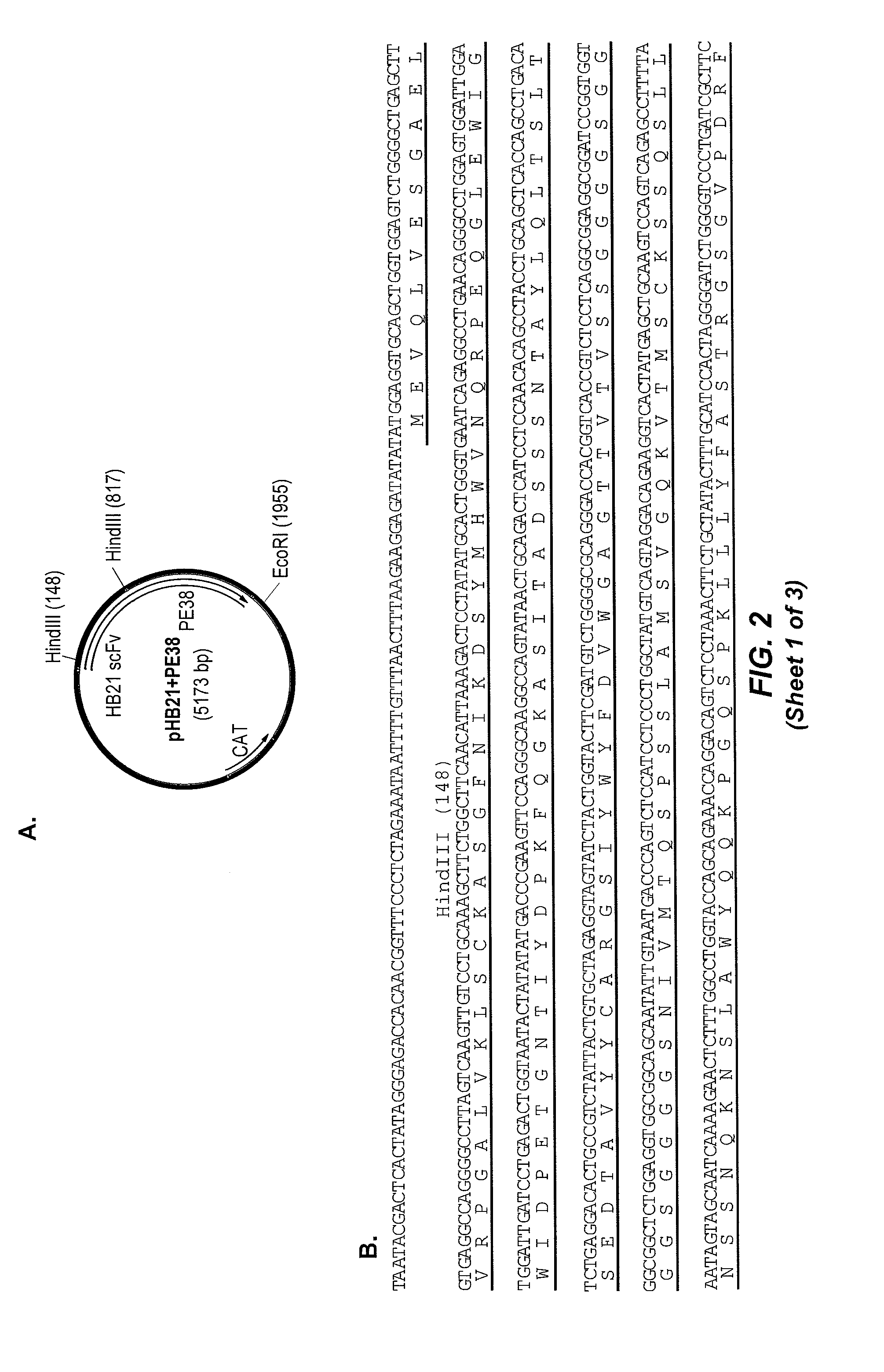
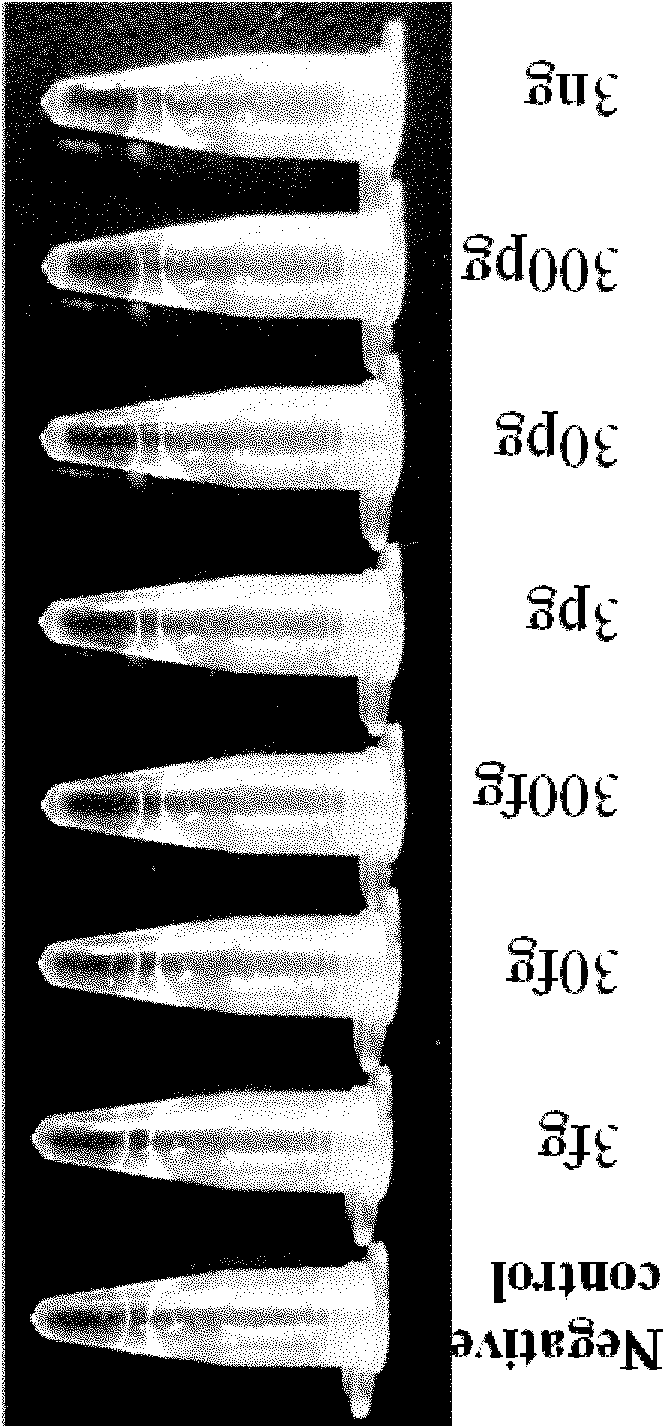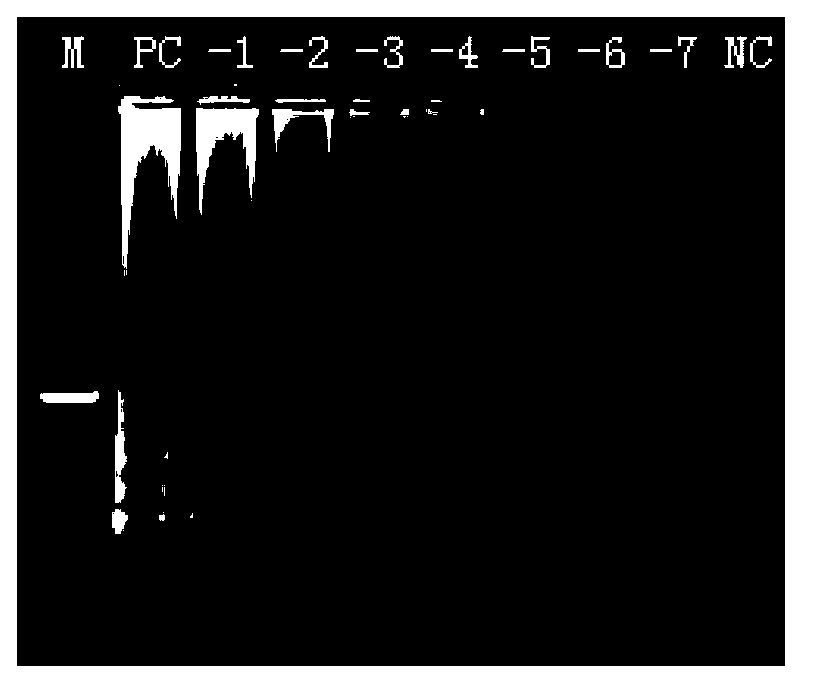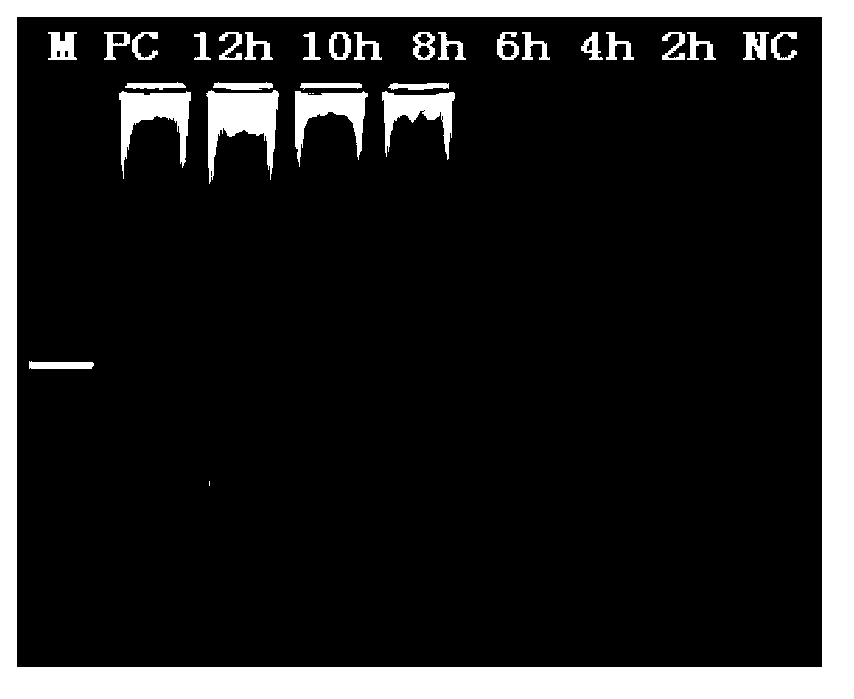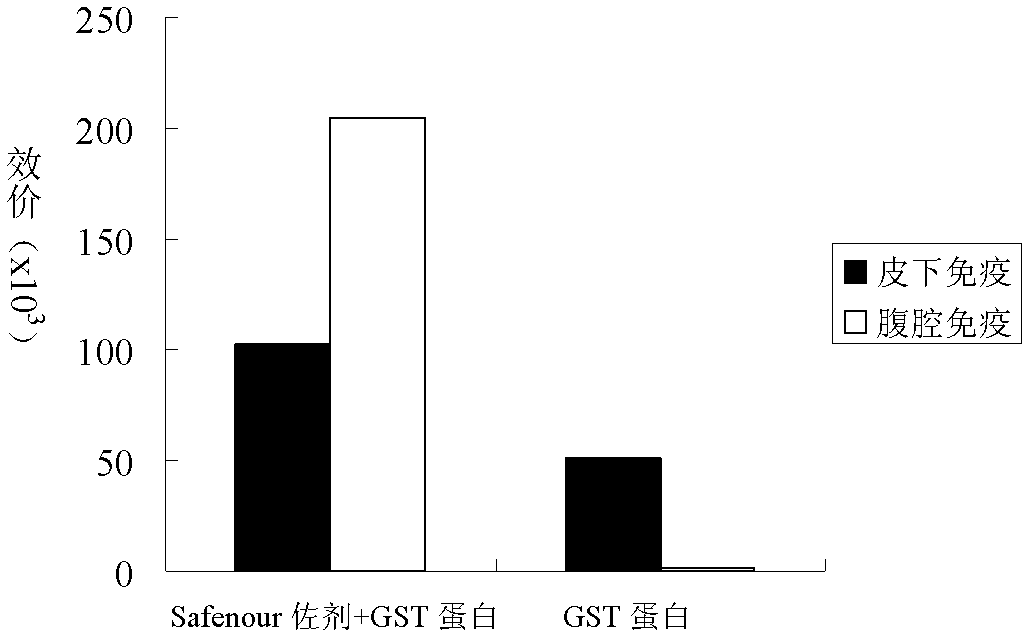Patents
Literature
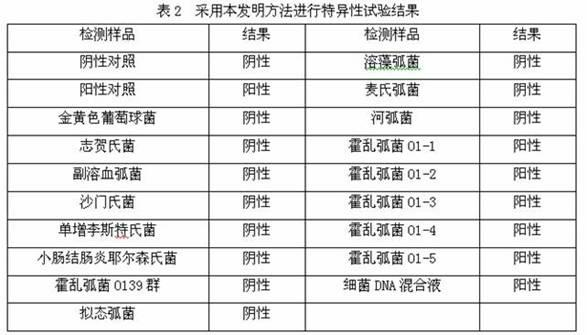
181 results about "Vibrio cholerae" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
Vibrio cholerae is a Gram-negative, comma-shaped bacterium. The bacterium's natural habitat is brackish or saltwater. Some strains of V. cholerae cause the disease cholera. V. cholerae is a facultative anaerobe and has a flagellum at one cell pole as well as pili. V. cholerae can undergo respiratory and fermentative metabolism. When ingested, V. cholerae can cause diarrhoea and vomiting in a host within several hours to 2–3 days of ingestion. V. cholerae was first isolated as the cause of cholera by Italian anatomist Filippo Pacini in 1854, but his discovery was not widely known until Robert Koch, working independently 30 years later, publicized the knowledge and the means of fighting the disease.
Broad-Spectrum Antibacterial and Antifungal Activity of Lactobacillus Johnsonii D115
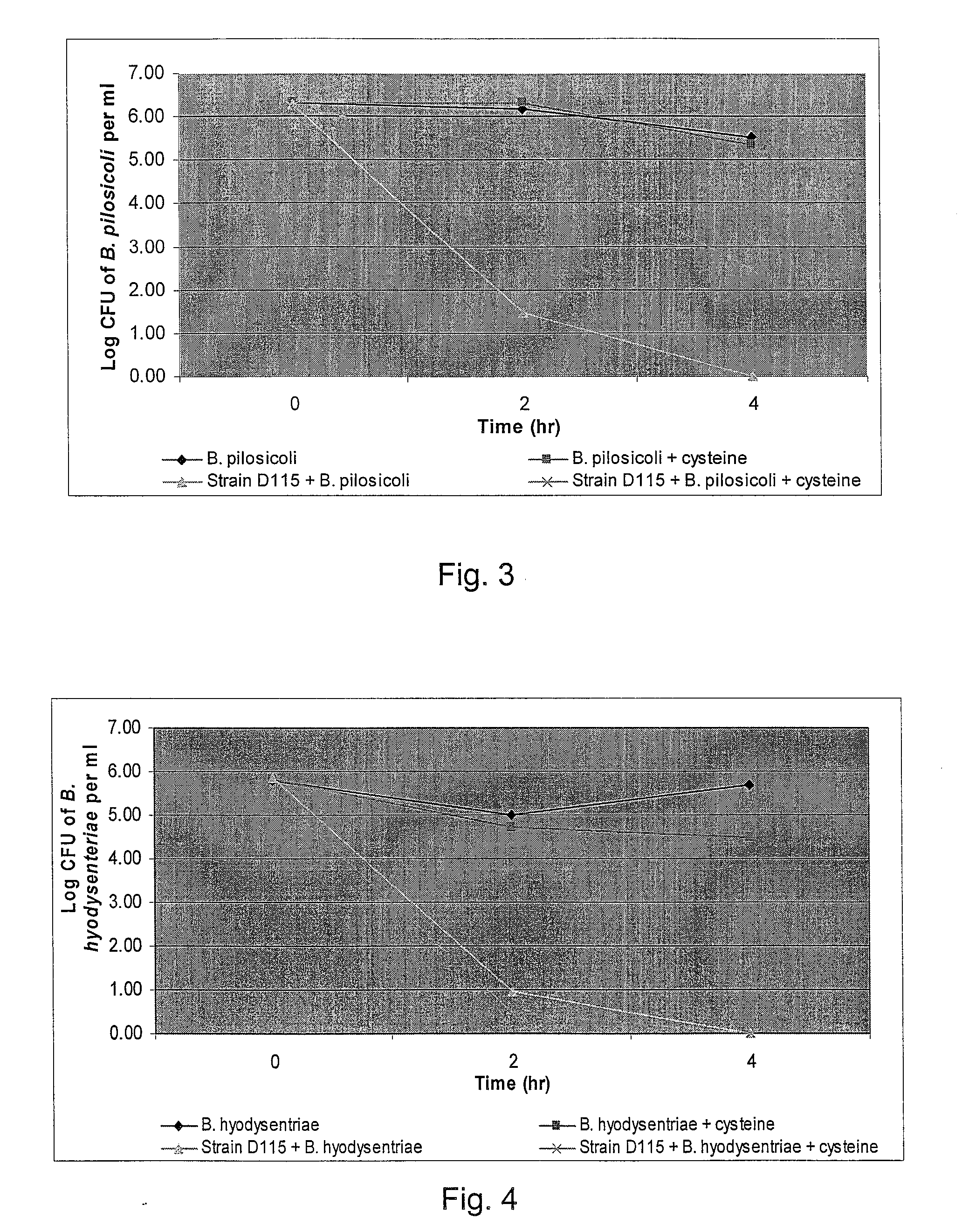
The present invention demonstrated the potential use of Lactobacillus johnsonii D115 as a probiotic, as a prophylactic agent or as a surface treatment of materials against human and animal pathogens such as Brachyspira pilosicoli, Brachyspira hyodysenteriae, Shigella sonnei, Vibrio cholera, Vibrio parahaemolyticus, Campylobacter jejuni, Streptococcus pneumoniae, Enterococcus faecalis, Enterococcus faecium, Clostridium perfringens, Yersinia enterocolitica, Escherichia coli, Klebbsiella pneumoniae, Staphylococcus aureus, Salmonella spp., Bacillus cereus, Aspergillus niger and Fusarium chlamydosporum. The proteineous antimicrobial compound was partially characterized and found to be heat tolerant up to 121° C. for 15 min, and acid tolerant up to pH1 for 30 min at 40° C. The compound is also stable to enzymatic digestion, being able to retain more than 60% antimicrobial activity when treated with pepsin and trypsin.
Owner:KEMIN IND INC
Pharmaceutical proteins, human therapeutics, human serum albumin, insulin, native cholera toxic b submitted on transgenic plastids
InactiveUS20030204864A1Eliminate needLarge biomassBiocidePeptide/protein ingredientsEscherichia coliInsulin-like growth factor
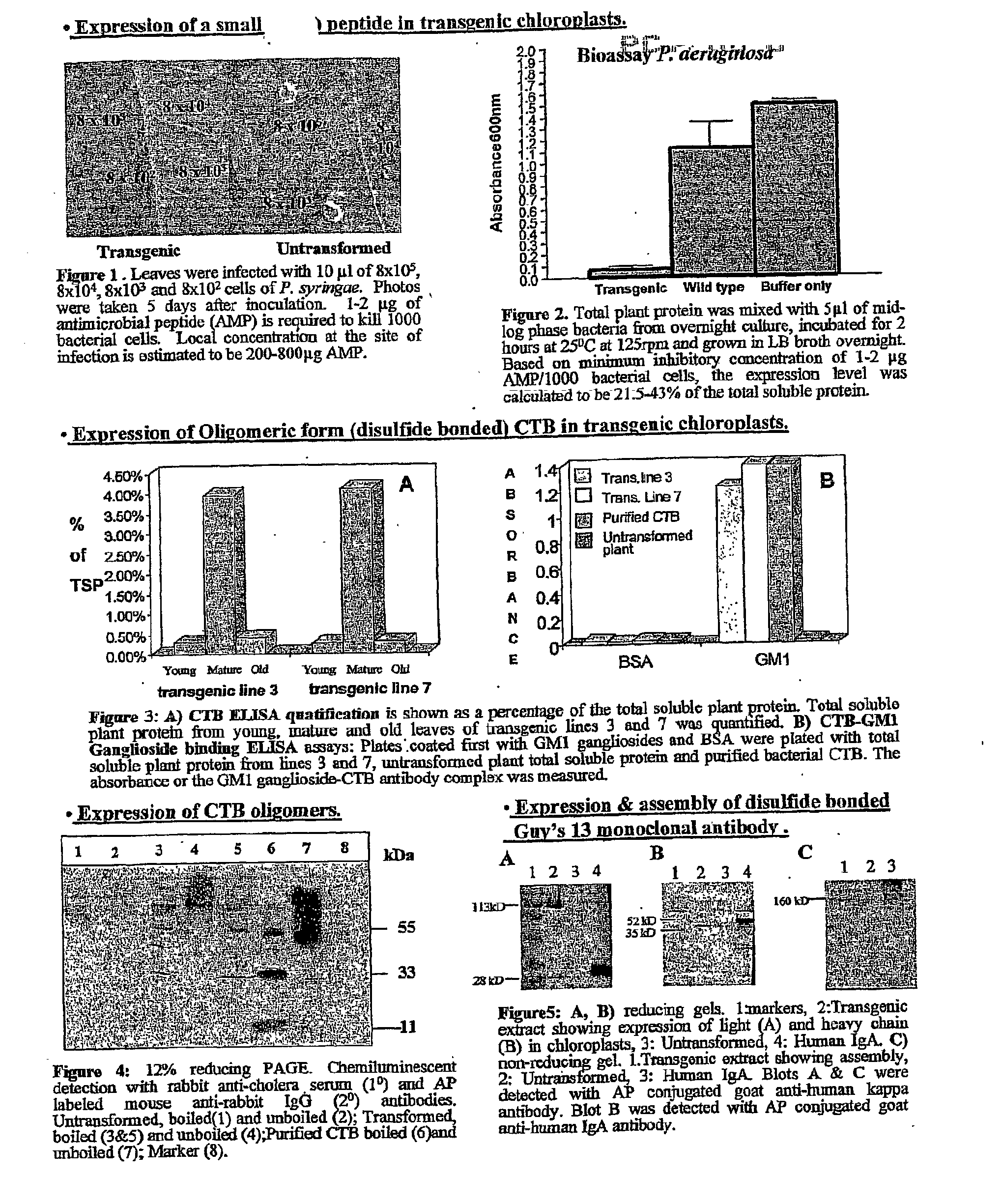
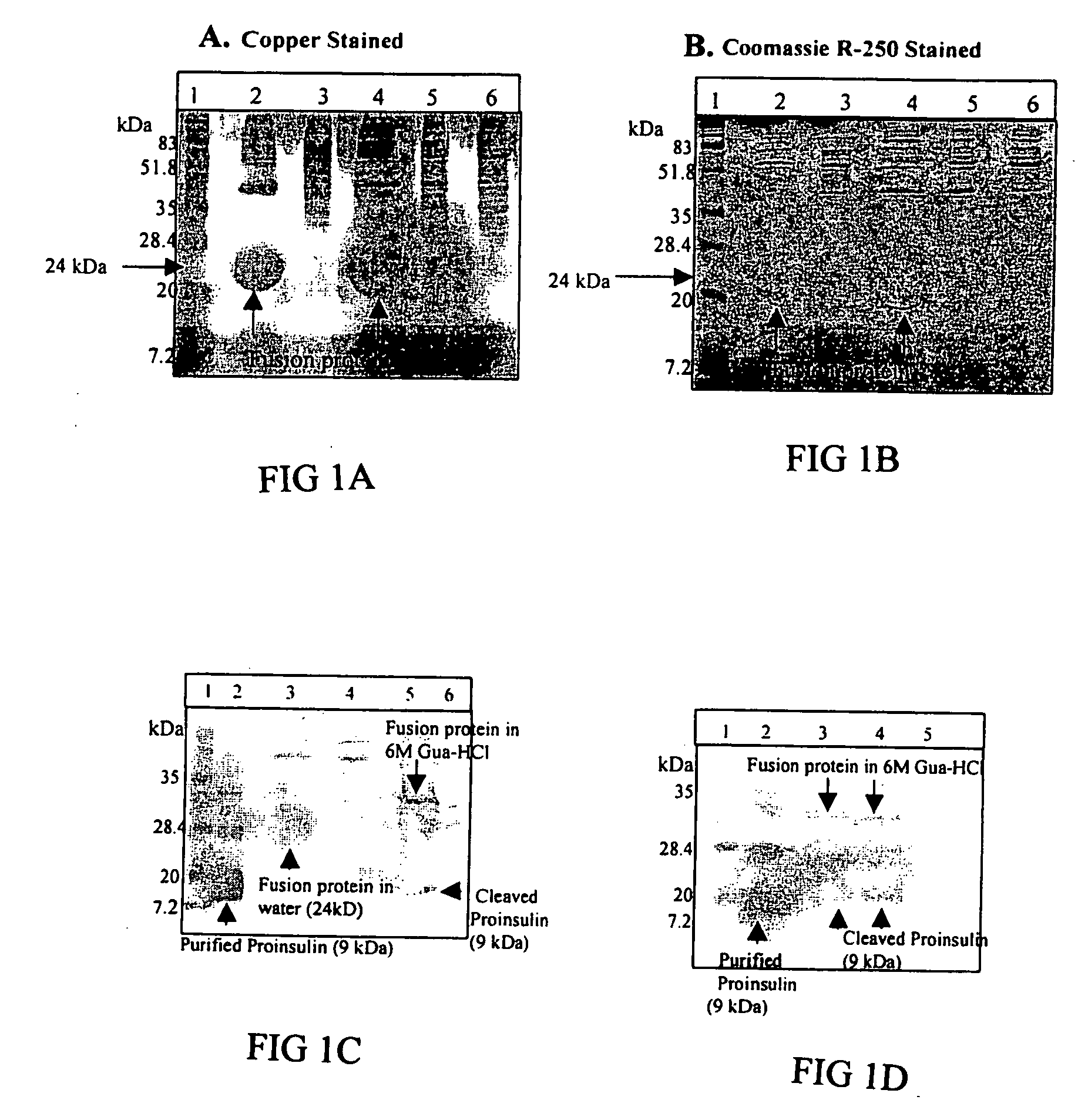
Transgenic chloroplast technology could provide a viable solution to the production of Insulin-like Growth Factor I (IGF-I), Human Serum Albumin (HSA), or interferons (IFN) because of hyper-expression capabilities, ability to fold and process eukaryotic proteins with disulfide bridges (thereby eliminating the need for expensive post-purification processing). Tobacco is an ideal choice because of its large biomass, ease of scale-up (million seeds per plant), genetic manipulation and impending need to explore alternate uses for this hazardous crop. Therefore, all three human proteins will be expressed as follows: a) Develop recombinant DNA vectors for enhanced expression via tobacco chloroplast genomes b) generate transgenic plants c) characterize transgenic expression of proteins or fusion proteins using molecular and biochemical methods d) large scale purification of therapeutic proteins from transgenic tobacco and comparison of current purification / processing methods in E. coli or yeast e) Characterization and comparison of therapeutic proteins (yield, purity, functionality) produced in yeast or E. coli with transgenic tobacco f) animal testing and pre-clinical trials for effectiveness of the therapeutic proteins. Mass production of affordable vaccines can be achieved by genetically engineering plants to produce recombinant proteins that are candidate vaccine antigens. The B subunits of Enteroxigenic E. coli (LTB) and cholera toxin of Vibrio cholerae (CTB) are examples of such antigens. When the native LTB gene was expressed via the tobacco nuclear genome, LTB accumulated at levels less than 0.01% of the total soluble leaf protein. Production of effective levels of LTB in plants, required extensive codon modification. Amplification of an unmodified CTB coding sequence in chloroplasts, up to 10,000 copies per cell, resulted in the accumulation of up to 4.1% of total soluble tobacco leaf protein as oligomers (about 410 fold higher expression levels than that of the unmodified LTB gene). PCR and Southern blot analyses confirmed stable integration of the CTB gene into the chloroplast genome. Western blot analysis showed that chloroplast synthesized CTB assembled into oligomers and was antigenically identical to purified native CTB. Also, GM1,-ganglioside binding assays confirmed that chloroplast synthesized CTB binds to the intestinal membrane receptor of cholera toxin, indicating correct folding and disulfide bond formation within the chloroplast. In contrast to stunted nuclear transgenic plants, chloroplast transgenic plants were morphologically indistinguishable from untransformed plants, when CTB was constitutively expressed. The introduced gene was stably inherited in the subsequent generation as confirmed by PCR and Southern blot analyses. Incrased production of an efficient transmucosal carrier molecule and delivery system, like CTB, in transgenic chloroplasts makes plant based oral vaccines and fusion proteins with CTB needing oral administration a much more practical approach.
Owner:AUBURN UNIV +1
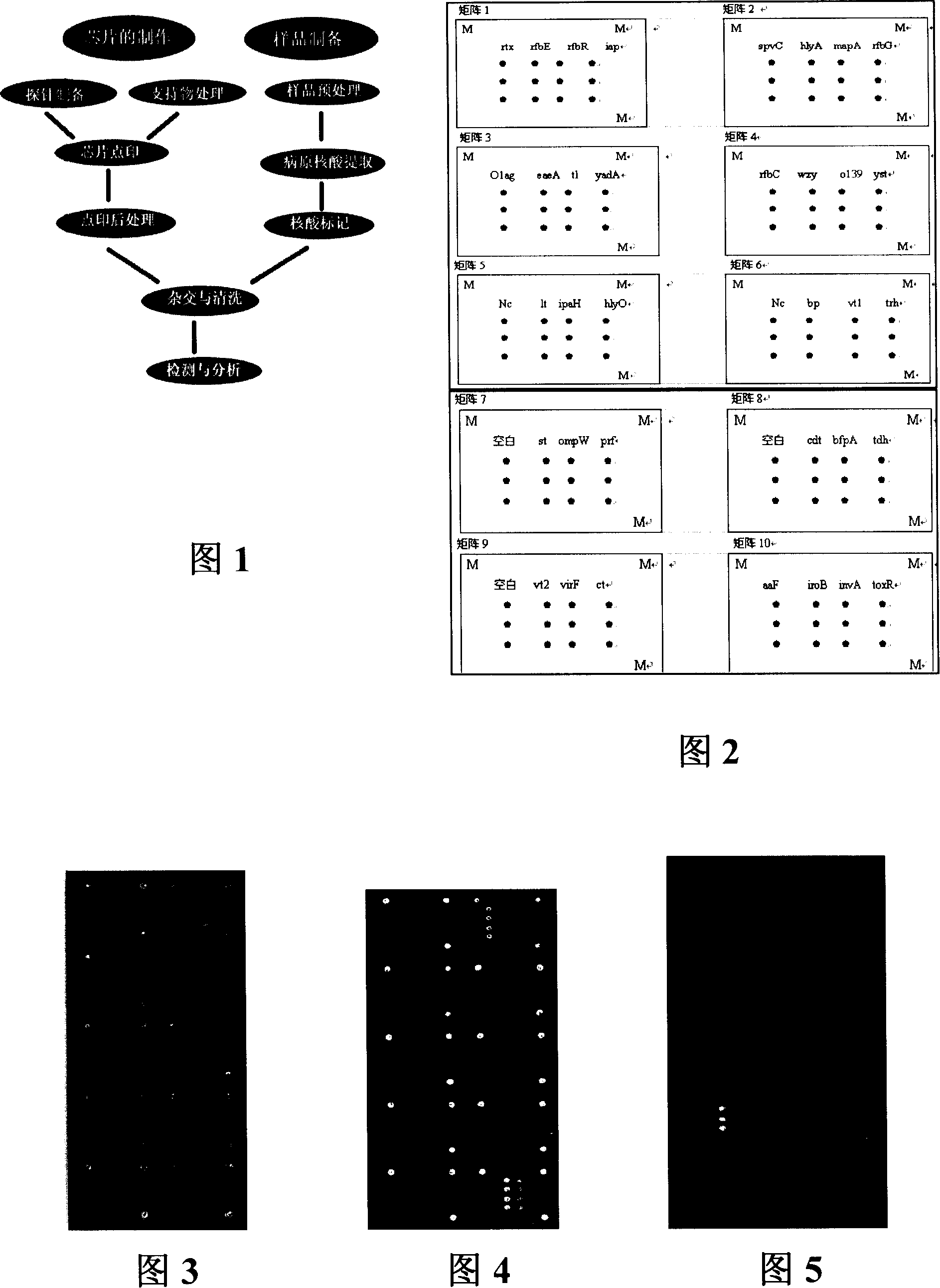
Pathogenic microorganism DNA detecting chip and preparation method and application thereof
InactiveCN101113476ASensitive hybridization detectionAccurate readingMicrobiological testing/measurementAgainst vector-borne diseasesESCHERICHIA COLI ANTIGENNucleic Acid Probes
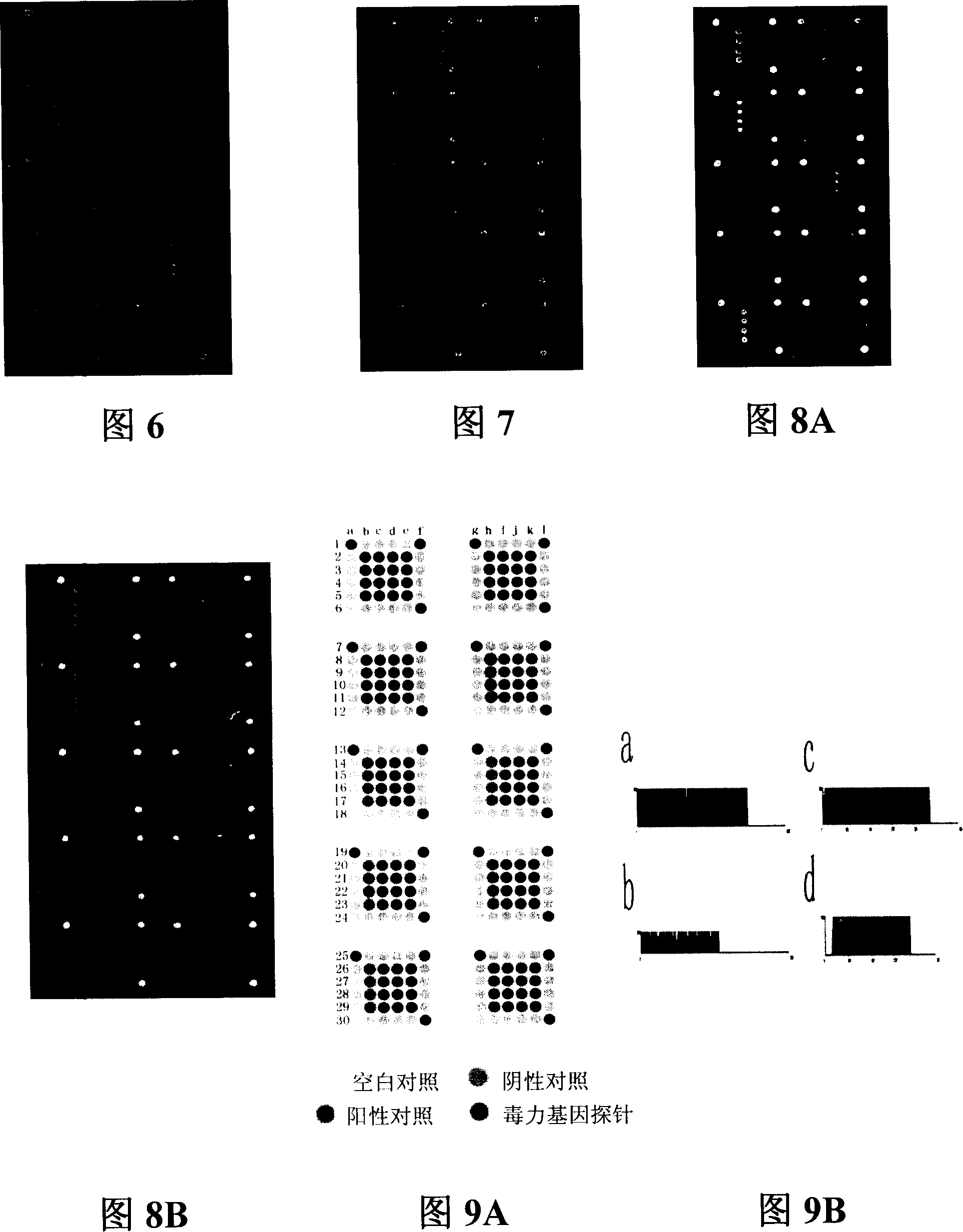
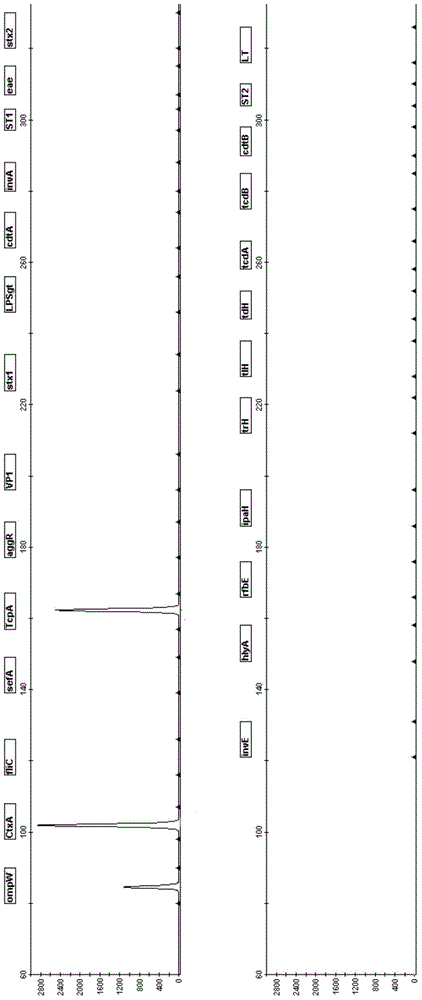
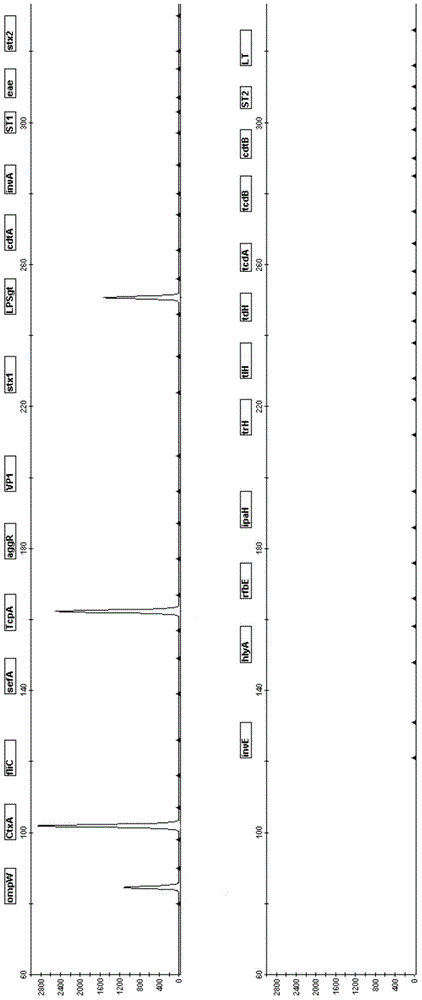
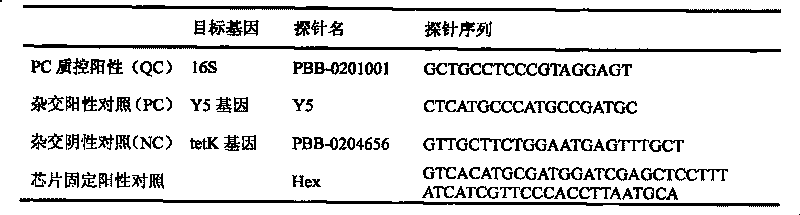
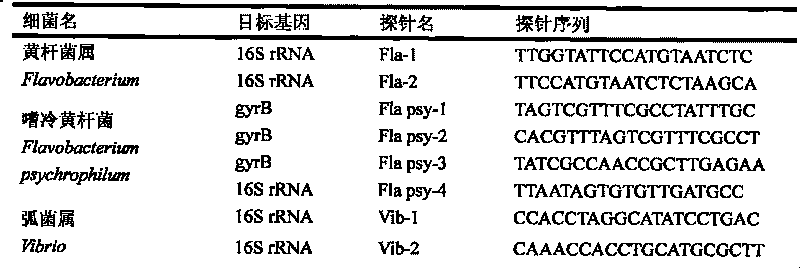
The invention relates to a pathogenic microorganism DNA detection chip and the chip comprises a carrier and a nucleic acid probe on the carrier. The nucleic acid probe is provided with a target detection probe used for detecting target gene of pathogenic microorganism. The pathogenic microorganism comprises but not limits to vibrio cholerae, pathogenic escherichia coli, campylobacter jejuni, Yersinia enterocolitica, parahemolytic vibrio, salmonella, and shigella and Listeria monocytogenes. The invention also relates to a preparation method of the pathogenic microorganism DNA detection chip and provides applications of the pathogenic microorganism DNA detection chip in detection of pathogenic microorganism. Also a pathogenic microorganism DNA detection chip kit is provided.
Owner:ICDC CHINA CDC
Fourteen-food-borne pathogenic bacterium multiplex PCR detection primer set and kit
ActiveCN103484546ATo achieve the detection effectMicrobiological testing/measurementAgainst vector-borne diseasesBacteroidesEscherichia coli
The invention discloses a fourteen-food-borne pathogenic bacterium multiplex PCR detection primer set and a kit. The detection primer set is composed of primer pairs for detecting salmonella, Shigella, Vibrio parahemolyticus, campylobacter jejuni, campylobacter coli, staphylococcus aureus, bacillus cereus, Listeria monocytogenes, yersinia enterocolitica, enterobacter sakazakii, Escherichia coli, vibrio cholerae, Escherichia coli O157, aeromonas hydrophila and internal positive control. A multiplex PCR detection method based on an ordinary PCR platform is built, multiplex PCR reactions are carried out on genomic DNA, extracted from a sample to be tested, of bacteria in the same reaction system through the primer pairs acquired through analysis and design, and whether the food-borne pathogenic bacteria are contained in the sample or not is judged through the electrophoretic analysis of reaction products.
Owner:北京卓诚惠生生物科技股份有限公司
Immunotoxins and uses thereof
The invention provides novel recombinant immunotoxins comprising domain III of cholix toxin and exotoxin from Vibrio cholerae. The present invention further provides methods for using the compositions of the present invention to (i) induce apoptosis in a cell bearing one or more surface markers (ii) inhibit unwanted growth, hyperproliferation or survival of a cell bearing one or more cell surface markers, (iii) treat a condition, such as a cancer, (iv) provide therapy for a mammal having developed antibodies to Pseudomonas exotoxin A, and (v) provide therapy for a mammal having developed a disease caused by the presence of cells bearing one or more cell surface marker.
Owner:UNITED STATES OF AMERICA
Oligonucleotide probe kit for detecting common intestine trac kpathogenic bacteria and its use
InactiveCN1683565AQuick checkAccurate detectionMicrobiological testing/measurementAgainst vector-borne diseasesAntigenBio engineering
The present invention belongs to the field of microbe detecting technology. The oligonucleotide probe for detecting common intestinal tract pathogenic bacteria is designed on 16S rRNA and 23S rRNA of bacteria, ipaH of dysentery bacillus giant plasmid, VipR of Salmonella typhi and other gene sequence, has length of 25-50 bp, and relatively high sensitivity and specificity. The oligonucleotide probe is suitable for detection based on nucleic acid hybridization principle, especially detection based on gene chip principle. Under certain use condition, it can detect Listeria, parahemolutic vibrio, Campylobacter, etc. It may be used in many aspects, such as disease diagnosis, environment detection, food poisoning detection, etc.
Owner:RADIOLOGY INST ACAD OF MILITARY MEDICINE SCI PLA
Modified coconut oils with broad antimicrobial spectrum
InactiveUS20100016430A1Improve antibacterial propertiesAntibacterial agentsBiocideEscherichia coliCandida famata
Owner:MALAYSIAN AGRI RES & DEV INST MARDI
Cholera toxin virulence gene detection kit and detection method thereof
InactiveCN102094090AHigh sensitivityReduce testing costsMicrobiological testing/measurementPositive controlVibrio cholerae
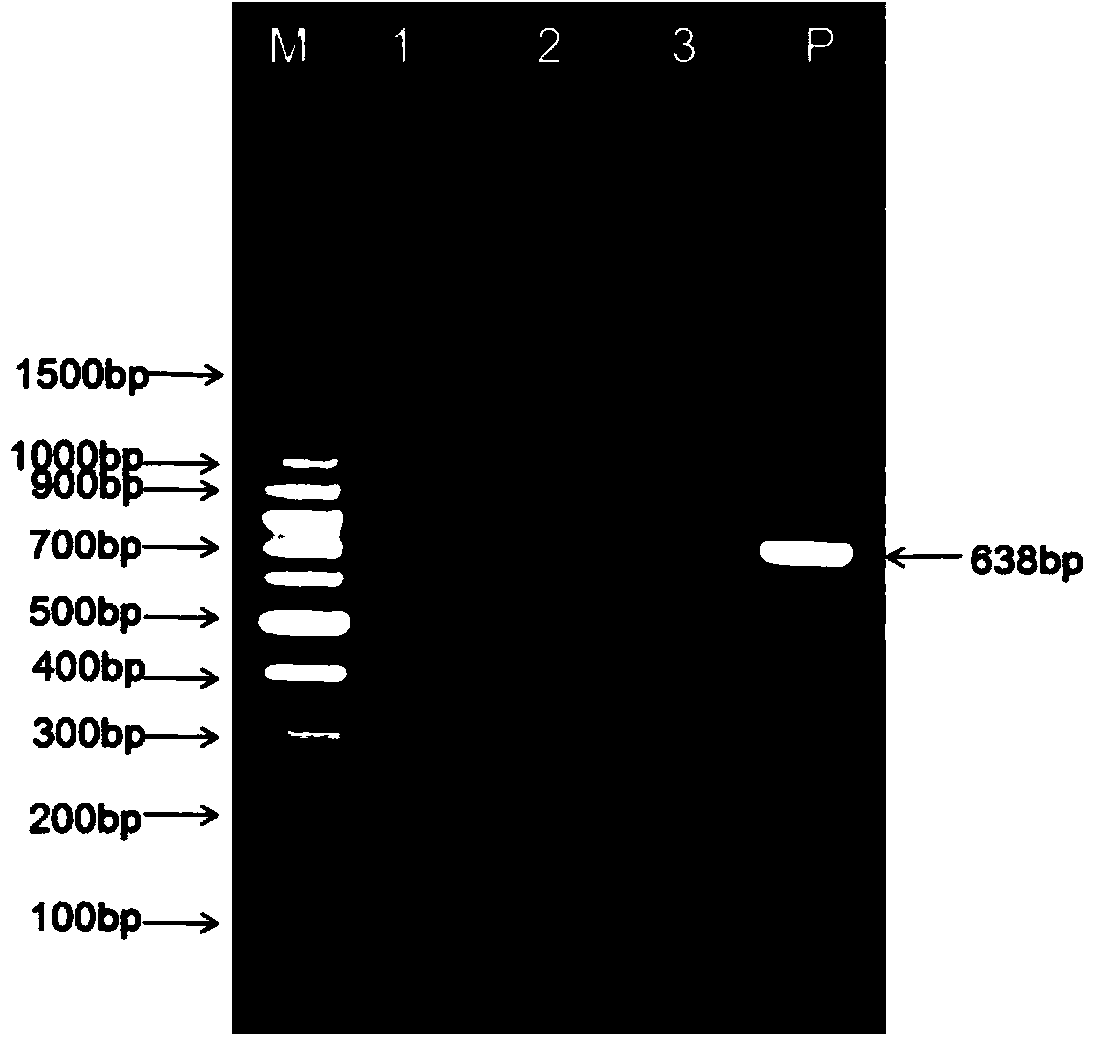
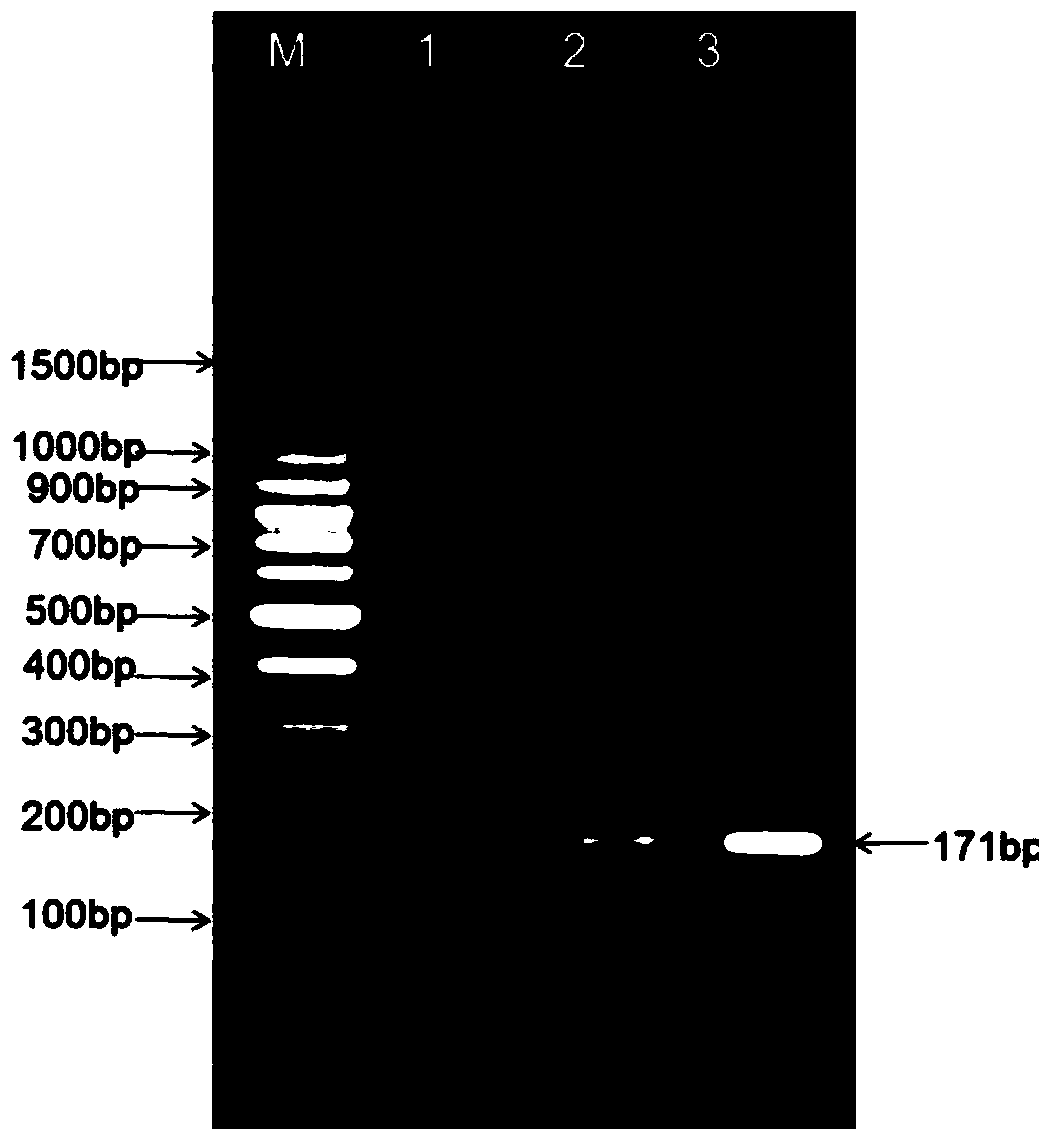
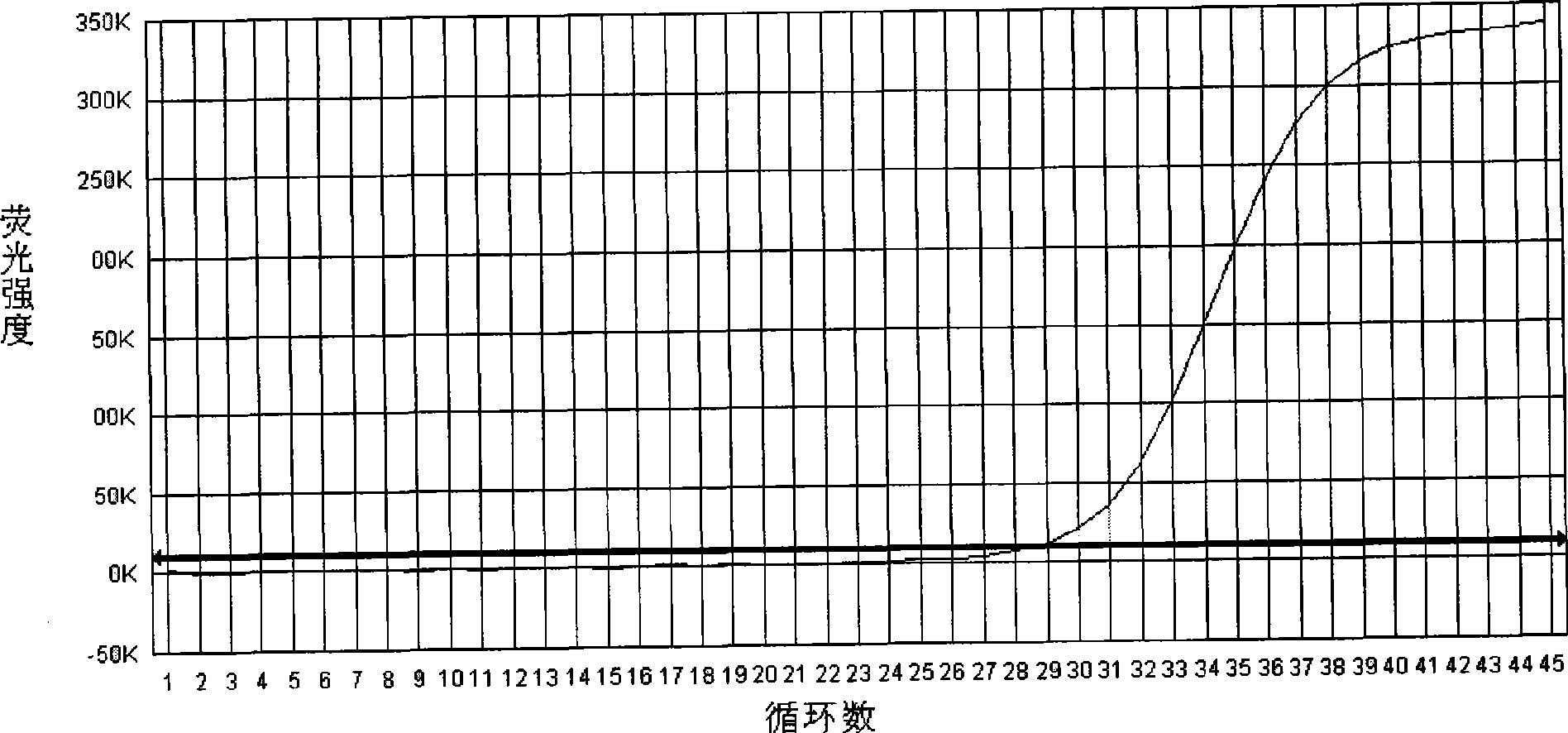
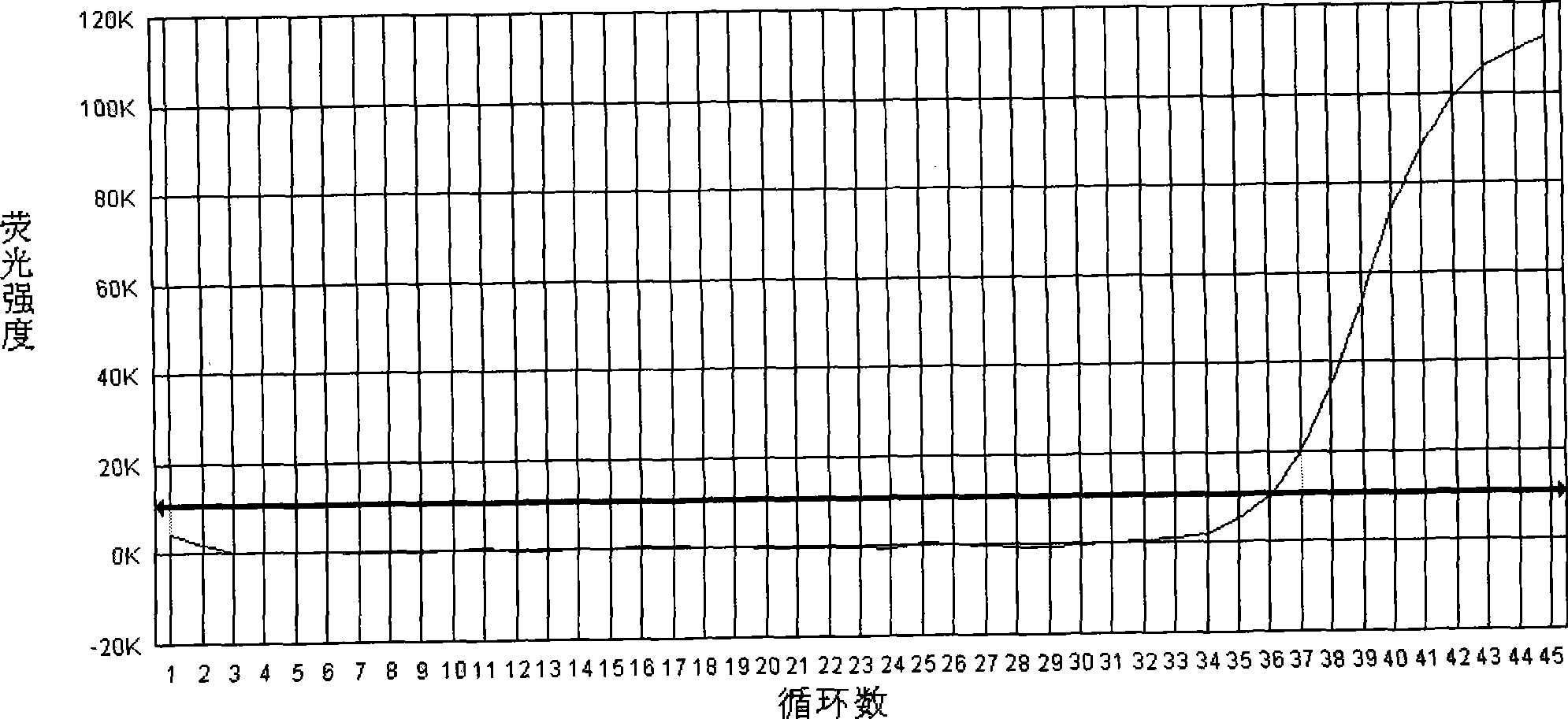
The invention relates to a cholera toxin virulence gene detection kit and a detection method thereof. The kit of the invention contains three pairs of primers which are designed by using vibrio cholera ctxA gene as target gene on the basis of the loop-mediated isothermal amplification technology, namely inner primers FIP / BIP, outer primers F3 / B3 and ring primers LF / LB and also contains Bst DNA polymerases, reaction solution, sample pretreatment solution, coloring liquid, stabilizing solution and positive control. The method for detecting the cholera toxin virulence gene comprises the following steps: extracting bacterial DNA, performing the loop-mediated isothermal amplification of the cholera toxin virulence gene and coloring for detection. The kit of the invention has high amplificationefficiency, specificity and sensitivity, low omission ratio and obvious coloring effect and is suitable for the rapid detection of toxigenic vibrio cholera.
Owner:EAST CHINA NORMAL UNIV +1
Food-originated pathogenic bactenium quick detection gene chip and its application
InactiveCN1536090AMicrobiological testing/measurementAgainst vector-borne diseasesFood poisoningBeta-hemolytic streptococcus
The present invention provides a gene chip for quickly detecting pathogens from food source, discloses the preparation method of said gene chip and provides 26 oligonucleotide probe sequences for detection. Said gene chip can quickly, accurately and high-effectively detect and identify the class of the pathogens in food, and its detection range includes staphylococcus aureus, Shiga's bacillus, salmonella, colibacillus 0157, bacillus proteus, mononuclear hyperplastic listerella, enterocolitis yersinia, aeruginous pseudomonads, vibrio parahaemolyticus, vibrio cholerae, bacillus cereus, beta hemolytic streptococcus, coconut fermentation pseudomonads, boticin, vibrio jejuni and bacillus perfringens, etc.
Owner:INST OF HYGIENE & ENVIRONMENTAL MEDICINE PLA ACAD OF MILITARY MEDICAL
Multiplex PCR based primer pair and kit for detecting multiple intestinal pathogens
ActiveCN104531898AQuick checkMultiple testing sites at one timeMicrobiological testing/measurementMicroorganism based processesEnteroinvasive E. coliEscherichia coli
The invention relates to a multiplex PCR based primer pair and kit for detecting intestinal pathogens, particularly relates to a multiplex PCR based primer pair and kit for detecting 14 intestinal pathogens, and belongs to the technical field of PCR application. The 14 intestinal pathogens comprise vibrio cholerae (group O1 phage, group O139 phage and group non-O1 / O139 phage), listeria monocytogenes, enteropathogenic escherichia coli (EPEC), enterohemorrhagic escherichia coli (EHEC), enterotoxigenic escherichia coli (ETEC), enteroinvasive escherichia coli (EIEC), enteroaggregative escherichia coli (EAEC), shigella, intestinal virus EV71, enterohemorrhagic escherichia coli O157:H7, clostridium difficile, vibrio parahaemolyticus, salmonella enteritidis and salmonella typhimurium. The multiplex PCR based primer pair and kit can change the situation that only a few intestinal pathogens can be detected in one time and can be used for detecting 14 intestinal pathogens and 26 genes simultaneously.
Owner:AGCU SCIENTECH
Gene chip of aquatic product cultivation pathogenic bacterium
InactiveCN101691608AReduce volumeRapid Test InterpretationNucleotide librariesMicrobiological testing/measurementBacteroidesPositive control
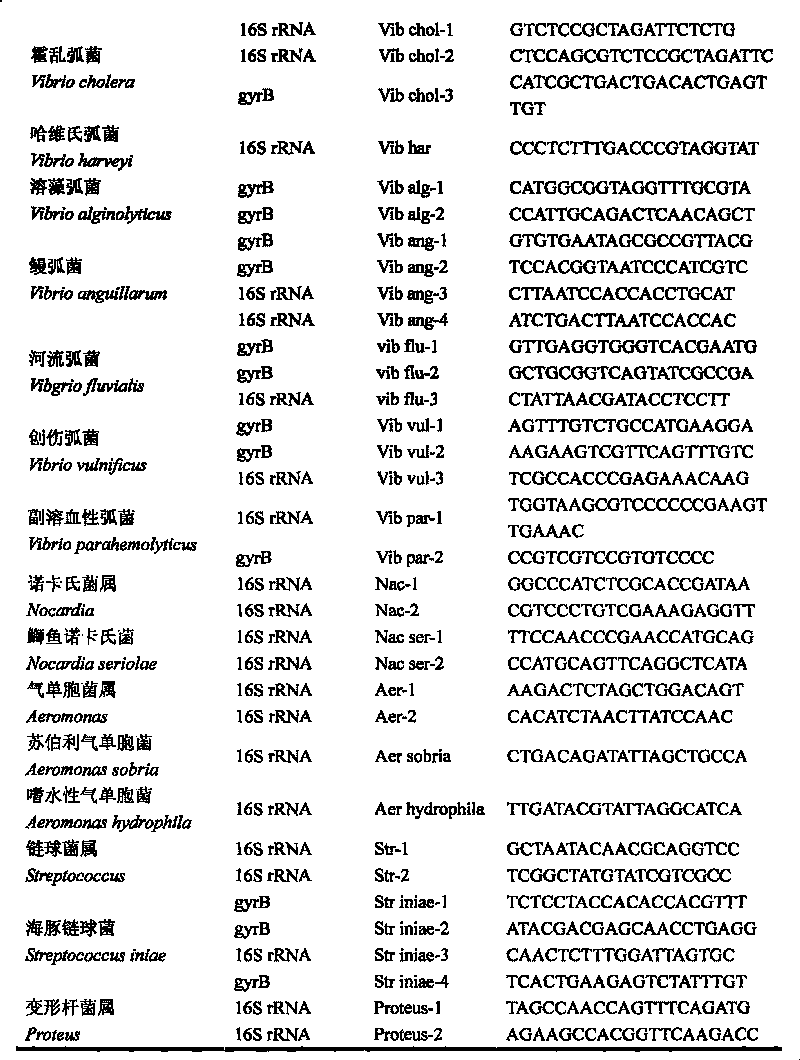
The invention discloses a gene chip of aquatic product cultivation pathogenic bacterium, comprising a solid phase carrier which is modified chemically, a detection probe and a quality control probe are distributed on the solid phase carrier in a dot matrix way; the detection probe comprises specificity 16S rDNA sequences and / or gyrB gene sequences of vibrio, comma bacillus, vibrio harveyi, vibrio alginolyicus, vibrio anguillarum, vibrio parahemolyticus, nocardia, nacardia seriolea, aeromonas, hydrophilic aeromonas, streptococcus and dolphin streptococcus, which are to be detected, the quality control probe includes PCR positive, chip fixed positive control, chip hybridizing negative control, chip hybridizing positive control and chip hybridizing blank control; the gene chip has the advantages of small volume and high flux, can detect known and unknown germs of the vibrio, the nocardia, the aeromonas and the streptococcus, and can detect specific germs with multiple kinds, and the simpleness and rapidness and specificity of the germs can be detected, and automatic detection can be carried out after detection software is additionally arranged.
Owner:NINGBO UNIV +2
Gene chip of main pathogenic microorganism in drinking water and testing kit
InactiveCN101748192ALow costImprove throughputMicrobiological testing/measurementAgainst vector-borne diseasesEscherichia coliBacteroides
The invention provides a gene chip of main pathogenic microorganism in drinking water and a testing kit, which mainly aims at 11 kinds of bacteria of colibacillus / Shigella, salmonella, vibrio cholera, vibrio parahaemolyticus, staphylococcus aureus, enterococcus faecails, pseudomonas aeruginosa, legionella pneumophilia, pneumobacillus, yersinia enterocolitica and the like, and L.interrogans. The gene chip comprises a solid phase carrier and a oligonucleotide probe fixed on the solid phase carrier, wherein the oligonucleotide probe contains gyrB gene with tremendous evolutionary advantage, ITS gene and DNA segment selected from 16srRNA gene or complementary DNA segment. The gene chip and the testing kit of the invention can test the main pathogenic microorganism in drinking water, and has the characteristics of simple operation, high throughput, high accuracy, strong repeatability and the like, and can be used for clinical test for the water quality monitoring department.
Owner:NANKAI UNIV
Culture medium for composite enrichment of salmonella, Vibrio parahaemolyticus and Vibrio cholerae, and preparation thereof
InactiveCN101412977AShorten enrichment timeBacteriaMicrobiological testing/measurementBacteroidesSulfite salt
The invention relates to a complex enrichment medium used for salmonella, vibrio parahaemolyticus and comma bacillus and a method for preparing the same. The formulation components of the complex enrichment medium in weight portion are as follows: 0.5 to 1.5 portions of buffer peptone water, 1000 portions of distilled water, 1.0 to 5.0 portions of glucose, 1.0 to 5.0 portions of mannitol, 0.03 to 0.06 portion sodium pyruvate, 0.5 to 2.5 portions of anhydrous sodium sulfite, 1.0 to 10.0 portions of sodium citrate, 5.0 to 25.0 portions of sodium chloride, 1 to 5 portions of cholate, and 0.0005 to 0.0025 portion of potassium tellurite. The method comprises the following steps: firstly, adding the buffer peptone water and the like into the distilled water, and sterilizing the mixture after heating and melting; and secondly, cooling the mixture to 50 DEG C, and then adding the potassium tellurite into the mixture to be mixed evenly. The complex enrichment medium can integrate two steps of primary enrichment and selective enrichment of bacterial detection to shorten the enrichment time to 24 hours, can perform enrichment on three target pathogens at the same time, and can inhibit the growth of other microorganisms.
Owner:SOUTH CHINA UNIV OF TECH
Primer, detection method and detection reagent kit for detecting cholera vibrio
InactiveCN101153332AStrong specificityIncreased sensitivityMicrobiological testing/measurementAgainst vector-borne diseasesFood borneVibrio cholerae
The invention relates a technique for fast detecting food-borne pathogens based on a loop-mediated isothermal amplification, LAMP technique. A primer for diction of comma bacillus can augment the specific base sequence of a target gene which is the ompW-GenBank (accession no. AF001009) of the comma bacillus, and the primer is complementary to a part of or a complementary chain fragment of the nucleic acid sequence on the 14-220 loci on the target gene. The invention provides a primer having specificity to a specific gene fragment of the comma bacillus, and through detecting whether or not the detecting specimen in a reagent box of the primer unit contains the specific gene fragment of the comma bacillus, determines whether the comma bacillus exists in the specimen or not.
Owner:ZHUHAI DISEASE PREVENTION & CONTROL CENT
LAMP (loop-mediated isothermal amplification) detection kit of vibrio cholerae and detection method thereof
InactiveCN103160606AHigh detection sensitivityShort reaction timeMicrobiological testing/measurementMicroorganism based processesVibrio choleraeMicrobiology
The invention discloses an LAMP (loop-mediated isothermal amplification) detection kit of vibrio cholerae and a detection method thereof and specifically relates to a group of primers for detecting the vibrio cholerae, which have oligonucleotide sequences shown in SEQ ID No. 1 to SEQ ID No. 6 in a sequence table, as well as the kit containing the primers and the detection method thereof. The kit disclosed by the invention is high in sensitivity, strong in specificity, low in cost and simple and convenient to operate.
Owner:BEIJING ENTRY EXIT INSPECTION & QUARANTINE BUREAU INSPECTION & QUARANTINE TECH CENT
Microbial preparation, aquatic feed and aquaculture method
InactiveCN109566919AIncrease food intakeIncrease growth rateFood processingClimate change adaptationVibrio parahaemolyticusPseudomonas
The invention discloses a microbial preparation, an aquatic feed and an aquaculture method and relates to the technical field of aquaculture. The microbial preparation comprises bdellovibrio and probiotic. Optimally, the probiotic is selected from any one of lactobacillus bulgaricus, lactobacillus delbrueckii, enterococcus faecalis, lactobacillus casei, lactobacillus acidophilus, rhodopseudomonaspalustris, clostridium butyricum, bacillus laterosporus, bacillus pumilus, bacillus coagulans, bacillus licheniformis, bacillus subtilis and lactobacillus plantarum. The microbial preparation has thecharacteristics of capability of increasing food intake of aquaculture animals, capability of promoting growth rate, survival rate and immunity of organisms, and the like. Besides, the microbial preparation is capable of effectively preventing and treating the problems of diseases caused by pathogenic bacteria, such as vibrio parahaemolyticus, vibrio harveyi, luminous vibrio, vibrio alginolyticus,vibrio campbellii, vibrio fluvialis, vibrio vulnificus, aeromonas, vibrio cholerae, pseudomonas and salmonella.
Owner:SHANGHAI LYUAO BIOTECH CO LTD
Short peptide for inhibiting Shiga toxin and application thereof
ActiveCN101016332AAvoid toxicityPlay a protective effectPeptide/protein ingredientsAntinoxious agentsDiseaseEscherichia coli
The invention discloses a making method of short peptide to inhibit shigellosis with amino acid as list 1, which is characterized by the following: adding chemical group, amino acid, peptide, protein, PEG, marked material to N and C ends to do kinds of chemical or biological decoration; optimizing the stability or other applying property and using scale; fitting for treat shigellosis, enterorrhagia escherichia coli O157 and cholera vibrio.
Owner:MICROBE EPIDEMIC DISEASE INST OF PLA MILITARY MEDICAL ACAD OF SCI
Immunogenic detoxified mutants of cholera toxin
InactiveUS20050136076A1Improved stability characteristicsImprove stabilityVirusesBacteriaEscherichia coliVibrio cholerae
Owner:NOVARTIS AG
Gene chip for detecting six kinds of diarrhea pathogens and its prepn process and kit
InactiveCN101045944AAccurate acquisitionNo pollution in the processMicrobiological testing/measurementAgainst vector-borne diseasesVibrio parahaemolyticusDiarrhea
The present invention relates to one kind of gene chip for detecting six kinds of diarrhea pathogens, and the gene chip includes solid carrier and oligonucleotide probe fixed on the carrier. The oligonucleotide probe includes DNA or cDNA segments selected from nucleotide sequences corresponding to the genomes of Shigella, haemorrhagic colibacillus, invasive colibacillus, Vibrio parahaemolyticus, Vibrio cholreae and salmonella. The said chip together with sample treating reagent, hybridizing reagent, color reagent and the specification constitutes the detection kit. The present invention has high detection efficiency and high detection accuracy.
Owner:IPE BIOTECHNOLOGY CO LTD
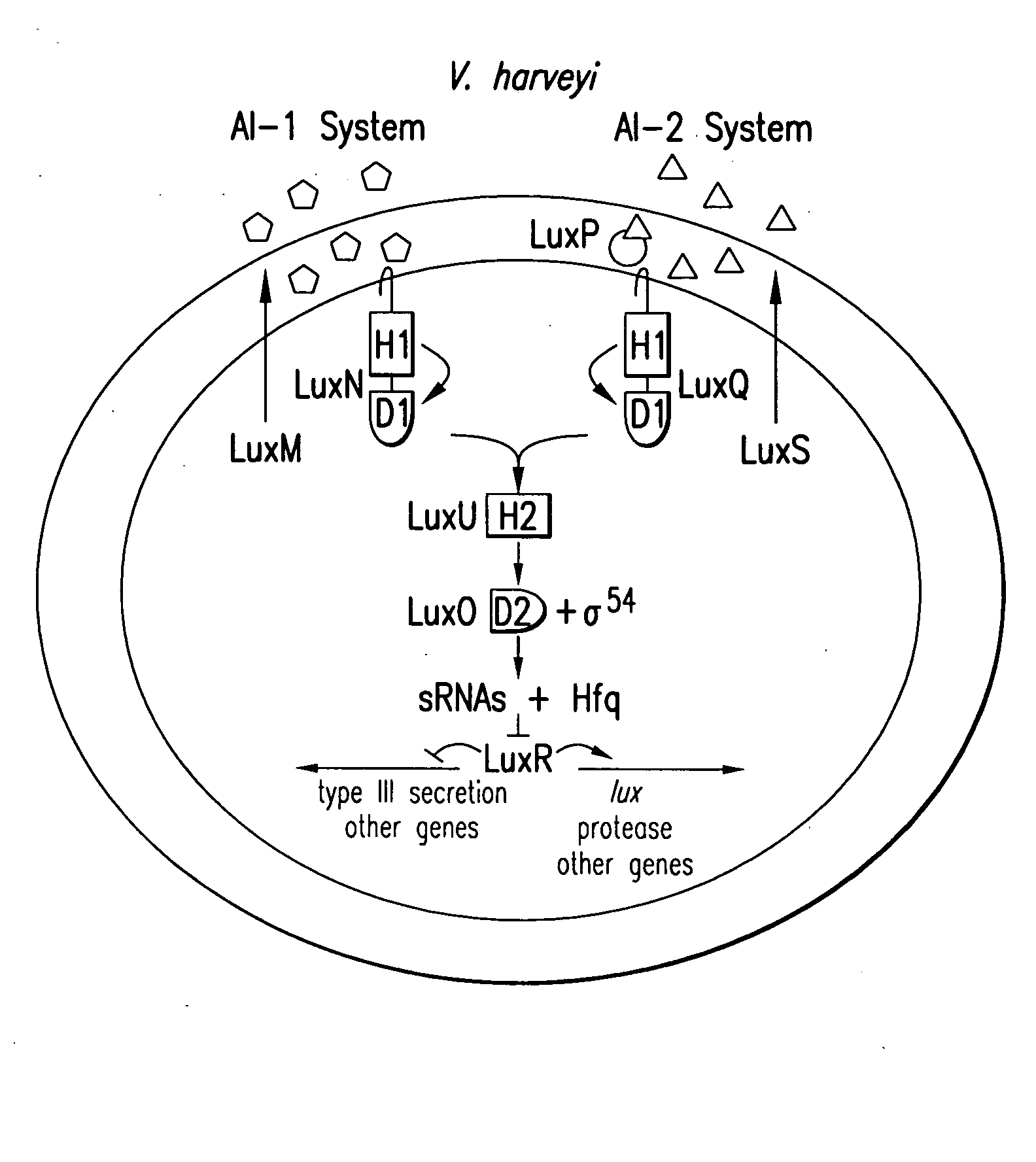
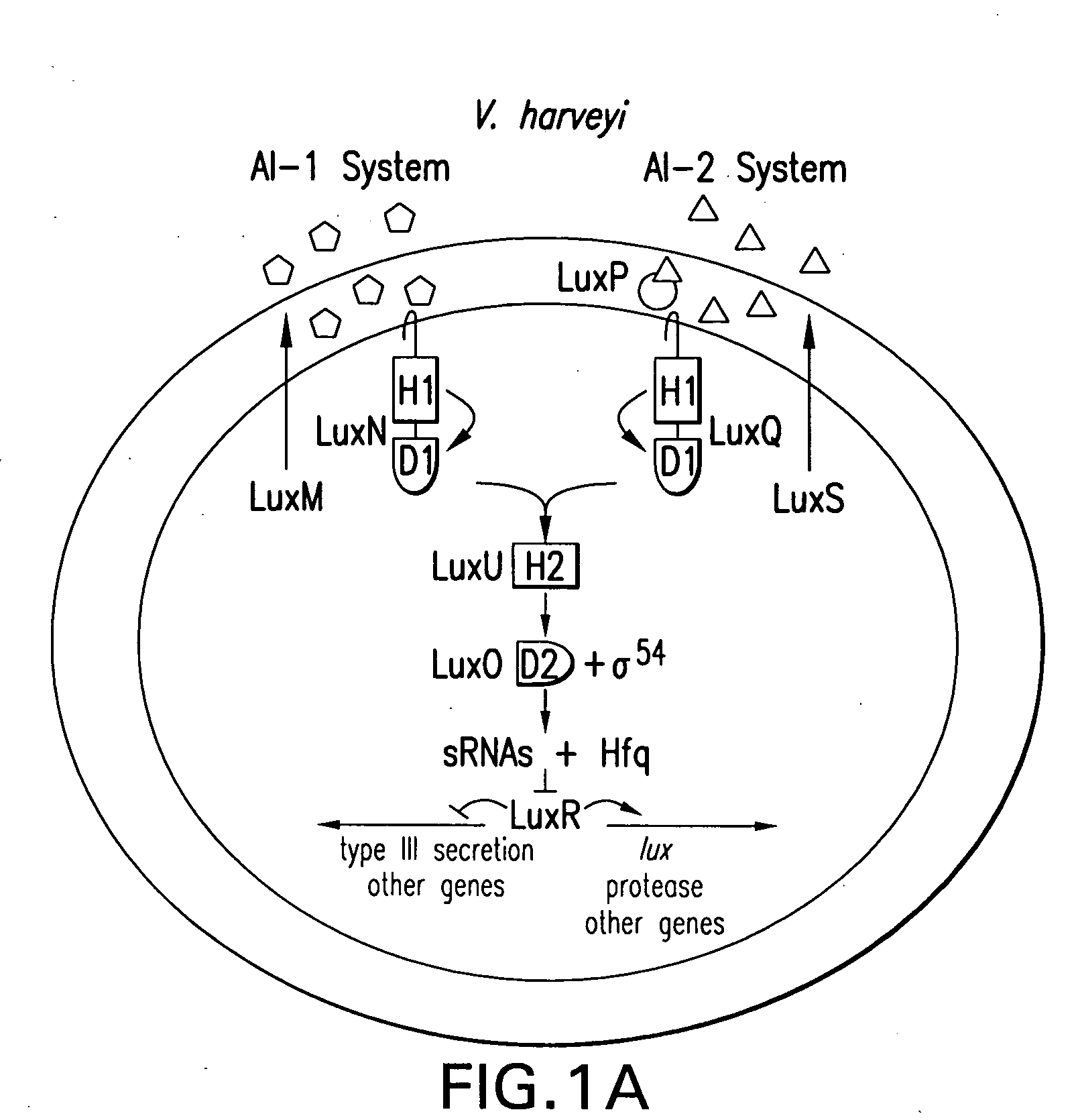
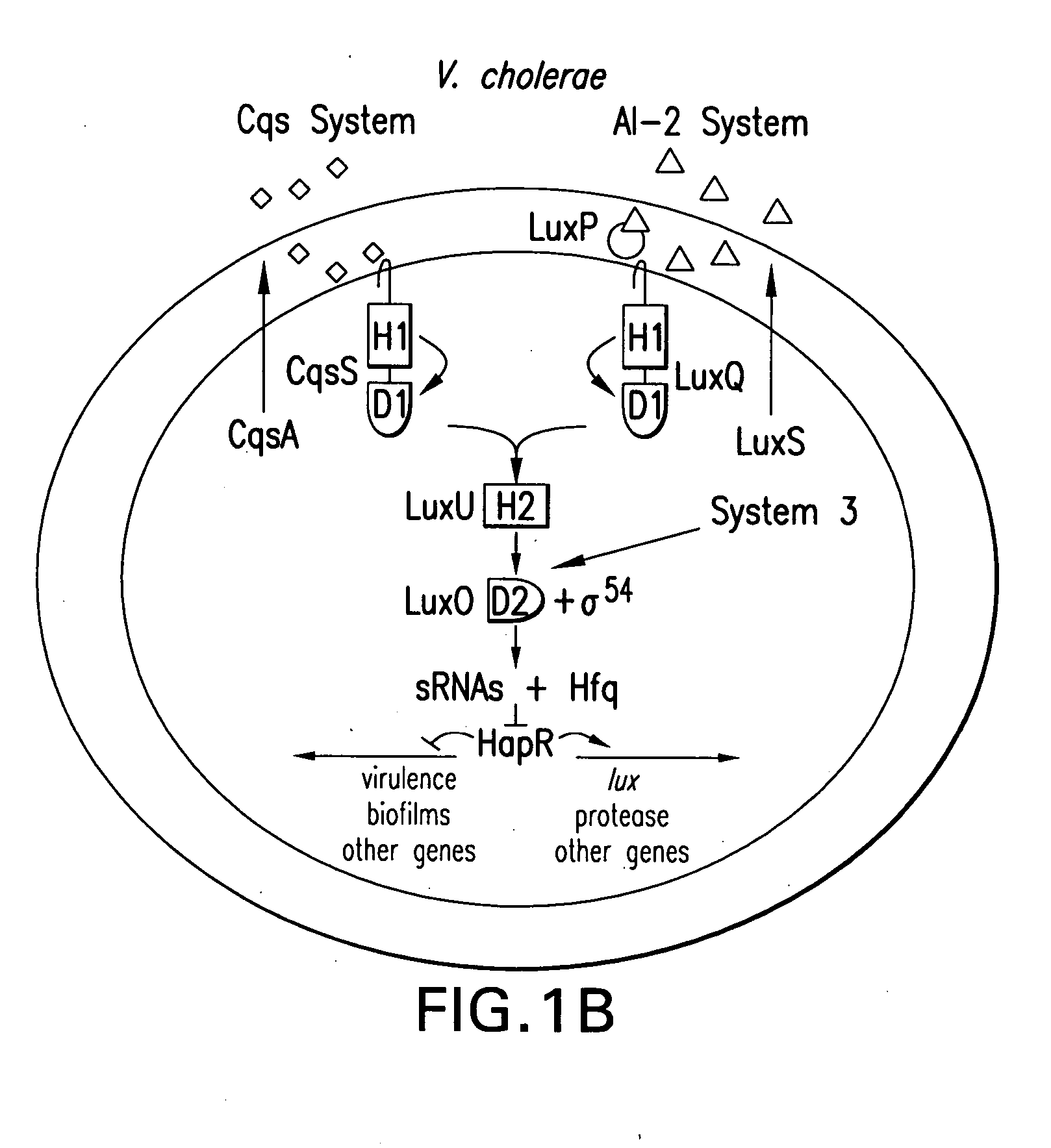
Small RNAs and bacterial strains involved in quorum sensing
Quorum-sensing bacteria communicate with extracellular signal molecules called autoinducers to allow community-wide synchronization of gene expression. The present invention relates to the identification the Vibrio harveyi and Vibrio cholerae protein Hfq as mediating interactions between small, regulatory RNAs (sRNAs) and specific messenger RNA (mRNA) targets. Accordingly, the present invention provides nucleic acids encoding the Vibrio sRNAs, strains having various deletions and mutations of one or more qrr genes encoding these sRNA as well as methods of identifying quorum-sensing regulators. Additionally, the invention relates to an isolated V. harveyi Hfq protein and conservative amino acid substitutions thereof as well as nucleic acids encoding those proteins, recombinant methods of producing those proteins and antibodies against those proteins.
Owner:THE TRUSTEES FOR PRINCETON UNIV
Detection kit for vibrio cholerae O1 group and detection method thereof
ActiveCN101824482AEffective expansionHigh yieldMicrobiological testing/measurementAgainst vector-borne diseasesPositive controlPolymerase L
The invention relates to a detection kit for a vibrio cholerae O1 group, which comprises BstDNA polymerase, reaction liquid, sample pretreatment liquid, coloration liquid, stabilizing solution and positive control, and also comprises two pairs of primers which take vibrio cholerae RfbN genes as target genes and two pairs of primers which are designed based on the loop-mediated isothermal amplification technology, namely inner primers FIP / BIP and outer primers F3 / B3. The detection kit for the vibrio cholerae O1 group has the advantages of comprehensive detection effect, high specificity and low omission factors, and is suitable for the quick detection of the vibrio cholerae O1 group.
Owner:GUANGZHOU HUAFENG BIOTECH +1
Vibrio cholerae typing and virulence gene detection kit and detection method
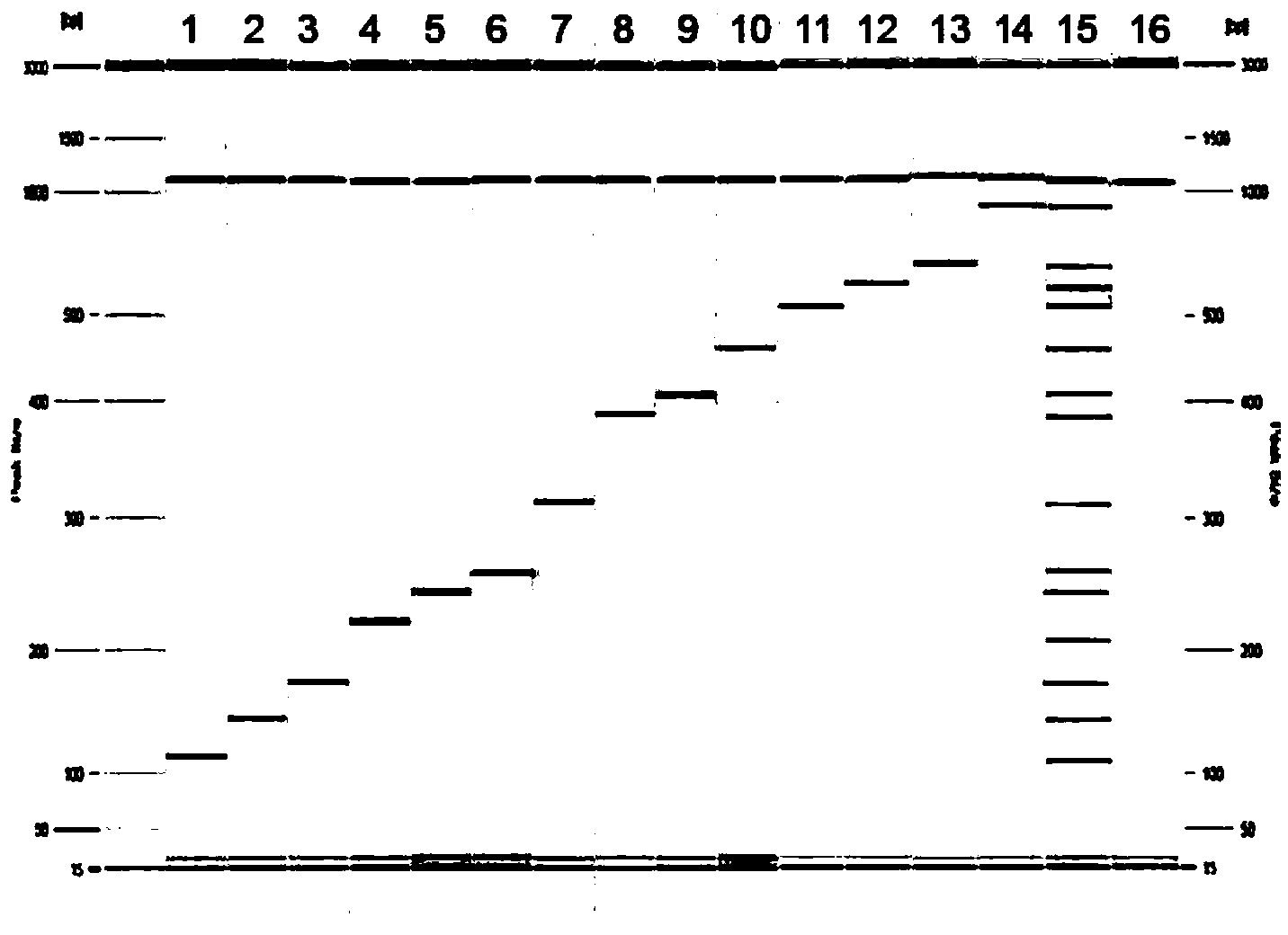
ActiveCN101967516ADetection helpsStrong specificityMicrobiological testing/measurementFluorescence/phosphorescenceVirulent characteristicsEnzyme system
The invention relates to a vibrio cholerae typing and virulence gene detection kit and a vibrio cholerae typing and virulence gene detection method. The kit comprises DNA extracting solution, PCR reaction solution, a Taq enzyme system, a positive quality control product and a negative quality control product, wherein the PCR reaction solution consists of PCR reaction buffer, four pairs of forward and reverse primers for the specificity of vibrio cholerae and four probes for the specificity of the vibrio cholerae; the primers and probes are designed according to the specificity conservative areas of nucleic acid sequences of a hemolysin gene, an O-antigen gene of O139 type vibrio cholerae, the O-antigen gene of O1 type vibrio cholerae and the virulence gene of cholera toxin (CTX). The detection kit and the detection method ensure a reliable and stable result, are easy and fast to operate and can realize the typing of the vibrio cholerae and fast judgment about whether the vibrio cholerae has a virulence gene or not, thereby facilitating the tracing and detection of the public health-related events caused by the vibrio cholerae and facilitating the routine monitoring of the vibrio cholerae.
Owner:DAAN GENE CO LTD +1
Composite gene chip for food-borne pathogenic bacteria detection
InactiveCN101397586AExplicitly excludeClear and distinctMicrobiological testing/measurementAgainst vector-borne diseasesEscherichia coliFood borne
The invention relates to a composite gene chip for detecting food pathogenic microbes, which comprises eight parallel sub-arrays constructed by detection probe groups for detecting each microbe in the eight food pathogenic microbes, blank lines are respectively arranged among the eight sub-arrays to be as intervals, thus constructing a big array; the eight food pathogenic microbes are listeria monocytogenes, comma bacillus, vibrio parahaemolyticus, salmonella, shigella, staphylococcus aureus, campylobacter jejuni and escherichia coli. The gene chip can clearly and accurately detect the species of eight target food pathogenic microbes, and also can definitely exclude other off-target microbe samples out of the eight target food pathogenic microbes. Each food pathogenic microbe detection sub-array on the chip can clearly and definitely distinguish off-target microbe samples from target microbe samples.
Owner:广东省疾病预防控制中心
Saccharide derivatives
Disclosed are novel saccharide derivatives which inhibit binding of toxins, such as heat-labile enterotoxin or cholera toxin, to their receptors either in vitro or in vivo. Additionally, disclosed are compounds which inhibit binding of enterovirulent organisms (e.g., bacteria, virus, fungi, and the like), such as Vibrio cholerae and enterotoxigenic strains of Escherichia coli, to their cell surface receptors.
Owner:SYNSORB BIOTECH INC
Oral composition and preparation method thereof
ActiveCN104688648AImprove antioxidant capacityHigh activityCosmetic preparationsToilet preparationsAdditive ingredientToothpaste
The invention provides an oral composition which is an oral preparation prepared by taking Periplaneta americana extract and Punicosides as active ingredients, wherein the weight ratio of the Periplaneta americana extract to Punicosides is 1:1-1:5. The invention also provides a preparation method and application of the oral composition. According to the oral composition disclosed by the invention, toothpaste can perform synchronous nursing on the oral cavity, teeth and gingiva and can effectively solve dental ulcer, the Punicosides have high in-vivo anti-oxidation and free radical cleaning activities, and the oral composition has a broad-spectrum antibacterial effect, has obvious effects of inhibiting staphylococcus aureus, hemolytic streptococcus, comma bacillus and shigella dysenteriae and has the effects of astringing, stopping bleeding, improving eyesight and the like.
Owner:耿福能
Multiplex fluorescent quantitative PCR detection method for main pathogenic bacteria in aquatic product
ActiveCN102121051ARapid specific detectionSensitive and specific detectionMicrobiological testing/measurementMicroorganism based processesSpecific detectionVibrio parahemolyticus
The invention discloses a multiplex fluorescent quantitative polymerase chain reaction (PCR) detection method for main pathogenic bacteria in an aquatic product. The method is characterized by comprising the following steps of: (1) performing enrichment culture on a sample to be detected in enrichment solution for 4 to 8 hours; (2) extracting genome DNA from the cultured sample by adopting a boiling method; (3) performing multiplex fluorescent quantitative PCR detection by using specific primers and TaqMan probes; and (4) performing result judgment according to the detected specific S amplification curve and threshold value (Ct value). Compared with the prior art, the method has the advantages that: one-tube multi-detection actual requirement can be met, and a solid technical guarantee can be provided for quick, sensitive and specific detection of vibrio parahaemolyticus, vibrio cholerae and listeria monocytogenes.
Owner:中华人民共和国舟山出入境检验检疫局
A kind of bacillus amyloliquefaciens wh3 and its preparation method and application
InactiveCN102286408AOral lowLow injection toxicityBacteriaMicroorganism based processesFreund adjuvantSclerotinia
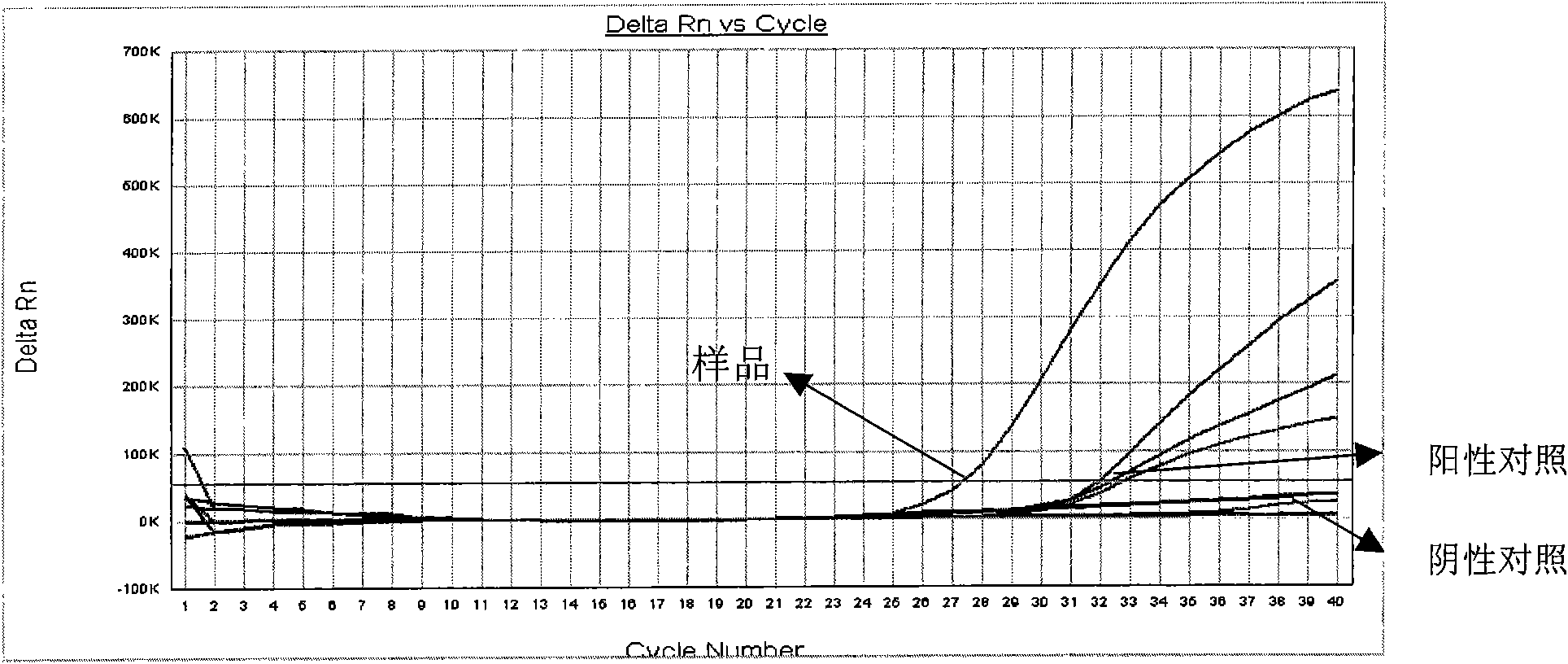
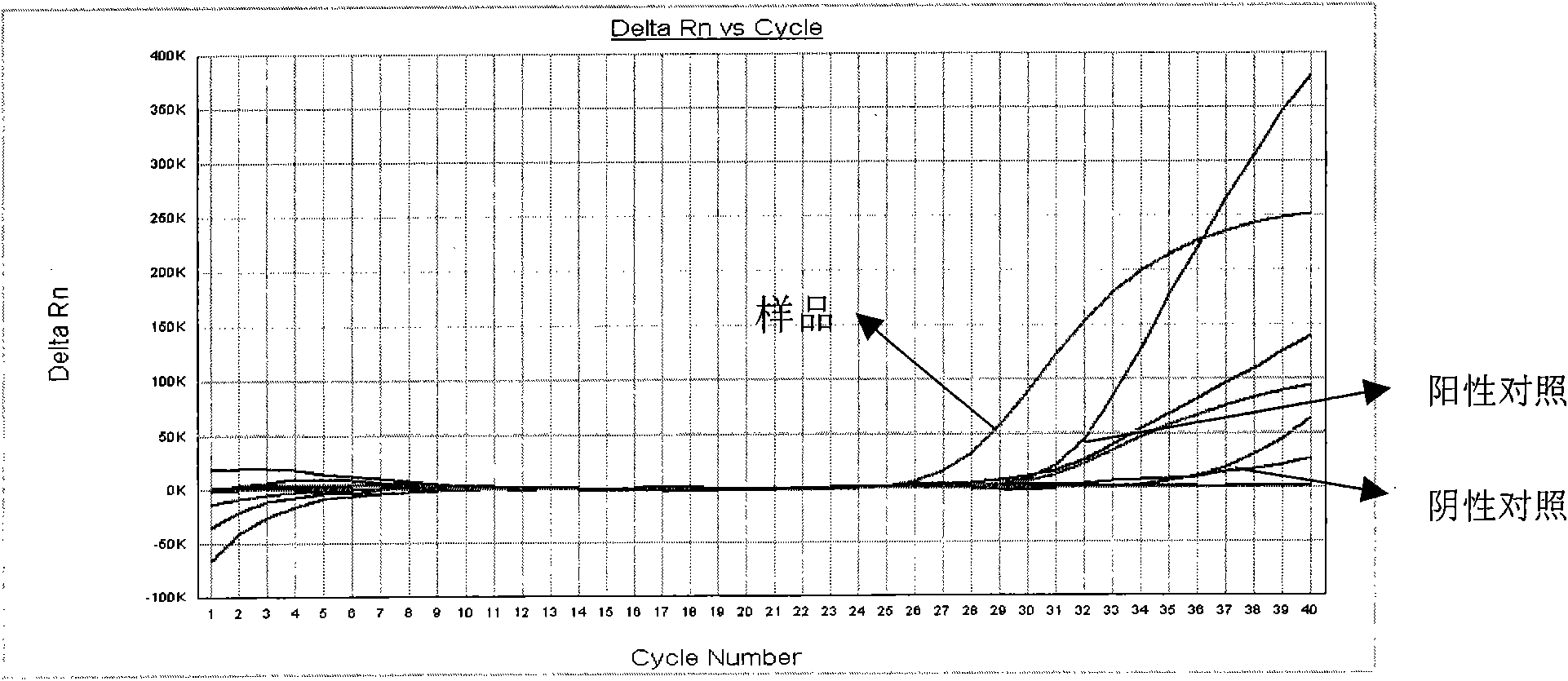
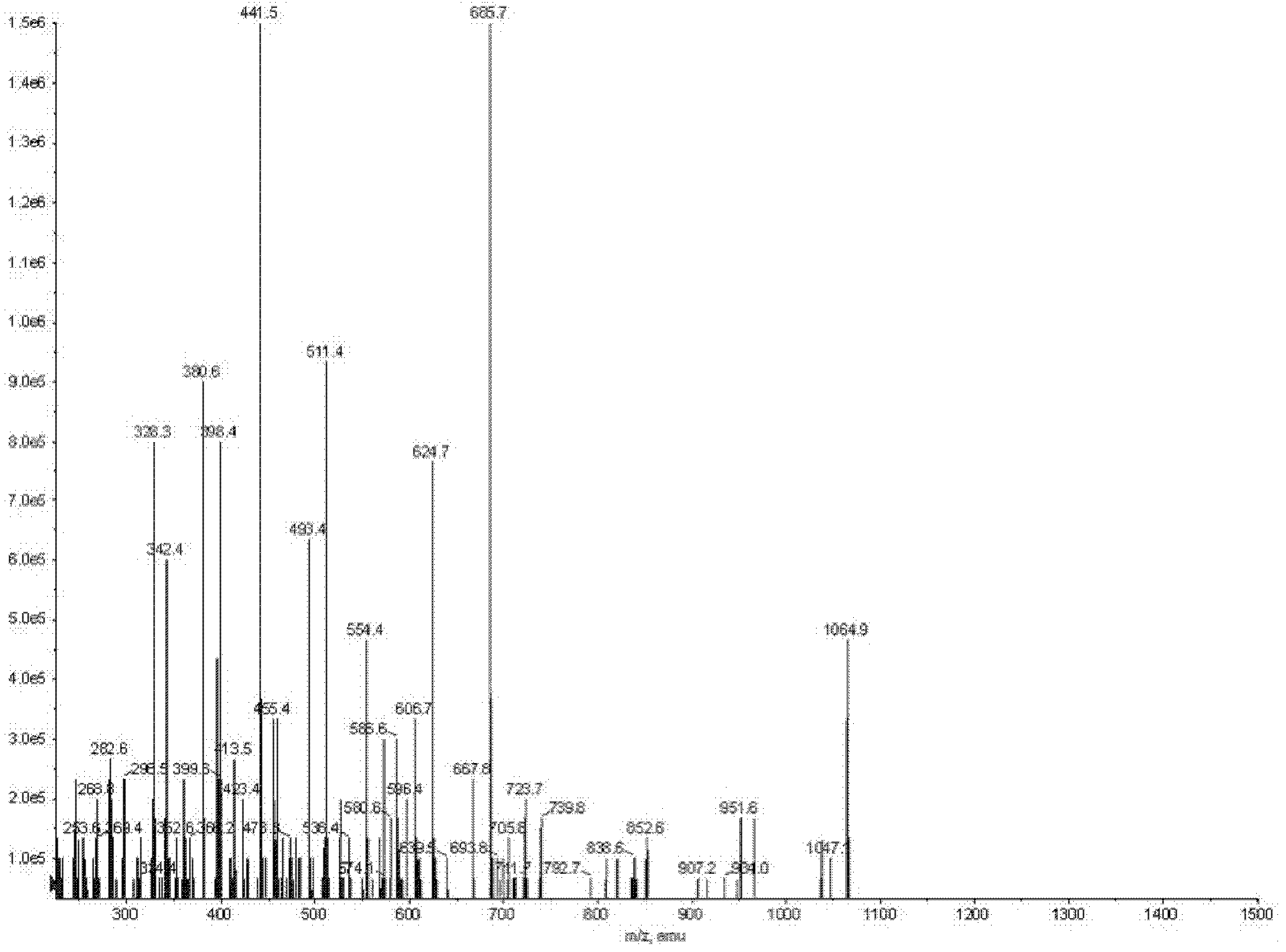
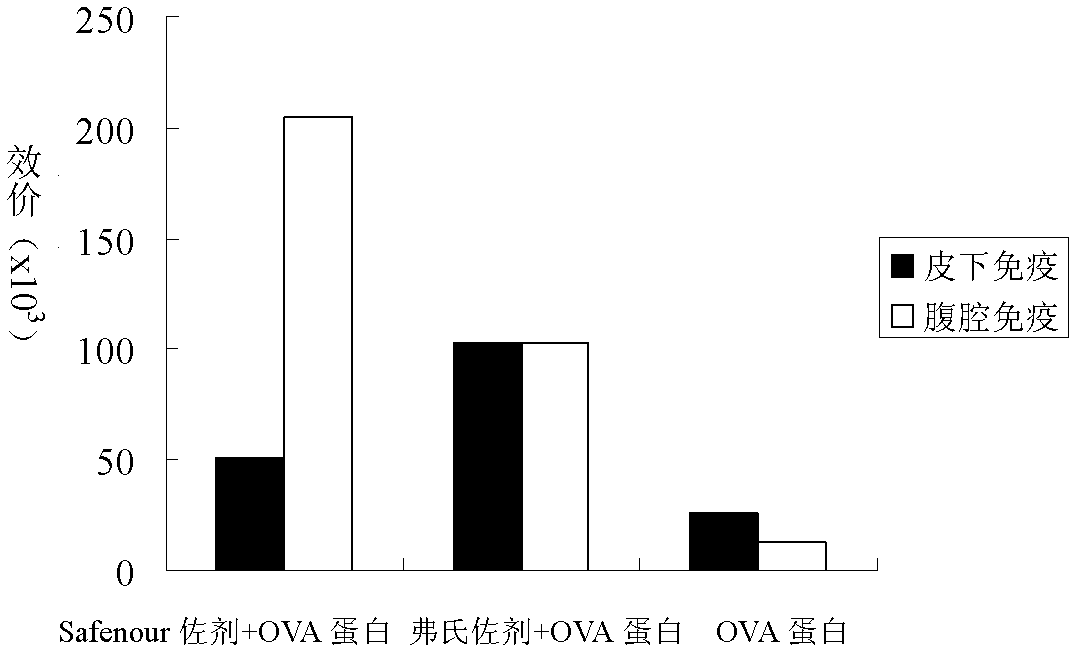
The invention discloses a Bacillus amyloliquefaciens WH3 strain, and a preparation method and application thereof. The preparation method comprises the following steps: 1, separation and identification of bacteria: separating bacteria resistant to rape sclerotinia rot from rape seedlings, and carrying out 16SrDNA and morphological identification to determine that the WH3 strain obtained by separation is Bacillus amyloliquefaciens; 2, separation, purification and identification of an antifungal active substance Safenour: fermenting the WH3 strain, extracting the antifungal active substance, separating and purifying through a sephadex column, and carrying out MALDI-TOF (matrix-assisted laser desorption / ionization-time of flight ) mass spectrometry on the active antifungal substance to inferthat the substance is a ring type polypeptide; and 3, application of the strain in the preparation of vaccines and immunological adjuvants. After the Safenour used as the adjuvant is mixed with a protein antigen and mice are respectively immunized through oral administration and injection of the mixture, effective body fluid and cellular immune response can be activated, and a high-titer specificantibody can be detected in the blood serum. The Safenour has low production cost and high stability, does not need to be emulsified when mixed with the antigen, and has a better immunoenhancement effect in comparison with Freund adjuvants and cholera toxin B subunits.
Owner:武汉光谷世傲生物科技有限公司
Pharmaceutical proteins, human therapeutics, human serum albumin insulin, native cholera toxic B submitted on transgenic plastids
InactiveUS20060117412A1Eliminate needLarge biomassSerum albuminDepsipeptidesInsulin-like growth factorEscherichia coli
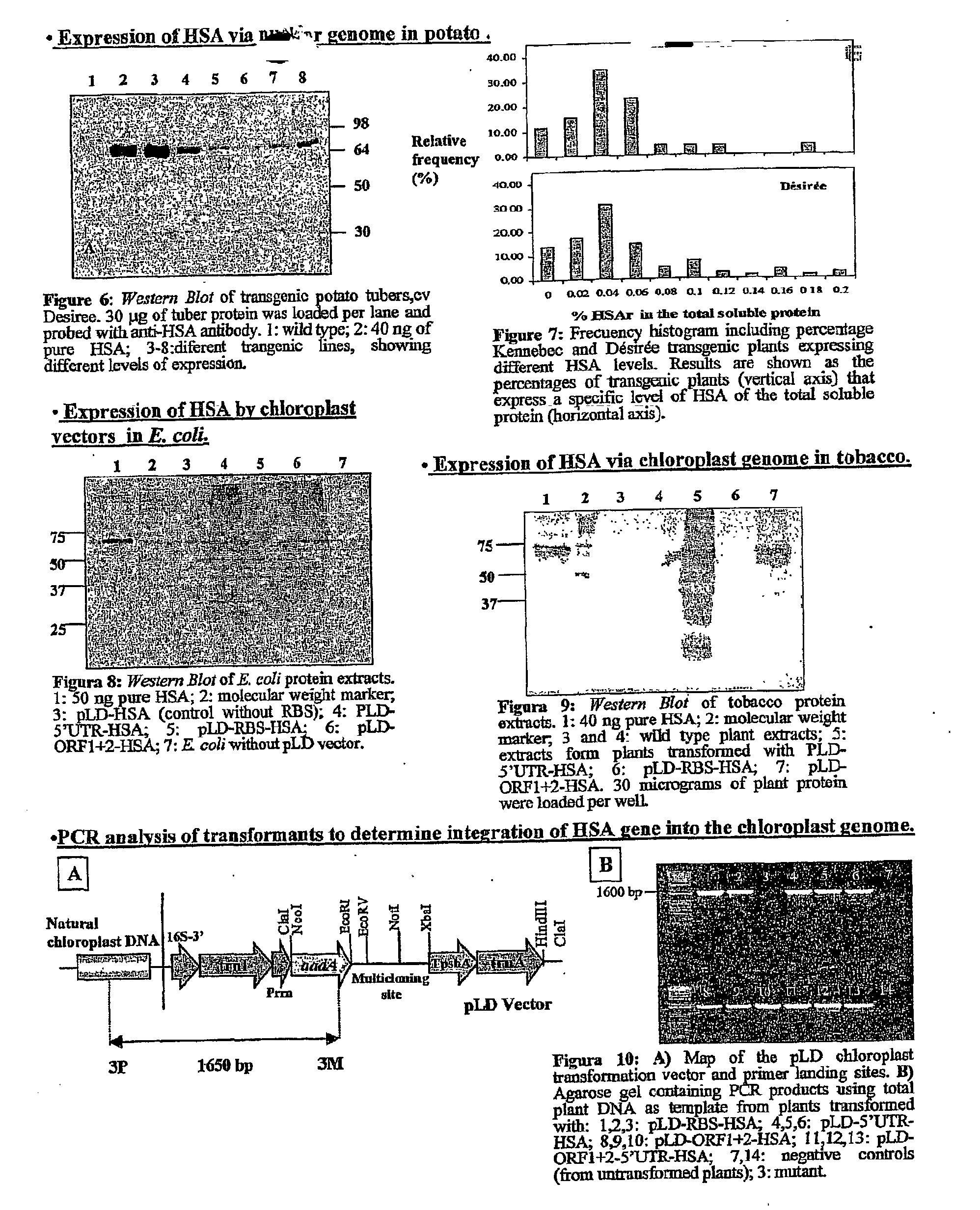
Transgenic chloroplast technology could provide a viable solution to the production of Insulin-like Growth Factor I (IGF-I), Human Serum Albumin (HSA), or interferons (IFN) because of hyper-expression capabilities, ability to fold and process eukaryotic proteins with disulfide bridges (thereby eliminating the need for expensive post-purification processing). Tobacco is an ideal choice because of its large biomass, ease of scale-up (million seeds per plant), genetic manipulation and impending need to explore alternate uses for this hazardous crop. Therefore, all three human proteins will be expressed as follows: a) Develop recombinant DNA vectors for enhanced expression via tobacco chloroplast genomes b) generate transgenic plants c) characterize transgenic expression of proteins or fusion proteins using molecular and biochemical methods d) large scale purification of therapeutic proteins from transgenic tobacco and comparison of current purification / processing methods in E. coli or yeast e) Characterization and comparison of therapeutic proteins (yield, purity, functionality) produced in yeast or E. coli with transgenic tobacco f) animal testing and pre-clinical trials for effectiveness of the therapeutic proteins. Mass production of affordable vaccines can be achieved by genetically engineering plants to produce recombinant proteins that are candidate vaccine antigens. The B subunits of Enteroxigenic E. coli (LTB) and cholera toxin of Vibrio cholerae (CTB) are examples of such antigens. When the native LTB gene was expressed via the tobacco nuclear genome, LTB accumulated at levels less than 0.01% of the total soluble leaf protein. Production of effective levels of LTB in plants, required extensive codon modification. Amplification of an unmodified CTB coding sequence in chloroplasts, up to 10,000 copies per cell, resulted in the accumulation of up to 4.1% of total soluble tobacco leaf protein as oligomers (about 410 fold higher expression levels than that of the unmodified LTB gene). PCR and Southern blot analyses confirmed stable integration of the CTB gene into the chloroplast genome. Western blot analysis showed that chloroplast synthesized CTB assembled into oligomers and was antigenically identical to purified native CTB. Also, GM1-ganglioside binding assays confirmed that chloroplast synthesized CTB binds to the intestinal membrane receptor of cholera toxin, indicating correct folding and disulfide bond formation within the chloroplast. In contrast to stunted nuclear transgenic plants, chloroplast transgenic plants were morphologically indistinguishable from untransformed plants, when CTB was constitutively expressed. The introduced gene was stably inherited in the subsequent generation as confirmed by PCR and Southern blot analyses. Incrased production of an efficient transmucosal carrier molecule and delivery system, like CTB, in transgenic chloroplasts makes plant based oral vaccines and fusion proteins with CTB needing oral administration a much more practical approach.
Owner:DANIELL HENRY
Gene chip for detecting important pathogenic bacteria in aquatic product and kit thereof
InactiveCN102311993AImprove accuracyGood repeatabilityMicrobiological testing/measurementAgainst vector-borne diseasesBacillus cereusStaphylococcus aureus
The invention relates to a gene chip for detecting important pathogenic bacteria in an aquatic product and a kit thereof. The gene chip comprises a solid phase carrier and an oligonucleotide probe; the oligonucleotide probe comprises one or more of the following nucleotide sequences: (1) DNA (Deoxyribonucleic Acid) sequences selected from proteus mirabilis 1, proteus vulgaris, salmonella, vibrio parahaemolyticus, vibrio cholerae, Listeria monocytogenes, staphylococcus aureus, streptococcus pyogenes, vibrio vulnificus, bacillus cereus cluster, Shigella and pathogenic Y. enterocolitica ail gene; (2) complementary DNA sequences of the DNA sequences selected in the (1); (3) complementary RNA (Ribonucleic Acid) sequences of the DNA sequences in the (1) or (2). The kit comprises the gene chip. The gene chip and the kit can be used for detecting the important pathogenic bacteria in an aquatic product, are convenient to operate, high in precision and strong in repeatability.
Owner:TIANJIN BIOCHIP TECH CO LTD
Bacillus pumilus and application thereof in prevention and control of comma bacillus in aquatic product
InactiveCN102994417AImprove water qualityAvoid harmBacteriaMicroorganism based processesAquaculture industryBacillus pumilus
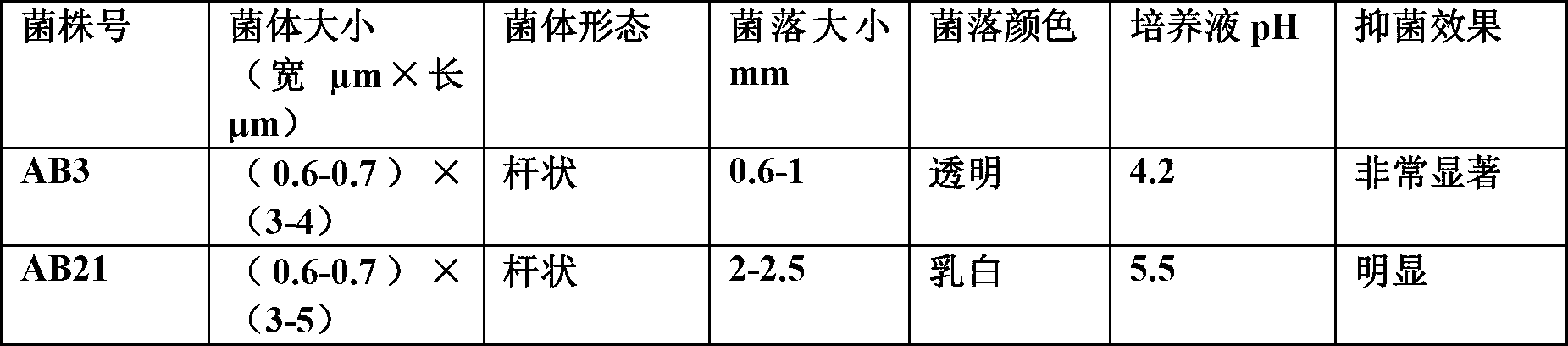
The invention discloses bacillus pumilus AB3. The bacterial strain has a preservation name called bacillus pumilus AB3, of which the preservation number is CGMCC NO.6408, and has the capability of inhibiting the growth or the reproduction of comma bacillus. The bacteria which serve as the comma bacillus antagonistic bacteria can be prepared to antibacterial agents, and the antibacterial agents can be applied to aquaculture, can be used as beneficial microorganism preparations replacing chemical sanitizers, are used for improving the water quality of fishery industry and removing the comma bacillus in an aquatic product cultivation system so as to prevent the damage of the comma bacillus to aquaculture objects and promote the development of the aquaculture industry, and can avoid carrying the comma bacillus in the aquatic products to guarantee the human health safety.
Owner:苏州市水生动物疫病预防控制中心