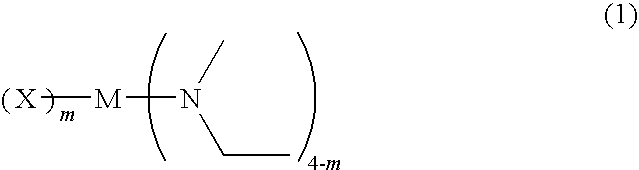Patents
Literature
55results about How to "Stable deposition rate" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
Electroless gold plating bath, electroless gold plating method and electronic parts
ActiveUS20080138507A1Stable deposition rateGood lookingAnti-corrosive paintsLiquid/solution decomposition chemical coatingWater solubleElectroless plating
An electroless gold plating bath includes a water-soluble gold compound, a completing agent, an aldehyde compound, and an amine compound represented by R1—NH—C2H4—NH—R2 or (CH2—NH—C2H4—NH—CH2)n—R4 (wherein R1 to R4 represent —OH, —CH3, —CH2OH, —C2H4OH, —CH2N(CH3)2, —CH2NH(CH2OH), CH2NH(C2H4OH), —C2H4NH(CH2OH), —C2H4NH(C2H4OH), —CH2N(CH2OH)2, —CH2N(C2H4OH)2, —C2H4N(CH2OH)2 or —C2H4N(C2H4OH)2, and n is an integer of 1 to 4).The electroless gold plating can be carried out without corrosion of an underlying metal to be plated at a stable deposition rate. Because of the high deposition rate and the immersion and reduction types, thickening of a plated coating is possible in one solution and the color of the coating is not degraded to provide a good appearance while keeping a lemon yellow color inherent to gold.
Owner:C UYEMURA & CO LTD
Method for preparing tungsten trioxide (WO3) electrochromic films
InactiveCN103246119AUniform depositionUniform dense depositionNon-linear opticsRadio frequency magnetron sputteringIndium tin oxide
WO3 films have the advantages of being large in visible light transmissivity adjusting range, high in coloration efficiency, short in response time, good in circulation stability, friendly to environment and the like. Electrochromic devices based on WO3 films can be widely used on buildings, automobiles and the like to serve as energy-saving smart windows. The invention relates to a WO3 photoelectric functional material and provides a method for preparing WO3 electrochromic films which are good in film quality, uniform in thickness and short in color-changing response time. According to the method, WO3 serves as a target, and electrochromic WO3 films are prepared on transparent conductive indium tin oxide (ITO) glass substrates through a radio frequency magnetron sputtering method. The method is simple in preparation process, easy to control, good in repeatability, stable in deposition rate and capable of being produced on a large scale conveniently.
Owner:NANJING UNIV OF SCI & TECH
Electroless tin plating formula for printed circuit board
ActiveCN106939417AComplexation effectiveStrong complexationLiquid/solution decomposition chemical coatingThioureaTin plating
The invention discloses an electroless tin plating formula for a printed circuit board. The formula comprises the following components with the following concentrations: 10-30g / L of stannous ions, 70-130g / L of thiourea, 30-70g / L of methanesulfonic acid, 30-60g / L of citric acid, 20-50g / L of tetrasodium iminodisuccinate, 10-30g / L of carbohydrazide, 10-50mg / L of interfacial agent, 0.05-0.15g / L of bismuth acetate and 20-40g / L of polyamino polyether methylene phosphonate, wherein the component solutions are uniformly mixed and then prepared into an electroless tin plating solution; and the electroless tin plating solution has the following characteristics: tin is stable in tin deposition rate, bivalent tin is not liable to be oxidized into tetravalent tin, and the solution is high in stability. The formula disclosed by the invention contains many complexing agents, the stannous ions are not liable to be oxidized into tetravalent tin, the oxidization of oxygen dissolved in the plating solution can be cancelled by virtue of a reducing agent, the plating solution contains bismuth acetate, a plating layer is a tin-bismuth alloy, and the alloyed plating layer is not liable to grow tin whiskers; and moreover, the electroless tin plating solution has the following characteristics: tin is stable in tin deposition rate, bivalent tin is not liable to be oxidized into tetravalent tin, and the solution is high in stability.
Owner:SHENZHEN CHENGGONG CHEM
Electroplating bath
ActiveCN103789806AReduce in quantityImprove uniformityTanksFinal product manufactureProduction lineIndium
The invention belongs to the technical field of preparation of solar cells, and in particular relates to an electroplating bath, which is used for depositing elemental copper, gallium, indium or selenium or a copper, gallium, indium or selenium alloy on the surface of a substrate. The electroplating bath comprises an electroplating branch bath and a master bath, wherein the electroplating branch bath is provided with an accommodation cavity for containing an electroplating solution; anodes immersed in the electroplating solution are hung on the lateral surfaces of the electroplating branch bath; a conveying device for carrying the substrate to synchronously move is arranged on the middle part of the electroplating branch bath; the anodes are arranged on the two sides of the conveying device; parts, opposite to the anodes, on the two lateral surfaces of the conveying device are immersed in the electroplating solution; the substrate is attached to the lateral surfaces of the conveying device, and is opposite to the anodes when passing through the electroplating branch bath. According to the electroplating bath, the length of an electroplating production line is reduced, and a small space is occupied; the elemental copper, gallium, indium or selenium or the copper, gallium, indium or selenium alloy is deposited on the front surface of the substrate only, so that few process steps are required; the anodes are opposite to the substrate, so that the uniformity and smooth finish of the surface of a deposited CIGS (copper, indium, gallium and selenium) absorption layer are ensured.
Owner:SOLTRIUM TECH LTD SHENZHEN
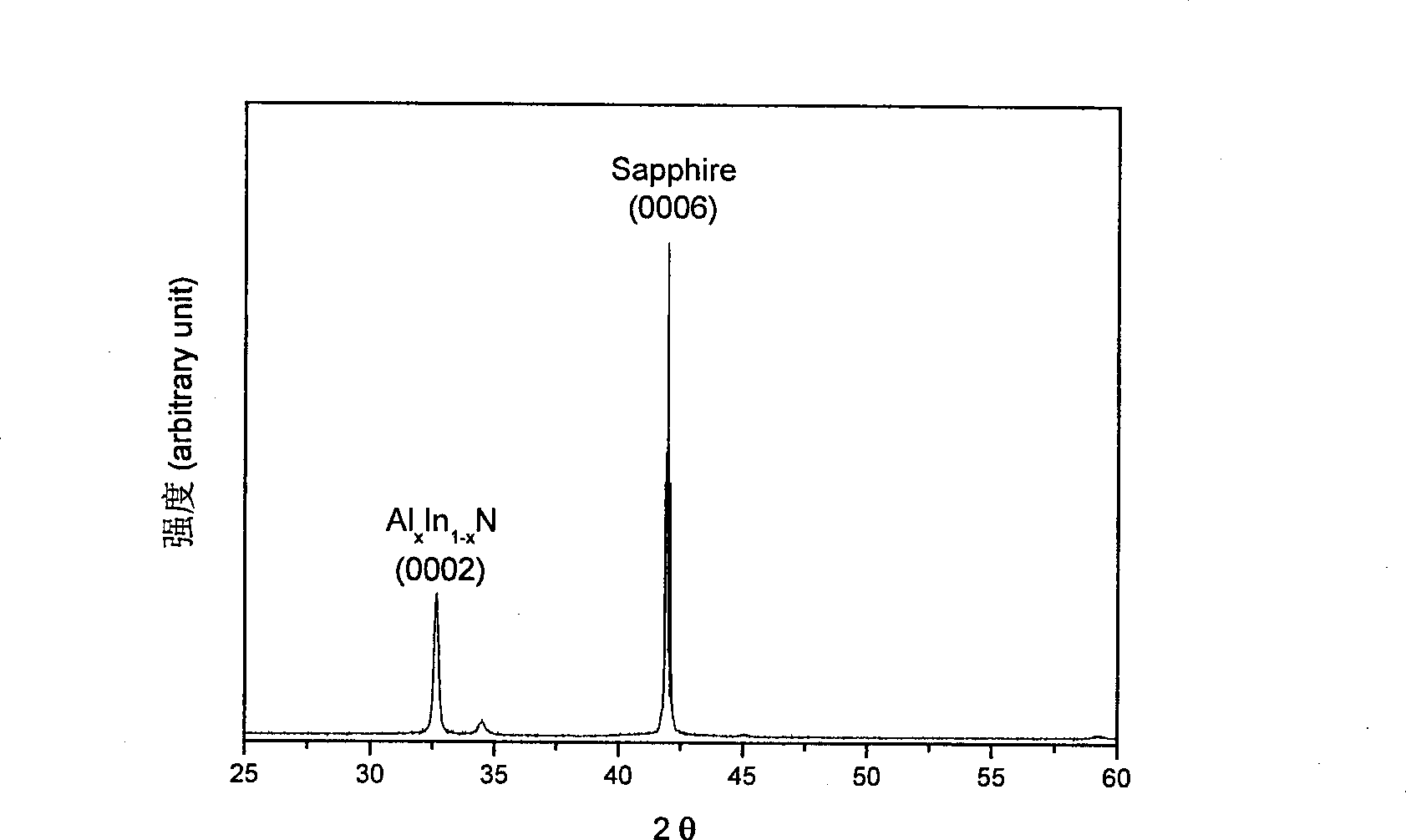
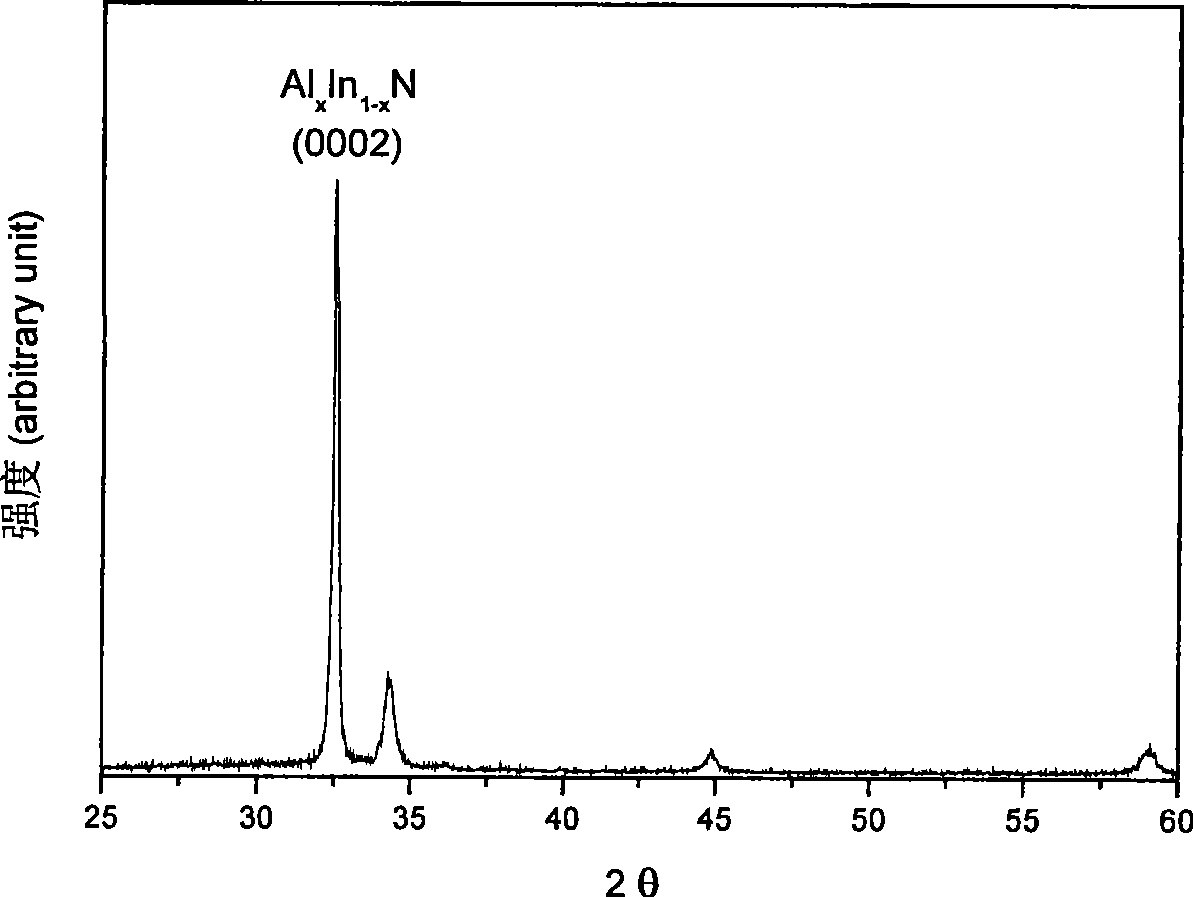
Method for preparing AlxIn1-xN film
InactiveCN101423927AReduce lattice mismatchAdequate responseVacuum evaporation coatingSputtering coatingSputteringNitrogen
The invention discloses a method for preparing AlxIn1-xN thin films. The method comprises the following steps: (1) a substrate is completely cleaned by washing at normal temperature and normal pressure and placed in an atmosphere of nitrogen to be dried; (2) the treated substrate is placed in a sputtering chamber, and a buffer layer AIN is grown on the substrate under a vacuum condition by a sputtering method which uses Al as a target material and carries out sputtering for 20 to 30 minutes under conditions of a flow ratio of N2 to Ar of 1 to 9, a DC sputtering power of between 50 to 60w and the temperature of the substrate during sputtering controlled between 400 to 500 DEG C; and (3) after growth of the buffer layer AIN, the target material is changed into Al-In(1:1) alloy and an AlxIn1-xN thin film is grown under a vacuum condition by a sputtering method which carries out sputtering for 10 to 30 minutes under conditions of a flow ratio of N2 to Ar of 3 to 1, a DC sputtering power of between 80 to 160w and the temperature of the substrate during sputtering controlled between 250 to 350 DEG C.
Owner:SICHUAN NORMAL UNIVERSITY
Process of mechanical copper plating and mechanical copper alloy plating
ActiveCN105112988APotential stabilityProcess stabilityElectrolysis componentsDual effectPolyethylene glycol
The invention discloses a process of mechanical copper plating and mechanical copper alloy plating and belongs to the technical field of surface plating of a steel product. The process of mechanical copper plating and mechanical copper alloy plating comprises the steps of adjusting the pH value of a plating solution by means of universal mechanical plating equipment, then adding copper powder or copper alloy powder and a sedimentation accelerating agent into the plating solution, and finally a cooper coating or a copper alloy coating is prepared on the surface of the steel product along with the rotation of a plating cylinder of the mechanical plating equipment. The sedimentation accelerating agent adopted by the process is composed of diatomite, sodium gluconate, triethanolamine, stannous salt, ammonium dihydrogen phosphate, polyethylene glycol, OP-10, inorganic acid and a defined amount of water. By adopting the process of mechanical copper plating and mechanical copper alloy plating, the copper powder or the copper alloy powder can subside on the surface of the steel product rapidly, the process is simple, the operation is convenient, and the dual-effect of corrosion prevention and decoration of the steel product can be achieved.
Owner:KUNMING UNIV OF SCI & TECH
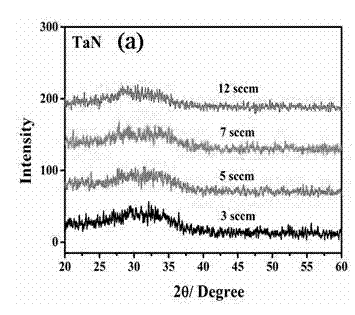
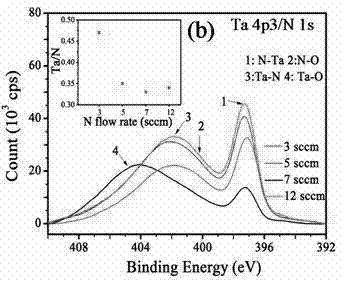
Method for preparing high thermal stability double layer diffusion impervious layer material
InactiveCN102392216APrevent purityAvoid pollutionVacuum evaporation coatingSputtering coatingDiffusionElectrical performance
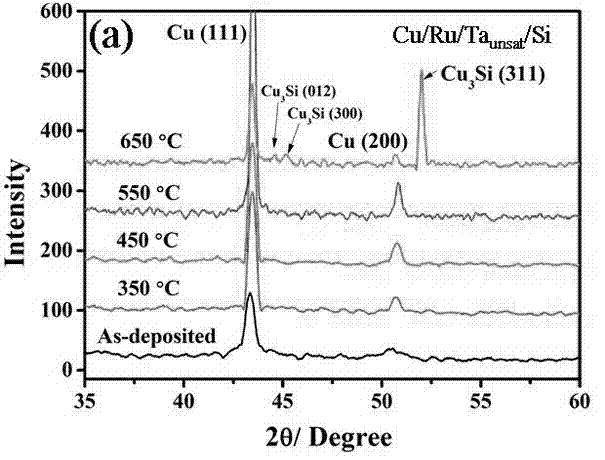
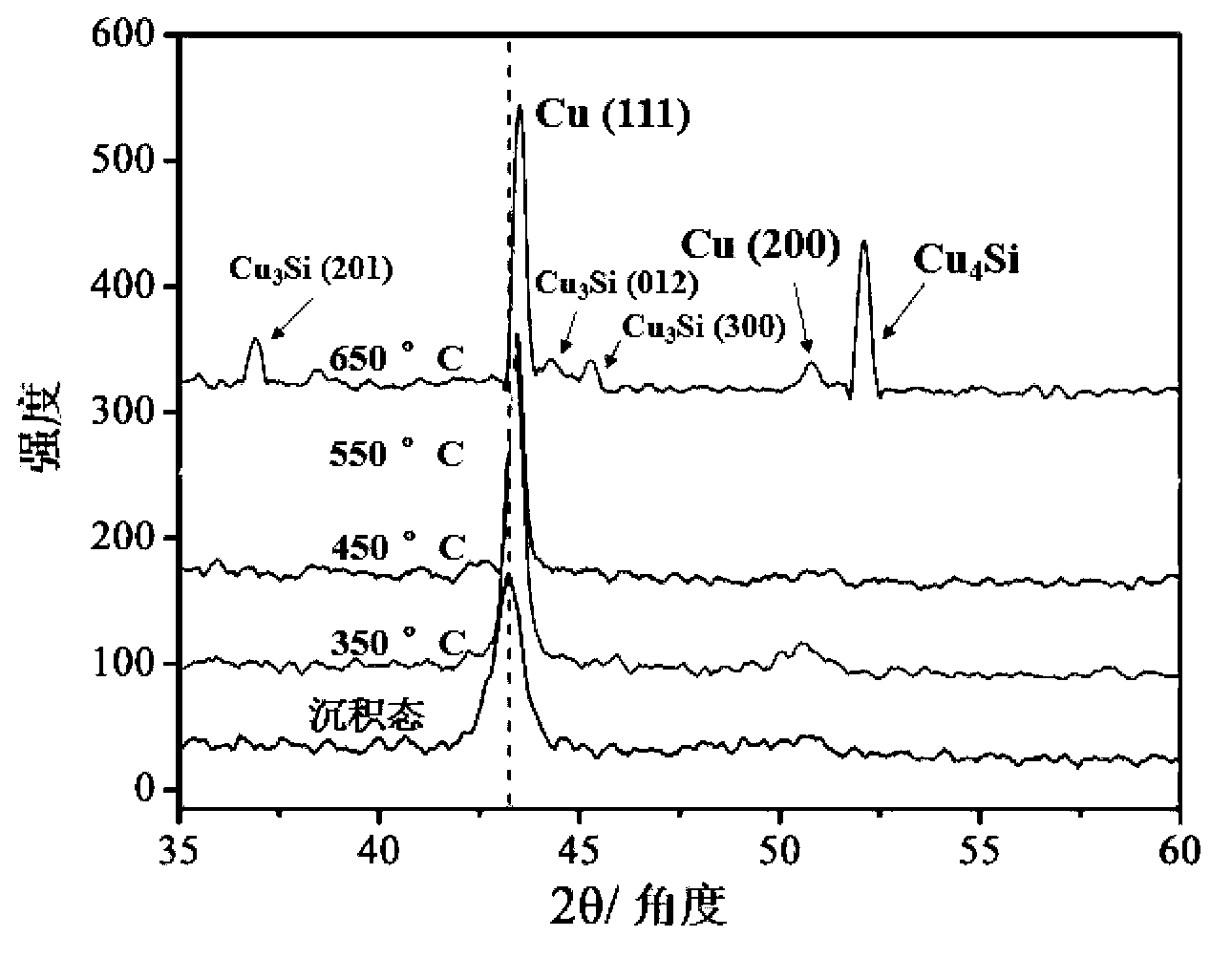
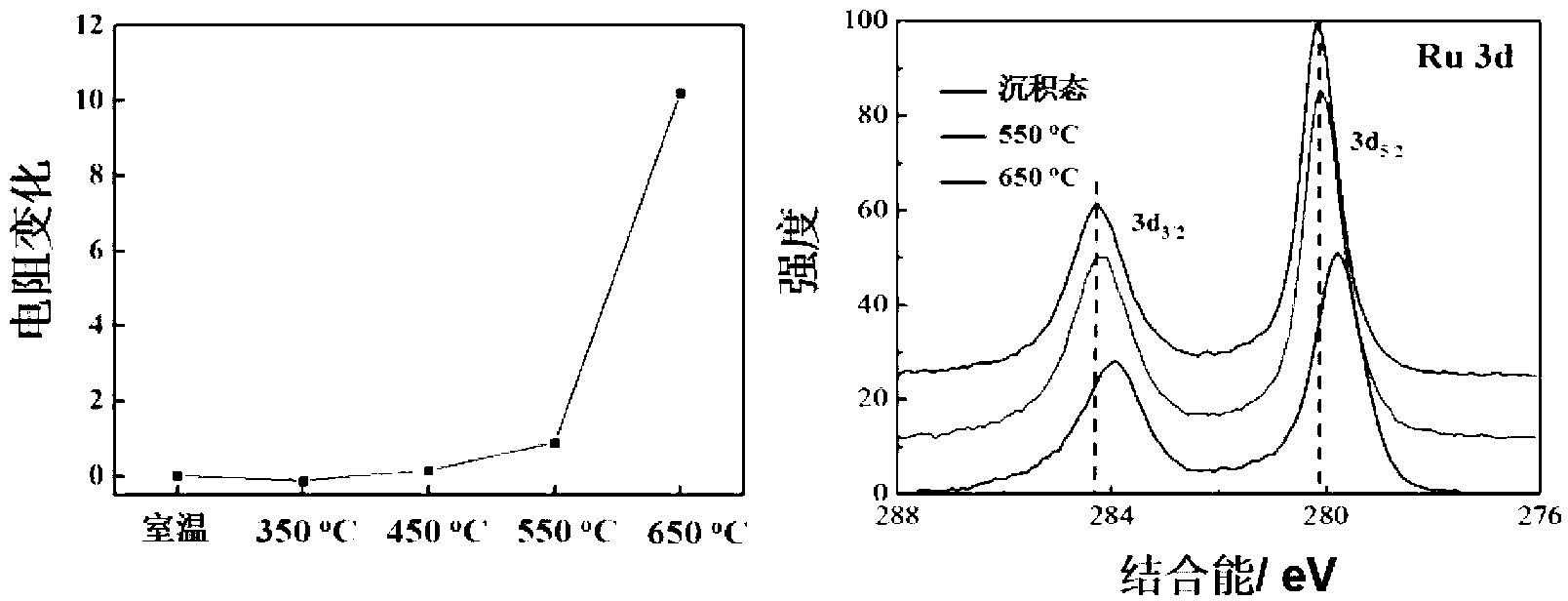
The invention provides a method for preparing a high thermal stability double layer diffusion impervious layer material. The method is characterized in that a direct current magnetron sputtering process is employed, accurate control of N content in an amorphous TaN film is realized by regulating N air flow rate, a high performance double layer Ru / TaN diffusion impervious layer structure with different N content is prepared, the effective work is stably as high as 650 DEG C. N atoms in gap of the TaN film are diffused during the annealing process and then raise the diffusion impervious performance of the Ru / TaN bilaminar membrane. The thermal stability double layer diffusion impervious layer material can be obtained by accurately controlling the N content, and is capable of keeping excellent electrical performance and better ensuring its practical application. The invention has the advantages of simple operation, good repeatability and good realization effect.
Owner:NANJING UNIV
Method for preparing transition metal-doped ZnO nanowire through hydrothermal method
InactiveCN106186040AStable deposition rateFilm thickness is easy to controlZinc oxides/hydroxidesNanotechnologyZno nanowiresSol-gel
The invention discloses a method for preparing a transition metal-doped ZnO nanowire through a hydrothermal method. The method comprises the steps that quartz is adopted as a substrate, and ZnO seed crystals are prepared through a magnetron sputtering method or a sol-gel method; a ZnO target is adopted as a target material; a ZnO nano-array grows by adopting the hydrothermal method; a growth solution is prepared from an original solution and a catalyst, wherein a zinc salt and transition metal salt aqueous solution with the concentration of 0.04-0.08 mol / L is taken as the original solution, hexamethylenetetramine is taken as the catalyst, 50 ml of the growth solution is taken to be added into a sealed reaction kettle, the quartz substrate with the prepared ZnO seed crystals is perpendicularly suspended in the sealed reaction kettle containing the growth solution, and then the reaction kettle is placed in an oven; after 3-5 h, growth is finished, and the reaction kettle is taken out; the reaction kettle is naturally cooled to room temperature, washed and blow-dried, and then the transition metal-doped ZnO nanowire is obtained. The transition metal-doped ZnO nanowire is stable in deposition rate, capable of easily controlling the membrane thickness, good in repeatability and high in adhesion strength with the substrate, has the high transmittance and good electrical property, is low in cost and safe in operation and is applied to the fields of gas-sensitive materials, sensors and optical detectors and the like.
Owner:YANAN UNIV
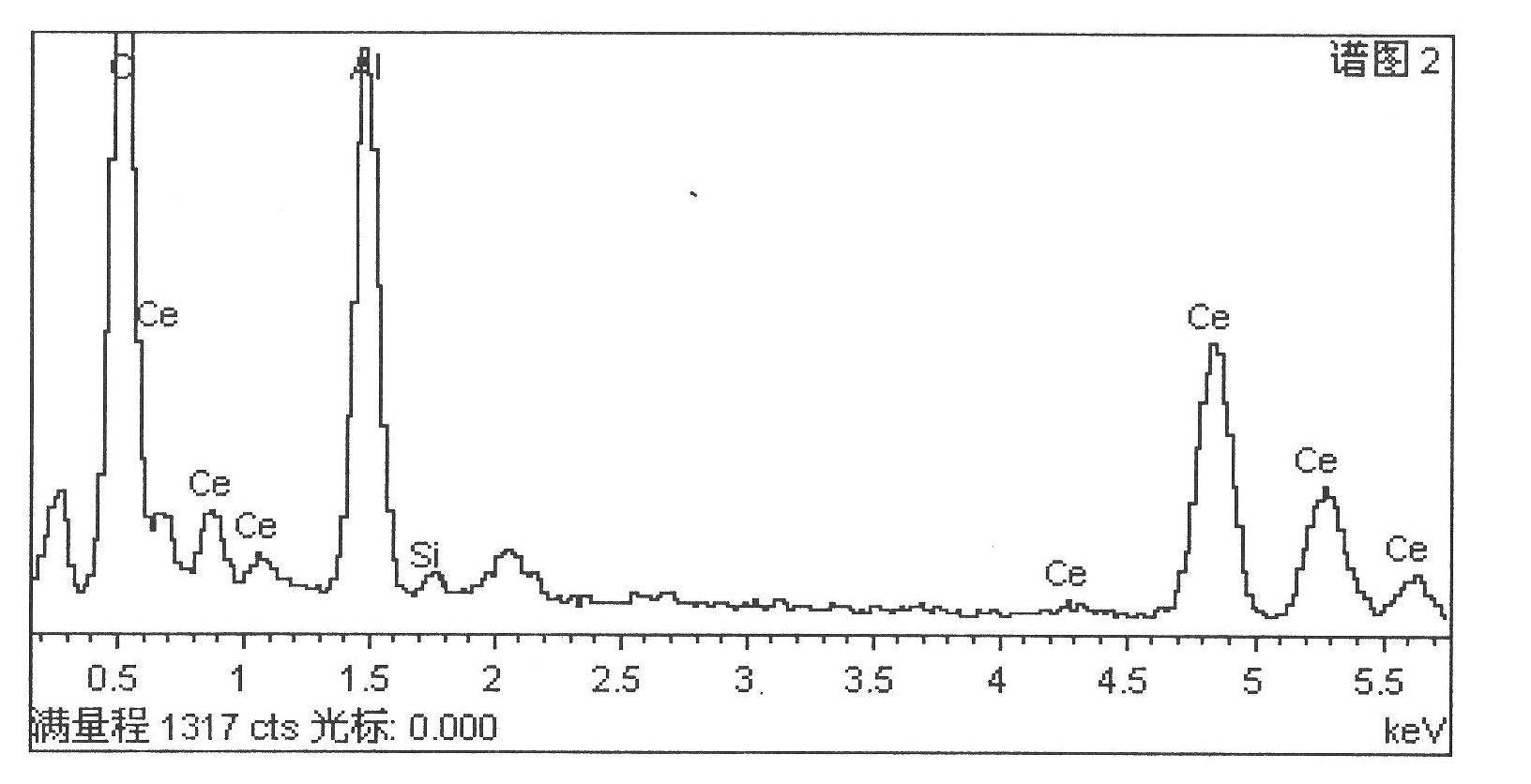
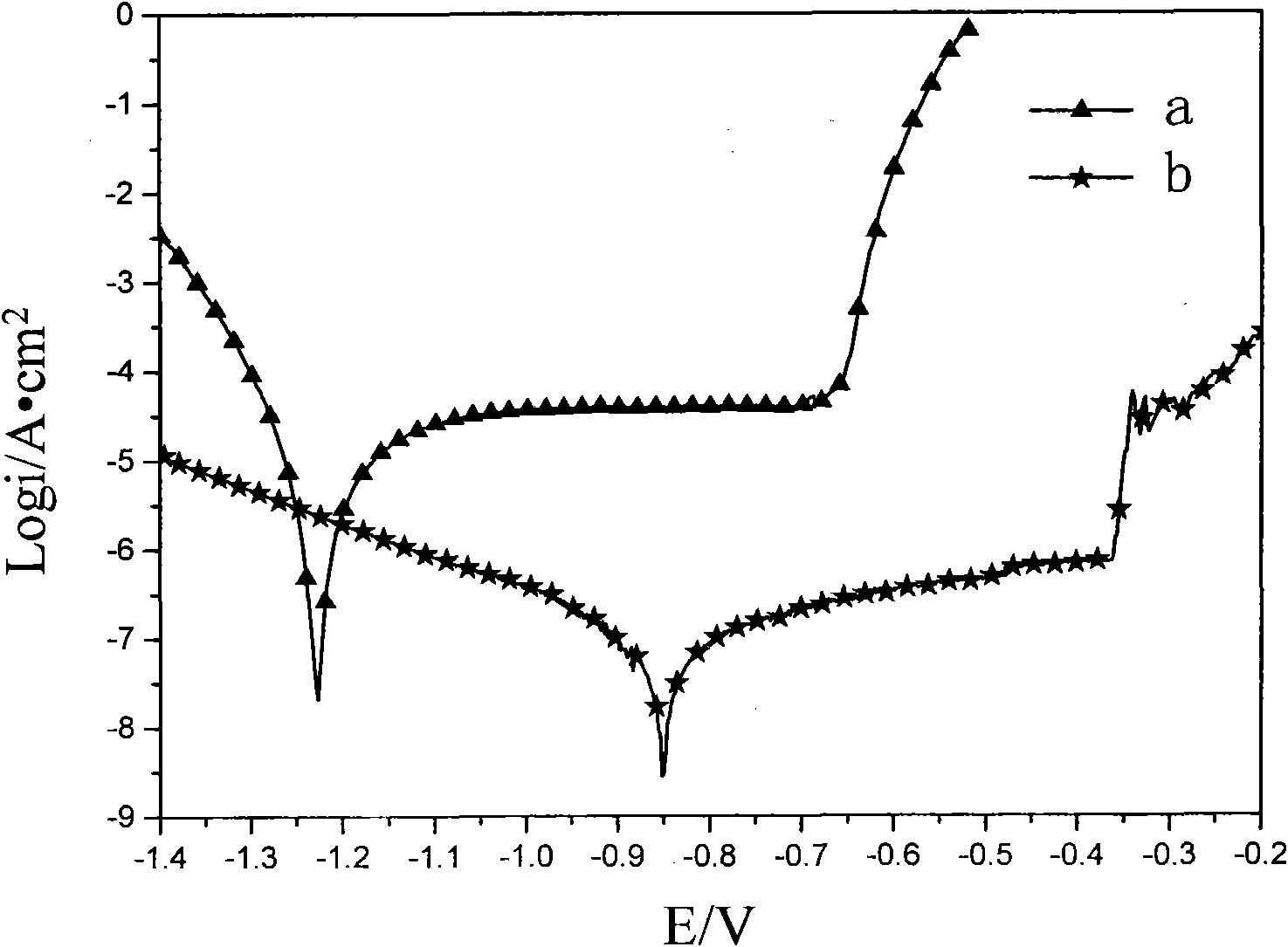
Preparation process of cerium-doped silicon corrosion-resisting membrane on surface of aluminium alloy
InactiveCN102021554AIncreased corrosion potentialImprove corrosion resistanceMetallic material coating processes5052 aluminium alloyCe element
The invention relates a preparation process of a cerium-doped silicon corrosion-resisting membrane on a surface of aluminium alloy. The preparation process is characterized in that surface pretreatment is carried out on aluminium alloy, and then the aluminium alloy is immersed in cerium salt conversion liquid dissolved with organosilicon, so as to carry out membrane formation reaction on the surface of the aluminium alloy, thus the corrosion-resisting membrane is formed; and then the aluminium alloy is taken out, is cleaned by deionized water and is dried. The invention has the advantages that the process is simple, the membrane formation speed is rapid, the preparation time of the corrosion-resisting membrane is shortened, the main constituents of the corrosion-resisting membrane layer are cerium dioxide and hydrated cerium oxide, and the uniformity and compactness of the surface of the membrane are good. After the cerium-doped silicon corrosion-resisting membrane is formed on the surface of the aluminium alloy, the corrosion resistance of the aluminium alloy is improved remarkably.
Owner:OCEAN UNIV OF CHINA
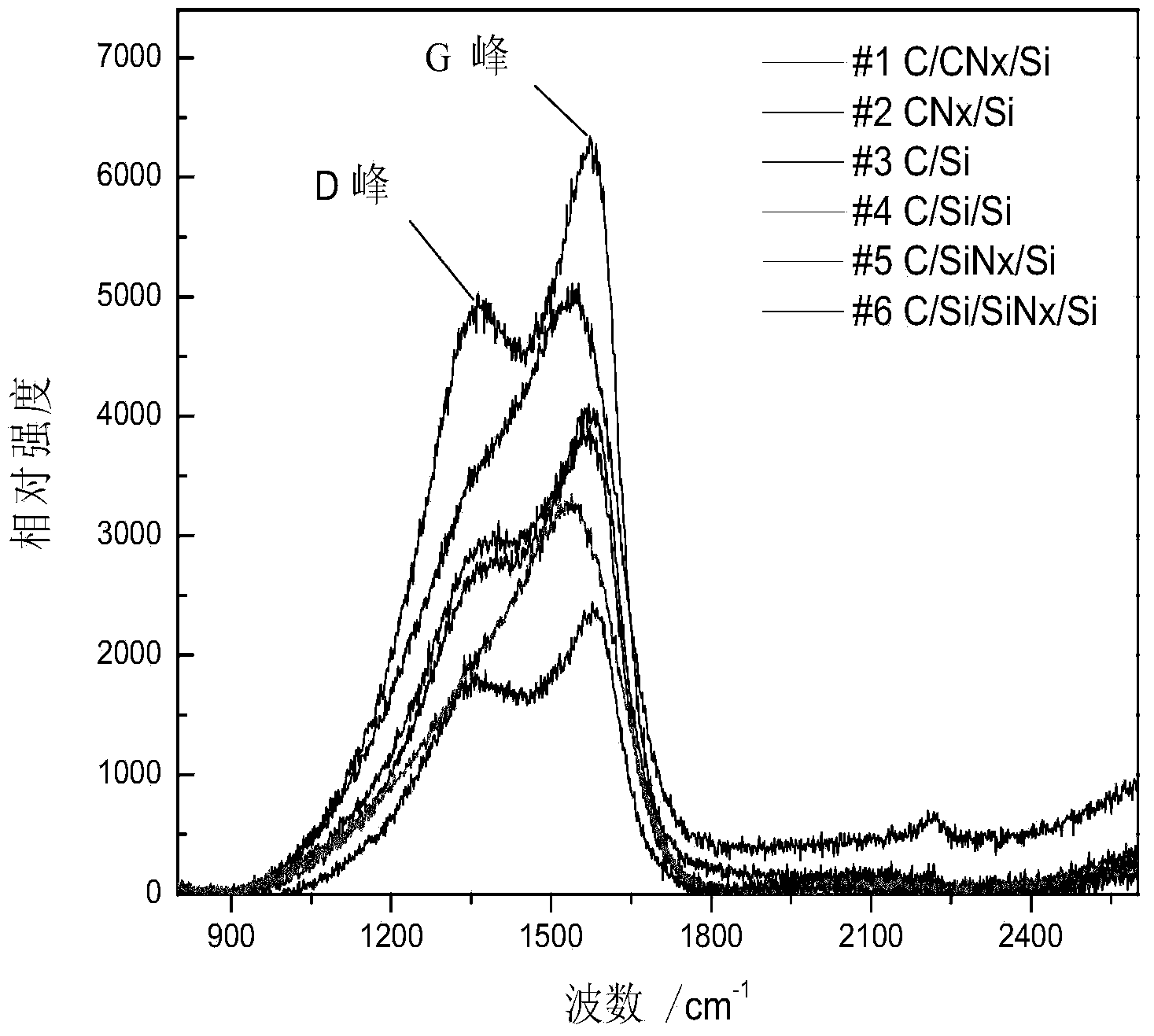
Method of preparing diamond-like membrane by film sputtering in SiNx middle layer
InactiveCN103849847ASimple preparation processGood repeatabilityVacuum evaporation coatingSputtering coatingSputteringOptoelectronics
The invention discloses a method of preparing a diamond-like film by magnetron sputtering in a SiNx middle layer. The method comprises the following steps: after cleaning a substrate Si chip, firstly forming a buffer layer SiNx thin film on the surface of the chip; and then, by taking a high purity carbon target as a target and Ar as a working gas, forming the diamond-like film on the SiNx thin film. The preparation process disclosed by the invention is simple and good in repeatability. The film can be deposited at low temperatures. The method is energy-saving, and the stress residue of the film cooled is reduced to prevent the film from falling. The diamond-like film prepared by the method disclosed by the invention has the advantages of low internal stress, good film quality, uniformity and compactness and the like.
Owner:SOUTHWEAT UNIV OF SCI & TECH
Method for plating nickel on surfaces of ceramic particles
InactiveCN107675149ALarge real surface areaHigh bonding strengthLiquid/solution decomposition chemical coatingChemical platingMegasonic cleaning
The invention provides a method for plating nickel on surfaces of ceramic particles. The method comprises the following steps: fused alumina zirconia ceramic particles are polished by using an abrasive paper; and after a preset roughness is reached, the fused alumina zirconia ceramic particles are put in anhydrous alcohol, are oscillated and cleaned by using ultrasonic waves, are put in rougheningliquid for roughening after drying, are sensitized after cleaning and drying, are put in acetone solution after sensitization, are cleaned by using the ultrasonic waves, are put in activating liquidfor activation after drying, are put in chemical plating liquid for plating after cleaning and drying, and are flushed and dried after plating to obtain finished products. The method for plating nickel on the surfaces of the ceramic particles increases real surface areas of the fused alumina zirconia ceramic particles and nickel metal, so that the bonding strength between the ceramic particles andthe nickel metal is improved; the reaction efficiency is improved; the nickel plating time is shortened; and the surfaces of the fused alumina zirconia ceramic particles after plating reach the metalgloss, are uniform in nickel plating and excellent in corrosion resistance, and achieve a certain wear resistance.
Owner:ANHUI FENGXING WEAR RESISTANT MATERIALS CO LTD +1
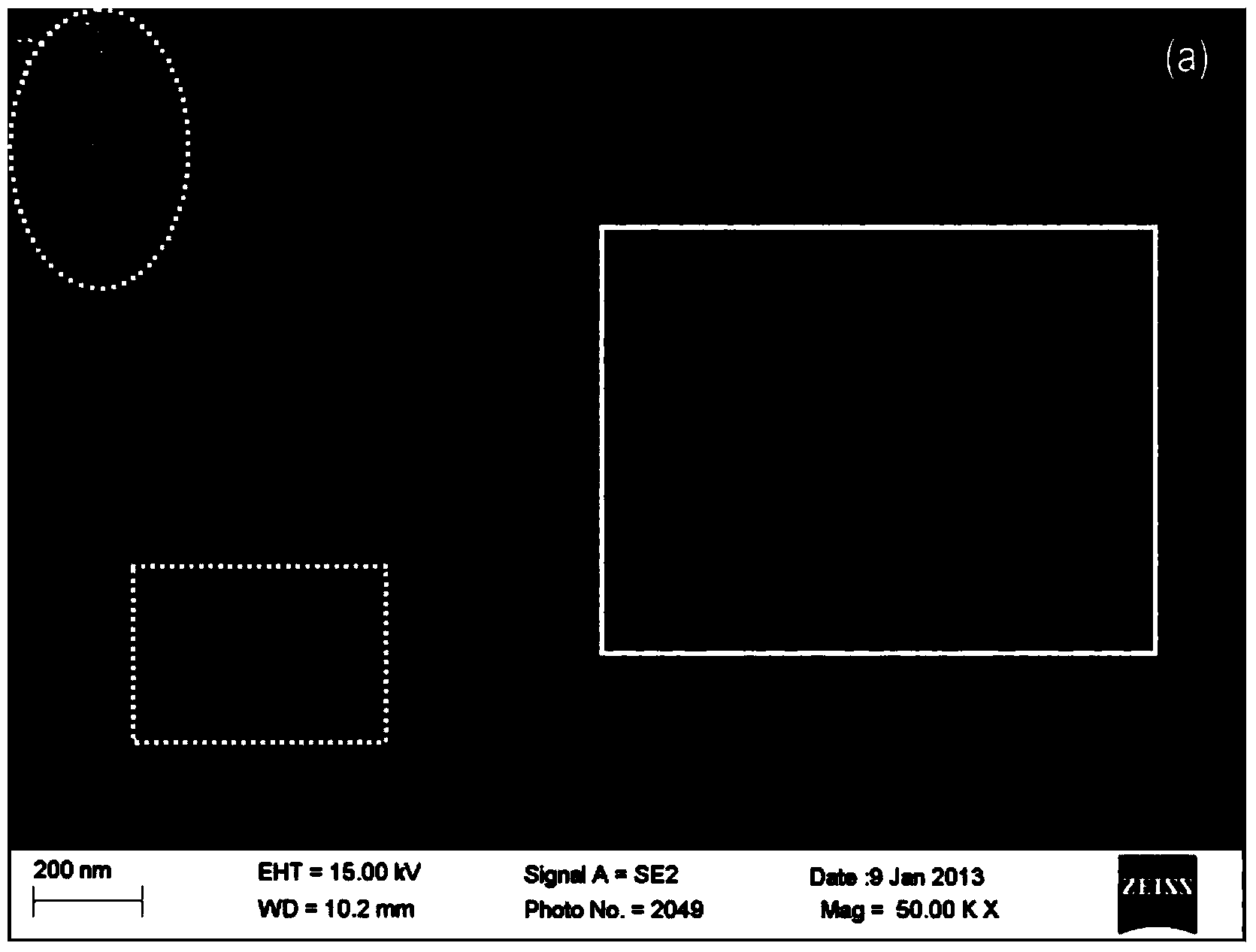
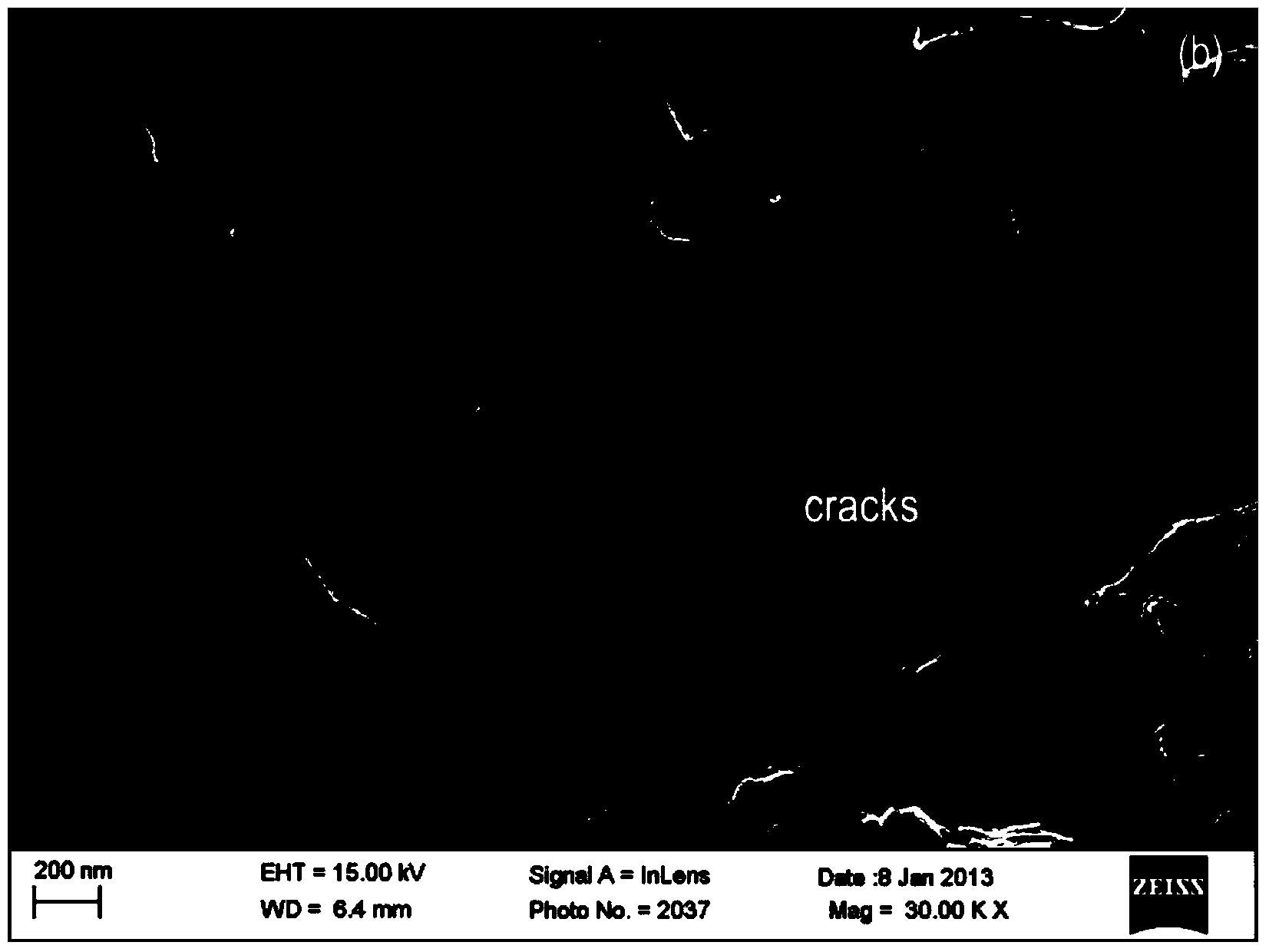
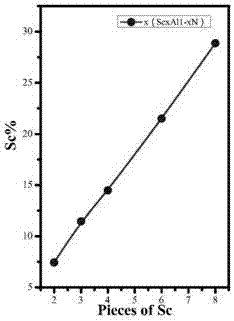
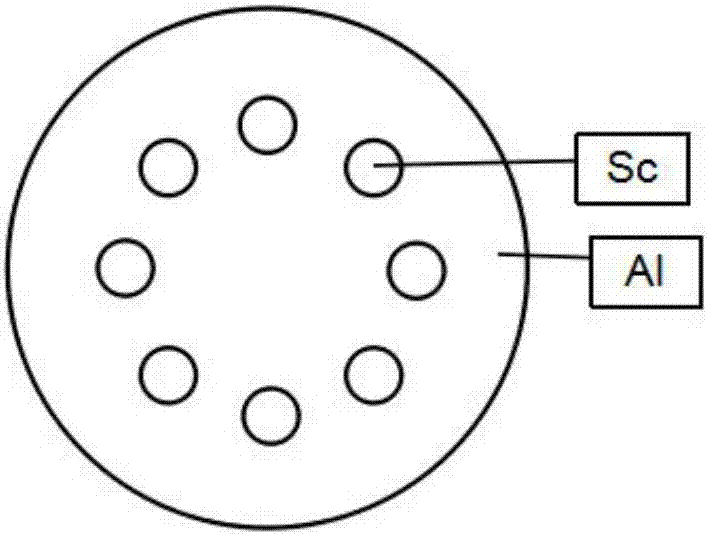
Process for preparing high-C-axis-orientation ScAlN thin film
InactiveCN106917088AShorten the timeExperiment operation is simpleVacuum evaporation coatingSputtering coatingRadio frequency magnetron sputteringDesalination
The invention discloses a process for preparing a high-C-axis-orientation ScAlN thin film and a preparation manner for lowering the process difficulty. In the vacuum environment, N2 gas is ionized through radio frequency so that local plasmas can be generated on a substrate, and a nitride layer is formed on the substrate mentioned above through the plasmas; by means of a radio frequency magnetron sputtering manner, a Sc-doped AlN thin film is prepared and formed on the substrate with a desalination layer under the room temperature condition, and the relative molecule content of the doping amount of the element Sc in the ScAlN thin film ranges from 0% to 28.87%; and regulation and control are achieved according to requirements, the relative molecule content values of the doping amount of the element Sc in the prepared thin film include 0, 7.45%, 11.45%, 14.49%, 21.50% and 28.87%. By means of the process, orientation of the ScAlN thin film can be optimized, and meanwhile the preparation difficulty is lowered.
Owner:UNIV OF ELECTRONICS SCI & TECH OF CHINA
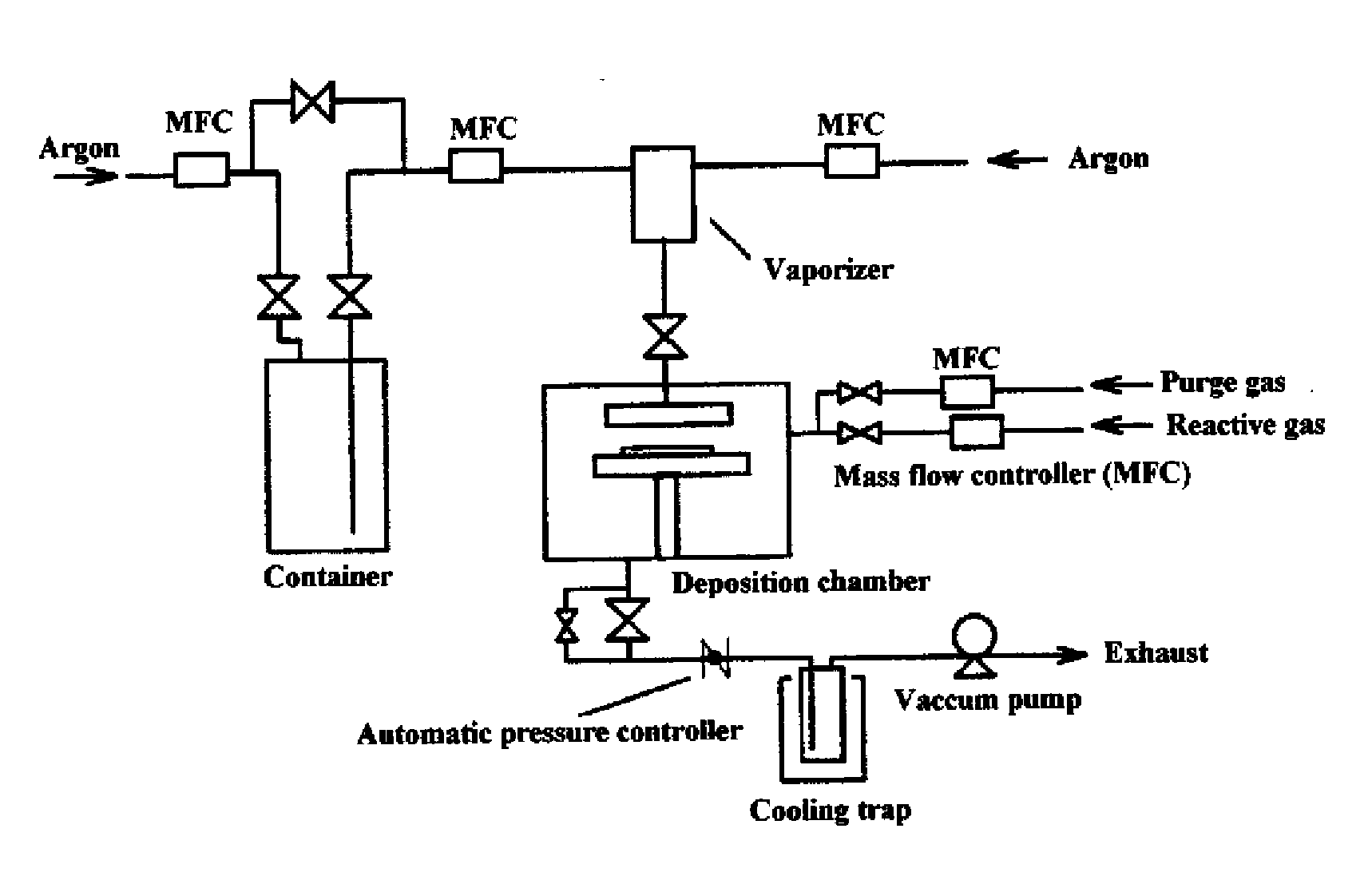
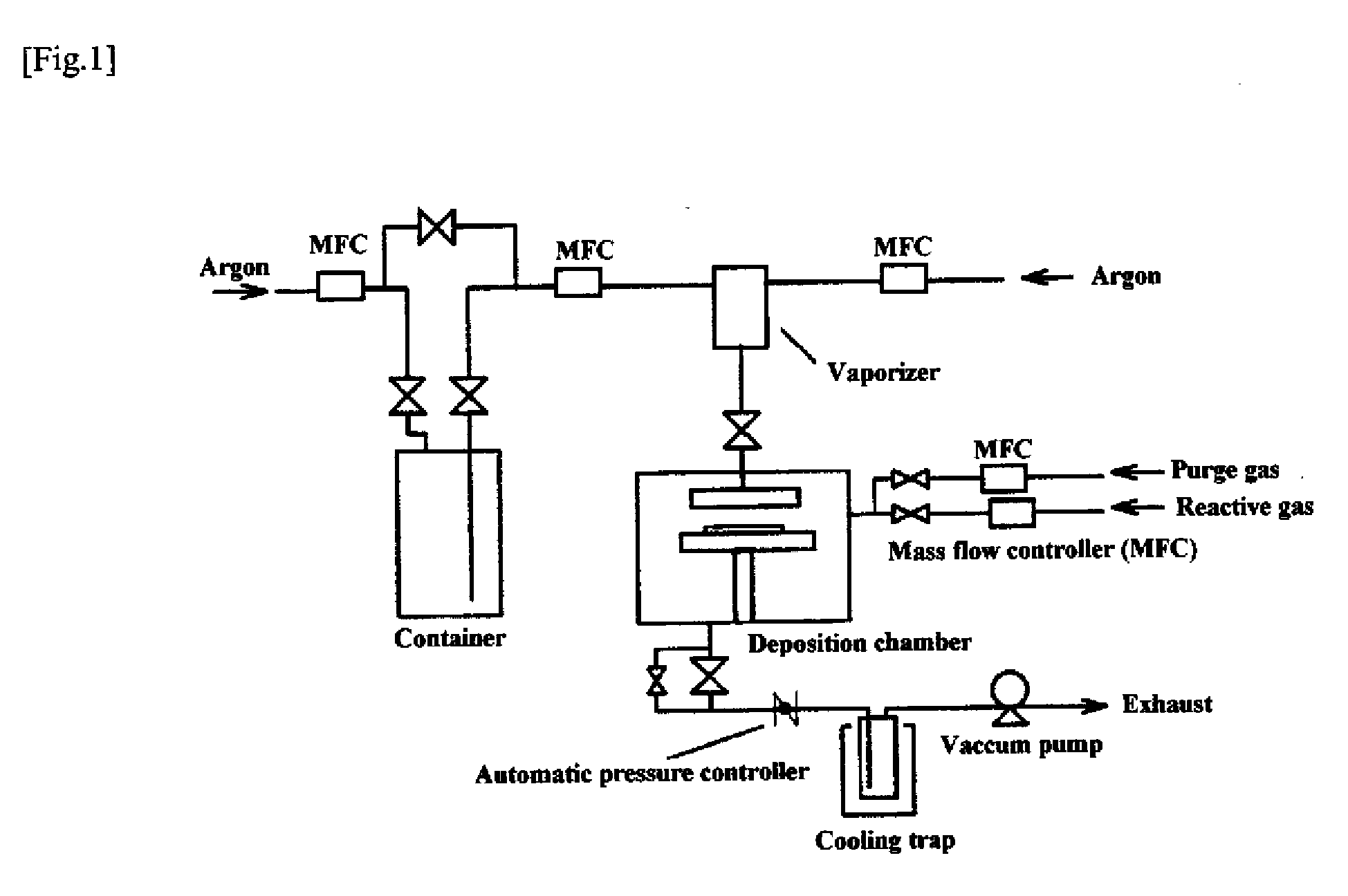
Vapor deposition source and vapor deposition apparatus
ActiveCN101041891AStable deposition rateHigh deposition rateElectrical apparatusElectroluminescent light sourcesCrucibleEngineering
Owner:CANON KK
Preparation method of diffusion impervious layer free Cu(Ru) alloy material with high thermal stability
InactiveCN103266304APrevent oxidationQuality assuranceVacuum evaporation coatingSputtering coatingAlloySingle crystal
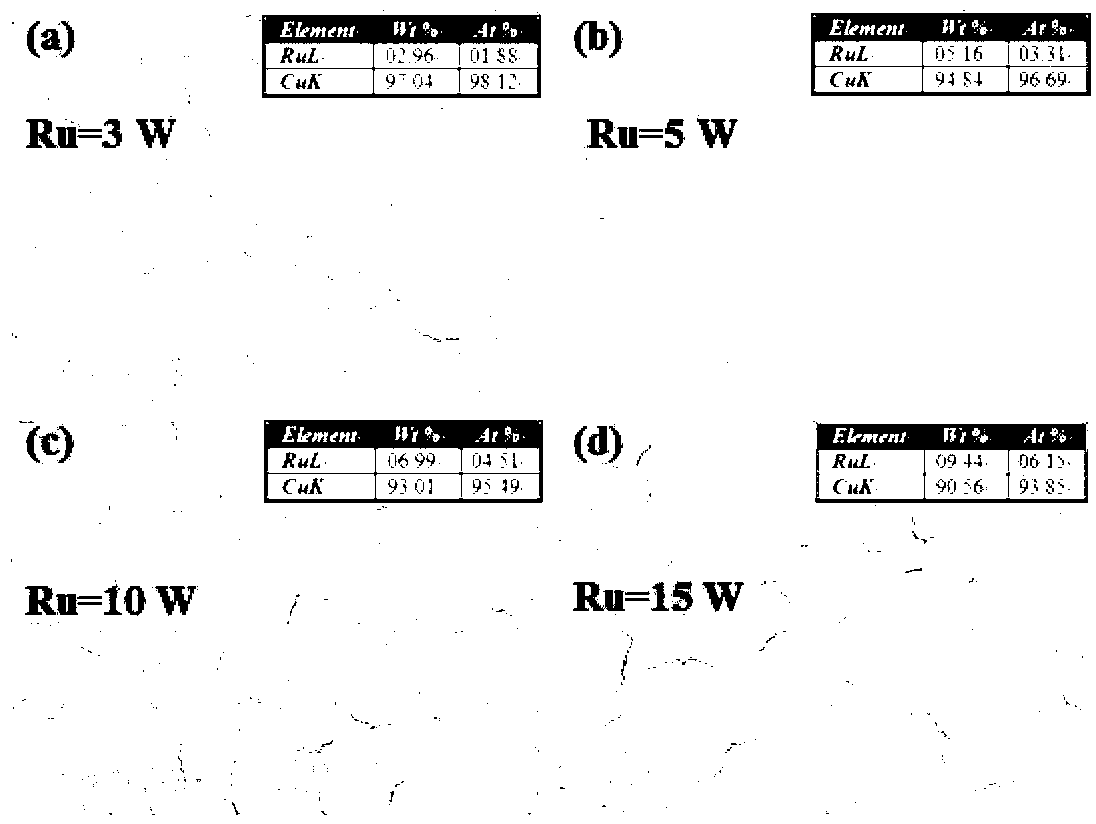
The invention relates to a preparation method of a diffusion impervious layer free Cu(Ru) alloy material with high thermal stability. The method is characterized by comprising the following steps: using a single crystal Si(111) wafer as a substrate material; selecting a high-purity Cu target and a high-purity Ru target; fixing the power of the Cu target at 80+ / -10; changing the sputtering power of the Ru target in a range of (3-15)+ / -1W so as to change the content of solid solution phase Ru in a Cu(Ru) alloy interconnecting material; and obtaining the alloy material through a direct current magnetic control confocal sputtering method. The diffusion impervious layer free Cu(Ru) alloy material with high thermal stability can be obtained, excellent electric performance can be maintained, and the actual application of the alloy material is better ensured. The method provided by the invention is simple to operate, good in repeatability and excellent in implementing effect.
Owner:JIANGSU UNIV OF SCI & TECH
Preparation method of ceramic particle metal composite material
ActiveCN107598136APacked tightlyPrevent peelingFoundry mouldsLiquid/solution decomposition chemical coatingChemical platingLost-foam casting
The invention provides a preparation method of a ceramic particle metal composite material. The preparation method comprises the following steps that ceramic particles are subjected to surface pretreatment and then placed into a chemical plating solution, and thus nickel-plated ceramic particles are obtained; an electric heating resistance wire is used for cutting a foam plate; the nickel-plated ceramic particles are washed and dried, and then coats the surface of a foam specimen, after drying, the foam specimen is bonded with a cross gate, a sprue and an ingate, and thus a lost foam casting white foam is obtained; the prepared lost foam casting white foam is placed into a sand box filled with dry sand, the dry sand is vibrated to be compacted and scraped to be flat, a plastic thin film isused for covering, a pouring cup is placed, and the negative pressure is pumped; after the dry sand is firm and is formed, metal liquid is cast; and after casting, vacuum relief, cooling and solidification then polishing are conducted. According to the preparation method of the ceramic particle metal composite material, the Rockwell hardness, impact toughness and overall abrasion resistance of the prepared composite material are greatly improved, no element diffusion exists between the ceramic particles and a metal substrate at the composite interface, and the bonding strength between the ceramic particles and metal is high.
Owner:安徽省凤形新材料科技有限公司 +1
Preparation method of ZnO/SnS solar cell element containing ZnO:Al window layer
InactiveCN102456768AImprove crystal qualityHigh precisionFinal product manufactureVacuum evaporation coatingSputteringRadio frequency magnetron sputtering
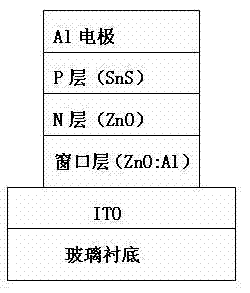
The invention relates to a preparation method of a ZnO / SnS solar cell element containing a ZnO:Al window layer, belonging to the technical field of the manufacturing process of solar cell elements. The solar cell element is of a multilayer structure, and comprises a glass substrate base material coated with an ITO (Indium-Tin Oxide) film, a ZnO:Al film window layer, a ZnO layer as an N layer, an SnS as a P layer and an Al membrane electrode at the uppermost layer. The main layer is the window layer ZnO:Al; the ZnO:Al film is prepared by using the traditional general JC500-3 / D type radio frequency magnetron sputtering device. The processing condition of the sputtering is as follows: the sputtering power is 100W-200W, the working vacuum air pressure is 0.3 Pa-0.9 Pa, the temperature is a room temperature, the sputtering time is 40-120 minutes, and the target-substrate distance is 8cm. The film has relatively good transparent electrical conductibility and infrared reflection power.
Owner:SHANGHAI UNIV
Preparation methods of optical dielectric thin film, Al2O3, silicon-containing thin film and laser cavity surface film
InactiveCN107254667AQuality improvementReach clean upVacuum evaporation coatingSputtering coatingHigh energyEvaporation
The invention discloses preparation methods of an optical dielectric thin film, Al2O3, a silicon-containing thin film and a laser cavity surface film. The preparation method of the optical dielectric thin film comprises the steps that a substrate is placed in a cavity of electron beam evaporation equipment and vacuumized; the surface of the substrate is bombarded under a high-energy electric field through ionized argon and ionized nitrogen; a target material with an optical dielectric is bombarded by high-energy electrons in batches, and optical dielectric particles are pre-molten step by step; and the optical dielectric particles are deposited on the substrate, and thus preparation of the optical dielectric thin film is completed. According to the preparation method of the optical dielectric thin film, the annealing process is not needed, the effects of the annealing process on the optical and mechanical properties and the like of the optical dielectric thin film are avoided, accordingly, the high-quality optical dielectric thin film is obtained, and the foundation is laid for preparation of various anti-reflection films and high reflectivity films.
Owner:INST OF SEMICONDUCTORS - CHINESE ACAD OF SCI
Chemical palladium solution applied to PCB chemical gold palladium clad layer
ActiveCN110318047AExtended service lifeControl deposition rateLiquid/solution decomposition chemical coatingPrinted circuit manufactureWire widthPh buffering
The invention discloses a chemical palladium solution applied to a PCB chemical gold palladium clad layer. Main salt, a coordination agent, a reducing agent, a stabilizer, a pH buffering agent and a surface active agent are included. The mass concentration serves as the unit, the components include, by proportion, 0.4-0.6 g / L of the palladium in the main salt according to the concentration, 0.1-30g / L of the coordination agent, 0.1-25 g / L of the reducing agent, 1-150 mg / L of the stabilizer, 1-25 g / l of the pH buffering agent, 0.1-10 mg / L of the surface active agent and the balance pure water.A palladium layer prepared through the palladium solution can meet good welding and bonding requirements, and meanwhile the chemical palladium solution is applicable to surface treatment of a circuitwith the wire width / wire distance being 0.5 mil or below or even smaller.
Owner:深圳市溢诚电子科技有限公司
Metal compound, material for chemical vapor phase growth, and process for forming metal-containing thin film
ActiveUS20100247765A1Reduced residual carbonStable deposition rateGroup 4/14 organic compounds without C-metal linkagesTitanium organic compoundsDeposition temperatureHalogen
A novel metal compound of general formula (1), a material for chemical vapor phase growth containing the compound, and a process for forming a metal-containing thin film by chemical vapor phase growth using the material. Among the compounds of formula (1), those wherein X is a chlorine atom are preferred because of inexpensiveness and high volatility. When M is titanium, those wherein m is 1 are preferred as having a greater difference between a volatilization temperature (vapor temperature) and a deposition temperature (reaction temperature), which provides a broader process margin.In formula (1), M is titanium, zirconium, or hafnium; X is a halogen atom; and m is 1 or 2.
Owner:ADEKA CORP
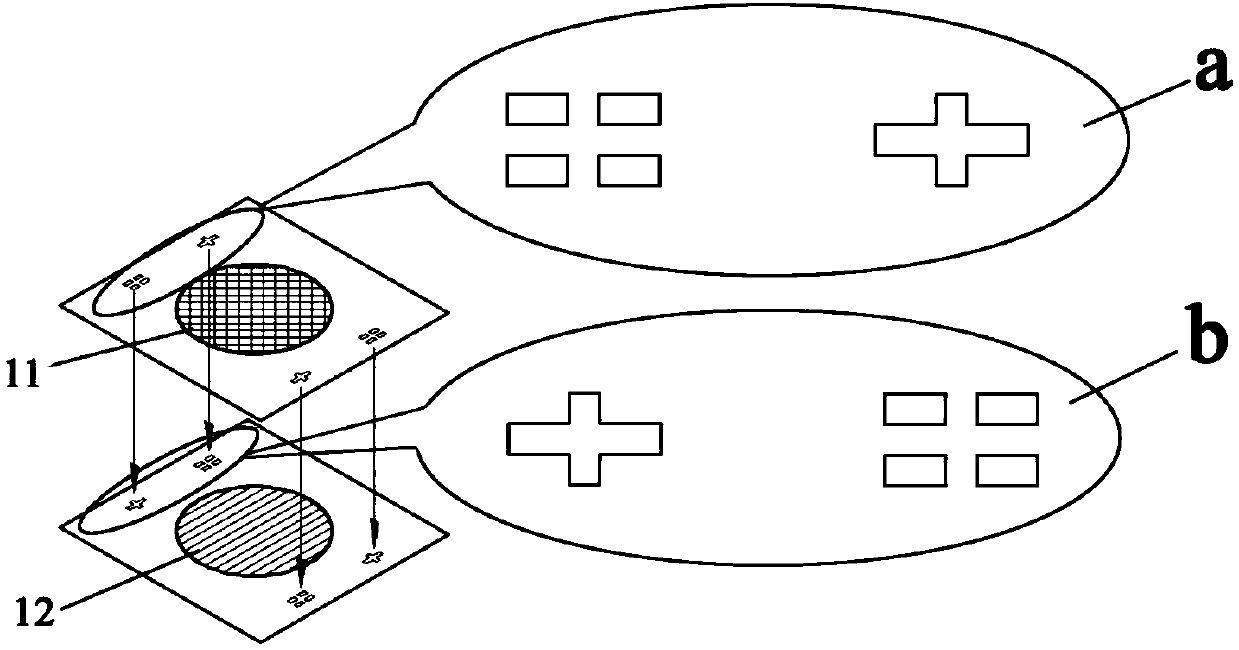
Quartz bonding method based on gold-tin co-crystal
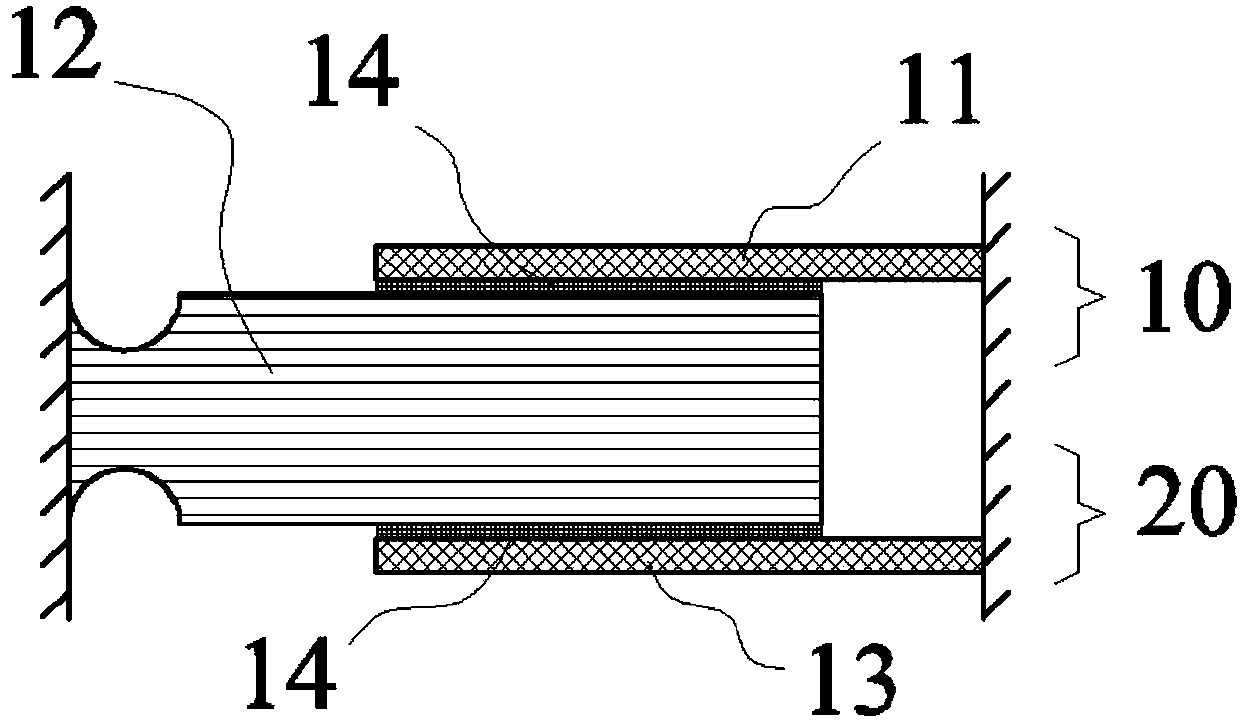
InactiveCN105502967AAchieve precision machiningAchieve precise alignmentOptoelectronicsPrepared Material
The invention relates to a quartz bonding method based on gold-tin co-crystal. The method comprises the following steps: preparing materials; cleaning the prepared materials; depositing thin films: depositing a Cr / Au thin film on one (11) of two parts on the lateral sides in sequence, depositing a Cu / Au / Sn / Au thin film on one side, which is corresponding to the part (11), of a middle part (12), then depositing a Cr / Au thin film on the other one (13) of two parts on the lateral sides in sequence, depositing a Cr / Au / Sn / Au thin film on the other side, which is corresponding to the part (13), of the middle part (12) in sequence; and carrying out thermo-compression bonding. The provided method can precisely control the ratio of components of a solder layer, can achieve precise processing of patterned solder, and is capable of precisely aligning quartz sensitive structural members.
Owner:BEIJING AUTOMATION CONTROL EQUIP INST
A kind of preparation method of wo3 electrochromic thin film
InactiveCN103246119BUniform depositionUniform dense depositionNon-linear opticsRadio frequency magnetron sputteringIndium tin oxide
Owner:NANJING UNIV OF SCI & TECH
Method for manufacturing window layers of copper, zinc, tin and sulfur solar cells
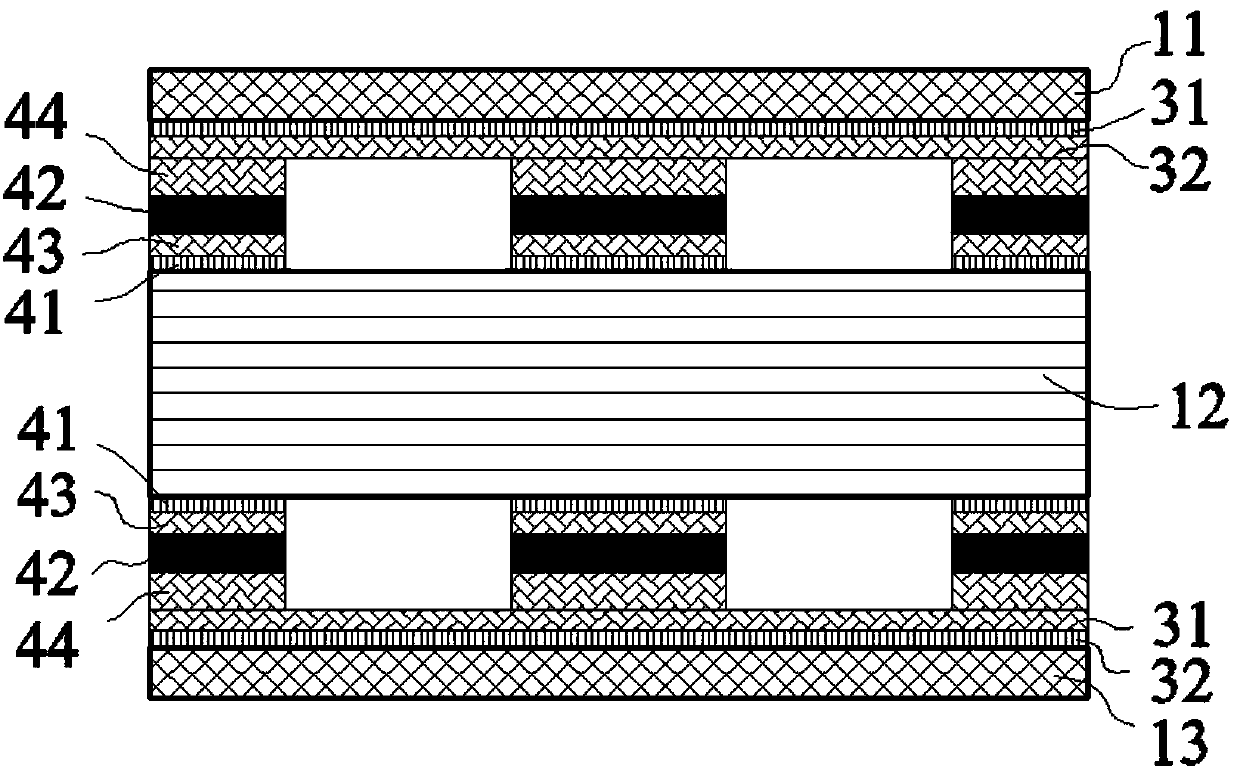
InactiveCN103236466AStable deposition rateFilm thickness is easy to controlFinal product manufactureSemiconductor devicesThin membraneSeed crystal
The invention discloses a method for manufacturing window layers of copper, zinc, tin and sulfur solar cells, and belongs to the technical field of processes for manufacturing thin-film devices of solar cells. The method is characterized by comprising steps of manufacturing a ZnO seed crystal by means of magnetron sputtering; growing a ZnO nano-array by a hydrothermal process; and vulcanizing and growing a ZnS shell structure by the hydrothermal process. The method has the advantages that the adhesive strength of a substrate and a ZnO@ZnS nano-array shell structure manufactured by the hydrothermal process is high, the ZnO@ZnS nano-array shell structure is high in transmittance and excellent in electrical property and can be used as a window layer of a CZTS (copper, zinc, tin and sulfur) solar cell, and the method can be excellently connected with a follow-up process for manufacturing the cell.
Owner:SHANGHAI UNIV
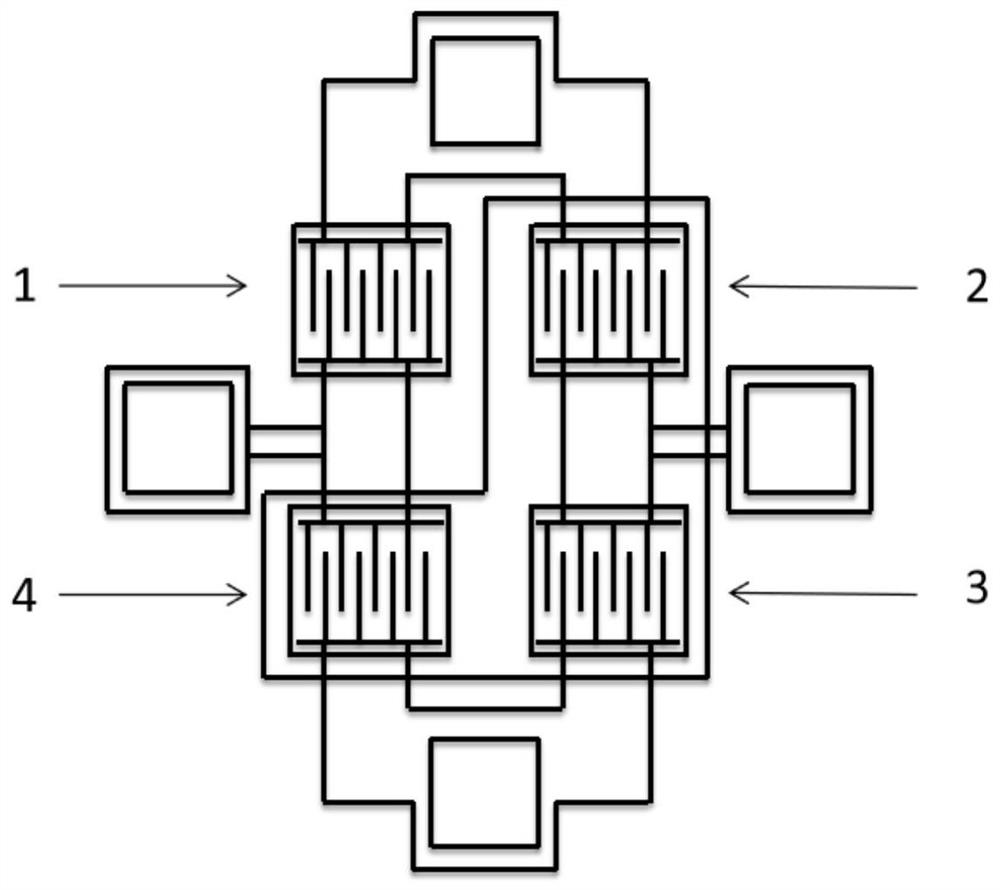
Zinc oxide electric bridge ultraviolet detection unit and preparation method thereof
ActiveCN112510108ALow costImprove bindingFinal product manufactureSolid-state devicesUltravioletEngineering
The invention provides a zinc oxide electric bridge ultraviolet detection unit and a preparation method thereof. The zinc oxide electric bridge ultraviolet detection unit comprises four ultraviolet photoresistors, which are arranged in an electric bridge structure, and are respectively a first ultraviolet photoresistor, a second ultraviolet photoresistor, a third ultraviolet photoresistor and a fourth ultraviolet photoresistor, wherein ZnO shading layers are arranged on the second ultraviolet photoresistor, the third ultraviolet photoresistor and the fourth ultraviolet photoresistor. The zincoxide electric bridge ultraviolet detection unit provided by the invention has the characteristics of small size and low cost, and has the advantage of being easier to combine with a back-end processing circuit.
Owner:XI AN JIAOTONG UNIV
Electroplating solution and electroplating technology of workpiece inner hole hard chrome
The invention discloses an electroplating solution of workpiece inner hole hard chrome. The electroplating solution is prepared from the following raw materials: H2SO4, CrO3 and Cr<3+>, wherein a proportion of the H2SO4 to the CrO3 is (0.8 to 1.2):100. The invention also provides an electroplating technology of the workpiece inner hole hard chrome. The electroplating technology comprises the following steps: (1) workpiece pretreatment; (2) preheating; (3) reversed etching; (4) activating; (5) chrome plating; (6) out of groove of workpiece, and recycling and cleaning;(7) unhanging and polishing; (8) secondary electroplating; (9) checking. According to the electroplating solution and the electroplating technology of the workpiece inner hole hard chrome, provided by the invention, after components and proportion of the electroplating solution are regulated, deposition speeds of the upper end and the lower end of a chrome layer are approximate, and the generation speed of the Cr<3+> is relatively reduced; after the electroplating technology is regulated, the product quality is increased, and the design requirements are met.
Owner:CHANGSHA DALISHEN HYDRAULIC ENG
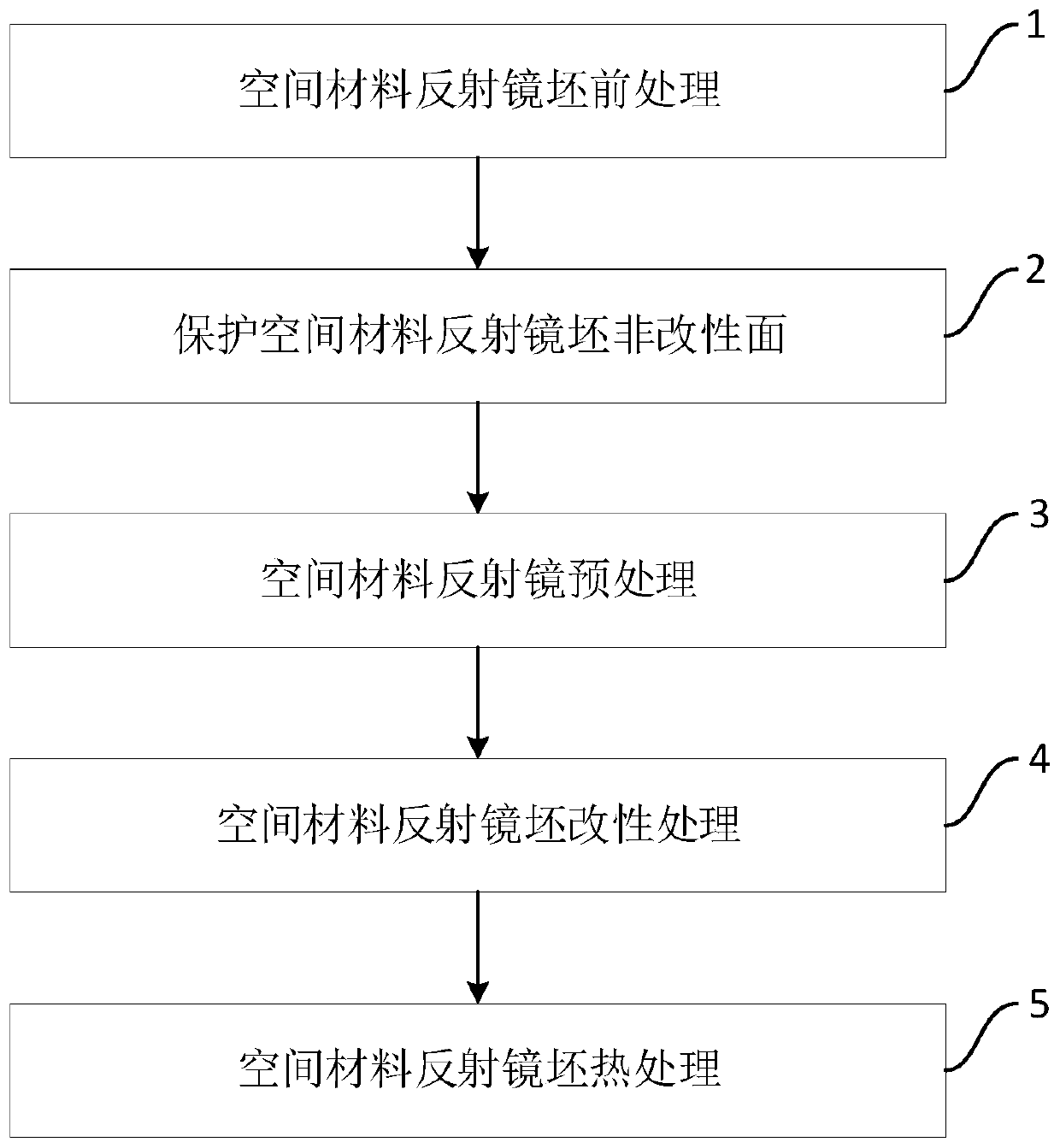
Modification treatment method for surface of space material reflector blank
InactiveCN110777370AAchieve surface modificationStable deposition rateLiquid/solution decomposition chemical coatingInvestigating material hardnessThermal dilatationAlloy
The invention discloses a modification treatment method for surface of a space material reflector blank. The modification treatment method comprises the steps that S1, the reflector blank is pretreated, specifically, a zinc dipping solution is used for treating the modified surface of the space material reflector blank, and a transition layer is generated on the modified surface of the space material reflector blank; and S2, modification treatment is conducted, specifically, the pretreated space material reflector blank is treated through a modification treatment solution, and a nickel-phosphorus alloy modified layer is deposited on the modified surface of the space material reflector blank. By means of the modification treatment method for the surface of the space material reflector blank, the nickel-phosphorus alloy modified layer with the microhardness being HV 400-780, the melting point being not lower than 890 DEG C, the thermal expansion coefficient being (7.8-9.4) * 10<-6> / DEG Cand the porosity being lower than 5% can be deposited on the surface of the reflector blank through a chemical deposition method, the surface defect grade of the nickel-phosphorus alloy modified layer is IV, the modification treatment method is suitable for optical ultra-precision machining, and an ultra-smooth surface with the root mean square value being 0.049 lambda, the roughness being superior to 2nm and the surface defect grade being IV can be obtained through optical precision polishing.
Owner:CHANGCHUN UP OPTOTECH
Method and apparatus for producing large uniform thickness nanomaterial sheets
ActiveUS9902123B1High pressure capabilityStable deposition rateTextiles and paperPress ramFluid controlVariable size
A batch-automated microfiltration press for producing large uniform thickness nanomaterial sheets includes a filtration envelope of variable size that is defined between an upper platen having a fixed position and a lower platen disposed in spaced apart, parallel relation to the upper platen. The upper platen has a plurality of input nozzles and a plurality of flow dispersers associated with each of the input nozzles. Nanoparticles are deposited onto a fine filter membrane positioned in the filtration envelope. Flow channels and drain holes are formed in the lower platen. Each of the flow channels has a non-linear path of travel. A closed-loop fluid control system maintains a predetermined rate of nanoparticle deposition onto the fine filter membrane. A motion control system controls the raising and the lowering of the lower platen, maintaining a constant displacement instead of constant pressure, enabling the production of clean, undamaged sheets of nanomaterial papers.
Owner:INNOVATECH ENG
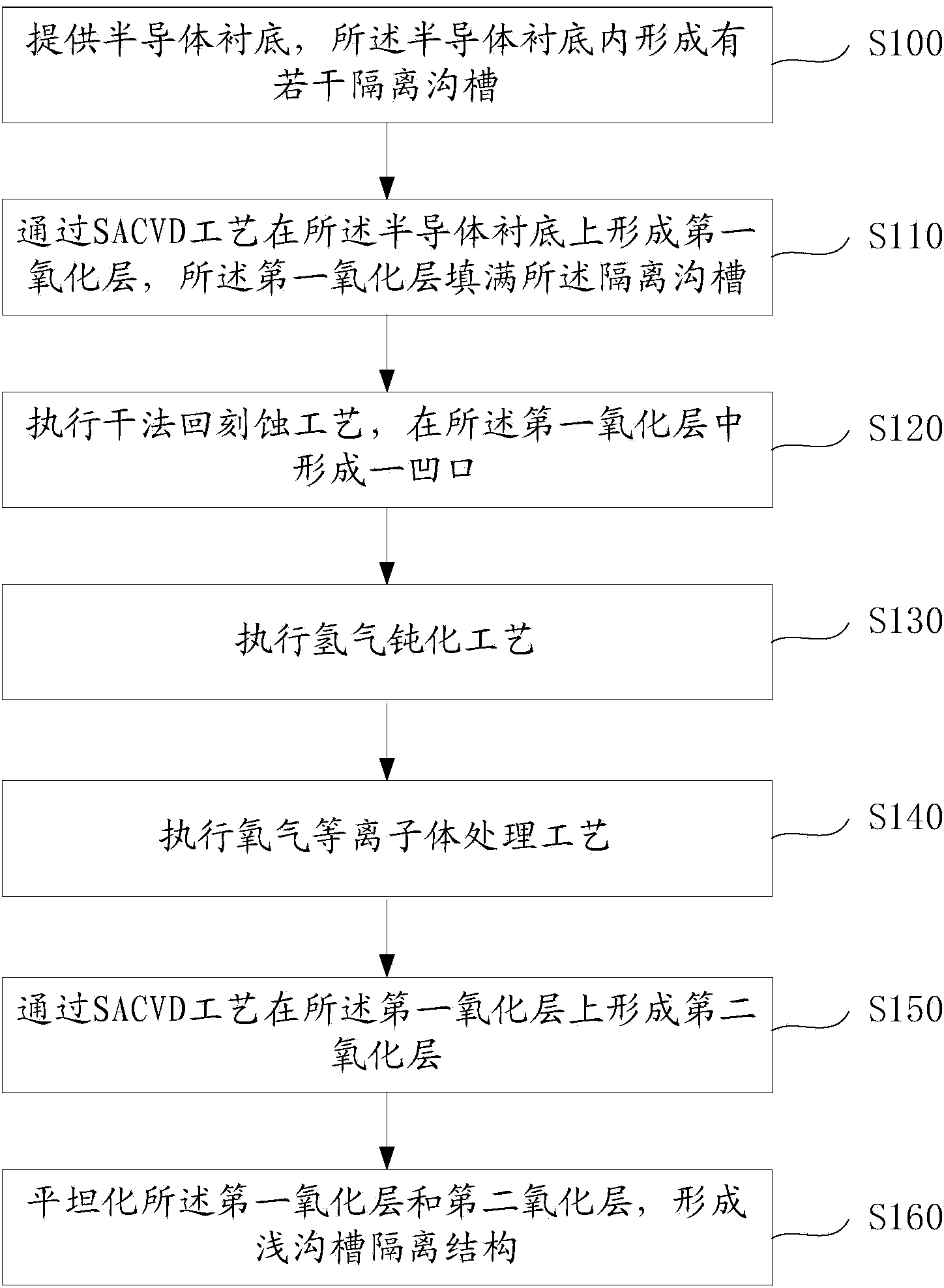
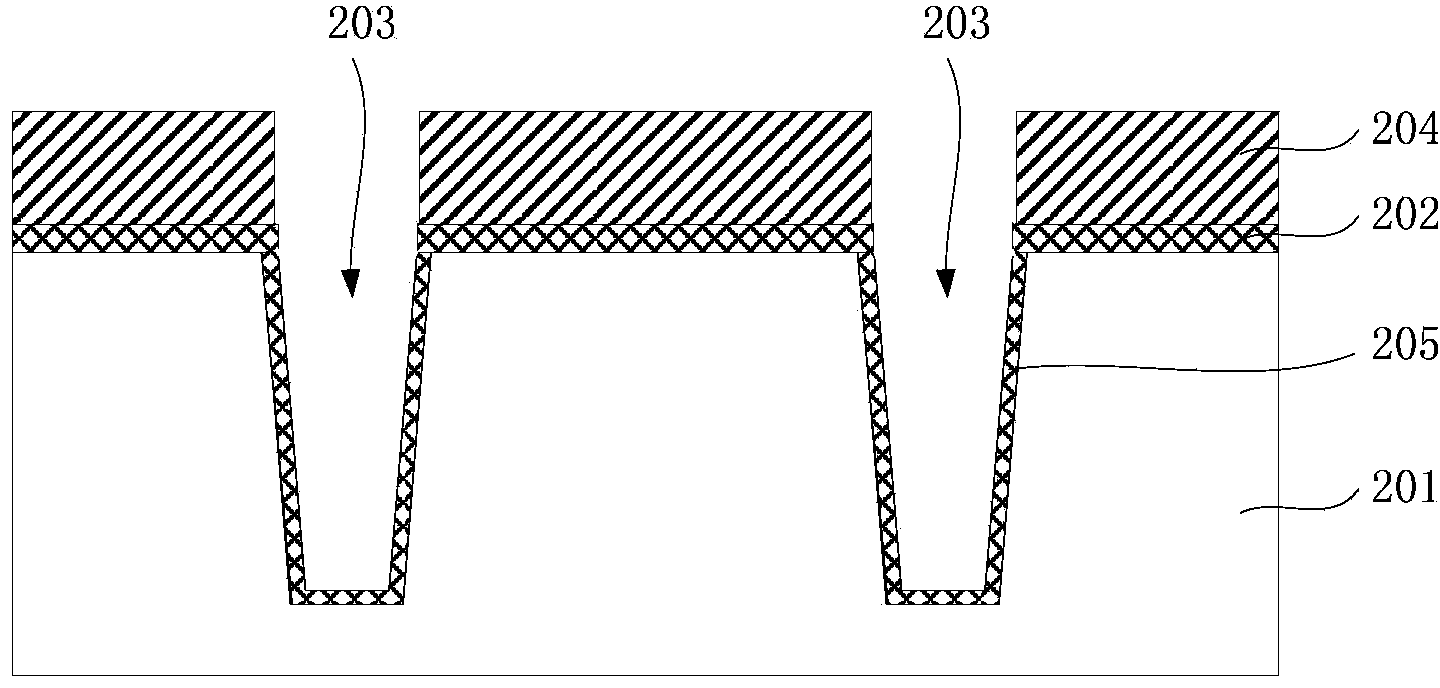
Method for forming shallow trench isolation structure
ActiveCN103545243AReduce vulnerabilityElimination of unsaturated bondsSemiconductor/solid-state device manufacturingIsolation effectO2 plasma
The invention provides a method for forming a shallow trench isolation structure. After a first oxidation layer is formed through the SACVD technology, the dry etching technology is executed to eliminate a fragile face of the first oxidation layer, and then a second oxidation layer is formed through the SACVD technology. The shallow trench isolation structure formed with the method is good in isolation effect, and a semiconductor device comprising the shallow trench isolation structure is good in stability and not prone to electric leakage and breakdown. Besides, the hydrogen passivation technology is added, unsaturated bonds on the surface of a film can be eliminated with the hydrogen passivation technology, and thus the deposition rate of the subsequent technology is stable, and finally the thickness uniformity of the film is improved. Furthermore, the oxygen plasma processing technology is adopted, hydrogen bonds on the surface and the surface layer of the structure are effectively removed through O2 plasma to eliminate the Q-time effect of the deposition rate of the subsequent manufacturing process, and thus the structure is more stable before the deposition of the subsequent manufacturing process.
Owner:SHANGHAI HUALI MICROELECTRONICS CORP
A kind of preparation method of ceramic particle metal composite material
ActiveCN107598136BLarge real surface areaHigh bonding strengthFoundry mouldsLiquid/solution decomposition chemical coatingChemical platingParticulate metal
The invention provides a preparation method of a ceramic particle metal composite material. The preparation method comprises the following steps that ceramic particles are subjected to surface pretreatment and then placed into a chemical plating solution, and thus nickel-plated ceramic particles are obtained; an electric heating resistance wire is used for cutting a foam plate; the nickel-plated ceramic particles are washed and dried, and then coats the surface of a foam specimen, after drying, the foam specimen is bonded with a cross gate, a sprue and an ingate, and thus a lost foam casting white foam is obtained; the prepared lost foam casting white foam is placed into a sand box filled with dry sand, the dry sand is vibrated to be compacted and scraped to be flat, a plastic thin film isused for covering, a pouring cup is placed, and the negative pressure is pumped; after the dry sand is firm and is formed, metal liquid is cast; and after casting, vacuum relief, cooling and solidification then polishing are conducted. According to the preparation method of the ceramic particle metal composite material, the Rockwell hardness, impact toughness and overall abrasion resistance of the prepared composite material are greatly improved, no element diffusion exists between the ceramic particles and a metal substrate at the composite interface, and the bonding strength between the ceramic particles and metal is high.
Owner:安徽省凤形新材料科技有限公司 +1
Electroplating tank
ActiveCN103789806BReduce in quantityImprove uniformityTanksFinal product manufactureIndiumSolar cell
The invention belongs to the technical field of preparation of solar cells, and in particular relates to an electroplating bath, which is used for depositing elemental copper, gallium, indium or selenium or a copper, gallium, indium or selenium alloy on the surface of a substrate. The electroplating bath comprises an electroplating branch bath and a master bath, wherein the electroplating branch bath is provided with an accommodation cavity for containing an electroplating solution; anodes immersed in the electroplating solution are hung on the lateral surfaces of the electroplating branch bath; a conveying device for carrying the substrate to synchronously move is arranged on the middle part of the electroplating branch bath; the anodes are arranged on the two sides of the conveying device; parts, opposite to the anodes, on the two lateral surfaces of the conveying device are immersed in the electroplating solution; the substrate is attached to the lateral surfaces of the conveying device, and is opposite to the anodes when passing through the electroplating branch bath. According to the electroplating bath, the length of an electroplating production line is reduced, and a small space is occupied; the elemental copper, gallium, indium or selenium or the copper, gallium, indium or selenium alloy is deposited on the front surface of the substrate only, so that few process steps are required; the anodes are opposite to the substrate, so that the uniformity and smooth finish of the surface of a deposited CIGS (copper, indium, gallium and selenium) absorption layer are ensured.
Owner:SOLTRIUM TECH LTD SHENZHEN
Model compensating method for controlling thin film deposition rate in physical gaseous phase thin film deposition process
ActiveCN102492931BTo achieve thickness control requirementsStable deposition rateVacuum evaporation coatingSputtering coatingRelational modelGas phase
The invention discloses a model compensating method for controlling thin film deposition rate in physical gaseous phase thin film deposition process, which is characterized by creating a relational model between the deposition rate and target material consumption amount on the basis of off-line measurement. Process control parameters including electric power in the thin film deposition process iscompensated by using the relational model, and relatively stable thin film deposition rate and thin film performance of products in different batches are achieved. The model compensating method is used for compensating main control parameters, so that stable thin film deposition rate is achieved, and adjustment of thin film micro-morphology and material performance is expected to be achieved through control of deposition rate. The model compensating method does not need to use complicated real-time observation systems.
Owner:上海鼎弈材料科技有限公司