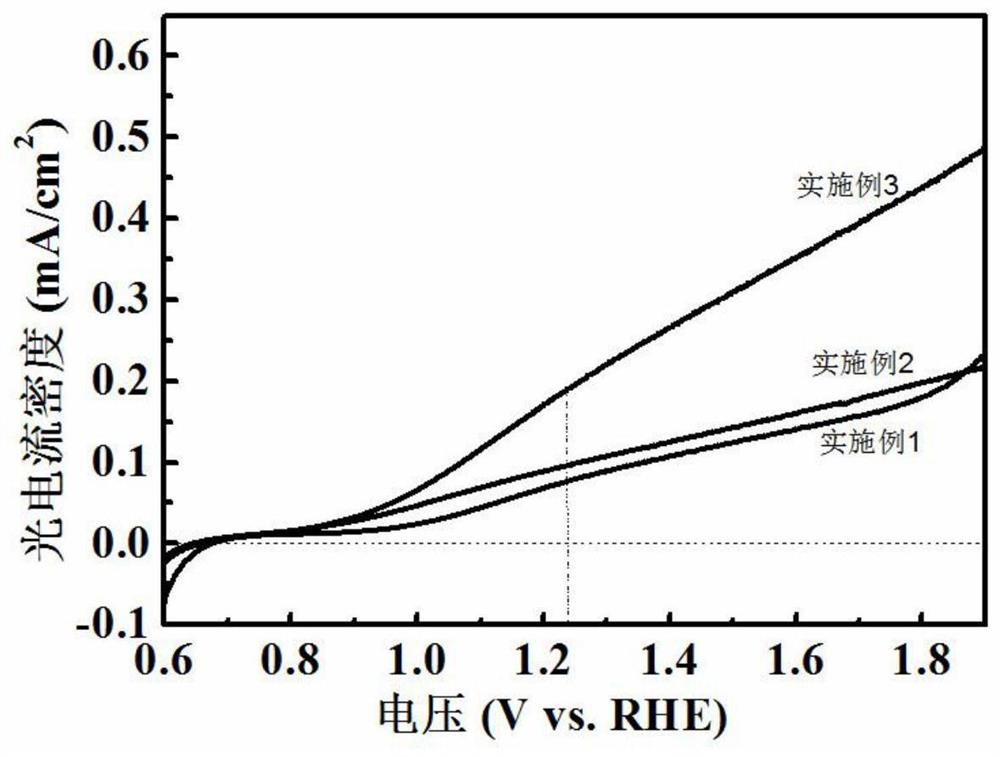
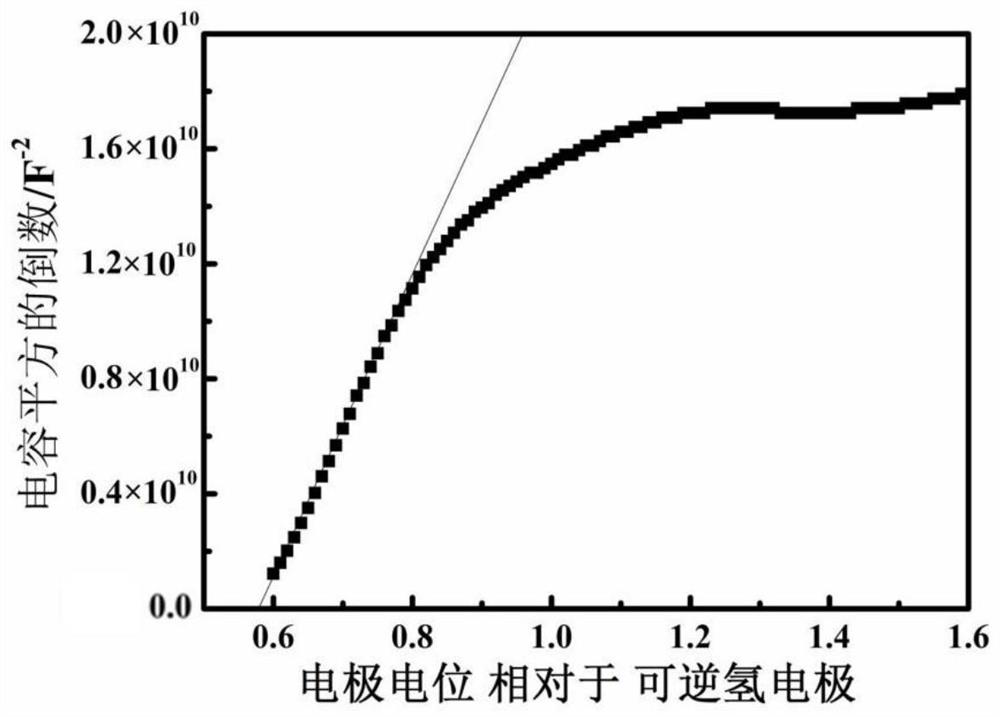
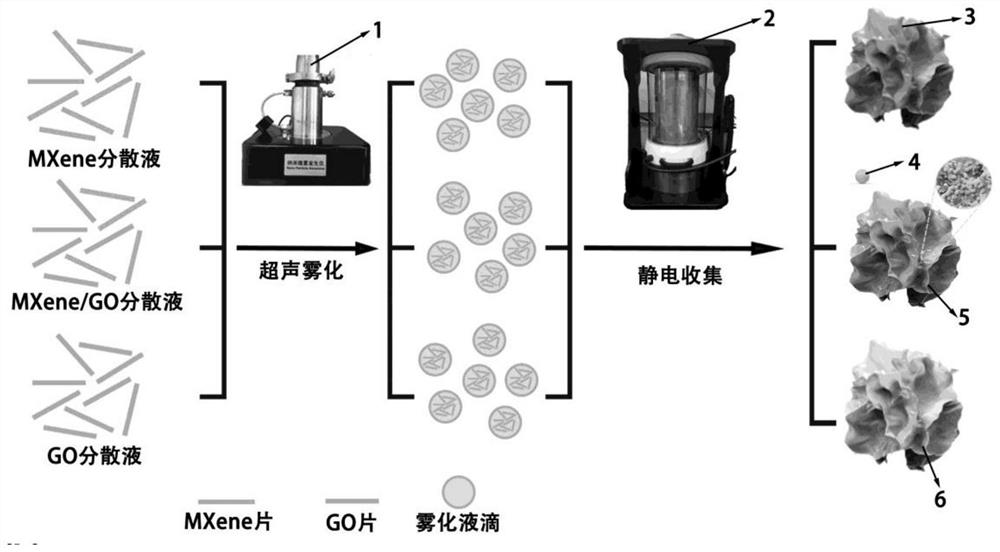
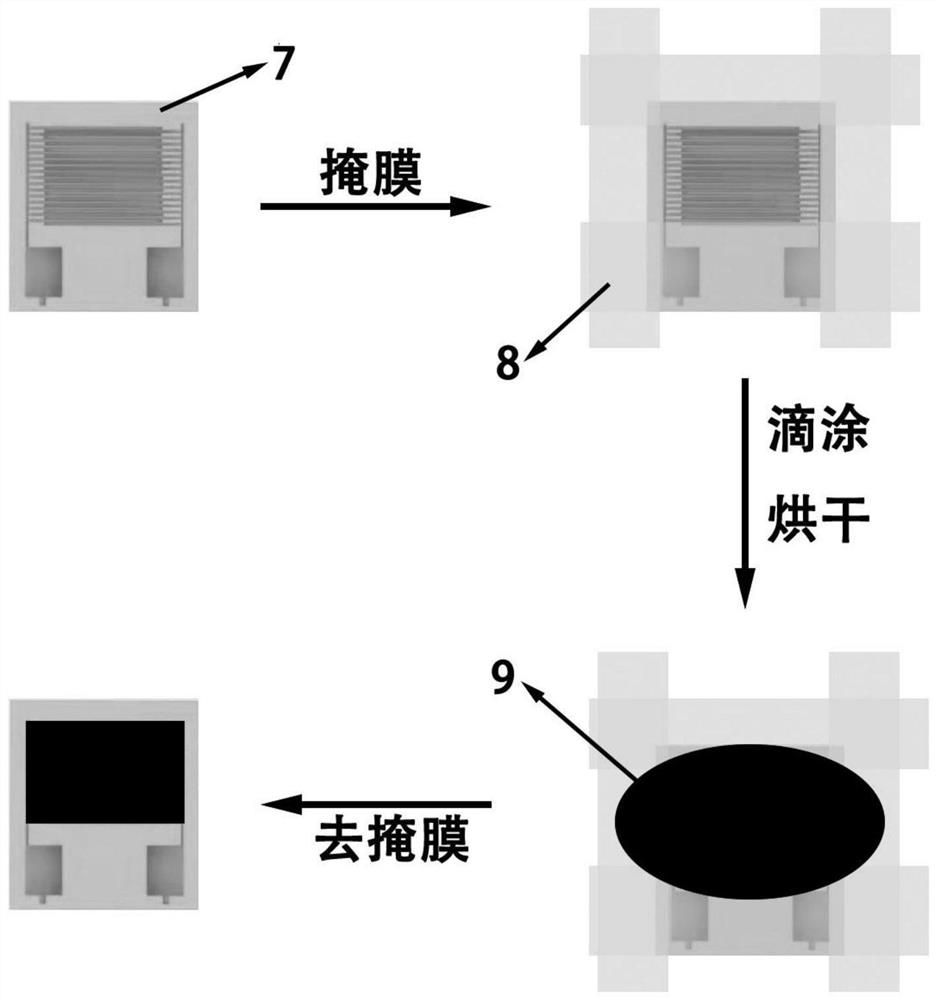
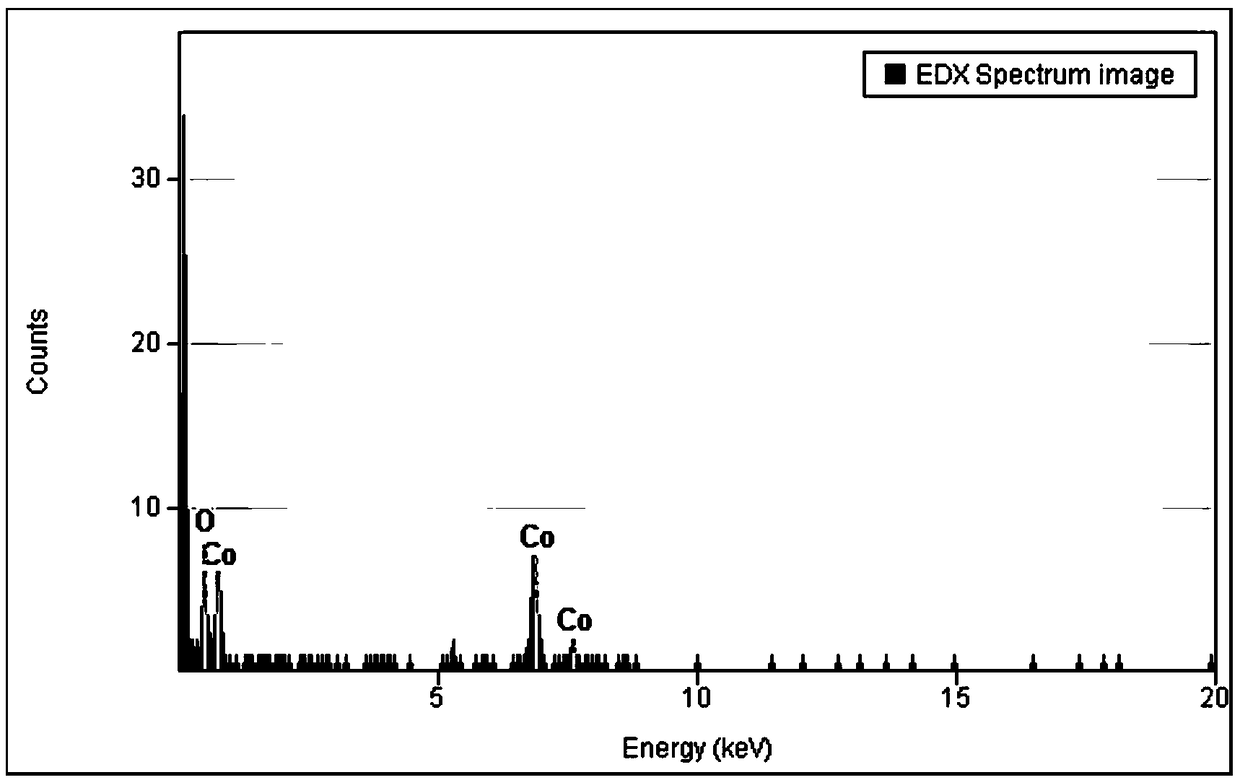
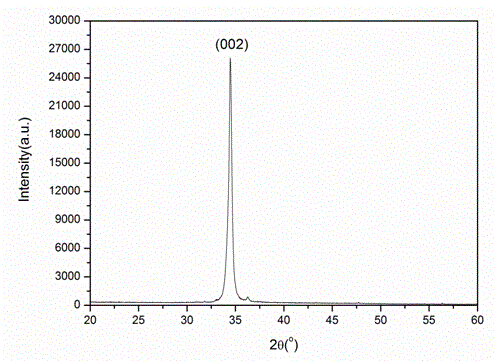
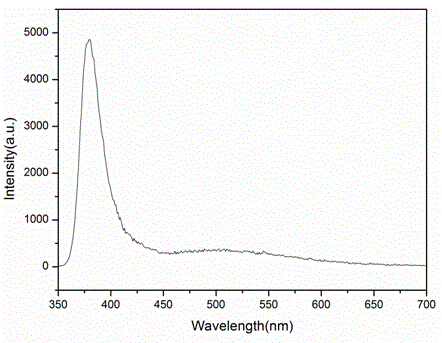
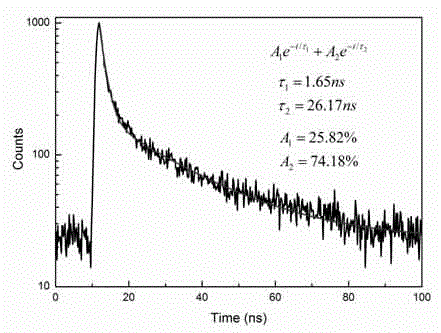
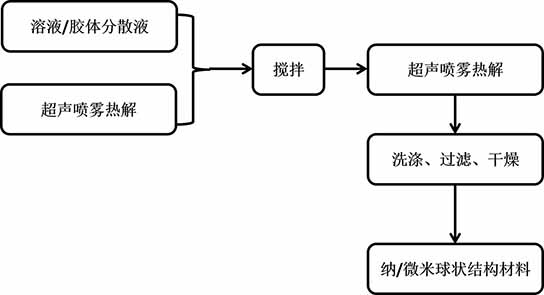
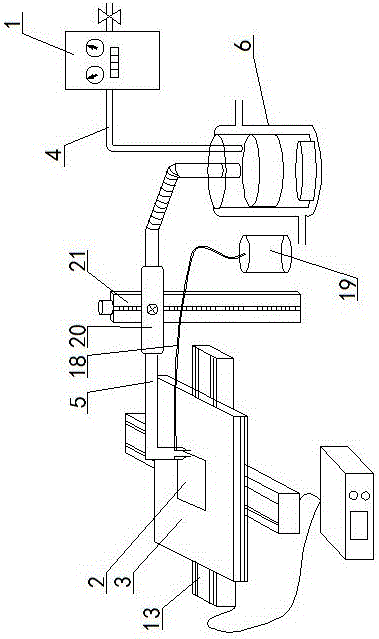
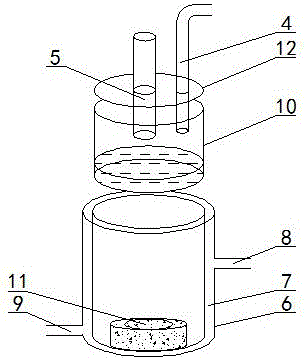
Patents
Literature
52 results about "Ultrasonic spray pyrolysis" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
Controllable Synthesis of Porous Carbon Spheres, and Electrochemical Applications Thereof
InactiveUS20110082024A1Good dispersionExcellent ORR activityMaterial nanotechnologyReactant parameters controlColloidal silicaSmall droplet
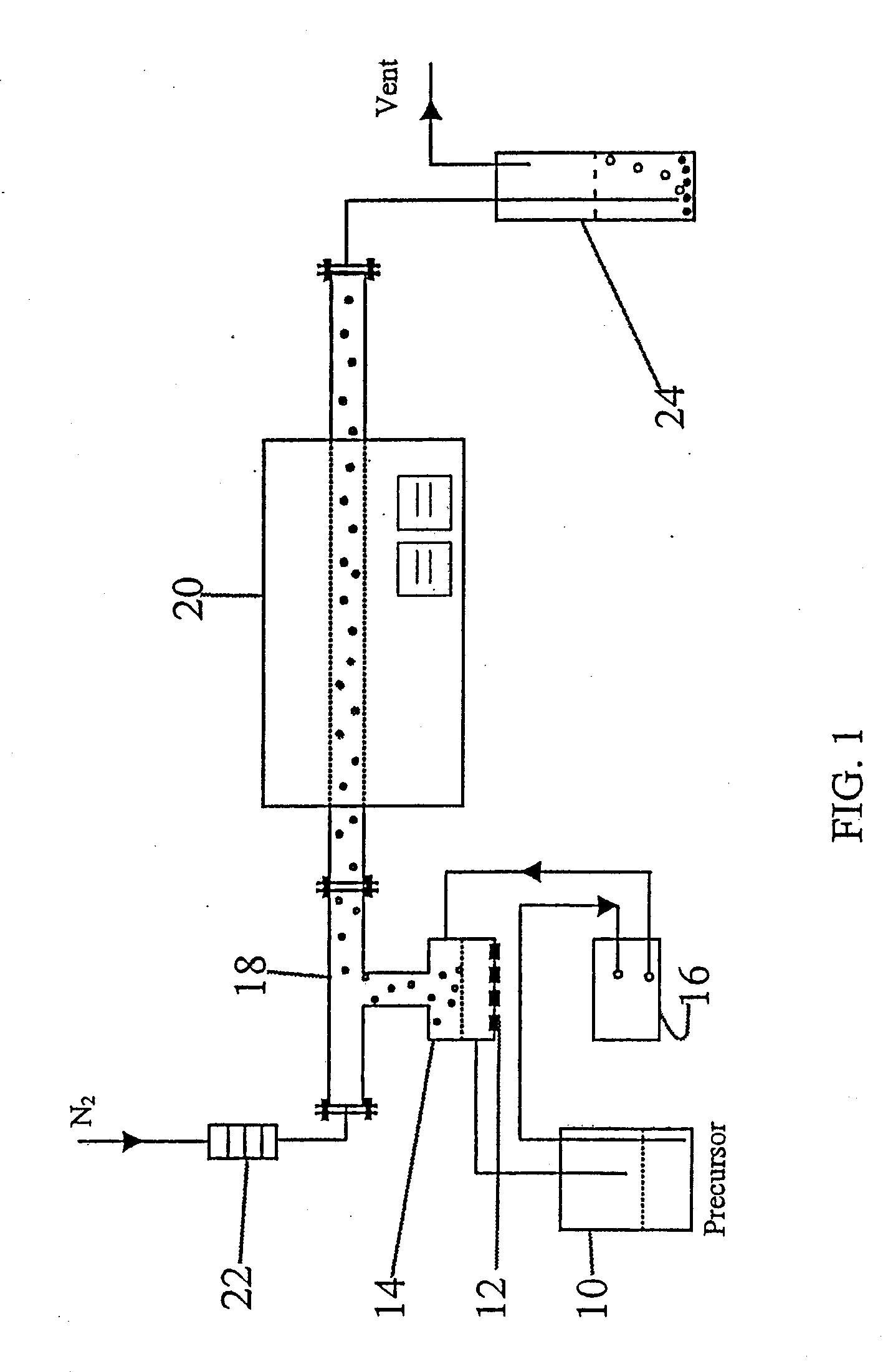
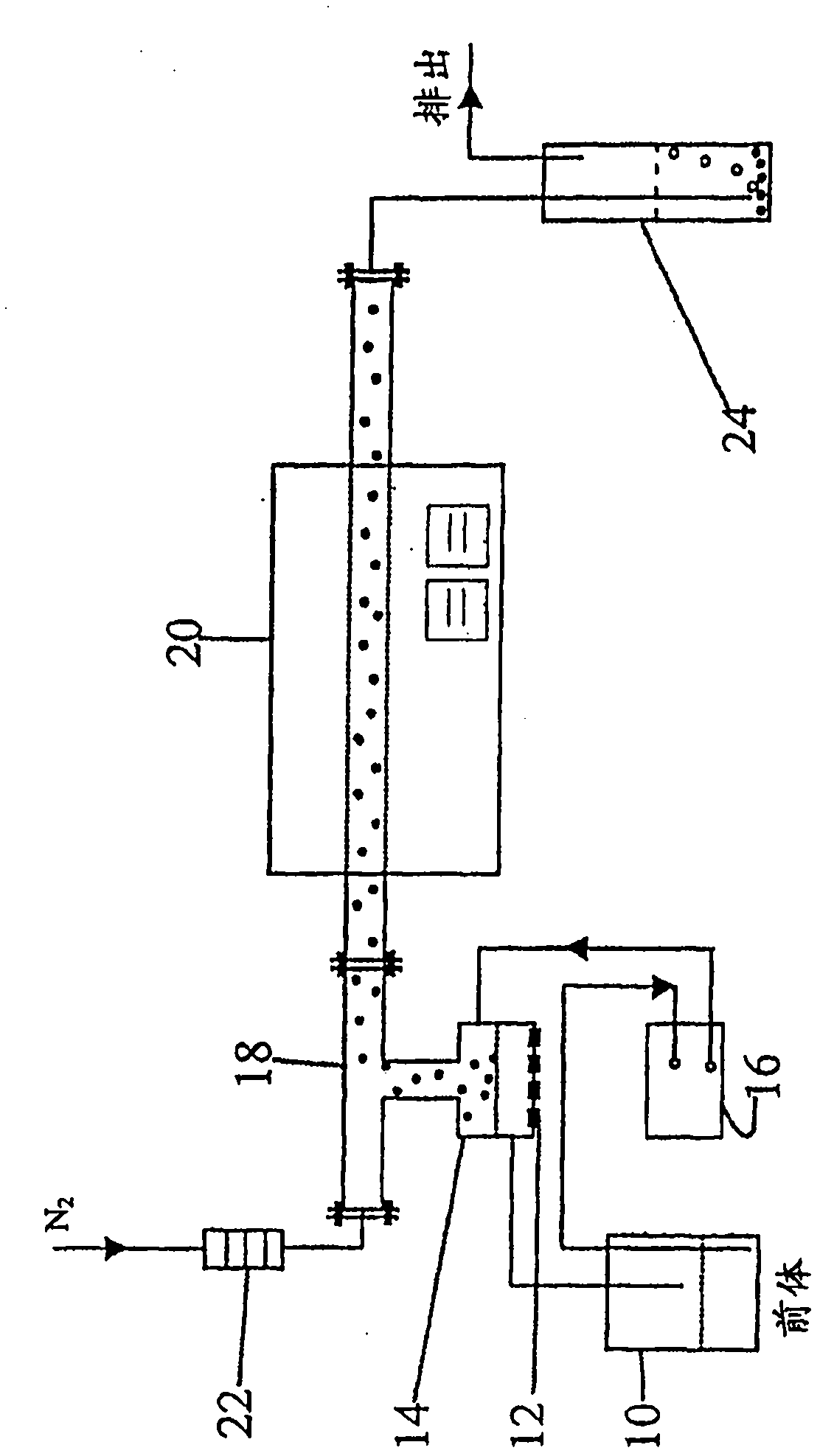
The invention disclosed relates to porous carbon of spherical morphology having tuned porosity and to a method of making same, comprising: (a) providing a precursor solution, by combining in an aqueous solution a colloidal silica template material and a water-soluble pyrolyzable carbon source, wherein the particle size of the colloidal silica template and the colloidal silica / carbon source weight ratio are controlled, (b) atomizing the precursor solution into small droplets by ultrasonic spray pyrolysis (c) directing the droplets into a high temperature furnace operating at a temperature of 700-1200° C., under an inert gas atmosphere, where the droplets are transformed into solid spherical composite carbon / silica particles, (d) collecting the resulting composite carbon / silica particles exiting from the furnace, and (e) removing the silica from the particles, to provide substantially pure porous carbon of spherical morphology having tuned porosity defined by surface area and pore size. The porous carbon according to the invention is used as catalyst supports in PEM fuel cells, as electrodes in supercapacitors and lithium in batteries, for hydrogen storage and as earners for drug delivering.
Owner:NAT RES COUNCIL OF CANADA
F, Mn codoped deposit nano SnO2 transparent thermal-insulation film
The invention discloses an F, Mn co-doping deposition nano-SnO2 transparent heat insulating thin film, wherein the F, Mn element co-doping effect is utilized to increase the number charge carriers in the thin film material, thus increasing the index of thin film reflection to infrared rays. The invention can prevent the excess doping of one single element, it can be applied to chemical CVD or supersonic spraying thermal dissociation deposition.
Owner:雷智 +1
Hydrothermal preparation method for NiS2 with controllable shape
InactiveCN102633309AEasy to operateMild reaction conditionsNanotechnologyNickel sulfidesNickel saltReaction temperature
The invention discloses a hydrothermal preparation method for NiS2 with a controllable shape, and the method mainly comprises the following steps of: mixing nickel salt, a sulphur source and a complexing agent in a certain molar ratio, transferring the mixture into a high-pressure reactor, adding distilled water, and stirring to adequately dissolve soluble solids; adjusting the pH value of the reaction system, sealing the reactor, and reacting at a certain temperature; and washing the obtained product by use of distilled water and ethanol respectively, centrifugally separating, and drying. The hydrothermal preparation method for NiS2 with a controllable shape disclosed by the invention has the advantages of being simple in operation by using a hydrothermal method, moderate in reaction condition, narrow in particle size distribution, high in product purity, easy in industrial production, and the like. The particle shape and size of NiS2 can be controlled by adjusting the synthesis conditions such as hydrothermal reaction temperature, reaction time, complexing agent type and pH value, and the prepared NiS2 can be approximately-cube-shaped, spherical, sheet-shaped and the like. Compared with a solid-state reaction method, an ultrasonic spray pyrolysis method, a gamma-ray irradiation method and an organic solvent hot method, the NiS2 prepared by the hydrothermal method has the advantages of being low in reaction temperature and equipment demand, less in toxicity on a human body, high in safety factor, low in production cost, capable of meeting the requirements of energy conservation and emission reduction, and the like.
Owner:SHENYANG LIGONG UNIV
Controllable synthesis of porous carbon spheres, and electrochemical applications thereof
The invention disclosed relates to porous carbon of spherical morphology having tuned porosity and to a method of making same, comprising: (a) providing a precursor solution, by combining in an aqueous solution a colloidal silica template material and a water-soluble pyrolyzable carbon source, wherein the particle size of the colloidal silica template and the colloidal silica / carbon source weight ratio are controlled, (b) atomizing the precursor solution into small droplets by ultrasonic spray pyrolysis, (c) directing the droplets into a high temperature furnace operating at a temperature of 700-1200 0C, under an inert gas atmosphere, where the droplets are transformed into solid spherical composite carbon / silica particles, (d) collecting the resulting composite carbon / silica particles exiting from the furnace, and (e) removing the silica from the particles, to provide substantially pure porous carbon of spherical morphology having tuned porosity defined by surface area and pore size. The porous carbon according to the invention is used as catalyst supports in PEM fuel cells, as electrodes in supercapacitors and lithium in batteries, for hydrogen storage and as earners for drug delivering.
Owner:NAT RES COUNCIL OF CANADA
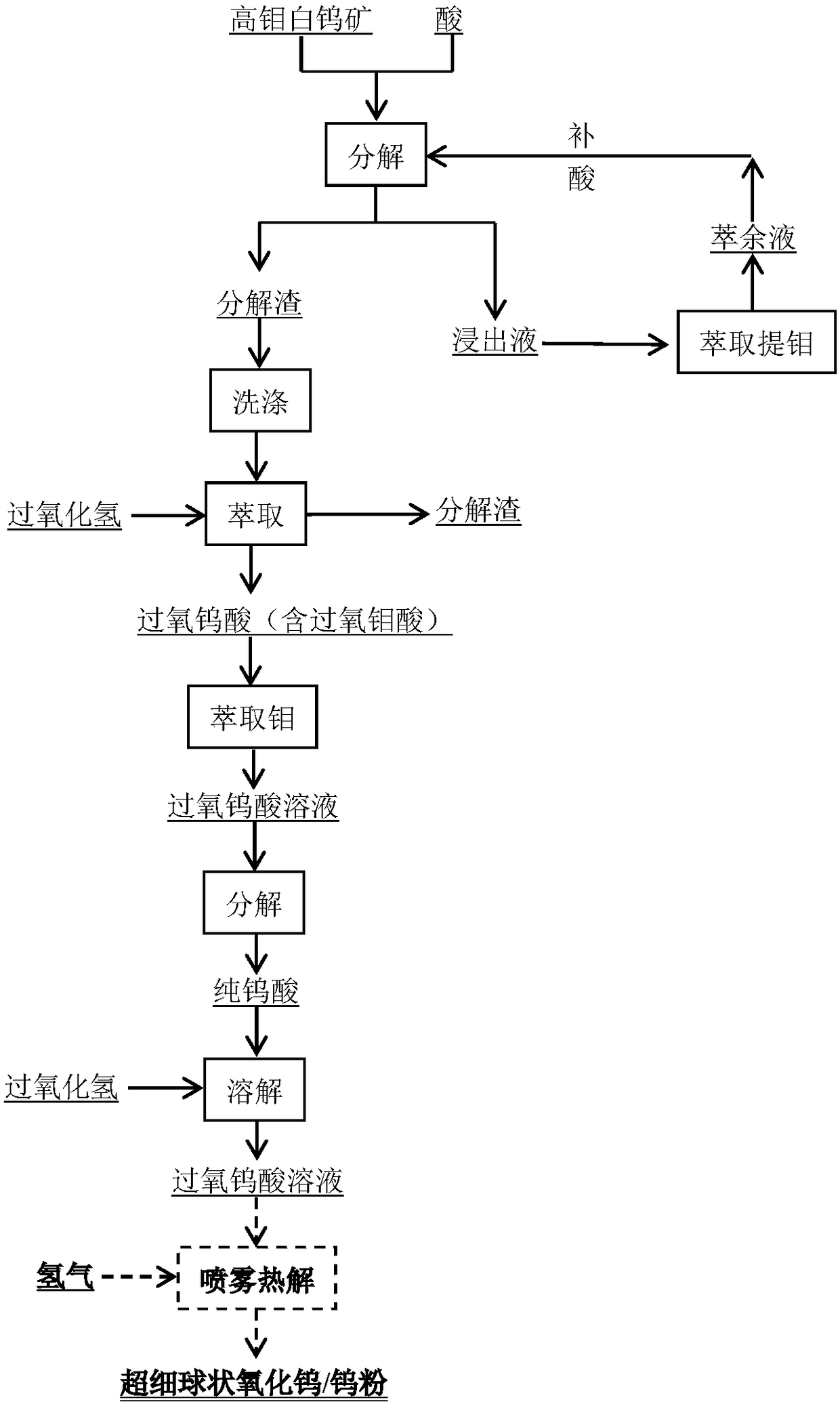
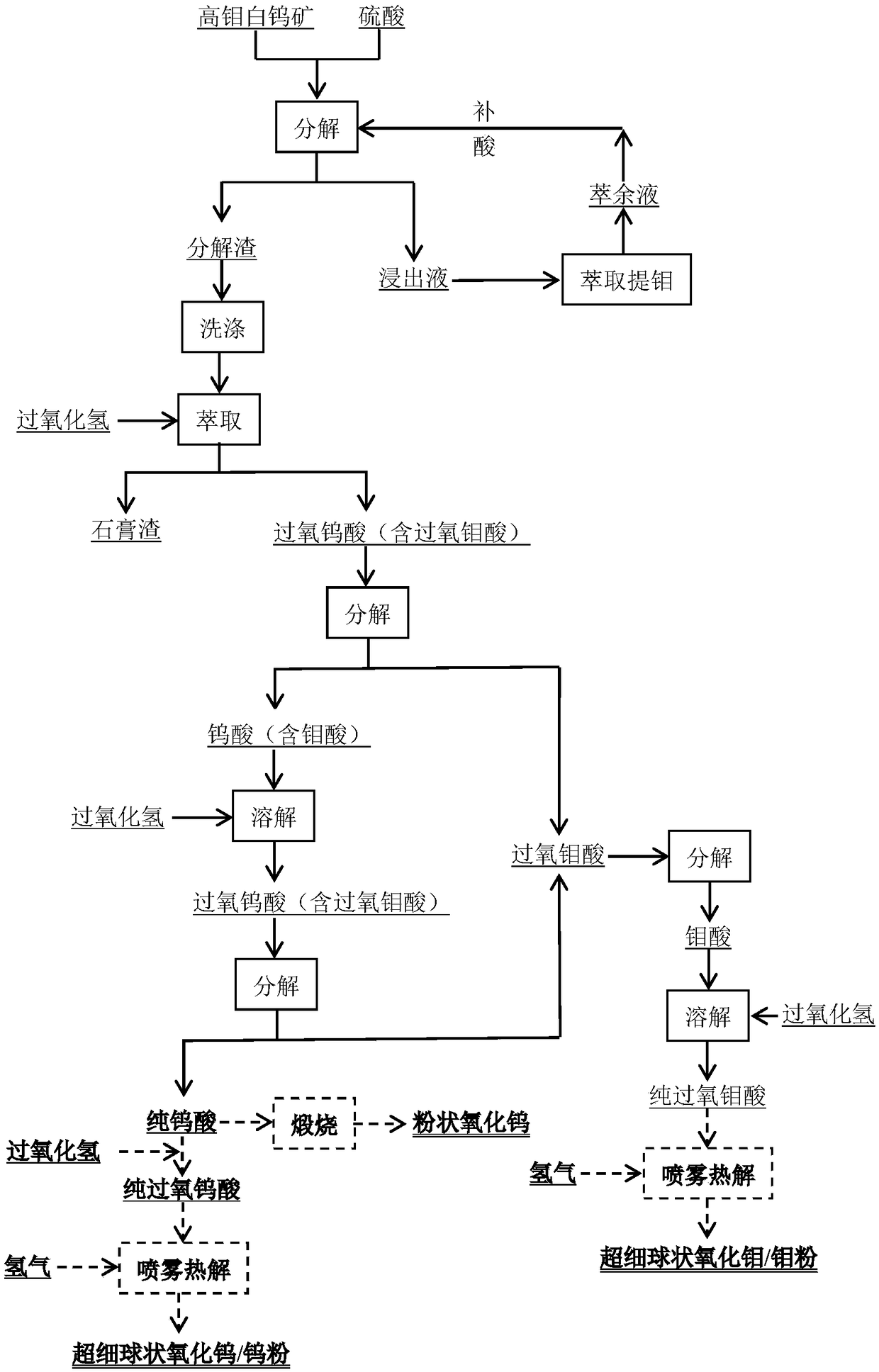
Method for extracting tungsten and molybdenum through acid decomposition of high-molybdenum scheelite
ActiveCN108425010AReduce break down costReduce manufacturing costProcess efficiency improvementDecompositionUltrasonic spray pyrolysis
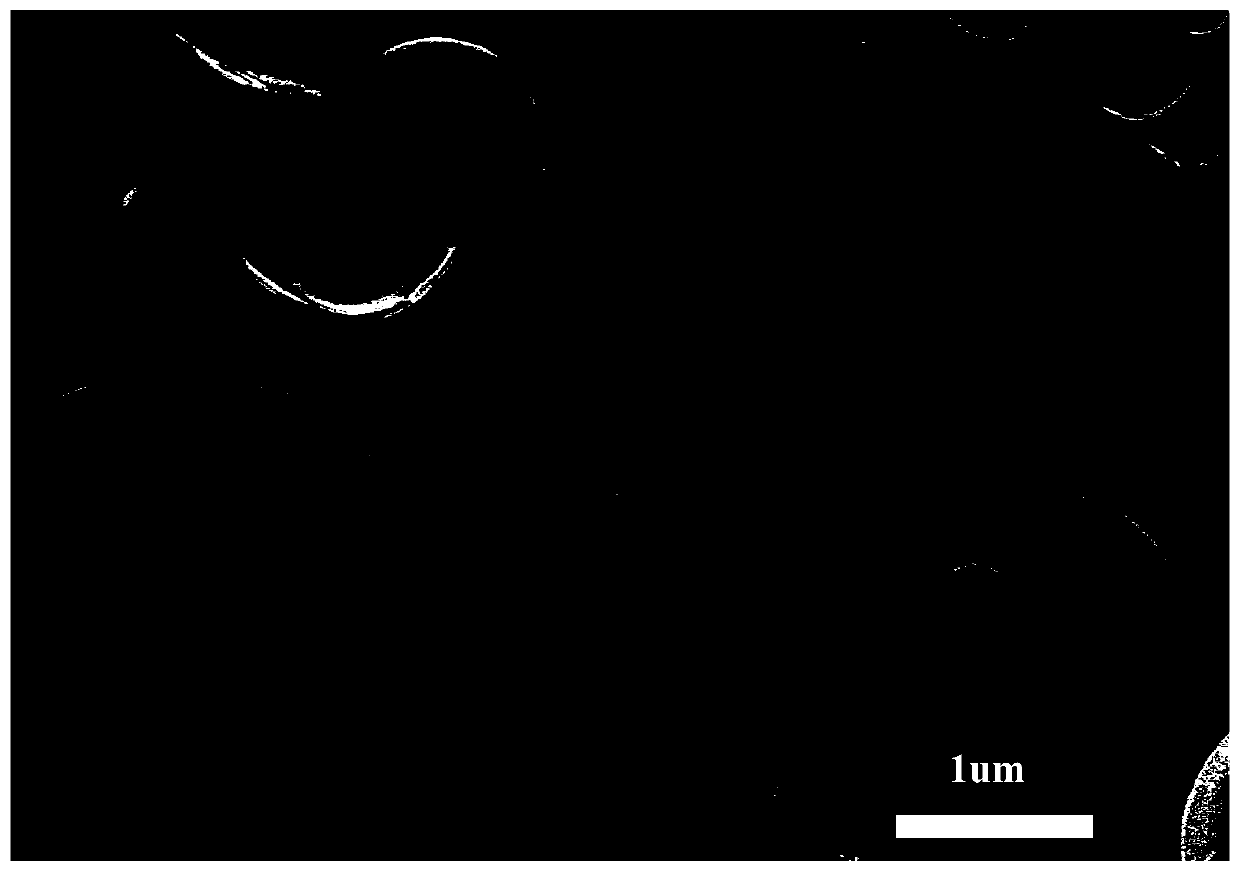
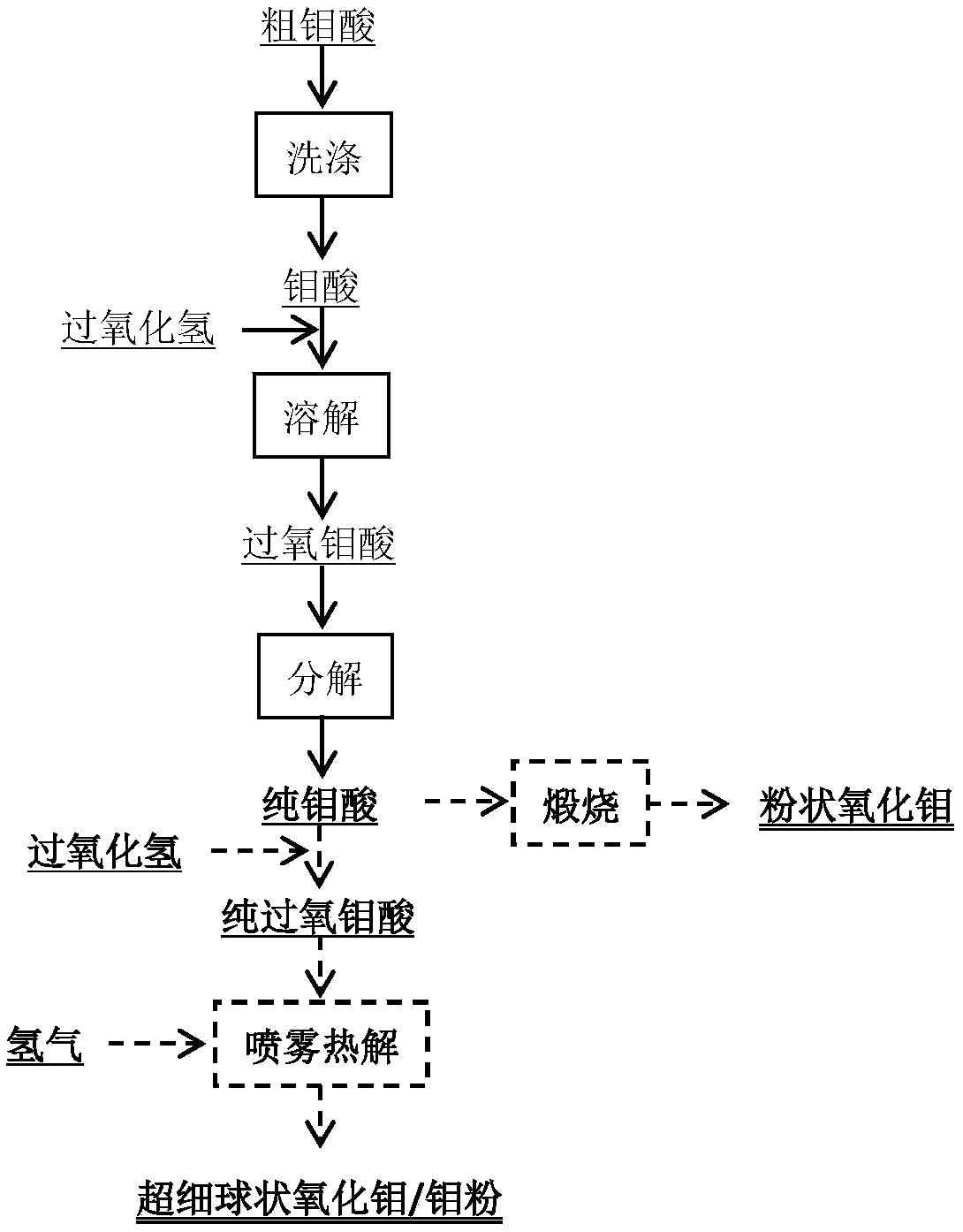
The invention relates to a method for extracting tungsten and molybdenum through acid decomposition of high-molybdenum scheelite. The method comprises the following steps: leaching the high-molybdenumscheelite with an acid solution, and extracting a leachate to extract molybdenum; extracting a leaching residue with hydrogen peroxide to obtain a solution containing peroxotungstic acid and peroxymolybdic acid, and heating or introducing SO2 to obtain tungstic acid and a solution containing the peroxomolybdic acid; extracting the tungstic acid with hydrogen peroxide, and heating or introducing SO2 to obtain tungstic acid and a solution containing the peroxomolybdic acid; repeating for several times till pure tungstic acid is obtained; preparing tungsten trioxide or tungsten powder from the pure tungstic acid; and heating or introducing SO2 into the solution containing the peroxomolybdic acid, dissolving obtained molybdic acid in a hydrogen peroxide solution to prepare a peroxomolybdic acid solution, and performing ultrasonic spraying pyrolysis to prepare molybdenum trioxide powder or molybdenum powder. By the method, the separation effect is good and the operation process is simple and easy to control, so that the method is easy to popularize and apply industrially.
Owner:CENT SOUTH UNIV
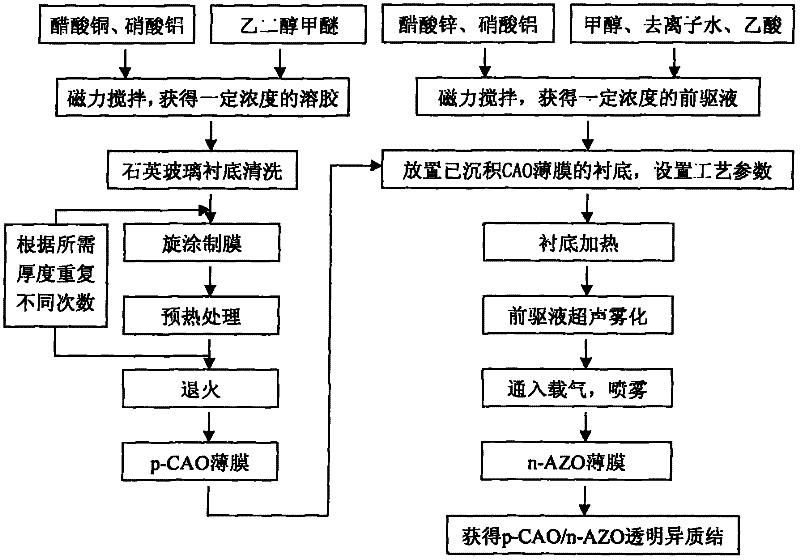
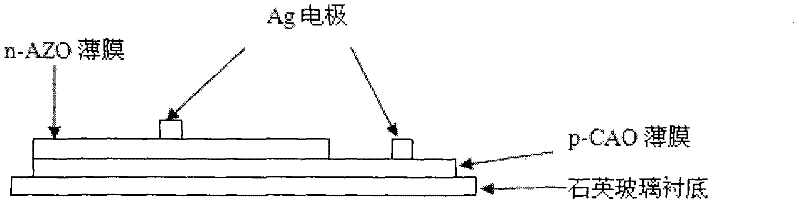
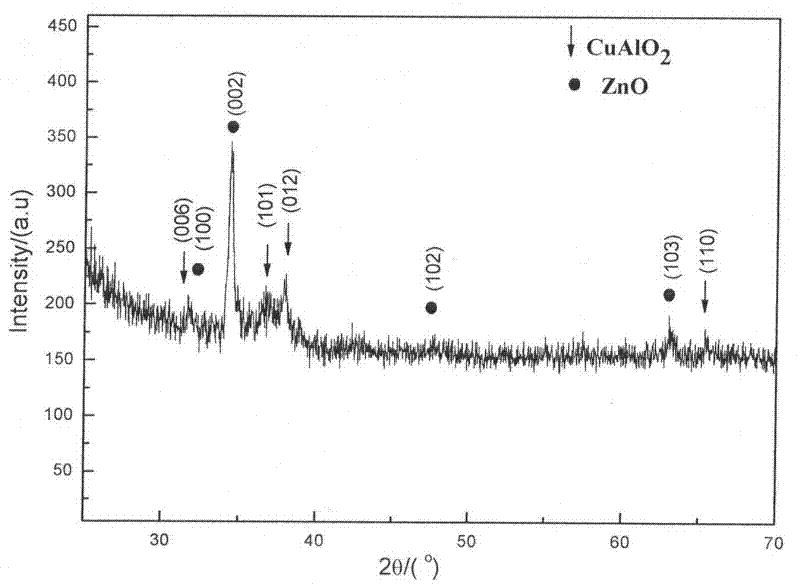
Preparation method of p-CuAlO2/n-ZnO:Al transparent thin film heterojunction of glass substrate
InactiveCN102244010AWith p-n rectification characteristicsExcellent electrical performanceSemiconductor/solid-state device manufacturingHeterojunctionFree cooling
The invention discloses a preparation method for a p-CuAlO2 / n-ZnO:Al transparent thin film heterojunction of a glass substrate. The preparation method comprises the following steps of: (1) preparing a p-CAO transparent conductive thin film by adopting a Sol-Gel process and preparing an n-AZO transparent conductive thin film by adopting an ultrasound spray pyrolysis (USP) process; (2) performing glue averaging, layering and preheating for many times during preparation of the p-CAO thin film; (3) annealing the p-CAO thin film under an argon environment; (4) depositing the n-AZO transparent conductive thin film on the substrate covered with the CAO thin film by adopting the USP process; and (5) heating the substrate during deposition of the n-AZO transparent conductive thin film, making the temperature of the substrate not more than 320 DEG C and naturally cooling the sample to obtain the p-CuAlO2 / n-ZnO:Al(p-CAO / N-AZO) transparent thin film heterojunction. The method is novel and simple and the requirement of a large-area film forming process can be met; furthermore, the prepared p-CAO / n-AZO heterojunction has a fully transparent structure, can realize a p-n node function and has high photoelectric performance.
Owner:GUILIN UNIV OF ELECTRONIC TECH
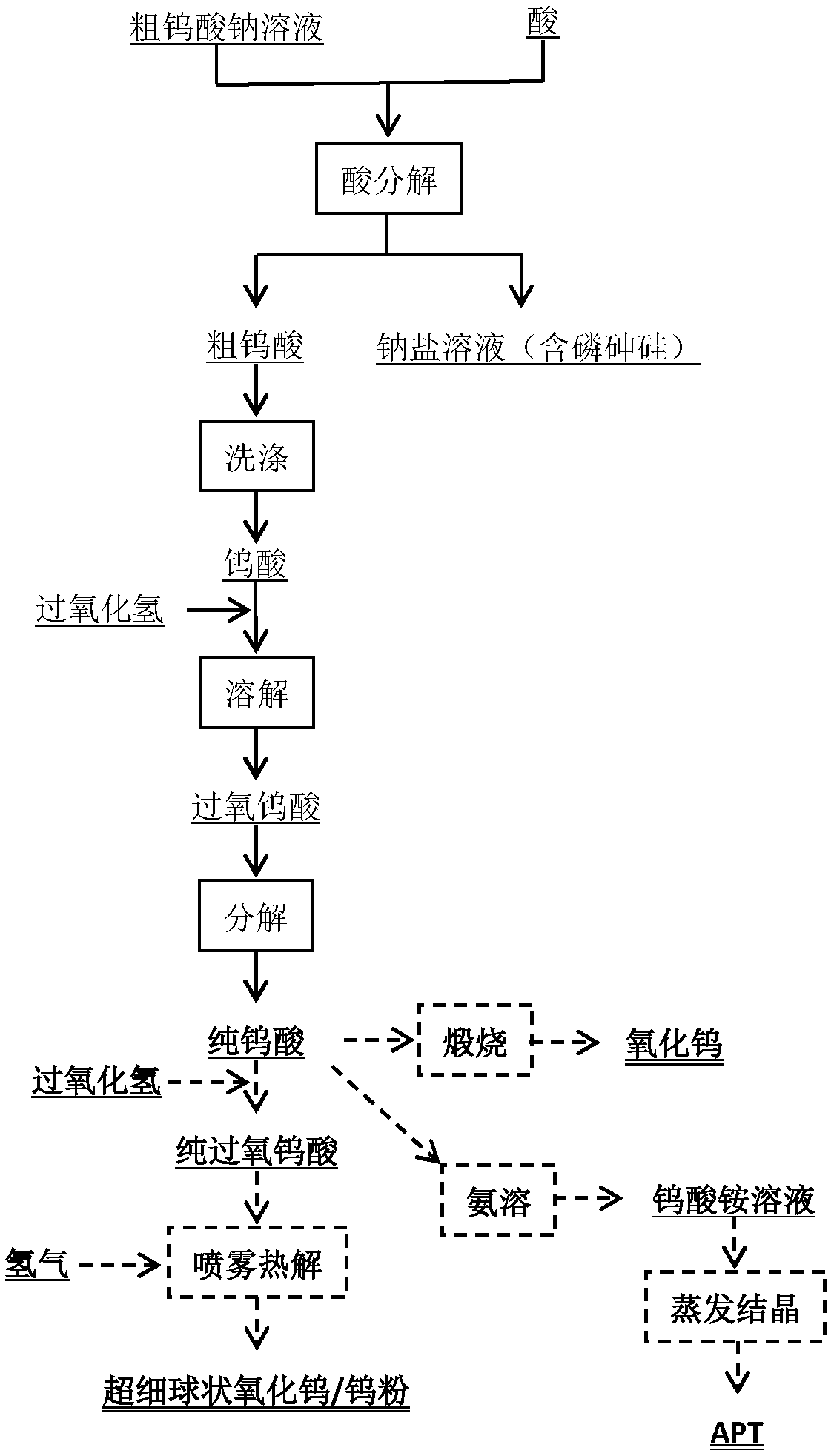
Method of producing tungsten product from crude sodium tungstate solution
ActiveCN108516588AReduce environmental costsAvoid emissionsTungsten compounds preparationTungsten oxides/hydroxidesUltrasonic spray pyrolysisSodium tungstate
The invention relates to a method of producing a tungsten product from a crude sodium tungstate solution. The method includes steps of: adding a proper amount of acid to the crude sodium tungstate solution to generate precipitate, and filtering the solution to obtain crude tungstic acid and a filtrate; recycling tungsten from the filtrate through a weak-alkaline anion exchange resin or with an extraction agent; extracting the crude tungstic acid with H2O2, and heating a peroxytungstic acid solution or feeding SO2 to prepare tungstic acid; repeating the step for several times until preparing pure tungstic acid; preparing WO3 or tungsten powder from the pure tungstic acid; dissolving the tungstic acid in H2O2 solution to obtain the peroxytungstic acid solution; and performing ultrasonic spray pyrolysis to obtain tungsten oxide powder or tungsten powder. The method has great separation effect, is simple and controllable in operation process, and is easy to promote and apply in industry.
Owner:CENT SOUTH UNIV
Integrative ultrasonic spray pyrolysis large-area wide-temperature zone coating device
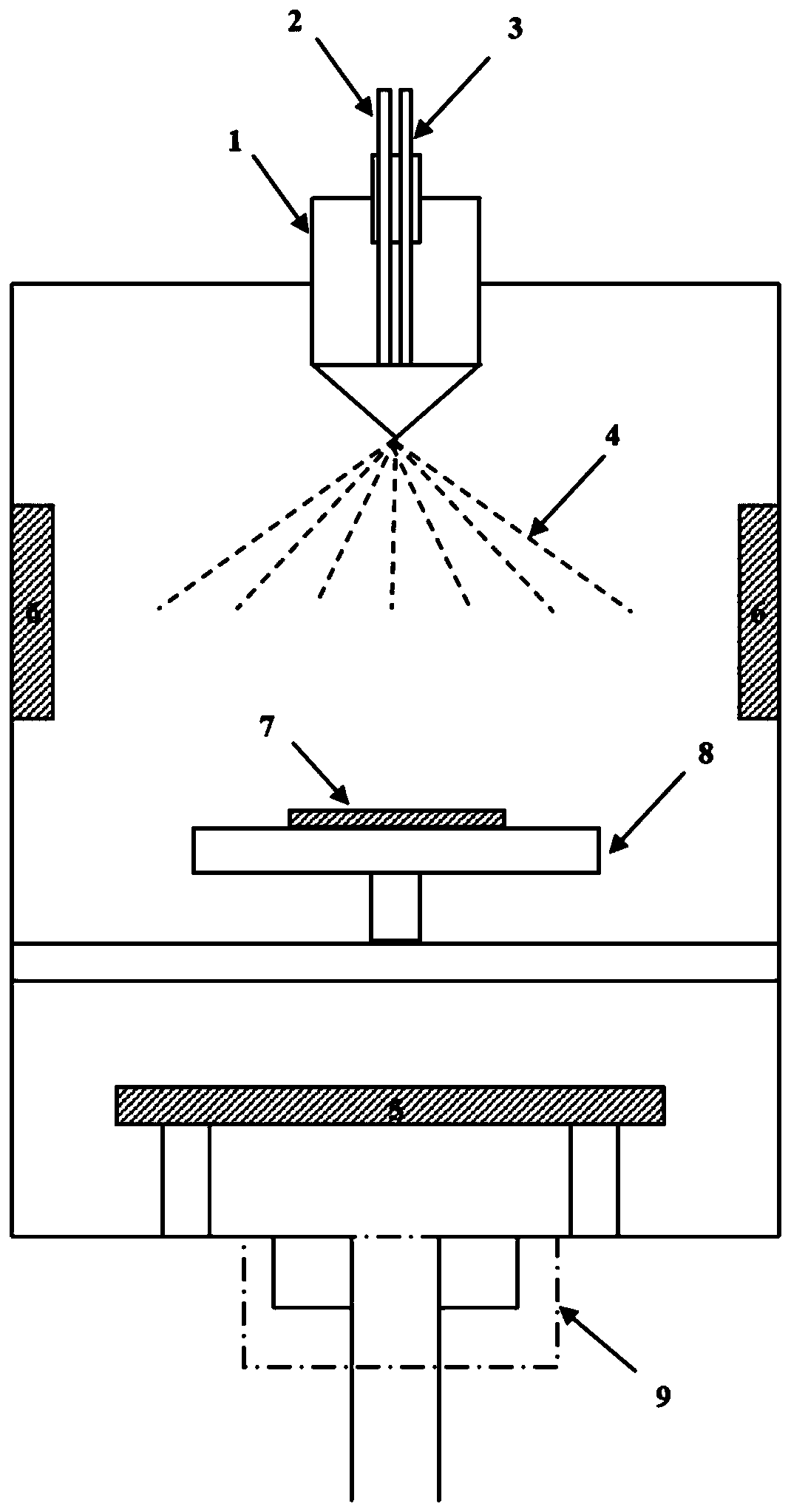
InactiveCN101759372AOvercome the problem of usingOvercoming frequencyLiquid/solution decomposition chemical coatingSpray nozzleEngineering
The invention discloses an integrative ultrasonic spray pyrolysis large-area wide-temperature zone coating device, comprising a frame, an air tank and an infusion pump arranged on the frame. A three-dimensional motion mechanism and a control module for controlling the three-dimensional motion mechanism to do three-dimensional motion are arranged above the frame. A temperature-controllable heating platform is arranged in the frame and under the three-dimensional motion mechanism. An integrative ultrasonic spray nozzle system capable of high-temperature coating is arranged between the heating platform and the three-dimensional motion mechanism and is toward the heating platform, and the infusion pump is communicated with the integrative ultrasonic spray nozzle system which is glidingly connected with the three-dimensional motion mechanism. Utilizing an integrative ultrasonic spray nozzle, the coating device solves the problems of high ultrasonic frequency, uneven gas spray and complex organization existing in the traditional ultrasonic spray mode. Utilizing a peripheral cooling gas nozzle, the coating device solves the problem that the integrative ultrasonic spray nozzle is usually used in low temperature and realizes high-temperature film formation.
Owner:GUANGZHOU INST OF ENERGY CONVERSION - CHINESE ACAD OF SCI
Method for extracting tungsten-molybdenum from high-molybdenum scheelite
ActiveCN108425011AReduce break down costReduce manufacturing costProcess efficiency improvementMolybdateTungstate
The invention relates to a method for extracting tungsten-molybdenum from high-molybdenum scheelite. The method comprises the following steps: leaching high-molybdenum scheelite with a sulfuric acid solution, and extracting molybdenum from the leaching liquid; extracting leaching residues with hydrogen peroxide so as to obtain a solution with peroxy-tungstate and peroxy-molybdate, and heating or introducing SO2 so as to obtain a solution with tungstate and peroxy-molybdate; dissolving the tungstate with hydrogen peroxide, heating the obtained solution, and introducing SO2 so as to obtain tungstate and a solution with peroxy-molybdate; repeating the operation for multiple times till pure tungstate is prepared; preparing tungsten trioxide or tungsten powder from the pure tungstate; heating the solution with the peroxy-molybdate, or introducing SO2, dissolving the obtained molybdate with a hydrogen peroxide solution so as to obtain a peroxy-molybdate solution, and performing ultrasonic spray pyrolysis, so as to obtain a molybdenum trioxide powder or a molybdate powder. The method is good in separation effect, simple in operation process, easy in operation process control, and easy inindustrial popularization and application.
Owner:CENT SOUTH UNIV
Active carbon material with spherical structure and preparation method and applications thereof
ActiveCN107808955ALarge specific surface areaImprove electrochemical performanceCell electrodesSecondary cellsLithium–sulfur batterySpray pyrolysis
The invention discloses an active carbon material with a spherical structure. The active carbon material is prepared by subjecting saccharides and a carbon material to ultrasonic dispersion, spray pyrolysis, and carbonization, wherein the mass ratio of saccharides to the carbon material is 1-5:1. Through an ultrasonic spray pyrolysis method, the active carbon material is prepared and is taken as an active substance carrier for the cathode material of a lithium sulfur battery. The prepared active carbon material has a spherical structure, which is formed by the self-supporting effect of the carbon material; cavities exist in the spheres, the specific surface area is large, the dispersibility is good, and the electrochemical properties of the cathode material are improved effectively. Moreover, the operation of the preparation method is simple, the cost is low, and the preparation method is energy saving and environmentally friendly. The pore structure of the prepared active carbon material is abundant. The active carbon material is taken as an active substance carrier for a lithium sulfur battery, which has the advantages of high electrochemical specific capacity, excellent cycle performance, stable structure, and environmental friendliness.
Owner:HUNAN UNIV OF TECH
Preparation of low-resistance / high-resistance composite film through ultrasonic spray pyrolysis
InactiveCN1544691APromote decompositionPromote oxidation reactionLiquid/solution decomposition chemical coatingHigh resistanceElectrical resistance and conductance
The invention prepares transparent low-distance and high-distance films by ultrasonic spray thermolysis technique. It uses a technique of ultrasonic spray deposition, and has the characters of large area and high deposition ratio. By improving the film forming device by ultrasonic spray thermolysis, it changes the spout from one point to one slot, which makes gas uniformly ejected on a line. The sample moves at uniform speed in the direction of the vertical spraying slot so that the thickness of the deposited film can be made uniform and consistent. It can be used in preparation of the transparent low-resistance films and transparent intrinsic high-resistance films of solar energy batteries and LCDs.
Owner:SICHUAN UNIV
Method for directly preparing samarium-iron alloy powder through spray pyrolysis reduction method
ActiveCN108274016AMix and disperse evenlyPromote formationTransportation and packagingMetal-working apparatusDispersityIron salts
The invention discloses a method for directly preparing samarium-iron alloy powder through a spray pyrolysis reduction method. A mixed aqueous solution of samarium salt and iron salt is used as a spray precursor solution, spherical samarium-iron composite oxides are prepared through the ultrasonic spray pyrolysis method, and samarium-iron alloy is obtained through hydrogen prereduction and then calcium reduction or directly through calcium reduction. According to the method, the cost of raw materials is low, the mole ratio of samarium and iron in the samarium-iron alloy can be easily obtainedby adjusting the proportion of the samarium salt and the iron salt in the precursor solution, the samarium and iron dispersity during reduction diffusion is good, the reduction temperature is low, thereaction time is short, samarium volatilization can be effectively reduced, and agglomeration of the samarium-iron alloy powder can be effectively restrained. According to the method, the pure-phasesamarium-iron alloy powder with the size being 0.5-10 micrometers and in the form of nearly spherical particles can be directly prepared without the need of a follow-up ball-milling crushing process,samarium-iron alloy grains are complete, following improvement on the performance of samarium iron nitrogen is facilitated, and samarium iron nitrogen has good flowing machinability when combined withmacromolecules.
Owner:ZHEJIANG UNIV OF TECH
FeS2 nano-material with three-dimensional structure, and preparation method and application thereof
ActiveCN107123807AImprove electrochemical activityGood electrical propertiesMaterial nanotechnologyCell electrodesUltrasonic spray pyrolysisHydrothermal synthesis
The invention discloses a FeS2 nano-material with a three-dimensional structure. The FeS2 nano-material is FeS2 nanochains with a three-dimensional porous structure or FeS2 nano-pedals with a three-dimensional structure. The invention also provides a method for preparing the FeS2 nano-material. For example, the FeS2 nanochains are prepared through a magnetic field-assisted ultrasonic spray pyrolysis method; or, the FeS2 nano-pedals are prepared through a hydrothermal synthesis method. The FeS2 nano-material provided by the invention has a three-dimensional structure, has high electrochemical activity, excellent electric properties and high mechanical strength, and has advantages of pure phase, high degree of crystallization, no impurity and the like. In addition, the preparation technology is simple and low-cost, has high output, and is suitable for large-scale production.
Owner:SUZHOU INST OF NANO TECH & NANO BIONICS CHINESE ACEDEMY OF SCI
Process for preparing nitrigen-aluminium co-blended hole zinc oxide thin film material
InactiveCN1783433AEasy to operateAbundant Al raw materialsLaser detailsSemiconductor/solid-state device manufacturingInorganic saltsNitrogen
This invention relates to a NAL common doped cavity ZnO film and a preparing technology, which applies the supersonic spray pyrogenation method and takes the mixed solution of the organic or inorganic solution of Zn, the organic or inorganic solution of N and the inorganic solution of Al as the precursor to control the electric performance of type-P ZnO film by regulating and controlling the substrate temperature at 600-900deg.C, the movability can reach to 103cm at the maximum.
Owner:SHANGHAI INST OF CERAMIC CHEM & TECH CHINESE ACAD OF SCI
Method for synthesis of silica loaded nickel-platinum catalyst by ultrasonic spray pyrolysis
ActiveCN103223347AHigh activityGood dispersionCatalyst activation/preparationMetal/metal-oxides/metal-hydroxide catalystsDispersitySilicon dioxide
The invention relates to a method for synthesis of a silica loaded nickel-platinum catalyst by ultrasonic spray pyrolysis. The method includes: step 1. preparing a nickel-platinum-containing soluble compound solution; step 2. atomizing the nickel-platinum-containing soluble compound solution; step 3. vaporizing silicon tetrachloride; step 4. bringing the atomized nickel-platinum liquid drops and silicon tetrachloride vapor to undergo a pyrolytic reaction so as to generate nanoscale silica, simultaneously making the active nickel-platinum-containing soluble compound form catalyst particles under high temperature and loading them on the nano-silica; and step 5. collecting and drying the catalyst. The method provided in the invention solves the problems that a silica carrier is expensive and the loading process is complex, has a simple synthesis process, and the obtained nano-silica loaded nickel-platinum catalyst has high activity and good dispersity.
Owner:TSINGHUA UNIV
Ultrasound spray pyrolysis preparation method of MgO buffer layer for coating conductor
InactiveCN104120411AHigh speedReduce manufacturing costSuperconductors/hyperconductorsLiquid/solution decomposition chemical coatingElectrical conductorMetallurgy
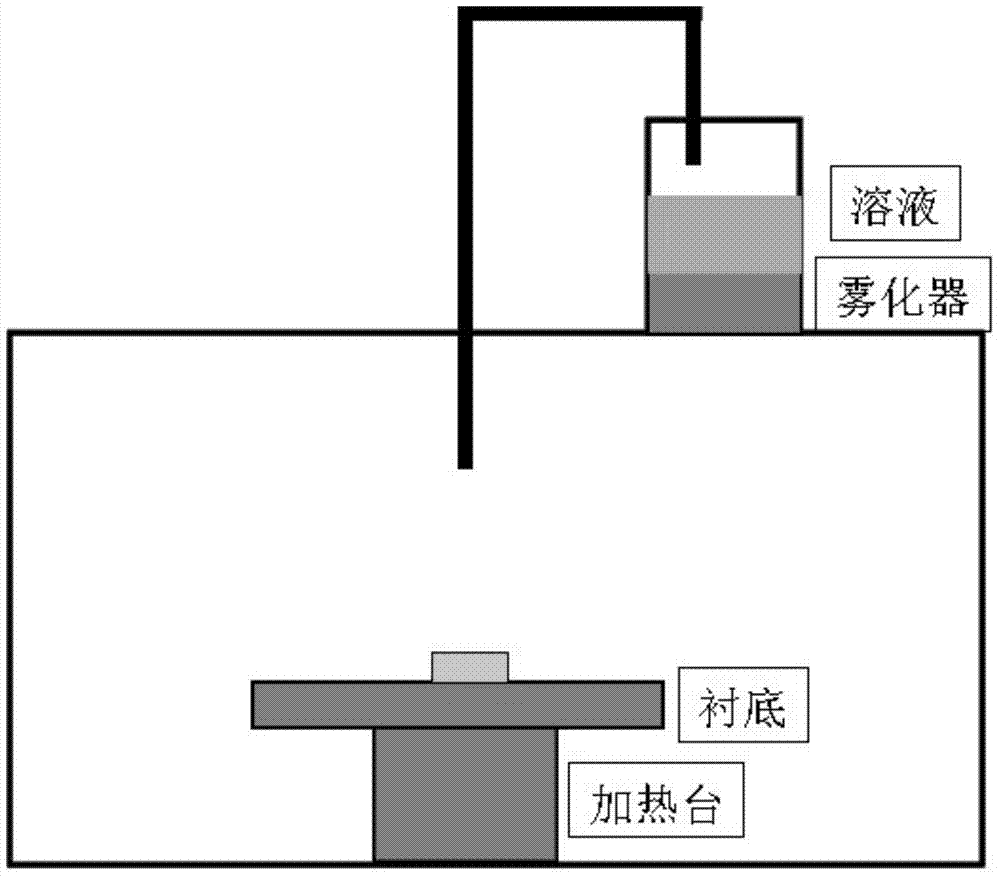
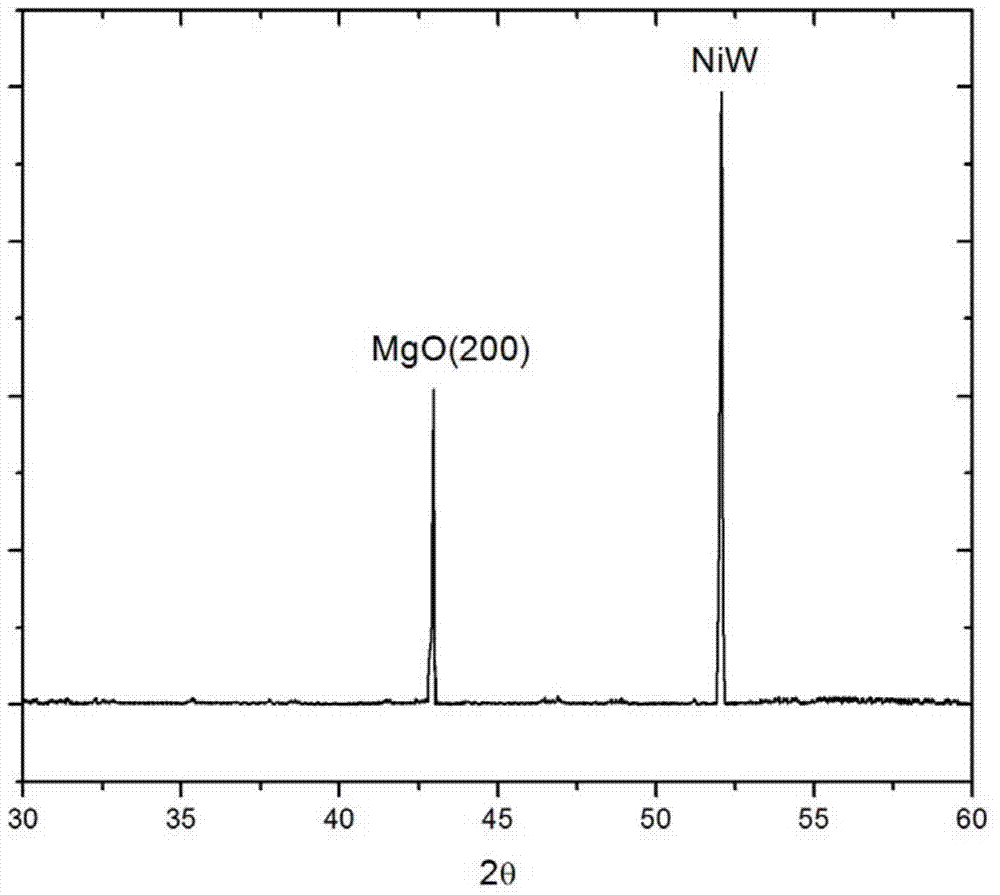
The invention discloses an ultrasound spray pyrolysis preparation method of a MgO buffer layer for a coating conductor. The ultrasound spray pyrolysis preparation method comprises the steps of: with a magnesium nitrate solution as a precursor solution, epitaxially growing the MgO buffer layer on a NiW alloy substrate by using an ultrasound spray pyrolysis technology; fixing the cleaned NiW alloy substrate on a heating platform, and heating the cleaned NiW alloy substrate to 580-630 DEG C; with the magnesium nitrate solution as the precursor solution, atomizing and spraying the precursor solution on the NiW alloy substrate in a way of spraying for 20-60s, stopping for 40-120s and then spraying again by using an ultrasound atomizer, and finally preserving the heat for 30-60min to obtain the MgO buffer layer, wherein the spraying operation lasts for 10-30min in total. The method is free of vacuum systems, high in film forming speed and capable of reducing the production cost and increasing the production efficiency.
Owner:NANJING UNIVERSTIY SUZHOU HIGH TECH INST
Improved process of ultrasonic spray pyrolysis deposition of one or more electrochromic and/or electrolytic films on a substrate
ActiveUS20170363928A1High layer uniformityEasy to coverEnergy inputOxide conductorsElectricityElectrolysis
A process of making an electrochromic or an electrolytic film by Ultrasonic Spray Pyrolysis (USP) deposition on a substrate comprising:mixing a surfactant to an aqueous precursor solution comprising an electrochromic component or an electrolytic component to provide a spray solution;introducing the spray solution into an ultrasonic spray deposition nozzle at a constant flow rate between 0.1 mL / min and 2 mL / min and applying an ultrasonic frequency between 80 and 120 kHz to generate atomized droplets of the precursor solution;entraining the atomized droplets with a controlled jet of air as gas carrier at a pressure between 0.50 to 2.0 psi, onto a pre-heated substrate at a temperature of 200 to 450° C.;thermally converting the atomized droplets when depositing onto the pre-heated substrate to generate an electrochromic or an electrolytic film.
Owner:INISMA +1
Method for preparing porous carbon materials with different morphologies through ultrasonic spray pyrolysis regulation and control
InactiveCN110790254ANice appearanceUniform size distributionCarbon preparation/purificationPorous carbonOxide composite
The invention discloses a method for preparing porous carbon materials with different morphologies by ultrasonic spray pyrolysis regulation and control. The method comprises the following steps: dissolving a soluble metal salt and a soluble sugar in water to prepare a precursor solution; ultrasonically atomizing the precursor solution into fog drops, and then introducing the fog drops into pyrolysis equipment by using a carrier gas for pyrolysis to obtain a metal oxide composite porous carbon material; and dissolving and cleaning the metal oxide in the metal oxide composite porous carbon material with an acid to obtain the porous carbon materials with different morphologies. The porous carbon materials with different morphologies can be regulated and controlled by changing the proportion of precursors, and the porous carbon materials with different specific surface areas and pore diameters are obtained. The prepared material is attractive in appearance and uniform in size distribution,the method has the advantages of being simple in process, easy to control in reaction, low in cost and free of pollution, a large number of good porous carbon materials with different appearances canbe continuously prepared, and the method is very suitable for large-scale production.
Owner:HUNAN UNIV
Method for extracting molybdenum from crude molybdic acid and preparing molybdenum product
ActiveCN108728674AReduce environmental costsImprove product added valueProcess efficiency improvementDecompositionDissolution
The invention relates to a method for extracting molybdenum from crude molybdic acid and preparing a molybdenum product. The method comprises the steps that the crude molybdic acid is dissolved with ahydrogen peroxide solution, and an obtained peroxo-molybdic acid solution is heated or SO2 is led into the obtained peroxo-molybdic acid solution until molybdic acid is completely decomposed from peroxo-molybdic acid; filtering is carried out to obtain the molybdic acid; dissolution is carried out by hydrogen peroxide, and decomposition and purification are carried out; the steps of dissolution,decomposition and purification are repeated several times until pure molybdic acid is prepared; calcinations is carried out to prepare molybdenum trioxide; and or the obtained pure molybdic acid is dissolved with hydrogen peroxide, filtering is carried out to obtain a pure peroxo-molybdic acid solution, and then ultrasonic spray pyrolysis is carried out to prepare molybdenum trioxide powder or toprepare molybdenum powder. According to the method, the separation effect is good, the operation process is simple and easy to control, and the method is easy to be industrialized and popularized.
Owner:CENT SOUTH UNIV
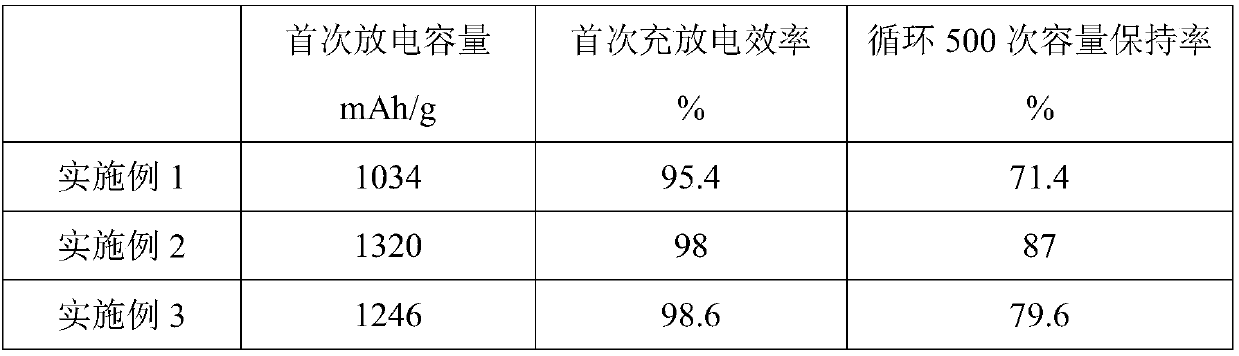
Preparation method of anode material LiNi0.5Mn0.5O2 of battery
The invention discloses a preparation method of an anode material LiNi0.5Mn0.5O2 of a battery. The preparation method includes the following steps: a, dissolving nickel nitrate, manganous nitrate and lithium nitrate with the molar ratio of 0.5: 0.5: (1.05-1.2) in deionized water to form solution, adding proper amount of NH4NO3 into the solution and evenly stirring the solution; b, performing ultrasound spray pyrolysis on the solution prepared in the step a to obtain powder, wherein the ultrasonic frequency is 1.8-2 MHz, and the pyrolysis temperature is 950-990 DEG C; and c, calcining the powder obtained in the step b for 5-12 hours at the temperature of 700-900 DEG C to obtain finished products. Nitrate of nickel, manganese and lithium serves as the raw materials, and the deionized water serves as the solvent, so that the raw materials are fully mixed in molecular state, the raw materials are evenly mixed by means of ultrasonic spray pyrolysis, and the preparation steps are simplified. The preparation method has the advantages of being even in raw material mixing and simple in preparation method.
Owner:成都兴能新材料股份有限公司
Preparation method of novel composite flat plate electric heating film
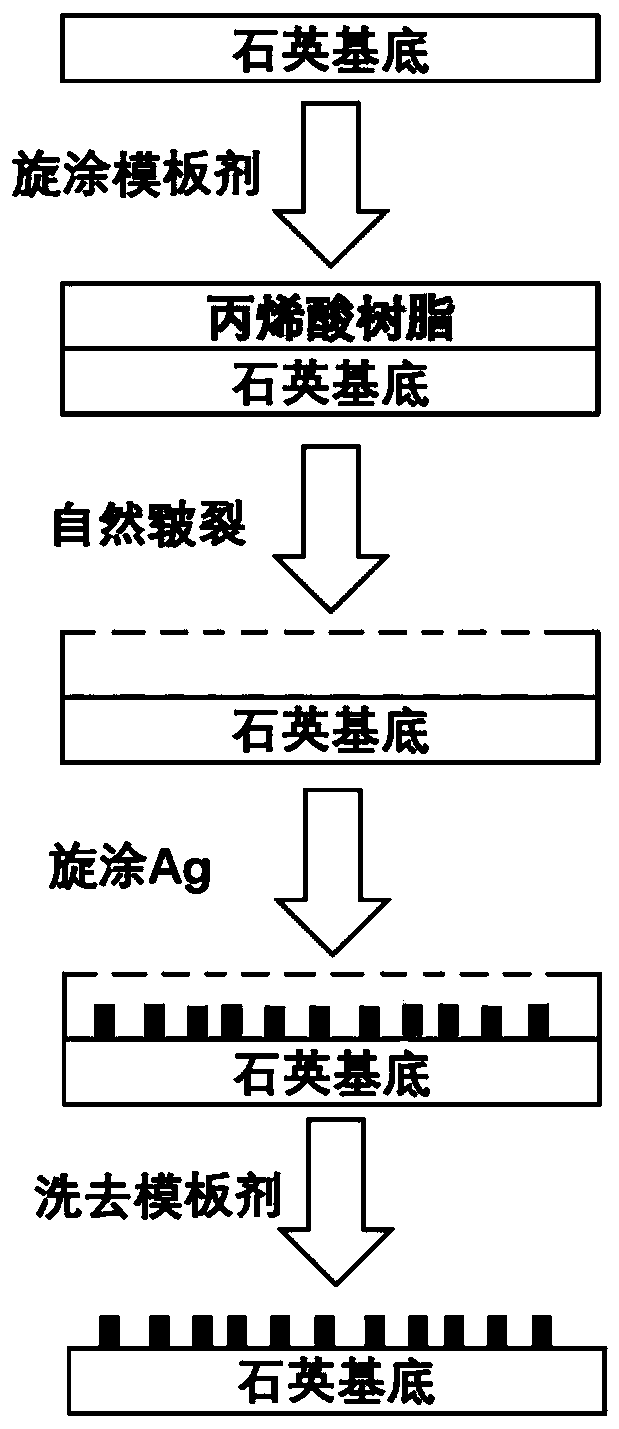
InactiveCN109862631AThickness is easy to controlUniform structureOhmic-resistance electrodesSpecial surfacesAcrylic resinLow voltage
The invention discloses a preparation method of a novel composite flat plate electric heating film. The method comprises the steps of dissolving butyltin trichloride (MBTC), SbCl3 and NH4F which are used as a precursor in a solvent prepared from ethyl alcohol and water according to a proportion so as to use as a first precursor liquid; dispersing silver nanoparticle in deionized water to use as asecond precursor liquid; preparing acrylic resin to form a template agent, dropwise adding the template agent onto a quartz substrate, and drying to form a chapped template network; dropwise adding and spin-coating the second precursor liquid onto a substrate for forming a template, and performing annealing after drying; depositing the first precursor liquid onto the substrate and performing thermal processing by a method of ultrasonic spray pyrolysis; and fabricating an electrode to from an electric heating film device. The method has the advantages that the novel composite flat plate electric heating film has relatively high oxidization-resistant performance by a tin oxide thin film at the outermost layer, and stable film layer resistance during the application process can be maintained;the silver nanoparticle has excellent electric and heat conversion efficiency, a relatively high temperature can be achieved under a low voltage; and meanwhile, the novel composite flat plate electric heating film has excellent light transmittance with the adoption of the quartz substrate, and the light transmittance reaches 80% or above.
Owner:ZHEJIANG UNIV
Manufacturing method for semiconductor gas-sensing device of testing carbon dioxide
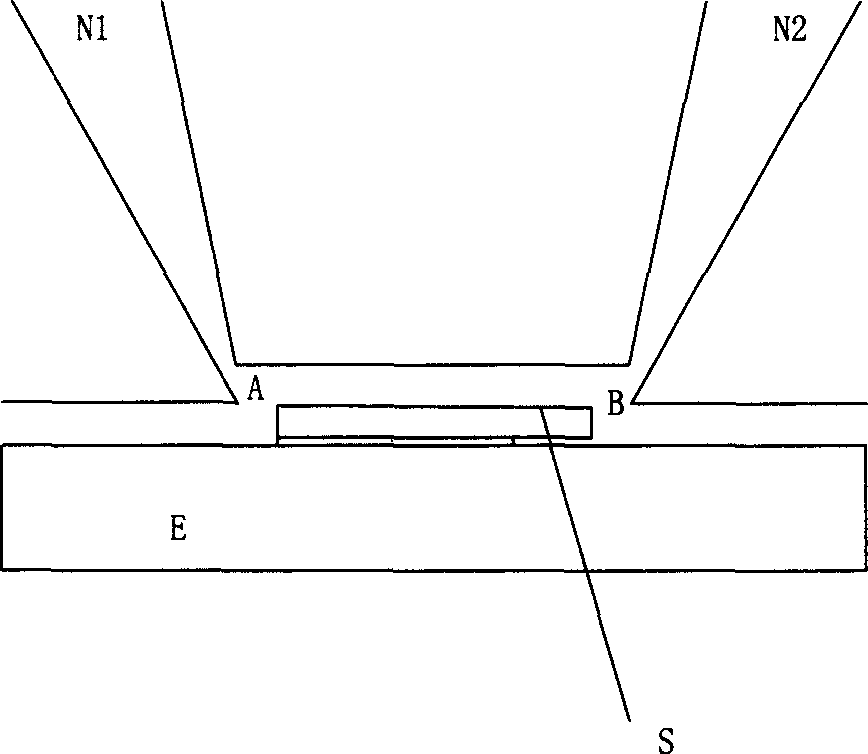
InactiveCN1424779ASimple structureSimple processSolid-state devicesMaterial resistanceIndiumNitrogen gas
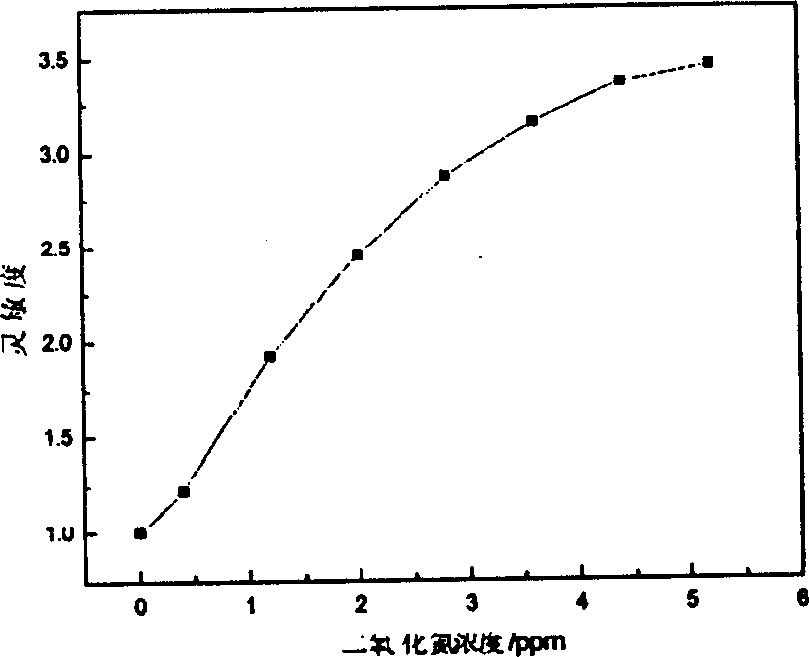
A gas-sensitive element of semiconductor sensor for detecting NO2 gas is an indium tin oxide film on quartz substrate having Au electrodes and heating layer of ruthenium oxide resistor. Its preparing steps includes dissolving indium in nitric acid, adding SnCl4, diluting with deionized water to obtain reaction liquid, loading said quartz substrate in tubular furnace, atomizing said reaction by ultrasonic oscillator, carrying the atomized reaction liquid into tubular furnace by N2, and spraying it toward quartz substrate. It has high sensitivity.
Owner:SHANGHAI UNIV
Copper tungstate photo-anode film and preparation method thereof
InactiveCN112695304AUniform thicknessImproved visible light responsivenessLiquid/solution decomposition chemical coatingElectrodesTungstateAmmonium metatungstate
The invention relates to the technical field of photoelectrocatalysis, and provides a preparation method of a copper tungstate photo-anode film. The preparation method comprises the following steps: mixing copper nitrate trihydrate, ammonium metatungstate and water to obtain a precursor solution; carrying out ultrasonic spray pyrolysis coating on the obtained precursor solution to form a copper tungstate precursor film on the substrate; and carrying out heat treatment on the obtained copper tungstate precursor film to obtain the copper tungstate photo-anode film. According to the preparation method, the precursor solution can be atomized into nanoscale small liquid drops by adopting an ultrasonic spray pyrolysis coating method, the solid film with uniform thickness is finally formed on the substrate through reaction processes such as solvent evaporation and solute thermal decomposition, crystallization of the precursor film can be promoted during heat treatment, and the uniformity of the structure of the copper tungstate photo-anode film is further improved, so that the visible light responsiveness of the copper tungstate photo-anode film can be improved. Experimental results show that the copper tungstate photo-anode film prepared by the preparation method provided by the invention is uniform in thickness and has excellent photoelectric properties.
Owner:NORTH CHINA UNIVERSITY OF SCIENCE AND TECHNOLOGY
Room-temperature high-selectivity NO2 sensor based on three-dimensional Ti3C2Tx/rGO composite folded ball and preparation method thereof
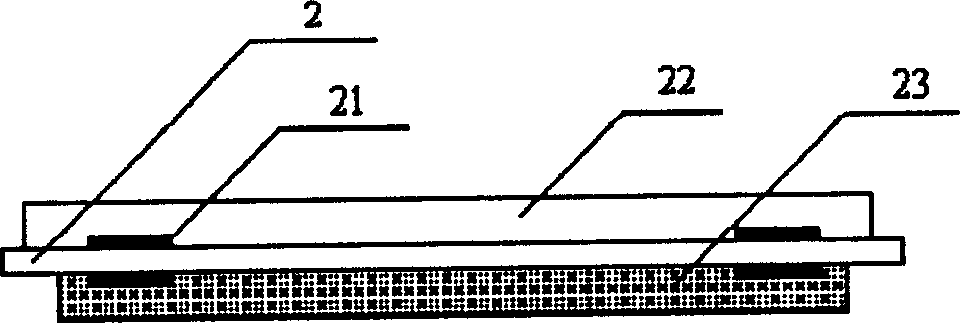
InactiveCN113834863AHigh selectivityReduce specific surface area lossMaterial resistancePolyimide substrateHeterojunction
The invention discloses a room-temperature high-selectivity NO2 sensor based on a three-dimensional Ti3C2Tx / rGO composite folded ball and a preparation method thereof, and belongs to the technical field of gas sensors. The sensor is composed of a polyimide substrate with an Au interdigital electrode and sensitive electrodes prepared on the interdigital electrode and the substrate. The three-dimensional Ti3C2Tx / rGO composite folded ball is synthesized by adopting an ultrasonic spray pyrolysis technology, and on the basis of forming an anti-aggregation pleated ball to reduce the loss of the specific surface area, a uniform rGO / TiO2 heterojunction is also formed on the folded ball, so that sensing sites are increased. The material shows the property of a p-type semiconductor, and the response direction of NO2 is opposite to that of VOCs and NH3, so that the selectivity of NO2 is further enhanced undoubtedly. Meanwhile, the three-dimensional Ti3C2Tx / rGO composite folded ball has higher NO2 response and lower detection limit compared with a pure Ti3C2Txfolded ball and a pure rGO folded ball.
Owner:JILIN UNIV
Preparation method of wrinkly nano-rGO composite material, and preparation method of wrinkly nanometer metal oxide
InactiveCN109174102AIncreased free volumeImprove stress resistanceManganese oxides/hydroxidesNickel oxides/hydroxidesShielding gasSpray pyrolysis
The invention discloses a preparation method of a wrinkly nano-rGO composite material. The preparation method of the wrinkly nano-rGO composite material comprises the following steps: (1) adding a metal salt to a GO dispersion, and performing uniform mixing to prepare a mixed solution; (2) performing ultrasonic spray pyrolysis on the mixed solution, and collecting prepared black powder to obtain the wrinkly nanometer metal oxide / rGO composite material. A preparation method of the wrinkly nanometer metal oxide is characterized in that the wrinkly nanometer metal oxide / rGO composite material iscalcined in a protective gas atmosphere to obtain the wrinkly nanometer metal oxide with a wrinkly spitball-shaped microstructure. The nanometer metal oxide / rGO composite material is prepared in a one-step manner through combining a spray pyrolysis process with a template process with GO and a metal salt as a precursor, and is calcined to prepare the wrinkly nanometer metal oxide. The preparationmethods have the advantages of simple process, short cycle, easily available raw materials and low cost, and the prepared metal oxide powder has a large surface area and rich pore structures.
Owner:UNIV OF SHANGHAI FOR SCI & TECH
Preparation method of ultra-fast-flickering ZnO film
InactiveCN104911706AImprove crystal orientationStrong ultraviolet emissionPolycrystalline material growthFrom chemically reactive gasesO-Phosphoric AcidUltraviolet
The invention relates to a preparation method of an ultra-fast-flickering ZnO film, which comprises the following steps: acid treatment on monocrystalline silicon wafer: measuring concentrated sulfuric acid and concentrated phosphoric acid, mixing the concentrated sulfuric acid and concentrated phosphoric acid, heating, putting the monocrystalline silicon wafer into the acid mixture, keeping the temperature for 10-40 minutes, naturally cooling, taking out the monocrystalline silicon wafer, cleaning with ethanol, carrying out ultrasonic cleaning, and carrying out blow-drying; and by using the treated monocrystalline silicon wafer as a substrate material, preparing a 40-60nm-thick ZnO film as a seed crystal layer on the substrate by a magnetron sputtering film deposition technique, and depositing a ZnO film on the ZnO seed crystal layer by an ultrasonic spray pyrolysis process, wherein the spray delivering carrier gas is N2, the deposition temperature is 370-390 DEG C, and the thickness of the ZnO film is 150-250nm. The ZnO film obtained by the method has the advantages of (002) preferred orientation characteristic, strong ultraviolet emission and ultra-fast flickering.
Owner:SHANGHAI INST OF TECH
Method for preparing micro-nano structure Prussian blue sodium battery positive electrode material through ultrasonic spray pyrolysis
InactiveCN112864382ALow costSimple processSecondary cellsPositive electrodesNano structuringElectrical battery
The invention provides a method for preparing a micro-nano structure Prussian blue sodium ion battery positive electrode material through ultrasonic spray pyrolysis. The method particularly relates to the following process: (1) preparation of a precursor: firstly preparing a nano-particle morphology electrode material by using a precipitation method, mixing the nano-particle morphology electrode material with deionized water, stirring, standing, filtering and re-dispersing the nano-particle morphology electrode material into the deionized water; and (2) ultrasonic spray pyrolysis: mixing the suspension prepared in the step (1) with LiOH, KOH, Na2CO3 and the like), putting the mixture into an ultrasonic atomizer, introducing the atomized solution into a high-temperature tubular furnace (heating to a target temperature of 200-400 DEG C), and collecting a product, thereby preparing the mesoporous spherical electrode material which has a proper specific surface area and is relatively uniform in size distribution and consists of nanoparticles. The preparation method disclosed by the invention is simple, low in raw material price and suitable for industrial production.
Owner:SHENGJIAN NO 83 FACTORY SHANDONG PROV
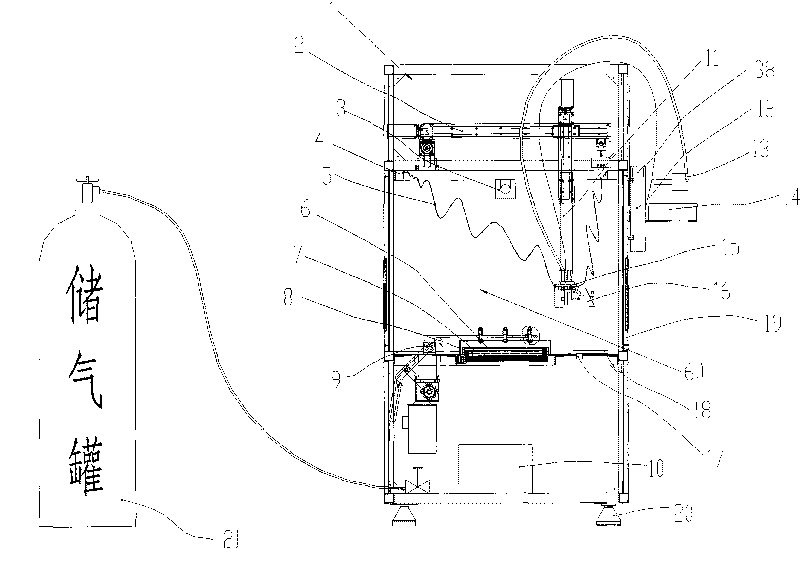
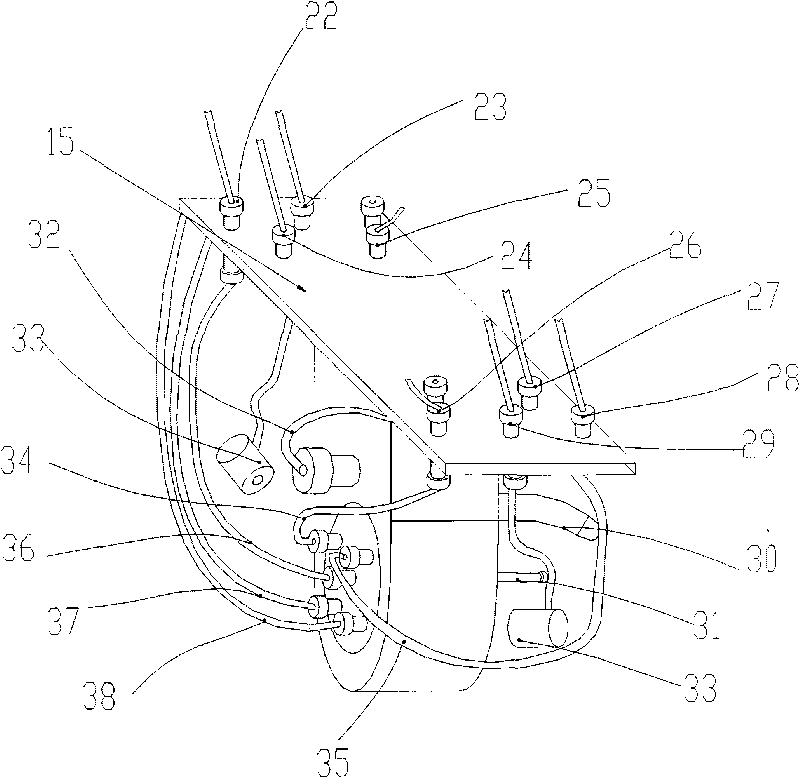
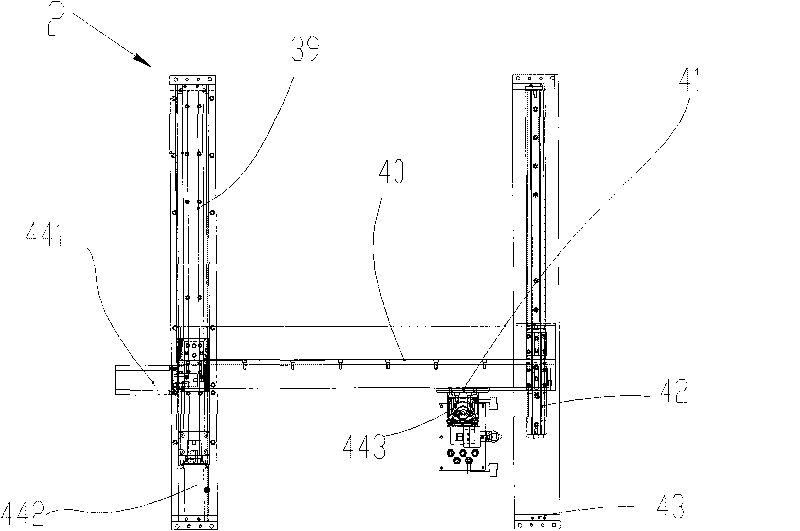
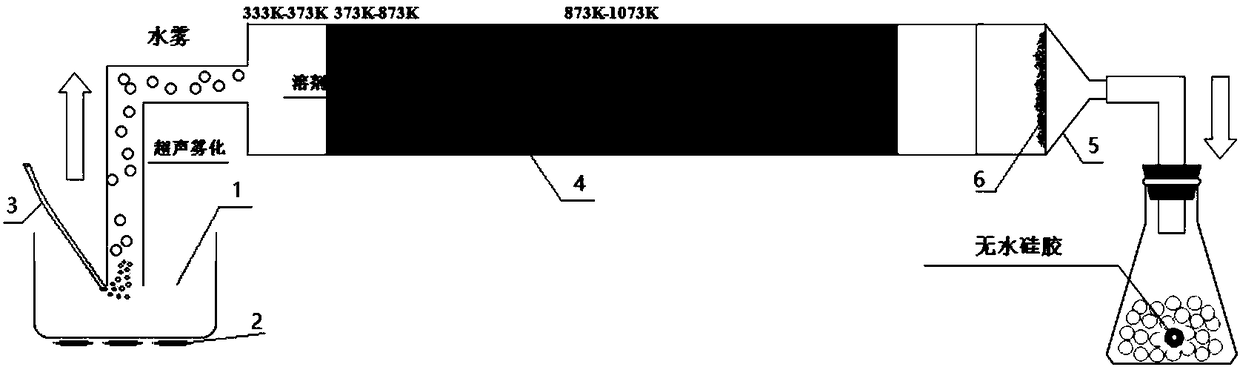
An ultrasonic spray pyrolytic coating device
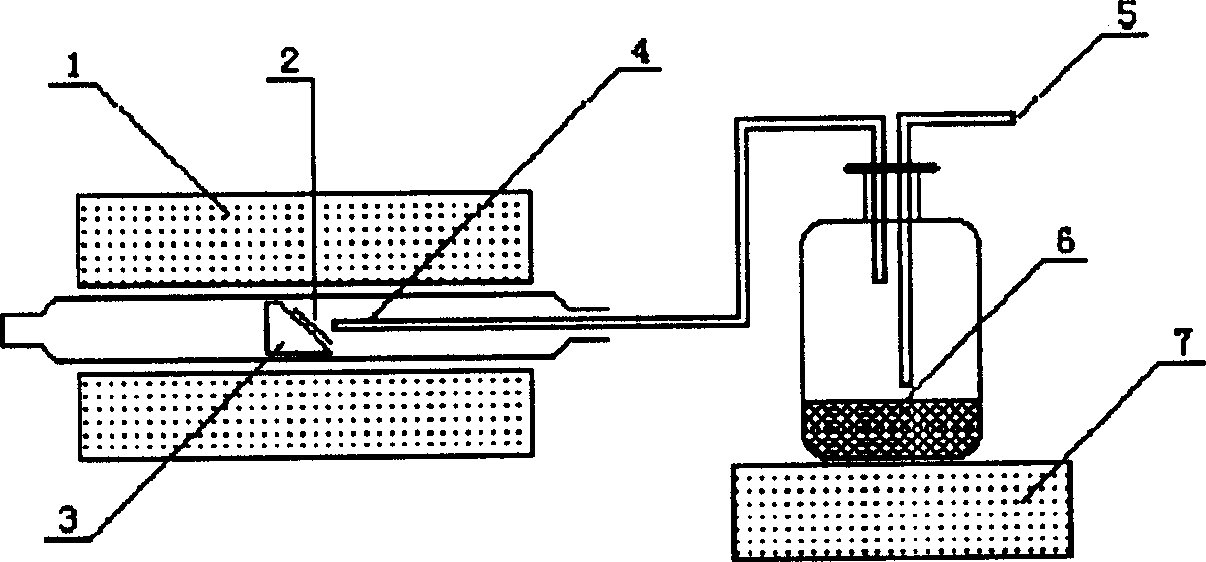
ActiveCN104762613BControl areaControl thicknessLiquid/solution decomposition chemical coatingSpray pyrolysisEngineering
The invention discloses an ultrasound spray pyrolysis film coating device. The ultrasound spray pyrolysis film coating device comprises a gas cabinet (1), an ultrasound atomizing device, a spray head and a heating platform (3) with a base plate (2). The ultrasound spray pyrolysis film coating device is characterized in that the gas cabinet (1) is communicated with the ultrasound atomizing device through a gas inlet pipe (4), the ultrasound atomizing device is communicated with the spray head through a pipeline (5), the spray head is located above the heating platform (3) and corresponds to the heating platform (3), an XY moving platform (13) is arranged below the heating platform (3), and a cooling device is arranged outside the ultrasound atomizing device. The ultrasound spray pyrolysis film coating device has the advantages of being ingenious in structural design, convenient to operate and low in cost. By means of the ultrasound spray pyrolysis film coating device, preparing of large films can be conducted for a long time, the area and the thickness of the films can be controlled, the water dropping problem in the film coating process is solved, the yield of the films is increased, and the quality of the films is improved.
Owner:重庆复源新材料科技有限公司
A method for directly preparing alumina-coated ferrite by high-pressure ultrasonic spray pyrolysis
ActiveCN109448979BThickness is easy to controlGood coating integrityInorganic material magnetismInductances/transformers/magnets manufactureThermal decomposition methodTube furnace
Owner:广州金意新材料有限公司
Preparation method for composite conductive film doped with tin oxide
InactiveCN108615585AIncrease the carrier concentrationImprove conductivityApparatus for manufacturing conducting/semi-conducting layersDopantOrganic solvent
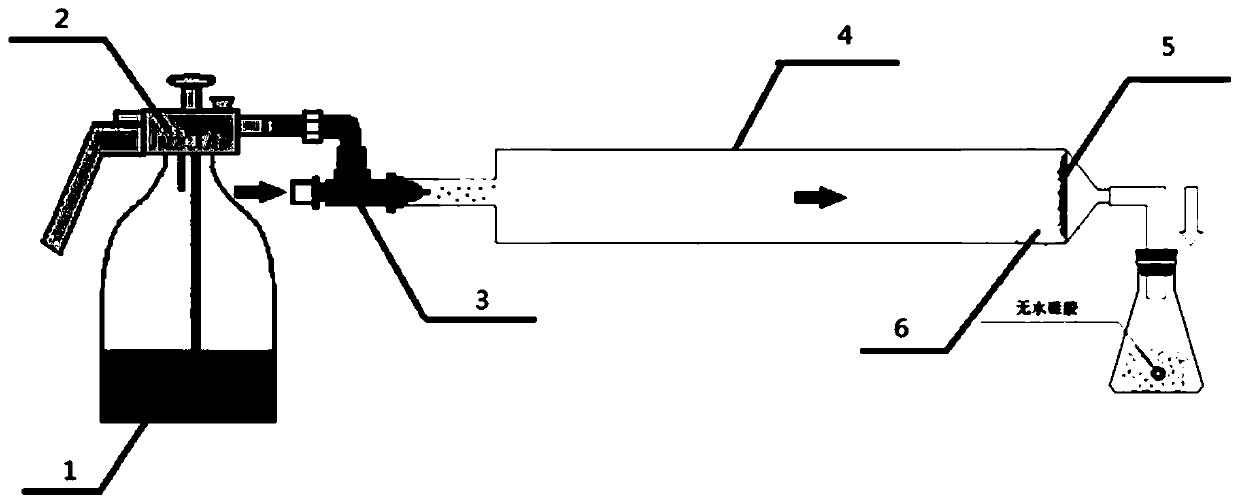
The invention relates to a preparation method for a composite conductive film doped with tin oxide, and the method comprises the steps: preparing mixed liquid of SnCl4, a dopant and an organic solvent, adding graphene to the mixed liquid for ultrasonic dispersion after the mixed liquid is placed quietly, coating the surface of the substrate with the mixed liquid through an ultrasonic spray pyrolysis coating machine, spraying the mixture containing no graphene, placing the substrate in an argon atmosphere for annealing under the temperature of 500 DEG C to obtain a tin oxide conductive film. The method solves a problem that the conductivity of the tin oxide conductive film is lower, employs simple equipment, and is easy to control. The prepared conductive film is uniform in surface, is compact in structure, is good in conductivity, and is firmly combined with the substrate.
Owner:YANCHENG INST OF TECH
Features
- R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
Why Patsnap Eureka
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Social media
Patsnap Eureka Blog
Learn More Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com