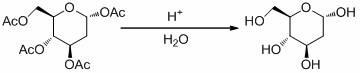
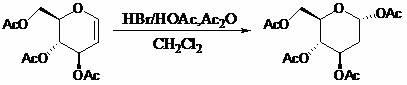Patents
Literature
125results about "Deoxy/unsaturated sugars" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
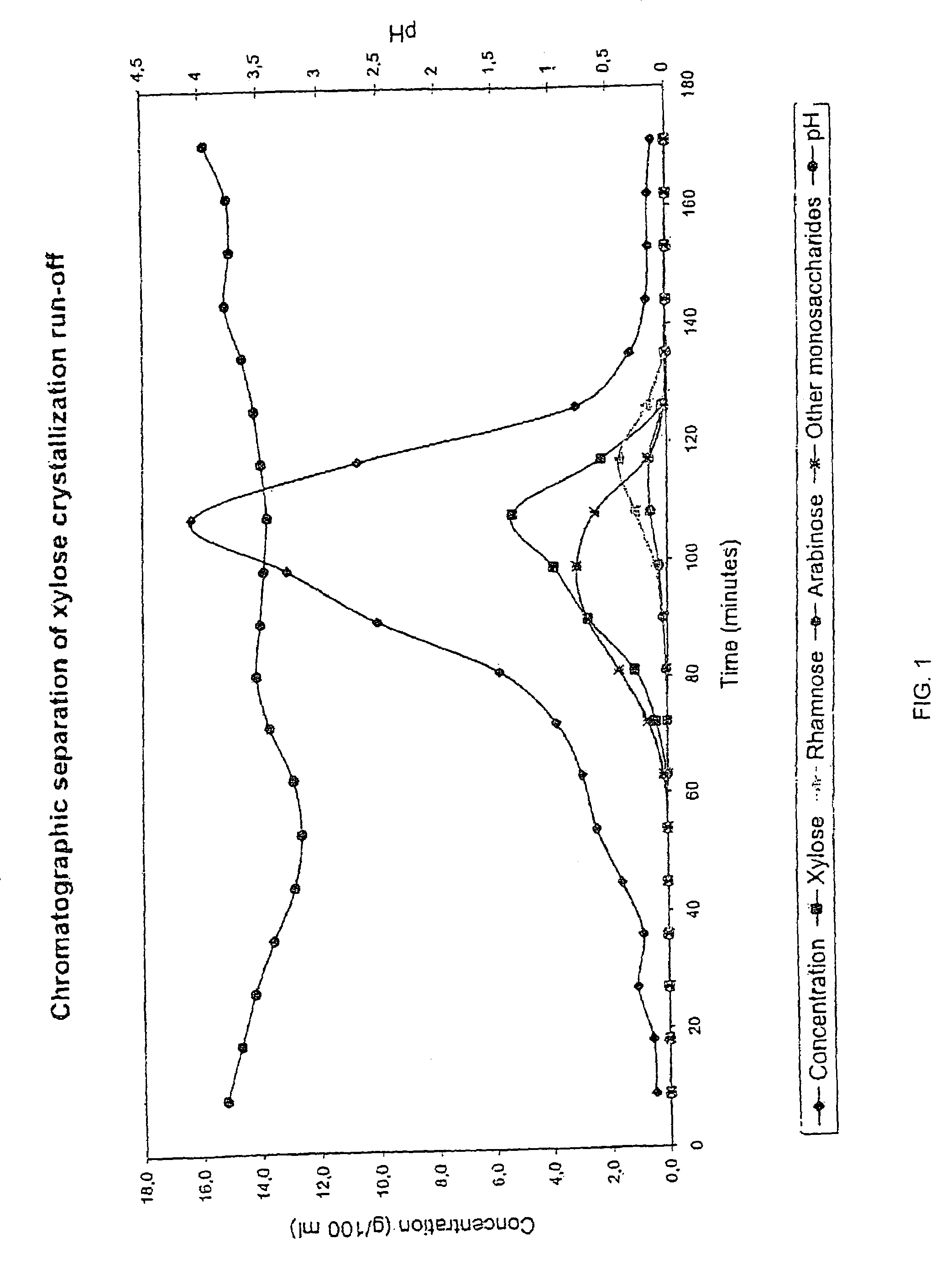
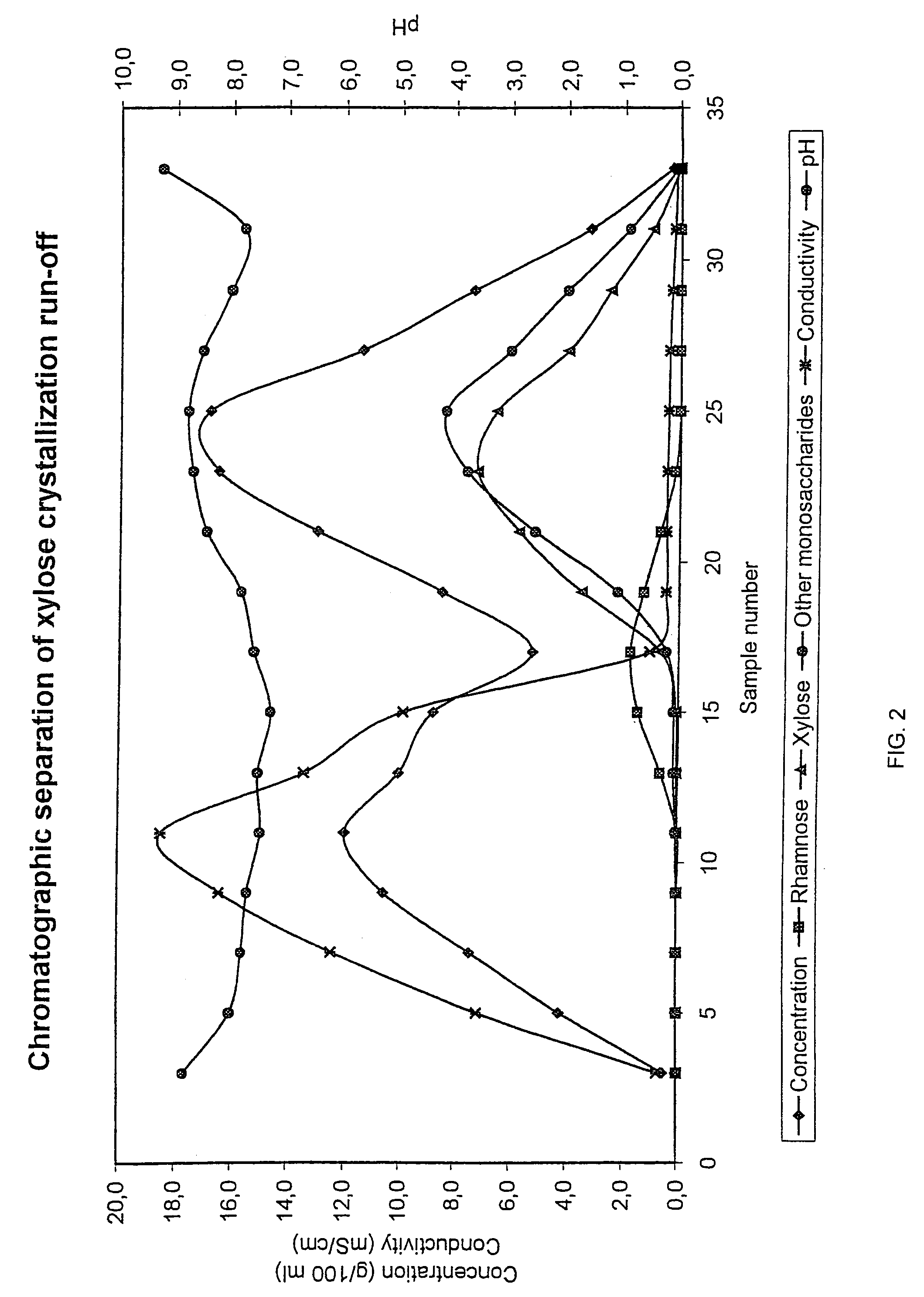
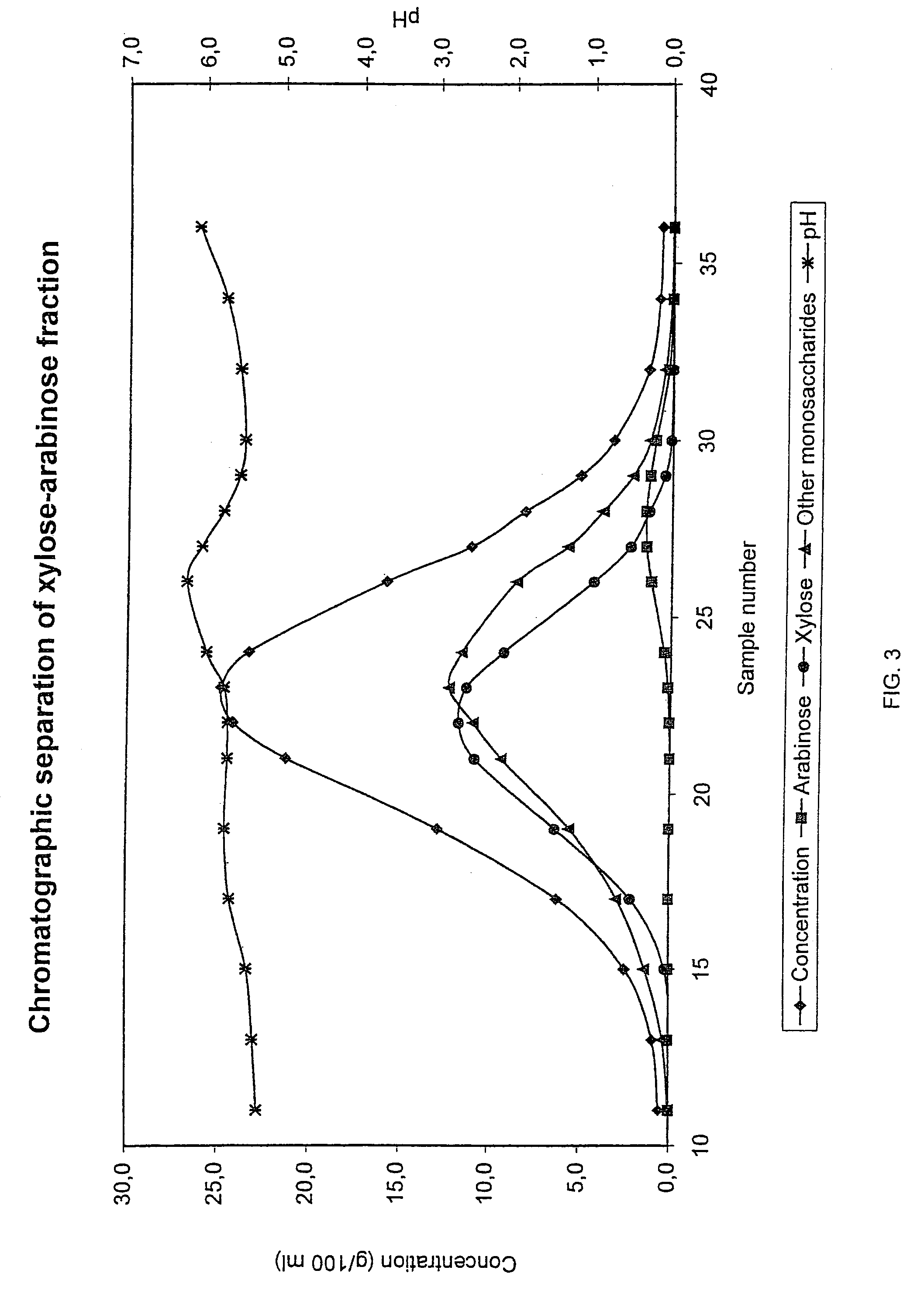
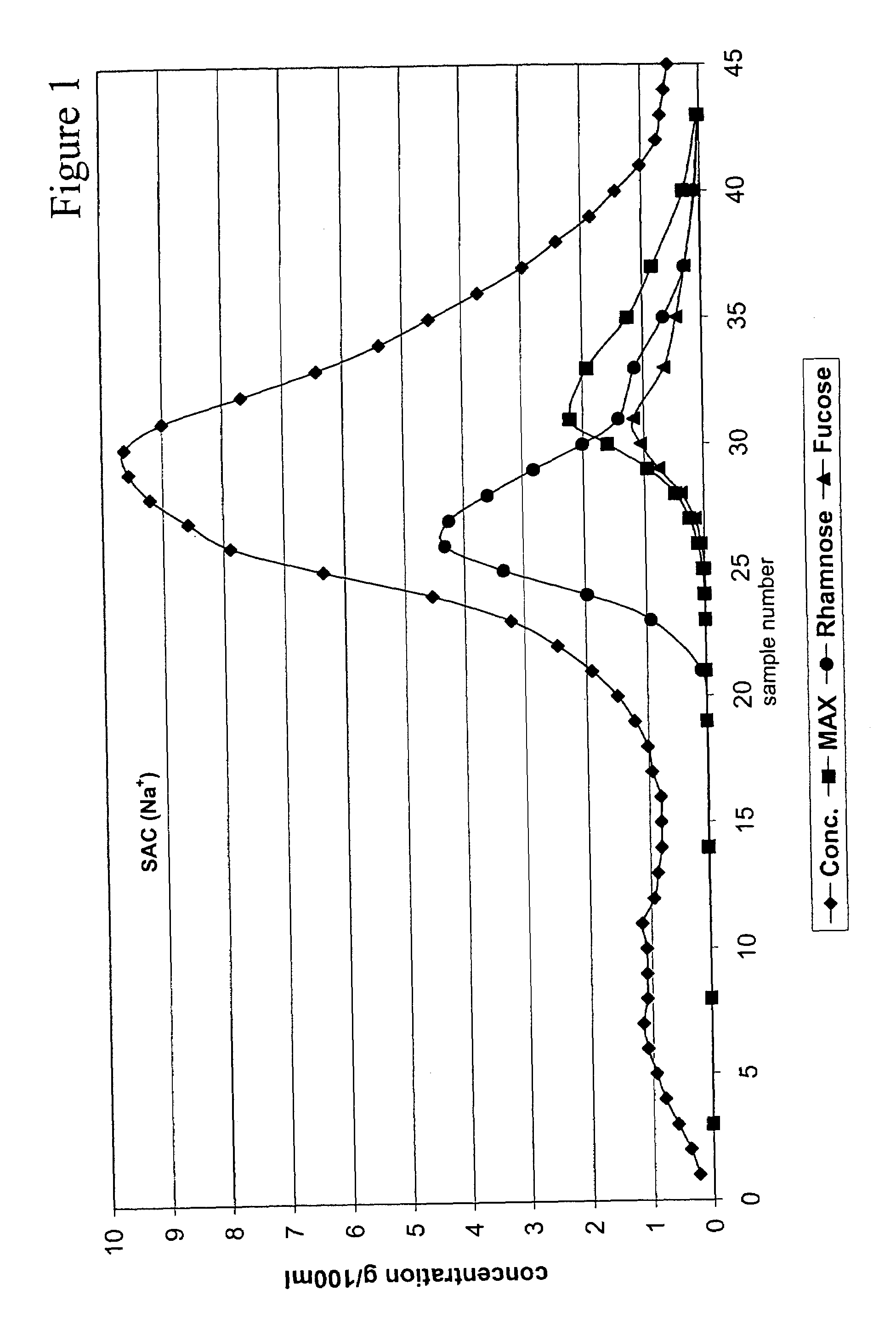
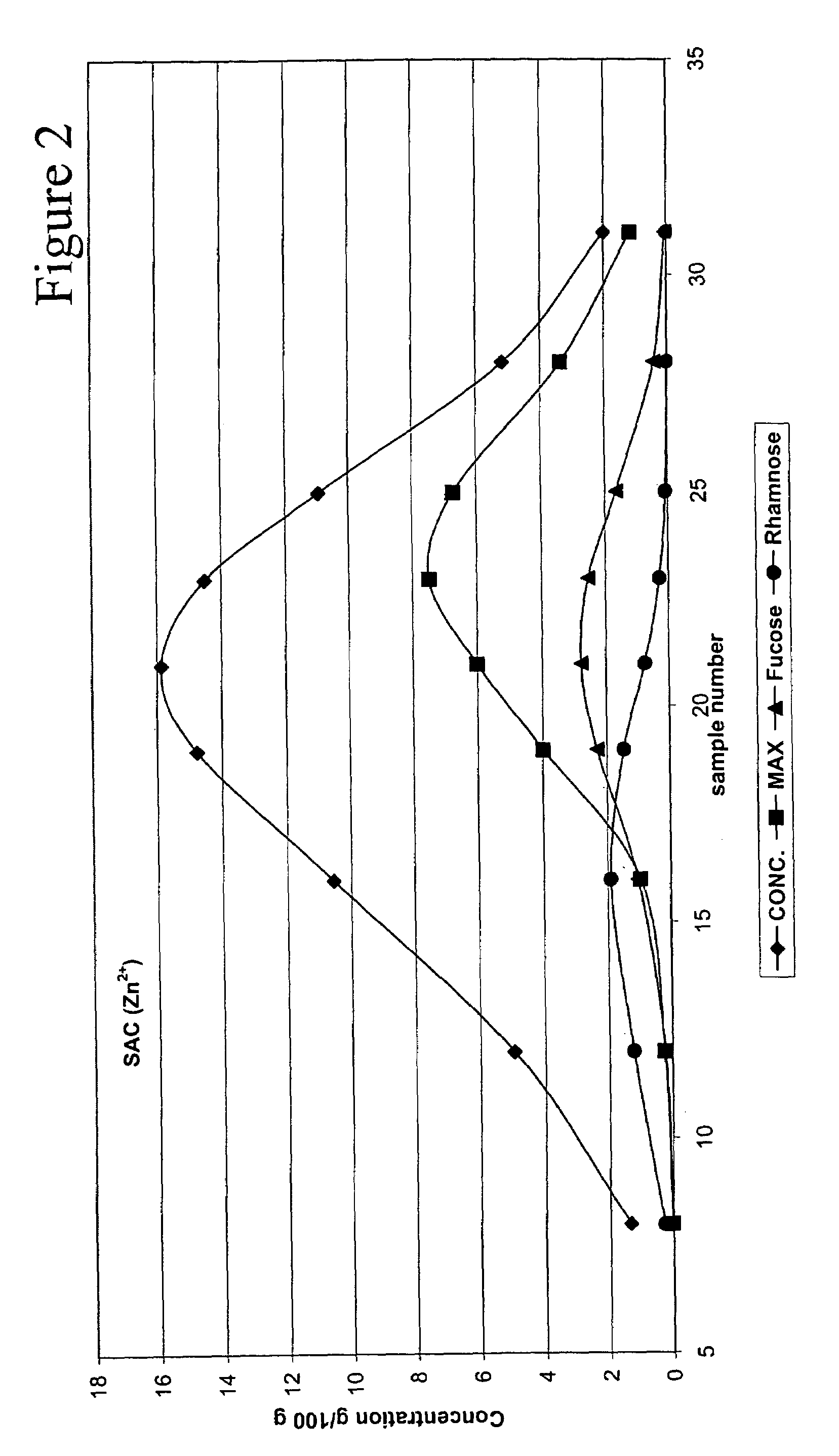
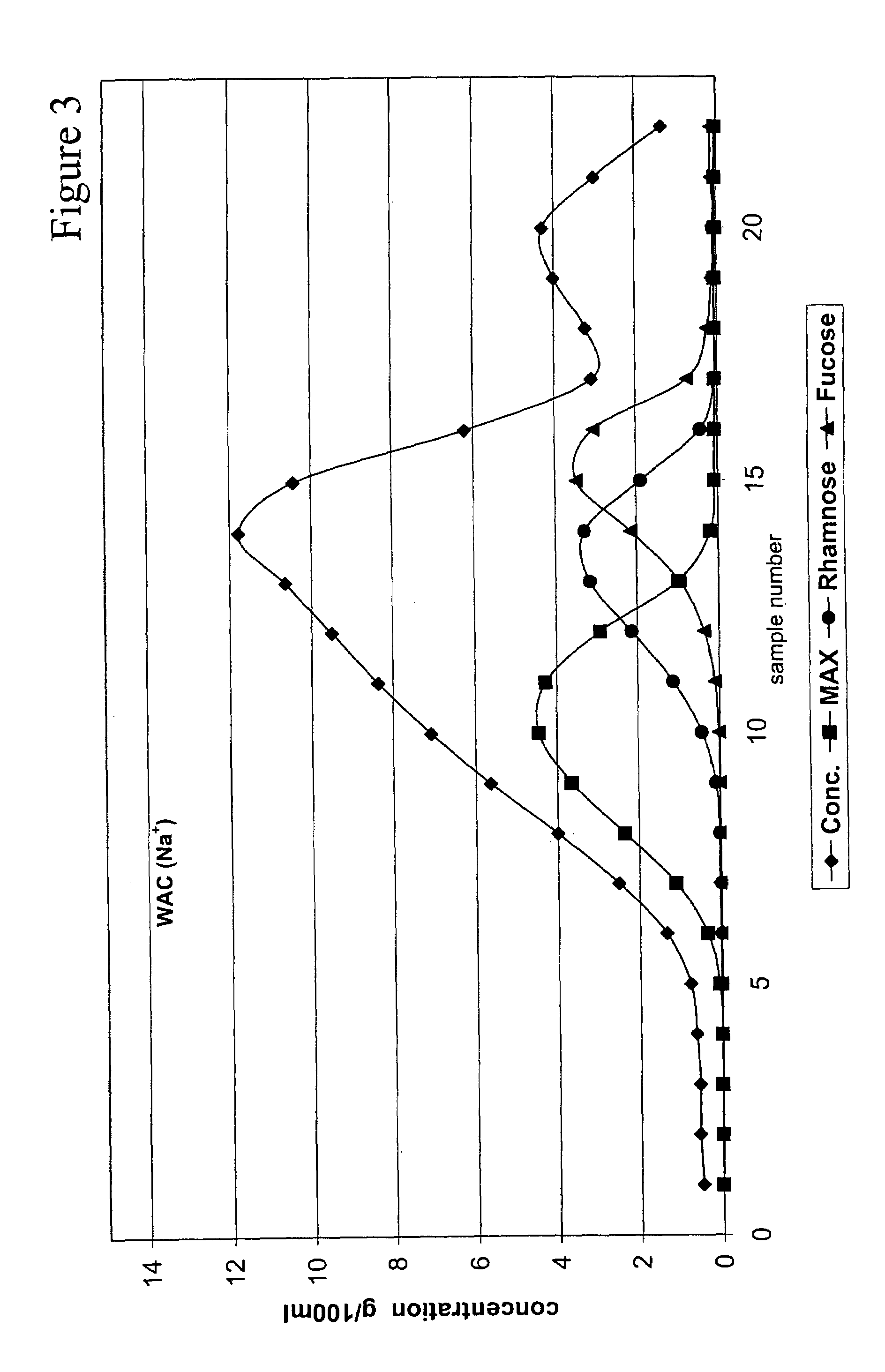
Method for recovering products
InactiveUS6987183B2Ion-exchanger regenerationSugar crystallisationChromatographic separationArabinose
The present invention is directed to a method comprising a multistep process for recovering one or more monosaccharides from a solution containing at least two monosaccharides selected from the group consisting of rhamnose, arabinose, xylose and mixtures thereof by using chromatographic separation comprising at least one step, where a weak acid cation exchange resin is used for the chromatographic separation.
Owner:DANISCO SWEETENERS
Separation of sugars
InactiveUS7037378B2Avoid insufficient puritySugar derivativesSugar productsChromatographic separationDeoxy sugar
Owner:DANISCO SWEETENERS
Method for recovering monosaccharide from solution using weakly acid cation exchange resin for chromatographic separation
InactiveCN1466632AChromatographic cation exchangersCation exchanger materialsChromatographic separationPhysical chemistry
The present invention relates to a method for recovering a monosaccharide selected from the group consisting of rhamnose, arabinose, xylose and mixtures thereof from a solution containing the same by a multistep process using chromatographic separation comprising at least one step, where a weakly acid cation exchange resin is used for the chromatographic separation.
Owner:DANISCO SWEETENERS
L-nucleic acid derivatives and process for the synthesis thereof
InactiveUS7125983B2Product can be usedOrganic active ingredientsSugar derivativesCombinatorial chemistryThymine
A novel method has been found to produce 2,2′-anhydro-1-(β-L-arabinofuranosyl)thymine as a novel useful intermediate compound. A novel method has been further found to produce thymidine from 2,2′-anhydro-1-(β-L-arabinofuranosyl)thymine. A novel method has been further found to L-2′-deoxyribose derivatives as a useful synthetic intermediate through L-2,2′-anhydro-5,6-dihydrocyclouridine derivative. According to these methods, synthesis of various L-nucleic acid derivatives, synthesis of which has been difficult till now.
Owner:MITSUI CHEM INC
Green preparation technology of dihydroquercetin
A green preparation technology of dihydroquercetin mainly comprises following steps: (1), preparing rough quercetin through hydrolysis of rutin NF11; (2), preparing fine quercetin by rough quercetin; (3), reacting fine quercetin under the catalysis of palladium-carbon catalyst by blowing hydrogen, or adding zinc powder or sodium borohydride to obtain dihydroquercetin reaction liquid; (4), preparing fine dihydroquercetin by dihydroquercetin reaction liquid; (5), extracting from acid liquor after rutin NF11 is hydrolyzed and filter pressed so as to obtain rhamnose. According to the invention, the rutin NF11 is taken as raw material to prepare dihydroquercetin, and meanwhile waste acid water is recycled, and the rhamnose is obtained through extraction, so that environmental pollution is avoided, the additional value of the technology is improved, and the whole process is green and environment-friendly.
Owner:李玉山
Method for preparing 2-deoxidation-L-ribose
ActiveCN102153600ALow toxicityWide variety of sourcesSugar derivativesSugar derivatives preparationBenzoic acidHigh pressure
The invention relates to a method for preparing 2-deoxidation-L-ribose based on L-arabinose as a raw material. The method comprises following seven steps: protection, group activation, transformation, deprotection and purification. Synthesis reaction conditions are common without high temperature and high pressure as well as supervacuum and are easy to control, and raw materials are available; in the process, acid catalysis, organic alkaline catalysis, strong acidic cation ion exchange resin, azodiisobutyronitrile catalysis, bezaldehyde and benzoic acid reduction, organic solvent extraction are carried out; the used medium is cheap and available and has no toxicity; and yield is high and cost is low, thus the preparation method is suitable for industrial production.
Owner:浙江一新制药股份有限公司
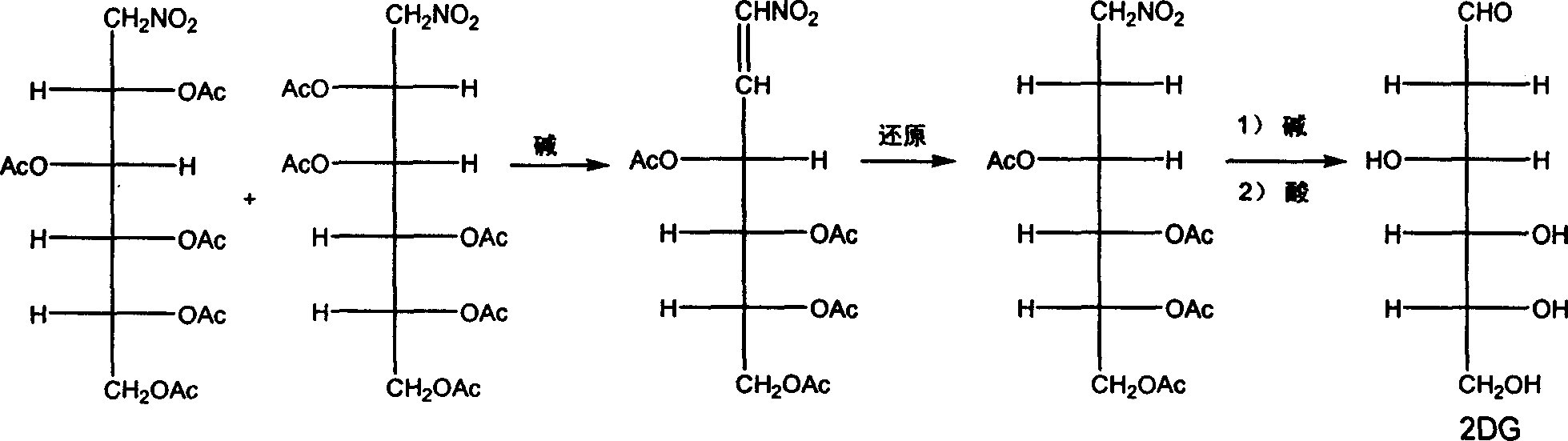
2-deoxidized glucose preparation method
InactiveCN1800193AOmit the process of isolating the intermediate D-glucal (III)Reduce production processSugar derivativesSugar derivatives preparationOxygenMethanol
The invention discloses a method for preparing for 2-deoxyglucose. The method uses D-acetylated dextroglucose and the absolute methanol to obtain the D-gluglucosan on the accelerating effect, and then it dose vacuum distillation to evaporate to dryness the methanol, it adds sulfuric acid water solution on the reacting machine to obtain the 2-deoxyglucose by hydrolyzing, it then uses normal method to do re-crystallization to the course product to obtain the 2-deoxyglucose product.
Owner:朱书汉
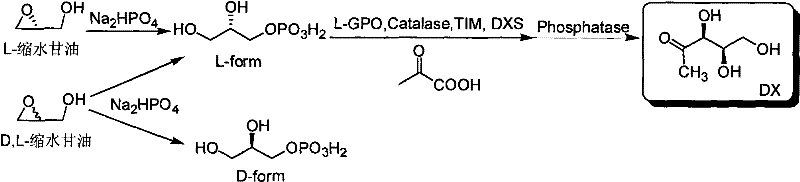
Method for preparing 1-deoxy-D-xylulose with chemical method-enzymatic method
InactiveCN101748170AHigh yieldImprove conversion rateSugar derivativesFermentationPhosphoric acidReaction step
The invention relates to a method for preparing 1-deoxy-D-xylulose with the combination of a chemical method and an enzymatic method, which comprises the steps as follows: in a water phase system under certain temperature, the glycidol can form glycerol 3-phosphate by inorganic phosphate through ring opening, then the generated L-glycerol 3-phosphate is oxidized by L-GPO to form DHAP under the existence of catalase, the DHAP is converted into DXP under the existence of pyruvic acid, TIM enzyme and DXS, and target molecule DX can be obtained by hydrolyzing the compound through phosphatase. The method organically couples the simple method for preparing the DHAP by taking the stable, cheap and easily available compound as raw material with the method for preparing the DX by using the enzymatic method, the target molecule DX can be obtained through one-step reaction, the yield is high, the purification step is simple and feasible, the DX with high purity (more than 95%) can be obtained, the price of the raw material is low, the nature is stable, and the technical problems of expensive and unstable reaction material, complicated reaction steps, longer time consuming and lower overall yield of the method for preparing DX in the background technology are solved.
Owner:NORTHWEST UNIV(CN)
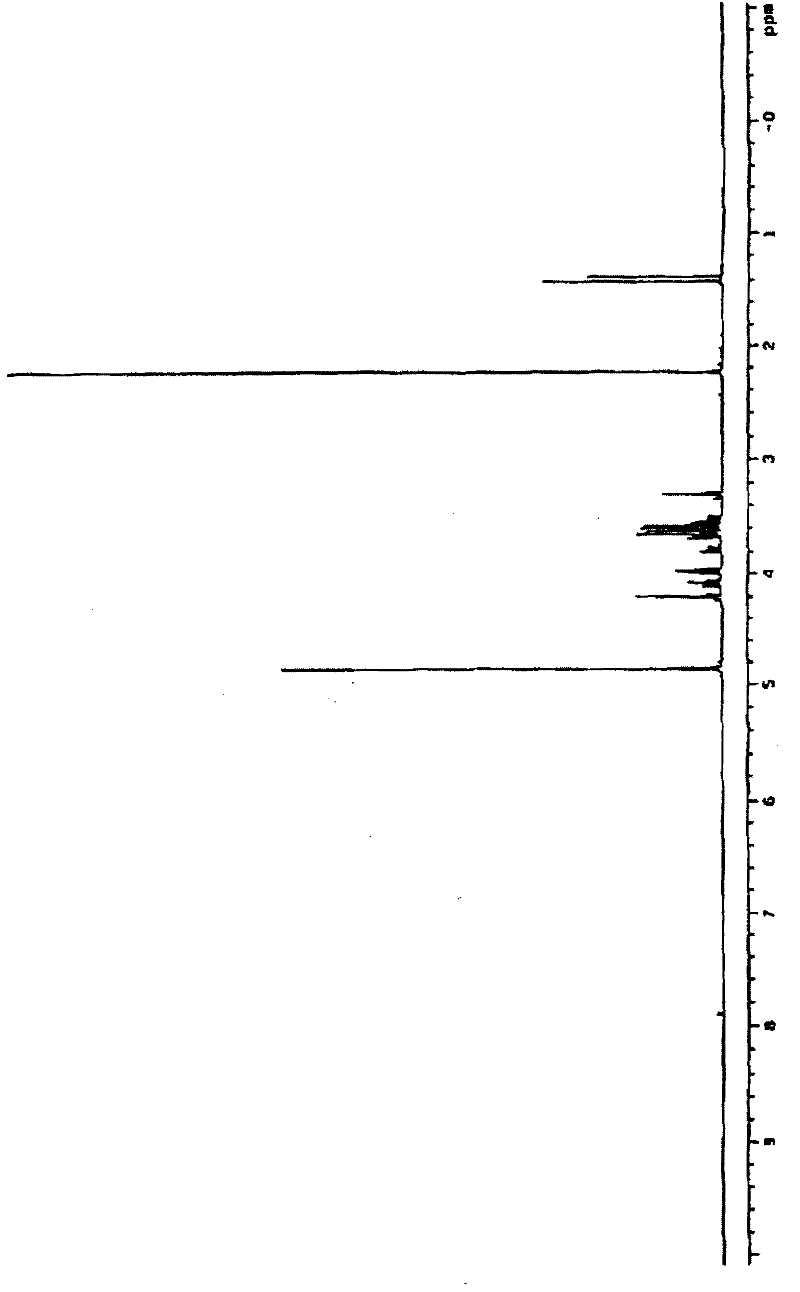
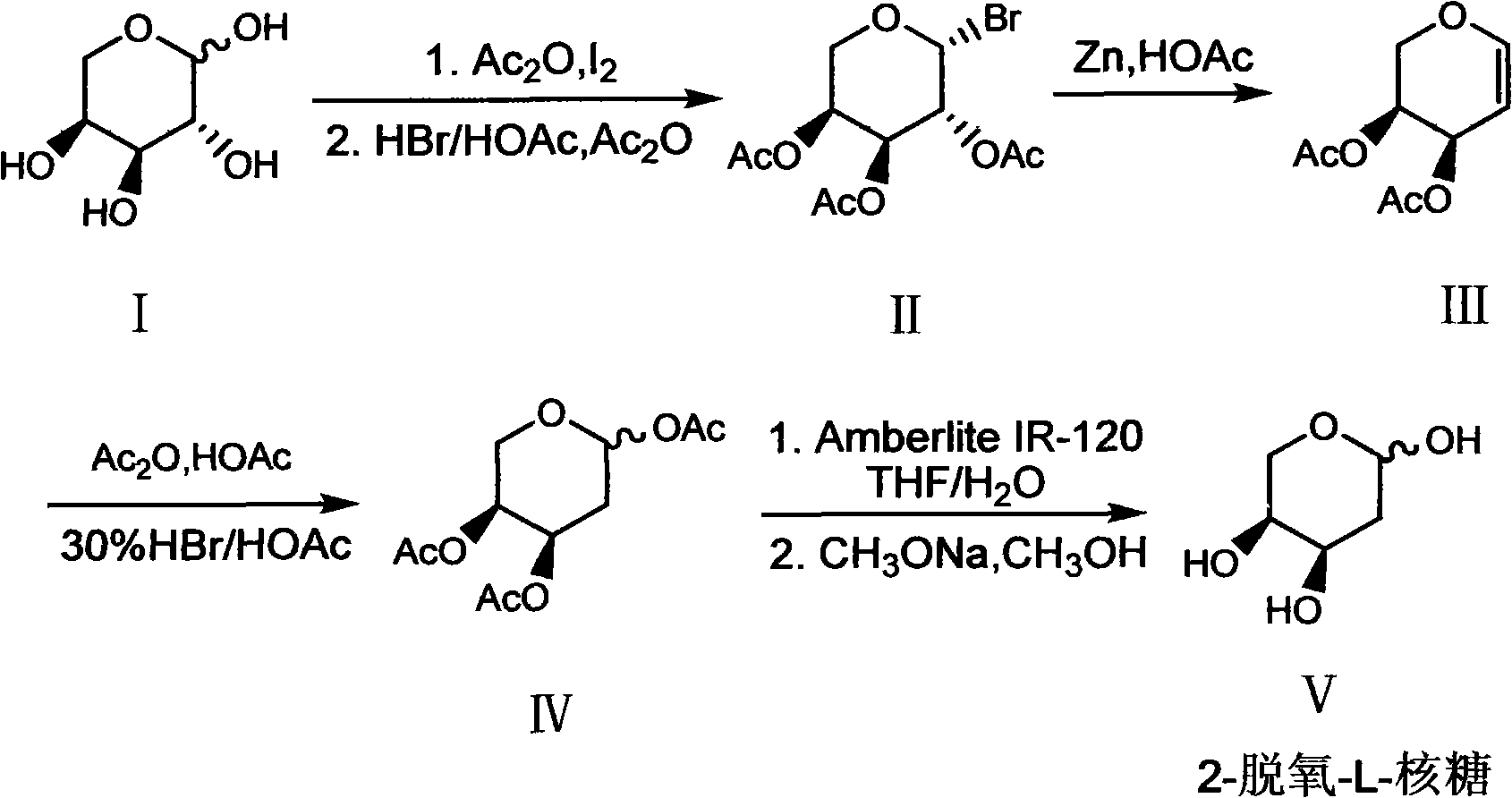
Preparation method of 2-deoxy-L-ribose
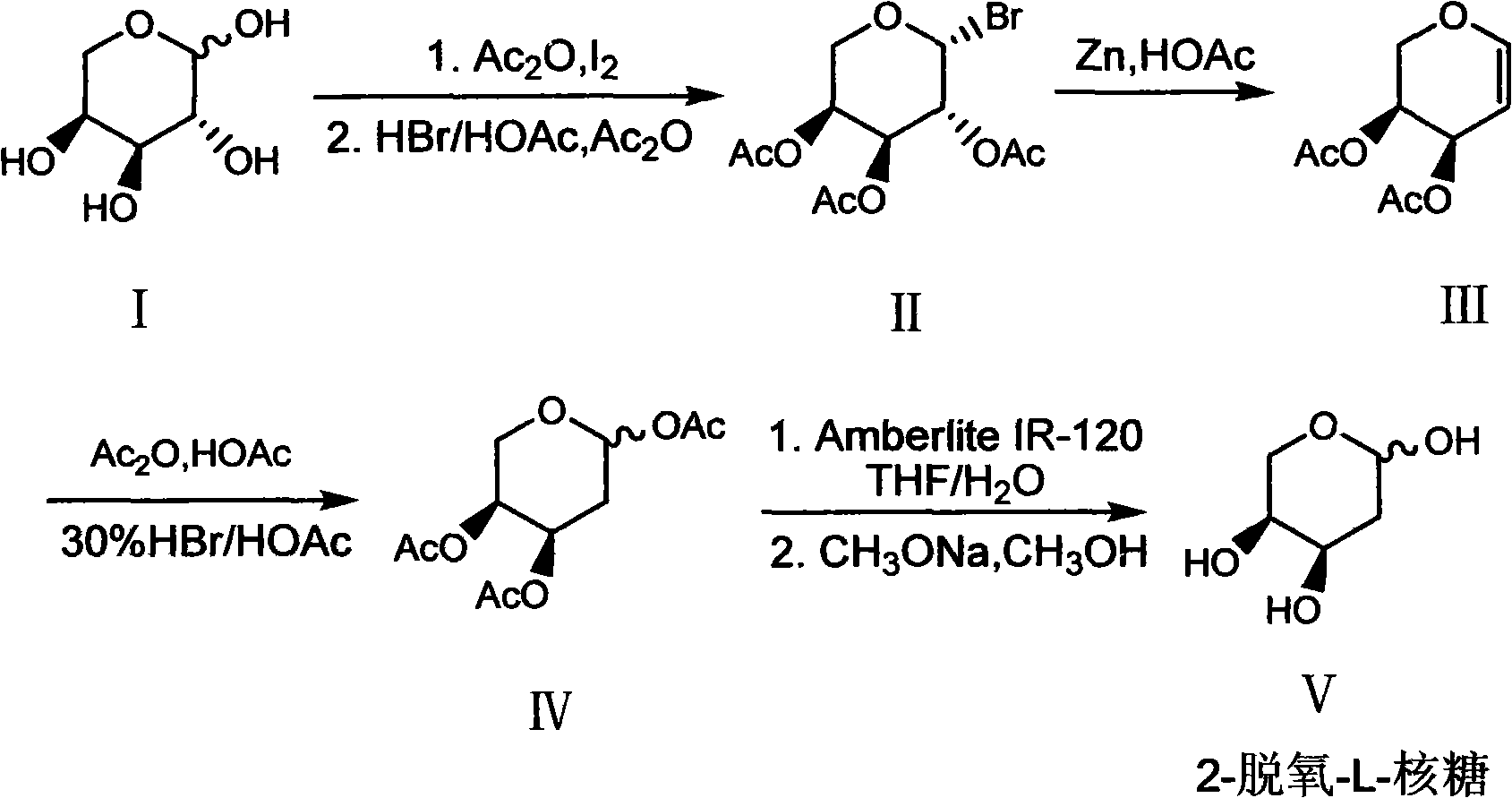
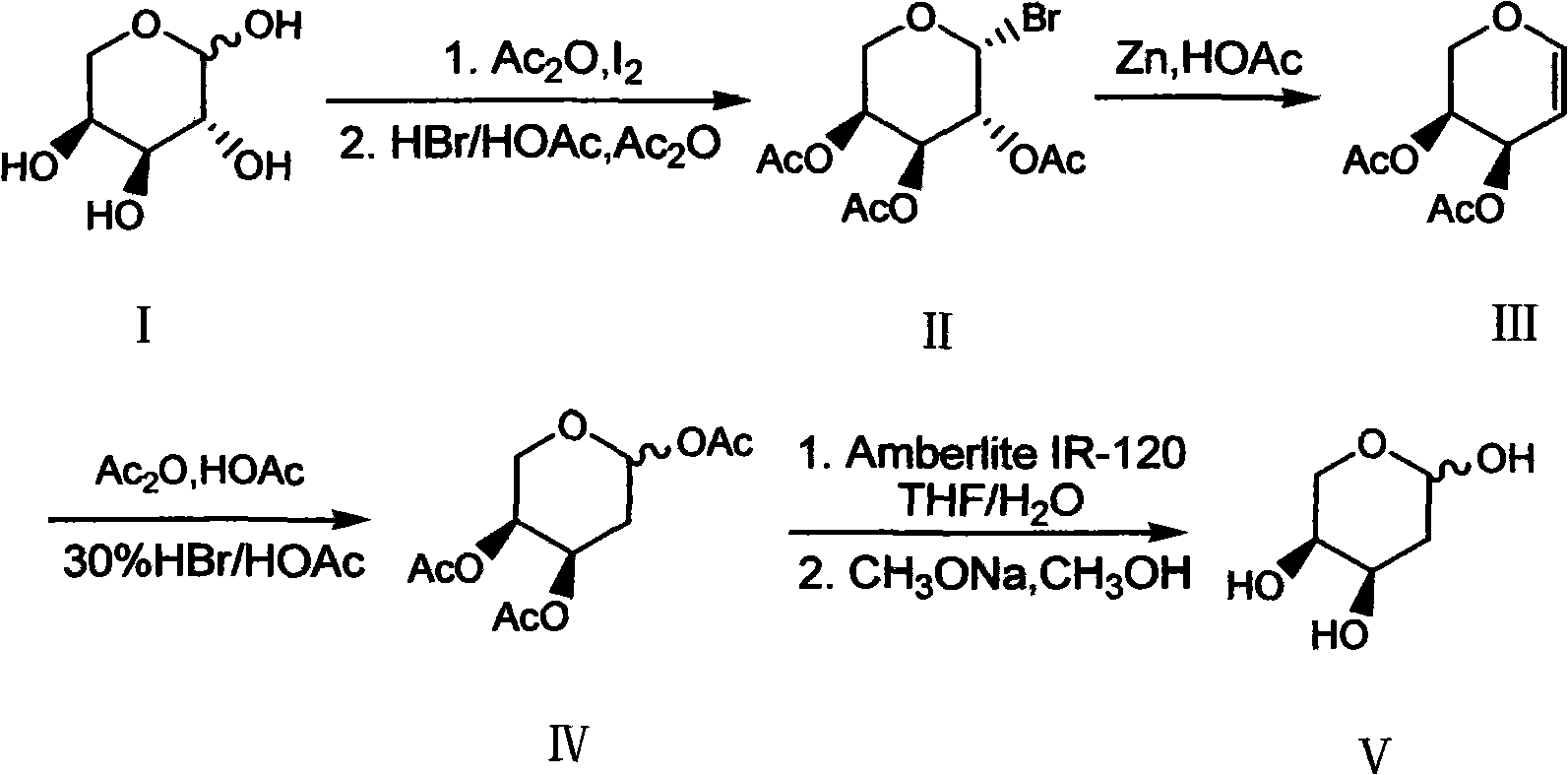
The invention discloses a synthesis method of 2-deoxy-L-ribose. The synthesis method comprises the following steps: in the presence of acetyl-protected hydroxyl, taking cheap L-arabinose as a raw material to generate beta-bromo-L-triacetyl arabopyranose through bromination under catalysis of iodine; performing elimination reaction in an acetic acid solution which takes zinc powder as a reducing agent to generate 3,4-diacetyl-L-arabinal; performing addition reaction in an acetic acid / acetic anhydride solution of hydrogen bromide to generate an intermediate 2-deoxy-1,3,4-triacetyl-L-ribose; andremoving acetyl stepwise by utilizing Amberlite IR-120 acid resin and a methanol solution containing sodium methylate to finally obtain the target product of the 2-deoxy-L-ribose. The synthesis method has the advantages that the production cost is greatly lowered by utilizing cheap and available raw materials and reagents, reaction conditions are more moderate, the operation process is simple andsafe, the yield of the obtained 2-deoxy-L-ribose is more ideal, and large-scale industrial production can be realized, thus solving the problem of demand quantity of the intermediate-2-deoxy-L-ribosein the field of pharmaceutical and chemical industry.
Owner:唐传生物科技(厦门)有限公司
Prepn apparatus and process for 2-fluoro-18 substituent-2 deoxy-beta-D-glucose
The present invention relates to the field of preparing positive electronic radioactive medicine, especially the preparation apparatus and process of 2-fluoro-8 substituent-2-deoxy-beta-D-glucose. The apparatus includes wind bath for heating and cooling the reaction bottle, and negative perssure transmission unit for ultimate product with reduced radioactivity loss, shortened time and raised synthesis efficiency. The technological process combined acid-alkali hydrolysis without possibility of elimination reaction in the 2-position, and it has short hydrolysis period and high radioactive purity and chemical purity of the prepared medicine.
Owner:派特(北京)科技有限公司
Method of Controlling the Proliferation of Vascular Endothelial Cells and Inhibiting Lumen Formation
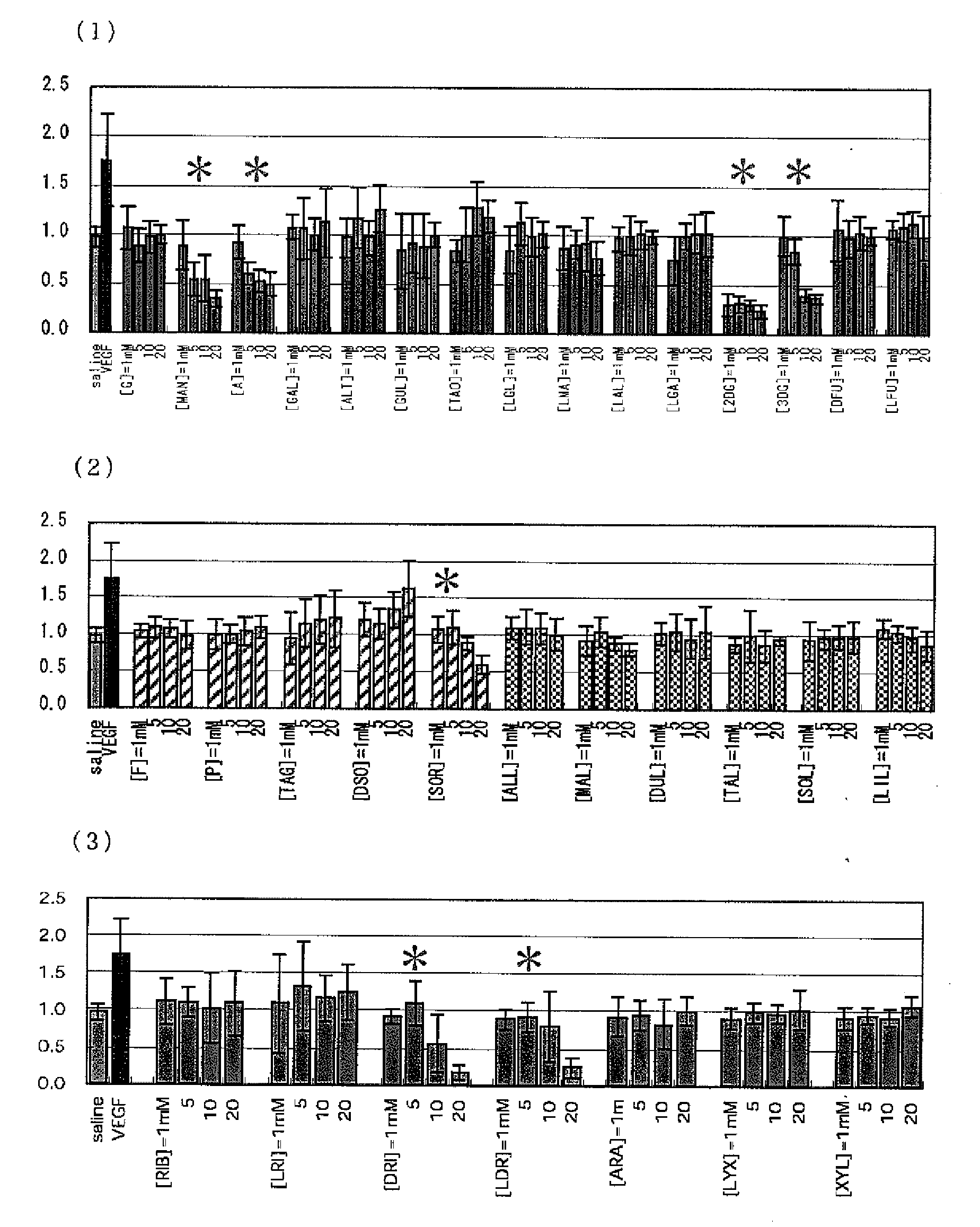
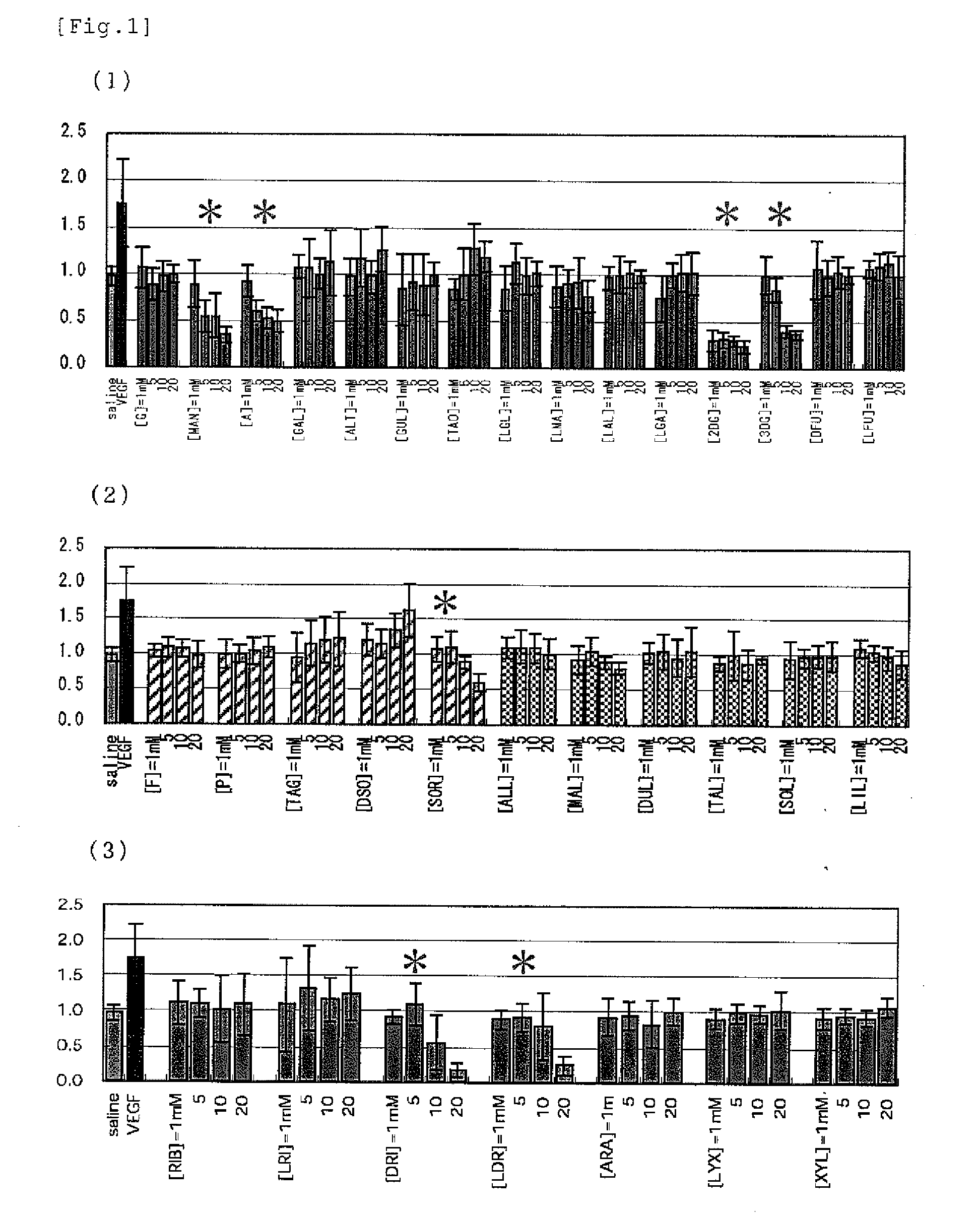
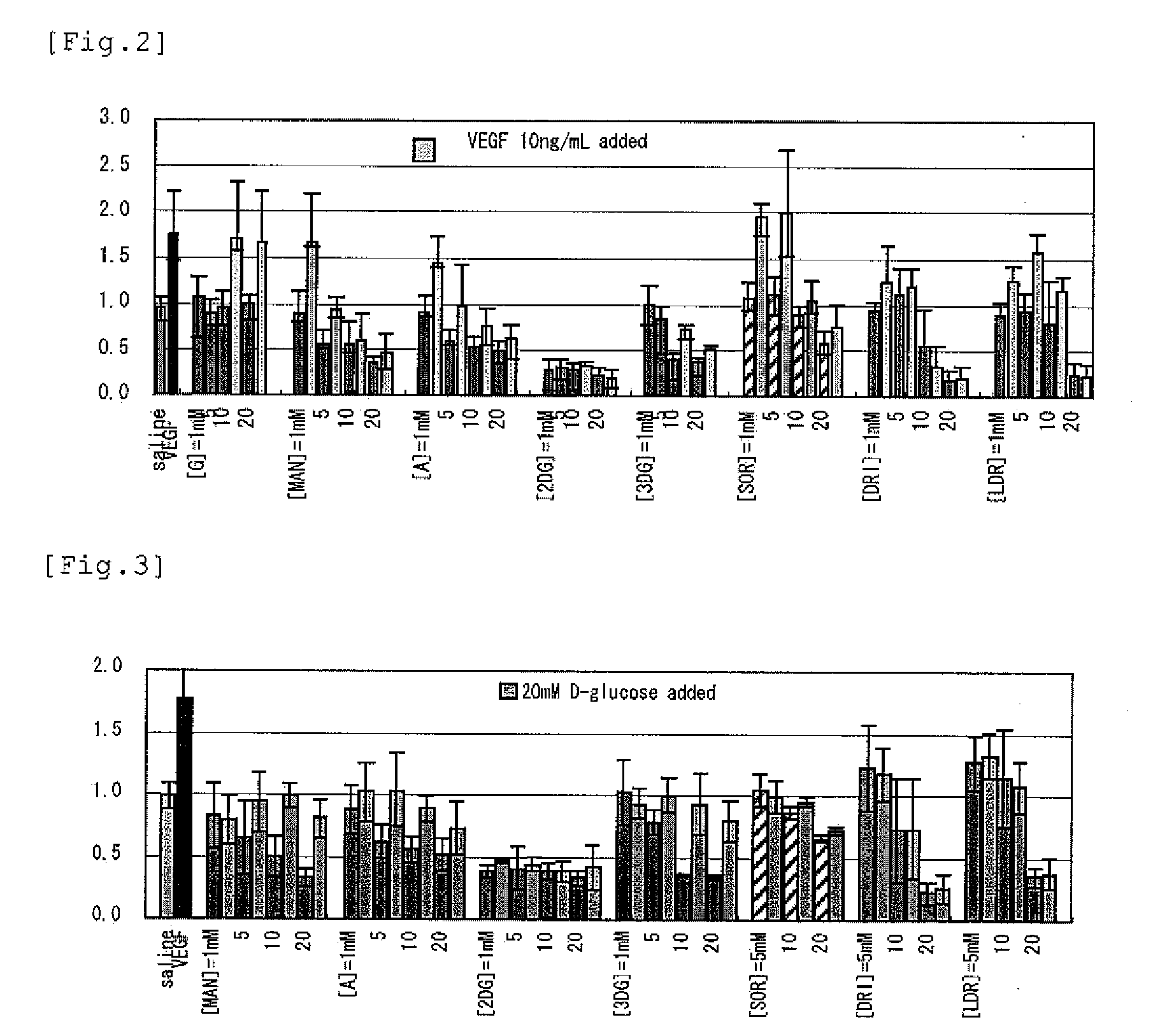
[PROBLEMS] To find out a specific rare sugar having effects of inhibiting the proliferation of vascular endothelial cells and lumen formation and utilize these effects. To provide this rare sugar as a preventive / remedy for diseases with angiogenesis, a cosmetic or a functional food.[MEANS FOR SOLVING PROBLEMS] A method of controlling the proliferation of vascular endothelial cells characterized by utilizing the vascular endothelial cell proliferation-controlling effect of D-mannose, D-allose, 2-deoxy-D-glucose, 3-deoxy-D-glucose, L-sorbose, 2-deoxy-D-ribose and / or 2-deoxy-L-ribose. A method of inhibiting lumen formation of vascular endothelial cells characterized by utilizing the vascular endothelial cell lumen formation-inhibiting effect of D-allose, D-altrose, D-gulose, D-talose, L-allose, 2-deoxy-D-glucose, 3-deoxy-D-glucose, D-ribose, L-ribose, 2-deoxy-D-ribose and / or 2-deoxy-L-ribose.
Owner:RARE SUGAR PRODN TECHN RES LAB
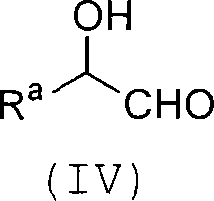
Process for producing carbon-diminished aldose compound
InactiveCN101090907AHigh industrial applicabilityImprove operational efficiencyOrganic compound preparationMonosaccharidesAtypical hyperphenylalaninemiaCompound a
A process by which 5-deoxy-L-arabinose, which is important as a material for the production of saproterin, a useful therapeutic agent for atypical hyperphenylalaninemia, can be industrially and efficiently produced even with a simple production apparatus. The process for 5-deoxy-L-arabinose production is characterized by reacting L-rhamnose with a C11-16 linear alkyl mercaptan compound in the presence of an acid catalyst to yield an L-rhamnose di(alkyl mercaptal), subsequently subjecting the resultant compound to an oxidation reaction to obtain a sulfonyl derivative, and then subjecting it to a carbon diminution reaction. This production process is applicable to a process for the production of a compound formed by removing one carbon atom from another aldol compound. Thus, the process is one for producing carbon-diminished compounds of general aldoses.
Owner:ASUBIO PHARMA
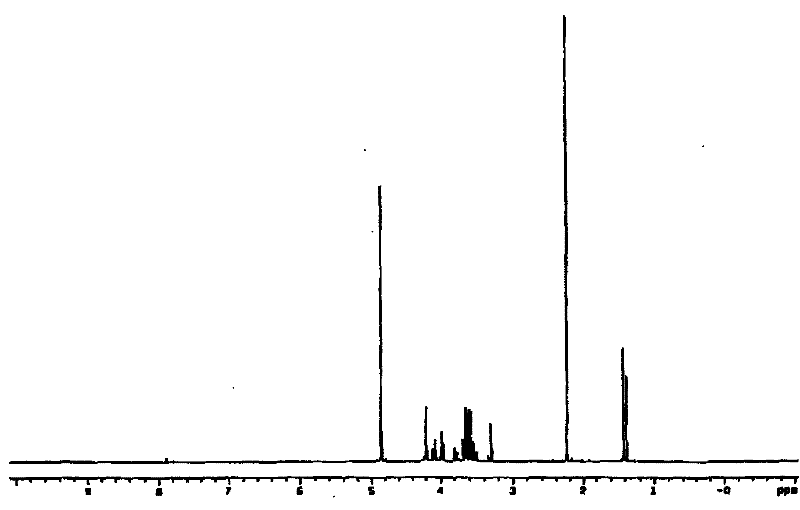
Method of preparing 2-deoxy-L-ribose
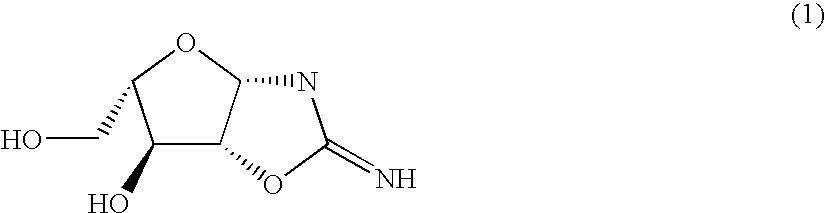
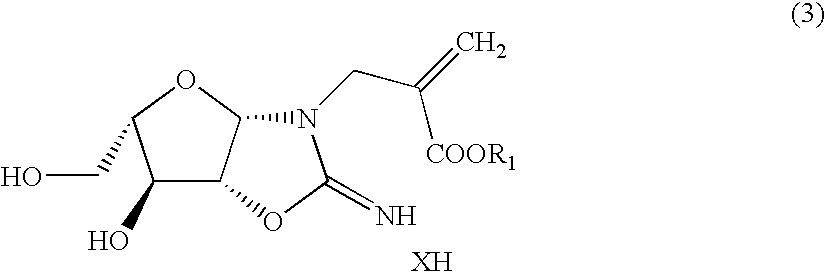
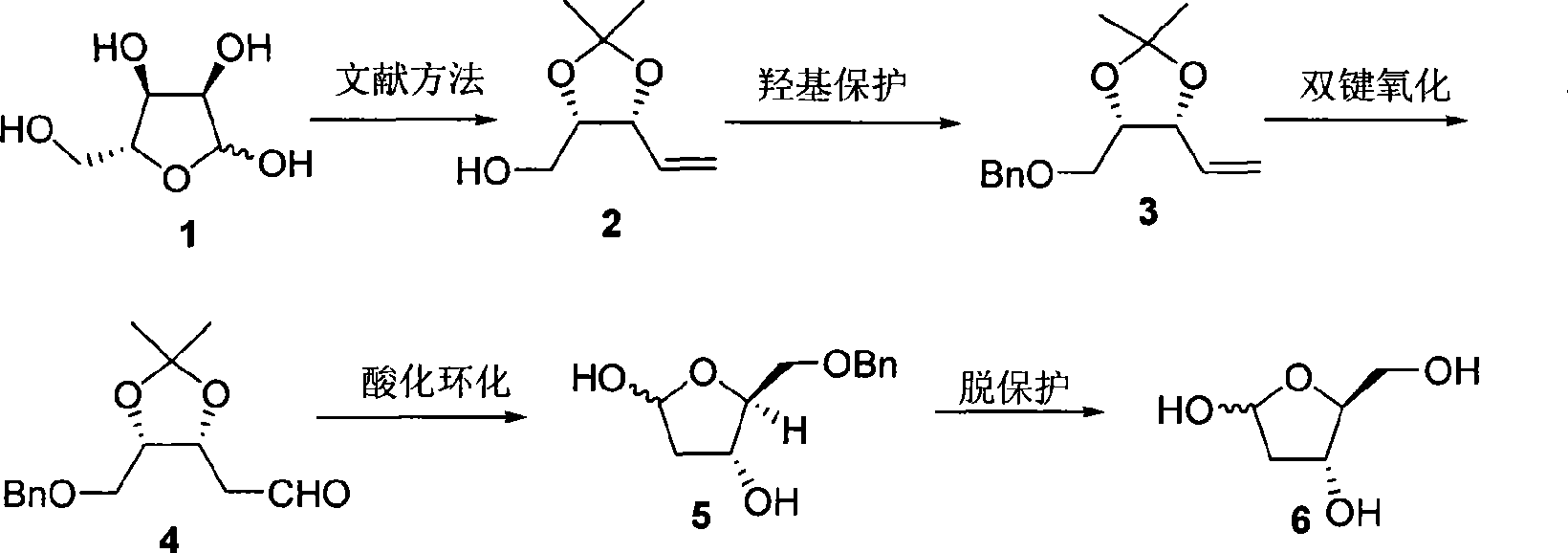
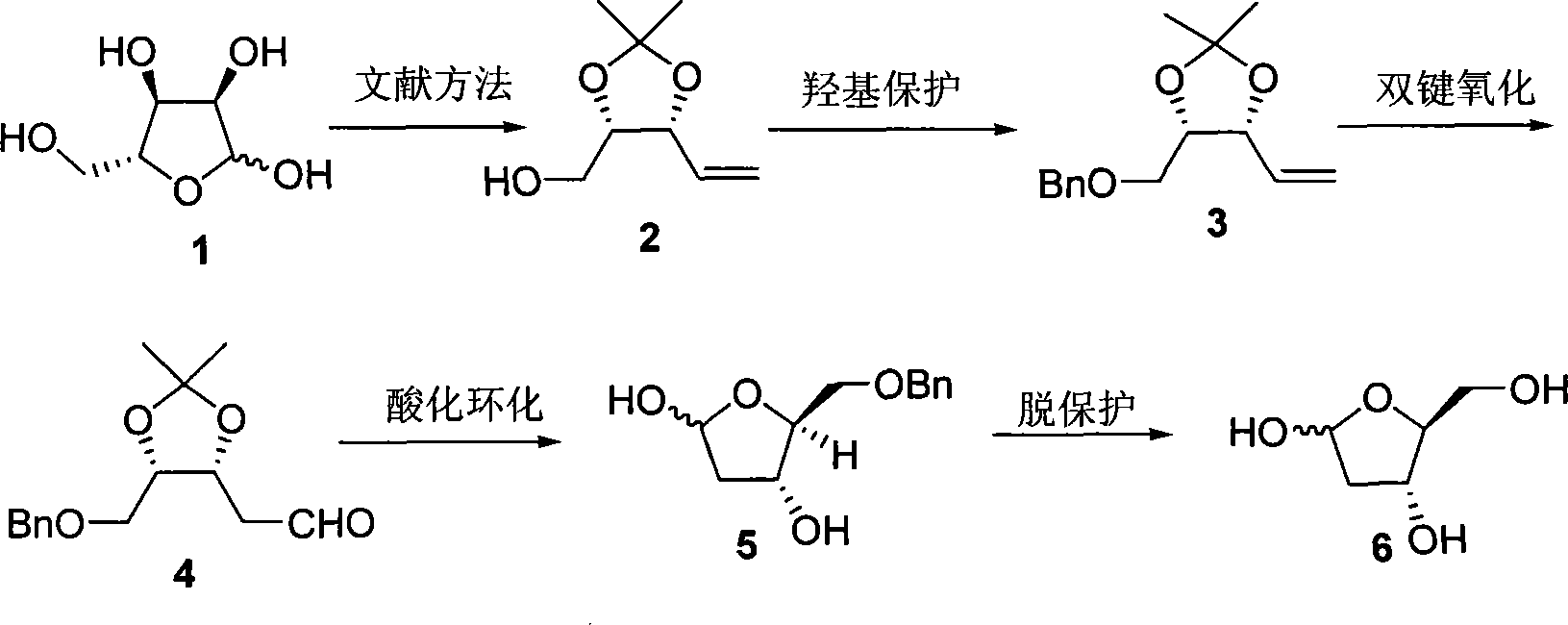
InactiveCN101125868AEasy to synthesizeHigh optical purityBulk chemical productionDeoxy/unsaturated sugarsDouble bondRibose
A preparation method of a 2-dioxidation-L-ribose relates to a non-natural deoxidation pentose. The invention provides the preparation method that takes D-(-)-ribose (1) as raw material and has simple technology, high optical activity and yield of the 2-dioxidation-L-ribose. (2S, 3R)-2, 3-O- isopropylidene-4-pentenol (2) is prepared through the raw material (1) by a literature method, hydroxide radical of the first carbon in (2) is protected to get (3R, 4S)-3, 4-O-isopropylidene-5- benzyloxy-4-pentene(3), then (3R,4S)-3, 4-O- isopropylidene-5- benzyloxy pentanal (4) is prepared through preferred oxidation at the end of the double linkage (3) in the existence of palladium chloride and cuprous chloride, acetonide protecting group in (4) is removed through catalysis of acid and phenmethyl 2-dioxidation-L-ribose (5) is obtained through cyclization, finally the 2-dioxidation-L-ribose (6) is prepared after the phenmethyl is removed through catalysis.
Owner:XIAMEN UNIV
Chemoenzymatic synthesis of heparin and heparan sulfate analogs
The present invention provides a one-pot multi-enzyme method for preparing UDP-sugars from simple sugar starting materials. The invention also provides a one-pot multi-enzyme method for preparing oligosaccharides from simple sugar starting materials.
Owner:RGT UNIV OF CALIFORNIA
Preparation method of 2-deoxidizing-D-glucose
InactiveCN102180914ASimple processGood choiceSugar derivativesSugar derivatives preparationState of artSynthesis methods
The invention discloses a preparation method of 2-deoxidizing-D-glucose, which is characterized in that in the method, an intermediate 2-deoxidizing-1,3,4,6-tetra-O-acetyl group-D-glucose is prepared by the selective addition reaction of acetyl glucose alkene, and acetyl groups are removed by the acid catalytic hydrolysis reaction of an intermediate 2-deoxidizing-acetyl groups-D-glucose, thus obtaining the 2-deoxidizing-D-glucose after purification. Compared with the prior art, the preparation method has the advantages of simple process, good selectivity, high yield and low cost, thus being a relatively green, environment-protective, economical and efficient synthesis method with broad application prospect.
Owner:EAST CHINA NORMAL UNIVERSITY
Preparation method of 2-deoxy-D-glucose
ActiveCN102924540AEasy to operateHigh yieldSugar derivativesSugar derivatives preparationHydrogenD-Glucose
The invention discloses a preparation method of 2-deoxy-D-glucose, relating to the field of production of active pharmaceutical ingredients. The preparation method comprises the following steps of: a, taking 3, 4, 6-triacetyl glucose alkenes as raw materials, removing acetyl protection groups in the presence of alkalis and then performing benzyl protection to obtain an intermediate I shown as a formula II; b, acid-hydrolyzing the intermediate I obtained in the step a to obtain an intermediate II shown as a formula III; and c, performing hydrogenolysis on the intermediate II obtained in the step b to de-protect and re-crystallizing to obtain the 2-deoxy-D-glucoses shown as a formula I. Compared with the prior art, the preparation method of the 2-deoxy-D-glucoses has the advantages of simple preparation process, easy purification for products, high yield, low cost and the like, and is more beneficial to achieving large-scale industrial production.
Owner:SHANDONG LUKANG SHELILE PHARMA
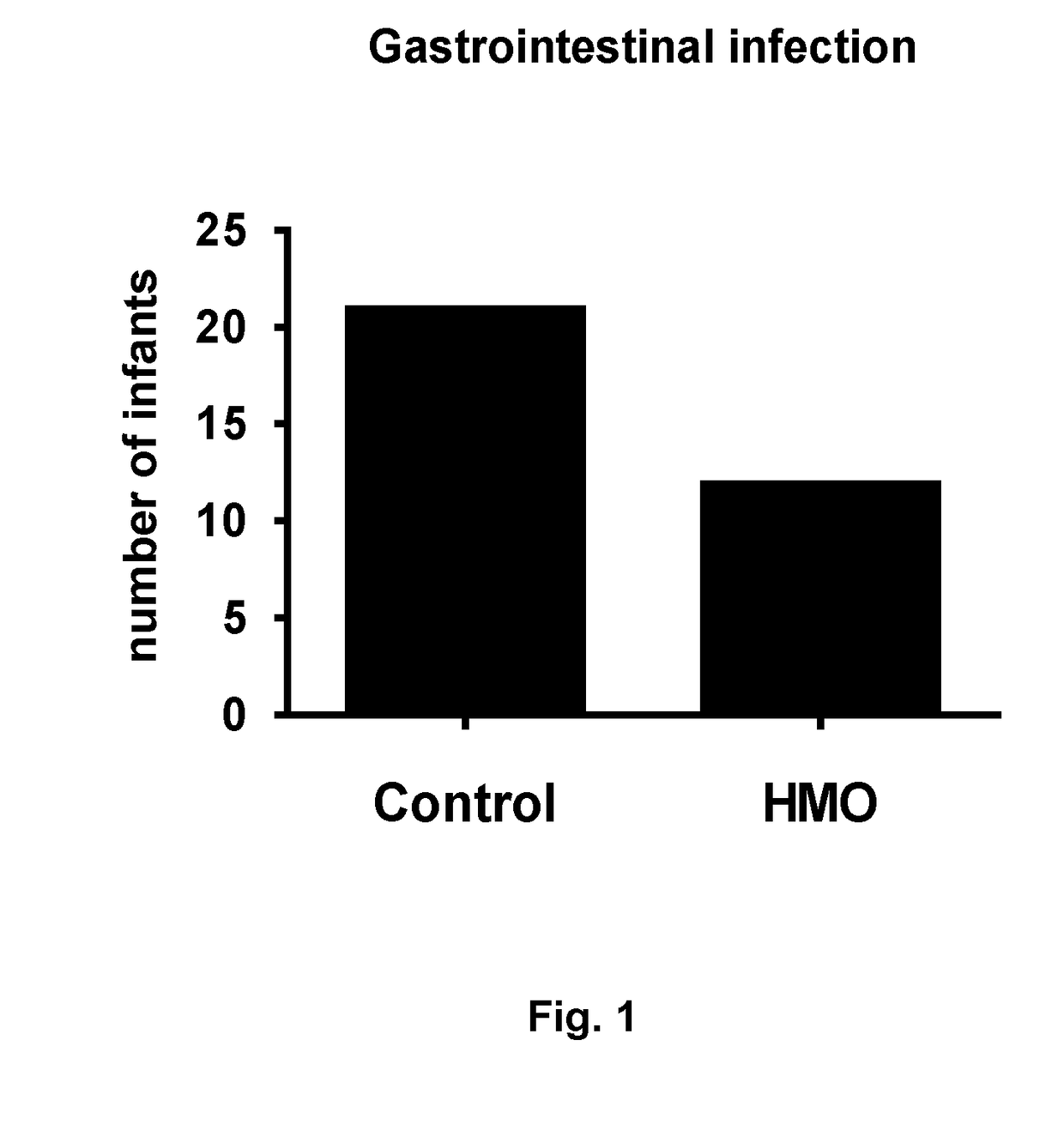
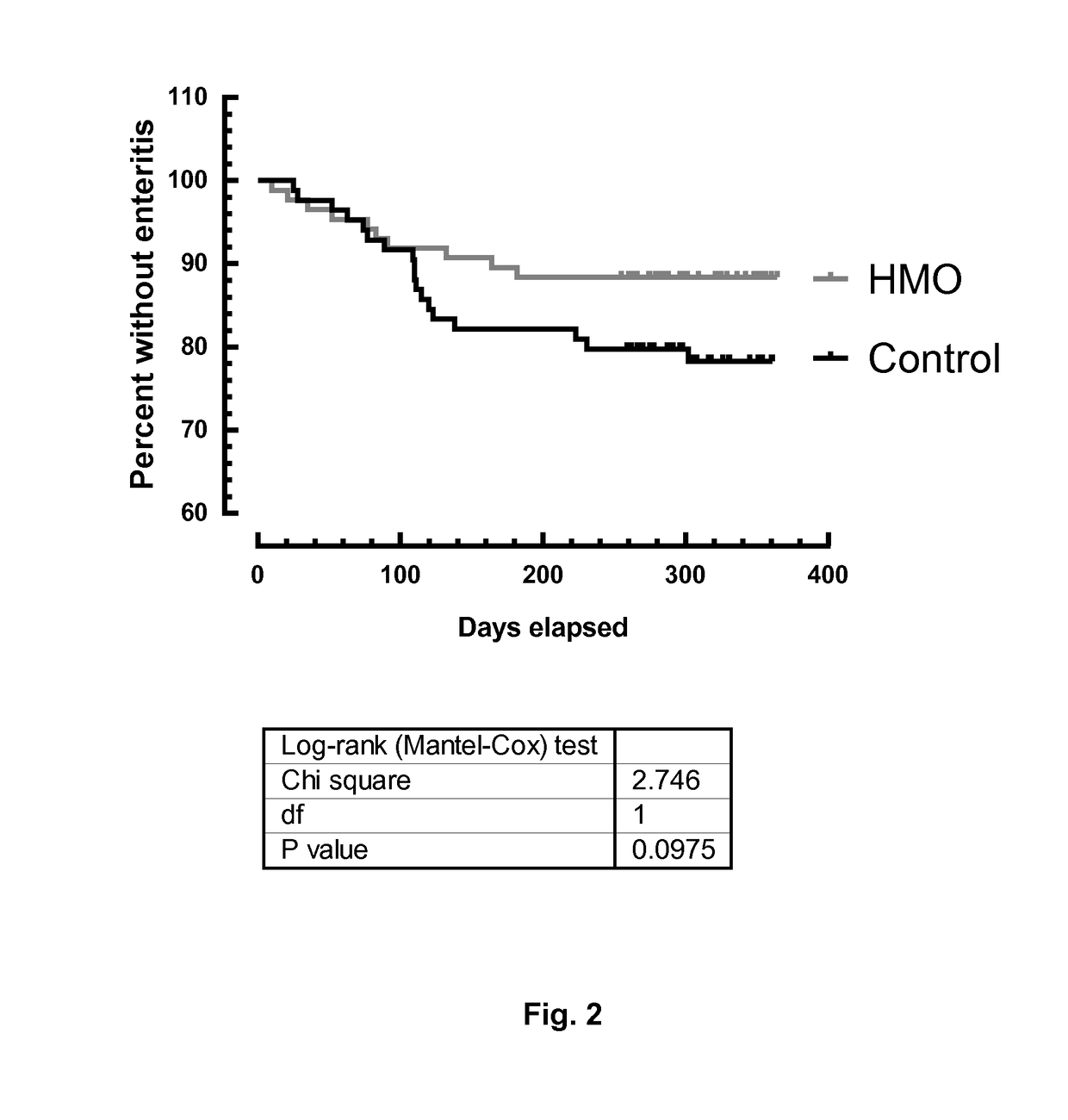
Nutritional compositions with 2fl and lnnt for use in inducing a gut microbiota close to the one of breast fed infants
PendingUS20180220690A1Eliminate and decrease populationReduced virulenceEsterified saccharide compoundsOrganic active ingredientsFucosylationIntestinal microorganisms
The present invention relates to a nutritional composition comprising at least one fucosylated oligosaccharide and at least one N-acetylated oligosaccharide for promoting or inducing a global gut microbiota that is closer to the one of infants fed exclusively with human breast milk, in comparison to infants fed with a conventional nutritional composition. The composition can be an infant formula and is in particular intended for infants between 0 and 12 months of age fed predominantly with infant formula. It promotes a healthy intestinal flora and has beneficial short and long terms effects.
Owner:SOC DES PROD NESTLE SA
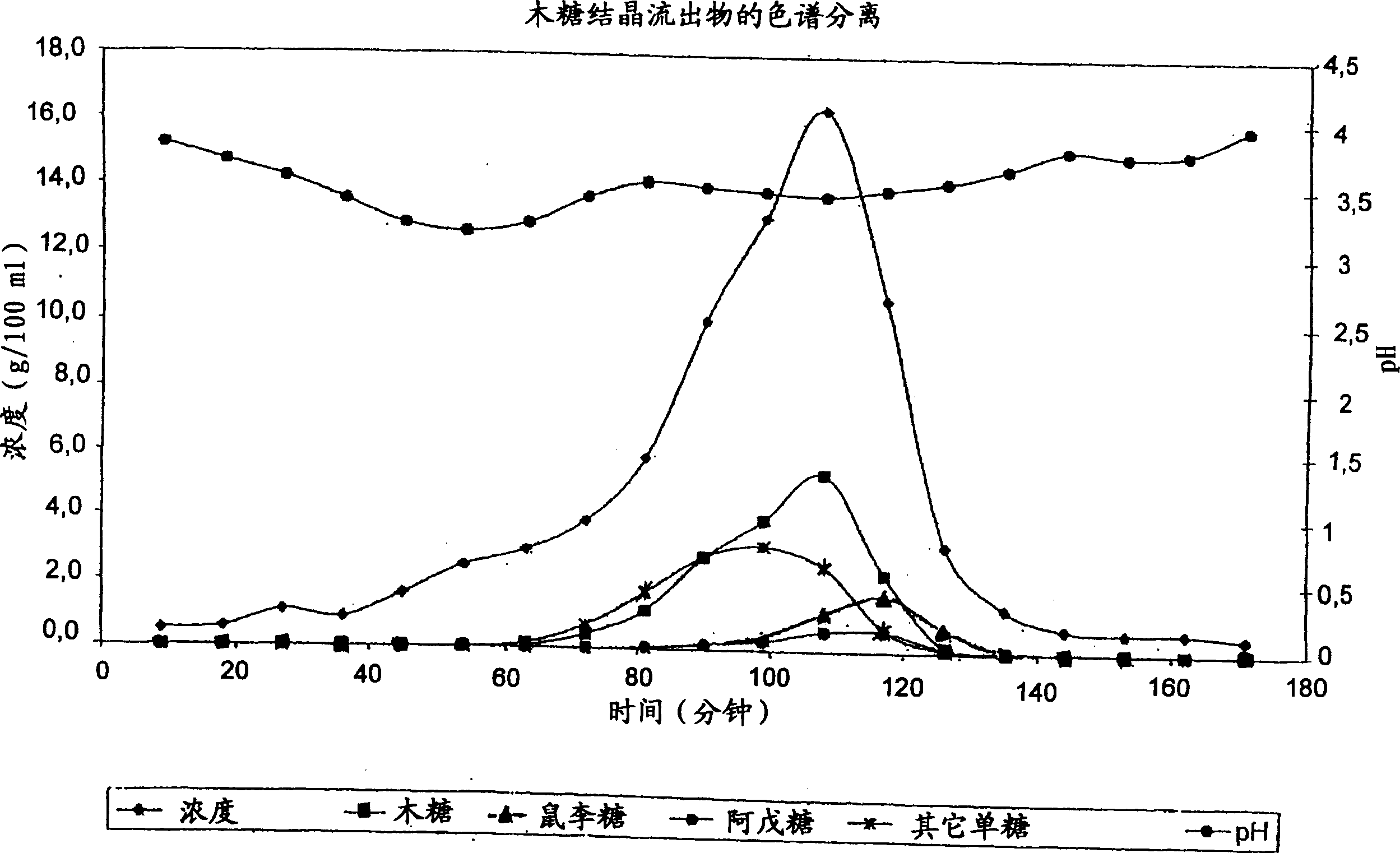
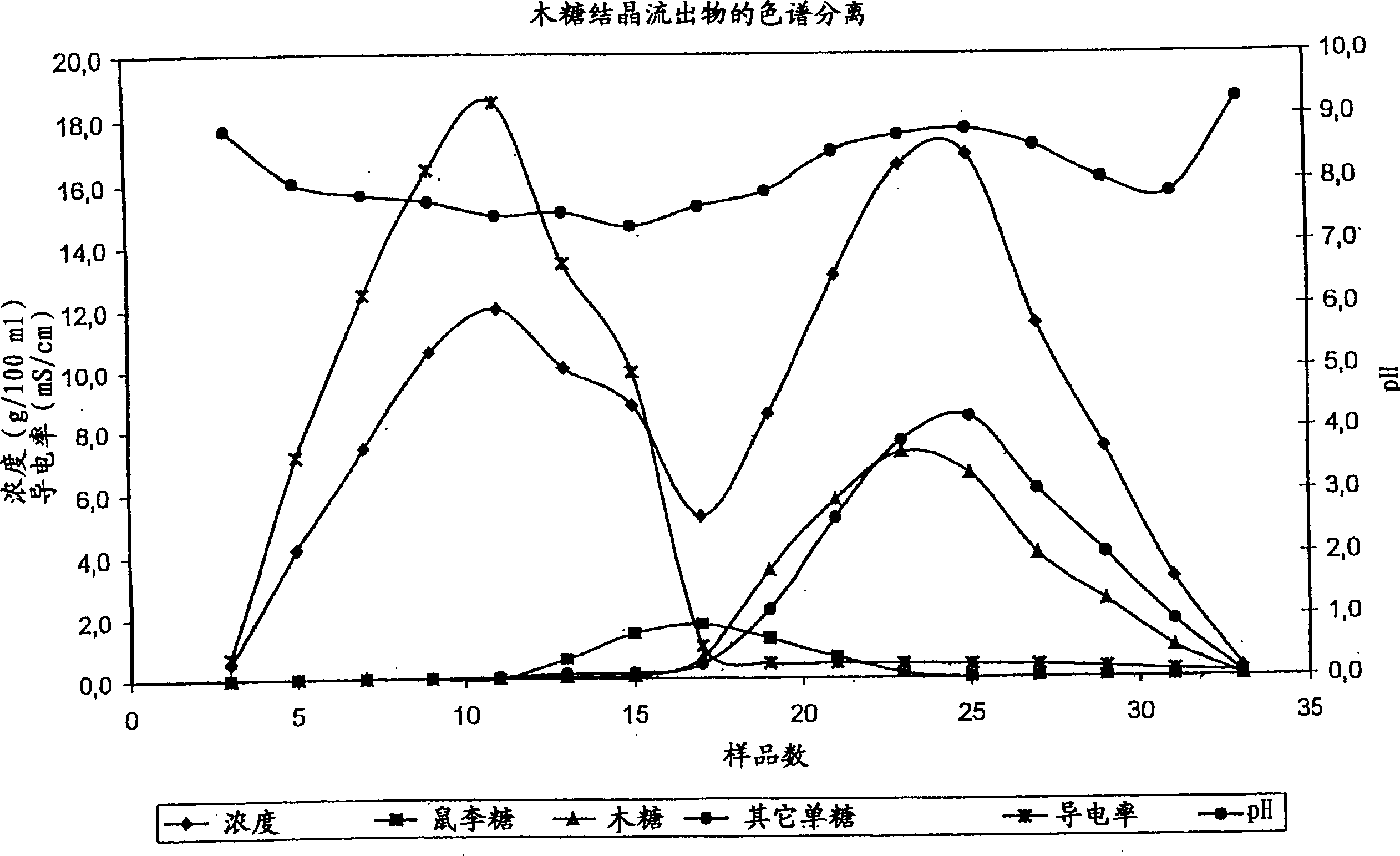
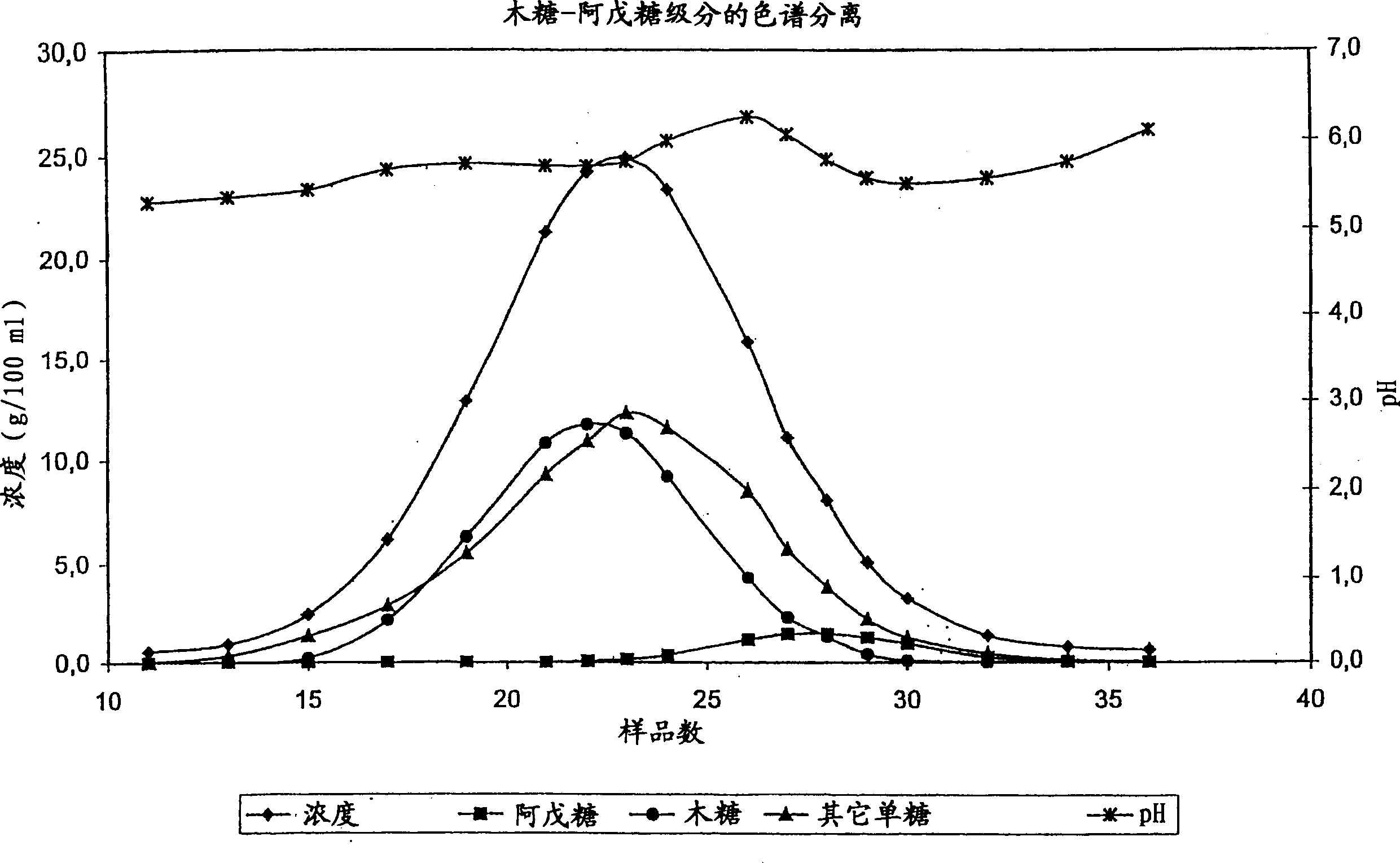
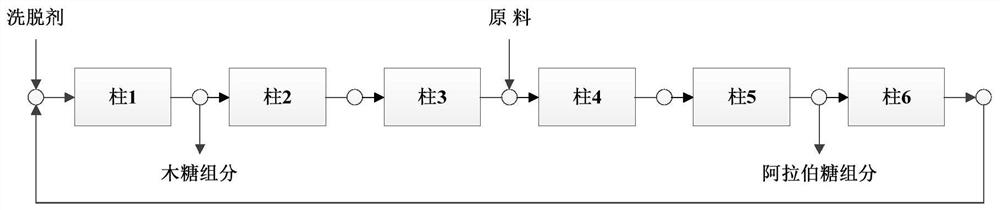
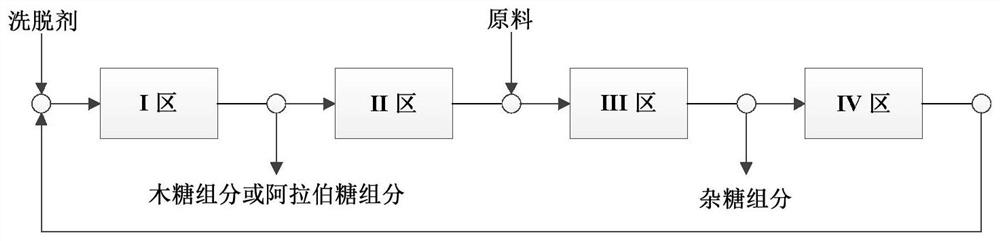
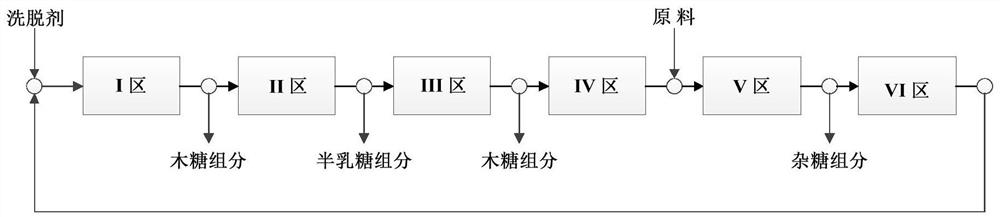
Method for simultaneously preparing xylose, arabinose and galactose by utilizing intermittent simulated moving bed chromatography
ActiveCN111747997AFully tap the valueHigh yieldSugar derivativesSugar derivatives preparationXylose fermentationBiochemical engineering
The invention discloses a method for simultaneously preparing xylose, arabinose and galactose by utilizing intermittent simulated moving bed chromatography, and belongs to the technical field of foodindustry separation. The method comprises the following steps: (1) pretreating xylose mother liquor; and (2) separating xylose, arabinose and galactose by intermittent simulated moving bed chromatography. According to the invention, xylose, arabinose and galactose can be simultaneously prepared from xylose mother liquor or xylose fermentation liquor with high purity and high yield; the problems that the sugar component recovery rate is not high, the conventional chromatography does not meet the separation requirement, the existing chromatography multi-component separation system is complex, the fixed phase demand is large, the separation function area is subjected to cross contamination and the like are solved; the problems that the crystallization yield of xylose and arabinose is relatively low due to galactose, galactose is difficult to consume by a fermentation method and the like are also solved, full recovery of high-value saccharides is realized, and the resource utilization rateis increased.
Owner:JIANGNAN UNIV
New method for preparing 2-deoxy-D-glucose
InactiveCN1775791AImprove performanceGood antiviral effectDeoxy/unsaturated sugarsD-Glucose2-Deoxy-D-glucose
The invention discloses a method of preparing 2-deoxidation-D-glucose. It solves the problems of the complex manufacturing technique and high cost for existing 2-deoxidation-D-glucose. The method includes the following steps: preparing 1.2-deoxidation-2-phenylenemethylamino-D-glucose; preparing 2.2-deoxidation-2- phenylenemethylamino-3,4,5,6-four-O-acetyl-D-glucose; preparing 4.2-deoxidation -2-chorine-3,4,5,6-four-O-acetyl-D-glucose; preparing 5.2-deoxidation-3,4,5,6- four-O-acetyl-D-glucose; preparing 6.2-deoxidation-D-glucose. The raw material of the method is far ranging, and low cost. It has great economic, social, and environment benefit.
Owner:YUHUAN MARINE BIOCHEM
Preparation method of 2-deoxy-L-ribose
ActiveCN103694279AEasy to purifyHigh yieldSugar derivativesSugar derivatives preparationHydration reactionPolyolefin
The invention discloses a preparation method of 2-deoxy-L-ribose. The preparation method comprises the following steps: preparing beta-bromo-L-triacetyl arabinose by using L-arabinose as a starting material, acetyl bromide serving as a protecting group and a brominating reagent, adding the beta-bromine-L-tri-O-acetyl-arabinose into a reaction system containing zinc powder, sodium bisulfate, copper sulfate and water, reacting at normal temperature, and then diluting, filtering, washing, concentrating under reduced pressure and rectifying the reaction products to produce acetylated acetylated L-arabinal with the purity of larger than 99%; dissolving the acetylated acetylated L-arabinal into ethanol, adding potassium carbonate into the mixture to react, so as to generate L-arabinose olefin as an intermediate; performing hydration addition on the acetylated L-arabinal under composite catalyst action of a sulphoalkyl pyrazine sulphate-long-chain polyolefin sulfonic acid resin, so as to produce the 2-deoxy-L-ribose. The used raw materials are conventional reagents, while the reaction conditions are milder, the operation method and the post-treatment processes are simple, and the product yield and purity are relatively desirable.
Owner:JIANGXI SUKEER NEW MATERIAL
Synthesis of trichlorosucrose
InactiveCN1176094CSimple processHigh purityEsterified saccharide compoundsSugar derivativesAcetic acidSulfate
The present invention discloses the synthesis of trichlorosucrose. Sucrose material is mixed with N, N-dimethyl formamide solution, made to produce ester exchange reaction with ethyl acetate in the action of solid sulfate catalyst, which may be adsorbed on polymer carrier, to produce sucrose-6-acetate. Sucrose-6-acetate is then chlorohydrinated to produce trichlorosucrose. The present invention has the advantages of simple technological process, high product purity, low production cost, etc. and is suitable for industrial production.
Owner:广东广业清怡食品科技股份有限公司
Compositions for use in the prevention or treatment of gastrointestinal infections/inflammations in infants or young children
ActiveUS20180036323A1Organic active ingredientsVitamin food ingredientsGastrointestinal inflammationNutrition
The present invention relates to a nutritional composition comprising at least one fucosylated oligosaccharide and at least one N-acetylated oligosaccharide in a particular ratio, for use in preventing and / or treating gastrointestinal infections and / or gastrointestinal inflammations in an infant or a young child.
Owner:SOC DES PROD NESTLE SA
Simple synthesis of D-and L-deoxyribose
InactiveCN1349999ARaw materials are cheap and easy to getThe synthetic route is simpleChemical recyclingBulk chemical productionPropanolSynthesis methods
The present invention relates to a simple high-effective synthesis method of D-and L-desoxyribose, which includes the following steps: using cheap easily-available D-high-alkyne propanol or L-high-alkyne propanol as raw material, protecting hydroxyl group, using Lindlar catalyst to make selective hydrogenation, using ozone to make oxidation according to different substituent making it react with acid, then react with alkali or react with reducing agent or lewis acid to remove protecting group and making cyclization reaction to obtain the invented product.
Owner:SHANGHAI INST OF ORGANIC CHEM CHINESE ACAD OF SCI
Method for preparing z-deoxy-D-glucose
InactiveCN1850835AReasonable designFew reaction stepsDeoxy/unsaturated sugarsNef reactionDouble bond
This invention relates to 2-deoxygenation-D-glucose preparation method. The method is that D-arabinose is reacted with nitromethane, and the reaction prodect is material, 2 bit acetoxy is selective stripped to get nitryl sugar ene. Borohydride isused to selectively reduce conjugated carbon carbon double bond of the sugar ene to get saturated nitryl sugar. Then the goal compound 2-deoxygenation-D-glucose I sogt after Nef reaction. In this invention, the selectivity is good, yield is improved, the material source is wide, cost is low, and its technique design is reasonable.
Owner:WUHAN UNIV OF TECH
Method for producing a 2-deoxy-L-ribose
InactiveCN1668626AEasy to synthesizeLow toxicitySugar derivativesSugar derivatives preparationD-GlucoseRibose
The present invention relates to a economic synthetic method of 2-deoxy-L-ribose from 2-deoxy-D-ribose with easy reaction, separation and purification. The present invention consists of four(4) steps including protection, activation 3-and 4-OH groups, inversion and deprotection step. In respect to the cost for equipment, reagent and operation, by the present invention, 2-deoxy-L-ribose can be produced more economically because the invention uses 2-deoxy-L-ribose which is abundant in nature and easily synthesized from D-glucose, and adopt simple and yielding process.
Owner:SAMCHULLY PHARM CO LTD
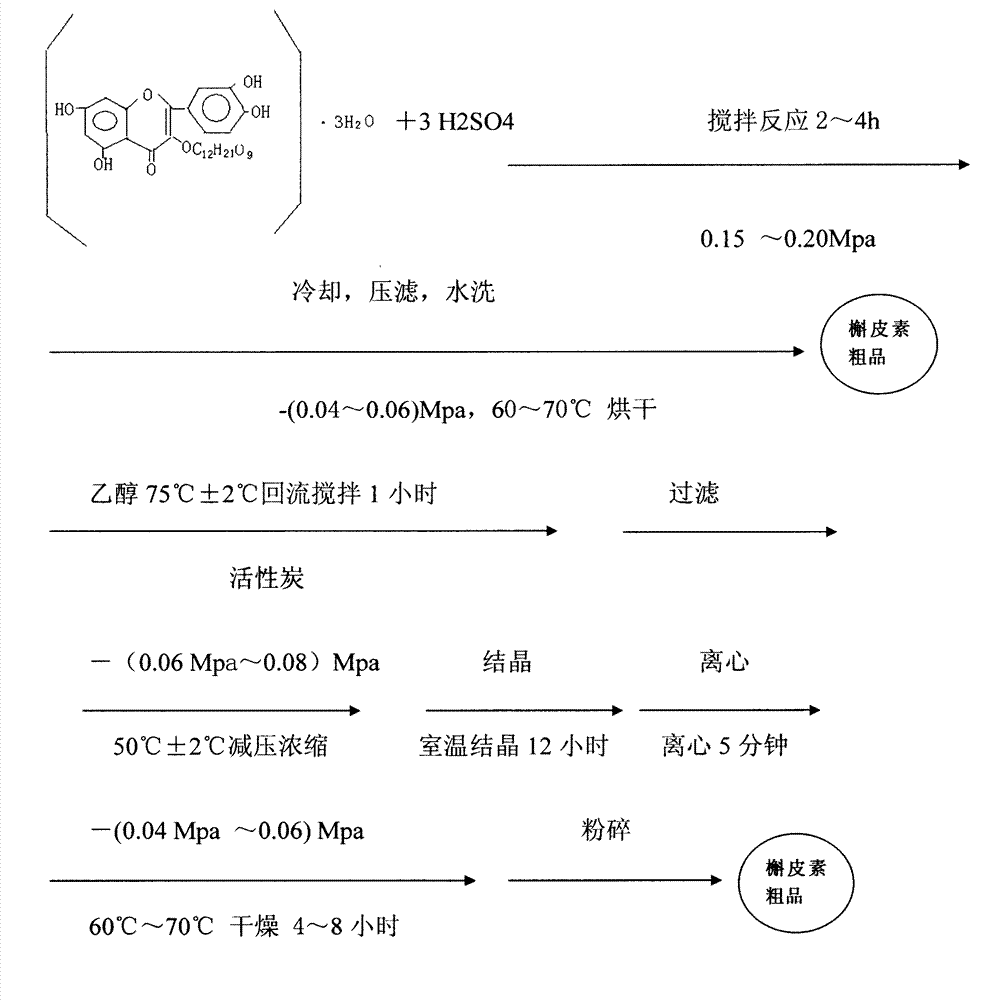
Method for preparing quercetin and rhamnose through rutin
InactiveCN103965153AAvoid pollutionRealize the effect of environmental protection and energy savingSugar derivativesSugar derivatives preparationFiltrationBud
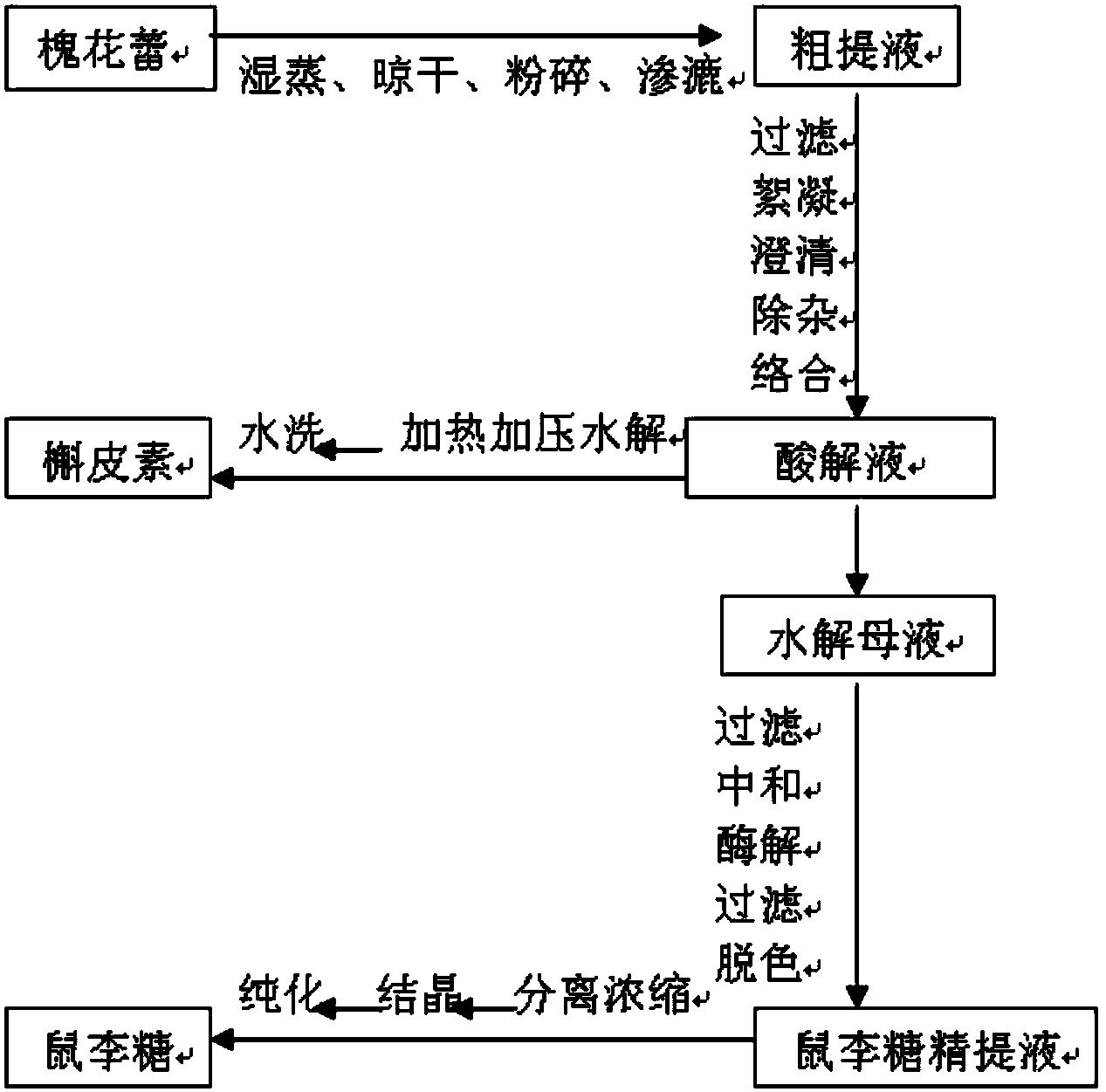
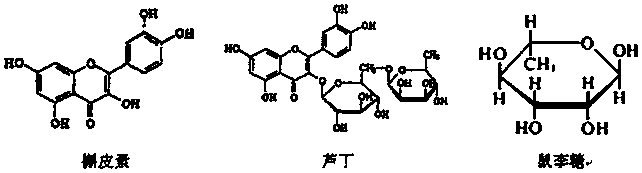
The invention belongs to the field of flavonoids compound extraction, and particularly relates to a method for preparing quercetin and rhamnose through rutin. The preparation method comprises the following steps: steaming collected sophora flower buds through steam, air-drying, smashing and seeping the steamed sophora flower buds, removing colloid and chlorophyll impurities through flocculation, performing protection complexing treatment through an antioxidant and a complexing agent, adding a mixed acid to adjust the mixture to be acidic, and heating, pressurizing and hydrolyzing to obtain the quercetin; performing impurity filtration and neutralization on hydrolysis mother liquor subjected to quercetin extraction, degrading glucose through glucose degrading enzyme, and performing decoloring vacuum centrifugal concentration and crystallization after a solution is filtered and separated to obtain a product, namely the rhamnose. Under detection of HPLC (high performance liquid chromatography), the content of the quercetin is over 95 percent; all impurity requirements meet an international standard requirement; in a technical process, rutin hydrolysis mother liquor is also subjected to comprehensive treatment, and rhamnose is obtained through production; the production cost of the production is lowered, and the environmental pollution is alleviated.
Owner:罗俊
Sarcopyramis nepalensis extract and application thereof
InactiveCN104804056AAchieve pharmacological activityDigestive systemGlycoside steroidsCytokine NetworkAlcohol
The invention discloses a sarcopyramis nepalensis extract. A preparation method of the sarcopyramis nepalensis extract comprises the following steps: taking whole herb of sarcopyramis nepalensis; after crushing the taken whole herb of the sarcopyramis nepalensis, extracting the crushed whole herb of the sarcopyramis nepalensis with ethyl alcohol of 95% for six times, wherein the extraction time each time is one week; performing the filtration, and performing the concentration so as to obtain a medicinal extract. The extract can be used for medicines for nourishing a liver and reducing transaminase, the sarcopyramis nepalensis extract disclosed by the invention can widely affect all the cytokine levels, pharmacological activities of nourishing the liver and reducing the transaminase are realized through regulating the balance of a cytokine network, and scientific basis is provided for the clinical application of the sarcopyramis nepalensis extract.
Owner:王燕燕
Method for synthesizing 2-deoxy-L-ribose
InactiveCN101407530AEasy to operatePost-processing is simpleDeoxy/unsaturated sugarsOrganic acidAcetic acid
The invention discloses a method for synthesizing 2-deoxidization-L-ribose, and the method is characterized in that 1-methyl-3, 4-O-isopropyl-2-deoxidization-beta-L-gum sugar reacts in water solution of organic acid with acidity more than that of acetic acid. The final product can be obtained in one step of reaction by the method, and the method has the advantages of simple reaction operation, easy post processing, without the need of column chromatography, high yield and purity, thus being suitable for industrialization production.
Owner:SHANGHAI INST OF PHARMA IND
Nutritional compositions with 2FL and LNnT for use in inducing a gut microbiota close to the one of breast fed infants
ActiveCN107847509ADiversity impactAvoid dissonanceEsterified saccharide compoundsOrganic active ingredientsFucosylationIntestinal microorganisms
The present invention relates to a nutritional composition comprising at least one fucosylated oligosaccharide and at least one N-acetylated oligosaccharide for promoting or inducing a global gut microbiota that is closer to the one of infants fed exclusively with human breast milk, in comparison to infants fed with a conventional nutritional composition. The composition can be an infant formula and is in particular intended for infants between 0 and 12 months of age fed predominantly with infant formula. It promotes a healthy intestinal flora and has beneficial short and long terms effects.
Owner:SOC DES PROD NESTLE SA
Method for preparing 2-deoxy-D-glucose
ActiveCN106496288ARaw materials are cheap and easy to getDoes not involve separationSugar derivativesSugar derivatives preparationAcetic anhydrideSilanes
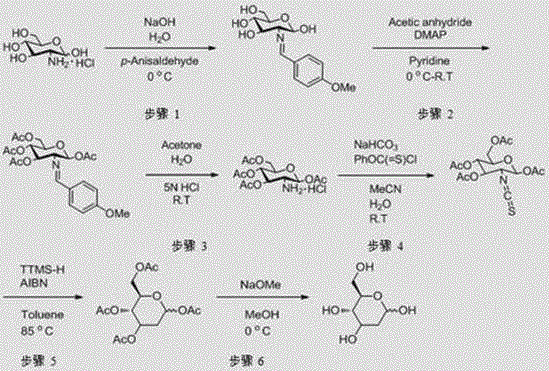
The invention discloses a method for preparing 2-deoxy-D-glucose. According to the method, D-glucosamine which is low in price and easy to obtain serves as a starting material, p-anisaldehyde is used for protecting amino, pyridine serves as a solvent under catalysis of 4-dimethylaminopyridine (DMAP), acetic anhydride serves as a reactant for acetylation protection of all hydroxyl groups on D-glucosamine, protecting groups on the amino are removed under the acidic condition to obtain 1,3,4,6-tetra-o-acetyl-2-glucosamine hydrochloride, and the amino is further converted into isothiocyano with PhOC(=S)Cl; AIBN serves as an initiator, a free radical reaction is conducted by means of tris(trimethylsilyl)silane (TTMS-H) and 1,3,4,6-tetra-o-acetyl-2-glucosamine hydrochloride, and finally acetyl is removed to obtain 2-deoxy-D-glucose. According to the novel synthetic route, raw materials are easy to obtain, operation is easy, no chromatographic column separation is involved, the total yield is high, cost is low, and the method is suitable for large-scale production. The preparation method is simple in product aftertreatment, the product is easy to separate, and large-scale production is easy to achieve.
Owner:JINAN SAMUEL PHARM CO LTD