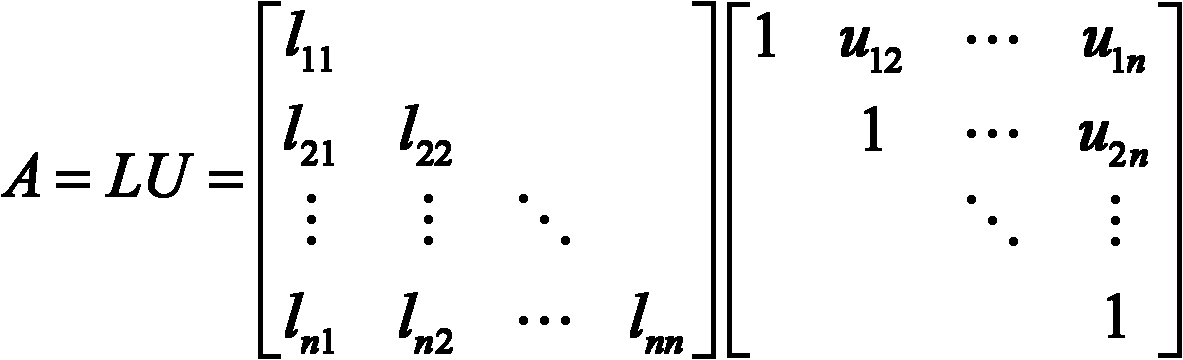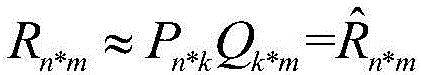Patents
Literature
374results about How to "Speed up decomposition" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
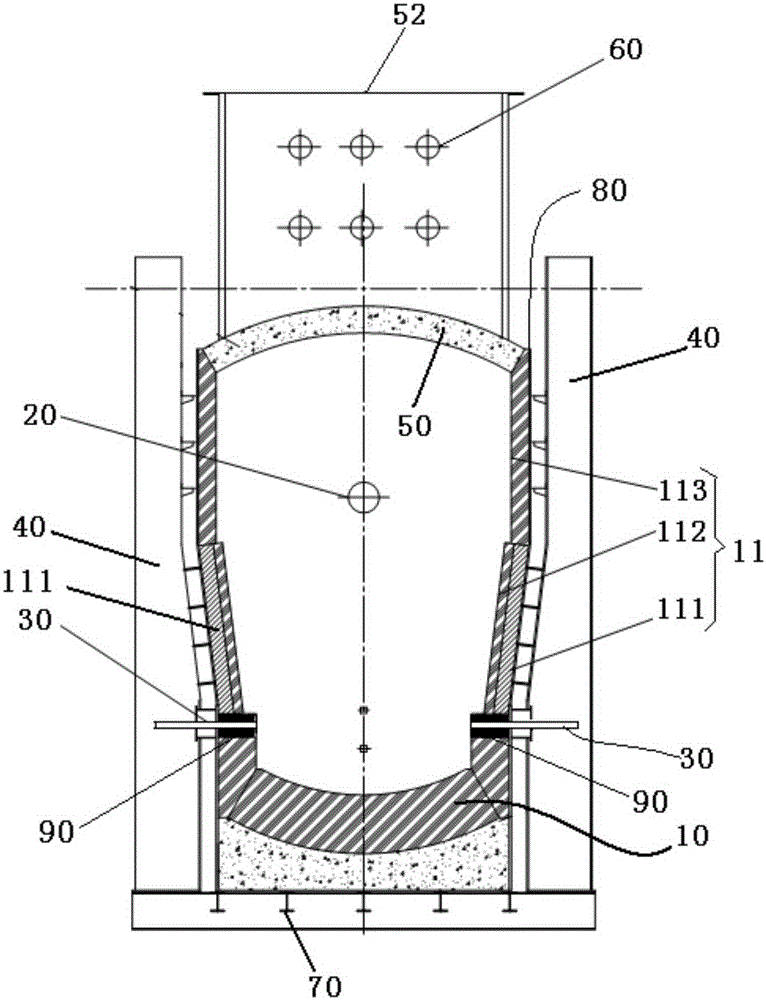
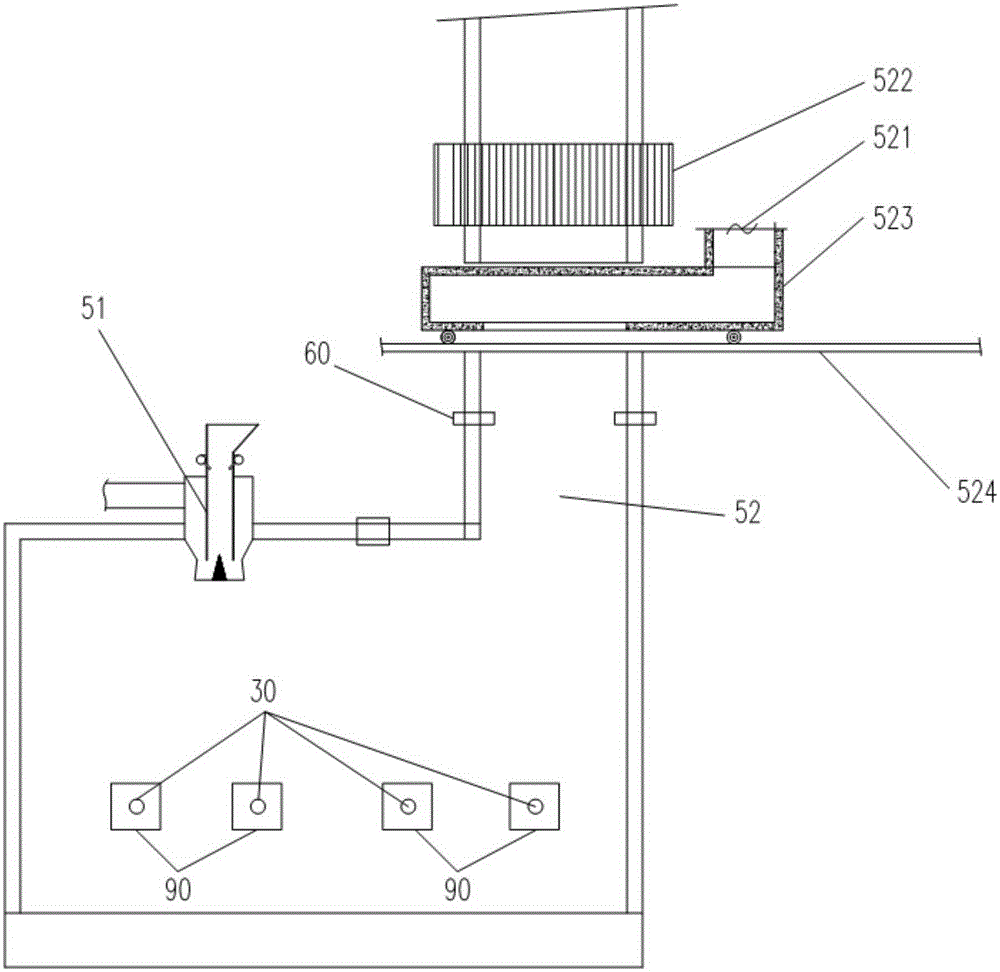
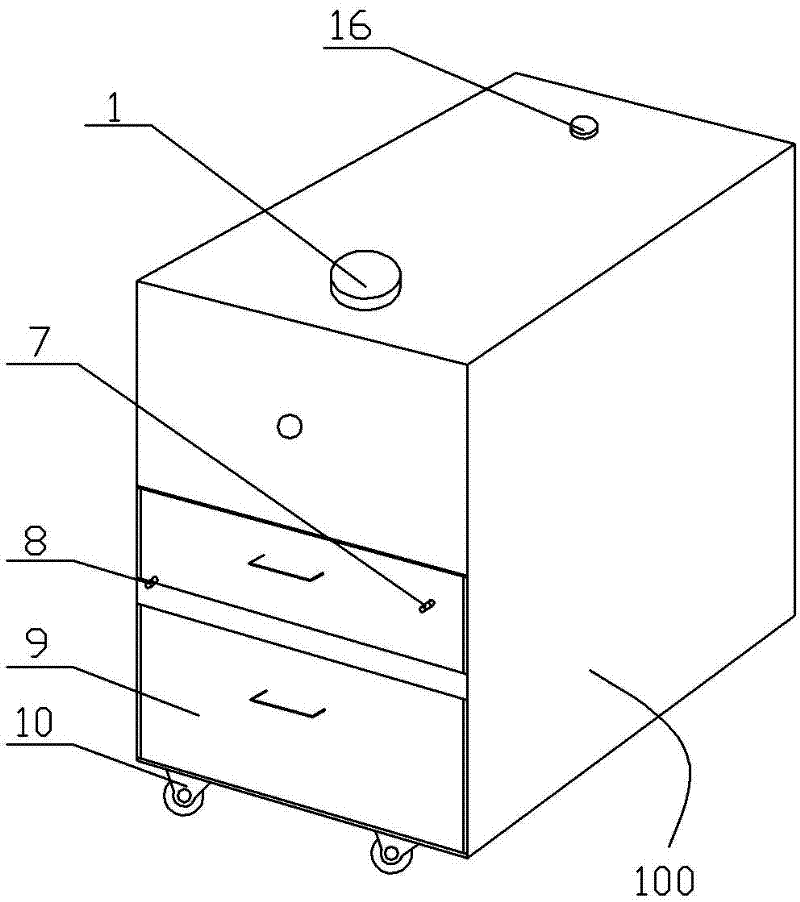
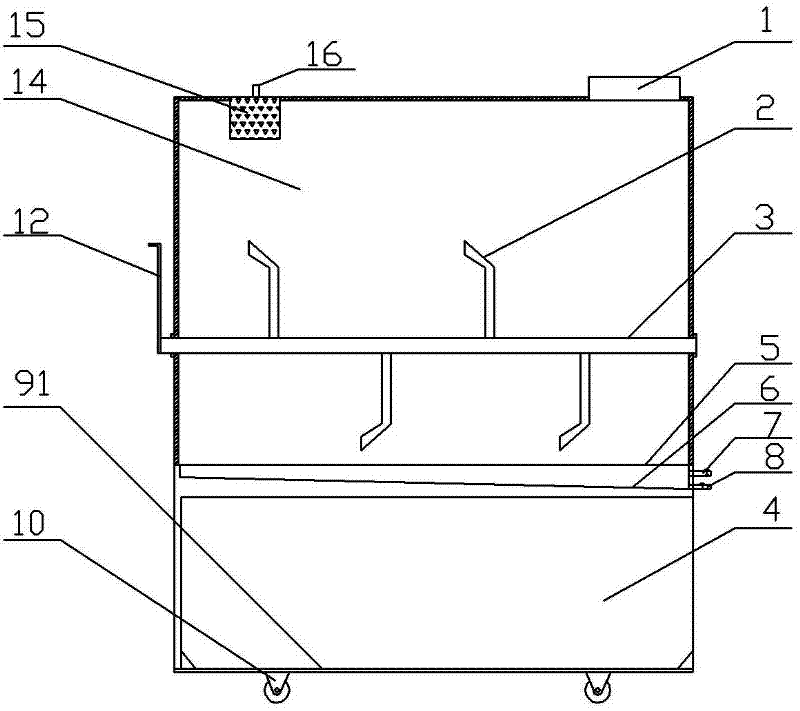
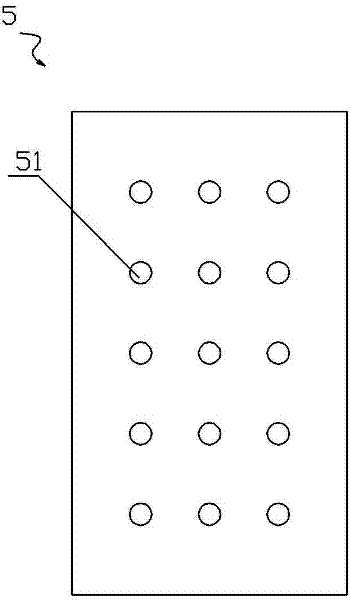
Natural gas hydrate water discharge gas production exploitation device and exploitation method of natural gas hydrate water discharge gas production exploitation device
ActiveCN103410488ASpeed up decompositionReduce moisture contentConstructionsFluid removalElectricityVapor–liquid separator
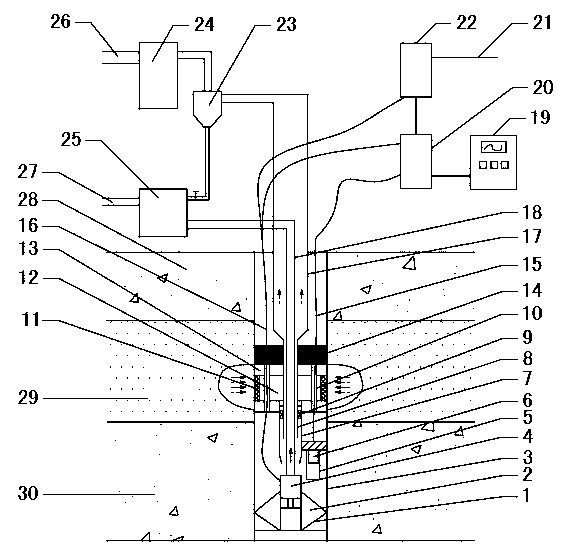
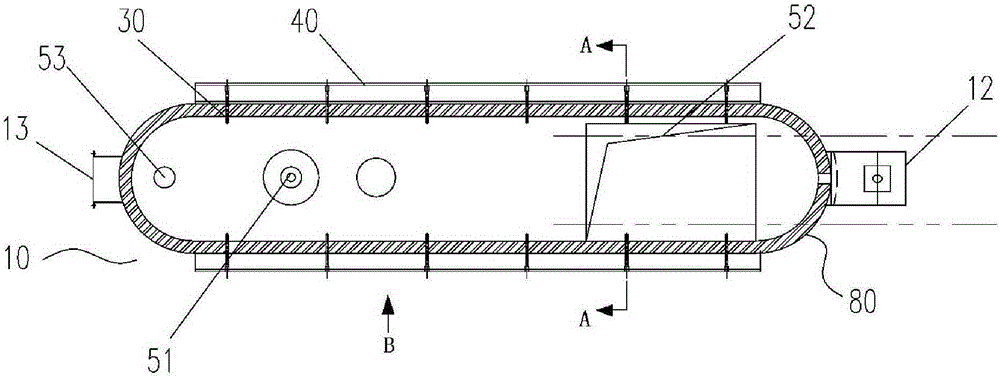
The invention provides a natural gas hydrate water discharge gas production exploitation device and an exploitation method of the natural gas hydrate water discharge gas production exploitation device, and belongs to the technical field of natural gas hydrate exploitation. A feeding cavity (12) communicated with a natural gas hydrate layer (29) is connected with an in-well gas-liquid separator (7), an exhaust cavity (8) of the in-well gas-liquid separator (7) is communicated with a gas production sleeve pipe (17), and a water discharge opening of the in-well gas-liquid separator (7) is communicated with a liquid storage cavity (2) arranged at the lower part. According to the exploitation method adopting the device, the natural gas hydrate layer (29) is firstly heated, the in-well gas-liquid separator (7) is utilized for separating gas-water mixture generated in the natural gas hydrate decomposition process, meanwhile, a liquid level monitoring device is utilized for regulating the water discharge speed of an electric submersible pump (4), and the decomposition speed of the natural gas hydrate is controlled. The device and the method have the advantages that the natural gas hydrate exploitation well liquid accumulation can be effectively prevented, the gas production efficiency is improved, the work is safe and reliable, the environment is protected, the service life is long, and the like.
Owner:CHINA UNIV OF PETROLEUM (EAST CHINA)
Metal alloy catalyst for formaldehyde purification and preparation method thereof
ActiveCN104646029AReduce dosageSpeed up decompositionDispersed particle separationMetal/metal-oxides/metal-hydroxide catalystsCatalytic oxidationBULK ACTIVE INGREDIENT
The invention discloses a metal alloy catalyst for formaldehyde purification and a preparation method thereof. The catalyst takes a metal alloy as an active center and takes a metal oxide as a carrier; the metal alloy is formed by compounding a precious metal A and a transition metal B; the precious metal A refers to any one of Pt, Ru, Rh, Pd and Ag; and the transition metal B refers to any one or a mixture of at least two out of Fe, Ni, Mn and Co. The preparation method comprises the following steps: synthesizing composite precious metal alloy nanoparticles by taking a precursor solution of the precious metal and the transition metal as a raw material under the CO atmosphere; loading the nanoparticles serving as active ingredients onto the carrier, reducing by virtue of hydrogen or a reducing solution to obtain the metal alloy catalyst. According to the catalyst disclosed by the invention, the indoor low-concentration harmful gas such as formaldehyde can be subjected to catalytic oxidation so as to prepare nontoxic and harmless CO2 and H2O under the room temperature conditions without light, electricity and other external auxiliary equipment, so that green purification of the formaldehyde is realized, and the catalyst has high stability.
Owner:HUIZHOU RES INST OF SUN YAT SEN UNIV
User abnormal behavior detection method based on neural network clustering
ActiveCN105224872AFacilitate accurate diagnosisImprove signal-to-noise ratioCharacter and pattern recognitionPlatform integrity maintainanceHidden layerNerve network
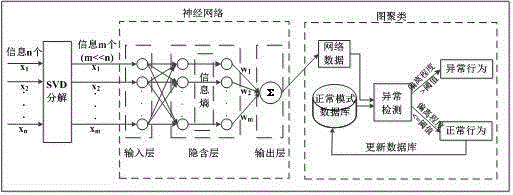
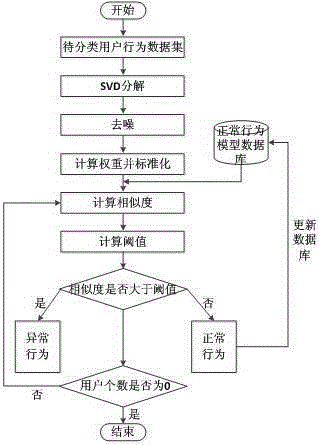
The invention discloses a user abnormal behavior detection method based on neural network clustering. The method comprises: firstly, performing SVD decomposition and denoising on a behavior data set matrix of a user, and inputting the denoised matrix to an input layer of a neural network, and then calculating weight of all attributes of each user in a hidden layer of the neural network, and standardizing the weight, outputting the weight in an output layer, and finally respectively calculating similarity value of each user and each user in a normal behavior model database and a threshold value, if the similarity value is larger than the threshold value, behavior being abnormal behavior, and carrying out corresponding prompt and precautionary measures, otherwise, the behavior being normal behavior, and combining the behavior in the normal model database to upgrade the database in real time. Through cooperation of each part, the method effectively realizes high detection rate and low false alarm rate.
Owner:HENAN UNIV OF SCI & TECH
Microbial straw rotting agent and preparation method thereof
The invention provides a microbial straw rotting agent, which adopts the strains having mutual synergistic action without generating antagonistic action as production strains: cellulase preparation, Fomes annosus, Bacillus subtilis, Thermobifida fusca, Bacillus pumilus and Saccharomyces cerevisiae. The microbial straw rotting agent has high decomposition effect on the main components of the straws, has biological control function, and can inhibit growth of crop sheath blight, rice blast, wheat root rot, strawberry gray mold and other pathogens. The product has the advantages of short manufacturing period, high production efficiency and low cost, and is safe and convenient to use.
Owner:安徽莱姆佳生物肥业有限公司
Cleaning control method and control device for air conditioner and air conditioner
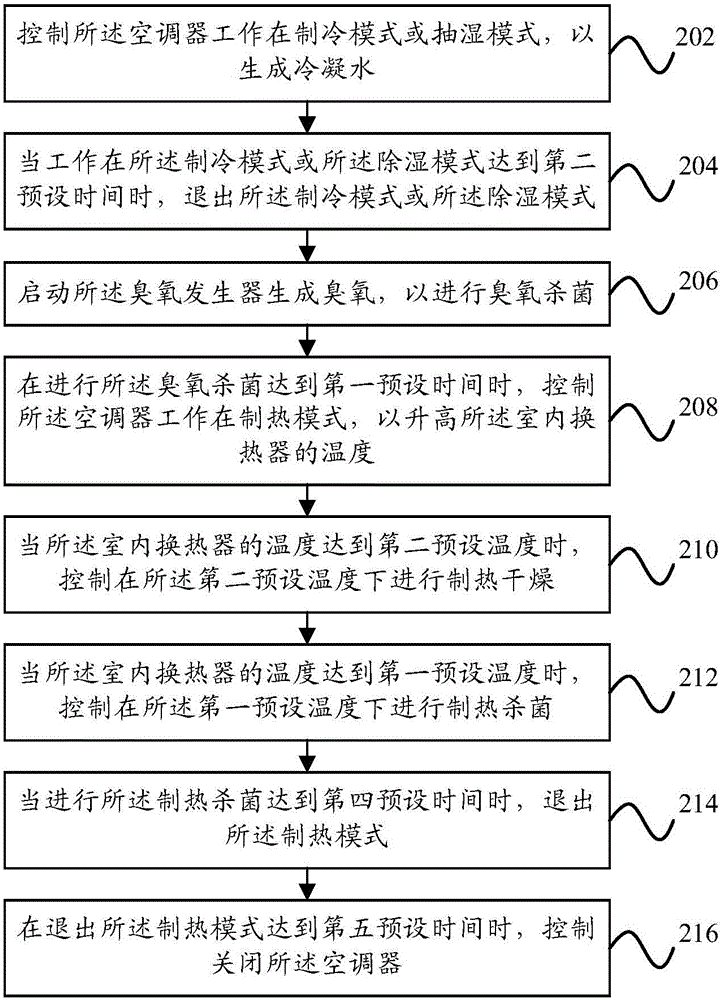
InactiveCN106123226ASpeed up decompositionImprove comfort experienceMechanical apparatusSpace heating and ventilation safety systemsEngineeringOzone Sterilization
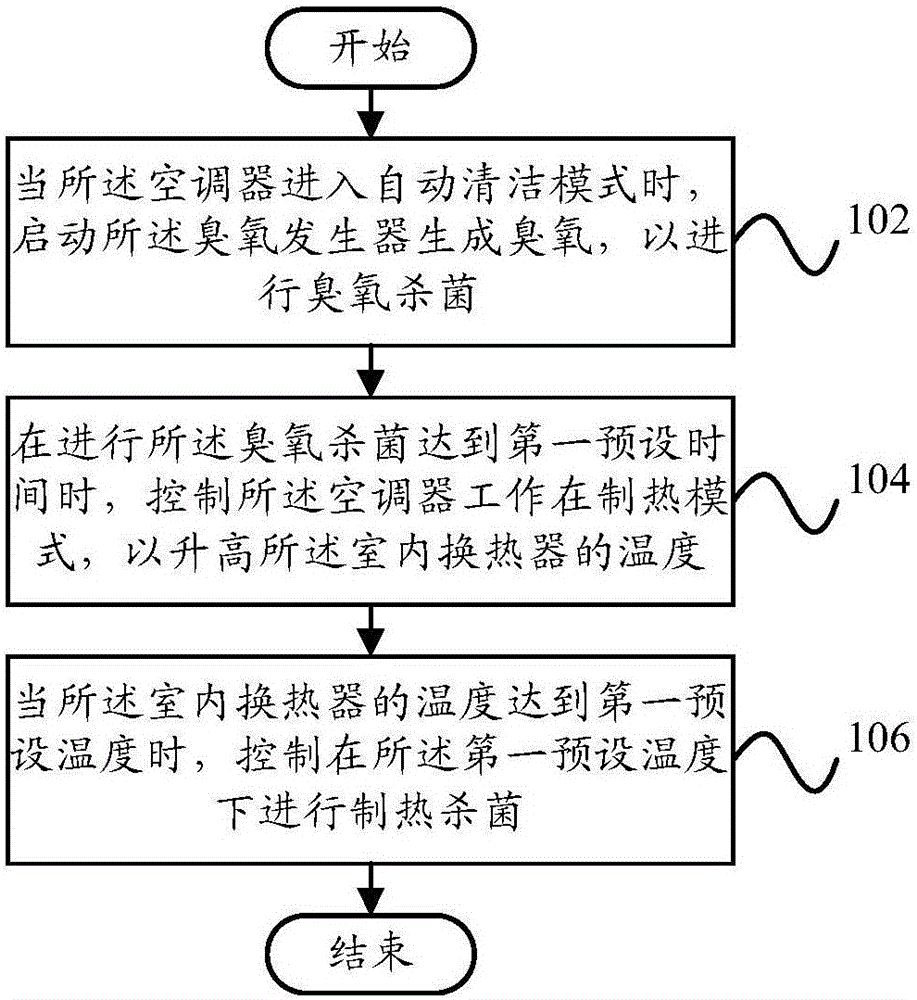
The present invention provides a cleaning control method for an air conditioner, a cleaning control device for the air conditioner, and an air conditioner. The air conditioner includes an ozone generator, an indoor heat exchanger, a damper, and an indoor fan. The cleaning control method includes: when When the air conditioner enters the automatic cleaning mode, the ozone generator is activated to generate ozone for ozone sterilization; when the ozone sterilization reaches a first preset time, the air conditioner is controlled to work in the heating mode to Raise the temperature of the indoor heat exchanger; when the temperature of the indoor heat exchanger reaches a first preset temperature, control to perform heating and sterilization at the first preset temperature. Through the technical solution of the present invention, the cleaning and sterilization effect of the air conditioner can be effectively improved, and thorough and effective sterilization can be realized, thereby improving the user's comfortable experience.
Owner:GD MIDEA AIR-CONDITIONING EQUIP CO LTD +1
Quick LU factorization method for circuit sparse matrix in circuit simulation
ActiveCN102142052ASpeed up decompositionShorten the timeSpecial data processing applicationsAlgorithmLU decomposition
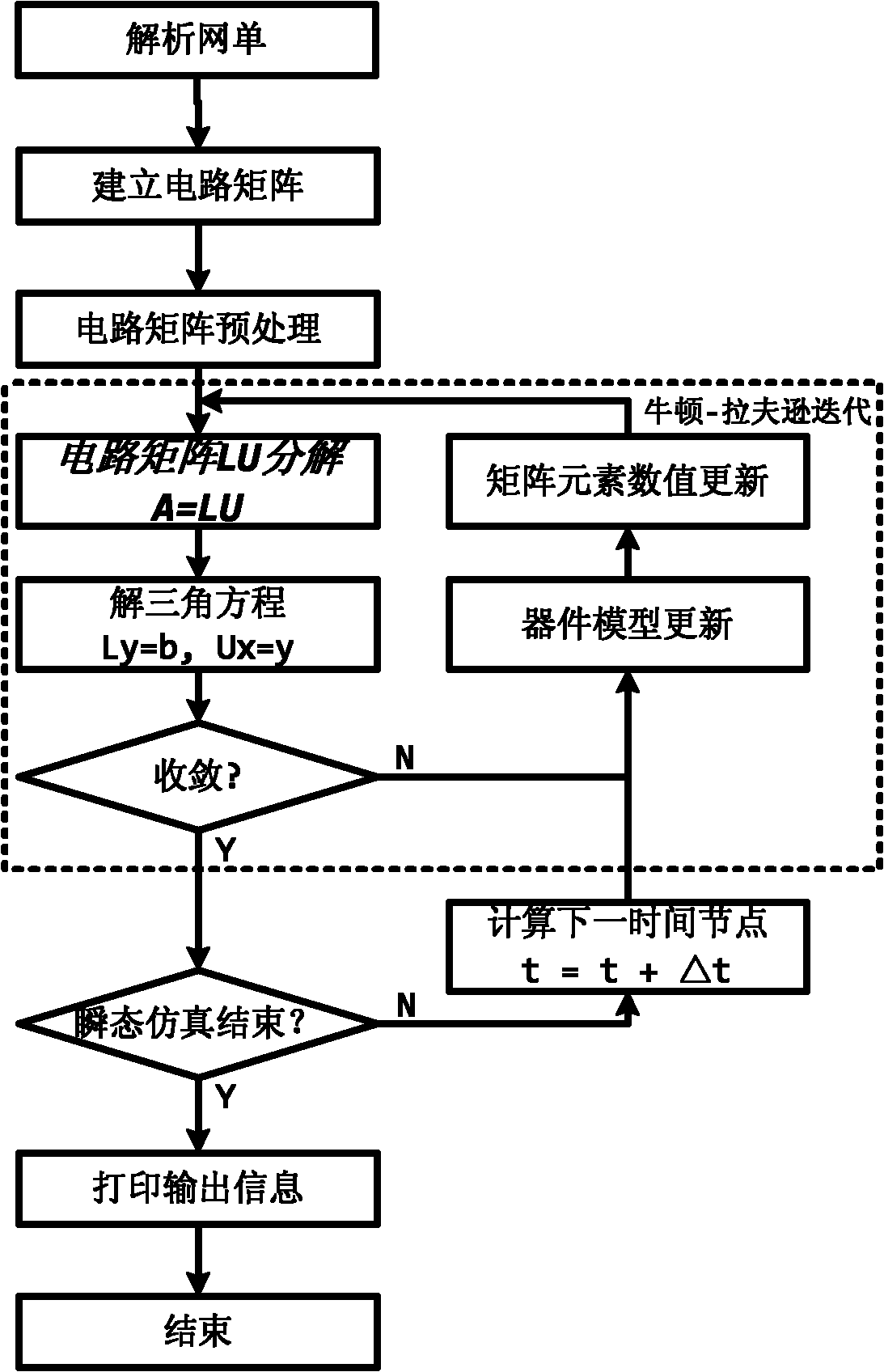
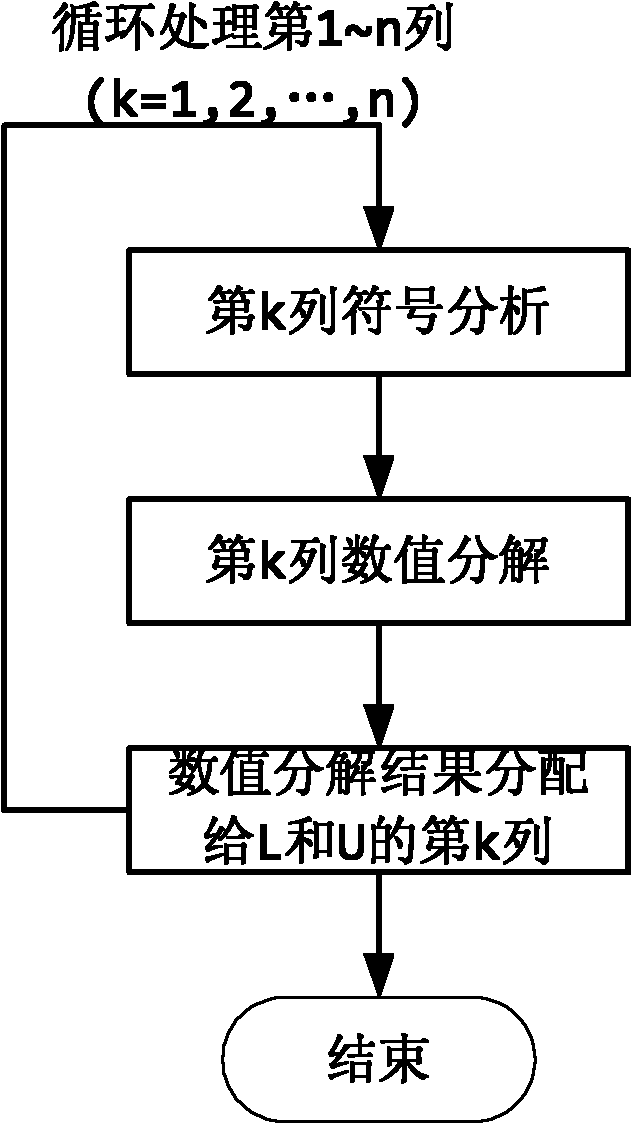
The invention relates to a quick LU factorization method for a circuit sparse matrix in circuit simulation, and belongs to the technical field of electronic design automation (EDA). The method comprises the following steps of: symbolic analysis and the calculation of LU factorization, wherein in the step of the symbolic analysis, a preprocessed matrix is analyzed to forecast each row of a non-zero structure of matrixes L and U formed after the matrix is subjected to the LU factorization; and in the step of the calculation of the LU factorization, based on the non-zero structures, obtained by the symbolic analysis, of the matrixes L and U, each row of the matrix is subjected to numerical solving and numerical distribution to obtain the matrixes L and U after the LU factorization. The method is characterized in that the symbolic analysis is separated from the circulation of the LU factorization, and the symbolic analysis is executed beyond the circulation of Newton-Raphson iteration in the process of the circuit simulation, namely is executed once, so the complexity of the circuit simulation can be reduced effectively, and the speed of the LU factorization can be improved to accelerate the integral circuit simulation.
Owner:TSINGHUA UNIV
Efficient straw-decomposing inoculant and preparation method thereof
ActiveCN103031254AAccelerate the rate of decaySpeed up decompositionFungiBacteriaPaecilomyces lilacinusGeoponic
The invention belongs to the technical field of microorganism, in particular to a compound microbial preparation used for directly decomposing crop straws, namely an efficient straw-decomposing inoculant. The efficient straw-decomposing inoculant comprises the following raw materials by weight percentage: 20-30 percent of bacillus subtilis, 10-15 percent of white light gray streptomycete, 5-10 percent of endomycopsis fibuligera, 5-10 percent of candida utilis, 10-15 percent of aspergillus, 10-15 percent of paecilomyces lilacinus, 10-15 percent of trichoderma harzianum and 20-30 percent of thermophilus streptomycete. The efficient straw-decomposing inoculant is organically combined with soil no-tillage, shifting and fallow to achieve the comprehensive purposes of protecting the environment, improving soil, increasing the utilization rate of fertilizers, increasing the quantity and the quality of agricultural products and developing the ecological agriculture.
Owner:鹤壁市人元生物技术发展有限公司
Preparation method of natural high-fiber odorless cattle and sheep feed
InactiveCN105685486AImprove internal environmentImprove the environmentFood processingAnimal feeding stuffFiberPassion fruit
The invention relates to a preparation method of a natural high-fiber odorless cattle and sheep feed. The prepared mixed probiotic dry powder and chopped straw feed are mixed and fermented for more than 10 days to obtain the product; the straw feed is bagasse or sugarcane leaves, cornstalks or corncobs, green pasture and mulberry stems; and the fermentation probiotics comprises saccharomyces cerevisiae, EM (Effective microorganisms) and bacillus subtilis. The preparation method comprises the steps of: mixing and crushing the following components of the feed in parts by weight: 20-30 parts of soybean meal, 10-20 parts of peanut bran, 5-10 parts of passion fruit leaf, 15-20 parts of mulberry stem, 1-3 parts of momordica grosvenori and 10-20 parts of corn; mixing with 10-25 parts by weight of running water and stirring for 15min; pouring the prepared mixed probiotic raw powder and trichoderma viride in a ratio of 1kg / t; barreling and sealing; fermenting naturally for 3-7 days; and when the fermented product emits sweet fragrance, pouring the fermented product into a stirrer, and adding 20-40 parts by weight of rice bran and stirring uniformly to obtain the fermented probiotic dry powder.
Owner:刘健
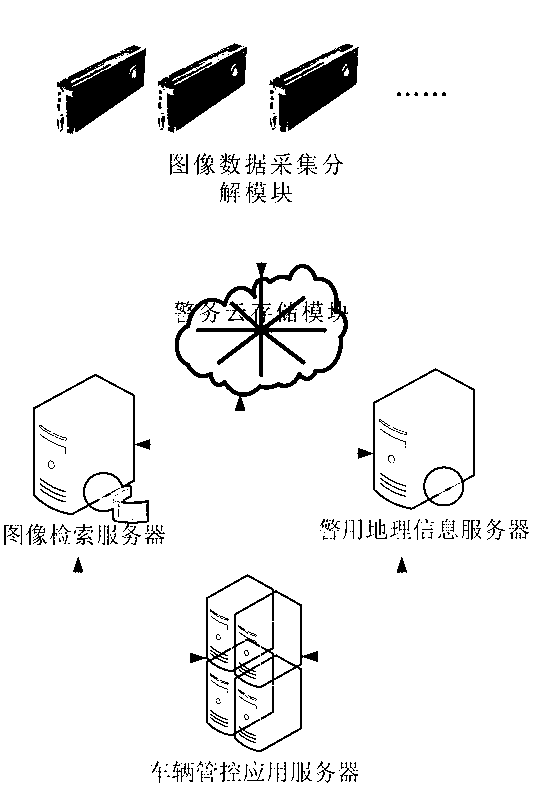
Police service cloud image recognition vehicle management and control system based on geographic space-time constraint
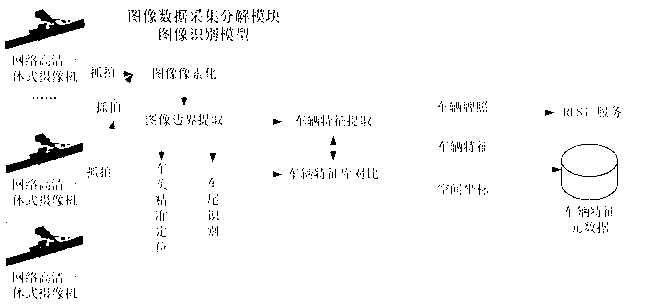
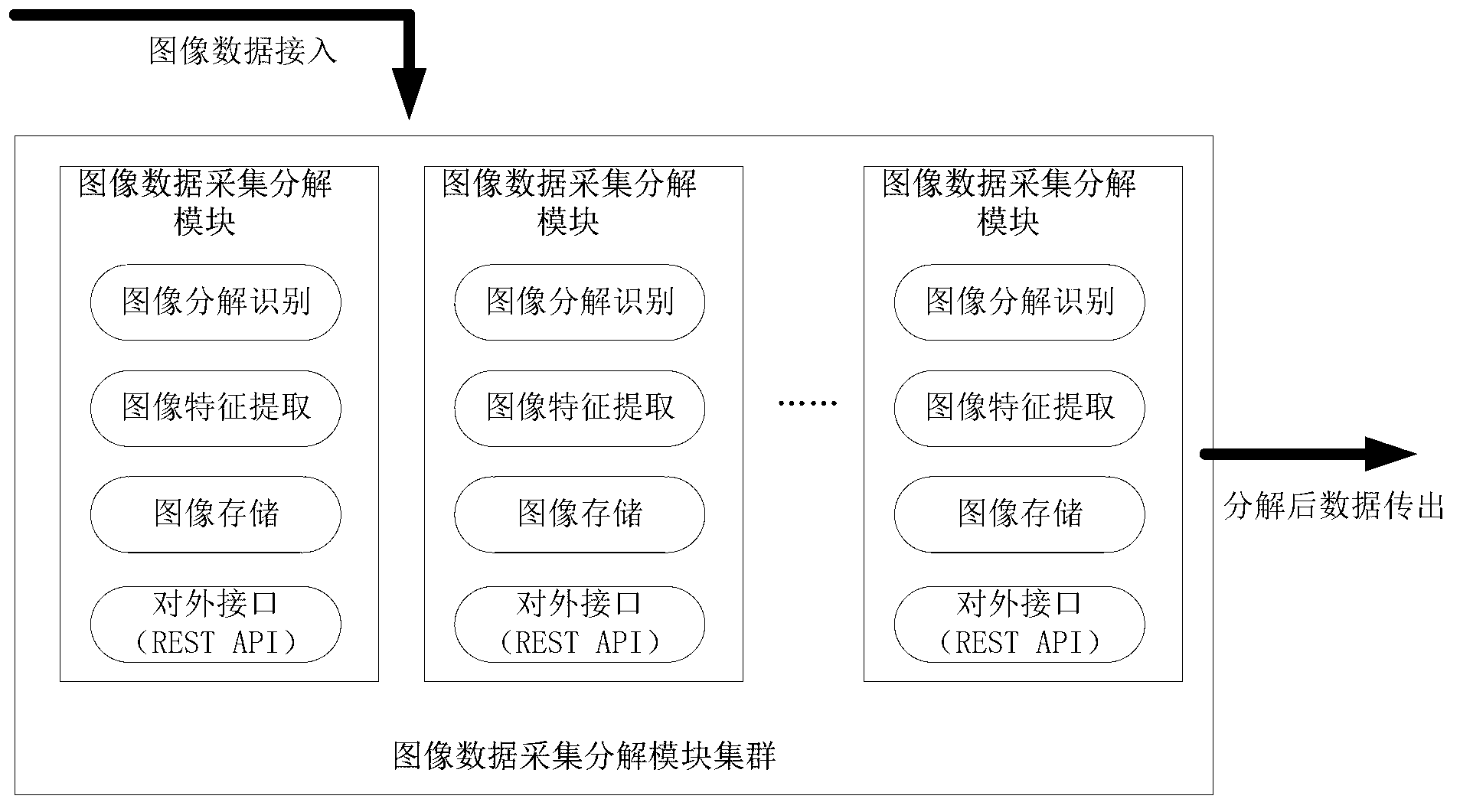
ActiveCN103294775ASpeed up decompositionReduce computing pressureCharacter and pattern recognitionSpecial data processing applicationsDecompositionImage retrieval
The invention discloses a police service cloud image recognition vehicle management and control system based on geographic space-time constraint. The system comprises an image data collection and decomposition module, an image retrieval module, a data storage module and a geographic information service module. The system is characterized in that the image data collection and decomposition module is responsible for collecting road images, extracting vehicle characteristics in the images and transmitting vehicle characteristic information in the images to the data storage module, the data storage module is used for storing the road images and image characteristic element data concerning the vehicle characteristic information in the images, the image retrieval module is used for responding to image retrieval requests of requesters, acquiring retrieval results meeting the image retrieval requests in an image retrieval mode mixing the image characteristic element data with geographic space-time constraint according to the image retrieval requests of the requesters and feeding the retrieval results back to the requesters, and the geographic information service module is used for responding to vehicle positioning requests of the requesters, acquiring vehicle space position information through a map server served by a geographic information platform and feeding the vehicle space position information back to the requesters.
Owner:SUZHOU XIANGYI NETWORK TECH
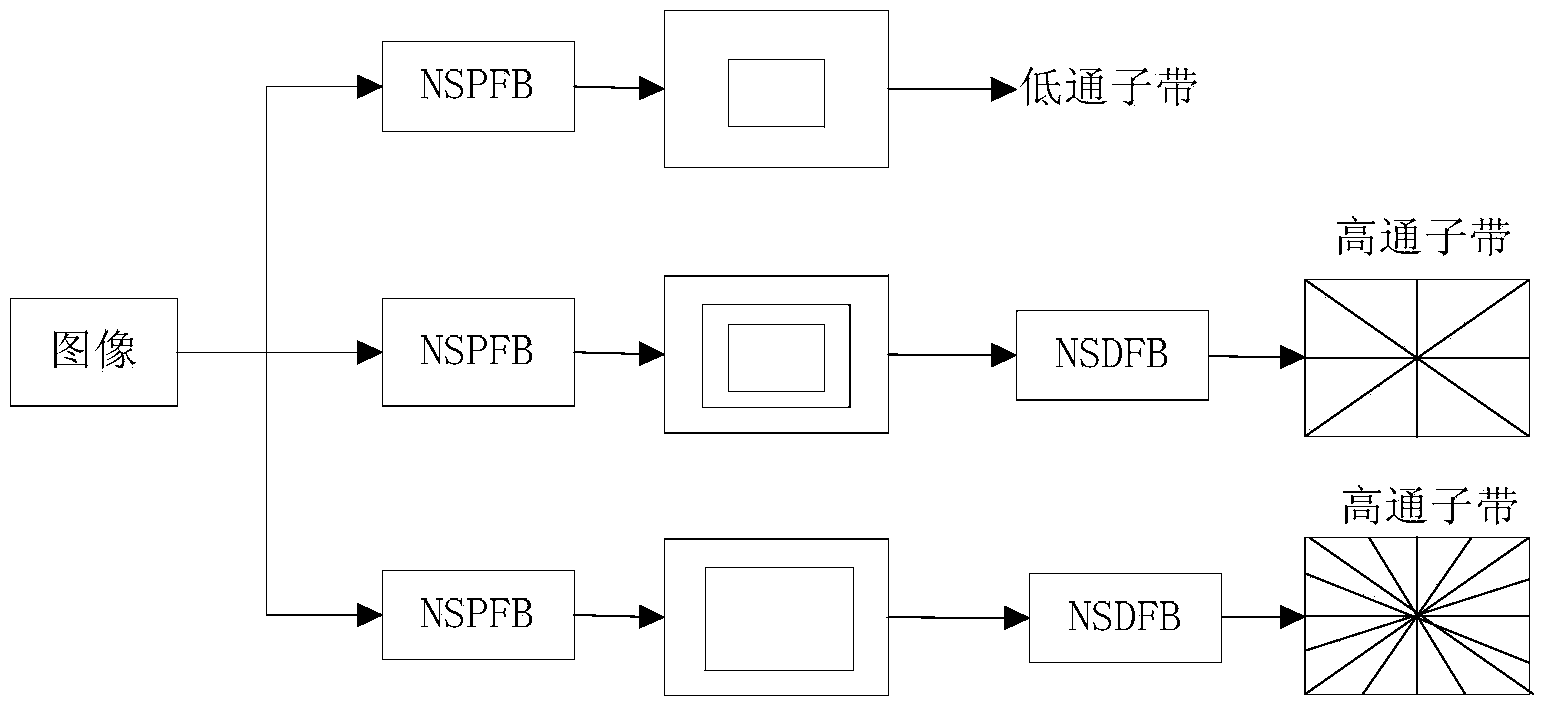
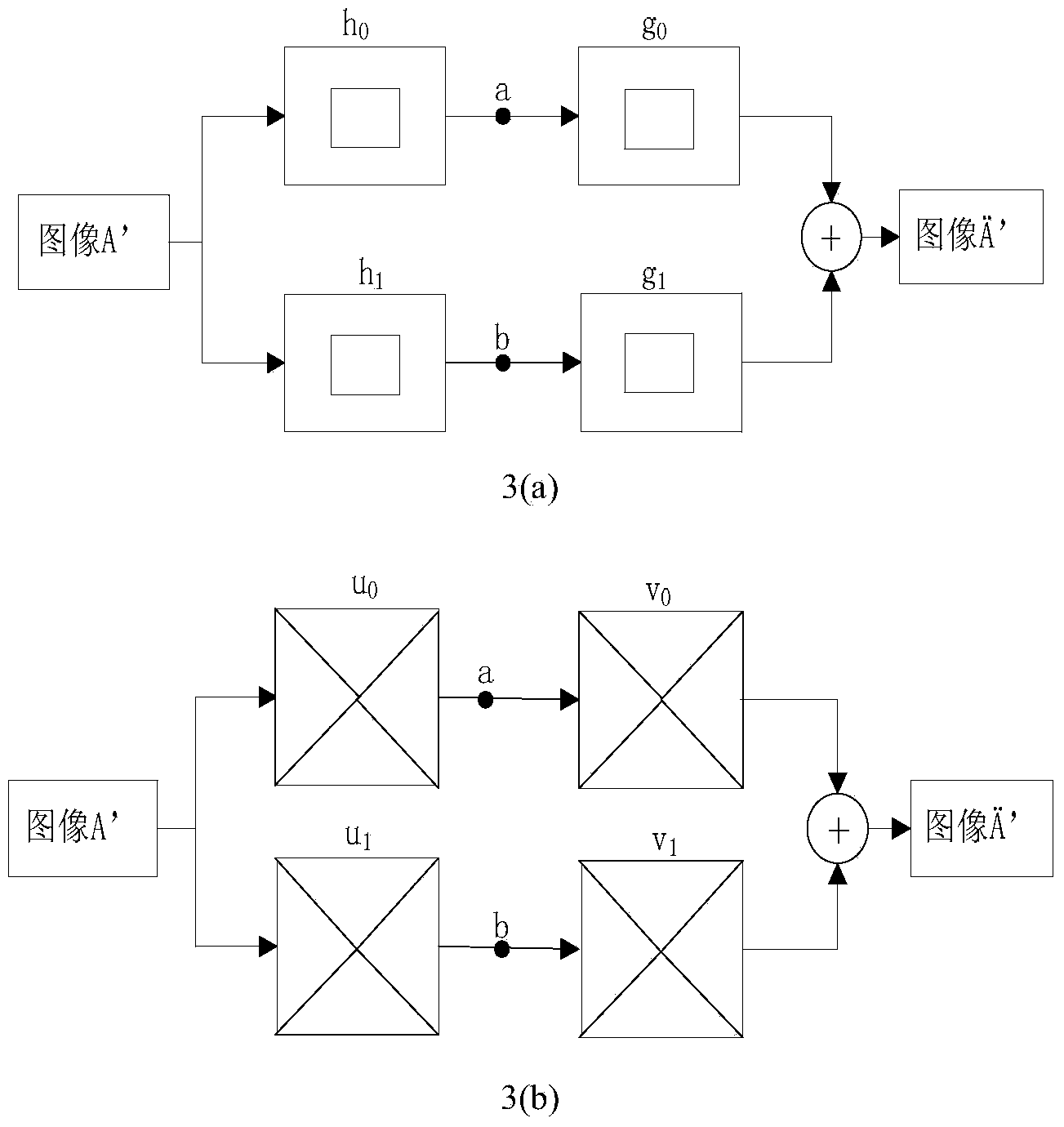
Contourlet transformation-adaptive medical image fusion method based on non-sampling
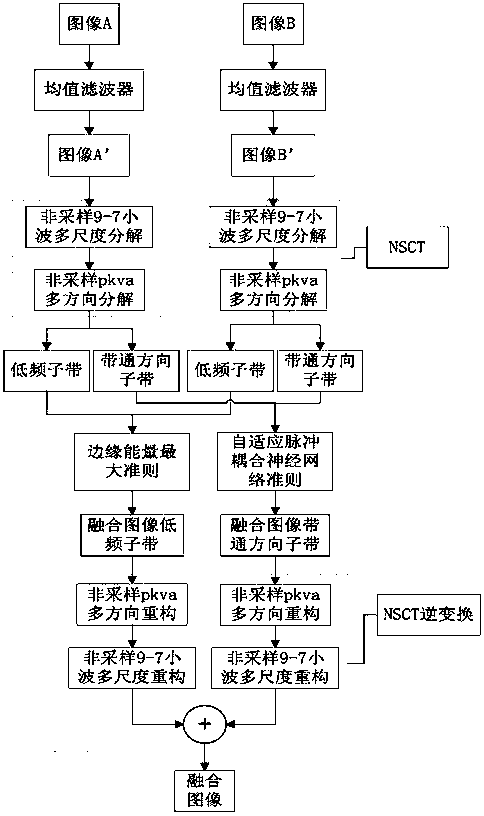
InactiveCN104282007ALimited universalityFast decompositionImage enhancementGeometric image transformationImaging processingContourlet
The invention relates to a contourlet transformation-adaptive medical image fusion method based on non-sampling and belongs to the field of image processing. The method comprises the steps that firstly, a source image is subjected to arithmetic average filtering and then is decomposed through an orthogonal 9-7 wavelet filter and a pkva filter during non-sampling to obtain low-frequency sub-band coefficients and all band-pass direction sub-band coefficients; secondly, the low-frequency sub-band coefficients are selected and fused according to the edge information maximum criterion, all the band-pass sub-band coefficients are selected and fused through an adaptive PCNN model based on a visual neuron model; lastly, a final fused image is obtained by means of inverse transformation of NSCT. According to the contourlet transformation-adaptive medical image fusion method based on non-sampling, the algorithm is very effective and correct, the edge and space texture information of the fused image is clear, color distortion is low, the false contour phenomenon does not exist, and feature information of the source image is well reserved.
Owner:CHANGCHUN UNIV OF SCI & TECH
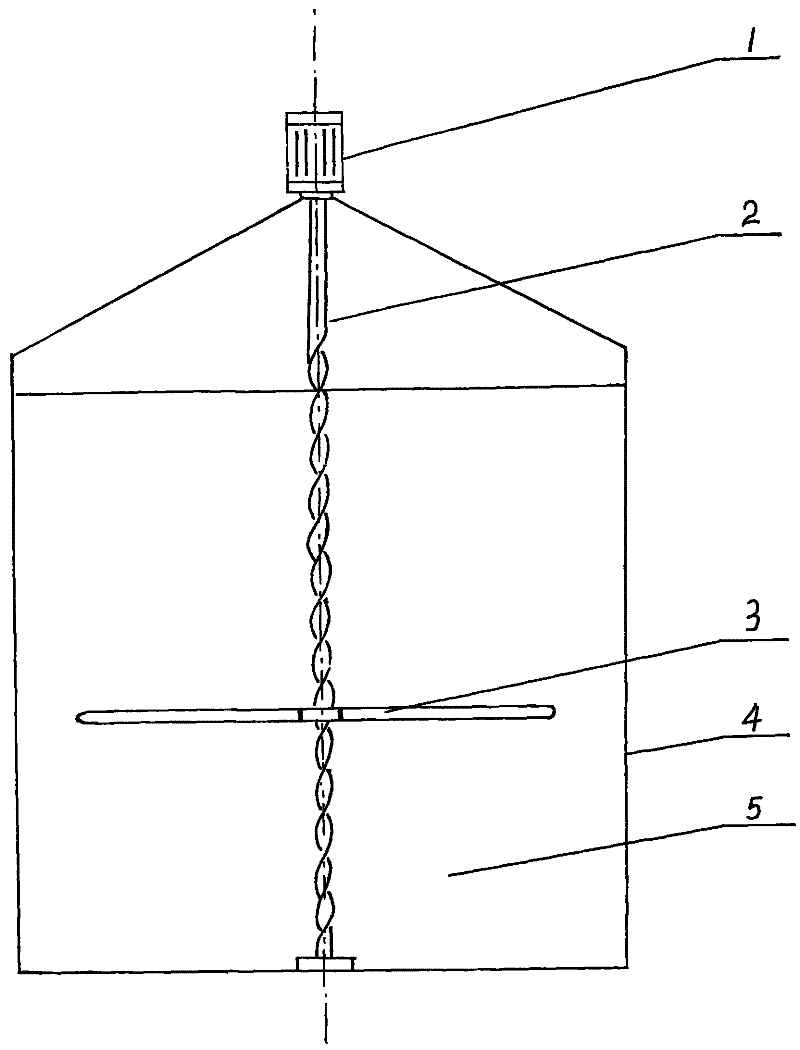
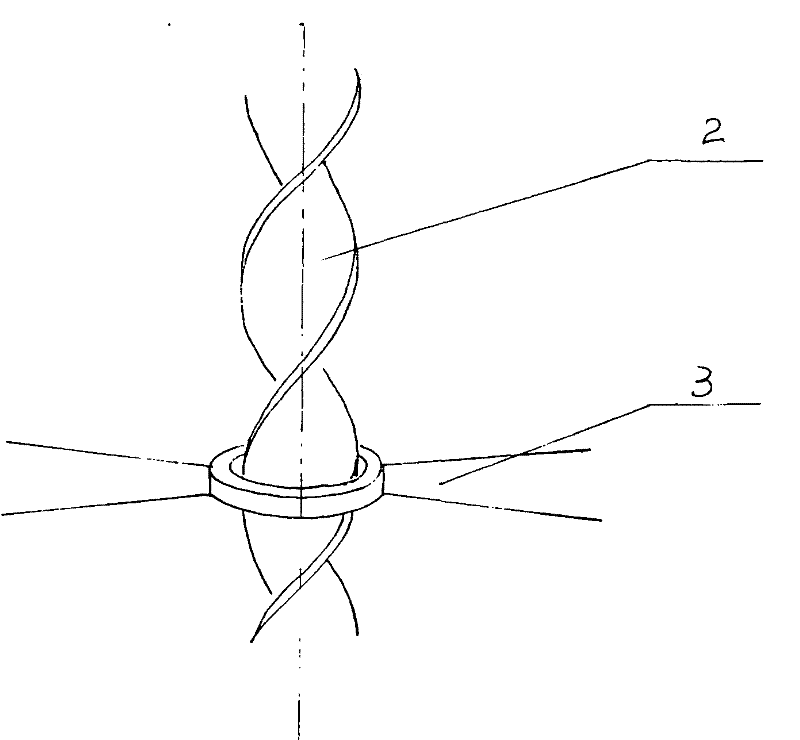
Paddle floating-type stirring machine
InactiveCN102188922ASimple structureEasy to processRotary stirring mixersMaterial distributionMixed materials
The invention relates to a paddle floating-type stirring machine. The stirring machine mainly comprises a variable frequency motor (1), a spiral shaft (2), a floating-type stirring paddle (3), wherein the variable frequency motor (1) and the spiral shaft (2) are in a fixed connection state, the spiral shaft (2) and the floating-type stirring paddle (3) are assembled and are in a free sliding state along the spiral shaft (2). The stirring machine is mainly arranged in a fermentation tank body (4) or other containers and is used for uniformly distributing and stirring solid-liquid mixed materials (5); the floating-type stirring paddle (3) can move up and down along the spiral shaft (2) along with variation of rotating speed of the variable frequency motor (1), thus the problem that materials can not be stirred or uniformly distributed at different parts as required as the paddle is fixed in the traditional stirring machine can be effectively solved; and the problems that gas production is influenced, material distribution is nonuniform and stirring is nonuniform as the materials crust in the fermentation tank body (4) or containers can be effectively solved, the structure is compact, the stirring effect is full, and the product cost is low. The stirring machine provided by the invention is mainly used in fermentation and related industries.
Owner:董保成 +1
Environment-friendly straw fermenting agent
InactiveCN102517217APromote growth and developmentHigh in nutrientsFungiBacteriaCelluloseStreptomyces hygroscopicus
The invention relates to an environment-friendly straw fermenting agent. The environment-friendly straw fermenting agent is prepared from the following seven raw materials in percentage by weight: 25 percent of bacillus subtilis, 15 percent of trichoderma viride, 18 percent of saccharomyces cerevisiae, 12 percent of aspergillus niger, 10 percent of streptomyces hygroscopicus, 10 percent of trichoderma koningii and 10 percent of arthrobacter. The environment-friendly straw fermenting agent can accelerate the decomposition speed of cellulose and can completely decompose crop straws within aboutten days to return to the field, so that the straws are corroded, blackened and composted. The environment-friendly straw fermenting agent can accelerate release of nutrient elements in the straws, provides more nutrients for growth of the crops at the later stage, maintains and improves soil fertility, improves soil organic matter, improves physicochemical property of the soil, increases the yield of the crops by about 10 percent, and can be widely applied to returning straws of the crops such as rice, wheat, corn and the like to the field.
Owner:宋保德
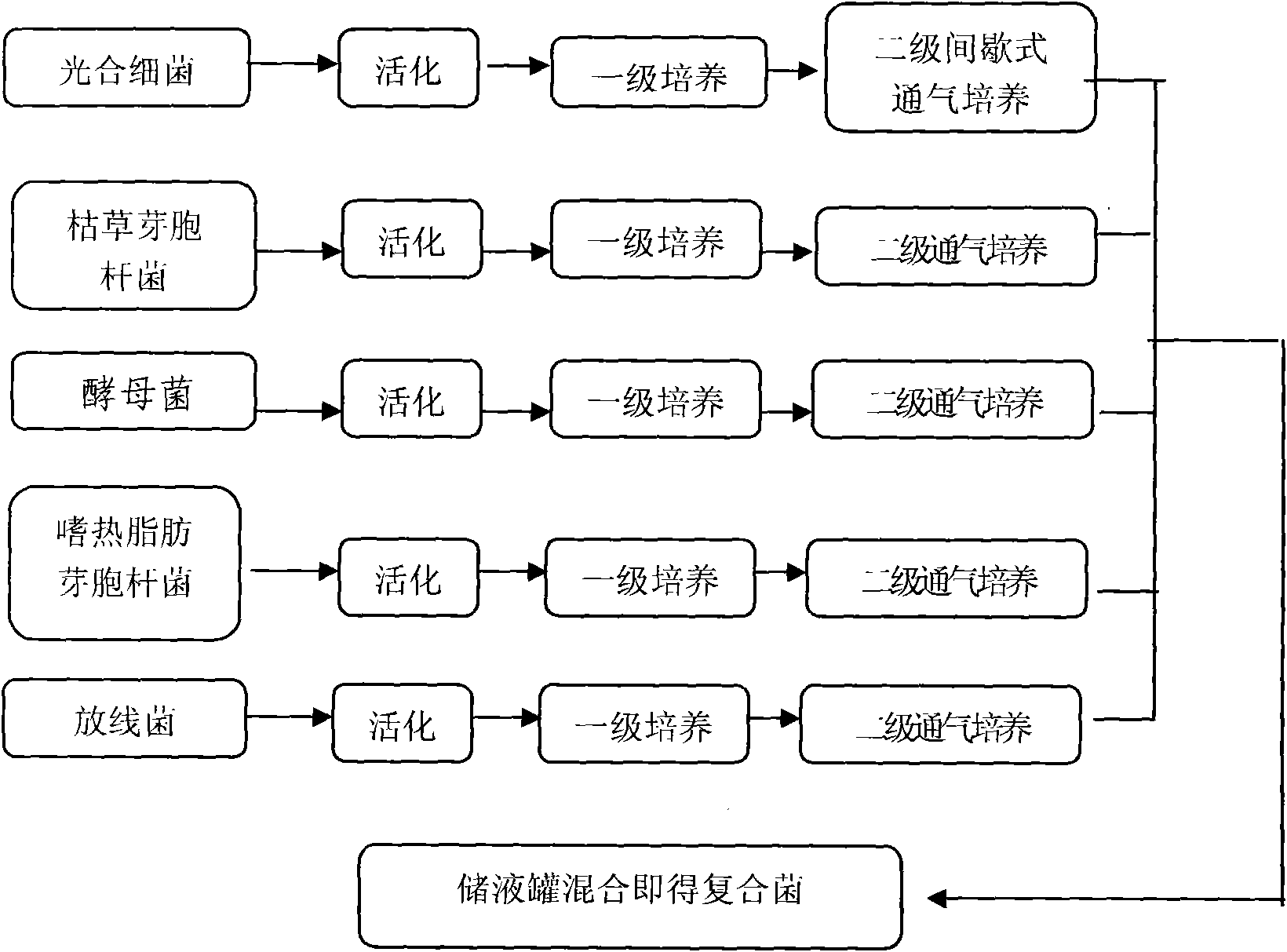
Microorganism fertilizer for promoting rice production, preparation method and application thereof
The invention also relates to a microorganism fertilizer. The composite microorganism preparation comprises the components based on the volume ratio: 5-15% of photosynthetic bacteria, 10-20% of bacillus subtilis, 15-35% of saccharomycete, 25-45% of thermophilic bacillus and 3-15% of actinomycete. The invention also relates to the application of the microorganism fertilizer in the aspects of improving the resistance of seeds, preventing plant diseases and insect pests, purifying the water quality of rice field and increasing the rice production, and a using method thereof. The invention also relates to the application of the microorganism fertilizer in the aspect of accelerating rice stubble rotten degree, and a using method thereof.
Owner:辽宁三色微谷科技有限公司
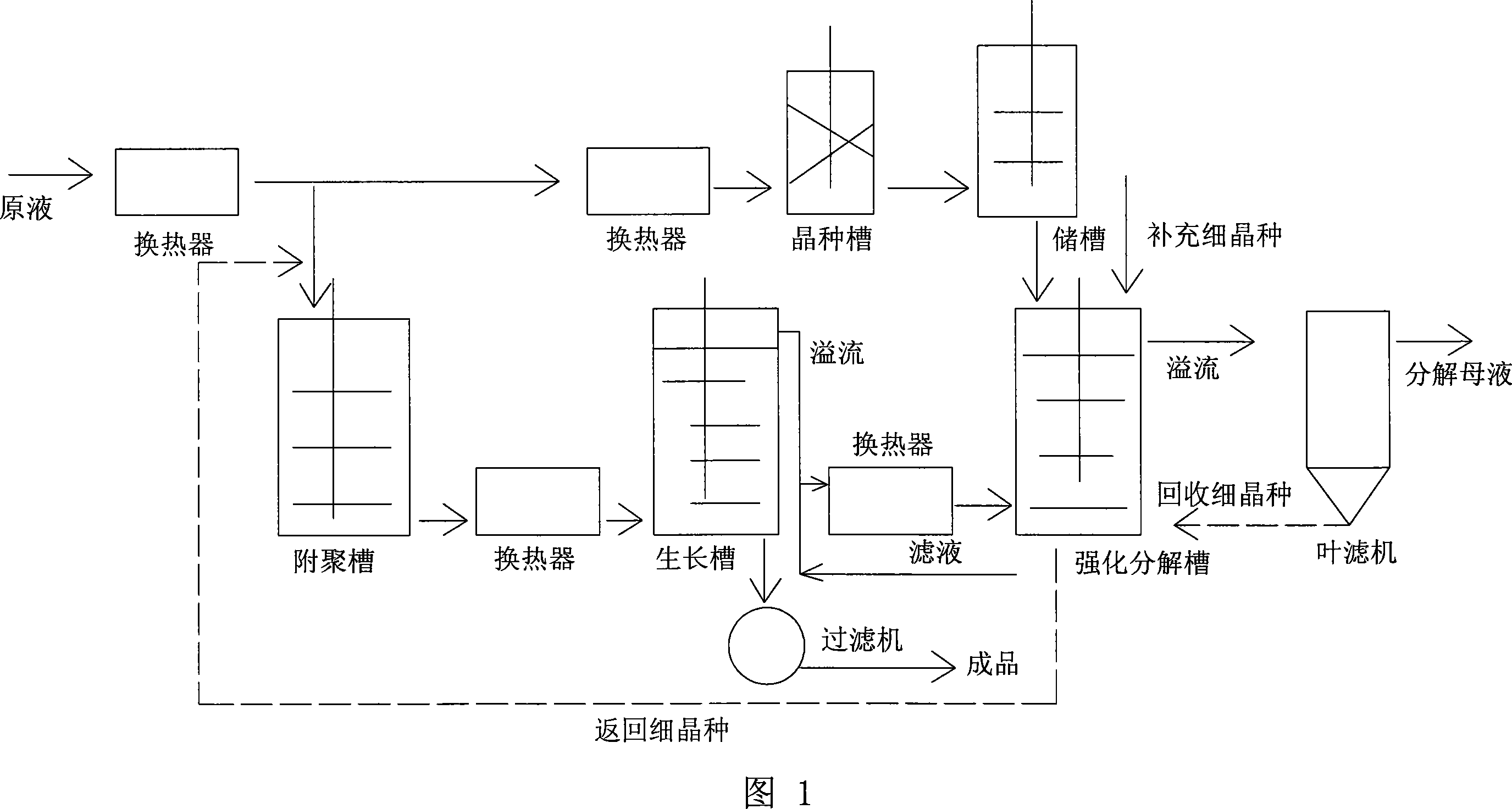
Three stages, two seeds technique of seed crystal decomposition
ActiveCN101092242AGood qualityControllable processSolution crystallizationAlkali-metal aluminates/aluminium-oxide/aluminium-hydroxide preparationDecomposerChemistry
This invention relates to a three-stage two-seed seed crystal decomposing process. The process comprises: passing the mother liquor through an agglomeration decomposer and a seed crystal growth decomposer in order, adding decomposing seed crystal into the seed crystal growth decomposer, then passing through a reinforced decomposer, adding fine seed crystal into the reinforced decomposer, sending the overflow liquid of the reinforced decomposer to a leaf filter to obtain decomposed mother liquor, recovering fine seed crystal from the leaf filter, returning to the reinforced decomposer, returning seed crystal from the reinforced decomposer to the agglomeration decomposer, and sending part of the mother liquor via the seed crystal growth decomposer to the reinforced decomposer for circulation. The process has such advantages as reasonable procedures, low energy consumption, low cost, easy control, high decomposition rate and high product quality.
Owner:GUIZHOU BRANCH CHINA ALUMINUM IND
Water-soluble asymmetric indocyanine fluorescent dye and preparation method thereof
InactiveCN103073909AEasy to separateGood water solubilityMethine/polymethine dyesLuminescent compositionsAcetic anhydrideFiltration
The invention discloses a water-soluble asymmetric indocyanine fluorescent dye. A general formula of the dye is as shown in the specification, wherein n is equal to 1, 2 or 3. A preparation method comprises the steps that (1), N-(3,5-2(2-(2-methoxyethoxy) oxethyl) oxethyl) benzyl-2,3,3-trimethyl-5-sulfonic group-3H-indole is synthesized; 2,3,3-trimethyl-5-sulfonic group-3H-indole, organic sylvine, and 3,5-2(2-(2-methoxyethoxy) oxethyl) oxethyl) oxethyl benzylbromide are added to an alcohol solvent in sequence, and conduct backflow reaction for 20-40h under inert gas shielding; the solvent is distilled off after the reaction; column separation is conducted; a red solid product is obtained; and (2), N-carboxyl benzyl-2,3,3-trimethyl-5-sulfonic group-3H-indole, the product and a condensing agent are dissolved in an acetic acid / acetic anhydride mixed solvent; pyridine or pyrimidine is added, and reacts at 60-100 DEG C for 1-6h under the inert gas shielding; ethyl acetate is added after the reaction for dissolving out a precipitate; suction filtration is conducted; the product is separated by taking a methanol / acetonitrile mixed solvent as an eluent, and using a reverse phase silica gel filling separation column; three color strips are obtained; the second color strip is accepted; and the water-soluble asymmetric indocyanine fluorescent dye is obtained.
Owner:HENAN UNIVERSITY
Improved type side-blowing molten pool smelting furnace
ActiveCN105698529ADry fastSpeed up decompositionIncreasing energy efficiencyCrucible furnacesSlagStraight segment
The invention provides an improved type side-blowing molten pool smelting furnace. The improved type side-blowing molten pool smelting furnace comprises a long round furnace body, a furnace cylinder, a furnace top, a furnace body frame structure and a plurality of multi-channel spray guns. The multi-channel spray guns are arranged on the two sides of the long round furnace body correspondingly. The long round furnace body is formed by a furnace body protective plate in an enclosed mode. A water sleeve is installed on the lower portion of the inner side of the furnace body protective plate. A first firebrick layer is further inlayed in the inner side of the water sleeve. A second firebrick layer is arranged on the water sleeve and the first firebrick layer and installed on the inner side of the furnace body protective plate. The furnace cylinder is arranged at the bottom of the long round furnace body. The long round furnace body is provided with a metal discharge outlet and a slag outlet. The furnace body frame structure comprises steel vertical pillars, cross beams and pulling rods. The multi-channel spray guns are arranged on the two sides of the middle straight segment of the long round furnace body, and the output ends of the multiple multi-channel spray guns extend into the long round furnace body. By means of the improved type side-blowing molten pool smelting furnace, the problem that in the prior art, the temperature in a molten pool smelting furnace is difficult to control can be solved.
Owner:CHINA ENFI ENGINEERING CORPORATION
Method for cultivating selenium-rich sweet potatoes
InactiveCN105993576ASufficient for absorptionReduce dosageAnimal corpse fertilisersExcrement fertilisersSite managementPlant Tubers
The invention belongs to the technical field of agriculture, and particular discloses a method for cultivating selenium-rich sweet potatoes. The method comprises the steps of land selection, seed selection, sowing, field management and pest control. According to field management, intertillage weeding and topdressing, reasonable irrigation and ridging are included; according to topdressing, selenium-rich organic fertilizer and selenium-rich leaf fertilizer are dressed, and 2 kg to 4 kg of the organic fertilizer and 1 kg to 2 kg of the leaf fertilizer are applied to each sweet potato. The organic fertilizer and the leaf fertilizer are applied together, growth of the sweet potatoes can be promoted, and the planting period can be shortened. During intertillage, the tubers of the sweet potatoes begin to grow, tuber growing is a guarantee of yield increasing, and therefore organic fertilizer application is critical during intertillage. According to the method, the organic fertilizer and the leaf fertilizer which can promote tuber growth and add selenium into the tubers are specially designed for the sweet potatoes, therefore, it is guaranteed that the sweet potatoes can absorb and utilize sufficient selenium in the whole growth process, the yield of the sweet potatoes is increased, and selenium in the sweet potatoes can be increased.
Owner:蒋丽红
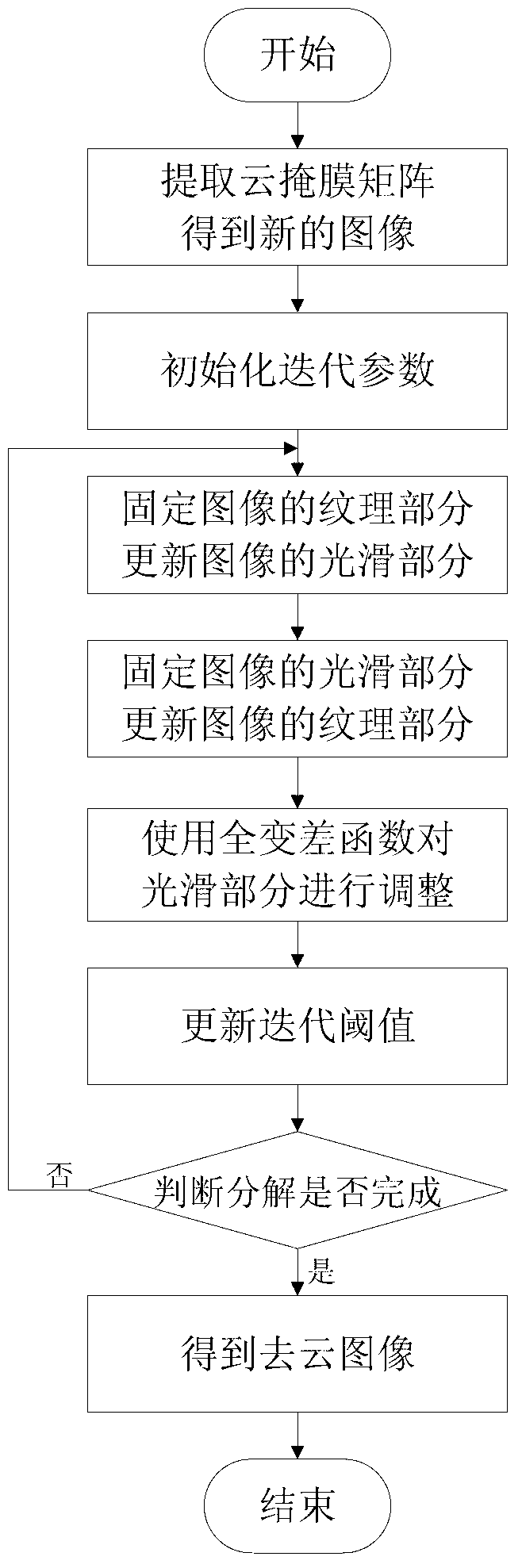
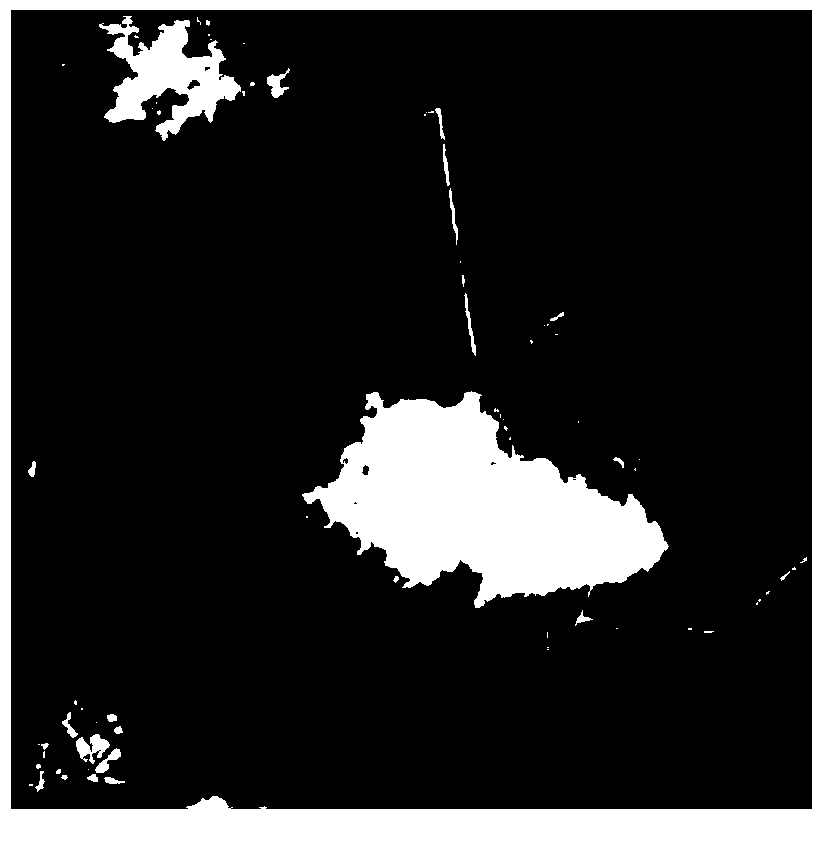
Remote sensing image cloud removing method based on sparse representation
InactiveCN102800058ASpeed up decompositionQuality improvementImage enhancementDecompositionRate of convergence
The invention discloses and particularly relates to a remote sensing image cloud removing method based on sparse representation. The remote sensing image cloud removing method comprises the following steps of: extracting a cloud mask matrix to acquire a new image; initializing iteration parameters; fixing a texture part of the image and updating a smooth part of the image; fixing the smooth part of the image and updating the texture part of the image; adjusting the smooth part by virtue of a total variation function; updating a iteration threshold; and judging whether decomposition is completed. The remote sensing image cloud removing method based on the sparse representation can adjust the parameters of the total variation adjusting function of the image, namely, a convergence rate of an algorithm is adjusted by changing the parameters of the total variation adjusting function when the block coordinate relaxation algorithm is adopted foe decomposing the image, so that the efficiency of the algorithm and the decomposing effect of the image are improved. The remote sensing image cloud removing method has an obvious thick cloud removing effect on the premise of not destroying original information of the image as much as possible.
Owner:HARBIN ENG UNIV
Air conditioner indoor unit
ActiveCN106382680ALarge air volumeIncrease air speedMechanical apparatusLighting and heating apparatusPhysical healthEngineering
The invention relates to an air conditioner indoor unit. The air conditioner indoor unit comprises ion wind generating devices and purifying devices. The ion wind generating devices are arranged on flowing paths, from a fan assembly to air outlets, in a unit shell, and air subjected to heat exchange through a heat exchange device is promoted by electric field force to flow to the air outlets through an air duct of the fan assembly. Each purifying device is arranged on the flowing path from the corresponding ion wind generating device to the corresponding air outlet and used for removing nitric oxide and ozone in air. The purifying devices are arranged outside the corresponding ion wind generating devices. Since high-voltage discharging devices are arranged inside the ion wind generating devices, a certain amount of harmful gas, mainly O3 and NO2, is generated in the working process of the ion wind generating devices, and in order to prevent the harmful gas from affecting physical health of users after being exhausted from the air conditioner indoor unit, the purifying devices are arranged outside the corresponding ion wind generating devices to remove the harmful gas and improve the air cleanliness.
Owner:QINGDAO HAIER AIR CONDITIONER GENERAL CORP LTD +2
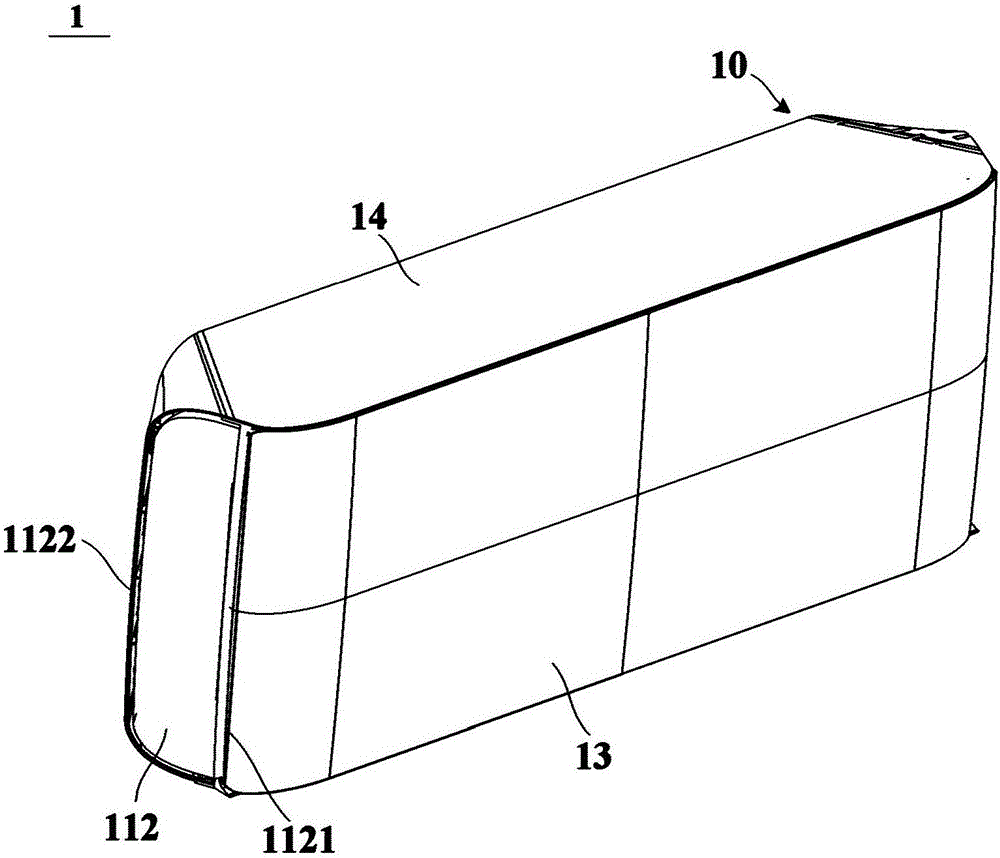
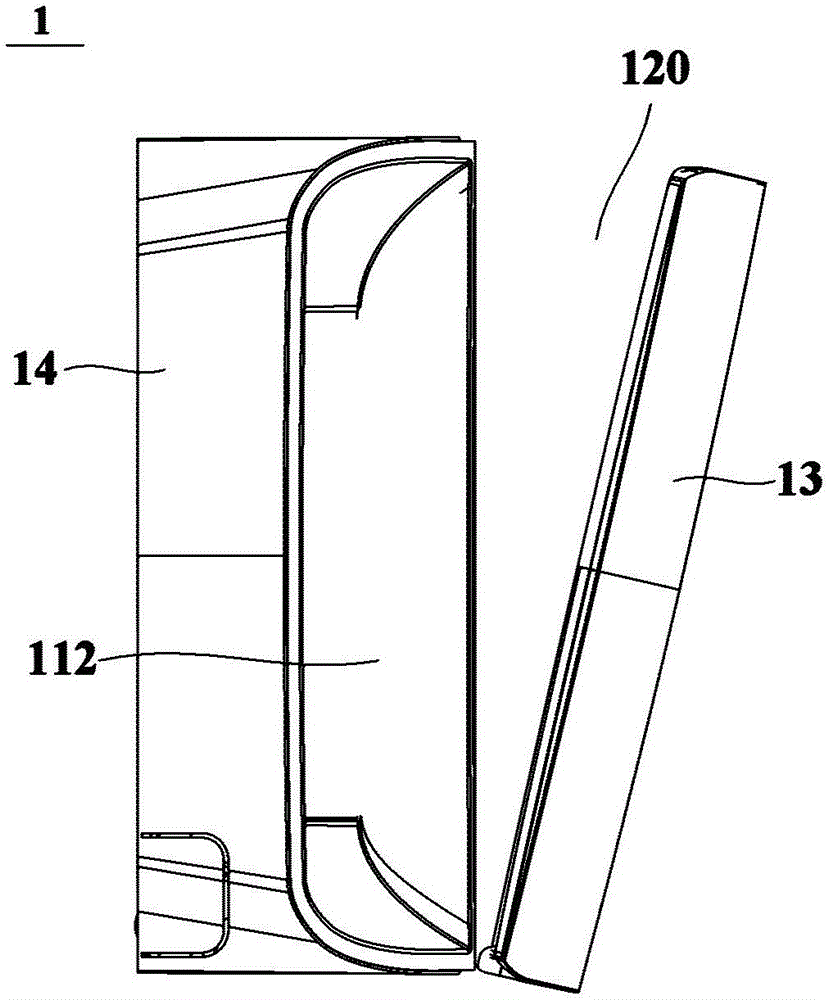
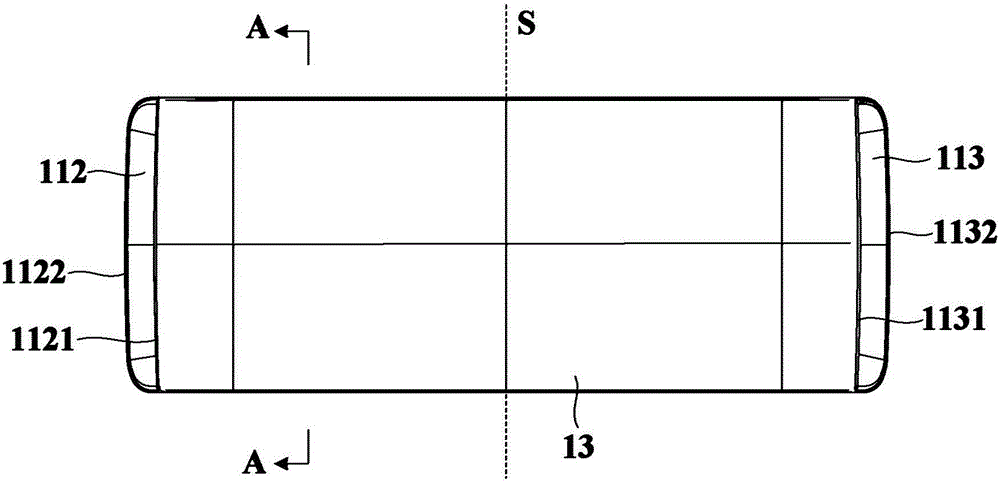
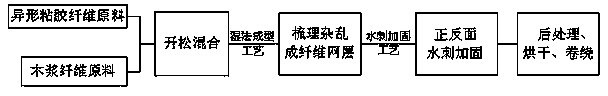
Water-flushing degradable non-woven fabric and production method thereof
InactiveCN103437066AImprove processing efficiencyImprove toughnessNon-woven fabricsHydrogenNonwoven fabric
The invention relates to a water-flushing degradable non-woven fabric and a production method thereof. Profiled viscose fibers and wood pulp fibers are directly used as raw materials and are opened and mixed, then the profiled viscose fibers and the wood pulp fibers are mutually twisted through wet process formation and spun-lacing reinforcement, hydrogen bonds are formed between the profiled viscose fibers and the wood pulp fibers and finally drying is performed to obtain the water-flushing degradable non-woven fabric. By adopting the scheme, the strength of the water-flushing degradable non-woven fabric is high, the water-flushing degradable non-woven fabric can be quickly and fully degraded under the effect of water flushing and the production process is simple.
Owner:HANGZHOU NBOND NONWOVENS
Organic garbage treatment box
InactiveCN102584356ASpeed up decompositionAchieve harmless disposalClimate change adaptationOrganic fertilisersSmall footprintBody positions
The invention discloses an organic garbage treatment box, which comprises a box body, wherein an inner cavity of the box body is partitioned by a partition layer arranged in the middle of the box body through a drawable support; the partition layer and the box body positioned above the partition layer encircle a compost fermentation bin; a material inlet and a gas exhaust port are formed on the upper part of the compost fermentation bin; the partition layer comprises a partition plate which is used for bearing solid garbage for composting and provided with a plurality of liquid penetration holes and a collection tank for collecting waste liquid falling from the liquid penetration holes in the partition plate; an inclined structure which is inclined towards one end of the collection tank to concentratively discharge the waste liquid is arranged at the bottom of the collection tank; and an earthworm breeding bin which is provided with an opening on the top and used for accommodating composts falling from the compost fermentation bin after the partition layer is taken out from the box body and breading earthworms is arranged below the partition layer in the box body. The organic garbage treatment box is easy to operate, occupies a small area, has high garbage treatment efficiency, and is suitable for individual farmer families.
Owner:XINYANG NORMAL UNIVERSITY
High efficiency coal additive
The high efficiency coal additive consists of desulfurizer, leavening agent, combustion adjuvant, smoke suppressor and corrosion inhibitor mixed together. The desulfurizer contains CaO, NH4HCO3, ZnO and SiO2; the leavening agent contains NaHCO3, KAl(SO4)2 .12H2O and acidified coal gangue; the combustion adjuvant contains Fe2O3, MgO, KCl, KMnO4 and NaNO3; the corrosion inhibitor is humic acid and NaNO3; and the smoke suppressor is also KMnO4. The high efficiency coal additive is mixed directly with coal for boiler, furnace, kiln, oven, etc. Detection shows that the high efficiency coal additive can result in desulfurizing rate of 85 %, save coal rate by14.77 %, raise heat efficiency by 9.19 %, lower carbon content of coal slag by 14.86 %,and reduce smoke, scale and corrosion by 90 %.
Owner:郴州市宇洁环保建材有限公司
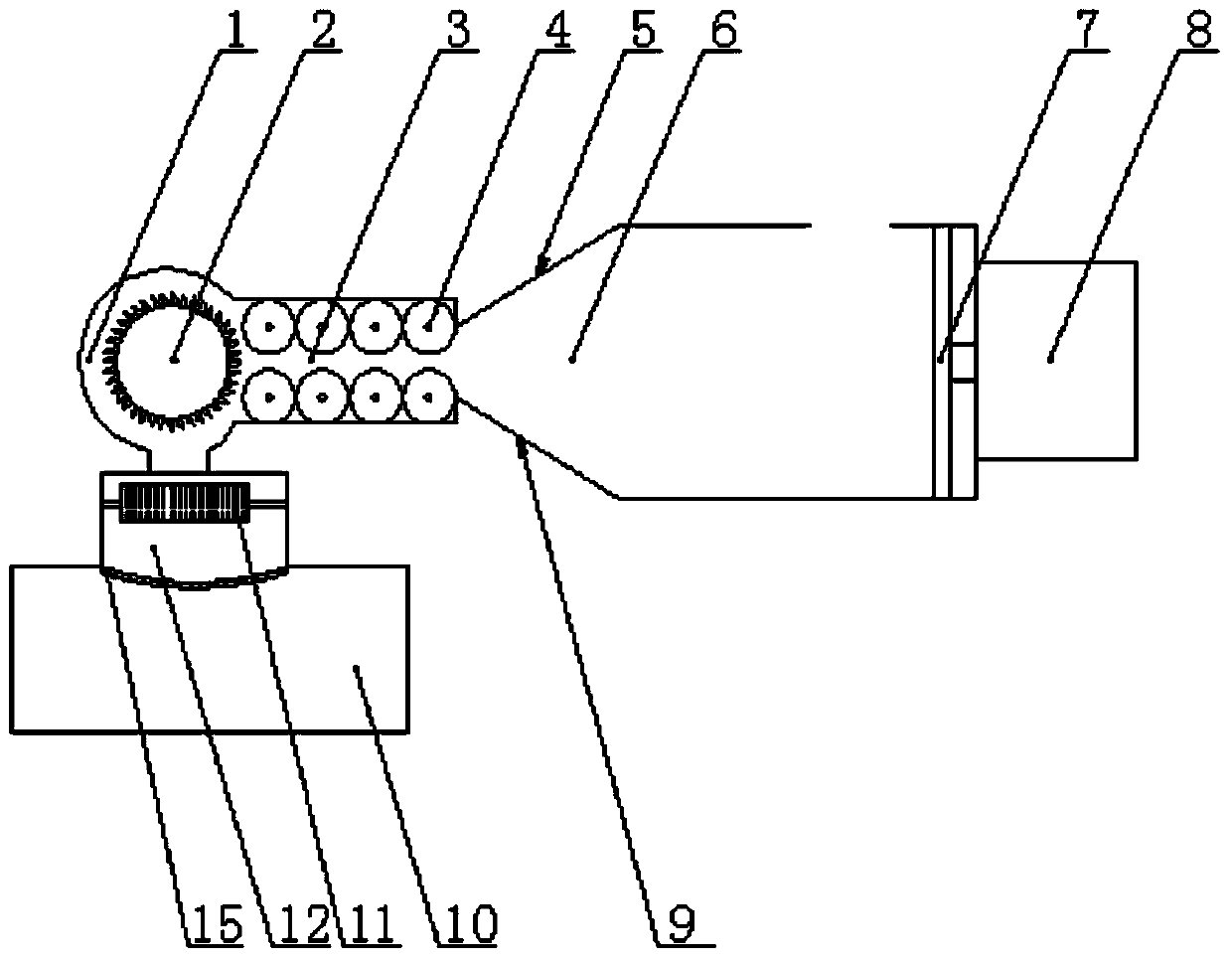
Waste plastic crushing device
InactiveCN103737748AReduce work intensitySpeed up decompositionPlastic recyclingMechanical material recoveryReciprocating motionEngineering
The invention provides a waste plastic crushing device. The waste plastic crushing device comprises a compressing chamber, a heating chamber, a crushing chamber and a discharging chamber which are connected sequentially, wherein a feed port is formed in the top of the compressing chamber; a briquette which can reciprocate towards the opposite side is arranged on one side in the compressing chamber; a plurality of pressing rollers are arranged on the upper and lower parts in the heating chamber respectively; serrated knifes are distributed on each pressing roller; the crushing chamber is composed of a transverse crushing chamber and a vertical crushing chamber; one side of the transverse crushing chamber is communicated with the heating chamber; and the bottom part of the transverse crushing chamber is communicated with the vertical crushing chamber; due to the adoption of the waste plastic crushing device, the problems that the crushing is incomplete, the noise is high, the wearing of a tool bit is serious and splashing materials are likely to cause a safety hidden in a crushing process of a traditional plastic crushing device are solved; the waste plastics are compressed, heated, crushed and degraded, the degrading speed of the waste plastics is significantly improved, on the premise that the complete crushing is guaranteed, the working strength of the tool bit is lowered, the working efficiency is improved, and the crushing cost is reduced.
Owner:TIANJIN HUAQING BAISHENG PLASTIC INDAL
R wave detection algorithm based on extremum field mean mode decomposition and improved Hilbert enveloping
InactiveCN103190901AImprove detection accuracyFast operationDiagnostic recording/measuringSensorsEcg signalElectricity
The invention discloses an R wave detection algorithm based on extremum field mean mode decomposition and improved Hilbert enveloping and belongs to the technical field of weak biological signal processing. An electrocardio signal pre-processing algorithm based on the extremum field mean mode decomposition and the improved Hilbert enveloping and an R wave detection algorithm based on slope threshold are provided. Detection criteria are set according to wave form characteristics and time domain distribution characters of electrocardio signals, and positions of R waves with most obvious characters and highest information amount in the electrocardio signals are detected. An extremum field mean mode decomposition algorithm improves empirical mode decomposition speed and can effectively restrain mode superimposition and boundary effect. The improved Hilbert enveloping can effectively restrain interference of noise and other characteristic waves an can also enhance energy of the R waves. The R wave detection algorithm based on the extremum field mean mode decomposition and the improved Hilbert enveloping can also detect positions of R points accurately even if interference of strong noise and large P / T waves exists. A Massachusetts institute of technology-Beth Israel hospital (MIT-BIH) data base is used for detecting the R wave detection algorithm. Sensitivity of the R wave detection algorithm is 99.94%, and positive predictive rate is 99.87%.
Owner:TIANJIN POLYTECHNIC UNIV
Magnetic resonance image denoising method based on improved multi-path matching pursuit algorithm
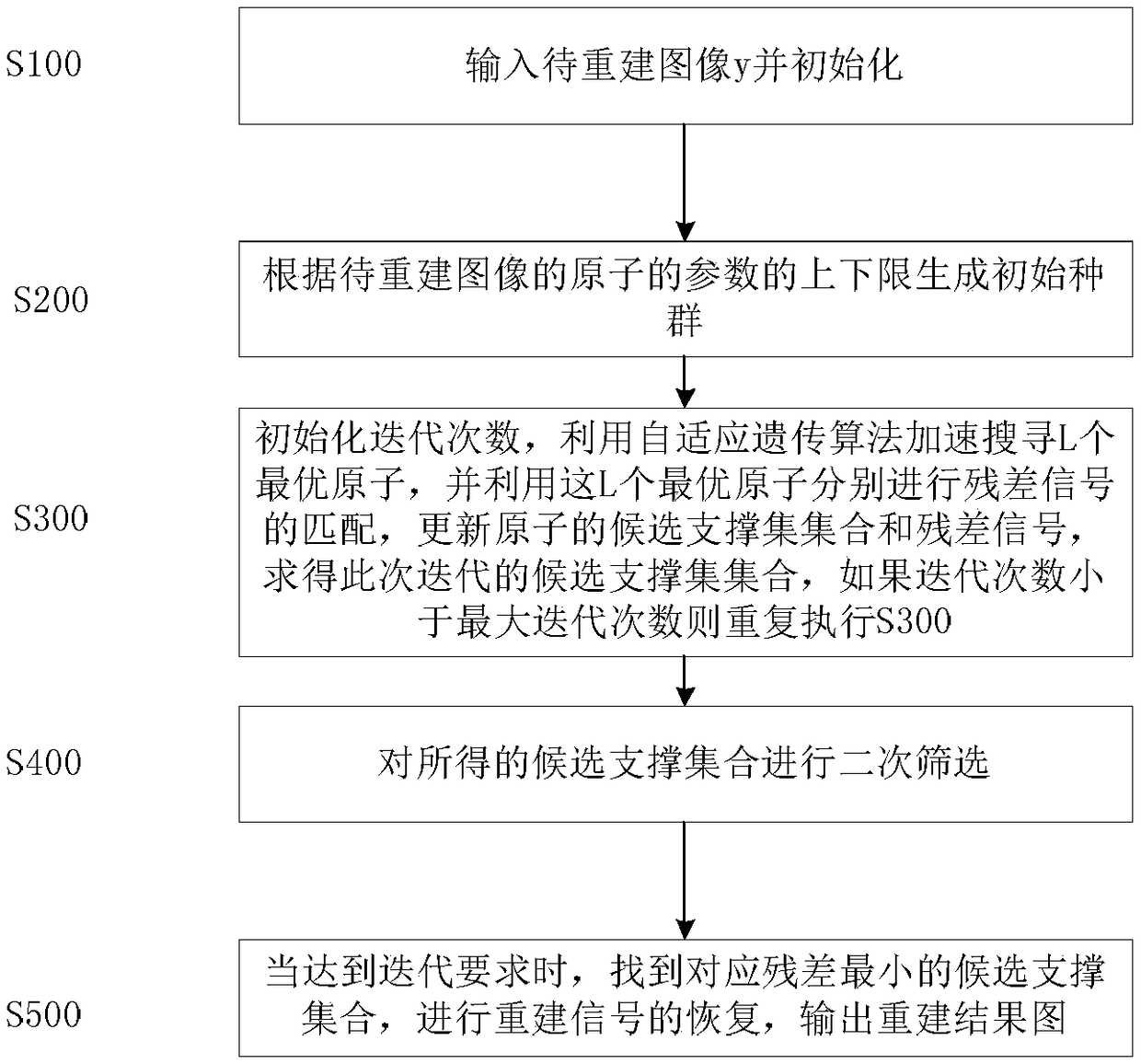
ActiveCN109191399AThe principle is simpleImprove efficiencyImage enhancementImage analysisPattern recognitionMatching pursuit algorithms
A magnetic resonance image denoising method relates to image processing. In this method, the adaptive genetic algorithm (AGA) is introduced into the iterative search of multiple candidate atoms matched with the local image features in multipath matching pursuit algorithm. The method combines the advantages of adaptive genetic algorithm and multi-path matching pursuit algorithm, thereby avoiding the shortcomings that genetic algorithm is easy to fall into the local optimum and getting the best matching parameters with high precision, reducing the computational load of multi-path matching pursuit algorithm and overcoming the shortcomings that the conventional method cannot be popularized and applied because of too much computational load.
Owner:SHAANXI NORMAL UNIV
Low heat value gas oxygen-enriched burning process in rotary lime kiln
The oxygen-enriched low heat value gas burning process in rotary lime kiln includes adopting low heat value gas of (1700-2000)x4.18 kJ / cu m, such as converter gas and coke oven gas, burning in oxygen-enriched air of oxygen content 23-25 % to reach over 100 deg.c higher calcining temperature, compressing oxygen in a compressor and blending oxygen into the comburant air for burning, and increasing the oxygen content to reinforce burning, concentrate high temperature area and speed the decomposition of limestone.
Owner:ACRE COKING & REFRACTORY ENG CONSULTING CORP DALIAN MCC
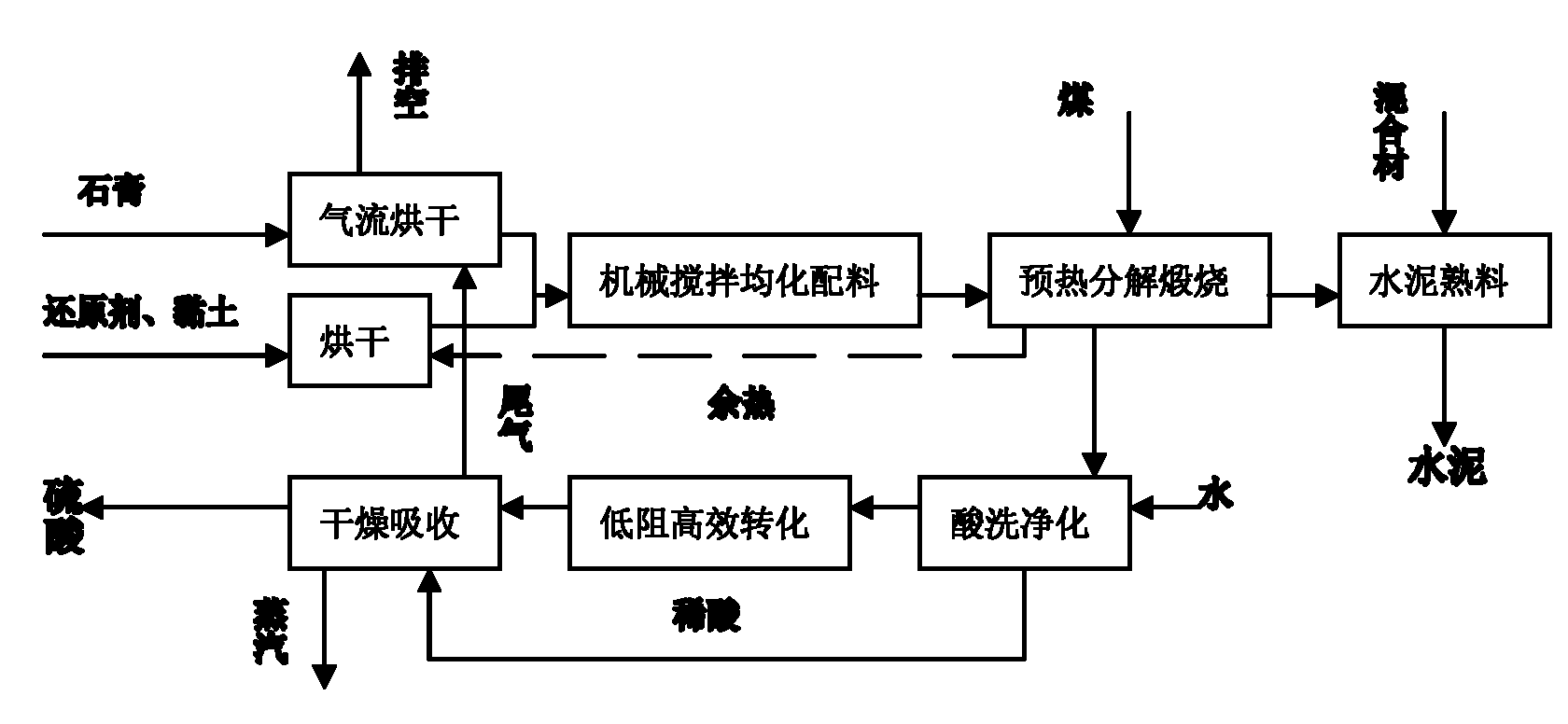
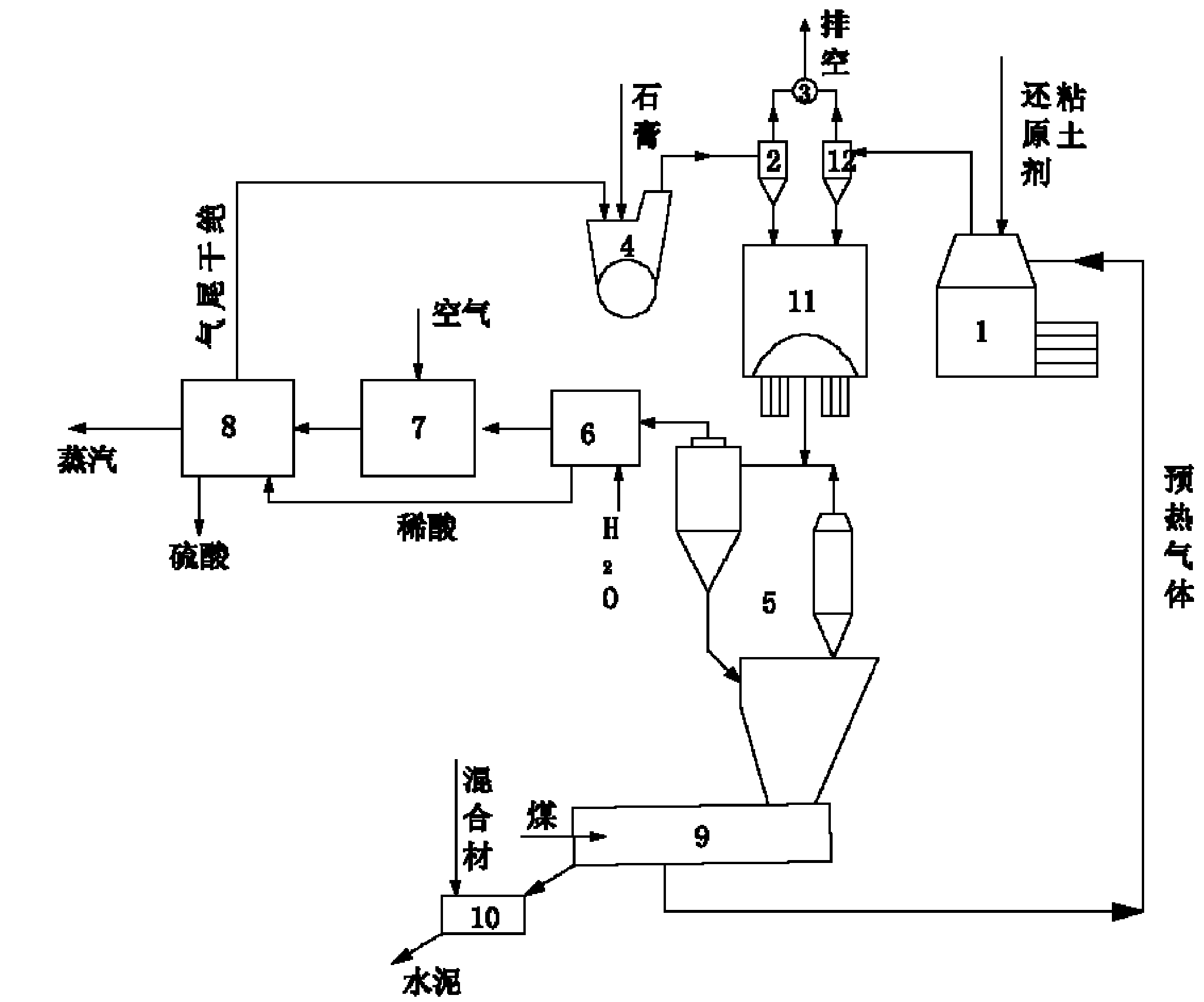
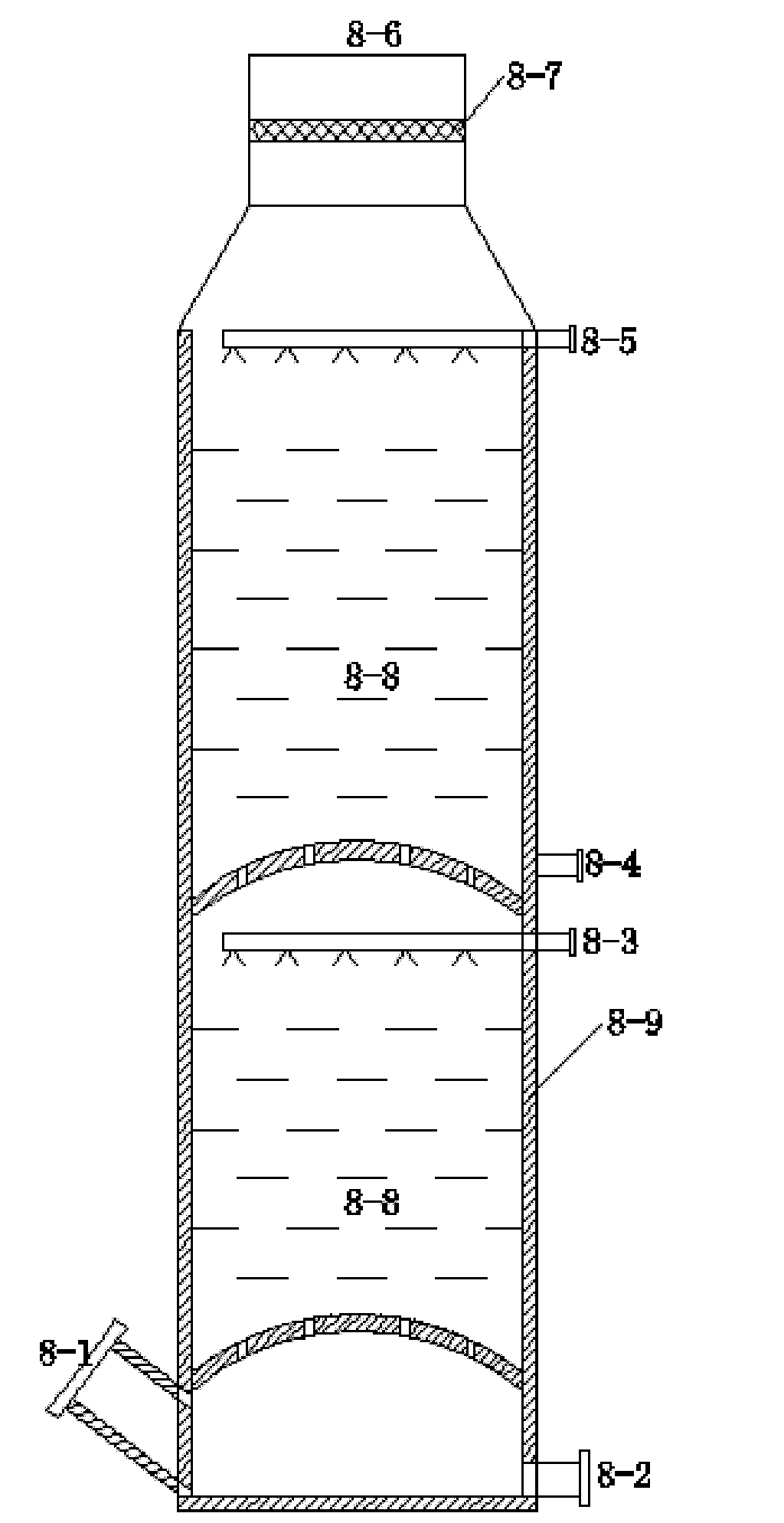
Improved production process for preparing sulfuric acid and cement from gypsum
The invention relates to an improved production process for preparing sulfuric acid and cement from gypsum. Raw materials comprise dihydrate gypsum, clay and a reducing agent. The process comprises the following steps of: drying the raw materials; independently grinding; mechanically stirring and homogenizing; preheating green stock; decomposing and calcining through a rotary kiln; acid-washing and purifying kiln gas; drying through SO2; performing a double-conversion double-absorption process; and grinding the cement to prepare sulfuric acid and cement products, wherein a dihydrate gypsum process is adopted; the raw materials are dried by absolute dry tail gas at the temperature of between 70 and 85 DEG C which is discharged out of a sulfuric acid absorption device to obtain the dihydrate gypsum with free water content of less than 5 weight percent; and the dihydrate gypsum is then directly homogenized with the clay and the reducing agent in the presence of CaCl2 or Na2SO4 to produce the green stock. In the process, the heat consumption is reduced by 30 percent, the power consumption is reduced by 20 percent, the investment is reduced by 25 to 40 percent, and the cost is reduced by 25 to 30 percent. The process overcomes the disadvantages of high power consumption and heat consumption, large investment, low operation ratio and the like in the conventional method, 0.4 to 0.5 tonnage of steam serving as a byproduct is produced by every tonnage of sulfuric acid, and the process has obvious economic benefit.
Owner:南京创能电力科技开发有限公司
Microorganism mixed culture for degrading kitchen wastes, method for producing microorganism mixed culture for degrading kitchen wastes and method for degrading wastes through utilizing culture
InactiveCN102911869AImprove decomposition efficiencySpeed up decompositionBacteriaClimate change adaptationBacillus licheniformisLichen
The invention discloses a microorganism mixed culture for degrading kitchen wastes, a method for producing the microorganism mixed culture for degrading kitchen wastes and a method for degrading wastes through utilizing the culture. The mixed culture comprises at least two or a plurality of bacillus smithii, bacillus stearothermophilus spore and bacillus licheniformis; and the bacillus smithii accounts for 80-90% of the quantity of active viable bacteria in the mixed culture, the bacillus stearothermophilus spore accounts for 0-10% of the quantity of the active viable bacteria in the mixed culture, and the bacillus licheniformis accounts for 0-10% of the quantity of the active viable bacteria in the mixed culture. The method for degrading wastes through utilizing the culture comprises: wood chips which carry the culture are added into a container which is used for heating and is provided with a stirring device, so that the water content of the wood chips can reach 50-70%; the wood chips with the water content of 50-70% are put in the kitchen wastes to be stirred uniformly and heated to 40-80 DEG C, so that the kitchen wastes are degraded; and discharge is carried out after 24 hours. The mixed culture provided by the invention can effectively turn wastes which are mainly kitchen wastes into organic fertilizers.
Owner:王欢
Method for plating copper and tin on metal circuit board through pattern plating
InactiveCN103052270AFlat surfaceOvercoming technical bias against tin platingConductive pattern reinforcementTinningCopper plating
The invention discloses a method for plating copper and tin on a metal circuit board through pattern plating. The method comprises steps of workpiece pre-treating, micro-etching, primary cleaning, copper plating, secondary cleaning, tin plating and third cleaning. According to the method, after copper is plated, workpieces are cleaned, and tin plating operation is performed immediately; a tin welding plane is smoother through tin plating operation, so that the uniformity of a product is improved, and batch treatment can be performed; a tin plating process is adopted, so that the conventional process concept is changed, the technical bias that a PCB (Printed Circuit Board) cannot be plated with tin is eliminated, the performance of the product is improved, and the production efficiency of the product is increased; a degreasing agent of CS-4-A or CS-4-B series is taken as a mailing component in a degreasing process, so that the decomposing speed of grease can be increased, and the efficiency is further increased; and a bronzing agent is added into a copper plating solution, so that the copper surface of the product is smooth and complete and has high flatness, and the quality of the product is improved.
Owner:SICHUAN HAIYING ELECTRONICS TECH
Key-element-based matrix decomposition and fine tuning method
InactiveCN105976070AImprove decomposition efficiencySpeed up decompositionAdvertisementsForecastingQR decompositionAlgorithm
The invention provides a key-element-based matrix decomposition and fine tuning method. The method comprises: scoring information of a commodity by a user is collected and a sparse scoring matrix R is formed based on the scoring information; according to the sparse scoring matrix R, two low-dimensional matrixes P and Q are initialized, multiplying of the low-dimensional matrixes P and Q is carried out to obtain a predicted matrix R^, and an objective function based on minimization of a difference value between the sparse scoring matrix R and the predicted matrix R^; and the objective function is optimized by an iterative process to obtain the low-dimensional matrixes P and Q and the predicted matrix R^, and fine tuning and updating are carried out on the predicted matrix R^, thereby obtaining a final predicted matrix. According to the invention, in order to overcome defects in the existing matrix decomposition algorithm, a quick matrix decomposition and fine tuning algorithm is put forward based on improvement of the algorithm. With the method, the decomposition efficiency and the decomposition speed of the matrix are increased and the recommendation accuracy is enhanced.
Owner:BEIJING JIAOTONG UNIV