Patents
Literature
110 results about "O-aminophenol" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
Method for preparing palladium/carbon catalyst for hydrogenation
InactiveCN1698953AGood dispersionHigh microcrystalline contentOrganic compound preparationCatalyst activation/preparationPhenanthrolineReducing agent
This invention relates to a method for preparing a hydrogenation palladium or carbon catalyst. The catalyst uses the particles or shaping active carbon as the carrier, loading active component metal Pd, wherein the Pd content is 0.2-5wt%. The preparing method comprises the following steps: a) acid cleaning the carrier active carbon; then flushing with water until neutral; drying; b) preparing Pd solution with water-soluble Pd compound and a complex builder; using Pd solution to immerse or spray the carrier active carbon, loading the Pd compound on the active carbon and then getting the catalyst prosoma, wherein the Pd compound content is 15-20wt% computed by Pd, the molecular ratio of complex builder and Pd is (0.01-1):1, the complex builder is any one from 8-bioquin, 2,3,4-trihydroxy-4-sulfonic acid azobenzene, ortho phenanthroline, o-aminophenol, o-hydroxybenzoic acid sodium or salicylaldehyde benzaldoxime; c) aging the catalyst prosoma, then deacidizing and getting the catalyst.
Owner:SINOPEC SHANGHAI PETROCHEMICAL CO LTD
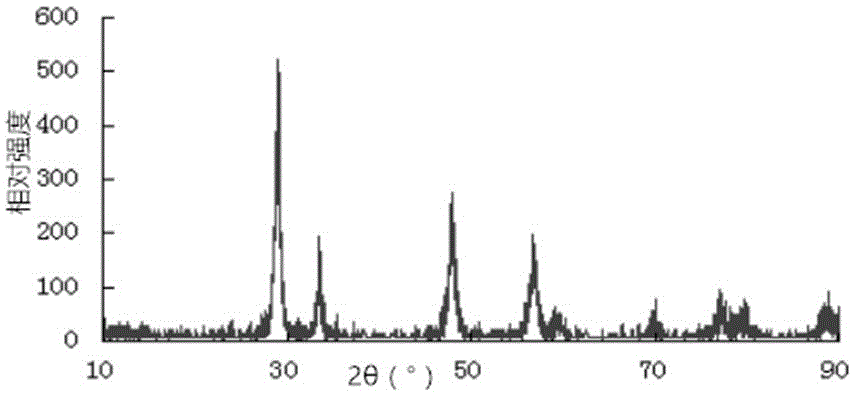
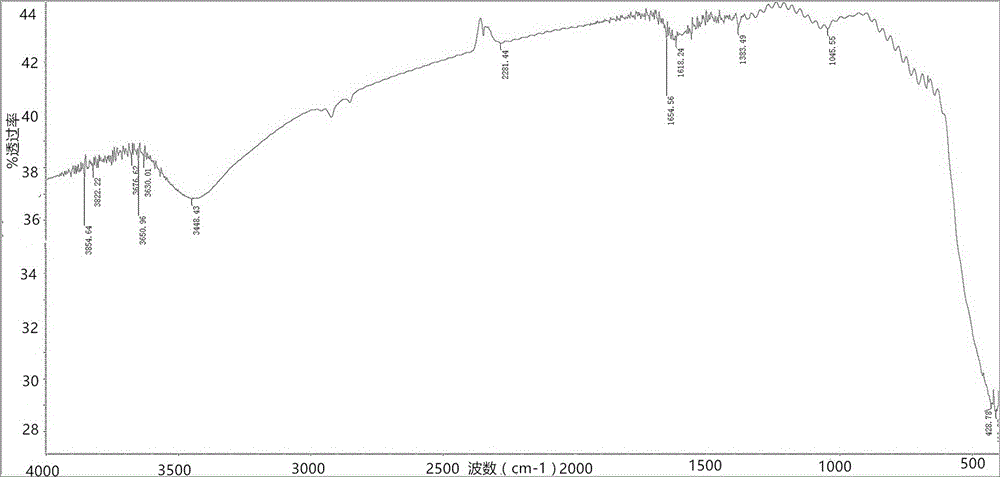
Method for preparing single-molecular magnet [Dy2(saph)2(NO3)2(CH3OH)4]
InactiveCN104557994AStrong ferromagnetismThe synthesis method is simpleGroup 3/13 organic compounds without C-metal linkagesOrganic/organic-metallic materials magnetismSalicylaldehydeRare earth
The invention discloses a method for preparing a single-molecular magnet [Dy2(saph)2(NO3)2(CH3OH)4] and relates to a method for preparing a single-molecular magnet. The invention aims at solving the problems that the synthetic method of the conventional rare earth complex single-molecular magnet is low in yield and the synthetic method of the complex is complicated. The method comprises the following steps: dissolving o-aminophenol salicylaldehyde in acetonitrile, thereby obtaining a solution A; dissolving dysprosium nitrate in methanol, thereby obtaining a solution B; mixing the solution A and the solution B, adding a triethylamine solution into the mixed solution, stirring under room temperature condition, thereby obtaining a preform; and volatilizing a solvent out of the preform, thereby obtaining the complex. The [Dy2(saph)2(NO3)2(CH3OH)4] prepared by the method disclosed by the invention is a single-molecular magnet with good ferromagnetic properties, the yield of the preparation method disclosed by the invention is high and is over 52.84 percent, and the synthetic method of the single-molecular magnet is simple and high in repeatability. The invention belongs to the field of preparation of single-molecular magnets.
Owner:HEILONGJIANG UNIV
Method for preparing catalyst for refining of crude terephthalic acid
ActiveCN100402144CLower redox potentialRestore will notOrganic compound preparationCarboxylic compound preparationNitrite ionPhenanthroline
This is a kind of producing method of catalyst used for refining coarse terephthalic acid (CTA). The catalyst makes grains or molding active carbon as carrier, the content of loading active component metal Pd is 0.2 - 4wt%. It orderly includes: have acid wash to carrier active carbon; wash carrier active carbon with water to neutral; have soakage with watery solution containing nitrite ion to carrier active carbon; dry carrier active carbon to get rid of water; prepare Pd solution with water-soluble Pd compound, a kind of complexing agent and water to infuse or insufflate the carrier active carbon; the content of Pd compound in the Pd solution counted by Pd is 15 - 20wt%; the mol proportion of complexing agent and Pd is (0.01 - 1) : 1, the complexing agent is got from anyone of 8-hydroxyquinoline, 2, 3, 4-trihydroxyl-4-sulfonic azobenzol, o-phenanthroline, o-Aminophenol, o-hydroxybenzoic sodium or o-hydroxybenzaldehyde oxime; after aginged, have deoxidizing treatment with reducing agent to get activator products.
Owner:CHINA PETROLEUM & CHEM CORP
Glassy carbon electrode (GCE) modified by conductive copolymer carbon nanotube composite, preparation method thereof and method for removing perchlorat from water
InactiveCN101634038AIncrease electrode areaEasy to operateWater/sewage treatmentElectrodesCarbon nanotubeAniline
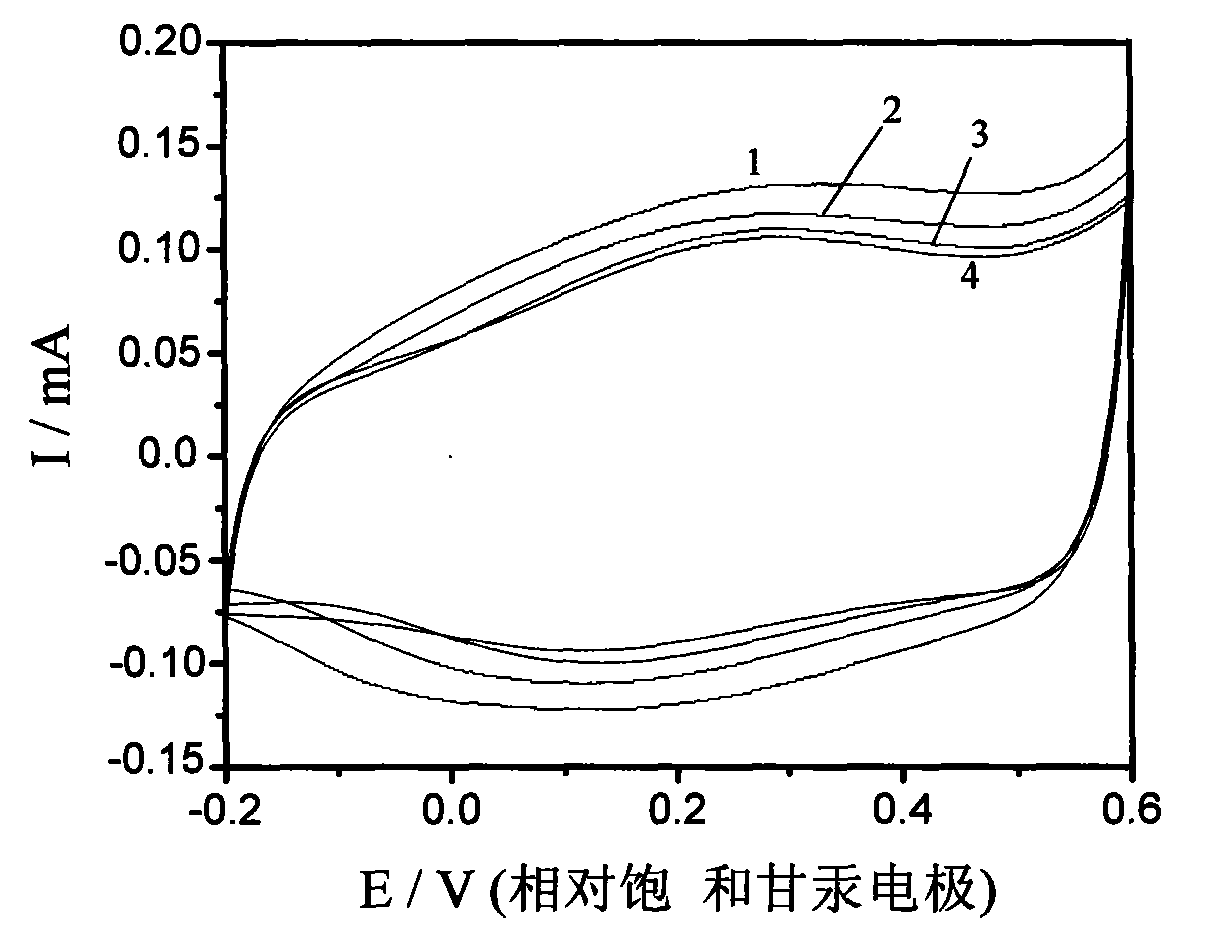
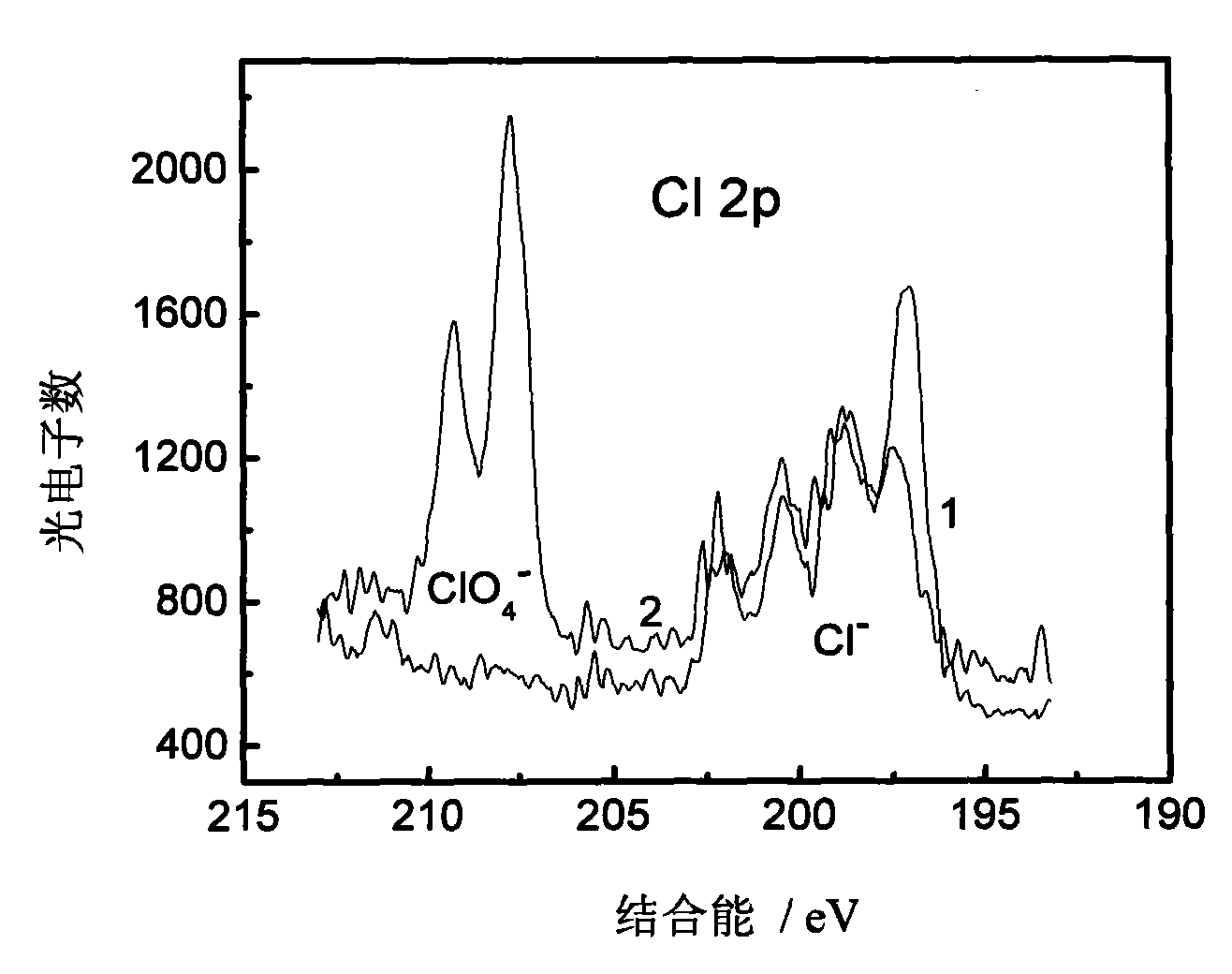
The invention discloses a glassy carbon electrode (GCE) modified by aniline-co-aminophenol copolymer carbon nanotube composite, a preparation method thereof and a method for removing perchlorat from water. The preparation method of the glassy carbon electrode comprises the following steps: dispersing carbon nanotubes in DMF; taking a proper amount of mixed liquor to drip onto the clean surface of the glassy carbon electrode, and air drying; carrying out electrolytic polymerization on a three-electrode system consisting of a glassy carbon working electrode modifying the carbon nanotube, a platinum counter electrode and a calomel reference electrode in the mixed liquor of hydrochloric acid, aniline and o-aminophenol through a constant bit method; thus, generating aniline-co-aminophenol copolymer carbon nanotube composite on the surface of the carbon nanotube modified glassy carbon electrode after a certain time. The modified electrode is washed by diluted hydrochloric acid and secondary water successively, forms the three-electrode system with the platinum counter electrode and the calomel reference electrode, and is doped in the water body containing perchlorat. The perchlorat is doped on the glassy carbon electrode modified by the aniline-co-aminophenol copolymer carbon nanotube composite, which can be seen clearly in an X-ray photoelectron spectroscopy.
Owner:NANJING UNIV
Preparation and application of melamine molecular imprinting electrochemical sensor
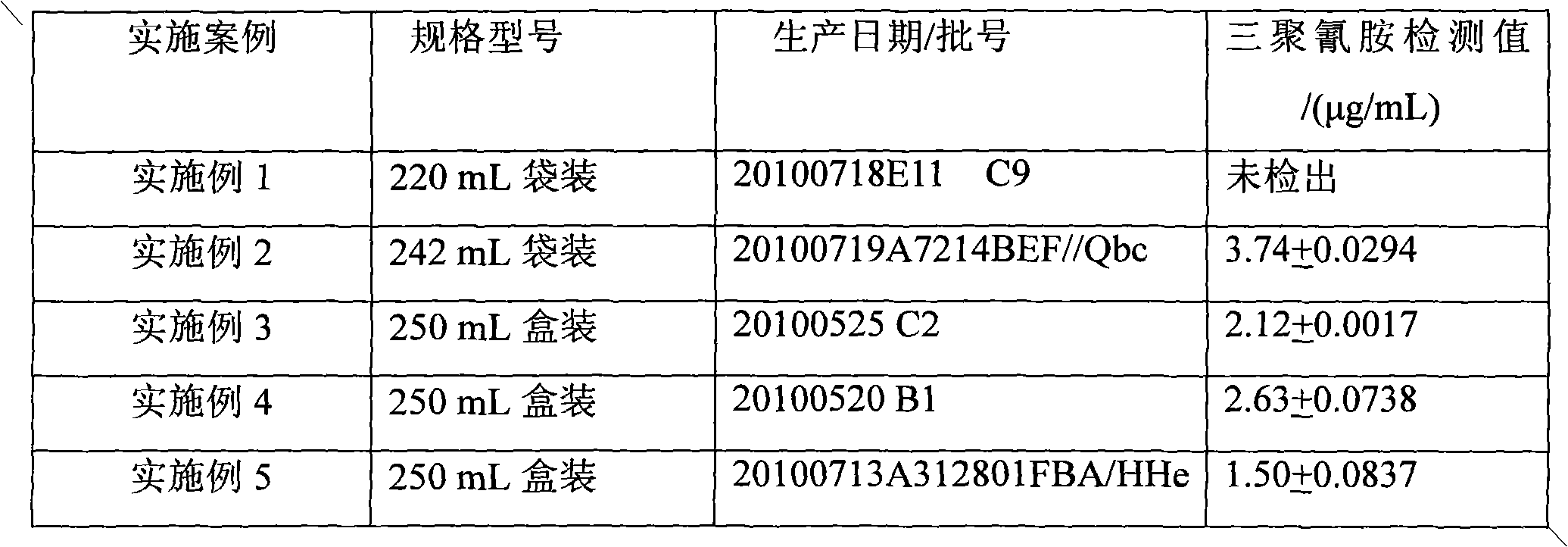
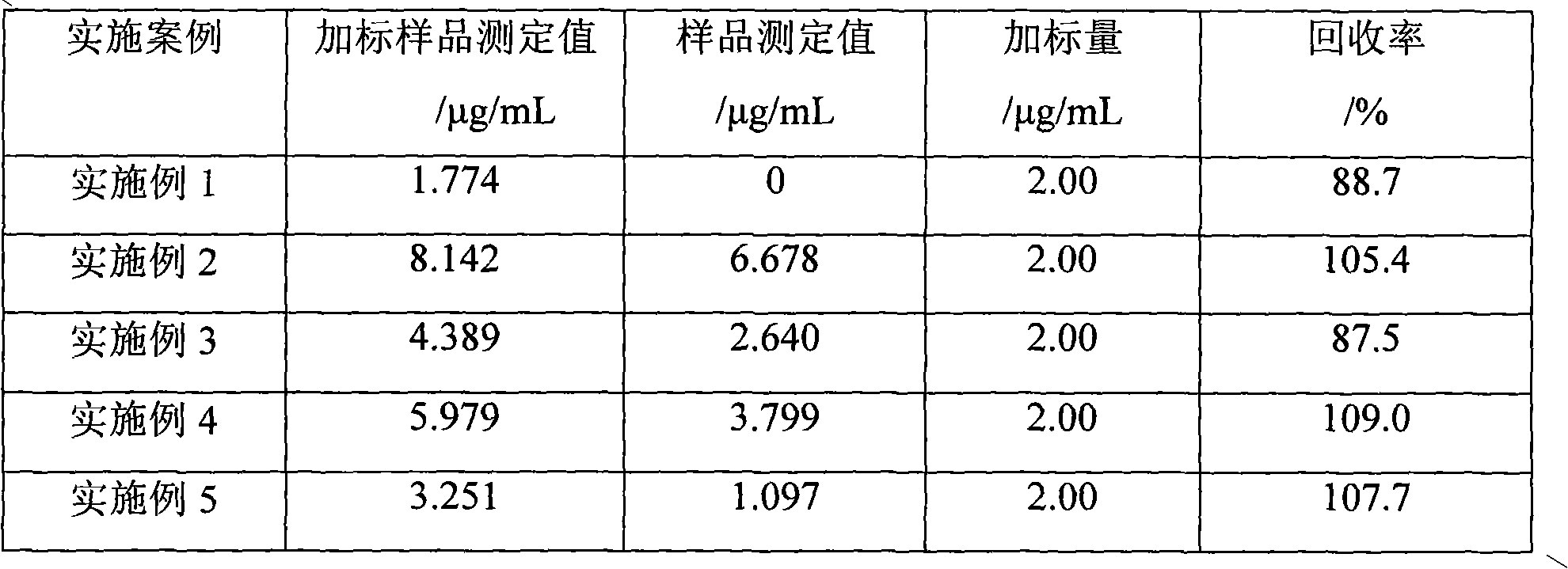
The invention relates to a molecular imprinting electrochemical sensor, which is used for determining melamine in liquid milk and belongs to the technical field of food safety detection. The melamine in the liquid milk is taken as an object of study, and a molecularly imprinted polymer is prepared by electropolymerization on a gold electrode by using the cyclic voltammetry method. The invention constructs a new method for selectively detecting the melamine by taking mixed solution of equimolar hydroxyphenol and o-aminophenol as functional monomers, taking the melamine as template molecules and taking K3Fe(CN)6 as a probe between an imprinting electrode and base solution. The reaction conditions are respectively as follows: the monomer / template mass ratio is 0.55 / 1, the pH value is 5.5, and the temperature is 30 DEG C. The molecularly imprinted polymer imprinted polymer thin film which leaves template molecular configuration holes is prepared by using methanol solution to carry out ultrasonic elution for 6min and removing the template molecules from the electrode. The detection is carried out through the differential pulse method, and the linear range is 0.50-7.50mg / kg. The response time is 10 seconds. The molecular imprinting electrochemical sensor can be used for detecting an actual liquid milk sample without pretreatment, the selectivity is good, and the recovery rate of adding standard is 87.5-109%.
Owner:BEIJING TECHNOLOGY AND BUSINESS UNIVERSITY
Cubic nano CeO2 and application thereof in degrading o-aminophenol
InactiveCN105731514AImprove stabilityEasy to storeRare earth metal oxides/hydroxidesMaterial nanotechnologyCerium(IV) oxideSURFACTANT BLEND
The invention belongs to the technical field of water treatment, and particularly relates to a cubic nano cerium oxide (CeO2) and application thereof as a mimic oxidase in degrading o-aminophenol. In the CeO2 preparation process, Ce(NO3)3.6H2O is used as a cerium source, urea is used as a precipitant, and CTAB (cetyltrimethylammonium bromide) is used as a surfactant. A precursor is firstly prepared, and the precursor is calcined to obtain the CeO2. Compared with the natural oxidase, the cerium oxide provided by the invention has the advantages of high stability, low denaturation tendency, wide application range and high reutilization ratio, is easy for storage and transportation, also has high-efficiency oxidase catalytic activity, and thus, can well substitute the existing natural oxidase. When the cerium oxide provided by the invention is used in degrading o-aminophenol, the maximum degradation rate can reach 70% or above.
Owner:ZHENGZHOU UNIVERSITY OF AERONAUTICS
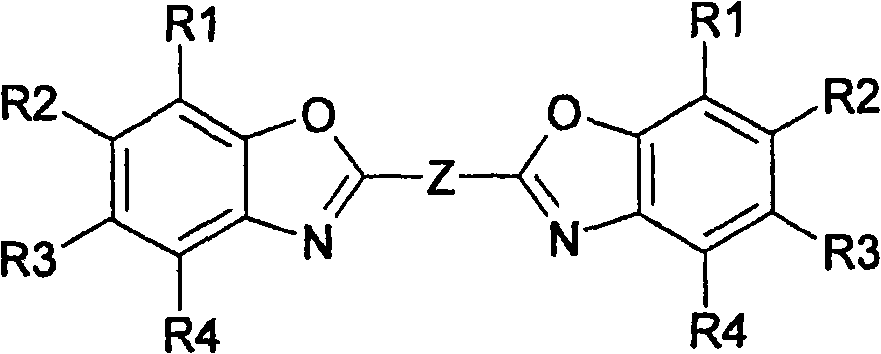
Method for producing bisbenzoxazoles
The invention relates to a method for producing bisbenzoxazoles that are interconnected by means of a system of conjugated double bonds, according to which o-aminophenols are reacted with dicarboxylic acids, the carboxyl groups of which are interconnected via a double bond or a system of conjugated double bonds, to form an ammonium salt, said ammonium salt being converted in the presence of dehydrogenating catalysts and solvents with a low dielectric loss into benzoxazol by means of microwave radiation.
Owner:CLARIANT FIANCE (BVI) LTD
Preparation method of single-molecular magnet [Dy2(saph)2Cl2].4CH3OH
InactiveCN103641850AStrong ferromagnetismThe synthesis method is simpleOrganic/organic-metallic materials magnetismGroup 3/13 element organic compoundsSalicylaldehydeSynthesis methods
The invention discloses a preparation method of a single-molecular magnet [Dy2(saph)2Cl2].4CH3OH and relates to the preparation method of the single-molecular magnet [Dy2(saph)2Cl2].4CH3OH, which is used for solving the problems that an existing synthesis method for a rear-earth cluster compound single-molecular magnet is lower in yield, complex in synthesis method of a coordination compound and incapable of carrying out mass production. The preparation method comprises the following steps: I, dissolving o-aminophenol salicylaldehyde acetal into acetonitrile to obtain liquor A; dissolving dysprosium chloride into methanol to obtain liquor B; mixing the liquor A with the liquor B, adding triethylamine with concentration of 0.001mol / L to obtain mixed liquor; II, stirring the mixed liquor obtained in the step I under the room temperature to obtain a preform; and III, volatilizing the preform in the solvent to obtain the single-molecular magnet [Dy2(saph)2Cl2].4CH3OH. The preparation method disclosed by the invention is simple, and high in yield which reaches over 45.76%. The preparation method disclosed by the invention is applied to the field of preparing the single-molecular magnets.
Owner:HEILONGJIANG UNIV
Method for preparing catalyst for refining of crude terephthalic acid
InactiveCN1709568AGood dispersionHigh microcrystalline contentMetal/metal-oxides/metal-hydroxide catalystsNitrite ionPhenanthroline
This is a kind of producing method of catalyst used for refining coarse terephthalic acid (CTA). The catalyst makes grains or molding active carbon as carrier, the content of loading active component metal Pd is 0.2 - 4wt%. It orderly includes: have acid wash to carrier active carbon; wash carrier active carbon with water to neutral; have soakage with watery solution containing nitrite ion to carrier active carbon; dry carrier active carbon to get rid of water; prepare Pd solution with water-soluble Pd compound, a kind of complexing agent and water to infuse or insufflate the carrier active carbon; the content of Pd compound in the Pd solution counted by Pd is 15 - 20wt%; the mol proportion of complexing agent and Pd is (0.01 - 1) : 1, the complexing agent is got from anyone of 8-hydroxyquinoline, 2, 3, 4-trihydroxyl-4-sulfonic azobenzol, o-phenanthroline, o-Aminophenol, o-hydroxybenzoic sodium or o-hydroxybenzaldehyde oxime; after aginged, have deoxidizing treatment with reducing agent to get activator products.
Owner:CHINA PETROLEUM & CHEM CORP
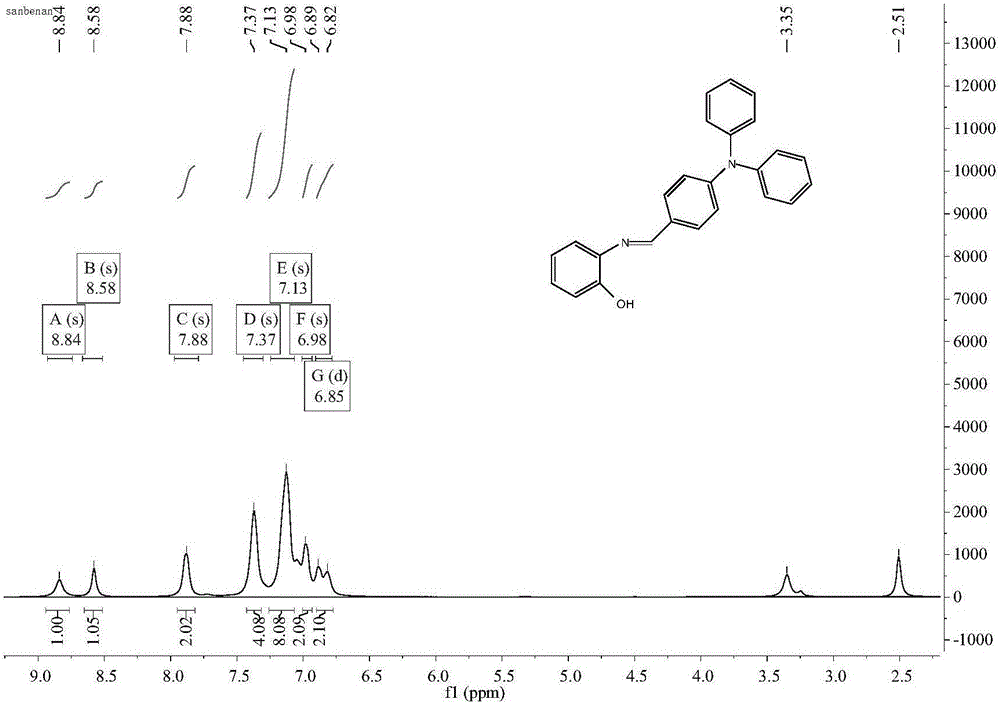
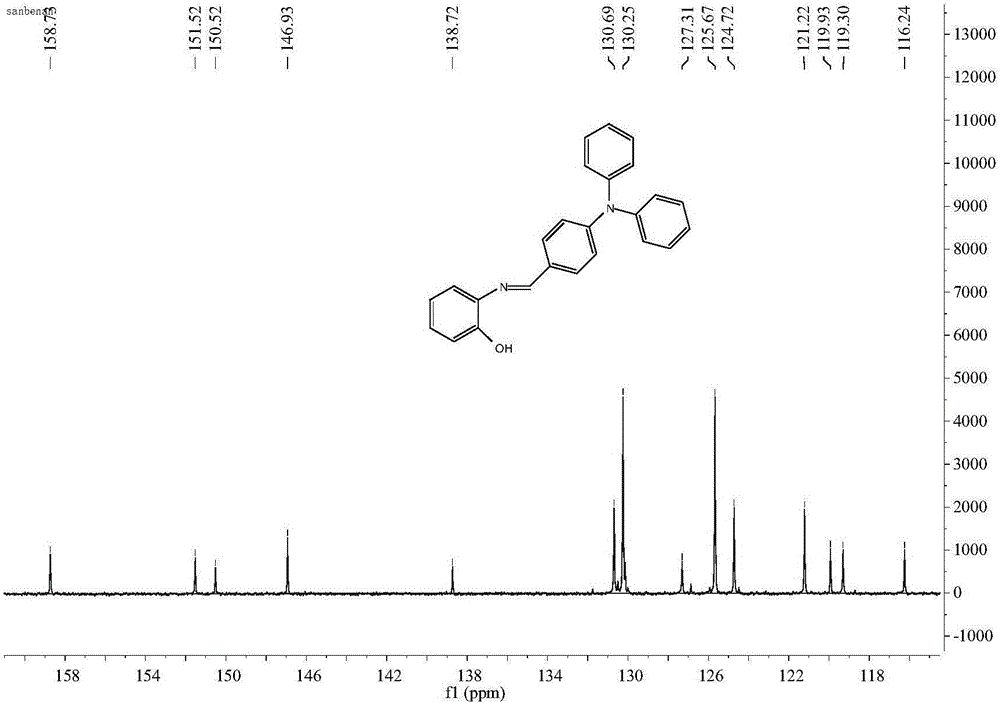
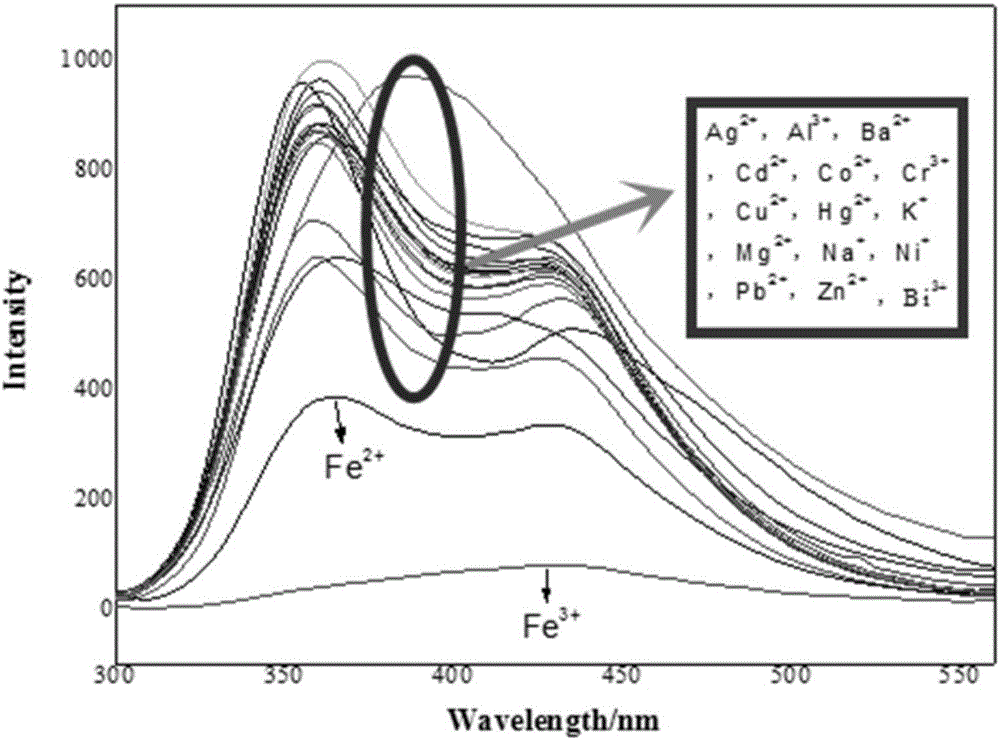
Iron ion fluorescent probe compound as well as preparation method and application thereof
InactiveCN106083645AGood choiceHigh sensitivityMaterial analysis by observing effect on chemical indicatorFluorescence/phosphorescenceOrganic solventLuminous intensity
The invention relates to the field of fluorescent probe compounds and discloses an iron ion fluorescent probe compound as well as a preparation method and application thereof. The fluorescent probe compound has the advantages of high luminous intensity, high sensitivity and wide pH adaptability. The fluorescent probe compound disclosed by the invention is a triphenylamine Schiff base type derivative with the chemical name of 2-hydroxyl-N-imidogenaniline-5-N,N-diphenylaniline. The invention further discloses the preparation method of the fluorescent probe compound, comprising the following steps: (a) taking ortho-aminophenol and 4-diphenylaminobenzaldehyde to react in an organic solvent to obtain a reaction solution; and (b) carrying out post-treatment on the reaction solution to obtain the fluorescent probe compound. The invention further discloses application of the fluorescent probe compound to detection of Fe<3+> ions.
Owner:GUANGDONG UNIV OF TECH
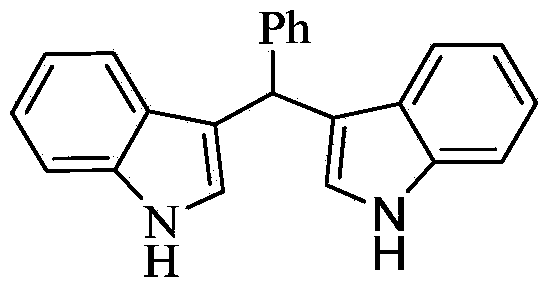
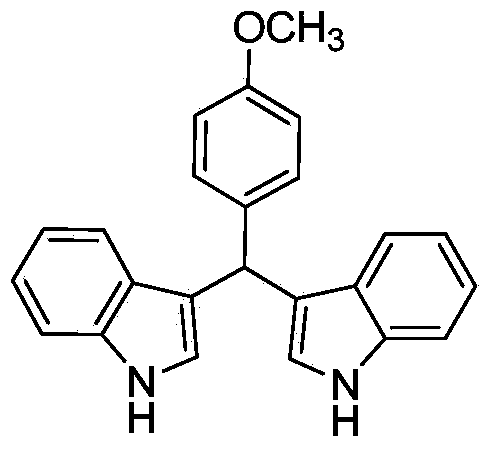
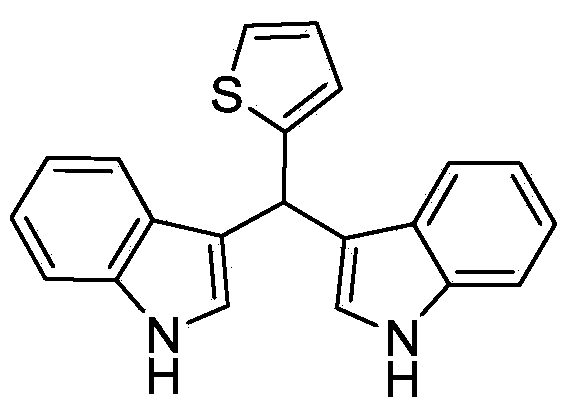
Method for preparing diindolyl methane derivatives
The invention discloses a method for preparing diindolyl methane derivatives. The method comprises the following step: with titanocene dichloride and a ligand as catalysts, reacting indole and aldehyde to obtain the diindolyl methane derivative under an alkaline condition, wherein the ligand can be 8-hydroxyquinoline, catechol and o-aminophenol and the alkali is aniline, pyridine, pyrrole or triethylamine. The method is easy to operate, the reaction is high-efficiency and mild, the catalysts are low in price, non-toxic, stable to air and water, and substrate applicability is good; therefore, the method can be widely applied to preparation of diindolyl methane derivatives.
Owner:SHAANXI NORMAL UNIV +1
Green method for preparing heterocyclic compounds from nitriles
The invention provides a green method for preparing heterocyclic compounds from nitriles. The method is characterized in that nitriles and compounds such as o-aminophenol, aminothiophenol, beta-amino alcohol, beta-amino thioalcohol and the like are catalyzed by a non-transition metal catalyst to synthesize the heterocyclic compounds such as benzoxazole, benzothiazole, oxazoline, thiazoline and the like by one step. The method has the beneficial effects that the non-transition metal catalyst with low price, water solubility and low toxicity is used, inert gas shielding is not needed, and the reaction can be directly carried out in the air to obtain the target heterocyclic compounds; the reaction conditions are simple and mild, the reaction is easy to operate, and the problems of transitional hydrolysis of nitriles and generation of byproducts are avoided, so that the products are simple and easy to separate and purify and have high recovery rates; and the method has lower requirements for the reaction conditions, wide range of application and good research and industrial application prospects.
Owner:WENZHOU UNIVERSITY
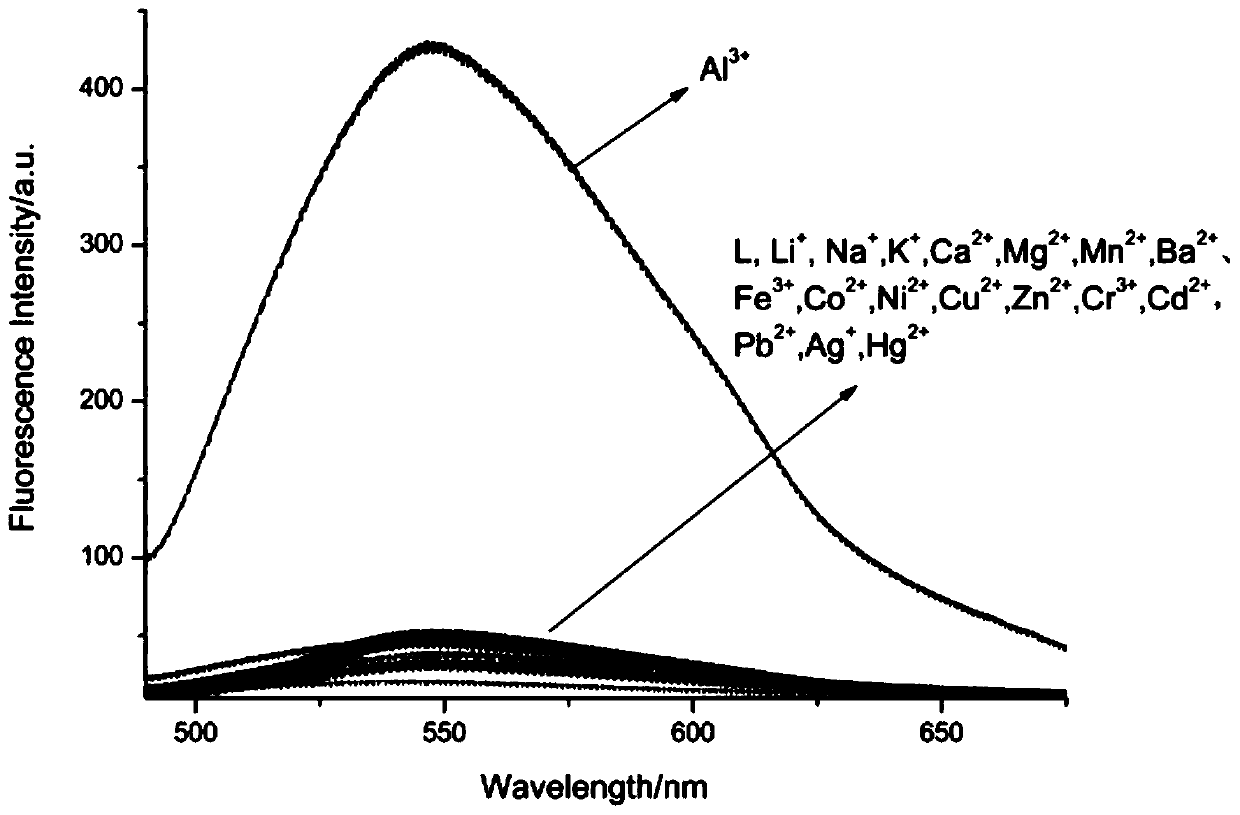
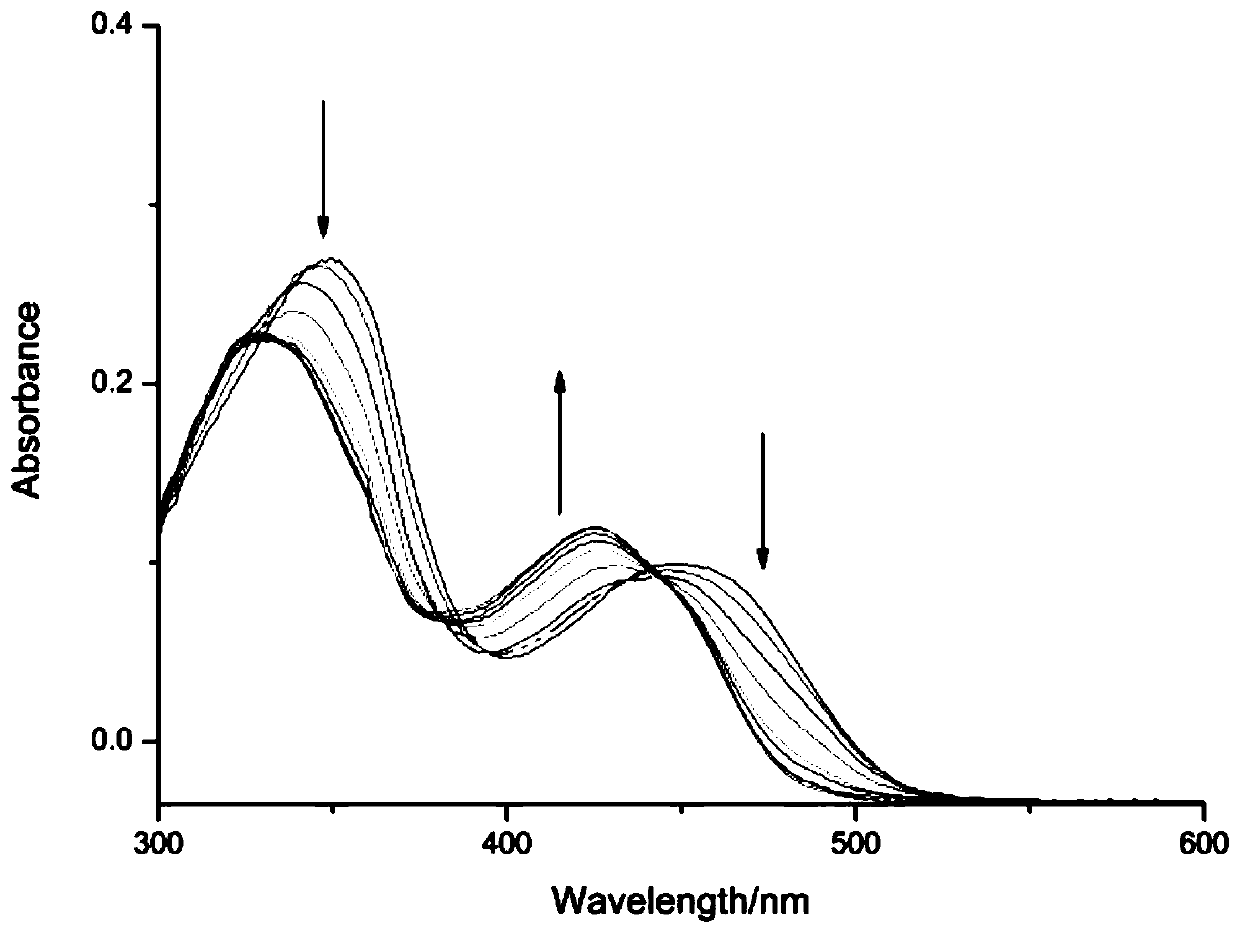
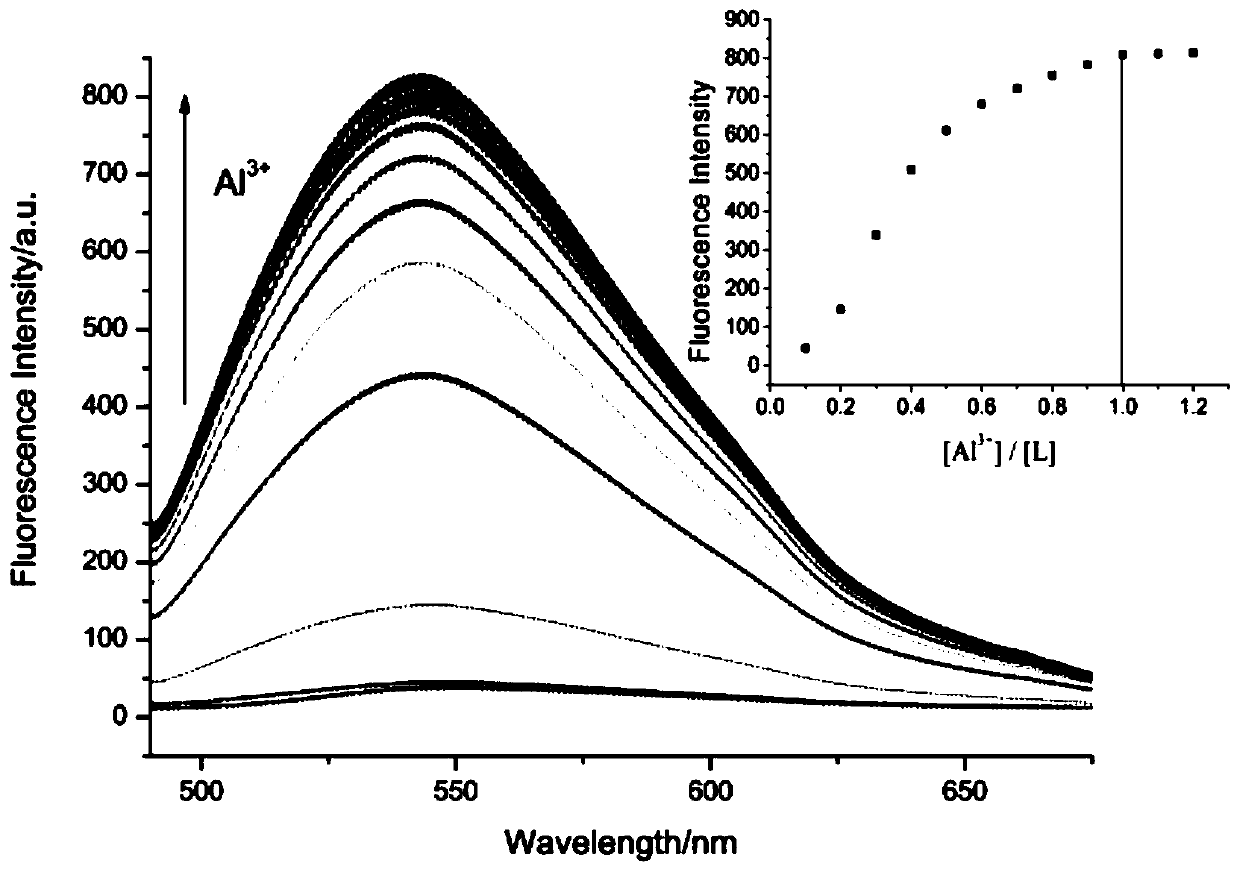
Aluminum ion fluorescent probe and preparation method thereof
PendingCN109912555AGood choiceGood reversibilityOrganic chemistryFluorescence/phosphorescenceAluminum IonAcetic acid
The invention discloses an aluminum ion fluorescent probe and a preparation method thereof. The method comprises the following steps: synthesizing 7-hydroxycoumarin-8-aldehyde by taking hexamethylenetetramine, 7-hydroxycoumarin and glacial acetic acid, and synthesizing the aluminum ion fluorescent probe by utilizing 7-hydroxycoumarin-8-aldehyde and ortho-aminophenol. The structure of the aluminumion fluorescent probe is represented by a nuclear magnetic spectrum and a mass spectrum; the spectral quality of the fluorescent probe is analyzed through an ultraviolet spectrum and a fluorescent spectrum; the result indicates that the fluorescent probe has high selectivity and is not interfered by other co-existing ions when identifying aluminum ions; and the probe generates coordination interaction with the aluminum ions according to the ratio of 1:1 and the reaction has reversibility. Therefore, the fluorescent probe can be applied to detection on the aluminum ions well.
Owner:YULIN UNIV
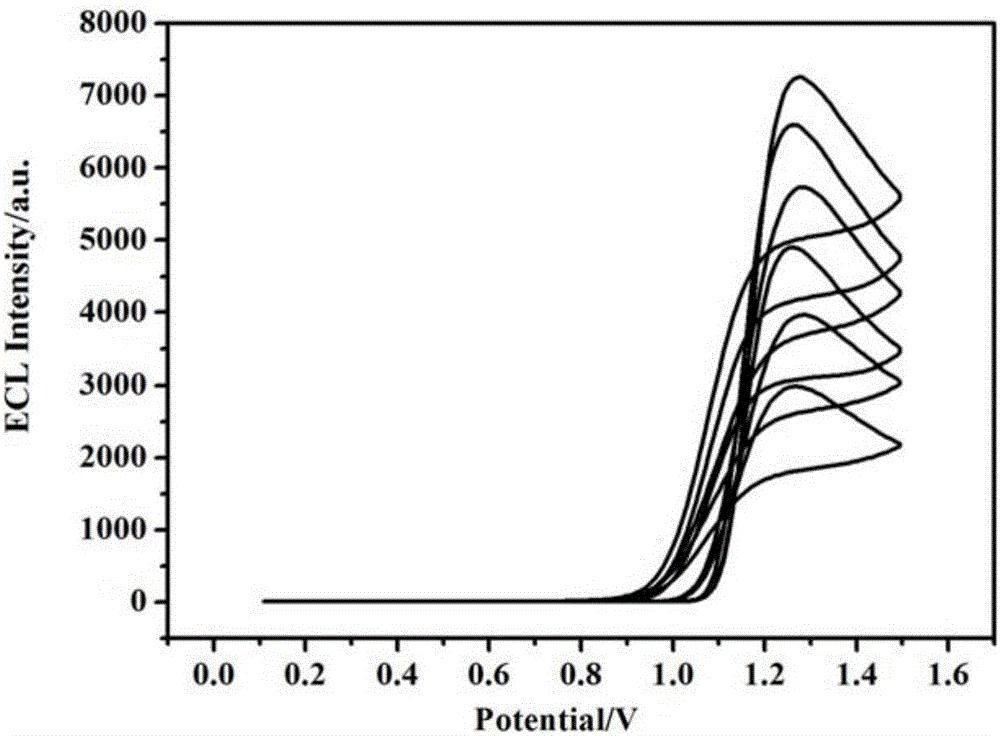
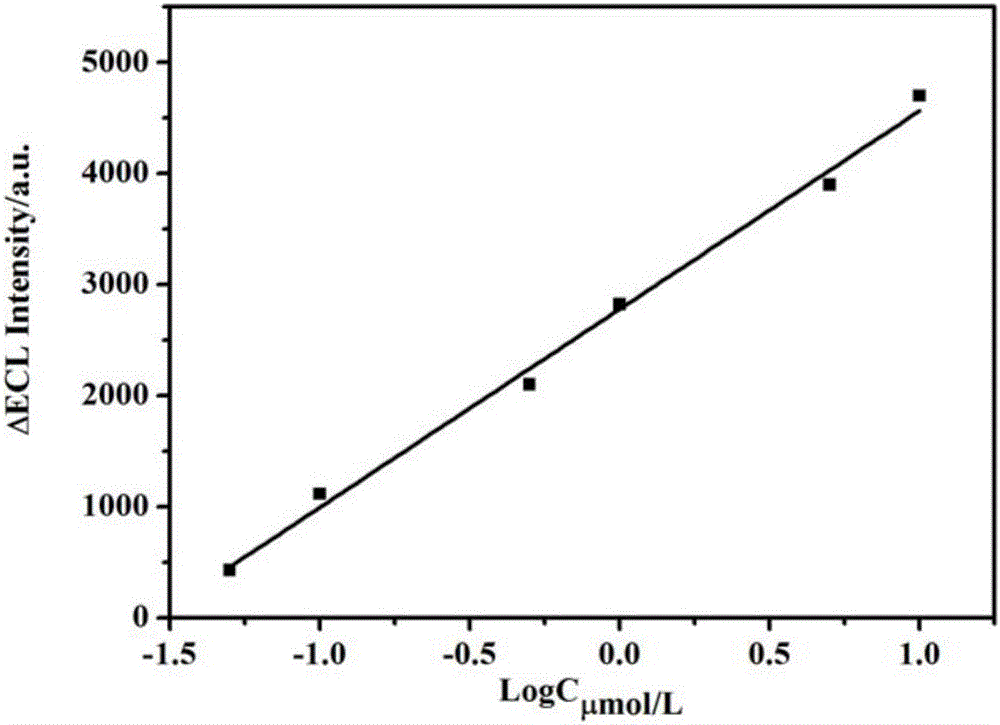
Method for fixation of tris bipyridine ruthenium and electrochemiluminescence detection of malachite green
ActiveCN106556593AEasy to fixEasy to prepareChemiluminescene/bioluminescenceMaterial electrochemical variablesMalachite greenMalachite green stain
Belonging to the field of electrochemiluminescence detection, the invention relates to a method for detection of malachite green. A poly(o-aminophenol-sulphonic acid)-tris bipyridine ruthenium modified glassy carbon electrode is adopted as the working electrode, a platinum electrode is taken as the auxiliary electrode, and a saturated calomel electrode is taken as the reference electrode to compose a three-electrode system to detect malachite green by electrochemiluminescence method. The method has a detection range of 5.0*10<-8>-1.0*10<-5>mol / L, and a minimum detection limit of 1.5*10<-8>mol / L. The method for detection of malachite green provided by the invention has the advantages of low cost, high sensitivity, simple operation and good selectivity.
Owner:CHANGZHOU UNIV
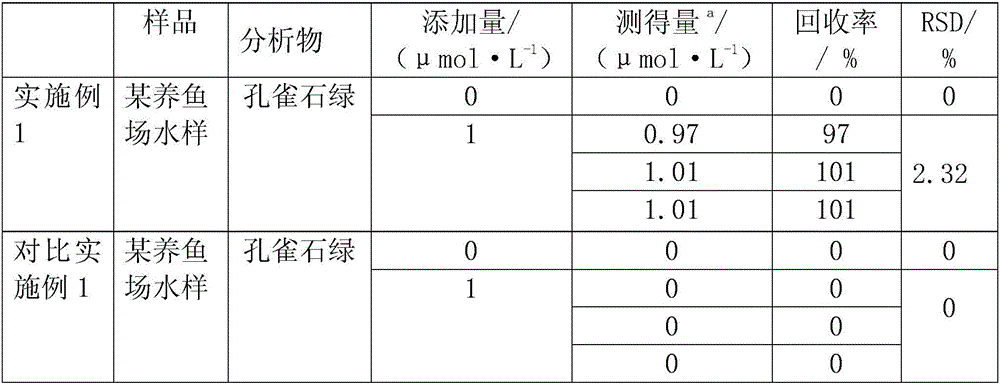
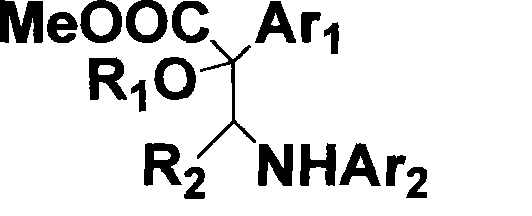
Method for synthesizing alpha-alkoxyl-beta-amino ester derivative
InactiveCN101531606ASimple and safe operationShort reaction timeOrganic compound preparationAmino-carboxyl compound preparationAmino estersDistillation
The invention provides a method for synthesizing an alpha-alkoxyl-beta-amino ester derivative, and relates to a process for synthesizing an intermediate compound of an anticancer medicament. The method comprises the following steps: weighing rhodium acetate, o-aminophenol, aldehyde, alkoxytitanium and a diazo compound according to molar ratio of 0.01:1.0:1.0:1.1:2.0; dissolving the aldehyde, the o-aminophenol, the alkoxytitanium, the rhodium acetate and a dehydrating agent in an organic solvent; stirring the mixture for 2 to 3 hours at room temperature to prepare a reaction system; dissolving the diazo compound into the solvent consisting of the organic solvent by a sampling pump to be injected into the reaction system; dripping saturated NaHCO3 aqueous solution to perform a quenching reaction after 2 to 3 hours of reaction; removing the solvent by reduced pressure distillation to prepare a crude product of the alpha-alkoxyl-beta-amino ester derivative with two chiral centers; and carrying out column chromatography on the crude product by ethyl acetate and sherwood oil according to the ratio of 1:50-1:20 to obtain a pure product. The method has convenient and safe operation, short reaction time and high efficiency; the reaction condition at room temperature is mild; raw materials have wide resources; and the product can be widely applied to synthesis of intermediate compound of the anticancer medicament.
Owner:EAST CHINA NORMAL UNIV
Method for preparing catalyst for refining of crude terephthalic acid
ActiveCN1709570AImprove anchoring abilityHigh affinityMetal/metal-oxides/metal-hydroxide catalystsNitrite ionPhenanthroline
This is a kind of producing method of catalyst used for refining coarse terephthalic acid (CTA). The catalyst makes grains or molding active carbon as carrier, the content of loading active component metal Pd is 0.2 - 4wt%. It orderly includes: have acid wash to carrier active carbon; wash carrier active carbon with water to neutral; have soakage with watery solution containing nitrite ion to carrier active carbon; dry carrier active carbon to get rid of water; prepare Pd solution with water-soluble Pd compound, a kind of complexing agent and water to infuse or insufflate the carrier active carbon; the content of Pd compound in the Pd solution counted by Pd is 15 - 20wt%; the mol proportion of complexing agent and Pd is (0.01 - 1) : 1, the complexing agent is got from anyone of 8-hydroxyquinoline, 2, 3, 4-trihydroxyl-4-sulfonic azobenzol, o-phenanthroline, o-Aminophenol, o-hydroxybenzoic sodium or o-hydroxybenzaldehyde oxime; after aginged, have deoxidizing treatment with reducing agent to get activator products.
Owner:CHINA PETROLEUM & CHEM CORP
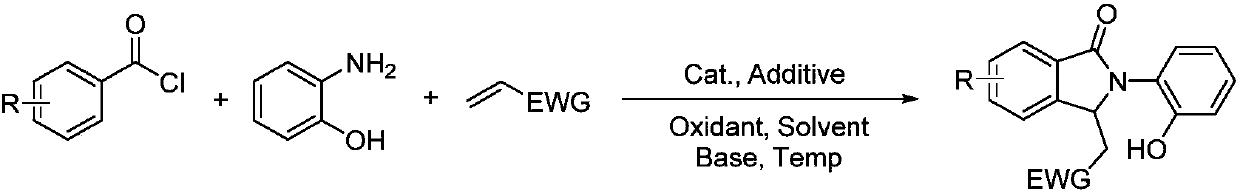
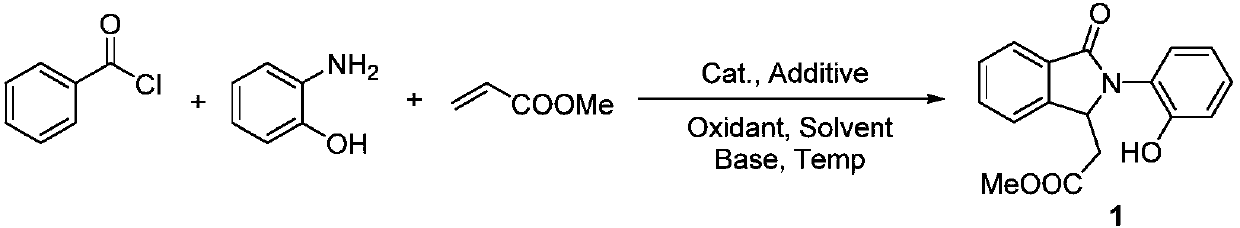
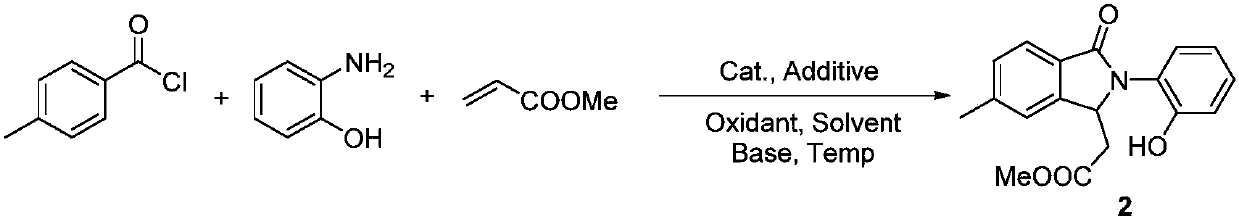
Synthesis method of polysubstituted isoindoline derivative
The invention belongs to the field of organic synthesis and in particular relates to a synthesis method of a polysubstituted isoindoline derivative. The synthesis method comprises the following steps:after sufficiently mixing aryl chloride, o-aminophenol, olefin with an electron withdrawing group, a catalyst, an additive, an oxidant, an alkali and a solvent, raising temperature and reacting; after reaction is finished, carrying out post-treatment to obtain a target product.
Owner:CHANGZHOU UNIV
OAP-MIP multiple binding site affinity membrane chlortetracycline (CTC) sensor and preparation method thereof
InactiveCN105092671AMeet the requirements of trace analysisQuick responseMaterial electrochemical variablesFunctional monomerBinding site
The invention relates to a chlortetracycline (CTC) molecularly imprinted (MIP) affinity membrane sensor and a preparation method thereof. The specific affinity membrane is formed based on an electropolymerization method, and CTC as a template molecule and o-aminophenol (OAP) as a functional monomer form a molecularly imprinted polymer membrane on the surface of a glassy carbon electrode by an electrochemistry method. CTC contains a phenolic hydroxyl group, an enol group and a dimethylamino group and the hydroxyl group in the OAP structure can be used as a latent coordination site so that OAP amino group, and phenolic hydroxyl group and enol group of CTC undergo intermolecular hydrogen bond interaction. OAP hydroxyl group and CTC dimethylamino group undergo a reaction so that multiple binding sites between OAP and CTC exist and the prepared molecularly imprinted polymer has very high CTC selectivity and bonding capability. The sensor can be used for on-site fast detection, does not need specimen pre-treatment, and has a low cost, simple processes and high sensitivity. The sensor has a detection linear range of 20-610nM and sensitivity of 15nM.
Owner:TIANJIN UNIVERSITY OF SCIENCE AND TECHNOLOGY
Catechol detection sensor, preparing process and application thereof
InactiveCN1667406AHigh sensitivityReduce the degree of oxidationThiol preparationMaterial electrochemical variablesPolymer scienceConductive polymer
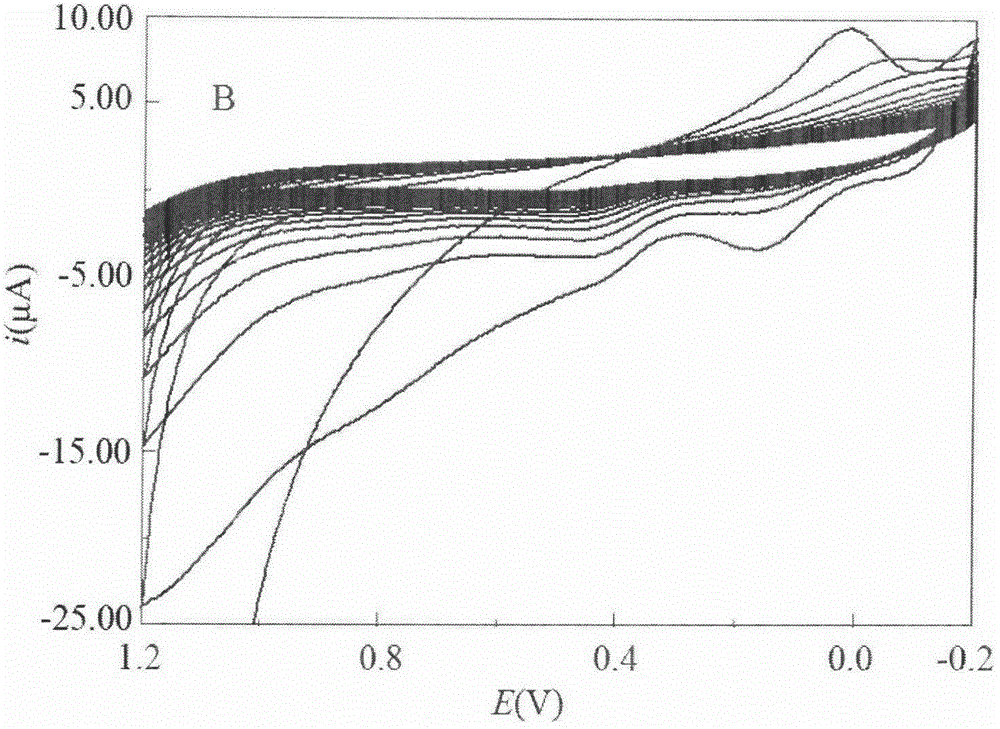
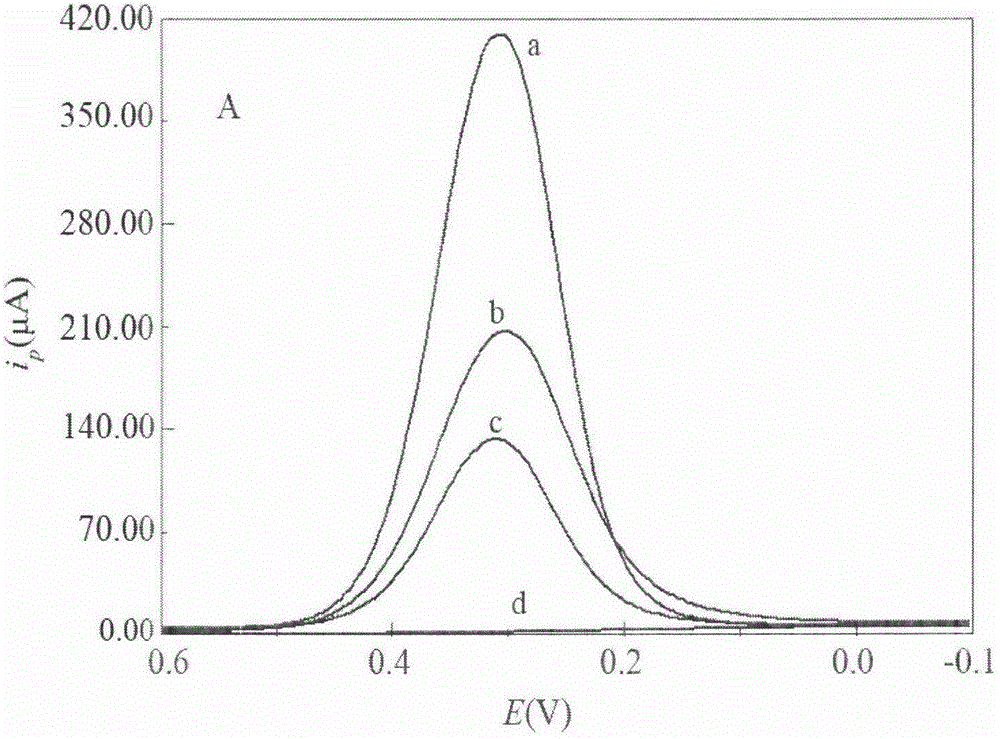
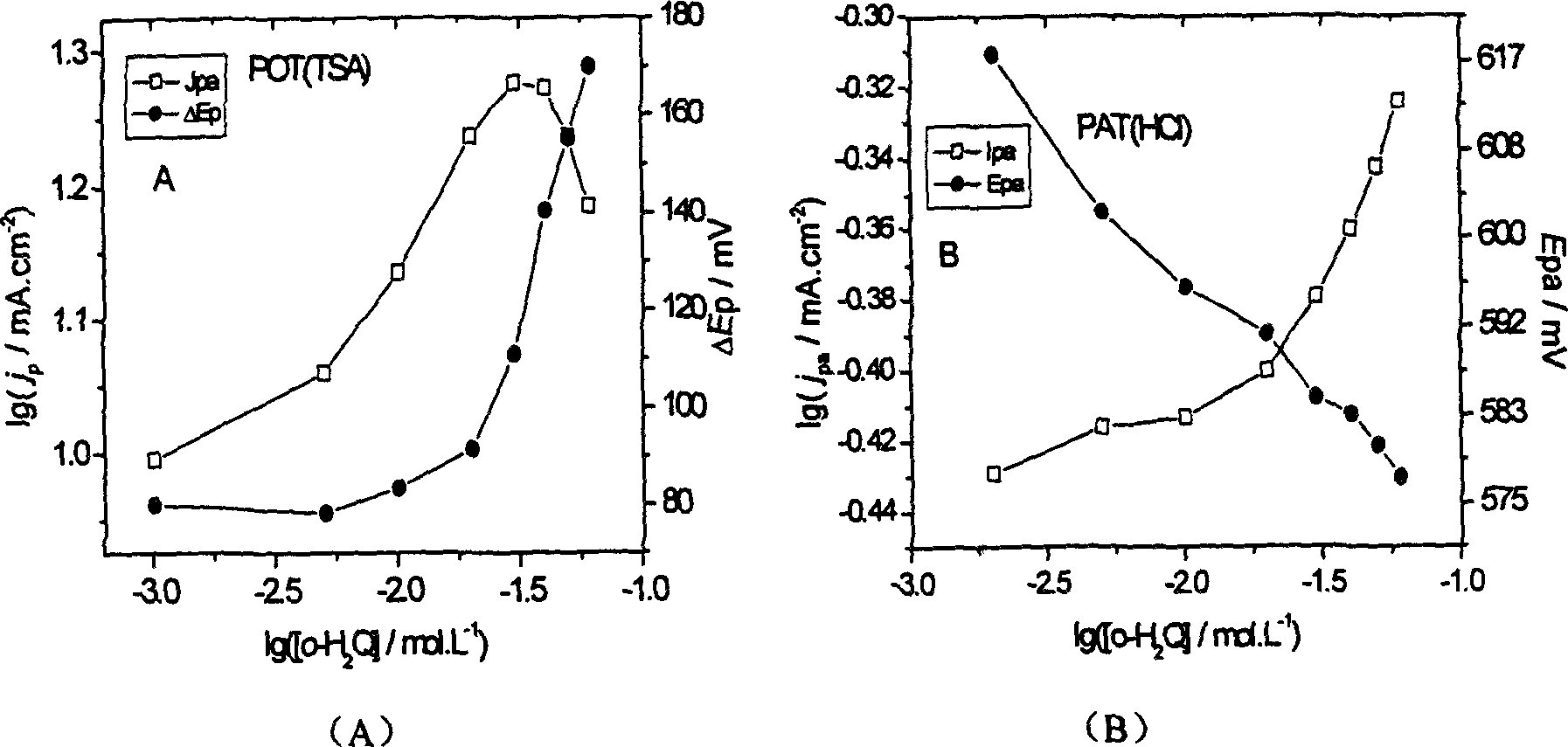
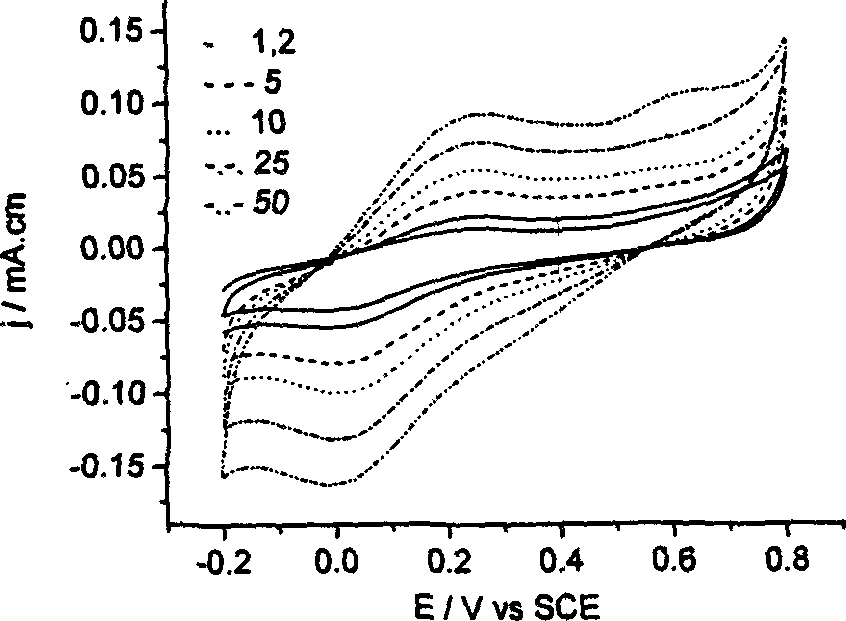
The invention discloses a catechol detection sensor, which relates to electron conductive polymer o-aminophenol polymer membrane modification electrode and its process method, and is applied to detection for water pollutant and neurotransmitter catechol.The sensor comprises inert electrode base and conductive polymer modification membrane, wherein, conductive polymer modification membrane is emulsion polymerisate polymer o-aminophenol and polymer adhesion amount on the surface of base electrode is 0.3í½1.5mgíñcm-2.The method comprises following steps: putting PAT(HCl) mixed with alcaine in N,N-dimethyl formamide; coating polymer liquor on electrode surface and drying it; then conducting periodic voltampere scan for polymer membrane modification electrode in 1.0mol / L acid medium.
Owner:XIAMEN UNIV
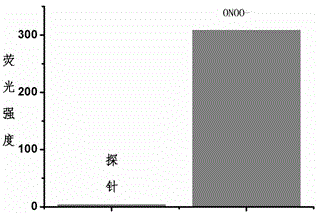
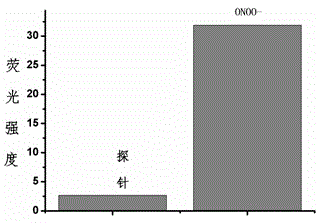
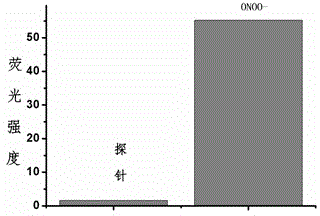
Fluorescent probe for detecting peroxynitrite, and preparation and application of fluorescent probe
InactiveCN104893711AImprove stabilityLong-term storage and useOrganic chemistryFluorescence/phosphorescencePeroxynitriteNitroso
The invention provides a fluorescent probe for detecting peroxynitrite. The probe has a general structure formula Ia or Ib. The probe is prepared by: (1) stirring R4-substituted o-aminothiophenol or R4-substituted o-aminophenol and R1 / R2-substituted benzaldehyde in a methanol solution at a room temperature for two hours to obtain Ia; and (2) performing oxidative condensation of R4-substituted o-aminothiophenol or o-aminophenol and R2-substituted o-hydroxybenzaldehyde to obtain a benzothiazole or benzoxazole intermediate, coupling triflate converted by the phenolic hydroxyl group of the intermediate and R3-substituted p-hydroxybenzene tert-butyldimethylsilylester / p-aminobenzene tert-butyldimethylsilylester, and performing methylation of aniline nitrogen atoms and hydroxy elimination of silylester protecting group to obtain Ib. The fluorescent probe provided in the invention can have fast specific reaction with peroxynitrite to generate a product with strong fluorescence, so that specific detection of peroxynitrite is realized. The general structure formula of the probe in the invention is shown in the description.
Owner:ZHEJIANG UNIV
High-efficiency energy-saving diesel fuel additive
InactiveCN104987902AIncrease profitImprove protectionLiquid carbonaceous fuelsIsobutanolOil additive
A high-efficiency energy-saving diesel fuel additive is prepared from the following raw materials in parts by weight: 4-8 parts of sodium carboxymethylcellulose, 2-7 parts of 2-methyl cellosolve, 4-6 parts of tritolyl phosphate, 5-8 parts of calcium peroxide, 20-26 parts of ethyl alcohol, 5-7 parts of ferrocene, 3-7 parts of petroleum ether, 5-7 parts of zinc primary alcohol dialkyl thiophosphate, 4-6 parts of graphite, 5-7 parts of N,N-disulfoxide salicyl propane diamine, 4-7 parts of isobutanol, 8-12 parts of ethelene glycol alkyl monophenyl ether, 3-7 parts of 3-hydroxybutyraldehyde, 4-8 parts of cyclopentane, 2-6 parts of triethyl orthoacetate, 4-7parts of ortho aminophenol, 2-4 parts of a modified additive and 4-6 parts of spermine. The high-efficiency energy-saving diesel fuel additive has the benefits that the utilization ratio of diesel oil is improved, an excellent protective effect is played for a fuel oil system, the heat efficiency is improved, the oil consumption is effectively reduced, and an efficient energy-saving effect can be played.
Owner:QINGDAO JINGSHITONG PRECISION MACHINERY MFG
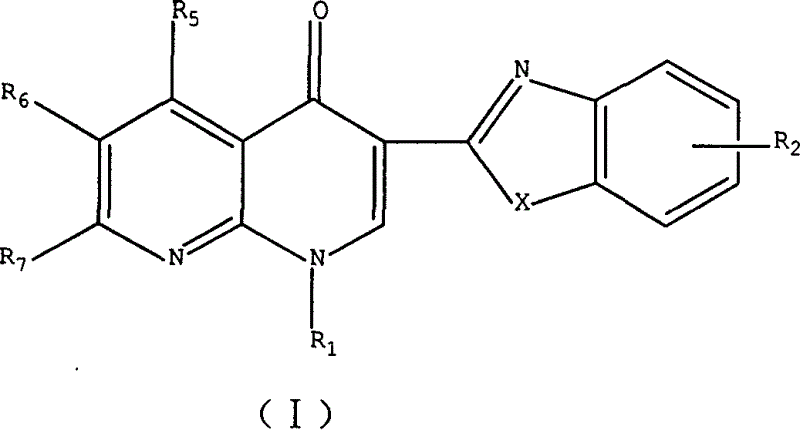
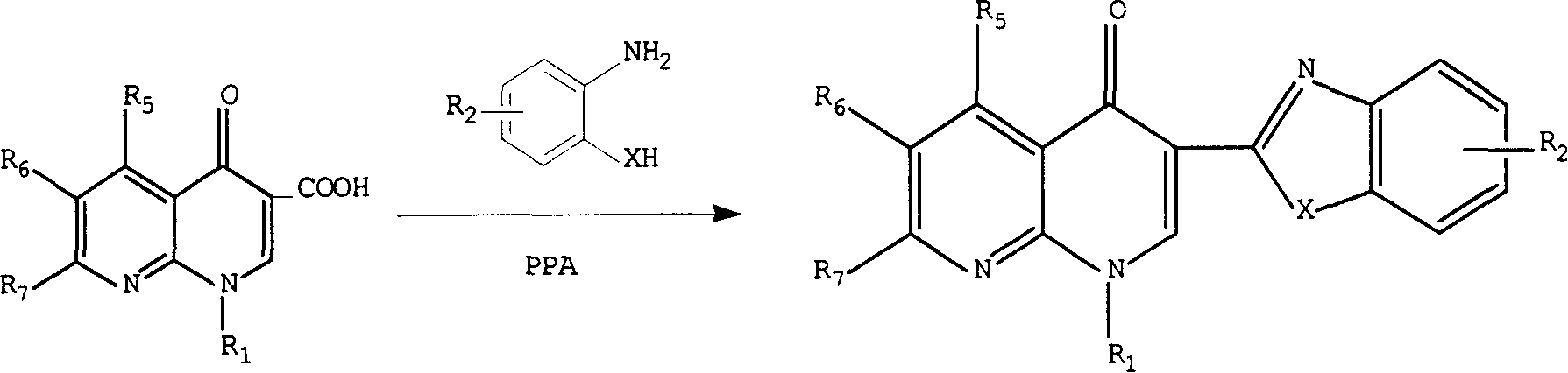
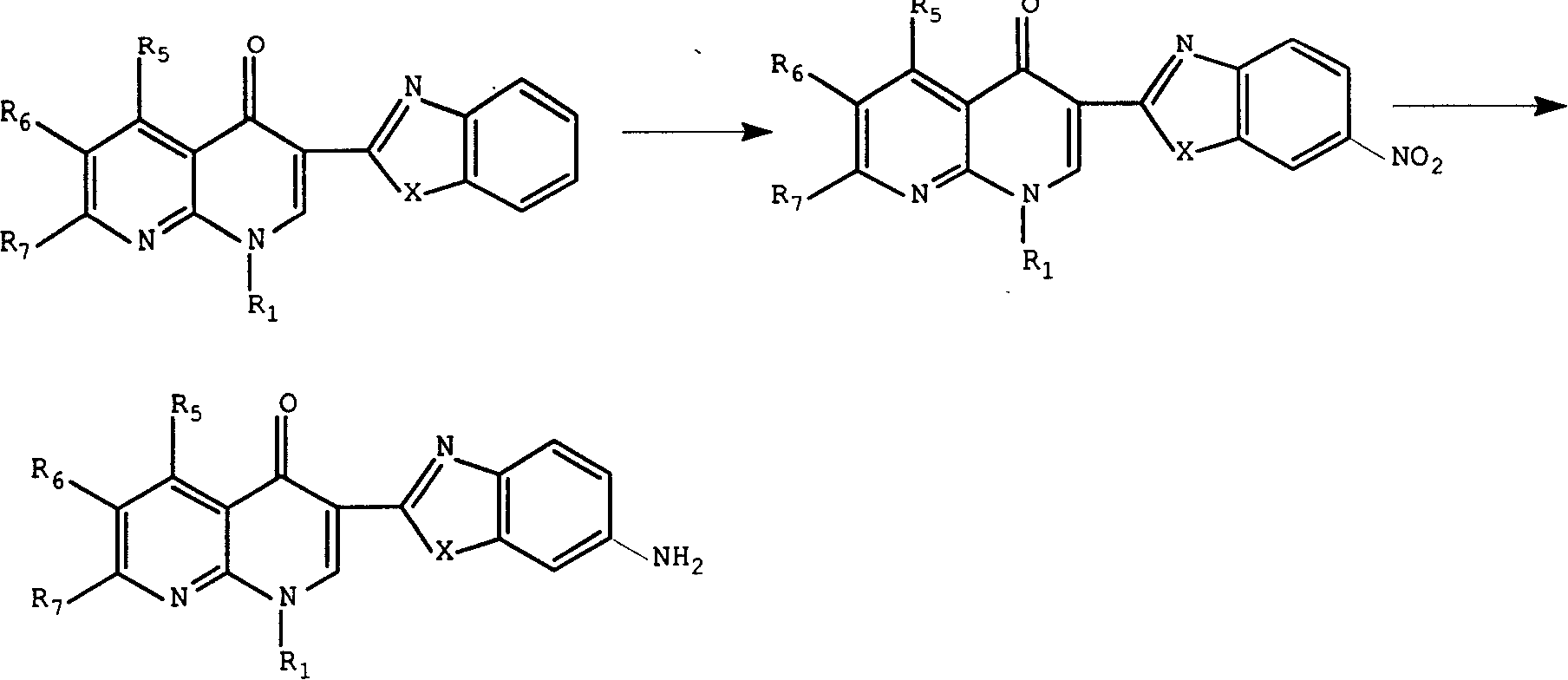
3-substituted nalidixic acid analog compound and its preparation method and uses in pharmacy
InactiveCN1733764AGood antitumor activityEasy to prepareOrganic active ingredientsOrganic chemistryNalidixic acidKetone
Disclosed is a 3-substituted nalidixic acid analog compound and its preparation method and uses in pharmacy, wherein the compound has antineoplastic activity and a general formula (I) with the 3-position substituted by benzimidazole and benzothiazole, the preparation process of the compound comprises condensing various substituted naphthyridine ketone-3-formylic acid, o-phenylenediamine, o-aminophenol, o-aminobenzenethiol and into polyphosphoric acids. The compound can be used in preparing anti-cancer medicaments.
Owner:CHINA PHARM UNIV
Benzoxazine monomer containing ethynyl groups and norbornene and preparation method and application of benzoxazine monomer
ActiveCN109293648ALow curing temperatureReduce processing temperatureOrganic chemistryPolymer scienceNorbornene
The invention belongs to the technical field of thermosetting resin, and discloses benzoxazine monomer containing ethynyl groups and norbornene and a preparation method and application of the benzoxazine monomer. According to the preparation method, ortho-norbornene-functionalized phenol is firstly synthesized by using norbornene dianhydride and o-aminophenol, and then the benzoxazine monomer containing ethynyl groups and norbornene is prepared by using the ortho-norbornene-functionalized phenol, ethynyl-containing aniline and paraformaldehyde in a low-polarity solvent. In addition to curablecross-linking of oxazine rings, curing cross-linking can also be performed on carbon-carbon double bonds and ethynyl groups in norbornene groups; the curing temperature of the norbornene groups is reduced significantly through introduction of the ethynyl groups, and is reduced from 350 DEG C to 220 DEG C, so that the processability of the resin is improved; not only can a new variety be added to benzoxazine compounds, but also the thermodynamic properties of the resin material can be improved significantly by a multiple-crosslinked network system formed through curing of the resin; and the preparation method has the advantages of simpleness, high operability, high yield, low production cost and easy industrial production.
Owner:镇江利德尔复合材料有限公司
Method for synthesizing benzoxazole compound by using nitration by-products of aromatic hydrocarbon and application of benzoxazole compound
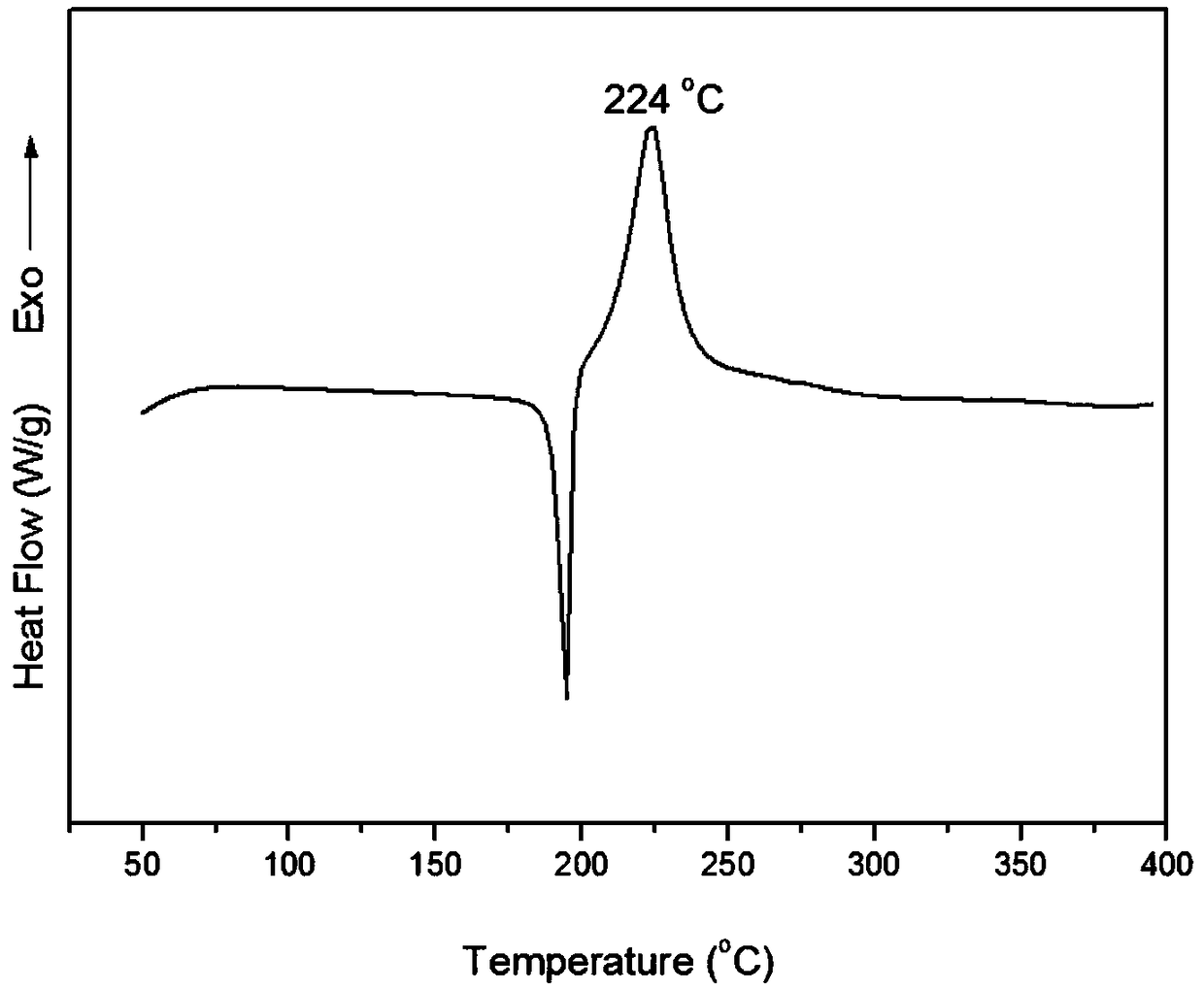
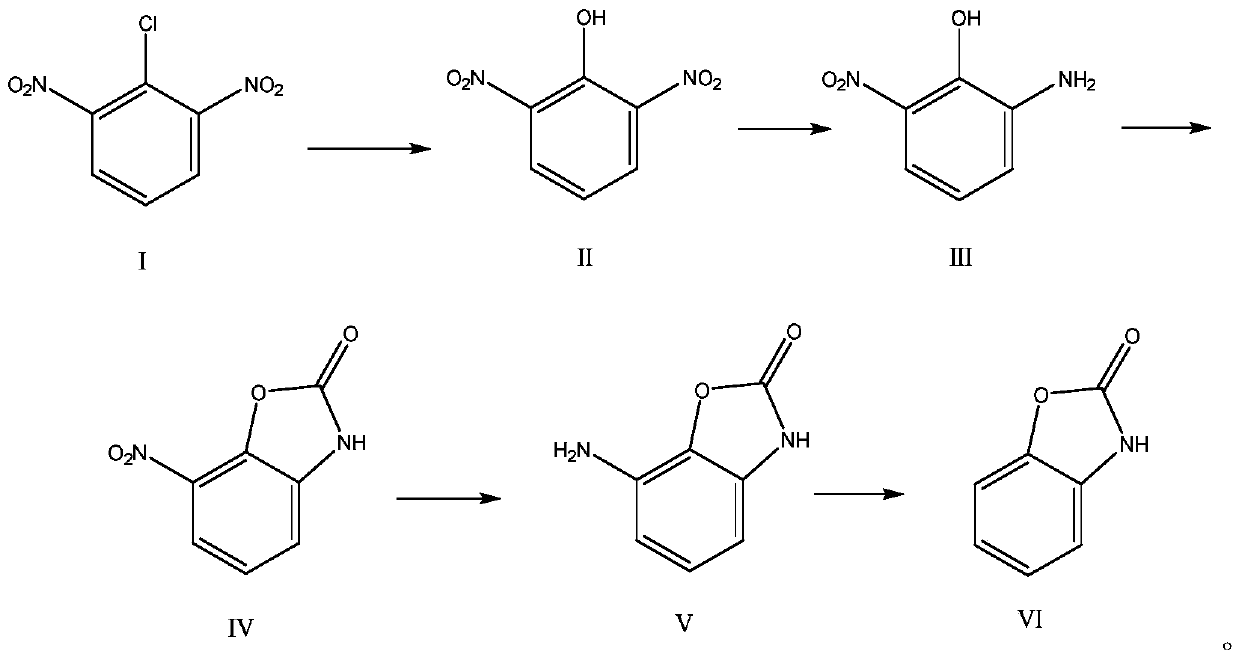
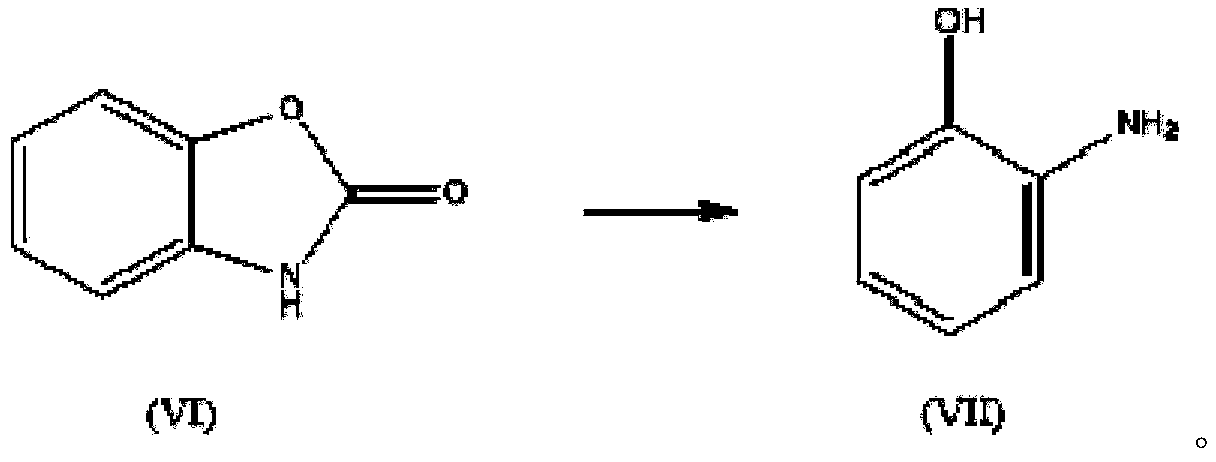
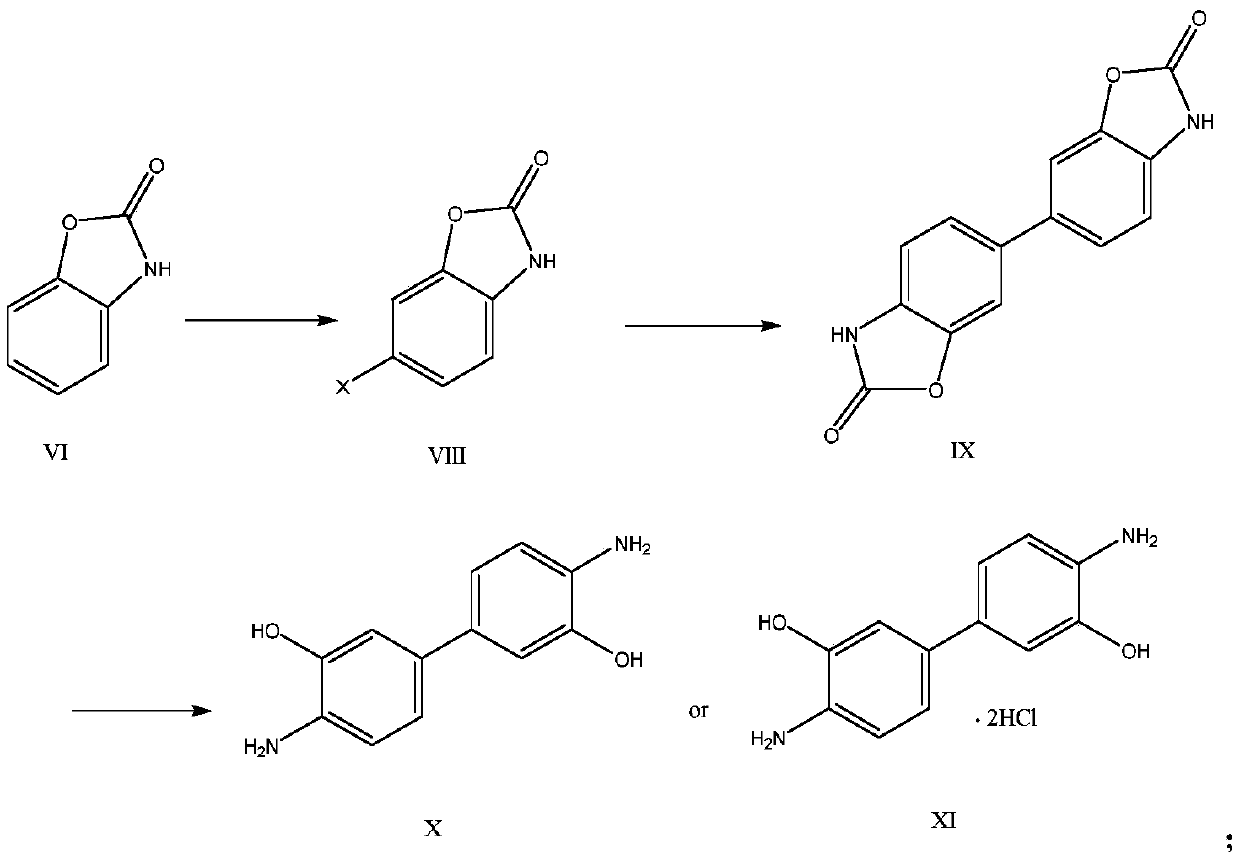
InactiveCN110156712AReduce Industrial Hazardous WasteImprove analysis efficiencyOrganic compound preparationAmino-hyroxy compound preparationBenzoxazoleNitration
The invention discloses a method for synthesizing a benzoxazole compound by using nitration by-products of aromatic hydrocarbon and application of the benzoxazole compound. The method comprises the main process: performing step-by-step crystallization on nitration products of a 2,4-dinitrochlorobenzene production enterprise so as to obtain 2,4-dinitrochlorobenzene, 2,6-dinitrochlorobenzene (I) anda small amount of residues, adopting the obtained 2,6-dinitrochlorobenzene (I) as the main starting material, and performing hydrolysis, selective catalytic hydrogenation reduction, cyclization, halogenation, carbon-carbon coupling and other processes so as to synthesize the benzoxazole compound, wherein the obtained compound can be used as a main raw material to synthesize a series of chemical intermediates with important application, and the chemical intermediates include o-aminophenol, 2-amino-4-nitrophenol, 2-amino-5-nitrophenol and hydrochloride of o-aminophenol, 2-amino-4-nitrophenol, 2-amino-5-nitrophenol. Through the method, the industrial by-products are converted into high value-added aromatic aminophenol products, industrial hazardous waste of the 2,4-dinitrochlorobenzene production enterprise is reduced, the scope of products of the enterprise is widened, and the economic benefits of enterprise are increased.
Owner:邵玉田
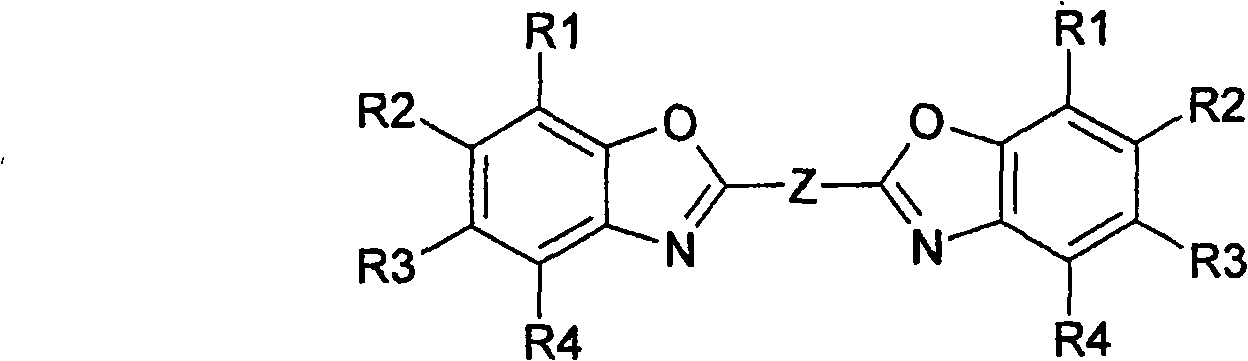
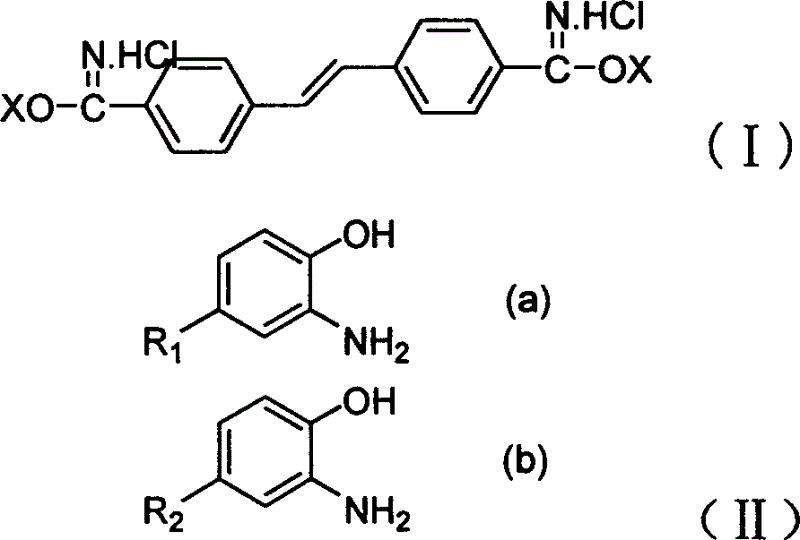
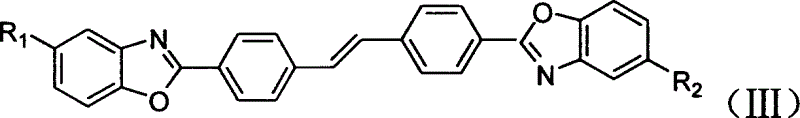
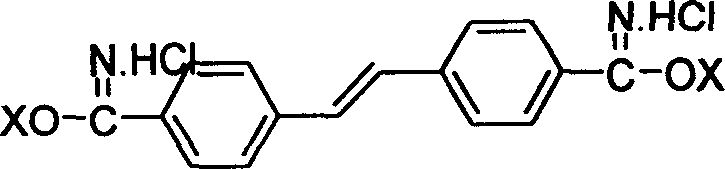
Fluorescent brightening agent and synthesis method of its mixture
A fluorescent brightening agent is prepared from distyrene-4,4'-biimine hydrochloride and o-aminophenol or its derivative through condensation and ring-closing reaction in the medium of organic acid and alcohol to obtain bibenzoxazyl distyrene compound.
Owner:SHENYANG RES INST OF CHEM IND +1
Method for treating o-nitrophenol production wastewater
InactiveCN110980864ANo secondary pollutionRecycling without distillationWater treatment parameter controlOrganic compound preparationO-nitrochlorobenzeneChlorobenzene
The invention belongs to the field of chemical wastewater treatment and particularly relates to a method for treating o-nitrophenol production wastewater. For wastewater produced after o-nitrochlorobenzene is hydrolyzed to prepare o-nitrophenol, ethanol is adopted as a desorbent in the method; and under the premise of utilizing existing process equipment and basis procedures, valuable o-nitrophenol can be effectively recovered without the need of toxic / expensive chemical agents, special treatment equipment or extra energy consumption. On this basis, the method can achieve effective utilizationof an o-nitrophenol recovery product, improve the overall process efficiency for preparing the o-aminophenol and the recovery efficiency of salts and other by-products, and avoid secondary pollution.The invention further provides a method for producing o-aminophenol related to the aforementioned method for treating wastewater.
Owner:HUBEI HONGXIN CHEM CO LTD
Binol-diform-o-aminophenol type Schiff base, and synthesis method and application thereof
ActiveCN107602413AStrong fluorescenceHigh fluorescence recognition performanceFluorescence/phosphorescenceLuminescent compositionsFluorescenceFiltration
The invention provides Binol-diform-o-aminophenol type Schiff base, and a synthesis method and an application thereof, and relates to a 2,2'-dihydroxy-1,1'-dinaphthalene-3,3'-diformaldehyde-o-aminophenol type Schiff base, and a synthesis method and an application thereof. The invention aims to solve a problem that Schiff base prepared from 2-hydroxy-1-naphthaldehyde-o-aminophenol in a plane structure has no recognition capability on metals. The structure formula of the 2,2'-dihydroxy-1,1'-dinaphthalene-3,3'-diformaldehyde-o-aminophenol type Schiff base is represented as the description. The method includes the steps of: 1) preparing a crude product of the 2,2'-dihydroxy-1,1'-dinaphthalene-3,3'-diformaldehyde-o-aminophenol type Schiff base; and 2) concentrating the crude product, dissolvingthe crude product in methanol, performing suction filtration, and washing and drying the product to obtain a pure product. Compared with analogues having plane structures, the synthesized Schiff basehas significantly enhanced fluorescent recognition capability. The compound can be used for detecting cerium ions in trace amount.
Owner:HARBIN UNIV OF SCI & TECH
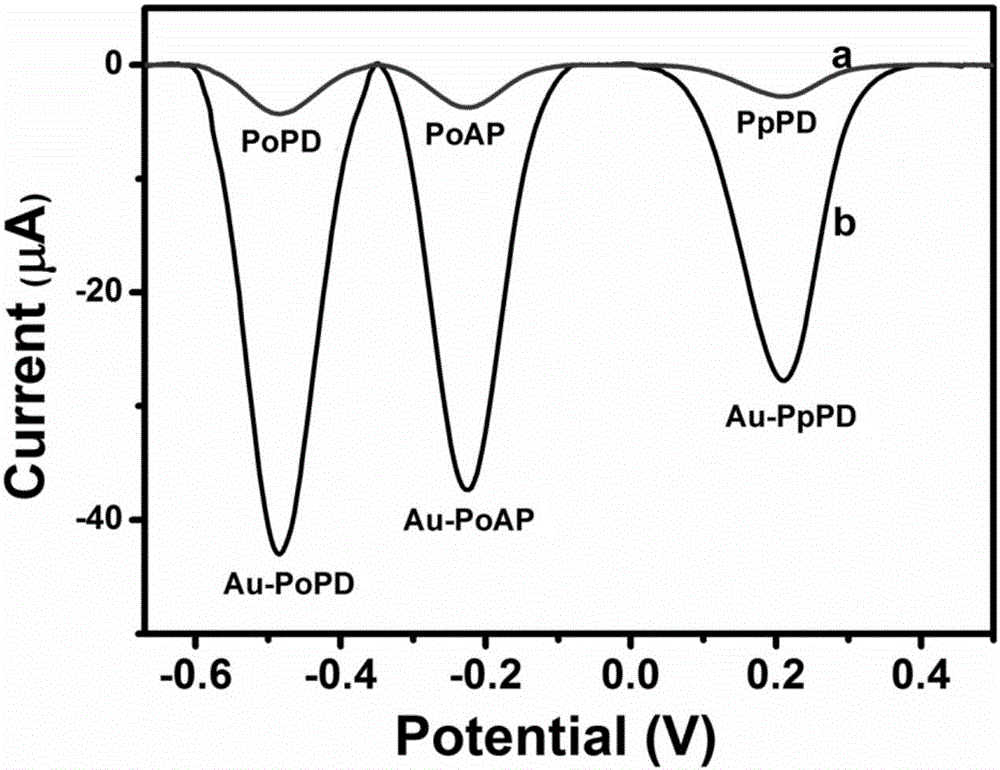
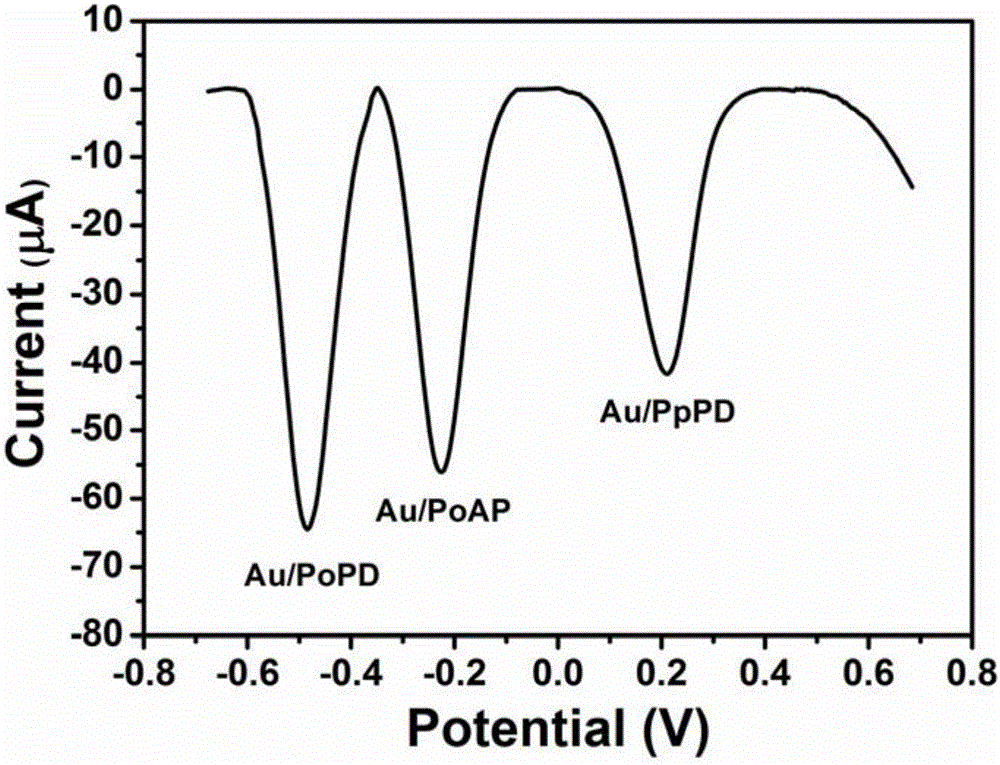
Gold/poly(o-aminophenol), gold/poly(p-phenylenediamine) and gold/poly(o-phenylenediamine) nanoparticles as well as preparation method and application thereof
InactiveCN105036069AResolved electrochemical signalAchieving Simultaneous DetectionMaterial nanotechnologyIndividual molecule manipulationComposite nanoparticlesP-Phenylenediamine
The invention discloses gold / poly(o-aminophenol), gold / poly(p-phenylenediamine) and gold / poly(o-phenylenediamine) nanoparticles as well as a preparation method and an application thereof. O-aminophenol, p-phenylenediamine and o-phenylenediamine are oxidized and polymerized by use of chloroauric acid respectively, to prepare a novel conductive composite nanomaterial having electrochemical activity; the preparation method of the composite nanomaterial is mild and easily controllable in preparation conditions, and simple and fast in preparation process; the gold / poly(zero-aminophenol), gold / poly(p-phenylenediamine) and gold / poly(o-phenylenediamine) composite nanoparticles possess single and differentiable electrochemical signals respectively, are high in electrical conductivity and large in specific surface area, and contain functional groups such as an amino group and an imino group; the composite nanoparticles can be taken as a good electrochemical sensing material and have wide application prospect in the field of electrochemical detection and analysis.
Owner:CAPITAL NORMAL UNIVERSITY
Poly-o-aminophenol antibacterial material and preparation method thereof
The invention discloses a preparation method of a poly-o-aminophenol antibacterial material. According to the preparation method, with o-aminophenol as a raw material, the poly-o-aminophenol high polymer antibacterial material is synthesized by carrying out chemical oxidation on the raw material in an acid solution; and the poly-o-aminophenol antibacterial material has the microparticle diameter of 1-50mu m and has chrysanthemum-like and spherical microstructure; and the poly-o-aminophenol antibacterial material prepared by using the preparation method has excellent bactericidal effects on escherichia coli, staphylococcus aureus, bacillus subtilis, bacillus megatherium and bacillus cereus, good environmental suitability, excellent organic solubility, heat stability and favorable conductivity, is controllable in size and morphology, low in cost, simple in preparation process and easy to industrialize and can be widely applied to the fields, such as medicines, agriculture and sanitation.
Owner:KUNMING UNIV OF SCI & TECH
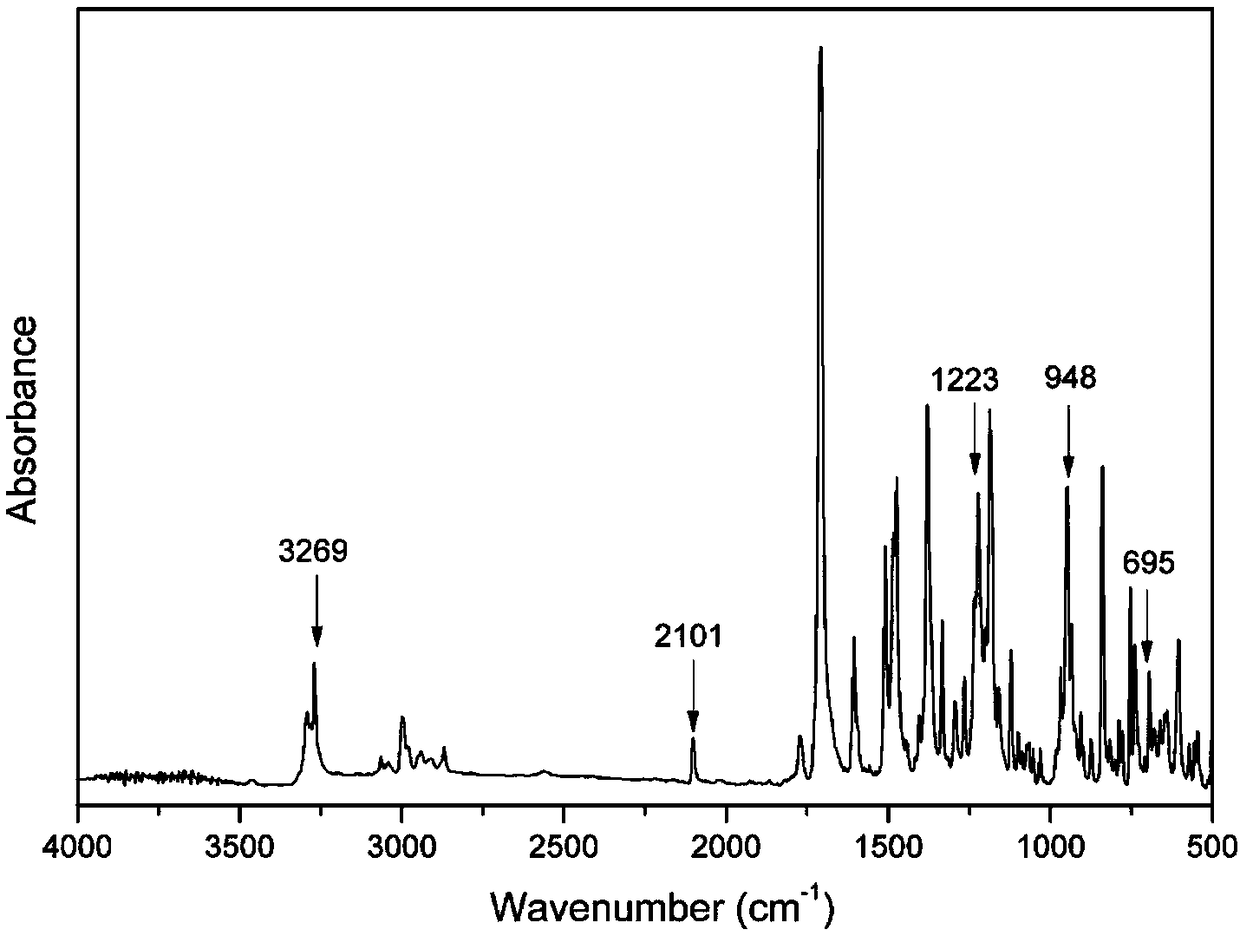
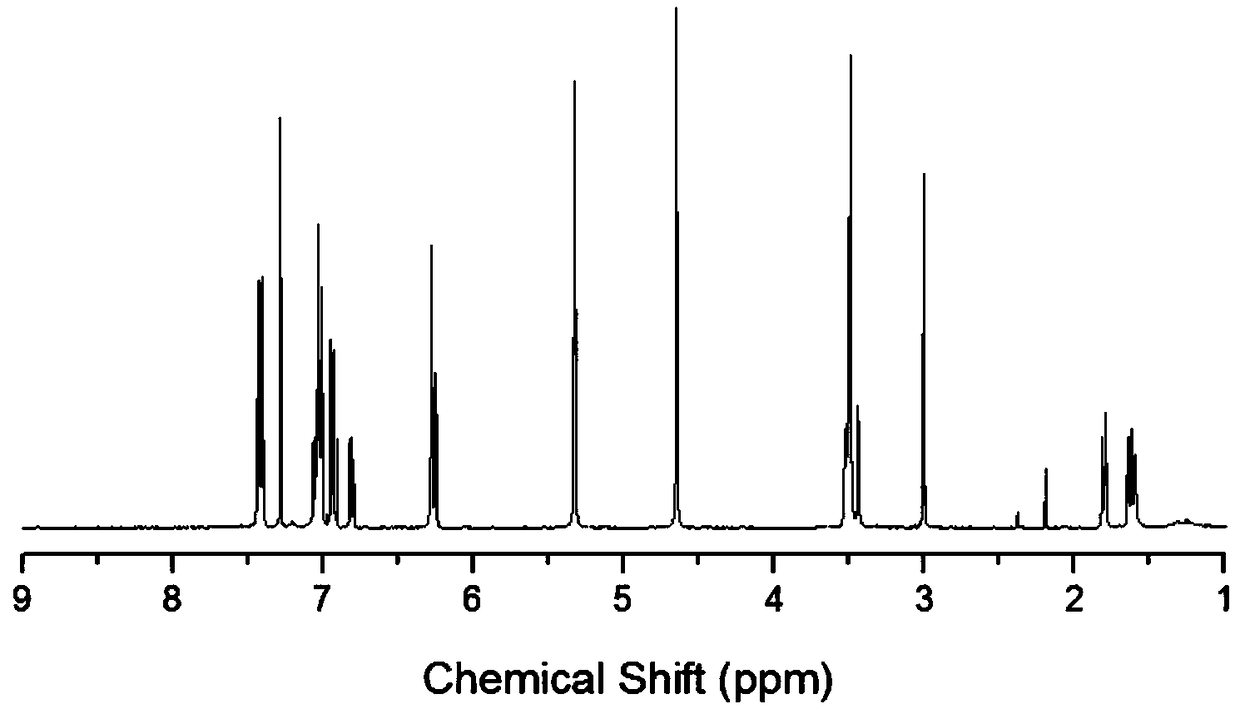
5-aryl-5H-dibenzo[b,f] azepine-10(11H) ketone compounds and preparation method thereof
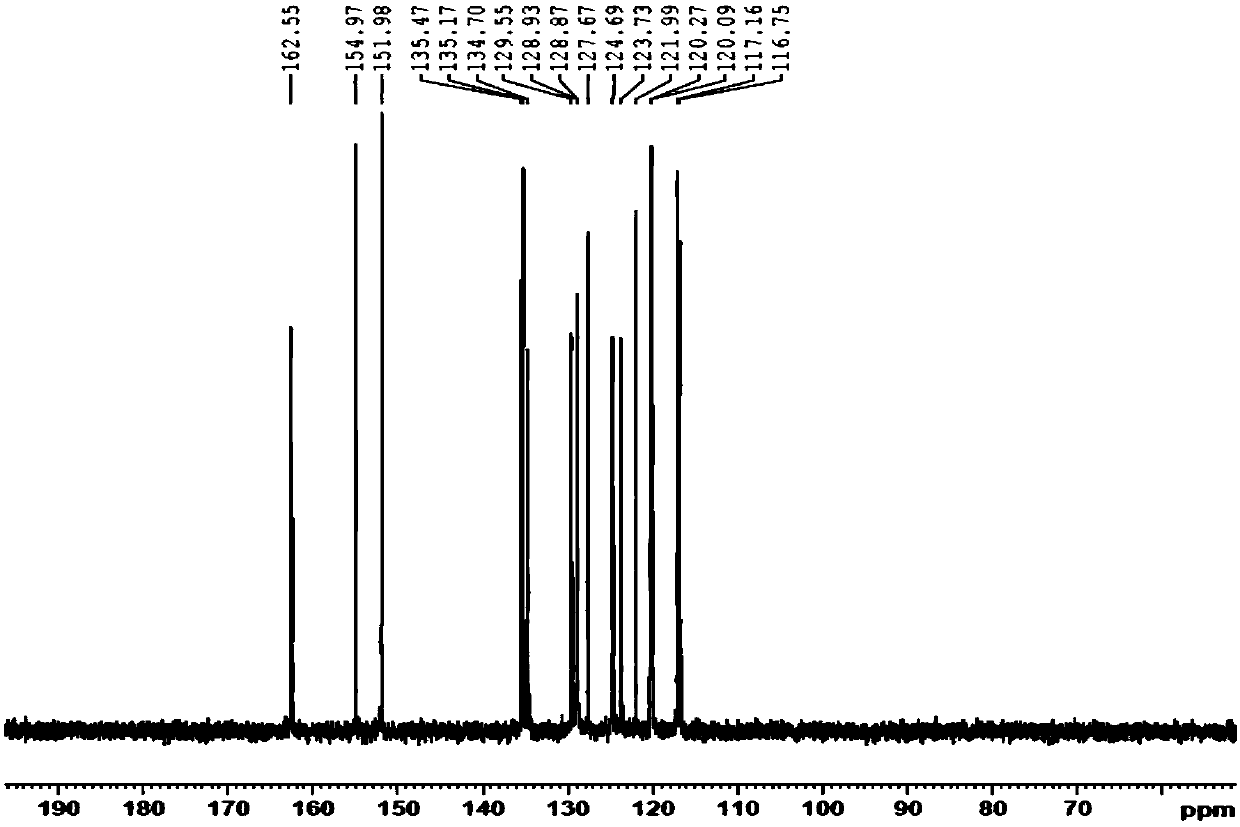
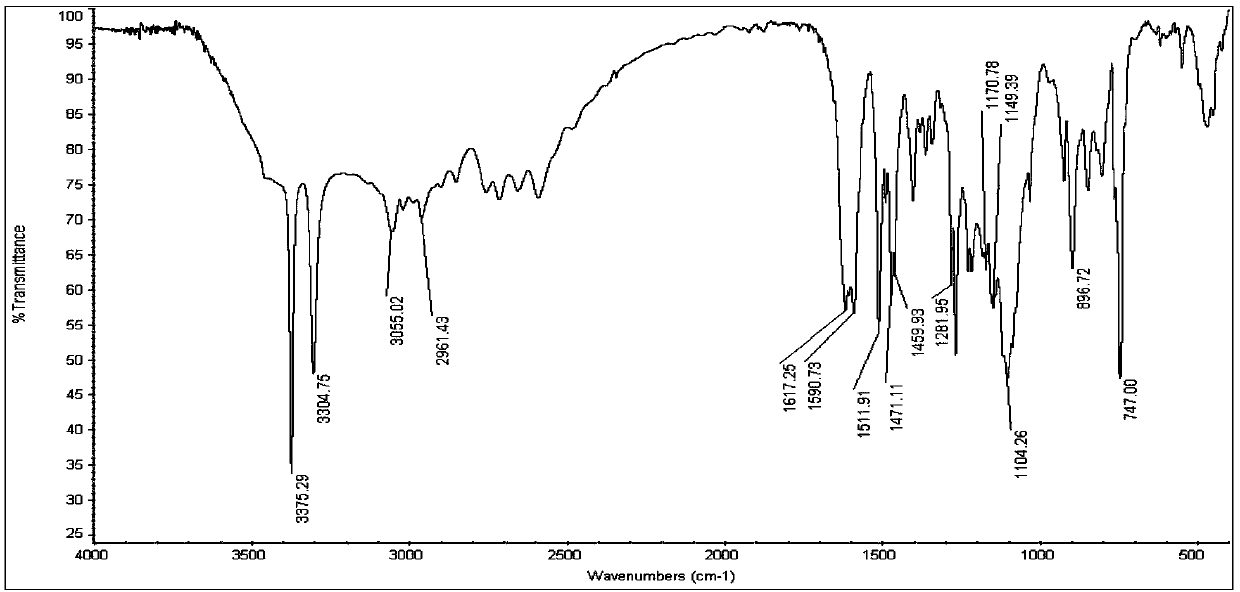
The invention discloses 5-aryl-5H-dibenzo[b,f]azepine-10(11H) ketone compounds and a preparation method thereof. The preparation method comprises the following steps: reacting N-aryl-o-aminophenol compounds with o-bromophenylacetic acid compounds in the presence of an alkali, a catalyst and microwave to obtain a triarylamine intermediate substituted by carboxyl and hydroxyl; performing intramolecular acylation reaction and cyclization of the triarylamine intermediate in the presence of a catalyst to obtain the 5-aryl-5H-dibenzo[b,f] azepine-10(11H) ketone compounds. The method disclosed by the invention has the advantages of convenient and simple operation, short reaction time and convenient post treatment; and the ketone compounds are conveniently prepared by using simple raw materials, and therefore sustainable development and utilization can be achieved.
Owner:GUANGZHOU CHEM CO LTD CHINESE ACADEMY OF SCI



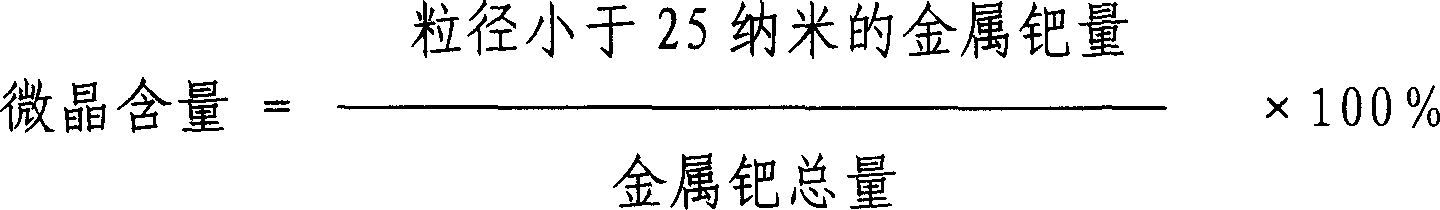
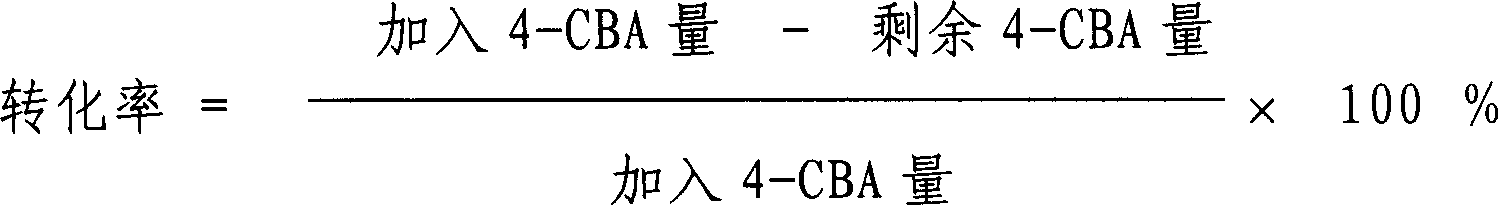
![Method for preparing single-molecular magnet [Dy2(saph)2(NO3)2(CH3OH)4] Method for preparing single-molecular magnet [Dy2(saph)2(NO3)2(CH3OH)4]](https://images-eureka.patsnap.com/patent_img/57537909-c307-494d-836f-821538d86323/HDA0000634118620000011.PNG)
![Method for preparing single-molecular magnet [Dy2(saph)2(NO3)2(CH3OH)4] Method for preparing single-molecular magnet [Dy2(saph)2(NO3)2(CH3OH)4]](https://images-eureka.patsnap.com/patent_img/57537909-c307-494d-836f-821538d86323/HDA0000634118620000012.PNG)
![Method for preparing single-molecular magnet [Dy2(saph)2(NO3)2(CH3OH)4] Method for preparing single-molecular magnet [Dy2(saph)2(NO3)2(CH3OH)4]](https://images-eureka.patsnap.com/patent_img/57537909-c307-494d-836f-821538d86323/HDA0000634118620000021.PNG)


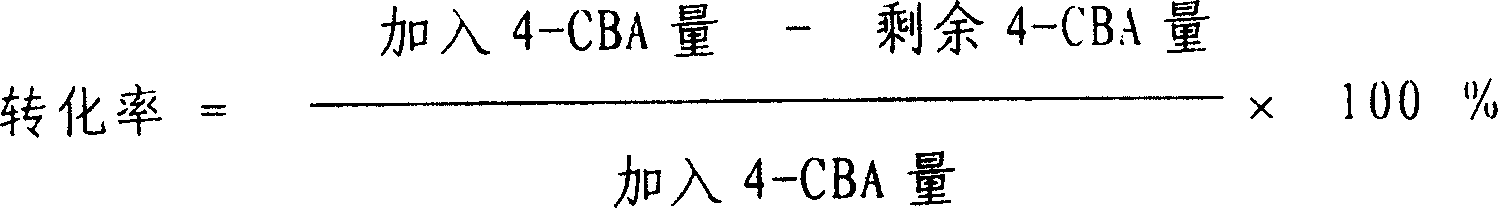

![Preparation method of single-molecular magnet [Dy2(saph)2Cl2].4CH3OH Preparation method of single-molecular magnet [Dy2(saph)2Cl2].4CH3OH](https://images-eureka.patsnap.com/patent_img/692108a6-82a0-447f-bbdc-cab60749ad61/HDA0000447506500000011.PNG)
![Preparation method of single-molecular magnet [Dy2(saph)2Cl2].4CH3OH Preparation method of single-molecular magnet [Dy2(saph)2Cl2].4CH3OH](https://images-eureka.patsnap.com/patent_img/692108a6-82a0-447f-bbdc-cab60749ad61/HDA0000447506500000012.PNG)
![Preparation method of single-molecular magnet [Dy2(saph)2Cl2].4CH3OH Preparation method of single-molecular magnet [Dy2(saph)2Cl2].4CH3OH](https://images-eureka.patsnap.com/patent_img/692108a6-82a0-447f-bbdc-cab60749ad61/HDA0000447506500000021.PNG)


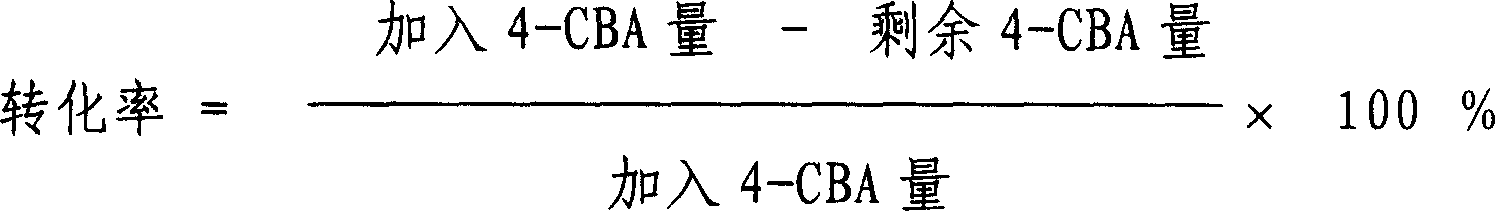
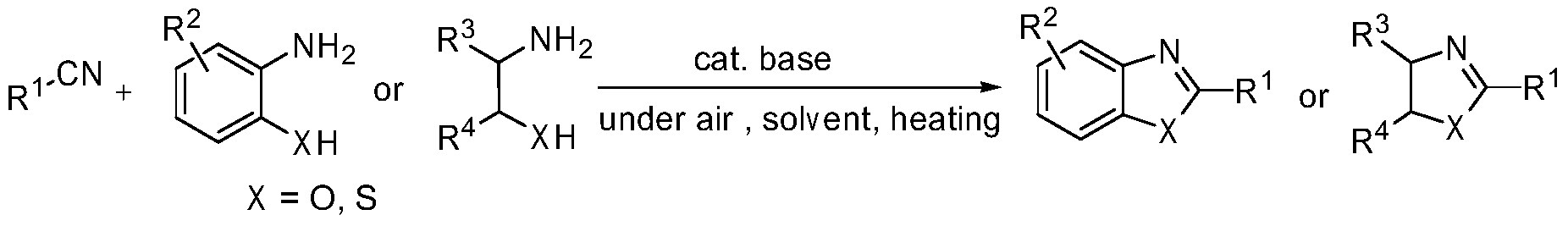
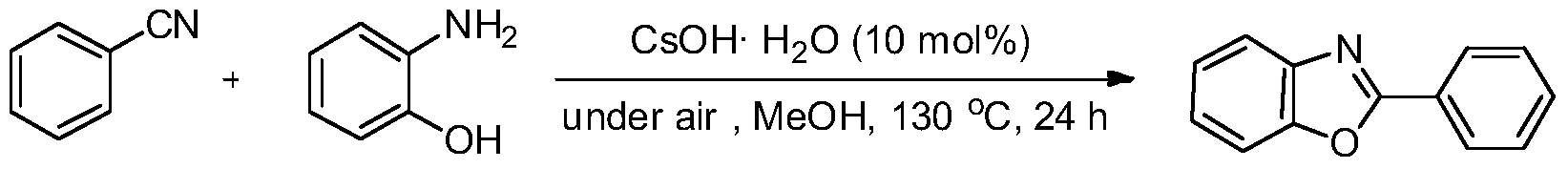
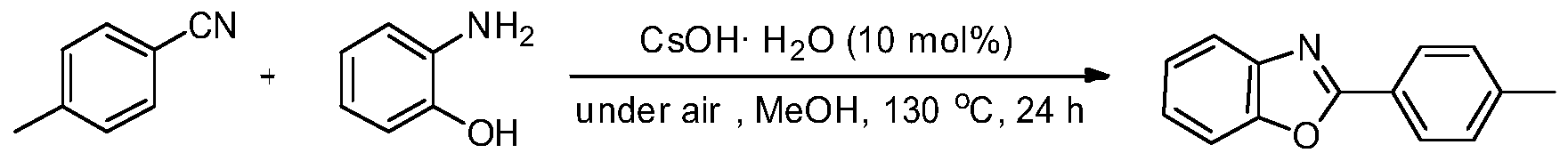
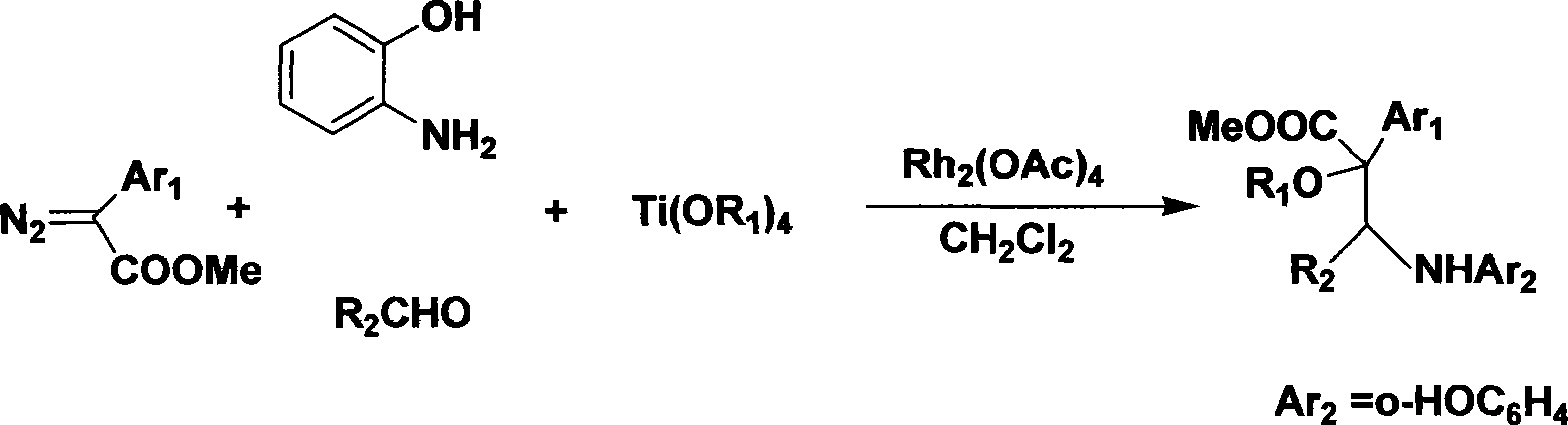





![5-aryl-5H-dibenzo[b,f] azepine-10(11H) ketone compounds and preparation method thereof 5-aryl-5H-dibenzo[b,f] azepine-10(11H) ketone compounds and preparation method thereof](https://images-eureka.patsnap.com/patent_img/f9637518-4801-434e-9304-240db46d8ef6/BDA0000606982660000011.PNG)
![5-aryl-5H-dibenzo[b,f] azepine-10(11H) ketone compounds and preparation method thereof 5-aryl-5H-dibenzo[b,f] azepine-10(11H) ketone compounds and preparation method thereof](https://images-eureka.patsnap.com/patent_img/f9637518-4801-434e-9304-240db46d8ef6/BDA0000606982660000021.PNG)
![5-aryl-5H-dibenzo[b,f] azepine-10(11H) ketone compounds and preparation method thereof 5-aryl-5H-dibenzo[b,f] azepine-10(11H) ketone compounds and preparation method thereof](https://images-eureka.patsnap.com/patent_img/f9637518-4801-434e-9304-240db46d8ef6/BDA0000606982660000022.PNG)