Patents
Literature
111results about How to "High refining yield" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
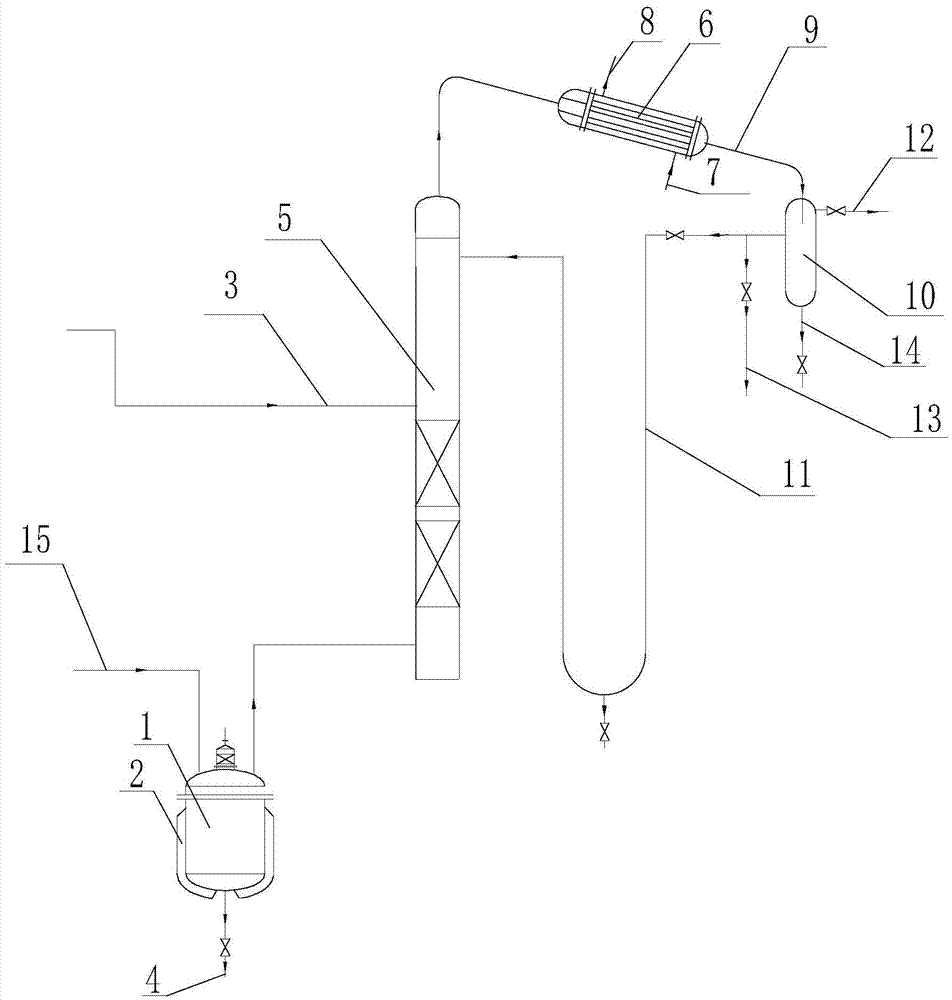
Method for refining long-carbon chain dicarboxylic acid
ActiveCN104496793AMeet the technical requirements of useLess investmentCarboxylic compound separation/purificationEngineeringCarboxylic acid
The invention relates to a method for refining long-carbon chain dicarboxylic acid. A long-chain dicarboxylic acid crude product prepared by a fermentation method is used as a raw material, acetic acid is used as a solvent, and the refining and purification are performed by recrystallization treatment. The method comprises the process steps of bleaching filtration, cooling crystallization, centrifugal separation, centrifugal material washing, secondary centrifugation and drying, and is characterized in that a. the long-chain dicarboxylic acid crude product to be refined does not need to be dried; b. the acetic acid solvent is recycled by an azeotropic distillation method, and isopropyl acetate is used as an azeotropic dehydrating agent; and c. the isopropyl acetate is used as a centrifugal material washing solvent for replacing the acetic acid. The purification method of the long-carbon chain dicarboxylic acid provided by the invention has the advantages of distillation equipment investment conservation, low operating power consumption, good product color, high purity, high refining yield and low production cost, the long-carbon chain dicarboxylic acid to be refined does not need to be dried and dehydrated, and the acetic acid solvent is completely recyclable. The quality of the refined product can meet the technical requirements of use of condensed monomer raw materials used by synthesis of high polymer materials.
Owner:山东广通新材料有限公司
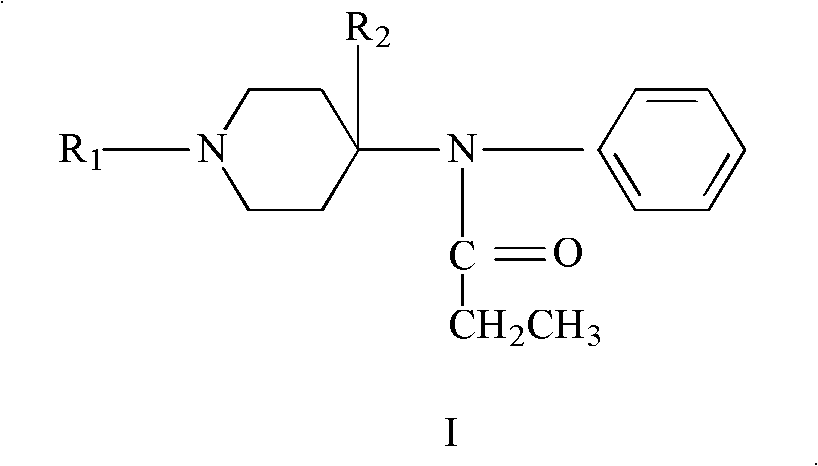
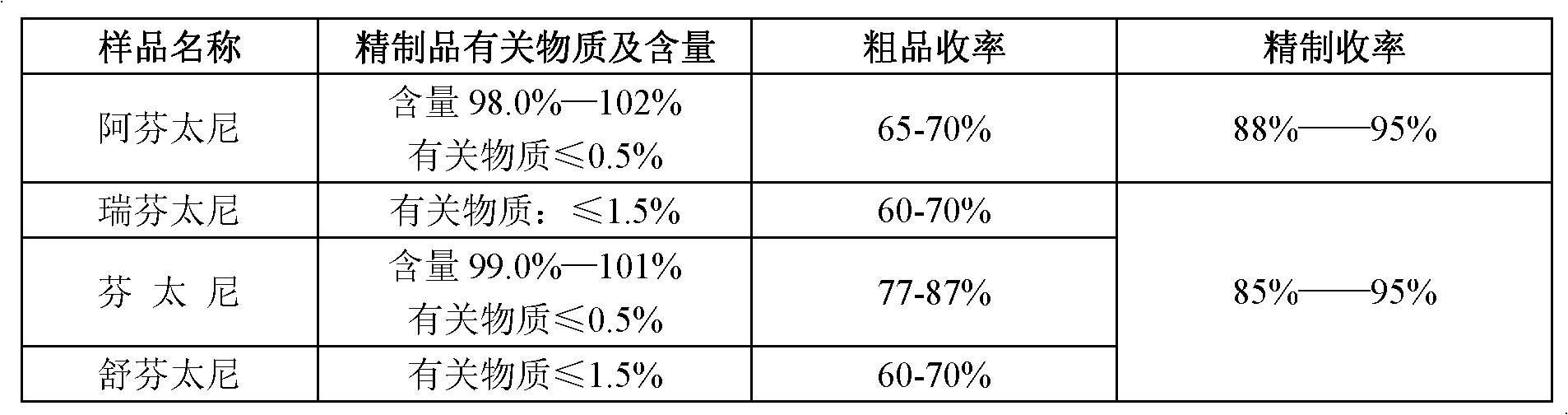
Refining method of 4-phenylaminopiperidine analgesic
The invention discloses a refining method of 4-phenylaminopiperidine analgesic, comprising the following steps of firstly preparing an acid salt from the 4-phenylaminopiperidine analgesic, purifying the acid salt after recrystallization, alkalizing the salt again to obtain free basic groups, adding another acid to the mixture to obtain another acid salt, re-crystallizing and refining the obtainedanother acid salt by a second solvent to obtain the acid salt. According to the invention, the process is simple, the used agents are simply and easily obtained, and only twice salifying operation and recrystallization operation are needed; and by utilizing the refining method, the content of impurities is remarkably reduced, the purity reaches the weight standard associated with the pharmacopoeia, the purity and the yield of fentanyl compound acid salts prepared from fentanyl compound free basic groups are remarkably improved, the production cost is reduced, and the production efficiency is remarkably improved.
Owner:YICHANG HUMANWELL PHARMA
Manufacturing method of proPiconazole
ActiveCN102225935AEmission reductionPrecise meteringOrganic chemistryChemical recyclingPropiconazoleBromine
The invention discloses a manufacturing method of proPiconazole. The manufacturing method comprises that 2,4-dichloroacetophenone and 1,2-pentanediol undergo a cyclization reaction, and then bromine and the reaction products of the cyclization reaction undergo a bromination reaction to form 2-bromomethyl-(2,4-dichlorophenyl)-4-propyl-1,3-dioxolane. The manufacturing method is characterized in that solid heteropolyacid catalysts are utilized in the cyclization reaction, and are filtered and recycled after the cyclization reaction is finished; then the bromination reaction is caused and the 2-bromomethyl-(2,4-dichlorophenyl)-4-propyl-1,3-dioxolane, 1,2,4-triazole and potassium carbonate undergo a condensation reaction to form the proPiconazole after the bromination reaction is finished. Through the manufacturing method, new impurities which can be introduced because catalysts take part in a bromination reaction are reduced. In the invention, because a potassium triazole preparation process, a condensation reaction process and a solvent removing process are carried out simultaneously during a condensation reaction, problems of long reaction time and adhesion of 1,2,4-triazole on a kettle are solved, and a reaction time and a production cost are reduced.
Owner:山东潍坊双星农药有限公司
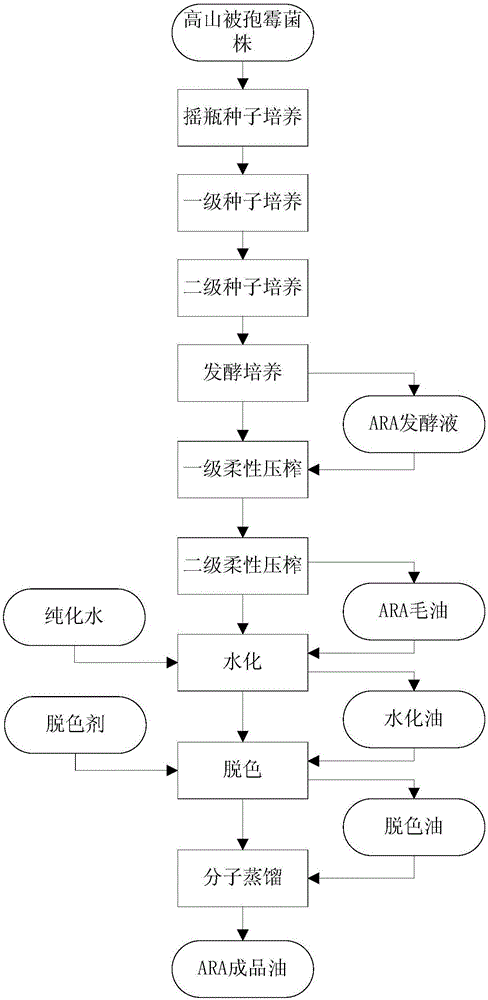
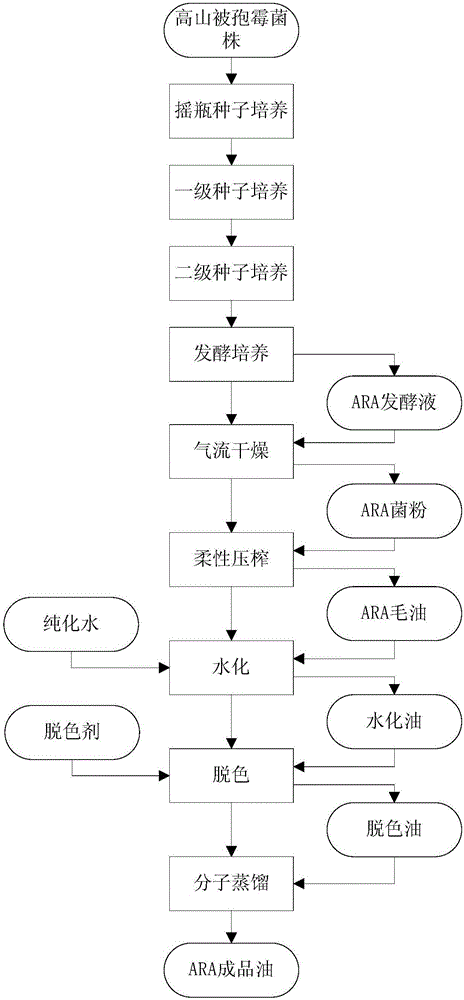
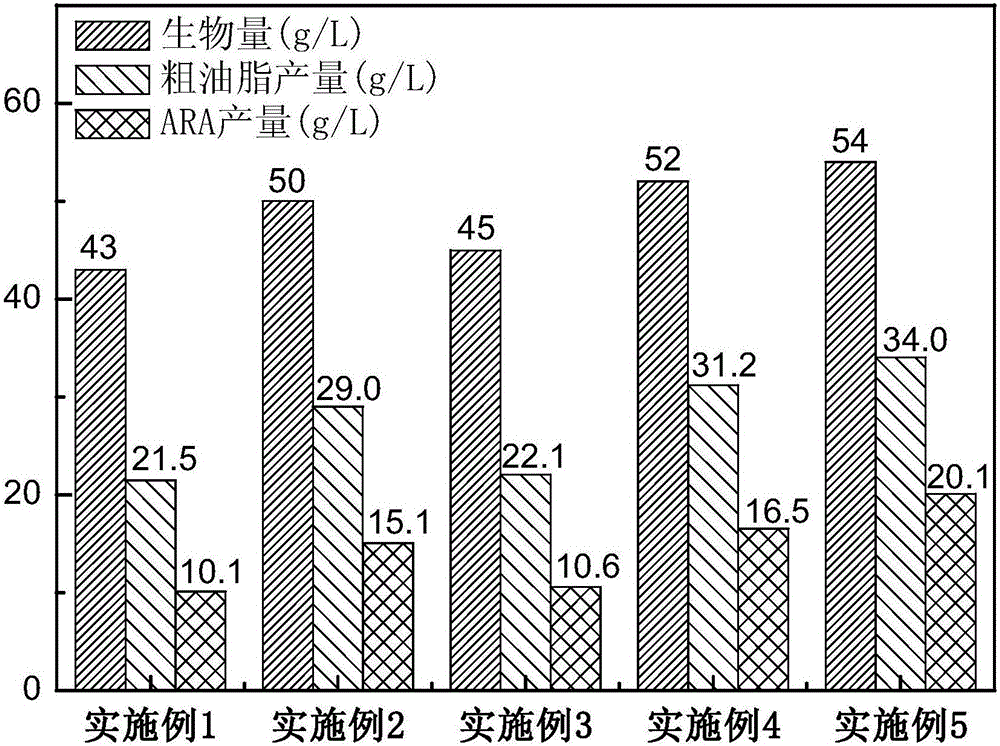
Method for producing ARA (arachidonic acid) through microbial fermentation
ActiveCN106755151AConducive to large-scale industrial productionReduce the cost of fermentation productionFatty-oils/fats refiningMicroorganism based processesMicroorganismBiotechnology
The invention belongs to the field of fermentation engineering, relates to a method for producing ARA (arachidonic acid) through microbial fermentation, and particularly relates to a method for producing compound fat containing the ARA through industrial fermentation of a mortierella alpine strain. More particularly, the invention relates to a method for culturing microorganisms for producing the ARA, wherein the method for culturing the microorganisms for producing the ARA comprises the steps of controlling the fermentation temperature to be 20 to 25 DEG C after culturing in a fermentation tank for 100 to 140h; and / or, stopping ventilation or reducing the ventilatory capacity by not less than 50 percent at the 8th to 10th day of the culturing in the fermentation tank. The method for preparing the ARA has the advantages that the yield is high, the purity is high, and the large-scale industrial production of the ARA is facilitated.
Owner:INNER MONGOLIA KINGDOMWAY PHARMA LTD +1
Separation method for refining coarse piperyene
ActiveCN1796340AHigh refining yieldHigh purityDistillation purification/separationCyclopenteneHydrazine compound
This is a process for the refinery and separation of crude m-pentadiene, which is obtained through the separation of the by-product C5 fraction of ethane manufacturing via petroleum pyrolysis, including the steps of: 1) directing the crude m-pentadiene into an extractive rectification column for extractive rectification, and collecting an overhead product of a mixed stream substantially enriched in cyclopentene and cyclopententane, and a bottom product of a mixed stream substantially enriched in m-pentadiene and the extractant, which is dimethyl formamide, N-methyl pyrrolidone, ethyl hydrazine or an aqueous solution of ethyl hydrazine containing 5-15 wt.% water; 2) directing the said bottom stream into a refinery column for refinery, and colleting an overhead product of the refined m-pentadiene, and a bottom product of the extractant, which is recovered and recycled; 3) subjecting the process 1 wherein said overhead stream to a hydrogenation reaction, and obtaining the hydrogenation product containing cyclopententane; and 4) removing the light components whose boiling point is lower than that of cyclopententane from the hydrogenation product, and obtaining the refinery product of cyclopententane. This invention ensures a high recovery and refinery rate to obtain refined m-pentadiene product with purity higher than 95%, and also obtains a byproduct of cyclopententane.
Owner:SINOPEC SHANGHAI PETROCHEMICAL CO LTD
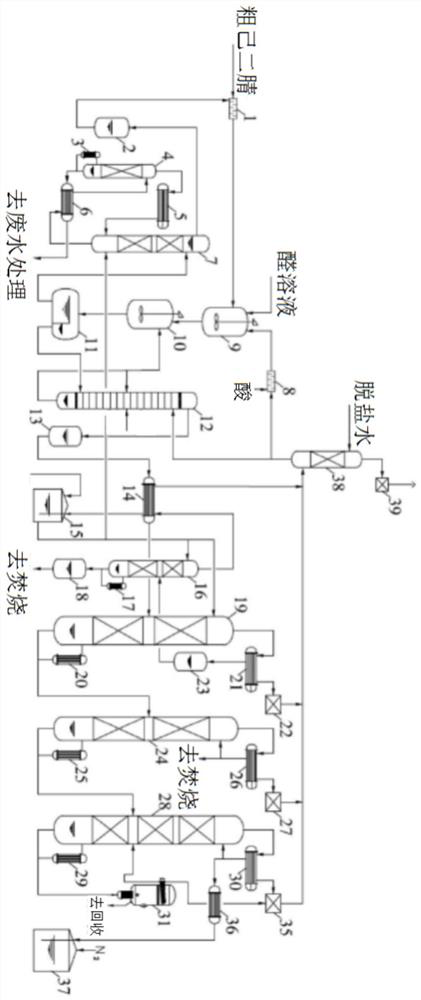
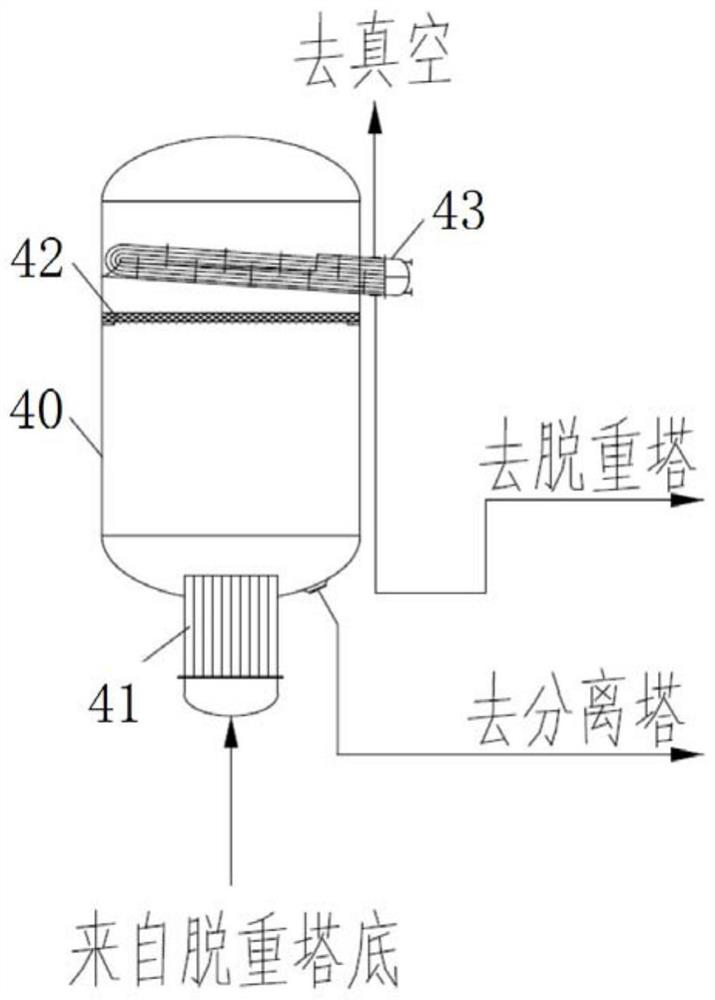
Refining method of long chain dicarboxylic acid
ActiveCN110002992ASimple preparation stepsScalable productionCarboxylic compound separation/purificationAcetic acidThermal insulation
The invention provides a refining method of long chain dicarboxylic acid. The long chain dicarboxylic acid is refined with a long chain dicarboxylic acid crude product prepared with a biological method as a raw material and low-concentration acetic acid as a solvent. Specifically, the long chain dicarboxylic acid crude product and the low-concentration acetic acid are mixed firstly, stirred, heated and decolorized in a normal pressure environment, filtration is performed with thermal insulation, filtrate is cooled slowly, crystallization and separation are performed, a dicarboxylic acid intermediate is obtained and suspended with water with melting point temperature higher than that of monoacid with chain length corresponding to that of the long chain dicarboxylic acid, thermal insulation,cooling, separation and drying are performed in a negative pressure environment, and a long chain dicarboxylic acid refined product is obtained. The defects that existing solvent refining methods arelow in yield and the content of solvent residues in products is high are overcome. The refined long chain dicarboxylic acid completely meets the raw material quality requirement of polymer material synthesis.
Owner:张艾琳
Synthesis method of lornoxicam
Owner:BEIJING JINCHENG TAIER PHARMA CO LTD
Method for preparing high-purity losartan
The invention relates to a method for preparing high-purity losartan. The method comprises steps as follows: (1), a losartan crude product is added to an organic solvent or a mixed solvent of an organic solvent and water, and the mixture is heated to reach 20-80 DEG C and stirred; (2), the system is cooled directly or cooled after water is added or cooled to 0-5 DEG C after a part of solvent is evaporated, a material is precipitated, filtered and dried, and losartan is obtained, wherein the organic solvent used in the step (1) is any one of tetrahydrofuran, butanone, acetone and methyl alcohol or is a mixed solvent of any one of the four solvents and water. The losartan obtained with the method has high purity, any individual impurity can be reduced to 0.2% or even under 0.1%, the purity of the losartan can reach 99.5%, the cost of the method is lower, the refining yield is high, and the method is very simple in operation, environment-friendly and suitable for industrial production.
Owner:ZHEJIANG HUAHAI PHARMA CO LTD
Production method of bromhexine hydrochloride
ActiveCN103333074AReduce wasteImprove product quality stabilityOrganic compound preparationAmino compound preparationAcetic acidAlcohol
The invention relates to a production method of bromhexine hydrochloride. The production method is characterized by comprising the following steps of: 1) carrying out a condensation reaction between 3,5-dibromo-2-aminobenzalcohol and N-methyl cyclohexane in the presence of glacial acetic acid as a catalyst, thereby generating N-methyl-N-cyclohexyl-2-amino-3,5-dibromobenzylamine; 2) salifying the generated N-methyl-N-cyclohexyl-2-amino-3,5-dibromobenzylamine with hydrochloric acid to obtain bromhexine hydrochloride; and 3) decoloring and refining the obtained bromhexine hydrochloride in an alcoholic solution to obtain a pure product, wherein an azeotropic water-carrying agent is added to the condensation reaction in the step 1), and water in the reaction mixed solution in the reaction process is removed immediately through distillation.
Owner:ZHEJIANG WANBANG PHARMA
Manufacturing method of proPiconazole
ActiveCN102225935BEmission reductionPrecise meteringOrganic chemistryChemical recyclingPropiconazoleBromine
The invention discloses a manufacturing method of proPiconazole. The manufacturing method comprises that 2,4-dichloroacetophenone and 1,2-pentanediol undergo a cyclization reaction, and then bromine and the reaction products of the cyclization reaction undergo a bromination reaction to form 2-bromomethyl-(2,4-dichlorophenyl)-4-propyl-1,3-dioxolane. The manufacturing method is characterized in that solid heteropolyacid catalysts are utilized in the cyclization reaction, and are filtered and recycled after the cyclization reaction is finished; then the bromination reaction is caused and the 2-bromomethyl-(2,4-dichlorophenyl)-4-propyl-1,3-dioxolane, 1,2,4-triazole and potassium carbonate undergo a condensation reaction to form the proPiconazole after the bromination reaction is finished. Through the manufacturing method, new impurities which can be introduced because catalysts take part in a bromination reaction are reduced. In the invention, because a potassium triazole preparation process, a condensation reaction process and a solvent removing process are carried out simultaneously during a condensation reaction, problems of long reaction time and adhesion of 1,2,4-triazole on a kettle are solved, and a reaction time and a production cost are reduced.
Owner:山东潍坊双星农药有限公司
Method for refining crude lenalidomide product
InactiveCN102993169AAid in crystallizationReduce volatilityOrganic chemistryActivated carbonMaterials science
The invention discloses a method for refining a crude lenalidomide product. The method comprises the following steps of: (1) adding the crude lenalidomide product to a mixed solution of isopropanol and water; (2) heating, after the crude lenalidomide product is completely dissolved, adding activated carbon and nanometer ceramic powder and carrying out heat preservation for the first time; (3) filtering when the solution is hot, and cooling filtrate till crystals precipitate; and (4) carrying out heat preservation for the second time, carrying out suction filtering after the crystal precipitation is finished, and finally drying. Due to adoption of the technical scheme, the method for refining the crude lenalidomide product has the beneficial effects of low isopropanol toxicity, high safety, simpleness in process, no need of special equipment and environment requirements, suitability for industrial production, high product content and high refining yield; and the purify of the lenalidomide is remarkably improved.
Owner:NANTONG YUANYA PRECISION MACHINERY
Waste water zero discharge acrylic acid refining device and process
InactiveCN107857701AAchieving zero emissionsAchieve emissionsCarboxylic compound separation/purificationAcetic acidAbsorption column
The invention relates to an acrylic acid refining device with zero discharge of waste water. The device includes an absorption tower, a dehydration tower, an acetic acid removal tower, a weight removal tower, a first circulation system and a second circulation system; the bottom outlet of the absorption tower is connected to the inlet of the dehydration tower through a first storage tank; The top outlet of the dehydration tower is connected with the first circulation system; the bottom outlet of the dehydration tower is connected with the inlet of the deacetic acid tower through the second storage tank; the top outlet of the dehydration tower is connected with the The second circulation system is connected; the bottom outlet of the deacetic acid tower is connected with the weight removal tower through the third storage tank. The invention also relates to a process for refining acrylic acid with zero discharge of waste water. Beneficial effects: the present invention can realize zero discharge of waste water in the true sense.
Owner:张华堂
Refining method of crystal type A azilsartan
The invention relates to the field of medicine, and discloses a refining method of crystal type A azilsartan. The method comprises the following steps: S1, mixing ethyl acetate and absolute ethyl alcohol, heating, and then adding an azilsartan crude product, stirring to be completely dissolved, carrying out filter-pressing; S2, stirring the filtrate in the S1 at 20-25 DEG C to crystallize, filtering, granulating the filter residue through a 24-mesh sieve; drying the raw materials by use of a boiling drying and gradient warming way; drying for 50-70min at an ambient temperature condition, and then drying for 50-70min after warming 40+ / -2 DEG C, wherein the granularity control D (90) is 300-400 microns; drying for 2-3h after warming to 50+ / -2 DEG C, and the granularity control D (90) is 100-200 microns, cooling to obtain an azilsartan finished product. By use of the refining method disclosed by the invention, the dosage of a required organic solvent in refining is greatly reduced; and meanwhile, the stable crystal type A azilsartan is obtained since the raw materials are dried by use of the boiling drying and gradient warming way, the refining yield of 90% or above is acquired, the purity is 99.7% or above, the solvent residue (ethyl alcohol) is less than 0.3%, and the drying time is shortened to 3-6h.
Owner:湖南千金湘江药业股份有限公司
Method for refining long-chain dibasic acid under protection of inert gas
ActiveCN110041193ALow residual acetic acidHigh refining yieldCarboxylic compound separation/purificationThermal insulationFiltration
The invention provides a method for refining long-chain dibasic acid under protection of inert gas. The long-chain dibasic acid is prepared from a long-chain dibasic acid crude product prepared with abiological method as a raw material with low-concentration acetic acid as a solvent. The method specifically comprises the following steps: mixing the long-chain dibasic acid crude product and low-concentration acetic acid, performing stirring, heating and decoloration, performing filtration without thermal insulation, cooling filtrate slowly, performing crystallization and separation to obtain adibasic acid intermediate, re-suspending the intermediate with water at the temperature higher than the melting point temperature of corresponding monobasic acid under protection of the inert gas, and performing thermal insulation, cooling, separation and drying to obtain a long-chain dibasic acid refined product. The method overcomes the defects that existing solvent refining methods are low inyield and the content of solvent residues in products is high. The refined long-chain dibasic acid can completely meet the raw material quality requirement of polymer material synthesis.
Owner:张艾琳
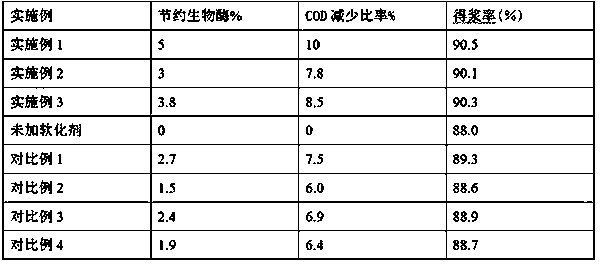
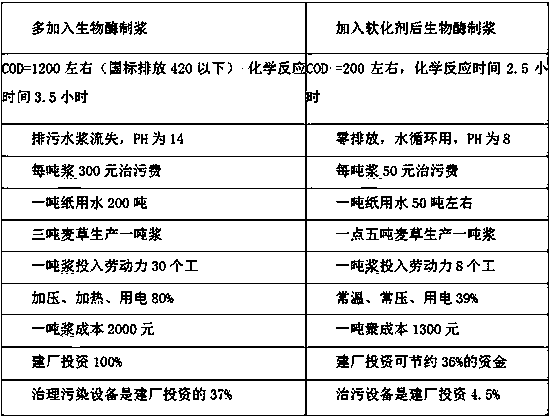
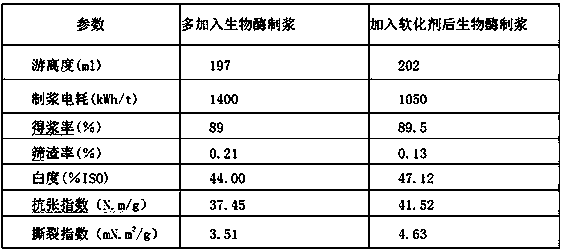
Special softener for bio-enzyme pulping and preparation method thereof
ActiveCN108411674AReduce dosageGood spreadabilityCellulose treatment using microorganisms/enzymesFiberChemical oxygen demand
The invention discloses a special softener for bio-enzyme pulping, which is prepared from the following components: a penetrant BS; sodium diethylhexyl sulfosuccinate; isomeric alcohol ethoxylate; peregal O; fatty alcohol-polyoxyethylene ether; alkyl sodium sulfonate; coconut diethanol amide; sodium hexametaphosphate; sulfamic acid; sodium citrate; and water. The softener ensures that wood chip and straw fibers are easy to mill, reduces the energy consumption of pulping, prolongs the service life of a mill, reduces the dosage of bio-enzyme, reduces the discharge of COD (chemical oxygen demand)in wastewater, and not only can bring economic benefit, but also has environmental protection benefit. The invention further discloses a preparation method for the softener. The manufacturing processis simple, paper products with higher physical strength can be produced, moreover, yield pulp is high, the softener has wider applicability, and can replace more chemical pulps for papermaking, and more resources can be saved; furthermore, the loss of wood chip and straw fibers is reduced, the pulping yield is higher, the COD content in wastewater is reduced, and the invention is of great significance in saving energy, reducing discharge and saving resources.
Owner:上海锴晨实业有限公司
Novel method for fine purification of theobromine
InactiveCN101195619AHigh refining yieldImprove product qualityOrganic chemistryTheobromineReducing agent
The invention relates to a refining method of santheose, which is characterized in that dissolves liquid alkali into crude santheose solution, decolourizes, fitlers, and adds reduction agent into filter liquor, acidifies at 60-80DEG C until pH=1-5, filters and dries to obtain santheose final product. The inventive method has refining yield of 90%, standard product color, high product quality, low cost and industrialization suitability.
Owner:PERRIGO TRADING (SHANGHAI) CO LTD
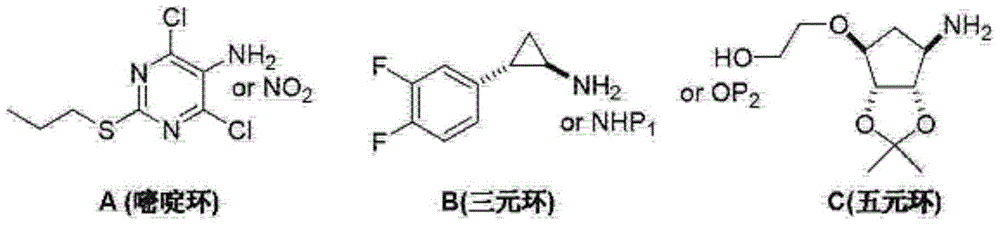
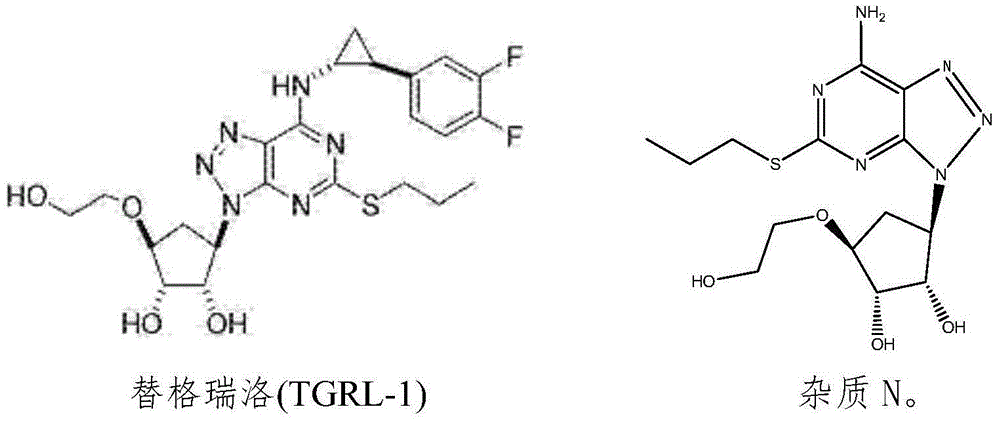
Ticagrelor crystal and medicinal composition comprising same
ActiveCN105153167AHigh refining yieldThe process steps are simpleOrganic active ingredientsOrganic chemistryTicagrelorImpurity
The invention provides a ticagrelor crystal and a medicinal composition comprising the same. The purity of the ticagrelor crystal is greater than 99 percent, and an impurity N is less than 0.1 percent. The crystal is prepared by adopting a one-pot method and is obtained by adopting a further crystal refining method. By adopting the methods, the ticagrelor with the purity being obviously improved can be obtained, and the raw material and medicinal composition with excellent quality are further acquired.
Owner:HUIZHOU XINLITAI PHARMA +1
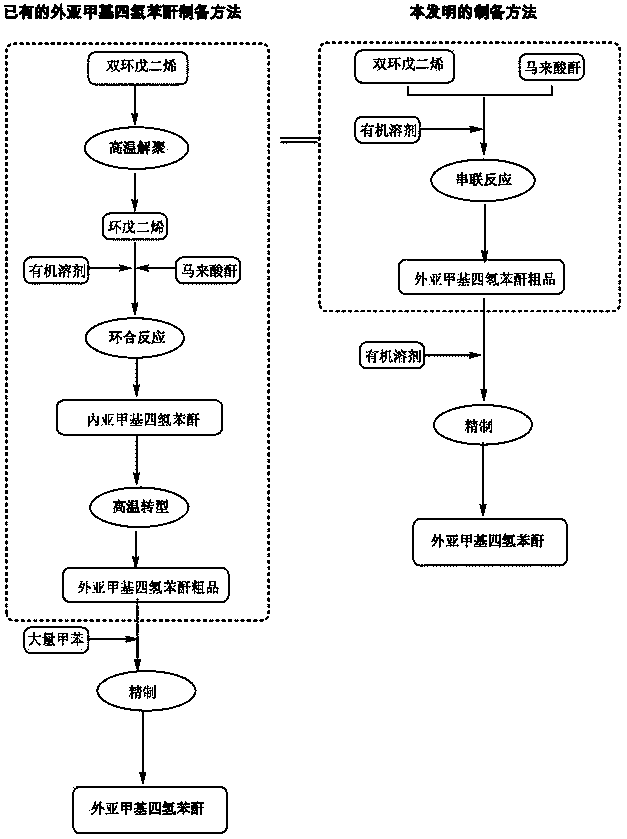
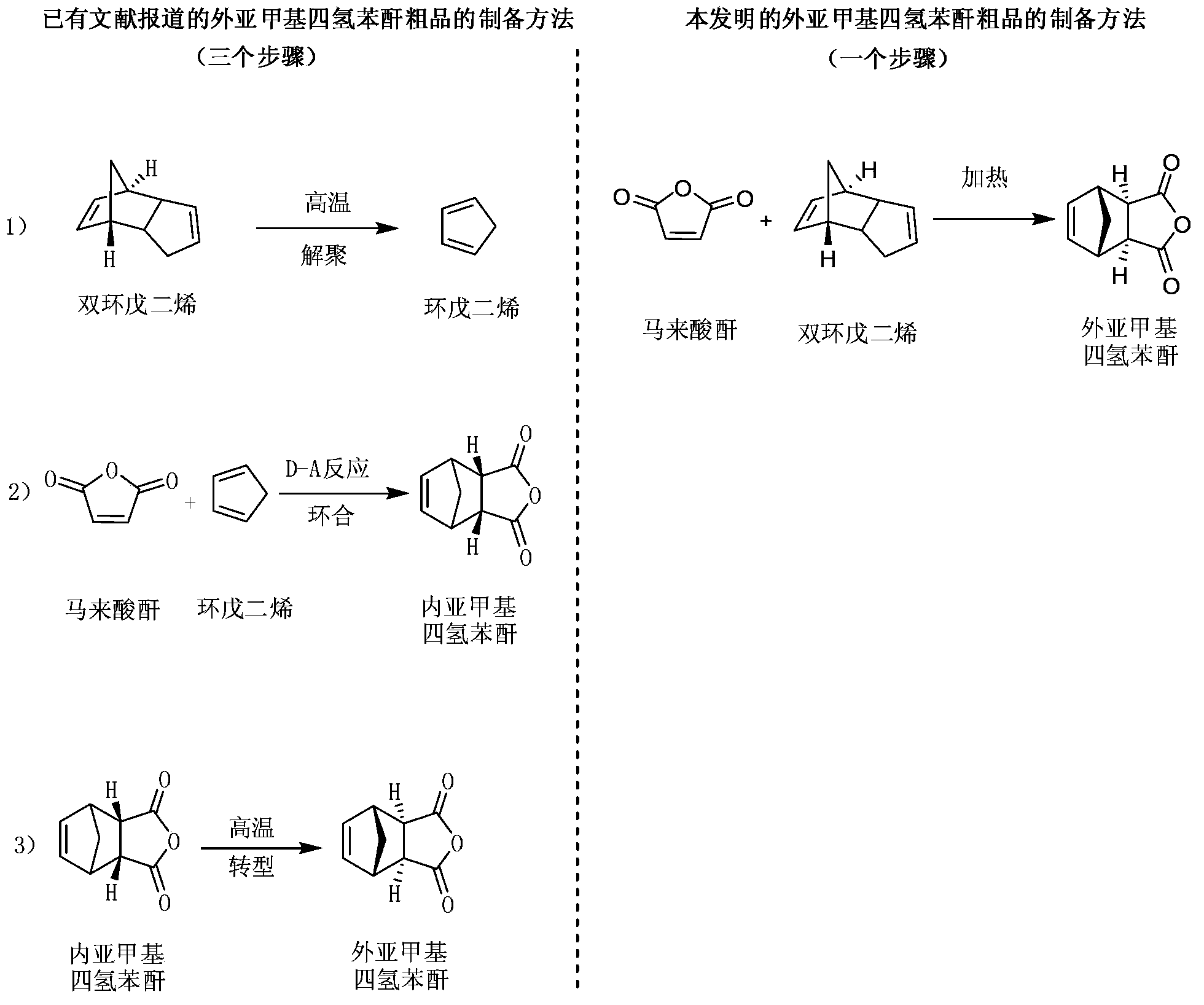
Preparation and refining method of exo-methylene tetrahydrophthalic anhydride and its use in preparation of tandospirone
ActiveCN103848801ASimple manufacturing processIncrease profitOrganic chemistryChemical recyclingDepolymerizationHigh energy
The invention relates to a preparation and refining method of exo-methylene tetrahydrophthalic anhydride and its use in preparation of tandospirone. The preparation and refining method realizes high-efficiency combination of depolymerization of dicyclopentadiene, cycloaddition of cyclopentadiene and maleic anhydride, and transformation of endo-methylene tetrahydrophthalic anhydride, is free of a single dicyclopentadiene depolymerization process, is free of a reaction intermediate product separation process, effectively solves the problem that the existing exo-methylene tetrahydrophthalic anhydride preparation method has defects of complex dicyclopentadiene depolymerization processes, low conversion rate, low yield, serious environmental pollution, high energy consumption, long production cycle and harsh storage condition requirements, and has the advantages of high yield, good quality, eco-friendly solvent, low cost and good industrial production applicability.
Owner:SICHUAN CREDIT PHARMA
Preparation method of palbociclib intermediate
ActiveCN112898299AMild reaction conditionsSimple post-processingOrganic chemistryAcetic anhydrideKetone
The invention discloses a preparation method of a palbociclib intermediate. The method comprises the following steps: preparing 5-bromine-2-chloro-N-cyclopentylamine pyrimidine-4 amine from 5-bromine-2,4-dichloropyrimidine and cyclopentylamine by taking solvents such as dichloromethane and water as solvents and taking inorganic base as an acid-binding agent; with DIEA as an acid-binding agent, DMF as a solvent and TBAB as a phase transfer catalyst, in the presence of water, catalyzing with a trace amount of palladium, and carrying out normal hexane reflux dehydration; further subjecting the acetic anhydride to dehydration cyclization, such that 2-chloro-8-cyclopentyl-5-methylpyridino[2,3-D]pyrimidine-7-(8H)-ketone is obtained; and reacting the obtained compound with NBS (N-bromosuccinimide) in acetonitrile to obtain the 6-bromo-2-chloro-8-cyclopentyl-5-methylpyridino[2, 3-D]pyrimidine-7(8H)-ketone. The method is mild in reaction, simple and convenient to operate, recyclable in solvent, less in environmental pollution, high in yield, low in cost, high in product quality and suitable for industrial production.
Owner:SHANDONG BOYUAN PHARM CO LTD
Refining separation method for coarse piperyene
ActiveCN1796341AHigh refining yieldHigh purityDistillation purification/separationCyclopenteneAcetonitrile
This is a process for the refinery and separation of crude m-pentadiene, which is obtained through the separation of the by-product C5 fraction of ethane manufacturing via petroleum pyrolysis, including the steps of: 1) directing the crude m-pentadiene into an extractive rectification column for extractive rectification, and collecting an overhead product of a mixed stream substantially enriched in cyclopentene and cyclopententane, and a bottom product of a mixed stream substantially enriched in m-pentadiene and the extractant, which is dimethyl formamide, N-methyl pyrrolidone, ethyl hydrazine or an aqueous solution of ethyl hydrazine containing 5-15 wt.% water; 2) directing the said bottom stream into a refinery column for refinery, and colleting an overhead product of the refined m-pentadiene, and a bottom product of the extractant, which is recovered and recycled; 3) directing the process 1 wherein said overhead stream into a refinery column for refinery, and collecting an overhead product of cyclopentene, and a bottom product of cyclopententane. This invention ensures a high recovery and refinery rate to obtain refined m-pentadiene product with a purity higher than 95%, and also obtains a mixture of cyclopentene and cyclopententane with a high utility value.
Owner:SINOPEC SHANGHAI PETROCHEMICAL CO LTD
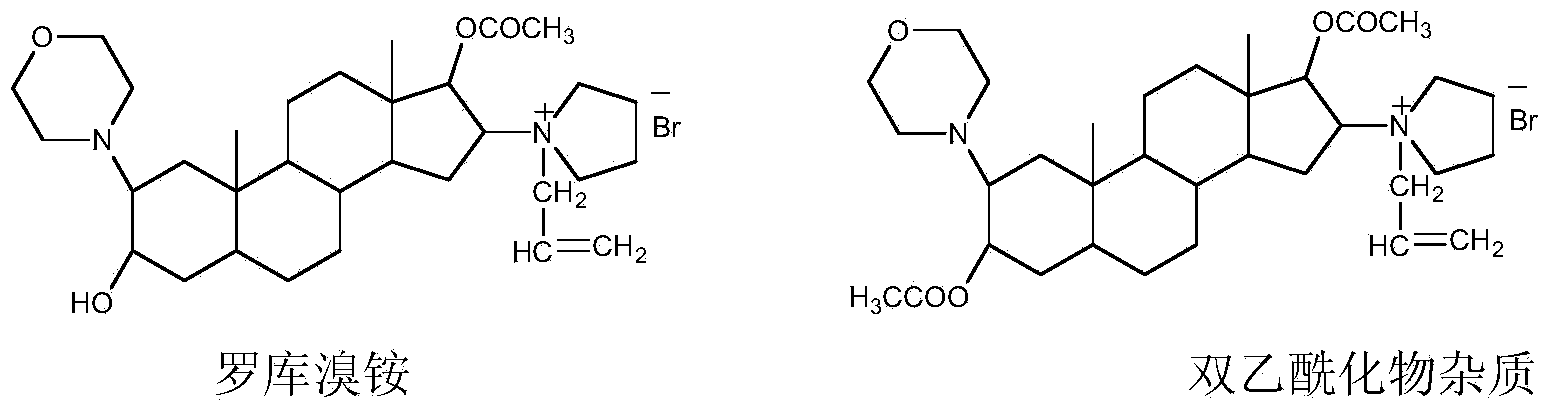
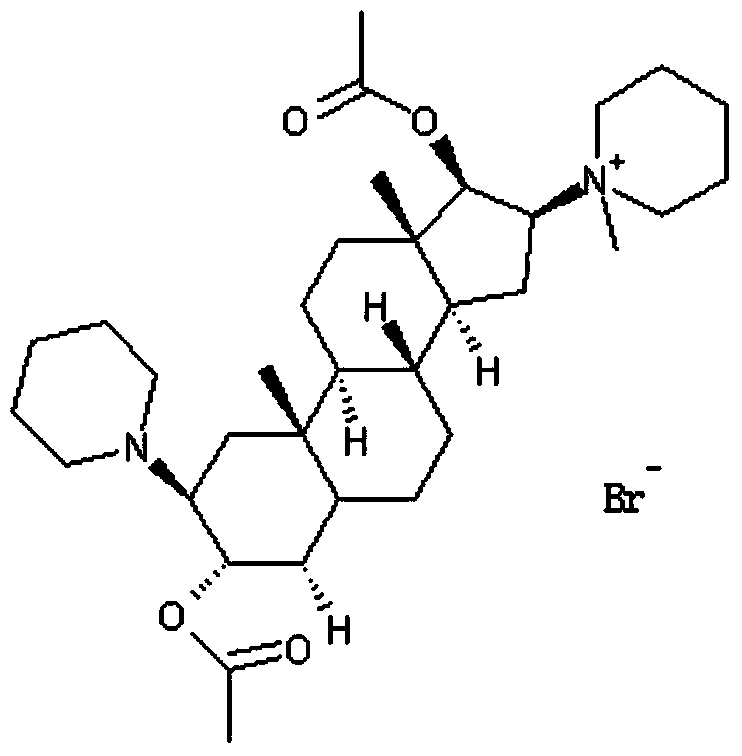
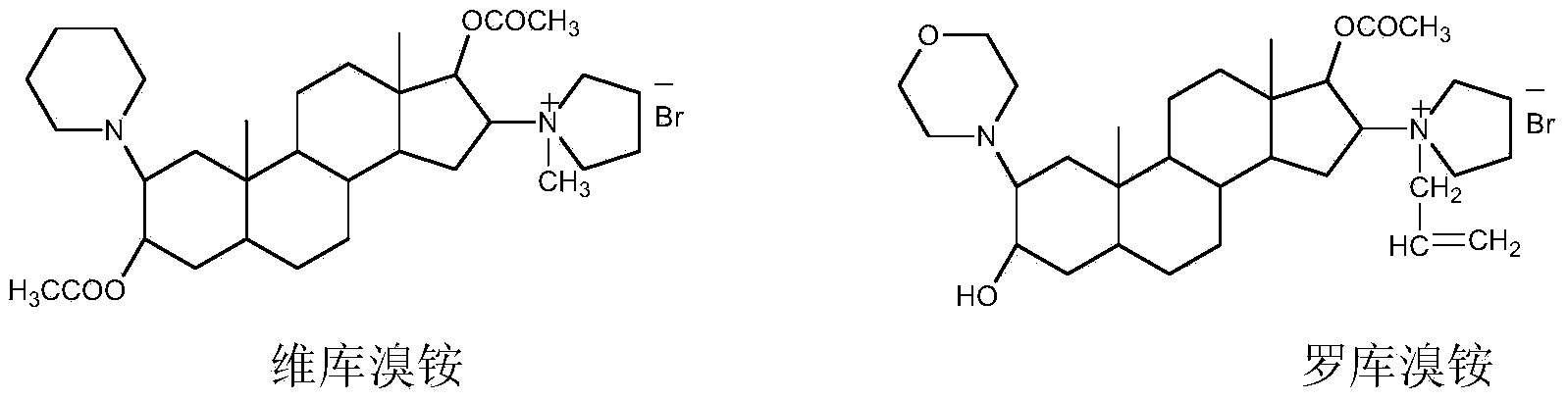
Method for refining steroid muscle relaxant
The invention discloses a method for refining a steroid muscle relaxant. The method for refining the steroid muscle relaxant comprises the following steps: firstly mixing the steroid muscle relaxant with water and acid to obtain an acidic salt aqueous solution, and washing by adopting an organic solvent A; then alkalifying the acidic salt aqueous solution, extracting the alkalified aqueous solution by adopting the organic solvent A, drying the collected extraction organic solvent A, filtering, concentrating to small volume, slowly dropping the concentrated organic solvent A into an organic solvent B, filtering, and drying to obtain a refined steroid muscle relaxant product. The method for refining the steroid muscle relaxant is simple in technology, good in controllability and strong in operability, industrialization production is easy to realize, only is once salifying alkalization operation needed, impurity content is obviously reduced, a better economic benefit during the industrialization production can be obtained, and production efficiency can be obviously improved.
Owner:YICHANG HUMANWELL PHARMA
Refining method of sodium esomeprazole
The invention discloses a refining method of a sodium esomeprazole crude product by adopting a mixed solvent re-crystallization method. The method is simple and convenient to operate, high in product purity, low in content of a single impurity (the content of sulphone is less than 0.1 percent), high in yield, and more suitable for industrially largely producing the sodium esomeprazole.
Owner:LUNAN BETTER PHARMA
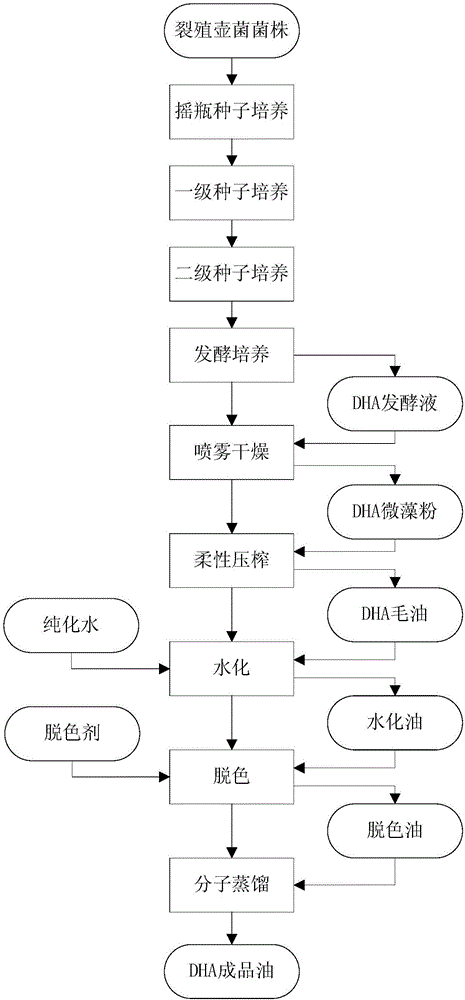
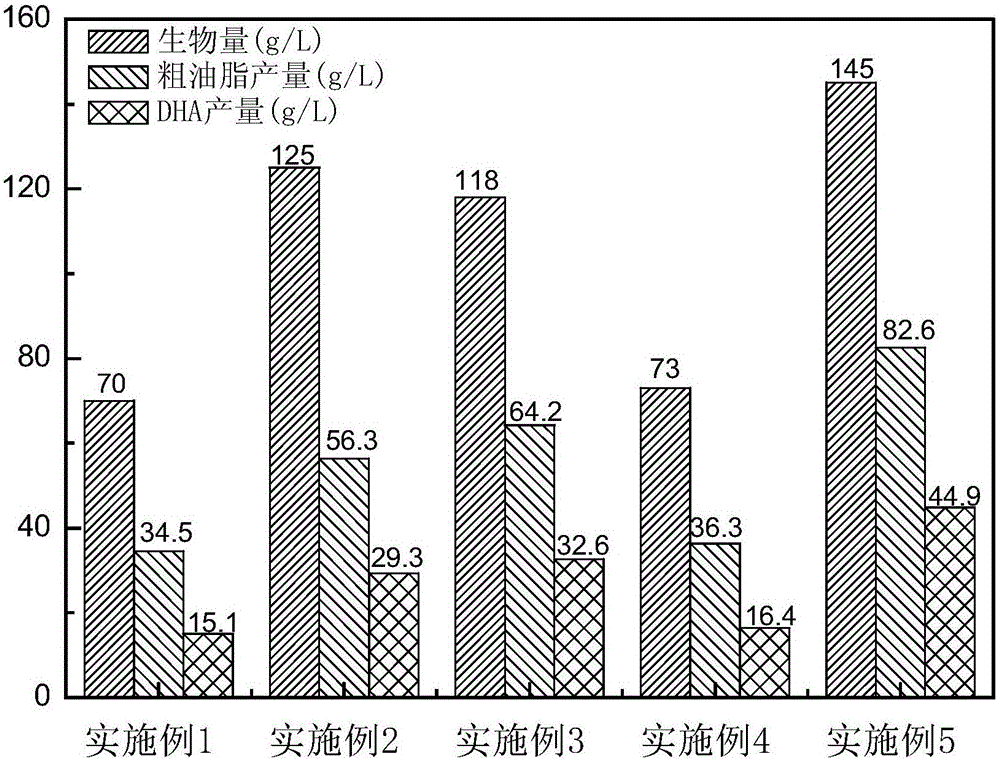
Method for producing DHA (Docosahexaenoic Acid) by utilizing microorganism fermentation
ActiveCN106636235AIncrease productionLow costFatty-oils/fats refiningMicroorganism based processesBiotechnologyMicroorganism
The invention belongs to the field of fermentation engineering, relates to a method for producing DHA (Docosahexaenoic Acid) by utilizing microorganism fermentation and in particular relates to a method for producing mixed oil containing the DHA by utilizing industrial fermentation of schizochytrium strains. More specifically, the invention relates a method for cultivating microorganisms for producing the DHA, wherein the dissolved oxygen saturation is controlled to be 5 percent to 10 percent from 36h to 60h of a culture process of a fermentation tank, and / or a nitrogen source is not added any more from 36h to 60h of the culture process of the fermentation tank. When the method provided by the invention is used for preparing the DHA, the yield is high and the purity is high; large-scale industrial production of the DHA is facilitated.
Owner:INNER MONGOLIA KINGDOMWAY PHARMA LTD +1
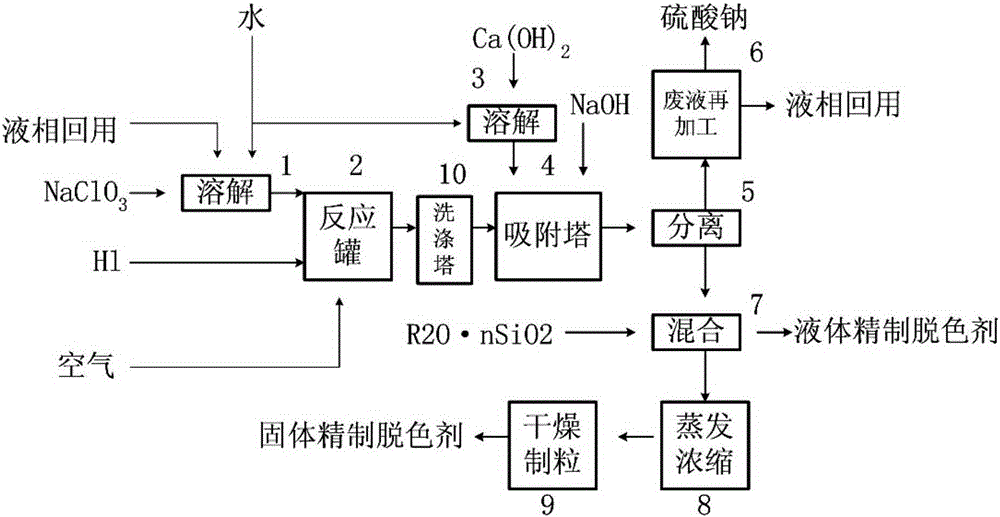
Preparation and application of refined decolorizing agent and cleaning for edible oil
InactiveCN105251440ASuitable for purification and decolorizationEliminate residueOther chemical processesFatty-oils/fats refiningEnvironmental resistanceChemical reaction
Provided are preparation and application of a refined decolorizing agent and cleaning for edible oil. Composite available chlorine gas is obtained through a chlorate aqueous solution under the condition of taking inorganic acid or organic acid as an oxidation reductant, a secondary chemical reaction is conducted for adsorption through stabilizer carrier hydroxide and a silicon compound, and a granular composite finished product of the refined decolorizing agent is obtained through concentration, fluidized drying and granulation; particularly, acid reaction mother liquor is not taken as waste liquor to be discharged, and the reaction mother liquor is reused. The preparation and application of the refined decolorizing agent and cleaning for the edible oil can be used for animal and vegetable oil refined decolorizing and specially for edible oil refined decolorizing, and the industry can respond to the important contribution on the aspects of energy conservation and emission reduction provided by the country; the edible oil industry can save energy by over 80 percent, and discharge of the waste clay residue containing oil is reduced by 100 percent, and the preparation and application of the refined decolorizing agent and cleaning for the edible oil can be further used for other food and organic matter decolorizing. The product has important environmental and economic value, wide application and promotion prospects are achieved, and the product takes the leading position in the world.
Owner:TIANJIN JINLONGCHANG TECH CO LTD
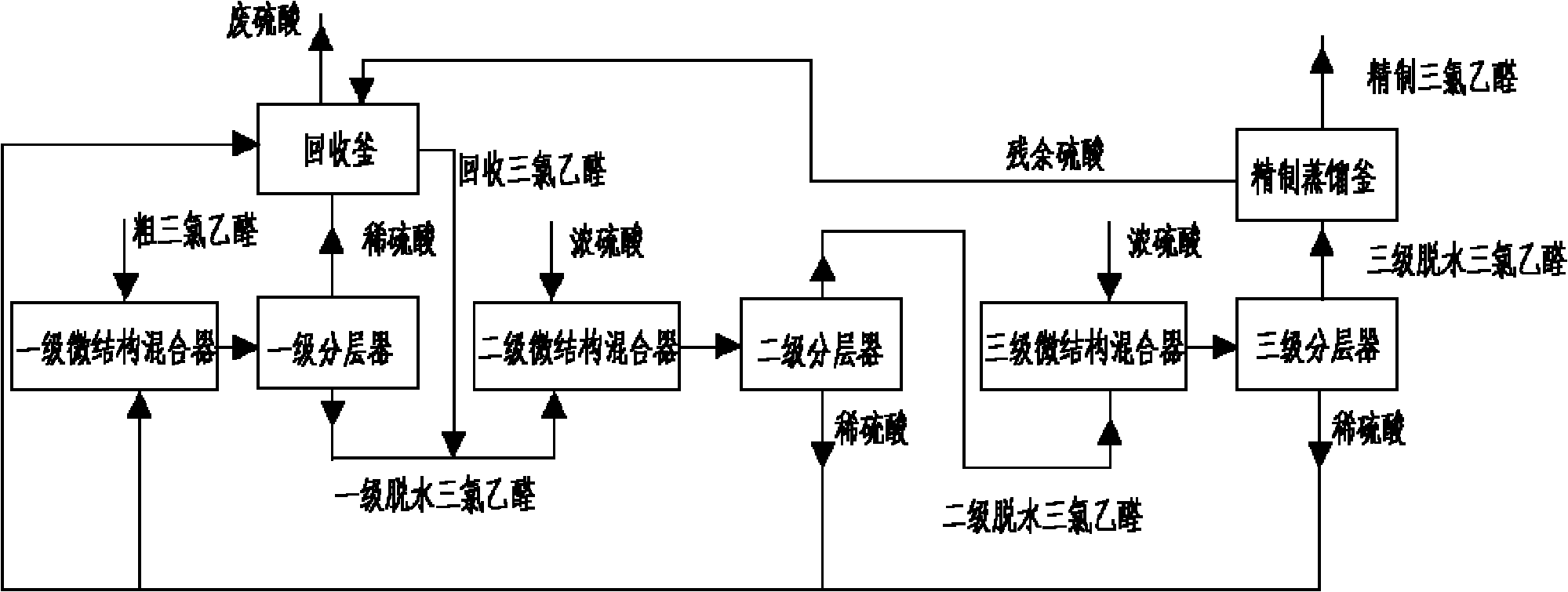
Method for purifying coarse trichloroacetaldehyde
InactiveCN102030623AEasy to dehydrateGuaranteed dehydrationCarbonyl compound separation/purificationDistillationMicrostructure
The invention discloses a method for purifying coarse trichloroacetaldehyde. The method comprises the following steps that: the coarse trichloroacetaldehyde is fully mixed with dilute sulphuric acid in a primary microstructure mixer; trichloroacetaldehyde obtained by a primary demixer is fully mixed with concentrated sulfuric acid in a secondary microstructure mixer; trichloroacetaldehyde obtained after being demixed by a secondary demixer is continuously and fully mixed with concentrated sulfuric acid in a third microstructure mixer; trichloroacetaldehyde containing a small amount of sulfuric acid obtained after being demixed by a third demixer is rectified to obtain anhydrous trichloroacetaldehyde; after sulfuric acid demixed by the secondary demixer and the third demixer is mixed, a part of sulfuric acid is used as the dilute sulphuric acid for the primary microstructure mixer, and the residual sulfuric acid is mixed with the sulfuric acid demixed by the primary microstructure mixer and the residual sulfuric acid of a refining distillation kettle to distill and recycle the residual trichloroacetaldehyde; and the recycled trichloroacetaldehyde is mixed with the trichloroacetaldehyde demixed by the primary demixer to enter the secondary microstructure mixer. By the method, the coarse trichloroacetaldehyde is purified, the consumption of steam and the sulfuric acid can be reduced, and the refining yield of the trichloroacetaldehyde is improved.
Owner:XIANGTAN UNIV
Refining technology of caffeine in caffeine production process
The invention relates to a refining technology of caffeine in a caffeine production process. The refining technology comprises the following steps: oxidizing caffeine refining material liquid after primary refining is completed, carrying out secondary refining on the oxidized refining material liquid after decoloration through a carbon fiber filter, after the secondary refining is completed, pressing the material liquid in an intermediate tank after decoloration through the carbon fiber filter, and then pressing in a finished product crystallizing tank from the intermediate tank. The refining technology effectively reduces the use of organic solvent chloroform during carbon treatment, and the COD (Chemical Oxygen Demand) is not increased. The use amount of active carbon in the refining process is reduced, and the consumption of caffeine in the refining process is lowered. The amount of the active carbon is reduced, and the refining yield is greatly promoted. Active carbon fibers can be regenerated through hot water boiling, and the treatment method is simple. The subsequent carbon treatment cost is greatly reduced.
Owner:SHANDONG XINHUA PHARMA CO LTD
Refining method of adiponitrile
PendingCN113429316AHigh purityHigh refining yieldCarboxylic acid nitrile purification/separationProcess engineeringHydrolysis
The invention provides a refining method of adiponitrile, which is characterized in that ICCP in crude adiponitrile is hydrolyzed and converted into 2-cyanocyclopentanone, and then the 2-cyanocyclopentanone is rectified and removed to obtain high-purity adiponitrile. The principle of the invention is as follows: under the pressure of 2.0 KPa, the boiling point of 2-cyanocyclopentanone is 137-140 DEG C, the boiling point of ICCP is 152-154 DEG C, and the boiling point of adiponitrile is 168 DEG C, so that the 2-cyanocyclopentanone is separated from the adiponitrile more easily than the ICCP in the subsequent rectification separation process, and the amount of the adiponitrile taken out when the 2-cyanocyclopentanone is distilled out from the tower top is much lower than the amount of the adiponitrile taken out when the ICCP is separated. Therefore, the refining method of the adiponitrile provided by the invention is simple; The ICCP in the crude adiponitrile is converted into the 2-cyanocyclopentanone, so that the removal effect of the ICCP is good, the refining yield of the adiponitrile is higher, and the purity of the refined adiponitrile product is also high. The method can better meet the quality requirements of downstream products on adiponitrile; specifically, the content of adiponitrile in the obtained product is 99.9% or above, and the content of ICCP is less than 50 ppm.
Owner:瑞典国际化工技术有限公司
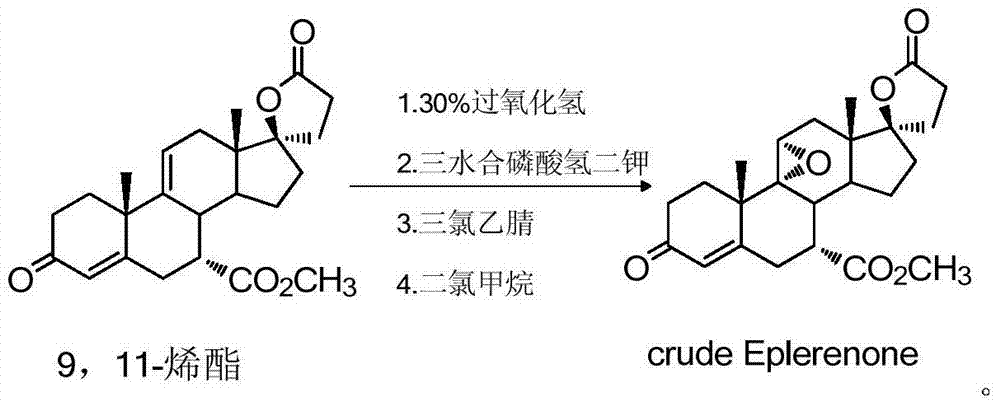
Method for preparing and refining eplerenone
InactiveCN104262450AShort reaction timeReduce manufacturing costSteroidsTrichloroacetonitrileHydrogen phosphate
The invention discloses a method for preparing and refining eplerenone. The method for preparing and refining eplerenone comprises the following steps: taking 9,11-alkenyl ester to react at 0-30 DEG C in the presence of potassium hydrogen phosphate trihydrate, trichloroacetamide and hydrogen peroxide so as to prepare an eplerenone crude product; carrying out re-crystallization by 2-butanone to obtain an eplerenone fine product with a purity of greater than 99.9%. The method is simple to operate; poisonous and harmful trichloroacetonitrile is not used; the use amount of hydrogen peroxide is greatly reduced; the production period is greatly shortened; the yield and purity of products are greatly improved; the method is suitable for industrial application.
Owner:JIANGSU SINOBIOPHARMA
Method for preparing high-purity white 2-hydroxy quinoxaline crystal
InactiveCN102010377AReduce dosageThe refining process is simpleOrganic chemistryQuinoxalinePotassium borohydride
The invention discloses a method for preparing 2-hydroxy quinoxaline by condensing glyoxylic acid and o-phenylenediamine. The method is characterized by comprising the following steps of: condensing industrial glyoxylic acid solution and dry ice-precooled o-phenylenediamine at the temperature of between -6 and 0 DEG C to obtain a crude product of the 2-hydroxy quinoxaline; heating and refluxing a colored crude product for 1 to 4 hours in a mixed solvent of N,N-dimethylformamide, diethylene glycol or N-methyl pyrrolidone and methanol in the presence of a reducing agent such as sulfur dioxide, sodium hypophosphite or potassium borohydride; dissolving impurities in the mixed solvent; converting a fine-powder crude product into white crystals; and filtering, washing and drying to obtain a refined product of the 2-hydroxy quinoxaline. Through liquid chromatography measurement, the content is 99.5 percent, the melting point is 270 to 272 DEG C and the refining yield is 97.5 percent. The 2-hydroxy quinoxaline has a simple refining process; a high-purity white crystal product can be obtained; the refining yield is high; the dosage of refining solvent is less; and the solvent can be recycled for use by heating, refluxing and discoloring by using activated carbon.
Owner:TIANJIN VOCATIONAL INST
Synthetic method of azithromycin impurity F
ActiveCN108727445AThe method steps are simpleHigh yieldSugar derivativesSugar derivatives preparationAzithromycinQuality control
The invention belongs to the technical field of chemistry, and particularly relates to a synthetic method of azithromycin impurity F. The combination method of the azithromycin impurity F-(2R,3S,4R,5R,8R,10R,11R,12S,13S,14R)-13-[(2,6-dideoxy-3-C-methyl-3-O-methyl-alpha-L-nucleus-pyranohexyl pyrranosyl)oxy]-2-ethyl-3,4,10-trihydroxy-3,5,6,8,10,12,14-heptamethyl-11-[[3,4,6-trideoxy-3-(N-methylformamido)-beta-D-pyranohexyl]oxy]-1-oxa-6-azacyclopentadecane-15 ketone in (formula I) comprises the main steps as follows: enabling azithromycin (compound II) to be subjected to 3'-N demethylation, performing formylation by using ethyl formate to generate a target product; purifying and then obtaining oxidized impurity with purity greater than 99.5%. The synthesized high-purity azithromycin impurity Fserves as impurity standard substance for finished product detection, is conducive to reinforcing location and nature determination of impurity, and improves quality control on azithromycin bulk drugs.
Owner:HEC PHARM