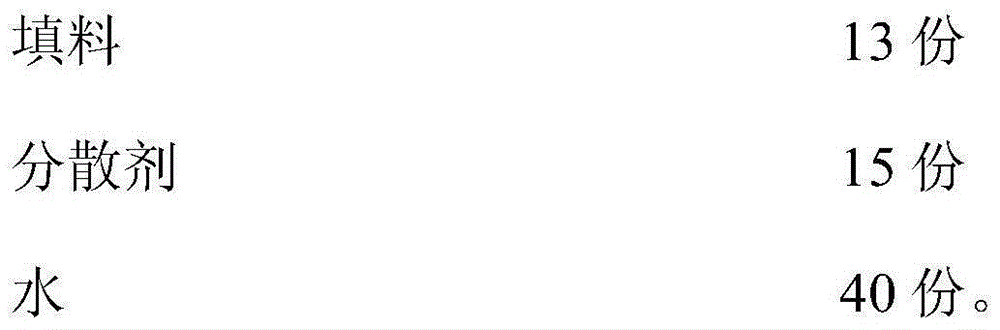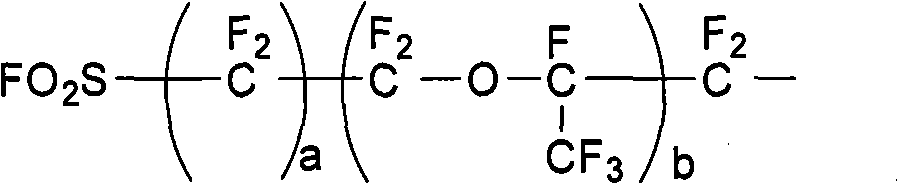Patents
Literature
191 results about "Ester sulfate" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
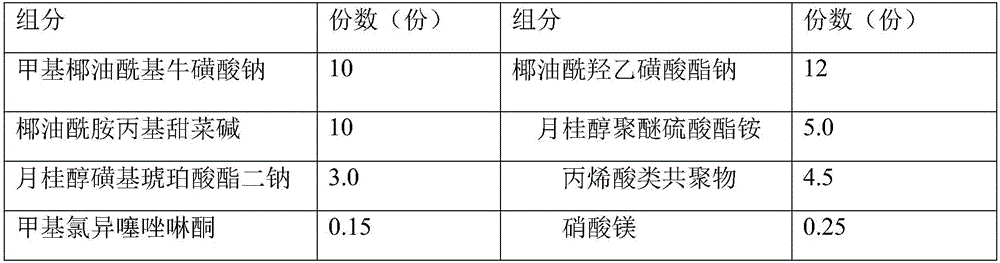
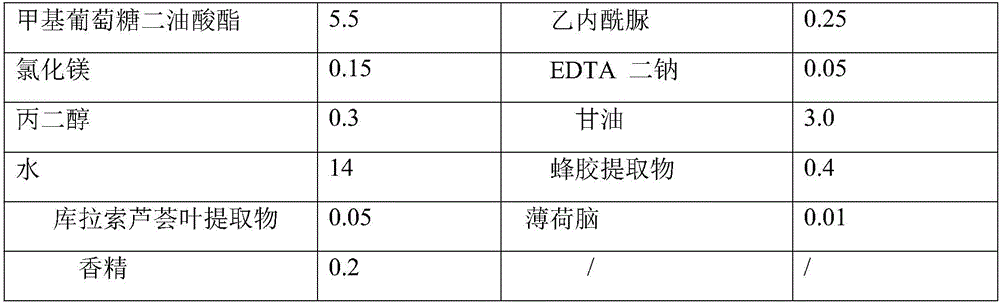
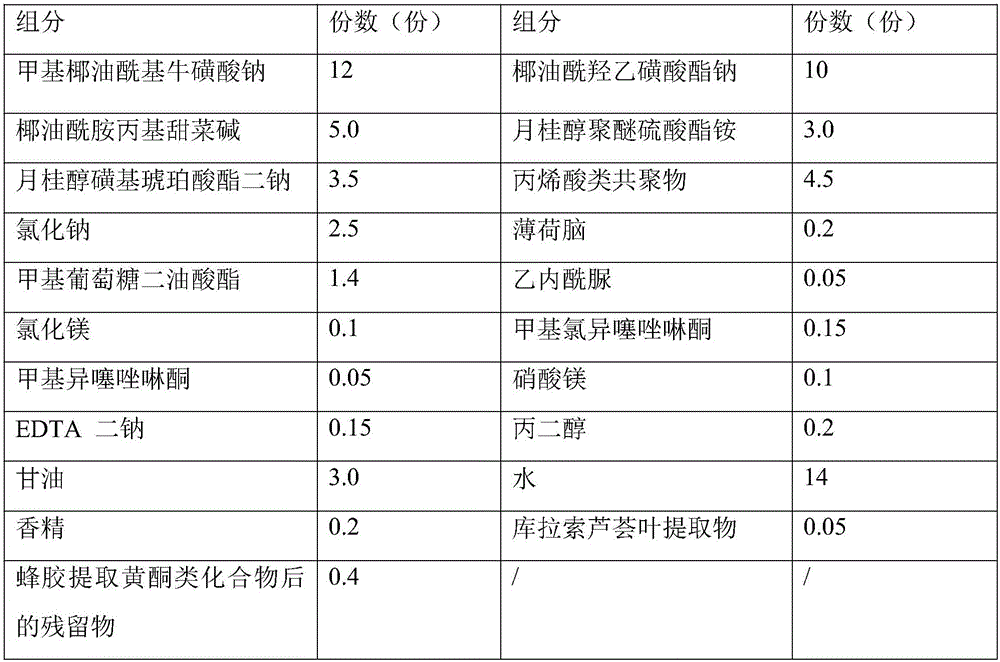
Facial cleanser and preparation method thereof
ActiveCN106038330APromote absorptionCosmetic preparationsToilet preparationsSODIUM METHYL COCOYL TAURATECocamidopropyl betaine
The invention discloses a facial cleanser and a preparation method thereof. The problem of skin tension caused by using the present facial cleanser for cleaning skin is solved. According to the technical scheme, the facial cleanser comprises the following components in parts by weight: 8-22 parts of sodium methyl cocoyl taurate, 3-12 parts of sodium cocoyl isethionate, 2-5 parts of lauryl sulfo succinate disodium, 2-10 parts of cocamidopropyl betaine, 1-5 parts of lauryl alcohol polyether ammonium sulfate, 3-10 parts of a thickening agent, 0.1-0.5 part of a preservative, 0.05-0.2 part of EDTA disodium, 0.2-0.6 part of propylene glycol, 2-12 parts of glycerinum, 0.1-0.5 part of essence and 10-18 parts of water. The prepared facial cleanser belongs to an amino acid facial cleanser, has the characteristics of safety, no stimulation, skin-relieving effect and deep skin-cleaning effect, and can be used by a user for a long term.
Owner:浙江高妍科技有限公司
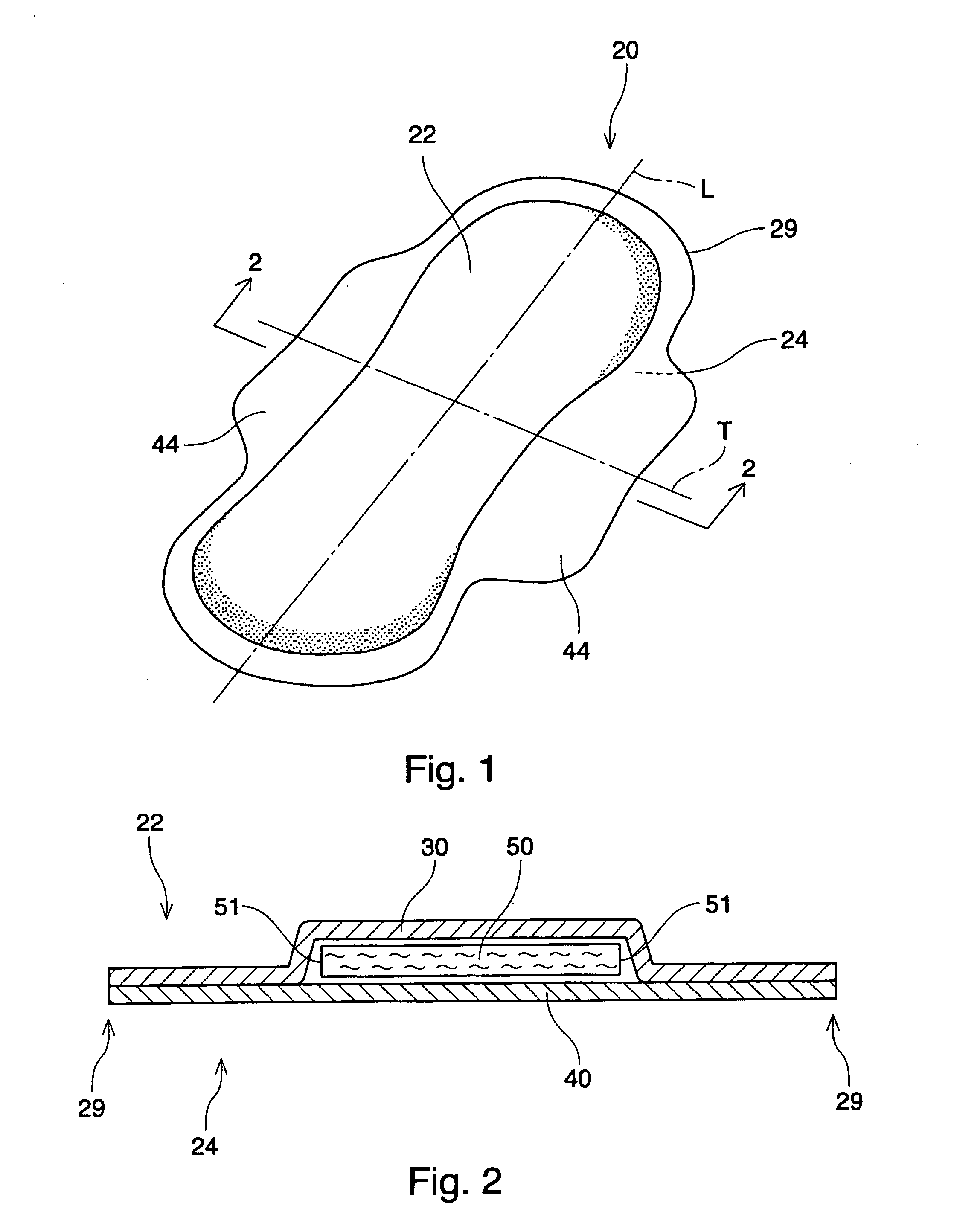
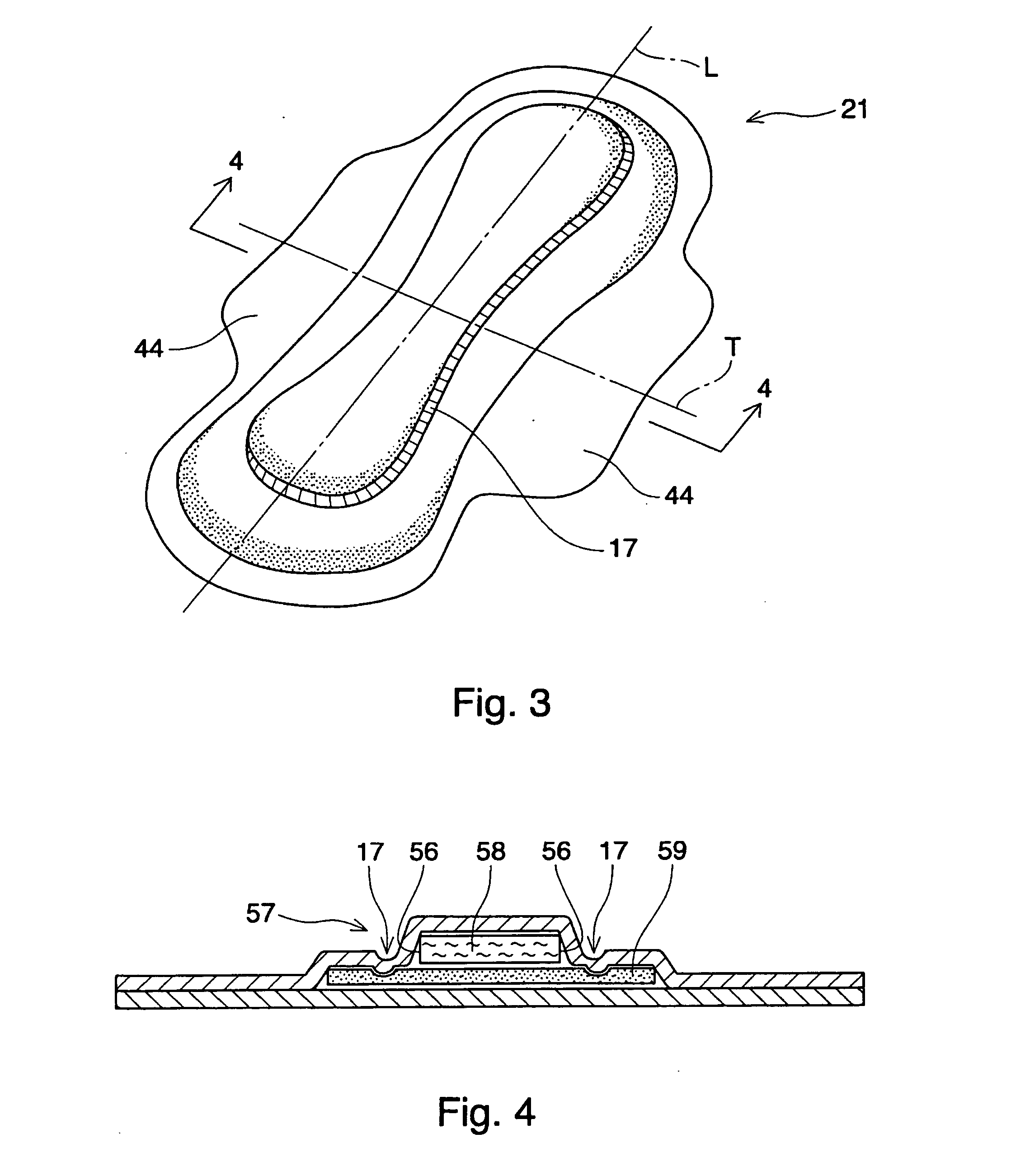
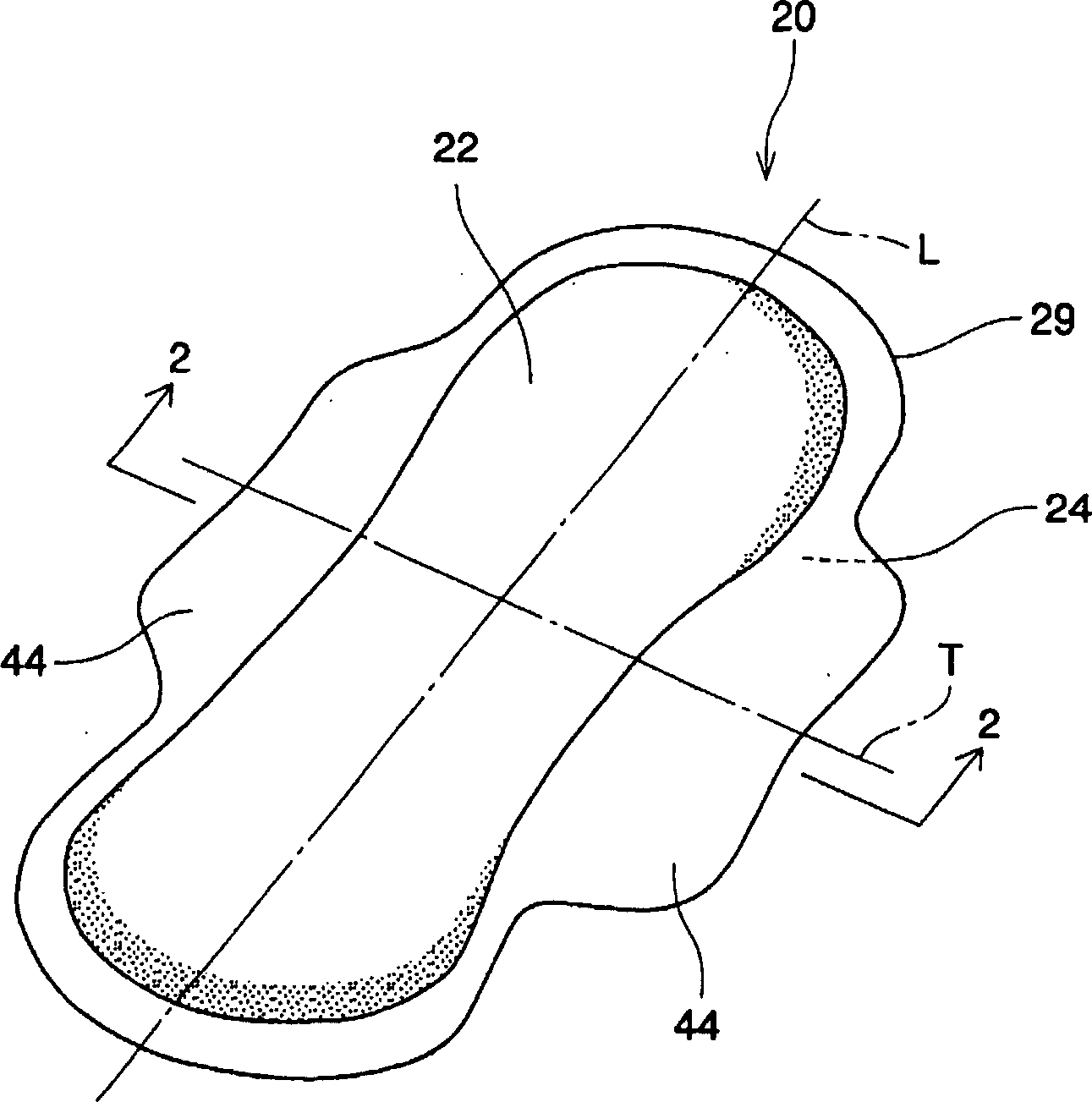
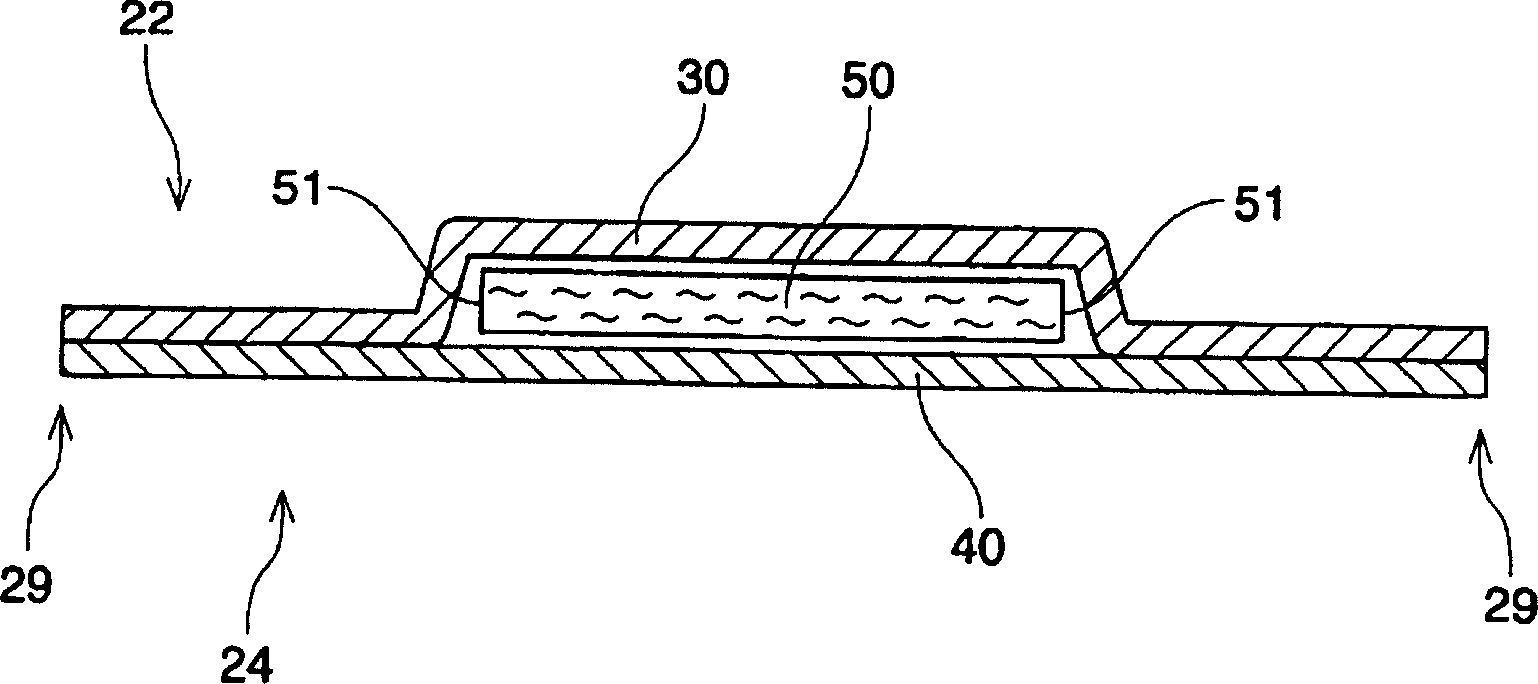
Absorbent article including airlaid mixture material containing thermoplastic fibers treated with phosphate ester or sulfate ester
InactiveUS20050054253A1Superior fluid handlingLow stiffnessAbsorbent padsBaby linensPhosphoric Acid EstersPolymer science
The invention is directed to an absorbent article, comprising: a topsheet; a backsheet; an airlaid mixture material disposed between the topsheet and the backsheet. In one aspect of the invention, the airlaid mixture material includes a) cellulosic fibers, and b) thermoplastic fibers having a surface treated with a surfactant including a phosphate ester, a sulfate ester, or a derivative thereof. The cellulosic fibers and the thermoplastic fibers are bonded together to form a thermally bonded airlaid matrix which preferably has dry density of from about 0.04 to about 0.11 g / cc under pressure of 20 gf / cm2.
Owner:THE PROCTER & GAMBLE COMPANY
Method for preparing cyclic sulfate by directly oxidizing hydrogen peroxide
PendingCN111909129ALess impuritiesHigh purityOrganic chemistryMetal/metal-oxides/metal-hydroxide catalystsPtru catalystCatalytic oxidation
The method comprises the following steps: dropwise adding hydrogen peroxide into a mixture of cyclic sulfite, an organic solvent and a solid catalyst to carry out catalytic oxidation reaction, filtering out the solid catalyst after the reaction is finished, standing filtrate for layering, taking an organic layer, and performing distilling and concentrating to obtain a cyclic sulfate product. Cheaphydrogen peroxide is used for directly catalyzing and oxidizing cyclic sulfite to prepare cyclic sulfate, so that on one hand, the reaction is mild and easy to control, and the reaction conversion rate is high; on the other hand, no waste salt is generated, the evaporation capacity of water is small, energy consumption is low, generated waste water is little, and the production process is more environmentally friendly; the used solid catalyst contains an active component, an active auxiliary agent and an oxide carrier, and can be recycled, so that the consumption of noble metals is reduced, and the production cost is greatly reduced; the cyclic sulfate prepared by the method is few in impurities, high in purity and wide in market prospect.
Owner:CHANGSHU CHANGJI CHEM +1
Fast-permeation, low-form, and easy-degradation mercerizing penetrating agent, and preparation raw materials and preparation process thereof
The invention belongs to the technical field of chemical engineering, and relates to an alkali-resistant penetrating agent used in cotton fabric mercerizing. The invention especially relates to a fast-permeation, low-form, and easy-degradation mercerizing penetrating agent, and preparation raw materials and a preparation process thereof. The invention assists in solving technical problems such as unreasonable technologies of prior art. The preparation raw materials comprises the components of, by mass: 45-50 parts of fatty alcohol sulfate salt, 45-50 parts of fatty alcohol ether sulfate salt, and 1-5 parts of an emulsification agent. Compared with prior art, the penetrating agent, the preparation raw materials and the preparation process have the advantages that: medium-carbon and low-carbon alcohols are adopted as main raw materials, and fatty alcohol sulfate salt is synthesized; separation, purification and structural characterization are carried out; and reasonable formulation is carried out. The preparation process is simple and easy to implement. The product has high quality and low cost.
Owner:ZHEJIANG DAHE CHEM
Antibacterial hand sanitizer and preparation method thereof
The invention discloses an antibacterial hand sanitizer and a preparation method thereof. The antibacterial hand sanitizer comprises (A) 0.05-12% of fungicide coconut oil alcohol polyether ester sulfate zinc; (B) 5-20% of anionic surfactant; (C) 5-20% of ampholytic surfactant; (D) 1-10% of humectants; (E) 0.05-0.5% of chelating agent; (F) the balance of water in the weight of a composition. The antibacterial hand sanitizer containing the fungicide coconut oil alcohol polyether ester sulfate zinc disclosed by the invention has the advantages of being low in thrill, strong in hard water resistance, high in detersive power and the like besides the bacteriostatic efficacy. The influence of the fungicide coconut oil alcohol polyether ester sulfate zinc on the surfactant is small, so that the selectable surfactant in the formula is less limited; the formula can be simply adjusted, so as to adapt to variable market requirements.
Owner:GUANGZHOU LIBY
Bubble bath oil and preparation method thereof
InactiveCN110897937AGood skin careGood moisturizing effectCosmetic preparationsToilet preparationsGlycerolEngineering
The invention discloses bubble bath oil and a preparation method thereof. The components of the bubble bath oil comprise, by weight, 30 to 55% of sweet almond oil, 1 to 4% of cocamidopropyl DEA, 15 to25% of laurinol polyether sulfate TIPA salt, 3 to 8% of propylene glycol, 20 to 35% of lauryl alcohol polyether-4, 1 to 4% of PEG-7 glyceryl cocoate, and 0.5 to 1% of auxiliary component. The bubblebath oil has the advantages that dirt and excessive sebum on the skin can be removed; the grease balance of the skin can be maintained; the itching and dry skin state can be improved; the lubricatingfeeling of the skin can be improved; the nourishing effect is realized during cleaning; and real two-in-one effect of washing and protection is realized.
Owner:广州蜜妆生物科技有限公司
Application of poly mannuronic acid propyl ester sulfate in preparing anti- H1N1 influenza A virus medication
InactiveCN102743409AAvoid infectionPrevent proliferationOrganic active ingredientsAntiviralsCanine kidneyPolymannuronic acid
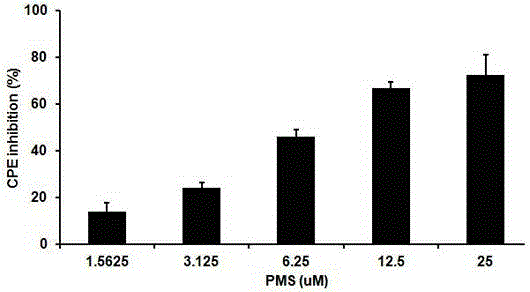
The invention provides applications of poly mannuronic acid propyl ester sulfate (PMS) in preparing anti- H1N1 influenza A virus medication. Experiments of the invention prove that PMS not only has great inhibition effect on the neuraminidase of the influenza A virus, but also has relatively good protection effect on canine kidney epithelial cells infected with the H1N1 influenza A virus, and can reduce the replication of the H1N1 virus with an effect that equal to the positive control medication ribavirin. Additionally, the PMS can effectively reduce the death rate of mice infected with the H1N1 influenza A virus and the survival time of the mice are prolonged. The poly mannuronic acid propyl ester sulfate provided by the invention has significant activity of inhibiting the neuraminidase of the H1N1 influenza A virus, and is proved to have good anti- H1N1 influenza A virus activity both in vivo and in vitro experiments, which shows the wide application prospect of the poly mannuronic acid propyl ester sulfate in preparing anti-H1N1 influenza A virus medication.
Owner:OCEAN UNIV OF CHINA
Heavily foaming detergent mixtures containing fatty acid polyglycol ester sulphates
InactiveUS6235696B1Inorganic/elemental detergent compounding agentsCosmetic preparationsAlkaline earth metalActive agent
Highly foaming detergent mixtures comprising: (a) at least one fatty acid polyglycol ester sulfate of the general formula (I):wherein R1CO represents an acyl group and R1 is selected from the group consisting of linear, branched, saturated and unsaturated chains having from about 6 to about 22 carbon atoms, x is an integer having a value of from 1 to 3, each AO independently represents a CH2CH2O, CH2CH(CH3)O or CH(CH3)CH2O group, and X is selected from the group consisting of an alkali metal, an alkaline earth metal, ammonium, an alkylammonium, an alkanolammonium or a glucammonium; and (b) at least one additional surfactant selected from the group consisting of anionics, nonionics, cationics, amphoterics and zwitterionics are disclosed. The disclosed detergent mixtures exhibit advantageous foaming performance and a high level of foam stability even in the presence of hard water and / or oil.
Owner:COGNIS DEUT GMBH
Sulfonated changium root polysaccharides and preparing method and use thereof
InactiveCN1844160ANovel preparation processLow costOrganic active ingredientsAntineoplastic agentsChangiumSulfated polysaccharides
In this invention, we extract raw polysaccharides from changium root named A B C respectively, and purify to get 6 polysaccharides I,II,III,IV,V,VI. After sulfation, we get sulfated polysaccharides A1, B1,C1. A1 has two sulfate groups on each monosaccharide at the average while B1 and C1 have three. The sulfated polysaccharides' molecular weight is more than 2 million D. The glycoside link is alpha type. The whole procedure of changium root sulfated polysaccharides is as followings: extract raw polysaccharides from changium root, and then get the polysaccharides sulfated. It is advantageous in simplied-manipulation and low cost. The pharmacological experiments prove its obvious suppression on rat transplanting tumor(S-180) and the reduction of platelet aggregation ratio and whole blood viscosity. It also reduces coagulant time.
Owner:NANJING UNIVERSITY OF TRADITIONAL CHINESE MEDICINE
Infiltration improvement additive, pole piece, infiltration method of pole piece, battery and preparation method of battery
ActiveCN111640946AImprove wettabilityHigh porosityFinal product manufacturePositive electrodesElectrolytic agentPhosphoric Acid Esters
The invention provides an infiltration improvement additive, a pole piece, an infiltration method of the pole piece, a battery and a preparation method of the battery. The infiltration improvement additive is one or a mixture of more than two of carbonic ester substances, sulfate ester substances and phosphate ester substances. According to the infiltration improvement additive, the infiltration improvement additive can be used for pole piece preparation, can be dissolved into an electrolyte to become part of the electrolyte in the final lithium ion battery preparation process, enables the pole piece to be filled with the electrolyte, and improves the infiltration performance of the pole piece. Meanwhile, the position occupied by an original additive is enabled to become pores, the porosity of the pole piece is increased, and the storage of the electrolyte is facilitated.
Owner:TIANJIN ENERGIES
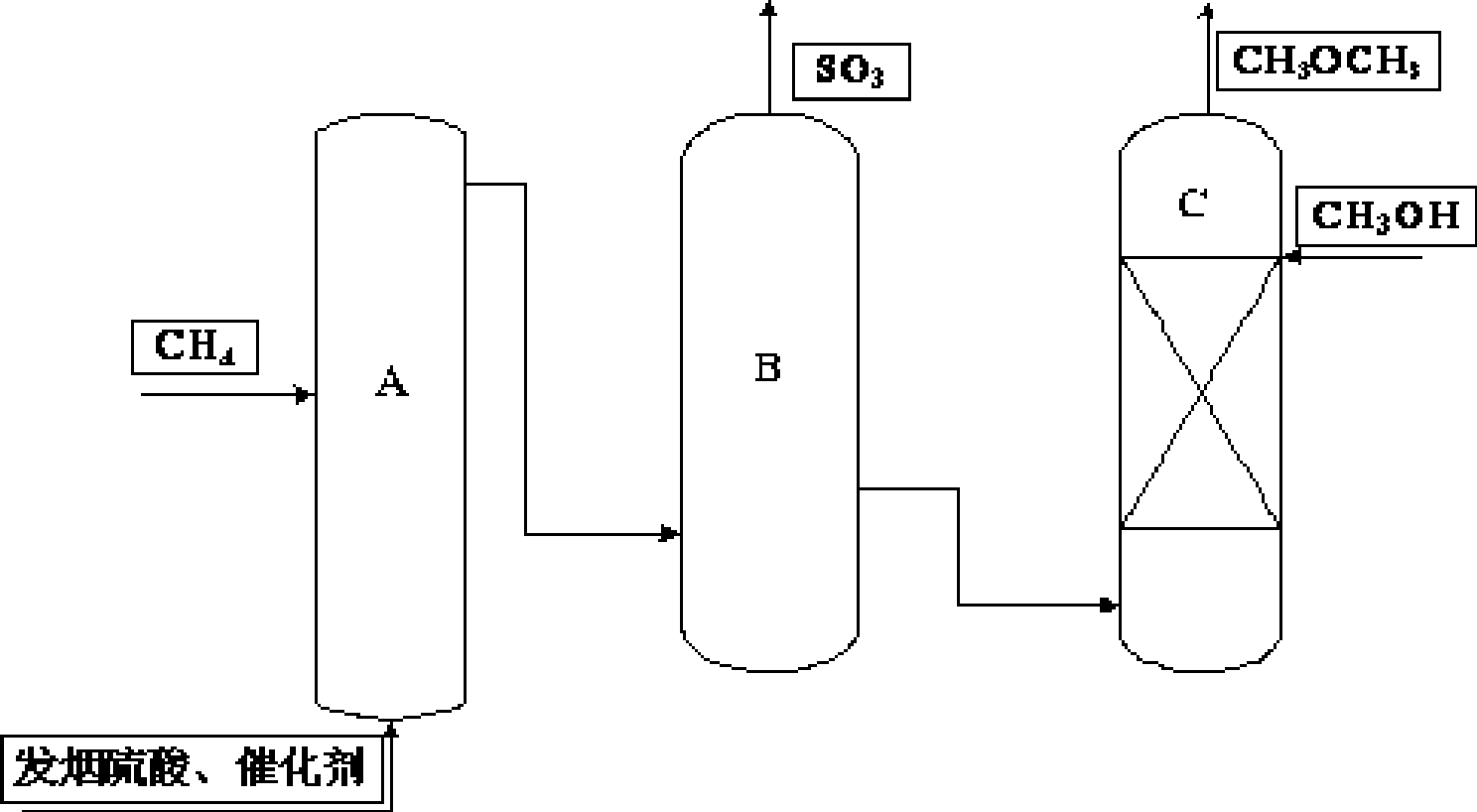
Process for preparing dimethyl ether
InactiveCN101486629AFully alcoholyzedControl join rateEther preparation by ester reactionsAlcoholSulfate
The invention discloses a method for preparing dimethyl ether. The method leads methane to be activated and synthesized into sulfate material which is then added into an alcoholysis reactor together with the methanol; the temperature of the alcoholysis reactor is controlled between 35 DEG C and 180 DEG C and alcoholysis is conducted for 0.5 hour to 5 hours; the dimethyl ether prepared is vaporized from the alcoholysis reactor and collected. The preparing method has the advantages of simple synthetic route, high utilization ratio of raw materials, few byproducts, mild reaction conditions, low energy consumption and being suitable for continuous production and pertains to the field of dimethyl ether preparation.
Owner:HANERGY TECH
Sulphated heparan sulfated for inhibiting cell proliferation
InactiveCN103288981AAvoid diversionPrevent relapseOrganic active ingredientsAntineoplastic agentsEthanol precipitationTrimethyloxamine
A heparan sulfated derivative is prepared from heparan sulfated by sulphating. A preparation method comprises: swelling heparan sulfated by anhydrous dimethyl formamide, adding a sulfur trioxide-trimethylamine complex, heating for esterification, dissolving, dialyzing, freeze-drying precipitation components of the reactant by distilled water, then dissolving by sodium chloride solution, precipitating by ethanol, centrifuging, removing a supernatant, drying, and then obtaining sulphated heparan sulfated. The heparan sulfated derivative can inhibit activities of tumor cells and umbilical vein endothelial cell proliferation.
Owner:SHENZHEN HEPALINK PHARMA GRP CO LTD
Water-soluble bio-based sulfate/sulfonate preparation process and uses of water-soluble bio-based sulfate/sulfonate as green environmental protection adhesive
ActiveCN104774575ABroaden the prospect of wide applicationHigh yieldCellulose adhesivesChitin adhesivesSolubilityPrice ratio
The present invention provides an adhesive, which comprises, by mass, 30-100% of cellulose sulfate, and less than or equal to 20% of chitosan and / or carboxymethyl chitosan salt. According to the present invention, the newly discovered sulfonation system and the abundant biomass raw material are utilized to develop cheap and green environmental protection water-soluble bio-based adhesive series products, wherein the material having the high performance price ratio is adopted to replace the existing urea-formaldehyde resin, polyvinyl alcohol, polyvinyl formal and soluble starch so as to fundamentally solve problems of fatal disadvantages of high formaldehyde residue, long term release, serious harm on human body health, and the like of the existing urea-formaldehyde resin, polyvinyl formal and other adhesives, and poor adhesion effect and high consumption of soluble starch and other natural high polymers; and the high-yield and stable bio-based sulfate / sulfonate adhesive is obtained, and the wide application prospects of the series of the adhesives can be expanded through the wide raw material sources, the low preparation cost, the excellent water solubility, the polymer performance, the formaldehyde-free green environmental protection, and other multiple advantages.
Owner:SOUTHWEST UNIV +1
Anti-hair-loss shampoo for women
InactiveCN108113932AGood antibacterial effectImprove the growing environmentCosmetic preparationsHair cosmeticsCetostearyl alcoholChemistry
The invention relates to a hair growing type special cosmetic and in particular relates to anti-hair-loss shampoo for women. The anti-hair-loss shampoo for women comprises the following raw material components in parts by weight: water, ammonium laureth sulfate, ammonium lauryl sulfate, sodium lauroyl sarcosine, cocamidopropyl betaine, glycol distearate, guar gum hydroxypropyl trimethyl ammonium chloride, sodium chloride, hydrolyzed wheat protein, essence, panthenol, cetostearyl alcohol, citric acid, disodium EDTA, methylparaben, methylisothiazolinone, an anti-hair-loss agent and an antioxidant. The anti-hair-loss shampoo for women adopts a natural plant formula, contains ingredients such as cacumen biotae extract, polygonum multiflorum root extract, fructus viticis extract, rhodiola roseaextract and ganoderma atrum extract, can clean scalp dirt, has better antibacterial effect on scalp, nourishes the scalp, is beneficial to improvement of a hair growing environment, improves toughness of hairs, reduces hair loss and broken hairs and has a protective effect on damaged hairs.
Owner:东莞权利得工业设计有限公司
Preparation method of perfluoroalkyl ethyl alcohol
The invention discloses a method for preparing perfluoroalkyl ethyl alcohol with perfluoroalkyl ethyl iodide, which comprises the following steps: (a) fuming sulphuric acid and the perfluoroalkyl ethyl iodide are mixed at 55-75DEG C, thus obtaining premixed reactants; (b) the premixed reactants are heated to 130DEG C at most to ensure the fuming sulphuric acid to react with the premixed reactants of the perfluoroalkyl ethyl iodide, thus forming sulfate; and (c) hydrolysis is carried out and the perfluoroalkyl ethyl alcohol is separated.
Owner:SHANGHAI 3F NEW MATERIAL
Polygonatum odoratum polysaccharide sulfation method
The present invention belongs to the technical field of sulfation, and specifically discloses a polygonatum odoratum polysaccharide sulfation method. The method comprises: dissolving dried polygonatum odoratum polysaccharide in a reaction solvent, adding a pyridine sulfur trioxide complex, carrying out a reaction for 4-9 h at a temperature of 40-80 DEG C, neutralizing, carrying out ethanol precipitation, carrying out dialysis, concentrating, and drying to obtain the polygonatum odoratum polysaccharide sulfate, wherein a mass volume ratio of the polygonatum odoratum polysaccharide to the reaction solvent is 1:10-1:500 g / ml, and a mass ratio of the polygonatum odoratum polysaccharide to the pyridine sulfur trioxide complex is 1:5-5:1.
Owner:GUANGDONG PHARMA UNIV
Preparation method of D-p-hydroxyphenylglycine methyl ester
PendingCN111153821AGuaranteed yieldGuaranteed quality indicatorsOrganic compound preparationOrganic chemistry methodsMethanolEster sulfate
Owner:SHANXI WEIQIDA PHARMA IND
Spectrophotometry for testing activity of pyruvic acid dehydrogenase system
InactiveCN101017134ALow priceSmall standard errorMicrobiological testing/measurementColor/spectral properties measurementsWavelengthMitophagy
This invention relates to one splitting photometry to test pyruvate dehydrogenase enzyme activity, which comprises the following steps: using splitting meter under situation of underlay of sodium ketopropionate and aid factor magnesium chloride and cocarboxylase using phenazine methyl ester sulfate as electron deliver part and thiazole blue as electron receiver restored by the product of pyruvate dehydrogenase through hydroxyethylation-cocarboxylase with maximum absorptive peak from length for 400 to 430 nanometer into 540 to 640 nanometer; through testing wave length for 540 to 640 nanometer absorptive add volume to determine the restore volume and defining enzyme activity.
Owner:HUAZHONG NORMAL UNIV
Anti-hair-loss and hair-strengthening shampoo and preparation method thereof
PendingCN113576988ABlood normalizationNormalization of blood circulationCosmetic preparationsHair cosmeticsSodium acetateDMDM hydantoin
The invention provides anti-hair-loss and hair-strengthening shampoo and a preparation method thereof, and the anti-hair-loss and hair-strengthening shampoo comprises the following components in percentage by mass: 1.6-2.3% of ammonium laureth sulfate, 2.2-3.6% of ammonium lauryl sulfate, 6.8-7.5% of sodium lauroamphoacetate, 4.5-5.3% of a cacumen biotae extract, 4.5-5.3% of a polygonum multiflorum root extract, 4.8-5.6% of a ginger root extract, 1.5-2.1% of polydimethylsiloxane, 5.5%-6.3% of sodium dodecyl benzene sulfonate, 0.5%-1.3% of cocamide MEA, 1.0%-1.6% of sodium lauroyl sarcosinate, 0.02%-0.1% of PCA sodium, 0.2%-0.5% of essence, 0.5%-1.0% of DMDM hydantoin, 0.6%-1.2% of guar gum hydroxypropyl trimethyl ammonium chloride, 0.05%-0.15% of pyridoxine HCl, 1.0%-1.5% of sodium chloride, 0.05%-0.15% of allantoin, 0.01%-0.08% of hydroxypropyl methyl cellulose, 0.8%-1.1% of citric acid, 1.8%-2.3% of EDTA disodium and the balance deionized water. By adding the polygonum multiflorum extract, the ginger root extract and the cacumen biotae extract, the shampoo can effectively clean scalp, promote generation of hair melanin, prevent and treat alopecia and nourish hair roots and hair follicles.
Owner:韩伟伟
Novel shampoo free of silicone oil and preparation method thereof
InactiveCN105662929ADoes not affect smoothnessDoes not affect combabilityCosmetic preparationsHair cosmeticsLanolinD-PANTHENOL
The invention belongs to the technical field of cosmetics and discloses a new type of shampoo without silicone oil. The formula of the new type of shampoo without silicone oil includes: sodium lauroyl glutamate 5-15%, alkyl sulfate monoethanolamine salt 3-9%, sodium lauryldimethylaminocaproate 1-5%, polyquaternium-7 0.5-3%, myristyl ether 0.1-1%, chamomile essential oil 1-5%, D pan Alcohol 1-3%, Campanola 0.1-1%, Lanolin 1-5%, Citric Acid 0.1-0.8%, EDTA 0.1-0.5%, Thickener 0.5-2%, Preservative 0.2-1% , essence 0.2-1%, and the balance pure water. The raw materials of the novel shampoo of the present invention do not contain silicone oil components, and the scalp will not be damaged during use. By adding water-soluble plant essential oil components and vitamin components instead of silicone oil, the hair can be cared for while washing the hair without affecting the smoothness and combability of the hair after shampooing, avoiding long-term contact of the scalp with washing Health hazards caused by silicone oil components in hair.
Owner:SICHUAN LIJIU INTPROP SERVICE CO LTD
Method for synthesizing reactive yellow DF 3RS
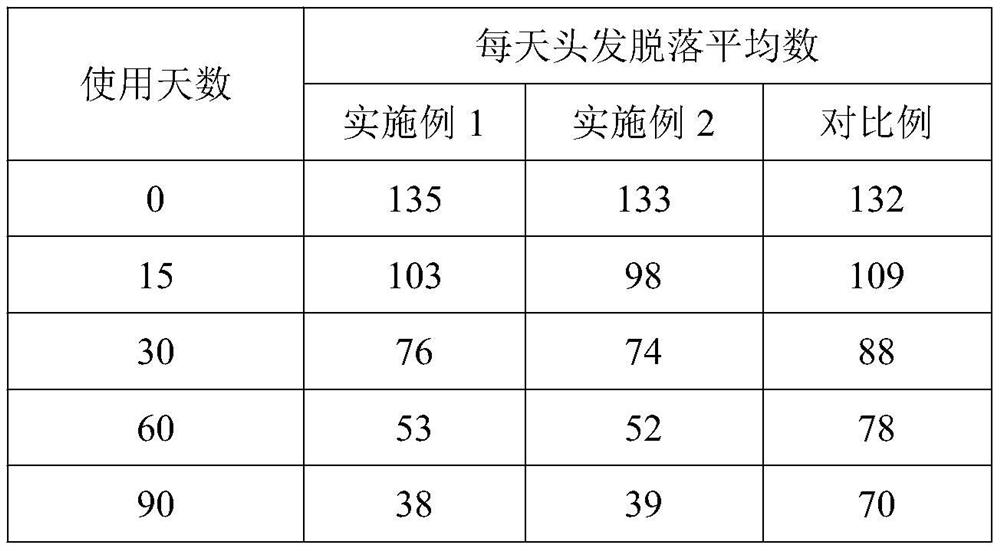
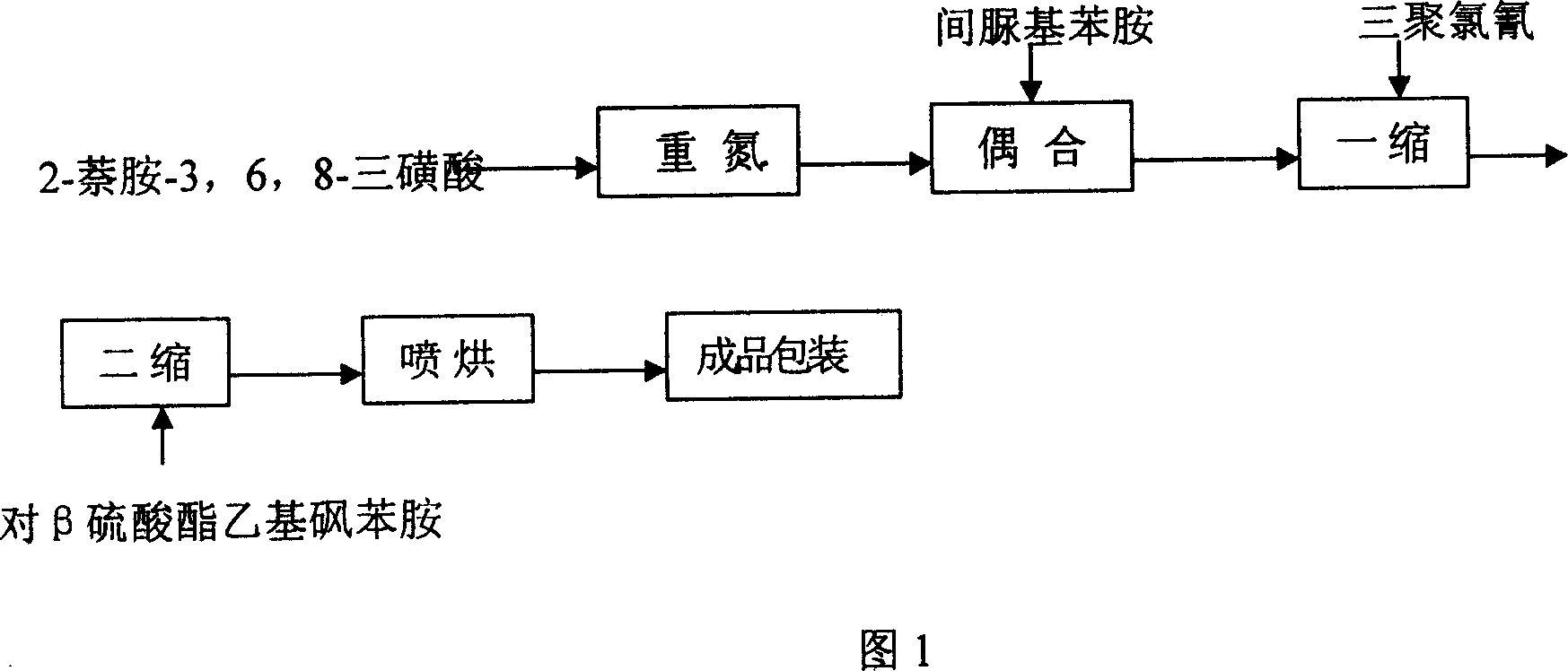
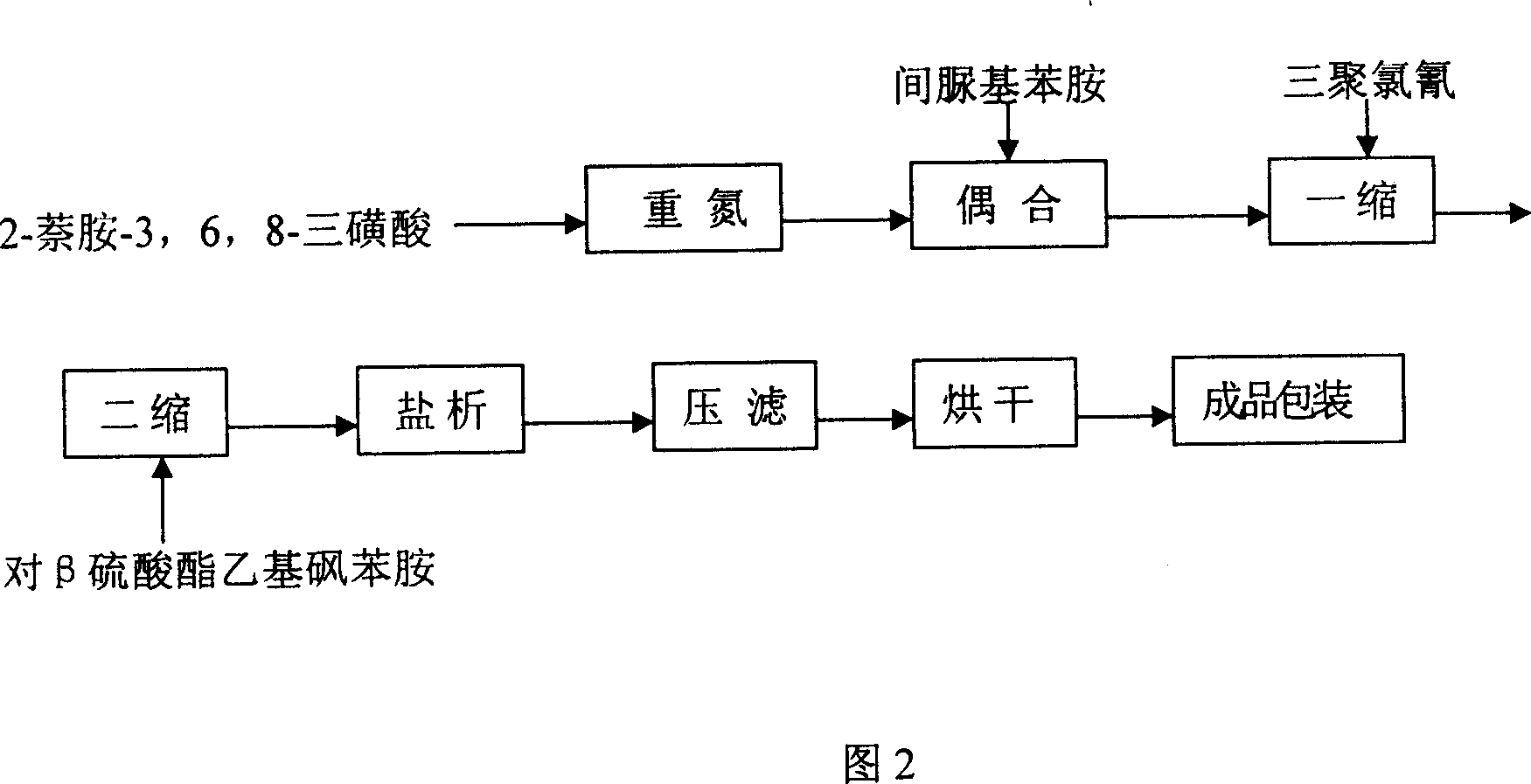
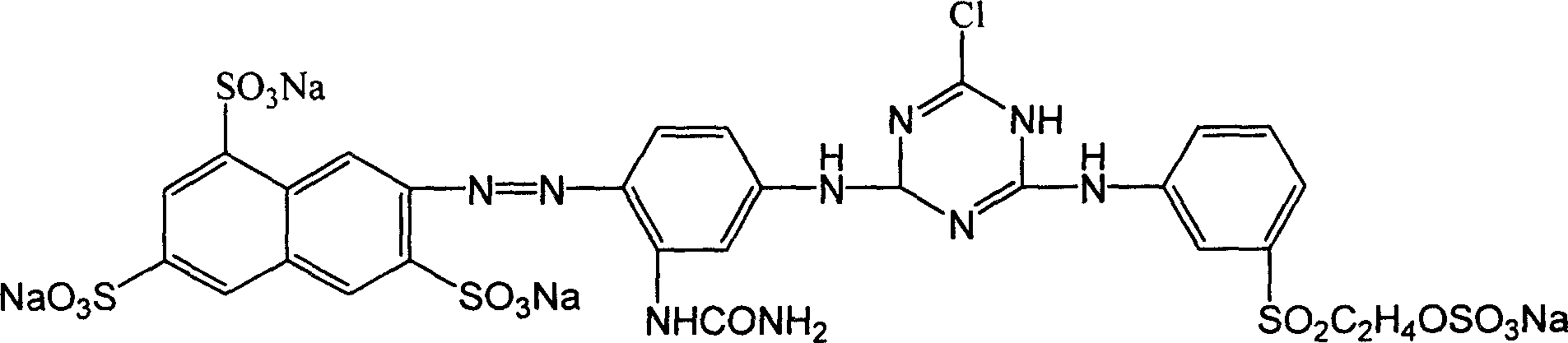
This invention discloses a method for synthesizing active yellow DF-3RS. The method comprises: (1) pulping 2-naphthylamine-3,6,8-trisulfonic acid, adding HCl and NaNO2, and reacting to obtain diazo salt; (2) pulping m-ureido-naphthylamine, adding NaHCO3 and the above diazo salt, and reacting to obtain a coupling compound; (3) adding the coupling compound into pulped cyanuric chloride suspension, and reacting to obtain a first condensate; (4) adding sulfate ethylsulfone naphthylamine, reacting, removing inorganic salts with an ionic membrane, and spray-drying to obtain yellow DF-3RS. The method can reduce the deterioration of the environment, and raise the product yield.
Owner:泰州市姜堰区东风染料化工厂
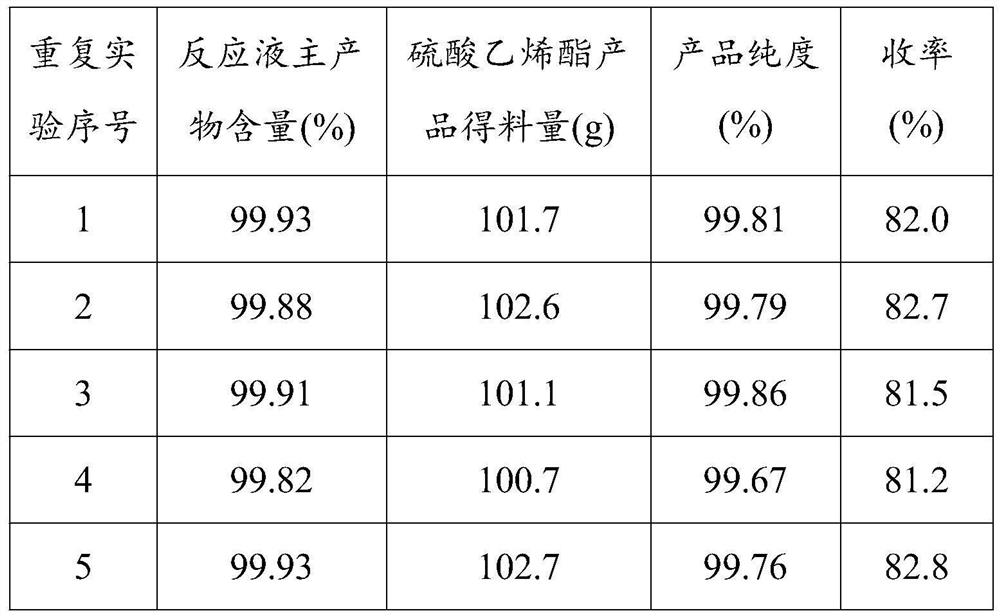
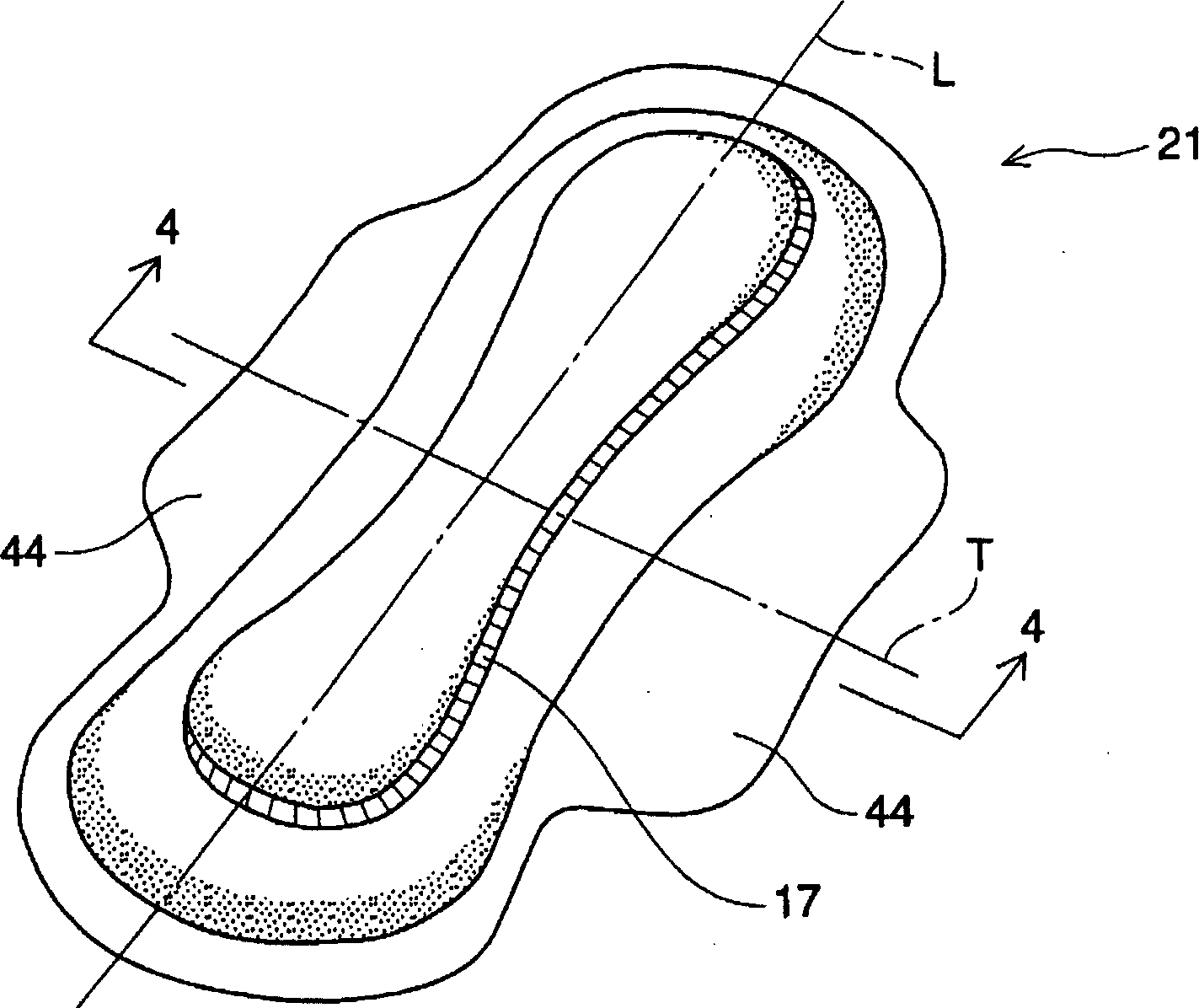
Absorbent article including airlaid mixture material containing thermoplastic fibers treated with phosphate ester or sulfate ester
InactiveCN1678262AExcellent fluid handling performanceImproved Fluid HandlingLayered productsAbsorbent padsPolymer chemistryPhosphate
The invention is directed to an absorbent article, comprising: a topsheet; a backsheet; an airlaid mixture material disposed between the topsheet and the backsheet. In one aspect of the invention, the airlaid mixture material includes a) cellulosic fibers, and b) thermoplastic fibers having a surface treated with a surfactant including a phosphate ester, a sulfate ester, or a derivative thereof. The cellulosic fibers and the thermoplastic fibers are bonded together to form a thermally bonded airlaid matrix which preferably has dry density of from about 0.04 to about 0.11 g / cc under pressure of 20 gf / cm<2>.
Owner:THE PROCTER & GAMBNE CO
Method for detecting parecoxib sodium sulfate genotoxic impurities
PendingCN111413440AHigh detection sensitivityHigh precisionComponent separationGas liquid chromatographicDiethylsulfate
The invention relates to a method for detecting parecoxib sodium sulfate genotoxic impurities. The method comprises the following steps of dissolving a dimethyl sulfate reference substance, a diethylsulfate reference substance and a diisopropyl sulfate reference substance to obtain a sulfate impurity reference substance solution, dissolving parecoxib sodium to be detected to obtain a sample solution to be detected, and carrying out gas chromatography-mass spectrometry determination on the sulfate impurity reference substance solution and the sample solution to be detected. The chromatographicconditions are as follows: a filler of the gas chromatographic column is selected from one of a non-polar filler, a weak polar filler and a medium polar filler; and mass spectrum conditions comprisethat an electrospray ion source and a positive ion scanning mode are selected. The method is high in detection precision, has very high specificity and durability, and is simple and convenient to operate. The separation degree between genotoxic impurities is greater than 2.0, and the method can be used for quality control of parecoxib sodium bulk drugs.
Owner:SHANGHAI CHENPON PHARMA TECH
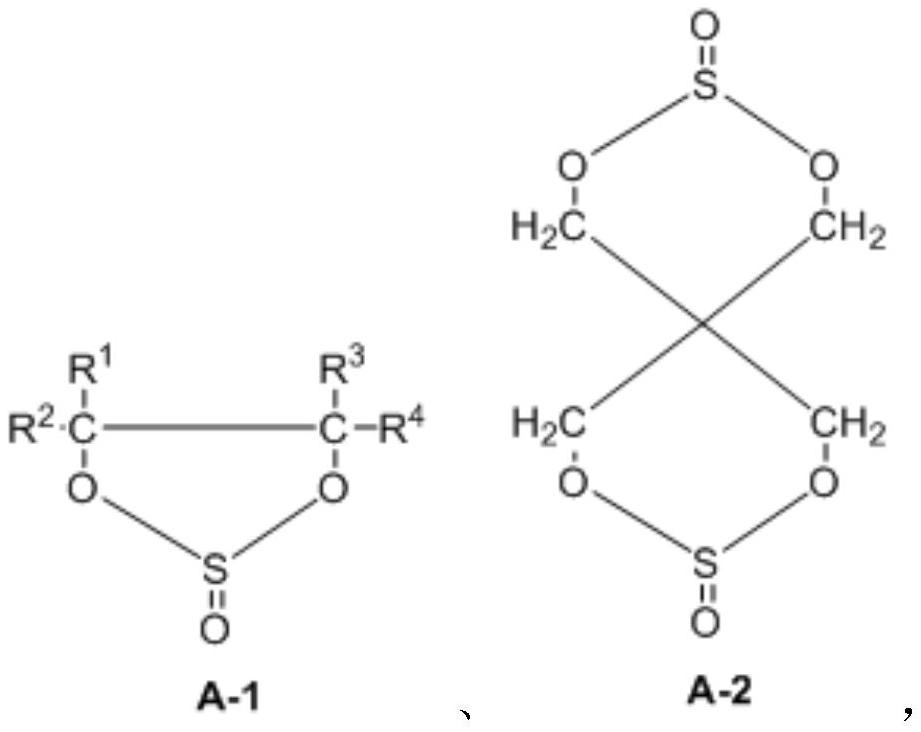
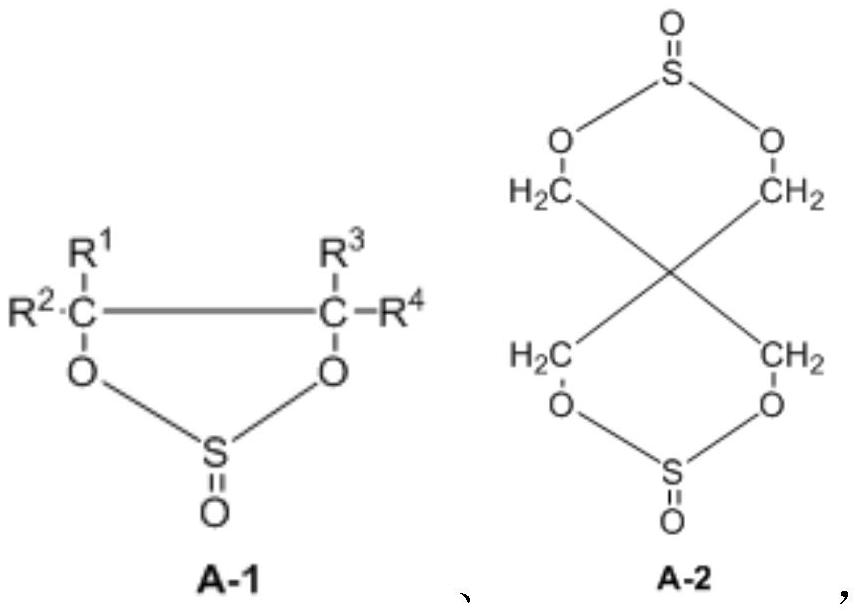
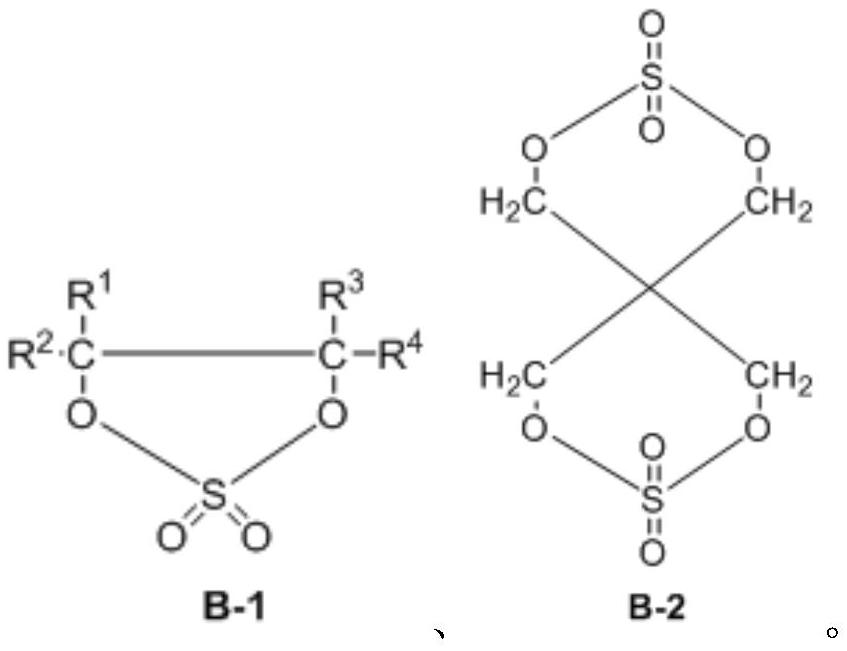
Preparation method of dicyclic sulfite and dicyclic sulfate
The invention provides a preparation method of dicyclic sulfite. The dicyclic sulfite is prepared by mixing a compound A and thionyl chloride and reacting; the compound A has a structure as shown in a formula (1) in the specification; the dicyclic sulfite has a structure as shown in a formula (2) in the specification; R1 to R8 are independently selected from H, a halogen atom, a cyano group, silane trifluoride or at least one X-substituted alkyl group with 1 to 3 carbon atoms when appearing at each time; and X is respectively and independently selected from H, F, silane group, cyano group or isocyanate group. The preparation method is mild in reaction, free of solvent participation, short in reaction time and easy in process control, and can be used for preparing high-yield and high-purity dicyclic sulfite.
Owner:JIUJIANG TINCI ADVANCED MATERIALS CO LTD
Finishing process for garment fabric
InactiveCN105064012AImprove antibacterial propertiesGood antifoulingFibre treatmentEster sulfateStearic acid
The invention discloses a finishing process for garment fabric. The process comprises steps as follows: 1) 4.4 parts of stearic acid by mass, 3.6 parts of clove essential oil by mass and 1.2 parts of dibutyl ester by mass are placed into 13.2 parts of deionized water by mass for even mixing; 2) then 1.8 parts of sodium ricinoleic acid n-butyl ester sulfate by mass and 2.7 parts of polyacrylamide by mass are added slowly and simultaneously, the mixture is continuously mixed until being fully and evenly mixed, and a finishing agent is prepared; 3) the fabric is subjected to padding treatment in an impregnation liquid containing the finishing agent and dried at the constant temperature. The fabric finished with the finishing process for the garment fabric has excellent antibacterial performance and antifouling performance and high washability.
Owner:ANHUI GUIGU E COMMERCE
Fluoropolymer having S-sulfate group and water/oil repellent composition containing the polymer
InactiveUS8394887B2Easy to waterExcellent oil-repellencyLayered productsLiquid repellent fibresPolymer scienceEster sulfate
A fluoropolymer comprising: (A) repeating unit derived from an S-sulfate monomer having an —S—SO3— group and carbon to carbon double bond; and (B) repeating unit derived from a fluoromonomer having a fluorine atom and carbon to carbon double bond. This fluoropolymer can be a constituent of water / oil repellent agent having excellent water / oil repelling capability, and is stable in air and permits an arbitrary control of crosslinking.
Owner:DAIKIN IND LTD
Flame retardant coating composition
The invention provides a water-based flame retardant coating. The water-based flame retardant coating comprises the following components in parts by weight: 20-30 parts of acrylate, 5-10 parts of sulfate, 0.2-0.4 part of potassium sulfate, 1-2 parts of nonylphenol polyoxyethylene ether, 8-12 parts of epoxy modified alcohol ester, 5-6 parts of tetraethoxysilane, 3-5 parts of a curing agent, 10-15 parts of filler, 12-20 parts of a dispersing agent and 30-45 parts of water. The water-based flame retardant coating has the beneficial effects that by applying an organophosphorus ester compound to the coating to form a synergistic flame retardant system with silicon, the coating has good compatibility and flame retardance; meanwhile, the coating has excellent adhesive property, is low in cost, has good component compatibility, is environmentally friendly and flame-retardant and has excellent application value.
Owner:WUXI JIABANG ELECTRIC POWER PIPE FACTORY
Hair lotion for preventing hair loss and promoting hair growth, containing ganoderma niger lloyd plant extract essence and preparation method of hair lotion
InactiveCN111700841APromote growthImprove hair qualityCosmetic preparationsHair cosmeticsBiotechnologyGlycerol
The invention discloses hair lotion for preventing hair loss and promoting hair growth, containing ganoderma niger lloyd plant extract essence and a preparation method of the hair lotion. The hair lotion comprises the following raw materials of water, a ganoderma niger lloyd extract, a ginseng root extract, a ligusticum wallichii extract, a fleeceflower root extract, a radix angelica sinensis extract, a Chinese soapberry seed peel extract, amine laureth sulfate, cocamidopropyl betaine, a Chinese honeylocust fruit extract, ammonium lauryl sulfate, polyquaternary ammonium salt-47, a camellia seed extract, cocamide methyl MEA, camellia seed oil, glycerine, a black mulberry extract, panthenol, sodium C10-16 pareth sulfate, a Chinese arborvitae twig and leaf extract, cetearyl alcohol, Guar hydroxypropyltrimonium chloride, a ginger root extract, PPG-3 myristyl ether, biotin, citric acid, a radix sophorae flavescentis extract, phenoxyethanol, essence, and an astragalus complanatus extract. The product is used, so that hair follicle can be activated, hair growth can be promoted, the hair can be nourished and cared, hair quality can be effectively improved, the pH of the hair can be effectively adjusted, and the hair lotion has the effects of diminishing inflammation and restraining bacteria, and can enable the head to be refreshing and clean.
Owner:梧州市华夏医药科技有限公司
Synthesis method of cyclic sulfate
Owner:HUNAN ASTAR BIO CHEM TECH
Method for preparing fluoro olefin
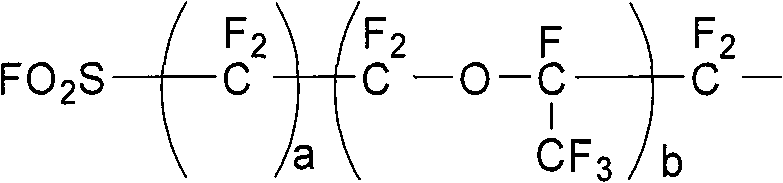
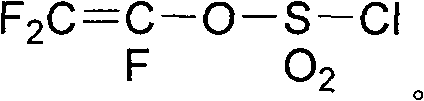
The invention discloses a method for preparing fluoro olefin, which comprises that: a perfluoro straight chain or branched chain acyl fluoride compound reacts with trifluoro vinyl ethyl sulfate in an organic solvent under the action of a catalyst so as to form a product having a general formula shown by the formula (I). The method avoids a pyrolysis route and uses no unconventional monomers, the used trifluoro vinyl ethyl sulfate can be prepared at a yield of above 95 percent, and a fluoro olefin monomer is prepared easily through an addition reaction, and a high-purity monomer can be obtained through rectification. CF2=CF-O-Rf (I).
Owner:SHANDONG DONGYUE WEILAI HYDROGEN ENERGY MATERIAL CO LTD