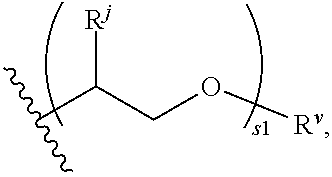
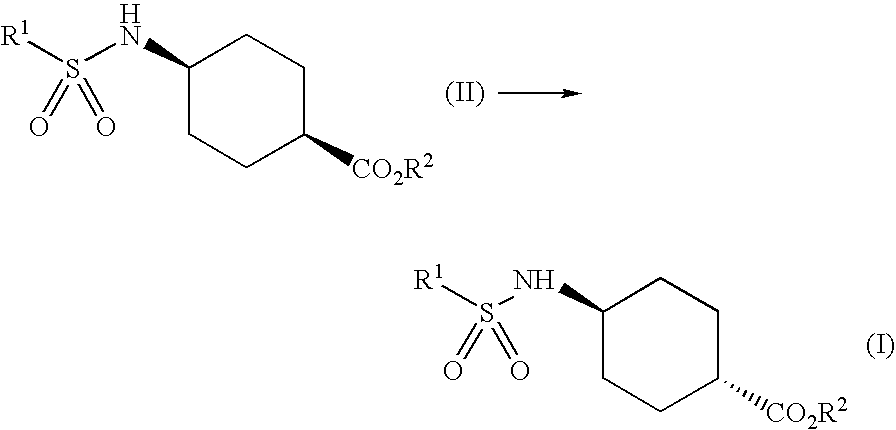
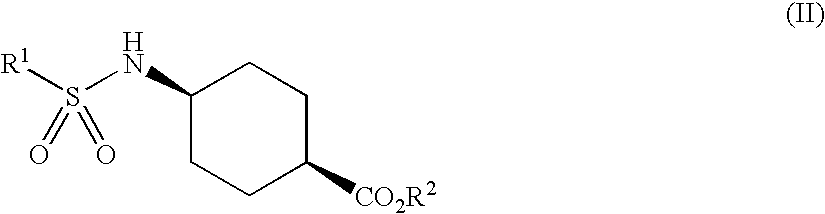
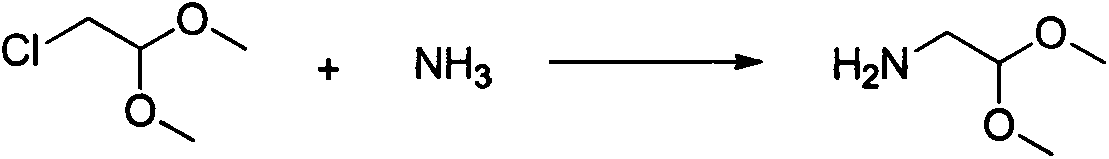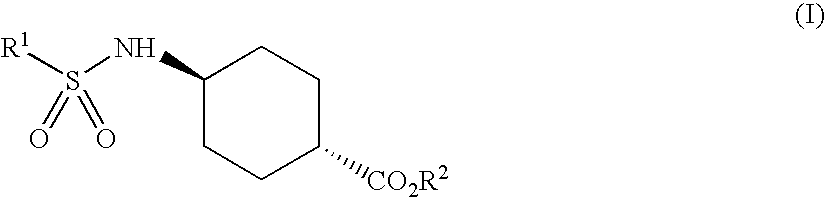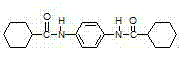Patents
Literature
119 results about "Cyclohexanecarboxylic acid" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
Cyclohexanecarboxylic acid is the organic compound with the formula C₆H₁₁CO₂H. It is the carboxylic acid of cyclohexane. It is an oily colorless oil that crystallizes near room temperature.
C7-C12 Secondary Alcohol Esters of Cyclohexanoic Acid
InactiveUS20100305250A1Easy to processGood low temperature flexibilityAlcoholCyclohexanecarboxylic acid
C7-C12 secondary alcohol esters of cyclohexanecarboxylic acids are especially useful as plasticizers for PVC and other plasticizable polymers. In embodiments, these plasticizers are useful in plastisol formulations to help improved processability of flexiblePVC material. In other embodiments these same plasticizers can be used in blends with other plasticizers to improve processability. In preferred embodiments these same plasticizers can be used to prepare flexible PVC compounds with enhanced low temperature flexibility, low toxicity, and improved resistance to outdoor aging.
Owner:EXXONMOBIL CHEM PAT INC
Compositions Based on C4-C7 Secondary Aliphatic Alcohol Esters of Cyclohexanecarboxylic Acids
InactiveUS20100310891A1Easy to processReduce solubilitySynthetic resin layered productsThin material handlingCyclohexanecarboxylic acidCarboxylic acid
C4-C7 secondary aliphatic alcohol esters of cyclohexanediacid are especially useful as plasticizers for PVC. In embodiments, these fast fusing plasticizers are useful in plastisol formulations to help improve the processability of flexible PVC material. In preferred embodiments these same plasticizers can be used in blends with other plasticizers, especially slower fusing plasticizers or plasticizers with reduced solvency, such as di-2-propylheptyl phthalate or di-isononyl cyclohexanedicarboxylic acid, to improve processability.
Owner:EXXONMOBIL CHEM PAT INC
Method for preparing praziquantel
InactiveCN103739601AThe synthesis process is simpleImproved post-treatment processOrganic chemistryDimethyl acetalCyclohexanecarboxylic acid
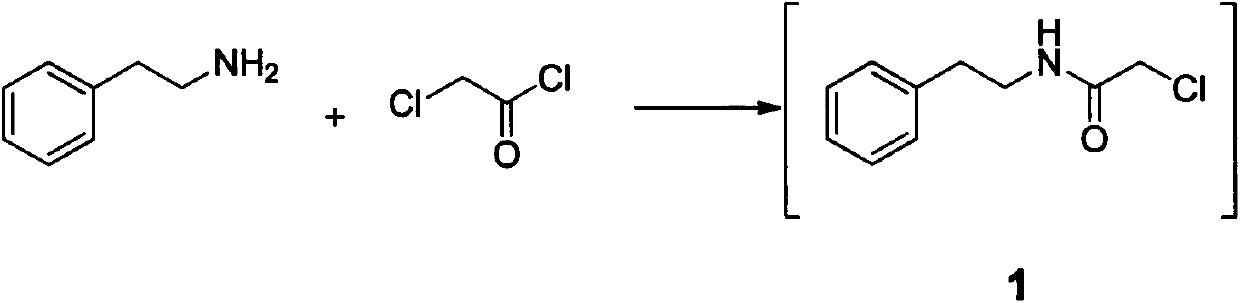
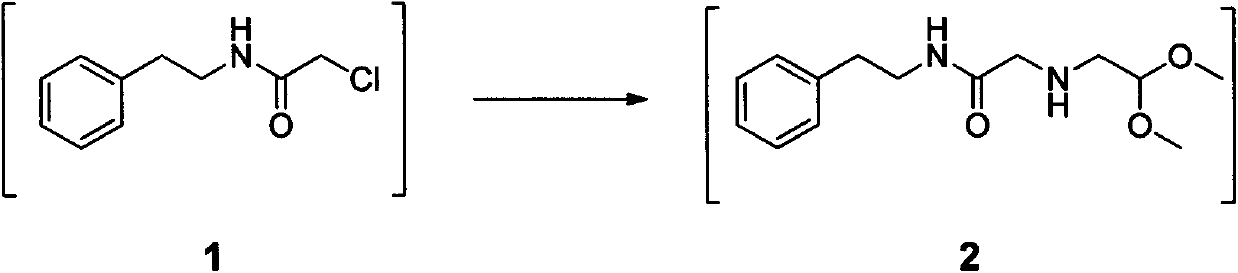
The invention relates to a method for preparing praziquantel, which is a one-pot method and comprises the following steps: performing an ammonolysis reaction of chloroacetaldehyde dimethyl acetal and an ammonia aqueous solution to generate aminoacetaldehyde dimethyl acetal; performing a condensation reaction of beta-phenylethylamine and chloroacetyl chloride in an organic solvent in alkaline environment to generate an intermediate 1; performing a condensation reaction of the intermediate 1 and the aminoacetaldehyde dimethyl acetal in an organic solvent to generate an intermediate 2; performing cyclization of the intermediate 2 in the presence of an acidic catalyst to generate an intermediate 3; performing a reaction of the intermediate 3 and cyclohexanecarboxylic acid chloride in an organic solvent in alkaline environment, and performing solvent crystallization to obtain the target product of praziquantel.
Owner:JIANGSU CHENGXIN PHARMA
Treatment of eye discomfort by topical administration of a cooling agent to the external surface of the eyelid
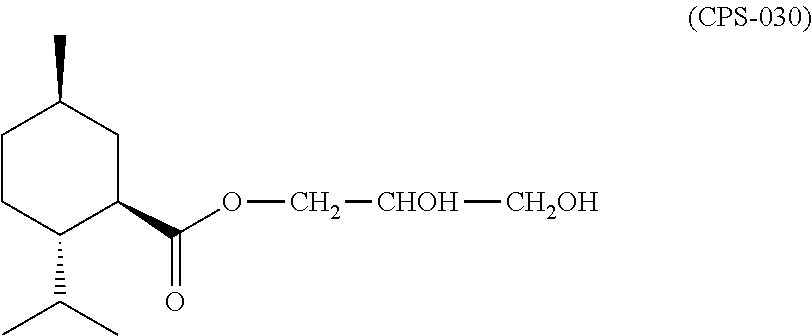
The present invention pertains generally to the field of ocular treatment, and more specifically to the use of a liquid cooling agent composition comprising a cooling agent for the treatment of (e.g., the alleviation of symptoms of; the amelioration of) eye discomfort. The preferred cooling agent is (1R,2S,5R)-2-isopropyl-5-methyl-cyclohexanecarboxylic acid 2,3-dihydroxy-propyl ester (referred to herein as CPS-030). The liquid cooling agent composition is topically administered to at least a portion of the external surface of the eyelid (preferably the closed eyelid) of the eye to be treated. Preferably, the liquid cooling agent composition is carried on or in a wipe, pad, or towelette, for example, an eye wipe.
Owner:WEI EDWARD TAK
Medical sodium alginate gel microsphere and preparation method and application thereof
InactiveCN103239730AControl releaseReduce erosionAntibacterial agentsPharmaceutical non-active ingredientsWater insolubleMicrosphere
The invention provides a medical sodium alginate gel microsphere and a preparation method and application of the medical sodium alginate gel microsphere. The medical sodium alginate gel microsphere consists of a composite medicine carrier and a water-insoluble medicine; the medicine is coated with the composite medicine carrier; and the composite medicine carrier is an ion crosslinking agent-sodium alginate-divalent metal ion, wherein the ion crosslinking agent is 4-aminomethylbenzoic acid or tranexamic acid. The preparation method comprises the following steps of: (1) mixing ion crosslinking agent aqueous solution with divalent metal ion solution in the same volume to obtain composite solidifying liquid; (2) dispersing medicine powder or an agent into sodium alginate aqueous solution; uniformly mixing; dropwise adding the mixture into the composite solidifying liquid obtained in step (1) through a high-voltage static droplet generating device or a syringe needle, so that the mixture drops are solidified into spheres; and (3) dehydrating gel microspheres which are washed with the distilled water; and drying at normal temperature. The medical sodium alginate gel microsphere can be used for treating tuberculosis, endocrine disease and tumor, and also can be used for treating local acute hemorrhage and chronic errhysis.
Owner:THE 309TH HOSPITAL OF CHINESE PEOPLES LIBERATION ARMY
Processes for making cyclohexane compounds
ActiveUS20150183699A1Promote resultsOther chemical processesOrganic compound preparationCyclohexanecarboxylic acidHydrogenation process
This invention relates to hydrogenation processes for making cyclohexane compounds. More specifically, this invention relates to hydrogenation processes in the presence of tertiary amide solvent compounds, as well as compositions that can result from such processes. The invention thus provides processes for making cyclohexanecarboxylic acid compounds and processes for making hydroxymethylcyclohexane compounds.
Owner:EASTMAN CHEM CO
Preparation method for levo-praziquantel
The invention relates to a preparation method for levo-praziquantel. The method includes the following steps that under the alkaline condition, a praziquantel amine phosphate compound serves as original raw material and is resolved by tartaric acid and derivatives of the tartaric acid, and the obtained levo-praziquantel amine tartrate compound is subjected to recrystallization; in the presence of an alkali compound, a hexamethylene formylation reaction is carried out on the recrystallized levo-praziquantel amine tartrate compound obtained and cyclohexanecarboxylic acid chloride to obtain a levo-praziquantel crude product, and the refined levo-praziquantel is obtained through decoloration, filtration, and filtrate recrystallization. The preparation method for the levo-praziquantel shortens a technological process, reduces generation of by-products and improves the quality and yield of the product. Recovered praziquantel amine compounds of the other configuration can be recycled after racemization, the resolving agent can be used repeatedly, and pollution to the environment is effectively reduced.
Owner:SHAOXING MINSHENG PHARMA
C7-C12 secondary alcohol esters of cyclohexanoic acid
InactiveUS8669311B2Good low temperature flexibilityImprove stabilityPlastic/resin/waxes insulatorsLavatory sanitoryAlcoholCyclohexanecarboxylic acid
C7-C12 secondary alcohol esters of cyclohexanecarboxylic acids are especially useful as plasticizers for PVC and other plasticizable polymers. In embodiments, these plasticizers are useful in plastisol formulations to help improved processability of flexible PVC material. In other embodiments these same plasticizers can be used in blends with other plasticizers to improve processability. In preferred embodiments these same plasticizers can be used to prepare flexible PVC compounds with enhanced low temperature flexibility, low toxicity, and improved resistance to outdoor aging.
Owner:EXXONMOBIL CHEM PAT INC
Prodrug bipyridylaminopyridines as syk inhibitors
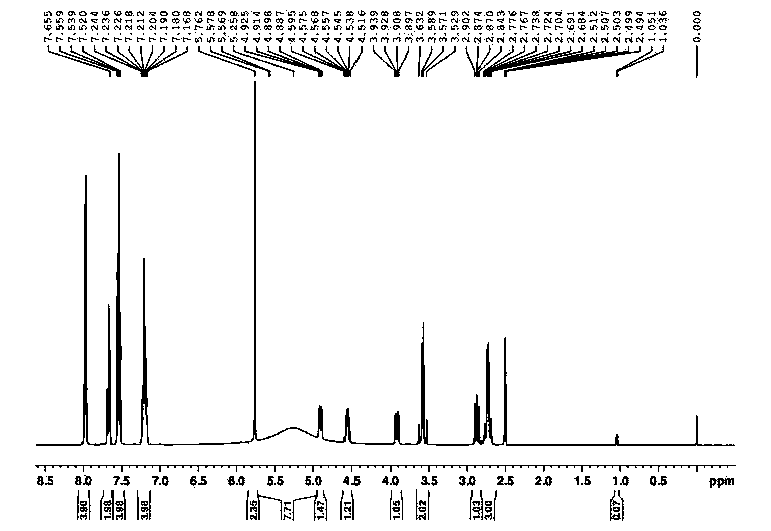
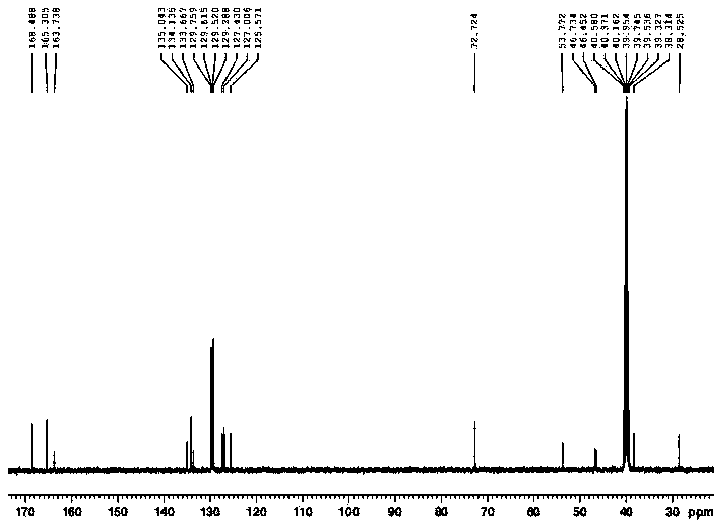
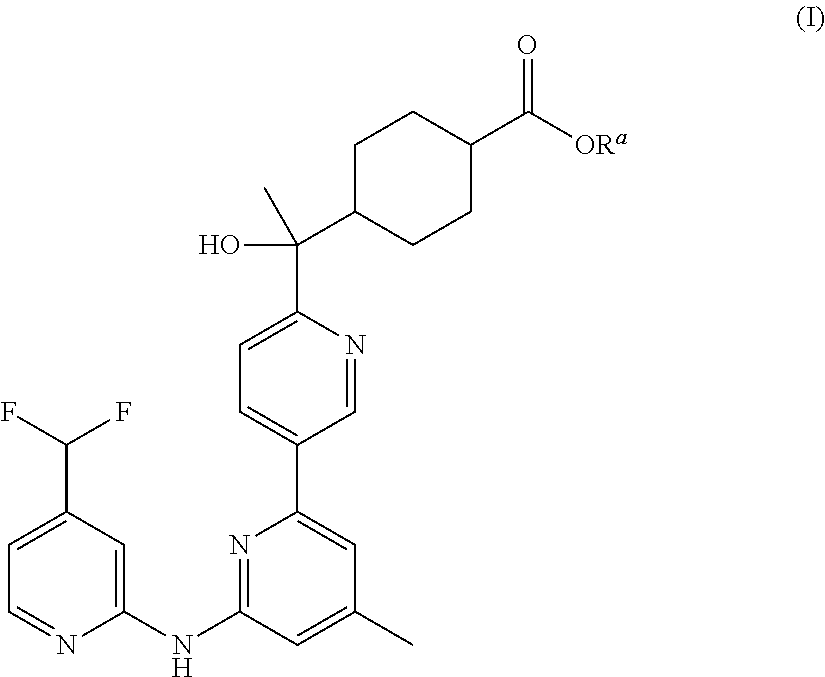
The present invention provides compounds of Formula (I), which are prodrugs of trans-4-[(1R)-(6-{[4-(difluoromethyl)pyridin-2-yl]amino}-4-methyl-2,3′-bipyridin-6′-yl)-1-hydroxyethyl]cyclohexanecarboxylic acid, a potent inhibitor of Syk. The compounds are useful in the treatment and prevention of diseases mediated by the enzyme, such as asthma, COPD, rheumatoid arthritis and cancer.
Owner:MERCK SHARP & DOHME CORP
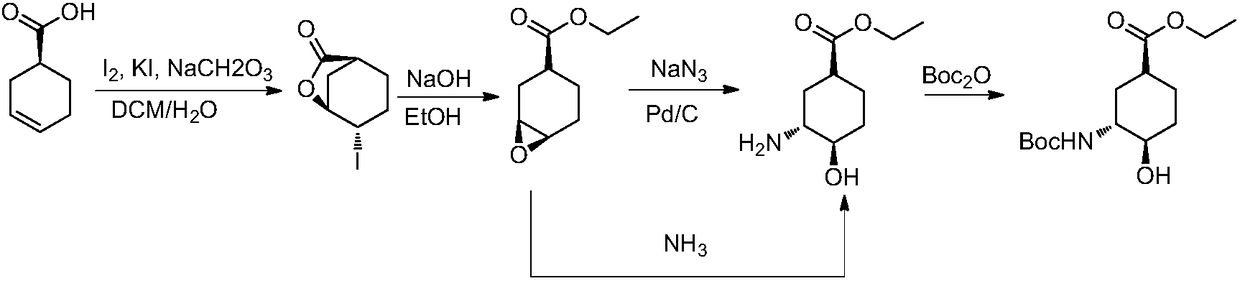
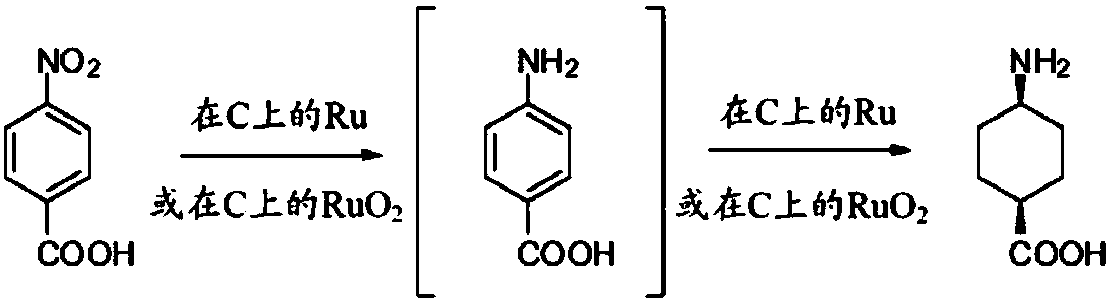
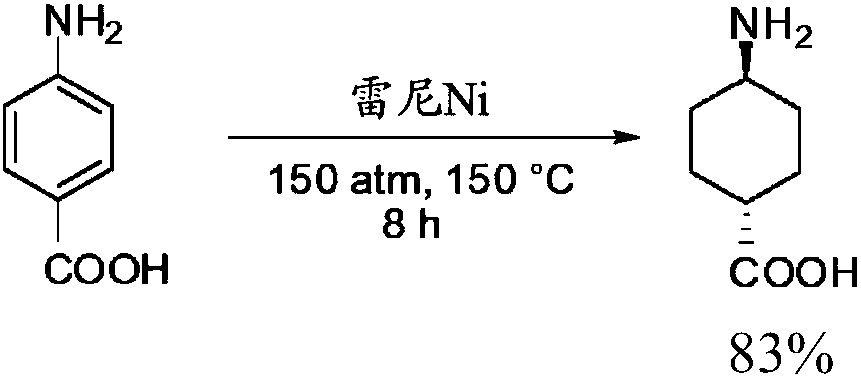
Process for trans-4-amino-1-cyclohexanecarboxylic acid derivatives
InactiveUS7314950B2Organic compound preparationOrganic chemistry methodsAcid derivativeCyclohexanecarboxylic acid
Owner:SHIONOGI & CO LTD
Catalyst for preparing cyclohexane carboxylic acid through benzoic acid hydrogenation and preparation method and application thereof
ActiveCN103599795AEasy to prepareRaw materials are widely availableCarboxylic preparation by ozone oxidationMetal/metal-oxides/metal-hydroxide catalystsBenzoic acidAdditive ingredient
The invention provides a catalyst for synthesizing cyclohexane carboxylic acid through benzoic acid hydrogenation. The catalyst contains a carrier, an assistant and an active ingredient, wherein the carrier is nickel foam, the assistant is activated carbon, and the active ingredient is a metallic element Pd. The invention further provides a preparation method of the catalyst and application of the catalyst as a monolithic catalyst in a fixed bed. According to the catalyst provided by the invention, the preparation method is simple, the raw materials are wide in source and are easily obtained, and the usage amount of precious metal is small; during application, the catalyst has strong carbon oxide poisoning resistance, high catalytic activity and good selectivity; compared with the traditional powder catalysts, the catalyst provided by the invention has the advantages that the problems of catalyst ingredient loss and catalyst wear are solved thoroughly, the separation of a reaction solution from the catalyst is not required, and the loading and replacing of the catalyst are very convenient and fast.
Owner:JIANGSU QINGQUAN CHEM CO LTD
Caprolactam production method
ActiveCN103012262AIncrease productionLow costLactams preparationBulk chemical productionPalladium on carbonBenzoic acid
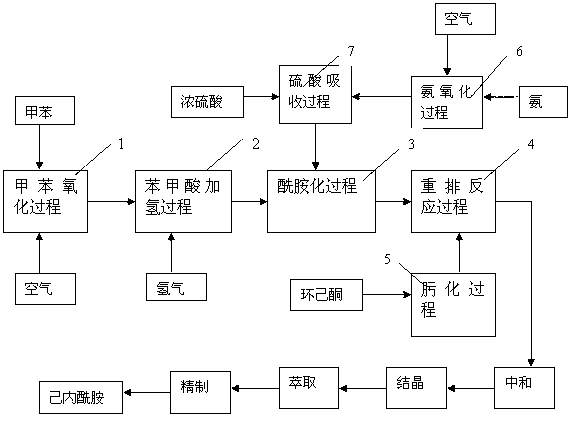
The present invention provides a caprolactam production method, which comprises the following processes that: toluene is oxidized under an effect of a cobalt salt catalyst to generate benzoic acid, benzoic acid is subjected to hydrogenation under an effect of a palladium carbon catalyst to generate cyclohexanecarboxylic acid, the cyclohexanecarboxylic acid and nitrosylsulfuric acid are subjected to an amidation reaction in an amidation reactor to generate an amide liquid, cyclohexanone is subjected to an oximation reaction to generate cyclohexanone oxime, the amide liquid and the cyclohexanone oxime are subjected to a rearrangement reaction in a rearrangement reactor, and the reaction product is subjected to processes such as neutralization, crystallization, extraction and refinement to prepare the ??caprolactam, wherein a rearrangement reaction temperature is 70-130 DEG C, pressure is 0-1.0 MPa, and a molar ratio of the acid in the amide liquid to the cyclohexanone oxime is 1-2:1. With the present invention, combination of the two processes such as a toluene method and a cyclohexanone-hydroxylamine method is achieved, a caprolactam yield is increased without increase of ammonium sulfate by-production, nicotinic acid consumption and ammonium sulfate by-production are reduced, the production process is simplified, and construction investment and production cost are saved.
Owner:CHINA PETROLEUM & CHEM CORP
Preparation method of edoxaban intermediate
ActiveCN107721866AEfficient manufacturingReduce manufacturing costCarbamic acid derivatives preparationOrganic compound preparationTert-Butyloxycarbonyl protecting groupLithium hydroxide
Owner:ASTATECH CHENGDU BIOPHARM CORP
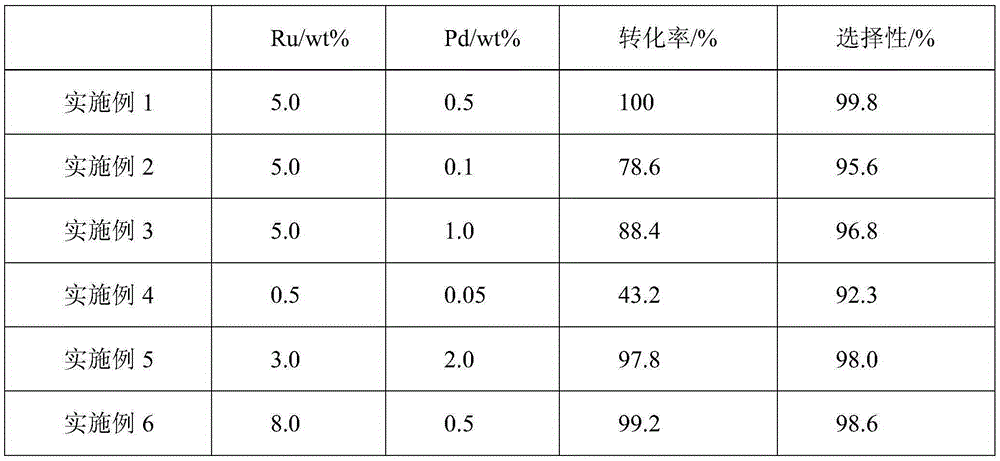
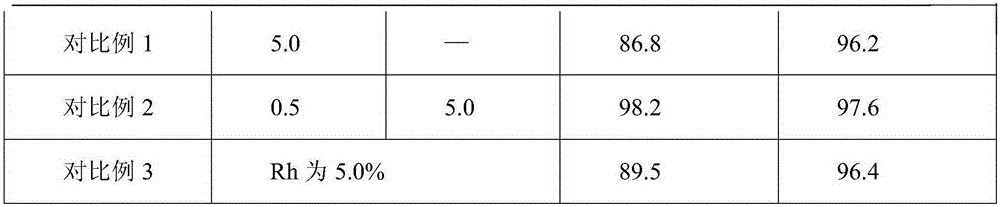
Ruthenium palladium/carbon catalyst of cyclohexanecarboxylic acid synthesized through benzoic acid hydrogenation and preparation method and application thereof
InactiveCN105251482AHigh reactivityHigh selectivityCarboxylic preparation by ozone oxidationMetal/metal-oxides/metal-hydroxide catalystsBenzoic acidActive component
The invention discloses a ruthenium palladium / carbon catalyst of cyclohexanecarboxylic acid synthesized through benzoic acid hydrogenation. The catalyst comprises a carbon material carrier, an active component of metal element Ru nanoparticles and an auxiliary of metal element Pd nanoparticles. The loading capacity of the Ru nanoparticles is 0.5-0.8%, and the loading capacity of the Pd nanoparticles is 0.05-2.0%. The invention further discloses a preparation method of the ruthenium palladium / carbon catalyst. Firstly, the carbon material carrier is prepared, then the active component ruthenium and the auxiliary palladium are loaded, a catalyst precursor is obtained, and finally the catalyst precursor is dried and reduced. The catalyst avoids use of a large amount of palladium, has high reaction activity, selectivity and stability, can be used circularly and remarkably reduces production cost. The catalyst is used for synthesizing cyclohexanecarboxylic acid through benzoic acid catalytic hydrogenation. The raw material conversion rate is 100%, the purity of a target product can be higher than 99.5%, process conditions are moderate, equipment is simple, three wastes are not emitted, and industrialization is easily achieved.
Owner:HIGH TECH RES INST NANJING UNIV LIANYUNGANG +1
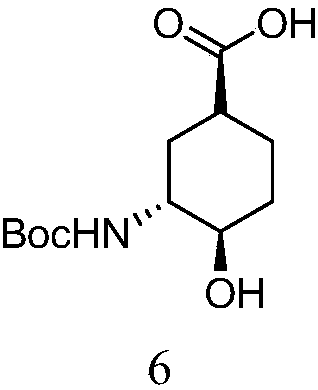
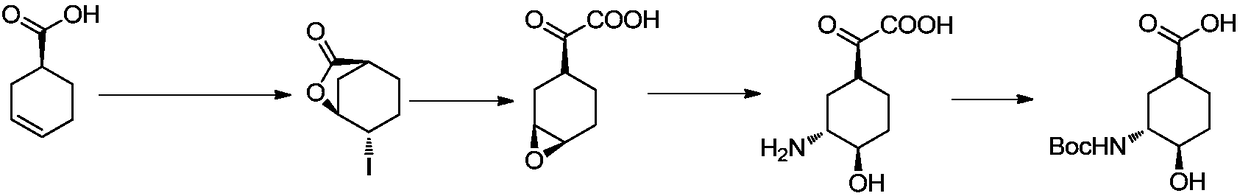
Preparation method for high-purity edoxaban intermediate
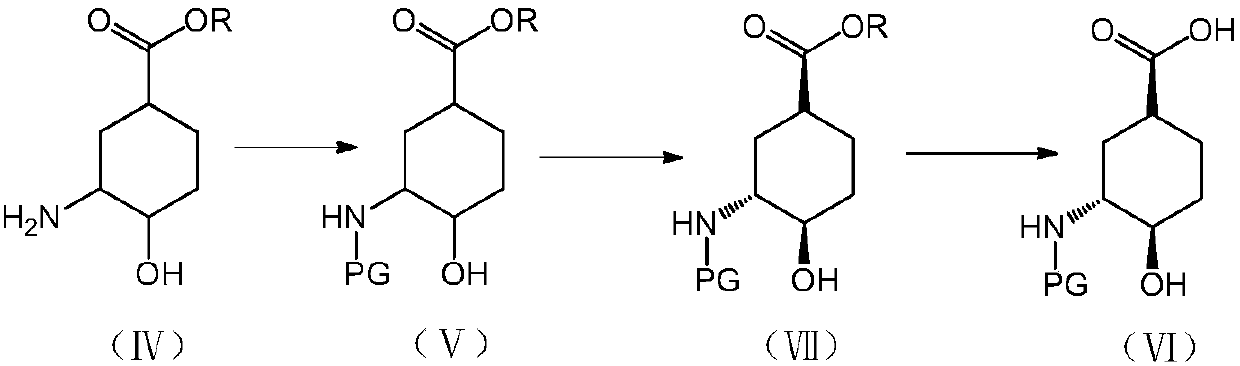
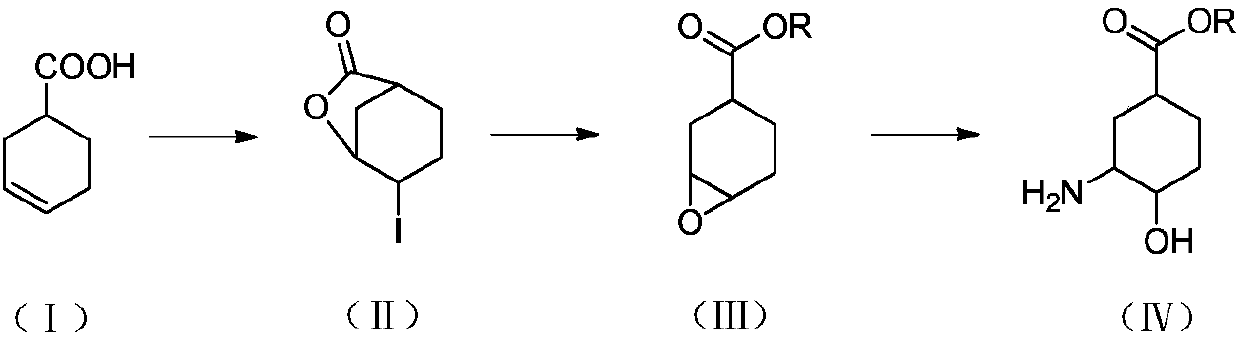
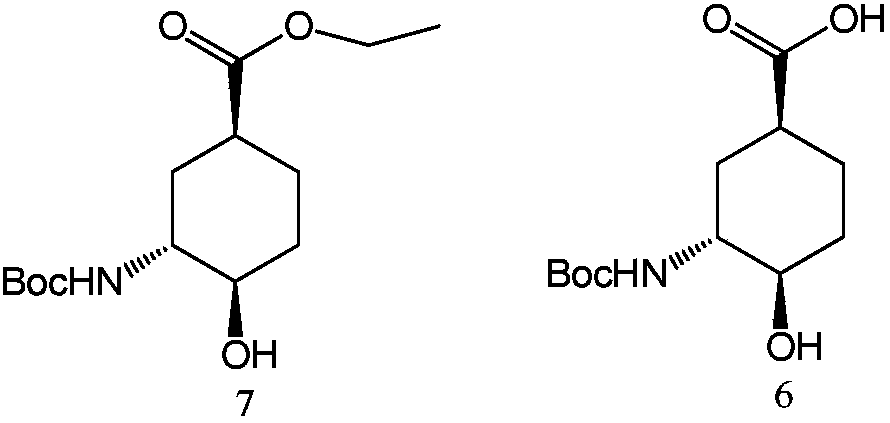
The invention discloses a preparation method for the high-purity edoxaban intermediate (1S,3R,4R)-3-tertbutyloxycarbonylamino-4-hydroxy-naphthenic acid. The preparation method comprises the followingsteps: with 3-amino-4-hydroxy-cyclohexane carboxylate as shown in a formula (IV) which is described in the invention as a raw material, subjecting the raw material to protection by an amino protectivegroup so as to obtain 3-tertbutyloxycarbonylamino-4-hydroxy-cyclohexane carboxylate as shown in a formula (V) which is described in the invention; and carrying out enzyme hydrolysis and splitting soas to obtain optically pure (1S,3R,4R)-3-tertbutyloxycarbonylamino-4-hydroxy-naphthenic acid as shown in a formula (VI) which is described in the invention. The preparation method provided by the invention has the advantages of simple operation, environmental protection, high selectivity, low cost and the like, can realize large-scale industrial production, and is convenient for industrial promotion and application.
Owner:ASTATECH CHENGDU BIOPHARM CORP
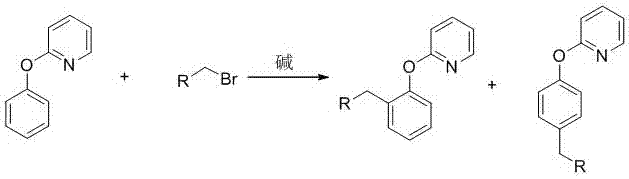
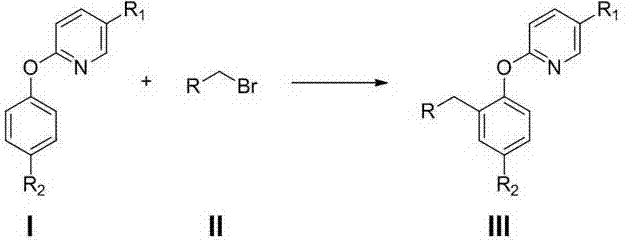
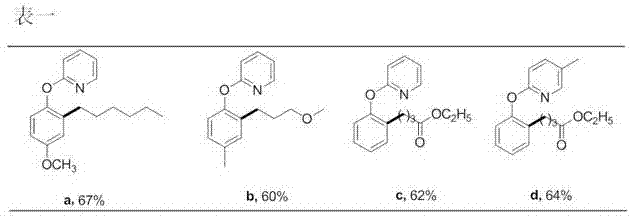
Preparation method of 2-(2-alkylphenoxy)pyridine derivative
InactiveCN107043350AHigh puritySynthetic raw materials are readily availableOrganic chemistryCyclohexanecarboxylic acidGlycol synthesis
A preparation method of a 2-(2-alkylphenoxy)pyridine derivative adopts a reaction of a 2-phenoxypyridine derivative and a primary bromoalkane compound. The method concretely comprises the following steps: directly adding the 2-phenoxypyridine derivative, the primary bromoalkane compound, a catalyst, an additive, an alkali and a solvent into a reaction device, stirring and heating the above added substances until the temperature is 90-130 DEG C, carrying out a reaction for 18-36 h, and separating the obtained product to obtain the 2-(2-alkylphenoxy)pyridine derivative, wherein the catalyst is dichloro(p-cymene)ruthenium dimer; the alkali is potassium carbonate or lithium carbonate; the additive is 1-adamantanecarboxylic acid or naphthenic acid; and the solvent is benzene or DMF or ethylene glycol dimethyl ether. The method has the advantages of easiness in obtaining of synthesis raw materials, simple process, and very good specificity of the reaction.
Owner:ANYANG NORMAL UNIV
Processes for making cyclohexane compounds
ActiveUS20150183706A1Promote resultsOther chemical processesCarboxylic preparation by ozone oxidationHydrogenation processCyclohexanecarboxylic acid
This invention relates to hydrogenation processes for making cyclohexane compounds. More specifically, this invention relates to hydrogenation processes in the presence of tertiary amide solvent compounds, as well as compositions that can result from such processes. The invention thus provides processes for making cyclohexanecarboxylic acid compounds and processes for making hydroxymethylcyclohexane compounds.
Owner:EASTMAN CHEM CO
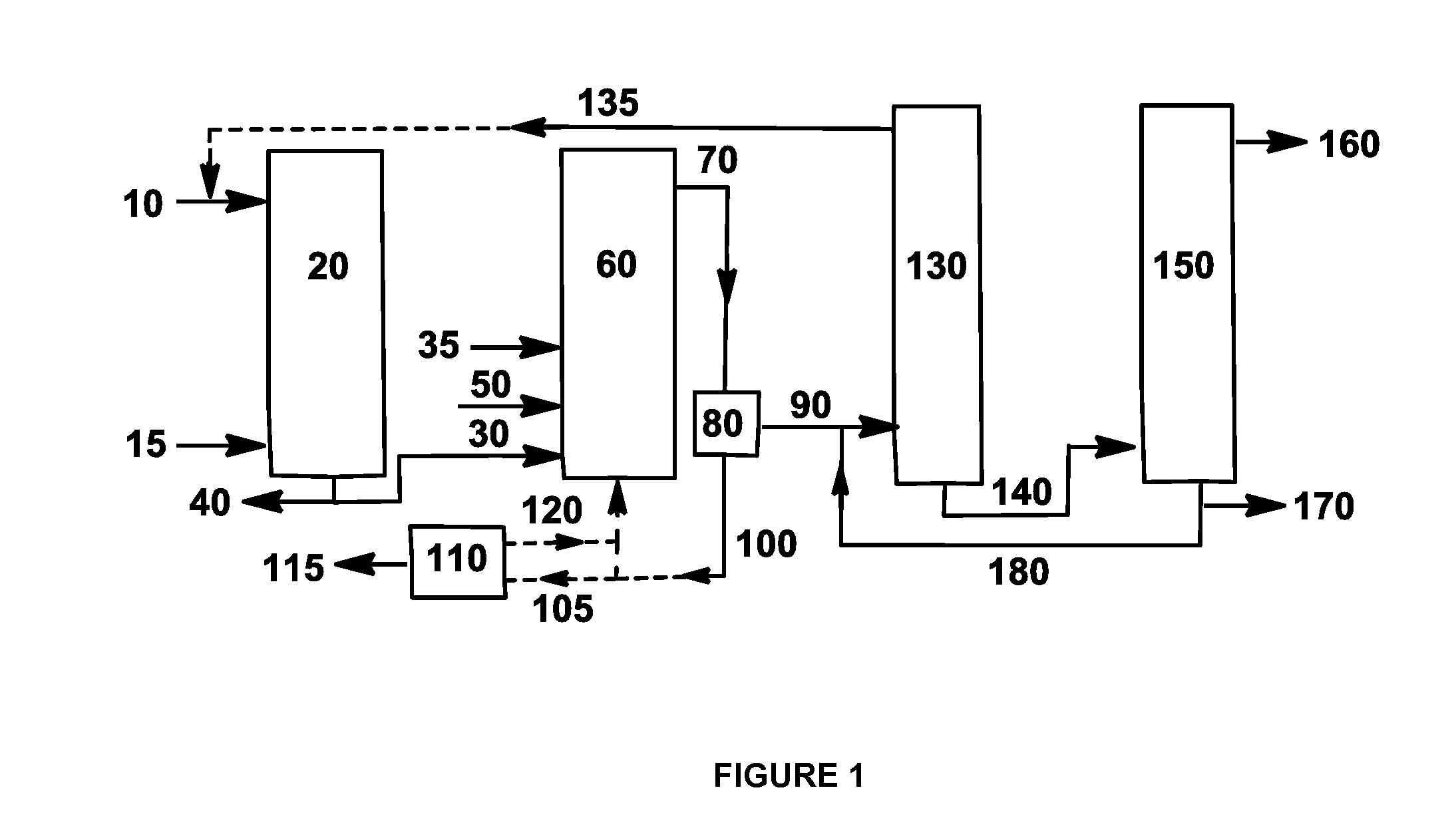
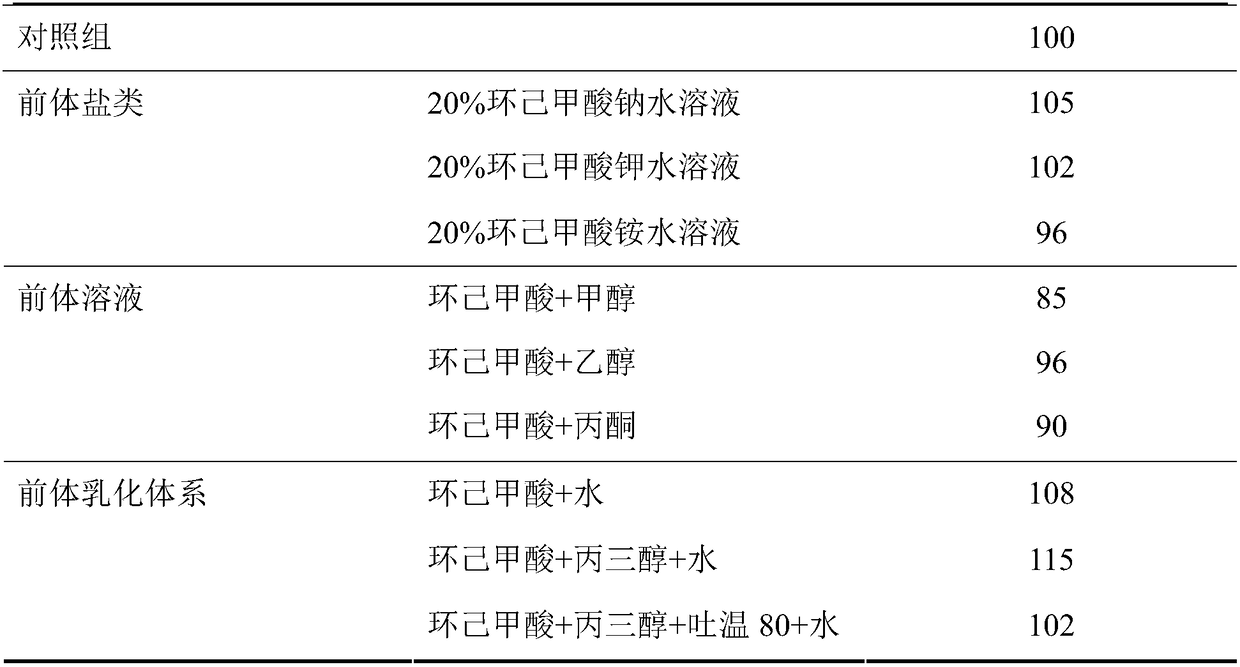
High-yield fermentation production method of doramectin
ActiveCN108396045ARaise the level of fermentationSimple methodMicroorganism based processesFermentationCyclohexanecarboxylic acidFermentation
The invention provides a high-yield fermentation production method of doramectin. The high-yield fermentation production method comprises the following steps: dissolving a doramectin precursor into water, methanol or ethanol or dispersing the doramectin precursor into a glycerol emulsifying system so as to be convenient for disinfection and material supplementation, wherein the doramectin precursor is cyclohexanecarboxylic acid or sodium cyclohexanecarboxylate; inoculating a strain seed solution into a fermentation culture medium and fermenting; adding the precursor which accounts for 0.02 to0.08 percent of the total weight of the fermentation culture medium at the early period of fermentation for induction; adding the precursor which accounts for 0.18 to 0.22 percent of the total weightof the fermentation culture medium at the middle period of the fermentation; adding the precursor which accounts for 0.02 to 0.08 percent of the total weight of the fermentation culture medium at thelater period of the fermentation. The method is simple and effective, and can be used for rapidly improving a fermentation level of the doramectin; the production cost is reduced and extra equipment and manpower do not need to be increased; the fermentation level of the doramectin can be relatively greatly improved and the production method is relatively suitable for commercial large-scale production.
Owner:HUBEI HONCH PHARMA
Method for preparing hexahydrophthalic anhydride
ActiveCN106674172AHigh yieldFlexible operationOrganic chemistryMetal/metal-oxides/metal-hydroxide catalystsFixed bedCyclohexanecarboxylic acid
The invention provides a method for preparing hexahydrophthalic anhydride, belonging to the field of synthesis of fine chemicals. The method comprises the steps: phthalic anhydride is heated to 50 DEG C and injected into a phthalic anhydride dissolving tower, the phthalic anhydride is dissolved with a solvent, the temperature of the dissolving tower is 50-70 DEG C, the dissolved phthalic anhydride solution is pressurized and injected into a constant-pressure reaction kettle or a fixed bed reactor filled with a selective hydrogenation catalyst to carry out selective hydrogenation reaction, the product directly enters a solvent recovery column after hydrogenation reaction, the solvent is distilled off at the tower top, the materials at the tower bottom enter a hexahydrophthalic anhydride rectifying tower, the hexahydrophthalic anhydride is obtained at the tower bottom, and a mixture of phthalide, o-toluic acid and o-methyl cyclohexanecarboxylic acid is obtained at the tower top; and the mixture obtained at the top of the rectifying tower is recrystallized to crystallize phthalide. The method has the advantages of simple operation, favorable economic benefit and industrial application prospects.
Owner:DALIAN UNIV OF TECH +1
Preparation method of amide polypropylene beta crystal form nucleating agent by taking p-phenylenediamine as skeleton
ActiveCN103539692AConvenient sourceStable storageOrganic compound preparationCarboxylic acid amides preparationIce waterCyclohexanecarboxylic acid
The invention discloses a preparation method of an amide polypropylene beta crystal form nucleating agent by taking p-phenylenediamine as a skeleton. The preparation method disclosed by the invention comprises the following steps of putting p-phenylenediamine into a flask, adding a solvent and an acid-binding agent in sequence, carrying out the temperature in an ice-water bath, dropping cyclohexanecarboxylic acid chloride, controlling the dropping temperature, increasing the temperature to reflux temperature after dropping, keeping the temperature for three hours, evaporating out the solvent, and washing through hot water and alcohol to obtain N-N'-1,4 benzene dicyclohexyl formamide. The preparation method disclosed by the invention has the benefits that raw materials are easily obtained, the process operation is simple, and the prepared product is high in yield and purity, has a good nucleating effect and is easy for industrialization.
Owner:山西科通化工有限公司
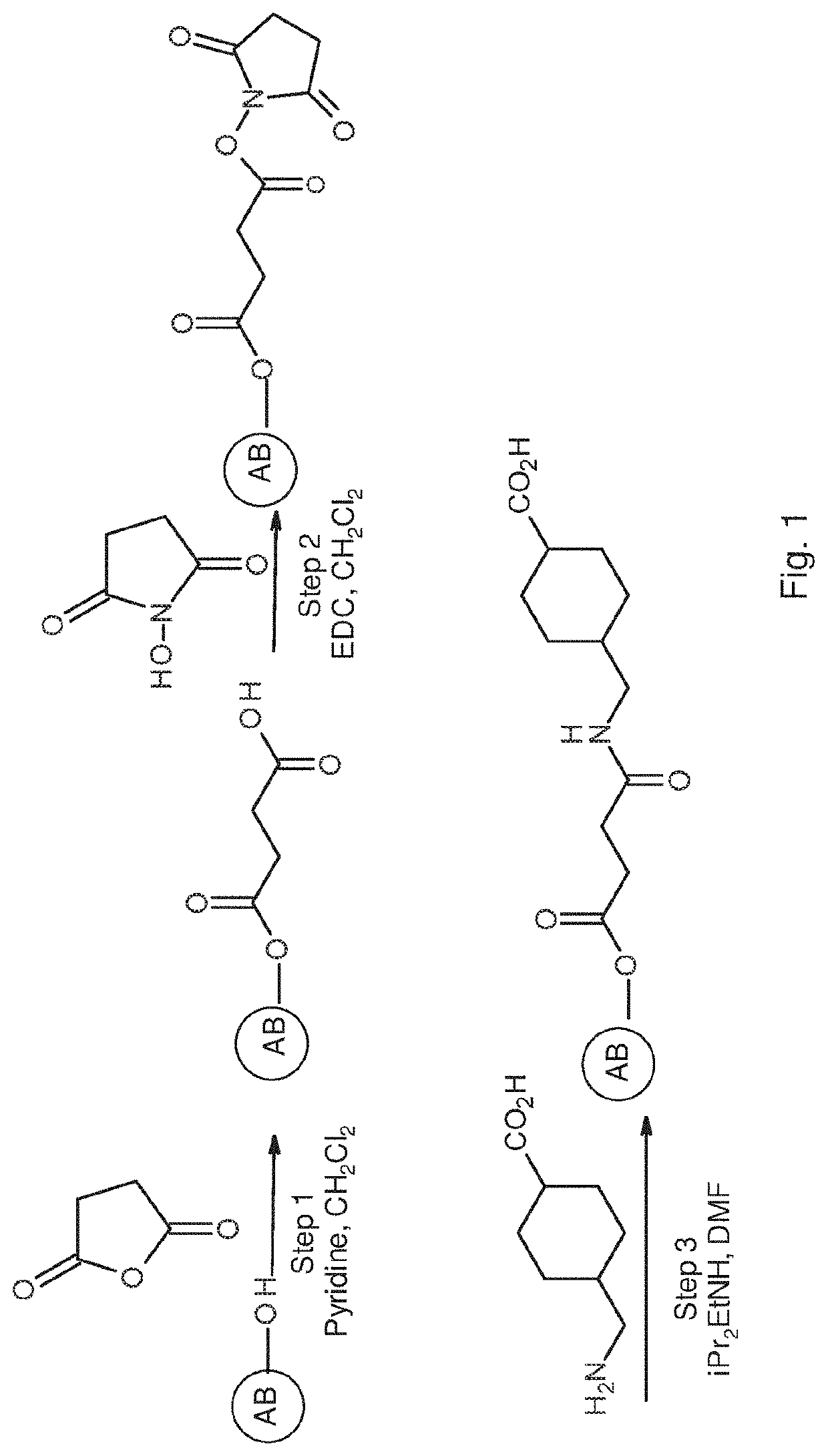
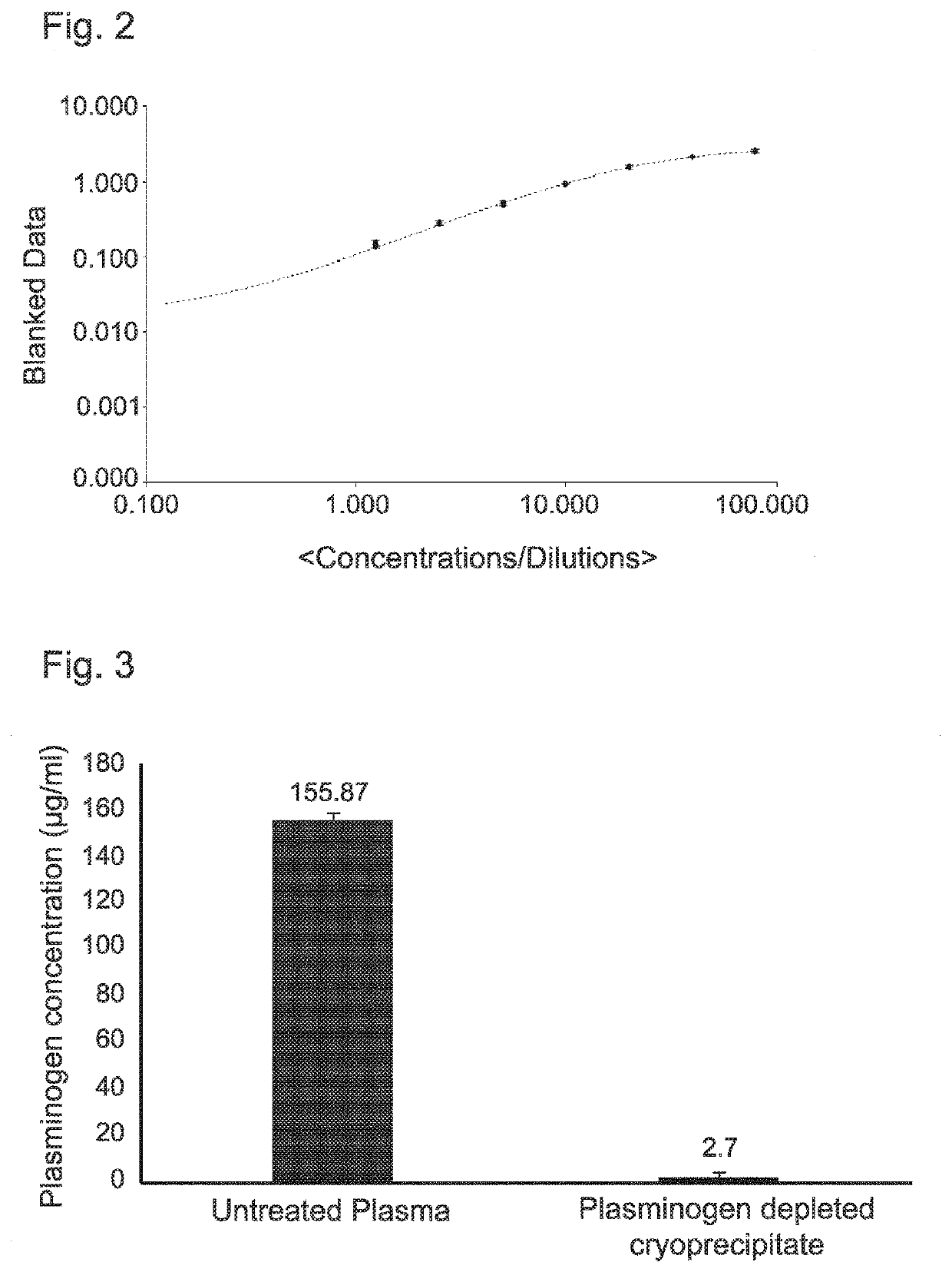
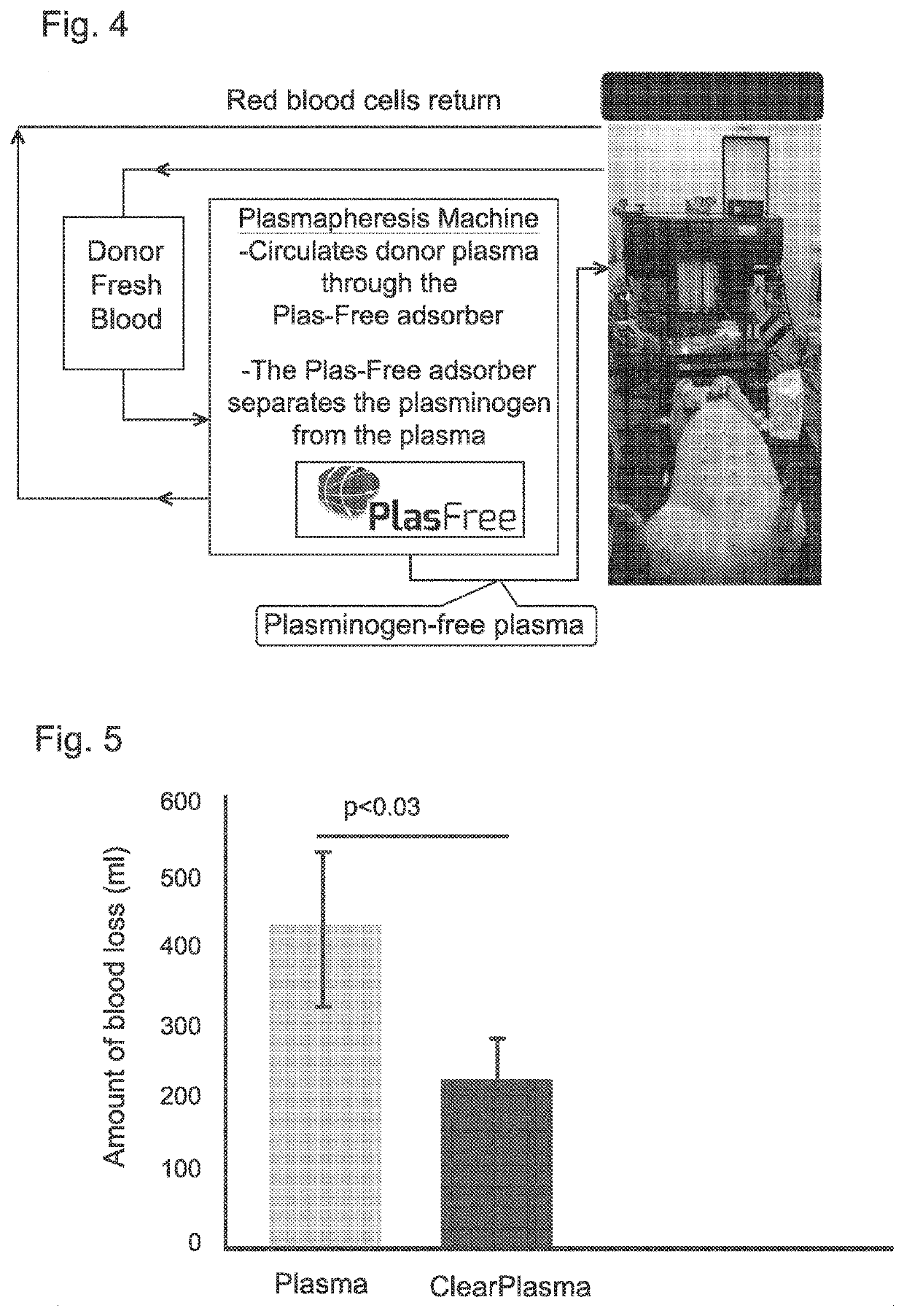
Extracorporeal device and matrix for removing fibrinolytic proteins from biological fluids, methods and uses thereof
PendingUS20210001023A1Treatment and prevention and prophylaxis and amelioration and inhibitionReduced fibrinolytic activityOrganic chemistryPeptide/protein ingredientsCyclohexanecarboxylic acidCarboxylic acid
The presently disclosed subject-matter provides specific compositions, conjugates, device, kits and systems for depleting fibrinolytic agents from biological fluids. The presently disclosed subject-matter further relates to the resulting biological fluid products that are devoid in fibrinolytic activity, therapeutic methods and uses thereof. The conjugates comprise a particle, at least one linker and at least one amino acid, derivative thereof or analog thereof being at least one of 4-(aminomethyl)-cyclo-hexane-carboxylic acid (tranexamic acid), epsilon-amino caproic acid, lysine, cyclohexanecarboxylic acid and 4-methyl-cyclohexanecarboxylic acid. A plurality of different conjugates (e.g. differing in particle size or type of linker) can be used.
Owner:PLAS FREE LTD
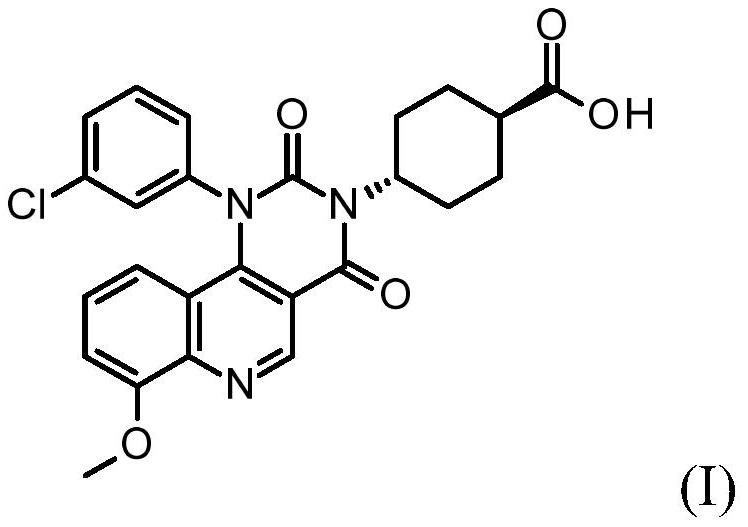
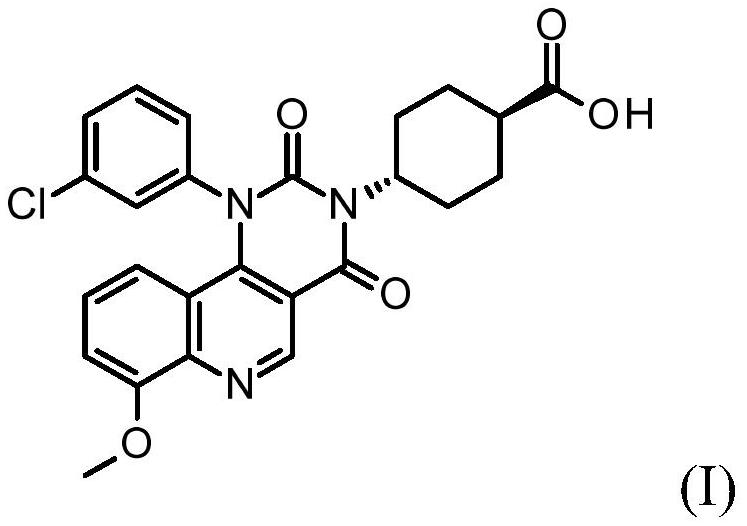
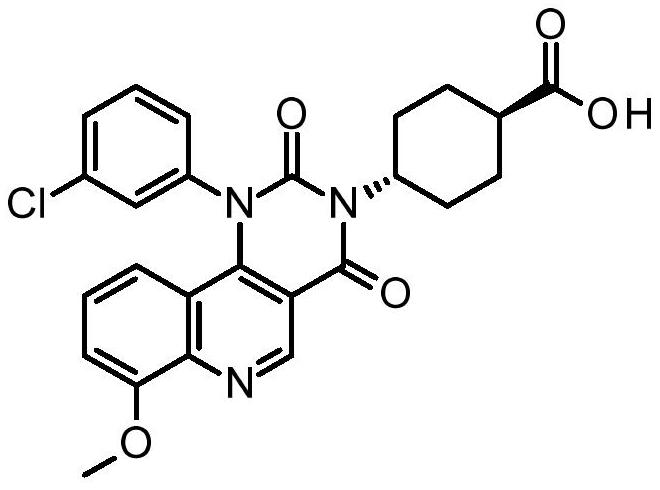
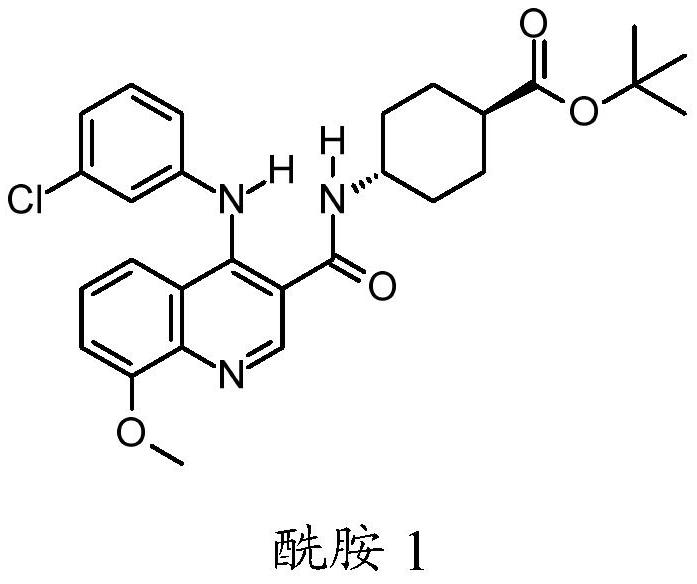
Quinoline derivatives, pharmaceutically acceptable salts thereof, and methods of use thereof
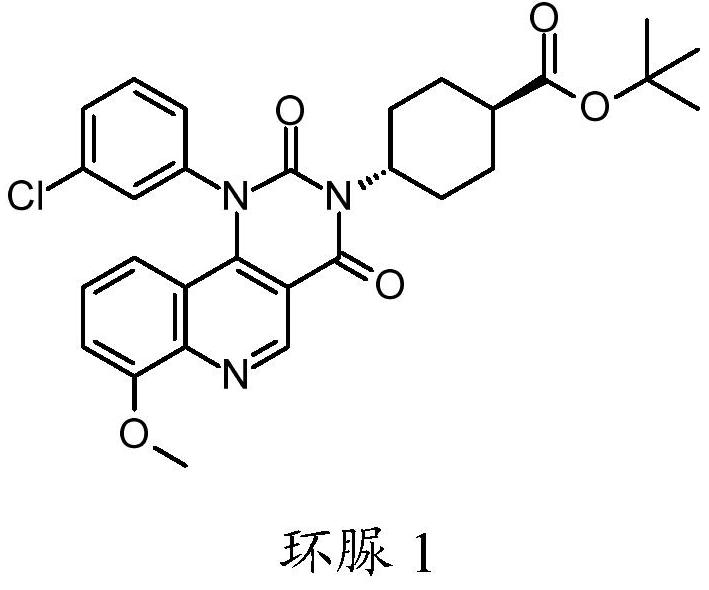
ActiveCN111808098AAntibacterial agentsOrganic active ingredientsChlorobenzeneCyclohexanecarboxylic acid
The present invention relates to compounds trans-4-[1-(3-chlorphenyl)-7-methoxy-2, 4-dioxo-pyrimido [5, 4-c] quinoline-3-yl] cyclohexanecarboxylic acid, pharmaceutically acceptable salts thereof, andtheir use as therapeutic agents, including in the treatment of inflammatory disorders.
Owner:VTV THERAPEUTICS LLC +1
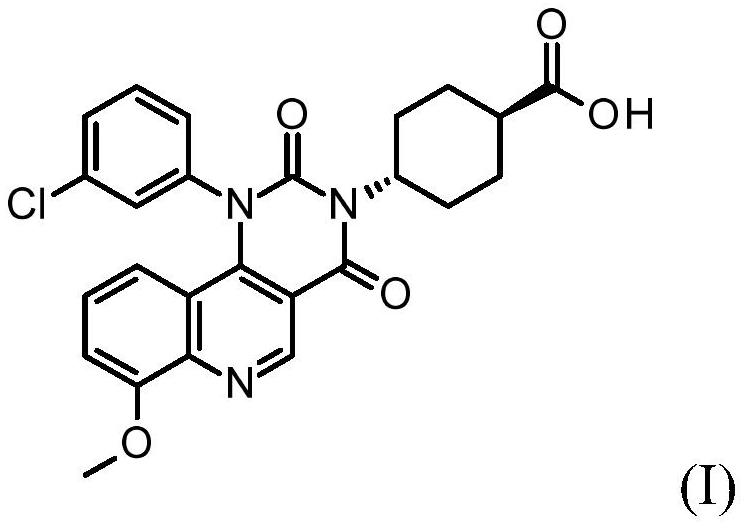
Quinoline derivatives, pharmaceutically acceptable salts thereof and methods of use thereof
ActiveCN111808098BAntibacterial agentsOrganic active ingredientsChlorobenzeneCyclohexanecarboxylic acid
The present invention relates to compound trans-4-[1-(3-chlorophenyl)-7-methoxy-2,4-dioxo-pyrimido[5,4-c]quinoline-3-yl] Cyclohexanecarboxylic acid, its pharmaceutically acceptable salts and their use as therapeutic agents, including in the treatment of inflammatory disorders.
Owner:VTV THERAPEUTICS LLC +1
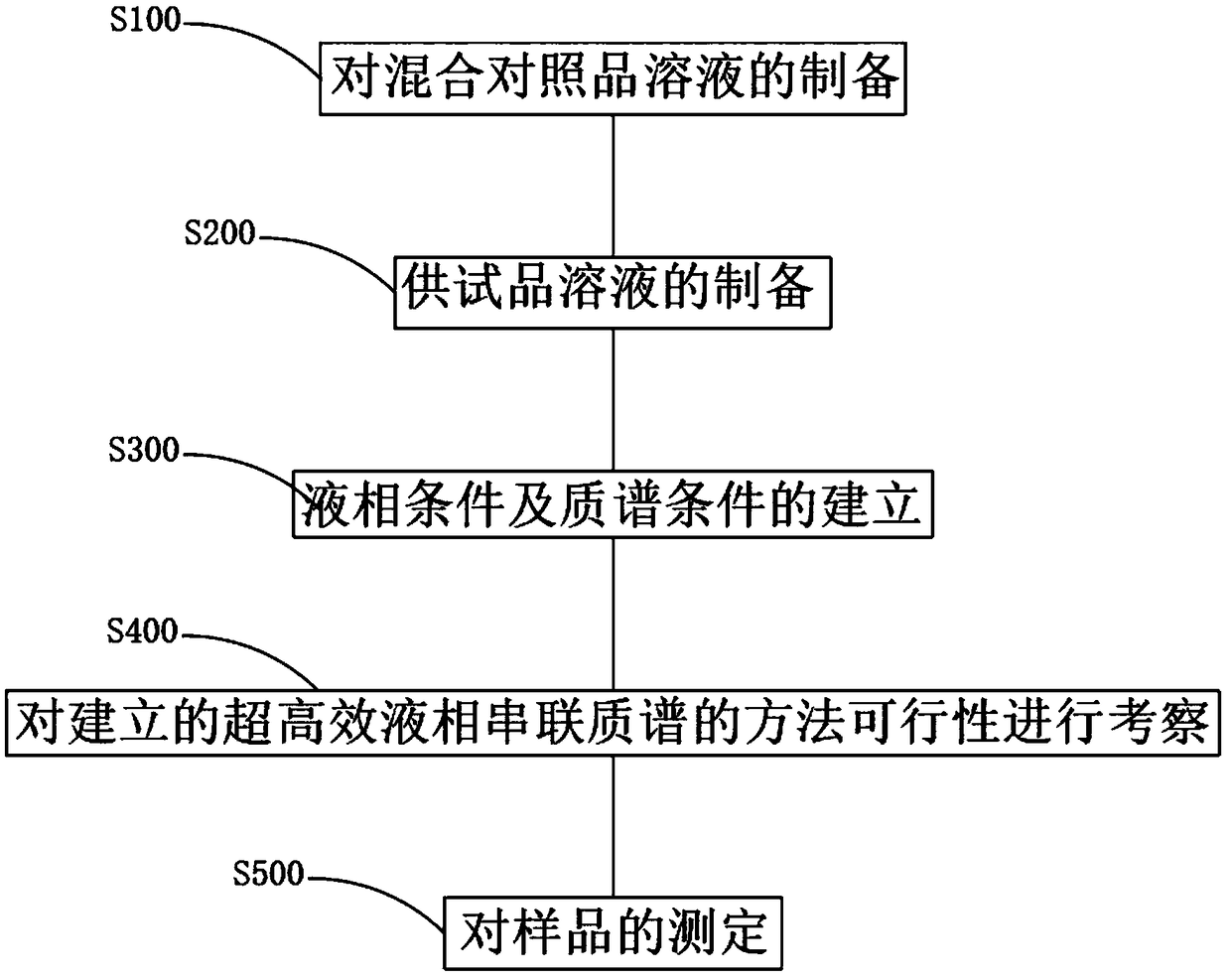
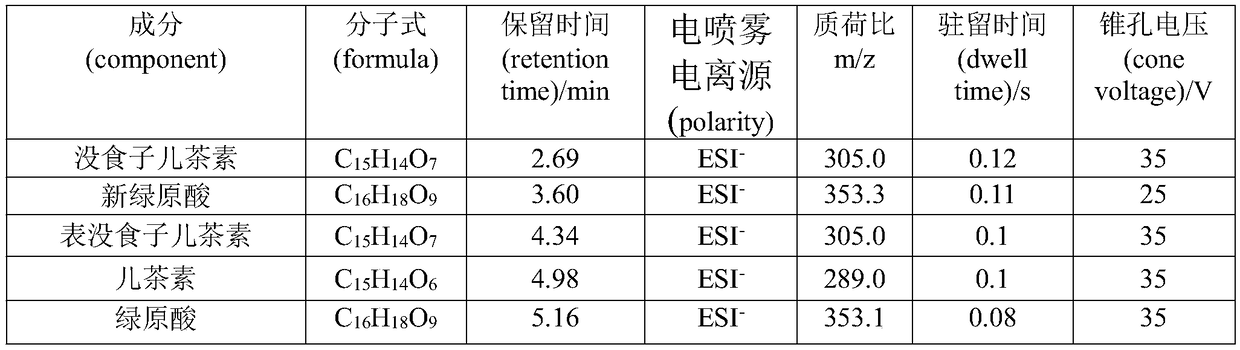
Multi-component content determination method of Miao medicine Laportea bulbifera
InactiveCN109270177ASimple methodFast wayComponent separationChlorogenic acidCyclohexanecarboxylic acid
The invention discloses a multi-component content determination method of Miao medicine Laportea bulbifera. The method comprises the following steps: S100, preparation of mixed reference solution, respectively and accurately weighing right amount of reference substances of Gallocatechin, neochlorogenic acid, Epigallocatechin, catechinic acid, chlorogenic acid, Cyclohexanecarboxylic acid, epicatechin, rutin, isoquercitrin, kaempferol-3-0-rutinoside and quercitrin; and simultaneously determining contents of eleven components including the Gallocatechin, the neochlorogenic acid, the Epigallocatechin, the catechinic acid, the chlorogenic acid, the Cyclohexanecarboxylic acid, the epicatechin, the rutin, the isoquercitrin, the kaempferol-3-0-rutinoside and the quercitrin. The method is simple, efficient, highly sensitive and excellent in specificity, and can provide baisis for quality control of Laportea bulbifera medicinal materials. Compared with the ordinary liquid phase, the method has the advantages of being accurate, reliable and highly sensitive, and having lower specificity detection limit and quantification limit, and is more effective in medicinal material component content analysis through a liquid chromatography-mass spectrometry analysis technology.
Owner:GUIZHOU MEDICAL UNIV
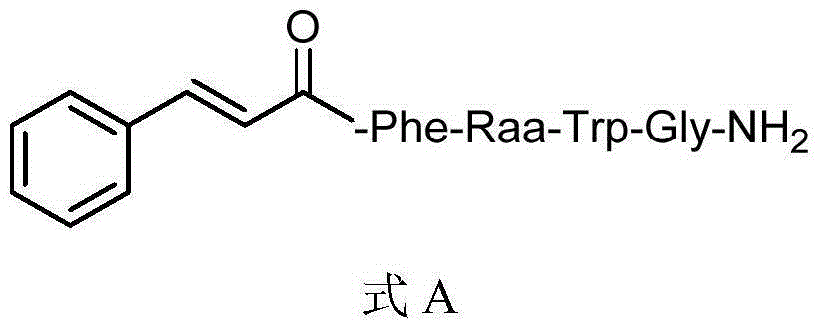
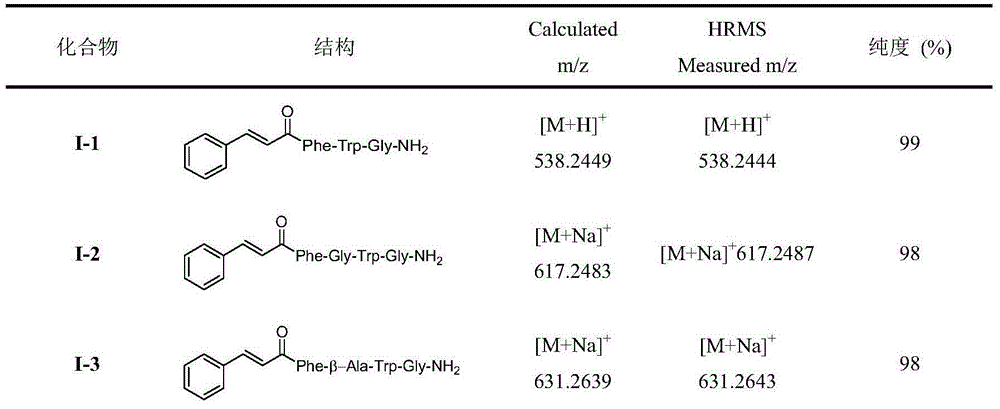
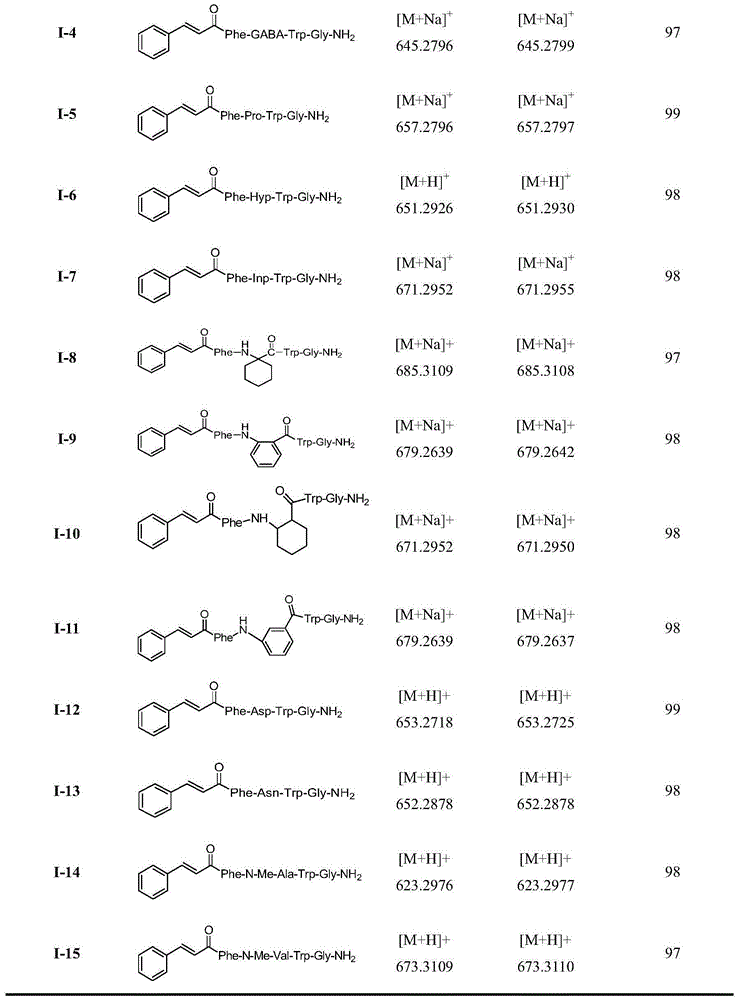
Insect kinin analogs and application thereof
The invention discloses insect kinin analogs and an application of the insect kinin analogs in control of pests and phytopathogen. A structural formula A of a prepared compound is shown in the specification, wherein Raa represents H and (non-natural) natural amino acids and comprises Gly, beta-Ala, GABA (gamma amino acid butyric acid), Pro, Hyp, Inp, 1-amino-cyclohexanecarboxylic acid, o-aminobenzoic acid, 2-amino-cyclohexanecarboxylic acid, m-aminobenzoic acid, Asp, Asn, N-Me-Ala, N-Me-Val and the like. The novel insect kinin analogs with the formula A are obtained with a method of modification of peptidomimetics, and the analogs are prepared with a solid-phase polypeptide synthesis method. A research proves that a target compound with the formula A has excellent insecticidal activity, particularly has a good control effect on aphids, also has better in-vitro antifungal activity for seven kinds of tested phytopathogen and has further development and application value.
Owner:CHINA AGRI UNIV
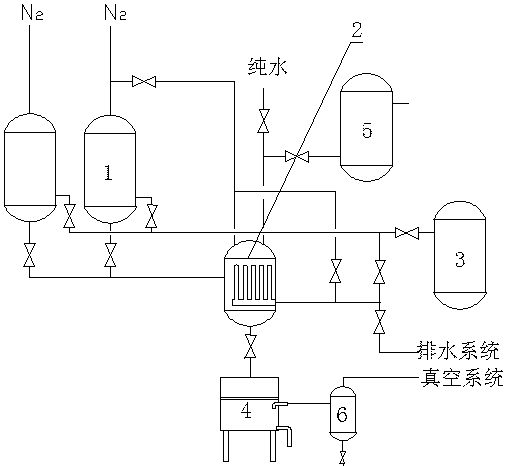
Palladium-carbon filtering and recycling system and method
PendingCN110918004AReduce lossReduce labor intensityFiltration circuitsChemical/physical processesPtru catalystFiltration
The invention discloses a palladium-carbon filtering and recycling system and method, the palladium-carbon filtering and recycling system comprises a hydrogenation reaction kettle (1), a filter (2) connected with the hydrogenation reaction kettle (1), a nitrogen back flushing mechanism, a drainage system and a cyclohexanecarboxylic acid crude product tank (3) which are connected with the filter (2), a palladium-carbon suction filter (4) connected with the filter (2), and a pure water pipe and a tail gas buffer tank (5) which are communicated with the filter (2), the palladium-carbon filteringand recycling method comprises the following steps: after the reaction is finished, replacing residual gas in the hydrogenation reaction kettle (1) with nitrogen, and conveying crude product cyclohexanecarboxylic acid obtained by the reaction to the cyclohexanecarboxylic acid crude product tank (3); when the activity of a catalyst becomes low and the catalyst cannot be continuously used, discharging filter residues from the filter (2) after the filter residues are filtered by the filter (2), starting pure water flushing until no palladium-carbon and cyclohexanecarboxylic acid residues exist ina pipeline and the filter (2), and enabling the filter residues and cleaning water to enter the palladium-carbon suction filter (4); and recycling the filter residues after vacuum filtration and dehydration through the palladium-carbon suction filter (4).
Owner:湖南双阳高科化工有限公司
Synthetic ester lubricating oil base oil with high viscosity index and preparation method thereof
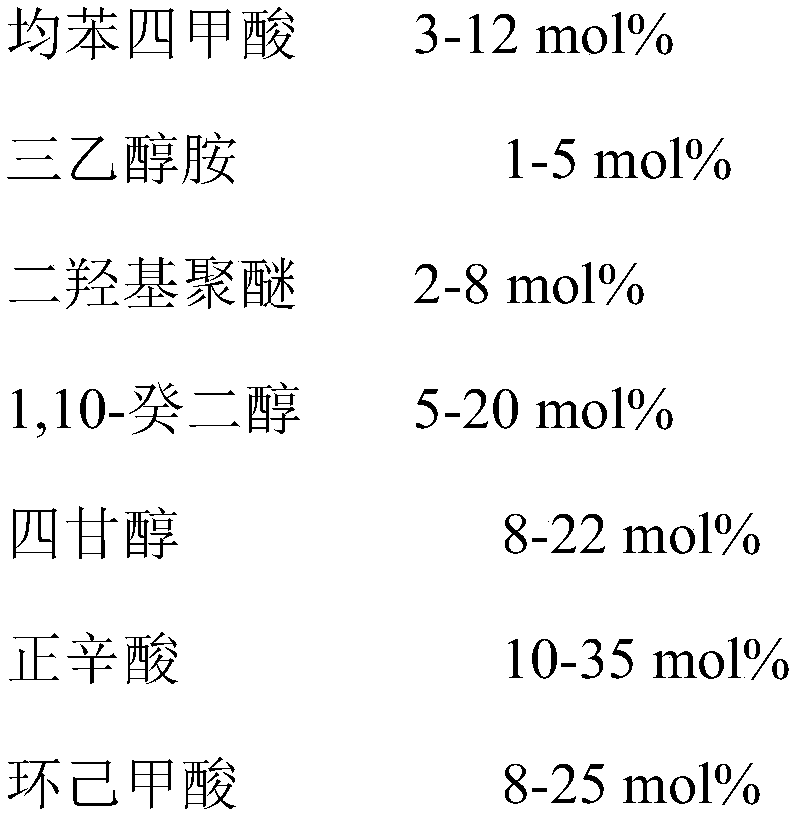
ActiveCN107686760AHigh viscosity indexMeet the use requirementsBase-materialsN-Octanoic acidOrganic solvent
The invention provides synthetic ester lubricating oil base oil with a high viscosity index and a preparation method thereof. The synthetic ester lubricating oil base oil is prepared by taking pyromellitic acid, triethanolamine, dihydroxy polyether, 1,10-decanediol, tetraethylene glycol, n-octanoic acid, cyclohexanecarboxylic acid and an organic solvent as main raw materials and carrying out sub-step esterification reaction. The synthetic ester lubricating oil base oil provided by the invention has a very high viscosity index and a relatively low pour point and the pour point can reach -45 DEGC; in a lubricating oil formula, a viscosity index modifier does not need to be additionally added and the requirements on high-viscosity lubricating oil can be met.
Owner:黄山市强力化工有限公司
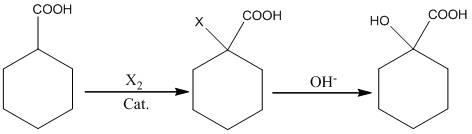
Preparation method of alpha-hydroxy-cyclohexanecarboxylic acid
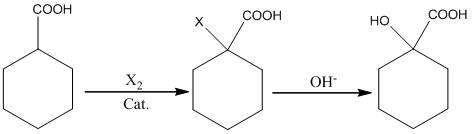
InactiveCN102675088AConvenient sourceLow priceOrganic compound preparationCarboxylic compound preparationDistillationCyclohexanecarboxylic acid
The invention relates to a synthesis process of corresponding hydroxy-cyclohexanecarboxylic acid from cycloalkyl formic acid. The method is characterized by performing halogenation on cyclohexanecarboxylic acid, subjecting the cyclohexanecarboxylic acid to hydrolization in an alkaline condition, and obtaining a corresponding hydroxy-cyclohexanecarboxylic acid product through acid neutralization, extraction and distillation. According to the preparation method, usage of highly toxic product of sodium cyanide is avoided and the synthesis process of hydroxy-cyclohexanecarboxylic acid is suitable for industrial application.
Owner:WENZHOU IND SCI RES INST
Process for the preparation of trans-4-amino-1-cyclohexanecarboxilic acid and its derivatives
ActiveCN108602758AOrganic compound preparationAmino-carboxyl compound preparationHydrogen pressureAcid derivative
The present invention relates to a one-pot process for the preparation of trans-4-amino-l-cyclohexanecarboxylic acid derivatives where the trans ratio is more than 75% by reaction of a 4-aminobenzoicacid derivative using an appropriate catayst and an appropriate solvent or solvent mixture under basic conditions. The process uses low hydrogen pressure and is therefore suitable for industrial application.
Owner:SIEGFRIED AG
Method and equipment for separating cyclohexanecarboxylic acid slurry
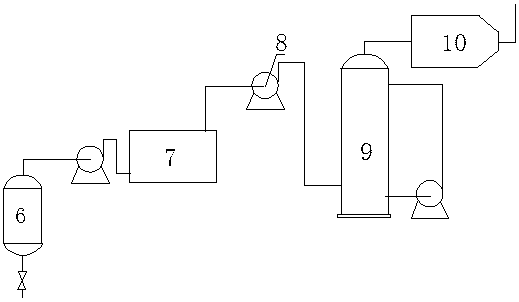
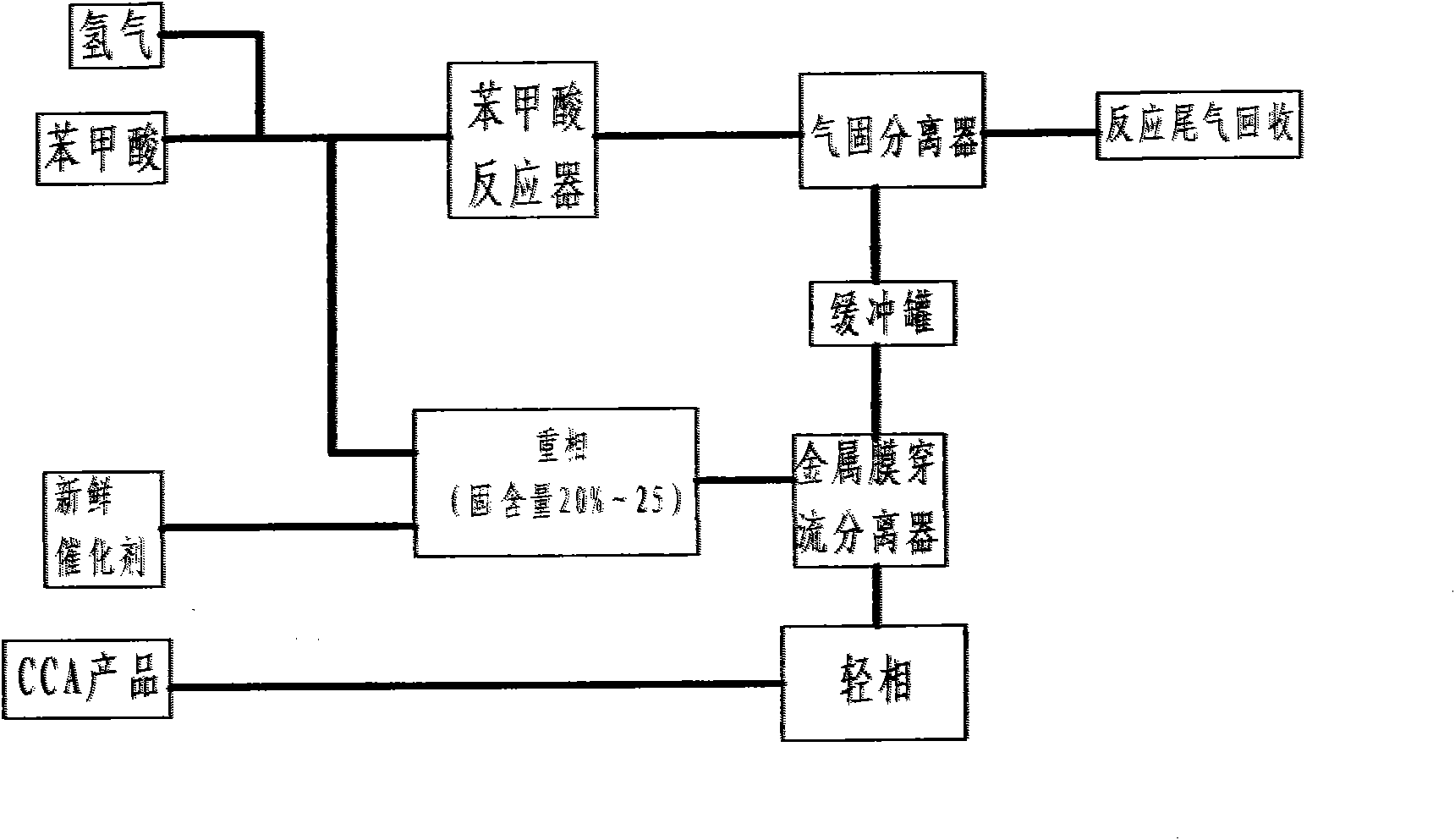
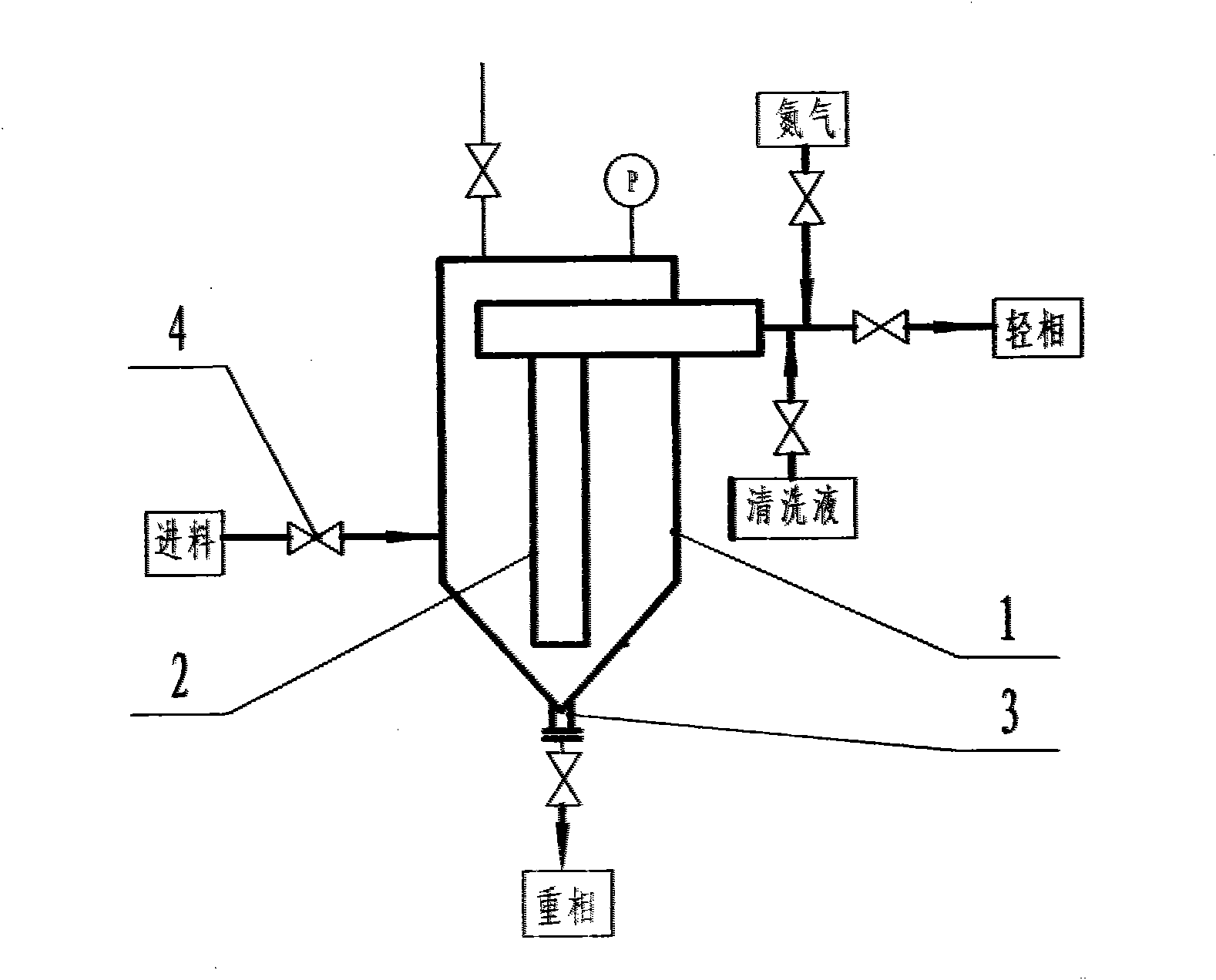
InactiveCN102050726AWon't wear outAvoid running damageCarboxylic preparation by ozone oxidationStationary filtering element filtersPalladium on carbonBuffer tank
The invention discloses a method and equipment for separating cyclohexanecarboxylic acid (CCA) slurry. The equipment mainly comprises a metal film penetrating separator; and a film pipe is arranged in the separator, and is provided with a stainless steel metal film with the aperture of 0.50 to 50 mu m. The method comprises the following steps of: introducing palladium-carbon catalyst-containing reaction mixed solution into an enclosed separator for separation through a buffer tank; and directly discharging the separated light phase into the next production procedure, concentrating the heavy phase in the separator, stopping feeding when the catalyst concentration reaches 10-30 weight percent, performing nitrogen stirring and clear liquid backwashing, and returning to a reactor. The separated light phase CCA product has the purity of 98-99 percent, and the catalyst content of zero. The separating process is automatically continuously carried out, power equipment is not required, the catalyst is not worn, the consumption of the catalyst is low, and the service life of the catalyst is long. The separating equipment is easy to operate, convenient to maintain, safe, environment-friendly and pollution-free.
Owner:CHINA PETROLEUM & CHEM CORP +2