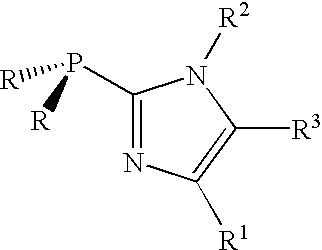
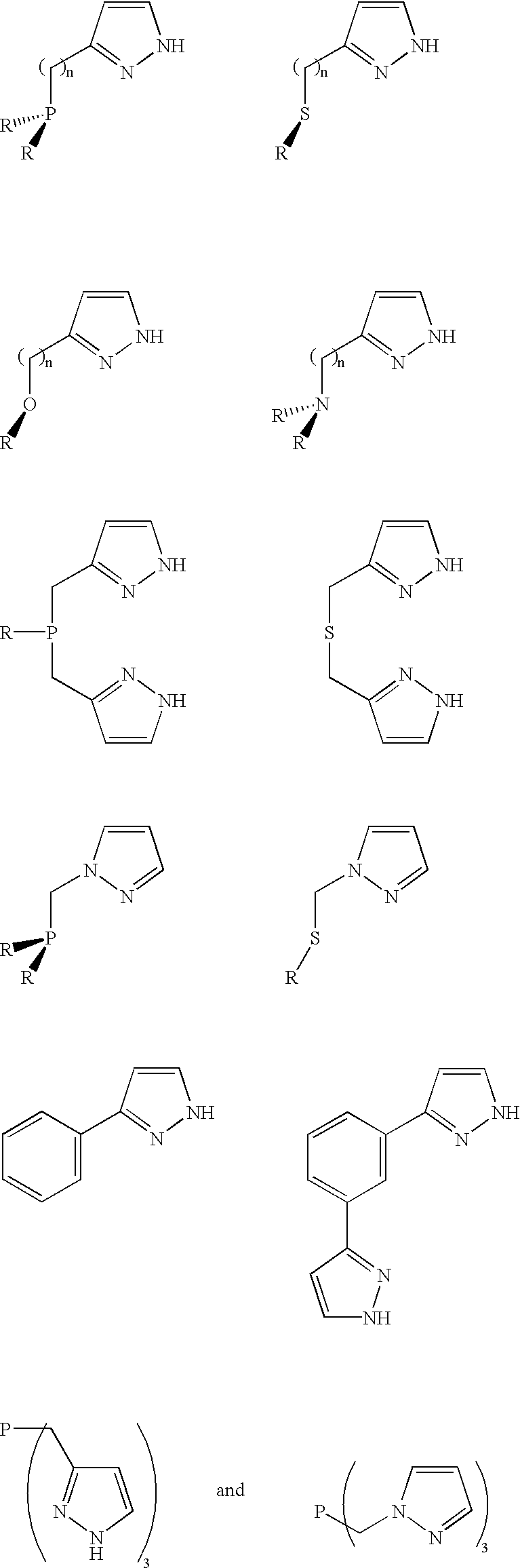
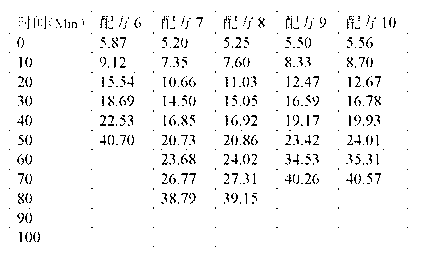Patents
Literature
104 results about "Imidazole ligand" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
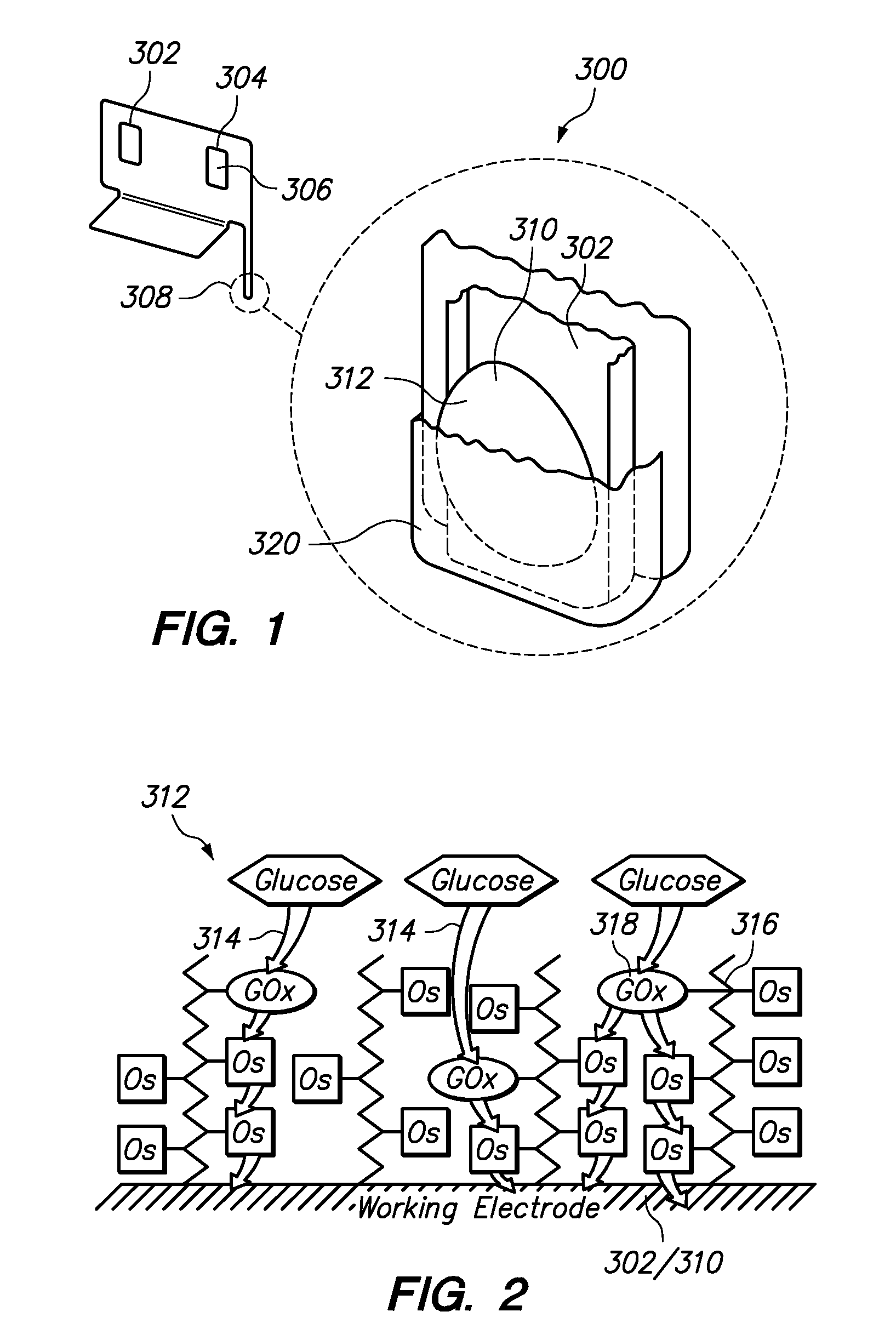
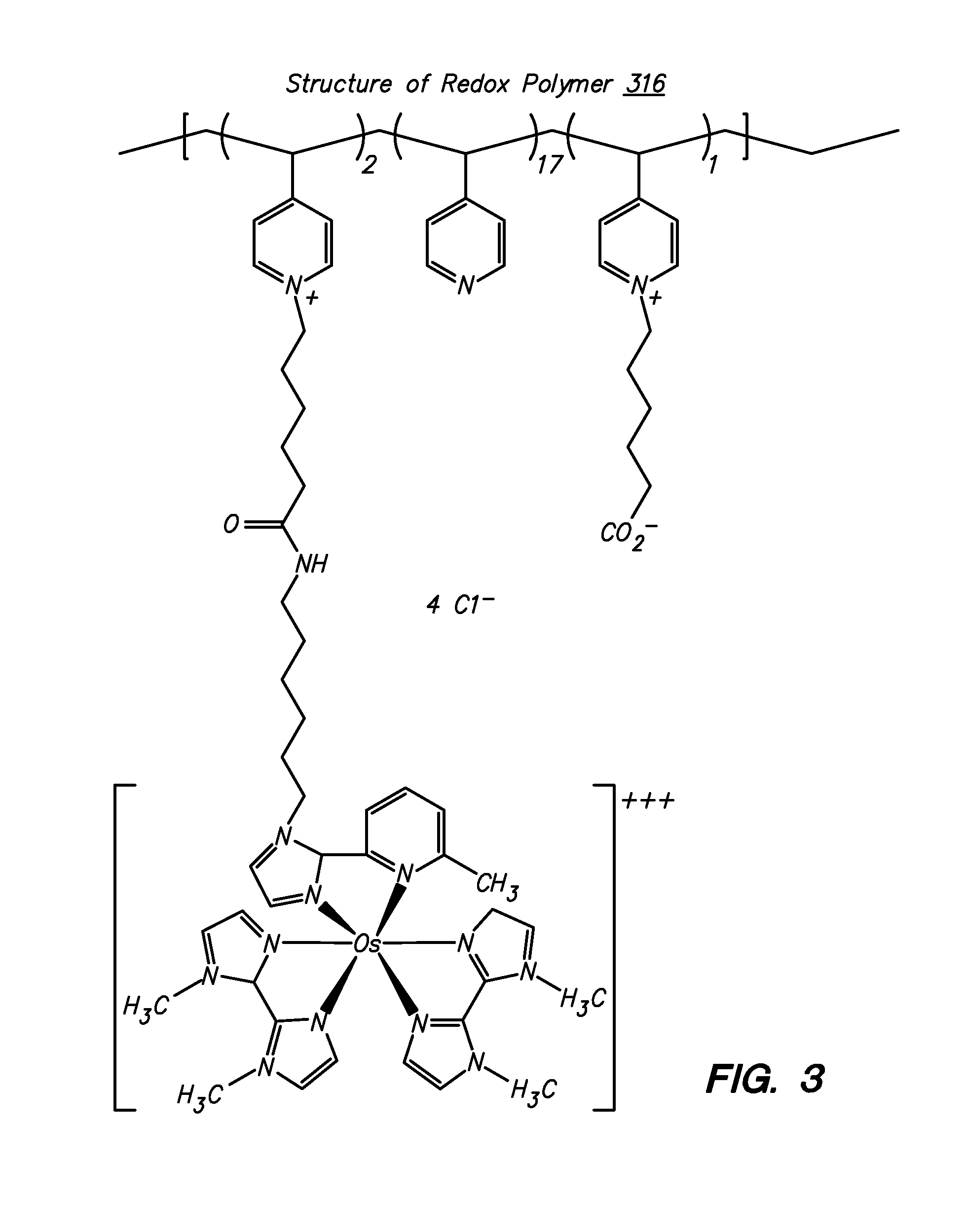
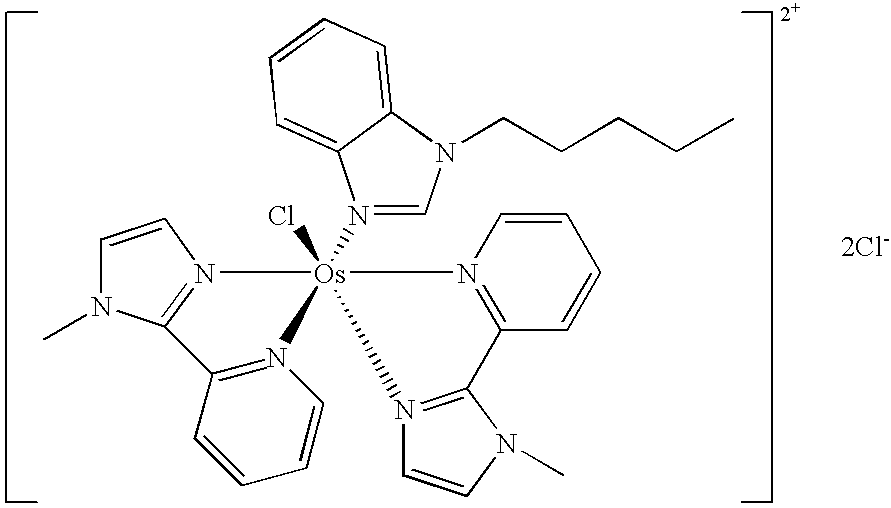
Transition metal complexes with (pyridyl)imidazole ligands
InactiveUS7074308B2Rapid electron exchangeFast dynamicsImmobilised enzymesBioreactor/fermenter combinationsOxidation-Reduction AgentRedox
Novel transition metal complexes of iron, cobalt, ruthenium, osmium, and vanadium are described. The transition metal complexes can be used as redox mediators in enzyme-based electrochemical sensors. The transition metal complexes include substituted or unsubstituted (pyridyl)imidazole ligands. Transition metal complexes attached to polymeric backbones are also described.
Owner:ABBOTT DIABETES CARE INC
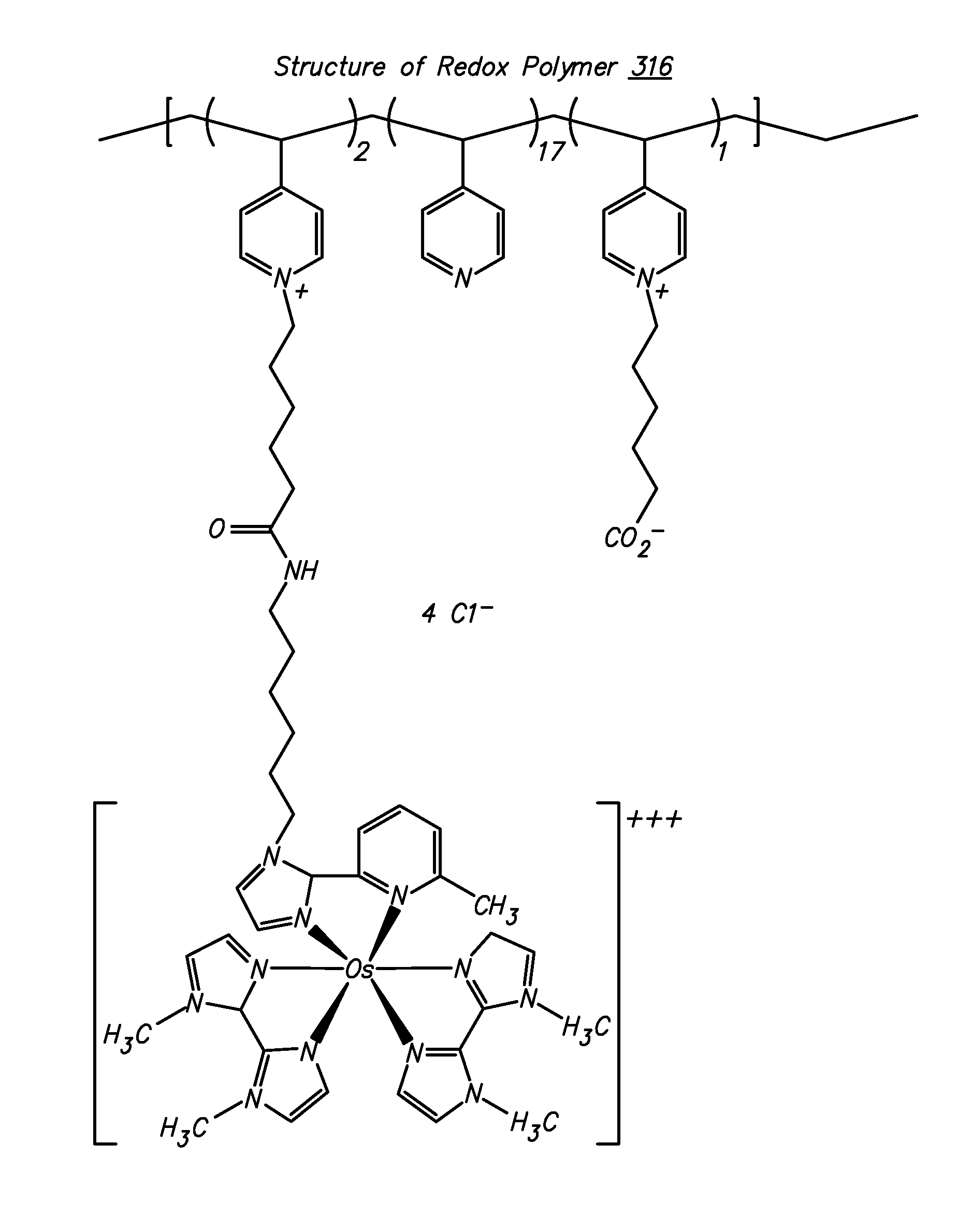
Redox polymers for use in analyte monitoring
InactiveUS8444834B2Immobilised enzymesBioreactor/fermenter combinationsHigh rateElectrochemical biosensor
Polymers for use as redox mediators in electrochemical biosensors are described. The transition metal complexes attached to polymeric backbones can be used as redox mediators in enzyme based electrochemical sensors. In such instances, transition metal complexes accept electrons from, or transfer electrons to, enzymes at a high rate and also exchange electrons rapidly with the sensor. The transition metal complexes include at least one substituted or unsubstituted biimidazole ligand and may further include a second substituted or unsubstituted biimidazole ligand or a substituted or unsubstituted bipyridine or pyridylimidazole ligand.
Owner:ABBOTT DIABETES CARE INC
Preparation method of nanometer-to-micrometer scale zeolitic imidazolate frameworks (ZIFs)
ActiveCN102731538AGood lookingPromotes deprotonationCobalt organic compoundsZinc organic compoundsOrganic fluidSolvent
The invention which belongs to the technical field of new materials relates to a synthetic method of ZIFs, and concretely discloses a method for synthesizing nanometer / micrometer scale ZIFs based on imidazole and its derivatives at room temperature in a concise, efficient and green manner. The method comprises the following steps: dissolving a single imidazole ligand or a mixture of two imidazole ligands in water or an organic liquid to form a solution, adding an alkaline substance to adjust the pH value of the solution, adding a metal salt to form a reaction system, and reacting to obtain the nanometer or micrometer scale ZIFs. The method which allows routine reactants to be utilized and the alkaline substance to be added to adjust the pH value of the system has the advantages of ligand application amount reduction, controllable product size, and high purity of the nanometer / micrometer scale ZIFs. The synthetic method has the characteristics of cheap and easily available raw materials and solvent, and no pollution, and has important meanings to the production and the application of the ZIFs.
Owner:CHINA UNIV OF PETROLEUM (EAST CHINA)
Calcium zinc heat stabilizer used for PVC, zinc-containing compound and application
ActiveCN103183690AHigh activityStable unstable chlorineZinc organic compoundsCompounds of zincZinc ion
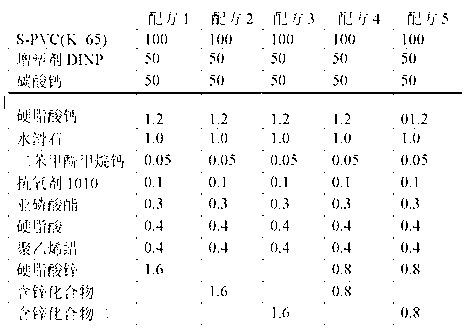
The invention relates to a calcium zinc heat stabilizer used for PVC. The calcium zinc heat stabilizer comprises a compound formed by a hydroxyl phenyl carboxylic acid, imidazole ligands and zinc ions, wherein the molecular formula of the compound is Zn(L1)2(L2)2, wherein L1 represents salicylic acid; L2 represents imidazole or L2 represents 1,2-dimethyl imidazole; a frequently-used fatty acid calcium, fatty acid zinc, an auxiliary stabilizer and a lubricating agent. Compared with a calcium zinc heat stabilizer prepared by using the fatty acid zinc and the like as raw materials, the calcium zinc heat stabilizer provided by the invention can effectively improve initial dyeing property and prolong zinc burnt blackening time of a PVC product, has easily available raw materials and is convenient for preparation.
Owner:SHENZHEN AIMSEA IND
Double-shell hollow spherical organic metal framework material and preparation method thereof
InactiveCN101805361ALarge specific surface areaGood chemical stabilityZinc organic compoundsMetal frameworkGranularity
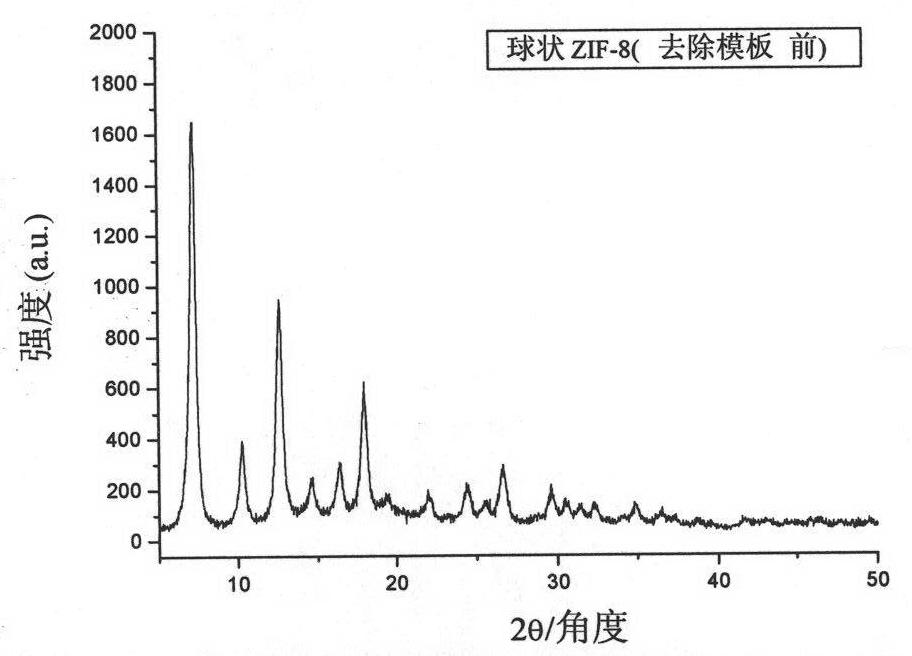
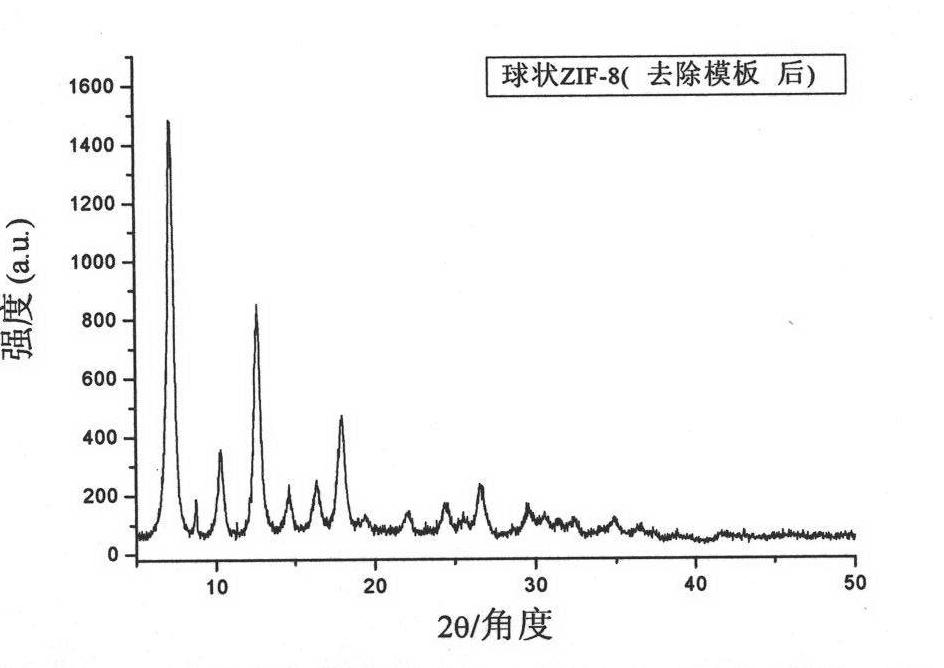
The invention belongs to an organic metal framework material, and particularly relates to a double-shell hollow spherical organic metal framework material (HS-ZIF-8) and a preparation method thereof. The material is of spherical particles, the granularity is 300 to 500 nm, and the material is provided with a double-layer hollow shell structure. The preparation method comprises the following stepsthat: sulfonated polystyrene pellets are scattered into methanol aqueous solution to be undertaken the ultrasonic treatment and be mixed for 5 to 15 minutes; zinc salt methanol solution is added and mixed; imidazole ligand is added; the mixed solution is mixed for 1 hour, held, centrifuged, washed by methanol and dried for 12 hours at the temperature of 40 DEG C to obtain white solid; under the protection of N2, the white solid is burned for 2 to 4 hours at the temperature of 300 to 400 DEG C to obtain the double-shell hollow spherical organic metal framework compound material. The double-shell hollow spherical organic metal framework compound material has the advantages that: the material has unique appearance feature, large specific surface area and high chemical and thermal stability, and has promising prospects in the fields such as gas storage, separation, sensor technology, nano parts and the like.
Owner:SHANGHAI NORMAL UNIVERSITY
Zeolitic imidazolate frameworks for kinetic separation of propane and propene
InactiveUS20110282067A1Different rate of diffusionDifferent relative diffusion rateGas treatmentDispersed particle separationPropaneImidazole ligand
Zeolitic Imidazolate Frameworks (ZIFs) characterized by organic ligands consisting of imidazole ligands that are either essentially all 2-chloroimidazole ligands or essentially all 2-bromoimidazole ligands are disclosed. Methods for separating propane and propene with the ZIFs of the present invention, as well as other ZIFs, are also disclosed.
Owner:RUTGERS THE STATE UNIV
Metallic ion-polyvinyl imidazol complex catalyst and preparation method and application thereof
InactiveCN101544755AIncrease local concentrationSolve the problem of low catalytic efficiencyEthylene HomopolymersWater soluble
The invention discloses a metallic ion-polyvinyl imidazole complex catalyst, which is a complex of a metallic ion and a polyvinyl imidazole ligand, wherein the molar ratio of imidazole groups to the metallic ion is 0.5 to 300; the metallic ion is a copper ion or manganese ion; and the polyvinyl imidazole ligand is a homopolymer of a N-vinyl imidazole monomer or copolymer of the N-vinyl imidazole monomer and another one or various water-soluble monomers and has a weight average molecular weight of 10<3> to 10<7>. The polyvinyl imidazole ligand has a large number of imidazole groups and can significantly increase the local concentration of the catalyst in a reaction medium after the complexation with the metallic ion, so the catalyst has greatly improved catalytic efficiency, achieves high PPO yield with a very small dosage when used in the preparation of PPO in a water medium, and solves the problem of the low catalytic efficiency of the prior polyphenylether catalyst prepared in the water medium.
Owner:ZHEJIANG UNIV
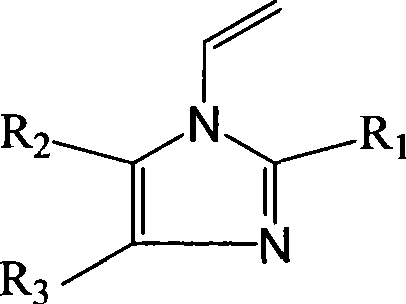
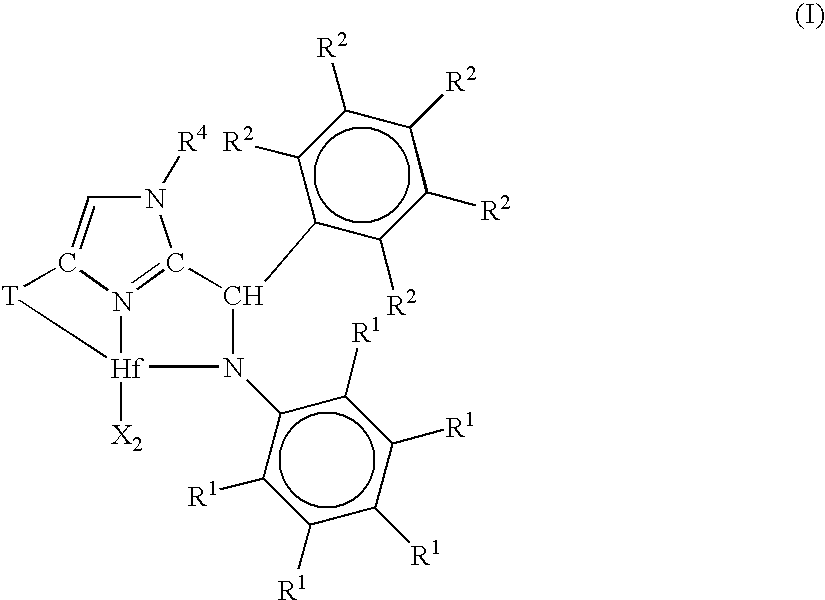
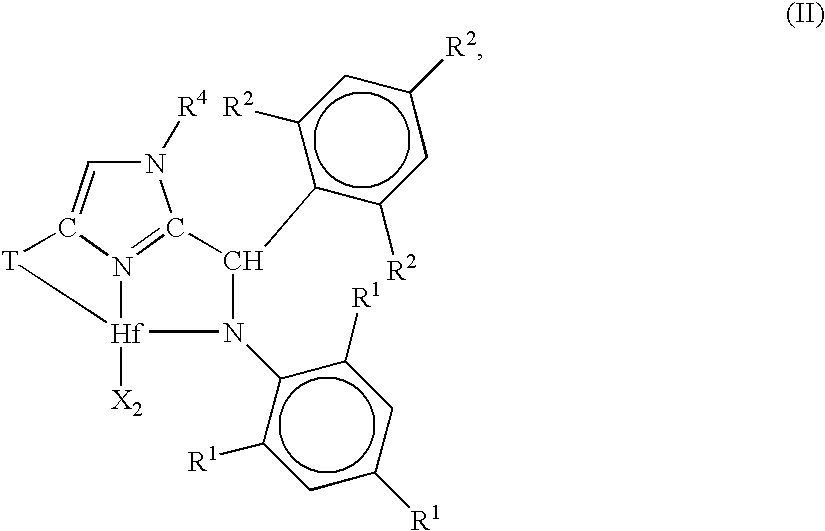
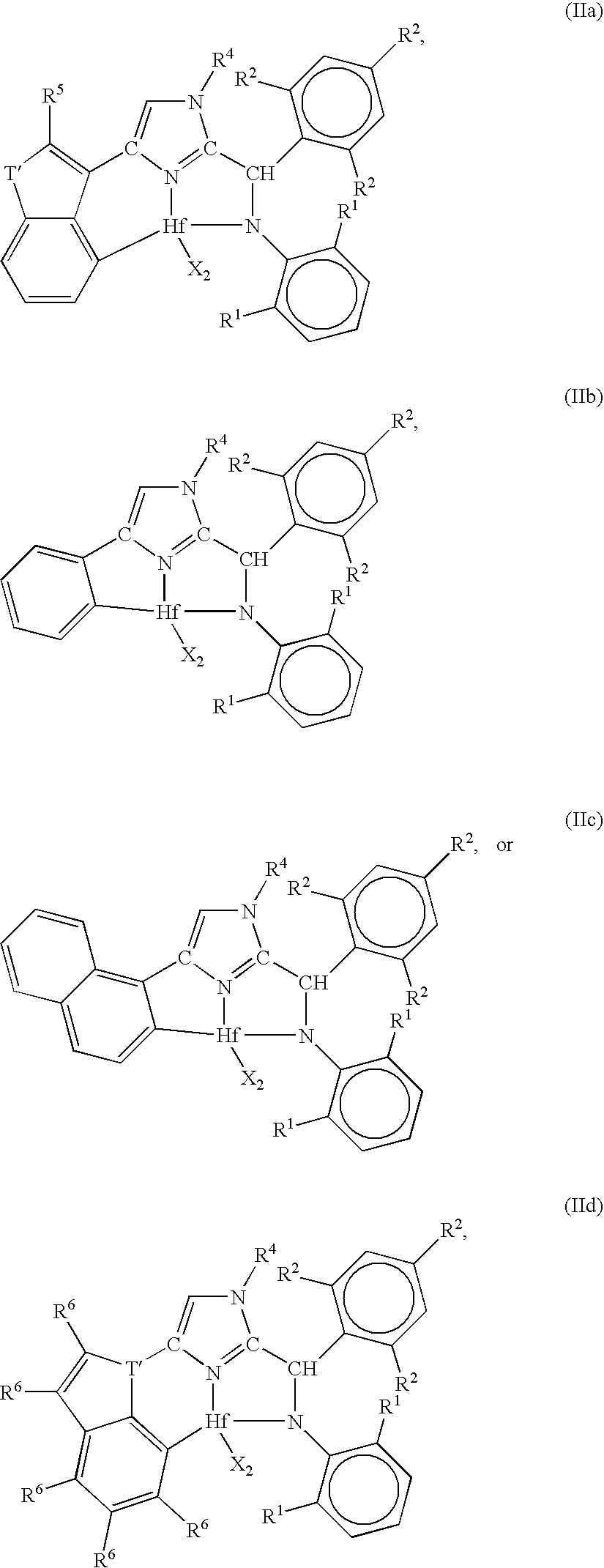
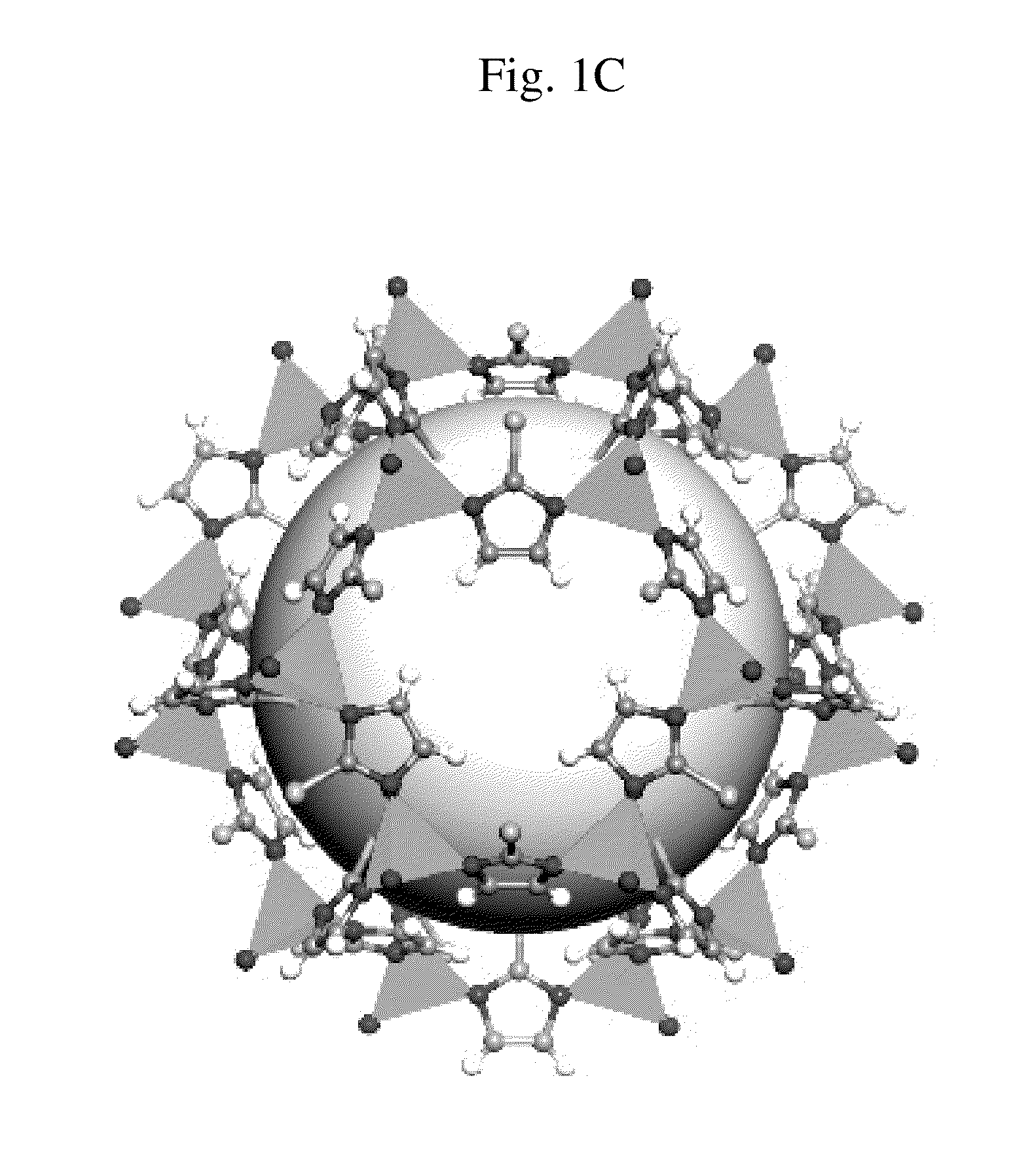
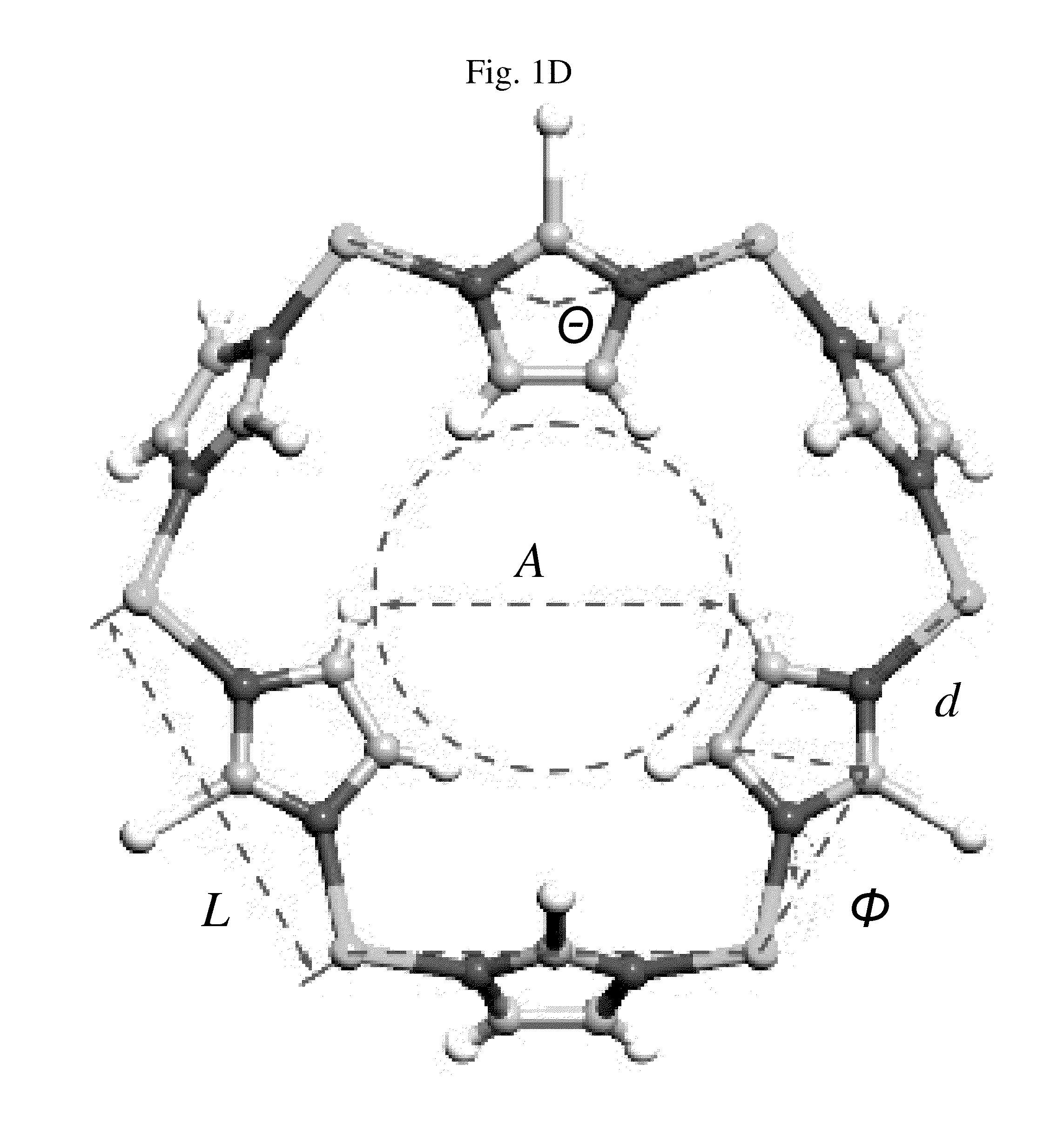
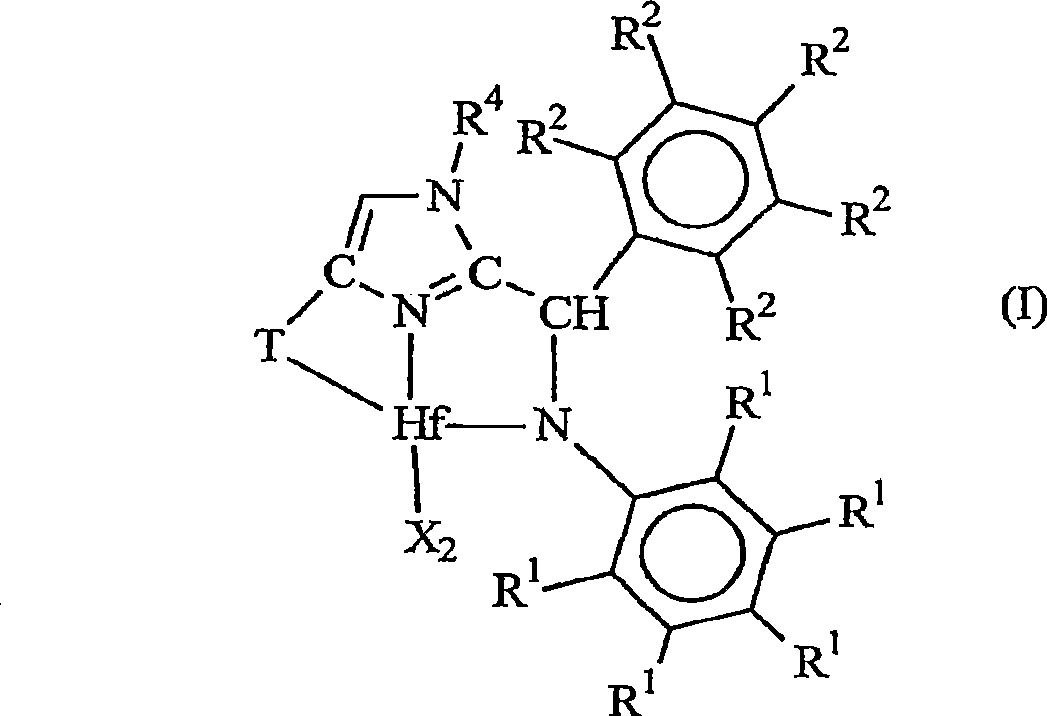
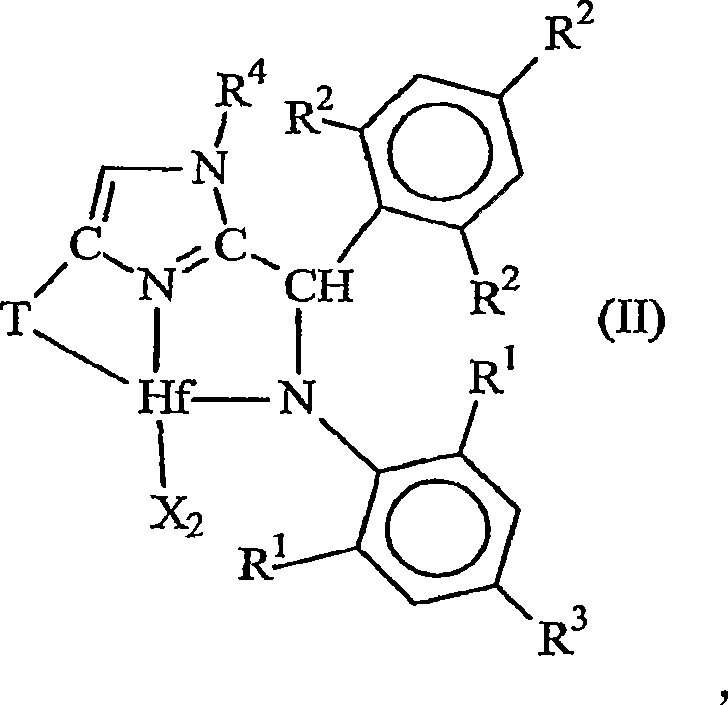
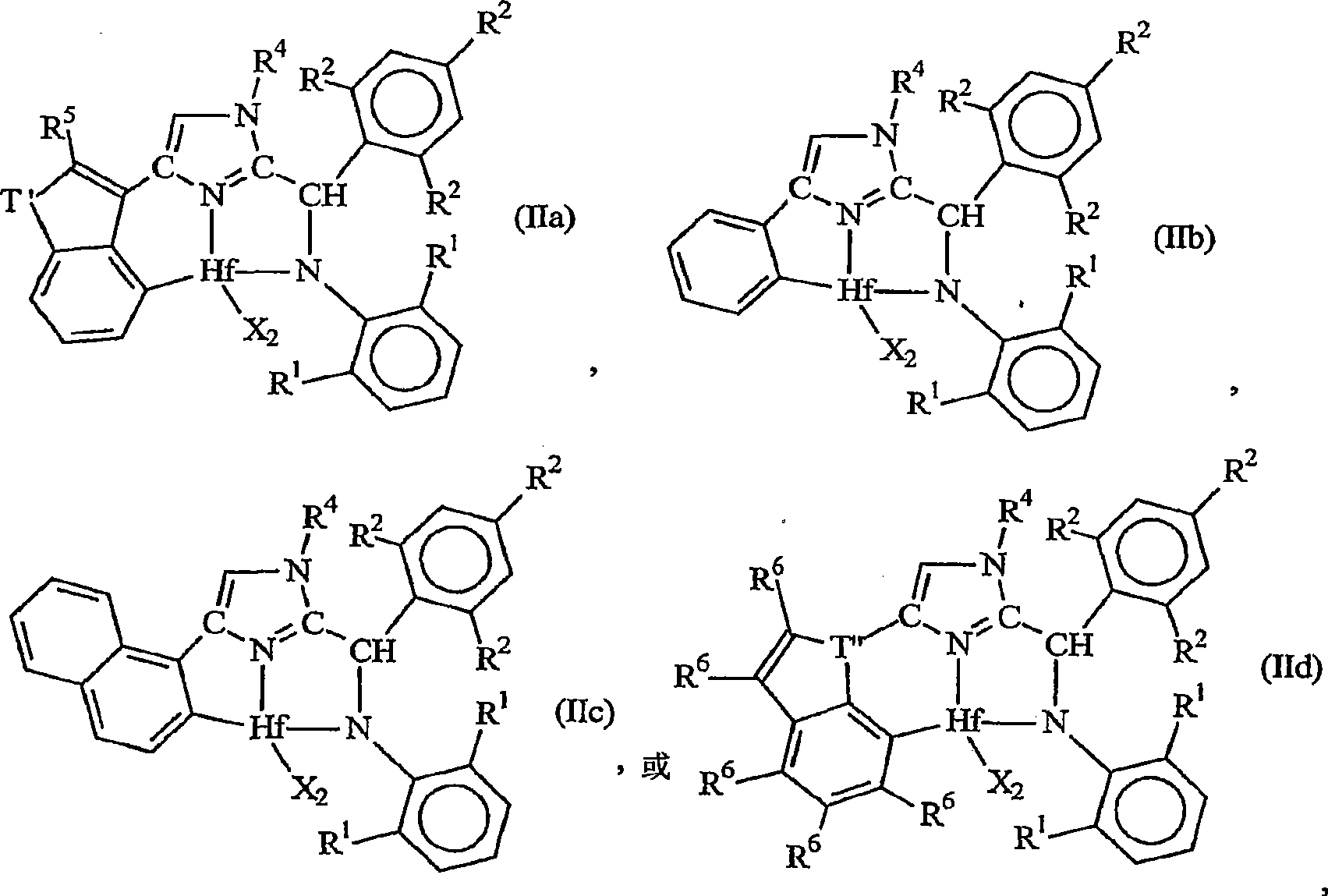
Ortho-metallated hafnium complexes of imidazole ligands
InactiveUS20090306318A1High molecular weightHigh catalytic efficiencyGroup 4/14 element organic compoundsGroup 8/9/10/18 element organic compoundsMetalationHafnium
Hafnium complexes of heterocyclic organic imidazol-2-yl ligands containing internal orthometallation and their use as components of olefin polymerization catalyst compositions, especially supported catalyst compositions, are disclosed.
Owner:DOW GLOBAL TECH LLC
Preparation method of high-corrosion-resistance modified epoxy resin composite coating
ActiveCN112341900AImprove mechanical propertiesAccelerated corrosionMaterial nanotechnologyAnti-corrosive paintsPolymer scienceWater methanol
The invention discloses a preparation method of a high-corrosion-resistance modified epoxy resin composite coating, which comprises the following steps: dissolving a metal salt and an imidazole ligandin absolute methanol, conducting stirring for 2-3 hours to obtain a white emulsion, carrying out centrifugal separation to obtain a white solid, alternately conducting washing with methanol and deionized water for 3-5 times, and carrying out vacuum drying at 60-80 DEG C to obtain a white powdery MOF nanometer material; mixing epoxy resin, the MOF nano material and a diluent, carrying out ball milling and stirring for 0.5-5 h, adding a curing agent, and conducting uniform mixing to obtain modified epoxy resin; coating the surface of a pretreated metal matrix with the modified epoxy resin, andconducting drying and curing to obtain the MOF modified epoxy resin composite coating on the surface of the metal matrix. According to the method, the epoxy resin is modified by the MOF nano material,so that the MOF nano material and the epoxy resin are chemically bonded, the problem of interfacial compatibility between the filler and the epoxy resin is solved, and the corrosion resistance and mechanical properties of the epoxy resin coating are improved.
Owner:SICHUAN UNIVERSITY OF SCIENCE AND ENGINEERING
Preparation method and application of heteroatom-doped metal carbon-containing material
InactiveCN107200346AMany active sitesLarge specific surface areaHybrid capacitor electrodesIron compoundsGas phaseOxygen
The invention relates to a preparation method and application of a heteroatom-doped metal carbon-containing material. The preparation method comprises the following steps: dissolving metal ions, an imidazole ligand compound and a water-soluble high molecular polymer into a solvent A respectively; adding a graphene oxide solution after a period of time; mixing and stirring; carrying out solid-liquid separation and calcining obtained sediment under the protection of inert gas to obtain a nitrogen-doped metal carbon-containing material; doping the nitrogen-doped metal carbon-containing material with other heteroatoms under a chemical vapor deposition condition; carrying out high-temperature calcining under inert protection gas and low pressure so as to obtain the heteroatom-doped metal carbon-containing material. The material provided by the invention can be used as an electrode material and is used for heavy metal detection, organic matter detection, active carbon detection or oxygen reduction reaction and the like.
Owner:北京至心安科技股份有限公司
Volatile Imidazoles and Group 2 Imidazole Based Metal Precursors
Sterically hindered imidazole ligands are described, along with their synthesis, which are capable of coordinating to Group 2 metals, such as: calcium, magnesium, strontium, in an eta-5 coordination mode which permits the formation of monomeric or dimeric volatile complexes.A compound comprising one or more polysubstituted imidazolate anions coordinated to a metal selected from the group consisting of barium, strontium, magnesium, radium or calcium or mixtures thereof. Alternatively, one anion can be substituted with and a second non-imidazolate anion.Synthesis of the novel compounds and their use to form BST films is also contemplated
Owner:VERSUM MATERIALS US LLC
Supported catalyst with surface active effect and application thereof in preparing polyphenyl ether in oil-water two-phase system
The invention discloses a supported catalyst with a surface active effect. The supported catalyst is a complex of imidazole ligands and metal ions, and the complex is grafted in nano-silica particlessubjected to surface modification, wherein the nano-silica particles subjected to surface modification are nano silicon dioxide particles which are grafted on the surface of a silane coupling agent subjected to hydrophobic treatment and the surfaces of polyvinyl imidazole ligands with a silane coupling agent at one end. The catalyst is high in catalytic efficiency and good in product selectivity,and can catalyze an oxidative coupling polymerization reaction of phenol monomers is catalyzed in an oil-water two-phase system; after the reaction is finished, the catalyst and a surfactant can be recycled through centrifugation or filtration separation, so that greenness and environment friendliness are achieved, and the catalyst meets the requirements of sustainable development better.
Owner:DALIAN INST OF CHEM PHYSICS CHINESE ACAD OF SCI
Semi-rigid double-imidazole organic-ligand-based transition metal compound containing N-S dicarboxylic acid, as well as synthetic method and application thereof
InactiveCN103408600ALow costAdjustable gear spacingOrganic-compounds/hydrides/coordination-complexes catalystsCobalt organic compoundsBulk crystalElectrical energy consumption
The molecular formula of a semi-rigid double-imidazole organic-ligand-based transition metal compound containing N-S dicarboxylic acid is [Co2 (L) 2 (2-CMSN) 2 (H2O)]. H2O; [Cd (L) (2-CMSN) (H2O)]; [Zn (L) (2-CMSN)]. 2 H2O; [Cd (L) (ADTZ) (H2O)]; [Zn (L) (ADTZ)]. H2O, wherein L represents 1, 4-bis ( imidazol-1-methylene ) benzene, 2-CMSN represents 2-carboxylic acid methylene group root, and ADTZ represents 2,2'-(1,3,4-thiadiazol oxazol-2,5-diyl) dithio diethyl dianion root. A synthetic method of the transition metal compound comprises the following steps: adding Co<2+> or Zn<2+> and Cd<2+> nitrate, semi-rigid double-imidazole ligands, dicarboxylic acid containing N and S into deionized water, stirring, using NaOH solution to adjust pH, pouring the mixture into a high-pressure reaction kettle, increasing the temperature to realize hydrothermal reaction, cooling to obtain a bulk crystal, and washing and drying the bulk crystal to obtain the target product. The synthetic method of the compound is simple, the synthesis productivity is high, the cost of synthetic raw materials is low, the synthetic period is short and the electrical energy consumption is low; the compound is high in affiliation capacity of water-soluble pollutants, good in catalysis and degradation effects and environmental-friendly, and can be used as photocatalytic materials.
Owner:BOHAI UNIV
Preparation method of Co3S4 nanosheet array modified by CeO2 nanoparticles
ActiveCN112164592AKeep porousRetain multiple active sitesHybrid capacitor electrodesHybrid/EDL manufactureCapacitanceNickel substrate
The invention provides a preparation method of a Co3S4 nanosheet array modified by CeO2 nanoparticles. The method comprises the following steps: mixing a cobalt salt, an imidazole ligand and a solvent, adding foamed nickel, and growing a zeolite imidazolate framework CoZIFL nanosheet array on a foamed nickel substrate through a reaction; then, adding the CoZIFL nanosheet array into a sulfur sourcesolution, and generating a Co3S4 nanosheet array through hydrothermal vulcanization; and adding cerium salt, a precipitator and a solvent into the precursor, and obtaining a CeO2 nanoparticle modified Co3S4 nanosheet array through solvothermal reaction. The CeO2 nanoparticle modified Co3S4 nanosheet array prepared by adopting the method can be directly used as an electrode of a supercapacitor, and compared with a Co3S4 nanosheet array precursor, the specific capacitance of the CeO2 nanoparticle modified Co3S4 nanosheet array prepared by adopting the method is improved by 114.8% (under the condition of 0.5 A / g).
Owner:宁波市大学科技园发展有限公司
Room temperature water-phase synthesis method for zeolite imidazole ester framework material
InactiveCN102898448AThe process steps are simpleHigh yieldCobalt organic compoundsZinc organic compoundsSurface-active agentsRaw material
The invention discloses a room temperature water-phase synthesis method for zeolite imidazole ester framework material, which has the advantages of being low in cost, high in reaction yield, mild in reaction conditions, low in price of raw materials, easy in obtaining of the raw materials as well as simple and clean in technology, and can be used for rapidly preparing the zeolite imidazole ester framework material. The room temperature water-phase synthesis method for the zeolite imidazole ester framework material comprises the following steps of: evenly mixing imidazole ligand and metal compound according to the chemical formula stoichiometric ratio of target coordination framework material with alkaline aqueous solution containing three-block non-ionic surface active agent at the room temperature, filtering or carrying out centrifugal separation, and drying to obtain the zeolite imidazole ester framework material.
Owner:NANJING FORESTRY UNIV
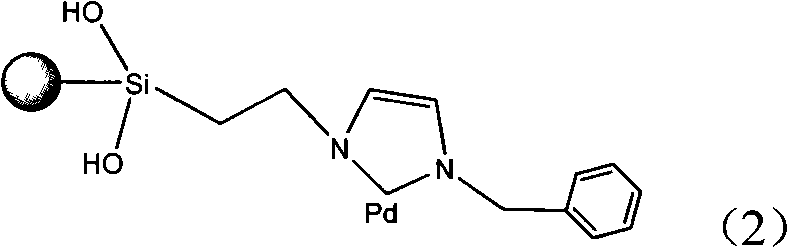
Method for degradation of organic halide in water with immobilized metallic catalyst
InactiveCN101314495AEliminate pollutionLow toxicityOrganic-compounds/hydrides/coordination-complexes catalystsEnergy based wastewater treatmentCatalyst degradationHydrogen
The invention provides a method for degrading organic halides in water by a metal-supported catalyst, comprising an imidazole ligand metal (palladium or nickel) catalyst supported on silica gel. The invention is characterized in that the method comprises the following steps that: the organic halides in the water are subject to the heterogeneous catalytic reduction reaction by the catalyst under the condition that a hydrogen donor is added at a temperature of between 10 and 90 DEG C, under normal pressure for 2 to 10 hours, thereby effectively degrading the organic halides in the water. Experiments show that the method catalyzes the organic halides in the water, such as bromobenzene, polychlorobiphenyl and so on, under a mild condition with a removal rate reaching over 90 percent.
Owner:KUNMING UNIV OF SCI & TECH
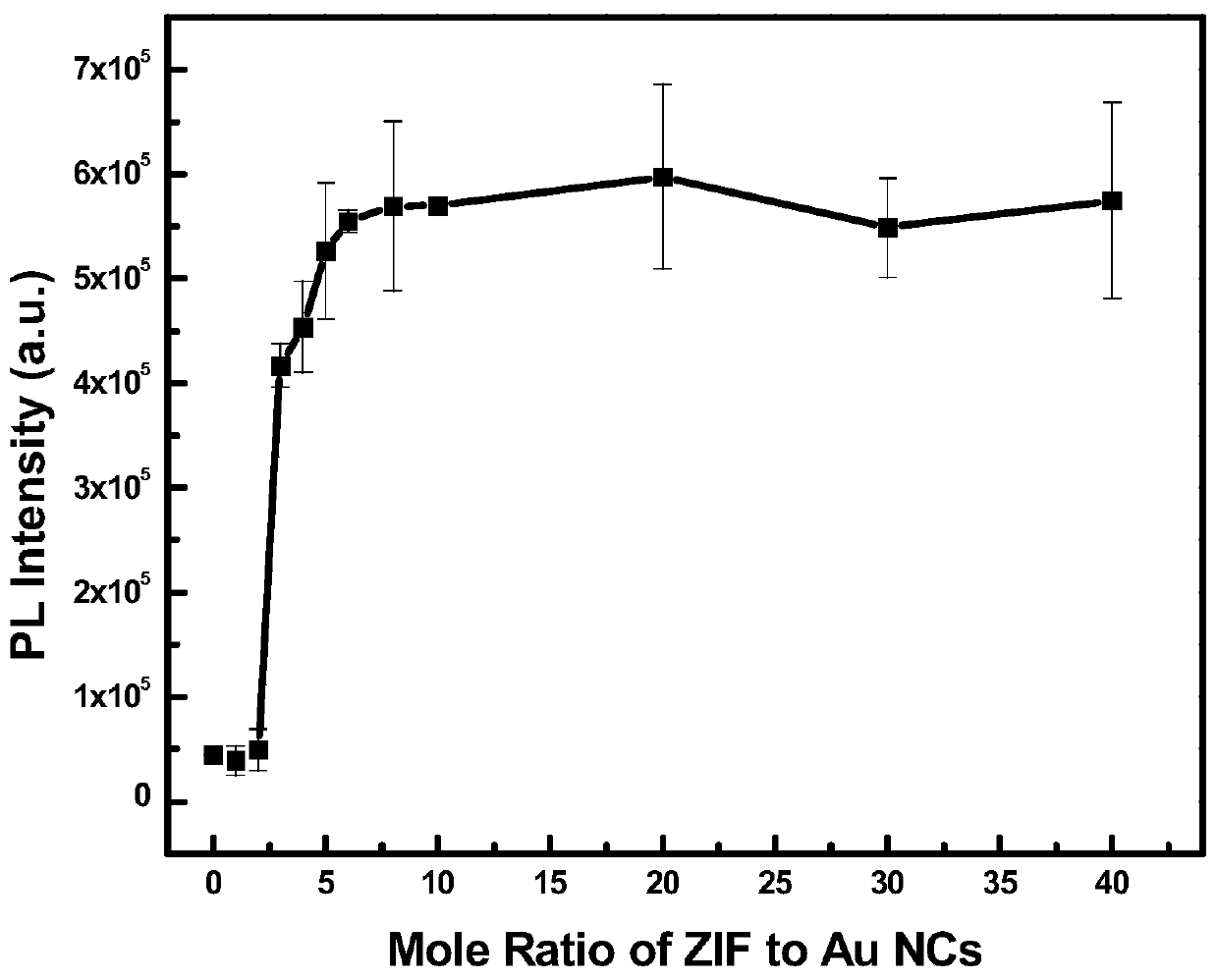
Preparation method of fluorescent reinforced metal nano cluster/zeolite imidazate framework composite nano material and application thereof
ActiveCN110628042ASimple processMild reaction conditionsMaterial nanotechnologyFluorescence/phosphorescenceLight emissionIon
The invention belongs to the technical field of fluorescent materials and in particular relates to a preparation method of a fluorescent reinforced metal nano cluster / zeolite imidazate framework composite nano material. The preparation method comprises the following steps: enabling a zeolite imidazate framework material and a metal nano cluster liquid to react under conditions ultrasonic waves, stirring or standing, and on the basis of competition coordination / electrostatic interactions of a surface ligand of metal nano clusters with a metal ion / imidazole ligand of the zeolite imidazate framework material, performing reassembling, so as to obtain a metal nano cluster / zeolite imidazate framework composite nano material. The invention is based on an aggregation-induced emission principle anda quantum confinement effect, the light emission performance of the metal nano clusters of the formed metal nano cluster / zeolite imidazate framework composite nano material is greatly improved, and multiple properties of a zeolite imidazate framework can be stilled maintained on the basis. The method is simple in process, mild in reaction condition, low in energy consumption, good in universalityand easy in on-scale popularization and application.
Owner:NORTHWESTERN POLYTECHNICAL UNIV +1
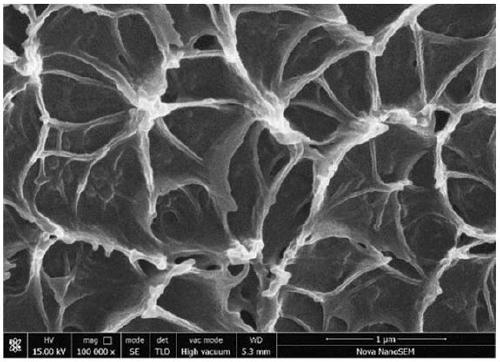
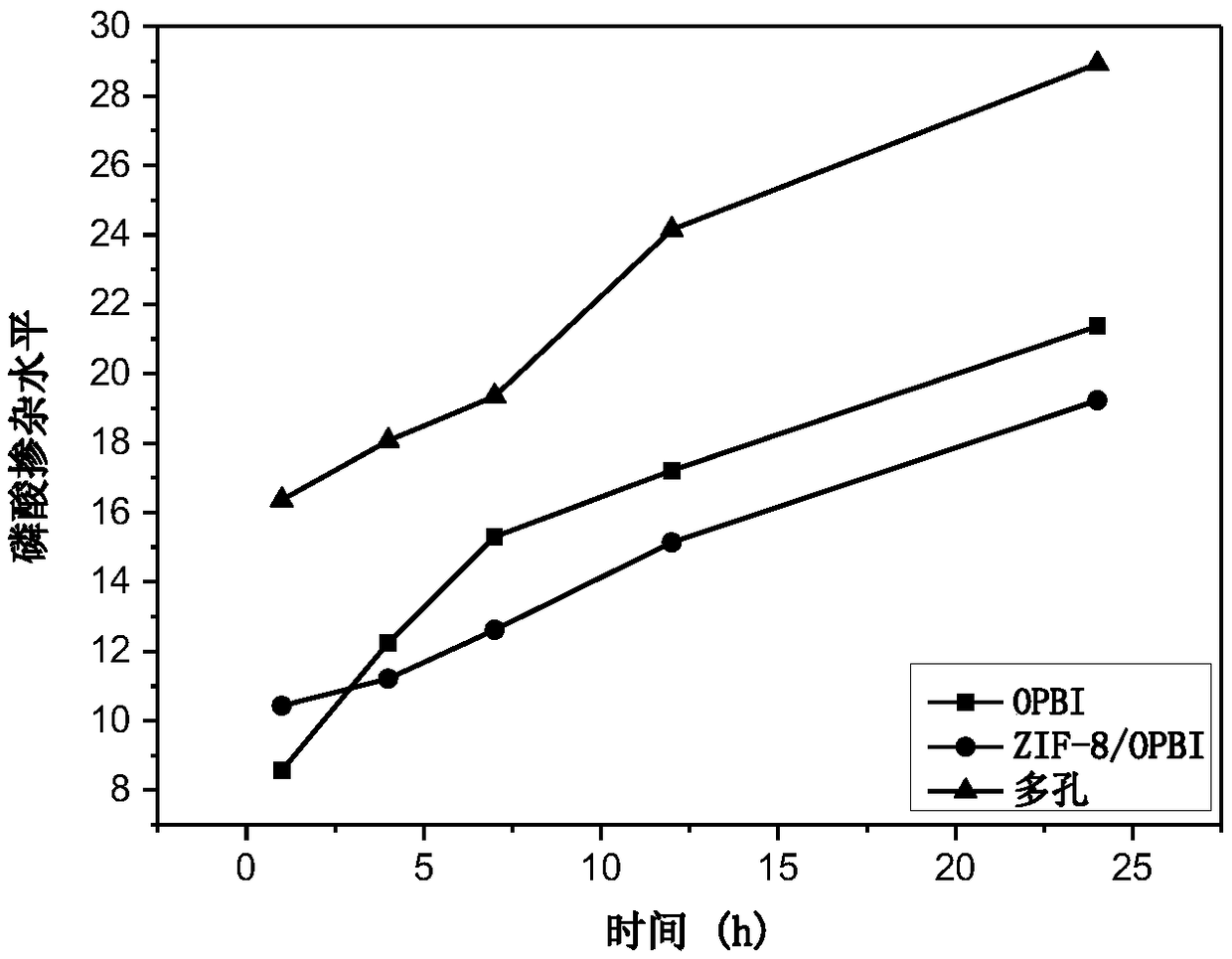
Polybenzimidazole film with nanometer porous structure and preparation method thereof
The invention relates to a polybenzimidazole film with a nanometer porous structure and a preparation method thereof and belongs to the technical field of special macromolecular functional films. Thepreparation method comprises the following steps: adding filler dispersion liquid into a polybenzimidazole solution, fully mixing and filtering, and then pouring onto a clean glass plate; heating andremoving solvent, thereby acquiring a polybenzimidazole film on the glass plate; soaking for a period of time in an etching agent; washing with boiled water and removing residual etching agent; drying, thereby acquiring the polybenzimidazole film with the nanometer porous structure. The polybenzimidazole film has high phosphoric acid doped level and high proton conductivity and can be applied to high-temperature fuel cells. According to the invention, a zeolite imidazate framework ZIFs series of materials are taken as pore-foaming agents and phosphoric acid is taken as the etching agent so asto in situ form the nanometer porous structure in the polybenzimidazole film. ZIFs materials can acquire different crystal sizes and morphologies by adjusting the molecular structure of imidazole ligand and the type of metal ions, so that the pore size, pore morphology, pore size distribution and pore connectivity of the prepared porous film all can be regulated.
Owner:JILIN UNIV
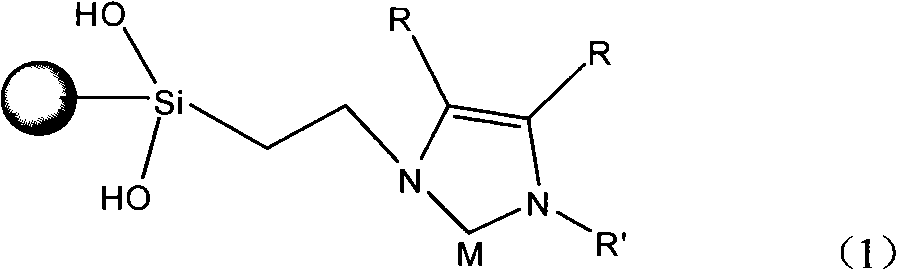
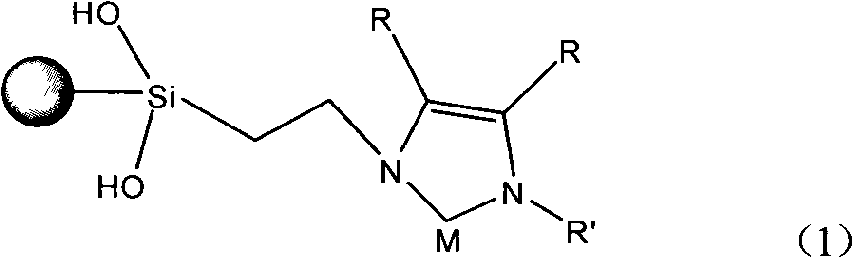
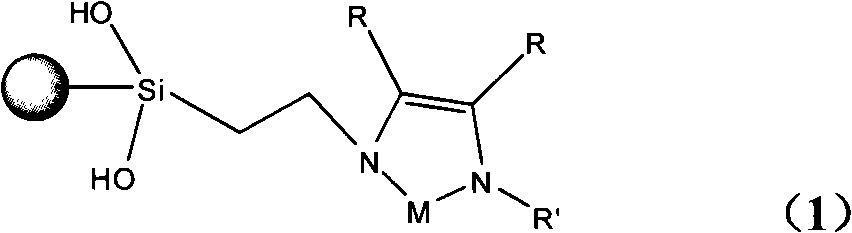
Glyoxaline ligand metallic catalyst supported on silica-gel and process for synthesizing the same
InactiveCN101279293AHigh activityImprove catalytic performanceOrganic-compounds/hydrides/coordination-complexes catalystsWater/sewage treatmentStructural formulaSilica gel
The present invention discloses a silica solid-carrying imidazole ligand metal catalyst with a structural formula on the right; wherein, the group on the left is a silica solid-carrying agent, R is selected from H atom and methyl; R' is selected from phenmethyl and aryl; M is selected from metal palladium or nickel. The catalyst uses imidazole carbine and transition metals to form a ligand compound. The catalyst can be used for degradation and dehalogenation of the organic halides in water; the reaction conditions of the catalyst are gentle and no secondary pollution is generated. The present invention provides an effective method for the treatment of waste water with organic halides included. The present invention also applies for a synthesizing method for the catalyst provided by the present invention.
Owner:KUNMING UNIV OF SCI & TECH
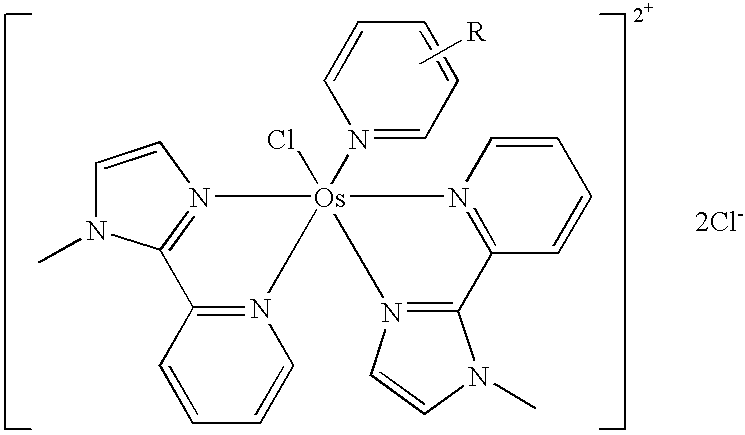
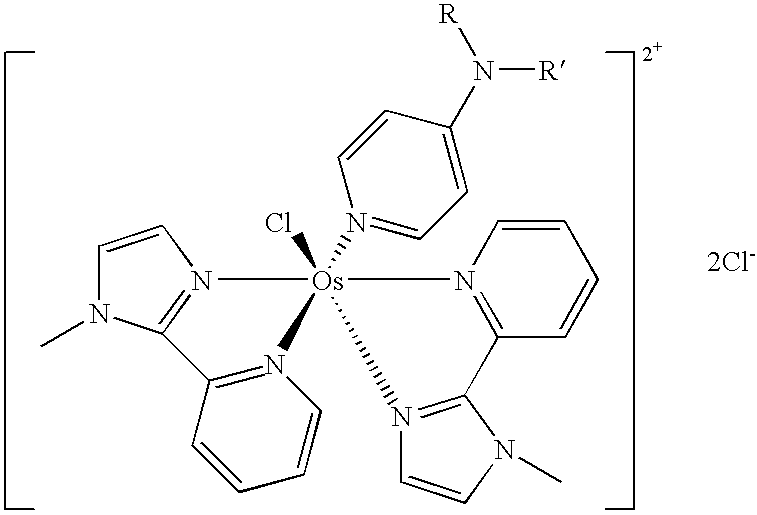
Transition metal complexes with pyridyl-imidazole ligands
InactiveUS20090095642A1Increase contactRapid electron exchangeImmobilised enzymesBioreactor/fermenter combinationsRedoxRedox mediator
Enzyme-based electrochemical sensors comprising transition metal complexes of iron, cobalt, ruthenium, osmium, and vanadium are described. The transition metal complexes can be used as redox mediators and include substituted or unsubstituted pyridyl-imidazole ligands. Transition metal complexes attached to polymeric backbones are also described.
Owner:ABBOTT DIABETES CARE INC
Organic electroluminescent materials and devices
A novel type of blue emitter is described based on the azaphenanthridine imidazole ligand. The preferred use of this moiety for generating blue phosphorescence is as part of a symmetric platinum tetradentate complex.
Owner:UNIVERSAL DISPLAY
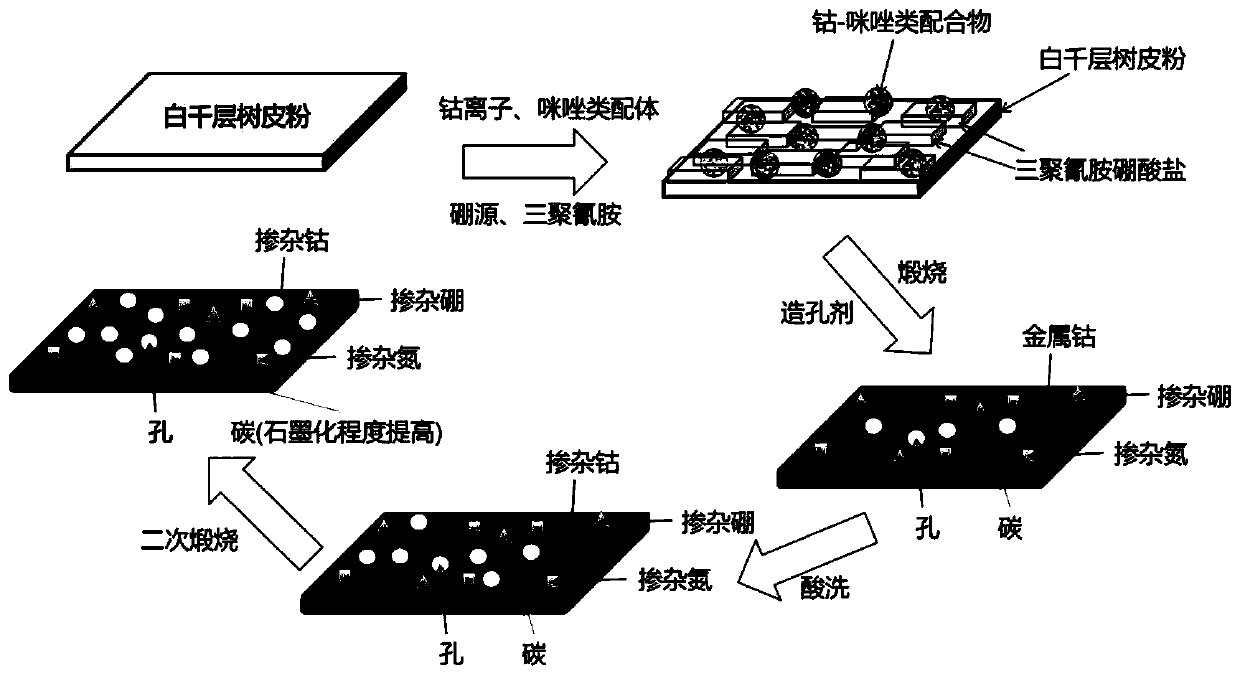
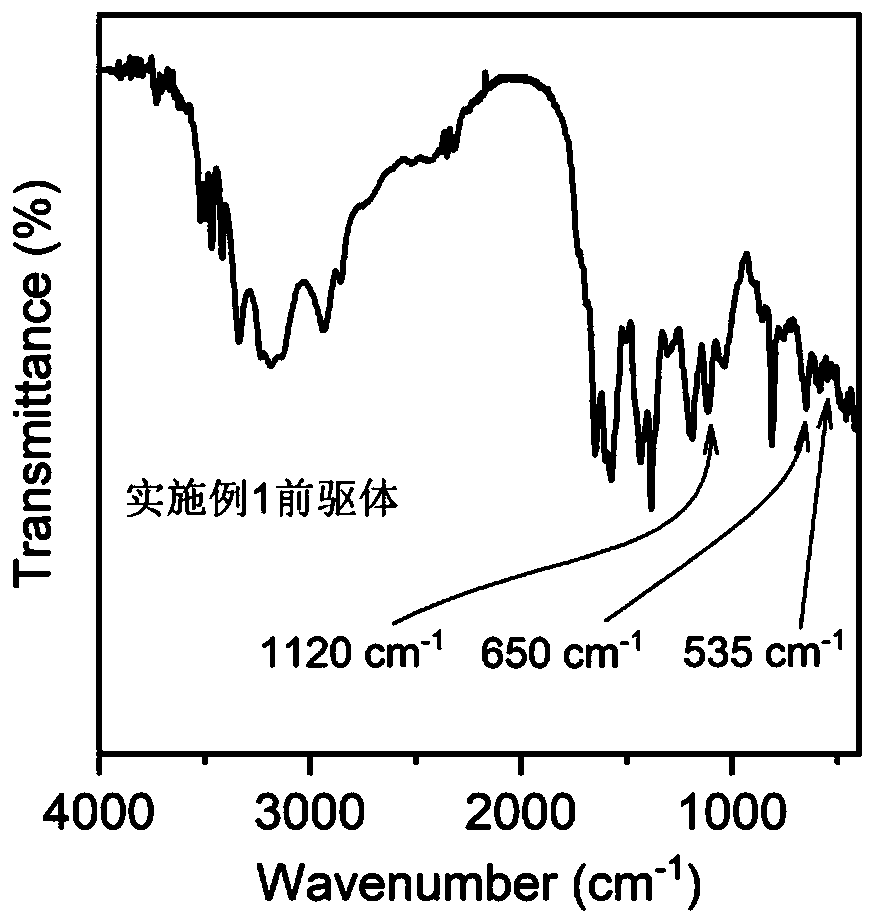
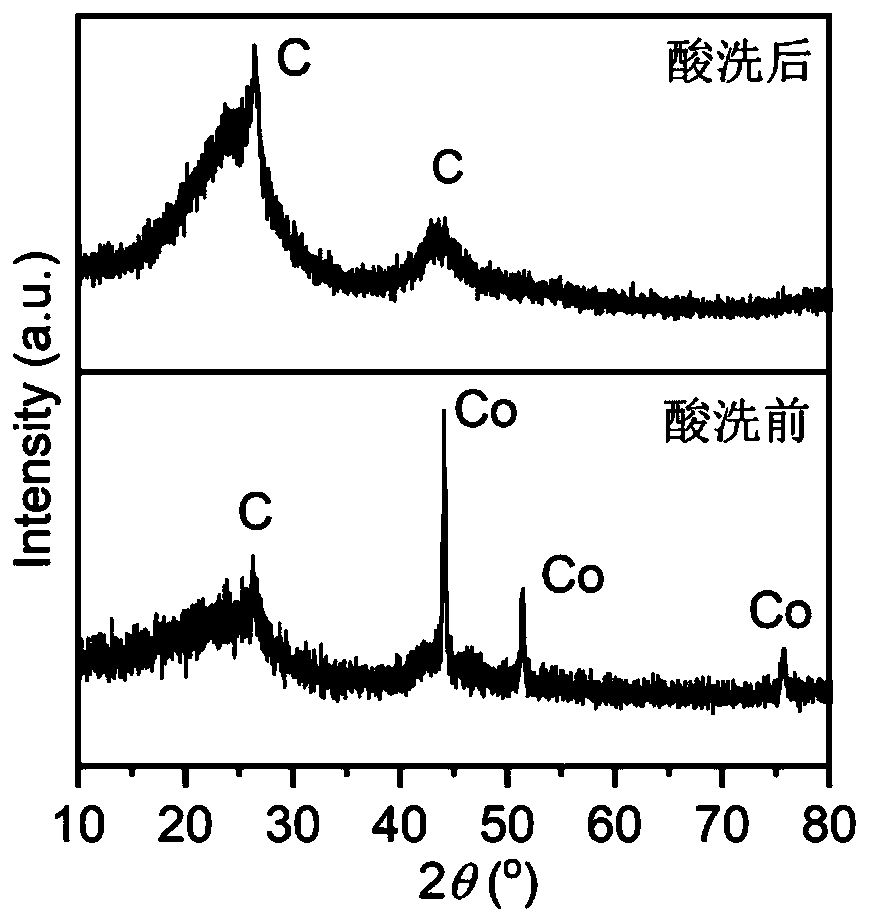
Non-noble metal electrocatalyst based on melaleuca leucadendron bark powder and preparation method and application thereof
ActiveCN110649269AEffective dispersionPrevent volatilizationCell electrodesPtru catalystCarbonization
The invention relates to a non-noble metal electrocatalyst based on melaleuca leucadendron bark powder and a preparation method and an application thereof. The preparation method comprises steps of S1, mixing an imidazole ligand material, a boron source, melamine, the melaleuca leucadendron bark powder and cobalt salt in solvent to obtain suspension, performing ultrasonic treatment, and obtaininga product after drying; S2, mixing the product with pore-forming agent, and performing carbonizing to obtain a carbonized product; and S3, pickling the carbonized product, and performing drying to obtain the non-noble metal electrocatalyst. The preparation method is advantaged in that the preparation method has simple process flow, low raw material cost, convenience in macro preparation, suitability for industrial production and the like, the prepared non-noble metal electrocatalyst has electrocatalytic oxygen reduction (ORR) performance similar to that of commercial Pt / C catalyst and even higher and more stable than that of the commercial Pt / C catalyst, and is expected to be applied to new energy devices of large zinc-air batteries, aluminum-air batteries and fuel batteries.
Owner:DONGGUAN UNIV OF TECH +1
Zeolitic imidazolate frameworks for kinetic separation of propane and propene
InactiveUS8796462B2Different rate of diffusionEnergy- and cost-efficientGas treatmentDispersed particle separationPropaneImidazole ligand
Zeolitic Imidazolate Frameworks (ZIFs) characterized by organic ligands consisting of imidazole ligands that are either essentially all 2-chloroimidazole ligands or essentially all 2-bromoimidazole ligands are disclosed. Methods for separating propane and propene with the ZIFs of the present invention, as well as other ZIFs, are also disclosed.
Owner:RUTGERS THE STATE UNIV
Ortho-metallated hafnium complexes of imidazole ligands
InactiveCN101460509AGroup 4/14 organic compounds without C-metal linkagesHafniumOlefin polymerization
Hafnium complexes of heterocyclic organic imidazol-2-yl ligands containing internal orthometallation and the use thereof as components of olefin polymerization catalyst compositions, especially supported catalyst compositions, are disclosed.
Owner:DOW GLOBAL TECH LLC
Metal organic framework based on imidazole sulfonic acid as well as preparation method and application
InactiveCN106883422ARealize functional integrationIncrease varietyOrganic chemistryOrganic-compounds/hydrides/coordination-complexes catalystsBenzaldehydeSynthesis methods
The invention provides a metal organic framework based on imidazole sulfonic acid as well as a preparation method and application. The metal organic framework based on the imidazole sulfonic acid is obtained by synthesizing and modifying a metal organic framework based on an imidazole ligand through a sulfonic compound; a chemical structural formula of the metal organic framework based on the imidazole ligand is [Zr6O4(OH)4L6]n, wherein n is a natural number more than 0; L is an organic ligand L and a chemical structural formula of the organic ligand L is show in the description; a synthesis method of the organic ligand L comprises the following steps: firstly, enabling 3-methyl-terephthalic acid to react to obtain an intermediate A; secondly, taking the intermediate A and bromosuccinimide as raw materials and reacting to obtain an intermediate B; thirdly, taking the intermediate B and imidazole as raw materials and reacting to obtain an intermediate C; finally, carrying out hydrolysis reaction on the intermediate C to prepare the organic ligand L. The metal organic framework based on the imidazole sulfonic acid has a catalytic effect on benzaldehyde under normal pressure and has the characteristics of moderate reaction conditions, short reaction time, less dosage of a catalyst and capability of being recycled and repeatedly utilized.
Owner:SHANDONG NORMAL UNIV
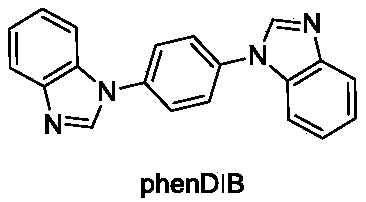
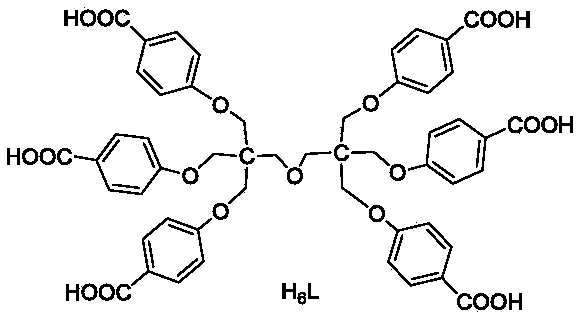
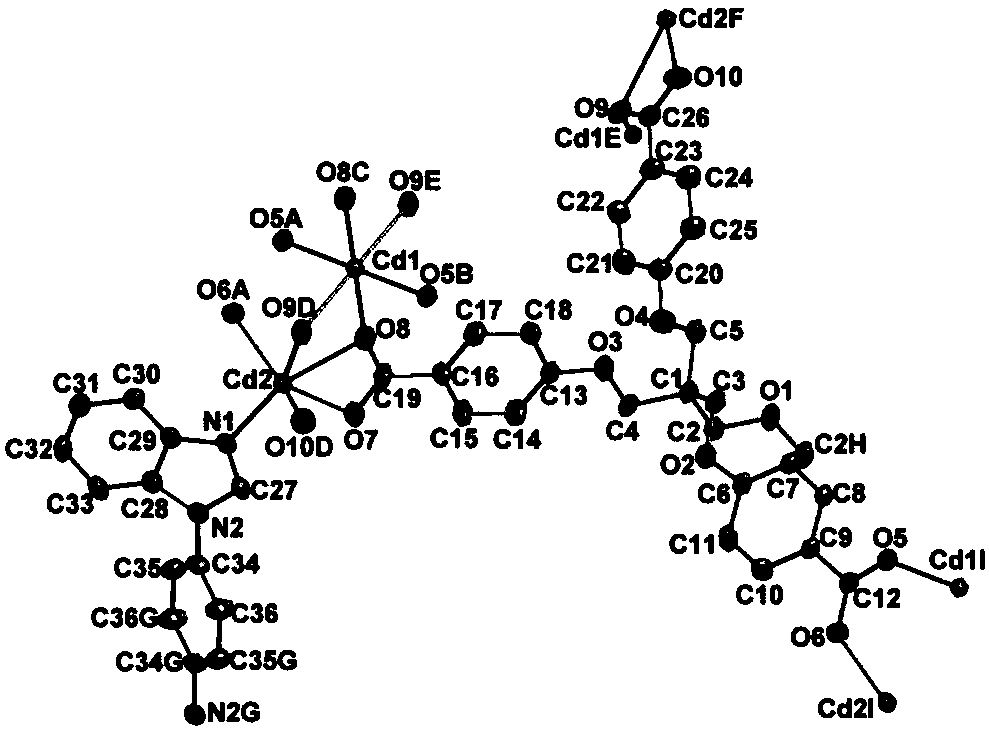
Cadmium metal organic framework complex and preparation method thereof
ActiveCN107778495AImprove stabilityHigh sensitivityFluorescence/phosphorescenceLuminescent compositionsSpace groupTetrachloride
The invention relates to a cadmium metal organic framework complex. The cadmium metal organic framework complex is cooperatively formed by a flexible carboxylate ligand and a rigid imidazole ligand, the chemical formula is {Cd3(L)(phenDIB).6DMF}n, and the molecular formula is C90H96Cd3N10O25; the cadmium metal organic framework complex is a monoclinic space group C2 / c; and cell parameters are as follows: a / angstrom is 29.403(3), b / angstrom is 20.8279(19), c / angstrom is 16.9132(17), alpha is 90 degrees, beta is 111.067(2) degrees, V / angstrom is 6665.2(16), the density is 1.412mg / m<3>, the lightabsorption coefficient is 0.730mm<-1>, and the number of molecules in a single cell is 4. By utilizing the cadmium metal organic framework complex, the high-sensitivity selective detection of high-toxicity carbon tetrachloride molecules and hexavalent heavy metal chromium ions can be realized; and the cadmium metal organic framework complex has good application prospects.
Owner:NINGBO UNIV
Method for preparing zeolitic imidazolate framework material (ZIF-8) nano-octa-tripod by using seed growth synthesis
The invention relates to a method for preparing a zeolitic imidazolate framework material (ZIF-8) nano-octa-tripod by using seed growth synthesis. The method comprises the following steps of (1) mixing and dissolving a zinc salt, an imidazole ligand, a surfactant and water at room temperature, preserving heat of the mixture at room temperature for 1-4 hours, washing the mixture to obtain ZIF-8 truncated dodecahedral seeds, and then dispersing the seeds into water to obtain a seed dispersion liquid; and (2) stirring, mixing and dissolving the zinc salt, the imidazole ligand, the surfactant, theseed dispersion and a solvent at room temperature, and preserving heat of the mixture at room temperature for 1-12 hours, and washing the mixture to obtain the ZIF-8 nano-octa-tripod product. The nano-octa-tripod ZIF-8 synthesized by the invention breaks through the long-standing bottleneck of the ZIF-8 polyhedron morphology and provides more possibilities for the product morphology of ZIF-8.
Owner:SHANDONG UNIV
Method of synthesizing and preparing zeolitic imidazolate framework material ZIF-8 nanometer six-foot body by using seed growth
The invention relates to a method of synthesizing and preparing a zeolitic imidazolate framework material ZIF-8 nanometer six-foot body by using seed growth. The method comprises the following steps of (1) mixing and dissolving zinc salt, an imidazole ligand, a surfactant and water at room temperature, performing thermal insulation for 2-4 h at room temperature, performing washing to obtain ZIF-8cube seeds, and then dispersing the ZIF-8 cube seeds into water to obtain a seed dispersion liquid; and (2) stirring, mixing and dissolving the zinc salt, the imidazole ligand, the surfactant, the seed dispersion liquid and a solvent at room temperature, performing thermal insulation for 1-12 h at room temperature, and performing washing to obtain a ZIF-8 nanometer six-foot body product. Accordingto the method provided by the invention, the ZIF-8 nanometer six-foot body is prepared by using a seed growth method, the seeds which are synthesized in advance instead of a precursor are selected asa starting point of crystal growth. Compared with a one-step synthesis method, the method can avoid a complex and difficultly controlled initial nucleation step and accurately controls the growth stage from angles of thermodynamics and dynamics.
Owner:SHANDONG UNIV
Preparation method and application of ammonium molybdate doped ZIF-67/chitosan composite catalyst
InactiveCN110368981AImprove production efficiencySimple preparation processPhysical/chemical process catalystsElectrodesArgon atmosphereCobalt
The invention discloses a preparation method and application of a molybdenum doped ZIF-67 / chitosan composite catalyst, and belongs to the technical field of metal organic complex nano materials and catalysis and the like. The preparation method includes the steps that the chitosan is mixed with cobalt nitrate hexahydrate and stirred into gel, and the gel is dropped into a mixed solution of ammonium molybdate and a dimethyl imidazole ligand by a needle to form pellets, and the pellets are activated in an oven, filtered, washed and calcined in an argon atmosphere to obtain the molybdenum doped ZIF-67 / chitosan composite catalyst. The preparation method has the advantages of low cost of raw materials, simple preparation process, low reaction energy consumption and industrial application prospect. The hybrid material is used for catalytic nitrogen fixation and has good electrocatalytic activity and electrochemical stability.
Owner:UNIV OF JINAN
Compositions and methods for hydration of terminal alkynes
InactiveUS6627758B2Facilitate one or more chemical reactionsEffective structureGroup 4/14 element organic compoundsGroup 8/9/10/18 element organic compoundsChemical reactionPtru catalyst
Owner:SAN DIEGO STATE UNIVERSITY