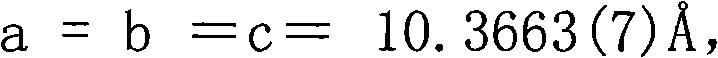Patents
Literature
123 results about "Non linear optical materials" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
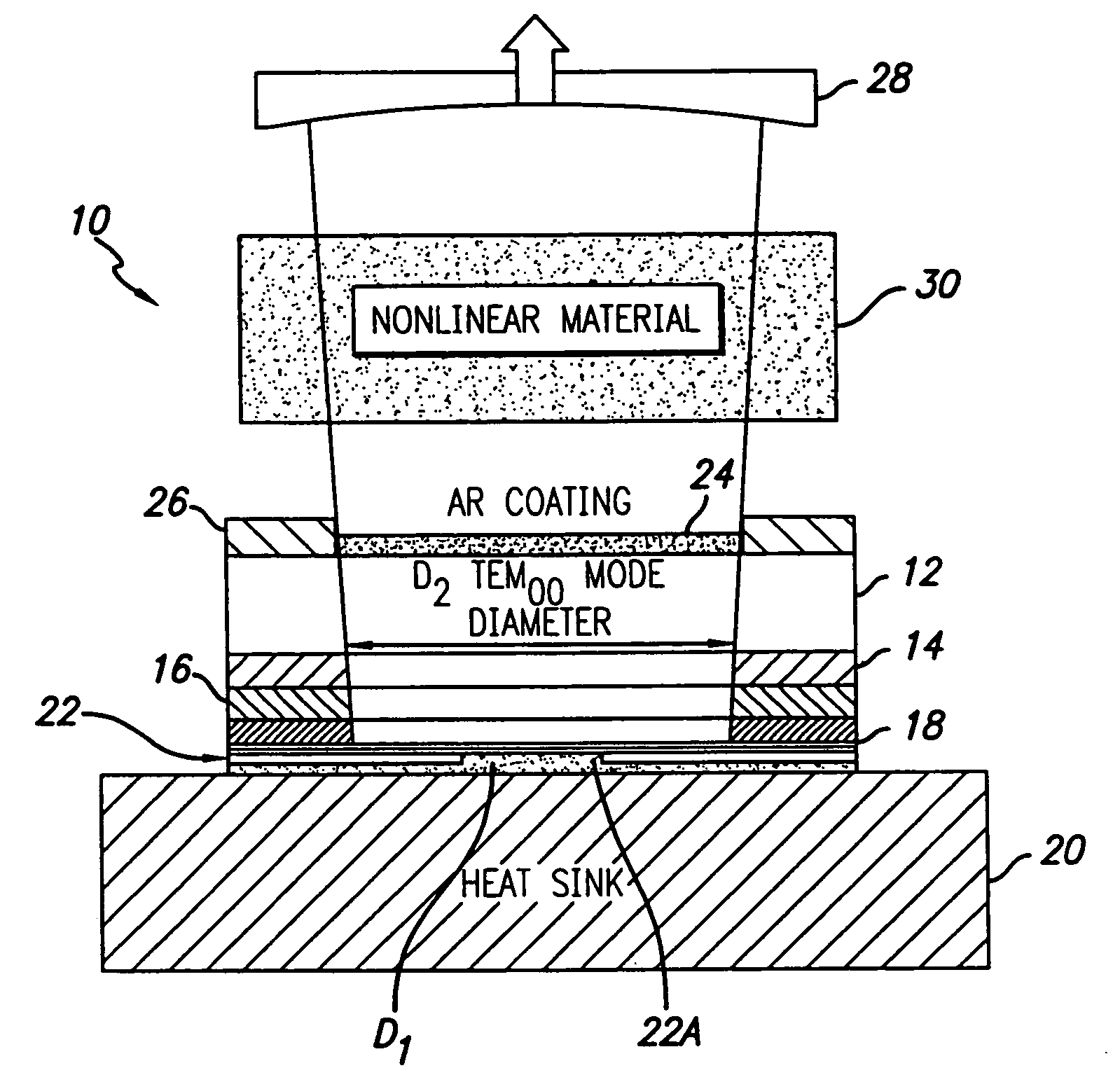
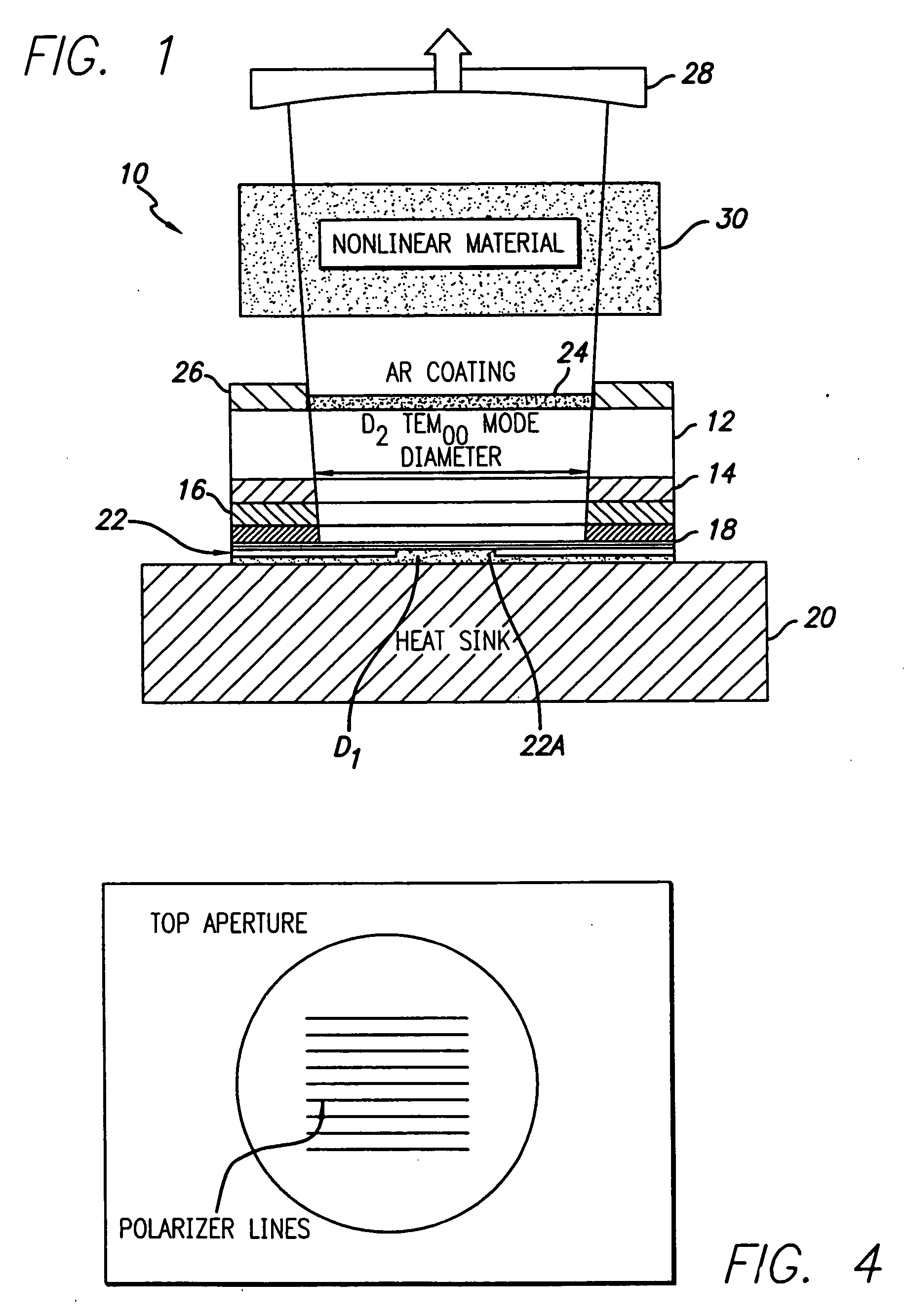
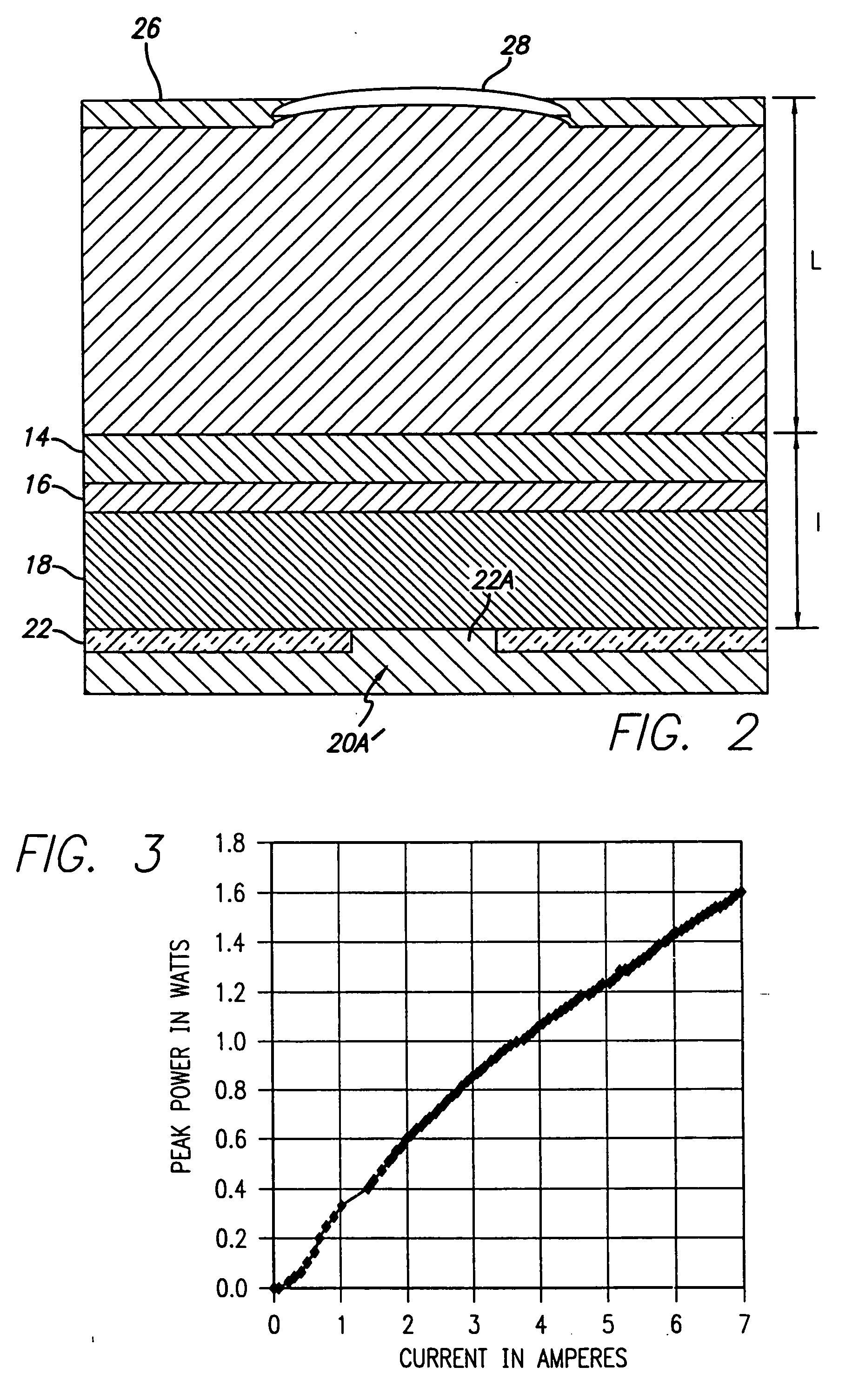
Coupled cavity high power semiconductor laser
InactiveUS20060029120A1Maximize circulating fundamental laser powerConvenient registrationOptical resonator shape and constructionSemiconductor lasersFundamental frequencyPartial reflection
An active gain region sandwiched between a 100% reflective bottom Bragg mirror and an intermediate partially reflecting Bragg mirror is formed on a lower surface of a supporting substrate, to thereby provide the first (“active”) resonator cavity of a high power coupled cavity surface emitting laser device. The reflectivity of the intermediate mirror is kept low enough so that laser oscillation within the active gain region will not occur. The substrate is entirely outside the active cavity but is contained within a second (“passive”) resonator cavity defined by the intermediate mirror and a partially reflecting output mirror, where it is subjected to only a fraction of the light intensity that is circulating in the gain region. In one embodiment, non-linear optical material inside each passive cavity of an array converts an IR fundamental wavelength of each laser device to a corresponding visible harmonic wavelength, and the external output cavity mirror comprises a Volume Bragg grating (VBG) or other similar optical component that is substantially reflective at the fundamental frequency and substantially transmissive at the harmonic frequency. The VBG used in an array of such devices may be either flat, which simplifies registration and alignment during manufacture, or may be configured to narrow the IR spectrum fed back into the active resonant cavity and to shape the spatial mode distribution inside the cavity, thereby reducing the size of the mode and compensating for any deformations in the semiconductor array.
Owner:ARASOR ACQUISITION +1
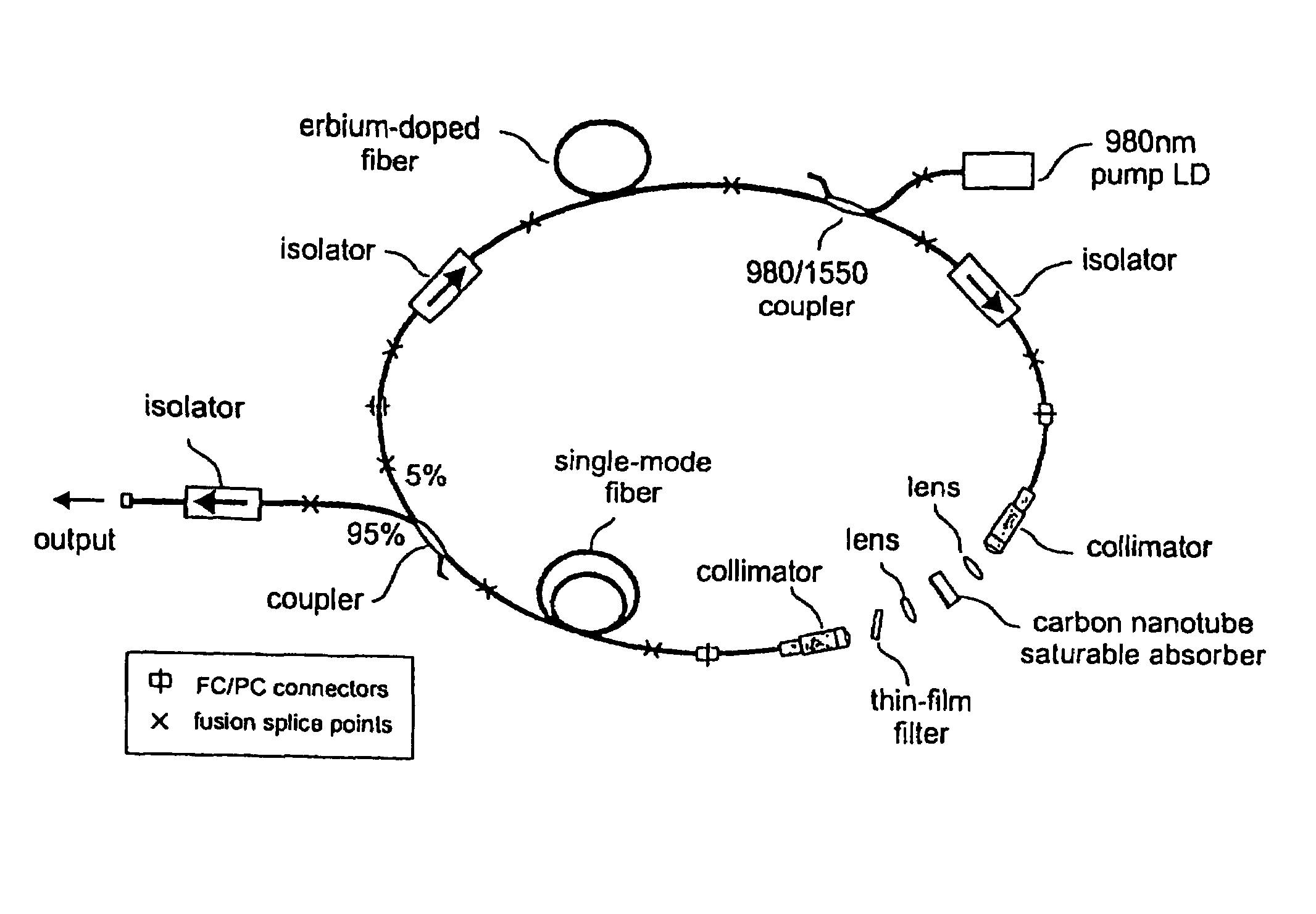
Optical pulse lasers
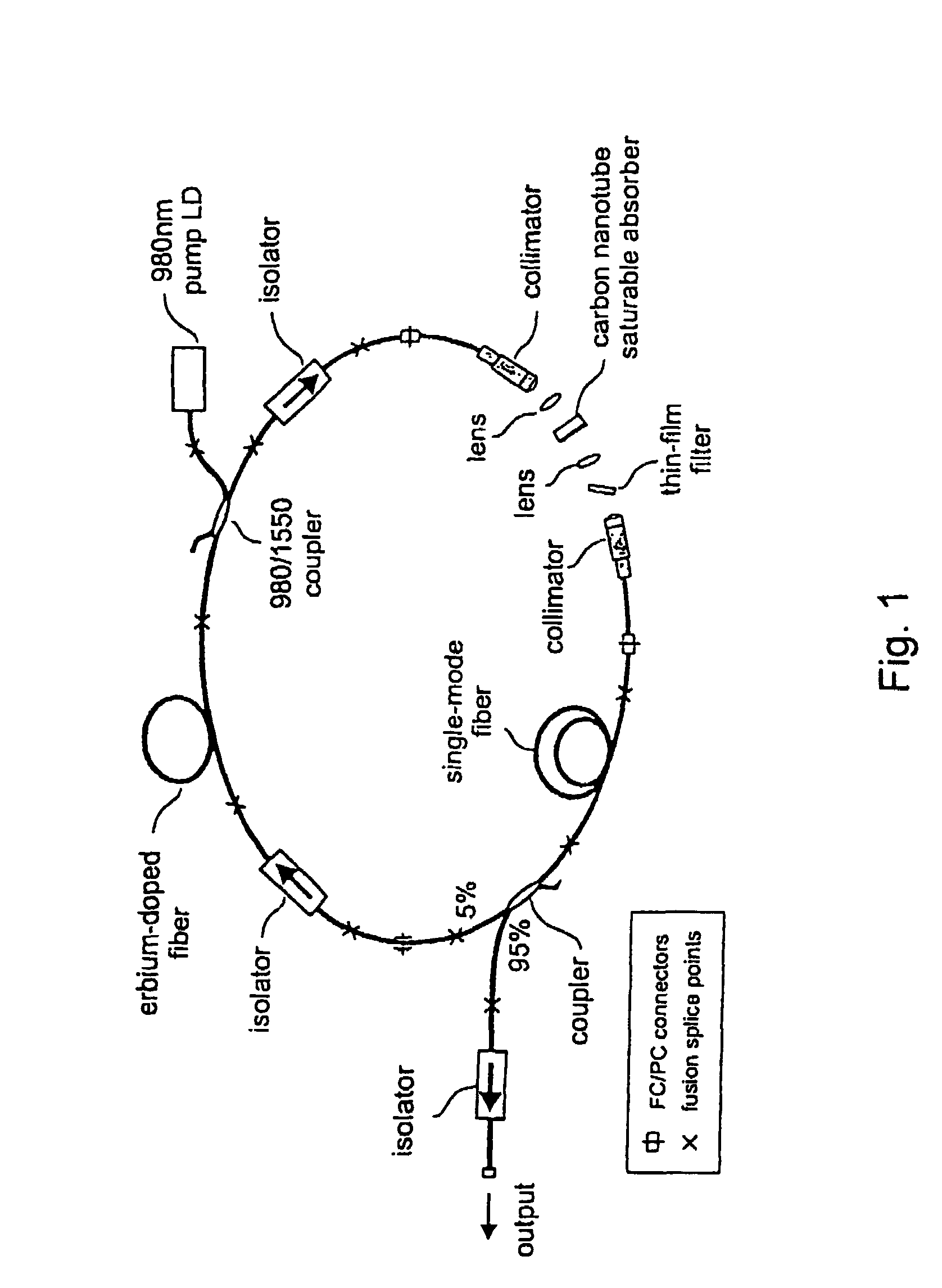
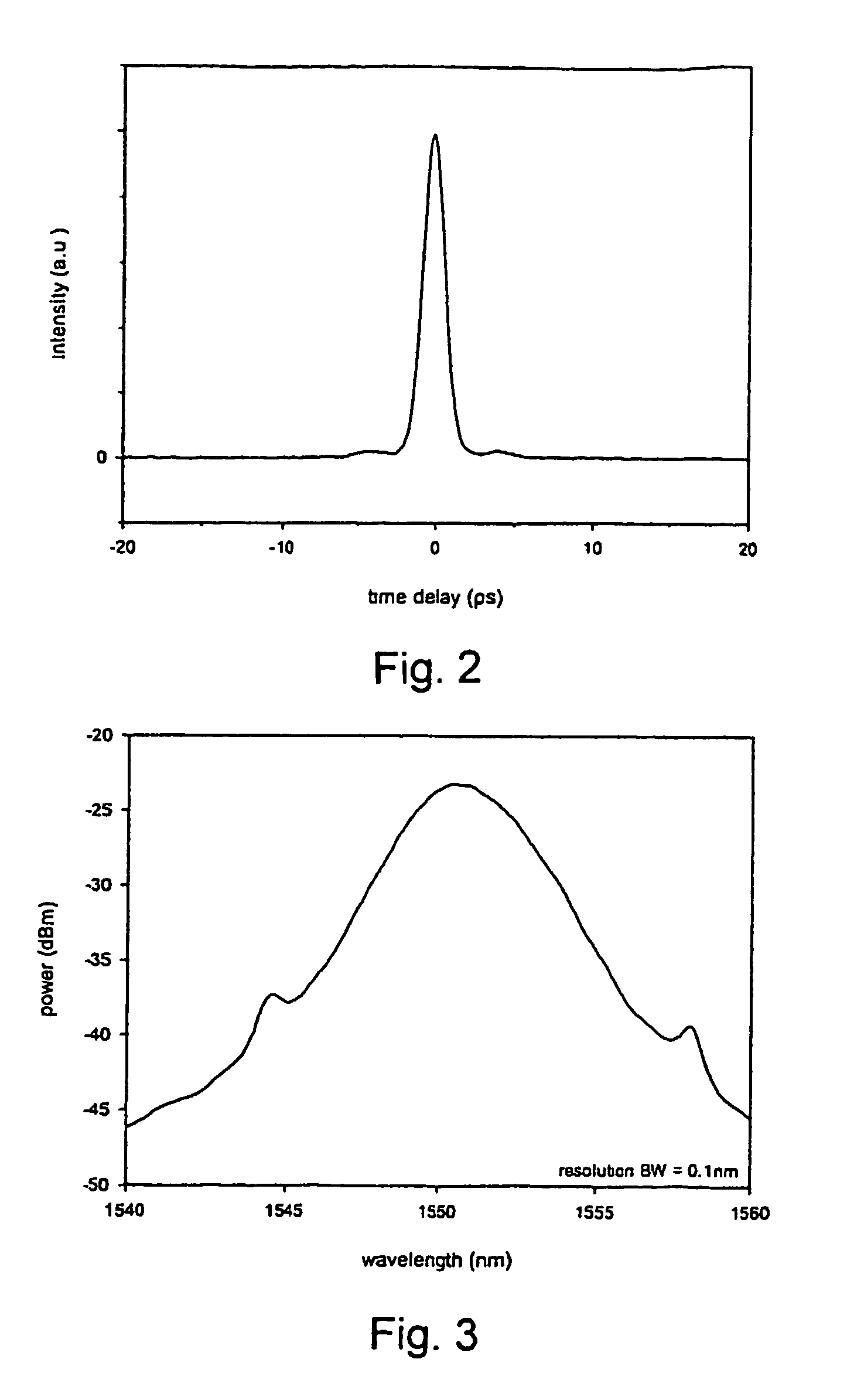
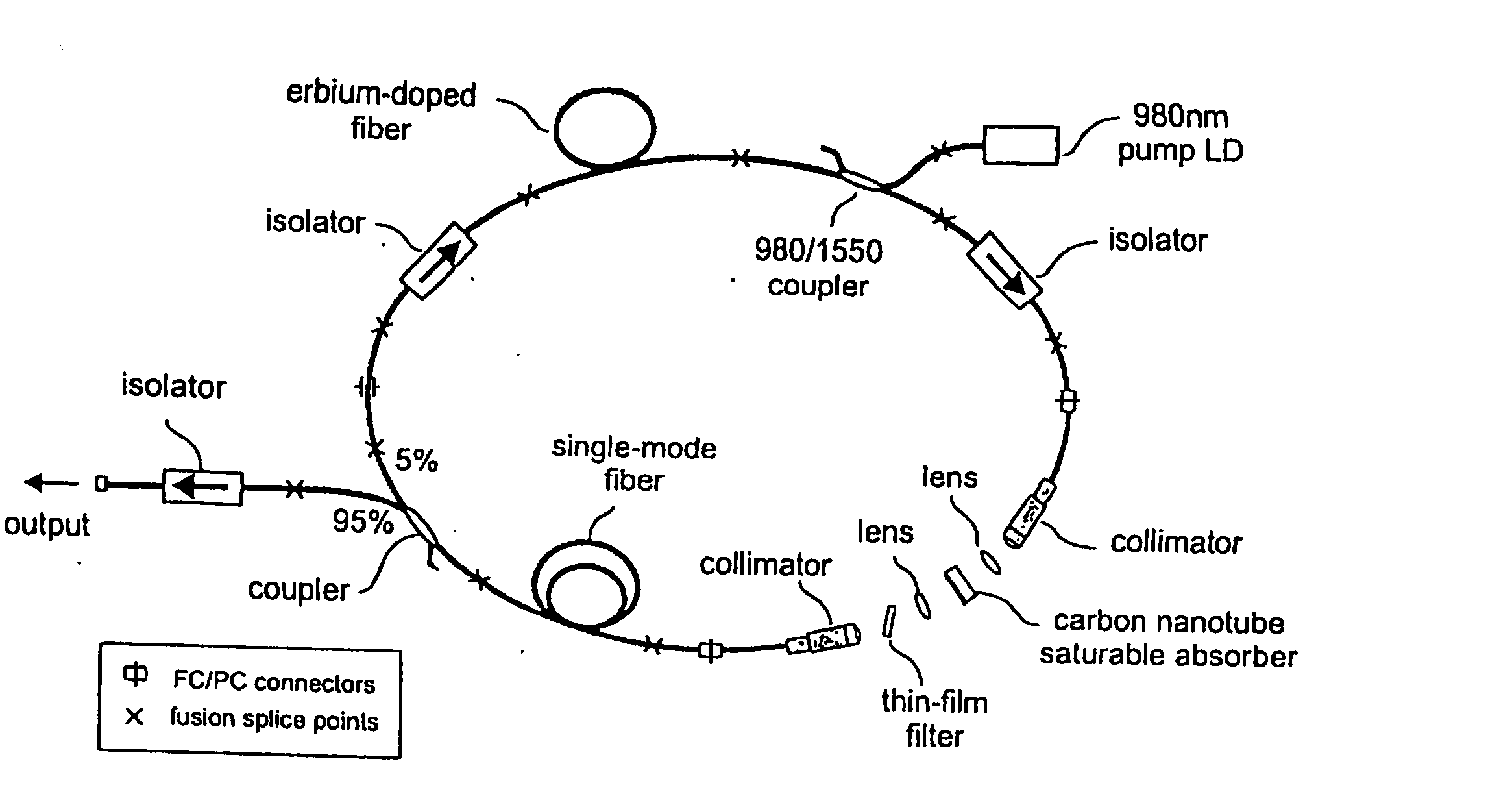
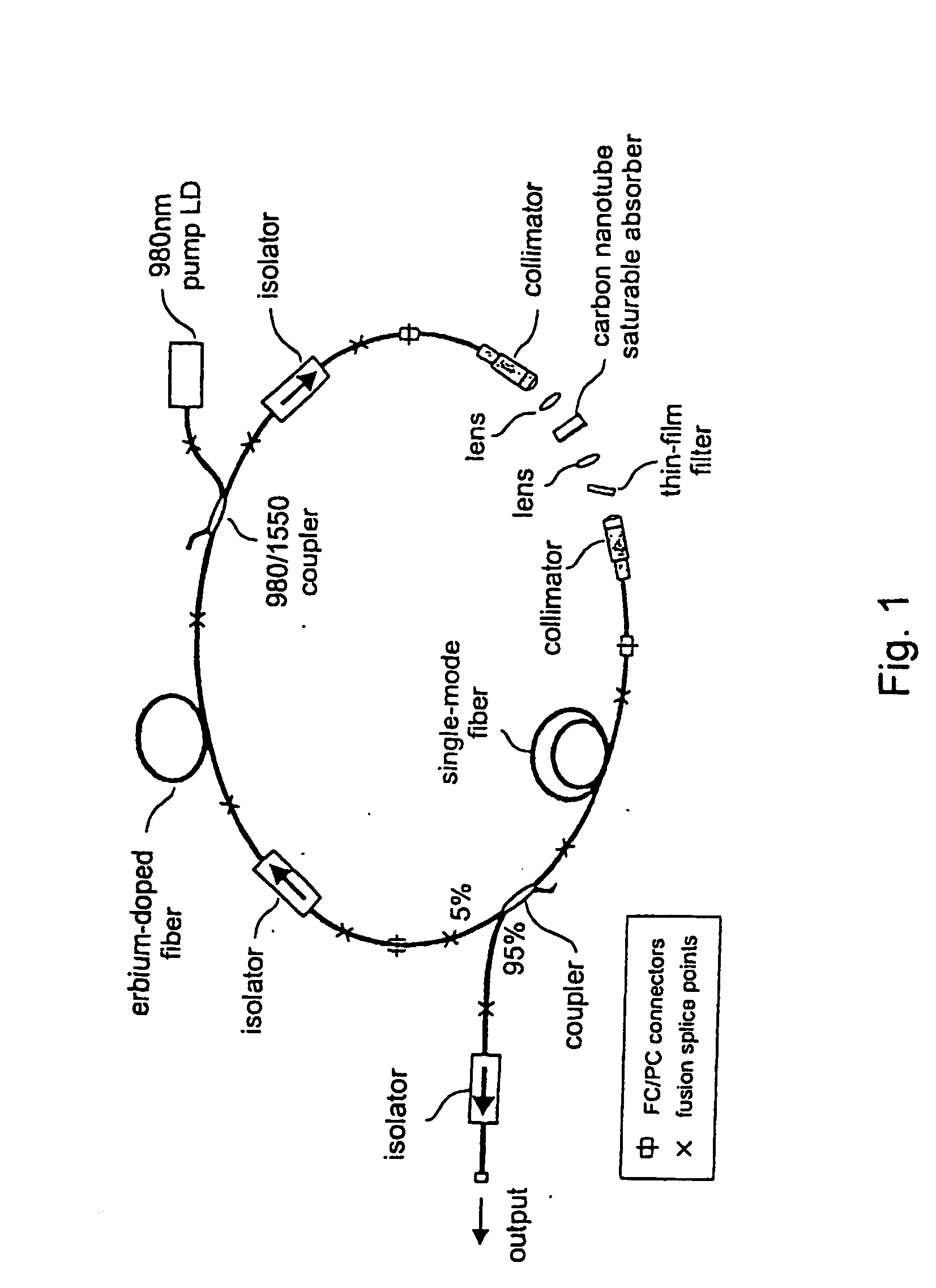
InactiveUS7372880B2Fast recovery timeHigh optical damage thresholdMirrorsNanoinformaticsCarbon nanotubeMode-locking
The present invention provides pulsed lasers which employ carbon nanotubes, particularly layers of carbon nanotubes, as saturable absorbers, mode lockers or for Q-switching elements. The present invention also provides methods and materials for mode-locking and Q-switching of lasers in which carbon nanotubes are employed as non-linear optical materials and / or saturable absorbers which facilitate mode-locking and / or Q-switching. The invention further provides mode locker and Q-switching elements or devices which comprise one or more layers containing carbon nanotubes which layer or layers function for mode locking and / or Q-switching.
Owner:NAT INST OF ADVANCED IND SCI & TECH +1
Optical pulse lasers
InactiveUS20060198399A1Easy to operateFast recovery timeMirrorsNanoinformaticsMode-lockingCarbon nanotube
The present invention provides pulsed lasers which employ carbon nanotubes, particularly layers of carbon nanotubes, as saturable absorbers, mode lockers or for Q-switching elements. The present invention also provides methods and materials for mode-locking and Q-switching of lasers in which carbon nanotubes are employed as non-linear optical materials and / or saturable absorbers which facilitate mode-locking and / or Q-switching. The invention further provides mode locker and Q-switching elements or devices which comprise one or more layers containing carbon nanotubes which layer or layers function for mode locking and / or Q-switching.
Owner:NAT INST OF ADVANCED IND SCI & TECH +1
Method for producing non-linear optical organic crystal film
InactiveUS6941051B2Semiconductor/solid-state device manufacturingOptical waveguide light guideOrganic crystalNon linear optical materials
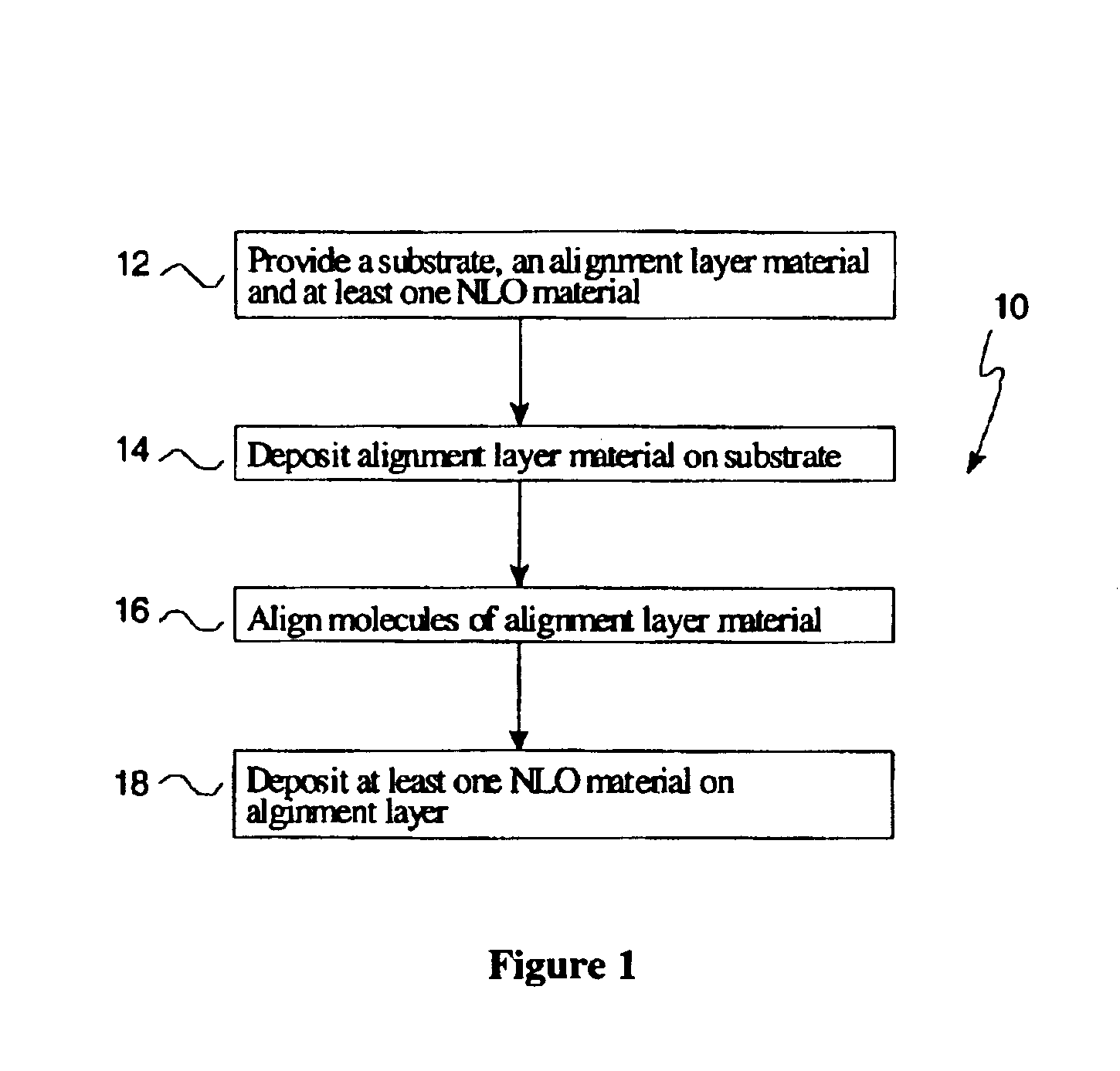
Crystalline non-linear optical films for nonlinear-optics applications are provided. One such crystalline non-linear optical film includes a substrate and an alignment layer material disposed on said substrate. The alignment layer includes elongated polymeric molecules aligned in a substantially uniaxial direction. Further, at least one non-linear optical material is disposed on the alignment layer.
Owner:REVEO
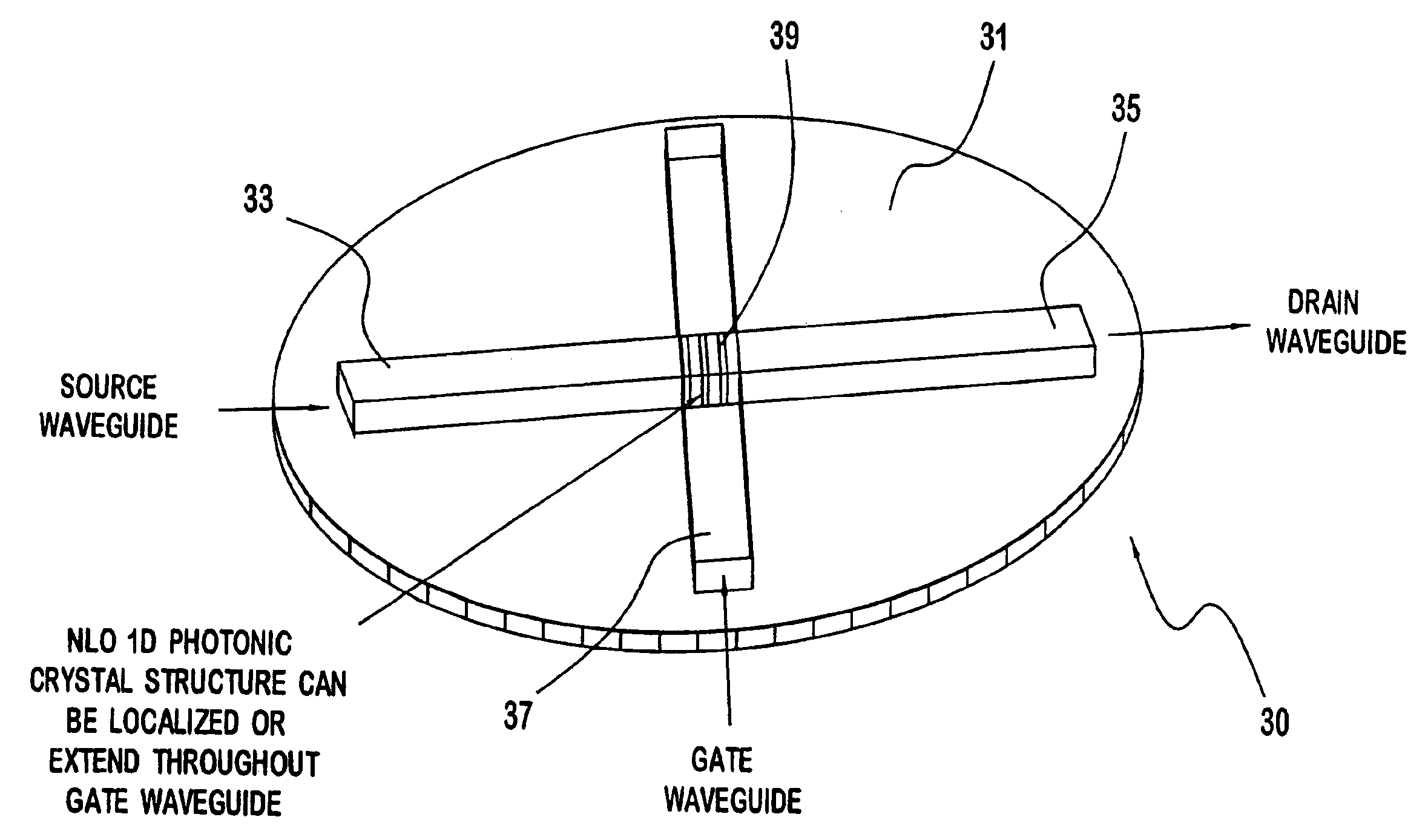
Non-linear photonic switch and method of making the same
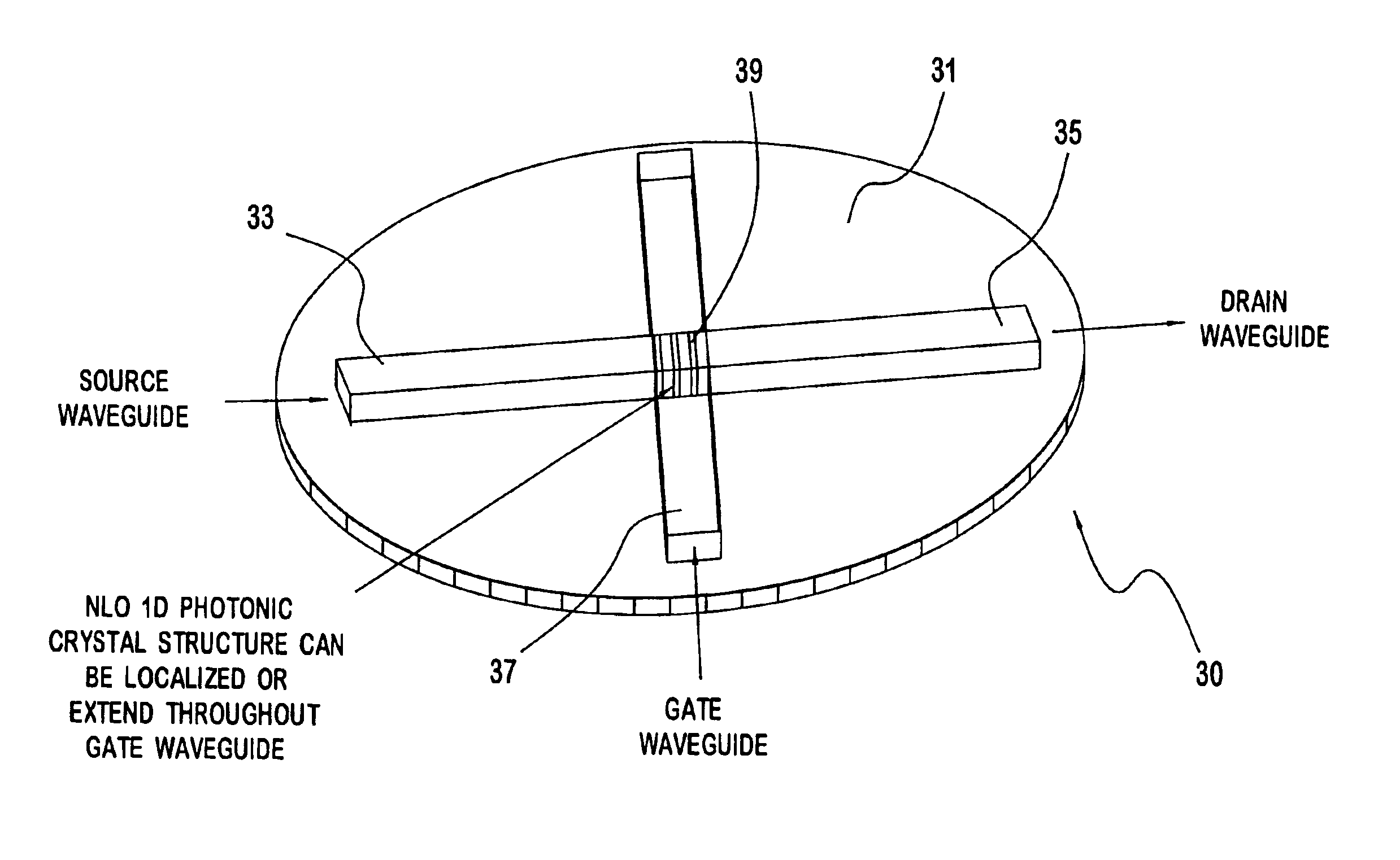
InactiveUS6937804B2Solid-state devicesSemiconductor/solid-state device manufacturingMicrocontact printingPhotonics
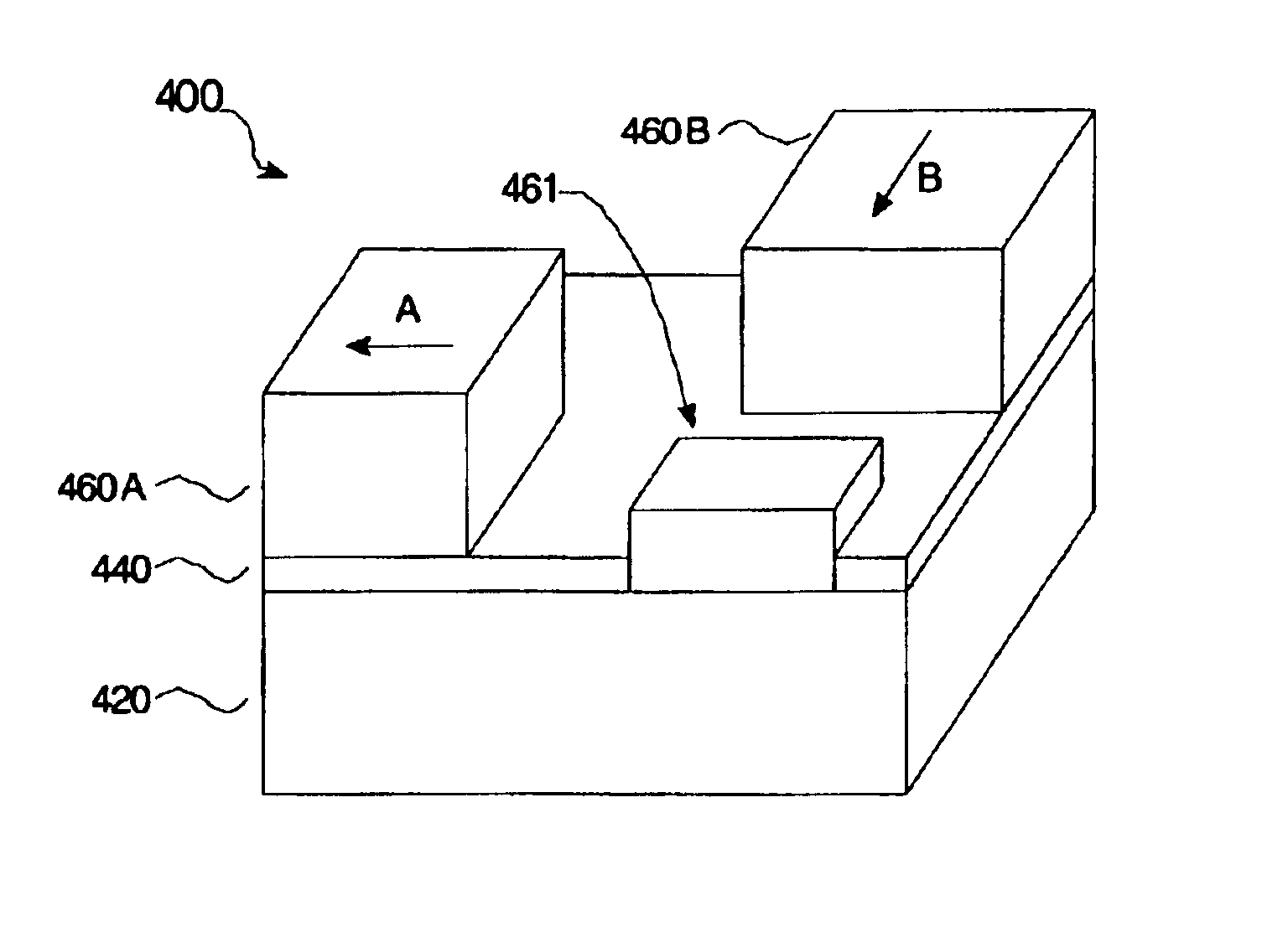
A photonic switch according to the present invention may be formed using one of a selected group of non-linear optical materials. Each of the materials within this group has a refractive index that demonstrates a substantial peak as a function of wavelength, where the peak occurs at a wavelength distinct from the wavelength of the input signal. A method of producing a photonic switch according to the present invention includes fabricating source and drain waveguides using micro-molding or micro-contact printing processes, or MIMIC (micro-molding in capillaries) of a UV-curable polymer. If desired, a gate waveguide may also be formed in part by these processes. The photonic switch also includes a photonic crystal formed from non-linear optical material, which may be formed, for example, using a block copolymer and nanoparticle composite in a MIMIC or μfluidics process. Such a process may employ a functionalized mold material to align the blocks.
Owner:SURFACE LOGIX INC
Preparation method of carbon-based titanium dioxide composite material of complex porous structure
InactiveCN101665679AHigh crystallinityHas a hierarchical fine structureOther chemical processesNon-linear opticsElectrical batteryCarbonization
The invention relates to a preparation method of carbon-based titanium dioxide composite material of complex porous structure. Based on the synthetic technology of biological templates, the method comprises the steps of using pollen as the material of biological templates, conducting ultrasonic pretreatment, then impregnating in titanium dioxide solution for impregnated optimization treatment, then carbonizing the mixture in air in advance and carbonating under nitrogen atmosphere. During the carbonated process, brookite phase titanium dioxide nanocrystal is produced in situ. Finally, a carbon-based brookite phase titanium dioxide composite material with complex porous structure is obtained. The composite material consists of brookite phase titanium dioxide crystal particles with the granularity less than 10nm and carbonized pollen matrix. The technology is simple, the cost is low, and the prepared carbon-based brookite phase titanium dioxide composite material with complex porous structure has favorable application prospect in the fields such as air-sensitive and pressure-sensitive elements, non-linear optical materials, solar cells, environment purification and the like.
Owner:SHANGHAI JIAO TONG UNIV
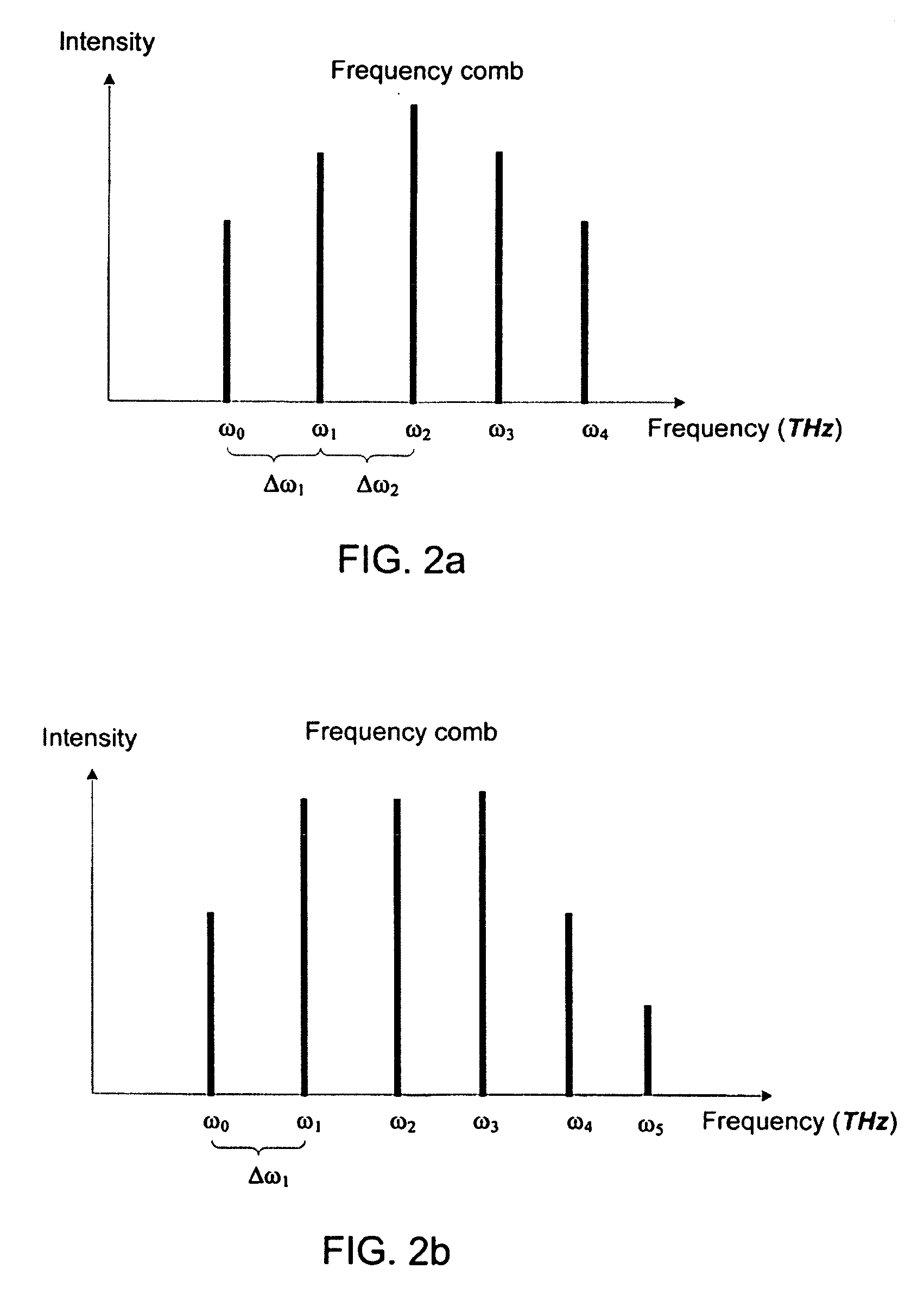
Speckle reduction in imaging applications and an optical system thereof
Speckle effect in imaging applications is reduced by generating additional speckle patterns on the screen such that the speckle patterns are overlapped and the overlapped speckle patterns average out on the screen to appear as a noise background to the viewers. The speckle patterns are generated by discrete optical signals of a visible frequency comb. A visible frequency comb having discrete optical signals is generated through modulation-instability processes, phase-conjugation processes, and Bragg-scattering processes using a non-linear optical material and a wavelength converter.
Owner:TEXAS INSTR INC
Conjugated ramification material of fluorine containing anthracene and pyrene at 9th position, preparation method and application
InactiveCN1686976AEasy to routeEasy injectionElectrical apparatusElectroluminescent light sourcesOrganic solar cellOrganic field-effect transistor
The present invention belongs to the field of photoelectric material, in the concrete, it is a 9-position anthracene and pyrene substituted fluorenic conjugated derivative material and its preparation method, and said material can be used in the field of organic electronics of organic / polymer electroluminescence material, organic integrated circuit, organic solar cell, organic field effect transistor, dye laser, organic non-linear optical material and fluorescent probe, etc. Said compound can utilize 9-position twice substitution structure to introduced the anthracene and pyrene into the high-effective luminescent conjugated fluorene system. Besides, said invention also provides several advantage of said conjugated derivative material.
Owner:FUDAN UNIV
V-shaped conjugated light-absorbing organic salt compound and its use
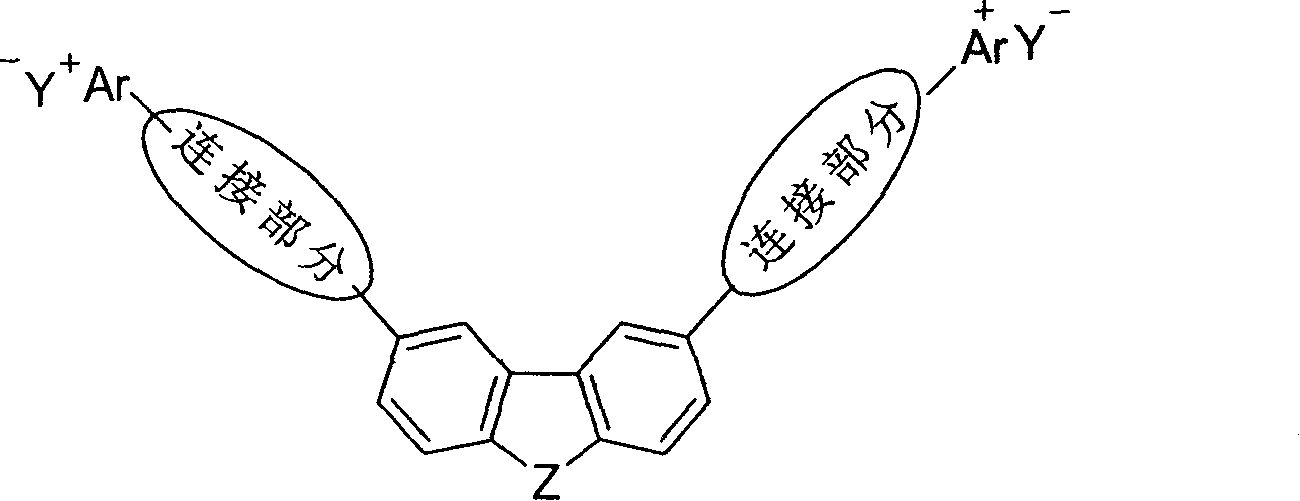
InactiveCN1887883AHigh reaction efficiencyNot easily affectedOrganic chemistryNon-linear opticsFluorescenceOrganic compound
The present invention belongs to the field of organic polymer chemistry and material chemistry, and is especially V-shaped conjugated light-absorbing organic salt compound and its use. The V-shaped conjugated light-absorbing organic salt compound is prepared with corresponding organic salt and heterocyclic dialdehyde, and through condensation or reaction with halogenated compound in V-shaped conjugation system. It has the structure as shown. The V-shaped conjugated light-absorbing organic salt compound may be as photsensitizer and / or initiator for single or double photon polymerization, as single or double photon bioluminescence probe with microM level use concentration, and as second order non-linear optical material; and its crystal may be used as electro-optical material and tera Hz beat frequency generator.
Owner:TECHNICAL INST OF PHYSICS & CHEMISTRY - CHINESE ACAD OF SCI
Blue-light emitting functional material and uses thereof
InactiveCN101250404AImprove performanceHigh color purityElectrical apparatusElectroluminescent light sourcesOligomerOrganic laser
The invention relates to a blue light emitting functional material and the application, which relates to star shooting shape oligomer blue light emitting functional material and the application. The material is monodisperse star shooting shape oligomer on the basis of minami carbazolyl element, the oligomer has the structure of a general formula 1, the material contains minami carbazolyl element, has monodisperse star shooting shape three-dimensional space structure, is easy to form stable amorphous state, and displays excellent filming property and light emitting property. The material has potential application in the fields such as organic electroluminescence, organic laser, organic non-linear optical material and the like. Particularly, organic electroluminescent devices which utilize the functional material to be blue luminescent material have improved luminescent brightness, device efficiency, color purity and spectroscopic stability.
Owner:NANJING UNIV OF POSTS & TELECOMM
Method for preparing TTF-TCNQ nano material
InactiveCN101100282AAchieve controlled growthEasy to makeNanostructure manufactureOrganic chemistryThermoelectric materialsAcetonitrile
The present invention discloses process of preparing nanometer TTF-TCNQ material. The process includes the following steps: 1. dispersing and dissolving TTF in normal hexane to form normal hexane solution of TTF; 2. dispersing and dissolving TCNQ in acetonitrile to obtain acetonitrile solution of TCNQ; and 3. adding the acetonitrile solution of TCNQ into normal hexane solution of TTF while stirring to obtain nanometer TTF-TCNQ material. The present invention synthesizes 1D nanometer TTF-TCNQ material as organic conductor, realizes the controllable growth of nanometer particle, and can obtain nanometer material with different controllable forms and structures. The preparation process is simple and high in yield, and the prepared nanometer material may be applied widely in sensor, field emission plane display, field effect device, non-linear optical material, piezoelectric material and other fields.
Owner:INST OF CHEM CHINESE ACAD OF SCI
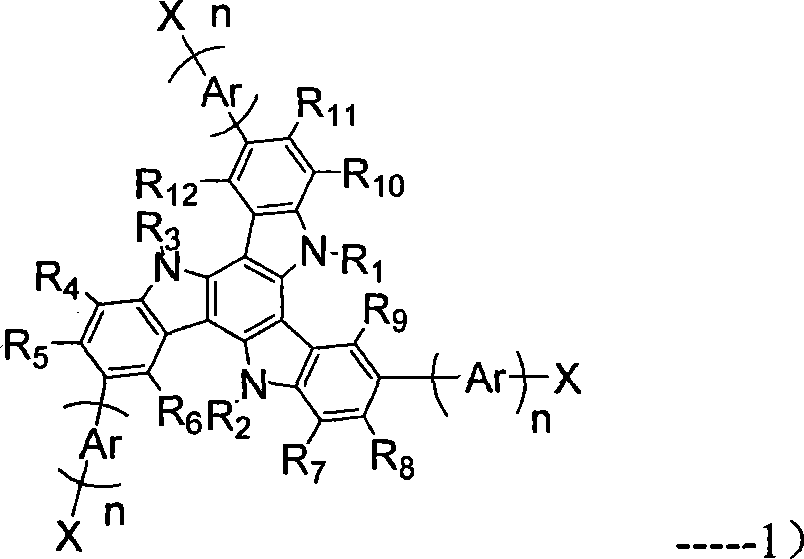
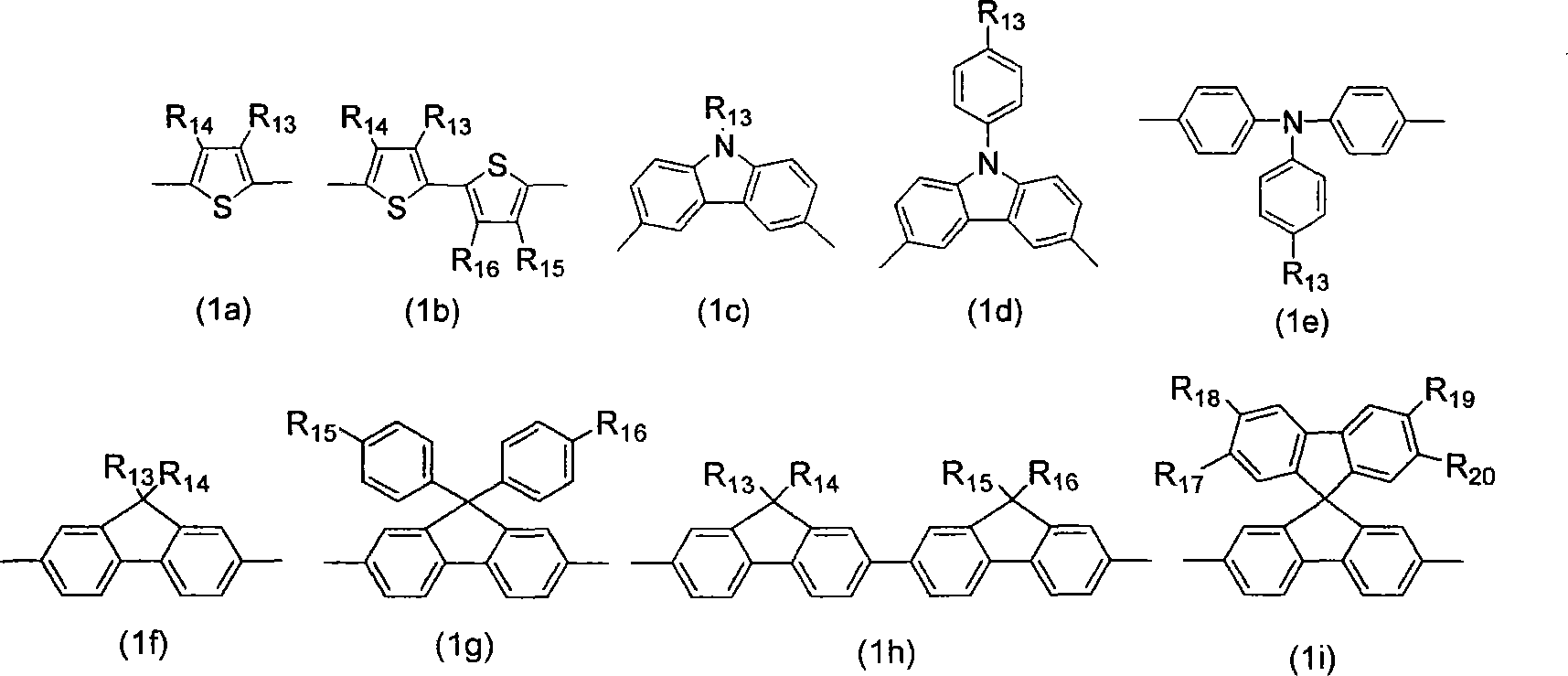
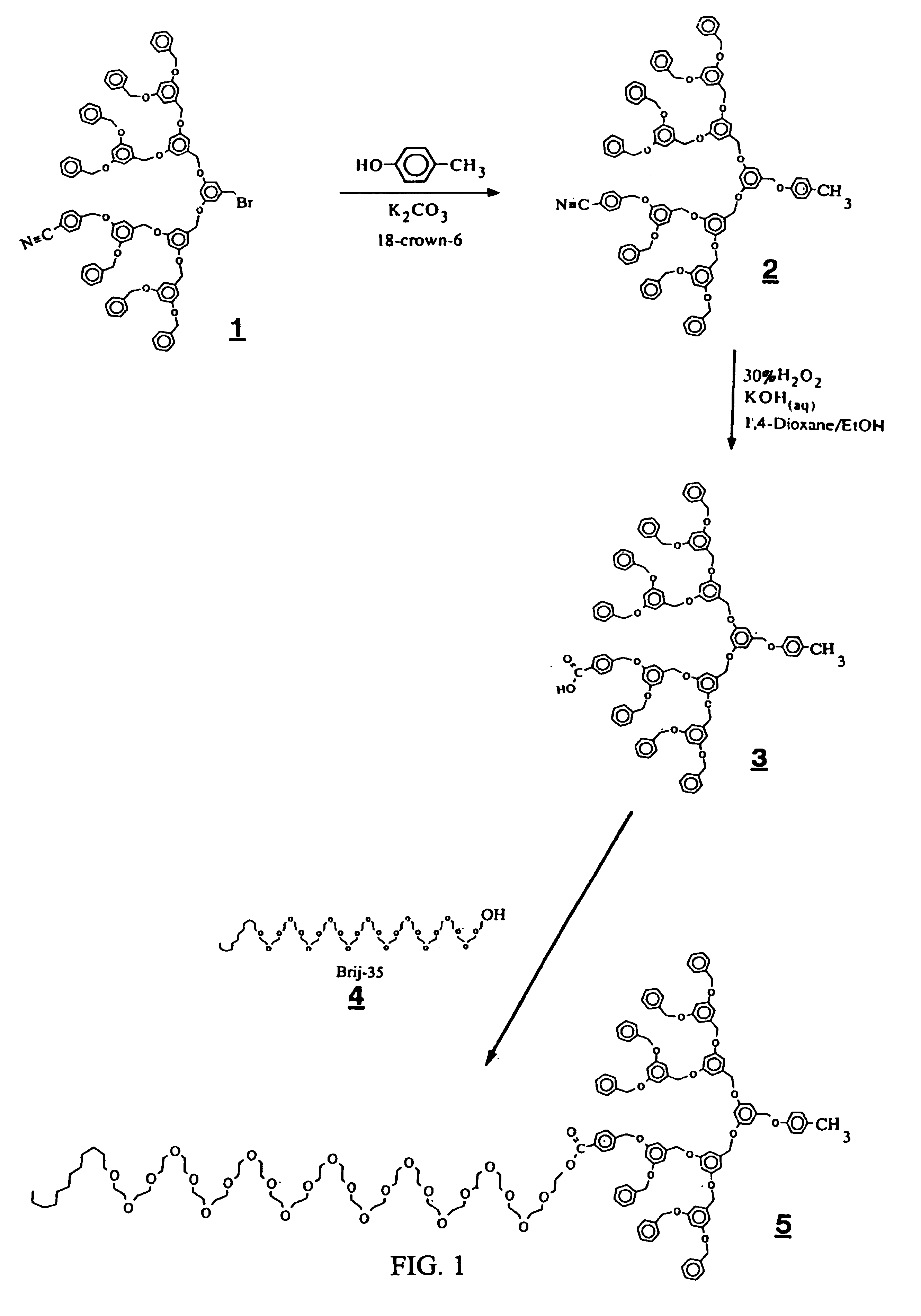
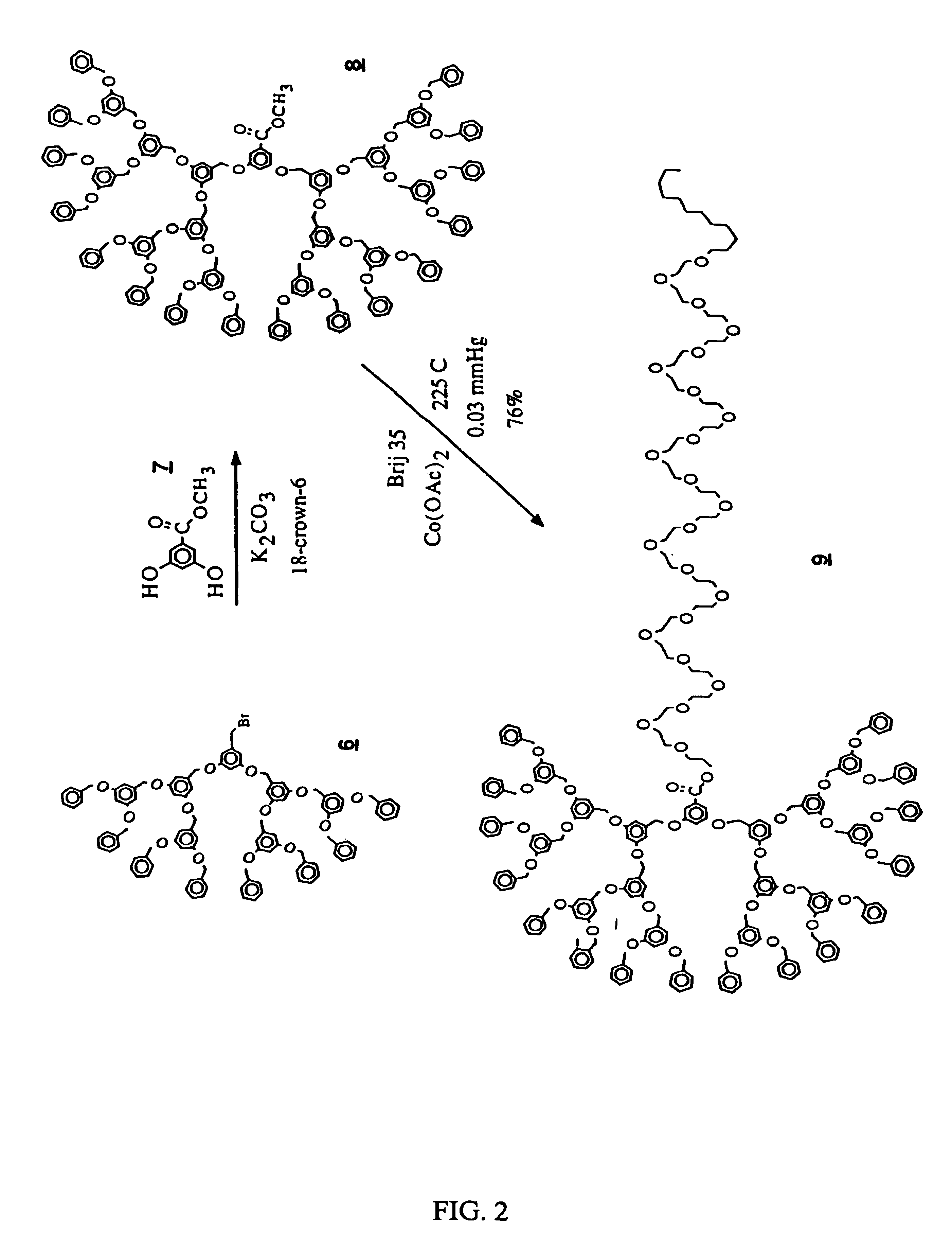
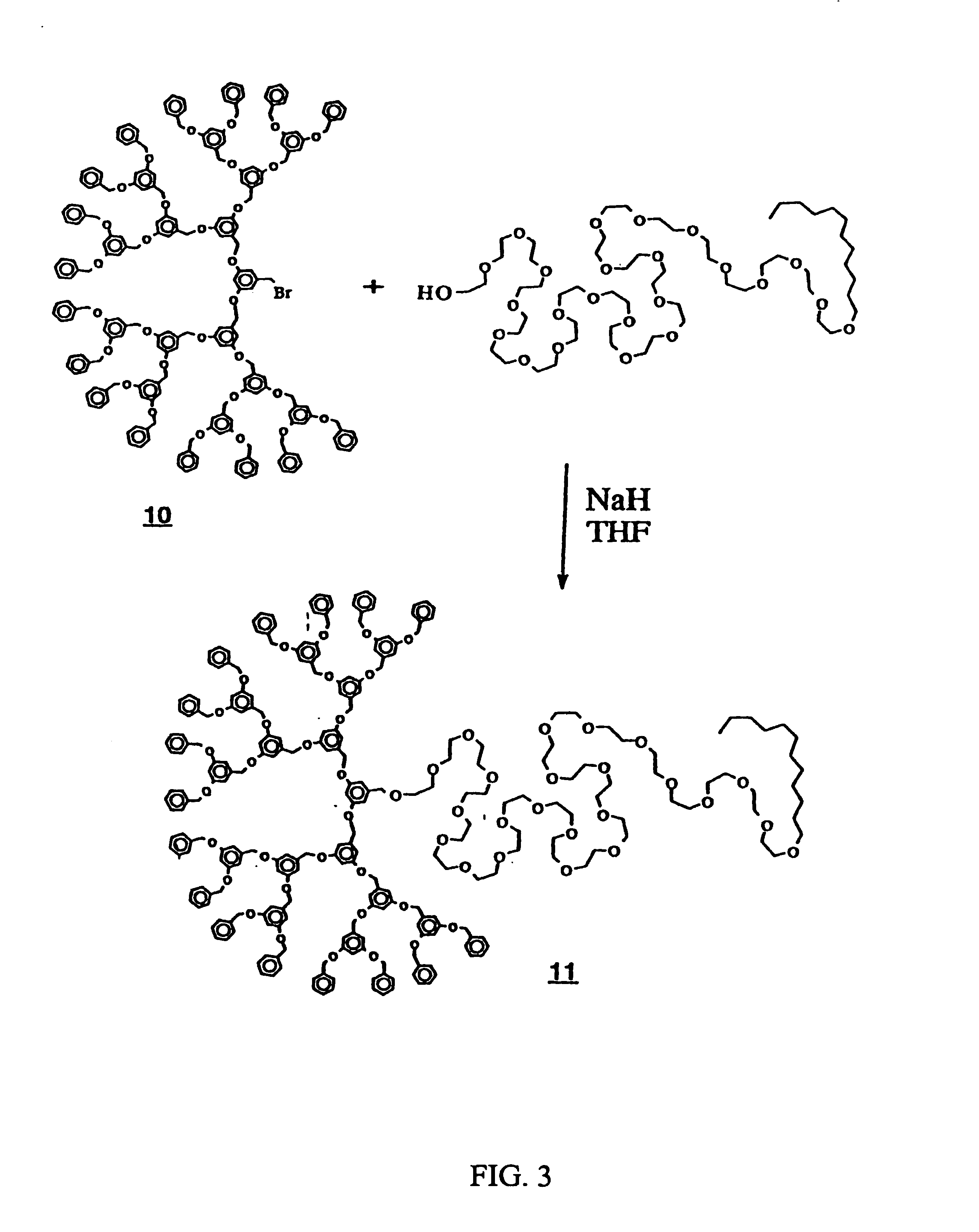
Dendritic based macromolecules
Macromolecules in the shape of three-dimensional kites, barbells, and other shapes are disclosed. The shaped macromolecules are prepared the reaction of one or more dendritic polymers and a linear polymer. The kite-shaped macromolecules are dendritic polymers attached at either their focal points or at an outer to a long carbon chain group. The barbell macromolecules have dendritic polymers at either end of the molecule, which polymers are connected together through the focal points of each by a suitable connecting substituent such as a linear polymer. The shaped macromolecules are used in specialty medical and technological applications including such as drug delivery agents, imaging materials, molecular devices, thin film devices, surface modifiers, transport agents, compatibilizers, rheology control agents, molecular ball bearings, molecular dipoles, non-linear optical materials, medical imaging agents, membrane and cell modifiers, complexing agents, adhesives, interface strengthening agents, and the like.
Owner:FRECHET JEAN M J +2
Preparation method of photocatalysis materials of biomorphic fine hiberarchy
InactiveCN101664675APromote crystallizationLow costPhysical/chemical process catalystsSolventSolar cell
The invention relates to a preparation method of photocatalysis materials of biomorphic fine hiberarchy. Nanometer titanium dioxide of biomorphic fine hiberarchy used as a photocatalysis material is obtained on the basis of the synthetic technology of biological templates by combining solvothermal technology, adopting butterfly wings with fine hiberachy as the material of the biological template and conducting solvothermal dipping treatment after pre-treating and activating the butterfly wings, as well as conducting ignition oxidation treatment. The preparation method is simple in process andlow in cost, and the obtained titanium dioxide nanometer material with biomorphic fine hiberarchy has excellent photocatalysis performance. The method can also be used for preparing air-sensitive andpressure-sensitive elements, non-linear optical materials, solar cells, environment purifying materials and the like, and the obtained materials have broad application prospect in the fields such as electronic and electrical industry, industries, environment protection, etc.
Owner:SHANGHAI JIAO TONG UNIV
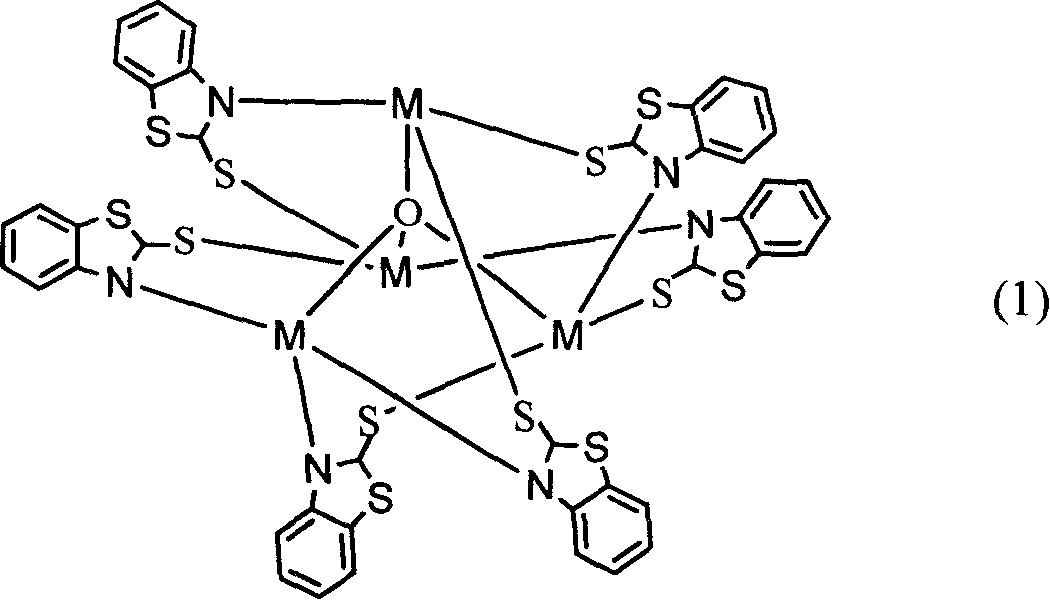
Process of preparing 2-thio benzothiazole transition metal cluster compound
A process for preparing the 2-mercapto benzothiazole cluster compound of transition metals features use of 'reflux method' with high output rate. Said cluster compound can be used for non-linear optical material and sulfurizing promoter.
Owner:QINGDAO UNIV OF SCI & TECH
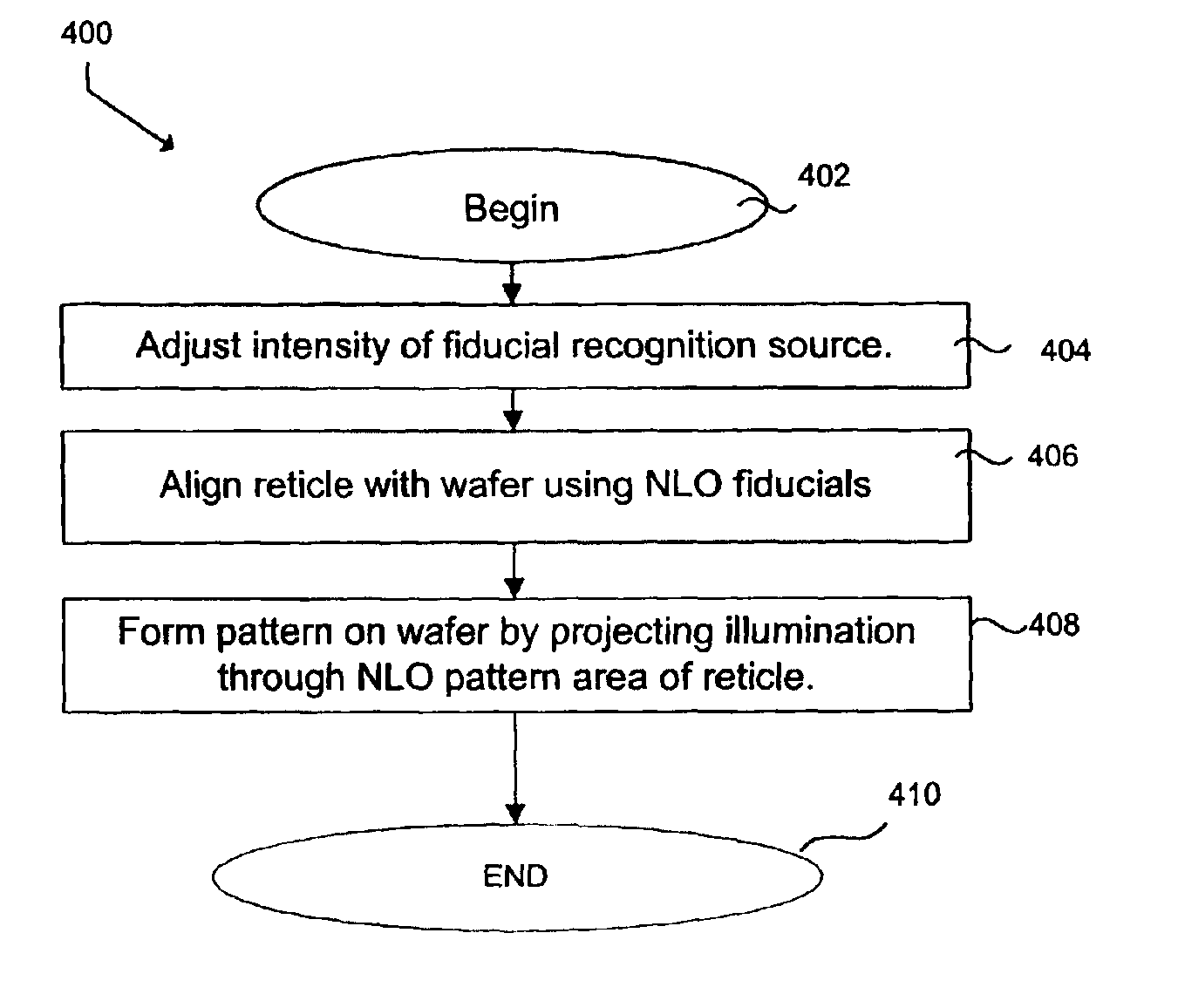
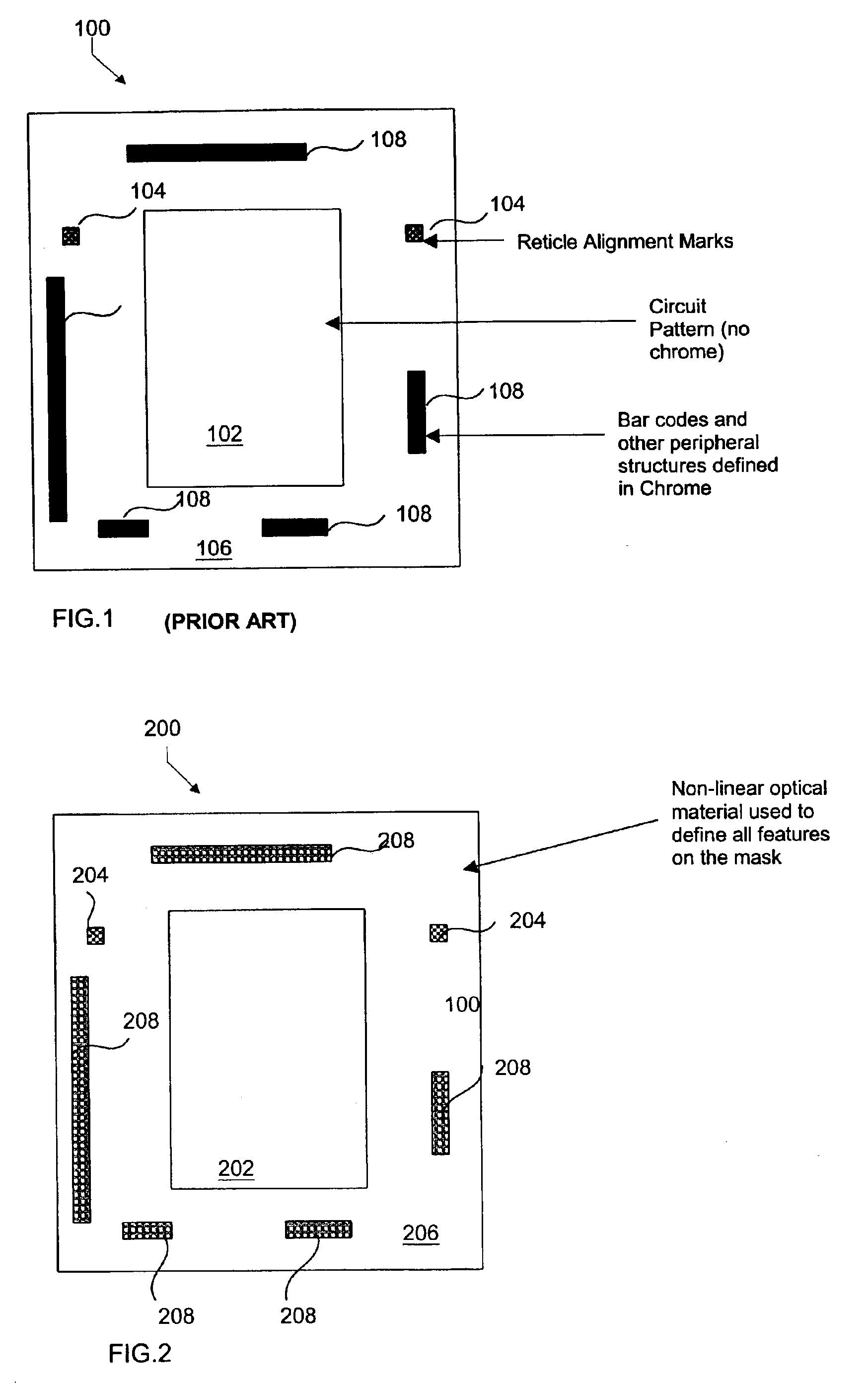
Chromeless phase shift mask using non-linear optical materials
InactiveUS6864020B1Avoid layeringSufficient opacityOriginals for photomechanical treatmentTransmittanceLength wave
An attenuated phase shift mask is formed using a non-linear optical material for both fiducial features and pattern features. The non-linear optical material selected has predetermined transmission at the actinic exposure wavelength and a smaller transmission at the fiducial recognition wavelengths.
Owner:BELL SEMICON LLC
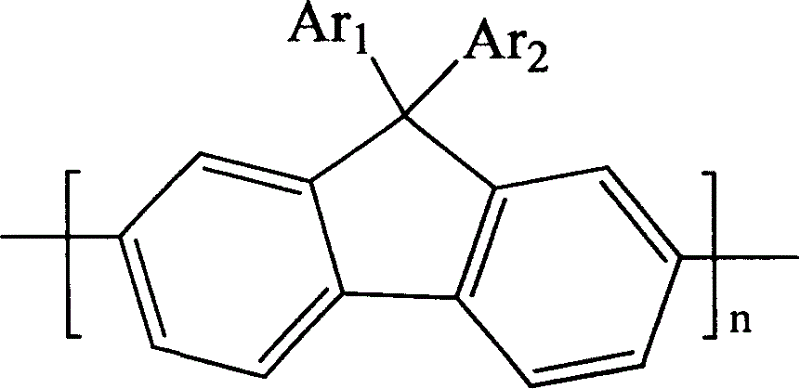
Spirocyclopentaphenanthrenefluorene-based compound and organoelectroluminescent device using the same
ActiveUS20050158583A1Solve low luminous efficiencyImprove thermal stabilitySolid-state devicesSemiconductor/solid-state device manufacturingSolubilityOrganic film
A spiro-based compound containing cyclopentaphenanthrene and fluorene, which is used for an organoelectroluminescent device and an organoelectroluminescent device having the spiro-based compound. The spiro-based compound containing cyclopentaphenanthrene and fluorene can be easily manufactured to have high solubility, high color purity, and good thermal stability. The spiro-based compound containing cyclopentaphenanthrene and fluorene is suitable as a substance for an organic film, in particular, an emission layer of an organoelectroluminescent device. Further, the spiro-based compound containing cyclopentaphenanthrene and fluorene can be used as an organic dye, or an electron material such as a non-linear optical material.
Owner:SAMSUNG DISPLAY CO LTD
Carbon nano tube/polymer/semiconductor nano particle composite material and preparation method thereof
InactiveCN102408644AEnsure Structural IntegrityStructural integrity is uniqueShieldingNano compositesOptical property
Owner:SHANGHAI NORMAL UNIVERSITY
Non-linear photonic switch
A photonic switch may be formed using one of a selected group of non-linear optical materials. Each of the materials within this group has a refractive index that demonstrates a substantial peak as a function of wavelength. The photonic switch includes a positive gain, and thus acts as a photonic transistor. In addition, a photonic switch is formed so that a gate signal is applied in a direction that is substantially perpendicular to the direction of the input signal so that there is no effective contamination of the input signal by the gate signal affecting the output signal.
Owner:SURFACE LOGIX INC
Nano silver shell/single dispersion SiO2 composite particle materia land its producing method and use
Owner:TECHNICAL INST OF PHYSICS & CHEMISTRY - CHINESE ACAD OF SCI
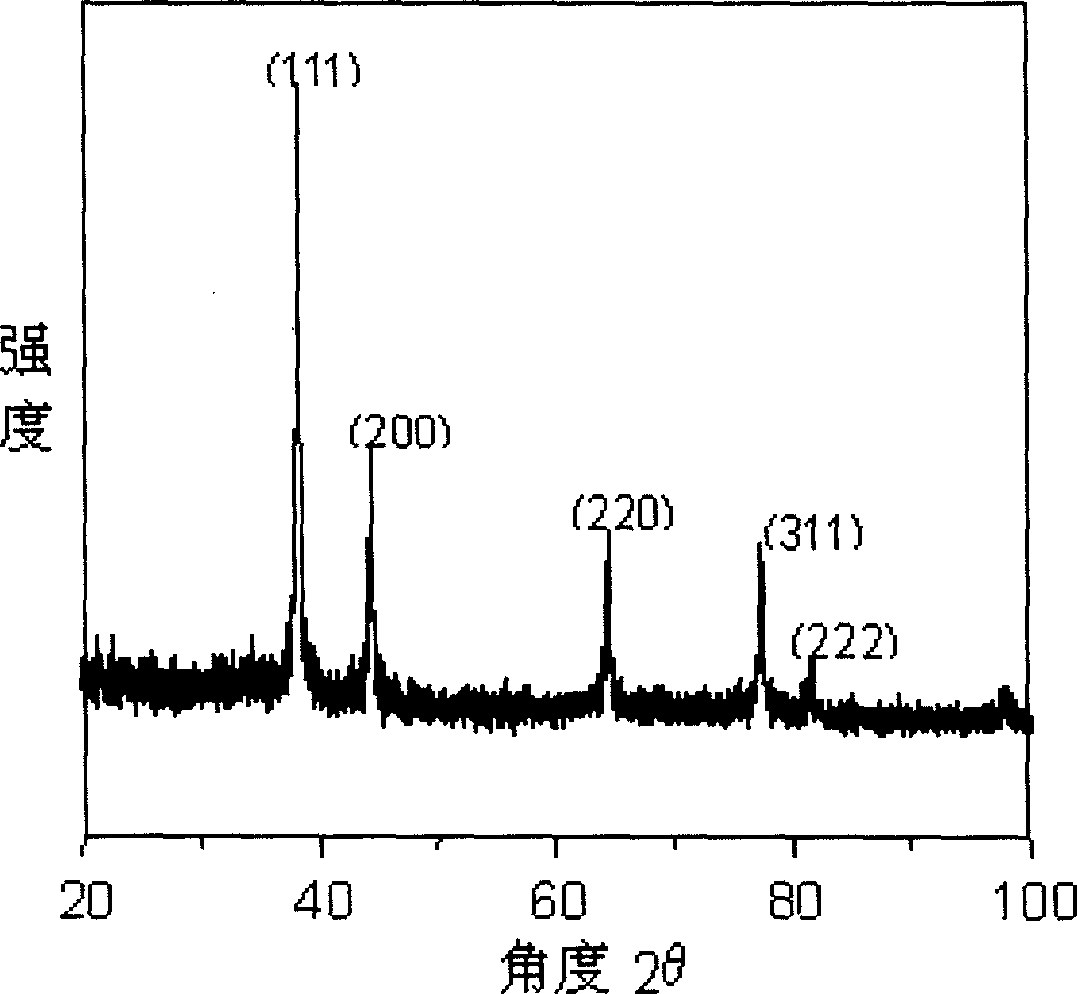
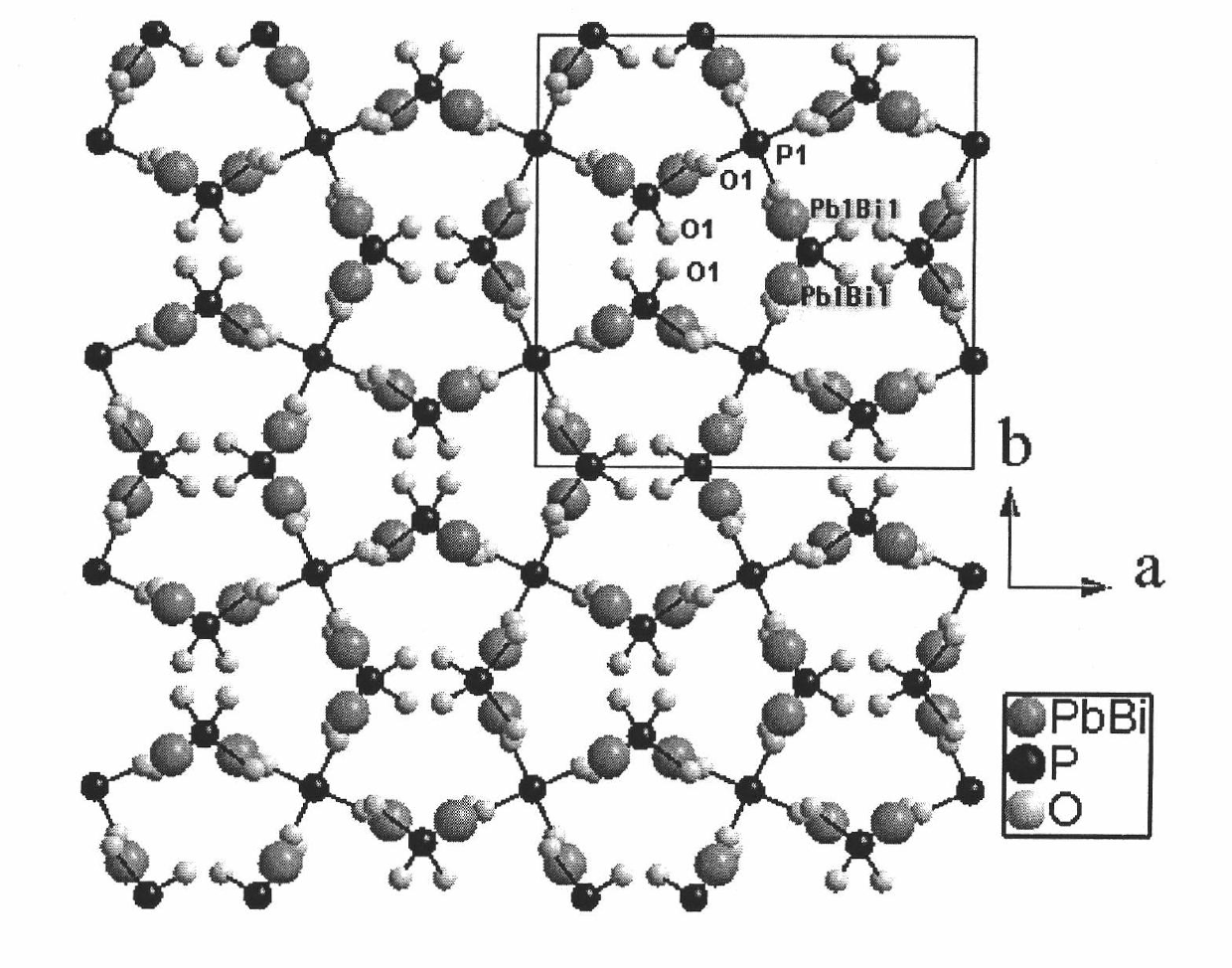
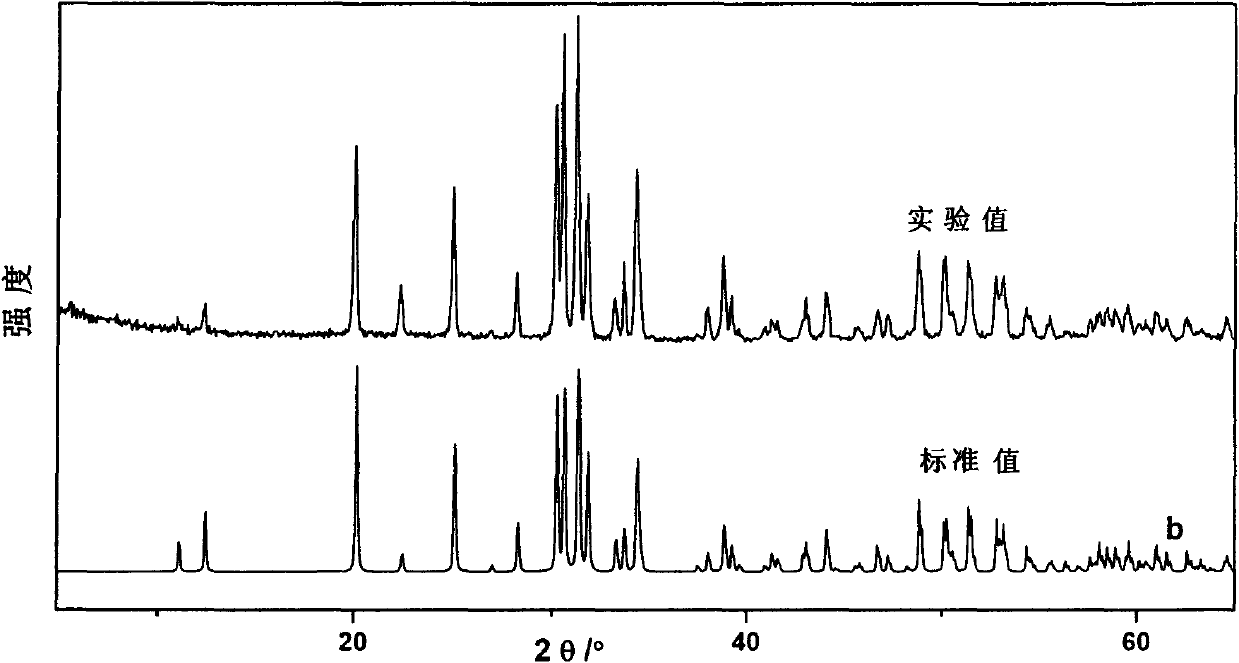
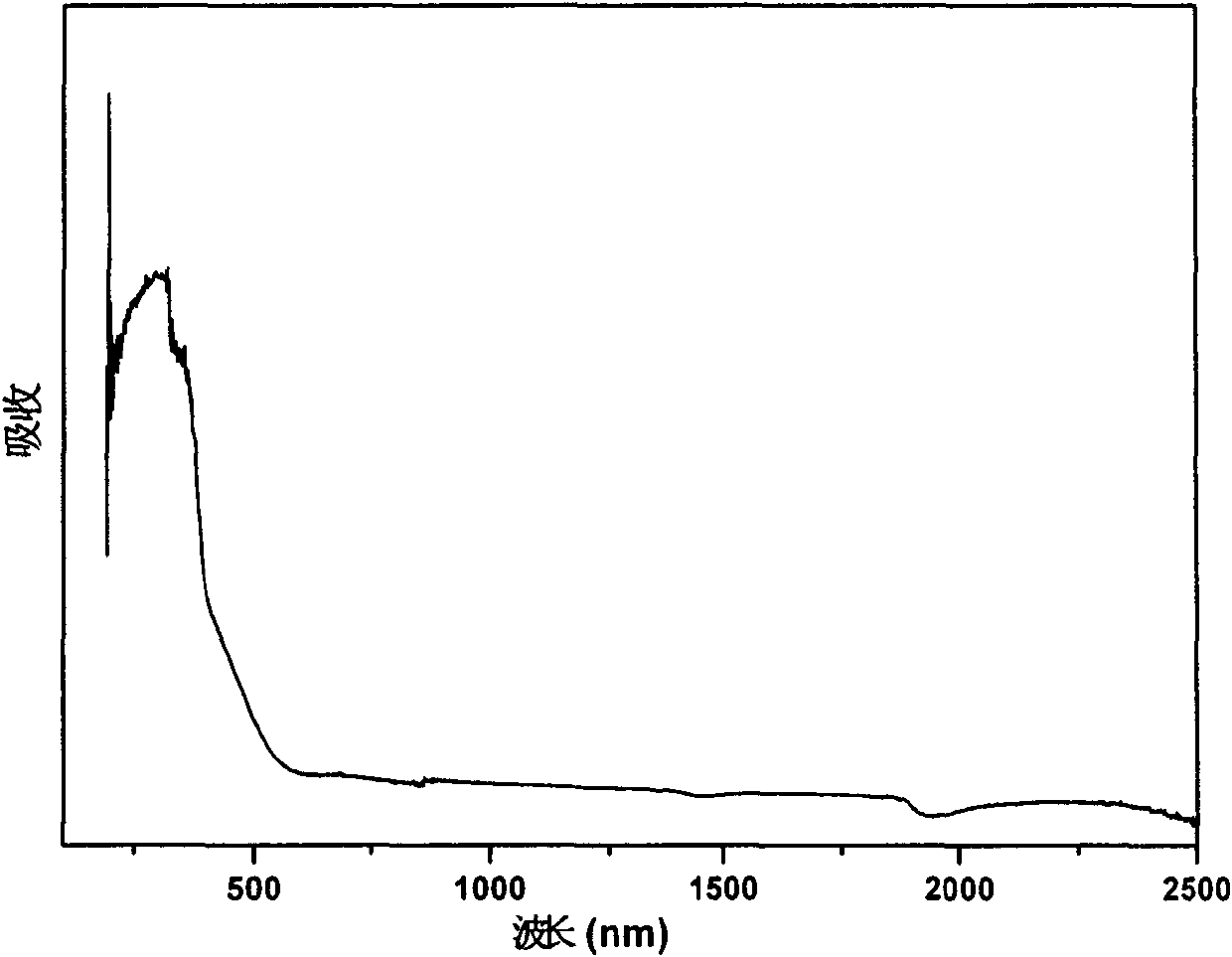
Non-linear optical material lead bismuth phosphate crystal
InactiveCN102086530ANonlinear Optical Performance ImprovementPolycrystalline material growthSingle crystal growth detailsSpace groupCell parameter
The invention discloses a non-linear optical material, namely a lead bismuth phosphate crystal, and relates to the field of non-linear optical materials. A chemical formula of lead bismuth phosphate is Pb3Bi (PO4)3; the molecular weight is 2230.98; the lead bismuth phosphate belongs to a cubic crystal system; the space group is I43d; and single-cell parameters, namely alpha, beta and gamma are 90.00 degrees, and Z is equal to 2. The lead bismuth phosphate crystal is prepared by adopting a high-temperature solid-state method. The lead bismuth phosphate has excellent non-linear optical performance; a powder secondary harmonic generation (SHG) coefficient of the lead bismuth phosphate is three times that of potassium dihydrogen phosphate (KDP); and the lead bismuth phosphate crystal can be used for deep water communication of submarines, laser blinding weapons, marine fish detection, optical disk recording, color laser printing, laser projection televisions, and optical computing or optical fiber communication.
Owner:FUJIAN INST OF RES ON THE STRUCTURE OF MATTER CHINESE ACAD OF SCI
Azobenzene polypeptide block copolymer and preparation method and application thereof
InactiveCN102093569ANew structureNon-linear opticsPolymer scienceAtom-transfer radical-polymerization
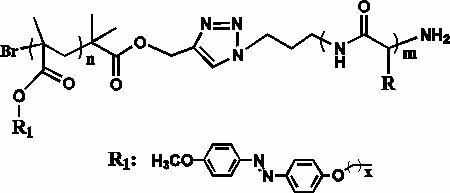
The invention discloses an azobenzene polypeptide block copolymer, a preparation method thereof and application thereof. One block of the azobenzene polypeptide block copolymer is an azobenzene radical-containing polymer and one block of the azobenzene polypeptide block copolymer is a polypeptide shown as the formula, wherein the number of methylene in an azobenzene radical is x which is equal to 2 to 9. The preparation method of the azobenzene polypeptide block copolymer comprises the following steps of: preparing the azobenzene radical-containing polymer, namely a homopolymer, by an atom transfer radical polymerization method; preparing the polypeptide by homopolymerizing an amino acid or an amino acid derivative compound; and connecting the azobenzene radical-containing polymer and the polypeptide through a click chemistry process to form the azobenzene polypeptide block copolymer. The method has the advantage that the azobenzene polypeptide block copolymer with a novel structure is prepared by the preparation method which is simple and feasible and in which the advantages of the click chemistry are fully used; and the azobenzene polypeptide block copolymer can be applied in the fields of non-linear optical materials and special photoresponse and storage devices and has wide application prospect.
Owner:EAST CHINA UNIV OF SCI & TECH +1
Color-changing polyurethane acrylic dyestuff resin and preparation method
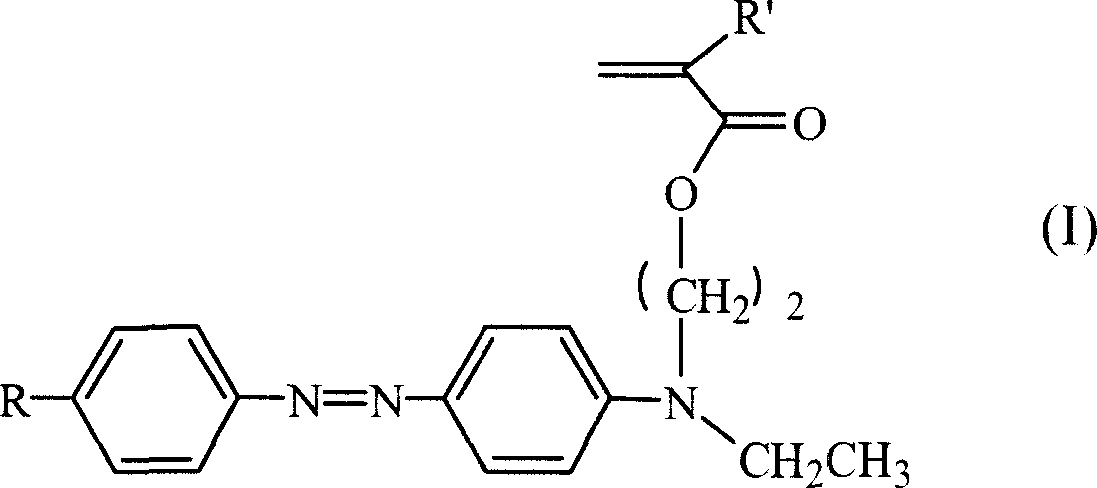
The invention relates to color-changing polyurethane acrylic dyestuff resin and a preparation method. According to the color-changing polyurethane acrylic dyestuff resin, a double-bond-containing polyurethane dyestuff prepolymer is prepared from macromolecular polyol, polybasic isocyanate, a dyestuff and a hydroxyl acrylate monomer through step-by-step polymerization; then an initiator and an acrylate monomer are added and are subjected to solution polymerization to prepare the color-changing polyurethane acrylic dyestuff resin. The color-changing dyestuff is very stable and has a bright color; the obtained polyurethane acrylic dyestuff resin has an obvious photochromic property; the defects in the aspect of application of a small molecular azo dyestuff and limitation to an application field are overcome; the dyestuff is joined in a polymer molecular chain through a chemical bonding manner and the advantages of the dyestuff of friction resistance, migration resistance, staining resistance, gloss retention, safety in utilization and the like are obviously improved. The synthesized polyurethane acrylic dyestuff resin has excellent performance of polyurethane and acrylate. The color-changing polyurethane acrylic dyestuff resin has excellent thermal stability and processing performance, and has a wide application prospect in the fields of advanced materials including information storage and protection materials, liquid crystal displays, non-linear optical materials and the like.
Owner:安徽科翌新材料有限公司
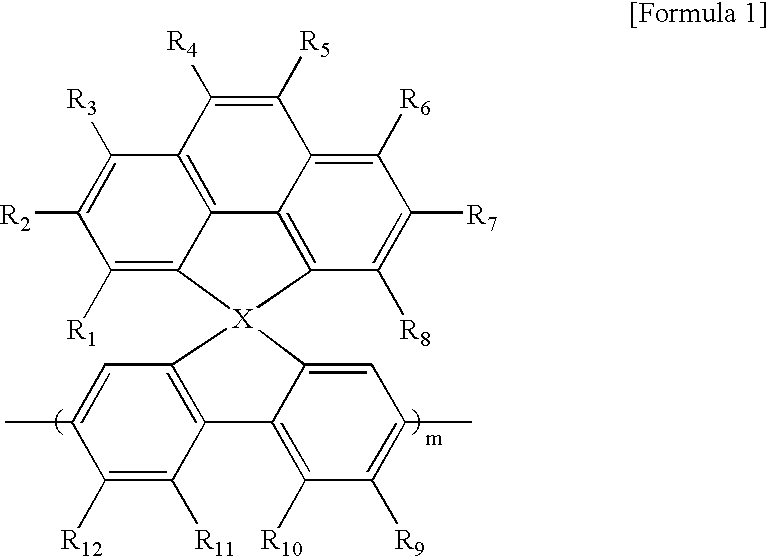
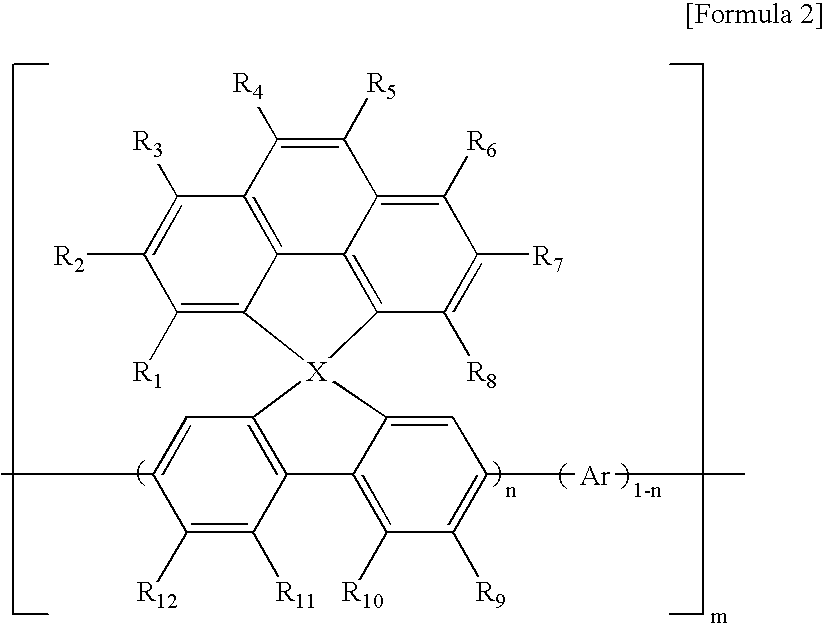
Nonlinear optical material with nano-sized dendrimer structure
The present invention relates to a non-linear optical material having a dendrimer structure; and, more particularly, to a non-linear optical material having organic chromophores at the ends and formed based on ester linkages and / or ether linkages. Since the non-linear optical material of the present invention is formed based on ester linkages and / or ether linkages, it is very stable. Also, because it is a dendrimer structure, it has the properties of a polymer while having a strong connection ability at the ends, and this makes the non-linear optical material easily adopt organic chromophores easily. As it is stable thermally and optically, it can be applied to optical communication usefully.
Owner:ELECTRONICS & TELECOMM RES INST
Furaldehyde-triphenylamine Schiff base and furaldehyde-triphenylamine poly Schiff base as well as preparation methods thereof
InactiveCN102212049APotential adjustableImprove adhesionOrganic chemistryElectrolysis componentsFuraldehydeTriphenyl diamine
The invention provides a furaldehyde-triphenylamine Schiff base and a furaldehyde-triphenylamine poly Schiff base as well as preparation methods thereof and relates to a Schiff base and a poly Schiff base as well as preparation methods thereof, wherein the Schiff base, poly Schiff base and method provided by the invention are used for solving the problems that in the furaldehyde-triphenylamine poly Schiff base obtained by a chemical oxidation method in the prior art, the Schiff base monomer is dissoluble, the poly Schiff base polymer is difficultly dissolved, and coating film processing is difficultly achieved. The molecular formulas of the two types of Schiff bases produced in the invention are shown in the specification, and the structural general formulas of the two types of Schiff bases are shown in the specification. In the preparation method, an electrochemistry polymerization method is adopted. The method comprises the following specific steps: 1, synthesis of monomers; and 2, synthesis of a polymer. The furaldehyde-triphenylamine poly Schiff base produced by the method can be used as a PH sensing material, an air-sensitive material, an electrochromism material, a hole transportation material, a non-linear optical material and other photoelectric materials as well as an anticorrosive material.
Owner:HEILONGJIANG UNIV
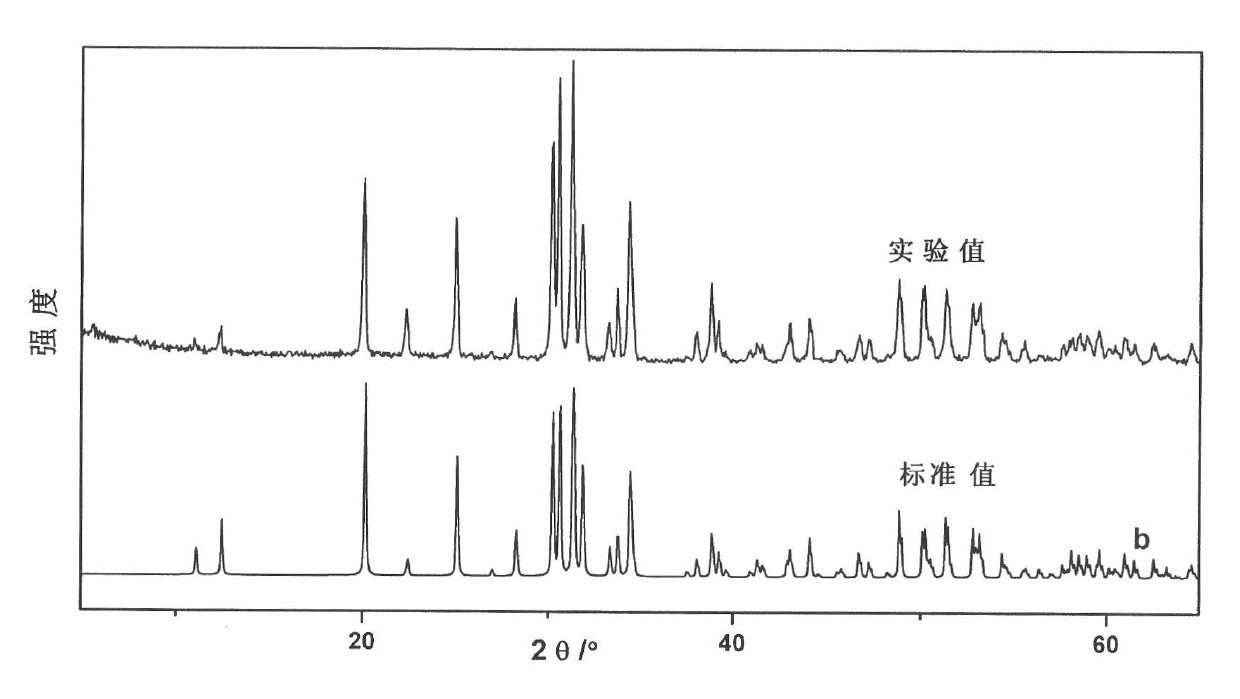
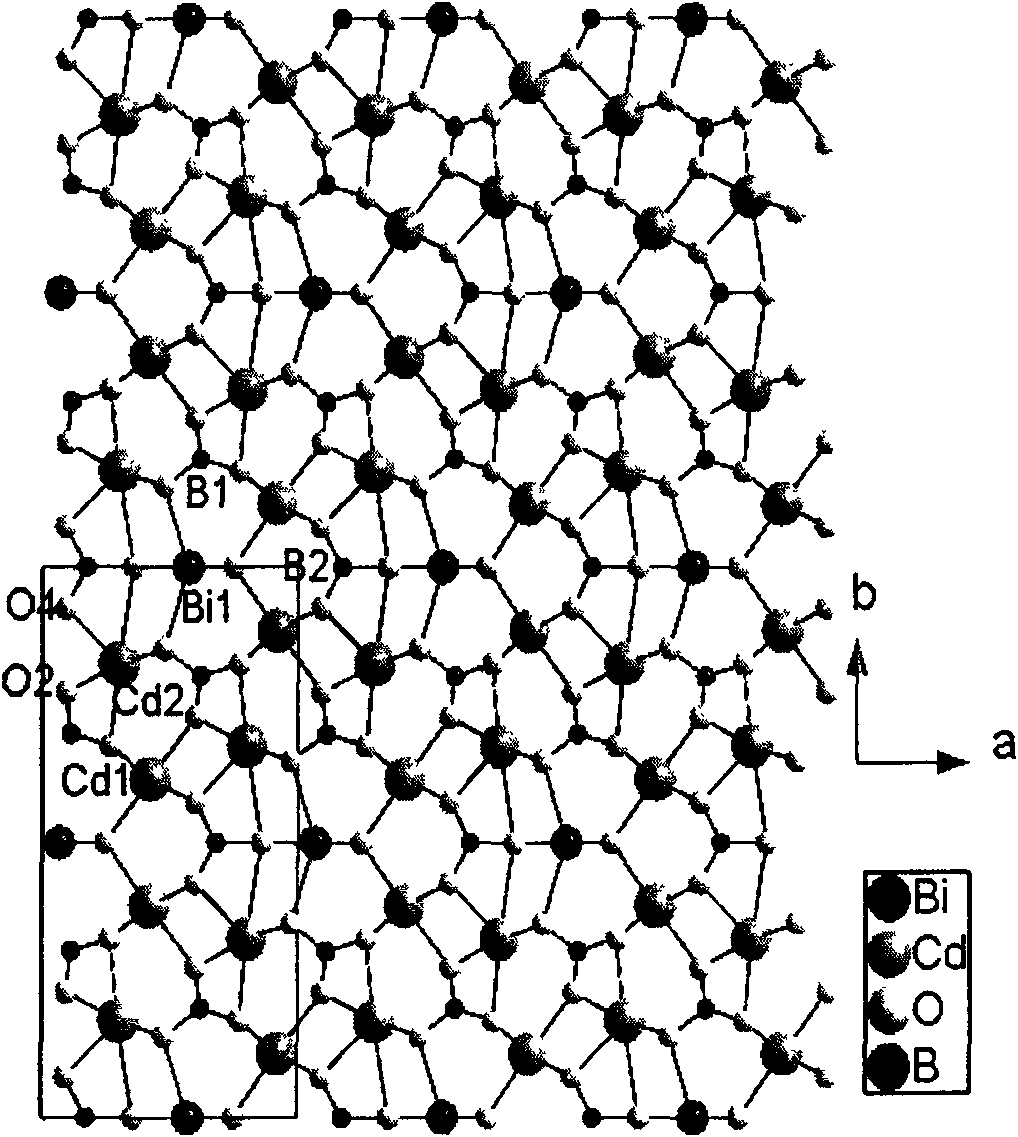
Non-linear optical crystal bismuth cadmium borate
ActiveCN102031562ANonlinear Optical Performance ImprovementPolycrystalline material growthFrom solid stateNonlinear optical crystalPhosphate
The invention provides an optical crystal bismuth cadmium borate compounded by three chromophores, and relates to the field of non-linear optical materials. The chemical formula of the bismuth cadmium borate is Cd3BiO(BO3)3, and the molecular weight is 851.05; the bismuth cadmium borate belongs to a monoclinic system, and the space group is Cm; the unit cell parameters are that: a is 8.044(2), b is 15.913(5), c is 3.4891(10) angstrom, alpha is 90.00 degrees, beta is 100.08(2) degrees, gamma is 90.00 degrees, V is 439.7(2) angstrom<3>, and Z is 2. The non-linear optical crystal bismuth cadmium borate is prepared by a high-temperature solid state method. The bismuth cadmium borate has excellent non-linear optical performance, and the second harmonic generation (SHG) coefficient of the bismuth cadmium borate powder is six times that of potassium dihydrogen phosphate (KDP).
Owner:FUJIAN INST OF RES ON THE STRUCTURE OF MATTER CHINESE ACAD OF SCI
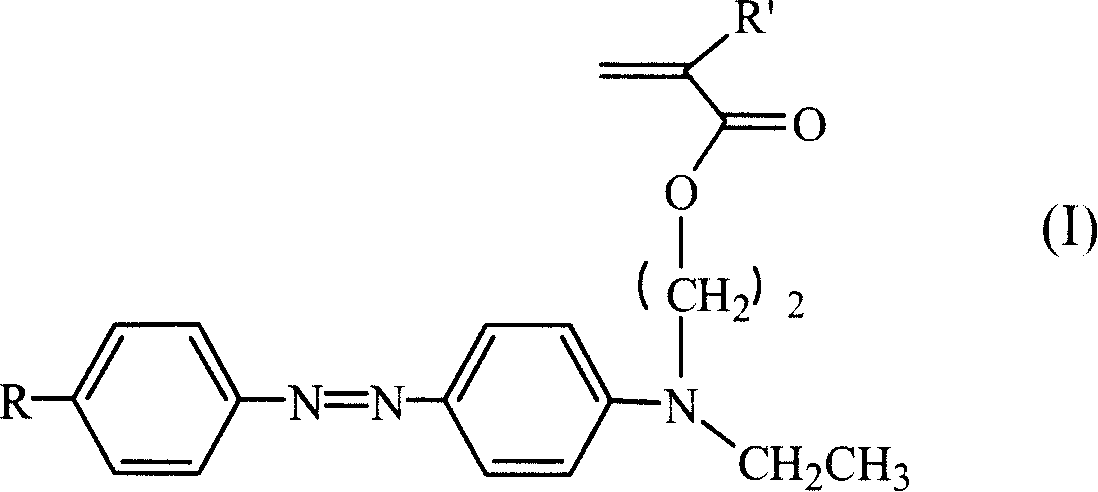
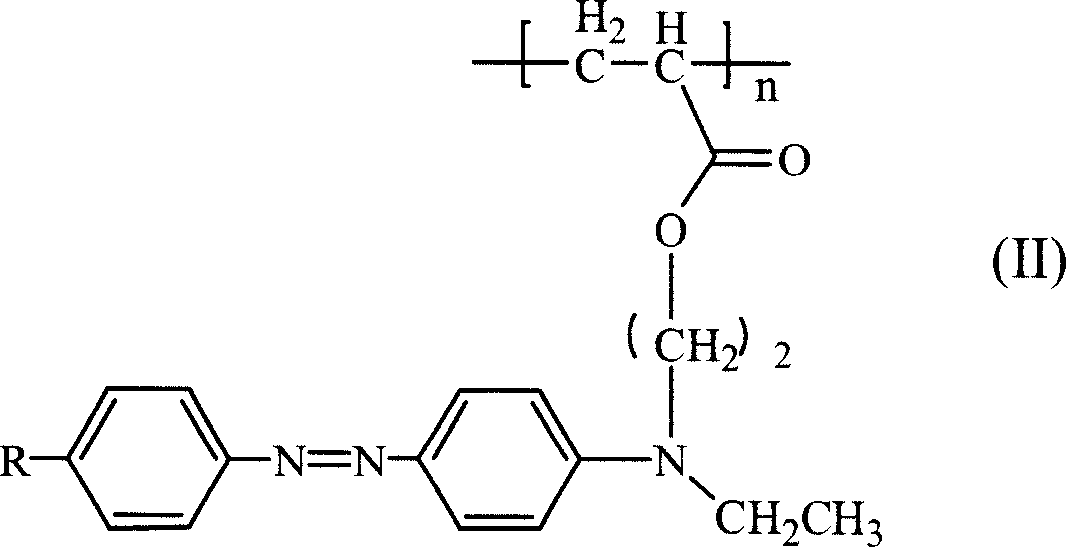
Third order non-linear optical material monomer, its polymer and production
ActiveCN1975552AImprove solubilityGood film formingNon-linear opticsSide chainAtom-transfer radical-polymerization
This invention discloses a kind of the azo monomer has intramolecularly push-and-pull electron structure and its polymer three order nonlinear optics material, after azo-coupler reaction of substituent aniline and N, N- ethyl hydroxyl ethyl aniline gaining the azo midbody, and reacts with (methyl) chlorine chloride then gains the crylic acid ester monomer with the functionality azo base group, and using the atom free radical polymerization (ATRP) method get the azo side chain type polymer whose molecule chain forms the push-and-pull electron structure. The polymer has good three order nonlinear optics functions and fairly good film function.
Owner:SUZHOU UNIV
Method for preparing nonlinear optical material of linkage nano carbon tubes of polythiophene methylene
InactiveCN1687251AImprove solubilityGood film formingPigment treatment with macromolecular organic compoundsOptical elementsSolubilityCarbon nanotube
The present invention discloses a preparation method of polythiophene methylene bonded carbon nano tube non-linear optical material. Said method includes the following processes: dissolving 3-octyl thiophene and 4-aminobenzaldehyde in dioxane, adding concetnrated sulfuric acid catalyst, under the protection of nitrogen gas synthesizing poly[(3-octyl thiophene)-2,5-di(4-aminobenzane)]; dissolving said compound in dioxane, adding tetrachlorobenzoquinone to obtain poly[(3-octyl thiophene)-2,5-di(4-aminobenzene)], then dissolving said compound in dioxane, and adding multiwall or single-wall carbon nano tube containing acyl chloride group so as to obtain the polythiophene methylene bonded carbon nano tube.
Owner:TIANJIN UNIV
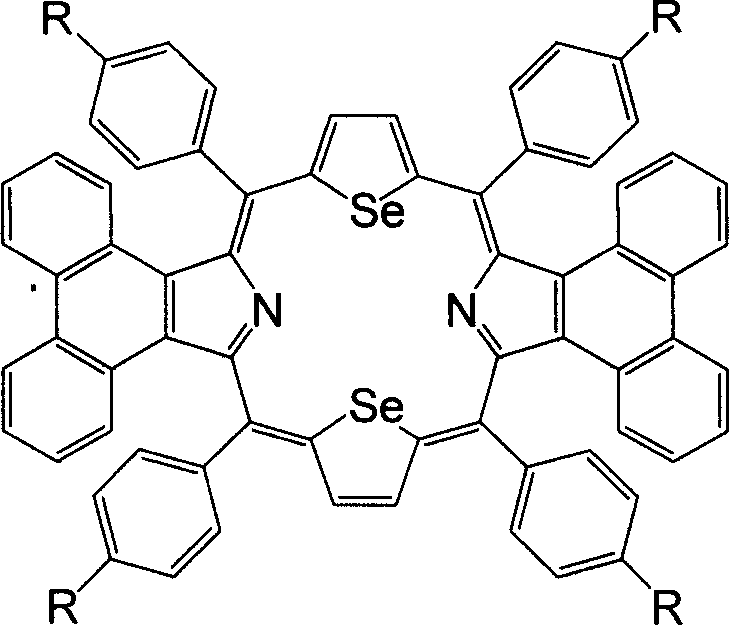
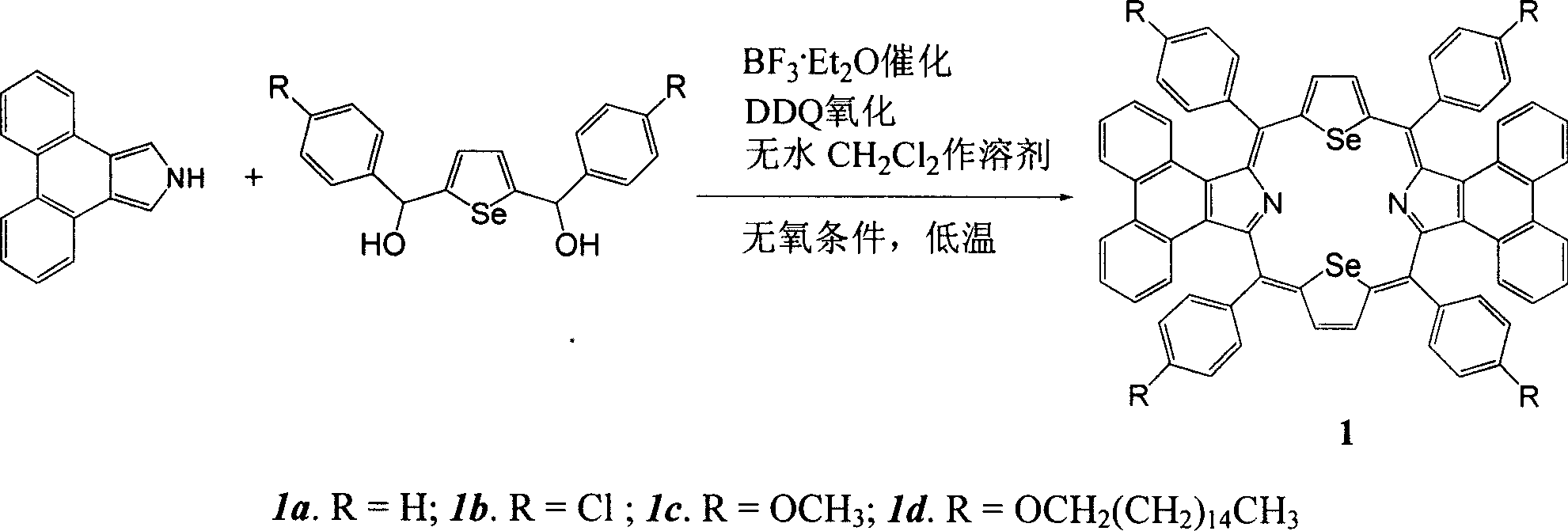
Synthesis of middle position-tetraaryldiphenanthrene diselenoporphyrin derivative and application thereof
InactiveCN101210024AImprove absorption efficiencyImprove conversion efficiencyOrganic chemistryLuminescent compositionsMolecular identificationOrganic solar cell
The invention discloses a 5,10,15,20-tetra-aryl diphenanthrene (9,10-b:9,10-1)-22,24-diseleno porphyrin compound which is obtained by the reaction of phenanthro-pyrrole and 2,5-di(aryl hydroxyl methyl) selenophen under low temperature and catalysis of BF3 Et2O in anaerobic atmosphere. The Soret spectral bands of the compound appear in 521nm, compared with tetraphenyl diseleno porphyrin without conjugated aromatic ring at beta-site, the Soret spectral bands are red shifted by 52nm and enter a green light zone above 500nm. The compound has wide application prospects in such areas as photodynamics therapy photosensitizer, OFETs (organic field effect tubes), molecular antenna, light energy converter, optical conversion material, molecular switch, molecular logic gate, molecular wire, organic solar battery, organic electroluminescence, non-linear optical material, optical storage, molecular identification and medicine and so on.
Owner:NANJING UNIV
Waveguide type optical device using large 3rd non-linear optical material and method for operating the same
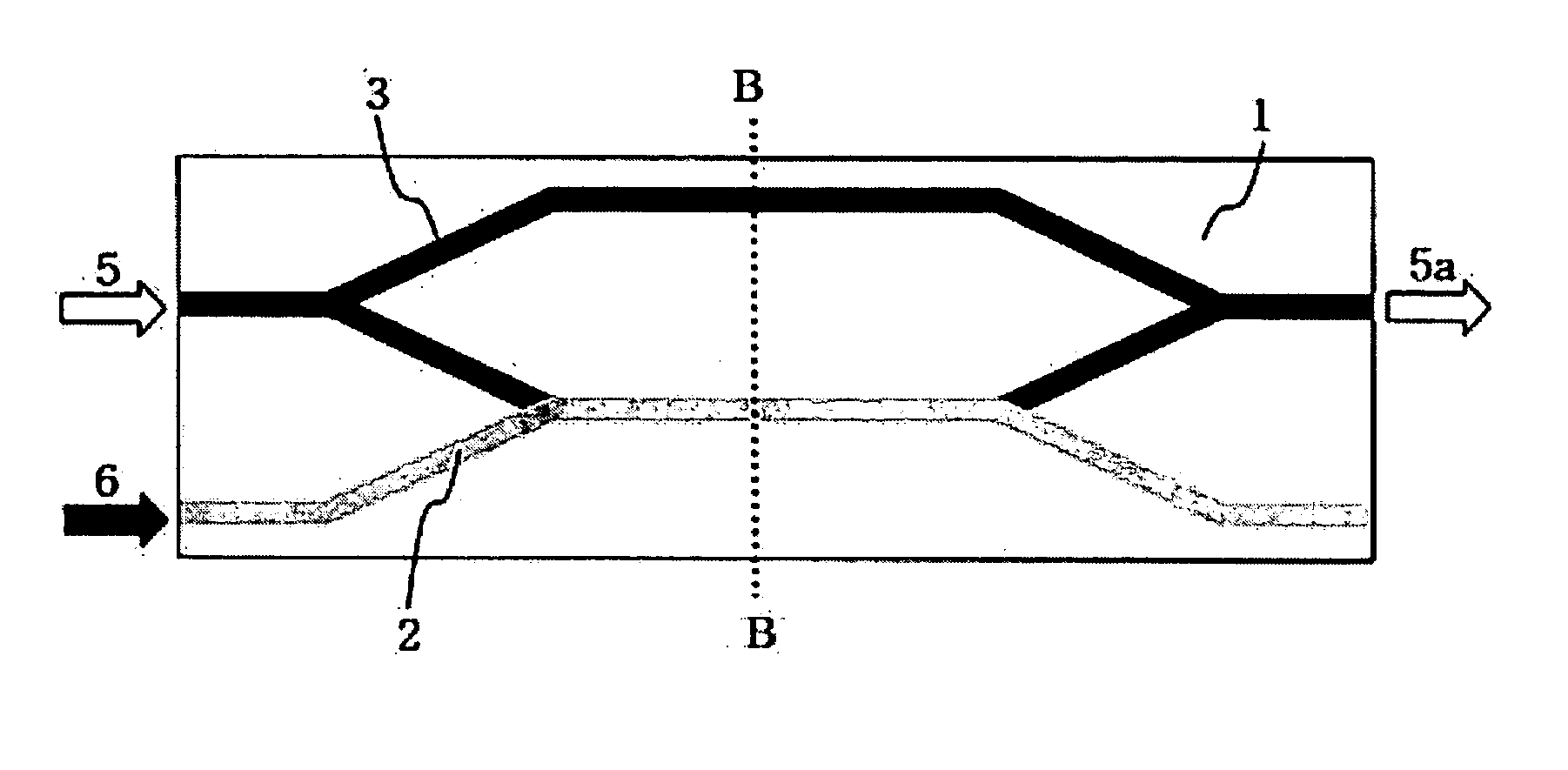
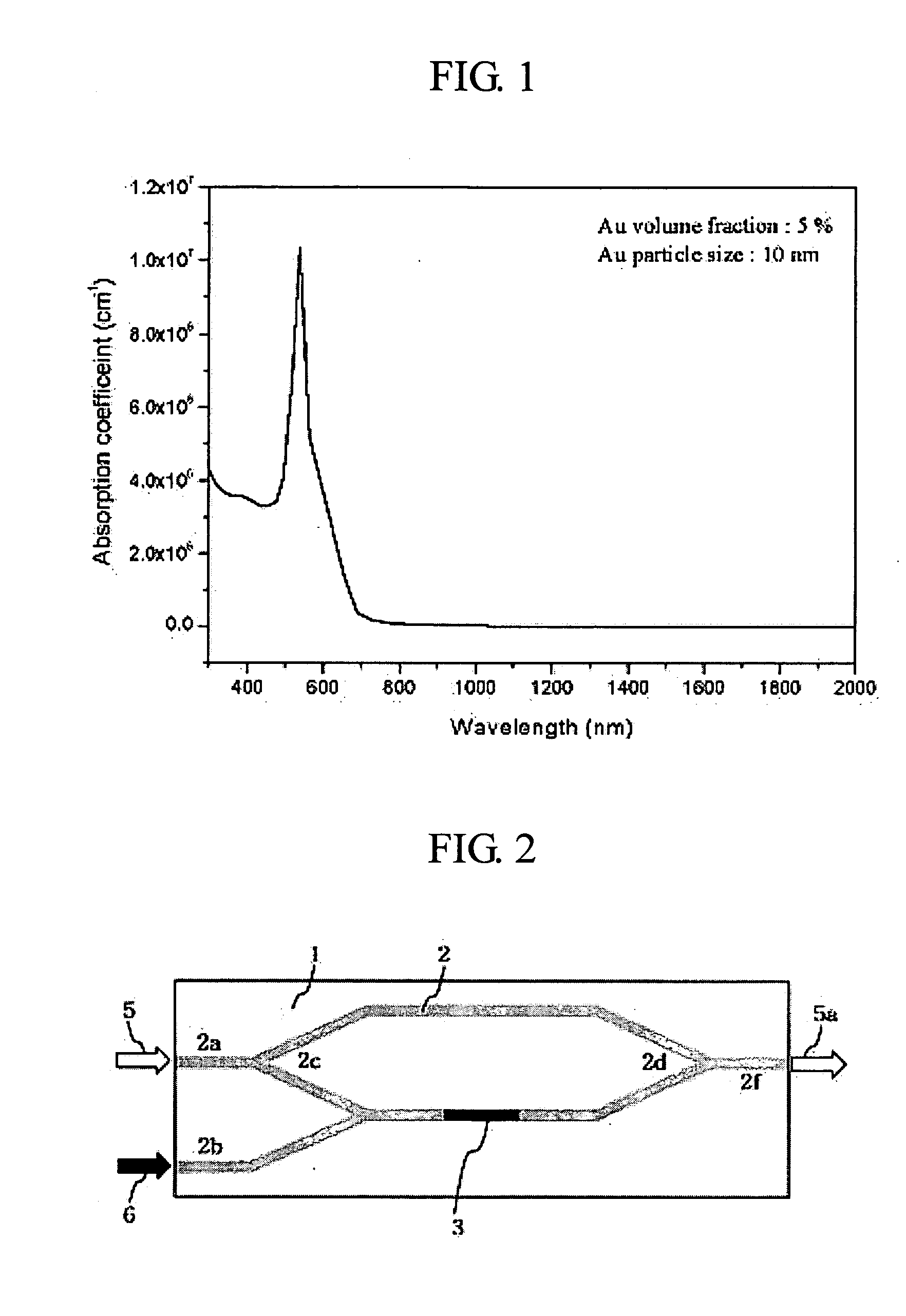
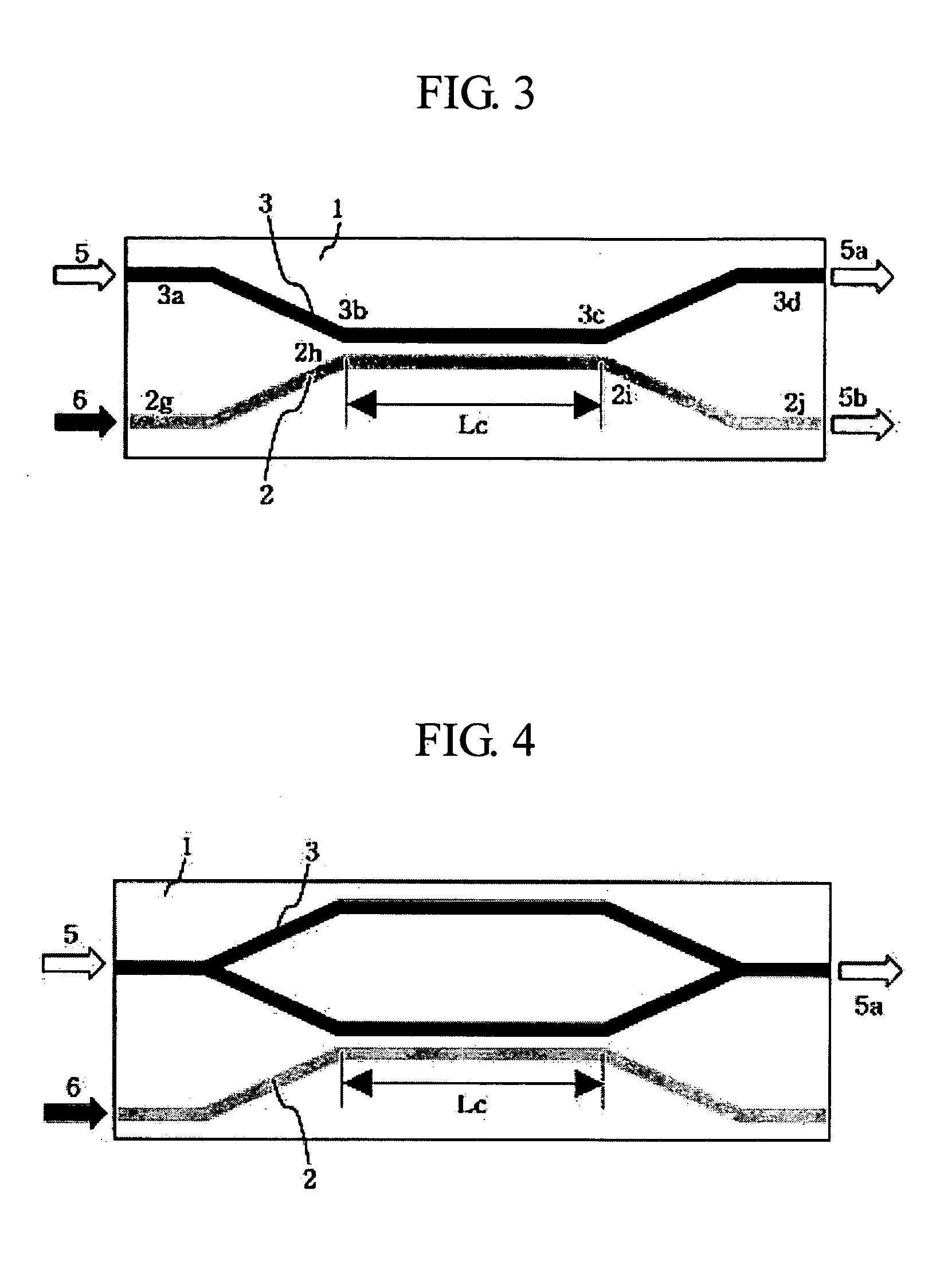
ActiveUS20050105864A1Easy to operateEasy to makeCoupling light guidesOptical waveguide light guideNonlinear phenomenaRefractive index
Disclosed is a waveguide type optical device utilizing a nonlinear refractive index change according to a large 3rd order nonlinear optical phenomenon. The waveguide type optical device includes a signal beam waveguide through which a signal beam propagates; and a pump beam waveguide through which a pump beam propagates, wherein the pump beam waveguide is disposed adjacent to the signal beam waveguide so that the pump beam can be coupled to the signal beam waveguide, the signal beam waveguide is made of nonlinear optical materials with large 3rd order nonlinear optical property and the pump beam waveguide is made of linear optical materials, and the wavelength range of the signal beam is different from that of the pump beam. By such a structure, the pump beam is coupled to one arm of the signal beam waveguide, thereby generating a 3rd nonlinear phenomenon on one arm of the waveguide through which the signal beam passes. Therefore, the waveguide optical device which implements an all-optical communication device operating as an optical device and can be integrated is provided.
Owner:KOREA INST OF SCI & TECH
Integrated biplane optical sensing core chip
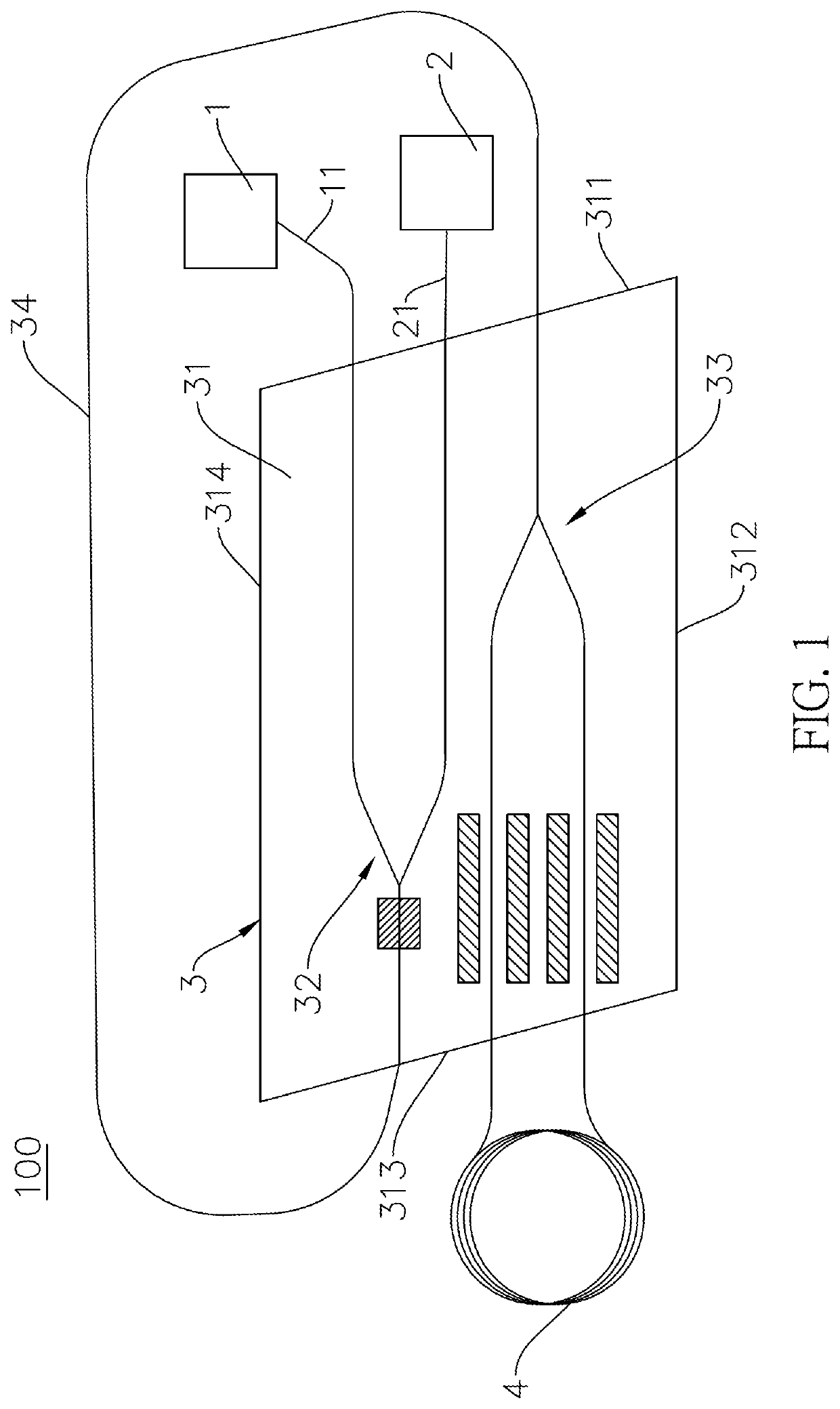
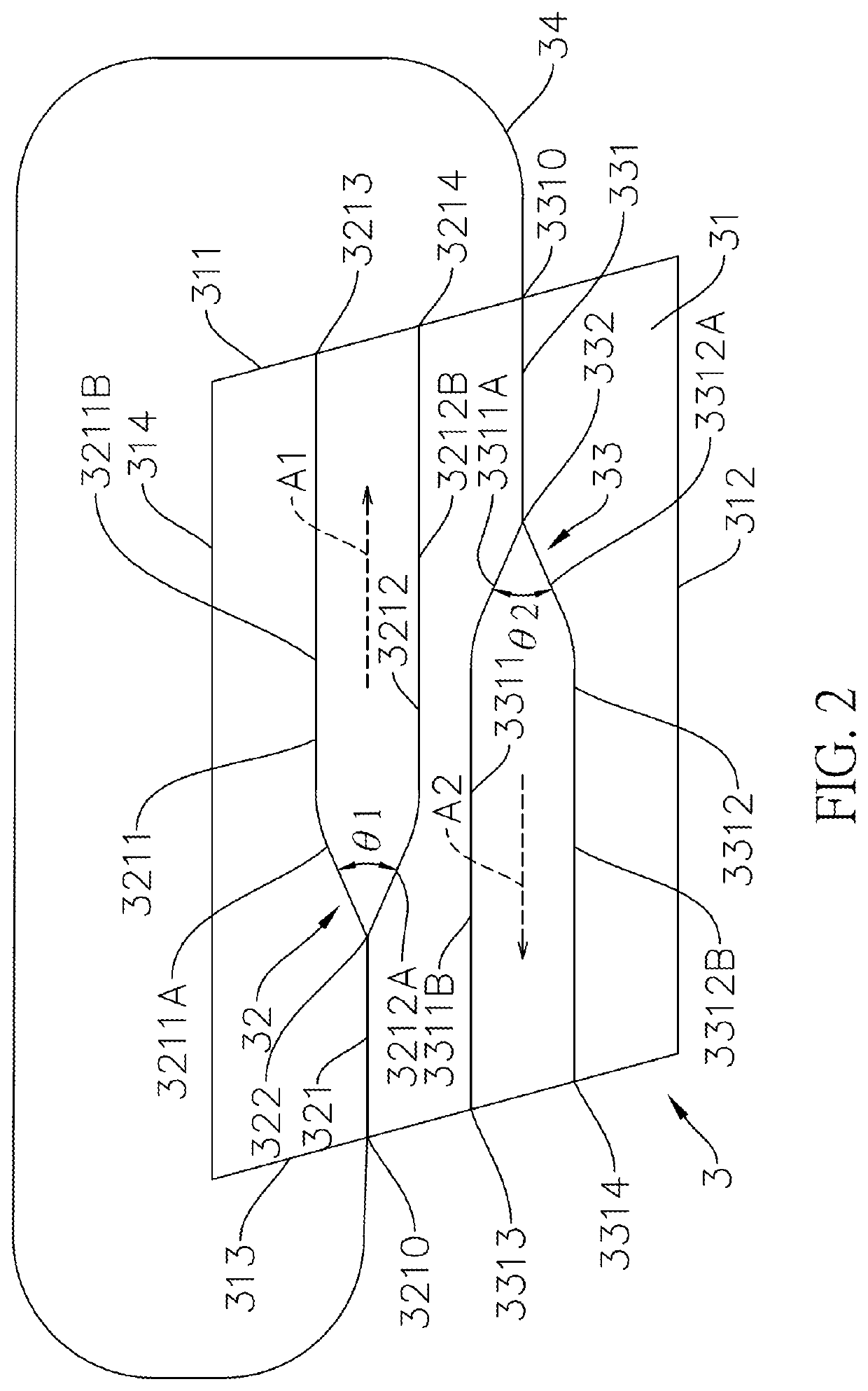
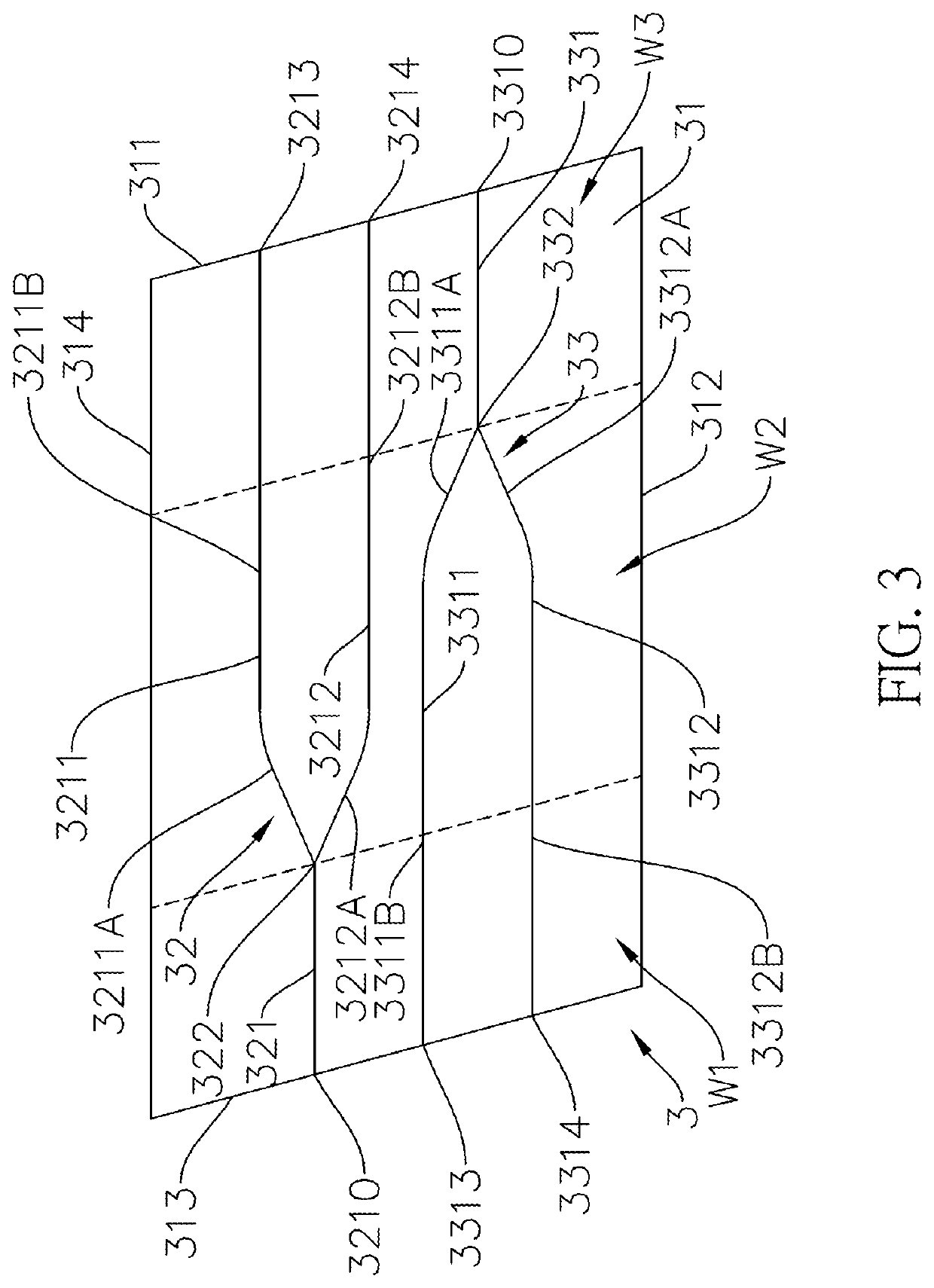
ActiveUS20210325602A1Meet the requirementsOptical light guidesNon-linear opticsGyroscopeCurrent sensor
An integrated biplane optical sensing core chip has a non-linear optical substrate, a first waveguide structure, a second waveguide structure and a waveguide coupled fiber. The non-linear optical substrate, the first waveguide structure and the second waveguide structure are made of the same nonlinear optical material. The first waveguide structure is connected to the second waveguide structure via a waveguide coupled fiber is outside and independent to the non-linear optical substrate. Therefore, the first waveguide structure and the second waveguide structure can overlap in the vertical direction, and can be set close to each other in the horizontal and vertical directions, so the integrated biplane optical sensing core chip can be miniaturized and can meet reciprocity. The integrated biplane optical sensing core chip is suitable for an optical fiber sensor, and the optical fiber sensor can be a fiber-optic gyroscope or a fiber-optic current sensor.
Owner:POLARIS PHOTONICS LTD