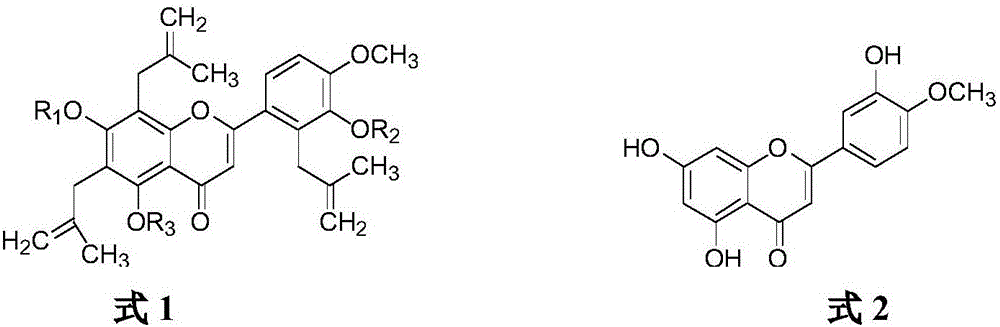Patents
Literature
46 results about "Diosmetin" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
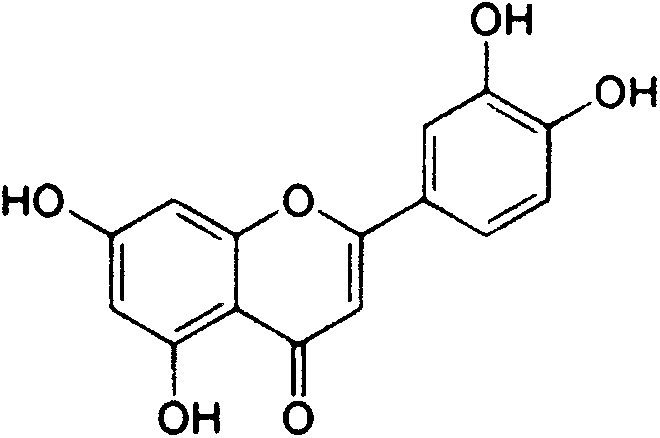
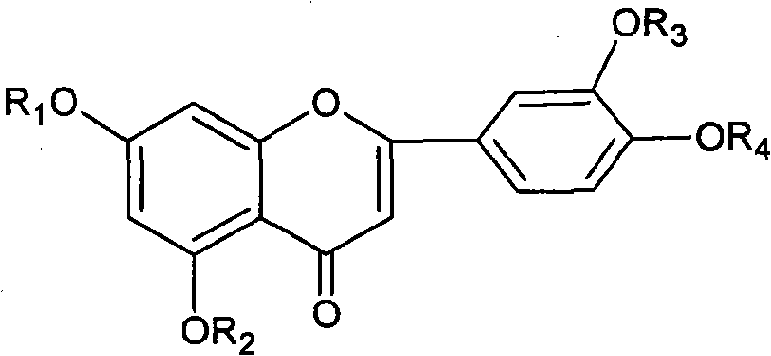
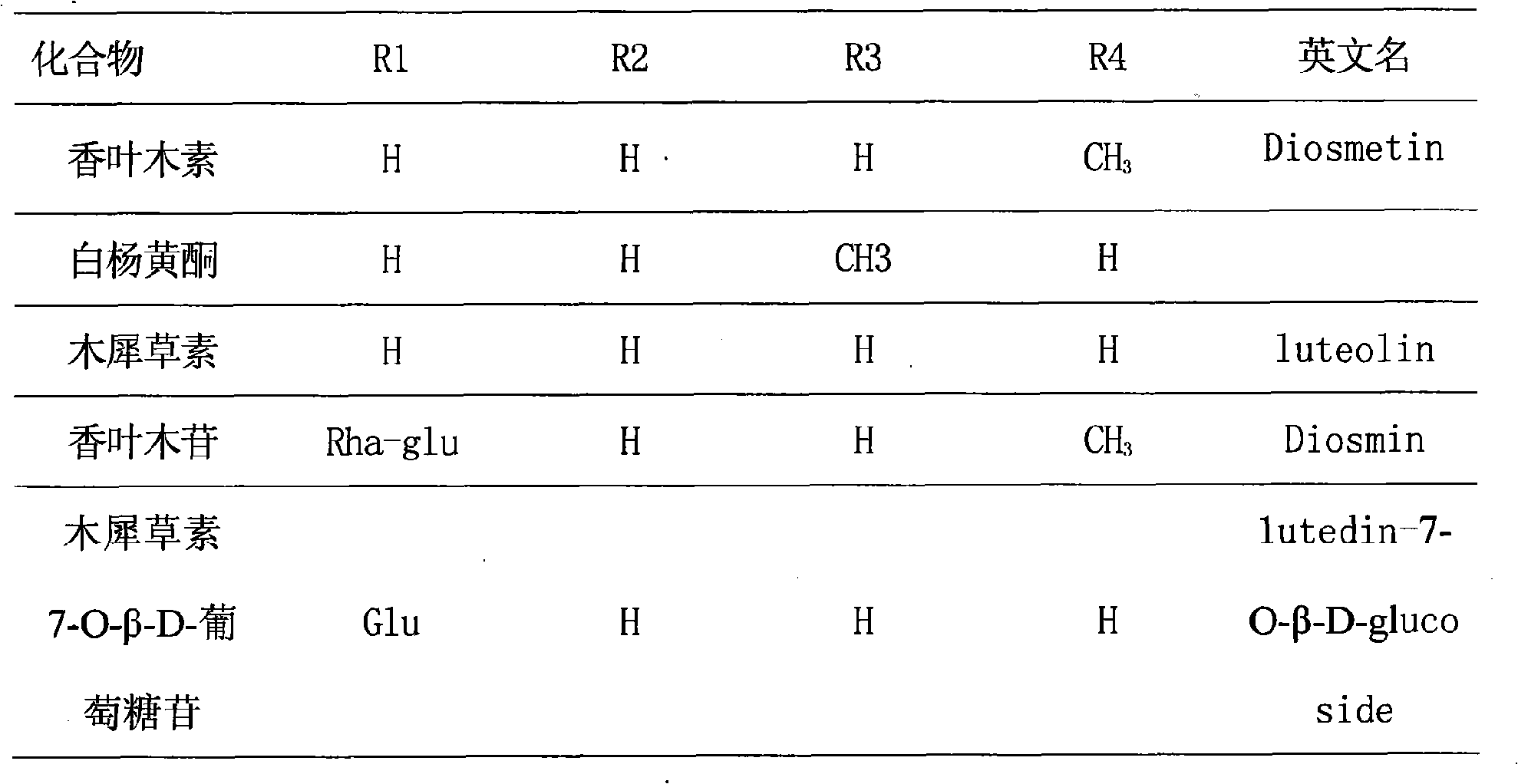
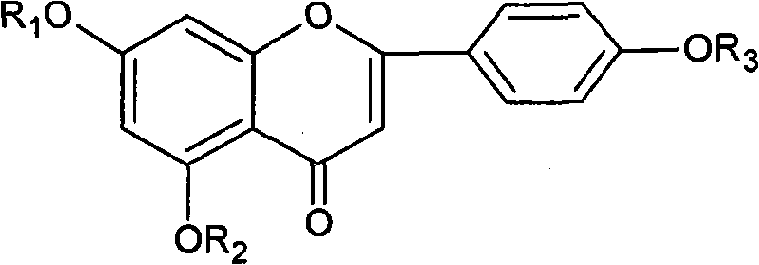
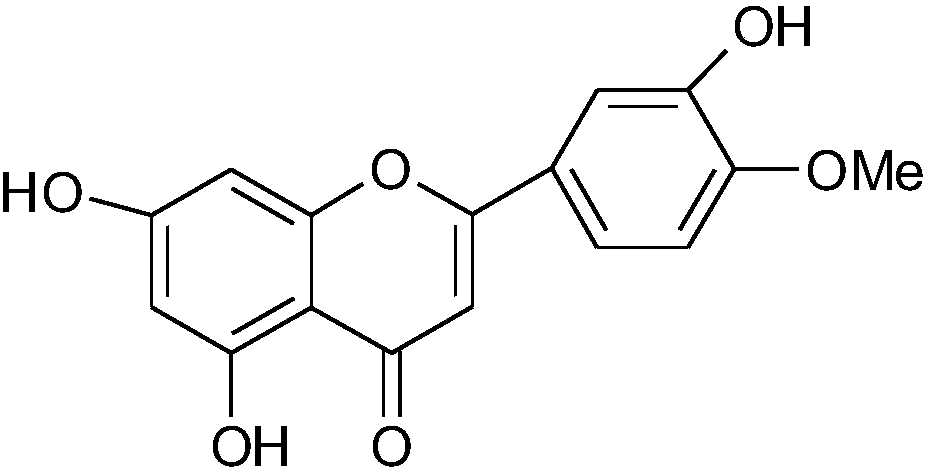
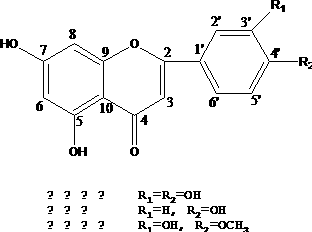
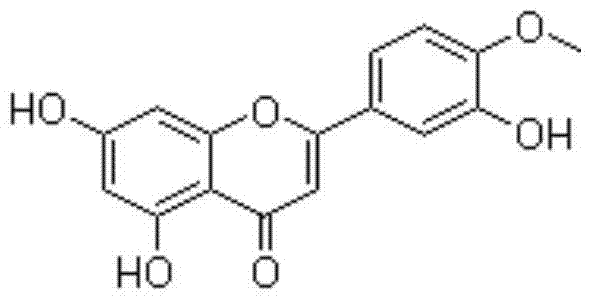
Diosmetin, also known as 5,7,3'-trihydroxy-4'-methoxyflavone, is an O-methylated flavone, a chemical compound that can be found in the Caucasian vetch. It has been found to act as a weak TrkB receptor agonist.
New technology for preparing luteolin by using hesperidin
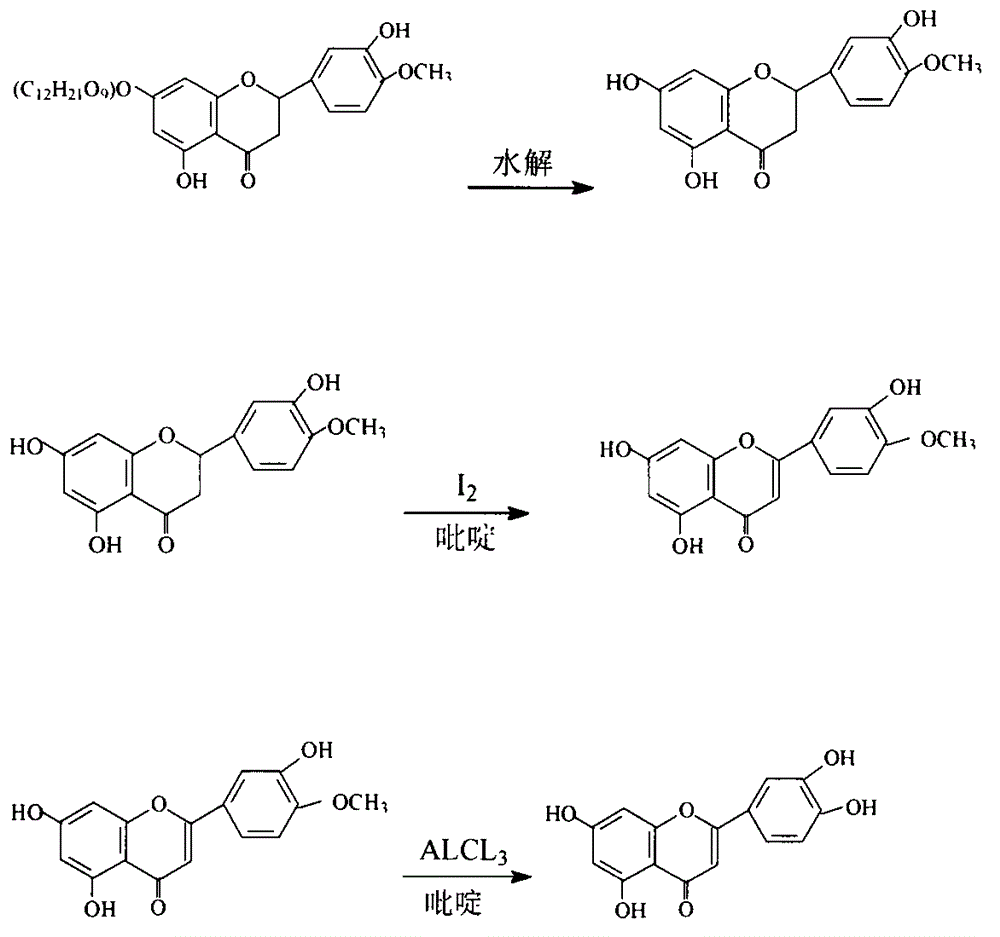
The invention provides new technology for preparing luteolin by using hesperidin. The method includes a first step of hydrolyzing hesperidin to obtain hesperetin, a second step of dehydrogenizing the hesperetin to obtain diosmetin, and a third step of demethylating the diosmetin to obtain the luteolin. Compared with the prior art that the hesperidin is firstly dehydrogenized and then hydrolyzed, the method enables iodine using amount to be greatly reduced, and production cost is low. The invention further provides new technology for preparing the diosmetin.
Owner:李玉山
Radical lobelia total flavones effective component and preparation thereof
The present invention discloses an effective component of total flavonoid extracted from Chinese medicine Chinese lobelia and a preparation method thereof. The preparation material mainly contains apigenin, luteolin, diosmetin, chrysin flavonoid, linarin, dafion, and indican compounds and other derivatives with the dafion as the mother nucleus. The present invention can be obtained by the methods of macroporous absorption resin, polyamide chromatography, solvent extraction and other methods.
Owner:石任兵
Method for preparing mono-glucoside through selective hydrolysis of flavone rutinoside or neohesperidoside
InactiveCN103819520AEasy to operateLow costSugar derivativesSugar derivatives preparationOrganosolvKetone
The invention relates to a method for preparing mono-glucoside through selective hydrolysis of flavone rutinoside or neohesperidoside, and belongs to the fields of chemistry and medicine. The method comprises the following steps: mixing flavone rutinoside or neohesperidoside with macroporous absorption resin in water or alkaline water solution, heating or adding acid; wherein in this process the diglucoside is dispersed, absorbed, and cured by the macroporous absorption resin; after drying, in the catalysis of acid or a dewatering agent, rhamnose in the molecule carries out condensation reactions with a ketone agent, and the rhamnose in the molecule is removed in the action of acid; eluting with an organic solvent or an alkaline water solution; and recycling the solvent or acidifying so as to obtain the flavone mono-glucoside. The method solves the problems that flavone diglucoside is hard to disperse in a ketone reagent and the selectivity of rhamnose hydrolysis is low. The method has the advantages of simple operation, easy recycling of organic solvent, low cost, high yield, and easiness in industrial production. Compounds such as hesperetin mono-glucoside, diosmetin mono-glucoside, hesperetin dihydrochlcone mono-glucoside, and the like, can be industrially produced from flavones such as flavone neohesperidoside through the method.
Owner:闻永举
Method for separating and purifying three flavonoid glycosides from trichosanthes bark
ActiveCN103304611ASimple compositionNot pollutedSugar derivativesSugar derivatives preparationFluid phaseTrichosanthes
The invention relates to a method for separating and purifying three flavonoid glycosides from trichosanthes bark. According to the method, three high-purity flavonoid glycosides, namely quercetin-3-O-beta-rutinoside, apigenin-7-O-beta-glucoside and diosmetin-7-O-beta-glucoside, are obtained from the trichosanthes bark through the following steps of: (1) preparing a crude trichosanthes bark extract; (2) extracting; (3) carrying out crude separation by using a polyamide column; and (4) carrying out separation and purification by using semi-preparative high-performance liquid chromatography: carrying out separation and purification on total flavonoid glycosides of the trichosanthes bark by using the semi-preparative high-performance liquid chromatography, wherein the mobile phase is methanol-water. The method disclosed by the invention has the advantages that the process flow is environment-friendly, the damage to the environment is not serious, and the comprehensive cost is low.
Owner:SHANDONG UNIV OF TRADITIONAL CHINESE MEDICINE +1
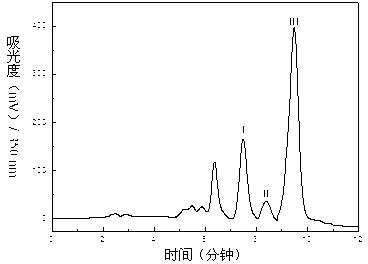
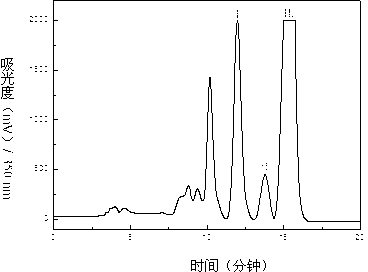
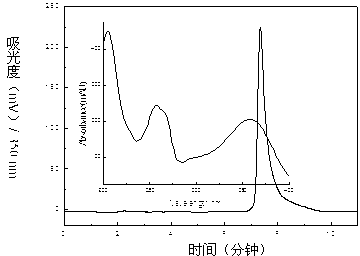
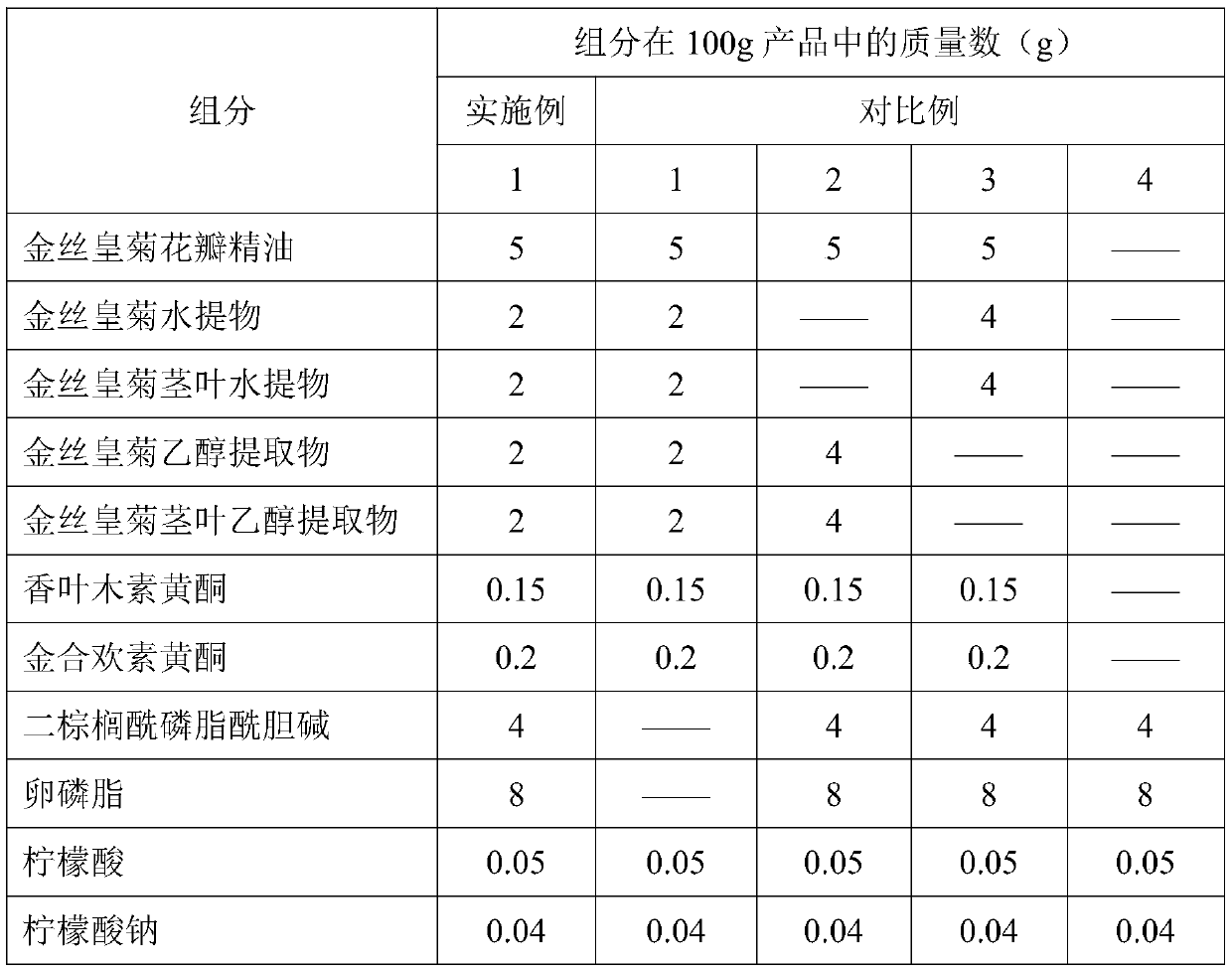
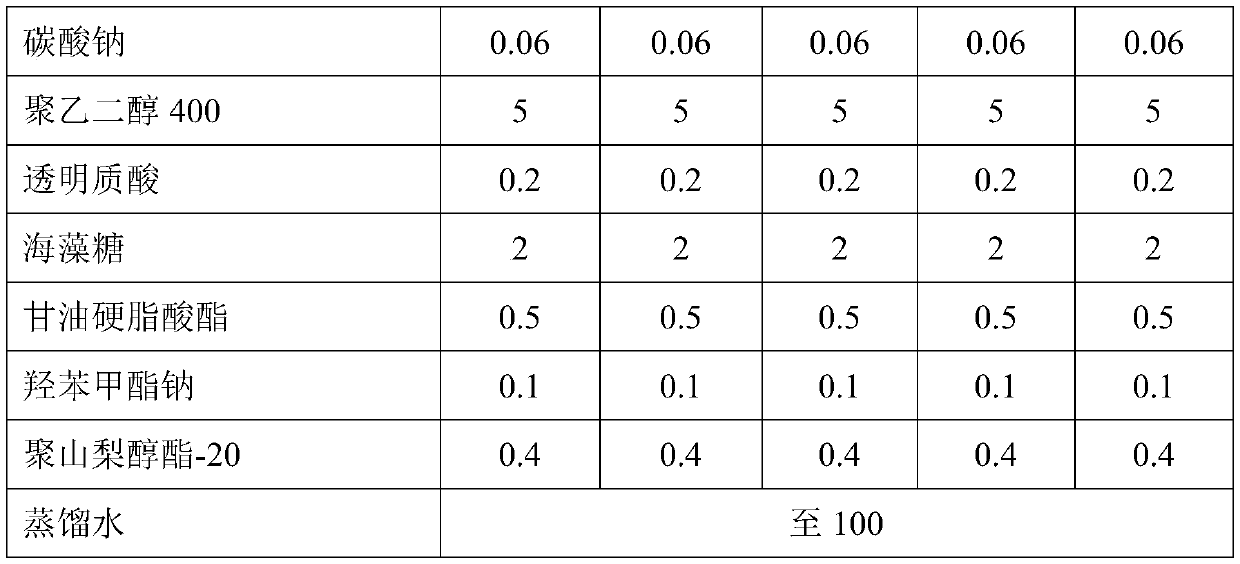
Dendranthema morifolium nanotechnology skin care product and preparation method and application thereof
ActiveCN111481481AUltimate skin care effectOptimal Screening of Nanotechnology Process ParametersCosmetic preparationsAntipyreticAcacetinPolyethylene glycol
The invention relates to a Dendranthema morifolium nanotechnology skin care product which is composed of the following raw materials: Dendranthema morifolium petal essential oil, a Dendranthema morifolium aqueous extract, a Dendranthema morifolium alcohol extract, a Dendranthema morifolium stem and leaf aqueous extract, a Dendranthema morifolium stem and leaf alcohol extract, and diosmetin and acacetin flavones separated from Dendranthema morifolium; dipalmitoyl phosphatidylcholine, lecithin, polyethylene glycol 400, hyaluronic acid, trehalose, glyceryl stearate, sodium methyl hydroxybenzoate,polysorbate-20, citric acid, sodium citrate, sodium carbonate and distilled water supplemented to 100 parts. According to the nanotechnology skin care product added with multiple active ingredients of Dendranthema morifolium, multiple components and proportions are reasonably and scientifically designed, nanotechnology process parameters are optimized and screened, and the skin care effect of Dendranthema morifolium is brought into play to the maximum extent.
Owner:江苏灵源沂岸科技股份有限公司
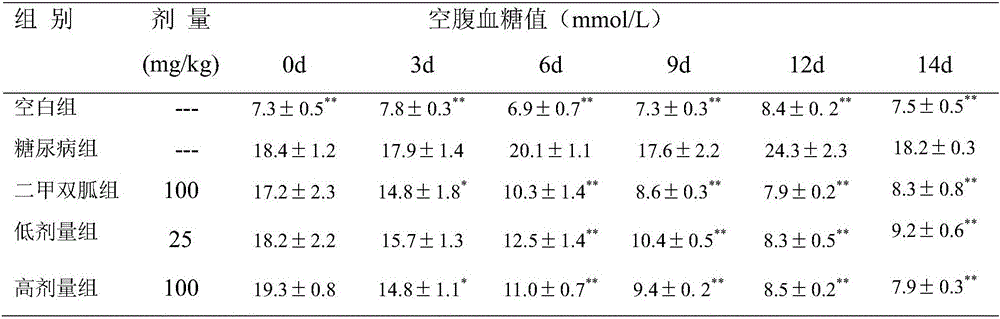
Application of diosmetin in preparation of medicine used for treating type 2 diabetes mellitus
InactiveCN106822087AExpand the scope of useImprove the level ofOrganic active ingredientsMetabolism disorderLevel insulinGlucose lowering
The invention relates to application of diosmetin in preparation of a medicine used for treating type 2 diabetes mellitus. Researches show that diosmetin can obviously improve glycometabolism of the type 2 diabetes mellitus, can improve insulin level and lower blood lipid level, can be used for preparing a blood-glucose-lowering medicine and provides a new medicine for treatment of the type 2 diabetes mellitus, thereby having significance on the treatment of diabetes mellitus.
Owner:SOUTHWEST UNIVERSITY
Recombinant escherichia coli and method for biosynthesizing diosmetin by utilizing recombinant escherichia coli
ActiveCN112725256AIncreased expression of solubleHigh catalytic activityBacteriaMicroorganism based processesEscherichia coliOrganosolv
The invention discloses recombinant Escherichia coli and a method for biosynthesizing diosmetin by using the recombinant Escherichia coli, the recombinant Escherichia coli is obtained by introducing a recombinant plasmid into host Escherichia coli, and the recombinant plasmid is constructed by connecting an encoding gene of AnFNSI after codon optimization and an expression vector pET-28a (+); The AnFNSI is flavone synthase 1 derived from angelica archang lica, the amino acid sequence of the AnFNSI is as shown in SEQ ID NO.1, and the amino acid sequence of the AnFNSI after codon optimization is as shown in SEQ ID NO.4. The recombinant Escherichia coli is used as a whole-cell catalyst to catalyze a substrate hesperetin to biosynthesize diosmetin. The method is green and efficient, does not need to use a large amount of organic solvents or toxic chemical reagents in the reaction process, and is low in cost, high in safety and easy for large-scale production.
Owner:HUNAN AGRI PRODS PROCESSING INST
Diosmetin having anti-inflammatory activity and application of diosmetin in preparation
The invention relates to a diosmetin having anti-inflammatory activity and application of the diosmetin in preparation of anti-inflammatory drugs. The diosmetin can remarkably inhibit inflammations caused by a free radical medium and a lipid medium and can be applied to the preparation of the anti-inflammatory drugs.
Owner:SOUTHWEST UNIVERSITY
Preparation method of corn silk flavonoid, and product and application thereof
ActiveCN105497050AReduce extraction timeImprove efficiencyOrganic active ingredientsAntipyreticChlorogenic acidCorn silk
The invention discloses a preparation method of corn silk flavonoid. The preparation method comprises the following steps of extraction of the corn silk flavonoid and purification of the corn silk flavonoid, wherein the extraction method of the corn silk flavonoid comprises the steps: washing, drying, smashing and sieving corn silks; extracting the corn silk flavonoid by taking an ethanol solution as an extract solvent through microwave assisted extraction, wherein the ethanol concentration of the extract solvent is 40 to 60 percent, the extract time is 15 to 25 minutes, a material-to-liquid ratio is 1:45 to 1:25, the temperature is 60 to 80 DEG C, and the extract power is 400 to 500 W; obtaining a corn silk flavonoid crude extract by drying an extract solution; preparing the corn silk flavonoid crude extract into a solution of which the concentration is 1.0 to 1.5 mg / mL and using AB-8 macroporous resin to do the absorption, wherein the adsorption time is 2 hours; eluting by using 4BV of 55 to 65% ethanol solution, wherein the eluting flow rate is 1.5 to 2 mL / min; obtaining a purified corn silk flavonoid product through concentrating and drying, wherein the purified corn silk flavonoid product contains apigenin, diosmetin, luteolin, chlorogenic acid and rutin. The preparation method for extracting the corn silk flavonoid, disclosed by the invention, has the advantages of simpleness and efficiency, and the extracted corn silk flavonoid has a remarkable anti-inflammation effect and can be applied to development of anti-inflammation medicine and healthcare products.
Owner:JILIN INST OF CHEM TECH
Semi-synthesis method of diosmetin
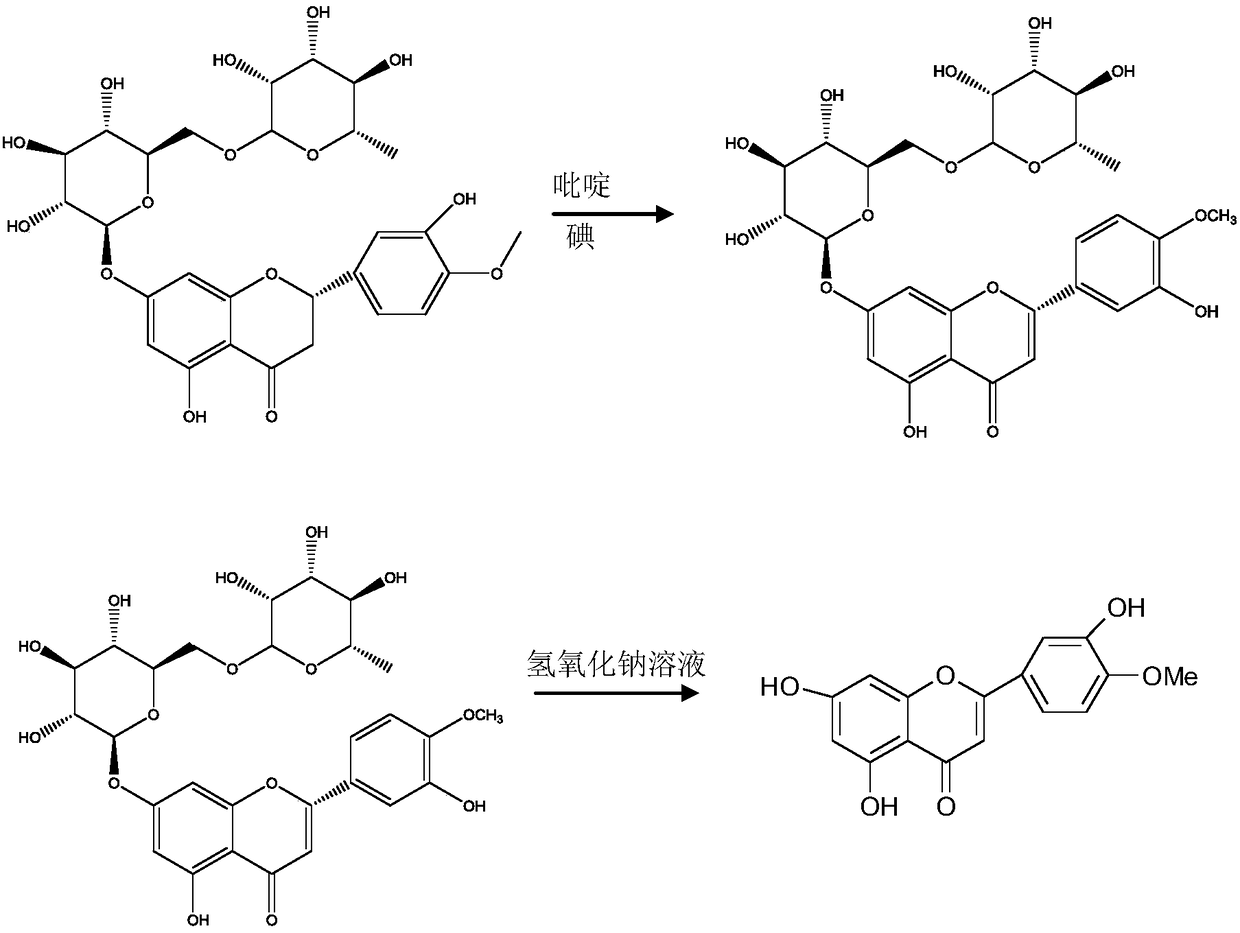
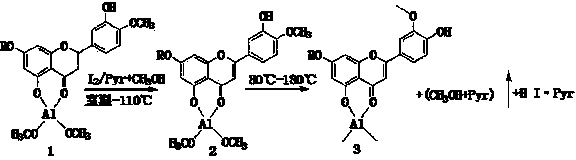
The invention provides a semi-synthesis method of diosmetin. A purpose of the present invention is to solve the problems of high production cost and low synthesis efficiency of the existing synthesismethod. According to the present invention, neohesperidin, pyridine and iodine are heated and react to prepare neodiosmin, and the neodiosmin is hydrolyzed with a sodium hydroxide solution to preparediosmetin; and the semi-synthesis method has characteristics of short process time, simple post-treatment, low production cost, high product purity and high yield, and is suitable for the industrial production of diosmetin.
Owner:SHAANXI JIAHE PHYTOCHEM
Method for separation and purification of luteolin, apigenin and diosmetin in trichosanthes peel
The invention relates to a method for separation and purification of luteolin, apigenin and diosmetin in trichosanthes peel. The method utilizes trichosanthes peel as a raw material and comprises the following steps of 1, preparing trichosanthes peel crude extract, 3, carrying out crude separation by a polyamide column, and 4, carrying out separation purification by semi-preparation high performance liquid chromatography. The method is green and environmentally friendly, does not produce damage on the environment and has a low comprehensive cost.
Owner:SHANDONG UNIV OF TRADITIONAL CHINESE MEDICINE +2
Method for separating and purifying diosmetin
InactiveCN102304113AReduce energy consumptionReduce pollutionOrganic chemistryBulk chemical productionReflux extractionAlcohol
The invention discloses a method for separating and purifying diosmetin. The method comprises the following steps of: taking peppermint pyrrosia petiolosa, from which volatile oil is extracted, as a raw material; adding the peppermint pyrrosia petiolosa into a CO2 supercritical extraction tank to extract; obtaining extract; adsorbing the extract by using aqueous solution through macro-porous adsorption resin; eluting an alcohol-water solution; collecting eluent; decompressing; concentrating; carrying out level reflux extraction on a concentrated solution by using a mixed solvent; mixing extraction solutions; decompressing and recovering the solvent; adding; crystallizing; and drying. The diosmetin is separated and purified by using the method disclosed by the invention. The industrial scale is easy realized. The content of the product is high.
Owner:NANJING ZELANG MEDICAL TECH
Semi-synthesis method of luteolin and galuteolin as well as luteolin rutinoside
InactiveCN103833714AAvoid absorptionQuick joinSugar derivativesSugar derivatives preparationDehydrogenationHydrolysis
The invention relates to a semi-synthesis method of luteolin and galuteolin as well as luteolin rutinoside by using hesperidin, a semi-synthesis method of the galuteolin by using hesperetin glucoside, and a semi-synthesis method of the luteolin by using hesperetin, and belongs to the field of chemistry and medicines. The semi-synthesis method comprises the following steps: enabling the hesperidin, the hesperetin glucoside and the hesperetin to be subjected to complexation in pyridine alcohol fluid, dehydrogenizing by using iodine, directly distilling alcohol and pyridine, maintaining for a period of time in an airtight distilled state, and carrying out demethylation reaction, so that the hesperidin is generated into the luteolin rutinoside; generating the galuteolin by demethylation of diosmetin glucoside, and generating the luteolin by diosmetin; and hydrolyzing the luteolin rutinoside, so that the luteolin rutinoside is transformed into luteolin and galuteolin. The semi-synthesis method has the advantages that two steps of dehydrogenation and demethylation are combined into one step, and reaction conditions of dehydrogenation and demethylation are mild and easy to control; few reagents are used and green and environmentally friendly; and the demethylation yield is high, and the industrial production is easy. Compared with disclosed documents and patients, the semi-synthesis method has great advantages in the production of luteolin and glucosides of the luteolin.
Owner:迁西县板栗产业研究发展中心
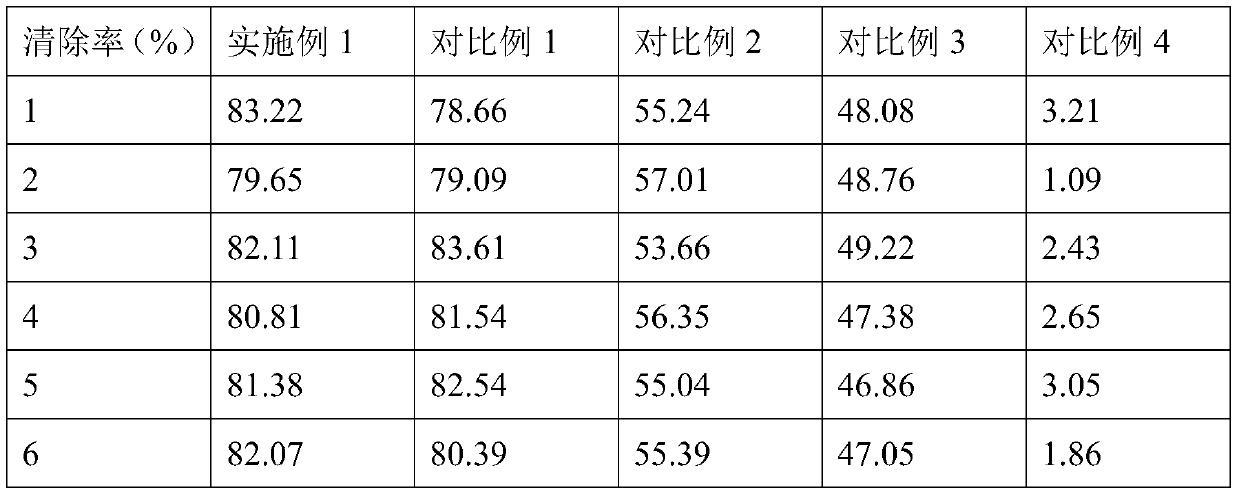
Composition with blood sugar reduction function and application
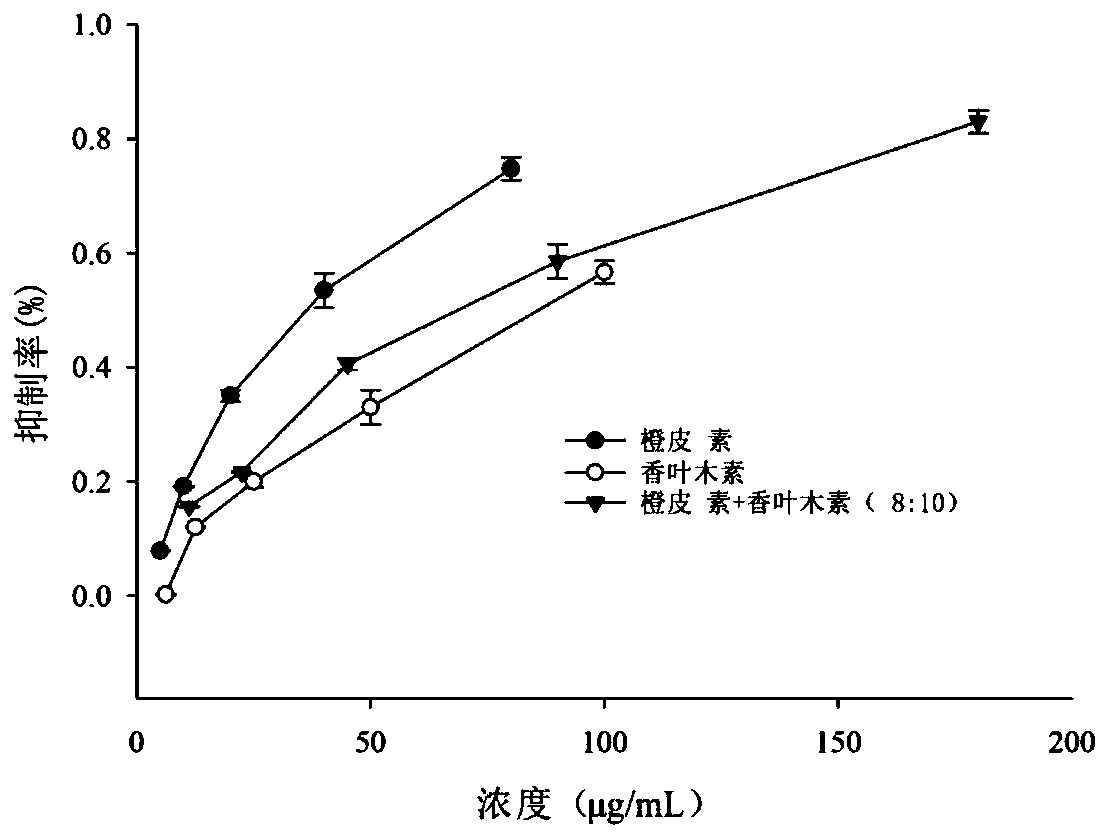
ActiveCN111388462AGood synergySmall doseOrganic active ingredientsMetabolism disorderPharmaceutical drugDiosmetin
The invention discloses a composition with a blood sugar reduction function and application and belongs to the technical field of medicines. The composition with the blood sugar reduction function consists of hesperetin and diosmetin in a mass concentration ratio of (2:10)-(8-10), and preferably the mass concentration ratio of the hesperetin to the diosmetin is (3:10)-(8:10). The composition of the hesperetin and the diosmetin, which is disclosed by the invention, has a remarkable synergism in inhibiting alpha-glucosidase, has an effect prior to those of separately used flavonoids, in addition, is capable of reducing dosages of medicines and reducing drug resistance, and has wide application prospects in preparing medicines, healthcare products or foods for treating diabetes.
Owner:ZHENGZHOU FRUIT RES INST CHINESE ACADEMY OF AGRI SCI
Medicine composition for treating hyperplasia of mammary glands and preparation method thereof
InactiveCN105456570AGood treatment effectEther/acetal active ingredientsUnknown materialsPiper wallichiiMammary gland structure
The invention discloses a medicine composition for treating the hyperplasia of mammary glands and a preparation method thereof. The medicine composition is prepared from 20-30 parts of traditional Chinese medicine preparation, 5-10 parts of folium ginkgo extract, 1-5 parts of chondroitin sulfate, 0.1-0.3 parts of pterostilbene and 0.1-1 part of diosmetin. The traditional Chinese medicine preparation is prepared from folium cycas, Japanese ardisia, dysosma versipellis, fiveleaf akebia fruit, pseudo-ginseng, piper wallichii, purple perilla and scorpions according to the weight ratio of (3-5):(2-6):(2-5):(1-3):(1-3):(3-5):(1-2):(0.1-0.5) through decocting, filtering and concentrating. The medicine composition has functions of promoting blood circulation to remove blood stasis, stimulating menstrual flow to remove stasis, dredging channels by soothing liver, dredging latex vessels, promoting qi circulation to relieve pain, expelling wind and relieving muscular spasm, clearing away heat to remove blood stasis and the like, and has an excellent effect on treating the hyperplasia of mammary glands.
Owner:THE AFFILIATED HOSPITAL OF QINGDAO UNIV
Extraction and assay method of total flavones of buchnera cruciata
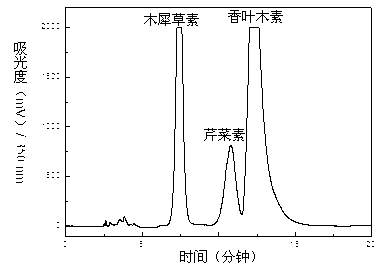
The invention discloses an extraction and assay method of total flavones of buchnera cruciata. The total flavones of buchnera cruciata is extracted with a column filled with D101 macroporous resin in a manner of ethanol elution; with rutin as a reference substance, sodium nitrite, aluminum nitrate and sodium hydroxide are added to measure light absorbency to successively determine the content of the total flavones; the content of luteolin, apigenin and diosmetin in the total flavones of buchnera cruciata are detected through HPLC, wherein acetonitrile-0.1% phosphoric acid is a mobile phase, Agilent ZORBAX Eclipse XDB-C18 is a chromatographic column, and detection wave length is 350 nm. The total flavones of buchnera cruciata have the effects of anticoagulation, inflammation resistance, sedation and allergy resistance, and are low in toxicity. The extraction and assay method provides reference data for development on medicinal value of the total flavones of buchnera cruciata.
Owner:GUANGXI INST OF CHINESE MEDICINE & PHARMA SCI
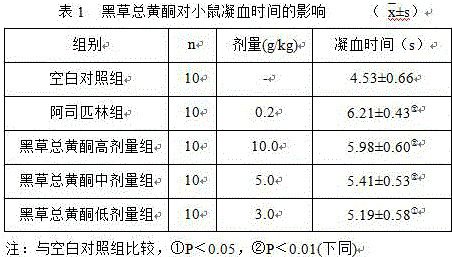
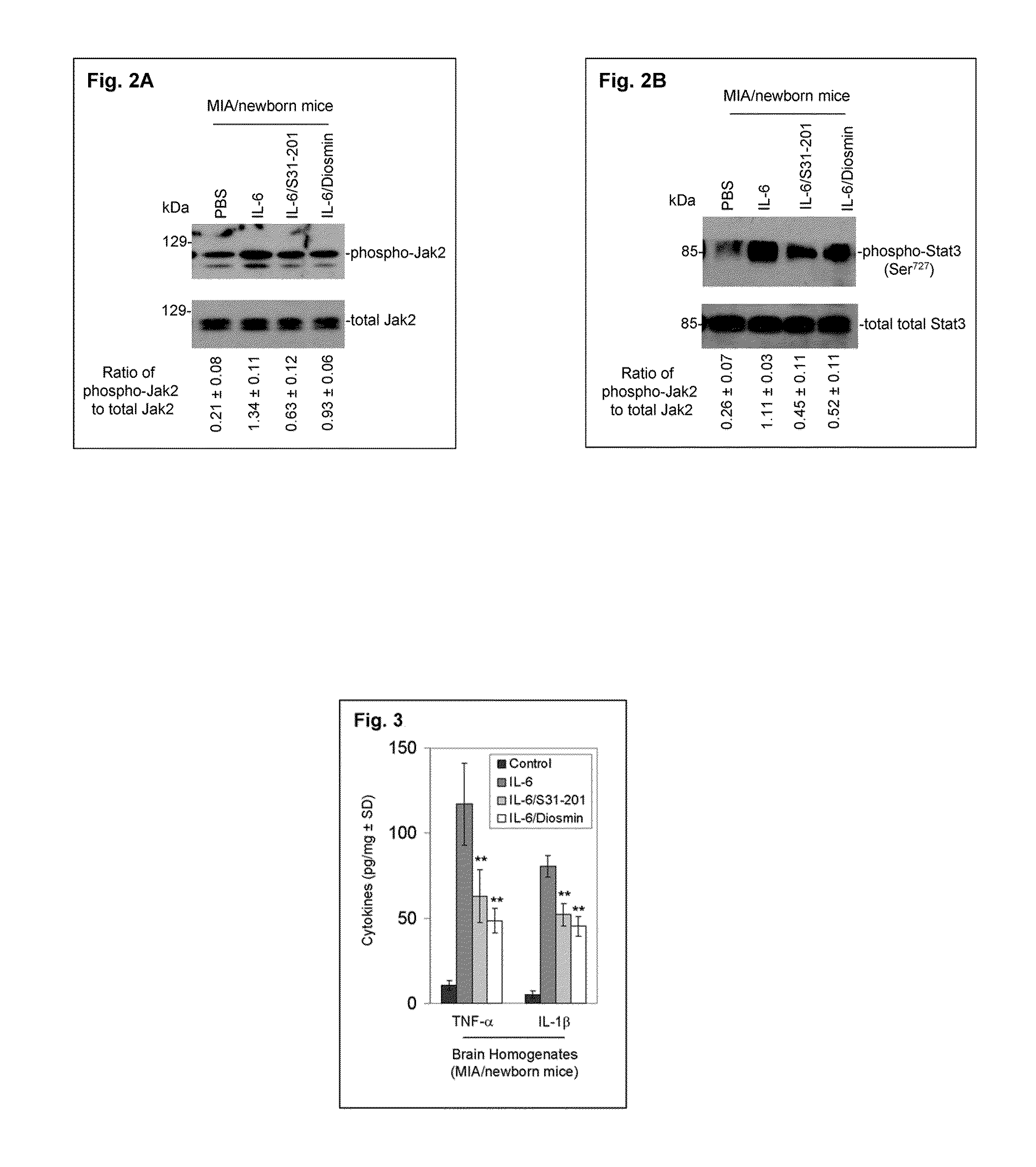
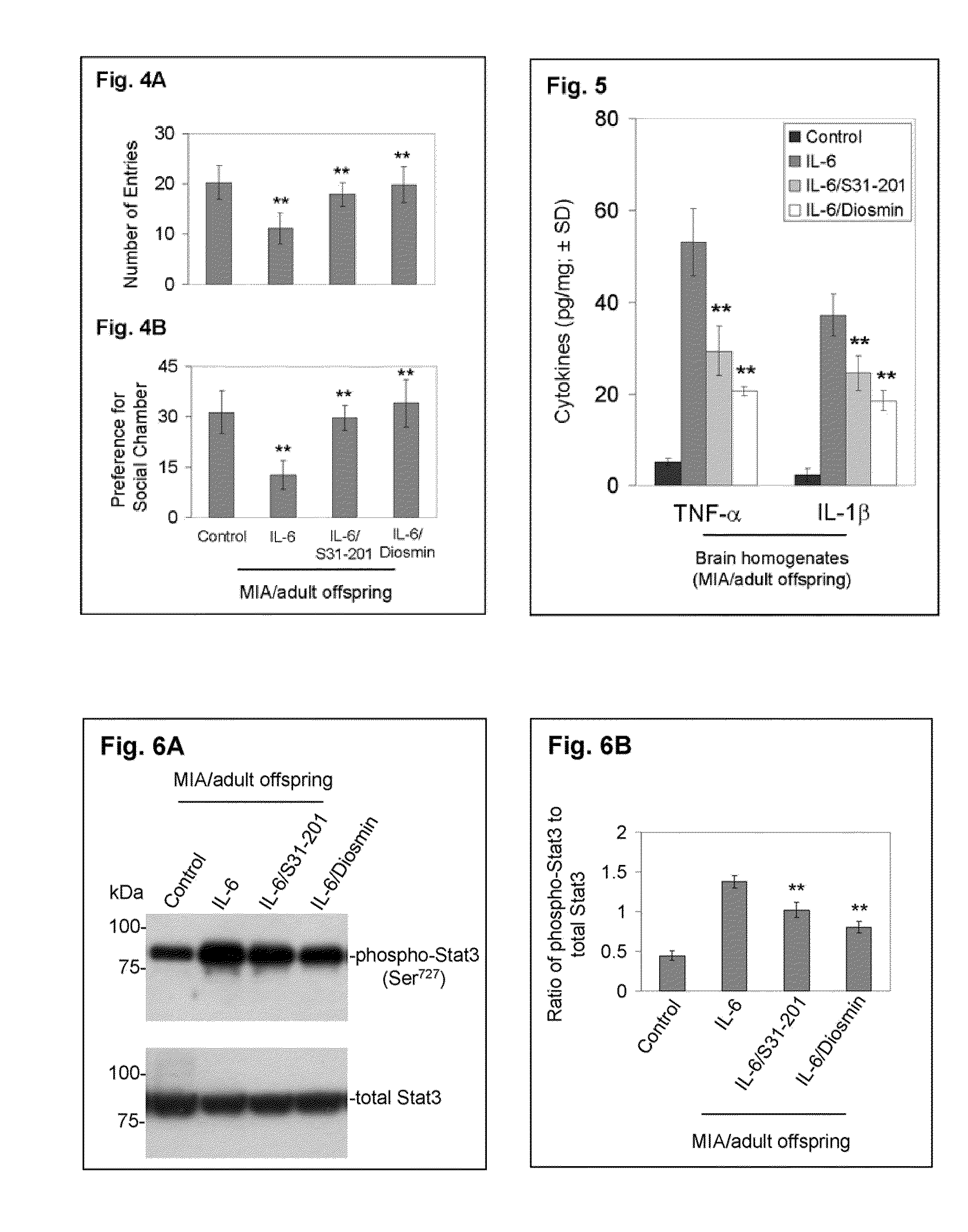
Luteolin and diosmin/diosmetin as novel stat3 inhibitors for treating autism
The present invention includes methods for the treatment of autoimmune disorders such as autism, schizophrenia, and type 1 diabetes. Flavonoids, luteolin, diosmin, and diosmin's aglycone form, diosmetin, were found to inhibit activation / phosphorylation of STAT3 induced by IL-6 in cultured neuronal cells. Furthermore, mice treated with diosmin showed a significant reduction of autistic phenotype induced by IL-6 through inhibition of STAT3 activation.
Owner:UNIV OF SOUTH FLORIDA
Compound anti-oxidization anti-gelling agent for oil product and preparation method of compound anti-oxidization anti-gelling agent
ActiveCN105219456AImprove performancePrevent discolorationLiquid carbonaceous fuelsPhosphoric Acid EstersPolymer science
The invention relates to a compound anti-oxidization anti-gelling agent for an oil product and a preparation method of the compound anti-oxidization anti-gelling agent and belongs to the technical field of oil gas additives. The compound anti-oxidization anti-gelling agent for the oil product is prepared from the following components in parts by weight: 1 to 2 parts of butylated hydroxyanisole, 0.5 to 1.5 parts of alkyl glycoside, 2 to 5 parts of stearic acid, 0.1 to 0.2 parts of triethylene glycol bi[beta-(3-tertiary butyl-4-hydroxy-5-methyl phenyl)propionate, 0.1 to 0.2 parts of bi(2,4-bitertiary butyl phenyl)pentaerythritol biphosphite, 1 to 2 parts of diosmetin, 1 to 3 parts of nano calcium carbonate, 15 to 19 parts of ethanol and 5 to 10 parts of glycerol. The compound anti-oxidization anti-gelling agent for the oil product has good stability; the performance of the compound anti-oxidization anti-gelling agent is not influenced after the compound anti-oxidization anti-gelling agent is stored for a long period; the oil product is prevented from color change when the oil product is stored; original colloid in the oil product can be completely removed and the formation of colloids is inhibited; the quality of the oil product is effectively improved.
Owner:重庆索银新能源科技有限公司
Application of diosmetin(4-O-methyl) glucoside compound in preparation of lipid-lowering drugs
ActiveCN112358516APromote proliferationStrong lipid-lowering activityOrganic active ingredientsSugar derivativesCell Differentiation processAglycone
The invention discloses an application of a diosmetin(4-O-methyl) glucoside compound as a lipid-lowering drug; in a biological activity test, the compound shows down-regulation or inhibition of mouse3T3-L1 precursor adipocyte proliferation and significant reduction of the number of lipid droplets generated in a cell differentiation process, and aglycones of the diosmetin(4-O-methyl) glucoside compound also have the functions, but the effect of the compound is more obvious; compared with the aglycones, the compound has the advantages that the water solubility of the compound is remarkably enhanced, the conversion rate of the compound is high, and the compound has wider application value.
Owner:INST OF CROP SCI CHINESE ACAD OF AGRI SCI
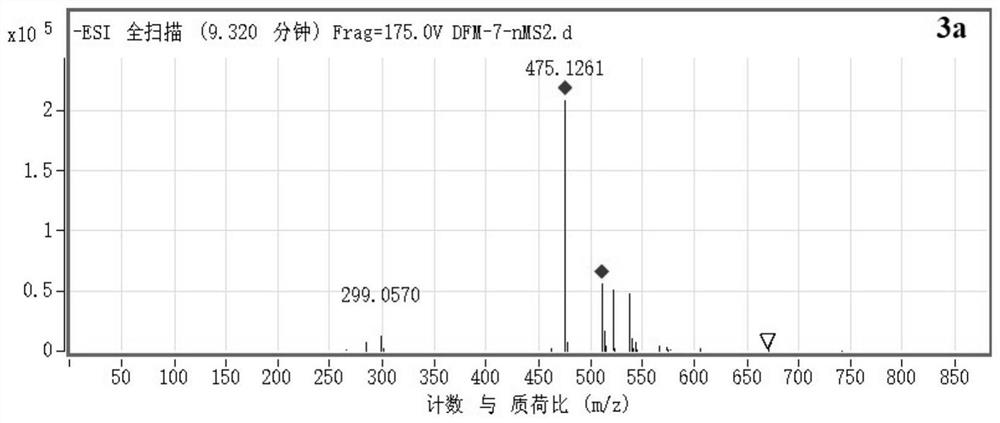
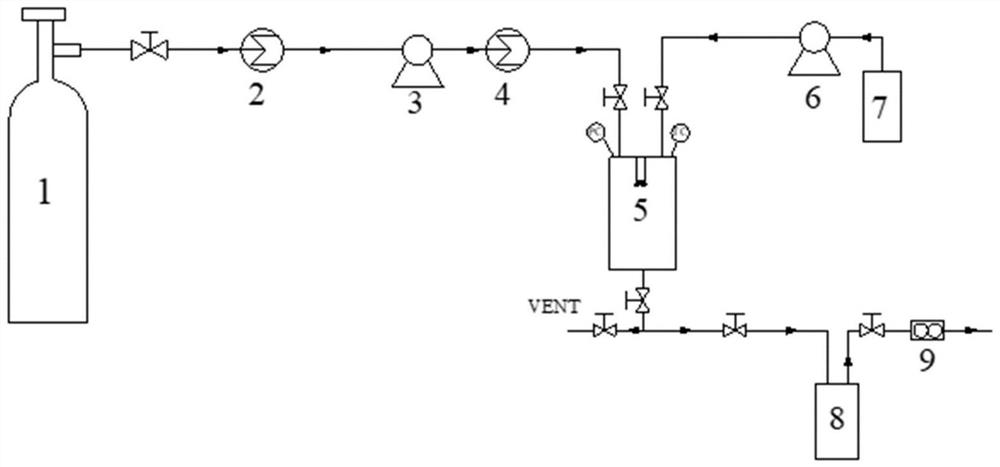
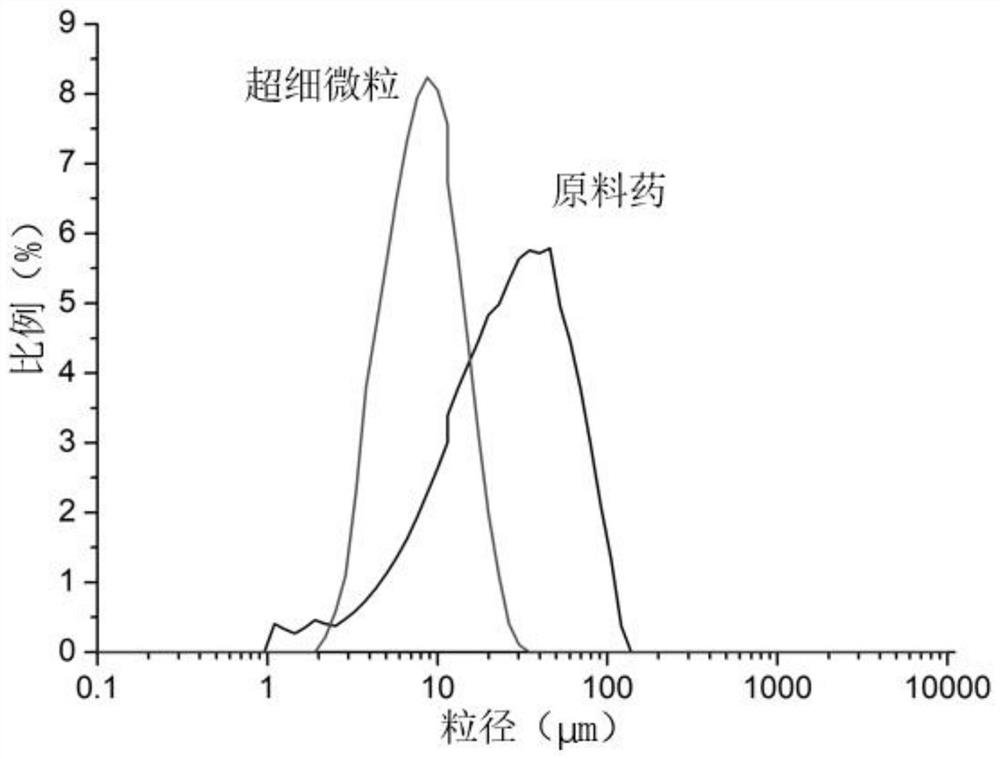
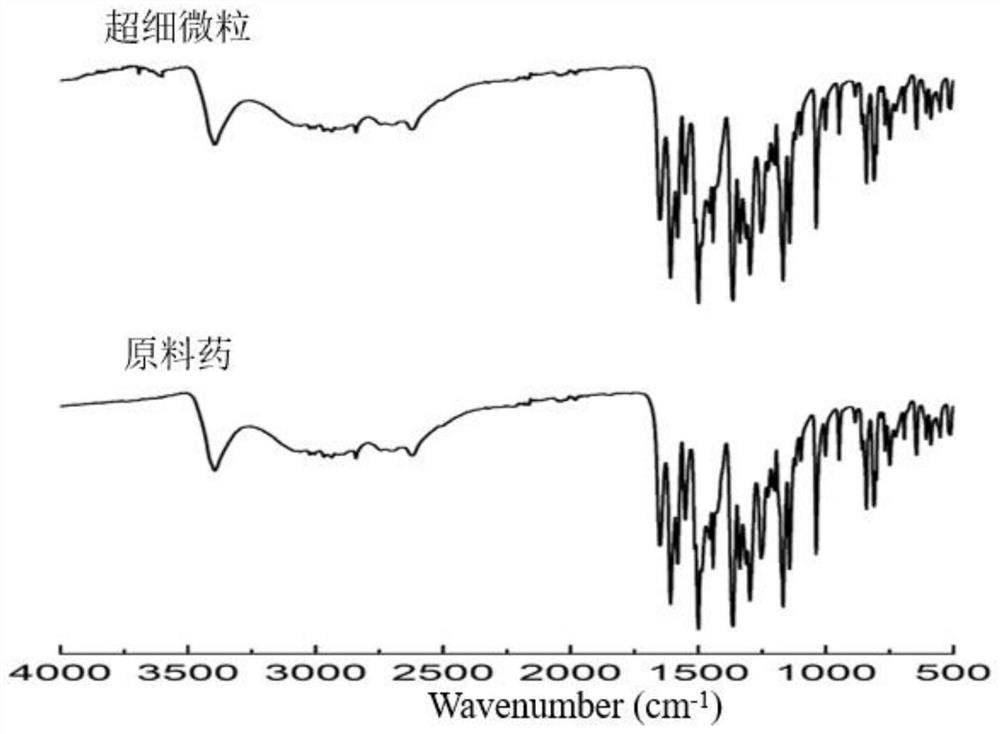
Method for preparing diosmetin derivative ultrafine particles
ActiveCN112500385AImprove bioavailabilitySmall particle sizeProductsOrganic chemistryOrganic solventMetallurgy
The invention discloses a method for preparing diosmetin derivative ultrafine particles, the diosmetin derivative is 7-O-farnesenyl-3'-O-methyl diosmetin, and the method comprises the following steps:step S1, dissolving the diosmetin derivative in an organic solvent to obtain a compound solution; S2, adjusting the temperature of the crystallization kettle to an experimental value, introducing CO2into the crystallization kettle, and pressurizing to a set experimental value; S3, continuously introducing CO2, keeping the temperature and the pressure in the crystallization kettle unchanged, andintroducing the compound solution into the crystallization kettle; and S4, after the compound solution is completely introduced, continuously introducing CO2, meanwhile, adjusting the flow rate of CO2through a rotor flow meter to be maintained within a certain flow rate range, finally, relieving the pressure to standard atmospheric pressure, taking down and opening the crystallization kettle, andcollecting ultrafine particles. According to the method, the 7-O-farnesenyl-3'-O-methyl diosmetin with smaller particle size and more uniform distribution can be obtained so that the bioavailabilityof the diosmetin is improved.
Owner:CHINA PHARM UNIV
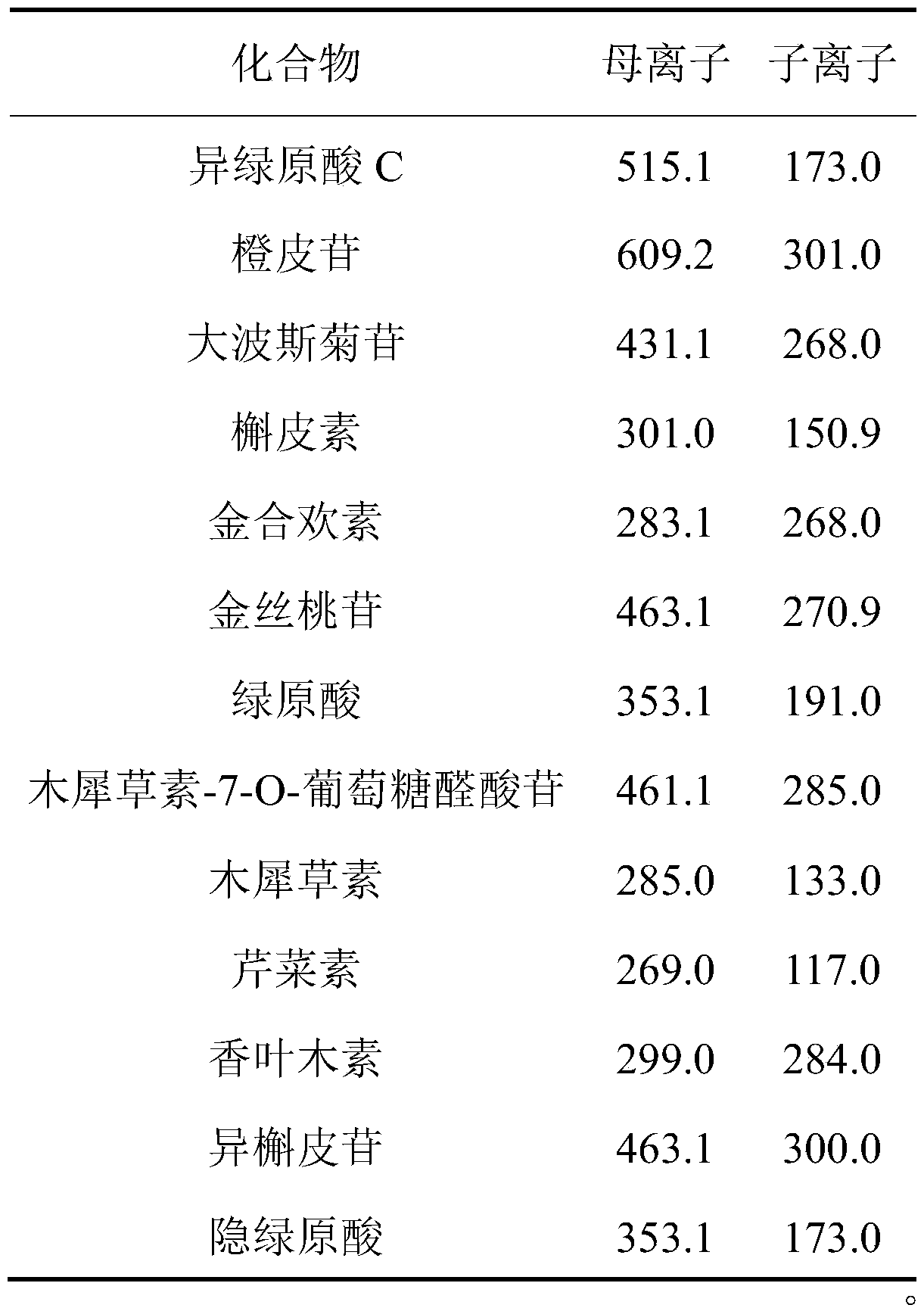
Method for determining content of chemical components in chrysanthemum
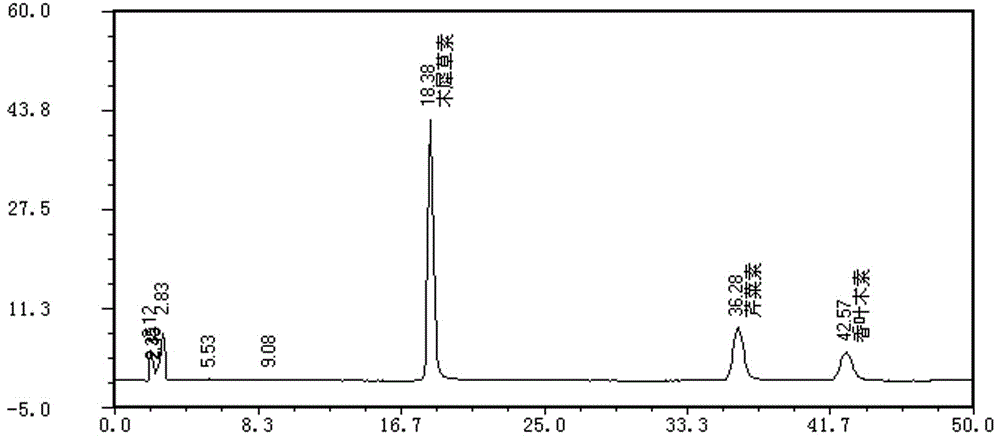
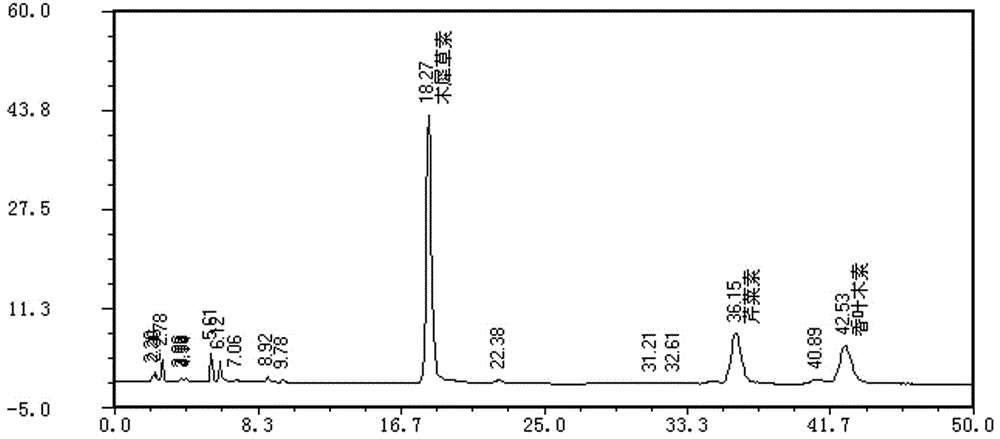
ActiveCN111537653AHigh sensitivityFast analysisComponent separationMedicinal herbsIsochlorogenic acid
The embodiment of the invention provides a method for determining the content of chemical components in chrysanthemum, which adopts ultra-high performance liquid chromatography-mass spectrometry to determine the content of 13 chemical components in chrysanthemum at the same time. The 13 chemical components comprise isochlorogenic acid C, hesperidin, coreopsis tinctoria glycoside, quercetin, farnetin, hyperoside, chlorogenic acid, luteolin-7-O-glucuronide, unuronide, luteolin, apigenin, diosmetin, isoquercitrin and cryptochlorogenic acid. By adopting the method disclosed by the invention, the contents of 13 chemical components in the chrysanthemum can be simultaneously determined by reasonably selecting chromatographic conditions and mass spectrometric conditions. And the method has the advantages of simplicity, convenience, high sensitivity, high analysis speed, strong specificity and the like, so that the method can be used for controlling the quality of the chrysanthemum medicinal material.
Owner:TIANJIN UNIV OF TRADITIONAL CHINESE MEDICINE
Flavonoid treatment of glycogen synthase kinase-based disease
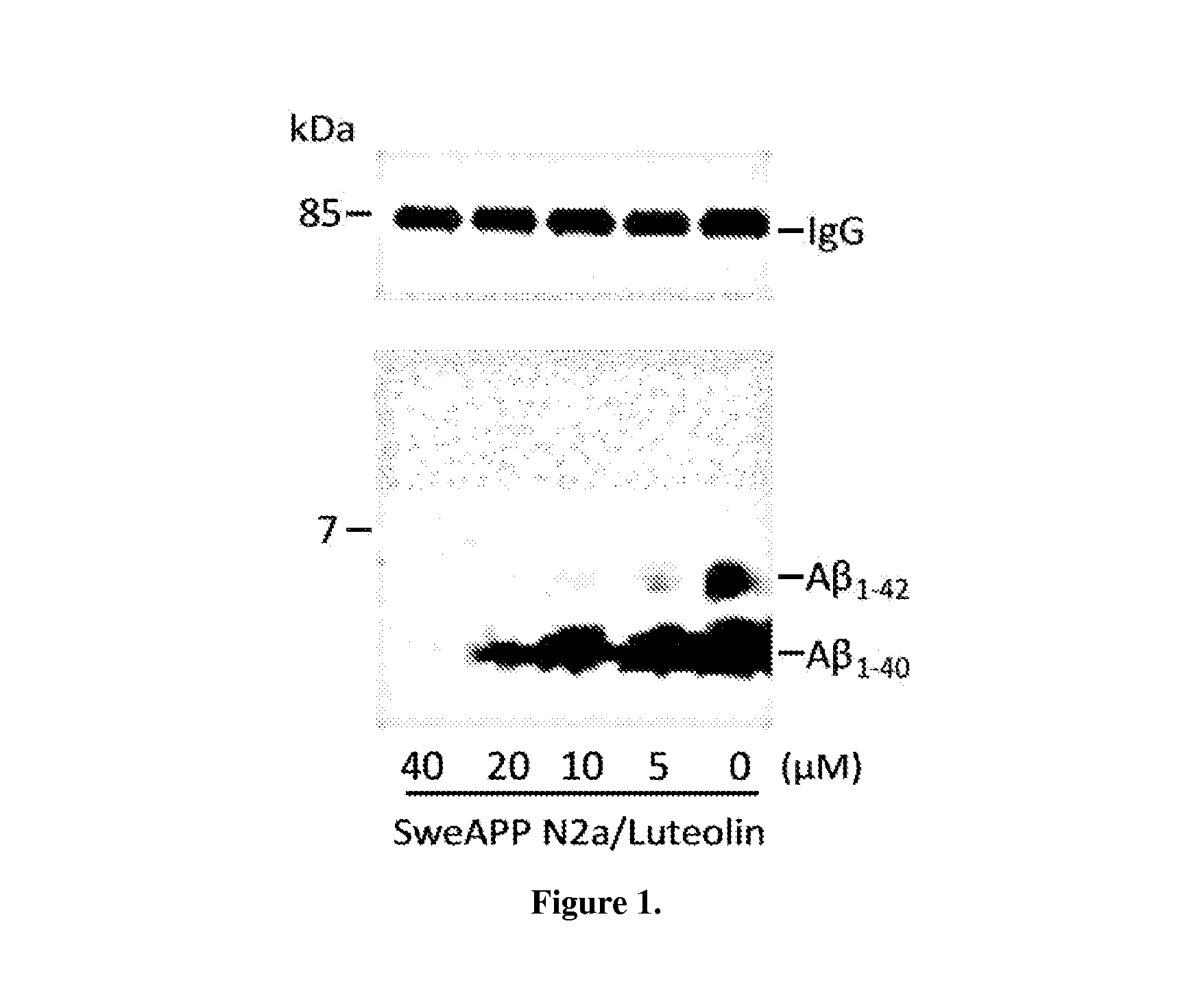
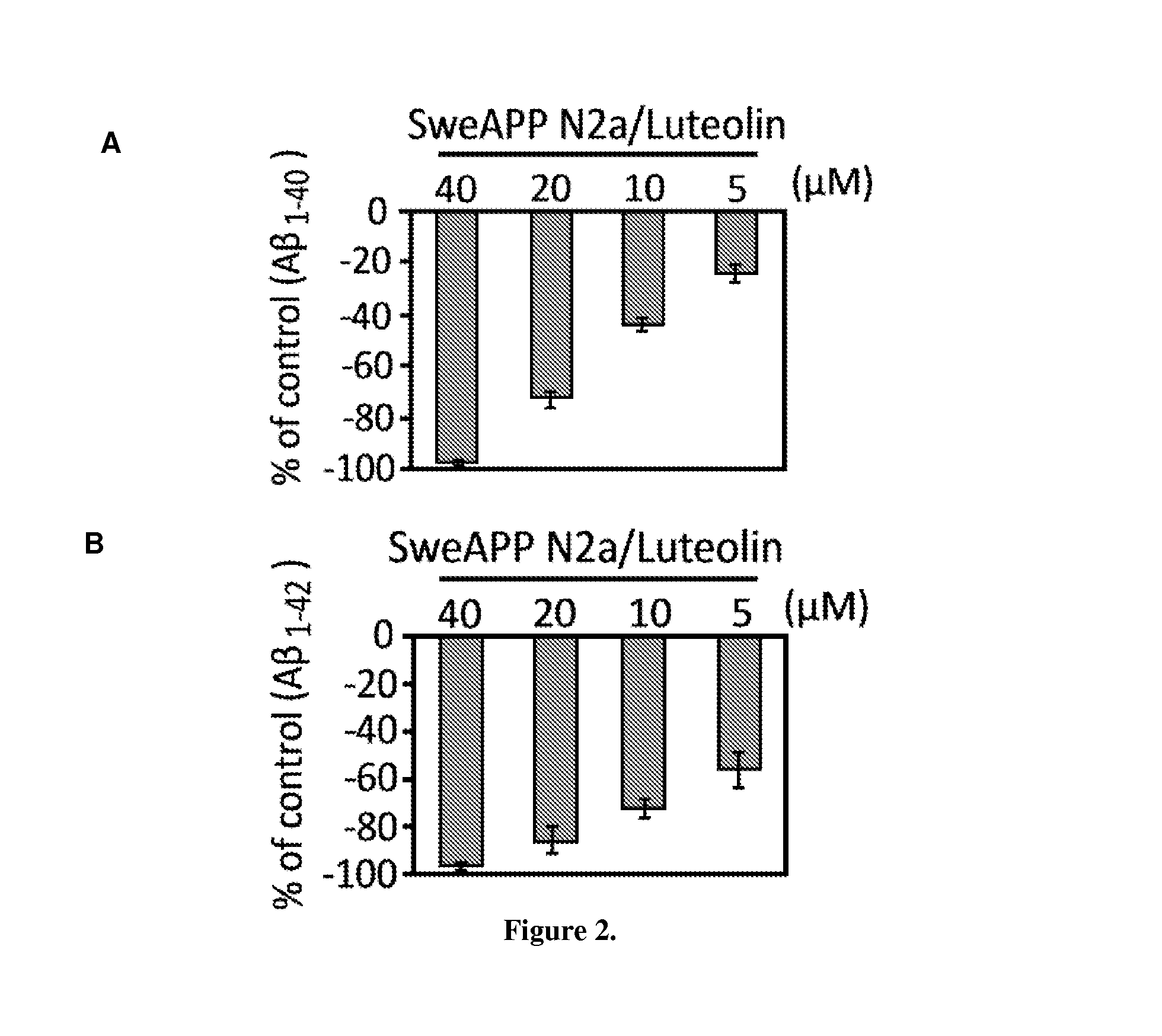
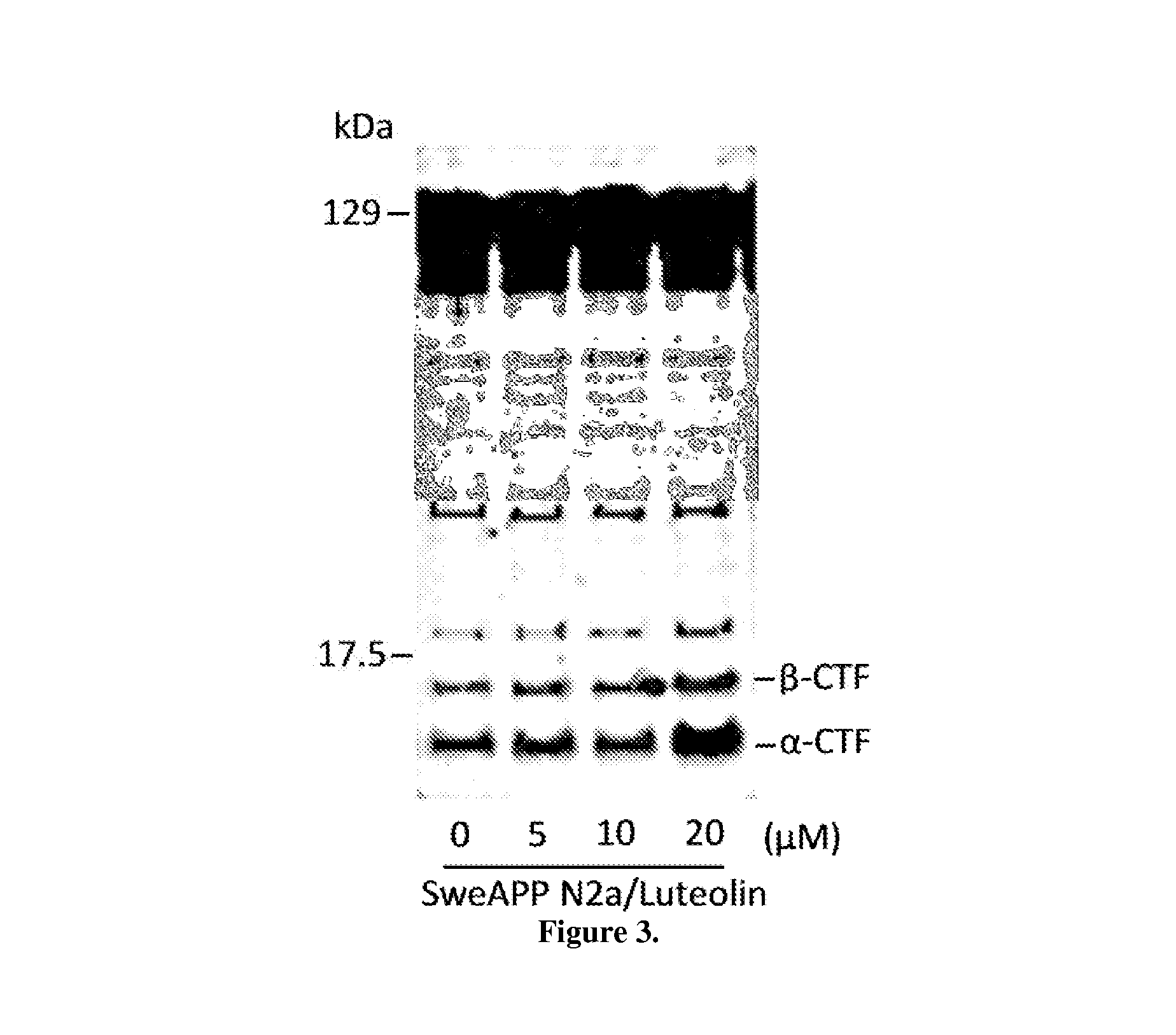
The flavonoid luteolin reduces amyloid-β peptide (Aβ) generation. Luteolin is also a selective GSK-3 inhibitor that 1) decreases amyloidogenic γ-secretase APP processing, and 2) promotes presenilin 1 (PS1) carboxyl-terminal fragment (CTF) phosphorylation. GSK-3α activity is essential for both PS1 CTF phosphorylation states and PS1-APP interaction. These findings were validated in vivo, using a Tg2576 Alzheimer's Disease model system. Luteolin treatment decreased soluble Aβ levels, reduced GSK-3 activity, and disrupted PS1-APP association. In addition, Tg2576 mice treated with diosmin, a glycoside of a flavone structurally and functionally similar to luteolin (diosmetin), displayed significantly reduced Aβ pathology as well.
Owner:UNIV OF SOUTH FLORIDA
Artemisia ordosica root extract and preparation method and application thereof
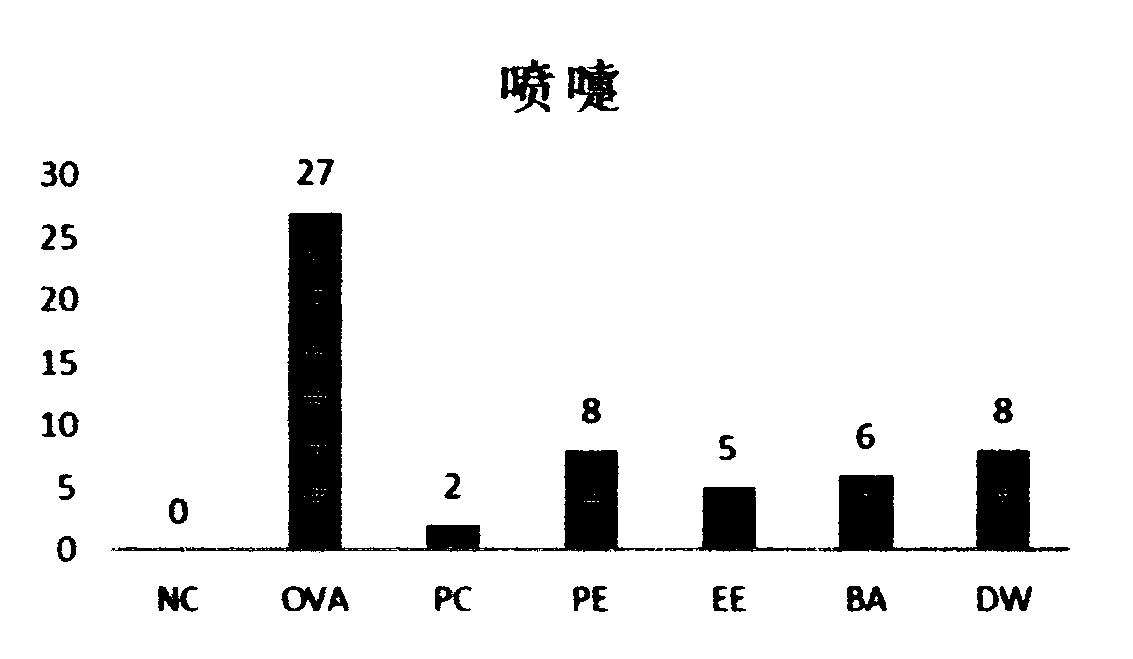
ActiveCN110960565AReduce allergic rhinitis symptomsReduce infiltrationRespiratory disorderImmunological disordersMorinAcacetin
The present invention discloses an artemisia ordosica root extract and a preparation method and an application thereof. The preparation method of the artemisia ordosica root extract comprises the following steps: drying and crushing artemisia ordosica roots and adding an ethanol solution for ultrasonic extraction; then mixing each extract and conducting concentrating and drying to obtain an extract A; and (2) dissolving the extract A with distilled water, and sequentially conducting extraction with petroleum ether, ethyl acetate and n-butanol; and collecting an ethyl acetate layer extract, andconducting concentration under reduced pressure and drying to obtain a finished product. The artemisia ordosica root extract contains 50-90% of total flavonoids, and eriodictyol, isosakuranetin, naringenin, hydroxygenkwanin, genkwanin, acacetin, diosmetin, glycitein, isorhamnetin, morin and quercetin with the total content of more than 2%. The artemisia ordosica root extract can obviously reduceallergic rhinitis symptoms of guinea pigs, also reduces levels of inflammatory factors of histamine, ovalbumin specific IgE, IL-2 / 4 / 10, IFN-gamma, ICam-1, etc. in serum of the guinea pigs, and can also reduce infiltration of eosinophilic granulocyte of nasal mucosa of nasal cavities of the guinea pigs.
Owner:肖斌 +8
Semi-synthetic method for diosmetin
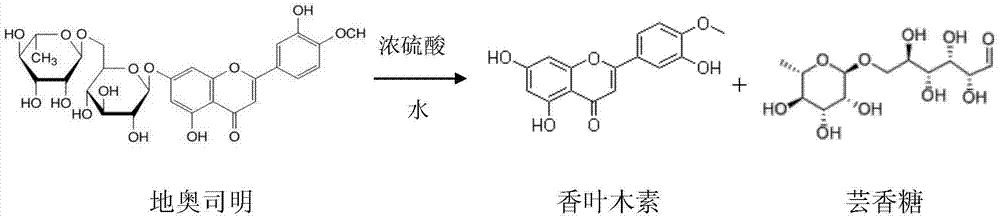
InactiveCN104119308AReduce manufacturing costNo other chemical side reactions will occurSugar derivativesDisaccharidesAlcoholDiosmin
The invention discloses a semi-synthetic process for diosmetin. The semi-synthetic process comprises the following steps of adding water and concentrated sulfuric acid into diosmin for hydrolyzing and carrying out suction filtration, washing a filter cake to obtain a diosmetin crude product; adding 50-60% alcohol into the diosmetin crude product, heating and refluxing, filtering, concentrating filtrate under reduced pressure, cooling and crystallizing to obtain a primary diosmetin pure product; adding 90-95% alcohol into the primary diosmetin pure product, heating and refluxing, filtering, concentrating filtrate under reduced pressure, cooling and crystallizing to obtain diosmetin. The process disclosed by the invention takes water as solvent to prepare diosmetin by one step, so that the synthesis process is simple, materials are easily available, reaction conditions are stable, operation is simple and convenient, production cost is lower, a product yield reaches 60-65%, purity reaches 98-99%, and therefore, the semi-synthetic process can be used for preparing large-scale production of diosmetin.
Owner:CHENGDU RUNDE PHARMA
Medicine composition for treating cardiovascular and cerebrovascular diseases and preparation method of medicine composition
InactiveCN105412850AAgainst spasmsGood healing effectEther/acetal active ingredientsUnknown materialsDiseaseHeadaches
The invention discloses a medicine composition for treating cardiovascular and cerebrovascular diseases. The medicine composition is prepared from 10 to 15 parts of Japanese ardisia herb, 10 to 15 parts of turmeric root-tuber, 8 to 12 parts of leaves of fruticose dracaena, 8 to 12 parts of dysosma versipellis, 5 to 8 parts of kudzu vine roots, 5 to 8 parts of astragalus membranaceus, 5 to 8 parts of angelica roots, 1 to 4 parts of whole scorpions, 0.1 to 0.5 part of pterostilbene and 0.1 to 0.5 part of diosmetin. The medicine provided by the invention has the remarkable effects of improving hyperkinesia fatigues and high summer heat, can be used for relieving muscle spasm, and has relatively good effects on angioneuralgia caused by vasospasm and symptoms including headache, dizziness, tinnitus, limb numbness and the like caused by hypertension; the medicine composition has the effects of improving brain and coronary circulation, lowering myocardium oxygen consumption, prolonging the life of brain cells and enhancing the intelligence, has a remarkable curative effect on myocardium tension and also has a certain curative effect on aged diminished intelligence; the medicine composition can realize the effect of relaxing vascular smooth muscles and can be used for preventing and treating vasospasm related diseases including the hypertension, heart and cerebral vessel vasospasm related diseases, stenocardia, myocardial infarction and the like.
Owner:THE AFFILIATED HOSPITAL OF QINGDAO UNIV
Application of diosmetin in preparation of medicine for treating prostatic cancer
PendingCN114404406APrevent proliferationPrevent drynessOrganic active ingredientsAntineoplastic agentsApoptosisPharmaceutical drug
The invention discloses an application of diosmetin in preparation of a medicine for treating prostatic cancer. The invention relates to an application of diosmetin in preparation of a medicine for treating prostatic cancer, and discloses that diosmetin can inhibit proliferation of prostatic cancer PC-3 cells and promote apoptosis of the prostatic cancer PC-3 cells, and also can significantly inhibit dryness, migration, adhesion and colony formation of the PC-3 cells.
Owner:遵义医科大学第二附属医院
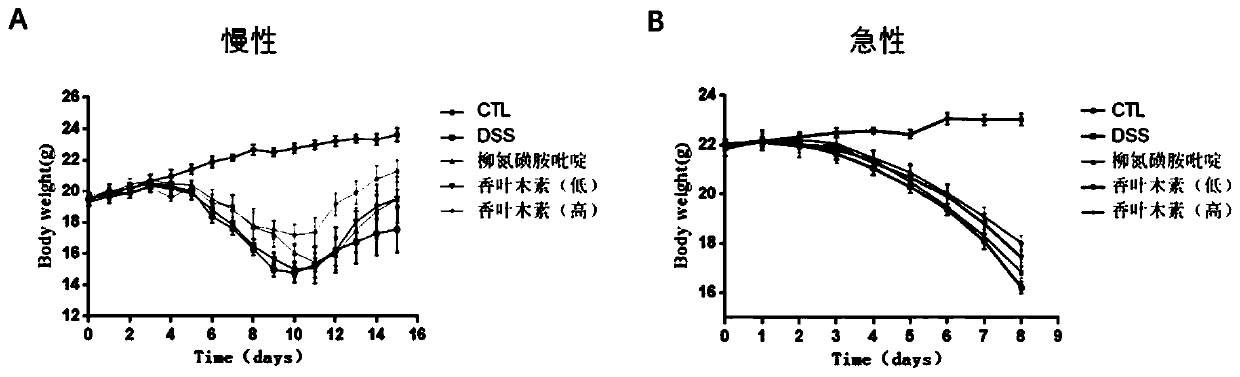
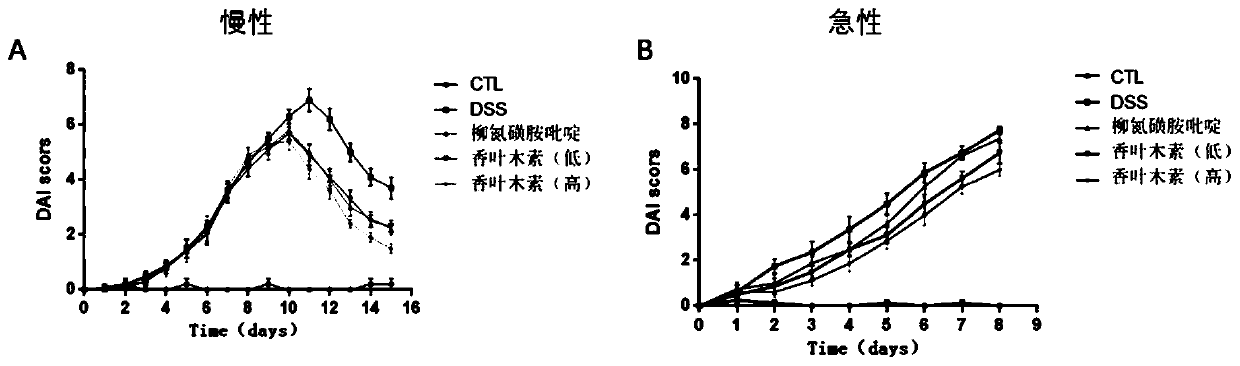
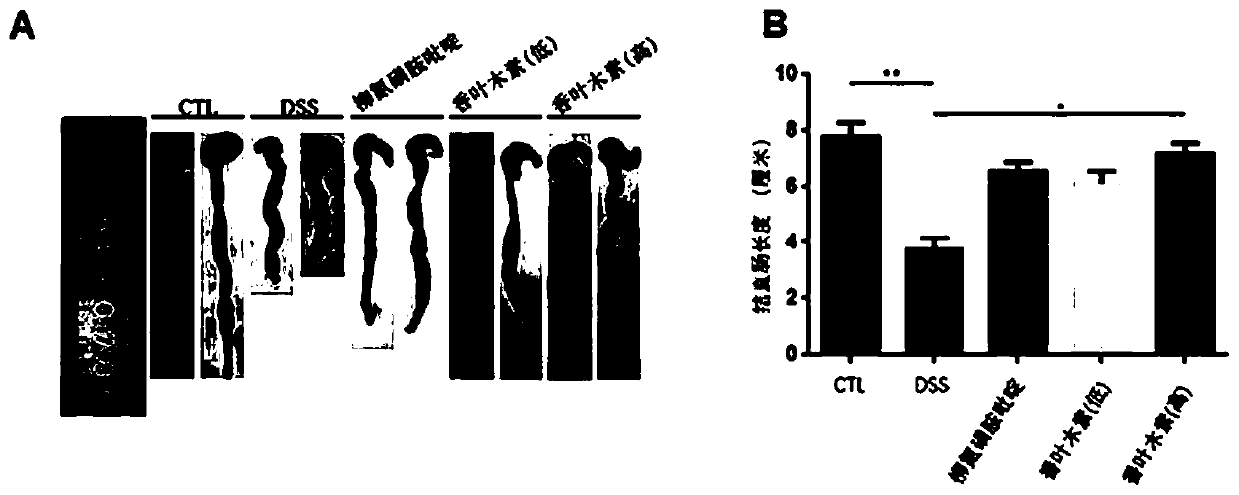
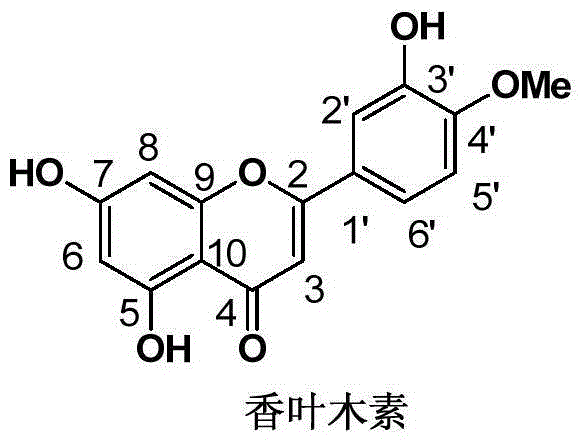
Application of diosmetin in preparation of medicament for treating inflammatory bowel disease
InactiveCN111374971AGood effectNo adverse reactionOrganic active ingredientsDigestive systemBowels diseasesEfficacy
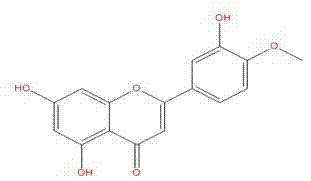
The invention provides application of diosmetin in preparation of a medicament for treating inflammatory bowel disease (IBD), and belongs to the technical field of medicines. The diosmetin has a structure shown in the following formula (I): The diosmetin has a good effect on IBD, has no adverse reaction, can slow down ulcerative enteritis of mice induced by dextran sodium sulfate (DSS), and provides a good application prospect for treating, alleviating or improving IBD.
Owner:NANKAI UNIV
Method for extracting and separating diosmetin from water chestnut peel
InactiveCN104529976AScientific and reasonable designEasy to separateOrganic chemistryChromatographic separationAcetic acid
The invention discloses a method for extracting and separating diosmetin from a water chestnut peel and belongs to the field of natural organic chemistry. The method is characterized by comprising the following steps: extracting diosmetin from the water chestnut peel by taking an acetone water solution as an extraction solvent; extracting the extract by using ethyl acetate; performing chromatography separation on the extract by virtue of intermediate-pressure MCI and a polyamide column; repeatedly purifying by using Sephade x LH-20 gel chromatography to obtain a diosmetin product of which the content is larger than 95%. The method disclosed by the invention opens up a new way for extracting and separating the diosmetin from natural plants, and is good in separation effect, high in product purity and capable of changing the water chestnut peel which is always considered as waste into a valuable material; the used MCI resin, polyamide and gel can be reused; the raw materials are easily available; resources are sufficient; the production cost is low; large-scale production can be realized; the requirements of medical industry are satisfied.
Owner:HEZHOU UNIV
Method for on-line enrichment and purification of diosmetin in dandelion
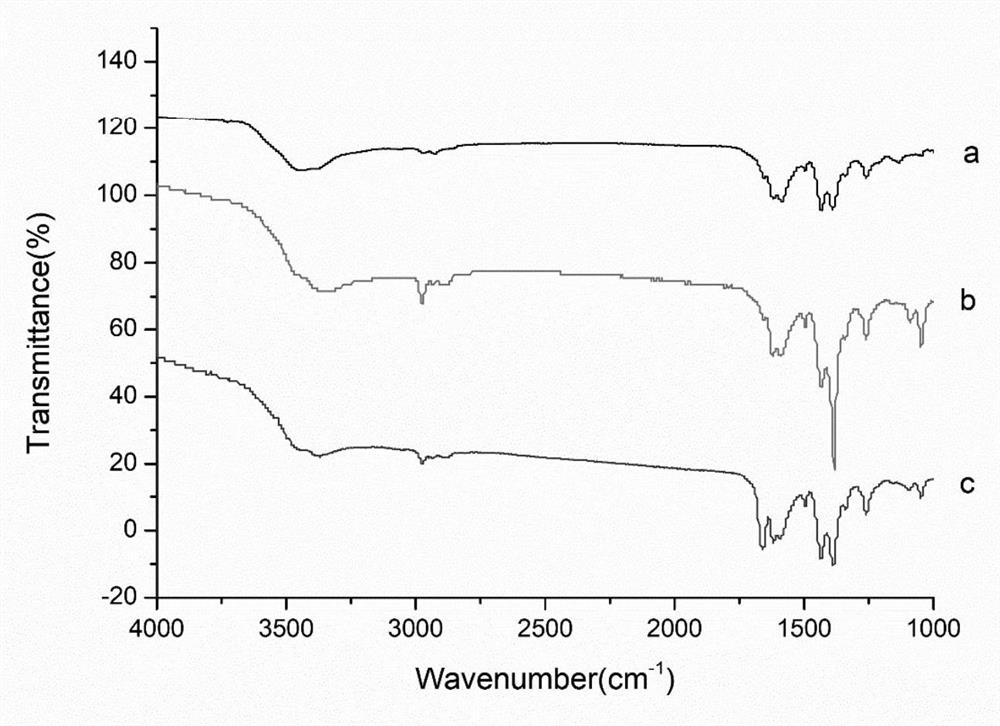
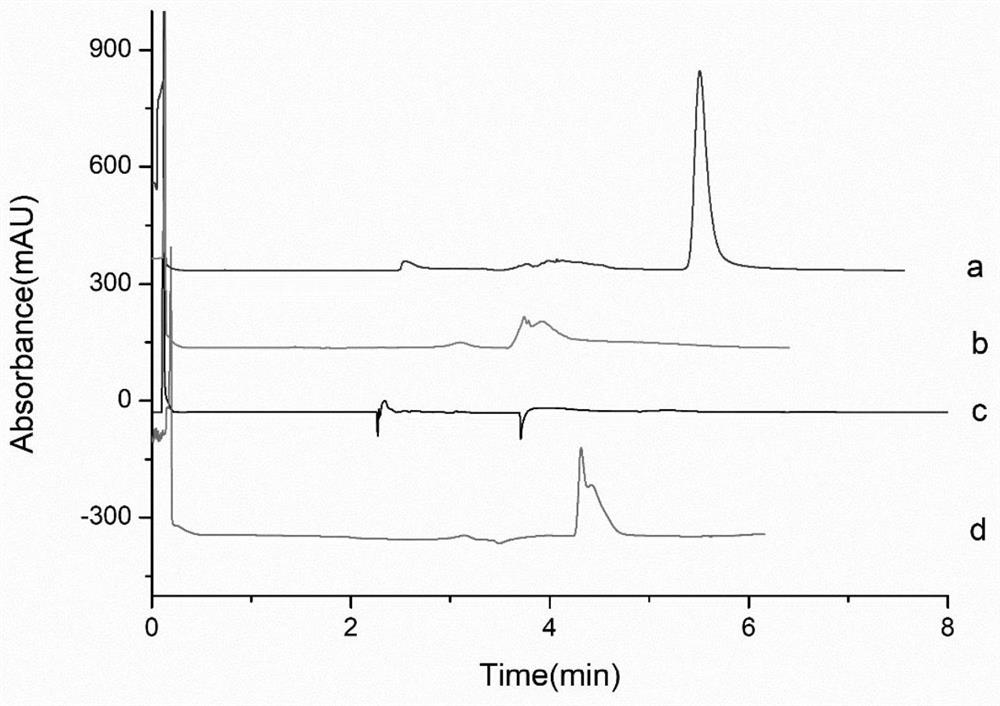
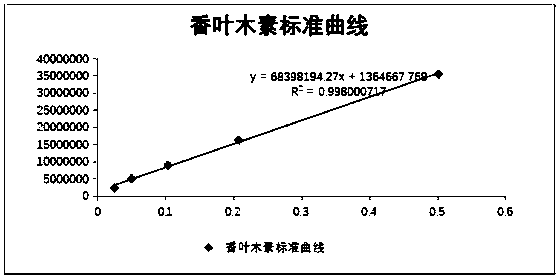
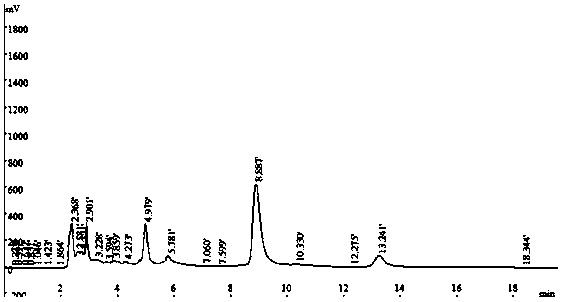
ActiveCN114405063AOptimized areaImprove the deficiency of low porosityOrganic chemistryOther chemical processesSynthetic materialsPhysical chemistry
The invention provides a method for on-line enrichment and purification of diosmetin in dandelion. The method comprises the following steps: (a) synthesis of an MOF material; (b) derivatization of the MOF material; (c) preparing the monolithic column; (d) enriching and purifying diosmetin on line; and (e) separating the diosmetin. According to the prepared MOF / polymer monolithic column, the defects that a traditional polymer monolithic column is small in specific surface area and low in porosity are overcome, the adsorption performance of the MOF / polymer monolithic column on diosmetin is enhanced, and the material can effectively separate and enrich diosmetin. Meanwhile, by adopting a mode of combining normal phase chromatography and reversed phase chromatography, the separation effect of diosmetin and impurities is enhanced, and high-purity diosmetin can be effectively prepared. The method has the advantages of simplicity in operation, economy and effectiveness.
Owner:HEBEI UNIVERSITY
Method for preparation of diosmetin from peanut shells
Belonging to the field of natural compound extraction, separation and purification, the invention provides a method for preparation of diosmetin from peanut shells. The method utilizes microwave-assisted extraction, employs an iron salt to remove luteolin and other components, and then performs purification by preparative high performance liquid chromatography to obtain diosmetin with purity of greater than 95%. The method is simple and rapid, and expands the sources of diosmetin.
Owner:BEIJING INSTITUTE OF TECHNOLOGYGY