Patents
Literature
147results about How to "Meet training requirements" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
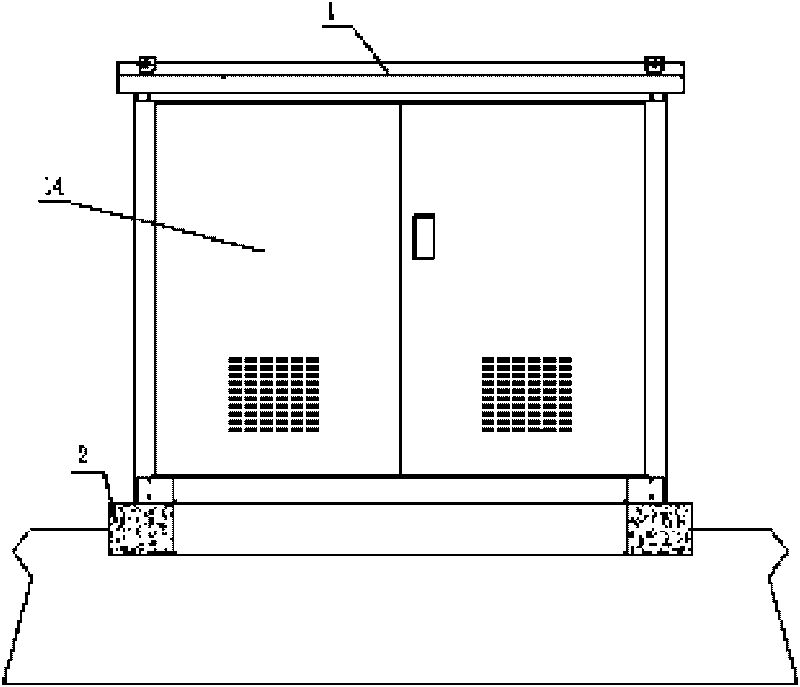
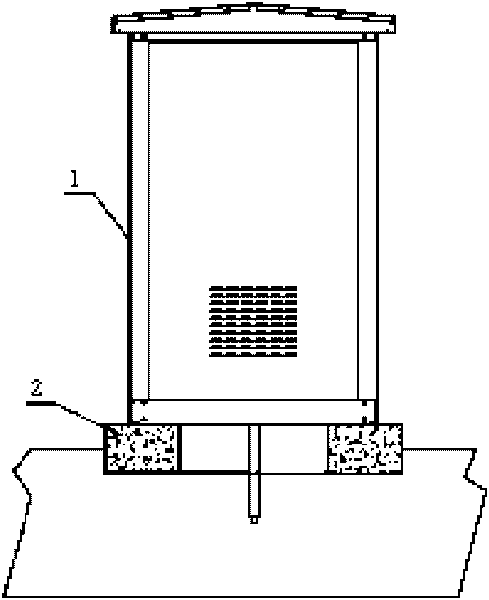
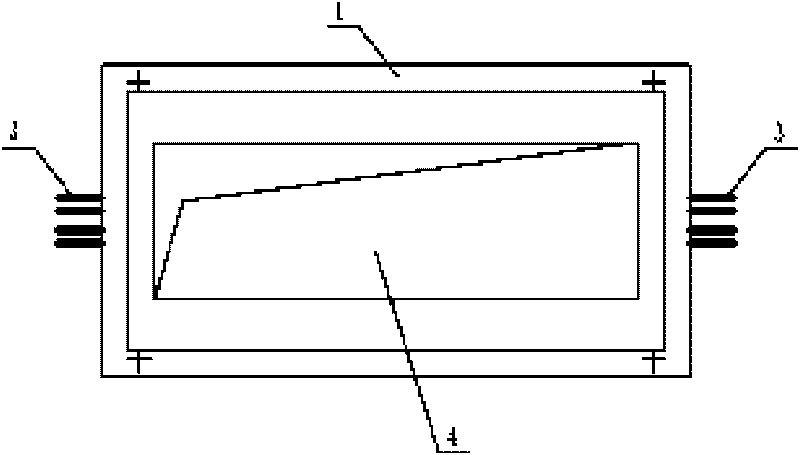
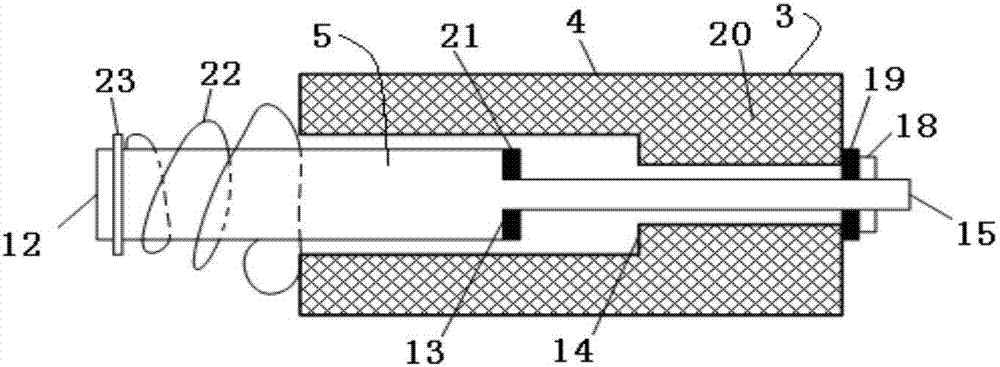
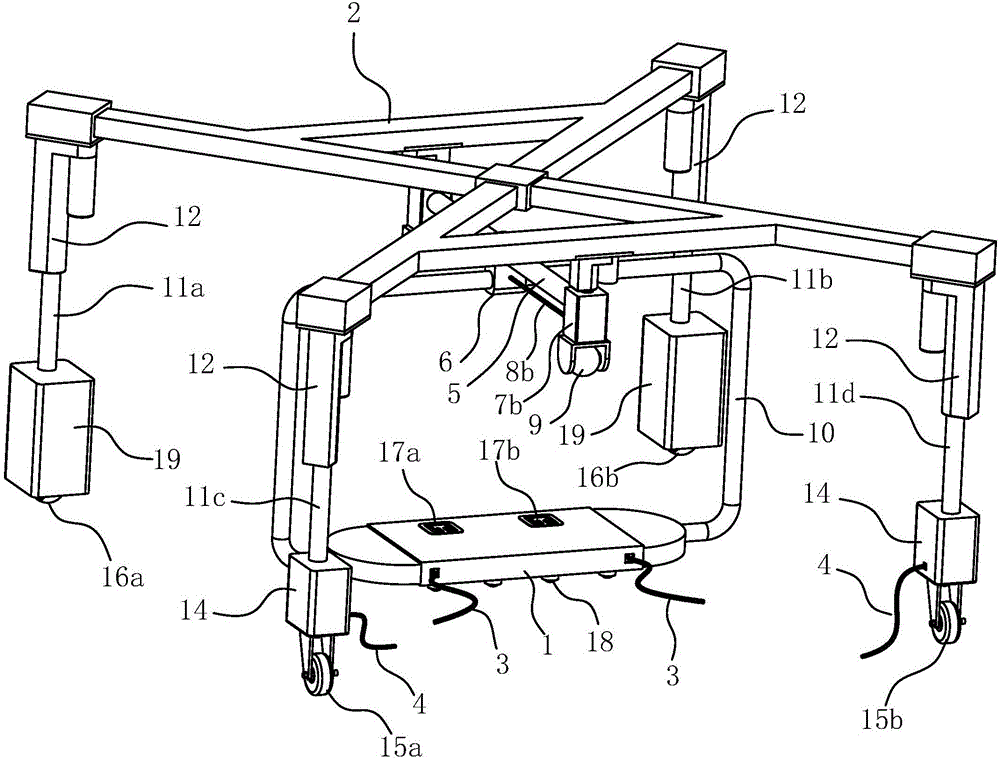
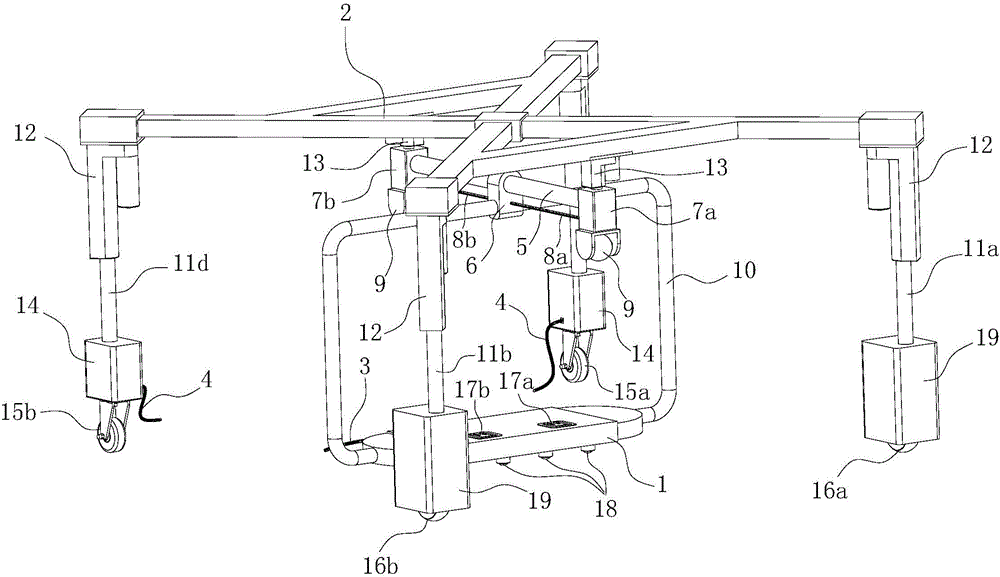
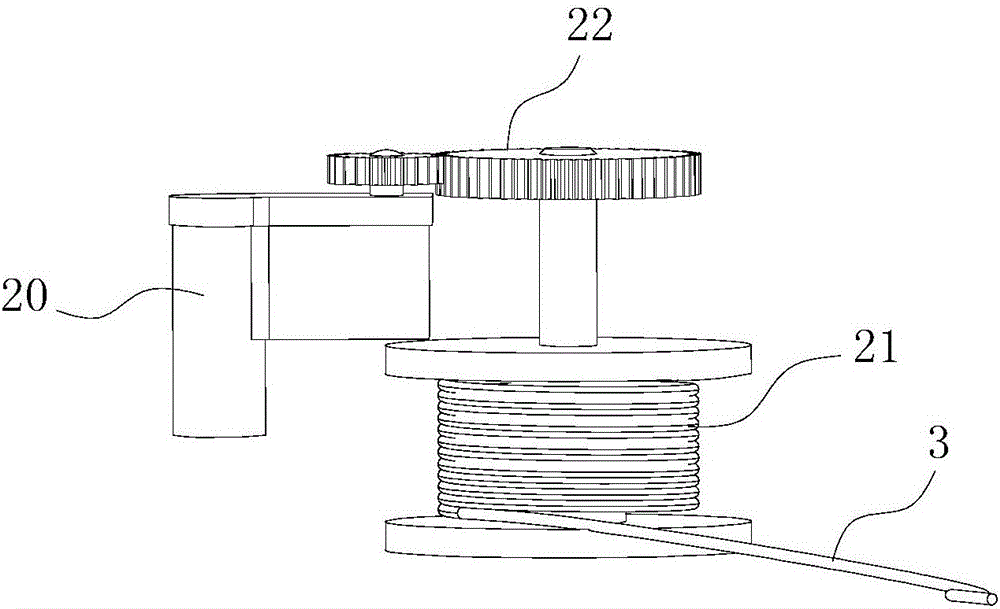
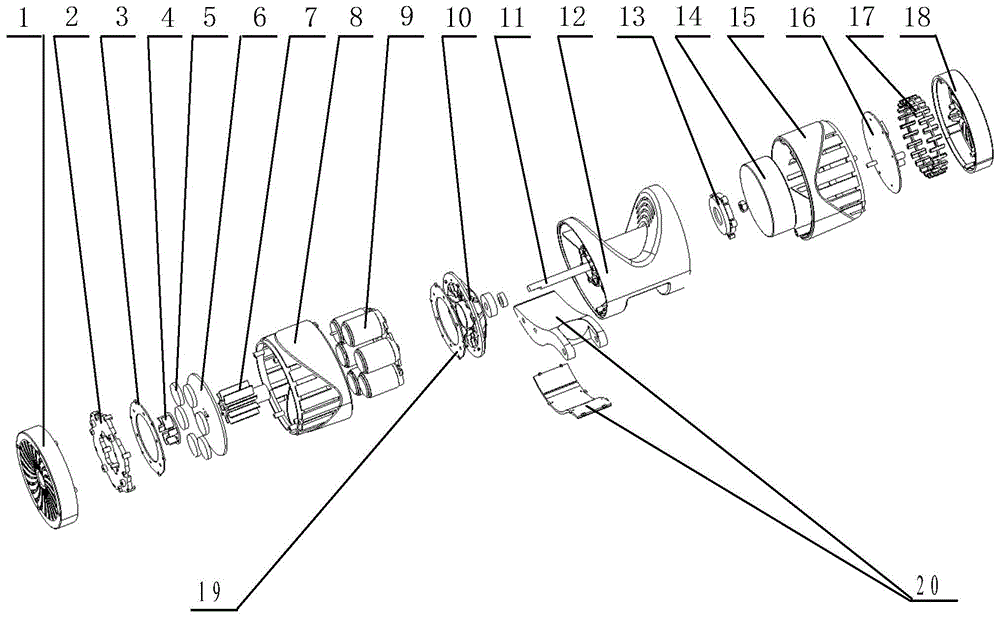
Cable fault simulation and location system
ActiveCN101699540AEasy to debugSimple structureEducational modelsFault location by pulse reflection methodsHigh-voltage cableEngineering
The invention relates to a cable fault simulation and location system, which comprises a cable fault simulation case, an electrode mounting plate arranged in the cable fault simulation case, three incoming line electrodes and three outgoing line electrodes, wherein the three incoming line electrodes are connected with a three-phase wire core in an incoming line cable respectively; the three outgoing line electrodes are connected with a three-phase wire core in an outgoing line cable respectively; a resistance box for simulating disconnection faults, low-resistance faults, high-resistance faults and flashover faults between the incoming line electrodes and the outgoing line electrodes is arranged in the cable fault simulation case; the tail end of the outgoing line cable connected with the outgoing line electrodes is provided with a high-voltage cable terminal; and the high-voltage cable terminal is used for connecting a cable fault location detector. The cable fault location detector comprises a central control unit, an impulse transmission circuit for transmitting impulse signals to a tested cable, a reflection receiving circuit for receiving reflected signals from the tested cable and a high-speed data acquisition circuit, wherein an output end of the reflection receiving circuit is connected with an input end of the high-speed data acquisition circuit.
Owner:CHANGZHOU POWER SUPPLY OF JIANGSU ELECTRIC POWER +1
Ganoderma lucidum potted landscape cultivation method
InactiveCN102318505AMeet training requirementsReliable growthHorticultureFertilizer mixturesAgricultural scienceGossypium
The invention provides a Ganoderma lucidum potted landscape cultivation method, which can solve the problem that the growth of Ganoderma lucidum fruit bodies is influenced because of insufficient nutrient compositions in substitute materials in the existing Ganoderma lucidum substitute material bag cultivation method, and can ensure the reliable growth of the Ganoderma lucidum fruit bodies at thesame time by controlling factors such as temperature, humidity, illumination and the like. The method comprises the following steps of medium preparation, medium bagging and sterilization, inoculation and mushroom development and fruit body culture management. The method is characterized in that cotton seed hulls are used as a culture medium which comprises 85 parts to 90 parts by weight of cotton seed hulls, 10 parts to 15 parts by weight of any one, any two or three of corn powder, wheat powder and soybean powder, and 1 part to 2 parts by weight of gypsum powder; or sawdust is used as the culture medium which comprises 90 parts to 92 parts by weight of sawdust, 8 parts to 10 parts by weight of any one, any two or three of corn powder, wheat powder and soybean powder and 1 part to 2 parts by weight of gypsum powder.
Owner:无锡飞凤生物科技有限公司
Culture medium for straw mushroom
ActiveCN103467187AMeet training requirementsReduce manufacturing costFertilizer mixturesDrug biotransformationMushroom
The invention provides a culture medium for straw mushroom, which comprises the following raw and auxiliary materials in parts by weight: 300-600 parts by weight of cane trash, 100-400 parts by weight of dregs, 0-200 parts by weight of straw, 0-200 parts by weight of corncob, 0-10 parts by weight of peanut bran, 5-10 parts by weight of compound fertilizer, 5-10 parts by weight of plaster, 5-8 parts by weight of lime, and 4-8 parts by weight of urea. According to the invention, cane trash and dregs, two primary by-products of the sugar industry, are used for cultivate the straw mushroom; the recipe is reasonable; the physical and chemical properties are reliable and can meet the cultivation requirement of the straw mushroom; the culture medium has the advantages of increasing comprehensive utilization of sugar refinery by-product, reducing straw mushroom manufacturing cost, reducing environment pollution, and accelerating the development of domestic fungus industry; the biotransformation ratio of straw mushroom cultivated through the culture medium is higher than 40%.
Owner:GUANGXI UNIV
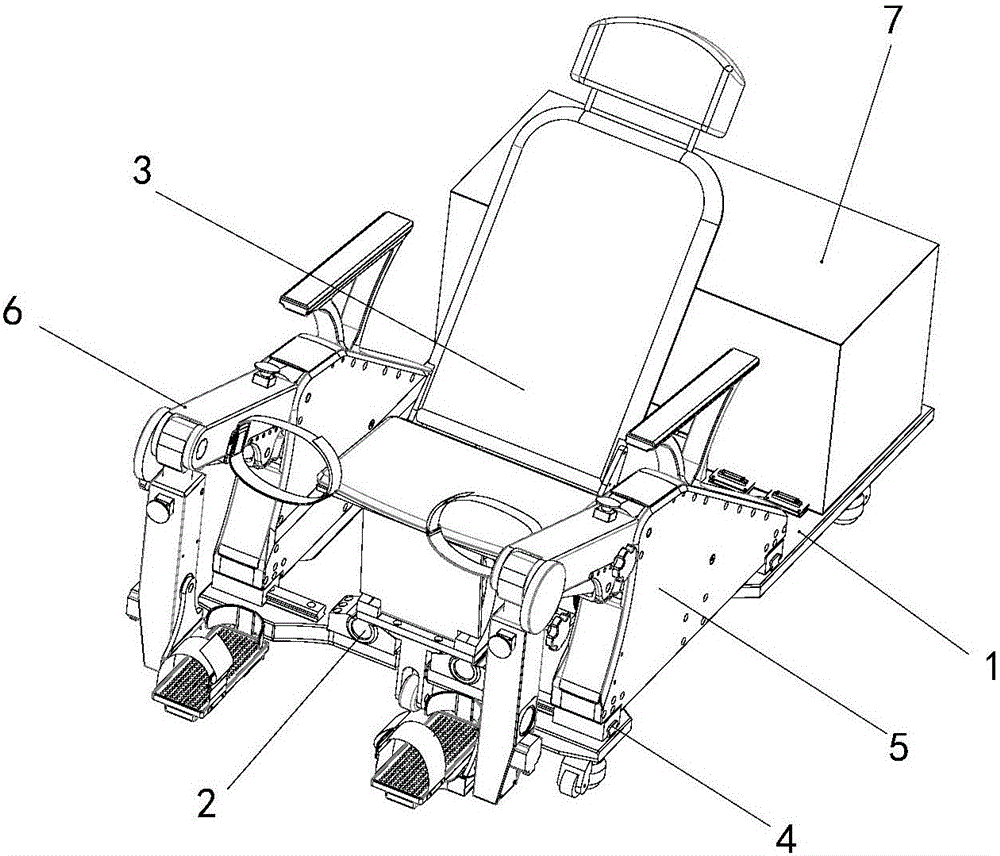
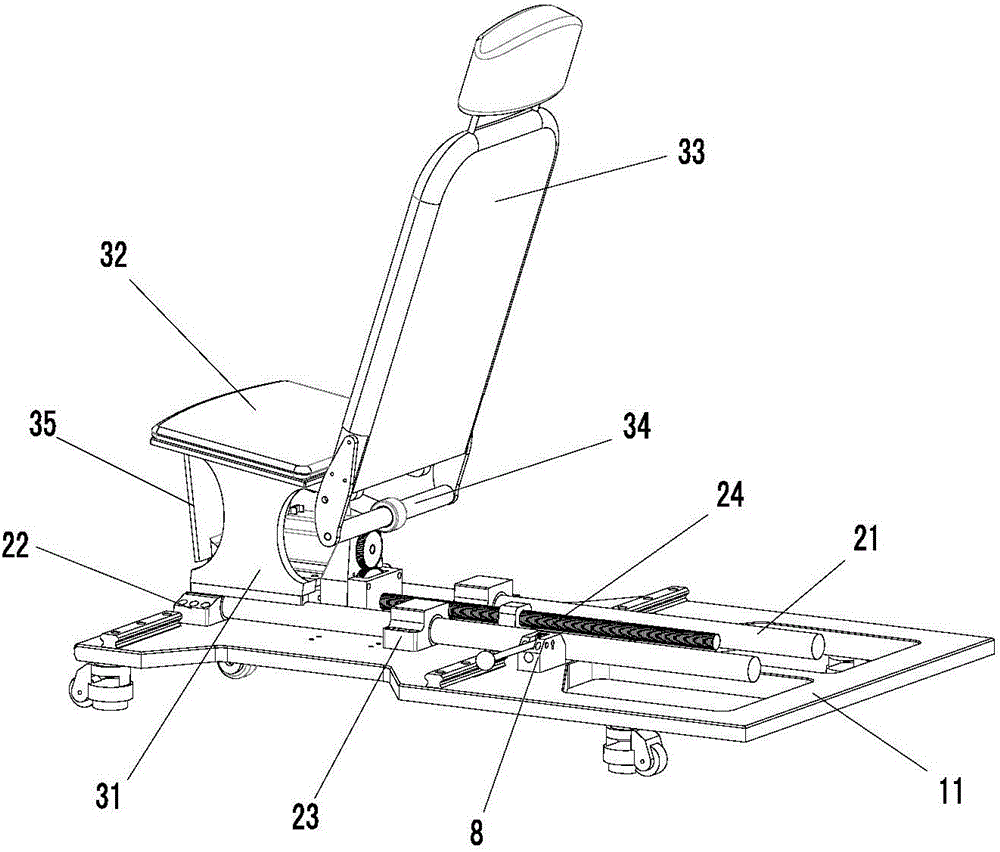
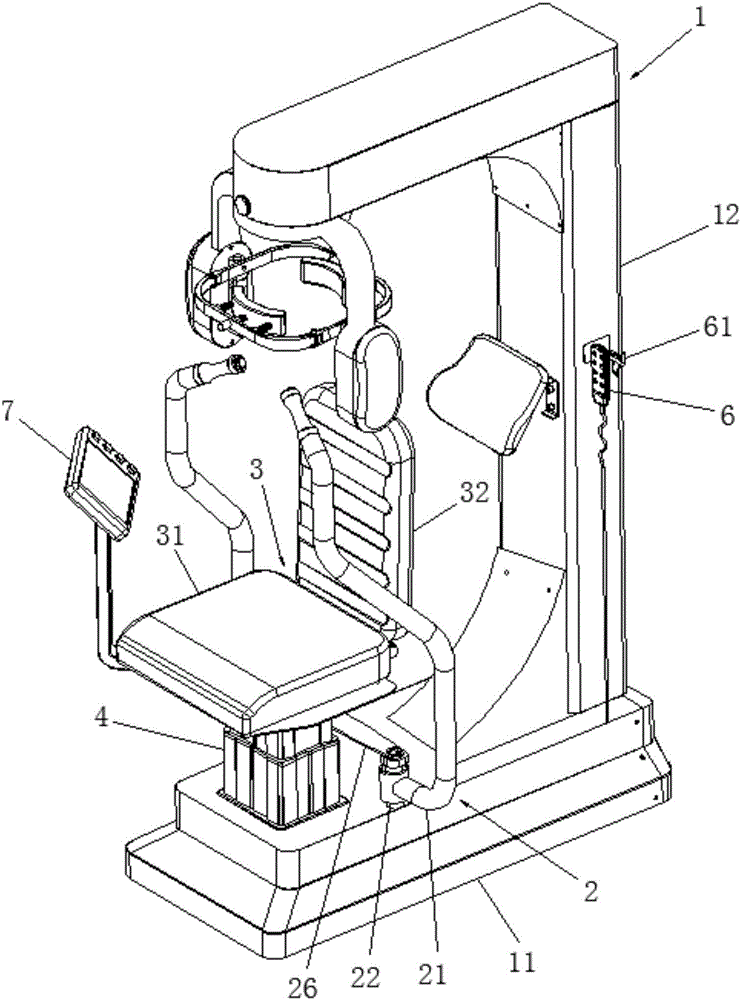
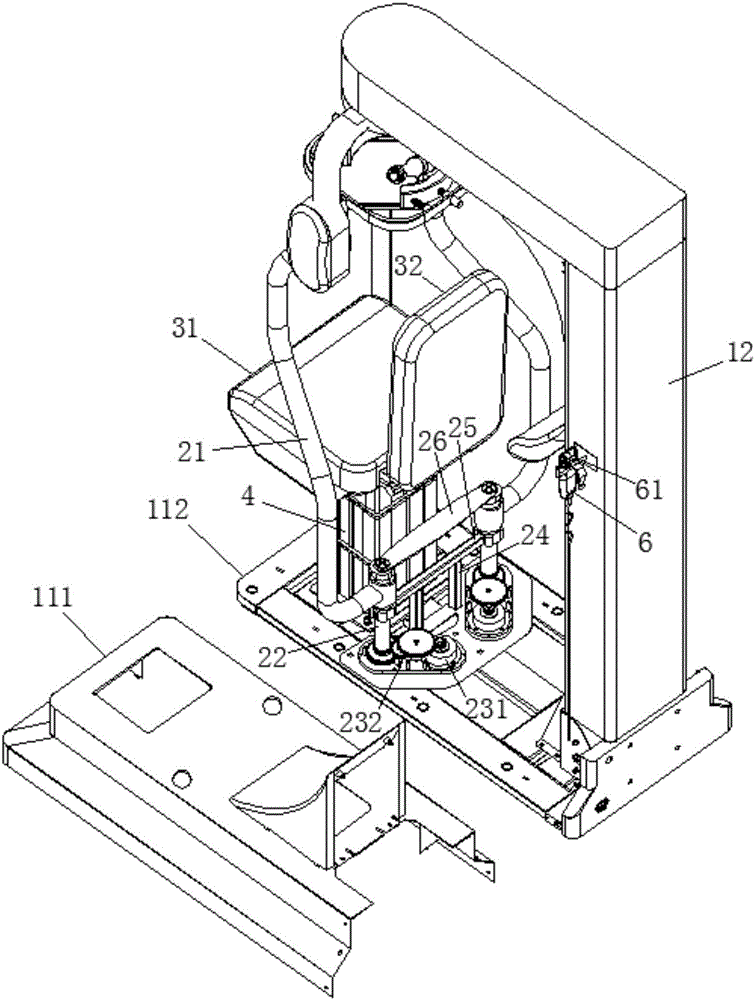
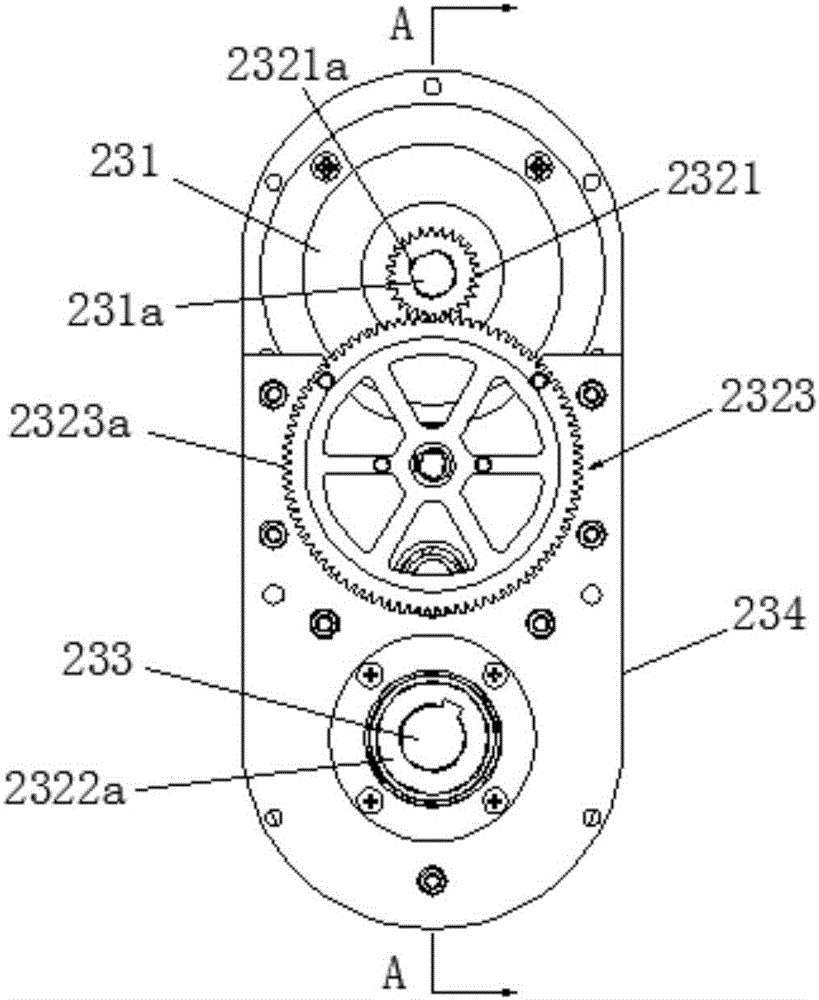
Lower limb exoskeleton rehabilitation training device provided with movable chair
A lower limb exoskeleton rehabilitation training device provided with a movable chair mainly comprises a base, a chair frontward and backward adjustment mechanism, the chair, a mechanical leg leftward and rightward adjustment mechanism, two mechanical leg fixing platforms, two mechanical legs and a counter weight control box. Because the mechanical leg leftward and rightward adjustment mechanism is arranged, the training requirements of training people having different widths between the hip joints can be met in the manner that the width between the two mechanical arms can be adjusted. The lower limb exoskeleton rehabilitation training device is combined with a chair movement and transmission mechanism, before a patient gets on the lower limb exoskeleton rehabilitation training device, the chair is moved to the front end of the base, and therefore the influence generated by the fact that the mechanical legs occupy space when the patient gets on and gets off the lower limb exoskeleton rehabilitation training device is overcome. Because thigh mechanisms, shank mechanisms and ankle joint mechanisms of the two mechanical legs are each provided with a drive mechanism, exercise training and movement range practices of one joint can be achieved, and coordinate exercise training of one leg and the double legs can also be achieved. Through independent hip joint adjusting mechanisms, independent knee joint adjusting mechanisms and independent ankle joint adjusting mechanisms, adjustment of the shanks and the thighs can be achieved, and therefore personalized training is achieved.
Owner:SHANDONG CLASSIC MEDICAL DEVICE TECH CO LTD
Primary culture method for liver cells of crucian carp
InactiveCN102304492AReduce contentSufficient quantityVertebrate cellsArtificial cell constructsRed blood cellDigestion
The invention provides a new primary culture method for liver cells of crucian carp, which mainly comprises the following steps: a, separating liver tissues from crucian carp; b, digesting cut liver tissues by using a stepwise digestion process and obtaining inner cell mass; and c, purifying and culturing obtained inner cell mass. In the method disclosed by the invention, the raw material is obtained from common crucian carp aquatic experimental animals, in-vitro tissue blocks are soaked in chlorhexidine gluconate (chlorhexidine) to be disinfected for reducing pollution. The tissues are digested by a stepwise digestion process, blood cell lysing solution is used to reduce the blood cell content in cells, and the liver cells are purified by percoll gradient centrifugation. The liver cells are purified by combined material preparation, the stepwise digestion process, blood cell lysing solution and percoll. The method can culture adequate liver cells, the survival rate of the liver cellsis over 90 percent, and the method meets requirements of primary culture.
Owner:SHANGHAI OCEAN UNIV +1
Traditional Chinese medicine extract capable of improving CIK cell proliferation rate as well as preparation method and application of same
InactiveCN102755512APrescription scouringQuality is easy to controlImmunological disordersAntineoplastic agentsBiotechnologyFormulary
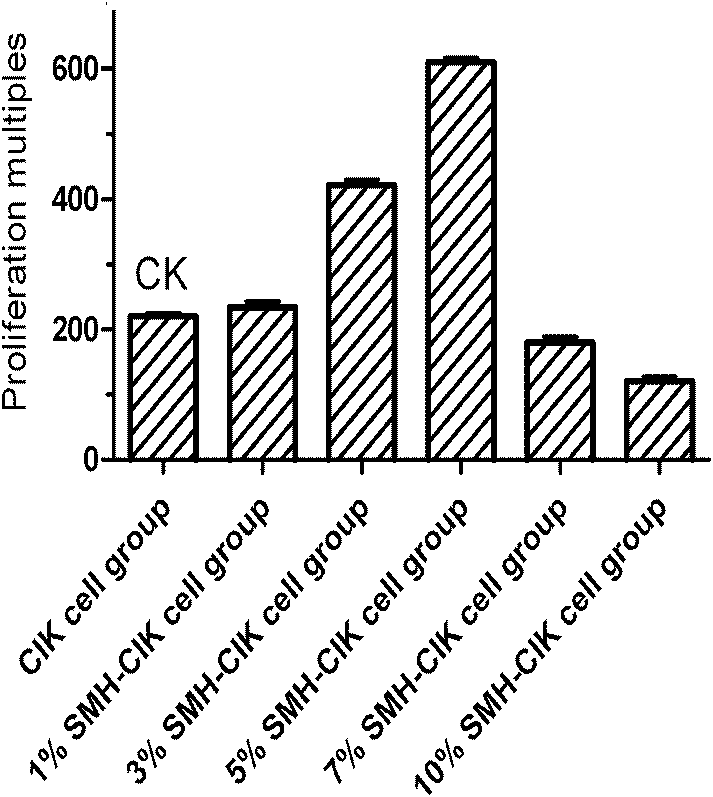
The invention relates to a traditional Chinese medicine extract capable of obviously improving CIK (Cytokine-Induced Killer) cell proliferation rate. The extract is prepared from the raw materials of red ginseng, radix ophiopogonis and milk vetch according to a certain weight ratio. A formula of the extract comprises the steps as follows: the red ginseng, the radix ophiopogonis and the milk vetchare added with water and decocted, and are subjected to ethanol precipitation and filtered; ethanol is recovered; the extract liquid is enriched with macroporous resin columns, eluted to be colorlesswith distilled water and eluted with ethanol; the eluant is collected; the extract liquid is filtered with a microporous filtration film; and the filter liquor is condensed, the pH value is adjusted,the filter liquor is filtered again with the microporous filtration film, sterilized, and subpackaged, and then the traditional Chinese medicine extract is obtained. Pharmacological experiments show that the traditional Chinese medicine extract can remarkably improve the proliferation rate of CIK cells.
Owner:CHINA JILIANG UNIV
Method for automatically identifying side slope rock types
InactiveCN110232419AMeet training requirementsRealize automatic identification and classificationCharacter and pattern recognitionNeural architecturesNetwork modelTest set
The invention discloses a method for automatically identifying the type of side slope rock. The classification of rock slope rocks is crucial to the analysis of slope stability. The conventional manual method is low in efficiency and is influenced by subjective factors. The method comprises the following steps of establishing a convolutional neural network model for rock slope image set analysis,performing feature information extraction and compression on 80000 rock slope images through convolution operation and pooling operation respectively, and then training the network model to realize automatic identification and classification of rock slope rocks. The rock slope images in the training set and the test set are adopted to test and analyze the model, the accuracy rate of the training set reaches 98%, the accuracy rate of the test set reaches 90%, it is shown that the trained network model has good robustness, and an ideal training effect can be achieved. Finally, a network model established by deep learning is adopted to realize the rapidness and automation of rock identification of the rock slope.
Owner:NORTHEASTERN UNIV
Belly loose muscle exercising and training device
InactiveCN109938993AAvoid strainPromote recoveryGymnastic exercisingVibration massageMuscle strainsFoot supports
The invention relates to a belly loose muscle exercising and training device which comprises a base plate, a fixing support, a foot support, an exercising device and an auxiliary device. The fixing support is installed in the middle of the upper end of the base plate. An annular groove is formed in the middle of the fixing support, a yoga ball is arranged inside the annular groove, the foot support is installed between the base plate and the left end of the fixing support, the exercising device is installed at the upper end of the fixing support, and the auxiliary device is installed between the base plate and the right end of the fixing support. The problems that when an existing belly exercising instrument is in use, the belly exercising effect is poor, vibration massage cannot be performed on the belly initiatively, loose muscles cannot be protected, and belly muscles of the user are likely to be strained can be solved, and the device has the advantages that the using effect is good, the requirement for training of different strengths can be met, vibration massage can be performed on the belly, meanwhile, the loose muscles can be protected, and belly muscle strains are avoided.
Owner:JILIN UNIV
Agaricus blazei murill cultivating base stock
ActiveCN103467188AMeet training requirementsReduce manufacturing costFertilizer mixturesSludgeAgaricus blazei
The invention discloses agaricus blazei murill cultivating base stock, which is prepared from the following raw auxiliary materials in part by weight: 300-600 parts of bagasse, 300-500 parts of lime sludge, 0-100 parts of straws, 0-200 parts of corncobs, 0-10 parts of peanut bran, 5-20 parts of compound fertilizer, 10-50 parts of gypsum, 20-50 parts of lime and 0-5 parts of urea. According to the invention, two kinds of main secondary products as bagasse and lime sludge in the sugar industry are adopted to cultivate agaricus blazei murill, the formula is reasonable, the physical and chemical properties are reliable, and the requirement for cultivating agaricus blazei murill is completely met, the base stock has the advantages of improving comprehensive utilization of secondary products of sugar factories, reducing production cost of agaricus blazei murill and environmental pollution and facilitating the development of edible mushroom industry, and the biotransformation efficiency when the cultivating base stock is adopted to cultivate agaricus blazei murill reaches 38% above.
Owner:GUANGXI UNIV
Fighting trainer
InactiveUS20120149534A1Less oscillating amplitudeRapidly bounce backBall sportsCombat sportsEngineeringElastance
Owner:FU DICKSON +1
Serum-free cartilage cell culture solution
ActiveCN104630138AClear outlineGood refractive indexSkeletal/connective tissue cellsGlucanTransferrin
A serum-free cartilage cell culture solution is obtained by adding exogenous additives to a basic culture solution. The basic culture solution is a DMEM culture solution or an F12 culture solution or a DMEM culture solution-F12 culture solution mixed culture solution, and the exogenous additives are nonessential amino acids, glucan, vitamin C, glutamine, sodium pyruvate, insulin or insulin-like growth factor, transferrin, growth hormone, triiodothyronine, hydrocortisone, dexamethasone, sodium selenite, beta-mercaptoethanol, lipid concentrate, progesterone, succinimide, a basic fibroblast growth factor, an epidermal growth factor, a transforming growth factor, a bone morphogenetic protein and a platelet derived growth factor respectively. The serum-free cartilage cell solution can avoid potential risks due to the use of serum, improves the extracellular matrix secreting ability of cartilage cells, makes the growth propagation state approach the growth propagation state of the cells in a serum-containing culture solution, and allows the dedifferentiation rate and the matrix secreting ability of low density inoculation cartilage cells to be obviously better than those of the cells in the serum-containing culture solution respectively.
Owner:SHAANXI RUISHENG BIOTECH
Agaricus bisporus culture medium material
ActiveCN103467189AReasonable formulaReliable physical and chemical propertiesFertilizer mixturesFertilizerSugar mill
An agaricus bisporus culture medium material is composed of the following raw materials and auxiliary materials in weight part: 200-500 parts of bagasse, 300-500 parts of filter mud, 0-200 parts of straw, 0-200 parts of maize stamen, 0-10 parts of peanut bran, 5-10 parts of compound fertilizer, 5-10 parts of gypsum, 5-8 parts of lime, and 4-8 parts of urea. According to the agaricus bisporus culture medium material, the main byproducts of the sugar industry, namely the bagasse and the filter mud are used for cultivating agaricus bisporus, the prescription is reasonable, the physical and chemical property is reliable, the requirement for culture of the agaricus bisporus is completely met, and the agaricus bisporus culture medium material has the advantages of capabilities of improving comprehensive utilization of byproducts of a sugar mill, lowering the production cost of the agaricus bisporus, reducing the environment pollution and facilitating development of the edible mushroom industry. The biotransformation efficiency of the agaricus bisporus cultured by utilizing the culture medium material can reach more than 65%.
Owner:GUANGXI UNIV
Upper limb training aid
ActiveCN106310603AImprove securityNo risk of strainMuscle exercising devicesPower flowUpper limb training
The invention discloses an upper limb training aid. The technical problem that an existing upper limb training aid is prone to injury a trainee is solved. The upper limb training aid comprises a rack, the rack comprises a base, two upper limb driving devices for being driven by arms are arranged on the base, and each upper limb driving device comprises a force applying handle, a rotary shaft connected with the force applying handle and a damping unit arranged in the base. Each damping unit comprises a magnetic damping motor outputting damping torque and a torque transmitting mechanism connected with the magnetic damping motor, the output ends of the torque transmitting mechanisms are connected with the rotary shafts, the upper limb training aid further comprises a master control system, the master control system comprises a CPU module and a control circuit, the input end of the control circuit is electrically connected with the CPU module, the output end of the control circuit is electrically connected with the magnetic damping motors, and the CPU modules control the magnitude of current input into the magnetic damping motors through the control circuit to control damping torque output by the magnetic damping motors.
Owner:PLA NAVY GENERAL HOSIPTAL +1
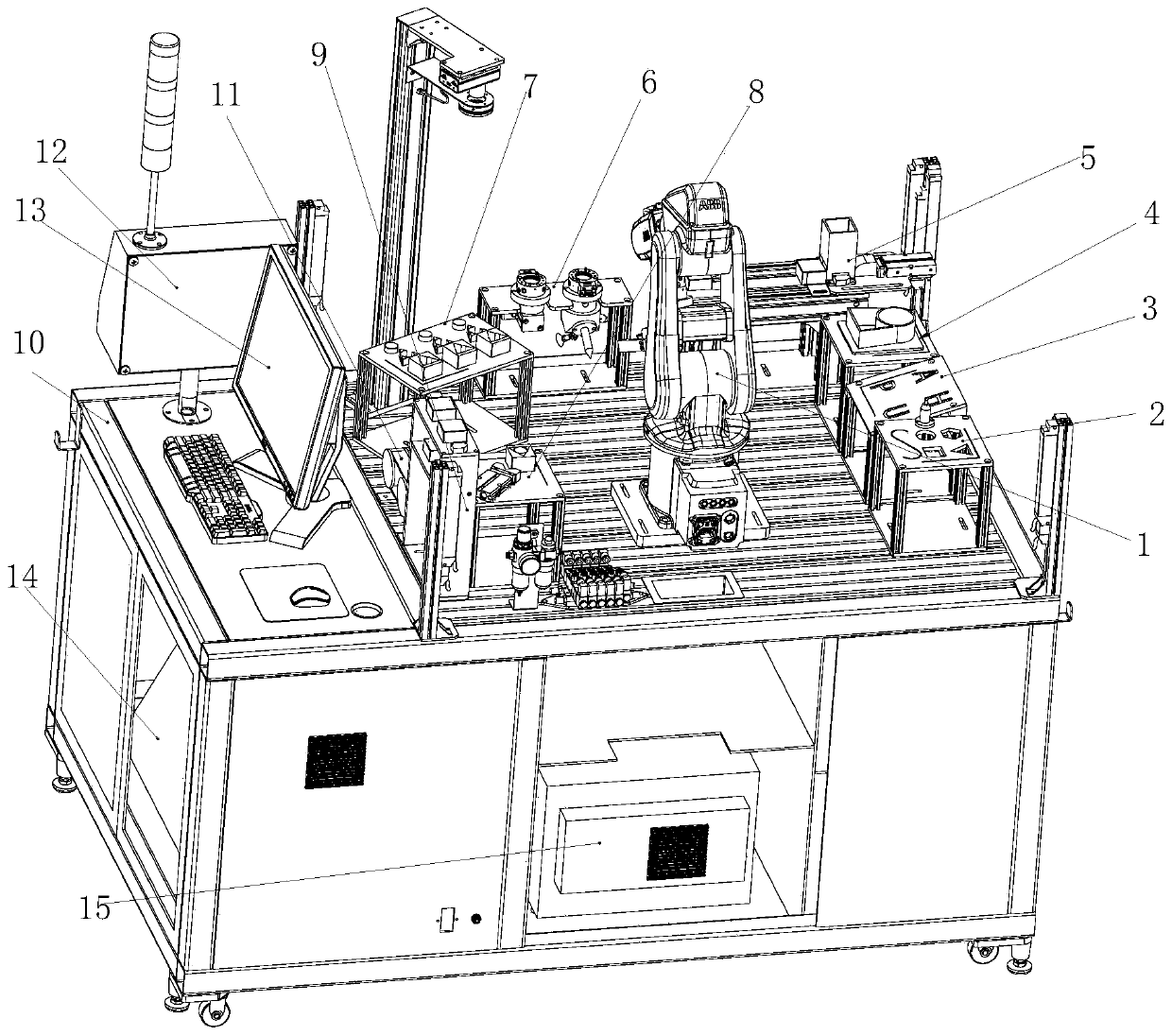
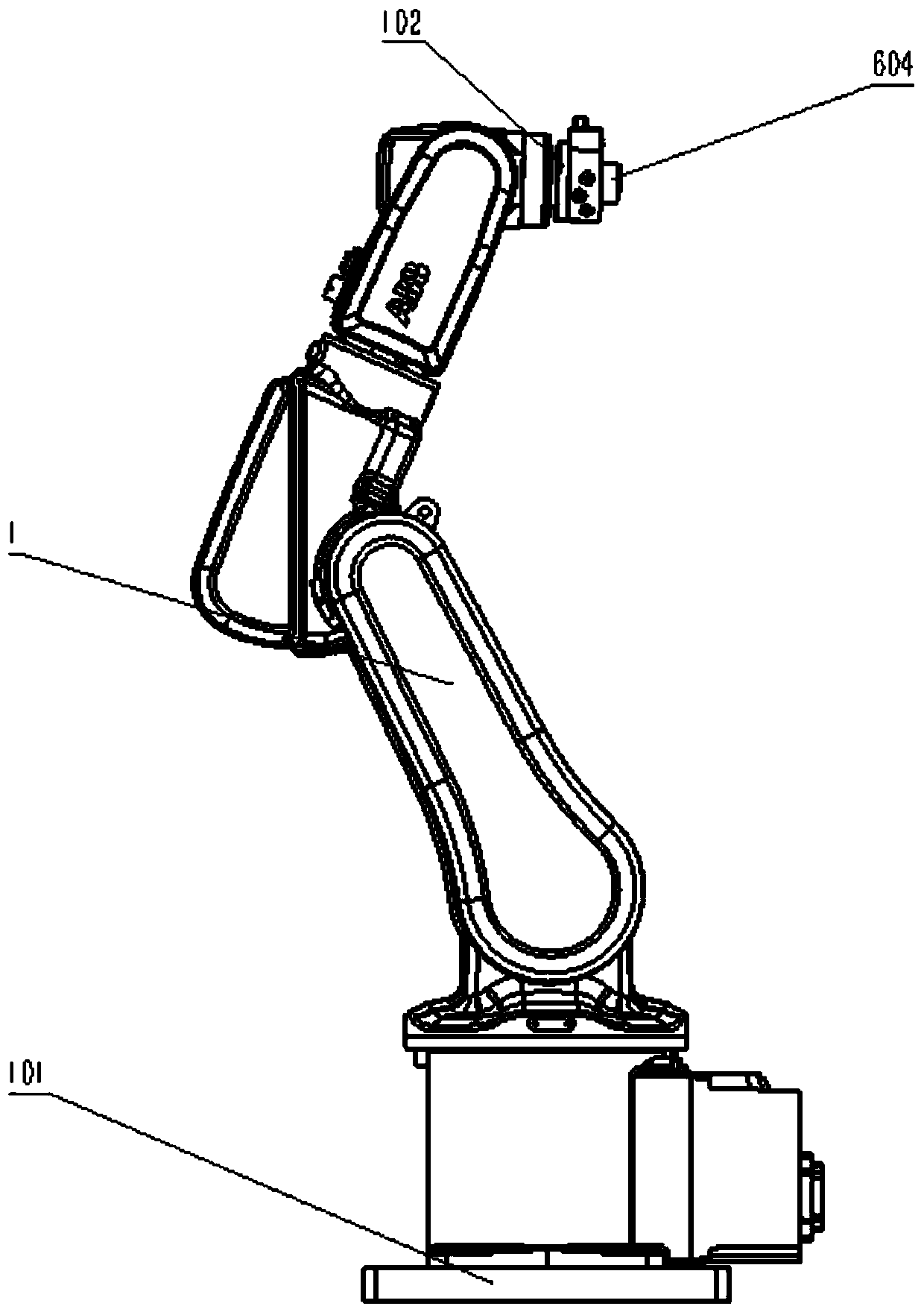
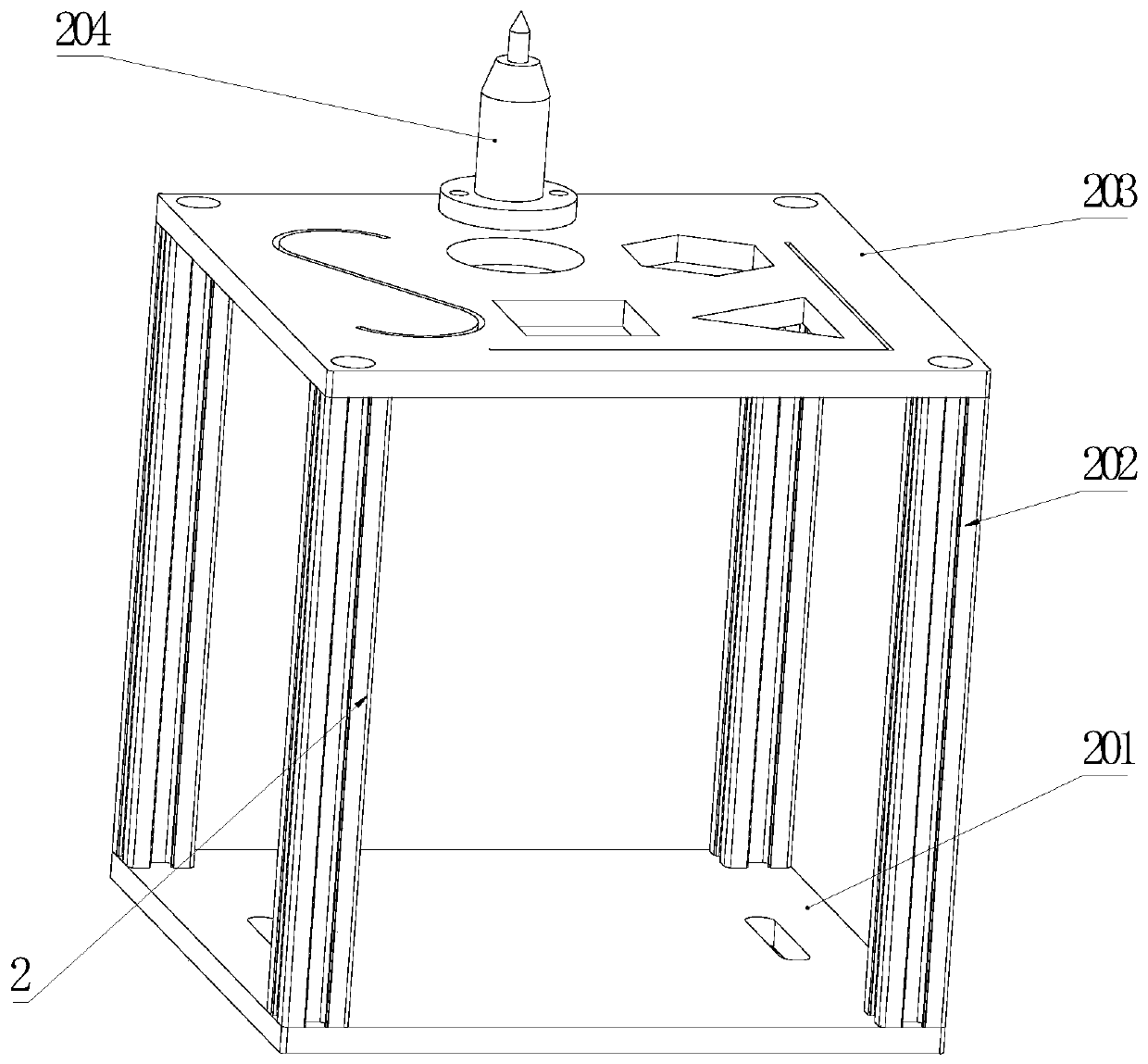
Industrial robot comprehensive practical training platform
PendingCN111445776AMeet training requirementsMeet basic teaching needsProgramme-controlled manipulatorGripping headsControl systemControl engineering
The invention relates to the field of robot teaching. The invention particularly relates to an industrial robot comprehensive practical training platform. The platform comprises an industrial robot, and a basic practical training module, a simulated laser engraving module, a simulated welding module, a simulated polishing module, a robot assembling module, a master control cabinet, a computer, a control system and a robot control cabinet which are circumferentially distributed around the industrial robot, the platform also comprises a quick-change clamp module, a robot vision module, an intelligent storage module, a conveying and sorting module, and a comprehensive practical training platform. According to the industrial robot comprehensive basic teaching platform, common application casesof an industrial robot are integrated into the design and layout of the practical training platform in a scene reproduction mode, basic teaching requirements can be met, various applications can be combined, modules are centralized, and functions are powerful; a clamp is additionally provided with an anti-collision sensor on the traditional basis, a safety light curtain is additionally arranged on a safety fence, double guarantees are achieved, and the practical training requirement of industrial robot technical talents is met.
Owner:WUHU ANPU ROBOT IND TECH RES INST +1
Apparatus capable of controlling density of CO2 for culturing growth of plant
InactiveCN101091444AControl concentrationHigh control precisionCarbon compoundsGreenhouse cultivationGrowth plantBuffer tank
The present invention discloses a plant growth culture equipment capable of controlling CO2 concentration. It includes a sealed plant growth chamber component with opening device. It is made of transparent material, one end of its outet tube is communicated with the internal cavity of said plant growth chamber component, and its another end is connected with CO2 gas balance component by successively means of three-way switching valve I, tube I, air pump and air outlet tube, the CO2 gas balance component is connected with internal cavity of said plant growth chamber component by successively means of tube II, air buffer tank, tube IV, CO2 detection component, tube III, three-way switching valve II and inlet tube; two ends of connecting tube are respectively connected with three-way switching valve I and three-way switching valve II. The application of said invention can raise accurate control of CO2 concentration.
Owner:ZHEJIANG UNIV
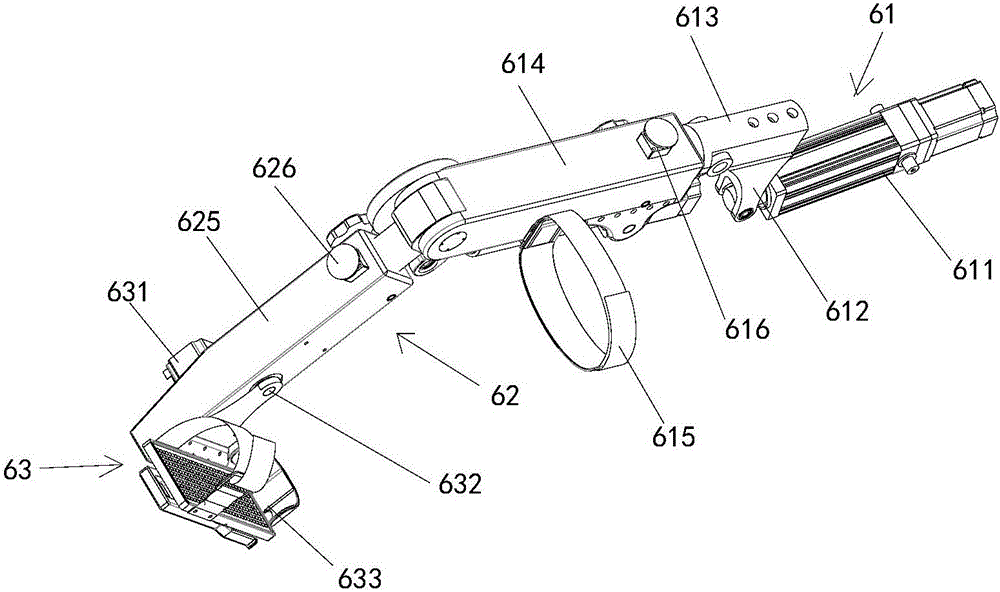
Laser simulation shooting device having one-way recoil force
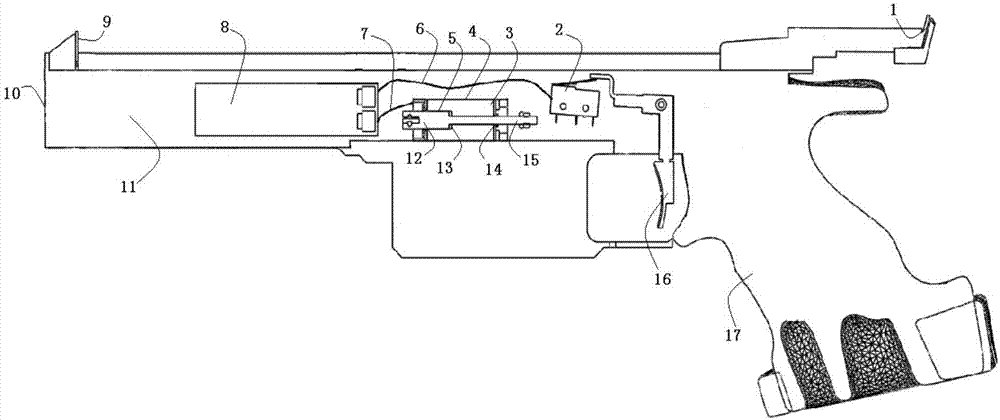
ActiveCN106871714AImprove laser simulation shooting experienceMeet training requirementsTraining adaptationClassical mechanicsGun barrel
The invention discloses a laser simulation shooting device having one-way recoil force. By providing real shooting recoil force feedback, the improvement of a laser simulation shooting experience of a user is facilitated, so that the training requirement of professional simulation shooting is met. The laser simulation shooting device is characterized in that the laser simulation shooting device comprises a gun barrel and a trigger; the gun-barrel is internally provided with a one-way recoil force executing mechanism; the one-way recoil force executing mechanism is connected with a control mechanism; and the control mechanism is connected with the trigger.
Owner:SUOOTER TECH BEIJING LTD
Industrial monitoring video image sharpening method based on GAN network
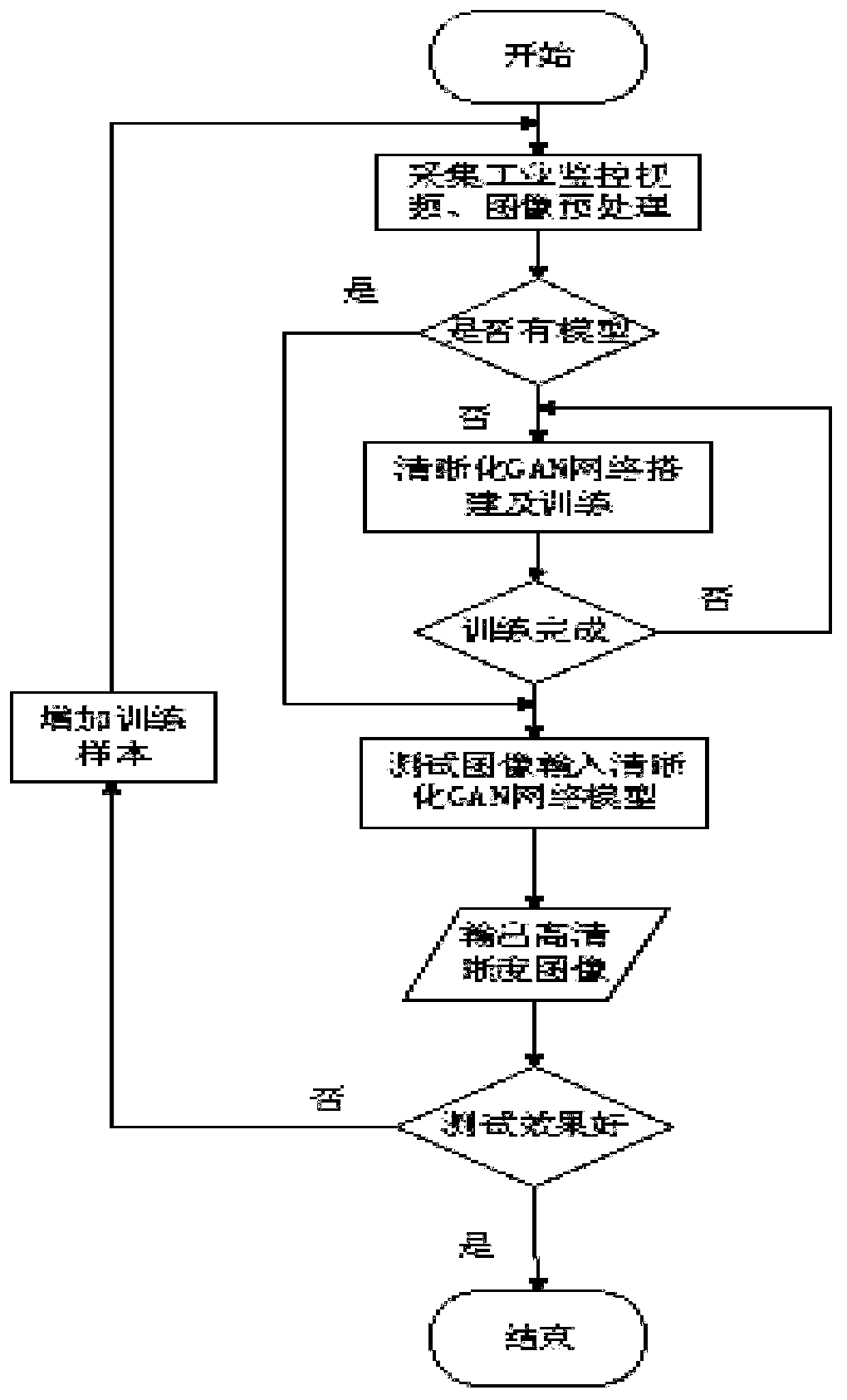
ActiveCN110660025ARecognizableRich in detailsImage enhancementImage analysisIndustrial monitoringEngineering
The invention discloses an industrial monitoring video image sharpening method based on a GAN network. The industrial monitoring video image sharpening method comprises the following steps: step 1, acquiring an original industrial video image; 2, preprocessing the image data; 3, detecting whether a trained model is contained or not; 4, building a GAN network; 5, perfomring model training; 6, performing model testing to obtain a corresponding high-definition image; 7, checking the test effect, and if the model can generate a high-definition image according to the test image, determining that the model training is better and can meet the actual application requirements; if the test effect is not good, restarting the step 1 to add training samples, and performing training again; the method has the advantages of simple training process, small model, good effect and high calculation efficiency, and is very suitable for recovery and sharpness processing of the monitoring image in a complex industrial environment.
Owner:XIAN UNIV OF TECH
Defect image sample generation method and device and panel defect detection method
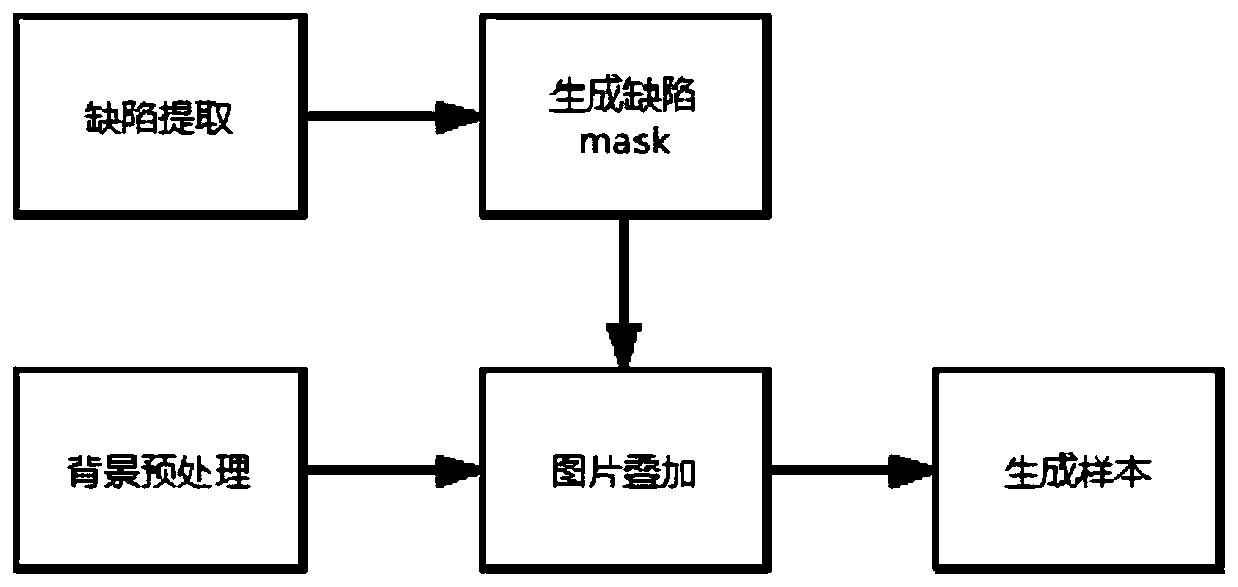
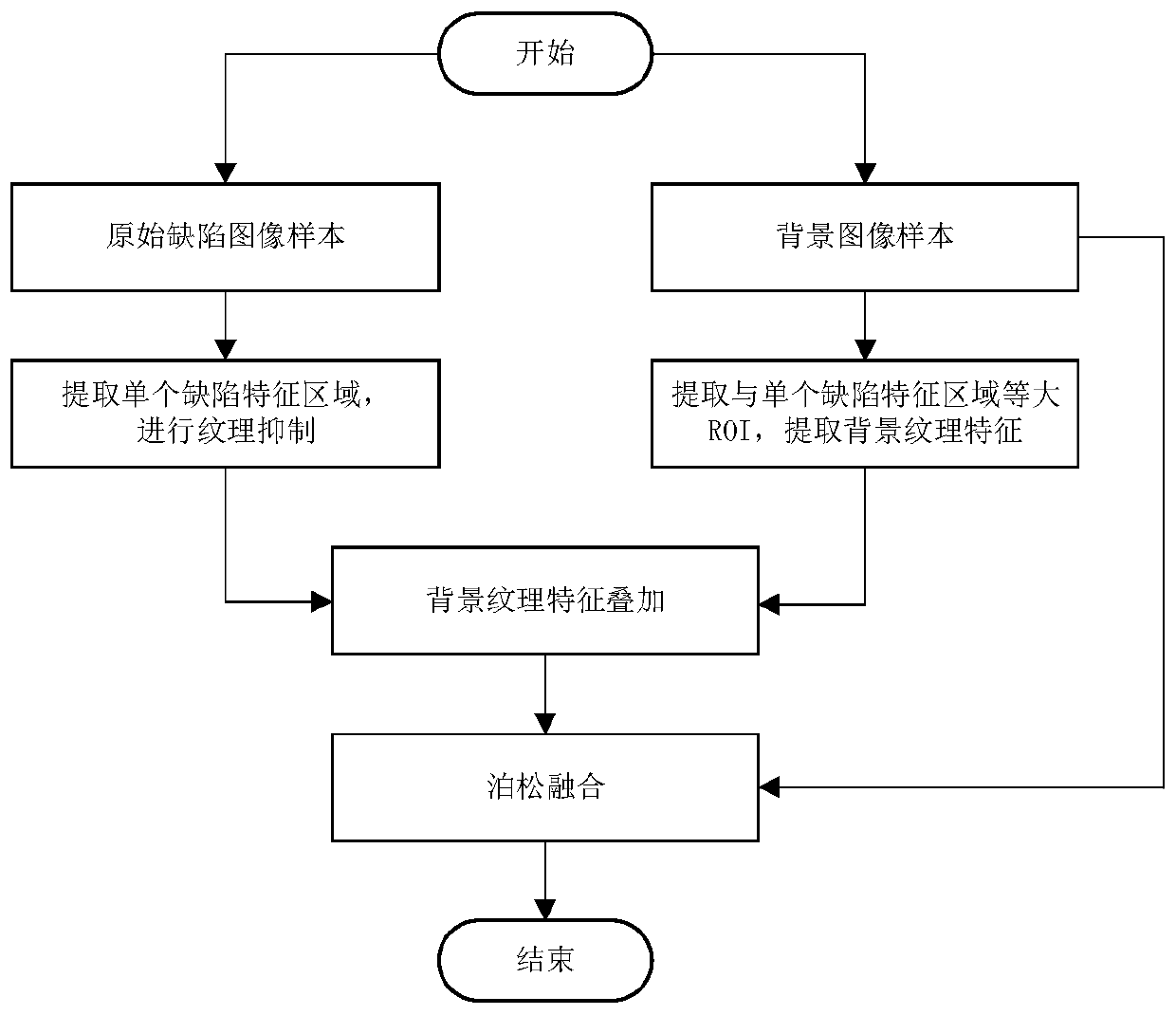
ActiveCN111524100AProminent visual defectGood fusion effectImage enhancementImage analysisRegion of backgroundDeep learning
The invention provides a defect image sample processing method and device and a panel defect detection method. The method comprises the following steps: extracting a defect feature region on an original defect image sample, and performing texture suppression on the defect feature region to obtain a first simulated defect image; extracting an ROI (Region Of Interest) equal to the defect feature region in area from the background image sample; extracting background texture features and superposing the background texture features into the first simulated defect image to obtain a second simulateddefect image; and finally, fusing the second simulated defect image and the background image sample to generate a defect image sample. According to the effective method for synthesizing the defect image sample by simulating any scene provided by the invention, the defect area is enabled to have the texture of the existing background through a background texture feature extraction and coverage method, the pixel distribution is closer to the background area, and the image is more natural; therefore, the generated defect image sample can meet the training requirement of a deep learning model, andthe problem of insufficient training samples is solved.
Owner:WUHAN JINGLI ELECTRONICS TECH +1
Multifunctional three-wheel ball serving machine
InactiveCN108543296AMeet different serving requirementsMeet training requirementsSport apparatusChinese charactersEngineering
The invention discloses a multifunctional three-wheel ball serving machine, which comprises a master control module, a launching mechanism, an angle regulating mechanism and a storage mechanism, wherein the launching mechanism is connected to the angle regulating mechanism and the storage mechanism; the launching mechanism, the angle regulating mechanism and the storage mechanism are connected tothe master control module; the launching mechanism comprises a first ball serving wheel, a second ball serving wheel and a third ball serving wheel which are mutually independent in rotating speed; the first ball serving wheel, the second ball serving wheel and the third ball serving wheel are arranged in a pin-shaped form (a Chinese character, pin), so that a ball serving passage from which ballsare launched is defined; and the first ball serving wheel, the second ball serving wheel and the third ball serving wheel are used for squeezing and rubbing the balls by rotating speeds which are mutually different, so that the balls are launched in different ball serving modes. According to the multifunctional three-wheel ball serving machine provided by the invention, conventional fixed-point ball serving can be achieved, and in addition, such functions as launching leftwards and rightwards rotating balls, random ball, side-rotating balls, high-pressure balls and the like can be achieved; therefore, user's training requirement can be satisfied.
Owner:WUYI UNIV
Condensed water-free multilayer culture shelf
InactiveCN103329806BEasy to self-regulateThe distance is easy to adjustPlant tissue cultureHorticulture methodsThermal insulationTransmittance
The invention discloses a condensed water-free multilayer culture shelf. One culture unit of the condensed water-free multilayer culture shelf comprises a frame, a workbench, a light source frame, a fan and spring bolts. Characteristics are that: the frame is provided with holes at equal intervals; the workbench is formed by thermal insulation boards, and a space is arranged between the upper thermal insulation board and the lower thermal insulation board; the light source frame is arranged 3 to 5cm away from the upper thermal insulation board of the workbench; a fan board provided with the fan is arranged right above the light source frame and is close to the light source frame; and four corners under the light source frame are provided with four spring bolts respectively. The condensed water-free multilayer culture shelf is capable of making up deficiencies of existing technologies; compared with existing culture shelf, the condensed water-free multilayer culture shelf enables that no condensed water is generated on culture dish walls, so that the number of vitrified plantlets is decreased, light transmittance is increased, and the demand of plantlets for a certain illumination intensity is satisfied.
Owner:SHANGHAI JIAO TONG UNIV
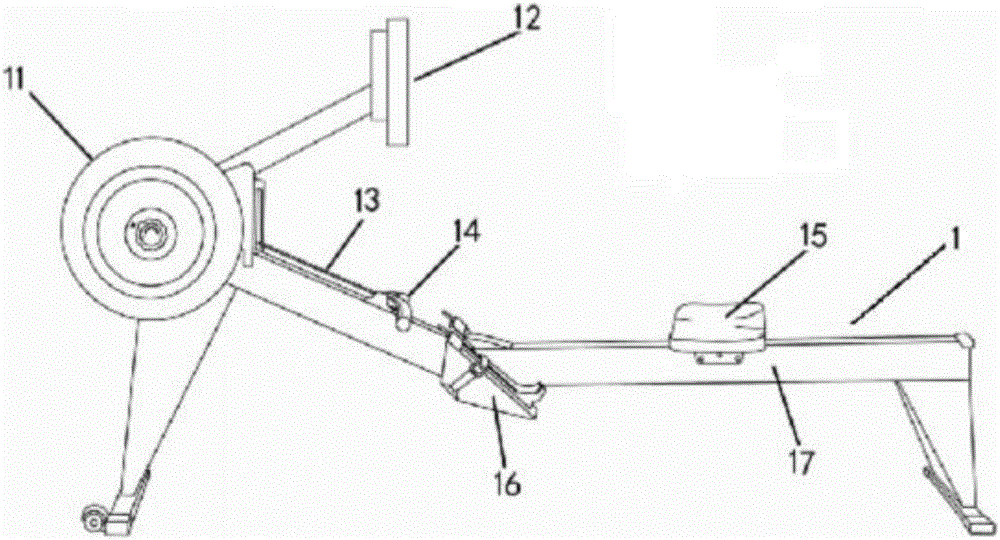
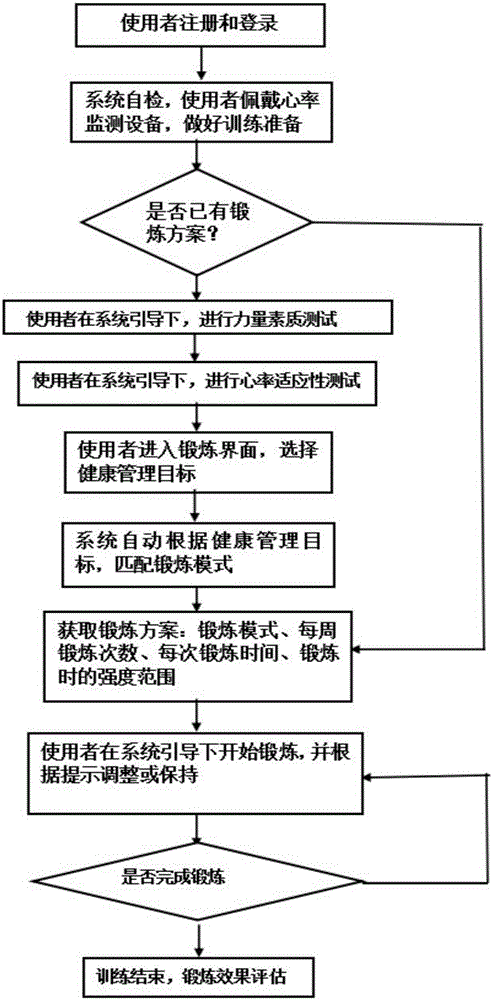
Body building method based on indoor rowing machine and heart rate monitoring
InactiveCN106512293AEfficient and safe fitness processMeet training requirementsMovement coordination devicesCardiovascular exercising devicesPhysical medicine and rehabilitationControl system
The invention discloses a body building method based on an indoor rowing machine and heart rate monitoring. Strength quality parameters of a user exercising in the indoor rowing machine are acquired, the heart rate applicability level of a user in unit time and under various strength requirements is monitored, the best exercise model, the weekly exercise frequency, exercise time for each time and the exercise strength range of the user are acquired in a pattern matching mode according to the health management objective of the user, parameters such as the dynamic heart rate, the oaring strength, the oaring frequency, consumed calories and power of the user are recorded in exercises, acoustic / optical / electric signals are used for guiding the user to do scientific and safe body-building exercises, the user can finish strength quality and heart rate applicability testing only by conforming to real-time guiding of a control system, the user can conveniently do exercises according to the body building method given by the system, and the body building method is conveniently popularized in the field of popular body building.
Owner:合肥中科博谐科技有限公司
Infant crawling trainer and control system and control method thereof
ActiveCN106621202ASo as not to damageWon't hurtDiagnosticsGymnastic exercisingBrain developmentMuscle strength
The invention belongs to the field of medical rehabilitation machinery and particularly relates to an infant crawling trainer and a control system and control method thereof. The trainer adopts two working modes, wherein the mode I is a function of training a crawling posture of a newborn infant, the mode II is a function of assisting crawling of an older infant, the training requirements of different ages of infants are met, and the usage value of the trainer is increased. The mode I can replace the work of an adult in newborn infant crawling training and can make infant crawling training unmanned, one adult can care multiple infants simultaneously, and the crawling training efficiency is improved. The mode II can give an assisting force to an infant who already masters the crawling posture but cannot independently complete crawling because of other reasons in crawling, so that the infant masters a crawling skill in the crawling process and exercises muscle strength, the posture keeping ability is improved, and brain development is promoted. The crawling mastering ability of a normal infant can be improved, and the curing possibility of infants possibly suffering from cerebral palsy can be also improved.
Owner:HEFEI UNIV OF TECH
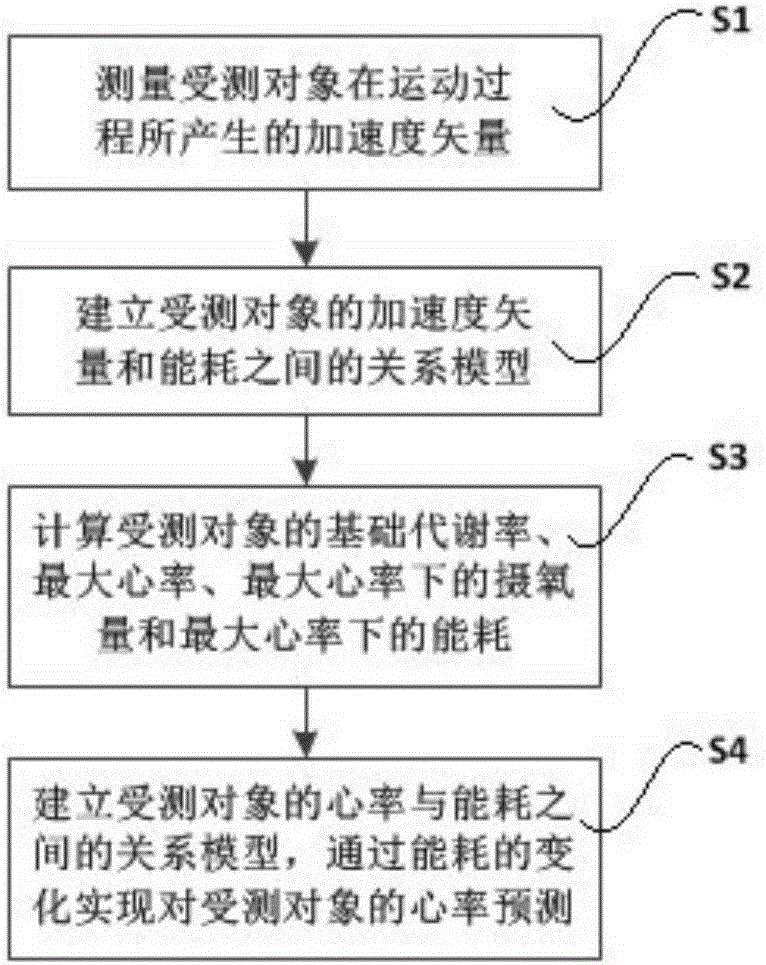
Exercise process heart rate predicting method and device based on acceleration sensor
ActiveCN105286842AEasy to operateMeet training requirementsMeasuring/recording heart/pulse rateRelational modelMedicine
The invention provides an exercise process heart rate predicting method and device based on an acceleration sensor. The exercise process heart rate predicting method based on the acceleration sensor comprises the following steps that 1, acceleration vectors generated by a measured object in the exercise process are collected and calculated through the acceleration sensor; 2, by analyzing the changes of acceleration vector values in a time axis, the energy consumption changes of the measured object in the exercise process are calculated, and a relation model of the acceleration vectors and energy consumption is built; 3, basic information of the measured object is collected, the basal metabolic rate, the maximum heart rate, the oxygen intake under the maximum heart rate and the energy consumption of the measured object are obtained through calculation; 4, a relation model of the heart rate and the energy consumption of the measured object in the exercise process is built, and prediction for the heart rate of the measured object is achieved. According to the exercise process heart rate predicting method and device based on the acceleration sensor, the heart rate of the measured object in the exercise process can be predicted through the acceleration sensor, the method is simple and easy to operate, and the measuring precision can meet the training requirements.
Owner:SHEN ZHEN FENGJING NETWORKS TECH CO LTD +1
Free combat training bag with adjustable softness, height and angle
ActiveCN111530050AEasy to operateMeet training requirementsSport apparatusElectric machineryRubber ring
The invention discloses a free combat training bag with adjustable softness, height and angle. The training bag comprises a base and a leather training bag, and the bottom of the leather training bagis connected with the top of the base; a rubber ring is arranged in the leather training bag; the bottom of the base is connected with a rotating motor; the bottom of the rotating motor is connected with a base plate; the bottom of the base plate is connected with a linear motor; the bottom of the linear motor is connected with a fixed seat connected with the ground; three fixing rods are arrangedon the top face of the fixing base in a circumferential array with the axis of the fixing base as the center, through holes corresponding to the fixing rods respectively and matched with the fixing rods in diameter are formed in the base disc, arc-shaped holes corresponding to the fixing rods are formed in the base, and the width of the arc-shaped holes is matched with the diameters of the fixingrods. A strip-shaped inflation bag is arranged in the center of the leather training bag, and an air pump connected with the inflation bag through an inflation pipe is arranged at the bottom of the base. The free combat training bag is adjustable in softness, height and angle, and can be suitable for trainees with different horizontal abilities and figures.
Owner:LIAONING INST OF SCI & TECH
Intelligent football training system
InactiveCN106422259AGuaranteed light intensityMeet training requirementsGymnastic exercisingBall sportsRadio frequencyLoudspeaker
The invention discloses an intelligent football training system. The system comprises a football training ground frame, a data collection controller, external equipment and a single chip microcomputer controller. A fixed plate is fixedly installed at the top of the football training ground frame; a protective net is fixedly installed at the bottom of the fixed plate; a recovery groove is formed in the bottom of the protective net; the data collection controller is composed of a heart rate sensor, a speed sensor and an acceleration sensor; the input end of the external equipment is connected with the output end of a wireless radio frequency receiving and transmitting module through the Internet; the input end of the single chip microcomputer controller is electrically connected with the output end of a light intensity sensor, the output end of a served ball counting device and the output end of a power supply module; the output end of the single chip microcomputer controller is electrically connected with the input ends of a football launcher, a loudspeaker, a camera, first illuminating lamps, second illuminating lamps, third illuminating lamps and LED flickering lamps. The intelligent degree is high, and body motion parameters are combined with training.
Owner:HUNAN UNIV OF SCI & ENG
Balance rehabilitation training robot
ActiveCN108096794AMeet training requirementsEasy to operateStiltsAgainst vector-borne diseasesEngineeringRehabilitation training
The invention discloses a balance rehabilitation training robot. The rehabilitation training robot comprises a control panel device, an overhung projector and a balance training device, wherein the balance training device comprises two balance training platform device for a left foot or a right foot to train independently or for the left foot and the right foot to train cooperatively; each training platform device comprises a retractable platform and a sliding mechanism arranged on the bottom of the retractable platform; each retractable platform comprises a knee protecting device, a pedal plate device, three retractable devices, a center pillar device and a bottom fixing plate; each center pillar device and the corresponding retractable devices are arranged on the corresponding bottom fixing plate; each pedal plate device is arranged above the corresponding center pillar device and the corresponding retractable devices; each knee protecting device is arranged above the corresponding pedal device; and a patient stands above the pedal devices, and rehabilitation training in various modes can be implemented automatically or by others. Independent movement or combined movement in pairs can be implemented, twenty to thirty different training modes can be achieved, and various training requirements are met. The balance rehabilitation training robot is simple to operate and high in practicality, scene interaction is carried out on projection of a projector, and thus, the balance rehabilitation training robot is quite interesting.
Owner:GUANGDONG YISHENG IND CO LTD
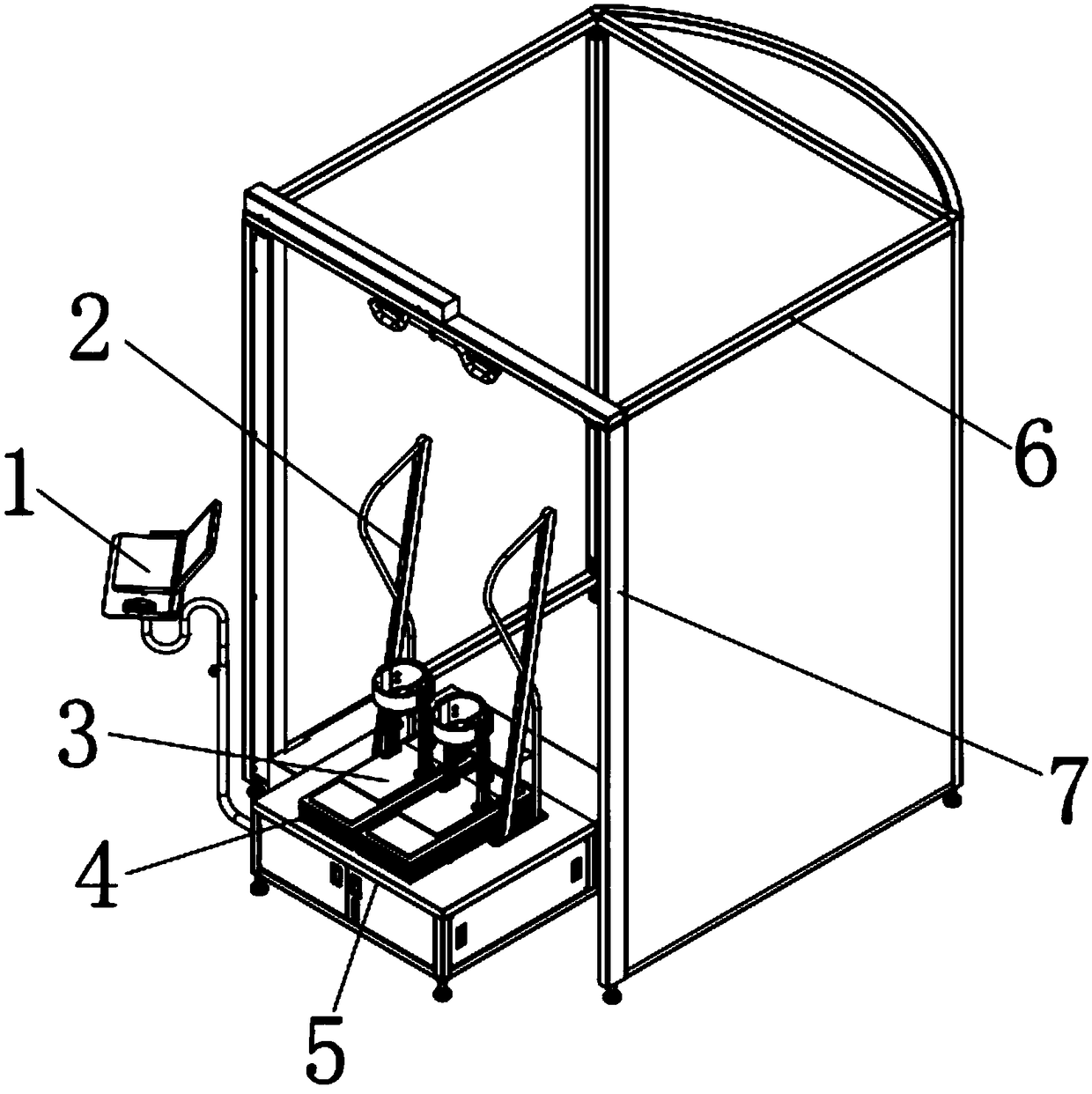
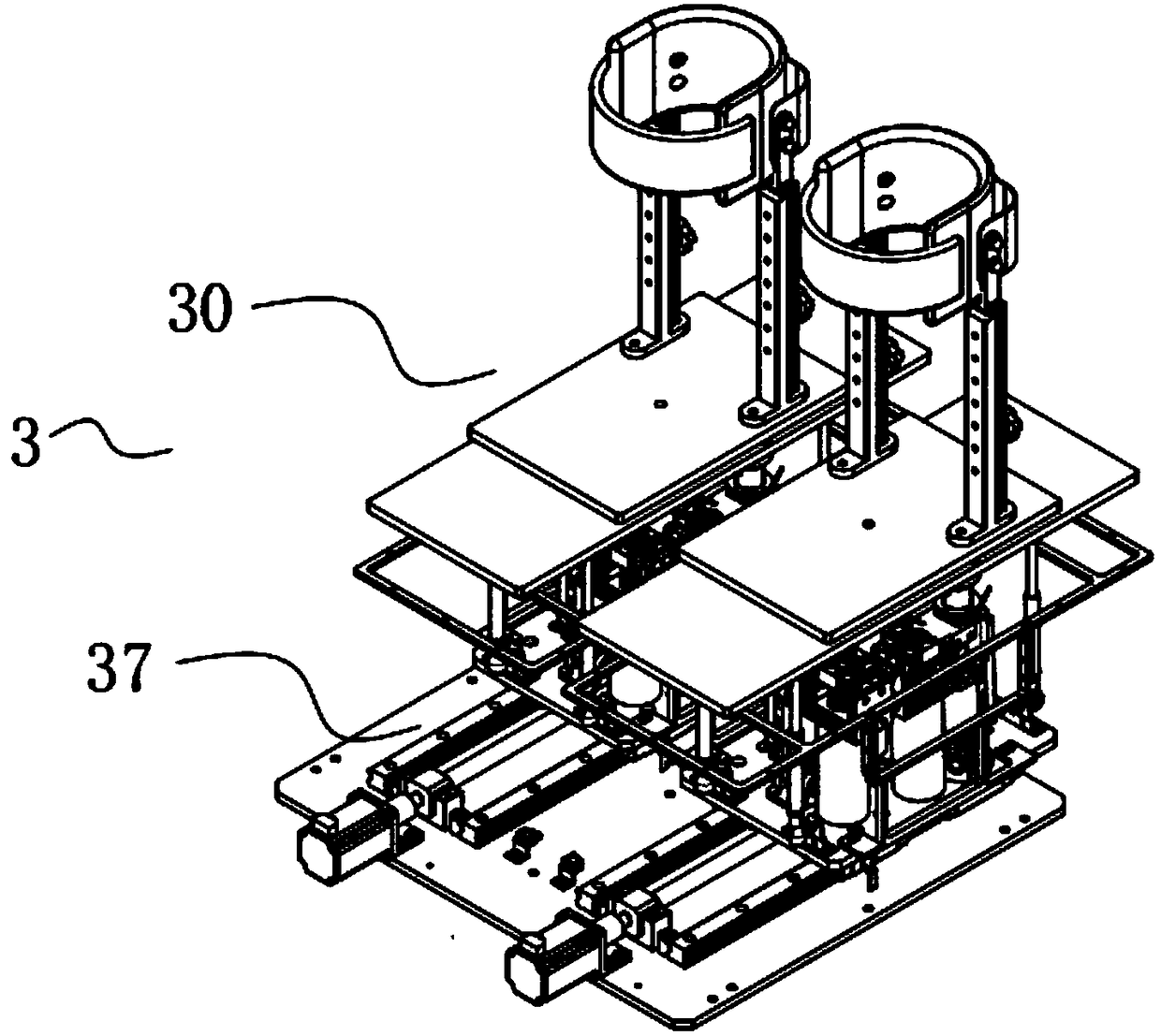
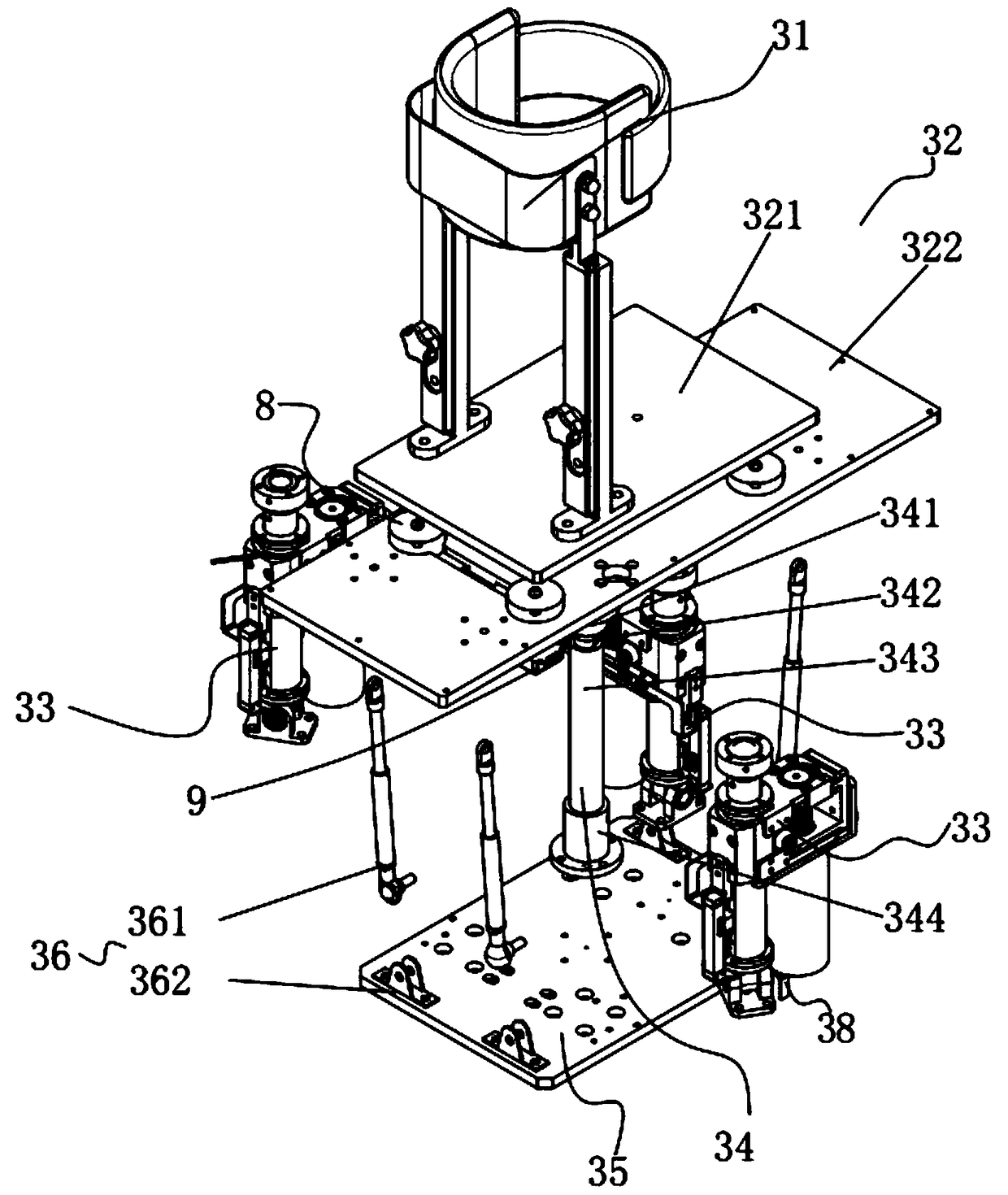
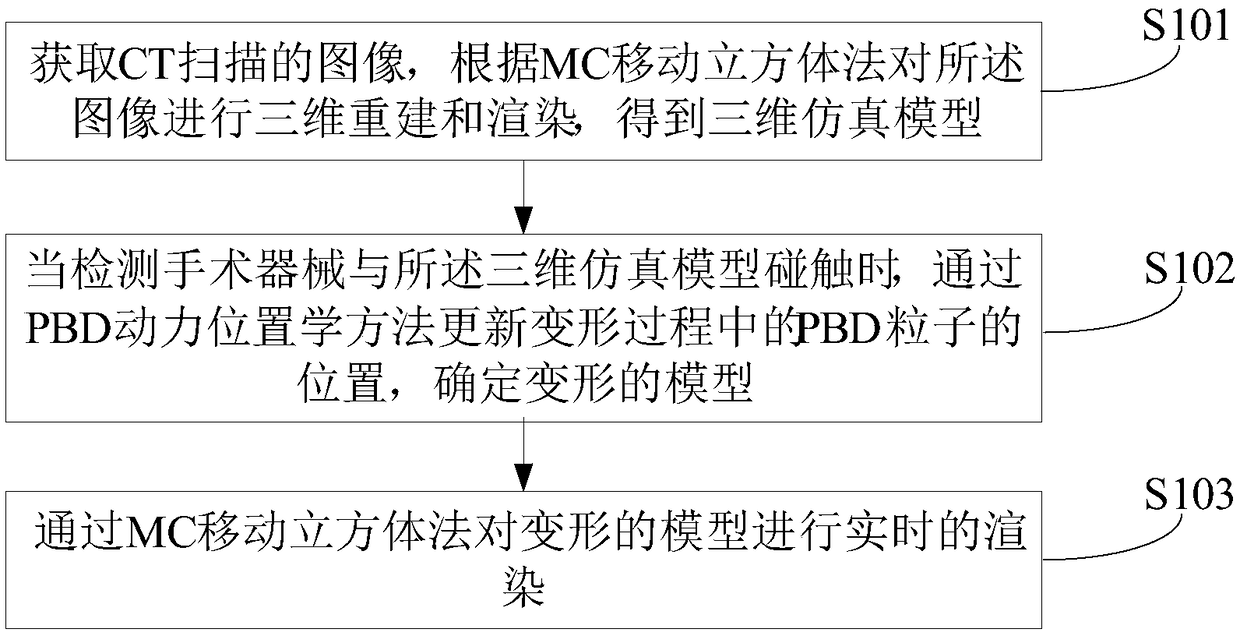
Surgical simulation method, device and system
ActiveCN109243614AMeet training requirementsMedical simulationMechanical/radiation/invasive therapiesThree dimensional modelDynamic method
The invention provides a surgical simulation method comprising the steps: acquiring a CT scanning image and performing three-dimensional reconstruction and rendering on the image according to an MC moving cube method so as to obtain a three-dimensional simulation model; when detecting the contact between the surgical instrument and the three-dimensional simulation model, the PBD particle positionin the contact process is continuously updated through the PBD position dynamics method to determine the deformed model; and real-time rendering is performed on the deformed model through the MC moving cube method. After the three-dimensional model corresponding to the scanning image is reconstructed by the MC moving cube method, the position of the PBD particles in the contact process is updatedby the PBD position dynamics method to determine the deformed model, and then real-time rendering is performed on the deformed model through the MC moving cube method to generate the touch deformationeffect of the three-dimensional simulation model in real time so as to effectively meet the training requirements of the surgery trainer for the complete video-assisted thoracoscopic lobectomy simulation scene.
Owner:SHENZHEN INST OF ADVANCED TECH
Stepless regulation magnetic control device for bicycle exercise equipment
ActiveCN104587661AMeet training requirementsMaximize the effect of magnetic resistanceMuscle exercising devicesRemote controlDrive shaft
The invention belongs to the field of exercise instruments, and relates to a stepless regulation magnetic control device for bicycle exercise equipment. The stepless regulation magnetic control device comprises a transmission device, a magnetic control group and a non-magnetic conductive metal rotary table, and is characterized in that the non-magnetic conductive metal rotary table is arranged on a transmission shaft of the transmission device and can synchronously rotate along with the transmission shaft; uniformly-distributed holes are formed in the ring surface of the non-magnetic conductive metal rotary table; the magnetic control group comprises a permanent magnet magnetic control group and an electromagnet magnetic control group; each magnetic control group comprises a plurality of identical permanent magnets or electromagnets with staggered magnetic poles; the magnets of the two magnetic control groups are annularly arranged and fixed on the two sides of the non-magnetic conductive metal rotary table respectively and are provided with electromagnet regulation and control devices. The stepless regulation magnetic control device can realize stepless regulation and also meet the training requirements of more body builders; the body builders can quantify all training indexes according to own demands to achieve the optimal fitness effect; the stepless regulation magnetic control device further has the advantages of light weight, small size, high convenience, simple and convenient operation, lower power consumption, and realizing of wireless remote control; the stepless regulation magnetic control device is an economical and practical updating substitute product with a higher performance-to-cost ratio.
Owner:JINHUA TIANYU TOOLS
Method for building psoriasis basic research models, and gas-liquid level transmembrane device
InactiveCN101570739APurification culture environmentGuaranteed observation cycle requirementsMicrobiological testing/measurementTissue cultureGas phaseBasic research
The invention provides a method for building psoriasis basic research models, and a gas-liquid level transmembrane device. The method utilizes a psoriasis streptococcal-induced mechanism and a T cell-mediated excessive proliferation mechanism, innovatively adopts a serum-free medium added with psoriasis peripheral blood T lymphocytes and hemolytic streptococcus antigens, and cultures psoriasis skin lesions in vitro. Characteristic tissue morphology of the psoriasis skin lesions can be maintained for as long as one week. The gas-liquid level transmembrane device used for culturing psoriasis in vitro skin lesions comprises the serum-free medium containing psoriasis peripheral blood T cells and the hemolytic streptococcus antigens in the method for building psoriasis basic research models, as well as sterilizing lens-wiping paper for manufacturing gas-liquid level, wherein tissue blocks and a gas phase are borne on the sterilizing lens-wiping paper; the gas phase is at 37 DEG C with 5 percent of CO2 saturation. The device not only can completely simulate gas-liquid junction environment for human skin growth, but also can reach the requirements of in vitro skin tissue culture. In addition, the device is easy to prepare, saves expenses, and has the advantages of repeatability, controllability and the like.
Owner:刘晓明 +1
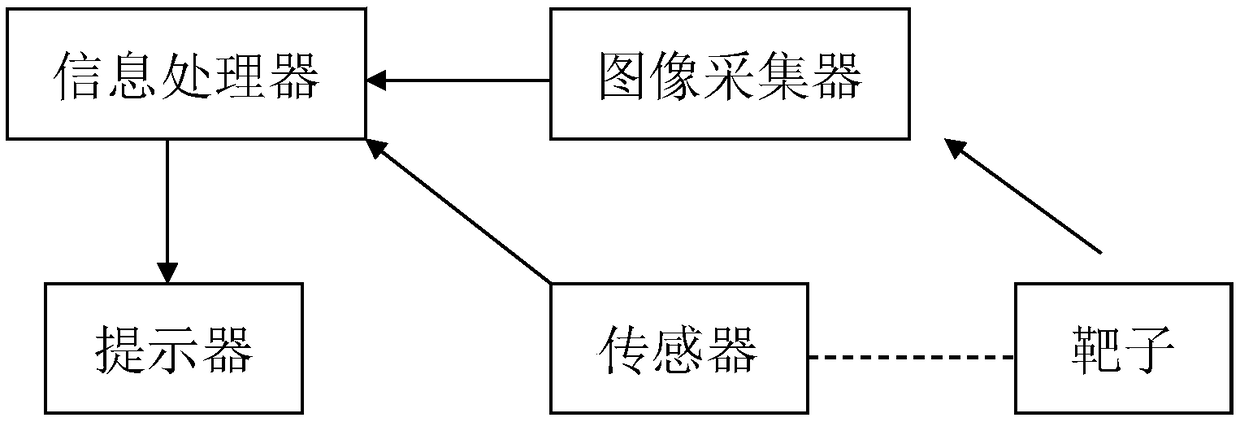
Self-adaptive archery auxiliary training system
The invention discloses a self-adaptive archery auxiliary training system which comprises an information processor, an image collector, a sensor and a prompter; the sensor comprises a gyroscope whichis arranged on the bow arm of the bow and is used for measuring the pose angle of the bow in real time, the image collector is used for acquiring the target surface information, the information processor analyzes, calculates and judges the information acquired by the gyroscope and the image collector, the prompter guides the shooting action of the archery personnel; The system guides the shootingaction of the archery personnel through the information processor, the image collector, the prompter and the sensor, and the pose angle of the bow is measured by arranging the gyroscope on the bow arm, the system has a simple structure, is not easy to loosen, is convenient to install and adjust, and reduces the cost.
Owner:HARBIN INST OF TECH