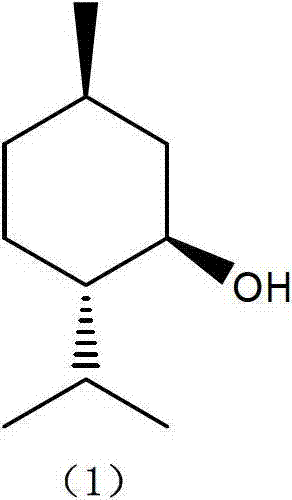
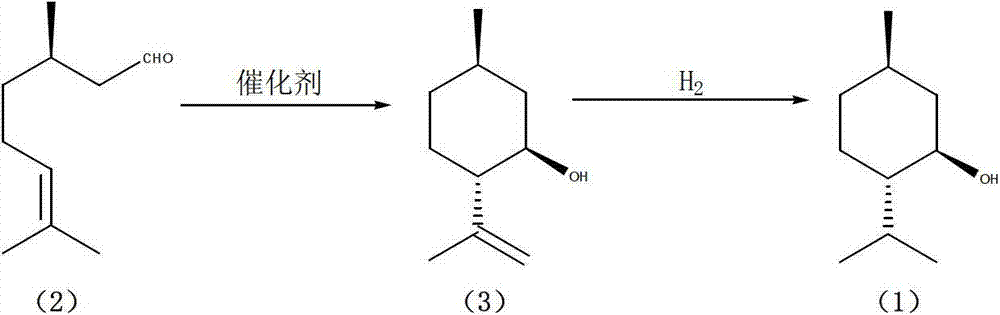
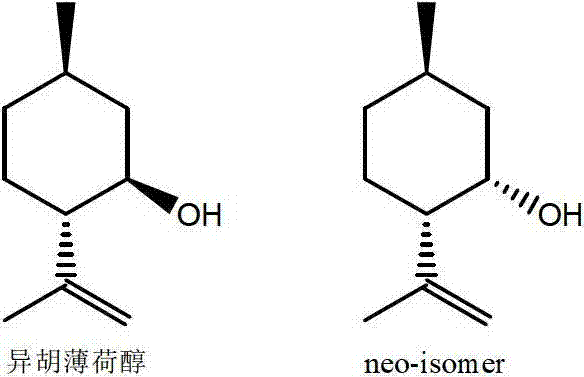
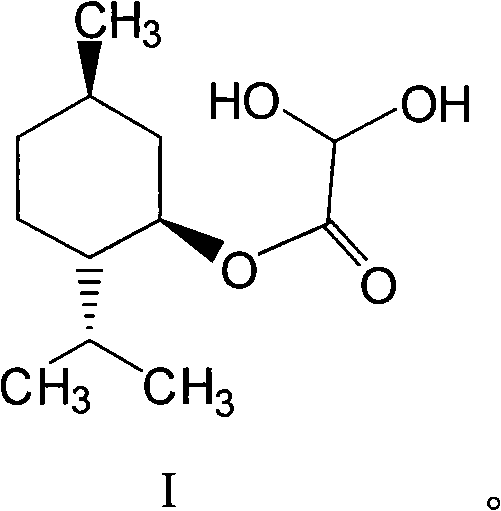
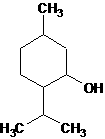
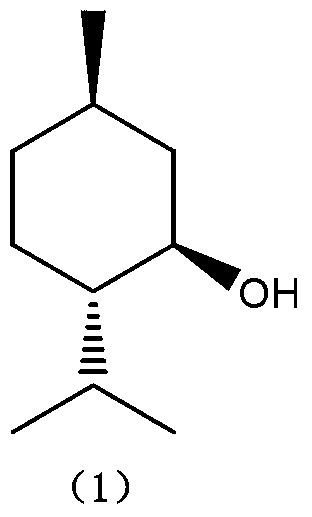
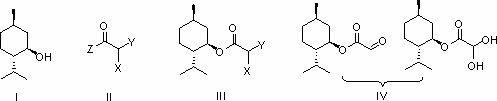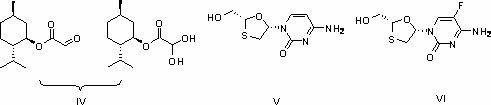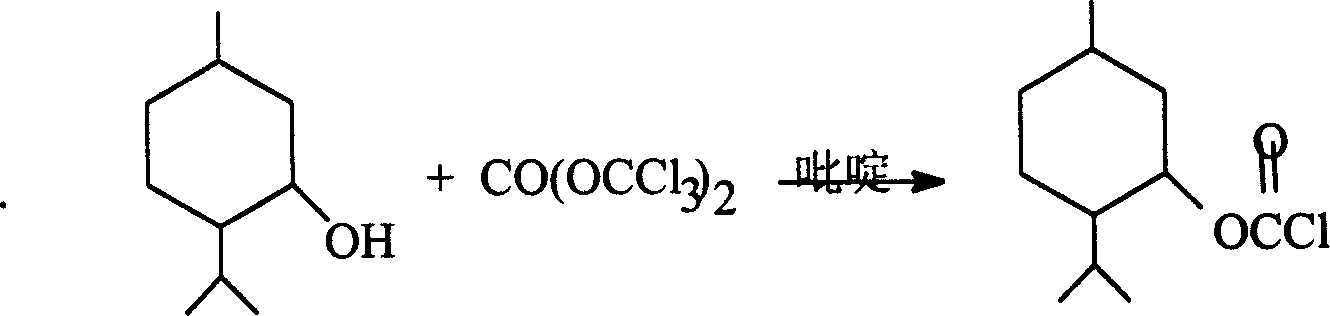Patents
Literature
127 results about "L menthol" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
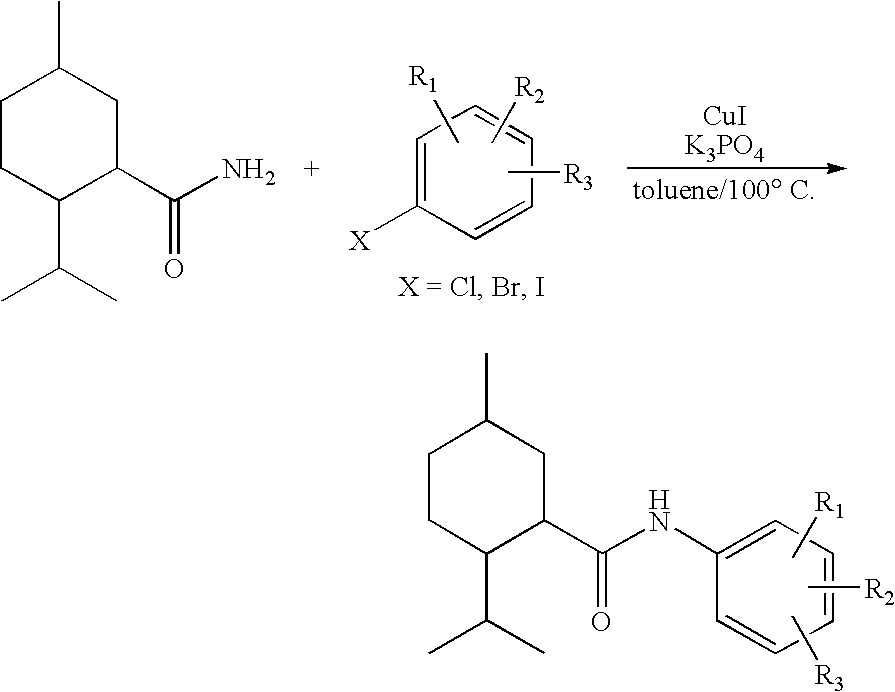
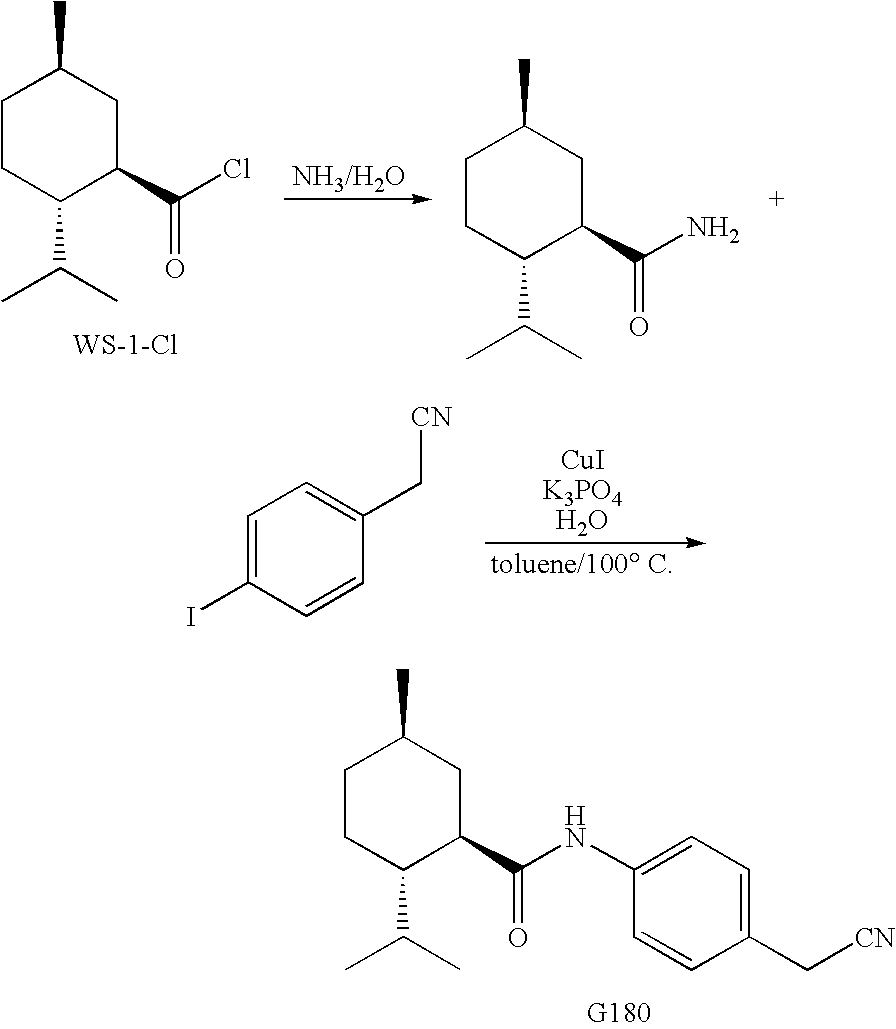
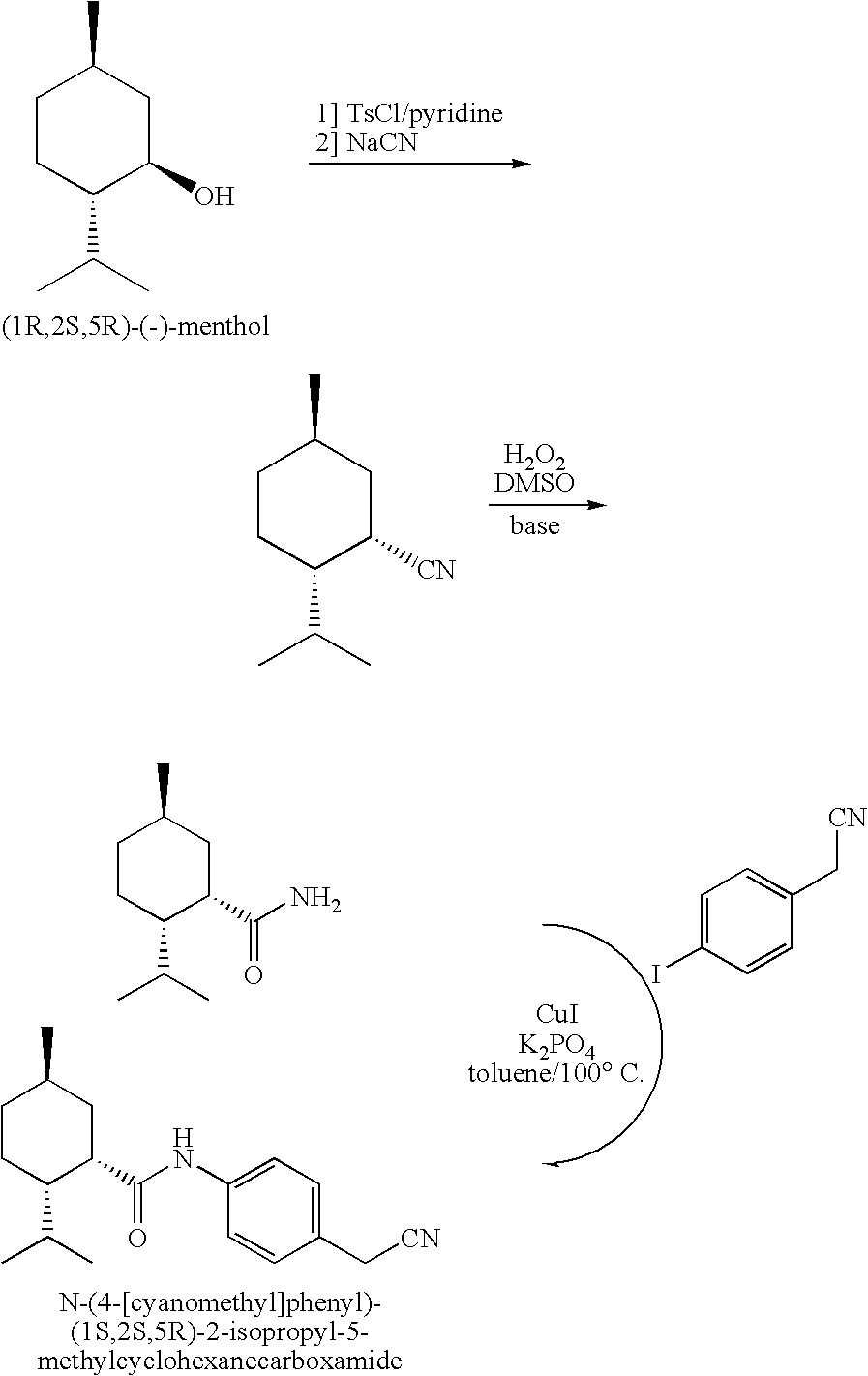
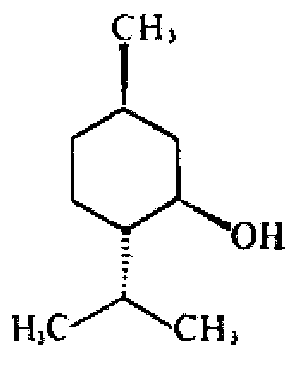
Synthesis of Cyclohexane Derivatives Useful as Sensates in Consumer Products
ActiveUS20100076080A1Improve cooling effectPotent and long lasting cooling effectBiocideCosmetic preparationsCooling effectL menthol
The present invention provides synthetic routes for preparing various isomers of cyclohexane-based coolants, such as menthyl esters and menthanecarboxamide derivatives, in particular those substituted at the amide nitrogen, for example with an aromatic ring or aryl moiety. Such structures have high cooling potency and long lasting sensory effect, which make them useful in a wide variety of consumer products. One synthetic route involves a copper catalyzed coupling of a primary menthanecarboxamide with an aryl halide, such reaction working best in the presence of potassium phosphate and water. Using this synthetic route, specific isomers can be prepared including the menthanecarboxamide isomer having the same configuration as l-menthol and new isomers such as a neoisomer having opposite stereochemistry at the carboxamide (C-1) position. The neoisomer unexpectedly has potent and long lasting cooling effect. Preparation schemes for neoisomers of other menthyl derivatives which are useful as coolants, including esters, ethers, carboxy esters and other N-substituted carboxamides are also provided.
Owner:THE PROCTER & GAMBLE COMPANY
Preparation method of cooling mint type cigarette filter additive and application of cooling mint type cigarette filter additive
The invention belongs to the technical field of mint type cigarettes and particularly relates to a preparation method of the cooling mint type cigarette filter additive and application of the cooling mint type cigarette filter additive. The cooling mint type cigarette filter additive comprises the following components of L-menthol, sorbitol, glycerin, propylene glycol, peppermint oil, spearmint oil. The preparation method of the cooling mint type cigarette filter additive comprises the following step of evenly mixing the L-menthol, the sorbitol, the glycerin, the propylene glycol, the peppermint oil and the spearmint oil to obtain the cooling mint type cigarette filter additive. The invention further provides the application of the cooling mint type cigarette filter additive in preparation of cooling mint type cigarette filters. Coolness and fragrance of the cooling mint type cigarette filter additive can remain for about one year and last long in the process of smoking cigarettes, smokers feel naturally and gently cool when smoking cigarettes, and irritation is reduced remarkably.
Owner:CHINA TOBACCO GUANGXI IND
Method for synthesizing L-menthol
InactiveCN101602651AWide variety of sourcesLow pricePreparation by isomerisationPreparation by hydrogenationIsomerizationOrganic synthesis
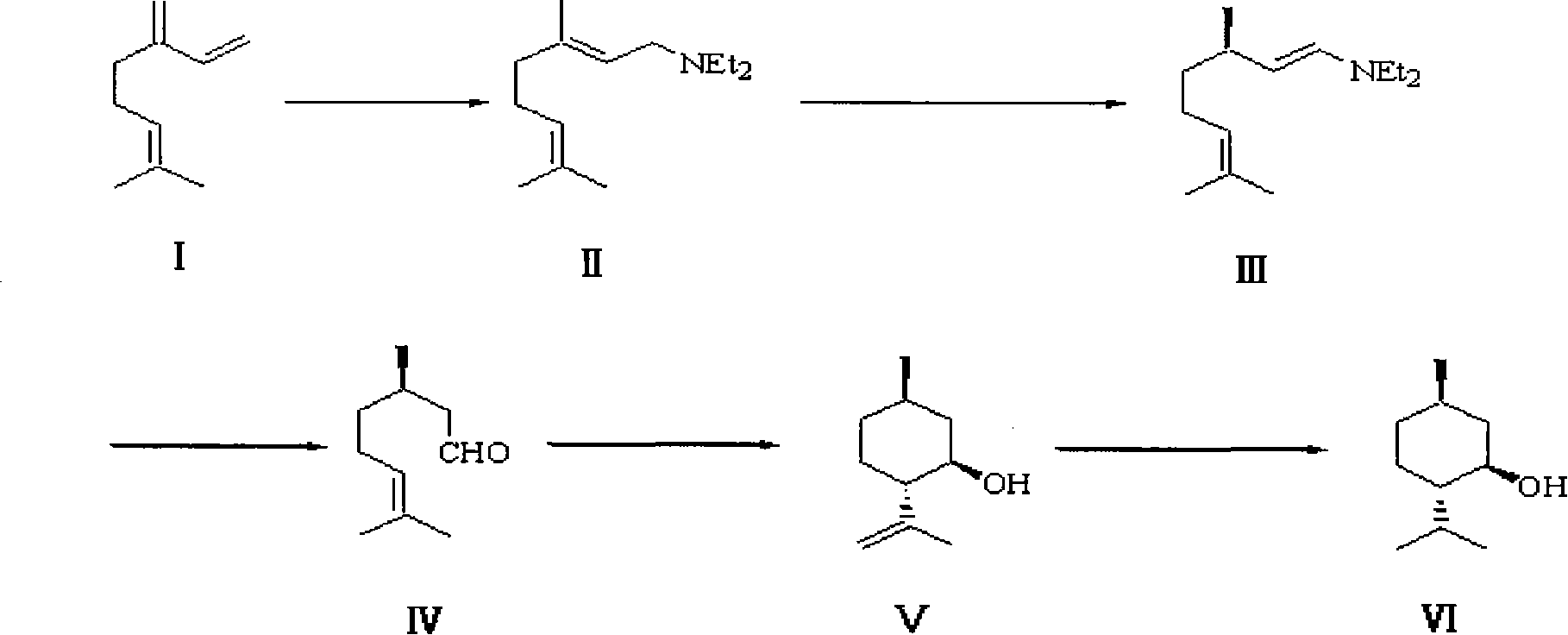
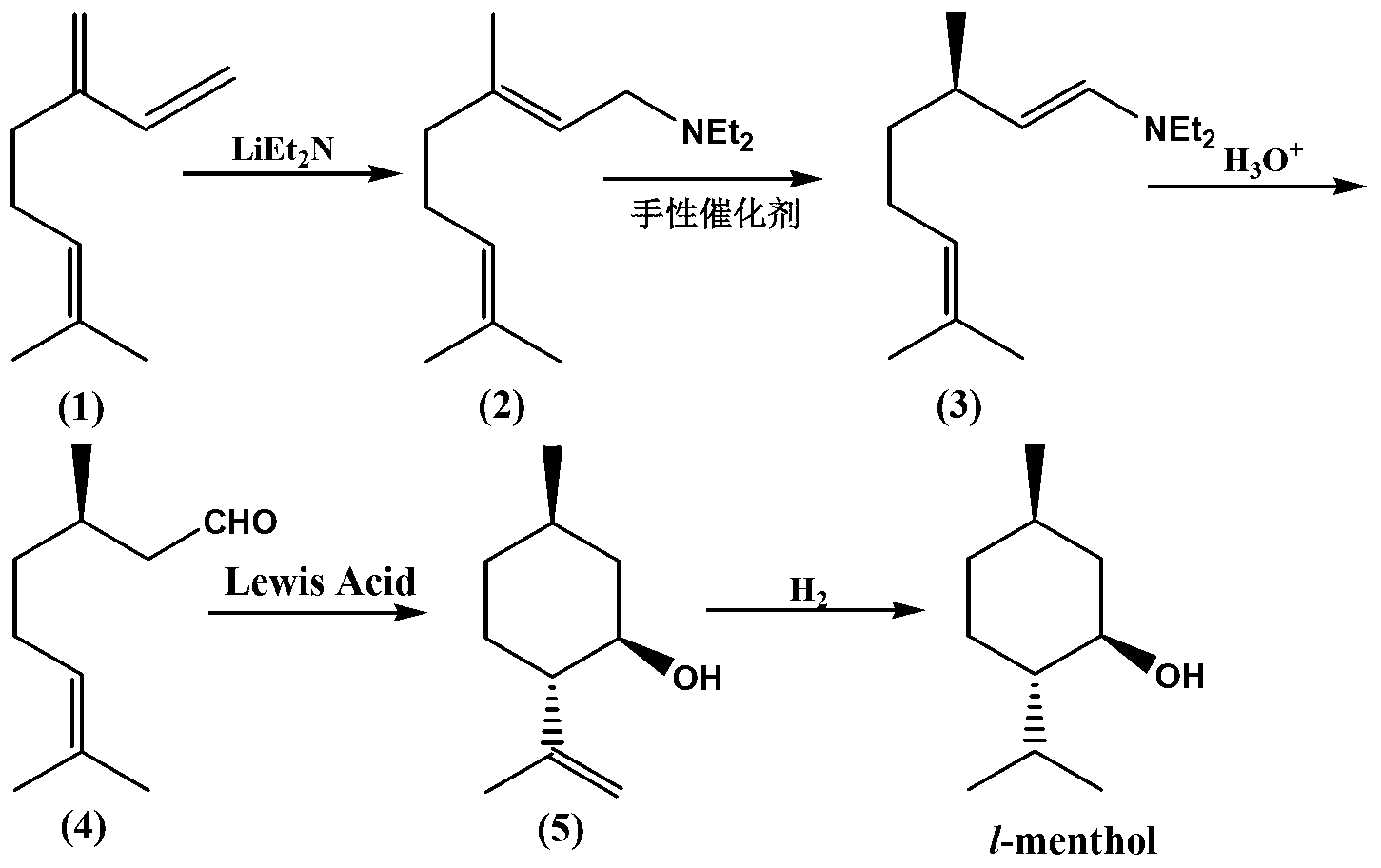
The invention belongs to the technical field of organic synthesis, in particular to a method for synthesizing L-menthol. The method for synthesizing L-menthol takes myrcene as an initial raw material and comprises the following steps: amination reaction: diethylamine and myrcene carry out amination reaction to obtain geranylamine; isomerization reaction: the geranylamine carries out isomerization reaction under the catalytic action of chiral catalyst to obtain intermediate enamine; hydrolysis reaction: the enamine carries out hydrolysis reaction in an acidic medium to produce intermediate D-citronellal; cyclization reaction: the D-citronellal generates L-isopulegol by lewis acid catalyst and organic solvent; and hydrogenation reaction: the L-isopulegol is hydrogenated to obtain L-menthol. The method for synthesizing L-menthol has the yield of more than 90 percent, the GC purity of higher than 99.0 percent and the specific rotation of -49 degrees to -50 degrees, the raw material myrcene has wide sources and lower cost, and the whole method has simple technology, good repeatability and high yield and is quite suitable for large-scale industrialized production.
Owner:SHANGHAI WANXIANG FLAVORS & FRAGRANCES
Preparation methods of glyoxylic acid L-menthyl alcohol ester and monohydrate of glyoxylic acid L-menthyl alcohol ester
ActiveCN102516078AEasy to operateMild reaction conditionsPreparation from carboxylic acid halidesFenchyl alcoholL menthol
The invention relates to preparation methods of glyoxylic acid L-menthyl alcohol ester and a monohydrate of the glyoxylic acid L-menthyl alcohol ester, and belongs to the technical field of fine chemical industry. The glyoxylic acid L-menthyl alcohol ester is prepared by the following steps: reacting L-menthol with monohalogen or dihalogen acetyl halide or anhydride to generate monohalogen or dihalogen acetic acid L-menthyl alcohol ester; and reacting in the presence of a pro-oxidant to obtain the glyoxylic acid L-menthyl alcohol ester. The monohydrate of the glyoxylic acid L-menthyl alcohol ester is prepared by the following steps: cooling and diluting a reaction solution using monohalogen acetic acid L-menthyl alcohol ester as a raw material; treating by using a dimethylsulfoxide (DMSO) solution of P2O5 and triethylamine to obtain the glyoxylic acid L-menthyl alcohol ester; washing, extracting and concentrating a reaction solution using dihalogen acetic acid L-menthyl alcohol ester as a raw material to obtain the glyoxylic acid L-menthyl alcohol ester; and treating by using sodium hydrogen sulfite and formaldehyde to obtain the monohydrate of the glyoxylic acid L-menthyl alcohol ester. The total yield of the glyoxylic acid L-menthyl alcohol ester synthesized by the technology can reach over 72 percent; the total yield of the monohydrate of the glyoxylic acid L-menthyl alcohol ester can reach over 80 percent; the purity can reach over 99.5 percent; the technical process is simple and convenient; the raw materials are available; the yield is high; the product purity is high; and the process is suitable for industrial production.
Owner:ZHEJIANG XIANFENG TECH +1
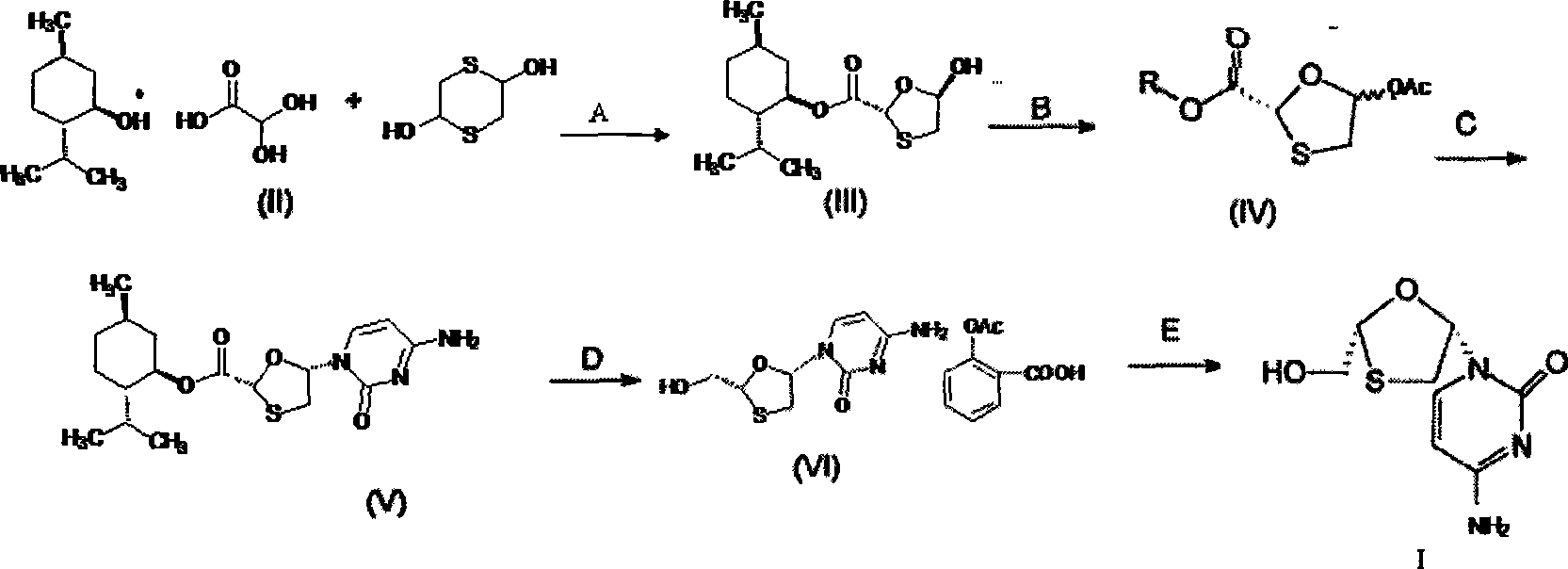
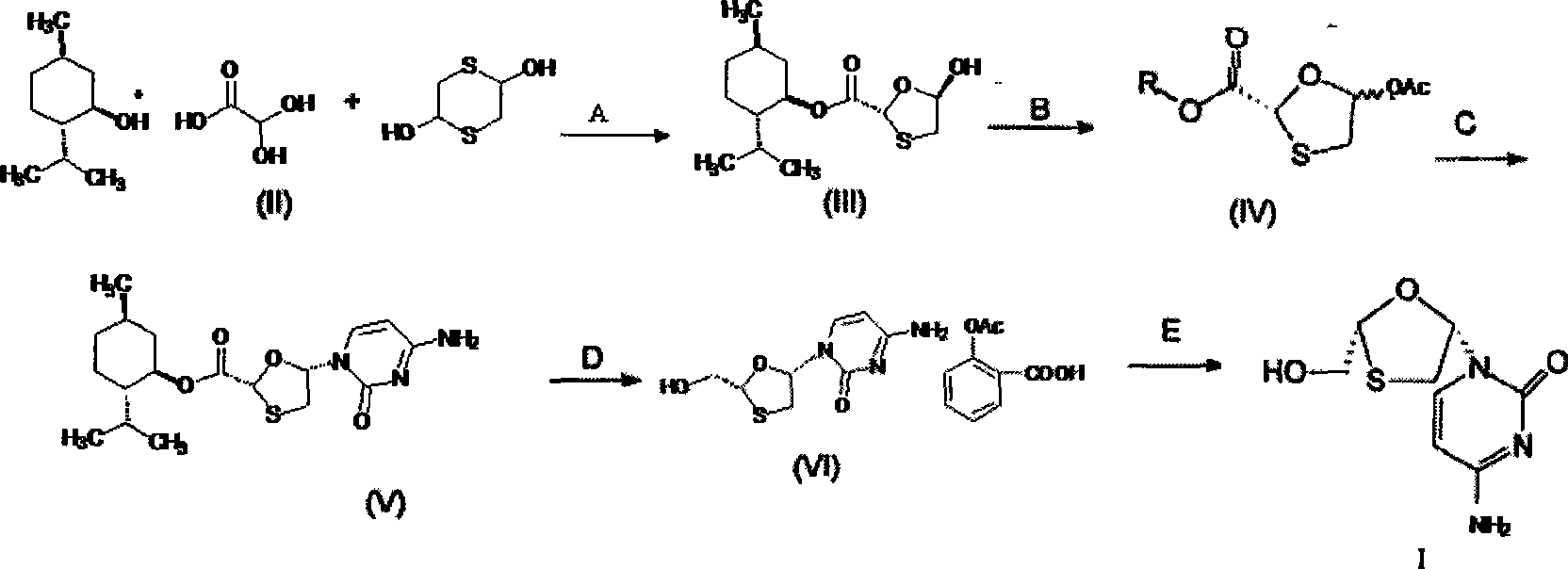
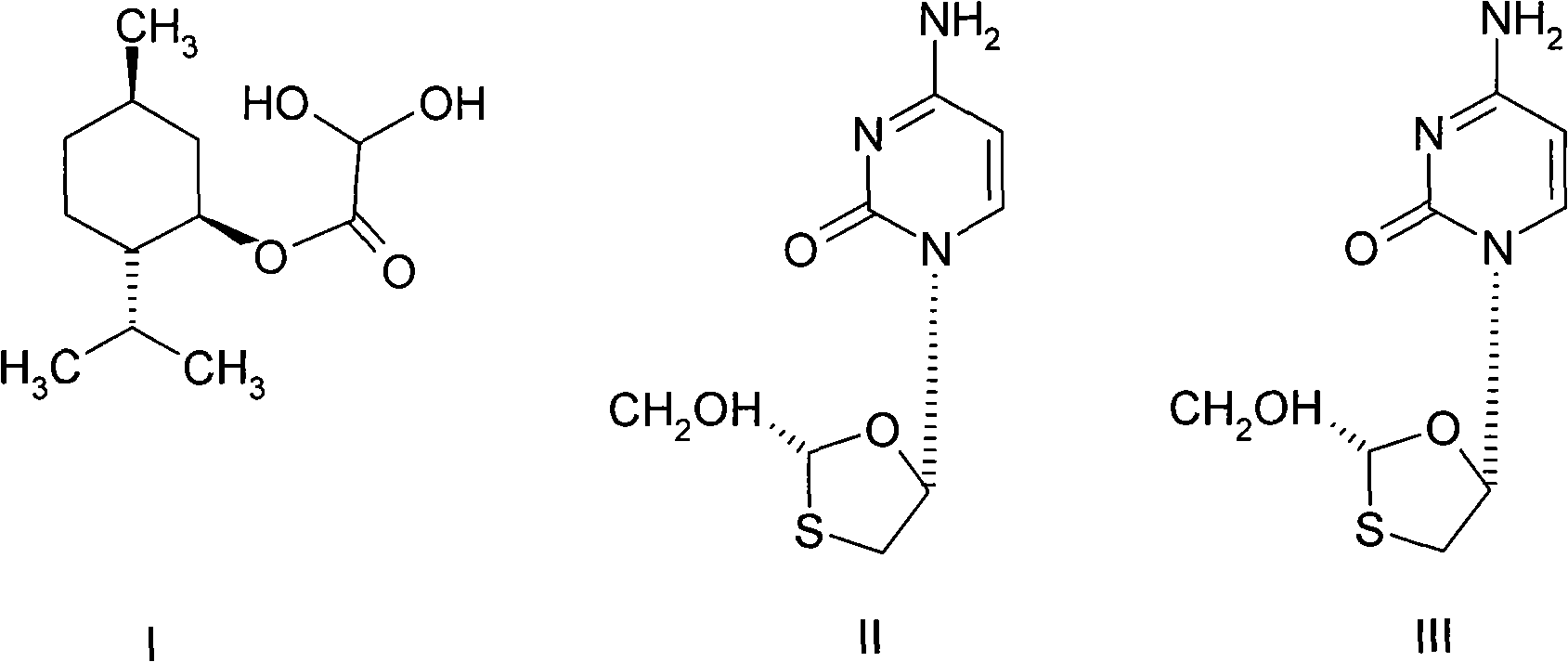
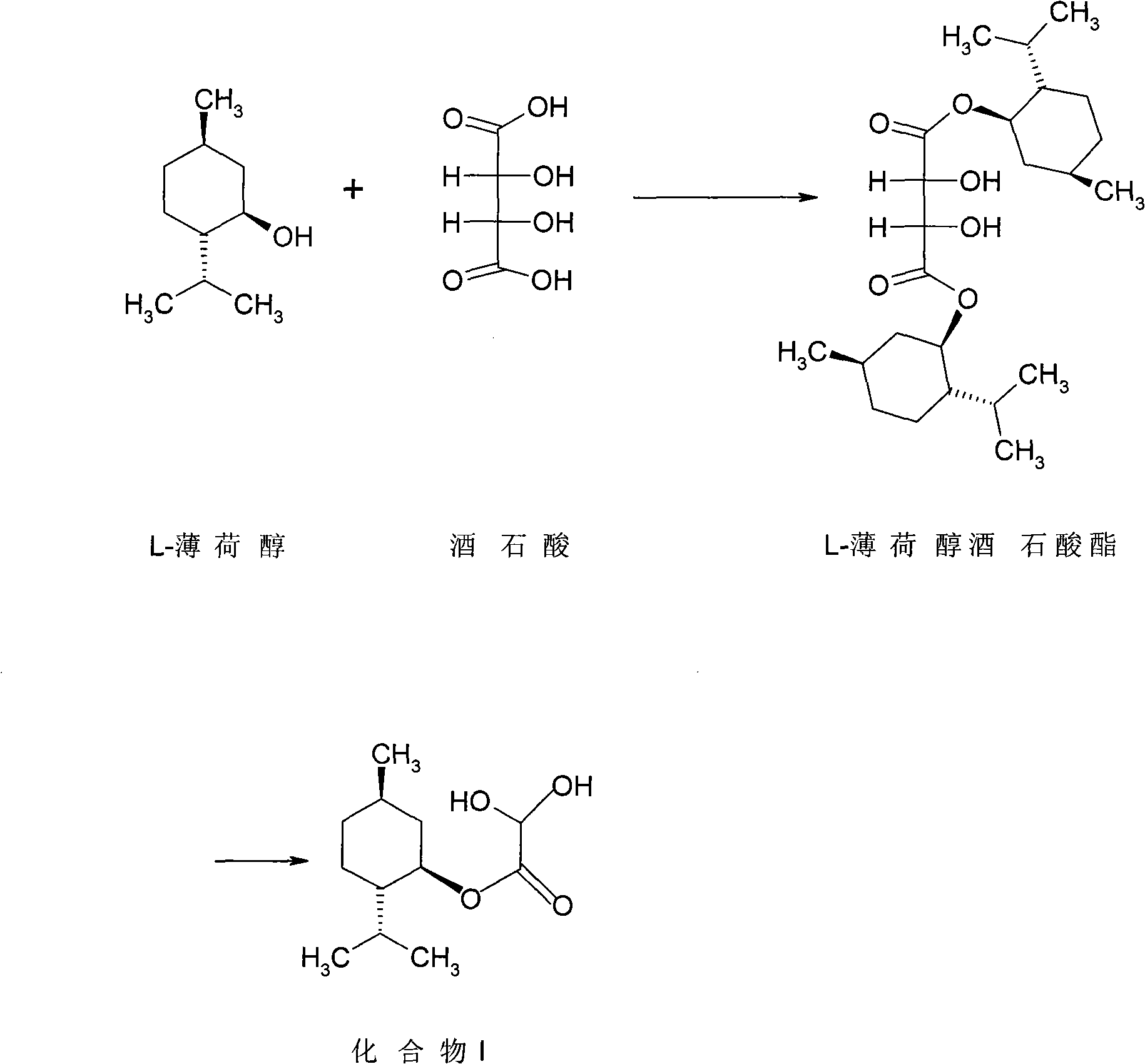
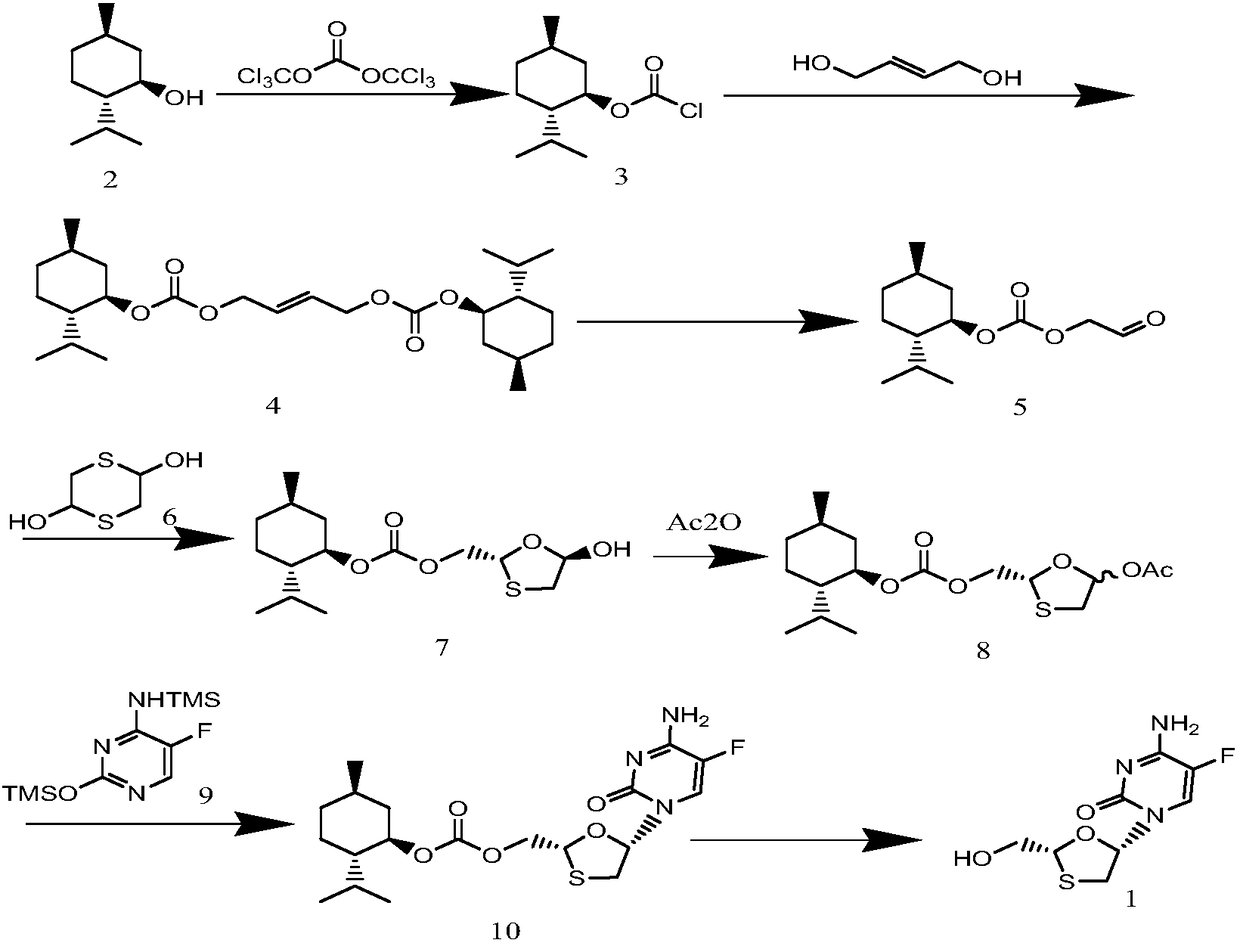
Lamivudine diastereoselective synthesis method
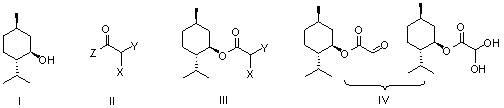
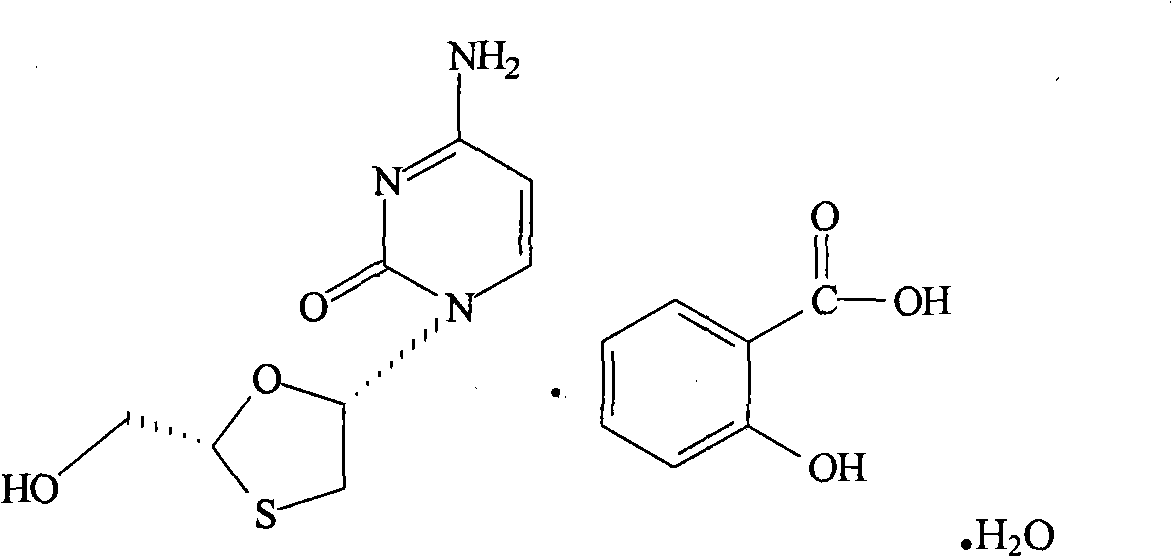
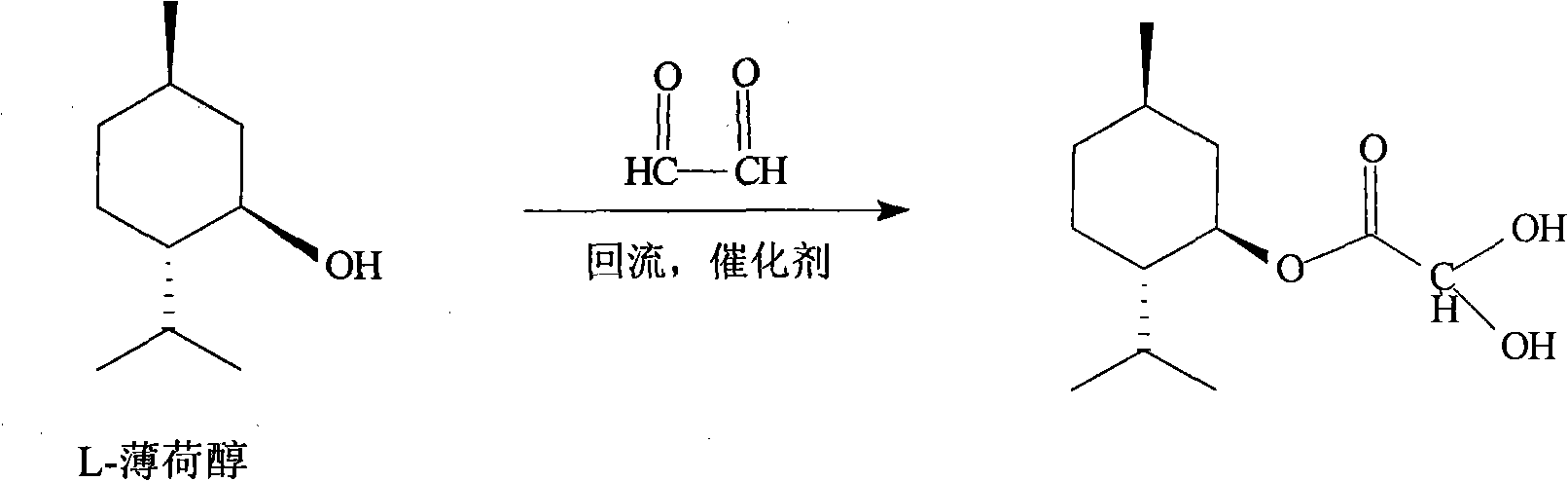
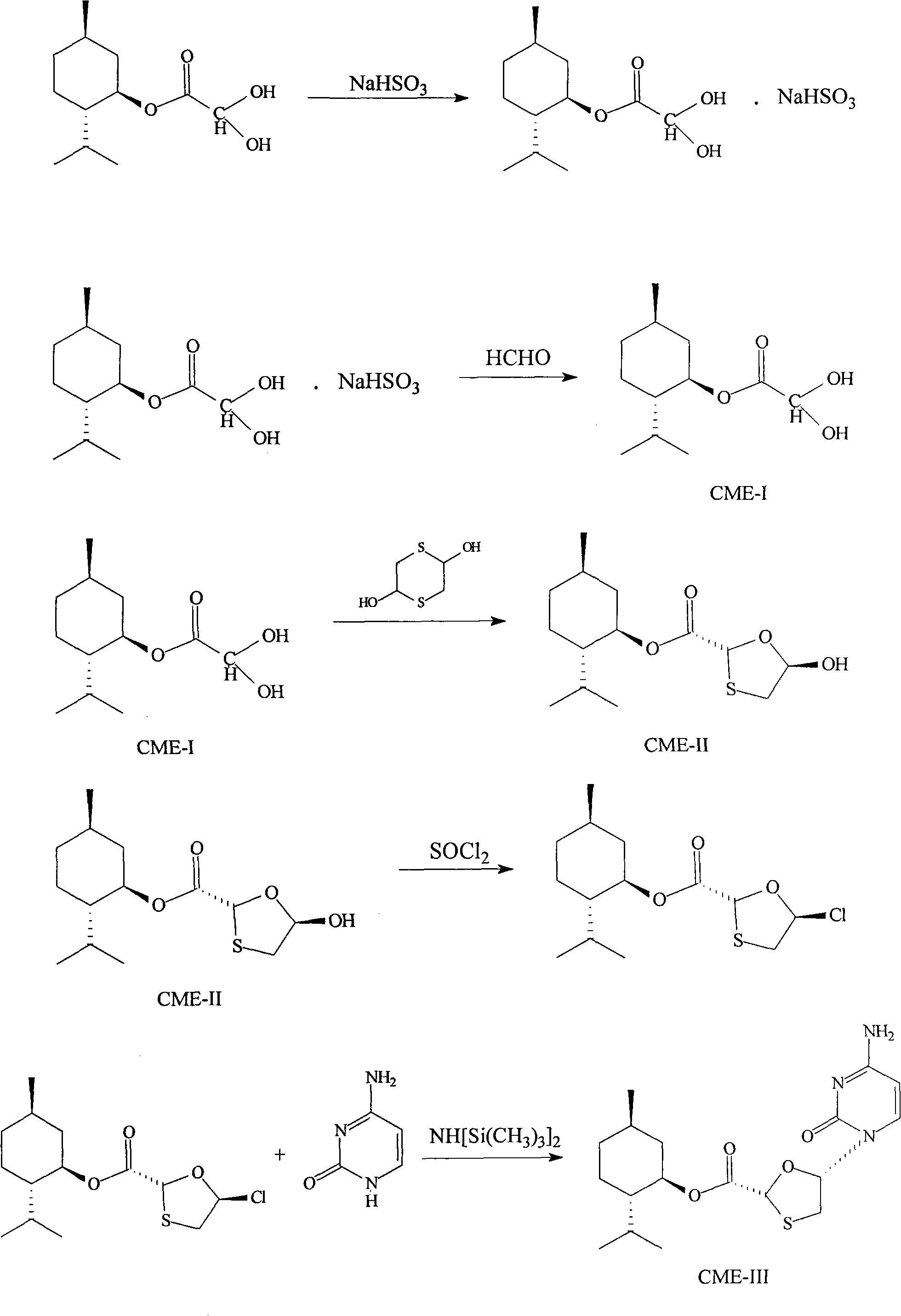
A lamivudine diastereoselective synthesis method, which takes chiral auxiliary agent L-menthol as the initial material, synthesizes trans-5-hydroxyl-1, 3-oxygen thiacyclopentane-2-carboxylic acid-(1'R, 2'S, 5'R) menthol ester under the action of concentrated sulfuric acid, choose triethanolamine to obtain trans-isomer trans-5-hydroxyl-1, 3-oxygen thiacyclopentane-2-carboxylic acid-(1'R, 2'S, 5'R) menthol ester, and let the trans-isomer to react with acylating agent to obtain trans-5-acetoxy-1, 3-oxygen thiacyclopentane-2-carboxylic acid-(1'R, 2'S, 5'R) menthol ester, glycosidate with cytosine under the action of alkali to obtain 5S-cytosine-1'-radical-1, 3-oxygen thiacyclopentane-2-carboxylic acid-(1'R, 2'S, 5'R) menthyl ester, and then deoxidize with a deoxidizer, and salifying with aspirin, to ionize and liberate lamivudine. Since triethanolamine is added as in the course of reaction interconverting agent, the yield of lamivudine is increased greatly. Aspirin is added in the course of reaction, so that the lamivudine forms an aspirin salt that has poor water solubility, and therefore can effectively separate and liberate lamivudine from the medium.
Owner:湖南千金湘江药业股份有限公司
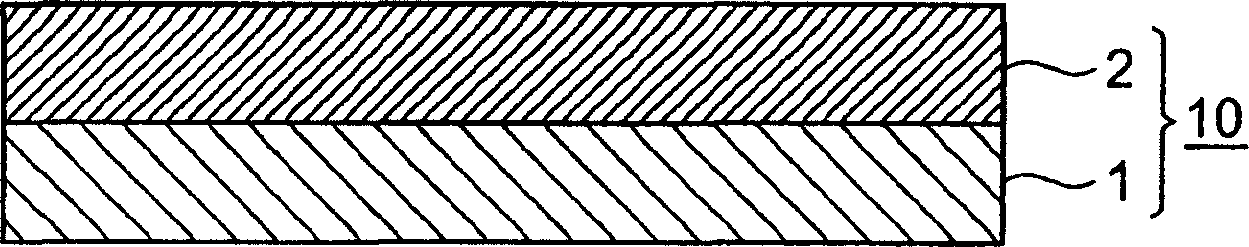
Warm poultice
Warm poultice (10) comprising support (2) and, disposed on at least one major surface of the support (2), pressure sensitive adhesive layer (1), the pressure sensitive adhesive layer (1) comprising a warm feeling imparting substance, L-menthol and, as a residual stimulation reducing agent, polyethylene glycol.
Owner:HISAMITSU PHARM CO INC
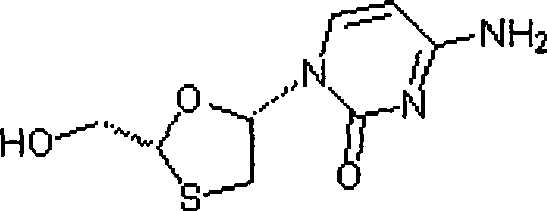
Synthesis and preparation process of lamivudine intermediate HDMS
InactiveCN101830893ALow costStep synthesis yield increaseOrganic chemistryOrganic synthesisGas phase
The invention discloses a synthesis and preparation process of a lamivudine intermediate HDMS, which belongs to the field of organic synthetic medicaments. The synthesis and preparation process is characterized in that a preparation method of glyoxylic acid menthol ester comprises the following steps of: under the catalysis of concentrated sulfuric acid, performing a reflux reaction on L-menthol and glyoxylic acid, wherein a reactive solvent is cyclohexane; judging the reaction end point through a gas phase chromatography, and then cooling and washing the mixture; reacting an organic layer with solution of sodium bisulfite at the temperature of between 20 and 30 DEG C; judging the reaction end point through the gas phase chromatography, and then washing the mixture by using the cyclohexane; reacting an aqueous layer with methanol at the temperature of between 20 and 30 DEG C; and washing the mixture after filtering and drying the mixture to obtain the glyoxylic acid menthol ester. By using the synthesis and preparation process, the condition is milder, the operation is simpler and more convenient and the reaction processes are easier to control, so the product production yield and the quality stability are improved, and the cost of the lamivudine intermediate HDMS is greatly reduced.
Owner:ANHUI PROVINCE YIFAN SPICE
Method for preparing L-menthol
InactiveCN103086845AAvoid purification difficultiesAvoid Yield ProblemsPreparation by isomerisationMolecular sieve catalystsZinc bromideMolecular sieve
The invention discloses a method for preparing L-menthol, which comprises the following steps:(1) reacting d-citronellal in a solvent in the presence of a catalyst, and collecting isopulegol from the reaction product, wherein the catalyst is a zinc-bromide-modified NaY molecular sieve; and carrying out high-pressure hydrogenation on the obtained isopulegol to obtain the target product L-menthol. The obtained isopulegol can be subjected to high-pressure hydrogenation to obtain the target product L-menthol without purification. The invention has the characteristics of mild reaction conditions, high stereoselectivity, high yield and the like, and is simple to operate; and the catalyst is simple to recover, and can be used repeatedly. The method disclosed by the invention avoids the problems of difficulty in product purification, low yield and the like in the traditional synthesis technique of the compounds, and greatly lowers the production cost.
Owner:上海统益生物科技有限公司
Method for preparing L-menthol glyoxylic ester monohydrate with solid acid as catalyst
InactiveCN101274892AEasy to separateEmission reductionPhysical/chemical process catalystsOrganic compound preparationSolid acidSodium bisulfate
The invention discloses a method for preparing the hydrate of L-menthol glyoxylic ester by taking a solid acid as a catalyst. The preparation method takes cyclohexane as a water-carrying agent; a reaction liquid A is obtained by the reflux heating and the water diversion of L-menthol and glyoxylic acid aqueous solution under the catalysis of the solid acid and filtered to remove the solid acid, and an organic phase obtained from separation adjusts pH to 4.9 to 5.1; a sodium bisulfate aqueous solution is added to fully react with the A and the obtained reaction liquid B is separated to obtain a water phase to adjust the pH to 7.1 to 7.5; formaldehyde aqueous solution is added and hydrolysis is carried out to obtain the hydrate of L-menthol glyoxylic ester. The method of the invention takes the solid acid as the catalyst, which is easy to be operated, adopts the catalyst which is easy to be separated and can be recycled, thus reducing waste acid emission during the production process, controlling the formation of side products better and having the advantages of high yield and high product purity, etc.
Owner:浙江教育学院 +2
Composite plant extract and application thereof in storage and mildew prevention of food grains
ActiveCN108029762ANo pollution in the processSimple production processEdible seed preservationNatural extract food ingredientsSnow moldEugenol
The invention discloses a composite plant extract and an application thereof in storage and mildew prevention of food grains. The composite plant extract comprises effective components of cinnamic aldehyde, citral, eugenol and mint oil (containing 33% of L-menthol). One-year corn mildew prevention experiment proves that the composite plant extract disclosed by the invention has obvious restrainingeffects on the growth of moulds of aspergillus flavus, fusarium and the like, and the production of fungus toxins, hinders and controls the quality of corn from becoming bad, and also has the effectof maintaining the physiological activity of the corn, and a high-efficient good-quality product is provided for mildew prevention, quality guaranteeing and safe storage of the corn.
Owner:INST OF AGRO FOOD SCI & TECH CHINESE ACADEMY OF AGRI SCI
Oral cavity care solution
InactiveCN104042780AInhibit manufacturingNo side effectsCosmetic preparationsToilet preparationsSodium bicarbonateVitamin C
The invention discloses an oral cavity care solution which comprises the following components in percentage by weight: 10%-12% of plant extracts, 2%-3% of sodium bicarbonate, 1%-2% of vitamin C, 1%-2% of L-menthol and the balance of water, wherein the total content of the components is 100%. According to the oral cavity care solution, the plant extracts are adopted, the integral components form alkalescence, so that the PH value of a product is approach to that of an oral ecosystem; lentinus edodes, green tea and honeysuckle flowers are all edible, especially, the lentinan contained in the lentinus edodes can inhibit the bacteria inside a mouth from making dental plaque; the effects of the sterilization, inflammation elimination, halitosis removal and the like are achieved according to the natural mechanism effects of disinfection, sterilization, trichomonad resistance and convergence of the sodium bicarbonate.
Owner:王玉果
Adhesive and plaster
InactiveUS20060263420A1Reliably exhibitPromote percutaneous absorptionBiocideSurgical adhesivesElastomerAdhesive
An adhesive comprising a base with tackiness, an oil and ketoprofen, wherein the adhesive contains no L-menthol, the base is composed at least of a tacky composition comprising an elastomer and a tackifier and / or a tacky polymer containing an unsaturated monomer with a total of 5 or more carbon atoms as the monomer unit, the oil is an oil that is compatible with the tacky composition and the tacky polymer, and the oil content is 150-175 parts by weight with respect to 100 parts by weight as the total of the tacky composition and the tacky polymer.
Owner:HISAMITSU PHARM CO INC
Formula of mint fragrant tobacco additive essence
ActiveCN102204717AGreat tasteAdd natural sweetnessTobacco treatmentEssential-oils/perfumesNicotiana tabacumEugenol
The invention discloses a formula of mint fragrant tobacco additive essence. The formula of the mint fragrant tobacco additive essence consists of the following components in percentage by mass: 1 to 5 percent of L-menthol, 1 to 5 percent of ethyl maltol, 1 to 10 percent of isoamyl isovalerate, 1 to 5 percent of coke fragrant furanone, 5 to 15 percent of ethyl berry ketone, 1 to 5 percent of isophorone, 0.1 to 0.2 percent of phenylethyl isovalerate, 1 to 5 percent of eugenol type basil oil, 1 to 5 percent of ethyl phenoxyisobutyrate, 0.1 to 2 percent of 2-methyl ethyl butyrate, 15 to 25 percent of honey extract, 5 to 15 percent of glycerol, 5 to 15 percent of edible ethanol and 20 to 40 percent of propylene glycol. Because the menthol is added in the formula of the mint fragrant tobacco additive essence, the tobacco can generate cool and refreshing smell of mint, the tobacco fragrance can be improved, the smoking taste is corrected, the smoking taste of the tobacco is finally improved, natural sweet fragrance of the tobacco is increased, the dry, astringent and bitter mouthfeel of customers is improved, and the requirements of the customers are met.
Owner:GUANGZHOU AOJIAN PERFUME
Preparation method of a variety specifications of dementholized peppermint oil and menthol by combined azeotropic distillation and vacuum distillation technology
ActiveCN108046993AReduce lossesHigh purityOrganic compound preparationHydroxy compound preparationWater vaporPeppermints
The invention provides a preparation method of a variety specifications of dementholized peppermint oil and menthol by combined azeotropic distillation and vacuum distillation technology. The method comprises the following steps that (1), azeotropic distillation to remove front distillate: peppermint trichomes oil is taken, after azeotropic agent water is added, azeotropic distillation is carriedout, front distillate is separated and detected, when olefin content in the front distillate is less than 1%, the azeotropic distillation is stopped; oil and water separation is carried out, oil phasewaits for treatment; (2), vacuum distillation to obtain post distillate: the above front distillate removed oil phase is subjected to vacuum distillation to obtain post distillate, the post distillate is detected, when the content of L-menthol is more than 50% in the distillate, the vacuum distillation is stopped; (3), atmospheric vapor distillation. The method reduces the loss of the front distillate, at the same time, the aroma of the front distillate can withstand 150 DEG C without being damaged by high temperature, the quality of dementholized peppermint oil is well guaranteed. And a variety specifications of dementholized peppermint oil can be prepared by the method, at the same time, the high content of menthol in the redundant post distillate can also be applied to the physical crystallization process to produce high purity natural menthol crystals.
Owner:ANHUI FENGLE PERFUME
Preparation method of L-menthol
InactiveCN103468779AImprove single synthesis efficiencyReduce energy consumptionMicroorganism based processesFermentationCyclodextrinHigh pressure
The invention discloses a preparation method of L-menthol. The preparation method comprises the following steps: 1) adding 3-methyl-6-isopropyl-phenol and a composite noble metal catalyst to a high-pressure kettle, wherein the composite noble metal catalyst is formed by the combination of a Raney nickel catalyst RTH211, palladium carbon, cyclodextrin and Cu / SiO2; closing the high-pressure kettle; filling hydrogen to replace the air in the kettle for two to three times; finally filling hydrogen to enable pressure in the kettle to be 5.0 to 8.0MPa; controlling the temperature to be 100 to 150 DEG C for 4 to 6 hours, thus obtaining a mixture of menthol; rectifying to obtain D,L-menthol; 2) performing esterification reaction on D,L-menthol and an acylation reagent under the effect of alkali so as to obtain D,L-menthol ester; 3) performing hydrolysis reaction on D,L-menthol ester under the effect of hydrolytic enzyme, thus obtaining L-menthol, wherein the hydrolytic enzyme is cellularized burkholderia cepacia (B.cepacia ATCC25416). The preparation method has the advantages that the production raw materials are simple and easy to obtain; the prepared L-menthol is high in yield and lower in cost and suitable for mass production and use.
Owner:阜阳市百富安香料有限公司
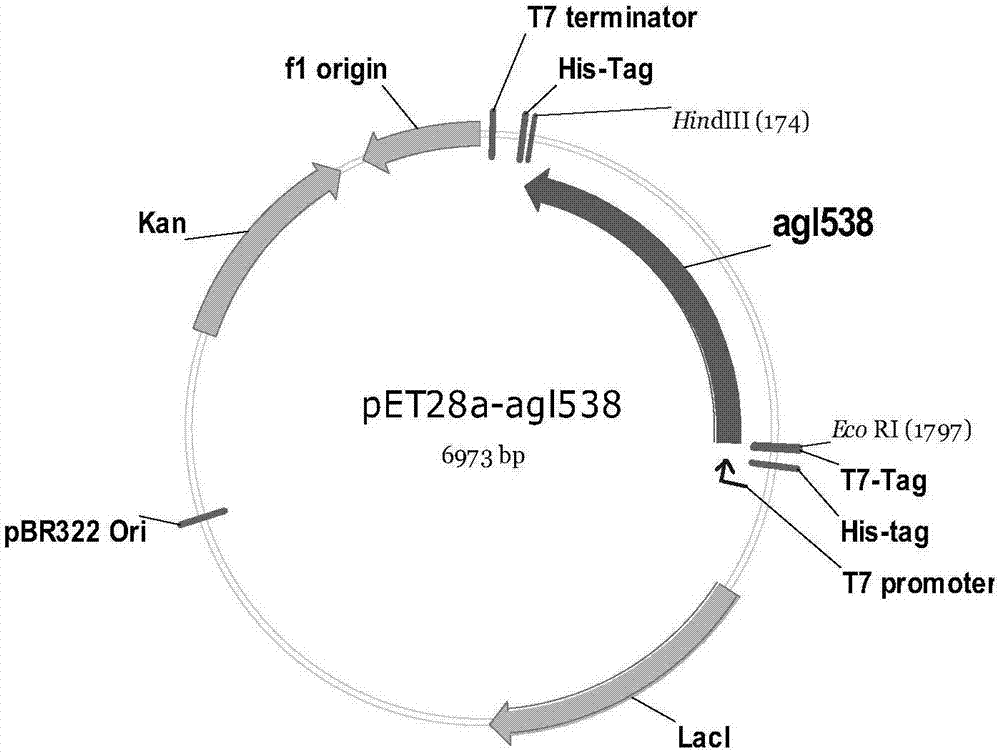
Recombinant escherichia coli containing alpha-glucosidase gene and application of recombinant escherichia coli
ActiveCN107400654AEfficient productionHigh yieldBacteriaMicroorganism based processesEscherichia coliAlglucerase
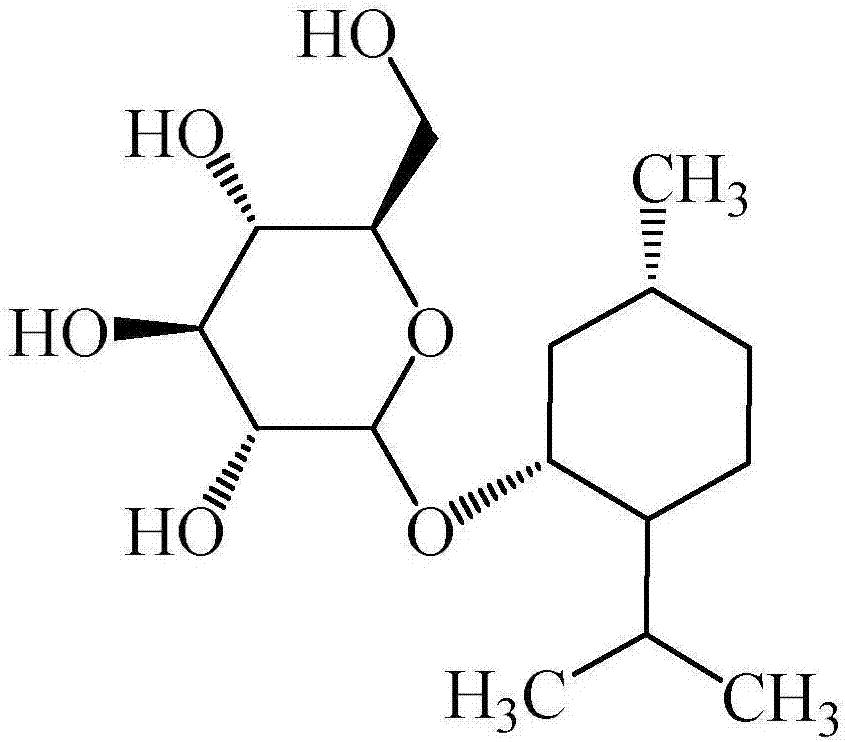
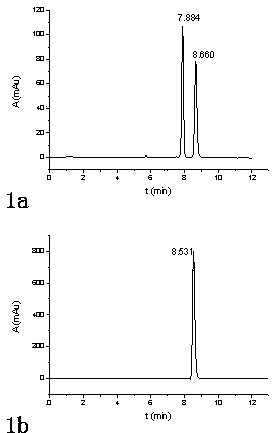
The invention discloses recombinant escherichia coli containing an alpha-glucosidase gene and application of the recombinant escherichia coli. The recombinant escherichia coli is obtained by transferring the alpha-glucosidase gene as shown in SEQ ID NO.1 into an escherichia coli host cell. The recombinant escherichia coli containing the alpha-glucosidase gene for producing L-menthol-alpha-glucoside can efficiently synthesize alpha-glucosidase in the cell; L-menthol is taken as a substrate, and maltose is taken as an auxiliary substrate to efficiently catalyze glycosylation reaction of L-menthol, the reaction is performed for 10-24 hours to obtain L-menthol-alpha-glucoside converted mash with concentration greater than 10%, wherein average production strength is greater than 5g.L<-1>.h<-1>, and a substrate conversion rate is greater than 95%; and the L-menthol-alpha-glucoside product is high in concentration, is high in conversion rate, and is beneficial for recycling and purifying L-menthol-alpha-glucoside.
Owner:深圳杉海创新技术有限公司
Method for splitting DL-menthol by means of pre-column derivatization high performance liquid chromatography
InactiveCN103910607ASplit validMeet separation requirementsOrganic compound preparationHydroxy compound preparationAlcoholUltraviolet absorption
The invention discloses a method for splitting DL-menthol by means of pre-column derivatization high performance liquid chromatography. The method comprises the following steps: firstly, carrying out a precolumn derivatization esterification reaction on DL-menthol and aromatic acid to obtain corresponding ultraviolet absorption DL-mint alcohol; then, preparing the DL-mint alcohol ester to a 0.2-1.0mg / mL sample solution by adopting an opposite mobile phase system formed by an aqueous phase and an organic phase by using an amylase chiral chromatographic column through the high performance liquid chromatograph; controlling the flow rate at 0.4-1.0mL / min, the sample size at 2-10 microlitre, the detecting wave length at 220-280nm and the temperature of the wave length at 20-35 DEG C for chromatographic column separation so as to separate the DL-mint alcohol ester; and finally, respectively hydrolyzing to obtain D-menthol and L-menthol. The method for detecting and splitting menthol is convenient, practical and high in sensitivity.
Owner:SHANGHAI INST OF TECH
Method for preparing optical pure L-menthol
ActiveCN108083980AControlling Optical PurityHigh optical purityOrganic compound preparationOrganic-compounds/hydrides/coordination-complexes catalystsL mentholPhosphine
The invention provides a method for preparing optical pure L-menthol. Under the action of a transition metal catalyst, L-isopulegol in isopulegol is selectively hydrogenated, and the optical pure L-menthol is prepared. The transition metal catalyst comprises a transition metal compound and a chiral phosphine ligand. High stereo-selective synthesis of the L-menthol is realized, and the optical purity of a product can reach up to 99ee%.
Owner:WANHUA CHEM GRP CO LTD +1
Method for preparing emtricitabine
ActiveCN109438432AStarting materials are cheap and readily availableMild reaction conditionsOrganic chemistry methodsEmtricitabineL menthol
The invention discloses a method for preparing emtricitabine. The method comprises the following steps: refining so as to obtain 5S-(5'-flucytosine-1')-1,3-oxythiacyclopentane-2-carbethoxy-(1'R,2'S,3'R)-menthyl lactate; under a weak alkali and solvent condition, removing a chiral aid L-menthol, thereby obtaining a product of emtricitabine. The initial raw material used in the method is low in price, easy to obtain, mild in reaction condition, high in atom utilization rate and simple and convenient in operation process, the reagent is environmentally friendly, the obtained product is high in chemical purity, meets medicine standards, and is applicable to industrial production of emtricitabine.
Owner:WUHAN INSTITUTE OF TECHNOLOGY
Method for synthesizing L-menthy chloroformate
InactiveCN1616405ALow requirements for production equipmentEasy to transportPreparation from phosgene or haloformatesHexahydropyridineRoom temperature
The present invention discloses a kind of L-menthyl chloroformate synthesizing process. Under room temperature and in the presence of catalyst, L-menthol and triphosgene are chloroacylated to obtain L-menthyl chloroformate. The process has reaction temperature of 25 deg.c, reaction time of 10-12 hr and catalyst pyridine, triethylamine or hexahydropyridine. The present invention has the advantages of substituting phosgene with less toxic solid triphosgene, easy control of the reaction, and relatively high safety and environment friendship.
Owner:上海香料研究所
Preparation of chiral covalent organic framework material having L-menthol as chiral source
ActiveCN109134875AHigh efficiency of chiral resolutionReduce energy consumptionOther chemical processesL mentholTwo step
The invention discloses a preparation method of a hyperbranched polymer having (s)-(-)-alpha-phenethylamine as a chiral source. Thionyl chloride, 3,5-dibromobenzoic acid and (S)-(-)-alpha-phenethylamine are subjected as materials to two-step organic synthetic reaction to obtain white needle crystal A; 1,3,5-tribromophenyl, Pd(PPh3)2Cl2, CuI, PPh3 and ethynyl trimethylsilane are subjected as raw materials to two-step reaction to synthesize compound B; the white needle crystal A, the compound B, Pd(PPh3)2Cl2, PPh3 and CuI are subjected as materials to heating via a microwave reactor by using triethylamine and dimethylformamide as solvents, and stirring is carried out for 17 min; the crude product is purified and dried to obtain yellow solid; the finished product is tested. The novel nano-level hyperbranched polymer herein can provide massive catalytic active sites containing chiral groups in asymmetric catalytic reaction, and asymmetric synthetic reactive catalytic efficiency is improved.
Owner:QIQIHAR UNIVERSITY
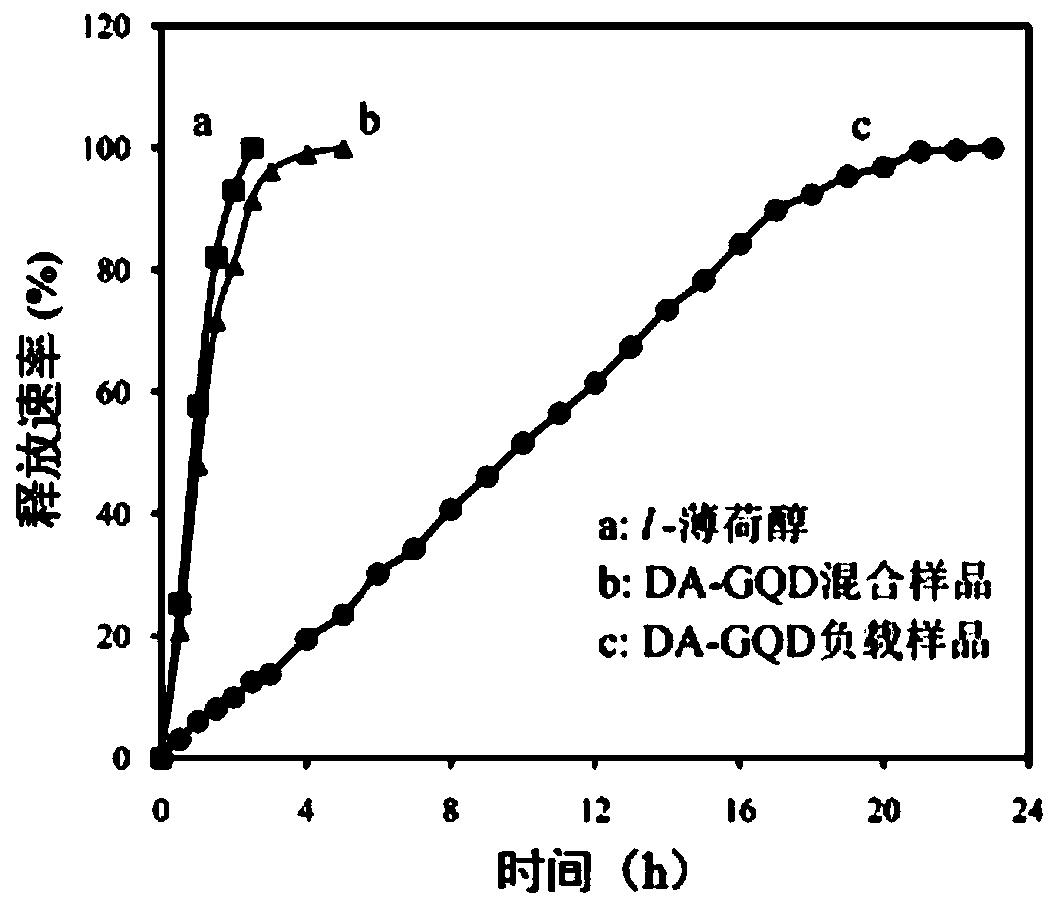
L-menthol slow-release material based on graphene quantum dots and preparation method thereof
ActiveCN110200821ASmall particle sizeGood dispersionCosmetic preparationsTobacco preparationPickering emulsionGraphite
The invention discloses an L-menthol slow-release material based on graphene quantum dots and a preparation method thereof, which belong to the field of slow release materials. The method utilizes functionalized graphene quantum dots as a particle emulsifier, and the functionalized graphene quantum dots are dispersed at a menthol / water interface to form a Pickering emulsion; the L-menthol is covered by small graphite sheets, and L-menthol supported by the sustained-release material obtained after cooling and crystallization has good sustained-release properties. The method of the invention hasthe advantages of simple process, effective and high loading of L-menthol can be realized, and effective sustained release is realized; the method achieves low cost and industrial production, and theobtained L-menthol sustained-release material can be used in the fields of food, tobacco, daily chemical and medicine.
Owner:JIANGNAN UNIV
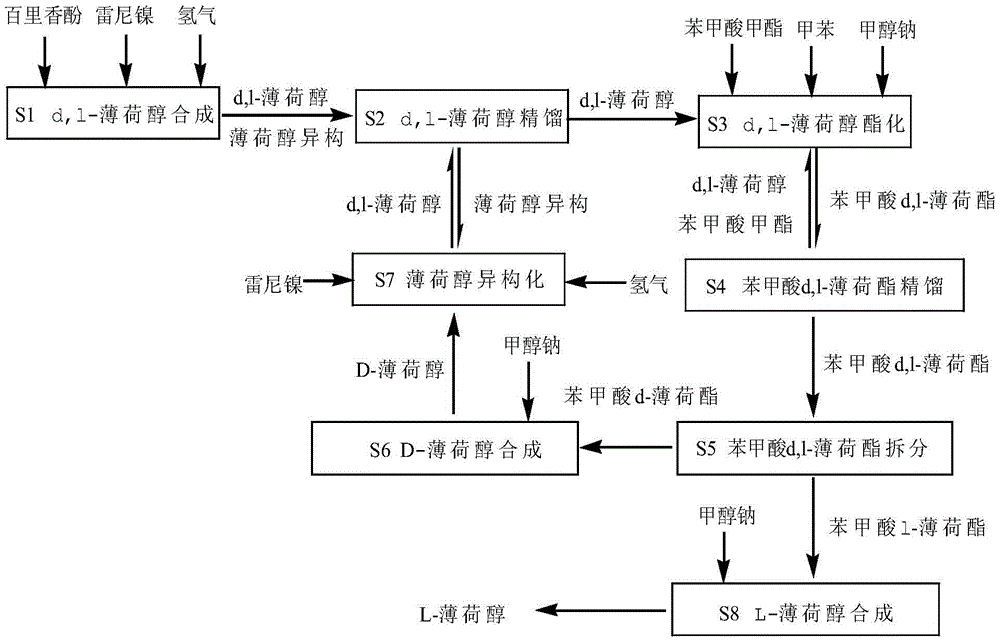
A synthetic process of L-menthol
ActiveCN105461516AHigh yieldLow costOrganic compound preparationPreparation by hydrogenationBenzoic acidIsomerization
The invention relates to the field of spice synthesis and particularly relates to a synthetic process of L-menthol. The process includes steps of d,l-menthol synthesizing, d,l-menthol rectification, d,l-menthol esterification, d,l-menthyl benzoate rectification, d,l-menthyl benzoate resolution, D-menthol synthesizing, menthol isomerization and L-menthol synthesizing. The process adopts thymol that is a simple, easily available and cheap chemical product as a raw material. Esterification conditions are optimized and the esterification and rectification are performed at the same time so as to allow the esterification to be converted into a way beneficial to d,l-menthyl benzoate production, thus increasing the esterification yield. Crystallization and resolution are optimized by utilization of the d,l-menthyl benzoate. Preparation of the L-menthol by the process is characterized by being high in yield, low in cost, simple and convenient in operation, suitable for continuous and large-scale production, and the like. According to the process, operation of the process is cyclic with a whole system being sealed, and the process is free of waste water, energy-saving and environmental friendly.
Owner:ANHUI HAIHUA CHEM
Preparation method of lamivudine
ActiveCN109438431ARaw materials are cheap and easy to getShort stepsOrganic chemistryL mentholSolvent
The invention discloses a preparation method of lamivudine. The method comprises the steps of obtaining pure 5S-(cytosinyl-1')-1,3-oxathiolane-2-ethoxycarbonyl-(1'R,2'S,3'R)-menthyl ester through refining; removing chiral aid L-menthol in the presence of a weak base and a solvent to obtain a lamivudine product. The raw materials are cheap and readily available, the reagents used are environmentally friendly, the steps are short, the reaction conditions are mild, the atomic utilization rate is high, the yield is high, the obtained product has high chemical purity and meets the medicinal standards, and the preparation method is suitable for large-scale production of lamivudine.
Owner:WUHAN INSTITUTE OF TECHNOLOGY
Superhigh-pressure crystallization separation method of L-menthol from peppermint oil
ActiveCN103274902ACrystalline Energy Saving and High EfficiencyControl crystallization temperatureHydroxy compound separation/purificationFiltrationPeppermints
A superhigh-pressure crystallization separation method of L-menthol from peppermint oil is provided. In the method, the peppermint oil is used as a raw material, and the method comprises crystallization and separation. The method is characterized in that: a pressure of 100 to 700MPa is applied to the peppermint oil in a superhigh-pressure vessel, and is kept for 0.25-2 hours at a temperature below 42 DEG C; ice-sand like mixed crystals of the L-menthol are obtained after pressure relief; L-menthol crystals are obtained through squeezing filtration of the mixed crystals. The L-menthol purity in the crystals is up to 99.5%. Through use of the method, selective crystallization, according to L-menthol content in the peppermint oil, and different requirements of L-menthol inventory level in a mother liquid of the peppermint oil, can be realized.
Owner:潘见
Synthesis method for d, l-menthol and stereoisomers thereof
InactiveCN103058825ALow priceEasy to makePreparation by hydrogenationRaney catalystsSynthesis methodsReaction temperature
The invention discloses a synthesis method for d, l-menthol and stereoisomers thereof. The synthesis method comprises: a compound Raney nickel is used as a catalyst for the catalytic hydrogenation of compounds containing at least one double bond, connecting to an oxygen atom at 3 - position, and having a menthane carbon skeleton, such as thymol, and d, l-menthol and / or a mixture of stereoisomers thereof; and after the reaction is completed, d, l-menthol, d, l-neomenthol and the d, l-iso-menthol are separately obtained via vacuum distillation in a packed tower with a number of theoretical plates of 60-100; wherein the reaction is carried out under conditions of a pressure being 0.1-3.5MPa and a temperature being 50-200 DEG C. In the whole reaction process of the present invention, water or an organic solvent are not used, the catalyst can be reused, and by-products of hydrogenolysis of hydroxyl in menthol is rarely produced, thereby preventing the generation of hydrocarbons which can not be repeatedly used. The hydrogenation reaction is carried out under low pressure, the reaction temperature is low, so requirement for the reaction equipment is not high; and the operating conditions is mild, thereby reducing the overall cost, and making the whole reaction process more environmental and safe. The thymol conversion rate is of 99.99%, and the yield is greater than 99%.
Owner:江苏宏邦化工科技有限公司
Paeonol solid dispersion air freshener
The invention relates to a paeonol solid dispersion air freshener. The air freshener is prepared by a solvent volatilization method or melting method and comprises the following components in part by mass: 1 to 40 parts of paeonol, 0 to 5 parts of L-menthol, 1 to 90 parts of inorganic carrier material, and 0 to 1 part of beta-cyclodextrin included essence and spice, wherein the inorganic carrier material is talcpowder, diatomite, bentonite or attapulgite and the like with high specific surface area. The air freshener is used for freshening and sterilizing air in families and automobiles and can also be used for the mold prevention of textiles, paper and the like.
Owner:ANHUI RONGJING FENGDAN BIOTECH +1
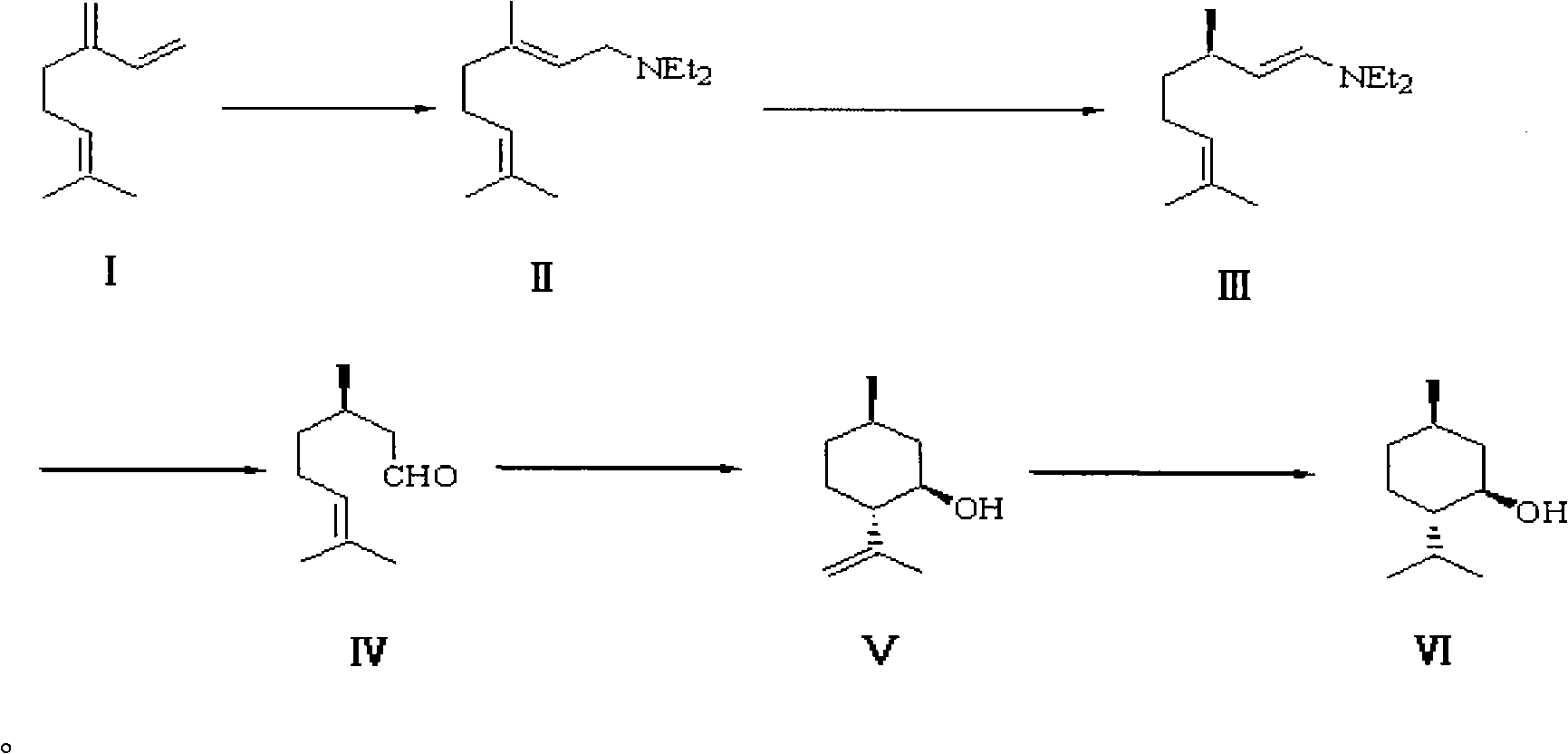
Method for preparing L-menthol intermediate d-citronellal
ActiveCN103254047AEasy to recycleAvoid recycling difficultiesAmino preparation from aminesCarbonyl compound preparation by hydrolysisL mentholSolvent
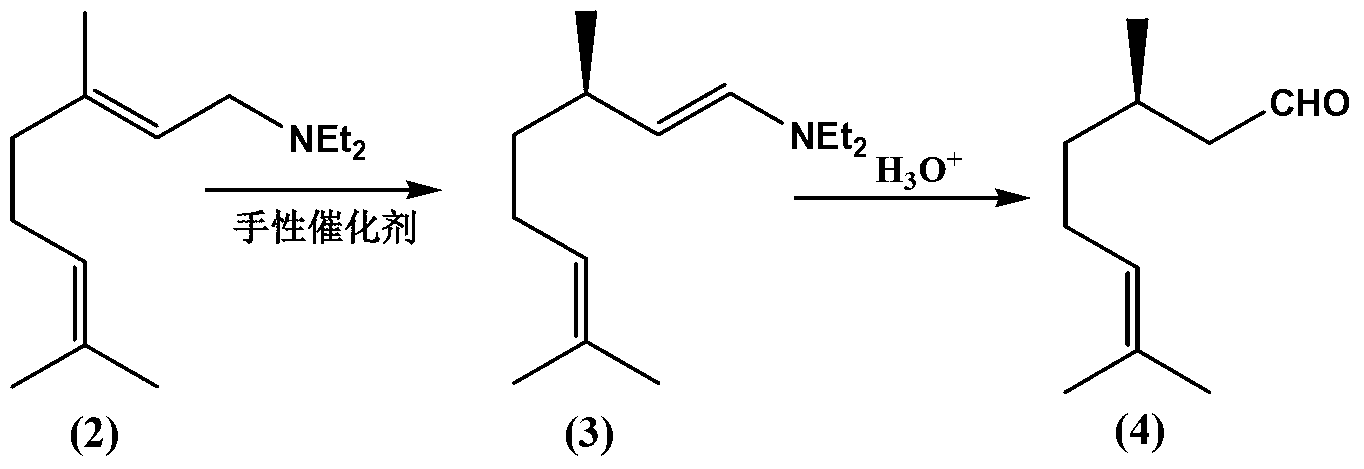
The invention discloses a method for preparing L-menthol intermediate d-citronellal. The method comprises the following steps of: (1) enabling geranyl amine (2) to react in a solvent A in the existence of a chiral catalyst, and then collecting myrcene amine (3) from a reaction product; and (2) carrying out acidic hydrolysis on the myrcene amine (3) to obtain d-citronellal (4). The structure of the chiral catalyst is as follows: [Rh(PS-Binap)2]<+>Y<->. The method has the characteristics of being mild in reaction condition, simple in operation, high in stereoselectivity, high in yield, simple in catalyst recovery and the like, and can be applied mechanically in cycle.
Owner:上海统益生物科技有限公司
Il-8 production promoters and use thereof
InactiveUS20070082068A1Increase polarityEasy to produceAntibacterial agentsBiocideHumuleneHouttuynia
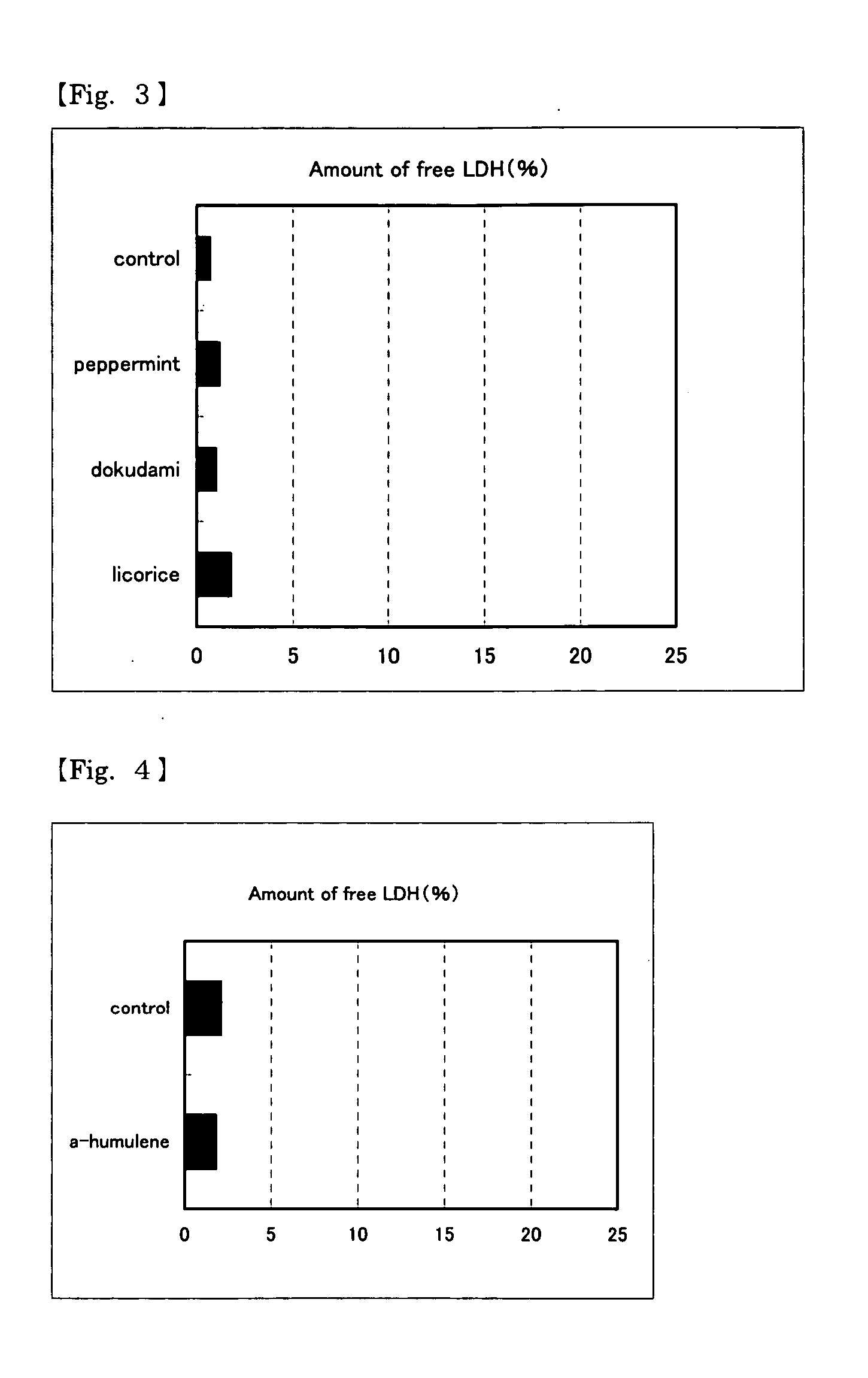
An object of the present invention is to provide IL-8 production promoters derived from safe food materials and to provide immunostimulants or preventive or treating agents of infectious disease comprising the IL-8 production promoter. The present inventors have found that a composition comprising, as an active ingredient, at least one plant extract from peppermint, dokudami (houttuynia herb), or licorice promotes IL-8 production, and that a composition comprising, as an active ingredient, at least one compound selected from the group consisting of α-humulene, α-pinene, β-pinene, and L-menthol promotes IL-8 production. These IL-8 production promoters can be used in foods and drinks supplemented with immunostimulants or preventive or treating agents of infectious disease.
Owner:KANEKA CORP
Novel preparation technology of tasimelteon intermediate
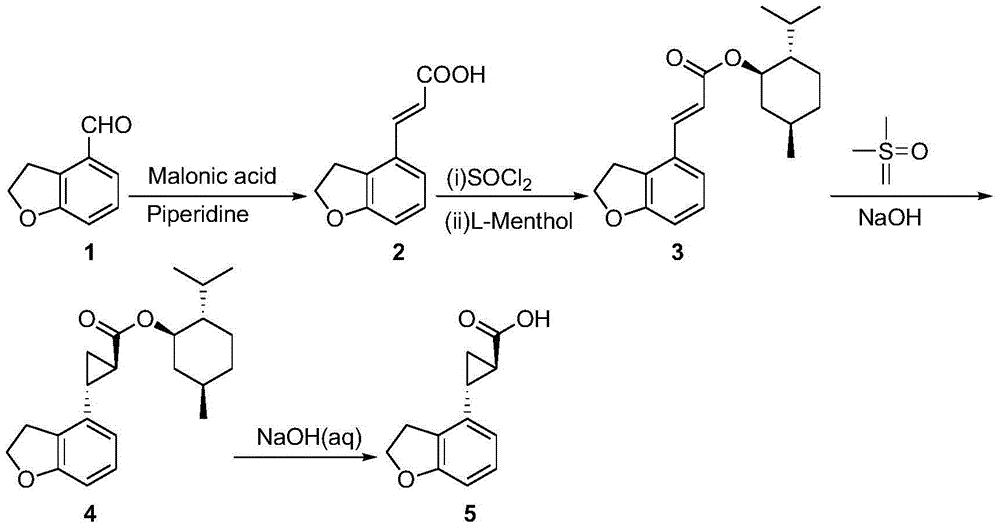
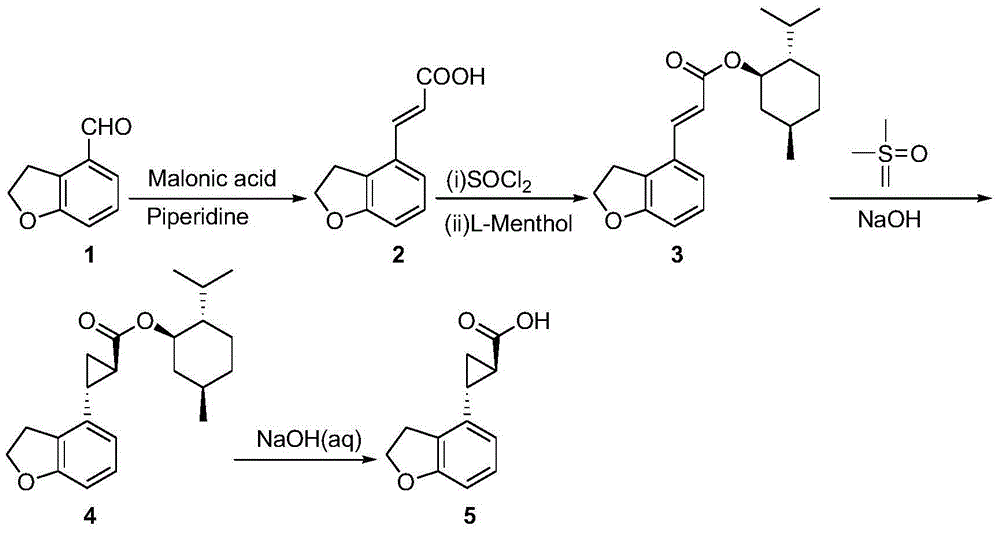
The invention provides a novel preparation technology of a tasimelteon intermediate namely (1R,2R)-(2,3-dihydrobenzofuran-4-yl)cyclopropyl formic acid. The technology comprises the following steps: (1) carrying out reactions between 2,3-dihydrobenzofuran-4-formaldehyde and malonic acid in the presence of piperidine to generate 2,3-dihydrobenzofuran-4-acrylic acid; (2) converting 2,3-dihydrobenzofuran-4-acrylic acid into acyl chloride in the presence of thionyl chloride, then reacting the acyl chloride with L-menthol to generate 2,3-dihydrobenzofuran-4-acrylic acid L-menthol; (3) carrying out reactions between 2,3-dihydrobenzofuran-4-acrylic acid L-menthol and dimethyl sulfur oxide in the presence of sodium hydroxide to generate (1R,2R)-(2,3-dihydrobenzofuran-4-yl)cyclopropyl formic acid L-menthol; (4) hydrolyzing the (1R,2R)-(2,3-dihydrobenzofuran-4-yl)cyclopropyl formic acid L-menthol in the action of a sodium hydroxide water solution so as to obtain the (1R,2R)-(2,3-dihydrobenzofuran-4-yl)cyclopropyl formic acid. The novel preparation technology is developed on the basis of the conventional technologies, and has the advantages of easily-available raw materials, low cost, simple operation, and suitability for industrial massive production.
Owner:苏州莱克施德药业有限公司