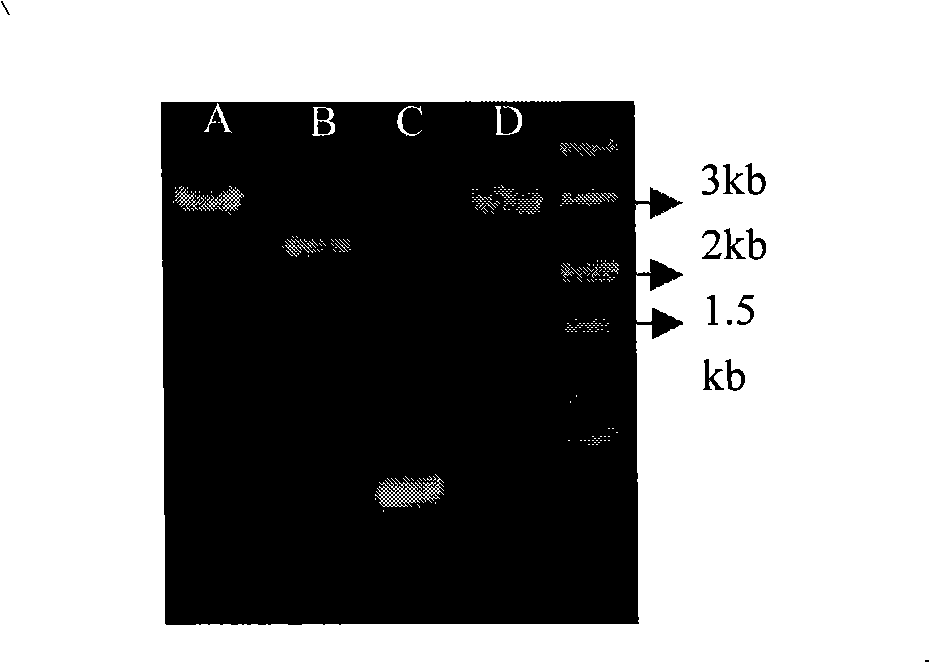
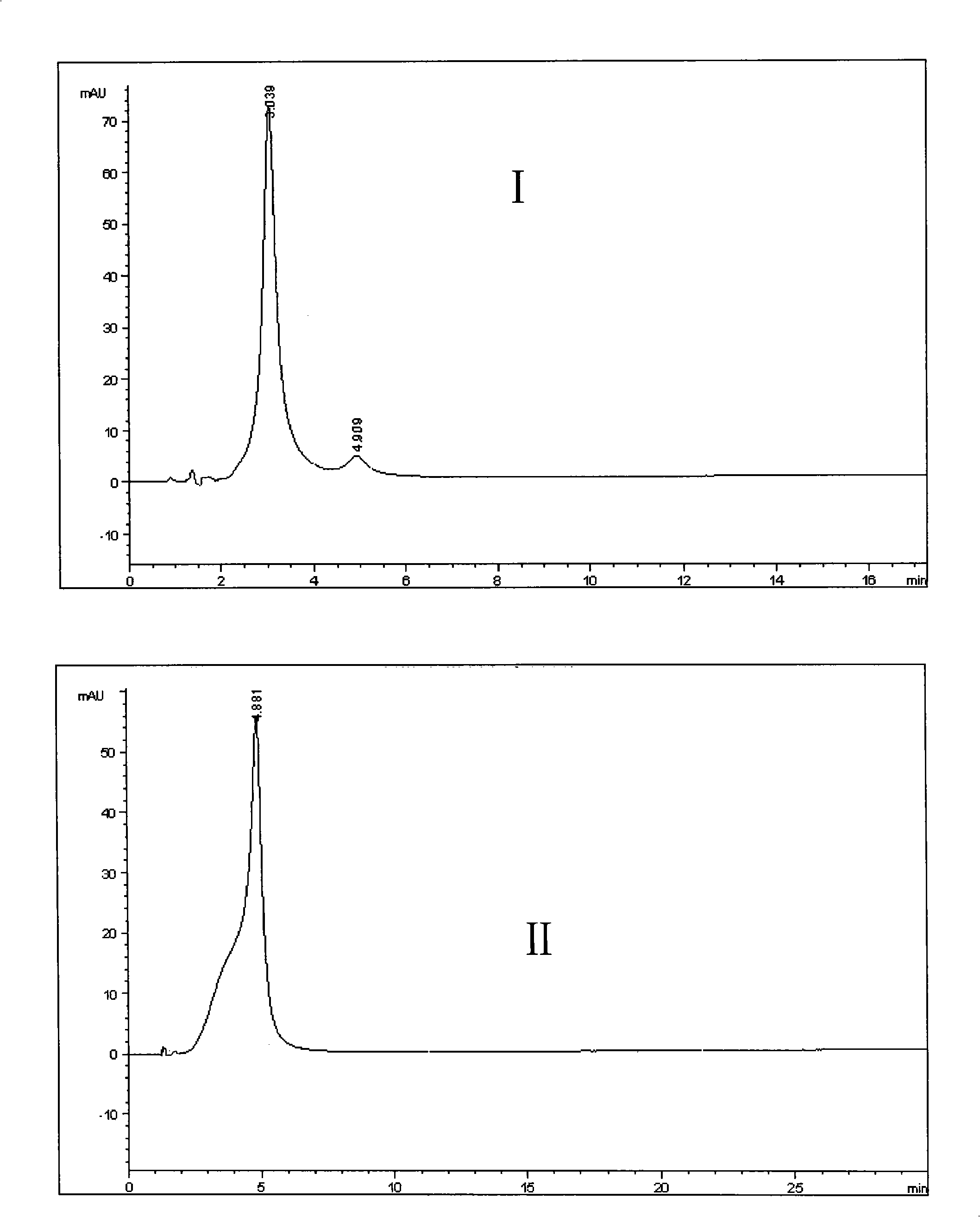
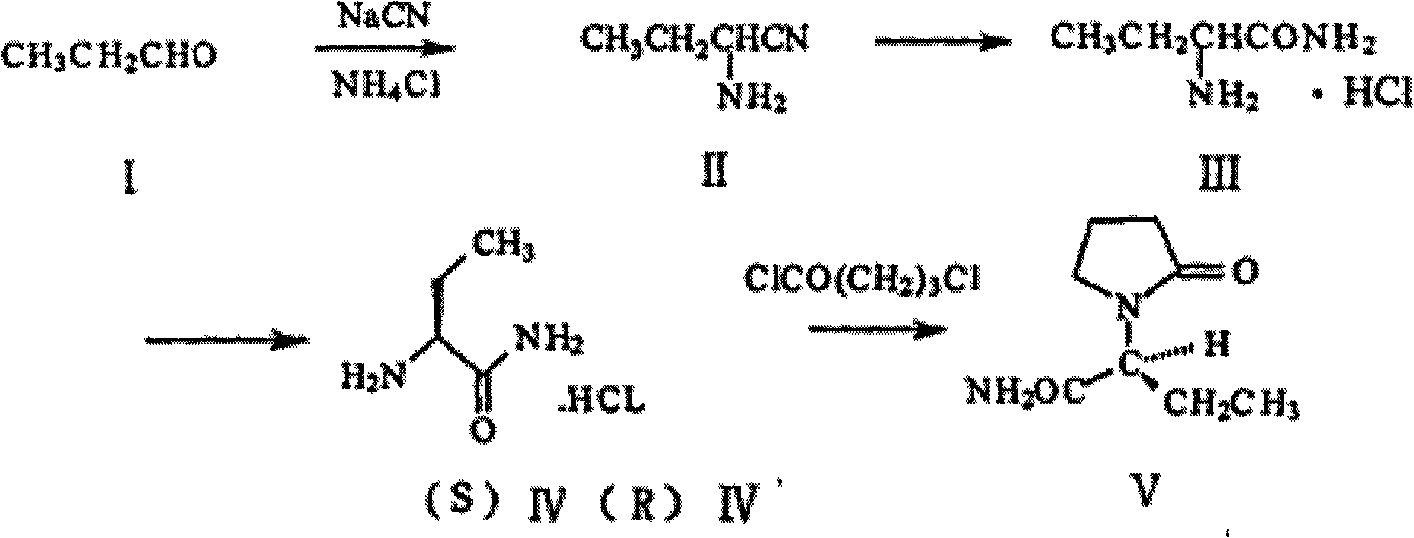
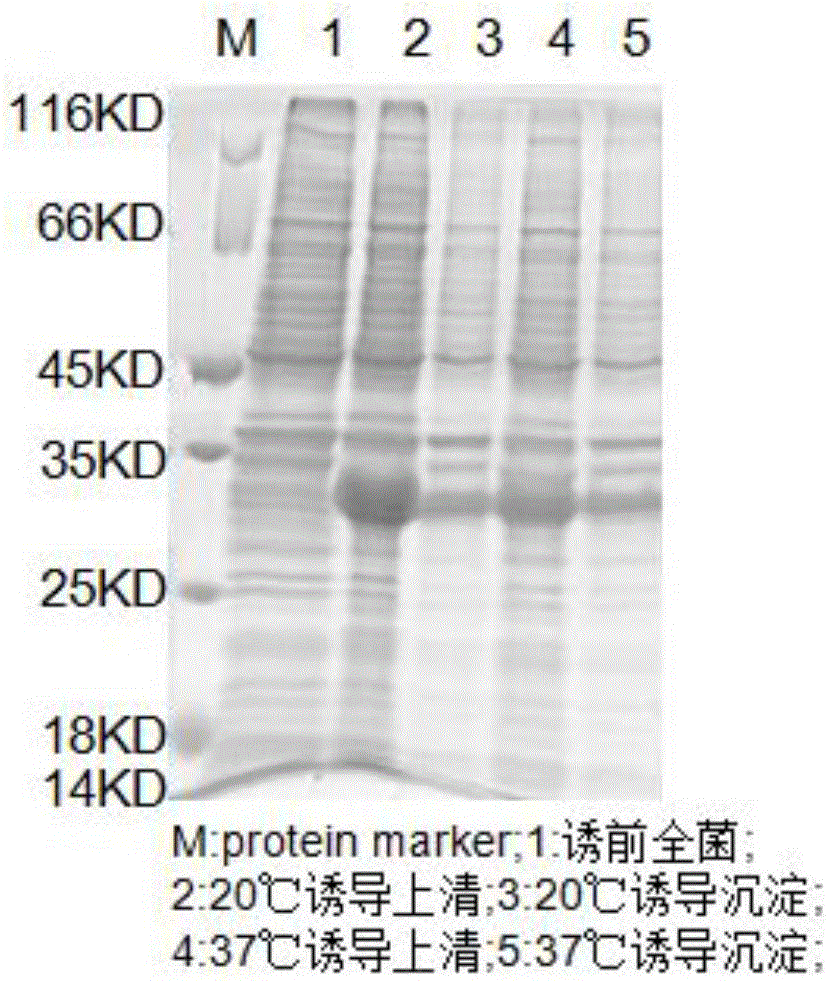
Patents
Literature
46results about How to "Simple separation and purification" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
Immunomagnetic bead purification kit and method for 12 kinds of swine common viruses and germs
InactiveCN105779398AEfficient separationSimple separation and purificationBacteriaMicrobiological testing/measurementBiotinStreptomycin
The invention provides an immunomagnetic bead purification kit and method for 12 kinds of swine common viruses and germs. The kit comprises a reagent A, a reagent B and a reagent C, wherein the reagent A is streptomycin immunomagnetic suspension; the reagent B is a 12-biotinylation monoclonal antibody mixed solution; and the reagent C is a 10* immunomagnetic bead separation and purification system buffer solution. The method comprises the following steps of (1) performing a pathogen suspension preparation process; (2) performing a biotin antibody preparation process; and (3) performing a pathogen immunological purification process. The immunomagnetic bead purification kit and method for 12 kinds of swine common viruses and germs provided by the invention have the advantages that 12 kinds of viruses and germs such as PPV (Porcine Parvovirus), SS-II (Streptococcus Suis Type 2), PRV (Porcine Pseudorabies Virus) and the like in a sample can be simultaneously, fast and effectively purified. The detection method has the advantages of high accuracy, high specificity, high sensitivity and high stability, and the subsequent fast diagnosis and effective detection work is facilitated.
Owner:BEIJING YISEN BIOTECH
Method for catalytic degradation of hexachlorobenzene
ActiveCN103977744AHigh catalytic activityLarge amount of processingHydrocarbon from halogen organic compoundsAmmonium halidesSolventChemistry
The invention discloses a method for catalytic degradation of hexachlorobenzene. The method comprises the following steps: firstly, filling a catalyst into a fixed bed reactor, and carrying out reduction treatment on the catalyst; secondly, mixing hexachlorobenzene steam and preheated hydrogen uniformly to obtain a mixed gas, introducing the mixed gas into the fixed bed reactor after reduction treatment, and carrying out catalytic hydrodechlorination reaction on the catalyst, thereby obtaining the mixed gas of benzene steam, hydrogen chloride gas and unreacted hydrogen; and thirdly, feeding the mixed gas of benzene steam, hydrogen chloride gas and unreacted hydrogen into a condenser for condensing, converting benzene into liquid, then feeding the hydrogen chloride gas and unreacted hydrogen in the mixed gas into an absorption tower for absorbing hydrogen chloride by ammonia water, compressing the unreacted hydrogen by a compressor, and returning for recycling. A solvent is not required to be added in catalytic hydrogenation and degradation of hexachlorobenzene by the method provided by the invention, benzene and ammonium chloride are produced as byproducts, and zero emission of pollutants can be reached, and the method is an environmental-friendly technology for degrading hexachlorobenzene efficiently.
Owner:XIAN CATALYST NEW MATERIALS CO LTD +1
Method for preparing deoxidized violacein and special recombinant bacterium
InactiveCN101319219AEasy to pick upSimple separation and purificationBacteriaMicroorganism based processesTryptophanGene cluster
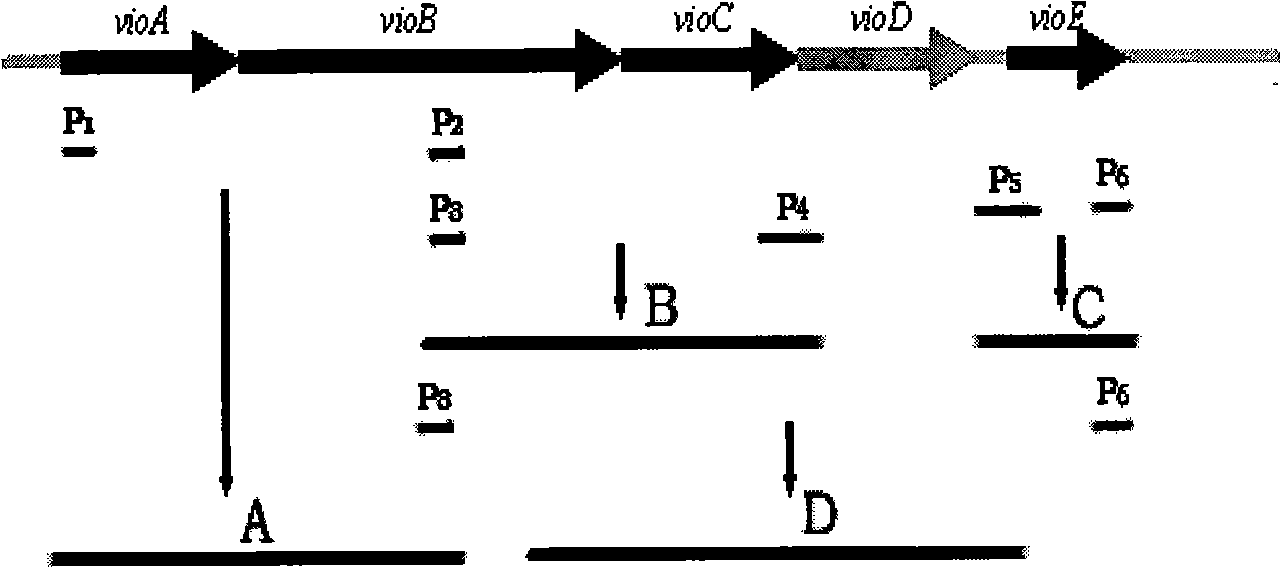
The invention discloses a method for producing deoxidized violacein and special recombination strains thereof. The method comprises the following steps of: obtaining the recombination strains through inducting a deoxidized violacein synthesis associated gene cluster into bacillus coli BL21-CodonPlus(DE3)-RIL, producing the deoxidized violacein through the recombination strains which take tryptophan as a substrate to ferment, wherein the deoxidized violacein synthesis associated gene cluster is a recombination gene cluster which is obtained through getting rid of a VioD gene from a violacein synthetic gene cluster consisting of VioA, VioB, VioC, VioD and VioE, and the recombination strains are obtained through inducting the gene cluster into bacillus coli BL21-CodonPlus(DE3)-RIL and are capable of producing the deoxidized violacein. The method for producing the deoxidized violacein has a yield as high as 0.71g / L fermentation liquor with convenient extraction and simple separation and purification.
Owner:TSINGHUA UNIV
A kind of process improvement method of preparing 2-aminobutyronitrile
InactiveCN102295580AIncrease concentrationTake advantage ofCarboxylic acid nitrile preparationOrganic compound preparationPolymer scienceCyanide
This article relates to a process improvement method for the preparation of levetiracetam intermediate 2-aminobutyridine from antiepileptic drugs. The method of the invention improves the traditional Strecker reaction, and remarkably improves the reaction efficiency through the airtight ammonia pressure condition and the use of excess cyanide.
Owner:ZHEJIANG HUAHAI PHARMACEUTICAL CO LTD +1
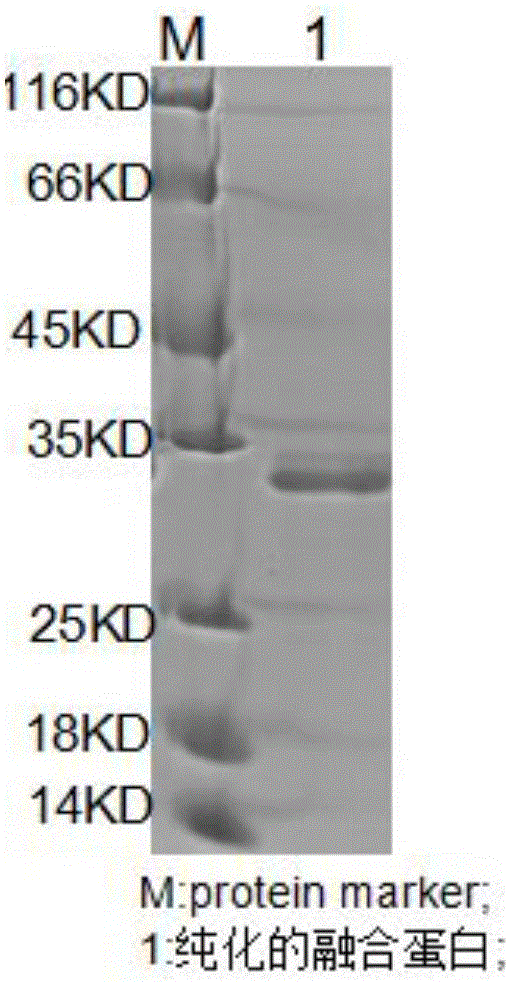
Fusion protein expression purification method
InactiveCN106636163AHigh expressionSimple separation and purificationPeptide preparation methodsFermentationClelands ReagentAgar
The invention discloses a fusion protein expression purification method, which comprises the following experimental articles: 1ul of recombinant pET32a plasmids, BL21 (DE3) bacterial strains, 100ug / ml of penbritin, 0.5mM of IPTG (isopropyl-beta-d-thiogalactoside), PBS (Phosphate Buffer solution), 220rpm, 2M urea,50mM of Tris, 300mM of NaCl, 1mM of DTT (dithiothreitol), lysozyme, 0.2% of TritonX-100, 5mL of NI-NTA, EK (Enterokinase) enzyme, nickel agarose affinity chromatography and a SK3071 non-interference albumen concentration measurement kit. The fusion protein expression purification method comprises the following experiment process: the prokaryotic expression of fusion protein, the purification of the fusion protein and the analysis of the purified fusion protein. According to the fusion protein expression purification method, (50mM of imidazole) eluant which contains target protein is dialyzed into 1M urea, the 1M urea is dialyzed at a temperature of 4DEG C overnight, the EK enzyme is added into a sample subjected to dialysis overnight, protein subjected to enzyme digestion is subjected to the nickel agarose affinity chromatography to remove a label and the EK enzyme, and the purity of the purified fusion protein is detected and analyzed through SDS-PAGE (Sodium Dodecyl Sulfate Polyacrylamide Gel Electrophoresis) electrophoresis and gray analysis. Therefore, compared with a non-fusion expression carrier, the purified fusion protein has the advantages of high expression quantity, simple foreign protein separation and purification, high recovery rate, simple purification and purification technology and low cost.
Owner:上海柏根生物科技有限公司
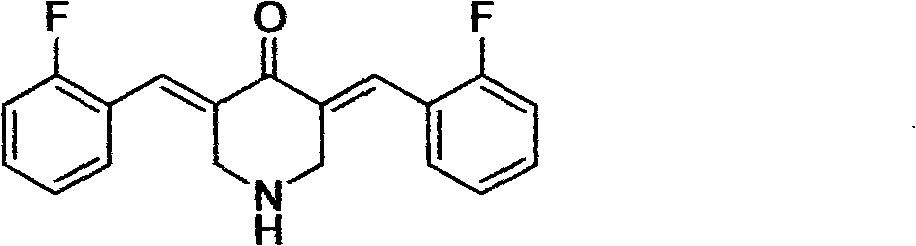
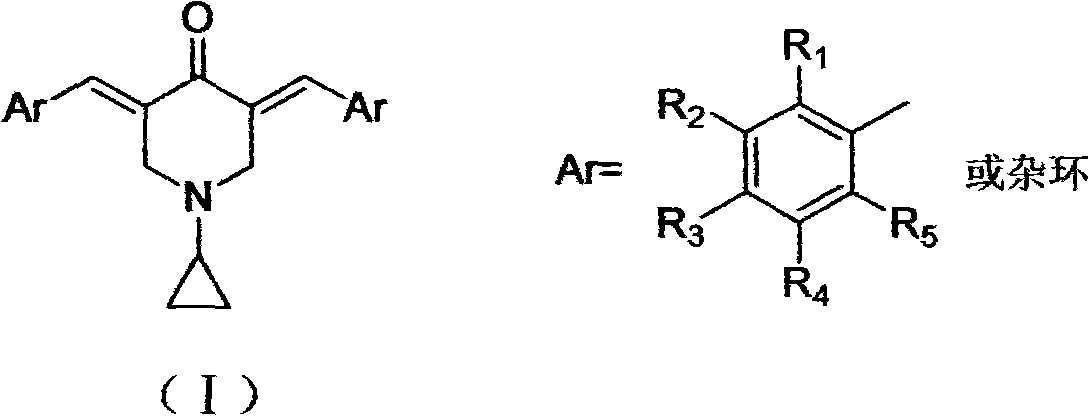
3,5-(E)-dibenzylidene-N-cyclopropyl piperidine-4-ketone and application of the 3,5-(E)-dibenzylidene-N-cyclopropyl piperidine-4-ketone in preparing antitumour drugs
ActiveCN102153508AShort reaction timeSimple separation and purificationOrganic active ingredientsOrganic chemistryDrugKetone
The invention discloses 3,5-(E)-dibenzylidene-N-cyclopropyl piperidine-4-ketone and application of the 3,5-(E)-dibenzylidene-N-cyclopropyl piperidine-4-ketone in preparing antitumor drugs. The 3,5-(E)-dibenzylidene-N-cyclopropyl piperidine-4-ketone compound can be obtained by Claisen-Schimidt reaction of aromatic aldehyde and N-cyclopropyl-4-piperidine ketone. The 3,5-(E)-dibenzylidene-N-cyclopropyl piperidine-4-ketone compound can obviously restrain the proliferation of various tumor cells and can be used for preparing drugs for treating leukocythemia, colorectal cancer, lover cancer, skin cancer, gastric cancer, breast cancer, prostatic cancer or other malignant tumor.
Owner:FUJIAN MEDICAL UNIV
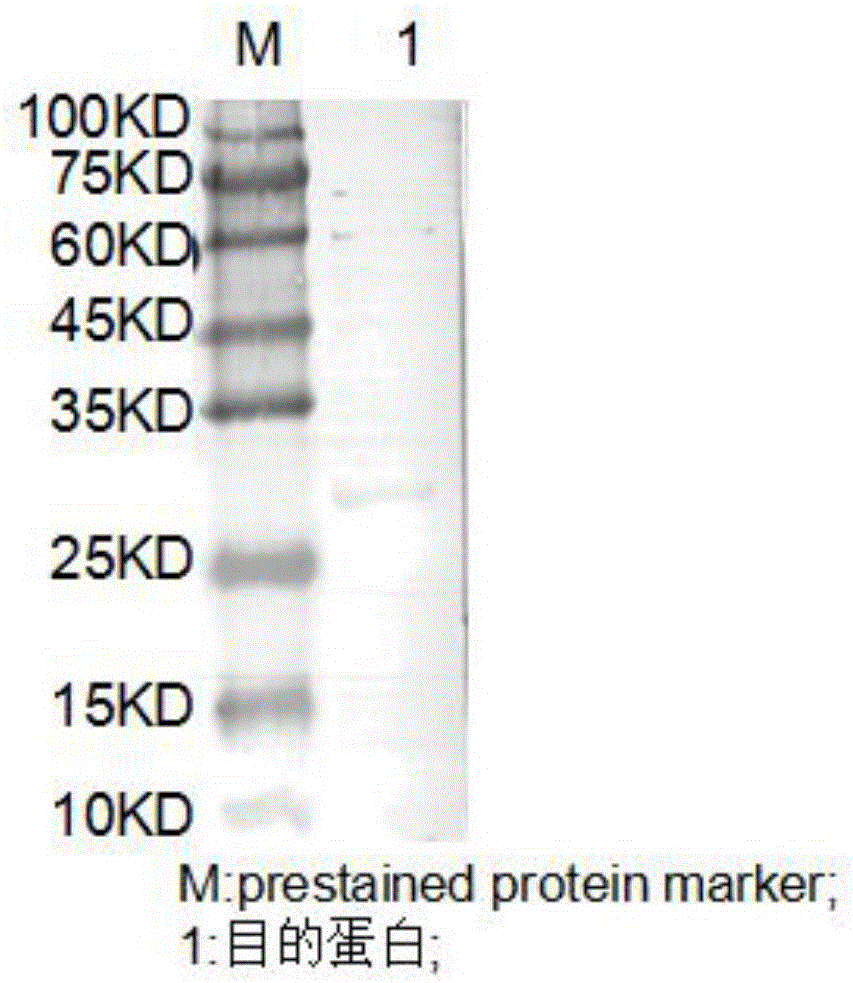
Fusion protein of human serum albumin (HAS) and human mutant hepatocyte growth factor (HGF), and preparation method and application thereof
InactiveCN108424459AHigh expressionSimple separation and purificationPeptide/protein ingredientsHepatocyte-growth/scatter/tumor-cytotoxic factorMutantHalf-life
The invention discloses a fusion protein of a HAS and human mutant HGF, and a preparation method and application thereof. The fusion protein comprises a first region composed of the HSA and a second region composed of the human mutant HGF, wherein the human mutant HGF refers to a mutation of amino acid R at site 494 of a HGF into N, D, K, Q or G. According to the invention, the HSA-HGF fusion protein is prepared from a HGF mutant which is biologically active without the action of proteolytic enzymes, and the fusion protein improves the expression level of the HGF in a CHO system and is simplified in separation and purification while maintaining the biological activity of the HGF. The fusion protein of the invention prolongs the in-vivo half-life of the HGF and increases the expression level of the HGF while maintaining the biological activity of the HGF, and has good application prospects in the pharmaceutical field.
Owner:SHANGHAI JIAOTONG UNIV SCHOOL OF MEDICINE +1
Preparation method of wood fiber oil-absorption material
InactiveCN106750368ASimple separation and purificationEasy to operateOther chemical processesFiberOrganic solvent
The invention provides a preparation method of a wood fiber oil-absorption material. An esterification reaction is carried out on wood fiber and an esterification reagent under the conditions of a pyridine catalysis effect and ball-milling, so that the wood fiber oil-absorption material is obtained, an organic solvent does not need to be used in the reaction process, the product separation and purification treatment is simple, and the preparation technology is convenient in operation, short in modification time, high in reaction degree and better in oil absorption effect; the absorption rate for diesel oil is 9.86 g / g, and the absorption rate for soybean oil is 10.21 g / g.
Owner:SOUTH CHINA UNIV OF TECH
Enzyme catalysis method for continuous synthesis of lauroyl maltose under organic phase
InactiveCN1995366AIncrease productionSimple separation and purificationFermentationEnzyme reactorChemistry
The invention discloses a continuous synthesizing method of maltose laurate in the organic phase, which is characterized by the following: using stirring tank typed enzyme reactor to synthesize maltose laurate; affirming optimum manufacturing condition; maintaining the reactor production at 10g / (L .d) for 8-10d; obtaining the purity of product to 95%.
Owner:JIANGNAN UNIV
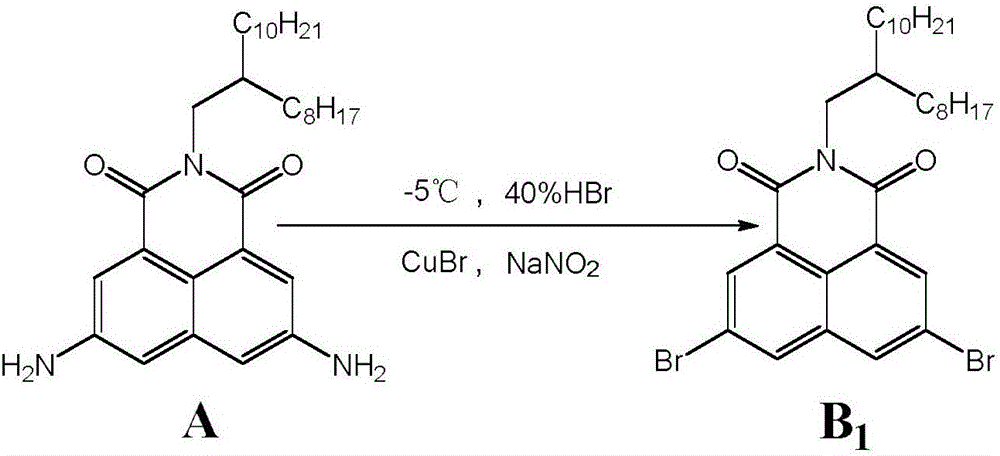
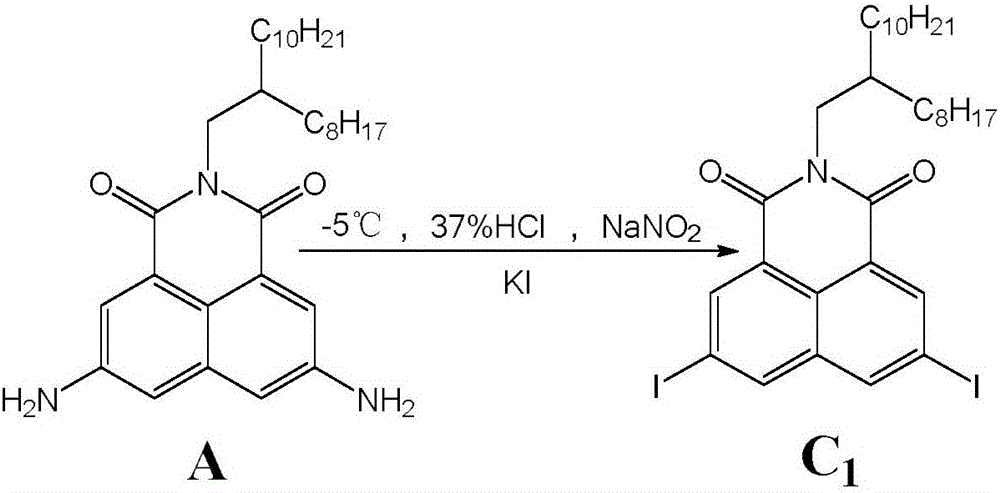
Synthesis method of 3,6-halogen atom-substituted 1,8-naphthalimide
ActiveCN106749017ASimple separation and purificationMild reaction conditionsOrganic chemistrySolventHalogen
The invention discloses a synthesis method of 3,6-halogen atom-substituted 1,8-naphthalimide. The synthesis method comprises the following steps of introducing halogen atoms to 3 and 6 bits of 1,8-naphthalimide; taking 3,6-diamido-1,8-naphthalimide, sodium nitrite and cuprous halide as raw materials, wherein the molar ratio of the 3,6-diamido-1,8-naphthalimide to the sodium nitrite is 1:(1-10), and the molar ratio of the 3,6-diamido-1,8-naphthalimide to the cuprous halide is (1:1) to (1:10); and reacting for 1-48h at a low temperature of -10 DEG C to 10 DEG C under a mixed solvent to obtain the 3,6-halogen atom-substituted 1,8-naphthalimide. The 3,6-halogen atom-substituted 1,8-naphthalimide is greatly prepared through mild conditions. The method has the advantages of being simple and convenient to separate and purify and mild in reaction conditions.
Owner:NANCHANG UNIV
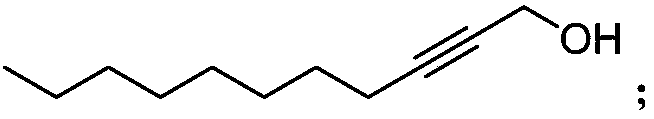
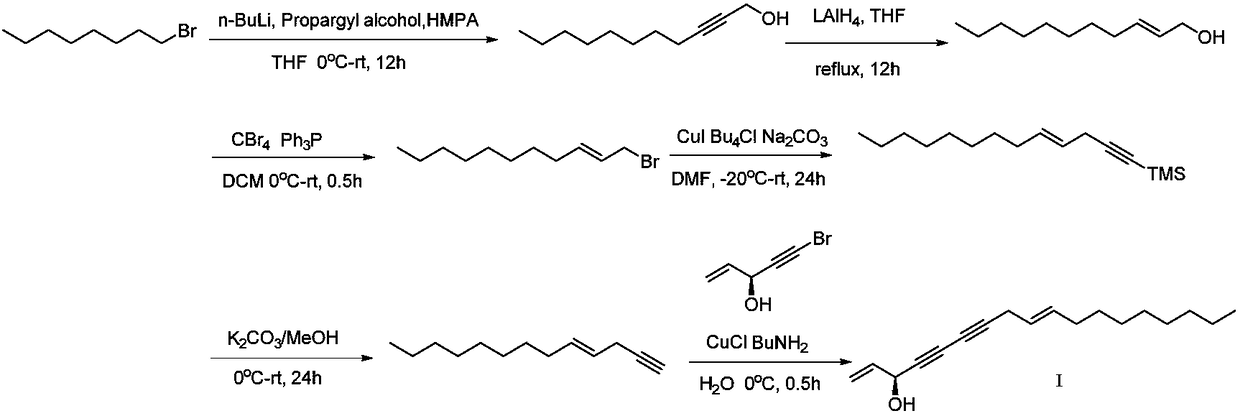
Synthetic method of highly-activity chiral acetylenic alcohol (S,E)-1,9-diene-4,6-diacetyl-3-octadecyl alcohol
InactiveCN108440242AFeeding ratio is reasonableHigh purityGroup 4/14 element organic compoundsOrganic compound preparationCouplingAcetylenic alcohol
The invention discloses a synthetic method of highly-activity chiral acetylenic alcohol (S,E)-1,9-diene-4,6-diacetyl-3-octadecyl alcohol. The synthetic method disclosed by the invention comprises thefollowing steps: bromooctane is taken as a starting raw material and is reacted in a reasonable proportion to synthesize required alkynol; and the efficient synthesis of active molecule (S,E)-1,9-diene-4,6-diyne-3-octadecanol is realized through reduction, bromination, coupling reactions, desilication and docking with a chiral molecule. According to the method, the yield in the reaction process ishigh, a synthetic route is simple, the raw material is simple and easy to obtain, the synthetic cost is low, reaction conditions are mild, the separation and purification operations are simple, and ahigh-purity target compound can be directly obtained; a chiral center is introduced in the last step, so that the possibility of racemization of the substance in the reaction is avoided; and in addition, the synthesized active molecule (S,E)-1,9-diene-4,6-diyne-3-octadecanol has a wide application prospect.
Owner:HENAN AGRICULTURAL UNIVERSITY
Pseudomonas aeruginosa bacteriophage lysin, coding gene thereof, recombinant expression vector as well as preparation method and application thereof
InactiveCN108103050AStrong lytic activitySimple separation and purificationAntibacterial agentsFungiRecombinant expressionAntibacterial agent
The invention belongs to the field of bioengineering technology, and discloses pseudomonas aeruginosa bacteriophage lysin, a coding gene thereof, a recombinant expression vector as well as a preparation method and an application thereof. An amino acid sequence of the pseudomonas aeruginosa bacteriophage lysin is shown as SEQ ID No.1, and a nucleotide sequence of the pseudomonas aeruginosa bacteriophage lysin is shown as SEQ ID No.2; the recombinant expression vector is constituted by recombining the nucleotide sequence of the pseudomonas aeruginosa bacteriophage lysin and a vector; and the recombinant expression vector is transformed into a host bacterium, so that a recombinant engineering bacterium is constituted. The pseudomonas aeruginosa bacteriophage lysin provided by the invention has the advantages that the pseudomonas aeruginosa bacteriophage lysin is relatively strong in lysis activity on pseudomonas aeruginosa in vitro. With the application of the preparation method of the pseudomonas aeruginosa bacteriophage lysin provided by the invention, the mass expression and preparation of the pseudomonas aeruginosa bacteriophage lysin can be achieved, and the preparation method issimple in separation and purification and is low in cost.
Owner:SICHUAN INDAL INST OF ANTIBIOTICS CHINA NAT PHARMA GROUP CORP
Preparation of tans-3,5- dihydroxy-4'- acetylamido-stilbene
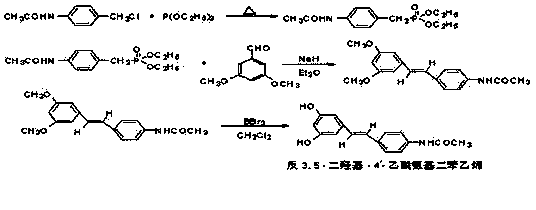
InactiveCN103387508ASimple separation and purificationQuick responseOrganic compound preparationCarboxylic acid amides preparationPhosphorous acidTriethylphosphite
The invention relates to preparation of a novel resveratrol derivative trans-3,5-dihydroxy-4'- acetylamido-stilbene, and belongs to the fields of organic chemistry and food chemistry. The invention aims to solve the technical problems by providing a preparation process of the resveratrol derivative tans-3,5-dihydroxy-4'- acetylamido-stilbene, wherein the preparation process has mild reaction conditions, high reaction selectivity, little side reaction, simple separation of products and high yield. The preparation process is characterized in that raw materials including 3,5-dimethoxybenzaldehyde, p-acetamido benzyl chloride, triethyl phosphite and boron tribromide are subjected to three step of chemical reactions to obtain the product trans-3,5-dihydroxy-4'- acetylamido-stilbene.
Owner:CHANGSHA UNIVERSITY OF SCIENCE AND TECHNOLOGY
Separation culture method of sponge symbiosis blue alga
ActiveCN103849586AHigh puritySimple separation and purificationBacteriaMicroorganism based processesMicrosphereAntibiotic Y
The invention discloses a separation culture method of sponge symbiosis blue algae. The method is characterized by comprising the following steps: selecting a sample with blue-green or yellow sponge from ocean, soaking the sponge sample, subsequently removing the impurities and then cutting into pieces, adding the sponge pieces and sterilized seawater containing antibiotics into a culture bottle till the lower parts of the sponge pieces are soaked in the water and the upper parts are exposed out of the water level, further treating the culture bottle in an carbon dioxide environment under certain temperature and illumination conditions, cutting the sponge pieces into pieces, taking the sponge pieces of dark color, stirring the sponge pieces, then mixing the sponge pieces with the sterilized seawater, putting into a cell culture plate for culturing till blue alga colonies grow out from the cell culture plate, diluting the sterilized seawater with the blue alga colonies, selecting a microsphere with only one algal filament till each hole of the cell culture plate contains the microsphere with one algal filament, and performing temporary rearing on the cell culture plate so as to obtain a product. The method has the advantages that a great deal of symbiosis glue algae can be rapidly separated with good culture effect and operation is convenient.
Owner:NINGBO UNIV
Residual oil treatment method for recovering sterol
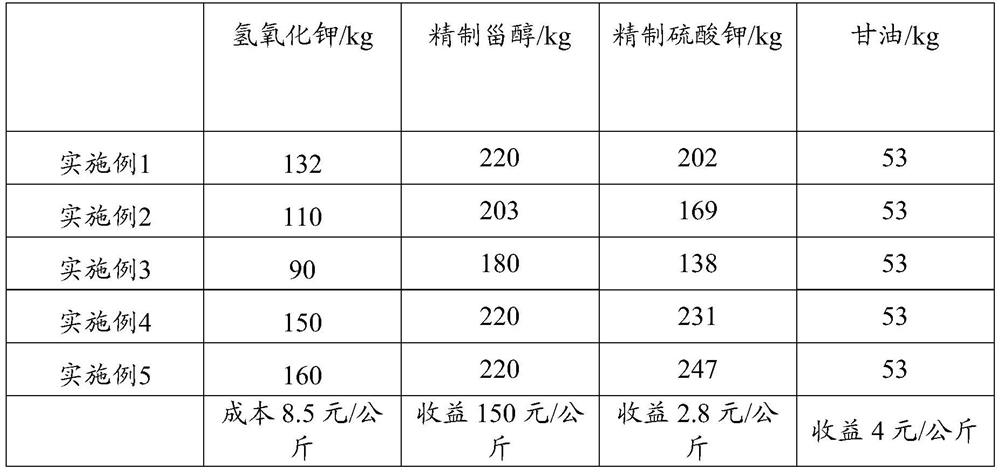
InactiveCN111718391AImprove practicalityHigh yieldAlkali metal sulfite/sulfate purificationSteroidsRefluxSterol
The invention provides a residual oil treatment method for recovering sterol, which comprises the following steps: heating potassium hydroxide and methanol for reflux, and reacting to obtain a mixed solution, wherein the heating reflux temperature is 65 DEG C, and the heating reflux time is 4 hours; mixing residual oil and the mixed solution for transesterification, and a transesterification product is obtained, wherein the temperature of the transesterification reaction is 80 DEG C, and the time is 4 hours; acidifying the transesterification product, and standing for separation to obtain an upper-layer oil phase and a lower-layer water phase; carrying out water washing treatment on the upper-layer oil phase, and then standing for separation to obtain a water washing solution and a secondupper-layer oil phase, wherein the temperature of the water for washing treatment is 80 DEG C; combining the lower-layer water phase and the water washing liquid, and then carrying out evaporation separation to respectively obtain water, methanol, crude potassium salt and glycerol; washing and drying the crude sylvite to obtain refined sylvite; and sequentially carrying out cold separation crystallization, filtration and refining treatment on the second upper-layer oil phase to obtain the refined sterol. The method is low in sterol recovery cost.
Owner:FUJIAN GLANNY BIO ENG
Method for preparing glucosamine hydrochloride through mushroom dregs for producing glutamic acid
ActiveCN106188168ALess quantityReduce manufacturing costSugar derivativesSugar derivatives preparationAlcoholGlucosamine Hydrochloride
The invention discloses a method for preparing glucosamine hydrochloride through mushroom dregs for producing glutamic acid. The method comprises the following steps that firstly, the wet glutamic acid mushroom dregs are taken, an ethanol solution with the volume being 2-3 times that of the glutamic acid mushroom dregs is added into the glutamic acid mushroom dregs, reflux extracting and filtering are carried out, and filter residues are obtained; secondly, water is added into the filter residues, ultrasonic extracting and filtering are carried out, and filtrate is obtained; thirdly, the filtrate is evaporated to dryness, a dry substance is obtained, water and hydrochloric acid are sequentially added into the dry substance, hydrolyzing is carried out for 3-5 hours under the condition that the temperature ranges from 15 DEG C to 30 DEG C, after hydrolyzing is completed, methyl alcohol is added, standing is carried out, and liquid supernatant is poured out; fourthly, the pH of the liquid supernatant is adjusted to 7-7.2, filtering is carried out, and white precipitate is obtained; methyl alcohol is added into the white precipitate, stirring is carried out for dissolving, methyl alcohol liquid supernatant is obtained, methyl alcohol is added into residual white precipitate, the operation is repeated multiple times, the methyl alcohol liquid supernatant is mixed and concentrated to be dry, and the glucosamine hydrochloride is obtained. The method for efficiently preparing glucosamine hydrochloride through the mushroom dregs for producing the glutamic acid is established for the first time, the number of kinds of needed reagents and the amount of the needed reagents are small, and the preparation cost of the glucosamine hydrochloride is reduced.
Owner:SHANDONG NORMAL UNIV
Method for accelerating biological production of 5-aminovaleric acid
ActiveCN106191089ASimple separation and purificationReduce consumptionHydrolasesPeptidesHydrolase GeneAminopentamide
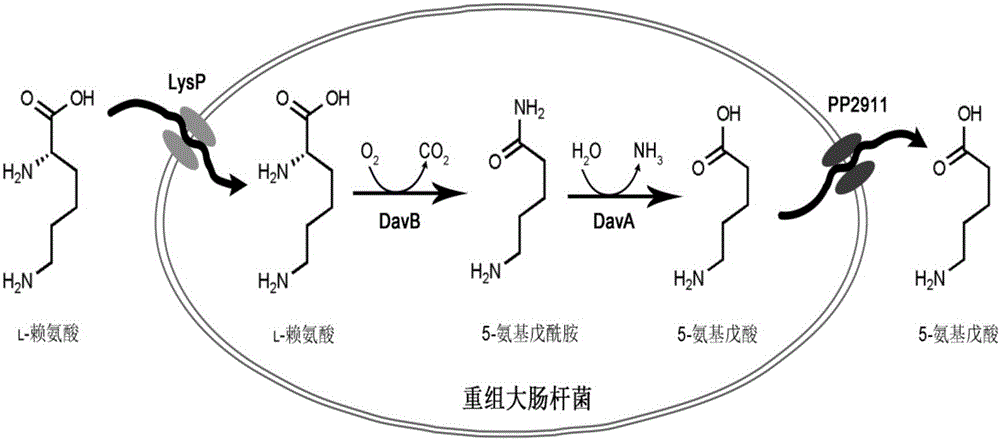
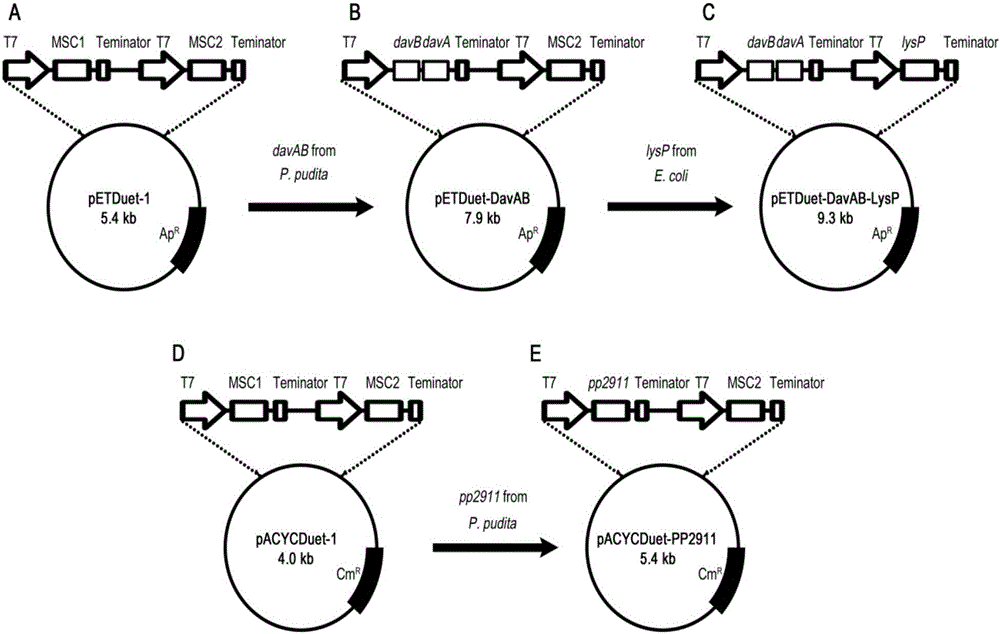
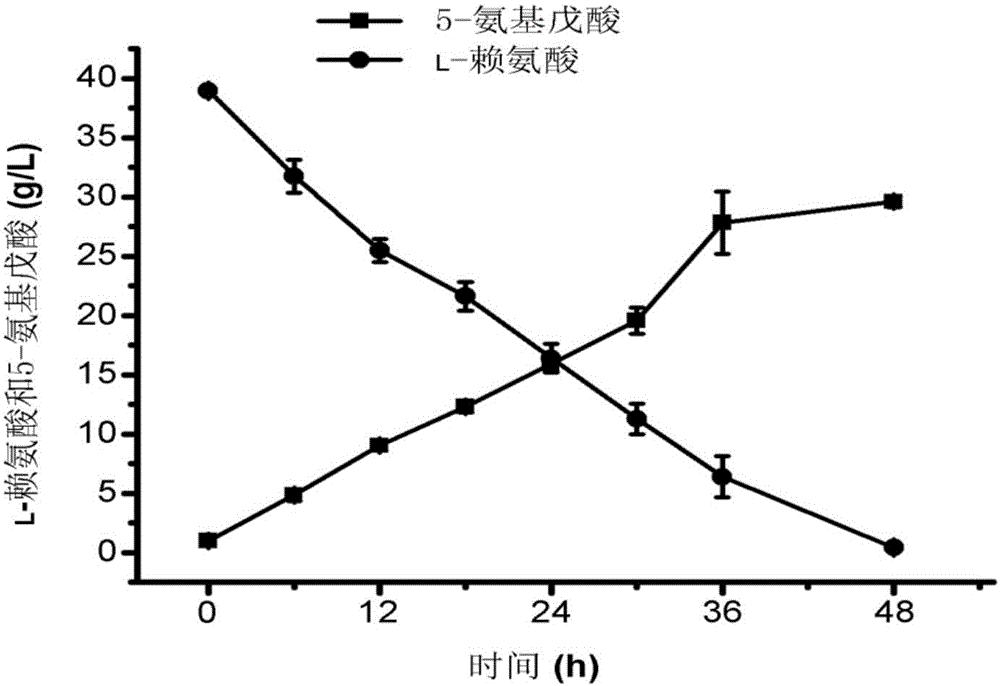
The invention discloses a method for accelerating biological production of 5-aminovaleric acid. Through co-expression of a lysine specific permease gene lysP and a 4-aminobutyric acid transporter gene pp2911 in an engineering bacterium that an L-lysine 2-monooxygenase gene davB and a [delta]-amino valeramide hydrolase gene davA are co-expressed, the speed and the yield of producing the 5-aminovaleric acid by catalyzing L-lysine through the engineering bacterium are improved. In comparison with a recombinant bacterium that either the L-lysine 2-monooxygenase gene davB or the [delta]-amino valeramide hydrolase gene davA is expressed, the method, which achieves the co-expression of the lysine specific permease gene lysP and the 4-aminobutyric acid transporter gene pp2911, can improve the yield of the 5-aminovaleric acid by virtue of the genetically engineered bacterium by 67.3%, and the conversion rate of the 5-aminovaleric acid can reach 0.94mol / mol or above. The method disclosed by the invention has a broad application prospect for the industrial production of the 5-aminovaleric acid.
Owner:SHANDONG UNIV
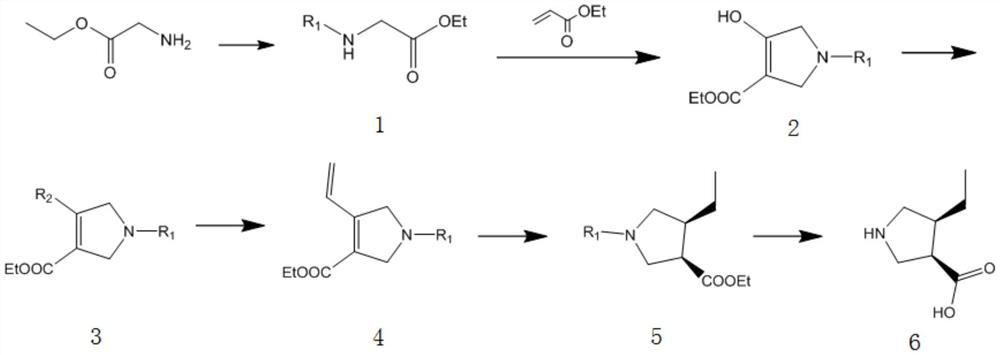
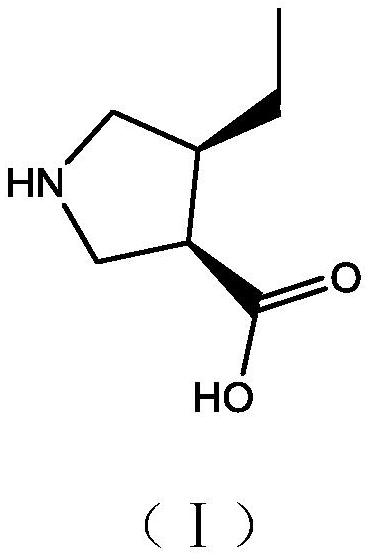
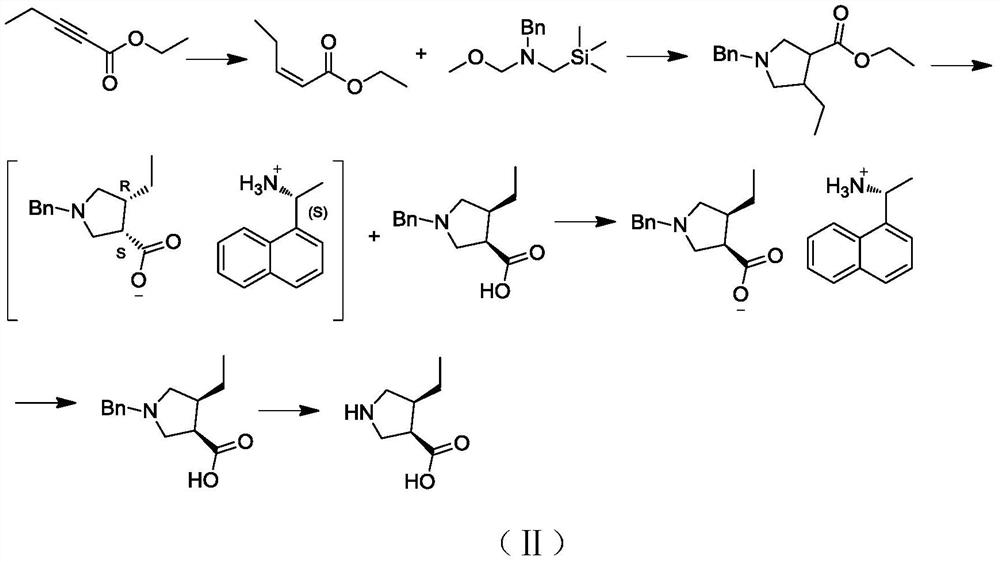
A kind of preparation method and application of (3r,4s)-4-ethylpyrrolidine-3-carboxylic acid compound
ActiveCN111072543BHigh yieldHigh chiral purityOrganic chemistryBulk chemical productionGlycineDrugs synthesis
The embodiment of the invention discloses a preparation method and application of a (3R,4S)-4-ethylpyrrolidine-3-carboxylic acid compound, which relates to the technical field of drug synthesis. In the present invention, ethyl glycine is used as a raw material, and (3R,4S)-4-ethylpyrrolidine-3-carboxylate is synthesized sequentially through amino group protection, ring closure, substitution, coupling, catalytic hydrogenation, hydrolysis and deprotection acid. The target product synthesized by this reaction has high yield and chiral purity, and also has the advantages of simple and easy-to-obtain raw materials, simple synthesis route, mild reaction conditions, simple separation and purification operation, and low synthesis cost.
Owner:北京海美桐医药科技有限公司
Orexin-A pegylation modifier and preparation method thereof
InactiveCN103804483AGood product uniformitySimple separation and purificationNervous disorderPeptide/protein ingredientsChromatographic columnPhosphate
The invention discloses an Orexin-A pegylation modifier which is connected with polyethylene glycol by an N-terminal amino covalent bond of Orexin-A. A preparation method of the Orexin-A pegylation modifier comprises the steps of enabling the Orexin-A and methoxy polyethylene glycol propionaldehyde or methoxy polyethylene glycol succinamide carbonate to undergo a reaction for 0.5-24 hours under the conditions that the pH is 4-9, and the temperature is 4-37 DEG C, wherein the molar ratio of the Orexin-A to the methoxy polyethylene glycol propionaldehyde or the methoxy polyethylene glycol succinamide carbonate is 1: 5-1: 50; then, adding small molecule amino acid to stop the reaction; desalting the product obtained in the first step by a Sephadex G-50 chromatographic column; after that, sampling the protein collected after desalting onto a QAE-Sephadex A-50 anion exchange column; and carrying out elution by a 10mM PB (phosphate buffer) solution, collecting the eluting peak, concentrating and carrying out freeze drying to obtain the Orexin-A pegylation modifier. The Orexin-A pegylation modifier is good in stability, long in half-life period and low in toxic and side effects.
Owner:FOURTH MILITARY MEDICAL UNIVERSITY
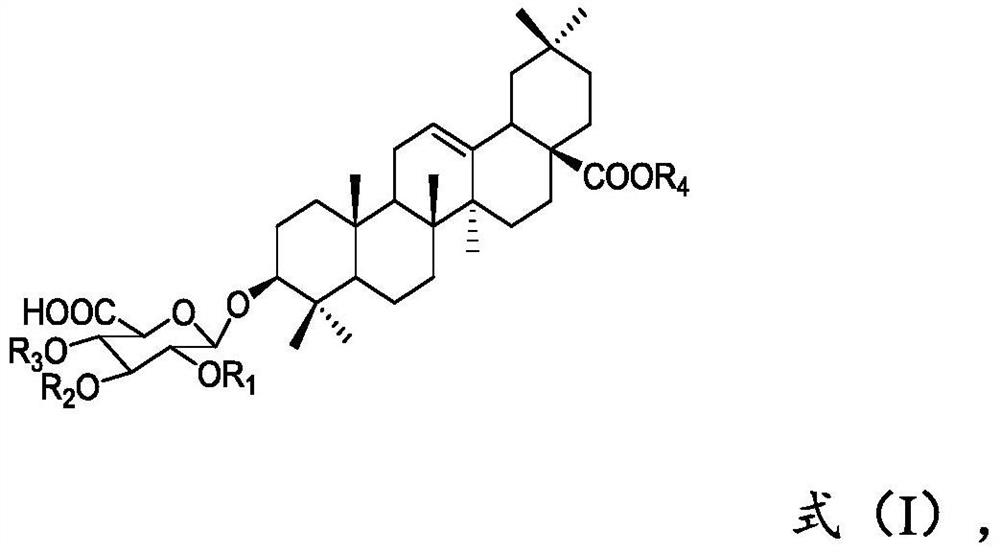
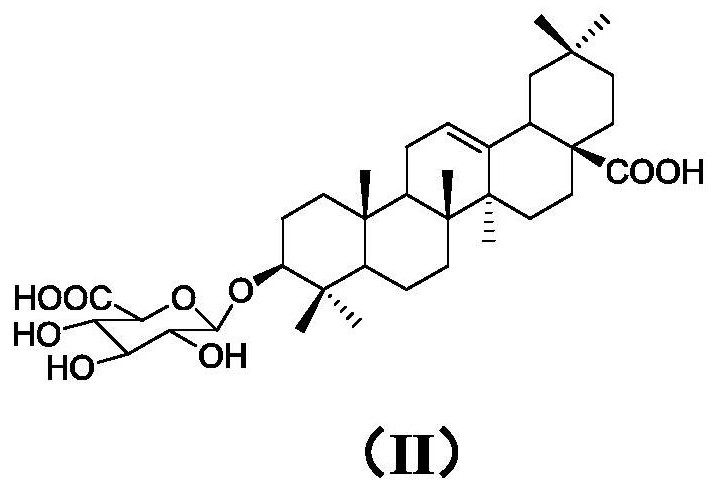
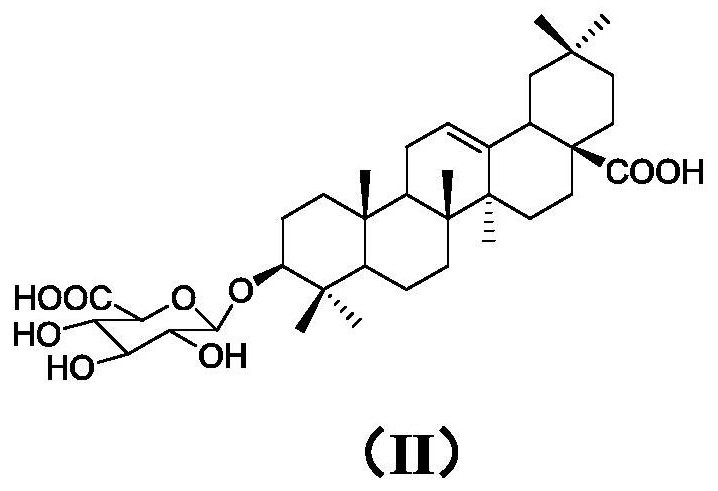
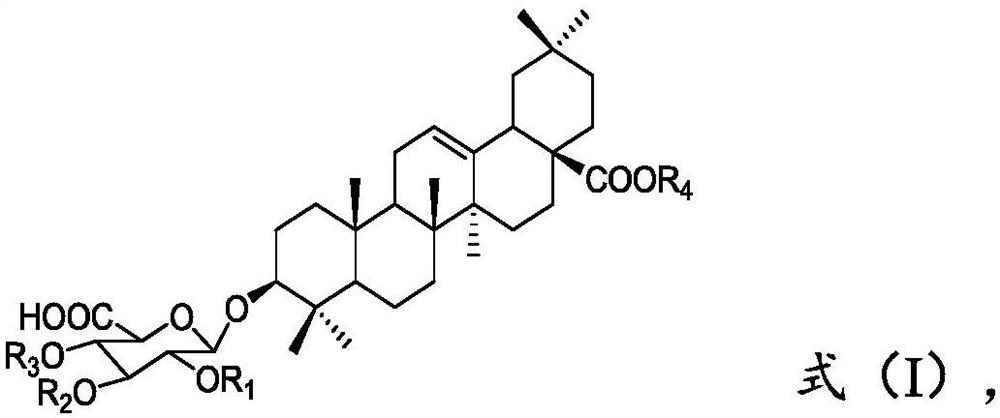
Preparation method of momordin Ib
ActiveCN111848716ASimple separation and purificationSuitable for industrial productionSteroidsTriterpeneElution
The invention provides a preparation method of momordin Ib. The preparation method comprises the following steps: firstly, mixing and reacting a plant containing a triterpenoid saponin derivative witha structural unit shown in a formula (I) or a plant extract containing the triterpenoid saponin derivative with the structural unit shown in the formula (I) with acid; (2) dissolving the momordin Ibin water to obtain an extracting solution, adjusting the pH value of the extracting solution in the step (1) to 6.5-7.5 by using alkali, and separating by using column chromatography to obtain a sample containing the momordin Ib; and finally, recrystallizing the obtained sample containing the momordin Ib to obtain a pure product of the momordin Ib. According to the invention, a plant or a plant extract rich in a triterpenoid saponin derivative with a structural unit shown as a formula (I) is used as a raw material for preparing momordin Ib; results show that the method not only is simple in separation and purification operation, but also can realize large-scale acquisition of momordin Ib, and solvents for elution and recrystallization in each step can be recycled for multiple times and thus the method is suitable for industrial production.
Owner:XISHUANGBANNA TROPICAL BOTANICAL GARDEN CHINESE ACAD OF SCI +1
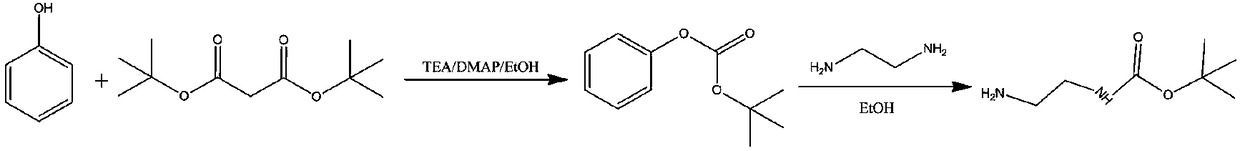
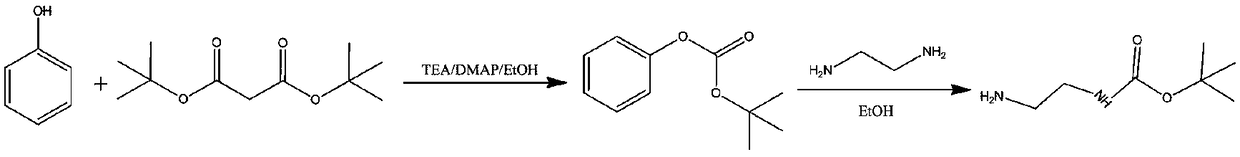
Preparation method of N-Boc-1, 2-diaminoethane
InactiveCN108689885ALow costSimple separation and purificationCarbamic acid derivatives preparationOrganic compound preparationOrganic solventReaction temperature
The invention relates to a preparation method of N-Boc-1, 2-diaminoethane. The preparation method comprises 1) dissolving phenol, a catalyst and a base in a first organic solvent, and adding di-tert-butyl dicarbonate into the solution with the first solvent drop by drop for a reaction to obtain tert-butyl phenyl carbonate, and 2) dissolving 1, 2-diaminoethane in the first organic solvent, and adding tert-butyl phenyl carbonate into the solution drop by drop for a reaction to obtain a final product. The preparation method has simple separation and purification processes, has a small reaction temperature range and a high reaction yield and realizes a low raw material cost.
Owner:湖南华腾制药有限公司
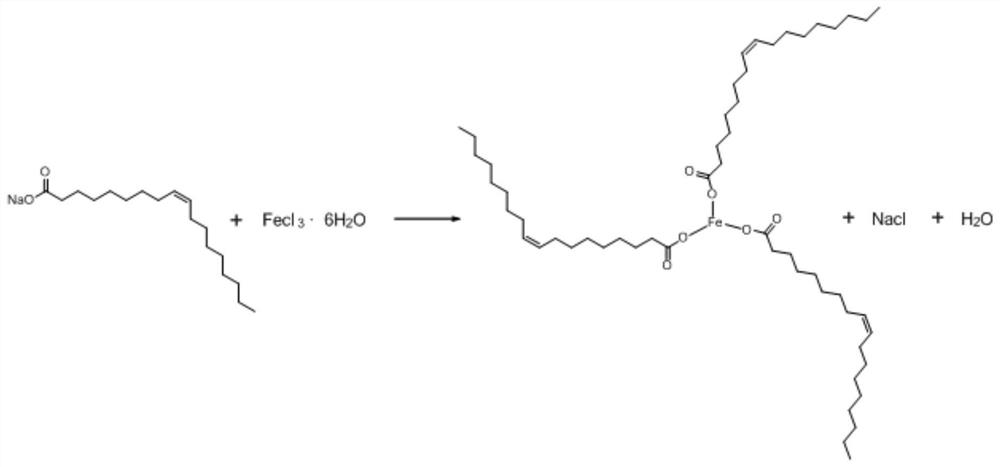
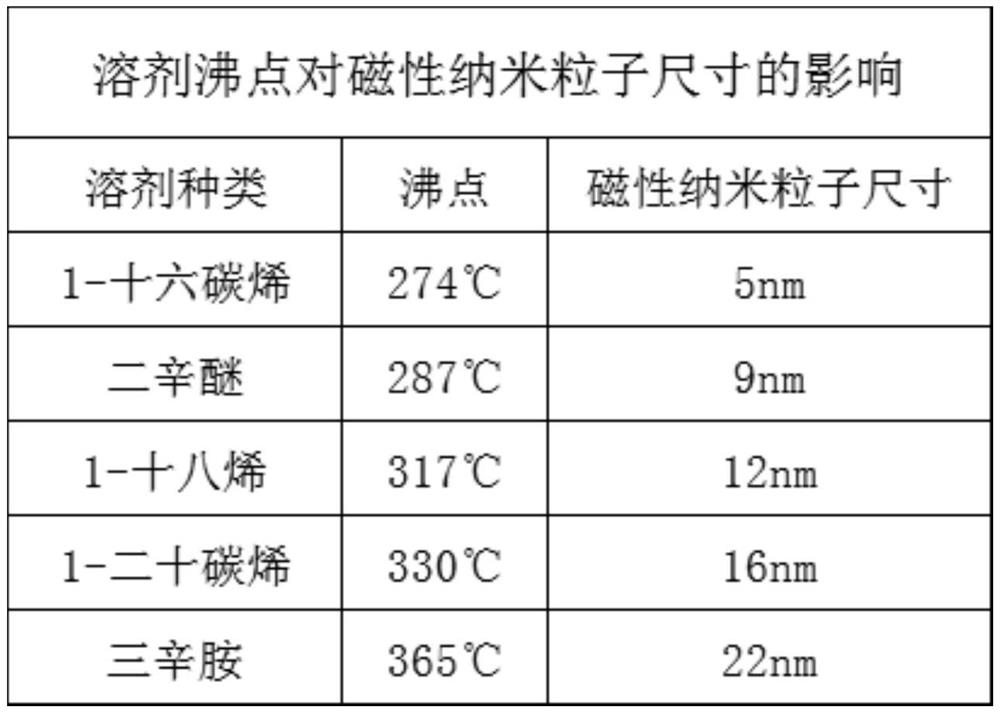
Method for simply and conveniently preparing monodisperse magnetic nanoparticles in quantity
InactiveCN112723418AEasy to manufactureSimple separation and purificationMaterial nanotechnologyIron oxides/hydroxidesHydration reactionMagnetite Nanoparticles
The invention relates to a method for simply and conveniently preparing monodisperse magnetic nanoparticles on a large scale. The method comprises the following steps: step 1, carrying out a reflux reaction on sodium oleate and ferric chloride hexahydrate in a mixed solvent of absolute ethyl alcohol, deionized water and normal hexane to prepare an iron oleate complex; step 2, taking water as a solvent, and extracting and purifying the ferric oleate complex for multiple times; and step 3, dissolving the purified waxy ferric oleate complex and oleic acid in a 1-octadecene solvent, carrying out high-temperature decomposition, then carrying out precipitation by using ethanol, and carrying out centrifugation and drying to obtain monodisperse magnetic nanoparticles. According to the method for simply and conveniently preparing the monodisperse magnetic nanoparticles on a large scale, the reaction conditions are convenient to control, product separation and purification are simple and convenient, large-scale preparation of the magnetic nanoparticles can be simply and conveniently carried out, and the sizes of the magnetic nanoparticles can be regulated and controlled through the concentration of oleic acid and the boiling point of a reaction solvent; and magnetic nanoparticles with different sizes can be conveniently prepared.
Owner:金华诺科生物科技有限公司
A method for preparing o-fluoroaniline by hydrogenation of o-fluoronitrobenzene
ActiveCN104130129BEfficient hydrogenationHigh catalytic activityOrganic compound preparationAmino compound preparationHydrogenLiquid state
The invention discloses a method for preparing o-fluoroaniline by hydrogenating o-fluoronitrobenzene. The method comprises the following steps: 1, filling a fixed bed reactor with a catalyst, introducing air and heating; 2, uniformly mixing o-fluoronitrobenzene steam and preheated hydrogen to obtain mixed gas, introducing the mixed gas into the fixed bed reactor, and performing a catalytic hydrogenation reaction in the presence of the catalyst; and 3, delivering the material subjected to the catalytic hydrogenation reaction into a condenser, condensing the material to convert the o-fluoronitrobenzene steam in the material into a liquid state, and separating the liquid-state o-fluoronitrobenzene in an oil-water separator to obtain oil-state o-fluoroaniline. The o-fluoronitrobenzene is catalytically hydrogenated by adopting the method, the treatment amount is large, the o-fluoronitrobenzene is completely converted, the molar conversion rate of the o-fluoronitrobenzene is 100 percent, and the molar percentage of a defluorinated product is less than 0.1 percent; and the product separating and purifying operation is simple at low energy consumption, and no toxic or harmful ions are doped in the product.
Owner:XIAN CATALYST NEW MATERIALS CO LTD
Method for preparing selenium substitution papain
A process for preparing selenopapain from papain and sodium selenohydride through dissolving papain in phosphate solution, adding excessive sodium selenohydride, seleno substitution under protection of N2, freeze drying, dissolving in phosphate solution centrifugal removal of Se, removing salt, collecting protein peaks, and freeze drying. Its advantages are simple process high activity of glutathion peroxidase, and excellent antioxidizing power.
Owner:JILIN UNIV
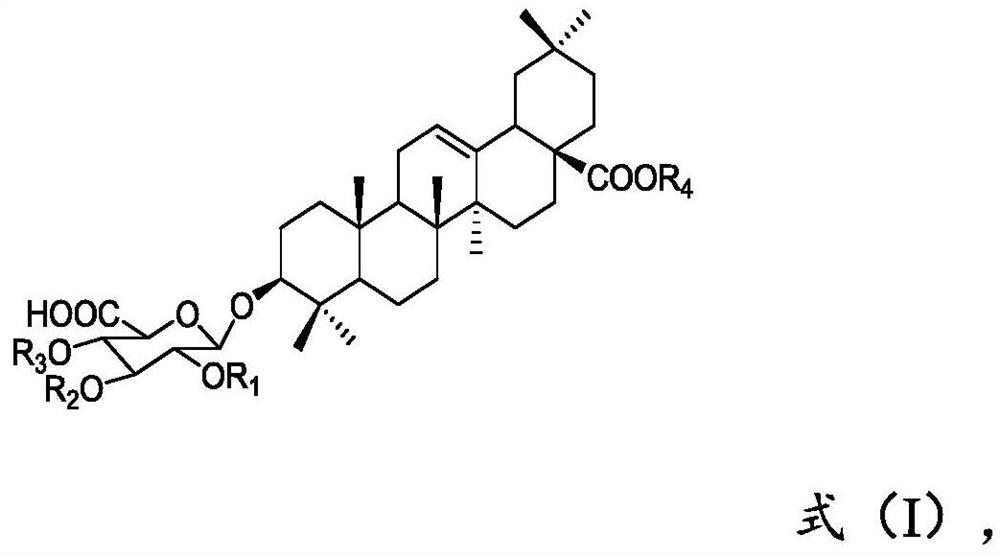
A kind of preparation method of momordin Ib
ActiveCN111848716BSimple separation and purificationSuitable for industrial productionSteroidsTriterpeneMomordin
The present invention provides a kind of preparation method of momordin Ib, by at first the plant that contains the triterpene saponin derivative of formula (I) structural unit or the plant extract that contains the triterpene saponin derivative of formula (I) structural unit and acid Mixing the reaction to obtain an extract, then adjusting the pH of the extract in step 1) to 6.5 to 7.5 with alkali, and then separating by column chromatography to obtain a sample containing momordin Ib; Crystallized to obtain pure momordin Ib. Among them, the present invention uses plants or plant extracts rich in triterpene saponin derivatives with structural units of formula (I) as raw materials for preparing momordin Ib. It was found that the method not only has simple separation and purification operations, but also can realize momordin Ib A large amount is obtained, and the solvents eluted and recrystallized in each step can be recycled many times, which is suitable for industrial production.
Owner:XISHUANGBANNA TROPICAL BOTANICAL GARDEN CHINESE ACAD OF SCI +1
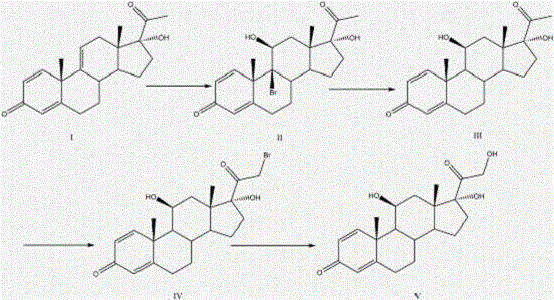
A kind of preparation method of prednisolone
The application relates to a method for synthetizing prednisolone. Double bond bromination, reductive debromination, bromination and hydrolysis reaction are carried out on a compound with a structure of formula I so as to generate the prednisolone. The method is high in yield, is easy to operate, and is applicable to industrial production.
Owner:李竞
A kind of preparation method of Apixaban
ActiveCN105732622BAdaptable to conditionsFast aminolysisOrganic chemistryP-NitroanilineNitro reduction
The invention discloses a preparation method of apixaban.The method comprises the steps that paranitroaniline serves as the raw material, paranitroaniline and delta-valerolactone are subjected to amidation ring-opening, substituting and ring-closing reactions under the action of AlMe3, and a compound 8 is obtained; the compound 8 is subjected to alpha-position dichloro substituting and condensation-elimination reactions, and a compound 7 is obtained; the compound 7 and a compound 6 are subjected to [3+2] cyclization-elimination and nitro reduction, and a compound 4 is obtained; the compound 4 sequentially reacts with delta-valerolactone and ammonium chloride under the action of AlMe3, and a compound 3 is obtained; the compound 3 is subjected to substituting and ring closing, and apixaban is obtained.According to the preparation method of apixaban, paranitroaniline and delta-valerolactone which are low in price are adopted to serve as the raw materials, operation of the whole route is simple, conditions of each reaction are mild, and the synthesizing method is easy to operate, high in yield and purity and suitable for industrial production.
Owner:SHANDONG LUOXIN PHARMA GRP CO LTD
Synthesis method of N-aryl hydroxylamine acid
InactiveCN102702027AMild operating conditionsSimple separation and purificationOrganic chemistryChemistryOrganic solvent
The invention discloses a synthesis method of N-aryl hydroxylamine acid, wherein the synthesis method comprises the following steps of mixing aniline with aromatic aldehyde, reacting the aniline and the aromatic aldehyde at the room temperature to obtain an intermediate product; dissolving the obtained intermediate product into an organic solvent, adding M-chloroperbenzoic acid, and conducting the reaction at the room temperature, so as to obtain the N-aryl hydroxylamine acid. In comparison with the traditional acyl chloride production methods, the production method provided by the invention has the advantages that the operation condition is milder, the reaction is carried out at the room temperature without rigorous de-watering operations and devices, the separation and purification operations of the obtained intermediate product or final product are simple, and the yield of the final product is as high as 80%, so that the method provided by the invention is suitable for industrial production.
Owner:LANZHOU UNIVERSITY
A kind of synthetic method of Apixaban
InactiveCN103896940BSimple separation and purificationLower synthesis costOrganic chemistryEthyl chloroformateEthyl ester
The invention discloses a synthetic method of novel Apixaban. The method comprises the following steps: (i) hydrolyzing an Apixaban precursor compound (II) to obtain a carboxylic acid product; and (ii) mixing the carboxylic acid product obtained in the step (i) with ethyl chloroformate, reacting in the presence of diisopropylethylamine at the temperature of 0-5 DEG C for 3-5 hours; then introducing ammonia gas and reacting to obtain an ammonolysis product, namely, Apixaban. By adopting the method for preparing Apixaban by using the Apixaban precursor compound (II), the yield of Apixaban can be up to 93 percent. In the entire synthesis route, the minimum yield in each step is over 76 percent at least, and the total yield is about 33 percent. In the entire process, the use of precious catalysts or auxiliary reagents is avoided.
Owner:CHEMFUTURE PHARMATECH JIANGSU
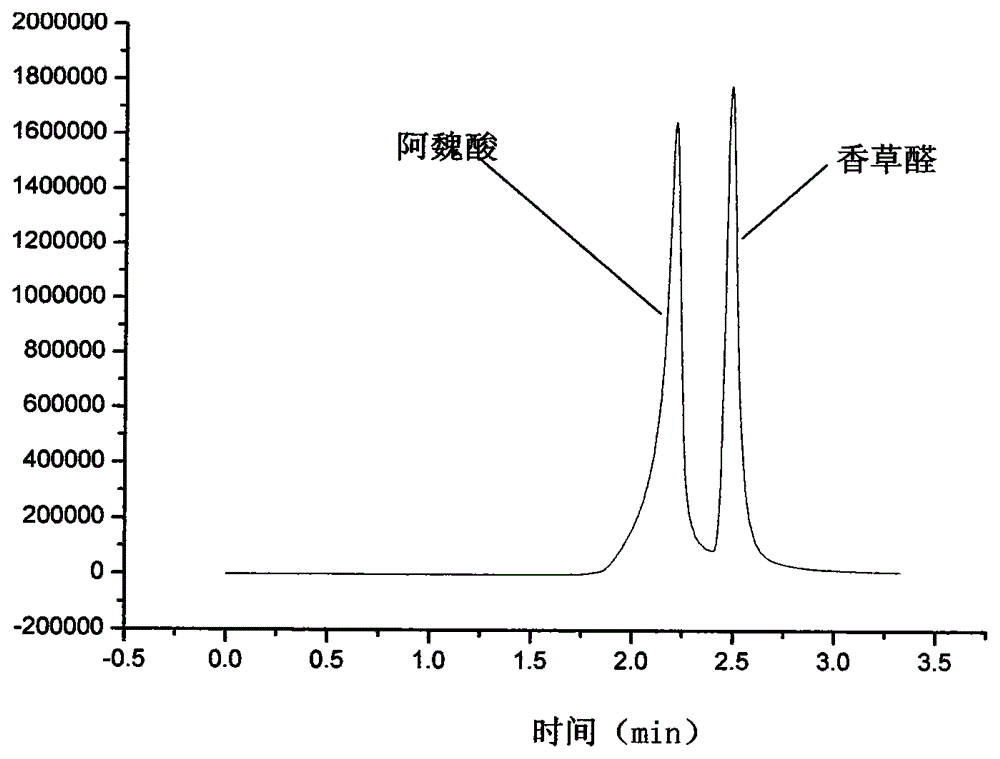
Method for synthesizing ferulic acid by microwave radiation
InactiveCN104151156AShort reaction timeSimple separation and purificationOxygen-containing compound preparationOrganic compound preparationSolventAcetic anhydride
The invention discloses a method for synthesizing ferulic acid by microwave radiation. The method comprises the following steps: adding vanillic aldehyde and anhydrous potassium carbonate to acetic anhydride, evenly mixing the raw materials, and carrying out microwave radiation until a reaction mixture is completely molten into liquid; cooling the molten liquid to room temperature and then adding ice water to mix evenly; standing the mixture for over 24 hours, adding ice water again, fully washing and carrying out suction filtration to obtain a filter cake; hydrolyzing the filter cake in an NaOH solution, and carrying out suction filtration; acidizing the obtained filtrate until the pH is 2-3, carrying out suction filtration, and recrystallizing the filtrate cake by using absolute ethyl alcohol, so as to obtain yellow needle crystal; drying the yellow needle crystal to obtain ferulic acid. Acetic anhydride is a reactant, and also is a solvent; acetic anhydride can react to the maximal extent; anhydrous potassium carbonate is non-toxic and pollution-free; acetic anhydride is taken as the solvent, and can be removed in a washing manner during impurity removal; the requirements of green chemistry are met, therefore, the method disclosed by the invention is short in reaction time, and easy in separation and purification, the productive rate can be up to 68%, and a new direction is provided for preparation of ferulic acid.
Owner:黄金凤