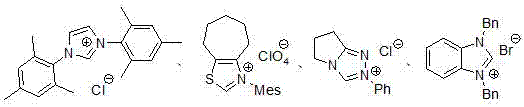Patents
Literature
31results about How to "Harsh reaction conditions" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
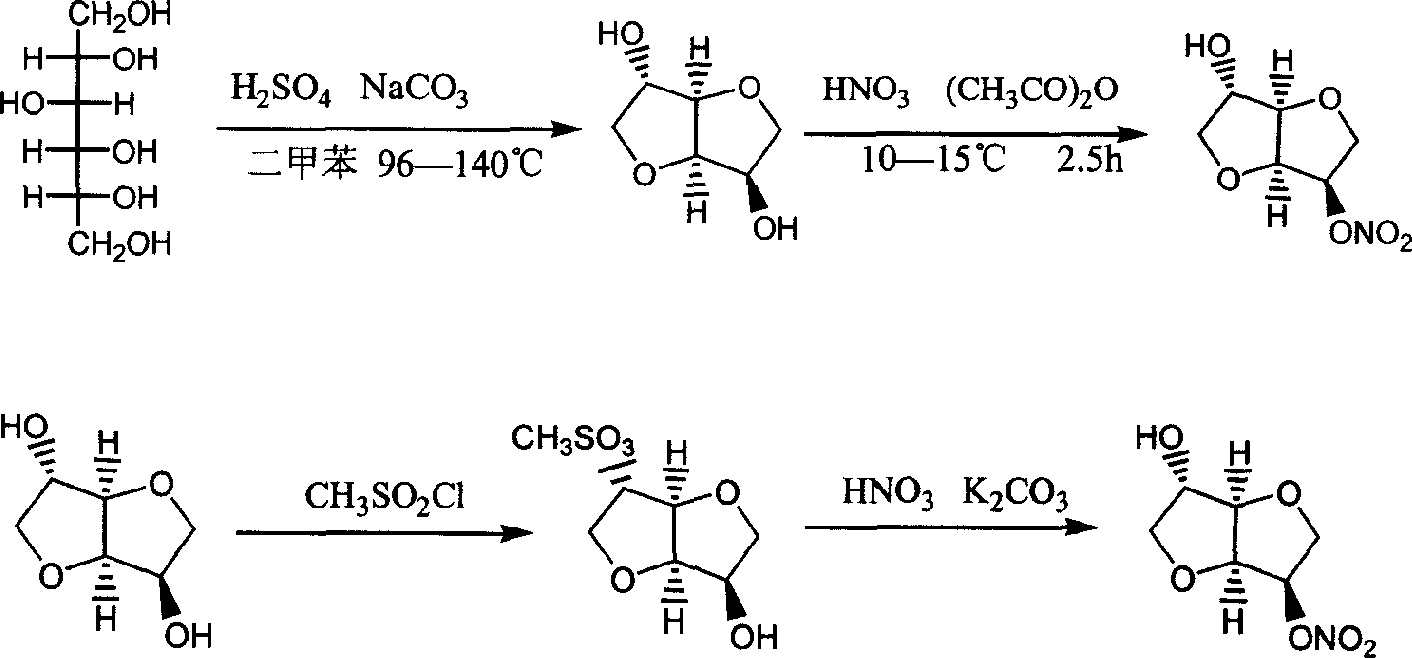
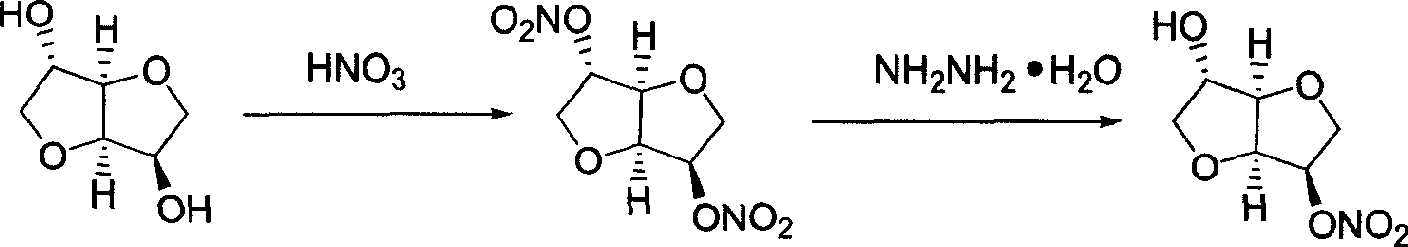
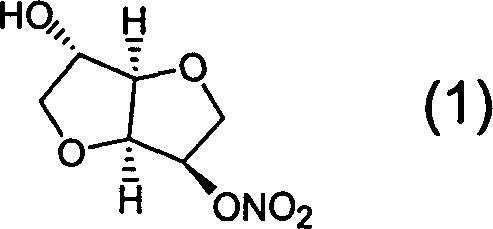
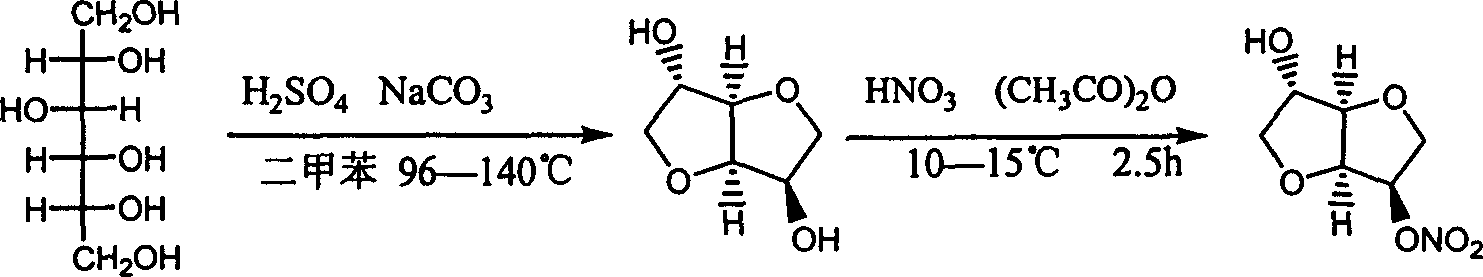
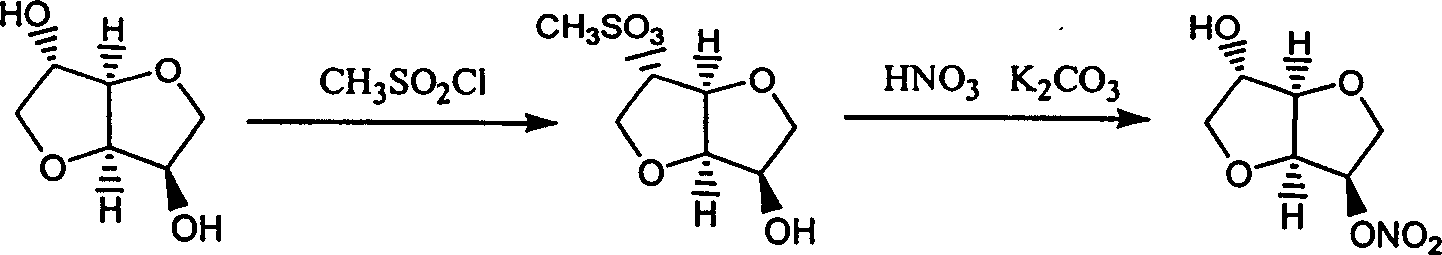
Prepn process of isosorbide mononitrate
ActiveCN1609108ALow boiling pointReduce the temperatureOrganic chemistryCardiovascular disorderRutheniumIsosorbide mononitrate
The preparation process of isosorbide mononitrate as angina pectoris resisting medicine includes the following steps: directly nitrating sorbitol with fuming nitric acid and concentrated sulfuric acid in 0-0.5 time to obtain 2, 5-dinitro isosorbate; and the subsequent catalytic hydrogenation with ruthenium complex as catalyst and selective reduction of 2-nitro group. The present invention has simple process and high yield.
Owner:SHANDONG NEWTIME PHARMA
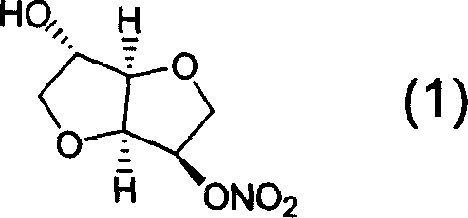
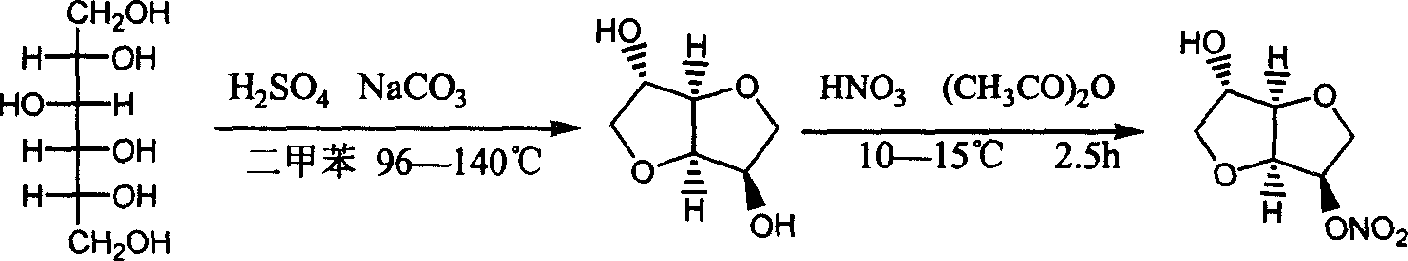
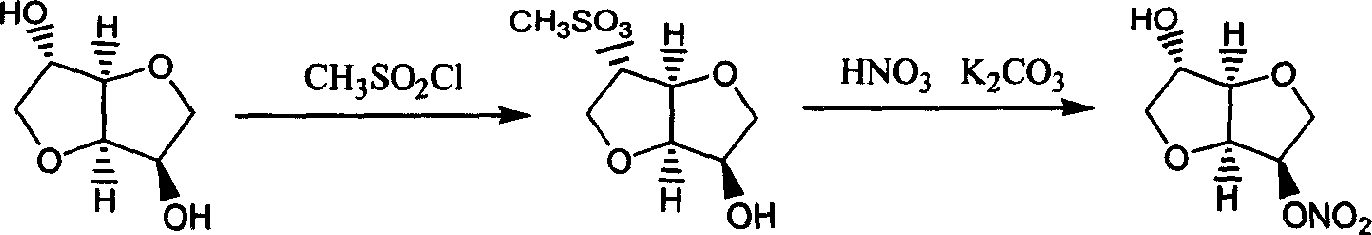
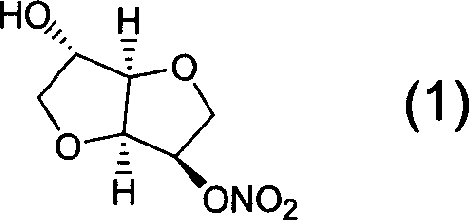
Preparation process of isosorbide mononitrate
InactiveCN1618798AHarsh reaction conditionsLow yieldOrganic chemistryCardiovascular disorderAnginaAcetic oxide
A process for preparing the isosorbide mononitrate from sorbitol includes dewatering sorbitol by p-methylphenyl sulfonic acid to obtain anhydrosorbitol, protecting by acetic oxide under existance of N,N-dimethylaminopyridine, nitrating by nitric acid / acetic oxide / acetic acid system, and removing protection by potassium carbonate-methanol system. It can be used to treat angina pectoris.
Owner:LUNAN PHARMA GROUP CORPORATION
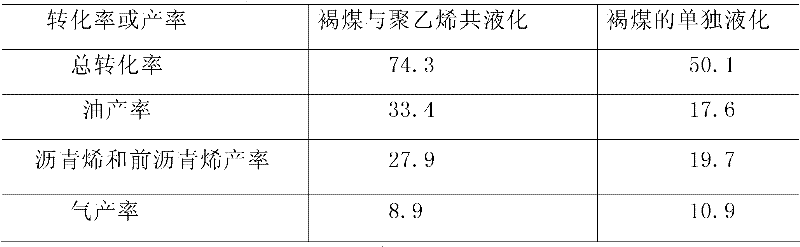
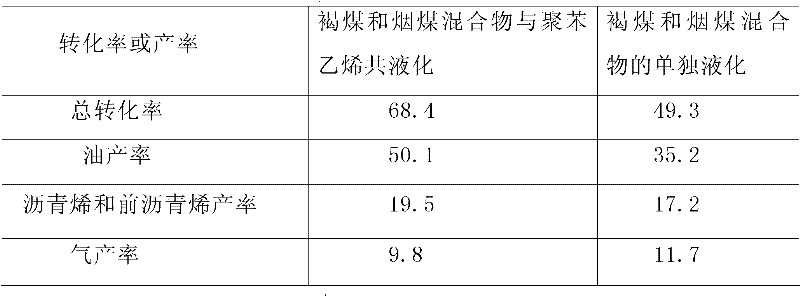
Method for co-liquefaction of coal and waste plastics under mild condition
InactiveCN102344823AHigh yieldIncrease profitLiquid hydrocarbon mixture productionDepolymerizationGas phase
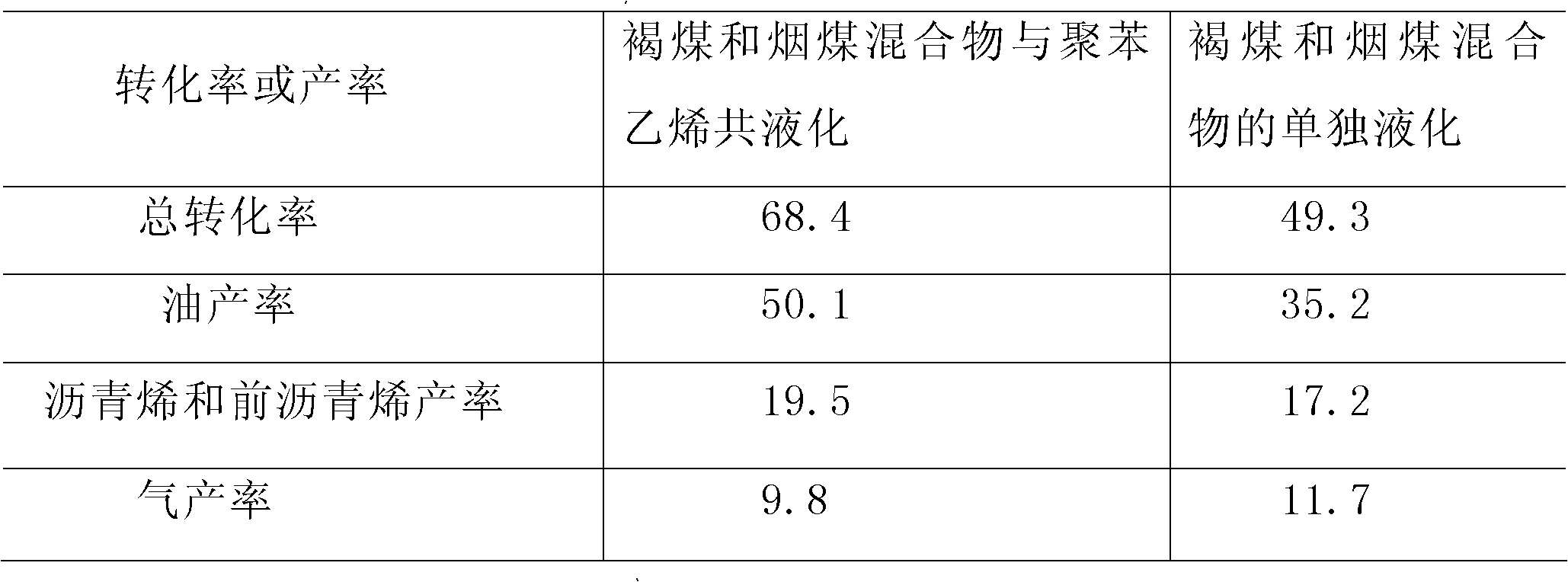
The invention is a method for co-liquefaction of coal and waste plastics under a mild condition, which comprises the following steps: adding coal, waste plastics, lignosulfonate, a catalyst, and a solvent into an autoclave, introducing hydrogen to increase the pressure to 1.0-10.0 Mpa, heating to 300-450 DEG C for a liquefaction reaction, collecting the gas after the hydrogenation liquefaction test in the autoclave, analyzing the gas phase composition by a gas chromatography, collecting all the other liquefaction products in a filter paper cylinder, performing hexane and tetrahydrofuran solvent extraction in order by a soxhlet extraction device, wherein the substance soluble in hexane is oil, and the substances soluble in tetrahydrofuran but insoluble in hexane are asphaltene and preasphaltene, and calculating the total conversion rate of the liquefaction by using the amount of the insoluble substances in tetrahydrofuran. The invention allows the coal to perform a depolymerization reaction at a low temperature, and to be co-liquefied with waste plastics; the extraction rate of coal by solvents such as pyridine and the like is increased, and the coal liquefaction process is accelerated; not only the hydrogen consumption and production cost for coal liquefaction are greatly reduced, but also white pollution caused by waste plastics is solved.
Owner:LIUPANSHUI NORMAL UNIV
Preparation method of draxxin
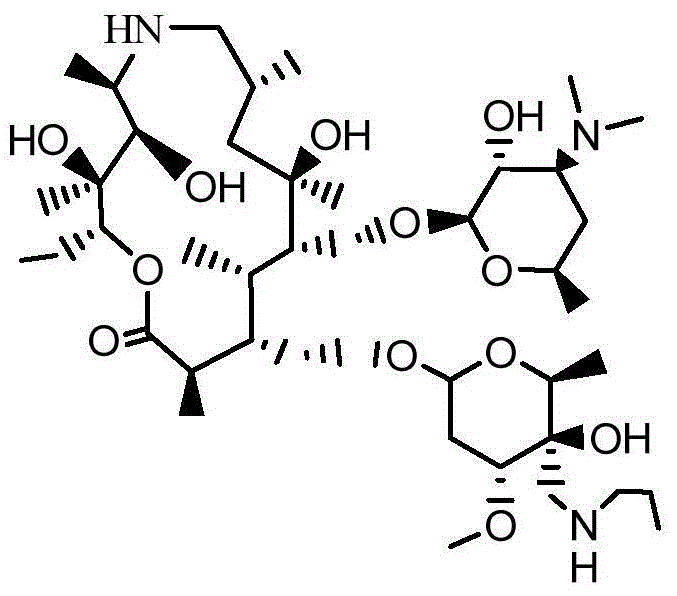
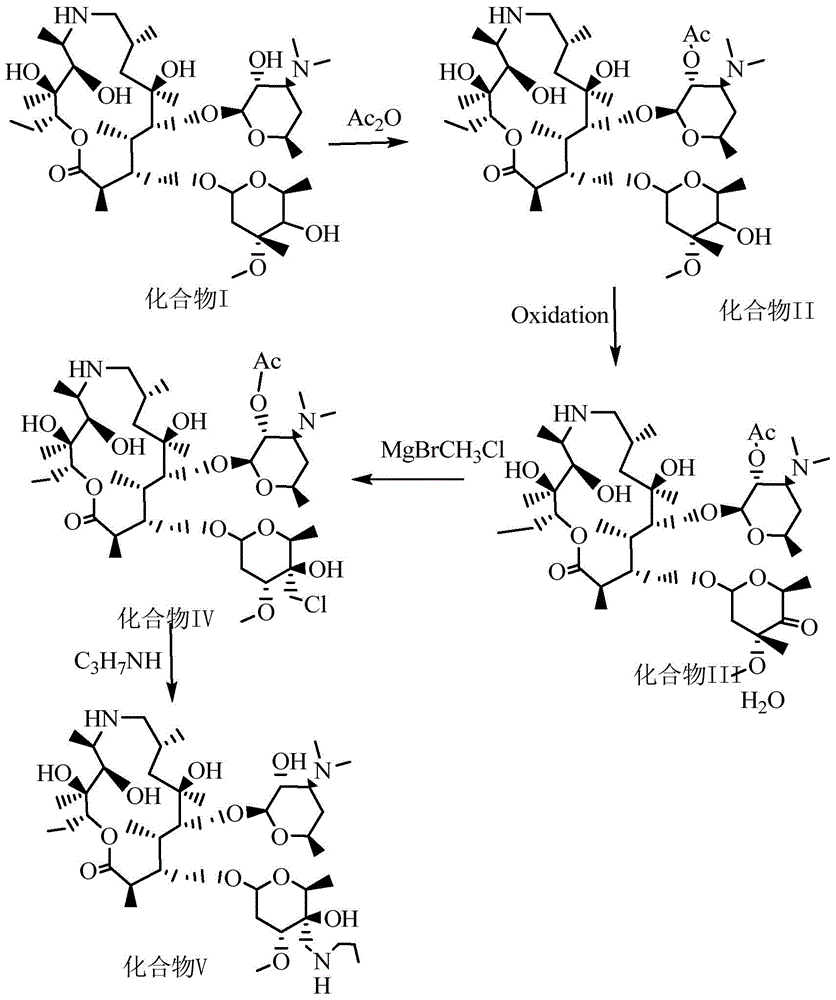
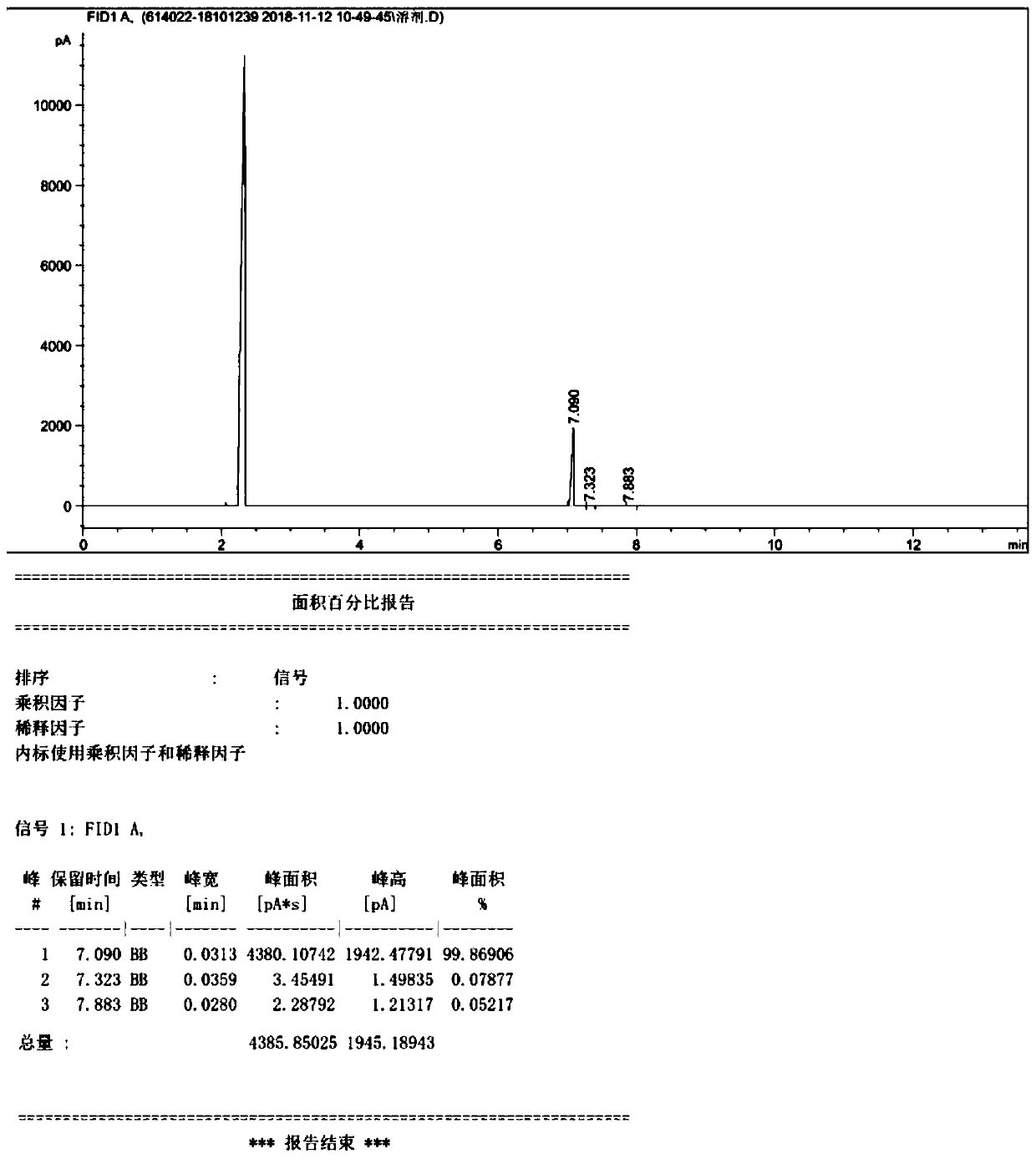
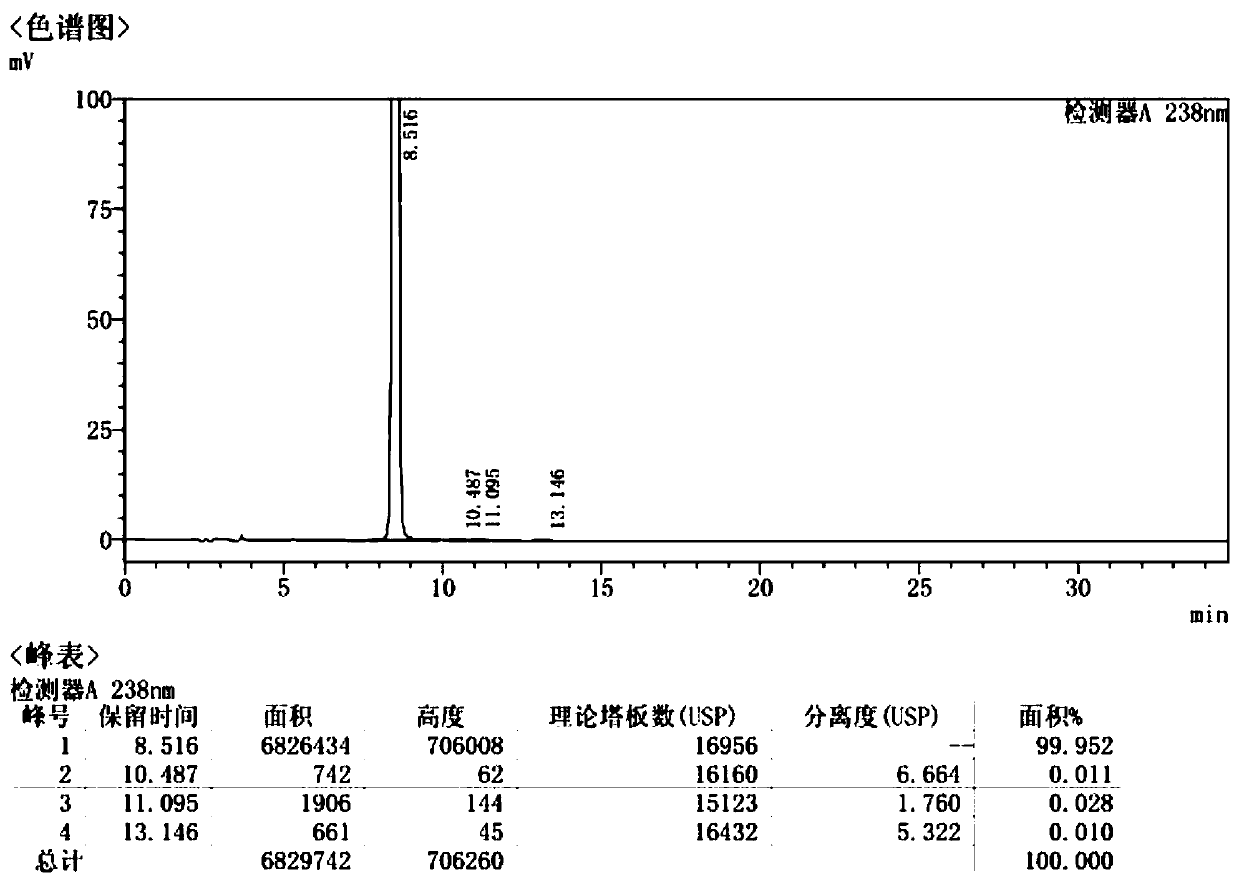
InactiveCN104861018AAvoid ultra-low temperature reaction processHarsh reaction conditionsSugar derivativesSugar derivatives preparationGrignard reagentRoom temperature
The invention discloses a preparation method of draxxin. The preparation method comprises the following steps: the Grignard reaction is adopted, hydroxyl protection ketone reacts with a Grignard reagent, a chloromethyl is introduced in the position of a carbonyl, the carbonyl is reduced to hydroxyl, and then the hydroxyl reacts with n-propylamine to prepare draxxin, so that the harsh reaction condition during the carbonyl epoxidation process is avoided. According to the preparation method of draxxin, the temperature range during the epoxidation reaction process is controlled to be from the room temperature to 80 DEG C, the reaction is fast, the operating time is shortened by 15% or above, the comprehensive yield is increased by 5% or above, the operation is simple and convenient, and the enlarged production is facilitated.
Owner:RINGPU TIANJIN BIOLOGICAL PHARMA
Preparation method of high purity 2-benzyloxy bromoethane
InactiveCN109896934AShort stepsSimple and fast operationOrganic chemistryOrganic compound preparationBromoethaneUmeclidinium bromide
The invention relates to a preparation method of an important intermediate 2-benzyloxy bromoethane (formula I) of umeclidinium bromide. The conditions of the preparation method are mild and controllable; post-treatment is simple; yield is high; and the purity of prepared 2-benzyloxy bromoethane is high.
Owner:SHANDONG ACADEMY OF PHARMACEUTICAL SCIENCES
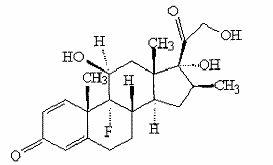
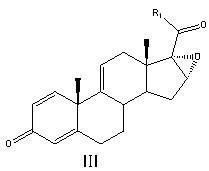
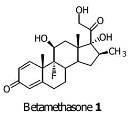
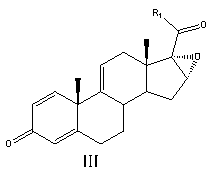
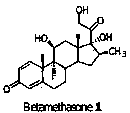
Preparation method of betamethasone intermediate
The invention relates to a preparation process of a betamethasone intermediate. The structural formula of the betamethasone intermediate is shown as a formula I, and is prepared by undergoing a ketone group protection reaction, a Grignard reaction and a hydrolysis deprotection reaction on a compound III; and the ketone group protection reaction comprises making a compound II, diazanyl carboxylic ether and an acid react to obtain a compound IV. The method has high yield, and contributes to protecting the environment; and the use of toxic substances is avoided. The structural formula of the compound III is shown as a formula III.
Owner:HUNAN NORCHEM PHARMACEUTICAL CO LTD
Method for efficiently dehalogenating waste plastics
ActiveCN105295088APromote conversionIncrease the number ofPlastic recyclingReaction temperatureEthyl Chloride
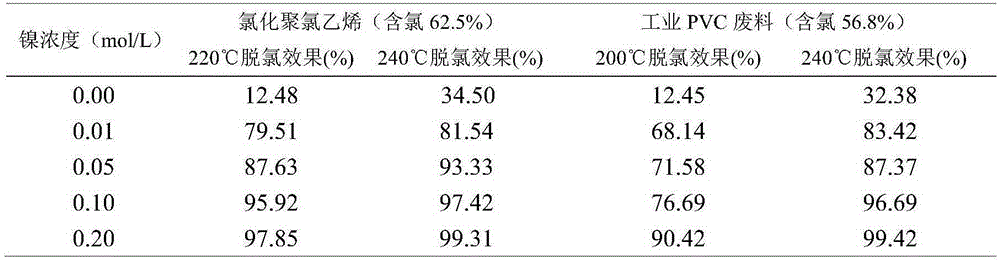
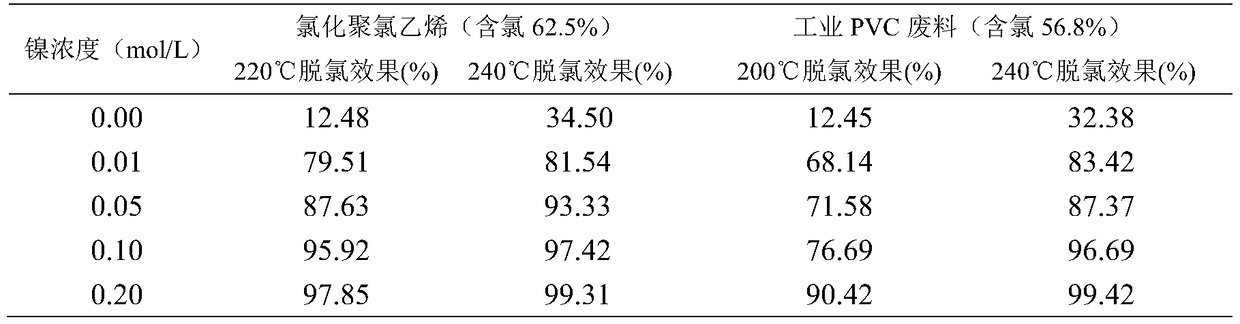
The invention discloses a method for efficiently dehalogenating waste plastics. The method comprises the following three steps: 1. quenching and tempering: adding an alkaline chemical raw material to the waste plastics, then adding a solution of a Ni<2+> chemical raw material / waste material and stirring and uniformly mixing the materials; 2. hydrothermal treatment: carrying out hydrothermal treatment on the prepared waste plastics at 200-240 DEG C for 10-60 minutes; 3. dehydration: carrying out solid-liquid separation on a mixture obtained after hydrothermal treatment, thus obtaining coke. The method has the beneficial effects that the function of Ni<2+> in plastic dehalogenation is confirmed for the first time; the method has the characteristics that the adding proportion of the alkaline chemical raw material is extremely small; the cost is low; the process is simple; the halogen removal efficiency is high; the reaction temperature is low; the time is short; the halogen containing rate of the waste plastics is reduced to about 0 from 56.8-74%; secondary pollution can not be caused; the plastics excluding halogen and some chemical raw materials after separation can be recycled, thus avoiding the problems of chlorine corrosion and dioxin pollution during halogen-containing plastic burning and providing beneficial conditions for harmless treatment and recycling of the halogen-containing plastics.
Owner:CHINA UNIV OF MINING & TECH
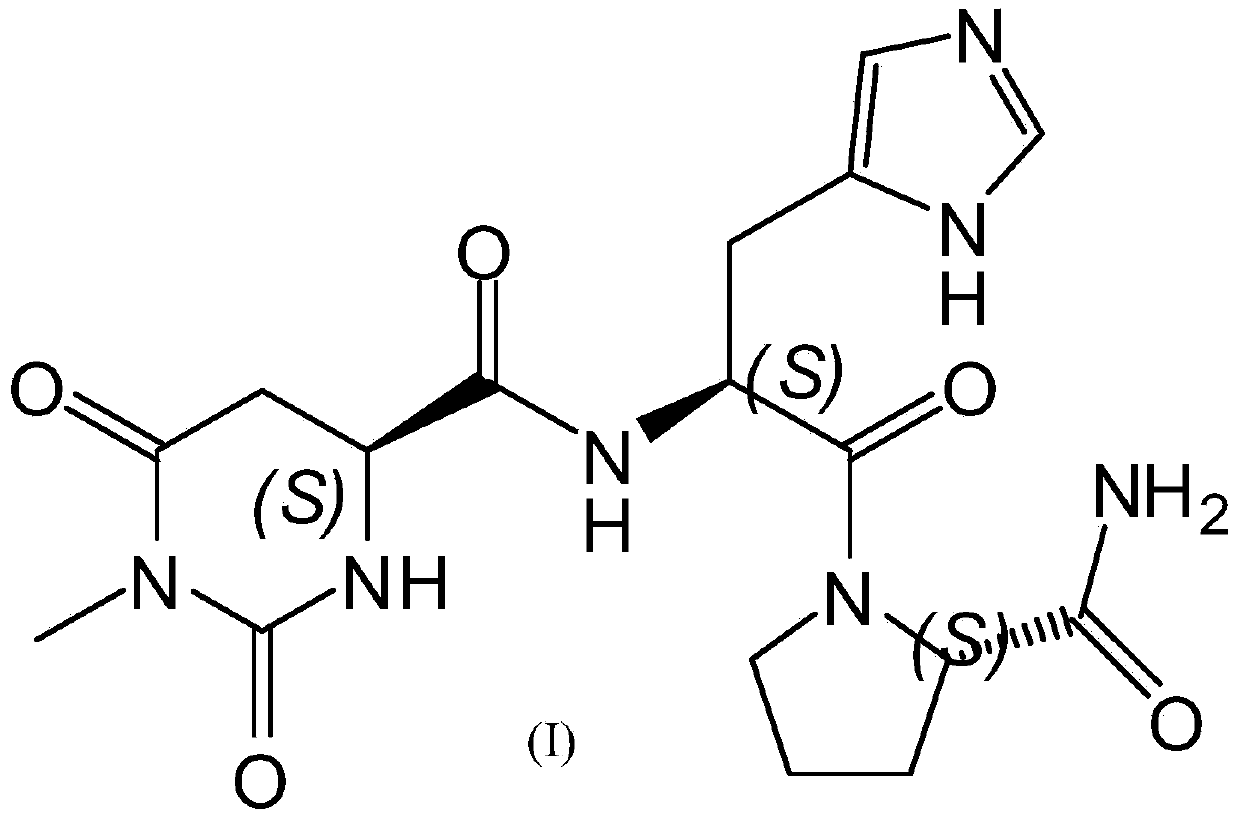
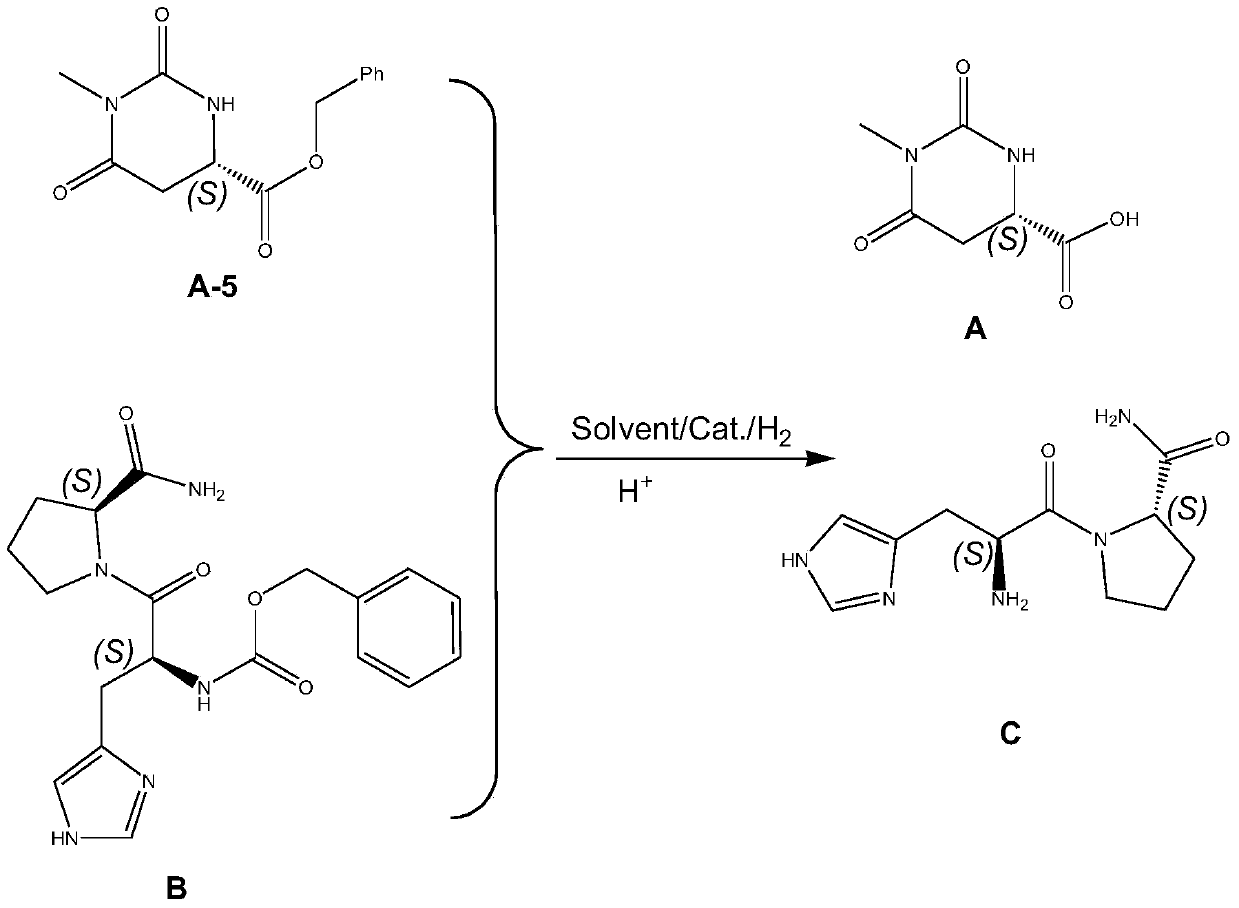
Preparing method for taltirelin and midbody of taltirelin
ActiveCN105254573AAvoid using effectsAvoid toxic reagents such as methyl iodideOrganic chemistryL-AspartateTaltirelin
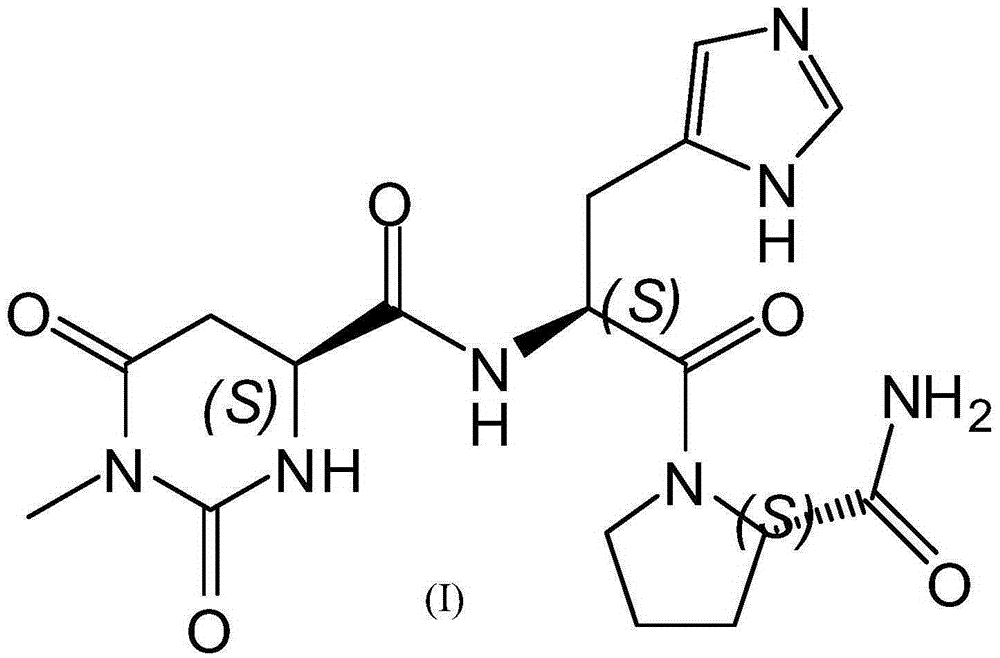
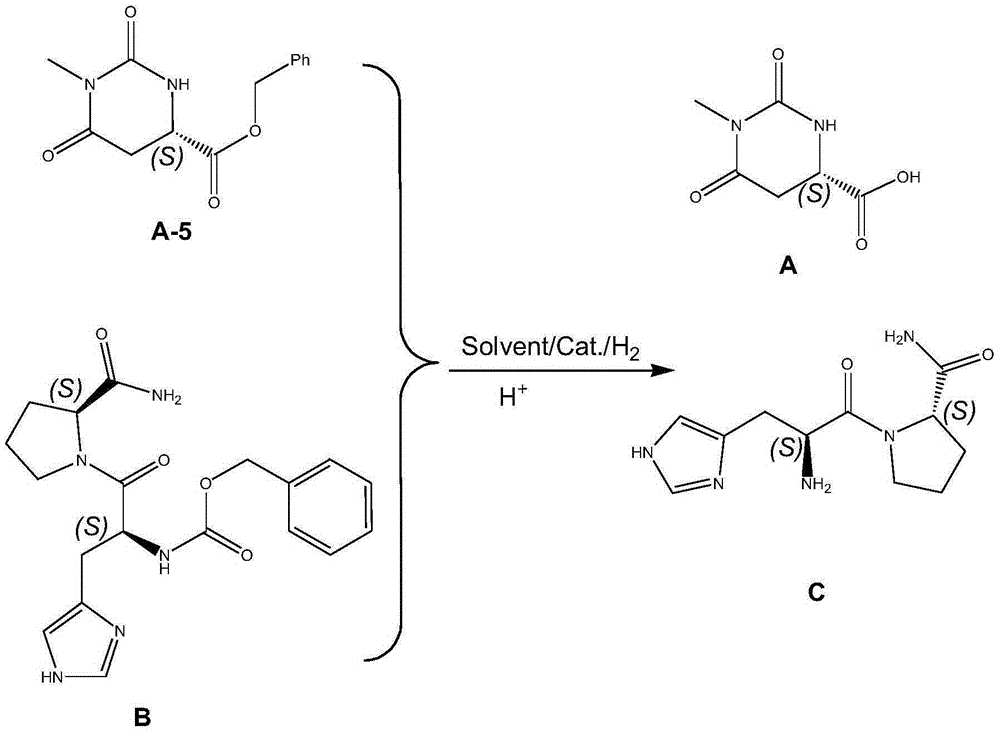
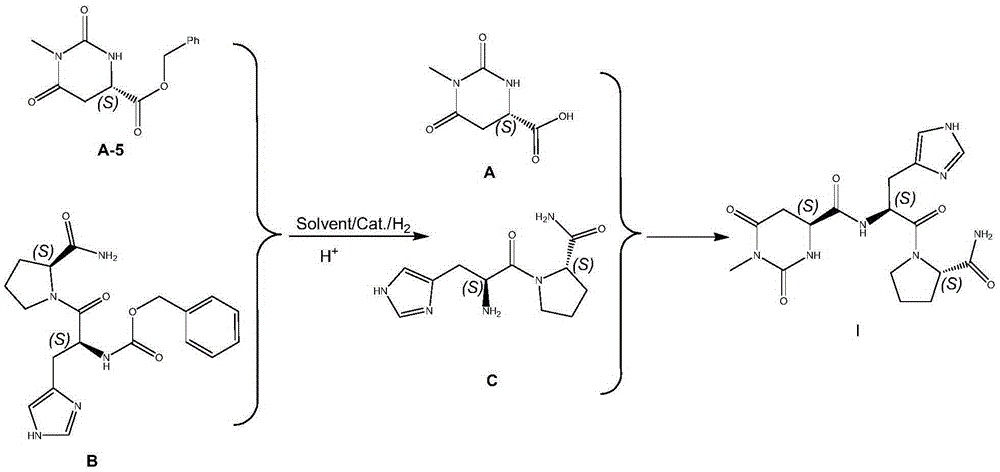
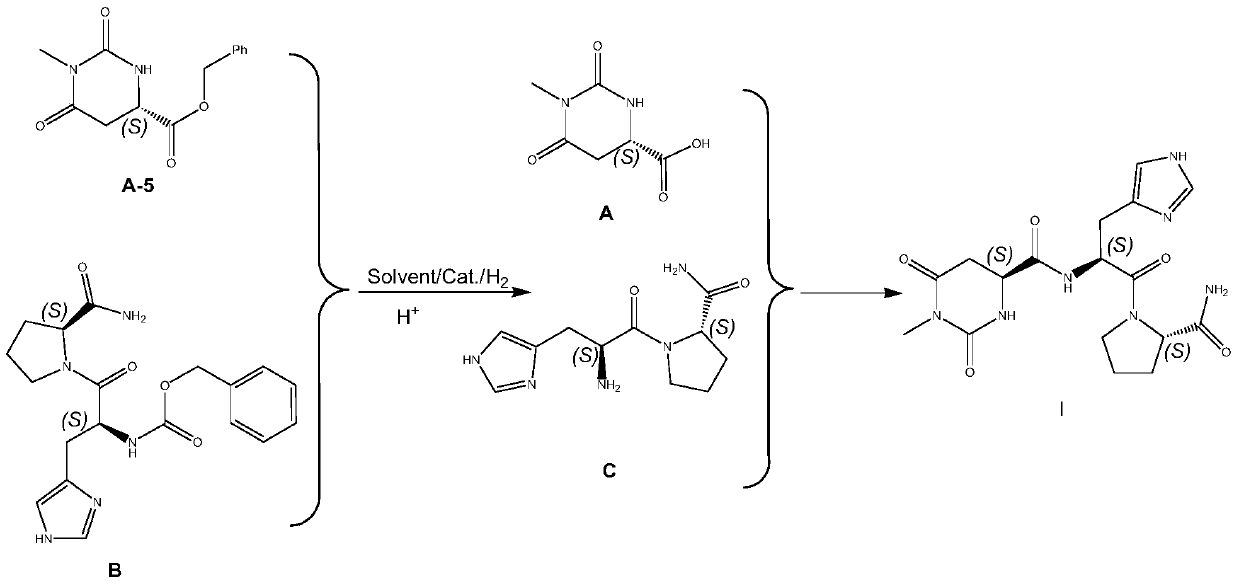
The invention discloses a preparing method for taltirelin and a midbody of taltirelin. The preparing method comprises the steps that L-aspartic acid-4-methyl ester or other hydrochloride serves as raw materials, a compound A-5 is obtained through ammonolysis, carboxyl protection and cyclization, the compound A-5 and a compound B are simultaneously subjected to deprotection through a one-pot reaction under the acid condition, and 1-methyl-L-4,5-dihydro orotic acid (a compound A) and a compound C are obtained. The compound A and the compound C can be used for a next-step condensation reaction through separation or without separation, and taltirelin shown in the formula I is prepared through the condensation reaction. According to the technical scheme, the prepared compound A and compound C or taltirelin has the advantages that the utilization rate of the raw materials is high, operation changes gradually, the preparation steps are few, the reaction condition is mild, the requirement for production equipment is low, the yield is high, and cost is low.
Owner:重庆莱美隆宇药业有限公司
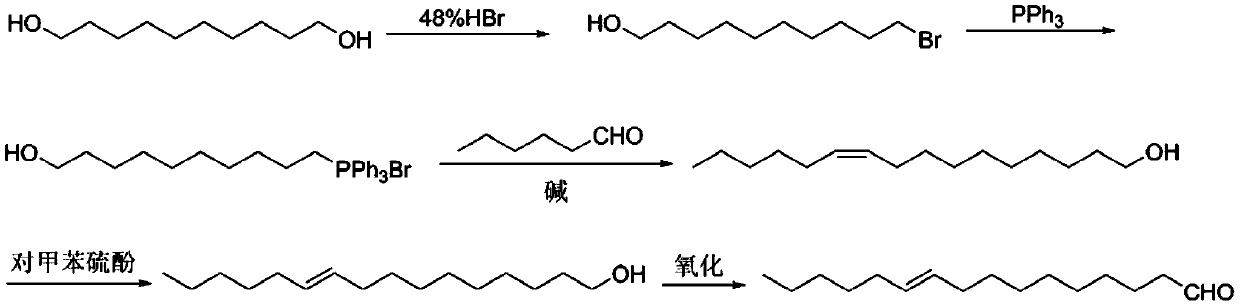
Industrial synthesis method of dichocrocis punctiferalis sex pheromone
ActiveCN111253228AMild conditionsSuitable for mass productionOrganic compound preparationOrganic chemistry methodsDichocrocis punctiferalisChemical synthesis
The invention belongs to the field of chemical synthesis, and particularly relates to an industrial synthesis method of a dichocrocis punctiferalis sex pheromone. The method comprises the following steps: taking 1, 10-decanediol as a raw material; carrying out a single-side bromination reaction to prepare 10-bromodecanol; then reacting the 10-bromodecanol with triphenylphosphine to obtain 10-hydroxydecyltriphenylphosphine salt; performing a Wittig reaction with n-hexaldehyde under the action of alkali to obtain cis-based 10-hexadecene-1-ol; performing isomerization on the cis-based enol underthe action of p-methylthiophenol to obtain trans-based 10-hexadecene-1-ol; and finally performing oxidation under the action of an oxidant to obtain the final product 10-hexadecenal. The method is mild in reaction condition and suitable for large-scale production.
Owner:CHANGZHOU UNIV
Chitin deacetylase and chitin nanofiber dispersion liquid as well as preparation method and application of chitin nanofiber dispersion liquid
The invention belongs to the technical field of preparation of chitin nanofibers and relates to chitin deacetylase and chitin nanofiber dispersion liquid as well as a preparation method and application of the chitin nanofiber dispersion liquid. The chitin deacetylase is prepared from a penicillium oxalicum 3.5705 strain through purification, wherein the collection number of the penicillium oxalicum strain is CGMCC No. 3.5705. The preparation method of the chitin nanofiber dispersion liquid comprises the following steps: Performing partial deacetylation modification on chitin by using the chitin deacetylase, to obtain partial chitosan; treating the partial chitosan under a mechanical effect, to obtain the chitin nanofiber dispersion liquid. According to the chitin deacetylase and the chitinnanofiber dispersion liquid as well as the preparation method and application of the chitin nanofiber dispersion liquid provided by the invention, the chitin is subjected to partial deacetylation modification by a novel biological enzyme process instead of traditional concentrated base pyrolysis chemical treatment, and the reaction is mild, nontoxic and pollution-free; the preparation method is green and environmentally friendly, the yield of chitin nanofiber is increased, and efficient utilization of the chitin biomass resources is realized.
Owner:NANJING FORESTRY UNIV
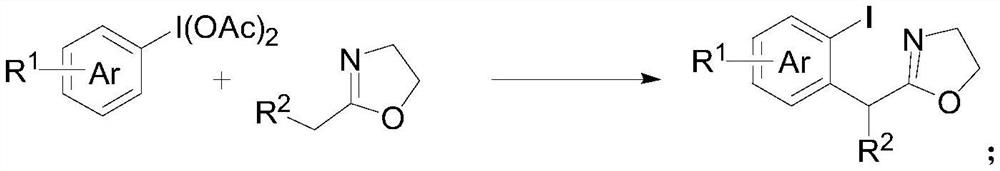
Method for preparing alpha-arylcarbonyl compound and product
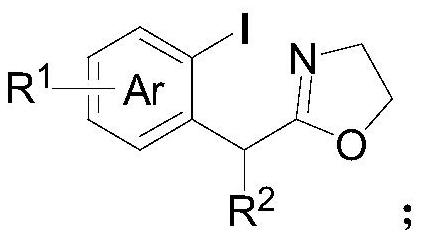
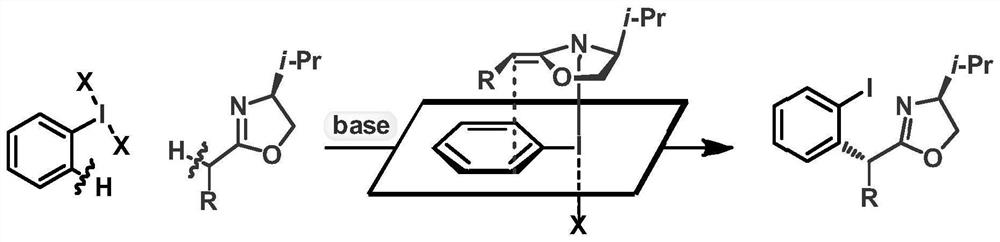
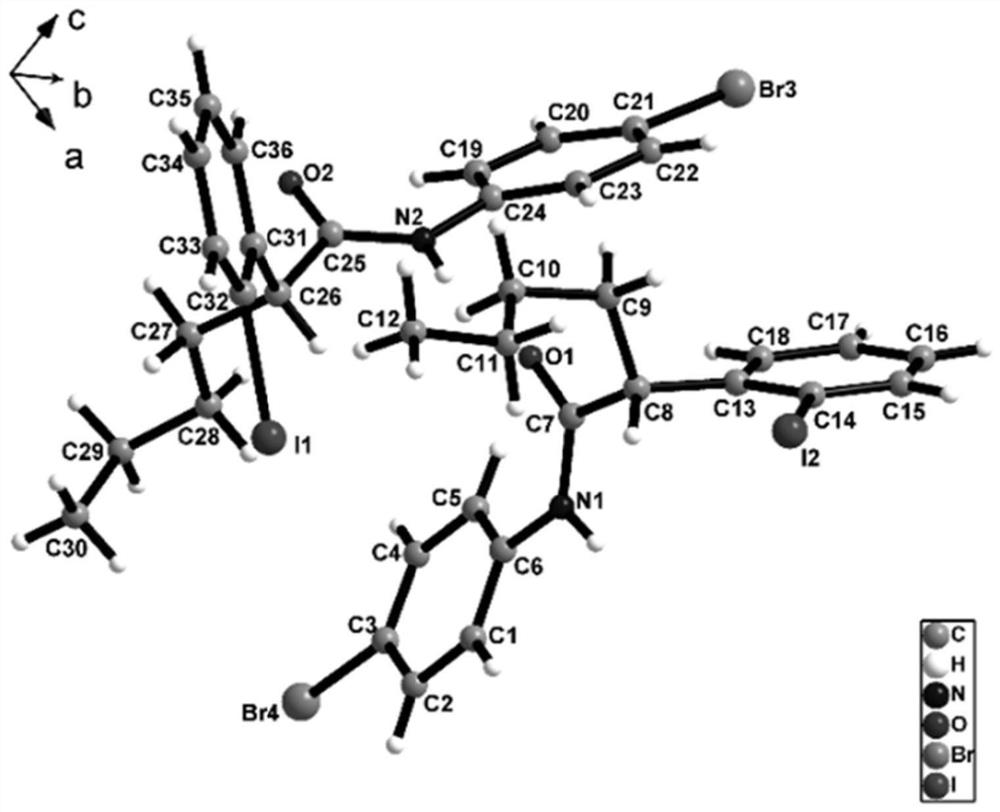
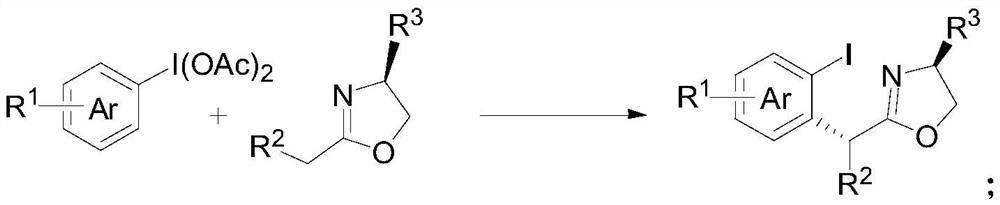
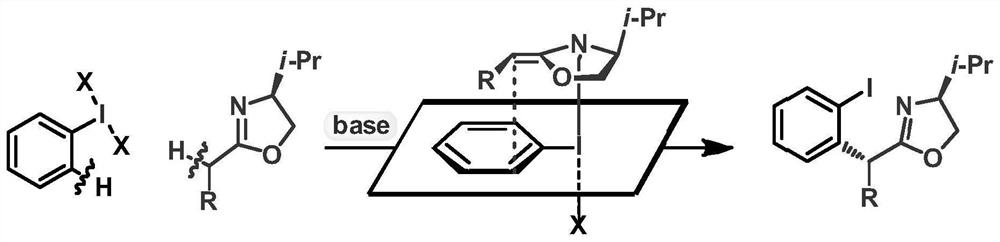
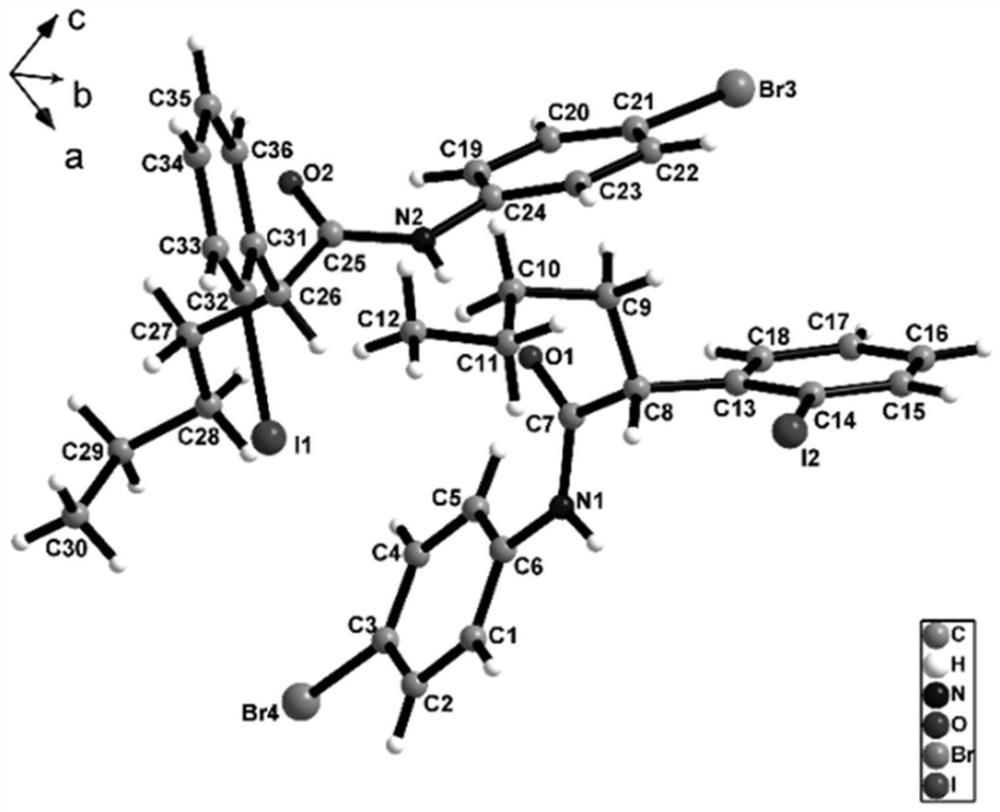
ActiveCN113135869ARaw materials are cheap and easy to getHarsh reaction conditionsOrganic chemistryArylHalogen
The invention discloses a method for preparing an alpha-aryl carbonyl compound, which comprises the following steps: activating an R1 substituted iodobenzene diacetate compound by Lewis acid, and then reacting with an R2 substituted oxazoline compound in the presence of alkali to obtain the alpha-aryl carbonyl compound, wherein R1 is selected from H, one or more alkyl groups, one or more halogenated alkyl groups, alkoxycarbonyl, halogen or multi-substitution formed by the combination of the above atoms, and R2 is selected from H, alkyl and substituted alkyl, wherein a substituent group is selected from cycloalkyl, halogen, alkoxy, heterocycloalkyl, substituted heterocycloalkyl, aryl, cyano, aryloxy and carbonyl. The method has the advantages of mild reaction conditions, easy product separation and simple operation.
Owner:ZHEJIANG NORMAL UNIVERSITY
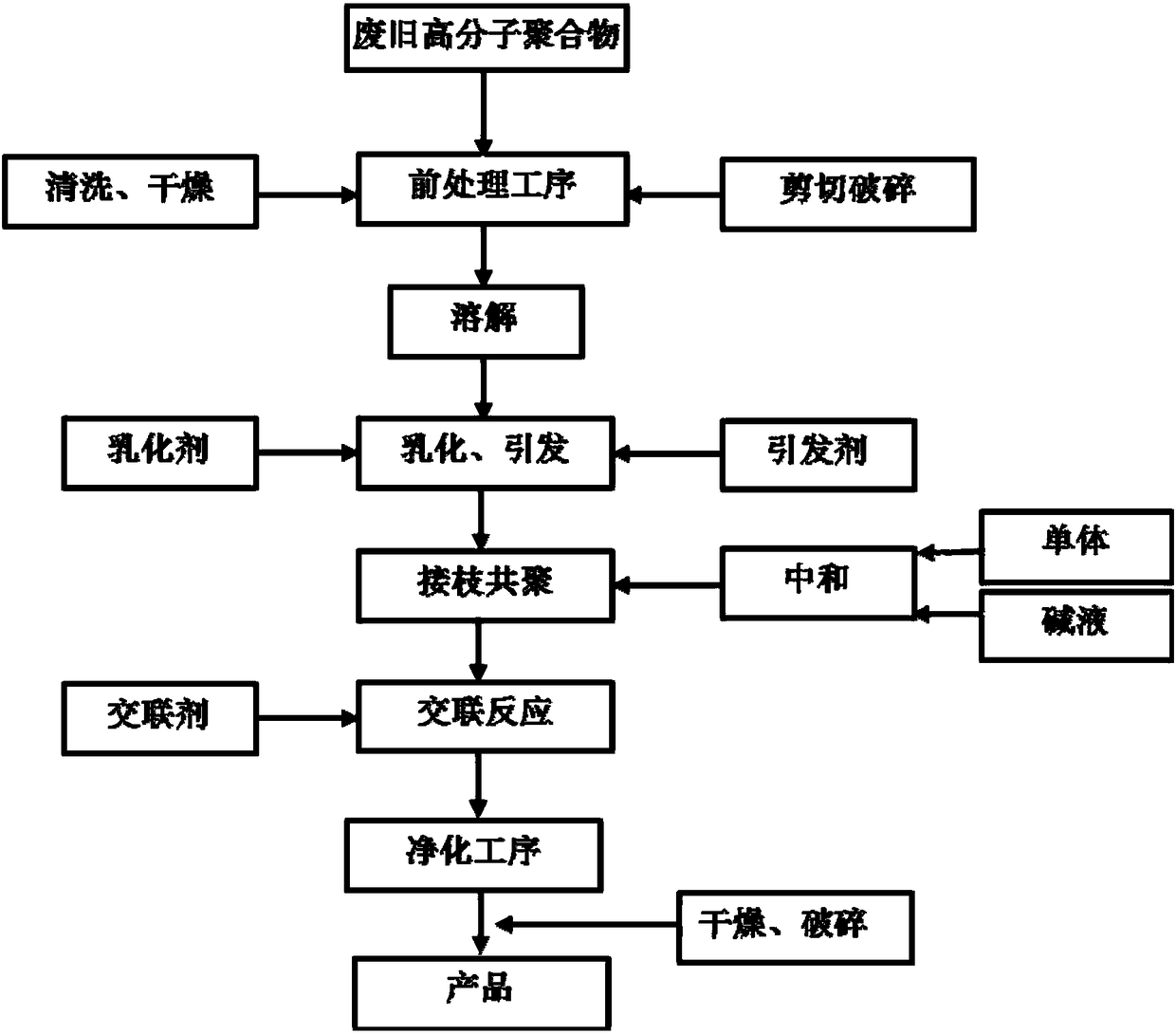
Method for preparing water-absorbent resin from waste macromolecular polymer
The invention provides a process for preparing multifunctional water-absorbent resin from waste macromolecular materials and aims at practical problems of complex recovery process, high cost, proneness to secondary pollution and the like of a traditional waste macromolecular polymer recovery process. The process includes a dissolution procedure, an emulsification initiating procedure, a graft copolymerization procedure, a cross-linking reaction procedure and a product purification procedure. The synthesized novel material is efficient in moisture absorption performance, great in moisture retention capacity and high in recyclability and has a high application value in fields of agriculture and forestry gardening, environmental pollution control, building interior curing agents, drying agents and the like.
Owner:RES CENT FOR ECO ENVIRONMENTAL SCI THE CHINESE ACAD OF SCI
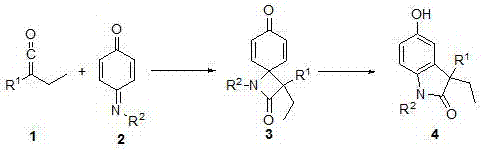
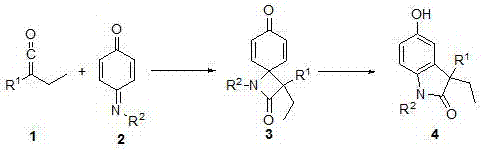
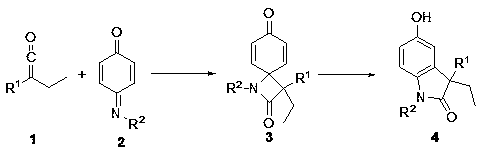
Synthesis method of 3-ethyl-5-hydroxy-1,3-diaryl indolone
The invention discloses a novel method for synthetizing 3-ethyl-5-hydroxy-1,3-diaryl indolone through reaction of 1,3-bis-(2, 4, 6-trimethylphenyl)-imidazole azacyclo carbene catalysis aryl ethyl ketone and N-aryl quinone imine. An imidazole azacyclo carbene catalyst is used; the [2+2] cycloaddition reaction is effectively catalyzed; an obtained four-membered ring product effectively promoted to be subjected to aromatization by a catalytic amount of Lewis acid; an aromatized indolone type product is obtained. The method has the advantages that the conditions are mild; the operation is simple;green and efficient effects are achieved; the atom utilization rate is high, and the like.
Owner:NORTHWEST UNIV(CN)
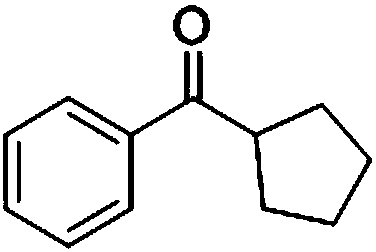
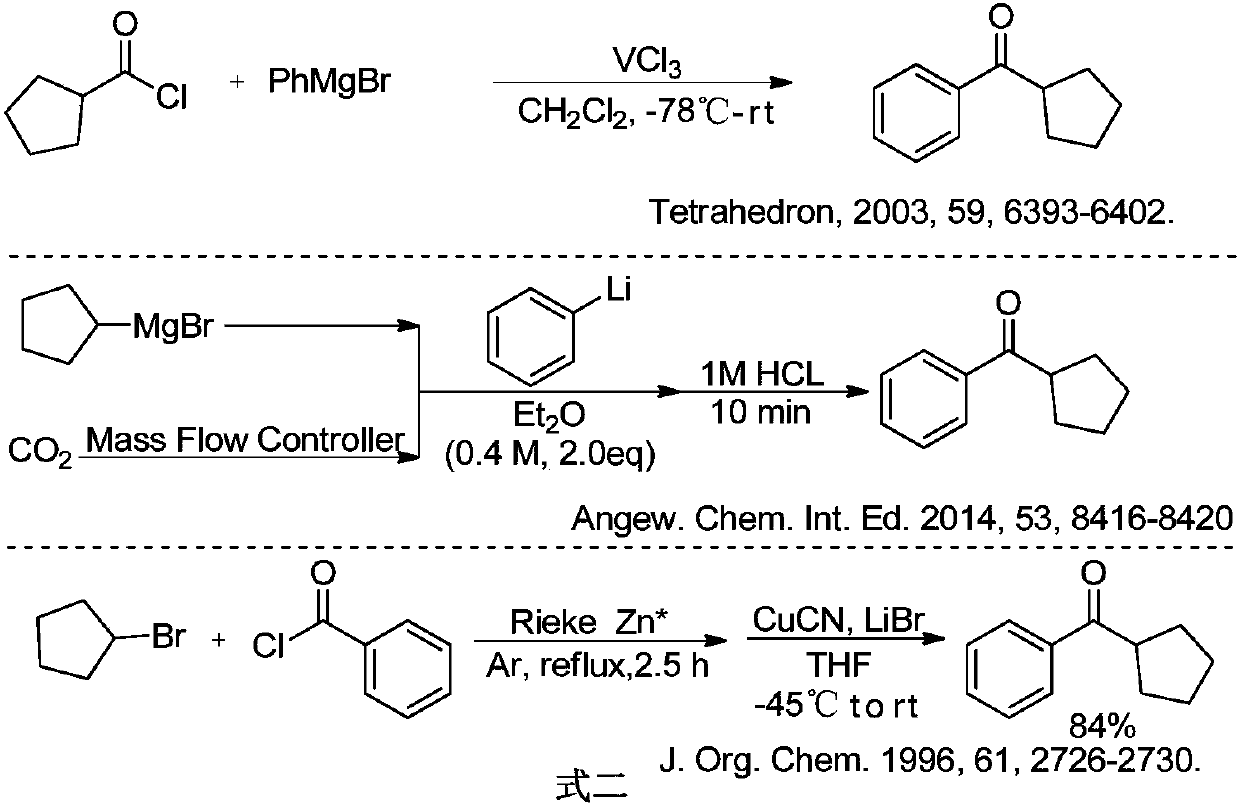
Preparation method of cyclopentyl phenyl ketone
ActiveCN105753682AReduce pollutionIncrease generation costOrganic compound preparationPreparation from carboxylic acid esters/lactonesMethyl acetateEthyl acetate
The invention provides a preparation method of cyclopentyl phenyl ketone.The preparation method includes hydrolyzing 2-cyclopentyl benzoylacetate as a raw material in basic solvents to obtain a product, namely the cyclopentyl phenyl ketone, wherein the 2-cyclopentyl benzoylacetate is 2-cyclopentyl methyl benzoylacetate or 2-cyclopentyl ethyl benzoylacetate.The preparation method of the cyclopentyl phenyl ketone has the advantages of environment-friendly solvents, little environmental pollution, easiness in operation, high yield, short technological period and low preparation cost.
Owner:CHENGDU ORGANIC CHEM CO LTD CHINESE ACAD OF SCI +1
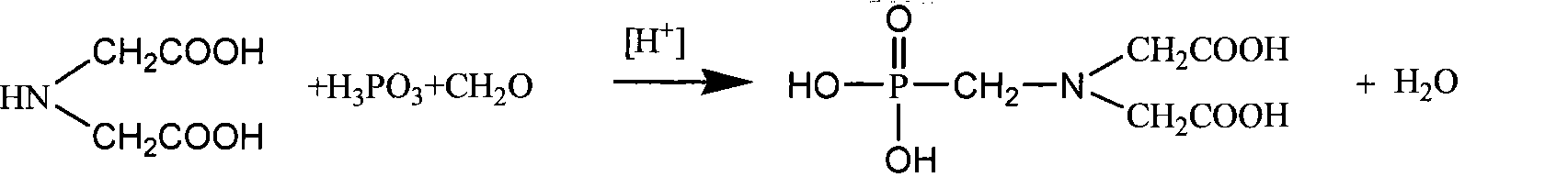
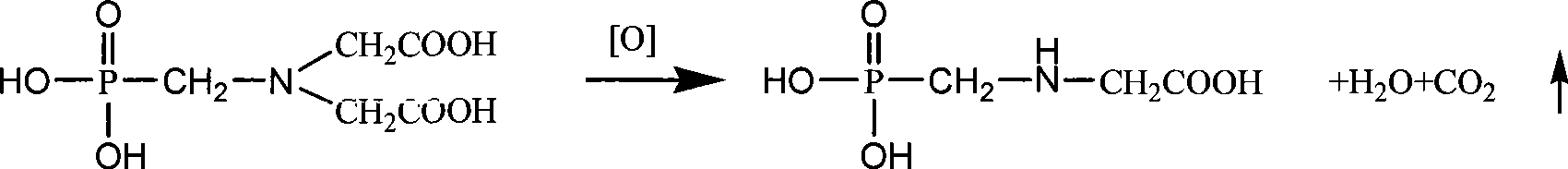
Production of iminodiacetic acid by microorganism catalytic processes and bacterial strain thereof
ActiveCN101392276BReduce outputLow impurity contentBacteriaMicroorganism based processesBacterial strainFermentation
Owner:ZHEJIANG UNIV OF TECH
Method for co-liquefaction of coal and waste plastics under mild condition
InactiveCN102344823BMild reaction conditionsHarsh reaction conditionsLiquid hydrocarbon mixture productionDepolymerizationGas phase
The invention is a method for co-liquefaction of coal and waste plastics under a mild condition, which comprises the following steps: adding coal, waste plastics, lignosulfonate, a catalyst, and a solvent into an autoclave, introducing hydrogen to increase the pressure to 1.0-10.0 Mpa, heating to 300-450 DEG C for a liquefaction reaction, collecting the gas after the hydrogenation liquefaction test in the autoclave, analyzing the gas phase composition by a gas chromatography, collecting all the other liquefaction products in a filter paper cylinder, performing hexane and tetrahydrofuran solvent extraction in order by a soxhlet extraction device, wherein the substance soluble in hexane is oil, and the substances soluble in tetrahydrofuran but insoluble in hexane are asphaltene and preasphaltene, and calculating the total conversion rate of the liquefaction by using the amount of the insoluble substances in tetrahydrofuran. The invention allows the coal to perform a depolymerization reaction at a low temperature, and to be co-liquefied with waste plastics; the extraction rate of coal by solvents such as pyridine and the like is increased, and the coal liquefaction process is accelerated; not only the hydrogen consumption and production cost for coal liquefaction are greatly reduced, but also white pollution caused by waste plastics is solved.
Owner:LIUPANSHUI NORMAL UNIV
Method for reducing graphene oxide by utilizing microbial fermentation liquor
The invention provides a method for reducing graphene oxide by utilizing microbial fermentation liquor, and belongs to the field of material synthesis. Genetically engineered Escherichia coli for heterologous expression of [FeFe] hydrogenase is constructed first, sterile fermentation liquor is obtained through treatment, and the sterile fermentation liquor is applied to reduction of graphene oxide. Therefore, biological green reduction of graphene oxide at normal temperature and normal pressure is realized.
Owner:JIANGSU UNIV
A method for efficient dehalogenation of waste plastics
ActiveCN105295088BPromote conversionIncrease the number ofPlastic recyclingHalogenReaction temperature
The invention discloses a method for efficiently dehalogenating waste plastics. The method comprises the following three steps: 1. quenching and tempering: adding an alkaline chemical raw material to the waste plastics, then adding a solution of a Ni<2+> chemical raw material / waste material and stirring and uniformly mixing the materials; 2. hydrothermal treatment: carrying out hydrothermal treatment on the prepared waste plastics at 200-240 DEG C for 10-60 minutes; 3. dehydration: carrying out solid-liquid separation on a mixture obtained after hydrothermal treatment, thus obtaining coke. The method has the beneficial effects that the function of Ni<2+> in plastic dehalogenation is confirmed for the first time; the method has the characteristics that the adding proportion of the alkaline chemical raw material is extremely small; the cost is low; the process is simple; the halogen removal efficiency is high; the reaction temperature is low; the time is short; the halogen containing rate of the waste plastics is reduced to about 0 from 56.8-74%; secondary pollution can not be caused; the plastics excluding halogen and some chemical raw materials after separation can be recycled, thus avoiding the problems of chlorine corrosion and dioxin pollution during halogen-containing plastic burning and providing beneficial conditions for harmless treatment and recycling of the halogen-containing plastics.
Owner:CHINA UNIV OF MINING & TECH
Method for preparing chiral alpha-arylcarbonyl compound and product
ActiveCN113135870ARaw materials are cheap and easy to getHarsh reaction conditionsOrganic chemistry methodsCarbonyl groupCombinatorial chemistry
The invention discloses a method for preparing a chiral alpha-aryl carbonyl compound, which comprises the following steps: activating an R2 substituted iodobenzene diacetate compound by Lewis acid, and then reacting with an R3 stereosubstituted oxazoline compound in the presence of alkali to obtain the chiral alpha-aryl carbonyl compound, wherein R1 is selected from H, one or more alkyl groups, one or more halogenated alkyl groups, alkoxycarbonyl, halogen or multi-substitution formed by the combination of the above atoms, R2 is selected from H, alkyl and substituted alkyl, wherein a substituent group is selected from cycloalkyl, halogen, alkoxy, heterocycloalkyl, substituted heterocycloalkyl, aryl, cyano, aryloxy and carbonyl, and R3 is selected from alkyl, substituted alkyl and aryl. The method has the advantages of mild reaction conditions, good stereoselectivity, easy product separation and simple operation.
Owner:ZHEJIANG NORMAL UNIVERSITY
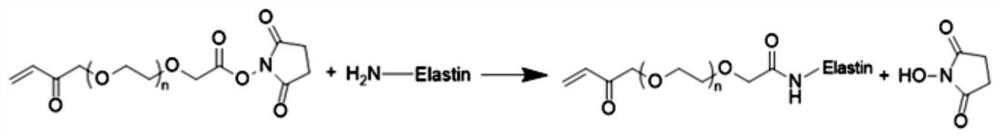
Method for culturing organoid by using elastin hydrogel
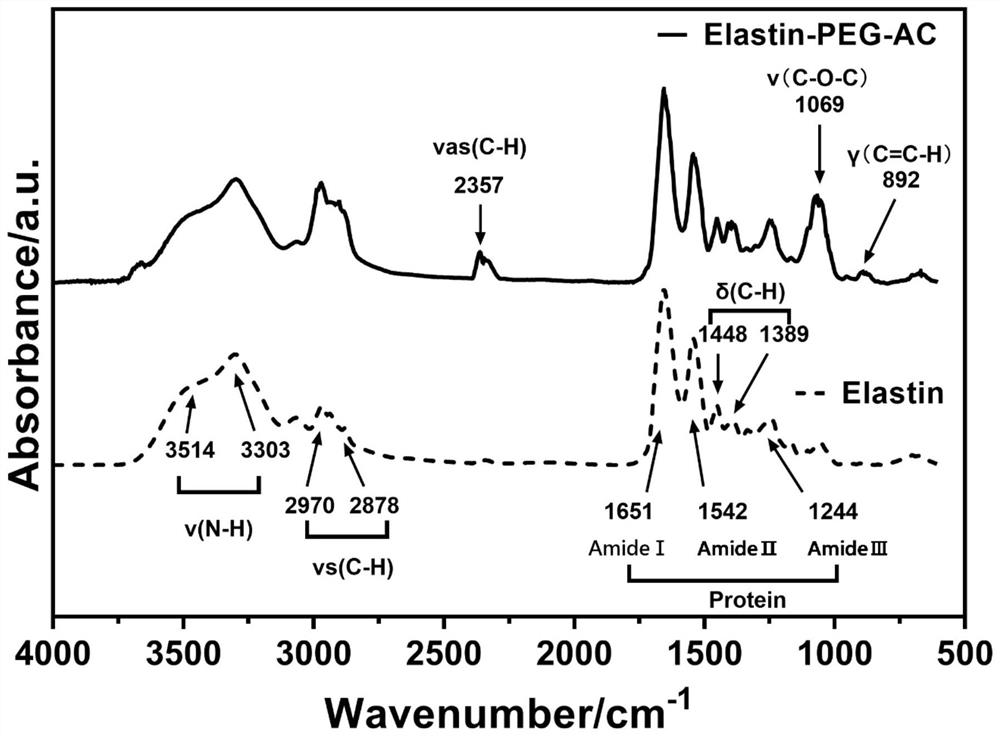
PendingCN114874975AImprove preparation efficiencyModified grafting efficiency is highGastrointestinal cellsCulture processPhotoinitiatorMolecular biology
The invention discloses a method for culturing an organoid by using elastin hydrogel, which comprises the following steps: dissolving soluble elastin in deionized water or a buffer solution to prepare an elastin aqueous solution; the preparation method comprises the following steps: firstly, preparing an elastin, then adding acrylate-polyethylene glycol-N-hydroxysuccinimide or methacrylate-polyethylene glycol-N-hydroxysuccinimide to obtain modified elastin, and dialyzing, freezing and freeze-drying the modified elastin in deionized water to obtain dry modified elastin; dissolving the dry-state modified elastin in a phosphate physiological buffer solution, adding a photoinitiator, mixing with an organoid cell mass, and irradiating under a light source with a certain wavelength to obtain an elastin hydrogel / cell compound; and culturing in an organoid culture medium, and amplifying the cells in the hydrogel to obtain the organoid. The method disclosed by the invention is simple in process, rapid in reaction and high in elastin content, and the cultured organoid is stable in quality.
Owner:THE SEVENTH AFFILIATED HOSPITAL SUN YAT SEN UNIV SHENZHEN
A method and product for preparing chiral α-aryl carbonyl compound
ActiveCN113135870BRaw materials are cheap and easy to getHarsh reaction conditionsOrganic chemistry methodsArylHalogen
The invention discloses a method for preparing a chiral α-aryl carbonyl compound, comprising: R 2 The substituted iodobenzene diacetate compound was activated with Lewis acid and then reacted with R 3 Stereo-substituted oxazoline compounds react in the presence of a base to obtain the chiral α-aryl carbonyl compound; the R 1 R 2 selected from H, alkyl, substituted alkyl, wherein the substituent is selected from cycloalkyl, halogen, alkoxy, heterocycloalkyl, substituted heterocycloalkyl, aryl, cyano, aryloxy, carbonyl; R 3 Selected from alkyl, substituted alkyl, aryl. The method has mild reaction conditions, good stereoselectivity, easy separation of products and simple operation.
Owner:ZHEJIANG NORMAL UNIVERSITY
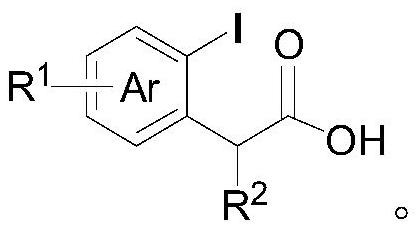
A kind of preparation method of deacetylceftiamidine
Owner:GUANGZHOU BAIYUSN TIANXIN PHARMA
Supported composite metal catalyst suitable for preparing acetamide through acetonitrile hydration reaction as well as preparation method and application of supported composite metal catalyst
ActiveCN112495391AEffectively regulate acidity and alkalinityHigh activityOrganic compound preparationCatalyst activation/preparationHydration reactionPtru catalyst
The invention relates to the field of acetonitrile hydration reaction catalysts. A supported composite metal catalyst suitable for preparation of acetamide through acetonitrile hydration reaction comprises 0.5-20 wt% of a metal oxide, 0.1-1 wt% of an additive and the balance SiO2, the metal oxide is one or more of Ni and Mn metal oxides and Mg, Ca, In, Ga, Bi and Zr oxides, and the additive is phosphorus or boron. The invention also relates to a preparation method and application of the catalyst. The efficient and low-cost catalytic method is developed for preparation of acetamide by acetonitrile hydration, the process is simple, no three wastes are generated, the target product selectivity is 99%, the product acetamide purity is 99.7%, the water content is 0.5%, and the use standard of pharmaceutical enterprises can be completely met.
Owner:SHANXI INST OF COAL CHEM CHINESE ACAD OF SCI
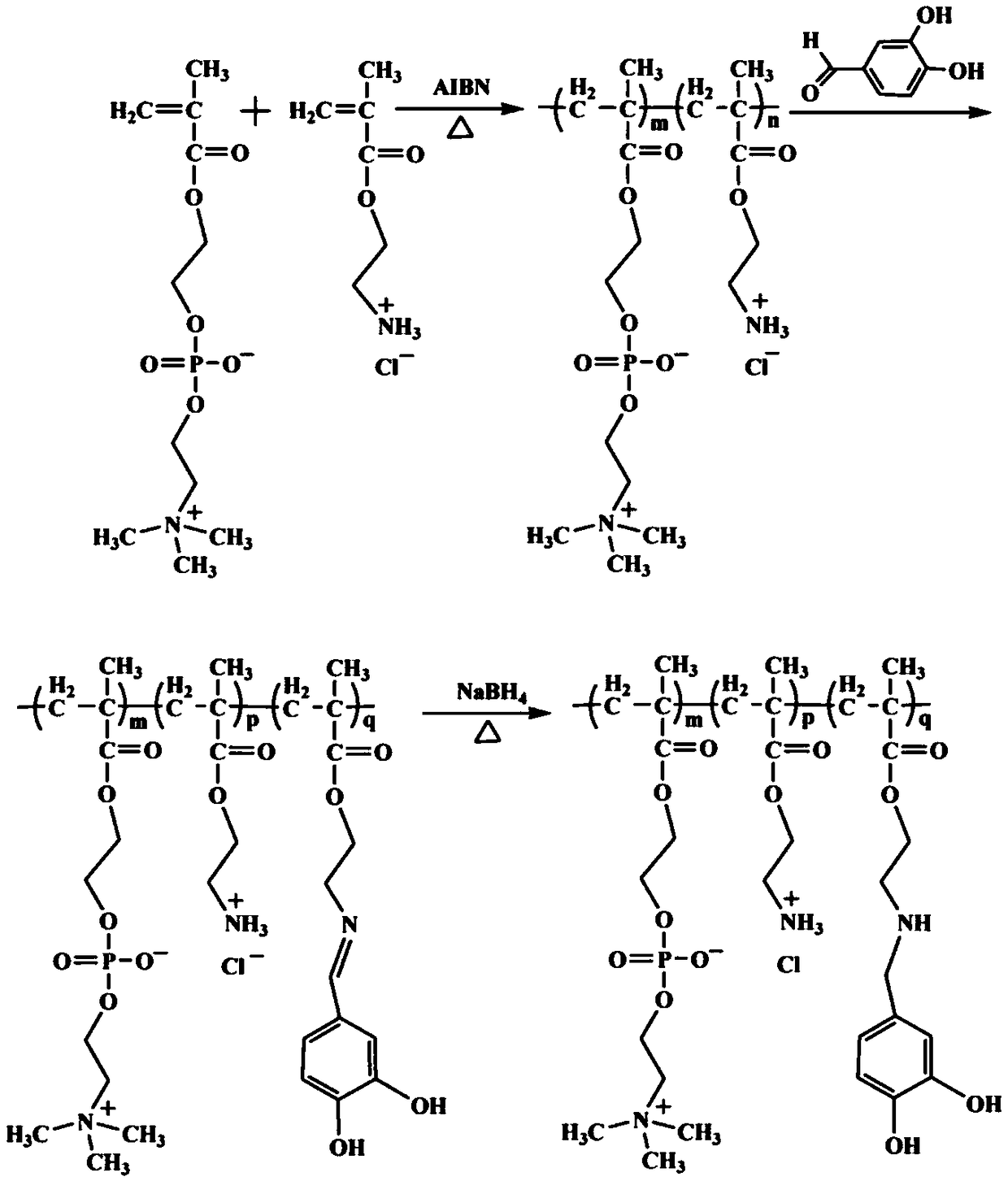
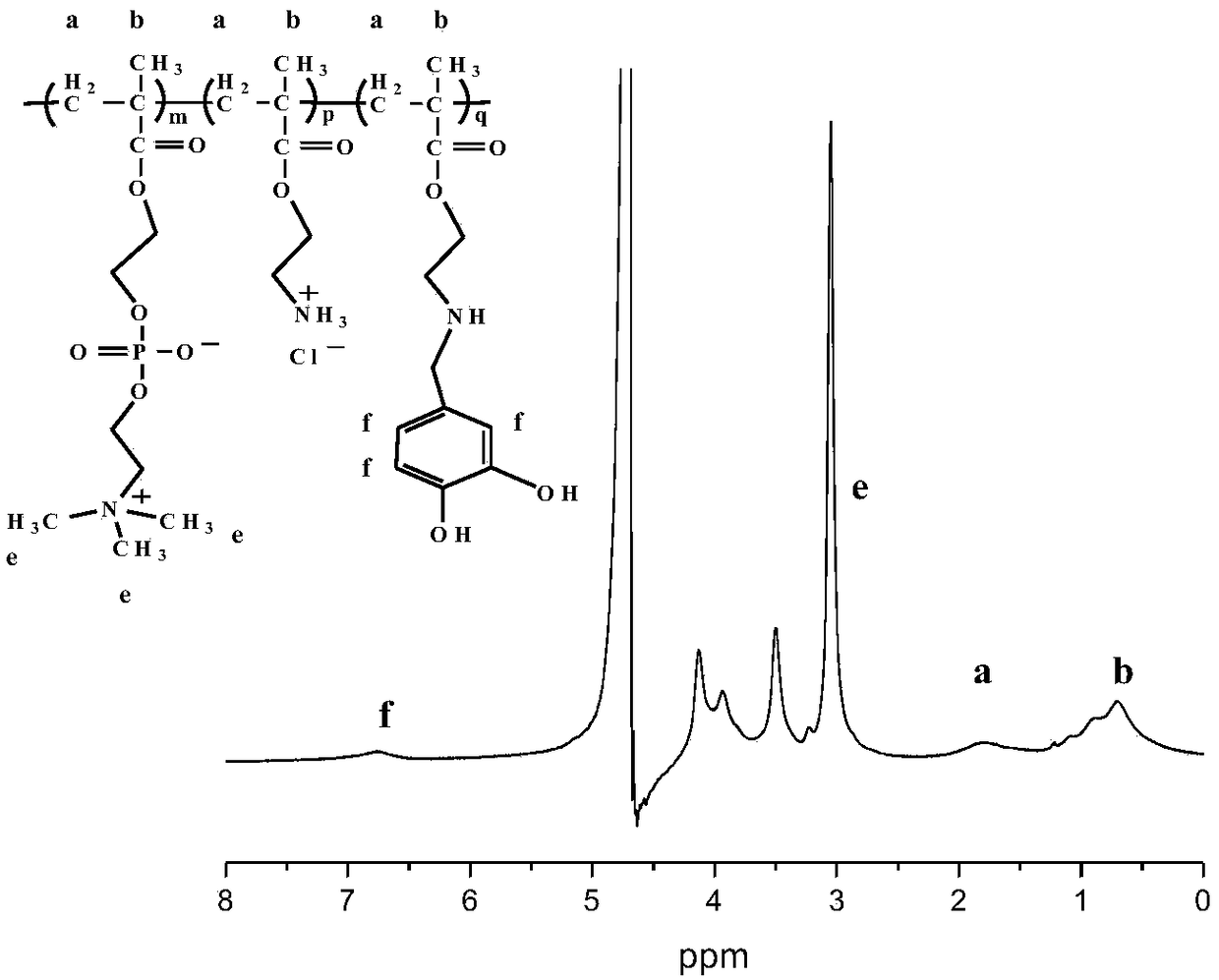
A method for synthesizing double biomimetic polymers based on the reaction of aldehyde groups and amino groups
The invention discloses a method for synthesizing a bionic polymer based on a reaction of aldehyde group and amino. The method comprises the following steps: synthesizing an amino-containing phosphorylcholine polymer; dissolving the amino-containing phosphorylcholine polymer into a polar solvent so as to obtain a polymer solution; heating the polymer solution to the temperature of 20-50 DEG C under nitrogen protection, adding 3,4-dihydroxy benzaldehyde into the polymer solution, and grafting under temperature maintaining and stirring conditions by utilizing the reaction of aldehyde group and amino; reducing with sodium borohydride, dialyzing the reaction product; and finally, performing freeze drying on the dialyzed reaction product, thereby obtaining the bionic polymer. The preparation method disclosed by the invention is simple, mild in conditions and controllable in grafting ratio, the o-diphenol hydroxyl has an adhesive effect of clam-imitated attachment protein dopamine, and the phosphorylcholine has anti-fouling performance of a cell outer membrane imitated structure. The polymer has dual bionic characteristics and is expected to have wide application prospects in the field of material surface modification.
Owner:XIAN UNIV OF SCI & TECH
Preparation process of isosorbide mononitrate
ActiveCN1257906CLow boiling pointReduce the temperatureOrganic chemistryCardiovascular disorderNitrationDimethyl isosorbide
Owner:SHANDONG NEWTIME PHARMA
A kind of synthetic method of 3-ethyl-5-hydroxyl-1,3-diarylindolinone
Owner:NORTHWEST UNIV
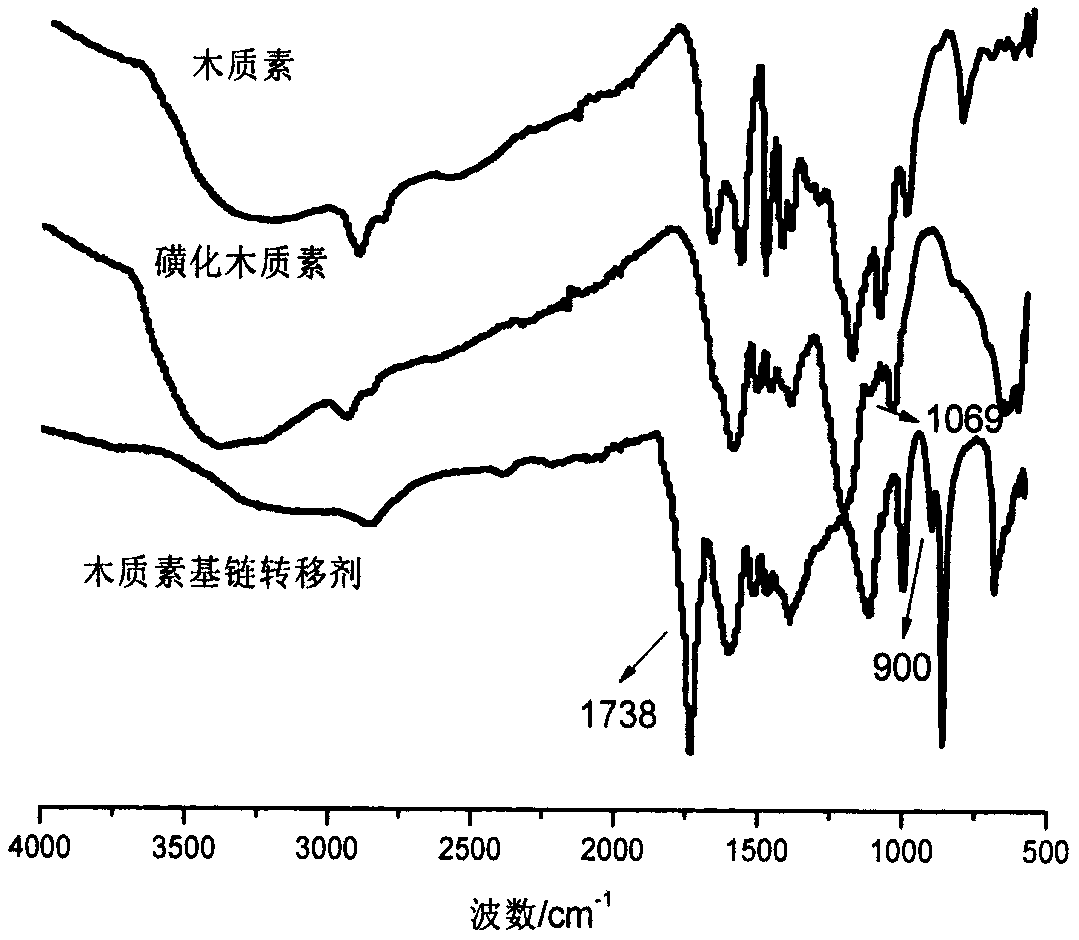
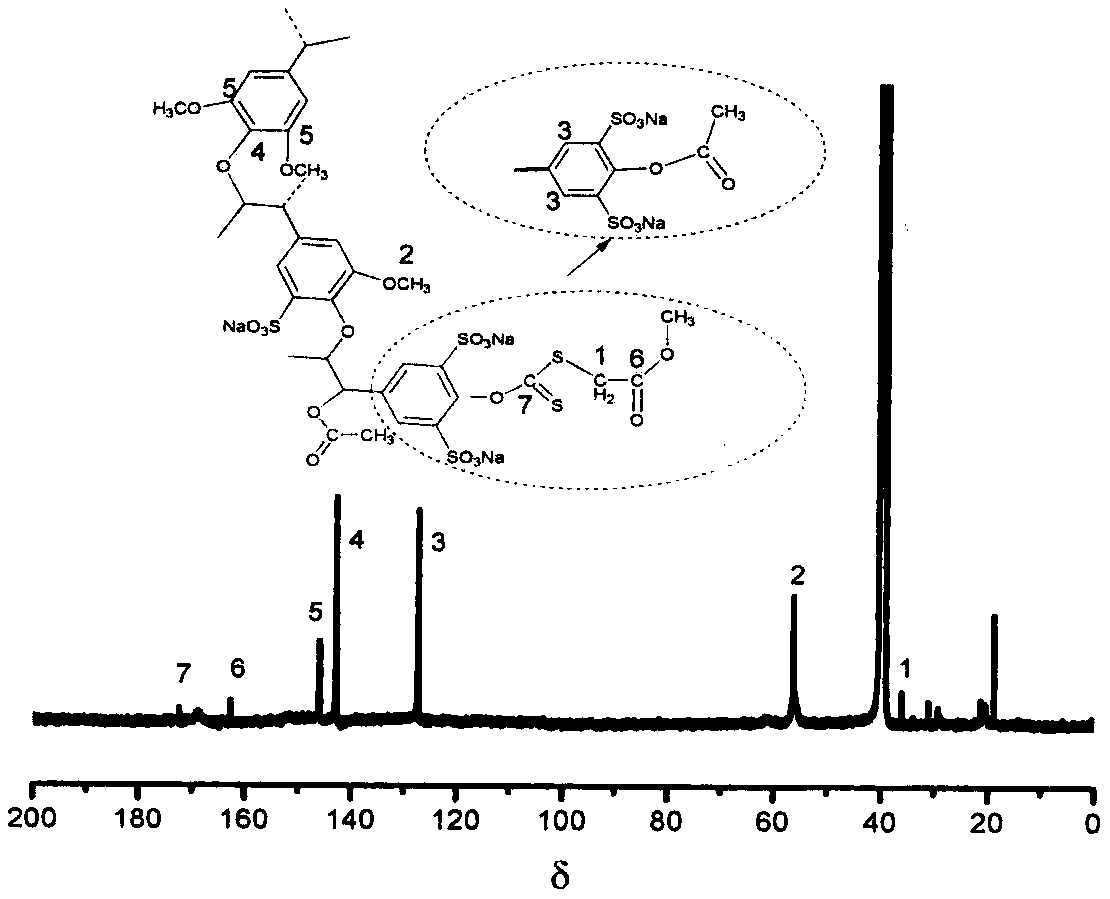
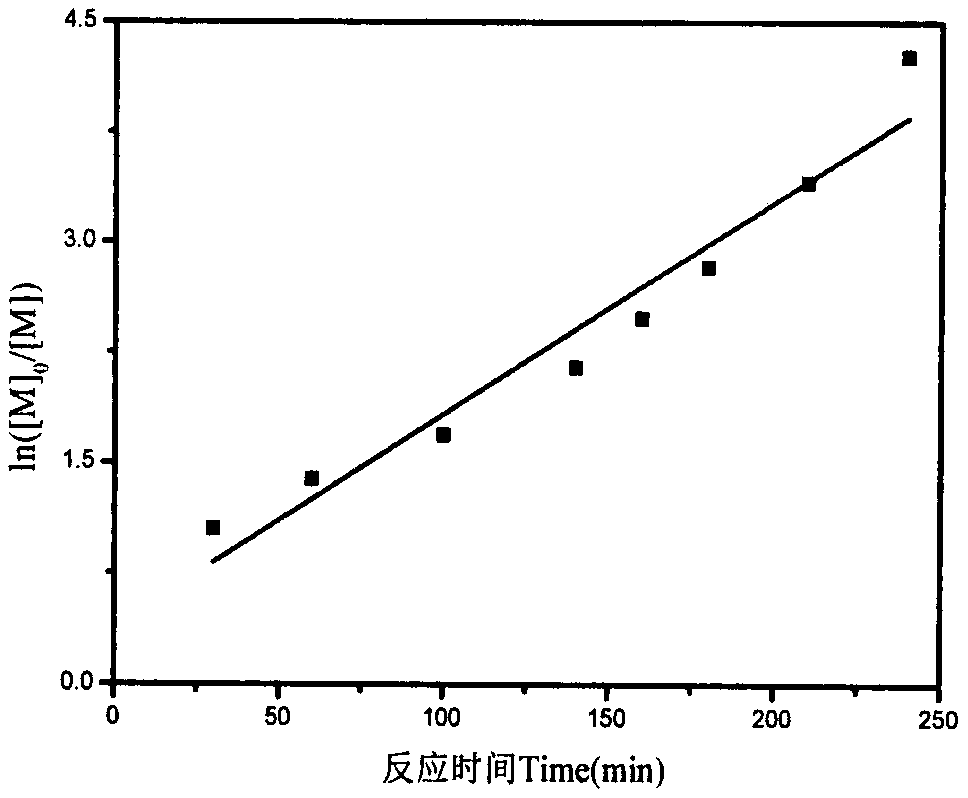
Water-soluble lignin base chain transferring agent and preparation method thereof
The invention relates to a water-soluble lignin base chain transferring agent and a preparation method thereof. The water-soluble lignin base chain transferring agent comprises the following steps: (1) sulfonating lignin by treating trichloromethane as a solvent and chlorosulfonic acid as a sulfonating agent; and (2) feeding sulfonated lignin and an alkaline solution into a reaction bottle to fully solve; dropwise adding CS2 to the bottle; reacting for 4-8h; dropping bromo ester to continuously react for a period of time; precipitating and washing a product; and drying to obtain the water-soluble lignin base chain transferring agent which is high in water soluble performance. According to the method, the problem of low solubility of a lignin product in a water solution can be released; andmoreover, the operation is convenient and easy to carry out; the cost is low; and the prepared water-soluble lignin base chain transferring agent is high in polymerizing controllability in polymerizing reaction.
Owner:INST OF CHEM IND OF FOREST PROD CHINESE ACAD OF FORESTRY
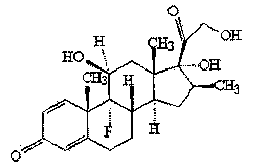
Preparation method of betamethasone intermediate
Owner:HUNAN NORCHEM PHARMACEUTICAL CO LTD
A kind of preparation method of tatirelin and its intermediate
Owner:重庆莱美隆宇药业有限公司
A kind of preparation method of phenylcyclopentyl ketone
ActiveCN105753682BReduce pollutionIncrease generation costOrganic compound preparationPreparation from carboxylic acid esters/lactonesKetoneMethyl acetate
Owner:CHENGDU ORGANIC CHEM CO LTD CHINESE ACAD OF SCI +1
Features
- R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
Why Patsnap Eureka
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Social media
Patsnap Eureka Blog
Learn More Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com