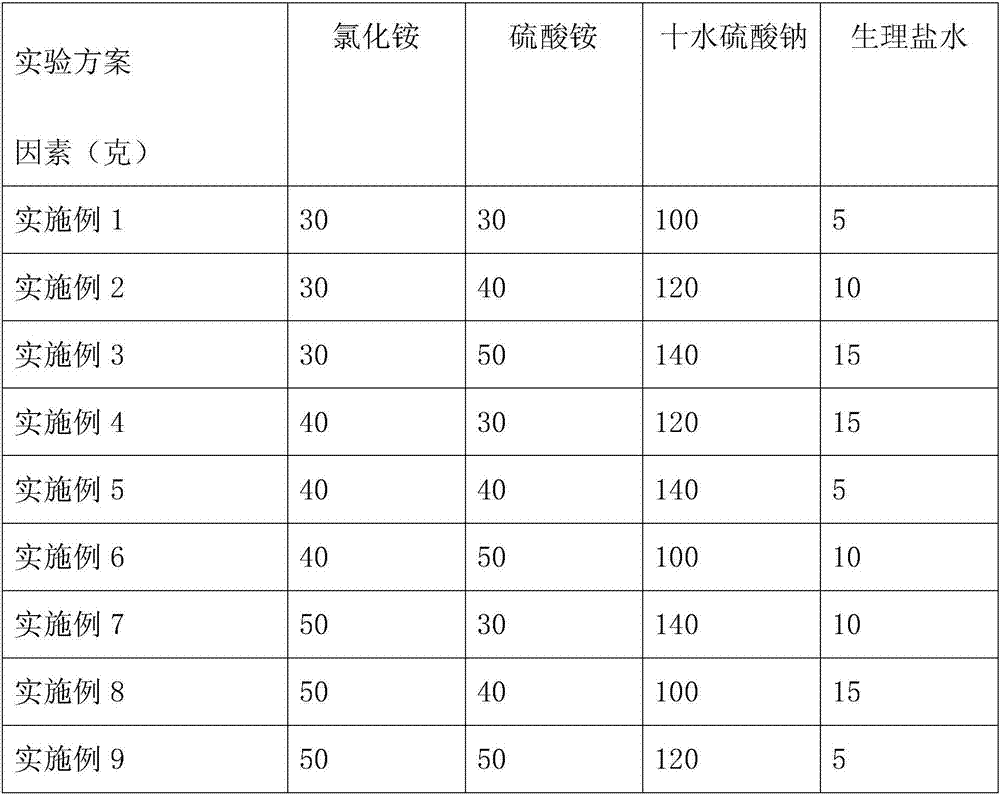
The invention relates to a cold therapy bag and cold therapy underpants containing the same. The cold therapy bag comprises, in parts by weight, 30-50 parts of ammonium chloride, 30-50 parts of ammonium sulfate, 100-140 parts of sodium sulfate decahydrate and 5-15 parts of normal saline, wherein ammonium chloride and ammonium sulfate are packed individually or multi-packed, and sodium sulfate decahydrate and normal saline are packed individually. The cold therapy bag can maintain the low temperature at about 10 DEG C for 2 hours or longer and is suitable for lasting cold therapy of burning or scalding. The cold therapy underpants containing the cold therapy bag are particularly suitable for nursing a special part such as the burnt perineum part.
Owner:常淑芳
Sustained-release freshener for disposable hygienic product, preparation method and application thereof
ActiveCN101785751AAvoid contactAvoid stimulationCosmetic preparationsToilet preparationsSocial benefitsPreservative
The invention relates to a combination of a sustained-release freshener used for disposable hygiene product, a preparation method and the application thereof. Raw materials of the combination are mainly are follows (proportion by weight of each component is shown in brackets): a freshening factor (10 to 30%), a sustained-release agent (30 to 50%), a preservative (0.1 to 1%), an inclusion agent (10 to 20%), an additive (1 to 25%) and a medical solvent (10 to 30%). With simple manufacturing process, cheap raw materials, good effect, strong adaptability and obvious economic as well as social benefits, the combination of the sustained-release freshener overcomes the stimulation to a human body due to high content of the freshening factor and improves the problem that the content utilizing rate of effective components of the common freshener is low.
Owner:TIANJIN SINOSH NEW MATERIAL TECH
Heat dissipation device using latent heat functional fluid and heat dissipation method thereof
ActiveCN102548361AReduce areaExtend cooling timeSemiconductor/solid-state device detailsSolid-state devicesStopped workConductive materials
The invention relates to a device for heat dissipation of an electronic apparatus with a discontinuous work heat source and a corresponding heat dissipation method. The device comprises a housing made of a heat conductive material; a cover plate made of a heat conductive material, connected with the housing and closed with the housing to from an inner chamber; a liquid injection tube for injecting a latent heat functional fluid; and a latent heat functional fluid filled in the inner chamber. The latent heat functional fluid stores the heat that is not discharged immediately inside when the discontinuous work heat source works and diffuses heat when the heat source stops working so as to realize non-synchronous operation of heat production and heat dissipation. Through the invention, partial storage and partial dissipation of heat are realized by phase change latent heat of the latent heat functional fluid. Heat stored is dissipated continuously when the heat source stops working, heat production and heat dissipation are carried out in a non-synchronous manner and the heat dissipation time is prolonged so as to obtain the technical effect of reducing the area of heat dissipation fins or even realizing fin free heat dissipation.
Owner:HUAZHONG UNIV OF SCI & TECH
Drug reagent storage device of moveable clinical diagnosis and treatment equipment
ActiveCN104192428ALight in massImprove cooling efficiencyShock-sensitive articlesPhysicsHeat conservation
The invention discloses a drug reagent storage device of moveable clinical diagnosis and treatment equipment. The drug reagent storage device comprises a cubic box and a cover which can be opened and closed and located at the upper end of the box. The box and the cover are respectively provided with a heat preservation and insulation layer, a heat-insulated storage chamber is arranged on the right side in the box, and a liquid nitrogen tank is stored in the storage chamber; a cooling pipe and a safety exhaust pipe are arranged at the upper end of the liquid nitrogen tank, the safety exhaust pipe is provided with a safety valve, and the cooling pipe is provided with a control valve used for controlling the discharge speed of nitrogen; a sliding block used for adjusting the cooling rate is arranged in the box. Liquid nitrogen serves as a cooling source, and the drug reagent storage device has the characteristics of small mass, high cooling efficiency and long refrigeration time and is convenient to move, carry and use; the refrigeration rate and the temperature in the box can be effectively controlled through adjustment of cooing pipe sets and the control valve, effective storage of drug reagents is guaranteed, and the drug reagent storage device has excellent carrying and using effects.
Owner:BEIJING BEIKEN HUAYE SCI & TECH DEV
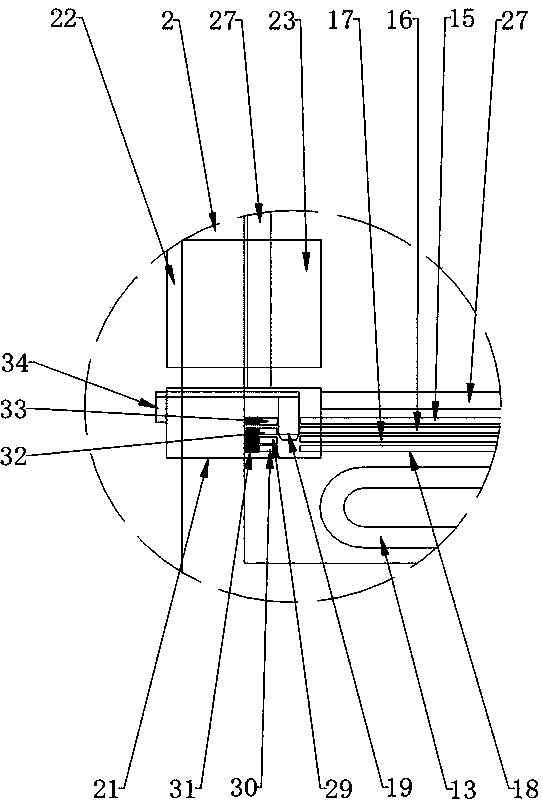
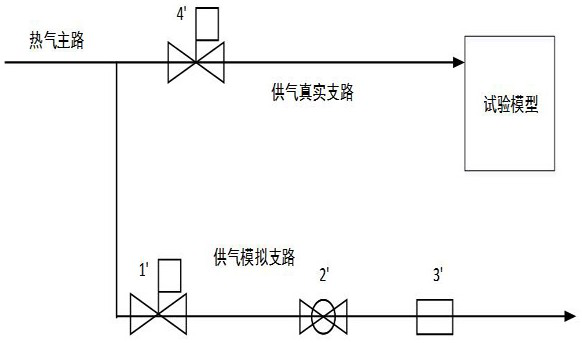
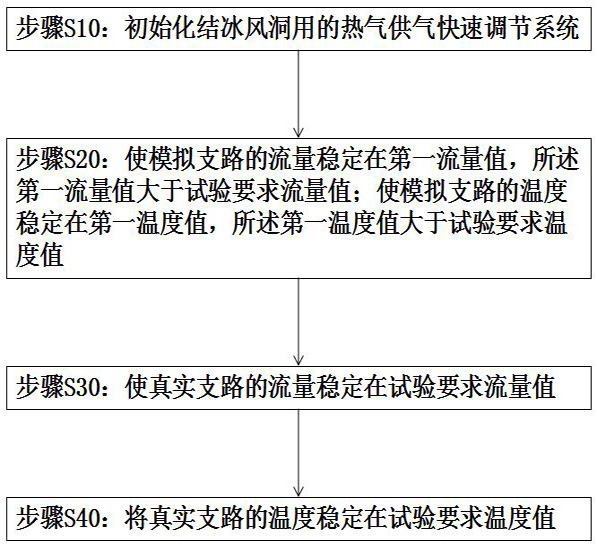
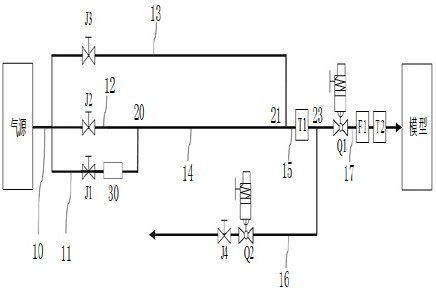
Hot air supply rapid adjusting system for icing wind tunnel and air supply method
ActiveCN112798213AArrive quicklyExtend cooling timeAerodynamic testingPipeline systemsTest efficiencyEngineering
The invention is suitable for the technical field of wind tunnel tests, and provides a hot air supply rapid adjusting system for an icing wind tunnel and an air supply method, and the air supply method of the hot air supply rapid adjusting system for the icing wind tunnel comprises the following steps: initializing the hot air supply rapid adjusting system for the icing wind tunnel; enabling the flow of the simulation branch to be stabilized at a first flow value, wherein the first flow value is greater than the flow value required by the test; enabling the temperature of the simulation branch to be stabilized at a first temperature value, wherein the first temperature value is greater than the temperature value required by the test; enabling the flow of a real branch to be stabilized at a flow value required by the test; and stabilizing the temperature of the real branch at the temperature value required by the test. Compared with the prior art, the temperature stabilization time of the inlet of the model is shortened, sudden temperature drop is prevented, large flow change is avoided, and the test efficiency is improved.
Owner:LOW SPEED AERODYNAMIC INST OF CHINESE AERODYNAMIC RES & DEV CENT
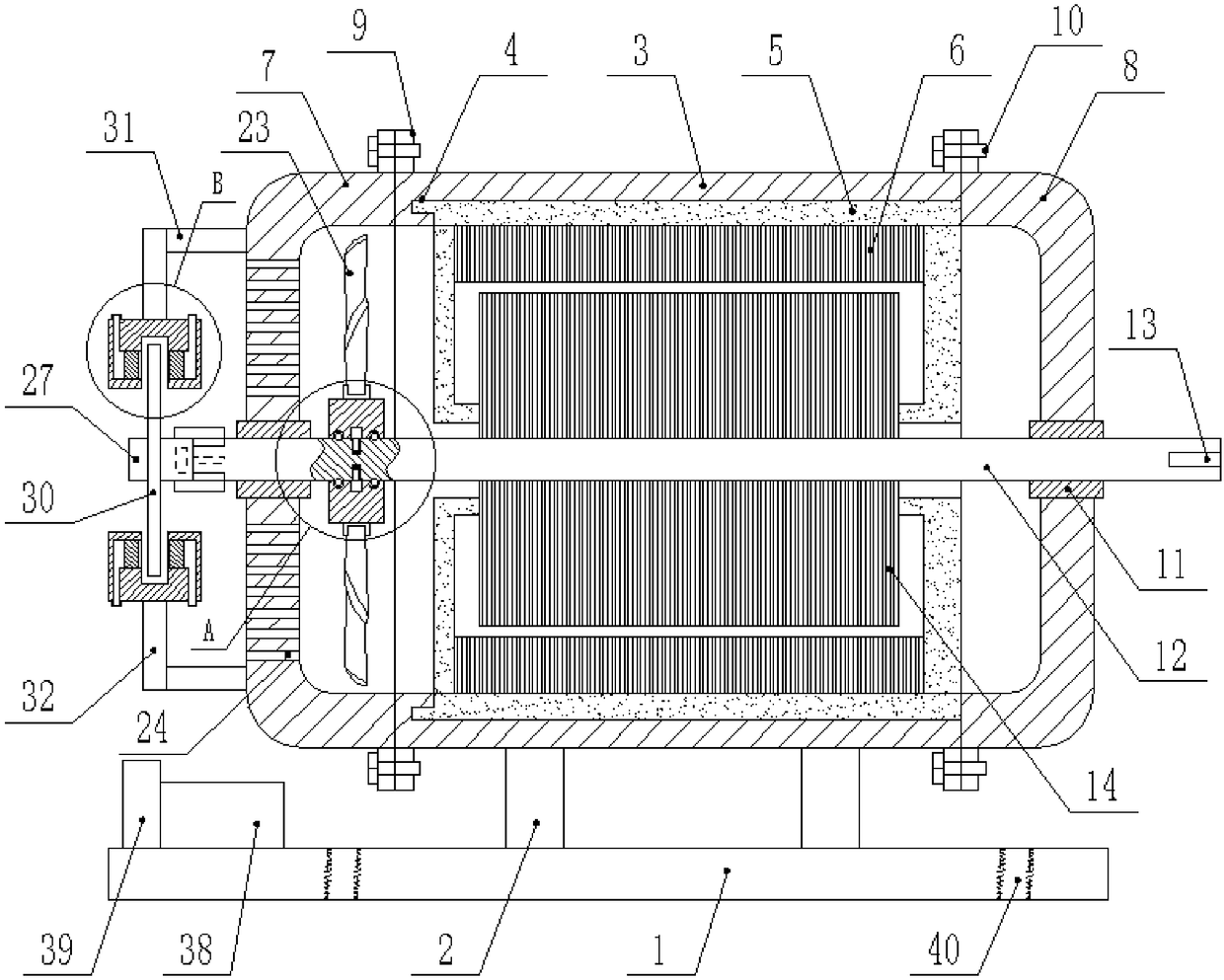
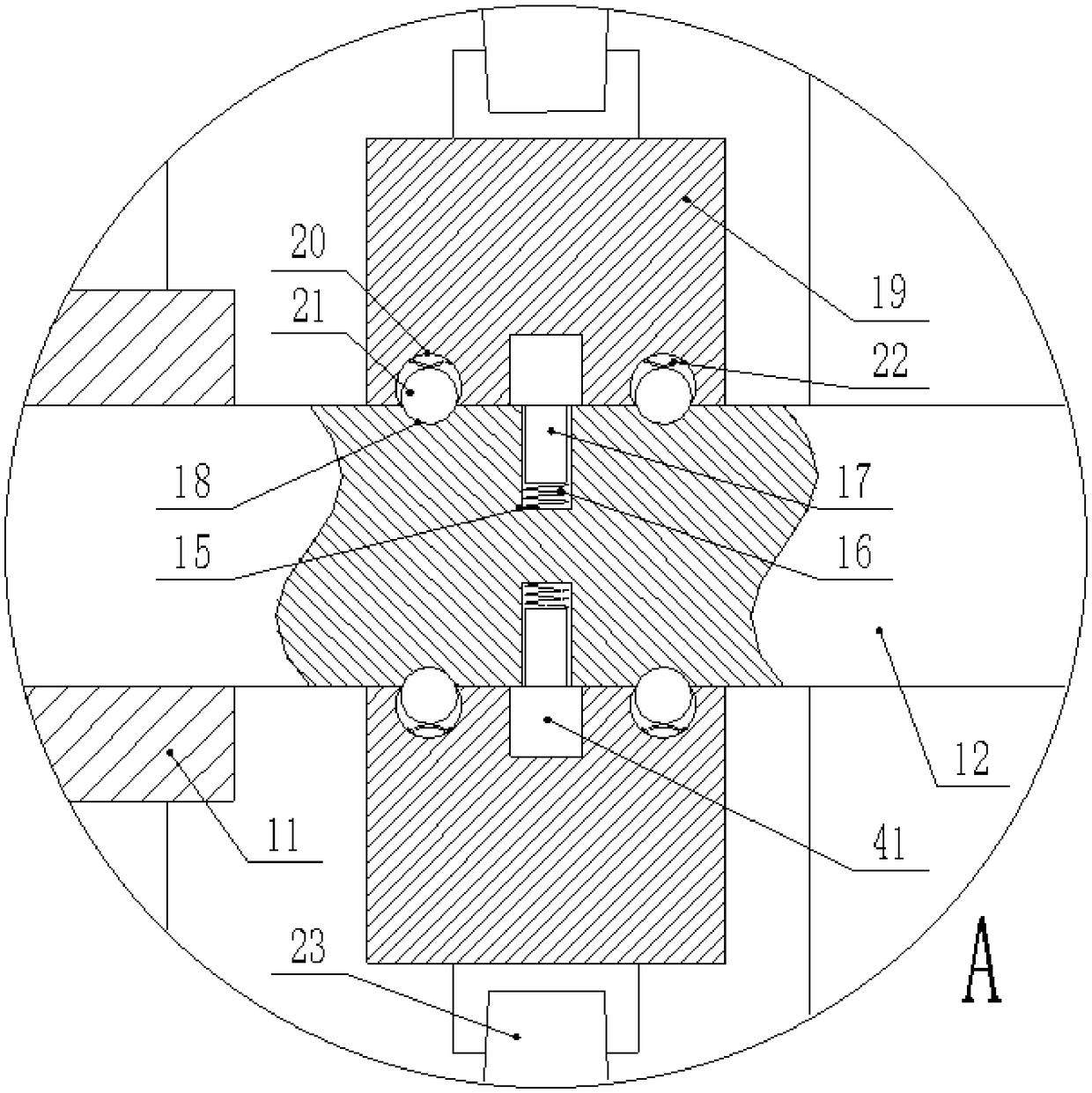
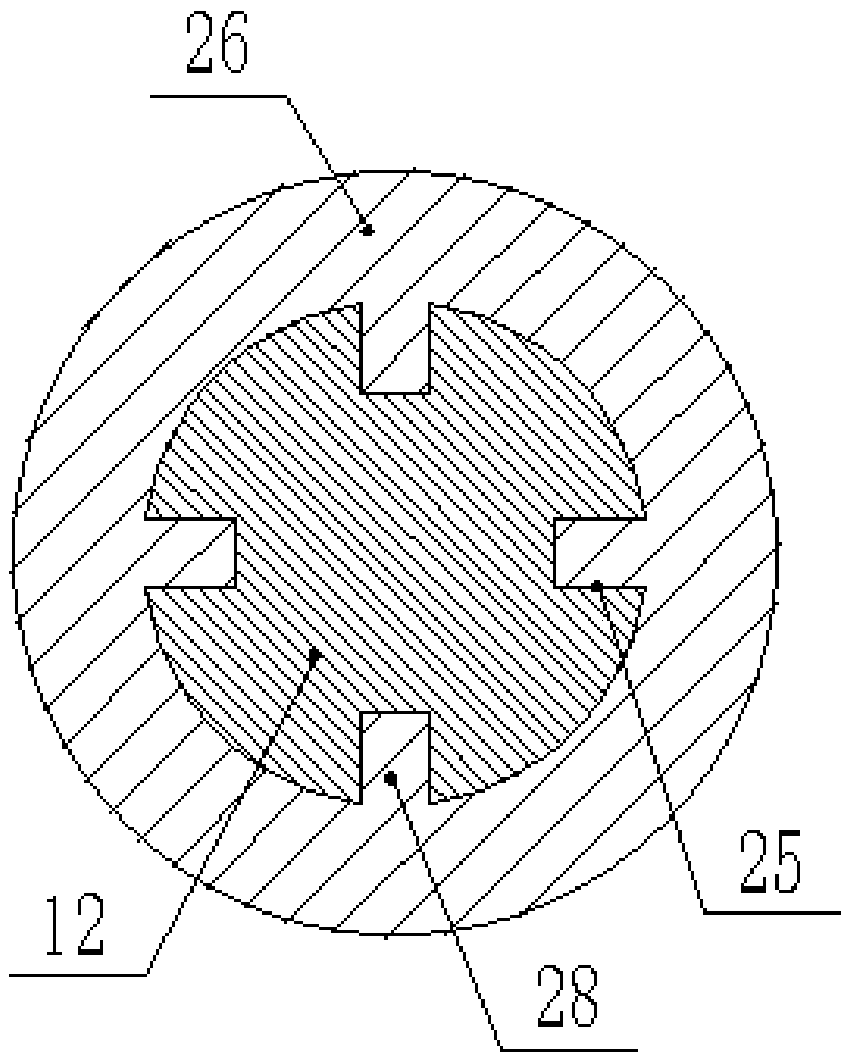
Novel motor convenient to disassemble and maintain
InactiveCN108808985AEasy to disassembleEasy to disassemble and repairMagnetic circuit rotating partsMechanical energy handlingArchitectural engineeringStator
The invention discloses a novel motor convenient to disassemble and maintain. The motor comprises a bottom plate, wherein a plurality of fixing rods are vertically fixed at the upper end of the bottomplate; a motor casing is fixed at the tops of the fixing rods; a fixing groove is formed in the inner wall of the motor casing; a fixing plate glidingly connected with the fixing groove is horizontally inserted into the fixing groove; a groove for clamping the fixing plate is formed in the inner side of the fixing groove which is matched with the fixing plate; multiple uniformly arranged stator cores are fixed on the inner wall of the fixing plate; a rear casing body and a front casing body are arranged at two ends of the motor casing respectively; multiple mutually corresponding fixing blocks are fixed on the outer walls of the motor casing, the rear casing body and the front casing body respectively; fixing bolts in threaded connection with the fixing blocks are inserted into the fixingblocks; a bearing fixedly connected with the rear casing body and the front casing body is inserted into centers of the rear casing body and the front casing body. The fixing plate is inserted into the fixing groove for fixation, the fixing plate can be dismounted and mounted easily by opening the front casing body, the stator cores are convenient to dismount and maintain by dismounting and mounting the fixing plate, and the motor is time-saving, labor-saving, efficient and fast.
Owner:张志通
Phase change cold storage agent for air conditioner and its producing method
InactiveCN1944566ASimple configurationShorten ice making timeHeat-exchange elementsHeat conductingPotassium
The present invention discloses one kind of phase change cold accumulating agent for air conditioner and its production process. The phase change cold accumulating agent consists of potassium chloride 1-4 wt%, nucleator 0.1-0.5 wt%, surfactant 0.05-0.5 wt%, titania grain as heat conducting enhancer 0.01-1 wt% and pure water for the rest, and has phase change temperature region of -1 deg.c to -8 deg.c. The phase change cold accumulating agent has the advantages of simple recipe, low cost, long cold releasing time, etc; and using the phase change cold accumulating agent in air conditioner can reach the aims of 'removing peak to fill the valley', raising efficiency and saving energy.
Owner:DONGGUAN GUANGDA REFRIGERATION
Novel quick-cooling chemical ice bag and preparation method thereof
ActiveCN105038721AExtended cooling timeExtended service lifeHeat-exchange elementsAmmonium chloride mixturePVA - Polyvinyl alcohol
The invention discloses a novel quick-cooling chemical ice bag and a preparation method of the novel quick-cooling chemical ice bag. The novel quick-cooling chemical ice bag is prepared from the following components in parts by weight: 20 to 30 parts of ammonium chloride, 50 to 60 parts of barium hydroxide, 30 to 40 parts of ammonium nitrate, 2 to 6 parts of polyvinyl alcohol, 10 to 20 parts of ferric chloride and 15 to 20 parts of sodium chloride. The preparation method of the novel quick-cooling chemical ice bag comprises the steps of accurately weighing according to the weight ratio of each component, adding the weighed polyvinyl alcohol into the barium hydroxide and stirring, wherein the stirring speed is 150 to 250 r / min, and the temperature can be heated up to be 70 to 80 DEG C; stopping heating after the solution becomes transparent, enabling the solution to be naturally cooled to room temperature, and obtaining solution A; sequentially adding the ammonium chloride, the ammonium nitrate and the ferric chloride in the solution A under a stirring state, and obtaining solution B by uniformly stirring the mixed solution; adding the sodium chloride in the solution B, and sealing and packaging the obtained solution in a bag when the mixed solution is uniformly stirred and becomes transparent. According to the novel quick-cooling chemical ice bag disclosed by the invention, the refrigerating speed is fast, and the refrigerating temperature is low.
Owner:江苏爱舍伦医疗科技集团股份有限公司
Method for reducing hot stack influences of metal SLM three-dimensional printing
InactiveCN105537590AMinimize the effect of heat accumulationExtend cooling timeAdditive manufacturing apparatusIncreasing energy efficiencyEngineeringThree dimensional data
A method for reducing hot stack influences of metal SLM three-dimensional printing specifically includes the following steps of firstly, dividing a three-dimensional data model into sheets with certain thicknesses, fine dividing each sheet into scanning lines at certain intervals, and completing before-processing data treatment; secondly, numbering the scanning lines at intervals in the sequence of a scanning route; thirdly, focusing a laser beam to fuse metal powder in a processing area spot by spot according to the scanning route under the control of a printing host to form metal sheet layers; fourthly, completing scanning of each metal sheet layer according to the scanning route to gradually form a three-dimensional metal object and finally complete three-dimensional printing. Printing time of a plurality of scanning lines exists between each scanning line and the previous scanning line in the printing process, cooling time is greatly prolonged, the hot stack effect is avoided or reduced, and processing quality stability in the whole scanning area is ensured; the printing process is still continuously conducted, and the overall printing time can not be prolonged.
Owner:合肥中加激光技术有限公司
Process method for reducing noise of film capacitor
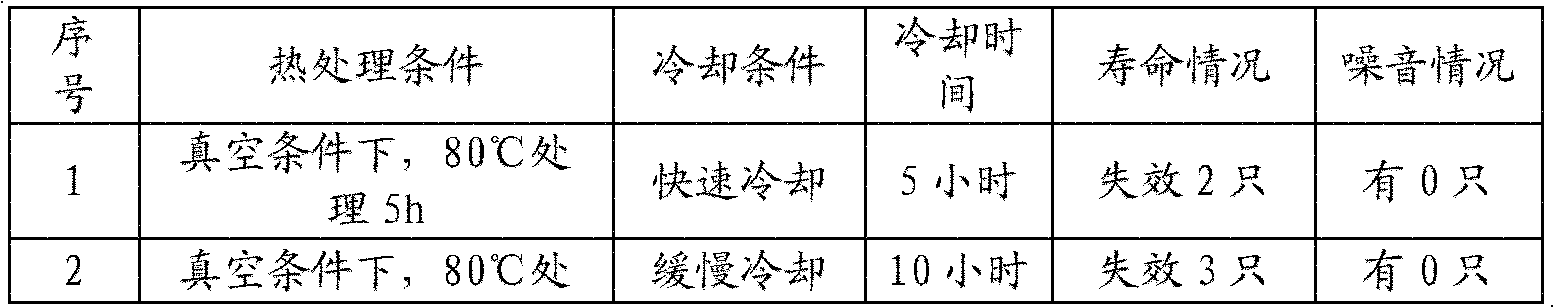
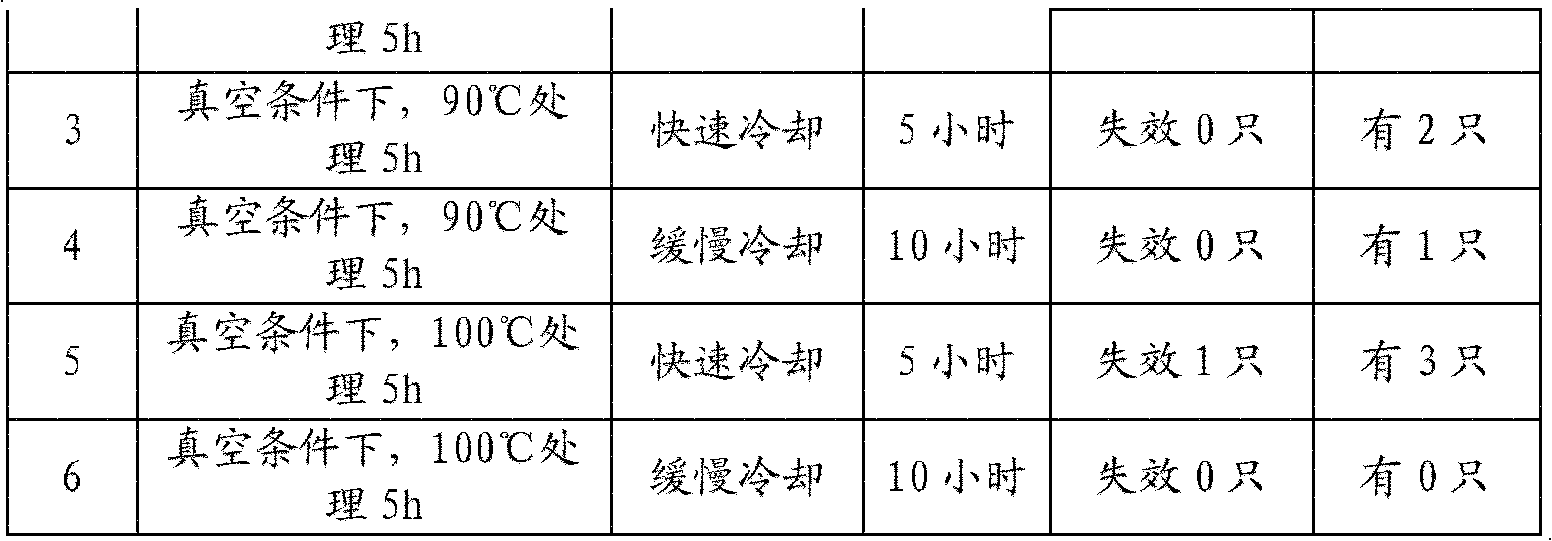
ActiveCN102129910AReduce gapExtend cooling timeThin/thick film capacitorStacked capacitorsProcess engineeringHeat setting
The invention relates to a heat treatment method for reducing the noise of a film capacitor, in particular to the heat treatment method for a film capacitor core. The heat treatment method comprises the following process steps of: firstly, carrying out heat setting treatment on the wound film capacitor core in a heat setting tank at the constant treatment temperature of 100DEG C for 6 hours undera vacuum condition; secondly, keeping the film capacitor core subjected to heat setting treatment in the heat setting tank for 2 hours without heating under a non-vacuum condition; thirdly, transferring the film capacitor core from the heat setting tank to a heat-radiating chamber, controlling the temperature of the heat-radiating chamber to be 30-40DEG C, keeping the relative humidity of the heat-radiating chamber below 60 percent and slowly dissipating heat for 10-12 hours; and fourthly, transferring the film capacitor core from the heat-radiating chamber to an assembly workshop, controlling the temperature of the assembly workshop to be 25-30DEG C and keeping the temperature for over 2 hours and then starting entering a next assembly work procedure.
Owner:ZHUHAI GREE XINYUAN ELECTRONICS
Pressure-controlling method of sterilizing kettle in sterilization cooling stage
The invention discloses a sterilizing method by adopting a sterilizing kettle, particularly to pressure-controlling method of a sterilizing kettle in sterilization cooling stage, which comprises the following steps of: introducing high-pressure air into the sterilizing tank until the pressure therein reaches the initial value; introducing cooling water into the sterilizing tank until the water level therein reaches the set value; introducing compressed air into the sterilizing tank continuously, opening a circulation pump at intervals according to the pressure in the sterilizing tank, wherein when the pressure in the sterilizing tank is greater than the set value, the circulation pump is opened, and when the pressure in the sterilizing tank is less than the set value, the circulation pump is stopped; when the cooling process is completed, emptying the cooling water, and reducing the sterilizing pressure in the tank to atmospheric pressure. By continuously air-intaking and opening, the pressure in the sterilizing tank can be continuously compensated; the circulation pump is opened at intervals according to the pressure in the sterilizing tank, the cooling operation can be conducted in the premise of ensuring the pressure in the tank, therefore great pressure drop occuring at the moment when the pump is opened for intruductino fo the cooling water and in the temperature dropping process can be effectively prevented. The method can be suitable for the sterilization operations of cans, flexible packages and other types of packaged food.
Owner:CHENGDU XIWANG FOOD
Device and control method for preparing wind energy, ocean energy and solar energy
InactiveCN102635510AStrong complementarityReduce instabilityWind energy with electric storageEnergy storageInstabilityFlywheel
The invention discloses a device and a control method for preparing wind energy, ocean energy and solar energy, wherein the device comprises a fan, an impellor, a solar battery pack, a storage battery, a commercial power interface, an air tank, an air compressor, a sensor, a transmission and a controller, the corresponding transmission gears are selected according to the real-time pressure of the air tank, so that a drive electromotor can operate within the rotation speed range with the highest efficiency so as to reduce the number of air compression stages required without the transmission; the instability of energy supply of wind energy, ocean energy and solar energy can be eliminated by adopting commercial power supplement measures, and a flywheel is arranged between an electromotor and the air compressor, thus the rotation speeds of the electromotor and the air compressor can be more stable; and meanwhile, direct-current electricity energy is provided for the drive electromotor of each stage of air compressors by utilizing the wind energy, ocean energy and solar energy, the complementation of multiple kinds of renewable energy sources is realized, the instability of single renewable energy source is relieved, and greater available power can be obtained.
Owner:JIANGSU UNIV
Cold storage type water bed mattress having water bed effect
InactiveCN102973047AOvercome the problem of not being soft and being hardImprove insulation effectFluid mattressesBedsRefrigeration compressorIce water
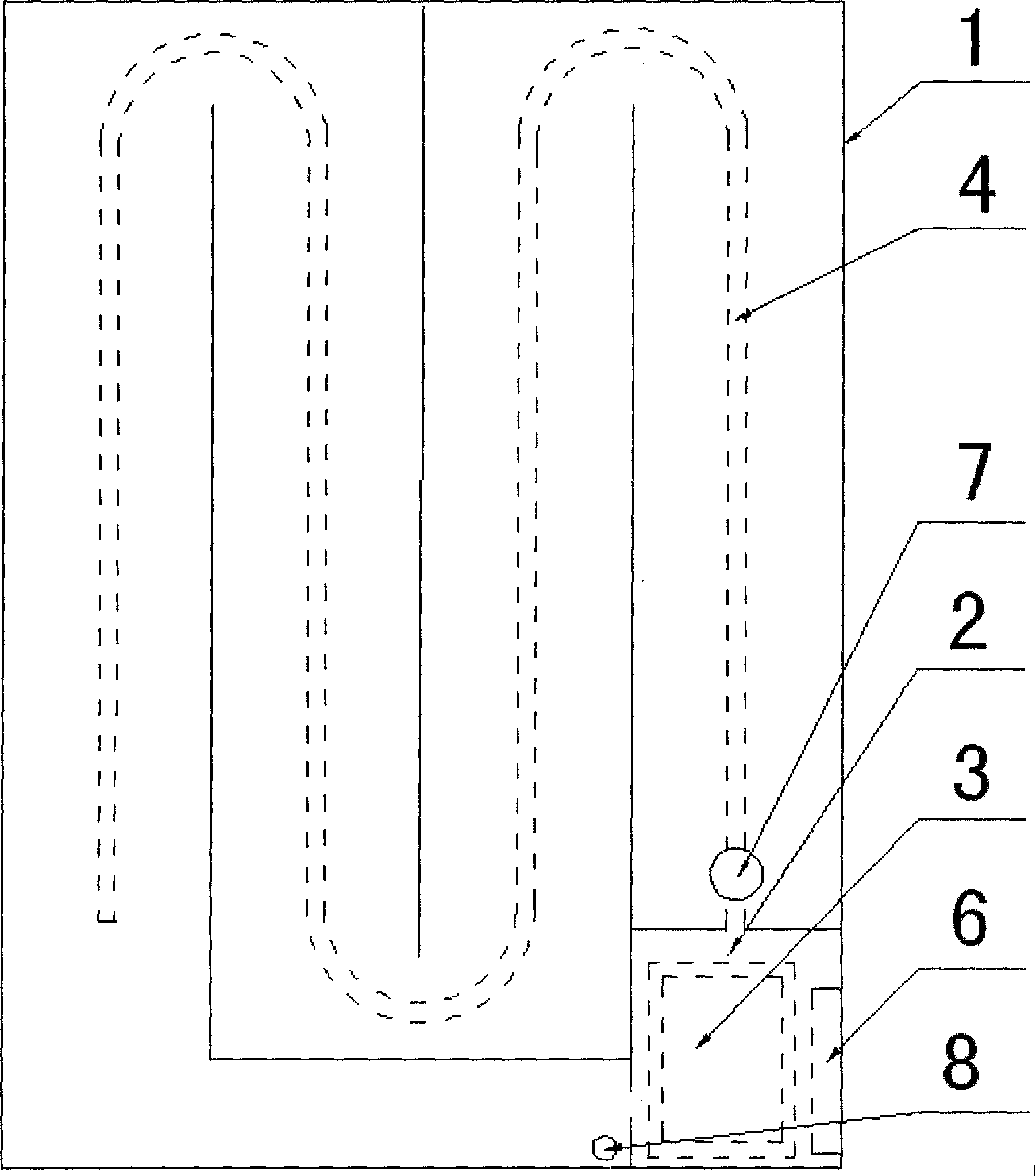
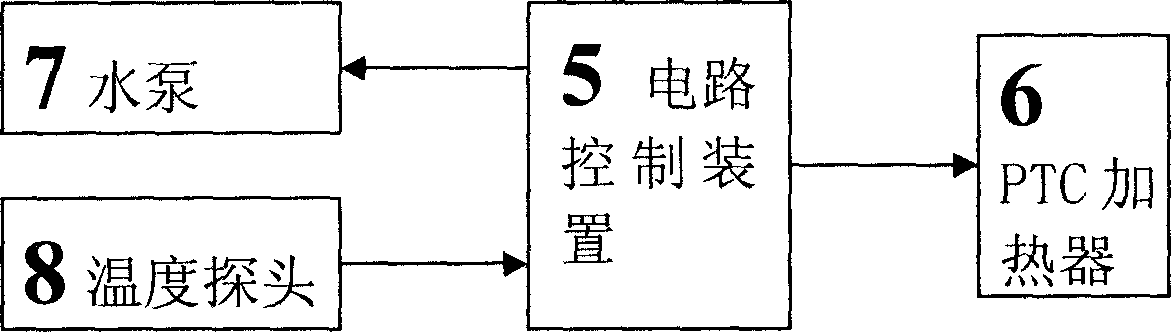
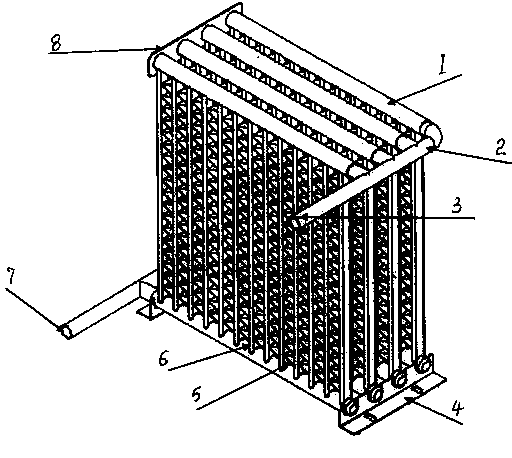
The invention relates to a cold storage type water bed mattress having a water bed effect. The cold storage type water bed mattress comprises a water bed mattress, a temperature adjustment bag, an ice bag, a U-shaped water pipe, a PTC heater and the like, wherein the internal of the water bed mattress is divided into a plurality of parallel closed small water pads, the head and the tail of the small water pads are communicated, the small water pads are filled with water, the U-shaped water pipe and a water pump are connected in series, the U-shaped water pipe, the water pump and the temperature adjustment bag form a water circulation temperature adjustment system, an empty chamber is formed between the inner layer and the outer layer of the temperature adjustment bag, when normal temperature water passes through the empty chamber, cold generated by the ice bag filled in the inner layer or heat generated by the PTC heater is absorbed to form ice water or hot water, and the ice water or the hot water is rapidly and uniformly sprinkle the whole water bed mattress through small holes arranged between the U-shaped water pipe at intervals so as to achieve a purpose of cooling (heating). According to the present invention, an effect that the water bed adopts buoyancy force to achieve a human body physiological curve meeting effect is provided, characteristics of small size, light weight, consumption saving and good thermal insulation are provided, and the cold storage type semiconductor cooling device or refrigeration compressor method is adopted to achieve a cooling effect so as to provide advantages of simple structure, low cost, easy implement, and easy popularization and application.
Owner:顾晓烨
Multilayer micro-channel condenser
InactiveCN103743164AExtend cooling timeImprove cooling efficiencyEvaporators/condensersEngineeringElectrical and Electronics engineering
The invention discloses a multilayer micro-channel condenser which aims at increasing the heat dissipation area and reducing the weight of the condenser. According to the technical scheme of the multilayer micro-channel condenser, every two groove type holes are separated through a blocking block; pipe openings which are formed at two ends of micro-channel pipes are provided with unit collecting pipes which are in correspondingly welding connection with groove type openings which are formed in the unit collecting pipes; aluminum fins are welded between every two micro-channel pipes to form a heat dissipation unit; abutting openings which are machined on a header collecting pipe are connected with the plurality of heat dissipation units to form the condenser after welding; a reinforcing plate is welded at the sealed end of the upper portion unit collecting pipes of the condenser. Compared with condensers with the same size, the multilayer micro-channel condenser is high in heat dissipation efficiency and the heat dissipation efficiency is improved by 2 to 3 times.
Owner:HENAN KELONG GRP
Lithium ion battery
ActiveCN103904354AShort circuit heat is smallSlow down heat productionLarge-sized flat cells/batteriesWound/folded electrode electrodesProduction rateElectricity
The invention provides a lithium ion battery, which comprises a shell, a cover plate, an electrolyte and a pole piece. A first lug and a second lug are led out from the pole piece; the pole piece is connected with a short circuit protection component, and the short circuit protection component has a three-layer structure and comprise a conductive layer in the middle and insulating layers on both sides of the conductive layer; one end of the conductive layer is electrically connected with the first lug of the pole piece, and the other end is a free end; and the outermost ring current collector is the one where the second lug is in. According to the lithium ion battery provided by the invention, the short circuit protection component arranged on the pole piece in the battery can initiatively convert complex short circuit inside the battery into large-area short circuit between positive / negative current collectors when the battery generates abnormal deformation under external force, so as to reduce internal short circuit heat in the battery to the minimum and shorten the heat conduction path; therefore, heat production rate of the battery lowers down, heat dissipation time of the battery is increased, and safety and reliability of the battery in harsh usage conditions are enhanced.
Owner:BYD CO LTD
Cold accumulation bed and magnetic refrigeration system
PendingCN110345680ARealize magnetizationAchieve demagnetizationDomestic cooling apparatusMachines using electric/magnetic effectsHeating timeRelative motion
The invention provides a cold accumulation bed and a magnetic refrigeration system. The cold accumulation bed comprises a first chamber and a second chamber, wherein the first chamber comprises a first region and a second region, the second chamber comprises a third region and a fourth region, a first magnetic working medium and a fourth magnetic working medium both made of positive effect magneto-thermal materials are arranged in the first region and the fourth region respectively, and second magnetic working medium and third magnetic working medium both made of negative effect magneto-thermal materials are arranged in the second region and the third region respectively; and the cold accumulation bed is further provided with a first magnetic generating device, wherein the cold accumulation bed and the first magnetic generating device can move relative to each other, when being opposite to the first region and the third region, the first magnetic generating device is not opposite to the second region and the fourth region, and when being opposite to the second region and the fourth region, the first magnetic generating device is not opposite to the first region and the third region. According to the cold accumulation bed and the magnetic refrigeration system, magnetizing and demagnetizing effects on the magnetic working media can be realized at the same time, the refrigeratingtime or the heating time of the system is prolonged, and the problem that the refrigerating time of a flow path of the single cold accumulation bed is short is solved.
Owner:GREE ELECTRIC APPLIANCES INC
Multifunctional temperature self-regulating film and face mask and eye mask manufactured therefrom
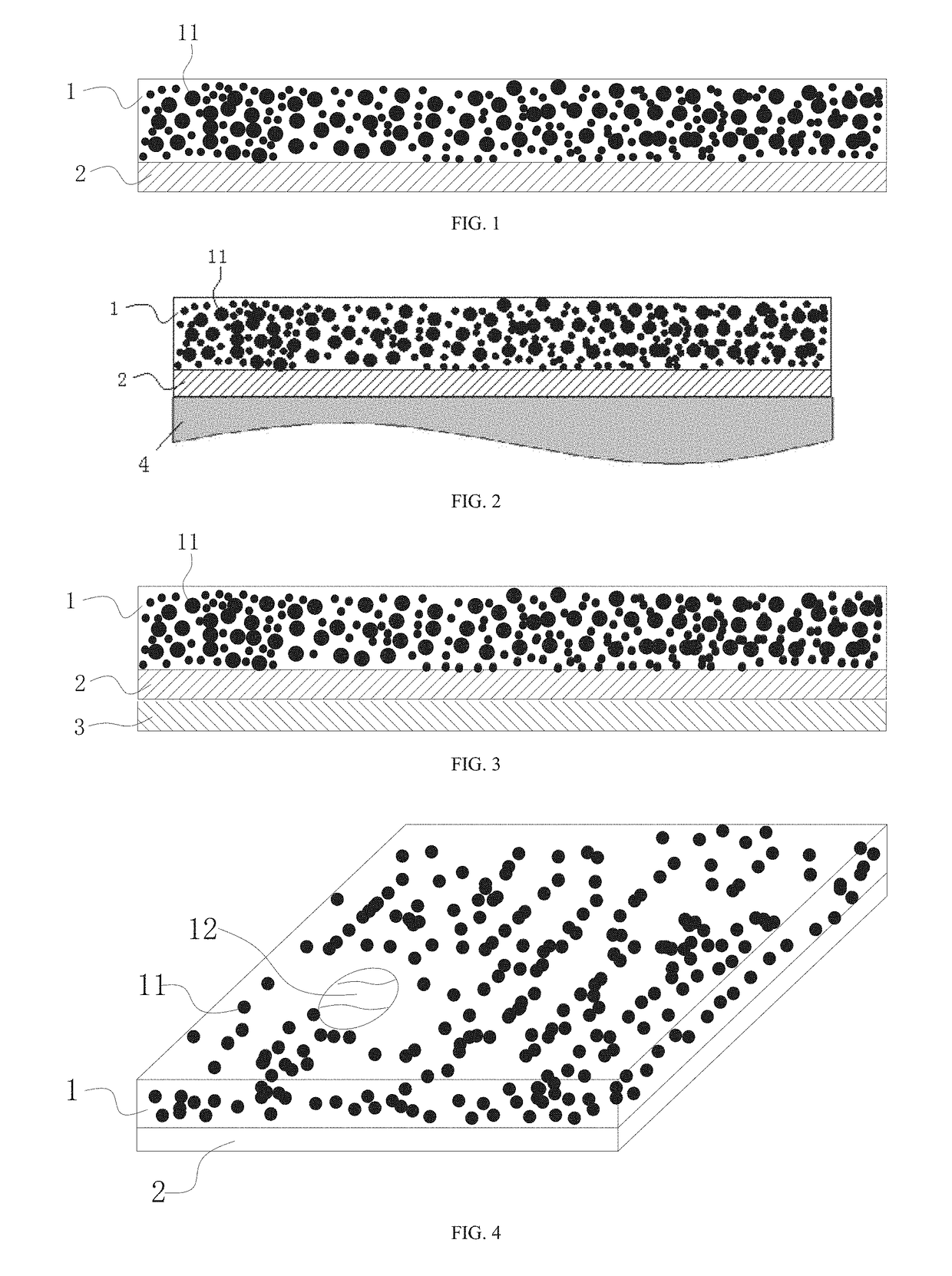
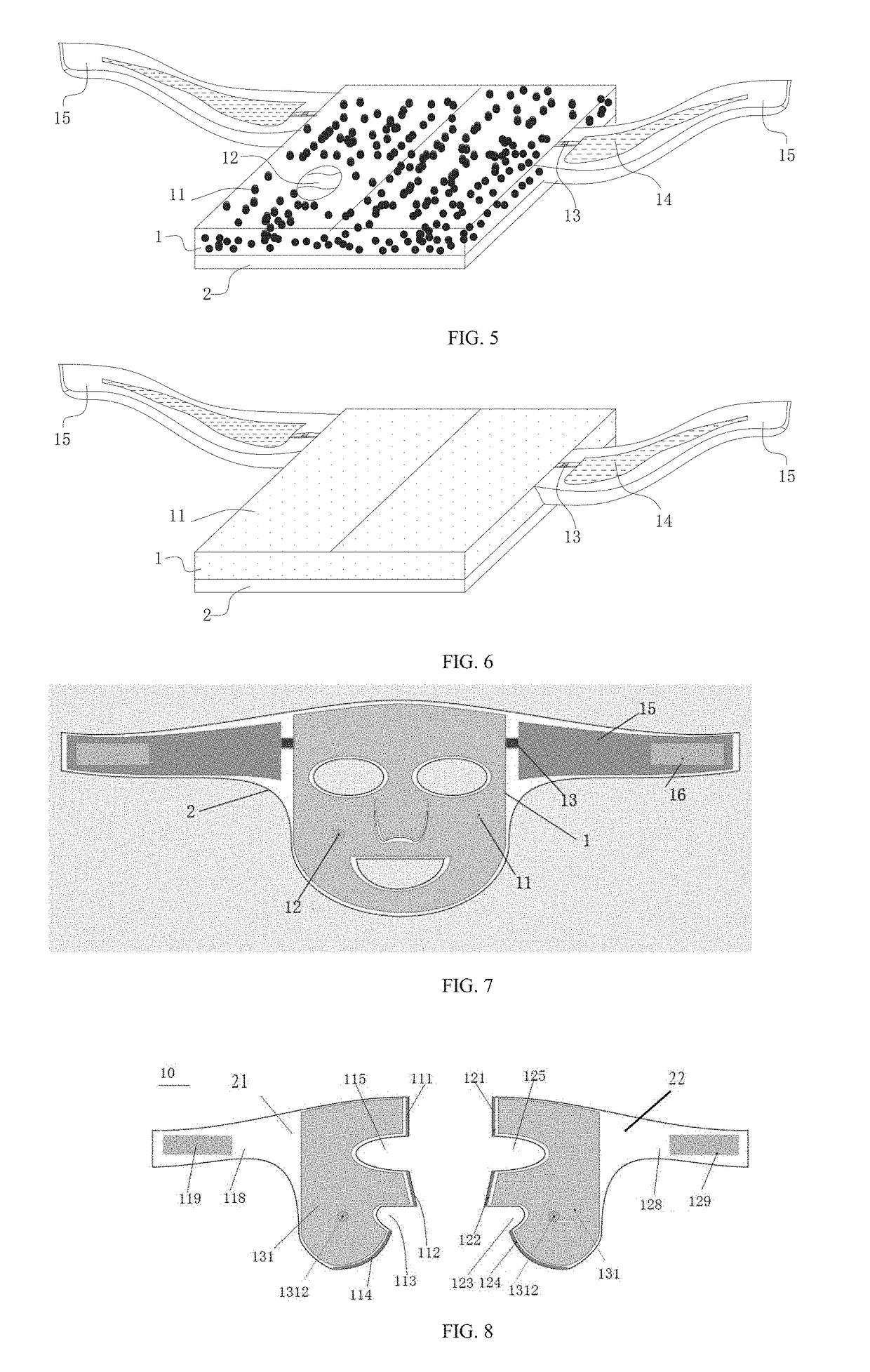
InactiveUS20170231884A1Reduce peak heating temperature temperatureReduce cooling temperatureCosmetic preparationsToilet preparationsFacial skinThermal insulation
The present invention discloses a multifunctional temperature self-regulating multilayer film, from outside to inside comprising at least: a carrier layer (1) loaded with functional material, and a thermal-insulation layer (2) directly contacted with the skin (4); wherein the carrier layer (1) loaded with functional material has a sealed cavity sealed with a flexible water-impermeable material, the sealed cavity is at least loaded with a refrigerating or heat-generating chemical material (11); wherein thermal-insulation layer (2) is made of a material having waterproof and thermal-insulation properties. The present multilayer film can be manufactured into a face mask or eye mask for use on facial skin.
Owner:HANDY TECH ZHUHAI LTD
High-power LED heat accumulating type solid-liquid phase change heat dissipation device
InactiveCN104344289AReduce dependenceExtend cooling timePoint-like light sourceLighting heating/cooling arrangementsLed packagingHeat sink
The invention discloses a high-power LED heat accumulating type solid-liquid phase change heat dissipation device which comprises a heat sink cavity, wherein the bottom of the heat sink cavity is used for being connected with an LED packaging part; a plurality of heat radiating fins are arranged on the outer side wall of the heat sink cavity; a plurality of metal pin fin structures are connected to the inner surface of the heat sink cavity in a direction perpendicular to heat flow; in addition, the part between every two adjacent metal pin fin structures is filled with a phase change material of which a melting point is lower than normal operation temperature of the LED packaging part. According to the high-power LED heat accumulating type solid-liquid phase change heat dissipation device disclosed by the invention, passive cooling is realized by using the characteristics that the phase change material absorbs heat during melting and the temperature is kept unchanged in the phase change process; besides, an effective heat conduction coefficient of the phase change material can be improved and natural convection of the phase change material is intensified, and further response rate of phase change latent heat storage is improved; the filling volume of the phase change material can be adjusted according to the power size and the working time, and normal work of the LED is guaranteed.
Owner:GUANGDONG XIAN JIAOTONG UNIV ACADEMY
3D-printing metal powder material preparation method capable of reducing hollow powder
InactiveCN106735268AReduce oxygen contentRaise the outlet temperatureAdditive manufacturing apparatusMaterials preparationNitrogen
The invention discloses a 3D-printing metal powder material preparation method capable of reducing hollow powder. The 3D-printing metal powder material preparation method includes A, melting stainless steel or aluminum alloy under protection of micro-positive pressure nitrogen into stainless steel or aluminum alloy liquid; B, taking nitrogen as an atomizing gas, controlling the atomizing pressure as 1.5-2MPa when the aluminum alloy serves as a raw material, controlling the atomizing pressure as 3-4MPa when the stainless steel serves as the raw material, and atomizing the melted stainless steel or aluminum alloy liquid to crush and cool the liquid into spherical or near-spherical stainless steel or aluminum alloy particles; C, collecting the spherical or near-spherical particles obtained in the step B, and treating the spherical or near-spherical particles to obtain 3D-printing metal powder. By the 3D-printing metal powder material preparation method, quantity of the hollow powder is reduced greatly, and 3D printing quality and mechanical property are improved.
Owner:NANTONG JINYUAN INTELLIGENT TECH CO LTD
Micro-channel side air-out condenser and mounting method thereof
InactiveCN106066102APracticalIncrease the scope of applicationEvaporators/condensersHeat exchanger casingsElectrical and Electronics engineeringHeat transfer
The invention discloses a micro-channel side air-out condenser and a mounting method thereof. The condenser comprises a first collecting main, a second collecting main, a flat pipe, separation plates, end covers, side plates and fins, wherein pipe joints are arranged on the first collecting main; inlet / outlet pipes are connected with the pipe joints; mounting brackets are arranged on both the first collecting main and the second collecting main; each of the mounting brackets comprises a fixed part and a connection part; the fixed parts and the connection parts are integrated; mounting holes are formed in the fixed parts; and the connection parts are arranged on the first collecting main. The mounting method of the condenser comprises the following steps: (a) part preparation; (b) part integration; (c) preliminary splicing; (d) end sealing; and (e) airtightness detection. The micro-channel side air-out condenser is compact in structure, flexible in connection, simple to mount, stable in performance, good in airtightness, large in heat transfer area so as to enlarge the heat exchange area of the air side, high in heat exchange efficiency, energy-saving, cost-reducing and wide in applicability.
Owner:浙江富源制冷设备股份有限公司
High-efficiency refrigerating system and refrigerating method thereof
ActiveCN107327993AMake full use of cooling capacityExtend cooling timeSpace heating and ventilation safety systemsLighting and heating apparatusCooling towerFully developed
The invention relates to a high-efficiency refrigerating system and a refrigerating method thereof. The high-efficiency refrigerating system comprises a first free cooling tower circulating device, tail end equipment and a second free cooling tower circulating device, wherein the first free cooling tower circulating device is used for natural cooling and refrigerating, and the tail end equipment is refrigerated. A heat exchanger is arranged between the first free cooling tower circulating device and the tail end equipment, a water cooling main engine is arranged between the second free cooling tower circulating device and the tail end equipment, and a first water outlet pipe is connected with the heat exchanger; a second water outlet pipe is connected with the water cooling main engine, a first water freezing circulating pump and a second water freezing circulating pump are arranged on the first water outlet pipe and the second water outlet pipe correspondingly, and a branching pipe is arranged between the first water outlet pipe and the second water outlet pipe in a communicating mode; and a first regulating valve is arranged on the branching pipe, and a second regulating valve is further arranged on the second water outlet pipe. According to the high-efficiency refrigerating system and the refrigerating method thereof, the cooling capacity of a cooling tower is fully developed, the cooling efficiency of a whole air-conditioning system is improved, and the operating energy consumption of the air-conditioning system is saved.
Owner:SHENZHEN DAS INTELLITECH CO LTD
Environmental-protection molding sand cooling recovery device
InactiveCN108436030AGuaranteed homogeneityReduce the temperatureMould handling/dressing devicesCooling effectEngineering
The invention discloses an environmental-protection molding sand cooling recovery device. The device comprises a tower body, and a first separation plate, a second separation plate and a blanking collecting plate arranged in the tower body in sequence from top to bottom; a crushing bin is fixedly connected with a fixed inclined surface end, a slide inclined surface end and a first driving cam in sequence from left to right; a first return spring is arranged between the slide inclined surface end and the first separation plate; the first driving cam is matched with the right side surface of theslide inclined surface end, and can butt against the slide inclined surface end to move to the left side to realize pressing of the first return spring; when the first return spring is reset, the slide inclined surface end can be driven to move to the right side; a discharge port is formed in the lower part of the blanking collecting plate; and the tower body is provided with a cooling intake endon the left side wall and an air collecting device on the right side wall. The device cools molding sand through the cooling intake end and the air collecting device, achieves better cooling effect,and is compact in structure, convenient to use, energy-saving, environment-friendly and suitable for wide application in cooling recovery of the molding sand.
Owner:李同贵
High-speed welding method of thin metal sheet
InactiveCN108326419APlay a fixed roleExtend cooling timeLaser beam welding apparatusThin metalThermal deformation
The invention relates to the field of welding, and provides a high-speed welding method of a thin metal sheet. The method comprises the steps of S1, dividing a welding track into a plurality of secondary welding sections; S2, arranging a plurality of welding points in each secondary welding section in advance; and sequentially marking the plurality of welding points; and S3, welding the first welding point in each welding section; then welding the second welding point in each welding section; sequentially welding in the same way until the N welding point in each secondary welding section is welded, and thus finishing sequential connection of the plurality of secondary welding sections and obtaining a complete welding line. According to the method, the thin metal plate can be fixed andprevented from welding deformation through the plurality of welding track sections; the alternative welding manner is performed, so that the heat dissipating time of the welding points can be increased, and the accumulated heat is decreased, and as a result, the weld penetration caused by thermal deformation and heat accumulation can be prevented; and meanwhile, low secondary section welding speedrealizes high overall welding speed.
Owner:HANS LASER TECH IND GRP CO LTD
Refrigerating device, refrigerating box and refrigerating system
InactiveCN107514850AAchieve refrigerationExtend cooling timeMechanical apparatusDomestic refrigeratorsEngineeringRefrigerant
The invention provides a refrigerating device, a refrigerating box and a refrigerating system. The refrigerating device comprises a container and a cold storage part. The container is used for containing a secondary refrigerant, and a liquid inlet and a liquid outlet are formed in the container. The secondary refrigerant can flow into the container from the liquid inlet and can flow out of the container from the liquid outlet. The cold storage part is arranged in the container and contains a cold storage agent. The cold storage part is used for making contact with the secondary refrigerant so as to achieve heat exchange. By means of the refrigerating device, the refrigerating box and the refrigerating system, transporting convenience can be improved.
Owner:PROADVANCER LOGISTICS TECH SUZHOU CO LTD
Particle analyzer, constant-temperature reaction system and constant-temperature reaction method
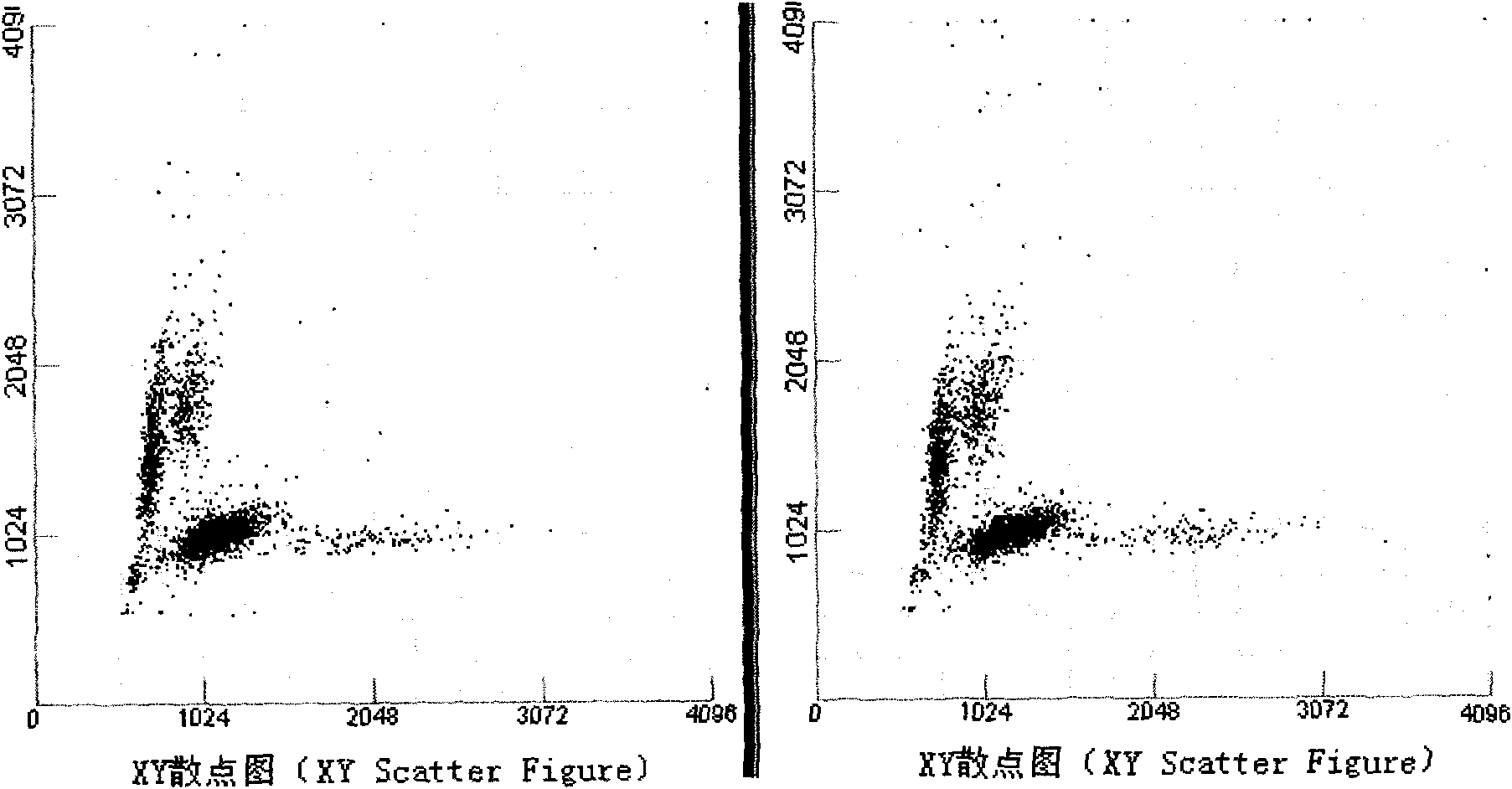
ActiveCN101799467AExtend heating timeExtend cooling timeBiological testingFluorescence/phosphorescenceTemperature controlFluorescence
The invention discloses a constant-temperature reaction system. The constant-temperature reaction system comprises a reaction tank, a dye pipe and a constant-temperature control device, wherein the reaction tank is used for providing a reaction place for a reactant; the dye pipe is used for delivering a fluorescent dye; one end of the dye pipe is communicated with the reaction tank; and the constant-temperature control device, the reaction tank and the dye pipe are arranged in such a manner that the constant-temperature control device controls the temperatures of the reaction tank and the dyepipe at a set temperature. Constant-temperature control is performed on the fluorescent dye according to the set temperature before the fluorescent dye enters the reaction tank, so heating or coolingtime of the dye is prolonged, the temperature of the dye is closer to the set temperature, the effect of ambient temperature on the dye is reduced, and the correctness and the stability of a measuring result are finally enhanced.
Owner:SHENZHEN MINDRAY BIO MEDICAL ELECTRONICS CO LTD +1