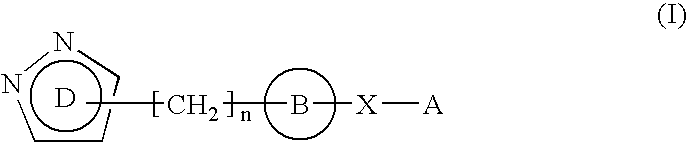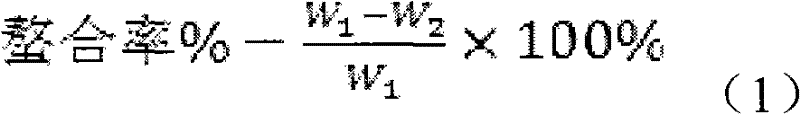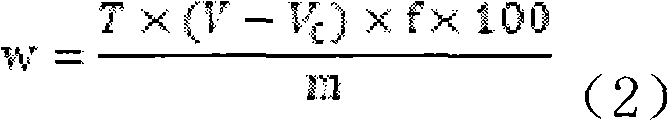Patents
Literature
3846 results about "Calcium EDTA" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
Short answer. Calcium Disodium EDTA (Ethylenediaminetetraacetic Acid) is a chemical chelating agent found in many household foods and beverages. Research shows that in addition to the short-term side effects, the chemical may build up in the body, causing significant toxicity after long-term use.
Malleable putty and flowable paste with allograft bone having residual calcium for filling bone defects
The invention is directed toward a malleable bone putty and a flowable pastel composition for application to a bone defect site to promote new bone growth at the site which comprises a new bone growth inducing compound of partially demineralized lyophilized allograft bone material having a residual calcium content ranging from 4 to 8% dry weight. The bone powder has a particle size ranging from about 100 to about 800 microns and is mixed in a high molecular weight hydrogel carrier containing a sodium phosphate saline buffer, the hydrogel component of the carrier ranging from about 1.00 to 50% of the composition and having a molecular weight of about at least 700,000 Daltons. The composition has a pH between 6.8-7.4 contains about 25% to about 35% bone powder and can be additionally provided with BMP's.
Owner:MUSCULOSKELETAL TRANSPLANT FOUND INC
Calcium based neutral and bioresorbable bone graft
InactiveUS20030055512A1Improve mechanical propertiesReduce decreaseSurgical adhesivesBone implantSulfateBone implant
An injectable and moldable putty comprising biodegradable calcium-based compounds including calcium sulfate, hydroxyapatite, and tricalcium phosphate is invented. The putty hardens into a solid body when mixed with water, saline, serum, or other neutral aqueous solutions. The hardening time of the putty can be tailored in order to meet the specific requirements of various dental or orthopedic applications. The pH of the putty is neutral during and after mixing. The invented putty may be used as bone graft, bone implant, or implantable drug delivery device.
Owner:BERKELEY ADVANCED BIOMATERIALS
Compositions and Methods for Improving Overall Tooth Health and Appearance
ActiveUS20080247973A1Avoid lostSuperior anticariesCosmetic preparationsToilet preparationsAmmonium compoundsStaining
Disclosed are oral care compositions comprising selected surface-active organophosphate compounds and methods of use to provide protection of teeth from erosion caused by the action of chemicals, such as harsh abrasives and acids. The surface-active organophosphate compounds are substantive to teeth, the phosphate groups binding the calcium in teeth and thus preventing loss of calcium from dissolution when contacted with acids. The organophosphate compound may also deposit a protective surface coating that prevents contact of teeth with erosive challenges. Selected organophosphate compounds contain one or more phosphate groups and are combined in the oral care composition with one or more of a fluoride ion agent, an antimicrobial agent preferably selected from quaternary ammonium compounds and polyvalent metal salts, an anticalculus agent and additional surfactant, to provide benefits including superior anti-erosion, anticaries, antiplaque and anti-staining as demonstrated by enhanced fluoride uptake, remineralization, resistance to acid demineralization and antimicrobial activities, resulting in improved overall tooth health, structural integrity and appearance.
Owner:THE PROCTER & GAMBLE COMPANY
Enhanced efficacy antiperspirant compositions containing strontium or calcium
InactiveUS6923952B2Improve efficacySimple compositionCosmetic preparationsToilet preparationsStrontiumWater soluble
Owner:THE GILLETTE CO
Solid pharmaceutical dosage forms comprising bisphosphonates and modified amino acid carriers
Owner:NOVO NORDISK NORTH AMERICA OPERATIONS AS
Deposition of calcium-phosphate (CaP) and calcium-phosphate with bone morphogenic protein (CaP+BMP) coatings on metallic and polymeric surfaces
InactiveUS20080097618A1Reduce the amount of solutionHigh densityImpression capsBone implantPolymeric surfaceSolubility
The invention is a medical implantable device which is coated by the method according to the invention. The surface of the substrate used for the implantable device, in the raw condition, following a cleaning regime and physiochemical pretreatments, is coated using a biomimetic process in a supersaturated calcium phosphate solution (SCPS) to obtain the desired coating coverage and morphology maintaining a ratio of calcium to phosphorus pH, as well as solution temperature plays a major role in yielding precipitation of the proper phase of CaP so that composition, morphologies, crystal structures, and solubility characteristics are optimal for the deposition process. The biomimetic coating adds the attribute of osteoconductivity to the implant device. To maximize bone growth, the implant must also induce bone growth, or possess the attribute of osteoinductivity. This attribute is acquired by the use of therapeutic agents, i.e. bone morphogenic proteins (BMP), growth factors, stem cells, etc. The preparation of the SCPS solution is slightly altered so that during the immersion of the implant in the SCPS, the therapeutic agents are co-precipitated and bonded with the CaP directly on the underlying surface of the implant device. A final dipping process into a BMP solution provides an initial burst of cellular activity. For delivering stem and / or progenitor cell, after drying the dipped solution of BMP, the cells are cultured on the surface of the implant.
Owner:HERKOWITZ HARRY N
Betaine with Calcium and/or Strontium Antiperspirants
InactiveUS20070020211A1Good curative effectImprove skinCosmetic preparationsToilet preparationsAntiperspirantsBetaine
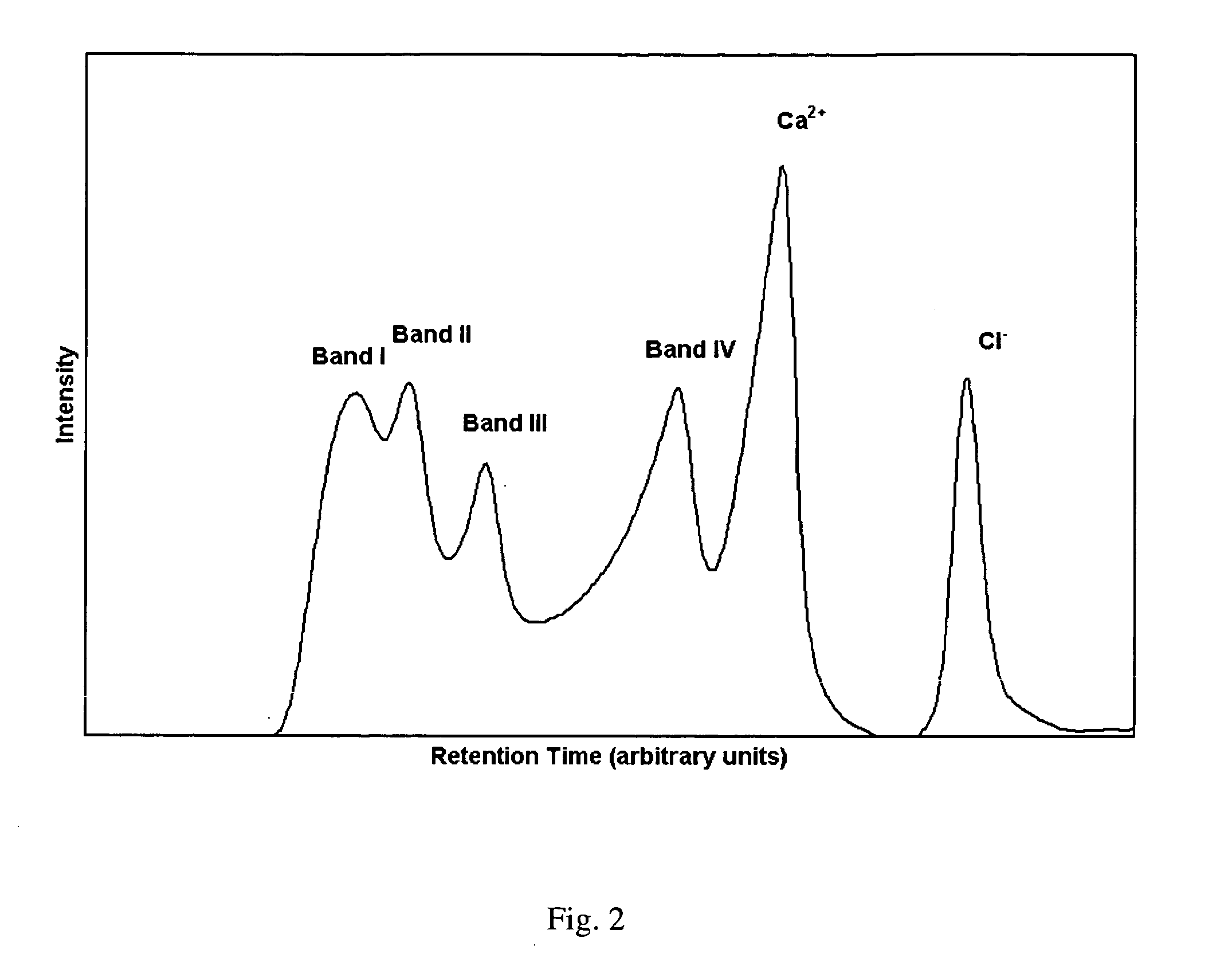
Aluminum and aluminum-zirconium antiperspirant compositions comprising basic aluminum chlorides that have a particular molecular size distribution defined by having an SEC-HPLC Band III / II ratio of at least 0.5, having SEC-HPLC Band III plus Band II area of at least 70% of the total area and having SEC-HPLC Band I content no more than 5% and containing betaine (trimethylglycine), calcium and / or strontium are disclosed. Also disclosed are the methods of making these compositions and the use thereof in consumer acceptable antiperspirant vehicles such as aerosols, gels, roll-on, sticks and soft solids.
Owner:SUMMIT RES LAB
Alkaline land soil repair agent
InactiveCN101417286AReduce alkalinityImprove biological activityContaminated soil reclamationOrganic fertilisersNatural organic matterAlkali soil
The invention relates to a restoration agent for saline-alkali land soil, which consists of free gypsum dehydration, nature organic substance, microbial fertilizer and inorganic nutrient with weight ratio of 60-70:25-35:1-4:3-6; the content of dihydrate calcium sulfate in the free gypsum dehydration is over 80 percent; the nature organic substance consists of weathered coal and grass carbon with weight ratio of 1.0:0.93; the content of effective bacteria in the microbial fertilizer is 2.0 hundred millions / g; and the inorganic nutrient consists of urea, ordinary superphosphate and potassium chloride with weight ratio of 1.0:2.42:075. The restoration agent can utilize calcium ions in the free gypsum dehydration for replacing sodium ions in the soil so as to lower the alkalinity in the soil and provide calcium and sulfur elements for crops, while the nature organic substance can lower and regulate the slat content and the alkalinity in the soil. In addition, the nature organic substance, the microbial fertilizer and the inorganic nutrient can supply large amount of nutrient elements such as nitrogen, phosphorus, potassium and the like to the crop for normal growth, thereby being capable of improving the yield of the crop.
Owner:TIANJIN INST OF AGRI RESOURCES & ENVIRONMENTAL
Modified ligands of calcium-dependent binding proteins
InactiveUS6316409B1High affinityImprove efficiencyOrganic active ingredientsBiocideWild typeCalcium dependent
The present invention relates to ligands capable of binding a calcium dependent binding protein, that comprise an amino acid sequence corresponding to that of a wild type ligand for the calcium dependent binding protein with modification which results in enhanced affinity of the ligand for the calcium dependent binding protein.
Owner:MEDICAL RESEARCH COUNCIL
Strontium fortified calcium nano-and microparticle compositions and methods of making and using thereof
InactiveUS20080317807A1Good biocompatibilityPrevent looseningHeavy metal active ingredientsBiocideDiseasePhosphate
Compositions containing strontium fortified calcium nanoparticles and / or microparticles, and methods of making and using thereof are described herein. The strontium fortified calcium compounds contain calcium ions, calcium atoms, strontium ions, strontium atoms, and combinations thereof and one or more anions. Exemplary anions include, but are not limited to, citrate, phosphate, carbonate, and combinations thereof. The particles can be formulated for enteral or parenteral administration by incorporating the particles into a pharmaceutically carrier. The compositions can further contain one or more active agents useful for bone diseases or disorders, such as vitamin D, growth factors, and combinations thereof. The compositions can be used to treat or prevent one or more bone diseases or disorders of the bone, such as osteoporosis. Alternatively, the particles can be coated onto a substrate, such as the surface of an implant. The coatings can be used to improved biocompatibility of the implant, prevent loosening of the implant, reducing leaching of metal ions from metallic implants, and reduce corrosion. The coatings can be applied to the substrate using a variety of techniques well known in the art. In one embodiment, the coating is applied using electrophoretic deposition. The use of nano- and / or microparticles that provide high surface area helps to improve interfacial strength between the coating and the implant, which allows for the use of lower sintering temperatures. Lowering sintering temperatures minimizes or prevents thermal decomposition of the coating material and / or degradation of the implant material.
Owner:THE UNIVERSITY OF HONG KONG
Carbonated fortified milk-based beverage and method for suppressing bacterial growth in the beverage
InactiveUS6866877B2Increase attractivenessFast absorptionMilk preparationVitamin food ingredientsAdditive ingredientPasteurization
Dairy or non-diary based fortified carbonated beverage solutions that supply essential nutrients in the human diet. The solution contains per 354 ml, calcium, magnesium and potassium ions in the form of salts and optionally vitamins A, D, C, lutein, zeaxanthin and folic acid in specified amounts to provide dietary supplementation. Sweeteners, stabilizers, flavors and carbonation can also be added to enhance flavor, taste, mouth-feel, ingredient solubilization and product appearance. A method of making the beverages is also described. A method of using carbonation to reduce bacterial counts and reduce degradation of essential nutrients in milk-based beverages with or without pasteurization is also disclosed.
Owner:MAC FARMS
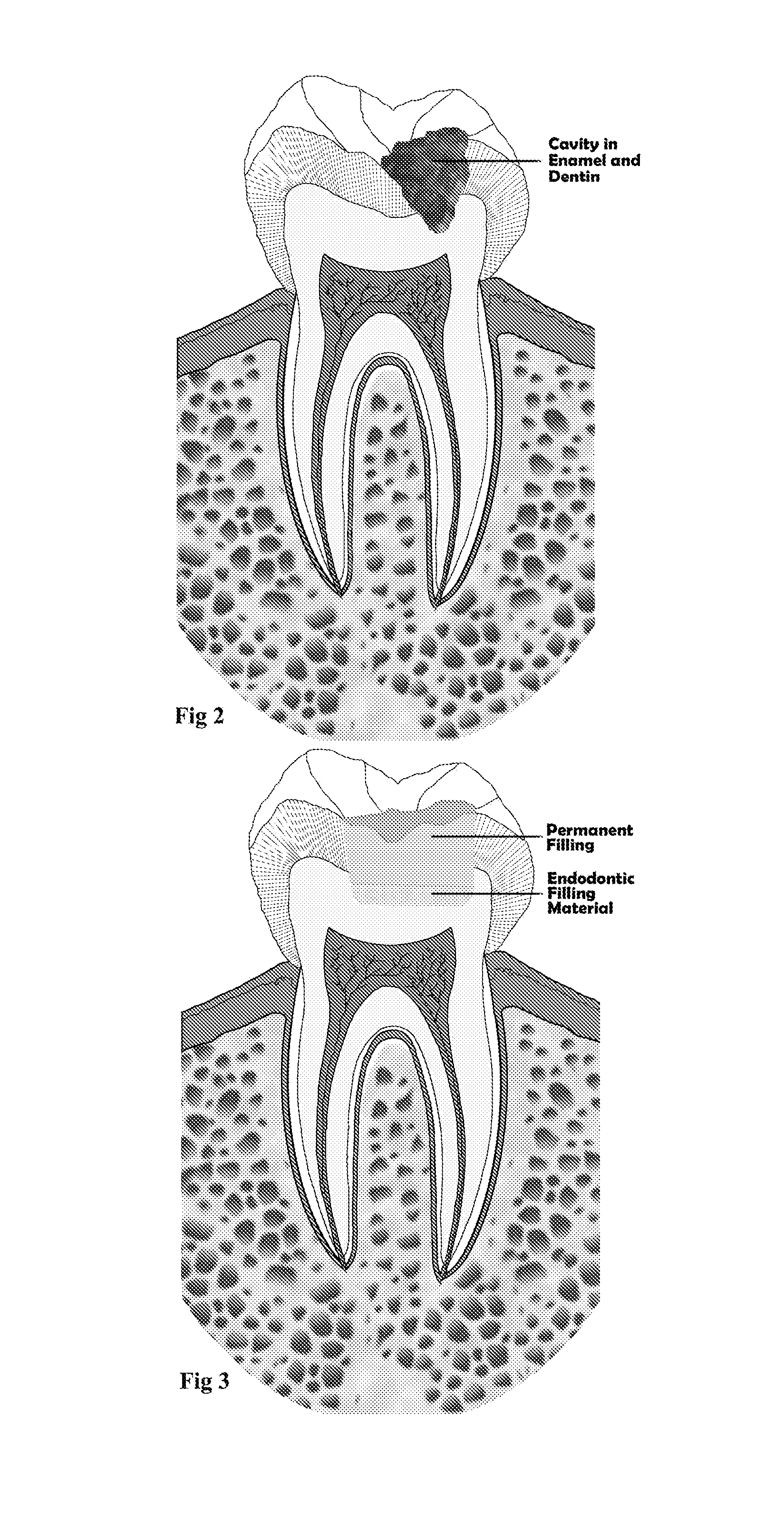
Endodontic filling material
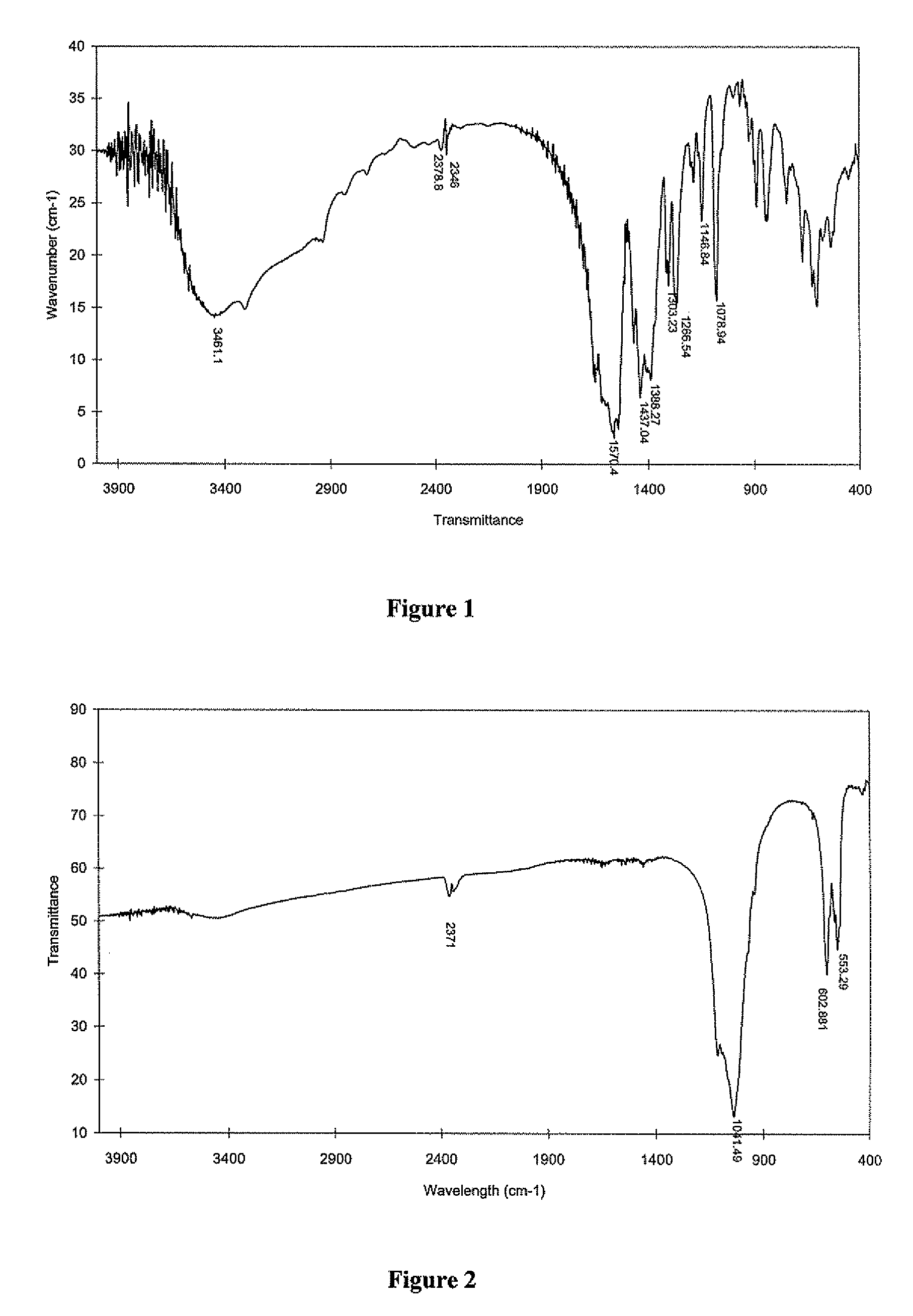
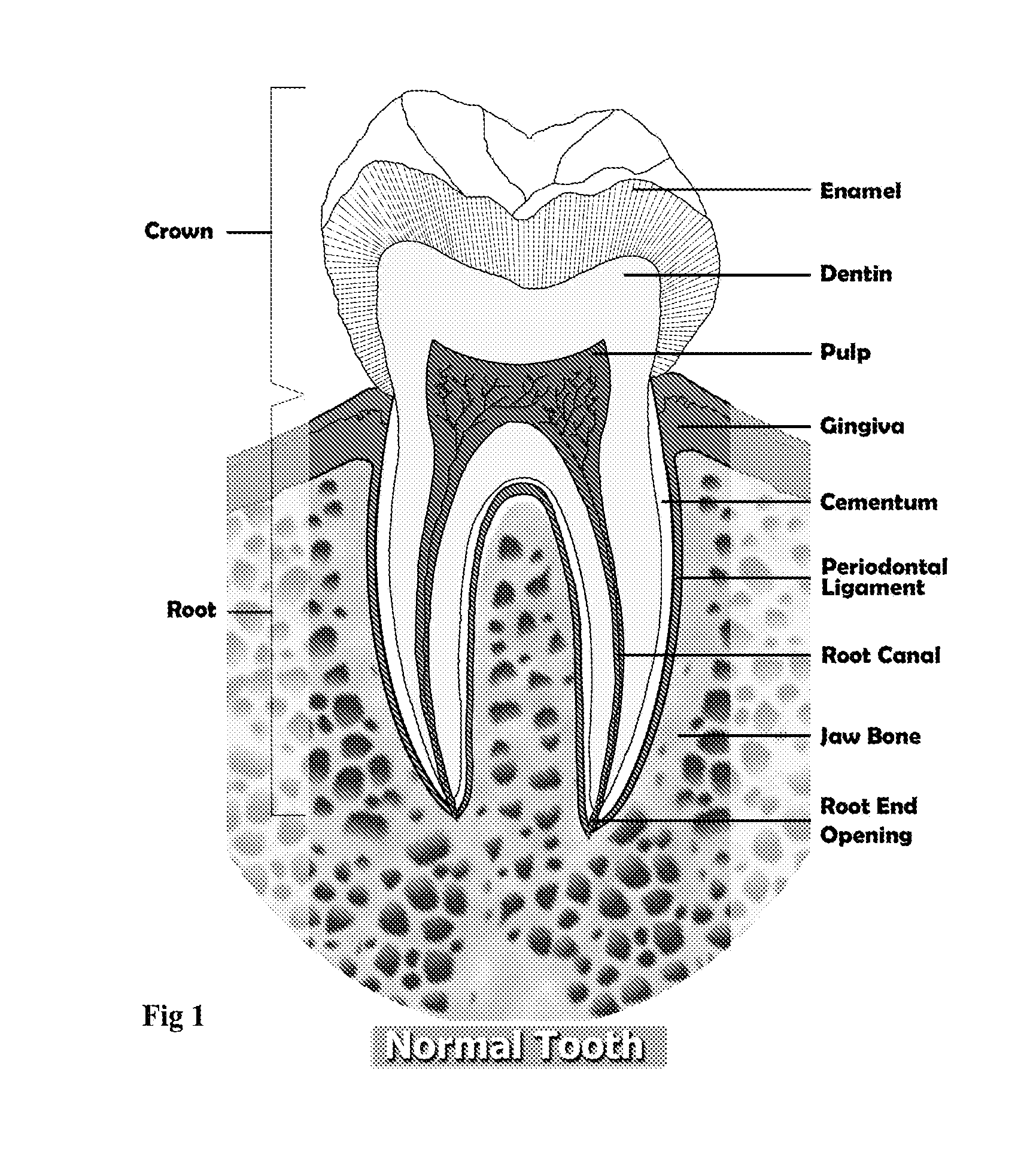
InactiveUS20080206716A1Improve sealingEasily manipulationAntibacterial agentsOther chemical processesCalcium silicateHigh concentration
The present invention is a bioactive endodontic material and its use for filling the tooth and bone cavities. The present invention, by using calcium salt, calcium oxide, calcium silicate, and calcium phosphate compounds as essential constituents, and mixing them with a water base solution, prepares a bioactive calcium and phosphate enriched material. The enriched material (cement) comprises high concentration of water-soluble calcium and phosphate, and immediately forms hydroxyapatite during and after setting. The cement is biocompatible, antibacterial, capable to form an effective seal against reentrance of microorganisms into the filled cavity, compatible to handle and set in an aqueous environment, and able to stimulate hard tissue healing.
Owner:ASGARY SAEED
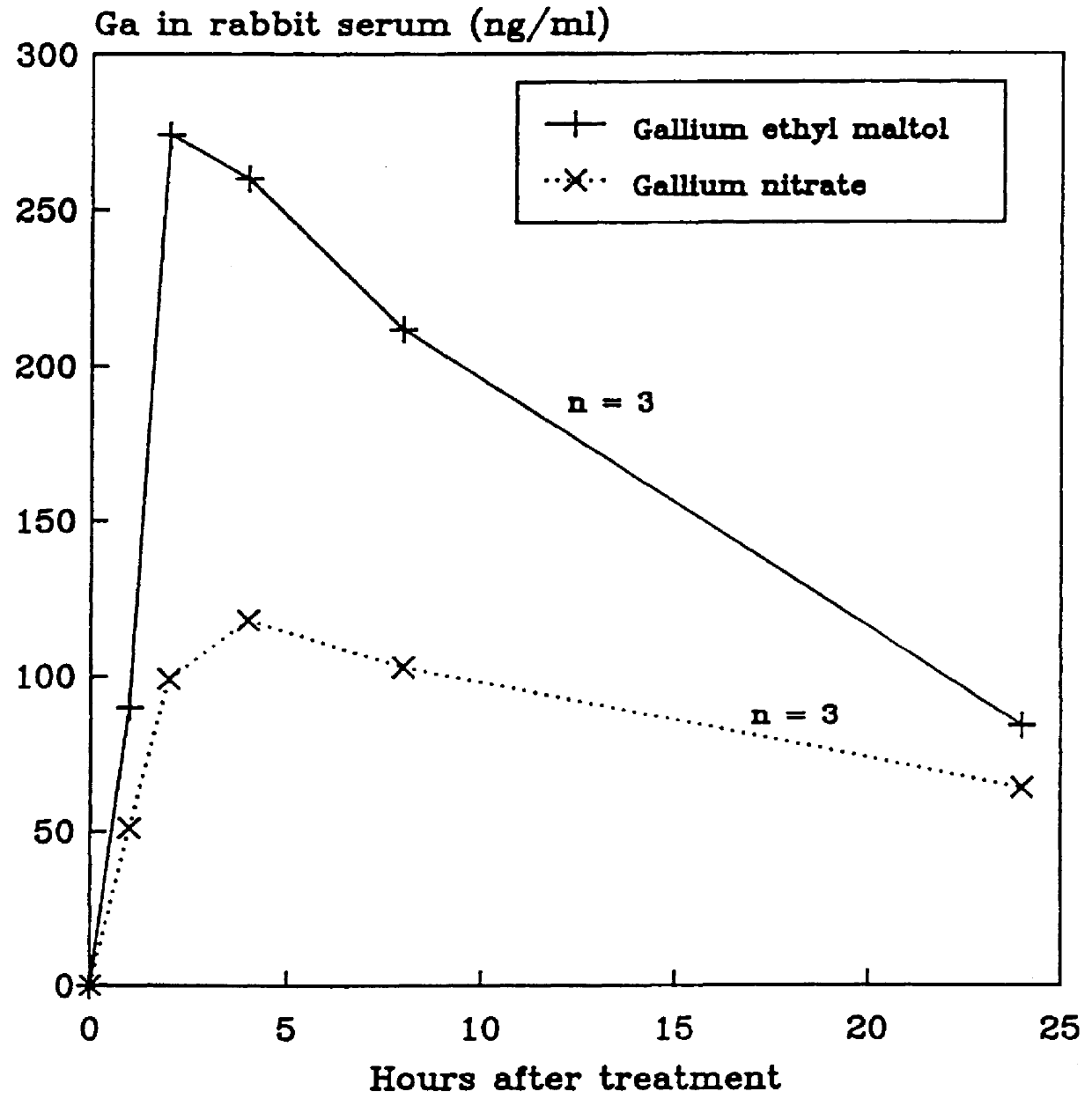
Gallium complexes of 3-hydroxy-4-pyrones to treat cancer
Disclosed are pharmaceutical compositions that comprise gallium complexes of 3-hydroxy-4-pyrones. These compositions provide enhanced gallium bioavailability particularly when orally administered as compared to the gallium bioavailability achieved by use of pharmaceutical compositions containing gallium salts. Compositions included in this invention are useful in providing gallium to humans and other mammals for a wide variety of medical and veterinary applications, including the treatment, prevention, or diagnosis of hypercalcemia, certain cancers, certain disorders of calcium homeostasis, and certain bone diseases including osteoporosis, osteopenia, and Paget's disease.
Owner:BERNSTEIN LAWRENCE RICHARD
Carbonated fortified milk-based beverage and method for suppressing bacterial growth in the beverage
InactiveUS20030113408A1Great tasteIncrease bodyMilk preparationVitamin food ingredientsAdditive ingredientPasteurization
Dairy or non-diary based fortified carbonated beverage solutions that supply essential nutrients in the human diet. The solution contains per 354 ml, calcium, magnesium and potassium ions in the form of salts and optionally vitamins A, D, C, lutein, zeaxanthin and folic acid in specified amounts to provide dietary supplementation. Sweeteners, stabilizers, flavors and carbonation can also be added to enhance flavor, taste, mouth-feel, ingredient solubilization and product appearance. A method of making the beverages is also described. A method of using carbonation to reduce bacterial counts and reduce degradation of essential nutrients in milk-based beverages with or without pasteurization is also disclosed.
Owner:MAC FARMS
Pyrazole derivative
The present invention is directed to drugs, in particular, pyrazole derivatives represented by the following general formula (I) which have a calcium release-activated calcium channel inhibitory effect and medicinal compositions, in particular, calcium release-activated calcium channel inhibitors containing the above compounds as the active ingredient, wherein each substituent is defined in the specification.The present invention also relates to a pharmaceutical composition containing an effective amount of the compound of formula (I) and a pharmaceutically effective carrier.The present invention further relates to methods of treatment of diseases associated with calcium release-activated calcium channels, diseases associated with IL-2 production, and methods of treatment of allergic, inflammatory or auto-immune diseases.
Owner:ASTELLAS PHARMA INC
Methods and compositions for the treatment of diseases characterized by calcification and/or plaque formation
The invention provides methods and compositions that include a nutraceutical supplement, antibiotic, and metal chelating agent that is administered to a patient to treat or prevent pathological calcification and or plaque formation as associated with Nanobacteria Calcifying Nano-Particles and / or diseases caused there-from, The method includes the administration of a therapeutically effective nutraceutical supplement, tetracycline HCL, and ethylenediaminetetraacetic acid calcium di-sodium salt to a patient in order to prevent and treat calcific disease.
Owner:CIFTCIOGLU NEVA
Compositions and methods for calcium fortification of dairy products and oleaginous foods
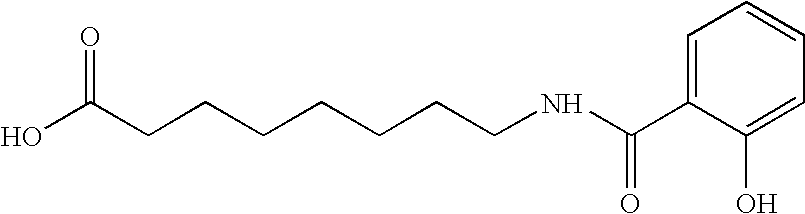
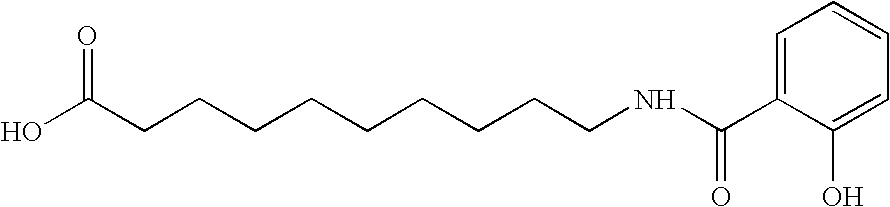
InactiveUS6299914B1Easy to meetLessen the shelf-life of the dairy productEdible oils/fats ingredientsMilk preparationSuspending AgentsCalcium EDTA
Calcium amino acid chelate complexes for fortification of dairy products and oleaginous foods, dairy products and oleaginous foods fortified with calcium amino acid chelate complexes, and methods of preparing calcium amino acid chelate complexes are disclosed and described. The calcium amino acid chelate complexes of the present invention are stable, palatable, and bioavailable. Preparation steps include reacting a calcium compound, an amino acid ligand, a pH adjuster, and, optionally, a stabilizing / suspending agent in an aqueous environment.
Owner:ALBION INT
Production of porous structure with biological activity on surface of pure-titanium tooth implantation
InactiveCN1817319AImprove clinical successHas bone conductionDental implantsImpression capsBone tissueMedicine
A process for preparing the bioactive porous structure on the surface of Ti explant for tooth includes such steps as blasting sand, double acid treating, treating with H2O2 and heat treating. Its surficial TiO2 layer can induce the crystallization of hydroxy apatite, and has high compatibility to bone tissue and high successful rate.
Owner:ZHEJIANG UNIV
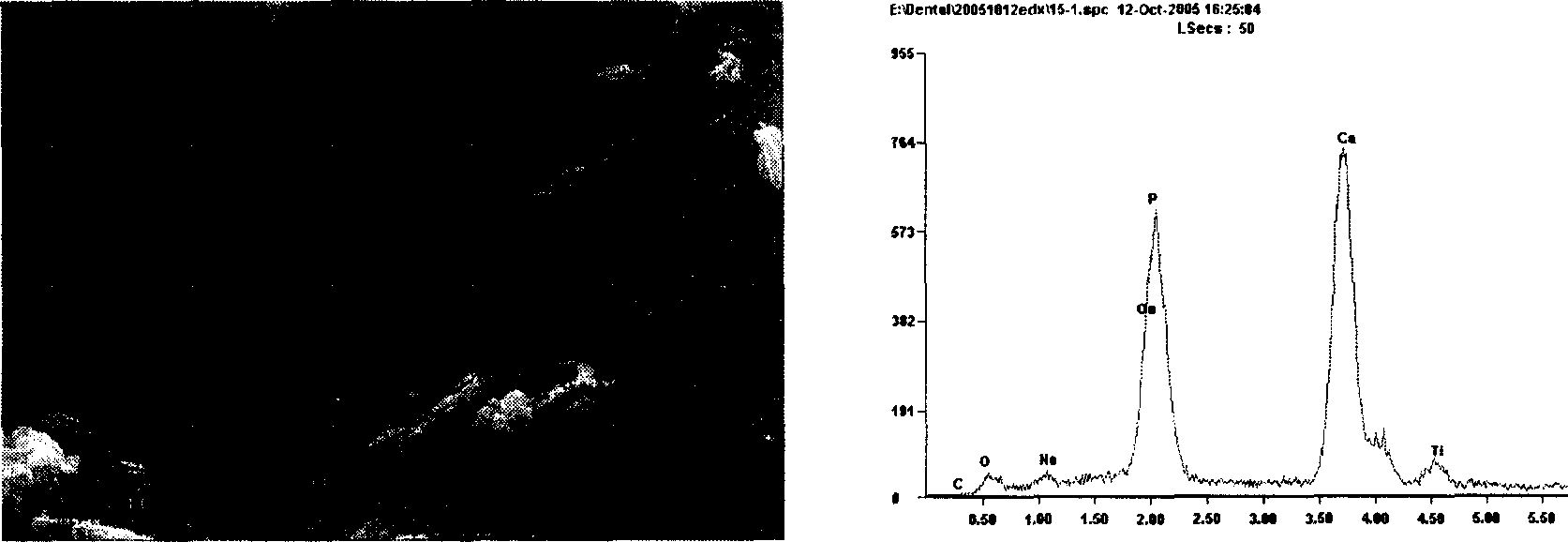
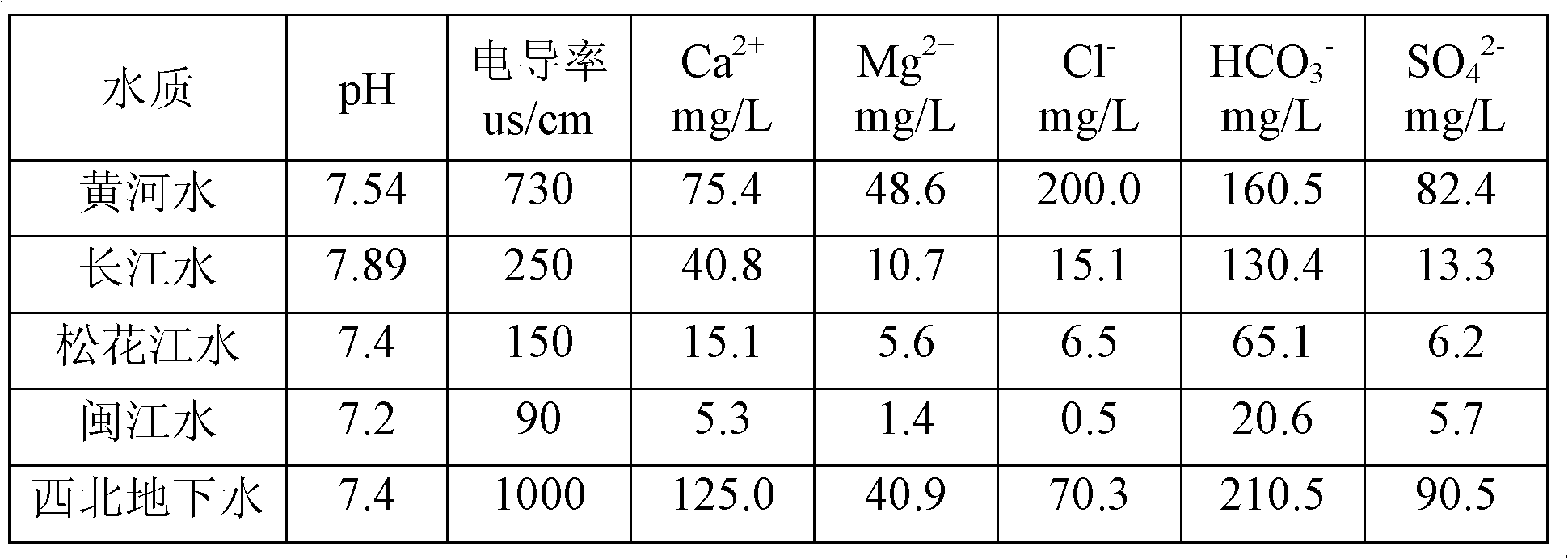
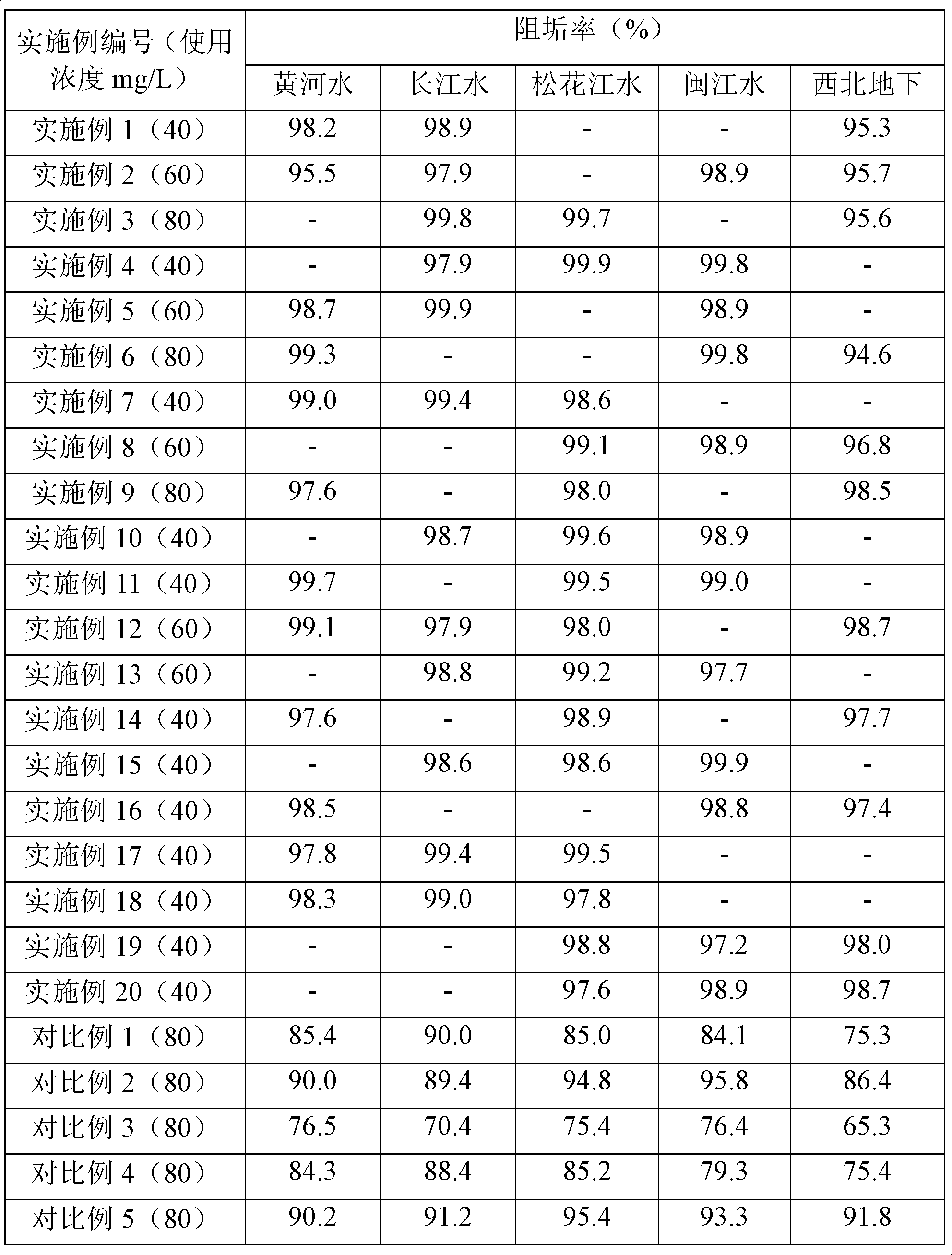
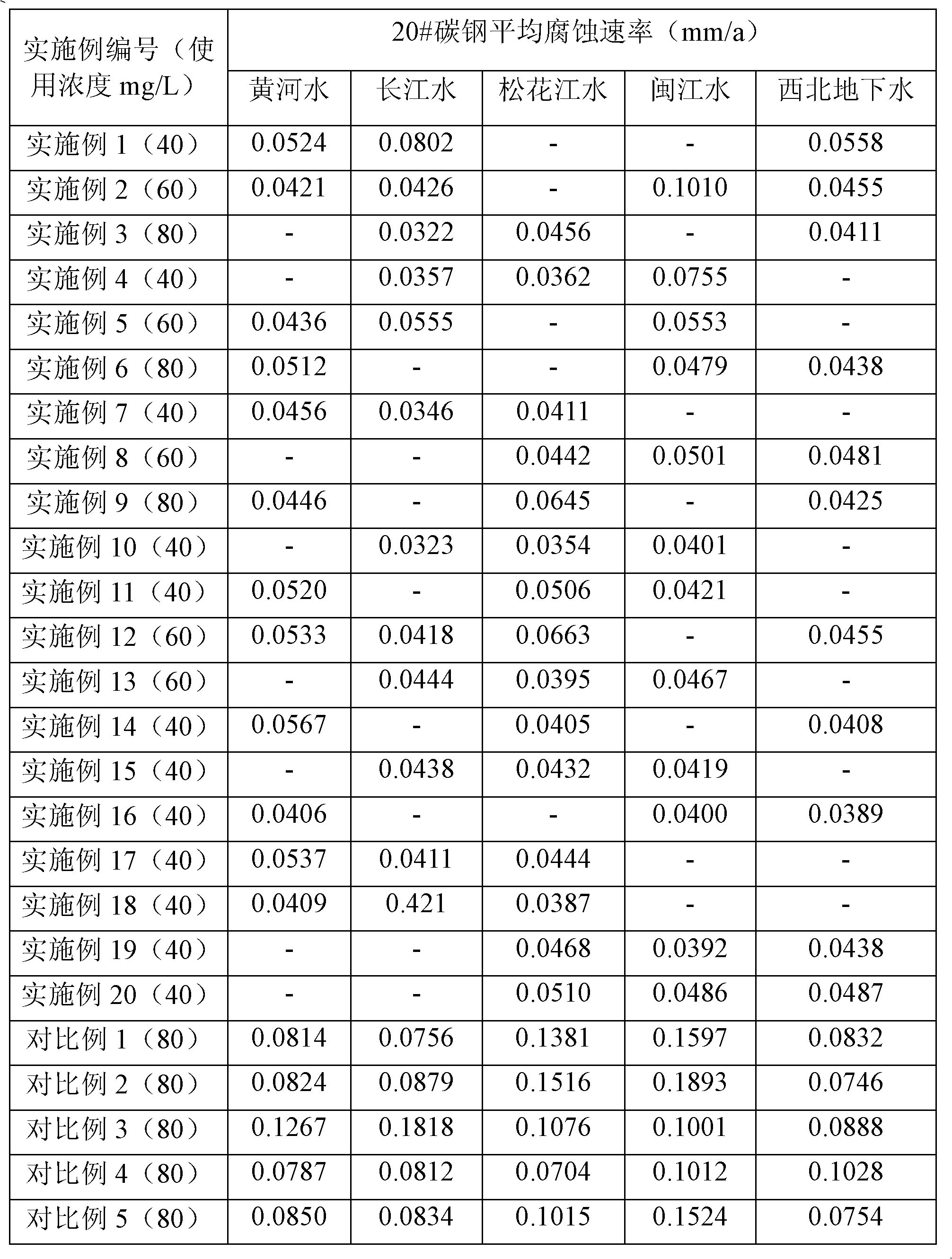
Composition used in scale-inhibition corrosion inhibitor, scale-inhibition corrosion inhibitor, and application thereof
ActiveCN103253777AHigh calcium and alkali toleranceGood scale and corrosion inhibitionScale removal and water softeningHigh concentrationTungstate
The invention discloses a composition used in a scale-inhibition corrosion inhibitor, a scale-inhibition corrosion inhibitor, and an application thereof. The composition used in the scale-inhibition corrosion inhibitor comprises cyclodextrin and / or a cyclodextrin derivative, a carboxylic-acid-group-containing homopolymer, a sulfonic-acid-group-containing copolymer, and selectively hydroxycarboxylic acid with a weight ratio of 1:0.2-20:0.04-15:0.2-30:0-20. The scale-inhibition corrosion inhibitor provided by the invention is environment-friendly. According to the invention, the application of phosphorus-containing compound or non-renewable molybdate or tungstate is prevented, such that the scale-inhibition corrosion inhibitor is phosphorus-free and nitrogen-free. The scale-inhibition corrosion inhibitor has high tolerance to calcium and alkali, resistance to corrosion of high-concentration chloride ion and sulfate ion, and wide applicability to water quality. Therefore, technical condition is provided for operation of circulation water under high concentration multiple and for reducing sewage discharge. The scale-inhibition corrosion inhibitor provided by the invention has excellent scale inhibition and corrosion inhibition performances in circulation water system application. With the scale-inhibition corrosion inhibitor, a dose is low, scale-inhibition treatment process is simple and substantial effect is provided. The scale-inhibition corrosion inhibitor is suitable for popularization.
Owner:CHINA PETROLEUM & CHEM CORP +1
Nutrient clusters for food products and methods of preparation
Nutrient clusters for food products, such as for addition to Ready-To-Eat cereals, are made in the form of aggregates or clusters comprising a first particulate component; a nutrient powder blend, and sufficient binder to adhere the powder to the particulates. The nutrient clusters are in the form of pieces each weighing from about 0.3 to 5 g. and having a moisture content of about 2% to 10%. Such nutrient clusters are prepared by applying a liquid binder to the particulates to form sticky particulates, adding a powdered nutrient blend, and curing the mixtures to form hardened dried nutrient clusters. The nutrient cluster can contain 100% US recommended daily allowance of essential vitamins and minerals and can contain added macronutrients such as soy proteins, soluble fiber, and / or calcium in nutritionally dense form in as little as 5 to 15 g of nutrient clusters. The clusters find particular suitability for use in providing to-order customized cereal products in response to particular customer requirements for nutrition.
Owner:GENERAL MILLS INC
Small molecule polypeptide Ca-chelate of fishbone and preparation method
InactiveCN102406176AHigh calcium chelation rateIncreased bioabsorptionFood preparationSolubilitySide effect
The invention discloses small molecule polypeptide Ca-chelate of fishbone, which belongs to the field of functional food or additive. The method for preparing the small molecule polypeptide Ca-chelate of the fishbone comprises the following steps: boiling the fishbone at high temperature and high pressure; pulping to obtain the fishbone paste; utilizing the compound protease to execute enzymolysis; centrifuging to obtain the liquid supernatant and the sediment; filtering the liquid supernatant to obtain the small molecule polypeptide liquid of the fishbone; executing the composite acid acidification through citric acid and lactic acid for the sediment so as to obtain the fishbone calcium liquid; mixing the small molecule polypeptide liquid of the fishbone with the fishbone calcium liquid; executing the chelation at 40-50 degrees centigrade while the pH value is 7.5-8.5; and drying to obtain the small molecule polypeptide Ca-chelate of the fishbone. The invention has high Ca-chelate rate which reaches over 90%, and has high biological absorption rate of the calcium which reaches over 70%. The invention has no side effects, has good flavor, can be directly absorbed from intestinal mucosa after being eaten, solves the problems that the solubility of the traditional calcium source is bad, the absorption rate is low and the side effect is large, and solves the problem of producing calculi. In addition, the invention has a simple preparation method and low cost.
Owner:SHANDONG RONGXIN AQUATIC PROD FOOD GROUP
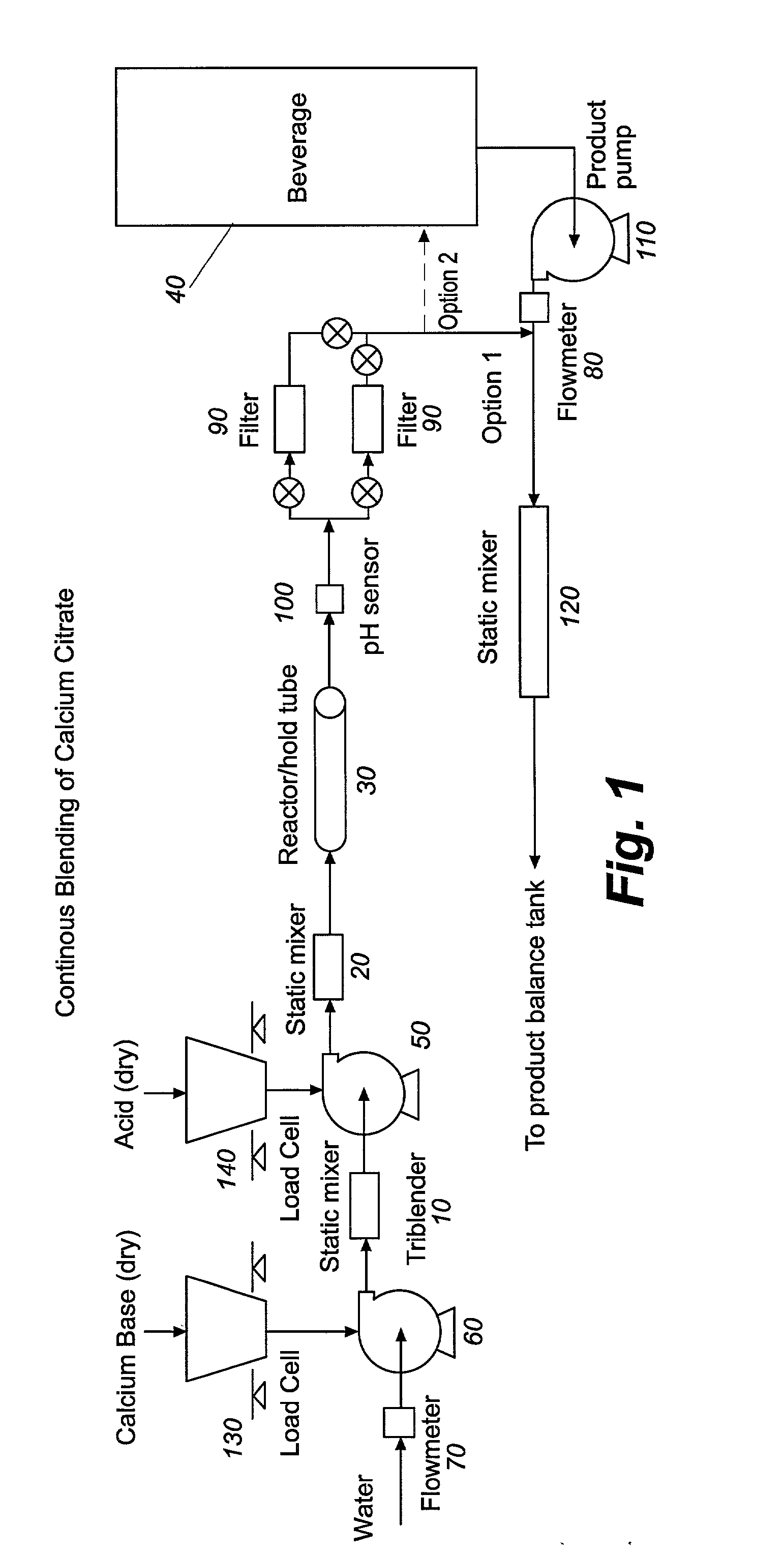
Process apparatus, and composition for calcium fortification of beverages
InactiveUS20020122866A1Inhibit productionReduce presenceFood ingredientsFood preparationCalcium hydroxideSufficient time
Process, apparatus and composition for calcium fortification of beverages are provided. The methods comprise continuously blending in water a calcium source, preferably calcium hydroxide, and an acid source for a time sufficient to produce soluble calcium salts, but with minimal precipitation. The acid source is preferably citric acid. The calcium salt solution remains in an in-line retaining reaction hold tube for the necessary period to produce calcium salt in solution without significant precipitation, and is then continuously and immediately added to a non-dairy beverage, such as orange juice, to calcium fortify the beverage. The pH of the calcium salt solution can be monitored to optimize the amount of soluble salt and minimize the amount of precipitate.
Owner:THE COCA-COLA CO
Calcium zeolite hemostatic agent
ActiveUS7595429B2Promote formationReduce heatBiocideInanimate material medical ingredientsCalcium contentCalcium EDTA
A composition for promoting the formation of clots in blood comprises a binder and a zeolite disposed in the binder, the zeolite having an adjusted calcium content. A method of forming a blood-clotting composition comprises the steps of providing a zeolite, combining the zeolite with a binder, and adjusting a calcium content of the zeolite to have an amount of calcium such that upon application of the composition to a wound, a heat of hydration is reduced and thereby heat transferred to the wound is reduced. A method of clotting blood flowing from a wound comprises the steps of applying a zeolite to the wound and maintaining the zeolite in contact with the wound for a predetermined amount of time. The zeolite preferably has an adjusted calcium content and is capable of producing a controllable blood clotting effect on the wound.
Owner:TELEFLEX LIFE SCI LTD
Formula milk powder for promoting absorption of fatty acid and calcium and preparation method thereof
The invention discloses a formula milk powder for promoting the absorption of fatty acid and calcium and a preparation method thereof. Raw cow milk, lactose, 1,3-Dioleoyl 2-palmitoyl triglyceride and demineralized whey powder as main materials are added with concentrated whey albumen powder, Alpha-lactalbumin powder, oligosaccharide, walnut oil, casein phosphopeptide, docosahexaenoic acid, arachidonic acid, nucleotide, lutein, inositol, carnitine and the like as well as vitamins, mineral substances and other nutrients needed for strengthening infants, and fat humanization, protein humanization and carbohydrate humanization are realized. The powdery product is produced by the processes of blending, homogenization, concentration, spray-drying, packaging and the like. According to the physiological characteristics and nutritional demand of the infants, the invention reinforces the calcium, the 1,3-Dioleoyl 2-palmitoyl triglyceride, other nutrient ingredients and the like, and aiming at the oversea clinical test conclusion of the 1,3-Dioleoyl 2-palmitoyl triglyceride, the final test conclusion of comparison with breast milk and infant formula milk powder sold on the market in the process of a clinical feeding test is that the feeding result of the designed formula approximates the feeding result of the breast milk and is better than the feeding result of an infant formula milk powder group sold on the market.
Owner:HEILONGJIANG FEIHE DAIRY
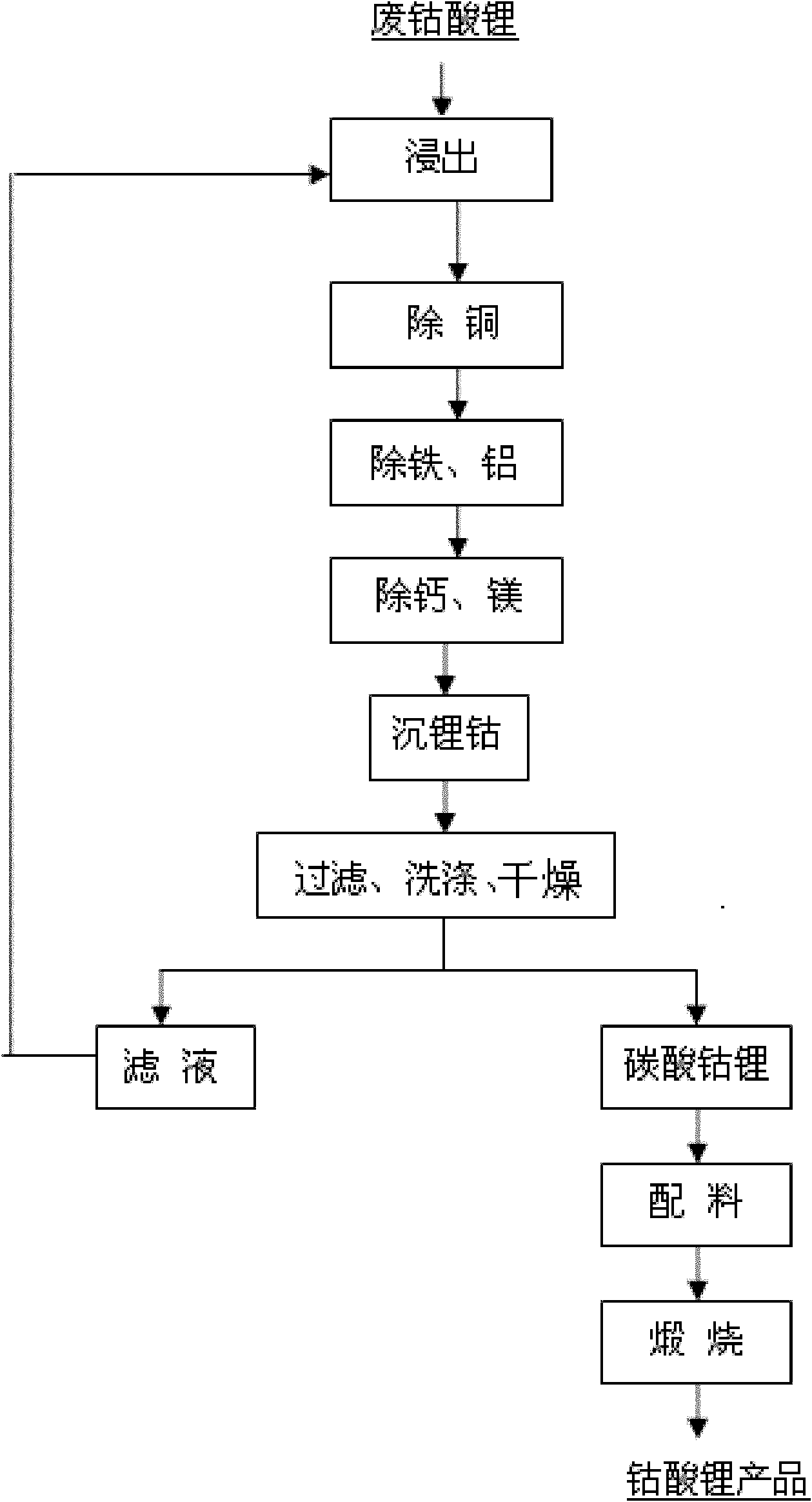
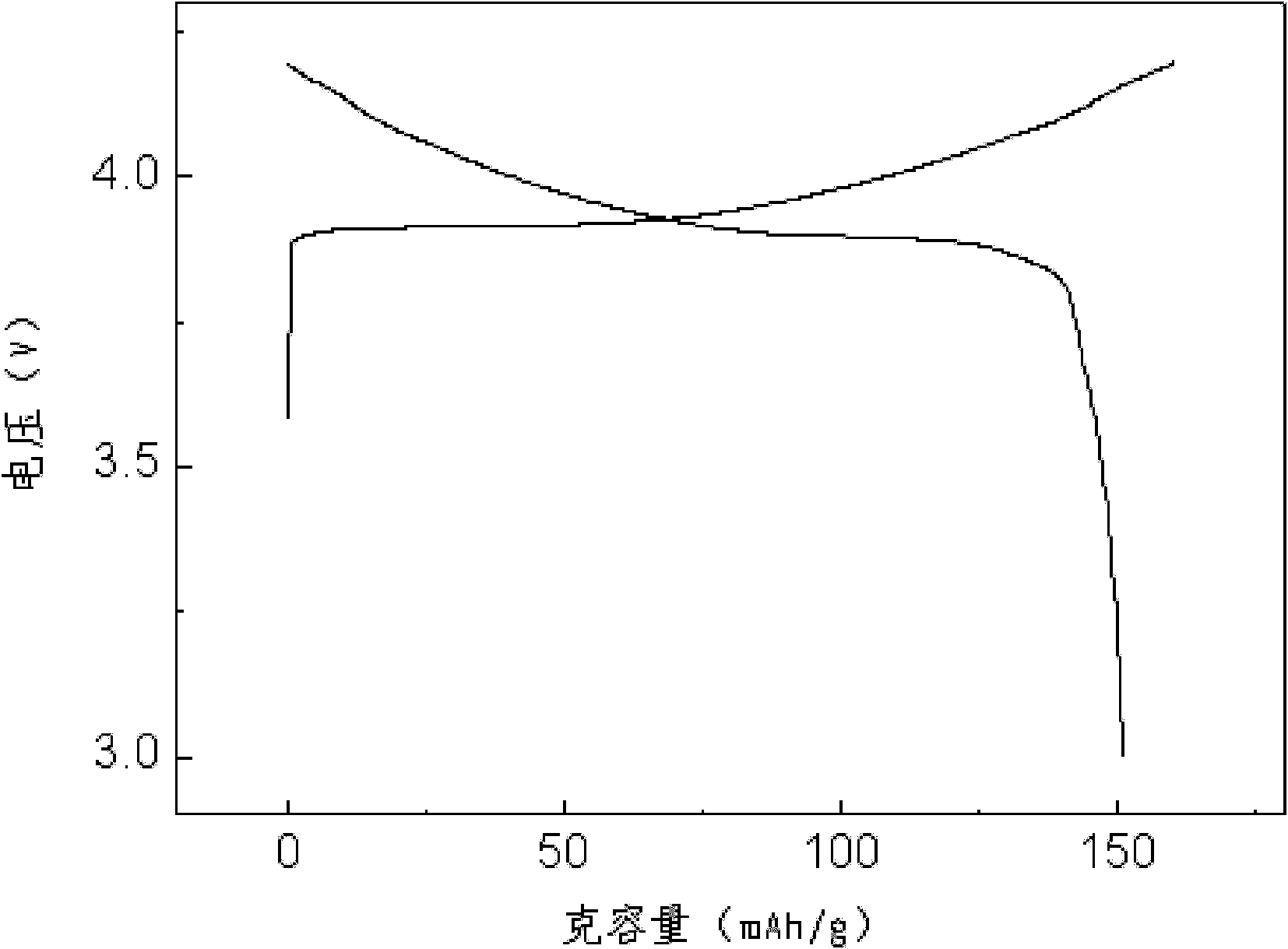
Method for recovering cobalt and lithium from waste lithium cobaltite and preparing lithium cobaltite
InactiveCN102101701AReduce consumptionSimple processWaste accumulators reclaimingCobalt compoundsLithium carbonateEconomic benefits
The invention discloses a method for recovering cobalt and lithium from waste lithium cobaltite and preparing lithium cobaltite. The method comprises the following steps: (1) leaching out the cobalt and lithium in the waste lithium cobaltite by using acid and a reducing agent, so as to obtain lixivium; (2) removing copper, iron, aluminum, calcium and magnesium in the lixivium by using a chemical method; (3) precipitating the cobalt and lithium in a solution without impurities with carbonate; (4) drying the cobalt lithium carbonate, and blending corresponding cobalt salt and / or lithium salt according to cobalt-lithium ratio; and (5) calcining so as to obtain lithium cobaltite product. The lithium cobaltite obtained by the method is good in property, cobalt and lithium recoveries are above 99% and above 96% respectively. The method has the advantages of simple process, low cost and high economic benefits, and is easy to carry out industrial production.
Owner:HUNAN BRUNP RECYCLING TECH
Polyaluminum calcium hydroxychlorides and methods of making the same
InactiveUS20080131354A1Improve efficiencyReduce sludge generationCosmetic preparationsChloride preparationAluminum hydrateHigh pressure
A process is provided for the preparation of a novel polyaluminum calcium hydroxychloride composition of enhanced efficiency for water treatment, paper sizing, and antiperspirant applications. The polyaluminum calcium hydroxychloride compositions are prepared via the acidification of higher basicity reaction products to form mid-to-high basicity final products. Two methods are provided for the preparation of higher basicity products, contemplated in the first, and an aqueous acid solution, such as hydrochloric acid, aluminum chloride, or a mixture thereof, is mixed with a strong alkaline calcium salt such as calcium oxide or calcium carbonate and aluminum powder at temperatures greater than 60° C. In a second method, bauxite, aluminum hydrate, or aluminum metal, and calcium aluminate are mixed with hydrochloric acid and are reacted at high temperatures and / or elevated pressures for a certain period of time whereby, when the reaction is complete, the mixtures are filtered to obtain clear solutions. The final PAC-Ca solutions may be dried to powder to have a wide range of basicities from about 40% to about 80%. At lower basicities, the polyaluminum calcium hydroxychloride sales are used preferably as antiperspirants, while at higher basicities, the solutions are used preferably as water treatment chemicals.
Owner:GENERAL CHEM LLC
Calcium Hydroxyapatite Based Sulfonate Grease Compositions and Method of Manufacture
An overbased calcium sulfonate grease composition comprising a reduced amount of overbased calcium sulfonate, calcium hydroxyapatite, base oil, one or more converting agents, and one or more complexing acids if a complex grease is desired. The calcium sulfonate grease composition improves thickener yield and expected high temperature utility as demonstrated by dropping point. A method of making the composition comprising the steps of mixing the calcium sulfonate and base oil, adding the calcium carbonate either before or after conversion, adding one or more converting agents, and adding one or more complexing acids. All or a portion of one or more of the complexing acids may be added with or prior to the one or more converting agents.
Owner:NCH CORP
Composite bone graft substitute cement and articles produced therefrom
ActiveUS7754246B2High mechanical strengthQuick changeAntibacterial agentsPeptide/protein ingredientsParticulatesTri calcium phosphate
The invention provides a particulate composition adapted for forming a bone graft substitute cement upon mixing with an aqueous solution, including i) a calcium sulfate hemihydrate powder having a bimodal particle distribution and a median particle size of about 5 to about 20 microns, wherein the calcium sulfate hemihydrate is present at a concentration of at least about 70 weight percent based on the total weight of the particulate composition; ii) a monocalcium phosphate monohydrate powder; and iii) a β-tricalcium phosphate powder having a median particle size of less than about 20 microns. Bone graft substitute cements made therefrom, a bone graft substitute kit comprising the particulate composition, methods of making and using the particulate composition, and articles made from the bone graft substitute cement are also provided.
Owner:AGNOVOS HEALTHCARE
Nano silver calcium alginate antibacterial dressing
InactiveCN1935268AWith liquid absorption functionIt also has the function of absorbing liquidAbsorbent padsBandagesFiberCalcium alginate
The present invention relates to a nano silver calcium alginate antibacterial dressing. It is a product made up by using calcium alignate fibre on which the nano silve granules are adsorbed, the nano silver content is 0.01-1% of total weight of said dressing. Besides, said invention also provides its preparation method and concrete steps.
Owner:上海川本卫生材料销售有限公司
Method of Manufacturing Calcium Sulfonate Greases Using Delayed Addition of Non-Aqueous Converting Agents
ActiveUS20160115416A1Quality improvementGreat thickener yieldThickenersAdditivesSulfonateDelay periods
A method of manufacturing an overbased calcium sulfonate grease comprising a reduced amount of overbased calcium sulfonate, water and at least one non-aqueous converting agent, where at least a portion of the non-aqueous converting agent is added after one or more delay periods relative to the addition of the water. A delay period may involve the period of time it takes to adjust the temperature of the mixture, a period of time during which the mixture is held at a temperature or within a range of temperatures, and multiples and any combination thereof. These calcium sulfonate greases have improved thickener yield and high dropping points compared to greases of substantially similar composition made without a delay between the additions of water and a non-aqueous converting agent, particularly when a poor quality overbased calcium sulfonate is used.
Owner:NCH CORP