Patents
Literature
112 results about "Cadmium Metal" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
Cadmium is primarily found in zinc-containing ores, but it may also be found in lead and copper ores. The primary mineral form of the metal is greenockite or cadmium sulfide.
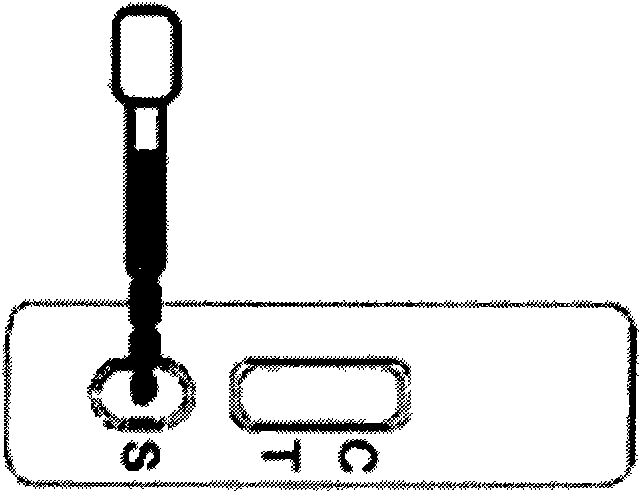
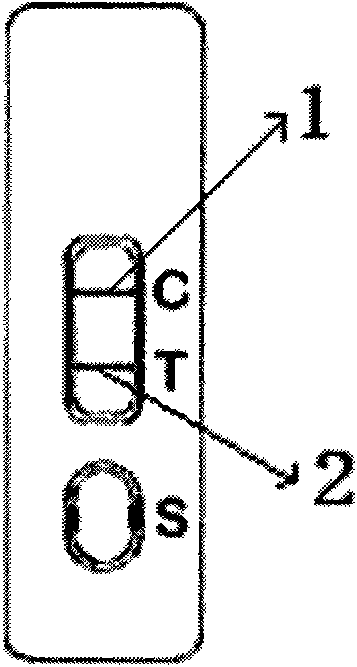
Test paper for rapidly detecting immunochromatography of cadmium ion colloidal gold and preparation method and application thereof
The invention discloses test paper for rapidly detecting the immunochromatography of cadmium ion colloidal gold and a preparation method and application thereof. The test paper consists of a sample pad, a colloidal gold combination pad, a nitrocellulose membrane, absorbent paper and a plastic base plate; The sample pad, the colloidal gold combination pad, the nitrocellulose membrane and the absorbent paper are sequentially stuck on the plastic base plate; an anti-cadmium ion monoclonal colloidal gold label is coated on the colloidal gold combination pad; a goat anti-rat IgG antibody and a complete antigen Cd-iEDTA-BSA of heavy metal cadmium are sequentially coated on the nitrocellulose membrane; the goat anti-rat IgG antibody is used as a quality control line; and the complete antigen Cd-iEDTA-BSA of the heavy metal cadmium is used as a test line. The test paper detects residual metal cadmium in ambient soil, a water body and an aquatic product based on the immunology theory of antigen antibody; and compared with the prior detection system, the test paper has the advantages of rapidness, cheapness, convenience, sensitivity and speciality, can give a result within five minutes and is convenient to carry along to carry out field monitoring.
Owner:JINAN UNIVERSITY
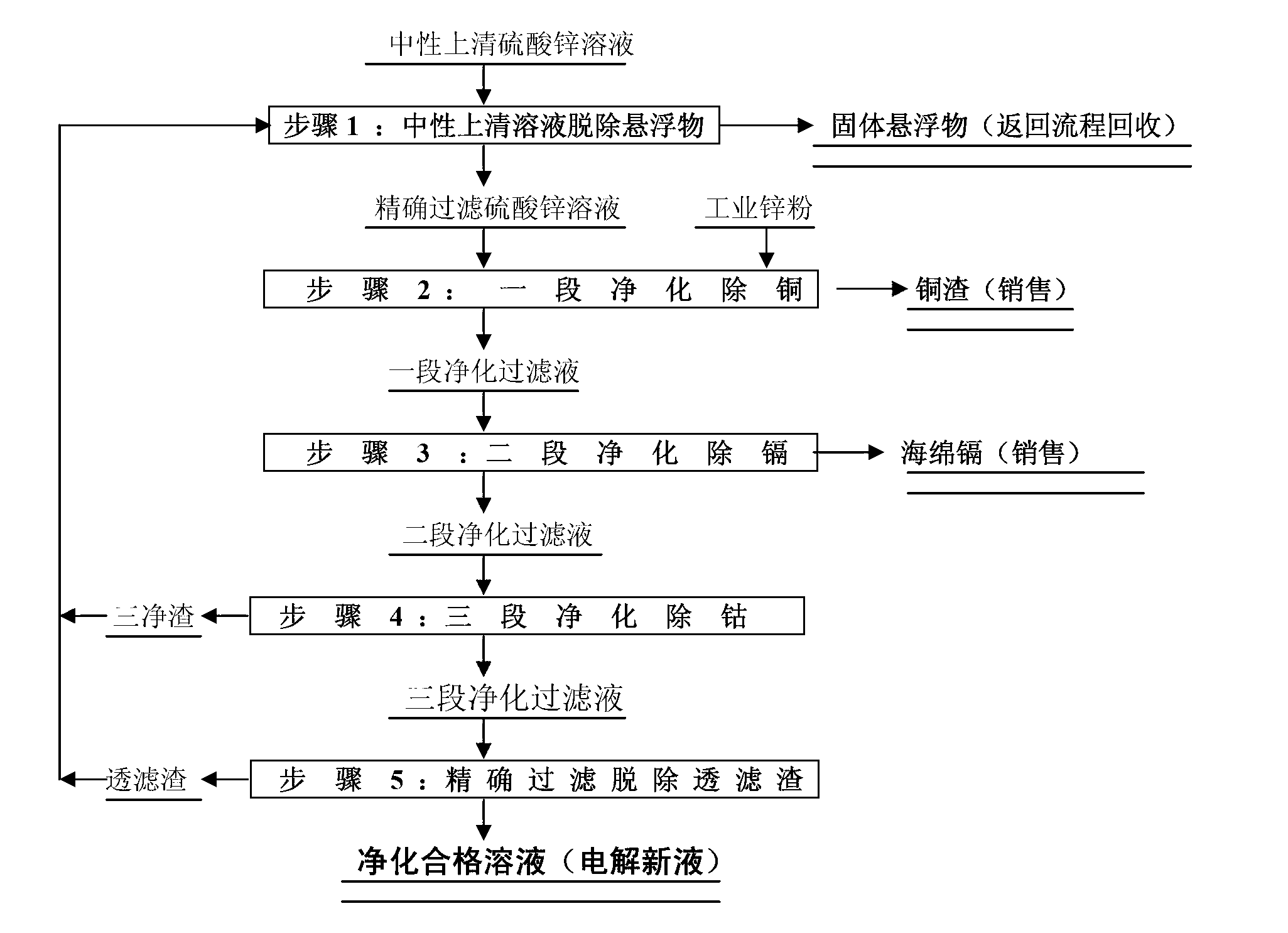
Method for purifying zinc sulfide solution
InactiveCN103194600AEfficient recyclingReduce consumptionProcess efficiency improvementElectrolysisAntimony trioxide
A method for purifying zinc sulfide solution comprises the following steps of: firstly, removing suspended solids from zinc sulfide solution by using a sean filter at a normal temperature; secondly, adding zinc powder by 120% of solution copper metal weight, stirring for 90 minutes at normal temperature for removing copper, and filtering; thirdly, adding zinc powder by 120-150% of solution cadmium metal weight, stirring for 60 minutes at a normal temperature for removing cadmium, and filtering; fourthly, increasing the temperature of the solution to the range of 82-94 DEG C, adding zinc powder in a proportion of 1 g / L of solution, and simultaneously adding some antimonous oxide, stirring for 50-70 minutes, removing cobalt and nickel impurities, and filtering; and finally, removing few suspended solids from the filtered solution by the sean filter. The method provided by the invention is capable of directly producing copper slag and sponge cadmium product; and the solution is capable of meeting the quality standard requirements of fresh electrolyte even though no extra zinc powder is added to the solution to remove residual cadmium; therefore, the consumption of the zinc powder is effectively reduced.
Owner:吴鋆
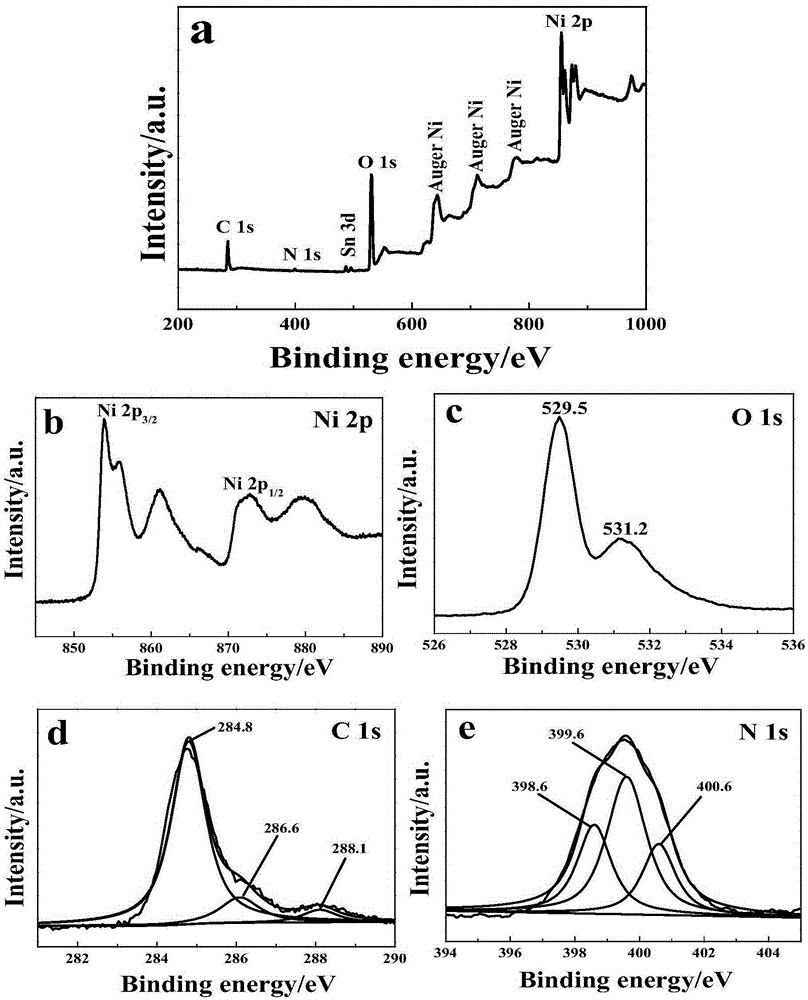
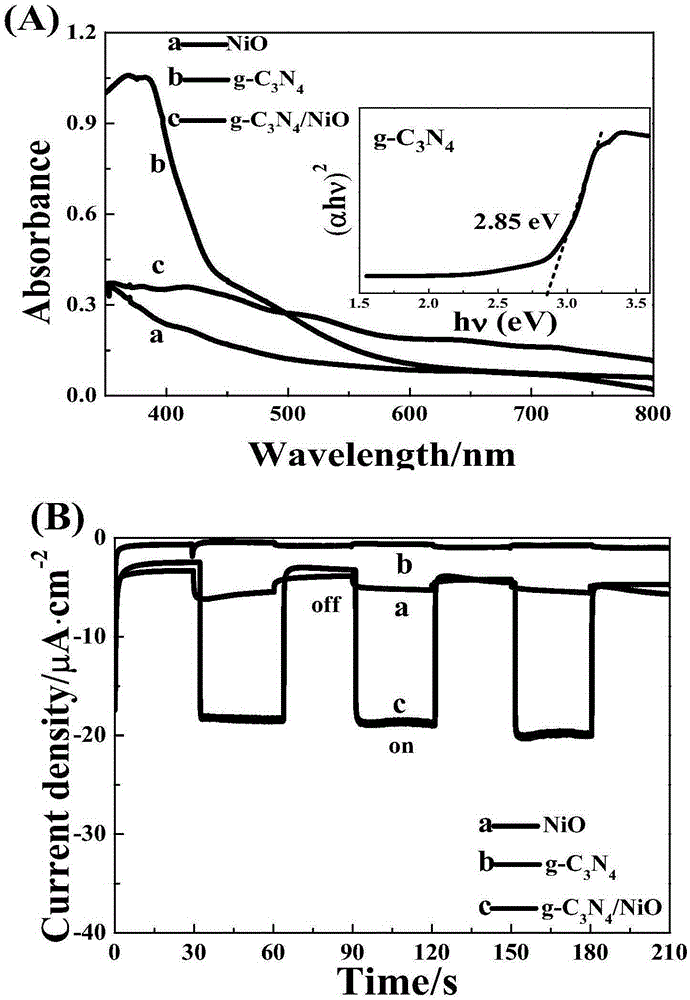
Novel and stable g-C3N4/NiO photoelectric cathode preparation method
ActiveCN105336498ASimple manufacturing methodEfficient responseLight-sensitive devicesPhotovoltaic energy generationThioureaSulfur
The invention discloses a novel and stable g-C3N4 / NiO photoelectric hydrogen production electrode preparation method, and belongs to the technical field of inorganic chemistry and photoelectrocatalysis. The g-C3N4 light absorber is nonmetal and is made of nontoxic and easily available materials. The preparation method solves the problem that in the prior art, usually a toxic cadmium metal chalcogenide as the light absorber, and utilizes the g-C3N4 as the photoelectric cathode light absorption agent and cocatalyst for the first time. The preparation method includes: 1 taking FTO electro-conductive glass as the substrate and using a hydrothermal method to prepare ordered NiO films; and 2 immersing the NiO / FTO electrode obtained by the last step in the saturated thiourea solution for 4h, and then placing the NiO / FTO electrode in a muffle furnace for calcining for 2h by 500DEG C, and then taking out and obtaining the g-C3N4 / NiO / FTO electrode. The preparation method is simple and easy to operate and is good for large-scale application.
Owner:JIANGNAN UNIV
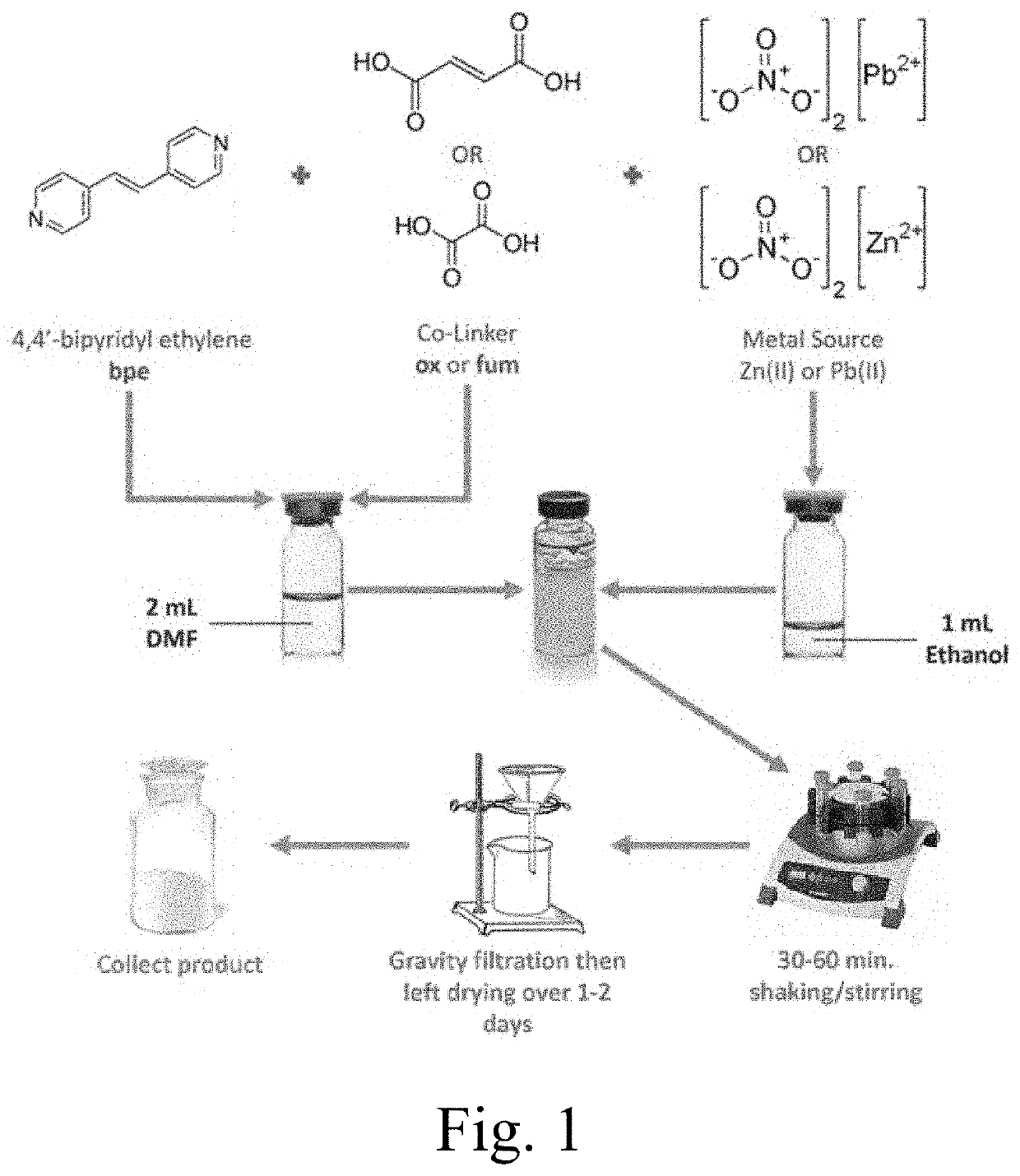
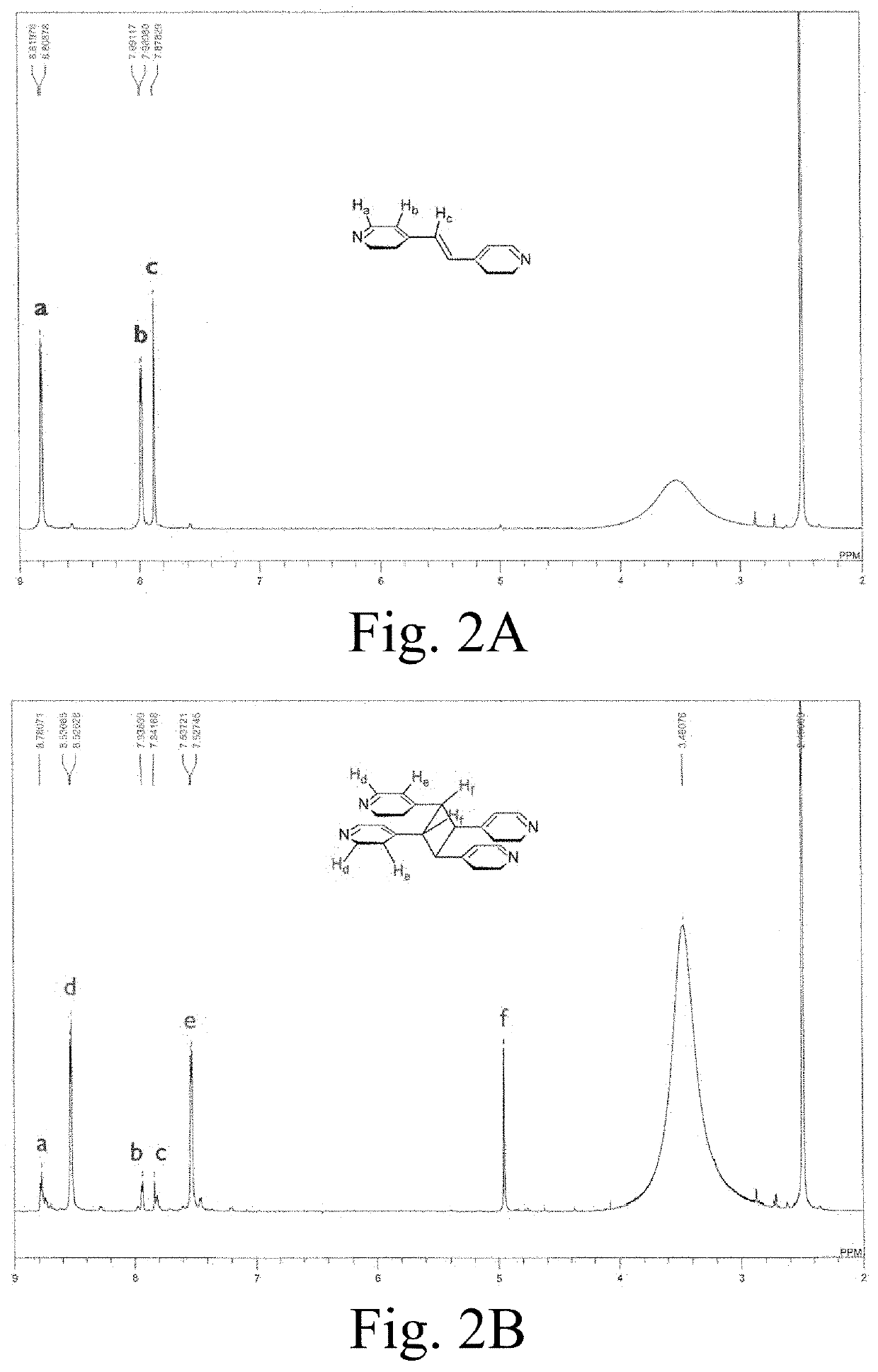
4,4'-bipyridyl-ethylene mofs of lead, zinc, or cadmium
ActiveUS20200269225A1Maintain good propertiesStructural topology of MOFs can be modifiedOrganic compound preparationGroup 4/14 organic compounds without C-metal linkagesOXALIC ACID DIHYDRATEPhysical chemistry
Metal-organic frameworks (MOFs) may have Zn(II), Pb(II), and / or Cd(II) as a central metal ion; a 4,4′-bipyridylethylene (bpe) ligand as a first ligand; and fumaric acid (fum) and / or oxalic acid (ox) as a second ligand, wherein the 4,4′-bipyridylethylene ligands are stacked in the MOF, and wherein a distance between two consecutive 4,4′-bipyridylethylene ligands is less than 5 Å. Cycloadditions, particularly photoinduced [2+2] cycloadditions may be catalyzed by such MOFs, and / or the conversion of photoinduced [2+2] cycloadditions in inventive MOFs may be increased by mechanical force, such as by grinding.
Owner:KING FAHD UNIVERSITY OF PETROLEUM AND MINERALS
Metallic fast reactor fuel element irradiation test device
ActiveCN110600150ARegulation and control of irradiation temperatureReduce the impactNuclear energy generationNuclear monitoringNeutron irradiationNuclear engineering
The invention discloses a metallic fast reactor fuel element irradiation test device. The device can be used for carrying out neutron irradiation test on metallic fast reactor fuel in a thermal spectrum research reactor and has the functions of neutron spectrum hardening, metallic fuel irradiation temperature control, fuel element heat release power measurement and the like. The device comprises aprotection tube, a cadmium tube, an irradiation test piece, a related gas pipeline, a thermocouple and the like. According to the device, a cadmium metal layer is adopted to absorb thermal neutrons to reduce the thermal neutron flux rate in the device, so that the neutron energy spectrum of a test fuel element is close to that of a fast neutron reactor; and the temperature difference is established between air gaps and a liquid metal layer outside a fuel rod, and the gas components in the air gaps in each irradiation part are independently adjusted, so that the temperature of fuel core bodiesin different irradiation parts are controlled within a required temperature range.
Owner:NUCLEAR POWER INSTITUTE OF CHINA
Cadmium metal organic frame compound with catalytic performance and preparation method and use thereof
InactiveCN102516274AOrganic compound preparationOrganic-compounds/hydrides/coordination-complexes catalystsMetal clustersPtru catalyst
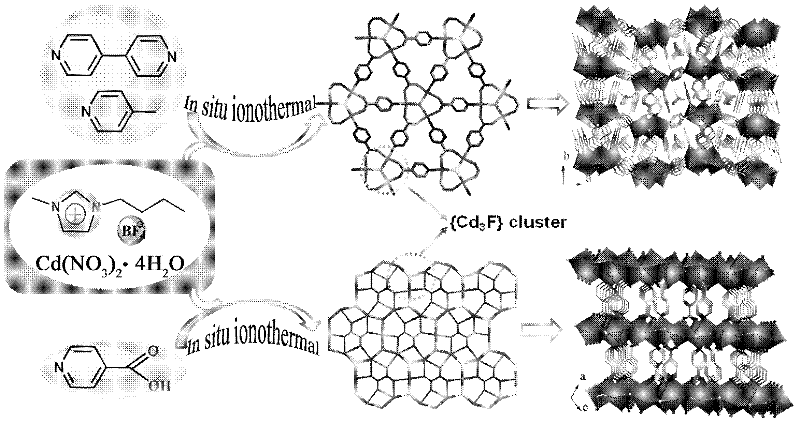
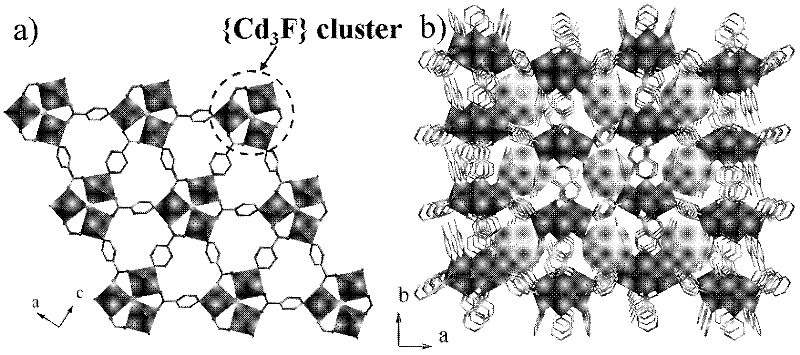
The invention provides a cadmium metal organic frame compound with catalytic performance and a preparation method and use thereof. A type of rarely-reported metal organic frame compound built by a three-core metal cluster formed by taking F as a central bridging cadmium ion and a carboxylic acid ligand generated through an in-situ reaction is synthesized thermally by using an ionic liquid. The ionic liquid plays multiple roles of a solvent, a soft template, an effective oxidation reaction accelerate and a ligand precursor in an ionic thermal synthesis process. The type of metal organic frame compound can be used for catalyzing a tetrahydrobenzene oxidation reaction, and the selectivity of a tetrahydrobenzene peroxide can be about 96 percent. Moreover, a catalyst is very easy to separate from a reaction system in a simple filtering way, the catalytic activity is kept, and the catalyst can be recycled repeatedly.
Owner:FUJIAN INST OF RES ON THE STRUCTURE OF MATTER CHINESE ACAD OF SCI
Cadmium metal complex with dye catalytic light degradation property and preparation method of cadmium metal complex
InactiveCN104693224ASimple processHigh yieldOrganic-compounds/hydrides/coordination-complexes catalystsCadmium organic compoundsCadmium acetateCadmium Metal
The invention discloses a cadmium coordination polymer adopting 1,3-bi(pyridine-3-methoxyl) benzene and 5-methyl isophthalic acid as ligands, and belongs to the chemical field. The chemical formula of the compound is as follows: [Cd2(5-Me-1,3-BDC)2(bpmb)2]n, wherein 5-Me-1,3-H2BDC is 5-methyl isophthalic acid; bpmb is 1, 3-bi(pyridine-3-methoxyl) benzene. The cadmium metal complex is prepared by adopting cadmium acetate dehydrate, 5-methyl isophthalic acid and 1,3-bi(pyridine-3-methoxyl) benzene. The preparation method of the complex is simple in process and relatively high in yield. The cadmium metal complex is stable in light catalytic degradation dye performance at a normal temperature and can be used for further developing a novel dye degradation material, so that novel vitality can be brought to the field of the material science.
Owner:ANYANG NORMAL UNIV
Composite soil conditioner and application thereof
InactiveCN106010576AEffective pH adjustmentEffective chelationAgriculture tools and machinesOther chemical processesSoil conditionerCadmium Metal
The invention relates to the technical field of agricultural environment conservation and repair, in particular to a composite soil conditioner. The composition soil conditioner is prepared from, by weight, 30-60 parts of limestone flour, 25-50 parts of decomposed straw, 10-30 parts of sepiolite and 1-10 parts of K2HPO4. The composite soil conditioner comprises four effective ingredients, the pH of soil can be stably kept at a certain level, and particularly, the pH of acid soil can be effectively adjusted; phosphate radical and humic acid in the conditioner can effectively chelate and deposit available cadmium in soil, and pollution brought by cadmium metal to soil and crops is reduced.
Owner:HUNAN TAIGU BIOTECH
Sepia pharaonis neuropeptide GnPH procreation regulation preparation
InactiveCN106749587APrevent loss of activity due to dehydrationAvoid degradationPeptide preparation methodsAnimals/human peptidesDiseaseSepia pharaonis
The invention provides a sepia pharaonis neuropeptide GnPH procreation regulation preparation. The sepia pharaonis neuropeptide GnPH procreation regulation preparation is prepared by the steps: 1) pre-treatment: taking live sepia pharaonis, dissecting the live sepia pharaonis to take a brain tissue and preserving the brain tissue in a preservation solution at an ultra-low temperature; 2) taking of supernatant: taking the brain tissue, adding saline of the same volume to the brain tissue, homogenizing and centrifuging an obtained mixture to take supernatant A; adding distilled water of the same volume to the obtained mixture, centrifuging the mixture with the distilled water to take supernatant B; and combining the supernatant A and the supernatant B to obtain supernatant C; 3) column chromatography: carrying out column chromatography, chromatography and developing on the supernatant C and collecting components to obtain neuropeptide; and 4) catalysis: enabling the neuropeptide to have substitution reaction with a GnRH derivative under the catalysis of ammonium nitrate and cadmium metal to generate the sepia pharaonis neuropeptide GnPH procreation regulation preparation. The sepia pharaonis neuropeptide GnPH procreation regulation preparation, provided by the invention, has the advantages that the tissue can be preserved in a frozen manner and the preservation effect is excellent; the brain tissue degradation of the sepia pharaonis can be effectively prevented; the extraction rate of the neuropeptide is higher; the prepared control agent does not need to be continuously or repeatedly injected for a long time; and the preparation is applied to drugs for preventing or treating hormone-dependent diseases.
Owner:ZHEJIANG OCEAN UNIV
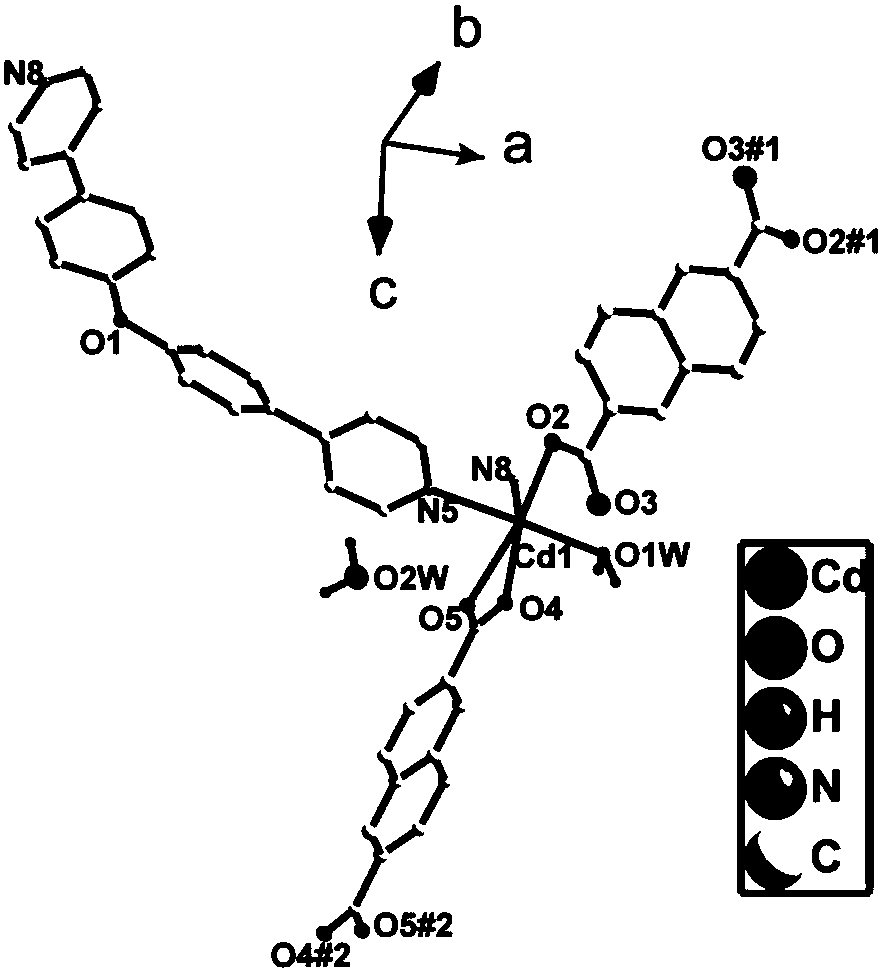
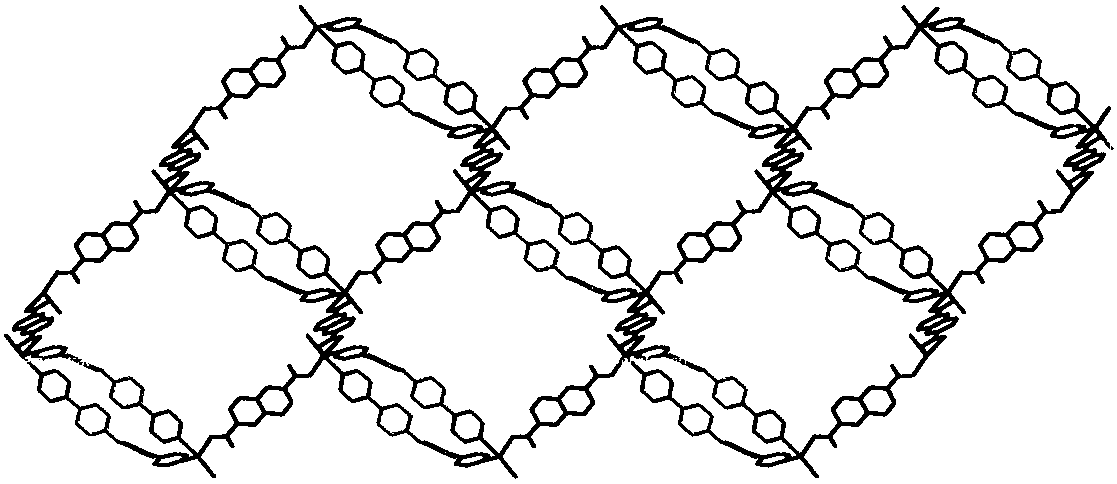
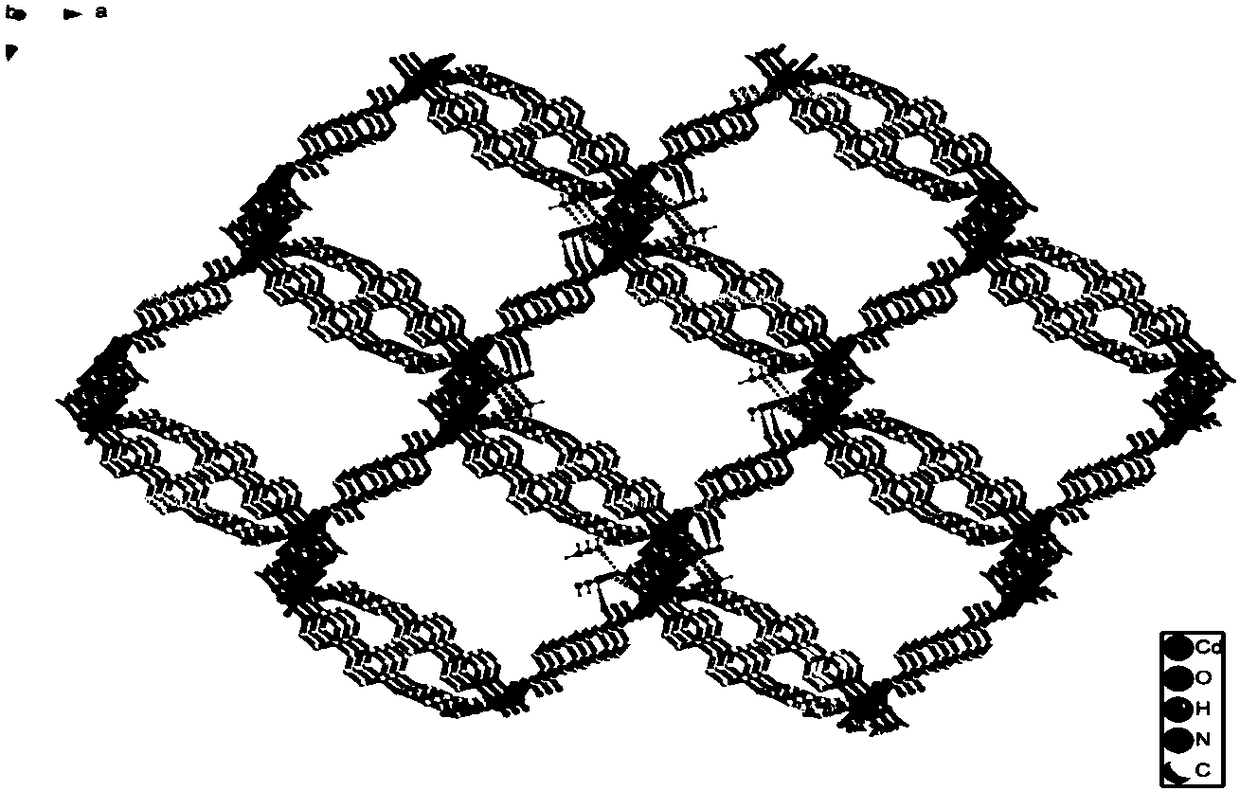
Cadmium metal organic complex with fluorescence property as well as preparation method and application thereof
ActiveCN108623621AExcellent fluorescence performanceEasy to operateOrganic chemistry methodsFluorescence/phosphorescenceSpace groupFluorescence
The invention discloses a cadmium metal organic complex with fluorescence property as well as a preparation method and application thereof. The complex has a chemical formula of [Cd3(BPDPE)(BDC)3.(DMF)2]2.DMF.2H2O, wherein the BPDPE is 4,4'-dipyridyl diphenyl ether, and the BDC is terephthalic acid anion. The complex belongs to an orthorhombic system, Pbcn space groups, and cell parameters are that a is equal to 11.5580 (11) angstroms, b is equal to 16.8150 (15) angstroms, c is equal to 55.775 (5) angstroms, alpha is equal to 90 degrees, beta is equal to 90 degrees, gamma is equal to 90 degrees, and V is equal to 10256.7 (17) angstroms<3>. The preparation method of the cadmium metal organic complex with fluorescence property provided by the invention is simple in operation, high in yield and excellent in reproducibility, and the prepared cadmium organic complex is stable in structure at a room temperature, has excellent fluorescence property, and is capable of detecting metal ions andsmall organic molecules.
Owner:ANHUI UNIV OF SCI & TECH
Synthesis and application of cadmium metal organic complex second-order nonlinear optical material
InactiveCN104311584APolycrystalline material growthCadmium organic compoundsMonopotassium phosphateSingle crystal
The invention relates to a cadmium metal organic complex second-order nonlinear optical material and synthesis and application thereof. The invention aims at synthesizing a second-order nonlinear optical responsive metal complex of cadmium in non-centrosymmetry by selecting a 3, 5-pyrazol dioctyl phthalate in centrosymmetry. By using cadmium chloride and 3, 5-pyrazol dioctyl phthalate as reaction raw materials and a mixed solution of acetonitrile and water as a reaction solvent, a single crystal of the complex (H3O)[Cd3(Hpada)2(pada)(H2O) under a solvothermal condition is obtained. The single crystal of the complex is flaky and easy to prepare by a device. The second-order nonlinear optical responsive strength is about 1 / 2 of KDP (monopotassium phosphate) in phase match and can be used for manufacturing a nonlinear optical device.
Owner:UNIV OF JINAN
Cadmium (II) metal organic complex based on 5-amino isophthalic acid influenced by solvent and preparation method thereof
InactiveCN106478697AGood fluorescence propertiesNo protectionOrganic chemistry methodsGroup 2/12 organic compounds without C-metal linkagesN dimethylformamideSolvent
The invention relates to a cadmium (II) metal organic complex based on 5-amino isophthalic acid influenced by solvent and preparation method thereof. The metal organic complex is a cadmium metal organic complex with repeated unit of [(Me2NH2)2Cd2(NH2-BDC)3(H2O)].DMF.8H2O. The NH2-BDC in the chemical formula [(Me2NH2)2Cd2(NH2-BDC)3(H2O)].DMF.8H2O is 5-amino isophthalic acid ligand. The DMF is N, N-dimethylformamide. The Me2NH2 is produced by the degeneration of solvent DMF. The Cd(II) metal organic complex emits blue fluorescent light, and can be used as fluorescent material.
Owner:ZHAOQING UNIV
Method for removing cadmium metal in source water by using hydrated manganese dioxide
InactiveCN1884120AStrong electrostatic attractionNo change in processingWater/sewage treatment by flocculation/precipitationWater sourceManganese
The invention discloses a metal cadmium removing method in the water source through hydrated manganese dioxide, which comprises the following steps: putting potassium permanganate and manganese sulfate at 1:1.5-1.54 molar rate in the water with nonattainment cadmium; flocculating; flowing water in the incidence sedimenting pool; filtering the sedimented water through filtering pool. The invention makes Cd2+ density far lower than drinking water standard at 0.005mg / L, which removes cadmium through adsorbing property of hydrated manganese dioxide.
Owner:HARBIN INST OF TECH
Electrocoppering method for TC2 titanium alloy
The invention discloses an electrocoppering method for TC2 titanium alloy. The method comprises the following steps of (1), selecting Emery with the particle size not larger than 60 meshes for performing abrasive blasting on the TC2 titanium alloy, wherein the abrasive blasting pressure ranges from 0.3 MPa to 0.5 MPa; (2), performing activating treatment on the TC2 titanium alloy in acid solutions after abrasive blasting, and reducing the thickness of the surface of the TC2 titanium alloy by two micrometers to seven micrometers; (3), placing the activated TC2 titanium alloy in the sulfuric acid and hydrochloric acid mixed solutions with the temperature of 18 DEG C to 25 DEG C for 30 min to 60 min for hydrotreating; (4), performing copper plating on the hydrogenated TC2 titanium alloy. According to the method, the binding force of the surface of the TC2 titanium alloy and a plating layer of the TC2 titanium alloy is ensured and improved, the electrical conductivity of the TC2 titanium alloy materials is improved, contact corrosion is prevented from happening to the TC2 titanium alloy materials and cadmium metal, and then the comprehensive performance of the TC2 titanium alloy materials is improved.
Owner:SHENYANG AIRCRAFT CORP
Method for preparing cadmium metal from copper and cadmium slag leachate
ActiveCN103773964APromote generationPromote growthProcess efficiency improvementSocial benefitsWorking environment
Owner:BAIYIN NONFERROUS GROUP
Cadmium metal organic complex and preparation method thereof
InactiveCN108558919AGood lookingEasy to operateOrganic chemistry methodsGroup 2/12 organic compounds without C-metal linkagesDiphenyl etherSpace group
The invention discloses a cadmium metal organic complex. The chemical formula of the cadmium metal organic complex is [Cd(BPDPE)(NDC) (H2O)] H2O, wherein BPDPE is 4,4'-dipyridine diphenyl ether, NDC is 2,6-naphthalene diacid anions, the cadmium metal organic complex belongs to a triclinic system and a P-1 space group, and cell parameters are shown in the description. The disclosed cadmium metal organic complex can be stable at room temperature for several months, and has good stability. The invention further discloses a preparation method of the cadmium metal organic complex. The method is simple, high in yield and good in repeatability.
Owner:ANHUI UNIV OF SCI & TECH
Talaromycesveerkampii strain for degrading cellulose and adsorbing cadmium and application of strain
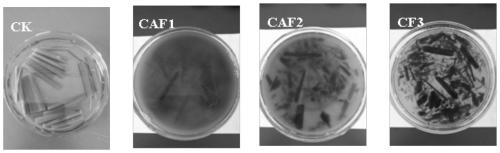
PendingCN110295118APromote degradationStrong colonization abilityFungiContaminated soil reclamationCelluloseBioremediation
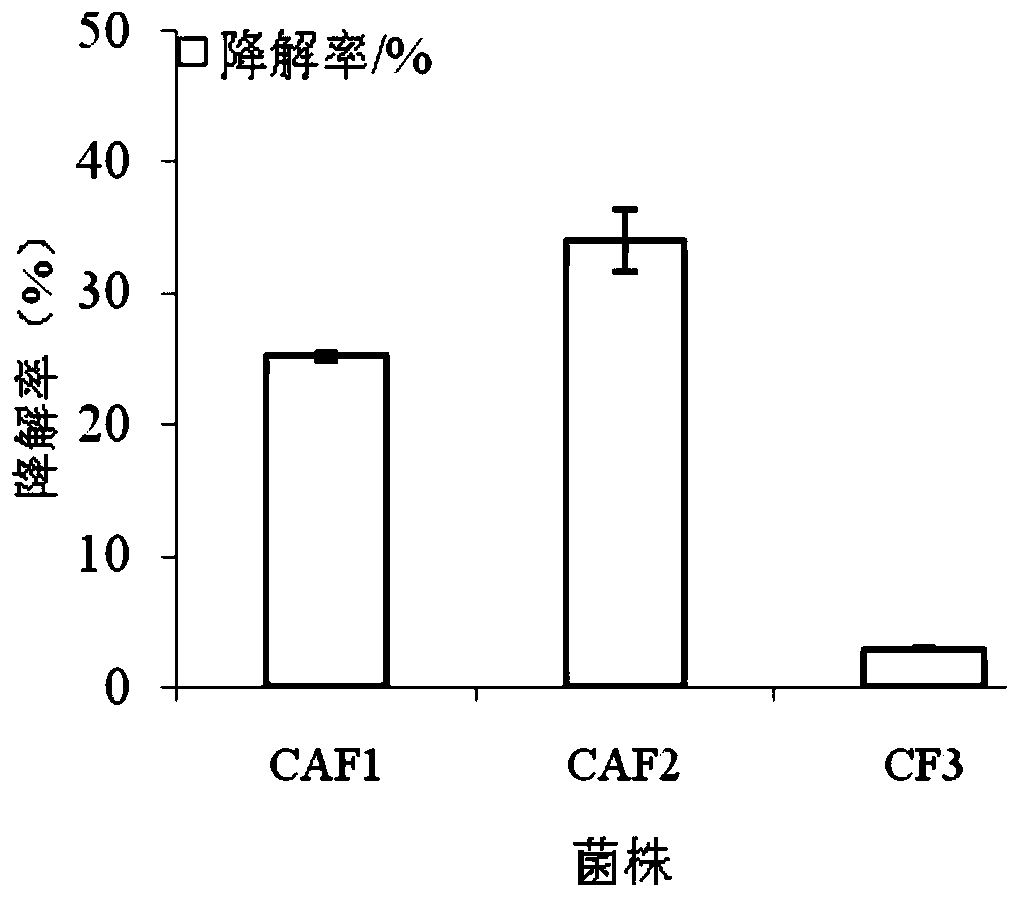
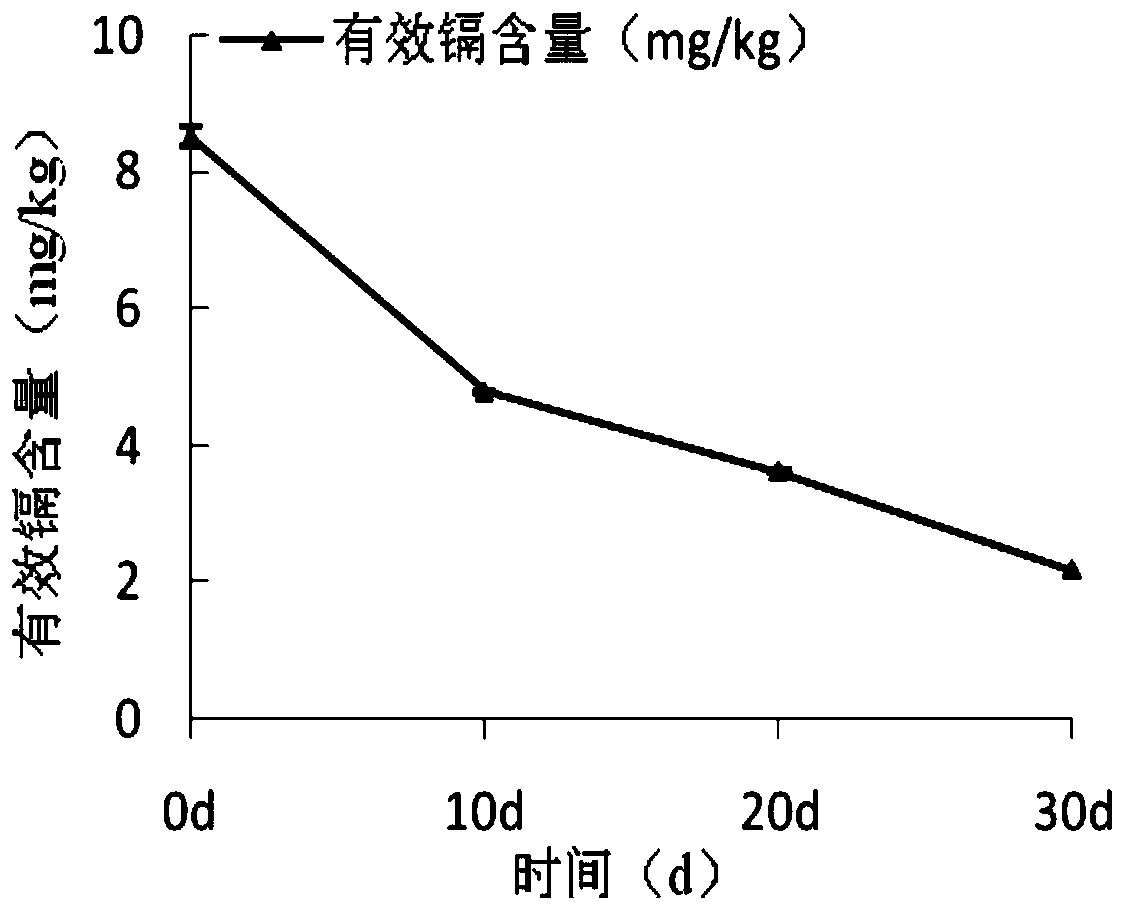
The invention provides a talaromycesveerkampii strain for degrading cellulose and adsorbing cadmium. The strain is classified and named as talaromycesveerkampii CAF2, and the preservation number of the strain is ACCC 32732. The cadmium ion concentration tolerance range of the talaromycesveerkampii strain is 0-100 mg / L. The talaromycesveerkampii strain can produce cellulase and has a good degradingeffect on rice straw, and when the strain is applied to degrading the rice straw, in the 15 th day, half of the rice straw is degraded; meanwhile, the strain has the capability of adsorbing heavy metal cadmium, and when the strain is applied to cadmium-polluted soil treatment, in the 30 th day, the adsorption rate of the talaromycesveerkampii strain on cadmium exceeds 74%. The strain is applied to bioremediation of cadmium-polluted rice fields, and can achieve the effect of solving soil heavy cadmium metal pollution while the rice straw is degraded.
Owner:湖南慧谷农业生态研究院有限公司
Buffer layer of flexible thin film solar cell and preparation method thereof
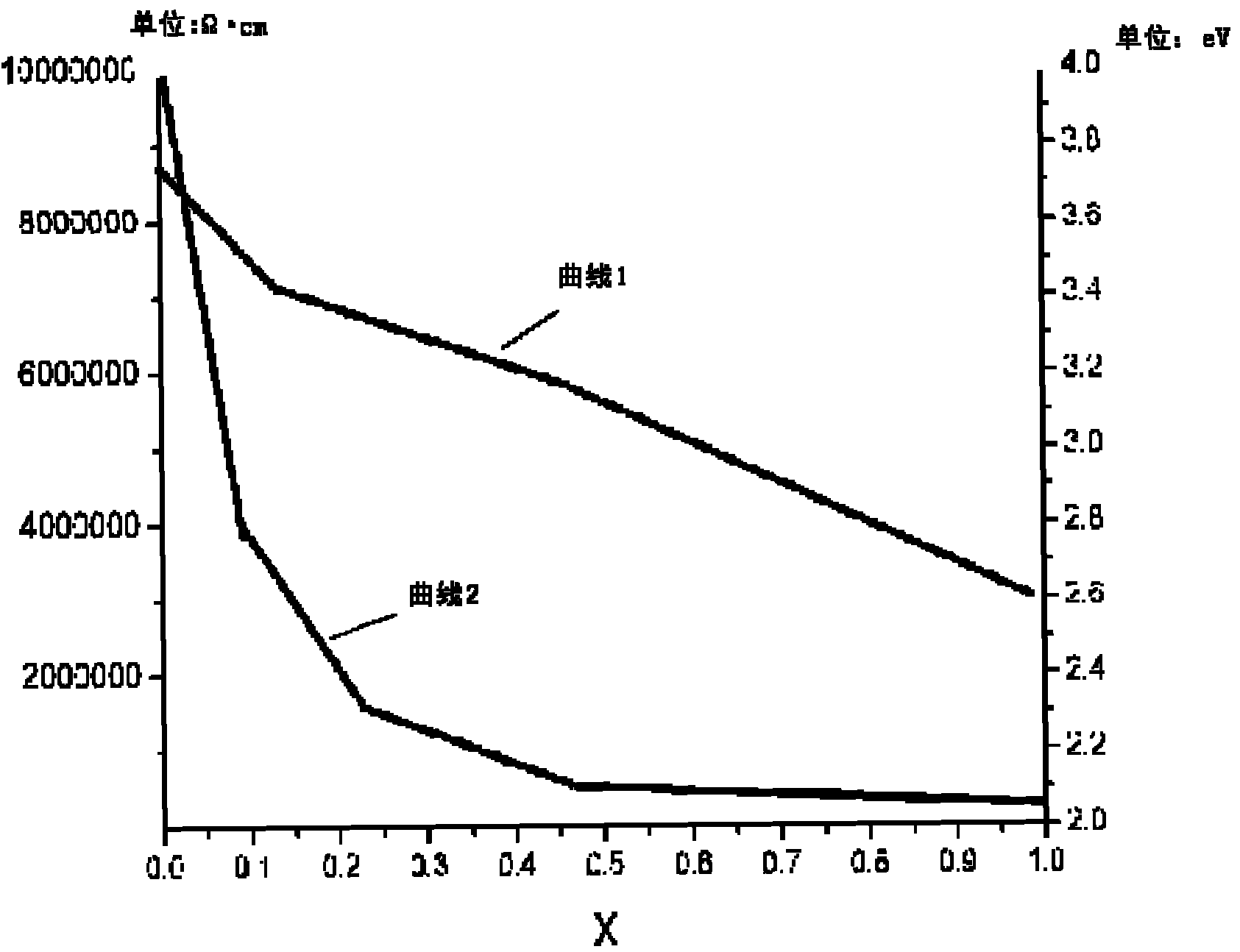
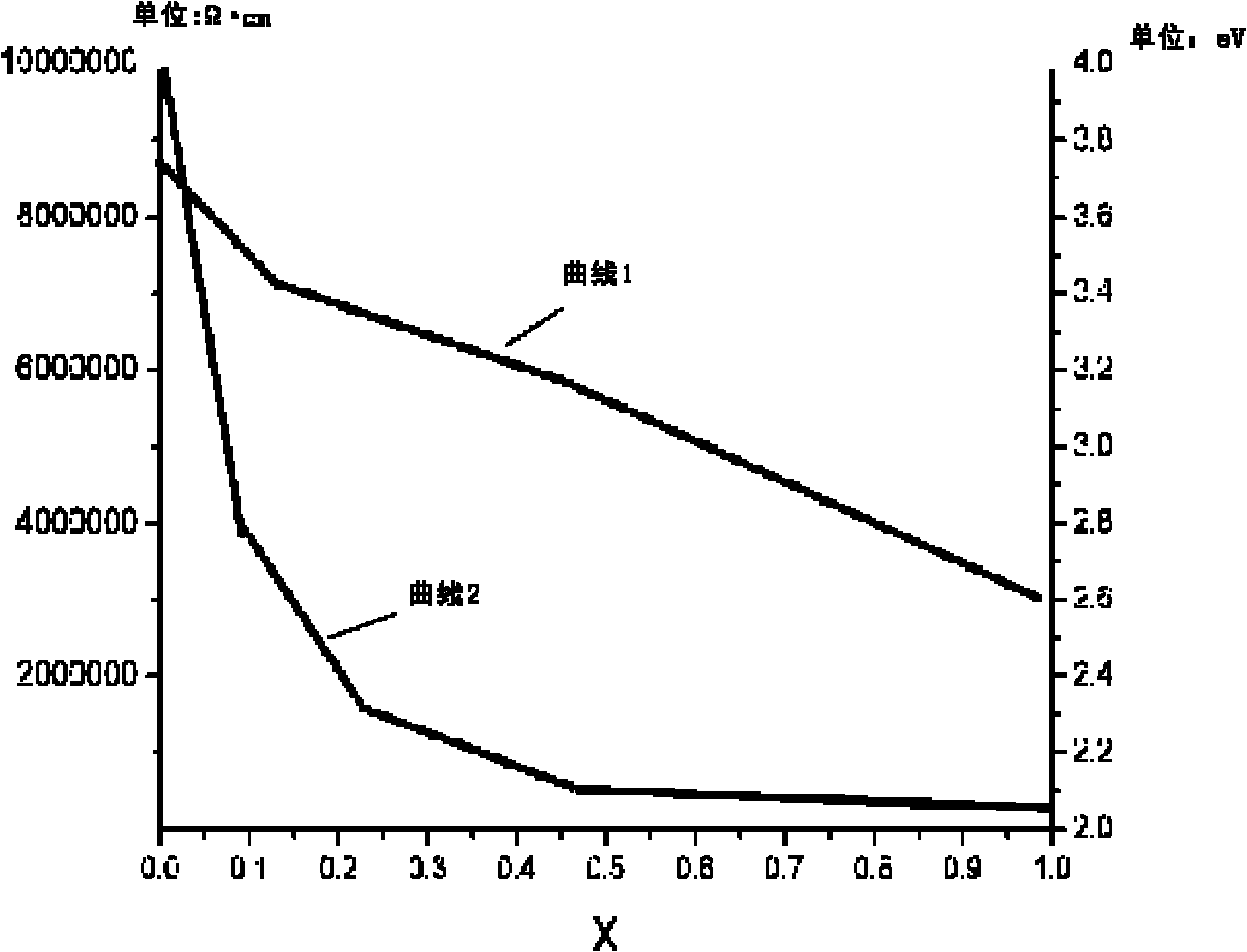
ActiveCN101980366AImproved band gapHigh resistivityFinal product manufactureSemiconductor devicesChemical reactionCadmium Metal
The invention provides a buffer layer of a flexible thin film solar cell and a preparation method thereof. The buffer layer of the flexible thin film solar cell is a Zn1-xMxS thin film, wherein M may be doped metal ions; and x is more than 0 and less than 1. The preparation method for the buffer layer of the flexible thin film solar cell comprises the following step of: Z, chemically reacting a Zn-containing compound with an S-containing compound and adding the metal ions in the chemical reaction for metal ion doping to form the buffer layer on a flexible substrate. The buffer layer of the flexible thin film solar cell and the preparation method thereof have the advantages of solving the problem of environmental pollution of cadmium metal and improving the optical and electrical properties of the thin film, along with proper forbidden gap.
Owner:SHENZHEN DANBANG INVESTMENT GROUP
Method for treating cadmium containing soil by utilizing low-accumulation cadmium crops
InactiveCN106941801AReduce absorptionReduce accumulationSoil-working methodsSoil conditionerCadmium Metal
The invention discloses a method for treating cadmium containing soil by utilizing low-accumulation cadmium crops. The method comprises the following steps: selecting a sweet potato variety with low cadmium absorption, high tolerance and high yield; applying lime and a ZH-type soil conditioner, adjusting the pH value and the like of soil, and restoring the soil in a polluted area; performing a mode of intercropping corn and sweet potato, and guaranteeing growth and yield of the sweet potato by adopting a special sweet potato standardized cultivation technology; and after harvesting of spring cropping and autumn cropping, performing autumn planting of the sweet potato so as to guarantee adequate yield and full utilization of land. The invention has the following beneficial effects: absorption and accumulation of crops in a cadmium polluted area to cadmium metal are reduced; the cost of treatment is low; and no secondary pollution occurs.
Owner:湖南省作物研究所
Efficient composite Fenton reagent and preparation method thereof
ActiveCN108191039AEasy to prepareEasy to operateWater treatment compoundsWater contaminantsFenton reagentCerium
The invention discloses an efficient composite Fenton reagent. Ferrous oxalate is used as a main catalyst; other metal salts which are one or several kinds of materials from copper metal salts, cobaltmetal salts, aluminum metal salts, manganese metal salts, nickel metal salts, cadmium metal salts, chromium metal salts, cerium metal salts and palladium metal salts, are added; The reagent has the advantages that the cost during the sewage treatment can be obviously reduced; a complicated device is not needed; the oxidization efficiency is obviously improved; meanwhile, the requirement on the use environment is reduced; good oxidization capability is realized under the acidic and basic conditions; meanwhile, the preparation method of the composite Fenton reagent is simple; the operation is convenient.
Owner:WUHAN TEXTILE UNIV
Cadmium metal organic framework material as well as preparation method and application thereof
InactiveCN110483801ASimple preparation processImprove securityOther chemical processesWater contaminantsProtonationReaction temperature
The invention provides a cadmium metal organic framework material as well as a preparation method and application thereof. The cadmium metal organic framework material is a three-dimensional porous material, and belongs to a monoclinic system, and the space group is P21 / c; and cell parameters are as follows: alpha / deg = 90.0, beta / deg = 101.2196 (11) and gamma / deg = 90.0. The cadmium metal organicframework material disclosed by the invention is a three-dimensional porous material, and a preparation process is simple, strong in operability, low in reaction temperature and high in process safety. An anionic cadmium metal organic framework material is constructed by using a pentacarboxylic acid ligand, and carboxyl on an intermediate benzene ring does not participate in coordination and extends into pore channels in a deprotonation form, so that -COO- exposed in the pore channels and cationic dye generate strong electrostatic interaction. The anionic cadmium metal organic framework material has columnar pores, and can separate dye according to the molecular size and electrification of dye molecules. The dye subjected to adsorption separation can be desorbed, recycled and utilized.
Owner:LINYI UNIVERSITY
Preparation method for cadmium sulfide quantum dots
The invention discloses a preparation method for cadmium sulfide quantum dots. The preparation method is characterized by including steps of uniformly mixing thioacetamide liquor with the concentration of 0.05-0.15mol / L and potassium chloride liquor with the concentration of 1-3mol / L, adding thiohydracrylic acid and water to stir, and adjusting a pH (potential of hydrogen) value to 10-13 so as to obtain electrolyte; leading high-purity nitrogen into the electrolyte and maintaining for 20-40 min to remove air; and inserting a cadmium metal wire of a working electrode and a platinum wire of an auxiliary electrode into the electrolyte to stir, then applying phase step oxidation current with 10-1000 microamperes onto the cadmium working electrode, electrolyzing for 60-240min, and obtaining cadmium sulfide quantum dot stock solution after electrolyzing. The preparation method has the advantages that the method is simple, convenient, environment-friendly and fast and is low in cost, water solubility of products is good, and the prepared cadmium sulfide quantum dots are uniform in size and good in fluorescence and electrochemiluminescence performance.
Owner:NINGBO UNIV
Fertilizer for reducing rice cadmium content, preparation method and application thereof
InactiveCN109020707AReduce contentInhibition of migratory operationAlkali orthophosphate fertiliserAmmonium orthophosphate fertilisersAdditive ingredientAlcohol sugars
The invention discloses a fertilizer for reducing a rice cadmium content, a preparation method and application thereof. The fertilizer for reducing the rice cadmium content is prepared from the following ingredients in weight ratio: 10% to 16% of amino acid, 2% to 3% of compound sugar alcohol, 25% to 28% of monopotassium phosphate, 1.3% to 1.6% of instant boron, 14% to 21% of a metal chelating agent, 0.6% to 0.9% of a complexing agent, 2% to 4% of a surface active agent and the balance of water. According to the fertilizer for reducing the rice cadmium content, the preparation method and the application thereof disclosed by the invention, the content of cadmium metal in the rice can be effectively reduced, the content can be lower than the maximum limit standard of the national cereal cadmium content, and the rice quality is improved.
Owner:JIANGSU HONGYU ENVIRONMENTAL TECH CO LTD
Preparation method of cadmium metal organic complex
InactiveCN103772419AFluorescentLuminescent compositionsCadmium organic compounds6-aminonicotinic acidFluorescence
The invention discloses a preparation method of a cadmium metal organic complex. The preparation method is characterized by comprising the steps: adding a mixture of 0.2mmol of 6-aminonicotinic acid and 0.1mmol of Cd(NO3)2.4H2O into 10ml of distilled water, adding a NaOH solution dropwise until a pH value is 2-4, fully mixing, putting a mixture into a stainless steel reaction kettle with polytetrafluoroethylene as a liner, carrying out hydrothermal reaction with 2.9-3.2d under the condition of 430-440K, and slowly cooling to the room temperature to obtain a colorless tabular crystal, i.e., the cadmium metal organic complex. The cadmium metal organic complex has the fluorescence and can be applied to fluorescent materials.
Owner:宿州学院
Iron ion fluorescent probe porous material and preparation method thereof
InactiveCN105969340AImprove solubilityImprove uniformityLuminescent compositionsFluorescenceThermal stability
The invention belongs to the technical field of synthesis of functional porous coordination polymer materials, and particularly relates to an iron ion fluorescent probe porous material and a preparation method thereof. According to the method, 2-(4-carboxyphenoxy) terephthalic acid serves as an organic raw material and is subjected to a solvothermal reaction with cadmium metal salt, and a novel porous coordination polymer material with the fluorescence property and the iron ion fluorescent probe function is obtained through a ternary mixed solvent system. The material has the advantages of being good in dissolubility, high in thermal stability, high in yield, high in purity, high in crystalline quality and the like.
Owner:SHAANXI UNIV OF SCI & TECH
Synthesis of three-dimensional cadmium complex and application of three-dimensional cadmium complex as fluorescent probe
InactiveCN111848655AHigh puritySimple processOrganic chemistry methodsFluorescence/phosphorescenceBenzoic acidFluoProbes
The invention provides a cadmium metal complex, a preparation method and application of the cadmium metal complex in detection of ferric ions (Fe < 3 + >) and carbonate ions (CO3 < 2->). The chemicalformula of the complex is [Cd (L) (1, 10-Phen)] n, H2L is equal to 3, 3 '-(1H, 1' H-2, 2 '-biimidazole-1, 1'-dimethylene) benzoic acid, and 1, 10-Phen is equal to 1, 10-phenanthroline. The cadmium metal complex is of a three-dimensional structure formed by self-assembling cadmium ions, L2 <-> and 1, 10-Phen ligands. Experimental results show that the cadmium complex has good fluorescence property,can efficiently detect iron ions and carbonate ions in water, is stable in structure and reusable, and has potential application value in the field of environmental pollution detection. The complex is simple in synthesis process, mild in reaction condition and convenient to use, and a new means is provided for detection of harmful substances in the environment.
Owner:YANCHENG TEACHERS UNIV
Method for reclaiming metal cadmium from cadmium-containing waste material
InactiveCN101191158AAccelerate evaporationEasy to condenseProcess efficiency improvementReducing atmosphereOrganic matter
The invention relates to a method for reclaiming cadmium metals from cadmiferous waste materials, comprising the following steps: organic substances and water in the cadmiferous waste materials are removed; cadmium oxides in the cadmiferous waste materials after removal of the organic substances and the water are reduced to elementary cadmiums in reducing atmosphere; the elementary cadmiums are then evaporated into cadmium vapor; the cadmium vapor is condensed into cadmium metals, wherein, the reducing atmosphere is formed by reducing gas which comprises main air flow and auxiliary air flow; the main air flow and the auxiliary air flow conversely blows towards the cadmiferous waste materials after removal of the organic substances and the water; linear velocity of the auxiliary air flow is higher than that of the main air flow. The method for reclaiming the cadmium metals from the cadmiferous waste materials provided by the invention has the advantages of low energy consumption and simple procedures.
Owner:浙江文国重工机械有限公司
Cadmium metal organic framework complex and preparation method thereof
ActiveCN107778495AImprove stabilityHigh sensitivityFluorescence/phosphorescenceLuminescent compositionsSpace groupTetrachloride
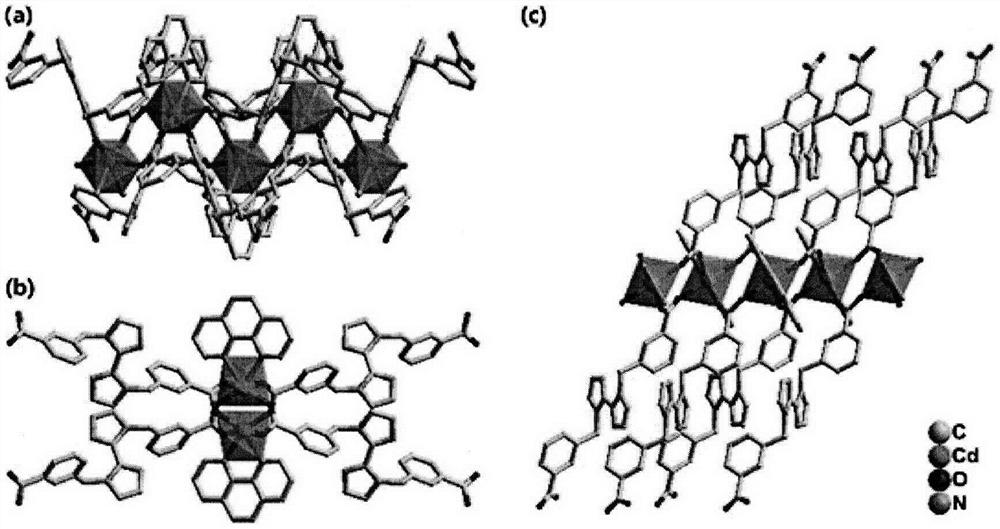
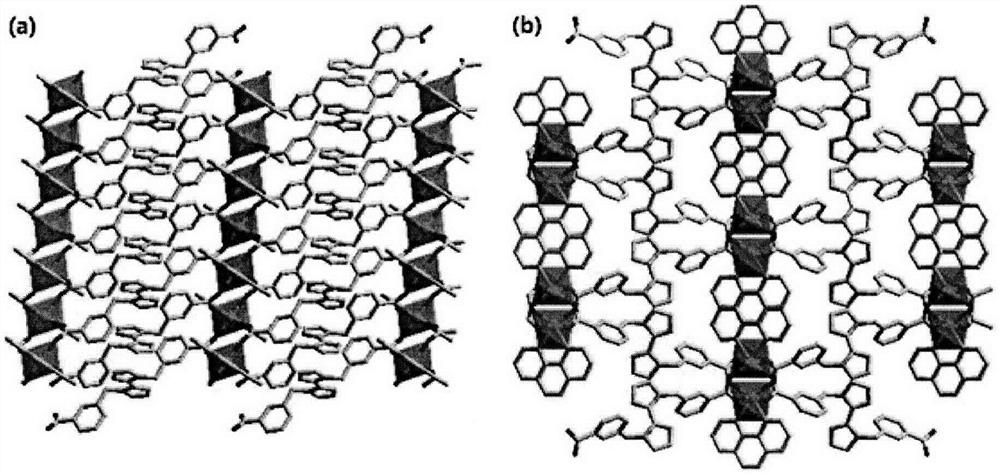
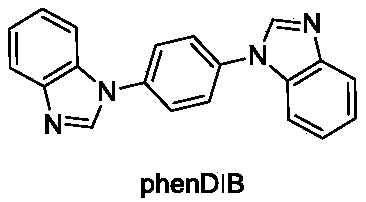
The invention relates to a cadmium metal organic framework complex. The cadmium metal organic framework complex is cooperatively formed by a flexible carboxylate ligand and a rigid imidazole ligand, the chemical formula is {Cd3(L)(phenDIB).6DMF}n, and the molecular formula is C90H96Cd3N10O25; the cadmium metal organic framework complex is a monoclinic space group C2 / c; and cell parameters are as follows: a / angstrom is 29.403(3), b / angstrom is 20.8279(19), c / angstrom is 16.9132(17), alpha is 90 degrees, beta is 111.067(2) degrees, V / angstrom is 6665.2(16), the density is 1.412mg / m<3>, the lightabsorption coefficient is 0.730mm<-1>, and the number of molecules in a single cell is 4. By utilizing the cadmium metal organic framework complex, the high-sensitivity selective detection of high-toxicity carbon tetrachloride molecules and hexavalent heavy metal chromium ions can be realized; and the cadmium metal organic framework complex has good application prospects.
Owner:NINGBO UNIV
Method for preparing cadmium sulphide/graphite oxide composite material in solid phase
The invention relates to a method for preparing a cadmium sulphide / graphite oxide composite material in a solid phase and belongs to the field of preparation of nano materials. The method provided by the invention comprises that cadmium metal salt, graphite oxide and thioacetamide react at room temperature, and low heat solid-phase synthesis of the cadmium sulphide / graphite oxide composite material is achieved. The low heat solid-phase chemical method for preparing the cadmium sulphide / graphite oxide composite material has the characteristics of easy operation, no solvent, high yield, low cost and simple synthetic process; and the cadmium sulphide / graphite oxide composite material prepared by the invention has large specific surface area and high reaction activity and has a broad application prospect in the fields of solar cells, photocatalysis and the like.
Owner:XINJIANG UNIVERSITY
Preparation and photocatalytic application of polyacid group cadmium metal hybrid material constructed by phosphomolybdic acid
ActiveCN113457742AExcellent photocatalytic water splitting hydrogen production performanceHigh catalytic activityOrganic-compounds/hydrides/coordination-complexes catalystsHydrogen productionPhosphomolybdic acidCadmium Metal
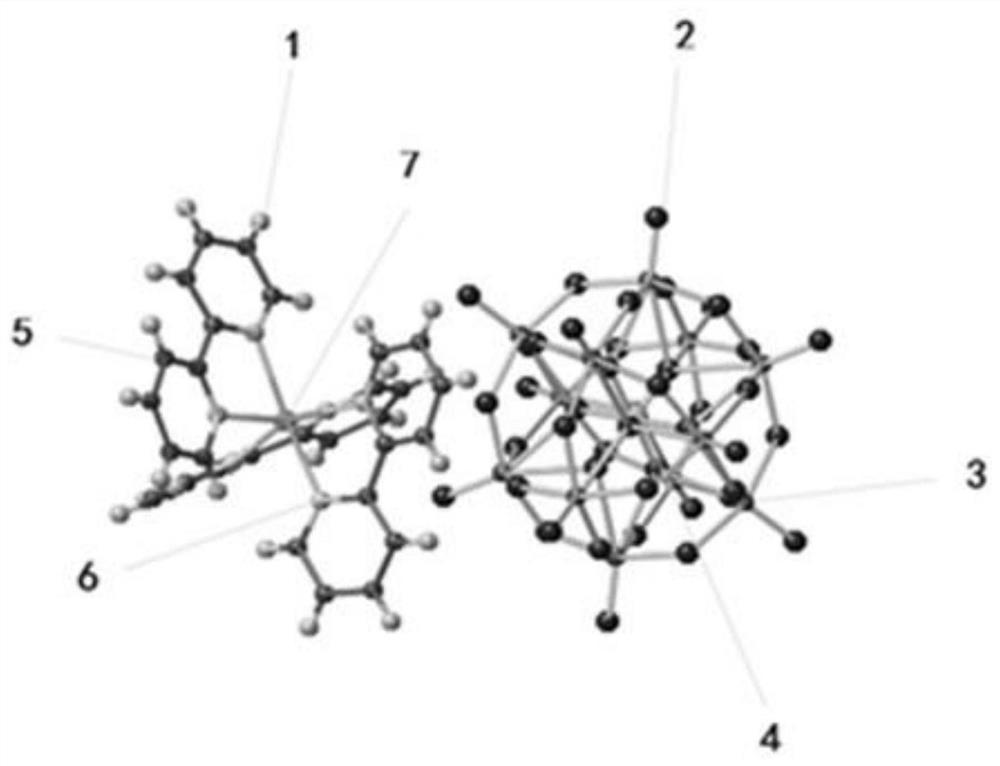
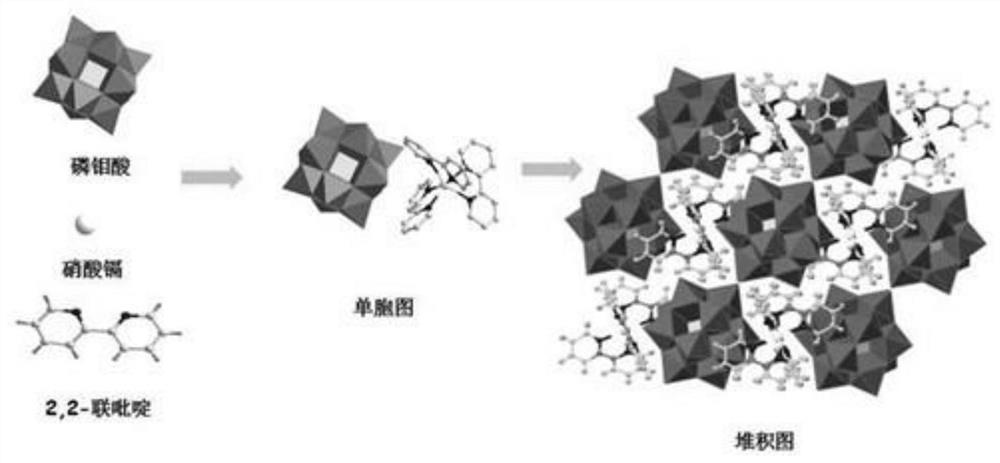
The invention discloses preparation and photocatalytic application of a polyacid-based cadmium metal hybrid material constructed by phosphomolybdic acid, and relates to a polyacid-based cadmium metal organic hybrid material constructed by phosphomolybdic acid. The invention aims to solve the problems that an existing photocatalytic hydrogen production material does not produce hydrogen or the hydrogen production amount is low and the like due to the fact that the energy gap of the photocatalytic hydrogen production material synthesized in the prior art is relatively wide, photo-induced electron holes are easy to compound, the surface reaction of proton reduction is relatively difficult and the like. The invention designs and develops a polyacid group cadmium metal organic hybrid material constructed by phosphomolybdate, the chemical formula of the polyacid group cadmium metal organic hybrid material constructed by phosphomolybdate is (PMo12O40)[Cd(C10H8N2)3], and C10H8N2 is 2,2-dipyridyl. The synthesis method comprises the following steps: dissolving phosphomolybdic acid, cadmium nitrate and an organic ligand (2,2-dipyridyl) into deionized water, adjusting the pH value to 1.5, and conducting reacting for 4 days at the temperature of 160 DEG C. According to the invention, the polyacid group cadmium metal organic hybrid material constructed by phosphomolybdate can be obtained.
Owner:HARBIN UNIV OF SCI & TECH