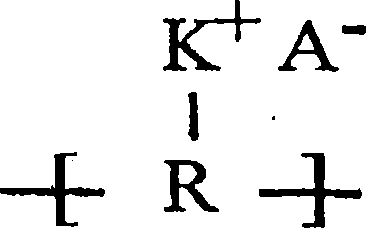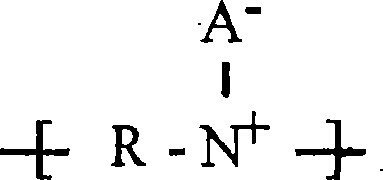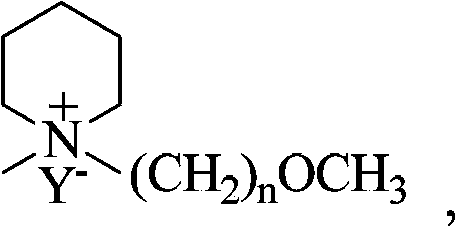Patents
Literature
75 results about "Cation composition" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
Ionic polymer membranes
InactiveUS20060049102A1Stable materialLarge specific surface areaCellsFatty/oily/floating substances removal devicesCarboxylic acidStructural unit
Compositions and processes are disclosed for economical separation of fluid mixtures. Broadly, the present invention discloses ionic polymer compositions that are useful for perm-selective membrane separations. More particularly, ionic polymers of the invention comprise a plurality of repeating structural units having as a constituent part thereof organic ionic moieties consisting of nitrogen containing anions and / or cations. In the form of non-porous membranes, ionic polymers of the invention facilitate recovery of purified organic and inorganic products from fluid mixtures by means of perm-selective membrane separations. The present invention also provides methods for forming the ionic polymers, for example by treating selected nitrogen-containing organic polymers with acids, or treating a polymeric material comprising a plurality of carboxylate groups with an amine. Ionic polymer compositions of the invention are particularly useful for simultaneous recovery of a permeate product of an increased concentration, and a desired non-permeate stream, from a fluid mixture containing at least two compounds of different boiling point temperatures.
Owner:BP CORP NORTH AMERICA INC +1
Ionic liquid prepared through diimine (vikane) and (perfluoroalkglsulfonyl fluorosulfonyl group) imine alkali salt
InactiveCN101747243APromote hydrolysisLow melting pointElectrolytic capacitorsGroup 5/15 element organic compoundsSulfoniumLithium-ion battery
The invention provides an ionic liquid consisting of the anions of diimine (vikane) and (perfluoroalkglsulfonyl fluorosulfonyl group) imine and the cation of sulfonium salt, ammonium salt or phosphor salt, as well as method for preparing the ionic liquid through diimine (vikane) and (perfluoroalkglsulfonyl fluorosulfonyl group) imine alkali salt. The ionic liquid can be used as an electrolyte material and applied on the fields like secondary lithium ion batteries, super capacitors, etc.
Owner:HUAZHONG UNIV OF SCI & TECH
Sintering resistant, low conductivity, high stability thermal barrier coating/environmental barrier coating/environmental barrier coating system for a ceramic-matrix composite (CMC) article to improve high temperature capability
ActiveUS20060121295A1Sufficient thermalSufficient environmental protectionLiquid surface applicatorsBlade accessoriesCoating systemThermal barrier coating
In accordance with an embodiment of the invention, a thermal barrier coating (TBC) for inclusion in a thermal barrier coating / environmental barrier coating system (TBC / EBC system) for use on a silicon containing material substrate is provided. The TBC comprises a compound having a primary constituent portion and a stabilizer portion stabilizing said primary constituent. The primary constituent portion of the TBC comprises hafnia present in an amount of at least about 5 mol % of the primary constituent. The stabilizer portion of said thermal barrier coating comprises at least one metal oxide comprised of cations with a +2 or +3 valence present in the amount of about 10 to about 40 mol % of the thermal barrier coating.
Owner:GENERAL ELECTRIC CO
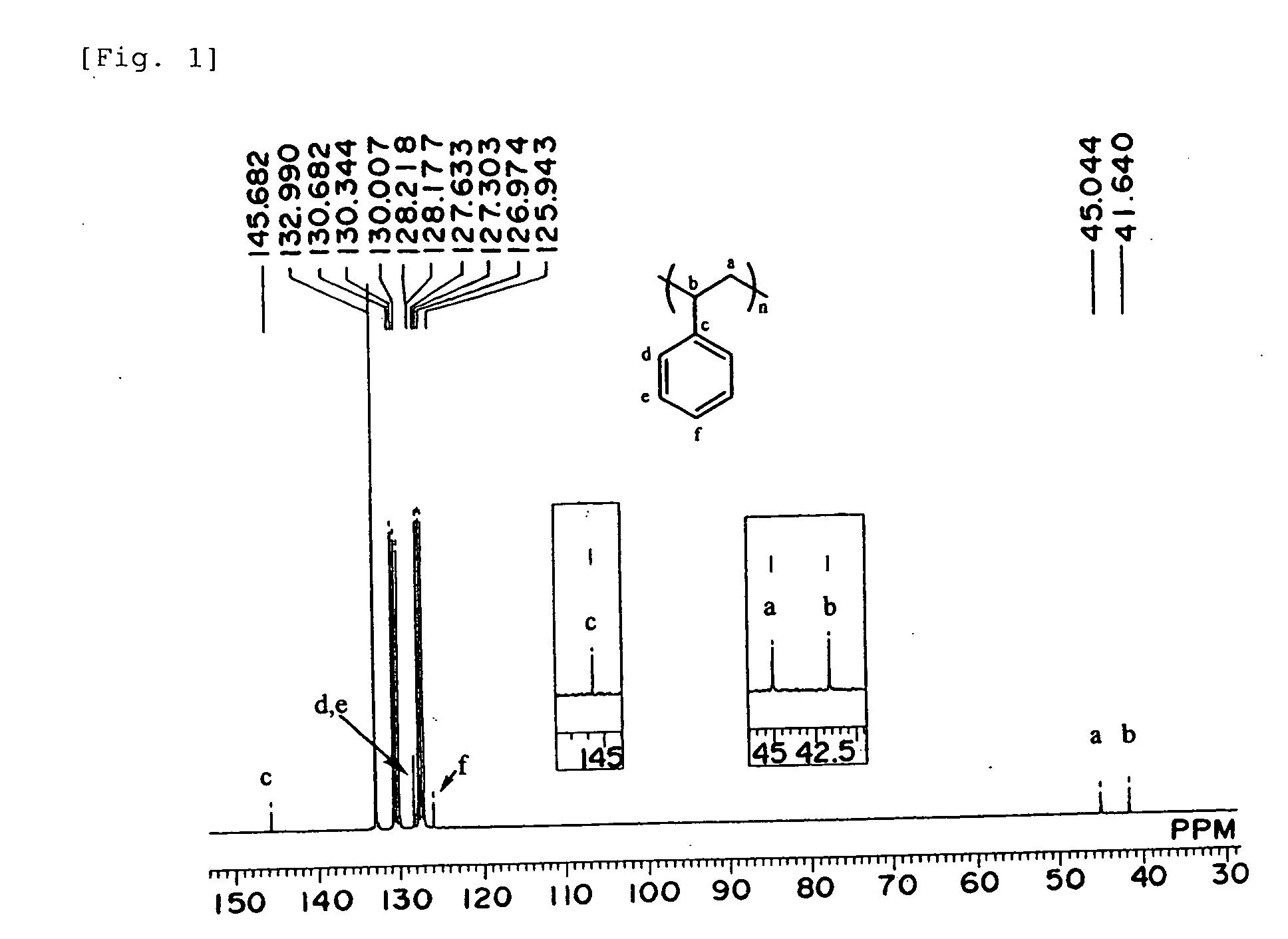
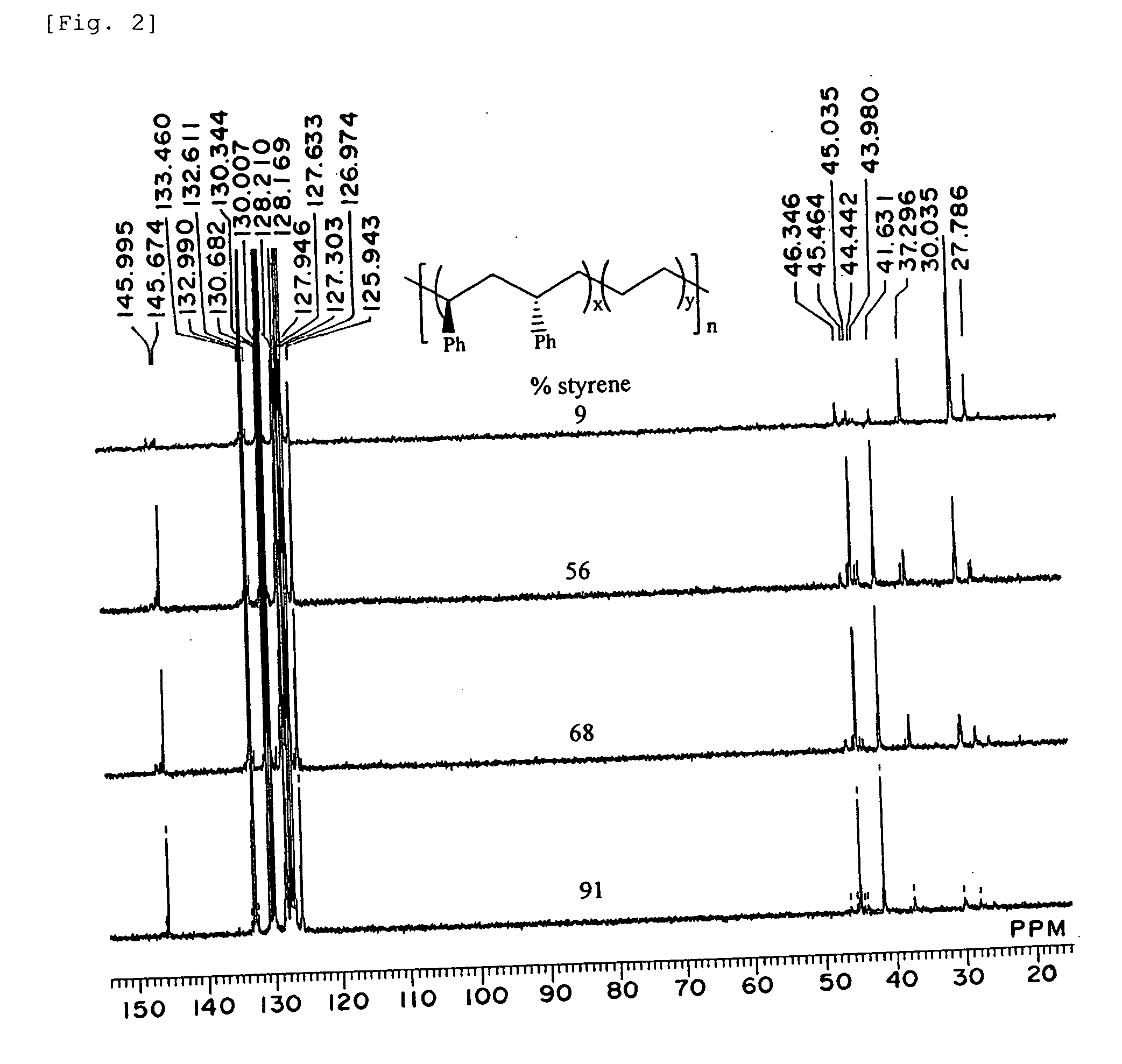
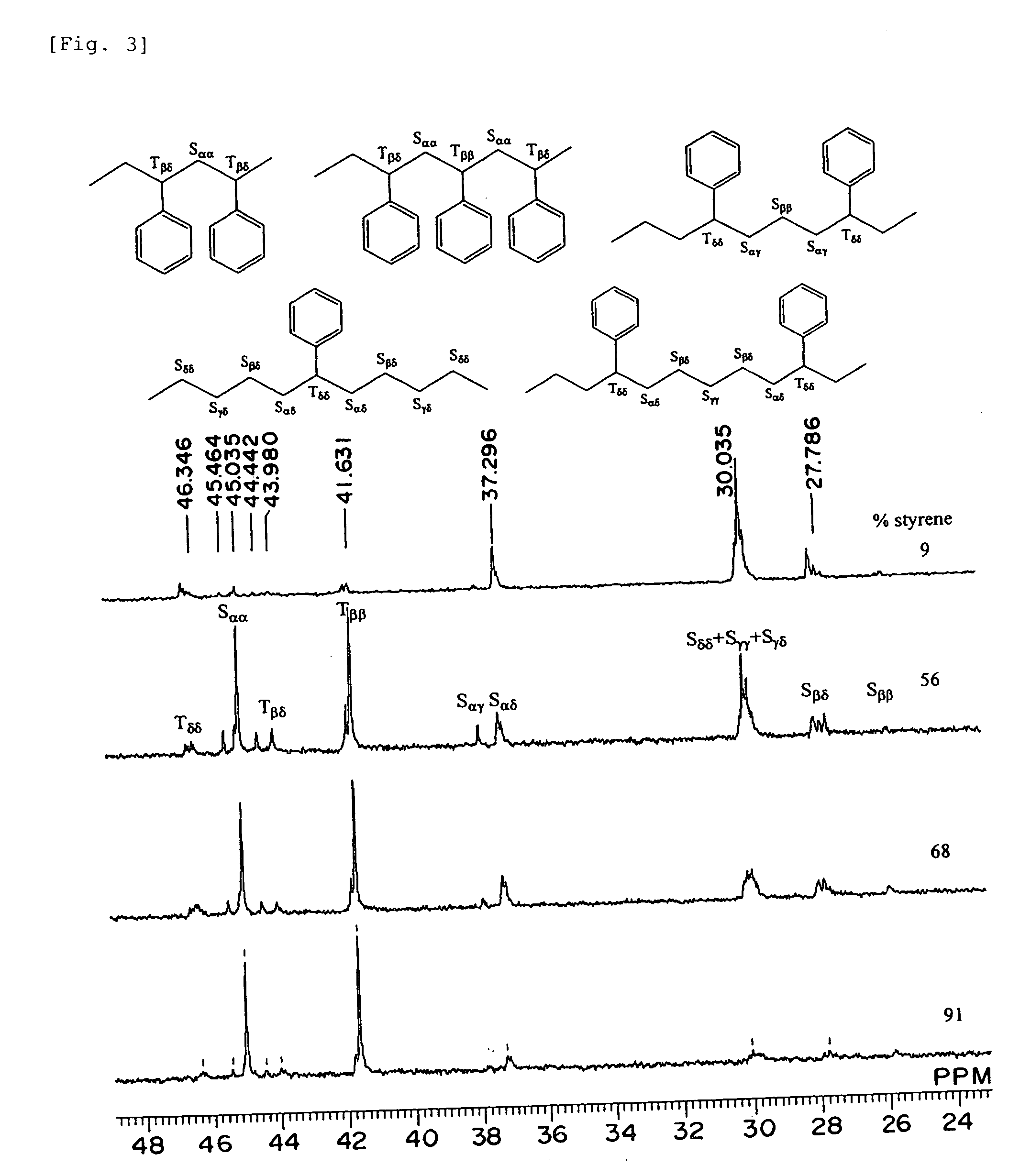
Polymerization Catalyst Compositions Containing Metallocene Complexes and Polymers Produced by Using the Same
InactiveUS20070232758A1Quality improvementWide applicationOrganic-compounds/hydrides/coordination-complexes catalystsPolymerization catalystsMetal
The present invention provides a novel catalyst composition comprising a metallocene complex, and a novel producing method for various polymer compounds. Preferably, the invention provides a novel polymer compound, and a producing method thereof. Specifically, the invention provides a polymerization catalyst composition, comprising: (1) a metallocene complex represented by the general formula (I), including: a central metal M which is a group III metal atom or a lanthanoid metal atom; a ligand Cp* bound to the central metal and including a substituted or unsubstituted cyclopentadienyl derivative; monoanionic ligands Q1 and Q2; and w neutral Lewis base L; and (2) an ionic compound composed of a non-ligand anion and a cation: where w represents an integer of 0 to 3.
Owner:RIKEN
Biological protein hair fiber preparation method by dissolving animal hair and cellulose material mixture
InactiveCN1884642AReduce lossesReduce direct lossesConjugated cellulose/protein artificial filamentsCelluloseAdjuvant
The invention discloses a biological protein wool fiber preparing method, which comprises the following steps: blending animal fur and fiber to produce soluble liquid in the ion liquid with positive and negative ion of fusing condition at 100 deg.c and positive ion to form salt; solidifying in the water, carbinol, alcohol, cyanoethyl or (NH4)2SO4 solution; adding adjuvant in the soluble liquid; solidifying in the water, carbinol, alcohol, cyanoethyl or 30 percent (NH4)2SO4 solution; evolving biological protein fur fiber.
Owner:INST OF PROCESS ENG CHINESE ACAD OF SCI
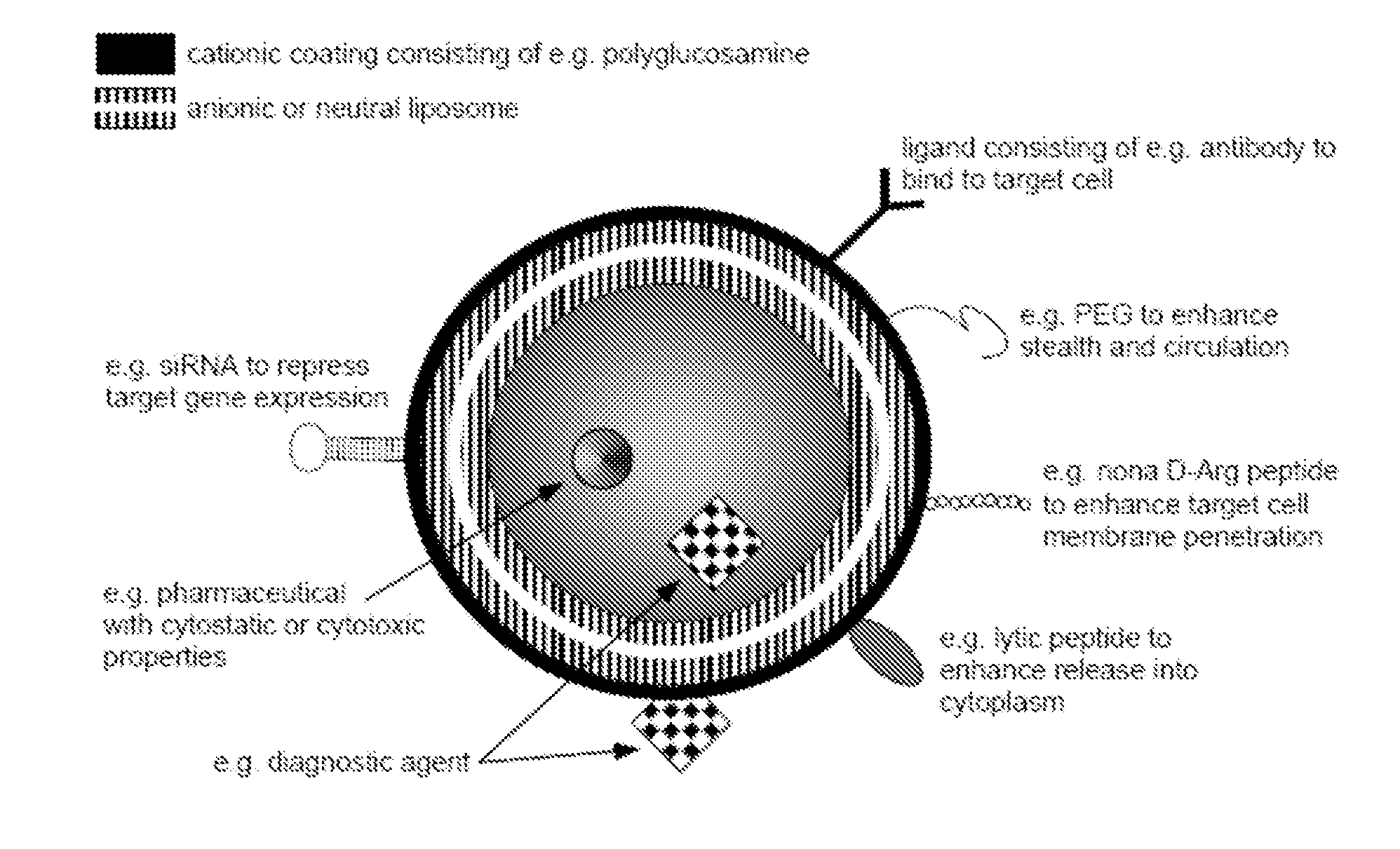
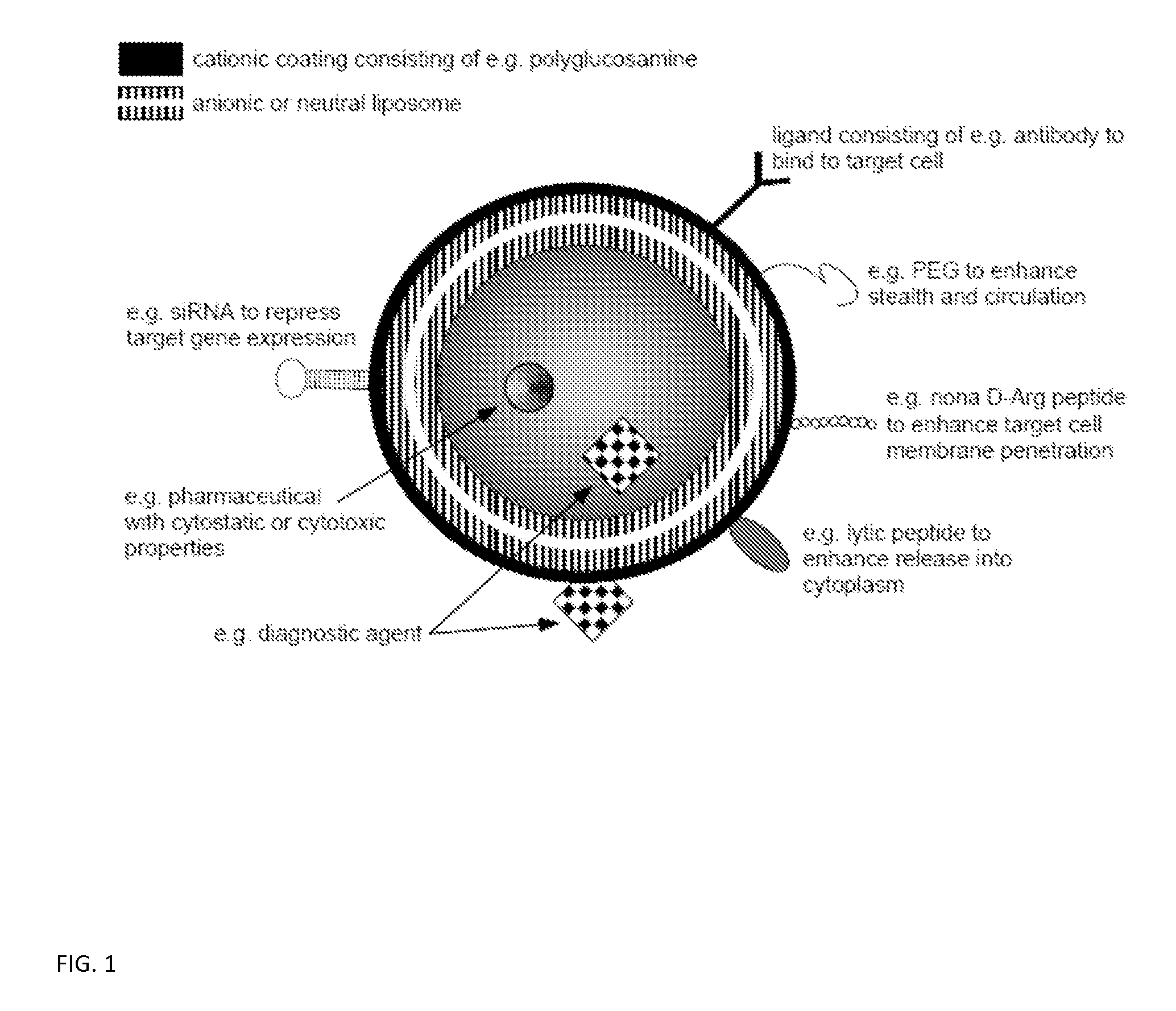
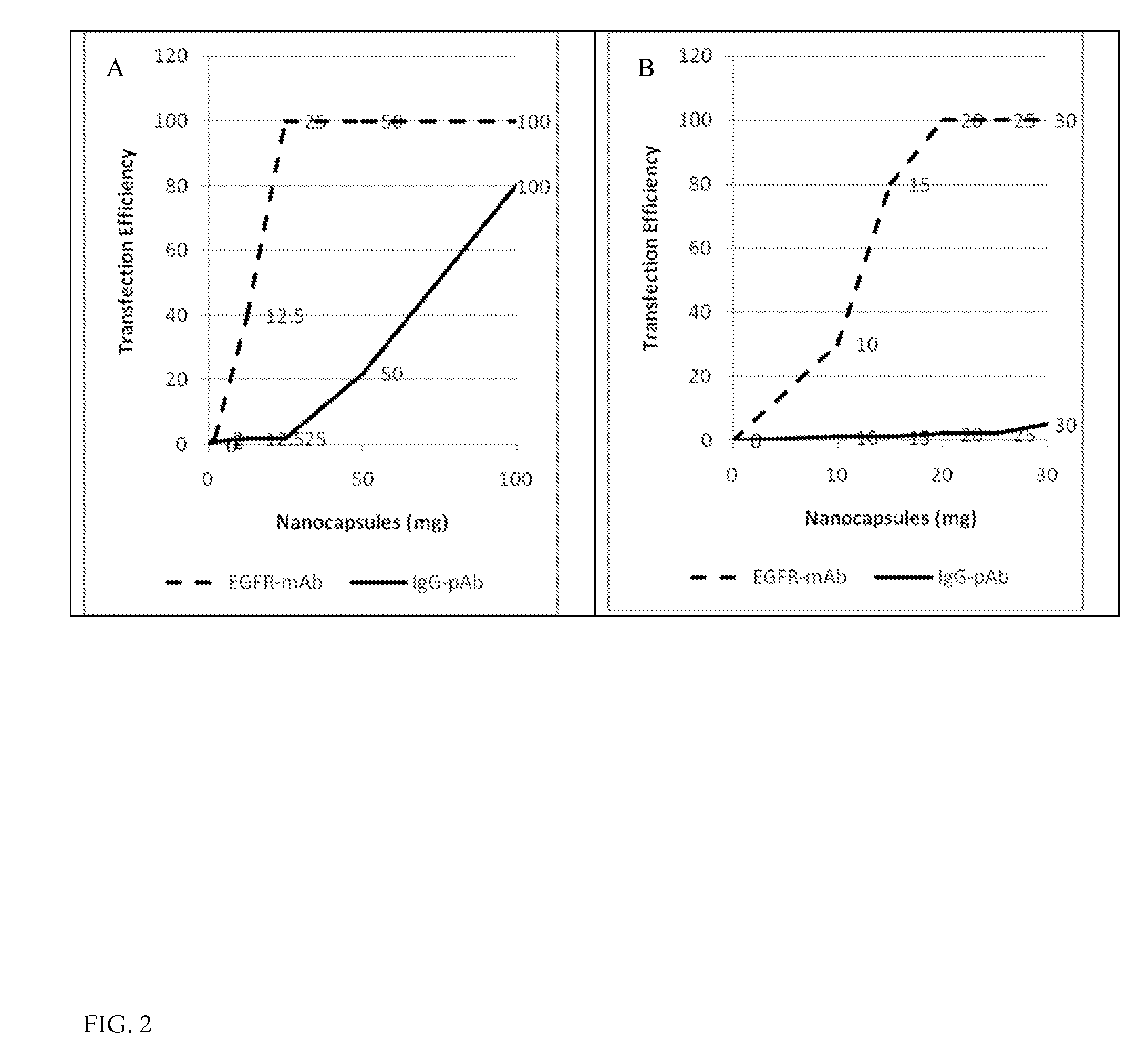
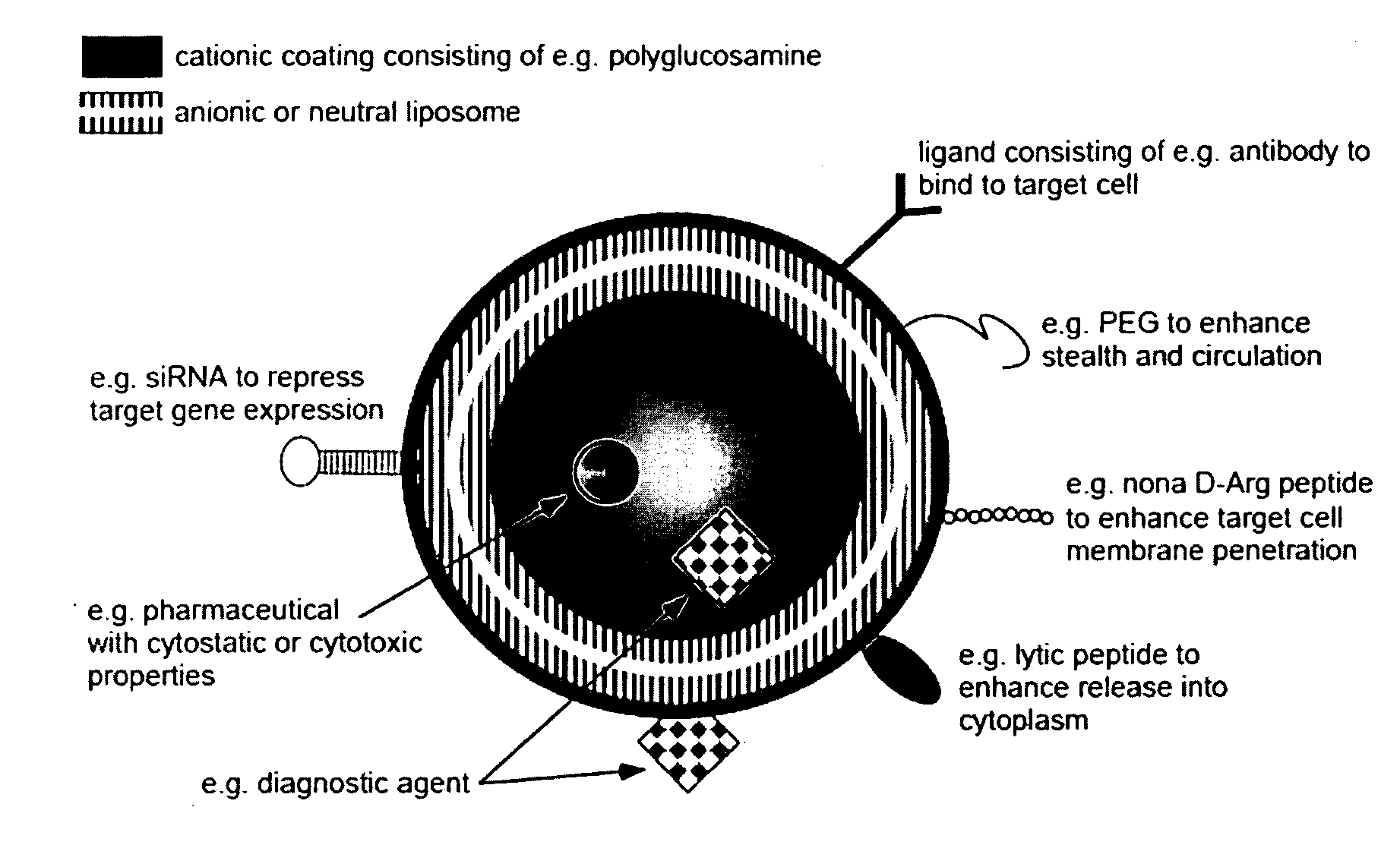
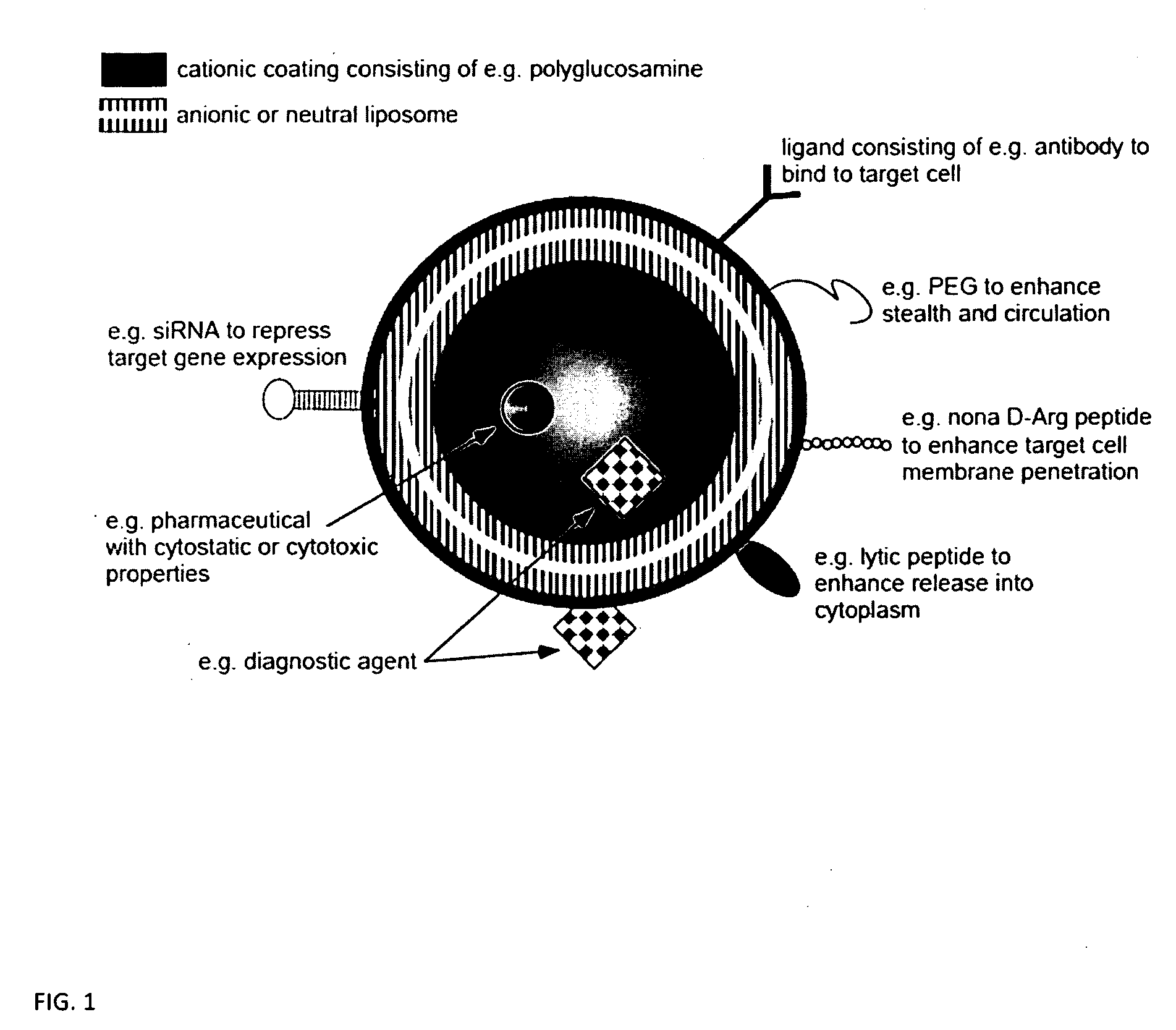
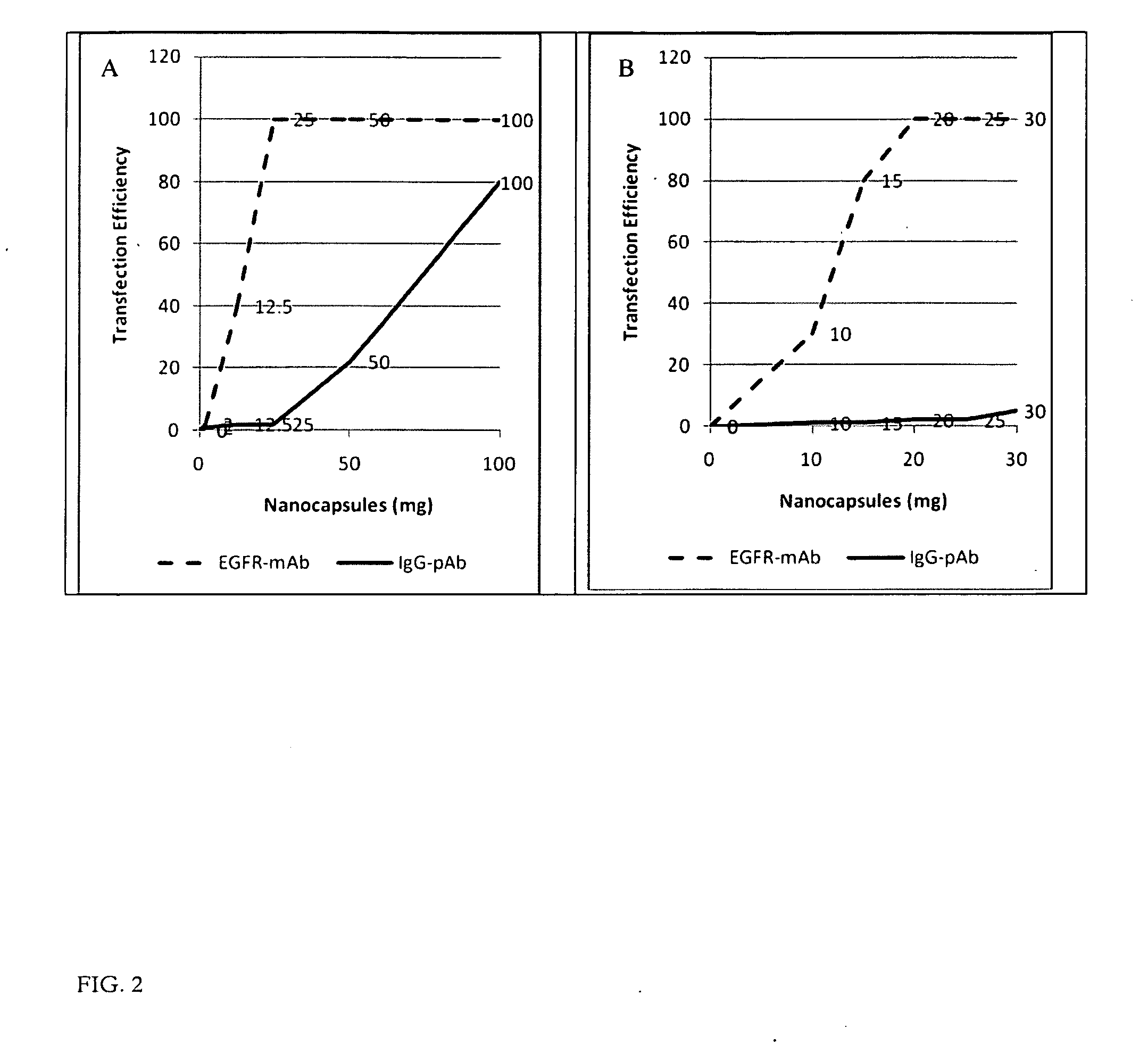
Ligand Targeted Nanocapsules for the delivery of RNAi and other Agents
InactiveUS20100105139A1Prolong blood circulation timeReduce systemic toxicityPolypeptide with localisation/targeting motifVectorsDiagnostic agentCell membrane
A carrier system for the delivery of therapeutic and / or diagnostic agents is described. The carrier system is comprised of ligands and a biodegradable polycation for complexing polyanionic molecules such as RNAi, said polycation forming a coating on the outer surface of anionic or neutral liposomes. Also disclosed is a method for using the composition to deliver to target cells and enhance cell membrane penetration of therapeutic and / or diagnostic agents.
Owner:SPANJAARD REMCO ALEXANDER
Ionic Polymer Membranes
InactiveUS20080223785A1Stable materialLarge specific surface areaMembranesSemi-permeable membranesPermselective membraneStructural unit
Compositions and processes are disclosed for economical separation of fluid mixtures. Broadly, the present invention discloses ionic polymer compositions that are useful for perm-selective membrane separations. More particularly, ionic polymers of the invention comprise a plurality of repeating structural units having as a constituent part thereof organic ionic moieties consisting of nitrogen containing anions and / or cations. In the form of non-porous membranes, ionic polymers of the invention facilitate recovery of purified organic and inorganic products from fluid mixtures by means of perm-selective membrane separations. The present invention also provides methods for forming the ionic polymers, for example by treating selected nitrogen-containing organic polymers with acids, or treating a polymeric material comprising a plurality of carboxylate groups with an amine. Ionic polymer compositions of the invention are particularly useful for simultaneous recovery of a permeate product of an increased concentration, and a desired non-permeate stream, from a fluid mixture containing at least two compounds of different boiling point temperatures.
Owner:MILLER JEFFREY T +3
Method for producing field effect transistor, field effect transistor, display device, image sensor, and X-ray sensor
Owner:SAMSUNG DISPLAY CO LTD
Glass, glass material for pressing moulding,optical member blank and optical membe
ActiveCN105693087AImprove thermal stabilityImprove homogeneityOptical elementsPolymer scienceRefractive index
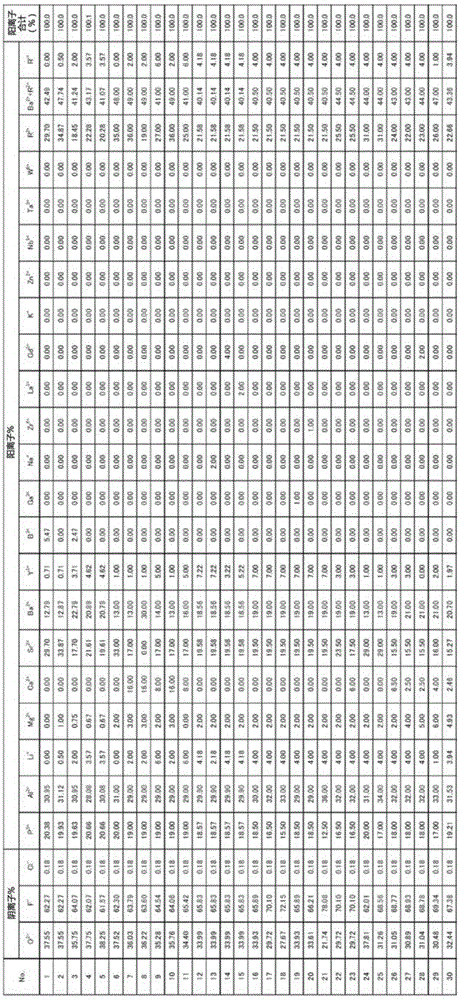
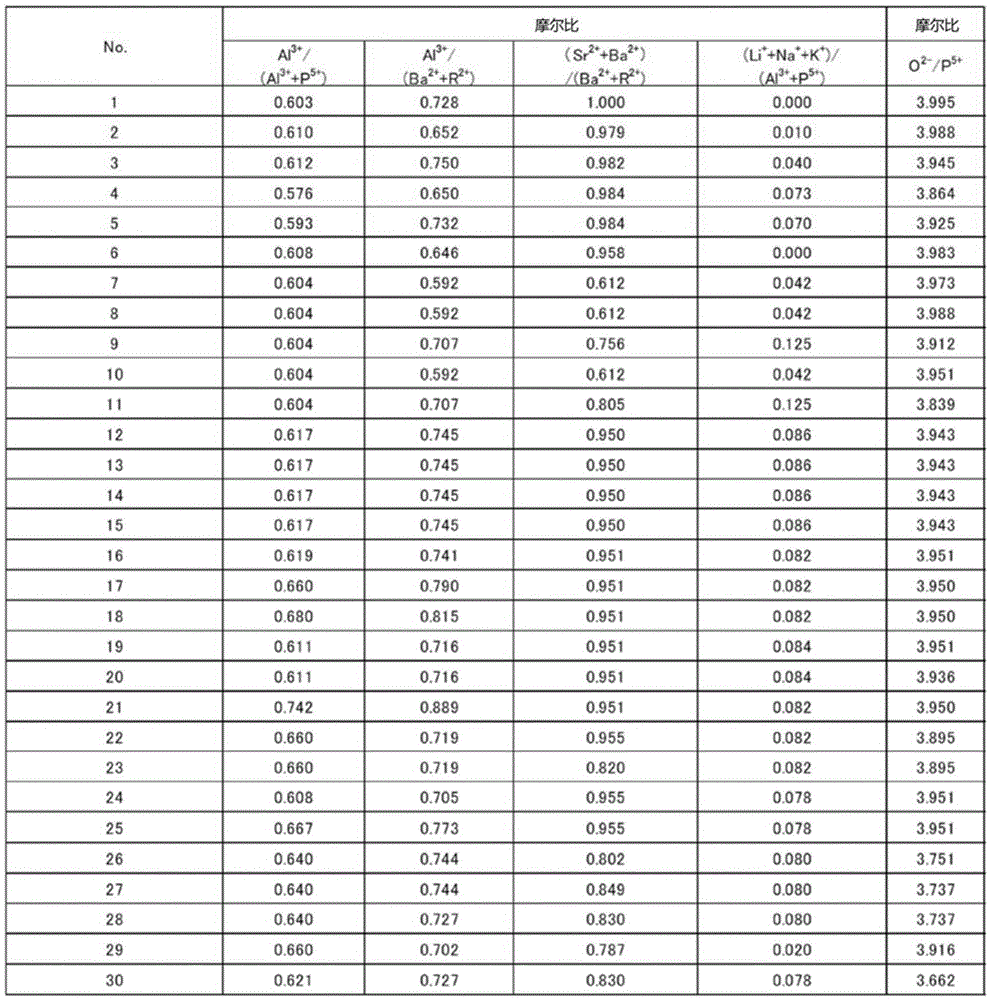
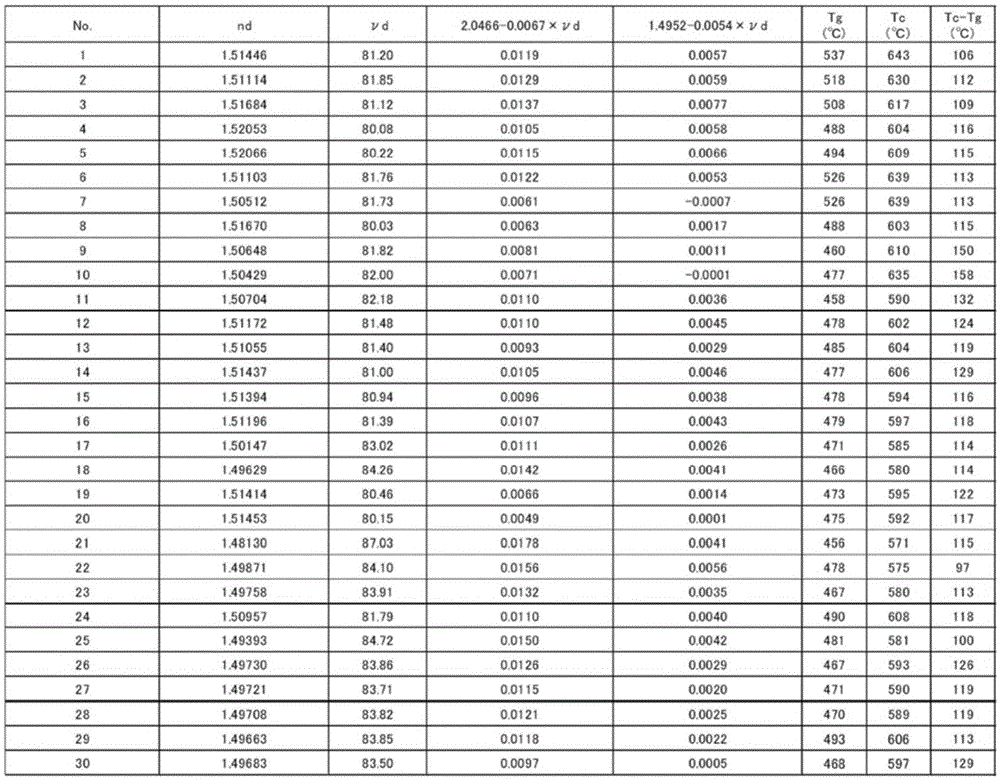
The present invention provides a thermal stability, homogeneity and acid resistance of excellent high refractive index low dispersion fluoride phosphate glass. A glass, which, as a cation composition contains at least P5 +, Al3 +, Ba2 + and R2 + R2 + is described from magnesium 2 +, Ca2 + and Sr2 + and Zn2 + more than one, as anion composition containing O2 - and F - at least, in the cation composition proportion, relative to the content of Al3 + Al3 + and the molar ratio of the total content of P5 + Al3 + / (Al3 + + P5 +) in the range of 0.56 ~ 0.56, relative to the content of Al3 + Ba2 + and the molar ratio of the total content of R2 + Al3 + / +) (Ba2 + + R2 more than 0.58, the total content of Ba2 + and Sr2 + relative to Ba2 + and the molar ratio of the total content of R2 + (Ba2 + + Sr2 +) / (Ba2 + + + R2) of 0.60 above, Li +, Na + and K + relative to the total content of Al3 + and the molar ratio of the total content of P5 + (Li++ Na++ K +) / (P5 + + Al3 +) less than 0.39, the range of the refractive index nd for more than 1.47 and 1.53 below, and the abbe number argument d range is 80 ~ 95.
Owner:HOYA CORP
Method for producing field effect transistor, field effect transistor, display device, image sensor, and x-ray sensor
Owner:SAMSUNG DISPLAY CO LTD
Process for producing conjugated diene polymer
InactiveUS20110263803A1Efficient productionOrganic-compounds/hydrides/coordination-complexes catalystsNon-coordinating anionHigh activity
Disclosed is a process for producing a conjugated diene polymer having a very high content of cis-1,4 structures by using an yttrium compound-containing catalyst that is relatively easy to handle and has a high activity. Specifically disclosed is a process for producing a conjugated diene polymer containing cis-1,4 structures at a ratio of 99% or higher, which is characterized by polymerizing a conjugated diene in the presence of a catalyst produced from (A) a specific yttrium compound, (B) an ionic compound consisting of a non-coordinating anion and a cation, and (C) an organoaluminum compound.
Owner:UBE IND LTD
Preparation method of porous carbon material based on ionic heat process
ActiveCN103395769ALarge specific surface areaRaw materials are cheap and easy to getCarbon preparation/purificationPorous carbonBiological materials
The invention discloses a preparation method of a porous carbon material based on an ionic heat process. The preparation method comprises the following steps: (1) adding a biological material into ionic liquid to form a mixed solution, wherein the ionic liquid consists of anions and cations, and the cations are one of formulas (I) to (IV); and (2) heating the solution to 140 to 260 DEG C, preserving heat for 10 to 48 hours, cooling and performing aftertreatment to obtain the porous carbon material. According to the preparation method of the porous carbon material based on the ionic heat process, the preparation process is simple, energy consumption is low, any severe conditions are not needed, products are easy to purify, the product structure is easy to adjust, and a great potential application value is achieved.
Owner:ZHEJIANG UNIV
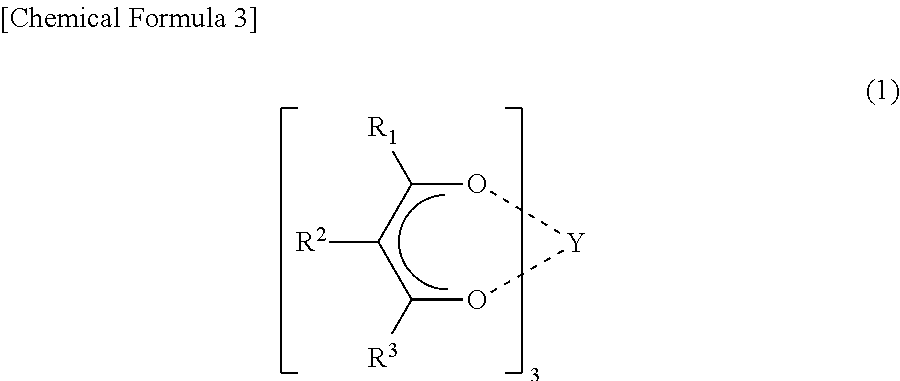
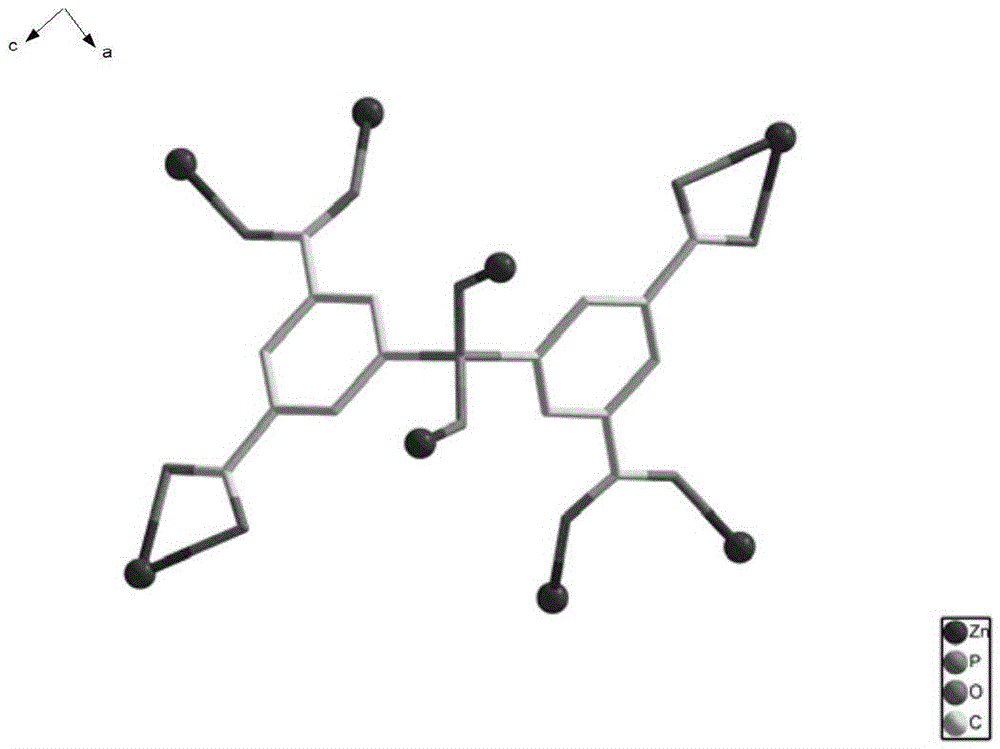
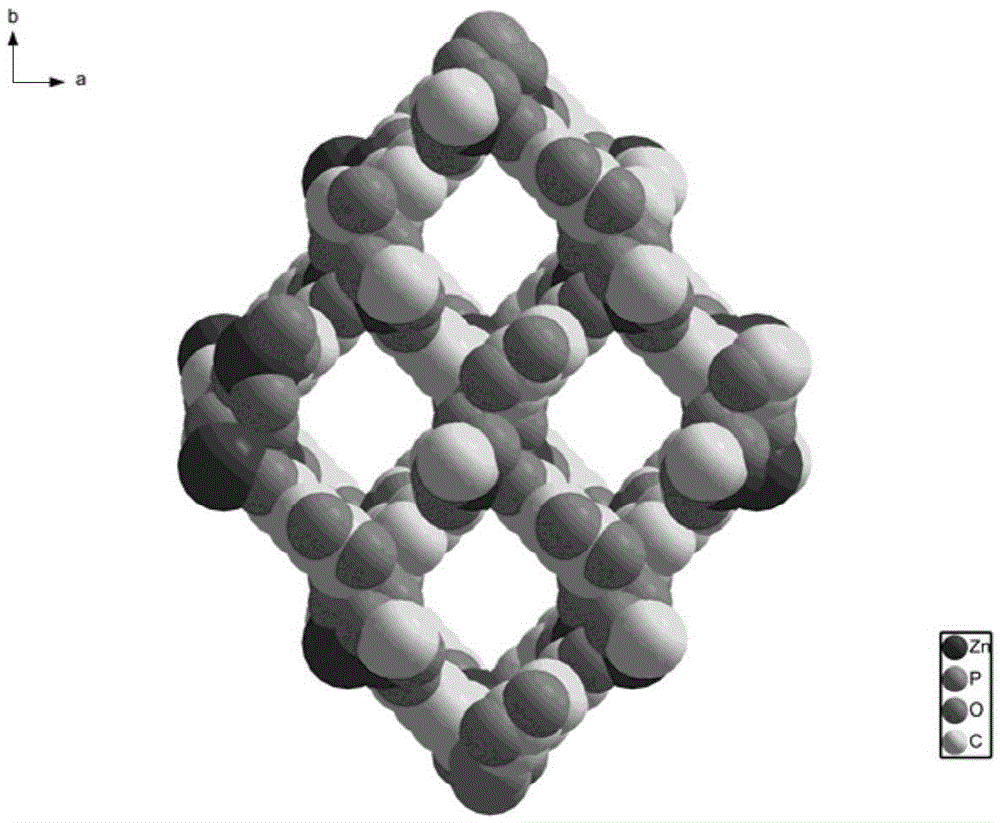
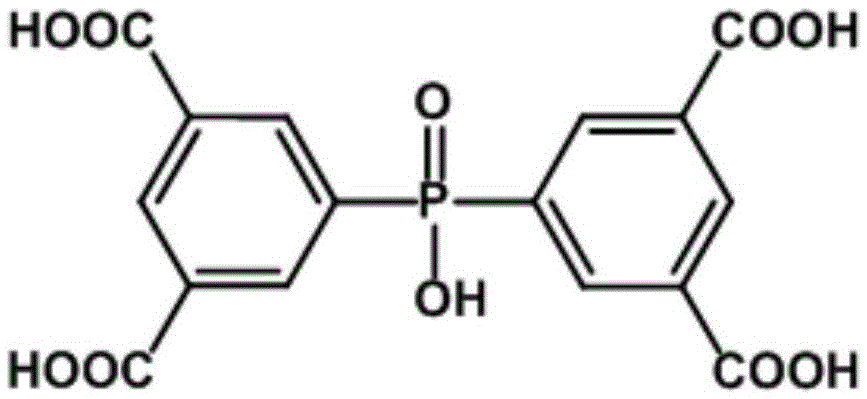
Preparation of a metal-organic framework material used for natural-gas purification
The invention provides preparation of a metal-organic framework material used for natural-gas purification. A microporous zinc-based zinc-organic framework material is prepared by a solvothermal synthesis method. The material is high in water stability, comprises bis(3,5-dicarboxyphenyl) phosphinate ions, zinc ions and a dimethyl ammonium cation, and is simple in preparation. The material has high adsorption capacities for light hydrocarbons comprising methane, ethylene, acetylene, ethane and propane at room temperature under atmospheric pressure. The methane can be selectively separated from the ethylene, the acetylene, the ethane and the propane through the material. The material can be used for natural-gas purification and is low in cost, convenient in operation and good in application effects.
Owner:FUJIAN INST OF RES ON THE STRUCTURE OF MATTER CHINESE ACAD OF SCI
Ligand Targeted Nanocapsules for the delivery of RNAi and other Agents
InactiveUS20100104622A1Prolong blood circulation timeReduce systemic toxicityPharmaceutical non-active ingredientsCapsule deliveryDiagnostic agentCell membrane
A carrier system for the delivery of therapeutic and / or diagnostic agents is described. The carrier system is comprised of ligands and a biodegradable polycation for complexing polyanionic molecules such as RNAi, said polycation forming a coating on the outer surface of anionic or neutral liposomes. Also disclosed is a method for using the composition to deliver to target cells and enhance cell membrane penetration of therapeutic and / or diagnostic agents.
Owner:SPANJAARD REMCO ALEXANDER
Method for preparing liquid alkylated diphenylamine compound by ionic liquid catalyst
InactiveCN101863774ALight color and stablePost-processing is simpleChemical recyclingAmino compound preparation by condensation/addition reactionsDistillationAniline
The invention discloses a method for preparing liquid alkylated diphenylamine compound by an ionic liquid catalyst. Alkylate reaction is carried out on diphenylamine and alkene under the action of the catalyst; the catalyst is separated and recovered after the reaction finishes; steps of washing, pressure distillation for removing excessive alkene and water post treatment are carried out on the product; and then light yellow liquid alkylated diphenylamine compound is obtained. Pressure of the alkylated reaction is 1 atmospheric pressure and temperature thereof is 80-150 DEG C. The catalyst is ironic liquid catalyst, which is composed of metal halogenide anions and quaternary cations. Dioctyl diphenylamine content in liquid alkylated diphenylamine compound is less than 25% wt and diphenylamine content therein is less than 1%wt. Residual diphenylamine content in the obtained product is less than 1%wt and the ioctyl diphenylamine in the product is less than 25%wt; color and luster of the product is light and stable. The invention has simple and easy post-processing, does not generate a large amount of aluminiferous waste water and is easy to carry out industrialized promotion; in addition, the invention has high catalyst efficiency, repeatable use and easy separation from the product.
Owner:JIANGSU UNIV OF SCI & TECH
Metal coordination polycation gene vector and preparation method and application thereof
ActiveCN106496555AImprove bindingIncrease forceOther foreign material introduction processesStimuli responsiveCell membrane
The invention discloses a metal coordination polycation gene vector. The metal coordination polycation gene vector consists of a coordinated metal, a coordinated unit, a stimuli-responsive unit and polycations, and the structural general formula of the metal coordination polycation gene vector is as shown in the specification, wherein M represents the coordinated metal; a dimethyl pyridylamine component represents the coordinated unit; a disulfide bond component represents the stimuli-responsive unit; a ball represents the polycations; n represents the quantity of metal ion ligands grafted by one polycation molecule, n is 1 to 200; and the metal coordination polycation gene vector is used for non-virus gene vectors. The metal coordination polycation gene vector has the advantage that the metal ion ligands containing stimuli-responsive radicals are modified on the polycations, so that the DNA binding capacity of the polycations and the acting force with cytomembranes are greatly improved; after the compound enters cells, the metal ion ligands and the polycations are disconnected, DNAs are released and expressed so as to show extremely high transfection efficiency, so that the application of the polycations in the aspect of the non-virus gene vectors is greatly expanded due to metal coordination monomers in the invention.
Owner:NANKAI UNIV
Ionic polymer membranes
Owner:BP CORP NORTH AMERICA INC +1
Water- and oil-repellent, antistatic composition
InactiveUS7361706B2Improve featuresEfficient combinationElectroconductive/antistatic filament manufactureOther chemical processesPolymer scienceDivalent metal
A water- and oil-repellent, antistatic composition comprises (a) at least one nonpolymeric ionic salt consisting of (i) at least one cation selected from the group consisting of monovalent metal cations, divalent metal cations, and organic onium cations, and (ii) at least one weakly coordinating anion, the conjugate acid of the anion having an acidity greater than or equal to that of a hydrocarbon sulfonic acid, and with the proviso that the anion is organic or fluoroorganic when the cation is a metal; (b) at least one fluorochemical repellency-imparting additive or repellent; and (c) at least one insulating material. The composition exhibits good antistatic and repellency characteristics.
Owner:3M INNOVATIVE PROPERTIES CO
Developing member, method of producing developing member, process cartridge, and electrophotographic apparatus
ActiveUS20170139346A1High quality imagingElectrographic process apparatusEngineeringCation composition
A developing member includes a conductive substrate, a conductive elastic layer on the conductive substrate, and a plurality of insulating domains on the conductive elastic layer, wherein the surface of the developing member includes the surfaces of the insulating domains and an exposed portion of the conductive elastic layer not covered with the insulating domains. The conductive elastic layer contains a resin and an anion, wherein the resin has a specific cationic structure synthesized from an ion conductive agent composed of a cation including at least one reactive functional group and an anion and a compound that can react to the ion conductive agent.
Owner:CANON KK
Lithium-sulfur battery
InactiveCN106876777ASolve the problem of dissolving into the electrolyte and diffusing to the negative electrodeImprove instabilityLi-accumulatorsSolvent moleculeAll solid state
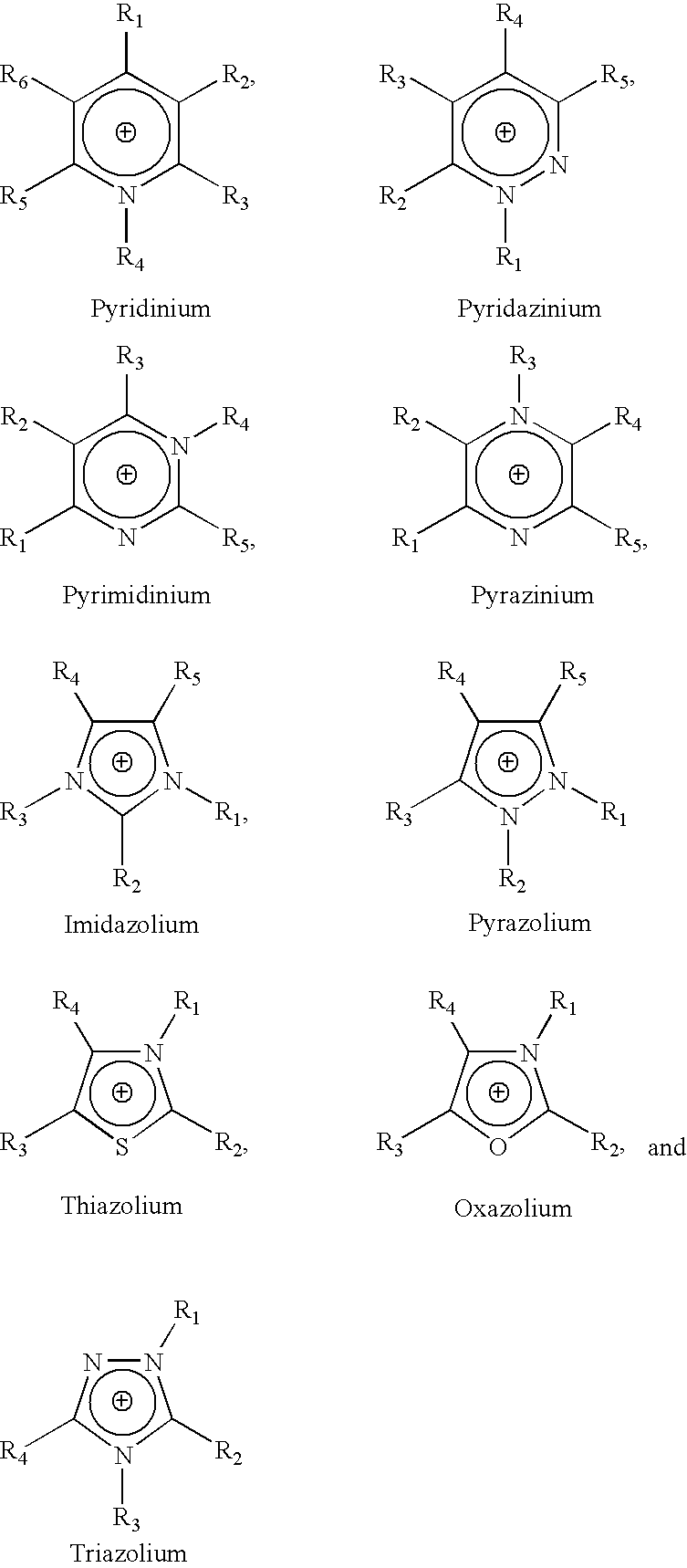
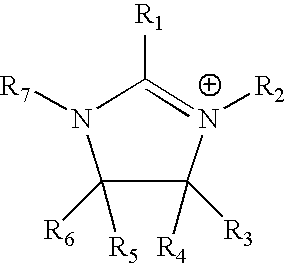
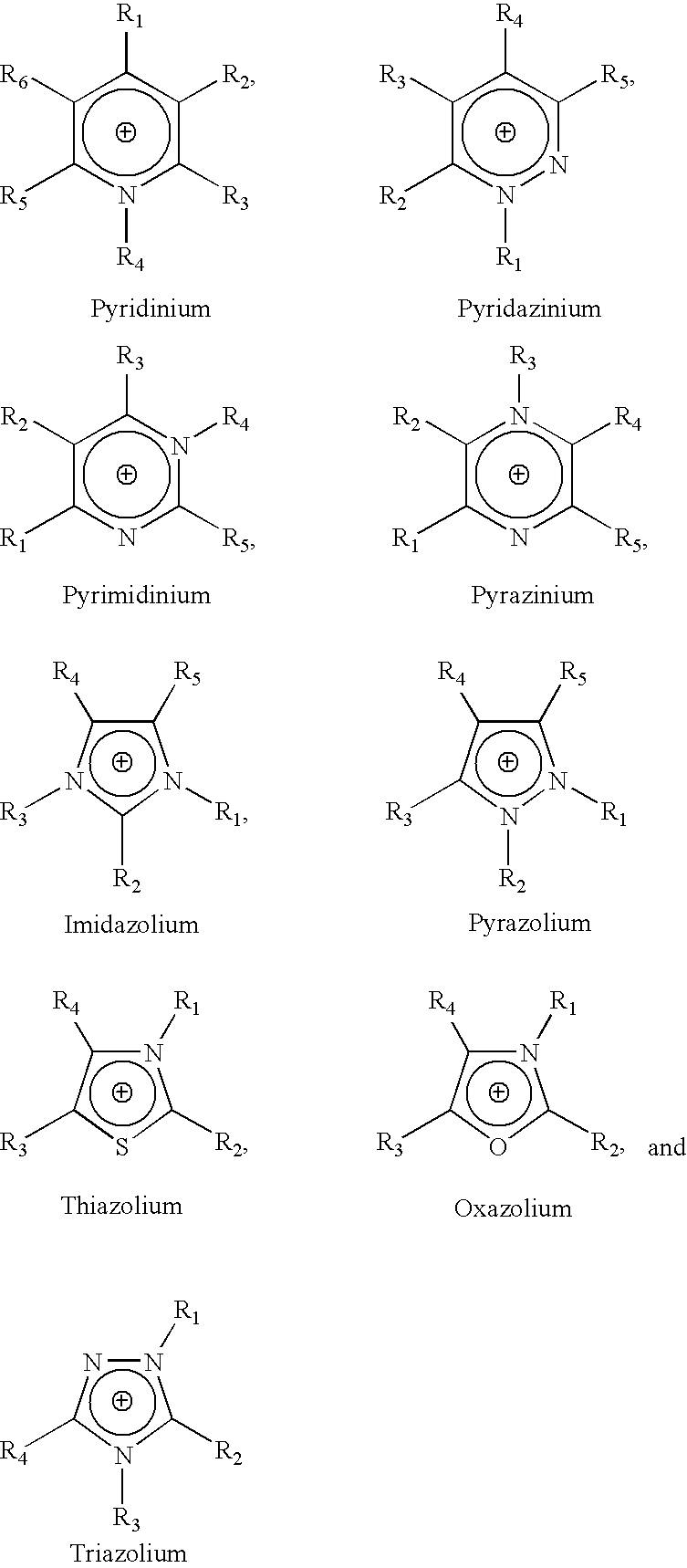
The invention relates to a lithium-sulfur battery. An electrolyte of the lithium-sulfur battery consists of solvent molecules, solute positive ions, and solute negative ions; the molar concentration of the solute in the electrolyte is 0.1-10M; the solute positive ions comprise one or more than two kinds of alkali metal positive ions, alkaline earth metal positive ions, quaternary ammonium ions, pyridinylamino ions, imidazolylamino ions, quaternary phosphonate positive ions, pyrimidinium positive ions, pyrazolium positive ions, pyridazinium positive ions, thiazolium positive ions, oxazolium positive ions, triazolium positive ions and phosphonium and ammonium positive ions, and the molar concentration of the solute positive ions is 0.1-10M. The charging process of the lithium-sulfur battery is opposite to that of a conventional lithium-sulfur battery, so that the problem that lithium polysulfide is dissolved into the electrolyte and diffused to a negative electrode is solved fundamentally; and the electrolyte is in a liquid state, so that the problem of electrode interface mass transfer existing in an all-solid-state electrolyte does not exist.
Owner:DALIAN INST OF CHEM PHYSICS CHINESE ACAD OF SCI
Copolymer and method of manufacturing the same
Disclosed is a method of manufacturing a copolymer of a conjugated diene compound and an olefin other than the conjugated diene compound, where a conjugated diene compound and an olefin other than the conjugated diene compound is copolymerized in the presence of a polymerization catalyst composition containing components: (A) a rare earth element compound or a reactant of the rare earth element compound and a Lewis base; and (B) at least one selected from a group consisting of an ionic compound (B-1) composed of a non-coordinating anion and a cation, an aluminoxane (B-2), and at least one kind of halogen compound (B-3) selected from among a Lewis acid, a complex compound of a metal halide and a Lewis base, and an organic compound containing active halogen (when containing at least one of (B-1) and (B-3), the polymerization catalyst composition further contains component (C): an organometallic compound).
Owner:BRIDGESTONE CORP
Novel composite quaternary phosphonium salt and preparation method thereof and fluorescence application
ActiveCN109810139AImprove performanceReduce consumptionGroup 5/15 element organic compoundsLuminescent compositionsChemical structurePhosphonium salt
The invention belongs to the technical field of functional material synthesis, and particularly relates to a novel composite quaternary phosphonium salt with fluorescence performance. The novel composite quaternary phosphonium salt is a compound composed of tetrachloro-manganese anions and benzyl tri(4-methoxyl)phenyl phosphonium cations, the chemical structure is shown in a formula (I) (please see the specifications for the formula), the abbreviation is [BzT(4-MeO)PP]2[MnCl4], the novel composite quaternary phosphonium salt shows the good green fluorescence effect under ultraviolet irradiation, especially at 360 nm, has the high fluorescence effect, and is an optical material with excellent performance. The invention further provides a preparation method of the novel composite quaternaryphosphonium salt with the fluorescence performance. The method has the advantages of high yield, cost saving, easy operation and facilitating industrial production.
Owner:SOUTH CHINA AGRI UNIV
Method for preparing furfural through ionic liquid catalysis
ActiveCN110256377AEasy to peelImprove solubilityOrganic chemistryOrganic-compounds/hydrides/coordination-complexes catalystsKetoneSolvent
The invention discloses a method for preparing furfural through catalysis of ionic liquid, and relates to the field of furfural preparation. The method comprises the following steps: mixing carbohydrate, a composite solvent and an acid bifunctional ionic liquid catalyst; and carrying out catalytic reaction on the mixture at 120-200 DEG C for 60-300 minutes to synthesize furfural. The carbohydrate is monosaccharide, disaccharide, polysaccharide or biomass rich in pentose monomers; wherein the composite solvent is composed of water and a ketone organic solvent; the acid bifunctional ionic liquid catalyst is a compound composed of metal halide anions having Lewis acidity and imidazolyl cations having acidity. The reaction system is economical and environment-friendly, the operation conditions are mild, the complexity of the technology for preparing furfural from pentose monomer source carbohydrate is effectively simplified, and the furfural preparation cost is greatly reduced under the condition that the preparation efficiency is equivalent to that of the prior art.
Owner:ZHEJIANG UNIV
Powdery, cross-linked absorbent polymers method for the production thereof and their use
The invention relates to crosslinked polymerizates which are capable of absorbing, which are based on partially neutralized, monoethylenically unsaturated monomers that carry acidic groups, which exhibit improved properties, in particular, with regard to their ability to transport liquids when in a swollen state, and which have been subsequently crosslinked on the surface thereof with a combination consisting of an organic crosslinker compound, with the exception of polyols, and of a cation provided in the form of a salt in an aqueous solution.
Owner:EVONIK DEGUSSA GMBH
Resist composition and patterning process
ActiveUS20210063879A1High resolutionWide focus marginSemiconductor/solid-state device manufacturingPhotosensitive material processingPolymer scienceImage resolution
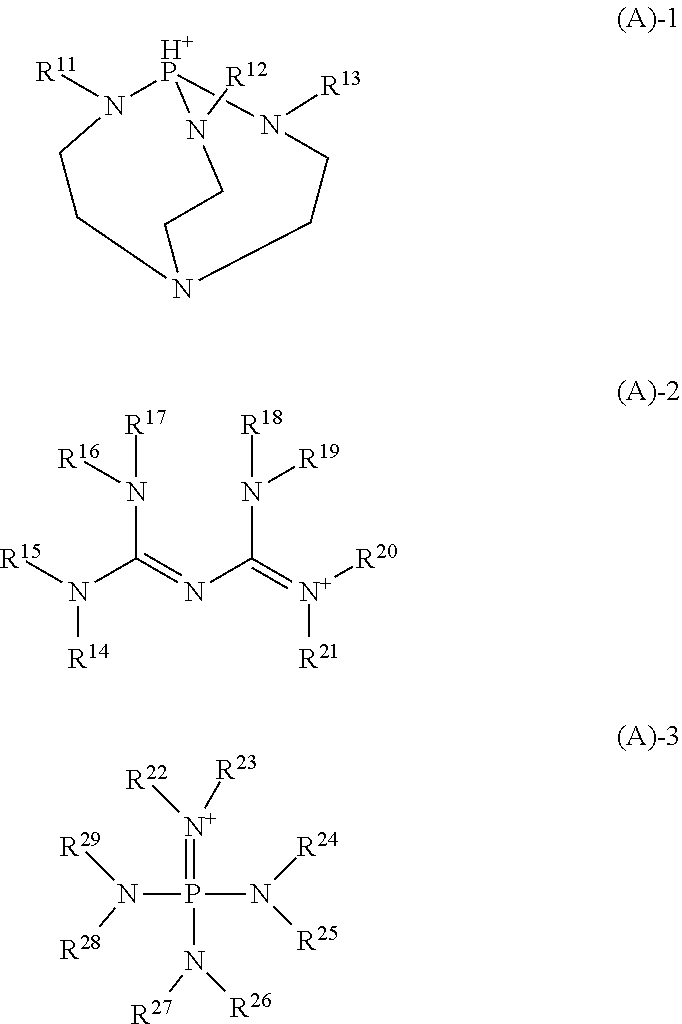
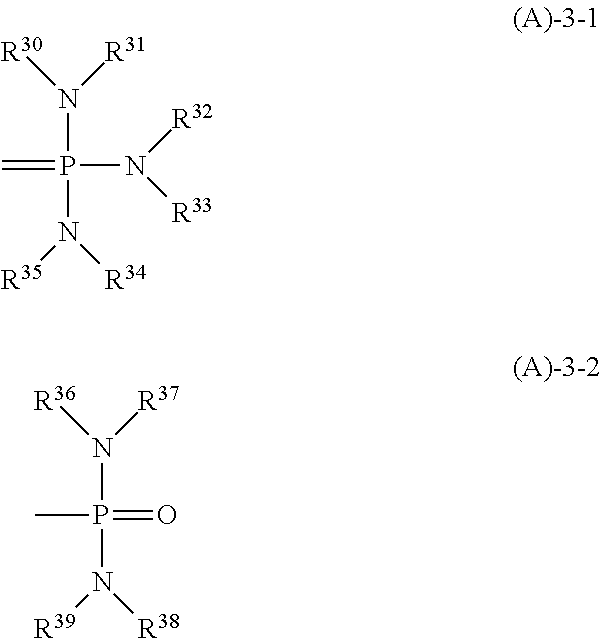
A resist composition comprising a base polymer and a salt is provided. The salt consisting of an anion derived from a carboxylic acid having an iodized or brominated hydrocarbyl group and a cation derived from a 2,5,8,9-tetraaza-1-phosphabicyclo[3.3.3]undecane, biguanide or phosphazene compound. The resist composition exerts a high sensitizing effect and an acid diffusion suppressing effect, causes no film thickness loss after development, and is improved in resolution, LWR and CDU when a pattern is formed therefrom by lithography.
Owner:SHIN ETSU CHEM IND CO LTD
Color correction material, film-forming composition, and optical filter
InactiveUS20110049446A1Improve heat resistanceExcellent lightfastnessOrganic chemistryMethine/polymethine dyesClay mineralsCyanine
A color correction material contains at least one type of cyanine compound composed of a cyanine cation represented by general formula (I) below (wherein the symbols are as defined in the description) and a q-valent anion represented by pAnq− (wherein the symbols are as defined in the description), and at least one type of layered clay mineral; or a color correction material containing a clay mineral composite obtained by intercalating a cyanine cation represented by general formula (I) below, or preferably the cyanine cation and an organic cation, into a layered clay mineral. Also described is a film-forming composition containing the color correction material and an optical filter having a layer composed of the film-forming composition.Ring A represents a benzene ring or a naphthalene ring; X represents CR3R4, NR5, an oxygen atom, a sulfur atom, or a selenium atom.
Owner:ADEKA CORP
Solid-phase synthesis method of thymalfasin
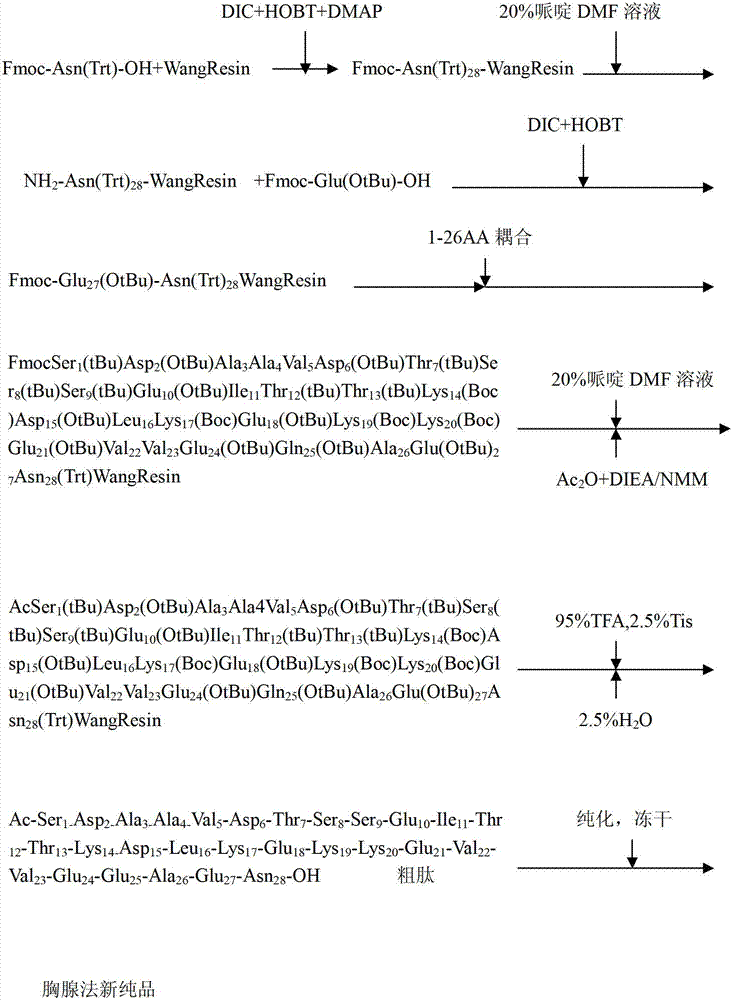
InactiveCN103923210AGood effectDifficult sequence to overcomeThymosin peptidesPeptide preparation methodsCombinatorial chemistrySolvent
The invention relates to a solid-phase synthesis method of thymalfasin. The solid-phase synthesis method comprises the process steps: solid-phase linear synthesis of linear thymalfasin is carried out, a DMF solution of a salt composed of bulky anions and monovalence cations is used as a reaction solvent in a difficult sequence, hydrogen bond formation is interfered, and a beta structure of a peptide chain is destroyed. The process can overcome the polypeptide synthesized difficult sequence, improves the coupling rate of the difficult sequence so as to improve the yield of thymalfasin, reduces the production cost, and is beneficial for industrialized production.
Owner:SHANDONG NEWTIME PHARMA
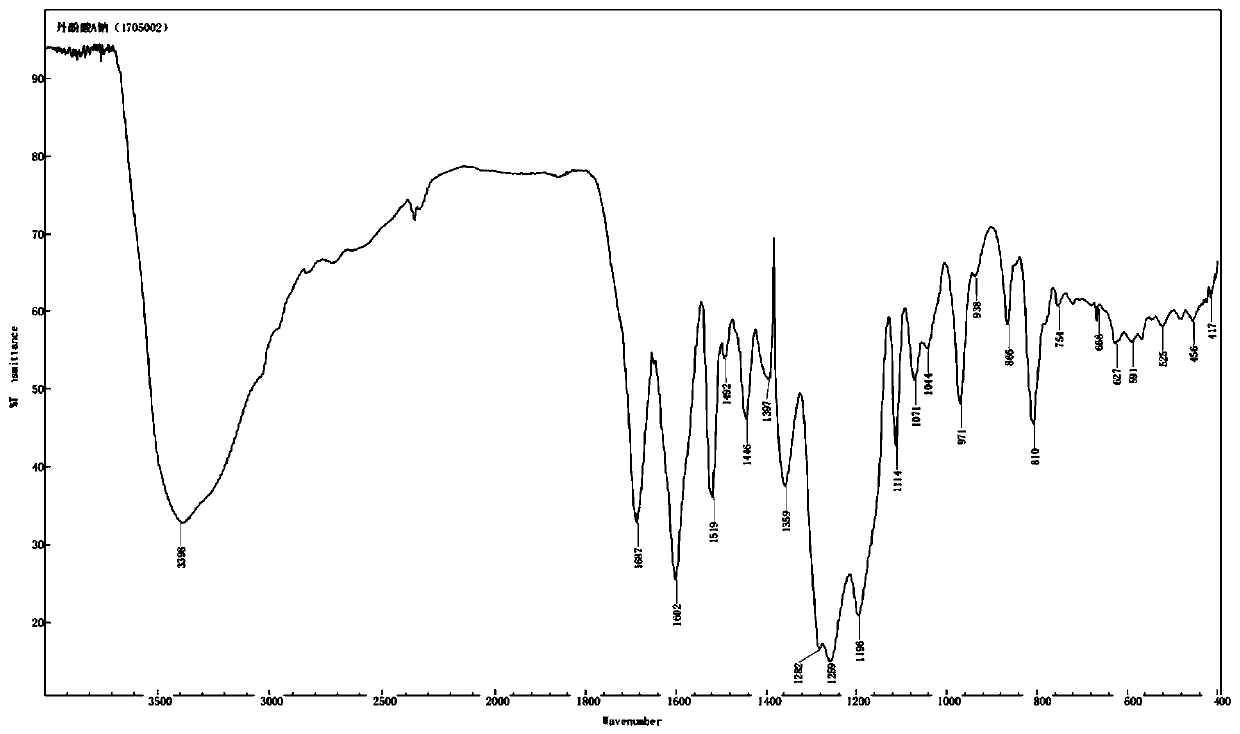
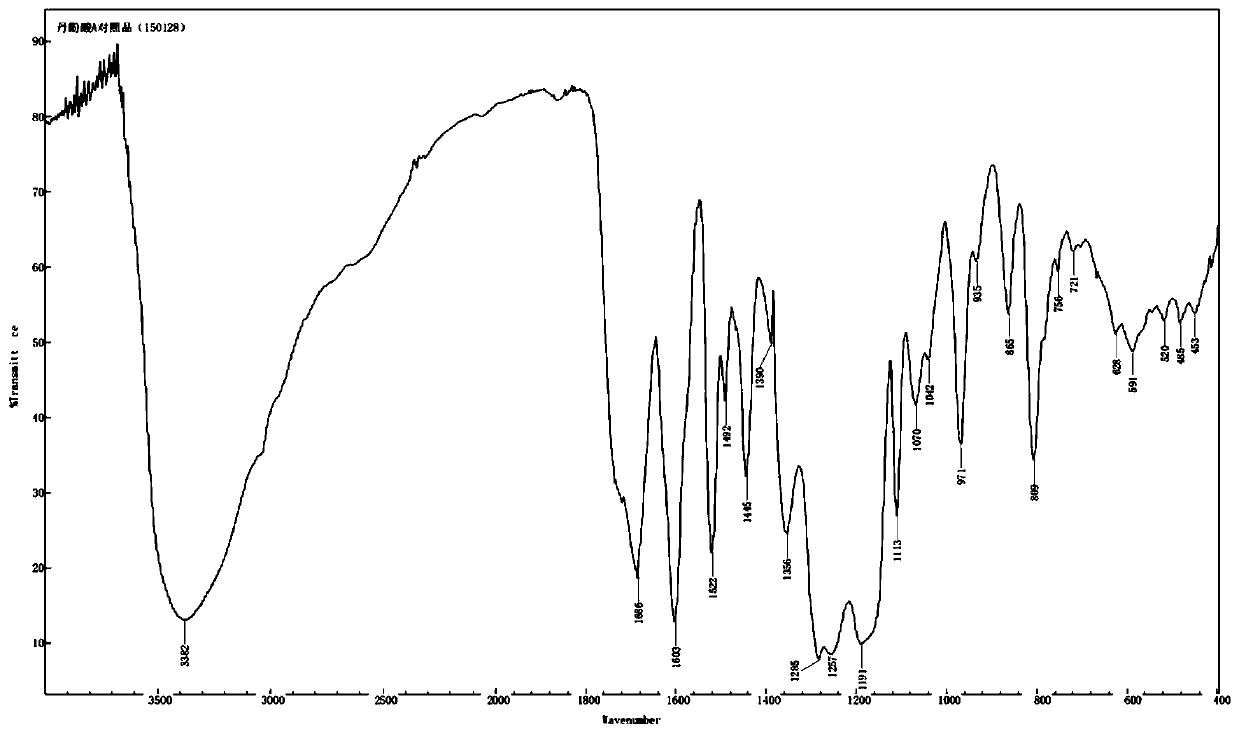
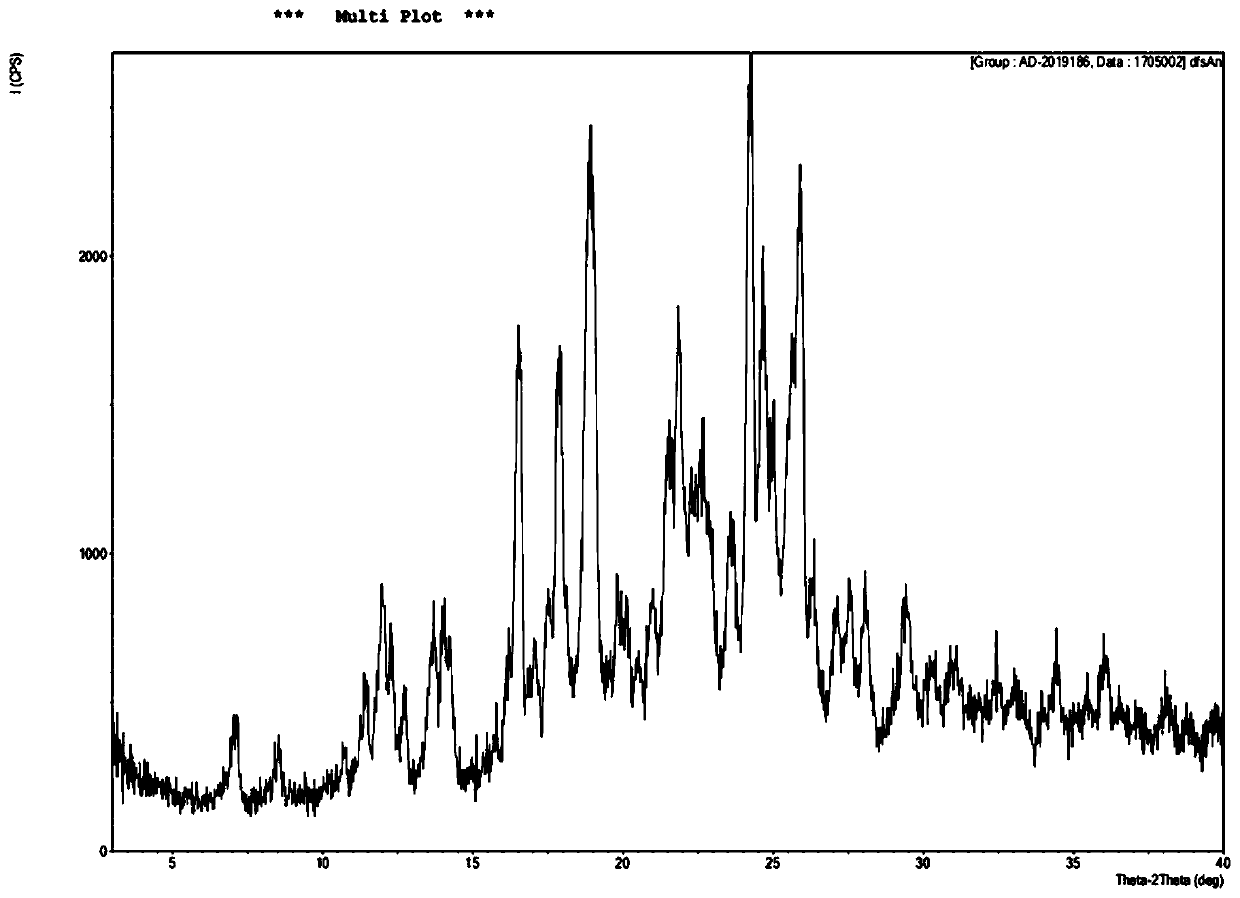
Salvianolic acid A salt complex and preparation method and application thereof
InactiveCN110403928AAnti-myocardial ischemiaInhibit aggregationOrganic active ingredientsBlood disorderCurative effectSalvianolic acid
The invention provides a salvianolic acid A salt complex. The salvianolic acid A salt complex is prepared from diad salvianolic acid A and a positive ion, the salvianolic acid A salt complex has the chemical construction (being defined as in the specification); and the salvianolic acid A salt complex has the pharmacological activity of anti-myocardial ischemia treating, anti-myocardial ischemia reperfusion injury treating, and platelet aggregation inhibition, and can be applied to preparation of medicines with the related curative effect. The preparation method of the salvianolic acid A salt complex is simple, easy to produce, and more stable relative to the salvianolic acid A.
Owner:CHIATAI QINGCHUNBAO PHARMA
Capillary electrophoresis test system applied to soil solution cation analysis
InactiveCN101603942AIncrease viscosityEasy to separateMaterial analysis by electric/magnetic meansHigh concentrationCapillary electrophoresis
A capillary electrophoresis separation test system applied to soil solution cation composition and concentration thereof. The invention is characterized in that the system contains imidazole of 5-10mM, the concentration of HIBA in a buffer solution is 8-9mM, the concentration of 18-crest-6 is 2-5mM, and the pH value of the test system is 3.5-4.0. The invention aims at providing the capillary electrophoresis separation test system for cation composition in soil in-situ solution. The test system focuses on optimizing the composition of buffer solution to be tested and formulation thereof, thus being widely applicable for fast test of soil solution, in particular for the test requirement featuring complex proportion of composition, unsuitable proportional absorption and higher concentration for partial composition.
Owner:INST OF SOIL SCI CHINESE ACAD OF SCI
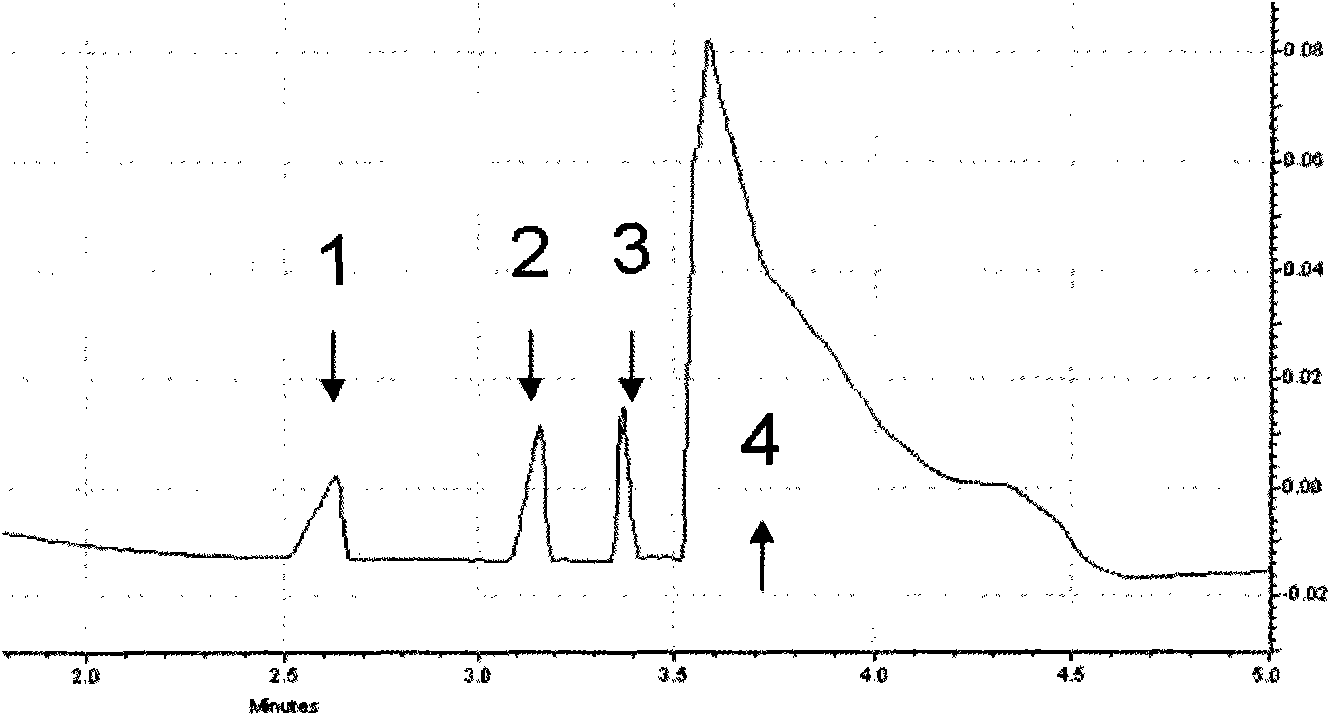
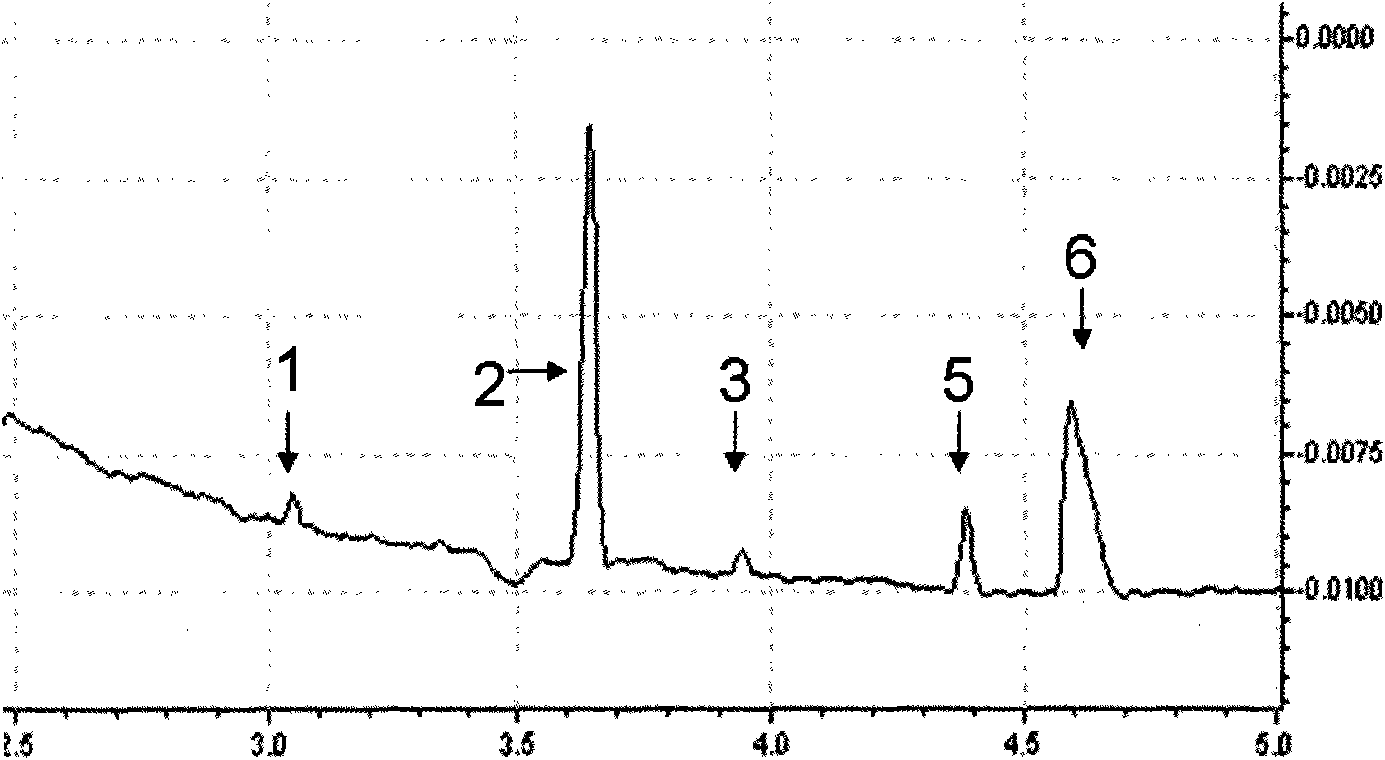
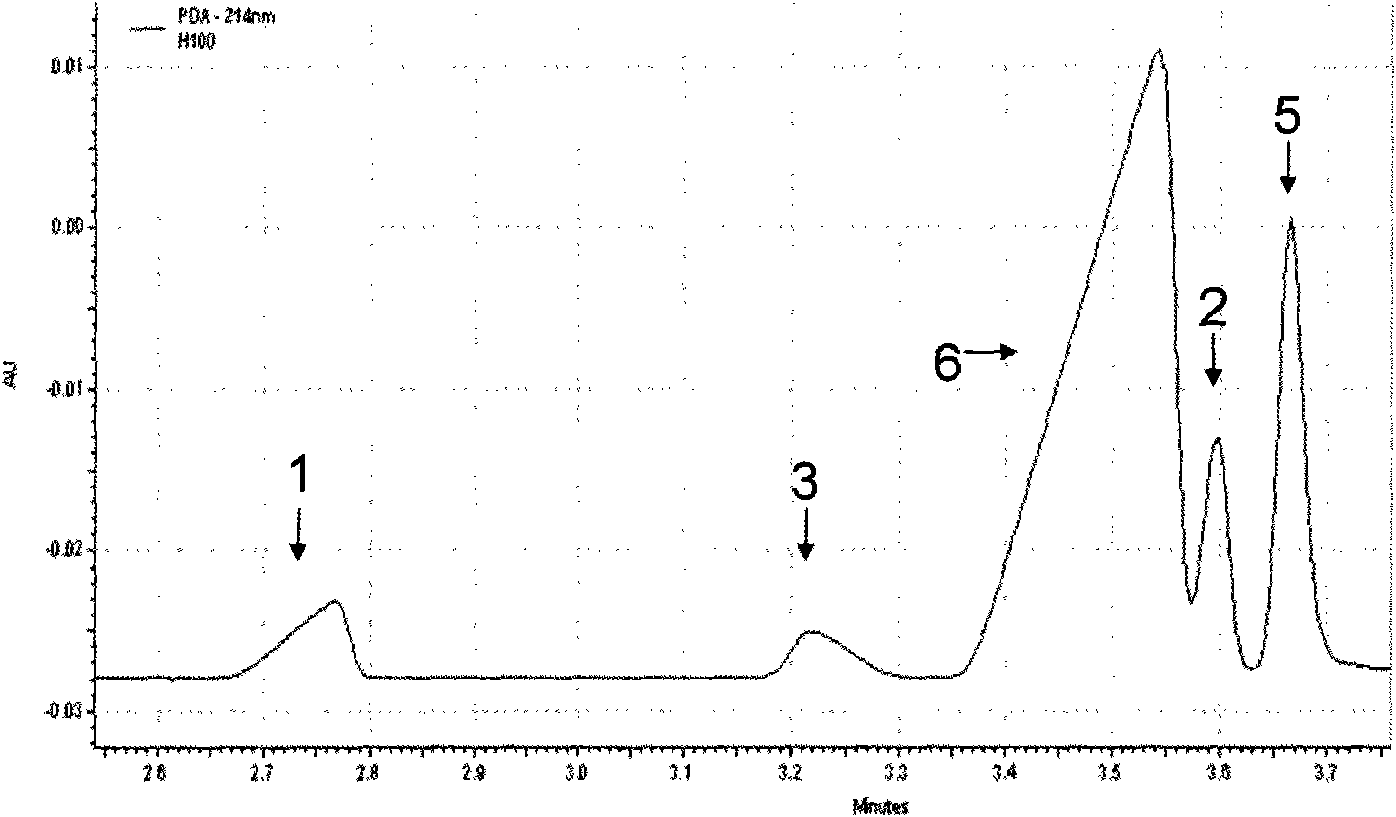
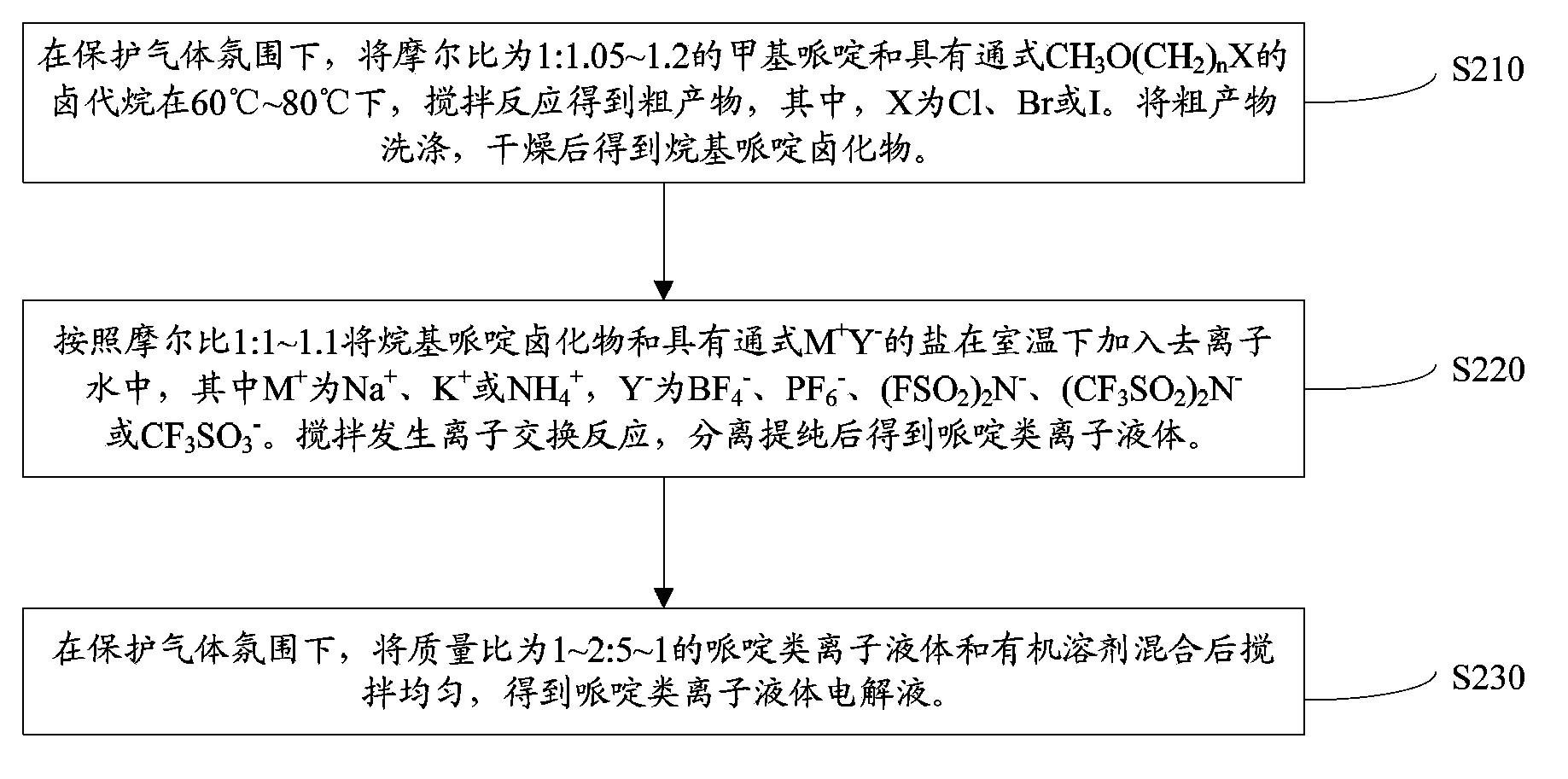
Piperidines ionic liquid, and preparation method and application thereof
InactiveCN103788017AHybrid capacitor electrolytesSulfonic acids salts preparationOrganic solventRoom temperature
The invention discloses a piperidines ionic liquid which has the following general structural formula defined in the description, wherein n is 2, 3 or 4, and Y<-> represents BF4<->, PF6<->, (CF3SO2)2N<-> or CF3SO3<->. The piperidines ionic liquid is composed of an anion and a cation at the room temperature, has good conductivity, moreover, is not easily volatilized, is not burnt, and has good thermal stability. When a piperidines ionic liquid electrolytic solution is used in a super capacitor, because an organic solvent of the piperidines ionic liquid electrolytic solution is evenly mixed with a large amount of the piperidines ionic liquid, the piperidines ionic liquid electrolytic solution is safer to use compared with a traditional organic electrolytic solution. The invention also discloses a preparation method and an application of the piperidines ionic liquid.
Owner:OCEANS KING LIGHTING SCI&TECH CO LTD +2