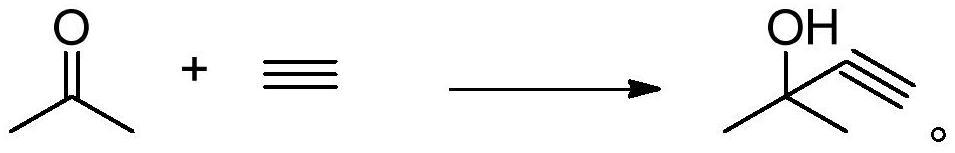Patents
Literature
52 results about "Hexyne" patented technology
Efficacy Topic
Property
Owner
Technical Advancement
Application Domain
Technology Topic
Technology Field Word
Patent Country/Region
Patent Type
Patent Status
Application Year
Inventor
The hexynes are a subgroup from the group of alkynes. It consists of several isomeric compounds having the formula C₆H₁₀.
Sulfur-containing bis-imidazoline type carbon dioxide corrosion inhibitor and preparation method thereof
InactiveCN102321463AImprove protectionGood water solubilityOrganic chemistryBorehole/well accessoriesCarbon dioxide corrosionActive agent
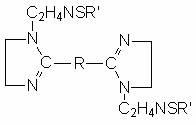
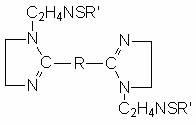
The invention discloses a sulfur-containing bis-imidazoline type carbon dioxide corrosion inhibitor and a preparation method thereof. The corrosion inhibitor comprises the following components in percentage by weight: 20-50 percent of sulfur-containing bis-imidazoline quaternary ammonium salt, 1-5 percent of nonionic surfactant, 40-70 percent of low molecular alcohol solvent and 5-10 percent of alkynol, wherein a sulfur-containing bis-imidazoline compound has a general formula of a structure which is shown in the specification; the number of carbon atoms in R of the formula is 5 or 8; the nonionic surfactant is fatty alcohol-polyoxyethylene ether or alkylphenol ethoxylate; the low molecular alcohol solvent is methanol, ethanol, isopropanol or ethylene glycol; and the alkynol is propargyl alcohol or hexynol. In the invention, the sulfur-containing bis-imidazoline compound which is generated through dehydration between dicarboxylic acid and polyamine molecule, dehydration reaction in the molecule and further reaction with the sulfur-containing compound is adopted; the molecule contains a plurality of active adsorption centers such as two diazo five-membered heterocyclic rings, a sulfur-containing group and the like; and a formed organic adsorption membrane has better protection effect on carbon steel.
Owner:UNIV OF SCI & TECH BEIJING
Ink-jet ink compositions comprising a latex polymer and diol additives
A solvent system has been found which aids in start-up, drop ejection, decap and high frequency firing above 10 kHz for inks which contain latex polymers. Two solvents work in conjunction with each other: 3-hexyne-2,5-diol and 1,2-octanediol. These two solvents in combination improve printability in latex polymer-containing ink-jet inks. Such ink-jet inks also include one or more pigments and a vehicle comprising at least one organic, water-miscible solvent and water.
Owner:HEWLETT PACKARD DEV CO LP
In-situ compatibilization polyolefin/polystyrene alloy and preparation method and application thereof
The invention discloses an in-situ compatibilization PO / PS (polyolefin / polystyrene) alloy and a preparation method and application thereof. The alloy is prepared by 45-65% of polyolefin, 10-30% of polystyrene, 2-5% of compatibilization initiation mother liquor and 20-25% of processing agent, and the compatibilization initiation mother liquor is obtained by dissolving crosslinking agent and crosslinking agent auxiliaries to oily carrier., wherein the crosslinking agent is at least one of 2, 5-dimethyl-2, 5-di (tertiary butyl peroxide) hexine, 2, 5-dimethyl-2, 5-di (tertiary butyl peroxide) hexane or dicumyl peroxide, and the crosslinking agent auxiliaries is at least one of liquid polybutadiene, prussic acid ester type substances, isocyanate substances or acrylate substances. Compared withthe dynamic sulfuration in-situ compatibilization method and the common compatibilization method, the preparation of the in-situ compatibilization PO / PS alloy is capable of effectively reducing production cost of the PO / PS alloy, the PO / PS alloy prepared has lower molding shrinkage rate, higher modulus and excellent overall performance and can be used as substitutes for acrylonitrile butadiene styrene partially.
Owner:KINGFA SCI & TECH CO LTD +2
Process for synthesizing sex pheromone of pine caterpillar
ActiveCN102613177AEasy to operateSynthetic raw materials are readily availableBiocidePreparation by isomerisationGrignard reagentIodide
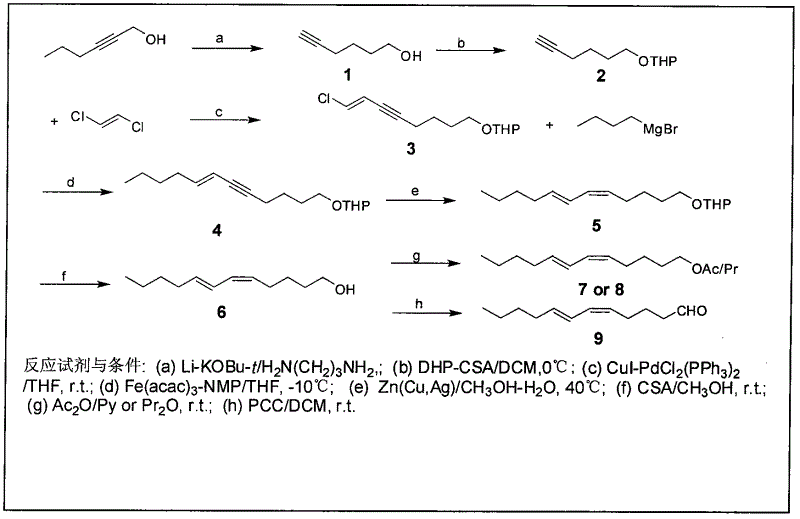
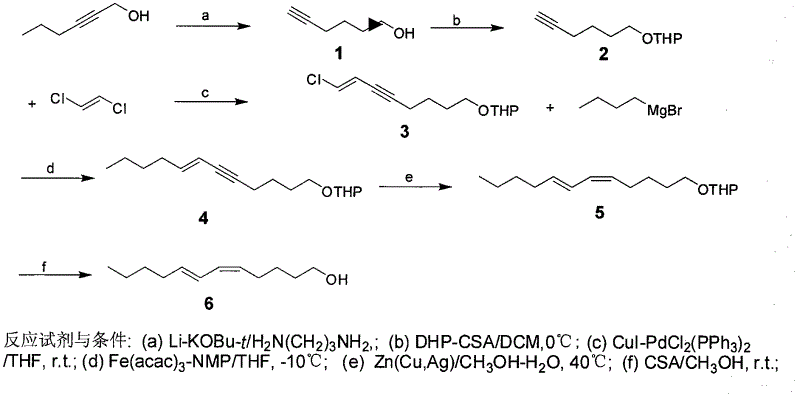
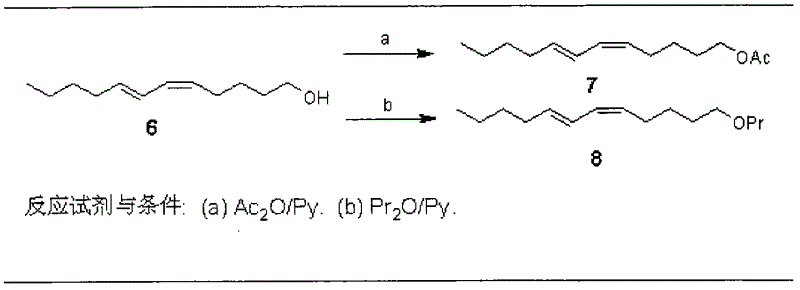
The invention discloses a process for synthesizing sex pheromone of pine caterpillar, which employs 2-hexyne-1-alcohol as an initial raw material, three-bond positional transference is carried out under the effect of lithium and propane diamine to obtain 5-hexyne-1-alcohol; under acidic condition, 5-hexyne-1-alcohol is reacted with dihydropyran to obtain 1-THP-5-hexyne-1-alcohol protected by THP on hydroxyl, Under co-catalysis of metal palladium and cuprous iodide, 1-THP-5-hexyne-1-alcohol and trans-dichloroethylene are subjected to coupling reaction to generate conjugate enyne(7E)-1-THP-8-chlorine-5-alkyne-7-alkene-1-octanol; under the catalysis of metallic iron, (7E)-1-THP-8-chlorine-5-alkyne-7-alkene-1-octanol and a n-Butyl bromide grignard reagent are further subjected to coupling reaction to obtain (7E)-1-THP-5-alkyne-7-alkene-1-dodecanol, under the catalytic reduction of metal zinc, (5Z, 7E)-1-THP-dodecanol dienol; under the camphor sulfonic acid condition, (5Z, 7E)-1-THP-dodecanol dienol removes the THP protective group to obtain the final target product (5Z, 7E)-dodecanol dienol. The method of the invention has the advantages of easily available synthesis raw materials, low cost, mild reaction condition, easy operation, high yield and good stereoselectivity.
Owner:WENZHOU MEDICAL UNIV +1
Separation method of 2-methyl-3-butyn-2-ol
ActiveCN111807929AHigh purityImprove conversion rateOrganic compound preparationHydroxy compound separation/purificationHexyneDistillation
The invention belongs to the technical field of acetylene alcohol production, and particularly relates to a separation method of 2-methyl-3-butyn-2-ol, which comprises the following steps: pretreatinga reaction solution containing 2-methyl-3-butyn-2-ol to remove unreacted acetone and contained salt from the reaction solution to obtain a 2-methyl-3-butyn-2-ol crude product, with the content of 2,5-dimethyl-3-hexyn-2,5-diol in the reaction solution containing 2-methyl-3-butyn-2-ol being not higher than 0.1 wt%; separating and purifying the 2-methyl-3-butyn-2-ol crude product by virtue of membrane separation treatment and reduced pressure distillation treatment, so as to obtain a 2-methyl-3-butyn-2-ol product. According to the method disclosed by the invention, water in the product can be removed and the content of the 2,5-dimethyl-3-hexyn-2,5-diol can be reduced under the condition that the investment and the energy consumption are reduced, so that the 2-methyl-3-butyn-2-ol with relatively high purity and conversion rate is obtained.
Owner:WANHUA CHEM GRP CO LTD
Process for synthesizing 3-hexyne-2,5-diol through slurry bed based on low pressure method
ActiveCN102875332AReduce generationQuick responseOrganic compound preparationHydroxy compound preparationHexynePtru catalyst
The invention discloses a process for synthesizing 3-hexyne-2,5-diol through a slurry bed based on a low pressure method, which is characterized by comprising the following steps: mixing an acetaldehyde water solution and a catalyst in a reaction kettle of the slurry bed, and stirring to obtain a slurry solution; introducing acetylene into the slurry system from the lower part of the slurry solution, and reacting; and concentrating, separating, and performing reduced pressure distillation to obtain the 3-hexyne-2,5-diol. The method has the advantages of simple process, good safety, high yield and high purity of the prepared product, and is easy to realize industrial production.
Owner:LONGCHANG TIANKANG FINE CHEM CO LTD
Semi-bright nickel plating bath and method of using same
A nickel plating bath for plating a semi-bright nickel deposit on a substrate comprising a) nickel sulfateions; b) a soluble salt of chloroacefic acid, acetic acid, glycolic acid, proprionic acid, benzoic acid, salicylic acid or chlorobenzoic acid; and c) at least one diol selected from the group consisting of hexyne diol, butyne diol and combinations of the foregoing. The semi-bright nickel plating bath described herein produces sulfur-free semi-bright deposits over a very wide current density range. The plating bath described herein is at least substantially free of coumarin and produces desirable leveling characteristics. The bath also requires no aldehydes to achieve simultaneous thickness and electrolytic potential (STEP) and has extremely low stress and excellent ductility.
Owner:MACDERMID ACUMEN INC
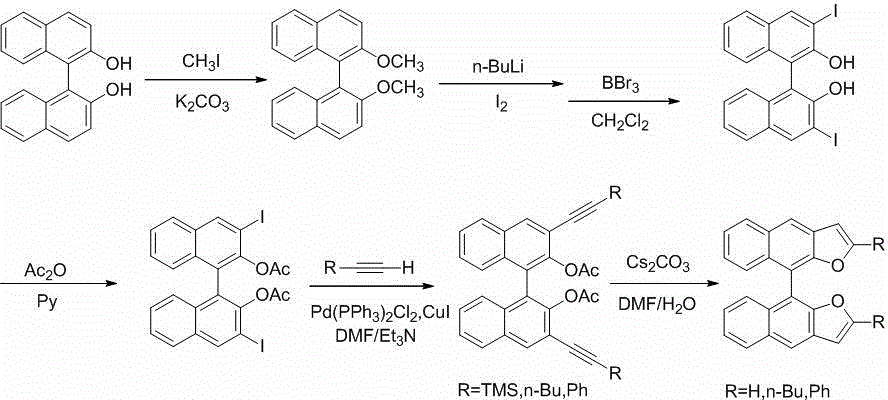
(S)-/(R)-difurodinaphthalene as well as derivatives thereof and preparation method
The invention discloses (S)- / (R)-difurodinaphthalene as well as derivatives thereof and a preparation method. The compounds have two spatial configurations which are (S)- / (R)-, and in the structure, the site 2 of furan has substituent groups which are hydrogen, and linear or branched alkyl groups or phenyl groups of C4 or below. The preparation method comprises the following steps of: reacting (S)- / (R)-binaphthol as a raw material and potassium carbonate as alkali with iodomethane in acetone so as to obtain (S)- / (R)-2, 2'-dimethoxy-1, 1'-dinaphthalene; then carrying out diiodo-reaction of a product obtained in the previous step, removing protecting groups of phenolic hydroxyl groups, and esterifying with acetic anhydride, thus obtaining (S)- / (R)-3, 3'-diiodo-2, 2'-diacetoxy-1, 1'-dinaphthalene; carrying out Sonogashira coupled reaction of (S)- / (R)-3, 3'-diiodo-2, 2'-diacetoxy-1, 1'-dinaphthalene with alkyne under the catalysis action of palladium to obtain a series of (S)- / (R)-3, 3'-di(trimethylsilylacetylene, hexynyl or phenyl ethynyl)-2, 2'-diacetoxy-1, 1'- dinaphthalene; and finally under the action of cesium carbonate, carrying out ring closing reaction to obtain the (S)- / (R)-double furodinaphthalene and derivatives thereof. A series of compounds provided by the invention broaden the range of organic electroluminescence materials.
Owner:EAST CHINA NORMAL UNIV
Removal liquid for removing blockage of weighting agent in drilling liquid
InactiveCN104073229AIncrease concentrationLess amount of methanesulfonic acidDrilling compositionHexyneActive agent
The invention discloses removal liquid for removing blockage of a weighting agent in drilling liquid. The removal liquid comprises the following components by weight percent: 5%-10% of methane sulfonic acid derivatives, 2%-5% of erythorbic acid, 0.05%-0.1% of a nonionic surfactant, 0.1%-0.5% of a corrosion inhibitor and the balance of water, wherein the methane sulfonic acid derivatives are amino-methane sulfonic acid, ethyl methane sulfonate, methyl methane sulfonate or a mixture of amino-methane sulfonic acid, ethyl methane sulfonate and methyl methane sulfonate; the nonionic surfactant is fatty alcohol-polyoxyethylene ether, dinonylphenol polyoxyethylene ether or a mixture of fatty alcohol-polyoxyethylene ether and dinonylphenol polyoxyethylene ether; the corrosion inhibitor is propynol, methyl butynol, hexynol or a mixture of propynol, methyl butynol and hexynol. According to the removal liquid, the blockage of stratum pore throat caused by weighting agent particles in drilling slurry is well removed, and especially the blockage of barite particles can be effectively removed, so that an oil-gas well is smoothly exploited after being completed; the removal liquid can be used for carbonate rock and sandstone reservoirs.
Owner:SOUTHWEST PETROLEUM UNIV
Preparation method of all-inorganic CsPbBr3 perovskite nanocrystal with controllable morphology
ActiveCN111204797AExcellent optical propertiesSolution to short lifeNanoopticsLuminescent compositionsHexyneNanocrystal
The invention relates to a preparation method of an all-inorganic CsPbBr3 perovskite nanocrystal with controllable morphology, and belongs to the technical field of preparation of inorganic nano luminescent materials. Specifically, a thermal injection method commonly used for synthesizing perovskite is adopted, and the innovation point is that 5-hexynoic acid is used as a surface ligand to passivate a product when the material is prepared. The morphology of the CsPbBr3 perovskite is regulated and controlled by controlling the use amount and the synthesis temperature of the ligand, and the method has the advantages of being rapid in synthesis, easy and convenient to operate and high in repeatability and is a reliable method for synthesizing the perovskite nano material. By regulating and controlling the morphology of the CsPbBr3 perovskite nanocrystal, carefully controlled emission of light with different wavelengths and high brightness can be realized under excitation. The CsPbBr3 perovskite nanocrystal has a wide application prospect in the aspects of LED lamps, screen display and the like.
Owner:NANJING UNIV OF TECH
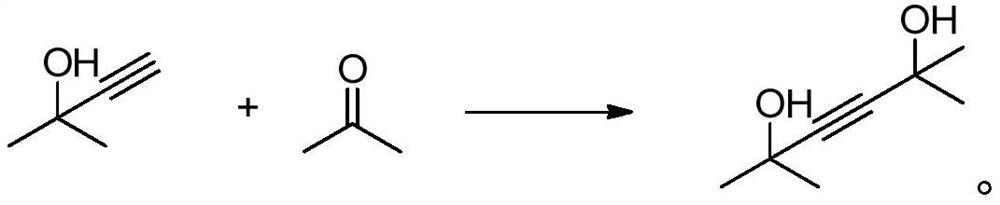
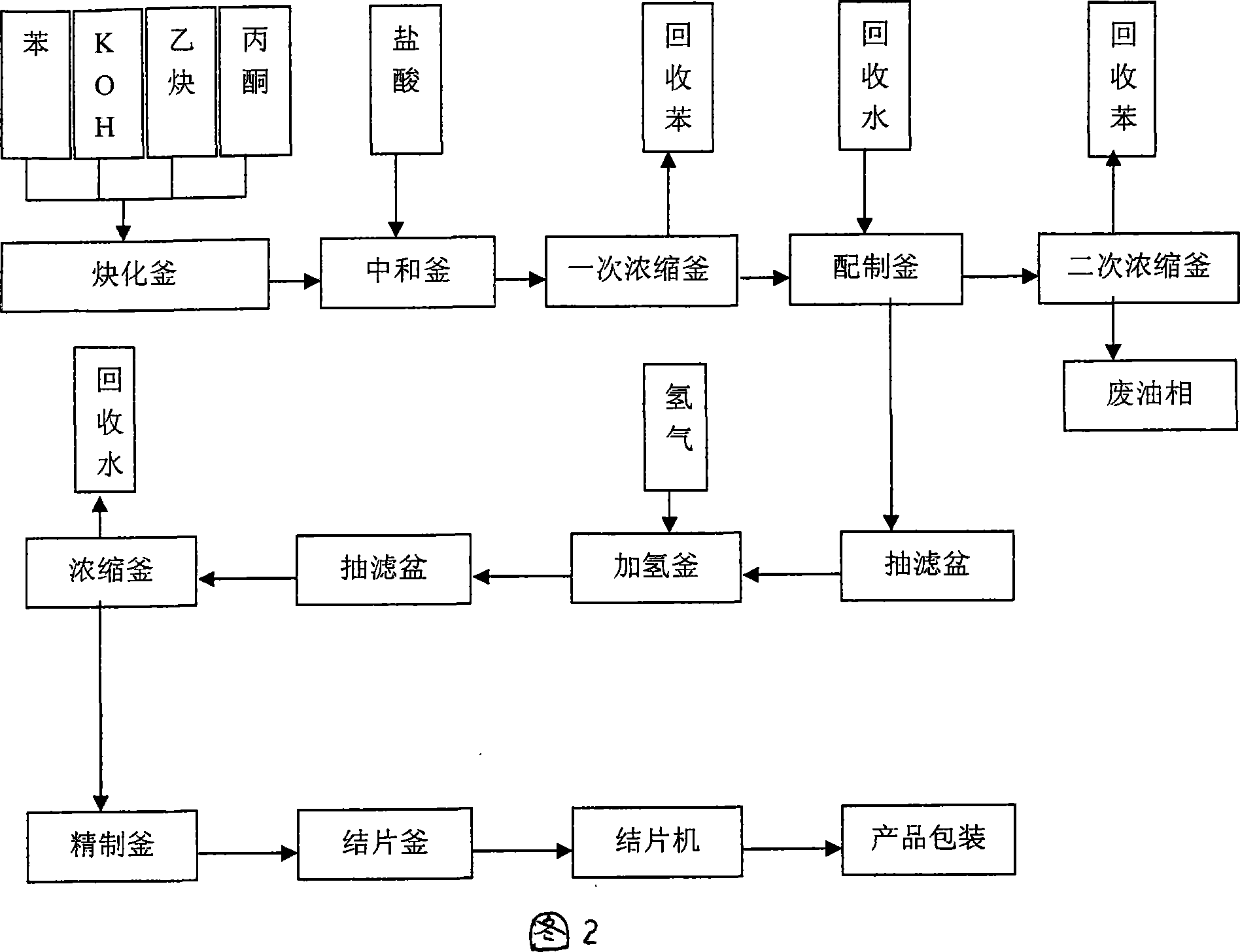
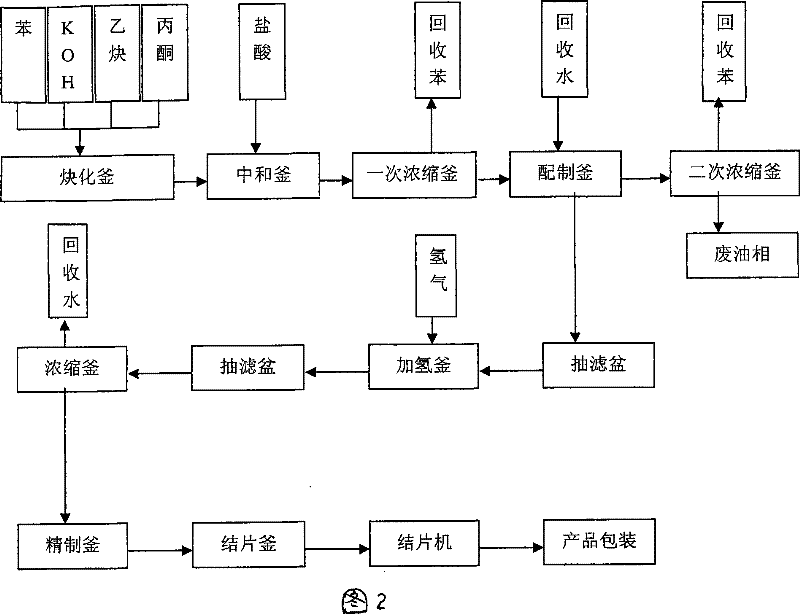
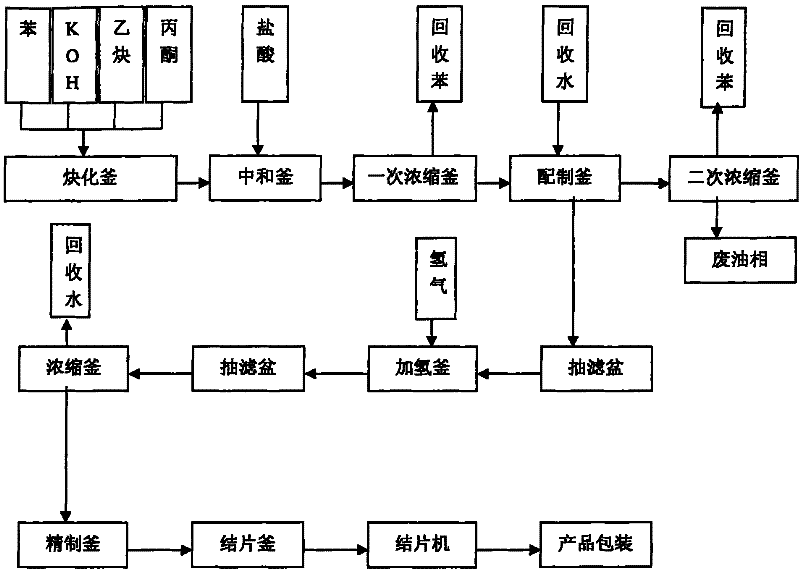
Technique for producing 2,5-dimethyl-2,5-hexandiol by extraction and decompression distillation method
ActiveCN101234950AEfficient separationImprove product qualityOrganic compound preparationHydroxy compound preparationHexynePotassium hydroxide
The invention discloses a new technology for producing 2, 5-dimethyl-2, 5-hexanediol using the method of extraction and reduced-pressure distillation. The method adopts acetylene and acetone, which are condensed with potassium hydroxide in benzene solvent and then neutralized with hydrochloric acid to get a feed solution containing hexyne-diol, oil phase and benzene. After condensation and extraction operation for twice, catalyzed hydrogenation reaction is carried out, and afterwards, the reaction product is distilled twice under reduced pressure to get the target product. The method reduces procedures such as crystallization, centrifugalization, activated carbon adsorption, etc. in prior art and develops a completely new production process, which has the advantages of high product quality, low production cost and improved working environment.
Owner:四川众邦新材料股份有限公司
Preparation method of CsPbBr3 spindle type perovskite micro-particles with adjustable size
ActiveCN111592035AHigh luminous purityLuminous lifetime adjustableLuminescent compositionsLead compoundsHexynePhysical chemistry
The invention discloses a preparation method of CsPbBr3 spindle type perovskite micro-particles with an adjustable size. The preparation method comprises the following steps: firstly, by taking 5-hexynoic acid and 10-undecynoic acid as surface ligands, respectively preparing a 5-hexynoic acid CsPbBr3 perovskite nanocrystal solution and a 10-undecynoic acid CsPbBr3 perovskite nanocrystal solution;respectively mixing the 5-hexynoic acid CsPbBr3 perovskite nanocrystal solution and the 10-undecynoic acid CsPbBr3 perovskite nanocrystal solution according to different volume ratios to obtain mixedsolutions; and finally, reacting the mixed solutions under the irradiation of an ultraviolet lamp to obtain the CsPbBr3 spindle type perovskite micro-particles with different length-width ratios. Thepreparation method is simple, the CsPbBr3 spindle type perovskite micro-particles with higher luminous purity and adjustable luminous life can be prepared at one time; the spindle type perovskite CsPbBr3 micro-particles prepared by the method disclosed by the invention are adjustable in length-width ratio, smaller in band gap, narrow in light-emitting half-peak width, high in color purity, long inlight-emitting service life and excellent in photoelectric property.
Owner:NANJING UNIV OF TECH
Technique for producing 2,5-dimethyl-2,5-hexandiol by extraction and decompression distillation method
ActiveCN101234950BReduce the impact on physical healthImprove securityOrganic compound preparationHydroxy compound preparationHexynePotassium hydroxide
The invention discloses a new technology for producing 2, 5-dimethyl-2, 5-hexanediol using the method of extraction and reduced-pressure distillation. The method adopts acetylene and acetone, which are condensed with potassium hydroxide in benzene solvent and then neutralized with hydrochloric acid to get a feed solution containing hexyne-diol, oil phase and benzene. After condensation and extraction operation for twice, catalyzed hydrogenation reaction is carried out, and afterwards, the reaction product is distilled twice under reduced pressure to get the target product. The method reduces procedures such as crystallization, centrifugalization, activated carbon adsorption, etc. in prior art and develops a completely new production process, which has the advantages of high product quality, low production cost and improved working environment.
Owner:四川众邦新材料股份有限公司
Low-density heat-conducting gel and preparation method thereof
The invention discloses low-density heat-conducting gel and a preparation method thereof, and belongs to the technical field of heat-conducting gel, wherein problems that in the prior art, the densityof heat conduction gel is high, the weight of parts is increased in the application process, and the cost is high are solved. The low-density heat-conducting gel is prepared from the following raw materials: hollow microspheres with silver-plated surfaces and aluminum oxide. The low-density heat-conducting gel is prepared from the following raw materials: a component A: vinyl silicone oil, dimethicone, hollow microspheres with silver-plated surfaces, aluminum oxide and a chloroplatinic acid-isopropanol complex; a component B: vinyl silicone oil, dimethyl silicone oil, side hydrogen-containingsilicone oil, terminal hydrogen-containing silicone oil, hollow microspheres with silver-plated surfaces, aluminum oxide, 3,5-dimethyl-1-hexyne-3-ol and carbon black. The low-density heat-conductinggel disclosed by the invention is good in heat-conducting property, low in density, low in cost and wide in applicability.
Owner:广州布鲁奥申新材料科技有限公司
Preparation method of low-current-noise high-resistance-value dispersive chip resistor slurry
ActiveCN112466585AMeet small size requirementsReduce current noiseNon-conductive material with dispersed conductive materialCable/conductor manufactureHexyneActive agent
The invention discloses a preparation method of low-current-noise high-resistance-value dispersive chip resistor paste, and the method comprises the following steps: respectively carrying out surfacetreatment on a conductive phase, glass powder and an inorganic additive powder material by using a small-molecular alkynediol surfactant, adding the materials into an organic carrier, and preparing the resistor paste with the fineness of less than or equal to 5 [mu]m by using a three-roller machine, wherein the small-molecular alkynediol surfactant is 2, 5-dimethyl-3-hexyne-2, 5-diol, 2, 4, 7, 9-tetramethyl-5-decyne-4, 7-diol or 3, 6-dimethyl-4-octyne-3, 6-diol. According to the invention, a small molecular alkynediol surfactant (relatively low in boiling point, volatile and non-decomposable)is adopted to carry out surface treatment on powder in the chip resistor slurry, so that the powder agglomeration phenomenon is greatly reduced, and the chip resistor prepared from the chip resistor slurry is compact in surface and free of cracking, pinholes and other undesirable phenomena; meanwhile, the outstanding advantages of low current noise and good resistance dispersity are achieved.
Owner:西安宏星电子浆料科技股份有限公司
Process for synthesis of 3-hexyne- 2, 5-diol by slurry bed acidity control method
InactiveCN105061143AHigh selectivityIncrease production rateOrganic compound preparationHydroxy compound preparationHexynePtru catalyst
The invention discloses a process for synthesis of 3-hexyne- 2, 5-diol by slurry bed acidity control method. The process includes: adding an acetaldehyde aqueous solution and a catalyst into a slurry bed reaction kettle and conducting mixing to obtain a slurry solution, then introducing acetylene from the lower part of the slurry solution into a slurry system to carry out reaction, thus obtaining a generated solution; in the reaction process, adding an alkali solution repeatedly to control the pH value of the reaction solution at 5.5-7.0; cooling the generated solution, conducting first standing settlement layering, then conveying the generated solution from standing settlement to a high level settler to carry out second standing settlement layering, and carrying out concentration and separation on the supernatant, and subjecting the remaining concentrated liquid to vacuum distillation, thus obtaining the 3-hexyne- 2, 5-diol. In the synthesis process, the low concentration alkali solution is added to control the pH value of the reaction solution at 5.5-7.0, so that the whole reaction has high selectivity and high production rate, thus being more conducive to industrial production.
Owner:四川隆桥化工集团有限公司
Method for synthesizing, extracting and purifying 3-hexyne-2, 5-diol
PendingCN113666802AEasy to transportEasy to storeOrganic compound preparationHydroxy compound separation/purificationHexynePtru catalyst
A method for synthesizing, extracting and purifying 3-hexyne-2, 5-diol adopts paraldehyde as an acetaldehyde raw material source and potassium hydroxide as a catalyst to react with acetylene of which the pressure is lower than 0.15 MPa in an aliphatic hydrocarbon or aromatic hydrocarbon organic solvent to generate the 3-hexyne-2, 5-diol. The method comprises the following steps: carrying out two-phase separation and hydrolysis on a reaction endpoint material which is a liquid-liquid two-phase system to obtain an aqueous solution containing a target product and potassium hydroxide, extracting by using an alcohol organic solvent with small water solubility, separating the target product from potassium hydroxide, and rectifying to obtain the target product 3-hexyne-2, 5-diol. The synthesis reaction process is carried out under the safe acetylene pressure of 0.15 MPa, the explosion danger is eliminated, and the method has the characteristics of safe production, high product yield, high product purity and the like.
Owner:四川众邦新材料股份有限公司
In-situ compatibilization polyolefin/polystyrene alloy and preparation method and application thereof
The invention discloses an in-situ compatibilization PO / PS (polyolefin / polystyrene) alloy and a preparation method and application thereof. The alloy is prepared by 45-65% of polyolefin, 10-30% of polystyrene, 2-5% of compatibilization initiation mother liquor and 20-25% of processing agent, and the compatibilization initiation mother liquor is obtained by dissolving crosslinking agent and crosslinking agent auxiliaries to oily carrier., wherein the crosslinking agent is at least one of 2, 5-dimethyl-2, 5-di (tertiary butyl peroxide) hexine, 2, 5-dimethyl-2, 5-di (tertiary butyl peroxide) hexane or dicumyl peroxide, and the crosslinking agent auxiliaries is at least one of liquid polybutadiene, prussic acid ester type substances, isocyanate substances or acrylate substances. Compared with the dynamic sulfuration in-situ compatibilization method and the common compatibilization method, the preparation of the in-situ compatibilization PO / PS alloy is capable of effectively reducing production cost of the PO / PS alloy, the PO / PS alloy prepared has lower molding shrinkage rate, higher modulus and excellent overall performance and can be used as substitutes for acrylonitrile butadiene styrene partially.
Owner:KINGFA SCI & TECH CO LTD +2
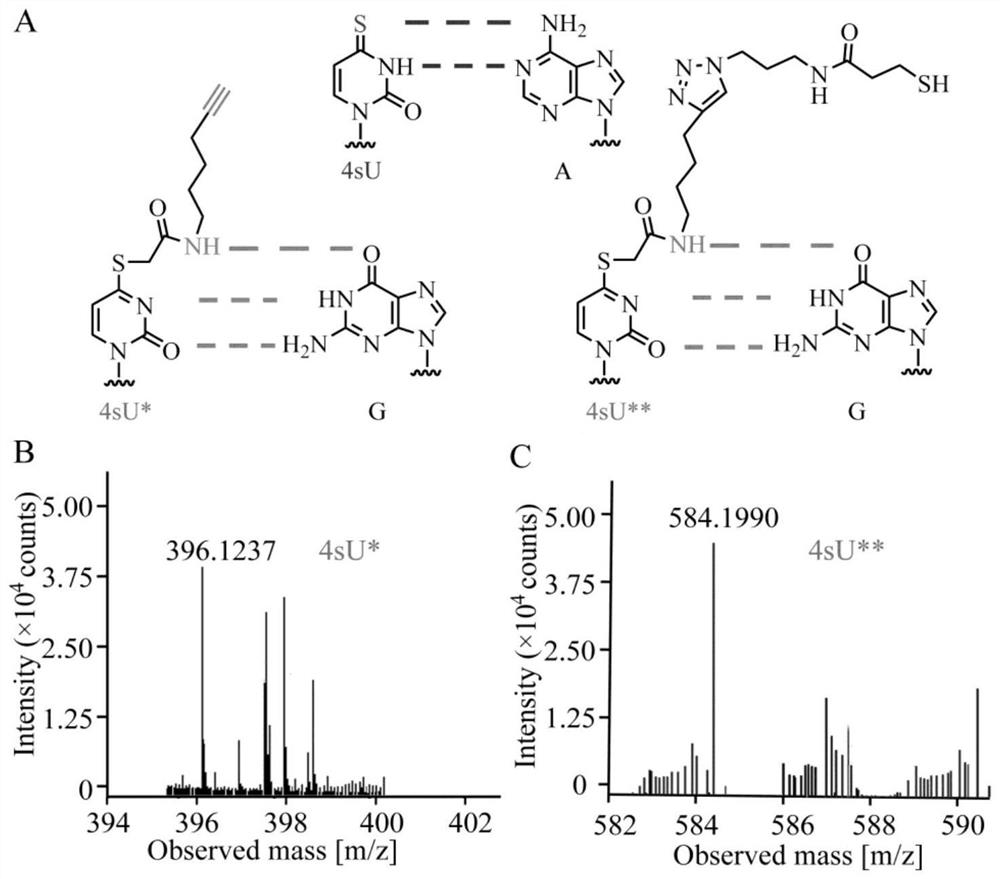
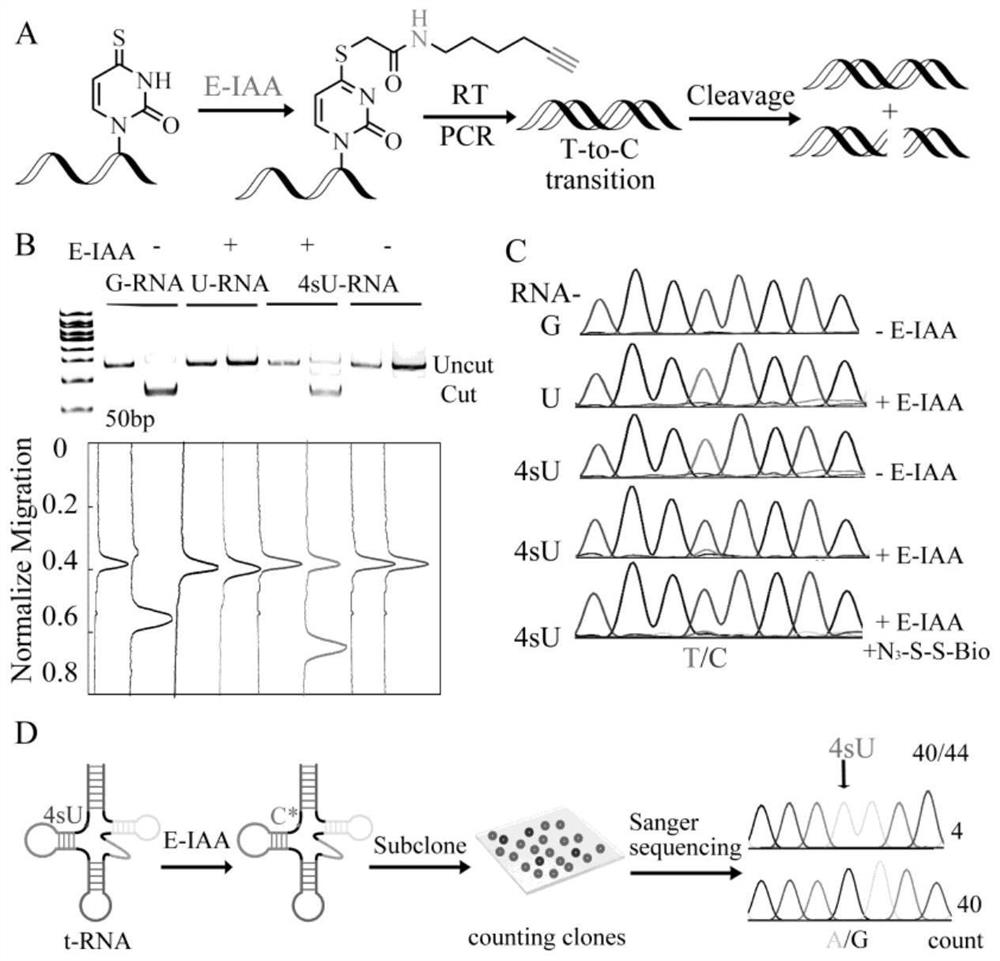
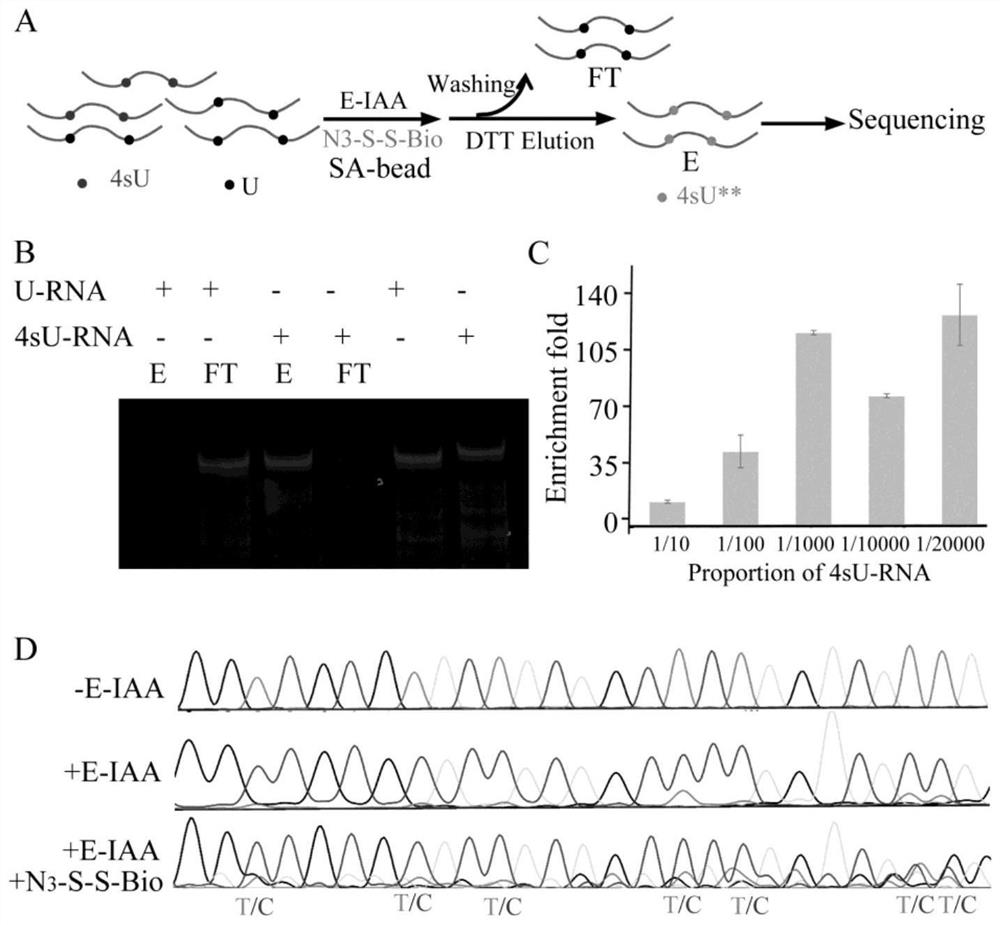
Difunctional marking method of 4-sulfydryl uracil and application of difunctional marking method in sequence enrichment and single base resolution sequencing
PendingCN114350770AAchieve sequence enrichmentAchieve resolutionOrganic chemistryMicrobiological testing/measurementHexyneBiotin
The invention discloses a bifunctional labeling method of 4-sulfydryl uracil and application of the bifunctional labeling method in sequence enrichment and single base resolution sequencing, and belongs to the technical field of nucleic acid chemistry. N-hexyl-5-alkynyl-2-iodoacetamide (E-IAA) is utilized to perform nucleophilic substitution reaction on 4-sulfydryl uracil so as to realize labeling of double functions, and the double functions are reflected as follows: alkynyl at the tail end can be efficiently crosslinked with an azide group with biotin under a mild condition, so that enrichment of a sequence containing 4-sulfydryl uracil is realized; the N-H bond at the near end can change the original base hydrogen bond pairing mode of 4-sulfydryl uracil, and mutation from T to C is generated at the position of 4-sulfydryl uracil, so that single base distinguishing of 4-sulfydryl uracil and nucleic acid derivatives is realized. The bifunctional labeling reaction efficiency is high, and the biological compatibility is good.
Owner:XI AN JIAOTONG UNIV
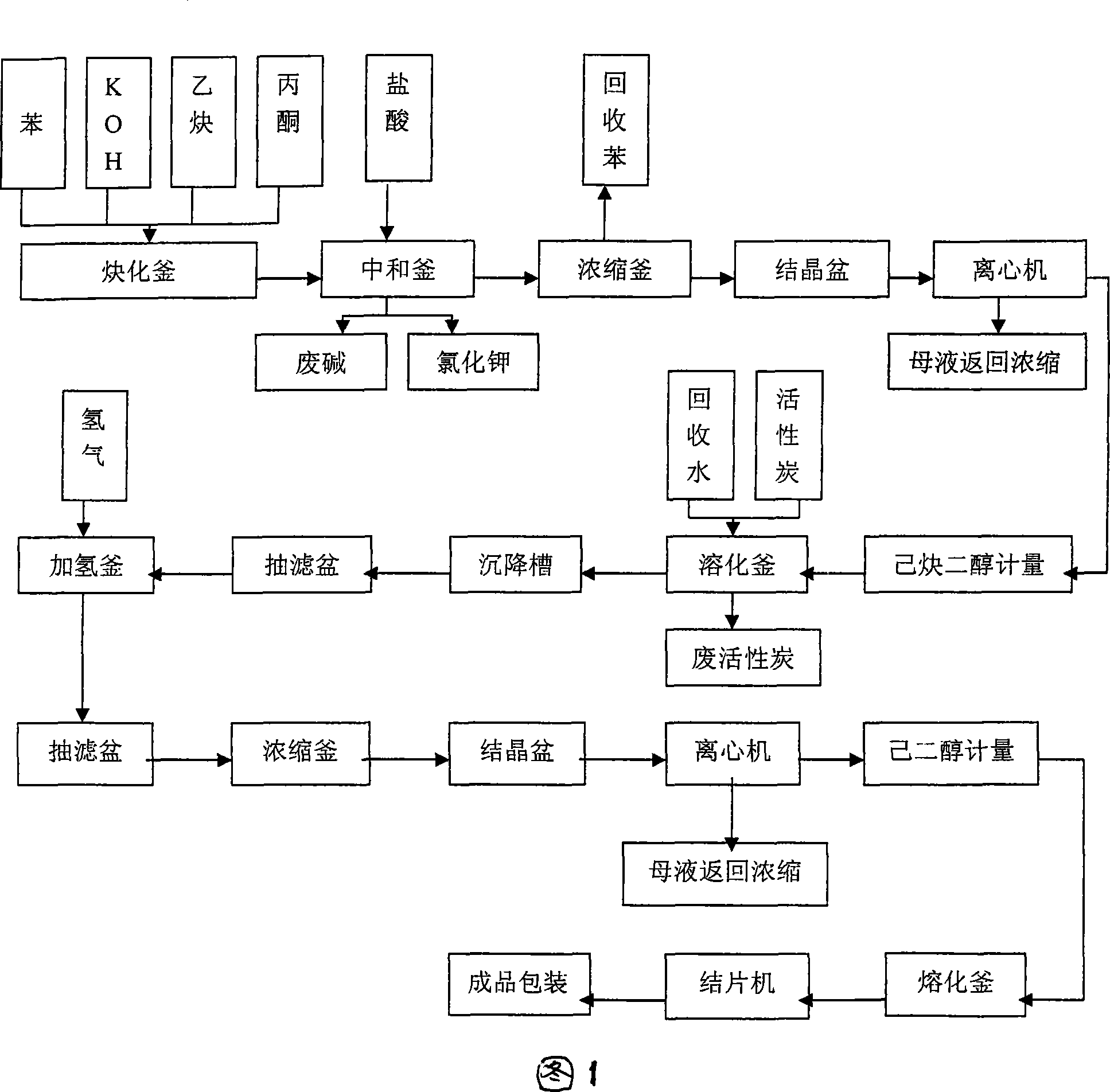
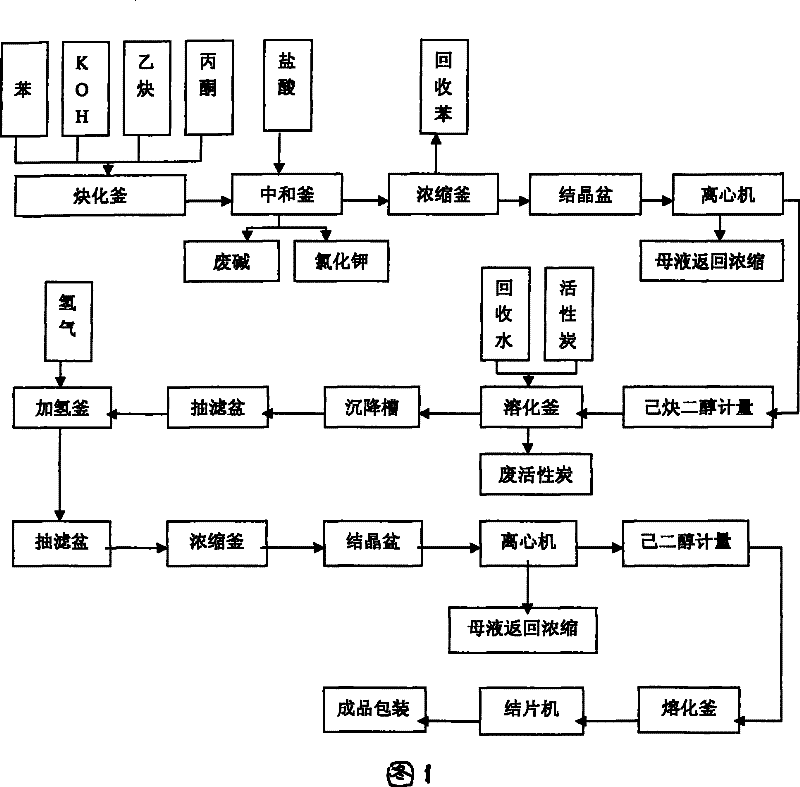
Industrialized production method and device for 2,5-dimethyl-2,5-hexylene glycol
PendingCN109608306ANo pollution in the processHidden negative costOrganic compound preparationPreparation by hydrogenationChemical industryHexyne
The invention discloses an industrialized production method and device for 2,5-dimethyl-2,5-hexylene glycol, and belongs to the technical field of chemical industry production. The production method includes the steps that with benzene being a solvent and potassium hydroxide being a catalyst, acetylene and acetone are used as main raw materials for an ethynylation reaction to prepare hexyne diol,and then a catalytic hydrogenation reaction is conducted, wherein after the ethynylation reaction, in the materials, carbon dioxide is adopted for conducting a neutralizing reaction with potassium hydroxide; potassium carbonate obtained through the neutralizing reaction is recycled. Carbon dioxide is adopted for replacing hydrogen chloride to neutralize potassium hydroxide, the usage amount of carbon dioxide is 35 kilograms for each ton of produced DHAG alcohol, the cost approximates to 80 RMB, but no special requirements for production equipment exist, the equipment is not corroded much moreand does not need to be replaced frequently, potassium carbonate generated after the reaction can be used as side products, implicit negative costs are high, the environment is not polluted, and the effect of environmental protection and economic benefits are improved greatly.
Owner:泸州宏达有机化工厂
Heatproof flexible composite insulating material and preparation method thereof
InactiveCN105679411AImprove heat resistanceHigh heat resistancePlastic/resin/waxes insulatorsQuartz/glass/vitreous enamelsYttriumAdhesive
The invention discloses a heatproof flexible composite insulating material and a preparation method thereof. The material is composed of inorganic ceramic fiber paper and a polyimide film bonded to the inorganic ceramic fiber paper via an adhesive. The adhesive is prepared by adding a filling material, including shell powder and yttrium nitrate, into mixed melting product of a phenolic compound, a bisphenol A dicyanate monomer, epoxy resin E44, pentaerythritol tetraacrylate, a dodecylbenzene sulfonic acid, iso-tridecanol polyoxyethylene ether, an N-Acetyl-1,3-phenylenediamine and butyl acetyne. The price of the heatproof flexible composite insulating material is reasonable is reasonable, so that the material can be applied to insulation systems of special motors, electrical appliances electronic transformers whose heatproof level is higher than H level (180 DEG C) in a more economical manner.
Owner:SUZHOU JIN TENG ELECTRONICS TECH
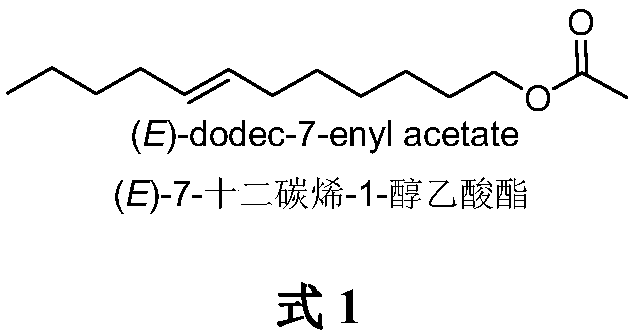
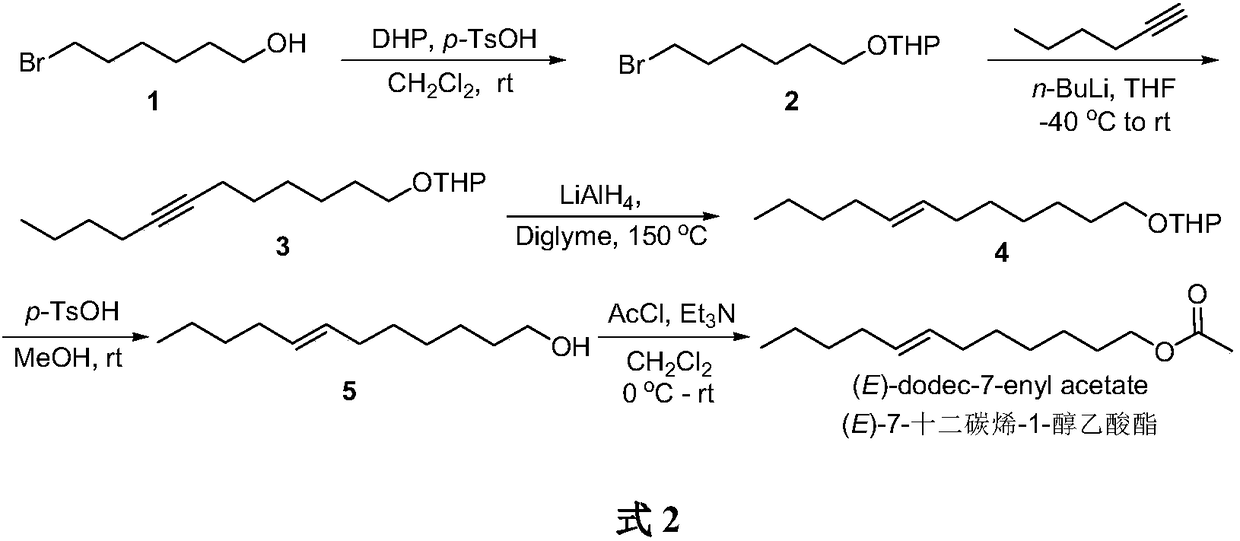
Method for synthesizing (E)-7-dodecen-1-ol acetate
InactiveCN109020937APreparation from carboxylic acid halidesOrganic chemistry methodsHexyneP-Toluenesulfonic acid
The invention belongs to the technical field of insect pheromone synthesis, and discloses a novel method for synthesizing (E)-7-dodecen-1-ol acetate. The method comprises the following steps: reactinga starting raw material 6-bromo-1-hexanol with 2,3-dihydropyran to obtain 1-tetrahydropyranyloxy-6-bromohexane, and carrying out a coupling reaction on the 1-tetrahydropyranyloxy-6-bromohexane and 1-hexyne in the presence of n-butyllithium to generate 1-tetrahydropyranyloxy-7-dodecyne; reducing the 1-tetrahydropyranyloxy-7-dodecyne by lithium aluminum hydroxide in diethylene glycol dimethyl etherto obtain (E)-tetrahydropyranyloxy-7-dodecene; and removing tetrahydropyran protecting groups by using p-toluenesulfonic acid to synthesize (E)-7-dodecen-1-ol acetate, and finally reacting the (E)-7-dodecen-1-ol acetate with acetyl chloride to obtain the target product (E)-7-dodecen-1-ol acetate. The method has the advantages of simple synthesis route, mild reaction conditions, and realization ofthe total yield reaching 49%.
Owner:CHINA AGRI UNIV
Semi-Bright Nickel Plating Bath and Method of Using Same
A nickel plating bath for plating a semi-bright nickel deposit on a substrate comprising a) nickel sulfateions; b) a soluble salt of chloroacefic acid, acetic acid, glycolic acid, proprionic acid, benzoic acid, salicylic acid or chlorobenzoic acid; and c) at least one diol selected from the group consisting of hexyne diol, butyne diol and combinations of the foregoing. The semi-bright nickel plating bath described herein produces sulfur-free semi-bright deposits over a very wide current density range. The plating bath described herein is at least substantially free of coumarin and produces desirable leveling characteristics. The bath also requires no aldehydes to achieve simultaneous thickness and electrolytic potential (STEP) and has extremely low stress and excellent ductility.
Owner:MACDERMID ACUMEN INC
Preparation method of chilo suppressalis sex pheromone cis-11-hexadecenal
InactiveCN110922311AEasy to operateMild reaction conditionsOrganic compound preparationOrganic chemistry methodsHexyneP-Toluenesulfonic acid
The invention discloses a preparation method of chilo suppressalis sex pheromone cis-11-hexadecenal. The preparation method comprises the steps of (1) allowing 10-bromine-1-decanol and dihydropyran react to generate 10-bromine-1-decanol tetrahydropyran in a dichloromethane solution; (2) allowing the 10-bromine-1-decanol tetrahydropyrane obtained in the step (1) and 1-hexyne and n-butyllithium to react in a tetrahydrofuran solvent under the protection of nitrogen to generate 11-hexadecene tetrahydropyrane, and reacting the 11-hexadecene tetrahydropyrane with 1-hexyne and n-butyllithium to generate 11-hexadecene tetrahydropyrane; (3) carrying out a reduction reaction on the 11-hexadecene tetrahydropyran obtained in the step (2) and nickel chloride and sodium borohydride to generate cis-11-hexadecene tetrahydropyran in a methanol solution; (4) in methanol, enabling the cis-11-hexadecene tetrahydropyran obtained in the step (3) to react with p-toluenesulfonic acid to generate cis-11-hexadecenol; and (5) in dichloromethane, carrying out an oxidation reaction on the cis-11-hexadecenol obtained in the step (4) and pyridinium chlorochromate to generate the cis-11-hexadecenal. The method issimple to operate and mild in reaction condition. Medicines and reagents used in the reaction are conventional products, are low in price and are easy to obtain, and the reaction cost is reduced.
Owner:JILIN INST OF CHEM TECH
Method for co-production of dimethyl decynediol and methyl hexynol
PendingCN113816835ALow viscosityTake advantage ofOrganic compound preparationHydroxy compound separation/purificationHexynePtru catalyst
The invention relates to a method for co-production of dimethyl decynediol (4, 7-dimethyl-5-decyne-4, 7-diol) and methyl hexynol (3-methyl-1-hexyne-3-alcohol), which comprises the following steps: by taking 2-pentanone and acetylene as raw materials, potassium hydroxide or potassium isobutoxide or potassium tert-butoxide as a catalyst and an organic solvent as a dispersing agent, reacting 2-pentanone with normal-pressure or low-pressure acetylene at a relatively low temperature to generate a methyl hexynol-catalyst complex, mixing the methyl hexynol-catalyst complex with 2-pentanone, and quickly reacting through a high-temperature reaction tube to generate the dimethyl decynediol-catalyst complex; hydrolyzing the reaction product to remove potassium hydroxide, and fractionating to collect each part. The method has the characteristics of low reaction temperature, short reaction time, less side reaction, less catalyst consumption and the like.
Owner:四川众邦新材料股份有限公司
Cable material with strong stability as well as preparation method thereof
InactiveCN104312066AUniform plasticizationIncreased plasticization ratePlastic/resin/waxes insulatorsHexyneAcrylic rubber
The invention discloses a cable material with strong stability. The cable material with strong stability is prepared from the following raw materials in parts by weight: 3-5 parts of diethyl ethylphosphonate, 6-8 parts of benzo-hydroperoxide tert-butyl ester, 15-25 parts of N550 carbon black, 12-18 parts of chlorinated polyethylene, 3-4 parts of potassium titanate whiskers, 2-4 parts of dodecanamide, 2-5 parts of ammonium polyphosphate, 3-5 parts of m-phenylene dibenzoate, 4-7 parts of a rare earth stabilizer, 4-8 parts of polyimide, 5-6 parts of expansible graphite, 2-4 parts of 2, 5-bi(tert-butyl peroxide)-2, 5-dimethyl hexyne, 3-4 parts of zinc isoocatanoate, 45-65 parts of acrylic rubber and 8-12 parts of auxiliaries. According to the cable material disclosed by the invention, by adding raw materials such as the rare earth stabilizer, the cable material is uniformly plastified, so that the plastifying rate is improved. Meanwhile, the extruding and processing manufacturability is excellent and the product quality stability of the cable material is improved.
Owner:安徽电信器材贸易工业有限责任公司
An LED encapsulating material, a preparing method thereof and application of the material
InactiveCN106046380AEfficient packagingImprove transmittanceSemiconductor devicesChemical structurePlatinum
The invention relates to an LED encapsulating material. The material is prepared from a vinyl compound A, a cyclohexyl methylsiloxane-carbonyl-platinum composite, 3,5-dimethyl-1-hexyn-3-ol, a hydride B and phenyl vinyl methylsilane. The chemical structural formula of the vinyl compound A is shown in the specification. The chemical structural formula of the hydride B is shown in the specification. The chemical structural formula of the material is shown in the specification, wherein x, y, u and v are not more than 2000 and not less than 0, Me represents methyl CH<3>- and Ph represents phenyl C<6>H<5>-. Compared with the prior art, the material has advantages of excellent transmissivity and refractive indexes in each waveband, excellent heat resistance, low optical performance degradation, good fluidity before heating and curing, convenience for effective encapsulating for light emitting diodes, a simple preparing method, and simple, convenient and efficient applications.
Owner:NINGBO XIAYUAN TECH
Medically efficient, nontoxic and non-side-effect organic silicon material for dental restoration
ActiveCN103271831AModerate vulcanization speedConvenience impressionImpression capsDental impression compositionsSide effectVulcanization
The invention discloses a medically efficient, nontoxic and non-side-effect organic silicon material for dental restoration. The organic silicon material is formed by mixing a component A with a component B according to the weight ratio being 1:1, wherein the component A comprises: 67 to 72 parts of first vinyl silicone oil, 217 to 229 parts of quartz powder, 70 to 75 parts of cyclohexanol alkynol, 33 to 36 parts of 50% of white carbon black and 50% of second vinyl silicone oil, 0.31 to 0.33 part of platinum catalyst and 0.78 to 0.82 part of PE (Poly Ethylene) mould releasing agent; and the component B comprises: 24 to 26 parts of the first vinyl silicone oil, 208 to 220 parts of the quartz powder; 69 to 73 parts of 50% of the white carbon black and 50% of the second vinyl silicone oil, 26 to 28 parts of silicone oil containing hydrogen, 15 to 17 parts of 2-bromine-4-cyano-6-methoxy phenyl ethyl ester, 0.02 to 0.03 part of chain-extended silicone oil containing the hydrogen and 36 to 39 parts of PE mould releasing agent. The organic silicon material is proper in vulcanization speed and convenient for impression.
Owner:SHANGHAI XINGDU NEW MATERIAL TECH CO LTD
Ligands for integrin αVβ8, synthesis and uses thereof
ActiveUS11332498B2High affinityHigh selectivityPeptide/protein ingredientsGeneral/multifunctional contrast agentsHexyneThreonine
Disclosed are compounds represented by the following general formula (I): Cyclo-(Arg-Gly-Asp-X1-X2-X3-X4-X5) (I) wherein the variables groups X1 to X5 have the following meanings X1: Leu, Ile, Nle, Val, Tyr, Phe; X2: D-amino acid such as D-Pro, N-Me-D-Phe; X3: Pro, N-Me-amino acid such as N-Me-Lys, N-Me-Lys(Ac); Pro-Rx3, N-Me-Lys-Rx4; X4: Gly, Ala, Ser, Thr; X5: Leu, Ala, Tyr, His, Ile, Nle, Val, Phe wherein Pro-Rx3 represents a proline residue that carries at the C-3, C-4 or C-5 carbon atom and preferably the C-4 carbon atom a functional group selected from —NH2, —OH, —NH—Ac, —NH-hexyne, and wherein Lys-Rx4 represents a lysine residue, wherein the ω-amino nitrogen atom carries a group of the formula -L4-R4, wherein L4 is selected from the group consisting of covalent bond, —C(O)—, and —C(O)—O—, . . . , and wherein R4 is selected from the group consisting of —(CH2)n-C≡CH with n=0, 1, 2, 3, 4, 5, 6, 7 or 8, or wherein the sub-sequence -X2-X3- represents a β-turn mimetic diffeωωring from the meanings above, or pharmaceutically acceptable salts, esters, solvates, polymorphs or modified forms thereof represented by the following general formula (II): (X0)n1L(X8)n2 wherein X0 represents the compound of the general formula (I) as specified above (excluding one hydrogen atom to allow bonding to the linker), L represents a linker, X8 represents the effector moiety and wherein n1 and n2 are each independently selected from the range of 1 to 5, preferably such that each of n1 and n2 represents 1, wherein n1+n2 represents the number of valencies of the linker and is preferably in the range of from 2 to 6, more preferably 2-5, with the proviso that each of n1 and n2 is at least 1, as well as uses therefore in therapy and imaging.
Owner:TECH UNIV MUNCHEN
Method for synthesizing sex pheromone of anarsia lineatella
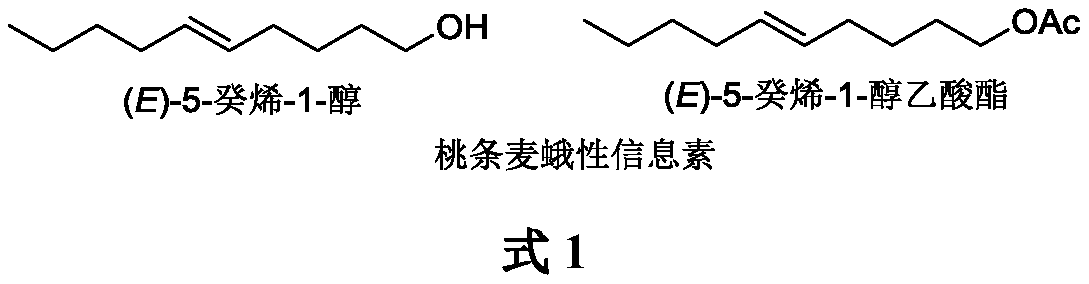
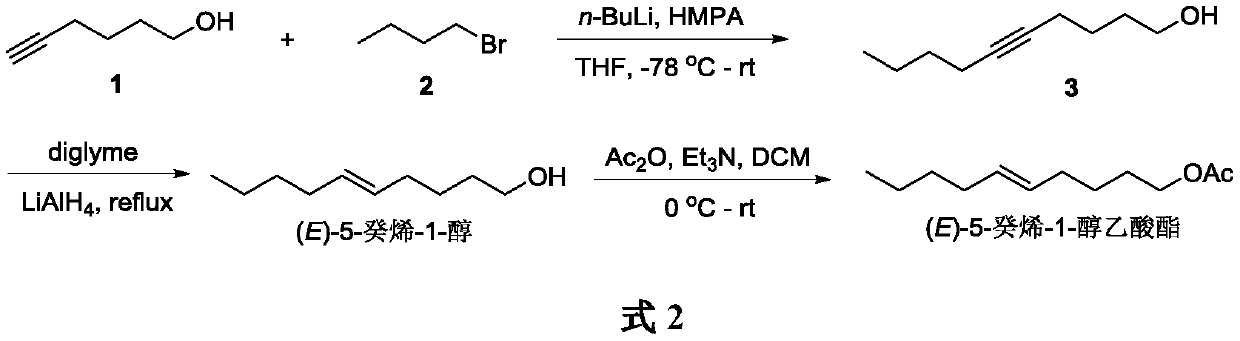
InactiveCN111116360AOrganic compound preparationPreparation by hydrogenationHexyneAnarsia lineatella
The invention belongs to the technical field of green pesticide synthesis, and discloses a novel method for synthesizing sex pheromone of anarsia lineatella. The method comprises the following steps:with 5-hexyn-1-ol as a raw material, subjecting 5-hexyn-1-ol and1-bromobutane to a nucleophilic substitution reaction in the presence of n-butyllithium and HMPA to generate 5-decyn-1-ol, then performing reduction via lithium aluminum hydride to obtain (E)-5-decen-1-ol, and finally performing an acetylation reaction to obtain (E)-5-decen-1-ol acetate. According to the invention, a triple bond of alkyne is reduced into an E-type double bond by lithium aluminum hydride, so the method has the advantages of simple synthetic route, mild reaction conditions, good environmental compatibility and the like.
Owner:CHINA AGRI UNIV