Patents
Literature
34 results about "2-bromoethanol" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
2-Bromoethanol can be used for the preparation of 2-bromoethyl glycosides from acetylated sugars. It can also be used as a starting material to synthesize 2-bromoethyl methoxymethyl ether. 2-Bromoethanol is used in selective reduction of nitroarenes (PcFe(II)/NaBH 4 /2-bromoethanol catalyst system).
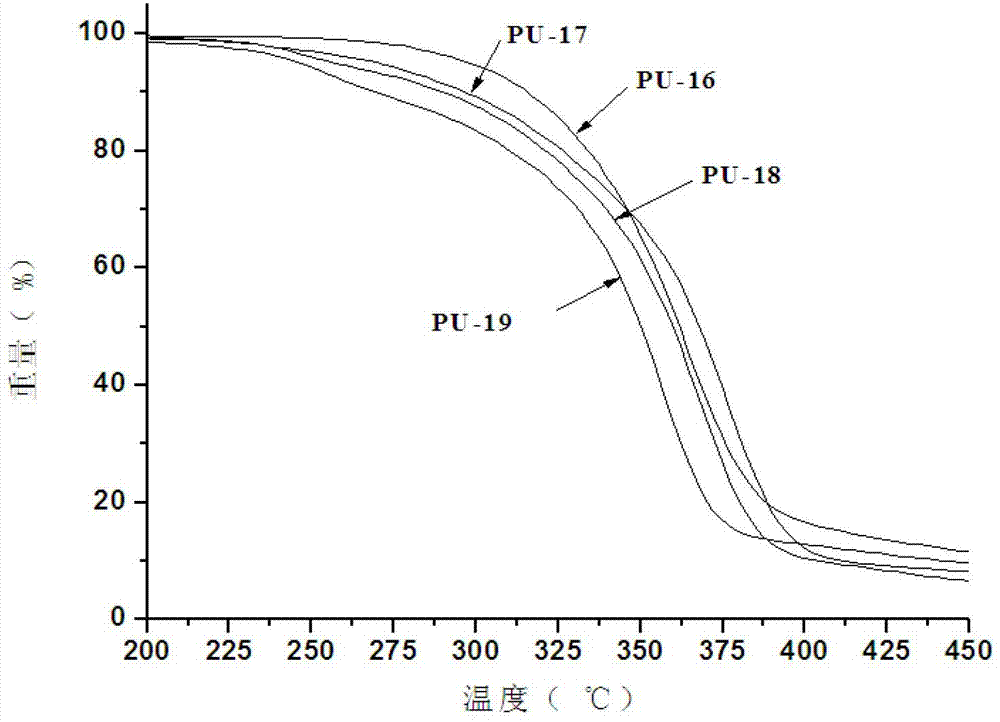
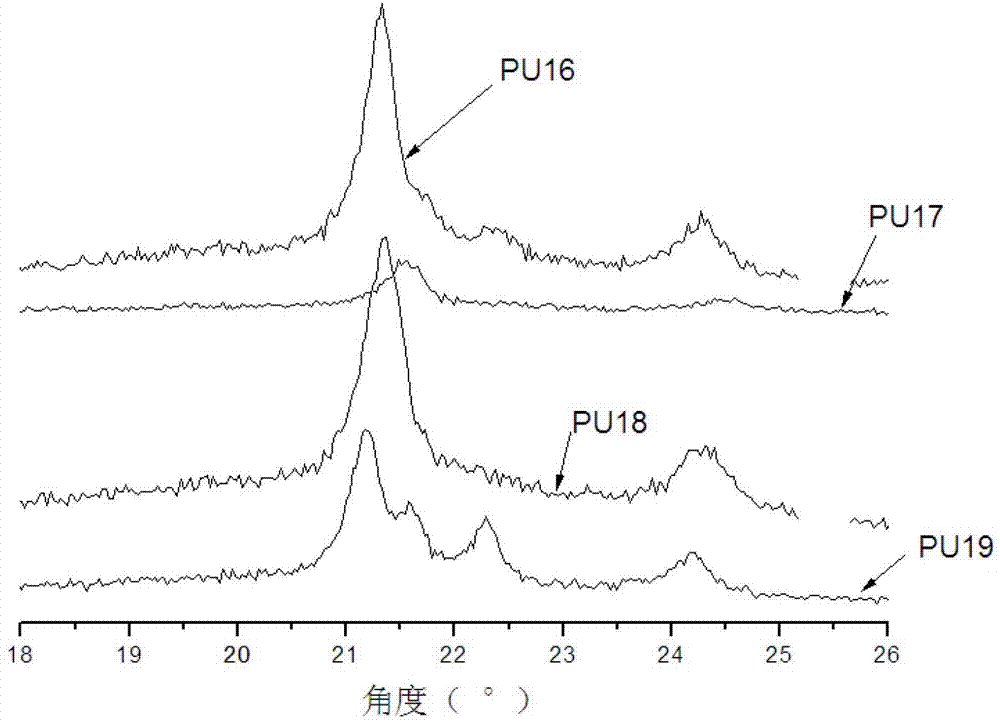
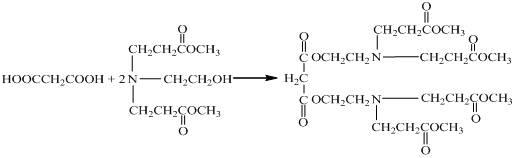
Hyperbranched polymer, hyperbranched type water reducing agent of polycarboxylic acid series and preparation method and application thereof
ActiveCN102002134AGood dispersionAvoid reunionAtom-transfer radical-polymerizationPolyethylene glycol
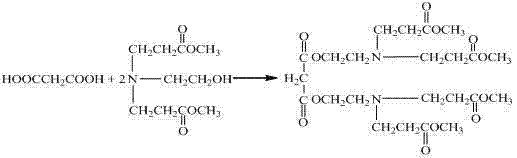
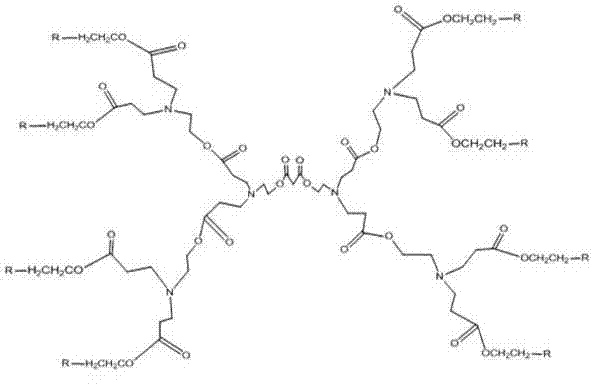
The invention provides a hyperbranched polymer and a preparation method thereof, wherein the preparation method comprises the steps of taking methyl acrylate, ethanolamine, 2-bromoethanol and propandioic acid as raw materials to synthesize a hyperbranched poly (amine-ester) nuclear molecule comprising a bromine terminal group; taking cuprous bromide and 2,2-bipyridyl as a catalyst and a coordinating agent; respectively grafting acrylic acid and single methyl oxygen radical sealed allyl polyethylene glycol into the hyperbranched poly (amine-ester) comprising bromine terminal group by means of an atom transfer radical polymerization method so as to obtain a hyperbranched polymer. The invention further provides a hyperbranched type water reducing agent of polycarboxylic acid series mainly comprising the hyperbranched polymer and the application thereof, wherein the water reducing agent improves the faults of existing water reducing agent of which the adding quantity is large and the cement adaptability is bad and the like,. and has the advantages that the adding quantity is low, the water reducing efficiency is high, the loss of slump is less, the compatibility to the cement is good, the frozen resistance is strong and the like, the properties of the product are stable, and the water reducing agent has no tendency of layering and precipitating and has no toxicity and pollution after storing for a long time.
Owner:江苏超力建材科技有限公司
Preparation method of light/pH-sensitive amphiphilic azobenzene polymer micelles
InactiveCN103193989ABreak the hydrophilic-hydrophobic balanceOrganic active ingredientsPharmaceutical non-active ingredientsPhenolPolypropylene
A preparation method of light / pH-sensitive amphiphilic azobenzene polymer micelles belongs to a field of functional polymer materials. The method comprises performing a radical polymerization for polymerizable monomer acrylic acid chloride to synthesize polypropylene chloride, using m-trifluoromethylaniline and phenol as raw materials to prepare m-trifluoromethylphenol containing azo groups, reacting the m-trifluoromethylphenol with 2-bromoethanol to synthesize 3-trifluoromethyl-4-ethanol oxygroup azobenzene, grafting the azo groups to the polypropylene chloride through an esterification reaction, hydrolyzing to obtain the amphiphilic polymer simultaneously containing the azo groups and carboxyl, and preparing the polymer into light / pH-sensitive polymer micelles through a micellization process. The polymer micelles are stable under a room temperature, and can be loaded with Nile red and other hydrophobic molecules; and the morphology of the micelles can be changed through ultraviolet irradiation and pH value adjusting, so that the loaded molecules can be released from the micelle. The polymer micelle not only has light responsiveness, but also has pH responsiveness, and has wide application prospect in fields of drug controlled release.
Owner:UNIV OF SCI & TECH BEIJING
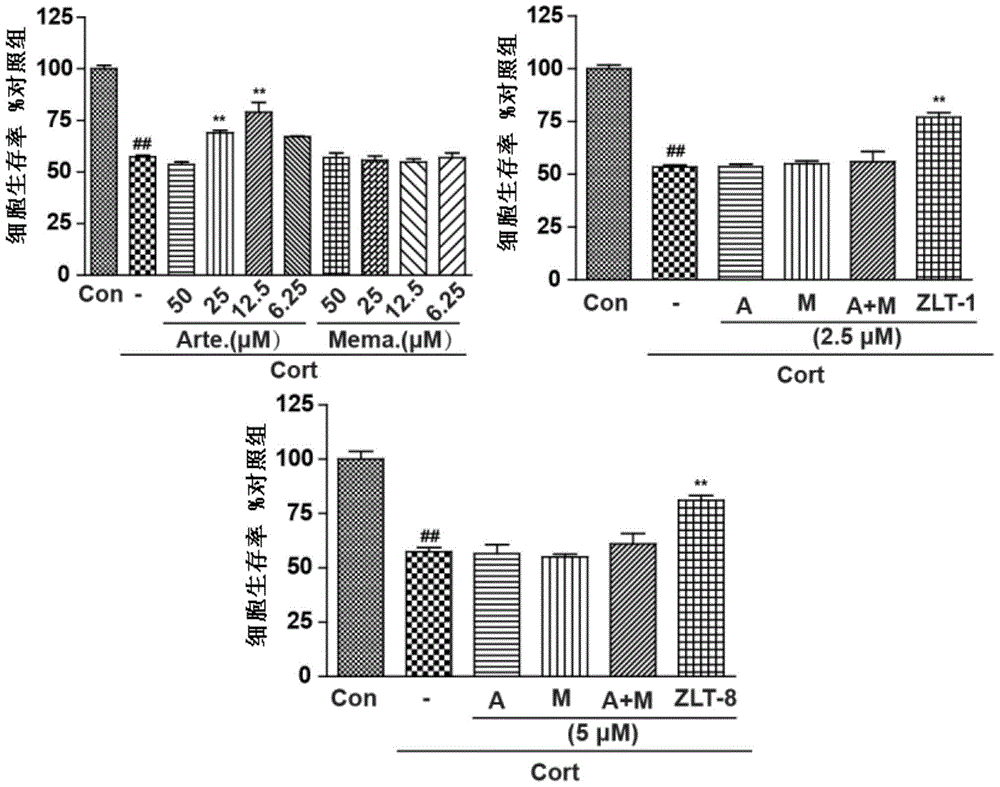
Dihydroarteannuin-memantine diad compounds, and synthesis method and application thereof
ActiveCN105732654ASimple preparation processSignificant effectOrganic active ingredientsNervous disorderMemantine HydrochlorideTreatment effect
The invention discloses dihydroarteannuin-memantine diad compounds, and a synthesis method and application thereof. The structure of the compounds is disclosed as Formula I. The synthesis method comprises the following steps: reducing arteannuin to obtain dihydroarteannuin, carrying out acetalation reaction on the dihydroarteannuin and 2-bromoethanol under the catalytic action of Lewis acid, and carrying out reaction on the acetalation reaction product and memantine hydrochloride to obtain the dihydroarteannuin-memantine diad compounds. The compounds are reported for the first time, have an therapeutic effect on neurodegenerative diseases, and can be used for preparing drugs for treating neurodegenerative diseases. Compared with other prior arts, the compounds disclosed by the invention have the advantages of simple preparation technique and better curative effect than memantine.
Owner:JINAN UNIVERSITY
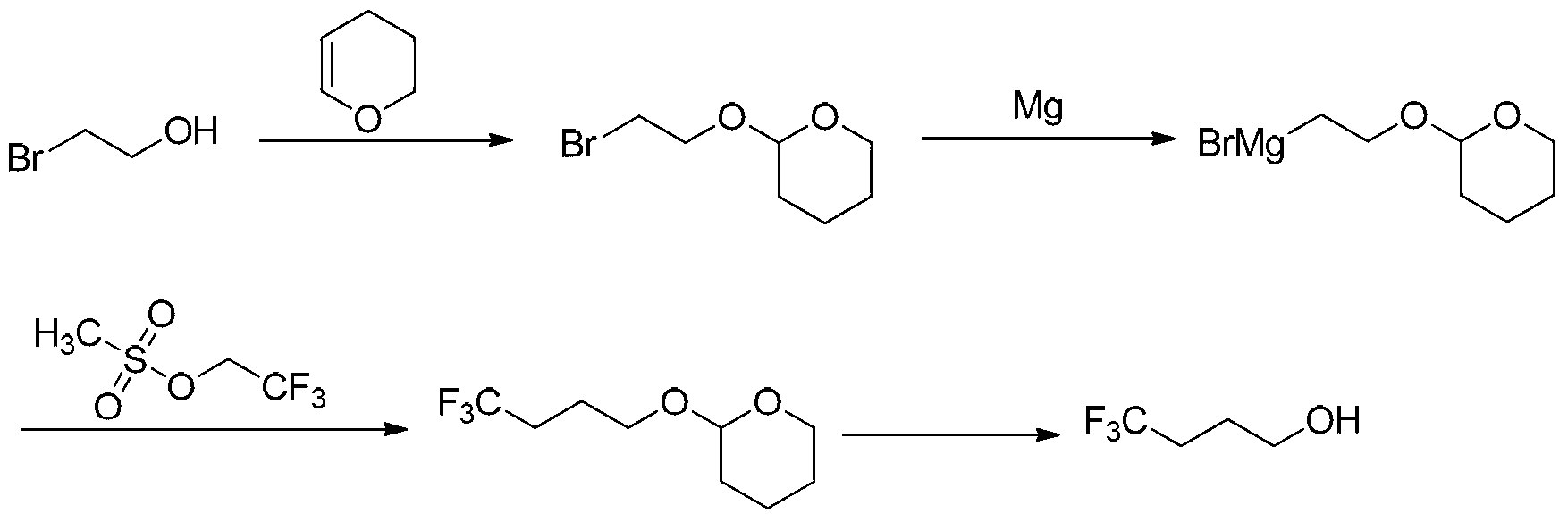
Method synthesizing 4, 4, 4-trifluoro butanol
ActiveCN103265403AShort process routeMild reaction conditionsPreparation by hydrolysisAlcoholGrignard reagent
The invention discloses a method for synthesizing 4, 4, 4-trifluoro butanol, wherein the method comprises: using 2-bromoethanol as a raw material, protecting a alcoholic hydroxyl group with 3,4-dihydro pyrans, preparing a Grignard reagent, carrying out coupling reaction of the Grignard reagent with 2,2,2-trifluoro ethyl methanesulfonates, and finally obtaining 4,4,4-trifluoro butanol by deprotection reaction. The method has advantages of short technology route, mild reaction condition, cheap and easily available raw materials, high reaction overall yield and low production cost, is in favor of industrialization production, and avoids usage of expensive raw materials like trifluoro butyric acid and lithium aluminium hydride.
Owner:XIAN CAIJING OPTO ELECTRICAL SCI & TECH
Method for synthesizing 3-tetrahydrofurfuryl alcohol
The invention relates to a method for synthesizing 3-tetrahydrofurfuryl alcohol. According to the technical scheme, 2-bromoethanol and diethyl malonate serve as starting materials, under the alkaline condition, 2-hydroxyethyl-diethyl malonate is generated through a reaction, hydrolysis is conducted on the 2-hydroxyethyl-diethyl malonate to generate 2-hydroxyethyl-malonate, catalyzing and high-pressure hydrotreatment are conducted on the 2-hydroxyethyl-malonate to generate 2-hydroxymethyl-1,4-butanediol, and dehydration cyclization is conducted on the 2-hydroxyethyl-1,4-butanediol to generate the 3-tetrahydrofurfuryl alcohol. According to the method for synthesizing the 3-tetrahydrofurfuryl alcohol, the yield can reach over 85 percent, the purity can reach above 95 percent, the reaction condition is mild, the cost is low, operation is easy, and the method has the advantage of being suitable for industrial production.
Owner:SHIJIAZHUANG XINGBAI BIOLOGICAL ENG
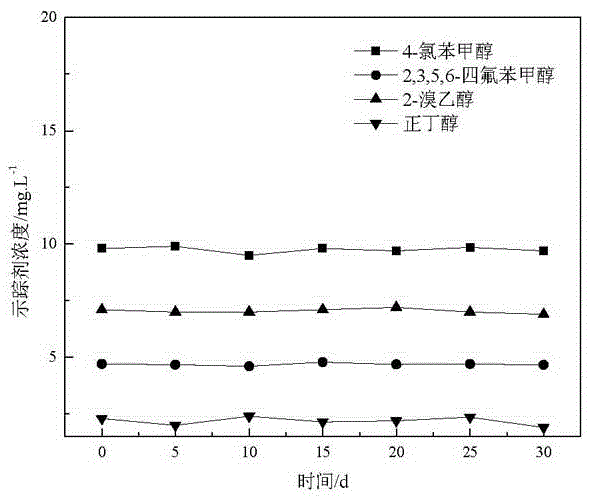
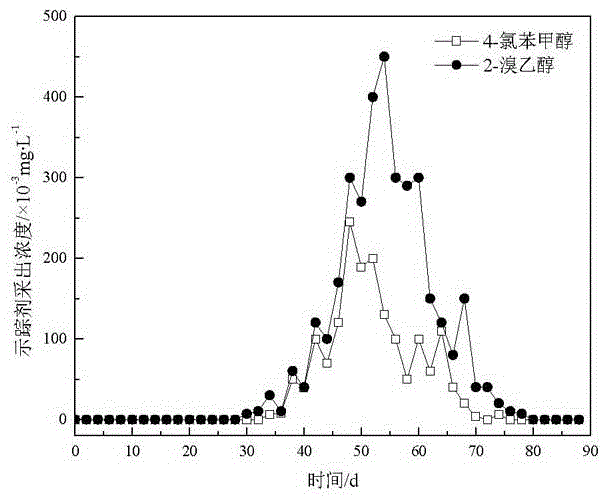
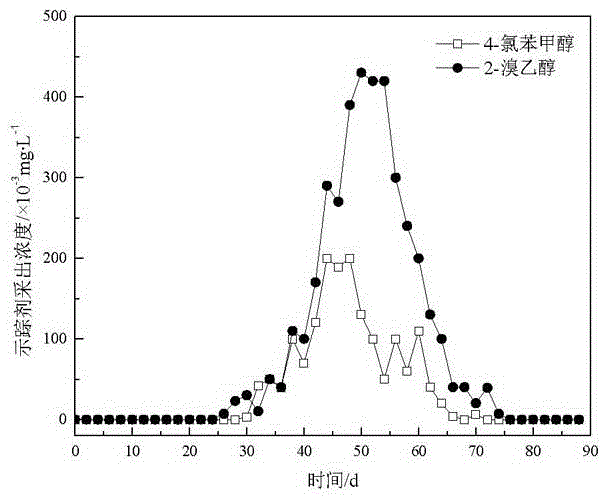
Application of halohydrin as water injection chemical tracer in oil field
InactiveCN106680147AHigh sensitivityLow detection limitComponent separationIndirect flow property measurementAlcoholBromine
The invention relates to the technical field of tracers and provides an application of halohydrin as a water injection chemical tracer in an oil field. The application of halohydrin as the water injection chemical tracer in the oil field is disclosed for the first time. 4-chlorobenzyl alcohol, 2,3,5,6-tetrafluorobenzyl alcohol and 2-bromoethanol have the characteristics of high sensitivity, low detection limit and good stability, 2-bromoethanol can cooperate with either of 4-chlorobenzyl alcohol and 2,3,5,6-tetrafluorobenzyl alcohol for use, not only can oil reservoir distribution and oil layer motion condition be monitored, but also downhole residual oil saturation can be determined; meanwhile, compared with traditional alcohol tracers, 4-chlorobenzyl alcohol, 2,3,5,6-tetrafluorobenzyl alcohol and 2-bromoethanol are resistant to bacterial and microbial erosion and are low in input and interference, greatly reduce the input and production cost and can be well applied as the water injection chemical tracer to tracing detection in the oil field.
Owner:XINJIANG UNIVERSITY
Compound N-2-methylsulfonyl ethyl group-2 beta-carbo methoxy group-3-beta-(4-chlorphenyl) nortropane, and synthetic method
InactiveCN1687062AReduce radiation exposureEasy to operateOrganic chemistrySynthesis methodsEthyl group
The compound N-2-methylsulfonylethyl-2 beta-carbomethoxyl-3 beta-(4-chlorophenyl) demethyltropane and its synthesis method relate to the field of synthesis technology of nuclear pharmaceuticals labelled precursor compound. The synthesis method of demethyltropane (MsOCC1T) uses 2 beta-carbomethoxy-3 beta-(4-chlorophenyl) demethyltropane as raw material, and said invention adopts literature synthesis method, then first step reaction uses 2 beta-carbomethoxyl-3-beta-(4-chlorophenyl) demethyltropane and 2-bromoethanol and makes them react to synthesize intermediate product N-(2-ethoxyl)-2 beta-carbomethoxyl-3 beta-(4-chlorophenyl) demethyltropane, and the second step reaction makes said intermediate product be reacted with methyl sulfoacid anhydride to synthesize target compound.
Owner:JIANGSU INST OF NUCLEAR MEDICINE
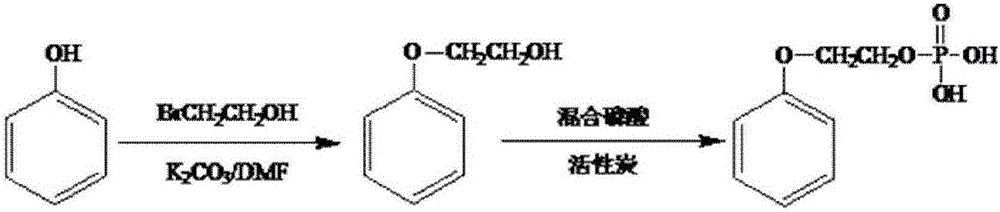
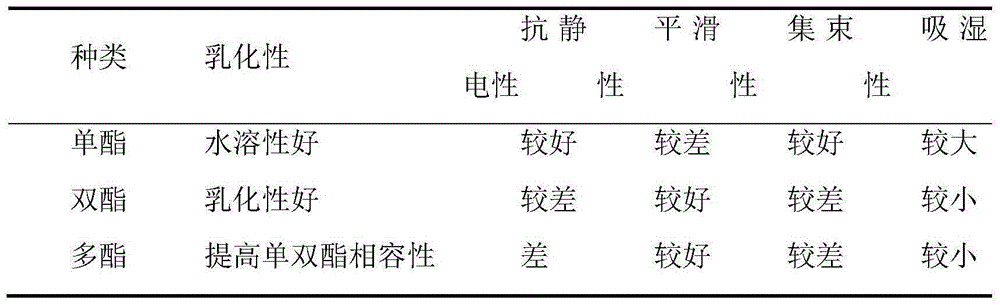
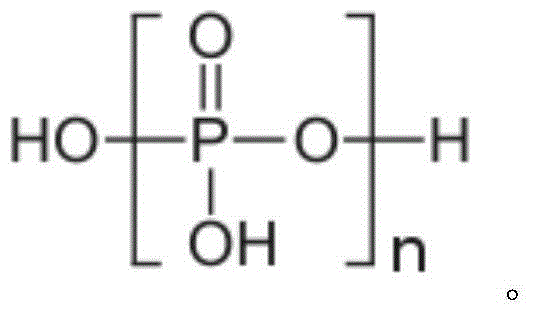
Preparation method of ethylene glycol monophenyl ether phosphomonoester surfactant
ActiveCN105080421AGood alkali resistanceImprove permeabilityTransportation and packagingMixingEthylene glycol monophenyl etherPolymer science
The invention discloses a preparation method of ethylene glycol monophenyl ether phosphomonoester surfactant and belongs to the technical field of surfactant. The preparation method disclosed by the invention comprises the steps of mixing phenol with 2-bromoethanol, dissolving the mixture in dimethylformamide, then adding and uniformly agitating potassium carbonate for reaction, adding distilled water to separate liquid, and increasing the temperature of solution at a lower layer to 160 to 180 DEG C to remove residual dimethylformamide in the solution; then adding polyphosphoric acid into the solution, agitating the solution and increasing the temperature to 60 to 80 DEG C, then adding 1.65 to 5g of catalyst for reaction, adding and uniformly agitating P2O5 for reaction, and adding distilled water with volume of 3 to 5 percent of total volume of the solution to perform hydrolysis for 1 to 3h at 60 to 80 DEG C to obtain the ethylene glycol monophenyl ether phosphomonoester surfactant. The preparation method of ethylene glycol monophenyl ether phosphomonoester surfactant has the advantages that the conversion rate of the prepared ethylene glycol monophenyl ether phosphomonoester is high, the monoester content highly reaches 86 to 86 percent, the alkali resistance of the surfactant is extremely high and the permeability is excellent.
Owner:马鞍山市恺鸾鑫实业有限责任公司
Preparation method and application of sulfate type photoresponsive foam control agent
ActiveCN110407723ATo achieve the purpose of controlSolve intractableSulfonic acids salts preparationDyeing processIce waterUltraviolet lights
The invention discloses a preparation method and an application of a sulfate type photoresponsive foam control agent. The preparation method comprises the following steps: (1) reacting p-nitrophenol,potassium hydroxide and water to obtain p-hydroxyazobenzene; and (2) dissolving the p-hydroxyazobenzene in DMF, adding an inorganic alkali and a catalyst, then dropwise adding DMF containing 1-bromoalkane to perform a reaction in order to obtain 4-hydroxy-4'-alkoxyazobenzene, dissolving the 4-hydroxy-4'-alkoxyazobenzene in DMF, adding the inorganic alkali and a catalyst, dropwise adding DMF containing 2-bromoethanol to perform a reaction in order to obtain 4-(2-hydroxyethoxy)-4'-alkoxyazobenzene, dropwise adding chlorosulfonic acid to the 4-(2-Hydroxyethoxy)-4'-alkoxyazobenzene in an ice waterbath, removing the ice water bath, carrying out a reaction at room temperature, and adjusting the pH vale to be more than 7 in order to obtain the sulfate type photoresponsive foam control agent. Thefoam control agent can realize the reversible conversion of a defoaming agent and a foam stabilizer under the irradiation of ultraviolet light and visible light in order to control foam and solve theproblem of residual foam.
Owner:JIANGNAN UNIV
Hyperbranched polymer, hyperbranched type water reducing agent of polycarboxylic acid series and preparation method and application thereof
ActiveCN102002134BGood dispersionAvoid reunionAtom-transfer radical-polymerizationPolyethylene glycol
The invention provides a hyperbranched polymer and a preparation method thereof, wherein the preparation method comprises the steps of taking methyl acrylate, ethanolamine, 2-bromoethanol and propandioic acid as raw materials to synthesize a hyperbranched poly (amine-ester) nuclear molecule comprising a bromine terminal group; taking cuprous bromide and 2,2-bipyridyl as a catalyst and a coordinating agent; respectively grafting acrylic acid and single methyl oxygen radical sealed allyl polyethylene glycol into the hyperbranched poly (amine-ester) comprising bromine terminal group by means of an atom transfer radical polymerization method so as to obtain a hyperbranched polymer. The invention further provides a hyperbranched type water reducing agent of polycarboxylic acid series mainly comprising the hyperbranched polymer and the application thereof, wherein the water reducing agent improves the faults of existing water reducing agent of which the adding quantity is large and the cement adaptability is bad and the like,. and has the advantages that the adding quantity is low, the water reducing efficiency is high, the loss of slump is less, the compatibility to the cement is good,the frozen resistance is strong and the like, the properties of the product are stable, and the water reducing agent has no tendency of layering and precipitating and has no toxicity and pollution after storing for a long time.
Owner:江苏超力建材科技有限公司
Solid heterogeneous catalyst used for preparation method using ethylene oxide, carbon dioxide and methanol to prepare dimethyl carbonate and ethylene glycol and preparation method of solid heterogeneous catalyst
InactiveCN107362826ALower conversion rateLow selectivityProductsOxygen-containing compound preparationMethyl carbonatePolystyrene
The invention discloses a solid heterogeneous catalyst used for a preparation method using ethylene oxide, carbon dioxide and methanol to prepare dimethyl carbonate and ethylene glycol. The solid heterogeneous catalyst comprises 1.0 part of carrier, 0.1-0.7 part of active component 1 and 0.5-1.5 parts of active component 2, wherein the carrier is modified resin material chlorinated polyethylene, chlorinated polypropylene or chloromethylated polystyrene; the active component 1 is imidazole; the active component 2 is a halogen-containing organic compound 2-bromoethanol or 1, 2-dibromoethane. A preparation method of the solid heterogeneous catalyst includes: preprocessing the carrier, and immobilizing the active component 1 and the active component 2 to obtain the solid heterogeneous catalyst. Compared with existing catalysts which are high in activity but low in product selectivity or high in product selectivity but low in activity, the solid heterogeneous catalyst is high in conversion rate and product selectivity.
Owner:SHAANXI COAL & CHEM TECH INST
Particle emulsifier based on colloid self-assembled by hydrophobic modified hyaluronic acid and preparation method of particle emulsifier
InactiveCN105199012AGood biocompatibilityGood moisturizing effectCosmetic preparationsToilet preparationsFreeze-dryingBiocompatibility Testing
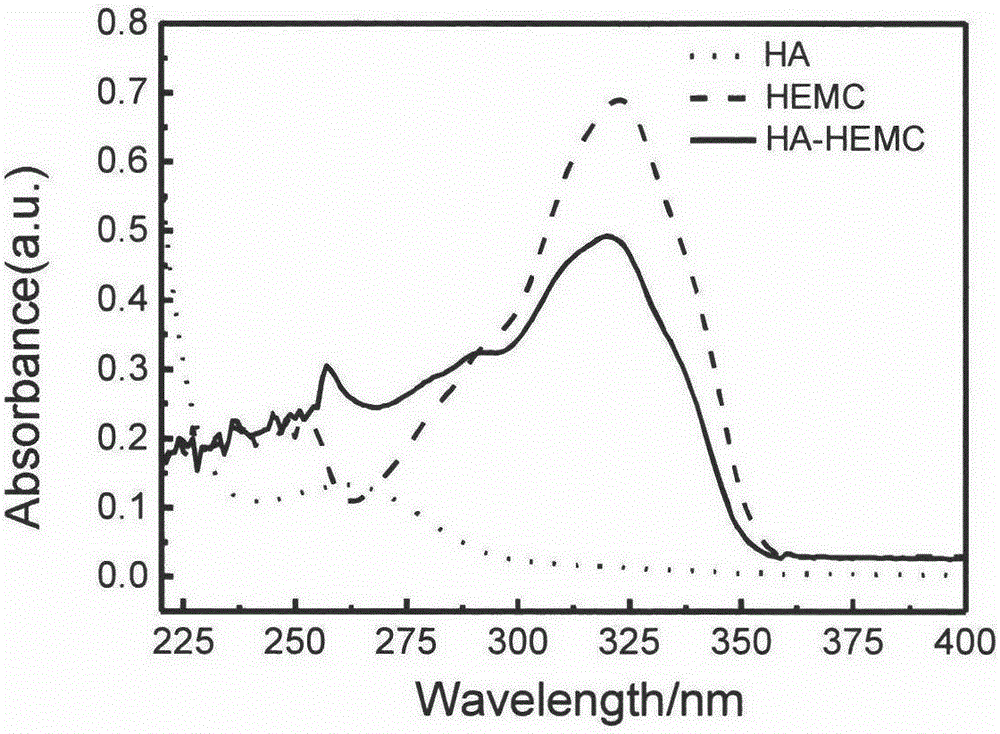
The invention discloses a particle emulsifier based on a colloid self-assembled by hydrophobic modified hyaluronic acid and a preparation method of the particle emulsifier, and belongs to the technical field of self-assembly of natural macromolecules and preparation of the particle emulsifier. The preparation method of the particle emulsifier comprises the following steps: step 1, HA (hyaluronic acid) and TBA-OH (tetrabutylammonium hydroxide) are compounded; step 2, 2-bromoethanol and 7-hydroxyl-4-methylcoumarin are adopted for a nucleophilic substitution reaction, and HEMC (7-(2-hydroxyethoxy)-4-methylcoumarin) is prepared; step 3, a compound obtained in the step 1 and HEMC have an esterification reaction under the action of a catalyst, and HA-HEMC (hydrophobic modified HA) is obtained; step 4, HA-HEMC is self-assembled in a selective solvent, a colloid dispersing agent is formed and freeze-dried, and the particle emulsifier is obtained. The particle emulsifier based on the colloid self-assembled by hydrophobic modified hyaluronic acid has good biocompatibility, can effectively stabilize an oil-water interface and can be applied to the fields of cosmetics, food, bio-medicine and the like.
Owner:JIANGNAN UNIV
Preparation method of formoterol key intermediate
The invention provides a preparation method of a formoterol key intermediate (R)-N-(2-(benzyloxy)-5-(2-bromo-1-hydroxyethyl)phenyl)formamide represented by a formula I, wherein asymmetric reduction isperformed by using 1-(4-(benzyloxy)-3-nitrophenyl)-2-bromoethanone (II) as a raw material and using (3aS-cis)-(-)-3,3a,8,8a-tetrahydro-2H-indeno[1,2-d]oxazole-2-isopropylborane (IV) as a catalyst toobtain a chiral alcohol intermediate (R)-1-(4-(benzyloxy)-3-nitrophenyl)-2-bromoethanol (III) with high enantioselectivity, and then nitro reduction and formylation one-pot reaction are performed to obtain a target product represented by the formula I. Compared with the traditional resolution method, the method of the invention has advantages of high chiral purity of the product, short productionperiod, easy operation, mild condition, convenient post-treatment and high yield, and is suitable for large-scale industrial production.
Owner:上海天慈中商药业有限公司
Process for producing fluorescent reagent
InactiveCN100386624CIncreased molar absorbanceHigh detection sensitivityComponent separationColor/spectral properties measurementsWater bathsMethylene Dichloride
The present invention relates to a preparation method of fluorescent reagent. Said method includes the following steps: using ortho-benzoxyaniline as mother body for synthesis, under the action of concentrated sulfuric acid reflux heating for 2-4hr in boiling water bath to form acridone; making acridone be reacted with 2-bromoethanol in dimethyl sulphoxide solvent to obtain 2-(N-acridone group)-ethyl alcohol; stirring 2-(C-acridone group)-ethyl alcohol and tricyanogen chloride in methylene dichloride solution at 0-5 deg.C and making them be reacted for 16-24 hr so as to obtain the target product. Said invention also provides the characteristics of said fluorescent reagent and its application range.
Owner:DALIAN INST OF CHEM PHYSICS CHINESE ACAD OF SCI
Method synthesizing 4, 4, 4-trifluoro butanol
ActiveCN103265403BShort process routeMild reaction conditionsPreparation by hydrolysisGrignard reagentPyran
The invention discloses a method for synthesizing 4, 4, 4-trifluoro butanol, wherein the method comprises: using 2-bromoethanol as a raw material, protecting a alcoholic hydroxyl group with 3,4-dihydro pyrans, preparing a Grignard reagent, carrying out coupling reaction of the Grignard reagent with 2,2,2-trifluoro ethyl methanesulfonates, and finally obtaining 4,4,4-trifluoro butanol by deprotection reaction. The method has advantages of short technology route, mild reaction condition, cheap and easily available raw materials, high reaction overall yield and low production cost, is in favor of industrialization production, and avoids usage of expensive raw materials like trifluoro butyric acid and lithium aluminium hydride.
Owner:XIAN CAIJING OPTO ELECTRICAL SCI & TECH
Methods of reducing pests by use of halogen substituted ethanol
InactiveUS7056864B2Reduces effective amountBiocideHydroxy compound active ingredientsHalogenNematode
A method for reducing pests in an object or area by applying to the object or area a pest reducing effective amount of halogen substituted ethanol (e.g., 2-iodoethanol, 2-bromoethanol, or mixtures thereof). The pests may be, for example, fungi, insects, nematodes, bacteria, weeds, or mixtures thereof. The object or area may be, for example, soil, structures, agricultural commodities, plants, or mixtures thereof.
Owner:US SEC AGRI +1
Preparation method for ticagrelor intermediate
InactiveCN106496180ARaw materials are cheap and easy to getReduce manufacturing costOrganic chemistryTicagrelorCycloaddition
The invention discloses a preparation method for a ticagrelor intermediate. The preparation method comprises the following steps: 1) subjecting o-substituted benzaldoxime and cyclopentadiene to a contact reaction so as to obtain a compound as shown in a formula I; 2) oxidizing the compound as shown in the formula I with hydrogen peroxide under the catalysis of osmium tetroxide so as to obtain a compound as shown in a formula II; 3) reacting the compound as shown in the formula II with acetone under the catalysis of p-toluenesulfonic acid so as to obtain a compound as shown in a formula III; 4) reacting the compound as shown in the formula III with 2-bromoethanol in the presence of alkali so as to obtain a compound as shown in a formula IV; and 5) reducing the compound as shown in the formula IV so as to obtain the ticagrelor intermediate. The method provided by the invention uses cheap and easily available raw material, reduces production cost, and does not need any chiral raw material; a cycloaddition reaction enables a highly stereoselective product to be obtained; and the method is short in steps and high in reaction yield and provides a novel approach for synthesis of ticagrelor.
Owner:QINGDAO YUNTIAN BIOTECH
Synthesis method of racemic-(3aR,7aR)-5-(t-butyloxycarboryl)-octahydro furan [3,2-c] pyridine-3a- carboxylic acid
InactiveCN107383034AReasonable reaction process designMethod route shortOrganic chemistry methodsFuranLithium hydroxide
The invention relates to a synthesis method of racemic-(3aR,7aR)-5-(t-butyloxycarboryl)-octahydro furan [3,2-c] pyridine-3a-carboxylic acid. The method mainly solves the technical problem that no proper industrial synthesis method exists in the prior art. The method comprises seven steps that: firstly, 2-bromoethanol and ethyl propiolate react in a solvent of methylene dichloride at the room temperature to obtain a compound 2; then, the compound 2 and ethyl cyanoacetate react in a solvent of N, N-dimethyl formamide at the room temperature over the night to obtain a compound 3; the compound 3 is subjected to catalytic hydrogenation to reduce the cyano groups to obtain amino groups; meanwhile, the exchange with the self ester is performed to obtain a compound 4; a compound 5 is obtained through carbobenzoxy chloride on the compound 4; the compound 5 is reduced by borane to obtain a compound 6; finally, the compound 6 uses a palladium hydroxide as a catalyst in ethanol; meanwhile, Boc estolide is added; hydrogen gas is introduced for overnight reaction to obtain a target compound 7; the compound 7 is hydrolyzed by water and lithium hydroxide in a mixed solution of methanol and water; a final target compound 8 is obtained.
Owner:上海药明康德新药开发有限公司 +4
Synthesis method of 7-chloroquinaldine
ActiveCN108822033AEasy to purifyLow priceOrganic chemistryOrganic-compounds/hydrides/coordination-complexes catalystsCrotonaldehydeSynthesis methods
The invention discloses a synthesis method of 7-chloroquinaldine. 2-nitrotoluene, SiO2-HEPIMBr, m-chloroaniline, crotonaldehyde, TEOS, 2-bromoethanol, imidazole and 3-chloropropyltriethoxysilane are taken as main raw materials. According to the synthesis process, m-chloroaniline and crotonaldehyde are subjected to Skraup reaction under action of an immobilized ion catalyst SiO2-HEPIMBr, and 7-chloroquinaldine is obtained. SiO2 immobilized hydroxyl ionic liquid HEPIMBr is prepared with a grafting method, in the reaction, few by-products are produced and no 5-isomer is produced, so that the separation and purification process is reduced, purity of a product is improved, yield of the product is increased, and higher market competitiveness is achieved.
Owner:马海红
Preparation method of phenoxybenzamine hydrochloride
ActiveCN102887830AAvoid residueRaw materials are easy to getOrganic compound preparationAmino-hyroxy compound preparationBrominePhenoxybenzamine Hydrochloride
The invention relates to a preparation method of phenoxybenzamine hydrochloride, which comprises the following steps: reacting phenol with methyl epoxyethane to obtain 1-phenoxy-2-hydroxy propane; reacting 2-nitrobenzenesulfonyl chloride with benzylamine to obtain N-2-nitrobenzenesulfonyl benzylamine; reacting the obtained 1-phenoxy-2-hydroxy propane with the N-2-nitrobenzenesulfonyl benzylamine to obtain N-benzyl-N-(1-methyl-2-phenoxyethyl)-2-nitro-benzsulfamide; converting the N-benzyl-N-(1-methyl-2-phenoxyethyl)-2-nitro-benzsulfamide into N-(1-methyl-2-phenoxyethyl) benzylamine; reacting the obtained N-(1-methyl-2-phenoxyethyl) benzylamine with 2-bromoethanol to obtain benzyl-(1-methyl-2-phenoxyethyl) ethanol; and dissolving benzyl-(1-methyl-2-phenoxyethyl) ethanol in a solvent, introducing hydrogen chloride gas, dropwisely adding thionyl chloride, and performing reflux reaction to obtain phenoxybenzamine hydrochloride.
Owner:SUZHOU HOMESUN PHARMA
Elastic conductive microsphere for anisotropic conductive adhesive film, and preparation method thereof
ActiveCN109705770AImprove conductivityAvoid breakingLiquid/solution decomposition chemical coatingElectrically-conducting adhesivesAnisotropic conductive adhesiveMicrosphere
The invention relates to the technical field of conductive adhesive film fillers, and provides an elastic conductive microsphere for an anisotropic conductive adhesive film, and a preparation method thereof. The method comprises the following steps: preparing 1-hydroxyethyl-3-vinylimidazolium bromide from 1-vinylimidazole and 2-bromoethanol, carrying out block copolymerization on the 1-hydroxyethyl-3-vinylimidazolium bromide and butadiene to obtain poly(ionic liquid)-butadiene elastic microspheres, and roughening, sensitizing, activating and chemically silvering the poly(ionic liquid)-butadiene elastic microspheres to obtain the elastic conductive microsphere for the anisotropic conductive adhesive film. Compared with conventional methods, the method for preparing the block copolymerized elastic microspheres from 1-hydroxyethyl-3-vinylimidazolium bromide and butadiene has the advantages of significant improvement of the elasticity of the microsphere by introducing a soft chain segment,reduction of breaking of the microsphere during crimping interconnection, broadening of the operable crimping pressure range, and reduction of the operation difficulty.
Owner:苏州纽劢特新材料科技有限公司
Dihydroartemisinin-memantine double compound and its synthesis method and application
ActiveCN105732654BSimple preparation processSignificant effectOrganic active ingredientsNervous disorderMemantine HydrochlorideSynthesis methods
The invention discloses dihydroarteannuin-memantine diad compounds, and a synthesis method and application thereof. The structure of the compounds is disclosed as Formula I. The synthesis method comprises the following steps: reducing arteannuin to obtain dihydroarteannuin, carrying out acetalation reaction on the dihydroarteannuin and 2-bromoethanol under the catalytic action of Lewis acid, and carrying out reaction on the acetalation reaction product and memantine hydrochloride to obtain the dihydroarteannuin-memantine diad compounds. The compounds are reported for the first time, have an therapeutic effect on neurodegenerative diseases, and can be used for preparing drugs for treating neurodegenerative diseases. Compared with other prior arts, the compounds disclosed by the invention have the advantages of simple preparation technique and better curative effect than memantine.
Owner:JINAN UNIVERSITY
A kind of preparation method of ethylene glycol phenyl ether phosphoric acid monoester surfactant
ActiveCN105080421BGood alkali resistanceImprove permeabilityTransportation and packagingMixingActive agentPhosphate
The invention discloses a preparation method of ethylene glycol phenyl ether phosphoric acid monoester surfactant, which belongs to the technical field of surfactants. The preparation method of the present invention is: mix and dissolve phenol and 2-bromoethanol in dimethylformamide, then add potassium carbonate and stir evenly, add distilled water after the reaction, and heat the lower layer solution to 160-180°C to evaporate the solution Residual dimethylformamide; add polyphosphoric acid to the solution, heat up to 60-80°C under stirring, then add 1.65-5g of catalyst, add P2O5 after the reaction and stir evenly, and then add 3% to 3% of the total volume of the solution after the reaction 15% distilled water is hydrolyzed at 60-80°C for 1-3 hours to obtain ethylene glycol phenyl ether phosphate monoester surfactant. The conversion rate of the ethylene glycol phenyl ether phosphoric acid monoester prepared by the invention is high, and the monoester content thereof is as high as 86% to 96%. The surfactant has extremely strong alkali resistance and has excellent permeability at the same time.
Owner:马鞍山市恺鸾鑫实业有限责任公司
New Etofibrate preparation method
The invention relates to a new preparation method of a blood lipid control medicine named 3-picolinic acid 2-(2-(4-chlorophenoxy acid)-2-methyl -1-propionyloxy) ethyl ester (Etofibrate). In the preparation method, the traditional 2-chlorohydrin is substituted with 2-bromethol, and nicotinic acid is adopted in the second reaction; and a solvent with high boiling point is avoided, therefore, the reaction is safe, convenient and economical, and is more suitable for industrial production.
Owner:大道隆达(北京)医药科技发展有限公司
Method of fabricating [F-18]FEONM precursor
ActiveUS10059661B1Increase fat solubilityIncrease carbonOrganic compound preparationSulfonic acid esters preparationBromineOxygen
A [F-18]FEONM precursor is synthesized. 2-bromoethanol is added to further connect an atom of oxygen at an N terminal of the precursor. Four atoms of carbon can be further connected. Thus, better fat-solubility is obtained along with the increase in carbon. Positioning in brain imaging becomes better.
Owner:INST NUCLEAR ENERGY RES ROCAEC
Synthetic method and application of hapten of tetrabromobisphenol A derivative (TBBPA-MHEE)
InactiveCN106588564ASimple methodMethod is feasibleOxygen-containing compound preparationOrganic compound preparationTetrabromobisphenol AEthyl acetate
The invention provides a synthetic method and application of a hapten of a tetrabromobisphenol A derivative (TBBPA-MHEE). The synthetic method comprises the following steps: (1) with TBBPA as a raw material, subjecting TBBPA, methyl bromoacetate and 2-bromoethanol to a substitution reaction so as to produce methyl-2-(2,6-dibromo-4-(2-(3,5-dibromo-4-(2-hydroxyethyoxy)phenyl)prop-2-yl)phenoxy)ethyl acetate; and (2) subjecting a reaction product of the previous step to saponification with sodium hydroxide and acidification with hydrochloric acid so as to obtain the hapten 2-(2,6-dibromo-4-(2-(3,5-dibromo-4-(2-hydroxyethyoxy)phenyl)prop-2-yl)phenoxy)acetic acid. According to the invention, the hapten of the tetrabromobisphenol A derivative (TBBPA-MHEE) is successfully designed and synthesized through repeated experiments; and the synthetic method provided by the invention is a novel synthetic method for the hapten and is simple, safe and practicable.
Owner:JIANGSU UNIV
A kind of preparation method of light/pH sensitive type amphiphilic azobenzene polymer micelle
InactiveCN103193989BBreak the hydrophilic-hydrophobic balanceOrganic active ingredientsPharmaceutical non-active ingredientsPolypropylenePhenol
A preparation method of light / pH-sensitive amphiphilic azobenzene polymer micelles belongs to a field of functional polymer materials. The method comprises performing a radical polymerization for polymerizable monomer acrylic acid chloride to synthesize polypropylene chloride, using m-trifluoromethylaniline and phenol as raw materials to prepare m-trifluoromethylphenol containing azo groups, reacting the m-trifluoromethylphenol with 2-bromoethanol to synthesize 3-trifluoromethyl-4-ethanol oxygroup azobenzene, grafting the azo groups to the polypropylene chloride through an esterification reaction, hydrolyzing to obtain the amphiphilic polymer simultaneously containing the azo groups and carboxyl, and preparing the polymer into light / pH-sensitive polymer micelles through a micellization process. The polymer micelles are stable under a room temperature, and can be loaded with Nile red and other hydrophobic molecules; and the morphology of the micelles can be changed through ultraviolet irradiation and pH value adjusting, so that the loaded molecules can be released from the micelle. The polymer micelle not only has light responsiveness, but also has pH responsiveness, and has wide application prospect in fields of drug controlled release.
Owner:UNIV OF SCI & TECH BEIJING
A kind of preparation method containing imidazolium cationic antistatic polyurethane
The invention discloses a preparation method of imidazole-containing cationic antistatic polyurethane. The preparation method provided by the invention is characterized by comprising the following steps: carrying out a reaction between imidazole and an acrylic acid ester compound at the mole ratio of 1:1-5 to obtain an esterified imidazole derivative; carrying out a reaction between the imidazole ester derivative and 2-bromoethanol at the mole ratio of 1:1-5 to obtain a hydroxyl-terminated imidazole ionic liquid, performing ester interchange between the hydroxyl-terminated imidazole ionic liquid and polyester or polyether glycol so as to obtain dihydroxy-terminiated imidazole cationic polyester or polyether glycol; and finally carrying out a reaction between 1-10 mol of dihydric alcohol with the molecular weight being 90-2000, 1-10 mol of dihydroxy-terminiated imidazole cationic antistatic long-chain dihydric alcohol and 1-20 mol of diisocyanate in a solvent in the presence of a catalyst so as to obtain the cationic antistatic polyurethane with the main chain containing imidazole. The imidazole-containing cationic polyurethane is prepared in the invention. The polyurethane has excellent antistatic performance. Electric property of the polyurethane will not be damaged by alkalinity or acidity, and electroconductibility of the polyurethane is not dependent on environment humidity. Thus, the polyurethane has a wider application field.
Owner:SICHUAN UNIV
Preparation method of-2-(N-carbazolyl)-ethoxy carbohydrazide
InactiveCN100412060CHigh fluorescence intensityHigh reactivityOrganic chemistryComponent separationHydrazine compoundPotassium hydroxide
The preparation process of 2-(N-carbazolyl)-ethoxy carbohydrazide includes the following steps: the reaction of carbazole and 2-bromo ethanol inside dimethyl sulfoxide solvent with potassium hydroxide as catalyst to produce 2-(N-carbazolyl)-ethanol; the reaction of 2-(N-carbazolyl)-ethanol and phosgene inside dichloro methane solvent under stirring for 24 hr to obtain 2-(N-carbazolyl)-ethyl chlorocarbonate; re-crystallization to purify 2-(N-carbazolyl)-ethyl chlorocarbonate; and reaction of purified 2-(N-carbazolyl)-ethyl chlorocarbonate and hydrazine hydrate to obtain the object product 2-(N-carbazolyl)-ethoxy carbohydrazide. The object product 2-(N-carbazolyl)-ethoxy carbohydrazide may be further re-crystallized to purify before being used for derivation reaction experiment.
Owner:DALIAN INST OF CHEM PHYSICS CHINESE ACAD OF SCI
Preparation method of phenoxybenzamine hydrochloride
ActiveCN102887830BAvoid residueRaw materials are easy to getOrganic compound preparationAmino-hyroxy compound preparationBrominePhenoxybenzamine Hydrochloride
The invention relates to a preparation method of phenoxybenzamine hydrochloride, which comprises the following steps: reacting phenol with methyl epoxyethane to obtain 1-phenoxy-2-hydroxy propane; reacting 2-nitrobenzenesulfonyl chloride with benzylamine to obtain N-2-nitrobenzenesulfonyl benzylamine; reacting the obtained 1-phenoxy-2-hydroxy propane with the N-2-nitrobenzenesulfonyl benzylamine to obtain N-benzyl-N-(1-methyl-2-phenoxyethyl)-2-nitro-benzsulfamide; converting the N-benzyl-N-(1-methyl-2-phenoxyethyl)-2-nitro-benzsulfamide into N-(1-methyl-2-phenoxyethyl) benzylamine; reacting the obtained N-(1-methyl-2-phenoxyethyl) benzylamine with 2-bromoethanol to obtain benzyl-(1-methyl-2-phenoxyethyl) ethanol; and dissolving benzyl-(1-methyl-2-phenoxyethyl) ethanol in a solvent, introducing hydrogen chloride gas, dropwisely adding thionyl chloride, and performing reflux reaction to obtain phenoxybenzamine hydrochloride.
Owner:SUZHOU HOMESUN PHARMA
























![Synthesis method of racemic-(3aR,7aR)-5-(t-butyloxycarboryl)-octahydro furan [3,2-c] pyridine-3a- carboxylic acid Synthesis method of racemic-(3aR,7aR)-5-(t-butyloxycarboryl)-octahydro furan [3,2-c] pyridine-3a- carboxylic acid](https://images-eureka.patsnap.com/patent_img/985c4940-3413-4eb4-ac6c-89b233e25011/972576DEST_PATH_IMAGE002.png)
![Synthesis method of racemic-(3aR,7aR)-5-(t-butyloxycarboryl)-octahydro furan [3,2-c] pyridine-3a- carboxylic acid Synthesis method of racemic-(3aR,7aR)-5-(t-butyloxycarboryl)-octahydro furan [3,2-c] pyridine-3a- carboxylic acid](https://images-eureka.patsnap.com/patent_img/985c4940-3413-4eb4-ac6c-89b233e25011/DEST_PATH_IMAGE001.png)














![Method of fabricating [F-18]FEONM precursor Method of fabricating [F-18]FEONM precursor](https://images-eureka.patsnap.com/patent_img/5f60d7fa-32fb-41ea-8b1c-d4af8ba5eef4/US10059661-D00001.png)
![Method of fabricating [F-18]FEONM precursor Method of fabricating [F-18]FEONM precursor](https://images-eureka.patsnap.com/patent_img/5f60d7fa-32fb-41ea-8b1c-d4af8ba5eef4/US10059661-C00001.png)
![Method of fabricating [F-18]FEONM precursor Method of fabricating [F-18]FEONM precursor](https://images-eureka.patsnap.com/patent_img/5f60d7fa-32fb-41ea-8b1c-d4af8ba5eef4/US10059661-C00002.png)