Patents
Literature
651results about "Carbonate preparation" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
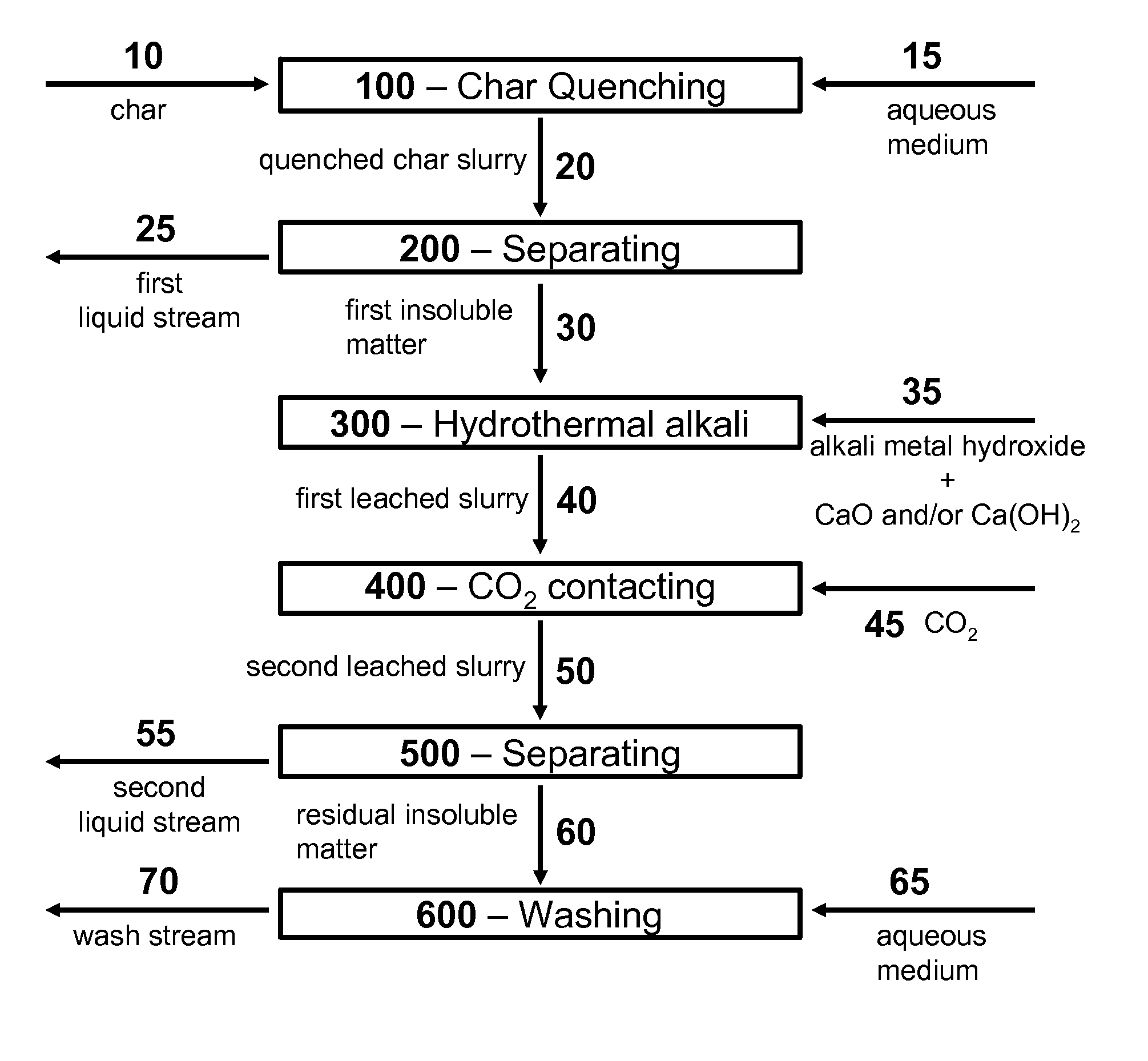
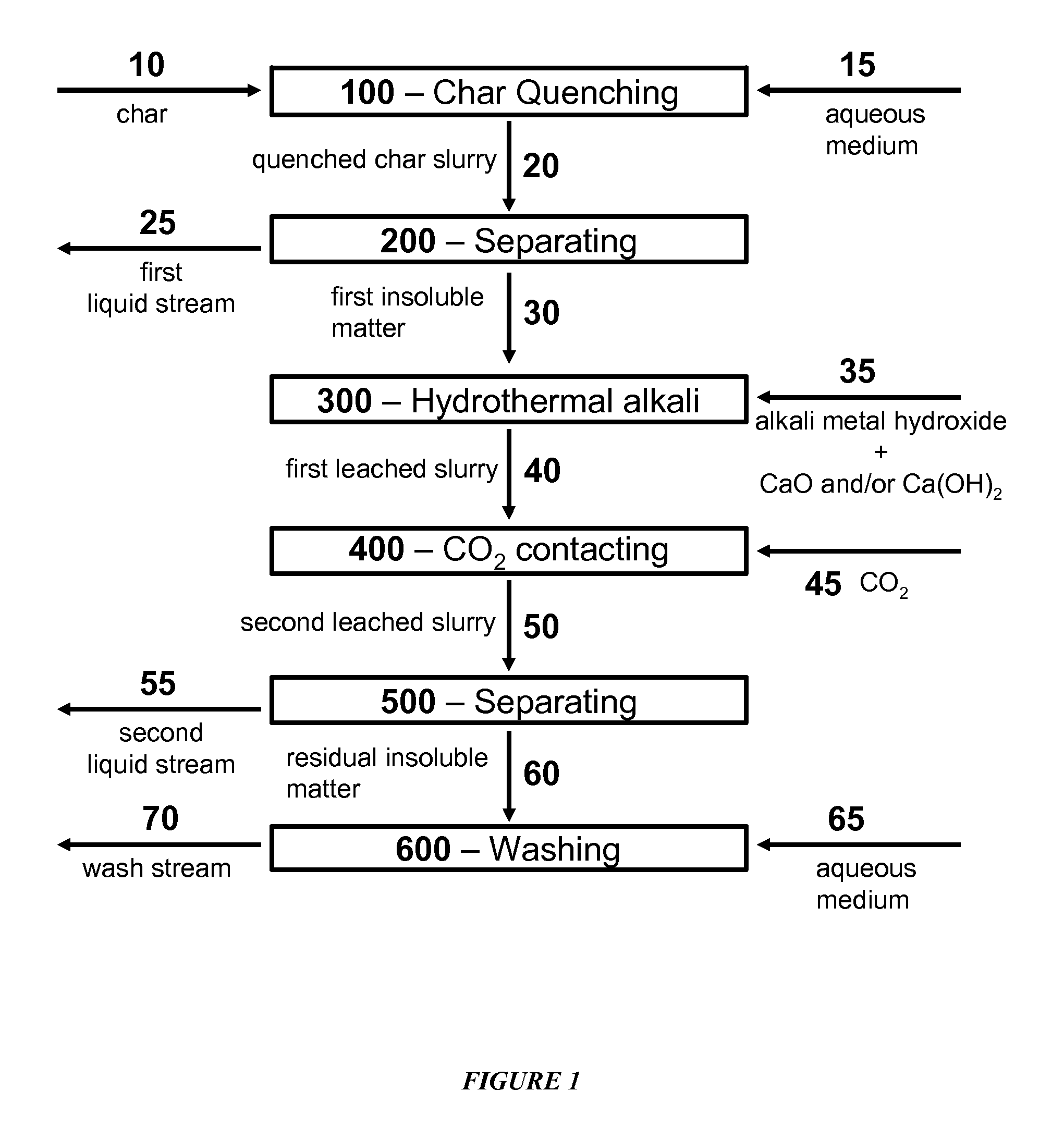
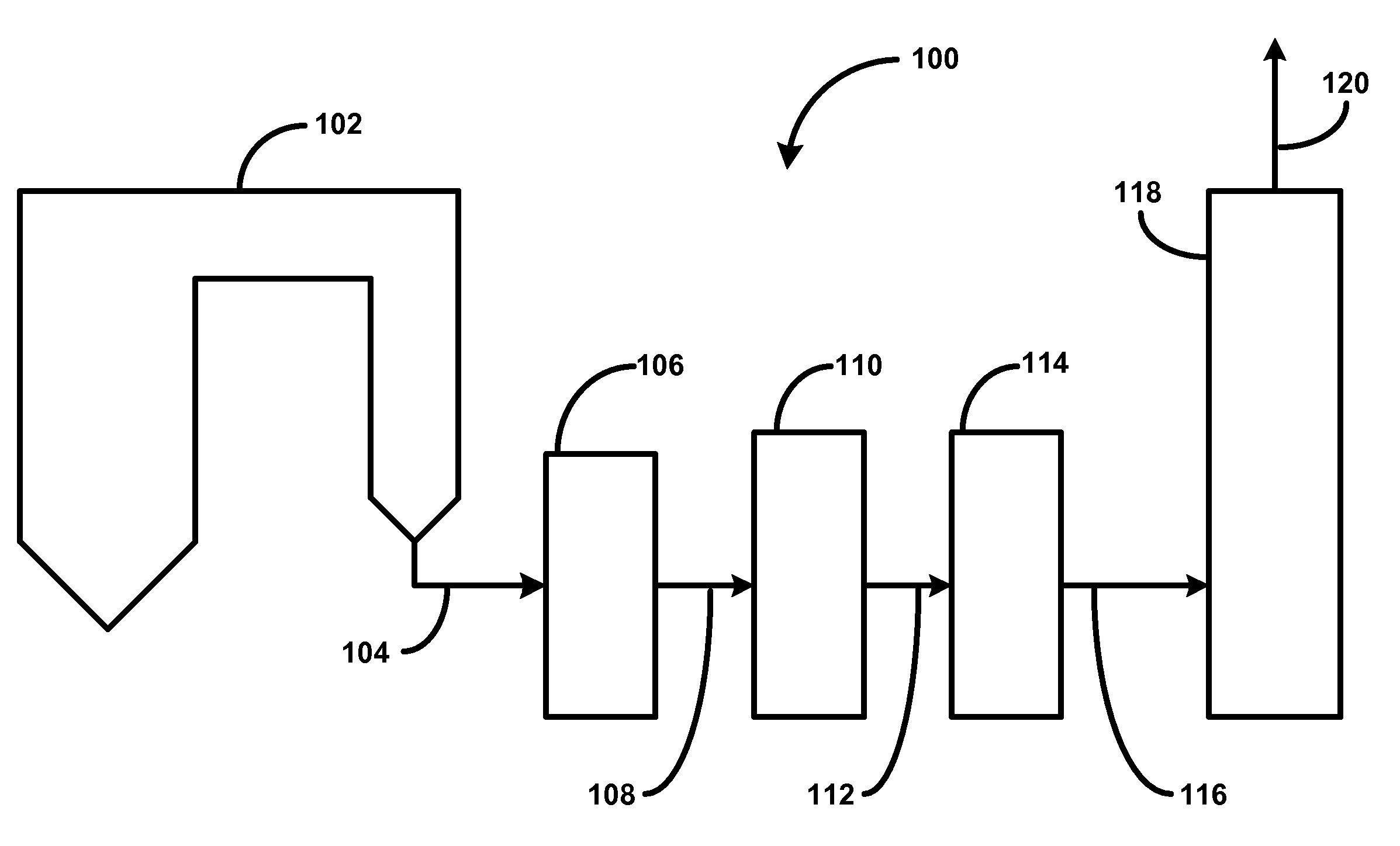
Catalytic Gasification Process with Recovery of Alkali Metal from Char
InactiveUS20090165383A1Quantity minimizationGaseous fuelsGasification processes detailsPhysical chemistryAlkali metal
Processes are described for the extraction and recovery of alkali metal from the char that results from catalytic gasification of a carbonaceous material. Among other steps, the processes of the invention include a hydrothermal leaching step in which a slurry of insoluble particulate comprising insoluble alkali metal compounds is treated with carbon dioxide and steam at elevated temperatures and pressures to effect the conversion of insoluble alkali metal compounds to soluble alkali metal compounds. Further, processes are described for the catalytic gasification of a carbonaceous material where a substantial portion of alkali metal is extracted and recovered from the char that results from the catalytic gasification process.
Owner:SURE CHAMPION INVESTMENT LTD
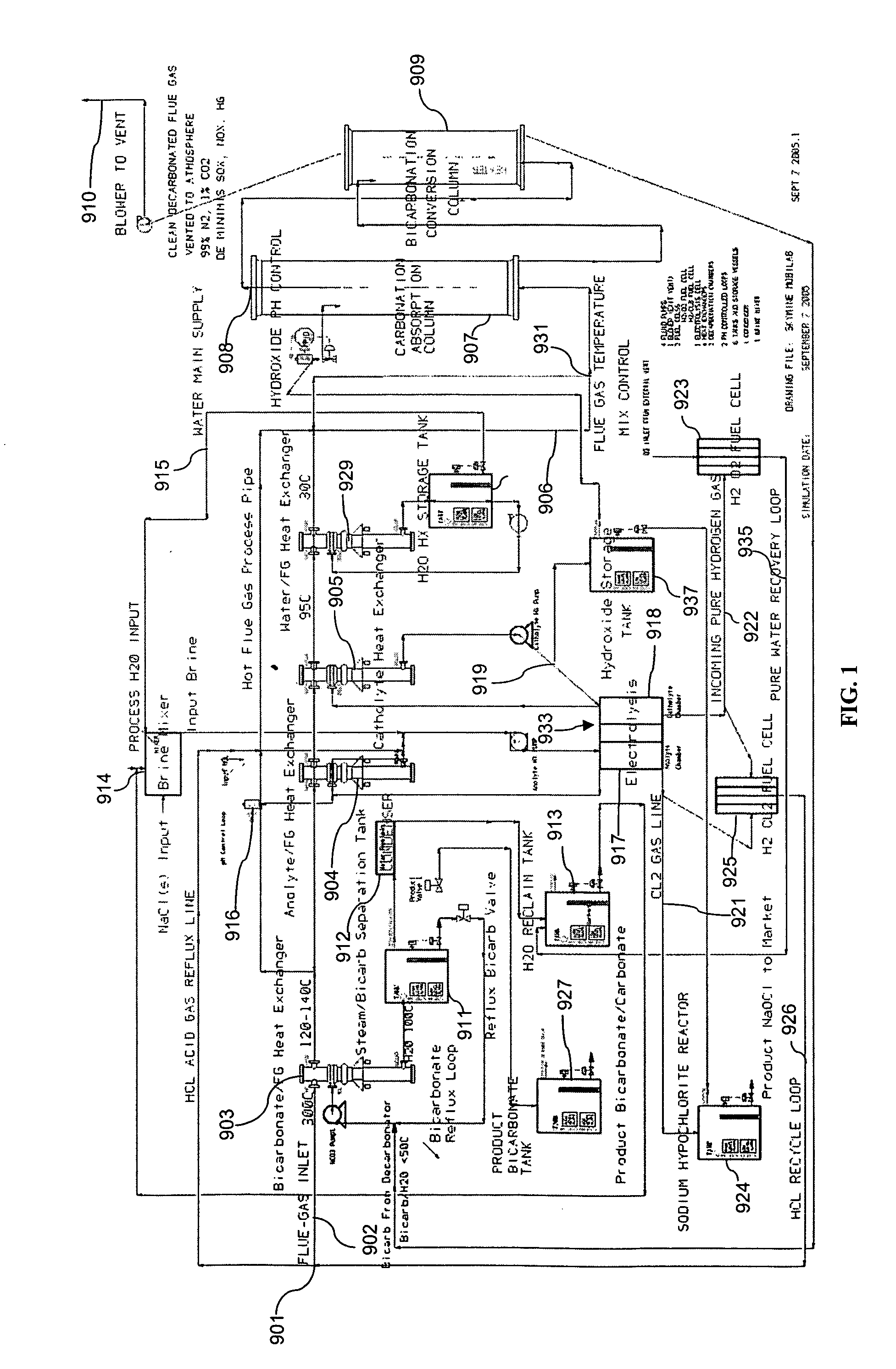
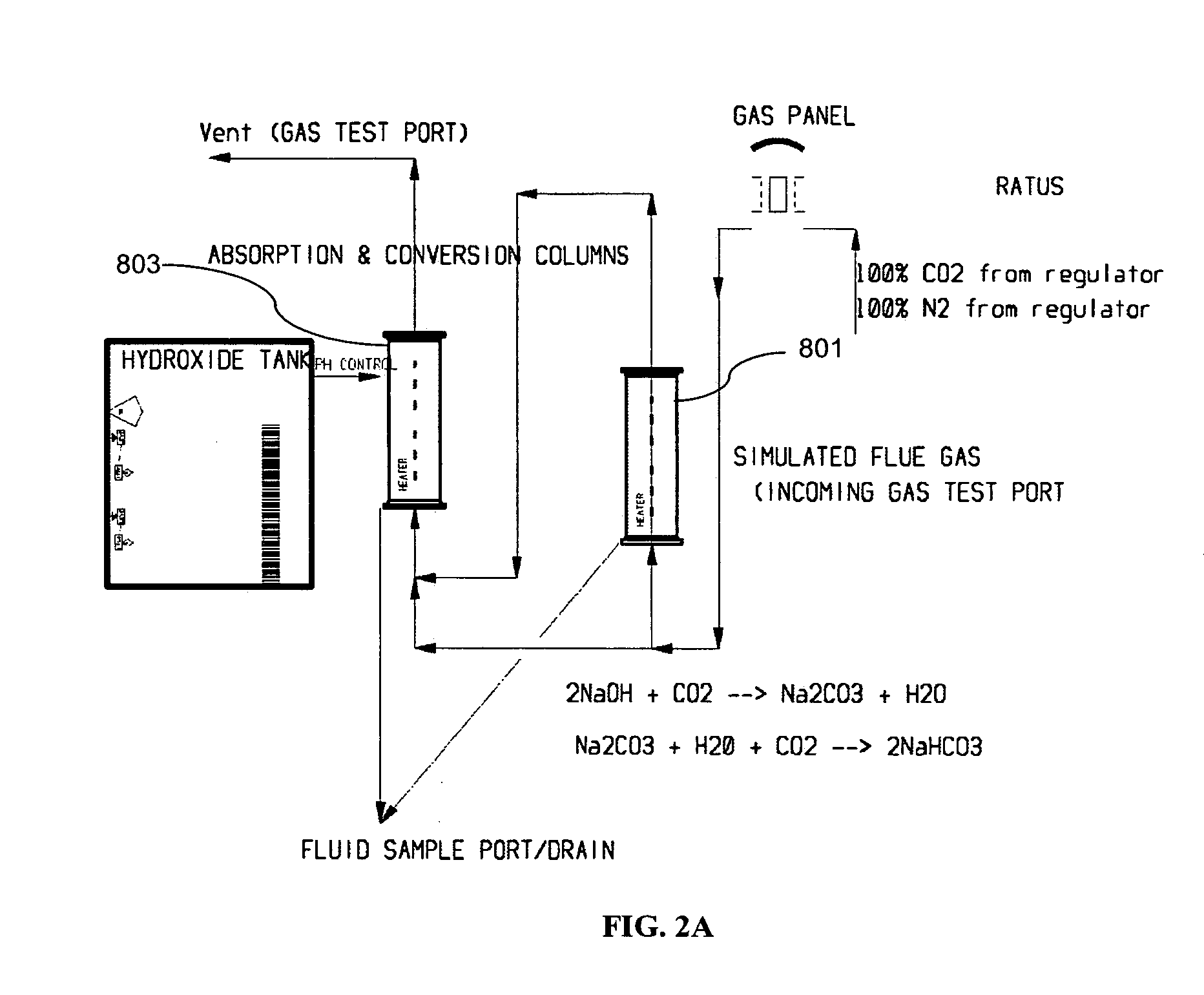
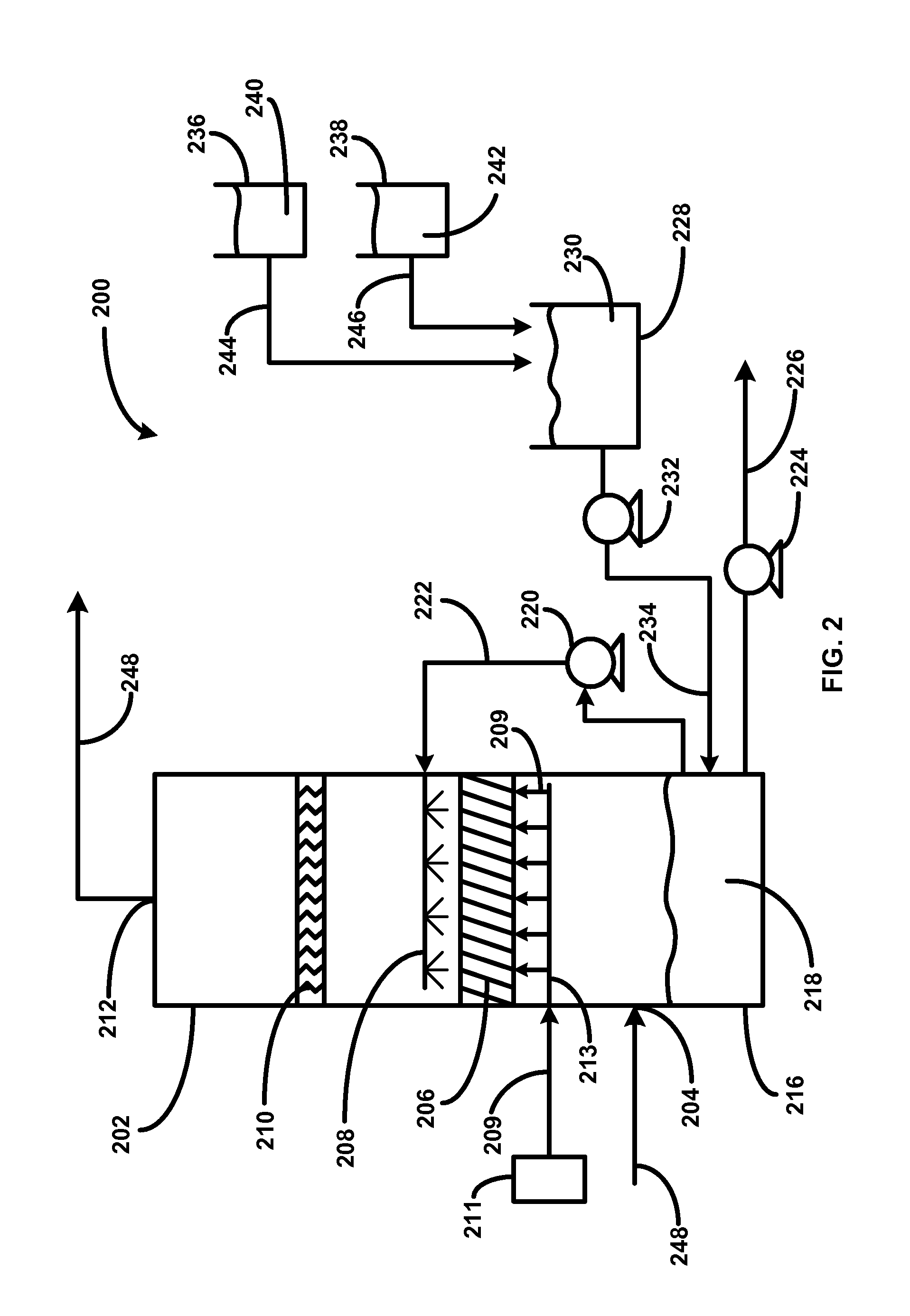
Removing carbon dioxide from waste streams through co-generation of carbonate and/or bicarbonate minerals
ActiveUS20060185985A1Improve ecologic efficiency of processEcologic efficiencyCalcium/strontium/barium carbonatesElectrolysis componentsElectrolysisWaste stream
Apparatuses and methods for removing carbon dioxide and other pollutants from a gas stream are provided. The methods include obtaining hydroxide in an aqueous mixture, and mixing the hydroxide with the gas stream to produce carbonate and / or bicarbonate. Some of the apparatuses of the present invention comprise an electrolysis chamber for providing hydroxide and mixing equipment for mixing the hydroxide with a gas stream including carbon dioxide to form an admixture including carbonate and / or bicarbonate.
Owner:CARBONFREE CHEM HLDG LLC
Systems and methods for carbon capture and sequestration and compositions derived therefrom
ActiveUS20090143211A1Promote formationCalcium/strontium/barium carbonatesNitrogen compoundsInterior spaceProduct formation
A method of sequestering a greenhouse gas is described, which comprises: (i) providing a solution carrying a first reagent that is capable of reacting with a greenhouse gas; (ii) contacting the solution with a greenhouse gas under conditions that promote a reaction between the at least first reagent and the greenhouse gas to produce at least a first reactant; (iii) providing a porous matrix having interstitial spaces and comprising at least a second reactant; (iv) allowing a solution carrying the at least first reactant to infiltrate at least a substantial portion of the interstitial spaces of the porous matrix under conditions that promote a reaction between the at least first reactant and the at least second reactant to provide at least a first product; and (v) allowing the at least first product to form and fill at least a portion of the interior spaces of the porous matrix, thereby sequestering a greenhouse gas.
Owner:RUTGERS THE STATE UNIV
Removing carbon dioxide from waste streams through co-generation of carbonate and/or bicarbonate minerals
ActiveUS7727374B2Calcium/strontium/barium carbonatesPhotography auxillary processesElectrolysisWaste stream
Apparatuses and methods for removing carbon dioxide and other pollutants from a gas stream are provided. The methods include obtaining hydroxide in an aqueous mixture, and mixing the hydroxide with the gas stream to produce carbonate and / or bicarbonate. Some of the apparatuses of the present invention comprise an electrolysis chamber for providing hydroxide and mixing equipment for mixing the hydroxide with a gas stream including carbon dioxide to form an admixture including carbonate and / or bicarbonate.
Owner:CARBONFREE CHEM HLDG LLC
Process for producing sodium bicarbonate for flue gas desulphurization
InactiveUS20100290967A1Electrolysis componentsVolume/mass flow measurementSodium bicarbonateFlue gas
Process for producing sodium bicarbonate for purifying flue gases, according to which an aqueous solution containing sodium sulfate is subjected to electrodialysis to produce a sodium hydroxide solution and a sodium bisulfate solution, the sodium hydroxide solution being carbonated in order to obtain sodium bicarbonate.
Owner:SOLVAY SA
Method and Apparatus for the Removal of Carbon Dioxide from a Gas Stream
The invention provides methods and apparatuses for removing carbon dioxide from a gas stream. In particular, the invention provides methods and apparatuses for absorbing carbon dioxide from a coal-fired boiler flue gas stream using an absorbing solution and for regeneration of an alkaline component used in the absorbing solution. In one embodiment, the invention provides a method for removing carbon dioxide from a gas stream by contacting a gas stream containing carbon dioxide with an alkaline liquid stream; absorbing at least a portion of the carbon dioxide into the alkaline liquid stream to produce absorbed carbon dioxide; and catalyzing a reaction of the absorbed carbon dioxide to a form of carbonate.
Owner:FLUEGEN
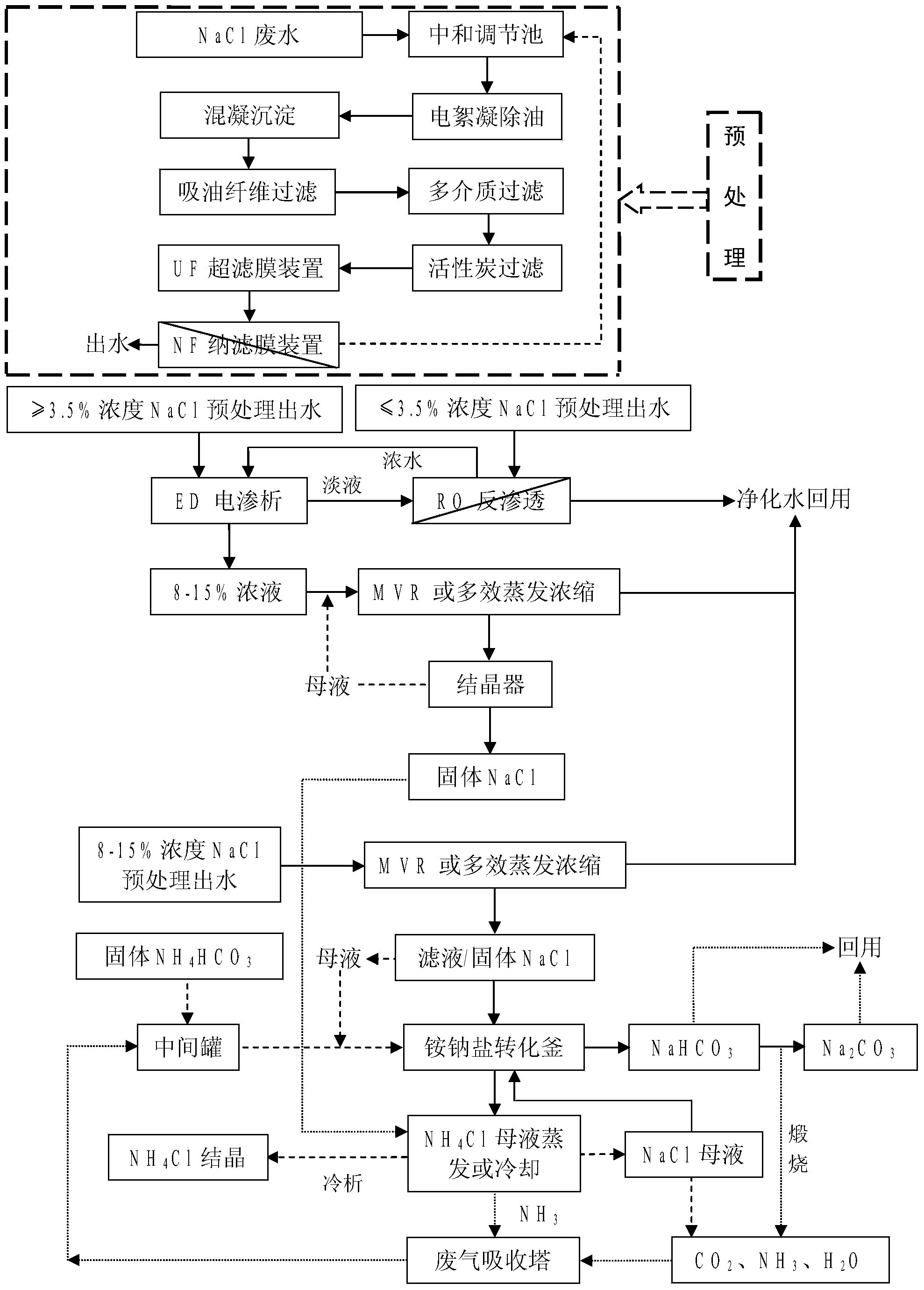
Combined treatment method for sodium chloride-containing wastewater generated in dressing and smelting of rare earth
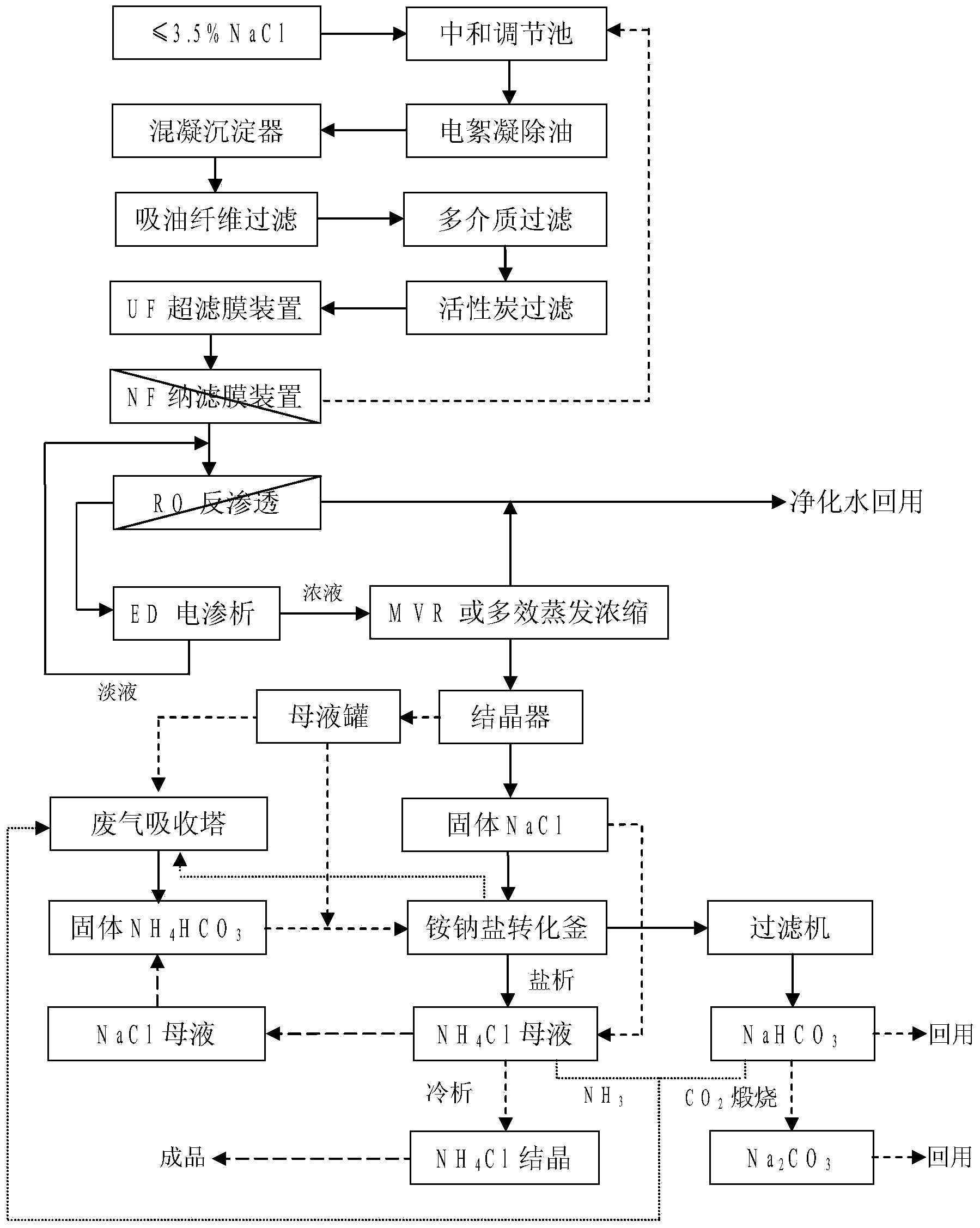
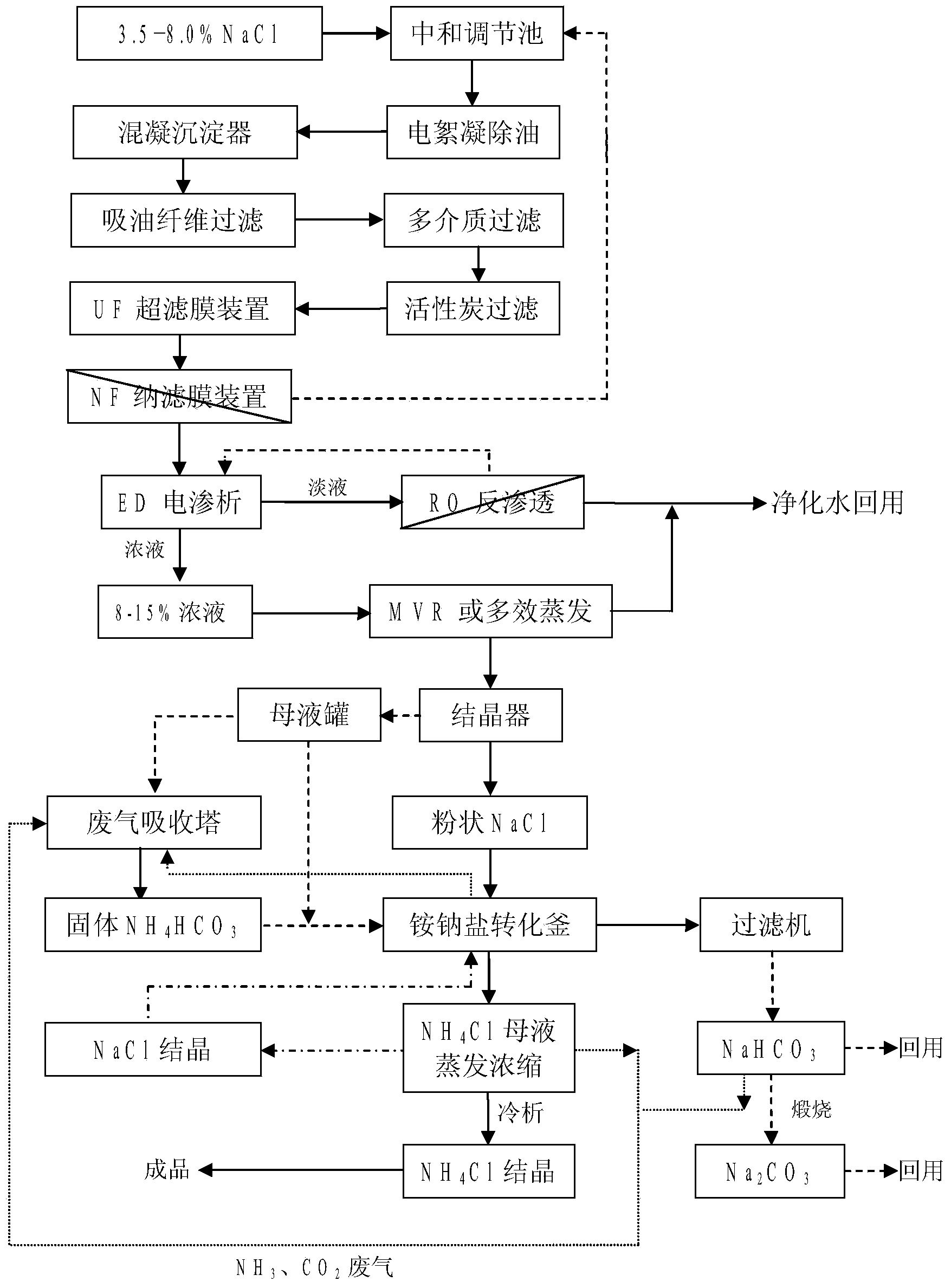
InactiveCN103449653AImprove environmental protection measuresAvoid volatile lossGeneral water supply conservationMultistage water/sewage treatmentSodium bicarbonateUltrafiltration
The invention relates to a combined treatment method for sodium chloride-containing wastewater generated in dressing and smelting of rare earth. According to the method, a series of desalination and concentration treatments such as deoiling, neutralizing, homogenizing, aeration, flocculation, ultrafiltration, nanofiltration, reverse osmosis, electroosmosis and MVR (mitral valve replacement) concentration are carried out on the wastewater, so that recyclable purified water is obtained; ammonium bicarbonate is added into the sodium chloride extracted from the wastewater, so that the sodium salt can be converted and regenerated into sodium bicarbonate, sodium carbonate and ammonium chloride products for dressing and smelting of the rare earth. According to the method, the recovery rate of the wastewater is high, the effluent quality is stable, the conversion rate of the sodium salt is high, and no three wastes are discharged; production equipment is compact, simple to operate, and easy to realize automation control; the problem that the sodium chloride-containing wastewater and other similar industrial wastewater generated in dressing and smelting of rare earth are difficult to recycle is solved.
Owner:YANSHAN UNIV
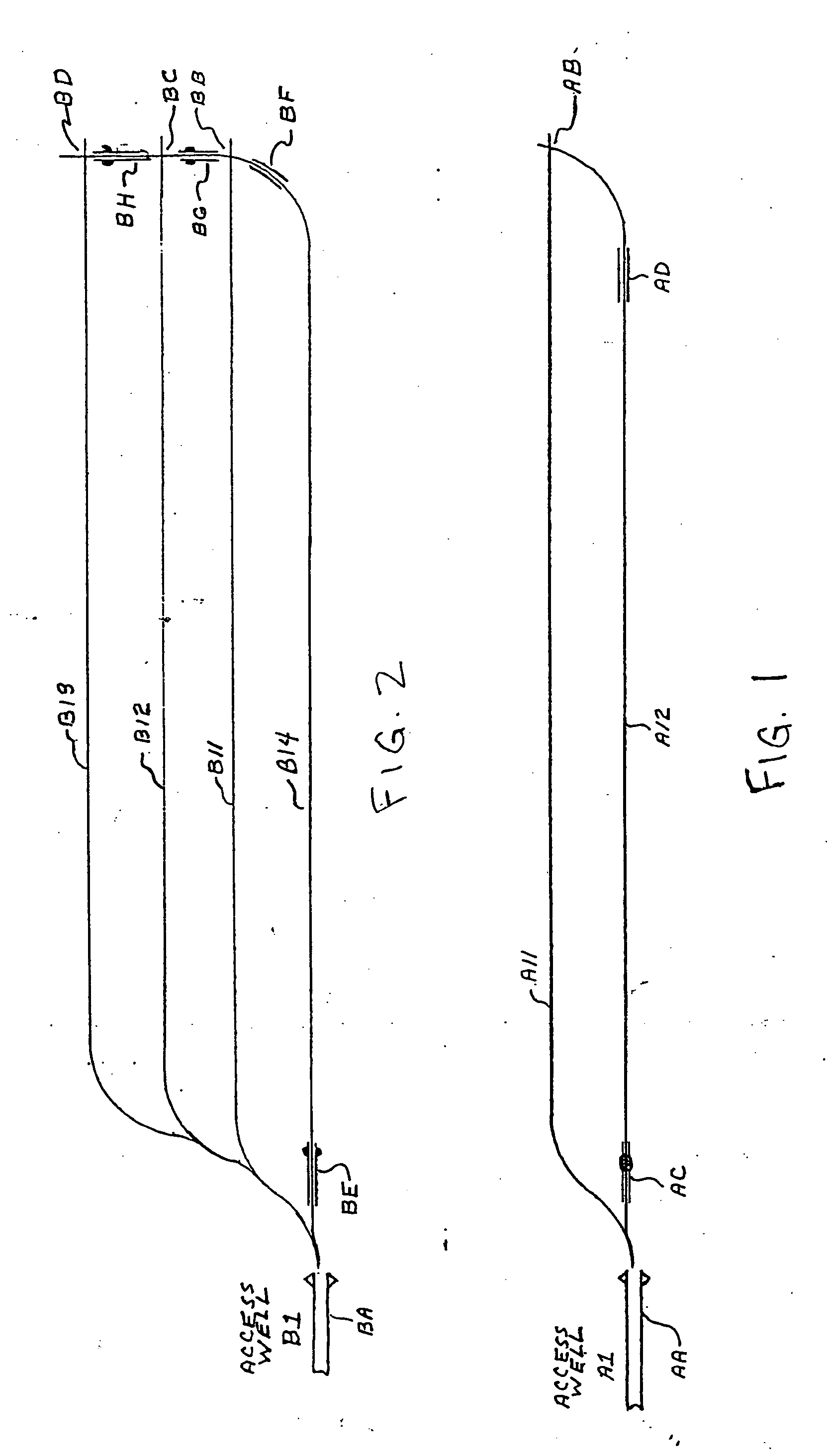
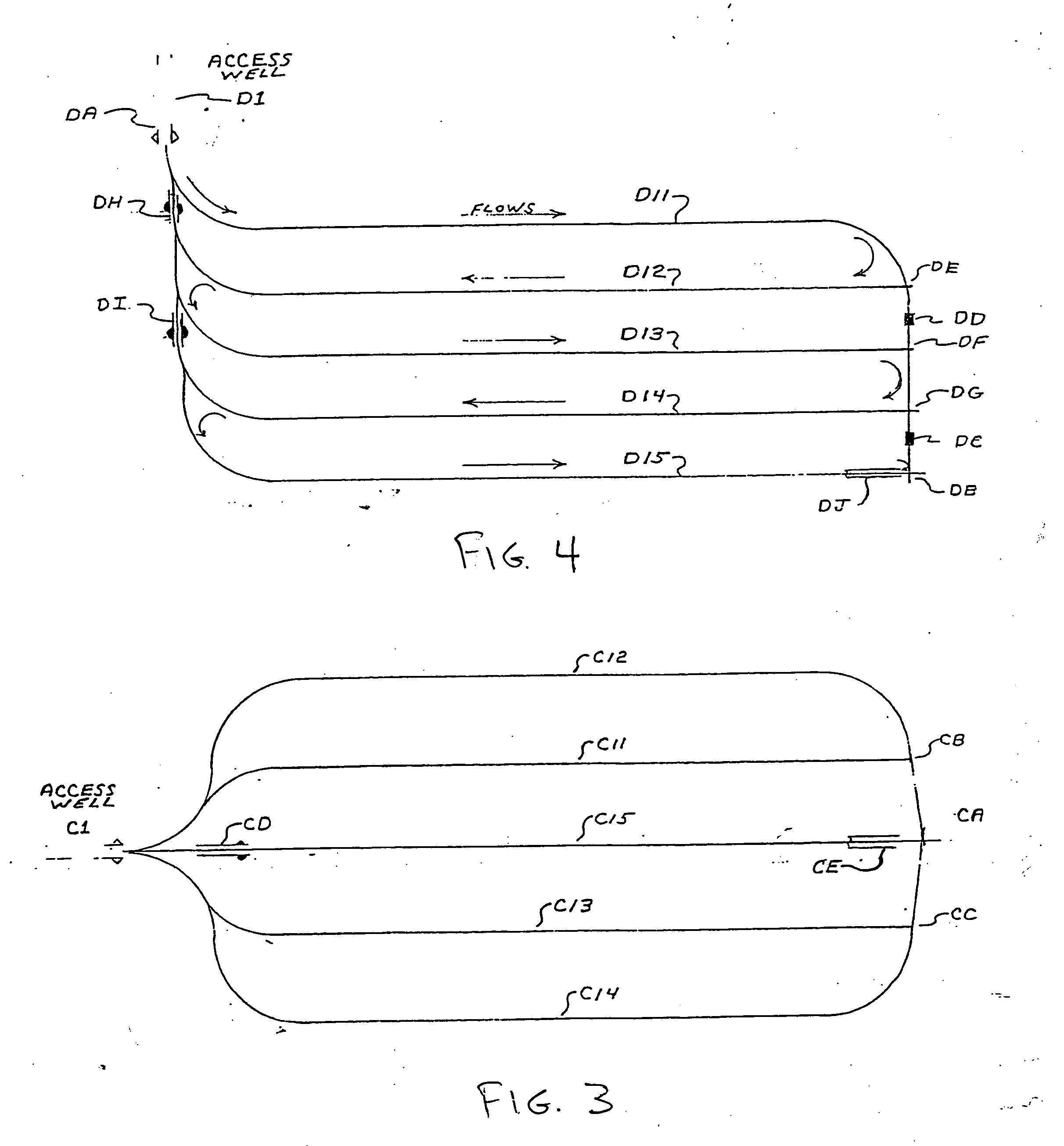
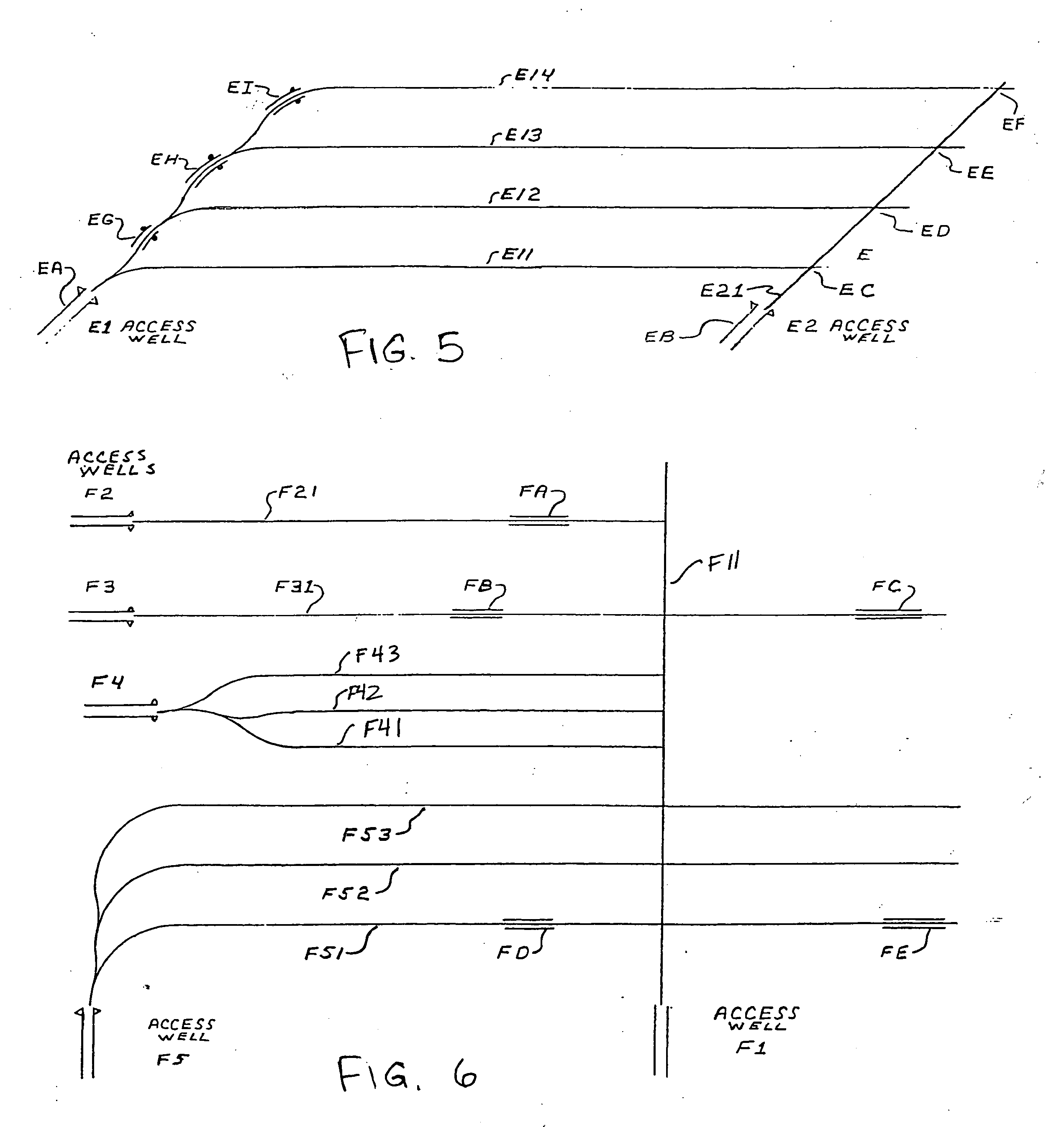
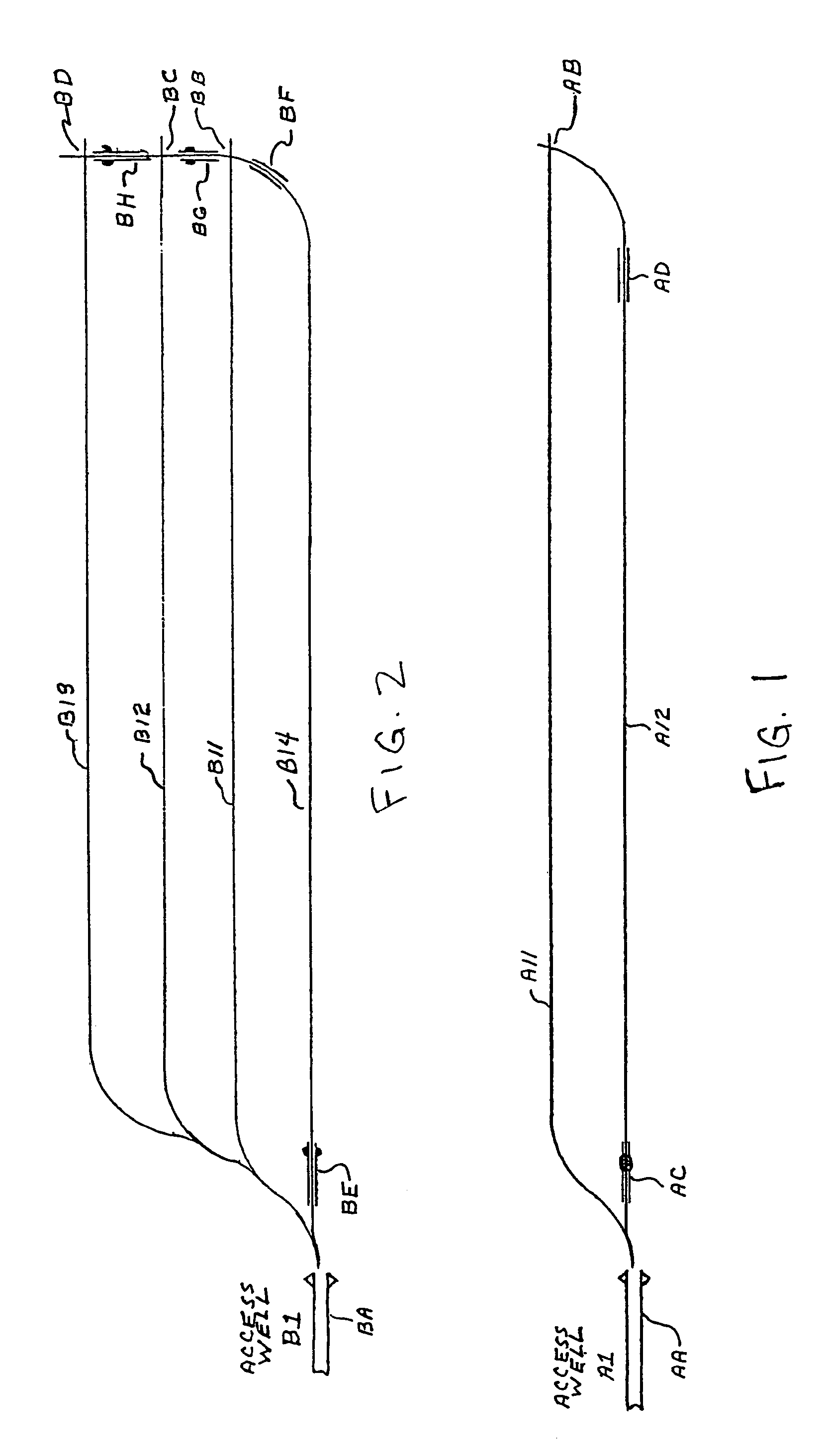
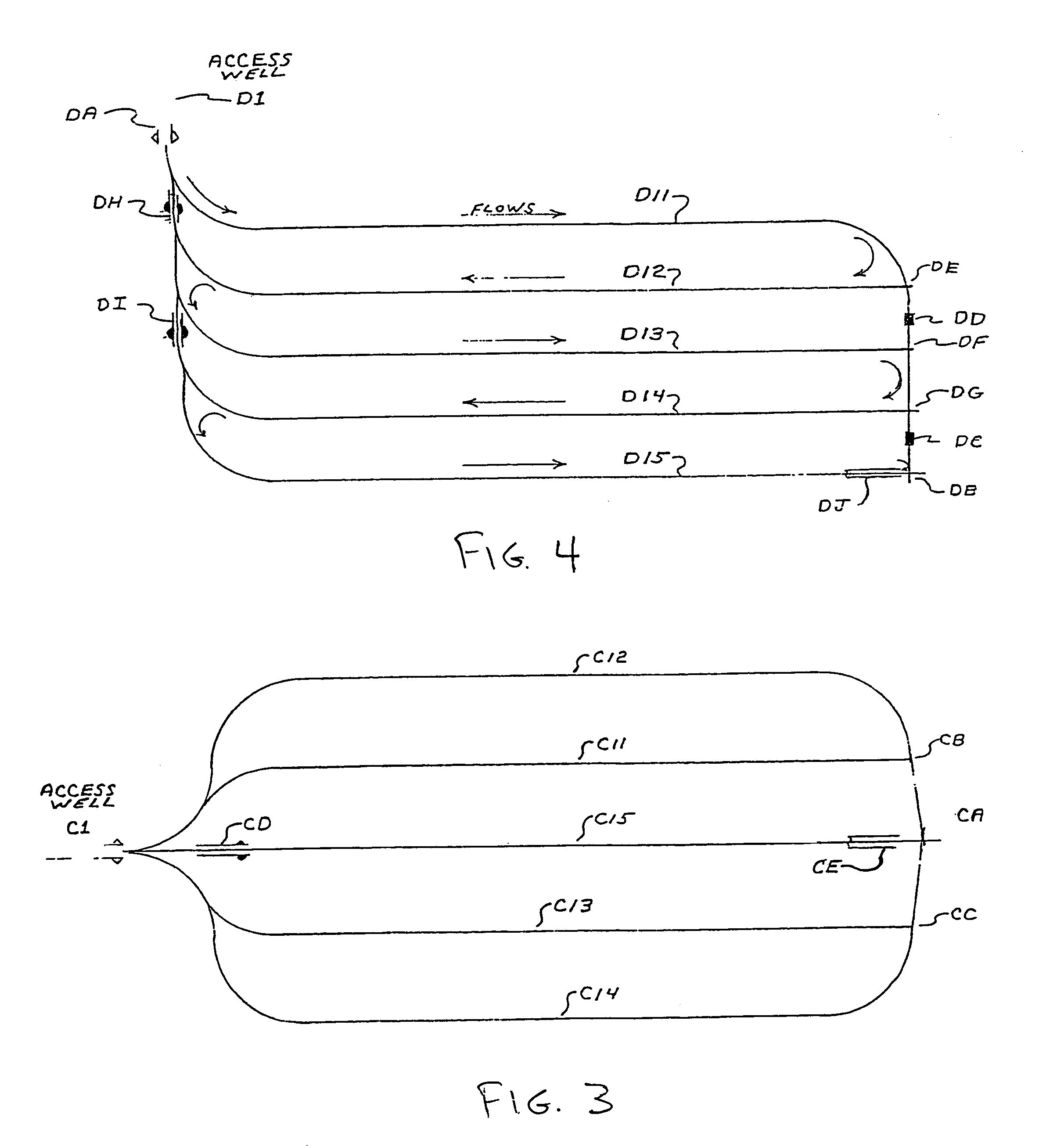
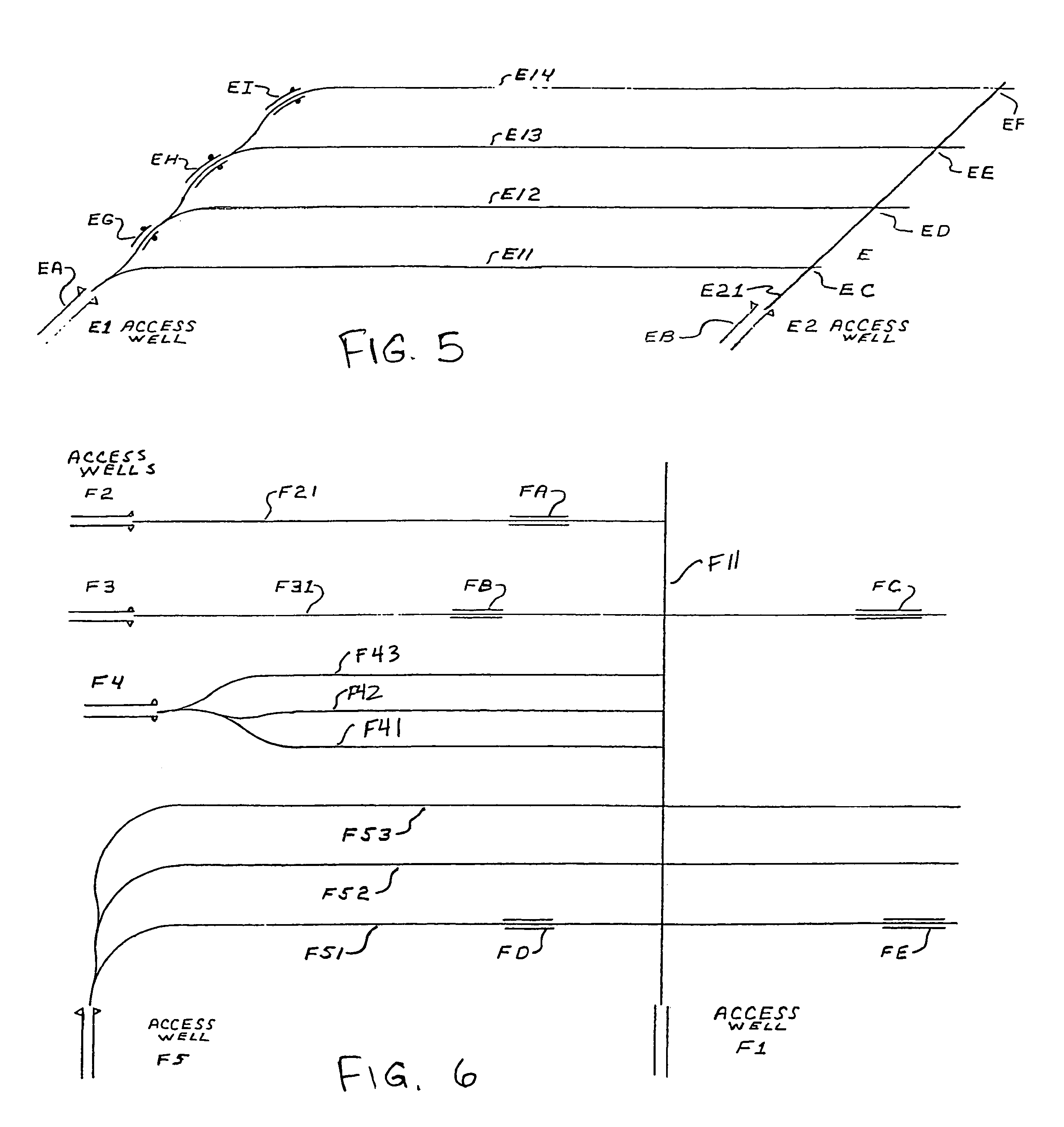
Methods for constructing underground borehole configurations and related solution mining methods
Disclosed are methods for solution mining of evaporite minerals, such as trona, comprising drilling an access well and at least two lateral boreholes; injecting a fluid; circulating the fluid through the lateral boreholes with a controlled fluid flow; and collecting a pregnant solution. Also disclosed are methods of solution mining that include injecting an aqueous solution into an underground trona cavity at a temperature sufficient to maintain at least a portion of the solution in the cavity in the Wegscheiderite solid phase region; removing aqueous solution from the cavity; and recovering alkaline values from the removed aqueous solution. Also disclosed are methods of solution mining that include injecting an aqueous solution into an underground trona cavity; removing aqueous solution from the cavity, wherein the temperature of the removed aqueous solution is at about the TWA point temperature; and recovering alkaline values from the removed aqueous solution.
Owner:SESQUI MINING
Process for production of dense soda, light soda, sodium bicarbonate and sodium silicate from solutions containing bicarbonate
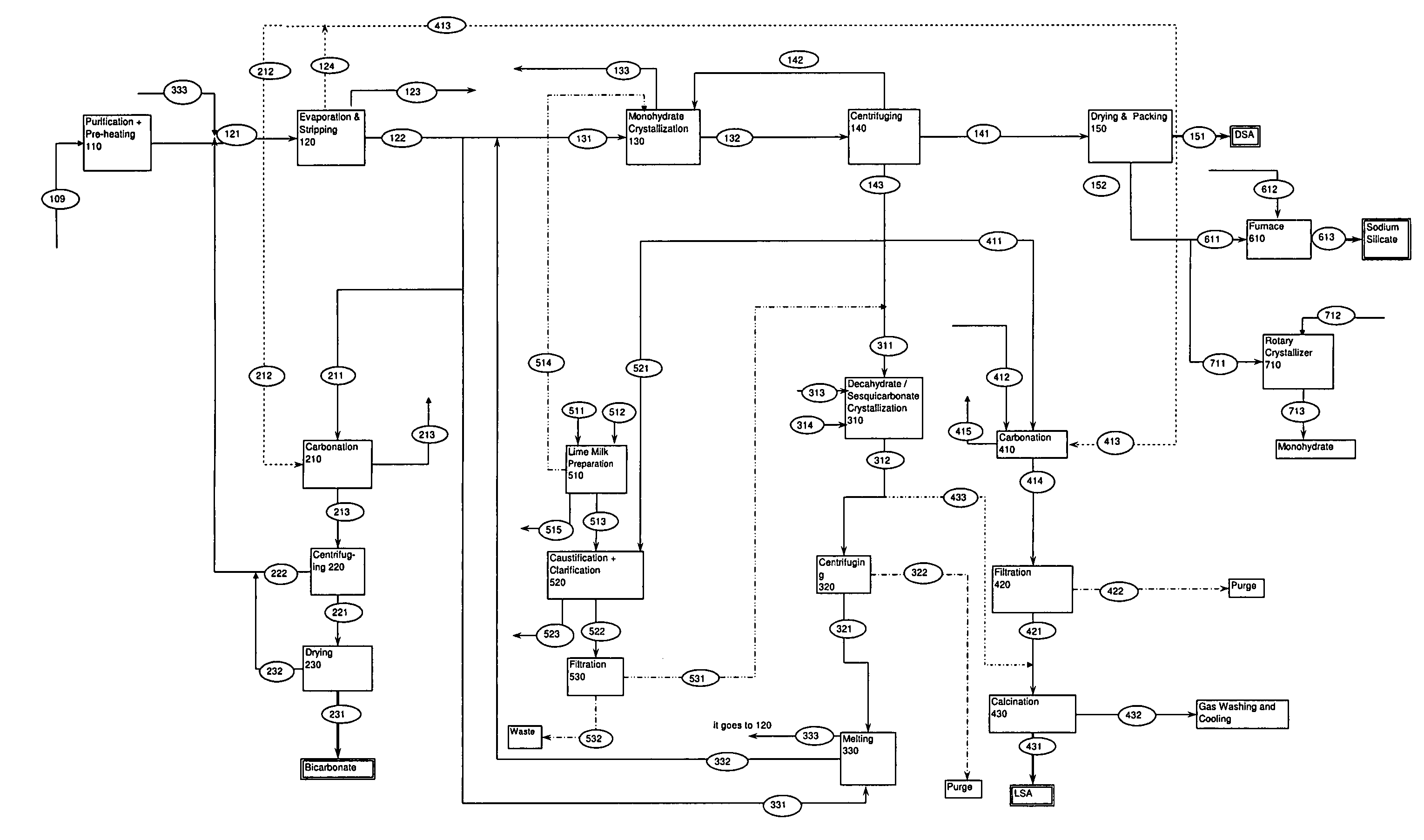
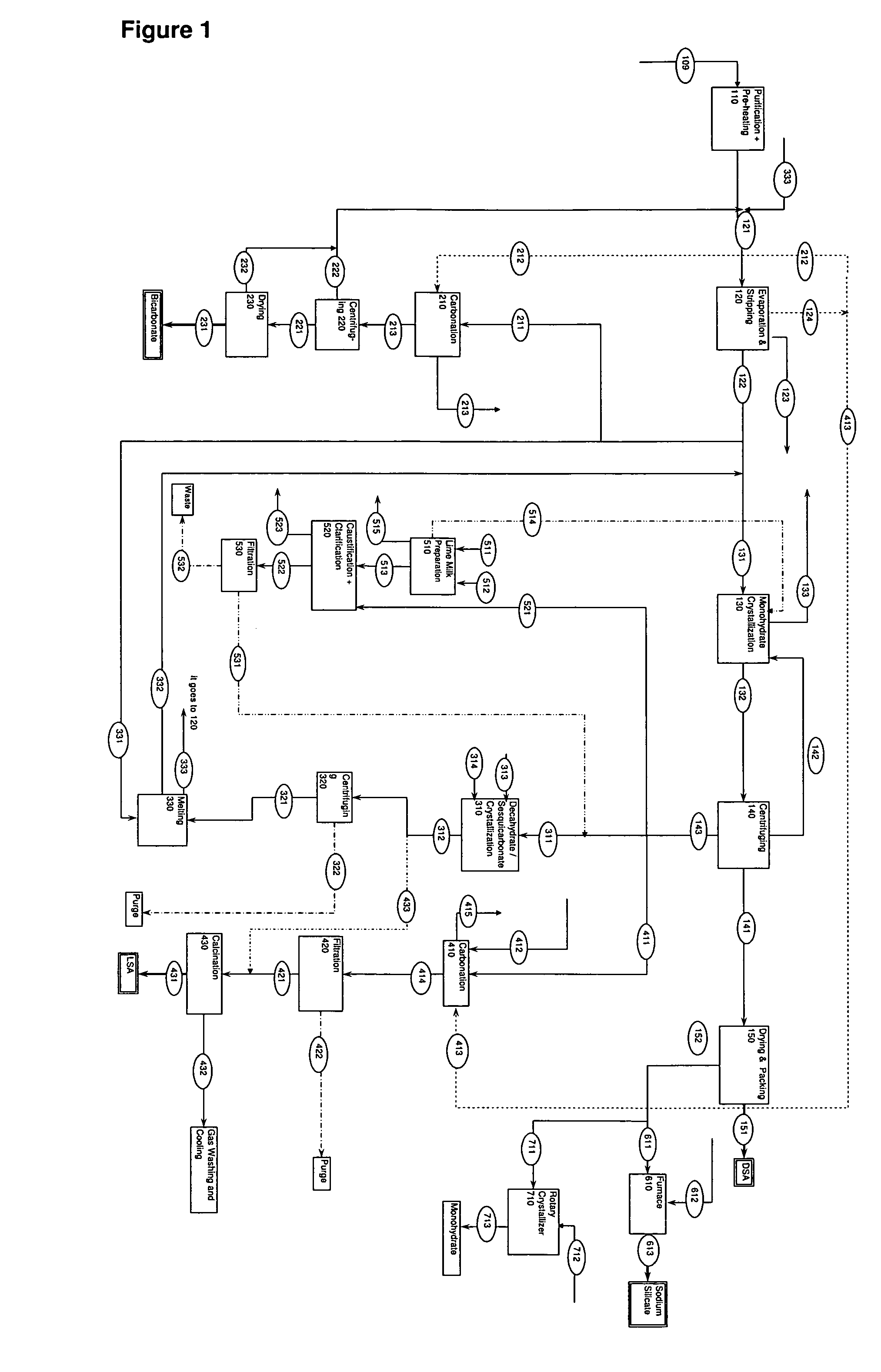
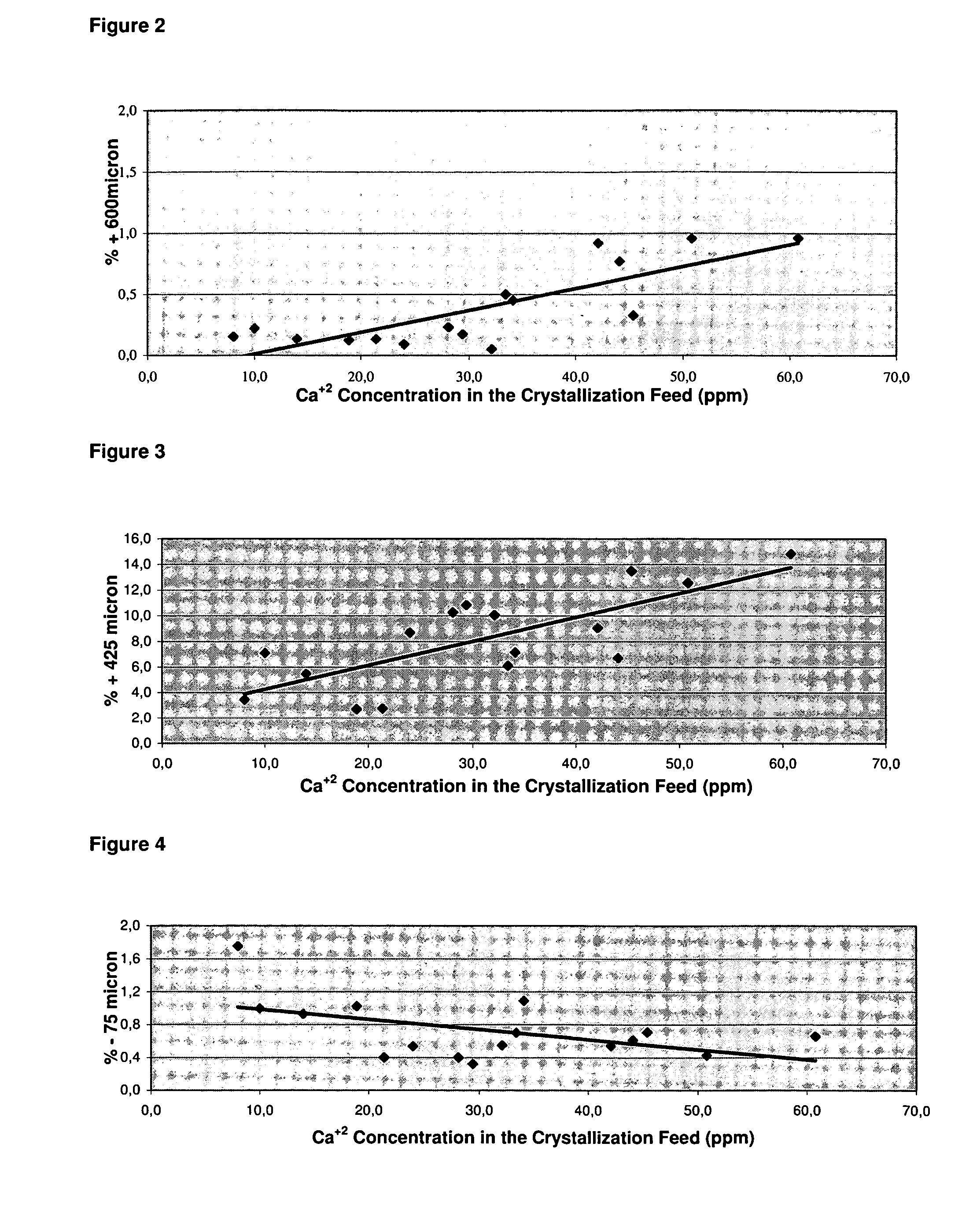
ActiveUS7507388B2Processing requirementLow production costCrystallization separationAlkali metal silicatesSodium bicarbonateEvaporation
A process related to sodium chemicals production, including the processing of bicarbonate containing solutions obtained by solution mining of trona, nahcolite or wegscheiderite reserves and the lake waters containing bicarbonates, includes the steps of purification, evaporation-decarbonation, crystallization, centrifuging, and drying.
Owner:ETI SODA URETIM PAZARLAMA NAKLIYAT & ELEKTRIK URETIM SANAYI & TICARET A S
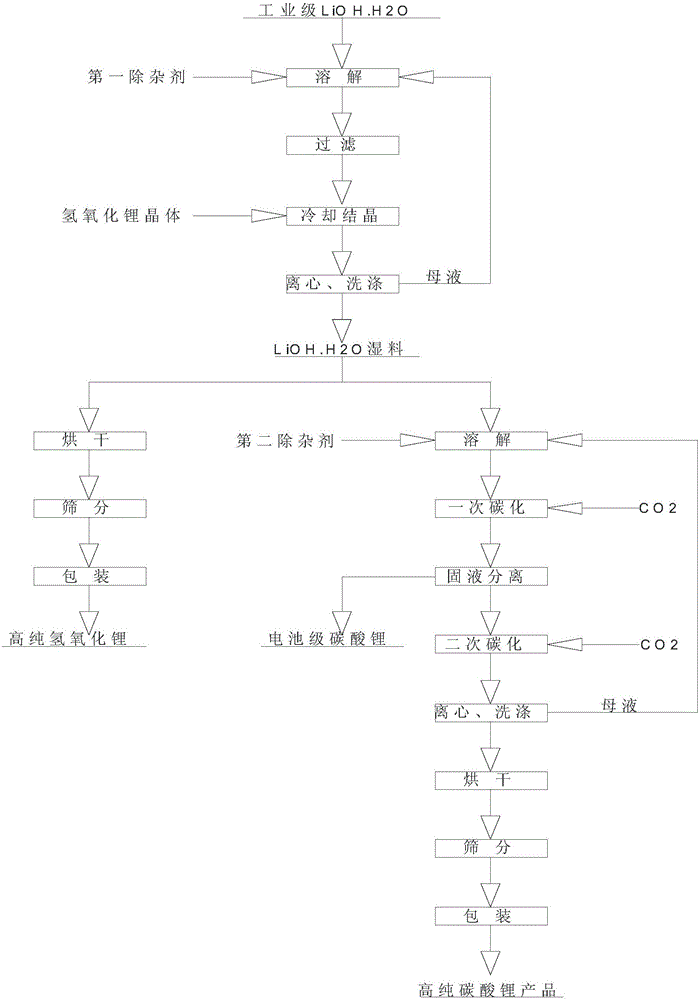
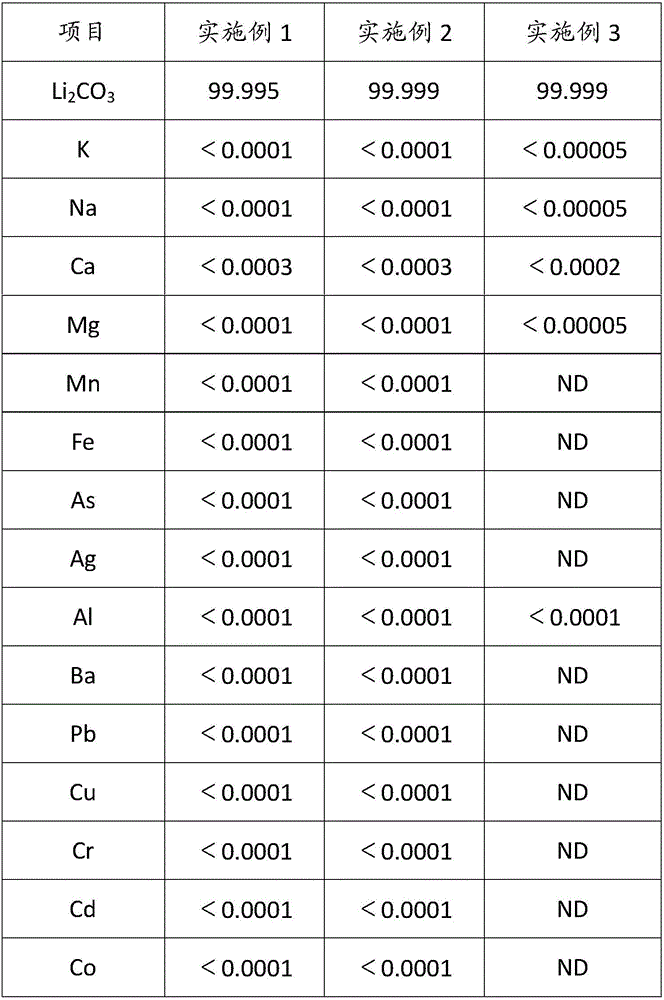
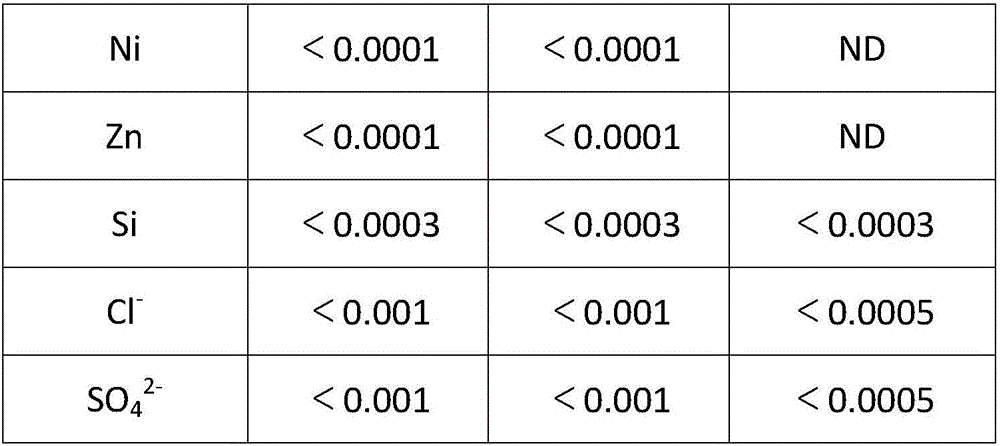
Method for composite production of high-purity lithium hydroxide monohydrate, high-purity lithium carbonate and battery grade lithium carbonate
ActiveCN106315625AHigh recovery rateAdjust outputAlkali metal oxides/hydroxidesCarbonate preparationLithium carbonatePhysical chemistry
The invention discloses a method for composite production of high-purity lithium hydroxide monohydrate, high-purity lithium carbonate and battery grade lithium carbonate. Industrial grade lithium hydroxide is dissolved into pure water or crystallization mother liquor, and a first solution is prepared; a first impurity removal agent is added into the first solution, then filtering is performed, a lithium hydroxide crystal is added into a filtrate, cooling crystallization is performed, solid-liquid separation is performed to obtain a high-purity lithium hydroxide monohydrate wet material and crystallization mother liquor; the high-purity lithium hydroxide monohydrate wet material is dissolved into pure water or carbonization mother liquor, and a second solution is prepared; carbon dioxide is introduced into the second solution to perform primary carbonization, and solid-liquid separation is performed to obtain a lithium carbonate wet material and a filtrate; a second impurity removal agent is added into the filtrate, after a heat preservation reaction is performed, carbon dioxide is introduced to perform secondary carbonization, then, introduction of the carbon dioxide is stopped, the reaction continues to be performed, then, solid-liquid separation is performed, obtained solids are washed, a high-purity lithium carbonate wet material and carbonization mother liquor are obtained, and after the high-purity lithium carbonate wet material is post-processed, a high-purity lithium carbonate product is obtained.
Owner:CHENGDU CHEMPHYS CHEM IND
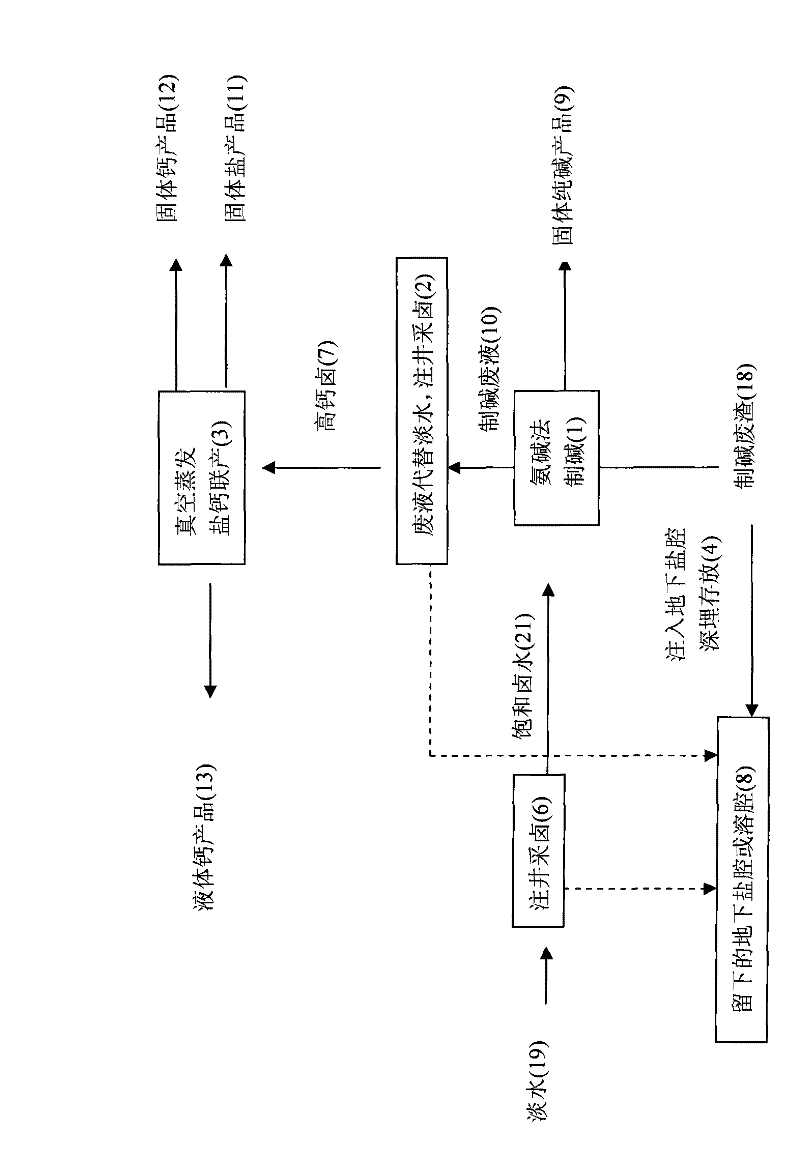
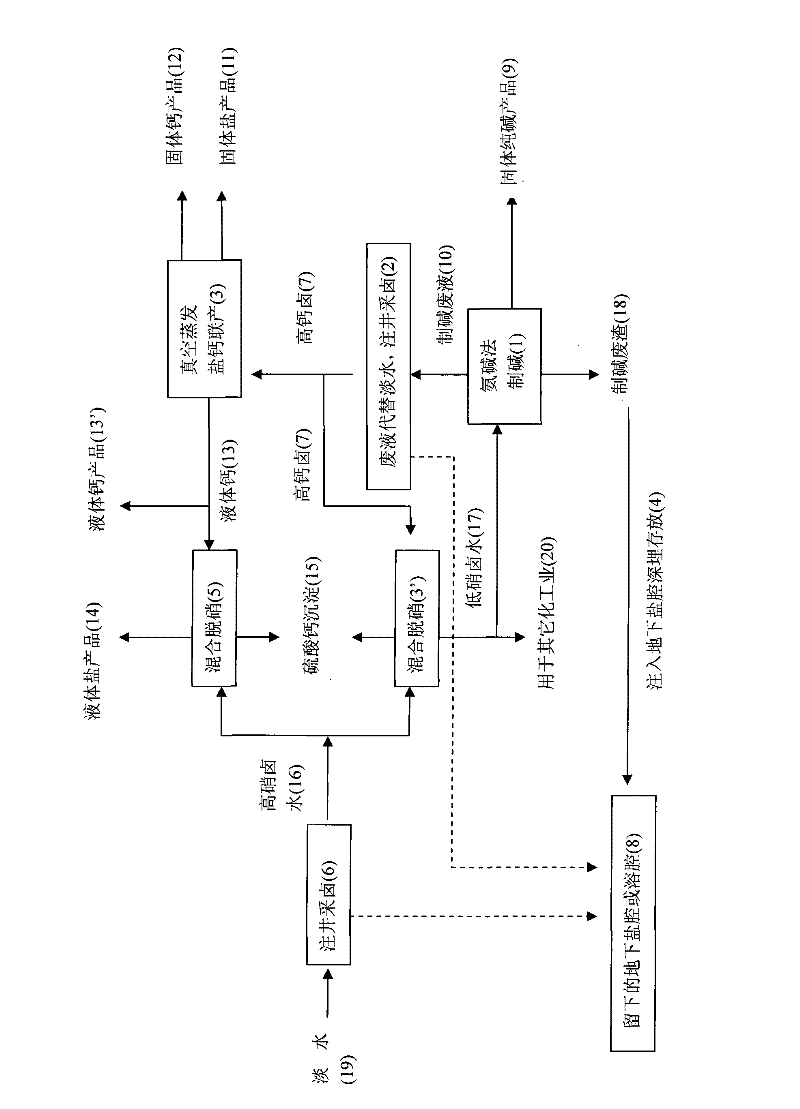
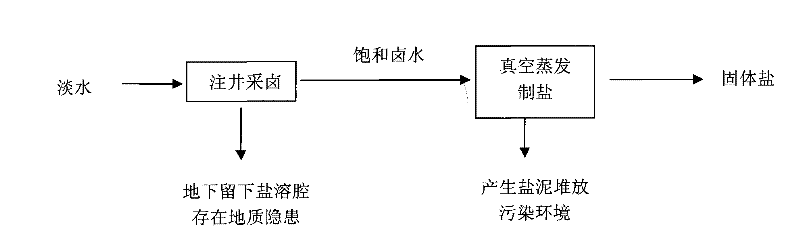
Salt, alkali and calcium combined cycle production technology by the use of well mineral salt
ActiveCN102205979ASolve processing problemsRealize energy saving and consumption reductionCalcium/strontium/barium chloridesAlkali metal chloridesDissolutionCalcium EDTA
The invention relates to a salt, alkali and calcium combined cycle production technology by the use of well mineral salt. Based on the exploitation of brine from well mineral salt, the salt, alkali and calcium combined cycle production technology by the use of well mineral salt is a cycle production technology, which focuses on the technology for making alkali from brine by the ammonia-soda process and takes account of the vacuum salt-making (including the brine refinement) and calcium-making technologies. Alkali-making wastewater replaces most fresh water to be injected into a salt mine well to collect brine, and the obtained high-calcium brine is taken as the raw material of the salt and calcium (including liquid calcium) co-production as well as the raw material for preparing alkali and other chemical production raw materials, wherein the liquid calcium can be used to produce liquid salt; alkali-making waste residues are injected into an underground dissolution cavern, namely a salt cavern which is formed by the exploitation of brine from well mineral salt, thus accomplishing the combined cycle production technology in which alkali-making and salt-making technologies are closely related with materials. The produced wastes are used as resources or undergo a harmless treatment, thus protecting the environment. In addition, the utilization rate of sodium chloride in rock salt reaches 100%, achieving maximum efficiency.
Owner:江苏苏盐井神股份有限公司 +1
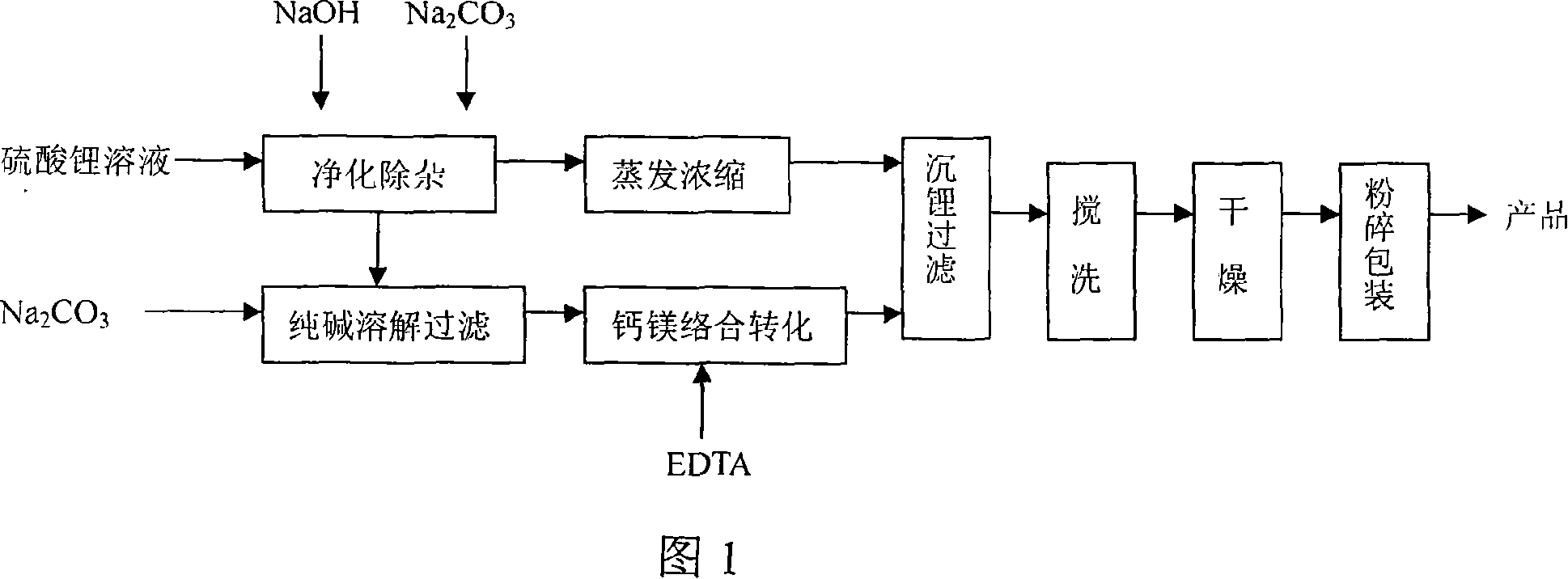
Method for producing low-magnesium battery-stage lithium carbonate from lithium sulfate solution
ActiveCN101125668ALower magnesium levelsAchieve preparationCarbonate preparationLithium carbonateChemistry
The invention provides a method to produce a low-magnesium battery grade lithium carbonate from lithium sulfate solution. The method comprises the steps that: (1) purification treatment of lithium sulfate solution as following: lithium sulfate solution is co-precipitated to lower the impurities ions Fe3+, Mg2+, Al3+ and Ca2+, and condensed and filtered to further eliminate impurities, and obtain purification residues and a pure finish solution of lithium sulfate; (2) soda is dissolved and added with purification residues obtained from the purified lithium sulfate solution, and used for a filtering media to filter calcium and magnesium and obtain purified soda solution; (3) the purified soda solution is added with complexant EDTA, and stirred for complexation reaction, and slowly added with condensed and impurities eliminated pure finish solution of lithium sulfate, and a crude lithium carbonate is prepared; (4) the crude lithium carbonate is stirred, cleaned, dried and smashed, thus obtaining the low-magnesium battery grade lithium carbonate. The method of the invention has the advantages of simple production technique, stable product quality and low cost, and expertly employs the waste residues in the process, not only solves the difficulty of impurities elimination for soda, but also enhances the recycling ratio of lithium, which is suitable for the production application of the positive pole material of Li-ion battery.
Owner:TIANQI LITHIUM CORP
Process for the treatment of waste metal chlorides
InactiveUS20060183958A1Low costMaximize recoveryTitanium tetrachlorideSilicon oxidesMetal chlorideOrganic chloride compound
A process is described for treating the residues from metal chlorination processes wherein valuable volatile metal chlorides or metalorgano chlorides are recovered while low volatility metal chlorides and chloride complexes are reacted with a neutralizing humectant. The resulting neutral, dry solid is suitable for land fill disposal or for recovery of valuable metal constituents by extractive metallurgy techniques.
Owner:REC SILICON
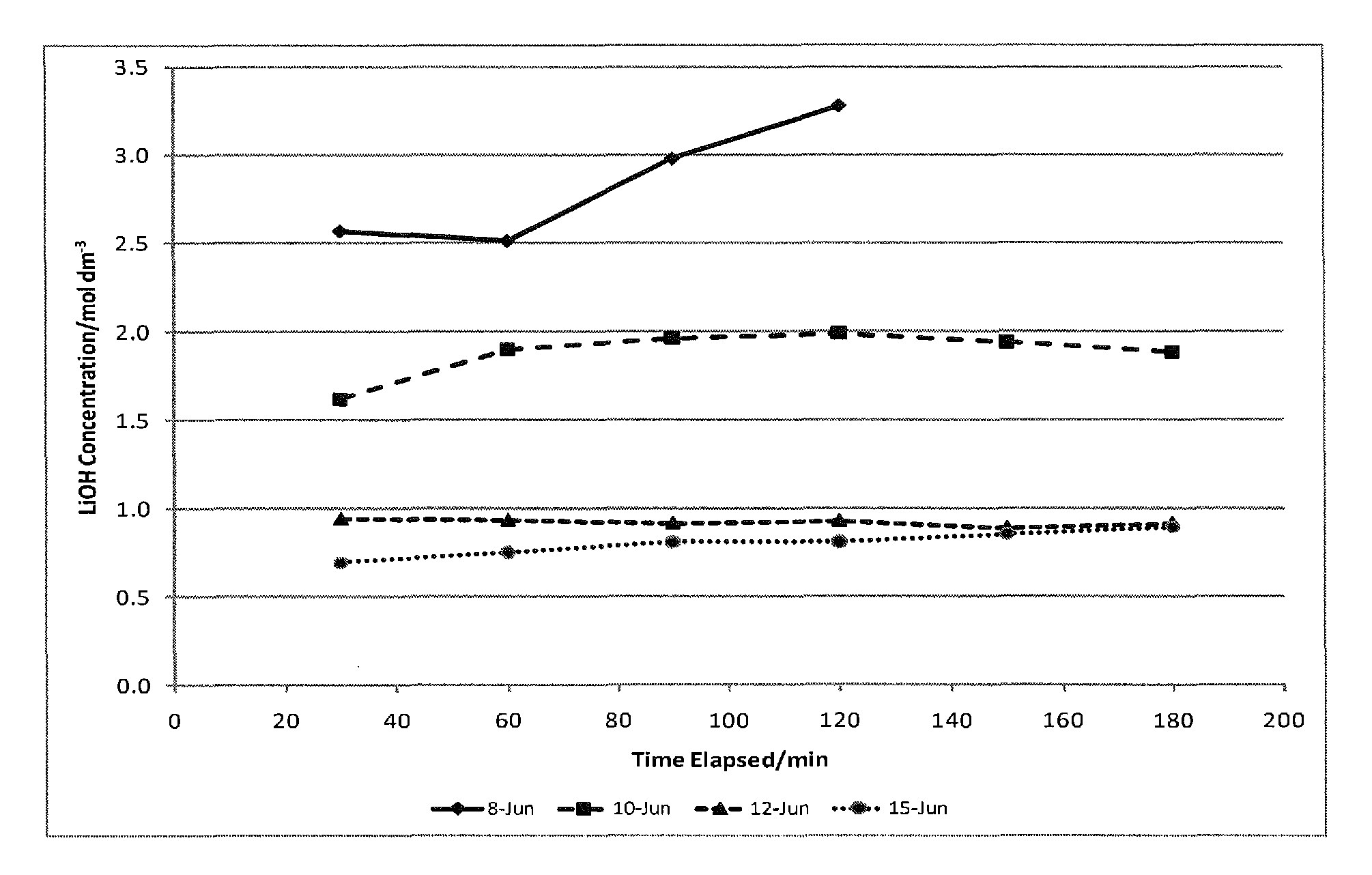
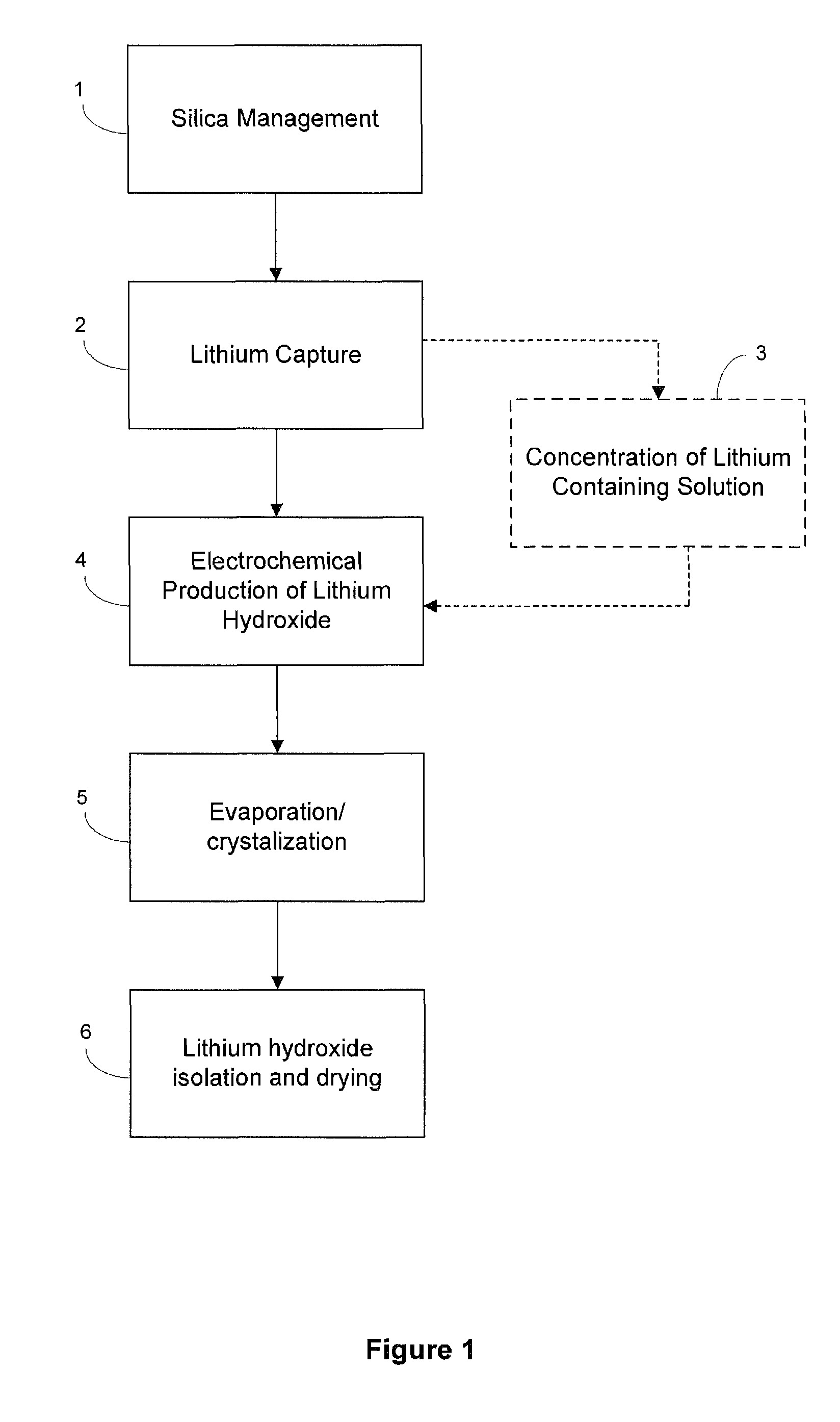
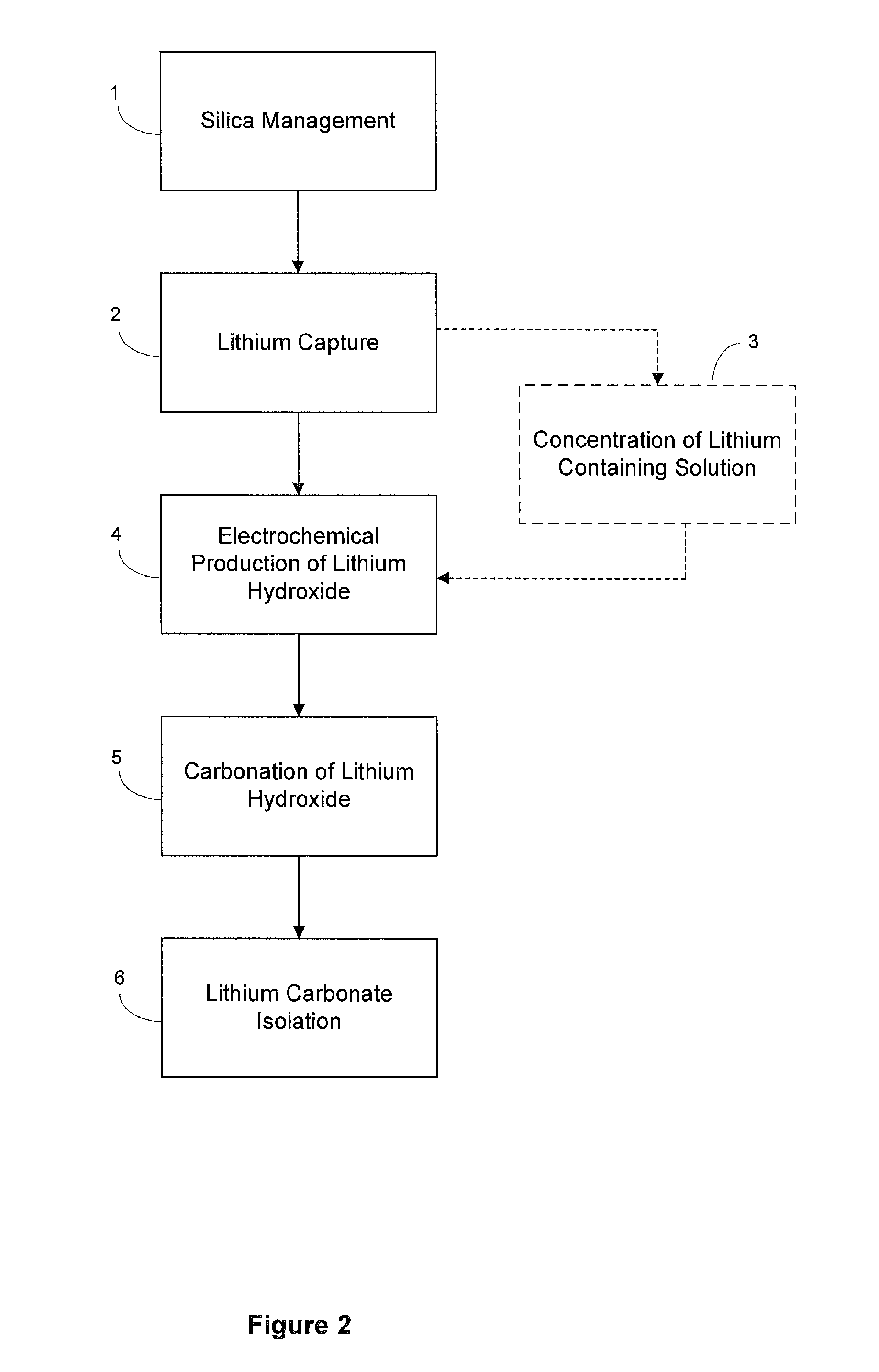
Preparation of lithium carbonate from lithium chloride containing brines
ActiveUS9034294B1Chloride preparationSolid sorbent liquid separationLithium carbonateLithium hydroxide
This invention relates to a method for the preparation of lithium carbonate from lithium chloride containing brines. The method can include a silica removal step, capturing lithium chloride, recovering lithium chloride, supplying lithium chloride to an electrochemical cell and producing lithium hydroxide, contacting the lithium hydroxide with carbon dioxide to produce lithium carbonate.
Owner:TERRALITHIUM LLC
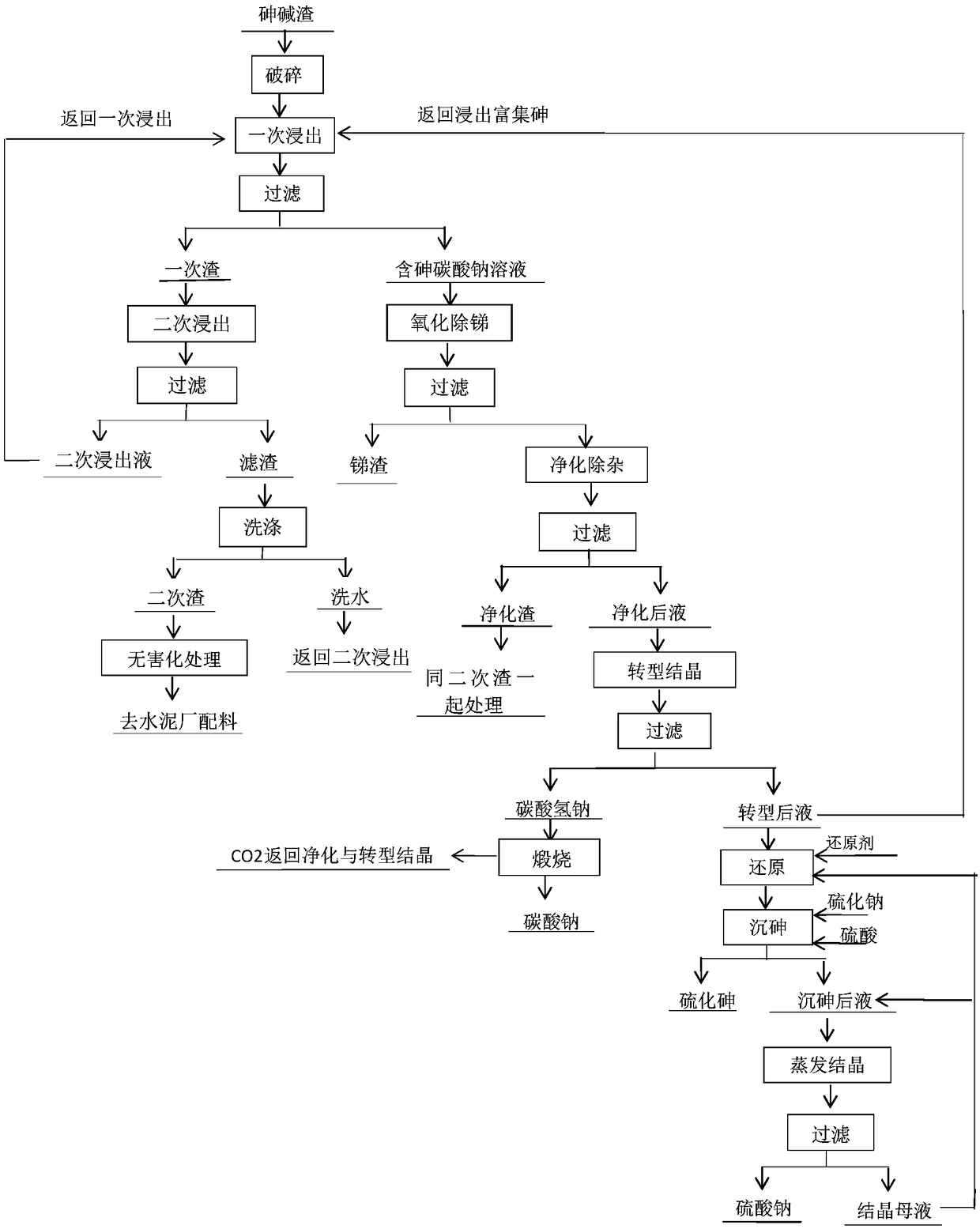
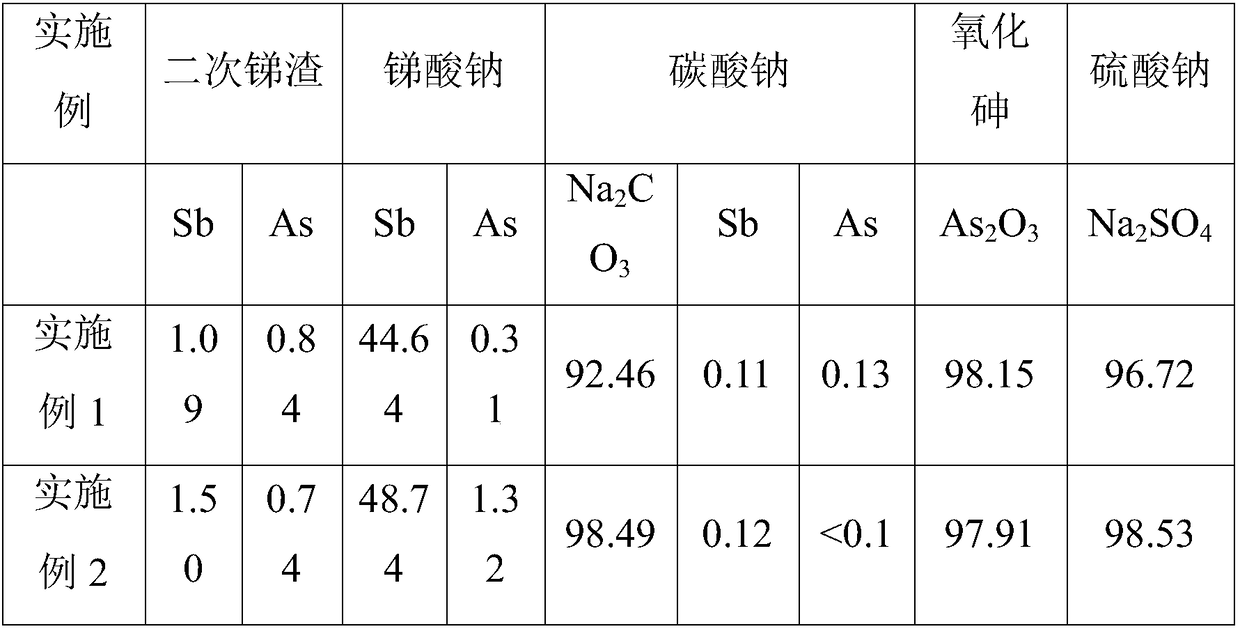
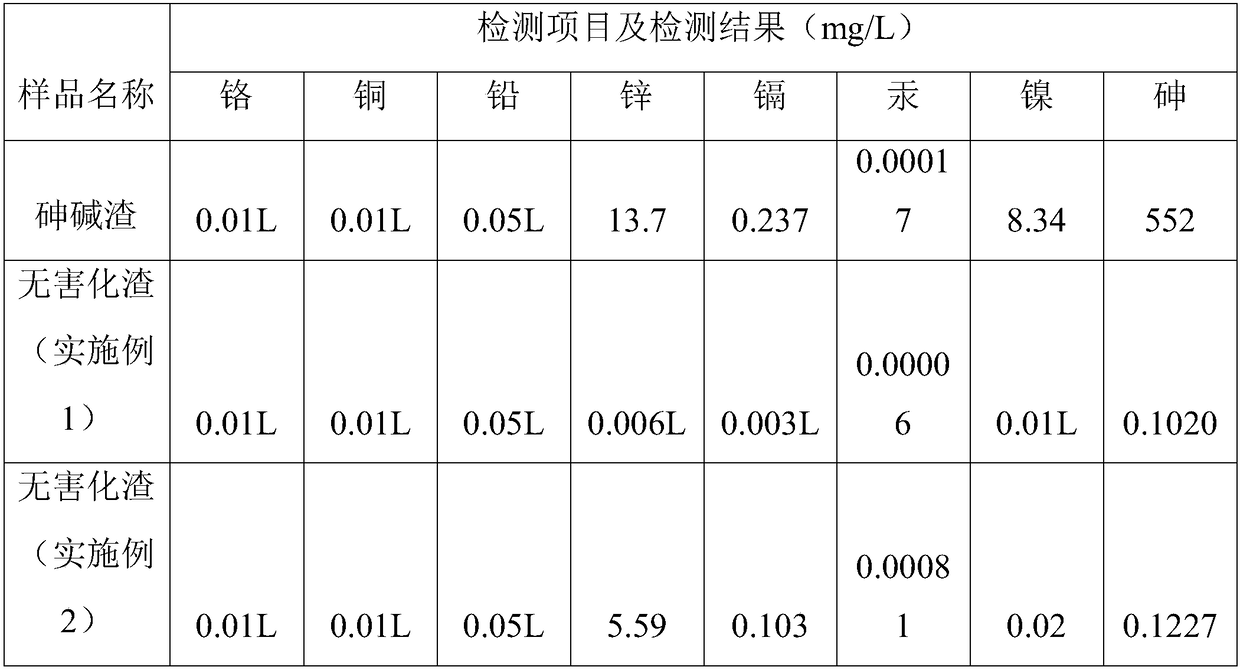
Wet-method recycling and harmless treatment process for antimony smelting arsenic alkali residue
PendingCN108441642ARealize green and clean cycle productionHigh recovery rateSolid waste disposalAlkali metal sulfite/sulfate purificationWastewaterImpurity
The invention belongs to the field of antimony smelting arsenic alkali residue treatment and particularly discloses a wet-method recycling and harmless treatment process for antimony smelting arsenicalkali residue. The process adopts a whole-wet-method treatment process and mainly comprises the eight working procedures of breaking, dissolving for leaching, oxidation for antimony outlet, purification for impurity removing, transition for crystallizing, reduction for arsenic sinking, evaporative crystallization and secondary residue harmless treatment. Recycling and harmless treatment of the arsenic alkali residue, zero emission of waste residue and waste water and up-to-standard discharge of exhaust gas are thoroughly achieved, the long-term plagued arsenic alkali residue environmental pollution problem in the antimony smelting industry in China is thoroughly solved, and the environmental risk caused by stacking the arsenic alkali residue is eliminated; and meanwhile, separation, enrichment and effective recovering of valuable elements are achieved.
Owner:CHENZHOU YANGTAO CHEM
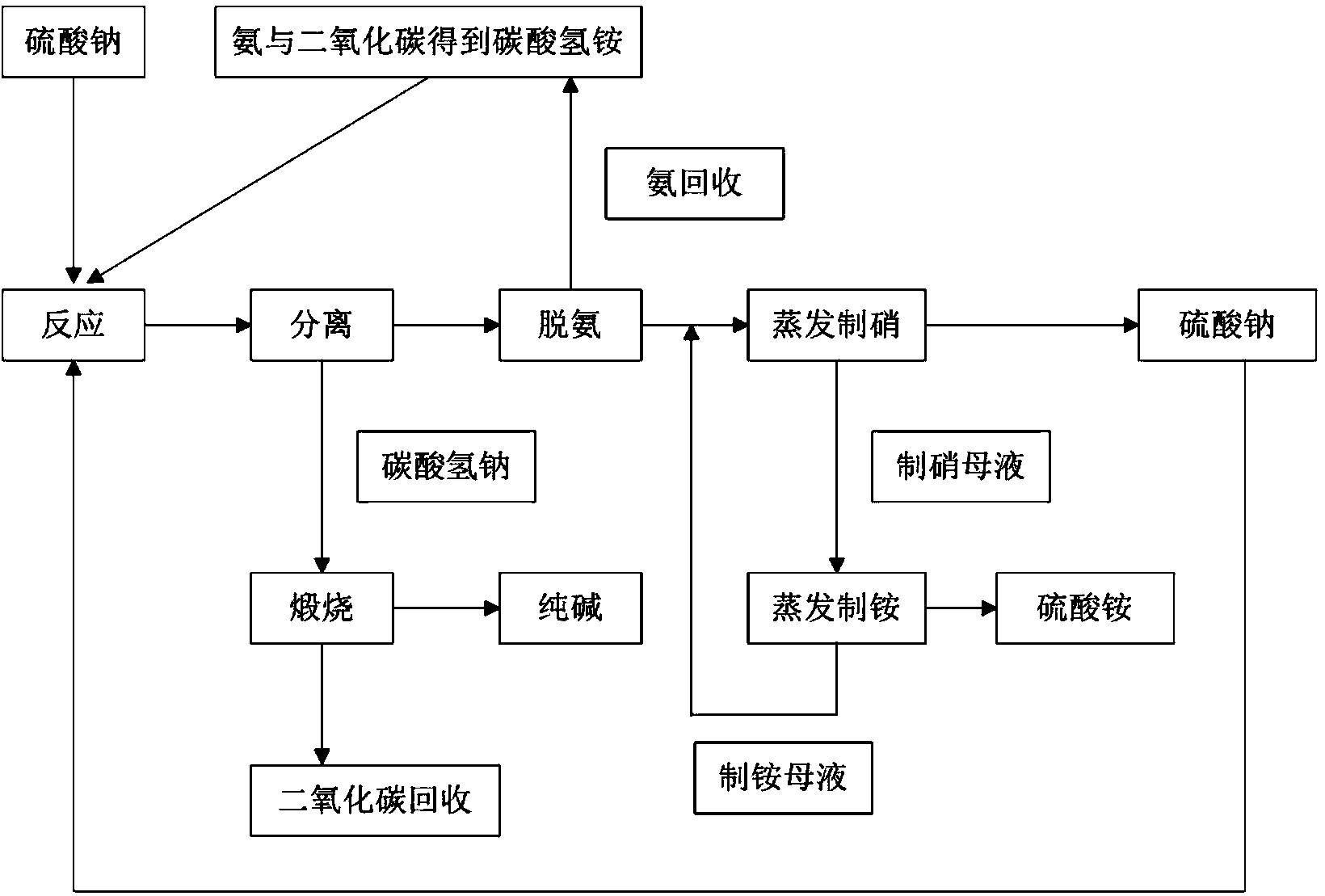
Thermal circulation technology of utilizing sodium sulfate solution or carrier to cogenerate sodium carbonate and ammonium sulfate
ActiveCN104355326AIncrease added valueReduce energy consumptionAmmonium sulfatesCarbonate preparationAmmonium Hydrogen CarbonateSodium sulfate
The invention provides a thermal circulation technology of utilizing sodium sulfate solution or a carrier to cogenerate sodium carbonate and ammonium sulfate. The thermal circulation technology comprises the following steps: regarding an ammonium hydrogen carbonate solution prepared from sodium sulfate, ammonia and carbon dioxide as a raw material to be conducted with a replacement reaction, then carrying out separation to obtain sodium hydrogen carbonate (which can be calcined to obtain a pure alkali product) and an alkali preparing mother solution containing ammonium sulfate, sodium sulfate, ammonium hydrogen carbonate and ammonium carbonate; preheating the alkali preparing mother solution to a high temperature to remove ammonium hydrogen carbonate and ammonium carbonate, and then obtaining the deaminized mother solution containing ammonium sulfate and sodium sulfate; evaporating the deaminized mother solution at a high temperature to obtain sodium sulfate and a niter preparing mother solution; evaporating the niter preparing mother solution at a low temperature to obtain ammonium sulfate and an ammonium preparing mother solution; circulating the sodium sulfate to the replacement reaction working procedure; circulating the ammonium preparing mother solution to the deaminized mother solution high-temperature evaporation working procedure. The main products and byproducts prepared by thermal circulation technology disclosed by the invention are high in quality and added value; the thermal circulation technology is well-adapted in raw materials, low in sodium sulfate theoretical use ratio, production cost and energy consumption, closed circulating, free of three-waste discharge, and capable of saving energy and reducing emission.
Owner:CHINA LIGHT IND INT ENG CO LTD
Method for high resource treatment of cyclohexanone waste saponification lye
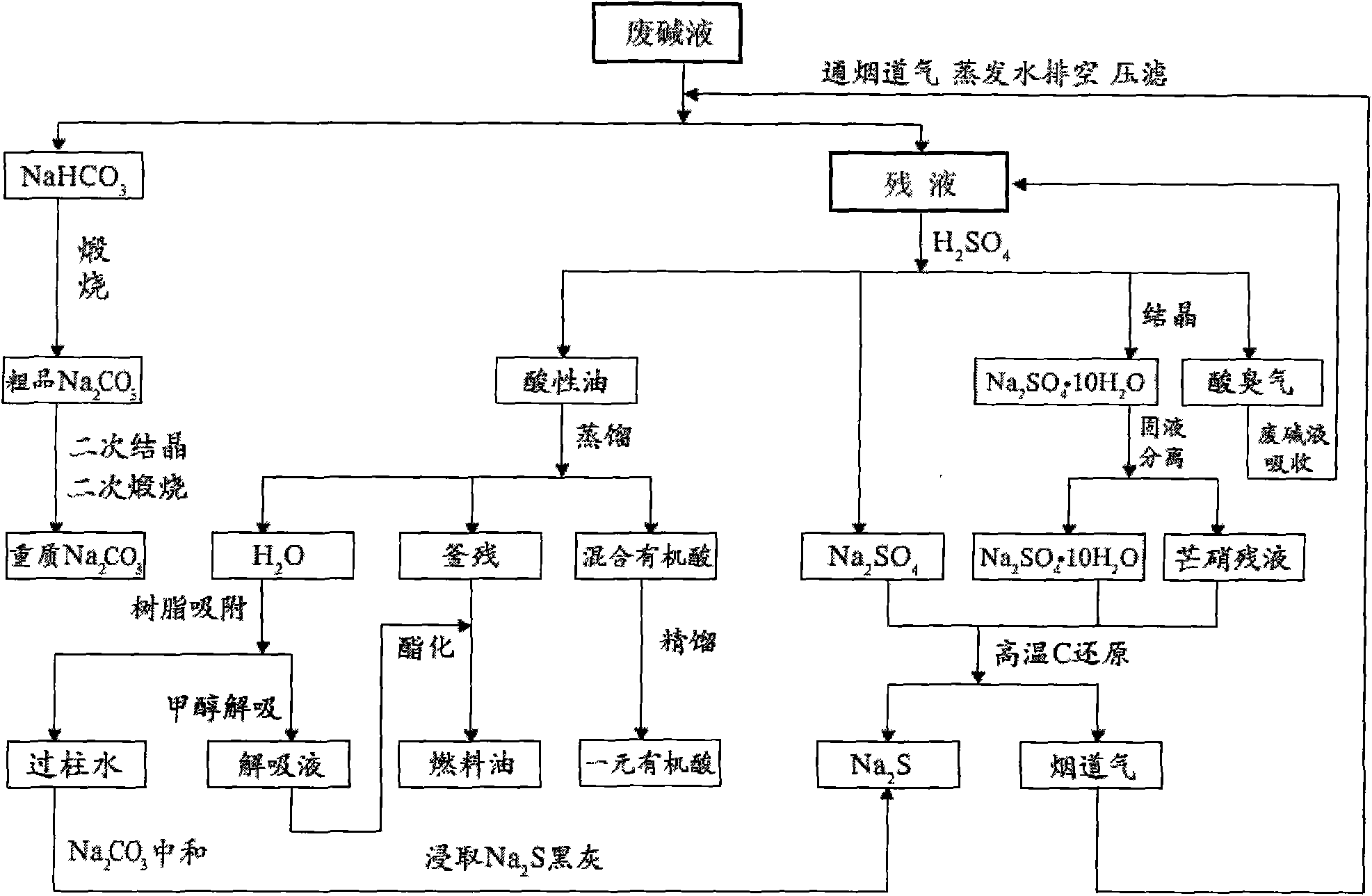
InactiveCN101570370AIncrease added valueEradicate pollutionCarbon compoundsSulfate/bisulfate preparationSodium bicarbonateCyclohexanone
The invention relates to a method for high resource treatment of cyclohexanone waste saponification lye. The method is improvement on a method for performing zero emission treatment on waste saponification lye produced by producing cyclohexanone through cyclohexane oxidation technology. By using the method, a process is set that sodium sulfate and pulverized coal are calcined to obtain sodium sulfide and carbon dioxide flue gas on the basis of the prior art, carbon dioxide in flue gas reacts with sodium hydroxide and sodium carbonate in initial waste lye to generate sodium hydrogencarbonate, and partial moisture is evaporated in the waste lye by high-temperature heat in the flue gas. The method utilizes the joint production of the cyclohexanone waste saponification lye to obtain chemical resources with higher level such as sodium carbonate, sodium sulfide, monoprotic organic acid, clean fuel oil and the like, and has more remarkable economic benefit; the method achieves the zero emission through the treatment of a waste with another waste, completely roots up the pollution of the waste lye on the environment, and has remarkable social benefit; and waste resources are circularly utilized, so the method has low energy consumption and mature process, is suitable for the large-scale treatment on the cyclohexanone waste saponification lye, and is also used for industrialized production of the chemical resources in the waste lye.
Owner:赵志军
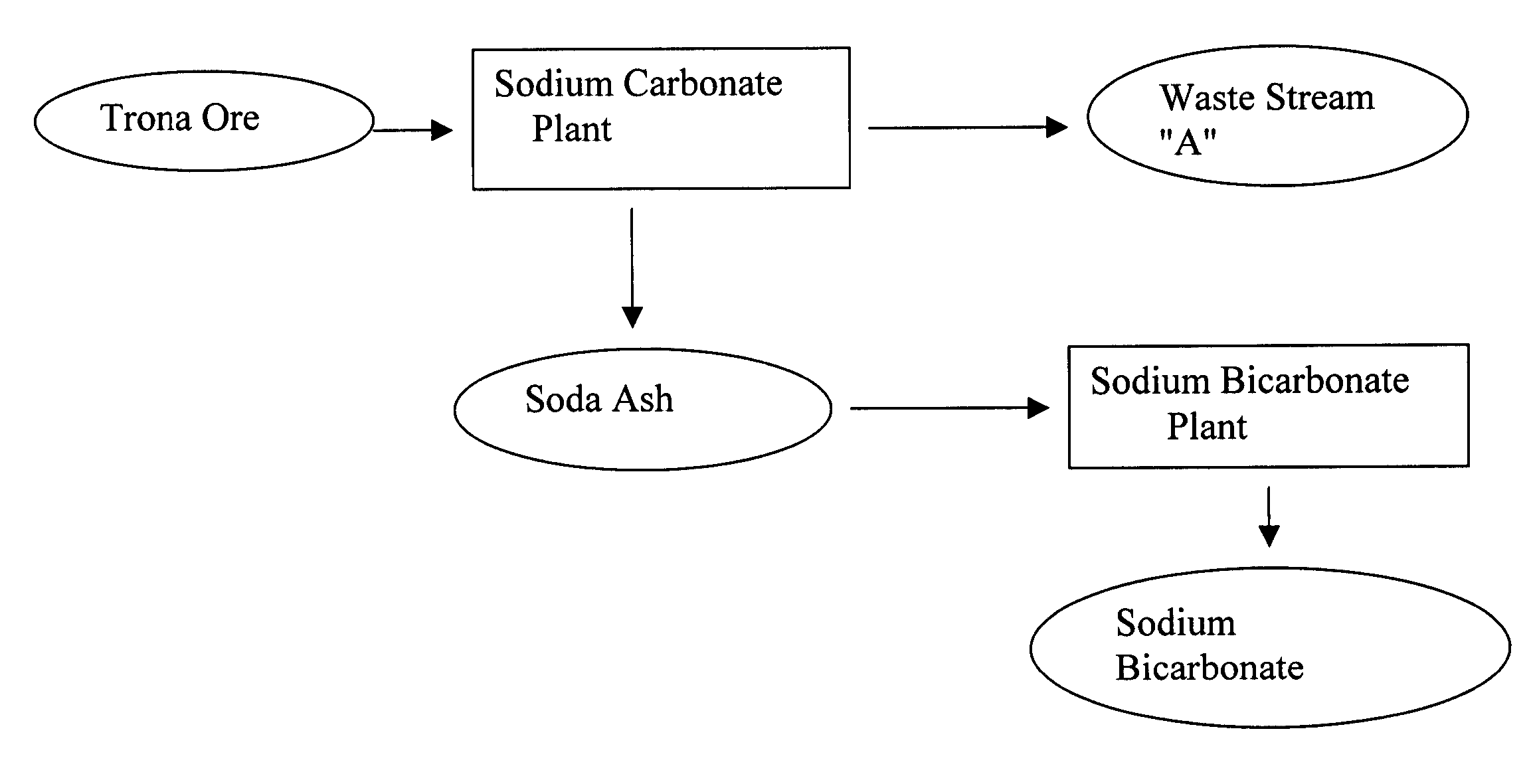
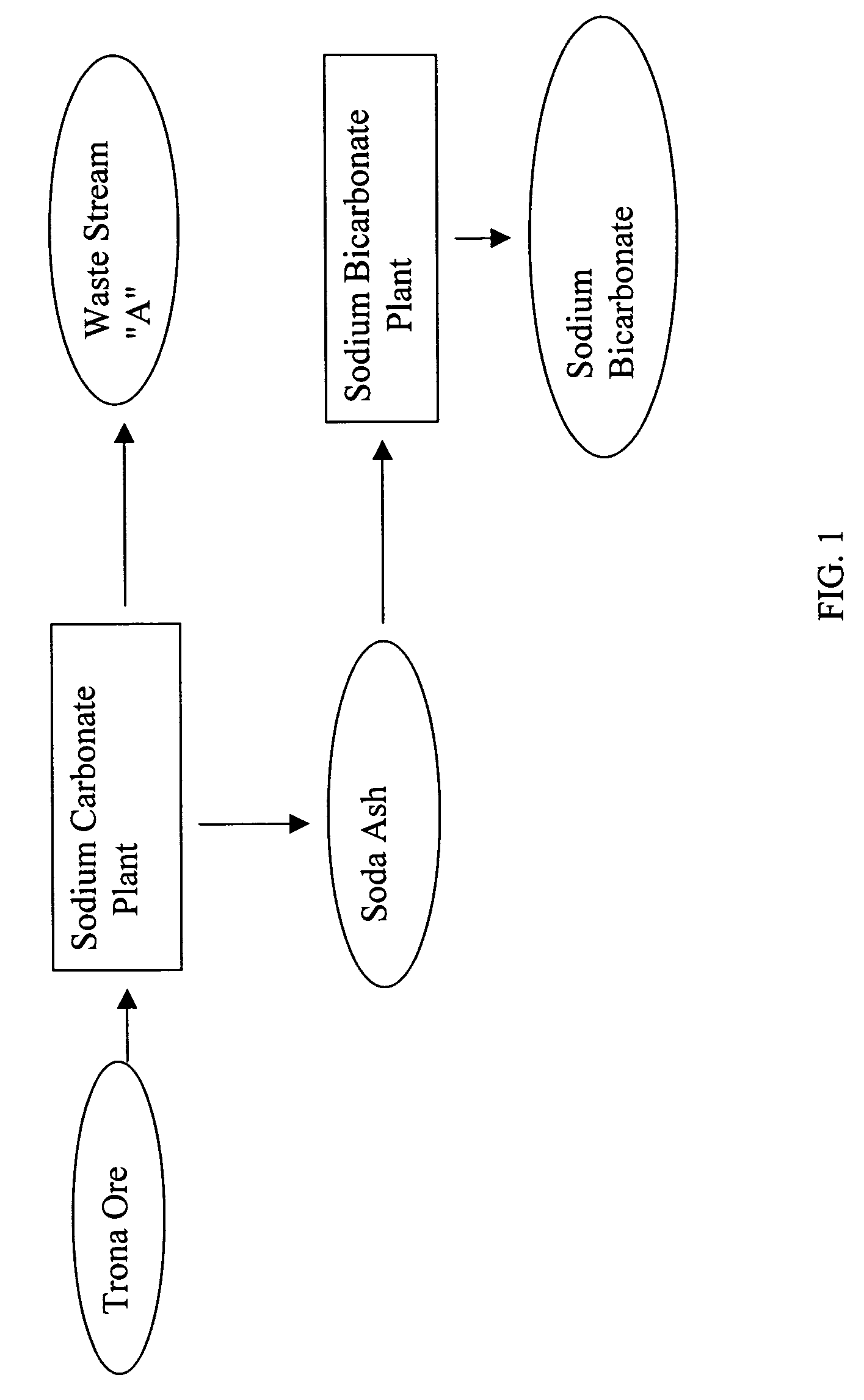
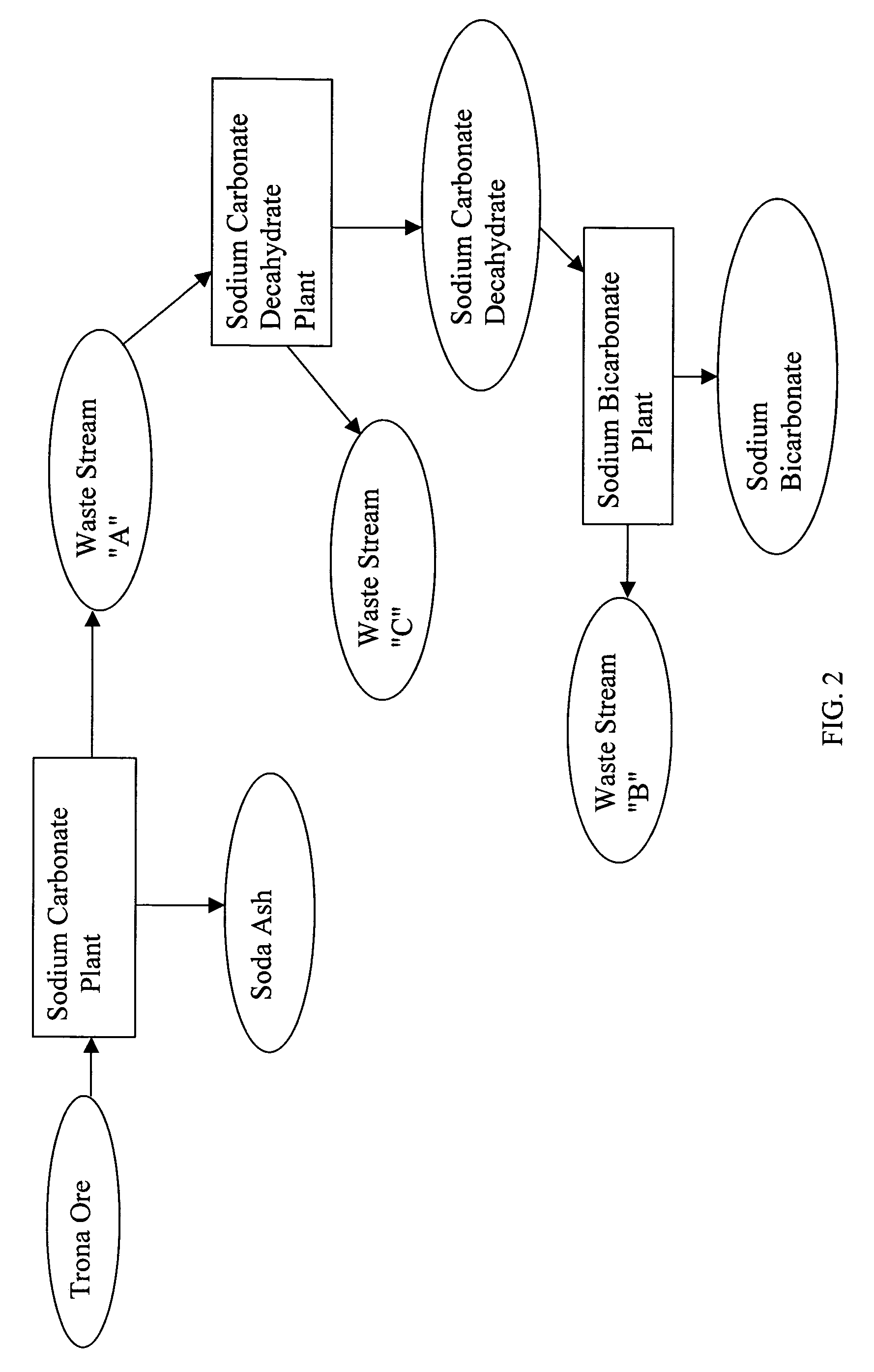
Sodium bicarbonate production method
InactiveUS20040057892A1Improvement in recovered sodium valuePromote recoveryBicarbonate preparationVarying alkali metal carbonate water contentWastewaterEngineering
A method of producing sodium bicarbonate having a high degree of purity and obtaining a net reduction in effluent waste water, as compared to prior processes, when starting from trona ore is disclosed. The process entails utilizing the waste-water effluent stream from the conversion of trona ore to sodium carbonate as the feed for the conversion of sodium carbonate to sodium bicarbonate.
Owner:CHURCH & DWIGHT CO INC
Method for preparing soda ash by converting sodium sulfate in low-grade rock salt or glauber salt ores
The invention discloses a method for mining low-grade rock salt ores and / or low-grade glauber salt ores by utilizing an alkali production waste liquor byproduct of an ammonia-alkali process and converting sodium sulfate in the ores into sodium chloride for alkali production. The method comprises the following steps of injecting the alkali production waste liquor into an underground cavity of the ores, performing chemical reaction on calcium chloride in the alkali production waste liquor and the sodium sulfate in the low-grade rock salt ores and / or the low-grade glauber salt ores to convert the sodium sulfate into sodium chloride required by alkali production, and mining the sodium chloride as brine for alkali production, precipitating and storing calcium sulfate generated by the reaction in the underground cavity, wherein the low-grade rock salt ores are low-grade rock salt ore resources abandoned by the salt and glauber salt co-production process, and every liter of brine mined by injecting fresh water into a mine of the ores contains 30 to 100 grams of Na2SO4 and 290 to 220 grams of NaCl; the low-grade glauber salt ores are low-grade glauber salt ore resources abandoned by the salt and glauber salt co-production process, and every liter of aqueous glauber salt mined by injecting the fresh water into a mine of the ores contains 200 to 100 grams of Na2SO4 and 120 to 220 grams of NaCl; every liter of alkali production waste liquor contains 95 to 115 grams of CaCl2 and 45 to 56 grams of NaCl.
Owner:江苏苏盐井神股份有限公司 +1
Methods for constructing underground borehole configurations and related solution mining methods
Disclosed are methods for solution mining of evaporite minerals, such as trona, comprising drilling an access well and at least two lateral boreholes; injecting a fluid; circulating the fluid through the lateral boreholes with a controlled fluid flow; and collecting a pregnant solution. Also disclosed are methods of solution mining that include injecting an aqueous solution into an underground trona cavity at a temperature sufficient to maintain at least a portion of the solution in the cavity in the Wegscheiderite solid phase region; removing aqueous solution from the cavity; and recovering alkaline values from the removed aqueous solution. Also disclosed are methods of solution mining that include injecting an aqueous solution into an underground trona cavity; removing aqueous solution from the cavity, wherein the temperature of the removed aqueous solution is at about the TWA point temperature; and recovering alkaline values from the removed aqueous solution.
Owner:SESQUI MINING
Technology for producing sodium bicarbonate from mirabilite
InactiveCN105712382AIncrease profitEmission reductionCalcium/strontium/barium sulfatesCarbonate preparationSodium bicarbonateDistillation
A technology for producing sodium bicarbonate from mirabilite comprises steps as follows: mirabilite and distillation waste liquor mixed liquor is used as a raw material, sodium carbonate is added to the solution after absorption of ammonia, magnesium, iron and calcium ions in the solution are removed after the solution is subjected to a precipitation reaction at 15-50 DEG C, and a refined solution is obtained; carbon dioxide is added to the refined solution for carbonatation, and sodium bicarbonate solids and heavy alkali mother liquor are obtained through separation after the refined solution is subjected to a replacement reaction at 15-50 DEG C; lime is added to the heavy alkali mother liquor, and ammonia gas, carbon dioxide, calcium sulfate and distillation waste liquor are obtained after distillation reaction at 15-120 DEG C; ammonia gas and carbon dioxide are recovered to ammonia absorption and carbonatation procedures respectively; the distillation waste liquor and mirabilite in Step A are mixed to be recycled. The technology has the characteristics that mirabilite is directly utilized, ammonia is recycled, the comprehensive utilization rate of distillation waste liquor waste is high, the production cost is low, the waste residue discharge is low, the technology is environment-friendly, and the like.
Owner:CHINA LIGHT IND INT ENG CO LTD
Technology for combined production of sodium carbonate and ammonium chloride through sodium sulfate type brine thermal cycle method
InactiveCN105000579AAdaptableThe main product is of high qualityAmmonium halidesCarbonate preparationSodium bicarbonateSodium sulfate
The invention discloses a technology for combined production of sodium carbonate and ammonium chloride through a sodium sulfate type brine thermal cycle method. The technology comprises the following steps: (1) carrying out a metathesis reaction on sodium sulfate type brine, ammonia and carbon dioxide as raw materials, separating to obtain sodium bicarbonate and an alkali production mother liquor containing ammonium chloride, sodium sulfate, sodium chloride, ammonium bicarbonate and ammonium carbonate, and calcining the obtained sodium bicarbonate to obtain a sodium carbonate product; (2) preheating the alkali production mother liquor to carry out high temperature removal of ammonium bicarbonate and ammonium carbonate in order to obtain a deaminized mother liquor containing ammonium chloride, sodium sulfate and sodium chloride; (3) adding lime with the amount equal to that of sodium sulfate to the deaminized mother liquor, and reacting to remove sodium sulfate in order to obtain calcium sulfate, ammonia and a denitrified mother liquor containing ammonium chloride and sodium chloride; and (4) evaporating the denitrified mother liquor, and separating to obtain ammonium chloride and an ammonium production mother liquor. The technology has the characteristics of high quality of main products, strong adaptability of the raw materials, low cost, low energy consumption, closed loop, and no discharge of three wastes.
Owner:CHINA LIGHT IND INT ENG CO LTD
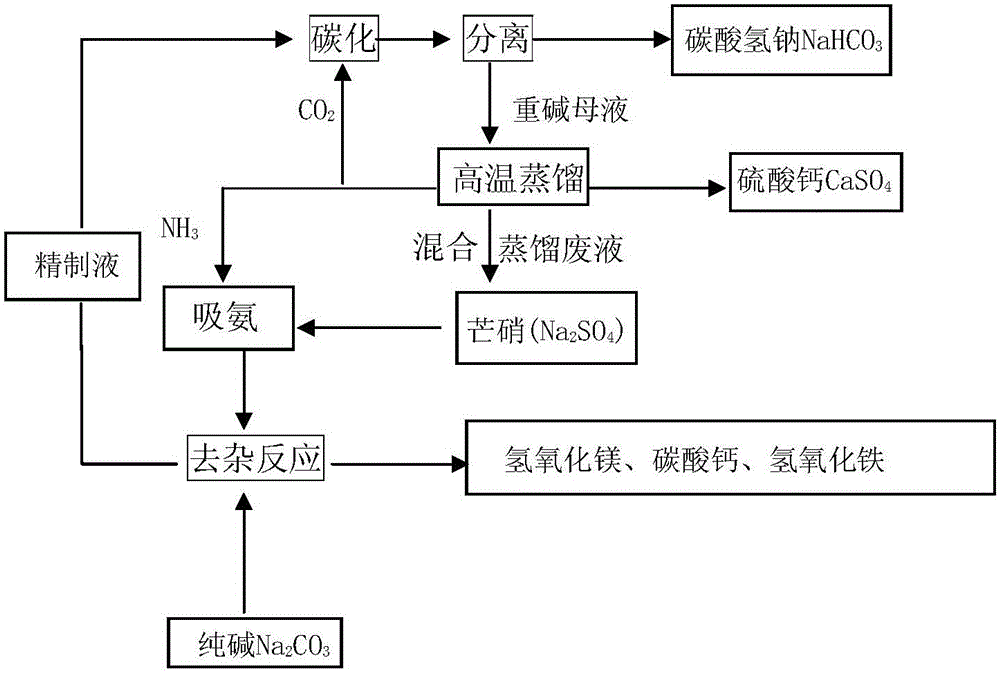
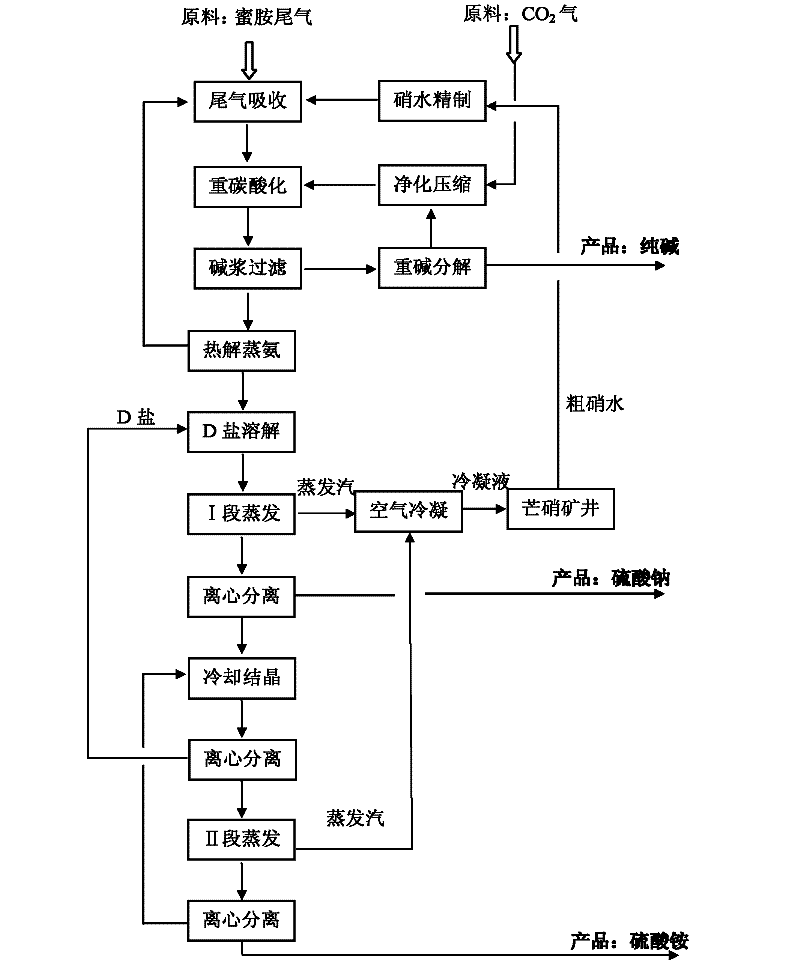
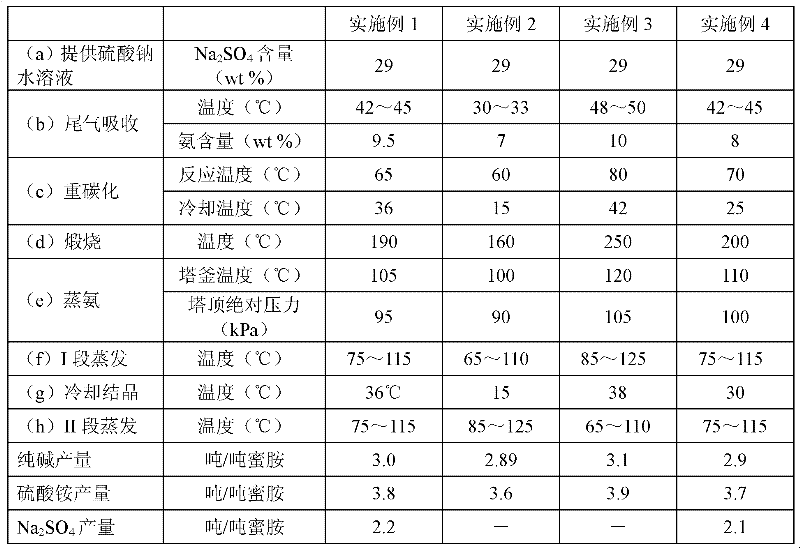
Methods for coproducing sodium carbonate and ammonium sulfate from melamine tail gas and mirabilite
InactiveCN102198953AIncrease profitReduce consumptionAmmonium sulfatesCarbonate preparationSodium bicarbonateSlurry
The invention relates to methods for coproducing sodium carbonate and ammonium sulfate from melamine tail gas and mirabilite. The method for producing the sodium carbonate comprises the following steps of: a, providing a sodium sulfate water solution; b, introducing melamine tail gas into the sodium sulfate water solution to form an ammonium carbonate-sodium sulfate water solution; c, introducing carbon dioxide into the ammonium carbonate-sodium sulfate water solution to obtain a suspension serous fluid of sodium bicarbonate crystals; and d, carrying out solid and liquid separation on the suspension serous fluid to obtain sodium bicarbonate and a mother solution I, calcining sodium bicarbonate solid to obtain the sodium carbonate. The method further comprises the following steps of: e, adding the mother solution I into an ammonia stilling tower, heating, forming tower top mixed gas containing ammonia and carbon dioxide, generating a deamination solution in the tower; f, evaporating the deamination solution for dewatering to obtain sodium sulfate crystals and a mother solution II; g, cooling the mother solution II for crystallizing to obtain Na2SO4.(NH4)2SO4.4H2O salt crystals and a mother solution III; and h, evaporating the mother solution III for dewatering, and separating out ammonium sulfate crystals. The methods have the advantages of high utilization rate of raw materials and low energy consumption.
Owner:SICHUAN GOLDEN ELEPHANT SINCERITY CHEM CO LTD +1
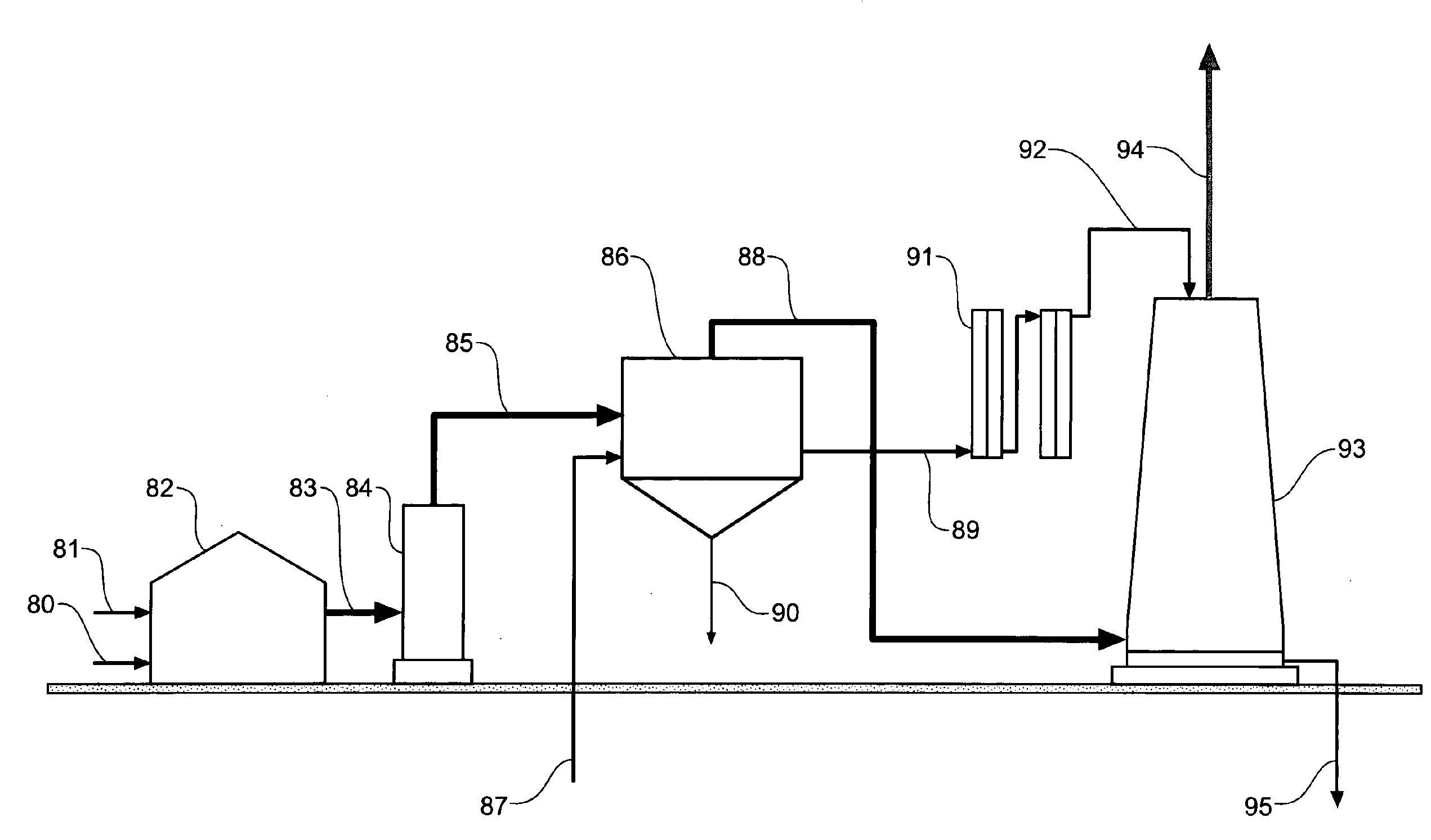
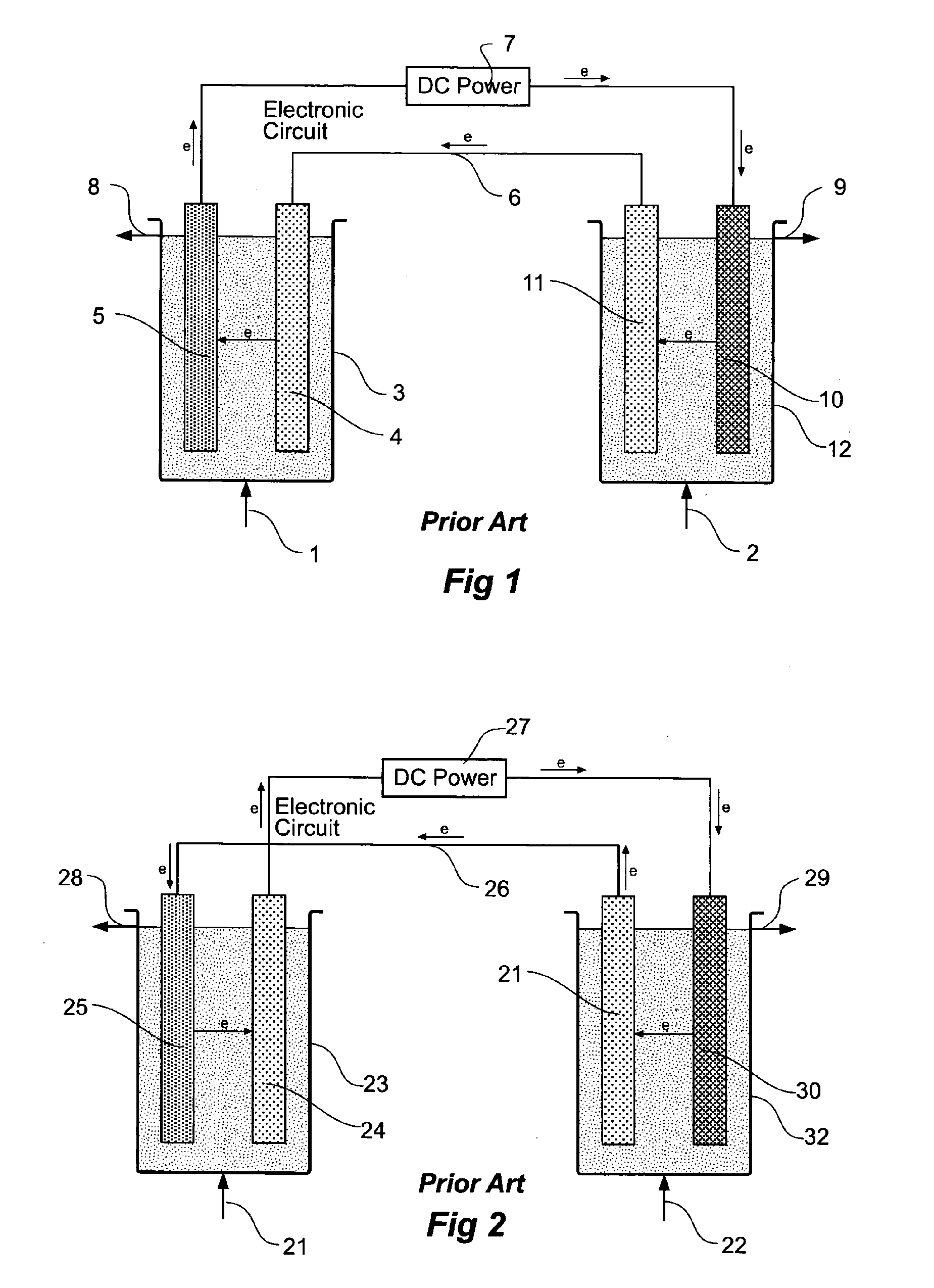
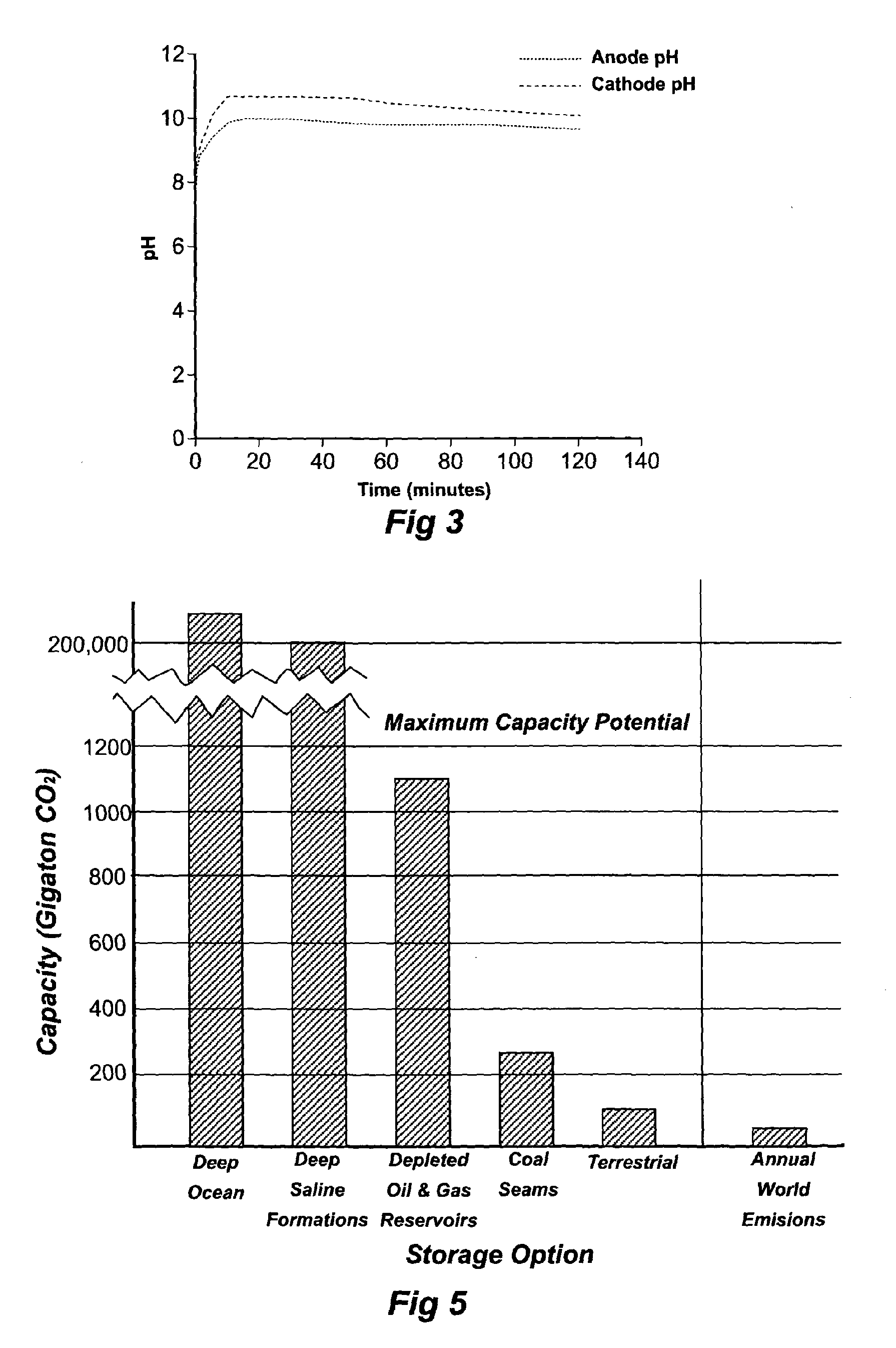
Carbon dioxide sequestration and capture
InactiveUS20100084283A1Calcium/strontium/barium carbonatesCellsCarbon dioxide productionAtmospheric air
A process to convert carbon dioxide into a stable substance with electrolytically activated seawater and use this process to sequester carbon dioxide from coal power plants (82) and similar carbon dioxide producing equipment, and capture and sequester carbon dioxide from the atmosphere. Electrolytically activated seawater (92) is produced using a unipolar electrolytic cell (91) and is sprayed into a contacting tower (93) or into the air.
Owner:GOMEZ RODOLFO ANTONIO M
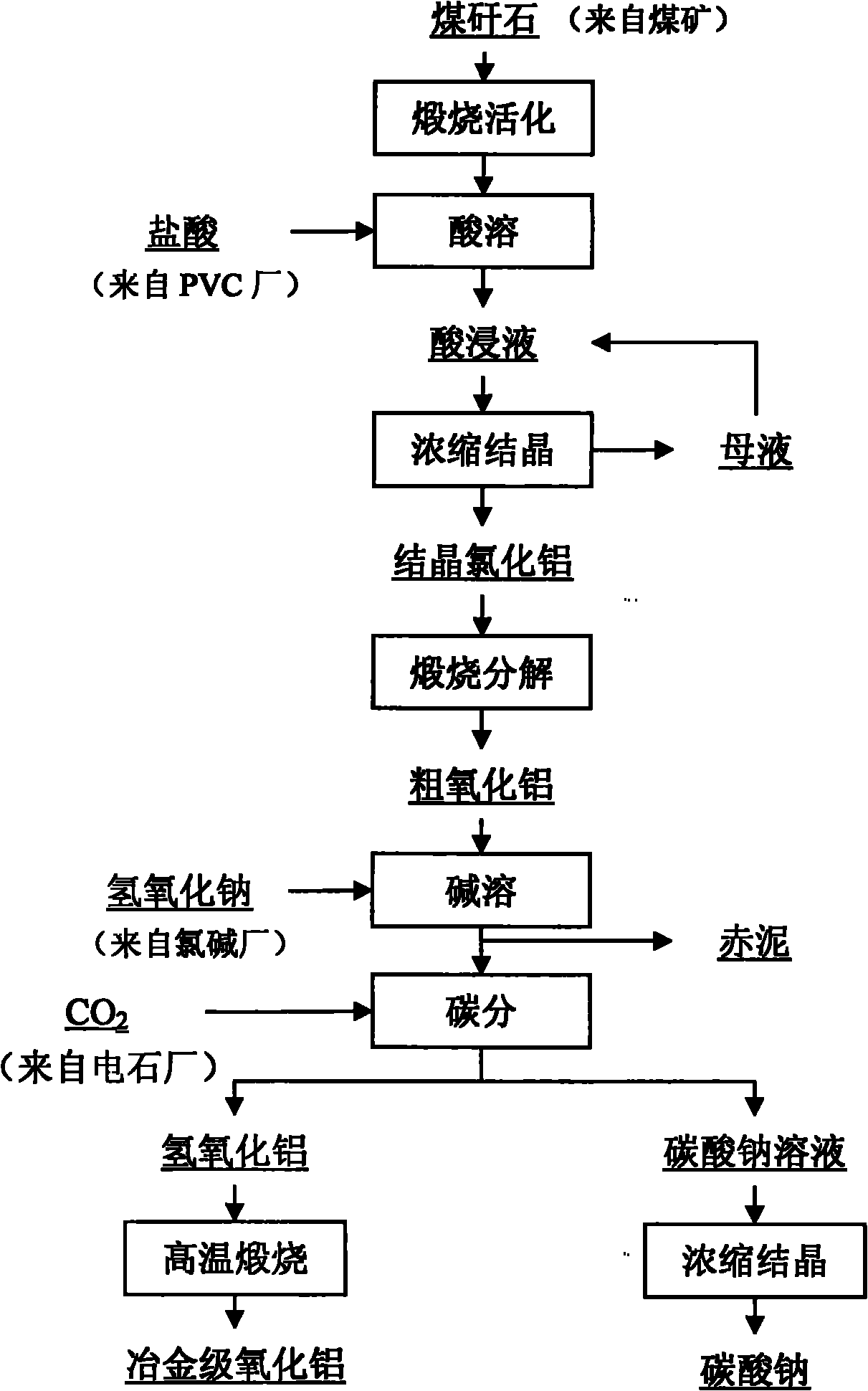
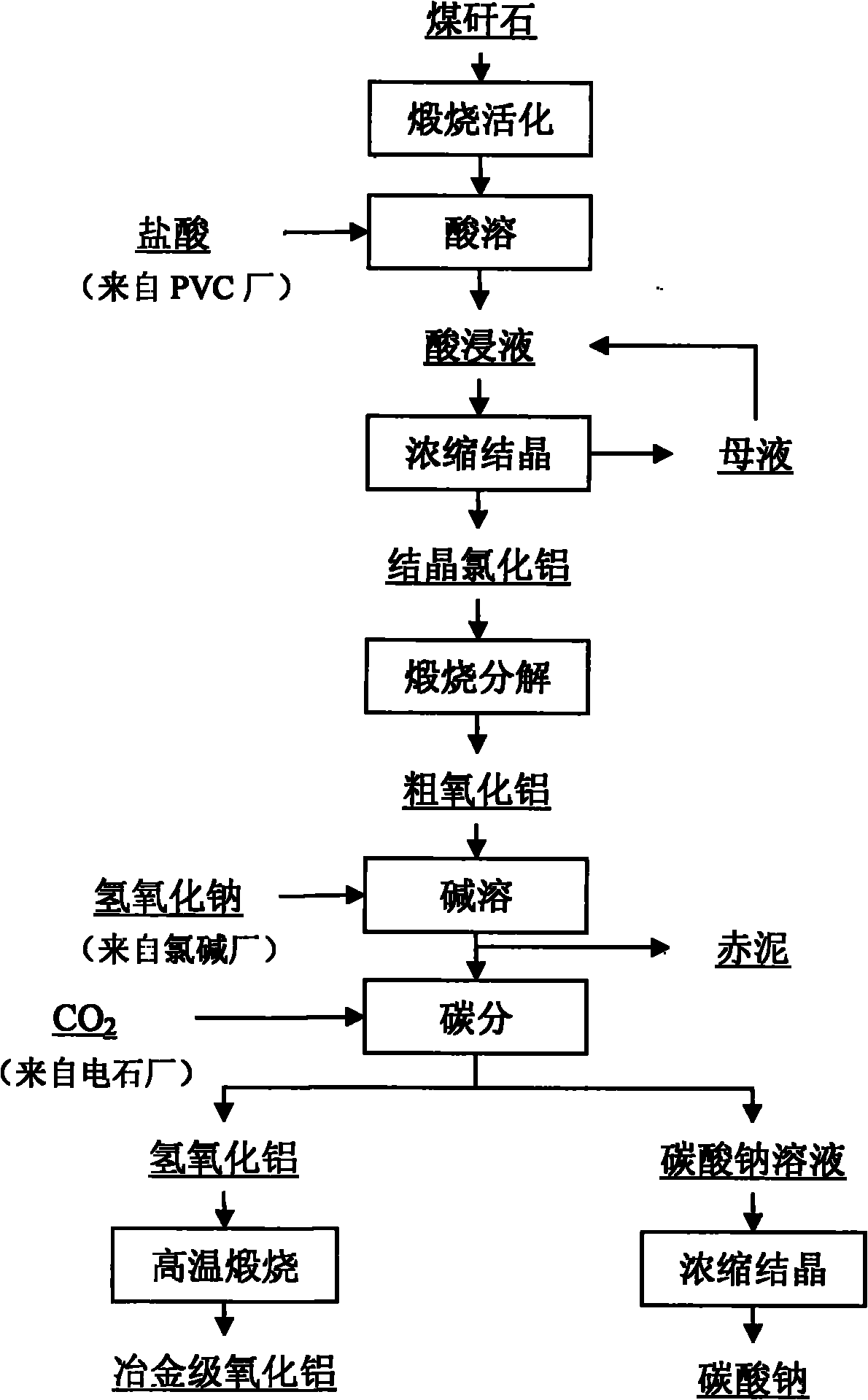
Method for co-producing aluminum oxide and sodium carbonate from coal gangue
InactiveCN101850998AHigh extraction rateReduce manufacturing costAluminium oxide/hydroxide preparationCarbonate preparationResource utilizationAluminium hydroxide
The invention relates to a method for co-producing aluminum oxide and sodium carbonate from coal gangue. The method comprises the following steps of: calcining and activating the coal gangue and dissolving the calcined and activated coal gangue in acid to prepare an aluminum chloride acid extract; concentrating and crystallizing the acid extract to prepare crystalline aluminum chloride; calcining and decomposing the crystalline aluminum chloride to prepare crude aluminum oxide; dissolving the crude aluminum oxide in alkali to prepare sodium metaaluminate; performing solid-liquid separation on iron and titanium insoluble substances and other impurities and washing the product to obtain solution of sodium metaaluminate; adding an aluminum hydroxide seed crystal into the solution of sodium metaaluminate to prepare aluminum hydroxide deposit and solution of sodium carbonate; performing solid-liquid separation to obtain an aluminum hydroxide crystal and solution of sodium carbonate; concentrating and crystallizing the aluminum hydroxide crystal and the solution of sodium carbonate to obtain sodium carbonate; and calcining the aluminum hydroxide to prepare metallurgical-grade aluminum oxide. In a production process, sodium hydroxide from a chlorine industry, hydrochloric acid which is a byproduct of a PVC industry and carbon dioxide discharged from a calcium carbide plant in a production place of the coal gangue are fully utilized, so that industry pollution is reduced, a useful product is obtained and comprehensive resource utilization is realized. The method has the advantages of simple production technology, easily-controlled production process, high aluminum oxide extraction ratio, low production cost and stable product quality.
Owner:CHANGCHUN CHAOWEI NEW MATERIALS TECH
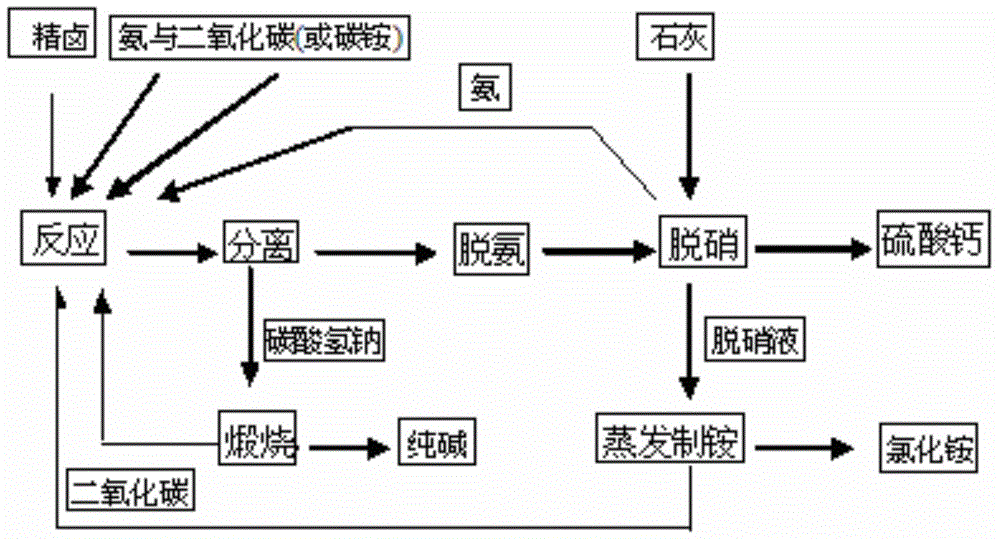
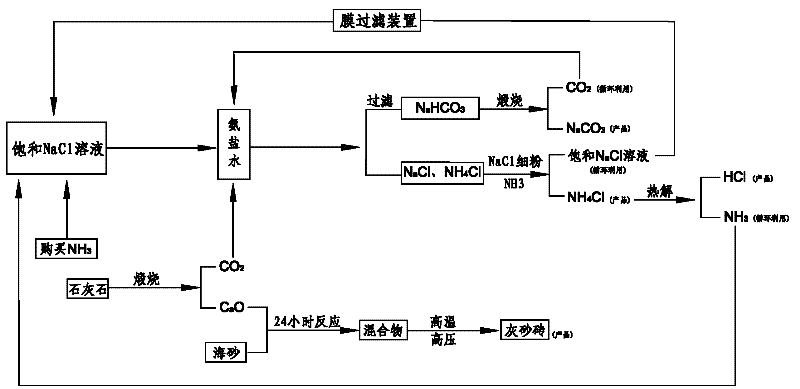
Comprehensive soda ash producing process and product application thereof
ActiveCN102531001AIncrease profitReduce manufacturing costCalcium/strontium/barium nitratesAmmonium halidesSodium bicarbonateProcess engineering
The invention relates to the technical field of soda ash production, and especially relates to a comprehensive soda ash producing process and product applications thereof; the process comprises the following process steps: introducing ammonia gas into a saturated sodium chloride solution to prepare ammoniacal brine; introducing carbon dioxide generated in limestone calcination into the ammoniacal brine to generate a mixture of sodium bicarbonate, sodium chloride and ammonium chloride; filtering the mixture to obtain sodium bicarbonate, sodium chloride and ammonium chloride; calcining the sodium bicarbonate to generate soda ash and carbon dioxide, recycling the carbon dioxide; adding sodium chloride fine powder and ammonia gas into the mixed filtrate of ammonium chloride and sodium chloride to generate ammonium chloride and sodium chloride solutions; separating impurities from the sodium chloride solution by a membrane filter, recycling the sodium chloride solution. The process has the advantages of low production cost, high soda ash whiteness, high sodium chloride utilization rate, no water waste or solid waste generation, environment protection, safety, simple processes, full use of geographic advantages in our country, low raw material purchase cost, waste change into things of value, high economic benefits, and good social benefits.
Owner:GUANGDONG DAZHONG AGRI SCI CO LTD
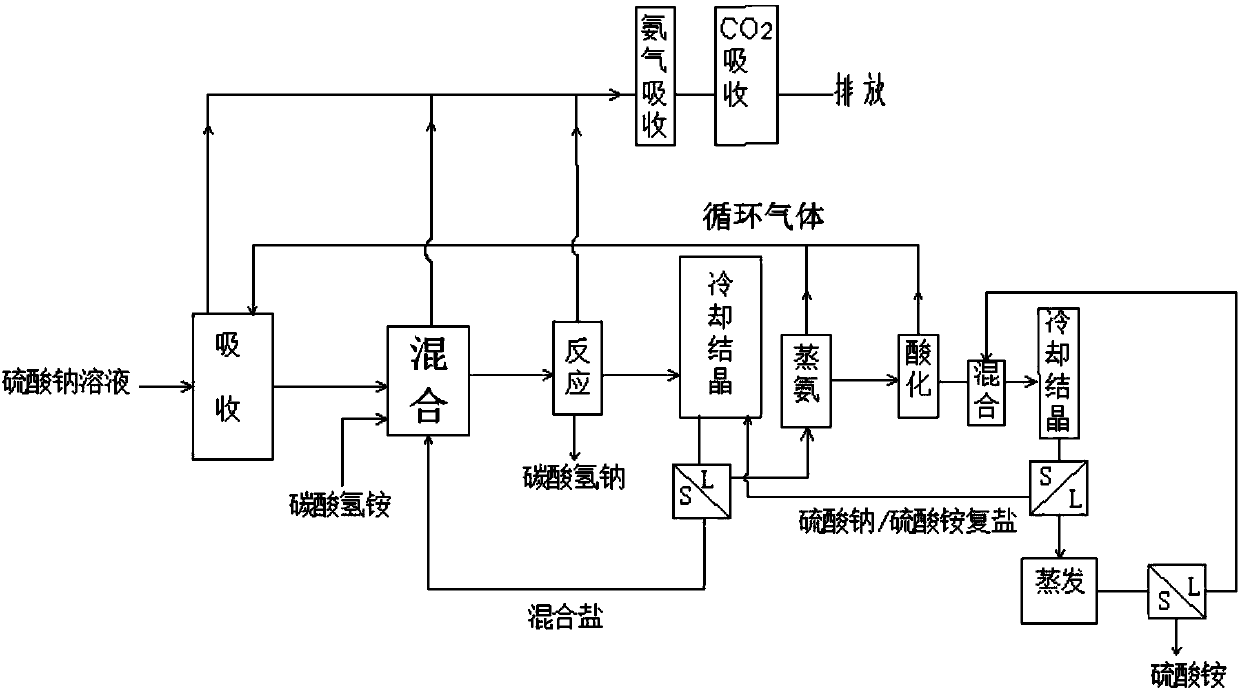
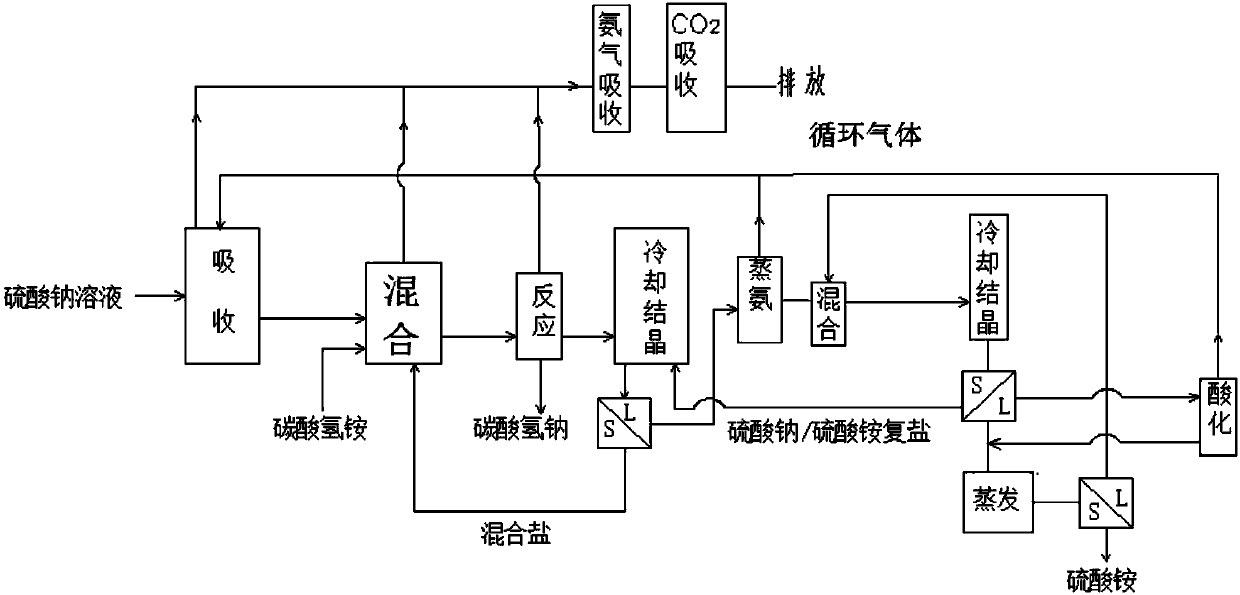
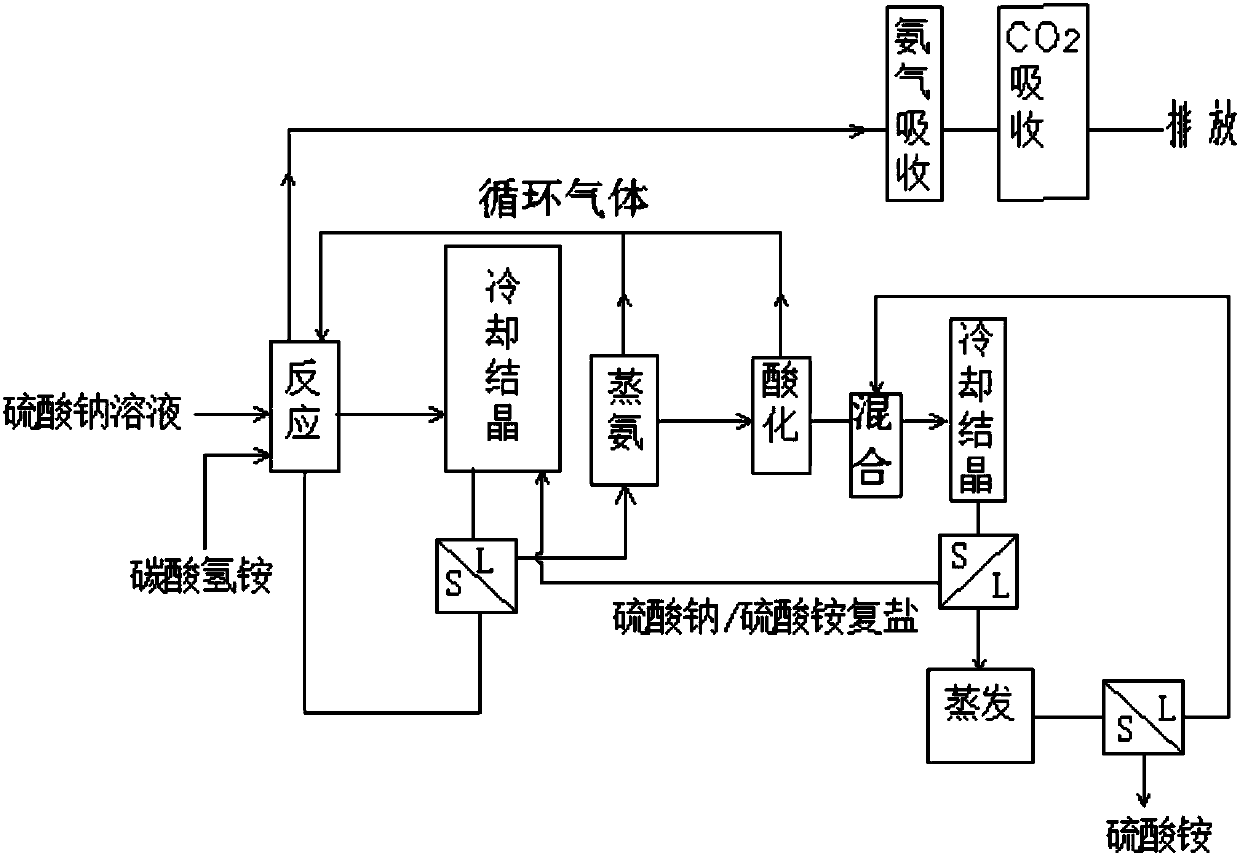
Method for producing sodium hydrogen carbonate and ammonium sulfate by using sodium sulfate solution
ActiveCN108046295ASolve pollutionImprove absorption rateCarbonate preparationAmmonia compoundsSodium bicarbonateHydrogen
The invention relates to a method for producing sodium hydrogen carbonate and ammonium sulfate by using a sodium sulfate solution, and belongs to the technical field of chemical engineering. The method comprises the following steps: a, absorbing circulating gas by using the sodium sulfate solution at -5 to 10 DEG C to obtain absorbing liquid; b, mixing the absorbing liquid, ammonium hydrogen carbonate and / or mixed salt at -5 to 20 DEG C to obtain mixed slurry; c, performing sealed reaction on the mixed slurry to obtain sodium hydrogen carbonate and reaction mother liquid; d, mixing the reaction mother liquid and complex salt to obtain salt mixture and primary frozen mother liquid after freezing; e, distilling the primary frozen mother liquid to obtain carbon dioxide, ammonium circulating gas and ammonium steaming mother liquid; f, adjusting the ammonium steaming mother liquid by acid to obtain an acidified solution and gas; g, mixing and freezing the acidified solution and crystallizedmother liquid to obtain complex salt and secondary frozen mother liquid; and h, evaporating and concentrating the secondary frozen mother liquid to separate out ammonium sulfate. The method is low inenvironmental pollution and high in raw material utilization rate.
Owner:SICHUAN UNIV
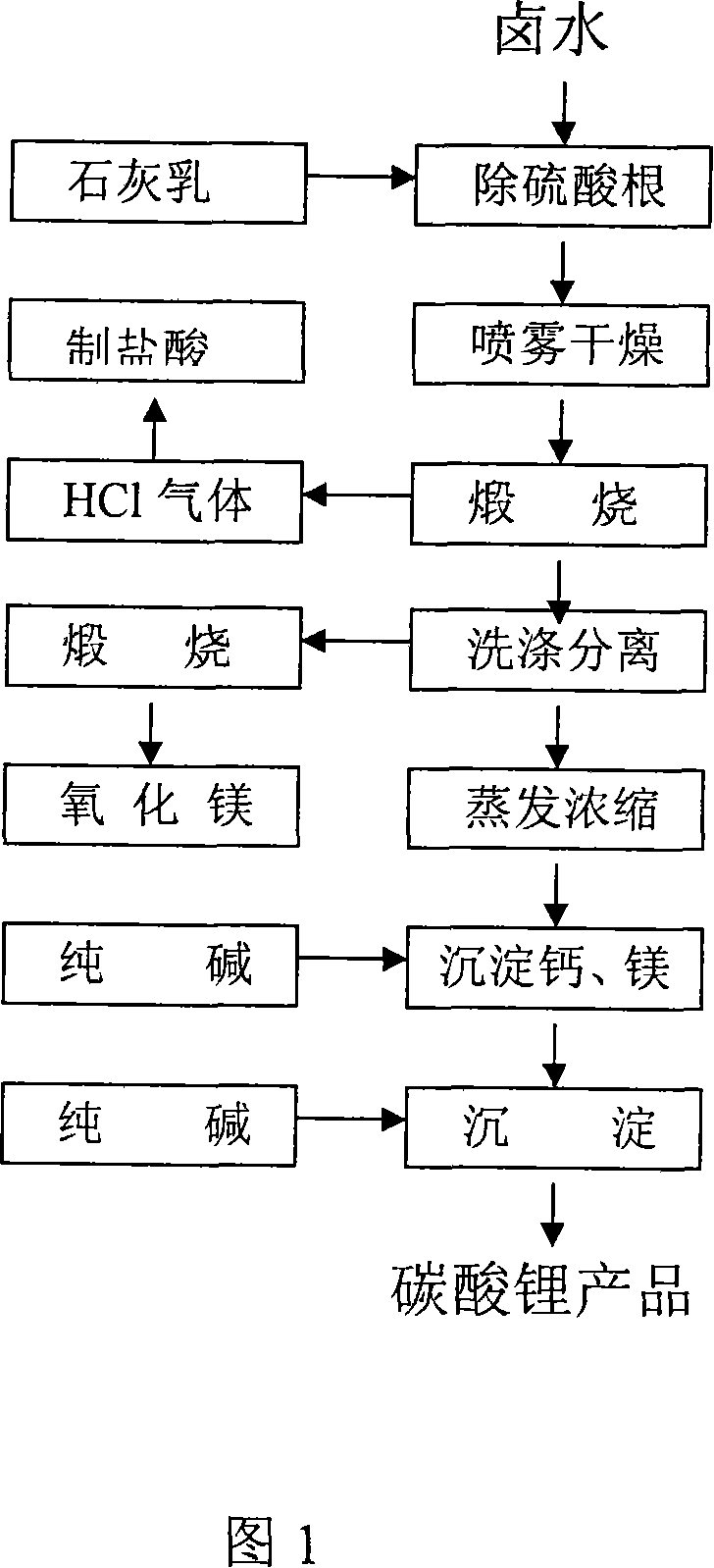
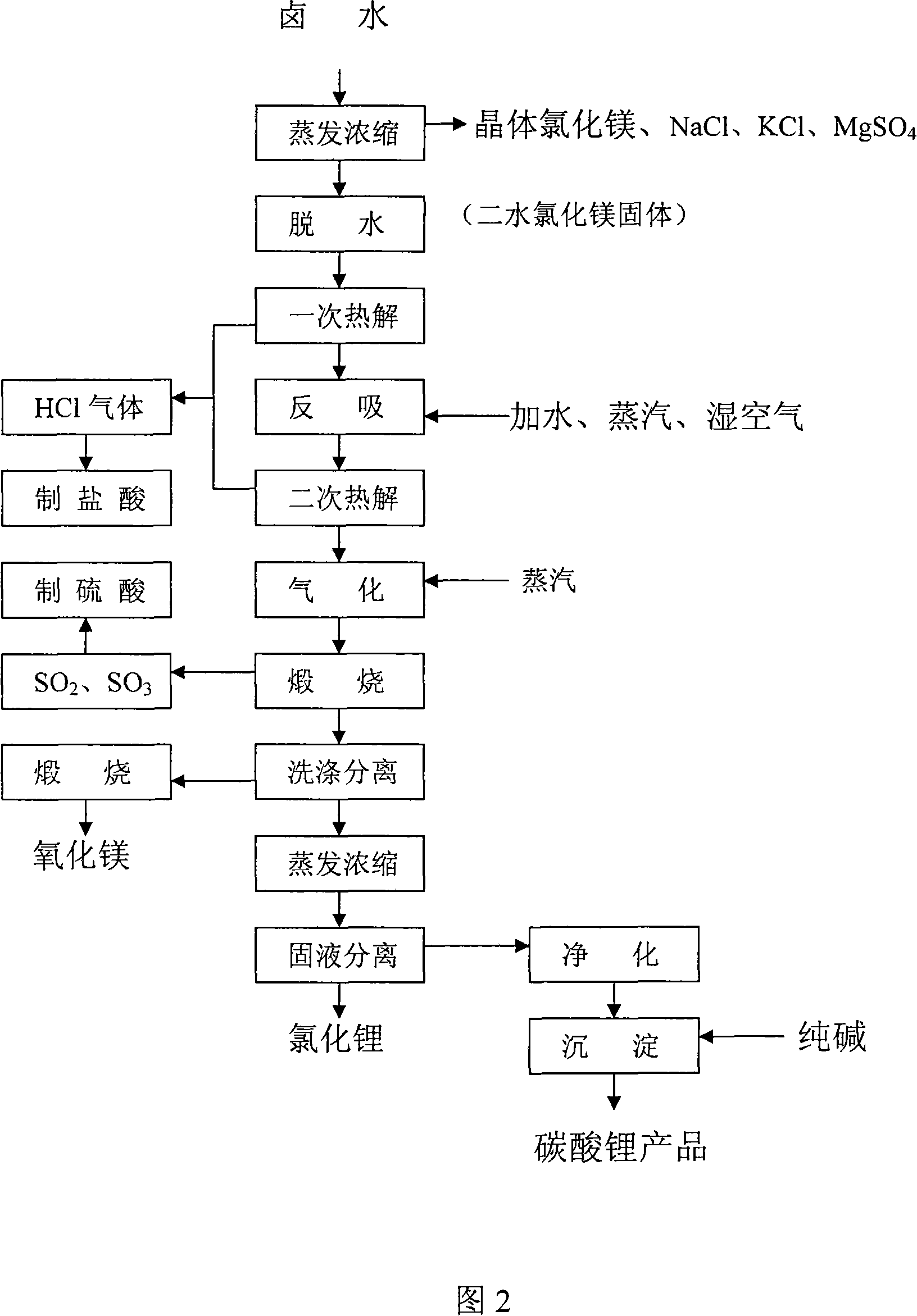
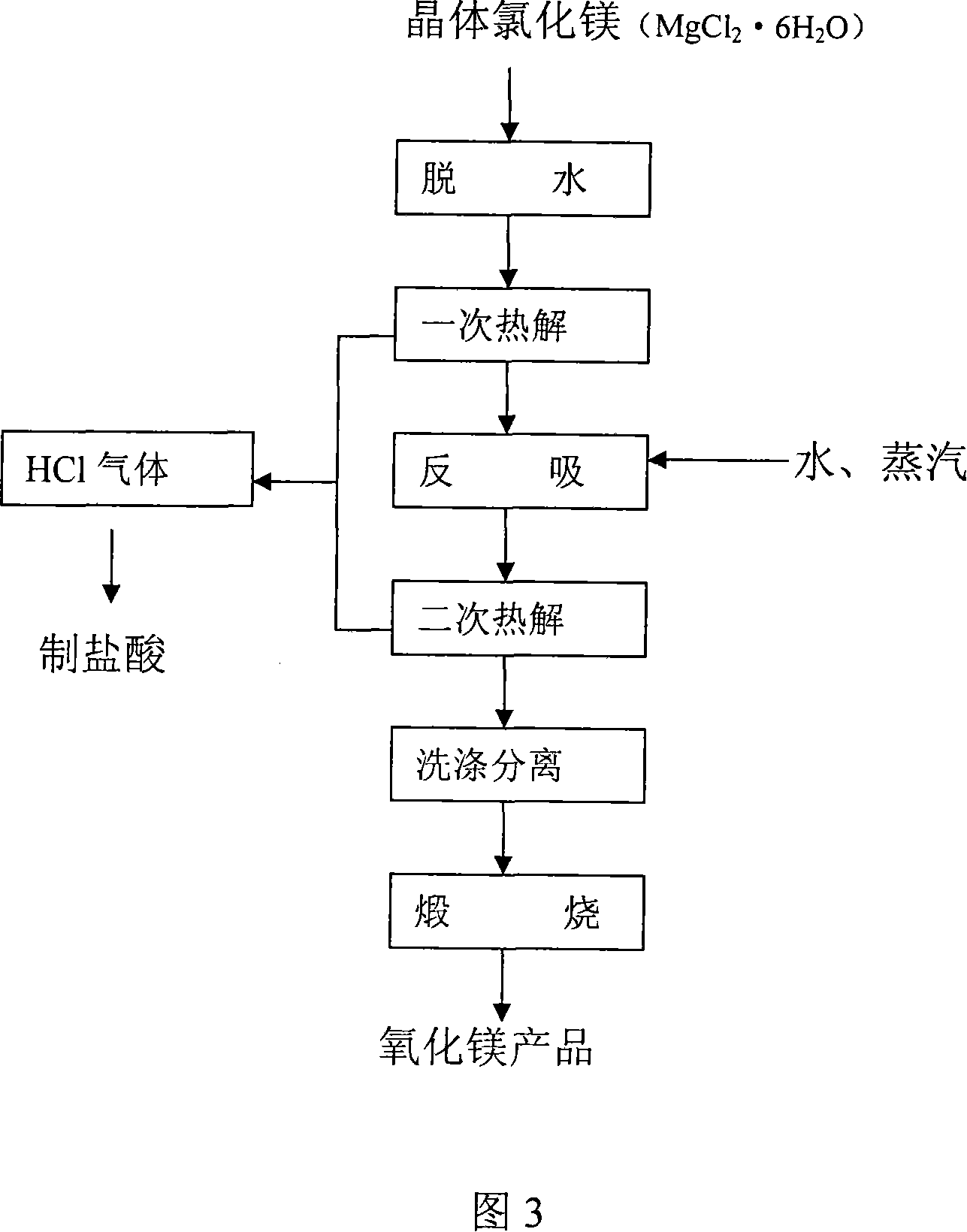
Process for producing high-purity magnesium oxide and lithium salt by using salt lake old brine
InactiveCN101117225ADon't talk about loss rateThe production process is easy to operateAlkali metal chloridesMagnesiaRequirements processMoisture absorption
The invention discloses a process for producing a high purity magnesium chloride, which is as follows: the crystal magnesium chloride separated from the salt lake old brine which is evaporated and concentrated or the residual liquid after the crystallization is processed by primary pyrolysis, counter moisture absorption and secondary pyrolysis. The invention also discloses a process for jointly producing the high purity magnesium chloride and the lithium salt, which is as follows: after the salt lake old brine is evaporated, concentrated and crystallized, the obtained residual liquid is processed by dehydration, primary pyrolysis, counter moisture absorption, secondary pyrolysis, gasification and calcination. The invention is widely adapted to the different salt lake brine, the extraction ratio of magnesium chloride and lithium salt is high. The invention is applied to magnesium chloride production alone, and is also applied to lithium salt joint production, thus the presently difficult technology problem of magnesium-lithium separation is completely resolved at home and abroad. The invention has the advantages of low requirement process, simple equipment, simple principle and easy mastery, short process for single product, low cost for comprehensive productions, .The invention is adapted to the production requirement of different size, and provides the new approach for comprehensive development of the salt lake in Chinese western region.
Owner:陈兆华
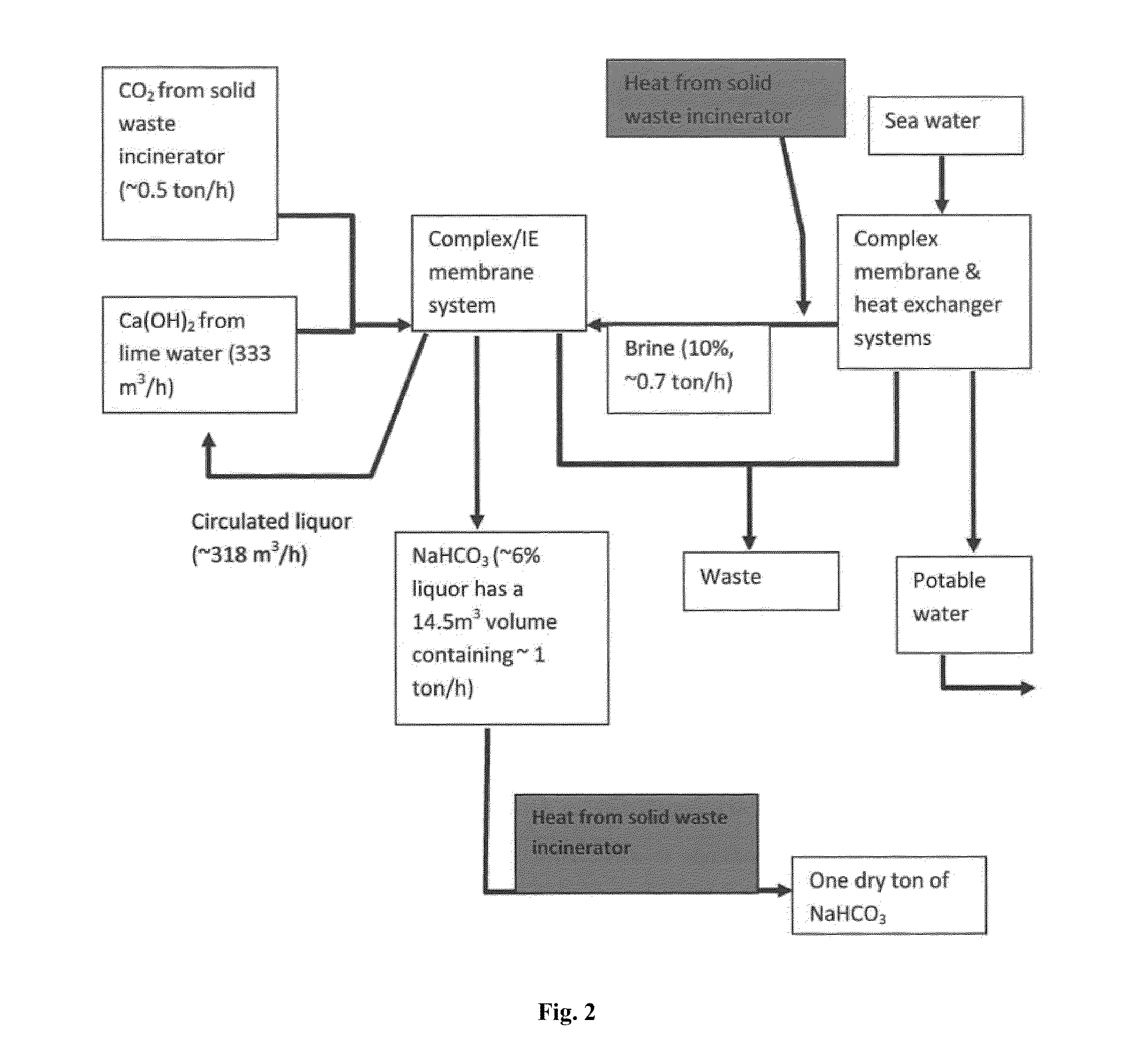
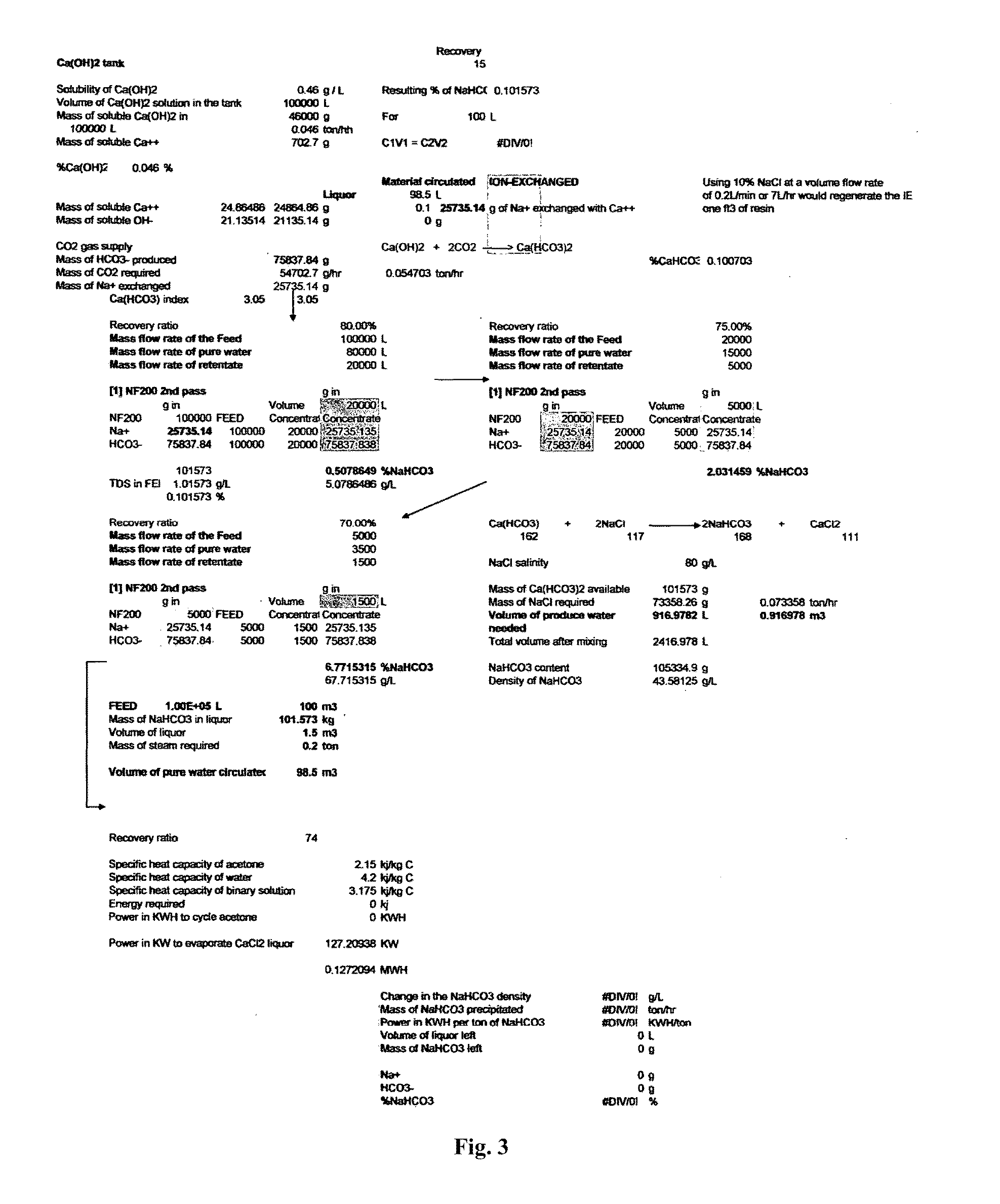
Combined solid waste, carbon dioxide quicklime sparging, brine water, and reverse osmosis/ion exchange processes for the production of soda chemicals
InactiveUS20110217227A1Calcium/strontium/barium carbonatesSemi-permeable membranesWaste processingChemical equation
The proposed invention uses a classical chemical equation where carbon dioxide CO2 is reacted with quick lime Ca(OH)2 to produce soda carb NaHCO3 and concentrating it to 6% using advanced membrane and resin technology. The invention requires three chemicals CO2, Ca(OH)2, and sodium chloride NaCl to produce NaHCO3. The output of many industrial processes lacks waste heat and in many instances CO2 and the present invention combines a solid waste processing unit to the above processes which allows the production of solid products or high % liquors. Availability of waste heat sources can lead to high efficiency in NaHCO3, Na2CO3, and NaOH production. The process is not chloro-alkali electrochemical or Solvay column ammonia processing technique. Advanced membrane uses technologies of reverse osmosis and nanofiltration systems while resin technology uses ion exchange systems. Therefore, we conveniently call it the solid waste-quicklime membrane SWQM process.
Owner:ENGSL FZE +1
Systems and methods for capture and sequestration of gases and compositions derived therefrom
ActiveUS20110129407A1Promote formationCalcium/strontium/barium carbonatesThin material handlingInterior spaceProduct formation
A method of sequestering a greenhouse gas is described, which comprises: (i) providing a solution carrying a first reagent that is capable of reacting with a greenhouse gas; (ii) contacting the solution with a greenhouse gas under conditions that promote a reaction between the at least first reagent and the greenhouse gas to produce at least a first reactant; (iii) providing a porous matrix having interstitial spaces and comprising at least a second reactant; (iv) allowing a solution carrying the at least first reactant to infiltrate at least a substantial portion of the interstitial spaces of the porous matrix under conditions that promote a reaction between the at least first reactant and the at least second reactant to provide at least a first product; and (v) allowing the at least first product to form and fill at least a portion of the interior spaces of the porous matrix, thereby sequestering a greenhouse gas.
Owner:RUTGERS THE STATE UNIV